| 83 Wooster Heights Road Danbury, Connecticut 06810 iqvia.com February 24, 2026 AstraZeneca PLC Legal & Secretary’s Department 1 Francis Crick Avenue Cambridge Biomedical Campus Cambridge CB2 0AA Dear Ladies and Gentlemen: IQVIA DATA DISCLOSURE FOR ANNUAL REPORT AND FORM 20-F INFORMATION 2025 In connection with the anticipated filing by AstraZeneca PLC (“AstraZeneca”) of a Form 20-F with the U.S. Securities and Exchange Commission, IQVIA Inc. (“IQVIA”) hereby authorizes AstraZeneca to refer to IQVIA and certain pharmaceutical industry data derived by IQVIA, as identified (highlighted in green) on the pages annexed hereto as Annex A, which are a selection of pages from AstraZeneca’s Annual Report and Form 20-F Information for the fiscal year ended December 31, 2025 (the “Annual Report”), each of which is incorporated by reference in the registration statement No. 333-278067 for AstraZeneca on Form F-3, and in the registration statements No. 333-293157; No. 333-277197, No. 333-240298, No. 333-226830, No. 333- 216901, No. 333-170381, No. 333-1 52767, No. 333-124689 and No. 333-09062 on Form S-8 for AstraZeneca. IQVIA’s authorization is subject to AstraZeneca’s acknowledgement and agreement that: 1) IQVIA has not undertaken an independent review of the information disclosed in the Annual Report or the Form 20-F other than to discuss its observations as to the accuracy of the information relating to IQVIA and certain pharmaceutical industry data derived by IQVIA; 2) AstraZeneca acknowledges and agrees that IQVIA shall not be deemed an “Expert” in respect of AstraZeneca’s securities filings, and AstraZeneca agrees that it shall not characterize IQVIA as such; and 3) AstraZeneca accepts full responsibility for the disclosure of all information and data, including that relating to IQVIA, set forth in the Annual Report and Form 20-F as filed with the SEC and agrees to indemnify IQVIA from any third party claims that may arise therefrom. Please indicate your agreement to the foregoing by signing in the space indicated below. Our authorization will not become effective until accepted and agreed by AstraZeneca. |
| Very truly yours, /s/ Matthew Gilmartin Name: Matthew Gilmartin Title: SVP, Deputy General Counsel ACCEPTED AND AGREED This 24 day of February 2026 AstraZeneca PLC /s/ Matthew Bowden Name: Matthew Bowden Title: Company Secretary |
| Annex A (See attached) |
| The external environment presents both challenges and opportunities that require us to adapt, innovate and build trust. A growing pharmaceutical sector The pharmaceutical sector continues to grow against a backdrop of increasing demand for healthcare. Global healthcare spending is projected to increase at an annual rate of 7.1% from 2025 to 2029. Healthcare in a Changing World Global trends Shifting economic power Economic power is shifting from the G7 to the largest emerging markets, such as China and countries with large populations, including India and Indonesia, altering global economic dynamics and creating new opportunities and challenges. For example, the G7 comprised some 65% of global GDP in 2000 which is expected to drop to less than one third by 2050. Global instability Continuing geopolitical tensions and shifting alliances are creating a more volatile global landscape, impacting international relations and stability. This includes the rise of economic nationalism, sustained strategic rivalry between the US and China, as well as conflicts, such as the war in Ukraine, and ‘grey zone’ conflict – the contested arena between routine diplomacy and open warfare. Changing populations The UN predicts the global population will reach 9.7 billion by 2050. Key trends include continued urbanisation, falling birth rates in many countries, notably South Korea, Japan and within Western Europe, and an ageing population, with those aged 65 and older set to triple by 2100. Furthermore, the ratio of retirees to workers will rise dramatically as the share of younger people declines, putting structural pressure on pay-as-you-go pensions and on health and long-term care financing. Population growth is also becoming more concentrated, with much of the growth coming from Africa and South Asia. <1/3 The G7’s share of global GDP fell from roughly two thirds (~65%) in 2000 to about half today, and is projected to shrink to less than one third by 2050. (Source: Global Trade Outlook, February 2023) 9.7 billion The UN predicts the global population will reach about 9.7 billion by 2050. (Source: United Nations) 10.0% Global pharmaceutical sales grew by 10.0% in 2025 (Source: IQVIA, IQVIA Midas Quantum Q3 2025) These risks are explored further in the Risk Overview from page 47 and Accessible and affordable healthcare from page 41. Against the background of broad structural trends, the pharmaceutical sector is navigating economic challenges and political uncertainty as well as the impacts of social changes and the climate crisis. Rapidly-advancing technologies offer both risks and benefits, while successful organisations are building trust with stakeholders. AstraZeneca Annual Report & Form 20-F Information 2025 6 Strategic Report Corporate Governance Financial Statements Sustainability Statement Additional Information Healthcare in a Changing World |
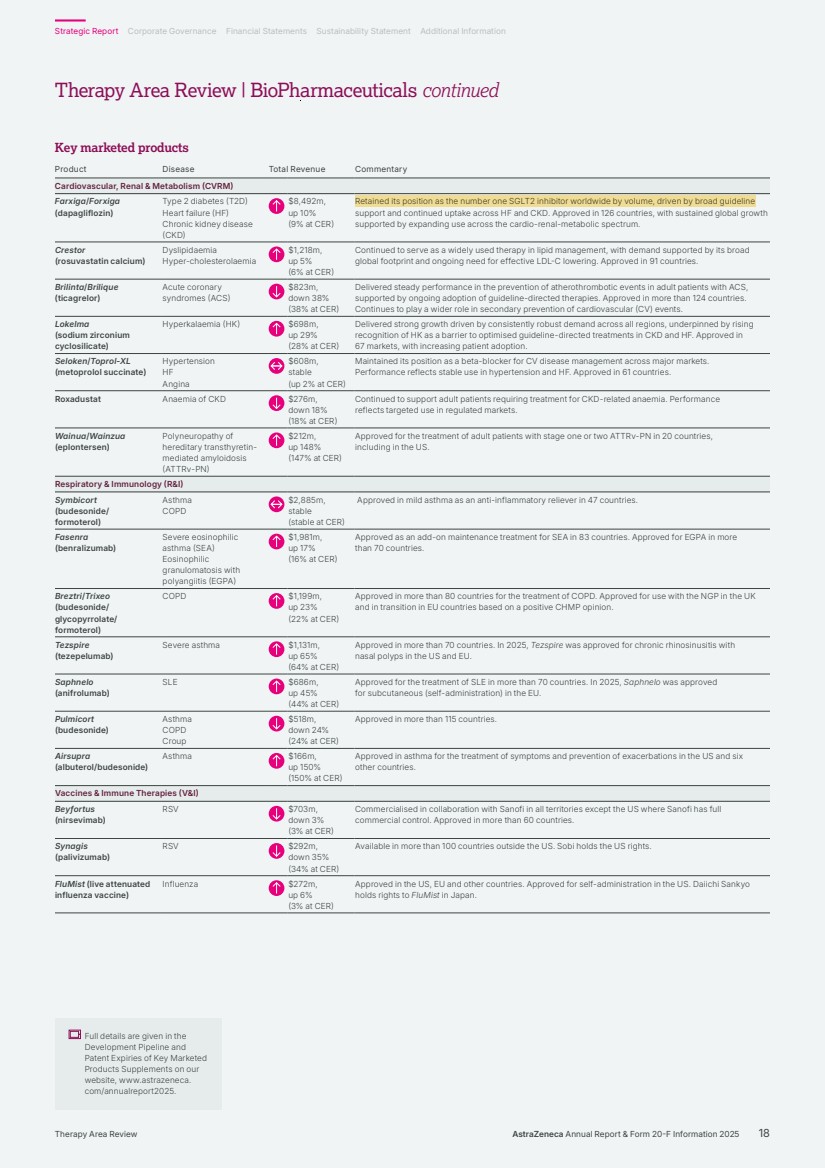
| Key marketed products Product Disease Total Revenue Commentary Cardiovascular, Renal & Metabolism (CVRM) Farxiga/Forxiga (dapagliflozin) Type 2 diabetes (T2D) Heart failure (HF) Chronic kidney disease (CKD) $8,492m, up 10% (9% at CER) Retained its position as the number one SGLT2 inhibitor worldwide by volume, driven by broad guideline support and continued uptake across HF and CKD. Approved in 126 countries, with sustained global growth supported by expanding use across the cardio-renal-metabolic spectrum. Crestor (rosuvastatin calcium) Dyslipidaemia Hyper-cholesterolaemia $1,218m, up 5% (6% at CER) Continued to serve as a widely used therapy in lipid management, with demand supported by its broad global footprint and ongoing need for effective LDL-C lowering. Approved in 91 countries. Brilinta/Brilique (ticagrelor) Acute coronary syndromes (ACS) $823m, down 38% (38% at CER) Delivered steady performance in the prevention of atherothrombotic events in adult patients with ACS, supported by ongoing adoption of guideline-directed therapies. Approved in more than 124 countries. Continues to play a wider role in secondary prevention of cardiovascular (CV) events. Lokelma (sodium zirconium cyclosilicate) Hyperkalaemia (HK) $698m, up 29% (28% at CER) Delivered strong growth driven by consistently robust demand across all regions, underpinned by rising recognition of HK as a barrier to optimised guideline-directed treatments in CKD and HF. Approved in 67 markets, with increasing patient adoption. Seloken/Toprol-XL (metoprolol succinate) Hypertension HF Angina $608m, stable (up 2% at CER) Maintained its position as a beta-blocker for CV disease management across major markets. Performance reflects stable use in hypertension and HF. Approved in 61 countries. Roxadustat Anaemia of CKD $276m, down 18% (18% at CER) Continued to support adult patients requiring treatment for CKD-related anaemia. Performance reflects targeted use in regulated markets. Wainua/Wainzua (eplontersen) Polyneuropathy of hereditary transthyretin-mediated amyloidosis (ATTRv-PN) $212m, up 148% (147% at CER) Approved for the treatment of adult patients with stage one or two ATTRv-PN in 20 countries, including in the US. Respiratory & Immunology (R&I) Symbicort (budesonide/ formoterol) Asthma COPD $2,885m, stable (stable at CER) Approved in mild asthma as an anti-inflammatory reliever in 47 countries. Fasenra (benralizumab) Severe eosinophilic asthma (SEA) Eosinophilic granulomatosis with polyangiitis (EGPA) $1,981m, up 17% (16% at CER) Approved as an add-on maintenance treatment for SEA in 83 countries. Approved for EGPA in more than 70 countries. Breztri/Trixeo (budesonide/ glycopyrrolate/ formoterol) COPD $1,199m, up 23% (22% at CER) Approved in more than 80 countries for the treatment of COPD. Approved for use with the NGP in the UK and in transition in EU countries based on a positive CHMP opinion. Tezspire (tezepelumab) Severe asthma $1,131m, up 65% (64% at CER) Approved in more than 70 countries. In 2025, Tezspire was approved for chronic rhinosinusitis with nasal polyps in the US and EU. Saphnelo (anifrolumab) SLE $686m, up 45% (44% at CER) Approved for the treatment of SLE in more than 70 countries. In 2025, Saphnelo was approved for subcutaneous (self-administration) in the EU. Pulmicort (budesonide) Asthma COPD Croup $518m, down 24% (24% at CER) Approved in more than 115 countries. Airsupra (albuterol/budesonide) Asthma $166m, up 150% (150% at CER) Approved in asthma for the treatment of symptoms and prevention of exacerbations in the US and six other countries. Vaccines & Immune Therapies (V&I) Beyfortus (nirsevimab) RSV $703m, down 3% (3% at CER) Commercialised in collaboration with Sanofi in all territories except the US where Sanofi has full commercial control. Approved in more than 60 countries. Synagis (palivizumab) RSV $292m, down 35% (34% at CER) Available in more than 100 countries outside the US. Sobi holds the US rights. FluMist (live attenuated influenza vaccine) Influenza $272m, up 6% (3% at CER) Approved in the US, EU and other countries. Approved for self-administration in the US. Daiichi Sankyo holds rights to FluMist in Japan. Full details are given in the Development Pipeline and Patent Expiries of Key Marketed Products Supplements on our website, www.astrazeneca. com/annualreport2025. AstraZeneca Annual Report & Form 20-F Information 2025 18 Strategic Report Corporate Governance Financial Statements Sustainability Statement Additional Information Therapy Area Review Therapy Area Review | BioPharmaceuticals continued |
| • the risk of failure to maintain supply of compliant, quality medicines • the risk of illegal trade in our Group’s medicines • the risk of reliance on third-party goods and services • the risk of failure in IT or cybersecurity • the risk of failure of critical processes • the risk of failure to collect and manage data and AI in line with legal and regulatory requirements and strategic objectives • the risk of failure to attract, develop, engage and retain a diverse, talented and capable workforce • the risk of failure to meet our sustainability targets, regulatory requirements or stakeholder expectations with respect to the environment • the risk of failure to meet regulatory and ethical expectations on commercial practices, including anti-bribery anti-corruption, anti-fraud and scientific exchanges • the risk of the safety and efficacy of marketed medicines being questioned • the risk of adverse outcome of litigation and/or governmental investigations • intellectual property-related risks to the Group’s products • the risk of failure to achieve strategic plans or meet targets or expectations • the risk of geopolitical and/or macroeconomic volatility disrupting the operation of our global business • the risk of failure in internal control, financial reporting or the occurrence of fraud • the risk of unexpected deterioration in the Group’s financial position. Certain of these factors are discussed in more detail, without limitation, in the Risk Supplement available on our website, www.astrazeneca.com/annualreport2025, and reproduced in AstraZeneca’s Form 20-F filing for 2025, available on the SEC website www.sec.gov. Nothing in this Annual Report should be construed as a profit forecast. Inclusion of Reported performance, Core financial measures and constant exchange rate growth rates AstraZeneca’s determination of non-GAAP measures, together with our presentation of them within our financial information, may differ from similarly titled non-GAAP measures of other companies. Statements of competitive position, growth rates and sales In this Annual Report, except as otherwise stated, market information regarding the position of our business or products relative to its or their competition is based upon published statistical sales data for the 12 months ended 30 September 2025 obtained from IQVIA, a leading supplier of statistical data to the pharmaceutical industry. Except as otherwise stated, these market share and industry data from IQVIA have been derived by comparing our sales revenue with competitors’ and total market sales revenues for that period, and except as otherwise stated, growth rates are given at CER. For the purposes of this Annual Report, unless otherwise stated, references to the world pharmaceutical market or similar phrases are to the 63 countries contained in the IQVIA database, which amounted to approximately 85% (in value) of the countries audited by IQVIA. Information on websites Information on or accessible through AstraZeneca websites (including www.astrazeneca.com and any websites referenced in this Annual Report) or any external/third-party websites does not form part of and is not incorporated into this Annual Report. Cautionary statement regarding forward‑looking statements The purpose of this Annual Report is to provide information to the members of the Company. The Company and its Directors, employees, agents and advisers do not accept or assume responsibility for any other person to whom this Annual Report is shown or into whose hands it may come and any such responsibility or liability is expressly disclaimed. In order, among other things, to utilise the ‘safe harbour’ provisions of the US Private Securities Litigation Reform Act of 1995 and the UK Companies Act 2006, we are providing the following cautionary statement: This Annual Report contains certain forward-looking statements with respect to the operations, performance and financial condition of the Group, including, among other things, statements about expected revenues, margins, earnings per share or other financial or other measures. Forward-looking statements are statements relating to the future which are based on information available at the time such statements are made, including information relating to risks and uncertainties. Although we believe that the forward-looking statements in this Annual Report are based on reasonable assumptions, the matters discussed in the forward-looking statements may be influenced by factors that could cause actual outcomes and results to be materially different from those predicted. The forward-looking statements reflect knowledge and information available at the date of the preparation of this Annual Report and the Company undertakes no obligation to update these forward-looking statements. We identify the forward-looking statements by using the words ‘anticipates’, ‘believes’, ‘expects’, ‘intends’ and similar expressions in such statements. Important factors that could cause actual results to differ materially from those contained in forward-looking statements, certain of which are beyond our control, include, among other things: • the risk of failure or delay in delivery of pipeline or launch of new medicines • the risk of failure to meet regulatory or ethical requirements for medicine development or approval • the risk of failures or delays in the quality or execution of the Group’s commercial strategies • the risk of pricing, affordability, access and competitive pressures Strategic Report Corporate Governance Financial Statements Sustainability Statement Additional Information Important information for readers of this Annual Report AstraZeneca Annual Report & Form 20-F Information 2025 228 Important information for readers of this Annual Report |