
2025 Annual Report
Leading
Antibody
Science
for Better
Futures
CVR No. 21 02 38 84 Genmab A/S Carl Jacobsens Vej 30 2500 Valby Denmark


 | Genmab 2025 Annual Report | 2 | |||||
Expanding the Reach of Our Medicines | ||
Our Reporting Suite | |
 | 2025 Corporate Governance Report |
 | 2025 Compensation Report |
Our Corporate Governance and Compensation Reports for 2025 can also be found on our website Genmab.com. | |

Genmab 2025 Annual Report | 3 | ||||||
In this section | ||
Expanding the Reach of Our Medicines | ||
 | Genmab 2025 Annual Report | 4 | |||||
Our 2030 Vision By 2030, our KYSO antibody medicines® are fundamentally transforming the lives of people with cancer and other serious diseases. | Our Core Purpose, Supporting Our 2030 Vision Our unstoppable team will improve the lives of patients through innovative and differentiated antibody therapeutics. | |
 |
1999 – 2009 | 2010 – 2020 | 2021 – 2025 |

Genmab 2025 Annual Report | 5 | ||||||

Genmab 2025 Annual Report | 6 | ||||||

Genmab 2025 Annual Report | 7 | ||||||

 | Genmab 2025 Annual Report | 8 | |||||
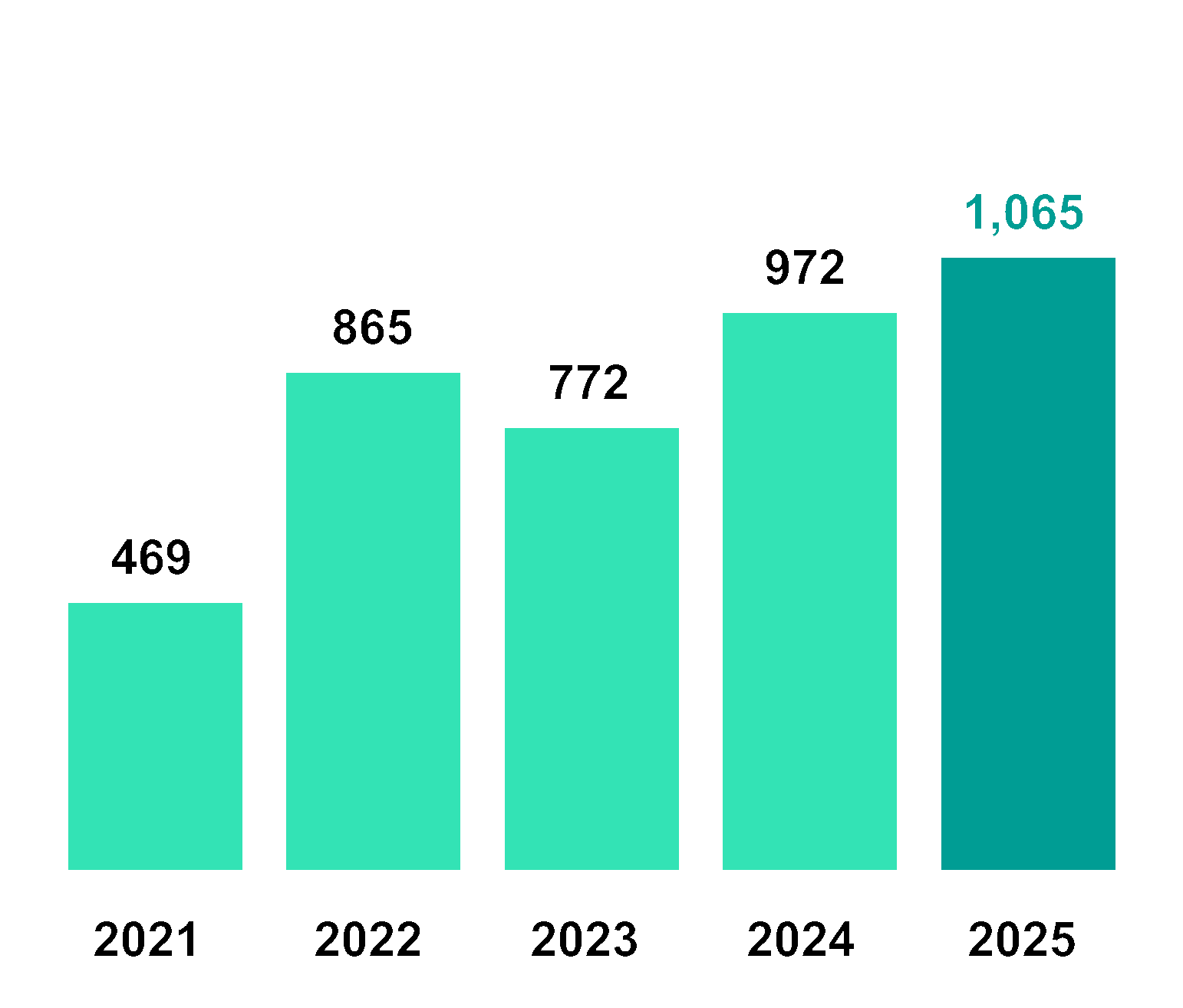
Financial | Operating Profit | ||||||
USD | USD | USD | (USD Million) | ||||
20.5B | 3,720M | 2,219M | |||||
2025 year-end market cap | 2025 revenue | 2025 adjusted operating expenses1, 72% invested in R&D | |||||
Liquidity and Capital Resources | |||||||
USD | USD | ||||||
1,715M | 5,847M | ||||||
Cash and cash equivalents | Shareholders’ equity | ||||||
1.Operating Expenses exclude 2025 charges related to: 1) acquisition and integration-related charges of $185 million and 2) amortization of intangible assets acquired through acquisitions of $13 million. | |||||||
 | Genmab 2025 Annual Report | 9 | |||||
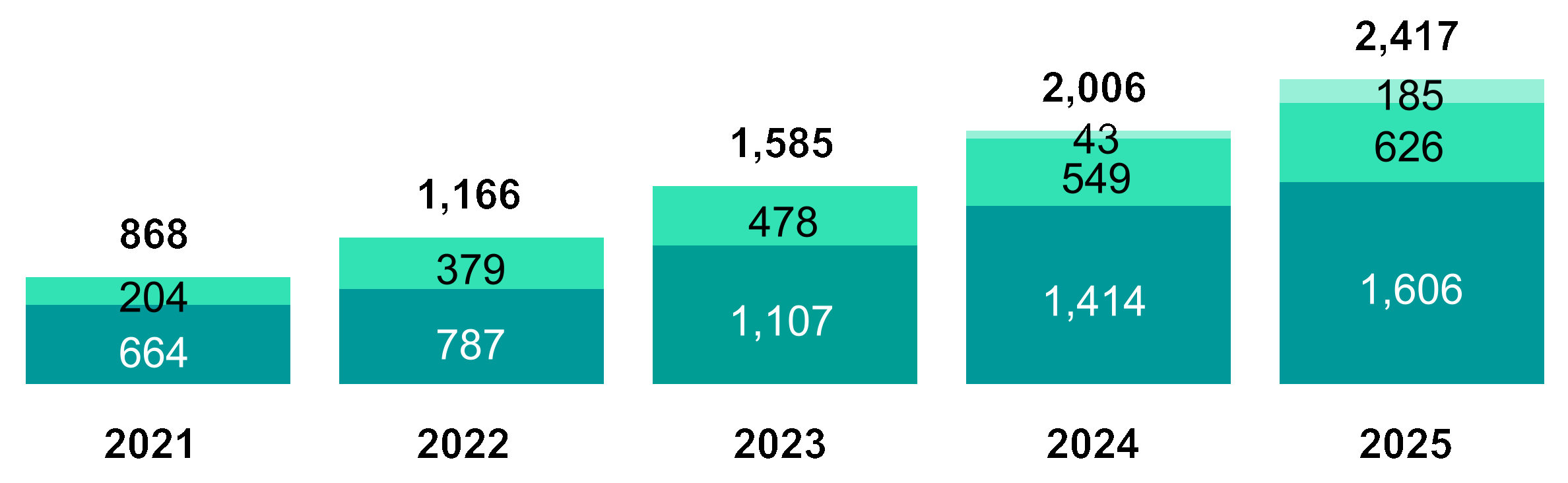
(USD Millions) | 2021 | 2022 | 2023 | 2024 | 2025 |
Income Statement | |||||
Revenue | 1,337 | 2,031 | 2,390 | 3,121 | 3,720 |
Cost of product sales | — | — | (33) | (143) | (238) |
Research and development expenses | (664) | (787) | (1,107) | (1,414) | (1,606) |
Selling, general and administrative expenses | (204) | (379) | (478) | (549) | (626) |
Acquisition and integration related charges | — | — | — | (43) | (185) |
Total costs and operating expenses | (868) | (1,166) | (1,618) | (2,149) | (2,655) |
Operating profit | 469 | 865 | 772 | 972 | 1,065 |
Net financial items | 153 | 96 | 45 | 354 | 139 |
Net profit | 470 | 750 | 631 | 1,133 | 963 |
Balance Sheet | |||||
Total non-current assets | 300 | 273 | 320 | 2,514 | 9,988 |
Marketable securities | 1,650 | 1,783 | 1,967 | 1,574 | — |
Cash and cash equivalents | 1,423 | 1,419 | 2,204 | 1,380 | 1,715 |
Total assets | 3,899 | 4,321 | 5,232 | 6,414 | 12,873 |
Borrowings | — | — | — | — | 5,274 |
Share capital | 10 | 10 | 10 | 10 | 10 |
Shareholders' equity | 3,405 | 3,915 | 4,687 | 5,137 | 5,847 |
Cash Flow Statement | |||||
Investment in acquisitions, net of cash acquired | — | — | — | (1,783) | (7,215) |
Cash flow from operating activities | 354 | 555 | 1,071 | 1,126 | 1,186 |
Cash flow from investing activities | (153) | (392) | (185) | (1,447) | (5,643) |
Cash flow from financing activities | (67) | (110) | (89) | (566) | 4,789 |
Investments in intangible assets | — | — | (1) | (17) | (18) |
Investments in tangible assets | (40) | (45) | (53) | (27) | (37) |
Financial Ratios and Other Information | |||||
Basic net profit per share | 7.19 | 11.47 | 9.67 | 17.66 | 15.50 |
Diluted net profit per share | 7.12 | 11.36 | 9.58 | 17.53 | 15.37 |
Year-end share market price | 2,630.00 | 2,941.00 | 2,155.00 | 1,492.50 | 2,027.00 |
Price/book value | 7.72 | 7.51 | 4.60 | 2.91 | 3.47 |
Shareholders' equity per share | 340.50 | 391.50 | 468.70 | 513.70 | 584.70 |
Equity ratio | 87% | 91% | 90% | 80% | 45% |
Shares outstanding | 65,718,456 | 65,961,573 | 66,074,535 | 66,187,186 | 64,238,408 |
Average number of employees (FTE)¹ | 1,022 | 1,460 | 2,011 | 2,535 | 2,694 |
Number of employees (FTE) at year-end | 1,212 | 1,660 | 2,204 | 2,682 | 3,029 |
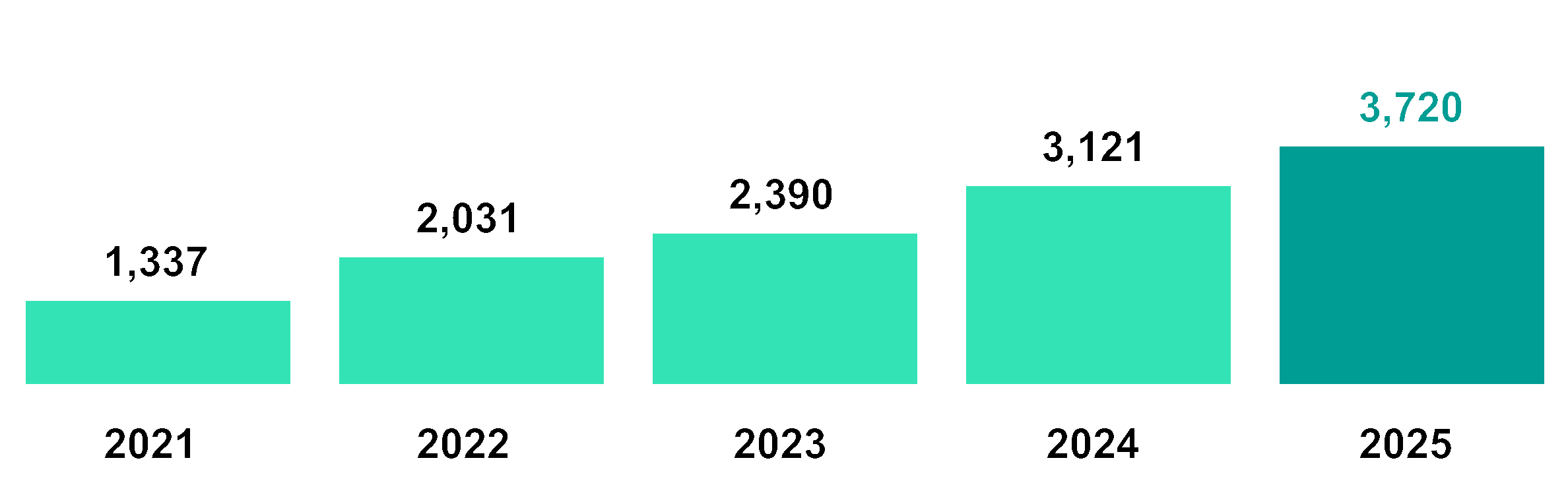
n | Research and development expenses | n | Selling, general and administrative expenses | n | Acquisition and integration related charges |
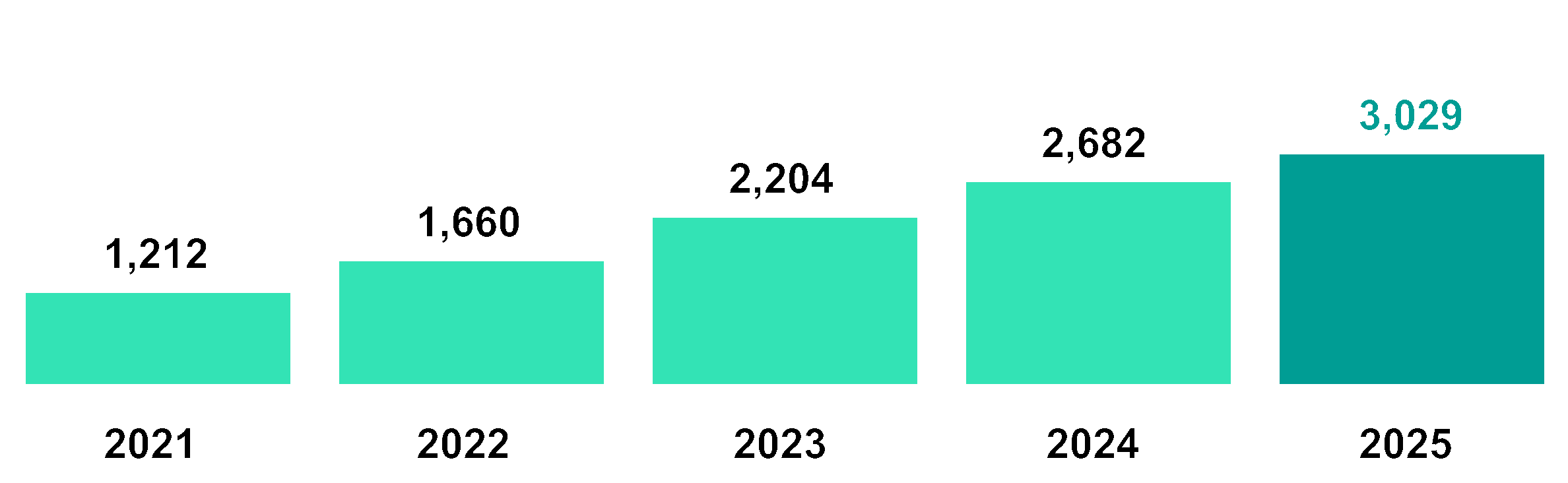

 | Genmab 2025 Annual Report | 10 | |||||
(USD millions) | 2025 Actual Result | 2025 Adjusted Result2 | 2026 Guidance2 | 2026 Guidance Mid-Point2 | |
Revenue | 3,720 | 3,720 | 4,065 - 4,395 | 4,230 | |
Royalties | 3,102 | 3,102 | 3,440 - 3,685 | 3,563 | |
Net product sales/ Collaboration revenue1 | 468 | 468 | 490 - 555 | 522 | |
Milestones/ Reimbursement revenue | 150 | 150 | 135 - 155 | 145 | |
Gross profit | 3,482 | 3,482 | 3,810 - 4,110 | 3,960 | |
Operating expenses | (2,417) | (2,219) | (2,710) - (2,910) | (2,810) | |
Operating profit | 1,065 | 1,263 | 900 - 1,400 | 1,150 |
 | Genmab 2025 Annual Report | 11 | |||||
 | Genmab 2025 Annual Report | 12 | |||||
Research Track record of success and investing for tomorrow | Development Scaled up capabilities to expand from early- to late-stage development | Technical Operations and Commercialization Capabilities in place to support commercialized medicines | ||||
Enabling functions: Supporting growth and managing risk | ||||||

 | Genmab 2025 Annual Report | 13 | |||||

Genmab 2025 Annual Report | 14 | ||||||

Inspired by Nature At Genmab, we are inspired by nature and understand how antibodies work. We are deeply knowledgeable about antibody biology and our scientists harness this expertise to create and develop differentiated investigational antibody medicines. We utilize a sophisticated and highly automated process to efficiently generate, select, produce, and evaluate antibody-based products. Our teams have established a fully integrated R&D enterprise and streamlined process to coordinate the activities of antibody product discovery, preclinical testing, manufacturing, clinical trial design and execution, and regulatory submissions across Genmab’s international operations. We have expanded our scientific focus to use data science and AI to aid in the discovery of new targets and biomarkers and bolster our in-depth precision medicine and translational laboratory capabilities. Through our expertise in antibody drug development, we pioneer technologies that allow us to create differentiated and potentially first-in-class or best- in-class investigational medicines with the potential to improve patients’ lives. Our antibody expertise has enabled us to create our cutting-edge technology platforms: DuoBody, HexaBody, DuoHexaBody® and HexElect®. With our acquisition of ProfoundBio we gained novel ADC technology platforms. We gained additional proprietary technology platforms as part of our acquisition of Merus. Additional information about our technologies is available on Genmab’s website, genmab.com/ antibody-science/antibody-technology-platforms. | ||

 | Genmab 2025 Annual Report | 15 | |||||
 | Genmab 2025 Annual Report | 16 | |||||


Follicular Lymphoma Follicular lymphoma is the second most common non-Hodgkin lymphoma and is considered incurable, underscoring the need for new treatments. | ||
 | ||
Cervical Cancer Support: CeMe™ The CeMe campaign in the U.S., created with Pfizer, creates connection and community for those affected by cervical cancer. “To be diagnosed with cervical cancer… knocked the wind out of me,” said Karen. “Sharing your story… lets people know that there are other people out there that can support you.” | ||
youtube.com/cemestories | ||
 | Genmab 2025 Annual Report | 17 | |||||

Tivdak Tivdak is the first and only ADC approved in the US, Europe, and Japan for the treatment of recurrent or metastatic cervical cancer after prior therapy and is the only ADC with demonstrated overall survival data in this setting compared to chemotherapy. | ||

Our Approach to Value, Access, and Pricing •Value: The value of our medicines is driven by our innovative science. •Access: Patient impact happens when our medicines reach the people who need them and help them live better. •Pricing: The price of our medicines reflects the innovation behind our science, its impact on patients, and our commitment to bringing that science to patients. | ||
Elevating Patient Voices Our Patient Advisory Council gives patients a seat at the table, ensuring their insights and experiences will help guide our work, from trial design to how we support and deliver our medicines. “Serving on Genmab’s Patient Advisory Council means the patient voice will have a direct influence on what comes next,” said Jim Zervanos. “It’s empowering and inspiring to know my experience will help shape decisions that can benefit others like me.” | ||
 | ||


 | Genmab 2025 Annual Report | 18 | |||||

Genmab 2025 Annual Report | 19 | ||||||
Pipeline At the end of 2025, Genmab’s proprietary pipeline of investigational medicines, where we are responsible for at least 50% of development, consisted of five antibody products in active clinical development. Our approved medicines are EPKINLY/TEPKINLY, which Genmab is co-developing and co-commercializing in the US and Japan in collaboration with AbbVie and Tivdak, which Genmab is co-developing globally and co-promoting in the US in collaboration with Pfizer and exclusively by Genmab outside of the US and China. In addition to our own pipeline, there are multiple investigational medicines in development by global pharmaceutical and biotechnology companies and six approved medicines powered by Genmab’s technology and innovations. BIZENGRI® (zenocutuzumab-zbco) was also added to our portfolio of royalty medicines as part of our acquisition of Merus. Beyond the investigational medicines in active clinical development, our pipeline includes multiple promising preclinical programs. An overview of the development status of our approved medicines and our late-stage investigational medicines is provided in the following sections. Detailed descriptions of dosing and efficacy and safety data from certain clinical trials have been disclosed in company announcements and media releases published via the Nasdaq Copenhagen stock exchange and may also be found in Genmab’s filings with the U.S. Securities and Exchange Commission (SEC). Additional information is available on Genmab’s website, genmab.com. The information accessible through our website is not part of and is not incorporated by reference herein. | ||

Genmab 2025 Annual Report | 20 | ||||||

Approved Product | Target | Developed By | Disease Indication(s)2 | |
EPKINLY (epcoritamab-bysp, epcoritamab) TEPKINLY (epcoritamab) | CD3xCD20 | Co-development Genmab/AbbVie | Approved in multiple territories including the US and Europe for adult patients with relapsed or refractory DLBCL after two or more lines of systemic therapy and in Japan for adult patients with certain types of relapsed or refractory large B-cell lymphoma (LBCL) after two or more lines of systemic therapy | |
Approved in multiple territories including the US, Europe and Japan for adult patients with relapsed or refractory FL after two or more lines of systemic therapy Approved in multiple territories including the US in combination with R2 for the treatment of adult patients with relapsed or refractory FL, following at least one prior systemic therapy | ||||
Tivdak (tisotumab vedotin-tftv, tisotumab vedotin) | TF | Co-development Genmab/ Pfizer | Approved in territories including the US, Europe and Japan for adult patients with recurrent/metastatic cervical cancer with disease progression on or after chemotherapy |

Genmab 2025 Annual Report | 21 | ||||||

Product | Developed By | Target(s) | Technology | Disease Indications | Most Advanced Development Phase | ||||||||
Preclinical | 1 | 2 | 3 | ||||||||||
Epcoritamab | Co-development Genmab/AbbVie | CD3, CD20 | DuoBody | Relapsed/refractory DLBCL | |||||||||
Relapsed/refractory FL | |||||||||||||
First line DLBCL | |||||||||||||
First line FL | |||||||||||||
Non-Hodgkin lymphoma (NHL) | |||||||||||||
Relapsed/refractory chronic lymphocytic leukemia (CLL) & Richter's Syndrome | |||||||||||||
Aggressive mature B-cell neoplasms in pediatric patients | |||||||||||||
Rinatabart Sesutecan (Rina-S, GEN1184) | Genmab | Folate receptor alpha (FRα) | ADC | PROC | |||||||||
Endometrial cancer | |||||||||||||
PSOC | |||||||||||||
NSCLC | |||||||||||||
Solid tumors | |||||||||||||
Petosemtamab | Genmab | Epidermal growth factor receptor (EGFR), leucine-rich repeat-containing G-protein coupled receptor 5 ( LGR5) | Biclonics® | Recurrent/metastatic head and neck squamous cell carcinoma (r/m HNSCC) | |||||||||
Advanced solid tumors including metastatic colorectal cancer (mCRC) | |||||||||||||
First line NSCLC with pembrolizumab | |||||||||||||
GEN1059 (BNT314) | Co-development Genmab/BioNTech SE (BioNTech) | Epithelial cell adhesion molecule (EpCAM), 4-1BB | DuoBody | Solid tumors | |||||||||
mCRC, in combination with pumitamig/chemo | |||||||||||||
GEN1057 | Genmab | Fibroblast activation protein alpha (FAPα), death receptor 4 (DR4) | DuoBody | Malignant solid tumors | |||||||||

Genmab 2025 Annual Report | 22 | ||||||

Approved Product | Discovered and/or Developed/Marketed By | Disease Indication(s)2 |
DARZALEX (daratumumab)/DARZALEX FASPRO (daratumumab and hyaluronidase-fihj) | J&J (Royalties to Genmab on global net sales) | Multiple myeloma |
Light-chain (AL) Amyloidosis | ||
Kesimpta (ofatumumab) | Novartis (Royalties to Genmab on global net sales) | Relapsing multiple sclerosis (RMS) |
TEPEZZA (teprotumumab-trbw) | Amgen (under sublicense from Roche, royalties to Genmab on global net sales) | Thyroid eye disease (TED) |
RYBREVANT (amivantamab/amivantamab-vmjw)/RYBREVANT FASPROTM (amivantamab and hyaluronidase-lpuj) | J&J (Royalties to Genmab on global net sales) | Advanced NSCLC with certain EGFR mutations |
TECVAYLI (teclistamab/teclistamab-cqyv) | J&J (Royalties to Genmab on global net sales) | Relapsed and refractory multiple myeloma |
TALVEY (talquetamab/talquetamab-tgvs) | J&J (Royalties to Genmab on global net sales) | Relapsed and refractory multiple myeloma |
BIZENGRI (zenocutuzumab-zbco) | Partner Therapeutics, Inc. (part of Genmab’s acquisition of Merus, royalties to Genmab on U.S. net sales) | Pancreatic adenocarcinoma and NSCLC that are advanced, unresectable or metastatic and harbor NRG1 gene fusions |
Product | Technology | Discovered and/or Developed By | Disease Indications | Most Advanced Development Phase | |||||
Preclinical | 1 | 2 | 3 | ||||||
Daratumumab | UltiMAb1 | J&J | Multiple myeloma | ||||||
AL Amyloidosis | |||||||||
Teprotumumab | UltiMAb | Amgen | TED | ||||||
Amivantamab | DuoBody | J&J | NSCLC | ||||||
Advanced or mCRC | |||||||||
Recurrent/metastatic head and neck cancer | |||||||||
Teclistamab | DuoBody | J&J | Multiple myeloma | ||||||
Talquetamab | DuoBody | J&J | Multiple myeloma | ||||||
Mim8 (denecimig) | DuoBody | Novo Nordisk | Hemophilia A | ||||||
Amlenetug (Lu AF82422) | UltiMAb | H. Lundbeck A/S (Lundbeck) | Multiple system atrophy | ||||||

Genmab 2025 Annual Report | 23 | ||||||


Genmab 2025 Annual Report | 24 | ||||||


Genmab 2025 Annual Report | 25 | ||||||


B-NHL Type | Stage | Development Phase | ||||||||
Preclinical | 1 | 2 | 3 | |||||||
DLBCL | Relapsed/Refractory | EPCORE DLBCL-1 | ||||||||
Front-line + R-CHOP | EPCORE DLBCL-2 | |||||||||
Relapsed/Refractory + lenalidomide, ASCT ineligible | EPCORE DLBCL-4 | |||||||||
Front-line +/- lenalidomide | EPCORE DLBCL-3 | |||||||||
FL | Relapsed/Refractory (Combo) | EPCORE FL-1 | ||||||||
Front-line +R2 | EPCORE FL-2 | |||||||||
DLBCL & FL | Outpatient | EPCORE NHL-6 | ||||||||
B-NHL | Relapsed/Progressive/Refractory | EPCORE NHL-1 | ||||||||
Relapsed/Progressive/Refractory (Japan) | EPCORE NHL-3 | |||||||||
Relapsed/Refractory Pediatric | EPCORE Peds-1 | |||||||||
Previously Untreated/Relapsed/Refractory (Combo) | EPCORE NHL-2 | |||||||||
Previously Untreated/Relapsed/Refractory (China) | EPCORE NHL-4 | |||||||||
Previously Untreated/Relapsed/Refractory (Combo) | EPCORE NHL-5 | |||||||||
CLL/Richter’s Syndrome | Relapsed/Refractory | EPCORE CLL-1 | ||||||||



Genmab 2025 Annual Report | 26 | ||||||


Genmab 2025 Annual Report | 27 | ||||||



Genmab 2025 Annual Report | 28 | ||||||


Genmab 2025 Annual Report | 29 | ||||||


Genmab 2025 Annual Report | 30 | ||||||
•Broad preclinical pipeline that includes both partnered products and in-house programs based on our proprietary technologies and/ or antibodies •Multiple new IND applications expected to be submitted over the coming years •Genmab has entered multiple strategic collaborations to support the expansion of our innovative pipeline, including our acquisition of ProfoundBio in 2024 and Merus in 2025 Our preclinical pipeline includes immune effector function enhanced antibodies developed with our HexaBody technology platform, bispecific antibodies created with our DuoBody technology platform and ADCs created with our ADC technology platforms. We are also collaborating with our partners to generate additional new antibody-based product concepts. A number of the preclinical programs are conducted in cooperation with our collaboration partners. Fourth Quarter Updates •November: IND submitted for GEN1079 •November: IND submitted for GEN1106 | ||

Genmab 2025 Annual Report | 31 | ||||||
In addition to Genmab’s own pipeline of investigational medicines, our innovations and proprietary technology platforms are applied in the pipelines of global pharmaceutical and biotechnology companies. These companies are running clinical development programs with antibodies created by Genmab or created using Genmab’s proprietary DuoBody bispecific antibody technology platform. The information in this section includes those therapies that have been approved by regulatory agencies in certain territories. Under the agreements for these medicines Genmab is entitled to certain potential milestones and royalties. | ||

Genmab 2025 Annual Report | 32 | ||||||


Genmab 2025 Annual Report | 33 | ||||||


Genmab 2025 Annual Report | 34 | ||||||


Genmab 2025 Annual Report | 35 | ||||||




Genmab 2025 Annual Report | 36 | ||||||
Platform | Principle | Applications | ||
DuoBody |  | Bispecific antibodies | Dual-targeting: •Recruitment (e.g., T cells) •Tumor heterogeneity | |
ADC Technology |  | Proprietary linker-drug platforms | •ADCs with more “antibody-like” PK •Pursue targets with clear opportunities for best- and/or first-in-class ADCs | |
HexaBody |  | Target-mediated enhanced hexamerization | Enhanced potency: •CDC •Target clustering, outside-in signaling, apoptosis | |
DuoHexaBody |  | Bispecific antibodies with target-mediated enhanced hexamerization | Dual-targeting + enhanced potency: •CDC •Target clustering, outside-in signaling, apoptosis | |
HexElect |  | Two co-dependent antibodies with target-mediated enhanced hexamerization | Dual-targeting + enhanced potency and selectivity: •Co-dependent unlocking of potency •New target space, previously inaccessible |

Genmab 2025 Annual Report | 37 | ||||||
 | Genmab 2025 Annual Report | 38 | |||||
Actual Result | Adjusted Result1 | Latest Guidance | |
Revenue | 3,720 | 3,720 | 3,500 - 3,700 |
Royalties | 3,102 | 3,102 | 2,945 - 3,090 |
Net product sales/Collaboration revenue | 468 | 468 | 425 - 465 |
Milestones/Reimbursement revenue | 150 | 150 | 130 - 145 |
Gross Profit | 3,482 | 3,482 | 3,280 - 3,460 |
Operating Expenses | (2,417) | (2,219) | (2,055) - (2,225) |
Operating Profit | 1,065 | 1,263 | 1,055 - 1,405 |
2025 | 2024 | 2023 | ||||
Royalties | 3,102 | 83% | 2,517 | 80% | 1,989 | 83% |
Net Product Sales | 398 | 11% | 253 | 8% | 61 | 3% |
Reimbursement revenue | 53 | 1% | 144 | 5% | 124 | 5% |
Milestone revenue | 97 | 3% | 145 | 5% | 171 | 7% |
Collaboration revenue | 70 | 2% | 62 | 2% | 45 | 2% |
Total revenue | 3,720 | 100% | 3,121 | 100% | 2,390 | 100% |
2025 | 2024 | 2023 | |
DARZALEX | 2,443 | 2,019 | 1,635 |
Kesimpta | 443 | 323 | 217 |
TEPEZZA | 105 | 106 | 102 |
Other | 111 | 69 | 35 |
Total royalties | 3,102 | 2,517 | 1,989 |
 | Genmab 2025 Annual Report | 39 | |||||
 | Genmab 2025 Annual Report | 40 | |||||
 | Genmab 2025 Annual Report | 41 | |||||
Percentage Change | Percentage Change | ||||
2025 | 2024 | 2023 | 2025/2024 | 2024/2023 | |
Research¹ | 267 | 310 | 219 | (14)% | 42% |
Development and contract manufacturing² | 598 | 516 | 337 | 16% | 53% |
Clinical³ | 607 | 478 | 476 | 27% | —% |
Other4 | 134 | 110 | 75 | 22% | 47% |
Total research and development expenses | 1,606 | 1,414 | 1,107 | 14% | 28% |
Percentage Change | Percentage Change | ||||
2025 | 2024 | 2023 | 2025/2024 | 2024/2023 | |
Epcoritamab | 303 | 414 | 192 | (27)% | 116% |
Rina-S | 230 | 46 | — | 400% | N/A |
Tisotumab vedotin | 23 | 38 | 41 | (39)% | (7)% |
Acasunlimab | 159 | 102 | 80 | 56% | 28% |
DuoBody-CD40x4-1BB | 50 | 75 | 59 | (33)% | 27% |
Other clinical stage programs | 23 | 73 | 109 | (68)% | (33)% |
Total third-party costs for clinical stage programs | 788 | 748 | 481 | 5% | 56% |
Preclinical projects | 245 | 216 | 164 | 13% | 32% |
Personnel, unallocated costs and overhead | 573 | 450 | 462 | 27% | (3)% |
Total research and development expenses | 1,606 | 1,414 | 1,107 | 14% | 28% |
 | Genmab 2025 Annual Report | 42 | |||||
2025 | 2024 | 2023 | |
Financial income: | |||
Interest and other financial income | 138 | 144 | 142 |
Gain on marketable securities | 112 | 237 | 157 |
Gain on other investments, net | — | 6 | — |
Foreign exchange rate gain | 158 | 258 | — |
Total financial income | 408 | 645 | 299 |
Financial expenses: | |||
Other interest expense | (34) | (18) | (10) |
Interest expense on borrowings | (27) | — | — |
Loss on marketable securities | (46) | (107) | (174) |
Loss on other investments, net | (1) | — | (4) |
Foreign exchange rate loss | (161) | (166) | (66) |
Total financial expenses | (269) | (291) | (254) |
Net financial items | 139 | 354 | 45 |
 | Genmab 2025 Annual Report | 43 | |||||
December 31 | ||
2025 | 2024 | |
Marketable securities | — | 1,574 |
Cash and cash equivalents | 1,715 | 1,380 |
Shareholders' equity | 5,847 | 5,137 |
Non-current borrowings | 5,001 | — |
Current borrowings | 273 | — |
 | Genmab 2025 Annual Report | 44 | |||||
 | Genmab 2025 Annual Report | 45 | |||||
Cash Flow ($ million) | 2025 | 2024 | 2023 |
Cash provided by operating activities | 1,186 | 1,126 | 1,071 |
Cash (used in) investing activities | (5,643) | (1,447) | (185) |
Cash provided by (used in) financing activities | 4,789 | (566) | (89) |
Changes in cash and cash equivalents | 332 | (887) | 797 |
Exchange rate adjustments | 3 | 63 | (12) |
 | Genmab 2025 Annual Report | 46 | |||||

 | Genmab 2025 Annual Report | 47 | |||||

 | Genmab 2025 Annual Report | 48 | |||||

 | Genmab 2025 Annual Report | 49 | |||||
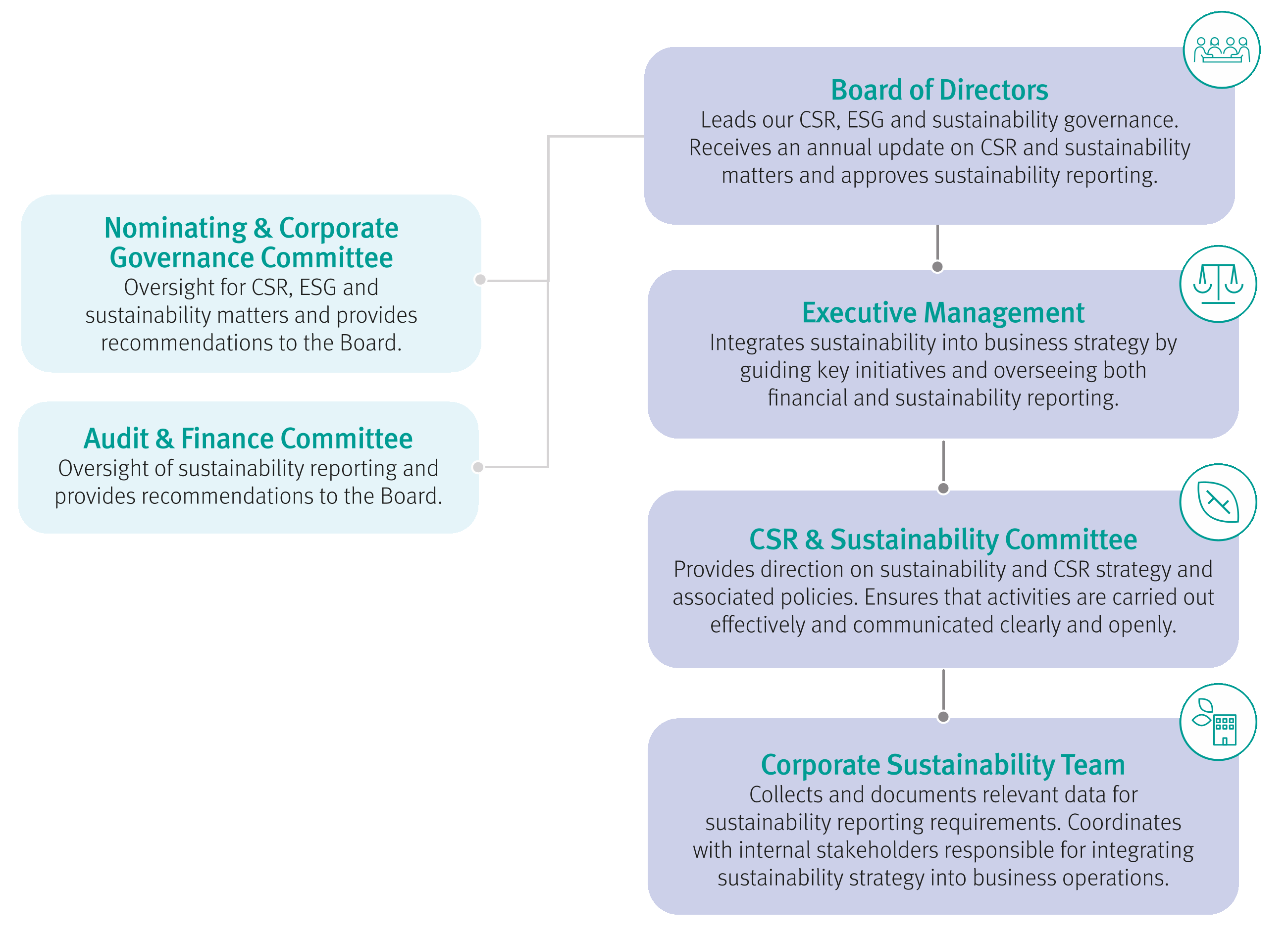
Effective ERM starts with strong governance | ||||
Board and Audit and Finance Committee: | The Board retains overall ERM/Risk oversight. The Audit and Finance Committee supports the Board by monitoring ERM activities and providing assurance that Management appropriately manages the risks of the business. | |||
Executive Management: | Maintains ultimate ownership of and accountability for management of top risks, enabling proper linkage of risk management to strategic initiatives and business decisions. | |||
GCRC: | Validates risk identification, prioritization, strategic and tactical ownership of risk mitigation plans and reporting. | |||
ERM Framework: | Routinely gathers risks, evaluates with risk sponsors, prioritizes, and reports to the GCRC, Executive Management and Board, driving risk discussions, and supporting risk sponsors and management in facilitating ERM processes, risk-intelligent decision-making, and key risk capabilities. | |||
Risk Sponsors and Business Champions: | Manage risks in the normal course of business, executing risk plans/mitigation activities, and monitoring and reporting key risk information. | |||

 | Genmab 2025 Annual Report | 50 | |||||
Risk related to | Risk areas | Mitigation | Risk trend | |
Business and Products | The identification and development of successful products is expensive and includes time-consuming clinical trials with uncertain outcomes and the risk of failure to obtain regulatory approval in one or more jurisdictions. | Genmab has a disciplined approach to investment, focusing on areas with the potential to maximize success, including new technologies and formats, scaling up to expand from early- to late-stage development and commercialization. Genmab has established various committees to ensure optimal selection of disease targets and formats of our antibody candidates, and to monitor progress of preclinical and clinical development. We strive to have a well-balanced product pipeline, continuing to search for and identify new product candidates, and closely monitoring the market landscape. |  | |
Genmab is dependent on the identification and development of new proprietary technologies and access to new third-party technologies. This exposes us to safety issues as well as other failures and setbacks related to use of such new or existing technologies. | Genmab strives to identify and develop new antibody-based products that harness new antibody technologies, such as the DuoBody, HexaBody, DuoHexaBody and HexElect technology platforms, ADC technology, and gain access to competitive and complementary new third-party technologies. We closely monitor our preclinical programs and clinical trials to mitigate any unforeseen safety issues or other failures, or setbacks associated with the use of these technology platforms. |  | ||
Genmab faces ongoing uncertainty about the successful commercialization of product candidates. This is a result of factors including immense competition on the basis of cost and efficacy as well as rapid technological change, which may result in others discovering, developing or commercializing competing products before and/or more successfully than us. | From early in the research phase and throughout development, commercial potential and product commercialization, associated risks are assessed to ensure that final products have the potential to be commercially viable. Genmab attempts to control commercial risks in part by regularly monitoring and evaluating current market conditions, competing products and new technologies, to potentially gain access to new technologies and products that may supplement our pipeline. Genmab also strives to ensure market exclusivity for its own technologies and products by seeking patent protection. Genmab engages with patients and caregivers to gather insights and improve patient outcomes. |  | ||
Genmab’s near- and mid-term prospects are substantially dependent on continued clinical and commercial success of DARZALEX. The impact of DARZALEX patent expirations will have an adverse impact on Genmab’s future royalty revenue. DARZALEX is subject to intense competition in the multiple myeloma therapy market. | Genmab focuses on its three-pronged strategy of focusing on our core competence, turning science into medicine and building a profitable and successful biotech to develop a broad pipeline of unique best-in-class or first-in-class antibody products with significant commercial potential. In addition, Genmab maintains a strong cash position, disciplined financial management, and a flexible and capital efficient business model to mitigate potential setbacks related to DARZALEX. To address the impact of DARZALEX Genmab patents expiration, Genmab intends to mitigate this risk through its strong foundation in science and investments in its late-stage assets EPKINLY, Rina-S and petosemtamab. Genmab manages and maintains efficient operations through focused prioritization and increased productivity. Beyond DARZALEX there are six additional medicines in our royalty portfolio that that drive revenue for the Company: Kesimpta, TEPEZZA, RYBREVANT, TECVAYLI, TALVEY and BIZENGRI. |  | ||
Genmab has exposure to product liability claims related to the use or misuse of our products and technologies. | Product liability claims and/or litigation could materially affect our business and financial position, and Genmab therefore strives to maintain internal processes for the review, approval, and compliant use of promotion materials and also maintains appropriate product liability insurance for our clinical trials and our approved products and other coverage required under applicable laws. |  | ||
Our core research and manufacturing activities are carried out at a limited number of locations. Any event resulting in Genmab’s or our vendors’/suppliers’ inability to operate these facilities could materially disrupt our business. | Genmab employs oversight and quality risk management principles. In addition, Genmab follows current Good Laboratory Practices (cGLP) and current Good Manufacturing Practices (cGMP) and requires that our vendors operate with the same standards. Genmab’s quality assurance (QA) department ensures that high-quality standards are set and monitors adherence to these practices. |  |

 | Genmab 2025 Annual Report | 51 | |||||
Risk related to | Risk areas | Mitigation | Risk trend | |
If we are unable to effectively manage Genmab’s fast-paced growth, or maintain our commercialization and other capabilities at adequate levels, and control operating costs within the scope of our overall business as well as properly integrate acquisitions, financial condition and net profits may be adversely affected. Any business disruption or failure to properly manage growth, maintain capabilities and transformation in a manner that reflects and supports our organizational strategies and priorities, while assuring ethical business practices, prudent risk management, and commercial compliance, could have a material adverse effect on our business, financial condition, results of operations and cash flows. | We have experienced rapid growth over the last several years. We anticipate additional growth as our pipeline advances and we continue product commercialization activities. Such growth, including maintaining and enabling R&D, commercialization, and support functions, has placed significant demands on our management and infrastructure, including new operational and financial systems, as well as extending manufacturing and commercial outsource arrangements. Our success will depend in part upon our ability to manage and maintain operations and integrate acquisitions effectively through leadership, focused prioritization, increased productivity and talent management to maintain our values-based, collaborative culture. As we continue to grow and evolve, we must continuously improve our operational, commercial, compliance, financial and management practices, and controls. |  | ||
Genmab is subject to government regulations on pricing/public reimbursement as well as other healthcare payer cost-containment initiatives; increased pressures by governmental and third-party payers to reduce healthcare costs. | Genmab strives to develop differentiated antibody medicines that bring meaningful impact to patients and health systems and are well-positioned to secure reasonable price reimbursement by government healthcare programs and private health insurers. The impact our science has on patients today and in the future, particularly those with few treatment options, drives the value of our medicines. Genmab’s U.S. Government Affairs & Policy department interacts with U.S. federal and state policymakers to advance policies aimed at improving patients’ lives through access to quality healthcare and innovative science. Genmab’s U.S. Market Access department educates payers on the value of our products and works across the healthcare system to help ensure all appropriate patients gain access to our innovative medicines. |  | ||
Strategic Collaborations | Genmab is dependent on existing partnerships with major pharmaceutical or biotech companies to support our business and develop and extend the commercialization of our products. | Our business may suffer if our collaboration partners do not devote sufficient resources to our programs and products, do not successfully maintain, defend and enforce their intellectual property rights or do not otherwise have the ability to successfully develop or commercialize our products, independently or in collaboration with others. Our business may also suffer if we are not able to continue our current collaborations or establish new collaborations. Genmab strives to be an attractive and respected collaboration partner, and to pursue a close and open dialogue with our collaboration partners to share ideas and align on best practices and decisions within clinical development and commercial operations to increase the likelihood that we reach our goals. |  | |
Genmab relies on a limited number of manufacturing organizations (CMO) and individual sites at those CMOs to produce and supply our product candidates. Genmab is also dependent on clinical research organizations to conduct key aspects of our clinical trials, and on collaboration partners to conduct some of our clinical trials. CMOs may be subject to or affected by various U.S. legislation, executive orders, regulations, or investigations. | Genmab oversees outsourcing and partnership relationships to ensure consistency with strategic objectives and service provider compliance with regulatory requirements, resources, and performance. This includes assessment of contingency plans, availability of alternative service providers and costs and resources required to switch service providers. We continually evaluate financial solvency and require our suppliers to abide by a code of conduct consistent with Genmab’s Code of Conduct. |  |

 | Genmab 2025 Annual Report | 52 | |||||
Risk related to | Risk areas | Mitigation | Risk trend | |
Regulation, Legislation, and Compliance | Genmab is subject to extensive legislative, regulatory and other requirements during preclinical and clinical development, commercialization, and post-marketing approval, including healthcare, marketing/labeling/ promotion, fraud and abuse, competition/antitrust laws, and regulations, as well as transparency, privacy and data protection and other requirements. Genmab is subject to strict disclosure obligations under applicable laws and regulations globally, including the EU Market Abuse Regulation and the U.S. Inflation Reduction Act (IRA). Being listed on the Nasdaq Global Select Market, we are subject to additional U.S. regulatory requirements, including U.S. securities laws and the U.S. Foreign Corrupt Practices Act, and may become more exposed to U.S. class actions. | To ensure compliance with applicable healthcare laws and regulations, Genmab has established a compliance program, including a Code of Conduct that is regularly evaluated and sets high ethical standards on which all colleagues receive regular training. Genmab also maintains a Speak Up Policy and Hotline for reporting and response to potential misconduct. Our head of Global Compliance reports directly to the Chief Legal Officer. Genmab is committed to transparency of clinical trial research and has published our Clinical Trial Transparency Declaration. Genmab is also committed to ensuring equal access to Genmab clinical trials and that patients participating in our trials are representative of those living with the disease being studied. Genmab respects the privacy, protection, and appropriate use of data by ensuring compliance with all applicable privacy and data protection laws, regulations, and other standards. In support of this commitment, Genmab established its Global Data Privacy Office supported by a cross-functional team of privacy subject matter experts, including a Data Protection Officer, who collaborate in the development and maintenance of a forward-looking Global Data Privacy Program that seeks to address shifts in both the internal and external environments, along with emerging challenges in the privacy and data protection regulatory landscape. The Program, through its policies, procedures, and centralized guidance for processing personal data, seeks to drive organizational accountability and empower Genmab colleagues, and our third party partners, to handle personal data consistent with our values of ethical behavior, integrity, fairness, inclusion, and transparency. To further support compliance with regulatory, legal, and other requirements applicable to our business and operations, including cGLP, current Good Clinical Practices (cGCP) and cGMP, Genmab’s QA department is staying abreast of and adhering to regulatory and legislative changes relevant to quality standards. Genmab has also established relevant procedures and guidelines to ensure transparency with respect to providing timely, adequate, and correct information to the market and otherwise complying with applicable securities laws and other legal and regulatory requirements. Genmab has an Internal Audit function that reports to the Audit and Finance Committee of the Board and administratively reports to the CFO. |  | |
Legislation, regulations, industry codes and practices, and their application may change from time to time. | To prevent unwarranted consequences of new and amended legislation, regulations, etc., Genmab strives to stay current with respect to all applicable legislation, regulations, industry codes and practices by means of its internal compliance function and related governance bodies as well as internal and external legal counsel. Also, internal procedures for review and refinement of contracts are ongoing to ensure contractual consistency and compliance with applicable legislation, regulation, and other standards. |  | ||
Intellectual Property | Genmab is dependent on protecting our own intellectual property rights to regain our investments and protect our competitive positions. We may become involved in lawsuits to protect or enforce our patents or other intellectual property which could result in costly litigation and unfavorable outcomes. Claims may be asserted against us that we infringe the intellectual property of third parties, which could result in costly litigation and unfavorable outcomes. | Genmab files and prosecutes patent applications to optimally protect its products and technologies. To protect trade secrets and technologies, Genmab maintains strict confidentiality standards and agreements for employees and collaborating parties. Genmab actively monitors third-party patent positions within our relevant fields to avoid violating any third-party patent rights. |  |

 | Genmab 2025 Annual Report | 53 | |||||
Risk related to | Risk areas | Mitigation | Risk trend | |
Finances | Genmab may have the inability to satisfy debt obligations. | Genmab's borrowings include two senior secured term loans, Term Loan A and Term Loan B, with nominal amounts of $1 billion and $2 billion, senior secured notes of $1.5 billion, senior unsecured notes of $1.0 billion as well as access to a revolving loan facility up to $500 million with a syndicate of lenders, which can be drawn down upon as another source of additional funding. The credit agreement requires Genmab to comply with certain financial maintenance and other covenants. A breach of the covenants under the credit agreement and the indentures governing the outstanding notes could result in an event of default which could result in Genmab having to repay the borrowings before their due dates. Because Genmab’s future commercial potential and operating profits are hard to predict, Genmab’s policy is to maintain a strong capital base so as to maintain investor, creditor and market confidence, and a continuous advancement of Genmab’s product pipeline and business in general. |  | |
Genmab is exposed to different kinds of financial risks, including currency exposure and changes in interest rates as well as changes in Danish, U.S. or foreign tax laws or related compliance requirements. | Genmab has established financial risk management guidelines to identify and analyze relevant risks, to set appropriate risk limits and controls, and to monitor the risks and adherence to limits. Please refer to Note 4.2 of the financial statements for additional information regarding financial risks. |  | ||
Management and Workforce | Genmab may have an inability to attract and retain suitably qualified team members as it continues to evolve. | To attract and retain our highly skilled team, including the members of Genmab’s Executive Management, Genmab offers competitive remuneration packages, including share-based remuneration. Genmab strives to create a positive, safe, and energizing working environment. Genmab has strong core values that nourish high-integrity and ethical behavior, respectful and candid tone and a culture which prizes diversity, as well as trust and teamwork. Genmab has implemented strategies such as diversifying recruitment efforts, cross-training employees, fostering a culture of knowledge sharing, investing in talent development programs, and promoting a supportive work environment that values employee well-being and career growth. Please refer to Note 4.6 of the financial statements for additional information regarding share-based compensation. |  | |
Cybersecurity | Genmab may be subject to malicious cyber attacks, and with the increased use of artificial intelligence within the biopharmaceutical industry, can lead to the theft or leakage of intellectual property, sensitive business data, or personal employee or patient data, with the result of significant business disruptions, negative impacts to patient or employee privacy, monetary loss or fines from authorities, or reputational damage. | Genmab has implemented security controls and processes to enhance the identification of potential data/systems security issues and mitigate the risk of security breaches. Genmab makes use of the National Institute of Standards and Technology (NIST) Cybersecurity Framework and other security standards to define and implement such security controls. Due to the continually changing threat environment, regular assessments are executed to ensure that implemented security controls and processes follow the threat profile of the Company and effectively support Genmab’s ambitious business strategy. The risk of security breaches is regarded as enterprise risk and the Company’s threat profile, the security program and security incidents are presented and discussed in meetings of the Global Compliance and Risk Committee and the Audit and Finance Committee of the Board. Genmab’s Cybersecurity Program, in conjunction with Genmab’s Global Data Privacy Program, collaborates to manage and mitigate any cybersecurity and data privacy threats to the personal data processed in our systems and by our third party partners. |  | |
Environment | Genmab could face transitional risks by its inability to manage the carbon footprint and energy mix from our business operations and physical risks from climate-related events that may impact our business operations or that of our third-party partners or suppliers. | Genmab has oversight and manages its carbon footprint Scope 1 and 2 emissions from its business operations. Genmab is committed to tracking the Scope 3 emissions carbon footprint by partnering with suppliers. Genmab makes use of scenario analysis to evaluate risks and opportunities due to the rapid pace of world climate change. Genmab’s work with climate strategy, carbon reduction targets, climate-related financial risk, relevant prevention, and mitigation measures are presented to and reviewed by the Board biannually. Refer to the sustainability statements for details of Genmab’s targets in the future to mitigate risks. |  |
 | Genmab 2025 Annual Report | 54 | |||||
 | Genmab 2025 Annual Report | 55 | |||||


 | Genmab 2025 Annual Report | 56 | |||||
As of December 31, 2025, there are nine members of Executive Management. | ||||||||
Jan G. J. van de Winkel, Ph.D. Dutch, 64, Male President & Chief Executive Officer Special Competencies Extensive antibody creation and development expertise, broad knowledge of the biotechnology industry and executive management skills. ESG Competencies: Social · Governance | Anthony Pagano American, 48, Male Executive Vice President & Chief Financial Officer Special Competencies Significant knowledge and experience in the life sciences industry particularly as it relates to corporate finance, corporate development, strategic planning, general management, treasury, accounting, and corporate governance. ESG Competencies: Social · Governance | Judith Klimovsky, M.D. Argentinian (U.S. Citizen), 69, Female Executive Vice President & Chief Development Officer Special Competencies Extensive expertise in oncology drug development from early clinical stages through to marketing approval, experience in clinical practice and leading large teams in pharmaceutical organizations. ESG Competencies: Social · Governance Current Board Positions: Member, Bio-Techne | Tahamtan Ahmadi, M.D., Ph.D. Iranian-German (U.S. Citizen), 53, Male Executive Vice President & Chief Medical Officer, Head of Experimental Medicines Special Competencies Significant expertise in global regulatory and clinical drug development across entire spectrum from pre-IND to life cycle management; drug discovery and translational research. ESG Competencies: Social · Governance | |||||
Christopher Cozic American, 48, Male Executive Vice President, Chief People Officer Special Competencies Expertise in strategic leadership, organization design, human resource management, policy development, employee relations, organizational development, and a heavy concentration in all aspects of corporate growth and expansion. ESG Competencies: Social · Governance Current Board Positions: Member, BioNJ | Martine J. van Vugt, Ph.D. Dutch, 55, Female Executive Vice President, Chief Strategy Officer Special Competencies Extensive knowledge of and experience in Corporate Strategy, Corporate and Business Development, as well as Portfolio, Project, and Alliance Management. ESG Competencies: Social · Governance | Rayne Waller American, 58, Male Executive Vice President & Chief Technical Operations Officer Special Competencies Expertise in all elements of technical operations from early-to-mid-stage product development through global manufacturing of both clinical and commercial products. ESG Competencies: Social · Governance | Brad Bailey American, 58, Male Executive Vice President & Chief Commercial Officer Special Competencies Extensive experience in strategic and operational commercial leadership roles across specialty biopharma, oncology, immunology, and other serious diseases. ESG Competencies: Social · Governance | Greg Mueller Canadian, British, 54, Male Executive Vice President, General Counsel & Chief Legal Officer Special Competencies Extensive international experience in Legal, IP, Compliance, Risk and Business Development matters, with deep experience in the pharmaceutical industry. ESG Competencies: Social · Governance |

 | Genmab 2025 Annual Report | 57 | |||||
Refer to our website for the Board of Directors diversity and skills matrix. | ||||||
Deirdre P. Connelly Female, Hispanic/American, 65 Board Chair (Independent, elected by the General Meeting); Member of the Audit and Finance Committee, the Compensation Committee and the Nominating & Corporate Governance Committee First elected 2017, current term expires 2026 Special Competencies and Qualifications Deirdre P. Connelly has more than 30 years’ experience as a corporate leader and board member in publicly traded companies with global operations. She has comprehensive knowledge and experience with business conduct, business turnaround and product development and has successfully directed the launch of more than 20 new pharmaceutical drugs. As a former HR executive, Deirdre P. Connelly also has valuable insight in corporate culture transformation, talent development and managing large organizations. She furthermore has significant experience with the development of governance and other sustainability related responsibilities from various leadership roles and as a board member. Deirdre P. Connelly is former President of U.S. Operations of Eli Lilly and Company and former President, North America Pharmaceuticals for GlaxoSmithKline. ESG Competencies: Social · Governance Current Board Positions Member: Lincoln Financial Corporation1, Macy’s Inc.2 Sarepta Therapeutics, Inc.3 1.Chair of Compensation Committee, Member of Audit Committee, Corporate Governance Committee and Executive Committee 2.Chair of Nominating and Corporate Governance Committee, Member of Compensation and Management Development Committee 3.Chair of Compensation Committee, Member of Nominating and Corporate Governance Committee | Pernille Erenbjerg Female, Danish, 58 Deputy Board Chair (Independent, elected by the General Meeting); Chair of the Audit and Finance Committee, Member of the Nominating and Corporate Governance Committee First elected 2015, current term expires 2026 Special Competencies and Qualifications Pernille Erenbjerg has broad executive management and business experience from the telecoms, media, and tech industries. She has extensive expertise with business conduct and in operation and strategic transformation of large and complex companies, including digital transformations and digitally based innovation, and has been responsible for major transformation processes in complex organizations including M&A. Pernille Erenbjerg furthermore has significant IT and cybersecurity expertise and sustainability related experience from various executive and non-executive positions. She has a Certified Public Accountant background (no longer practicing) and has a comprehensive all-around background within finance, including extensive exposure to public and private equity and debt investors. Pernille Erenbjerg is former CEO and President of TDC Group A/S. Pernille Erenbjerg is an audit committee financial expert based on her professional experience, including her background within accounting, her service in senior finance leadership at TDC Group A/S and as an audit committee chair or member at other public companies. ESG Competencies: Environmental · Social · Governance Current Board Positions Chair: KK Wind Solutions Member: RTL Group1, GlobalConnect, Nokia2 1.Chair of Audit Committee 2.Member of the Audit Committee and Corporate Governance and Nomination Committee | Anders Gersel Pedersen, M.D., Ph.D. Male, Danish, 74 Board Member (Non-independent, elected by the General Meeting); Chair of the Nominating and Corporate Governance Committee, Member of the Scientific Committee and Compensation Committee First elected 2003, current term expires 2026 Special Competencies and Qualifications Anders Gersel Pedersen has more than 30 years’ board and management experience in publicly traded, international pharmaceutical and biotech companies. He has significant knowledge and expertise in discovery and development of the product pipeline from preclinical activities to post-launch marketing studies as well as solid business experience. Anders Gersel Pedersen furthermore has extensive experience with the global pharmaceutical market and has built comprehensive knowledge and insight in governance, including business conduct, and the development of ESG and other sustainability related responsibilities from various leadership roles and as a board member. Anders Gersel Pedersen is former Executive Vice President of Research & Development of H. Lundbeck. ESG Competencies: Environmental · Social · Governance Current Board Positions Chair: Aelis Farma S.A.S. Member: Bond 2 Development GP Limited |

 | Genmab 2025 Annual Report | 58 | |||||
Refer to our website for the Board of Directors diversity and skills matrix. | ||||||
Paolo Paoletti, M.D. Male, Italian/American, 75 Board Member (Independent, elected by the General Meeting); Chair of the Scientific Committee and Member of the Compensation Committee First elected 2015, current term expires 2026 Special Competencies and Qualifications Paolo Paoletti has extensive experience in research, development and commercialization in the pharmaceutical industry, where he has been responsible for the development of several medicines approved globally and the related global commercial strategies. As an executive, he has led cross-functional teams on the development and registration of medicines and has been responsible for all compliance aspects for the R&D organization. Paolo Paoletti has successfully conducted submissions and approvals of new cancer drugs and new indications in the U.S., in Europe and in Japan. He furthermore has significant experience with governance, including business conduct, from various leadership roles and as a board member. Paolo Paoletti is former Vice President of Oncology Clinical Development with Eli Lilly and Company, former President of GSK Oncology with GlaxoSmithKline, and former CEO of GAMMADELTA Therapeutics. ESG Competencies: Environmental · Social · Governance Current Position, including Managerial Positions Member of the Investment Committee for Apollo Therapeutics Limited Scientific Advisor for 3B Future Health Fund Scientific Advisor for Sun Pharmaceuticals Current Board Positions None | Rolf Hoffmann Male, German/Swiss, 66 Board Member (Independent, elected by the General Meeting); Member of the Audit and Finance Committee and the Scientific Committee First elected 2017, current term expires 2026 Special Competencies and Qualifications Rolf Hoffmann has more than 30 years’ experience in senior management and as a board member in the life science industry worldwide. He has significant expertise with business conduct and in creating and optimizing commercial opportunities in global markets and has managed companies across multiple continents with multibillion-dollar P&L and cross- functional accountability. Rolf Hoffmann furthermore has knowledge and experience with governance, compliance and ensuring organizational efficiency from various management positions as well as from being a board member. Rolf Hoffmann has held a variety of sales and marketing and executive management positions with Eli Lilly and Company, and is former Senior Vice President, International Commercial Operations and former Senior Vice President, U.S. Commercial Operations with Amgen. ESG Competencies: Environmental · Social · Governance Current Position, including Managerial Positions Adjunct Professor of Strategy and Entrepreneurship at University of North Carolina Business School Current Board Positions Member: Semdor Pharma, Sun Pharmaceutical Industries Ltd.1, Priavoid GmbH2 1.Member of Nomination and Remuneration Committee 2.Deputy Chairman | Elizabeth A. O’Farrell Female, American, 61 Board Member (Independent, elected by the General Meeting); Chair of the Compensation Committee, Member of the Audit and Finance Committee First elected 2022, current term expires 2026 Special Competencies and Qualifications Elizabeth O’Farrell has solid financial experience from her 25-year career in finance leadership roles and as a board member. During her career, she has led multiple strategy, planning and resource allocation processes in multiple roles and in cross-functional teams. Elizabeth O’Farrell has significant knowledge and expertise in business conduct and with driving paradigm-changing contributions within finance and the enterprise through collaboration and influence. In addition to experience at Price Waterhouse and Whipple & Company Corporation, Elizabeth O’Farrell held various executive management positions at Eli Lilly and Company, including as former Chief Procurement Officer. Elizabeth O’Farrell is an audit committee financial expert based on her professional experience, including her service in senior finance leadership positions at Eli Lilly and as an audit committee chair or member at other public companies. She has also completed the Nasdaq Center for Board Excellence Cyber Security Program. ESG Competencies: Social · Governance Current Board Positions Chair: PDL BioPharma, Geron Corporation1 Member: LENSAR1, Karius1, SpyGlass Pharma1 1.Chair of Audit Committee |

 | Genmab 2025 Annual Report | 59 | |||||
Refer to our website for the Board of Directors diversity and skills matrix. | ||||||
Michael Kavanagh Male, American, 52 Board Member (Non-independent, elected by the employees) First elected 2025, current term expires 2028 Special Competencies and Qualifications Michael Kavanagh is a seasoned professional with over 25 years of experience in the pharmaceutical industry and has a track record of successful product launches in oncology commercialization. His extensive expertise in building commercial teams and driving market expansion has been demonstrated in the successful product launches at Genmab. Previously, he held senior leadership roles at Bristol Myers Squibb, leading commercialization efforts for hematology and oncology brands in the US and globally. ESG Competencies: Social · Governance Current Position, including Managerial Positions Senior Director, Head of Strategic Engagement, Oncology Marketing at Genmab | Martin Schultz Male, Danish, 50 Board Member (Non-independent, elected by the employees) First elected 2022, current term expires 2028 Special Competencies and Qualifications Martin Schultz has broad experience within clinical project management with a substantial understanding and knowledge of research and development. He furthermore has specific expertise in project management, strategic sourcing, vendor collaboration, contract, and budget governance. ESG Competencies: Social · Governance Current Position, including Managerial Positions Senior Director, Head of Development Business Partnership & Strategy at Genmab | Mijke Zachariasse, Ph.D. Female, Dutch, 52 Board Member (Non-independent, elected by the employees) First elected 2019, current term expires 2028 Special Competencies and Qualifications Mijke Zachariasse has broad experience in people and business management and expertise in building partnerships across sectors, the research funding landscape, operational excellence and organizational strategy and change. ESG Competencies: Environmental · Social · Governance Current Position, including Managerial Positions Vice President, Head of Protein and Cell Supply at Genmab |
C — Chair M — Member O — Observer F — Financial Expert | Review of BoD (incl. Committee Composition) Current Committee Composition | ||||
Name | A&FC | Compensation Comm. | NCGC | Scientific Comm. | |
Deirdre P. Connelly (Chair) | M | M | M | ||
Pernille Erenbjerg (Deputy Chair) (F) | C | M | |||
Anders Gersel Pedersen | M | C | M | ||
Paolo Paoletti | M | C | |||
Rolf Hoffmann | M | M | |||
Elizabeth O’Farrell (F) | M | C | |||
Mijke Zachariasse | O | ||||
Martin Schultz | O | ||||
Michael Kavanagh | |||||
 | Genmab 2025 Annual Report | 60 | |||||

Share Data | Denmark | US |
Number of shares at December 31, 2025 | 64,238,408 | 6,437,745 (represented by 64,377,450 American Depository Shares (ADSs)) |
Listing | Nasdaq Copenhagen | Nasdaq Global Select Market, New York |
Ticker Symbol | GMAB | GMAB |
Index Membership | OMX Nordic Large Cap Index OMX Copenhagen Benchmark Index OMX Copenhagen 25 Index (OMXC25) | Nasdaq Biotech Index |

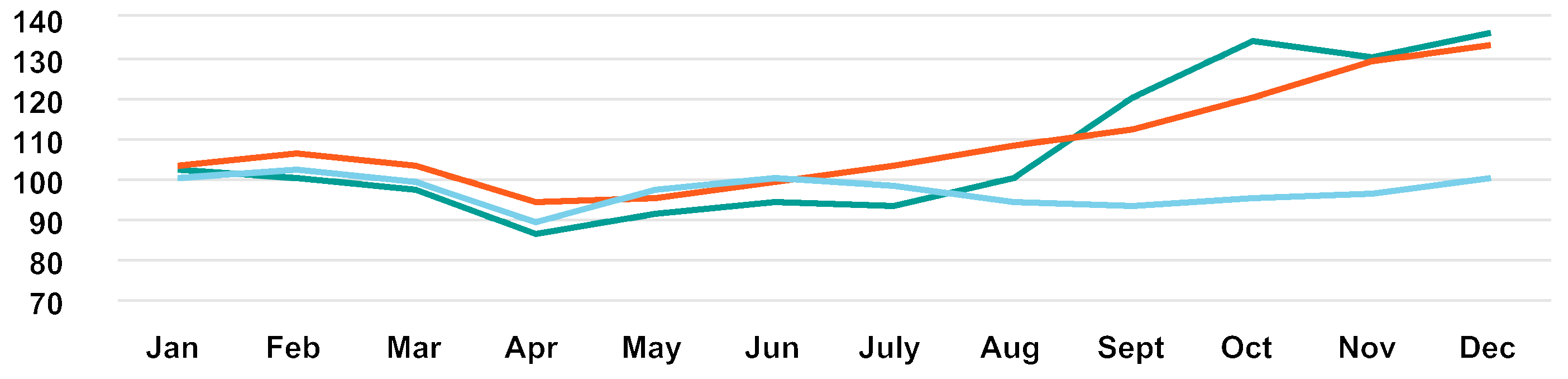
— Genmab | — OMXC25 | — Nasdaq Biotech |
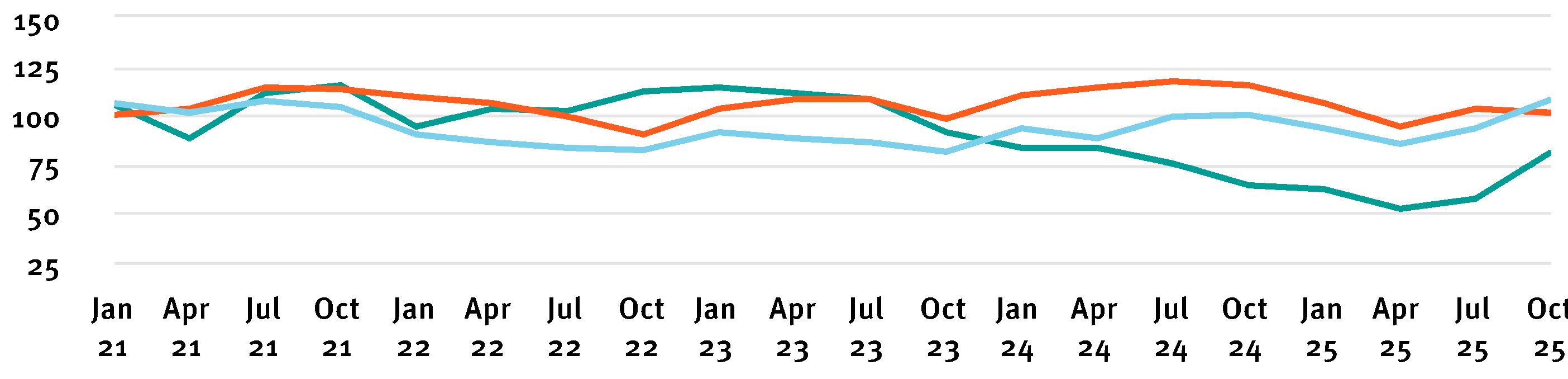
— Genmab | — OMXC25 | — Nasdaq Biotech |
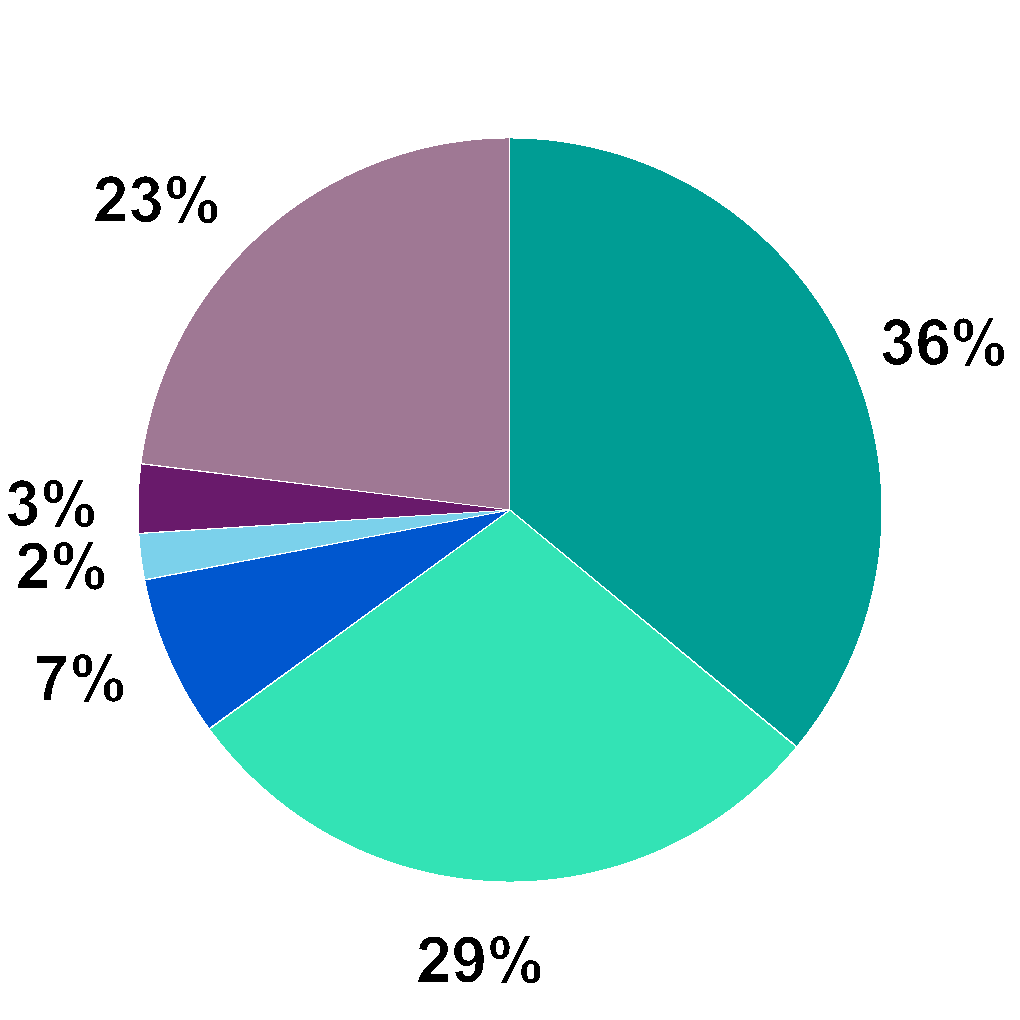
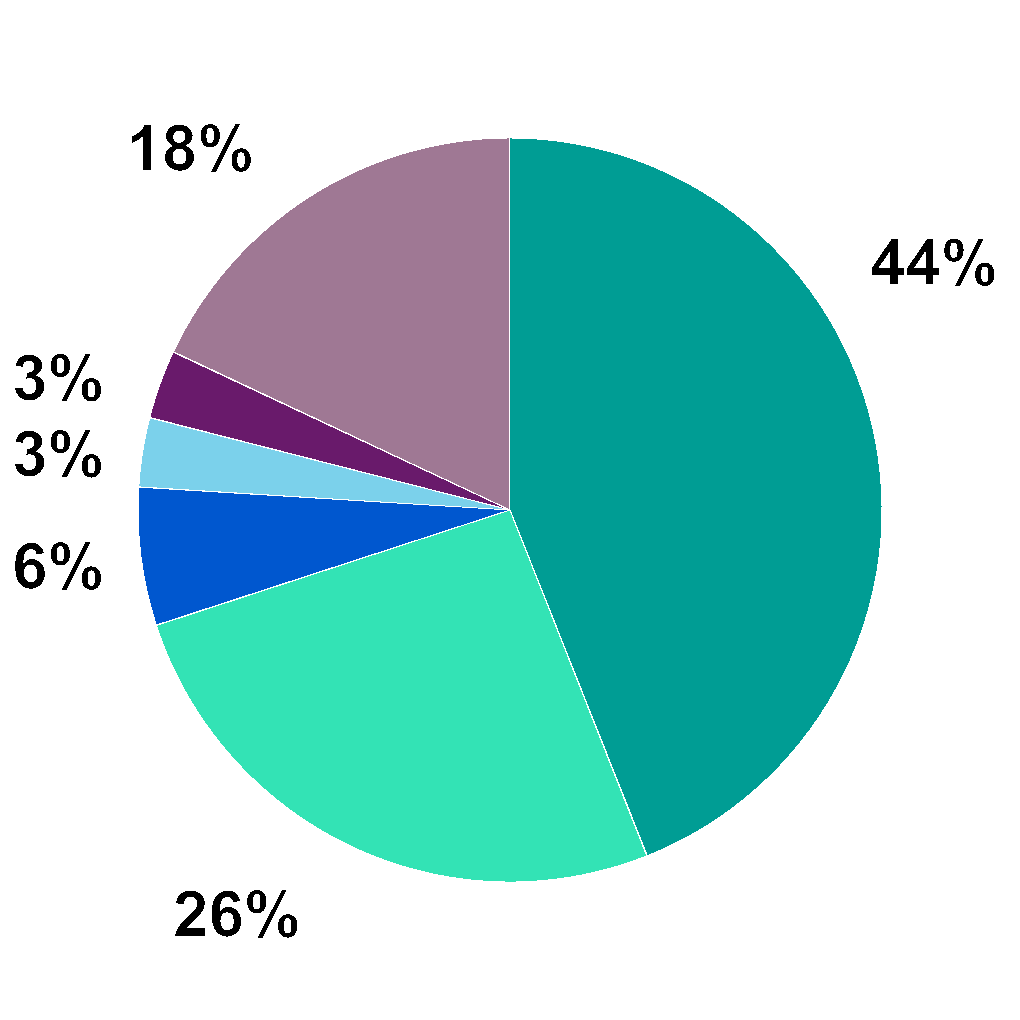
• | USA | • | Netherlands |
• | Denmark | • | Norway |
• | UK | • | Other3 |
 | Genmab 2025 Annual Report | 61 | |||||
Annual General Meeting 2026 | Thursday, March 19, 2026 |
Publication of the Interim Report for the first quarter 2026 | Thursday, May 7, 2026 |
Publication of the Interim Report for the first half 2026 | Thursday, August 6, 2026 |
Publication of the Interim Report for the first nine months 2026 | Thursday, November 5, 2026 |

 | Genmab 2025 Annual Report | 62 | |||||


 | Genmab 2025 Annual Report | 63 | |||||

Section | Disclosure Requirements Content | Disclosure Requirements # | Reference/Report | |
1.1 Basis for preparation | General basis for preparation of the sustainability statement | BP-1 | SUS | |
Disclosures in relation to specific circumstances | BP-2 | SUS | ||
1.2 Governance | The role of the administrative, management and supervisory bodies | GOV-1 | SUS, MR | |
Information provided to, and sustainability matters addressed by the undertaking’s administrative, management and supervisory bodies | GOV-2 | SUS, MR | ||
Sustainability-related performance in incentive schemes | GOV-3, E1, S1 | SUS | ||
Statement on due diligence | GOV-4 | SUS | ||
Risk management and internal controls over sustainability reporting | GOV-5 | SUS, MR | ||
1.3 Strategy | Strategy, business model and value chain | SBM-1 | SUS, MR, FS | |
Interests and views of stakeholders | SBM-2 | SUS | ||
Material impacts, risks and opportunities and how they interact with our strategy and business model | SBM-3 | SUS | ||
1.4 Impact, risk and opportunity management | Process to identify and assess material impacts, risks and opportunities | IRO-1 | SUS | |
Disclosure requirements in ESRS covered by the sustainability statement | IRO-2 | SUS |
 | Genmab 2025 Annual Report | 64 | |||||
 | Genmab 2025 Annual Report | 65 | |||||
Sustainability-related performance in incentive schemes | Unit | 2025 | 2024 |
Total remuneration to registered Executive Management | USDm | 15.4 | 11.4 |
Portion linked to climate-related performance goals | USDm | 0.5 | 0.3 |
% | 3% | 3% | |
Total variable remuneration to registered Executive Management | USDm | 12.9 | 9.1 |
Portion of variable remuneration linked to all sustainability-related performance goals | USDm | 1.1 | 0.9 |
% | 9% | 10% |
 | Genmab 2025 Annual Report | 66 | |||||
Core elements of Due Diligence | Sections in the Sustainability Statement | Does the disclosure relate to people and/or the environment? |
a) Embedding due diligence in governance, strategy, and business model | ESRS 2 GOV-1, GOV-2, GOV-3 | People and Environment |
ESRS 2 SBM-3: | ||
E1 | Environment | |
b) Engaging with affected stakeholders in all key steps of the due diligence | ESRS 2 GOV-1, GOV-2 | People and Environment |
ESRS 2 SBM-2 | People and Environment | |
ESRS 2 IRO-1 | People and Environment | |
ESRS 2 MDR-P: | ||
E1-2 | Environment | |
c) Identifying and assessing adverse impacts | ESRS 2 IRO-1: | |
E1 | Environment | |
d) Taking actions to address those adverse impact | ESRS 2 MDR-A: | |
E1-3 | Environment | |
e) Tracking the effectiveness of these efforts and communicating | ESRS 2 MDR-M: | |
E1-4 | Environment | |
ESRS 2 MDR-T: | ||
E1-4 | Environment |
 | Genmab 2025 Annual Report | 67 | |||||
Stakeholder Group | Description | Value Chain Location | How Engagement is Organized | Purpose of Engagement | Key Outcomes |
Academic, Scientific & Research Partners | Academic institutions, Contract Research Organizations (CROs) and research organizations collaborating on early-stage research, technology, and innovation. | Upstream, Own Operations | •Collaborative research programs and licensing agreements – Scientific conferences and workshops – Sponsorships and co-development meetings | •Drive innovation and access new technologies – Exchange knowledge and expertise | •Accelerated innovation – Publications and patents – Strengthened reputation and talent development |
Collaboration Partners | Companies partnering in co- development, licensing, or commercialization. | Upstream, Downstream | •Joint steering committees – Regular project reviews and team meetings | •Strategic alignment and innovation – Shared development responsibilities | •Successful product launches – Shared expertise and strengthened partnerships |
Contract Manufacturers, Suppliers & Quality Partners | Contract Manufacturing Organizations (CMOs), suppliers, and QA teams ensuring quality, reliability, and sustainability of materials and production. | Upstream, Own Operations | •Supplier qualification and audits – Regular communication and SOP adherence – Quality assurance reviews and training | •Ensure supply chain quality, safety, and compliance – Drive continuous improvement and sustainability | •Consistent product quality – Reduced compliance risks – Long-term, trusted supplier relationships |
Employees | Core internal stakeholders supporting research, development, and operations aligned with Genmab’s strategic goals and 2030 Vision. | Own Operations | •Engagement surveys and networks – Development dialogues and training – Works councils and employee representatives | •Foster inclusive, safe, and engaging workplace – Promote well-being and collaboration | •Action plans and improved engagement – Enhanced workplace culture and communication |
Healthcare Providers | Physicians, nurses, and medical institutions supporting clinical trials and patient care. | Upstream, Downstream | •Advisory boards – Clinical trials – Educational initiatives and feedback mechanisms | •Understand clinical needs – Enhance patient outcomes and research quality | •Improved clinical trial design – Increased therapy adoption and safety awareness |
Patients & Patient Organizations | End-users and advocacy groups providing insights on therapies and clinical trials. | Upstream, Downstream | •Patient Advisory Council – Focus groups and surveys – Support for patient organizations | •Embed patient perspectives in R&D – Improve education and awareness | •Enhanced trial design and patient experience – Safer, more effective therapies |
Payers | Insurance providers and health systems determining reimbursement and market access. | Downstream | •Advisory boards and meetings – Value assessment studies – Health economics collaborations | •Demonstrate therapeutic value and pricing rationale – Support equitable access | •Strengthened payer relationships – Improved access and affordability |
Regulatory Agencies | Authorities such as EMA, FDA, and MHLW, overseeing clinical trials and approvals. | Upstream, Downstream | •Submissions, reports, and regular meetings – Advisory consultations | •Ensure compliance and patient safety – Clarify approval pathways | •Regulatory approvals – Streamlined development and improved safety data |
Investors | Shareholders supporting Genmab’s financial growth and long-term strategy. | Upstream, Own Operations, Downstream | •Earnings calls, roadshows, and conferences – One-on-one investor meetings | •Build trust through transparency – Gather feedback on strategy and performance | •Increased investor confidence – Broader shareholder base |
Communities | Local and global communities where Genmab operates, benefiting from CSR and social initiatives. | Upstream, Own Operations, Downstream | •Community programs and partnerships – Employee volunteering | •Promote health awareness and social responsibility – Strengthen community trust | •Positive social impact – Enhanced employee morale and community relations |
 | Genmab 2025 Annual Report | 68 | |||||
Key: | U | Upstream | D | Downstream | MT | Medium-Term | ||
OO | Own operations | ST | Short-Term | LT | Long-Term |
Value Chain Location | Time Horizon | ||||||||
IRO Name | IRO Type | IRO Description | U | OO | D | ST | MT | LT | |
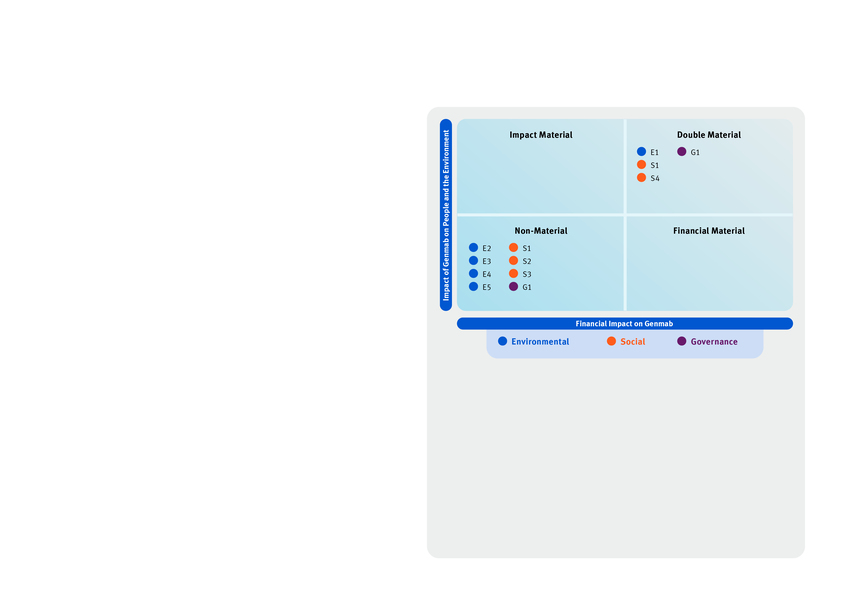
E1 - Climate Change | |||||||||
Climate Change - Adaptation, Mitigation and Energy | GHG emissions from own operations and value chain | Actual Negative Impact | Genmab’s business model centers on the research, development, and commercialization of innovative antibody therapies. These activities generate GHG emissions which have an actual negative impact on the environment. | • | • | • | • | • | |
Transitional and physical risks related to GHG emissions | Risk | Genmab faces potential transitional risks including loss of market access and higher costs from investments in green technologies, alongside reputational, regulatory, and financial pressures linked to the net-zero transition. Genmab also faces potential physical risks including disruption of supply chain and operations from extreme weather, heat waves, and flooding, though exposure is limited in our own operations due to Genmab’s asset-light model. | • | • | • | • | • | ||
Partner with value chain to reduce Scope 3 emissions | Opportunity | Genmab has an opportunity to partner with the value chain to reduce upstream emissions while driving efficiency and potential cost savings for both Genmab and its suppliers. This opportunity is linked to our Scope 3 supplier engagement target. | • | • | • | • | |||
S1 - Own Workforce | |||||||||
Working Conditions | |||||||||
Own Workforce - Working Conditions | Employee well-being and vitality | Actual Positive Impact | Genmab’s employees feel connected and motivated in a safe work environment enabling them to thrive and perform at their best. | • | • | • | • | ||
Attracting and retaining talent to enable continued innovation | Risk | As a science-driven innovation company, Genmab recognizes that our success depends on our ability to attract, develop, and retain exceptional talent. Our continued progress in research and development makes this especially critical. | • | • | • | • | |||
 | Genmab 2025 Annual Report | 69 | |||||
Key: | U | Upstream | D | Downstream | MT | Medium-Term | ||
OO | Own operations | ST | Short-Term | LT | Long-Term |
Value Chain Location | Time Horizon | ||||||||
IRO Name | IRO Type | IRO Description | U | OO | D | ST | MT | LT | |
Provide a voice to employees through our global engagement survey | Opportunity | Genmab has an opportunity through our annual Global Engagement Survey to assesses satisfaction, well-being, and workplace conditions, using results to drive improvement. Leaders are accountable for acting on feedback, ensuring employees feel heard, valued, and aligned with the Company’s goals—fostering a positive, supportive, and sustainable work environment. This opportunity is linked to our Global Engagement Survey target which is part of Executive Management performance criteria for incentive compensation. | • | • | • | • | |||
Safety in our facilities | Potential Negative Impact | Genmab recognizes there is a systemic potential negative impact around safety in our facilities due to potential work-related accidents, illnesses or fatalities that can arise in a laboratory setting. | • | • | • | • | |||
Equal Treatment and Opportunities for All | |||||||||
Own Workforce - Equal Treatment and Opportunities for All | Career development through training and skill building | Actual Positive Impact | Genmab’s focus on continuous learning fosters growth, collaboration, and morale while strengthening its ability to attract and retain top talent. | • | • | • | • | ||
Equal opportunity promoting innovation | Actual Positive Impact | Genmab’s team members encompass over 75 nationalities. We foster a global, inclusive culture, with access to equal opportunities, where a broad mix of perspectives across gender, age, and nationality drive innovation to meet the needs of patients, partners, and employees. | • | • | • | • | |||
S4 - Consumers and End-Users | |||||||||
Social inclusion of consumers and/or end-users | |||||||||
Consumers and End-Users - Social inclusion of consumers and/or end-users | Innovation for patients with unmet needs | Actual Positive Impact | From discovery through commercialization, Genmab’s antibody-based medicines have a meaningful positive impact on patients’ lives. As we expand our innovative capabilities to address cancer and other serious diseases, our continued investment in scientific excellence creates new opportunities to deliver breakthrough therapies that improve health outcomes and quality of life. | • | • | • | • | ||
Research and development risk | Risk | The identification and development of successful products is expensive and includes time-consuming clinical trials with uncertain outcomes and the risk of failure to obtain regulatory approval in one or more jurisdictions. | • | • | • | • | |||
Access and inclusion in clinical trials | Potential Negative Impact | Persistent inequities in cancer incidence and care continue to drive underrepresentation in clinical research. Expanding access for underrepresented groups ensures Genmab’s trials reflect real-world patients and generate more representative safety and efficacy data. | • | • | • | • | |||
Regulation, Legislation, and Compliance | Risk | Genmab is subject to extensive legislative, regulatory, and other requirements during preclinical and clinical development, commercialization, and post-marketing approval, including healthcare, marketing/labeling/promotion, fraud and abuse, competition/antitrust laws, and regulations, as well as transparency, privacy, and data protection and other requirements. | • | • | • | • | |||
Responsible and ethical marketing | Potential Negative Impact | Without responsible, ethical marketing, patients and healthcare professionals could receive incomplete or misleading information about Genmab’s therapies. This could undermine trust in our science, contributing to improper medicine use, and negatively affect patient well-being. It can also distort treatment decisions and harm the integrity of the broader healthcare ecosystem. | • | • | • | • | |||
Personal safety and information of consumers and/or end user | |||||||||
Consumers and End-Users - Personal safety and information of consumers and/or end user | Patient voice | Actual Positive Impact | Genmab incorporates patient and caregiver perspectives across the full product lifecycle, ensuring our innovations address the realities of serious illness. | • | • | • | • | ||
 | Genmab 2025 Annual Report | 70 | |||||
Key: | U | Upstream | D | Downstream | MT | Medium-Term | ||
OO | Own operations | ST | Short-Term | LT | Long-Term |
Value Chain Location | Time Horizon | ||||||||
IRO Name | IRO Type | IRO Description | U | OO | D | ST | MT | LT | |
Health and safety of patients | Potential Negative Impact | Any breakdown in Genmab’s safety and clinical oversight processes could expose trial participants to avoidable risks, including adverse events, inappropriate use of investigational medicines, or missed beneficial treatments. Patients depend on rigorous controls and accurate information to protect their health and ensure safe use of our therapies. | • | • | • | • | |||
Pharmacovigilance risks as a biotech company | Risk | Robust pharmacovigilance is essential for monitoring the safety and effectiveness of our therapies throughout their lifecycle. Any gaps or disruptions in these processes could delay the detection of adverse events, lead to regulatory non-compliance, and create reputational or financial consequences. | • | • | • | • | |||
Access to quality information | Potential Negative Impact | Limited transparency in clinical trials can restrict access to reliable information, compromising patient outcomes, research integrity, and trust. | • | • | • | • | |||
G1 - Business Conduct | |||||||||
Business Conduct - Corporate Culture Business Conduct - Corruption and bribery | Healthy and ethical corporate culture aligned with core values and purpose | Actual Positive Impact | Genmab has clear, core values, allowing a healthy and ethical culture to thrive and anti-corruption practices embedded in the ways of working. This is demonstrated by all employees’ attestation to our ethical standards and Code of Conduct. | • | • | • | • | ||
Organizational health risk | Risk | Misaligned or toxic culture, or failure to prevent corruption and bribery in operations and the supply chain, can result in financial, operational, legal, and reputational risks, including high employee turnover, reduced productivity, compliance breaches, and loss of trust with stakeholders and patients. | • | • | • | • | |||
Business Conduct - Privacy | Global data privacy | Potential Negative Impact | Genmab handles the data of patients, employees, business partners, healthcare professionals and other stakeholders. Despite prioritizing the privacy and protection of personal data, there is an inherent potential negative impact. | • | • | • | • | • | • |
Business Conduct - Protection of whistle- blower | Protection of whistleblowers | Potential Negative Impact | Failing to protect whistleblowers could discourage the reporting of incidents or unethical and unlawful behavior, potentially leading to negative impacts on patients and undermining trust in Genmab’s operations. | • | • | • | • | • | • |
Business Conduct - Animal Welfare | Animal welfare | Actual Negative Impact | As part of developing new therapies, Genmab conducts preclinical studies involving animals before testing in humans. Failure to ensure appropriate care and minimize potential adverse impacts during research could compromise animal welfare. | • | • | • | • | ||
Business Conduct - Management of relationships with suppliers (including payment practices | Management of suppliers | Potential Negative Impact | Without strong ethical standards for good supplier payment practices and responsible sourcing, Genmab could be prone to supply chain risks compromising Genmab’s ethical standards and patient’s access to treatment. Supplier relationship management is a key focus for Genmab, aimed at building strong, mutually beneficial partnerships. | • | • | • | • | ||
 | Genmab 2025 Annual Report | 71 | |||||
E1: Climate Change E2: Pollution E3: Water and Marine Resources E4: Biodiversity and Ecosystems E5: Circular Economy | S1: Own Workforce •Working conditions •Equal treatment and opportunities for all •Other work-related rights S2: Workers in the Value Chain S3: Affected Communities S4: Consumers and End- Users •Social inclusion of consumers and/or end-users •Personal safety and information of consumers and/or end-users | G1: Business Conduct •Corporate culture •Protection of whistle-blowers •Animal welfare •Management of relationships with suppliers •Corruption and bribery •Political engagement and lobbying | ||
Grey text denotes Non-Material | ||||
 | Genmab 2025 Annual Report | 72 | |||||

 | Genmab 2025 Annual Report | 73 | |||||
Section | Disclosure Requirement Content | Disclosure Requirement # | |
E1 Climate Change | |||
2.0 Climate Change Strategy | Transition plan for climate change mitigation | E1-1 | |
2.1 Climate Change IRO management | Policies related to climate change mitigation and adaptation | E1-2 | |
2.2 Climate Change Actions, Metrics and Targets | Actions and resources in relation to climate change policies | E1-3 | |
Targets related to climate change mitigation and adaptation | E1-4 | ||
Energy consumption and mix | E1-5 | ||
Gross Scopes 1, 2, 3 and total GHG emissions | E1-6 | ||
GHG removals and GHG mitigation projects financed through carbon credits | E1-71 | ||
Internal carbon pricing | E1-81 | ||
Anticipated financial effects from material physical and transition risks and potential climate-related opportunities | E1-92 | ||
EU Taxonomy | |||
2.3 EU Taxonomy | Reporting according to the EU Taxonomy | N/A | |
Actual Negative Impact | GHG emissions from own operations and value chain | |
Risk | Transitional and physical risks related to GHG emissions | |
Opportunity | Partner with value chain to reduce Scope 3 emissions |
 | Genmab 2025 Annual Report | 74 | |||||
 | Genmab 2025 Annual Report | 75 | |||||
Policy | IRO Mapping | Policy Content and Objectives | Scope of the Policy | Accountability | External Standards or Commitments | Stakeholder Consideration | Accessibility / Communication |
Commitment to the Environment and Sustainability | GHG emissions from own operations and value chain | Establishes our approach to managing material environmental topics. Its objective is to guide responsible environmental practices across all operations. | Applies to all employees, contractors, and operations globally | CSR & Sustainability Committee | Guided by the Paris Agreement of the United Nations Framework Convention on Climate Change | Developed with input from internal and external experts and stakeholders | Available on internet and intranet |
IRO | Key Actions in 2025 | Targets | Outcomes / Tracking Effectiveness | Stakeholder Involvement |
GHG emissions from own operations and value chain | We developed a sustainability roadmap as an integral part of Genmab’s sustainable climate-related strategy, in collaboration with an external expert consultant, focusing on quantifying investments, impacts, and feasibility to ensure structured and prioritized implementation of initiatives aligned with our sustainability targets. | Develop and execute on sustainable climate-related strategy by 2025.1 | Achieved in 2025. The development of our sustainability roadmap has enhanced our ability to govern and sequence sustainability initiatives. Genmab has executed on this strategy with significant progress on market-based Scope 2 GHG emission reductions in 2025. | Facility Management, R&D Operations, and External Environmental Sustainability Expert Consultant |
We expanded the use of renewable electricity to additional Genmab sites globally to advance our target of reducing Scope 2 emissions. Specifically, we began sourcing renewable electricity by use of unbundled renewable energy certificates at our sites in China during 2025. | Reduce Scope 1 and Scope 2 (market-based) emissions by 42% through a reduction in Scope 2 emissions by 2030 from a 2024 base year.2 Reduce Scope 1 and 2 (market-based) emissions by 90% by 2050 from a 2024 base year | In progress for both targets. We plan to continue using energy attribute certificates (EACs) as the primary decarbonization lever to reduce Scope 2 emissions to achieve our 2030 GHG emissions reduction target. Additional levers are under investigation, and their quantitative impacts will be disclosed when available. | Facility Management, Landlords and Utility Providers | |
Genmab monitored the climate ambitions of our top suppliers to ensure traction towards our 2030 target of at least 70% (by spend) of our suppliers having a science- aligned target. | Ensure 70% of suppliers by spend covering upstream purchases goods and services, capital goods and upstream transportation commit to have science-aligned targets by 2030.3 | In progress. The benchmarking confirmed that we remain on track to meet our 2030 target and enabled us to identify priority areas for supplier engagement, highlighting action hotspots where targeted collaboration will have the greatest impact. | Suppliers and Procurement. |
 | Genmab 2025 Annual Report | 76 | |||||
2025 | 20243 | |||
1 | Total fossil energy consumption | MWh | 4,469 | 5,120 |
Share of fossil sources in total energy consumption | % | 35% | 40% | |
2 | Consumption from nuclear sources | MWh | — | 92 |
Share of consumption from nuclear sources in total energy consumption | % | —% | 1% | |
3 | Fuel consumption for renewable sources, including biomass (also comprising industrial and municipal waste of biologic origin, biogas, renewable hydrogen, etc.) | MWh | — | — |
4 | Consumption of purchased or acquired electricity, heat, steam, and cooling from renewable sources | MWh | 8,143 | 7,414 |
5 | The consumption of self- generated non-fuel renewable energy | MWh | 117 | 77 |
6 | Total renewable energy consumption1 | MWh | 8,260 | 7,491 |
Share of renewable sources in total energy consumption | % | 65% | 59% | |
Total energy consumption2 | MWh | 12,729 | 12,703 |
Milestones and Target Years | |||||||
2025 | Base Year 20244 | % Change | 2030 | 2050 | Annual % Target/ Base Year3 | ||
Scope 1 GHG emissions1 | |||||||
Gross Scope 1 GHG emissions (tCO2eq) | 758 | 662 | 15% | 662 | 67 | —% | |
Scope 2 GHG emissions | |||||||
Gross location-based Scope 2 GHG emissions (tCO2eq) | 2,658 | 2,705 | (2)% | ||||
Gross market-based Scope 2 GHG emissions (tCO2eq) | 41 | 1,163 | (96)% | 397 | 116 | 7% | |
Total Scope 1 and market-based Scope 2 GHG emissions (tCO2eq) | 799 | 1,825 | (56)% | 1,059 | 183 | 7% | |
Significant Scope 3 GHG emissions2 | |||||||
Total Gross indirect (Scope 3) GHG emissions (tCO2eq) | |||||||
1 - Purchased Goods and services | 192,922 | 164,449 | 17% | ||||
2 - Capital goods | 7,746 | 5,519 | 40% | ||||
3 - Fuel and energy-related Activities (not included in Scope 1 or Scope 2) | 1,119 | 1,112 | 1% | ||||
4 - Upstream transportation and distribution | 5,877 | 5,425 | 8% | ||||
6 - Business travel | 10,784 | 10,559 | 2% | ||||
7 - Employee commuting | 1,002 | 946 | 6% | ||||
Total Scope 3 GHG emissions | 219,450 | 188,010 | 17% | ||||
Total GHG emissions | |||||||
Total GHG emissions (location- based) (tCO2eq) | 222,866 | 191,377 | 16% | ||||
Total GHG emissions (market- based) (tCO2eq) | 220,249 | 189,835 | 16% | ||||
 | Genmab 2025 Annual Report | 77 | |||||
GHG intensity per net revenue | 2025 | 2024 |
Total GHG emissions (location-based) per net revenue (tCO2eq/ USD million) | 59.9 | 61.3 |
Total GHG emissions (market-based) per net revenue (tCO2eq/ USD million) | 59.2 | 60.8 |
 | Genmab 2025 Annual Report | 78 | |||||


 | Genmab 2025 Annual Report | 79 | |||||
Financial Year (N) | 2025 | |||||||||||||||||||
KPI (1) | Total (2) | Proportion of Taxonomy eligible activities (3) | Taxonomy aligned activities (4) | Proportion of Taxonomy aligned activities (5) | Breakdown by environmental objectives of Taxonomy aligned activities | Proportion of enabling activities (12) | Proportion of transitional activities (13) | Not assessed activities considered non- material (14) | Taxonomy aligned activities in previous financial year (N-1) (15) | Proportion of Taxonomy aligned activities in previous financial year (N-1) (16) | ||||||||||
Climate Change Mitigation (6) | Climate Change Adaption (7) | Water (8) | Circular Economy (9) | Pollution (10) | Biodiversity (11) | |||||||||||||||
MUSD | % | MUSD | % | % | % | % | % | % | % | % | % | % | MUSD | % | ||||||
Turnover | 3,720 | 11% | 0 | —% | —% | —% | —% | —% | —% | —% | —% | —% | —% | 0 | —% | |||||
CapEx | 7,469 | 0.4% | 0 | —% | —% | —% | —% | —% | —% | —% | —% | —% | —% | 0 | —% | |||||
OpEx | 1,560 | —% | 0 | —% | —% | —% | —% | —% | —% | —% | —% | —% | —% | 0 | —% | |||||
Reported KPI | Turnover | ||||||||||||
Financial Year (N) | 2025 | ||||||||||||
Economic Activities (1) | Code (2) | Taxonomy eligible KPI (Proportion of Taxonomy eligible Turnover) (3) | Taxonomy aligned KPI (monetary value of Turnover) (4) | Taxonomy aligned KPI (Proportion of Taxonomy aligned Turnover) (5) | Environmental objective of Taxonomy aligned activities | Enabling Activity (12) | Transitional Activity (13) | Proportion of Taxonomy aligned in Taxonomy eligible (14) | |||||
Climate Change Mitigation (6) | Climate Change Adaption (7) | Water (8) | Circular Economy (9) | Pollution (10) | Biodiversity (11) | ||||||||
% | MUSD | % | % | % | % | % | % | % | (E where applicable) | (T where applicable) | % | ||
Manufacture of medicinal products | PPC 1.2 | 11% | 0 | —% | —% | —% | —% | —% | —% | —% | —% | ||
Sum of alignment per objective | % | % | % | % | % | % | |||||||
Total Turnover | 11% | 0 | —% | —% | —% | —% | —% | —% | —% | —% | |||

 | Genmab 2025 Annual Report | 80 | |||||
Reported KPI | CapEx | ||||||||||||
Financial Year (N) | 2025 | ||||||||||||
Economic Activities (1) | Code (2) | Taxonomy eligible KPI (Proportion of Taxonomy eligible CapEx) (3) | Taxonomy aligned KPI (monetary value of CapEx) (4) | Taxonomy aligned KPI (Proportion of Taxonomy aligned CapEx) (5) | Environmental objective of Taxonomy aligned activities | Enabling Activity (12) | Transitional Activity (13) | Proportion of Taxonomy aligned in Taxonomy eligible (14) | |||||
Climate Change Mitigation (6) | Climate Change Adaption (7) | Water (8) | Circular Economy (9) | Pollution (10) | Biodiversity (11) | ||||||||
% | (MUSD) | % | % | % | % | % | % | % | (E where applicable) | (T where applicable) | % | ||
Renovation of existing buildings | CCM 7.2 | 0.4% | 0 | —% | —% | —% | —% | —% | —% | —% | —% | ||
Sum of alignment per objective | % | % | % | % | % | % | |||||||
Total CapEx | 0.4% | 0 | —% | —% | —% | —% | —% | —% | —% | —% | |||

 | Genmab 2025 Annual Report | 81 | |||||

Section | Disclosure requirement content | Disclosure requirement # | |
3.0 Own Workforce IRO Management | Policies related to own workforce | S1-1 | |
Processes for engaging with own workers and workers’ representatives about impacts | S1-2 | ||
Processes to remediate negative impacts and channels for own workers to raise concerns | S1-3 | ||
3.1 Own Workforce Actions, Metrics and Targets | Taking action on material impacts on own workforce, and approaches to mitigating material risks and pursuing material opportunities related to own workforce, and effectiveness of those actions | S1-4 | |
Targets related to managing material negative impacts, advancing positive impacts, and managing material risks and opportunities | S1-5 | ||
Characteristics of the Company’s employees | S1-6 | ||
Characteristics of non-employee workers in the Company’s own workforce | S1-71 | ||
Collective bargaining coverage and social dialogue | S1-8 | ||
Diversity metrics | S1-9 | ||
Adequate wages | S1-10 | ||
Social protection | S1-11 | ||
Persons with disabilities | S1-121 | ||
Training and skills development metrics | S1-13 | ||
Health and safety metrics | S1-14 | ||
Work-life balance metrics | S1-151 | ||
Compensation metrics (pay gap and total compensation) | S1-16 | ||
Incidents, complaints and severe human rights impacts | S1-17 |
Actual Positive Impact | Employee well-being and vitality | |
Risk | Attracting and retaining talent to enable continued innovation | |
Opportunity | Provide a voice to employees through our global engagement survey | |
Potential Negative Impact | Safety in our facilities | |
Actual Positive Impact | Career development through training and skill building | |
Actual Positive Impact | Equal opportunity promoting innovation |
 | Genmab 2025 Annual Report | 82 | |||||
Policy/ Commitment | IRO Mapping | Policy/Commitment Content and Objectives | Scope | Accountability | External Standards or Commitments | Stakeholder Consideration | Accessibility / Communication |
Code of Conduct | Covers all Social S1 IROs | Defines our commitment to conducting business ethically and in compliance with applicable legal, regulatory, and industry code requirements. The objective is to translate the principles of the Code into clear global compliance expectations, guiding interactions with healthcare professionals, healthcare organizations, patients, patient advocacy groups, public officials, and other stakeholders, and supporting consistent, lawful, and ethical conduct across all business activities. | Applies to all employees, Board members, and third parties. | SVP, Global Compliance, Risk, and Data Privacy. | Supports alignment with: •Applicable global and local laws including but not limited to Foreign Corrupt Practices Act, False Claims Act, Anti-Kickback Statute, UK Bribery Act, Sarbanes-Oxley Act. •Regulations set forth by government agencies such the FDA and EMA. •GDPR and other applicable privacy and data protection guidelines. •Internationally recognized industry codes/ethical standards including EFPIA, PhRMA Codes of Practice, and the UN Convention against Corruption. | Reflects the expectations of employees, patients, business partners, regulators, and society by setting clear standards for ethical behavior, legal compliance, and responsible business conduct. | Available on internet, intranet, and integrated into onboarding and annual Code of Conduct training. |
Global Speak Up Policy | Employee well- being and vitality | Shares our commitment to fostering a culture of openness, integrity, and accountability where concerns can be raised without fear of retaliation. The objective is to encourage employees and other relevant stakeholders to report suspected misconduct, unethical behavior, or violations of laws, policies, or the Code of Conduct, and to ensure that such concerns are addressed promptly, fairly, and confidentially. | Applies to all employees and external parties, such as contractors, consultants, and other third parties. | SVP, Global Compliance, Risk, and Data Privacy. | Supports alignment with expectation of effective compliance programs and applicable laws/regulations (e.g. EU Whistleblower Protection Directive; Dutch Whistleblower Protection Act, GDPR, etc.). | Supports employees and third parties by providing safe and confidential channels to raise concerns, reinforcing trust, accountability, and ethical conduct. | Available on internet, intranet, and integrated into onboarding and annual Code of Conduct training. |
Human Rights Commitment | Employee well- being and vitality | Defines our responsibility to respect human dignity across our operations, guided by international human rights and labor standards. Its objectives are to prevent human rights impacts, ensure fair employment practices, prohibit forced or child labor, protect privacy, and uphold these standards across our operations and suppliers. Genmab does not discriminate based on race, ethnicity, color, religion, sex, gender identity and expression, national origin, age, disability, genetic information, sexual orientation, military, veteran or other protected status. | Applies to all employees and third parties acting on behalf of Genmab and extends to all workers in the value chain. | EVP, Chief People Officer. | Guided by human rights laws and the UN Guiding Principles, Genmab aligns with the International Bill of Human Rights and the ILO’s core labor standards. | Developed with consideration of employees, suppliers, patients, and other affected stakeholders. | Available on internet, intranet, and integrated into supplier expectations. |
 | Genmab 2025 Annual Report | 83 | |||||
Policy/ Commitment | IRO Mapping | Policy/Commitment Content and Objectives | Scope | Accountability | External Standards or Commitments | Stakeholder Consideration | Accessibility / Communication |
Global Workforce Culture Policy | Equal opportunity promoting innovation | Describes our commitment to fostering an equitable and inclusive workplace where all employees are valued and treated fairly. Its objectives are to ensure equal opportunities, prevent discrimination and harassment, and enable a safe environment that drives innovation, strong performance, and positive impact across our workforce and value chain. It covers inclusion for groups that may be at particular risk of vulnerability regardless of gender, race, ethnicity, religion, age, disability, and other protected characteristics, and as noted in our Code of Conduct, all forms of harassment and retaliation are unacceptable and counter to everything we stand for as a company. | Applies to all employees and suppliers; extends to all workers in the value chain. | EVP, Chief People Officer | Aligns with widely recognized external standards, local statutory frameworks, and commitments, including Title VII of the U.S. Civil Rights Act (and corresponding U.S. and state laws), the UN Guiding Principles on Business and Human Rights, Universal Declaration of Human Rights, International Labour Organization (ILO) Declaration on Fundamental Principles and Rights at Work, ISO 26000 - Social Responsibility, UN Sustainable Development Goals (SDGs), UN Global Compact Principles, and OECD Guidelines for Multinational Enterprises. | Developed with employee feedback to foster an inclusive, fair, and responsible workplace culture. | Available on internet and intranet. |
Corporate Social Responsibility (CSR) Policy | Career development through training and skill building | Outlines how the Company integrates its purpose, values, and vision into responsible business practices focused on patient-centered innovation, caring for employees and communities, business integrity, and environmental sustainability. Its objective is to guide ethical, transparent, and sustainable operations, supported by clear governance and oversight, and ensure all employees incorporate CSR principles into daily decisions. | Applies across all operations and employee groups. | SVP, Communications & Corporate Affairs | Aligns with UN Universal Declaration of Human Rights (UDHR) and UN Guiding Principles on Business and Human Rights (UNGPs), and International Labour Organization (ILO). | Developed considering employee welfare, governance, and long-term sustainability goals. | Available on internet and intranet. |
Health and Safety Commitment | Safety in our facilities | Details our dedication to maintaining safe, healthy, and supportive environments across all sites through proactive risk management, strong governance, and continuous improvement. Its objective is to prevent workplace injuries and illnesses, foster a shared culture of safety, and ensure resilient, high-performing operations that support our broader sustainability and business goals. | Applies across all sites; covers employees, contractors, and visitors. | EVP, Chief People Officer | Aligns with ILO Occupational Safety and health and safety standards; ISO 45001 and national and local OHS laws; UN Universal Declaration of Human Rights. | Developed with employee safety input and local operational oversight. | Available on internet and intranet. |
Global Lab Occupational Health and Safety Policy | Safety in our facilities | Defines our approach to maintaining safe R&D lab environments through site-level health and safety committees and prevention measures. Its objective is to protect employees by ensuring strong safety oversight, issue escalation, and continuous improvement across all lab facilities. | Applies to all R&D laboratories and related operations globally. | EVP, Chief Medical Officer | Aligns with applicable occupational health, safety and environmental legislation in China, the Netherlands, Denmark, and the United States, including the Dutch Working Conditions and environmental permitting framework, EU REACH/CLP, and relevant U.S. federal and state regulations such as OSHA, EPA, DOT, and applicable fire, biosafety and transport of dangerous goods requirements. | Developed considering employee safety needs in lab environments. | Available on intranet and communicated internally to all lab staff through site operations and training. |
 | Genmab 2025 Annual Report | 84 | |||||

 | Genmab 2025 Annual Report | 85 | |||||
IRO | Key Actions in 2025 | Targets | Outcomes/Tracking Effectiveness | Stakeholder Involvement |
Employee well- being and vitality Provide a voice to employees through our global engagement survey | Annual Employee Engagement Survey: Provided employees with a meaningful voice through our annual survey, offering insights into key areas such as career development, innovation, leadership, and the work environment. Global Well-Being: Strengthened our well-being pillars—emotional, financial, physical, and social—by supporting mental health and resilience; promoting financial security through initiatives such as Global Money Week; offering on- site and virtual fitness options; and fostering community connection. We also expanded our Global Well-Being Programs, providing additional workshops and resources focused on financial planning, work–life balance, and holistic self-care. Emotional and Mental Health: Continued offering programs and local resources to support emotional and mental well-being. We announced the launch of a new global resource through Spring Health—providing care navigation, coaching, in-person and virtual therapy, self-guided digital exercises, and educational content—which will be available to all employees beginning January 2026. Volunteering: Organized events throughout the year to connect with each other and our communities. In 2025, 770 team members volunteered 3,135 hours on Global Volunteer Day compared to 688 team members and 2,952 hours in 2024. Work-life Balance: Continued offering four additional days off and four meeting-free days annually, alongside paid time off, leave-of-absence policies, and family-related leave for all full-time employees in line with local regulations. Maintained the tradition of a late-December holiday office closure to support rest and connection outside of work. Total Rewards & Opportunities: Advanced our ongoing efforts to support employee understanding of Total Rewards & Opportunities, including salary growth potential, equity grants, and market-competitive benefits across well- being dimensions. We also launched a new global reward and recognition program and tool to facilitate timely feedback, offer opportunities for cash rewards, and strengthen a culture of appreciation across the organization. | Meet or exceed the global benchmark for (1) employee engagement score and (2) participation rate for the Global Employee Engagement Survey (Annual)1 | Target achieved for 2025. In 2025, Genmab achieved a 77% engagement score compared to 77% industry average, and an 89% global participation rate compared to an 80% industry average. The engagement score reflects overall favorability based on key questions designed to measure employee engagement, while the participation rate indicates how broadly employees are contributing feedback relative to industry norms. The favorability score is calculated by the percent of agreement responses when combining scale points 4-5. Genmab compares to the Life and Sciences Sector for the industry benchmark provided by Mercer. This provides a comparison of employees answering surveys conducted over a number of years for organizations within hospital, health systems, insurance, pharmaceuticals, and medical research and development industry. These results allow us to track progress year over year, identify patterns, and assess the effectiveness of our actions. Insights from the survey help us monitor Genmab’s areas of strength and opportunity. To deepen understanding of critical engagement issues, we also conduct focus groups and share survey results across the organization. People leaders are encouraged to review team-level feedback and develop targeted actions to improve the employee experience. Executive Management reviews the findings collectively to analyze trends, reflect on strengths and opportunities, and guide organizational priorities. In addition, Genmab regularly hosts functional and regional town halls to update teams on business progress and reinforce transparency. By consistently leveraging our global engagement survey and follow-up actions, Genmab creates a workplace where employees feel valued, motivated, and committed to achieving our shared goals. | Results are reviewed by Executive Management and people leaders, shared transparently across our internal network, and used collaboratively to guide action planning across teams. |
1.Executive Management received RSU grants in 2023, 2024 and 2025 with performance linked to sustaining at or better than the global benchmark for employee engagement. |
 | Genmab 2025 Annual Report | 86 | |||||
IRO | Key Actions in 2025 | Targets | Outcomes/Tracking Effectiveness | Stakeholder Involvement |
Safety in our facilities | Training: Provided mandatory safety training and continuous education, focusing on hazardous materials in our labs. Our chemical management meets all regulations, prioritizes employee and environmental safety, and monitors high-risk substances. In 2025, no chemical-related incidents required more than first aid. Annual Safe & Sound Day: Demonstrates Genmab’s continued focus on workplace health, safety, and employee well-being. Colleagues across our sites in the Netherlands, Denmark, and the US took part in coordinated activities designed to enhance safety awareness and practical response skills. The program included the launch of Genmab’s global Safety Culture Ladder, first aid and firefighter training, interactive safety games, CPR workshops, vendor fairs, and live safety demonstrations. These events fostered engagement and strengthened our shared commitment to a safe and supportive work environment. | None | We have formal committees responsible for monitoring and improving health and safety at all locations. Our chemical management team met all regulations, prioritized employee and environmental safety, and monitored high-risk substances. | Under the leadership of our Chief Medical Officer, our committees are responsible for ensuring compliance. |
Career Development through Training and Skill Building | Learning & Development: Leveraged multiple skills-based in-person workshops and digital learning paths for employees focused on specific skill development with focus areas including leadership, digital and AI, feedback, strategic planning, advance Excel skills, leading different generations, informal leadership, and business communications. Global Mentorship Program: Extended the Program to ensure an inclusive and supportive environment, designed to impact the sense of belonging, as employees connect with other colleagues outside their area of expertise as well as connecting with leaders who can provide insights and feedback not linked to any performance plan. The feedback can be used to develop skills and traits important to that specific individual. Sustainability Awareness Training: Launched training to all Genmab employees. | 1.100% of employees are provided access to training programs that meet the development needs across various career stages and learning styles (Annual) 2.100% of eligible employees are provided access to Genmab’s end of year performance process (Annual)2 3.Launch sustainability awareness training by 2025 | 1.Target achieved for 2025. The Global Talent and Culture team ensured that required trainings were offered to all employees. 2.Target achieved for 2025. The Global Talent and Culture team ensured all eligible employees were provided full access to Genmab’s year-end performance process, including providing internal resources and early engagement. 3.Target achieved for 2025. The Corporate Sustainability Team launched the sustainability awareness training for employees in 2025. | 1.Under the leadership of the Global Talent and Culture team, all employees are provided access. 2.Under the leadership of the Global Talent and Culture team, all eligible employees are provided access. 3.Under the leadership of the Corporate Sustainability Team, all employees are provided access to Sustainability Awareness training. |
Equal Opportunity Promoting Innovation | Culture Trainings: Offered a combination of culture workshops and masterclasses to our workforce. Employee Resource Groups (ERGs): Made ERGs available to all employees to foster a collaborative culture where unique perspectives drive innovation, engagement, and organizational strength—empowering employees to contribute meaningfully to our mission of developing the next generation of antibody medicines for patients in need. | Target between 40% to 60% gender representation by 2025 in the Other Management Levels at Genmab A/S only in accordance with the guidelines from the Danish Business Authority (DBA). In 2025, Genmab removed gender diversity targets at the Group level including those linked to Executive Management compensation. | Target not achieved for 2025. As of December 31, 2025, women represented 30% (seven) and men 70% (16) of managers in the “Other Management Levels” of Genmab A/S, as defined by the Danish Gender Balance Act. Other Management Levels are comprised of Executive Management and employees with personnel responsibilities who report to Executive Management. As Genmab does not currently have an equal gender representation in the Other Management Levels, with women being the underrepresented gender, the Board of Directors has decided to maintain a target for the proportion of women in the Other Management Levels of 40%, or, depending on the specific number of individuals to be included in the Other Management Levels at the given time, the percentage that comes closest to 40%, but not exceeding 49%, by 2028. | The Global Talent and Culture team, within the Human Resource team. |
 | Genmab 2025 Annual Report | 87 | |||||
December 31, | |||||||
2025 | 20242 | 2023 | |||||
Employees (Headcount) | Female | Male | Total | Female | Male | Total | Total |
Denmark | 345 | 254 | 599 | 326 | 228 | 554 | 495 |
Netherlands | 461 | 321 | 782 | 490 | 335 | 825 | 740 |
U.S. | 647 | 421 | 1,068 | 642 | 432 | 1,074 | 887 |
Japan | 62 | 158 | 220 | 55 | 131 | 186 | 140 |
China | 69 | 63 | 132 | 55 | 56 | 111 | — |
Other Countries | 14 | 4 | 18 | — | — | — | — |
Total Headcount | 1,598 | 1,221 | 2,819 | 1,568 | 1,182 | 2,750 | 2,262 |
December 31, | |||||||
20251 | 20242 | 2023 | |||||
Employees (FTEs)1 | Female | Male | Total | Female | Male | Total | Total |
Permanent | 1,517 | 1,168 | 2,685 | 1,498 | 1,136 | 2,634 | 2,159 |
Temporary | 32 | 24 | 56 | 28 | 20 | 48 | 45 |
Total FTEs | 1,549 | 1,192 | 2,741 | 1,526 | 1,156 | 2,682 | 2,204 |
December 31, | ||||||
FTEs (R&D vs. SG&A)1 | 20251 | 2024 | 2023 | |||
Research and development FTE | 1,880 | 1,886 | 1,541 | |||
Selling, general and administrative FTE | 861 | 796 | 663 | |||
December 31, | |||||||
Turnover | 2025 | 2024 | 2023 | ||||
# of FTEs leaving Genmab | 321 | 190 | 157 | ||||
Turnover Rate - Overall | 12% | 7% | 8% | ||||
Turnover Rate - Voluntary | 7% | 6% | 5% | ||||
Collective Bargaining Coverage | Social Dialogue | |
Coverage Rate | Employees - EEA | Workplace representation (EEA only) |
0-19% | ||
20-39% | ||
40-59% | ||
60-79% | ||
80-100% | Denmark, Netherlands |
 | Genmab 2025 Annual Report | 88 | |||||
2025 | 2024 | 2023 | |||||||
Female | Male | Total | Female | Male | Total | Female | Male | Total | |
Board of Directors, Shareholder-Elected1 | 3 | 3 | 6 | 3 | 3 | 6 | 3 | 3 | 6 |
% of total | 50% | 50% | 100% | 50% | 50% | 100% | 50% | 50% | 100% |
Board of Directors, Employee-Elected1 | 1 | 2 | 3 | 1 | 2 | 3 | 1 | 2 | 3 |
% of total | 33% | 67% | 100% | 33% | 67% | 100% | 33% | 67% | 100% |
Board of Directors, Total | 4 | 5 | 9 | 4 | 5 | 9 | 4 | 5 | 9 |
% of total | 44% | 56% | 100% | 44% | 56% | 100% | 44% | 56% | 100% |
Registered Executive Management | 0 | 2 | 2 | 0 | 2 | 2 | 0 | 2 | 2 |
% of total | —% | 100% | 100% | —% | 100% | 100% | —% | 100% | 100% |
Executive Management (Top Management), Total | 2 | 7 | 9 | 3 | 6 | 9 | 3 | 5 | 8 |
% of total | 22% | 78% | 100% | 33% | 67% | 100% | 38% | 62% | 100% |
Age | 2025 | 2024 | 2023 |
< 30 | 9% | 11% | 13% |
30 - 50 | 64% | 63% | 63% |
> 50 | 27% | 26% | 24% |
100% | 100% | 100% |

 | Genmab 2025 Annual Report | 89 | |||||
2025 | 2024 | |||
Training and skills development metrics | Female | Male | Female | Male |
% of Employees Who Have Participated in Performance Reviews/ Career Conversations | 100% | 100% | 100% | 100% |
Average Number of Training Hours | 5 | 5 | 10 | 8 |
Health and safety metrics | 2025 | 2024 |
Percentage of own workforce covered by the Health and Safety management system | 100% | 100% |
Number of fatalities | 0 | 0 |
Number of recordable work-related accidents | 2 | 0 |
Rate of recordable work-related accidents (accidents per million hours worked) | 0.4 | 0 |

Remuneration Metrics | 2025 | 2024 |
Gender pay gap - Overall | 9% | 12% |
Gender pay gap - Excluding Executive Management | 2% | 5% |
CEO pay ratio | 76 | 64 |
 | Genmab 2025 Annual Report | 90 | |||||
Incidents, complaints and severe human rights impact metrics | 2025 | 2024 |
Total number of incidents of discrimination reported, including harassment | 10 | 0 |
Substantiated number of incidents of discrimination, including harassment | 4 | 0 |
Number of complaints filed through channels for own workforce, excluding incidents of discrimination and harassment | 0 | 0 |
Number of complaints filed through National Contact Points for OECD Multinational Enterprises | 0 | 0 |
Fines, penalties, and compensation for damages as a result of the incidents and complaints of discrimination, including harassment (in USD) | 0 | 0 |
Number of severe human rights incidents for own workforce | 0 | 0 |
Fines, penalties, and compensation for damages as a result of severe human rights incidents (in USD) | 0 | 0 |


 | Genmab 2025 Annual Report | 91 | |||||
Section | Disclosure requirement content | Disclosure requirement # | |
4.0 Consumers & End-Users IRO Management | Policies related to consumers and end-users | S4-1 | |
Processes for engaging with consumers and end-users about impacts | S4-2 | ||
Processes to remediate negative impacts and channels for consumers and end-users to raise concerns | S4-3 | ||
4.1 Consumers & End-Users Actions, Metrics and Targets | Taking action on material impacts on consumers and end-users, and approaches to managing material risks and pursuing material opportunities related to consumers and end-users, and effectiveness of those actions | S4-4 | |
Targets related to managing material negative impacts, advancing positive impacts, and managing material risks and opportunities | S4-5 |
Actual Positive Impact | Innovation for patients with unmet needs | |
Risk | Research and development risk | |
Potential Negative Impact | Access and inclusion in clinical trials | |
Risk | Regulation, Legislation, and Compliance | |
Potential Negative Impact | Responsible and ethical marketing | |
Actual Positive Impact | Patient voice | |
Potential Negative Impact | Health and safety of patients | |
Risk | Pharmacovigilance risks as a biotech company | |
Potential Negative Impact | Access to quality information |
 | Genmab 2025 Annual Report | 92 | |||||
Policy/ Commitment | IRO Mapping | Policy/Commitment Content and Objectives | Scope | Accountability | External Standards or Commitments | Stakeholder Consideration | Accessibility / Communication |
Code of Conduct | Access and inclusion in clinical trials; Responsible and ethical marketing | Defines our commitment to conducting business ethically and in compliance with applicable legal, regulatory, and industry code requirements. The objective is to translate the principles of the Code into clear global compliance expectations, guiding interactions with healthcare professionals, healthcare organizations, patients, patient advocacy groups, public officials, and other stakeholders, and supporting consistent, lawful, and ethical conduct across all business activities. | Applies to all employees, Board members, and third parties. | SVP, Global Compliance, Risk, and Data Privacy | Supports alignment with: •Applicable global and local laws including but not limited to Foreign Corrupt Practices Act, False Claims Act, Anti-Kickback Statute, UK Bribery Act, Sarbanes-Oxley Act. •Regulations set forth by government agencies such as the FDA and EMA. •GDPR and other applicable privacy and data protection guidelines. •Internationally recognized industry codes/ethical standards including EFPIA, PhRMA Codes of Practice, and the UN Convention against Corruption. | Reflects the expectations of employees, patients, business partners, regulators, and society by setting clear standards for ethical behavior, legal compliance, and responsible business conduct. | Available on internet, intranet, and integrated into onboarding and annual Code of Conduct training. |
Human Rights Commitment | Health and safety of patients | Defines our responsibility to respect human dignity across our operations, guided by international human rights and labor standards. Its objectives are to prevent human rights impacts, ensure fair employment practices, prohibit forced or child labor, protect privacy, and uphold these standards across our operations and suppliers. Genmab does not discriminate based on race, ethnicity, color, religion, sex, gender identity and expression, national origin, age, disability, genetic information, sexual orientation, military, veteran or other protected status. | Applies to all employees and third parties acting on behalf of Genmab and extends to all workers in the value chain. | EVP, Chief People Officer | Guided by human-rights laws and the UN Guiding Principles, Genmab aligns with the International Bill of Human Rights and ILO’s core labor standards. | Developed considering employees, suppliers, patients, and other affected stakeholders. | Available on internet, intranet, and integrated into supplier expectations. |
Commitment to Quality | Health and safety of patients; Access to quality information | Reflects our commitment to developing, manufacturing, and delivering antibody therapies that meet applicable regulatory requirements and patient needs. The objective is to ensure the consistent delivery of safe, effective, and compliant products through a robust quality management system, adherence to global regulatory standards, protection of patient safety, and continuous improvement across the product lifecycle. Genmab maintains comprehensive quality and safety processes to identify and mitigate product-related risks and works with relevant authorities and stakeholders to support product safety and efficacy. | Applies to all operations and suppliers; extends to all workers in the value chain. | EVP, Chief Development Officer | Aligns with applicable US FDA and EU regulatory requirements, including Good Manufacturing Practice, and internationally recognized standards such as the ICH guidelines governing quality, clinical development, and product safety, and Japan PMDA. | Developed with employee feedback and engagement, emphasizing the importance of quality in our business. | Available on internet and intranet. |
 | Genmab 2025 Annual Report | 93 | |||||
Policy/ Commitment | IRO Mapping | Policy/Commitment Content and Objectives | Scope | Accountability | External Standards or Commitments | Stakeholder Consideration | Accessibility / Communication |
Clinical Trial Transparency Declaration | Access to quality information | Shares our commitment to the responsible, unbiased, and timely disclosure of clinical trial information. The objective of this declaration is to promote transparency in clinical research, advance scientific knowledge, and support informed decision-making by patients and healthcare professionals, while safeguarding patient privacy and intellectual property. The commitments apply to all Genmab-sponsored interventional clinical trials from Phase 1 onwards and, where applicable, to non- interventional studies and expanded access programs, in compliance with applicable legal and regulatory requirements. | Applies to all Genmab-sponsored interventional clinical trials from Phase 1 onwards conducted worldwide and, where applicable, to Genmab-sponsored non-interventional clinical studies and expanded access programs. | EVP, Chief Development Officer | Aligned with applicable global and national clinical trial disclosure requirements and internationally recognized standards, including WHO guidance on clinical trial registration, and PhRMA/EFPIA principles on responsible data sharing. | Addresses the needs of patients, healthcare professionals, regulators, and the scientific community by supporting informed decision-making through transparent and responsible disclosure of clinical trial information. | Available on internet and intranet. |
Commitment to Patients | Patient voice; Innovation for patients with unmet needs | Communicates Genmab’s commitment to placing patients at the center of its activities across research, development, and access to medicines. The objective is to improve patient outcomes by integrating patient perspectives, promoting inclusive and patient-centric clinical research, supporting access to prescribed medicines, and acting with integrity and transparency in all patient-related interactions. | Applies across global operations, including clinical development, medical writing, patient advocacy, and patient access and support programs. | SVP, Communications & Corporate Affairs | Aligned with patient-focused drug development guidance (e.g., ICH and FDA PFDD); established industry codes for ethical, transparent engagement (e.g., EFPIA/PhRMA, and GDPR); responsible interactions with patients, consumers, and healthcare stakeholders (e.g., UN Guiding Principles on Business and Human Rights and OECD Guidelines for Multinational Enterprises); and best practices for integrating the patient voice into research and healthcare decision-making (e.g., National Health Council Patient Activities Framework). | Addresses the needs and perspectives of patients, care partners, healthcare professionals, regulators, and patient organizations by incorporating patient insights, supporting equitable access to medicines, while maintaining ethical and transparent engagement. | Available on internet and intranet. |
Global Compliance Policy | Responsible and ethical marketing | Reflects our commitment to conducting business ethically and in compliance with applicable legal, regulatory, and industry code requirements. The objective is to provide a common set of ethical principles and compliance expectations governing Genmab’s interactions with healthcare professionals, healthcare organizations, patients, patient advocacy groups, public officials, and other stakeholders, and to support consistent, lawful, and ethical conduct across the business. | Applies to all employees, managers, board members, and third parties. | SVP, Global Compliance, Risk, and Data Privacy | Supports alignment with: •Applicable global and local laws including but not limited to Foreign Corrupt Practices Act, False Claims Act, Anti-Kickback Statute, UK Bribery Act, Sarbanes-Oxley Act. •Regulations set forth by government agencies such as the FDA and EMA. •GDPR and other applicable privacy and data protection guidelines. •Internationally recognized industry codes/ethical standards including EFPIA, PhRMA Codes of Practice, and the UN Convention against Corruption. | Addresses the expectations of patients, healthcare professionals, healthcare organizations, regulators, public authorities, and business partners by promoting ethical, transparent, and compliant interactions and safeguarding trust in Genmab’s activities. | Available on intranet and integrated into onboarding and annual Code of Conduct training. |
 | Genmab 2025 Annual Report | 94 | |||||
Patient Engagement and Safety Genmab’s mission is reflected in its patient engagement activities across clinical development, advocacy, and support programs. MyNavCare Patient Support™ offers personalized guidance, insurance navigation, and financial assistance to help eligible patients access therapies. Patient perspectives are incorporated through the Patient Advisory Council, informing clinical trial design and treatment delivery. Engagement is overseen by the SVP of Communications and Corporate Affairs, reporting to the CEO. | Clinical Trial Transparency Genmab collaborates with patient advocacy groups and healthcare professionals to integrate stakeholder perspectives in clinical trial design and execution. Genmab’s Clinical Trial Transparency Declaration outlines its commitment to openness, with oversight by the Development Operations team under the Chief Development Officer. |
Safety and Compliance Genmab’s Global Drug Safety team ensures therapies meet rigorous safety and efficacy standards through close collaboration with regulatory authorities and continuous monitoring. Oversight is led by the Chief Development Officer, supported by cross-functional teams. | Ethical Marketing Practices Genmab promotes responsible marketing and compliance with applicable laws and industry standards. Employees receive regular training under the Code of Conduct, with oversight by the Chief Commercial Officer. |
 | Genmab 2025 Annual Report | 95 | |||||
IRO | Key Actions in 2025 | Targets | Outcomes / Tracking Effectiveness | Stakeholder Involvement |
Innovation for patients with unmet needs | Training: All Market Access colleagues complete annual compliance training to ensure regulatory and ethical adherence. Training covers Data Privacy, FDA guidelines, PhRMA Code, Ad Promotion rules, Anti-bribery, Speak Up/whistleblower, Pharmacovigilance, Sunshine Act, and Conflict of Interest. The MyNavCare team receives additional annual training on Patient Support Services (PSS) policies and business rules. | None | Global Compliance monitors training completion to ensure accountability and uphold patient access to Genmab products. | Market access team, and the MyNavCare team, overseen by Vice President, U.S. Market Access. |
Access and inclusion in clinical trials | Patient and Study Coordinator Advisory Councils: Continued involvement to inform trial design and execution. Registration: Registered all Genmab-sponsored trials on ClinicalTrials.gov to ensure transparency. Trial Access: Conducted 13 trials in 27 countries, enrolling 818 patients in 2025; compared to 25 trials in 37 countries, enrolling 620 patients in 2024. Oversight: Ensured oversight by ethics committees, regulatory authorities, and data and safety monitoring boards. Compliance: Complied with global standards (cGLP, cGCP, cGMP, FELASA) and regulatory guidance (EMA, FDA, PMDA). Monitoring: Continued post-approval safety monitoring and regular review by internal and external committees. | None | We have a Patient Advisory Council and a Study Coordinator Advisory Council who provide input to our clinical trials. In addition, our clinical trials are reviewed by institutional review boards, ethics committees, regulatory authorities, and data and safety monitoring boards. | Patient Advocacy team working closely with the Patient and Study Coordinator Advisory Councils Overseen by the SVP, Communications and Corporate Affairs. |
Responsible and ethical marketing | Training: All Marketing colleagues completed annual compliance training to ensure regulatory and ethical adherence. All promotional materials went through medical, regulatory, and legal review to ensure medically accurate, on label, and responsible promotion. | None | Genmab has established a Code of Conduct that sets high ethical standards for employees, reinforced through regular training. This ensures that all marketing and sales efforts align with local and national regulations, which may limit direct engagement with consumers and end- users. Ethical marketing practices are the responsibility of our Chief Commercial Officer. | Global Marketing team, with oversight by the EVP, Chief Commercial Officer. |
 | Genmab 2025 Annual Report | 96 | |||||
IRO | Key Actions in 2025 | Targets | Outcomes / Tracking Effectiveness | Stakeholder Involvement |
Patient voice | US Patient Advisory Council: Met multiple times during the year with discussions focused on feedback for patient-facing content (brand messaging, Medical Information resources, and digital communications), reviewing three clinical-trial lay summaries, and two unbranded resource flashcards (hematology and gynecologic cancers). Members represented diverse tumor types, age groups, geographies, and socioeconomic backgrounds. European Patient Advocate Advisory Council: Launched in late 2025 as a complement to the U.S. patient council, met twice and reviewed a clinical trial lay summary, offering culturally relevant insights that shaped the document. Fourth annual Science Day: Convened leaders and patient advisors for collaborative scientific discussions on health literacy, regulatory policy, care partner support, and education needs for hematologic and gynecologic cancers. The event produced actionable insights that are shaping patient engagement, education, and plain language communications. MyNavCare Patient Support: Provided direct assistance to patients following the U.S. launch of EPKINLY, ensuring rapid and sustainable access to therapy. Patients and care partners received support through case management, insurance navigation, and financial assistance, while healthcare providers (HCPs) were supported with reimbursement education, billing guidance, and coverage information. Patient Assistance Program: Facilitated access for eligible uninsured or underinsured patients, reducing administrative burdens and ensuring timely support. | None | The effectiveness of these patient engagement programs is tracked via regular meetings with our patient advisory council and through scheduled updates with other internal teams. Under the leadership of the Vice President, U.S. Market Access, Genmab’s Market Access function oversaw MyNavCare operations, with effectiveness tracked through regular cross-functional and leadership reviews. | Patient Advocacy team working closely with the Patient and Study Coordinator Advisory Councils. Overseen by the SVP, Communications and Corporate Affairs. |
Health and safety of patients | Training: Provided annual training programs on pharmacovigilance for global drug safety and pharmacovigilance (GDS&PV) staff, including regular updates and assessments to ensure continuous learning and compliance with the latest regulations. Also, Genmab developed and distributed educational materials on safety requirements to stakeholders and organized workshops to enhance their understanding and implementation of safety protocols. These actions are linked to the personal safety and information of consumers and end-users. | None | Genmab has safety measures in place for products, patients, and healthcare providers. Genmab believes that patient safety plays a critical role in our business operations. Genmab has a Comprehensive Safety Program that is designed to identify and mitigate potential risks associated with our products, and to ensure that our products are safe and effective for their intended use. We work closely with regulatory agencies to ensure that our products meet all safety and efficacy standards. | Global Drug Safety & Pharmacovigilance team. Overseen by the EVP, Chief Development Officer. |
 | Genmab 2025 Annual Report | 97 | |||||
IRO | Key Actions in 2025 | Targets | Outcomes / Tracking Effectiveness | Stakeholder Involvement |
Access to quality information | Provided clinical trial information and results on publicly accessible registries, including ClinicalTrials.gov and the EU Clinical Trials Information System (CTIS), in compliance with evolving global transparency regulations and good practice. To support potential trial participants, we included clear and understandable trial descriptions in ClinicalTrials.gov registrations, written specifically for non-scientists. Similarly, for all protocols uploaded to the EU CTIS, we provided a lay protocol synopsis to help non-specialist audiences understand trial objectives and design. To ensure trial results were meaningfully returned to patients, all scientific result summaries submitted to the EU CTIS were accompanied by lay results summaries written in non-technical language. Visitors on ClinicalTrials.gov were also made aware of these lay results summaries via a link provided on the specific trial registration. | None | Genmab tracks the effectiveness of its transparency and patient engagement initiatives by evaluating feedback, monitoring communication channels, and incorporating learnings into future practices. Building on the Patient Advisory Council’s earlier contribution to the lay results summary template, the Council in 2025 reviewed three lay results summaries, providing valuable feedback that improved both content and tone. This collaboration deepened our understanding of critical aspects such as wording sensitivity and the right level of detail for non-scientific audiences. To further enhance clarity and consistency across all patient-facing materials, we developed and finalized an internal Plain Language Lexicon. The lexicon supports the use of health-literate terminology and a consistent tone across communications with patients, whether developed by disclosure, trial, or publication teams. We also hosted a health literacy workshop at Genmab’s Advisory Science Day, highlighting Genmab’s efforts to make complex scientific information more accessible and gathering valuable feedback to guide future initiatives. Requests and questions from consumers and end-users were received via Genmab’s publicly available mailbox, ClinicalTrials@genmab.com, and were triaged to ensure timely responses by the appropriate functions. Through these initiatives, Genmab continues to strengthen transparency, build trust, and ensure that clinical trial information is communicated responsibly and meaningfully to patients and the public. | Clinical Trial Disclosure & Transparency team, part of Development Operations that is overseen by the EVP, Chief Development Officer. |

 | Genmab 2025 Annual Report | 98 | |||||

Section | Disclosure requirement content | Disclosure requirement # | |
5.0 Business Conduct IRO Management including Actions, Metrics and Targets | Business conduct policies and corporate culture | G1-1 | |
Management of relationships with suppliers | G1-2 | ||
Prevention and detection of corruption and bribery | G1-3 | ||
Incidents of corruption or bribery | G1-4 | ||
Political influence and lobbying activities | G1-51 | ||
Payment practices | G1-6 |
Actual Positive Impact | Healthy and ethical corporate culture aligned with core values and purpose | |
Risk | Organizational health risk | |
Potential Negative Impact | Global data privacy | |
Potential Negative Impact | Protection of whistleblowers | |
Actual Negative Impact | Animal welfare | |
Potential Negative Impact | Management of suppliers |
 | Genmab 2025 Annual Report | 99 | |||||
 | Genmab 2025 Annual Report | 100 | |||||
 | Genmab 2025 Annual Report | 101 | |||||
IRO | Key Actions in 2025 | Targets | Outcomes / Tracking Effectiveness | Stakeholder Involvement |
Management of suppliers (potential negative impact) | Leadership transition: Responsibility for Supplier Code of Conduct implementation moved to a new Procurement VP in mid-2025. Process review: Conducted a review of the initial implementation phase to identify opportunities for a more structured and effective approach. Supplier identification: Identified over 130 suppliers requiring formal engagement on the Supplier Code of Conduct. Revised planning: Developed a phased implementation plan, targeting engagement with roughly half of suppliers in 2026 and the remainder in 2027. Monitoring and oversight: Planned regular progress tracking by the Global Procurement team. | Acceptance of Genmab’s Supplier Code of Conduct by 80% of suppliers by spend by 2025. The target supports key governance areas in the Supplier Code of Conduct, including legal compliance, anti-corruption, labor practices, human rights, supply chain, animal welfare, and information and IP security. | Target not achieved in 2025. The target was initially set for 2025; however, Genmab did not meet this target date due to a lack of systematic initiation and monitoring during the initial implementation phase. Responsibility has since moved to a new VP of Procurement (joined mid-2025), and a review has been conducted to establish a more structured approach to achieve this target. Target has been reset with a 2027 time horizon. | Key stakeholders include suppliers, the Global Procurement team, and the Legal and Compliance teams. |
 | Genmab 2025 Annual Report | 102 | |||||

Incidents of corruption or bribery | 2025 | 2024 |
Number of convictions for violation of anti-corruption and anti-bribery laws | 0 | 0 |
Amount of fines for violation of anti-corruption and anti-bribery laws (USD million) | 0 | 0 |

 | Genmab 2025 Annual Report | 103 | |||||
Disclosure Requirement | Data Point | SFDR Reference | Pillar 3 Reference | Benchmark Regulation Reference | EU Climate Law Reference | Material/ Not Material | Section, Paragraph or Page Reference |
ESRS 2 GOV-1 | 21 (d) | • | • | Material | GOV-1 Section | ||
ESRS 2 GOV-1 | 21 (e) | • | Material | GOV-1 Section | |||
ESRS 2 GOV-4 | 30 | • | Material | GOV-4 Section | |||
ESRS 2 SBM-1 | 40 (d) i | • | • | • | Not Material | ||
ESRS 2 SBM-1 | 40 (d) ii | • | • | Not Material | |||
ESRS 2 SBM-1 | 40 (d) iii | • | • | Not Material | |||
ESRS 2 SBM-1 | 40 (d) iv | • | Not Material | ||||
ESRS E1-1 | 14 | • | Material | ESRS E1-1 Section | |||
ESRS E1-1 | 16 (g) | • | • | Not Material | |||
ESRS E1-4 | 34 | • | • | • | Material | ESRS E1-4 Section | |
ESRS E1-5 | 38 | • | Not Material | ||||
ESRS E1-5 | 37 | • | Material | ESRS E1-5 Section | |||
ESRS E1-5 | 40-43 | • | Not Material | ||||
ESRS E1-6 | 44 | • | • | • | Material | ESRS E1-6 Section | |
ESRS E1-6 | 53-55 | • | • | • | Material | ESRS E1-6 Section | |
ESRS E1-7 | 56 | • | Not Material |
Disclosure Requirement | Data Point | SFDR Reference | Pillar 3 Reference | Benchmark Regulation Reference | EU Climate Law Reference | Material/ Not Material | Section, Paragraph or Page Reference |
ESRS E1-9 | 66 | • | Not Material | ||||
ESRS E1-9 | 66 (a); 66 (c) | • | Not Material | ||||
ESRS E1-9 | 67 (c) | • | Not Material | ||||
ESRS E1-9 | 69 | • | Not Material | ||||
ESRS E2-4 | 28 | • | Not Material | ||||
ESRS E3-1 | 9 | • | Not Material | ||||
ESRS E3-1 | 13 | • | Not Material | ||||
ESRS E3-1 | 14 | • | Not Material | ||||
ESRS E3-4 | 28 (c) | • | Not Material | ||||
ESRS E3-4 | 29 | • | Not Material | ||||
ESRS 2 - SBM 3 - E4 | 16 (a) i | • | Not Material | ||||
ESRS 2 - SBM 3 - E4 | 16 (b) | • | Not Material | ||||
ESRS 2 - SBM 3 - E4 | 16 (c) | • | Not Material | ||||
ESRS E4-2 | 24 (b) | • | Not Material | ||||
ESRS E4-2 | 24 (c) | • | Not Material | ||||
ESRS E4-2 | 24 (d) | • | Not Material |

 | Genmab 2025 Annual Report | 104 | |||||
Disclosure Requirement | Data Point | SFDR Reference | Pillar 3 Reference | Benchmark Regulation Reference | EU Climate Law Reference | Material/ Not Material | Section, Paragraph or Page Reference |
ESRS E5-5 | 37 (d) | • | Not Material | ||||
ESRS E5-5 | 39 | • | Not Material | ||||
ESRS 2- SBM3 - S1 | 14 (f) | • | Not Material | ||||
ESRS 2- SBM3 - S1 | 14 (g) | • | Not Material | ||||
ESRS S1-1 | 20 | • | Material | ESRS S1-1 Section | |||
ESRS S1-1 | 21 | • | Material | ESRS S1-1 Section | |||
ESRS S1-1 | 22 | • | Material | ESRS S1-1 Section | |||
ESRS S1-1 | 23 | • | Material | ESRS S1-1 Section | |||
ESRS S1-3 | 32 (c) | • | Material | ESRS S1-3 Section | |||
ESRS S1-14 | 88 (b); 88 (c) | • | • | Material | ESRS S1-14 Section | ||
ESRS S1-14 | 88 (e) | • | Material | ESRS S1-14 Section | |||
ESRS S1-16 | 97 (a) | • | • | Material | ESRS S1-16 Section | ||
ESRS S1-16 | 97 (b) | • | Material | ESRS S1-16 Section | |||
ESRS S1-17 | 103 (a) | • | Material | ESRS S1-17 Section | |||
ESRS S1-17 | 104 (a) | • | • | Not Material | |||
ESRS 2- SBM3 – S2 | 11 (b) | • | Not Material | ||||
ESRS S2-1 | 17 | • | Not Material |
Disclosure Requirement | Data Point | SFDR Reference | Pillar 3 Reference | Benchmark Regulation Reference | EU Climate Law Reference | Material/ Not Material | Section, Paragraph or Page Reference |
ESRS S2-1 | 18 | • | Not Material | ||||
ESRS S2-1 | 19 | • | • | Not Material | |||
ESRS S2-1 | 19 | • | Not Material | ||||
ESRS S2-4 | 36 | • | Not Material | ||||
ESRS S3-1 | 16 | • | Not Material | ||||
ESRS S3-1 | 17 | • | • | Not Material | |||
ESRS S3-4 | 36 | • | Not Material | ||||
ESRS S4-1 | 16 | • | Material | ESRS S4-1 Section | |||
ESRS S4-1 | 17 | • | • | Not Material | |||
ESRS S4-4 | 35 | • | Not Material | ||||
ESRS G1-1 | 10 (b) | • | Not Material | ||||
ESRS G1-1 | 10 (d) | • | Material | ESRS G1-1 Section | |||
ESRS G1-4 | 24 (a) | • | • | Material | ESRS G1-4 Section | ||
ESRS G1-4 | 24 (b) | • | Not Material |

Genmab 2025 Annual Report | 105 | ||||||

Genmab 2025 Annual Report | 106 | ||||||
Intangible Assets and Goodwill | ||
Acquisitions | ||
 | Genmab 2025 Annual Report | 107 | |||||
Note | 2025 | 2024¹ Restated | 2023¹ Restated | |
Revenue | 2.1, 2.2 | |||
Cost of product sales | 2.3 | ( | ( | ( |
Research and development expenses | 2.3, 3.1, 3.2 | ( | ( | ( |
Selling, general and administrative expenses | 2.3, 3.2 | ( | ( | ( |
Acquisition and integration related charges | 5.5 | ( | ( | |
Total costs and operating expenses | ( | ( | ( | |
Operating profit | ||||
Financial income | 4.5 | |||
Financial expenses | 4.5 | ( | ( | ( |
Net profit before tax | ||||
Corporate tax | 2.4 | ( | ( | ( |
Net profit | ||||
Other comprehensive income: | ||||
Amounts which may be re-classified to the income statement: | ||||
Exchange differences on translation of foreign operations | ( | |||
Total comprehensive income | ||||
Basic net profit per share | 2.5 | |||
Diluted net profit per share | 2.5 |
 | Genmab 2025 Annual Report | 108 | |||||
December 31, | January 1, | |||
Note | 2025 | 2024¹ Restated | 2024¹ Restated | |
Assets | ||||
Goodwill | 3.1, 5.5 | |||
Other intangible assets | 3.1, 5.5 | |||
Property and equipment | 2.2, 3.2 | |||
Right-of-use assets | 2.2, 3.3 | |||
Receivables | 2.2, 3.6 | |||
Deferred tax assets | 2.4 | |||
Other investments | 3.4 | |||
Total non-current assets | ||||
Corporate tax receivable | 2.4 | |||
Inventories | 3.5 | |||
Receivables | 3.6 | |||
Marketable securities | 4.2, 4.4 | |||
Cash and cash equivalents | ||||
Total current assets | ||||
Total assets | ||||
December 31, | January 1, | |||
Note | 2025 | 2024¹ Restated | 2024¹ Restated | |
Shareholders' Equity And Liabilities | ||||
Share capital | 4.7 | |||
Share premium | 4.7 | |||
Other reserves | ( | ( | ( | |
Retained earnings | ||||
Total shareholders' equity | ||||
Borrowings | 4.8 | |||
Lease liabilities | 3.3 | |||
Contract Liabilities | 3.7 | |||
Deferred tax liabilities | 2.4 | |||
Other payables | 3.8 | |||
Total non-current liabilities | ||||
Borrowings | 4.8 | |||
Corporate tax payable | 2.4 | |||
Lease liabilities | 3.3 | |||
Contract liabilities | 3.7 | |||
Other payables | 3.8 | |||
Total current liabilities | ||||
Total liabilities | ||||
Total shareholders' equity and liabilities | ||||
 | Genmab 2025 Annual Report | 109 | |||||
Note | 2025 | 2024¹ Restated | 2023¹ Restated | |
Cash flows from operating activities: | ||||
Net profit before tax | ||||
— | — | — | ||
Financial income | 4.5 | ( | ( | ( |
Financial expense | 4.5 | |||
Adjustment for non-cash transactions | ||||
Share-based compensation expense | 2.3, 4.6 | |||
Depreciation | 3.2, 3.3 | |||
Amortization | 3.1 | |||
Impairment losses | 3.1, 3.6 | |||
Change in operating assets and liabilities | ||||
Receivables | 3.6 | ( | ( | |
Inventories | 3.5 | ( | ( | ( |
Other payables | 3.8 | |||
Cash flows from operating activities before financial items | ||||
Interest received | ||||
Interest elements of lease payments | 3.3 | ( | ( | ( |
Interest paid | ( | |||
Corporate taxes paid | ( | ( | ( | |
Net cash provided by operating activities | ||||
Cash flows from investing activities: | ||||
Acquisition of business, net of cash acquired | 5.5 | ( | ||
Acquisition of assets, net of cash acquired | 5.5 | ( | ||
Investment in intangible assets | 3.1 | ( | ( | ( |
Investment in tangible assets | 3.2 | ( | ( | ( |
Marketable securities bought | 4.3, 4.4 | ( | ( | ( |
Marketable securities sold | 4.3, 4.4 | |||
Other investments bought | 3.4 | ( | ( | ( |
Net cash (used in) investing activities | ( | ( | ( | |
Note | 2025 | 2024¹ Restated | 2023¹ Restated | |
Cash flows from financing activities: | ||||
Warrants exercised | 4.6, 4.7 | |||
Principal elements of lease payments | 3.3 | ( | ( | ( |
Purchase of treasury shares | 4.7 | ( | ( | ( |
Payment of withholding taxes on behalf of employees on net settled RSUs | ( | ( | ( | |
Proceeds from issuance of borrowings | 4.8 | |||
Debt issuance costs paid | 4.8 | ( | ||
Net cash provided by (used in) financing activities | ( | ( | ||
Changes in cash and cash equivalents | ( | |||
Cash and cash equivalents at the beginning of the period | ||||
Exchange rate adjustments | ( | |||
Cash and cash equivalents at the end of the period | ||||
Cash and cash equivalents include: | ||||
Bank deposits | ||||
Short-term marketable securities | ||||
Cash and cash equivalents at the end of the period |
 | Genmab 2025 Annual Report | 110 | |||||
Share capital | Share premium | Translation reserves | Retained earnings | Shareholders' equity | |
Balance at December 31, 2022¹ Restated | ( | ||||
Net profit | — | — | — | ||
Other comprehensive income | — | — | — | ||
Total comprehensive income | — | — | |||
Transactions with owners: | |||||
Exercise of warrants | — | — | — | ||
Purchase of treasury shares | — | — | — | ( | ( |
Share-based compensation expenses | — | — | — | ||
Withholding taxes on behalf of employees on net settled RSUs | — | — | — | ( | ( |
Tax on items recognized directly in equity | — | — | — | ( | ( |
Balance at December 31, 2023¹ Restated | ( | ||||
Net profit | — | — | — | ||
Other comprehensive income | — | — | ( | — | ( |
Total comprehensive income | — | — | ( | ||
Transactions with owners: | |||||
Exercise of warrants | — | — | — | ||
Purchase of treasury shares | — | — | — | ( | ( |
Share-based compensation expenses | — | — | — | ||
Withholding taxes on behalf of employees on net settled RSUs | — | — | — | ( | ( |
Tax on items recognized directly in equity | — | — | — | ( | ( |
Share capital | Share premium | Translation reserves | Retained earnings | Shareholders' equity | |
Balance at December 31, 2024¹ Restated | ( | ||||
Net profit | — | — | — | ||
Other comprehensive income | — | — | — | ||
Total comprehensive income | — | — | |||
Transactions with owners: | |||||
Exercise of warrants | — | — | — | ||
Purchase of treasury shares | — | — | — | ( | ( |
Share-based compensation expenses | — | — | — | ||
Withholding taxes on behalf of employees on net settled RSUs | — | — | — | ( | ( |
Share Reduction | — | ( | — | ||
Tax on items recognized directly in equity | — | — | — | ||
Balance at December 31, 2025 | ( |

 | Genmab 2025 Annual Report | 111 | |||||
Section 2 - Result for the Year |
2.1 - Revenue |
2.2 - Information about Geographical Areas |
2.3 - Staff Costs |
2.4 - Corporate and Deferred Tax |
2.5 - Profit Per Share |
Section 3 - Operating Assets and Liabilities |
3.1 - Other Intangible Assets and Goodwill |
3.2 - Property and Equipment |
3.3 - Leases |
3.4 - Other Investments |
3.5 - Inventories |
3.6 - Receivables |
3.7 - Contract Liabilities |
3.8 - Other Payables |
Section 4 - Capital Structure, Financial Risk and Related Items |
4.3 - Financial Assets and Liabilities |
4.4 - Marketable Securities |
4.5 - Financial Income and Expenses |
4.6 - Share-Based Instruments |
4.8 - Borrowings |
Section 5 - Other Disclosures |
5.5 - Acquisitions |
 | Genmab 2025 Annual Report | 112 | |||||
Name | Domicile | Ownership and votes 2025 | Ownership and votes 2024 |
Genmab B.V. | Utrecht, the Netherlands | ||
Genmab Holding B.V. | Utrecht, the Netherlands | ||
Genmab Holding II B.V. | Utrecht, the Netherlands | N/A² | |
Genmab US, Inc. | Delaware, USA | ||
Genmab K.K. | Tokyo, Japan | ||
Genmab Germany GmbH | Munich, Germany | N/A² | |
Genmab UK Ltd | London, United Kingdom | N/A² | |
Genmab France SAS | Paris, France | N/A² | |
Genmab Finance LLC. | Delaware, USA | N/A² | |
ProfoundBio Inc. | Delaware, USA | ||
ProfoundBio US Co. | Delaware, USA | ||
ProfoundBio Limited | Hong Kong, China | ||
Genmab (Suzhou) Co., Ltd. | Suzhou, China | ||
Genmab (Beijing) Co., Ltd. | Beijing, China | ||
Merus B.V. | Utrecht, the Netherlands | N/A¹ | |
Merus US Inc. | Massachusetts, USA | N/A¹ |
 | Genmab 2025 Annual Report | 113 | |||||
2020 | 2021 | 2022 | 2023 | |
Average rate | NA | |||
Closing rate | N/A |
Q1 2024 | Q2 2024 | Q3 2024 | Q4 2024 | 2024 | |
Average rate | |||||
Closing rate |
Previously reported in DKK | Reported in USD | ||
December 31, 2024 | Presentation currency change | December 31, 2024 | |
Total assets | ( | ||
Total liabilities | ( | ||
Total shareholders' equity | ( |
Previously reported in DKK | Reported in USD | ||
January 1, 2024 | Presentation currency change | January 1, 2024 | |
Total assets | ( | ||
Total liabilities | ( | ||
Total shareholders' equity | ( |
Previously reported in DKK | Reported in USD | ||
December 31, 2024 | Presentation currency change | December 31, 2024 | |
Net profit | ( | ||
Comprehensive income | ( |
Previously reported in DKK | Reported in USD | ||
December 31, 2023 | Presentation currency change | December 31, 2023 | |
Net profit | ( | ||
Comprehensive income | ( |
Previously reported in DKK | Reported in USD | ||
Cash provided by (used in): | December 31, 2024 | Presentation currency change | December 31, 2024 |
Operating activities | ( | ||
Investing activities | ( | ( | |
Financing activities | ( | ( |
Previously reported in DKK | Reported in USD | ||
Cash provided by (used in): | December 31, 2023 | Presentation currency change | December 31, 2023 |
Operating activities | ( | ||
Investing activities | ( | ( | |
Financing activities | ( | ( |
 | Genmab 2025 Annual Report | 114 | |||||
Previously reported in DKK | Reported in USD | ||
December 31, 2024 | Presentation currency change | December 31, 2024 | |
Earnings per share - basic | ( | ||
Earnings per share - diluted | ( |
Previously reported in DKK | Reported in USD | ||
December 31, 2023 | Presentation currency change | December 31, 2023 | |
Earnings per share - basic | ( | ||
Earnings per share - diluted | ( |
 | Genmab 2025 Annual Report | 115 | |||||
 | Genmab 2025 Annual Report | 116 | |||||

 | Genmab 2025 Annual Report | 117 | |||||
Accounting policy | Key accounting estimates and judgements | Note reference | Risk |
Fair value and impairment assessment of other intangible assets and goodwill | Estimation of the fair value of other intangible assets acquired through acquisitions Subsequent assessment of impairment of other intangible assets Estimation regarding the valuation of goodwill and assessment of impairment of goodwill | Notes 3.1 and 5.5 | High |
Revenue recognition | Judgement in assessing whether a collaboration partner is a customer Estimation of partner net sales amounts in the calculation of royalties Estimation of variable consideration Judgement in assessing the nature of combined performance obligations within contracts | Note 2.1 | High |
Share-based compensation | Judgement in selecting assumptions required for valuation of warrant grants Estimation in developing forfeiture rate RSUs/warrants and probability of achievement for PSUs | Note 4.6 | Moderate |
Current and deferred income taxes | Judgement and estimation regarding valuation of deferred income tax assets | Note 2.4 | Moderate |
December 31, 2024 | Reclass | December 31, 2024 | |
Financial income: | |||
Gain on marketable securities | |||
Foreign exchange rate gain | ( | ||
Gain on other investments, net | ( | ||
Total financial income | ( | ||
Financial expenses: | |||
Loss on marketable securities | ( | ( | ( |
Foreign exchange rate loss | ( | ( | |
Loss on other investments, net | ( | ||
Total financial expenses | ( | ( | |
Net financial items |
December 31, 2023 | Reclass | December 31, 2023 | |
Financial income: | |||
Gain on marketable securities | |||
Foreign exchange rate gain | ( | ||
Gain on other investments, net | ( | ||
Total financial income | |||
Financial expenses: | |||
Loss on marketable securities | ( | ( | ( |
Foreign exchange rate loss | ( | ( | |
Loss on other investments, net | ( | ( | |
Total financial expenses | ( | ( | ( |
Net financial items | ( | ( |

 | Genmab 2025 Annual Report | 118 | |||||
2025 | 2024 | 2023 | |
Revenue by type: | |||
Royalties | |||
Net product Sales | |||
Reimbursement revenue | |||
Milestone revenue | |||
Collaboration revenue | |||
Total | |||
Revenue by collaboration partner: | |||
Janssen | |||
AbbVie | |||
Roche | |||
Novartis | |||
BioNTech | |||
Pfizer1 | |||
Other | |||
Total2 | |||
Royalties by product: | |||
DARZALEX | |||
Kesimpta | |||
TEPEZZA | |||
Other3 | |||
Total |
 | Accounting Policies |
 | Genmab 2025 Annual Report | 119 | |||||
Revenue | Non-current assets | Revenue | Non-current assets | Revenue | Non-current assets | |
2025 | 2024 | 2023 | ||||
Denmark | ||||||
Netherlands | ||||||
United States | ||||||
Japan | ||||||
China | ||||||
Total | ||||||
 | Accounting Policies |
 | Genmab 2025 Annual Report | 120 | |||||
2025 | 2024 | 2023 | |
Wages and salaries | |||
Share-based compensation | |||
Defined contribution plans | |||
Other social security costs | |||
Government grants related to research and development expenses | ( | ( | ( |
Total | |||
Staff costs are included in the Consolidated Statements of Comprehensive Income as follows: | |||
Cost of product sales | |||
Research and development expenses | |||
Selling, general and administrative expenses | |||
Total | |||
Average number of FTE | |||
Number of FTE at year-end |
 | Accounting Policies |
2025 | 2024 | 2023 | |
Current tax on profit | |||
Adjustment to deferred tax | ( | ( | |
Net increase (decrease) of unrecognized deferred tax assets for the year | ( | ||
Effect of exchange rate adjustment | ( | ||
Total tax for the period in the income statement |
2025 | 2024 | 2023 | |
Net profit before tax | |||
Tax at the Danish corporation tax rate of | |||
Tax effect of: | |||
Net increase (decrease) of unrecognized deferred tax assets for the year | ( | ||
Net of non-taxable income over non- deductible expenses | ( | ||
Other current and deferred tax adjustments | ( | ( | ( |
Effect of exchange rate adjustment | ( | ||
Total tax effect | ( | ( | |
Total tax for the period in the income statement | |||
Total tax for the period in shareholders' equity | ( | ||
Effective Tax Rate |
 | Genmab 2025 Annual Report | 121 | |||||
December 31, | December 31, | January 1, | |
2025 | 2024 | 2024 | |
Share-based instruments | |||
Deferred revenue | |||
Intangible assets | ( | ( | ( |
Liabilities | |||
Tax losses and credits carried forward | |||
Other temporary differences | |||
Total | ( | ( |
Share- based instruments | Deferred revenue | Intangible assets | Liabilities | Tax losses carried forward | Other temporary differences | Total | |
2025 | |||||||
Net deferred tax asset/(liability) at the beginning of the year | ( | ( | |||||
Recognised in Profit or Loss | ( | ( | ( | ||||
Acquired in Business Combinations | |||||||
Net deferred tax asset/(liability) at the end of the year | ( | ( | |||||
2024 | |||||||
Net deferred tax asset/(liability) at the beginning of the year | ( | ||||||
Recognised in Profit or Loss | ( | ||||||
Acquired in Business Combinations | ( | ( | |||||
Net deferred tax asset/(liability) at the end of the year | ( | ( |
 | Genmab 2025 Annual Report | 122 | |||||
 | Accounting Policies |
 | Management’s Judgements and Estimates |
2025 | 2024 | 2023 | |
Net profit | |||
(Shares) | |||
Weighted average number of shares outstanding | |||
Weighted average number of treasury shares | ( | ( | ( |
Weighted average number of shares excl. treasury shares | |||
Adjustments for share-based instruments, dilution | |||
Weighted average number of shares, diluted | |||
Basic net profit per share | |||
Diluted net profit per share |
 | Accounting Policies |

 | Genmab 2025 Annual Report | 123 | |||||
Goodwill | Licenses and Patents | Technology Platform | Acquired IPR&D | Total Intangible Assets | |
2025 | |||||
Cost at the beginning of the year | |||||
Additions for the year | |||||
Effect of exchange rate adjustment | |||||
Cost at the end of the year | |||||
Amortization and impairment losses at the beginning of the year | |||||
Amortization for the year | — | ||||
Impairment losses for the year | |||||
Effect of exchange rate adjustment | |||||
Amortization and impairment losses at the end of the year | |||||
Carrying amount at the end of the year | |||||
2024 | |||||
Cost at the beginning of the year | |||||
Additions for the year | |||||
Effect of exchange rate adjustment | ( | ( | ( | ||
Cost at the end of the year | |||||
Amortization and impairment losses at the beginning of the year | |||||
Amortization for the year | — | ||||
Impairment losses for the year | |||||
Effect of exchange rate adjustment | ( | ( | ( | ||
Amortization and impairment losses at the end of the year | |||||
Carrying amount at the end of the year |
 | Genmab 2025 Annual Report | 124 | |||||
 | Accounting Policies |
 | Genmab 2025 Annual Report | 125 | |||||
 | Management’s Judgements and Estimates |
Leasehold improvements | Equipment, furniture and fixtures | Assets under construction | Total property and equipment | |
2025 | ||||
Cost at January 1 | ||||
Additions for the year | ||||
Transfers between the classes | ( | |||
Disposals for the year | ( | ( | ( | |
Exchange rate adjustment | ||||
Cost at December 31 | ||||
Accumulated depreciation and impairment at January 1 | ( | ( | ( | |
Depreciation for the year | ( | ( | ( | |
Exchange rate adjustment | ( | ( | ( | |
Accumulated depreciation on disposals | ||||
Accumulated depreciation and impairment at December 31 | ( | ( | ( | |
Carrying amount at December 31 |
 | Genmab 2025 Annual Report | 126 | |||||
Leasehold improvements | Equipment, furniture and fixtures | Assets under construction | Total property and equipment | |
2024 | ||||
Cost at January 1 | ||||
Additions for the year | ||||
Acquisitions through business combinations | ||||
Transfers between the classes | ( | |||
Disposals for the year | ( | ( | ( | ( |
Exchange rate adjustment | ( | ( | ( | ( |
Cost at December 31 | ||||
Accumulated depreciation and impairment at January 1 | ( | ( | ( | |
Depreciation for the year | ( | ( | ( | |
Exchange rate adjustment | ||||
Accumulated depreciation on disposals | ||||
Accumulated depreciation and impairment at December 31 | ( | ( | ( | |
Carrying amount at December 31 |
2025 | 2024 | 2023 | |
Depreciation and impairment included in the income statement as follows: | |||
Research and development expenses | |||
Selling, general and administrative expenses | |||
Total |
 | Accounting Policies |
Equipment, furniture and fixtures | |
Leasehold improvements |
 | Genmab 2025 Annual Report | 127 | |||||
2025 | 2024 | 2023 | |
Right-of-use assets | |||
Balance at January 1 | |||
Additions to right-of-use assets1 | |||
Depreciation charge for the year | ( | ( | ( |
Exchange rate adjustment | ( | ||
Balance at December 31 | |||
Lease liabilities | |||
Current | |||
Non-current | |||
Total at December 31 | |||
Cash outflow for lease payments | |||
1.Additions to right-of-use assets in 2025 related to the Merus acquisition, modifications to existing leases, lease incentives, and adjustments to the provisions for contractual restoration obligations related to leases of Genmab offices. | |||
December 31, | December 31, | December 31, | |
2025 | 2024 | 2023 | |
Payment due | |||
Within 1 year | |||
1 – 3 years | |||
3 – 5 years | |||
More than 5 years | |||
Total |
 | Accounting Policies |
 | Genmab 2025 Annual Report | 128 | |||||
December 31, | December 31, | January 1, | |
2025 | 2024 | 2024 | |
Publicly traded equity securities | |||
Fund investments | |||
Privately held equity securities | |||
Total |
 | Accounting Policies |
December 31, | December 31, | January 1, | |
2025 | 2024 | 2024 | |
Raw materials | |||
Work in progress | |||
Finished goods | |||
Total inventories (gross) | |||
Allowances at year end | ( | ( | ( |
Total inventories (net) |
 | Accounting Policies |
 | Genmab 2025 Annual Report | 129 | |||||
December 31, | December 31, | January 1, | |
2025 | 2024 | 2024 | |
Receivables related to collaboration agreements | |||
Prepayments | |||
Trade receivables related to product sales | |||
Interest receivables | |||
Other receivables | |||
Total | |||
Non-current receivables | |||
Current receivables | |||
Total |
 | Accounting Policies |
December 31, | December 31, | January 1, | |
2025 | 2024 | 2024 | |
Contract liabilities at January 1 | |||
Consideration received | |||
Additions from asset acquisition | |||
Revenue recognized during the year | ( | ( | |
Exchange rate adjustment | ( | ||
Total | |||
Non-current contract liabilities | |||
Current contract liabilities | |||
Total |
 | Genmab 2025 Annual Report | 130 | |||||
December 31, | December 31, | January 1, | |
2025 | 2024 | 2024 | |
Liabilities related to collaboration agreements | |||
Staff cost liabilities | |||
Accounts payable | |||
Other liabilities | |||
Total | |||
Non-current other payables | |||
Current other payables | |||
Total |
 | Accounting Policies |

 | Genmab 2025 Annual Report | 131 | |||||
⬤ | credit risk; |
⬤ | foreign currency risk; and |
⬤ | interest rate risk |
 | Genmab 2025 Annual Report | 132 | |||||
Category | S&P | Moody’s | Fitch |
Short-term | A-2 | P-2 | F-2 |
Long-term | BBB- | Baa3 | BBB- |
 | Genmab 2025 Annual Report | 133 | |||||
2025 | 2024 | |||
Percent | ||||
USD | % | % | ||
DKK | % | % | ||
EUR | % | % | ||
GBP | % | % | ||
Total at December 31 | % | % | ||
2025 | 2024 | |
Year of Maturity | ||
2025 | ||
2026 | ||
2027 | ||
2028 | ||
2029 | ||
2030+ | ||
Total at December 31 |
 | Genmab 2025 Annual Report | 134 | |||||
December 31, | |||
Note | 2025 | 2024 | |
Financial assets measured at fair value through profit or loss | |||
Marketable securities | 4.4 | ||
Other investments | 3.4 | ||
Financial assets measured at amortized cost | |||
Receivables excluding prepayments | 3.6 | ||
Cash and cash equivalents | |||
Financial liabilities measured at amortized cost | |||
Borrowings | 4.8 | ( | |
Lease liabilities | 3.3 | ( | ( |
Other payables excluding provisions | 3.8 | ( | ( |
December 31, | |||||||||
2025 | 2024 | ||||||||
Note | Level 1 | Level 2 | Level 3 | Total | Level 1 | Level 2 | Level 3 | Total | |
Assets Measured at Fair Value | |||||||||
Marketable securities | 4.4 | — | — | — | — | — | |||
Other investments | 3.4 | ||||||||
Other Investments | |
Fair value at December 31, 2023 | |
Acquisitions | |
Fair value changes | |
Fair value at December 31, 2024 | |
Acquisitions | |
Fair value changes | ( |
Fair value at December 31, 2025 |
 | Accounting Policies |
 | Genmab 2025 Annual Report | 135 | |||||
•Level 1 - Quoted prices (unadjusted) in active markets for identical assets or liabilities |
•Level 2 - Inputs other than quoted prices included within Level 1 that are observable for the asset or liability, either directly (that is, as prices) or indirectly (that is, derived from prices) |
•Level 3 - Inputs for the asset or liability that are not based on observable market data (that is, unobservable inputs). |
Market value | Share | Market value | Share | Market value | Share | |
December 31, 2025 | % | December 31, 2024 | % | January 1, 2024 | % | |
USD portfolio | ||||||
Corporate bonds | ||||||
US government bonds and treasury bills | ||||||
Commercial paper | ||||||
Other | ||||||
Total USD portfolio | ||||||
DKK portfolio | ||||||
Kingdom of Denmark bonds and treasury bills | ||||||
Danish mortgage- backed securities | ||||||
Total DKK portfolio | ||||||
EUR portfolio | ||||||
European government bonds and treasury bills | ||||||
GBP portfolio | ||||||
UK government bonds and treasury bills | ||||||
Total portfolio | ||||||
Marketable securities |
 | Genmab 2025 Annual Report | 136 | |||||
 | Accounting Policies |
2025 | 2024¹ | 2023¹ | |
Financial income: | |||
Interest and other financial income | |||
Gain on marketable securities | |||
Gain on other investments, net | |||
Foreign exchange rate gain | |||
Total financial income | |||
Financial expenses: | |||
Other interest expense | ( | ( | ( |
Interest expense on borrowings | ( | ||
Loss on marketable securities | ( | ( | ( |
Loss on other investments, net | ( | ( | |
Foreign exchange rate loss | ( | ( | ( |
Total financial expenses | ( | ( | ( |
Net financial items |
 | Genmab 2025 Annual Report | 137 | |||||
 | Accounting Policies |
 | Genmab 2025 Annual Report | 138 | |||||
RSUs Granted in Periods | ||
Key Terms | December 2019 - February 2021 | From February 2021 |
Grants | RSUs are granted at no cost to employees. Number of shares granted is determined based on closing share price on the grant date. | |
Vesting (Settlement) | Cliff vesting – RSUs become fully vested on the first banking day of the month following a period of while also subject to the degree of fulfilment of the applicable performance criteria. After RSUs vest, the holder receives tax authority on behalf of the employee, Genmab withholds the number of RSUs that are equal to the monetary value of the employee’s tax obligation from the total number of RSUs that otherwise would have been issued to the employee upon vesting (“net settlement”). Genmab A/S may at its sole discretion in extraordinary circumstances choose to make a cash settlement instead of delivering shares. | |
Leaver | Leavers – Forfeit all unvested RSUs except when due to retirement, death, serious sickness, or serious injury, in which case granted but not yet vested RSUs shall remain outstanding and will be settled in accordance with their terms. Notwithstanding the above, the December 2020 RSU grant to members of the Board was made subject to pro-rata vesting upon termination of board services. Employees and Executive Management – RSUs remain outstanding and vest accordingly when the employment relationship is terminated by Genmab without cause. | Good-Leavers2 – May maintain a pro-rata portion of unvested RSUs. Bad-Leavers3 – Forfeit all unvested RSUs. Death – Forfeit all unvested RSUs. Voluntary leavers forfeit unvested RSUs. |
 | Genmab 2025 Annual Report | 139 | |||||
Number of RSUs held by the Board of Directors | Number of RSUs held by the Executive Management | Number of RSUs held by employees | Number of RSUs held by former members of the Executive Management, Board of Directors and employees | Total RSUs | Weighted Average Fair Value - RSUs Granted - DKK | Total Fair Value of RSUs Granted - DKK million | |
Outstanding at January 1, 2023 | |||||||
Granted¹ | |||||||
Settled | ( | ( | ( | ( | ( | ||
Transferred | — | ( | — | ||||
Forfeited | — | ( | ( | ( | ( | ||
Outstanding at December 31, 2023 | |||||||
Outstanding at January 1, 2024 | |||||||
Granted¹ | |||||||
Settled | ( | ( | ( | ( | ( | ||
Transferred | — | ( | ( | — | |||
Forfeited | — | ( | ( | ( | ( | ||
Outstanding at December 31, 2024 | |||||||
Outstanding at January 1, 2025 | |||||||
Granted¹ | |||||||
Settled | ( | ( | ( | ( | ( | ||
Transferred | ( | ( | — | ||||
Forfeited | ( | ( | ( | ( | ( | ||
Outstanding at December 31, 2025 |
 | Genmab 2025 Annual Report | 140 | |||||
Warrants Granted in Periods | |||
Key Terms | March 2017 - February 2021 | From February 2021 | |
Grants | Warrants are granted at no cost to employees. Granted at an exercise price equal to the closing share price on the grant date. | ||
Vesting (Exercisable) | Cliff vesting over | ||
Leaver | Leavers - Forfeit all unvested warrants; however, will be able to exercise pro-rata portion of warrants on a regular schedule in instances where the employment relationship is terminated by Genmab without cause. | Good-Leavers - May maintain a pro-rata portion of unvested warrants. Bad-Leavers - Forfeit all unvested warrants. Death - Forfeit all unvested warrants. Voluntary leavers forfeit all unvested warrants. | |
Lapse | 7th anniversary of grant date | ||
 | Genmab 2025 Annual Report | 141 | |||||
Number of warrants held by the Board of Directors | Number of warrants held by the Executive Management | Number of warrants held by employees | Number of warrants held by former members of the Executive Management, Board of Directors and employees | Total warrants | Weighted average exercise price - DKK | Weighted average share price at exercise date - DKK | Outstanding Warrants - % of Share Capital | |
Outstanding at January 1, 2023 | ||||||||
Granted¹ | — | |||||||
Exercised | — | ( | ( | ( | ( | |||
Expired | — | — | ( | ( | ( | |||
Forfeited | — | — | ( | ( | ( | |||
Transfers | — | ( | — | — | ||||
Outstanding at December 31, 2023 | ||||||||
Exercisable at year end | ||||||||
Exercisable warrants in the money at year end | ||||||||
Outstanding at January 1, 2024 | ||||||||
Granted¹ | — | |||||||
Exercised | — | ( | ( | ( | ( | |||
Expired | — | — | ( | ( | ( | |||
Forfeited | — | — | ( | ( | ( | |||
Transfers | — | ( | — | — | ||||
Outstanding at December 31, 2024 | ||||||||
Exercisable at year end | ||||||||
Exercisable warrants in the money at year end | — | |||||||
Outstanding at January 1, 2025 | ||||||||
Granted¹ | — | |||||||
Exercised | — | ( | ( | ( | ( | |||
Expired | — | — | ( | ( | ( | |||
Forfeited | — | — | ( | ( | ( | |||
Transfers | ( | ( | — | — | ||||
Outstanding at December 31, 2025 | ||||||||
Exercisable at year end | ||||||||
Exercisable warrants in the money at year end |
 | Genmab 2025 Annual Report | 142 | |||||
 | Accounting Policies |
 | Management’s Judgements and Estimates |
Weighted average | 2025 | 2024 | 2023 |
Fair value per warrant on grant date | |||
Share price | |||
Exercise price | |||
Expected dividend yield | |||
Expected stock price volatility | |||
Risk-free interest rate | |||
Expected life of warrants |
Total Fair Value of Amounts Granted | 2025 | 2024 | 2023 |
Total fair value of warrants granted | DKK | DKK | DKK |
 | Genmab 2025 Annual Report | 143 | |||||
March 12, 2025 | March 13, 2024 | April 13, 2021 | |
Authorization | Authorization | Authorization | |
Warrants issued | |||
Warrants reissued | |||
Warrants available for issue | |||
Warrants available for reissue |
Number of shares | Share capital | Share capital | Share Price Ranges1 | |
(DKK million) | (USD million) | |||
December 31, 2022 | ||||
Exercise of warrants | DKK | |||
December 31, 2023 | ||||
Exercise of warrants | DKK | |||
December 31, 2024 | ||||
Exercise of warrants | DKK | |||
Share capital reduction | ( | ( | ||
December 31, 2025 |
Number of shares | Share capital (USD million) | Proportion of share capital % | Cost (USD million) | |
Shareholding at December 31, 2022 | ||||
Purchase of treasury shares | ||||
Shares used for funding RSU program | ( | ( | ( | |
Shareholding at December 31, 2023 | ||||
Purchase of treasury shares | ||||
Shares used for funding RSU program | ( | ( | ( | |
Shareholding at December 31, 2024 | ||||
Purchase of treasury shares | ||||
Shares used for funding RSU program | ( | ( | ( | |
Reduction in share capital | ( | ( | ( | ( |
Shareholding at December 31, 2025 |
 | Genmab 2025 Annual Report | 144 | |||||
Current | Non-Current | ||||||
December 31, | December 31, | January 1, | December 31, | December 31, | January 1, | ||
2025 | 2024 | 2024 | 2025 | 2024 | 2024 | ||
Term Loan A (Secured) | |||||||
Term Loan B (Secured) | |||||||
Secured Notes | |||||||
Unsecured Notes | |||||||
Total Borrowings | |||||||
Interest | Maturity Date | Nominal Value | |
Term Loan A (Secured) | December 2030 | ||
Term Loan B (Secured) | December 2032 | ||
Secured Notes | December 2032 | ||
Unsecured Notes | December 2033 | ||
Total |
Term Loan A (Secured) | Term Loan B (Secured) | Secured Notes | Unsecured Notes | Total | |
Payments due | |||||
Within 1 year | |||||
1 – 3 years | |||||
3 – 5 years | |||||
More than 5 years | |||||
Total |
 | Genmab 2025 Annual Report | 145 | |||||
 | Accounting Policies |

 | Genmab 2025 Annual Report | 146 | |||||
2025 | 2024 | 2023 | |
Wages and salaries | |||
Share-based compensation expenses | |||
Defined contribution plans | |||
Total |
 | Genmab 2025 Annual Report | 147 | |||||
Base Board Fee | Committee Fees | Share-Based Compensation Expenses | Total | ||||||||||||
2025 | 2024 | 2023 | 2025 | 2024 | 2023 | 2025 | 2024 | 2023 | 2025 | 2024 | 2023 | ||||
Deirdre P. Connelly | |||||||||||||||
Pernille Erenbjerg | |||||||||||||||
Anders Gersel Pedersen | |||||||||||||||
Paolo Paoletti | |||||||||||||||
Rolf Hoffmann | |||||||||||||||
Elizabeth O’Farrell | |||||||||||||||
Mijke Zachariasse1 | |||||||||||||||
Martin Schultz1 | |||||||||||||||
Takahiro Hamatani2 | |||||||||||||||
Michael Kavanagh1 | |||||||||||||||
Total | |||||||||||||||
 | Genmab 2025 Annual Report | 148 | |||||
Base Salary | Defined Contribution Plans | Other Benefits | Annual Cash Bonus | Share-Based Compensation Expenses | Total | ||||||||||||||||||
2025 | 2024 | 2023 | 2025 | 2024 | 2023 | 2025 | 2024 | 2023 | 2025 | 2024 | 2023 | 2025 | 2024 | 2023 | 2025 | 2024 | 2023 | ||||||
Jan van de Winkel | |||||||||||||||||||||||
Anthony Pagano | |||||||||||||||||||||||
Anthony Mancini3 | |||||||||||||||||||||||
Judith Klimovsky | |||||||||||||||||||||||
Tahamtan Ahmadi | |||||||||||||||||||||||
Birgitte Stephensen1 | |||||||||||||||||||||||
Christopher Cozic1 | |||||||||||||||||||||||
Martine van Vugt2 | |||||||||||||||||||||||
Brad Bailey4 | |||||||||||||||||||||||
Rayne Waller4 | |||||||||||||||||||||||
Greg Mueller | |||||||||||||||||||||||
Total | |||||||||||||||||||||||
 | Genmab 2025 Annual Report | 149 | |||||
2025 | 2024 | 2023 | |
Audit fees | |||
Audit-related fees | |||
Total |
 | Genmab 2025 Annual Report | 150 | |||||
Total Consideration | |
Cash paid for outstanding shares | |
Cash for equity compensation attributable to pre-modification of equity awards1 | |
Cash paid by Genmab directly attributable acquisition related costs2 | |
Total consideration |
Amounts Recognized as of the Acquisition Date | |
Cash and cash equivalents | |
Other current assets1 | |
IPR&D intangible asset | |
Technology platform intangible asset | |
Licenses and patents | |
Other non-current assets2 | |
Non-current contract liabilities | ( |
Current contract liabilities | ( |
Other liabilities3 | ( |
Total identifiable net assets |
 | Genmab 2025 Annual Report | 151 | |||||
Total Consideration | |
Cash paid for outstanding shares | |
Cash for equity compensation attributable to pre-combination service | |
Total consideration | |
Cash acquired | ( |
Cash used for acquisition of asset |
Amounts Recognized as of the Acquisition Date | |
Cash and cash equivalents | |
Other current assets1 | |
Property and equipment | |
IPR&D intangible asset | |
Technology platform intangible asset | |
Other non-current assets2 | |
Deferred tax liability | ( |
Other current liabilities3 | ( |
Total identifiable net assets | |
Goodwill | |
Total consideration |
 | Genmab 2025 Annual Report | 152 | |||||
Consolidated Statements of Comprehensive Income (USD million): | Acquisition Date through December 31, 2024 |
Research and development expenses | |
Selling, general and administrative expenses | |
Acquisition and integration related charges¹ | |
Total |
(USD million) | Twelve Month Period Ended December 31, 2024 |
Revenue | |
Net Profit |
 | Accounting Policies |
 | Genmab 2025 Annual Report | 153 | |||||
 | Management’s Judgements and Estimates – Other Intangible Assets and Goodwill |
 | Genmab 2025 Annual Report | 154 | |||||
 | Genmab 2025 Annual Report | 155 | |||||
 | Genmab 2025 Annual Report | 156 | |||||

Genmab 2025 Annual Report | 157 | ||||||

 | Genmab 2025 Annual Report | 158 | |||||
(USD Millions) | Note | 2025 | 2024¹ Restated | |
Revenue | 2 | 3,659 | 3,213 | |
Cost of product sales | (116) | (72) | ||
Research and development expenses | 3, 5, 6 | (1,713) | (1,545) | |
Selling, general and administrative expenses | 3, 6 | (477) | (346) | |
Integration related charges | (1) | (5) | ||
Total costs and operating expenses | (2,307) | (1,968) | ||
Operating profit | 1,352 | 1,245 | ||
Financial income | 14, 18 | 420 | 2,508 | |
Financial expenses | 14, 18 | (257) | (1,767) | |
Net profit before tax | 1,515 | 1,986 | ||
Corporate tax | 4 | (311) | (356) | |
Net profit | 1,204 | 1,630 | ||
Other comprehensive income: | ||||
Amounts which may be re-classified to the income statement: | ||||
Exchange differences on translation of foreign operations | — | (236) | ||
Total comprehensive income | 1,204 | 1,394 |

 | Genmab 2025 Annual Report | 159 | |||||
December 31, | December 31, | ||
(USD Millions) | Note | 2025 | 2024¹ Restated |
ASSETS | |||
Intangible assets | 5 | 1,877 | 1,872 |
Property and equipment | 6 | 14 | 15 |
Right-of-use assets | 7 | 30 | 33 |
Investments in subsidiaries | 18 | 4,332 | 929 |
Receivables | 10 | 19 | 3 |
Receivables from subsidiaries | 10 | 3 | — |
Loans to subsidiaries | 10 | 4,258 | — |
Deferred tax assets | 4 | — | — |
Other investments | 8 | 25 | 25 |
Total non-current assets | 10,558 | 2,877 | |
Corporate tax receivable | 4 | — | 14 |
Inventories | 9 | 7 | 2 |
Receivables | 10 | 931 | 802 |
Receivables from subsidiaries | 10 | 446 | 135 |
Loans to subsidiaries | 10 | 79 | — |
Marketable securities | 13 | — | 1,574 |
Cash and cash equivalents | 1,358 | 1,259 | |
Total current assets | 2,821 | 3,786 | |
Total assets | 13,379 | 6,663 | |
December 31, | December 31, | ||
(USD Millions) | Note | 2025 | 2024¹ Restated |
SHAREHOLDERS' EQUITY AND LIABILITIES | |||
Share capital | 10 | 10 | |
Share premium | 1,920 | 1,961 | |
Other Reserves | (265) | (265) | |
Retained earnings | 5,055 | 4,108 | |
Total shareholders' equity | 6,720 | 5,814 | |
Borrowings | 15 | 5,001 | — |
Lease liabilities | 7 | 34 | 33 |
Contract liabilities | 11 | 65 | 67 |
Deferred tax liabilities | 4 | 364 | 306 |
Other payables | 12 | 3 | 3 |
Total non-current liabilities | 5,467 | 409 | |
Borrowings | 15 | 273 | — |
Corporate tax payable | 4 | 43 | — |
Payable to subsidiaries | 12 | 358 | 231 |
Lease liabilities | 7 | 3 | 2 |
Contract liabilities | 11 | 4 | 3 |
Other payables | 12 | 511 | 204 |
Total current liabilities | 1,192 | 440 | |
Total liabilities | 6,659 | 849 | |
Total shareholders' equity and liabilities | 13,379 | 6,663 |

 | Genmab 2025 Annual Report | 160 | |||||
(USD Millions) | Note | 2025 | 2024¹ Restated |
Cash flows from operating activities: | |||
Net profit before tax | 1,515 | 1,986 | |
Financial income | 14 | (420) | (2,508) |
Financial expense | 14 | 257 | 1,767 |
Adjustment for non-cash transactions | |||
Share-based compensation expense | 16 | 13 | |
Depreciation | 6 | 5 | |
Amortization | 15 | 5 | |
Impairment losses | 44 | 41 | |
Change in operating assets and liabilities | |||
Receivables | (121) | (169) | |
Inventories | (6) | 3 | |
Other payables | 248 | 50 | |
Cash provided by operating activities before financial items | 1,554 | 1,193 | |
Interest received | 131 | 131 | |
Interest elements of lease payments | 7 | (1) | (1) |
Interest paid | (5) | — | |
Corporate taxes (paid)/received | (186) | (46) | |
Net cash provided by operating activities | 1,493 | 1,277 | |
Cash flows from investing activities: | |||
Transactions with subsidiaries | (7,781) | (1,965) | |
Investment in intangible assets | 5 | (18) | (28) |
Investment in tangible assets | 6 | (1) | (1) |
Marketable securities bought | (991) | (1,248) | |
Marketable securities sold | 2,599 | 1,636 | |
Other investments bought | (2) | (6) | |
Net cash (used in) investing activities | (6,194) | (1,612) | |
(USD Millions) | Note | 2025 | 2024¹ Restated |
Cash flows from financing activities: | |||
Warrants exercised | 23 | 19 | |
Principal elements of lease payments | 7 | (2) | (2) |
Purchase of treasury shares | (430) | (560) | |
Payment of withholding taxes on behalf of employees on net settled RSUs | (18) | (16) | |
Proceeds from issuance of borrowings | 5,500 | — | |
Debt issuance costs paid | (273) | — | |
Net cash (used in) financing activities | 4,800 | (559) | |
Changes in cash and cash equivalents | 99 | (894) | |
Cash and cash equivalents at the beginning of the period | 1,259 | 2,145 | |
Exchange rate adjustments | — | 8 | |
Cash and cash equivalents at the end of the period | 1,358 | 1,259 | |
Cash and cash equivalents include: | |||
Bank deposits | 1,358 | 1,248 | |
Short-term marketable securities | — | 11 | |
Cash and cash equivalents at the end of the period | 1,358 | 1,259 |

 | Genmab 2025 Annual Report | 161 | |||||
(USD Millions) | Share capital | Share premium | Translation Reserves | Retained earnings | Shareholders' equity |
Balance at December 31, 2023¹ Restated | 10 | 1,942 | (29) | 2,950 | 4,873 |
Net profit | — | — | — | 1,630 | 1,630 |
Other Comprehensive Income | — | — | (236) | — | (236) |
Total Comprehensive Income | — | — | (236) | 1,630 | 1,394 |
Exercise of warrants | — | 19 | — | — | 19 |
Purchase of treasury shares | — | — | — | (560) | (560) |
Share-based compensation expenses | — | — | — | 105 | 105 |
Net settlement of RSUs | — | — | — | (16) | (16) |
Tax on items recognized directly in equity | — | — | — | (1) | (1) |
Balance at December 31, 2024¹ Restated | 10 | 1,961 | (265) | 4,108 | 5,814 |
Net profit | — | — | — | 1,204 | 1,204 |
Other Comprehensive Income | — | — | — | — | — |
Total Comprehensive Income | — | — | — | 1,204 | 1,204 |
Exercise of warrants | — | 23 | — | — | 23 |
Purchase of treasury shares | — | — | — | (430) | (430) |
Share-based compensation expenses | — | — | — | 124 | 124 |
Net settlement of RSUs | — | — | — | (18) | (18) |
Share reduction | — | (64) | — | 64 | — |
Tax on items recognized directly in equity | — | — | — | 3 | 3 |
Balance at December 31, 2025 | 10 | 1,920 | (265) | 5,055 | 6,720 |
 | Genmab 2025 Annual Report | 162 | |||||

 | Genmab 2025 Annual Report | 163 | |||||
Q1 2024 | Q2 2024 | Q3 2024 | Q4 2024 | YTD 2024 | |
Average rate | 0.1456 | 0.1443 | 0.1472 | 0.1433 | 0.1452 |
Closing rate | 0.1450 | 0.1435 | 0.1502 | 0.1400 | 0.1400 |
Previously reported in DKK | Presentation currency change | Reported in USD | |
December 31, 2024 | December 31, 2024 | ||
Total assets | 47,069 | (40,406) | 6,663 |
Total liabilities | 5,473 | (4,624) | 849 |
Total shareholders' equity | 41,596 | (35,782) | 5,814 |
Previously reported in DKK | Presentation currency change | Reported in USD | |
January 1, 2024 | January 1, 2024 | ||
Total assets | 37,325 | (31,792) | 5,533 |
Total liabilities | 4,451 | (3,791) | 660 |
Total shareholders' equity | 32,874 | (28,001) | 4,873 |
Previously reported in DKK | Presentation currency change | Reported in USD | |
December 31, 2024 | December 31, 2024 | ||
Net profit | 11,867 | (10,237) | 1,630 |
Comprehensive income | 11,867 | (10,473) | 1,394 |
Previously reported in DKK | Presentation currency change | Reported in USD | |
Cash provided by (used in): | December 31, 2024 | December 31, 2024 | |
Operating activities | (9,553) | 10,830 | 1,277 |
Investing activities | (11,746) | 10,134 | (1,612) |
Financing activities | (3,872) | 3,313 | (559) |
 | Genmab 2025 Annual Report | 164 | |||||
2024 | |||
Revised Balances | Effect of Error Correction | Previously Reported Balances | |
Income Statement: | |||
Revenue | 3,213 | — | 3,213 |
Cost of product sales | (72) | — | (72) |
Research and development expenses | (1,545) | (43) | (1,502) |
Selling, general and administrative expenses | (346) | (64) | (282) |
Integration related charges | (5) | — | (5) |
Operating profit | 1,245 | (107) | 1,352 |
Financial income/expense | 741 | — | 741 |
Net profit before tax | 1,986 | (107) | 2,093 |
Corporate tax | (356) | 24 | (380) |
Net profit | 1,630 | (83) | 1,713 |
Balance Sheet: | |||
Retained earnings | 4,108 | (83) | 4,191 |
Total shareholders' equity | 5,814 | (83) | 5,897 |
Deferred tax liabilities | 306 | (24) | 330 |
Payable to subsidiaries | 231 | 107 | 124 |
Total liabilities | 849 | 83 | 766 |
Total shareholders' equity and liabilities | 6,663 | — | 6,663 |
Cash Flow Statement: | |||
Net profit before tax | 1,986 | (107) | 2,093 |
Cash flows from operating activities before financial items | 1,193 | (107) | 1,300 |
Net cash provided by operating activities | 1,277 | (107) | 1,384 |
Transactions with subsidiaries | (1,965) | 107 | (2,072) |
Net cash (used in) investing activities | (1,612) | 107 | (1,719) |
 | Genmab 2025 Annual Report | 165 | |||||
($ Millions) | December 31, 2024 | Reclass | December 31, 2024 | ||
Financial income: | |||||
Gain on marketable securities | 53 | 184 | 237 | ||
Foreign exchange rate gain | 420 | (173) | 247 | ||
Gain on other investments, net | 17 | (1) | 16 | ||
Total financial income | 490 | 10 | 500 | ||
Financial expenses: | |||||
Loss on marketable securities | (21) | (84) | (105) | ||
Foreign exchange rate loss | (229) | 73 | (156) | ||
Loss on other investments, net | (1) | 1 | — | ||
Total financial expenses | (251) | (10) | (261) | ||
Net financial items | 239 | — | 239 | ||
2025 | 2024 | |
Revenue by type: | ||
Royalties | 3,102 | 2,517 |
Net product sales - External | 5 | — |
Net product sales - Intercompany | 208 | 180 |
Reimbursement revenue - External | 52 | 144 |
Reimbursement revenue - Intercompany | 125 | 165 |
Milestone revenue | 97 | 145 |
Collaboration revenue | 70 | 62 |
Total | 3,659 | 3,213 |
Revenue by collaboration partner: | ||
Janssen | 2,565 | 2,091 |
AbbVie | 66 | 58 |
Roche | 106 | 107 |
Novartis | 446 | 408 |
BioNTech | 40 | 127 |
Pfizer | 76 | 77 |
Other | 21 | — |
Total¹ | 3,320 | 2,868 |
Royalties by product: | ||
DARZALEX | 2,443 | 2,019 |
Kesimpta | 443 | 323 |
TEPEZZA | 105 | 106 |
Other² | 111 | 69 |
Total | 3,102 | 2,517 |
 | Genmab 2025 Annual Report | 166 | |||||
2025 | 2024 | |
Wages and salaries | 103 | 82 |
Share-based compensation | 16 | 13 |
Defined contribution plans | 9 | 7 |
Other social security costs | 3 | — |
Total | 131 | 102 |
Staff costs are included in the income statement as follows: | ||
Research and development expenses | 94 | 78 |
Selling, general and administrative expenses | 37 | 24 |
Total | 131 | 102 |
Average number of FTE | 521 | 492 |
Number of FTE at year-end | 556 | 519 |
2025 | 2024 | |
Current tax | ||
Current tax on profit | 254 | (12) |
Adjustment to deferred tax | 64 | 368 |
Effect of exchange rate adjustment | (7) | — |
Total tax for the period in the income statement | 311 | 356 |
A reconciliation of Genmab's effective tax rate relative to the Danish statutory tax rate is as follows: | ||
2025 | 2024 | |
Net profit before tax | 1,515 | 1,986 |
Tax at the Danish statutory corporation tax rate of 22% for all periods | 333 | 437 |
Tax effect of: | ||
Non-deductible expenses/non-taxable income and other permanent differences, net | (12) | (96) |
All other | (3) | 15 |
Effect of exchange rate adjustment | (7) | — |
Total tax effect | (22) | (81) |
Total tax for the period in the income statement | 311 | 356 |
Total tax for the period in shareholders' equity | (3) | 1 |
Effective Tax Rate | 20.5% | 17.9% |
2025 | 2024 | |
Share-based instruments | 9 | 5 |
Deferred revenue | 16 | 16 |
Intangible assets | (413) | (412) |
Tax losses and credits carried forward | — | 72 |
Other temporary differences | 24 | 13 |
Total deferred tax liabilities | (364) | (306) |
 | Genmab 2025 Annual Report | 167 | |||||
Licenses and Patents | Technology Platform | Acquired IPR&D | Total Intangible Assets | |
2025 | ||||
Cost at the beginning of the year | 187 | 173 | 1,650 | 2,010 |
Additions for the year | 33 | — | 31 | 64 |
Cost at the end of the year | 220 | 173 | 1,681 | 2,074 |
Amortization and impairment losses at the beginning of the year | 138 | — | — | 138 |
Amortization for the year | 3 | 12 | — | 15 |
Impairment losses for the year | 44 | — | — | 44 |
Amortization and impairment losses at the end of the year | 185 | 12 | — | 197 |
Carrying amount at the end of the year | 35 | 161 | 1,681 | 1,877 |
2024 | ||||
Cost at the beginning of the year | 162 | — | — | 162 |
Additions for the year | 35 | 180 | 1,712 | 1,927 |
Exchange Rate Adjustments | (10) | (7) | (62) | (79) |
Cost at the end of the year | 187 | 173 | 1,650 | 2,010 |
Amortization and impairment losses at the beginning of the year | 106 | — | — | 106 |
Amortization for the year | 5 | — | — | 5 |
Impairment losses for the year | 35 | — | — | 35 |
Amortization and impairment losses at the end of the year | 138 | — | — | 138 |
Carrying amount at the end of the year | 49 | 173 | 1,650 | 1,872 |
Leasehold improvements | Equipment, furniture and fixtures | Assets under construction | Total property and equipment | |
2025 | ||||
Cost at January 1 | 10 | 11 | — | 21 |
Additions for the year | — | 1 | — | 1 |
Transfers between the classes | 1 | — | — | 1 |
Disposals for the year | — | — | — | — |
Cost at December 31 | 11 | 12 | — | 23 |
Accumulated depreciation and impairment at January 1 | (1) | (6) | — | (7) |
Depreciation for the year | (1) | (2) | — | (3) |
Disposals for the year | — | — | — | — |
Exchange Rate Adjustments | — | 1 | — | 1 |
Accumulated depreciation and impairment at December 31 | (2) | (7) | — | (9) |
Carrying amount at December 31 | 9 | 5 | — | 14 |
 | Genmab 2025 Annual Report | 168 | |||||
Leasehold improvements | Equipment, furniture and fixtures | Assets under construction | Total property and equipment | |
2024 | ||||
Cost at January 1 | 12 | 12 | — | 24 |
Additions for the year | — | — | — | — |
Transfers between the classes | — | — | — | — |
Disposals for the year | (1) | (1) | — | (2) |
Cost at December 31 | 11 | 11 | — | 22 |
Accumulated depreciation and impairment at January 1 | (1) | (4) | — | (5) |
Depreciation for the year | (1) | (3) | — | (4) |
Disposals for the year | 1 | 1 | — | 2 |
Accumulated depreciation and impairment at December 31 | (1) | (6) | — | (7) |
Carrying amount at December 31 | 10 | 5 | — | 15 |
2025 | 2024 | ||
Depreciation and impairment included in the income statement as follows: | |||
Research and development expenses | 2 | 3 | |
Selling, general and administrative expenses | 1 | 1 | |
Total | 3 | 4 |
2025 | 2024 | |
Right-of-use assets | ||
Balance at January 1 | 33 | 34 |
Additions to right-of-use assets1 | — | 3 |
Depreciation charge for the year | (3) | (2) |
Exchange rate adjustment | — | (2) |
Balance at December 31 | 30 | 33 |
Lease liabilities | ||
Current | 3 | 2 |
Non-current | 34 | 33 |
Total at December 31 | 37 | 35 |
Cash outflow for lease payments | 3 | 3 |
2025 | 2024 | |
Payment due | ||
Less than 1 year | 5 | 4 |
1 to 3 years | 8 | 7 |
More than 3 years but less than 5 years | 9 | 8 |
More than 5 years | 25 | 26 |
Total at December 31 | 47 | 45 |
 | Genmab 2025 Annual Report | 169 | |||||
2025 | 2024 | |
Fund Investments | 23 | 23 |
Privately held equity securities | 2 | 2 |
Total at December 31 | 25 | 25 |
2025 | 2024 | |
Raw materials | 4 | 1 |
Work in progress | — | — |
Finished goods | 3 | 1 |
Total inventories (gross) at December 31 | 7 | 2 |
Allowances at year end | — | — |
Total inventories (net) at December 31 | 7 | 2 |
2025 | 2024 | |
Receivables related to collaboration agreements | 904 | 761 |
Prepayments | 14 | 15 |
Receivables from subsidiaries | 449 | 135 |
Loans to subsidiaries | 4,337 | — |
Interest receivables | 4 | 19 |
Other receivables | 28 | 10 |
Total at December 31 | 5,736 | 940 |
Non-current receivables | 4,280 | 3 |
Current receivables | 1,456 | 937 |
Total at December 31 | 5,736 | 940 |
2025 | 2024 | |
Deferred revenue at January 1 | 70 | 76 |
Customer payment received | 14 | — |
Revenue recognized during the year | (15) | (1) |
Exchange Rate Adjustments | — | (5) |
Total at December 31 | 69 | 70 |
Non-current deferred revenue | 65 | 67 |
Current deferred revenue | 4 | 3 |
Total at December 31 | 69 | 70 |
 | Genmab 2025 Annual Report | 170 | |||||
2025 | 2024 | |
Liabilities related to collaboration agreements | 42 | 18 |
Staff cost liabilities | 21 | 13 |
Accounts payable | 60 | 26 |
Payable to subsidiaries | 358 | 231 |
Other liabilities | 391 | 150 |
Total at December 31 | 872 | 438 |
Non-current other payables | 3 | 3 |
Current other payables | 869 | 435 |
Total at December 31 | 872 | 438 |
2025 | 2024¹ | |
Financial income: | ||
Dividend income from subsidiaries | — | 1,867 |
Interest and other financial income | 134 | 141 |
Interest from subsidiaries | 22 | — |
Gain on marketable securities | 112 | 237 |
Gain on other investments, net | — | 16 |
Foreign exchange rate gain | 152 | 247 |
Total financial income | 420 | 2,508 |
Financial expenses: | ||
Impairment of investment in subsidiaries | — | (1,491) |
Other interest expense | (30) | (13) |
Interest expense on borrowings | (27) | (1) |
Interest to subsidiaries | — | (1) |
Loss on marketable securities | (46) | (105) |
Loss on other investments, net | (2) | — |
Foreign exchange rate loss | (152) | (156) |
Total financial expenses | (257) | (1,767) |
Net financial items | 163 | 741 |
 | Genmab 2025 Annual Report | 171 | |||||
2025 | 2024 | |
Transactions with subsidiaries: | ||
Income statement: | ||
Net product sales | 208 | 180 |
Reimbursement revenue | 125 | 165 |
Cost of product sales | 7 | (4) |
Service fee costs | (1,064) | (914) |
Milestone costs | (67) | (79) |
Impairment intangible assets - Intercompany | (26) | — |
Impairment investment in subsidiaries | — | (1,491) |
Dividend income | — | 1,867 |
Financial income | 22 | — |
Financial expense | — | (1) |
Balance sheet: | ||
Intangible assets | 148 | 144 |
Receivables from subsidiaries - Non-Current | 3 | — |
Loans to subsidiaries - Non-Current | 4,258 | — |
Receivables from subsidiaries - Current | 446 | 135 |
Loans to subsidiaries - Current | 79 | — |
Current payables | (358) | (231) |
 | Genmab 2025 Annual Report | 172 | |||||
2025 | 2024 | |
Cost at January 1 | 2,678 | 777 |
Additions | 3,403 | 1,901 |
Cost at December 31 | 6,081 | 2,678 |
Impairment at January 1 | (1,749) | (286) |
Impairment for the year | — | (1,463) |
Impairment at December 31 | (1,749) | (1,749) |
Carrying amount at December 31 | 4,332 | 929 |
2025 | 2024 | |
Audit fees | 1.7 | 1.5 |
Audit-related fees | 0.8 | 0.3 |
Total | 2.5 | 1.8 |
 | Genmab 2025 Annual Report | 173 | |||||
Executive Management |
 |  |
Jan van de Winkel | Anthony Pagano |
(President & CEO) | (Executive Vice President & CFO) |
Board of Directors |
 |  |  |
Deirdre P. Connelly | Pernille Erenbjerg | Anders Gersel Pedersen |
(Chair) | (Deputy Chair) | |
 |  |  |
Rolf Hoffmann | Paolo Paoletti | Elizabeth O’Farrell |
 |  |  |
Mijke Zachariasse | Michael Kavanagh | Martin Schultz |
(Employee elected) | (Employee elected) | (Employee elected) |
 | Genmab 2025 Annual Report | 174 | |||||
Key audit matter | How our audit addressed the key audit matter |
Valuation of Acquired IPR&D Asset in the Merus N.V. Acquisition financial statements. The Company acquired Merus N.V. (“Merus”) for USD 8.017 billion on December 12, 2025. The Company accounted for the acquisition as an asset acquisition based on an asset concentration test in accordance with IFRS 3 Business Combinations, as substantially all of the fair value of the acquired assets is concentrated in a single identifiable asset. Intangible assets acquired primarily included an in- process research and development intangible asset (“Acquired IPR&D asset”). The Company allocated the cost price of the Acquired IPR&D asset using an income approach to estimate the fair value at the acquisition date. The fair value determination of the Acquired IPR&D asset required the Company to apply significant estimates and assumptions related to the forecasted future cash flows, such as probabilities of technical and regulatory success, and the determination of the discount rates. We identified the valuation of the Acquired IPR&D asset for the Merus acquisition as a key audit matter because of the high level of complexity and management judgement involved in determining the above outlined significant estimates and assumptions used by the Company to determine the fair value of the asset. This required a high degree of auditor judgement and an increased extent of effort when performing audit procedures to evaluate the reasonableness of management’s estimates and assumptions. | Our audit procedures related to the Company’s valuation of the Acquired IPR&D asset in the Merus acquisition included the following, among others: •We performed corroborative inquiries of key individuals from senior leadership, including research & development, and personnel involved in forecasting the future cash flows in determining the appropriateness of the probabilities of technical and regulatory success. •We tested the effectiveness of controls relating to management's review of the significant estimates and assumptions related to the forecasted future cash flows, including the determination of the probabilities of technical and regulatory success and discount rates applied. •We evaluated the probabilities of technical and regulatory success against external medical studies and industry benchmarks to determine if these were corroborative or contradictory to the probabilities of technical and regulatory success applied by management. •With the assistance of our valuation specialists, we evaluated the appropriateness of the valuation method and we tested the source information and inputs applied in the determination of the discount rates, including comparison to publicly available information of comparable companies, and tested the mathematical accuracy of the calculation. |
 | Genmab 2025 Annual Report | 175 | |||||
Revenue recognition of royalty revenue statements. The Company recognized royalty revenue, where revenue is recognized based on net sales by collaboration partners. The Company uses net sales provided by its collaboration partners as an input to its calculation of the amount of royalty revenue to recognize in each period. The preliminary net sales data provided by the collaboration partner may change once final net sales data is available. We identified the revenue recognition of royalty contracts for selected products as a key audit matter because of the significant estimation uncertainty related to the net sales data provided by collaboration partners. Specifically, the collaboration partner’s estimate of net sales could change based on the final net sales impacting the royalty revenue recognized in each period. This required a high degree of auditor judgement and an increased extent of effort when performing audit procedures to evaluate the reliability of management’s estimates of the net sales. Further, the contracts with the collaboration partners are complex and contain multiple clauses that directly impact revenue recognition, which require an increased extent of audit effort to ensure accurate and complete revenue recognition. | Our audit procedures related to the royalty revenue recognized based on the significant assumption of estimated net sales provided by the collaboration partners and the complex and multiple clauses in the contracts included the following, among others: •We tested the effectiveness of controls relating to management’s review of the estimated net sales used in the determination of royalty revenue recognition. •We tested the overall reliability of the estimated net sales reported by the collaboration partners by assessing the historical accuracy of the estimates. •We tested the recognition of royalty revenue by reconciling to the contract terms, cash receipts and royalty reports from collaboration partners or reported net sales. •We obtained external confirmations from collaboration partners on the estimated and actual net sales amounts reported. |
 | Genmab 2025 Annual Report | 176 | |||||
 | Genmab 2025 Annual Report | 177 | |||||
 |  |
Sumit Sudan | Niels Skannerup Vendelbo |
State Authorised Public Accountant | State Authorised Public Accountant |
Identification No (MNE) mne33716 | Identification No (MNE) mne34532 |
 | Genmab 2025 Annual Report | 178 | |||||
 | Genmab 2025 Annual Report | 179 | |||||
 |  |
Sumit Sudan | Niels Skannerup Vendelbo |
State Authorised Public Accountant | State Authorised Public Accountant |
Identification No (MNE) mne33716 | Identification No (MNE) mne34532 |

Genmab 2025 Annual Report | 180 | ||||||

Genmab 2025 Annual Report | 181 | ||||||
