
NON-EXCLUSIVE LICENSE AGREEMENT THIS NON-EXCLUSIVE LICENSE AGREEMENT (this “Agreement”) is entered into effective as of October ___ ___, 2025 (the “Effective Date”) by and between Lisata Therapeutics, Inc., a Delaware corporation (“Lisata”) and Redwood Bioscience, Inc., a Delaware corporation (“Catalent”). Lisata and Catalent are referred to in this Agreement individually as a “Party” and collectively as the “Parties.” Capitalized terms used in this Agreement have the meanings assigned to them in Section 1 below or where otherwise defined in this Agreement. WHEREAS, Lisata is a biotechnology company and has developed Certepetide and owns the Licensed Technology; WHEREAS, Catalent is a biotechnology company and has developed proprietary technology platforms related to the identification, assembly and generation of bioconjugate therapeutic products; and WHEREAS, subject to the terms and conditions of this Agreement, Lisata wishes to grant Catalent, and Catalent wishes to obtain from Lisata, a non-exclusive license under the Licensed Technology for the Field in the Territory. NOW THEREFORE, in consideration of the foregoing premises and the mutual covenants set forth below, and for other good and valuable consideration, the receipt and sufficiency of which are hereby acknowledged, the Parties agree as follows: 1. DEFINITIONS The following definitions will control the construction of each of the following items wherever they appear in this Agreement. 1.1 “Act” means the United States Federal Food, Drug, and Cosmetic Act, as amended to date and as may be further amended from time to time during the Term, and the regulations promulgated with respect thereto. 1.2 “Affiliate” means: (a) with respect to Lisata or any Third Party, any corporation, firm, partnership or other entity that controls, is controlled by or is under common control with such entity; and (b) with respect to Catalent, Redwood Bioscience, Inc. and any corporation, firm, partnership or other entity controlled by it. For the purposes of this definition, “control” means possession, directly or indirectly, of the power to direct the management and policies of such corporation, firm, partnership or other entity, whether through the ownership of 50% or more of the voting interests, through contract, or otherwise. 1.3 “Applicable Laws” means all laws, rules, regulations and guidelines within the Territory (including, the Act and all regulations promulgated thereunder), as existing as of the applicable time, in each case to the extent applicable and relevant to a Party or its performance under this Agreement. 1.4 “Business Day” means any day other than a Saturday, Sunday or other day on which banks in New York, New York are permitted or required to close by law or regulation. 1.5 “Catalent Business Sale” means: (a) any Change of Control of Catalent or (b) any transaction (other than a Licensed Product Sale) pursuant to which Catalent or any of its Affiliates assigns or otherwise transfers to a Third Party its rights to any assets of Catalent and/or Catalent Emeryville. 1.6 “Catalent Emeryville” means the business conducted by Catalent and/or its Affiliates at their Emeryville, California facility. Exhibit 10.19

2 1.7 “Catalent-Introduced Partner” means any Sublicensee or other Party to a Licensed Product Transaction that is not a Lisata-Introduced Partner. 1.8 “Catalent Net Sales” means, solely with respect to any Licensed Product that is commercialized by Catalent or any of its Affiliates (each, a “Catalent Selling Party”) (as opposed to being commercialized by a Sublicensee), the gross amounts invoiced for sales or other dispositions of such Licensed Product by or on behalf of such Catalent Selling Party to Third Parties, less the following deductions to the extent actually incurred, allowed, paid or accrued by the Catalent Selling Party, as determined in each case in compliance with generally accepted accounting principles in the United States (“GAAP”): (a) normal and customary trade, quantity, cash, or other discounts actually allowed and properly taken directly with respect to sales of such Licensed Product; (b) credits or allowances given or made for rejection or return of previously sold Licensed Products or for retroactive price reductions and billing errors; (c) rebates and chargeback payments granted to managed health care organizations, pharmacy benefit managers (or equivalents thereof), national, state/provincial, local, and other governments, their agencies and purchasers and reimbursers, or to trade customers; (d) fees paid to wholesalers, distributors, group purchasing organizations and Third Party payors, in each case with respect to Licensed Products; (e) costs of freight, carrier insurance, and other transportation charges directly related to the distribution of such Licensed Product; (f) that portion of the annual fee on branded prescription drug manufacturers imposed by the Patient Protection and Affordable Care Act, Pub. L. No. 111-148 (as amended), or similar sales-based taxes adopted in the future, in each case to the extent reasonably allocable to sales of such Licensed Product; (g) taxes, duties, or other governmental charges (including any tax such as a value added or similar tax, other than any taxes based on income) directly levied on or measured by the billing amount for such Licensed Product, as adjusted for rebates and refunds, reimbursements and credits; (h) any invoiced amounts that are written off by a Catalent Selling Party as uncollectable amounts; and (i) any other deductions taken by a Catalent Selling Party in calculating net sales in the ordinary course of its business, consistent with GAAP. In no event shall any particular amount identified above be deducted more than once in calculating Catalent Net Sales (i.e., no “double counting” of deductions). Sales of a Licensed Product between Catalent and its Affiliates for resale will be excluded from the computation of Catalent Net Sales, but the subsequent resale of such Licensed Product to a Third Party will be included within the computation of Catalent Net Sales. For clarity, Catalent Net Sales does not include any manufacturing fees or other amounts invoiced by Catalent or any of its Affiliates in connection with the supply of Licensed Product by Catalent or its Affiliate to a Third Party. In the event a Licensed Product is commercialized by a Catalent Selling Party as part of a Combination Product in a country, the Catalent Net Sales with respect to the Combination Product in such country shall be determined by multiplying the Catalent Net Sales amount for the Combination Product

3 during the applicable reporting period, calculated as set forth above, by the fraction A/(A+B), where A is the list price of the Licensed Product in such country when sold separately, and B is the list price of the other active ingredient(s) in the Combination Product in such country when sold separately, in each case in the same dosage and dosage form and in the same country as the Combination Product during the applicable reporting period. If the Licensed Product or other active ingredient(s) in the Combination Product is sold separately in such country, but not at an equivalent dosage as contained in the Combination Product, then the price when sold separately shall be adjusted proportionally based on dosage to determine the imputed price of a product containing the relevant dosage of the relevant active ingredient when sold separately, which dosage is equivalent to that of the Combination Product. If neither the Licensed Product nor the other active ingredient(s) were sold separately in such country during the applicable reporting period at any dosage strength, then A shall equal the Licensed Product’s fair market value contribution to the Combination Product’s selling price, and B shall equal the fair market value of the contribution to the Combination Product’s selling price of the other active ingredient(s) in the Combination Product. 1.9 “Catalent Representatives” means Catalent, its Affiliates and each of their respective employees and contractors. 1.10 “Certepetide” means that certain investigational drug known as certepetide that is under clinical development by Lisata as of the Effective Date, the chemical structure of which is depicted on Appendix A. 1.11 “Change of Control” means, with respect to a Party: (a) a merger, reorganization, consolidation, or other transaction or series of related transactions involving such Party (or involving any Affiliate that directly or indirectly controls such Party) in which the voting securities of such Party or such Affiliate outstanding immediately prior thereto cease to represent at least fifty percent (50%) of the combined voting power of the surviving entity immediately after such merger, reorganization, consolidation, or other transaction or series of related transactions; (b) any Person, or group of Persons acting in concert, acquires more than fifty percent (50%) of the voting equity securities or management control of such Party (or of any Affiliate that directly or indirectly controls such Party) or (c) the assignment or transfer to a Third Party of all or substantially all of the assets of such Party; provided, however, that an initial listing of a Party or any such Affiliate on a securities exchange shall not, by itself, constitute a Change of Control. 1.12 “Combination Product” means any pharmaceutical product containing (a) a Licensed Product and (b) at least one other active pharmaceutical ingredient, either co-formulated or packaged together and sold as a single unit for a single price. 1.13 “Compound” means: (a) Certepetide and (b) any other iRGD Peptide. 1.14 “Control” or “Controlled” means, with respect to any Know-How, Patents, or other intellectual property rights, the legal authority or right (whether by ownership, license, or otherwise but without taking into account any rights granted by one Party to the other Party pursuant to this Agreement) of a Party or any of its Affiliates to grant access, a license, or a sublicense of or under such Know-How, Patents, or other intellectual property rights to the other Party, or to otherwise disclose proprietary or trade secret information to such other Party, without breaching the terms of any agreement with a Third Party, or misappropriating the proprietary or trade secret information of a Third Party. 1.15 “Confidential Information” means any proprietary, nonpublic information related to the business, technology, finances, products, processes, customers, licensors or suppliers of a Party that is disclosed by such Party (“Disclosing Party”) to the other Party (“Receiving Party”) in connection with this Agreement. Confidential Information may include, without limitation, any and all nonpublic information, know how, data, designs, plans, specifications, structures, documents, trade secrets, ideas,

4 concepts, products, processes, prototypes, formulas, works in progress, systems, technologies, manufacturing or marketing techniques, business or financial information and other proprietary and nonpublic information of the Disclosing Party, as well as any information with respect to which the Disclosing Party has a confidentiality obligation to a Third Party. Confidential Information may be written, recorded or otherwise fixed in a tangible medium, electronically communicated, or orally or visually communicated, furnished, provided or disclosed by a Disclosing Party, or acquired by a Receiving Party, directly or indirectly, from the Disclosing Party. The terms and conditions of this Agreement shall be deemed to be Confidential Information of both Parties. 1.16 “Cover,” “Covering” or “Covers” means (a) as to a method, compound or product and a Patent, that, in the absence of a license granted under, or ownership of, such Patent, the making, using, selling, offering for sale or importation of such method, compound or product would infringe any Valid Claim of such Patent or, as to a pending claim included in such Patent, the making, using, selling, offering for sale or importation of such method, compound or product would infringe such Patent if such pending claim were to issue in an issued patent without modification, and (b) as to Know-How and a Patent, that, in the absence of a license granted under, or ownership of, such Patent, the use or practice of such Know-How would infringe such Patent or, as to a pending claim included in such Patent, the use or practice of such Know-How would infringe such Patent if such pending claim were to issue in an issued patent without modification. 1.17 “FDA” means the United States Food and Drug Administration and any successor agency or authority having substantially the same function. 1.18 “Field” means the prevention, treatment, diagnosis, detection, monitoring or pre- disposition treatment or control of any disease, disorder or condition in humans. 1.19 “First Commercial Sale” means, on a country-by-country basis, the first sale by Catalent or any of its Affiliates to a Third Party for end use of the Licensed Product in such country after all Regulatory Approvals have been granted with respect to the sale of the Licensed Product in such country. 1.20 “IND” means an investigational new drug application or equivalent application filed with the applicable Regulatory Authority, which application is required to commence human clinical trials in the applicable country. 1.21 “Initiation” means, with respect to a clinical trial of a Licensed Product, the first dosing of the first human subject pursuant to the protocol for such clinical trial. 1.22 “iRGD Peptide” means any internalizing peptide that (a) contains 9 amino acids and (b) includes an Arginine-Glycine-Aspartic acid sequence. 1.23 “Know-How” means any tangible and intangible information, techniques, technology, practices, inventions (whether patentable or not), discoveries, methods, knowledge, know-how, trade secrets, data, specifications, instructions, processes, formulae, compositions of matter, cells, cell lines, assays, animal models and other physical, biological, or chemical materials and expertise, and further including all biological, chemical, pharmacological, biochemical, toxicological, pharmaceutical, physical, analytical, quality control, safety, manufacturing, development, nonclinical and clinical data, regulatory documents, data and filings, instructions, processes, formulae, expertise and information. For the avoidance of doubt, Know-How excludes Patents. 1.24 “Licensed Know-How” means any Know-How that is (a) Controlled by Lisata or any of its Affiliates as of the Effective Date or at any time during the Term and (b) necessary or reasonably useful for the research, development, manufacture or commercialization of the Licensed Products in the Field.

5 1.25 “Licensed Patents” means any Patents that are (a) Controlled by Lisata or any of its Affiliates as of the Effective Date or at any time during the Term and (b) necessary or reasonably useful for the research, development, manufacture or commercialization of the Licensed Products in the Field. A list of the Licensed Patents as of the Effective Date is attached to this Agreement as Appendix B. 1.26 “Licensed Product” means any bioconjugate therapeutic product or product candidate (in all forms, presentations, formulations and dosage forms) that is developed by Catalent or any of its Affiliates, or by a Sublicensee pursuant to a Sublicense, that contains and/or is enabled by a Compound. 1.27 “Licensed Product Sale” means any transaction pursuant to which Catalent assigns or otherwise transfers to a Third Party its rights to any one or more Licensed Products, other than as part of a larger transaction that also includes the assignment or other transfer of any assets that do not consist of rights to a Licensed Product. 1.28 “Licensed Product Transaction” means any (i) Sublicense or (ii) Licensed Product Sale. 1.29 “Licensed Product Transaction Revenue” means any payments (including up-front fees, development and commercial milestone payments and royalties) that Catalent or any of its Affiliates receives from a Third Party during the Royalty Term in consideration for a Licensed Product Transaction, minus any transaction costs incurred in connection with such Licensed Product Transaction; provided, however, that if Catalent or any of its Affiliates receives any such payment on account of achievement of any Milestone Event (a “Third Party Milestone Payment”), then only that portion of such Third Party Milestone Payment that is in excess of that amount that is payable by Catalent under Section 5.1 for the corresponding Milestone Event shall be counted as Licensed Product Transaction Revenue. For clarity, Licensed Product Transaction Revenue shall exclude any amounts received by Catalent or any of its Affiliates for research and development or manufacturing services performed by Catalent or any of its Affiliates for any Third Party. 1.30 “Licensed Technology” means the Licensed Know-How together with the Licensed Patents. 1.31 “Lisata-Introduced Partner” means any Sublicensee or other Party to a Licensed Product Transaction that is first introduced to Catalent by Lisata, as demonstrated via bona fide, contemporaneous documentation; provided, however, that if Catalent or any of its Affiliates enters into a Licensed Product Transaction with a Lisata-Introduced Partner or any of its Affiliates (an “Initial Transaction Partner”), then such Initial Transaction Partner (and any of its Affiliates and any of its or their respective successors or assigns) shall be deemed to be Catalent-Introduced Partners (and not Lisata-Introduced Partners) with respect to any subsequent Licensed Product Transaction entered into with Catalent or any of its Affiliates. 1.32 “Lisata Representatives” means Lisata, its Affiliates and each of their respective employees and contractors. 1.33 “Patents” means (a) all patents, certificates of invention, applications for certificates of invention, priority patent filings, and patent applications, and (b) any renewals, divisions, continuations (in whole or in part), or requests for continued examination of any of such patents, certificates of invention and patent applications, and any and all patents or certificates of invention issuing thereon, and any and all reissues, reexaminations, extensions, supplementary protection certificates, divisions, renewals, substitutions, confirmations, registrations, revalidations, revisions, and additions of or to any of the foregoing. 1.34 “Phase 1 Clinical Trial” means a human clinical trial of a Licensed Product in patients and/or healthy volunteers with the primary objective of characterizing its safety, tolerability, and pharmacokinetics and identifying a recommended dose and regimen for future studies as described in US

6 21 CFR § 312.21(a) or a comparable clinical trial prescribed by the relevant Regulatory Authority in a country other than the United States. 1.35 “Phase 2 Clinical Trial” means a human clinical trial of a Licensed Product that is intended to initially evaluate the effectiveness of a Licensed Product for a particular indication or indications in patients with the disease or indication under study or would otherwise satisfy requirements of U.S 21 CFR § 312.21(b) or its foreign equivalent. The phase 2 portion of a Phase 1/2 clinical trial shall be deemed to be a Phase 2 Clinical Trial. 1.36 “Phase 3 Clinical Trial” means a human clinical trial in any country, the results of which could be used to establish safety and efficacy of a Licensed Product as a basis for a BLA or would otherwise satisfy requirements of U.S 21 CFR § 312.21(c), or its foreign equivalent. 1.37 “Regulatory Approval” means any and all approvals (including, where required, pricing and reimbursement approval), licenses, registrations, permits, notifications, and authorizations (or waivers), establishment license, registration or authorization of any Regulatory Authority that are necessary for the manufacture, use, storage, import, transport, promotion, marketing, distribution, offer for sale, sale, or other commercialization of any Licensed Product for use in the Field in any country or jurisdiction in the Territory. 1.38 “Regulatory Authority” means any national, international, regional, state or local regulatory agency, department, bureau, commission, council or other governmental entity with authority over the distribution, importation, exportation, manufacture, production, use, storage, transport, clinical testing, pricing, sale or reimbursement of any Licensed Product for use in the Field in any country or jurisdiction in the Territory. 1.39 “Right of Reference” means as that term is defined in US 21 CFR §314.3(b) or any analogous Applicable Laws recognized outside of the United States. 1.40 “Royalty Term” means, on a country-by-country and Licensed Product-by-Licensed Product basis, the period that begins on the First Commercial Sale of a given Licensed Product in such country by Catalent or any of its Affiliates and ends on the expiration of the last-to-expire Valid Claim of any Licensed Patent that Covers such Licensed Product in such country. 1.41 “Sublicensee” means any Third Party that is granted a sublicense under any of the rights granted under the License (in each case, subject to the requirements of Section 2.2), whether such sublicense is granted by Catalent or by any sublicensee of any of the rights granted under the License, through multiple tiers. 1.42 “Territory” means worldwide. 1.43 “Third Party” mean any person or entity other than Catalent, Lisata or any of their respective Affiliates. 1.44 “Upstream License Agreement” means that certain Exclusive License Agreement between Lisata (formerly Cend Therapeutics, Inc.) and The Sanford Burnham Prebys Medical Discovery Institute (“Burnham Institute”) dated December 1, 2015, amended with a First Amendment dated March 8, 2016, later amended with a Second Amendment dated May 10, 2019, and further amended with a Third Amendment dated October 6, 2020. 1.45 “Valid Claim” means (a) a claim of an issued and unexpired patent that has not been revoked or held unenforceable, unpatentable, or invalid by a decision of a court or other governmental agency of competent jurisdiction that is not appealable or has not been appealed within the time allowed

7 for appeal, and that has not been abandoned, disclaimed, denied, or admitted to be invalid or unenforceable through reissue, re-examination, disclaimer, or otherwise, or (b) a claim of a pending patent application that has not been cancelled, withdrawn, abandoned, or finally rejected by an administrative agency action from which no appeal can be taken and that has not been pending for more than seven (7) years from the date of filing of the earliest priority patent application to which such pending patent application is entitled to claim benefit, excluding, however, any pending application that does not have a reasonable bona fide basis for patentability (such reasonable bona fide basis to be determined by an outside counsel selected in good faith by the Parties, in the event that the Parties disagree as to whether there is a reasonable bona fide basis for patentability for such a claim). 1.46 Additional Definitions. Each of the following terms is defined in the Section set forth opposite such term: Term Section Agreement Preamble Assigned Licensed Products 13.5.2 Catalent Preamble Catalent Indemnitees 11.2 Catalent Selling Party 1.5 Effective Date Preamble Indemnitee 11.3.1 Indemnitor 11.3.1 Intellectual Property 7.1 JSC 4.6.1 Liabilities 11.1 License 2.1 Licensed Product Assignment 13.5.2 Lisata Preamble Lisata Indemnitees 11.1 Milestone Event 5.1 Milestone Payment 5.1 Out-Licensing Process 3.1 Pharmacovigilance Agreement 4.4 Parties Preamble Party Preamble Payments 5.5 Revenue Share Payments 5.2 Revenue Statement 5.4 ROFN 3.1 ROFN Notice 3.1 ROFN Period 3.1 Royalty Payments 5.3 Sublicense 2.2 Term 6.1 US GAAP 1.5 2. LICENSE

8 2.1 License Grant. Lisata hereby grants to Catalent a non-exclusive, royalty-bearing, non- transferable (except as permitted under Section 13.5) license under the Licensed Technology, with the right to grant sublicenses through multiple tiers (subject to Section 2.2 below), to use, research, develop, make, have made, offer for sale, sell, have sold, import, export and otherwise commercialize and exploit the Licensed Products for use in the Field in the Territory during the Term (the “License”). 2.2 Sublicenses. Catalent may grant sublicenses, through multiple tiers of Sublicensees, under the License (each, a “Sublicense”) to its Affiliates and Third Parties; provided, however, that Catalent will continue to be responsible and liable vis-à-vis Lisata for the performance of its obligations under this Agreement (including remaining responsible for all payments due to Lisata hereunder) and for all acts and omissions of any Sublicensee with respect to such obligations. Catalent will provide Lisata with copy of any such sublicense agreement within thirty (30) days following execution of such sublicense agreement. 3. RIGHT OF FIRST NEGOTIATION 3.1 Grant of ROFN. If Catalent initiates a specific, organized process to out-license a Licensed Product (including, without limitation, by engaging an investment bank, consultant, or other adviser to solicit offers, or otherwise creating a bespoke process for soliciting offers to license such Licensed Product) (an “Out-Licensing Process”), Catalent shall first offer Lisata the right to negotiate in good faith for the license of such Licensed Product, subject to the terms set forth in this Section 3 (the “ROFN”). The ROFN for a given Licensed Product shall commence upon written notice from Catalent to Lisata identifying the applicable Licensed Product (the “ROFN Notice”) and shall continue for up to sixty (60) days from the date of such ROFN Notice, unless terminated earlier by written notice to Catalent from Lisata that it elects to discontinue negotiations (the “ROFN Period”). Catalent shall have sole discretion in determining the structure, timeline, and terms of the out-licensing process with respect to each Licensed Product, and nothing herein shall require Catalent to alter such process to accommodate Lisata. Upon expiration or earlier termination of the ROFN Period, Catalent shall have no further obligation to Lisata under this Section 3.1 with respect to the Licensed Product that is the subject of the applicable ROFN Notice. 3.2 Non-Exclusive Negotiations Outside ROFN Process. If a Third Party expresses interest in licensing a Licensed Product outside of the Out-Licensing Process described in Section 3.1, Catalent may, but is not obligated to, inform Lisata. Any discussions in such circumstances shall be on a non- exclusive basis and shall not preclude Catalent from engaging in concurrent or preferential negotiations with Third Parties. No exclusive negotiation period shall apply in such circumstances. 3.3 Reservation of Rights. Nothing in this Section 3 shall restrict or limit Catalent’s right to (i) publicly disclose information regarding Licensed Products in its pipeline, (ii) engage in its general business model of out-licensing pipeline assets, or (iii) respond to or pursue unsolicited inquiries from Third Parties at any time, including during the ROFN Period (provided that Catalent has not at the time entered into a binding agreement to license the applicable Licensed Product to Lisata). 3.4 No Obligation to Agree. The Parties acknowledge that the ROFN does not obligate either Party to enter into any definitive agreement related to the applicable Licensed Product, and no such agreement shall be binding unless and until executed by both Parties. 3.5 Not Binding Following Catalent Business Sale. In the event of any Catalent Business Sale, Catalent’s obligations under this Section 3 shall terminate and shall not be binding on Catalent or the acquiring entity in such Catalent Business Sale, unless otherwise agreed in writing following the closing of such Catalent Business Sale.

9 4. DEVELOPMENT AND COMMERCIALIZATION OF LICENSED PRODUCTS 4.1 Diligence. Catalent, in its sole discretion, shall use commercially reasonable efforts to develop, obtain Regulatory Approval for one or more Licensed Products, and commercialize any Licensed Product for which Regulatory Approval has been obtained. Catalent can either itself perform these activities and/or work with Affiliates and/or Third Parties to perform such activities. 4.2 Development and Commercialization of Licensed Products. 4.2.1 Catalent Responsibilities. As between the Parties, Catalent will have sole responsibility for and discretion over all decisions related to the pre-clinical and clinical development, manufacture and commercialization of any Licensed Product, including, but not limited to, with respect to selection of any specific indication to develop, regulatory strategy, launch, marketing, pricing and all other matters. Catalent shall, in its sole discretion, be permitted to pursue and/or enter into any agreements within Third Parties related to the ownership and/or development and commercialization rights for any Licensed Product. 4.2.2 Lisata Responsibilities. Lisata will (a) provide Catalent and/or its designee with copies of all data, records, reports and other information in the possession or control of Lisata or its Affiliates as of the Effective Date or at any time during the Term that relate to any Compound and are relevant to, or may be useful for, the development of Licensed Products and (b) upon Catalent’s request, provide Catalent and/or its designee with reasonable access to persons knowledgeable about the Compounds, for the purpose of answering Catalent’s questions and providing other technical assistance related to the Compounds for the purpose of developing Licensed Products. Without limiting the foregoing, promptly following the creation or receipt of such data, records, reports or other information, Lisata will promptly provide Catalent with: (i) all clinical trial design information related to any clinical trial involving a Compound, (ii) all data, records and reports generated pursuant to any such clinical trial and (iii) all CMC information and other information related to the manufacture of any Compounds; in each case to the extent such data, records, reports or other information are the possession or control of Lisata or its Affiliates and may be disclosed to Catalent without violating any confidentiality obligations to Third Parties. For clarity, all data, records, reports and other information provided by Lisata will be deemed to be included in the Licensed Know-How that is licensed to Catalent pursuant to this Agreement. 4.3 Right of Reference. Lisata (on behalf of itself and its Affiliates) hereby grants to Catalent an irrevocable, permanent, royalty-free, transferable, sublicensable Right of Reference and right to use all submissions for Regulatory Approval submitted by or on behalf of Lisata or any of its Affiliates (and all resulting Regulatory Approvals) pertaining any Compound worldwide, for the purpose of seeking, obtaining, and maintaining Regulatory Approvals of Licensed Products for the Territory. 4.4 Adverse Events. Prior to commencement of any clinical studies related to a Licensed Product by or on behalf of Catalent, any of its Affiliates or any Sublicensee, the Parties shall negotiate in good faith and enter into a pharmacovigilance agreement on such reasonable and customary terms as are agreed upon by the Parties (the “Pharmacovigilance Agreement”) with respect to the Parties’ exchange of information regarding Compounds and Licensed Product-related adverse events and the reporting of such information to the appropriate Regulatory Authorities. 4.5 Provision of Initial Licensed Product Data to Lisata. Solely with respect to the first three (3) Licensed Products under development by Catalent (the “Initial Licensed Products”), Catalent will provide Lisata with data generated by Catalent and its Affiliates related to the Initial Licensed Products, solely to the extent Catalent is able to do so without violating any confidentiality obligations to Third Parties. Lisata may: (a) use any such data that is provided to Lisata by Catalent with respect to the Initial Licensed Products (the “Initial Licensed Product Data”) internally at Lisata for the purpose of exploring

10 potential licensing opportunities for the Compounds and (b) disclose the Initial Licensed Product Data to Third Parties in connection with such licensing opportunities, provided that: (i) such disclosures are limited to a subset of the Initial Licensed Product Data that Catalent and Lisata agree may be shared with Third Parties by Lisata, (ii) each such Third Party is approved by Catalent in writing (such approval not to be unreasonably withheld, conditioned, or delayed) and (iii) each such Third Party agrees to maintain the confidentiality of such Initial Licensed Product Data on terms that are at least as protective of such Initial Licensed Product Data as those set forth in Section 8. For clarity, Catalent shall remain the owner of all rights to any Initial Licensed Product Data and may use and disclose the Initial Licensed Product Data without restriction. 4.6 Joint Steering Committee. 4.6.1 Formation and Role. Within thirty (30) days after the Effective Date, the Parties shall establish a joint steering committee (the “JSC”) to coordinate, review and discuss the Parties’ activities related to this Agreement. For that purpose and to the extent reasonably necessary, the JSC shall (by itself or through discharging its responsibilities through one (1) or more subcommittees): (a) align on anticipated timelines for activities under this Agreement; (b) discuss the status, progress, and results of pre-clinical and clinical development activities conducted by or on behalf of either Party with respect to Compounds and/or Licensed Products; (c) facilitate communications and discussions between the Parties with respect to Licensed Products development; and (d) perform such other functions as appropriate to further the purposes of this Agreement, as agreed by the Parties in writing. In no event shall the JSC or any subcommittee of the JSC have any authority to: (i) amend, modify, or waive compliance with this Agreement; (ii) determine that a breach has occurred under this Agreement; or (iii) make any decision that is specified elsewhere in this Agreement as being made by one or both Parties. 4.6.2 JSC Members. The JSC shall have four (4) members, with one (1) representative from each Party appointed as a co-chairperson. Catalent shall appoint two (2) representatives to the JSC, and Lisata shall appoint two (2) representatives to the JSC. Each JSC representative may be an officer, employee, consultant or other representative of the applicable Party having sufficient experience and knowledge of matters arising within the scope of the JSC’s responsibilities to make decisions with respect thereto. The role of each co-chairperson shall be to convene and preside at the meetings of the JSC and to ensure the preparation of meeting minutes, but, except as set forth herein, each co-chairperson shall have no additional powers or rights beyond those held by other JSC representatives. The JSC may change its size from time to time; provided that the JSC shall consist at all times of an equal number of representatives of each Party. Each Party may replace any of its JSC representatives with a qualified employee of such Party at any time upon written notice to the other Party. 4.6.3 Meetings. Unless the Parties agree otherwise, the JSC shall meet on a periodic basis as is agreed upon by the JSC. The JSC may conduct such meetings in-person, by videoconference or by teleconference, as the Parties agree. Each Party may invite a reasonable number of participants, in addition to its representatives, to attend JSC meetings; provided that if either Party wishes to have any Third Party (including any consultant) attend such a meeting, such Party shall (a) obtain the other Party’s prior, written consent before permitting such Third Party to attend any JSC

11 meeting and (b) ensure that such Third Party is bound by confidentiality and non-use obligations consistent with the terms of this Agreement. Each Party is responsible for its own expenses incurred in connection with participating in and attending all such meetings. 4.6.4 Decision Making. Each Party shall have a single vote in the JSC regardless of the number of representatives appointed to the JSC or present at the meeting. There must be a minimum of two (2) representatives from each Party at any meeting of the JSC in order for any action taken at such meeting to be valid. The JSC shall act by unanimous consent of both Parties. If after reasonable discussion and good faith consideration of each Party’s view on a particular matter before the JSC, the JSC cannot reach a unanimous decision as to such matter within five (5) Business Days after such matter was first brought to the JSC for resolution, then Catalent shall have the final decision-making authority with respect to such matter. 5. FINANCIAL TERMS 5.1 Milestone Payments. Catalent will pay the one-time, non-refundable, non-creditable milestone payments set forth in the below table (each, a “Milestone Payment”) to Lisata within thirty (30) days after the first occurrence of each event set forth in the below table, provided that such event occurs during the Royalty Term (each, a “Milestone Event”). Milestone Event Milestone Payment Initiation of the first Phase 1 Clinical Trial for the first Licensed Product $0 Initiation of the first Phase 1 Clinical Trial for the second Licensed Product $2,250,000 Initiation of the first Phase 1 Clinical Trial for the third Licensed Product $2,500,000 Initiation of the first Phase 1 Clinical Trial for the fourth Licensed Product $2,750,000 Initiation of the first Phase 1 Clinical Trial for the fifth and each subsequent Licensed Product $3,000,000 5.2 Revenue Share Payments. Catalent will pay Lisata a portion of Licensed Product Transaction Revenue as set forth in Section 5.2.1 or Section 5.2.2 below (in each case, subject to Section 5.2.3) (as applicable, “Revenue Share Payments”), depending on whether such Licensed Product Transaction Revenue is received from a Catalent-Introduced Partner or a Lisata-Introduced Partner. 5.2.1 Catalent-Introduced Partners. Subject to Section 5.2.3, within forty-five (45) days following receipt of any Licensed Product Transaction Revenue by Catalent or any of its Affiliates from any Catalent-Introduced Partner, Catalent shall pay Lisata the applicable percentage of such Licensed Product Transaction Revenue set forth in the following table (with such percentage determined in accordance with the following table, based on the status of the appliable Licensed Product immediately before the closing of the applicable Licensed Product Transaction):
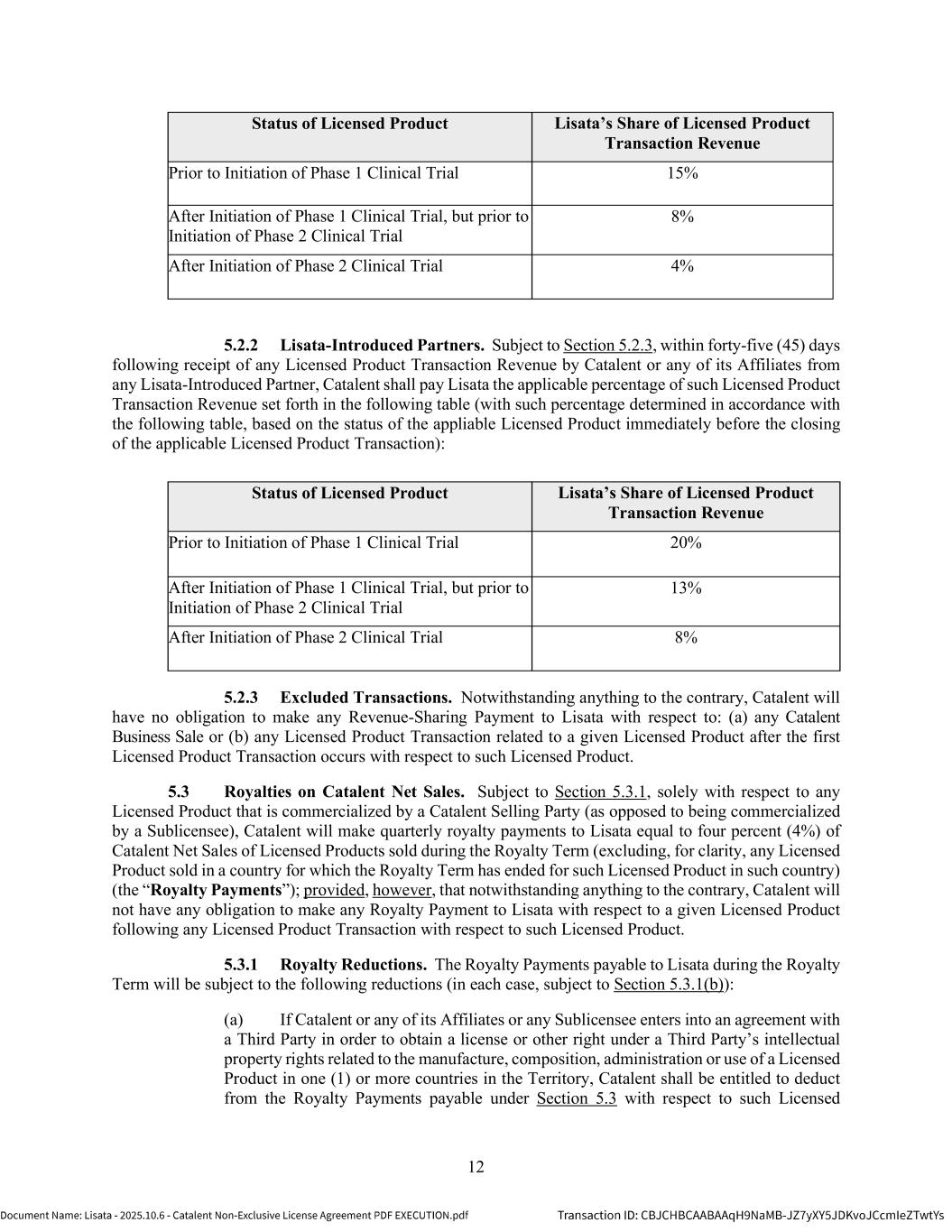
12 Status of Licensed Product Lisata’s Share of Licensed Product Transaction Revenue Prior to Initiation of Phase 1 Clinical Trial 15% After Initiation of Phase 1 Clinical Trial, but prior to Initiation of Phase 2 Clinical Trial 8% After Initiation of Phase 2 Clinical Trial 4% 5.2.2 Lisata-Introduced Partners. Subject to Section 5.2.3, within forty-five (45) days following receipt of any Licensed Product Transaction Revenue by Catalent or any of its Affiliates from any Lisata-Introduced Partner, Catalent shall pay Lisata the applicable percentage of such Licensed Product Transaction Revenue set forth in the following table (with such percentage determined in accordance with the following table, based on the status of the appliable Licensed Product immediately before the closing of the applicable Licensed Product Transaction): Status of Licensed Product Lisata’s Share of Licensed Product Transaction Revenue Prior to Initiation of Phase 1 Clinical Trial 20% After Initiation of Phase 1 Clinical Trial, but prior to Initiation of Phase 2 Clinical Trial 13% After Initiation of Phase 2 Clinical Trial 8% 5.2.3 Excluded Transactions. Notwithstanding anything to the contrary, Catalent will have no obligation to make any Revenue-Sharing Payment to Lisata with respect to: (a) any Catalent Business Sale or (b) any Licensed Product Transaction related to a given Licensed Product after the first Licensed Product Transaction occurs with respect to such Licensed Product. 5.3 Royalties on Catalent Net Sales. Subject to Section 5.3.1, solely with respect to any Licensed Product that is commercialized by a Catalent Selling Party (as opposed to being commercialized by a Sublicensee), Catalent will make quarterly royalty payments to Lisata equal to four percent (4%) of Catalent Net Sales of Licensed Products sold during the Royalty Term (excluding, for clarity, any Licensed Product sold in a country for which the Royalty Term has ended for such Licensed Product in such country) (the “Royalty Payments”); provided, however, that notwithstanding anything to the contrary, Catalent will not have any obligation to make any Royalty Payment to Lisata with respect to a given Licensed Product following any Licensed Product Transaction with respect to such Licensed Product. 5.3.1 Royalty Reductions. The Royalty Payments payable to Lisata during the Royalty Term will be subject to the following reductions (in each case, subject to Section 5.3.1(b)): (a) If Catalent or any of its Affiliates or any Sublicensee enters into an agreement with a Third Party in order to obtain a license or other right under a Third Party’s intellectual property rights related to the manufacture, composition, administration or use of a Licensed Product in one (1) or more countries in the Territory, Catalent shall be entitled to deduct from the Royalty Payments payable under Section 5.3 with respect to such Licensed

13 Product in such country(ies) fifty percent (50%) of all amounts paid to such Third Party pursuant to the terms of such agreement, in each case to the extent reasonably allocable to such Third Party right in such country. Catalent may carry forward to subsequent calendar quarters any amounts that it was not able to credit under this Section 5.3.1(a) on account of the royalty floor set forth in Section 5.3.1(b), and credit such carried forward amounts towards any future milestone or royalty payment hereunder. (b) Notwithstanding anything to the contrary, with respect to any Licensed Product in any calendar quarter, the operation of Section 5.3.1(a) will not reduce by more than fifty percent (50%) the Royalty Payment that would otherwise have been due under Section 5.3 with respect to such Licensed Product in the applicable country(ies) during a given calendar quarter. 5.4 Revenue Statements. Within forty-five (45) days after the end of each calendar quarter during which any Licensed Product Transaction Revenue or Catalent Net Sales are earned for which Lisata is entitled to receive a Revenue Share Payment or Royalty Payment under this Agreement, Catalent will deliver to Lisata a complete and accurate report, giving such particulars of the business conducted during the preceding quarter under this Agreement as are pertinent to an accounting of Revenue Share Payments and Royalty Payments that may be due to Lisata under this Agreement (the “Revenue Statement”). Each Revenue Statement will include the following information, broken down by country and Licensed Product: (a) in the case where Catalent or its Affiliates are commercializing a Licensed Product: (i) gross sales for such Licensed Product by Catalent and its Affiliates for such calendar quarter, (ii) deductions taken from such gross sales amounts and the calculation of Catalent Net Sales for such calendar quarter and (iii) calculation of Royalty Payments due based on such Catalent Net Sales; and (b) any Licensed Product Transaction Revenue received during such calendar quarter, broken down by source, and calculation of any applicable Revenue Share Payments. 5.5 Records and Audits. Catalent and its Affiliates shall keep true and accurate books and records in relation to the calculation of Revenue Share Payments and Royalty Payments under this Agreement (collectively, “Payments”). Catalent and its Affiliates shall keep such books and records for at least two (2) years following the calendar year to which they pertain. During the Term and for two (2) years thereafter, Catalent and its Affiliates will make such books of account available (no more than once per year upon at least thirty (30) days’ prior, written notice) for examination and audit by Lisata and/or its designated accounting firm during normal business hours. Lisata will be responsible for the cost of any such inspection; provided, however, that if an inspection shows for any year an underpayment in excess of five percent (5%) of the Payments payable with respect to any calendar year, then Catalent will reimburse Lisata for the reasonable, documented out-of-pocket expenses incurred to conduct the inspection, within thirty (30) days of receipt of notice from Lisata setting forth such costs. In the event that any such inspection reveals an underpayment in the amount of Payments that should have been paid by Catalent to Lisata, then the underpaid amount will be paid by Catalent within thirty (30) days after Lisata makes a demand therefor. Lisata will require its accounting firm to retain all information subject to review under this Section in strict confidence, and will treat all such information as Catalent’s Confidential Information. 5.6 Form of Payment; Currency Conversions. All Payments under this Agreement will be paid in United States dollars by wire transfer or electronic funds transfer to such bank account as Lisata may from time-to-time designate by notice. For the purposes of determining any Royalty Payments, Catalent Net Sales will first be determined in the currency in which such Catalent Net Sales are earned and then converted to its equivalent in United States currency. The buying rates of exchange for the currencies involved into the currency of the United States quoted by Citibank (or its successor in interest) in New

14 York, New York at the close of business on the last business day of the quarterly period in which the Catalent Net Sales were earned will be used to determine any such conversion. 5.7 Taxes. The amounts payable by Catalent to Lisata pursuant to this Agreement will not be reduced on account of any taxes unless required by Applicable Laws. Any taxes, duties, or other levies which Catalent is required by Applicable Laws to withhold on remittance of any payment(s) due under this Agreement will be deducted from such payment(s) to Lisata and timely paid to the appropriate taxing authority. Catalent will secure and send to Lisata proof of any such taxes, duties or other levies withheld and paid by Catalent for the benefit of Lisata, and cooperate, at Lisata’s expense, with any reasonable request to help ensure that amounts withheld and/or paid are reduced and/or recovered to the extent permitted by the relevant jurisdiction. 6. TERM AND TERMINATION 6.1 Term. The term of this Agreement will commence on the Effective Date and will remain in effect at all times thereafter, unless terminated earlier pursuant to Section 6.2 (the “Term”). 6.2 Termination. 6.2.1 Termination for Material Breach. Each Party shall have the right to terminate this Agreement immediately in its entirety upon written notice to the other Party if such other Party commits a material breach of this Agreement and has not cured such breach within ninety (90) days after notice of such breach from the non-breaching Party; provided, however, that if the breach is capable of being cured, but cure of such breach cannot reasonably be effected within such ninety (90)-day period, the breaching Party shall deliver to the non-breaching Party a plan reasonably calculated to cure such breach within a reasonable timeframe, but in any event within one hundred eighty (180) days from the date of the notice from the non-breaching Party of such material breach. So long as the breaching Party is diligently carrying out such plan, the non-breaching Party shall not have the right to terminate this Agreement prior to expiration of such one-hundred eighty (180) day period. If the alleged breaching Party disputes in good faith the existence or materiality of a breach specified in a notice provided by the other Party, and such alleged breaching Party provides the other Party notice of such dispute within thirty (30) days, then the other Party shall not have the right to terminate this Agreement under this Section 6.2.1 unless and until a court of competent jurisdiction, in accordance with Section 13.4, has determined that the alleged breaching Party has materially breached the Agreement and, if the breach is then curable, such Party fails to cure such breach within the applicable cure period set forth above following such decision. (a) Rights In Lieu of Termination for Material Breach. Notwithstanding anything to the contrary in the foregoing, if Catalent would otherwise have the right to terminate this Agreement pursuant to Section 6.2.1, then in lieu of such termination Catalent may, upon written notice to Lisata, elect to continue this Agreement in full force and effect, except that: (a) Catalent’s diligence obligations under Section 4.1 shall be deemed fully satisfied and (b) any consideration that would have been due to Lisata under Section 5 by Catalent accruing after such termination shall be reduced by fifty percent (50%). Notwithstanding the above, if Lisata disputes in good faith the existence or materiality of such breach, and provides Catalent notice of such dispute within thirty (30) days after receipt of Catalent’s notice, then the preceding subclauses (a) and (b) will not apply unless and until a court of competent jurisdiction, in accordance with Section 13.4, has determined that Lisata has committed such material breach and has not ceased the activities giving rise to such material breach or has otherwise failed to cure such breach within the applicable cure period set forth in Section 6.2.1.

15 6.2.2 Termination for Bankruptcy Events. Either Party may terminate this Agreement immediately upon written notice to the other Party if the other Party seeks protection under any bankruptcy, receivership or comparable proceeding, or if any such proceeding is instituted against the other Party and not dismissed within ninety (90) days. 6.2.3 Termination for Convenience by Catalent. Catalent may terminate this Agreement for convenience (either in its entirety, or in part with respect to any one or more countries in the Territory) at any time upon thirty (30) days prior written notice to Lisata. 6.2.4 Termination By Mutual Agreement. This Agreement may be terminated at any time if such termination is agreed upon by both Parties in writing. 6.3 Effect of Termination. 6.3.1 In the event of any termination of this Agreement, the License will immediately terminate; provided, however, that notwithstanding anything to the contrary, any Sublicenses granted prior to the effective date of any termination of this Agreement shall survive such termination and remain in effect in accordance with their terms. 6.3.2 In the event of any termination of this Agreement, each Party shall promptly return to the other Party, or delete or destroy, all relevant records and materials in such Party’s possession or control containing Confidential Information of the other Party; provided, however, that a Party may keep reasonable number of copies of such materials for archival purposes subject to continuing confidentiality obligations. 6.4 Survival. The following provisions will survive any termination of this Agreement: Section 1 (“Definitions”), Section 4.3 (“Right of Reference”), Section 5.5 (“Records and Audits”), Section 6.3 (“Effect of Termination”), this Section 6.4 (“Survival”), Section 7 (“Intellectual Property”), Section 8 (“Confidentiality”), Section 10 (“Representations, Warranties and Covenants; Disclaimer”), Section 11 (“Indemnification”), Section 12 (“Liability”) and Section 13 (“Miscellaneous Provisions”). 7. INTELLECTUAL PROPERTY 7.1 Definitions. For purposes hereof: A. “Catalent Invention” means any invention, work of authorship, Know-How or other item developed by either Party or any of their respective Representatives in connection with this Agreement (other than any Lisata Invention) that relates to any Licensed Product and/or any of Catalent’s bioconjugation technologies. B. “Lisata Invention” means any invention, work of authorship, Know-How or other item developed by either Party or any of their respective Representatives in connection with this Agreement that exclusively relates to Certepetide. 7.2 Ownership of Intellectual Property. 7.2.1 Generally. Except as expressly provided in Section 7.2.2 and Section 7.2.3, each Party shall retain ownership of all Patents and other intellectual property rights with respect to any invention, work of authorship, Know-How and other item that such Party owned prior to the Effective Date, or develops or acquires after the Effective Date outside of the scope of this Agreement. 7.2.2 Catalent Inventions. All Catalent Inventions shall be owned solely by Catalent and no right therein is granted to Lisata under this Agreement. Lisata hereby assigns (and shall cause each other Lisata Representative to assign) to Catalent all of Lisata’s and each other Lisata Representatives’

16 right, title, and interest in and to any Catalent Inventions. Lisata shall assist Catalent, at Catalent’s request and expense, in securing for Catalent any Patents to such Catalent Inventions, and performing all acts that may be reasonably required to vest in Catalent all right, title, and interest in such Catalent Inventions. 7.2.3 Lisata Inventions. All Lisata Inventions shall be owned solely by Lisata and no right therein is granted to Catalent under this Agreement (other than the rights granted to Catalent pursuant to the License). Catalent hereby assigns (and shall cause each other Catalent Representative to assign) to Lisata all of Catalent’s and each other Catalent Representatives’ right, title, and interest in and to any Lisata Inventions. Catalent shall assist Lisata, at Lisata’s request and expense, in securing for Lisata any Patents to such Lisata Inventions, and performing all acts that may be reasonably required to vest in Lisata all right, title, and interest in such Lisata Inventions. 7.2.4 Each Party shall be solely responsible for the costs associated with the protection of its respective intellectual property. 8. CONFIDENTIALITY 8.1 Confidential Information. The Receiving Party agrees that it shall not, without the prior written consent of the Disclosing Party: (a) disclose to any person the Disclosing Party’s Confidential Information, except to those of the Receiving Party’s and its Affiliates’ employees, representatives, consultants, service providers and contractors (and: (i) in the case of Catalent, any Sublicensees and (ii) in the case of Lisata, disclosure of a certain subset of Initial Licensed Product Data to Third Parties, if and to the extent permitted under Section 4.5) who need to know such information in connection with the Receiving Party’s exploitation of its rights or fulfillment of its obligations under this Agreement (and then only to the extent that such persons are under an obligation to maintain the confidentiality of the Confidential Information consistent with this Agreement) or (b) use any of the Disclosing Party’s Confidential Information for any reason other than in connection with the Receiving Party’s exploitation of its rights or fulfillment of its obligations under this Agreement. In addition, either Party may disclose the terms and conditions of this Agreement and other Confidential Information of the Disclosing Party on a confidential basis to existing and potential investors, Sublicensees, business partners, lenders and acquirers and their respective attorneys and advisors, in connection with licensing, financing and acquisition activities, and due diligence processes related to such activities. 8.2 Exceptions. The obligations under Section 8.1 will not apply to any information that the Receiving Party can show: (a) is now, or hereafter becomes, through no act or failure to act on the part of the Receiving Party, generally known or available to the public; (b) is known by the Receiving Party at the time of receiving such information, other than by previous disclosure of the Disclosing Party, or its Affiliates, employees, agents, consultants, or contractors; (c) is hereafter furnished to the Receiving Party without restriction by a Third Party who has no obligation of confidentiality with respect thereto; or (d) is independently discovered or developed by the Receiving Party without the use of the Disclosing Party’s Confidential Information. 8.3 Disclosures Required by Law. The Receiving Party may disclose the Disclosing Party’s Confidential Information to the limited extent required in order to comply with a subpoena or the order of a court or other governmental body, or as otherwise necessary to comply with applicable law; provided that the Receiving Party gives the Disclosing Party reasonable notice and the opportunity to seek confidential treatment, a protective order or similar remedies or relief prior to disclosure. 9. COMPLIANCE WITH APPLICABLE LAWS 9.1 Compliance with Applicable Laws. Each Party agrees that it will comply with all Applicable Laws in carrying out its responsibilities under this Agreement.

17 10. REPRESENTATIONS, WARRANTIES AND COVENANTS; DISCLAIMER 10.1 Mutual Representations and Warranties. Each Party makes the following representations and warranties to the other Party: 10.1.1 Organization. Such Party (i) is a corporation duly incorporated, validly existing and in good standing under the laws of the jurisdiction where such corporation was incorporated, and (ii) has all necessary corporate power and authority to own its properties and to conduct its business, as currently conducted. 10.1.2 Authorization. The execution and delivery of this Agreement and the consummation of the transactions contemplated hereby are within the company power of such Party, have been duly authorized by all necessary company proceedings of such Party, and this Agreement has been duly executed and delivered by such Party. 10.1.3 No Conflict. The execution and delivery of this Agreement and the consummation of the transactions contemplated hereby do not: (i) conflict with or result in a breach of any provision of such Party’s organizational documents; (ii) result in a material breach of any material agreement to which such Party is bound; (iii) result in a violation of any order to which such Party is subject; (iv) require such Party to obtain any material approval or consent from any governmental authority or other Third Party other than those consents and approvals which have been obtained prior to the date hereof; or (v) violate any Applicable Laws applicable to such Party in any material respect. 10.1.4 Enforceability. This Agreement constitutes the valid and binding obligation of such Party, enforceable against such Party in accordance with its terms, subject to bankruptcy, reorganization, insolvency and other similar laws affecting the enforcement of creditors’ rights in general and to general principles of equity (regardless of whether considered in a proceeding in equity or an action at law). 10.1.5 No Debarment. Neither Party nor its personnel involved in any manner with the development of any Compound or any Licensed Product have been debarred pursuant to the Act or excluded from any health care program sponsored by the United States Federal Government, including, but not limited to, Medicare or Medicaid, and it will notify the other Party immediately if it or any such personnel is debarred under the Act or excluded under any such health care program. 10.2 Additional Lisata Representations, Warranties and Covenants. Lisata represents, warrants and covenants to Catalent that, as of the Effective Date and at all times thereafter during the Term: 10.2.1 except for the Licensed Technology that is licensed to Lisata pursuant to the Upstream License Agreement, Lisata solely and exclusively owns, and at all times during the Term shall solely and exclusively own, all of the Licensed Technology that exists as of the Effective Date, free and clear of all liens and encumbrances; 10.2.2 Lisata has, and at all times shall maintain, the right to grant the License and all other rights and licenses it purports to grant to Catalent under this Agreement without conflict with the rights of any Third Party; 10.2.3 a list of all of the Licensed Patents as of the Effective Date is attached to this Agreement as Appendix B;

18 10.2.4 all issued Patents included in the Licensed Patents are, to Lisata’s knowledge, valid and enforceable; 10.2.5 there are no claims or assertions in writing received by Lisata or any of its Affiliates, or to Lisata’s knowledge, alleged or threatened: (a) alleging that any of the Licensed Patents are invalid or unenforceable; (b) regarding the inventorship of any of the Licensed Patents or alleging that additional or alternative inventors should be listed; or (c) alleging that the use of the Licensed Technology as contemplated by the License infringes or misappropriates, or would infringe or misappropriate, any Intellectual Property of any Third Party; 10.2.6 there are no pending or, to Lisata’s knowledge, alleged or threatened, inter partes reviews, post-grant reviews, interferences, re-examinations, or oppositions involving any of the Licensed Patents that are in or before any patent authority (or other governmental authority performing similar functions); 10.2.7 to Lisata’s knowledge: (a) the use of the Licensed Technology as contemplated by the License will not infringe or misappropriate any Intellectual Property of any Third Party IP and (b) no Third Party is infringing or misappropriating or has infringed or misappropriated or is threatening to infringe or misappropriate the Licensed Technology; 10.2.8 all Licensed Patents are filed and maintained properly and correctly and all applicable fees have been paid on or before the due date for payment or during any extension thereof; 10.2.9 none of the inventions included in Licensed Technology: (a) was conceived, discovered, developed, or otherwise made in connection with any research activities funded, in whole or in part, by any governmental agency, including, but not limited to, the federal government of the United States or any agency thereof, (b) is a “subject invention” as that term is described in 35 U.S.C. Section 201(e), or (c) is otherwise subject to the provisions of the Patent and Trademark Law Amendments Act of 1980, as amended, codified at 35 U.S.C. §§ 200-212, as amended, or any regulations promulgated pursuant thereto, including in 37 C.F.R. part 401; and 10.2.10 All activities conducted by or on behalf of Lisata or any of its Affiliates in connection with the development of any Compound have been conducted in accordance with Applicable Law. 10.3 Lisata Representations, Warranties and Covenants Regarding the Upstream License Agreement. Lisata represents, warrants and covenants to Catalent that, as of the Effective Date and at all times thereafter during the Term: 10.3.1 a true, complete and correct copy of the Upstream License Agreement has been provided or made available to Catalent prior to the Effective Date; 10.3.2 the Upstream License Agreement is valid, binding, enforceable, and in full force and effect; 10.3.3 (a) Lisata is in compliance in all material respects with the Upstream License Agreement, and, to Lisata’s knowledge, Burnham Institute is not in breach of the Upstream License Agreement; (b) no notice of breach, default, or termination has been received or given under the Upstream License Agreement and (c) no circumstances or grounds exist (including, but not limited to, the execution, delivery and performance of this Agreement) that would reasonably be expected to give rise to a claim by Burnham Institute of breach of, or a right of rescission, termination, revision or amendment of, the Upstream License Agreement;

19 10.3.4 Lisata has obtained all approvals and consents and will provide all notices required to be provided under the Upstream License Agreement in connection with the execution and delivery of this Agreement and the performance by Lisata of Lisata’s obligations hereunder; 10.3.5 Lisata shall at all times maintain in good standing the Upstream License Agreement and shall timely perform all obligations arising thereunder, including by making any payments required to be made under the Upstream License Agreement; 10.3.6 Lisata shall not agree to any amendment to the Upstream License Agreement that would adversely affect the rights sublicensed to Catalent under this Agreement without Catalent’s prior, written consent; and 10.3.7 (a) in the event that Lisata receives a notice or other communication alleging it is in breach (including a notice or other communication threatening termination) of the Upstream License Agreement, Lisata shall promptly provide Catalent with a copy of such notice and (b) without limiting any other right or remedy of Catalent under this Agreement and in order to prevent, ameliorate, mitigate, or cure a breach of the Upstream License Agreement, in the event that Lisata fails to perform any of its obligations under the Upstream License Agreement, Catalent may perform such obligation on behalf of Lisata, provided that Catalent shall be entitled to credit any out-of-pocket costs it incurs in performing any such obligation against any future payments otherwise owed to Lisata under this Agreement. 10.4 Disclaimer. EXCEPT AS SET FORTH IN SECTION 10.1, SECTION 10.2 AND SECTION 10.3, NEITHER PARTY MAKES, AND EACH HEREBY EXPRESSLY DISCLAIMS, ANY AND ALL REPRESENTATIONS AND WARRANTIES OF ANY KIND ARISING FROM OR RELATING TO THIS AGREEMENT OR SUCH PARTY’S PERFORMANCE HEREUNDER, THE LICENSED TECHNOLOGY, OR ANY LICENSED PRODUCT, EITHER EXPRESS OR IMPLIED, INCLUDING, BUT NOT LIMITED TO, ANY IMPLIED WARRANTIES OF MERCHANTABILITY, FITNESS FOR A PARTICULAR PURPOSE, NON-INFRINGEMENT AND ANY REPRESENTATIONS OR WARRANTIES ARISING FROM A COURSE OF DEALING, COURSE OF PERFORMANCE OR USAGE OF TRADE. 11. INDEMNIFICATION 11.1 Indemnification by Catalent. Subject to and except to the extent of any indemnification from Lisata pursuant to Section 11.2 below, Catalent will indemnify, defend and hold Lisata, its Affiliates, and its and their respective directors, officers, employees and agents (“Lisata Indemnitees”) harmless from and against all losses, damages, liabilities, settlements, penalties, fines, costs and expenses (including, without limitation, reasonable attorneys’ fees and expenses), (collectively, the “Liabilities”) to the extent such Liabilities arise out of or result from any claim, lawsuit or other action or threat by a Third Party arising out of: (a) any breach of this Agreement by Catalent or (b) any grossly negligent act or omission or willful misconduct by or on behalf of any of the Catalent Indemnitees; in each case, except to the extent that any of the foregoing arises out of or results from any Lisata Indemnitee’s negligence, willful misconduct or breach of this Agreement. 11.2 Indemnification by Lisata. Subject to and except to the extent of any indemnification from Catalent pursuant to Section 11.1 above, Lisata will indemnify, defend and hold Catalent, its Affiliates, and its and their respective directors, officers, employees and agents (“Catalent Indemnitees”) harmless from and against all Liabilities to the extent such Liabilities arise out of or result from any claim, lawsuit or other action or threat by a Third Party arising out of (a) any breach of this Agreement by Lisata or (b) any grossly negligent act or omission or willful misconduct by or on behalf of any of the Lisata

20 Indemnitees; in each case, except to the extent that any of the foregoing arises out of or results from any Catalent Indemnitee’s negligence, willful misconduct or breach of this Agreement. 11.3 Indemnification Procedures. 11.3.1 Identification of Indemnitor and Indemnitee. An “Indemnitor” means the indemnifying Party. An “Indemnitee” means the indemnified Party, its Affiliates, and their respective directors, officers, employees and agents. 11.3.2 Indemnification Procedures. An Indemnitee which intends to claim indemnification under Section 11.1 or Section 11.2 hereof will promptly notify the Indemnitor in writing of any claim, lawsuit or other action in respect of which the Indemnitee, its Affiliates, or any of their respective directors, officers, employees and agents intend to claim such indemnification; provided, that failure to provide such notice within a reasonable period of time will not relieve the Indemnitor of any of its obligations hereunder except to the extent the Indemnitee is prejudiced by such failure. The Indemnitee will permit, and will cause its Affiliates and their respective directors, officers, employees and agents to permit, the Indemnitor, at its discretion, to settle any such claim, lawsuit or other action and agrees to the complete control of such defense or settlement by the Indemnitor; provided, however, that in order for the Indemnitor to exercise such rights, such settlement will not adversely affect the Indemnitee’s rights under this Agreement or impose any obligations on the Indemnitee in addition to those set forth herein, including an admittance of fault. No such claim, lawsuit or other action will be settled without the prior written consent of the Indemnitor, which consent will not be unreasonably withheld, conditioned or delayed, and the Indemnitor will not be responsible for any legal fees or other costs incurred other than as provided herein. The Indemnitee, its Affiliates and their respective directors, officers, employees and agents will cooperate fully with the Indemnitor and its legal representatives in the investigation and defense of any claim, lawsuit or other action covered by this indemnification, all at the reasonable expense of the Indemnitor. The Indemnitee will have the right, but not the obligation, to be represented by counsel of its own selection and expense. 11.4 Insurance. In the event that any Licensed Product is commercialized, Catalent will procure and maintain (or will cause each Sublicensee to procure and maintain) from the date of first commercial sale of a Licensed Product until the date that is three (3) years after any expiration or termination of this Agreement, a products liability insurance policy with coverage of at least $5 million dollars per occurrence. Lisata and its Affiliates will be named as additional insureds within such products liability insurance policy. Catalent will provide to Lisata upon request, a copy of its insurance certificate evidencing such insurance. 12. LIABILITY 12.1 Liability Exclusion. NEITHER PARTY SHALL BE LIABLE TO THE OTHER PARTY FOR ANY INCIDENTAL, CONSEQUENTIAL, INDIRECT, SPECIAL, PUNITIVE OR EXEMPLARY DAMAGES OR ANY DAMAGES FOR LOSS OF BUSINESS, LOSS OF PROFITS OR THE LIKE, ARISING OUT OF OR RELATING TO THIS AGREEMENT OR SUCH PARTY’S PERFORMANCE HEREUNDER, THE LICENSED TECHNOLOGY, OR ANY LICENSED PRODUCT, EVEN IF SUCH PARTY HAS BEEN ADVISED OF THE POSSIBILITY OF SUCH DAMAGES AND REGARDLESS OF THE CAUSE OF ACTION (WHETHER IN CONTRACT, TORT, BREACH OF WARRANTY OR OTHERWISE), AND NOTWITHSTANDING ANY FAILURE OF THE ESSENTIAL PURPOSE OF ANY LIMITED REMEDY PROVIDED HEREIN. 12.2 Exceptions. Notwithstanding anything to the contrary, the liability exclusions set forth in Section 12.1 will not apply: (a) to damages arising from a Party’s fraud or willful misconduct or (b) the Parties’ respective indemnity and defense obligations under Section 11.

21 13. MISCELLANEOUS PROVISIONS 13.1 Rights in Bankruptcy. All rights and licenses granted under or pursuant to this Agreement by one Party to the other Party are, and otherwise will be deemed to be, for purposes of Section 365(n) of the U.S. Bankruptcy Code or comparable provision of applicable bankruptcy or insolvency laws, licenses of right to “intellectual property” as defined under Section 101 of the U.S. Bankruptcy Code or comparable provision of applicable bankruptcy or insolvency laws. The Parties agree that a Party that is a licensee of such rights under this Agreement will retain and may fully exercise all of its rights and elections under the U.S. Bankruptcy Code or comparable provision of applicable bankruptcy or insolvency laws. The Parties further agree that, in the event of the commencement of a bankruptcy proceeding by or against a Party to this Agreement under the U.S. Bankruptcy Code or comparable provision of applicable bankruptcy or insolvency laws, the other Party will be entitled to a complete duplicate of (or complete access to, as appropriate) any such intellectual property and all embodiments of such intellectual property, and same, if not already in its possession, will be promptly delivered to it (a) upon any such commencement of a bankruptcy or insolvency proceeding upon its written request therefor, unless the bankrupt Party elects to continue to perform all of its obligations under this Agreement, or (b) if not delivered under (a) above, following the rejection of this Agreement by or on behalf of the bankrupt Party upon written request therefor by the other Party. 13.2 Publicity. Neither Party nor any of their respective Affiliates, will, without the prior, written consent of the other Party, issue any press release or make any other public announcement concerning the existence of the Agreement, publicly disclose the terms of this Agreement, or use the other Party’s (or its Affiliate’s) name, trade name, trademark or other designation in a manner that could be construed as an endorsement of its products or services; provided, however, that notwithstanding the foregoing: (a) promptly after the Effective Date, the Parties will issue a joint press release substantially in the form attached to this Agreement as Appendix C and (b) either Party may publicly disclose the terms of this Agreement to the extent required by Applicable Laws or exchange rules. 13.3 Notices. Any notice or other communication required or permitted to be given hereunder (a) shall be in writing (email being sufficient) and (b) shall be deemed to have been received by the other Party (i) if personally delivered, (ii) one (1) Business Days after it is sent, if sent by overnight delivery using a nationally-recognized express courier service and providing proof of delivery, or (iii) upon sending, if sent by email with delivery receipt; in each case, to the other Party at the address set forth below; provided, however, that either Party, by written notice given in accordance with this Section to the other Party, may designate another address or person for receipt of notices hereunder, provided that notice of such a change shall be effective upon receipt: If to Catalent: Redwood Bioscience, Inc. c/o Catalent Pharma Solutions 5959 Horton Street Suite 400 Emeryville, CA 94608 Attn: Email: with a copy to:

22 Catalent Pharma Solutions, LLC 200 Crossing Blvd., 7th Floor, Bridgewater, NJ 08807 Attn: General Counsel (Legal Department) Email: GenCouns@catalent.com If to Lisata: Lisata Therapeutics, Inc. 110 Allen Road, 2nd Floor Basking Ridge, NJ 07920 Attn: General Counsel (Legal Department) Email: legal@lisata.com With a copy (which shall not constitute notice) to: Lisata Therapeutics, Inc. 110 Allen Road, 2nd Floor Basking Ridge, NJ 07920 Attn: Amy Jackson Ayala Email: aayala@lisata.com 13.4 Applicable Laws; Exclusive Forum. This Agreement, and all questions regarding the existence, validity, interpretation, breach, or performance of this Agreement, will be governed by, and construed and enforced in accordance with, the laws of the State of Delaware, without reference to its conflicts of law principles. The Parties hereby consent and agree to the exclusive jurisdiction of the state and federal courts located in Delaware, for all suits, actions or proceedings directly or indirectly arising out of or relating to this Agreement, and waive any and all objections to such courts, including but not limited to objections based on improper venue or inconvenient forum, and each Party hereby irrevocably submits to the jurisdiction of such courts in any suits, actions or proceedings arising out of or relating to this Agreement. 13.5 Assignment. 13.5.1 Neither Party may assign or transfer its interest under this Agreement without the prior, written consent of the other Party, such consent not to be unreasonably withheld, conditioned or delayed; provided, however, that notwithstanding the foregoing: (a) either Party may assign or transfer its interest under this Agreement, upon prior, written notice to the other Party but without any requirement to obtain the other Party’s consent: (i) to any of its Affiliates, (ii) pursuant to any merger involving such Party, (iii) pursuant to a sale of all or substantially all assets of such Party or its line of business to which this Agreement relates or (iv) in connection with any sale of equity interests or Change of Control transaction involving such Party and (b) Catalent may assign or transfer its interest under this Agreement, in whole or in part, pursuant to any Licensed Product Assignment, upon prior, written notice to Lisata but without any requirement to obtain Lisata’s consent. 13.5.2 “Licensed Product Assignment” means any assignment or transfer by Catalent to an Affiliate of Catalent or a Third Party of Catalent’s interest under this Agreement related to one or more Licensed Product(s) (whether as part of a Licensed Product Sale or as part of a Catalent Business Sale). “Assigned Licensed Products” means the Licensed Product(s) that are the subject of a Licensed Product Assignment. In the event of a Licensed Product Assignment that does not involve the assignment or transfer by Catalent of all of its interest under this Agreement:

23 (a) a new agreement shall be deemed to be formed between Lisata and the assignee or transferee in such Licensed Product Assignment (the “Acquiring Party”) which consists of all of the terms of this Agreement solely to the extent related to the applicable Assigned Licensed Product(s) (except that: (i) in the case where such Licensed Product Assignment is part of a Licensed Product Sale, such assignee or transferee will have no obligation to pay Lisata any applicable Revenue-Share Payments with respect to any Licensed Product Transaction Revenue from such Licensed Product Sale, (ii) such new agreement shall not include any of the terms set forth in Section 4.5 or Section 4.6 of this Agreement and (iii) for clarity, such new agreement shall not provide the Acquiring Party rights with respect to any product other than the applicable Assigned Licensed Product(s)) (the “Assigned Agreement”); (b) all references to “Catalent” in the Assigned Agreement shall be deemed to refer to the Acquiring Party, and all references to any “Licensed Product” in the Assigned Agreement shall be deemed to refer to the applicable Assigned Licensed Product(s); (c) this Agreement shall be remain in effect between Lisata and Catalent, provided that this Agreement will be deemed to be amended to provide that Catalent shall have no obligations to Lisata, and Lisata shall have no obligations to Catalent, with respect to the applicable Assigned Licensed Products, in each case to the extent such obligations arise after the effective date of the assignment or transfer of such Assigned Licensed Products to the Acquiring Party (except that, in the case where such Licensed Product Assignment is part of a Licensed Product Sale, Catalent will remain obligated to pay Lisata any applicable Revenue-Share Payments with respect to any Licensed Product Transaction Revenue from such Licensed Product Sale) (the “Retained Agreement”); (d) the Assigned Agreement and the Retained Agreement shall be deemed to be separate and independent agreements; (e) Catalent shall have no obligations or liabilities to Lisata, and Lisata shall have no obligations or liabilities to Catalent, under the Assigned Agreement; (f) the Acquiring Party shall have no obligations or liabilities to Lisata, and Lisata shall have no obligations or liabilities to the Acquiring Party, under the Retained Agreement; (g) no breach, termination or other acts or omissions of Lisata or the Acquiring Party under the Assigned Agreement shall have any effect on the Retained Agreement; and (h) no breach, termination or other acts or omissions of Lisata or Catalent under the Retained Agreement shall have any effect on the Assigned Agreement. 13.5.3 Any permitted assignment or transfer of this Agreement by either Party will be conditioned upon that Party’s permitted assignee or transferee agreeing in writing to comply with all the applicable terms and conditions contained in this Agreement (subject to Section 13.5.2, where applicable). Any purported assignment or transfer of this Agreement without a required consent will be void. No assignment or transfer of this Agreement will relieve any Party of responsibility for the performance of any obligation that accrued prior to the effective date of such assignment or transfer. This Agreement is binding on and inures to the benefit of the Parties and their respective permitted successors and assigns and transferees.

24 13.6 Further Assurances. The Parties agree to execute, acknowledge and deliver such further instruments and to take all such other incidental acts as may be reasonably necessary or appropriate to carry out the purpose and intent of this Agreement. 13.7 No Third-Party Beneficiaries. This Agreement will not confer any rights or remedies upon any person or entity other than the Parties named herein and their respective successors and permitted assigns. 13.8 Severability. If any part of this Agreement will be found to be invalid or unenforceable under applicable law in any jurisdiction, such part will be ineffective only to the extent of such invalidity or unenforceability in such jurisdiction, without in any way affecting the remaining parts of this Agreement in that jurisdiction or the validity or enforceability of the Agreement as a whole in any other jurisdiction. In addition, the part that is ineffective will be reformed in a mutually agreeable manner so as to as nearly approximate the intent of the Parties as possible. 13.9 Independent Contractors. Each of the Parties is an independent contractor and nothing herein contained will be deemed to constitute the relationship of partners, joint venturers, nor of principal and agent between the Parties. Neither Party will at any time enter into, incur, or hold itself out to Third Parties as having authority to enter into or incur, on behalf of the other Party, any commitment, expense, or liability whatsoever. 13.10 Waiver. No waiver of any term, provision or condition of this Agreement whether by conduct or otherwise in any one or more instances will be deemed to be or construed as a further or continuing waiver of any such term, provision or condition or of any other term, provision or condition of this Agreement. 13.11 Entirety; Amendments. This Agreement, including the Appendices attached hereto, constitutes the full understanding of the Parties and a complete and exclusive statement of the terms of their agreement with respect to the specific subject matter hereof and supersedes all prior written or oral agreements, arrangements, term sheets and understandings related to the specific subject matter hereof, including, but not limited to, that certain Binding Term Sheet between Catalent and Lisata dated August 25, 2025). No terms, conditions, understandings or agreements purporting to modify or vary the terms thereof will be binding unless hereafter made in a written instrument referencing amendment of this Agreement and signed by each of the Parties. 13.12 Interpretation. The captions and headings to this Agreement are for convenience only and are to be of no force or effect in construing or interpreting any of the provisions of this Agreement. Unless specified to the contrary, references to Articles, Sections or Appendices mean the particular Articles, Sections or Appendices to this Agreement and references to this Agreement include all Appendices hereto. Unless context otherwise clearly requires, whenever used in this Agreement: (a) the words “include” or “including” will be construed as incorporating, also, “but not limited to” or “without limitation;” (b) the word “day” or “year” means a calendar day or year unless otherwise specified; (c) the word “notice” will mean notice in writing (whether or not specifically stated) and will include notices, consents, approvals and other written communications contemplated under this Agreement; (d) the words “hereof,” “herein,” “hereby” and derivative or similar words refer to this Agreement (including any Appendices); (e) the word “or” will be construed as the inclusive meaning identified with the phrase “and/or”; (f) provisions that require that a Party, the Parties or a committee hereunder “agree,” “consent” or “approve” or the like will require that such agreement, consent or approval be specific and in writing, whether by written agreement, letter, approved minutes or otherwise; (g) words of any gender include the other gender; (h) words using the singular or plural number also include the plural or singular number, respectively; (i) references to any Applicable Laws, or article, section or other division thereof, will be deemed to include the then-current

25 amendments thereto or any replacement Applicable Laws thereto; and (j) neither Party or its Affiliates will be deemed to be acting “on behalf of” or “under authority of” the other Party under this Agreement. 13.13 Headings. All headings in this Agreement are for convenience of reference only and will not affect the interpretation of this Agreement. 13.14 Counterparts. This Agreement and any amendment hereto may be executed in any number of counterparts (which may be exchanged by emails of .PDF copies, DocuSign or other electronic means), each of which will for all purposes be deemed an original and all of which will constitute the same instrument. (signature page follows)

IN WITNESS WHEREOF, the Parties have executed this Agreement by their duly authorized representatives. LISATA THERAPEUTICS, INC. REDWOOD BIOSCIENCE, INC. By: ____________________________ By:_________________________________ Name: David J. Mazzo, PhD Name: Title: President and Chief Executive Officer Title: [signature page to Non-Exclusive License Agreement]
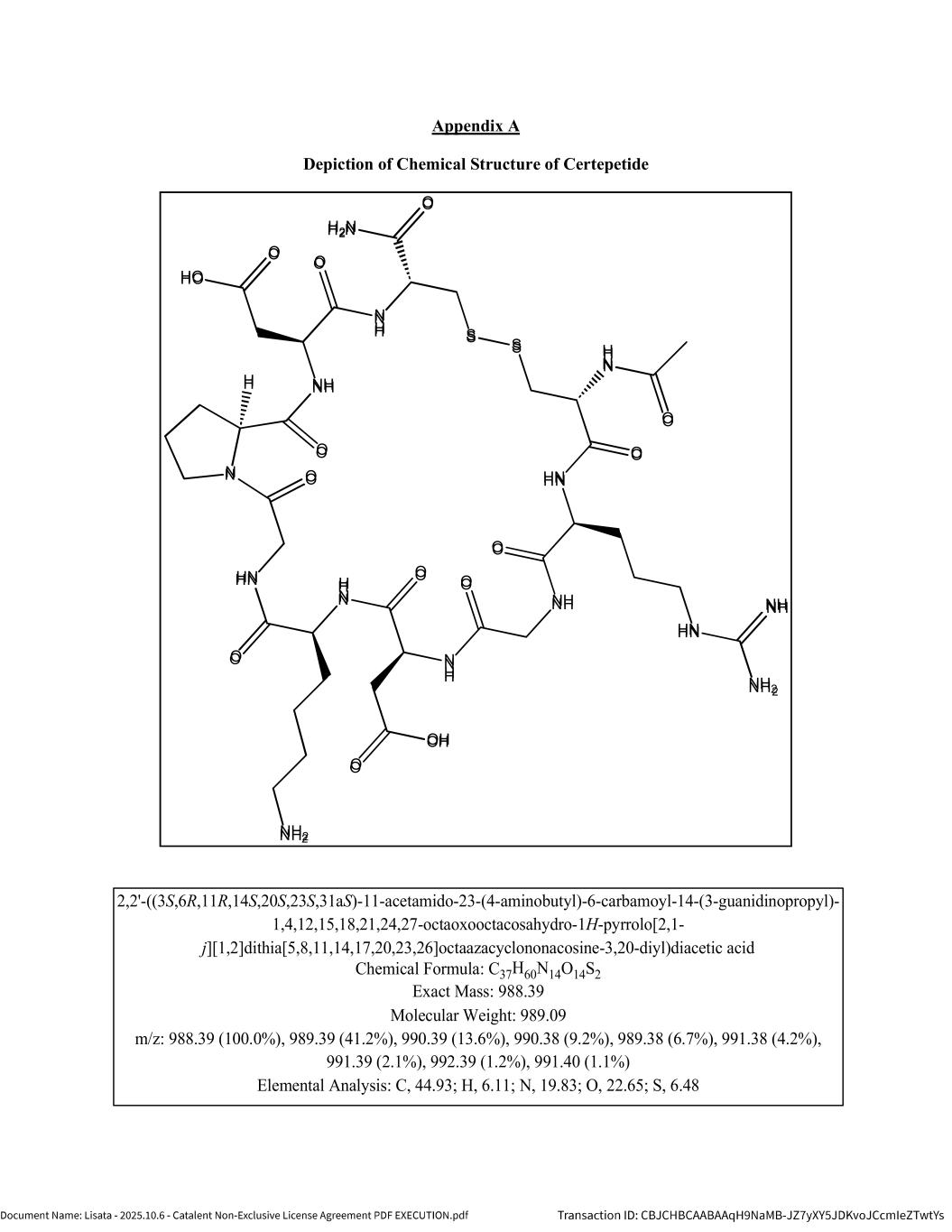
Appendix A Depiction of Chemical Structure of Certepetide O H N HN H N N H NH HN S S N H NH HO O O H2N O H N O O HN NH2 NH O O OH O O NH2 O O 2 NH2 2,2'-((3S,6R,11R,14S,20S,23S,31aS)-11-acetamido-23-(4-aminobutyl)-6-carbamoyl-14-(3-guanidinopropyl)- 1,4,12,15,18,21,24,27-octaoxooctacosahydro-1H-pyrrolo[2,1- j][1,2]dithia[5,8,11,14,17,20,23,26]octaazacyclononacosine-3,20-diyl)diacetic acid Chemical Formula: C37H60N14O14S2 Exact Mass: 988.39 Molecular Weight: 989.09 m/z: 988.39 (100.0%), 989.39 (41.2%), 990.39 (13.6%), 990.38 (9.2%), 989.38 (6.7%), 991.38 (4.2%), 991.39 (2.1%), 992.39 (1.2%), 991.40 (1.1%) Elemental Analysis: C, 44.93; H, 6.11; N, 19.83; O, 22.65; S, 6.48
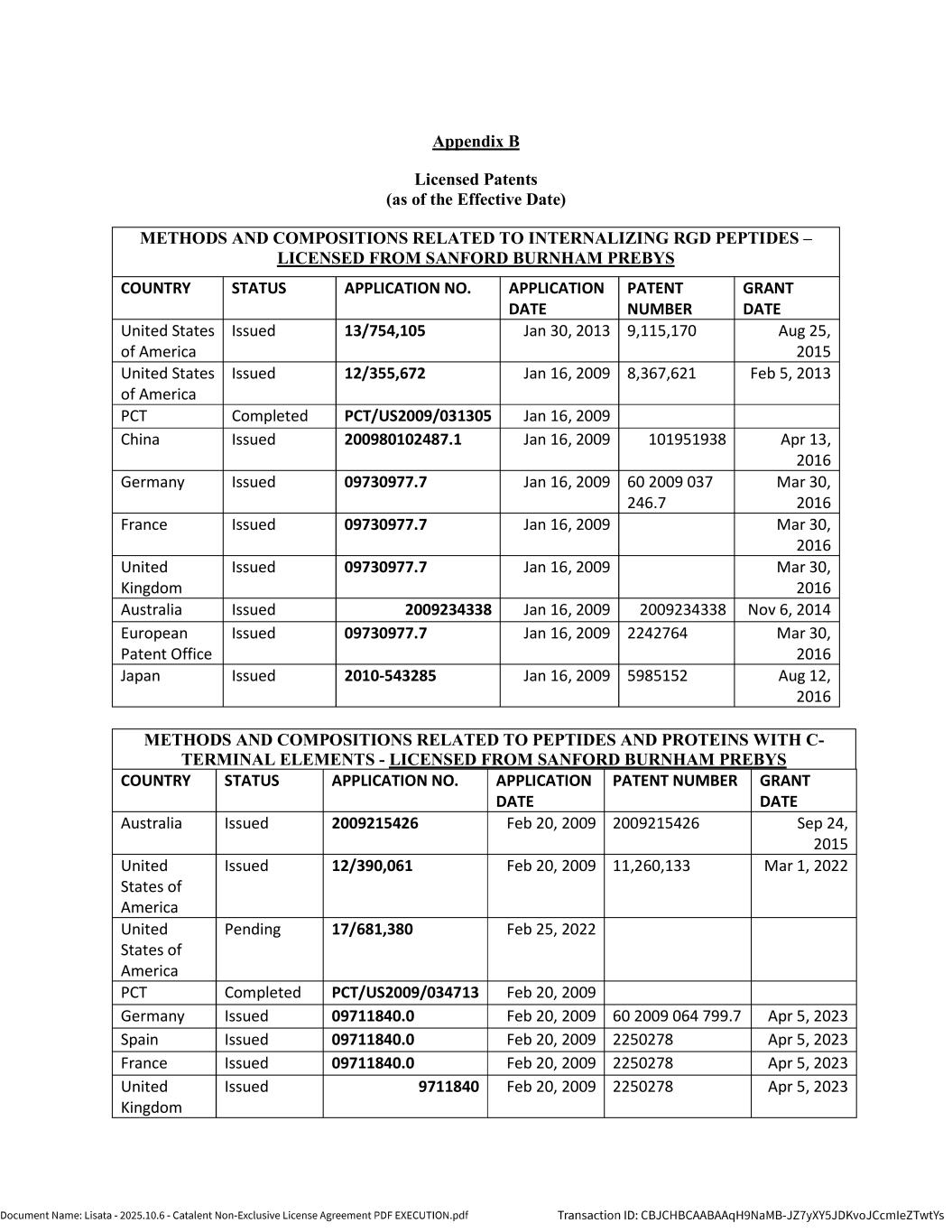
Appendix B Licensed Patents (as of the Effective Date) METHODS AND COMPOSITIONS RELATED TO INTERNALIZING RGD PEPTIDES – LICENSED FROM SANFORD BURNHAM PREBYS COUNTRY STATUS APPLICATION NO. APPLICATION DATE PATENT NUMBER GRANT DATE United States of America Issued 13/754,105 Jan 30, 2013 9,115,170 Aug 25, 2015 United States of America Issued 12/355,672 Jan 16, 2009 8,367,621 Feb 5, 2013 PCT Completed PCT/US2009/031305 Jan 16, 2009 China Issued 200980102487.1 Jan 16, 2009 101951938 Apr 13, 2016 Germany Issued 09730977.7 Jan 16, 2009 60 2009 037 246.7 Mar 30, 2016 France Issued 09730977.7 Jan 16, 2009 Mar 30, 2016 United Kingdom Issued 09730977.7 Jan 16, 2009 Mar 30, 2016 Australia Issued 2009234338 Jan 16, 2009 2009234338 Nov 6, 2014 European Patent Office Issued 09730977.7 Jan 16, 2009 2242764 Mar 30, 2016 Japan Issued 2010-543285 Jan 16, 2009 5985152 Aug 12, 2016 METHODS AND COMPOSITIONS RELATED TO PEPTIDES AND PROTEINS WITH C- TERMINAL ELEMENTS - LICENSED FROM SANFORD BURNHAM PREBYS COUNTRY STATUS APPLICATION NO. APPLICATION DATE PATENT NUMBER GRANT DATE Australia Issued 2009215426 Feb 20, 2009 2009215426 Sep 24, 2015 United States of America Issued 12/390,061 Feb 20, 2009 11,260,133 Mar 1, 2022 United States of America Pending 17/681,380 Feb 25, 2022 PCT Completed PCT/US2009/034713 Feb 20, 2009 Germany Issued 09711840.0 Feb 20, 2009 60 2009 064 799.7 Apr 5, 2023 Spain Issued 09711840.0 Feb 20, 2009 2250278 Apr 5, 2023 France Issued 09711840.0 Feb 20, 2009 2250278 Apr 5, 2023 United Kingdom Issued 9711840 Feb 20, 2009 2250278 Apr 5, 2023
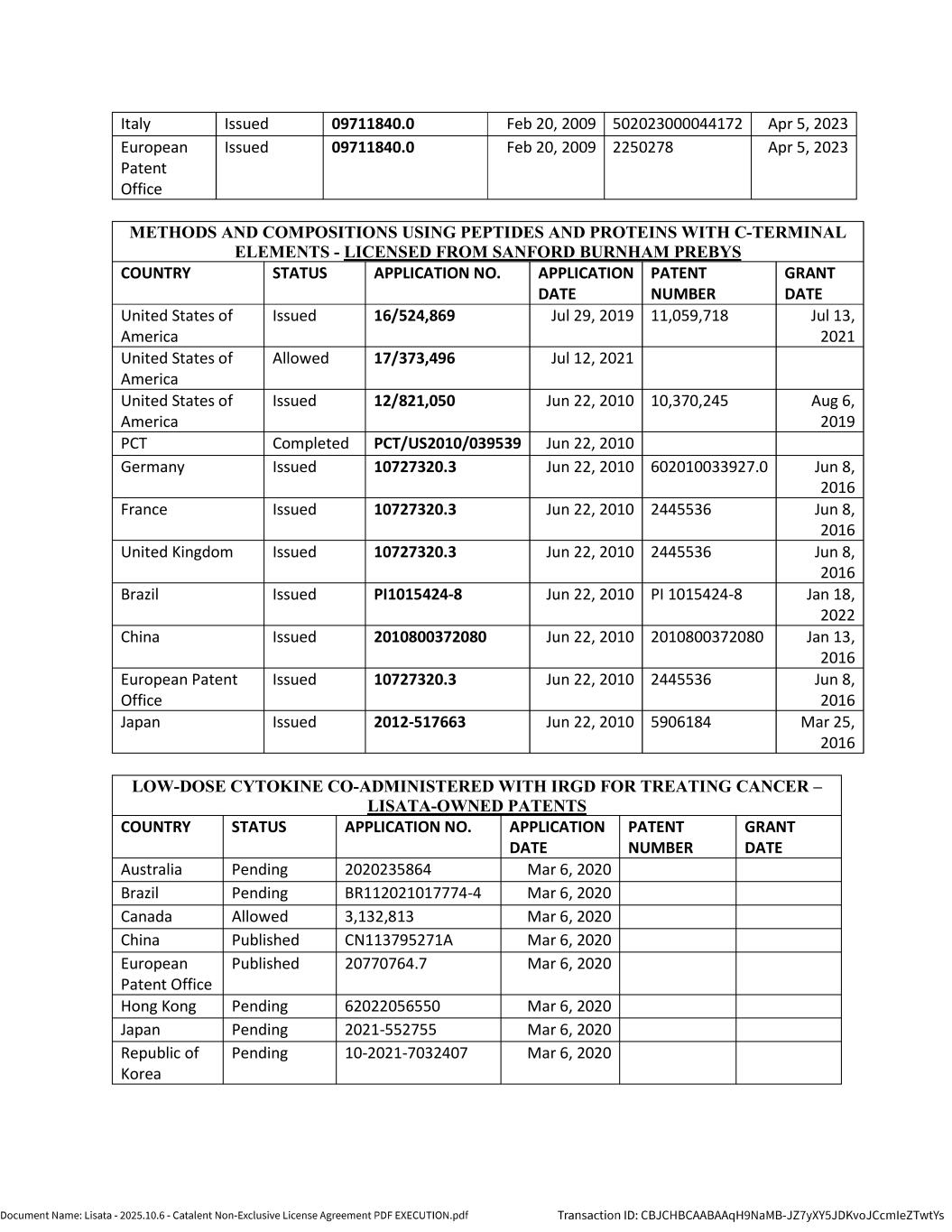
Italy Issued 09711840.0 Feb 20, 2009 502023000044172 Apr 5, 2023 European Patent Office Issued 09711840.0 Feb 20, 2009 2250278 Apr 5, 2023 METHODS AND COMPOSITIONS USING PEPTIDES AND PROTEINS WITH C-TERMINAL ELEMENTS - LICENSED FROM SANFORD BURNHAM PREBYS COUNTRY STATUS APPLICATION NO. APPLICATION DATE PATENT NUMBER GRANT DATE United States of America Issued 16/524,869 Jul 29, 2019 11,059,718 Jul 13, 2021 United States of America Allowed 17/373,496 Jul 12, 2021 United States of America Issued 12/821,050 Jun 22, 2010 10,370,245 Aug 6, 2019 PCT Completed PCT/US2010/039539 Jun 22, 2010 Germany Issued 10727320.3 Jun 22, 2010 602010033927.0 Jun 8, 2016 France Issued 10727320.3 Jun 22, 2010 2445536 Jun 8, 2016 United Kingdom Issued 10727320.3 Jun 22, 2010 2445536 Jun 8, 2016 Brazil Issued PI1015424-8 Jun 22, 2010 PI 1015424-8 Jan 18, 2022 China Issued 2010800372080 Jun 22, 2010 2010800372080 Jan 13, 2016 European Patent Office Issued 10727320.3 Jun 22, 2010 2445536 Jun 8, 2016 Japan Issued 2012-517663 Jun 22, 2010 5906184 Mar 25, 2016 LOW-DOSE CYTOKINE CO-ADMINISTERED WITH IRGD FOR TREATING CANCER – LISATA-OWNED PATENTS COUNTRY STATUS APPLICATION NO. APPLICATION DATE PATENT NUMBER GRANT DATE Australia Pending 2020235864 Mar 6, 2020 Brazil Pending BR112021017774-4 Mar 6, 2020 Canada Allowed 3,132,813 Mar 6, 2020 China Published CN113795271A Mar 6, 2020 European Patent Office Published 20770764.7 Mar 6, 2020 Hong Kong Pending 62022056550 Mar 6, 2020 Japan Pending 2021-552755 Mar 6, 2020 Republic of Korea Pending 10-2021-7032407 Mar 6, 2020
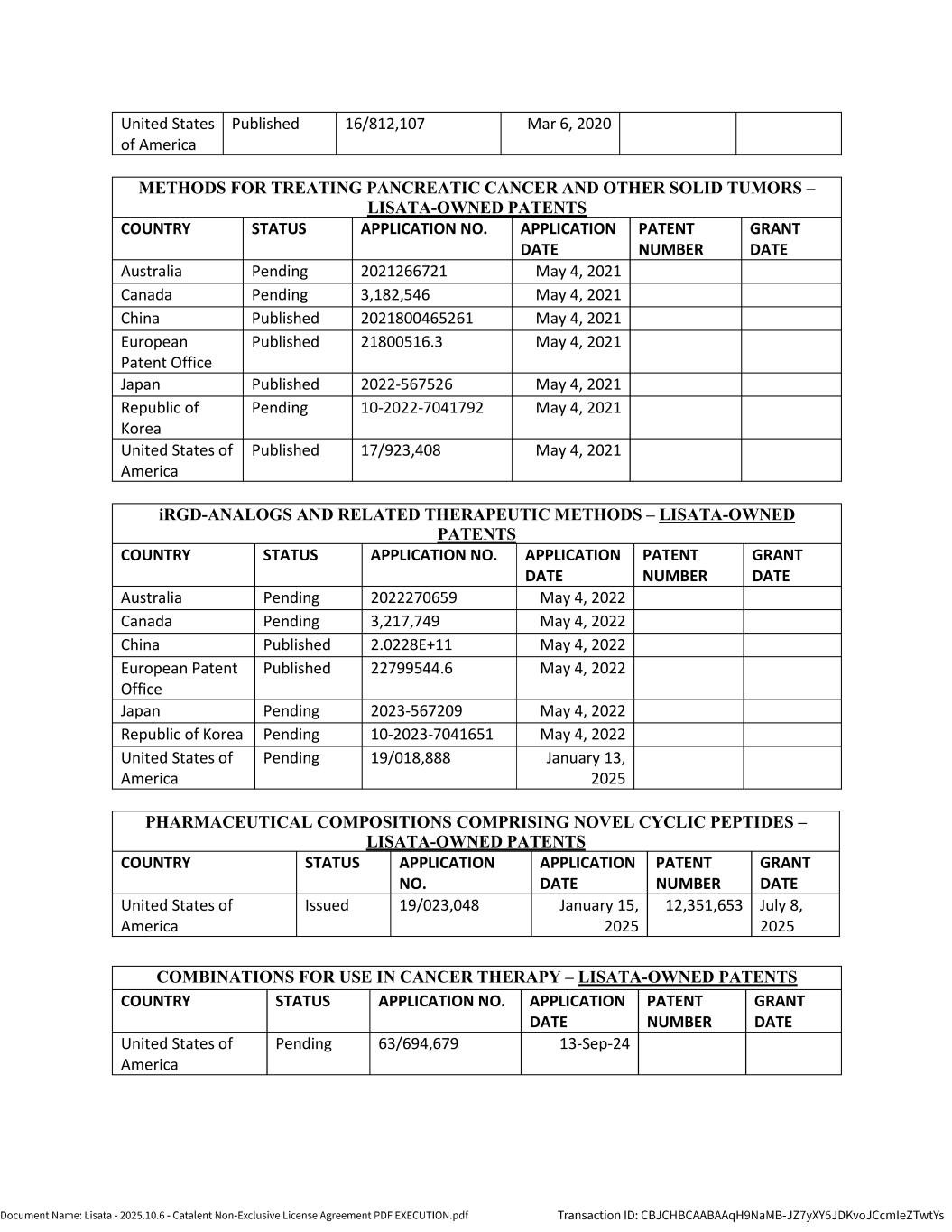
United States of America Published 16/812,107 Mar 6, 2020 METHODS FOR TREATING PANCREATIC CANCER AND OTHER SOLID TUMORS – LISATA-OWNED PATENTS COUNTRY STATUS APPLICATION NO. APPLICATION DATE PATENT NUMBER GRANT DATE Australia Pending 2021266721 May 4, 2021 Canada Pending 3,182,546 May 4, 2021 China Published 2021800465261 May 4, 2021 European Patent Office Published 21800516.3 May 4, 2021 Japan Published 2022-567526 May 4, 2021 Republic of Korea Pending 10-2022-7041792 May 4, 2021 United States of America Published 17/923,408 May 4, 2021 iRGD-ANALOGS AND RELATED THERAPEUTIC METHODS – LISATA-OWNED PATENTS COUNTRY STATUS APPLICATION NO. APPLICATION DATE PATENT NUMBER GRANT DATE Australia Pending 2022270659 May 4, 2022 Canada Pending 3,217,749 May 4, 2022 China Published 2.0228E+11 May 4, 2022 European Patent Office Published 22799544.6 May 4, 2022 Japan Pending 2023-567209 May 4, 2022 Republic of Korea Pending 10-2023-7041651 May 4, 2022 United States of America Pending 19/018,888 January 13, 2025 PHARMACEUTICAL COMPOSITIONS COMPRISING NOVEL CYCLIC PEPTIDES – LISATA-OWNED PATENTS COUNTRY STATUS APPLICATION NO. APPLICATION DATE PATENT NUMBER GRANT DATE United States of America Issued 19/023,048 January 15, 2025 12,351,653 July 8, 2025 COMBINATIONS FOR USE IN CANCER THERAPY – LISATA-OWNED PATENTS COUNTRY STATUS APPLICATION NO. APPLICATION DATE PATENT NUMBER GRANT DATE United States of America Pending 63/694,679 13-Sep-24

Appendix C Form of Joint Press Release

Lisata Therapeutics and Catalent Announce Global Antibody-Drug Conjugate (ADC) License Agreement Catalent gains global rights to evaluate certepetide and its analogs for use as SMARTag® payloads across multiple ADCs designed to address difficult-to-treat-diseases preclinical study results provide proof-of-concept support for leveraging certepetide- related biology through its use as an ADC payload TAMPA, Fla incorporate into s s developed using Catalent's SMARTag® Certepetide, a proprietary, internalizing RGD (arginyl-glycyl-aspartic acid) peptide, is being tested by Lisata as a cancer therapeutic to be used in combination with other anti-cancer agents to enhance tumor targeting and penetration and improve treatment outcomes. e gains certepetide and its analogs, including the ability to partner with third parties. As part of this collaboration, Catalent will ha in studies receive over $10 million in tiered study initiation milestone payments plus revenue sharing on future sales and partnerships. Preclinical work supporting incorporation of iRGD peptides into the SMARTag® We are excited about the opportunity to explore iRGD biology as it relates to ADC delivery to the tumor microenvironment. Early data suggest that incorporating iRGD peptides into ADCs improves efficacy and pharmacokinetics, leading us to be optimistic about the potential of iRGD as a novel payload class said Penelope Drake, Head of R&D, Bioconjugates at Catalent. About Lisata Therapeutics Lisata Therapeutics is a clinical-stage pharmaceutical company dedicated to the discovery, development and commercialization of innovative therapies for the treatment of advanced solid tumors and other certepetide, is an investigational drug designed to activate a novel uptake pathway that allows co-administered or tethered anti-cancer drugs

to selectively target and penetrate solid tumors more effectively. Lisata has already established noteworthy commercial and R&D partnerships based on its CendR Platform® technology. The Company expects to announce numerous milestones over the next 1.5 years and believes that its projected capital will fund operations through the fourth quarter of 2026, encompassing anticipated data milestones from its ongoing and planned clinical trials. For a comprehensive overview of certepetide's mechanism of action, please view our informative short film. For more information on the Company, please visit www.lisata.com. About Catalent Catalent, Inc. is a leading global contract development and manufacturing organization (CDMO) whose mission is to develop, manufacture, and supply products that help people live better and healthier lives. Catalent is dedicated to delivering unparalleled service to pharma, biotech, and consumer health customers, supporting product development, launch, and full life-cycle supply. With time-tested experience in development sciences, delivery technologies, and multi-modality manufacturing, Catalent supports the acceleration of development programs and the launch of more than a hundred new products every year. Powered by thousands of scientists and technicians and the latest technology platforms at more than 40 global sites, Catalent supplies billions of doses of life-enhancing and life- saving treatments for patients annually. For more information, visit www.catalent.com. Forward-Looking Statements

Catalent Contact:

Media Contact: Laura Hortas 609-240-7025 media@catalent.com