
COMBINING OUR STRENGTHS GETTING TO KNOW
BIOGEN Confidential - For Internal Integration Planning Purposes Only

Important Information The tender offer described in this communication has not yet
commenced. This communication is for informational purposes only and is neither an offer to purchase nor a solicitation of an offer to sell any Apellis Common Stock or any other securities, nor is it a substitute for the tender offer materials
that Biogen or Purchaser will file with the SEC. The terms and conditions of the tender offer will be published in, and the offer to purchase Apellis Common Stock will be made only pursuant to, the offer document and related offer materials
prepared by Biogen and Purchaser and filed with the SEC in a tender offer statement on Schedule TO at the time the tender offer is commenced. THE TENDER OFFER MATERIALS (INCLUDING AN OFFER TO PURCHASE, A RELATED LETTER OF TRANSMITTAL AND
CERTAIN OTHER TENDER OFFER DOCUMENTS) AND THE SOLICITATION/RECOMMENDATION STATEMENT ON SCHEDULE 14D-9, AS THEY MAY BE AMENDED FROM TIME TO TIME, WILL CONTAIN IMPORTANT INFORMATION. INVESTORS AND APELLIS SECURITYHOLDERS ARE URGED TO READ THESE
DOCUMENTS CAREFULLY WHEN THEY BECOME AVAILABLE BECAUSE THEY WILL CONTAIN IMPORTANT INFORMATION THAT SUCH PERSONS SHOULD CONSIDER BEFORE MAKING ANY DECISION REGARDING TENDERING THEIR COMMON STOCK. The tender offer materials, including the offer
to purchase and the related letter of transmittal and certain other tender offer documents, and the solicitation/recommendation statement (when they become available) and other documents filed with the SEC by Biogen or Apellis, may be obtained
free of charge at the SEC’s website at www.sec.gov or at Biogen’s website at https://www.biogen.com/ or at Apellis’ website at https://investors.apellis.com/news-releases. In addition, Biogen’s tender offer statement and other documents it will
file with the SEC will be available at https://investors.biogen.com/.

Cautionary Note Regarding Forward-Looking Statements This communication contains
forward-looking statements, relating to, among others, statements regarding the expected timetable for completing the proposed transaction, benefits of the proposed transaction, financing of the proposed transaction, costs and other anticipated
financial impacts of the proposed transaction. These forward-looking statements may be accompanied by such words as “aim,” “anticipate,” “assume,” “believe,” “contemplate,” “continue,” “could,” “estimate,” “expect,” “forecast,” “goal,”
“guidance,” “hope,” “intend,” “may,” “objective,” “outlook,” “plan,” “possible,” “potential,” “predict,” “project,” “prospect,” “should,” “target,” “will,” “would” or the negative of these words or other words and terms of similar meaning. Drug
development and commercialization involve a high degree of risk, and only a small number of research and development programs result in commercialization of a product. Results in early-stage clinical trials may not be indicative of full results
or results from later stage or larger scale clinical trials and do not ensure regulatory approval. You should not place undue reliance on these statements. Given their forward-looking nature, these statements involve substantial risks and
uncertainties that may be based on inaccurate assumptions and could cause actual results to differ materially from those reflected in such statements. These forward-looking statements are based on management’s current beliefs and assumptions
and on information currently available to management. Given their nature, we cannot assure that any outcome expressed in these forward-looking statements will be realized in whole or in part. We caution that these statements are subject to
risks and uncertainties, many of which are outside of our control and could cause future events or results to differ materially from those stated or implied in this document, including, among others, the delay or failure of the tender offer
conditions to be satisfied (or waived), including insufficient shares of Apellis common stock being tendered in the tender offer; the timing to consummate the proposed transaction; the risk that the conditions to closing of the proposed
transaction may not be satisfied or that the closing of the proposed transaction otherwise does not occur; the risk that a regulatory approval that may be required to consummate the proposed transaction is not obtained or is obtained subject to
conditions that are not anticipated or conditions that Biogen is not obligated to accept; the diversion of management time on transaction-related issues; expectations regarding regulatory approval of the transaction; results of litigation,
settlements and investigations; actions by third parties, including governmental agencies; global economic conditions; adverse industry conditions; potential business uncertainty, including changes to existing business relationships during the
pendency of the proposed transaction that could affect financial performance; legal proceedings; governmental regulation; the ability to retain management and other personnel; that all or any of the contingent consideration will become payable
on the terms described herein; the accuracy of Biogen’s estimates of the size and characteristics of the markets that may be addressed by its product candidates; Biogen’s ability to increase its manufacturing capabilities for its products and
product candidates; and other economic, business, or competitive factors. and any other risks and uncertainties that are described in other reports we have filed with the U.S. Securities and Exchange Commission, which are available on the SEC’s
website at www.sec.gov. These statements speak only as of the date of this press release and are based on information and estimates available to us at this time. Should known or unknown risks or uncertainties materialize or should underlying
assumptions prove inaccurate, actual results could vary materially from past results and those anticipated, estimated or projected. Investors are cautioned not to put undue reliance on forward-looking statements. A further list and description
of risks, uncertainties and other matters can be found in our Annual Report on Form 10-K for the fiscal year ended December 31, 2025, and in our subsequent reports on Form 10-Q. Except as required by law, we do not undertake any obligation to
publicly update any forward-looking statements whether as a result of any new information, future events, changed circumstances or otherwise.

4 IT’S GREAT TO MEET YOU Today we hope to: Share a little about us Get to know one
another Discover what unites us, as well as the unique strengths we both bring
5 UNLOCKING GREATER IMPACT ADVANCING IMMUNOLOGY Apellis' complement biology
expertise deepens our immunology expertise DELIVERING VALUE Apellis’ talent, expertise and products can enhance Biogen’s growth portfolio and accelerate Biogen’s entry into nephrology REACHING MORE & NEW PATIENTS Together we can serve
more patients in new specialty and rare disease areas with significant unmet needs Apellis brings unique strengths that complement Biogen’s expertise and capabilities
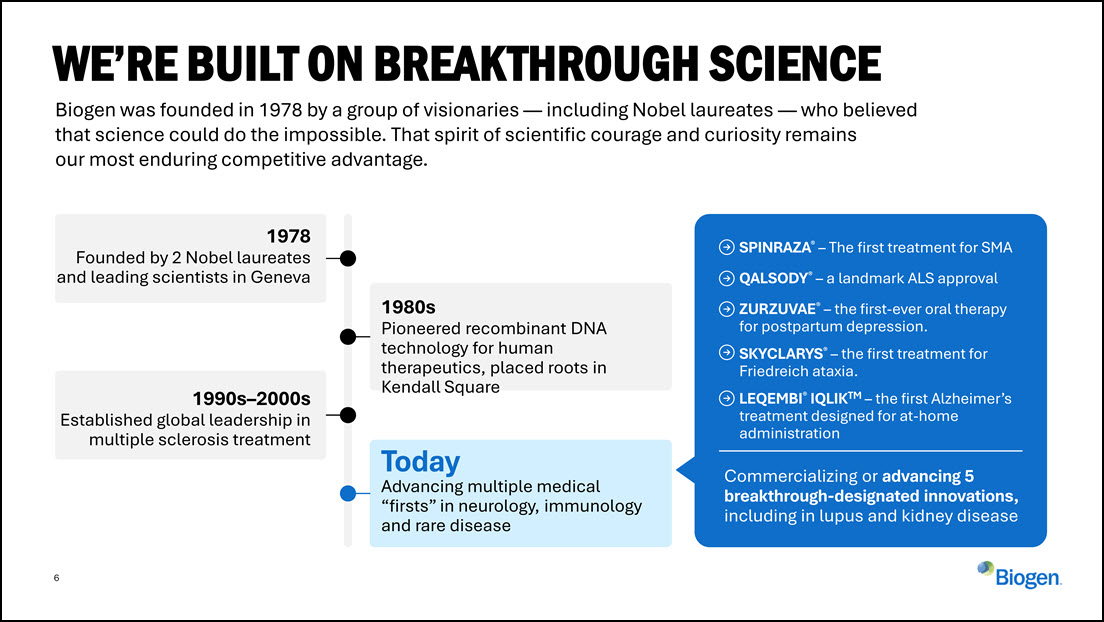
6 Biogen was founded in 1978 by a group of visionaries — including Nobel laureates
— who believed that science could do the impossible. That spirit of scientific courage and curiosity remains our most enduring competitive advantage. WE’RE BUILT ON BREAKTHROUGH SCIENCE 1978 Founded by 2 Nobel laureates and leading
scientists in Geneva 1980s Pioneered recombinant DNA technology for human therapeutics, placed roots in Kendall Square 1990s–2000s Established global leadership in multiple sclerosis treatment Today Advancing multiple medical “firsts”
in neurology, immunology and rare disease SPINRAZA® – The first treatment for SMA QALSODY® – a landmark ALS approval ZURZUVAE® – the first-ever oral therapy for postpartum depression. SKYCLARYS® – the first treatment for Friedreich
ataxia. LEQEMBI® IQLIKTM – the first Alzheimer’s treatment designed for at-home administration Commercializing or advancing 5 breakthrough-designated innovations, including in lupus and kidney disease

7 A LOOK INSIDE BIOGEN ~8K Employees worldwide 2028 New headquarters
in Kendall Square Global network across North America, Europe and Intercontinental region, with 45+ affiliates, and distribution partners in over an additional 60 countries 9 Manufacturing sites 8 in North Carolina, U.S., and 1 in
Solothurn, Switzerland 100+ Countries receive our products $9.89B Revenue in 2025

8 Kai Living with Multiple Sclerosis WE’RE COMMERCIALIZING PRODUCTS IN THE MOST
COMPLEX AND DEVASTATING DISEASES Cleo and Norma Living with Alzheimer’s Disease Elizabeth Living with Friedreich Ataxia 1 in 3 seniors die with AD or another dementia ALZHEIMER’S DISEASE #1 most common neurodegenerative
disease >55 million patients worldwide MULTIPLE SCLEROSIS 2.8 million estimated number of people living with MS worldwide >1 million treated patients, but no ability to completely halt or reverse disease progression POSTPARTUM
DEPRESSION 1 in 8 mothers report experiencing symptoms of PPD 500k estimated number of women in the U.S. who experience PPD ~50% of all PPD cases may go undiagnosed ~1 in 11k live births affected 85% of people living with SMA develop
symptoms after infancy AMYOTROPHIC LATERAL SCLEROSIS ~352,000 people living with ALS/motor neuron disease worldwide 3- to 5-year life expectancy from symptom onset ~15% of cases are genetic ALS FRIEDREICH ATAXIA ~15k Estimated number
of people living with FA worldwide 37 years average life expectancy SPINAL MUSCULAR ATROPHY
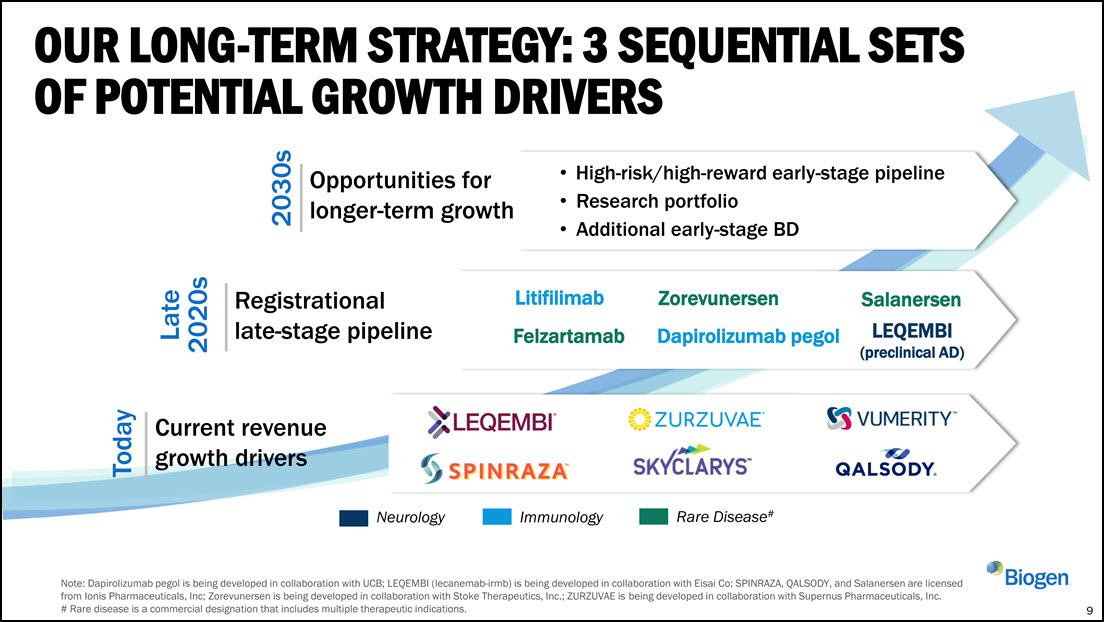
Litifilimab Felzartamab Zorevunersen Dapirolizumab pegol High-risk/high-reward
early-stage pipeline Research portfolio Additional early-stage BD 2030s Late 2020s Today Opportunities for longer-term growth Registrational late-stage pipeline Current revenue growth drivers OUR LONG-TERM STRATEGY: 3 SEQUENTIAL
SETS OF POTENTIAL GROWTH DRIVERS Salanersen LEQEMBI (preclinical AD) 9 Note: Dapirolizumab pegol is being developed in collaboration with UCB; LEQEMBI (lecanemab-irmb) is being developed in collaboration with Eisai Co; SPINRAZA, QALSODY,
and Salanersen are licensed from Ionis Pharmaceuticals, Inc; Zorevunersen is being developed in collaboration with Stoke Therapeutics, Inc.; ZURZUVAE is being developed in collaboration with Supernus Pharmaceuticals, Inc. # Rare disease is a
commercial designation that includes multiple therapeutic indications. Immunology Neurology Rare Disease#

10 Note: Dapirolizumab pegol is being developed in collaboration with UCB; LEQEMBI
(lecanemab-irmb) is being developed in collaboration with Eisai Co; SPINRAZA, QALSODY, and Salanersen are licensed from Ionis Pharmaceuticals, Inc; Zorevunersen is being developed in collaboration with Stoke Therapeutics, Inc.; ZURZUVAE is
being developed in collaboration with Supernus Pharmaceuticals, Inc. # Rare disease is a commercial designation that includes multiple therapeutic indications. C3G = complement 3 glomerulopathy; IC-MPGN = immune complex membranoproliferative
glomerulonephritis APELLIS PROVIDES THE POTENTIAL FOR ADDITIONAL GROWTH PRODUCTS 10 Apellis adds two commercial assets Broadens, diversifies, and strengthens our current revenue growth drivers Best-in-class medicine approved in
Geographic Atrophy Generated $587M of revenue in 2025 Regulatory filing for prefilled syringe planned for 1H 2026 Best-in-class medicine approved in C3G/IC-MPGN. Also marketed for PNH. Significant commercial potential that
accelerates Biogen’s expansion into nephrology 2030s Opportunities for longer-term growth High-risk/high-reward early-stage pipeline Research portfolio Additional early-stage BD Late 2020s Registrational late-stage
pipeline Litifilimab Felzartamab Zorevunersen Dapirolizumab pegol Salanersen LEQEMBI (preclinical AD) Immunology Neurology Rare Disease# Today Current revenue growth drivers
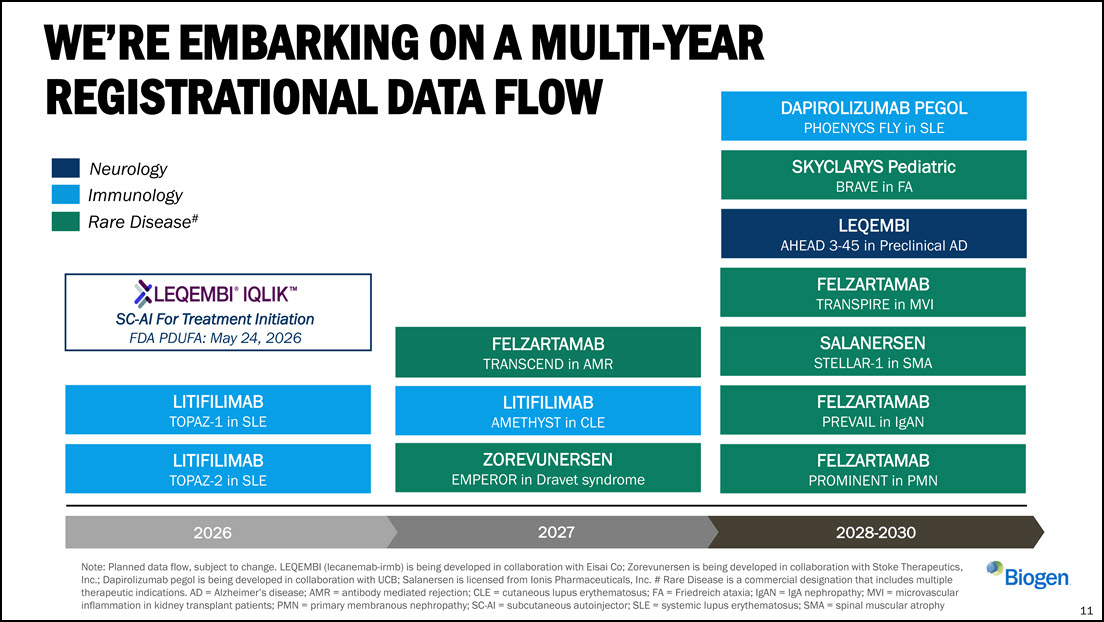
WE’RE EMBARKING ON A MULTI-YEAR REGISTRATIONAL DATA FLOW 11 Neurology Immunology
Rare Disease# 2026 2027 2028-2030 Note: Planned data flow, subject to change. LEQEMBI (lecanemab-irmb) is being developed in collaboration with Eisai Co; Zorevunersen is being developed in collaboration with Stoke Therapeutics, Inc.;
Dapirolizumab pegol is being developed in collaboration with UCB; Salanersen is licensed from Ionis Pharmaceuticals, Inc. # Rare Disease is a commercial designation that includes multiple therapeutic indications. AD = Alzheimer’s disease; AMR =
antibody mediated rejection; CLE = cutaneous lupus erythematosus; FA = Friedreich ataxia; IgAN = IgA nephropathy; MVI = microvascular inflammation in kidney transplant patients; PMN = primary membranous nephropathy; SC-AI = subcutaneous
autoinjector; SLE = systemic lupus erythematosus; SMA = spinal muscular atrophy LITIFILIMAB TOPAZ-2 in SLE LITIFILIMAB AMETHYST in CLE FELZARTAMAB TRANSPIRE in MVI ZOREVUNERSEN EMPEROR in Dravet syndrome SALANERSEN STELLAR-1 in
SMA LEQEMBI AHEAD 3-45 in Preclinical AD FELZARTAMAB PREVAIL in IgAN FELZARTAMAB TRANSCEND in AMR DAPIROLIZUMAB PEGOL PHOENYCS FLY in SLE FELZARTAMAB PROMINENT in PMN SKYCLARYS Pediatric BRAVE in FA LITIFILIMAB TOPAZ-1 in
SLE SC-AI For Treatment Initiation FDA PDUFA: May 24, 2026

12 OUR CULTURE MAKES BREAKTHROUGHS HAPPEN We boldly advance rigorous science to
drive innovation in medicine. We are humble and curious, integrating external and internal advances to successfully compete. We achieve high performance and have greater impact by being decisive and solution-oriented, while effectively
managing risk. We act with the highest integrity with each other and all who place their trust in us. We are open, embrace and leverage differences, and treat everyone with care and dignity. INCLUSIVE ETHICAL THINK BROADLY DRIVE
RESULTS PIONEER

13 Sources: Biogen , Apellis CULTURE COMPARISON: BIOGEN + APELLIS Biogen and
Apellis share themes of prioritizing science, being innovative and decisive, holding a patient-first focus, and embracing inclusion Science Focused Solution Oriented & Innovative Patient Centered, Passionate & Ethical Caring &
Inclusive Pioneer Boldly advance rigorous science Drive Results High performance; decisive and solution-oriented Think Broadly Humble & curious; integrate internal and external advances Ethical Act with the highest
integrity Inclusive Embrace differences; treat everyone with care and dignity We are Complement Scientific integrity, inclusion, curiosity, and collaboration. We are Fearless Take risks and push for productivity and innovation We love
what we do Passionate about work, purpose, and impact We Care Improve the lives of patients, families, and employees We Belong Embrace differences; learn fearlessly and evolve together We are Resourceful Act in the most agile and
efficient way Biogen Essentials Shared Themes Apellis Values

14 BIOGEN’S WELL-BEING COMMITMENT Well-Being Social Emotional Medical, vision
and dental plans Health advocates Wellness programs On-site fitness centers Wellness reimbursements Physical Financial Generous paid time off Summer hours Year-end shutdown Paid sabbatical Parental leave following local
regulations On-site childcare On-site cafeterias and cafés Employee & Family Solutions (EFS) Concierge services Commuter benefits Competitive compensation Retirement/ savings plans Long-term incentive plans No-cost life insurance
and disability coverage Financial advising Tuition reimbursement College planning services Legal consultations and referrals Employee Resource Groups Care Deeply Day for volunteering Charitable company matching Compost/Recycling
programs Community Lab

THANK YOU & QUESTIONS