.2
March 23, 2026 Topline Results of Phase 3b ENCORE Study of ARIKAYCE® in
Patients with Newly Diagnosed MAC Lung Infection Not Yet Treated With Antibiotics MAC: Mycobacterium avium complex lung disease | ARIKAYCE is approved in the U.S. as ARIKAYCE (amikacin liposome inhalation suspension), in Europe as ARIKAYCE
Liposomal 590 mg Nebuliser Dispersion, and in Japan as ARIKAYCE inhalation 590 mg (amikacin sulfate inhalation drug product)

Opening Remarks Closing Remarks Q&A Session 4-6 7-16 17 18 Will
Lewis Chair & CEO Martina Flammer Chief Medical Officer Study Results

Forward Looking Statements The forward-looking statements in this presentation
are based upon the Company’s current expectations and beliefs, and involve known and unknown risks, uncertainties and other factors, which may cause the Company’s actual results, performance and achievements and the timing of certain events
to differ materially from the results, performance, achievements or timings discussed, projected, anticipated or indicated in any forward-looking statements. Such risks, uncertainties and other factors include, among others, the following:
risk that topline data from the Company’s clinical trials, including the ENCORE trial, that the Company announces or publishes from time to time may change as more patient data become available or may be interpreted differently if additional
data are disclosed; failure to continue to successfully commercialize ARIKAYCE® in the U.S., Europe or Japan, or to maintain U.S., European or Japanese approval for ARIKAYCE; the Company’s inability to obtain full approval of ARIKAYCE from
the FDA, or the Company’s failure to obtain regulatory approval to expand ARIKAYCE's indication to a broader patient population; failure to obtain, or delays in obtaining, regulatory approvals for ARIKAYCE outside of the U.S., Europe and
Japan, including separate regulatory approval for the Lamira® Nebulizer System in each market and for each usage; failure to successfully commercialize ARIKAYCE in a broader patient population, if approved by applicable regulatory
authorities; uncertainties or changes in the degree of market acceptance of ARIKAYCE by physicians, patients, third-party payors and others in the healthcare community; the Company’s inability to obtain and maintain adequate reimbursement
from government or third-party payors for ARIKAYCE in a broader patient population, if approved, or acceptable prices for ARIKAYCE; inaccuracies in the Company’s estimates of the size of the potential markets for ARIKAYCE or in data the
Company has used to identify physicians, expected rates of patient uptake, duration of expected treatment, or expected patient adherence or discontinuation rates; failure of third parties on which the Company is dependent to manufacture
sufficient quantities of ARIKAYCE for commercial needs, or to comply with the Company’s agreements or laws and regulations that impact the Company’s business; the Company’s inability to create or maintain an effective direct sales and
marketing infrastructure or to partner with third parties that offer such an infrastructure for distribution of ARIKAYCE; development of unexpected safety or efficacy concerns related to ARIKAYCE; restrictions or other obligations imposed on
the Company by agreements related to ARIKAYCE, including the Company’s license agreement with PARI, and failure to comply with the Company’s obligations under such agreements; the cost and potential reputational damage resulting from
litigation to which the Company is or may become a party, including product liability claims; and delays in the execution of plans to build out an additional third-party manufacturing facility approved by the appropriate regulatory
authorities and unexpected expenses associated with those plans. The Company may not actually achieve the results, plans, intentions or expectations indicated by the Company's forward-looking statements because, by their nature,
forward-looking statements involve risks and uncertainties because they relate to events and depend on circumstances that may or may not occur in the future. For additional information about the risks and uncertainties that may affect the
Company's business, please see the factors discussed in Item 1A, "Risk Factors," in the Company's Annual Report on Form 10-K for the year ended December 31, 2025 and any subsequent Company filings with the Securities and Exchange Commission
(SEC). The Company cautions readers not to place undue reliance on any such forward-looking statements, which speak only as of the date of this press release. The Company disclaims any obligation, except as specifically required by law
and the rules of the SEC, to publicly update or revise any such statements to reflect any change in expectations or in events, conditions or circumstances on which any such statements may be based, or that may affect the likelihood that
actual results will differ from those set forth in the forward-looking statements. Additional Disclaimers: ARIKAYCE has not been approved for the treatment of all patients with MACLD. This presentation is not promotion or advertisement of
ARIKAYCE. Insmed and ARIKAYCE are registered trademarks of Insmed Incorporated. All other trademarks are property of their respective owner(s). FDA: Food & Drug Administration | U.S.: United States | MACLD: Mycobacterium avium complex
lung disease

Opening Remarks Will Lewis | Chair & CEO

ENCORE Demonstrates Clear Success PRO: patient reported outcome | MAC:
Mycobacterium avium complex lung disease | 1 ARIKAYCE + macrolide-based background regimen consisting of Azithromycin (AZI) and Ethambutol (ETH) | 2 Empty liposome placebo control + AZI + ETH Statistically significant improvement in
Respiratory Symptom Score at Month 13 Statistically significant greater rate of culture conversion by Months 6, 12, 13, and 15 Earlier, greater, and more durable culture conversion throughout the entire study period Patients on ARIKAYCE®
showed1… …versus patients on active multidrug comparator therapy2 Using a validated PRO, results demonstrate that ARIKAYCE improves respiratory symptoms and effectively clears MAC infection from the lungs

Next Steps: Results Support Actions to Seek Expansion of ARIKAYCE’s Label in the
U.S. and Japan Plan to file sNDA with FDA for potential U.S. label expansion in MACLD patients in 2H:26 Will also request conversion of ARIKAYCE’s current U.S. conditional label to traditional approval Intend to submit data to PDMA to
support potential Japanese label expansion in 2H:26 sNDA: supplemental new drug application | U.S.: United States | FDA: Food & Drug Administration | PDMA: Pharmaceuticals & Medical Device Agency | H: half | MAC / MACLD:
Mycobacterium avium complex lung disease | TAM: total addressable market | * if expanded label for ARIKAYCE is approved | † Total addressable Refractory MAC patients in the U.S., Europe (France, Germany, Italy, Spain, and United Kingdom), and
Japan | ‡ Additional addressable MAC patients in the U.S. and Japan if expanded ARIKAYCE label is approved | Note: References related to patient TAMs can be found in the Company's Investor Presentation Potential Label Expansion* Current TAM
30K† (Refractory MAC only) ~+200K‡ Patients* (MACLD) Addressable Patient Population

Study Results Martina Flammer | Chief Medical Officer
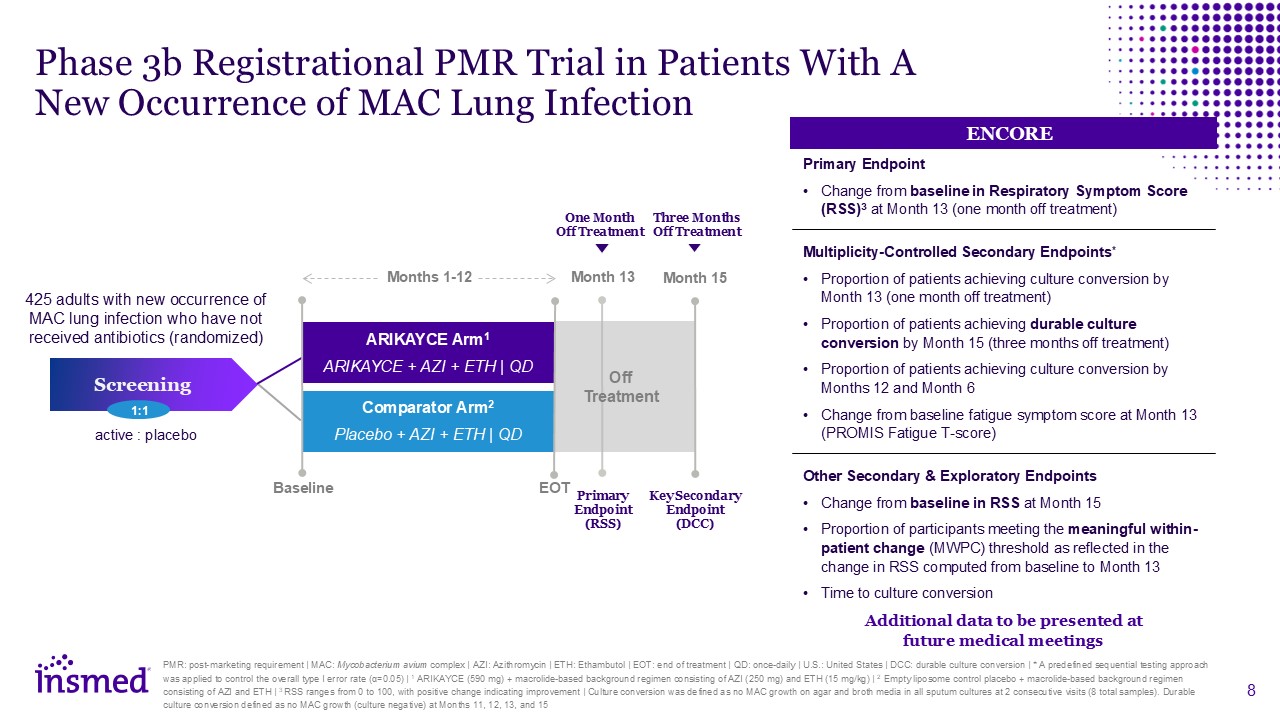
Phase 3b Registrational PMR Trial in Patients With A New Occurrence of MAC Lung
Infection PMR: post-marketing requirement | MAC: Mycobacterium avium complex | AZI: Azithromycin | ETH: Ethambutol | EOT: end of treatment | QD: once-daily | U.S.: United States | DCC: durable culture conversion | * A predefined sequential
testing approach was applied to control the overall type I error rate (α=0.05) | 1 ARIKAYCE (590 mg) + macrolide-based background regimen consisting of AZI (250 mg) and ETH (15 mg/kg) | 2 Empty liposome control placebo + macrolide-based
background regimen consisting of AZI and ETH | 3 RSS ranges from 0 to 100, with positive change indicating improvement | Culture conversion was defined as no MAC growth on agar and broth media in all sputum cultures at 2 consecutive visits (8
total samples). Durable culture conversion defined as no MAC growth (culture negative) at Months 11, 12, 13, and 15 Key Secondary Endpoint (DCC) 425 adults with new occurrence of MAC lung infection who have not received antibiotics
(randomized) ARIKAYCE Arm1 ARIKAYCE + AZI + ETH | QD Comparator Arm2 Placebo + AZI + ETH | QD Primary Endpoint (RSS) Month 13 Month 15 active : placebo Screening 1:1 One Month Off Treatment Three Months Off Treatment Off
Treatment Months 1-12 Baseline EOT ENCORE Primary Endpoint Change from baseline in Respiratory Symptom Score (RSS)3 at Month 13 (one month off treatment) Multiplicity-Controlled Secondary Endpoints* Proportion of patients achieving
culture conversion by Month 13 (one month off treatment) Proportion of patients achieving durable culture conversion by Month 15 (three months off treatment) Proportion of patients achieving culture conversion by Months 12 and Month
6 Change from baseline fatigue symptom score at Month 13 (PROMIS Fatigue T-score) Other Secondary & Exploratory Endpoints Change from baseline in RSS at Month 15 Proportion of participants meeting the meaningful within-patient change
(MWPC) threshold as reflected in the change in RSS computed from baseline to Month 13 Time to culture conversion Additional data to be presented at future medical meetings
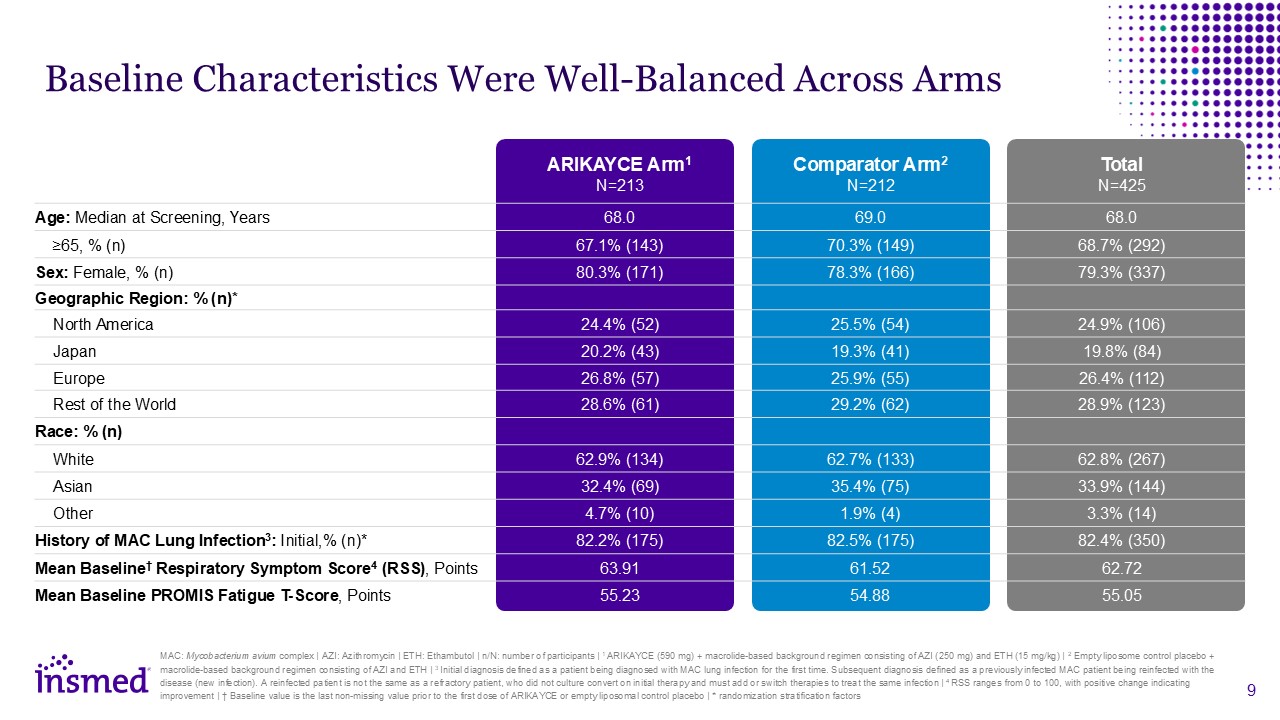
ARIKAYCE Arm1N=213 Comparator Arm2N=212 Total N=425 Age: Median at
Screening, Years 68.0 69.0 68.0 ≥65, % (n) 67.1% (143) 70.3% (149) 68.7% (292) Sex: Female, % (n) 80.3% (171) 78.3% (166) 79.3% (337) Geographic Region: % (n)* North America 24.4% (52) 25.5% (54) 24.9% (106) Japan 20.2%
(43) 19.3% (41) 19.8% (84) Europe 26.8% (57) 25.9% (55) 26.4% (112) Rest of the World 28.6% (61) 29.2% (62) 28.9% (123) Race: % (n) White 62.9% (134) 62.7% (133) 62.8% (267) Asian 32.4% (69) 35.4% (75) 33.9%
(144) Other 4.7% (10) 1.9% (4) 3.3% (14) History of MAC Lung Infection3: Initial,% (n)* 82.2% (175) 82.5% (175) 82.4% (350) Mean Baseline† Respiratory Symptom Score4 (RSS), Points 63.91 61.52 62.72 Mean Baseline PROMIS Fatigue
T-Score, Points 55.23 54.88 55.05 Baseline Characteristics Were Well-Balanced Across Arms MAC: Mycobacterium avium complex | AZI: Azithromycin | ETH: Ethambutol | n/N: number of participants | 1 ARIKAYCE (590 mg) + macrolide-based
background regimen consisting of AZI (250 mg) and ETH (15 mg/kg) | 2 Empty liposome control placebo + macrolide-based background regimen consisting of AZI and ETH | 3 Initial diagnosis defined as a patient being diagnosed with MAC lung
infection for the first time. Subsequent diagnosis defined as a previously infected MAC patient being reinfected with the disease (new infection). A reinfected patient is not the same as a refractory patient, who did not culture convert on
initial therapy and must add or switch therapies to treat the same infection | 4 RSS ranges from 0 to 100, with positive change indicating improvement | † Baseline value is the last non-missing value prior to the first dose of ARIKAYCE or
empty liposomal control placebo | * randomization stratification factors
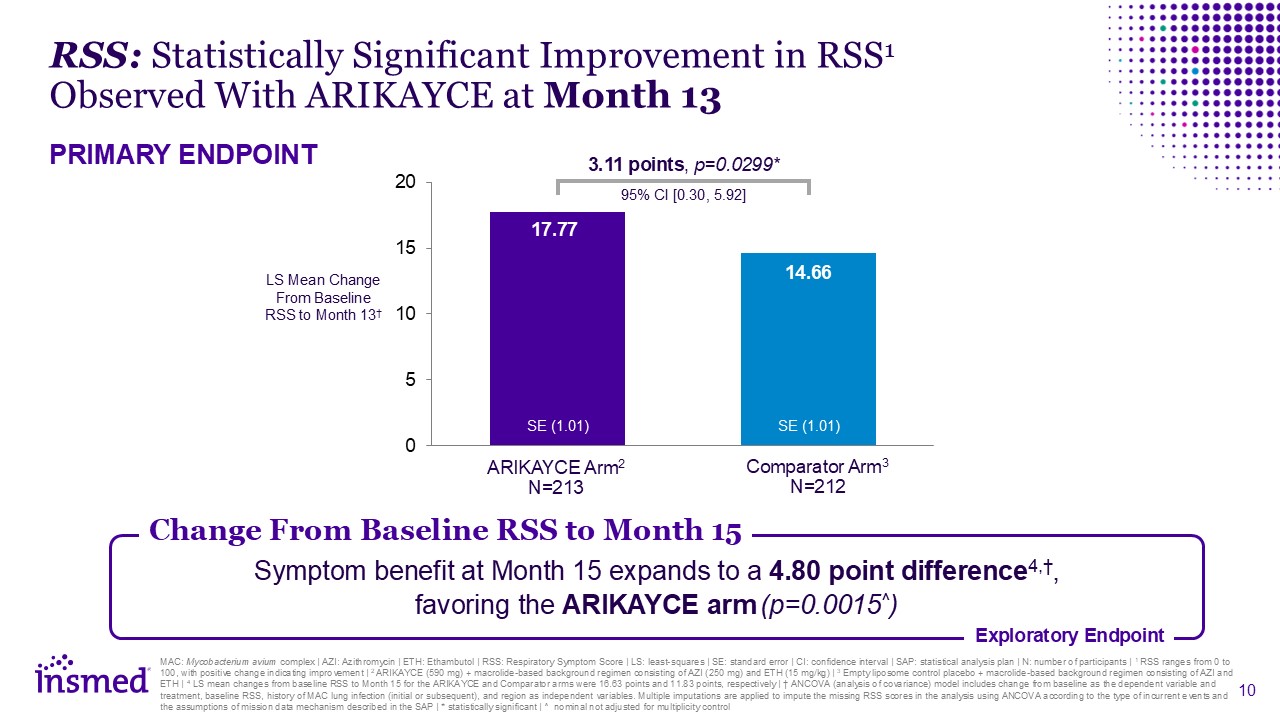
MAC: Mycobacterium avium complex | AZI: Azithromycin | ETH: Ethambutol | RSS:
Respiratory Symptom Score | LS: least-squares | SE: standard error | CI: confidence interval | SAP: statistical analysis plan | N: number of participants | 1 RSS ranges from 0 to 100, with positive change indicating improvement | 2 ARIKAYCE
(590 mg) + macrolide-based background regimen consisting of AZI (250 mg) and ETH (15 mg/kg) | 3 Empty liposome control placebo + macrolide-based background regimen consisting of AZI and ETH | 4 LS mean changes from baseline RSS to Month 15
for the ARIKAYCE and Comparator arms were 16.63 points and 11.83 points, respectively | † ANCOVA (analysis of covariance) model includes change from baseline as the dependent variable and treatment, baseline RSS, history of MAC lung infection
(initial or subsequent), and region as independent variables. Multiple imputations are applied to impute the missing RSS scores in the analysis using ANCOVA according to the type of incurrent events and the assumptions of mission data
mechanism described in the SAP | * statistically significant | ^ nominal not adjusted for multiplicity control Symptom benefit at Month 15 expands to a 4.80 point difference4,†, favoring the ARIKAYCE arm (p=0.0015^) Exploratory
Endpoint RSS: Statistically Significant Improvement in RSS1 Observed With ARIKAYCE at Month 13 PRIMARY ENDPOINT LS Mean Change From Baseline RSS to Month 13† ARIKAYCE Arm2 N=213 Comparator Arm3 N=212 3.11 points, p=0.0299* 95% CI
[0.30, 5.92] SE (1.01) SE (1.01) Change From Baseline RSS to Month 15
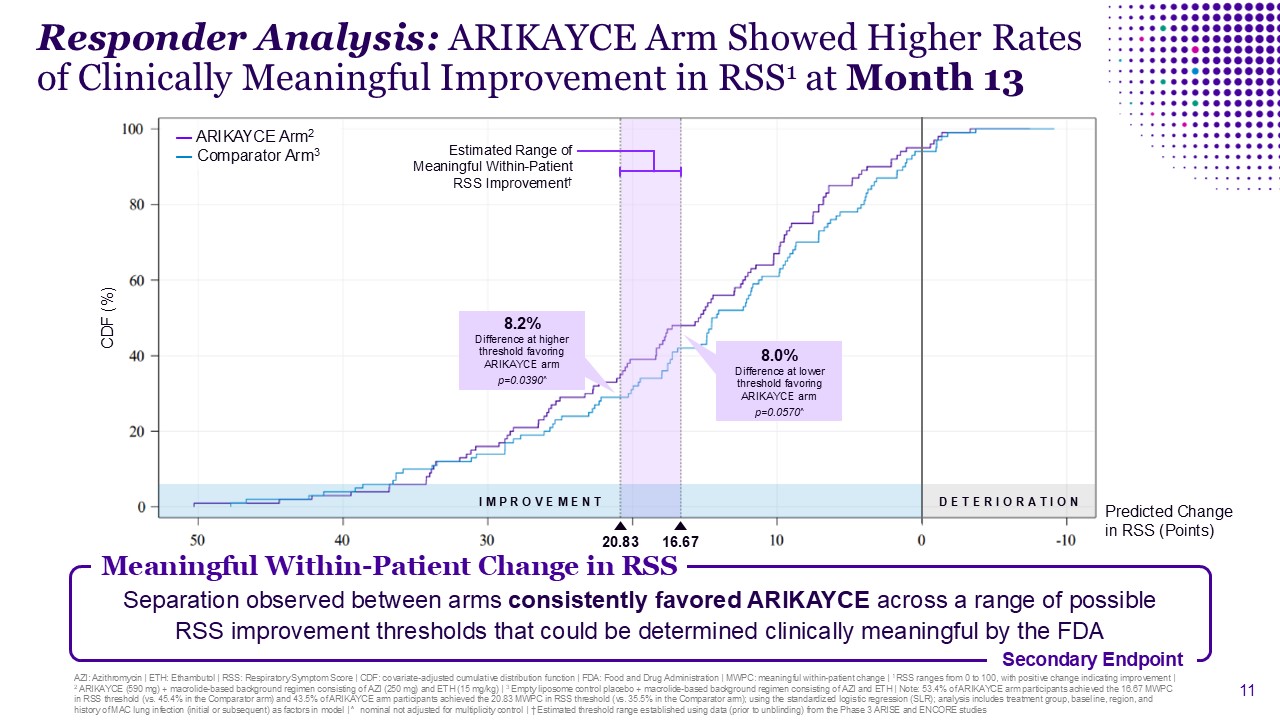
Responder Analysis: ARIKAYCE Arm Showed Higher Rates of Clinically Meaningful
Improvement in RSS1 at Month 13 Meaningful Within-Patient Change in RSS Secondary Endpoint Separation observed between arms consistently favored ARIKAYCE across a range of possible RSS improvement thresholds that could be determined
clinically meaningful by the FDA CDF (%) 16.67 — ARIKAYCE Arm2 — Comparator Arm3 8.0% Difference at lower threshold favoring ARIKAYCE arm p=0.0570^ 8.2% Difference at higher threshold favoring ARIKAYCE arm p=0.0390^ Predicted
Change in RSS (Points) I M P R O V E M E N T D E T E R I O R A T I O N Estimated Range of Meaningful Within-Patient RSS Improvement† 20.83 AZI: Azithromycin | ETH: Ethambutol | RSS: Respiratory Symptom Score | CDF: covariate-adjusted
cumulative distribution function | FDA: Food and Drug Administration | MWPC: meaningful within-patient change | 1 RSS ranges from 0 to 100, with positive change indicating improvement | 2 ARIKAYCE (590 mg) + macrolide-based background regimen
consisting of AZI (250 mg) and ETH (15 mg/kg) | 3 Empty liposome control placebo + macrolide-based background regimen consisting of AZI and ETH | Note: 53.4% of ARIKAYCE arm participants achieved the 16.67 MWPC in RSS threshold (vs. 45.4% in
the Comparator arm) and 43.5% of ARIKAYCE arm participants achieved the 20.83 MWPC in RSS threshold (vs. 35.5% in the Comparator arm); using the standardized logistic regression (SLR); analysis includes treatment group, baseline, region, and
history of MAC lung infection (initial or subsequent) as factors in model | ^ nominal not adjusted for multiplicity control | † Estimated threshold range established using data (prior to unblinding) from the Phase 3 ARISE and ENCORE studies
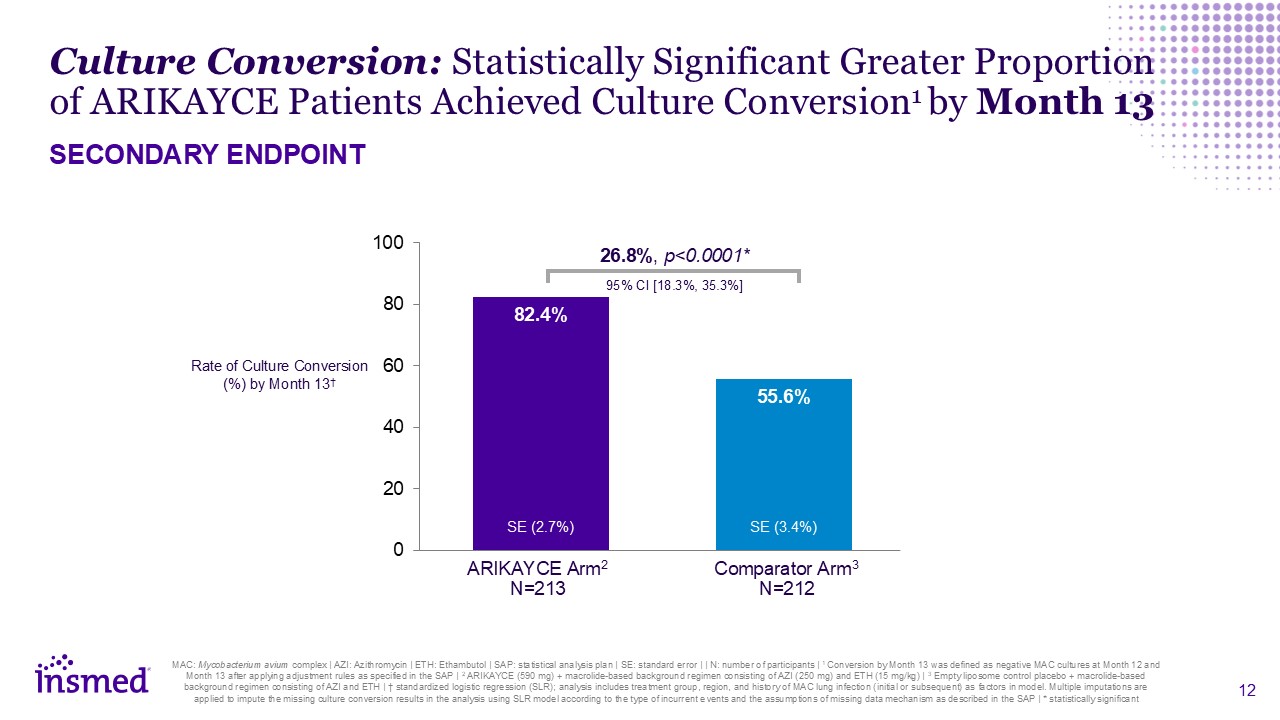
MAC: Mycobacterium avium complex | AZI: Azithromycin | ETH: Ethambutol | SAP:
statistical analysis plan | SE: standard error | | N: number of participants | 1 Conversion by Month 13 was defined as negative MAC cultures at Month 12 and Month 13 after applying adjustment rules as specified in the SAP | 2 ARIKAYCE (590
mg) + macrolide-based background regimen consisting of AZI (250 mg) and ETH (15 mg/kg) | 3 Empty liposome control placebo + macrolide-based background regimen consisting of AZI and ETH | † standardized logistic regression (SLR); analysis
includes treatment group, region, and history of MAC lung infection (initial or subsequent) as factors in model. Multiple imputations are applied to impute the missing culture conversion results in the analysis using SLR model according to
the type of incurrent events and the assumptions of missing data mechanism as described in the SAP | * statistically significant 26.8%, p<0.0001* 95% CI [18.3%, 35.3%] Rate of Culture Conversion (%) by Month 13† ARIKAYCE
Arm2 N=213 Comparator Arm3 N=212 Culture Conversion: Statistically Significant Greater Proportion of ARIKAYCE Patients Achieved Culture Conversion1 by Month 13 SECONDARY ENDPOINT SE (2.7%) SE (3.4%)
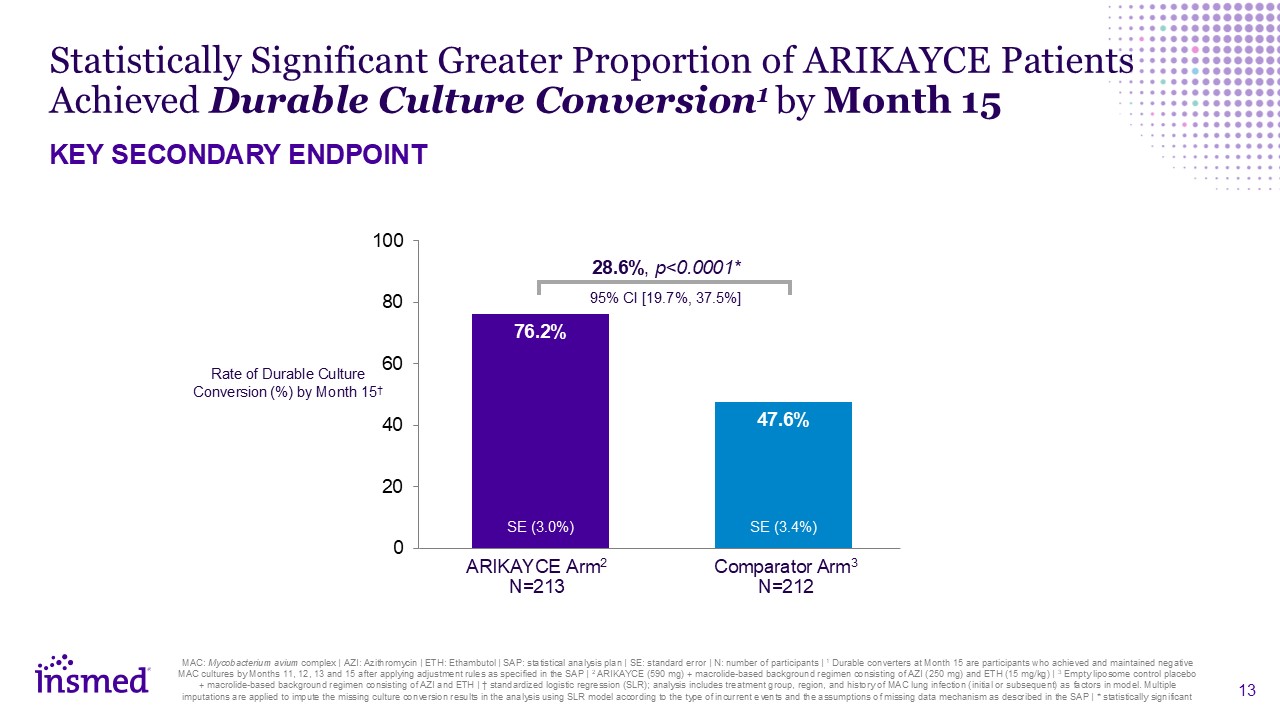
MAC: Mycobacterium avium complex | AZI: Azithromycin | ETH: Ethambutol | SAP:
statistical analysis plan | SE: standard error | N: number of participants | 1 Durable converters at Month 15 are participants who achieved and maintained negative MAC cultures by Months 11, 12, 13 and 15 after applying adjustment rules as
specified in the SAP | 2 ARIKAYCE (590 mg) + macrolide-based background regimen consisting of AZI (250 mg) and ETH (15 mg/kg) | 3 Empty liposome control placebo + macrolide-based background regimen consisting of AZI and ETH | † standardized
logistic regression (SLR); analysis includes treatment group, region, and history of MAC lung infection (initial or subsequent) as factors in model. Multiple imputations are applied to impute the missing culture conversion results in the
analysis using SLR model according to the type of incurrent events and the assumptions of missing data mechanism as described in the SAP | * statistically significant 28.6%, p<0.0001* 95% CI [19.7%, 37.5%] Rate of Durable Culture
Conversion (%) by Month 15† ARIKAYCE Arm2 N=213 Comparator Arm3 N=212 Statistically Significant Greater Proportion of ARIKAYCE Patients Achieved Durable Culture Conversion1 by Month 15 KEY SECONDARY ENDPOINT SE (3.0%) SE (3.4%)
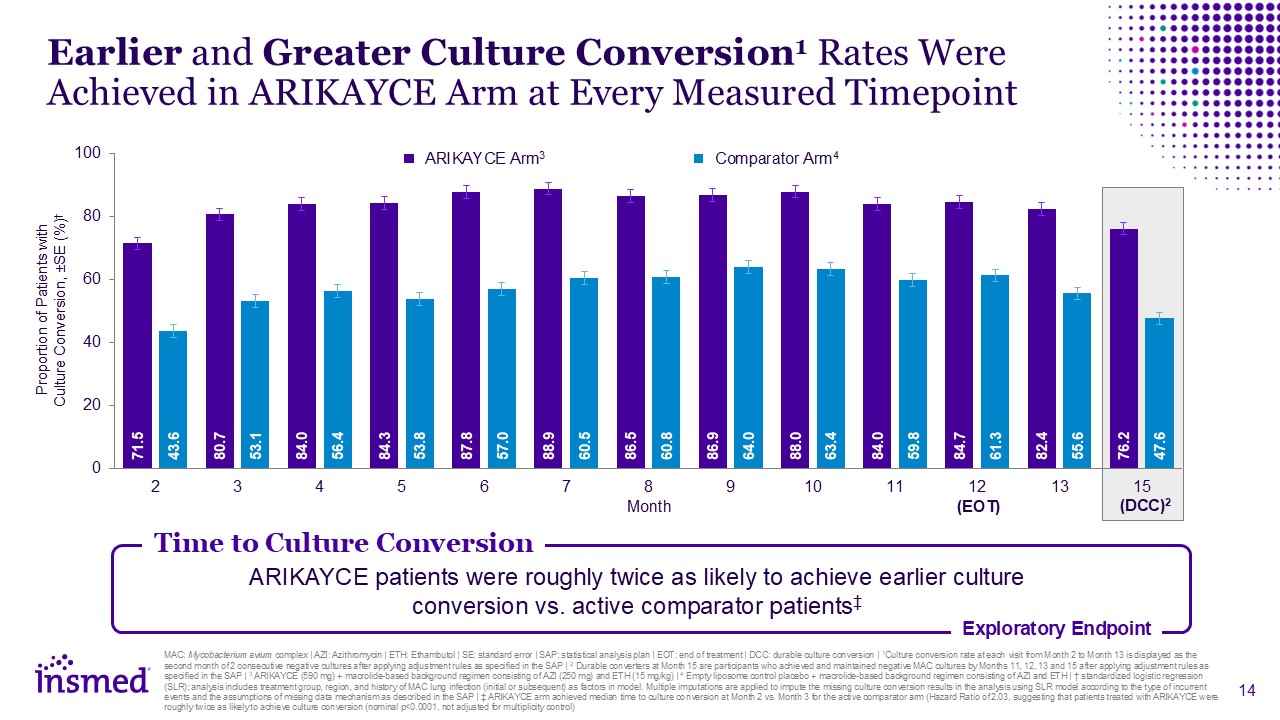
Earlier and Greater Culture Conversion1 Rates Were Achieved in ARIKAYCE Arm at
Every Measured Timepoint Proportion of Patients with Culture Conversion, ±SE (%)† Month ARIKAYCE Arm3 Comparator Arm4 MAC: Mycobacterium avium complex | AZI: Azithromycin | ETH: Ethambutol | SE: standard error | SAP: statistical analysis
plan | EOT: end of treatment | DCC: durable culture conversion | 1Culture conversion rate at each visit from Month 2 to Month 13 is displayed as the second month of 2 consecutive negative cultures after applying adjustment rules as specified
in the SAP | 2 Durable converters at Month 15 are participants who achieved and maintained negative MAC cultures by Months 11, 12, 13 and 15 after applying adjustment rules as specified in the SAP | 3 ARIKAYCE (590 mg) + macrolide-based
background regimen consisting of AZI (250 mg) and ETH (15 mg/kg) | 4 Empty liposome control placebo + macrolide-based background regimen consisting of AZI and ETH | † standardized logistic regression (SLR); analysis includes treatment group,
region, and history of MAC lung infection (initial or subsequent) as factors in model. Multiple imputations are applied to impute the missing culture conversion results in the analysis using SLR model according to the type of incurrent events
and the assumptions of missing data mechanism as described in the SAP | ‡ ARIKAYCE arm achieved median time to culture conversion at Month 2 vs. Month 3 for the active comparator arm (Hazard Ratio of 2.03, suggesting that patients treated
with ARIKAYCE were roughly twice as likely to achieve culture conversion (nominal p<0.0001, not adjusted for multiplicity control) Time to Culture Conversion Exploratory Endpoint ARIKAYCE patients were roughly twice as likely to achieve
earlier culture conversion vs. active comparator patients‡ (EOT) (DCC)2
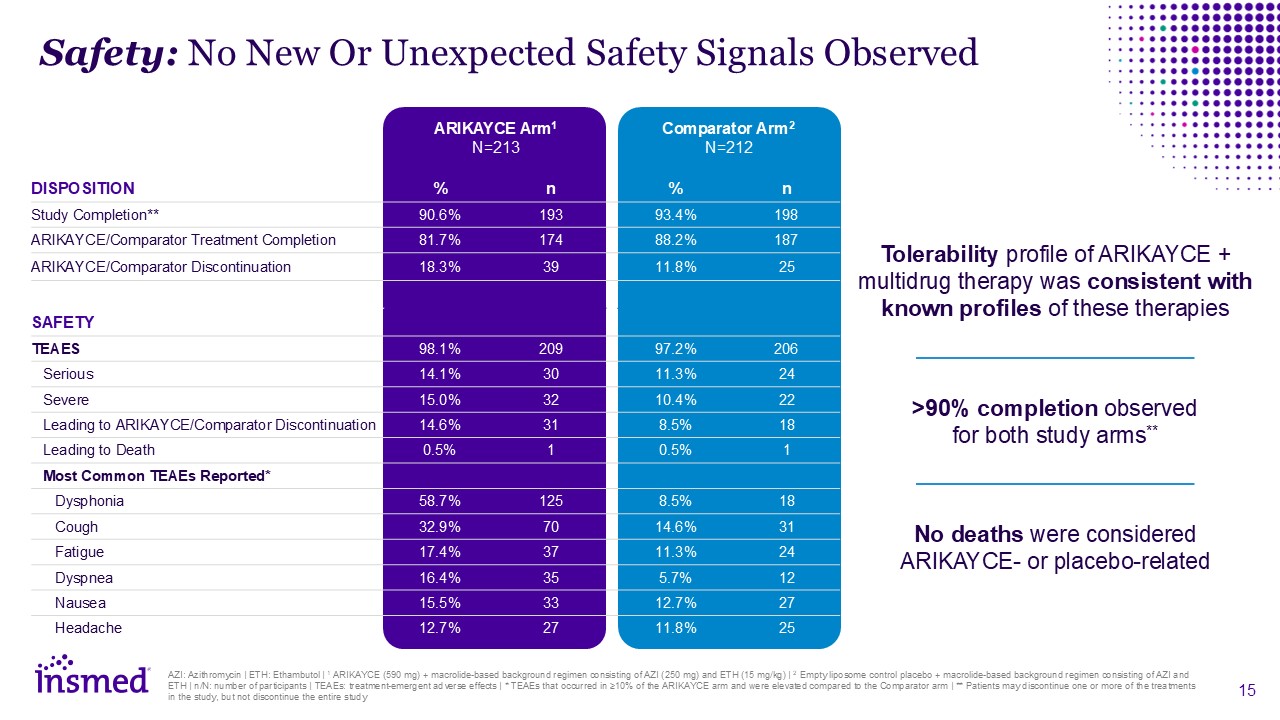
Safety: No New Or Unexpected Safety Signals Observed ARIKAYCE
Arm1 N=213 Comparator Arm2 N=212 DISPOSITION % n % n Study Completion** 90.6% 193 93.4% 198 ARIKAYCE/Comparator Treatment Completion 81.7% 174 88.2% 187 ARIKAYCE/Comparator
Discontinuation 18.3% 39 11.8% 25 SAFETY TEAES 98.1% 209 97.2% 206 Serious 14.1% 30 11.3% 24 Severe 15.0% 32 10.4% 22 Leading to ARIKAYCE/Comparator Discontinuation 14.6% 31 8.5% 18 Leading to
Death 0.5% 1 0.5% 1 Most Common TEAEs Reported* Dysphonia 58.7% 125 8.5% 18 Cough 32.9% 70 14.6% 31 Fatigue 17.4% 37 11.3% 24 Dyspnea 16.4% 35 5.7% 12 Nausea 15.5% 33 12.7% 27
Headache 12.7% 27 11.8% 25 AZI: Azithromycin | ETH: Ethambutol | 1 ARIKAYCE (590 mg) + macrolide-based background regimen consisting of AZI (250 mg) and ETH (15 mg/kg) | 2 Empty liposome control placebo + macrolide-based background
regimen consisting of AZI and ETH | n/N: number of participants | TEAEs: treatment-emergent adverse effects | * TEAEs that occurred in ≥10% of the ARIKAYCE arm and were elevated compared to the Comparator arm | ** Patients may discontinue one
or more of the treatments in the study, but not discontinue the entire study Tolerability profile of ARIKAYCE + multidrug therapy was consistent with known profiles of these therapies >90% completion observed for both study arms** No
deaths were considered ARIKAYCE- or placebo-related

RSS Improvement Statistically significant at Month 13* Strengthening of
improvement at Month 15^ Greater proportion of clinically meaningful RSS responders at Month 13 Culture Conversion Statistically significant greater conversion by Months 6, 12, 13 and 15* Earlier, greater, and more durable
conversion Results Show That Early Treatment with ARIKAYCE + Multidrug Therapy Can Significantly Improve Outcomes For MACLD Patients Treatment with ARIKAYCE1 vs. Comparator2 showed… Safety & Tolerability >90% completion in both
study arms No new or unexpected safety signals observed AZI: Azithromycin | ETH: Ethambutol | 1 ARIKAYCE (590 mg) + macrolide-based background regimen consisting of AZI (250 mg) and ETH (15 mg/kg) | 2 Empty liposome control placebo +
macrolide-based background regimen consisting of AZI and ETH | MAC / MACLD: Mycobacterium avium complex lung disease | RSS: Respiratory Symptom Score | * statistically significant | ^ not multiplicity controlled; nominally statistically
significant

Results bring new hope to patients and providers battling MACLD Early treatment
with ARIKAYCE may improve patient respiratory symptoms, achieve culture conversion sooner, and maintain conversion for longer Expect to file a sNDA for ARIKAYCE in patients with MACLD with the FDA in 2H:26 MAC / MACLD: Mycobacterium avium
complex lung disease | sNDA: supplemental new drug application | FDA: Food and Drug Administration | H: half Clear Success Unlocks Pathway to Engage with Regulatory Authorities

Q&A Session Will Lewis Chair & CEO Sara Bonstein Chief Financial
Officer Kevin Mange Chief Development Officer Martina Flammer Chief Medical Officer Thank you to the patients and investigators who participated in this study!
