
1 February 26, 2026 Q4 2025 & Full Year Financial Results Our Mission: Protect and Save Lives

2 Safe Harbor Statement/Trademarks 2 This presentation includes forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995, as amended. All statements, other than statements of historical fact, including statements regarding the future performance of the Company or any of our businesses, our business strategy, future operations, future financial position, future revenues and earnings, our ability to achieve the objectives of our restructuring initiatives, acquisitions and divestitures, including our future results, projected costs, prospects, plans and objectives of management, are forward-looking statements. We generally identify forward-looking statements by using words like “anticipate,” “believe,” “can,” “continue,” “could,” “estimate,” “expect,” “confident,” “commit,” “forecast,” “future,” “outlook,” “goal,” “intend,” “may,” “plan,” “position,” “possible,” “potential,” “predict,” “project,” “should,” “target,” “will,” “would,” and similar expressions or variations thereof, or the negative thereof, but these terms are not the exclusive means of identifying such statements. These forward-looking statements are based on our current intentions, beliefs, assumptions and expectations regarding future events based on information that is currently available. You should realize that if underlying assumptions prove inaccurate or unknown risks or uncertainties materialize, actual results could differ materially from our expectations. Readers are, therefore, cautioned not to place undue reliance on any forward- looking statement contained herein. Any such forward-looking statement speaks only as of the date of this press release, and, except as required by law, we do not undertake any obligation to update any forward-looking statement to reflect new information, events or circumstances. There are a number of important factors that could cause our actual results to differ materially from those indicated by such forward-looking statements, including, among others, the availability of USG funding for contracts related to procurement of our medical countermeasures (“MCM”) products, including CYFENDUS® (Anthrax Vaccine Adsorbed (AVA) Adjuvanted), previously known as AV7909, ACAM2000® (Smallpox (Vaccinia) Vaccine, Live), CNJ-016® (Vaccinia Immune Globulin Intravenous (Human) (VIGIV)), BAT® (Botulism Antitoxin Heptavalent (A,B,C,D,E,F,G)-(Equine)), BioThrax® (Anthrax Vaccine Adsorbed) EbangaTM (ansuvimab-zykl) and/or TEMBEXA® (brincidofovir) among others, as well as contracts related to development of medical countermeasures; our ability to meet our commitments to quality and compliance in all of our manufacturing operations; our ability to negotiate additional USG procurement or follow-on contracts for our MCM products that have expired or will be expiring; the commercial availability and impact of a generic and competitive marketplace on future sales of NARCAN® (naloxone HCL) Nasal Spray, over-the-counter NARCAN® Nasal Spray and KLOXXADO® Nasal Spray; our ability to perform under our contracts with the USG, including the timing of and specifications relating to deliveries; the ability of our contractors and suppliers to maintain compliance with current good manufacturing practices and other regulatory obligations; our ability to collect reimbursement for raw materials and payment of service fees from our Bioservices customers; the results of pending government investigations and their potential impact on our business; our ability to satisfy the conditions of our litigation settlement agreements, and the potential impact of such agreements, including the funds to resolve related litigation, on our business; our ability to comply with the operating and financial covenants required by (i) our term loan facility under a credit agreement, dated August 30, 2024, among the Company, the lenders from time to time party thereto and OHA Agency LLC, as administrative agent, (ii) our revolving credit facility under a credit agreement, dated September 30, 2024, among the Company, certain subsidiary borrowers, the lenders from time to time party thereto and Wells Fargo, National Association, as Agent, and (iii) our 3.875% Senior Unsecured Notes due 2028; our ability to maintain adequate internal control over financial reporting and to prepare accurate financial statements in a timely manner; our ability to maintain sufficient cash flow from our operations to pay our substantial debt, both now and in the future; our ability to invest in our business operations as a result of our current indebtedness; the impact of our share and debt repurchase programs; the procurement of our product candidates by USG entities under regulatory authorities that permit government procurement of certain medical products prior to FDA marketing authorization, and corresponding procurement by government entities outside the United States; the success of our commercialization, marketing and manufacturing capabilities and strategy; our ability to identify and acquire companies, businesses, products or product candidates that satisfy our selection criteria; our ability to attract and retain qualified personnel; our ability to adequately secure and protect our intellectual property rights; the impact of cybersecurity incidents, including the risks from the unauthorized access, interruption, failure or compromise of our information systems or those of our business partners, collaborators or other third parties; and the accuracy of our estimates regarding future revenues, expenses, capital requirements and need for additional financing. The foregoing sets forth many, but not all, of the factors that could cause actual results to differ materially from our expectations in any forward- looking statement. In addition, other risks and uncertainties not presently known to us or that we currently believe to be immaterial could affect the accuracy of any forward-looking statements. Readers should consider this cautionary statement, as well as the risks identified in our periodic reports filed with the Securities and Exchange Commission, when evaluating our forward-looking statements. Trademarks: Emergent®, BioThrax®, BaciThrax®, BAT®, Trobigard®, ANTHRASIL®, CNJ-016®, ACAM2000®, NARCAN®, CYFENDUS®, TEMBEXA® and any and all Emergent BioSolutions Inc. brands, products, services and feature names, logos and slogans are trademarks or registered trademarks of Emergent BioSolutions Inc. or its subsidiaries in the United States or other countries. All other brands, products, services and feature names or trademarks are the property of their respective owners, including KLOXXADO®, which is a registered trademark of Hikma Pharmaceuticals USA Inc.

3 Today’s Agenda Joe Papa President and CEO Rich Lindahl EVP, Chief Financial Officer Joe Papa President and CEO Multi-Year Transformation Plan 2025 Business Performance & Key Highlights Q4 & FY 2025 Financial Results FY 2026 Guidance 2026 Growth Catalysts Q&A Session TopicPresenter

4 Multi-Year Transformation Plan Joe Papa President and Chief Executive Officer
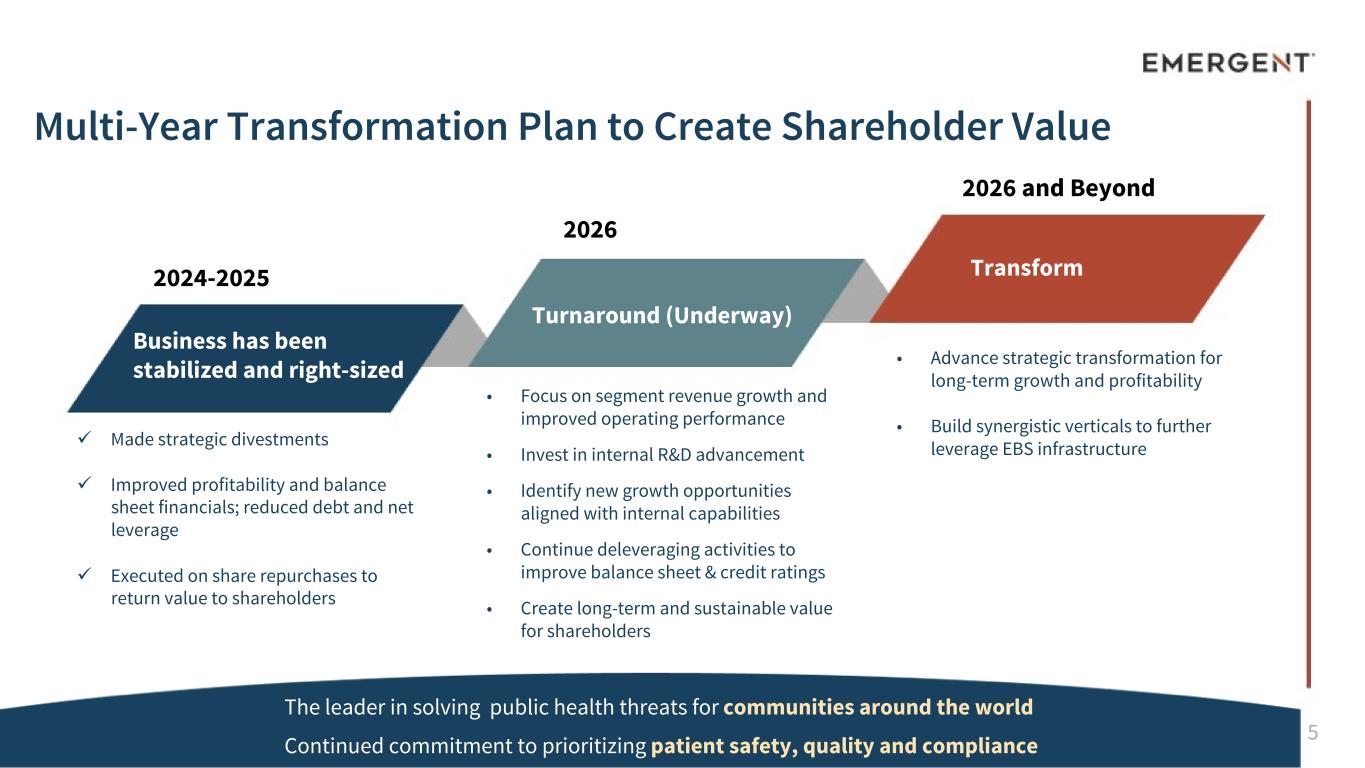
5 • Focus on segment revenue growth and improved operating performance • Invest in internal R&D advancement • Identify new growth opportunities aligned with internal capabilities • Continue deleveraging activities to improve balance sheet & credit ratings • Create long-term and sustainable value for shareholders ü Made strategic divestments ü Improved profitability and balance sheet financials; reduced debt and net leverage ü Executed on share repurchases to return value to shareholders • Advance strategic transformation for long-term growth and profitability • Build synergistic verticals to further leverage EBS infrastructure Business has been stabilized and right-sized Turnaround (Underway) Transform2024-2025 2026 2026 and Beyond Multi-Year Transformation Plan to Create Shareholder Value The leader in solving public health threats for communities around the world Continued commitment to prioritizing patient safety, quality and compliance

6 2025 Business Performance & Key Highlights Joe Papa President and Chief Executive Officer 6

77 ü Solid MCM performance reflects increased global demand and strategic diversification beyond the U.S. • 11+ contract awards and product orders received • Int'l MCM sales represent 34% of total MCM revenue in 2025 • Continued engagement with U.S. and allied governments ü Continued market leadership across naloxone category • Maintained market leadership; pricing stabilized • Differentiated commercial capabilities and innovative line extensions • 100+ million doses of NARCAN® Nasal Spray distributed across all channels in the U.S. and Canada since 2016 ü YTD activities support growth initiatives • Incremental investment in R&D and international growth • KLOXXADO® Nasal Spray 8 mg (U.S. and CAN commercial rights) • Swiss Rockets/Rocketvax investment ü FY 2025 Revenues of $743M with strong MCM sales while managing NARCAN® Q4 impact of prolonged U.S. government shutdown on public interest customers ü FY 2025 Adjusted EBITDA1 $205M, 28% margin, on high end of FY guidance range ü Improved cash balance to $205M, and liquidity of $305M increased +$105M vs. prior year with strong net working capital management • Strong cash position enabled $110M debt paydown • Strong YoY growth in Operating Cash Flow; grew $112M to $171M in 2025 ü Continued capital allocation to create value • Net leverage1 is 1.9x Adjusted EBITDA1, down from 3.3x in Q4 2024 • Repurchased a portion of EBS' unsecured bonds • Paid down $100M of the Term Loan • Continued share repurchases through Q4 1. See "End Notes: Non-GAAP Financial Measures" and "Appendix" for the definitions of non-GAAP terms and reconciliations to the most directly comparable GAAP financial measures. Strong Financial Performance Solid Business Impact & Growth Initiatives Strong Q4 & Full Year Performance on Both Top and Bottom Line

MCM Business: Supporting U.S. and International Governments' Biodefense Readiness and Supply Resilience Consistent and ongoing engagement with U.S. and allied governments International orders represent 34% of total MCM revenues in 2025 11+ MCM contract awards and product orders received NEW in Q1 2026 • $140M CAD in multi-product agreements in Canada • Up to $21.5M delivery order to supply BioThrax® (Anthrax Vaccine Adsorbed) to the USG 8

9 Naloxone Business: Broader Access, Awareness and Availability Helps Save Lives • Market-leading offerings, e.g. NARCANDirect®; platform features KLOXXADO® Nasal Spray 8 mg, Wall Units and Convenience Kits • Continued market leadership in U.S. public interest and Canada • Expanding consumer access through retail- enabling product availability and new lines/kits • Leadership and public awareness through Ready to Rescue campaign • FDA approved OTC NARCAN® Nasal Spray Carrying Case and Multipacks • Enabling workplace safety initiatives by securing 8 NARCAN® exclusive B2B distributors; expanding NARCANWorkplace.com • Executive Order: illicit fentanyl designated Weapon of Mass Destruction; launched the Great American Recovery initiative to address the addiction crisis1 • American Heart Association: updated guidelines include algorithm for treating individuals with naloxone in opioid overdose emergencies2 • New York: federally mandated first-aid supplies must include opioid antagonist3 1. White House Memo, accessed February 23, 2026; https://www.whitehouse.gov/presidential-actions/2025/12/designating-fentanyl-as-a-weapon-of-mass-destruction/ 2. American Heart Association Newsroom, accessed February 23, 2026; https://newsroom.heart.org/news/updated-cpr-guidelines-tackle-choking-response-opioid-related-emergencies-and-a-revised-chain-of-survival 3. NY A02725 Bill, accessed February 23, 2026; https://www.billtrack50.com/billdetail/1797019#:~:text=This%20bill%20requires%20employers%20who,where%20such%20emergencies%20might%20occur. OTC NARCAN® Nasal Spray 4 mg continues to be a standard of care NEW in Q1 2026Key stakeholders and actions drive naloxone access and availability

10 Q4 2025 & FY Financial Results Rich Lindahl EVP, Chief Financial Officer
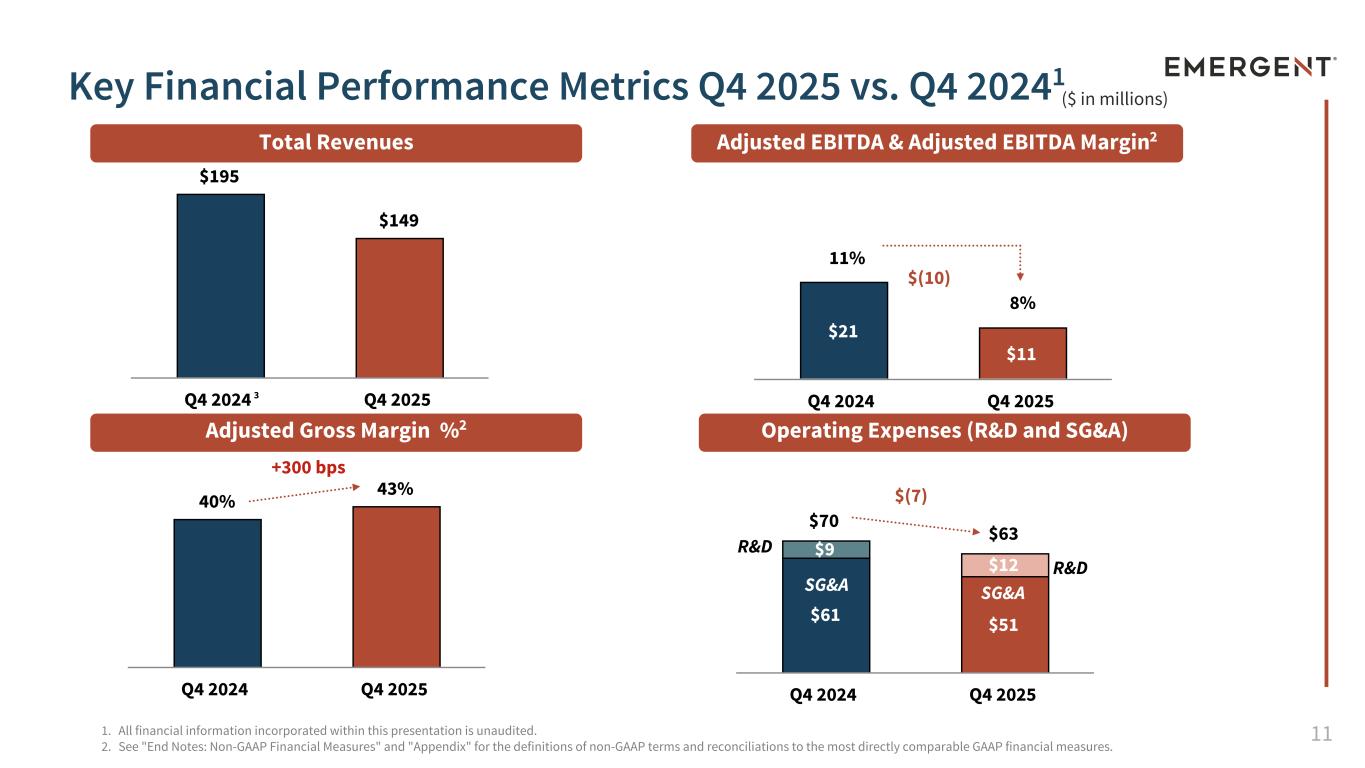
11 $70 $63 $61 $51 $9 $12 Q4 2024 Q4 2025 40% 43% Q4 2024 Q4 2025 $195 $149 Q4 2024 Q4 2025 $21 $11 Q4 2024 Q4 2025 ($ in millions) 1. All financial information incorporated within this presentation is unaudited. 2. See "End Notes: Non-GAAP Financial Measures" and "Appendix" for the definitions of non-GAAP terms and reconciliations to the most directly comparable GAAP financial measures. Total Revenues Adjusted EBITDA & Adjusted EBITDA Margin2 Adjusted Gross Margin %2 Operating Expenses (R&D and SG&A) SG&A SG&A Key Financial Performance Metrics Q4 2025 vs. Q4 20241 R&D SG&A R&D +300 bps 3 $(10) $(7) 8% 11%
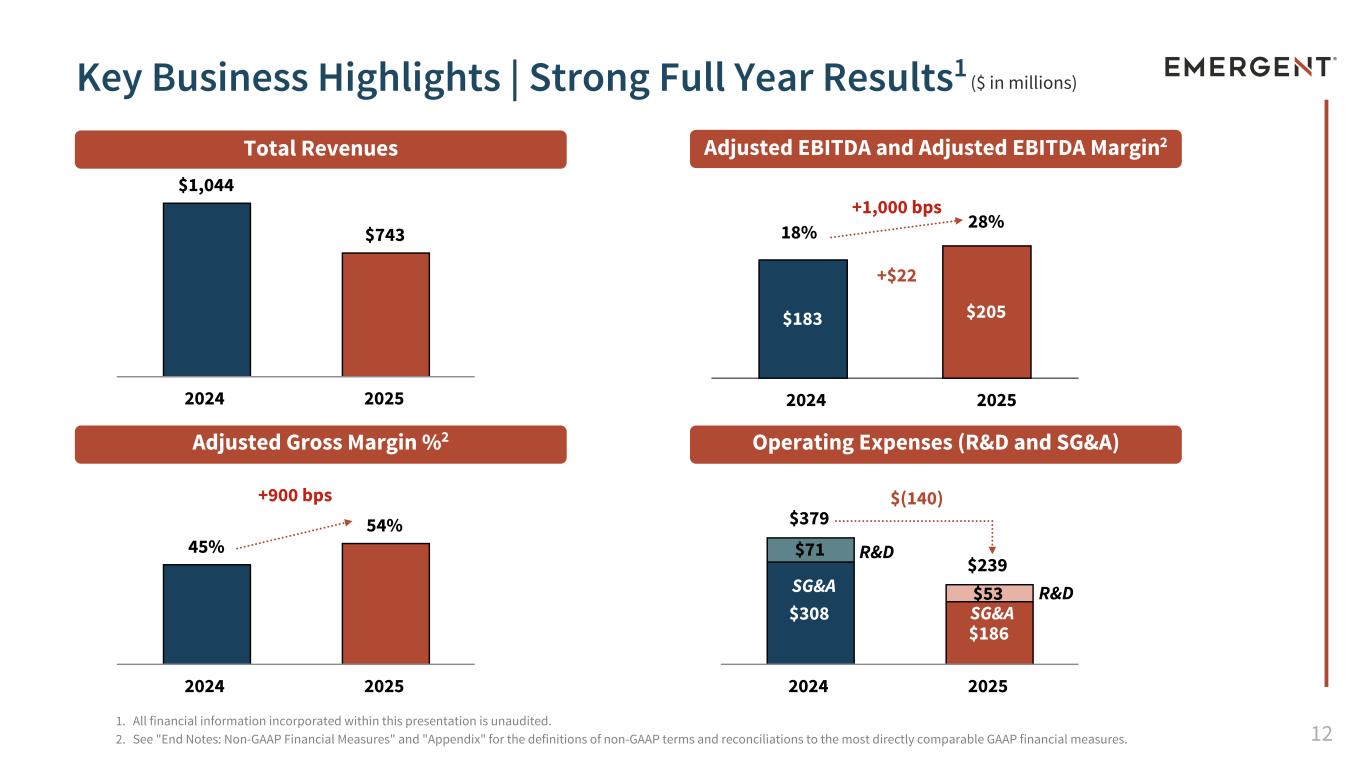
12 $183 $205 $379 $239 $308 $186 $71 $53 2024 2025 45% 54% 2024 2025 $1,044 $743 2024 2025 ($ in millions) 1. All financial information incorporated within this presentation is unaudited. 2. See "End Notes: Non-GAAP Financial Measures" and "Appendix" for the definitions of non-GAAP terms and reconciliations to the most directly comparable GAAP financial measures. SG&A SG&A R&D Key Business Highlights | Strong Full Year Results1 2024 2025 R&D $(140) +1,000 bps +$22 +900 bps 18% 28% Total Revenues Adjusted EBITDA and Adjusted EBITDA Margin2 Adjusted Gross Margin %2 Operating Expenses (R&D and SG&A)
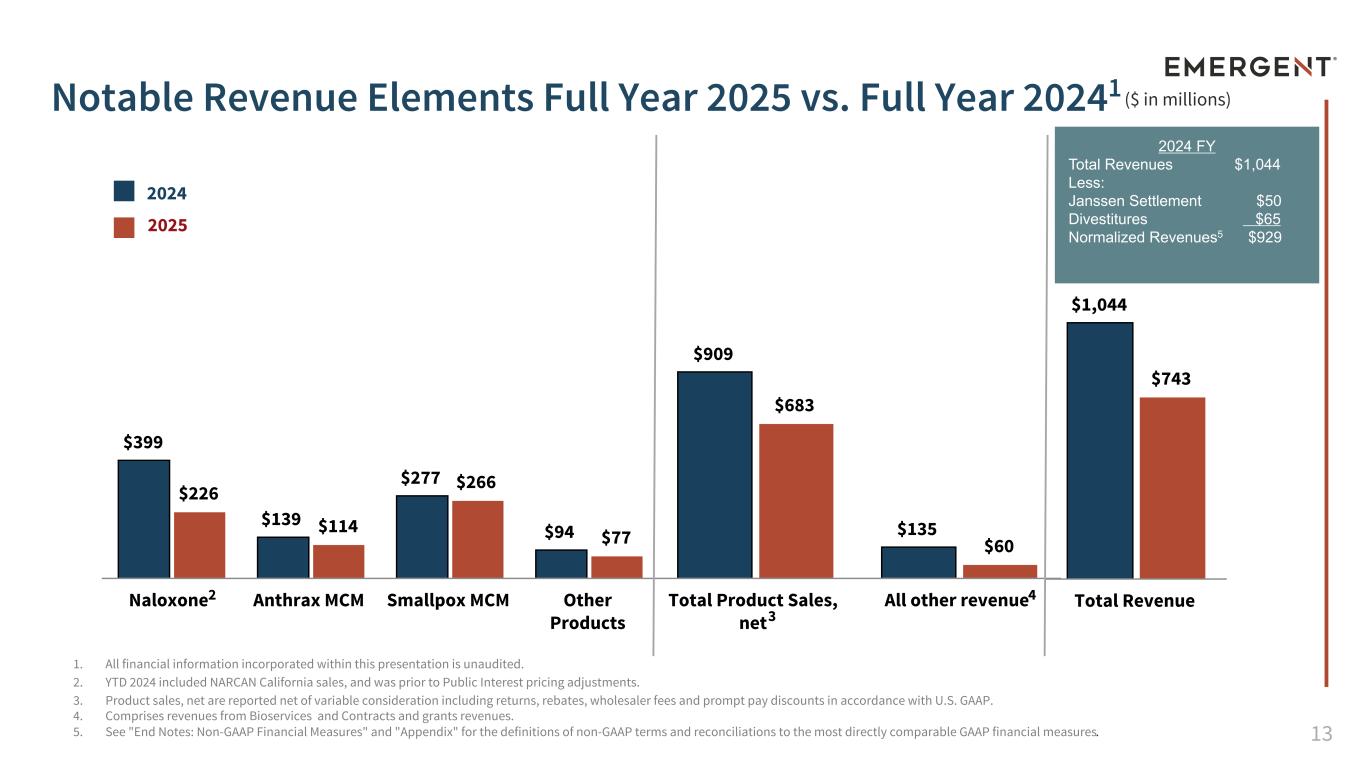
13 $1,044 $743 Total Revenue $909 $135 $683 $60 Total Product Sales, net All other revenue 1. All financial information incorporated within this presentation is unaudited. 2. YTD 2024 included NARCAN California sales, and was prior to Public Interest pricing adjustments. 3. Product sales, net are reported net of variable consideration including returns, rebates, wholesaler fees and prompt pay discounts in accordance with U.S. GAAP. 4. Comprises revenues from Bioservices and Contracts and grants revenues. 5. See "End Notes: Non-GAAP Financial Measures" and "Appendix" for the definitions of non-GAAP terms and reconciliations to the most directly comparable GAAP financial measures. Notable Revenue Elements Full Year 2025 vs. Full Year 20241 $399 $139 $277 $94 $226 $114 $266 $77 Naloxone Anthrax MCM Smallpox MCM Other Products 2024 2025 ($ in millions) 3 2 4 2024 FY Total Revenues $1,044 Less: Janssen Settlement $50 Divestitures $65 Normalized Revenues5 $929
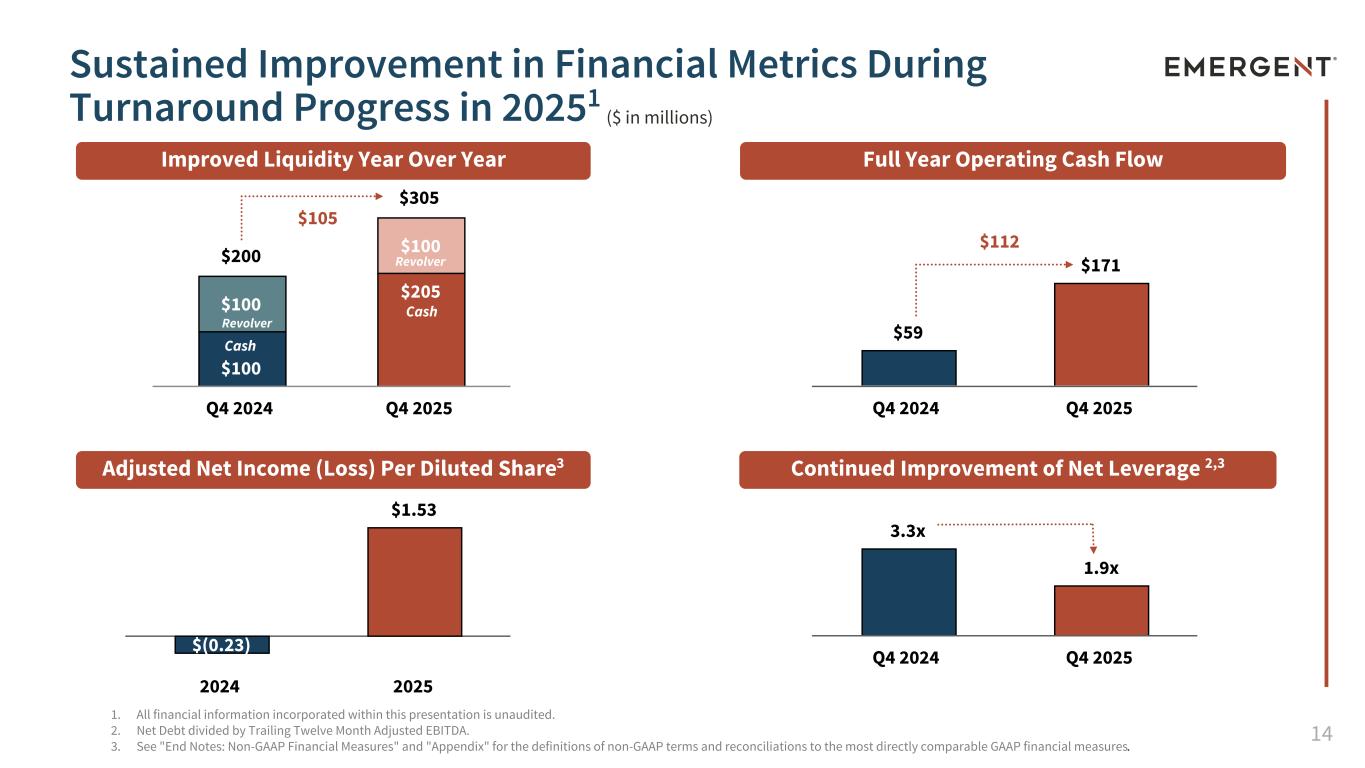
14 $(0.23) $1.53 2024 2025 3.3x 1.9x Q4 2024 Q4 2025 $200 $305 $100 $205 $100 $100 Q4 2024 Q4 2025 1. All financial information incorporated within this presentation is unaudited. 2. Net Debt divided by Trailing Twelve Month Adjusted EBITDA. 3. See "End Notes: Non-GAAP Financial Measures" and "Appendix" for the definitions of non-GAAP terms and reconciliations to the most directly comparable GAAP financial measures. Adjusted Net Income (Loss) Per Diluted Share3 Sustained Improvement in Financial Metrics During Turnaround Progress in 20251 Improved Liquidity Year Over Year Full Year Operating Cash Flow Continued Improvement of Net Leverage 2,3 ($ in millions) Cash Revolver $105 Cash Revolver $59 $171 Q4 2024 Q4 2025 $112
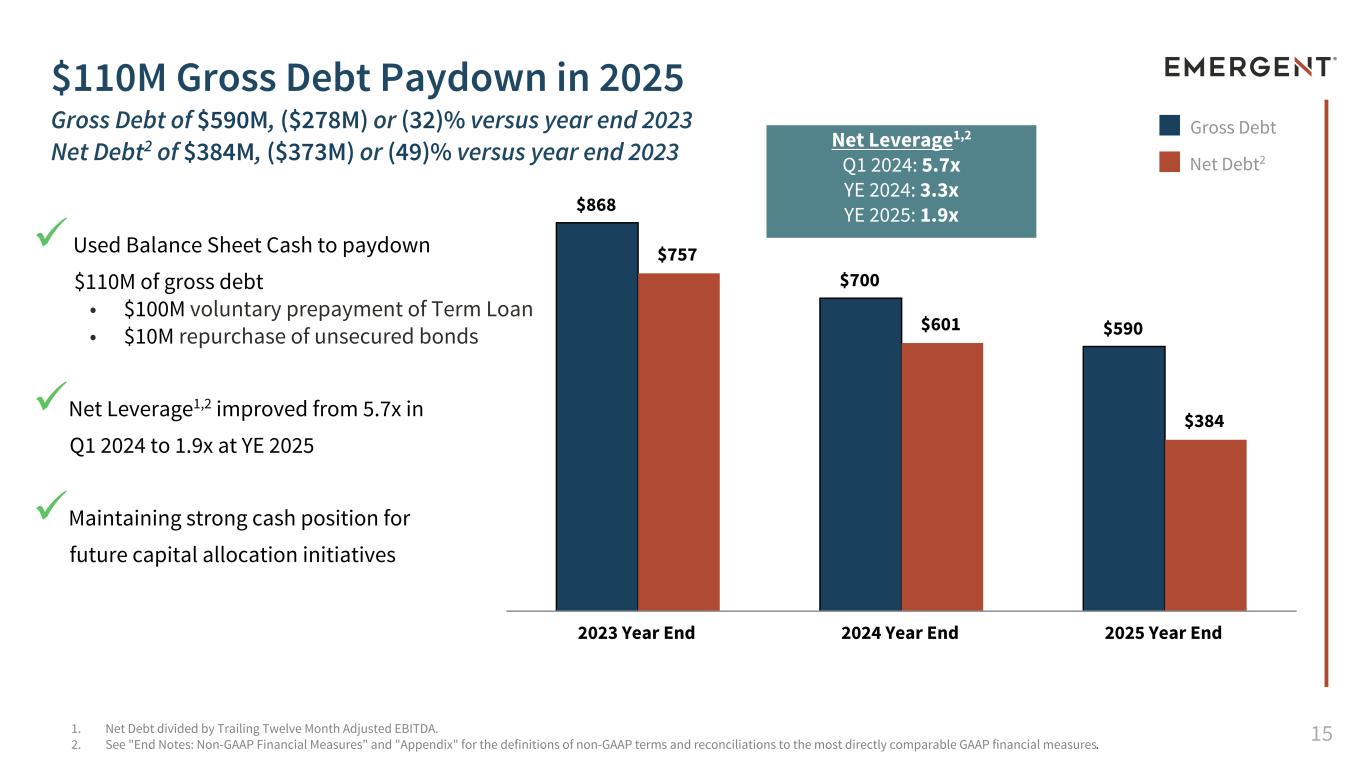
15 $110M Gross Debt Paydown in 2025 Gross Debt of $590M, ($278M) or (32)% versus year end 2023 Net Debt2 of $384M, ($373M) or (49)% versus year end 2023 1. Net Debt divided by Trailing Twelve Month Adjusted EBITDA. 2. See "End Notes: Non-GAAP Financial Measures" and "Appendix" for the definitions of non-GAAP terms and reconciliations to the most directly comparable GAAP financial measures. $868 $700 $590 $757 $601 $384 2023 Year End 2024 Year End 2025 Year End Gross Debt Net Debt2 Net Leverage1,2 Q1 2024: 5.7x YE 2024: 3.3x YE 2025: 1.9x ü Used Balance Sheet Cash to paydown $110M of gross debt • $100M voluntary prepayment of Term Loan • $10M repurchase of unsecured bonds üNet Leverage1,2 improved from 5.7x in Q1 2024 to 1.9x at YE 2025 üMaintaining strong cash position for future capital allocation initiatives

16 Growth Investments Capital Allocation Priorities During 2026 Turnaround Period Debt Repayment Share Repurchase • International MCM growth plan • KLOXXADO® Nasal Spray 8 mg • Swiss Rockets/Rocketvax investment • Internal R&D investments • Business development opportunities $110M Debt Paydown • $100M Term Loan paydown • Initiated $30M Bond Repurchase Program starting August 2025 • Repurchased $10.3M of EBS' unsecured bonds for $8.7M during 2025 ◦ Q4 $3.4M of bond repurchases for $2.9M • Net Debt1 $384M, a 36% reduction vs. last year • Net Leverage1 1.9x Initiated $50M Share Repurchase Program • 3.1M Shares repurchased during 2025 for $24.8M2 • Q4 0.8M shares repurchased for $9M Reauthorized a New Share Repurchase Program for $50M through March 2027 1. See "End Notes: Non-GAAP Financial Measures" and "Appendix" for the definitions of non-GAAP terms and reconciliations to the most directly comparable GAAP financial measures. 2. This amount is excluding the excise tax of $0.3M for the year ended December 31, 2025.

17 METRIC ($ in millions) FY 2025 Actuals FY 2025 as of October 29, 2025 FY 2025 as of August 6, 2025 FY 2025 as of May 7, 2025 FY 2025 as of March 3, 2025 Total revenues $743 $775 - $835 $765 - $835 $750 - $850 $750 - $850 Net income $53 $60 - $75 $40 - $65 $20 - $70 $16 - $66 Adjusted net income1 $87 $70 - $85 $45 - $70 $20 - $70 $20 - $70 Adjusted EBITDA1 $205 $195 - $210 $175 - $200 $150 - $200 $150 - $200 Adjusted gross margin %1 54% 52% - 54% 50% - 52% 48% - 51% 48% - 51% Segment Level Revenue MCM Products2 $457 $450 - $475 $440 - $475 $435 - $485 $435 - $485 Segment Level Revenue Commercial Products3 $226 $265 - $300 $265 - $300 $265 - $315 $265 - $315 Adjusted Net Income, Adjusted EBITDA, & Adjusted Gross Margin exceeded mid-point of Full Year Guidance Continued to Raise Midpoint of FY 2025 Revenue & Profitability Guidance Throughout the Year 1. See "End Notes: Non-GAAP Financial Measures" and "Appendix" for the definitions of non-GAAP terms and reconciliations to the most directly comparable GAAP financial measures. 2. Our MCM Products forecast excludes revenues related to RSDL®, which product was sold during the third quarter of 2024. 3. Our Commercial Products forecast consists of revenues for NARCAN® Nasal Spray and revenues from distribution of KLOXXADO® Nasal Spray 8 mg pursuant to an agreement with Hikma Pharmaceuticals PLC in January 2025

18 Rich Lindahl EVP, Chief Financial Officer FY 2026 Revenue & Profitability Guidance
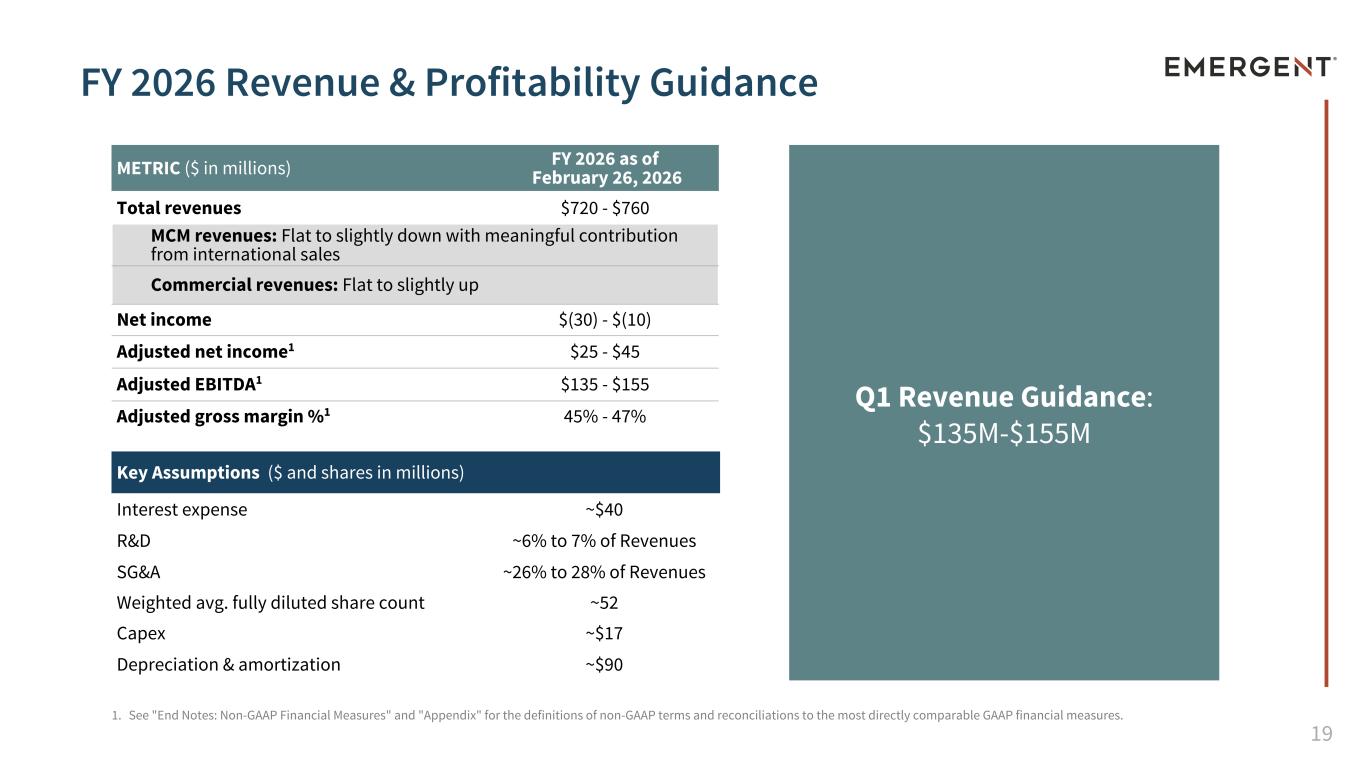
19 METRIC ($ in millions) FY 2026 as of February 26, 2026 Total revenues $720 - $760 MCM revenues: Flat to slightly down with meaningful contribution from international sales Commercial revenues: Flat to slightly up Net income $(30) - $(10) Adjusted net income1 $25 - $45 Adjusted EBITDA1 $135 - $155 Adjusted gross margin %1 45% - 47% Key Assumptions ($ and shares in millions) Interest expense ~$40 R&D ~6% to 7% of Revenues SG&A ~26% to 28% of Revenues Weighted avg. fully diluted share count ~52 Capex ~$17 Depreciation & amortization ~$90 1. See "End Notes: Non-GAAP Financial Measures" and "Appendix" for the definitions of non-GAAP terms and reconciliations to the most directly comparable GAAP financial measures. FY 2026 Revenue & Profitability Guidance Q1 Revenue Guidance: $135M-$155M

2 0 20 2025 Key Summary • Continued strong execution and progress on the turnaround phase of multi-year plan through 2025 • Increased gross margin & profit follow through from 2024, with a lean, efficient business model to build upon going forward • Strong operating cash flow in 2025 and expecting this trend to continue in 2026, allowing continued focus on key capital allocation strategies • Further deleveraging with $110M gross debt paydown • Net Debt1 declined 36% YoY and Net Leverage1 reduced from 3.3x to 1.9x at year end 2025 • Identifying opportunities to deliver value to shareholders • 3.1M shares repurchased in 2025 • Reauthorized share repurchase program through March 2027 1. See "End Notes: Non-GAAP Financial Measures" and "Appendix" for the definitions of non-GAAP terms and reconciliations to the most directly comparable GAAP financial measures.

21 2026 Business Outlook & Catalysts to Enable Growth Joe Papa President and Chief Executive Officer
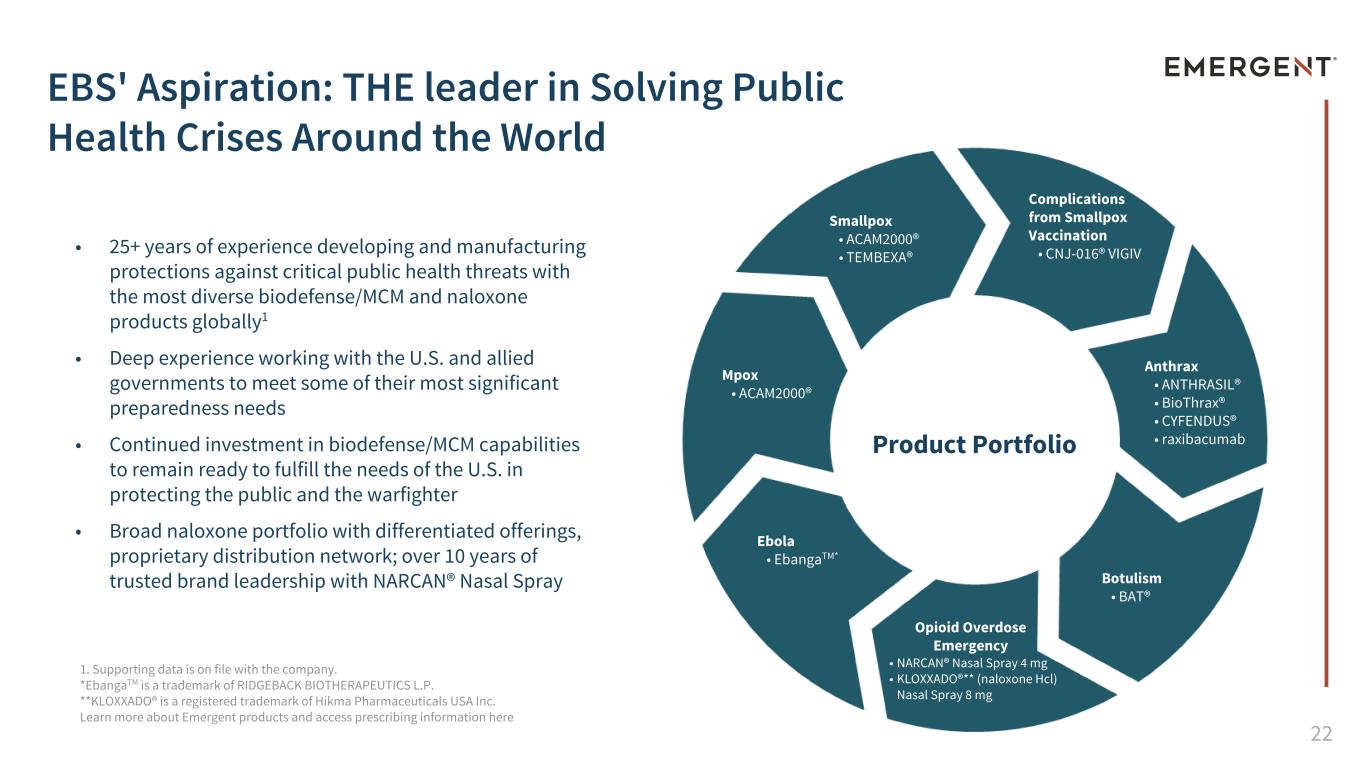
22 • 25+ years of experience developing and manufacturing protections against critical public health threats with the most diverse biodefense/MCM and naloxone products globally1 • Deep experience working with the U.S. and allied governments to meet some of their most significant preparedness needs • Continued investment in biodefense/MCM capabilities to remain ready to fulfill the needs of the U.S. in protecting the public and the warfighter • Broad naloxone portfolio with differentiated offerings, proprietary distribution network; over 10 years of trusted brand leadership with NARCAN® Nasal Spray Product Portfolio Smallpox • ACAM2000® • TEMBEXA® Ebola • EbangaTM* Botulism • BAT® Opioid Overdose Emergency • NARCAN® Nasal Spray 4 mg • KLOXXADO®** (naloxone Hcl) Nasal Spray 8 mg Anthrax • ANTHRASIL® • BioThrax® • CYFENDUS® • raxibacumab Complications from Smallpox Vaccination • CNJ-016® VIGIV 1. Supporting data is on file with the company. *EbangaTM is a trademark of RIDGEBACK BIOTHERAPEUTICS L.P. **KLOXXADO® is a registered trademark of Hikma Pharmaceuticals USA Inc. Learn more about Emergent products and access prescribing information here Mpox • ACAM2000® EBS' Aspiration: THE leader in Solving Public Health Crises Around the World
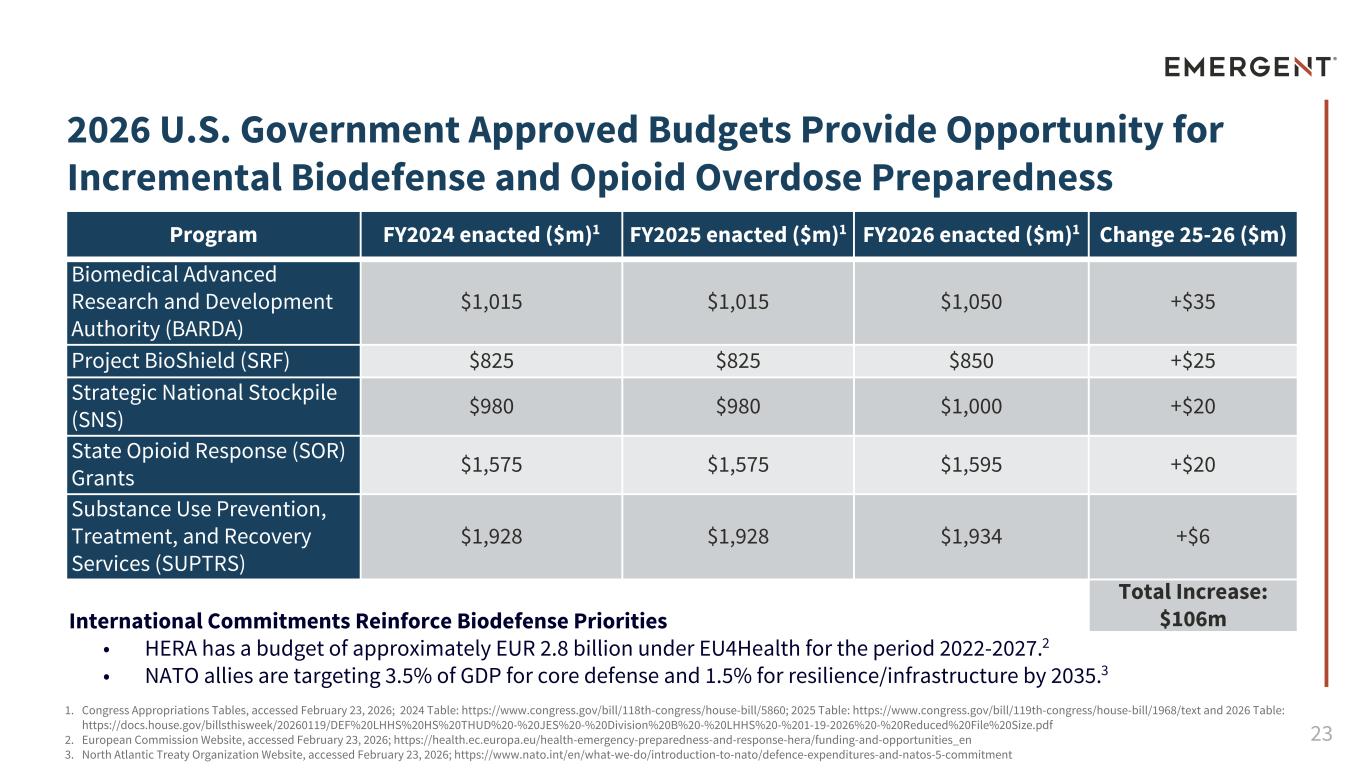
23 2026 U.S. Government Approved Budgets Provide Opportunity for Incremental Biodefense and Opioid Overdose Preparedness Program FY2024 enacted ($m)1 FY2025 enacted ($m)1 FY2026 enacted ($m)1 Change 25-26 ($m) Biomedical Advanced Research and Development Authority (BARDA) $1,015 $1,015 $1,050 +$35 Project BioShield (SRF) $825 $825 $850 +$25 Strategic National Stockpile (SNS) $980 $980 $1,000 +$20 State Opioid Response (SOR) Grants $1,575 $1,575 $1,595 +$20 Substance Use Prevention, Treatment, and Recovery Services (SUPTRS) $1,928 $1,928 $1,934 +$6 Total Increase: $106mInternational Commitments Reinforce Biodefense Priorities • HERA has a budget of approximately EUR 2.8 billion under EU4Health for the period 2022-2027.2 • NATO allies are targeting 3.5% of GDP for core defense and 1.5% for resilience/infrastructure by 2035.3 1. Congress Appropriations Tables, accessed February 23, 2026; 2024 Table: https://www.congress.gov/bill/118th-congress/house-bill/5860; 2025 Table: https://www.congress.gov/bill/119th-congress/house-bill/1968/text and 2026 Table: https://docs.house.gov/billsthisweek/20260119/DEF%20LHHS%20HS%20THUD%20-%20JES%20-%20Division%20B%20-%20LHHS%20-%###-##-####%20-%20Reduced%20File%20Size.pdf 2. European Commission Website, accessed February 23, 2026; https://health.ec.europa.eu/health-emergency-preparedness-and-response-hera/funding-and-opportunities_en 3. North Atlantic Treaty Organization Website, accessed February 23, 2026; https://www.nato.int/en/what-we-do/introduction-to-nato/defence-expenditures-and-natos-5-commitment

24 Multiple Growth Opportunities Ahead Drive organic growth through internal R&D programs • TEMBEXA® • EBANGA™ • raxibacumab With a stronger cash and liquidity position entering 2026, we plan to invest to enable sustainable, long- term growth. Exploring potential for government-funded R&D development programs. Selectively evaluating strategically suitable external programs. Launch additional line extensions for naloxone business • NARCAN® Nasal Spray Carrying Case • Multipack configurations Expand international MCM orders and opportunities Accelerate inorganic growth through external business development opportunities
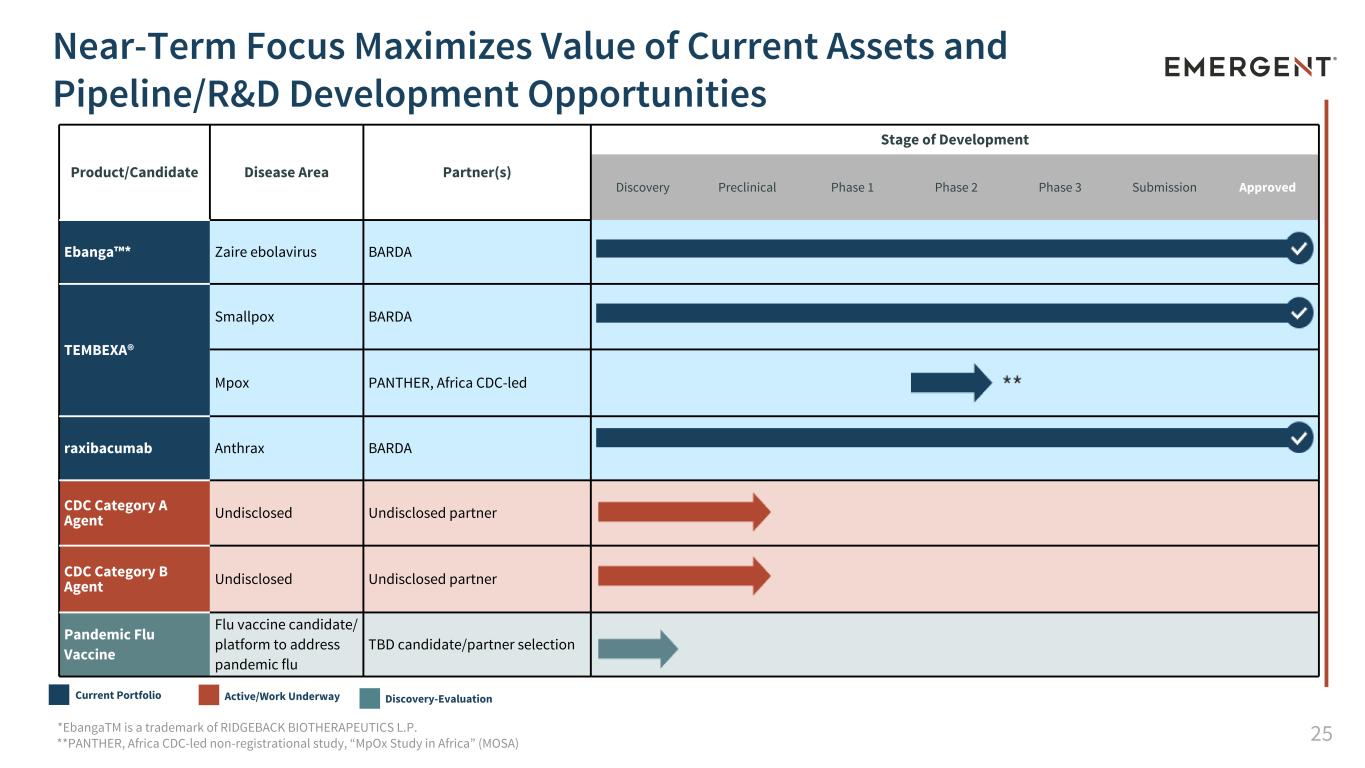
25 Product/Candidate Disease Area Partner(s) Stage of Development Discovery Preclinical Phase 1 Phase 2 Phase 3 Submission Approved Ebanga™* Zaire ebolavirus BARDA TEMBEXA® Smallpox BARDA Mpox PANTHER, Africa CDC-led raxibacumab Anthrax BARDA CDC Category A Agent Undisclosed Undisclosed partner CDC Category B Agent Undisclosed Undisclosed partner Pandemic Flu Vaccine Flu vaccine candidate/ platform to address pandemic flu TBD candidate/partner selection Near-Term Focus Maximizes Value of Current Assets and Pipeline/R&D Development Opportunities *EbangaTM is a trademark of RIDGEBACK BIOTHERAPEUTICS L.P. **PANTHER, Africa CDC-led non-registrational study, “MpOx Study in Africa” (MOSA) Current Portfolio Active/Work Underway Discovery-Evaluation

26 • Continued to deliver on multi-year transformation plan; on track to execute on key turnaround actions, financial targets to drive our business forward • Operated with lean, efficient and customer-focused business model • Introduced FY 2026 revenue and profitability guidance • Continue to repurchase shares and reduce debt • Naloxone and MCM products continue to deliver on EBS’ mission to protect and save lives • Pursuing organic and inorganic growth initiatives and creating shareholder value • Ongoing commitment to patient safety, quality and compliance across the enterprise Closing Remarks

27 Q&A Session Questions? Answers.

28

29 End Notes: Non-GAAP Financial Measures In this presentation, we sometimes use information derived from consolidated and segment financial information that may not be presented in our financial statements or prepared in accordance with generally accepted accounting principles in the United States (“GAAP”). Certain of these financial measures are considered not in conformity with GAAP (“non-GAAP financial measures”) under the United States Securities and Exchange Commission (“SEC”) rules. Specifically, we have referred to the following non- GAAP financial measures: • Adjusted Net Income (Loss) • Adjusted Net Income (Loss) per Diluted Share • Adjusted EBITDA • Adjusted EBITDA Margin • Adjusted Gross Margin • Adjusted Gross Margin % • Net Debt • Net Leverage Ratio • Normalized Revenue We define Adjusted Net Income (Loss) and Adjusted Net Income (Loss) per Diluted Share, which are non-GAAP financial measures, as net income (loss) and net income (loss) per diluted share, respectively, excluding the impact of non-cash amortization charges, impairments, severance and restructuring costs (benefits), inventory step-up provision, acquisition and divestiture costs, exit and disposal costs, gain on sale of business, settlement charges, net, contingent consideration milestones, changes in fair value of financial instruments, loss (gain) on debt extinguishment, other expense (income) items and tax effects. We use Adjusted Net Income (Loss) for the purpose of calculating Adjusted Net Income (Loss) per Diluted Share. Management uses Adjusted Net Income (Loss) per Diluted Share to assess total Company operating performance on a consistent basis. We believe that these non-GAAP financial measures, when considered together with our GAAP financial results and GAAP financial measures, provide management and investors with an additional understanding of our business operating results, including underlying trends. We define Adjusted EBITDA, which is a non-GAAP financial measure, as net income (loss) before depreciation and amortization, income taxes, interest expense, net, and excluding impairments, inventory step-up provision, changes in fair value of financial instruments, severance and restructuring costs (benefits), exit and disposal costs, acquisition and divestiture costs, gain on sale of business, settlement charges, net, contingent consideration milestones, loss (gain) on debt extinguishment, and other expense (income) items. We define Adjusted EBITDA Margin, which is a non-GAAP financial measure, as Adjusted EBITDA divided by Total Revenues. We believe that these non-GAAP financial measures, when considered together with our GAAP financial results and GAAP financial measures, provide management and investors with a more complete understanding of our operating results, including underlying trends. In addition, EBITDA is a common alternative measure of operating performance used by many of our competitors. It is used by investors, financial analysts, rating agencies and others to value and compare the financial performance of companies in our industry, although it may be defined differently by different companies. Therefore, we also believe that this non-GAAP financial measure, considered along with corresponding GAAP financial measures, provides management and investors with additional information for comparison of our operating results with the operating results of other companies.

30 End Notes: Non-GAAP Financial Measures (Continued) We define Adjusted Gross Margin, which is a non-GAAP financial measure, as Gross Margin excluding the impact of intangible asset amortization, inventory step-up provision, settlement charges, net, restructuring costs (benefits), and changes in the fair value of financial instruments. We define Adjusted Gross Margin %, which is a non-GAAP financial measure, as Adjusted Gross Margin as a percentage of revenues. We define Net Debt, which is a non-GAAP financial measure, as our total debt less our cash and cash equivalents. We believe this non-GAAP financial measure, when considered together with our GAAP financial results, provides management and investors with an additional understanding of the Company's ability to pay its debts. We define Net Leverage Ratio, which is a non-GAAP financial measure, as our Net Debt divided by our Trailing Twelve Month Adjusted EBITDA. We believe this non-GAAP financial measure, when considered together with our GAAP financial results, provides management and investors with an additional understanding of the Company's current borrowing capabilities. We define Normalized Revenues which is a non-GAAP financial measure, as the Total Revenues, less non-recurring revenue related to the Janssen settlement and divestitures of RSDL® and Camden. We believe this non-GAAP financial measure, when considered together with our GAAP financial results, provides management and investors with an additional understanding of the Company's revenues. Non-GAAP financial measures are not defined in the same manner by all companies and may not be comparable with other similarly titled measures of other companies. The determination of the amounts that are excluded from these non-GAAP financial measures are a matter of management judgment and depend upon, among other factors, the nature of the underlying expense or income amounts. Because non-GAAP financial measures exclude the effect of items that will increase or decrease the Company’s reported results of operations, management strongly encourages investors to review the Company’s consolidated financial statements and publicly filed reports in their entirety. For additional information on the non-GAAP financial measures noted here, please refer to the reconciliation tables provide in the Appendix to this presentation as well as the associated press release which can be found on the Company’s website at www.emergentbiosolutions.com.
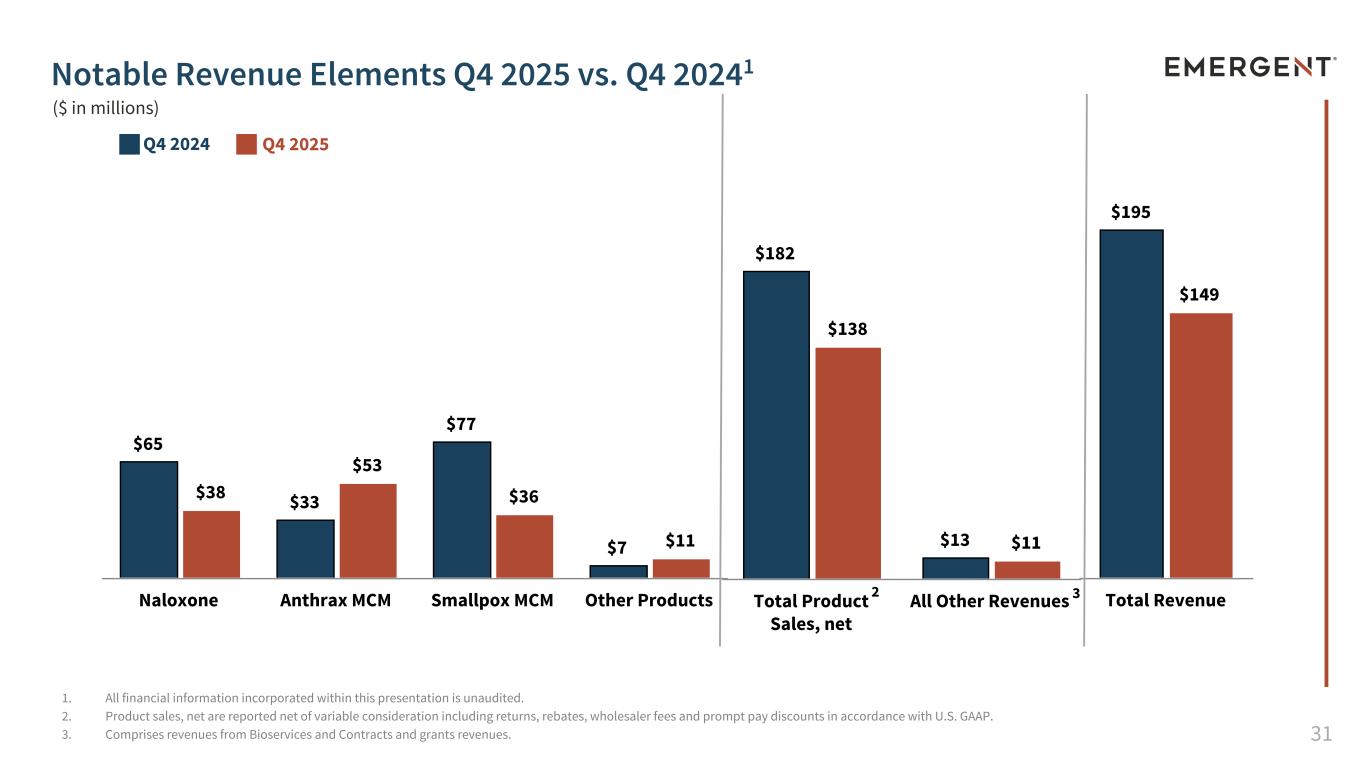
31 $65 $33 $77 $7 $38 $53 $36 $11 Naloxone Anthrax MCM Smallpox MCM Other Products $195 $149 Total Revenue $182 $13 $138 $11 Total Product Sales, net All Other Revenues 1. All financial information incorporated within this presentation is unaudited. 2. Product sales, net are reported net of variable consideration including returns, rebates, wholesaler fees and prompt pay discounts in accordance with U.S. GAAP. 3. Comprises revenues from Bioservices and Contracts and grants revenues. Notable Revenue Elements Q4 2025 vs. Q4 20241 Q4 2024 Q4 2025 3 ($ in millions) 2
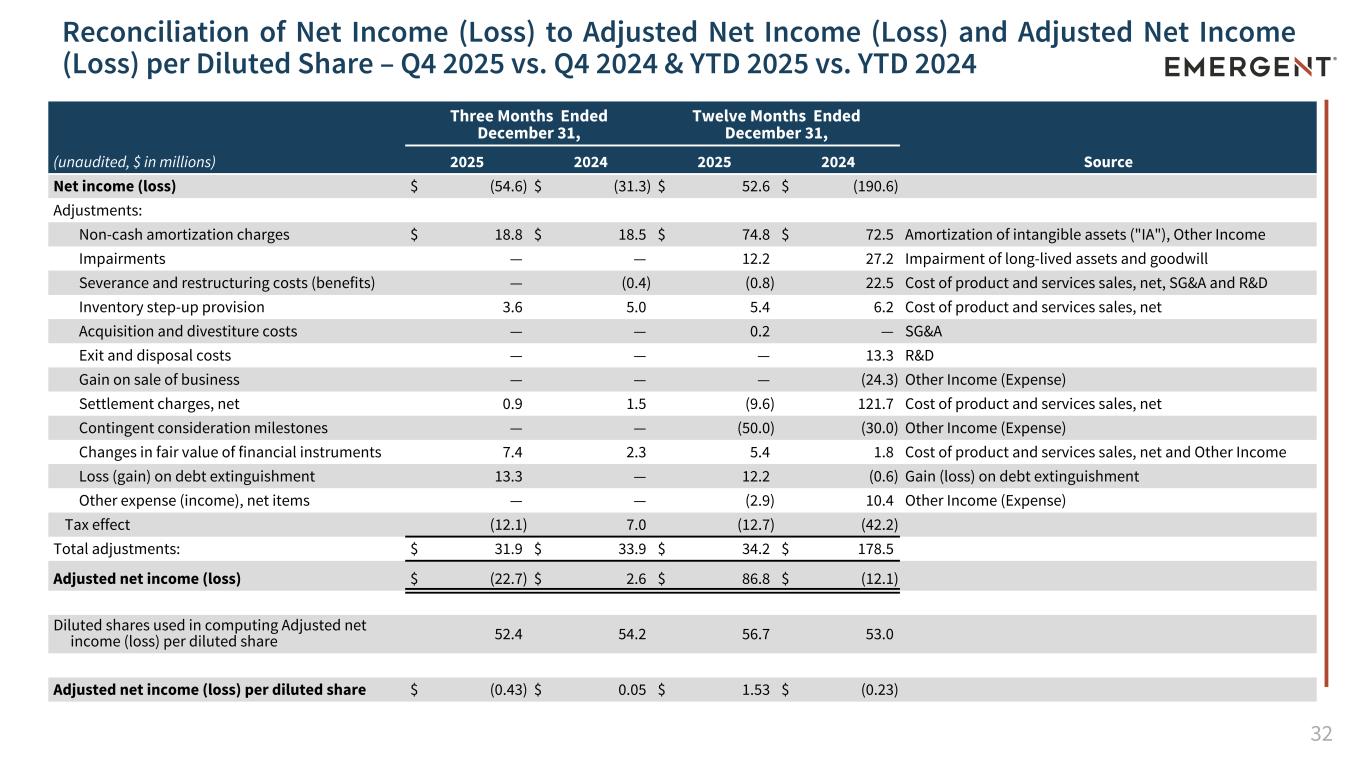
32 Reconciliation of Net Income (Loss) to Adjusted Net Income (Loss) and Adjusted Net Income (Loss) per Diluted Share – Q4 2025 vs. Q4 2024 & YTD 2025 vs. YTD 2024 (unaudited, $ in millions) Three Months Ended December 31, Twelve Months Ended December 31, 2025 2024 2025 2024 Source Net income (loss) $ (54.6) $ (31.3) $ 52.6 $ (190.6) Adjustments: Non-cash amortization charges $ 18.8 $ 18.5 $ 74.8 $ 72.5 Amortization of intangible assets ("IA"), Other Income Impairments — — 12.2 27.2 Impairment of long-lived assets and goodwill Severance and restructuring costs (benefits) — (0.4) (0.8) 22.5 Cost of product and services sales, net, SG&A and R&D Inventory step-up provision 3.6 5.0 5.4 6.2 Cost of product and services sales, net Acquisition and divestiture costs — — 0.2 — SG&A Exit and disposal costs — — — 13.3 R&D Gain on sale of business — — — (24.3) Other Income (Expense) Settlement charges, net 0.9 1.5 (9.6) 121.7 Cost of product and services sales, net Contingent consideration milestones — — (50.0) (30.0) Other Income (Expense) Changes in fair value of financial instruments 7.4 2.3 5.4 1.8 Cost of product and services sales, net and Other Income Loss (gain) on debt extinguishment 13.3 — 12.2 (0.6) Gain (loss) on debt extinguishment Other expense (income), net items — — (2.9) 10.4 Other Income (Expense) Tax effect (12.1) 7.0 (12.7) (42.2) Total adjustments: $ 31.9 $ 33.9 $ 34.2 $ 178.5 Adjusted net income (loss) $ (22.7) $ 2.6 $ 86.8 $ (12.1) Diluted shares used in computing Adjusted net income (loss) per diluted share 52.4 54.2 56.7 53.0 Adjusted net income (loss) per diluted share $ (0.43) $ 0.05 $ 1.53 $ (0.23)
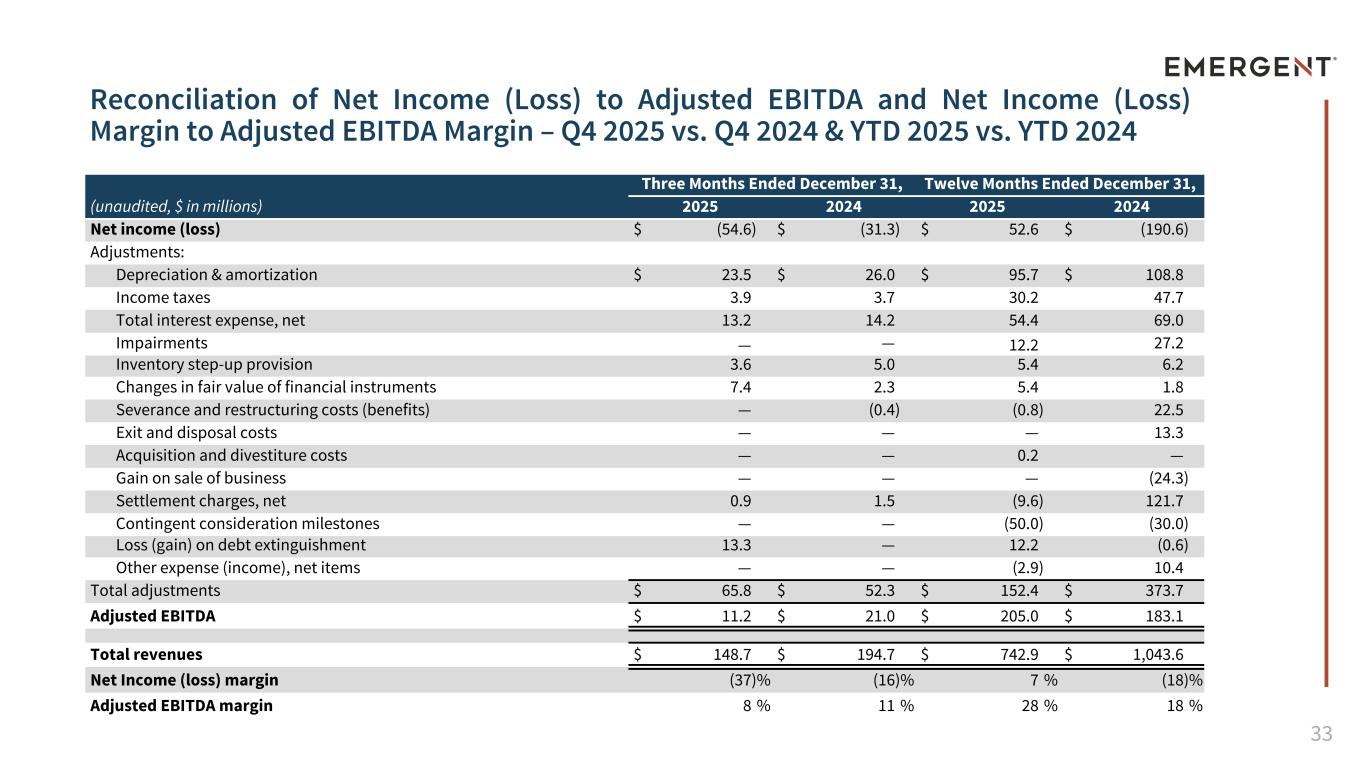
33 Reconciliation of Net Income (Loss) to Adjusted EBITDA and Net Income (Loss) Margin to Adjusted EBITDA Margin – Q4 2025 vs. Q4 2024 & YTD 2025 vs. YTD 2024 (unaudited, $ in millions) Three Months Ended December 31, Twelve Months Ended December 31, 2025 2024 2025 2024 Net income (loss) $ (54.6) $ (31.3) $ 52.6 $ (190.6) Adjustments: Depreciation & amortization $ 23.5 $ 26.0 $ 95.7 $ 108.8 Income taxes 3.9 3.7 30.2 47.7 Total interest expense, net 13.2 14.2 54.4 69.0 Impairments — — 12.2 27.2 Inventory step-up provision 3.6 5.0 5.4 6.2 Changes in fair value of financial instruments 7.4 2.3 5.4 1.8 Severance and restructuring costs (benefits) — (0.4) (0.8) 22.5 Exit and disposal costs — — — 13.3 Acquisition and divestiture costs — — 0.2 — Gain on sale of business — — — (24.3) Settlement charges, net 0.9 1.5 (9.6) 121.7 Contingent consideration milestones — — (50.0) (30.0) Loss (gain) on debt extinguishment 13.3 — 12.2 (0.6) Other expense (income), net items — — (2.9) 10.4 Total adjustments $ 65.8 $ 52.3 $ 152.4 $ 373.7 Adjusted EBITDA $ 11.2 $ 21.0 $ 205.0 $ 183.1 Total revenues $ 148.7 $ 194.7 $ 742.9 $ 1,043.6 Net Income (loss) margin (37) % (16) % 7 % (18) % Adjusted EBITDA margin 8 % 11 % 28 % 18 %
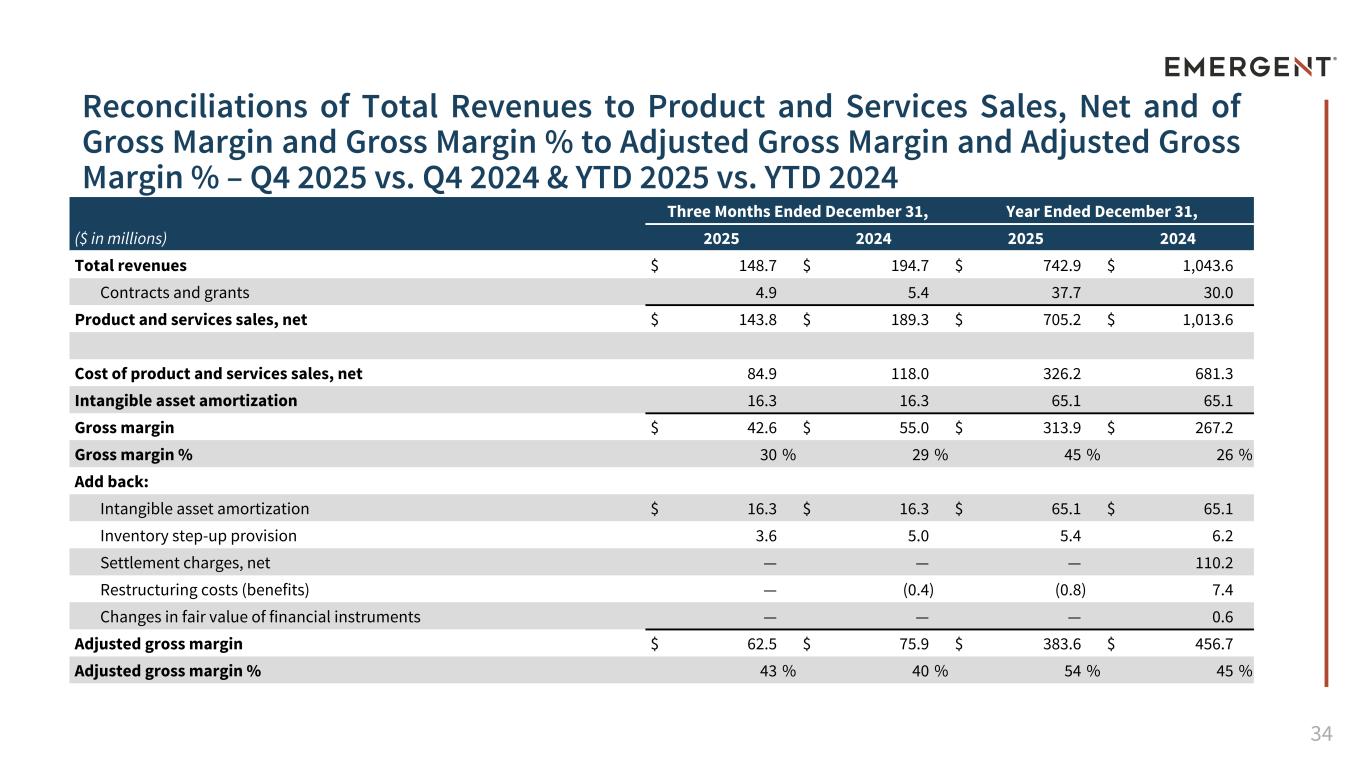
34 Reconciliations of Total Revenues to Product and Services Sales, Net and of Gross Margin and Gross Margin % to Adjusted Gross Margin and Adjusted Gross Margin % – Q4 2025 vs. Q4 2024 & YTD 2025 vs. YTD 2024 APPENDIX Three Months Ended December 31, Year Ended December 31, ($ in millions) 2025 2024 2025 2024 Total revenues $ 148.7 $ 194.7 $ 742.9 $ 1,043.6 Contracts and grants 4.9 5.4 37.7 30.0 Product and services sales, net $ 143.8 $ 189.3 $ 705.2 $ 1,013.6 Cost of product and services sales, net 84.9 118.0 326.2 681.3 Intangible asset amortization 16.3 16.3 65.1 65.1 Gross margin $ 42.6 $ 55.0 $ 313.9 $ 267.2 Gross margin % 30 % 29 % 45 % 26 % Add back: Intangible asset amortization $ 16.3 $ 16.3 $ 65.1 $ 65.1 Inventory step-up provision 3.6 5.0 5.4 6.2 Settlement charges, net — — — 110.2 Restructuring costs (benefits) — (0.4) (0.8) 7.4 Changes in fair value of financial instruments — — — 0.6 Adjusted gross margin $ 62.5 $ 75.9 $ 383.6 $ 456.7 Adjusted gross margin % 43 % 40 % 54 % 45 %
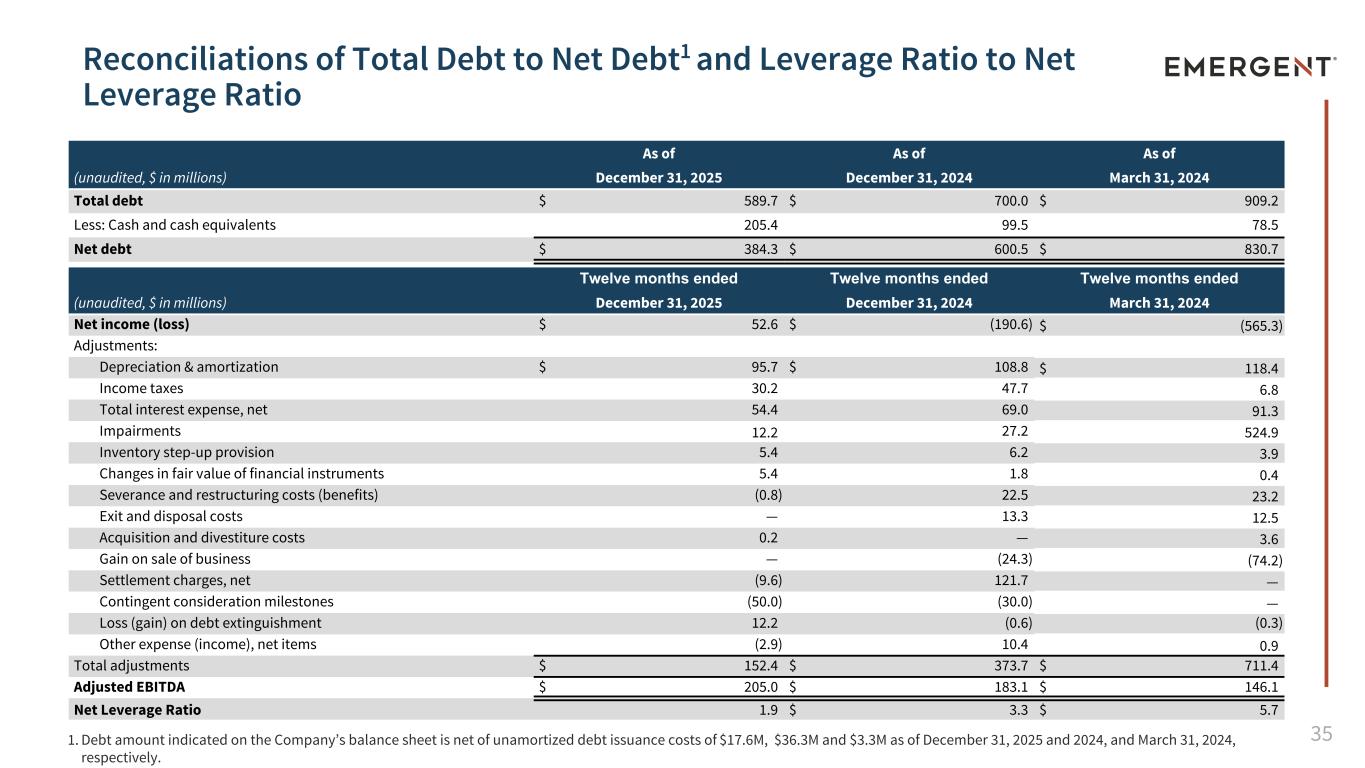
35 Reconciliations of Total Debt to Net Debt1 and Leverage Ratio to Net Leverage Ratio (unaudited, $ in millions) As of As of As of December 31, 2025 December 31, 2024 March 31, 2024 Total debt $ 589.7 $ 700.0 $ 909.2 Less: Cash and cash equivalents 205.4 99.5 78.5 Net debt $ 384.3 $ 600.5 $ 830.7 Twelve months ended Twelve months ended Twelve months ended (unaudited, $ in millions) December 31, 2025 December 31, 2024 March 31, 2024 Net income (loss) $ 52.6 $ (190.6) $ (565.3) Adjustments: Depreciation & amortization $ 95.7 $ 108.8 $ 118.4 Income taxes 30.2 47.7 6.8 Total interest expense, net 54.4 69.0 91.3 Impairments 12.2 27.2 524.9 Inventory step-up provision 5.4 6.2 3.9 Changes in fair value of financial instruments 5.4 1.8 0.4 Severance and restructuring costs (benefits) (0.8) 22.5 23.2 Exit and disposal costs — 13.3 12.5 Acquisition and divestiture costs 0.2 — 3.6 Gain on sale of business — (24.3) (74.2) Settlement charges, net (9.6) 121.7 — Contingent consideration milestones (50.0) (30.0) — Loss (gain) on debt extinguishment 12.2 (0.6) (0.3) Other expense (income), net items (2.9) 10.4 0.9 Total adjustments $ 152.4 $ 373.7 $ 711.4 Adjusted EBITDA $ 205.0 $ 183.1 $ 146.1 Net Leverage Ratio 1.9 $ 3.3 $ 5.7 1. Debt amount indicated on the Company’s balance sheet is net of unamortized debt issuance costs of $17.6M, $36.3M and $3.3M as of December 31, 2025 and 2024, and March 31, 2024, respectively.
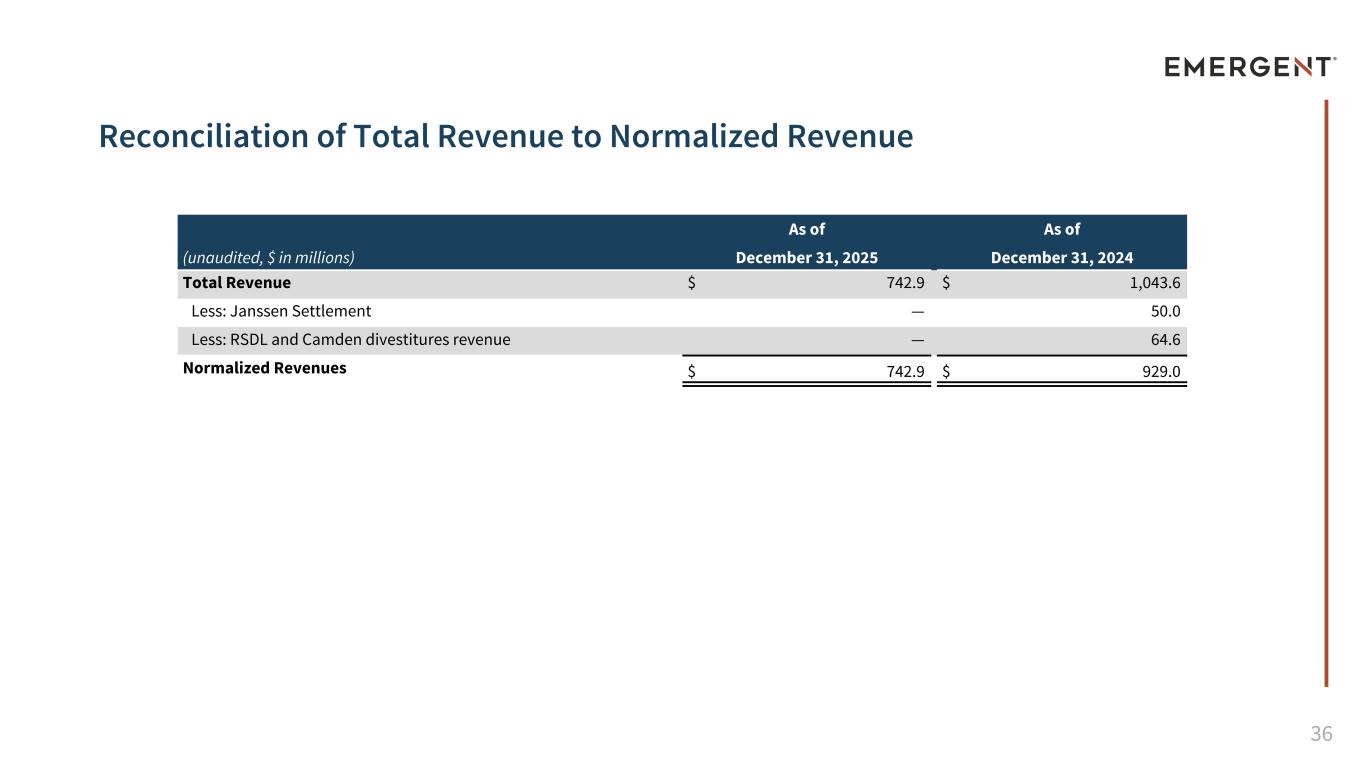
36 Reconciliation of Total Revenue to Normalized Revenue (unaudited, $ in millions) As of As of December 31, 2025 December 31, 2024 Total Revenue $ 742.9 $ 1,043.6 Less: Janssen Settlement — 50.0 Less: RSDL and Camden divestitures revenue — 64.6 Normalized Revenues $ 742.9 $ 929.0
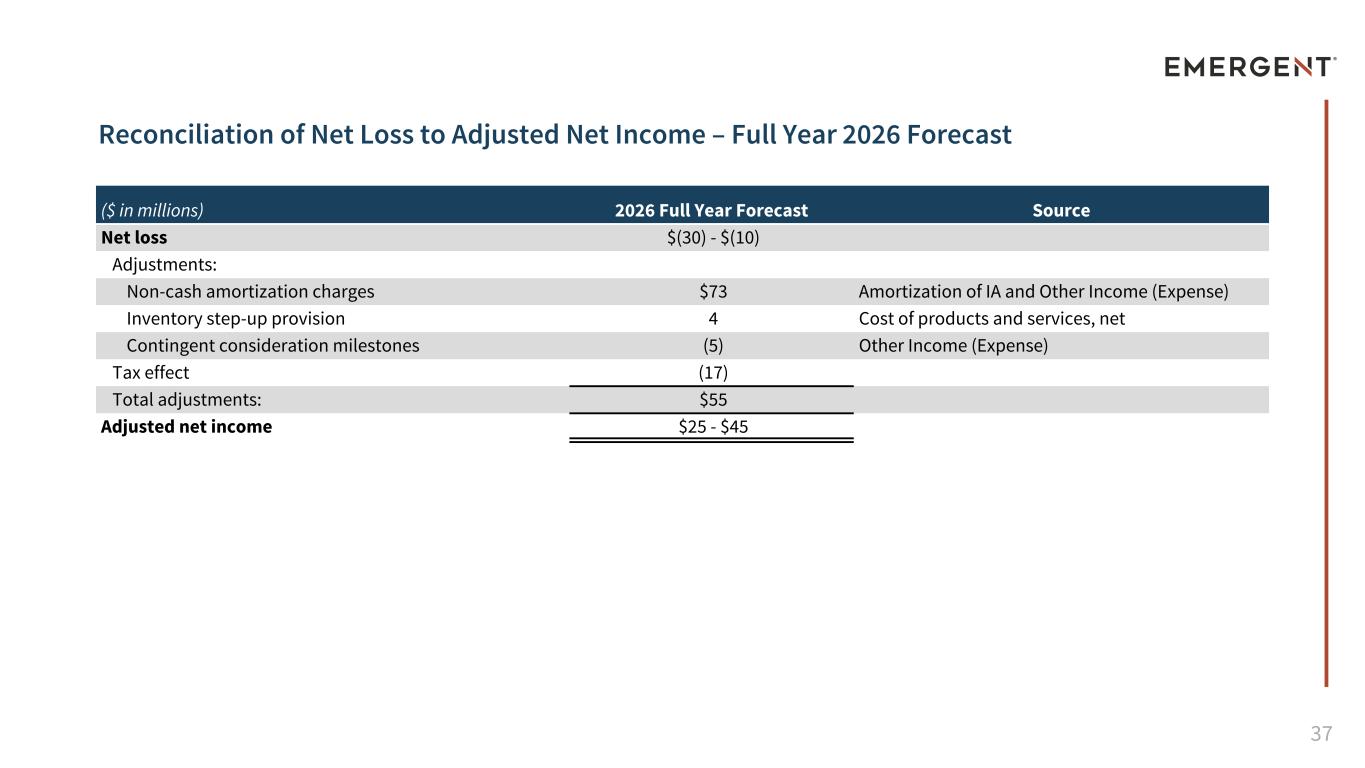
37 Reconciliation of Net Loss to Adjusted Net Income – Full Year 2026 Forecast ($ in millions) 2026 Full Year Forecast Source Net loss $(30) - $(10) Adjustments: Non-cash amortization charges $73 Amortization of IA and Other Income (Expense) Inventory step-up provision 4 Cost of products and services, net Contingent consideration milestones (5) Other Income (Expense) Tax effect (17) Total adjustments: $55 Adjusted net income $25 - $45
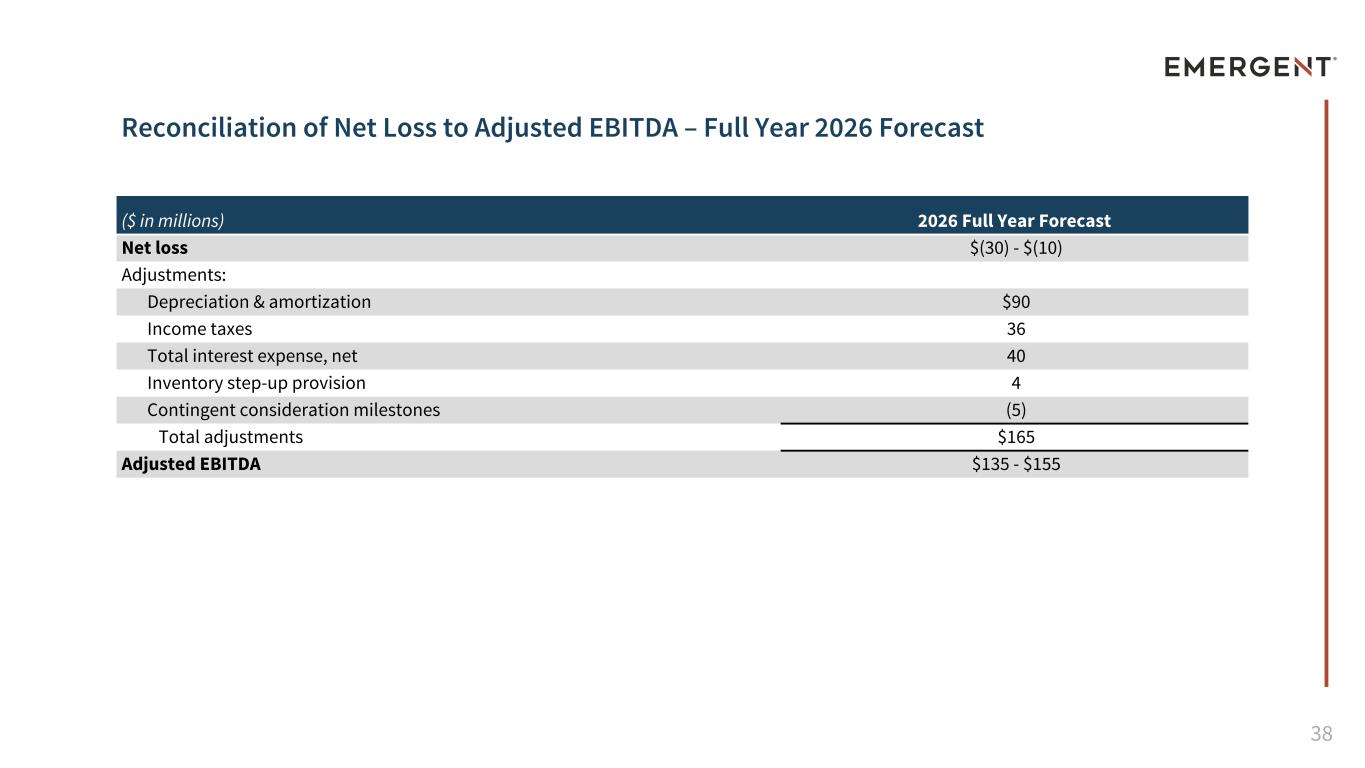
38 Reconciliation of Net Loss to Adjusted EBITDA – Full Year 2026 Forecast ($ in millions) 2026 Full Year Forecast Net loss $(30) - $(10) Adjustments: Depreciation & amortization $90 Income taxes 36 Total interest expense, net 40 Inventory step-up provision 4 Contingent consideration milestones (5) Total adjustments $165 Adjusted EBITDA $135 - $155
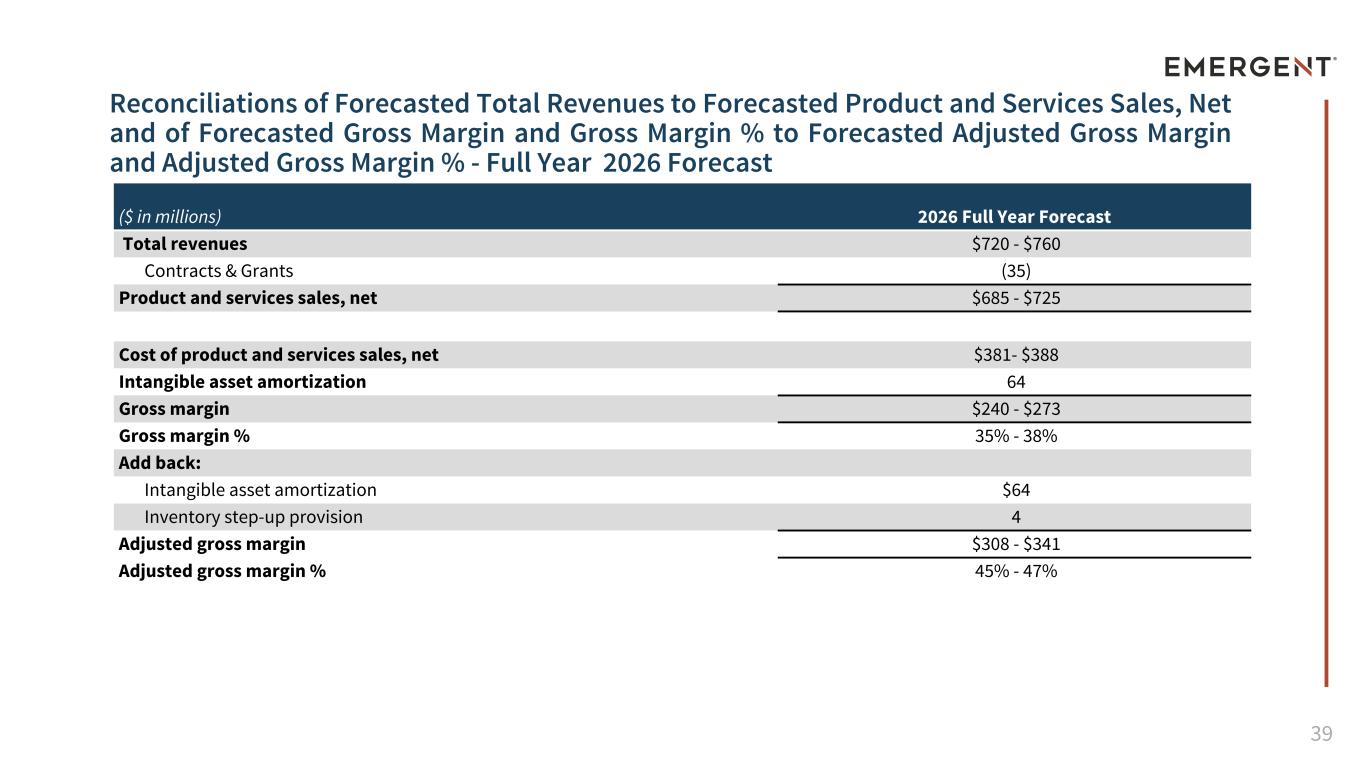
39 Reconciliations of Forecasted Total Revenues to Forecasted Product and Services Sales, Net and of Forecasted Gross Margin and Gross Margin % to Forecasted Adjusted Gross Margin and Adjusted Gross Margin % - Full Year 2026 Forecast ($ in millions) 2026 Full Year Forecast Total revenues $720 - $760 Contracts & Grants (35) Product and services sales, net $685 - $725 Cost of product and services sales, net $381- $388 Intangible asset amortization 64 Gross margin $240 - $273 Gross margin % 35% - 38% Add back: Intangible asset amortization $64 Inventory step-up provision 4 Adjusted gross margin $308 - $341 Adjusted gross margin % 45% - 47%
