
Niagen Bioscience, Inc. Earnings Presentation Fourth Quarter and Full Year 2025 Nasdaq: NAGE | March 4, 2026

2 This presentation and other written or oral statements made from time to time by representatives of Niagen Bioscience contain “forward-looking statements” within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended. Forward-looking statements reflect the current view about future events. Statements that are not historical in nature, such as 2026 financial outlook, and which may be identified by the use of words like “expects,” “anticipates,” “intends,” “estimates,” “plans,” “potential,” “possible,” “probable,” “believes,” “seeks,” “may,” “will,” “should,” “could,” “predicts,” “projects,” “continue,” “would” or the negative of these terms and other words of similar meaning, are forward-looking statements. Such statements include, but are not limited to, statements contained in this presentation relating to our expected sales, cash flows, planned investments, and financial performance, business, business strategy, expansion, growth, key drivers (including cost savings and increased investments), products and services we offer and their impact on our performance or products and services we may offer in the future and the timing of their development, sales and marketing strategy and capital outlook. Forward-looking statements are based on management’s current expectations and assumptions regarding our business, the economy and other future conditions and are subject to inherent risks, uncertainties and changes of circumstances that are difficult to predict and may cause actual results to differ materially from those contemplated or expressed. We caution you therefore against relying on any of these forward-looking statements. These risks and uncertainties include those risk factors discussed in Part I, “Item 1A. Risk Factors” of our Annual Report on Form 10-K for the year ended December 31, 2025, filed with the Securities Exchange Commission (the “Commission”), and in subsequent filings with the Commission. Any forward-looking statements are qualified in their entirety by reference to the factors discussed in these filings with the Commission. Should one or more of these risks or uncertainties materialize, or should the underlying assumptions prove incorrect, actual results may differ significantly from those anticipated, believed, estimated, expected, intended or planned. Important factors that could cause actual results to differ materially from those in the forward looking statements include but are not limited to: our relationships with major customers; a decline in general economic conditions nationally and internationally; the market and size of the vitamin mineral and dietary supplement market and the intravenous market; decreased demand for our products and services; market acceptance of our products; the ability to protect our intellectual property rights; impact of any litigation or infringement actions brought against us; competition from other providers and products; risks in product development; our ability to develop pharmaceutical business; inability to raise capital to fund continuing operations or new product development; changes in government regulation or regulatory priorities of government officials; the ability to complete customer transactions and capital raising transactions; inflationary conditions and adverse economic conditions; our history of operating losses; the growth and profitability of our product sales; our ability to maintain and grow sales, marketing and distribution capabilities; changing consumer perceptions of our products; our reliance on a single or limited number of third-party suppliers; risks of conducting business in China; unanticipated developments in and risks related to the Company’s ability to secure adequate quantities of pharmaceutical-grade Niagen in a timely manner; the Company’s ability to obtain appropriate contracts and arrangements with U.S. FDA-registered 503B outsourcing facilities required to compound and distribute pharmaceutical-grade Niagen to clinics; the Company’s ability to remain on the U.S. FDA Bulk Drug Substances Nominated for Use in Compounding Under Section 503B of the Federal Food, Drug, and Cosmetic Act Category 1 list; the Company’s ability to maintain and enforce the Company’s existing intellectual property and obtain new patents; whether the potential benefits of NRC can be further supported; further research and development and the results of clinical trials possibly being unsuccessful or insufficient to meet applicable regulatory standards or warrant continued development; the ability to enroll sufficient numbers of subjects in clinical trials; determinations made by the FDA and other governmental authorities, including with respect to products seeking to compete in our market; mislabeling or other misleading marketing practices by competitors; economic and market instability, including as a result of tariffs or trade conflicts; and the risks and uncertainties associated with our business and financial condition in general. Factors or events that could cause our actual results to differ may emerge from time to time, and it is not possible for us to predict all of them. We cannot guarantee future results, levels of activity, performance or achievements. Except as required by applicable law, including the securities laws of the United States, we do not intend to update any of the forward-looking statements to conform these statements to actual results. About Non-GAAP Financial Measures Niagen Bioscience’s non-GAAP financial measure, Adjusted EBITDA, is defined as net income before interest, provision for income taxes, depreciation, amortization, non-cash share-based compensation costs, severance and restructuring expense and other infrequent items, including the gain recognized in nonoperating income related to a royalty settlement, reversal of previously accrued royalties and license maintenance fees and recoveries of previously recognized credit losses from a legal settlement. Niagen Bioscience used this non-GAAP measures when evaluating its financial results as well as for internal resource management, planning and forecasting purposes. This non-GAAP measure should not be viewed in isolation from or as a substitute for Niagen Bioscience’s financial results in accordance with GAAP. Reconciliation of this non-GAAP measure to the most directly comparable GAAP measure is attached to this presentation. FDA Disclaimer Statements made in this presentation have not been evaluated by the Food and Drug Administration. Niagen Bioscience products are not intended to diagnose, treat, cure, or prevent any disease. The statements in this presentation are for investor relations and educational purposes only and not intended for consumers or vendors. Safe Harbor Statement

Table of Contents & Leadership Team 3 Rob Fried Chief Executive Officer Ozan Pamir Chief Financial Officer Carlos Lopez SVP, General Counsel Andrew Shao SVP, Global Regulatory & Scientific Affairs Michiko Kelley Chief Marketing Officer 1. Highlights 2. Financial Results & Outlook 3.Scientific Updates 4.Contact Information

(1) See slide 13 for the non-GAAP reconciliation 4 64.3% gross profit margin, +250bps vs PY $0.20 diluted EPS, +$0.09 vs PY +103% Y/Y net income growth $20.4 million Adjusted EBITDA(1), +139% vs PY $64.8 million cash, no outstanding borrowings FY 2025 Financials Met or Exceeded Guidance Across All Key Metrics +30% Y/Y net sales growth

Enterprise • Successfully rolled out Niagen Bioscience rebrand, refocusing organization around its leading NAD+ position • Authorized $10 million share repurchase program Tru Niagen and Niagen Ingredient • Successfully captured increasing demand for Tru Niagen products across direct-to-consumer platforms • Launched Tru Niagen Beauty, expanding into multibillion dollar beauty market • Expanded the presence of Niagen Plus, featuring pharmaceutical-grade Niagen, to over 1,200 clinics nationwide Pharmaceuticals • Acquired core NR patent portfolio from Queen’s University Belfast • Published first-ever peer-reviewed study highlighting potential of NR for rare genetic disorder Werner Syndrome • Secured U.S. Composition of Matter patent for NR salt forms • Appointed Dr. Pinchas Cohen to Scientific Advisory Board Operational Execution Drove 2025 Performance 5
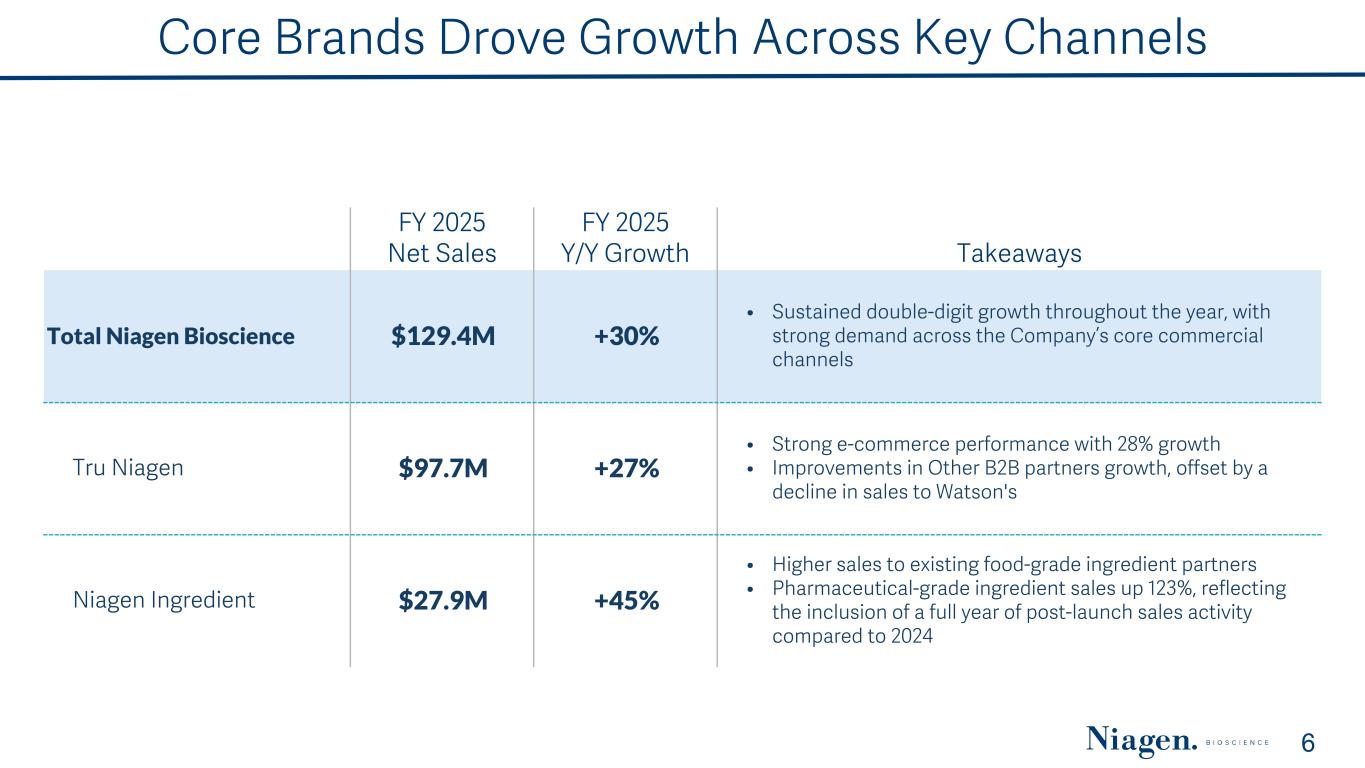
Core Brands Drove Growth Across Key Channels 6 FY 2025 Net Sales FY 2025 Y/Y Growth Takeaways Total Niagen Bioscience $129.4M +30% • Sustained double-digit growth throughout the year, with strong demand across the Company’s core commercial channels Tru Niagen $97.7M +27% • Strong e-commerce performance with 28% growth • Improvements in Other B2B partners growth, offset by a decline in sales to Watson's Niagen Ingredient $27.9M +45% • Higher sales to existing food-grade ingredient partners • Pharmaceutical-grade ingredient sales up 123%, reflecting the inclusion of a full year of post-launch sales activity compared to 2024
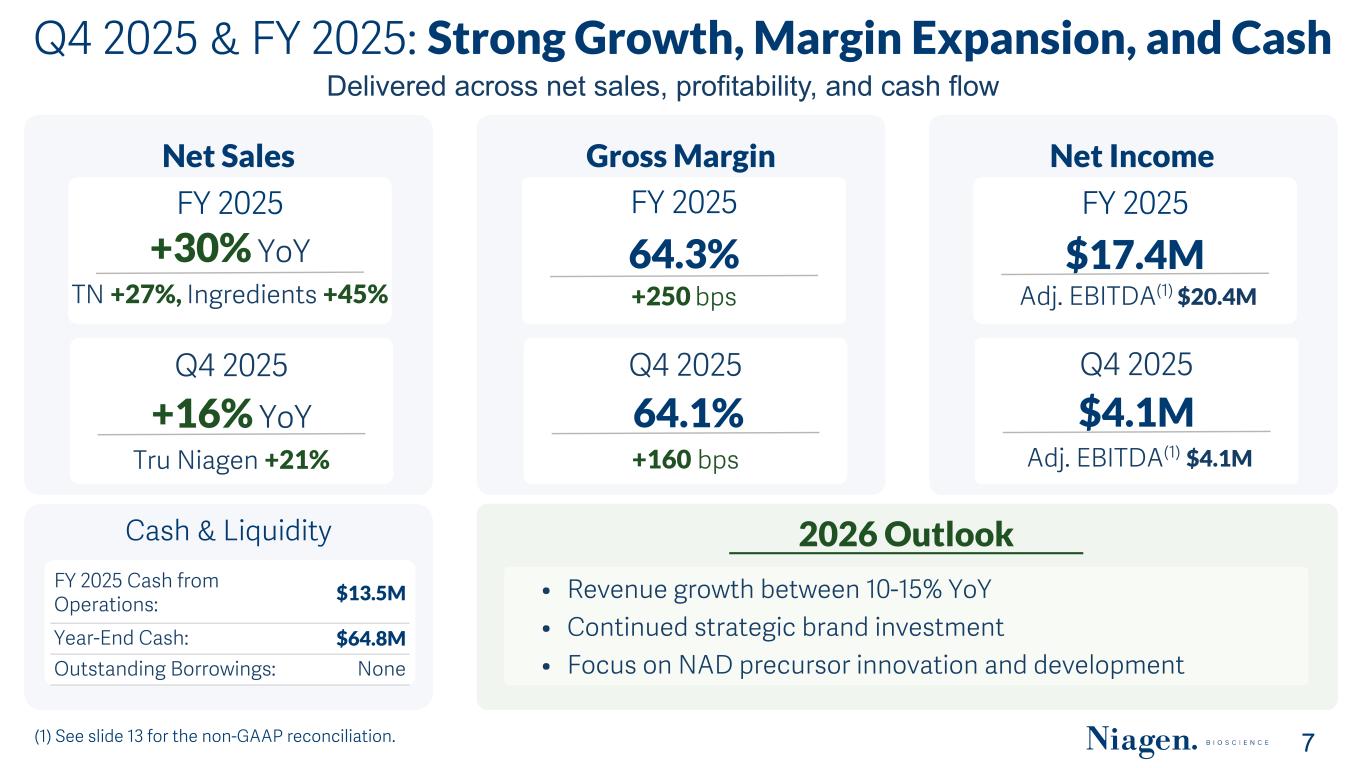
Q4 2025 & FY 2025: Strong Growth, Margin Expansion, and Cash (1) See slide 13 for the non-GAAP reconciliation. 7 Net Sales Q4 2025 +16% YoY Tru Niagen +21% FY 2025 +30% YoY TN +27%, Ingredients +45% Gross Margin Q4 2025 64.1% +160 bps FY 2025 64.3% +250 bps Net Income Q4 2025 $4.1M Adj. EBITDA(1) $4.1M FY 2025 $17.4M Adj. EBITDA(1) $20.4M Cash & Liquidity FY 2025 Cash from Operations: $13.5M Year-End Cash: $64.8M Outstanding Borrowings: None 2026 Outlook • Revenue growth between 10-15% YoY • Continued strategic brand investment • Focus on NAD precursor innovation and development Delivered across net sales, profitability, and cash flow

Financial Highlights 8
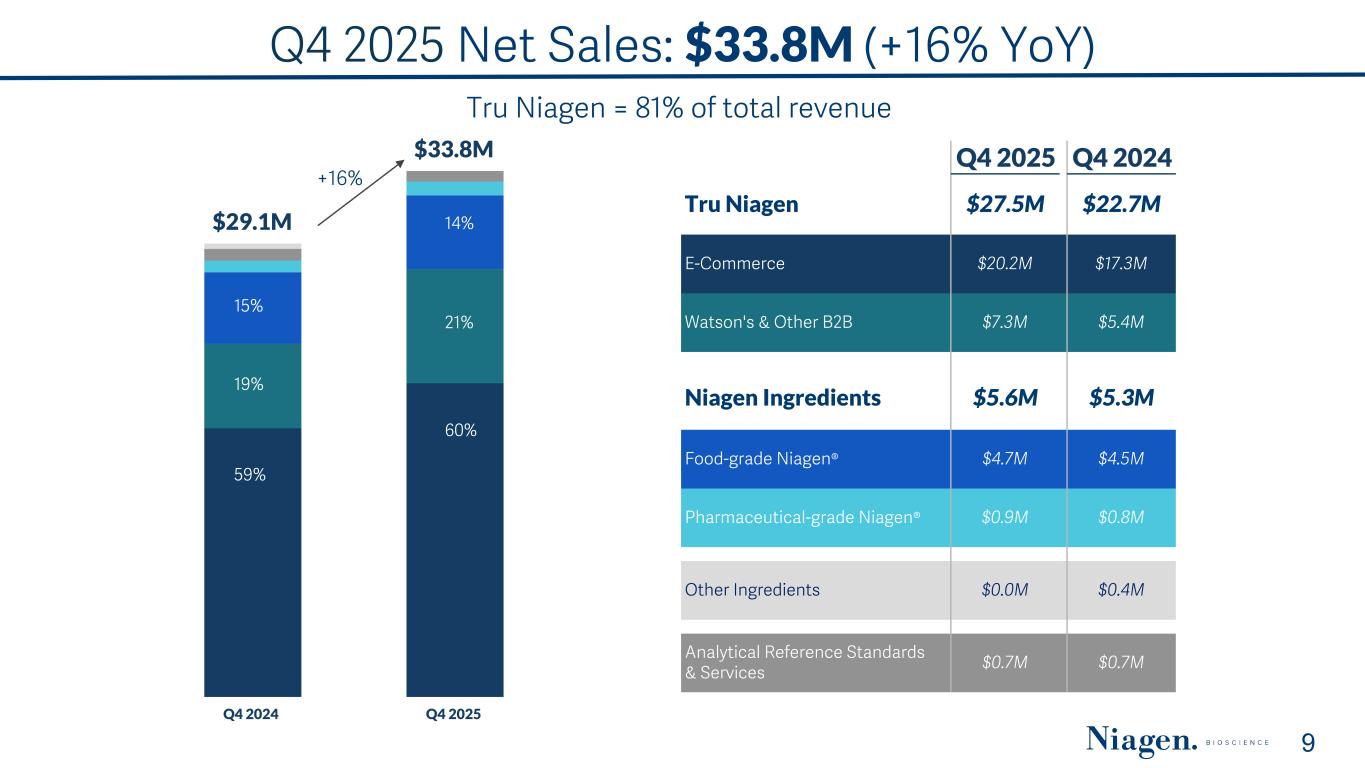
Q4 2025 Net Sales: $33.8M (+16% YoY) 9 Tru Niagen = 81% of total revenue Q4 2025 Q4 2024 Tru Niagen $27.5M $22.7M E-Commerce $20.2M $17.3M Watson's & Other B2B $7.3M $5.4M Niagen Ingredients $5.6M $5.3M Food-grade Niagen® $4.7M $4.5M Pharmaceutical-grade Niagen® $0.9M $0.8M Other Ingredients $0.0M $0.4M Analytical Reference Standards & Services $0.7M $0.7M $29.1M $33.8M Q4 2024 Q4 2025 59% 19% 15% 60% 21% 14% +16%
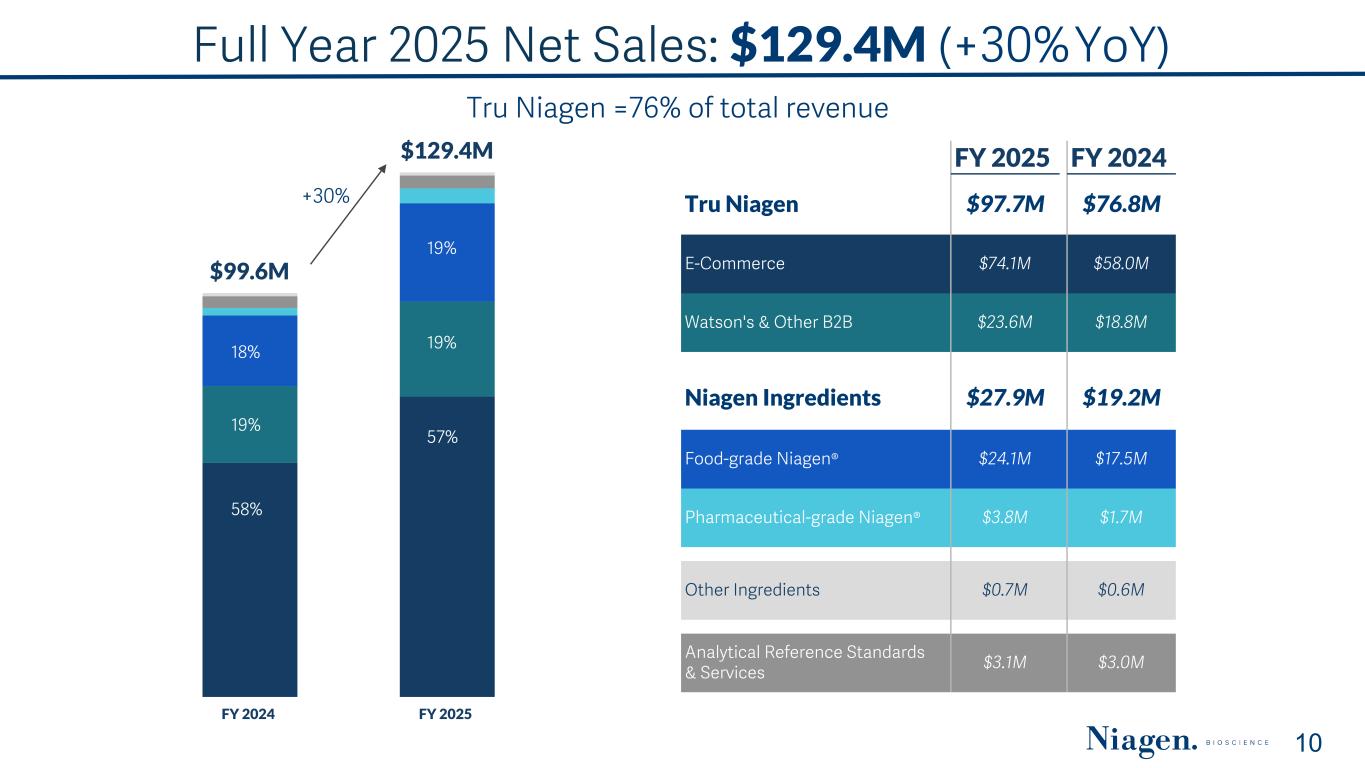
Full Year 2025 Net Sales: $129.4M (+30% YoY) 10 $99.6M $129.4M FY 2024 FY 2025 +30% 58% 19% 18% 57% 19% 19% Tru Niagen =76% of total revenue FY 2025 FY 2024 Tru Niagen $97.7M $76.8M E-Commerce $74.1M $58.0M Watson's & Other B2B $23.6M $18.8M Niagen Ingredients $27.9M $19.2M Food-grade Niagen® $24.1M $17.5M Pharmaceutical-grade Niagen® $3.8M $1.7M Other Ingredients $0.7M $0.6M Analytical Reference Standards & Services $3.1M $3.0M
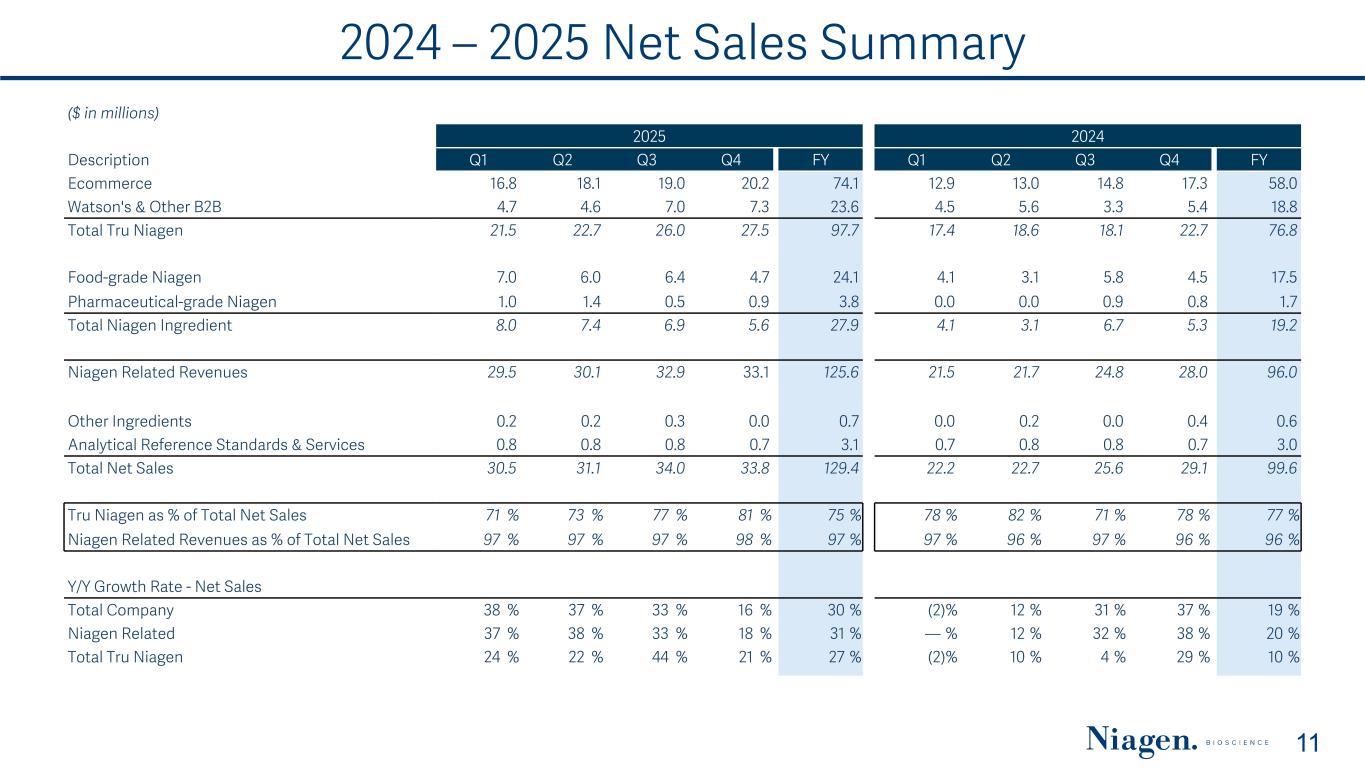
2024 – 2025 Net Sales Summary 11 ($ in millions) 2025 2024 Description Q1 Q2 Q3 Q4 FY Q1 Q2 Q3 Q4 FY Ecommerce 16.8 18.1 19.0 20.2 74.1 12.9 13.0 14.8 17.3 58.0 Watson's & Other B2B 4.7 4.6 7.0 7.3 23.6 4.5 5.6 3.3 5.4 18.8 Total Tru Niagen 21.5 22.7 26.0 27.5 97.7 17.4 18.6 18.1 22.7 76.8 Food-grade Niagen 7.0 6.0 6.4 4.7 24.1 4.1 3.1 5.8 4.5 17.5 Pharmaceutical-grade Niagen 1.0 1.4 0.5 0.9 3.8 0.0 0.0 0.9 0.8 1.7 Total Niagen Ingredient 8.0 7.4 6.9 5.6 27.9 4.1 3.1 6.7 5.3 19.2 Niagen Related Revenues 29.5 30.1 32.9 33.1 125.6 21.5 21.7 24.8 28.0 96.0 Other Ingredients 0.2 0.2 0.3 0.0 0.7 0.0 0.2 0.0 0.4 0.6 Analytical Reference Standards & Services 0.8 0.8 0.8 0.7 3.1 0.7 0.8 0.8 0.7 3.0 Total Net Sales 30.5 31.1 34.0 33.8 129.4 22.2 22.7 25.6 29.1 99.6 Tru Niagen as % of Total Net Sales 71 % 73 % 77 % 81 % 75 % 78 % 82 % 71 % 78 % 77 % Niagen Related Revenues as % of Total Net Sales 97 % 97 % 97 % 98 % 97 % 97 % 96 % 97 % 96 % 96 % Y/Y Growth Rate - Net Sales Total Company 38 % 37 % 33 % 16 % 30 % (2) % 12 % 31 % 37 % 19 % Niagen Related 37 % 38 % 33 % 18 % 31 % — % 12 % 32 % 38 % 20 % Total Tru Niagen 24 % 22 % 44 % 21 % 27 % (2) % 10 % 4 % 29 % 10 %
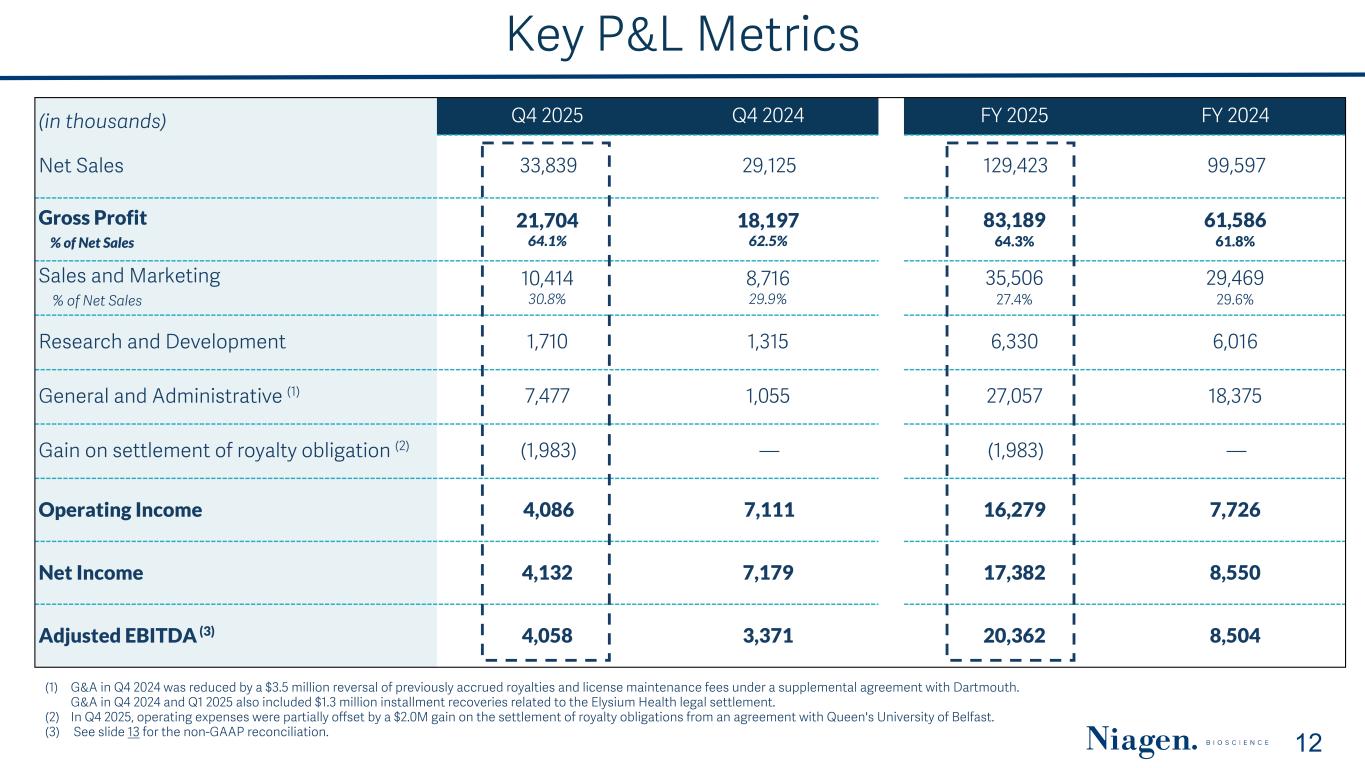
(in thousands) Q4 2025 Q4 2024 FY 2025 FY 2024 Net Sales 33,839 29,125 129,423 99,597 Gross Profit % of Net Sales 21,704 64.1% 18,197 62.5% 83,189 64.3% 61,586 61.8% Sales and Marketing % of Net Sales 10,414 30.8% 8,716 29.9% 35,506 27.4% 29,469 29.6% Research and Development 1,710 1,315 6,330 6,016 General and Administrative (1) 7,477 1,055 27,057 18,375 Gain on settlement of royalty obligation (2) (1,983) — (1,983) — Operating Income 4,086 7,111 16,279 7,726 Net Income 4,132 7,179 17,382 8,550 Adjusted EBITDA (3) 4,058 3,371 20,362 8,504 Key P&L Metrics 12 (1) G&A in Q4 2024 was reduced by a $3.5 million reversal of previously accrued royalties and license maintenance fees under a supplemental agreement with Dartmouth. G&A in Q4 2024 and Q1 2025 also included $1.3 million installment recoveries related to the Elysium Health legal settlement. (2) In Q4 2025, operating expenses were partially offset by a $2.0M gain on the settlement of royalty obligations from an agreement with Queen's University of Belfast. (3) See slide 13 for the non-GAAP reconciliation.
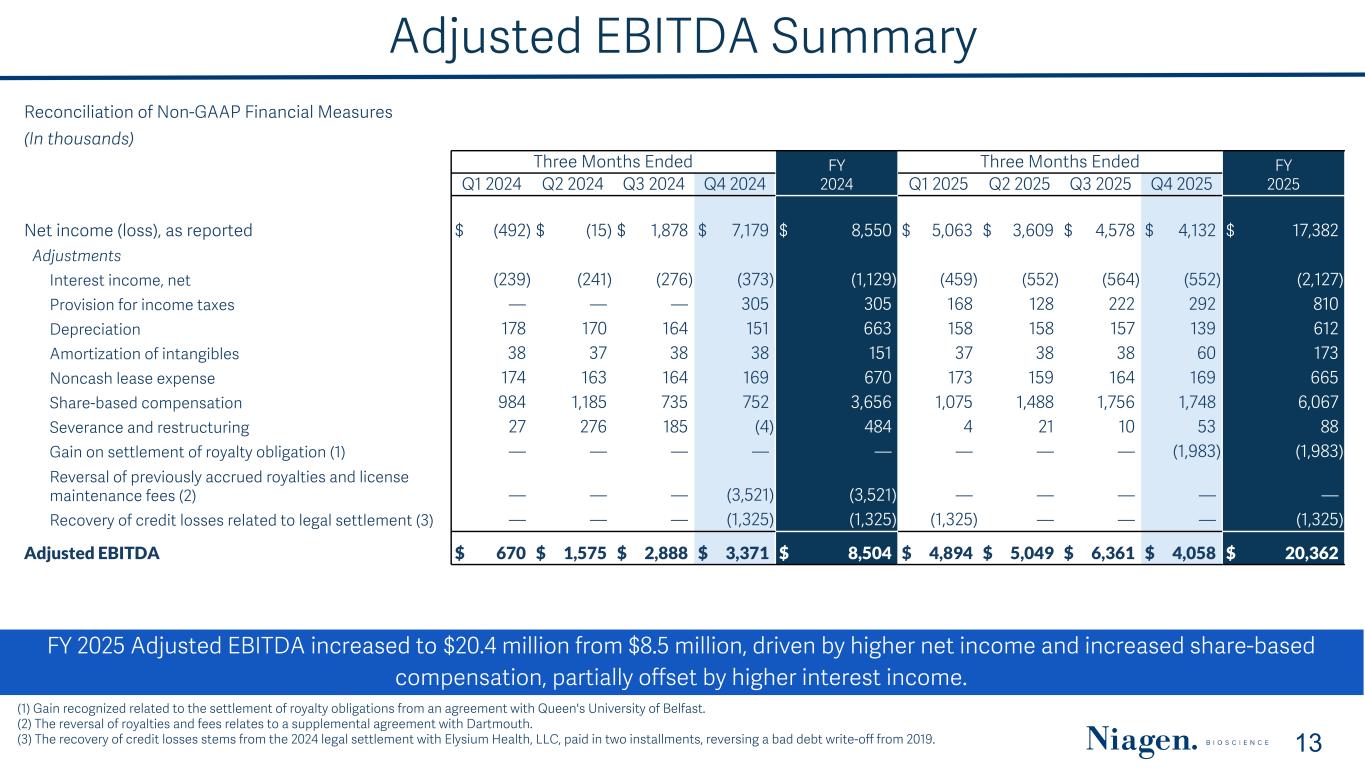
Adjusted EBITDA Summary FY 2025 Adjusted EBITDA increased to $20.4 million from $8.5 million, driven by higher net income and increased share-based compensation, partially offset by higher interest income. 13 (1) Gain recognized related to the settlement of royalty obligations from an agreement with Queen's University of Belfast. (2) The reversal of royalties and fees relates to a supplemental agreement with Dartmouth. (3) The recovery of credit losses stems from the 2024 legal settlement with Elysium Health, LLC, paid in two installments, reversing a bad debt write-off from 2019. Reconciliation of Non-GAAP Financial Measures (In thousands) Three Months Ended FY 2024 Three Months Ended FY 2025Q1 2024 Q2 2024 Q3 2024 Q4 2024 Q1 2025 Q2 2025 Q3 2025 Q4 2025 Net income (loss), as reported $ (492) $ (15) $ 1,878 $ 7,179 $ 8,550 $ 5,063 $ 3,609 $ 4,578 $ 4,132 $ 17,382 Adjustments Interest income, net (239) (241) (276) (373) (1,129) (459) (552) (564) (552) (2,127) Provision for income taxes — — — 305 305 168 128 222 292 810 Depreciation 178 170 164 151 663 158 158 157 139 612 Amortization of intangibles 38 37 38 38 151 37 38 38 60 173 Noncash lease expense 174 163 164 169 670 173 159 164 169 665 Share-based compensation 984 1,185 735 752 3,656 1,075 1,488 1,756 1,748 6,067 Severance and restructuring 27 276 185 (4) 484 4 21 10 53 88 Gain on settlement of royalty obligation (1) — — — — — — — — (1,983) (1,983) Reversal of previously accrued royalties and license maintenance fees (2) — — — (3,521) (3,521) — — — — — Recovery of credit losses related to legal settlement (3) — — — (1,325) (1,325) (1,325) — — — (1,325) Adjusted EBITDA $ 670 $ 1,575 $ 2,888 $ 3,371 $ 8,504 $ 4,894 $ 5,049 $ 6,361 $ 4,058 $ 20,362
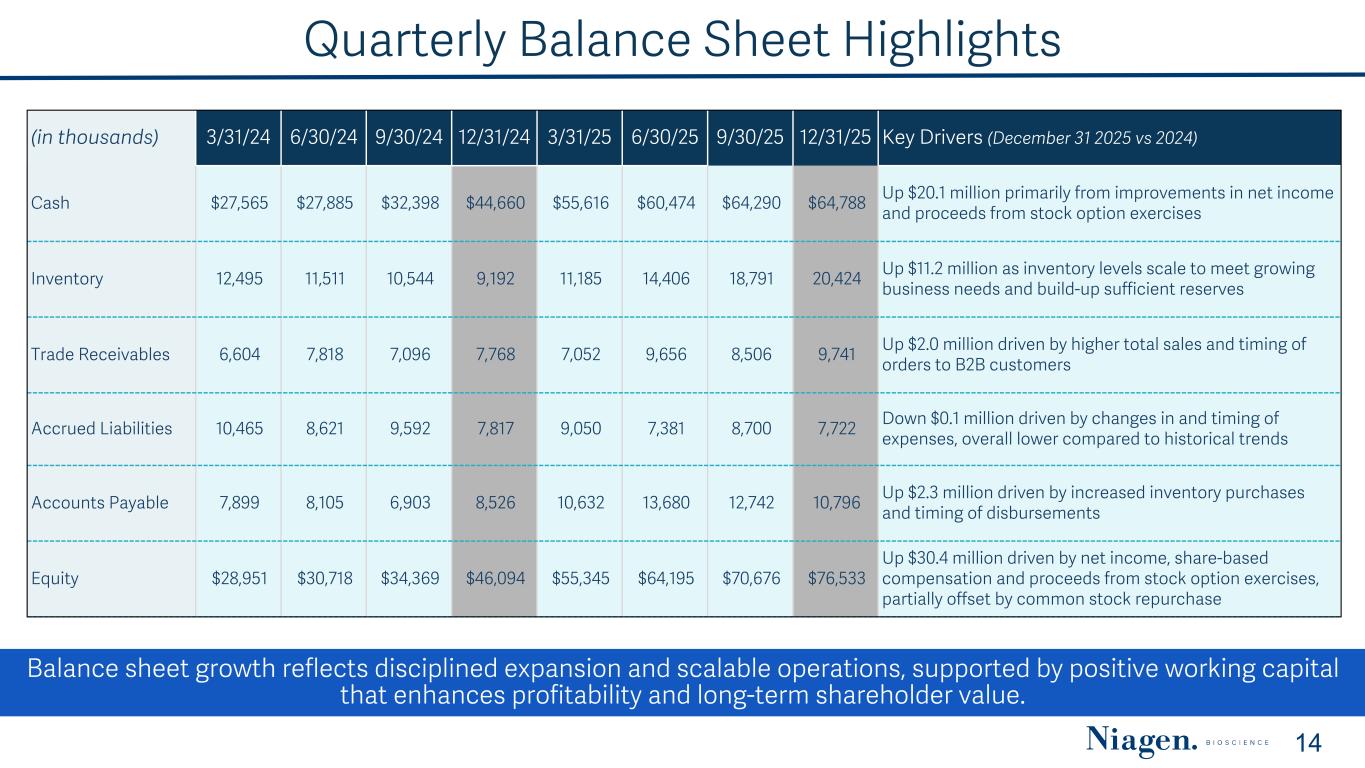
Quarterly Balance Sheet Highlights (in thousands) 3/31/24 6/30/24 9/30/24 12/31/24 3/31/25 6/30/25 9/30/25 12/31/25 Key Drivers (December 31 2025 vs 2024) Cash $27,565 $27,885 $32,398 $44,660 $55,616 $60,474 $64,290 $64,788 Up $20.1 million primarily from improvements in net income and proceeds from stock option exercises Inventory 12,495 11,511 10,544 9,192 11,185 14,406 18,791 20,424 Up $11.2 million as inventory levels scale to meet growing business needs and build-up sufficient reserves Trade Receivables 6,604 7,818 7,096 7,768 7,052 9,656 8,506 9,741 Up $2.0 million driven by higher total sales and timing of orders to B2B customers Accrued Liabilities 10,465 8,621 9,592 7,817 9,050 7,381 8,700 7,722 Down $0.1 million driven by changes in and timing of expenses, overall lower compared to historical trends Accounts Payable 7,899 8,105 6,903 8,526 10,632 13,680 12,742 10,796 Up $2.3 million driven by increased inventory purchases and timing of disbursements Equity $28,951 $30,718 $34,369 $46,094 $55,345 $64,195 $70,676 $76,533 Up $30.4 million driven by net income, share-based compensation and proceeds from stock option exercises, partially offset by common stock repurchase Balance sheet growth reflects disciplined expansion and scalable operations, supported by positive working capital that enhances profitability and long-term shareholder value. 14
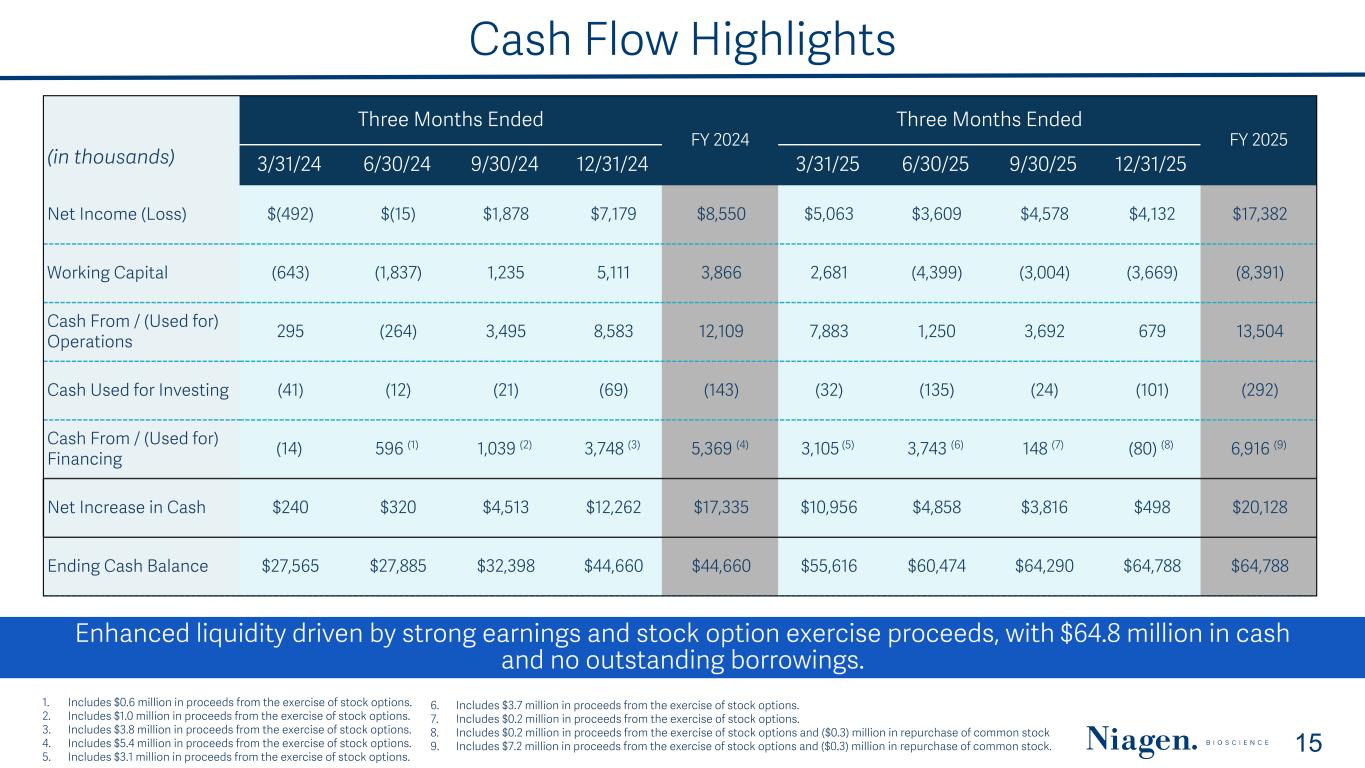
Cash Flow Highlights Three Months Ended FY 2024 Three Months Ended FY 2025 (in thousands) 3/31/24 6/30/24 9/30/24 12/31/24 3/31/25 6/30/25 9/30/25 12/31/25 Net Income (Loss) $(492) $(15) $1,878 $7,179 $8,550 $5,063 $3,609 $4,578 $4,132 $17,382 Working Capital (643) (1,837) 1,235 5,111 3,866 2,681 (4,399) (3,004) (3,669) (8,391) Cash From / (Used for) Operations 295 (264) 3,495 8,583 12,109 7,883 1,250 3,692 679 13,504 Cash Used for Investing (41) (12) (21) (69) (143) (32) (135) (24) (101) (292) Cash From / (Used for) Financing (14) 596 (1) 1,039 (2) 3,748 (3) 5,369 (4) 3,105 (5) 3,743 (6) 148 (7) (80) (8) 6,916 (9) Net Increase in Cash $240 $320 $4,513 $12,262 $17,335 $10,956 $4,858 $3,816 $498 $20,128 Ending Cash Balance $27,565 $27,885 $32,398 $44,660 $44,660 $55,616 $60,474 $64,290 $64,788 $64,788 Enhanced liquidity driven by strong earnings and stock option exercise proceeds, with $64.8 million in cash and no outstanding borrowings. 15 1. Includes $0.6 million in proceeds from the exercise of stock options. 2. Includes $1.0 million in proceeds from the exercise of stock options. 3. Includes $3.8 million in proceeds from the exercise of stock options. 4. Includes $5.4 million in proceeds from the exercise of stock options. 5. Includes $3.1 million in proceeds from the exercise of stock options. 6. Includes $3.7 million in proceeds from the exercise of stock options. 7. Includes $0.2 million in proceeds from the exercise of stock options. 8. Includes $0.2 million in proceeds from the exercise of stock options and ($0.3) million in repurchase of common stock 9. Includes $7.2 million in proceeds from the exercise of stock options and ($0.3) million in repurchase of common stock.
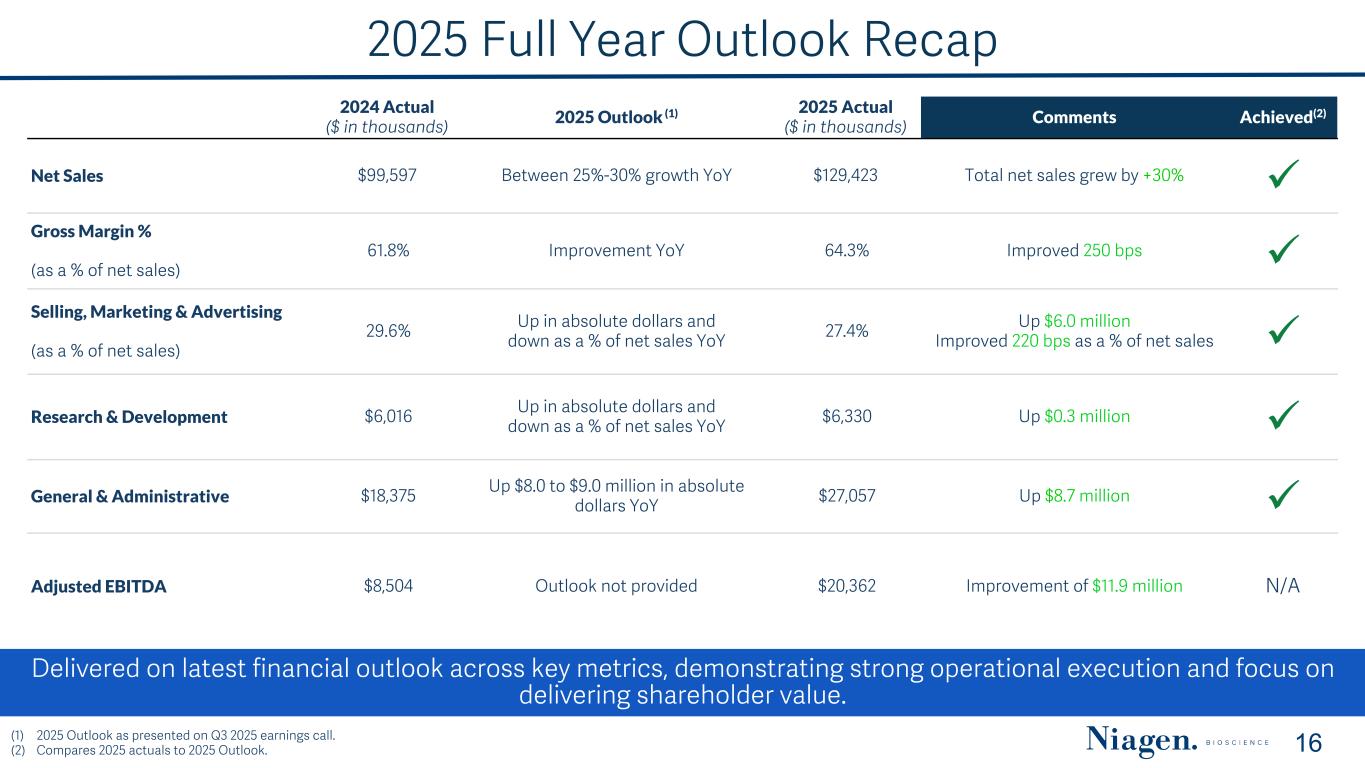
2025 Full Year Outlook Recap Delivered on latest financial outlook across key metrics, demonstrating strong operational execution and focus on delivering shareholder value. 16 2024 Actual ($ in thousands) 2025 Outlook (1) 2025 Actual ($ in thousands) Comments Achieved(2) Net Sales $99,597 Between 25%-30% growth YoY $129,423 Total net sales grew by +30% ü Gross Margin % (as a % of net sales) 61.8% Improvement YoY 64.3% Improved 250 bps ü Selling, Marketing & Advertising (as a % of net sales) 29.6% Up in absolute dollars and down as a % of net sales YoY 27.4% Up $6.0 million Improved 220 bps as a % of net sales ü Research & Development $6,016 Up in absolute dollars and down as a % of net sales YoY $6,330 Up $0.3 million ü General & Administrative $18,375 Up $8.0 to $9.0 million in absolute dollars YoY $27,057 Up $8.7 million ü Adjusted EBITDA $8,504 Outlook not provided $20,362 Improvement of $11.9 million N/A (1) 2025 Outlook as presented on Q3 2025 earnings call. (2) Compares 2025 actuals to 2025 Outlook.
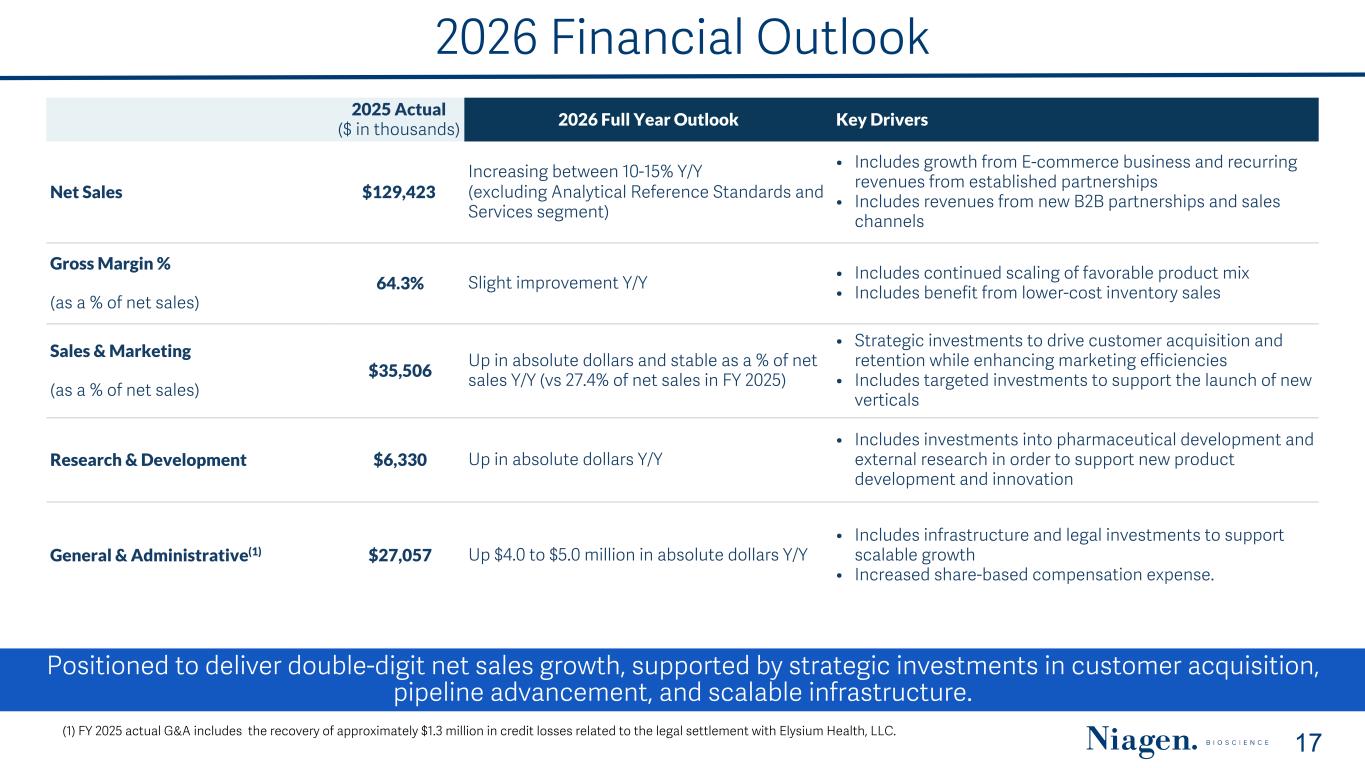
Positioned to deliver double-digit net sales growth, supported by strategic investments in customer acquisition, pipeline advancement, and scalable infrastructure. 2026 Financial Outlook 17(1) FY 2025 actual G&A includes the recovery of approximately $1.3 million in credit losses related to the legal settlement with Elysium Health, LLC. 2025 Actual ($ in thousands) 2026 Full Year Outlook Key Drivers Net Sales $129,423 Increasing between 10-15% Y/Y (excluding Analytical Reference Standards and Services segment) • Includes growth from E-commerce business and recurring revenues from established partnerships • Includes revenues from new B2B partnerships and sales channels Gross Margin % (as a % of net sales) 64.3% Slight improvement Y/Y • Includes continued scaling of favorable product mix • Includes benefit from lower-cost inventory sales Sales & Marketing (as a % of net sales) $35,506 Up in absolute dollars and stable as a % of net sales Y/Y (vs 27.4% of net sales in FY 2025) • Strategic investments to drive customer acquisition and retention while enhancing marketing efficiencies • Includes targeted investments to support the launch of new verticals Research & Development $6,330 Up in absolute dollars Y/Y • Includes investments into pharmaceutical development and external research in order to support new product development and innovation General & Administrative(1) $27,057 Up $4.0 to $5.0 million in absolute dollars Y/Y • Includes infrastructure and legal investments to support scalable growth • Increased share-based compensation expense.

The information contained in this documents is confidential, privileged and only for the information of the intended recipient and may not be used, published or redistributed without the prior written consent (2019) 18 The Science

Scientific Advisory Board 19
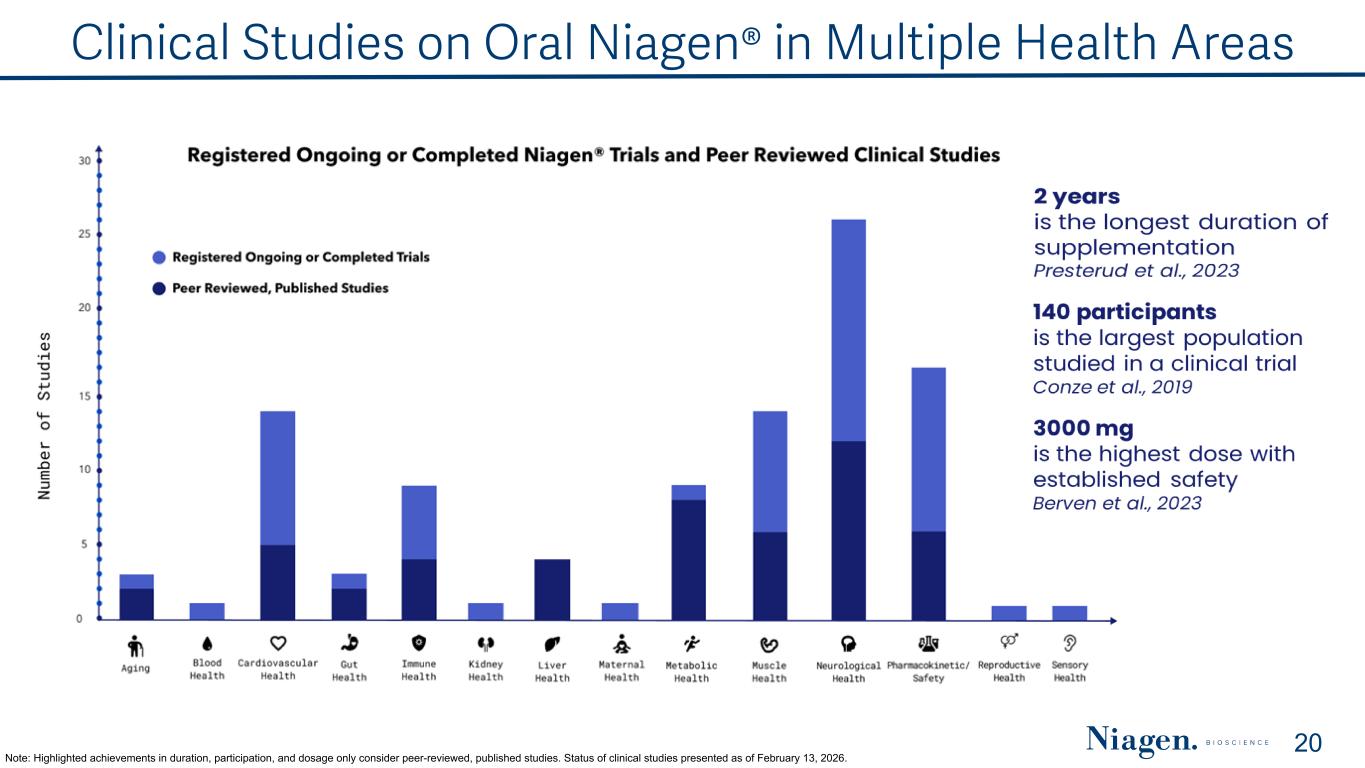
Clinical Studies on Oral Niagen® in Multiple Health Areas Note: Highlighted achievements in duration, participation, and dosage only consider peer-reviewed, published studies. Status of clinical studies presented as of February 13, 2026. 20

Contact Info: Niagen Bioscience Investor Relations: KCSA Strategic Communications Valter Pinto, Managing Director T: +1 (212) 896-1254 Niagen@kcsa.com www.niagenbioscience.com Where to purchase Tru Niagen® TruNiagen.com Find Health Clinics Offering Niagen® Plus NiagenPlus.com 21