
.3

Corporate Presentation February 2026

Forward Looking Statements 1 ©2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute Statements contained in this presentation regarding matters that are not historical facts are "forward looking statements" within the meaning of the Private Securities Litigation Reform Act of 1995. Protara may, in some cases, use terms such as “predicts,” “believes,” “potential,” “proposed,” “continue,” “designed,” “estimates,” “anticipates,” “expects,” “plans,” “intends,” “may,” “could,” “might,” “will,” “should” or other words or expressions referencing future events, conditions or circumstances that convey uncertainty of future events or outcomes to identify these forward - looking statements. Such forward - looking statements include but are not limited to, statements regarding Protara’s intentions, beliefs, projections, outlook, analyses or current expectations concerning, among other things: Protara’s business strategy, including its development plans for its product candidates and plans regarding the timing or outcome of existing or future clinical trials (including the timing of any particular phases of such trials and the timing of the announcement of any data produced during such trials or phases thereof); statements related to expectations regarding interactions with the U.S. Food and Drug Administration (FDA); Protara’s financial position; statements regarding the anticipated safety or efficacy of Protara’s product candidates; and Protara’s outlook for the remainder of the year and future periods. Because such statements are subject to risks and uncertainties, actual results may differ materially from those expressed or implied by such forward - looking statements. Factors that contribute to the uncertain nature of the forward - looking statements include: risks that Protara’s financial guidance may not be as expected, as well as risks and uncertainties associated with: Protara’s development programs, including the initiation and completion of non - clinical studies and clinical trials and the timing of required filings with the FDA and other regulatory agencies; general market conditions; changes in the competitive landscape; changes in Protara’s strategic and commercial plans; Protara’s ability to obtain sufficient financing to fund its strategic plans and commercialization efforts; having to use cash in ways or on timing other than expected; the impact of market volatility on cash reserves; failure to attract and retain management and key personnel; the impact of general U.S. and foreign, economic, industry, market, regulatory, political or public health conditions; and the risks and uncertainties associated with Protara’s business and financial condition in general, including the risks and uncertainties described more fully under the caption “Risk Factors” and elsewhere in Protara's filings and reports with the United States Securities and Exchange Commission. All forward - looking statements contained in this presentation speak only as of the date on which they were made and are based on management's assumptions and estimates as of such date. Protara undertakes no obligation to update any forward - looking statements, whether as a result of the receipt of new information, the occurrence of future events or otherwise, except as required by law.
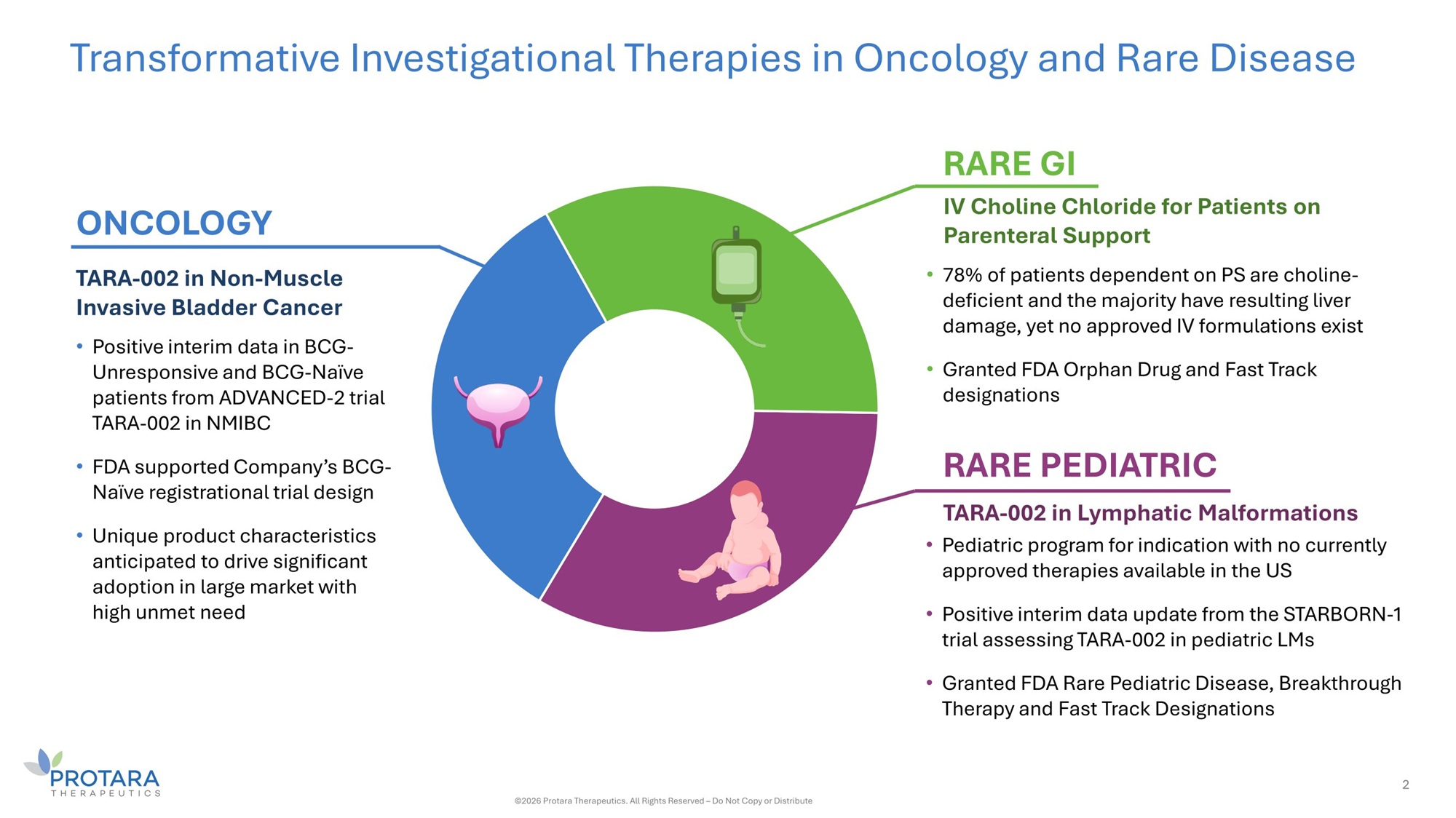
Transformative Investigational Therapies in Oncology and Rare Disease TARA - 002 in Non - Muscle Invasive Bladder Cancer • Positive interim data in BCG - Unresponsive and BCG - Naïve patients from ADVANCED - 2 trial TARA - 002 in NMIBC • FDA supported Company’s BCG - Naïve registrational trial design • Unique product characteristics anticipated to drive significant adoption in large market with high unmet need IV Choline Chloride for Patients on Parenteral Support • 78% of patients dependent on PS are choline - deficient and the majority have resulting liver damage, yet no approved IV formulations exist • Granted FDA Orphan Drug and Fast Track designations ONCOLOGY RARE GI RARE PEDIATRIC TARA - 002 in Lymphatic Malformations • Pediatric program for indication with no currently approved therapies available in the US • Positive interim data update from the STARBORN - 1 trial assessing TARA - 002 in pediatric LMs • Granted FDA Rare Pediatric Disease, Breakthrough Therapy and Fast Track Designations 2 ©2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute
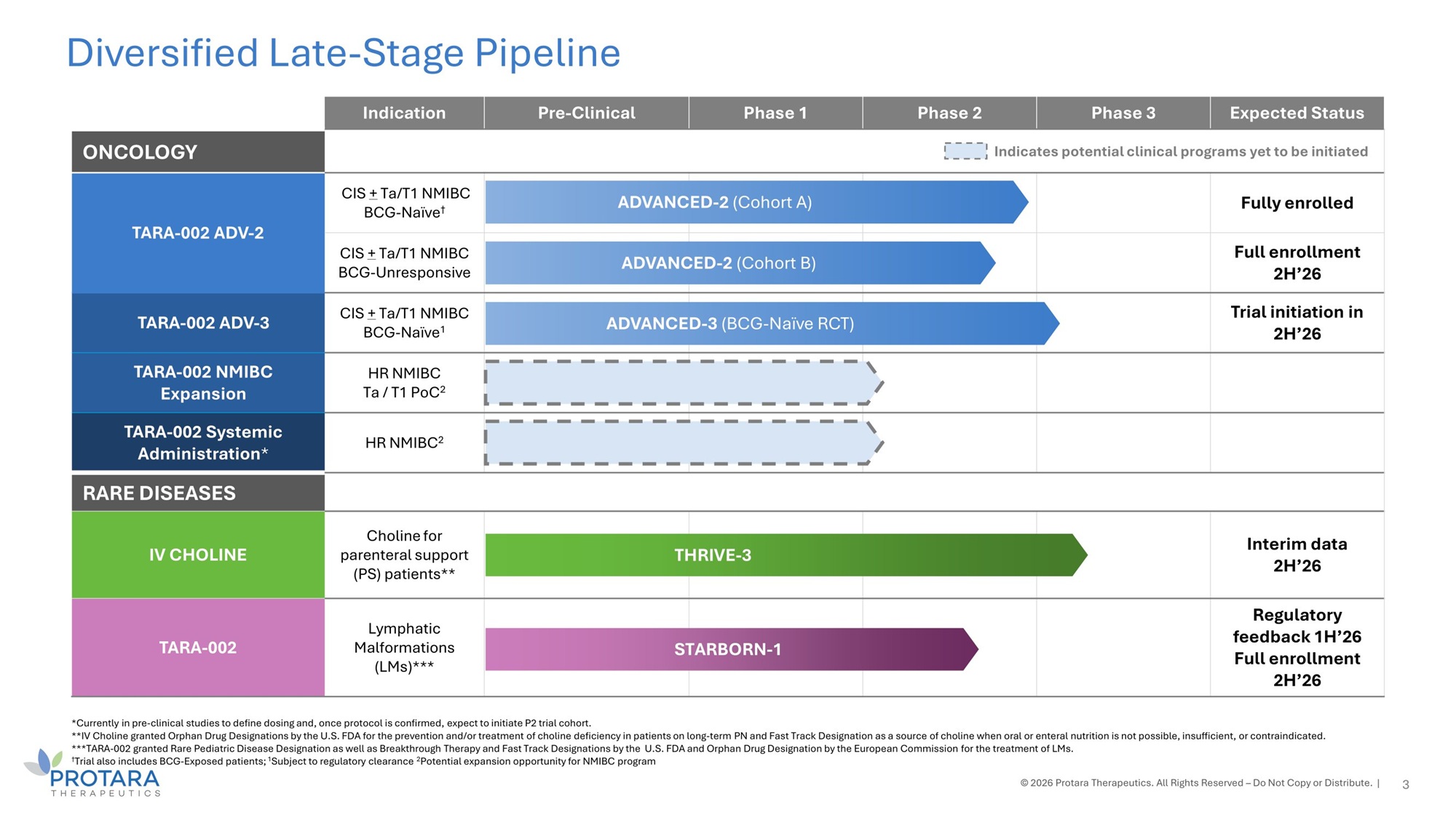
*Currently in pre - clinical studies to define dosing and, once protocol is confirmed, expect to initiate P2 trial cohort. **IV Choline granted Orphan Drug Designations by the U.S. FDA for the prevention and/or treatment of choline deficiency in patients on long - term PN and Fast Track Designation as a source of choline when oral or enteral nutrition is not possible, insufficient, or contraindicated. ***TARA - 002 granted Rare Pediatric Disease Designation as well as Breakthrough Therapy and Fast Track Designations by the U.S. FDA and Orphan Drug Designation by the European Commission for the treatment of LMs. † Trial also includes BCG - Exposed patients; 1 Subject to regulatory clearance 2 Potential expansion opportunity for NMIBC program © 2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute. | Diversified Late - Stage Pipeline Expected Status Phase 3 Phase 2 Phase 1 Pre - Clinical Indication Indicates potential clinical programs yet to be initiated ONCOLOGY Fully enrolled ED - 2 (Cohort A) ADVANC CIS + Ta/T1 NMIBC BCG - Naïve † TARA - 002 ADV - 2 Full enrollment 2H’26 ED - 2 (Cohort B) ADVANC CIS + Ta/T1 NMIBC BCG - Unresponsive Trial initiation in 2H’26 - 3 (BCG - Naïve RCT) ADVANCED CIS + Ta/T1 NMIBC BCG - Naïve 1 TARA - 002 ADV - 3 HR NMIBC Ta / T1 PoC 2 TARA - 002 NMIBC Expansion HR NMIBC 2 TARA - 002 Systemic Administration * RARE DISEASES Interim data 2H’26 HRIVE - 3 T Choline for parenteral support (PS) patients** IV CHOLINE Regulatory feedback 1H’26 Full enrollment 2H’26 TARBORN - 1 S Lymphatic Malformations (LMs)*** TARA - 002 3
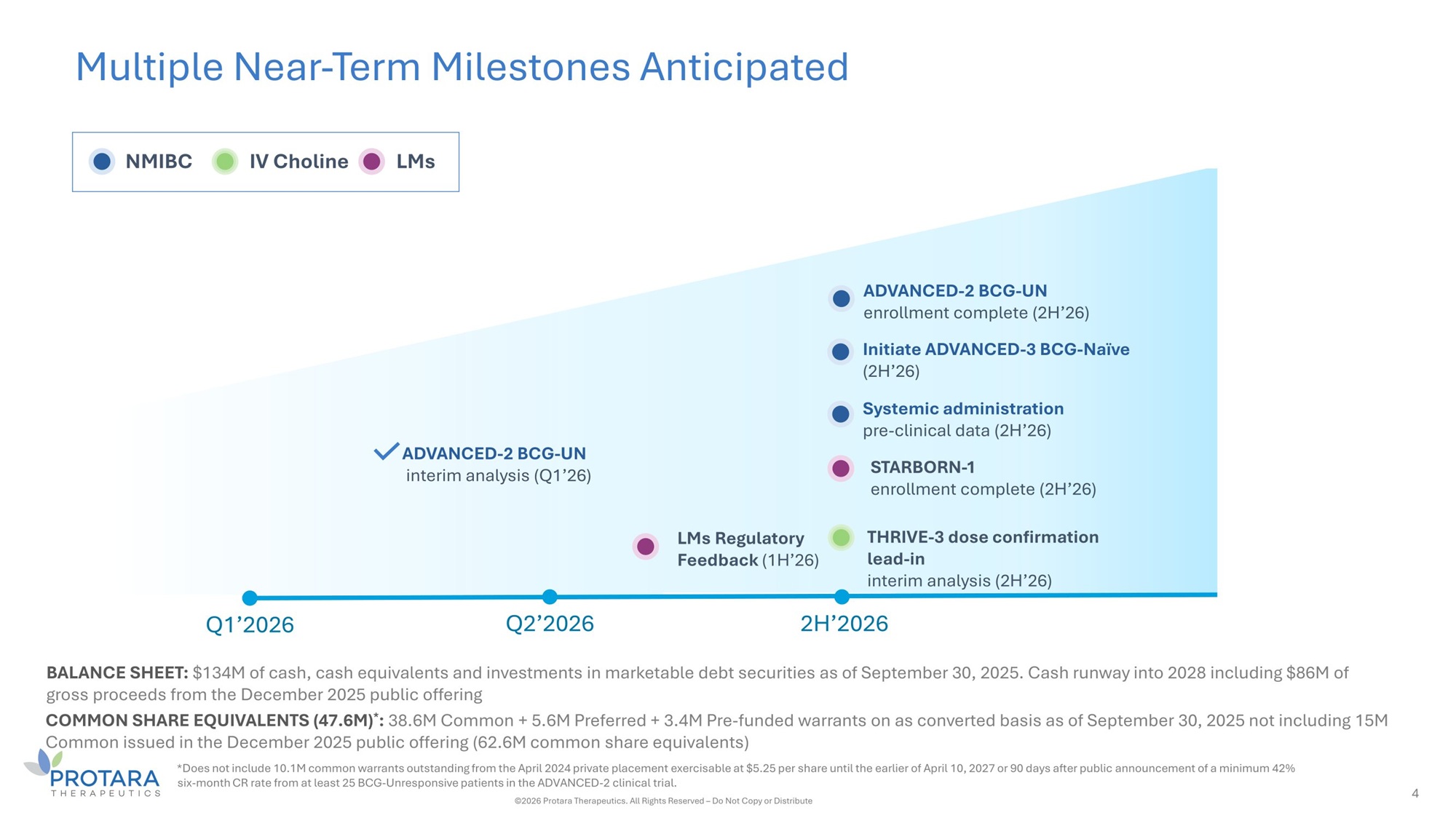
Multiple Near - Term Milestones Anticipated 4 ADVANCED - 2 BCG - UN interim analysis (Ǫ1’26) NMIBC IV Choline LMs Ǫ1’2026 Ǫ2’2026 2H’2026 BALANCE SHEET: $134M of cash, cash equivalents and investments in marketable debt securities as of September 30, 2025. Cash runway into 2028 including $86M of gross proceeds from the December 2025 public offering COMMON SHARE EǪUIVALENTS (47.6M) * : 38.6M Common + 5.6M Preferred + 3.4M Pre - funded warrants on as converted basis as of September 30, 2025 not including 15M Common issued in the December 2025 public offering (62.6M common share equivalents) *Does not include 10.1M common warrants outstanding from the April 2024 private placement exercisable at $5.25 per share until the earlier of April 10, 2027 or 90 days after public announcement of a minimum 42% six - month CR rate from at least 25 BCG - Unresponsive patients in the ADVANCED - 2 clinical trial. ©2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute THRIVE - 3 dose confirmation lead - in interim analysis (2H’26) LMs Regulatory Feedback (1H’26) ADVANCED - 2 BCG - UN enrollment complete (2H’26) Initiate ADVANCED - 3 BCG - Naïve (2H’26) Systemic administration pre - clinical data (2H’26) STARBORN - 1 enrollment complete (2H’26)

TARA - 002 Non - Muscle Invasive Bladder Cancer (NMIBC)
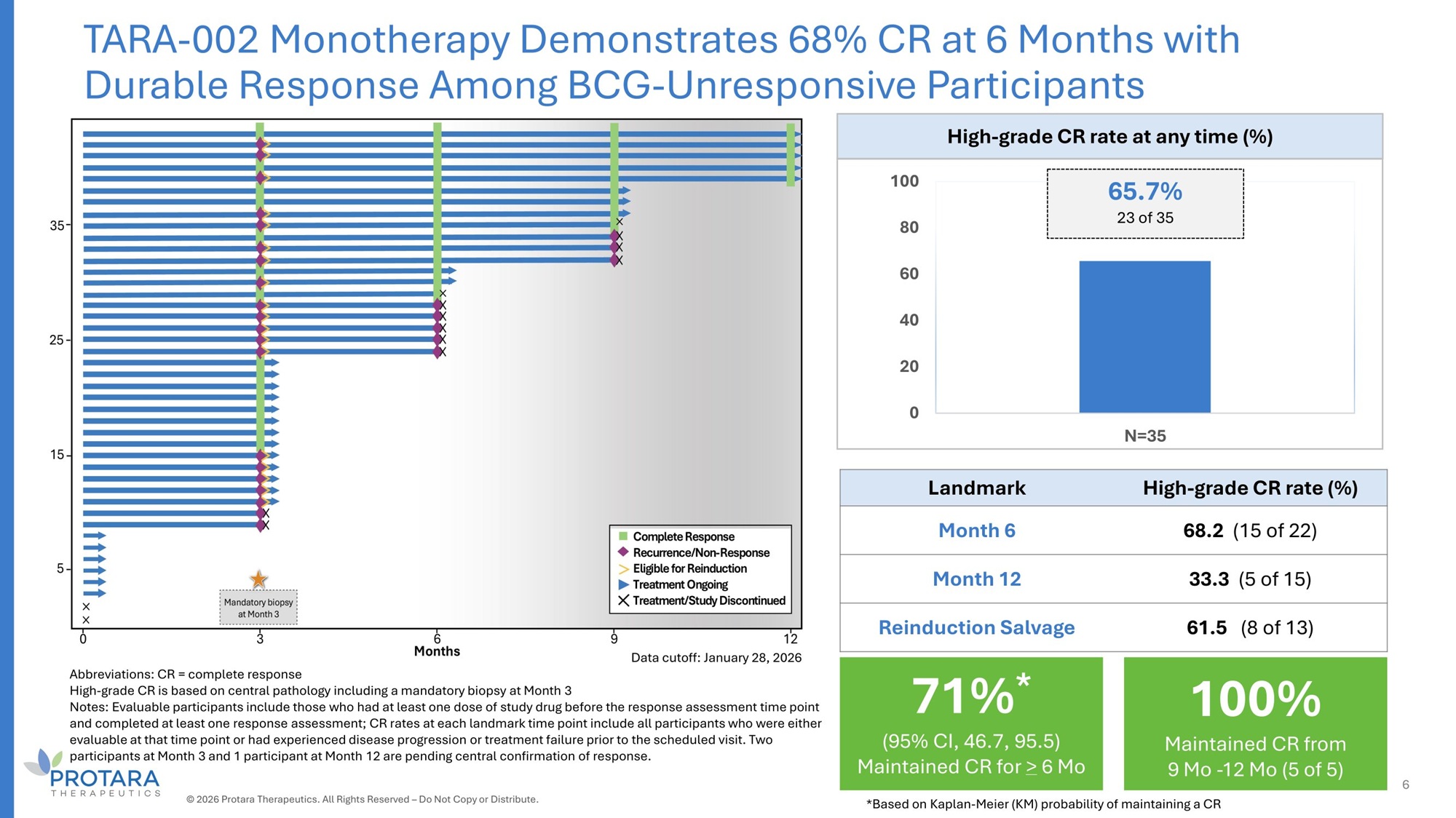
Abbreviations: CR = complete response High - grade CR is based on central pathology including a mandatory biopsy at Month 3 Notes: Evaluable participants include those who had at least one dose of study drug before the response assessment time point and completed at least one response assessment; CR rates at each landmark time point include all participants who were either evaluable at that time point or had experienced disease progression or treatment failure prior to the scheduled visit. Two participants at Month 3 and 1 participant at Month 12 are pending central confirmation of response. High - grade CR rate at any time (%) 65.7% 100 23 of 35 80 60 40 20 0 N=35 High - grade CR rate (%) Landmark 68.2 (15 of 22) Month 6 33.3 (5 of 15) Month 12 61.5 (8 of 13) Reinduction Salvage TARA - 002 Monotherapy Demonstrates 68% CR at 6 Months with Durable Response Among BCG - Unresponsive Participants Data cutoff: January 28, 2026 100% Maintained CR from 9 Mo - 12 Mo (5 of 5) 71% * (95% CI, 46.7, 95.5) Maintained CR for > 6 Mo 6 © 2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute. *Based on Kaplan - Meier (KM) probability of maintaining a CR
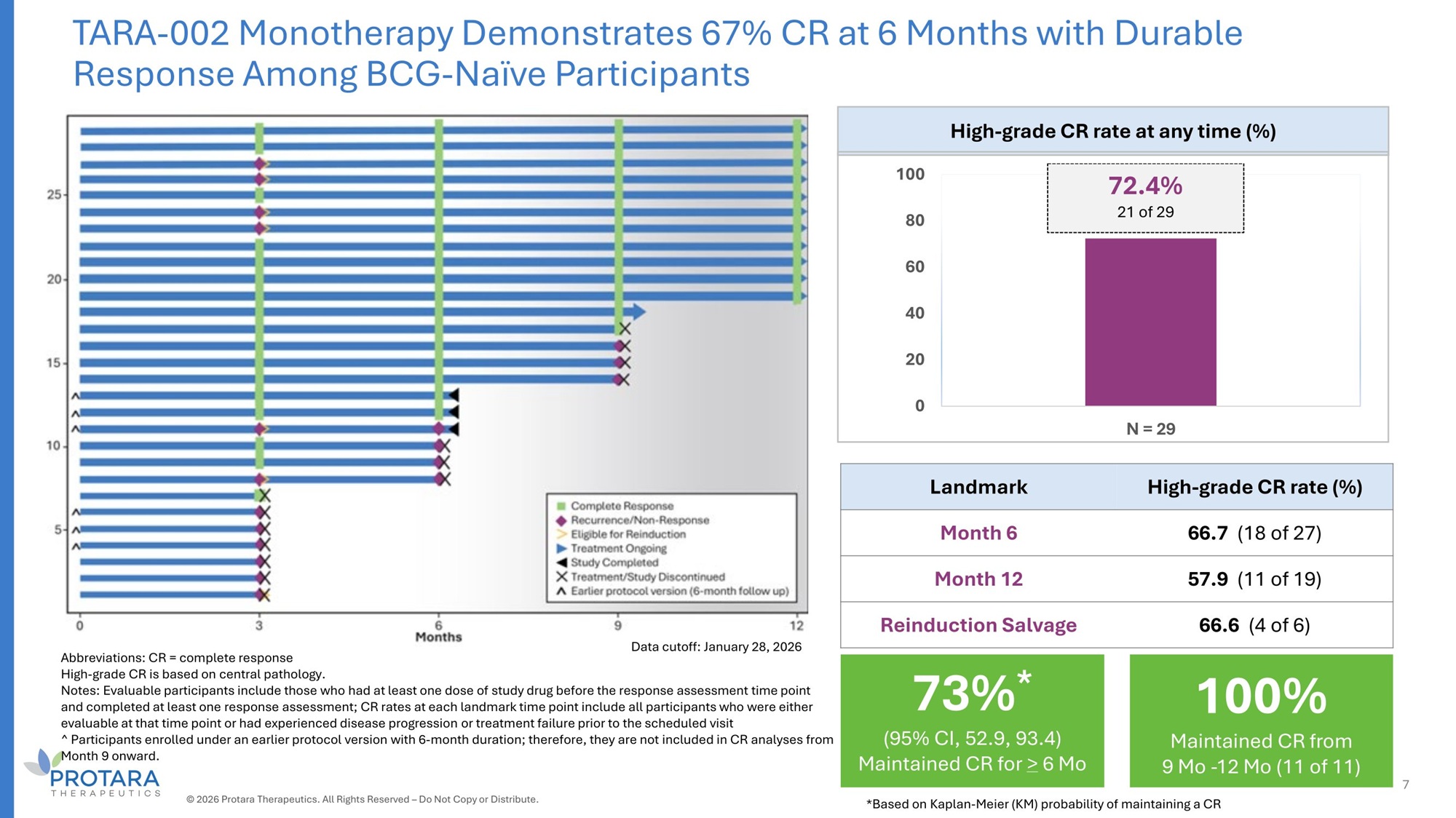
TARA - 002 Monotherapy Demonstrates 67% CR at 6 Months with Durable Response Among BCG - Naïve Participants High - grade CR rate at any time (%) 72.4% 100 21 of 29 80 60 40 20 0 N = 2G High - grade CR rate (%) Landmark 66.7 (18 of 27) Month 6 57.G (11 of 19) Month 12 66.6 (4 of 6) Reinduction Salvage Abbreviations: CR = complete response High - grade CR is based on central pathology. Notes: Evaluable participants include those who had at least one dose of study drug before the response assessment time point and completed at least one response assessment; CR rates at each landmark time point include all participants who were either evaluable at that time point or had experienced disease progression or treatment failure prior to the scheduled visit ^ Participants enrolled under an earlier protocol version with 6 - month duration; therefore, they are not included in CR analyses from Month 9 onward. Data cutoff: January 28, 2026 100% Maintained CR from 9 Mo - 12 Mo (11 of 11) 73% * (95% CI, 52.9, 93.4) Maintained CR for > 6 Mo 7 © 2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute. *Based on Kaplan - Meier (KM) probability of maintaining a CR
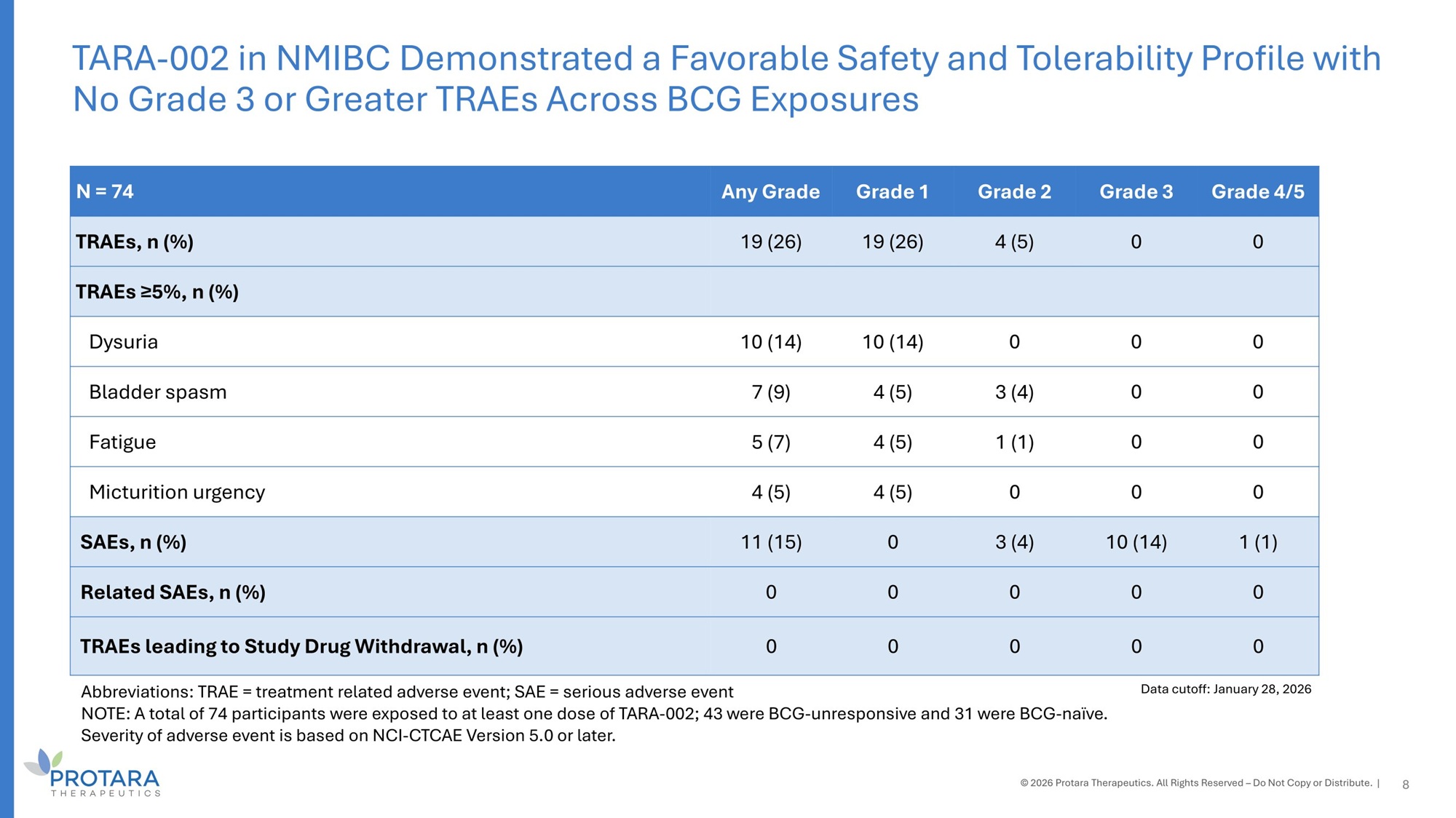
Grade 4/5 Grade 3 Grade 2 Grade 1 Any Grade N = 74 0 0 4 (5) 19 (26) 19 (26) TRAEs, n (%) TRAEs ≥5%, n (%) 0 0 0 10 (14) 10 (14) Dysuria 0 0 3 (4) 4 (5) 7 (9) Bladder spasm 0 0 1 (1) 4 (5) 5 (7) Fatigue 0 0 0 4 (5) 4 (5) Micturition urgency 1 (1) 10 (14) 3 (4) 0 11 (15) SAEs, n (%) 0 0 0 0 0 Related SAEs, n (%) 0 0 0 0 0 TRAEs leading to Study Drug Withdrawal, n (%) Abbreviations: TRAE = treatment related adverse event; SAE = serious adverse event NOTE: A total of 74 participants were exposed to at least one dose of TARA - 002; 43 were BCG - unresponsive and 31 were BCG - naïve. Severity of adverse event is based on NCI - CTCAE Version 5.0 or later. TARA - 002 in NMIBC Demonstrated a Favorable Safety and Tolerability Profile with No Grade 3 or Greater TRAEs Across BCG Exposures © 2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute. | 8 Data cutoff: January 28, 2026
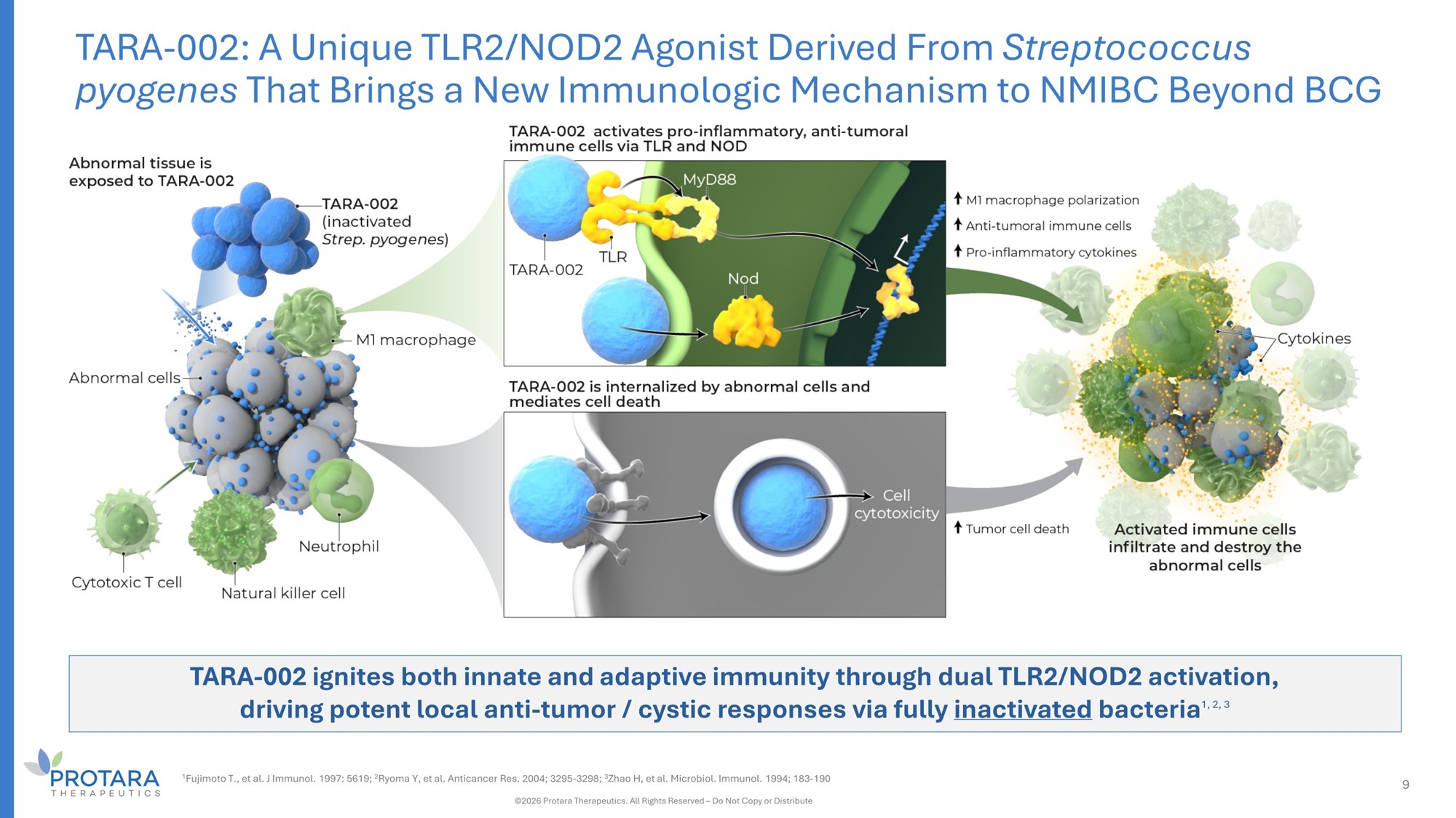
TARA - 002 ignites both innate and adaptive immunity through dual TLR2/NOD2 activation, driving potent local anti - tumor / cystic responses via fully inactivated bacteria 1, 2, 3 TARA - 002: A Unique TLR2/NOD2 Agonist Derived From Streptococcus pyogenes That Brings a New Immunologic Mechanism to NMIBC Beyond BCG 1 Fujimoto T., et al. J Immunol. 1997: 5619; 2 Ryoma Y, et al. Anticancer Res. 2004; 3295 - 3298; 3 Zhao H, et al. Microbiol. Immunol. 1994; 183 - 190 ©2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute 9
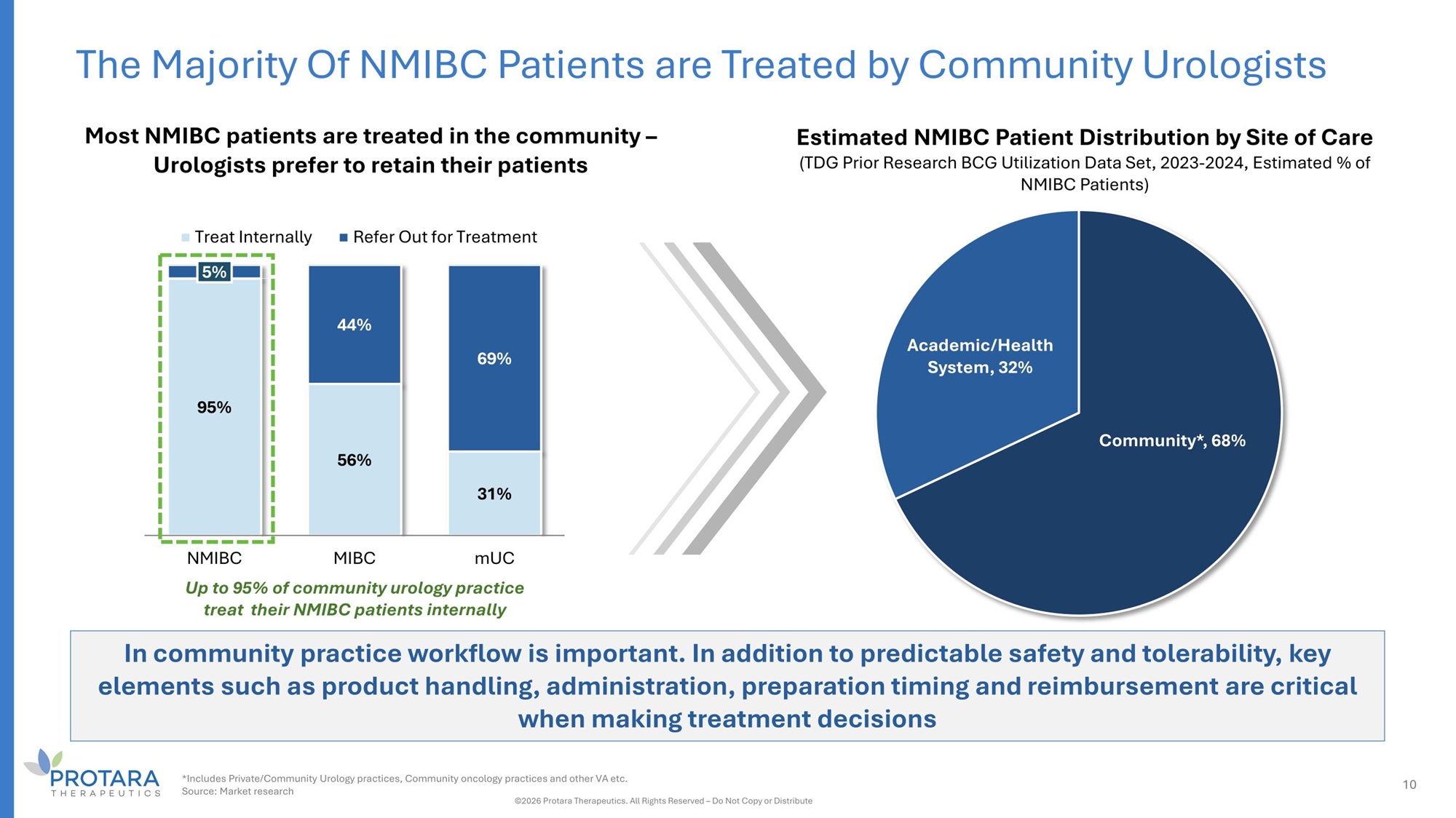
The Majority Of NMIBC Patients are Treated by Community Urologists Estimated NMIBC Patient Distribution by Site of Care (TDG Prior Research BCG Utilization Data Set, 2023 - 2024, Estimated % of NMIBC Patients) Community*, 68% Academic/Health System, 32% G5% 56% 31% 5% 44% 6G% NMIBC MIBC mUC Up to 35% of community urology practice treat their NMIBC patients internally Treat Internally Refer Out for Treatment Most NMIBC patients are treated in the community – Urologists prefer to retain their patients In community practice workflow is important. In addition to predictable safety and tolerability, key elements such as product handling, administration, preparation timing and reimbursement are critical when making treatment decisions *Includes Private/Community Urology practices, Community oncology practices and other VA etc. Source: Market research ©2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute 10
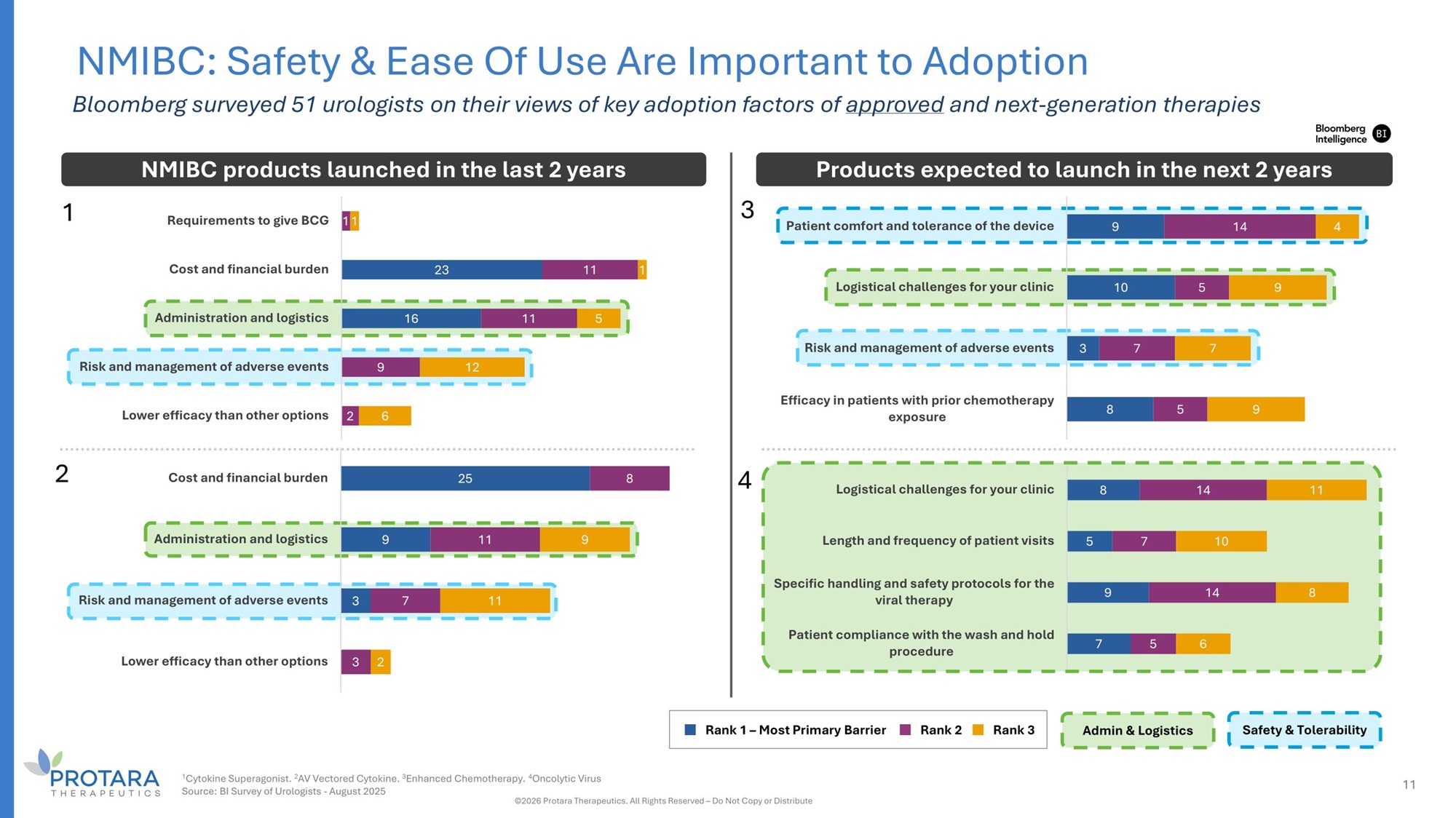
23 16 11 11 9 2 1 5 12 6 Requirements to give BCG 11 Cost and financial burden Administration and logistics Risk and management of adverse events Lower efficacy than other options NMIBC: Safety C Ease Of Use Are Important to Adoption Bloomberg surveyed 51 urologists on their views of key adoption factors of approved and next - generation therapies 9 10 8 14 5 7 5 4 9 7 9 Patient comfort and tolerance of the device Logistical challenges for your clinic Risk and management of adverse events 3 Efficacy in patients with prior chemotherapy exposure 25 9 3 8 11 7 9 11 Cost and financial burden Administration and logistics Risk and management of adverse events Lower efficacy than other options 3 2 8 5 9 7 14 7 14 5 11 10 8 6 Logistical challenges for your clinic Length and frequency of patient visits Specific handling and safety protocols for the viral therapy Patient compliance with the wash and hold procedure NMIBC products launched in the last 2 years Products expected to launch in the next 2 years Admin G Logistics Safety G Tolerability Rank 1 – Most Primary Barrier Rank 2 Rank 3 1 4 2 3 1 Cytokine Superagonist. 2 AV Vectored Cytokine. 3 Enhanced Chemotherapy. 4 Oncolytic Virus Source: BI Survey of Urologists - August 2025 ©2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute 11

TARA - 002 No vial thaw, simple preparation; no pre/post treatment protocol Preparation: can be conducted on open table or benchtop; no special handling Detalimogene voraplasmid No vial thaw, simple preparation; no pre/post treatment protocol Anktiva + BCG - TICE Risk of infection for patient and caregivers; urine decontamination burden Adstiladrin Vial thaw introduces preparation bottleneck; elevated biosafety procedures required; additional pre/post burden for patients Cretostimogene Complex process with unclear vial thaw, biosafety, and urine decontamination requirements TARA - 002 has reduced burden for patients and physicians Preparation: can be conducted on open table or benchtop; no special handling Preparation: mask/gown required to avoid infection 6 - hour period of required urine bleaching Antichollinergic premedication 3 – 10 hour vial thaw Preparation : “universal biosafety precautions” required ; USPI contains infection risk warning 48 - hour period of required urine bleaching Thaw process not disclosed Preparation: BSL2/2+ handling likely required Saline wash DDM wash Saline wash DDM re - infusion and dwell (15 min) Urine bleaching requirements not disclosed Inlexzo Requires insertion every three weeks, potential for patients to feel device in bladder and removal requires cystoscopy Assembly of the device on open table or benchtop Cystoscopy is encouraged prior to instillation to check for bladder mucosal integrity Removal of the device requiring cystoscopy Repeat the same procedure every 3 weeks 12 TARA - 002 Is Designed For Best - in - Class Ease of Use in NMIBC Definitions: USPI – U.S. prescribing information; DDM - dodecyl maltoside Source: Derived from product SOPIs and clinical trial publications. Not based on head - to - head or comparator studies. ©2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute
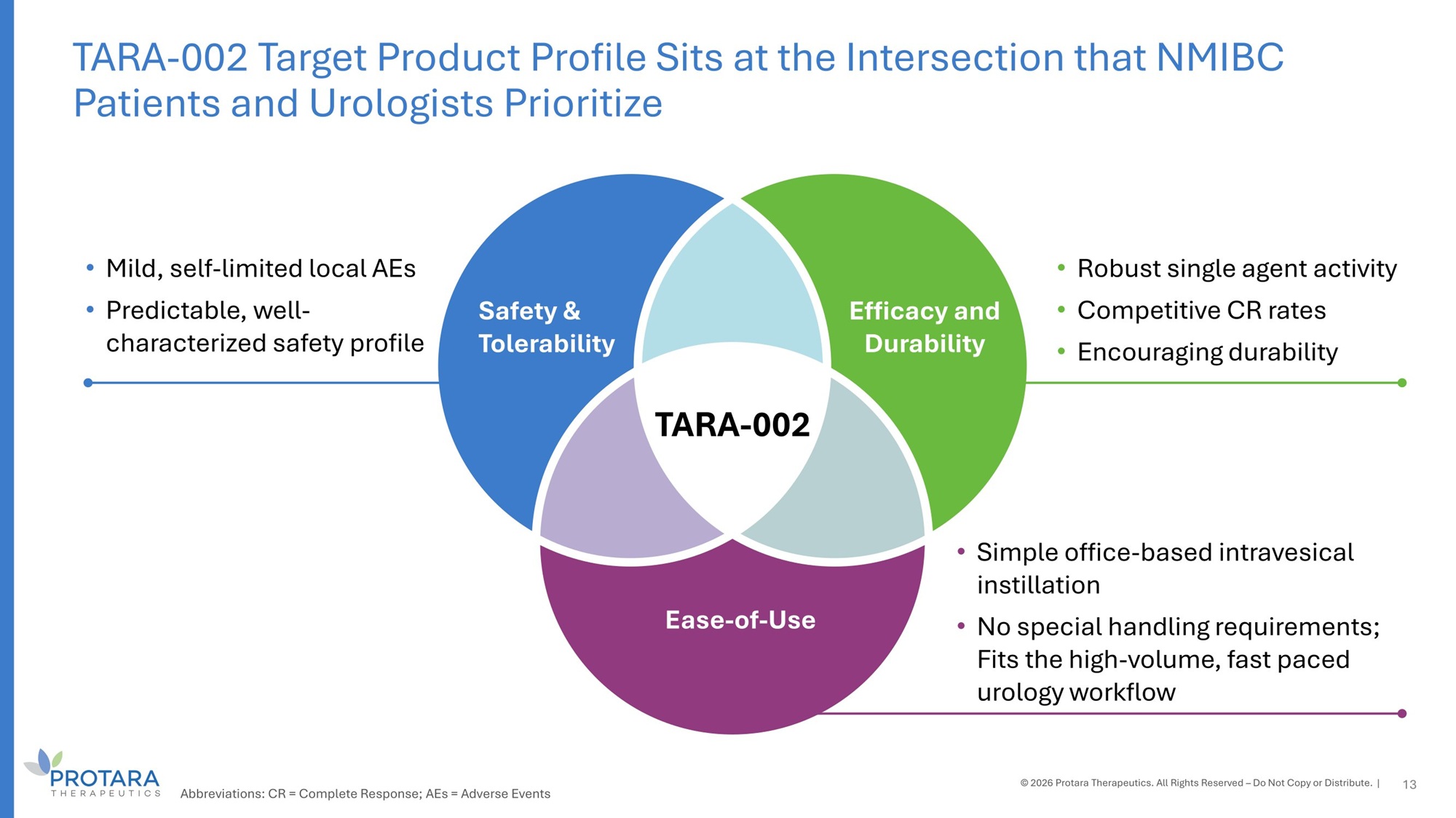
Safety G Tolerability Efficacy and Durability Ease - of - Use • Mild, self - limited local AEs • Predictable, well - characterized safety profile • Robust single agent activity • Competitive CR rates • Encouraging durability • Simple office - based intravesical instillation • No special handling requirements; Fits the high - volume, fast paced urology workflow TARA - 002 13 TARA - 002 Target Product Profile Sits at the Intersection that NMIBC Patients and Urologists Prioritize © 2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute. | Abbreviations: CR = Complete Response; AEs = Adverse Events

TARA - 002 Lymphatic Malformations (LMs)
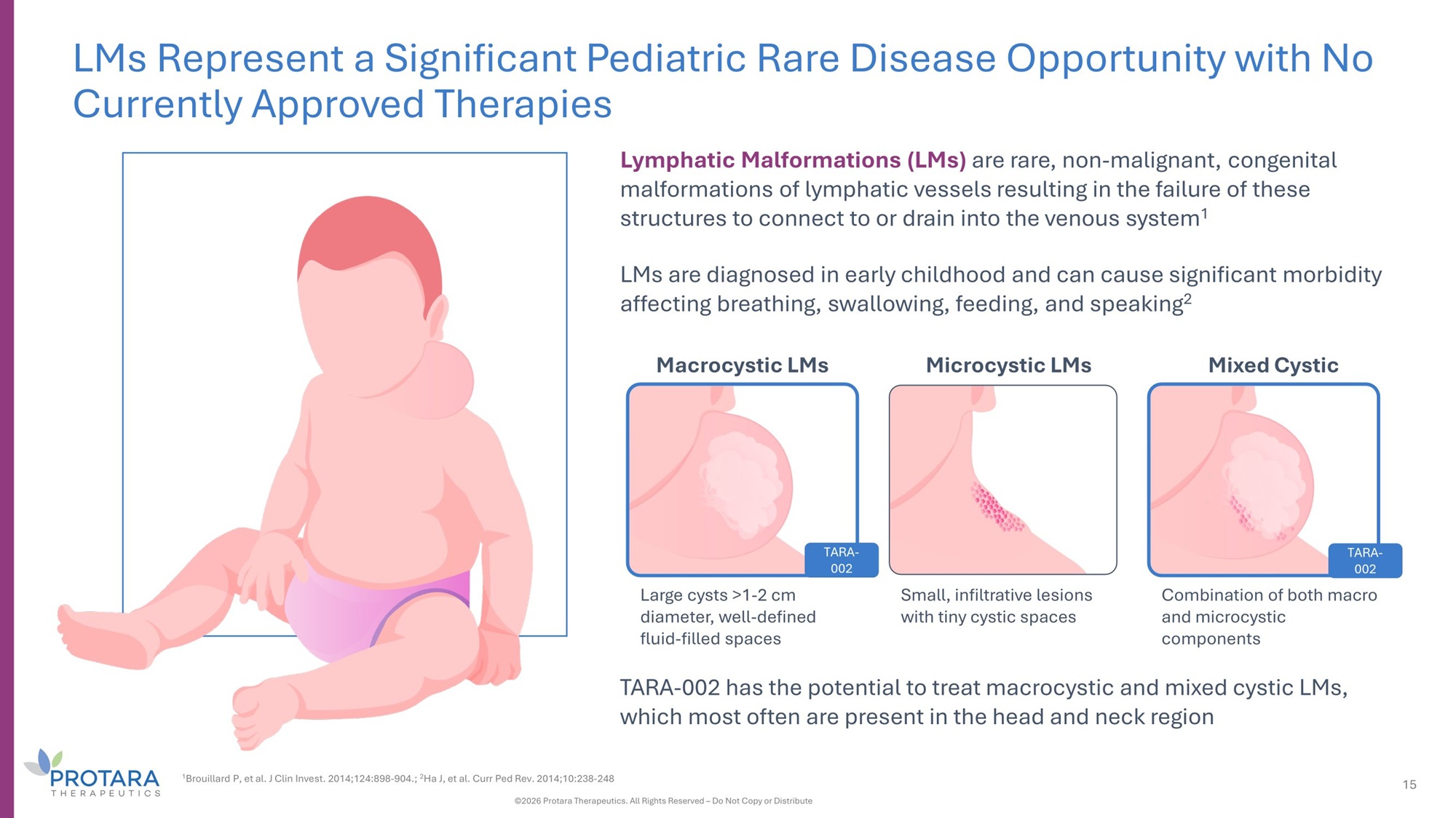
LMs Represent a Significant Pediatric Rare Disease Opportunity with No Currently Approved Therapies Lymphatic Malformations (LMs) are rare, non - malignant, congenital malformations of lymphatic vessels resulting in the failure of these structures to connect to or drain into the venous system 1 LMs are diagnosed in early childhood and can cause significant morbidity affecting breathing, swallowing, feeding, and speaking 2 Macrocystic LMs Microcystic LMs Mixed Cystic Large cysts >1 - 2 cm diameter, well - defined fluid - filled spaces Small, infiltrative lesions with tiny cystic spaces TARA - 002 Combination of both macro and microcystic components TARA - 002 has the potential to treat macrocystic and mixed cystic LMs, which most often are present in the head and neck region 1 Brouillard P, et al. J Clin Invest. 2014;124:898 - 904.; 2 Ha J, et al. Curr Ped Rev. 2014;10:238 - 248 ©2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute 15 TARA - 002
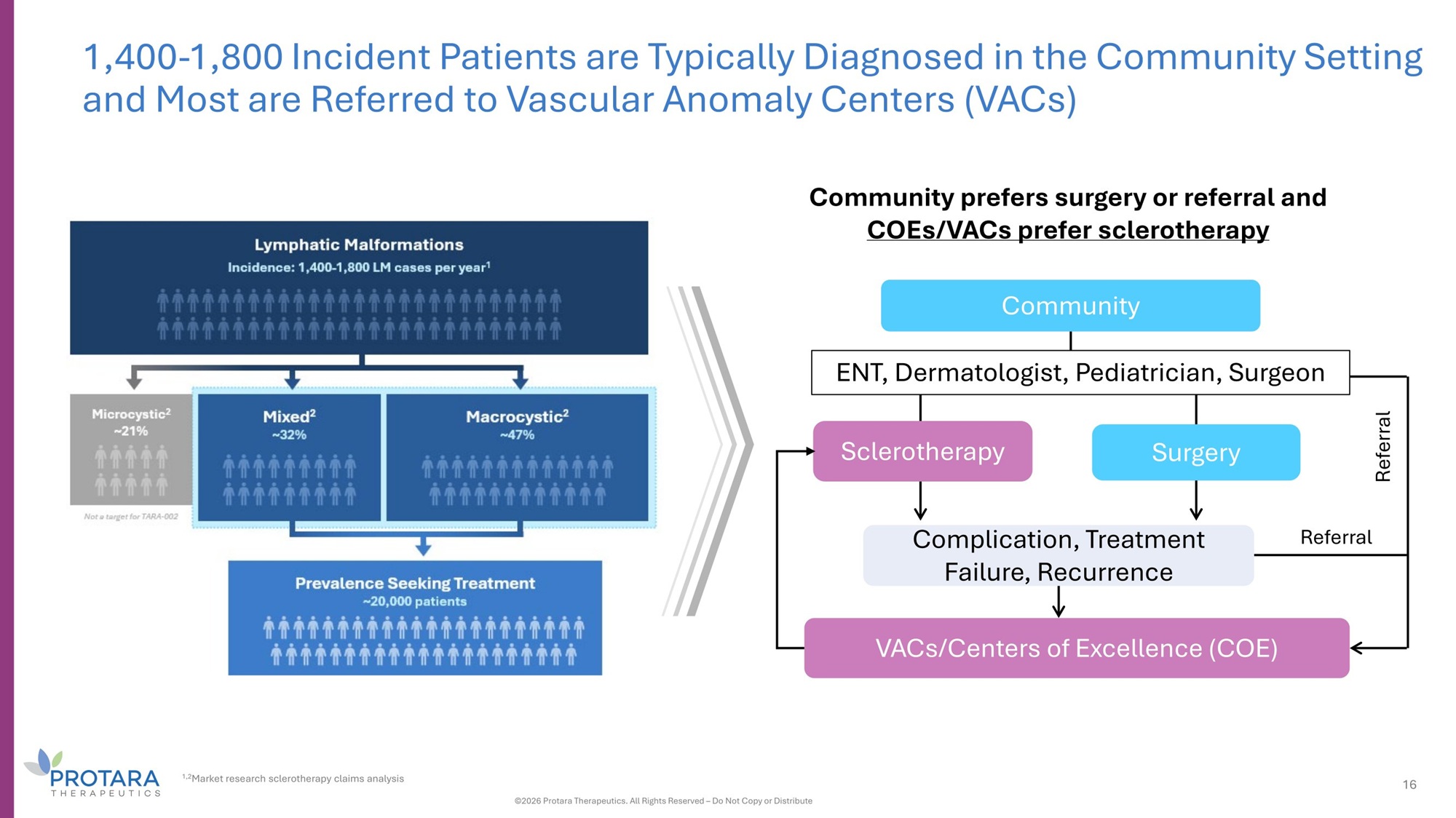
Referral 1,400 - 1,800 Incident Patients are Typically Diagnosed in the Community Setting and Most are Referred to Vascular Anomaly Centers (VACs) VACs/Centers of Excellence (COE) Referral Complication, Treatment Failure, Recurrence Community prefers surgery or referral and COEs/VACs prefer sclerotherapy Community ENT, Dermatologist, Pediatrician, Surgeon Sclerotherapy Surgery 1,2 Market research sclerotherapy claims analysis 16 ©2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute
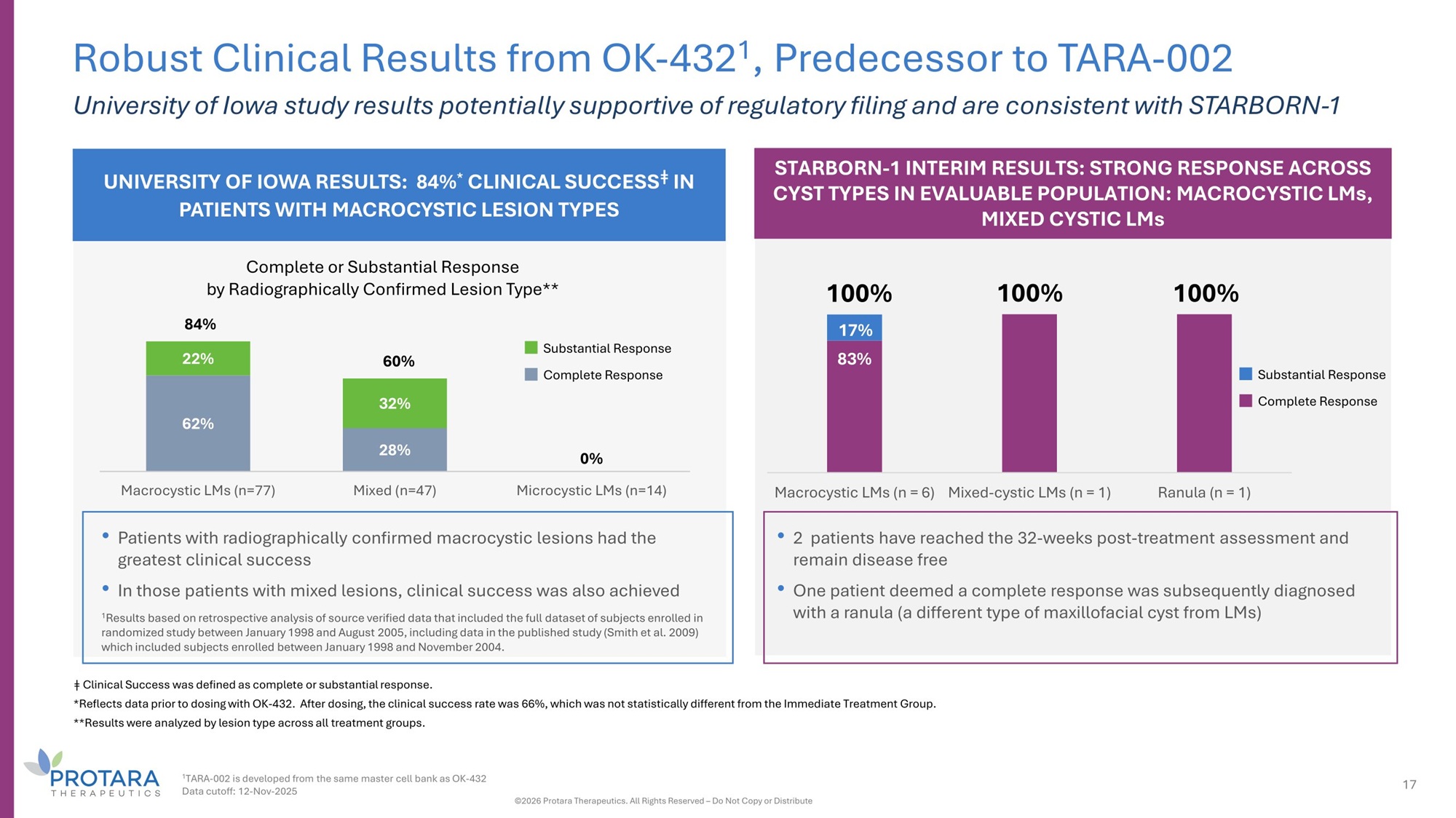
UNIVERSITY OF IOWA RESULTS: 84% * CLINICAL SUCCESS ǂ IN PATIENTS WITH MACROCYSTIC LESION TYPES 62% 28% 0% 22% 32% Macrocystic LMs (n=77) Mixed (n=47) Microcystic LMs (n=14) • Patients with radiographically confirmed macrocystic lesions had the greatest clinical success • In those patients with mixed lesions, clinical success was also achieved 1 Results based on retrospective analysis of source verified data that included the full dataset of subjects enrolled in randomized study between January 1998 and August 2005, including data in the published study (Smith et al. 2009) which included subjects enrolled between January 1998 and November 2004. Complete or Substantial Response by Radiographically Confirmed Lesion Type** 84% 60% Robust Clinical Results from OK - 432 1 , Predecessor to TARA - 002 University of Iowa study results potentially supportive of regulatory filing and are consistent with STARBORN - 1 Substantial Response Complete Response STARBORN - 1 INTERIM RESULTS: STRONG RESPONSE ACROSS CYST TYPES IN EVALUABLE POPULATION: MACROCYSTIC LMs, MIXED CYSTIC LMs ǂ Clinical Success was defined as complete or substantial response. *Reflects data prior to dosing with OK - 432. After dosing, the clinical success rate was 66%, which was not statistically different from the Immediate Treatment Group. **Results were analyzed by lesion type across all treatment groups. • 2 patients have reached the 32 - weeks post - treatment assessment and remain disease free • One patient deemed a complete response was subsequently diagnosed with a ranula (a different type of maxillofacial cyst from LMs) Substantial Response Complete Response 83% 1 TARA - 002 is developed from the same master cell bank as OK - 432 Data cutoff: 12 - Nov - 2025 17 ©2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute 17% Macrocystic LMs (n = 6) Mixed - cystic LMs (n = 1) Ranula (n = 1) 100% 100% 100%

TARA - 002 in LMs Has a Favorable Safety Profile and is Well - Tolerated Data cutoff: November 12, 2025 18 ©2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute Grade 4/5 Grade 3 Grade 2 Grade 1 Any Grade Number of Patients, n = 12 0 (0.0) 1 (8.3) 6 (50.0) 8 (66.7) 8 (66.7) TEAEs, n (%) 0 (0.0) 1 (8.3) 4 (33.3) 7 (58.3) 7 (58.3) Related TEAEs, n (%) Related TEAEs >10%, n (%) 0 (0.0) 1 (8.3) 2 (16.7) 0 (0.0) 3 (25.0) Swelling 0 (0.0) 0 (0.0) 1 (8.3) 2 (16.7) 3 (25.0) Fatigue 0 (0.0) 0 (0.0) 0 (0.0) 2 (16.7) 2 (16.7) Headache 0 (0.0) 0 (0.0) 0 (0.0) 2 (16.7) 2 (16.7) Injection site pain 0 (0.0) 0 (0.0) 0 (0.0) 2 (16.7) 2 (16.7) Injection site rash 0 (0.0) 0 (0.0) 0 (0.0) 2 (16.7) 2 (16.7) Pyrexia 0 (0.0) 0 (0.0) 0 (0.0) 0 (0.0) 0 (0.0) AESIs, n (%) 0 (0.0) 0 (0.0) 0 (0.0) 0 (0.0) 0 (0.0) Serious TEAEs, n (%) 0 (0.0) 0 (0.0) 1 (8.3) 0 (0.0) 1 (8.3) Related TEAEs leading to study drug withdrawal, n (%) • Most AEs were mild to moderate. • No SAEs or AESIs have occurred to date. • Related AEs and systemic reactions are in line with typical systemic responses to bacterial immunopotentiation and the established safety profile of OK - 432. AESI = adverse event of special interest; TEAE = treatment emergent adverse event
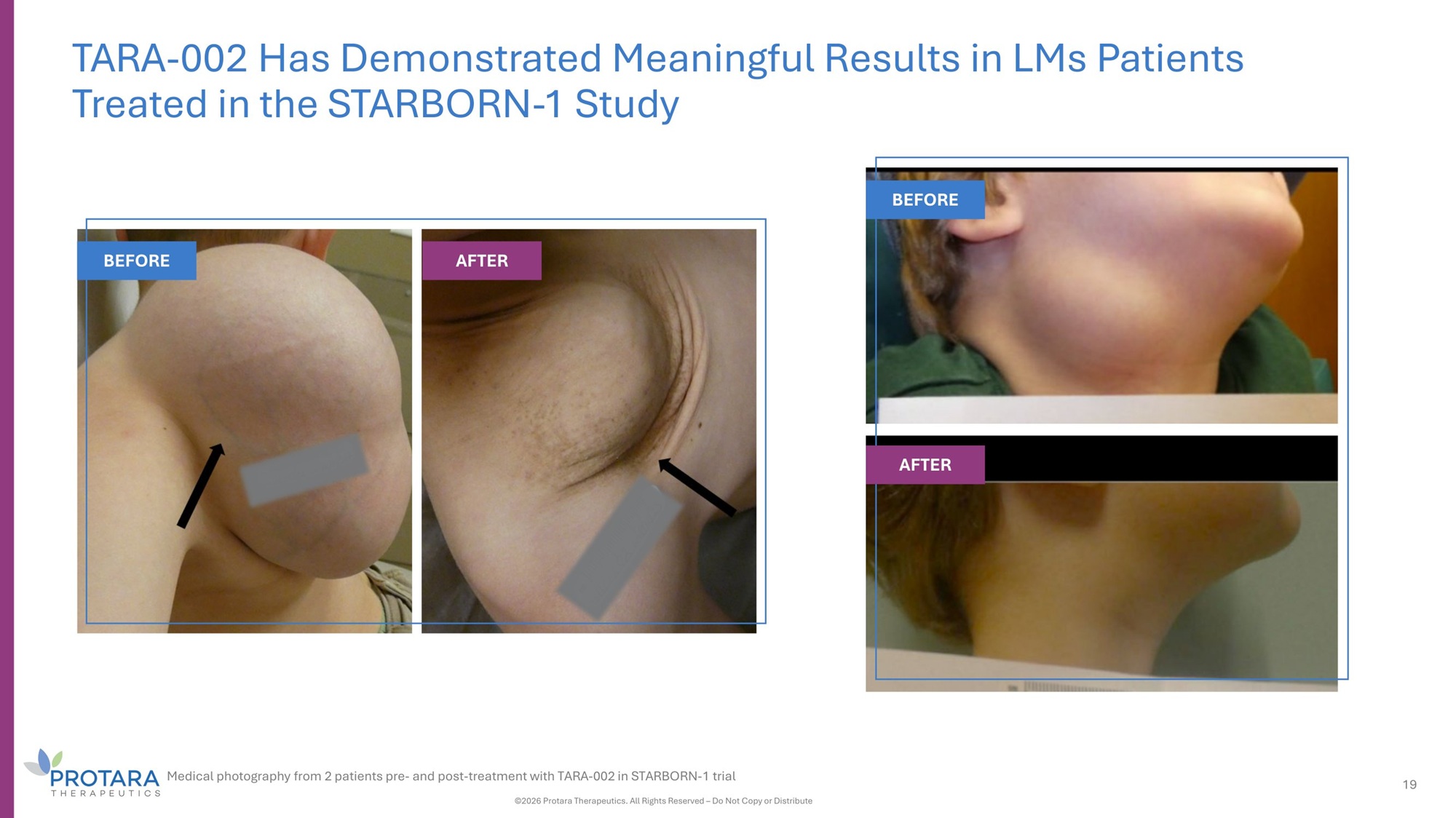
TARA - 002 Has Demonstrated Meaningful Results in LMs Patients Treated in the STARBORN - 1 Study BEFORE AFTER BEFORE AFTER Medical photography from 2 patients pre - and post - treatment with TARA - 002 in STARBORN - 1 trial ©2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute 19

TARA - 002 in LMs is a Late - Stage Opportunity with Significant Unmet Need STARBORN - 1 DATA INDICATE TARA - 002 HAS COMPARABLE SAFETY G EFFICACY TO OK - 432 • 100% of evaluable patients who received TARA - 002 achieved clinical success • TARA - 002 demonstrated favorable safety and tolerability profile with no serious adverse events reported • Clinical Success was achieved with one or two doses in 88% of patients • OK - 432, predecessor therapy to TARA - 002, has shown strong safety and efficacy results in 500+ U.S. pediatric LMs patients in University of Iowa - led study • OK - 432 approved in Japan as standard of care for LMs for 30 years HIGH UNMET NEED WITH POTENTIAL TO TREAT OTHER MAXILLOFACIAL CYSTS AND TO ACHIEVE RARE DISEASE PRICING • FDA Rare Pediatric Disease, Breakthrough Therapy and Fast Track Designations • FDA has invited us to apply to the Chemistry, Manufacturing, and Controls (CMC) Development and Readiness Pilot (CDRP) Program and we look forward to submitting a request to participate in this program • Historical literature and patient experience show TARA - 002 may also be effective in treating other maxillofacial cysts including ranula and thyroglossal duct cysts • LMs incidence/prevalence, unmet need and TARA - 002 product profile suggest rare disease pricing opportunity 20 ©2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute

IV Choline Chloride Phospholipid substrate replacement therapy for patients dependent on parenteral support (PS)
22 IV Choline Chloride is a Late - Stage Opportunity with High Unmet Medical Need HIGH UNMET NEED WITH LARGE MARKET OPPORTUNITY • ~30K patients on PS long term at home • 78% of PS - dependent patients are choline - deficient and 63% have some degree of liver damage 1 • Pricing analogues suggest significant market opportunity Has the potential to become the first approved IV choline formulation for PS patients POSITIVE CLINICAL DATA AND ALREADY INCLUDED IN MEDICAL GUIDELINES • Independently conducted Phase 2 data demonstrated significant improvement in serum choline concentrations and a pronounced impact on steatosis 2 • IV choline replacement for PS patients is included in US and EU medical guidelines, even though no therapy currently exists SINGLE PK STUDY REǪUIRED FOR REGULATORY APPROVAL • Single PK study demonstrating an increase in choline levels required for registration • FDA granted a targeted indication of source of choline when oral or enteral nutrition is not possible, insufficient or contra - indicated INTELLECTUAL PROPERTY PROTECTIONS • Both a compound patent and a method of treatment patent in U.S. to 2041 1 Micic D, Muhic E et al., JPEN J Parenter Enteral Nutr. 2025;49(6):e2733.; 2 Buchman A, et al. JPEN. 2001;5:260 - 268 ©2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute
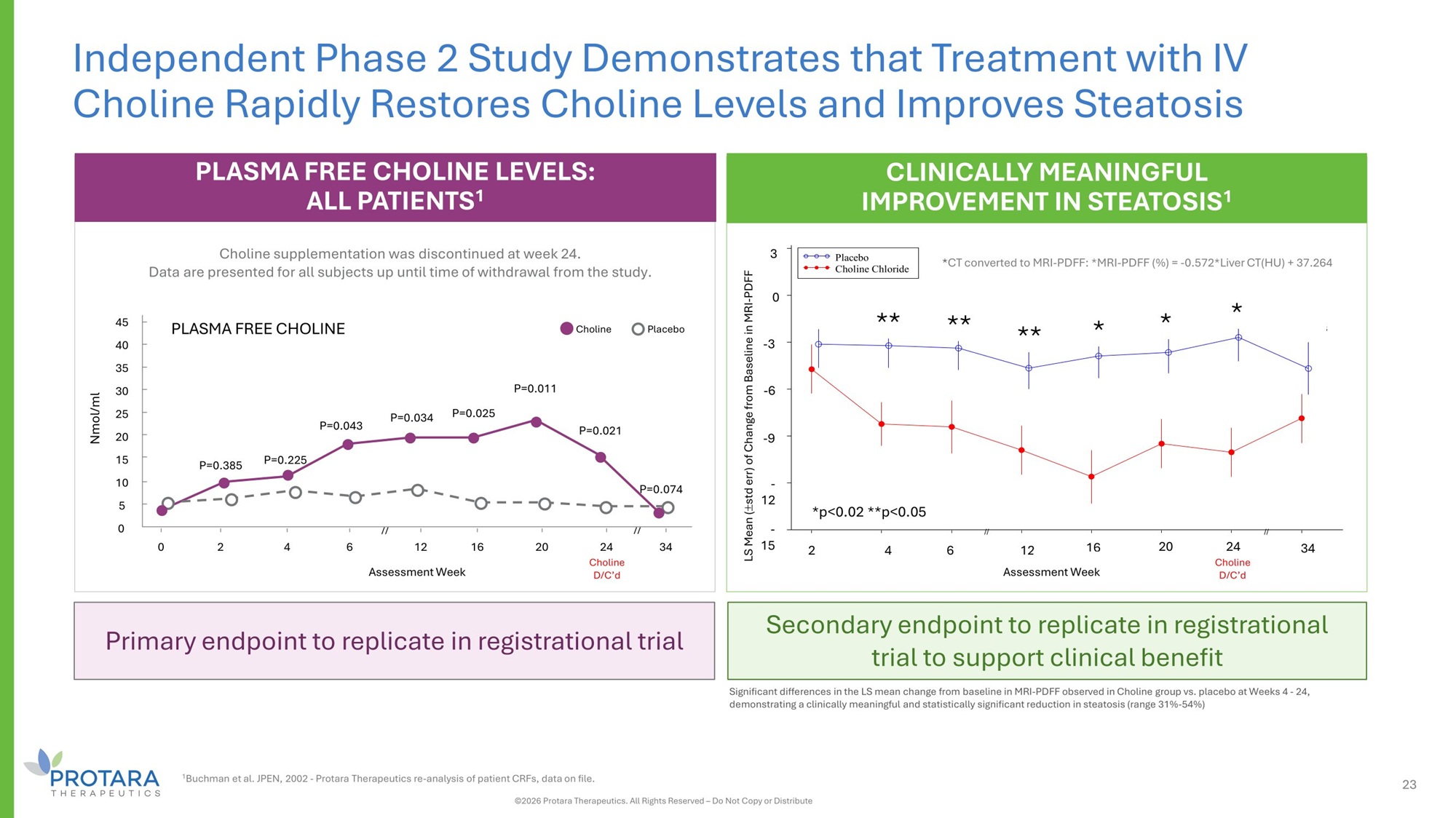
Significant differences in the LS mean change from baseline in MRI - PDFF observed in Choline group vs. placebo at Weeks 4 - 24, demonstrating a clinically meaningful and statistically significant reduction in steatosis (range 31% - 54%) PLASMA FREE CHOLINE LEVELS: ALL PATIENTS 1 Choline supplementation was discontinued at week 24. Data are presented for all subjects up until time of withdrawal from the study. PLASMA FREE CHOLINE Nmol/ml 0 2 4 6 12 Assessment Week 16 20 34 P=0.225 P=0.043 P=0.034 P=0.025 P=0.011 P=0.021 P=0.074 45 40 35 30 25 20 15 10 5 0 Choline // // P=0.385 Placebo 24 Choline D/C’d Primary endpoint to replicate in registrational trial Secondary endpoint to replicate in registrational trial to support clinical benefit Independent Phase 2 Study Demonstrates that Treatment with IV Choline Rapidly Restores Choline Levels and Improves Steatosis CLINICALLY MEANINGFUL IMPROVEMENT IN STEATOSIS 1 sses s ek C d LS Means stderr) hange Baseli (+/ - of c from ne of MRI - PDFF p= 0.4770 p= 0.0197 p= 0.0312 p= 0.0180 p= 0.0014 p= 0.0091 p= 0.0021 p= 0.1823 // // - 15 - 12 - 9 - 6 - 3 0 3 2 4 6 12 A 16 ment We 20 24 holine D/C' 34 Placebo Choline Chloride * * * ** ** ** - 3 3 0 - 6 - 9 - 12 - 15 *p<0.02 **p<0.05 2 4 6 12 16 20 34 * CT converted to MRI - PDFF: *MRI - PDFF (%) = - 0.572*Liver CT(HU) + 37.264 LS Mean ( std err) of Change from Baseline in MRI - PDFF Assessment Week 24 Choline D/C’d 23 ©2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute 1 Buchman et al. JPEN, 2002 - Protara Therapeutics re - analysis of patient CRFs, data on file.

24 Large Market Opportunity for IV Choline to Address a Clear, Guideline Supported Unmet Medical Need in PS US Patients requiring PS >110,000 per year Patients on long term PS at home ~30,000 ASPEN (US) and ESPEN (EU) guidelines recommend choline for patients on PS, yet no product is available to meet this need. HCPs expect to prescribe IV Choline to the majority of their long - term PS patients, driven by IV Choline’s expected efficacy and ASPEN recommendation IV Choline Market Research – Intent to treat IV Choline Commercial Analogs Market Research and Claims Analysis ©2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute

Appendix

TARA - 002 BCG Cytokines + -- - IFN - γ +++ + TNF - α + = IL - 12p70 - = IL - 8 + + IL - 6 +++ +++ IL - 1β + = IL - 10 + + IL - 4 = = IL - 13 - - - - IL - 2 26 TARA - 002 Demonstrates Differentiated Profile to BCG = : No change + : 2 - 5 fold upregulation +++ : ≥ 15 - fold upregulation - : 2 - 5 fold downregulation -- : 5 - 14 - fold downregulation --- : ≥ 15 - fold downregulation TARA - 002 treatment promotes differential pro - inflammatory TH1 - type cytokines than BCG in co - culture 1 TARA - 002 Potent Immune Activation with a Distinct Cytokine Signature • Distinct cytokine profile marked by strong TH1 activation (↑ IFN - γ, TNF - α, IL - 12p70), defining an immune signature different from BCG • Selectively downregulates IL - 8, a cytokine linked to tumor recurrence and progression in NMIBC ©2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute 1 Internal pre - clinical data.
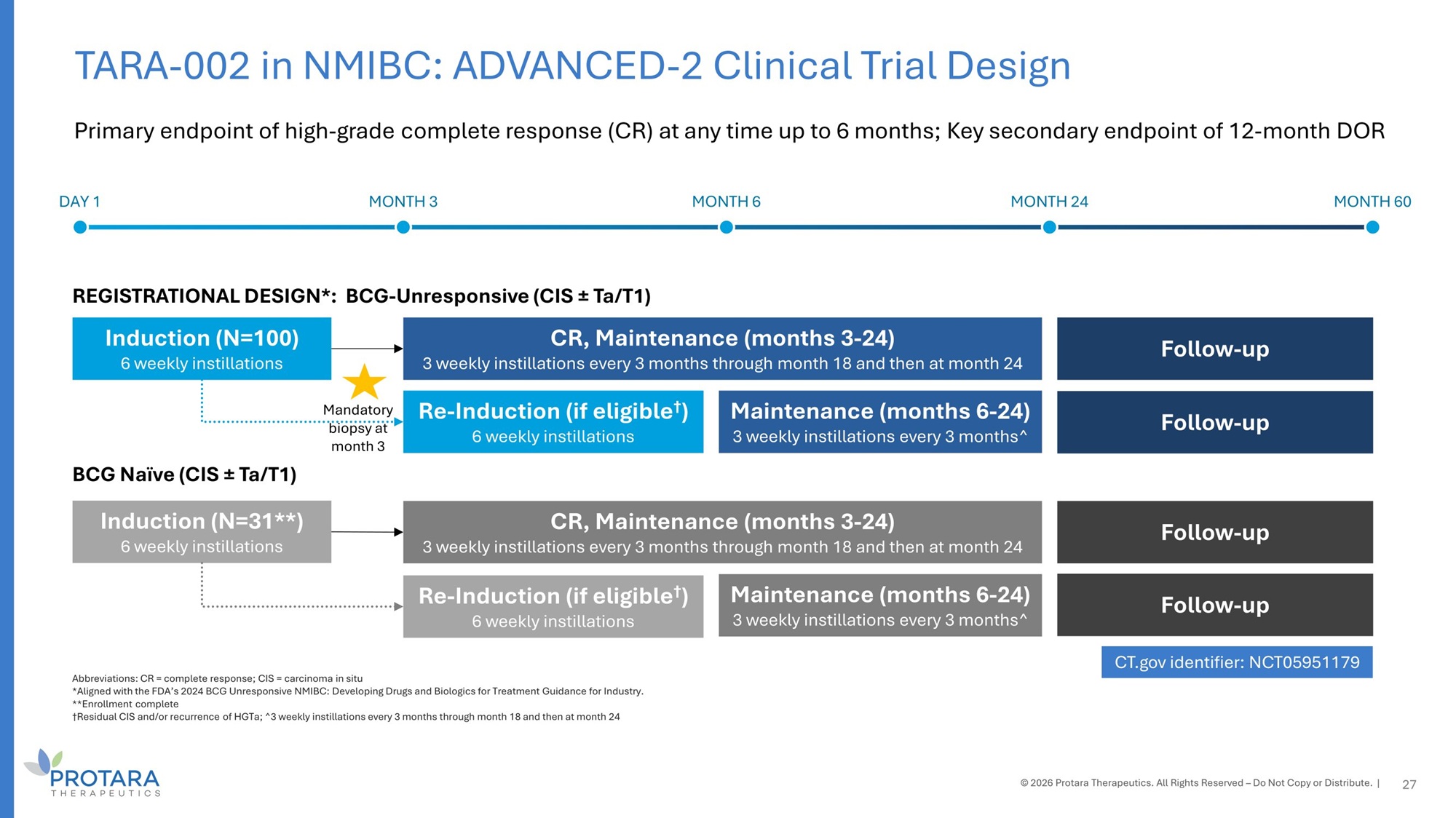
Abbreviations: CR = complete response; CIS = carcinoma in situ *Aligned with the FDA’s 2024 BCG Unresponsive NMIBC: Developing Drugs and Biologics for Treatment Guidance for Industry. **Enrollment complete †Residual CIS and/or recurrence of HGTa; ^3 weekly instillations every 3 months through month 18 and then at month 24 Primary endpoint of high - grade complete response (CR) at any time up to 6 months; Key secondary endpoint of 12 - month DOR 27 DAY 1 MONTH 3 MONTH 6 MONTH 24 MONTH 60 REGISTRATIONAL DESIGN*: BCG - Unresponsive (CIS “ Ta/T1) BCG Naïve (CIS “ Ta/T1) Induction (N=100) 6 weekly instillations Induction (N=31**) 6 weekly instillations CR, Maintenance (months 3 - 24) 3 weekly instillations every 3 months through month 18 and then at month 24 Follow - up Follow - up CR, Maintenance (months 3 - 24) 3 weekly instillations every 3 months through month 18 and then at month 24 Maintenance (months 6 - 24) 3 weekly instillations every 3 months^ Maintenance (months 6 - 24) 3 weekly instillations every 3 months^ Re - Induction (if eligible † ) 6 weekly instillations Re - Induction (if eligible † ) 6 weekly instillations Follow - up Follow - up CT.gov identifier: NCT05951179 © 2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute. | TARA - 002 in NMIBC: ADVANCED - 2 Clinical Trial Design Mandatory biopsy at month 3
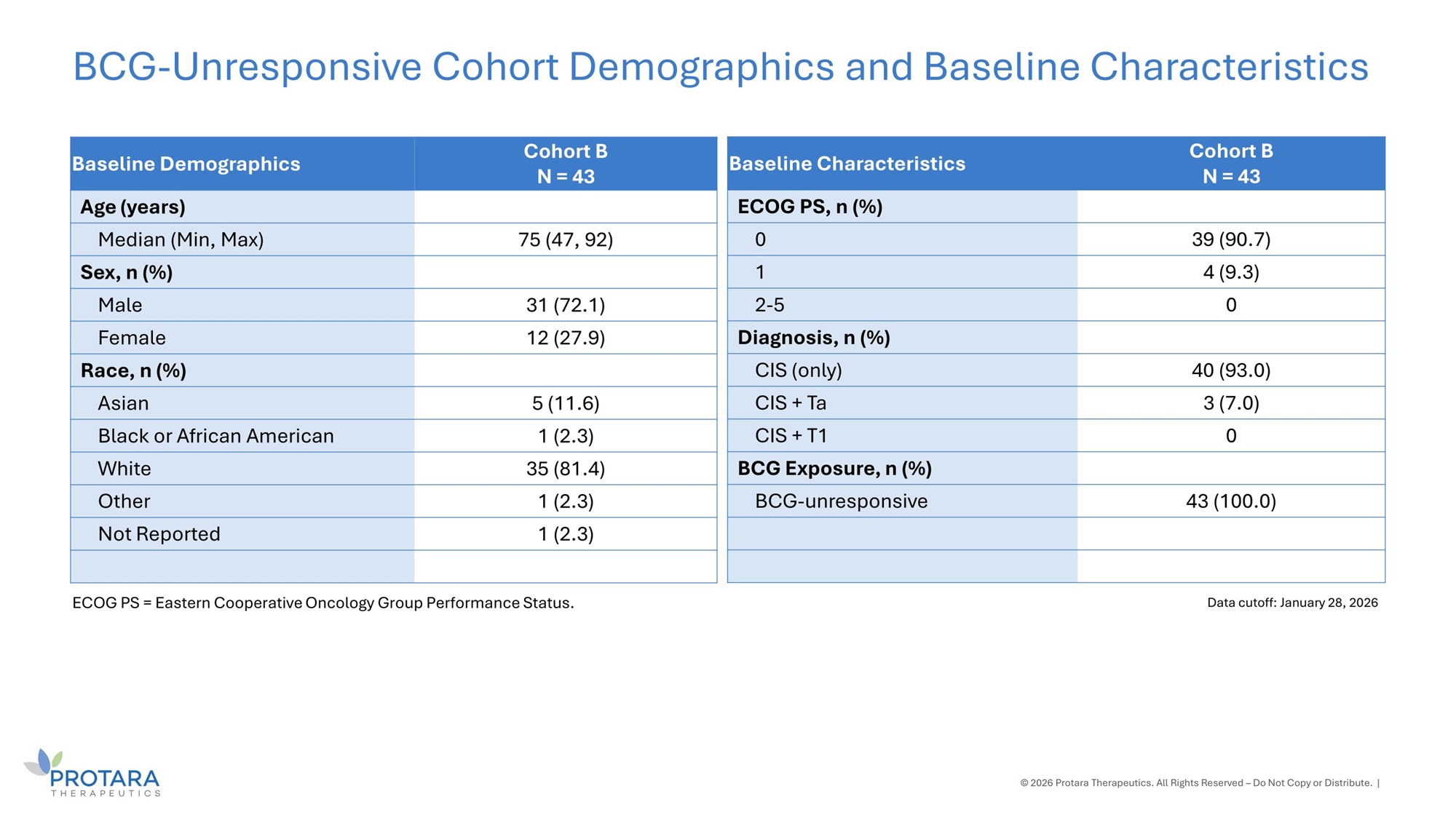
Cohort B N = 43 Baseline Demographics Age (years) 75 (47, 92) Median (Min, Max) Sex, n (%) 31 (72.1) Male 12 (27.9) Female Race, n (%) 5 (11.6) Asian 1 (2.3) Black or African American 35 (81.4) White 1 (2.3) Other 1 (2.3) Not Reported © 2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute. | BCG - Unresponsive Cohort Demographics and Baseline Characteristics Data cutoff: January 28, 2026 ECOG PS = Eastern Cooperative Oncology Group Performance Status. Cohort B N = 43 Baseline Characteristics ECOG PS, n (%) 39 (90.7) 0 4 (9.3) 1 0 2 - 5 Diagnosis, n (%) 40 (93.0) CIS (only) 3 (7.0) CIS + Ta 0 CIS + T1 BCG Exposure, n (%) 43 (100.0) BCG - unresponsive
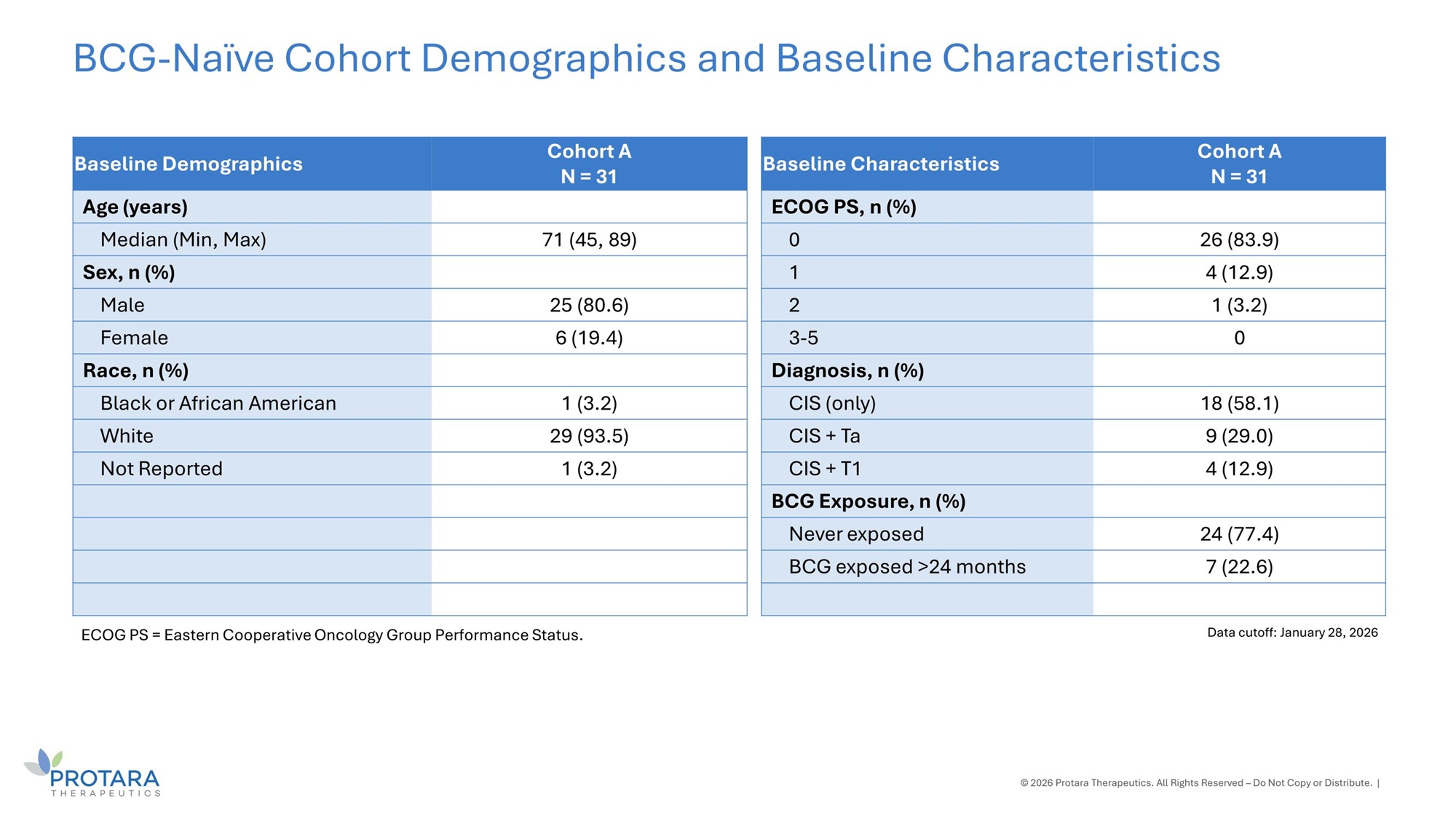
Cohort A N = 31 Baseline Demographics Age (years) 71 (45, 89) Median (Min, Max) Sex, n (%) 25 (80.6) Male 6 (19.4) Female Race, n (%) 1 (3.2) Black or African American 29 (93.5) White 1 (3.2) Not Reported © 2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute. | BCG - Naïve Cohort Demographics and Baseline Characteristics Data cutoff: January 28, 2026 ECOG PS = Eastern Cooperative Oncology Group Performance Status. Cohort A N = 31 Baseline Characteristics ECOG PS, n (%) 26 (83.9) 0 4 (12.9) 1 1 (3.2) 2 0 3 - 5 Diagnosis, n (%) 18 (58.1) CIS (only) 9 (29.0) CIS + Ta 4 (12.9) CIS + T1 BCG Exposure, n (%) 24 (77.4) Never exposed 7 (22.6) BCG exposed >24 months
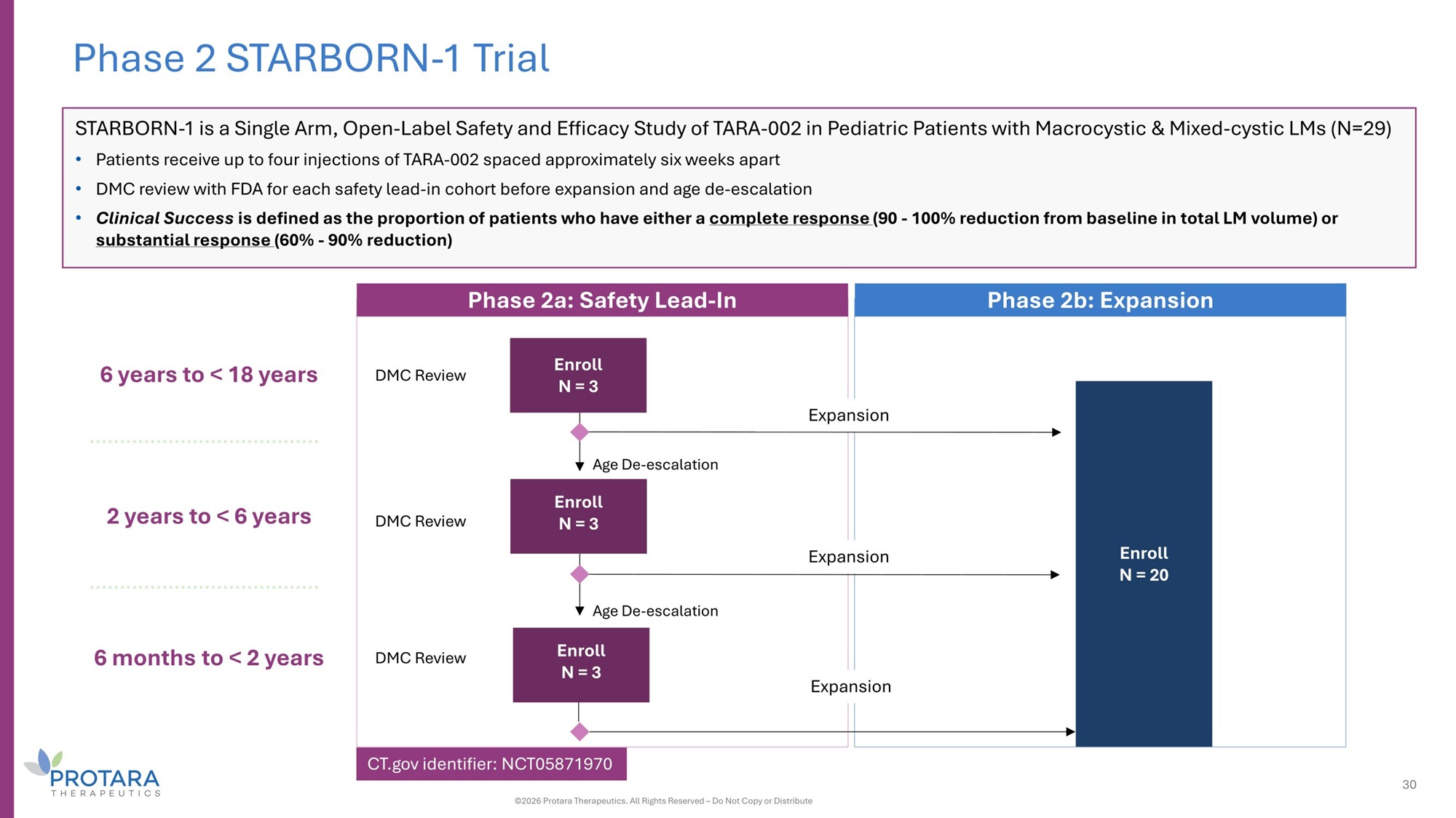
6 years to < 18 years 2 years to < 6 years 6 months to < 2 years Phase 2a: Safety Lead - In Phase 2b: Expansion Age De - escalation Age De - escalation DMC Review DMC Review DMC Review Phase 2 STARBORN - 1 Trial Enroll N = 20 Expansion Expansion Expansion Enroll N = 3 Enroll N = 3 Enroll N = 3 STARBORN - 1 is a Single Arm, Open - Label Safety and Efficacy Study of TARA - 002 in Pediatric Patients with Macrocystic C Mixed - cystic LMs (N=29) • Patients receive up to four injections of TARA - 002 spaced approximately six weeks apart • DMC review with FDA for each safety lead - in cohort before expansion and age de - escalation • Clinical Success is defined as the proportion of patients who have either a complete response (G0 - 100% reduction from baseline in total LM volume) or substantial response (60% - G0% reduction) 30 ©2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute CT.gov identifier: NCT05871970
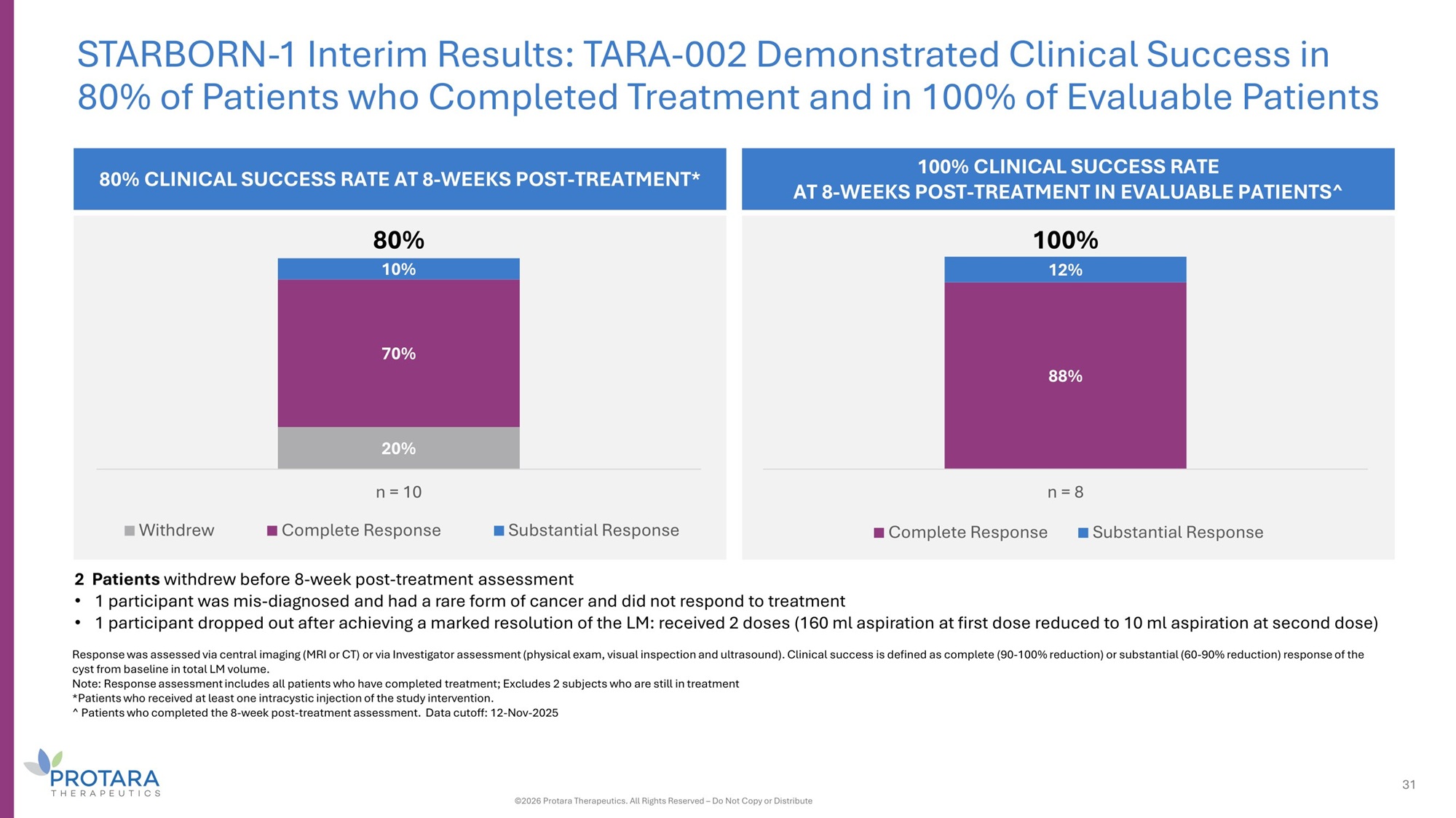
2 Patients withdrew before 8 - week post - treatment assessment • 1 participant was mis - diagnosed and had a rare form of cancer and did not respond to treatment • 1 participant dropped out after achieving a marked resolution of the LM: received 2 doses (160 ml aspiration at first dose reduced to 10 ml aspiration at second dose) Response was assessed via central imaging (MRI or CT) or via Investigator assessment (physical exam, visual inspection and ultrasound). Clinical success is defined as complete (90 - 100% reduction) or substantial (60 - 90% reduction) response of the cyst from baseline in total LM volume. Note: Response assessment includes all patients who have completed treatment; Excludes 2 subjects who are still in treatment *Patients who received at least one intracystic injection of the study intervention. ^ Patients who completed the 8 - week post - treatment assessment. Data cutoff: 12 - Nov - 2025 20% 70% 10% Withdrew n = 10 Complete Response Substantial Response 80% 80% CLINICAL SUCCESS RATE AT 8 - WEEKS POST - TREATMENT* STARBORN - 1 Interim Results: TARA - 002 Demonstrated Clinical Success in 80% of Patients who Completed Treatment and in 100% of Evaluable Patients 100% CLINICAL SUCCESS RATE AT 8 - WEEKS POST - TREATMENT IN EVALUABLE PATIENTS^ 88% 12% n = 8 Complete Response Substantial Response 31 ©2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute 100%
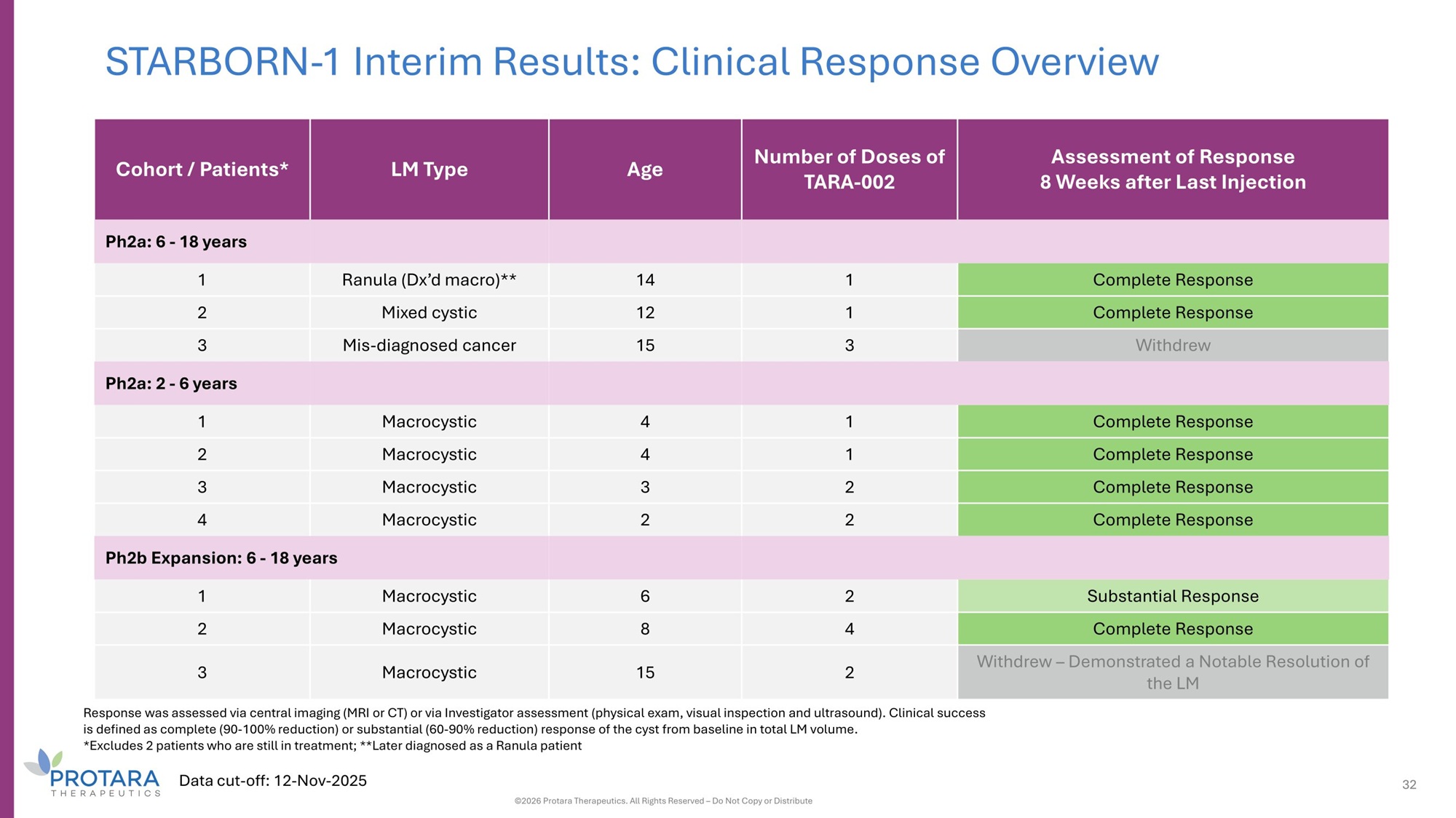
STARBORN - 1 Interim Results: Clinical Response Overview Assessment of Response 8 Weeks after Last Injection Number of Doses of TARA - 002 Age LM Type Cohort / Patients* Ph2a: 6 - 18 years Complete Response 1 14 Ranula (Dx’d macro)** 1 Complete Response 1 12 Mixed cystic 2 Withdrew 3 15 Mis - diagnosed cancer 3 Ph2a: 2 - 6 years Complete Response 1 4 Macrocystic 1 Complete Response 1 4 Macrocystic 2 Complete Response 2 3 Macrocystic 3 Complete Response 2 2 Macrocystic 4 Ph2b Expansion: 6 - 18 years Substantial Response 2 6 Macrocystic 1 Complete Response 4 8 Macrocystic 2 Withdrew – Demonstrated a Notable Resolution of the LM 2 15 Macrocystic 3 Response was assessed via central imaging (MRI or CT) or via Investigator assessment (physical exam, visual inspection and ultrasound). Clinical success is defined as complete (90 - 100% reduction) or substantial (60 - 90% reduction) response of the cyst from baseline in total LM volume. *Excludes 2 patients who are still in treatment; **Later diagnosed as a Ranula patient Data cut - off: 12 - Nov - 2025 32 ©2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute
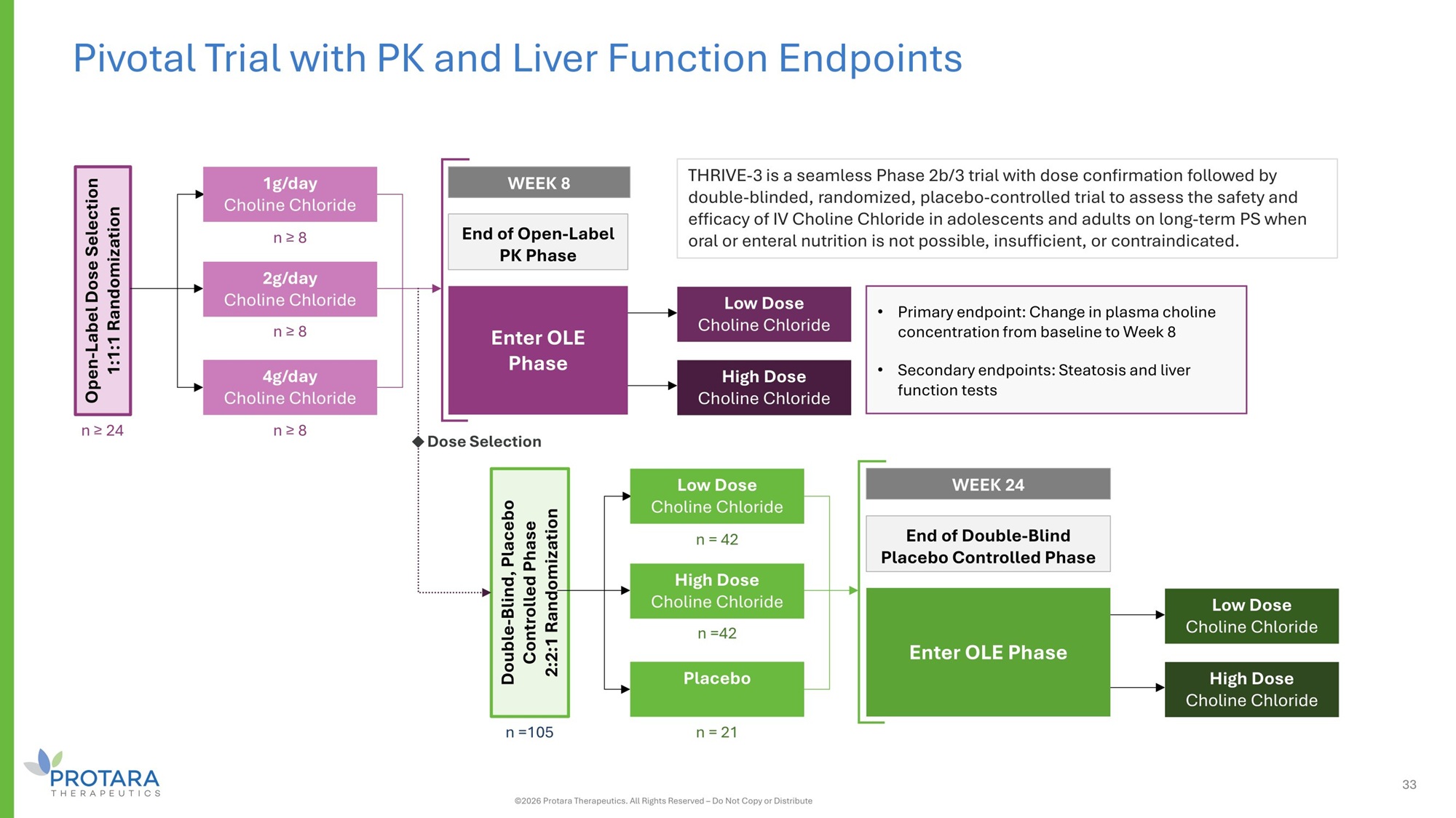
THRIVE - 3 is a seamless Phase 2b/3 trial with dose confirmation followed by double - blinded, randomized, placebo - controlled trial to assess the safety and efficacy of IV Choline Chloride in adolescents and adults on long - term PS when oral or enteral nutrition is not possible, insufficient, or contraindicated. Pivotal Trial with PK and Liver Function Endpoints • Primary endpoint: Change in plasma choline concentration from baseline to Week 8 • Secondary endpoints: Steatosis and liver function tests n ≥ 8 n ≥ 8 n ≥ 8 Open - Label Dose Selection 1:1:1 Randomization n ≥ 24 End of Open - Label PK Phase WEEK 8 1g/day Choline Chloride 2g/day Choline Chloride 4g/day Choline Chloride Enter OLE Phase Low Dose Choline Chloride High Dose Choline Chloride n = 42 n =42 n = 21 Double - Blind, Placebo Controlled Phase 2:2:1 Randomization n =105 End of Double - Blind Placebo Controlled Phase WEEK 24 Low Dose Choline Chloride High Dose Choline Chloride Placebo Enter OLE Phase Low Dose Choline Chloride High Dose Choline Chloride Dose Selection 33 ©2026 Protara Therapeutics. All Rights Reserved – Do Not Copy or Distribute