
.2

Interim Data Presentation from the ADVANCED - 2 Trial of TARA - 002 in Patients with NMIBC American Society of Clinical Oncology (ASCO) Genitourinary (GU) Cancers Symposium February 2026

2 Statements contained in this presentation regarding matters that are not historical facts are "forward looking statements" within the meaning of the Private Securities Litigation Reform Act of 1995. Protara may, in some cases, use terms such as “predicts,” “believes,” “potential,” “proposed,” “continue,” “designed,” “estimates,” “anticipates,” “expects,” “plans,” “intends,” “may,” “could,” “might,” “will,” “should” or other words or expressions referencing future events, conditions or circumstances that convey uncertainty of future events or outcomes to identify these forward - looking statements. Such forward - looking statements include but are not limited to, statements regarding Protara’s intentions, beliefs, projections, outlook, analyses or current expectations concerning, among other things: Protara’s business strategy, including its development plans for its product candidates and plans regarding the timing or outcome of existing or future clinical trials (including the timing of any particular phases of such trials and the timing of the announcement of any data produced during such trials or phases thereof); statements related to expectations regarding interactions with the U.S. Food and Drug Administration (FDA); Protara’s financial position; statements regarding the anticipated safety or efficacy of Protara’s product candidates; and Protara’s outlook for the remainder of the year and future periods. Because such statements are subject to risks and uncertainties, actual results may differ materially from those expressed or implied by such forward - looking statements. Factors that contribute to the uncertain nature of the forward - looking statements include: risks that Protara’s financial guidance may not be as expected, as well as risks and uncertainties associated with: Protara’s development programs, including the initiation and completion of non - clinical studies and clinical trials and the timing of required filings with the FDA and other regulatory agencies; general market conditions; changes in the competitive landscape; changes in Protara’s strategic and commercial plans; Protara’s ability to obtain sufficient financing to fund its strategic plans and commercialization efforts; having to use cash in ways or on timing other than expected; the impact of market volatility on cash reserves; failure to attract and retain management and key personnel; the impact of general U.S. and foreign, economic, industry, market, regulatory, political or public health conditions; and the risks and uncertainties associated with Protara’s business and financial condition in general, including the risks and uncertainties described more fully under the caption “Risk Factors” and elsewhere in Protara's filings and reports with the United States Securities and Exchange Commission. All forward - looking statements contained in this presentation speak only as of the date on which they were made and are based on management's assumptions and estimates as of such date. Protara undertakes no obligation to update any forward - looking statements, whether as a result of the receipt of new information, the occurrence of future events or otherwise, except as required by law. © 2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute. | Forward Looking Statements

Jesse Shefferman Co - founder, Director, Chief Executive Officer Neal Shore, MD Medical Director, START Carolinas/Carolina Urologic Research Center Introduction Jesse Shefferman Review of Updated TARA - 002 Data Carla Beckham, MD, PhD KOL Discussion Neal Shore, MD Closing Remarks Jesse Shefferman ǪGA All 3 Carla Beckham, MD , PhD Lead Medical Director and Head of Clinical Development of TARA - 002 in NMIBC and Board - Certified Urologist Agenda © 2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute. |

Key Clinical Updates TARA - 002 in NMIBC • No additional administration procedures or safety protocols required • Rapid administration typically performed by nurse 3. Next steps • Expect to complete enrollment in the BCG - Unresponsive registrational cohort of ADVANCED - 2 in 2H’26 • Expect to initiate enrollment in the BCG - Naive NMIBC ADVANCED - 3 registrational trial in 2H’26 TARA - 002 for Non - Muscle Invasive Bladder Cancer (NMIBC) 1. Interim data from the ADVANCED - 2 trial of TARA - 002 in patients with NMIBC • TARA - 002 demonstrated 66% CR rate at any time, 68% 6 - month landmark and 33% 12 - month landmark CR rate in patients with BCG - Unresponsive NMIBC 1 • TARA - 002 demonstrated 72% CR rate at any time, 67% 6 - month landmark and 58% 12 - month landmark CR rate in patients with BCG - Naïve NMIBC 1 • Favorable safety and tolerability profile observed with no Grade 3 or greater TRAEs 2. TARA - 002 has anticipated low burden on physicians G patients 1 Referencing data from poster presented at ASCO GU symposium with data cutoff of January 28, 2026 Abbreviation: CR = Complete Response © 2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute. | 4
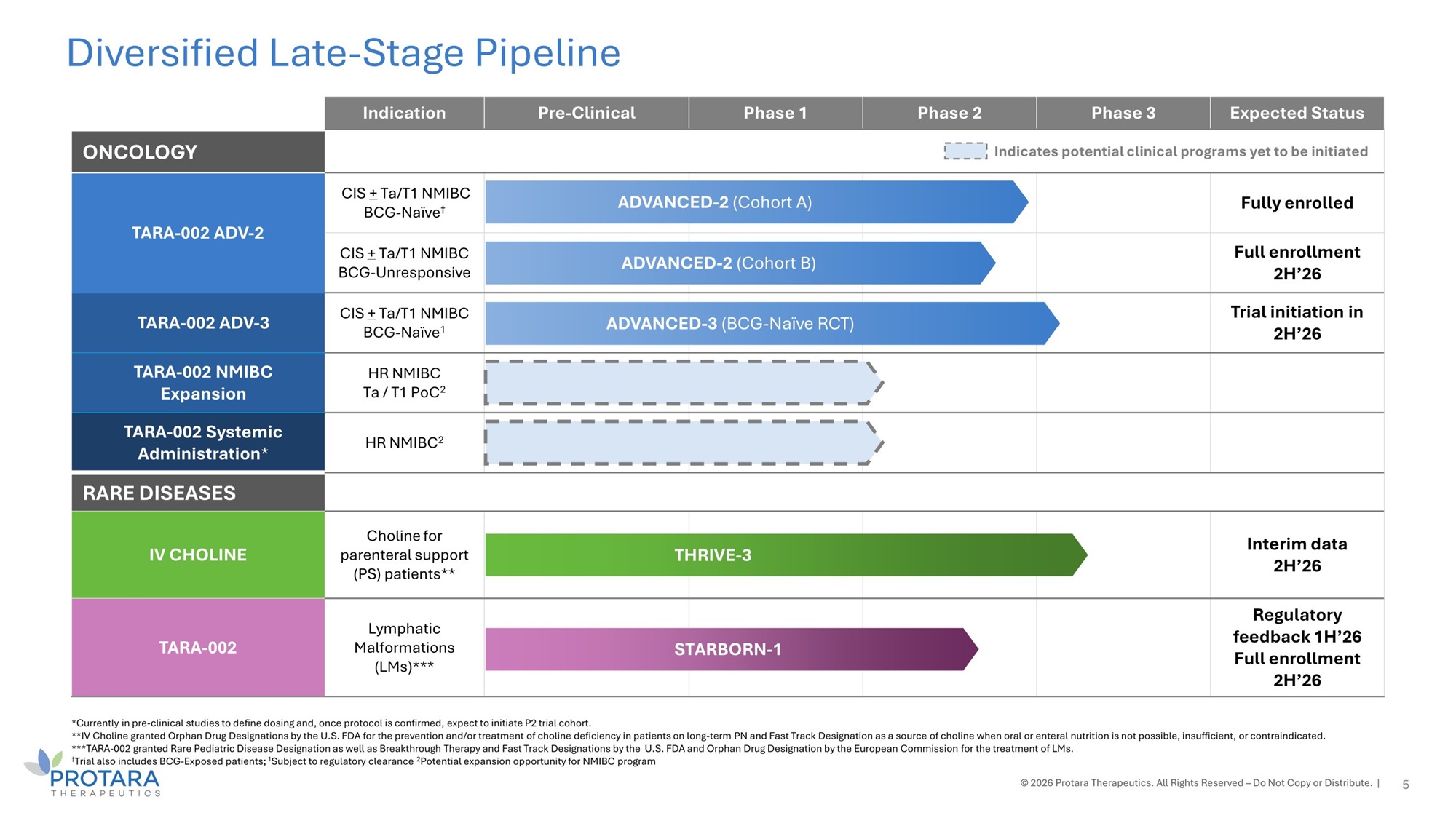
*Currently in pre - clinical studies to define dosing and, once protocol is confirmed, expect to initiate P2 trial cohort. **IV Choline granted Orphan Drug Designations by the U.S. FDA for the prevention and/or treatment of choline deficiency in patients on long - term PN and Fast Track Designation as a source of choline when oral or enteral nutrition is not possible, insufficient, or contraindicated. ***TARA - 002 granted Rare Pediatric Disease Designation as well as Breakthrough Therapy and Fast Track Designations by the U.S. FDA and Orphan Drug Designation by the European Commission for the treatment of LMs. † Trial also includes BCG - Exposed patients; 1 Subject to regulatory clearance 2 Potential expansion opportunity for NMIBC program © 2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute. | Diversified Late - Stage Pipeline Expected Status Phase 3 Phase 2 Phase 1 Pre - Clinical Indication Indicates potential clinical programs yet to be initiated ONCOLOGY Fully enrolled ED - 2 (Cohort A) ADVANC CIS + Ta/T1 NMIBC BCG - Naïve † TARA - 002 ADV - 2 Full enrollment 2H’26 ED - 2 (Cohort B) ADVANC CIS + Ta/T1 NMIBC BCG - Unresponsive Trial initiation in 2H’26 - 3 (BCG - Naïve RCT) ADVANCED CIS + Ta/T1 NMIBC BCG - Naïve 1 TARA - 002 ADV - 3 HR NMIBC Ta / T1 PoC 2 TARA - 002 NMIBC Expansion HR NMIBC 2 TARA - 002 Systemic Administration * RARE DISEASES Interim data 2H’26 HRIVE - 3 T Choline for parenteral support (PS) patients** IV CHOLINE Regulatory feedback 1H’26 Full enrollment 2H’26 TARBORN - 1 S Lymphatic Malformations (LMs)*** TARA - 002 5
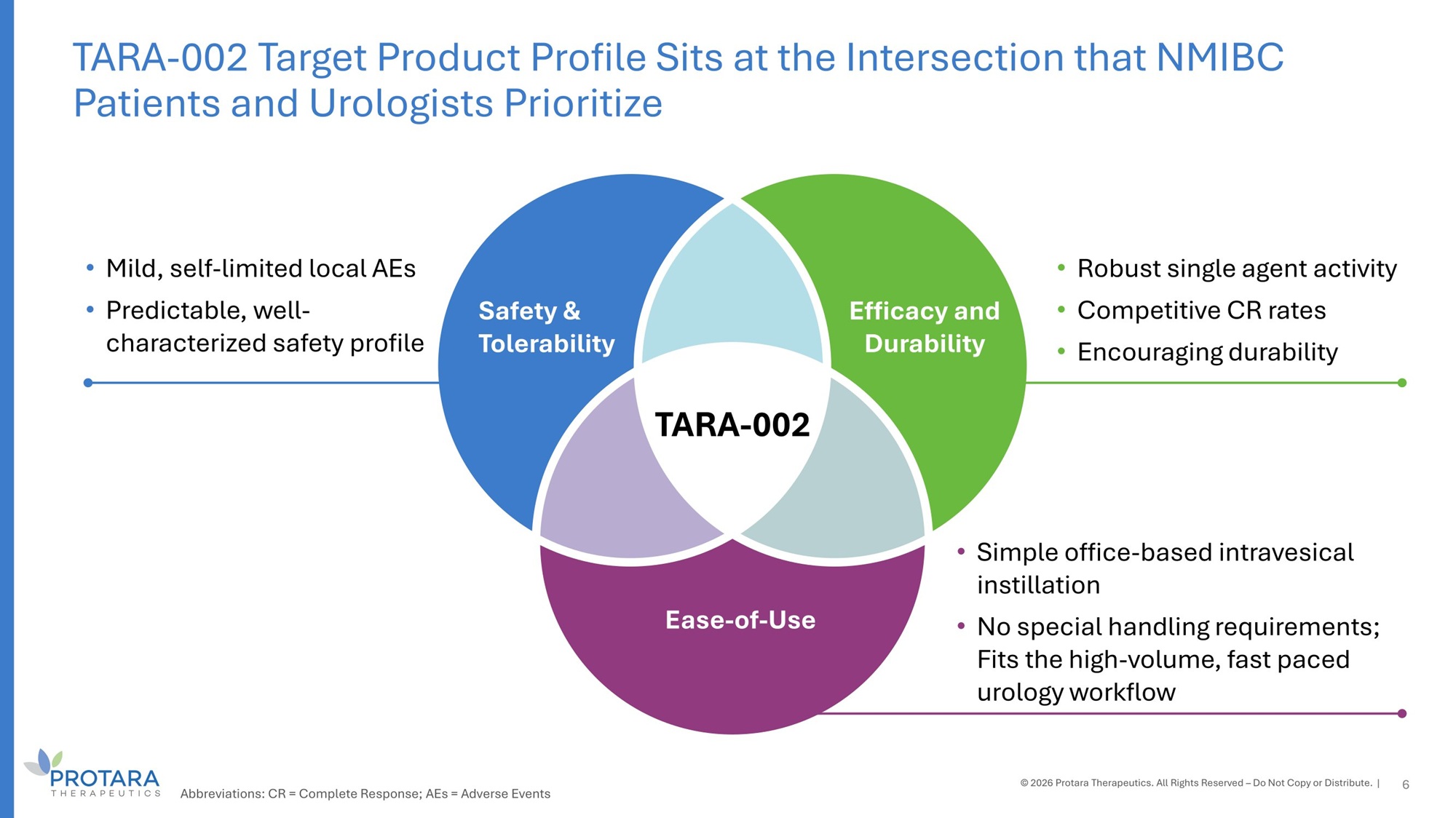
Safety G Tolerability Efficacy and Durability Ease - of - Use • Mild, self - limited local AEs • Predictable, well - characterized safety profile • Robust single agent activity • Competitive CR rates • Encouraging durability • Simple office - based intravesical instillation • No special handling requirements; Fits the high - volume, fast paced urology workflow TARA - 002 6 TARA - 002 Target Product Profile Sits at the Intersection that NMIBC Patients and Urologists Prioritize © 2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute. | Abbreviations: CR = Complete Response; AEs = Adverse Events

ADVANCED - 2 RESULTS
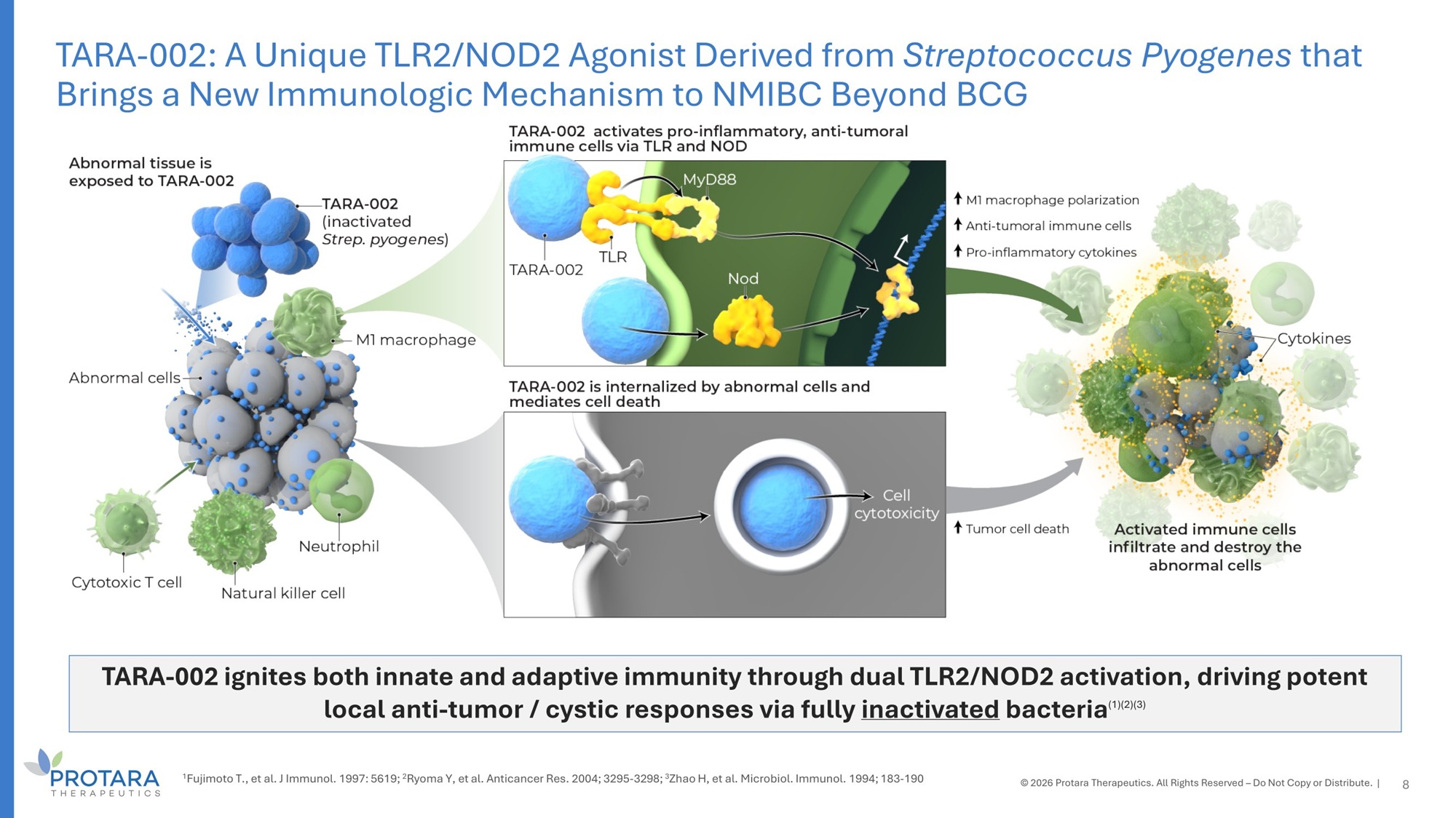
TARA - 002 ignites both innate and adaptive immunity through dual TLR2/NOD2 activation, driving potent local anti - tumor / cystic responses via fully inactivated bacteria (1)(2)(3) 8 1 Fujimoto T., et al. J Immunol. 1997: 5619; 2 Ryoma Y, et al. Anticancer Res. 2004; 3295 - 3298; 3 Zhao H, et al. Microbiol. Immunol. 1994; 183 - 190 TARA - 002: A Unique TLR2/NOD2 Agonist Derived from Streptococcus Pyogenes that Brings a New Immunologic Mechanism to NMIBC Beyond BCG © 2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute. |
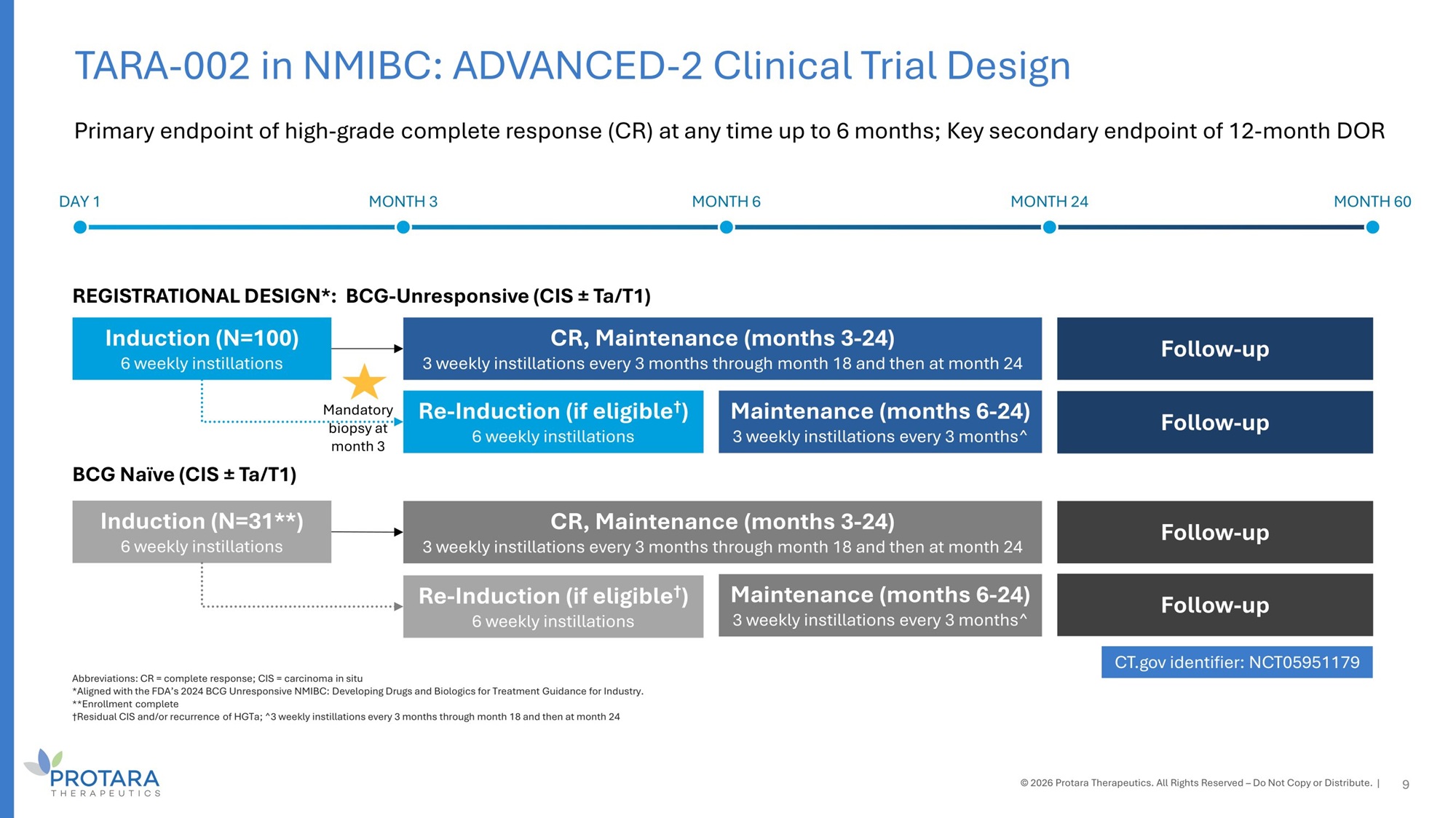
Abbreviations: CR = complete response; CIS = carcinoma in situ *Aligned with the FDA’s 2024 BCG Unresponsive NMIBC: Developing Drugs and Biologics for Treatment Guidance for Industry. **Enrollment complete †Residual CIS and/or recurrence of HGTa; ^3 weekly instillations every 3 months through month 18 and then at month 24 Primary endpoint of high - grade complete response (CR) at any time up to 6 months; Key secondary endpoint of 12 - month DOR 9 DAY 1 MONTH 3 MONTH 6 MONTH 24 MONTH 60 REGISTRATIONAL DESIGN*: BCG - Unresponsive (CIS “ Ta/T1) BCG Naïve (CIS “ Ta/T1) Induction (N=100) 6 weekly instillations Induction (N=31**) 6 weekly instillations CR, Maintenance (months 3 - 24) 3 weekly instillations every 3 months through month 18 and then at month 24 Follow - up Follow - up CR, Maintenance (months 3 - 24) 3 weekly instillations every 3 months through month 18 and then at month 24 Maintenance (months 6 - 24) 3 weekly instillations every 3 months^ Maintenance (months 6 - 24) 3 weekly instillations every 3 months^ Re - Induction (if eligible † ) 6 weekly instillations Re - Induction (if eligible † ) 6 weekly instillations Follow - up Follow - up CT.gov identifier: NCT05951179 © 2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute. | TARA - 002 in NMIBC: ADVANCED - 2 Clinical Trial Design Mandatory biopsy at month 3
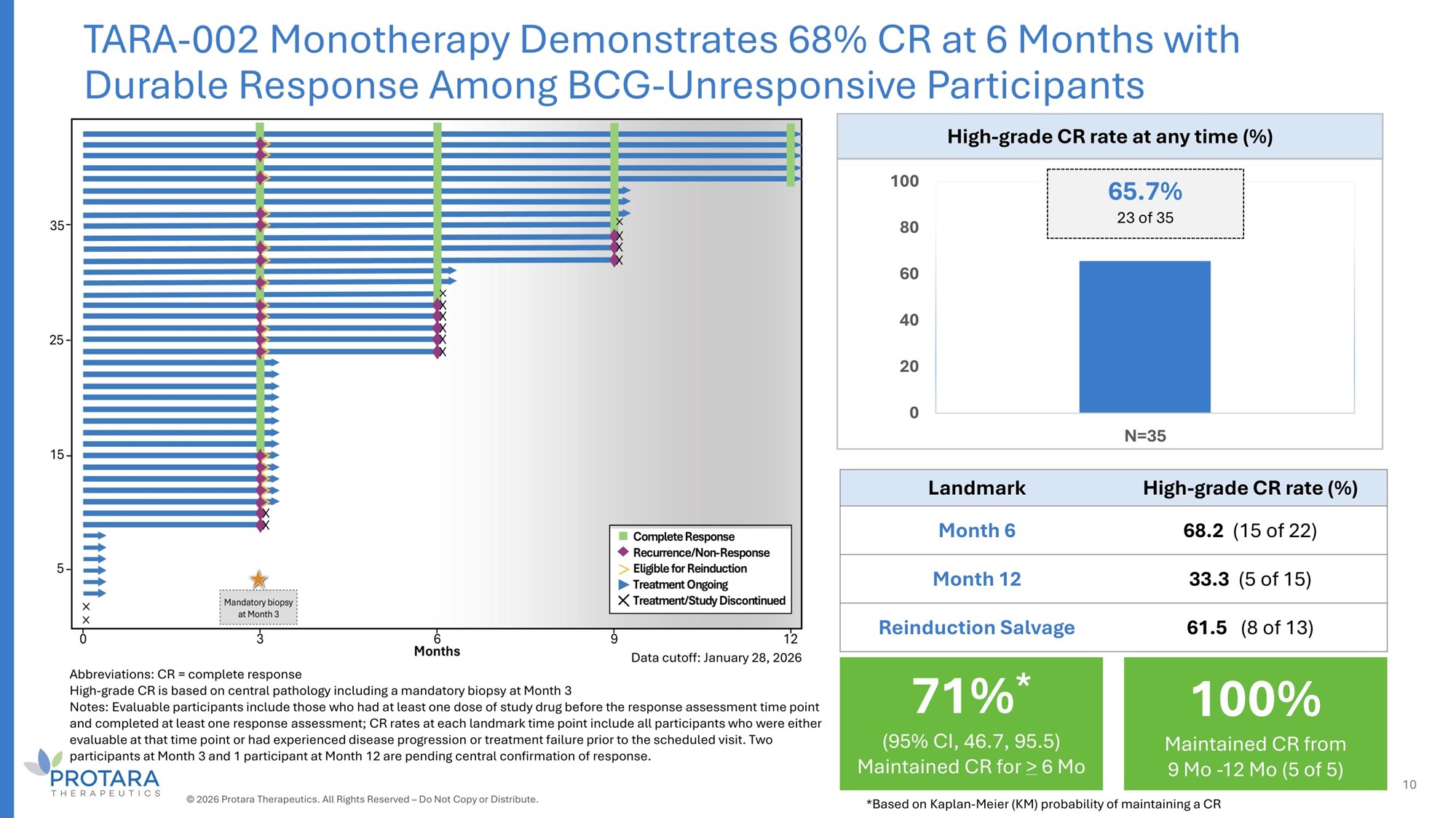
Abbreviations: CR = complete response High - grade CR is based on central pathology including a mandatory biopsy at Month 3 Notes: Evaluable participants include those who had at least one dose of study drug before the response assessment time point and completed at least one response assessment; CR rates at each landmark time point include all participants who were either evaluable at that time point or had experienced disease progression or treatment failure prior to the scheduled visit. Two participants at Month 3 and 1 participant at Month 12 are pending central confirmation of response. High - grade CR rate at any time (%) 65.7% 100 23 of 35 80 60 40 20 0 N=35 High - grade CR rate (%) Landmark 68.2 (15 of 22) Month 6 33.3 (5 of 15) Month 12 61.5 (8 of 13) Reinduction Salvage TARA - 002 Monotherapy Demonstrates 68% CR at 6 Months with Durable Response Among BCG - Unresponsive Participants Data cutoff: January 28, 2026 © 2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute. 10 100% Maintained CR from 9 Mo - 12 Mo (5 of 5) 71% * (95% CI, 46.7, 95.5) Maintained CR for > 6 Mo *Based on Kaplan - Meier (KM) probability of maintaining a CR
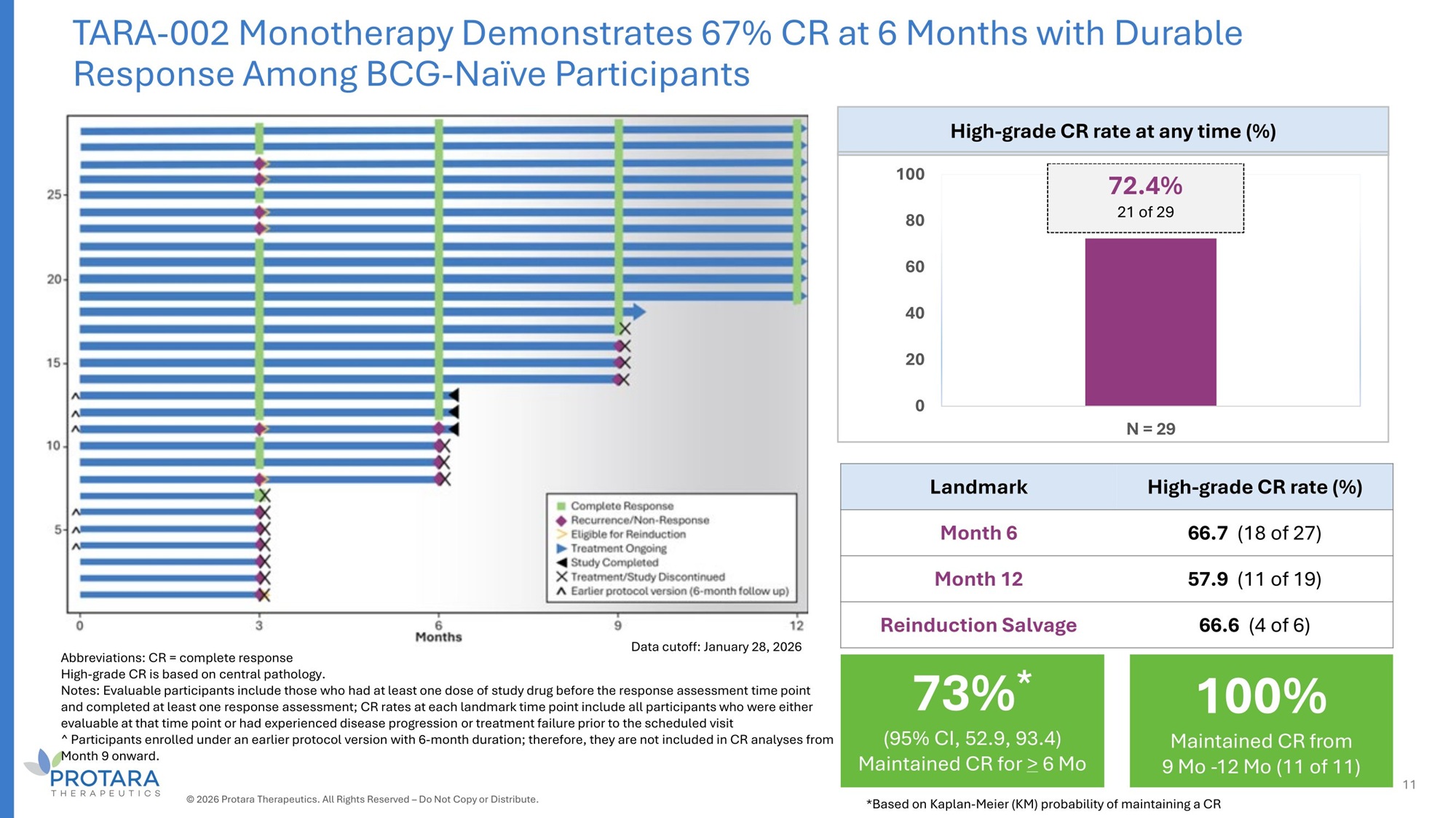
TARA - 002 Monotherapy Demonstrates 67% CR at 6 Months with Durable Response Among BCG - Naïve Participants High - grade CR rate at any time (%) 72.4% 100 21 of 29 80 60 40 20 0 N = 2G High - grade CR rate (%) Landmark 66.7 (18 of 27) Month 6 57.G (11 of 19) Month 12 66.6 (4 of 6) Reinduction Salvage Abbreviations: CR = complete response High - grade CR is based on central pathology. Notes: Evaluable participants include those who had at least one dose of study drug before the response assessment time point and completed at least one response assessment; CR rates at each landmark time point include all participants who were either evaluable at that time point or had experienced disease progression or treatment failure prior to the scheduled visit ^ Participants enrolled under an earlier protocol version with 6 - month duration; therefore, they are not included in CR analyses from Month 9 onward. Data cutoff: January 28, 2026 11 100% Maintained CR from 9 Mo - 12 Mo (11 of 11) 73% * (95% CI, 52.9, 93.4) Maintained CR for > 6 Mo © 2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute. *Based on Kaplan - Meier (KM) probability of maintaining a CR
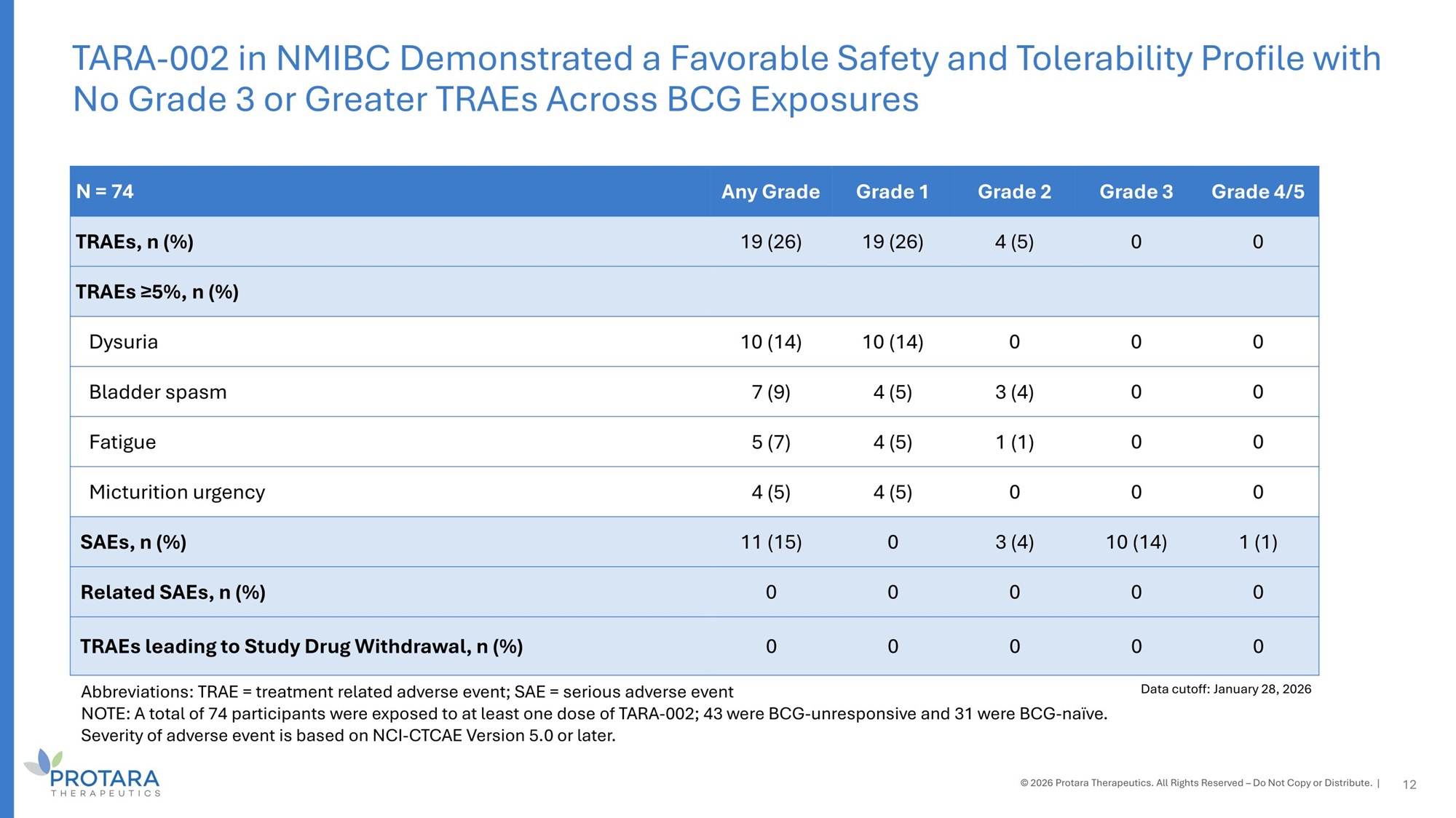
Grade 4/5 Grade 3 Grade 2 Grade 1 Any Grade N = 74 0 0 4 (5) 19 (26) 19 (26) TRAEs, n (%) TRAEs ≥5%, n (%) 0 0 0 10 (14) 10 (14) Dysuria 0 0 3 (4) 4 (5) 7 (9) Bladder spasm 0 0 1 (1) 4 (5) 5 (7) Fatigue 0 0 0 4 (5) 4 (5) Micturition urgency 1 (1) 10 (14) 3 (4) 0 11 (15) SAEs, n (%) 0 0 0 0 0 Related SAEs, n (%) 0 0 0 0 0 TRAEs leading to Study Drug Withdrawal, n (%) Abbreviations: TRAE = treatment related adverse event; SAE = serious adverse event NOTE: A total of 74 participants were exposed to at least one dose of TARA - 002; 43 were BCG - unresponsive and 31 were BCG - naïve. Severity of adverse event is based on NCI - CTCAE Version 5.0 or later. TARA - 002 in NMIBC Demonstrated a Favorable Safety and Tolerability Profile with No Grade 3 or Greater TRAEs Across BCG Exposures © 2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute. | 12 Data cutoff: January 28, 2026

KOL DISCUSSION Neal Shore, MD Medical Director, START Carolinas/Carolina Urologic Research Center

CLOSING REMARKS

ǪCA Jesse Shefferman Co - founder, Director, Chief Executive Officer Jacqueline Zummo, PhD Co - founder, Senior Vice President, Chief RCD Officer © 2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute. | 15 Neal Shore, MD Medical Director, START Carolinas/ Carolina Urologic Research Center Carla Beckham, MD, PhD Lead Medical Director and Head of Clinical Development of TARA - 002 in NMIBC Bill Conkling Chief Commercial Officer Leonardo Viana Nicacio, MD Chief Medical Officer Pat Fabbio Chief Financial Officer

THANK YOU

APPENDIX
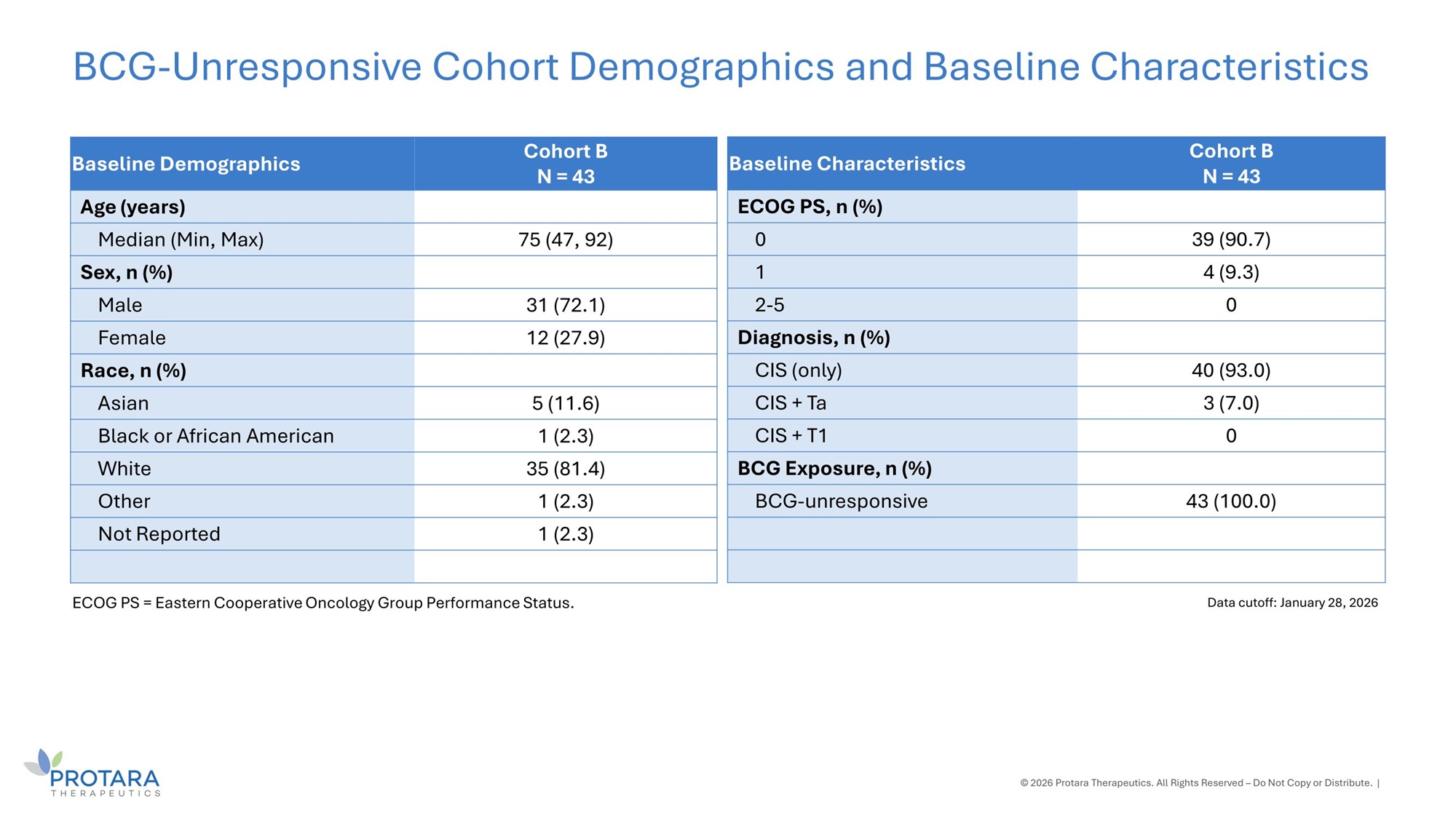
Cohort B N = 43 Baseline Demographics Age (years) 75 (47, 92) Median (Min, Max) Sex, n (%) 31 (72.1) Male 12 (27.9) Female Race, n (%) 5 (11.6) Asian 1 (2.3) Black or African American 35 (81.4) White 1 (2.3) Other 1 (2.3) Not Reported BCG - Unresponsive Cohort Demographics and Baseline Characteristics © 2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute. | Data cutoff: January 28, 2026 ECOG PS = Eastern Cooperative Oncology Group Performance Status. Cohort B N = 43 Baseline Characteristics ECOG PS, n (%) 39 (90.7) 0 4 (9.3) 1 0 2 - 5 Diagnosis, n (%) 40 (93.0) CIS (only) 3 (7.0) CIS + Ta 0 CIS + T1 BCG Exposure, n (%) 43 (100.0) BCG - unresponsive
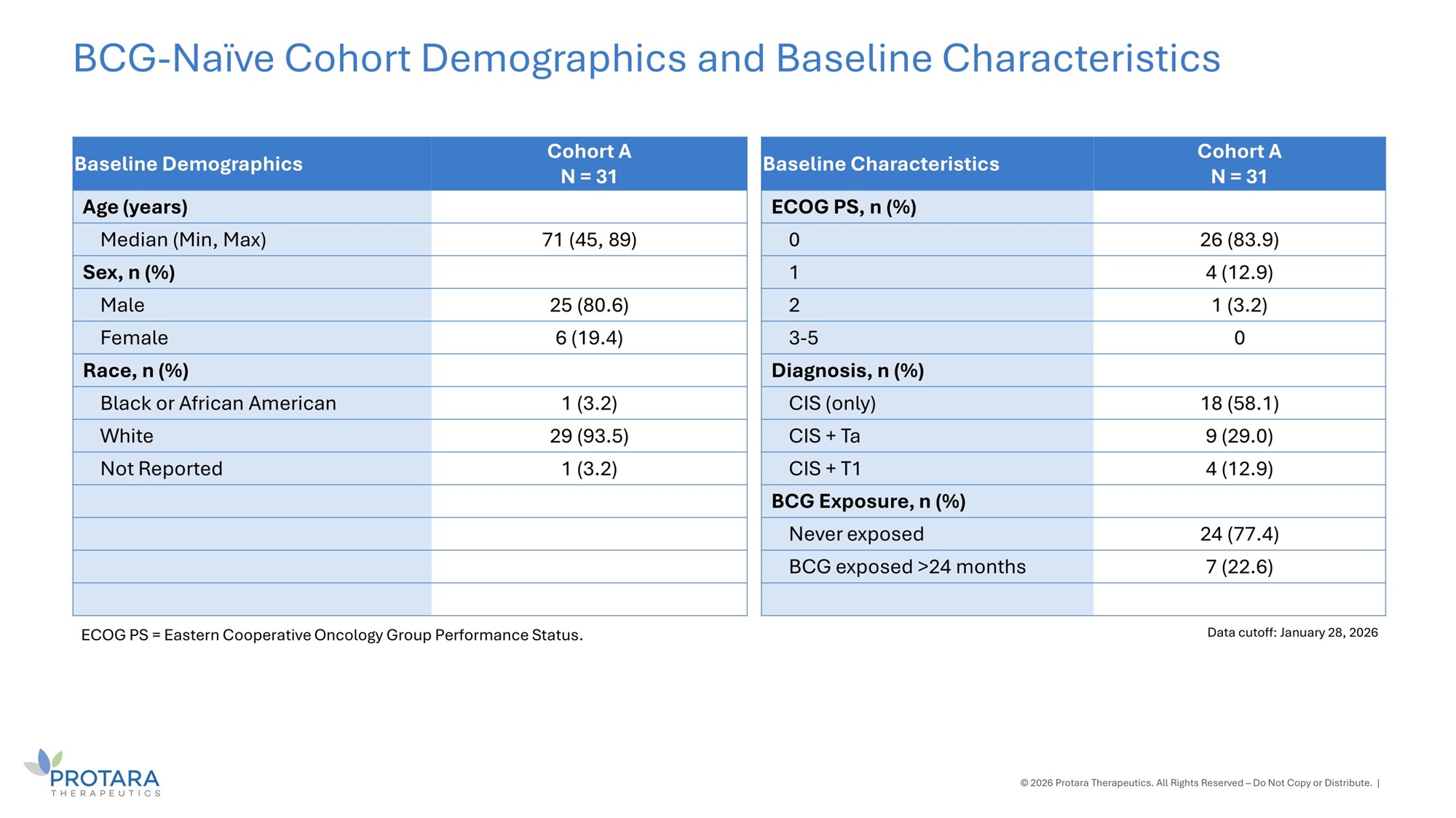
Cohort A N = 31 Baseline Demographics Age (years) 71 (45, 89) Median (Min, Max) Sex, n (%) 25 (80.6) Male 6 (19.4) Female Race, n (%) 1 (3.2) Black or African American 29 (93.5) White 1 (3.2) Not Reported BCG - Naïve Cohort Demographics and Baseline Characteristics © 2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute. | Data cutoff: January 28, 2026 ECOG PS = Eastern Cooperative Oncology Group Performance Status. Cohort A N = 31 Baseline Characteristics ECOG PS, n (%) 26 (83.9) 0 4 (12.9) 1 1 (3.2) 2 0 3 - 5 Diagnosis, n (%) 18 (58.1) CIS (only) 9 (29.0) CIS + Ta 4 (12.9) CIS + T1 BCG Exposure, n (%) 24 (77.4) Never exposed 7 (22.6) BCG exposed >24 months