Franchise Revenue Performance
U.S. Glaucoma
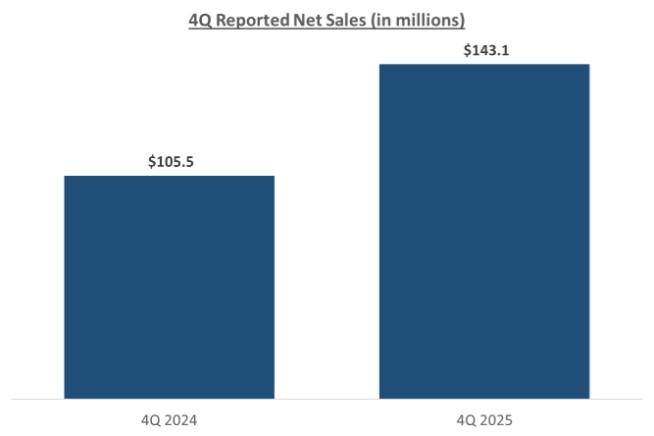
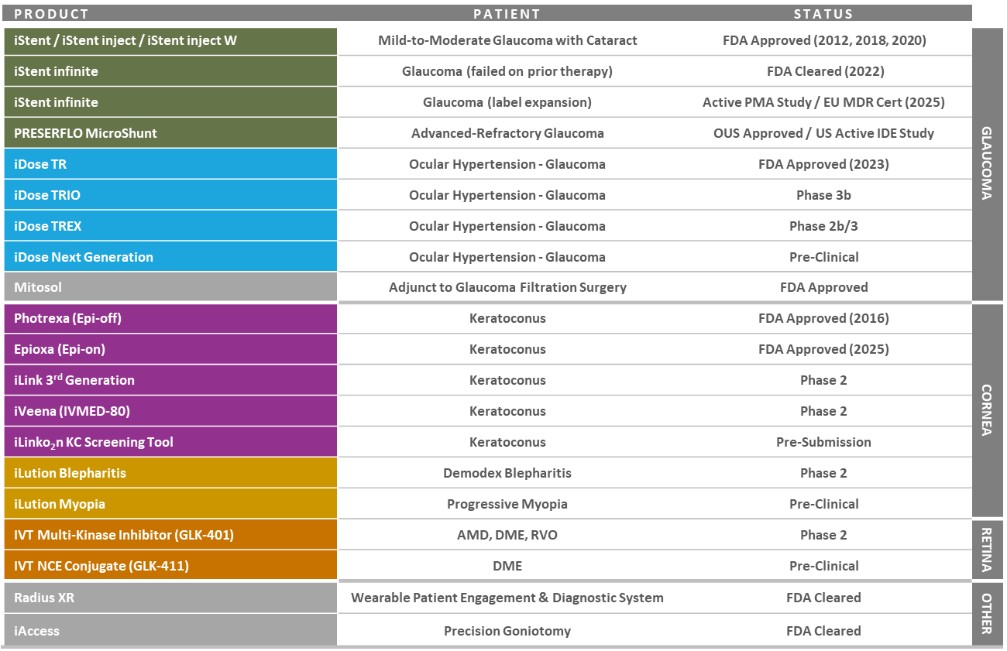
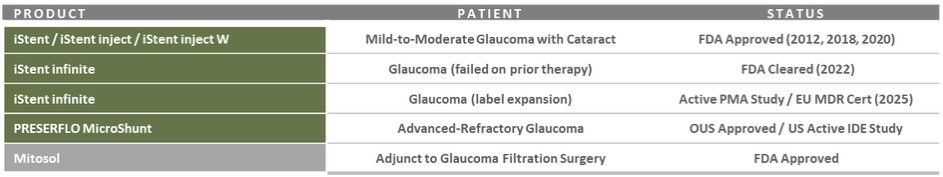
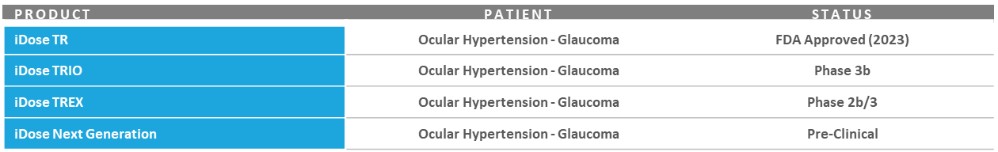
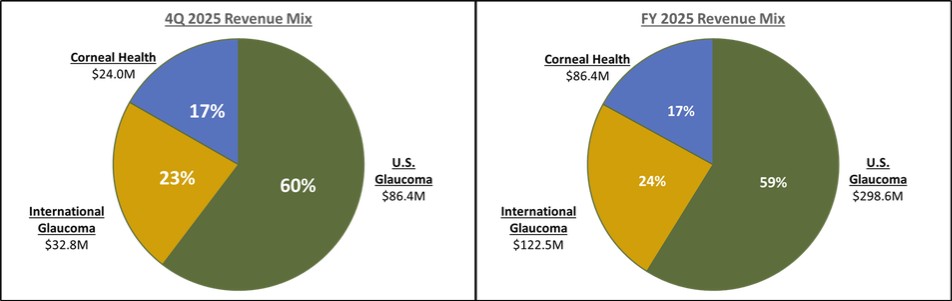
Our record fourth quarter U.S. Glaucoma net revenues were approximately $86.4 million, representing year-over-year growth of 53% versus 4Q 2024 driven by growing contributions from iDose TR, which generated sales of approximately $45 million in the third quarter.
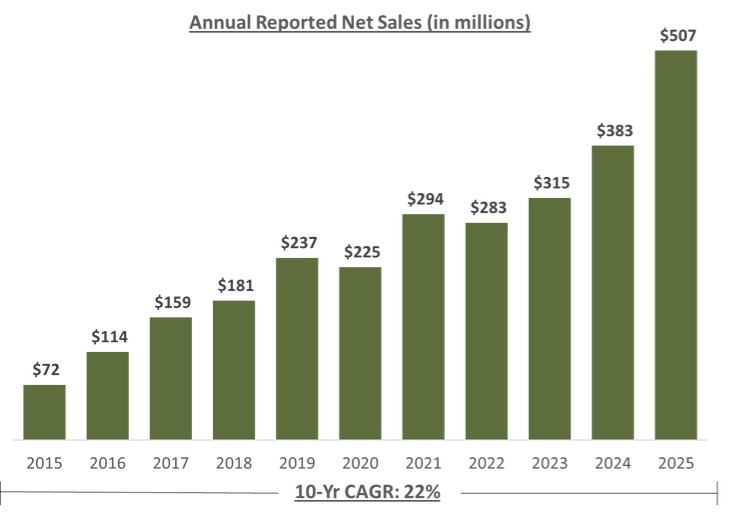
For fiscal year 2025, U.S. Glaucoma net revenues were approximately $298.6 million, representing year-over-year growth of 50% versus fiscal year 2024, including iDose TR sales of approximately $136 million.
During the fourth quarter, we successfully advanced execution of our detailed launch plans for iDose TR, a first-of-its-kind intracameral procedural pharmaceutical that was designed to continuously deliver glaucoma drug therapy for up to three years. Most importantly, clinical outcomes and product feedback from a growing number of cases and trained surgeons continue to be very positive and reaffirm our view that with the launch of iDose TR, we have the potential to reshape glaucoma management as we know it today.
International Glaucoma
Our record fourth quarter International Glaucoma net revenues were approximately $32.8 million, representing year-over-year growth of 18% on a reported basis, or 13% on a constant currency basis, versus 4Q 2024. The strong growth internationally during the fourth quarter was broad-based as we continue to scale our international infrastructure and increasingly drive MIGS forward as the standard of care in each region and major market in the world.
For fiscal year 2025, International Glaucoma net revenues were approximately $122.5 million, representing year-over-year reported growth of 18%, or 16% on a constant currency basis, versus fiscal year 2024.
We remain in the early stages of expanding our IG and product portfolio initiatives globally ahead of anticipated new product approvals and expanding market access in the years to come.
Corneal Health
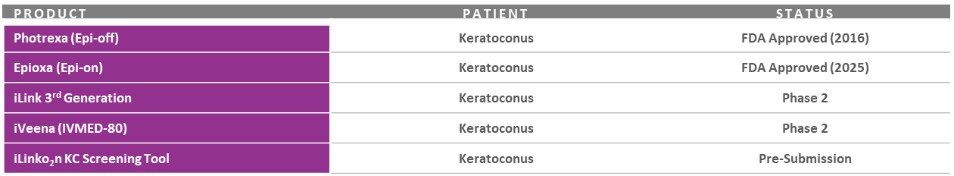
Our record fourth quarter Corneal Health net revenues were approximately $24.0 million, representing year-over-year growth of 12% versus 4Q 2024, including U.S. Photrexa® net sales of $21.4 million. As