
1 Fourth Quarter & Full Year 2025 Earnings Presentation March 2, 2026

2 Legal Information USE OF NON-GAAP FINANCIAL MEASURES To supplement Quanterix's preliminary financial information presented on a GAAP basis, Quanterix has provided certain non-GAAP financial measures, including adjusted EBITDA, adjusted EBITDA margin, adjusted cash usage, adjusted gross profit, adjusted gross margin, adjusted total operating expenses, and adjusted loss from operations. Management uses these non-GAAP financial measures to evaluate our operating performance in manner that allows for meaningful period-to-period comparison and analysis of trends in our business and our competitors. Management believes that presentation of these non-GAAP financial measures provides useful information to investors in assessing our operating performance within our industry and in order to allow comparability to the presentation of other companies in our industry. The non-GAAP financial measures presented herein should be considered in conjunction with, and not as a substitute for, the financial information presented in accordance with GAAP. For example, adjusted EBITDA excludes a number of expense items that are included in net loss and adjusted cash usage excludes certain actual cash payments. As a result, positive adjusted EBITDA or positive adjusted cash usage may be achieved even where we record a significant net loss or reduction in our cash and marketable securities balances in accordance with U.S. GAAP. Investors are encouraged to review the reconciliation of these non-GAAP financial measures to their most directly comparable GAAP financial measures set forth herein. Quanterix does not forecast many of the excluded items for internal use and therefore information reconciling forward-looking non-GAAP financial measures to U.S. GAAP financial measures is not available without unreasonable effort and is not provided. The occurrence, timing, and amount of any of the items excluded from U.S. GAAP to calculate non-GAAP financial measures could significantly impact our U.S. GAAP results. Please refer to our fourth quarter 2025 earnings release for additional discussion of non-GAAP financial measures. Unless otherwise specified, all information contained herein is provided as of Dec 31, 2025. CAUTIONARY STATEMENT REGARDING FORWARD-LOOKING STATEMENTS Statements included in this presentation that are not historical in nature or do not relate to current facts are intended to be, and are hereby identified as, forward-looking statements for purposes of the safe harbor provided by Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended. Forward-looking statements include, among other things, statements about Quanterix’s future business outlook, operations, strategy and financial performance, including statements related to our expectations about consistent profitable revenue growth and achieving cash flow breakeven performance, the development and commercialization of our products, the benefits and synergies we may realize from the acquisition of Akoya Biosciences Inc., and under the header “2026 Business Outlook.”. Words and phrases such as “may,” “approximately,” “continue,” “should,” “expects,” “projects,” “anticipates,” “is likely,” “look ahead,” “look forward,” “believes,” “will,” “intends,” “estimates,” “strategy,” “plan,” “could,” “potential,” “possible” and variations of such words and similar expressions are intended to identify such forward- looking statements. Forward-looking statements are subject to certain risks and uncertainties that are difficult to predict with regard to, among other things, timing, extent, likelihood and degree of occurrence, which could cause actual results to differ materially from anticipated results. Such risks and uncertainties include, among others, the following possibilities with respect to Quanterix’s future business, operations, strategy and financial performance: risks related to the impact of changes in U.S. government policies, including impacts of tariffs and reductions in federal research funding; risks associated with the anticipated timing for launch of, and features of, Quanterix’s next-generation instruments to upgrade its existing platforms; risks related to Quanterix’s ability to improve existing diagnostics and develop new diagnostic tests and tools; risks related to Quanterix’s ability to successfully penetrate the diagnostics market; risks related to Quanterix’s ability to retain and expand its customer base and achieve sufficient market acceptance of its products; risks related to the ability of Quanterix’s contract manufacturers and suppliers to reliably and consistently manufacture and supply our instruments; risks that Quanterix may fail to realize the anticipated benefits and synergies of its recent acquisitions of Emission, Inc. and Akoya Biosciences Inc.; risk that integrating Quanterix’s business with that of Akoya could be more difficult, costly or time-consuming than expected; risks that Quanterix’s estimates regarding expenses, future revenues, capital requirements, and needs for additional financing could be incorrect; risks related to Quanterix’s ability to maintain effective internal control over financial reporting and disclosure controls and procedures; and risks related to defects or other quality issues in Quanterix’s products that could lead to unforeseen costs, product recalls, adverse regulatory actions, negative publicity and litigation. Additional factors that could cause results to differ materially from those described above can be found in the periodic reports filed by Quanterix with the SEC, including the “Risk Factors” sections contained therein, which are available on the SEC’s website at www.sec.gov. All forward-looking statements, expressed or implied, included in this press release are expressly qualified in their entirety by the cautionary statements contained or referred to herein. If one or more events related to these or other risks or uncertainties materialize, or if Quanterix’s underlying assumptions prove to be incorrect, actual results may differ materially from what Quanterix anticipates. Quanterix cautions readers not to place undue reliance on any such forward- looking statements, which speak only as of the date they are made and are based on information available at that time. Quanterix does not assume any obligation to update or otherwise revise any forward-looking statements to reflect circumstances or events that occur after the date the forward-looking statements were made or to reflect the occurrence of unanticipated events except as required by federal securities laws.

3 Q4-25: Key Messages Revenue stabilization and execution of cost synergies demonstrate that cash flow breakeven performance is achievable in 2nd half of 2026 • Reported fourth quarter 2025 revenue of $43.9M and adjusted cash usage of $3M • Captured $74M of $85M cost synergy target Hitting key milestones and gaining momentum in Alzheimer’s Diagnostics • Filed 510K with FDA, gained approval from CMS for reimbursement rate of $897 Material weaknesses remediated • Sets stronger foundation for growth Delivered on guidance for year-end cash balance • Reported $122M of cash, significantly reduced adjusted cash usage
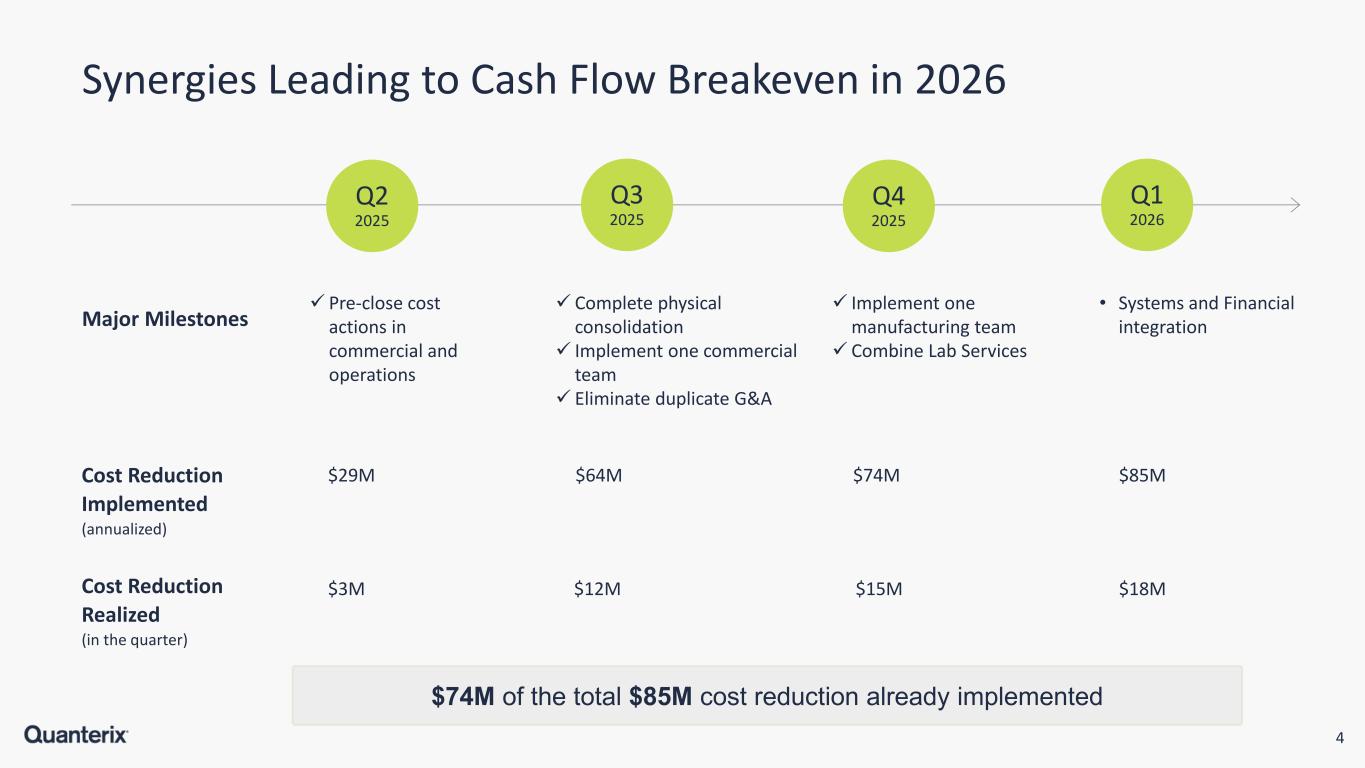
4 $74M of the total $85M cost reduction already implemented Synergies Leading to Cash Flow Breakeven in 2026 Major Milestones Cost Reduction Implemented (annualized) Complete physical consolidation Implement one commercial team Eliminate duplicate G&A Implement one manufacturing team Combine Lab Services • Systems and Financial integration $74M $85M $29M $64M Cost Reduction Realized (in the quarter) $3M $12M Pre-close cost actions in commercial and operations $15M $18M Q2 2025 Q3 2025 Q4 2025 Q1 2026
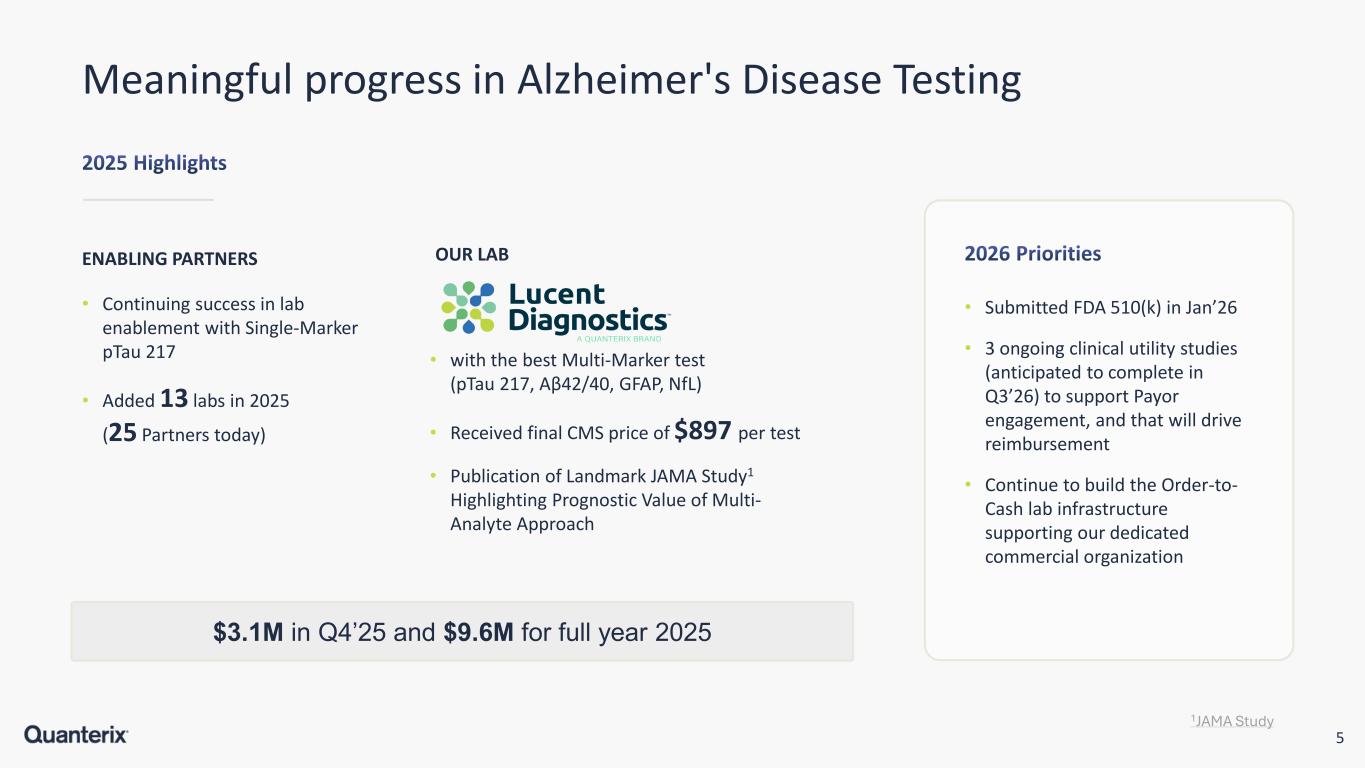
5 Meaningful progress in Alzheimer's Disease Testing ENABLING PARTNERS • Submitted FDA 510(k) in Jan’26 • 3 ongoing clinical utility studies (anticipated to complete in Q3’26) to support Payor engagement, and that will drive reimbursement • Continue to build the Order-to- Cash lab infrastructure supporting our dedicated commercial organization $3.1M in Q4’25 and $9.6M for full year 2025 • Continuing success in lab enablement with Single-Marker pTau 217 • Added 13 labs in 2025 (25 Partners today) OUR LAB • with the best Multi-Marker test (pTau 217, Aβ42/40, GFAP, NfL) • Received final CMS price of $897 per test • Publication of Landmark JAMA Study1 Highlighting Prognostic Value of Multi- Analyte Approach 1JAMA Study
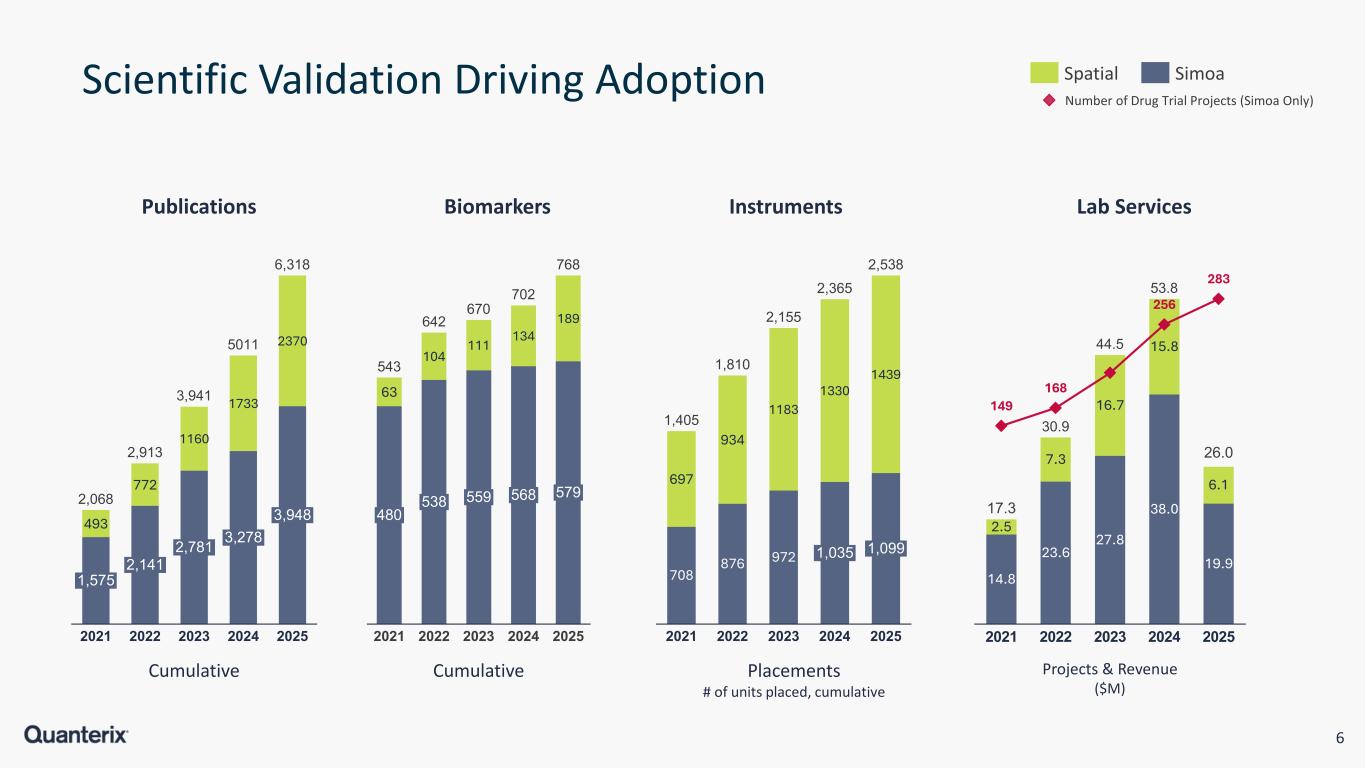
6 Scientific Validation Driving Adoption Spatial Simoa Number of Drug Trial Projects (Simoa Only) 493 772 1160 1733 2370 1,575 2021 2,141 2022 2,781 2023 3,278 2024 3,948 2025 2,068 2,913 3,941 5011 6,318 63 104 111 134 189 480 2021 538 2022 559 2023 568 2024 579 2025 543 642 670 702 768 708 876 972 697 934 1183 1330 1439 2021 2022 2023 1,035 2024 1,099 2025 1,405 1,810 2,155 2,365 2,538 14.8 23.6 27.8 38.0 19.9 2.5 7.3 16.7 15.8 6.1 149 168 256 283 2021 2022 2023 2024 2025 17.3 30.9 44.5 53.8 26.0 Publications CumulativeCumulative Projects & Revenue ($M) Placements # of units placed, cumulative Biomarkers Instruments Lab Services
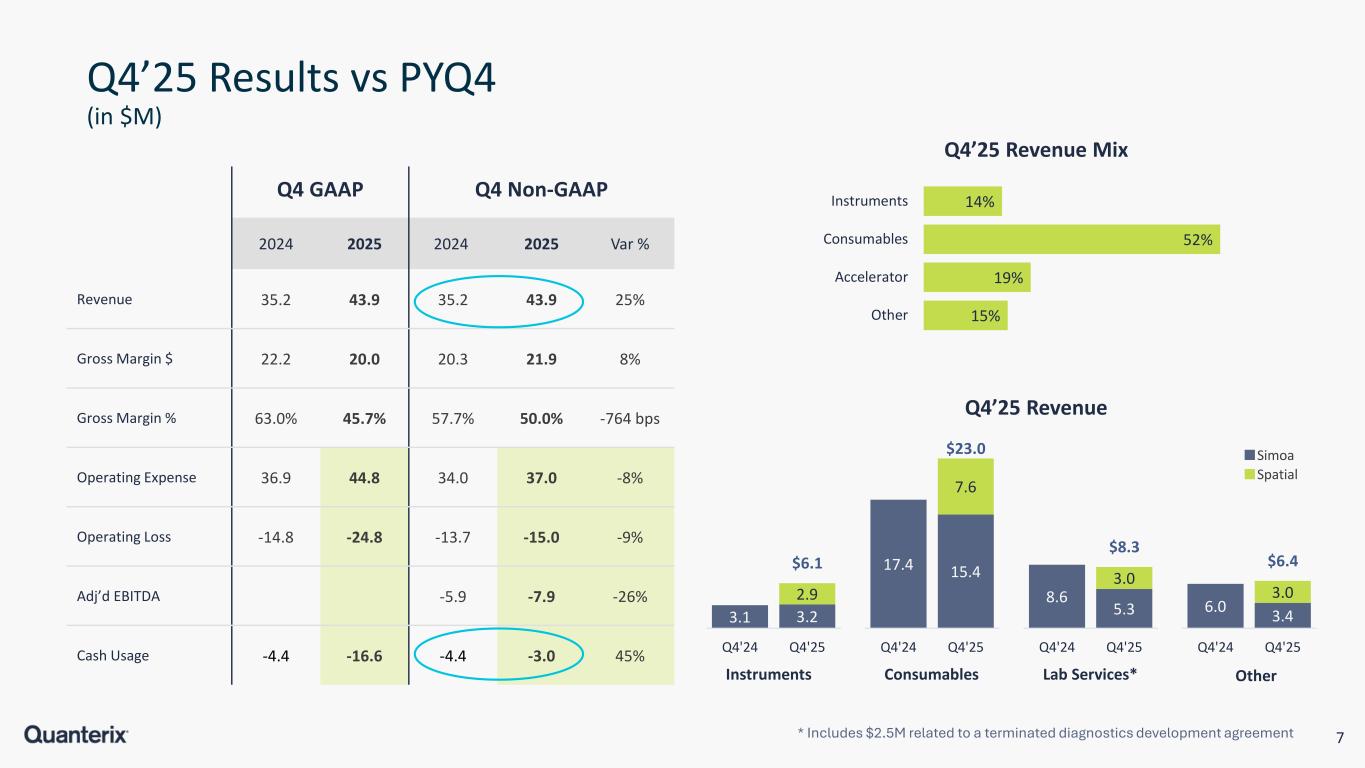
7 Q4’25 Results vs PYQ4 (in $M) Q4 GAAP Q4 Non-GAAP 2024 2025 2024 2025 Var % Revenue 35.2 43.9 35.2 43.9 25% Gross Margin $ 22.2 20.0 20.3 21.9 8% Gross Margin % 63.0% 45.7% 57.7% 50.0% -764 bps Operating Expense 36.9 44.8 34.0 37.0 -8% Operating Loss -14.8 -24.8 -13.7 -15.0 -9% Adj’d EBITDA -5.9 -7.9 -26% Cash Usage -4.4 -16.6 -4.4 -3.0 45% 14% 52% 19% 15% Instruments Consumables Accelerator Other Q4’25 Revenue Mix * Includes $2.5M related to a terminated diagnostics development agreement 3.1 3.2 2.9 $6.1 Q4'24 Q4'25 Instruments 17.4 15.4 7.6 $23.0 Q4'24 Q4'25 Consumables 8.6 5.3 3.0 $8.3 Q4'24 Q4'25 Lab Services* 6.0 3.4 3.0 $6.4 Q4'24 Q4'25 Other Q4’25 Revenue Spatial Simoa
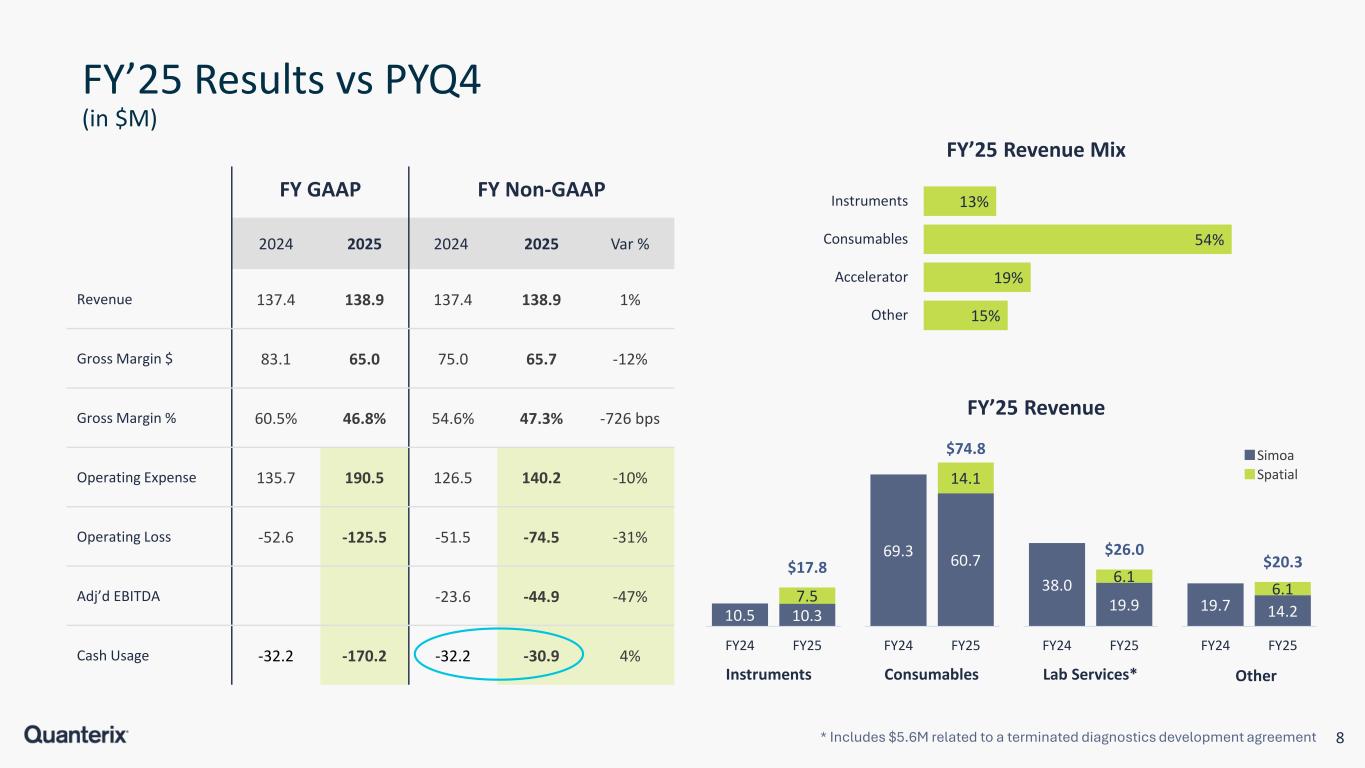
8 FY’25 Results vs PYQ4 (in $M) FY GAAP FY Non-GAAP 2024 2025 2024 2025 Var % Revenue 137.4 138.9 137.4 138.9 1% Gross Margin $ 83.1 65.0 75.0 65.7 -12% Gross Margin % 60.5% 46.8% 54.6% 47.3% -726 bps Operating Expense 135.7 190.5 126.5 140.2 -10% Operating Loss -52.6 -125.5 -51.5 -74.5 -31% Adj’d EBITDA -23.6 -44.9 -47% Cash Usage -32.2 -170.2 -32.2 -30.9 4% 13% 54% 19% 15% Instruments Consumables Accelerator Other FY’25 Revenue Mix * Includes $5.6M related to a terminated diagnostics development agreement 10.5 10.3 7.5 $17.8 FY24 FY25 Instruments 69.3 60.7 14.1 $74.8 FY24 FY25 Consumables 38.0 19.9 6.1 $26.0 FY24 FY25 Lab Services* 19.7 14.2 6.1 $20.3 FY24 FY25 Other FY’25 Revenue Spatial Simoa
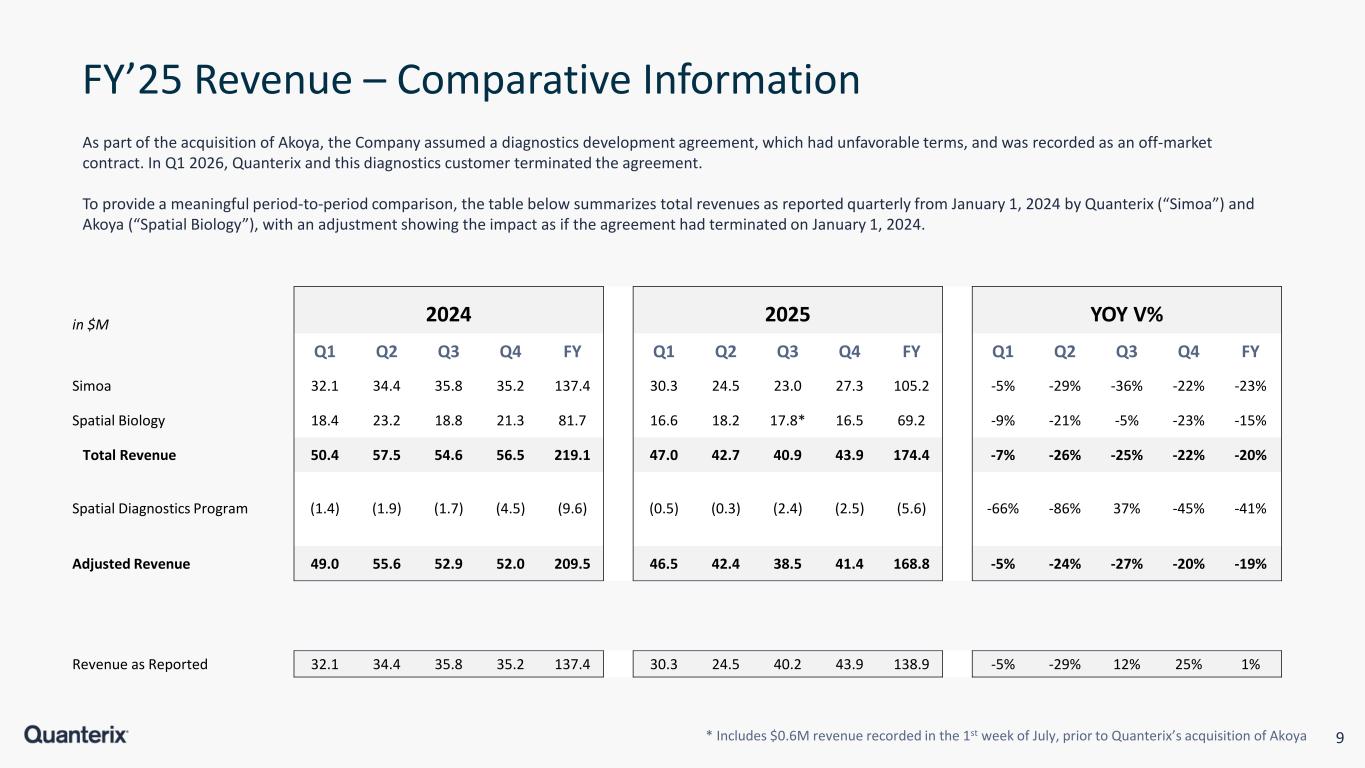
9 FY’25 Revenue – Comparative Information * Includes $0.6M revenue recorded in the 1st week of July, prior to Quanterix’s acquisition of Akoya As part of the acquisition of Akoya, the Company assumed a diagnostics development agreement, which had unfavorable terms, and was recorded as an off-market contract. In Q1 2026, Quanterix and this diagnostics customer terminated the agreement. To provide a meaningful period-to-period comparison, the table below summarizes total revenues as reported quarterly from January 1, 2024 by Quanterix (“Simoa”) and Akoya (“Spatial Biology”), with an adjustment showing the impact as if the agreement had terminated on January 1, 2024. in $M 2024 2025 YOY V% Q1 Q2 Q3 Q4 FY Q1 Q2 Q3 Q4 FY Q1 Q2 Q3 Q4 FY Simoa 32.1 34.4 35.8 35.2 137.4 30.3 24.5 23.0 27.3 105.2 -5% -29% -36% -22% -23% Spatial Biology 18.4 23.2 18.8 21.3 81.7 16.6 18.2 17.8* 16.5 69.2 -9% -21% -5% -23% -15% Total Revenue 50.4 57.5 54.6 56.5 219.1 47.0 42.7 40.9 43.9 174.4 -7% -26% -25% -22% -20% Spatial Diagnostics Program (1.4) (1.9) (1.7) (4.5) (9.6) (0.5) (0.3) (2.4) (2.5) (5.6) -66% -86% 37% -45% -41% Adjusted Revenue 49.0 55.6 52.9 52.0 209.5 46.5 42.4 38.5 41.4 168.8 -5% -24% -27% -20% -19% Revenue as Reported 32.1 34.4 35.8 35.2 137.4 30.3 24.5 40.2 43.9 138.9 -5% -29% 12% 25% 1%

10 2026 Guidance Full Year Revenue: $169 to $174 million 0–3% revenue growth after the effect of a terminated diagnostics development agreement Gross Margin GAAP gross margin between 45 to 49% Adjusted gross margin (Non-GAAP) between 49% to 53% Anticipate cash flow breakeven in the 2nd half of 2026 Exit the year with ~$100 million in cash, and no debt
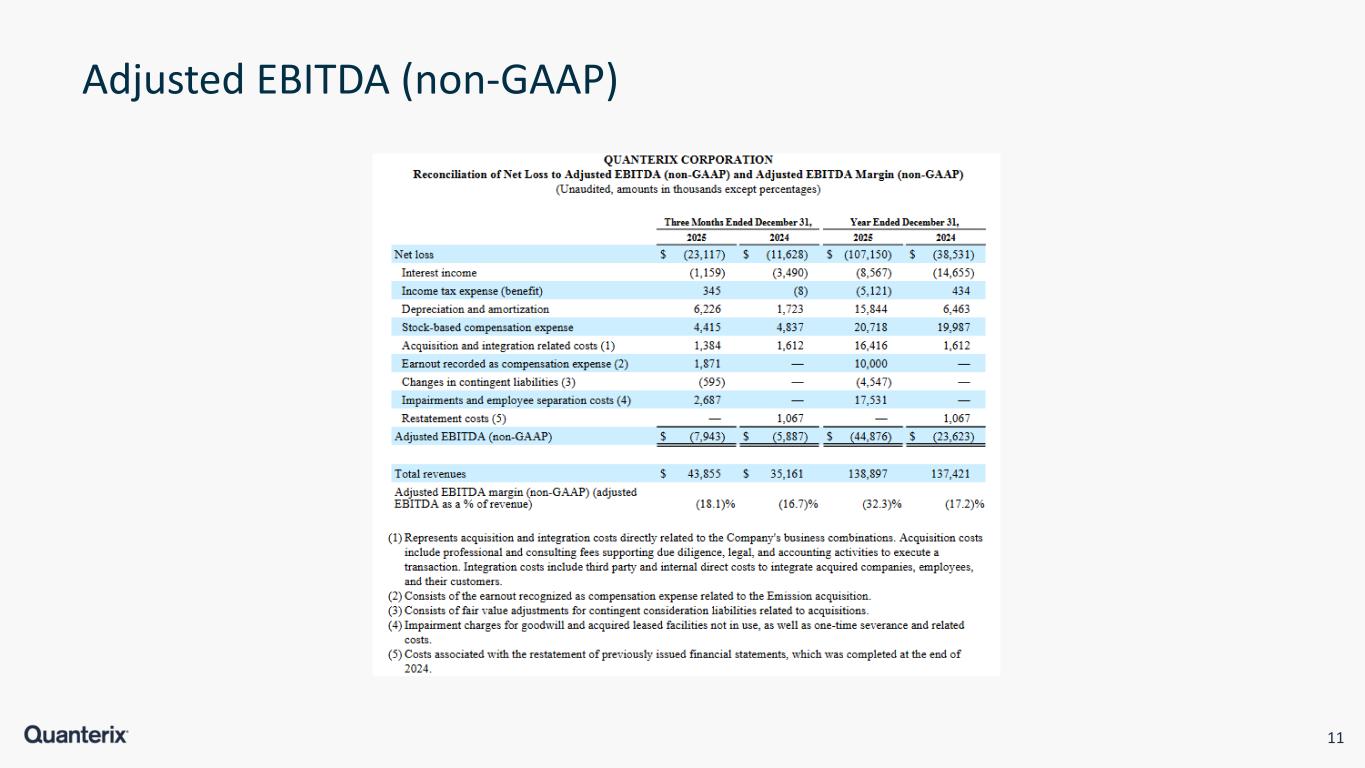
11 Adjusted EBITDA (non-GAAP)
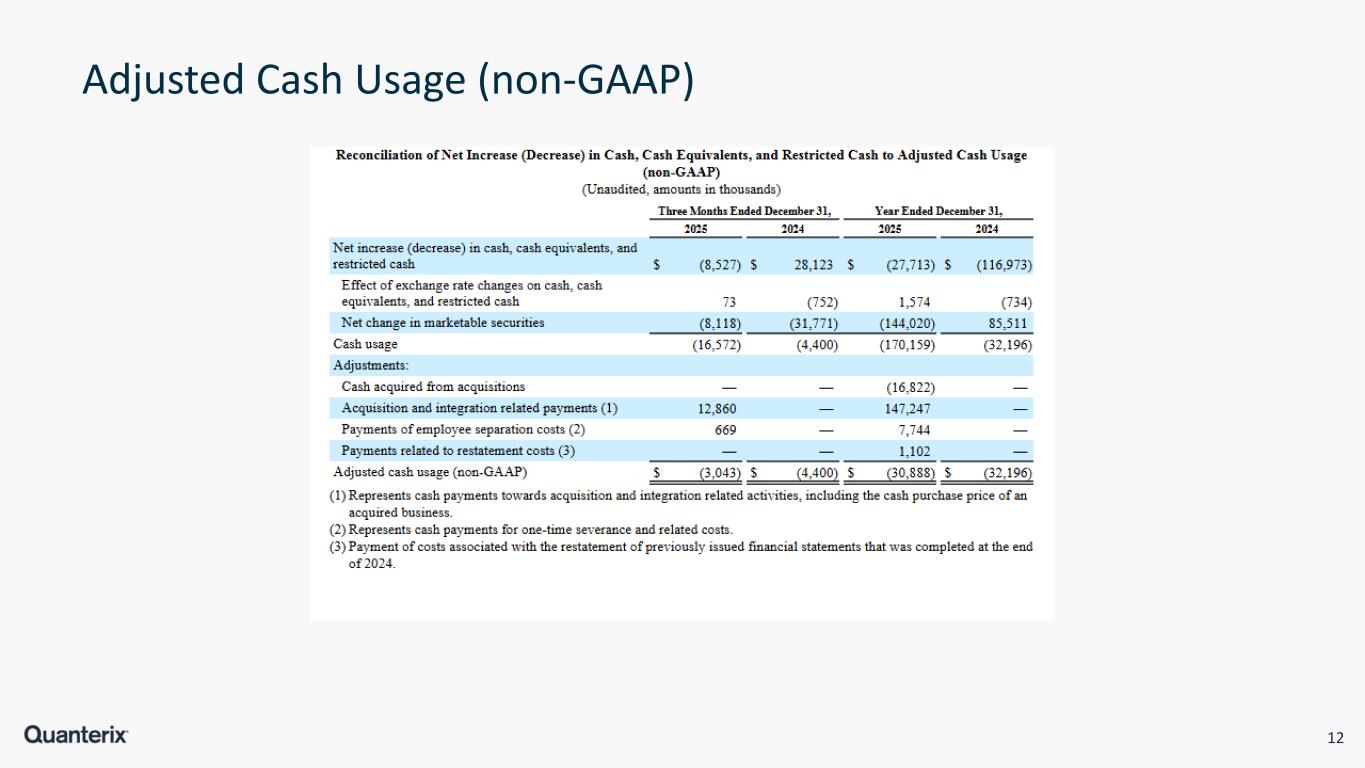
12 Adjusted Cash Usage (non-GAAP)
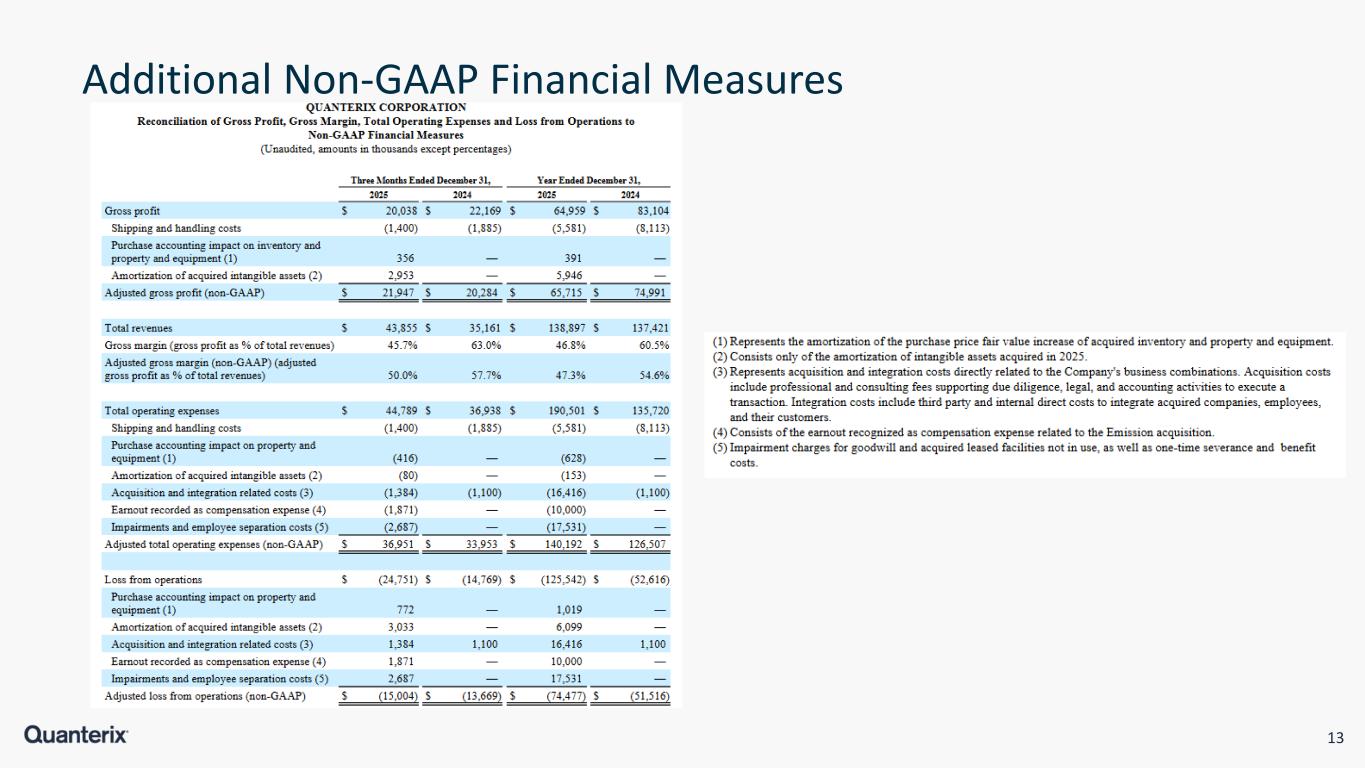
13 Additional Non-GAAP Financial Measures

Contact Us General inquiries 900 Middlesex Turnpike, Billerica, MA 01821 USA 617.301.9400 info@quanterix.com www.quanterix.com