
1 Exhibit 10.52 CERTAIN CONFIDENTIAL INFORMATION IN THIS EXHIBIT HAS BEEN OMITTED AND REPLACED WITH “[***]” BECAUSE IT IS NOT MATERIAL AND WOULD BE COMPETITIVELY HARMFUL IF PUBLICLY DISCLOSED. COMMERCIAL SUPPLY AGREEMENT THIS COMMERCIAL SUPPLY AGREEMENT (this “Agreement”) is made and entered into this 18th day of December, 2024 (the “Effective Date”), by and between Madrigal Pharmaceuticals, Inc., a Delaware corporation having a principal place of business at Four Tower Bridge, 200 Barr Harbor Drive, Suite 200, West Conshohocken, Pennsylvania, 19428, U.S.A. (“Madrigal” or “Customer”), and Corden Pharma GmbH (“Corden”), a German company having a principal place of business at Otto Hahn Strasse 68723 Plankstadt Germany (each a “Party” and collectively, the “Parties”). Recitals WHEREAS, Customer is a pharmaceutical company engaged in the registration, manufacturing, marketing, promotion and distribution of pharmaceuticals product; WHEREAS, Customer is expecting to obtain a Health Registration and all necessary permits and approvals from all relevant authorities for the completion of certain Phase III clinical studies and /or marketing and sale of the Product in the Territory; WHEREAS, the Parties entered into a Master Services Agreement effective as of June 20, 2022 (the “MSA”) to cover the technology transfer of the Product to Corden and perform certain development and/or manufacturing services, including the process validation batches; WHEREAS, subject to the terms and conditions contained in this Agreement, Customer desires to engage Corden to Manufacture and supply the Product for commercialization in the Territory following the receipt of required Health Registrations (as those terms are defined below) as well as to provide certain ancillary services regarding the performance of stability studies, as well as other services related to support of commercial Manufacture and supply as agreed by the Parties. WHEREAS, Corden is willing to perform such work for Customer according to the terms and conditions provided for in this Agreement; NOW, THEREFORE, for and in consideration of the foregoing premises and of the mutual covenants of the Parties hereinafter set forth, the Parties hereto agree as follows:

2 Table of Content 1. DEFINITIONS ............................................................................................................................... 3 2. SCOPE OF AGREEMENT ......................................................................................................... 9 3. FORECAST, ORDERS ............................................................................................................. 11 4. MANUFACTURE OF PRODUCT ............................................................................................ 15 5. PAYMENTS, TAXES ................................................................................................................. 19 6. WARRANTIES ............................................................................................................................ 20 7. CHANGES TO PRODUCTS / CHANGE CONTROL ........................................................... 21 8. HEALTH REGISTRATION APPROVAL SUPPORT; REGULATORY MATTERS ........... 23 9. QUALITY ..................................................................................................................................... 24 10. RECALL, INDEMNIFICATION, INSURANCE, SECURITY MEASURES ..................... 25 11. CONFIDENTIALITY .............................................................................................................. 28 12. PROPERTY COMPLIANCE ................................................................................................ 29 13. TERM, TERMINATION ......................................................................................................... 30 14. LEGALCOMPLIANCE ........................................................................................................... 33 15. FORCE MAJEURE ................................................................................................................ 33 16. MISCELLANEOUS ................................................................................................................ 34

3 1. DEFINITIONS The following words, terms and phrases, when used herein, shall have the following respective meanings: 1.1 “Affiliate” means a company, whether a corporation or other business entity, that is controlling, controlled by or under common control with such Party. A corporation or other business entity shall be regarded as in “control” of another corporation or entity if it owns or controls, directly or indirectly, fifty percent (50%) or more of the share capital and/or voting rights of such entity or the power to direct or cause the direction of the management or policies of such other corporation or entity, whether through the ownership of voting securities, by contract or otherwise. 1.2 “Agreement” has the meaning set forth in the Preamble. 1.3 “API” means active pharmaceutical ingredient which refers to the chemical substance contained in the Product elucidating pharmacological activity or generating some direct effect in the diagnosis, treatment or prevention of a disease in a patient. The API for the Product is MGL-3196 (resmetirom), as described in Appendix 4. 1.4 “API Specifications” means the Specifications for the API set forth in Appendix 4. 1.5 “Applicable Law(s)” means all ordinances, rules, regulations, laws, guidelines, guidance, or other requirements of any Governmental Authority in the Territory that may be in effect from time to time and that are applicable to a Party and its activities performed pursuant to this Agreement in the country where such activity is performed, including, but not limited to compliance with cGMP and any environmental or biohazard laws in the country where the Manufacture takes place. 1.6 “Background IP” means all Intellectual Property owned, conceived, developed, first reduced to practice or otherwise made or acquired by a Party prior to the Effective Date hereof, including all modifications, adjustments or improvements thereto, or generated after the Effective Date without the use of any Intellectual Property of the other Party. 1.7 “Batch” means a specific quantity of Product Manufactured with uniform character and quality produced in one process or a series of processes so that it could be expected to be homogeneous within specified limits. 1.8 “Batch Record” or “Executed Batch Record” (EBR) means the complete production record documenting the Manufacture of one Batch, in accordance with the Master Batch Record and contains a compilation of records containing the Manufacturing history and control of a Product, including the Certificate of Analysis. These records are generated by Corden’s Quality Control and reviewed and approved by Quality Assurance and are described more fully in the Quality Agreement. 1.9 “Binding Forecast” has the meaning set forth in Section 3.1(b). 1.10 “Business Day” means any day other than a Saturday or Sunday or a day that is a statutory holiday in Germany or the United States of America. 1.11 “Calendar Quarter” means the respective periods of three (3) consecutive calendar months ending on March 31, June 30, September 30 and December 31; provided, however, that (a) the first Calendar Quarter of the Term shall extend from the Effective Date to the end of the first complete Calendar Quarter thereafter; and (b) the last Calendar Quarter of the Term shall end upon the expiration or termination of this Agreement. 1.12 “Calendar Year” means the twelve-month period ending on December 31; provided, however, that (a) the first Calendar Year of the Term shall begin on the Effective Date and end on December 31, 20244; and (b) the last Calendar Year of the Term shall end on the effective date of expiration or termination of this Agreement. 1.13 “Certificate of Analysis” means a document signed by an authorized representative of Corden for each Batch of Product that provides full analytical results of the Batch, including the testing

4 methods applied to the Batch, the corresponding acceptance criteria, and the results of such testing and certifies (a) the conformity of the Batch of Product to the Manufacturer’s Release Specifications and (b) that Manufacturing and release records of such Batch of Product were reviewed by Corden and Manufacturing and release of such Batch of Product is in accordance with the Manufacturing Standards. 1.14 “Certificate of Compliance” means a document signed by an authorized representative of Corden certifying that a particular Batch of the Product was Manufactured in accordance with cGMP, Applicable Law, and the Manufacturer’s Release Specifications. 1.15 “cGMP”, “GMP” or “Current Good Manufacturing Practices” means the regulatory requirements for the current good manufacturing practices in the United States Code of Federal Regulations 21 CFR Part 210 & Part 211, European Union (“EU”) EudraLex Volume 4 Good Manufacturing Guidelines, the MHLW GMP/GQP ordinances and according regulations in Japan, as applicable, and all applicable rules, regulations, promulgations, policies and guidelines in effect at any given time during the applicable Term. 1.16 “Claims” has the meaning set forth in Section 10.2. 1.17 “CMC” means the chemistry, manufacturing, and controls section(s) and data in the Health Registrations that cover the chemical composition of the Product and its components and the control and Manufacturing Process for the Product, as amended from time to time. 1.18 “Commercially Reasonable Efforts” means with respect to the efforts to be expended, or considerations to be undertaken, by a Party with respect to any objective, activity or decision to be undertaken hereunder, reasonable, good faith efforts to accomplish such objective, activity or decision as such Party would normally use to accomplish a similar objective, activity or decision under similar circumstances. Such efforts shall be substantially equivalent to those efforts and resources commonly used by such Party for a product owned by it or to which it has rights, which product is at a similar stage in its development or product life and is of similar market and commercial potential as the Product. The term “Commercially Reasonable” shall have correlative meaning. 1.19 “Confidential Information” shall have the meaning set forth in Section 11.1. 1.20 “Continuous Improvement Program” shall have the meaning set forth in Section 5.5. 1.21 “Control” means, with respect to any Confidential Information, Intellectual Property or regulatory documentation, possession by a Party of the ability (whether by ownership, license or otherwise) to grant access, rights, title, possession, a license or a sublicense, as applicable, to such intellectual property right without violating the terms of any Third Party agreement, court order, or other arrangement or legal obligation. 1.22 “Corden” shall have the meaning set forth in the preamble. 1.23 “Corden Indemnitee” has the meaning set forth in Section 10.2. 1.24 “CSP” means any Third Party cloud storage providers or other Third Party hosted service providers. 1.25 “Customer” or “Madrigal” has the meaning set forth in the Preamble. 1.26 “Customer Indemnitee” has the meaning set forth in Section 10.3. 1.27 “Customer Material” means the API provided by or on behalf of Customer to Corden. 1.28 “Customer Material Specifications” means the Specifications for Customer Materials. 1.29 “Customer Release” means the release of cGMP Product by Customer for the use in humans. Such Customer Release shall be conducted by Customer through review of all appropriate documentation which signifies that the Product has been produced using approved processes, in

5 compliance with appropriate regulations, including but not limited to, cGMP, and meets the established specifications including the Manufacturer’s Release Specifications. 1.30 “Deficiency Cure Batch(es)” has the meaning set forth in Section 4.5(a). 1.31 “Delivery” or “Deliver” or “Delivered” means Corden’s notification to Customer for pick up of Product in accordance with Section 3.3 (a). 1.32 Intentionally Omitted. 1.33 Intentionally Omitted. 1.34 Intentionally Omitted. 1.35 “Effective Date” has the meaning set forth in the Preamble. 1.36 “Embargoed Country” or “Embargoed Party” has the meaning set forth in Section 14.2(a). 1.37 “Equipment” means all equipment and machinery used to (or otherwise necessary for), directly or indirectly, Manufacture of Product. 1.38 “Export Control Regulations” has the meaning set forth in Section 2.6(a) and Section 14.2(a). 1.39 “Facility” means Corden’s facility located at Otto Hahn Strasse 68723 Plankstadt Germany where the Product is being Manufactured (if and only if such facility is approved by applicable Regulatory Authority(es) for the Manufacture of Product). 1.40 “FDA” means the U.S. Food and Drug Administration and any successor agency(ies) or authority having substantially the same function. 1.41 “FDCA” means the United States Federal Food, Drug and Cosmetic Act of 1938 (21 U.S.C. §301 et seq.) and applicable regulations promulgated thereunder, as amended from time to time. 1.42 “Force Majeure” has the meaning set forth in Section 15. 1.43 Intentionally Omitted. 1.44 “Governmental Authority” means any supra-national, federal, national, regional, state, provincial or local entity responsible for granting approvals for the performance of services under this Agreement or for issuing or enforcing any Applicable Law, or for exercising authority with respect to the Manufacture of the Product or the conduct of Manufacturing services at any Facility or in granting Health Registration(s) and/or exercising authority with respect to the Product, including without limitation the FDA and the European Medicines Agency. 1.45 “Health Registration” means, with respect to a medicinal product containing Product, all registrations with and approvals from the relevant Governmental Authority necessary to market and sell such medicinal product in a given country or group of countries, including the technical, medical and scientific licenses, registrations, authorizations and/or approvals of such medicinal product (including any marketing authorizations, pricing approvals, reimbursement approvals, and labeling approvals, as applicable). With respect to the United States, the term Health Registrations shall include an NDA, for such medicinal product, as amended from time to time. When referring to a “Health Registration” for a Product in this Agreement, such term shall also refer to the Health Registration for the final finished form of the Product, as applicable. For clarity, Health Registrations shall exclude any permits specific to the Facility. 1.46 “Importer” or “Importer of Record” has the meaning set forth in Section 2.6(a). 1.47 “Indemnifying Party” has the meaning set forth in Section 10.6.

6 1.48 “Indemnitee” has the meaning set forth in Section 10.6. 1.49 “Initial Term” shall have the meaning set forth in Section 13.1. 1.50 “Intellectual Property” means any intellectual or industrial property, including, without limitation, patents, trademarks, trade names, service marks, domain names, copyrights, trade secrets, inventions, know-how, data, and any other intellectual property, in each case, whether statutory or non- statutory, registered or unregistered and including applications for registration, and all rights or forms of protection having equivalent or similar effect anywhere in the world that are owned or controlled by a Party as of the Effective Date or after the Effective Date was developed outside the scope of the activities set forth in a Product Addendum during the term of this Agreement. 1.51 “Inventions” means any discoveries, observations, innovations, inventions, improvements, modifications, works of authorship, developments, concepts, designs, data, know-how or trade secrets, whether or not protectable under patent, copyright, trade secrecy or other laws, that are conceived, developed, or reduced to practice or tangible medium of expression by either Party or jointly by the Parties in connection with the performance of the activities set forth in a Product Addendum during the Term of this Agreement. 1.52 “Latent Defect” means, (i) with respect to any Customer Materials supplied by Customer under this Agreement, defects in such Customer Materials that existed at the time of delivery but that could not be reasonably detected at the time such Customer Materials was tested in accordance with the pertinent Specifications; and (ii) with respect to Product supplied by Corden under this Agreement, defects in such Product that existed at the time of Delivery but that could not be reasonably detected at the time such Product was tested in accordance with the Testing Specifications or after Customer Release. 1.53 “Launch” has the meaning set forth in Section 3.1(b). 1.54 “Losses” shall have the meaning set forth in Section 10.2. 1.55 “Madrigal Europe” shall have the meaning set forth in Section 3.2(g). 1.56 “Manufacture” or “Manufacturing” means the manufacture, processing, packaging, labeling, quality control and testing of the Product performed up to and including to the Manufacturer’s Release by Corden in accordance with the terms of this Agreement and subject to any applicable Product Addendum executed pursuant hereto, including, as applicable, receipt (including testing) and storage of Customer Materials and Raw Materials, production, visual inspection, packaging, labeling, handling, warehousing, quality control testing (including in-process, release and stability testing), release, as applicable, of Product, and also including such activities as may be specified in the Master Batch Record for the Product. 1.57 “Manufacturing Process” means, with respect to the Product, the manufacturing process that is in effect on the effective date of a Product Addendum, which process shall not be changed by Corden or Customer during the Term except in accordance with the terms of this Agreement. 1.58 “Master Batch Record” means a controlled document of Corden, approved by authorized representatives of both Parties, that documents the Manufacturing Process. It includes all relevant process parameters to be met and equipment and Raw Materials to be used. 1.59 “Manufacturer’s Release” means Corden has: (i) Manufactured and/or packaged and/or labeled a Product according to the Master Batch Record; (ii) fulfilled its testing/analytical obligations as further set forth herein; (iii) had its quality department review and approve all Manufacturing and testing services performed by Corden; (iv) completed and made available to the Customer all documentation related to such Product pursuant to the Quality Agreement or any additional documentation reasonably requested from the Customer’s quality department; and (v) provided Customer with notice that such Product is available to be picked up by Customer’s carrier. 1.60 “Manufacturer’s Release Specifications” means the defined list of analytical test methods performed by Corden or on behalf of Corden and the respective acceptance criteria thereto for the Product as set forth in the applicable Product Addendum for Corden’s Manufacturer’s Release.

7 1.61 “Manufacturing Standards” has the meaning set forth in Section 4.2. 1.62 “Minimum Remaining Shelf-Life” means, with respect to Product, the minimum percentage of the maximum shelf-life for such Product that is required to be remaining at the time of - Delivery by Corden of such Product hereunder, which shall in all cases be eighty percent (80%) of the maximum shelf-life for the Product (based upon the shelf-life of the Product configuration with the shortest shelf-life) set forth in the Health Registration for the Product. 1.63 “Minimum Time” has the meaning set forth in Section 3.2(c). 1.64 “MSA” has the meaning set forth in the Recitals. 1.65 “Non-Binding Forecast” has the meaning set forth in Section 3.1(b). 1.66 “Non-Conforming Product” has the meaning set forth in Section 4.33(a). 1.67 “Note” has the meaning set forth in Section 2.6. 1.68 “Partner(s)” means licensees, sublicensees or distributors that Customer engages for the purpose of marketing, promoting, distributing, offering for sale and selling the Product. 1.69 “Party(ies)” has the meaning set forth in the Preamble. 1.70 “Person” means an individual, sole proprietorship, partnership, limited partnership, limited liability partnership, corporation, limited liability company, business trust, joint stock company, trust, incorporated association, joint venture or similar entity or organization, including a government or political subdivision, department or agency of a government. 1.71 “Product” means bulk drug product as more specifically described in the Product Addendum attached as Appendix 2. 1.72 “Product Addendum” means a written individual agreement between the Parties for Corden to provide Services with respect to the Product and Manufacture such Product for Customer’s commercial use according to the terms and conditions set forth in this Agreement. For the avoidance of doubt, a Project Addendum shall not be used to address activities undertaken pursuant to the MSA, 1.73 “Product Invention” means any Invention that relates exclusively to (a) the Product or any derivative thereof, (b) the use of the Product and/or (c) any process, method or procedure specifically related to the Processing of the Products including analytical methods directed solely to the Product (such as, but not limited to, assays that may only be used for testing the Products, and not to test any other product) and that can not be used without incorporating: (A) Customer Background Intellectual Property, (B) Customer Confidential Information, or (C) Customer Material. 1.74 Intentionally Omitted. 1.75 “Project Invention” means any Invention that relates exclusively to, incorporates, or contains claims requiring: (A) Corden Intellectual Property, or (B) Corden Confidential Information, or that (C) relates generally to manufacturing technologies, methods, processes or techniques or improvements to existing manufacturing technologies, methods, processes or techniques that are broadly applicable to developing or manufacturing biologic or pharmaceutical products generally or to experimental methods and that is not a Product Invention. 1.76 “Purchase Order” has the meaning set forth in Section 3.2(a). 1.77 “Quality Agreement” refers to a separate agreement, which shall be integrated by reference into this Agreement, that sets out the quality assurance standards for Corden’s Manufacture of Product and performance of Services. 1.78 “Quantity Statement” has the meaning set forth in Section 2.6(b).

8 1.79 “Raw Materials” means all raw materials, chemicals, components, excipients, packaging and labeling components, and other consumable items, which are useful or necessary for the Manufacture of the Product, as may be further specified in a bill of materials in the Product Addendum. 1.80 “Records” means Corden’s (or its Affiliate’s or subcontractor’s, as applicable) records related to performance of this Agreement, which shall include Manufacturing documents, executed Batch Records, test results, reports, and any other cGMP relevant documentation related to he performance of this Agreement. 1.81 “Regulatory Filing(s)” means all applications, filings, dossiers and the like submitted to a Governmental Authority for the purpose of obtaining a Health Registration from such Governmental Authority. 1.82 “Replenishment Period” has the meaning set forth in Section 4.6(c). 1.83 “Rolling Forecast” has the meaning set forth in Section 3.1(b). 1.84 “Rules” has the meaning set forth in Section 16.14(a). 1.85 “Services” means Manufacturing of Product using API and other Customer Material, and certain ancillary services in addition to and in support of the Manufacturing of Product, including, for example, sourcing of certain Raw Materials, analytical analysis, stability services, and quality assurance services provided by Corden. 1.86 “Specifications” means numerical limits, ranges or other acceptance criteria to which the Raw Materials, Product or in-process samples of Manufacture of the Product, must conform to in order for the Product to be acceptable for its intended use under this Agreement and the Quality Agreement, as set forth in the Manufacture’s Release Specifications, the API Specifications, the Customer Materials Specifications and the Testing Specifications, as applicable. 1.87 “Safety Stock” or “Materials Safety Stock” has the meaning set forth in Section 4.6(a). 1.88 “Supply Deficiency” has the meaning set forth in Section 4.4(b). 1.89 “Surge Capacity” has the meaning set forth in Section 3.2(d). 1.90 “Testing Specification” means the specific master document that lists the testing parameters, references to analytical procedures and their Specifications. Types of Testing Specifications include but are not limited to the Raw Materials, in-process controls, intermediates and Product. 1.91 “Term” shall have the meaning set forth in Section 13.1. 1.92 “Territory” means the following countries and/or jurisdictions, where Customer intends to market the Product: the United States of America, the member countries of the European Union, the United Kingdom, Australia, China and Japan, and any other country that the Parties agree in writing to add to this definition of Territory in an amendment to this Agreement, it being understood that any Regulatory Filings for such other countries shall be discussed in good faith and subject to mutual agreement. For clarity, the Territory shall not include countries that are targeted by the comprehensive sanctions, restrictions or embargoes administered by the United Nations, European Union, United Kingdom, or the United States. 1.93 “Third Party(ies)” means any Person other than (a) Customer, (b) Corden or (c) an Affiliate of Customer or Corden

9 2. SCOPE OF THE AGREEMENT 2.1 Manufacture of Product. Subject to the terms and conditions set forth herein, Corden agrees to Manufacture Product for Customer and to supply Customer with Product as may be agreed upon by the Parties in a Product Addendum in accordance with Customer’s orders as defined and forecasted pursuant to Section 3. 2.2 Additional Services. Corden shall provide such other services in connection with Manufacturing of Product as may be agreed upon by the Parties in a Product Addendum, including but not limited to stability studies (including registration stability), analytical validation completion based on identified gaps, placebo manufacture (if required), services arising from the preparation of a response to a question from a Regulatory Authority (to the extent necessary), additional validation protocols / testing (if required for future commercial batches), redevelopment (if required), scale up Batch size, process improvements. 2.3 Intentionally Omitted. 2.4 First Source. Pursuant to the terms of this Agreement, Customer hereby appoints Corden as its non-exclusive contract manufacturer for the Product in the Territory. Customer shall order and Corden shall supply, [***] during the Initial Term. In no event shall the forgoing preclude Customer from taking whatever steps necessary to qualify alternative suppliers for Product prior to the expiration of the initial Term nor from obtaining from alternative suppliers at any time during the Term any amount of Product for countries in the Territory that Corden is unable or unwilling to Manufacture and supply Product in accordance with the terms and conditions of this Agreement and any amount of Product other than [***] that is committed to Corden in accordance with this Section 2.4. 2.5 General Terms and Conditions. All Product Addenda are governed by the terms and conditions of this Agreement. The general terms and conditions of each Party and each of the Party’s Affiliates shall not apply under this Agreement and are hereby expressly excluded. Any reference in a Purchase Order to the general terms and conditions of any Party, Affiliates or Third Parties shall not become legally effective between the involved legal entities. In the event of any conflict between the terms of this Agreement and any Purchase Order Section 16.16 of this Agreement shall govern. 2.6 Sourcing of Customer Material and other Raw Materials. (a) Delivery, Import/Export. Unless otherwise agreed between the Parties in a Product Addendum, Customer will at its sole cost and expense deliver the Customer Material to the Facility DDP (Incoterms 2020). Customer’s obligation will include obtaining the release of the Customer Material from the applicable customs agency and Government Authority. Unless otherwise agreed in writing, Customer or Customer’s designated broker will be the “Importer” or “Importer of Record” (or equivalent, as understood under Applicable Laws) for Customer Materials imported to the Facility, and Customer is responsible for compliance with Applicable Laws (and the cost of compliance) relating to that role. For Customer Materials which may be subject to import or export to or from the EU or the U.S., Customer agrees to be responsible that its agents, brokers, vendors and carriers will comply with applicable requirements of the EU and the U.S. as well as international laws (hereinafter “Export Control Regulations”) imposing restrictions on trade including export, re-export, import, transfer, disclosure, supply or comparable transaction, regardless of the way of provision to other countries or parties. (b) Time of Delivery. Unless not otherwise agreed between the Parties in a Product Addendum, Customer or its designee shall deliver to the Facility at least forty (40) calendar days prior to the planned production start of Product ordered by Customer hereunder, such quantities of Customer Material as are reasonably forecasted by Corden pursuant to Section 3.1(f) for Corden to Manufacture the amount of Product

10 ordered by Customer, at no cost to Corden. Each delivery of Customer Material shall be accompanied by an appropriate certificate of analysis or equivalent documentation and a statement setting forth the amount of Customer Material being delivered (the “Quantity Statement”). Customer Materials shall be delivered in accordance with the provisions and standards set forth in the Quality Agreement. (c) Testing. Upon receipt of Customer Material, Corden shall conduct or perform (i) visual inspections in accordance with the Quality Agreement, including identification testing, (ii) testing in accordance with the applicable Testing Specifications, and (iii) a quantity check to confirm that the quantities delivered are as set forth in the applicable Quantity Statement. For Customer Material being API: each shipment of API will be accompanied by an API certificate(s) of analysis from the API manufacturer, confirming that the API conforms to the API Specifications. Customer will be responsible for disclosing to Corden all information available to it regarding health risks associated with the API. Corden will test the API, using test methods (as agreed upon by the Parties and set forth in the Quality Agreement), to verify that the API conforms to the API Specifications. Corden shall only release API for use in Manufacturing Product hereunder if Corden verifies that the API conforms to the API Specifications. Customer shall be responsible for qualification of the API vendor and for providing a Certificate of Compliance confirming that the API is compliant with the provisions outlined in the “Note for Guidance on minimizing the risk of transmitting spongiform encephalopathy agents via human and veterinary medicinal products” (EMEA/410/01, Rev.2 or update) (the “Note”) as such may be modified from time to time. (d) Inspection Period. Within twenty (20) calendar days after Corden’s receipt of Customer Material (including API) Corden shall inspect the Customer Material (“Inspection Period”). Upon expiration of the Inspection Period, Corden shall provide Customer with written confirmation that such shipment conforms with and to the applicable Customer Material Specifications to the extent required under the Quality Agreement, and the Quantity Statement. Corden shall maintain control samples of the Customer Material and records with respect to such testing and/or inspection, in accordance with Corden’s internal record retention policies and cGMP, and shall make such records available to Customer during normal business hours, upon Customer’s prior written request. (e) Non-Conforming Customer Material. In the event that Corden reasonably determines that any Customer Material does not conform with or to the Customer Material Specifications, Corden shall notify Customer thereof as soon as practicable but not later than two (2) Business Days after the conclusion of the Inspection Period, and Corden shall not use such Customer Material for the manufacture of the Product until the conformity of such shipment is established or negated as set forth in this Section 2.6(e). Notwithstanding the foregoing, Latent Defects are not subject to the above timelines and will be communicated in writing as soon as possible to Customer, but in no event more than thirty (30) days after the date Corden first becomes aware of the defect. In the event the Parties cannot agree as to whether any Customer Material conforms with or to the Customer Material Specifications within fourteen (14) calendar days after such notification is provided by Corden to Customer, the Parties shall designate an independent testing laboratory to determine same, the findings of which shall be binding on the Parties, absent manifest error, gross negligence, or fraud on the part of the testing laboratory. Any agreed delivery dates for any Purchase Order affected by non- conforming Customer Material which is subject to the aforementioned testing and investigation shall be extended commensurate with the time consumed for such testing and investigation and any subsequent remedy action. The costs and expenses of such laboratory testing shall be borne solely by the Party whose position is determined to have been in error or, if the testing laboratory cannot place the fault noticed and complained about on one Party, then the Parties shall share equally the costs and expenses of the testing laboratory. In the event that the Customer Material is ultimately agreed by the Parties or found by the testing laboratory not to conform with or to the

11 Customer Material Specifications, Customer shall, at its option, re-work or replace (or have re-worked or replaced) such non-conforming Customer Material at Customer’s cost and expense. At Customer’s discretion and at Customer’s sole cost and expense, Corden shall deliver to Customer (or its designee), or destroy, any rejected Customer Material. Notwithstanding the foregoing, if the Delivery dates as agreed in the Purchase Orders are delayed due to (i) Customer Material not meeting the Customer Material Specifications or (ii) Customer Material not delivered within the time as agreed in Section 2.6(b) or (iii) Corden requires additional time for testing of Customer Material that is returned to Corden after being reworked, then Corden’s time period for Manufacture and Delivery of the applicable portion of such Purchase Orders shall be appropriately extended. If Corden is unable to use such manufacturing capacity for another purpose despite its good faith efforts, then Customer agrees to compensate Corden for such the direct costs of such lost or idle capacity based on the Product Price of the applicable Product which was scheduled to be Manufactured during the time of idle capacity using the Customer Material which is not available according to the cases of subsection (i) to (iii) above. (f) Ownership. Customer shall at all times exclusively own and retain all right, title and interest in, to and under (i) any Customer Material delivered to Corden pursuant to this Agreement including any API contained in any Manufacturing in progress and in the Product Manufactured hereunder; and (ii) all documentation, information and data as well as all biological, chemical or other materials Controlled by Customer and furnished to Corden in connection with this Agreement and/or the Services (including without limitation, all samples and Customer Confidential Information). (g) Use of Customer Material. Any Customer Material under the control or in the possession of Corden shall be used by Corden solely and exclusively to manufacture Product to be supplied to Customer pursuant to this Agreement. Corden shall supply Customer with all of the Product that Corden manufactures using the Customer Material during the Term. (h) Inventory. Within five (5) Business Days after the end of each Calendar Quarter during the Term, Corden will provide Customer with an inventory report, which report will minimally include a description of Customer Material and Product then in its possession or control, including the lot number(s), quantity and inventory status of same. (i) Other Raw Materials. All Raw Materials (other than the Customer Material) required for the manufacture of the Products shall be procured and/or purchased by Corden for its own account, at the sole cost and expense of Corden. Prior to use of any such other Raw Materials, Corden shall ensure that such Raw Materials conforms with or to the applicable Raw Materials specifications. Corden shall maintain adequate inventory of such qualified Raw Materials to meet its obligations at Corden’s sole expense. If Customer wishes Corden to use a specific vendor(s) to purchase Raw Materials and this vendor is not an approved supplier currently used by Corden, it will be Customer’s responsibility to audit and approve such vendor, at Customer’s cost and expense. 3. FORECASTS, ORDERS 3.1 Forecasts. (a) Intentionally Omitted. (b) During the Term, but no later than six (6) months prior to the commercial launch of the Product in the Territory using Corden as registered commercial manufacturer (“Launch“), on a Calendar Quarter-by-Calendar Quarter basis, Customer shall provide
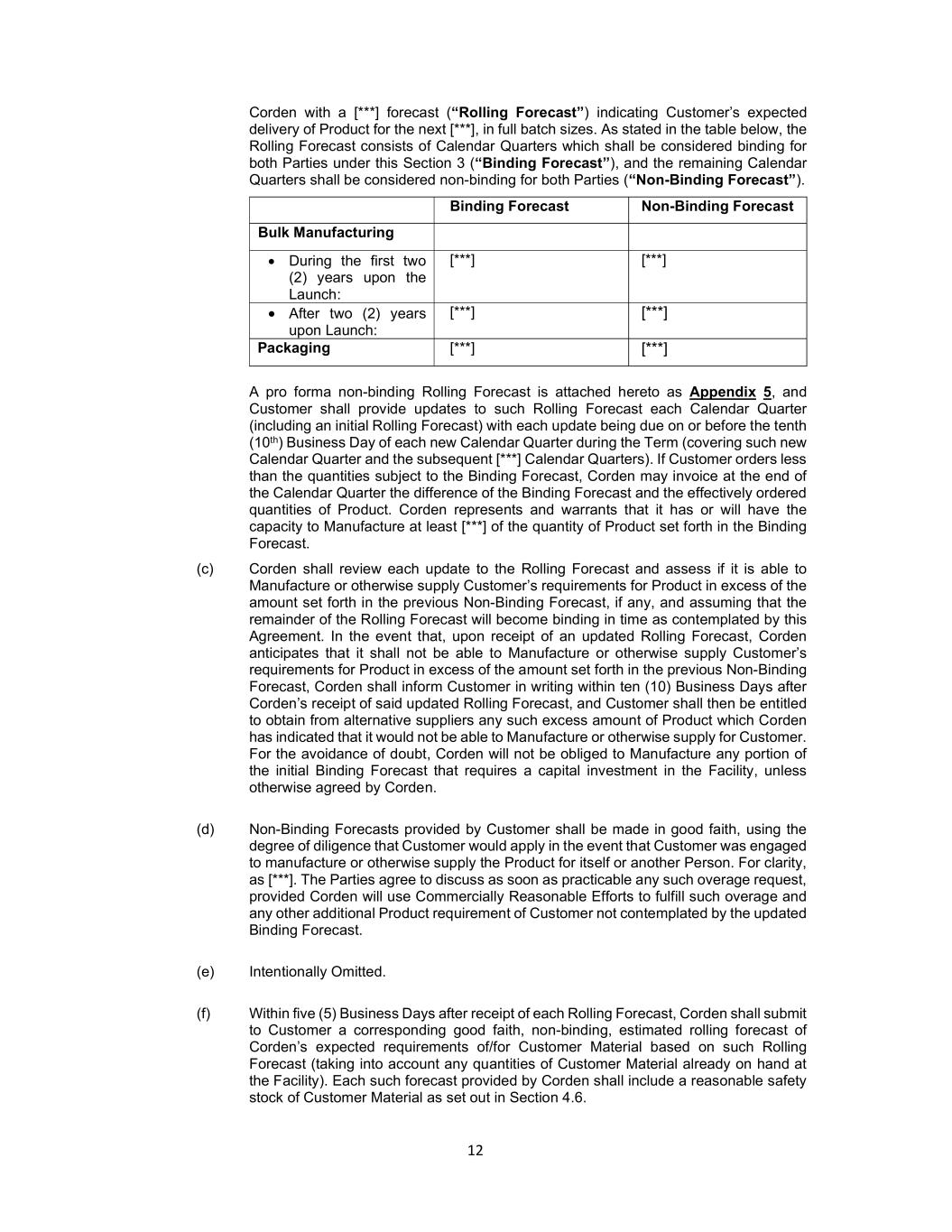
12 Corden with a [***] forecast (“Rolling Forecast”) indicating Customer’s expected delivery of Product for the next [***], in full batch sizes. As stated in the table below, the Rolling Forecast consists of Calendar Quarters which shall be considered binding for both Parties under this Section 3 (“Binding Forecast”), and the remaining Calendar Quarters shall be considered non-binding for both Parties (“Non-Binding Forecast”). Binding Forecast Non-Binding Forecast Bulk Manufacturing During the first two (2) years upon the Launch: [***] [***] After two (2) years upon Launch: [***] [***] Packaging [***] [***] A pro forma non-binding Rolling Forecast is attached hereto as Appendix 5, and Customer shall provide updates to such Rolling Forecast each Calendar Quarter (including an initial Rolling Forecast) with each update being due on or before the tenth (10th) Business Day of each new Calendar Quarter during the Term (covering such new Calendar Quarter and the subsequent [***] Calendar Quarters). If Customer orders less than the quantities subject to the Binding Forecast, Corden may invoice at the end of the Calendar Quarter the difference of the Binding Forecast and the effectively ordered quantities of Product. Corden represents and warrants that it has or will have the capacity to Manufacture at least [***] of the quantity of Product set forth in the Binding Forecast. (c) Corden shall review each update to the Rolling Forecast and assess if it is able to Manufacture or otherwise supply Customer’s requirements for Product in excess of the amount set forth in the previous Non-Binding Forecast, if any, and assuming that the remainder of the Rolling Forecast will become binding in time as contemplated by this Agreement. In the event that, upon receipt of an updated Rolling Forecast, Corden anticipates that it shall not be able to Manufacture or otherwise supply Customer’s requirements for Product in excess of the amount set forth in the previous Non-Binding Forecast, Corden shall inform Customer in writing within ten (10) Business Days after Corden’s receipt of said updated Rolling Forecast, and Customer shall then be entitled to obtain from alternative suppliers any such excess amount of Product which Corden has indicated that it would not be able to Manufacture or otherwise supply for Customer. For the avoidance of doubt, Corden will not be obliged to Manufacture any portion of the initial Binding Forecast that requires a capital investment in the Facility, unless otherwise agreed by Corden. (d) Non-Binding Forecasts provided by Customer shall be made in good faith, using the degree of diligence that Customer would apply in the event that Customer was engaged to manufacture or otherwise supply the Product for itself or another Person. For clarity, as [***]. The Parties agree to discuss as soon as practicable any such overage request, provided Corden will use Commercially Reasonable Efforts to fulfill such overage and any other additional Product requirement of Customer not contemplated by the updated Binding Forecast. (e) Intentionally Omitted. (f) Within five (5) Business Days after receipt of each Rolling Forecast, Corden shall submit to Customer a corresponding good faith, non-binding, estimated rolling forecast of Corden’s expected requirements of/for Customer Material based on such Rolling Forecast (taking into account any quantities of Customer Material already on hand at the Facility). Each such forecast provided by Corden shall include a reasonable safety stock of Customer Material as set out in Section 4.6.

13 3.2 Purchase Orders. (a) Customer shall place purchase orders covering the quantities of Product contained in each Binding Forecast (each, a “Purchase Order”) and Corden shall accept such Purchase Orders in full (to the extent consistent with the Binding Forecast) and deliver the quantities of Product covered by such Purchase Orders with such delivery dates as are not sooner than [***] following the date on which Corden receives the Purchase Order. Customer or its designee shall issue Purchase Orders that include timelines for delivery and quantity of Product to be supplied by Corden and such other details as may be agreed to by the Parties in writing in accordance with such specified lead times. Together with the first Rolling Forecast delivered hereunder, Customer will issue Purchase Orders covering the first [***] (i.e., the initial Binding Forecast). Thereafter, Customer will issue Purchase Orders with each subsequent Rolling Forecast that covers the new Calendar Quarter added to the latest Binding Forecast. Each Purchase Order will be confirmatory of, and supplemental to, the latest Binding Forecast rather than creating a new legal obligation. For the sake of clarity, [***]. (b) Purchase Orders for Product will contain the Batch size, which shall be [***], as specified in a Purchase Order. (c) Each Purchase Order placed by Customer must be accepted by Corden by way of a written Purchase Order confirmation. Corden will acknowledge acceptance of all Purchase Orders within five (5) Business Days after Corden’s receipt of such Purchase Order. Corden will accept all Purchase Orders to the extent that the Purchase Order requires Delivery not fewer than hundred twenty (120) days following the date on which Corden receives the Purchase Order (the “Minimum Time”) and the quantity of Product to be Delivered does not exceed the Binding Forecast plus the applicable Surge Capacity, subject only to the exception set forth in Section 3.1(c) above. Corden shall Deliver the Product covered by any such Purchase Orders on or before the scheduled delivery date as specified in the applicable Purchase Order confirmation. (d) Corden shall maintain the ability to increase the quantity of Product Manufactured such that it would be able to, within the delivery period under any applicable Purchase Order, Manufacture one hundred and fifteen percent (115%) of the quantity of Product that is the subject of the first two (2) Calendar Quarters of the most recent Rolling Forecast delivered pursuant to Section 3.1 (“Surge Capacity”). (e) Any change in or to any Purchase Order shall require the prior written agreement of Corden and Customer. Any accepted Purchase Orders pursuant to this Section 3.2 shall be firm and binding on the Parties and may not be cancelled, either totally or partially, unless agreed to by the Parties in writing. (f) Corden shall not be liable for any delay in Manufacturing/supplying Product to the extent such delay is due to circumstances caused by or within the direct control of Customer, or is due to Force Majeure. Corden shall promptly inform Customer of any circumstance which may cause delays in manufacturing/supplying Product and both Parties will use commercially reasonable efforts to mitigate the effects of any such delay. (g) For the avoidance of doubt, it is understood that Customer’s to be established Affiliate located within the European Economic Area (“Madrigal Europe”) may place Purchase Orders under this Agreement, and that if Madrigal Europe places such Purchase Orders, then Customer shall cause Madrigal Europe to comply with the provisions of this Agreement in connection with such performance. Any breach by Madrigal Europe of any of Customer’s obligations under this Agreement shall be deemed a breach by Customer, and Corden may proceed directly against Customer without any obligation to first proceed against Madrigal Europe.

14 3.3 Manufacturer’s Release; Delivery; Invoicing; Payment; Customer Release. (a) All Product shipments shall be accompanied by the appropriate documentation and Manufacturer’s Release Specifications as more fully described in the Quality Agreement. All Product containers shall be appropriately labelled with the name and presentation of the Product, traceable batch number, date of manufacturing, SAP Code, quantity of/in each Product containers and storage conditions. The packing slip for the Product shall also contain: item number, quantity of Product, shipment date and ship- to address as specified by Customer. [***]. Corden shall not Deliver Product unless and until such Product has been quality released by Corden in accordance with Section 3.3(b). (b) If, based upon the review performed by Corden, the Product conforms to the Manufacturer’s Release Specifications, Testing Specifications and was otherwise Manufactured according to Applicable Law, then a Certificate of Compliance will be completed by Corden. Corden shall confirm in writing to Customer that such Product has been subject to Manufacturer’s Release and cleared for Delivery, and Corden shall issue the corresponding invoice for the price of such quantity of Product specified in Appendix 3 (the “Product Price”). Corden will deliver to Customer at the time it notifies Customer of Manufacturer’s Release, the applicable Manufacturer’s Release Specifications and Testing Specifications results, the completed Manufacturing and bulk packaging Batch Record(s), Certificate of Analysis, Certificate of Compliance, along with a packing list including Product description, lot number, lot expiration date, and quantity available for shipment Corden will also deliver to Customer all raw data and other records in the possession or under the Control of Corden relating to the Manufacture of such Product, as well as summaries of all applicable analytical results in machine-readable format. Upon receipt of such Manufacturer’s Release documentation for each Purchase Order set forth in this Section 3.3(b), Customer will review as outlined in the Quality Agreement and raise concerns (if any) of noncompliance with the Specifications or cGMP within thirty (30) calendar days. During this period, Customer shall have the right to request reasonable additional clarifying information from Corden, which Corden shall provide promptly. Failure by Corden to provide such clarifying information available to Corden shall delay Customer’s review period for an equal number of days. When clearing any Product for Delivery, Corden shall do so in accordance with the instructions for shipping and packaging specified in the applicable Purchase Order accepted by the Parties or as otherwise agreed to by the Parties in the Quality Agreement. (c) Customer shall authorize shipment of the Product within fourteen (14) days following provision of the Manufacturer’s Release documentation to Customer in accordance with Section 3.3(b). Corden shall provide Customer with reasonable assistance to obtain and maintain any necessary export approvals, licenses and customs clearance applications, forms and other correspondence in connection with the Delivery of Product. In any case, Corden will invoice the Product upon Manufacturer’s Release and provision of the Manufacturer’s Release documentation to Customer in accordance with Section 3.3(b). In case Customer wants Corden to store Product that is the subject of Manufacturer’s Release according to Section 4.1(b), Corden will not have the Product anymore in its books but in Customer inventory. (d) Customer will review the provided documentation and evaluate the Product upon receipt, and may test it against the Manufacturer’s Release Specifications, and will notify Corden in writing of its acceptance or rejection of such Batch as promptly as possible after its receipt. If Customer intends to reject a Batch on the grounds that it is Non-Conforming Product, Customer shall notify such rejection to Corden in writing, such notice to be given within thirty (30) calendar days after receipt of the Product by Customer or its designee, to be accompanied by a sample of the Product analyzed by Customer together with all relevant documentation and a report indicating the methods used by Customer to evaluate same. If Customer does not report the failure to conform to the Manufacturer’s Release Specifications that should have been reasonably

15 detected by Customer when reviewing the Product supplied by Corden and testing it against the Manufacturer’s Release Specifications or Purchase Order terms for packaging within such period of thirty (30) calendar days after receipt of the Product, such Product shall be deemed to have been accepted by Customer as conforming to the applicable Manufacturer’s Release Specifications. Notwithstanding the forgoing, for a maximum period up to the shelf-life of the relevant Product, Customer or its designee reserves the right to reject Product as Non-Conforming Product if the reason such Product (a) does not conform with the applicable Manufacturing Standards, (b) is adulterated within the meaning of Section 501(a)(2)(B) of the FDCA, or (c) was not otherwise Manufactured in accordance with Applicable Law, because such Product contained a Latent Defect that was not reasonably detectable, unless such Latent Defect was directly attributable solely to a defect or nonconformity of the Customer Material used in the Manufacture of such Product, or was due to the negligent transportation of such Product from the Facility to Customer or its designee and further provided that Customer shall notify any Latent Defect to Corden promptly after it becomes aware of the defect but in any event within five (5) calendar days thereafter. If Customer rejects Product pursuant to this Section 3.3(d), the Parties shall resolve such matter using the procedure specified in Section 4.3. 4. MANUFACTURE OF PRODUCT 4.1 Storage and Handling. (a) Corden shall store and handle all Customer Materials and the Product in accordance with the relevant Specifications, the Quality Agreement, Applicable Law, and under other appropriate conditions, including without limitation, appropriate temperature, humidity, light and cleanliness conditions in order to avoid any material adverse effect on the identity, strength, quality and/or purity of the Products. In addition to the foregoing, Corden shall store and handle all Customer Materials and the Product so as to prevent the commingling of same with Corden’s own inventories and supplies, or those held by Corden for Third Parties. (b) Handling and storage of Customer Materials and Product are free of charge, in the case of Product, for twelve (12) weeks from the date the Product is the subject of Manufacturer’s Release for Delivery to Customer or its designee in accordance with Section 3.3(b). In the event that storage in excess of twelve (12) weeks is required, Corden shall offer Customer storage for such Product on consignment either at the Facility, or at a Third Party facility, at mutually acceptable financial terms. If Corden is unable to store any Product due to capacity constraints, Corden may use an Affiliate or qualified Third Party to store outside the Facility any Product under this Agreement. Customer acknowledges that such a consignment storage may lead to a Delivery of Product which is subject to local VAT, and that Customer shall assume all risk of loss or damage to Product while stored at a Third Party facility and that Customer shall be responsible for having appropriate insurance coverage in place for this risk. (c) In case any Product is returned to Corden after having been shipped by Corden, due to any cause which is not connected to the actions undertaken by Corden, the reshipment costs along with any warehousing fees will be for the account of Customer. 4.2 Manufacturing Standards. Corden shall perform all qualification and validation of the Facility, Equipment, and the Manufacturing Process in accordance with cGMP, Applicable Law, the Quality Agreement and Corden’s standard operating procedures, as well as other mutually agreed procedures. Once the Manufacturing Process has been validated by Corden, Corden shall Manufacture the Product in accordance with cGMP, Applicable Law, the Quality Agreement in order to meet the Manufacturer’s Release Specifications at the time of the Manufacturer’s Release (collectively, the “Manufacturing Standards”). 4.3 Non-Conforming Product(s).

16 (a) A Product that does not conform with or to the applicable Manufacturing Standards (as may be in effect from time to time during the Term) or that is adulterated within the meaning of Section 501(a)(2)(B) of the FDCA or similar provisions of any applicable laws in the country where the Manufacture takes place for any reason or that was not otherwise Manufactured in accordance with Applicable Law, shall be deemed to be a non-conforming Product (“Non-Conforming Product”). (b) In the event of any disagreement between the Parties regarding whether a Product is a Non-Conforming Product, the quality assurance representatives of the Parties will attempt to resolve any such disagreement in good faith. If the disagreement is not resolved in a reasonable time (which will not exceed thirty (30) calendar days after a notice of dispute is provided by one Party to the other Party), a representative sample of the Product and/or relevant documentation will be submitted for tests and final determination as to whether or not such Product is Non-Conforming Product. The Parties shall designate an independent testing laboratory or consultant or both to determine whether the relevant Product is a Non-Conforming Product, the findings of which testing laboratory shall be binding on the Parties, absent manifest error, gross negligence or fraud on the part of the testing laboratory. The independent testing laboratory shall be instructed to complete its analysis within ten (10) Business Days after its appointment using the test methods contained in the Manufacturer’s Release Specifications. The costs and expenses of such laboratory testing shall be borne solely by the Party whose position is determined to have been in error or, if the testing laboratory cannot place the fault noticed and complained about on one Party, then the Parties shall share equally the costs and expenses of the testing laboratory. If, after the later of (i) sixty (60) days from the date of receipt by Corden of Customer’s notice pursuant Section 3.3(d) or Section 4.3(a) and (ii) completion by the independent testing laboratory of its analysis of the relevant Product, the Parties have not agreed as to the payment of any outstanding fees related to such Product, the Parties shall commence arbitration pursuant to Section 16.14. (c) Notwithstanding any further right of Customer, as stipulated in this Agreement, in the event that any Product is ultimately agreed or found to be Non-Conforming Product and provided that such failure has been notified in accordance with Section 3.3.(d) and further provided that such failure is directly attributable to Corden and not due to the acts or omissions of Customer or any Third Party after Delivery of such Product but regardless of when it was discovered, then (save in respect of any claims by Third Parties, which shall be subject to the limitation set forth in Section 10.4 of this Agreement), Corden's liability shall be limited to, at Customer’s election, (i) Manufacturing new Product (in the same quantity as that which was deemed to be Non- Conforming Product), provided that Customer shall provide Customer Material needed for the new Product free of charge, or (iii) if possible according to Applicable Law, reworking or reprocessing the Non-Conforming Product, at Corden’s sole cost and expense, so that such Non-Conforming Product conforms to the applicable Manufacturer’s Release Specifications, (iii) only if (i) and (ii) are not possible, refunding in full the price paid by Customer for such Non-Conforming Product. 4.4 Inability to Supply. (a) Corden shall immediately notify Customer: (i) upon becoming aware of an event of Force Majeure or any other event that would render Corden unable to: (i) transfer the quantities that Corden is required to supply pursuant to any confirmed Purchase Order(s), or (ii) otherwise meet any of its supply obligations to Customer under this Agreement; and/or (iii) if Corden reasonably believes that it will not be able to meet any portion of the latest Binding Forecast provided by Customer following Corden’s receipt thereof. (b) In the event that Corden fails to: (i) transfer to Customer the quantities specified in the relevant confirmed Purchase Order; or (ii) otherwise meet any of its supply obligations to Customer hereunder, then in either such event the difference between the number of Batches transferred under a confirmed Purchase Order that meets the requirements

17 under this Agreement and the number specified in such Purchase Order shall constitute a “Supply Deficiency” for purposes of this Agreement, provided, however, that such Supply Deficiency represents more than ten percent (10%) of the amount specified in the confirmed Purchase Order. 4.5 Procedure to Cure Supply Deficiencies. If there is a Supply Deficiency, then, if requested by Customer and at Customer’s election, Corden shall promptly take the selected following steps to remedy the Supply Deficiency, in the following order of preference whenever practicable (i.e., with highest preference given to the remedy in paragraph (a) and the lowest preference given to the remedy in paragraph (d)): (a) use Commercially Reasonable Efforts to increase the length of a manufacturing campaign at the Facility in order to manufacture and transfer to Customer additional Batches that meet the relevant requirements under this Agreement to fully remedy such Supply Deficiency (“Deficiency Cure Batches,” and each such Batch, a “Deficiency Cure Batch”); (b) use Commercially Reasonable Efforts to utilize any capacity at the Facility which is not then contractually committed to the performance of manufacturing services for Third Party customers during the applicable Contract Year to manufacture and transfer to Customer such Deficiency Cure Batches that meet the relevant requirements under this Agreement; (c) coordinate and cooperate with Customer to re-schedule Manufacture and supply Batches of Product ordered hereunder that meet the relevant requirements under this Agreement in order to maximize Corden’s ability to Manufacture and supply to Customer such Deficiency Cure Batches that meet the relevant requirements under this Agreement while minimizing the disruption of manufacture at the Facility then in force and any contractual commitments to Third Party customers; and (d) use Commercially Reasonable Efforts to remedy the Supply Deficiency in subsequent periods, if any, by utilizing and dedicating excess capacity not contractually committed to Third Party customers to manufacture and transfer Deficiency Cure Batches that meet the relevant requirements under this Agreement and to reserve such capacity for Customer’s requirements until all of the issues surrounding the Supply Deficiency have been remedied to Customer’s complete satisfaction. If Corden is unable to remedy a Supply Deficiency using the procedures specified above after an aggregate period of six (6) months (or longer as agreed in writing by mutual agreement of the Parties), commencing with the date upon which such Supply Deficiency began (as specified in Section 4.4(b)), Customer shall have the right to: (i) cancel any outstanding Purchase Order until the Supply Deficiency has been rectified, and Customer shall have no obligation to Corden for any Purchase Order to the extent the Product has not been supplied prior the date of delivery of such cancelation notice; and/or (ii) have the Product manufactured by a Third Party supplier rather than by Corden. For the avoidance of doubt, it is understood and agreed that a Supply Deficiency does not relieve Customer of its obligations under Section 2.4 with respect to the purchase of Product from Corden in the event and to the extent that Corden is able to supply Customer with Product during such Supply Deficiency, provided that during such Supply Deficiency, Customer’s obligation under Section 2.4 is limited to the quantity of Product that Corden can Manufacture and Deliver in accordance with the terms of this Agreement. During a Supply Deficiency, the Parties shall confer regarding the amount of Product that Corden is able to supply Customer pursuant to a proposed Purchase Order prior to the date that Customer places any new Purchase Order, and Customer shall adjust the quantity of Product in the Purchase Order(s) it places during such Supply Deficiency accordingly. For the avoidance of doubt, Customer shall not be in breach of its obligations under Section 2.4 with respect to any such Purchase Order modified pursuant to this Section 4.5 adjustment procedure. Customer may continue to use a Third Party supplier to supply Product that Corden cannot supply until Corden notifies Customer that it is again able to supply Customer’s forecasted quantity of the

18 Product and substantiates such claim to Customer’s reasonable satisfaction. Upon such a showing, Customer shall commence purchasing from Corden at least the applicable percentage portion of Customer’s requirements for Product for which Customer substantiates its ability to supply, provided that: (1) Customer shall not be required to cancel any then outstanding purchase orders with the Third Party supplier to the extent such orders have been accepted by such Third Party supplier and are binding obligations of Customer and (2) Corden shall have paid all cancellation costs incurred by Customer in switching its purchases from such Third Party supplier to Corden. Customer shall use Commercially Reasonable Efforts to avoid significant cancellation fees in any contracts it enters with any Third Party supplier. Customer shall not order Product from a Third-Party supplier for delivery more than nine (9) months following the date of such order and/or in amounts in excess of Customer’s forecast provided to Corden for the period during which the Supply Deficiency is occurring (as well as an amount equal to the applicable Surge Capacity). 4.6 Materials Safety Stock. (a) Corden shall within nine (9) months of the Effective Date have a safety stock of each of the Materials set forth on Appendix 6 (“Safety Stock Materials”) in a quantity that is sufficient to Manufacture the quantity of Product equal to the first two (2) Calendar Quarters of Customer’s then most recent Binding Forecast, and thereafter throughout the Term Corden shall maintain a safety stock of such Safety Stock Materials in a quantity that is equal to the quantity of Safety Stock Materials required to Manufacture the quantity of Product equal to the first two (2) Calendar Quarters of Customer’s then most recent forecast (the “Safety Stock”). Corden will use Safety Stock to supply Product ordered by Customer, and will maintain the appropriate level of Safety Stock by promptly replenishing that quantity of Safety Stock Materials used in such supply in accordance with Section 4.6(c). If Customer has failed, for a period of two (2) consecutive Calendar Quarters to purchase a quantity of Product equal to or greater than the two (2) previous Calendar Quarters, then Corden may reduce the Safety Stock to a level reflecting the reduction in actual purchases by Customer for such two (2) Calendar Quarter period. Unless mutually agreed to otherwise, Corden will manage Safety Stock on a “First In, First Out” basis to fulfil Customer purchase orders for Product on a routine basis. Customer shall have the right to adjust Safety Stock levels from time to time. Notwithstanding the foregoing, if Safety Stock Materials become obsolete other than due to Corden’s negligence or willful misconduct, Customer shall reimburse Corden for the cost of such obsolete Safety Stock Materials, including but not limited to any related shipping or destruction costs. (b) If any of the Safety Stock Materials show a longer lead time than expected, Corden is allowed to order such Safety Stock Material outside the Binding Forecast at Customer’s cost and upon Corden’s request Customer shall provide the respective purchase order for such materials. (c) Corden shall replenish its Safety Stock of each of the Safety Stock Materials within thirty (30) days of use pursuant to Section 4.6(a) (the “Replenishment Period”). Corden shall within twenty (20) days of the expiration of the Replenishment Period notify Customer in writing of its inability to replenish the Safety Stock. 4.7 Quarterly Business Review Meetings. The Parties shall conduct a Quarterly Business Review meeting on a Calendar Quarter basis during the Term to exchange information to enable the Parties to review and discuss topics of interest, including but not limited to the following issues: (i) general business update; (ii) Quality performance and any open corrective actions; (iii) Delivery performance and any open corrective actions; (iv) Forecast and capacity review, including the acquisition of Materials; and (v) other existing and planned projects.

19 5. PAYMENTS, TAXES 5.1 Payments. Customer shall pay all invoices for the Product to be paid under this Agreement that are properly invoiced in accordance with Section 3.3 and Appendix 3 or Appendix 2 (if the Parties enter into a Product Addendum that includes fees for other activities). (a) Payments shall be made by Customer within [***] after the date the applicable invoice is received by Customer. All invoices and payments required to be paid hereunder shall be in Euro and all such payments shall be completed electronically and wired in immediately available funds to an account designated by Corden. Undisputed late payments shall bear interest at [***]. Failure by Customer to pay undisputed invoices within ninety (90) days after the date the applicable invoice is received by Customer shall be considered a breach of this Agreement by Customer. (b) Customer shall have the right to withhold payment of any portion of an invoice that is subject to justified warranty claims or other claims hereunder. If the claim is later found to be unjustified, Corden shall have the right to reinvoice Customer with the applicable interest included as per Section 5.1(a). (c) Commencing with Calendar Year 2028, Corden reserves the right to adjust the fees no more than once per calendar year to cover increases in raw materials, consumables, energy costs, and services supplied to Corden by Third Parties required for the performance of this Agreement based on the actual increase in the annual average monthly German index of producer prices of industrial products and non-durable goods during the preceding 12-month period, as published by the German Statistic Office. Any such change in the prices shall become effective on the anniversary of the Effective Date. For avoidance of doubt, the pricing provided in Appendix 3 shall not be subject to adjustment pursuant to this Section 5.1(c) during Calendar Years 2025, 2026, and 2027. 5.2 Taxes. Except for value added tax, any and all federal, provincial or municipal taxes, levies, charges or fees imposed upon or with respect to or measured by the production, sale or delivery by Corden to Customer of Product in accordance with Customer’s instructions, shall be for the account of Corden. If applicable, each invoice will show any applicable VAT required to be charged separately. In the event that Corden will be charged by local tax authority for importation VAT for Customer Materials supplied to Corden, Corden acknowledges that Corden will invoice Customer for such paid importation VAT as a pass- through cost for the importation VAT paid by Corden. Corden shall cooperate with Customer in its efforts to recover the VAT incurred and shall provide the required import documentation to support Customer in such activities. 5.3 Withholding Tax. If withholding of any taxes imposed by local laws or regulations of the country where one of the Parties has its registered office is required on account of any payments paid/to be paid under this Agreement, such taxes shall be borne by Customer. All payments by Customer shall be made free and clear of and without any deduction for or on account of withholding taxes. In case Customer is required to make such a payment subject to the deduction of local withholding taxes, the sum payable by Customer shall be increased to the extent necessary to ensure that Corden receives a sum net of any withholding or deduction of taxes equal to the sum which it would have received in case no such deduction or withholding of taxes has been made or required to be made. The Parties shall use their Commercially Reasonable Efforts to do all such acts and things and to sign all such documents as will enable them to take advantage of any applicable double taxation agreement or treaty. 5.4 Certification of Purchase Obligation. Within thirty (30) days of the end of each Calendar Year during the Term, Customer shall deliver to Corden a written report of the total kilograms of Product ordered by Customer from all sources for sale in the United Kingdom and the European Union Member Countries, certified by Madrigal’s Chief Financial Officer. If Madrigal fails to comply with its annual purchase obligations under Section 2.4 in any

20 Calendar Year, Madrigal shall promptly pay Corden the Product Price for the quantity of Product that Madrigal did not purchase, but should have purchased from Corden in such Calendar Year pursuant to the allocation provisions set forth in Section 2.4. 5.5 Continuous Improvement Program. Corden shall use Commercially Reasonable Efforts to identify and implement (and accept input from Customer regarding) continuous cost, quality and Customer service improvement programs by (i) seeking productivity improvements, (ii) minimizing waste and improving Product yields, (iii) purchasing quality Materials at lower cost, (iv) improving Manufacturing processes within the validated parameters for Product, (v) streamlining organizational processes, and (vi) reducing cycle times and lead times, with such cost improvement objectives obtained, determined or measured, among other things, by reference to industry or competitive developments (collectively, the “Continuous Improvement Program”). Progress against objectives shall be measured yearly. The Parties shall as part of the Quarterly Business Review Meetings pursuant to Section 4.7 discuss and use Commercially Reasonable Efforts to agree on (a) objectives for the Continuous Improvement Program, which will include specific objectives for each calendar year; and (b) the means of measuring and implementing the results of the Continuous Improvement Program. Corden shall use Commercially Reasonable Efforts to achieve the agreed objectives and targets identified for the relevant period. The up-front costs for any such agreed upon development improvements shall be borne by Customer. The net benefits of cost reductions and improved efficiencies shall be shared equally by the Parties, after first settling any disproportionate up-front cost allocation, and allocated to Customer via reductions to the Product Price under this Agreement. In such case, the Parties shall use Commercially Reasonable Efforts to discuss and agree on the amount of such reductions to the Product Price. 6. WARRANTIES 6.1 Mutual Representations and Warranties. Each Party hereby represents and warrants to the other Party that: (a) it is a corporation duly organized and validly existing under the laws of the state or other jurisdiction of incorporation or formation; and (b) the execution, delivery and performance of this Agreement by such Party has been duly authorized by all requisite corporate action; and (c) it has full corporate authority to enter into this Agreement and the Agreement is binding upon it in accordance with its terms; and (d) it is not under any obligation, contractual or otherwise, to any Person that conflicts with or is inconsistent in any material respect with the terms of this Agreement, or that would impede the diligent and complete fulfilment of its obligations hereunder. 6.2 Customer Warranties. Customer hereby represents and warrants that: (a) to its reasonable knowledge, as of the Effective Date, the Product as well as the Customer Background IP and all Customer Confidential Information (including for the avoidance of doubt the Manufacturing Process) and their use by Corden or its Affiliates in accordance with this Agreement do not infringe the Intellectual Property Rights of any Third Party; and (b) as of the Effective Date, Customer has the right to grant Corden the licenses stipulated under this Agreement, and will notify Corden if at any time during the term of this Agreement it no longer has the right to grant such licenses; and (c) the Customer Materials provided by Customer (i) have been manufactured and tested in accordance with cGMP or ISO guidelines (as applicable), (ii) meet the required specifications and (iii) are neither adulterated nor contaminated; (d) for avoidance of doubt, all Customer liability or indemnification obligations that might result from the representations and warranties under this Section 6.2 are always subject to the limitations set forth in Section 10 of this Agreement.

21 6.3 Corden Warranties. Corden hereby represents and warrants that: (a) the Product, if and to the extent it is required to be Manufactured under cGMP conditions, (i) will conform to the agreed Manufacturer’s Release Specifications; (ii) has been manufactured, stored, tested, labelled, and packaged in accordance with Applicable Laws including cGMP and any environmental or biohazard laws in the country where the Manufacture takes place; (iii) will not be adulterated within the meaning of Section 501 (a)(2)(B) of the FDCA or similar provisions of any Applicable Laws in the country where the Manufacture takes place; (iv) will not be an article that may not, under the provisions of the FDCA or similar provisions of any Applicable Laws in the country where the Manufacture takes place, be introduced into stream of commerce by Corden; (v) will have at least the Minimum Remaining Shelf-Life, as evidenced by expiry dating, remaining, except where Customer is the cause for any delay in Manufacture or Delivery of Product that results in less than the Minimum Remaining Shelf-Life remaining at the time of Delivery; and (vi) will be transferred to Customer free and clear of any security interests, liens or encumbrances; (b) to its reasonable knowledge at the Effective Date, there are no Third Party claims against Corden or its Affiliates asserting that the Corden Background IP and Corden Confidential Information to be used in the performance of the Services and/or Manufacturing infringe the Intellectual Property Rights of any Third Party, and Corden will promptly notify Customer in writing should it become aware of any claims asserting such infringement in the performance of any Product; and (c) neither Corden nor any of its representatives has been debarred, nor is subject to a pending debarment, and that neither Corden nor any of its representatives will use in any capacity in connection with the Services and/or Manufacturing under this Agreement any person, who has been debarred pursuant to section 306 of the FDCA. 21 U.S.C. § 335a, or who is the subject of a conviction described in such Section. Corden agrees to notify Customer in writing immediately if it comes to its knowledge that Corden or any person who is performing Services is debarred or is the subject of a conviction described in section 306 of the FDCA or if any action, suit, claim, investigation, or proceeding is pending, or to Corden’s knowledge, is threatened, relating to the debarment or conviction of Corden or any person performing Services under this Agreement. (d) for avoidance of doubt, all Corden liability or indemnification obligations that might result from the representations and warranties under this Section 6.3 are always subject to the limitations set forth in Section 10 of this Agreement. 6.4 Disclaimer of Warranties. EXCEPT AS EXPRESSLY SET FORTH IN THIS SECTION 6, NEITHER PARTY MAKES ANY WARRANTIES, REPRESENTATIONS OR GUARANTEES OF ANY KIND WHATSOEVER, EITHER EXPRESS OR IMPLIED, WRITTEN OR ORAL, EITHER IN FACT OR BY OPERATION OF LAW, BY STATUTE OR OTHERWISE, AND EACH PARTY SPECIFICALLY DISCLAIMS ANY OTHER WARRANTIES, INCLUDING WITHOUT LIMITATION, ANY EXPRESS OR IMPLIED WARRANTIES OF MERCHANTABILITY, QUALITY, NONINFRINGEMENT OR AS TO THE VALIDITY OF ANY PATENTS, OR FITNESS FOR A PARTICULAR PURPOSE OR USE. 7. CHANGES TO PRODUCTS / CHANGE CONTROL 7.1 Changes to the Products (including the Raw Materials and suppliers thereof), the Manufacturing Process or the Facility may only be made in accordance with the Quality Agreement. For the purpose of clarity, changes to the Facility under this Section 7.1 and in the Quality Agreement only refers to changes directly relating to the Manufacturing of Product and/or impacting the Product. Notwithstanding anything herein to the contrary or in the Quality Agreement, except as otherwise agreed to by Customer in writing or as may be required to comply with the Applicable Law (including cGMPs), Corden shall not amend, change, or supplement any of the following without Customer’s prior written consent: (1) the Product Specifications; (2) the Materials; (3) the specifications for Materials that have regulatory impact (e.g., specification is listed in the Regulatory Filing(s)) or the potential for quality impact on the Product; (4) the source of Materials that have regulatory impact (e.g., supplier is listed in the Regulatory Filing(s)) or the potential for quality impact on the Product;

22 (5) the equipment and machinery, other than in-kind replacements, used in the Manufacture of Product that have a direct impact on the quality of the Product; (6) the test methods used in connection with the Manufacture and release testing of Product that have regulatory impact (e.g., method is listed in the Regulatory Filing(s)) or the potential for quality impact on the Product; (7) the process for Manufacturing Product or Materials; or (8) the cleaning process or procedures used at any time on the equipment and machinery used in the manufacture of Product that have a direct impact on the quality of the Product. 7.2 Corden shall solely bear all of its actual and related costs resulting from: (a) changes to the Facility (including but not limited to changes related to Facility safety reasons) requested by Corden (on its own or on behalf of any approved subcontractor); (b) non-Product specific changes (i) required under Applicable Laws applicable in the country where the Facility is located; (ii) requested or required by the Government Authorities relating to the Facility and to the manufacture of pharmaceutical products; and/or (iii) related to the establishment and/or maintenance of cGMP; and (c) changes in the materials or suppliers of the Raw Materials requested by Corden and to the extent within Corden’s control. 7.3 Customer shall solely bear all actual and related costs resulting from: (a) changes requested by Customer (other than changes requested or required as set out in Section 7.2(b)); (b) changes requested or required by the Regulatory Authorities relating to the Product, Manufacturing of Product and marketing of the pharmaceutical products containing the Product (except as set forth in Section (7.2(b)); and (c) changes to the Product, Raw Materials and/or suppliers of Raw Materials for Product or the Manufacturing requested by Customer. 7.4 In the event that Applicable Laws change such that Corden’s expense for the Manufacture of the Product increases by more than ten (10) percent, Corden shall be entitled to add the additional, reasonable and documented expenses to the Price for Product apportioned to Customer on a pro-rata share of the increase based on Corden’s apportioned utilization of the Facility. 7.5 In the event and solely to the extent that a change requested by Customer (or by Corden in accordance with Section 7.2(c)) may reasonably be expected to have an impact on the Manufacturing Process and/or the Manufacturer’s Release Specifications, the warranty obligation of Corden pursuant to Section 6.3(a)(i) shall be waived, but solely to the extent relating to such change and solely until such time that real-time stability data is available to reasonably support that such change does not negatively impact the Manufacturing Process and/or the Manufacturer’s Release Specifications. Under no circumstance shall such change relieve Corden of its general obligations to comply with the terms of this Agreement and the Quality Agreement. 7.6 To the extent permitted by Applicable Law, Corden shall continue to supply Customer with Product approved under any Customer’s existing regulatory filings (including NDAs), as applicable, for the subject Product until such time as the Product Manufactured following such change is permitted under the amended regulatory filings therefor. 7.7 Corden shall Manufacture all Product supplied hereunder at the Facility. Manufacturing of Product may not be relocated from the Facility without Customer’s prior written consent (in its sole discretion).

23 8. HEALTH REGISTRATION APPROVAL SUPPORT; REGULATORY MATTERS 8.1 Filing and Maintenance of the Health Registrations. As between the Parties, Customer shall have the sole right to prepare and file for the Health Registrations, including the NDA and the CMC, with the applicable Governmental Authorities, and, for clarity, Corden shall have no right to do so except in the case of an inspection of the Facility and/or other request for information made by an applicable Governmental Authority. Customer may at its own discretion include a designation of Corden and the Facility as a Manufacturer and Manufacturing site of Product in the applicable Health Registrations. 8.2 CMC Information. Customer shall provide Corden with CMC information applicable to Corden to the extent necessary for Corden to Manufacture the Product in accordance with this Agreement, and Corden shall comply with all such CMC information in performing its activities hereunder. In the event Customer subsequently modifies any relevant Health Registration (or the CMC) which will affect the Manufacture of the Product hereunder, it shall promptly notify Corden and provide it with necessary information about such Health Registration (or CMC) supplements or amendments to the extent necessary for Corden to Manufacture the Product in accordance with this Agreement, and the provisions of Section 8.3 shall apply. 8.3 Regulatory Support for Maintaining Filings. Corden shall perform the activities (including tests) in connection with the receipt and maintenance of the CMC as reasonably requested by Customer from time to time, which activities shall be performed by Corden in compliance with all Applicable Laws and shall be covered in a Change Order. In all cases, Corden shall be prepared for any and all inspections, including pre-approval inspections, by Governmental Authorities. Without limitation of the foregoing, Corden shall provide Customer with information and assistance as Customer may reasonably request for purposes of applying for and maintaining all relevant Health Registrations for the Product including providing Customer with all reports, authorizations, certificates, methodologies, specifications and other documentation in the possession or under the control of Corden (or any of its Affiliates) relating to the pharmaceutical/technical development and/or Manufacture of the Product or any component thereof. Corden hereby grants Customer a right of reference to any regulatory approvals and/or Corden’s (and/or any of its Affiliates) site master file for use in connection with the Product. 8.4 Corden Approvals. Except as otherwise specifically set forth herein, Corden shall be responsible for obtaining and maintaining (and throughout the Term, Corden shall maintain in full force and effect) all permits and approvals from all Governmental Authorities or as otherwise may be required under Applicable Laws, in each case, to operate the Facility and shall ensure that its subcontractors operate in compliance with any applicable regulations. 8.5 Audit Reports and Other Information. Corden acknowledges that Governmental Authorities may, in conducting an inspection of Customer, request copies of reports of Customer audits of its suppliers. For clarity, in response to such a request, Customer shall have the right to provide to the Governmental Authority any report of any compliance audit conducted hereunder (including as may be conducted in accordance with the Quality Agreement) and any other information in connection with the activities hereunder, provided that such disclosure is made in compliance with the provisions set forth in Section 11 (Confidentiality). 8.6 Communication with Governmental Authorities. As between the Parties, Customer shall have the sole right to communicate with the appropriate Governmental Authorities relating to the Product, and Corden shall have no right to do so unless required to do so by Applicable Laws. Corden shall provide Customer or its Affiliate, in a timely manner, all information in Corden’s (or its Affiliate’s) possession or control concerning the Product which is reasonably requested by Customer (or its Affiliates) and which is reasonably necessary to meet Customer’s (and its Affiliate’s) regulatory obligations. Corden shall immediately notify Customer of any Governmental Authority request for samples of the Product, or Executed Batch Records or Master Batch Records or any other information

24 related to the Product and will not provide such material, records or information until such notification is made to Customer. 8.7 Records. Corden shall retain all Records related to: (a) the validation of Product (i.e., Manufacturing, packaging/labelling (if applicable), or Manufacturing Process until the Product is no longer marketed; (b) the Manufacture of Product for use in human clinical trials for a period of not less than five (5) years from the date of the completion of the applicable clinical trial to which said Records pertain (or such longer period as required by Applicable Law) and (c) Manufacture of Product that is subject to a Health Registration for five (5) years past the date such Product is delivered to Customer or one (1) year following the date of termination of this Agreement (or such longer period as required by Applicable Law. 9. QUALITY 9.1 Quality Agreement. Promptly after the Effective Date, the Parties shall enter into good faith discussions and use their respective good faith reasonable efforts to enter into a Quality Agreement within thirty (30) days after the Effective Date, but in any event prior to the Manufacture of Product for commercial purposes. Upon execution, such Quality Agreement shall be appended to this Agreement as Appendix 7. Such Quality Agreement shall be consistent with all Applicable Laws and regulations relating to the Manufacture of the Product and describe the respective quality assurance responsibilities and obligations of the Parties for the Manufacture of Product. 9.2 Quality Control. Corden shall maintain a quality assurance and quality control program in accordance with cGMP, Applicable Laws and the Quality Agreement. 9.3 Quality Audits and Regulatory Inspections. In accordance with the terms of the Quality Agreement, Corden shall (and shall reasonably cause its Affiliates and approved subcontractors to) permit (i) Customer or its designated representatives to audit the Facility and any facility where Products are being Manufactured, handled or stored, or Services are performed by approved subcontractors; and (ii) Government Authorities to inspect any such facilities. With the exception of Product-specific pre-approval audits and Product-specific regulatory inspections, for which the Parties shall agree a Service Fee upfront, all audits and inspections by Customer (but not more often than once every two Calendar Years except in the case of emergency or for-cause (and for clarity, cause may include a Supply Deficiency) in which case such once every two Calendar Years limit shall not apply) performed in accordance with the Quality Agreement shall be free of charge for Customer. Customer’s personnel shall comply with Corden’s procedures concerning cGMP, training, safety, hygiene, confidentiality and controlled access to facilities and documents. For practical reasons and in order to ensure a smooth Manufacturing Process, a maximum of two (2) Customer representatives or employees shall be permitted in the manufacturing area for no more than two (2) Business Days per quality audit. For the purpose of such audits, Customer or its authorized representative shall have access to the Records beginning on the Effective Date. In addition, Corden shall provide adequate and appropriate workspace for Customer or its authorized representatives to conduct such audits. Such audits shall be subject to prior notice by Customer as agreed by the Parties 9.4 Person in Plant. Customer shall have the right to have personnel at the Facility during regular business hours during a manufacturing campaign for the Product as set forth in the Quality Agreement. Such personnel may either be technical or quality personnel. Customer’s personnel shall comply with Corden’s procedures concerning cGMP, training, safety, hygiene, confidentiality and controlled access to facilities and documents as set forth under the Quality Agreement. 9.5 Retention Samples; Annual Product Evaluations. Corden is responsible for maintaining, retaining and storing retention samples of each Batch of Product Manufactured under this Agreement sufficient to perform quality analysis in accordance with the Product

25 Specifications and Applicable Laws. Such samples shall be retained for the longer of (i) the expiration period of the Product plus one (1) year or (ii) such longer period as may be required by Applicable laws. Corden will conduct annual product reviews for the Product as required under Applicable Laws to evaluate the quality standards of the Product to determine the need for changes in Product Specifications or Manufacturing or control procedures. Corden will provide Customer with a copy of such Product reviews to Customer in accordance with the Quality Agreement. 10. RECALL, INDEMNIFICATION, INSURANCE, SECURITY MEASURES 10.1 Investigation, Recall, Voluntary Withdrawal. In the event that any Governmental Authority in any country shall allege or prove that the Product does not comply with Applicable Law in such country where such Product is marketed, distributed and sold, the Party becoming aware of same shall notify the other Party in writing within 24 hours, and thereafter both Parties shall cooperate fully regarding the investigation and disposition of any such matter. If (a) such Product is adulterated within the meaning of Section 501(a)(2)(B) of the FDCA due to the acts or omissions of Corden and not due to the acts or omissions of Customer or any Third Party after delivery of such Product or (b) Customer is required or should deem it appropriate to voluntarily withdraw such Product, then to the extent that such recall or withdrawal is due to any negligence, fraud, recklessness or wrongful intentional acts or omissions by, or breach of any representation, warranty or covenant of or by Corden that could not have been reasonably detected by Customer during the Customer Release, then Corden shall bear the actual, documented and reasonable expenses of the Parties in carrying out the recall subject to the limitation of liability under Section 10.4, and Section 4.3(c) shall apply, to the extent the recall concerned Non-Conforming Product. 10.2 Indemnification by Corden. Subject to Sections 10.4 and Error! Reference source not f ound., Corden shall indemnify, defend and hold harmless Customer, its Affiliates, its sublicensees and distributors, and their respective directors, officers, employees, representatives and agents (each, a “Customer Indemnitee”), from and against any and all liabilities, damages, losses, costs or expenses whatsoever (including reasonable fees of attorneys and/or other professionals) (“Losses”) to which any Customer Indemnitee may become subject arising out of or resulting from claims, demands or actions (“Claims”) by Third Parties to the extent such Losses are based upon: (a) any breach of Corden’s obligations, representations and warranties set forth in Section 6 or the Quality Agreement, including the failure of Product to meet the Product warranties set forth in Section 6.3(a); (b) personal injury (including death) or property damage relating to or arising out of any Manufacture of Product by Corden or its Affiliates due to any negligence, fraud, recklessness or wrongful intentional acts or omissions by, or strict liability of, Corden or its Affiliates, and/or their respective directors, officers, employees or agents; (c) Corden’s or its Affiliates’ negligence, fraud, recklessness or wrongful intentional acts or omissions, or Corden’s or its Affiliates’ failure to comply with Applicable Law in the Manufacture of Product; or (d) the claimed infringement of any Third Party patent, trademark or other intellectual property right Corden was aware of and that is asserted due to any activities of Corden or any of its Affiliates relating to any of its Manufacturing processes or methods used in the Manufacture of the Product, except to the extent that such activities, processes or methods were specifically provided or required by Customer. 10.3 Indemnification by Customer. Subject to Sections 10.4 and 10.5, Customer shall indemnify, defend and hold harmless Corden and its Affiliates, and their respective directors, officers, employees and agents (each, a “Corden Indemnitee”), from and against any and all Losses

26 arising out of or resulting from Claims by Third Parties to the extent such Losses are based upon: (a) any claim that the use of the Customer Materials provided by Customer infringes the Intellectual Property of a Third Party; (b) any breach of Customer’s obligations, covenants, representations and warranties set forth in this Agreement or the Quality Agreement; (c) personal injury (including death) or property damage relating to or arising out of any use, distribution or sale of Product by Customer, its Affiliates or Partners (in each case after Delivery to Customer), unless due to any negligence, fraud, recklessness or wrongful intentional acts or omissions by, or strict liability of, Corden, its Affiliates or its Partners, and/or their respective directors, officers, employees or agents; or (d) Customer’s or its Affiliates’ negligence, fraud, recklessness or wrongful intentional acts or omissions in the manufacture or procurement of Customer Materials to the extent used by Corden in the Manufacture of Product. 10.4 Limitation on Indemnification and Liability. (a) Corden’s obligations under Section 10.2 and Customer’s obligations under Section 10.3 shall not apply to the extent that the Customer Indemnitee’s or the Corden Indemnitee’, as applicable, Losses are attributable to any act constituting negligence, fraud, recklessness, wrongful intentional act or omission on the part of the Customer Indemnitee or the Corden Indemnitee, as applicable or are subject to indemnification by the Customer under Section 10.3 or Corden under Section 10.2, as applicable. (b) Except for: (a) any unauthorized use of Customer’s Intellectual Property or Confidential Information; (b) Corden’s gross negligence, willful misconduct or fraud in connection with the Manufacture of a Product; or (c) Third Party Claims, including for personal injury or death for Product provided in breach of the warranties given in Section 6, in no event shall Corden’s maximum liability to Customer for: (i) the replacement costs of Non- Conforming Product under Section 4.3 including cost for replacement of Raw Material and Customer Material, or (ii) any other claim relating to any breach of Corden’s obligations, covenants, representations and warranties set forth in this Agreement or the Quality Agreement exceed one hundred fifty (150%) percent of the amount payable under the Purchase Order that gave rise to the Claim. 10.5 Consequential Damages. EXCEPT FOR EACH PARTY’S LIABILITY FOR FRAUD OR OTHER WILLFUL MISCONDUCT, AND EXCEPT AS OTHERWISE PROVIDED IN SECTIONS 10.2-10.3 WITH RESPECT TO THIRD PARTY CLAIMS, IN NO EVENT SHALL EITHER PARTY OR ITS AFFILIATES OR THEIR RESPECTIVE OFFICERS, DIRECTORS, EMPLOYEES OR AGENTS BE LIABLE TO THE OTHER PARTY OR ITS AFFILIATES OR ITS OR THEIR RESPECTIVE OFFICERS, DIRECTORS, EMPLOYEES OR AGENTS FOR ANY PUNITIVE, INDIRECT OR CONSEQUENTIAL DAMAGES OF ANY KIND, NATURE OR DESCRIPTION WHATSOEVER (INCLUDING, TO THE EXTENT CONSTITUTING CONSEQUENTIAL DAMAGES, ECONOMIC LOSSES OR LOST PROFITS) SUFFERED OR INCURRED BY SUCH PARTY ARISING UNDER OR IN CONNECTION WITH THIS AGREEMENT OR AS A RESULT OF ANY ACTIVITIES HEREUNDER, REGARDLESS OF WHETHER ARISING FROM BREACH OF CONTRACT, WARRANTY, TORT, STRICT LIABILITY OR OTHERWISE, EVEN IF SUCH PARTY IS ADVISED OF THE POSSIBILITY OF SUCH LOSS OR DAMAGE OR IF SUCH LOSS OR DAMAGE COULD HAVE BEEN REASONABLY FORESEEN. 10.6 Notice of Indemnification. Promptly after a Customer Indemnitee or a Corden Indemnitee (each, an “Indemnitee”) receives notice of a pending or threatened Claim, such Indemnitee

27 shall give written notice of the Claim to the Party from whom the Indemnitee is entitled to receive indemnification under Section 10.2 or Section 10.3 as applicable (the “Indemnifying Party”) as soon as reasonably practicable; provided that failure to give such notification shall not affect the indemnification provided under this Agreement, except to the extent the Indemnifying Party shall actually have been prejudiced by such failure in a material manner. Thereafter, the Indemnitee shall deliver to the Indemnifying Party, promptly after the Indemnitee’s receipt thereof, copies of all notices and documents (including court papers) received by the Indemnitee relating to the Claim. Upon receipt of notice from the Indemnitee, the Indemnifying Party shall have the duty to either compromise or defend, at its own expense and by counsel selected by the Indemnifying Party (reasonably satisfactory Io Indemnitee) such Claim. The Indemnifying Party will promptly (and in any event not more than twenty (20) days after receipt of the Indemnitee’s original notice) notify the Indemnitee in writing that it acknowledges its obligation to indemnify the Indemnitee with respect to the Claim pursuant to this Article 10 and of its intention either to compromise or defend such Claim. Once the Indemnifying Party gives such notice to the Indemnitee, the Indemnifying Party is not liable to the Indemnitee for the fees of other counsel or any other expenses subsequently incurred by the Indemnitee in connection with such defense, other than the Indemnitee’s reasonable out of pocket Third Party expenses related to its investigation and cooperation, except as otherwise provided in the next sentence. In any such proceeding the defense of which the Indemnifying Party shall have so assumed, the Indemnitee shall have the right to participate therein and retain its own counsel (without otherwise affecting the rights of the Parties under this Section 10.6) at its own expense unless (i) the Indemnitee and the Indemnifying Party shall have mutually agreed on the retention of counsel, (ii) the Indemnitee shall have reasonably concluded that there may be one or more legal defenses available to it which are different from or additional to those available to the Indemnifying Party, or (iii) the named Parties (including the impleaded Parties) include both the Indemnifying Party and the Indemnitee, and representation of both Parties by the same counsel would be inappropriate in the opinion of the Indemnifying Party’s counsel due to actual or potential differing interests between them; in any such case, one firm of attorneys separate from the Indemnifying Party’s counsel may be retained to represent Indemnitee at the Indemnifying Party’s expense. The Indemnitee shall reasonably cooperate with the Indemnifying Party and its legal representatives in the investigation and defense of any Claim. The Indemnifying Party shall keep the Indemnitee informed on a reasonable and timely basis as to the status of such Claim (to the extent the Indemnitee is not participating in the defense of such Claim) and conduct the defense of such Claim in a prudent manner. 10.7 Settlement. If an Indemnifying Party assumes the defense of a Claim, no compromise or settlement of such Claim may be effected by the Indemnifying Party without the Indemnitee’s written consent (such consent not to be unreasonably withheld, delayed or conditioned). Notwithstanding the foregoing, the Indemnitee’s consent shall not be required of a settlement where: (i) there is no finding or admission of any violation of law or any violation of the rights of any person and no effect on any other claims that may be made against the Indemnitee; (ii) the sole relief provided is monetary damages that are paid in full by the Indemnifying Party; (iii) the Indemnitee’s rights under this Agreement are not adversely affected; and (iv) there is a full release of the Indemnitee from such Claim. If the Indemnifying Party fails to assume defense of a Claim within a reasonable time, the Indemnitee may settle such Claim on such terms as it deems appropriate with the consent of the Indemnifying Party (such consent not to be unreasonably withheld, delayed or conditioned), and the Indemnifying Party shall be obligated to indemnify the Indemnitee for such settlement as provided in Section 10.2 or Section 10.3, as applicable. It is understood that only Customer and Corden may claim indemnification under this Agreement (on its own behalf or on behalf of its Indemnitees), and other Indemnitees may not directly claim indemnity under this Agreement. 10.8 Insurance. During the Term and for three (3) years thereafter, Corden shall, at its sole cost, obtain and maintain, with financially sound and reputable insurers, insurance coverage (including worker’s compensation) at or above the applicable statutory limits, comprehensive liability coverage with contractual liability, professional liability/errors and omissions coverage and such other coverage as may be reasonably necessary or useful in

28 connection with the conduct of its business to the extent insurable at reasonable terms covering its obligations and performance under this Agreement, including its storage and use of Customer Material, including, without limitation, endorsements for Product(s) liability, in such amounts and as at such terms and conditions as are customary for well-insured companies engaged in similar businesses and sufficient to support its obligations under this Agreement but in any event not less than the equivalent of [***] per occurrence and [***] per year in the aggregate per policy. 10.9 Security Measures. Corden shall take reasonable measures to protect the Facility, the Customer Material, the other Raw Materials, the Product and all work-in-progress from and against events, including but not limited to, theft, vandalism and terrorism. Corden agrees to notify Customer of any such events which threaten or negatively impact the Facility, the Customer Material, other Raw Materials, the Product, or any work-in-progress. 11. CONFIDENTIALITY 11.1 Generally. During the period from and after the Effective Date until the sixth (6th) anniversary of the expiration or termination of this Agreement, each Party shall keep confidential and shall not use for any purpose other than the performance of such Party’s obligations under this Agreement, and shall cause its Affiliates and such Party’s and its Affiliates’ respective directors, officers, employees and advisers to keep confidential and not to use for any purpose other than the performance of such Party’s obligations under this Agreement, all non-public information acquired from the other Party or its Affiliates, in connection with this Agreement and the transactions contemplated hereby, including, without limitation, all non-public information concerning the Process, Product Intellectual Property, the contents and existence of this Agreement and all Customer Material Specifications, Manufacturer’s Release Specifications and Testing Specifications and other quality standards hereunder, other than any information that: (i) is or hereafter becomes generally available to the public other than by reason of any default with respect to a confidentiality obligation; (ii) was already known to the receiving Party (which, in the case of Corden, shall mean known prior to the effectiveness of the Previous Manufacturing Agreement) as evidenced by prior written documents in the receiving Party’s possession; or (iii) is disclosed to the receiving Party by a Third Party who or which is not in default of any confidentiality obligation to the disclosing Party (such information to which none of the foregoing exceptions applies, “Confidential Information”). Each receiving Party shall transmit, and shall cause each of its Affiliates to transmit, Confidential Information only to those of its employees, agents or representatives who shall need same for the purpose of this Agreement and shall take all necessary measures to assure that such employees, agents or representatives do not reveal such Confidential Information to any Third Party without prior written authorization from the disclosing Party for as long as the receiving Party is obliged to hold such information in confidence hereunder, regardless of the respective terms of employment of such employees. Notwithstanding any other provision of this Agreement, Customer may disclose Confidential Information of Corden to a Partner; provided that such Partner is under confidentiality obligations at least as restrictive as those set forth herein. For clarity, all Customer Background Intellectual Property and Product Inventions, whether disclosed by Customer or its Affiliates to Corden or its Affiliates or developed under this Agreement, shall be the Confidential Information of Customer and not of Corden, with Customer considered the disclosing Party and Corden considered the receiving Party. All Corden Background Intellectual Property and Project Inventions, whether disclosed by Corden or its Affiliates to Customer or its Affiliates, owned by Corden prior to the Effective Date, or developed under this Agreement shall be the Confidential Information of Corden, and not of Customer, with Corden considered the disclosing Party and Customer considered the receiving Party. The terms of this Agreement shall be deemed the Confidential Information of both Parties. 11.2 Exceptions. The provisions of this Section 11 shall not apply to Confidential Information: (i) that is submitted by the receiving Party to Governmental Authorities to facilitate the issuance or maintenance of marketing approvals for the Product; provided that, to the extent permitted by Applicable Law, reasonable measures shall have been taken to ensure

29 confidential treatment of such Confidential Information; (ii) that is required to be disclosed in compliance with Applicable Laws or order by any court, supervisory, regulatory, judicial or Governmental Authority having competent jurisdiction (including without limitation SEC reporting and disclosures to a U.S. or foreign tax authority); provided that, to the extent permitted by Applicable Law, reasonable measures shall have been taken to ensure confidential treatment of such Confidential Information; or (iii) that is necessary to facilitate due diligence in connection with entering into a financing or similar arrangement with a bank or other credit institution. Each Third Party who receives Confidential Information pursuant to subclauses (i), (ii) and (iii) of this Section 11.2 shall be bound by the same confidentiality obligations set out in Section 11.1. 11.3 Remedies. Each Party shall be entitled, in addition to any other right or remedy it may have, at law, in equity or under this Agreement, to obtain temporary, preliminary and permanent injunctions, without the posting of any bond or other security, enjoining or restraining the other Party and its Affiliates from any violation or threatened violation of this Section 11. 12. PROPERTY RIGHTS 12.1 Background IP. Except as expressly set forth in this Agreement or as the Parties may otherwise agree in writing, each Party owns, and shall continue to own, its Background IP, without conferring or transferring any interests therein on the other Party. 12.2 Arising IP. (a) Subject to the obligations of confidentiality set forth in Section 11, each Party shall disclose to the other Party any and all Inventions made pursuant to the activities undertaken pursuant to this Agreement at least quarterly or as may otherwise be agreed to in writing by the Parties. (b) All Project Inventions shall be owned by Corden. To the extent that a Project Invention is patentable, Corden shall have the right but not the obligation to file, prosecute and maintain any patent or patent application claiming or covering any Project Invention. For the avoidance of doubt, as used in the definition, a Project Invention may include Inventions made solely by employees or consultants of Corden, employees or consultants of Madrigal or jointly by employees or consultants of Corden and employees or consultants of Madrigal. Corden hereby grants to Madrigal a perpetual, non- exclusive, fully paid-up worldwide license to use Project Inventions as necessary for Madrigal's use, distribution, import, export, sale, offer for sale, development or commercialization of the Product. (c) All Product Inventions shall be owned by Madrigal. Madrigal shall have the right but not the obligation to file, prosecute and maintain any patent or patent application claiming or covering any Product Invention. For the avoidance of doubt, as used in the definition, a Product Invention may include Inventions made solely by employees or consultants of Corden, employees or consultants of Madrigal or jointly by employees or consultants of Corden and employees or consultants of Madrigal. 12.3 Licenses. (a) Customer hereby grants to Corden a non-exclusive, royalty-free, limited, non- transferable, non-sublicensable license, during the Term, to use the Customer Background IP and Product Inventions, solely to the extent necessary to Manufacture and supply Product in accordance with this Agreement and to otherwise comply with its obligations hereunder. No other rights or licenses, either express or implied, to any other Intellectual Property Controlled by Customer are granted.

30 (b) Corden hereby grants to Customer a non-exclusive, worldwide, perpetual, fully paid-up and royalty-free, transferable, perpetual and irrevocable license, with the right to sublicense under the Corden Background IP and Project Inventions that Corden incorporates into the Manufacturing Process for the Product in the course of the activities performed pursuant to this Agreement for the sole purpose of manufacturing, using, selling, offering for sale, importing or otherwise exploiting the Product. No other rights or licenses, either express or implied, to any other Intellectual Property Controlled by Corden are granted. 12.4 Third Party Cloud Storage Services. Corden may utilize CSP in relation to the Services hereunder, provided that Corden shall notify Madrigal of the proposed CSP and establish a written relationship with such CSP with terms and conditions consistent with the terms and conditions of this Agreement as related to the Services hereunder. Corden shall, at all times, remain liable for the performance of the CSP. 12.5 Work at Third Party Facilities. Except as otherwise permitted under this Agreement, Corden will not transfer Madrigal supplied materials or use any Third-Party facilities or Intellectual Property in performing the Services without Madrigal's prior written consent. 12.6 Representatives. The obligations of this Article 12 shall apply to Corden's agents, employees, representatives, subcontractors, and affiliates involved in the Services to be performed hereunder. Corden agrees to cause such persons or entities to execute agreements ensuring compliance with this Agreement. 13. TERM, TERMINATION 13.1 Term, Extension. This Agreement shall commence on the Effective Date and shall continue for an initial term of five (5) years (“Initial Term”). If not terminated with eighteen (18) months’ written notice prior to the expiration of the Initial Term (or any subsequent renewal term), it shall be renewed for consecutive terms of two (2) year(s) each (the Initial Term, together with any such renewal term(s), the “Term”). 13.2 Termination for Cause. A Party shall have the right to terminate this Agreement upon the occurrence of any of the following events: (a) if the other Party commits a material breach of this Agreement, which (in the case of a breach capable of remedy) is not remedied within forty (40) Business Days after the receipt by the breaching Party of written notice from the non-breaching Party that identifies the breach and requires its remedy; provided that a breach of payment obligations shall be subject to a twenty (20) Business Day cure period; and provided further that if the breach is curable but not within forty (40) Business Days, then the breaching Party shall have such period of time as is necessary to cure the breach so long as the breaching Party has provided to the non-breaching Party a written plan that is reasonably calculated to effect a cure of such material breach, such plan is accepted by the non-breaching Party (such acceptance not to be unreasonably withheld, delayed or conditioned), and the breaching Party commits to and diligently carries out such plan as provided to the non-breaching Party; or (b) upon the filing or institution of bankruptcy, reorganization, liquidation, or receivership proceedings by or against the other Party (and if such proceedings are by or against Corden, Corden shall provide written notice of such proceedings to Customer within three (3) Business Days after such filing or institution, so that Customer may take possession of its property, including Product); provided, however, that in the event of any involuntary bankruptcy or receivership proceeding, such right to terminate shall only become effective if the proceeding is not dismissed within forty (40) Business Days after the filing thereof; or

31 (c) if the other Party ceases for any reason to carry on its business, or makes an assignment for the benefit of its creditors, or is the subject of any proposal for a voluntary arrangement. In addition, Corden shall promptly notify Customer in writing if Corden receives notice of the debarment or threatened debarment of any individual or entity utilized by Corden in connection with this Agreement, and Customer shall have the right to immediately terminate this Agreement upon written notice to Corden without further cost or liability, except for payments of accrued and unpaid obligations accrued before the date of termination if Corden is not able to replace such individual or entity with another individual or entity reasonably acceptable to Customer within reasonable time. 13.3 Termination by Customer. Customer shall have the right to terminate this Agreement in its entirety at any time if: (a) the European Medicines Agency or the FDA does not approve any required Health Registration, or other permit or license required to market and sell the Product, or any such approval, permit or license is deactivated, by the FDA or the European Medicines Agency; or (b) if Corden fails to satisfy validation of the Manufacturing Process as set out under the MSA; or (c) if any required license, permit or certificate of Corden related to the Manufacture of Product at the Facility is not approved or not issued, or is deactivated or withdrawn, by the FDA or the European Medicines Agency. 13.4 Termination for Force Majeure Event. A Party may terminate this Agreement upon written notice if Force Majeure has delayed performance by the other Party for more than ninety (90) consecutive days or an aggregate one hundred twenty (120) days in any 12-month period. 13.5 Termination by Mutual Agreement. The Parties may terminate this Agreement at any time upon mutual written agreement between the Parties. 13.6 Effects of Termination. All of the following effects of termination are in addition to the other rights and remedies that may be available to either of the Parties under this Agreement and shall not be construed to limit any such rights or remedies. (a) Expiration or termination of this Agreement for any reason shall not relieve Customer of payment obligations under this Agreement for Services performed prior to expiration or termination and payment associated with any close-out costs of any Purchase Orders and (except if such termination is due to an uncured material breach by Corden) all reasonably incurred future obligations to Third Parties and out-of-pocket expenses pre- incurred by Corden in connection with the performance of the Services, that cannot be reasonably avoided, cancelled or mitigated. For the avoidance of doubt, it is hereby clarified that in case of termination of this Agreement immediately after acceptance of such termination notice from Customer, (i) Corden will wind down any respective ongoing activities; and (ii) Corden shall neither incur any additional future obligations nor start any new activities, without receipt of Customer's specific written consent. (b) In the event that this Agreement is terminated by Customer in accordance with Section 13.2(a), Section 13.2(b), Section 13.2(c), Section 13.3(a), Section 13.3(b) or Section 13.3(c), Customer shall (in its discretion) either: (i) keep any or all outstanding Purchase Orders in place (on a Purchase Order-by-Purchase Order basis as determined by Customer), in which case Corden shall Manufacture and Deliver, in accordance with this Agreement, all quantities of Product ordered pursuant to such Purchase Orders (regardless of whether the Delivery Date for such Product is before or after such termination) and Customer shall pay the Product Price with respect to such Product which meet the representations, warranties and covenants set forth in this Agreement; or (ii) cancel any or all outstanding Purchase Orders (on a Purchase Order-by-Purchase Order basis as determined by Customer), and with respect to any such cancelled Purchase Orders, Customer shall have no further liability with respect thereto; provided that Customer shall only have the right to cancel Purchase Orders pursuant to this

32 Section 13.6(b) if this Agreement is terminated by Customer pursuant to Section 13.2(a) or Section 13.3. (c) In the event that this Agreement is terminated by Corden pursuant to Section 13.2(a) or by Customer pursuant to Section 13.3(a), Customer shall purchase: (i) the quantity of Safety Stock of Product existing as of the time of such termination (if any) (provided that all such Product meets the representations, warranties and covenants set forth in this Agreement), and in connection therewith, Corden shall Deliver all such quantities of Safety Stock in accordance with this Agreement, and Customer shall pay the applicable Product Price with respect to such Product and (ii) the quantity of Raw Materials in the possession of Corden or in transit to Corden, or subject to non- cancellable orders placed by Corden in reliance on Customer’s forecast, and in connection therewith Corden shall Deliver all such quantities of Raw Materials to Customer, and Customer shall pay Corden the costs of such Raw Materials. Notwithstanding the foregoing or anything to the contrary contained herein, from and after the delivery of any notice of termination pursuant to this Agreement, Corden shall not replenish (or otherwise add any additional quantities of Product to) any Safety Stock then being held for Customer. (d) Upon expiration or termination of this Agreement, Customer and Corden shall immediately settle all outstanding invoices and other monies owed to the other pursuant to this Agreement. The termination or expiration of this Agreement shall not affect the rights and obligations of the Parties accruing prior to such termination or expiration, including, but not limited to, Customer’s reimbursement to Corden for any work in progress and all non-cancelable commitments to purchase Raw Materials entered into by Corden specifically to conduct the Services hereunder that Corden cannot reasonably utilize in other projects and that meet the relevant specifications therefor that had been agreed upon by the Parties in writing. Subject to the foregoing, expiration or termination of this Agreement shall relieve and release the Parties from any liabilities and obligations under this Agreement, other than those specifically set forth in this Section 13.6 and those that survive termination in accordance with Section 13.7. (e) Upon written request of Customer, Corden shall promptly (within no more than thirty (30) days following receipt of such request) initiate transfer to Customer in writing of all Corden Background IP and Project Inventions that Corden incorporates into the Manufacturing Process for the Product pursuant to this Agreement, including, but not limited to, information concerning the test methods for the Product that are performed hereunder as may be required for Customer or a Third Party to perform Manufacturing or testing of the Product according to the processes and methods performed by Corden under this Agreement. Customer shall be entitled to use and transfer such information to one or more Third Parties for the sole purpose of Manufacturing or testing the Product. Customer agrees to pay for Corden’s actual cost of transfer including, but not limited to, Corden’s out of pocket expenses, working hours of its personnel, and the compilation of the technology transfer plan. To the extent that Corden designates specific items of such Corden Background IP or Project Inventions transferred pursuant to this Section 13.6(e) to be proprietary and confidential in writing at the time of transfer to Customer, Customer shall obtain confidentiality and nondisclosure agreements from Third Party(ies) to which Customer transfers such Corden Background IP and Project Inventions, in form and substance approved by Corden, which such approval shall not be unreasonably withheld and the Parties shall discuss in good faith if such confidentiality and non-disclosure agreement should be between Customer, Corden and such Third Party(ies) or between Customer and such Third Party(ies). 13.7 Survival. The following Sections shall survive the termination or expiration of this Agreement for any reason: Sections 1, 2.5, 2.6(f), 5.1-5.4, 6, 8.1, 8.5, 8.6, 8.7, 10.2-10.8, 11, 12, 13.6- 13.7, 14, 16.1-16.2, 16.5, and 16.7-16.19, each for the period specified therein, or, if no period is specified therein, then perpetually. Expiration or termination of this Agreement shall not relieve the Parties of any rights, causes of action, or obligations accruing prior to such expiration or termination.

33 14. LEGAL COMPLIANCE 14.1 Anti-Corruption. Each Party will conduct itself and undertake the arrangements contemplated by this Agreement in a manner which is consistent with the applicable anti- corruption legislation (national and foreign), including but not limited to the German laws regarding corruption, the U.S. Foreign Corrupt Practices Act and the U.K. Bribery Act 2010, as amended. Failure of a Party to comply with the provisions of this Section will be deemed a material breach of this Agreement. 14.2 Export Control. (a) Customer agrees and understands that the Product and other Corden deliverable may be subject to trade restrictions (e.g. export license requirements, sanctions programs in terms of embargoes or sanctioned party lists) by the Applicable Laws of France, the EU and the U.S. as well as international laws (hereinafter “Export Control Regulations”) imposing restrictions on trade including export, re-export, transfer, disclosure, supply or comparable transaction, regardless of the way of provision to other countries or parties. Customer shall comply with all such Export Control Regulations. In particular, Customer shall not, without first obtaining permission to do so from the appropriate authorities, (i) provide any such Product or Corden deliverable to any country, person or other party that is ineligible to receive such Product or Corden deliverable under the Export Control Regulations (hereinafter “Embargoed Country or Embargoed Party”); or (ii) provide any Product or Corden deliverable to a person or other party if Customer knows or has reason to assume that such person or other party intends to provide the Product or Corden deliverable to any such Embargoed Country or Embargoed Party, or intends to use or allow others to use the Product or Corden deliverable for activities related to military or otherwise restricted use. Customer shall cooperate fully with Corden in any official or unofficial audit or inspection related to this Agreement in connection with any Export Control Regulations. (b) Neither Party shall disclose Confidential Information to the other Party in violation of the Export Control Regulations. In the event that either Party knows, or has reason to believe that any Confidential Information to be disclosed hereunder is subject to the Export Control Regulations, such Party shall notify the other Party in advance of planned disclosure and before disclosure the Parties will reasonably cooperate to ensure compliance therewith. In no case shall such Confidential Information be disclosed to the other Party without its prior written agreement. 14.3 Data Protection. Each Party shall comply with applicable data protection laws, to the extent that such Party receives and/or processes and uses personal data in the performance of its obligations under this Agreement. Upon a Party’s request, the other Party agrees to enter into any additional agreements required by Applicable Law, and specifically the European General Data Protection Regulation 2016/679 (GDPR), relating to data use and protection. In case of transfer of personal data to recipients seated outside the European Union/European Economic Area, which do not provide for an adequate data protection level, such contractual arrangements may include (i) the European Union’s Standard Contractual Clauses/Standard Data Protection Clauses for the transfer of personal data to processors and/or (ii) any other agreement that competent data protection authorities have declared to be compulsory or acceptable to comply with data protection law obligations. 15. FORCE MAJEURE Neither Party shall be held liable or responsible to the other Party nor be deemed to be in default under, or in breach of any provision of, this Agreement for failure or delay in fulfilling or performing any obligation of this Agreement when such failure or delay is due to Force Majeure, and without the fault or negligence of the Party so failing or delaying. For purposes of this Agreement, “Force Majeure” is defined as unforeseeable causes beyond the control of a Party, including without limitation, acts of God; war; civil commotion; epidemics, quarantines, and the

34 destruction of production facilities or materials by fire, flood, earthquake, explosion or storm. In such event, the Party experiencing such Force Majeure event, shall immediately notify the other Party of such inability and of the period for which such inability is expected to continue. The Party giving such notice shall thereupon be excused from such of its obligations under this Agreement as it is thereby disabled from performing for so long as it is so disabled and for fifteen (15) days thereafter. To the extent possible, each Party shall use commercially reasonable efforts to minimize the duration of any event of Force Majeure. If Corden is unable to perform its obligations due to a Force Majeure event, Customer shall be entitled to obtain immediately any Customer Material, and/or other Raw Materials dedicated exclusively to the Product or work-in process involving Customer Material then in the custody of Corden so that Customer may arrange for the production or completion of Product by other manufacturers, in its discretion. If such Force Majeure event is expected to delay production for more than thirty (30) days, the Parties shall immediately consult with each other to consider how to best address such delay. 16. MISCELLANEOUS 16.1 Relationship of Parties. Nothing in this Agreement is intended or shall be deemed to constitute a partnership, agency, employer-employee or joint venture relationship between the Parties. The Parties’ obligations and rights in connection with the subject matter hereof are solely as specifically set forth in this Agreement, and they agree and acknowledge that they owe no fiduciary or other duties or obligations to each other by virtue of any relationship created by this Agreement. Without limiting the foregoing, the Parties also acknowledge and agree that if it should be determined by a court of competent jurisdiction that, notwithstanding the foregoing, such duties or obligations exist, the Parties hereby waive same and agree not to assert or rely on same in any proceeding. Neither Party shall incur any debts or make any commitments for the other, except to the extent, if at all, specifically provided herein. Customer shall sell the Product without participation of Corden in the negotiation or consummation of such sales, and, as between the Parties, Customer shall derive the entire income and incur the entire loss, as the case may be, from such sales. Corden shall only be entitled to the applicable properly invoiced Fees as set forth in this Agreement. 16.2 Assignment. Neither Party shall be entitled to assign its rights or delegate its obligations hereunder without the express prior written consent of the other Party, not to be unreasonably withheld, except that: (i) Customer may assign its rights and delegate its obligations hereunder, in whole or in part, to an Affiliate of Customer; (ii) Customer may assign its rights and delegate its obligations hereunder with respect to the Product to a Person who acquires all or substantially all of Customer’s business, rights or obligations with respect to the Product or the applicable Product line; and (iii) Customer may assign its rights and delegate all of its obligations hereunder in the event of Customer’s or its Affiliates’ merger or consolidation, sale of all or substantially all its assets or business or similar transaction. Any assignment not in accordance with this Section 16.2 shall be null and void. 16.3 Exception for Trade Receivables. Corden may, upon notice to Customer, sell its trade receivables to a Third Party or an Affiliate (so long as the recipients are not direct competitors of the Customer), provided that any such Affiliate or Third Party expressly acknowledges in writing that Customer’s obligations with respect to payment of such trade receivables shall at all times remain subject to the terms of this Agreement. Customer hereby grants permission for Corden to disclose the provisions of this Agreement to purchasers and prospective purchasers of receivables, or their Affiliates (so long as the recipients are not direct competitors of the Customer), and their respective agents, attorneys, auditors, rating agencies, and other advisors; provided that prior to making any such disclosure Corden has entered into a written confidentiality agreement with any such person or entity that contains terms consistent with Section 11.1 to protect the confidentiality of the terms of this Agreement.

35 16.4 Sub-contracting. Corden shall not sub-contract any of the work to be performed by Corden hereunder without the prior written consent of Customer. No such sub-contracting, even if approved by Customer, shall relieve Corden of any of its obligations hereunder. 16.5 Binding Effect; No Third Party Beneficiaries. Except as otherwise provided in this Agreement, this Agreement shall be binding on the successors and permitted assigns of the Parties, each of such permitted universal successors or assigns being deemed to be a Party hereunder in substitution of its respective predecessor. This Agreement is for the sole benefit of the Parties and their respective successors, Affiliates and permitted assigns, and in the case of Customer, as applicable, its Partners, and nothing herein expressed or implied shall give or be construed to give to any Person, other than the Parties and such assigns or Partners, any legal or equitable rights hereunder. 16.6 Further Actions. Each Party agrees to execute, acknowledge and deliver such further instruments, and to do all such other acts, as may be reasonably necessary or appropriate in order to carry out the purposes and intent of this Agreement. 16.7 Notices. Any notice, request or other communication required or permitted to be given under or in connection with this Agreement shall be deemed to have been sufficiently given solely if in writing and personally delivered or sent by email transmission (receipt verified, with an automatic “read receipt” not constituting verification of receipt for purposes of this Section 16.7) or express courier service for overnight delivery (with delivery tracking provided, signature required and delivery prepaid) to the Party for which such notice is intended, at the address set forth below for such Party: (a) In the case of Corden, to: Corden Pharma GmbH Attention: Managing Director Otto-Hahn-Strasse, 68723 Plankstadt, Germany Email: [***] (primary recipient) Copy to: Corden Pharma International Switzerland LLC Attn.: Legal Department Aeschenvorstadt 67-71, 4051 Basel, Switzerland [***] (b) In the case of Customer, to: Madrigal Pharmaceuticals, Inc. Four Tower Bridge 200 Barr Harbor Drive, Suite 200 Conshohocken, PA 19428 Attention: [***] Email: [***] (primary recipient) Copy to: Madrigal Pharmaceuticals, Inc. Four Tower Bridge 200 Barr Harbor Drive, Suite 200 Conshohocken, PA 19428 Attention: General Counsel or to such other address for such Party as it shall have specified by like notice to the other Party; provided that notices of a change of address shall be effective only upon receipt thereof. If delivered personally, the date of delivery shall be the date on which such notice or request has been given if delivered on any Business Day in the delivery location prior to 6 pm local time or on the next succeeding Business Day if delivered in person on a non-

36 Business Day or after 6 pm local time. If sent by express courier, the date of delivery shall be one (1) Business Day after having been delivered to the express courier for overnight delivery (with delivery tracking provided, signature required and delivery prepaid). If sent by email, the date of delivery shall be when the primary recipient, by an email sent to the email address for the sender stated in this Section 16.7 or by a notice delivered by another method in accordance with this Section 16.7, acknowledges having received that email, with an automatic “read receipt” not constituting acknowledgment of an email for purposes of this Section 16.7. All notices, requests or other communications required by this Agreement shall be in the English language. 16.8 Use of Name. Except as otherwise provided herein, neither Party shall have any right, express or implied, to use in any manner the name or other designation of the other Party or any other trade name or trademark of the other Party for any purpose in connection with the performance of this Agreement. 16.9 Public Announcements. Neither Party shall make any public announcement regarding this Agreement without the prior written consent of the other, which consent shall not be unreasonably withheld, conditioned or delayed; provided, that the provisions of this Section 16.9 will not prohibit (a) any disclosure required by any applicable legal requirement, including any legal requirement or listing standard of any exchange or quotation system on which the disclosing Party’s securities are listed or traded or to be listed or traded (in which case the disclosing Party will provide the other Party with the opportunity to review in advance the disclosure and to contest the same, including reasonable opportunity to seek a protective order or to seek confidential treatment of such disclosures under Rule 24b-2 of the Securities Exchange Act of 1934, as amended), (b) any disclosure made in connection with the enforcement of any right or remedy relating to this Agreement, (c) any disclosure made by Customer or Corden to their respective employees, collaborators, licensors, licensees, contract research organizations, business partners, investors, potential investors, lenders and potential lenders provided the person receiving the disclosure has undertaken a confidentiality obligation to Customer or Corden, as applicable, substantially similar to the confidentiality obligations the Parties have undertaken to each other under this Agreement, or (d) any disclosure made pursuant to a press release in a form mutually agreed to by the Parties (or any other subsequent disclosure containing substantially similar information). In the event of permitted public announcement, or of an announcement required by Applicable Law, regulation or a Governmental Authority, the Party making such announcement shall provide the other Party with a copy of the proposed text prior to such announcement sufficiently in advance of the scheduled release of such announcement to afford such other Party a reasonable opportunity to review and comment upon the proposed text, except where such prior disclosure is not permitted by Applicable Law or regulation or would otherwise jeopardize the timely delivery by the other Party of any required public announcement. Following approval of the proposed text, such text may be used in subsequent public announcements without further approval, to the extent it remains accurate, complete and not misleading. 16.10 Waiver. Any waiver by a Party of a breach of any provision of this Agreement shall not operate as, or be construed to be, a waiver of any other breach of such provision or of any breach of any other provision of this Agreement. The failure of a Party to insist upon strict adherence to any term of this Agreement on one or more occasions shall not be considered a waiver or deprive that Party of the right thereafter to insist upon strict adherence to that term or any other term of this Agreement. Any waiver must be in writing and signed by the duly authorized representative of the waiving Party. 16.11 Severability. Whenever possible, each provision of this Agreement shall be interpreted in such manner as to be effective and valid under Applicable Law, but if any provision of this Agreement is held to be illegal, invalid or unenforceable under present or future laws effective during the Term, then (i) in lieu of such illegal, invalid or unenforceable provision, the Parties, acting in good faith shall amend this Agreement to replace such provision (from the time of its invalidity, unenforceability, or illegality) with a new appropriate and permissible provision as similar in terms to the intention of the Parties with respect to such illegal, invalid

37 or unenforceable provision as may be possible, and (ii) such illegal, invalid or unenforceable provision shall not affect or impair the legality, validity and enforceability of the remaining provisions of this Agreement in any way. 16.12 Amendment. This Agreement may only be amended in writing and signed by the duly authorized representatives of both Parties. 16.13 Governing Law; English Original Controlling. This Agreement shall be governed by and interpreted in accordance with the laws of New York, U.S.A. without giving effect to any choice of law principles that would result in the application of the laws of any other jurisdiction. The application of the United Nations Convention on Contracts for the International Sale of Goods (CISG) shall be excluded. The English original of this Agreement shall prevail over any translation hereof. 16.14 Dispute Resolution. (a) All disputes, controversies, and claims directly or indirectly arising out of or in relation to this Agreement or the validity, interpretation, construction, performance, breach or enforceability of this Agreement shall be finally, exclusively and conclusively settled by binding arbitration, as provided in this Section 16.14, under the International Chamber of Commerce Rules of Arbitration which are in effect as of the Effective Date (“Rules”). (b) Any legal action or proceeding will be heard by one arbitrator appointed in accordance with said Rules. Unless the Parties otherwise agree to a different place of arbitration, the place of arbitration will be in Zurich, Switzerland. The arbitration language will be English. The arbitration award will be final and binding upon the Parties. Both Parties consent to the exclusive jurisdiction of such arbitration procedure and waive any objection to the propriety or convenience of such venues. Nothing in this clause shall preclude either Party from seeking interim or provisional relief, including a temporary restraining order, preliminary injunction or other interim equitable relief, if such Party thinks this is necessary to protect its interests. (c) The Parties agree (i) that any such award or order shall be a reasoned award, shall be in writing and in English, shall specify the factual and legal basis for the award and shall be final and binding; and (ii) that judgment or any arbitral award or order resulting from an arbitration conducted under this Section 16.14 may be entered in any court, in any country of competent jurisdiction, having jurisdiction thereof or having jurisdiction over the Parties or any of their respective assets. (d) Customer and Corden hereby irrevocably waive and exclude all rights of appeal, challenge or recourse to any court from any arbitral award or order resulting from any arbitration conducted under this Section 16.14 (except for initiating actions or proceedings to obtain a judgment recognizing or enforcing an arbitral award or order and except for actions or proceedings seeking interim, interlocutory or other provisional relief in any court having jurisdiction, but only on the ground that the award to which the applicant may be entitled may be rendered ineffectual without such provisional relief). (e) The arbitrator, in his or her discretion, may consolidate two or more arbitrations or claims between the Parties into one arbitration, or terminate any such consolidation and/or establish other arbitration proceedings for different claims that may arise in any one arbitration. Notwithstanding the foregoing, the arbitrator shall consolidate arbitrations and/or claims, if they determine that it would be more efficient to consolidate such arbitrations and/or claims than to continue them separately and (i) there are matters of fact or law that are common to the arbitrations and/or claims to be consolidated, (ii) there are related payment and performance obligations considered in the arbitrations and/or claims to be consolidated, or (iii) there is a danger of inconsistent awards.

38 16.15 Interpretation. Except as the context otherwise requires, (i) any reference in this Agreement to a Section, subsection, paragraph, clause or Exhibit will be deemed to be a reference to a Section, subsection, paragraph, clause or Exhibit of or to this Agreement, (ii) any definition of or reference to any agreement, instrument or other document refers to such agreement, instrument other document as from time to time amended, supplemented or otherwise modified (subject to any restrictions on such amendments, supplements or modifications set forth herein or therein); (iii) the words “herein,” “hereof” and “hereunder,” and words of similar import, refer to this Agreement in its entirety and not to any particular provision hereof; (iv) the word “include” (and variants thereof) shall be deemed to be followed by the phrase “without limitation” or words of similar import, and (v) except as otherwise expressly stated or unless the context otherwise requires, all references to the singular will include the plural and vice versa. 16.16 Entire Agreement; Priority of Documents. This Agreement together with the appendices and schedules attached hereto and thereto, contain the entire understanding and agreement among the Parties with respect to the subject matter hereof and supersede all prior oral and written understandings and agreements relating thereto. In the event of a conflict or ambiguity between any term of this Agreement, its Appendices, the Quality Agreement and any Purchase Order, the following order of precedence shall apply, whereas 1 equals to the highest priority. The document with the higher priority shall prevail over the document with the lower priority. 1. This Agreement; 2. Its Appendices (including any Product Addendum except to the extent such Product Addendum expressly provides that its terms are intended to supersede specific clauses of this Agreement); 3. Quality Agreement ( except to the extent such conflict relates to quality related matters, including compliance with cGMP, in which case the Quality Agreement shall be of highest priority and shall prevail over this Agreement, its Appendices and any Purchase Order), but, in the event of a conflict between any of the provisions of this Agreement and the Quality Agreement with respect to any commercial matters, including allocation of risk, liability and financial responsibility, the provisions of this Agreement shall control; 4. Any Purchase Order (except to the extent that such Purchase Order expressly and conspicuously contains terms intended to supersede the terms and conditions of this Agreement or its Appendices, in which case the Purchase Order supersedes this Agreement only with regard to such specific terms). 16.17 MSA. The Parties hereby agree that the MSA shall continue in full force and effect for the currently outstanding Statements of Work and for additional Statements of Work (as defined in the MSA), as necessary, and this Agreement shall have no impact upon the terms and conditions of the MSA, nor shall the terms and conditions of the MSA have any impact upon the terms and conditions of this Agreement. The Parties intend that the MSA shall remain in effect and not be replaced by this Agreement. To the extent there are any inconsistencies or conflicts between this Agreement and the MSA or a Statement of Work under the MSA, the terms and conditions of this Agreement shall control with respect to the transactions contemplated by this Agreement and the terms of the MSA shall control with respect to the transactions contemplated by the MSA and subject to a Statement of Work under the MSA, including for the avoidance of doubt the Product that is subject to Statement of Work Nos. 1, 2, 3, 4, and 5 under the MSA. 16.18 Descriptive Headings. The headings contained in this Agreement, in the table of contents to this Agreement and in any exhibits or schedules to this Agreement are for reference purposes only and shall not affect in any way the meaning or interpretation of this Agreement.

39 16.19 Counterparts. This Agreement is executed in two or more counterparts, each of which shall be deemed an original, but all of which together shall constitute one and the same instrument. The Parties agree that execution of this Agreement by industry standard electronic signature software or by exchanging executed signatures by facsimile, email, portable document format (.pdf) or by any other electronic means intended to preserve the original graphic and pictorial appearance of this Agreement shall have the same legal force and effect as the physical delivery of the paper document bearing original signature. [signature page follows]

40 IN WITNESS WHEREOF, the Parties hereto have caused this Agreement to be executed, as of the Effective Date, by their duly authorized representatives. CORDEN PHARMA GmbH MADRIGAL PHARMACEUTICALS, INC. By: /s/ Stephan Haitz By: /s/ Bill Sibold Name: Stephan Haitz Name: Bill Sibold Title: CCO Date: 12/17/2024 Title:CEO Date: 12/18/2024 By: /s/ Christian Merz Name: Christian Merz Title: Managing Director Date: 12/17/2024 Enclosures: Appendix 1 – Description of Product Appendix 2 – Product Addendum Appendix 3 – Product Price Appendix 4 – API Specifications Appendix 5 – Initial Rolling Forecast Appendix 6 – Safety Stock Materials Appendix 7 – Quality Agreement

FIRST AMENDMENT TO COMMERCIAL SUPPLY AGREEMENT by and between Madrigal Pharmaceuticals, Inc. (“Madrigal”) and Corden Pharma GmbH (“Corden”) This First Amendment to Commercial Supply Agreement (“Amendment”) is effective as of the last date of signature below (hereinafter “Effective Date”), and is made by and between Madrigal and Corden, to that certain Commercial Supply Agreement dated as of December 18, 2024 (the “Agreement”), and shall be deemed to be a part thereof. WHEREAS, the parties wish to modify the Agreement as set forth below; NOW THEREFORE, for good and valuable consideration, the receipt of which is hereby acknowledged; and subject to the terms and conditions of the Agreement, Madrigal and Corden hereby agree that the Agreement is amended as follows: 1. Strike Appendix 3 - Product Price of the Agreement in its entirety and replace it with the attached Appendix 3 Product Price Except as herein amended, all terms, covenants and provisions of the Agreement are ratified, reaffirmed, and shall remain in full force and effect. All references therein to the Agreement shall hereafter refer to the Agreement as amended by this Amendment. All capitalized, undefined terms shall have the same meaning as defined in the Agreement. This Amendment shall be deemed incorporated into, and a part of, the Agreement. IN WITNESS WHEREOF, the parties have executed this Amendment as of the last date of signature below. Acknowledged and Agreed to: Madrigal Pharmaceuticals, Inc. Corden Pharma GmbH By: /s/ Vincent Pisculli By: /s/ Christina Simons Signature Signature Vincent Pisculli Christina Simons Name Name VP, Supply Chain Dir. Project Management & Communication Title Title 3/4/2025 3/4/2025 Date Date Corden Pharma GmbH By: /s/ Christian Merz Signature Christian Merz Name Managing Director Title 3/4/2025 Date

SECOND AMENDMENT TO COMMERCIAL SUPPLY AGREEMENT by and between Madrigal Pharmaceuticals, Inc. (“Madrigal”) and Corden Pharma GmbH (“Company”) This Second Amendment to Commercial Supply Agreement (“Amendment”) is effective as of the last date of signature below (hereinafter “Effective Date”), and is made by and between Madrigal and Company, to that certain Commercial Supply Agreement dated as of December 18, 2024 and as amended (the “Agreement”), and shall be deemed to be a part thereof. WHEREAS, the parties wish to modify the Agreement as set forth below; NOW THEREFORE, for good and valuable consideration, the receipt of which is hereby acknowledged; and subject to the terms and conditions of the Agreement, Madrigal and Company hereby agree that the Agreement is amended as follows: 1. Change frequency of Forecasts (Section 3.1(b)) - Forecasts to change to monthly from quarterly forecasts with PO issued within 90 days. 2. Strike Appendix 3 - Product Price of the Agreement in its entirety and replace it with the attached Appendix 3 Product Price. Except as herein amended, all terms, covenants and provisions of the Agreement are ratified, reaffirmed, and shall remain in full force and effect. All references therein to the Agreement shall hereafter refer to the Agreement as amended by this Amendment. All capitalized, undefined terms shall have the same meaning as defined in the Agreement. This Amendment shall be deemed incorporated into, and a part of, the Agreement. IN WITNESS WHEREOF, the parties have executed this Amendment as of the last date of signature below. Acknowledged and Agreed to: Madrigal Pharmaceuticals, Inc. Corden Pharma GmbH By: /s/ Vincent Pisculli By: /s/ Christina Simons Signature Signature Vincent Pisculli Christina Simons Name Name VP, Supply Chain Director, Project Management & Communication Title Title 5/5/2025 5/4/2025 Date Date Corden Pharma GmbH By: /s/ Christian Merz Signature Christian Merz Name Managing Director Title 5/4/2025

THIRD AMENDMENT TO COMMERCIAL SUPPLY AGREEMENT by and between Madrigal Pharmaceuticals, Inc. (“Madrigal”) and Corden Pharma GmbH (“Corden”) This Third Amendment to Commercial Supply Agreement (“Amendment”) is effective as of the last date of signature below (hereinafter “Effective Date”), and is made by and between Madrigal and Corden, to that certain Commercial Supply Agreement dated as of December 18, 2024 (the “Agreement”), and shall be deemed to be a part thereof. WHEREAS, the parties wish to modify the Agreement as set forth below; NOW THEREFORE, for good and valuable consideration, the receipt of which is hereby acknowledged; and subject to the terms and conditions of the Agreement, Madrigal and Corden hereby agree that the Agreement is amended as follows: 1. Strike Appendix 3 - Product Price of the Agreement in its entirety and replace with the attached Appendix 3 Product Price Except as herein amended, all terms, covenants and provisions of the Agreement are ratified, reaffirmed, and shall remain in full force and effect. All references therein to the Agreement shall hereafter refer to the Agreement as amended by this Amendment. All capitalized, undefined terms shall have the same meaning as defined in the Agreement. This Amendment shall be deemed incorporated into, and a part of, the Agreement. IN WITNESS WHEREOF, the parties have executed this Amendment as of the last date of signature below. Acknowledged and Agreed to: Madrigal Pharmaceuticals, Inc. Corden Pharma GmbH By: /s/ Vincent Pisculli By: /s/ Oliver Schinzinger Signature Signature Vincent Pisculli Oliver Schinzinger Name Name VP, Supply Chain Director of Pharmaceutical Development Title Title 8/26/2025 8/25/2025 Date Date Corden Pharma GmbH By: /s/ Christian Merz Signature Christian Merz Name Managing Director Title 8/25/2025 Date

FOURTH AMENDMENT TO COMMERCIAL SUPPLY AGREEMENT by and between Madrigal Pharmaceuticals, Inc. (“Madrigal”) and Corden Pharma GmbH (“Corden”) (“Company”) This Fourth Amendment to Commercial Supply Agreement (“Amendment”) is effective as of the last date of signature below (hereinafter “Effective Date”), and is made by and between Madrigal and Company, to that certain Commercial Supply Agreement dated as of December 18, 2024 (the “Agreement”), and shall be deemed to be a part thereof. WHEREAS, the parties wish to modify the Agreement as set forth below; NOW THEREFORE, for good and valuable consideration, the receipt of which is hereby acknowledged; and subject to the terms and conditions of the Agreement, Madrigal and Company hereby agree that the Agreement is amended as follows: 1. Strike Appendix 3 - Product Price of the Agreement in its entirety and replace it with the attached Appendix 3 Product Price Except as herein amended, all terms, covenants and provisions of the Agreement are ratified, reaffirmed, and shall remain in full force and effect. All references therein to the Agreement shall hereafter refer to the Agreement as amended by this Amendment. All capitalized, undefined terms shall have the same meaning as defined in the Agreement. This Amendment shall be deemed incorporated into, and a part of, the Agreement. IN WITNESS WHEREOF, the parties have executed this Amendment as of the last date of signature below. Acknowledged and Agreed to: Madrigal Pharmaceuticals, Inc. Corden Pharma GmbH By: /s/ Vincent Pisculli By: /s/ Oliver Schinzinger Signature Signature Vincent Pisculli Dr. Oliver Schinzinger VP, Supply Chain Director R&D, Technology and Innovation 1/21/2026 1/21/2026 Date Date Corden Pharma GmbH By: /s/ Juergen Nauert Signature Juergen Nauert Finance Director 1/21/2026 Date