
Dykema Gossett PLLC
111 E. Kilbourn Ave.
Suite 1050
Milwaukee, WI 53202
www.dykema.com
Tel: 414-488-7300
Kate Bechen
Direct Dial: (414) 488-7333
Email: KBechen@dykema.com
 |
Dykema Gossett PLLC 111 E. Kilbourn Ave. Suite 1050 Milwaukee, WI 53202 www.dykema.com Tel: 414-488-7300 | |
Kate Bechen Direct Dial: (414) 488-7333 Email: KBechen@dykema.com |
August 4, 2025
U.S. Securities and Exchange Commission
Division of Corporate Finance
Office of Life Sciences
100 F Street, N.E.
Washington, D.C. 20549
Attention: Daniel Crawford, Tim Buchmiller, Tracie
Mariner, and Kevin Vaughn
| Re: | Scienture Holdings, Inc. | |
| Form 10-K for Fiscal Year Ended December 31, 2024 | ||
| File No. 1-39199 |
Dear Mr. Crawford, Mr. Buchmiller, Ms. Mariner and Mr. Vaughn:
This response letter (this “Response”) is submitted on behalf of Scienture Holdings, Inc. (the “Company”) in response to the comments that the Company received from the staff of the Division of Corporation Finance (the “Staff”) of the U.S. Securities and Exchange Commission (the “SEC”) in a letter addressed to Dr. Narasimhan Mani, dated July 24, 2025 (the “Comment Letter”), with respect to the Company’s Annual Report on Form 10-K for the fiscal year ended December 31, 2024 (the “Form 10-K”).
For reference purposes, each of the Staff’s numbered comments from the Comment Letter is set forth in bold text below, followed by the Company’s response to each comment. The responses below are based on information provided to Dykema Gossett PLLC by the Company.
Form 10-K for Fiscal Year Ended December 31, 2024
Part I
Item 1. Business
Current Business – Scienture LLC
Research and Development and Product Portfolio, page 6
| 1. | In future filings, please revise your pipeline table so the progress arrow associated with SCN-104 does not enter the “Phase 1” column as it appears you have not filed an IND and commenced the Phase 1 trial for the product candidate. For SCN-106, to the extent you believe you can make an NDA/BLA submission without progressing through all three phases of clinical trials, as depicted in the table, please add appropriate disclosure that makes that clear. Revise to disclose the design and results of the Phase 3 clinical trial for SCN-106, or otherwise revise your pipeline table to indicate the product candidate’s current stage of development as it currently appears to state you have completed Phase 3 clinical development. |
Response: In response to the Staff’s comments, the Company advises the Staff that it will revise the pipeline table associated with the Company’s product portfolio in future filings to better depict the status of and requirements associated with its products as follows:
Research and Development and Product Portfolio
Scienture, LLC is committed to the development of innovative product candidates in the CNS and CVS therapeutic areas. The process by which Scienture, LLC intends to bring its product candidates to market and the anticipated launch dates of its product candidates is depicted in the following table. The progress of Scienture, LLC’s products through this process to date is represented by the green bars shown below.
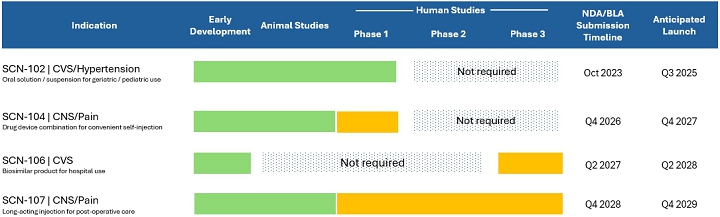
In addition, the Company will supplement its disclosures in future filings to clarify that, based on conversations with the United States Food and Drug Administration, the Company’s management believes that SCN-106 may be approved without advancing though all three clinical trial phases as depicted in the pipeline table above and further described below:
SCN-106 (Potential Biosimilar)
Scienture, LLC is developing a potential biosimilar, SCN-106, based on Cathflo Activase, a reference product that is a thrombolytic agent that binds to fibrin in clots and converts entrapped plasminogen to plasmin. SCN-106 is a sterile, purified glycoprotein that is synthesized using the complementary DNA for natural human tPA obtained from a Chinese hamster ovary cell-line.
Scienture, LLC is working with Anthem Biosciences Pvt, Ltd. (“Anthem”) to develop a biosimilar product that utilizes the same mechanism(s) of action for the proposed condition of use, and has the same route of administration, dosage form, and strength as the reference product. In this regard, Scienture, LLC entered into a Master Services Agreement with Anthem on October 29, 2024 (the “Anthem Agreement”). The following is a summary of the Anthem Agreement, which is qualified in its entirety by the full text of the Anthem Agreement, which is filed as an exhibit to this Annual Report.
Under the Anthem Agreement, Anthem has agreed to diligently complete the services associated with SCN-106 as included in work orders to be attached to the Anthem Agreement. In performing these services, Anthem has agreed to strictly comply with the specifications in the Anthem Agreement, the work order, standard operating procedures approved in writing by Scienture, LLC, and relevant professional standards, and any regulatory authority requirements, including current Good Laboratory Practices (“GLPs”) and current Good Manufacturing Practices (“GMPs”) promulgated by the FDA, and any other applicable laws, rules, and regulations. In carrying-out its services, Anthem will only allow those employees and personnel under Anthem’s direct control to perform such services and will obtain the Scienture, LLC’s consent prior to delegating or subcontracting any portion of the services. Anthem is required to provide prompt written reports to Scienture, LLC the status of the services provided by Anthem under the Anthem Agreement and any work order. Under the Anthem Agreement, Scienture, LLC is responsible for paying Anthem the amounts designated on any attached work order. These amounts are to be paid on the schedule stated on the work order and Anthem is responsible for invoicing Scienture, LLC for such amounts. Undisputed late payments incur interest at the rate of 18% per annum payable until the date of actual payment.
Any project or work order in effect under the Anthem Agreement may be terminated by Scienture, LLC without cause upon thirty (30) days’ prior notice to Anthem. Anthem may terminate the Anthem Agreement without cause upon thirty (30) days’ prior notice to Scienture, LLC. However, Anthem is responsible for delivering all services and deliverables under the Anthem Agreement then required to be performed by Anthem under a work order prior to any such termination. Either party may terminate the Anthem Agreement upon the breach of the Anthem Agreement by the other party if the breach remains uncured for a period of thirty (30) days. In the event that performance by Anthem or Scienture, LLC under the Anthem Agreement is delayed due to an event beyond the control of Anthem or Scienture, LLC for a period of ninety (90) days, then the other party can terminate the Anthem Agreement upon written notice.
With respect to projects to be performed by Anthem under the Anthem Agreement, any and all materials relating to such projects are the property of Scienture, LLC, and are to be protected as such by Anthem. Furthermore, Anthem has agreed to irrevocably assign to Scienture, LLC all right, title, and interest in and to any “Program Technology” (as defined in the Anthem Agreement) and to make any assignments necessary to ensure that Scienture, LLC has such ownership interest. The Anthem Agreement also contains customary confidentiality obligations, representations and warranties, indemnification provisions, and anti-assignment provisions. The Anthem Agreement may only be amended upon the written consent of both parties.
The CMC development program is focused on establishing the analytical similarity of SCN-106 to the reference product. Multiple clones of CHO cells have been produced to synthesize lots of SCN-106 which were screened for similarity to the reference product for several key biochemical quality attributes as well as overall protein yield and finalization of a lead clone.
Scienture, LLC completed a Biosimilar Initial Advisory meeting with the FDA in June 2023 to discuss the CMC, non-clinical, and clinical studies required for regulatory approval. As a result of this meeting, Scienture, LLC learned that its analytical strategy for initiating analytical similarity studies between SCN-106 and a proposed biosimilar product is acceptable. Scienture, LLC also learned that SCN-106 is suitable for further development and received guidance from the FDA on a comparable clinical study needed to demonstrate biosimilarity of SCN-106 and the reference product. In this regard, Scienture, LLC was informed that no additional safety, PK, toxicology or dose range finding studies will be required due to the method of use (very limited exposure) and the availability of an extensive amount of data on the original brand product. The only clinical requirement is a comparative phase 3 clinical study in the sensitive population to demonstrate that there are no clinically meaningful differences between SCN-106 and the currently marketed product.
Thank you for your review and consideration of the matters set forth in this Response. If you have any questions, please contact the undersigned at (414) 488-7333 or KBechen@dykema.com.
| Sincerely, | |
| Dykema Gossett PLLC | |
| /s/ Kate Bechen | |
| Kate Bechen, Esq. |
| cc: | Narasimhan Mani |
| Co-Chief Executive Officer and President | |
| Scienture Holdings, Inc. |