
A Fully-Integrated Biopharmaceutical Company Featuring a Robust Pipeline of
Degraders and DACs

This presentation contains “forward-looking statements” within the meaning of the
federal securities laws, including Section 27A of the United States Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended, regarding the current plans, expectations and strategies of GYRE
Therapeutics, Inc. (“Gyre”), Cullgen Inc. (“Cullgen”) and their respective subsidiaries, which statements are subject to substantial risks and uncertainties and are based on management’s estimates and assumptions. All statements, other than
statements of historical facts included in this presentation, are forward-looking statements, including statements concerning the potential merger between Gyre and Cullgen (the “Merger”) and the potential benefits thereof. Gyre or Cullgen’s
plans, objectives, goals, strategies, future events, or intentions relating to Gyre or Cullgen’s products and markets, the safety, efficacy and clinical benefits of Gyre or Cullgen’s product candidates, the anticipated timing and design of any
planned and ongoing preclinical studies and clinical trials, Gyre or Cullgen’s research and development efforts, plans and objectives of management for future operations and future results of anticipated product development efforts, potential
addressable market size and Gyre or Cullgen’s liquidity and capital resources and business trends. In some cases, you can identify forward-looking statements by terms such as “believe,” “can,” “could,” “anticipate”, “design,” “estimate,”
“expect,” “forecast,” “intend,” “may,” “might,” “plan,” “target”, “potential,” “predict,” “objective,” “should,” “strategy,” “will,” “would,” “forthcoming,” or the negative of these terms, and similar expressions that are predictions of or
indicate future events and future trends. These forward-looking statements may include express or implied statements relating to: the estimated future financial performance, financial position and financial impacts of the Merger; the
satisfaction of closing conditions to the Merger; the timing of completion of the Merger; the synergies that may be achieved between Gyre and Cullgen in connection with the Merger; the synergies that may be achieved between Gyre and Cullgen in
connection with the Merger; the therapeutic potential and utility, efficacy and clinical benefits of the product candidates of the combined company, including for the treatment of fibrosis, pain and solid tumors; the risk/benefit profile of the
product candidates of the combined company, including the potential of CG001419 to reduce the risk of addiction associated with other pharmaceutical therapies for the treatment of pain; expectations regarding Gyre or Cullgen’s research and
development efforts, including timing of initiation of Phase 2 trials for the product candidates of the combined company; Gyre or Cullgen’s expectations regarding the advancement of product candidates into IND-enabling studies; and Gyre and
Cullgen’s expectations, hopes, beliefs, intentions and strategies; and other statements that are not historical fact. These statements involve known and unknown risks, uncertainties and other factors that could cause Gyre or Cullgen’s actual
results to differ materially from the forward-looking statements expressed or implied in this presentation, in addition to those risks and uncertainties, such as the uncertainties inherent in the clinical drug development process, the
regulatory approval process, the timing of any regulatory filings, the potential for substantial delays, the risk that earlier study results may not be predictive of future study results, manufacturing risks, competition from other therapies or
products and the impacts of current macroeconomic and geopolitical risks. A discussion of these and other factors, is set forth in Gyre’s Annual Report on Form 10-K for the year ended December 31, 2024 filed with the Securities and Exchange
Commission (the”SEC”) on March 17, 2025 and elsewhere in such other filings and in Gyre’s periodic reports and subsequent disclosure documents filed with the SEC. Gyre and Cullgen cannot assure you that it will realize the results, benefits or
developments that it expects or anticipates or, even if substantially realized, that they will result in the consequences or affect Gyre or Cullgen or its business in the way expected. Forward-looking statements are not historical facts and
reflect management’s current views with respect to future events. Given the significant uncertainties, you should evaluate all forward-looking statements in the context of these risks and uncertainties and not place undue reliance on these
forward-looking statements as predictions of future events. All forward-looking statements in this presentation apply only as of the date made and are expressly qualified in their entirety by the cautionary statements included in this
presentation. Gyre and Cullgen have no intention to publicly update or revise any forward-looking statements to reflect subsequent events or circumstances, except as required by law. Certain information contained in this presentation and
statements made orally during this presentation relate to or is based on studies, publications, surveys and other data obtained from third-party sources and Gyre or Cullgen’s own internal estimates and research. While Gyre and Cullgen believe
these third-party studies, publications, surveys and other data to be reliable as of the date of this presentation, it has not independently verified, and makes no representation as to the adequacy, fairness, accuracy or completeness of, any
information obtained from third-party sources. In addition, no independent sources have evaluated the reasonableness or accuracy of Gyre or Cullgen’s internal estimates or research, and no reliance should be made on any information or
statements made in this presentation relating to or based on such internal estimates and research. This presentation contains, trademarks, trade names and service marks of other companies which are the property of their respective owners. This
presentation concerns a discussion of investigational drugs that are under preclinical and/or clinical investigation, and which have not yet been approved for marketing by the U.S. Food and Drug Administration. They are currently limited by
Federal law to investigational use, and no representations are made as to their safety or effectiveness for the purposes for which they are being investigated. Forward-looking Statements

Gyre Therapeutics: At-A-Glance
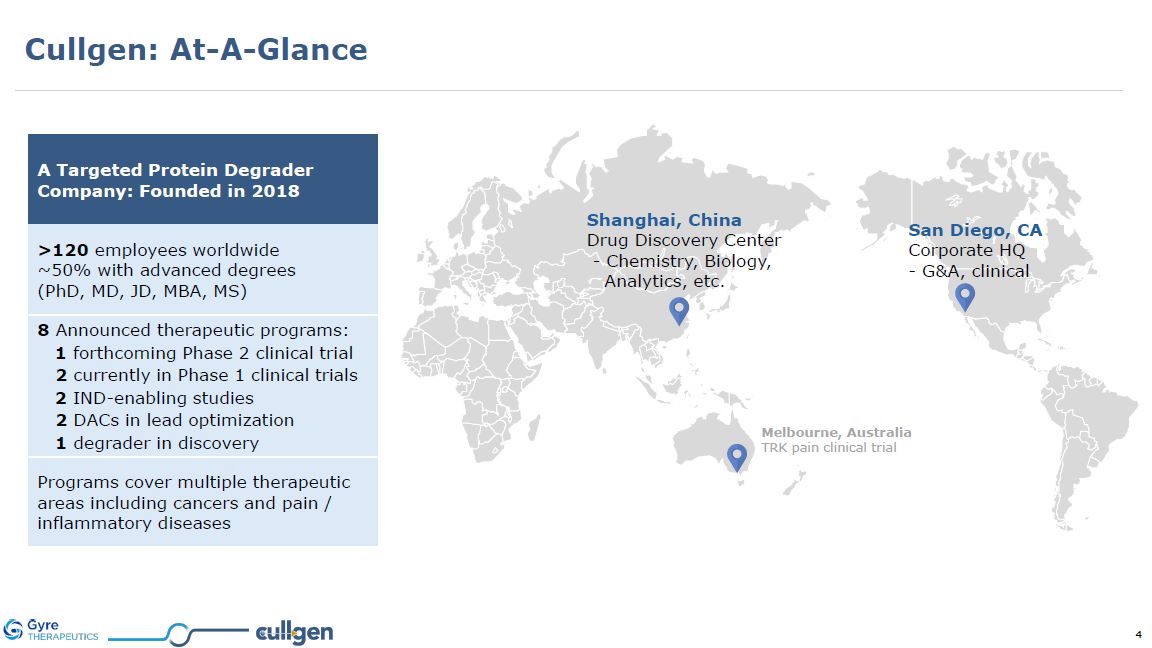
A Targeted Protein Degrader Company: Founded in 2018 >120 employees
worldwide ~50% with advanced degrees (PhD, MD, JD, MBA, MS) 8 Announced therapeutic programs: 1 forthcoming Phase 2 clinical trial 2 currently in Phase 1 clinical trials 2 IND-enabling studies 2 DACs in lead optimization 1 degrader in
discovery Programs cover multiple therapeutic areas including cancers and pain / inflammatory diseases San Diego, CA Corporate HQ - G&A, clinical Shanghai, China Drug Discovery Center - Chemistry, Biology, Analytics,
etc. Melbourne, Australia TRK pain clinical trial Cullgen: At-A-Glance
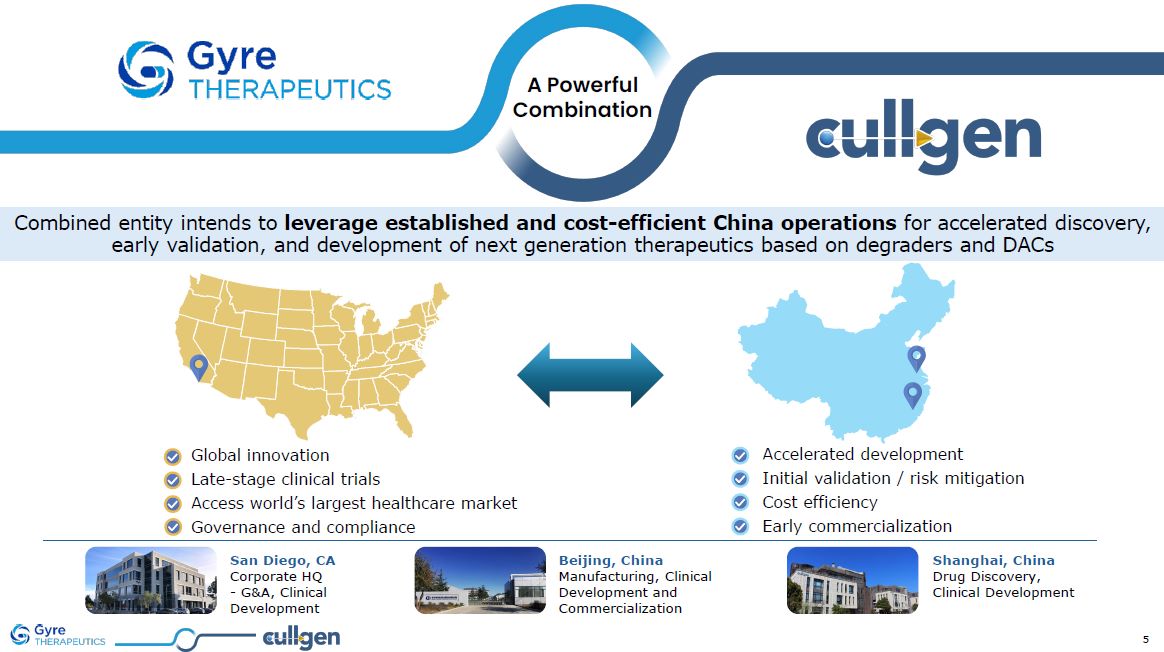
Cullgen Inc. Combined entity intends to leverage established and
cost-efficient China operations for accelerated discovery, early validation, and development of next generation therapeutics based on degraders and DACs San Diego, CA Corporate HQ - G&A, Clinical Development Shanghai, China Drug
Discovery, Clinical Development Beijing, China Manufacturing, Clinical Development and Commercialization A Powerful Combination Accelerated development Initial validation / risk mitigation Cost efficiency Early
commercialization Global innovation Late-stage clinical trials Access world’s largest healthcare market Governance and compliance
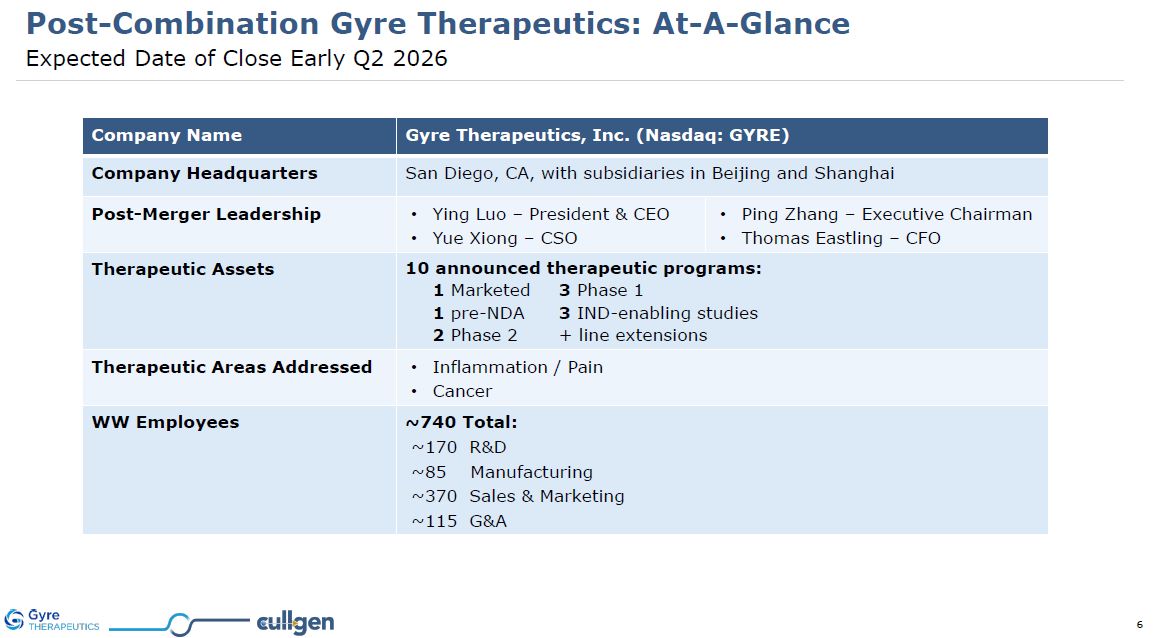
Post-Combination Gyre Therapeutics: At-A-Glance Expected Date of Close Early Q2
2026 Company Name Gyre Therapeutics, Inc. (Nasdaq: GYRE) Company Headquarters San Diego, CA, with subsidiaries in Beijing and Shanghai Post-Merger Leadership Ying Luo – President & CEO Yue Xiong – CSO Ping Zhang – Executive
Chairman Thomas Eastling – CFO Therapeutic Assets 10 announced therapeutic programs: 1 Marketed 3 Phase 1 1 pre-NDA 3 IND-enabling studies 2 Phase 2 + line extensions Therapeutic Areas Addressed Inflammation / Pain Cancer WW
Employees ~740 Total: ~170 R&D ~85 Manufacturing ~370 Sales & Marketing ~115 G&A
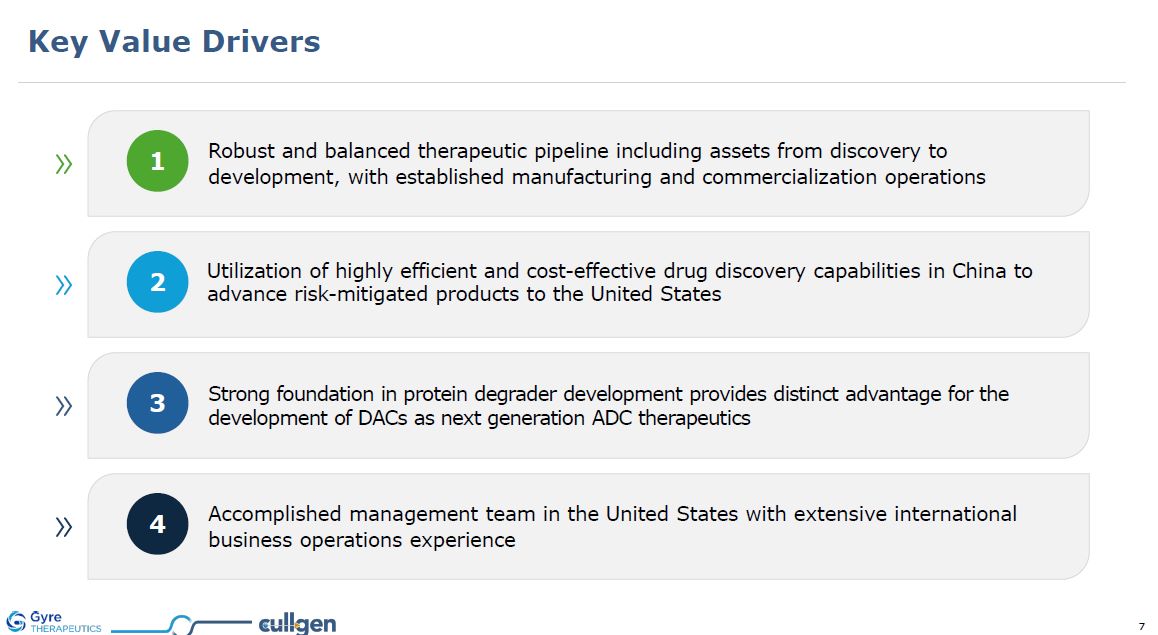
Key Value Drivers 2 3 1 Robust and balanced therapeutic pipeline including
assets from discovery to development, with established manufacturing and commercialization operations 4 Utilization of highly efficient and cost-effective drug discovery capabilities in China to advance risk-mitigated products to the United
States Accomplished management team in the United States with extensive international business operations experience Strong foundation in protein degrader development provides distinct advantage for the development of DACs as next generation
ADC therapeutics

Broad Product Portfolio from Discovery to Commercialization
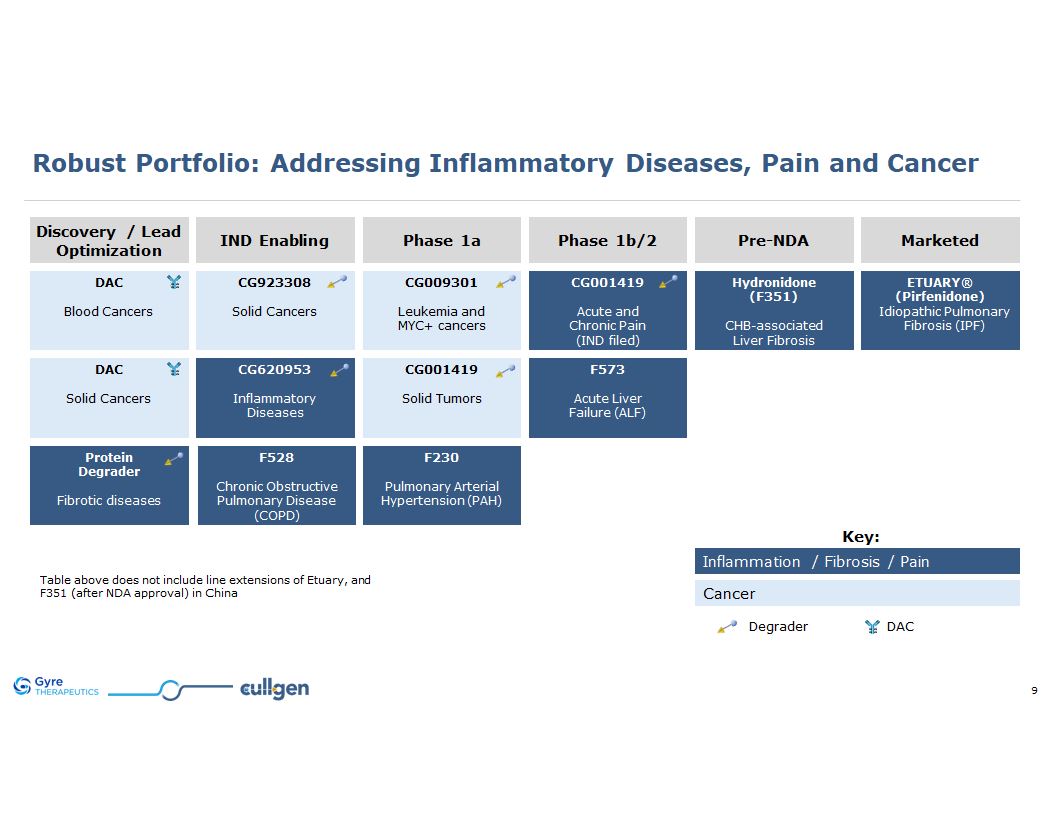
Key: Inflammation / Fibrosis / Pain Cancer Robust Portfolio: Addressing
Inflammatory Diseases, Pain and Cancer Discovery / Lead Optimization IND Enabling Phase 1a Phase 1b/2 Pre-NDA Marketed ETUARY® (Pirfenidone) Idiopathic Pulmonary Fibrosis (IPF) F528 Chronic Obstructive Pulmonary
Disease (COPD) CG001419 Acute and Chronic Pain (IND filed) CG009301 Leukemia and MYC+ cancers CG620953 Inflammatory Diseases F573 Acute Liver Failure (ALF) Hydronidone (F351) CHB-associated Liver Fibrosis F230 Pulmonary
Arterial Hypertension (PAH) CG923308 Solid Cancers DAC Blood Cancers Protein Degrader Fibrotic diseases CG001419 Solid Tumors DAC Solid Cancers Table above does not include the following line extensions: ETUARY for DKD ETUARY for
RILI ETUARY for PD F351 for MASH Degrader DAC
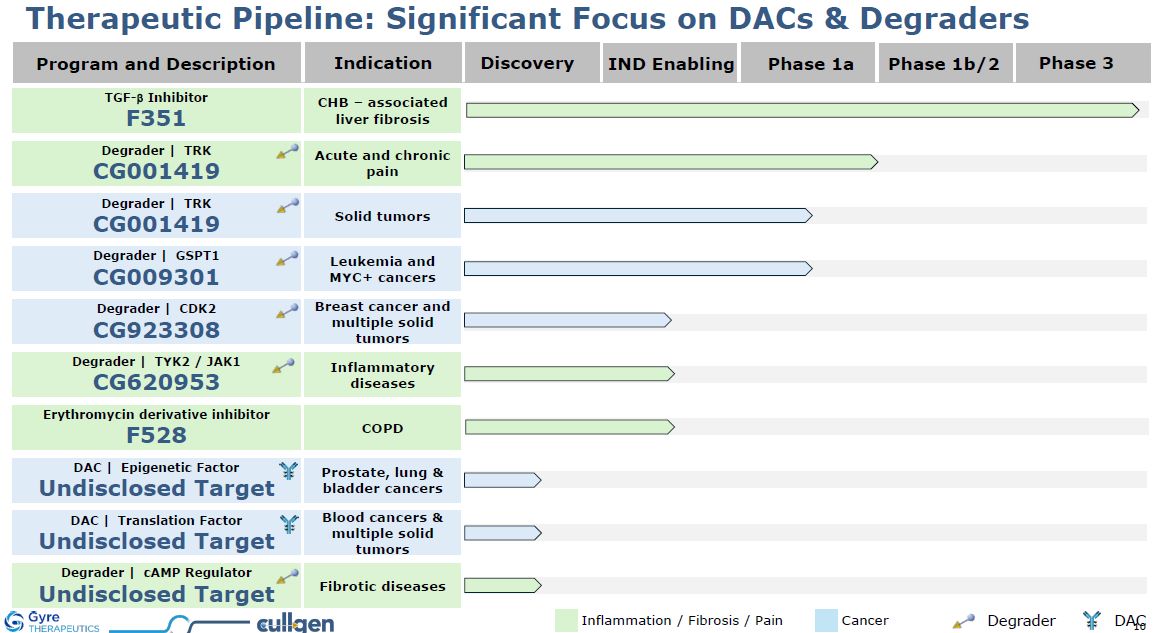
Program and Description Discovery IND Enabling Phase 1a Phase 1b/2 Phase
3 Indication DAC | Epigenetic Factor Undisclosed Target Prostate, lung & bladder cancers Degrader | TRK CG001419 Acute and chronic pain Degrader | TRK CG001419 Solid tumors Degrader | GSPT1 CG009301 Leukemia and MYC+
cancers Degrader | CDK2 CG923308 Breast cancer and multiple solid tumors Degrader | TYK2 / JAK1 CG620953 Inflammatory diseases DAC | Translation Factor Undisclosed Target Blood cancers & multiple solid tumors Degrader | cAMP
Regulator Undisclosed Target Fibrotic diseases Therapeutic Pipeline: Significant Focus on DACs & Degraders TGF-b Inhibitor F351 CHB – associated liver fibrosis Inflammation / Fibrosis / Pain Cancer Erythromycin derivative
inhibitor F528 COPD Degrader DAC

TPD and DAC Platform Technology to Revolutionize Drug Discovery
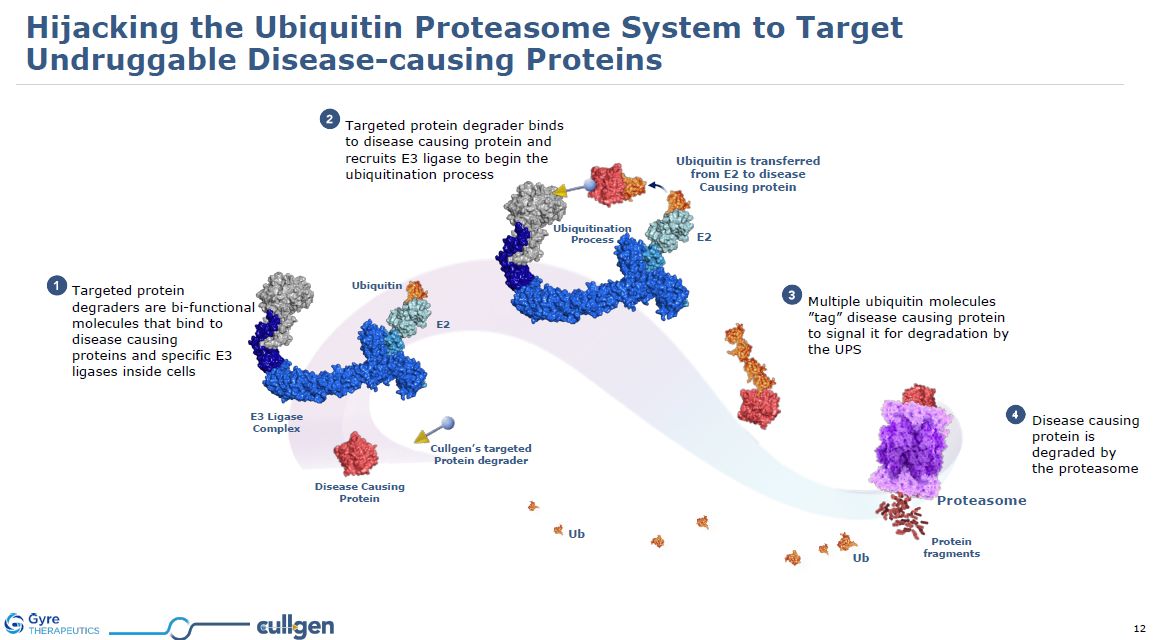
Targeted protein degraders are bi-functional molecules that bind to disease
causing proteins and specific E3 ligases inside cells Targeted protein degrader binds to disease causing protein and recruits E3 ligase to begin the ubiquitination process Multiple ubiquitin molecules ”tag” disease causing protein to signal
it for degradation by the UPS Disease causing protein is degraded by the proteasome Protein fragments Ub Ub Ubiquitin is transferred from E2 to disease Causing protein Cullgen’s targeted Protein
degrader Proteasome Ubiquitination Process E2 Disease Causing Protein E3 Ligase Complex Ubiquitin E2 Hijacking the Ubiquitin Proteasome System to Target Undruggable Disease-causing Proteins

Degrader-Antibody Conjugates (DACs) Combine the Advantages of ADCs and TPD and
Represent the Next-Generation of ADCs ADC (Antibody drug conjugate) DAC (Degrader antibody conjugate) TPD (Targeted protein degradation) E3 ligase E3 ligase Reduced toxicity through dual target selectivity at the cell surface
(antibody-tumor associated antigen) and intracellularly (degrader-target protein). The catalytic mechanism of action of TPDs ensures small quantity of degrader delivered by the antibody to achieve sufficient efficacy. Extended half-life,
reduced systemic clearance, improved solubility, and bypassing the need for oral bio-availability or cell permeability optimization. HIGH POTENCY IMPROVED pk IMPROVED SAFETY
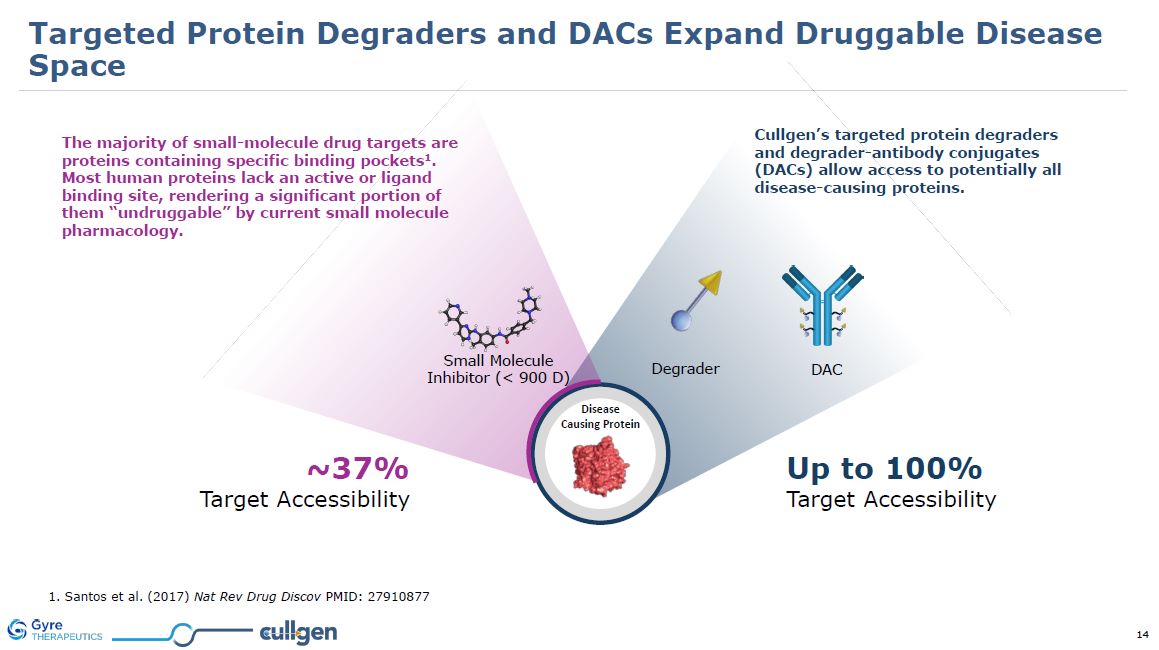
Disease Causing Protein 1. Santos et al. (2017) Nat Rev Drug Discov PMID:
27910877 Cullgen’s targeted protein degraders and degrader-antibody conjugates (DACs) allow access to potentially all disease-causing proteins. ~37% Target Accessibility Up to 100% Target Accessibility DAC Degrader Small
Molecule Inhibitor (< 900 D) The majority of small-molecule drug targets are proteins containing specific binding pockets1. Most human proteins lack an active or ligand binding site, rendering a significant portion of them “undruggable” by
current small molecule pharmacology. Targeted Protein Degraders and DACs Expand Druggable Disease Space
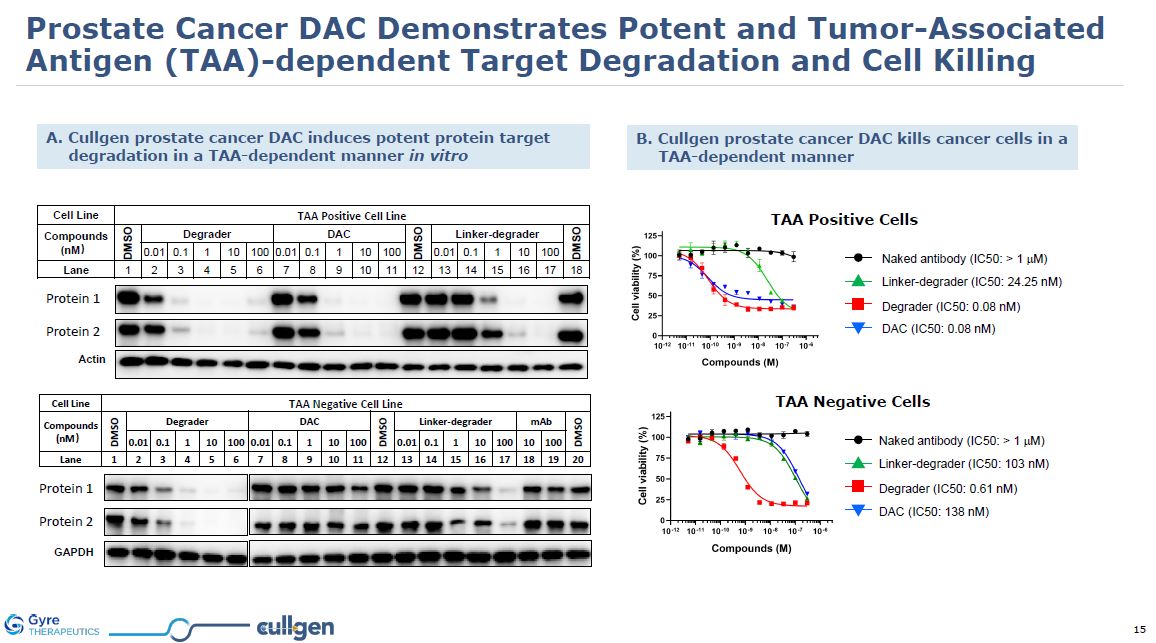
Cell Line TAA Positive Cell
Line Compounds (nM) DMSO Degrader DAC DMSO Linker-degrader DMSO 0.01 0.1 1 10 100 0.01 0.1 1 10 100 0.01 0.1 1 10 100 Lane 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 Cell Line TAA Negative Cell
Line Compounds (nM) DMSO Degrader DAC DMSO Linker-
degrader mAb DMSO 0.01 0.1 1 10 100 0.01 0.1 1 10 100 0.01 0.1 1 10 100 10 100 Lane 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 p300 CBP Actin p300 CBP GAPDH A. Cullgen prostate cancer DAC
induces potent protein target degradation in a TAA-dependent manner in vitro B. Cullgen prostate cancer DAC kills cancer cells in a TAA-dependent manner Linker-degrader (IC50: 24.25 nM) Naked antibody (IC50: > 1 mM) Degrader (IC50: 0.08
nM) DAC (IC50: 0.08 nM) TAA Positive Cells Linker-degrader (IC50: 103 nM) Naked antibody (IC50: > 1 mM) Degrader (IC50: 0.61 nM) DAC (IC50: 138 nM) TAA Negative Cells Protein 1 Protein 2 Protein 1 Protein 2 Prostate Cancer DAC
Demonstrates Potent and Tumor-Associated Antigen (TAA)-dependent Target Degradation and Cell Killing
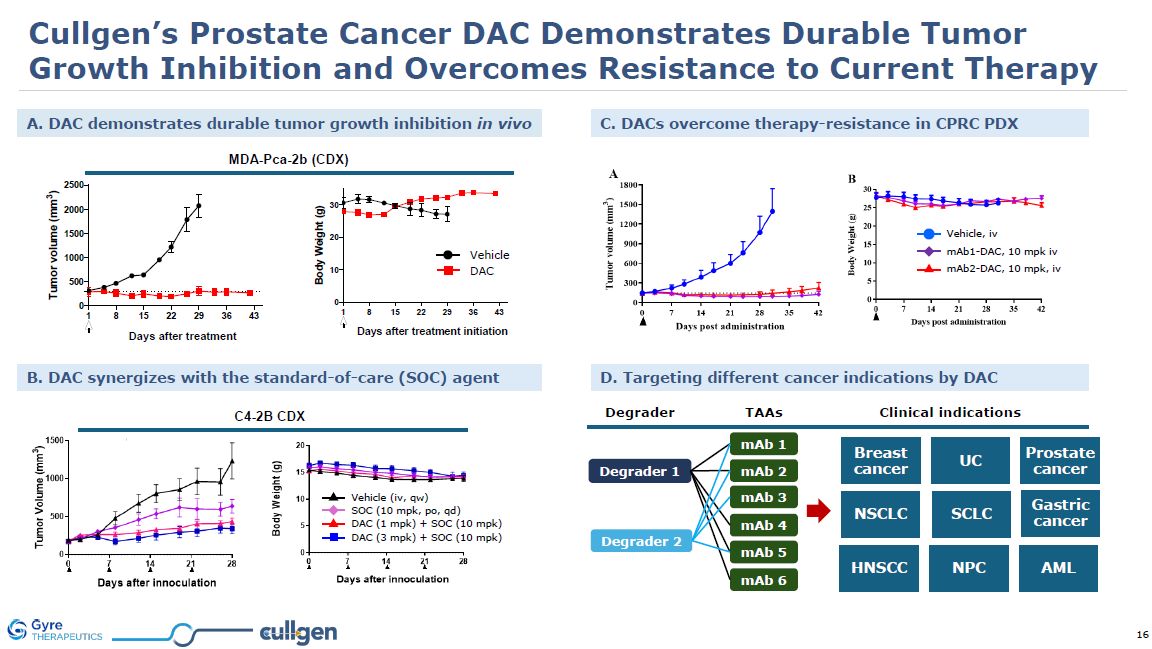
Cullgen’s Prostate Cancer DAC Demonstrates Durable Tumor Growth Inhibition and
Overcomes Resistance to Current Therapy MDA-Pca-2b (CDX) B. DAC synergizes with the standard-of-care (SOC) agent C. DACs overcome therapy-resistance in CPRC PDX A. DAC demonstrates durable tumor growth inhibition in
vivo DAC Vehicle mAb1-DAC, 10 mpk iv mAb2-DAC, 10 mpk, iv Vehicle, iv C4-2B CDX SOC (10 mpk, po, qd) Vehicle (iv, qw) DAC (1 mpk) + SOC (10 mpk) DAC (3 mpk) + SOC (10 mpk) D. Targeting different cancer indications by
DAC Degrader TAAs Degrader 1 Degrader 2 mAb 1 mAb 2 mAb 3 mAb 4 mAb 5 mAb 6 Clinical indications Breast cancer UC Prostate cancer SCLC Gastric cancer HNSCC NPC AML NSCLC

Leading Inflammation/Fibrosis Therapeutic ProgramsExpanding F351’s Global Market
PotentialTyk2 for Rheumatoid Diseases
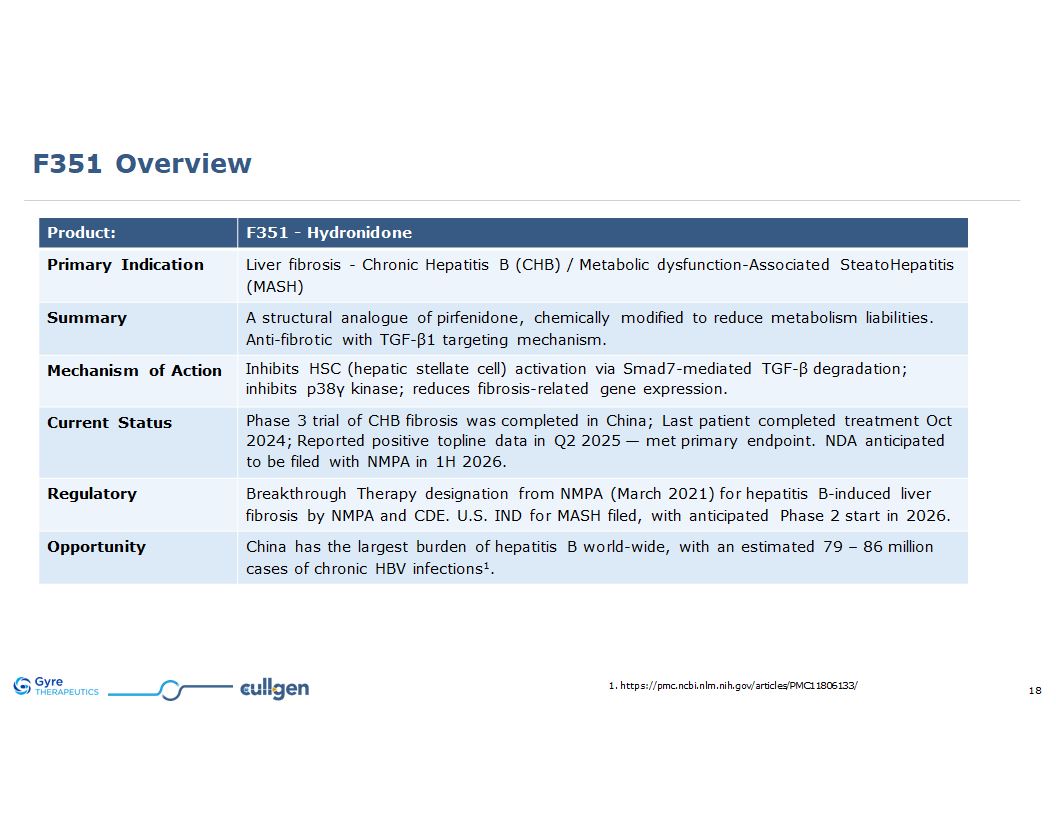
F351 Overview Product: F351 - Hydronidone Primary Indication Liver fibrosis -
Chronic Hepatitis B (CHB) / Metabolic dysfunction-Associated SteatoHepatitis (MASH) Summary A structural analogue of pirfenidone, chemically modified to reduce metabolism liabilities. Anti-fibrotic with TGF-β1 targeting mechanism. Mechanism
of Action Inhibits HSC (hepatic stellate cell) activation via Smad7-mediated TGF-β degradation; inhibits p38γ kinase; reduces fibrosis-related gene expression. Current Status Phase 3 trial of CHB currently on-going in China; Last patient
completed treatment Oct 2024; Reported positive topline data in Q2 2025 — met primary endpoint. NDA anticipated to be filed with NMPA in 1H 2026. Regulatory Breakthrough Therapy designation from NMPA (March 2021) for hepatitis B-induced liver
fibrosis by NMPA and CDE. U.S. IND for MASH filed, with anticipated Phase 2 start in 2026. Opportunity China has the largest burden of hepatitis B world-wide, with an estimated 79 – 86 million cases of chronic HBV infections1. 1.
https://pmc.ncbi.nlm.nih.gov/articles/PMC11806133/

Primary Endpoint Met with High Statistical Significance Key Secondary Endpoint
Reduction in Liver Inflammation Favorable Safety Profile ≥1-stage fibrosis regression at Week 52: Hydronidone: 52.85% (n=123) vs. Placebo: 29.84% (n=124) Delta: 23.01% P = 0.0002 (ITT1 analysis with central blinded pathology
review) Consistent with fibrosis regression rates observed in Phase 2 ≥1-grade inflammation improvement without fibrosis progression at Week 52: Hydronidone: 49.57% (n=123) vs. Placebo: 34.82% (n=124) Delta: 14.75% P =
0.0246 Reinforces anti-inflammatory activity Serious Adverse Events Hydronidrone: 4.88% (6/123) vs Placebo: 6.45% (8/124) No discontinuations, dose interruption or dose reduction due to adverse events F351 Phase 3 Results Demonstrate New
Global Potential in Liver Fibrosis and Cirrhosis Breakthrough Therapy Designation Priority Review of NDA (China NMPA, 2021, 2026) NDA expected to be filed with NMPA in 1H 2026
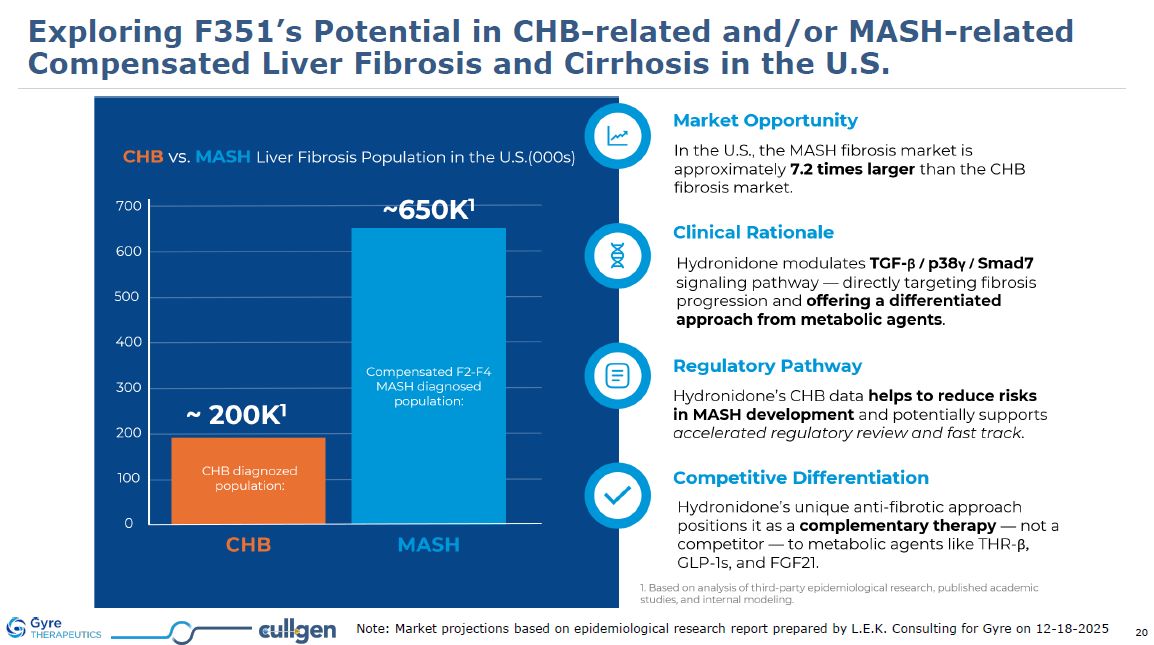
Exploring F351’s Potential in CHB-related and/or MASH-related Compensated Liver
Fibrosis and Cirrhosis in the U.S. Note: Market projections based on epidemiological research report prepared by L.E.K. Consulting for Gyre on 12-18-2025
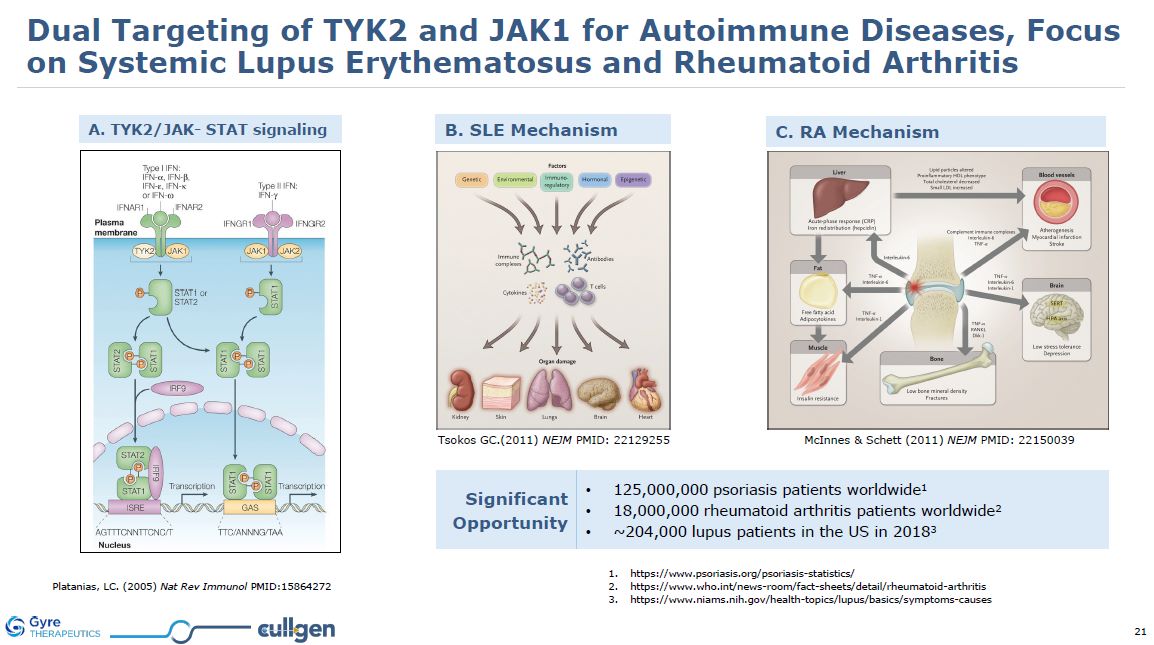
A. TYK2/JAK- STAT signaling Tsokos GC.(2011) NEJM PMID: 22129255 McInnes &
Schett (2011) NEJM PMID: 22150039 B. SLE Mechanism C. RA Mechanism Platanias, LC. (2005) Nat Rev Immunol PMID:15864272 Significant Opportunity 125,000,000 psoriasis patients worldwide1 18,000,000 rheumatoid arthritis patients
worldwide2 ~204,000 lupus patients in the US in
20183 https://www.psoriasis.org/psoriasis-statistics/ https://www.who.int/news-room/fact-sheets/detail/rheumatoid-arthritis https://www.niams.nih.gov/health-topics/lupus/basics/symptoms-causes Dual Targeting of TYK2 and JAK1 for Autoimmune
Diseases, Focus on Systemic Lupus Erythematosus and Rheumatoid Arthritis
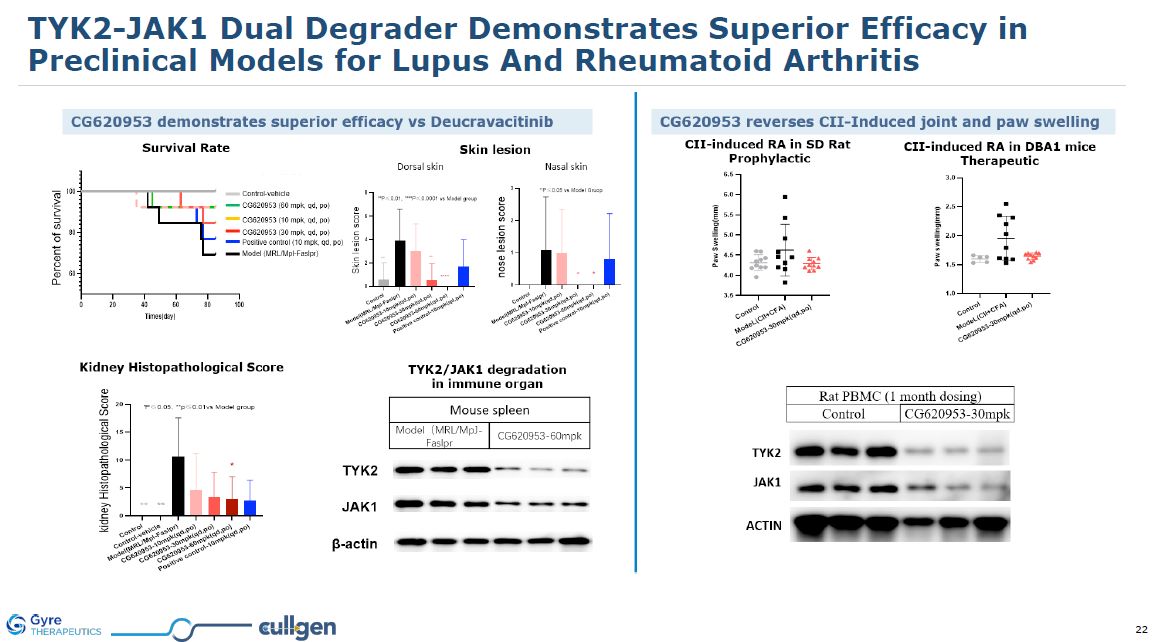
TYK2-JAK1 Dual Degrader Demonstrates Superior Efficacy in Preclinical Models for
Lupus And Rheumatoid Arthritis

Pain Therapeutic Program CG001419 for Acute and Chronic Pain
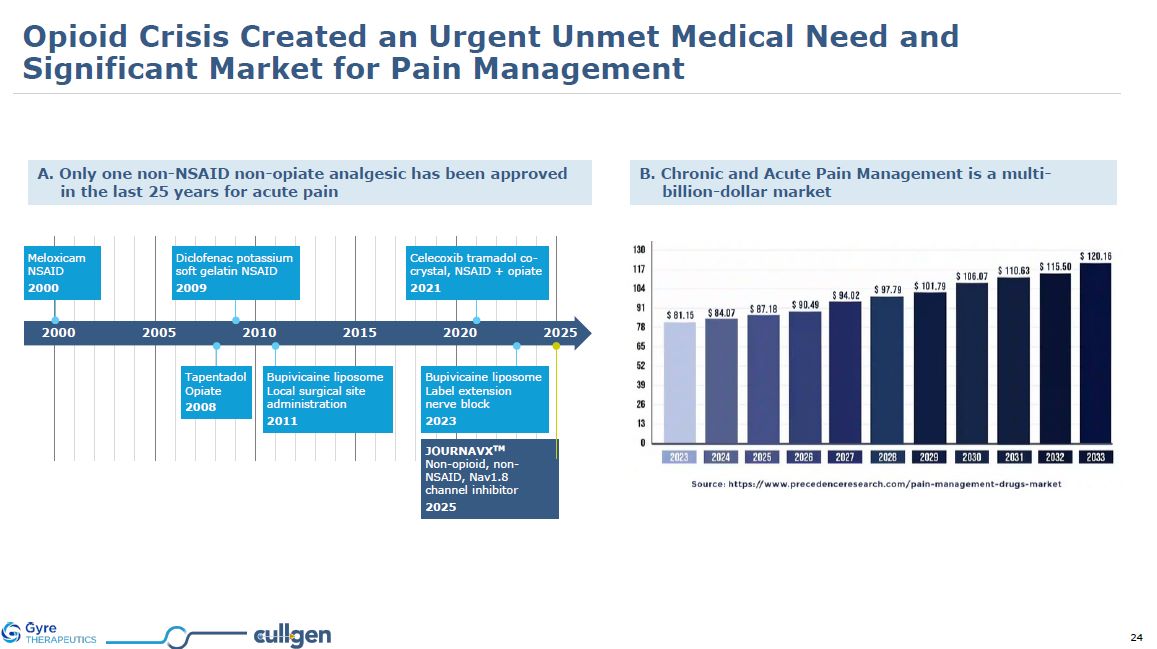
A. Only one non-NSAID non-opiate analgesic has been approved in the last 25 years
for acute pain B. Chronic and Acute Pain Management is a multi-billion-dollar market 2000 2005 2010 2015 2020 2025 Meloxicam NSAID 2000 Diclofenac potassium soft gelatin NSAID 2009 Tapentadol Opiate 2008 Bupivicaine liposome
Local surgical site administration 2011 Celecoxib tramadol co-crystal, NSAID + opiate 2021 Bupivicaine liposome Label extension nerve block 2023 JOURNAVXTM Non-opioid, non-NSAID, Nav1.8 channel inhibitor 2025 Opioid Crisis Created
an Urgent Unmet Medical Need and Significant Market for Pain Management
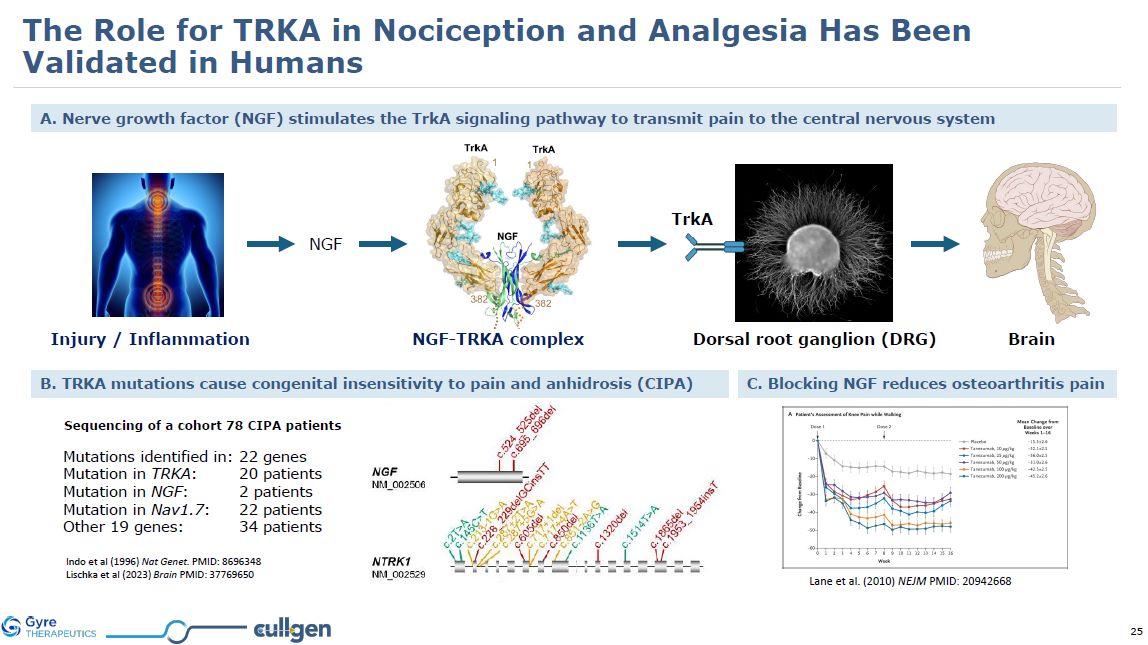
A. Nerve growth factor (NGF) stimulates the TrkA signaling pathway to transmit
pain to the central nervous system TrkA Dorsal root ganglion (DRG) NGF Injury / Inflammation Brain NGF-TRKA complex Lane et al. (2010) NEJM PMID: 20942668 C. Blocking NGF reduces osteoarthritis pain B. TRKA mutations cause congenital
insensitivity to pain and anhidrosis (CIPA) Mutations identified in: 22 genes Mutation in TRKA: 20 patients Mutation in NGF: 2 patients Mutation in Nav1.7: 22 patients Other 19 genes: 34 patients Indo et al (1996) Nat Genet. PMID:
8696348 Lischka et al (2023) Brain PMID: 37769650 Sequencing of a cohort 78 CIPA patients The Role for TRKA in Nociception and Analgesia Has Been Validated in Humans
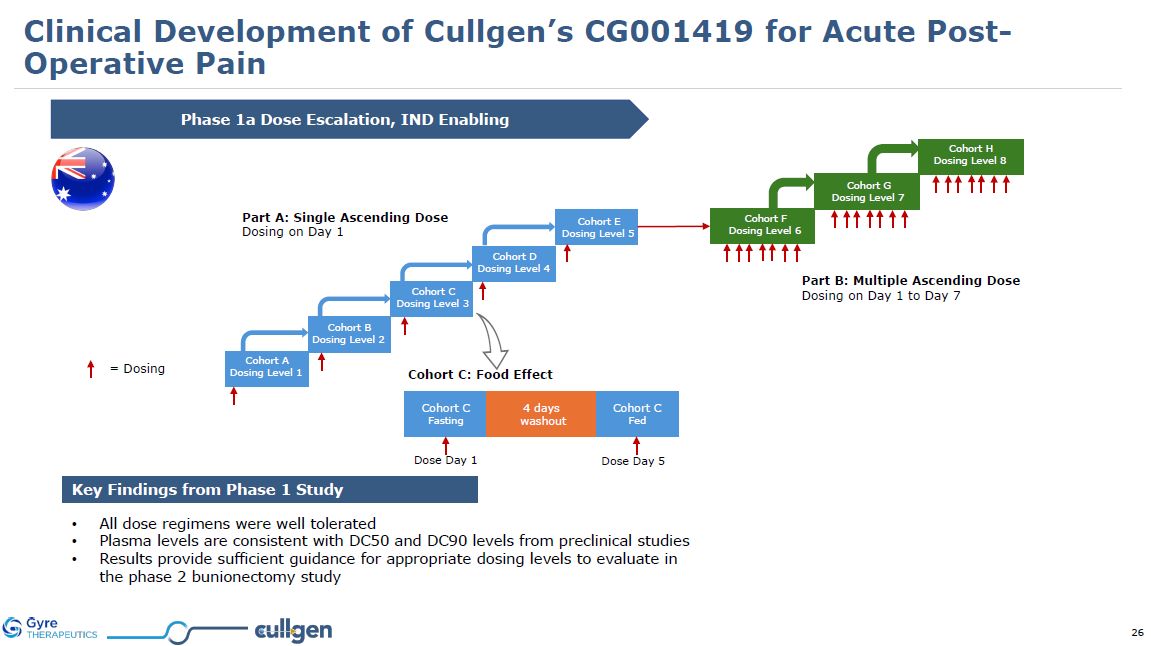
Phase 1a Dose Escalation, IND Enabling Key Findings from Phase 1 Study All dose
regimens were well tolerated Plasma levels are consistent with DC50 and DC90 levels from preclinical studies Results provide sufficient guidance for appropriate dosing levels to evaluate in the phase 2 bunionectomy study = Dosing Part
A: Single Ascending Dose Dosing on Day 1 Part B: Multiple Ascending Dose Dosing on Day 1 to Day 7 Cohort C: Food Effect Cohort A Dosing Level 1 Cohort B Dosing Level 2 Cohort C Dosing Level 3 Cohort D Dosing Level 4 Cohort
E Dosing Level 5 Cohort F Dosing Level 6 Cohort G Dosing Level 7 Cohort H Dosing Level 8 Cohort C Fasting Cohort C Fed 4 days washout Dose Day 1 Dose Day 5 Clinical Development of Cullgen’s CG001419 for Acute Post-Operative
Pain
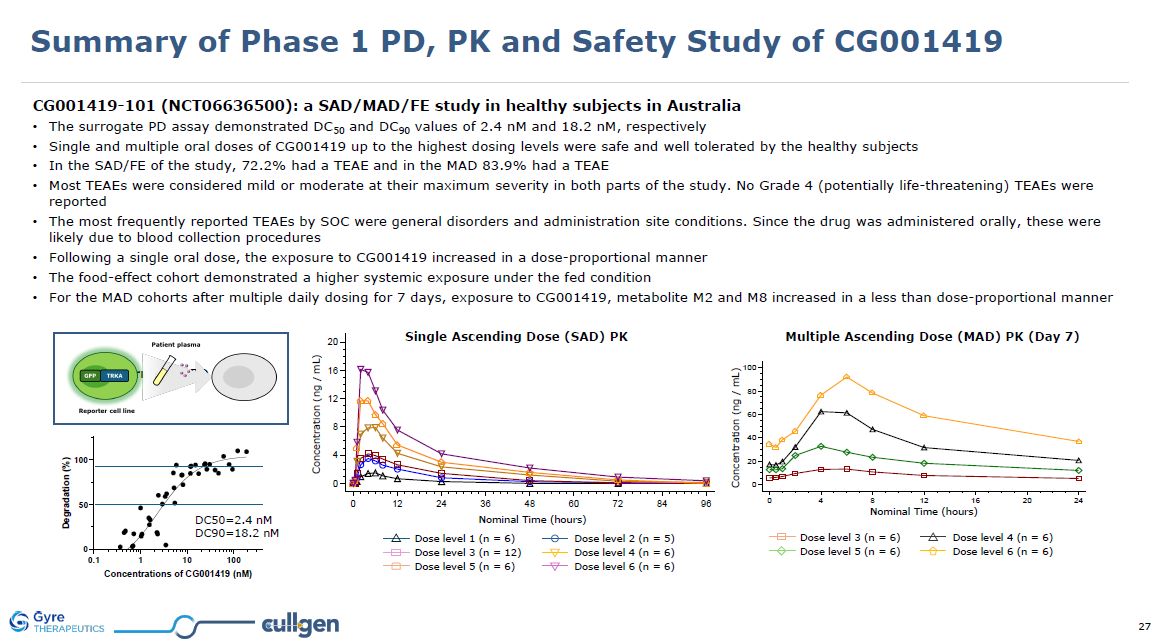
Summary of Phase 1 PD, PK and Safety Study of CG001419 CG001419-101
(NCT06636500): a SAD/MAD/FE study in healthy subjects in Australia The surrogate PD assay demonstrated DC50 and DC90 values of 2.4 nM and 18.2 nM, respectively Single and multiple oral doses of CG001419 up to the highest dosing levels were
safe and well tolerated by the healthy subjects In the SAD/FE of the study, 72.2% had a TEAE and in the MAD 83.9% had a TEAE Most TEAEs were considered mild or moderate at their maximum severity in both parts of the study. No Grade 4
(potentially life-threatening) TEAEs were reported The most frequently reported TEAEs by SOC were general disorders and administration site conditions. Since the drug was administered orally, these were likely due to blood collection
procedures Following a single oral dose, the exposure to CG001419 increased in a dose-proportional manner The food-effect cohort demonstrated a higher systemic exposure under the fed condition For the MAD cohorts after multiple daily dosing
for 7 days, exposure to CG001419, metabolite M2 and M8 increased in a less than dose-proportional manner Single Ascending Dose (SAD) PK Dose level 1 (n = 6) Dose level 3 (n = 12) Dose level 5 (n = 6) Dose level 2 (n = 5) Dose level 4 (n
= 6) Dose level 6 (n = 6) Nominal Time (hours) Concentration (ng / mL) Multiple Ascending Dose (MAD) PK (Day 7) Dose level 5 (n = 6) Dose level 3 (n = 6) Dose level 6 (n = 6) Dose level 4 (n = 6) Nominal Time (hours) Concentration (ng
/ mL) DC50=2.4 nM DC90=18.2 nM Surrogate PD assay
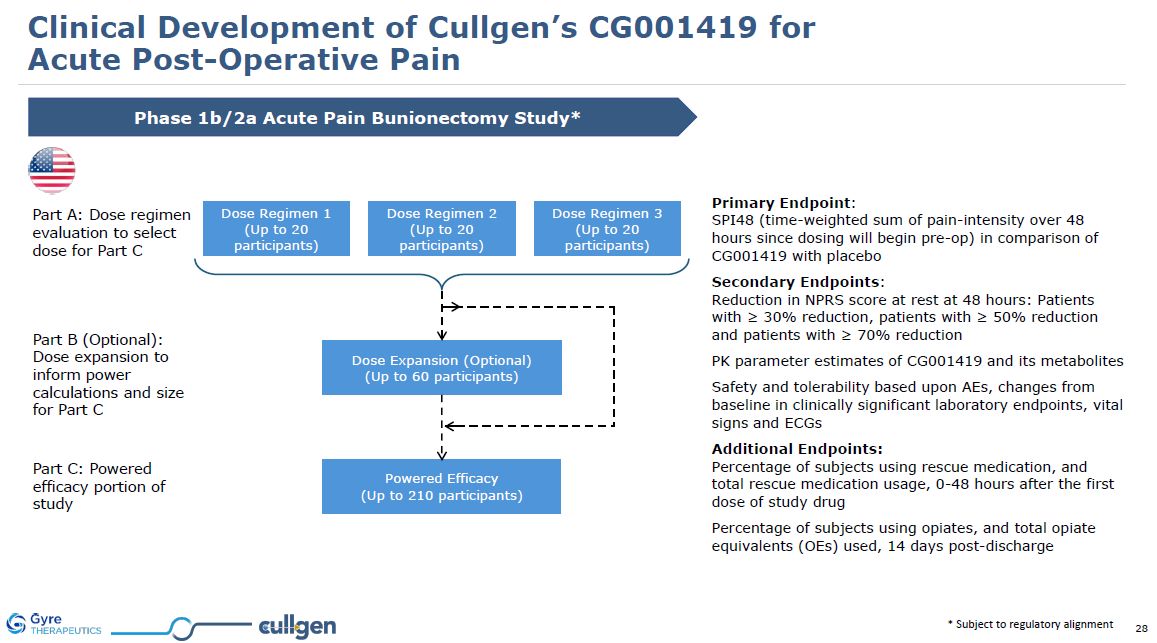
* Subject to regulatory alignment Phase 1b/2a Acute Pain Bunionectomy
Study* Part A: Dose regimen evaluation to select dose for Part C Part B (Optional): Dose expansion to inform power calculations and size for Part C Part C: Powered efficacy portion of study Primary Endpoint: SPI48 (time-weighted sum of
pain-intensity over 48 hours since dosing will begin pre-op) in comparison of CG001419 with placebo Secondary Endpoints:Reduction in NPRS score at rest at 48 hours: Patients with ≥ 30% reduction, patients with ≥ 50% reduction and patients with
≥ 70% reduction PK parameter estimates of CG001419 and its metabolites Safety and tolerability based upon AEs, changes from baseline in clinically significant laboratory endpoints, vital signs and ECGs Additional Endpoints:Percentage of
subjects using rescue medication, and total rescue medication usage, 0-48 hours after the first dose of study drug Percentage of subjects using opiates, and total opiate equivalents (OEs) used, 14 days post-discharge Clinical Development of
Cullgen’s CG001419 for Acute Post-Operative Pain Dose Regimen 1 (Up to 20 participants) Dose Regimen 2 (Up to 20 participants) Dose Regimen 3 (Up to 20 participants) Dose Expansion (Optional) (Up to 60 participants) Powered
Efficacy (Up to 210 participants)
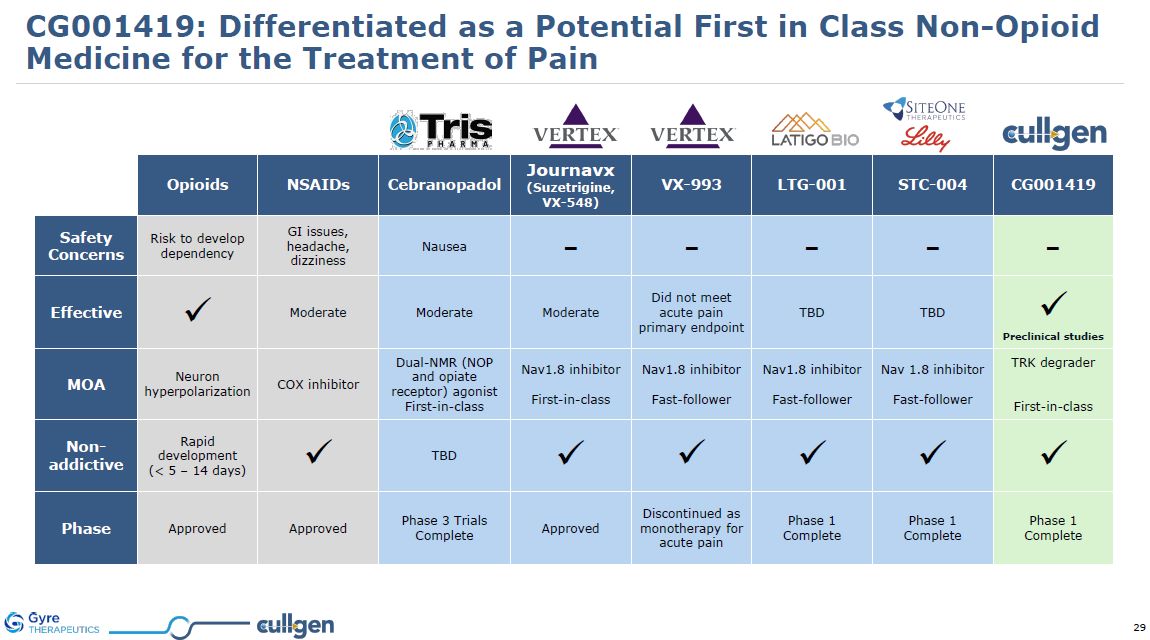
CG001419: Differentiated as a Potential First in Class Non-Opioid Medicine for the
Treatment of Pain Opioids NSAIDs Cebranopadol Journavx (Suzetrigine, VX-548) VX-993 LTG-001 STC-004 CG001419 Safety Concerns Risk to develop dependency GI issues, headache,
dizziness Nausea - - - - - Effective Moderate Moderate Moderate Did not meet acute pain primary endpoint TBD TBD Preclinical studies MOA Neuron hyperpolarization COX inhibitor Dual-NMR (NOP and opiate receptor)
agonist First-in-class Nav1.8 inhibitor First-in-class Nav1.8 inhibitor Fast-follower Nav1.8 inhibitor Fast-follower Nav 1.8 inhibitor Fast-follower TRK degrader First-in-class Non-addictive Rapid development (< 5 – 14
days) TBD Phase Approved Approved Phase 3 Trials Complete Approved Discontinued as monotherapy for acute pain Phase 1 Complete Phase 1 Complete Phase 1 Complete

Cancer ProgramsCG001419: for pan-TRK Cancers CG009301: GSPT1 Degrader for AML and
MYC+ Cancers
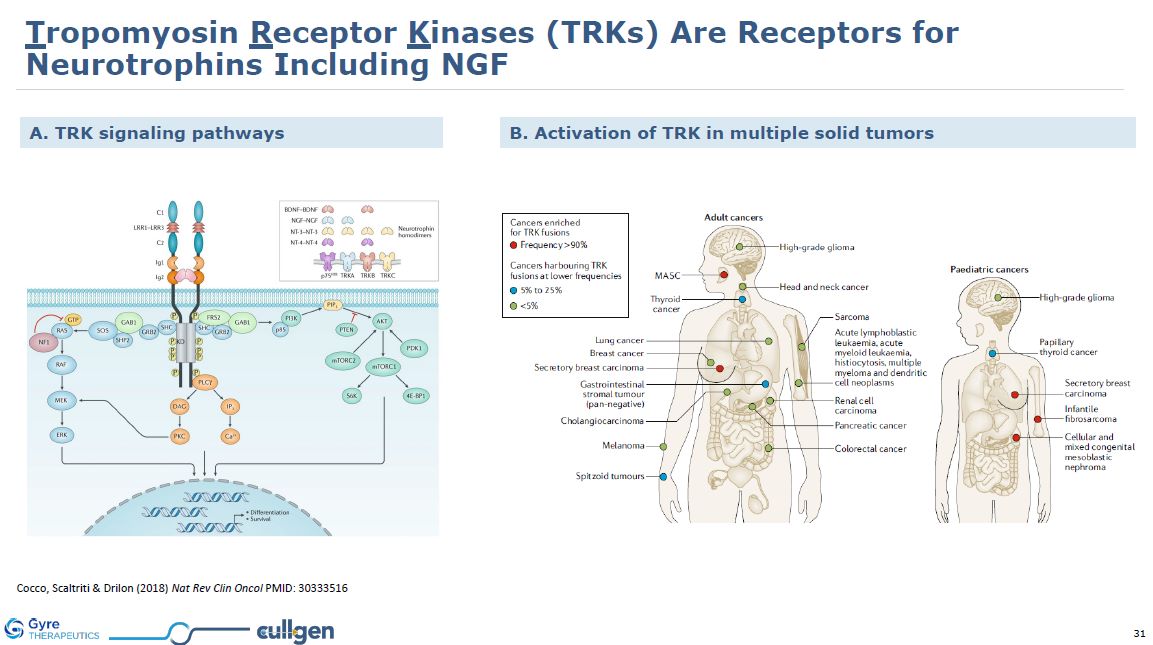
Cocco, Scaltriti & Drilon (2018) Nat Rev Clin Oncol PMID: 30333516 B.
Activation of TRK in multiple solid tumors A. TRK signaling pathways Tropomyosin Receptor Kinases (TRKs) Are Receptors for Neurotrophins Including NGF
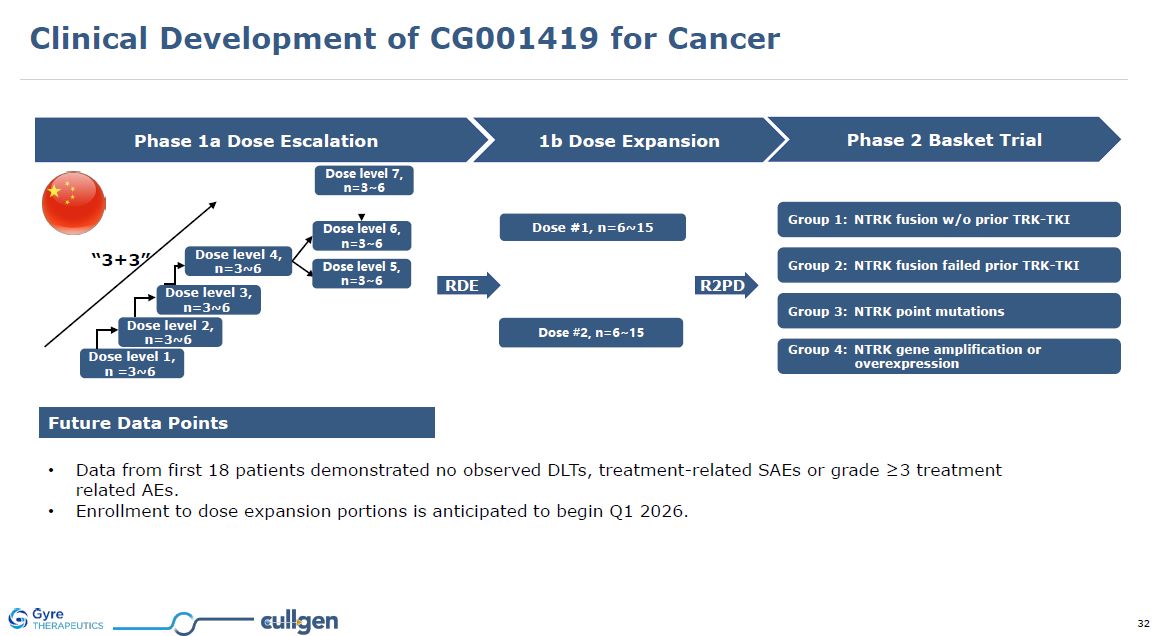
Future Data Points Data from first 18 patients demonstrated no observed DLTs,
treatment-related SAEs or grade ≥3 treatment related AEs. Enrollment to dose expansion portions is anticipated to begin Q1 2026. RDE Phase 1a Dose Escalation 1b Dose Expansion Phase 2 Basket Trial Dose level 1, n =3~6 Dose level 2,
n=3~6 Dose level 3, n=3~6 Dose level 4, n=3~6 “3+3” Group 1: NTRK fusion w/o prior TRK-TKI Group 2: NTRK fusion failed prior TRK-TKI Group 3: NTRK point mutations Group 4: NTRK gene amplification or overexpression Dose #1,
n=6~15 R2PD Dose #2, n=6~15 Dose level 5, n=3~6 Dose level 6, n=3~6 Dose level 7, n=3~6 Clinical Development of CG001419 for Cancer
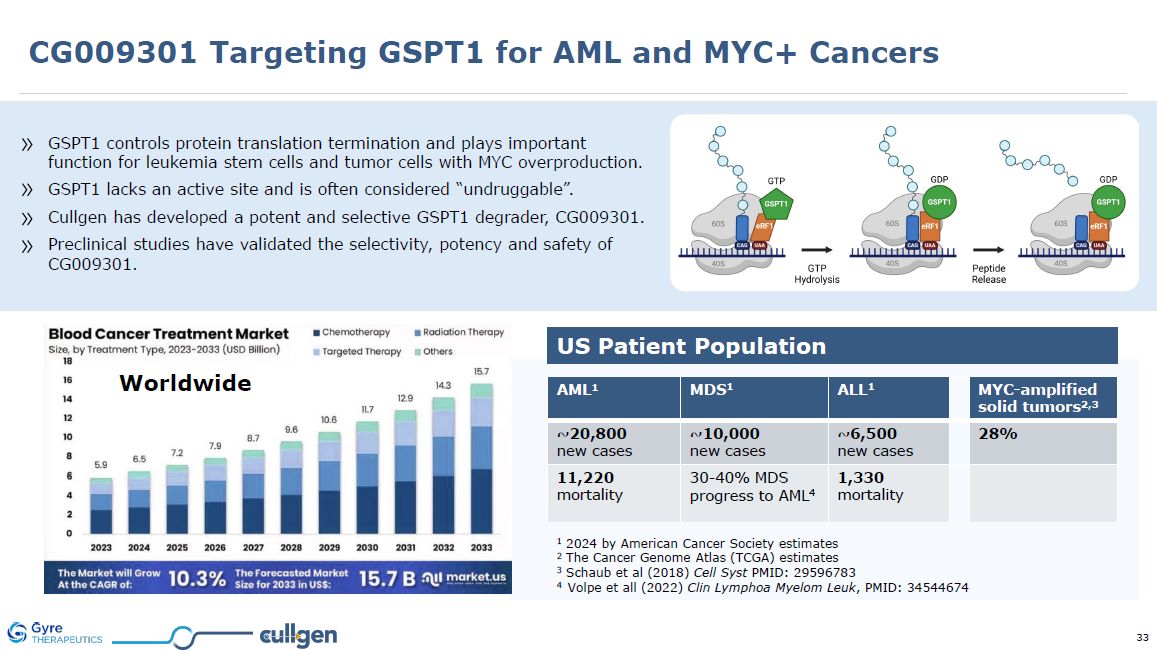
GSPT1 controls protein translation termination and plays important function for
leukemia stem cells and tumor cells with MYC overproduction. GSPT1 lacks an active site and is often considered “undruggable”. Cullgen has developed a potent and selective GSPT1 degrader, CG009301. Preclinical studies have validated the
selectivity, potency and safety of CG009301. US Patient Population 1 2024 by American Cancer Society estimates 2 The Cancer Genome Atlas (TCGA) estimates 3 Schaub et al (2018) Cell Syst PMID: 29596783 4 Volpe et all (2022) Clin Lymphoa
Myelom Leuk, PMID: 34544674 AML1 MDS1 ALL1 MYC-amplified solid tumors2,3 ~20,800 new cases ~10,000 new cases ~6,500 new cases 28% 11,220 mortality 30-40% MDS progress to AML4 1,330 mortality Worldwide CG009301 Targeting
GSPT1 for AML and MYC+ Cancers
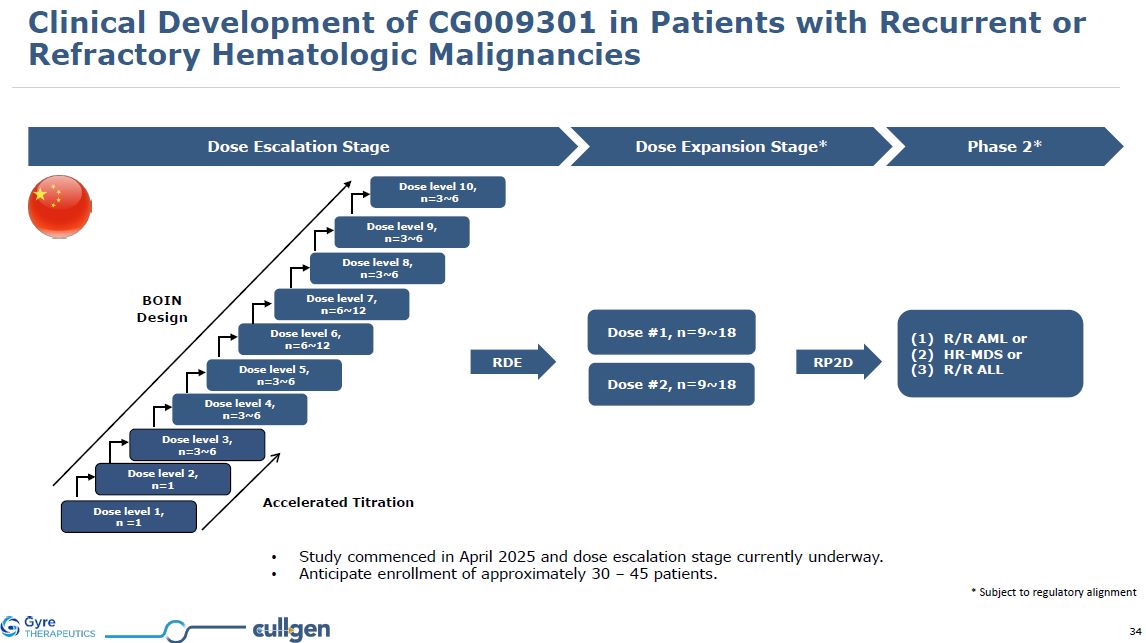
Dose Escalation Stage Dose Expansion Stage* Dose level 1, n =1 Dose level
2, n=1 Dose level 3, n=3~6 Dose level 6, n=6~12 Dose level 7, n=6~12 BOIN Design Dose #1, n=9~18 Dose #2, n=9~18 Dose level 4, n=3~6 Dose level 5, n=3~6 Accelerated Titration Phase 2* R/R AML or HR-MDS or R/R
ALL RDE Dose level 8, n=3~6 Dose level 9, n=3~6 Dose level 10, n=3~6 Study commenced in April 2025 and dose escalation stage currently underway. Anticipate enrollment of approximately 30 – 45 patients. * Subject to regulatory
alignment RP2D Clinical Development of CG009301 in Patients with Recurrent or Refractory Hematologic Malignancies
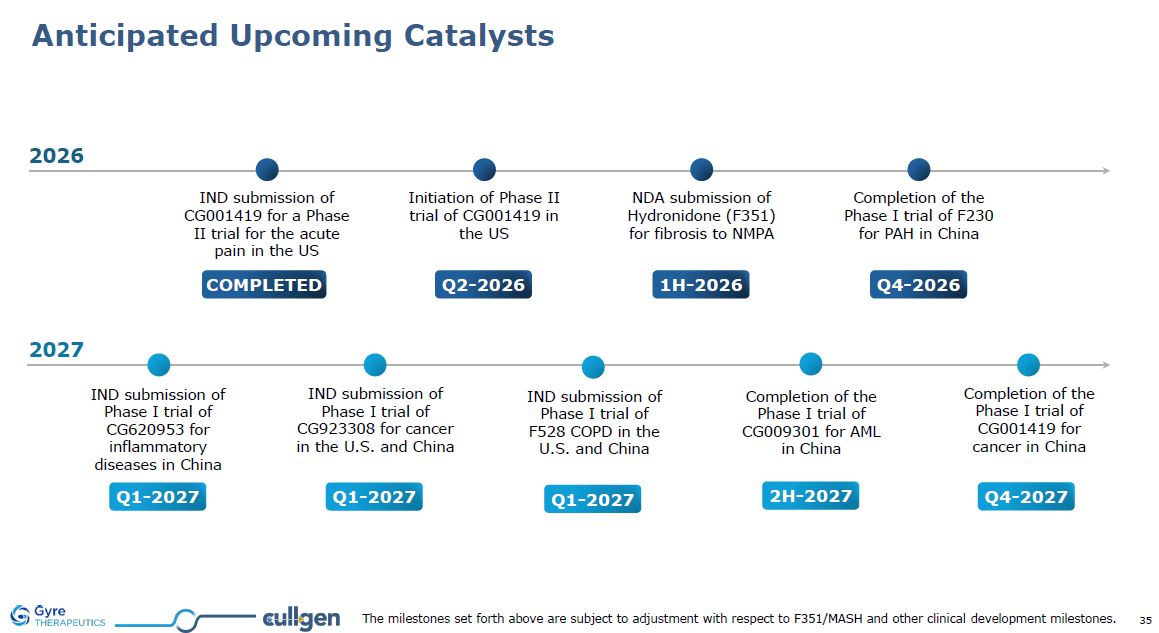
Anticipated Upcoming Catalysts Q1-2027 IND submission of Phase I trial of
CG620953 for inflammatory diseases in China 2H-2027 Completion of the Phase I trial of CG009301 for AML in China Q1-2027 IND submission of Phase I trial of CG923308 for cancer in the U.S. and China Q1-2027 IND submission of Phase I trial
of F528 COPD in the U.S. and China COMPLETED IND submission of CG001419 for a Phase II trial for the acute pain in the US Initiation of Phase II trial of CG001419 in the US Q2-2026 Q4-2026 Completion of the Phase I trial of F230 for PAH
in China 1H-2026 NDA submission of Hydronidone (F351) for fibrosis to NMPA 2026 2027 Q4-2027 Completion of the Phase I trial of CG001419 for cancer in China The milestones set forth above are subject to adjustment with respect to
F351/MASH and other clinical development milestones.

Investment Summary: Gyre’s Acquisition of Cullgen 2 3 1 Robust and balanced
therapeutic pipeline including assets from discovery to development, with established manufacturing and commercialization operations 4 Utilization of highly efficient and cost-effective drug discovery capabilities in China to advance
risk-mitigated products to the United States Accomplished management team in the United States with extensive international business operations experience Strong foundation in protein degrader development provides distinct advantage for the
development of DACs as next generation ADC therapeutics

Future Board of Combined Company Ping Zhang, MBA Executive Chairman 20 years
experience in healthcare investment with senior postings in Japan and China. Managing director of String Capital. Executive director of GNI Group Ltd. Renate Parry, PhD 25 years of research experience in global pharma. Developed 3 novel
drugs for oncology and fibrosis into clinical development Rodney Nussbaum, CPA Managing director of Atago Advisory. Former Senior Partner of E&Y Japan and Asia Pacific. Former partner of Arthur Anderson. Dan Weng, MD, MA CEO of Medelis.
Former CEO of EPS International. Held executive positions at MedPace, ICON, PharmaNet and Quintiles Ying Luo, PhD, CEO >30 years of biotech experience. PhD from U. Connecticut. Management at Aviron, Clontech, and Rigel. Founded Shanghai
Genomics and led GNI IPO (TSE 2160). >40 research articles and >20 patents. David Epstein, PhD Founder of PairX and Black Diamond (NASDAQ BDTX). Vice Dean of Duke-NUS Medical School at Singapore. CSO of OSI Pharmaceuticals. Developed
Izervay in Archemix Gordon Carmichael, PHD Professor of Genetics and Genome Sciences at the University of Connecticut Health Center. Published 110 papers. on kinase signaling in oncogenesis, transcriptional and post-transcriptional gene
regulation, long noncoding RNAs, stem cell biology, innate immunity, RNA modifications

Ying Luo, PhDPresident and CEO >30 years of biotech experience. PhD from U.
CT. President of GNI Group. Founded Shanghai Genomics and led GNI IPO (TSE 2160). Responsible for 6 IND approvals and 1 class 1 drug approval (Etuary) by China FDA. Author of >37 research articles and >20 patents. Yue Xiong,
PhDCSO William R. Kenan Distinguished Professor, UNC Chapel Hill. Pew Scholar. AACR Gertrude B. Elion Cancer Research Award. >220 papers. Discovery of Cyclin D, CKD4, p21, and ROC1/2. Thomas EastlingCFO >25 years experience in global
health care, financial services and investment banking, with senior postings in New York, London, Tokyo and China. Previously CFO of GNI Group Ltd. Future Leadership Team of Combined Company Ping Zhang, MBA Executive Chairman 20 years
experience in healthcare investment with senior postings in Japan and China. Managing director of String Capital. Executive director of GNI Group Ltd.

Thank Thank You