
Q4 and 2025 Financial Results February 24, 2026 1

Disclaimer 2 Forward-looking Statements This presentation contains “forward-looking” statements as defined in the Private Securities Litigation Reform Act of 1995, which are statements that are not historical facts and relate to future conditions, events, or results. These statements are based on management's current expectations or predictions of future conditions, events, or results based on various assumptions and management's estimates of trends and economic factors in the markets in which we are active, as well as our business plans. Words such as “expects,” “anticipates,” “objectives,” “intends,” “plans,” “believes,” “seeks,” “estimates,” “projects,” “forecasts,” “continue,” “may,” “should,” “will,” “goals,” and variations of such words and similar expressions are intended to identify such forward-looking statements. Forward-looking statements include, but are not limited to, statements related to: clinical development activities, including expansion into prostate applications; commercial growth initiatives, including planned expansion of breast and extremities sales specialists; market development opportunities; expectations regarding disciplined, profitable growth and margin improvement; financial guidance and outlook for 2026, including projected revenue growth, net cash flow, gross margins, and other operating performance metrics; and statements regarding our training and education initiatives, reimbursement and market access efforts, and research and development activities. Actual results or events could differ materially from those described in any forward-looking statements as a result of various factors, including, without limitation, risks related to global supply chain conditions, inflationary pressures, hospital staffing challenges, product development and product potential, clinical enrollment timing and outcomes, regulatory processes and approvals, financial performance, sales growth, surgeon and product adoption, market awareness of our products, data validation, our visibility at and sponsorship of conferences and educational events, geopolitical and macroeconomic conditions, including armed conflicts and government actions or policies that may affect our business, tax position, or regulatory processes, as well as those risk factors described under Part I, Item 1A., “Risk Factors,” of our most recent Annual Report on Form 10-K, subsequent Quarterly Reports on Form 10-Q, and other filings made from time to time with the Securities and Exchange Commission. Forward-looking statements are not a guarantee of future performance, and actual results may differ materially from those projected. Forward-looking statements speak only as of the date they are made and, except as required by applicable law, we assume no responsibility to publicly update or revise any forward-looking statements. About Non-GAAP Financial Measures To supplement our condensed consolidated financial statements, we use the non-GAAP financial measures of EBITDA, which measures earnings before interest, income taxes, depreciation and amortization, EBITDA margin, Adjusted EBITDA, which further excludes non-cash stock compensation expense, and Adjusted EBITDA margin. We also use the non-GAAP financial measures of Adjusted Net Income and Adjusted Net Income Per Common Share - diluted which excludes noncash stock compensation expense from Net (Loss) Income and Net (Loss) Income Per Common Share - diluted, respectively. We also use the Operational Cashflow metric, which corresponds to Net change in cash, cash equivalents, restricted cash, and investments, less cashflow from issuance or repayment of long-term debt. These non-GAAP measures are not based on any comprehensive set of accounting rules or principles and should not be considered a substitute for, or superior to, financial measures calculated in accordance with GAAP and may be different from non-GAAP measures used by other companies. In addition, these non-GAAP measures should be read in conjunction with our financial statements prepared in accordance with GAAP. The reconciliations of the non-GAAP measures to the most directly comparable financial measures calculated and presented in accordance with GAAP should be carefully evaluated. We use these non-GAAP financial measures for financial and operational decision-making and as a means to evaluate period-to-period comparisons. We believe that these non-GAAP financial measures provide meaningful supplemental information regarding our performance and that both management and investors benefit from referring to these non-GAAP financial measures in assessing our performance and when planning, forecasting, and analyzing future periods. We believe these non-GAAP financial measures are useful to investors because (1) they allow for greater transparency with respect to key metrics used by management in its financial and operational decision-making and (2) they are used by our institutional investors and the analyst community to help them analyze the performance of our business, the Company’s cash available for operations, and the Company’s ability to meet future capital expenditure and working capital requirements.

3 Michael Dale President and Chief Executive Officer Q4 & 2025 Business Highlights and 2026 Goals

Agenda 4 Q4 and 2025 Financials and 2026 Guidance Lindsey Hartley, Chief Financial Officer Q&A Michael Dale, Lindsey Hartley, Jens Kemp, Chief Marketing Officer Rick Ditto, VP Global Health Economic, Reimbursement & Policy Q4 and 2025 Business Highlights and 2026 Goals Michael Dale, President and Chief Executive Officer

Strategic Priorities 01 GROWTH 15–20% Revenue CAGR + Operating Leverage 02 MARKET DEVELOPMENT Elective & Planned Procedures + Prostate market development 03 COMMERCIAL EXPANSION Infrastructure and Sales Force expansion 04 COMMERCIAL EXCELLENCE Continuous business model and customer creation process optimization by market 05 STANDARD OF CARE Clinical evidence generation for societal support, standard of care & coverage requirements 06 INNOVATION Product development to drive better benefit versus risk profiles in nerve care 5

6 Strategic Priorities 2025 Business Highlights 0 1 G R OW TH 15–20% Revenue CAGR + Operating Leverage Q4 Revenue $59.9M, +21.3% YoY Full-Year Revenue $225.2M, +20.2% YoY Capital Structure Raised $133.3M; retired $69.7M term loan 2026 Target Disciplined profitable growth; improving margins 0 2 M A R K E T D E V E L O PM EN T Elective & Planned + Prostate Extremities Solid traumatic & chronic growth; most mature market OMF / H&N High double-digit growth; quality-of-life recognition growing Breast Fast-growing; accelerating Resensation adoption Prostate 100+ procedures; 10 sites; surgical technique standardized 2026 Prostate Meaningful clinical signals expected in 2H 2026 0 3 C OMME R C I A L E X P A N S I O N Infrastructure + Sales Force Growth Breast 21 reps, 2 regional directors Extremities 117 reps, 15 regional directors OMF / H&N 3 field-based market development managers Prostate Added 3 clinical development managers and 1 director 2026 Breast / Ext. Grow to ~30 breast reps; ~130 extremity reps 6

7 Strategic Priorities 2025 Business Highlights 2026 2026 2026 0 4 C OMME R C I A L E X C E L L E N C E HiPo Accounts, Productivity & Education HiPo Revenue 61% of growth from HiPo accounts Productivity +21% avg. HiPo account productivity Active Accounts 679 HiPo accounts; +131 active surgeons Education Exceeded surgeon training targets across all markets HiPo & Training 60% growth from HiPo; +18% productivity; 100+ new surgeons 0 5 S T A N D A R D O F C A R E Evidence, Coverage & Avance® FDA BLA Exclusivity 12 years of U.S. market exclusivity Avance® BLA Approved First & only FDA-approved biologic for peripheral nerve repair Societies AAHS & ASRM recognize allograft as standard of care (1) Coverage +19.8M lives added; commercial coverage now above 65% Payer & Coverage Pursue near-universal US coverage (est. 2H 2028) 0 6 I N NO V A T I O N R&D + Therapeutic Reconstruction Program Updates Detailed updates on individual R&D programs in 2H 2026 Ease of Coaptation R&D focused on faster, more consistent nerve coaptation Chronic Injuries Advancing non-transected and chronic nerve repair solutions Therapeutic Reconstruction Next-gen technologies to improve nerve regeneration Clinical Studies BLA enables prioritized breast & mixed/motor nerve studies 7 (1) The American Association of Hand Surgery (“AAHS”) and the American Society for Reconstructive Microsurgery (“ASRM”) released official position statements recognizing nerve allograft as a standard medical practice option for the treatment of peripheral nerve defects during the third quarter of 2025.

8 Lindsey Hartley Chief Financial Officer Q4 & 2025 Financials and 2026 Guidance Discussion
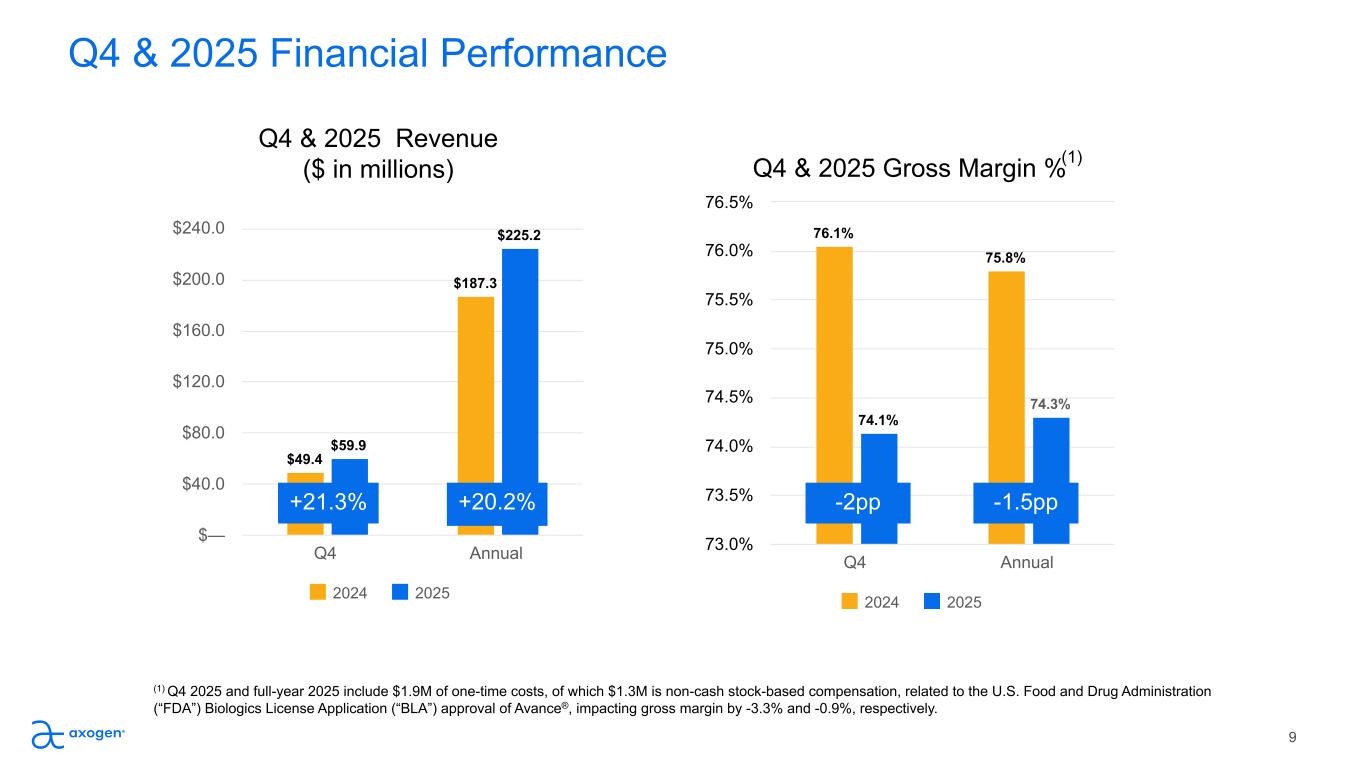
Q4 & 2025 Financial Performance 9 Q4 & 2025 Revenue ($ in millions) $49.4 $187.3 $59.9 $225.2 2024 2025 Q4 Annual $— $40.0 $80.0 $120.0 $160.0 $200.0 $240.0 Q4 & 2025 Gross Margin % 76.1% 75.8% 74.1% 74.3% 2024 2025 Q4 Annual 73.0% 73.5% 74.0% 74.5% 75.0% 75.5% 76.0% 76.5% -2pp+21.3% (1) Q4 2025 and full-year 2025 include $1.9M of one-time costs, of which $1.3M is non-cash stock-based compensation, related to the U.S. Food and Drug Administration (“FDA”) Biologics License Application (“BLA”) approval of Avance®, impacting gross margin by -3.3% and -0.9%, respectively. (1) -1.5pp+20.2%
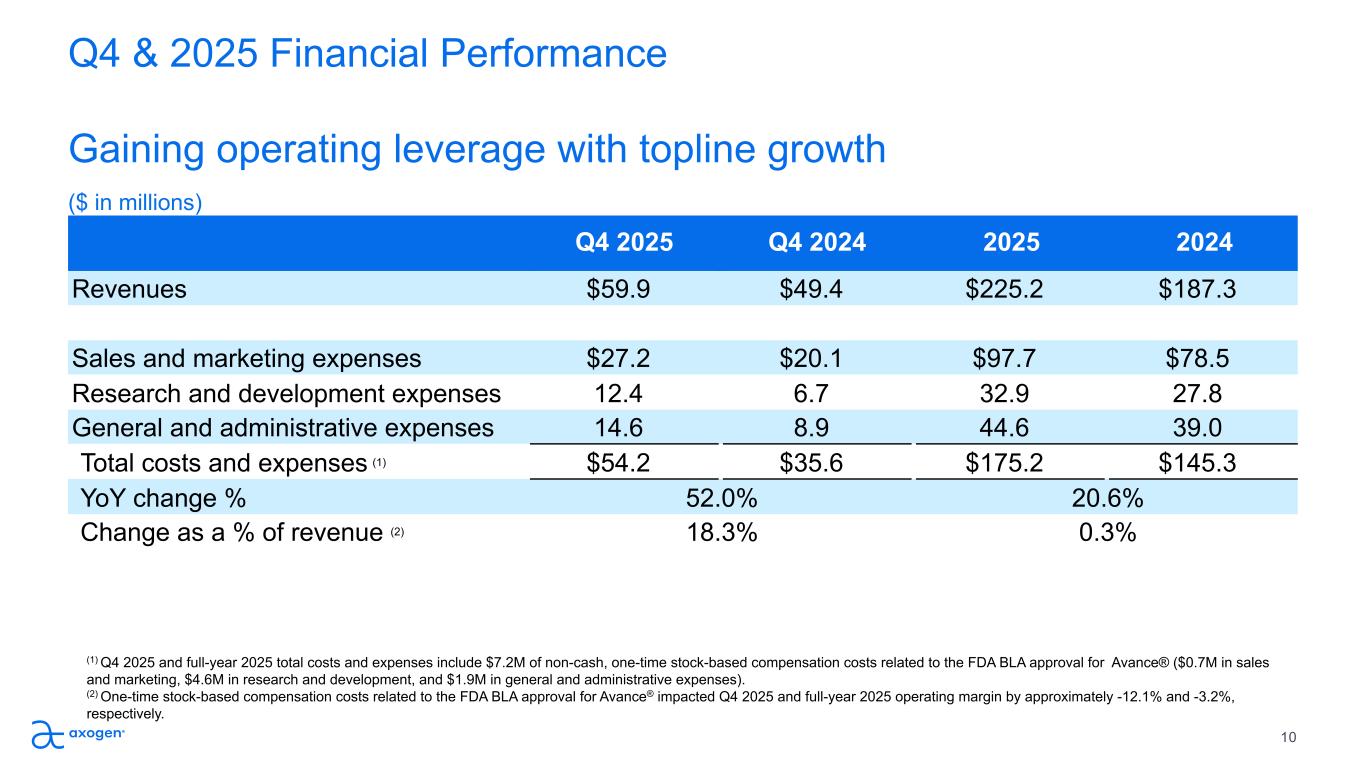
Q4 & 2025 Financial Performance Gaining operating leverage with topline growth ($ in millions) 10 Q4 2025 Q4 2024 2025 2024 Revenues $59.9 $49.4 $225.2 $187.3 Sales and marketing expenses $27.2 $20.1 $97.7 $78.5 Research and development expenses 12.4 6.7 32.9 27.8 General and administrative expenses 14.6 8.9 44.6 39.0 Total costs and expenses (1) $54.2 $35.6 $175.2 $145.3 YoY change % 52.0% 20.6% Change as a % of revenue (2) 18.3% 0.3% (1) Q4 2025 and full-year 2025 total costs and expenses include $7.2M of non-cash, one-time stock-based compensation costs related to the FDA BLA approval for Avance® ($0.7M in sales and marketing, $4.6M in research and development, and $1.9M in general and administrative expenses). (2) One-time stock-based compensation costs related to the FDA BLA approval for Avance® impacted Q4 2025 and full-year 2025 operating margin by approximately -12.1% and -3.2%, respectively.
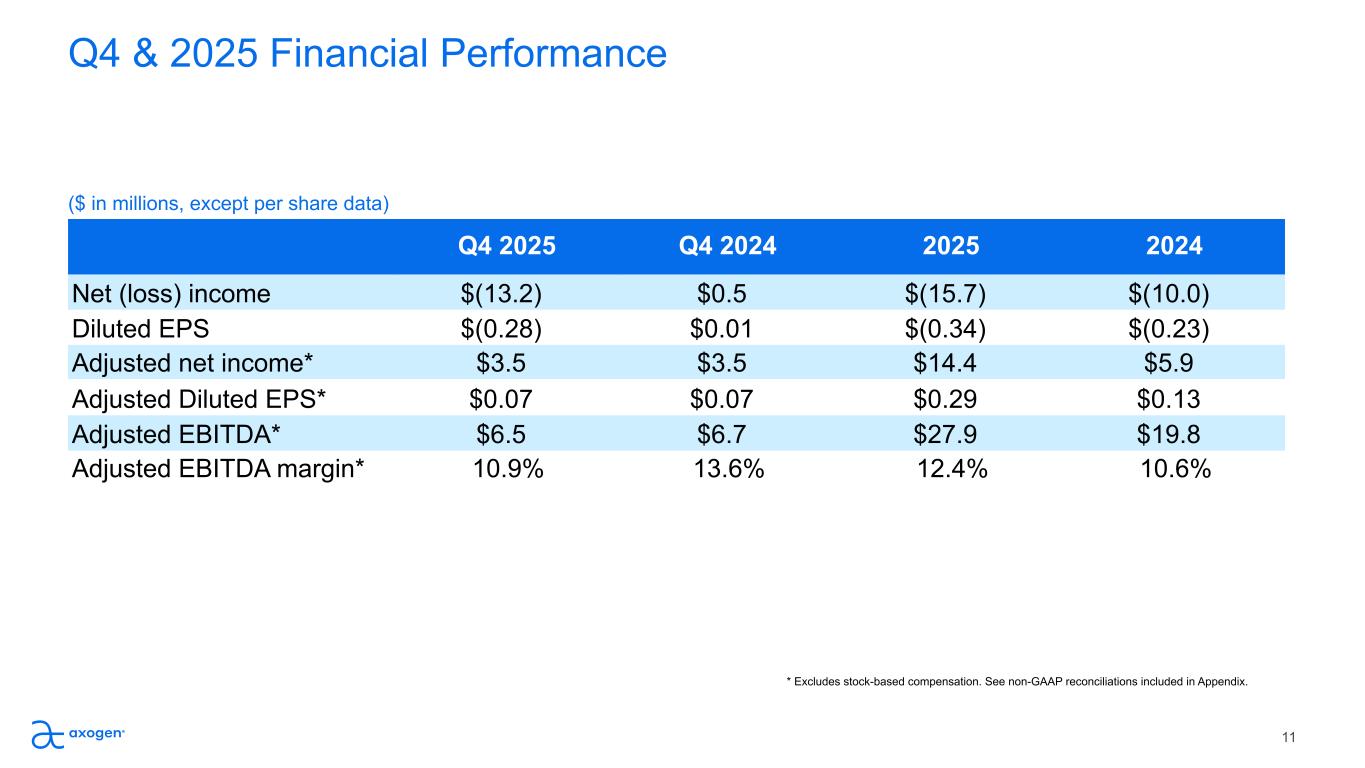
Q4 & 2025 Financial Performance ($ in millions, except per share data) 11 * Excludes stock-based compensation. See non-GAAP reconciliations included in Appendix. Q4 2025 Q4 2024 2025 2024 Net (loss) income $(13.2) $0.5 $(15.7) $(10.0) Diluted EPS $(0.28) $0.01 $(0.34) $(0.23) Adjusted net income* $3.5 $3.5 $14.4 $5.9 Adjusted Diluted EPS* $0.07 $0.07 $0.29 $0.13 Adjusted EBITDA* $6.5 $6.7 $27.9 $19.8 Adjusted EBITDA margin* 10.9% 13.6% 12.4% 10.6%

Q4 and 2025 Financial Performance Operating cash flow ($ in millions) 12 * Cash, cash equivalents, restricted cash, and investments. December 31, 2025 Q4 Change 2025 Change Operational cash* $45.5 +$5.7 +$6.0

Guidance for the Full-Year 2026 Revenue growth of at least 18% or $265.7 million Gross margin of 74% to 76% Net free cash flow positive 13

Michael Dale President and Chief Executive Officer Jens Kemp Chief Marketing Officer Rick Ditto VP, Global Health Economics, Reimbursement & Policy 14 Lindsey Hartley Chief Financial Officer Q&A

Thank you

Appendix 16
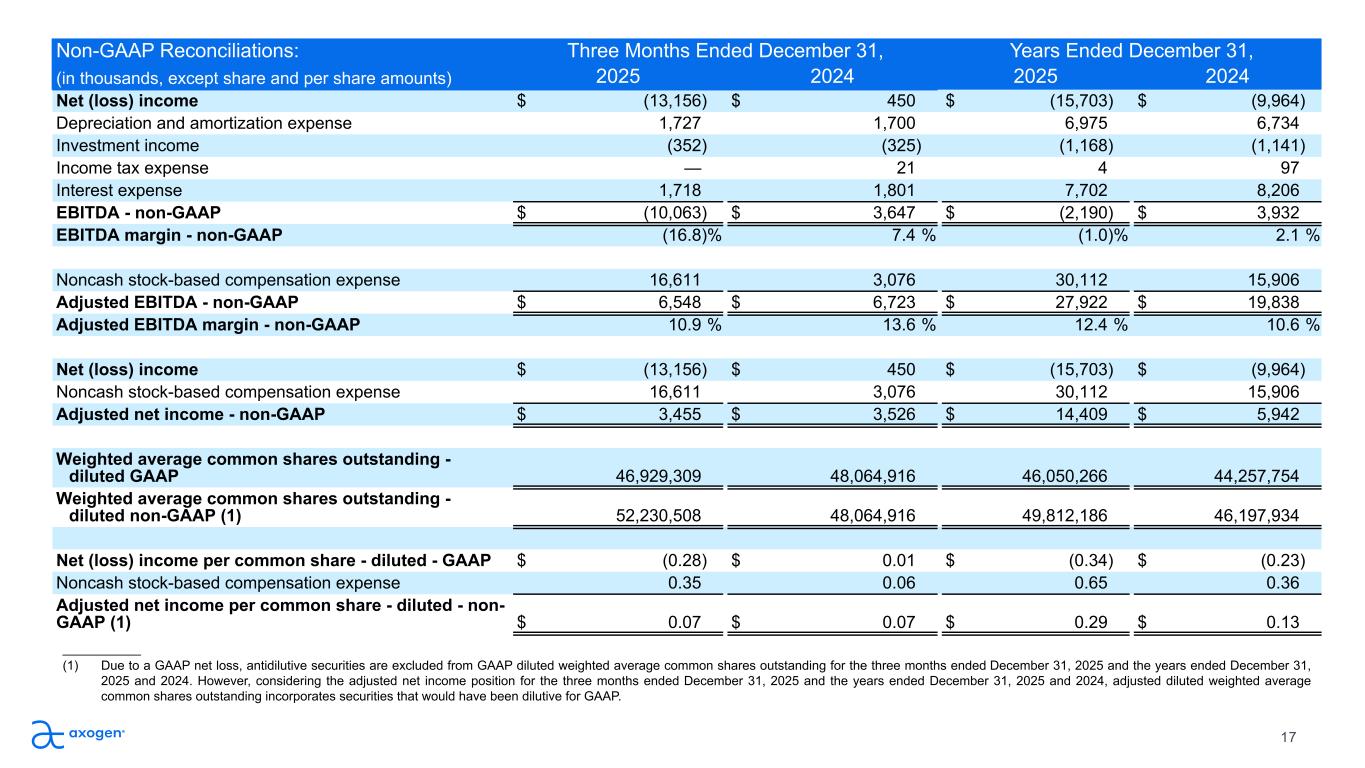
17 Non-GAAP Reconciliations: Three Months Ended December 31, Years Ended December 31, (in thousands, except share and per share amounts) 2025 2024 2025 2024 Net (loss) income $ (13,156) $ 450 $ (15,703) $ (9,964) Depreciation and amortization expense 1,727 1,700 6,975 6,734 Investment income (352) (325) (1,168) (1,141) Income tax expense — 21 4 97 Interest expense 1,718 1,801 7,702 8,206 EBITDA - non-GAAP $ (10,063) $ 3,647 $ (2,190) $ 3,932 EBITDA margin - non-GAAP (16.8) % 7.4 % (1.0) % 2.1 % Noncash stock-based compensation expense 16,611 3,076 30,112 15,906 Adjusted EBITDA - non-GAAP $ 6,548 $ 6,723 $ 27,922 $ 19,838 Adjusted EBITDA margin - non-GAAP 10.9 % 13.6 % 12.4 % 10.6 % Net (loss) income $ (13,156) $ 450 $ (15,703) $ (9,964) Noncash stock-based compensation expense 16,611 3,076 30,112 15,906 Adjusted net income - non-GAAP $ 3,455 $ 3,526 $ 14,409 $ 5,942 Weighted average common shares outstanding - diluted GAAP 46,929,309 48,064,916 46,050,266 44,257,754 Weighted average common shares outstanding - diluted non-GAAP (1) 52,230,508 48,064,916 49,812,186 46,197,934 Net (loss) income per common share - diluted - GAAP $ (0.28) $ 0.01 $ (0.34) $ (0.23) Noncash stock-based compensation expense 0.35 0.06 0.65 0.36 Adjusted net income per common share - diluted - non- GAAP (1) $ 0.07 $ 0.07 $ 0.29 $ 0.13 ___________ (1) Due to a GAAP net loss, antidilutive securities are excluded from GAAP diluted weighted average common shares outstanding for the three months ended December 31, 2025 and the years ended December 31, 2025 and 2024. However, considering the adjusted net income position for the three months ended December 31, 2025 and the years ended December 31, 2025 and 2024, adjusted diluted weighted average common shares outstanding incorporates securities that would have been dilutive for GAAP.
