| are trademarks of MaxCyte, Inc. in the U.S.A. Driving the Next Generation of Cell-Based Therapies MaxCyte Corporate Presentation NASDAQ: MXCT March 2026 © 2026 MaxCyte, Inc. All Rights Reserved |
| Forward Looking Statement Disclaimer 2 © 2026 MaxCyte, Inc. All Rights Reserved Certain statements in this document (this “Presentation”) are, or may be deemed to be, forward-looking statements within the meaning of the “safe harbor” provisions of the Private Securities Litigation Reform Act of 1995, including but not limited to statements regarding our expected potential future revenue. These statements about us and our industry involve substantial known and unknown risk, uncertainties and assumptions, that may cause our actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward-looking statements. All statements other than statements of historical facts contained in this Presentation are forward-looking statements. The words "may," "might," "will," "could," "would," "should," "expect," "plan," "anticipate," "intend," "believe," "expect," "estimate," "seek," "predict," "future," "project," "potential," "continue," "target" and similar words or expressions are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words. Any forward-looking statements in this Presentation are based on management's current expectations and beliefs and are subject to a number of risks, uncertainties and important factors that may cause actual events or results to differ materially from those expressed or implied by any forward-looking statements contained in this Presentation, including, without limitation, statements regarding the Company's future growth, results of operations, performance, future capital and other expenditures (including the amount, nature and sources of funding thereof), competitive advantages, business prospects and opportunities. These and other risks and uncertainties are described in greater detail in the section entitled "Risk Factors" in our Annual Report on Form 10-K for the year ended December 31, 2025, as well as discussions of potential risks, uncertainties, and other important factors in the other filings that we make with the Securities and Exchange Commission from time to time. These documents are available, without charge, on the Securities and Exchange Commission website and through the Investor Menu, Financials section under "SEC filings" on the Investors page of our website at http://investors.maxcyte.com. No statement in this Presentation is intended to be, or intended to be construed as, a profit forecast or profit estimate or to be interpreted to mean that earnings per Company share for the current or future financial years will necessarily match or exceed the historical earnings per Company share. As a result, no undue reliance should be placed on such statements. Any forward-looking statements represent our views only as of the date of this Presentation and should not be relied upon as representing our views as of any subsequent date. We explicitly disclaim any obligation to update any forward-looking statements, whether as a result of new information, future events or otherwise, except as required by law. No representations or warranties (expressed or implied) are made about the accuracy of any such forward-looking statements. This Presentation contains Adjusted EBITDA, which is a non-GAAP measure defined as earnings before interest, taxes, depreciation, amortization, goodwill impairment and one-time restructuring charges. MaxCyte believes that Adjusted EBITDA provides useful information to management and investors relating to its results of operations. The company’s management uses these non-GAAP measures to compare the company’s performance to that of prior periods for trend analyses, and for budgeting and planning purposes. The company believes that the use of Adjusted EBITDA provides an additional tool for investors to use in evaluating ongoing operating results and trends and in comparing the company’s financial measures with other companies, many of which present similar non-GAAP financial measures to investors, and that it allows for greater transparency with respect to key metrics used by management in its financial and operational decision-making. This Presentation also contains Non-GAAP Gross Margin, which we define as Gross Margin when excluding SPL program related revenue and reserves for excess and obsolete inventory. The Company believes that the use of Non-GAAP Gross Margin provides an additional tool to investors because it provides consistency and comparability with past financial performance, as Non-GAAP Gross Margin excludes non-core revenues and inventory reserves, which can vary significantly between periods and thus affect comparability. Management does not consider these Non-GAAP financial measures in isolation or as an alternative to financial measures determined in accordance with GAAP. The principal limitation of these Non-GAAP financial measures is that they exclude significant revenues and expenses that are required by GAAP to be recorded in the Company’s financial statements. Non-GAAP measures should be considered in addition to results prepared in accordance with GAAP, but should not be considered a substitute for, or superior to, GAAP results. A reconciliation table of Gross Margin, the most comparable GAAP financial measure to Non-GAAP Gross Margin, is included in the appendix of this Presentation. The Company urges investors to review the reconciliation and not to rely on any single financial measure to evaluate its business. |
| MaxCyte at a Glance 3 © 2026 MaxCyte, Inc. All Rights Reserved Our Mission We power the future of cell and gene therapy with innovative, scalable cell engineering solutions that enable our customers to deliver advanced therapies to patients Base-editing (CRISPR), CRISPR, ARCUS, RNA-Based Engineering, TALENS, Zinc Finger Nucleases (ZFNs) 13 Clinical and Commercial Therapies Supported 2025 Revenue • Genetic diseases, solid tumors, infectious disease, Hematological • Malignancies, autoimmune disease 31 SPL Customers $156M Cash & Cash Equivalents1 $33.0M 2025 Non-GAAP Adjusted Gross Margins 81% 2 1. As of December 31, 2025 2. Excluding SPL Program-related revenue and reserves for excess and obsolete inventory. See appendix for reconciliation to GAAP gross margins |
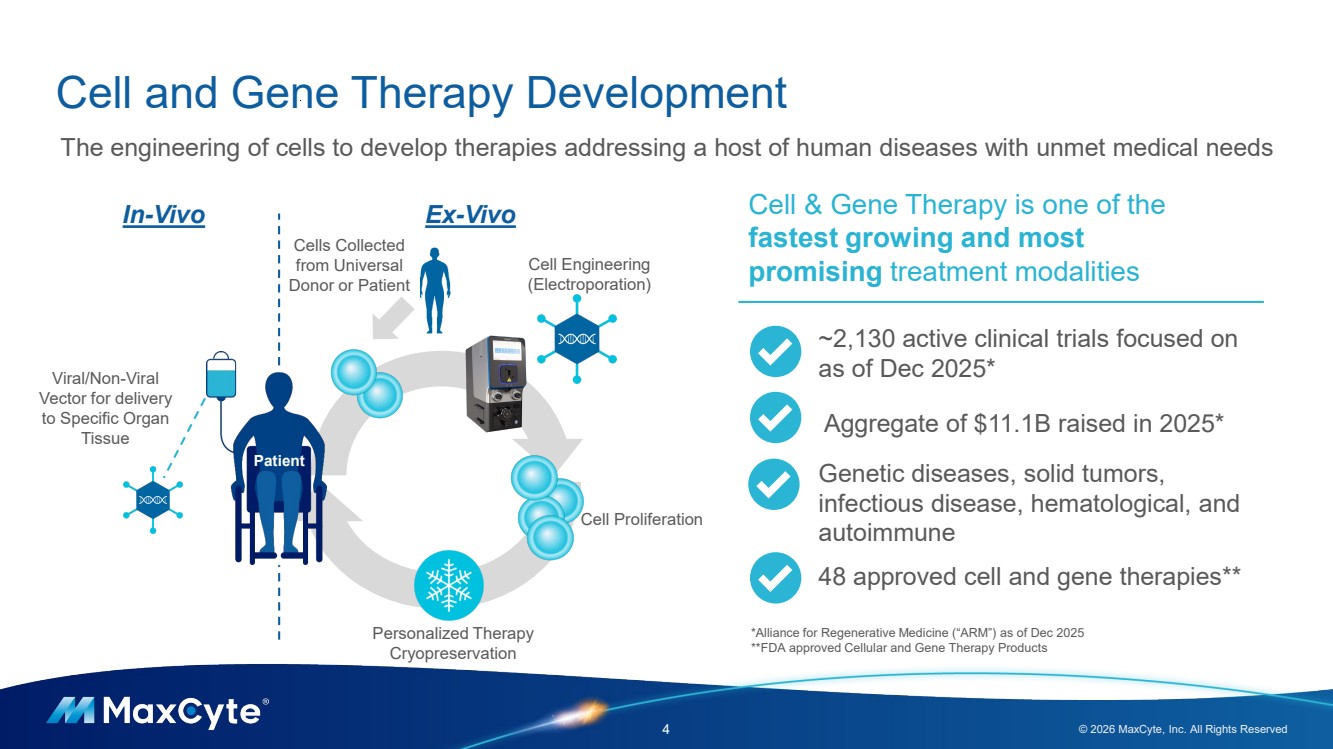
| Cell and Gene Therapy Development 4 © 2026 MaxCyte, Inc. All Rights Reserved Cell Engineering (Electroporation) Cell Proliferation In-Vivo Ex-Vivo Viral/Non-Viral Vector for delivery to Specific Organ Tissue The engineering of cells to develop therapies addressing a host of human diseases with unmet medical needs Cells Collected from Universal Donor or Patient Personalized Therapy Cryopreservation Patient Cell & Gene Therapy is one of the fastest growing and most promising treatment modalities ~2,130 active clinical trials focused on as of Dec 2025* Aggregate of $11.1B raised in 2025* Genetic diseases, solid tumors, infectious disease, hematological, and autoimmune 48 approved cell and gene therapies** *Alliance for Regenerative Medicine (“ARM”) as of Dec 2025 **FDA approved Cellular and Gene Therapy Products |
| Addressing the Challenges of Cell & Gene Therapy Development 5 © 2026 MaxCyte, Inc. All Rights Reserved Next-generation cell therapy programs have become increasingly complex requiring multiple edits Regulatory risk increases with new unknowns (donor cells, next-gen approaches, new indications) Vein-to-vein manufacturing times are high; optimizations needed to deliver medicines to patients faster Lack of industry standard for cell engineering process development causes costly and inconsistent manufacturing runs Many steps in the cell engineering process with lack of support or safety assessments before regulatory review |
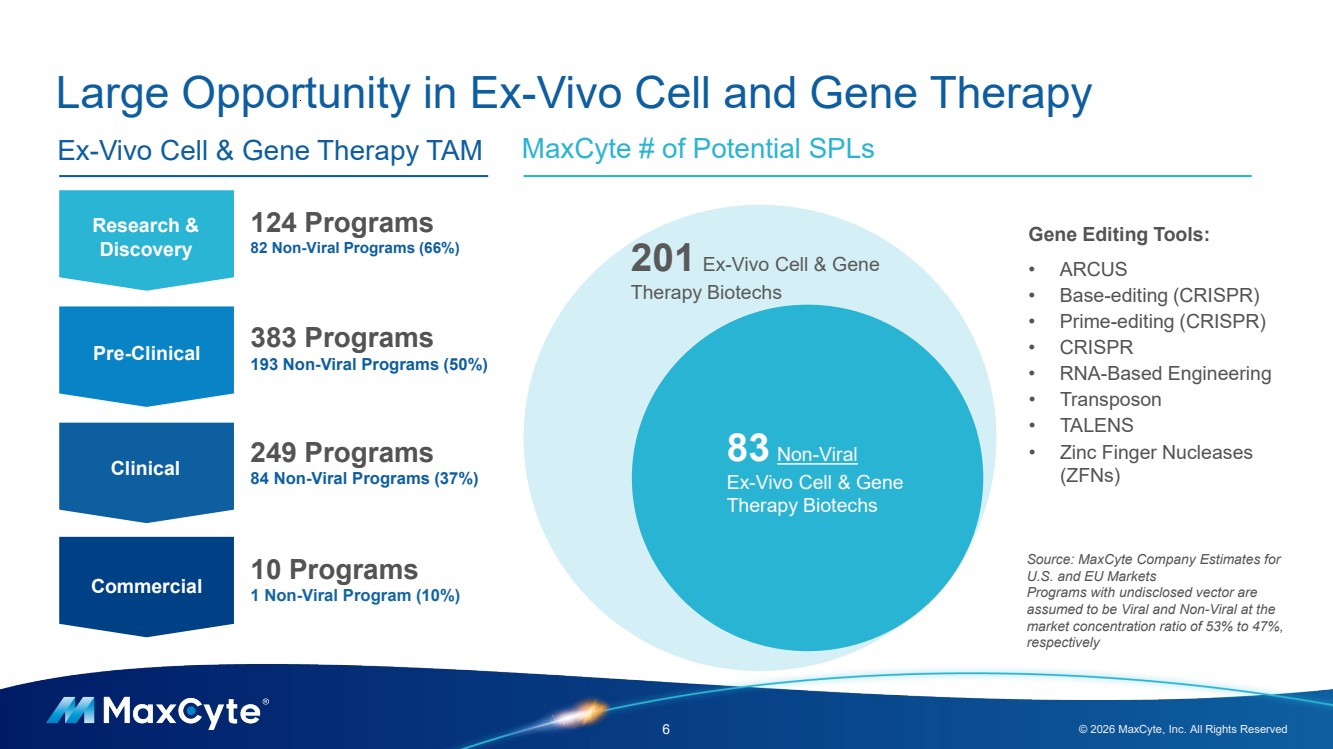
| Large Opportunity in Ex-Vivo Cell and Gene Therapy 6 © 2026 MaxCyte, Inc. All Rights Reserved MaxCyte # of Potential SPLs Research & Discovery Pre-Clinical Clinical Commercial 124 Programs 82 Non-Viral Programs (66%) 383 Programs 193 Non-Viral Programs (50%) 249 Programs 84 Non-Viral Programs (37%) 10 Programs 1 Non-Viral Program (10%) Ex-Vivo Cell & Gene Therapy TAM 201 Ex-Vivo Cell & Gene Therapy Biotechs 83 Non-Viral Ex-Vivo Cell & Gene Therapy Biotechs Gene Editing Tools: • ARCUS • Base-editing (CRISPR) • Prime-editing (CRISPR) • CRISPR • RNA-Based Engineering • Transposon • TALENS • Zinc Finger Nucleases (ZFNs) Source: MaxCyte Company Estimates for U.S. and EU Markets Programs with undisclosed vector are assumed to be Viral and Non-Viral at the market concentration ratio of 53% to 47%, respectively |
| The Expert Platform Enabling Non-Viral Cell Engineering 7 © 2026 MaxCyte, Inc. All Rights Reserved Full scale RUO Small/mid-scale RUO Large Scale RUO/cGMP Full scale RUO/cGMP 75 thousand to 700 million cells 75 thousand to 20 billion cells 5 billion to 200 billion cells 75 thousand to 20 billion cells High Performance • >90% transfection efficiencies (depending on cell type and molecule) • >90% cell viabilities • Computer-controlled system for reproducible results Flexibility • Single, fully-defined, animal component-free electroporation buffer for all cell types • Pre-loaded library of validated, cell-specific protocols Scalability – Ability to Transfect • 75,000 to 7 million cells in seconds • Up to 20 billion cells in less than 30 minutes • And up to 200 billion cells in less than 30 minutes with the high scale VLx High Quality • Sterile, single-use processing assemblies (PAs) • Closed, cGMP-compliant, ISO-certified, and CE marked instruments • Supported by US FDA Master File and global equivalents Key Applications: Ex-Vivo Engineered Cell Therapies Customer Base: Leading global cell therapy developers and academic translational centers Additional Electroporation Applications in Drug Discovery Viral Vector Production – Transfect adherent or suspension cells to produce a variety of viral vectors. Cell Based Assays – Produce assay-ready cells faster with scalable electroporation. Gene Editing – Navigate the complexities of genome engineering with highly efficient delivery. Antibody & Protein Production – Accelerate biotherapeutic development with transient expression for gram-scale protein production. Vaccine Development – Innovate vaccine research with our adaptable platform for production of recombinant proteins, virus-like particles and more. Research & discovery 96-well platform RUO 100 thousand to 10 million cells Launched Q1-26 |
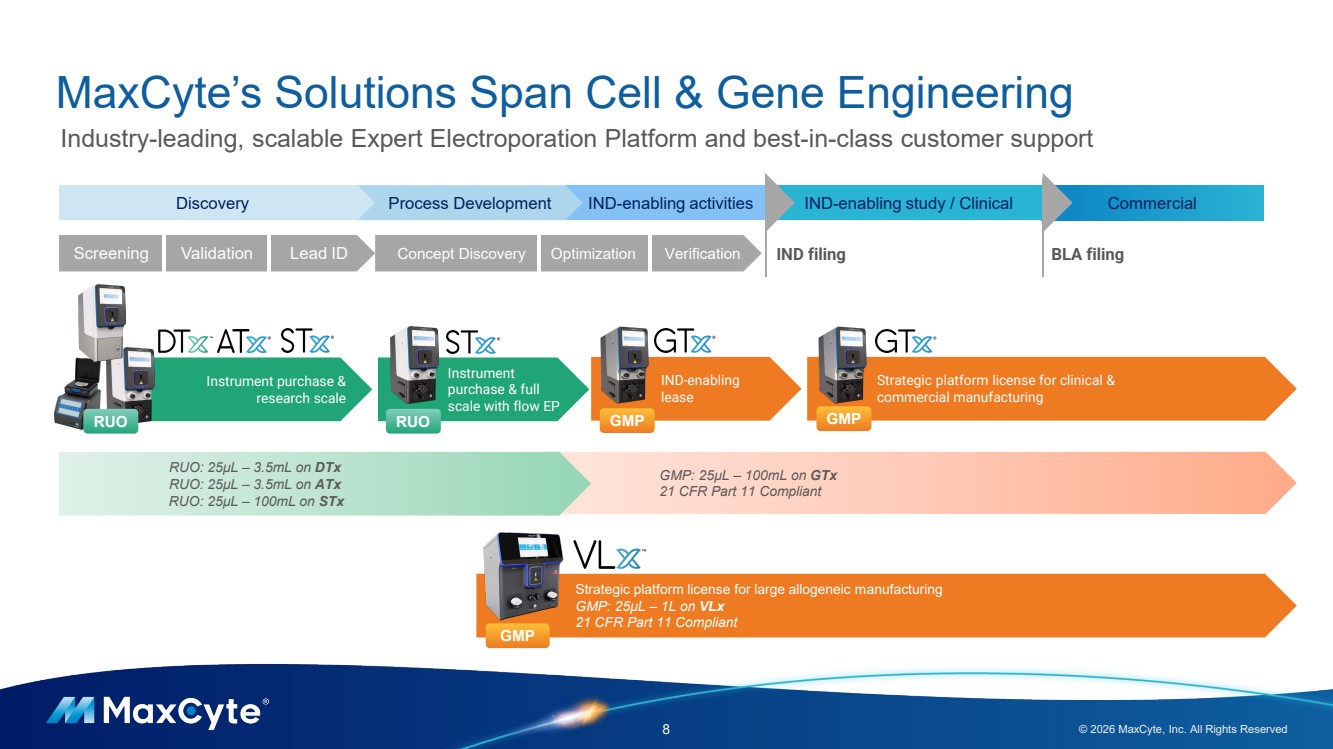
| Instrument purchase & research scale GMP: 25µL – 100mL on GTx 21 CFR Part 11 Compliant MaxCyte’s Solutions Span Cell & Gene Engineering 8 © 2026 MaxCyte, Inc. All Rights Reserved Industry-leading, scalable Expert Electroporation Platform and best-in-class customer support Commercial Strategic platform license for clinical & commercial manufacturing IND-enabling lease Discovery Process Development IND-enabling activities IND-enabling study / Clinical IND filing BLA filing Instrument purchase & full scale with flow EP RUO RUO GMP GMP Screening Validation Lead ID Concept Discovery Optimization Verification RUO: 25µL – 3.5mL on DTx RUO: 25µL – 3.5mL on ATx RUO: 25µL – 100mL on STx Strategic platform license for large allogeneic manufacturing GMP: 25µL – 1L on VLx 21 CFR Part 11 Compliant GMP |
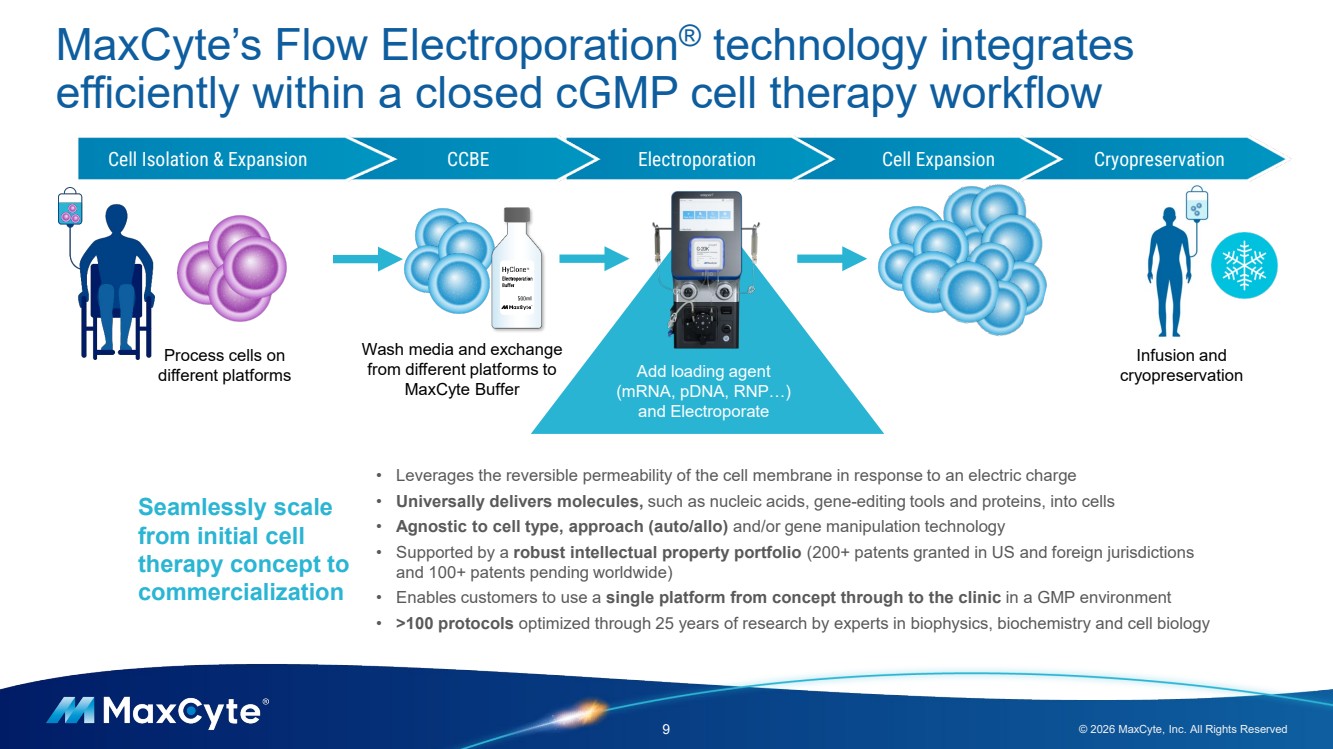
| MaxCyte’s Flow Electroporation® technology integrates efficiently within a closed cGMP cell therapy workflow 9 © 2026 MaxCyte, Inc. All Rights Reserved Cell Isolation & Expansion CCBE Electroporation Cell Expansion Cryopreservation Add loading agent (mRNA, pDNA, RNP…) and Electroporate Process cells on different platforms Wash media and exchange from different platforms to MaxCyte Buffer Infusion and cryopreservation Seamlessly scale from initial cell therapy concept to commercialization • Leverages the reversible permeability of the cell membrane in response to an electric charge • Universally delivers molecules, such as nucleic acids, gene-editing tools and proteins, into cells • Agnostic to cell type, approach (auto/allo) and/or gene manipulation technology • Supported by a robust intellectual property portfolio (200+ patents granted in US and foreign jurisdictions and 100+ patents pending worldwide) • Enables customers to use a single platform from concept through to the clinic in a GMP environment • >100 protocols optimized through 25 years of research by experts in biophysics, biochemistry and cell biology |
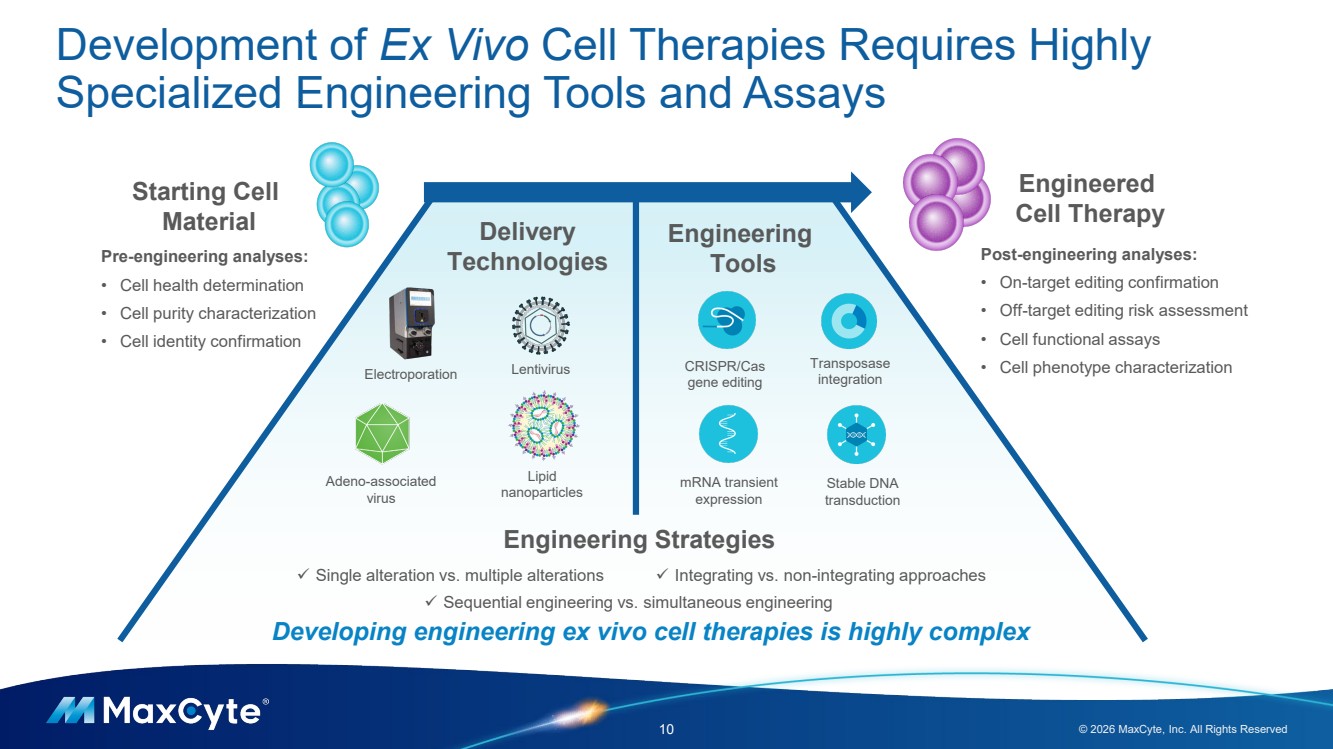
| Development of Ex Vivo Cell Therapies Requires Highly Specialized Engineering Tools and Assays 10 © 2026 MaxCyte, Inc. All Rights Reserved Lentivirus Adeno-associated virus Lipid nanoparticles Electroporation Engineering Tools Delivery Technologies CRISPR/Cas gene editing Transposase integration mRNA transient expression Stable DNA transduction Engineered Cell Therapy Starting Cell Material Post-engineering analyses: • On-target editing confirmation • Off-target editing risk assessment • Cell functional assays • Cell phenotype characterization Engineering Strategies Pre-engineering analyses: • Cell health determination • Cell purity characterization • Cell identity confirmation Single alteration vs. multiple alterations Sequential engineering vs. simultaneous engineering Integrating vs. non-integrating approaches Developing engineering ex vivo cell therapies is highly complex |
| MaxCyte’s Solutions are Uniquely Positioned to Support Cell Therapy Development 11 © 2026 MaxCyte, Inc. All Rights Reserved Flow Electroporation technology facilitates multiplex and sequential engineering without the payload and capacity limitations of viral approaches 23+1 Field Application Scientists support our customers in their development process Expert platform provides industry leading transfection efficiency & cell viability at high scale in 30 minutes or less, enabling manufacturers to quickly scale up production Regulatory Support Superior Results Complex Engineering Optimization Scientific Support FDA Master File can be referenced in regulatory filings to accelerate and de-risk regulatory review MaxCyte technology allows plug and play processes with rapid optimization delivering reproducible outcomes and the ability to seamlessly scale up from pre-IND to the clinic and commercialization 1. As of December 31, 2025 |
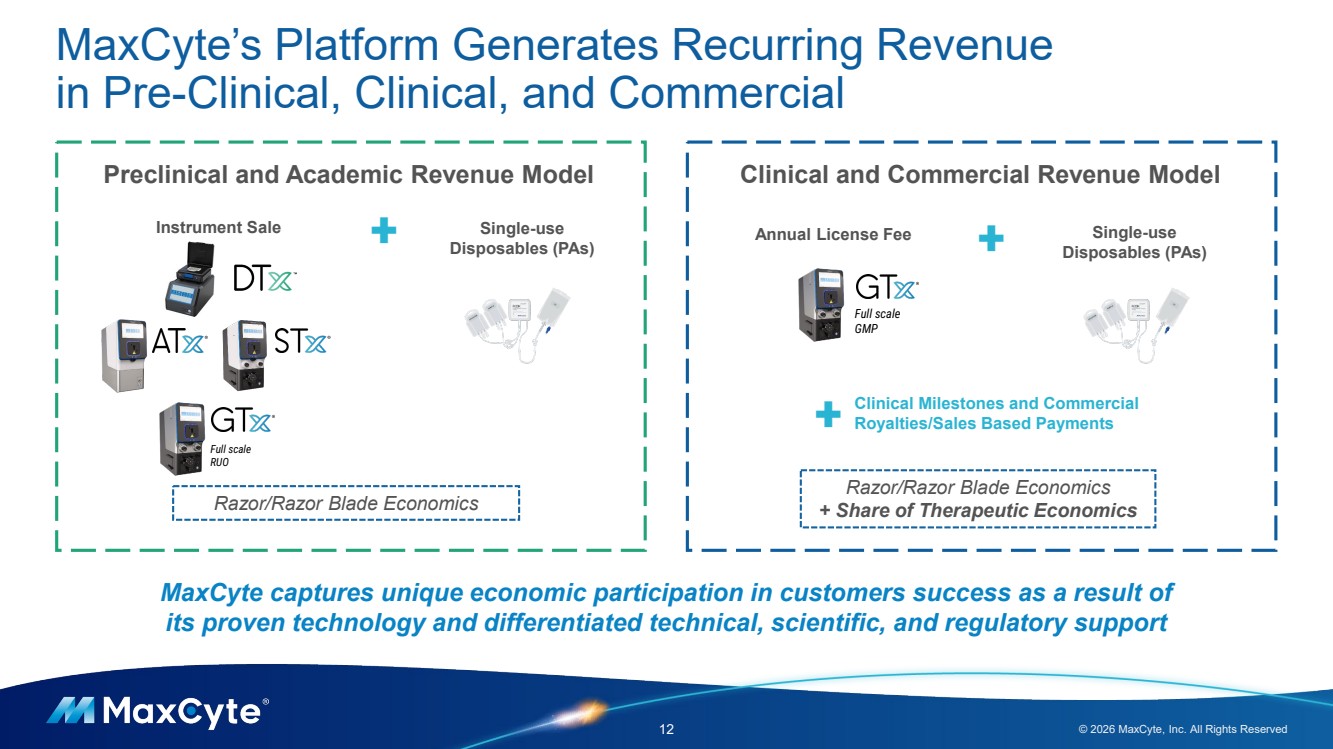
| MaxCyte’s Platform Generates Recurring Revenue in Pre-Clinical, Clinical, and Commercial 12 © 2026 MaxCyte, Inc. All Rights Reserved Razor/Razor Blade Economics Full scale RUO Preclinical and Academic Revenue Model Instrument Sale Clinical and Commercial Revenue Model Annual License Fee Razor/Razor Blade Economics + Share of Therapeutic Economics Clinical Milestones and Commercial Royalties/Sales Based Payments Single-use Disposables (PAs) Full scale GMP Single-use Disposables (PAs) + + + MaxCyte captures unique economic participation in customers success as a result of its proven technology and differentiated technical, scientific, and regulatory support |
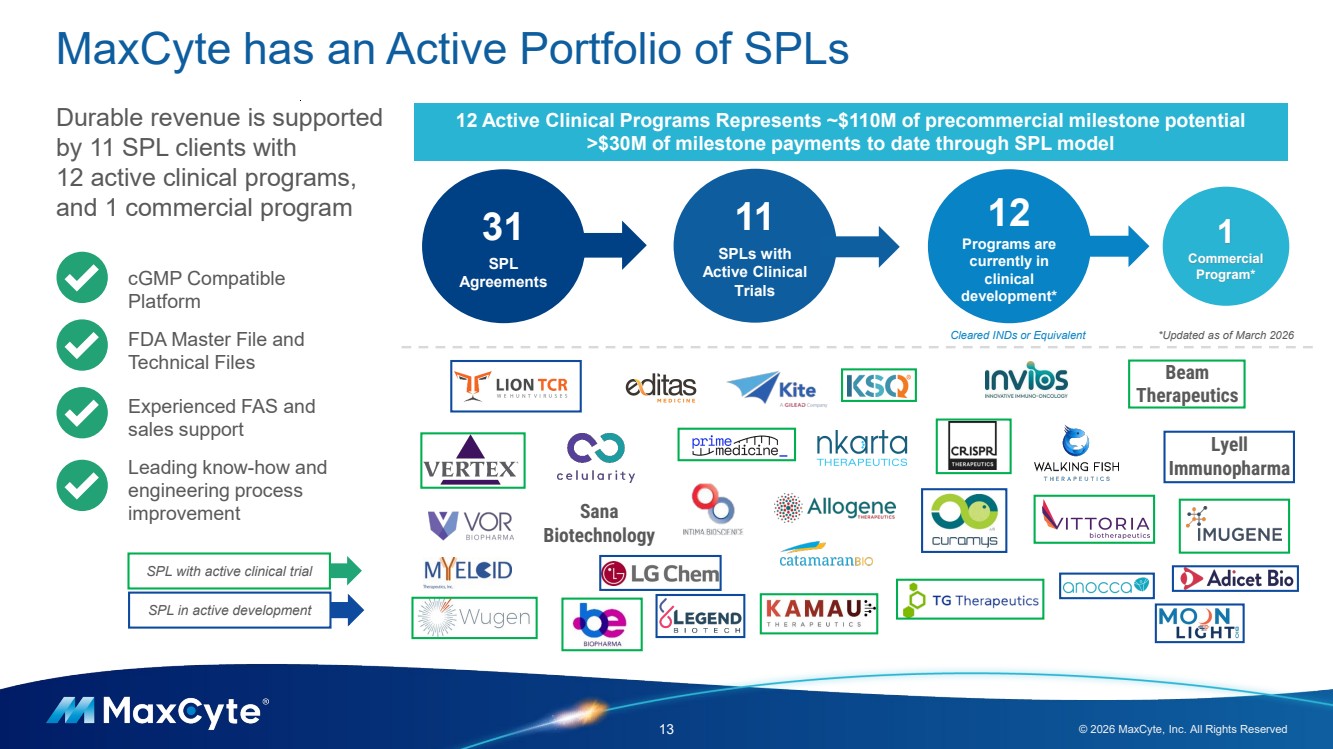
| Lyell Immunopharma MaxCyte has an Active Portfolio of SPLs 13 © 2026 MaxCyte, Inc. All Rights Reserved 1 Commercial Program* 31 SPL Agreements 12 Programs are currently in clinical development* Cleared INDs or Equivalent *Updated as of March 2026 Durable revenue is supported by 11 SPL clients with 12 active clinical programs, and 1 commercial program Sana Biotechnology Beam Therapeutics cGMP Compatible Platform FDA Master File and Technical Files Experienced FAS and sales support Leading know-how and engineering process improvement 12 Active Clinical Programs Represents ~$110M of precommercial milestone potential >$30M of milestone payments to date through SPL model 11 SPLs with Active Clinical Trials SPL with active clinical trial SPL in active development |
| Typical MaxCyte SPL Economics 14 © 2026 MaxCyte, Inc. All Rights Reserved Early Clinical: (Phase 1/2) Years 1-3 • Mid 6-figure up to low 7-figure milestones • 1-3+ instruments + disposables Mid-late Clinical: (Phase 2/3) Years 3-5+ • 7-figure milestone • Increasing instrument and disposables usage Approval: Year 5+ • Multiple 7-figure milestones based on geographic region (e.g. US and Europe) Commercial Phase • Royalty and/or sales-based payments • Annual instrument fees and disposable sales Instruments and Processing Assemblies Milestones Royalties & Sales-Based Payments Pre-commercial milestones in early clinical, mid-late clinical and product approval Significant development milestones and high-value participation in future commercial success of partners Recurring revenues from lease of instruments and sales of single-use disposables Royalites and Sales-based payments upon partner’s product commercialization |
| Differentiated Commercial Relationships Expand Sales Funnel 15 © 2026 MaxCyte, Inc. All Rights Reserved Highly Technical Employees Field Application Scientists (FAS) and Commercial Team Unparalleled scientific support to customers Customer relationships at early stages of cell & gene therapy development Signed SPL *Updated as of December 31, 2025 MaxCyte has a team of 23+ highly trained FAS* Global teams providing scientific, technical, and regulatory expertise FAS works with prospective customers to optimize and implement cell engineering methods, processes, and applications MaxCyte grows its sales funnel by leading with scientific, technical, and regulatory expertise 50 Advanced Degrees and 23 PhDs* Support academic and translational institutions, biotech companies, and pharma companies in discovery and pre-clinical |
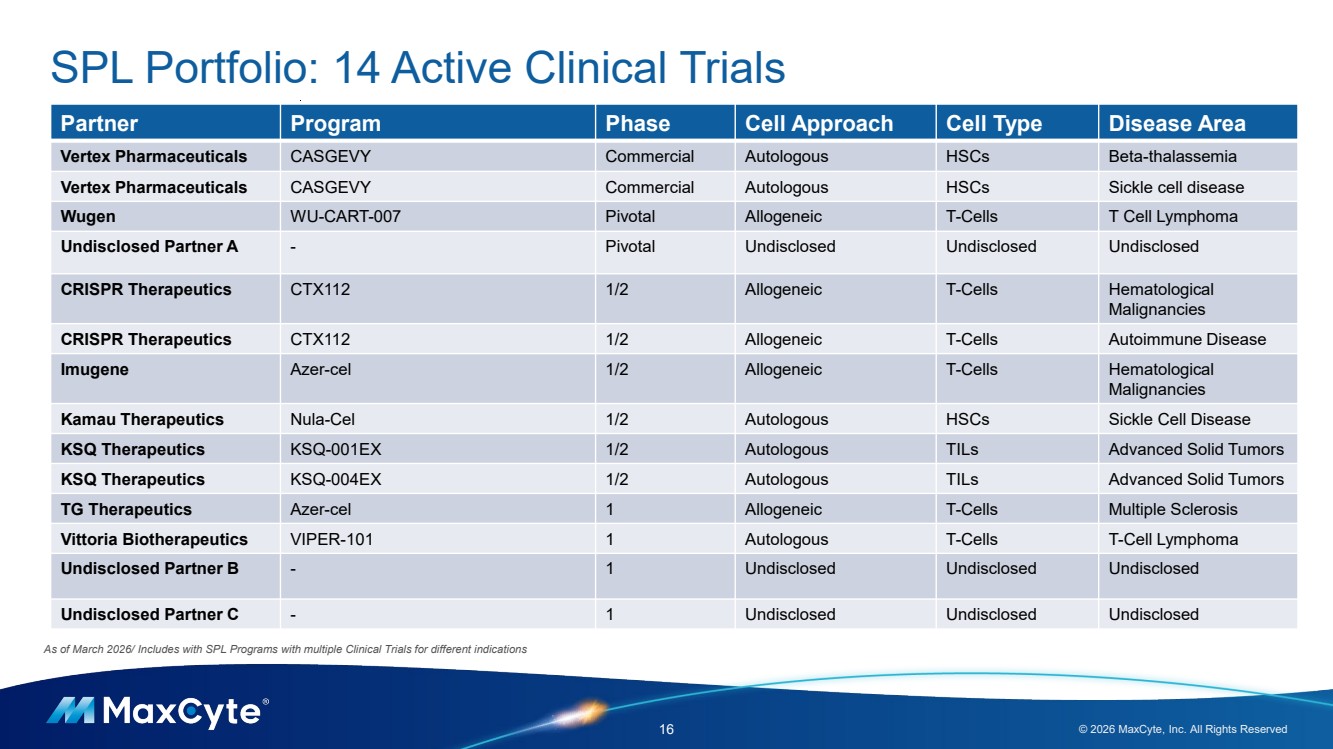
| 16 © 2026 MaxCyte, Inc. All Rights Reserved As of March 2026/ Includes with SPL Programs with multiple Clinical Trials for different indications SPL Portfolio: 14 Active Clinical Trials Partner Program Phase Cell Approach Cell Type Disease Area Vertex Pharmaceuticals CASGEVY Commercial Autologous HSCs Beta-thalassemia Vertex Pharmaceuticals CASGEVY Commercial Autologous HSCs Sickle cell disease Wugen WU-CART-007 Pivotal Allogeneic T-Cells T Cell Lymphoma Undisclosed Partner A - Pivotal Undisclosed Undisclosed Undisclosed CRISPR Therapeutics CTX112 1/2 Allogeneic T-Cells Hematological Malignancies CRISPR Therapeutics CTX112 1/2 Allogeneic T-Cells Autoimmune Disease Imugene Azer-cel 1/2 Allogeneic T-Cells Hematological Malignancies Kamau Therapeutics Nula-Cel 1/2 Autologous HSCs Sickle Cell Disease KSQ Therapeutics KSQ-001EX 1/2 Autologous TILs Advanced Solid Tumors KSQ Therapeutics KSQ-004EX 1/2 Autologous TILs Advanced Solid Tumors TG Therapeutics Azer-cel 1 Allogeneic T-Cells Multiple Sclerosis Vittoria Biotherapeutics VIPER-101 1 Autologous T-Cells T-Cell Lymphoma Undisclosed Partner B - 1 Undisclosed Undisclosed Undisclosed Undisclosed Partner C - 1 Undisclosed Undisclosed Undisclosed |
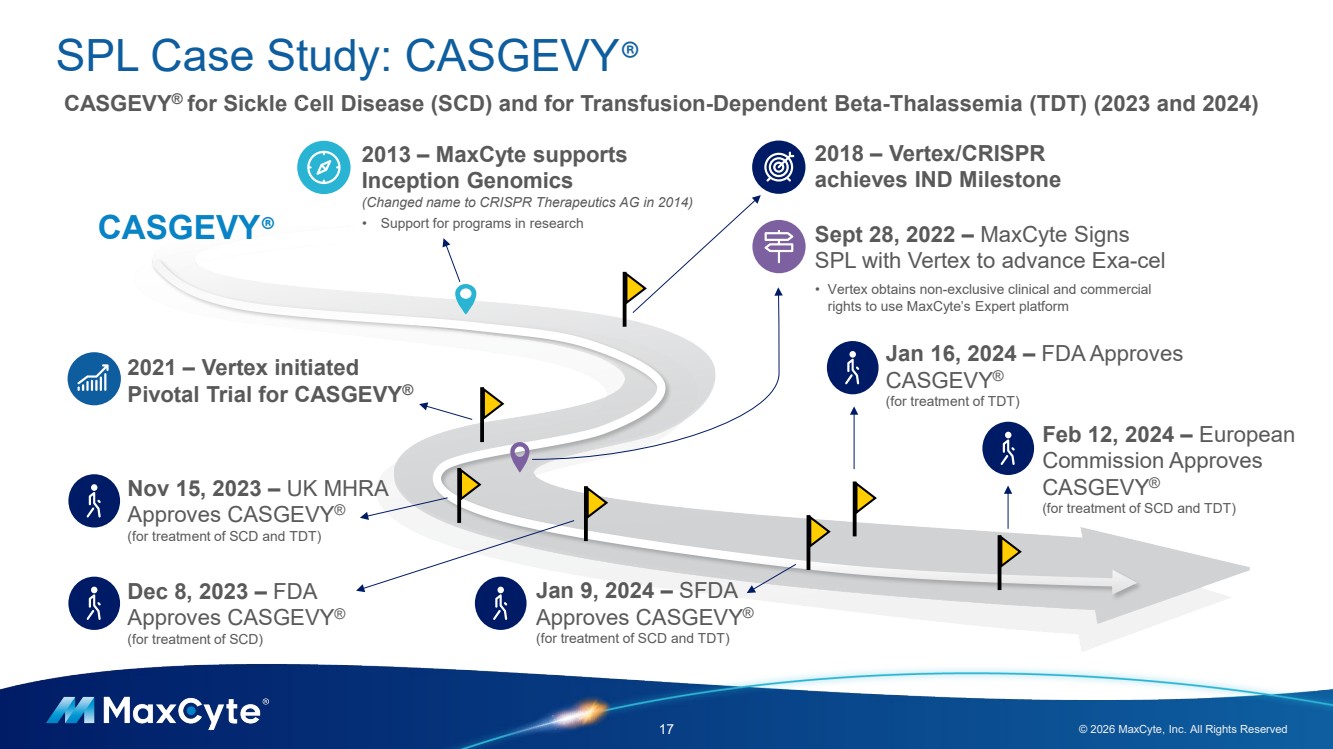
| SPL Case Study: CASGEVY® 17 © 2026 MaxCyte, Inc. All Rights Reserved CASGEVY® for Sickle Cell Disease (SCD) and for Transfusion-Dependent Beta-Thalassemia (TDT) (2023 and 2024) 2013 – MaxCyte supports Inception Genomics (Changed name to CRISPR Therapeutics AG in 2014) • Support for programs in research 2018 – Vertex/CRISPR achieves IND Milestone Sept 28, 2022 – MaxCyte Signs SPL with Vertex to advance Exa-cel • Vertex obtains non-exclusive clinical and commercial rights to use MaxCyte’s Expert platform Dec 8, 2023 – FDA Approves CASGEVY® (for treatment of SCD) 2021 – Vertex initiated Pivotal Trial for CASGEVY® Jan 16, 2024 – FDA Approves CASGEVY® (for treatment of TDT) Feb 12, 2024 – European Commission Approves CASGEVY® (for treatment of SCD and TDT) Nov 15, 2023 – UK MHRA Approves CASGEVY® (for treatment of SCD and TDT) CASGEVY® Jan 9, 2024 – SFDA Approves CASGEVY® (for treatment of SCD and TDT) |
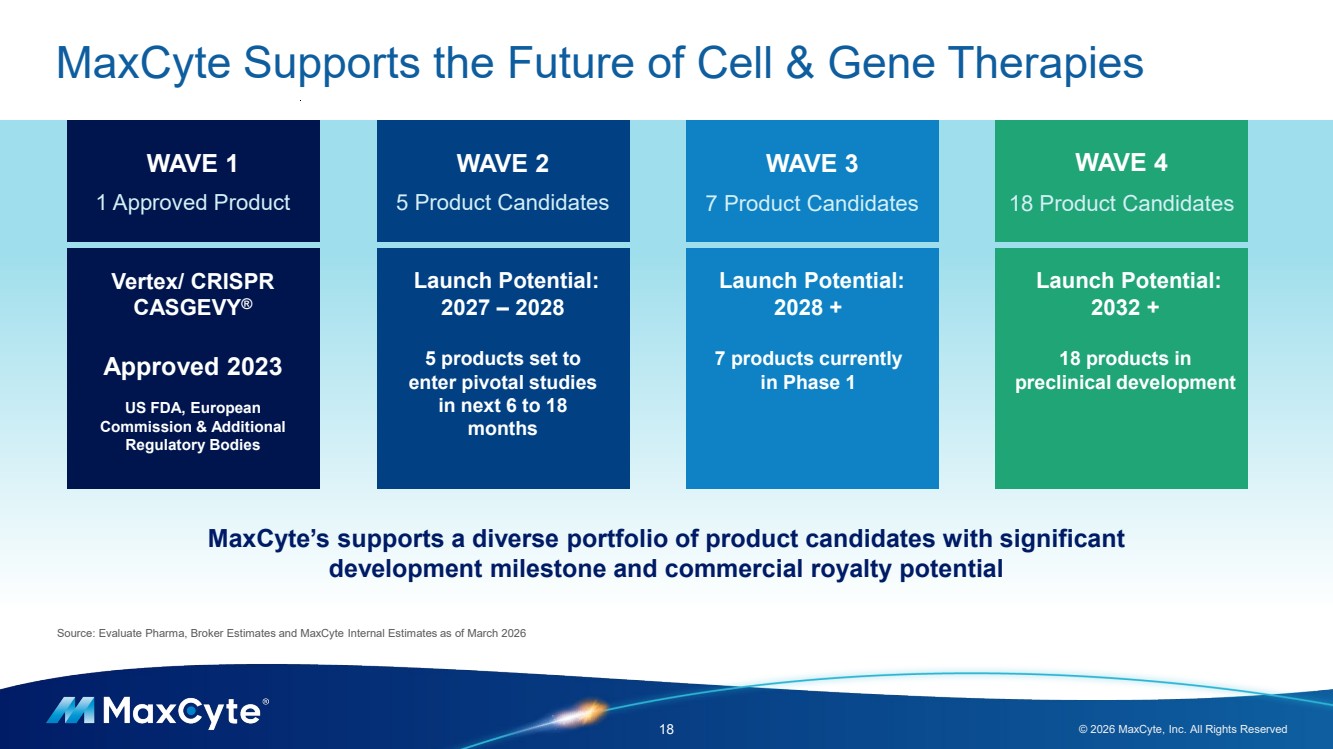
| MaxCyte Supports the Future of Cell & Gene Therapies 18 © 2026 MaxCyte, Inc. All Rights Reserved WAVE 1 1 Approved Product WAVE 2 5 Product Candidates WAVE 3 7 Product Candidates WAVE 4 18 Product Candidates Vertex/ CRISPR CASGEVY® Approved 2023 US FDA, European Commission & Additional Regulatory Bodies Launch Potential: 2027 – 2028 5 products set to enter pivotal studies in next 6 to 18 months Launch Potential: 2028 + 7 products currently in Phase 1 Launch Potential: 2032 + 18 products in preclinical development Source: Evaluate Pharma, Broker Estimates and MaxCyte Internal Estimates as of March 2026 MaxCyte’s supports a diverse portfolio of product candidates with significant development milestone and commercial royalty potential |
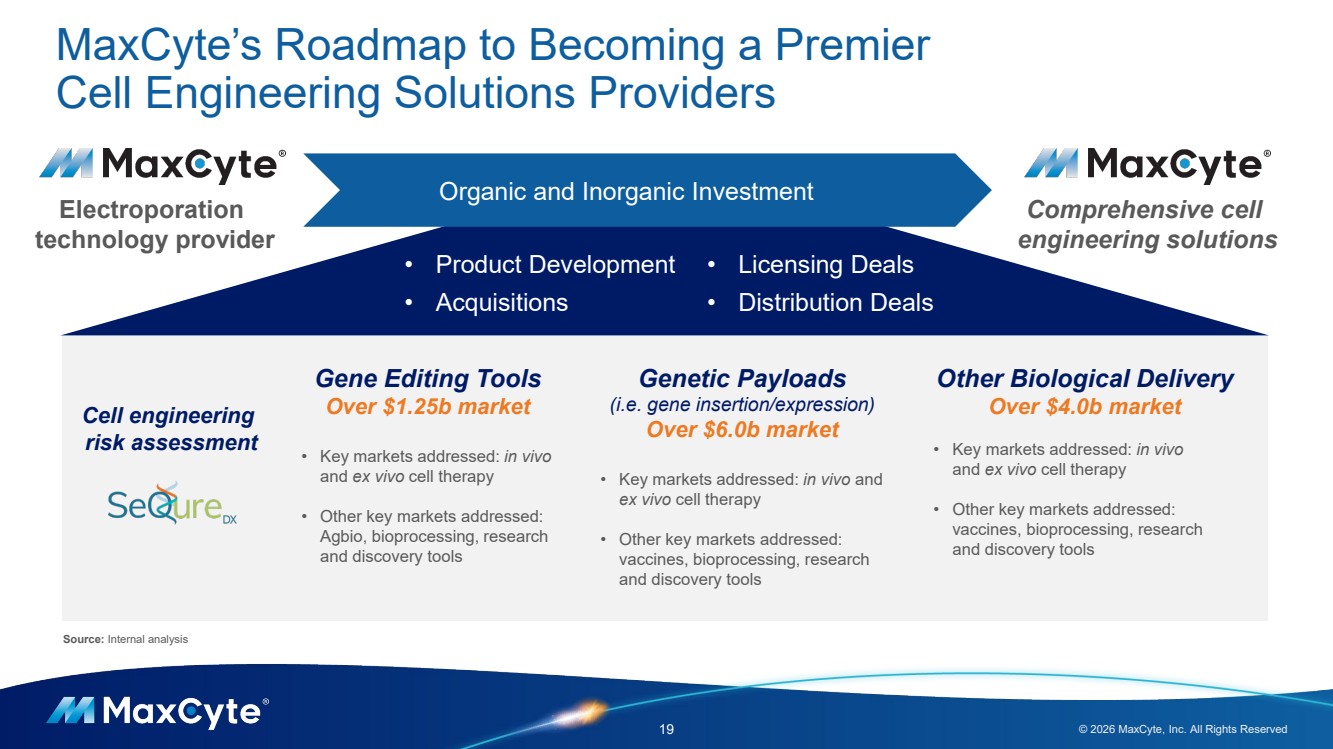
| MaxCyte’s Roadmap to Becoming a Premier Cell Engineering Solutions Providers 19 © 2026 MaxCyte, Inc. All Rights Reserved Electroporation technology provider Comprehensive cell engineering solutions Gene Editing Tools Over $1.25b market • Key markets addressed: in vivo and ex vivo cell therapy • Other key markets addressed: Agbio, bioprocessing, research and discovery tools Genetic Payloads (i.e. gene insertion/expression) Over $6.0b market • Key markets addressed: in vivo and ex vivo cell therapy • Other key markets addressed: vaccines, bioprocessing, research and discovery tools Other Biological Delivery Over $4.0b market • Key markets addressed: in vivo and ex vivo cell therapy • Other key markets addressed: vaccines, bioprocessing, research and discovery tools Cell engineering risk assessment Organic and Inorganic Investment • Product Development • Acquisitions • Licensing Deals • Distribution Deals Source: Internal analysis |
| Editing Assessment Services Strategy 20 © 2026 MaxCyte, Inc. All Rights Reserved MaxCyte provides ex-vivo and in-vivo developers with best-in-class on-target and off-target risk assessment services Use Cases: candidate guide screening, guide RNA selection Use Cases: Off-target risk assessment, guide RNA selection, IND filings Highly sensitive Universal for all editors Population-scale variant assessment Multiple orthogonal methods Variant effect prediction GLP-grade Guide Screening Rapid turnaround time Comprehensive report Minimize risk from variation at on-target locus Guide RNA Selection Low-cost per guide Evaluate multiple guides Reduce program risk through early profiling and selection Use Cases: Cellular Editing Assessment, IND filings Sensitive detection methods Relevant cell types On- and off-target analysis Biologic impact assessment GLP-grade Increases program likelihood of success Decreases risk of unexpected costs or program delays Aligns with most recent FDA guidance for gene edited therapies Quicker time to clinic and safer therapies Discovery Pre-Clinical Development and IND-Enabling Studies BENEFITS |
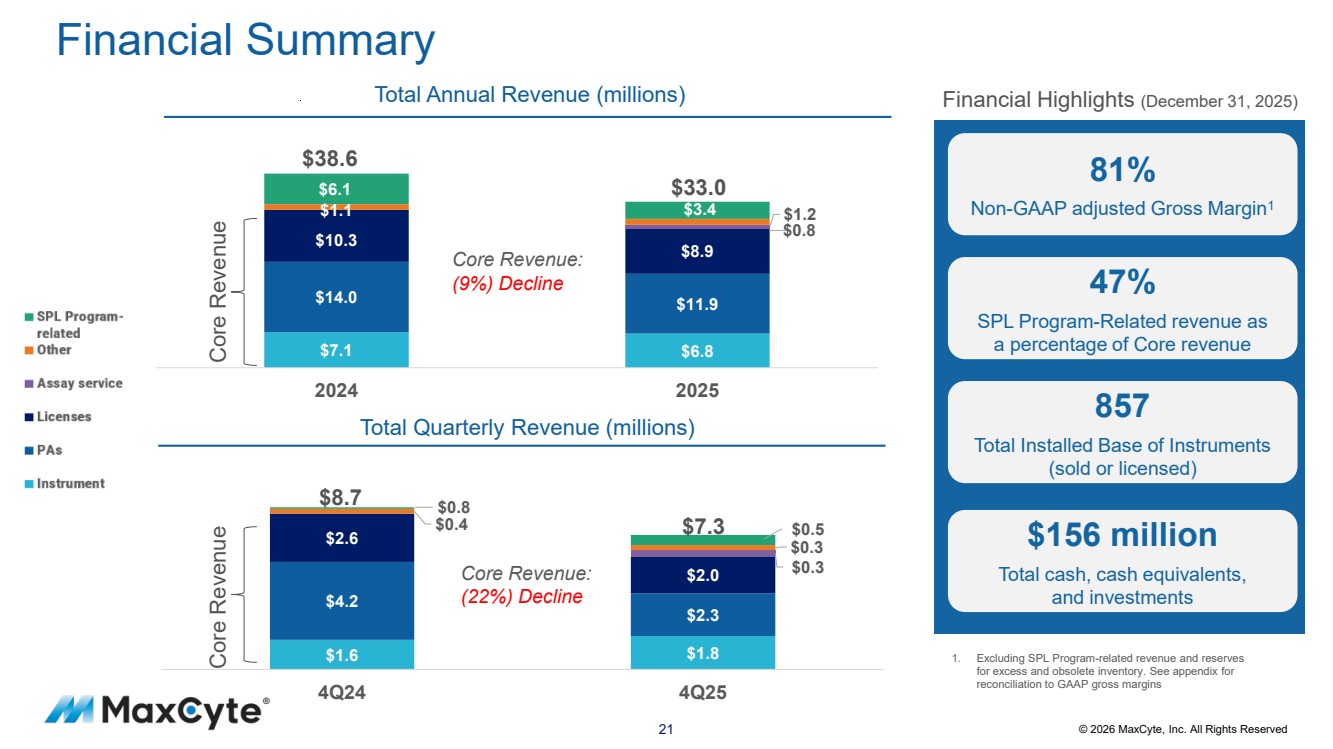
| 21 © 2026 MaxCyte, Inc. All Rights Reserved Financial Summary $1.6 $1.8 $4.2 $2.3 $2.6 $2.0 $0.3 $0.4 $0.3 4Q24 4Q25 Core Revenue: (22%) Decline $7.1 $6.8 $14.0 $11.9 $10.3 $8.9 $1.1 $1.2 $6.1 $3.4 2024 2025 Core Revenue: (9%) Decline 1. Excluding SPL Program-related revenue and reserves for excess and obsolete inventory. See appendix for reconciliation to GAAP gross margins Financial Highlights (December 31, 2025) 81% Non-GAAP adjusted Gross Margin1 47% SPL Program-Related revenue as a percentage of Core revenue 857 Total Installed Base of Instruments (sold or licensed) $156 million Total cash, cash equivalents, and investments Total Annual Revenue (millions) Core Revenue Total Quarterly Revenue (millions) Core Revenue $38.6 $33.0 $8.7 $7.3 $0.5 $0.8 $0.8 |
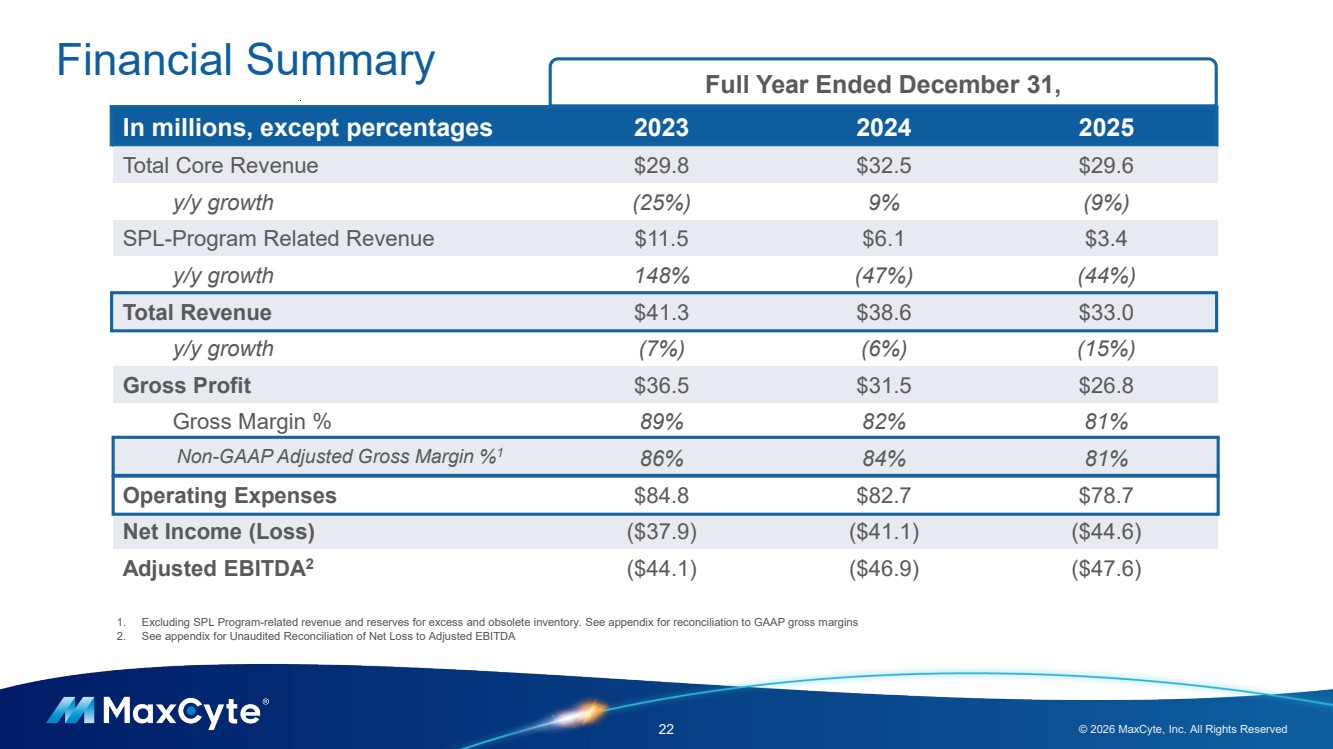
| Financial Summary 22 © 2026 MaxCyte, Inc. All Rights Reserved In millions, except percentages 2023 2024 2025 Total Core Revenue $29.8 $32.5 $29.6 y/y growth (25%) 9% (9%) SPL-Program Related Revenue $11.5 $6.1 $3.4 y/y growth 148% (47%) (44%) Total Revenue $41.3 $38.6 $33.0 y/y growth (7%) (6%) (15%) Gross Profit $36.5 $31.5 $26.8 Gross Margin % 89% 82% 81% Non-GAAP Adjusted Gross Margin %1 86% 84% 81% Operating Expenses $84.8 $82.7 $78.7 Net Income (Loss) ($37.9) ($41.1) ($44.6) Adjusted EBITDA2 ($44.1) ($46.9) ($47.6) Full Year Ended December 31, 1. Excluding SPL Program-related revenue and reserves for excess and obsolete inventory. See appendix for reconciliation to GAAP gross margins 2. See appendix for Unaudited Reconciliation of Net Loss to Adjusted EBITDA |
| Disciplined Management is Committed to Growth Investment and Efficient Spending 23 © 2026 MaxCyte, Inc. All Rights Reserved MaxCyte is well capitalized and funded to achieve profitability with existing capital Alignment of spending and resources to growth areas Organic investment in new products and product enhancements Reduction of annual cash burn excluding one-time and non-cash items Realize operating leverage on existing cost base Inorganic investment to solve critical pain points in Cell & Gene Therapy Healthy balance sheet ~$156M of cash, cash equivalents, and investments1 1. As of December 31, 2025 |
| All other trademarks are the property of their respective owners. ©2025 MaxCyte, Inc. All rights reserved. For a complete list of MaxCyte trademarks in the United States and other countries, please visit maxcyte.com/trademarks. Thank you! Any questions? ir@maxcyte.com 24 © 2026 MaxCyte, Inc. All Rights Reserved |
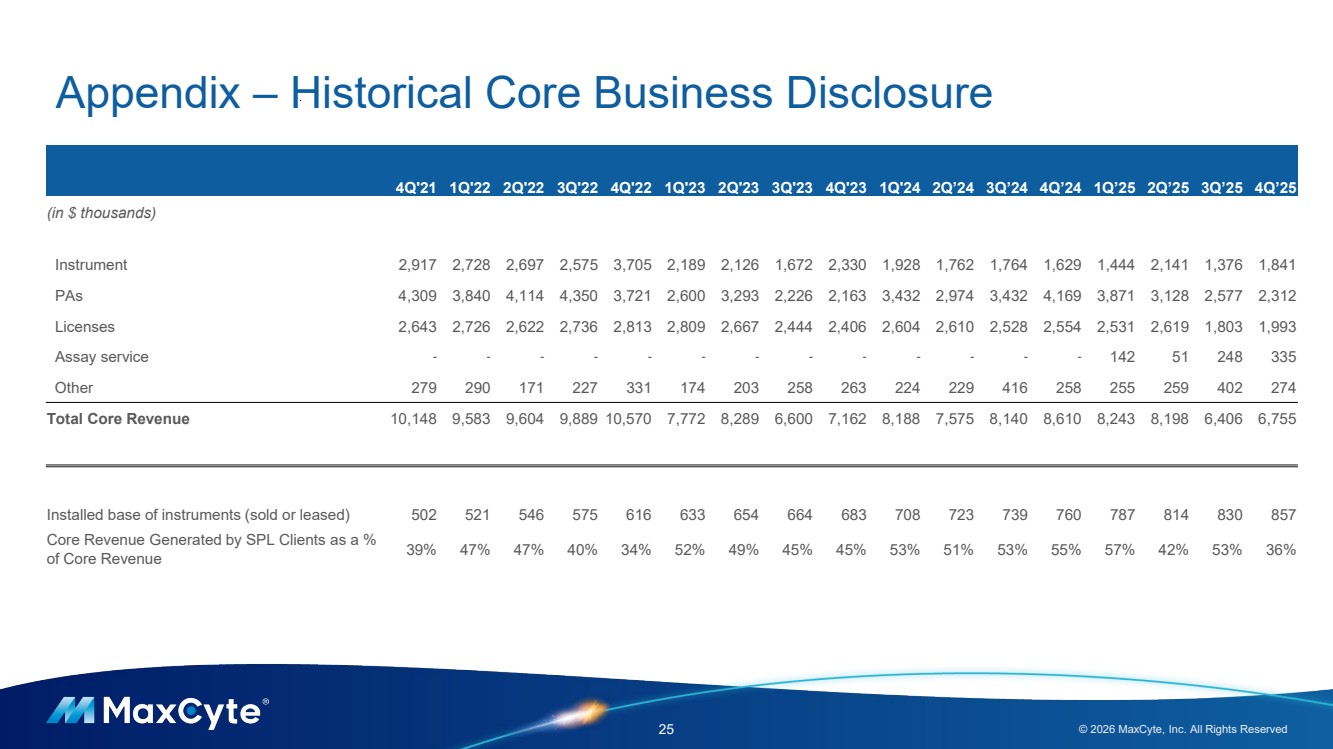
| Appendix – Historical Core Business Disclosure 25 © 2026 MaxCyte, Inc. All Rights Reserved 4Q'21 1Q'22 2Q'22 3Q'22 4Q'22 1Q'23 2Q'23 3Q'23 4Q'23 1Q'24 2Q’24 3Q’24 4Q’24 1Q’25 2Q’25 3Q’25 4Q’25 (in $ thousands) Instrument 2,917 2,728 2,697 2,575 3,705 2,189 2,126 1,672 2,330 1,928 1,762 1,764 1,629 1,444 2,141 1,376 1,841 PAs 4,309 3,840 4,114 4,350 3,721 2,600 3,293 2,226 2,163 3,432 2,974 3,432 4,169 3,871 3,128 2,577 2,312 Licenses 2,643 2,726 2,622 2,736 2,813 2,809 2,667 2,444 2,406 2,604 2,610 2,528 2,554 2,531 2,619 1,803 1,993 Assay service - - - - - - - - - - - - - 142 51 248 335 Other 279 290 171 227 331 174 203 258 263 224 229 416 258 255 259 402 274 Total Core Revenue 10,148 9,583 9,604 9,889 10,570 7,772 8,289 6,600 7,162 8,188 7,575 8,140 8,610 8,243 8,198 6,406 6,755 Installed base of instruments (sold or leased) 502 521 546 575 616 633 654 664 683 708 723 739 760 787 814 830 857 Core Revenue Generated by SPL Clients as a % of Core Revenue 39% 47% 47% 40% 34% 52% 49% 45% 45% 53% 51% 53% 55% 57% 42% 53% 36% |
| Appendix – Unaudited Reconciliation of Gross Margin to Non-GAAP Adjusted gross margin 26 © 2026 MaxCyte, Inc. All Rights Reserved 1. Adjustments include the exclusion of SPL program related revenue from Revenue, and the exclusion of reserves for excess and obsolete inventory from Cost of Goods Sold. Unaudited Reconciliation of Gross Margin to Non-GAAP Adjusted gross margin (in thousands, except for percentages) (Unaudited) Three months ended December 31, 2025 Twelve months ended December 31, 2025 GAAP Adjustments Non-GAAP GAAP Adjustments Non-GAAP Revenue $ 7,300 $ (546) $ 6,754 $ 8,693 $ (83) $ 8,610 Cost of Goods Sold 1,610 (151) 1,459 2,281 (916) 1,365 Gross Margin 5,690 395 5,295 6,412 833 7,245 Gross Margin % 78% 78% 74% 84% Year ended December 31, 2025 Year ended December 31, 2024 GAAP Adjustments Non-GAAP GAAP Adjustments Non-GAAP Revenue $ 33,026 $ (3,423) $ 29,603 $ 38,627 $ (6,115) $ 32,512 Cost of Goods Sold 6,222 (676) 5,546 7,100 (1,771) 5,329 Gross Margin 26,804 (2,747) 24,057 31,527 (4,344) 27,183 Gross Margin % 81% 81% 82% 84% |
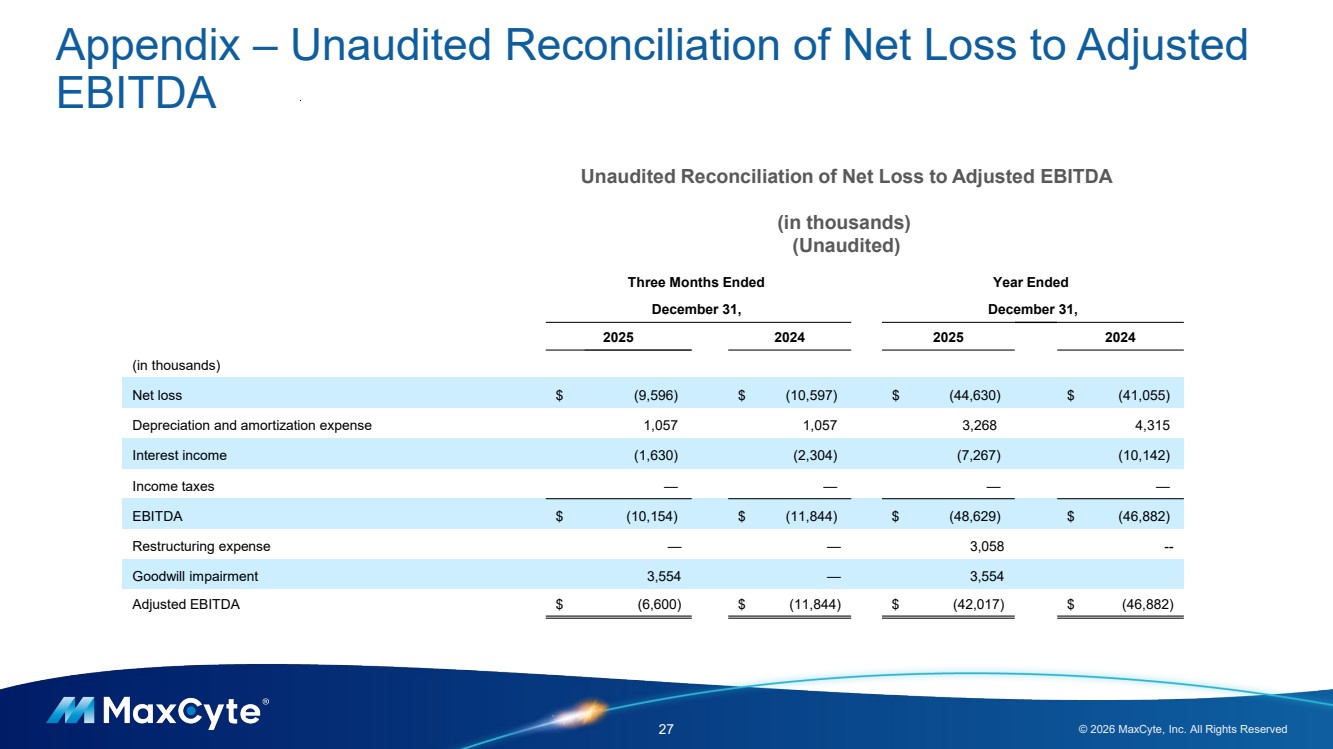
| Appendix – Unaudited Reconciliation of Net Loss to Adjusted EBITDA 27 © 2026 MaxCyte, Inc. All Rights Reserved Unaudited Reconciliation of Net Loss to Adjusted EBITDA (in thousands) (Unaudited) Three Months Ended Year Ended December 31, December 31, 2025 2024 2025 2024 (in thousands) Net loss $ (9,596) $ (10,597) $ (44,630) $ (41,055) Depreciation and amortization expense 1,057 1,057 3,268 4,315 Interest income (1,630) (2,304) (7,267) (10,142) Income taxes — — — — EBITDA $ (10,154) $ (11,844) $ (48,629) $ (46,882) Restructuring expense — — 3,058 -- Goodwill impairment 3,554 — 3,554 Adjusted EBITDA $ (6,600) $ (11,844) $ (42,017) $ (46,882) |