

Acquisition of Terns Pharmaceuticals Merck & Co., Inc., Rahway, N.J., USA March 25, 2026

Disclaimer Forward-looking statements This presentation of Merck & Co., Inc., Rahway, N.J., USA (the “company”) contains forward-looking statements, including, but not limited to, statements related to the potential benefits of and future plans for TERN-701; the ability of the company and Terns Pharmaceuticals, Inc. ( Terns ) to complete the transactions contemplated by the transaction agreement, including the parties' ability to satisfy the conditions to the consummation of the transaction contemplated thereby, statements about the expected timetable for completing the transaction, the company's and Terns' beliefs and expectations and statements about the benefits sought to be achieved in the company's proposed acquisition of Terns, the potential effects of the acquisition on both the company and Terns, and the possibility of any termination of the transaction agreement, as well as the expected benefits and success of the company's product candidates. Risks and uncertainties include but are not limited to, unanticipated delays in or negative results from Terns' clinical studies and other risks related to clinical development, delays in or unanticipated action by regulatory authorities, risks related to government contracts, having to use cash in ways other than as expected and other risks, uncertainties associated with Terns' business in general; the risk that competing offers or acquisition proposals will be made; the possibility that various conditions to the consummation of the proposed transaction contained in the transaction agreement may not be satisfied or waived (including, but not limited to, the failure to obtain a sufficient number of tendered shares from Terns' shareholders); the effects of disruption from the transactions contemplated by the transaction agreement and the impact of the announcement and pendency of the transactions on Terns' business; the risk that shareholder litigation in connection with the transaction may result in significant costs of defense, indemnification and liability; general industry conditions and competition; general economic factors, including interest rate and currency exchange rate fluctuations; the impact of pharmaceutical industry regulation and health care legislation in the United States and internationally; global trends toward health care cost containment; technological advances, new products and patents attained by competitors; challenges inherent in new product development, including obtaining regulatory approval; the company’s ability to accurately predict future market conditions; manufacturing difficulties or delays; financial instability of international economies and sovereign risk; dependence on the effectiveness of the company’s patents and other protections for innovative products; and the exposure to litigation, including patent litigation, and/or regulatory actions. The company undertakes no obligation to publicly update any forward-looking statement, whether as a result of new information, future events or otherwise, except to the extent required by law. Additional factors that could cause results to differ materially from those described in the forward-looking statements can be found in the company’s Annual Report on Form 10-K for the year ended December 31, 2025, Terns’ Quarterly Report on Form 10-Q for the quarter ended September 30, 2025 and Annual Report on Form 10-K for the year ended December 31, 2024, and other filings with the Securities and Exchange Commission (SEC) available at the SEC’s Internet site (www.sec.gov). Additional information regarding the proposed transaction This presentation is for informational purposes only and is neither an offer to purchase nor a solicitation of an offer to sell any securities of Terns. After the planned tender offer is commenced, investors and security holders are urged to read carefully the tender offer statement on Schedule TO (including an Offer to Purchase, a related letter of transmittal and other tender offer documents) to be filed by Merck and the solicitation/recommendation statement on Schedule 14D-9 to be filed by Terns, as may be amended from time to time, which may be obtained at www.sec.gov. 2

Strategic Rationale Rob Davis Chairman & Chief Executive Officer Scientific Overview Dr. Dean Li President, Merck Research Laboratories Ag Ag Agen en end d da a a Commercial Opportunity Jannie Oosthuizen EVP & President, Oncology and MSD International Financial Overview Caroline Litchfield Chief Financial Officer 3

Strategic Rationale Rob Davis Chairman & Chief Executive Officer

Advancing science-led strategy and building upon growing hematology pipeline with acquisition of Terns Pharmaceuticals Science-driven business development that strengthens and complements hematology pipeline TERN-701 an investigational next-generation, allosteric TKI for treatment of certain patients with chronic myeloid leukemia Clinical differentiation with potential best-in-class profile driven by high selectivity and improved therapeutic index $53 cash per share, representing total equity value of approximately $6.7B, or $5.7B net of cash acquired; Multibillion potential commercial opportunity and driver of expected to close in 2Q 2026 growth in the next decade, creating long-term shareholder value 5 5

Scientific Overview Dr. Dean Li President, Merck Research Laboratories
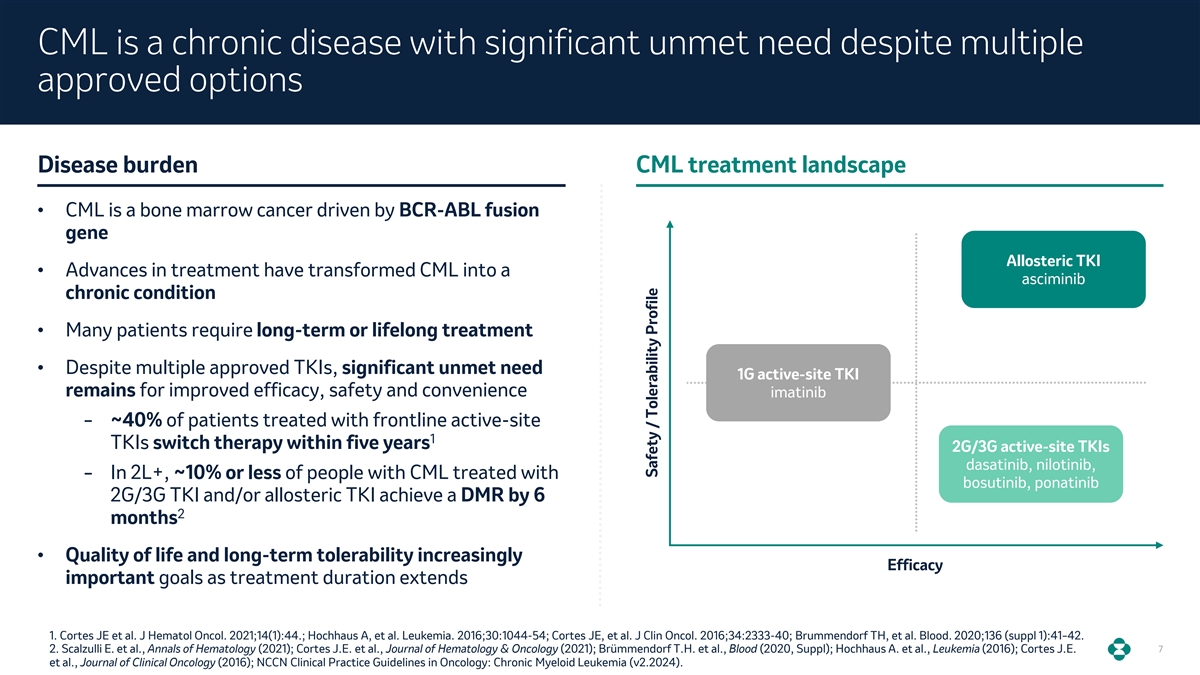
CML is a chronic disease with significant unmet need despite multiple approved options Disease burden CML treatment landscape • CML is a bone marrow cancer driven by BCR-ABL fusion gene Allosteric TKI • Advances in treatment have transformed CML into a asciminib chronic condition • Many patients require long-term or lifelong treatment • Despite multiple approved TKIs, significant unmet need 1G active-site TKI remains for improved efficacy, safety and convenience imatinib ‒ ~40% of patients treated with frontline active-site 1 TKIs switch therapy within five years 2G/3G active-site TKIs dasatinib, nilotinib, ‒ In 2L+, ~10% or less of people with CML treated with bosutinib, ponatinib 2G/3G TKI and/or allosteric TKI achieve a DMR by 6 2 months • Quality of life and long-term tolerability increasingly Efficacy important goals as treatment duration extends 1. Cortes JE et al. J Hematol Oncol. 2021;14(1):44.; Hochhaus A, et al. Leukemia. 2016;30:1044-54; Cortes JE, et al. J Clin Oncol. 2016;34:2333-40; Brummendorf TH, et al. Blood. 2020;136 (suppl 1):41–42. 2. Scalzulli E. et al., Annals of Hematology (2021); Cortes J.E. et al., Journal of Hematology & Oncology (2021); Brümmendorf T.H. et al., Blood (2020, Suppl); Hochhaus A. et al., Leukemia (2016); Cortes J.E. 7 et al., Journal of Clinical Oncology (2016); NCCN Clinical Practice Guidelines in Oncology: Chronic Myeloid Leukemia (v2.2024). Safety / Tolerability Profile
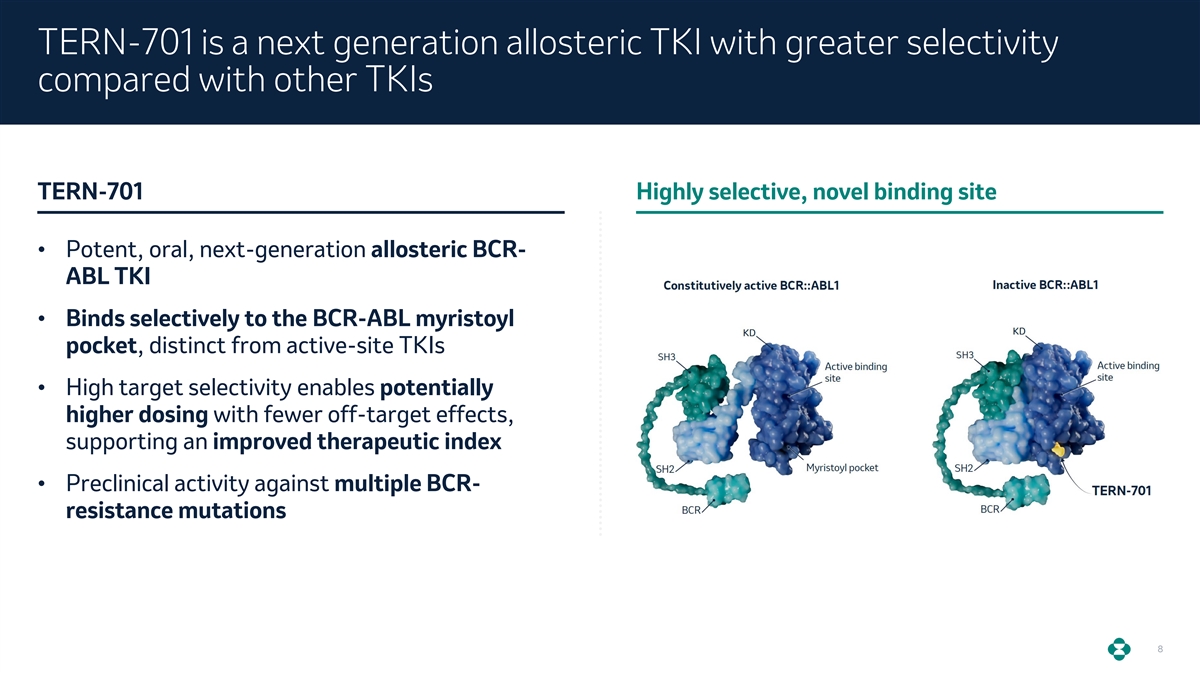
TERN-701 is a next generation allosteric TKI with greater selectivity compared with other TKIs TERN-701 Highly selective, novel binding site • Potent, oral, next-generation allosteric BCR- ABL TKI • Binds selectively to the BCR-ABL myristoyl pocket, distinct from active-site TKIs • High target selectivity enables potentially higher dosing with fewer off-target effects, supporting an improved therapeutic index • Preclinical activity against multiple BCR- resistance mutations 8

Phase 1/2 CARDINAL study demonstrates encouraging results in certain patients with CML 1 TERN-701 Phase 1 : MMR and DMR Merck evaluated data on intent-to-treat at 24 achievement by 24 weeks at doses ≥ 320mg weeks at doses ≥ 320mg 3 • Robust MMR and DMR observed • Best-in-class potential efficacy − MMR 75% (53.3% to 90.2%) − MMR potential of ~2x that of approved TKIs including asciminib 2 − DMR 36% (18.6% to 55.9%) − DMR potential of ~2-3x that of approved − Heavily pretreated population with a TKIs including asciminib high level of disease burden − Unprecedented speed to achieve MMR & • Clinical response in prior asciminib DMR treatment failures • Clinical response in prior asciminib treatment • Observed favorable safety and tolerability failures profile based on data available to date • Observed favorable safety and tolerability profile based on data available to date 1. Jabbour E., et al. Oral presentation at 67th ASH Annual Meeting and Exposition; December 6‒9, 2025; Presentation #901 2. Included patients with baseline BCR::ABLIS >0.01% achieving MR4, BCR::ABL1IS ≤0.01%; MR4.5, BCR::ABL1IS ≤0.0032%; and MR5, BCR::ABL1IS ≤0.001 3. Cross-trial comparison 9
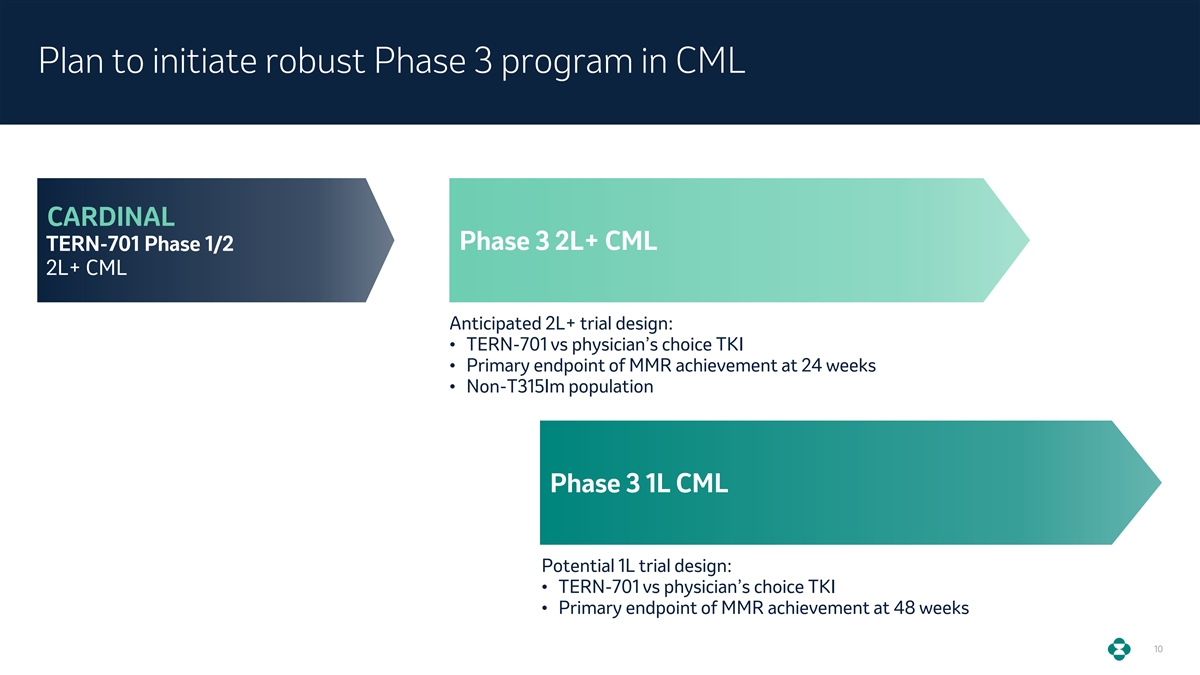
Plan to initiate robust Phase 3 program in CML CARDINAL Phase 3 2L+ CML TERN-701 Phase 1/2 2L+ CML Anticipated 2L+ trial design: • TERN-701 vs physician’s choice TKI • Primary endpoint of MMR achievement at 24 weeks • Non-T315Im population Phase 3 1L CML Potential 1L trial design: • TERN-701 vs physician’s choice TKI • Primary endpoint of MMR achievement at 48 weeks 10
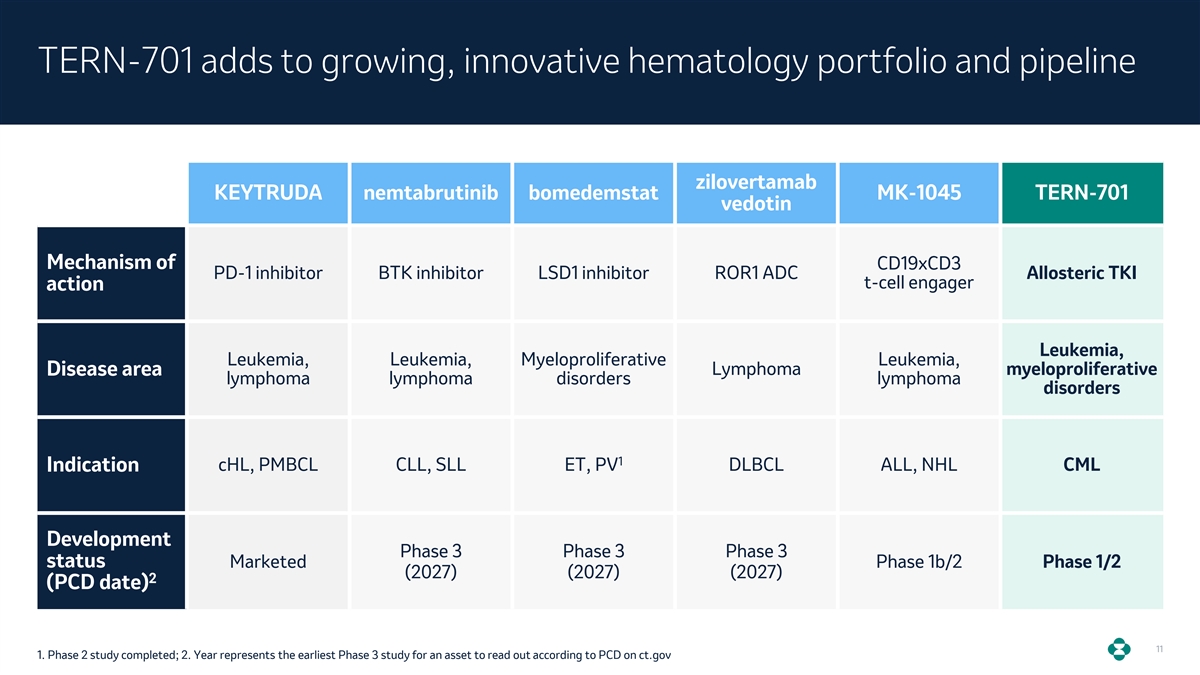
TERN-701 adds to growing, innovative hematology portfolio and pipeline zilovertamab KEYTRUDA nemtabrutinib bomedemstat MK-1045 TERN-701 vedotin Mechanism of CD19xCD3 PD-1 inhibitor BTK inhibitor LSD1 inhibitor ROR1 ADC Allosteric TKI t-cell engager action Leukemia, Leukemia, Leukemia, Myeloproliferative Leukemia, Lymphoma myeloproliferative Disease area lymphoma lymphoma disorders lymphoma disorders 1 Indication cHL, PMBCL CLL, SLL ET, PV DLBCL ALL, NHL CML Development Phase 3 Phase 3 Phase 3 status Marketed Phase 1b/2 Phase 1/2 (2027) (2027) (2027) 2 (PCD date) 11 1. Phase 2 study completed; 2. Year represents the earliest Phase 3 study for an asset to read out according to PCD on ct.gov

Commercial Opportunity Jannie Oosthuizen EVP & President, Oncology and MSD International

CML is a chronic disease with increasing prevalence and high remaining unmet need Treatment landscape • CML has become a chronic disease that can require life-long treatment • As patients live longer, quality of life and tolerability have become important treatment goals ~18K • Prevalence expected to increase over time with New patients diagnosed annually aging population 1 with CML • Duration of treatment expected to increase driven by higher allosteric TKI use 13 1. CancerMPact®, Oracle Life Sciences. CML incidence in US, EU5, JP in 2025

TERN-701 represents a potentially meaningful and durable commercial opportunity Potential best-in-class, next-generation allosteric TKI Strategic fit within growing hematology portfolio and pipeline Multibillion non-risk adjusted Significant remaining unmet need in CML peak commercial opportunity by mid-2030s Compelling commercial opportunity in a serious, chronic disease with long duration of therapy Patent exclusivity in the U.S. anticipated to extend into 2040s 14

Financial Overview Caroline Litchfield Chief Financial Officer

To be updated Financial overview of Terns Pharmaceuticals acquisition • Merck has agreed to acquire all outstanding shares of Terns Pharmaceuticals for a purchase price of $53.00 per share • Total equity value of ~$6.7 billion, or ~$5.7 billion net of acquired cash, cash equivalents, and marketable securities Transaction Details • Expected to finance the transaction primarily through new debt issuance • Expected to close in 2Q 2026, subject to the tender of a majority of Terns' outstanding shares and regulatory approvals • Potential value creating transaction delivering growth early in the next decade • Expected to be accounted for as an asset acquisition, resulting in a charge to 2026 research and development expense of ~$5.8 billion or ~$2.35 per share, included in GAAP and non-GAAP results Financial Impact • In addition, expected to negatively impact non-GAAP EPS by ~$0.17 in first 12 months, representing costs associated with advancing TERN-701 and assumed cost of financing • No impact to credit rating expected • Retain significant capacity within strong investment-grade credit rating to pursue additional business Capital Allocation development Priorities • Remain committed to funding and growing dividend over time Unchanged • Continue to expect ~$3 billion of share repurchases during 2026, as previously communicated 16

Q&A Rob Davis Dr. Dean Li Chairman & Chief President, Executive Officer Merck Research Laboratories Jannie Oosthuizen Caroline Litchfield EVP & President, Chief Financial Officer Oncology and MSD International Dr. Marjorie Green Peter Dannenbaum SVP, Head of Global SVP, Investor Oncology Clinical Relations Development

Appendix 18

Acronyms ALL = Acute lymphocytic leukemia MMR = Major molecular response ATP = Adenosine Triphosphate MR = Molecular response BCR = Breakpoint Cluster Region NHL = Non-Hodgkin Lymphoma cHL = Classic Hodgkin Lymphoma PMBCL = Primary mediastinal B-cell lymphoma CLL = Chronic lymphocytic leukemia PV = Polycythemia vera CML = Chronic myeloid leukemia SH2 = Src-homology 2 domain DLBCL = Diffuse large B-cell lymphoma SH3 = Src- homology 3 domain DMR = Deep molecular response SLL = Small lymphocytic lymphoma ET = Essential thrombocythemia TKI = Tyrosine kinase inhibitor KD = Kinase Domain 19