

.2 Phase 3 X-TOLE2 Study: Topline Results MARCH 09, 2026 NASDAQ: XENE xenon-pharma.com

On Today’s Call Ian Mortimer Chris Kenney, MD Darren Cline Tucker Kelly President & Chief Medical Officer Chief Commercial Officer Chief Financial Officer Chief Executive Officer MARCH 2026 2

Forward Looking Statement/Safe Harbor This slide presentation and the accompanying oral commentary contain forward-looking statements (within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended, and the Private Securities Litigation Reform Act of 1995 and Canadian Securities laws) that involve risks, uncertainties and assumptions. If the risks or uncertainties ever materialize or the assumptions prove incorrect, our results may differ materially from those expressed or implied by such forward- looking statements. All statements other than statements of historical fact could be deemed forward-looking and include statements regarding the timing of and potential results from clinical studies; the potential efficacy, safety profile, future development plans in current and anticipated indications, addressable market, regulatory success and commercial potential of our and our partners’ product candidates; the efficacy of our clinical study designs; our ability to successfully develop and achieve milestones in our azetukalner and other pipeline and development programs, including the anticipated filing of INDs and NDAs; the timing and results of our interactions with regulators, including the timing of any NDA submission; our ability to successfully develop and obtain regulatory approval of azetukalner and our other product candidates; anticipated timing of topline data readout from our clinical studies of azetukalner; and our expectation that we will have sufficient cash to fund operations into the second half of 2027. These forward-looking statements are based on current assumptions that involve risks, uncertainties and other factors that may cause the actual results, events, or developments to be materially different from those expressed or implied by such forward-looking statements. These risks and uncertainties, many of which are beyond our control, include, but are not limited to: clinical studies may not demonstrate safety and efficacy of any of our or our collaborators’ product candidates; promising results from pre-clinical development activities or early clinical study results may not be replicated in later clinical studies; our assumptions regarding our planned expenditures and sufficiency of our cash to fund operations may be incorrect; our ongoing discovery and pre-clinical efforts may not yield additional product candidates; any of our or our collaborators’ product candidates, including azetukalner, may fail in development, may not receive required regulatory approvals, or may be delayed to a point where they are not commercially viable; we may not achieve additional milestones in our proprietary or partnered programs; regulatory agencies may impose additional requirements or delay the initiation or completion of clinical studies; the impact of market, industry, and regulatory conditions on clinical study enrollment; the impact of competition; the impact of expanded product development and clinical activities on operating expenses; the impact of new or changing laws and regulations; the impact of unstable economic conditions in the general domestic and global economic markets; adverse conditions from geopolitical events; as well as the other risks identified in our filings with the U.S. Securities and Exchange Commission and the securities commissions in British Columbia, Alberta, and Ontario. These forward- looking statements speak only as of the date hereof and we assume no obligation to update these forward-looking statements, and readers are cautioned not to place undue reliance on such forward-looking statements. Xenon and the Xenon logo are registered trademarks or trademarks of Xenon Pharmaceuticals Inc. in the US, Canada, and elsewhere. All other trademarks belong to their respective owner. MARCH 2026 3

Opening Remarks Ian Mortimer President & Chief Executive Officer

Thank you to our partners in the epilepsy community, including patient advocates, clinicians and study participants. Hadley, living with epilepsy

POSITIVE TOPLINE DATA Azetukalner in Focal Onset Seizures MARCH 2026 6

Azetukalner’s Differentiated Profile in FOS Robust Clinical Data Well-Documented Safety Profile Compelling double-blind efficacy data in FOS 800+ patient years of data in FOS patients, patients from two placebo-controlled studies with some dosed for more than 5 years Consistent safety profile between X-TOLE and Durable long-term seizure reduction and seizure freedom data in ongoing X-TOLE OLE X-TOLE2 studies Ease-of-Use Novel Mechanism Once-daily dosing Highly potent K 7.2/7.3 potassium channel V opener No titration needed Differentiated mechanism may allow for No meaningful DDIs with other ASMs or rational polytherapy anticipated monitoring requirements FOS, focal onset seizures; DDIs, drug-drug interactions; ASMs, anti-seizure medications Source: 1. “Long-Term Safety and Efficacy of Azetukalner, a Novel, Potent KV7 Potassium Channel Opener, in Adults With Focal Epilepsy: ≥48-Month Interim Analysis of the MARCH 2026 7 7 Ongoing 7-Year X-TOLE Open-Label Extension.” 2025 Annual Meeting of the American Epilepsy Society (AES). 2. French JA et al. JAMA Neurol.2023;80(11):1145-1154.

X-TOLE2 Results Summary Positive Phase 3 study results support an anticipated NDA submission in Q3 2026 PRIMARY ENDPOINT WAS MET: Highly statistically significant, dose-dependent reduction from baseline in median monthly FOS frequency (MPC) over the 12-week treatment period vs. placebo Responder rate 50: Statistically significant, dose-dependent (15 and 25 mg) increase in number of responders with >50% reduction in monthly FOS frequency Onset of efficacy: Rapid onset of response as assessed by statistically significant MPC achieved within one week for AZK 25 mg vs. placebo Overall health status: Significant improvements in PGI-C/CGI-C for both 15 and 25 mg treatment groups vs. placebo Safety and tolerability: AZK profile consistent with Phase 2b X-TOLE study AZK, azetukalner; PGI-C, Patient Global Impression of Change; CGI-C, Clinical Global Impression of Change; MPC, median percent change; FOS, focal onset seizure MARCH 2026 8

Topline Results from Phase 3 X-TOLE2 Study Chris Kenney, MD Chief Medical Officer
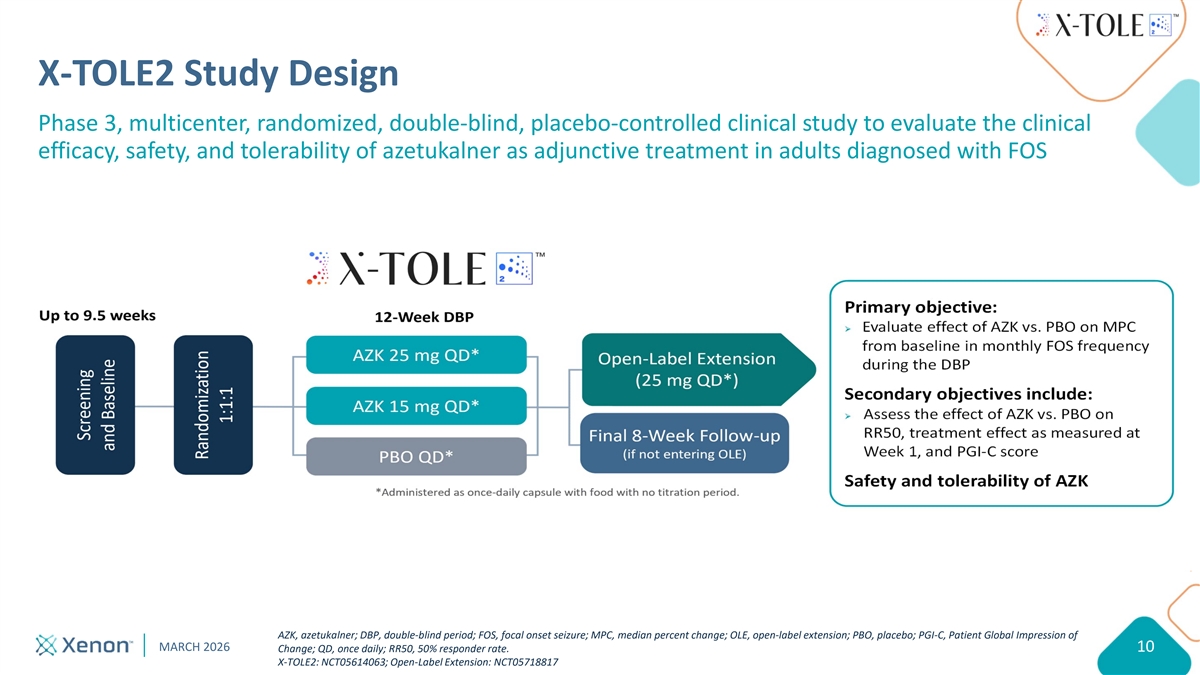
X-TOLE2 Study Design Phase 3, multicenter, randomized, double-blind, placebo-controlled clinical study to evaluate the clinical efficacy, safety, and tolerability of azetukalner as adjunctive treatment in adults diagnosed with FOS Primary objective: Ø Evaluate effect of AZK vs. PBO on MPC from baseline in monthly FOS frequency during the DBP Secondary objectives include: Ø Assess the effect of AZK vs. PBO on RR50, treatment effect as measured at Week 1, and PGI-C score Safety and tolerability of AZK AZK, azetukalner; DBP, double-blind period; FOS, focal onset seizure; MPC, median percent change; OLE, open-label extension; PBO, placebo; PGI-C, Patient Global Impression of MARCH 2026 Change; QD, once daily; RR50, 50% responder rate. 10 X-TOLE2: NCT05614063; Open-Label Extension: NCT05718817
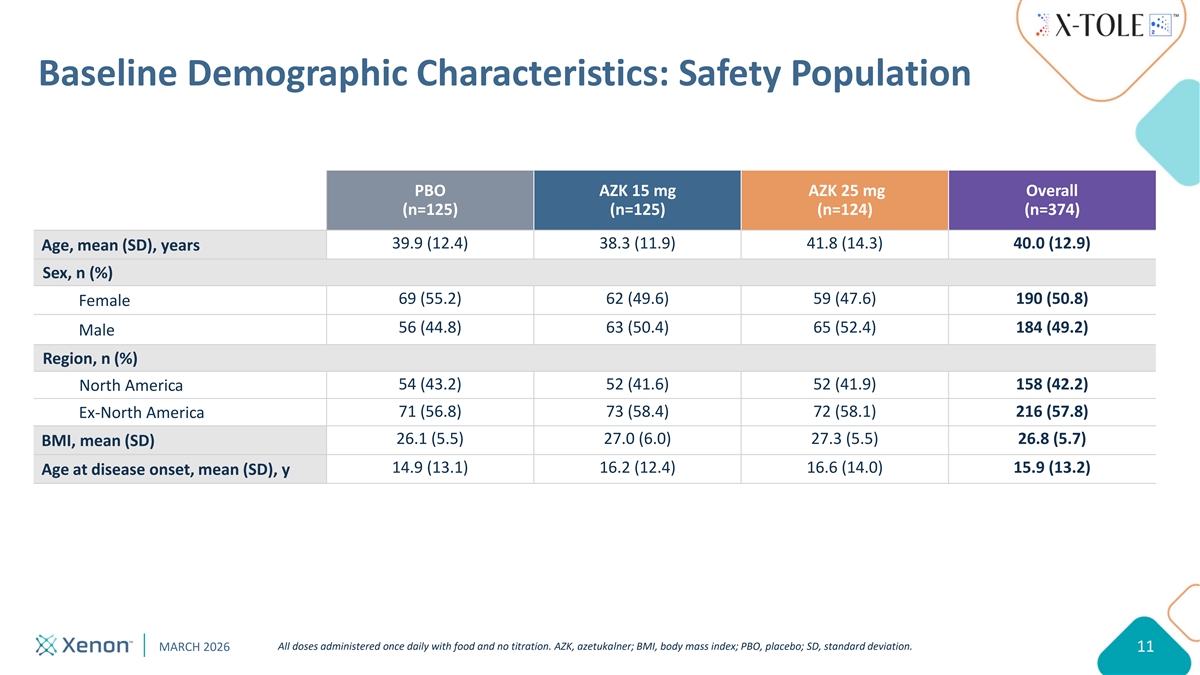
Baseline Demographic Characteristics: Safety Population PBO AZK 15 mg AZK 25 mg Overall (n=125) (n=125) (n=124) (n=374) 39.9 (12.4) 38.3 (11.9) 41.8 (14.3) 40.0 (12.9) Age, mean (SD), years Sex, n (%) 69 (55.2) 62 (49.6) 59 (47.6) 190 (50.8) Female 56 (44.8) 63 (50.4) 65 (52.4) 184 (49.2) Male Region, n (%) 54 (43.2) 52 (41.6) 52 (41.9) 158 (42.2) North America 71 (56.8) 73 (58.4) 72 (58.1) 216 (57.8) Ex-North America 26.1 (5.5) 27.0 (6.0) 27.3 (5.5) 26.8 (5.7) BMI, mean (SD) 14.9 (13.1) 16.2 (12.4) 16.6 (14.0) 15.9 (13.2) Age at disease onset, mean (SD), y All doses administered once daily with food and no titration. AZK, azetukalner; BMI, body mass index; PBO, placebo; SD, standard deviation. MARCH 2026 11
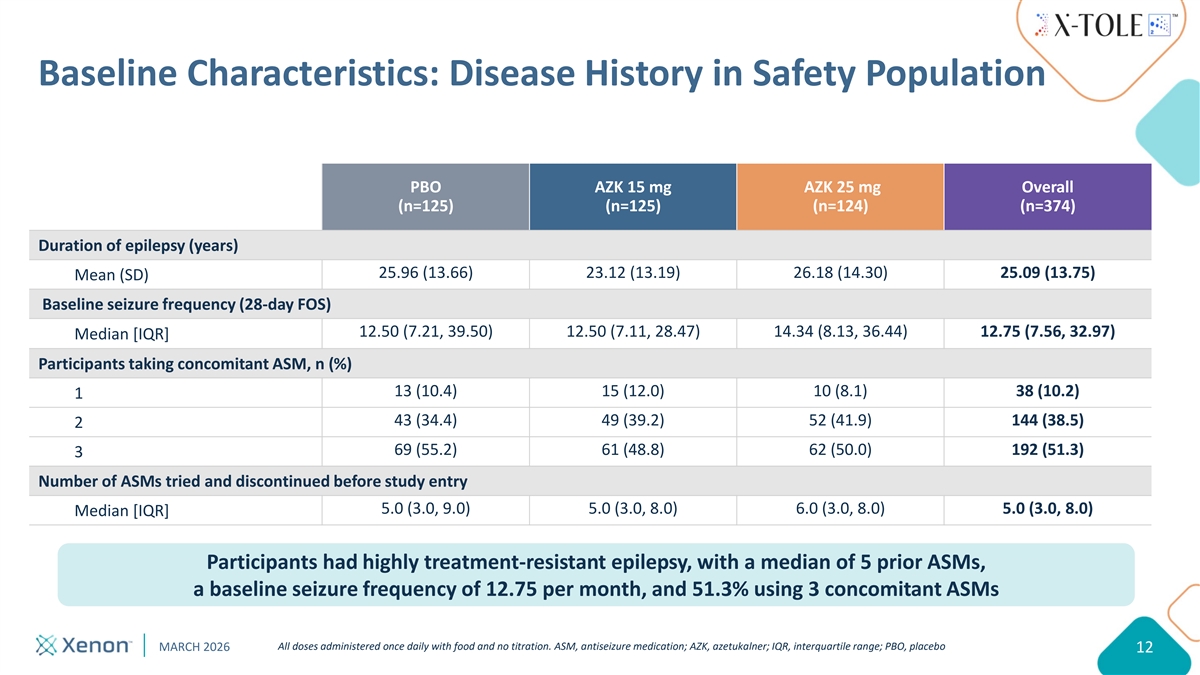
Baseline Characteristics: Disease History in Safety Population PBO AZK 15 mg AZK 25 mg Overall (n=125) (n=125) (n=124) (n=374) Duration of epilepsy (years) 25.96 (13.66) 23.12 (13.19) 26.18 (14.30) 25.09 (13.75) Mean (SD) Baseline seizure frequency (28-day FOS) 12.50 (7.21, 39.50) 12.50 (7.11, 28.47) 14.34 (8.13, 36.44) 12.75 (7.56, 32.97) Median [IQR] Participants taking concomitant ASM, n (%) 13 (10.4) 15 (12.0) 10 (8.1) 38 (10.2) 1 43 (34.4) 49 (39.2) 52 (41.9) 144 (38.5) 2 69 (55.2) 61 (48.8) 62 (50.0) 192 (51.3) 3 Number of ASMs tried and discontinued before study entry 5.0 (3.0, 9.0) 5.0 (3.0, 8.0) 6.0 (3.0, 8.0) 5.0 (3.0, 8.0) Median [IQR] Participants had highly treatment-resistant epilepsy, with a median of 5 prior ASMs, a baseline seizure frequency of 12.75 per month, and 51.3% using 3 concomitant ASMs All doses administered once daily with food and no titration. ASM, antiseizure medication; AZK, azetukalner; IQR, interquartile range; PBO, placebo MARCH 2026 12
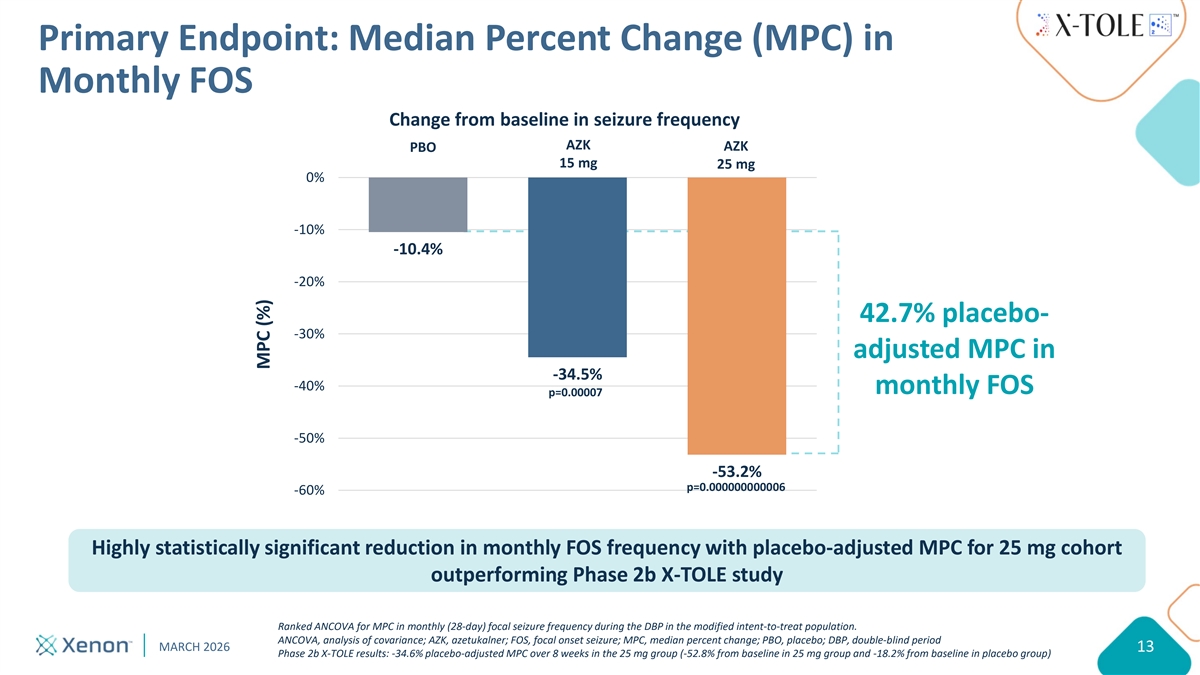
Primary Endpoint: Median Percent Change (MPC) in Monthly FOS Change from baseline in seizure frequency AZK AZK PBO 15 mg 25 mg 0% -10% -10.4% -20% 42.7% placebo- -30% adjusted MPC in -34.5% -40% monthly FOS p=0.00007 -50% -53.2% p=0.000000000006 -60% Highly statistically significant reduction in monthly FOS frequency with placebo-adjusted MPC for 25 mg cohort outperforming Phase 2b X-TOLE study Ranked ANCOVA for MPC in monthly (28-day) focal seizure frequency during the DBP in the modified intent-to-treat population. ANCOVA, analysis of covariance; AZK, azetukalner; FOS, focal onset seizure; MPC, median percent change; PBO, placebo; DBP, double-blind period MARCH 2026 13 Phase 2b X-TOLE results: -34.6% placebo-adjusted MPC over 8 weeks in the 25 mg group (-52.8% from baseline in 25 mg group and -18.2% from baseline in placebo group) MPC (%)
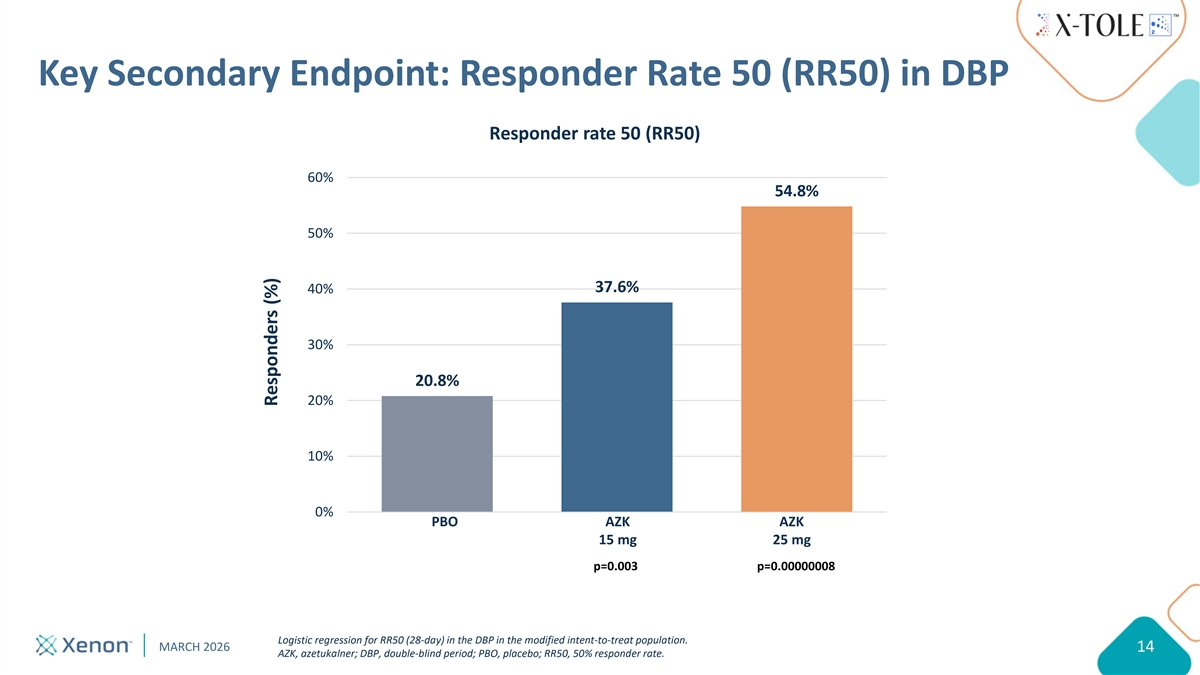
Key Secondary Endpoint: Responder Rate 50 (RR50) in DBP Responder rate 50 (RR50) 60% 54.8% 50% 37.6% 40% 30% 20.8% 20% 10% 0% PBO AZK AZK Placebo AZK (15mg) AZK (25mg) 15 mg 25 mg p=0.003 p=0.00000008 Logistic regression for RR50 (28-day) in the DBP in the modified intent-to-treat population. MARCH 2026 14 AZK, azetukalner; DBP, double-blind period; PBO, placebo; RR50, 50% responder rate. Responders (%)
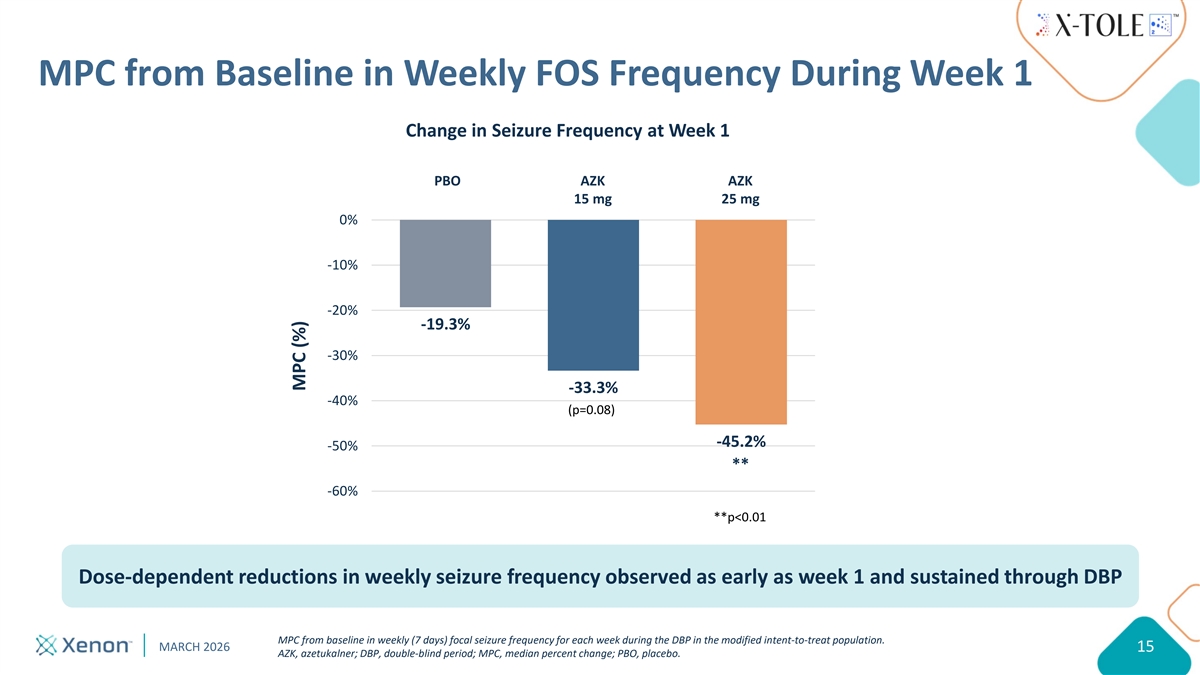
MPC from Baseline in Weekly FOS Frequency During Week 1 Change in Seizure Frequency at Week 1 PBO AZK AZK 15 mg 25 mg 0% -10% -20% -19.3% -30% -33.3% -40% (p=0.08) -45.2% -50% ** -60% **p<0.01 Dose-dependent reductions in weekly seizure frequency observed as early as week 1 and sustained through DBP MPC from baseline in weekly (7 days) focal seizure frequency for each week during the DBP in the modified intent-to-treat population. MARCH 2026 15 AZK, azetukalner; DBP, double-blind period; MPC, median percent change; PBO, placebo. MPC (%)
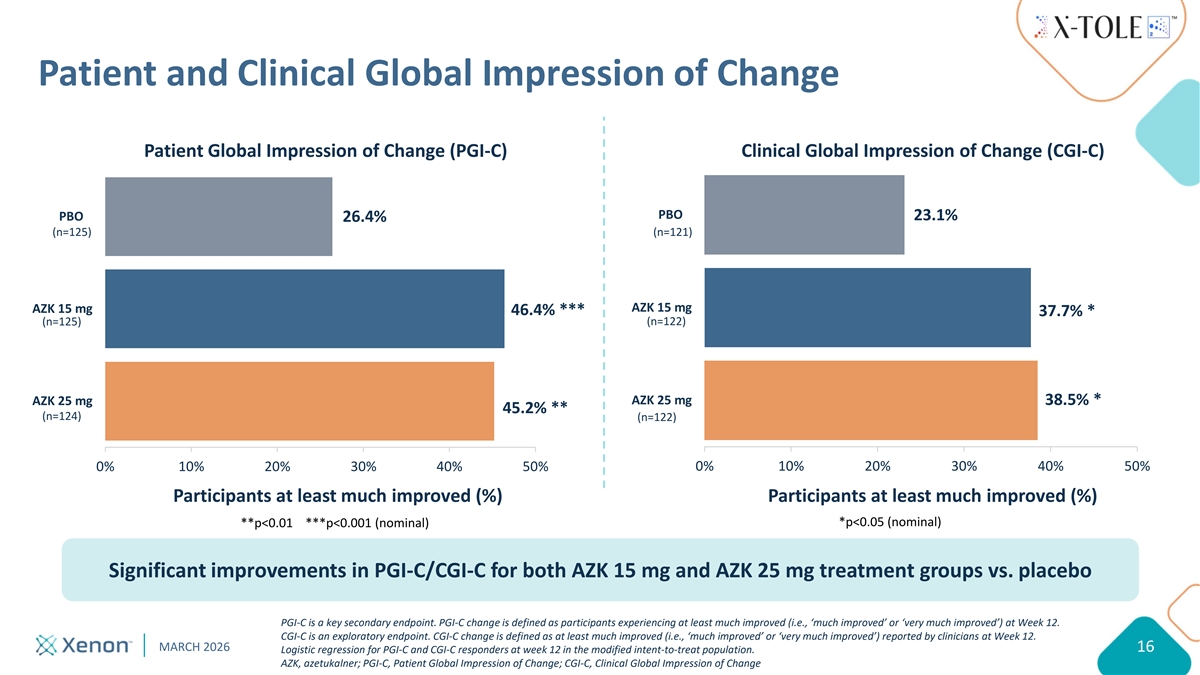
Patient and Clinical Global Impression of Change Patient Global Impression of Change (PGI-C) Clinical Global Impression of Change (CGI-C) PBO 23.1% PBO 26.4% (n=125) (n=121) AZK 15 mg AZK 15 mg 46.4% *** 37.7% * (n=122) (n=125) AZK 25 mg AZK 25 mg 38.5% * 45.2% ** (n=124) (n=122) 0% 10% 20% 30% 40% 50% 0% 10% 20% 30% 40% 50% Participants at least much improved (%) Participants at least much improved (%) *p<0.05 (nominal) **p<0.01 ***p<0.001 (nominal) Significant improvements in PGI-C/CGI-C for both AZK 15 mg and AZK 25 mg treatment groups vs. placebo PGI-C is a key secondary endpoint. PGI-C change is defined as participants experiencing at least much improved (i.e., ‘much improved’ or ‘very much improved’) at Week 12. CGI-C is an exploratory endpoint. CGI-C change is defined as at least much improved (i.e., ‘much improved’ or ‘very much improved’) reported by clinicians at Week 12. MARCH 2026 16 Logistic regression for PGI-C and CGI-C responders at week 12 in the modified intent-to-treat population. AZK, azetukalner; PGI-C, Patient Global Impression of Change; CGI-C, Clinical Global Impression of Change
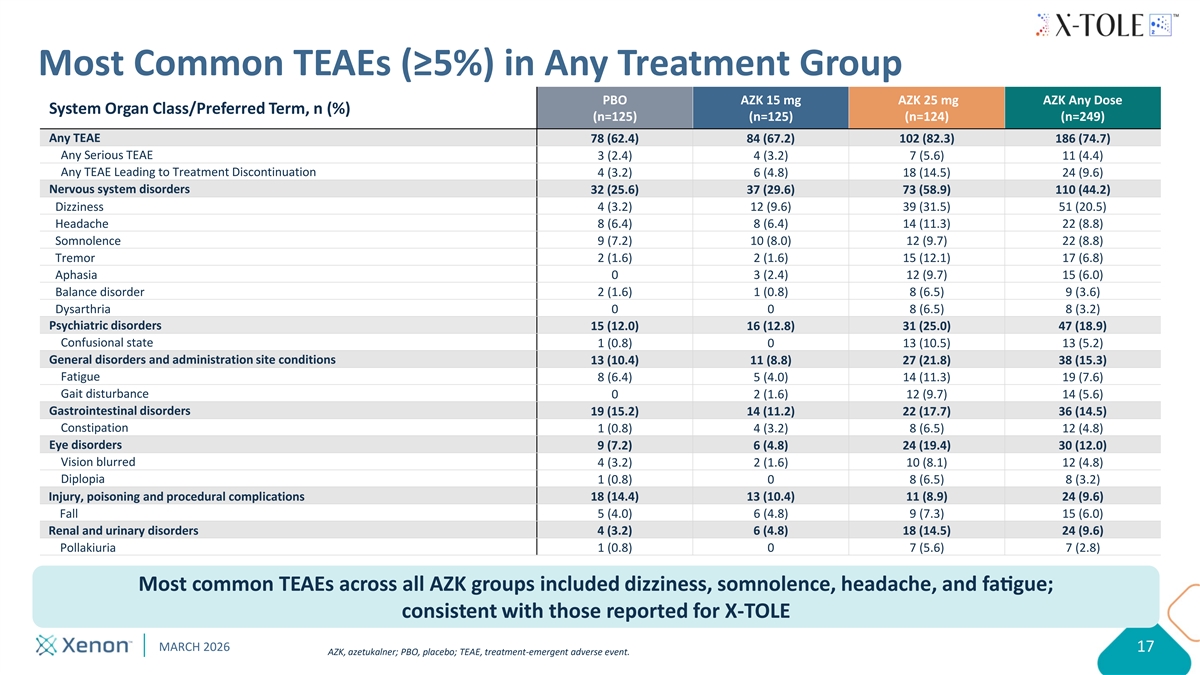
Most Common TEAEs (≥5%) in Any Treatment Group PBO AZK 15 mg AZK 25 mg AZK Any Dose System Organ Class/Preferred Term, n (%) (n=125) (n=125) (n=124) (n=249) Any TEAE 78 (62.4) 84 (67.2) 102 (82.3) 186 (74.7) Any Serious TEAE 3 (2.4) 4 (3.2) 7 (5.6) 11 (4.4) Any TEAE Leading to Treatment Discontinuation 4 (3.2) 6 (4.8) 18 (14.5) 24 (9.6) Nervous system disorders 32 (25.6) 37 (29.6) 73 (58.9) 110 (44.2) Dizziness 4 (3.2) 12 (9.6) 39 (31.5) 51 (20.5) Headache 8 (6.4) 8 (6.4) 14 (11.3) 22 (8.8) Somnolence 9 (7.2) 10 (8.0) 12 (9.7) 22 (8.8) Tremor 2 (1.6) 2 (1.6) 15 (12.1) 17 (6.8) Aphasia 0 3 (2.4) 12 (9.7) 15 (6.0) Balance disorder 2 (1.6) 1 (0.8) 8 (6.5) 9 (3.6) Dysarthria 0 0 8 (6.5) 8 (3.2) Psychiatric disorders 15 (12.0) 16 (12.8) 31 (25.0) 47 (18.9) Confusional state 1 (0.8) 0 13 (10.5) 13 (5.2) General disorders and administration site conditions 13 (10.4) 11 (8.8) 27 (21.8) 38 (15.3) Fatigue 8 (6.4) 5 (4.0) 14 (11.3) 19 (7.6) Gait disturbance 0 2 (1.6) 12 (9.7) 14 (5.6) Gastrointestinal disorders 19 (15.2) 14 (11.2) 22 (17.7) 36 (14.5) Constipation 1 (0.8) 4 (3.2) 8 (6.5) 12 (4.8) Eye disorders 9 (7.2) 6 (4.8) 24 (19.4) 30 (12.0) Vision blurred 4 (3.2) 2 (1.6) 10 (8.1) 12 (4.8) Diplopia 1 (0.8) 0 8 (6.5) 8 (3.2) Injury, poisoning and procedural complications 18 (14.4) 13 (10.4) 11 (8.9) 24 (9.6) Fall 5 (4.0) 6 (4.8) 9 (7.3) 15 (6.0) Renal and urinary disorders 4 (3.2) 6 (4.8) 18 (14.5) 24 (9.6) Pollakiuria 1 (0.8) 0 7 (5.6) 7 (2.8) Most common TEAEs across all AZK groups included dizziness, somnolence , headache , and fague; consistent with those reported for X-TOLE MARCH 2026 17 AZK, azetukalner; PBO, placebo; TEAE, treatment-emergent adverse event.

Serious Adverse Events and Additional Safety Findings Serious Adverse Events Additional Safety Findings: No severe allergic rashes (SJS, DRESS) occurred Incidence of SAEs was low and similar across AZK groups: No signals of retinal pigment epithelium or macular abnormalities PBO (2.4%) No signals of cardiovascular events AZK 15 mg (3.2%) No meaningful weight gain occurred AZK 25 mg (5.6%) No deaths occurred Four non-serious TEAEs of urinary retention were reported SAEs reported in >1 participant: Dysarthria (2) – One participant in PBO and two in 25 mg with no dose reduction Tremor (2) – One participant in 15 mg was hospitalized for a psychiatric event, which Confusional state (2) included catheterization and discontinuation Fall (2) All reported from the AZK 25 mg group AZK, azetukalner; PBO, placebo; TEAE, treatment-emergent adverse event; SJS, Stevens-Johnson Syndrome; DRESS, Drug Reaction with Eosinophilia and Systemic Symptoms. MARCH 2026 18

X-TOLE2 Summary of Safety and Tolerability Summary of Safety and Tolerability Treatment-Emergent Adverse Events Most common TEAEs across all TEAEs resulted in AZK groups included: discontinuation in: AZK was generally well tolerated with a dose-dependent incidence of TEAEs consistent with X-TOLE Dizziness (20.5%) 3.2% in PBO Somnolence (8.8%) 4.8% in AZK 15 mg Incidence of TEAEs for AZK 15 mg was similar to PBO Headache (8.8%) 14.5% in AZK 25 mg Fague (7.6%) Most common TEAEs across all AZK groups were consistent with those reported for X-TOLE Most common TEAEs leading to discontinuation across No individual TEAE led to discontinuation in all AZK groups: >5% of participants Dizziness (3.2%) Gait disturbance (1.2%) Incidence of SAEs was low and similar across AZK groups Headache (1.6%) Coordination abnormal (1.2%) Fatigue (1.6%) Speech disorder (1.2%) AZK, azetukalner; PBO, placebo; TEAE, treatment-emergent adverse event; SAE, serious adverse event. MARCH 2026 19
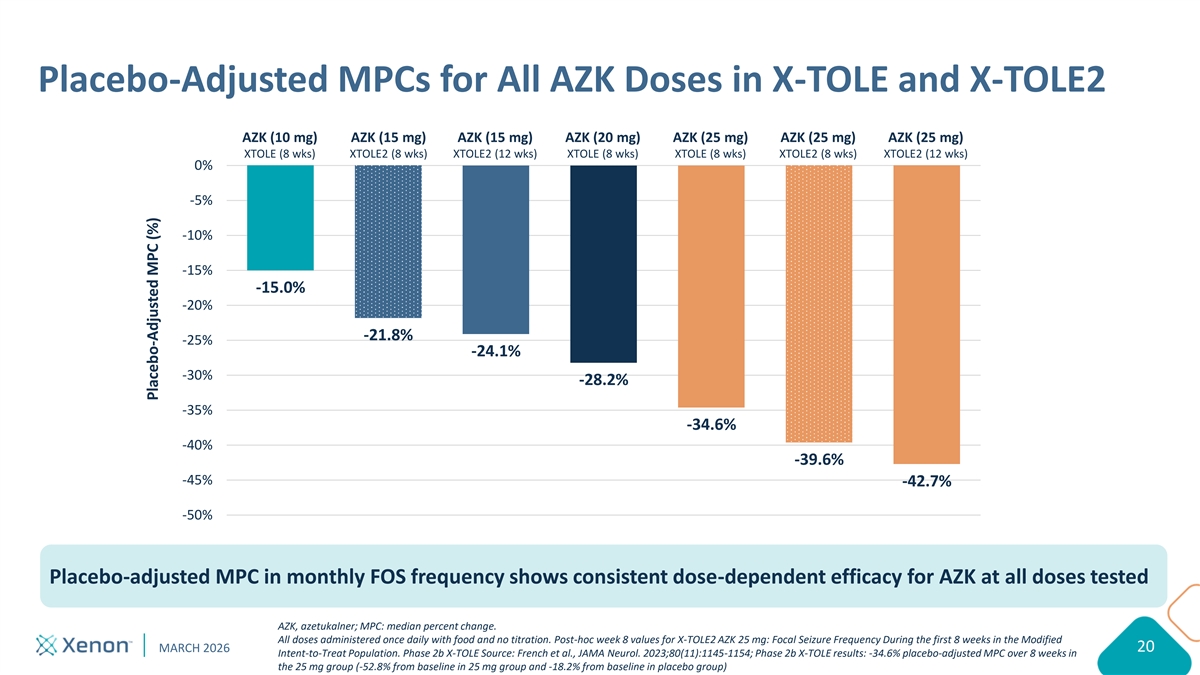
Keep content within provided margin guidelines: 12.5” width Please do not move or change the size of the Placebo-Adjusted MPCs for All AZK Doses in X-TOLE and X-TOLE2 Title box Use eyedropper tool to select colors from the expanded color palette: AZK (10 mg) AZK (15 mg) AZK (15 mg) AZK (20 mg) AZK (25 mg) AZK (25 mg) AZK (25 mg) XTOLE (8 wks) XTOLE2 (8 wks) XTOLE2 (12 wks) XTOLE (8 wks) XTOLE (8 wks) XTOLE2 (8 wks) XTOLE2 (12 wks) 0% -5% -10% -15% -15.0% -20% Keep content -21.8% -25% within provided -24.1% margin guidelines: -30% -28.2% 5.5” height -35% -34.6% -40% -39.6% -45% -42.7% -50% Placebo-adjusted MPC in monthly FOS frequency shows consistent dose-dependent efficacy for AZK at all doses tested AZK, azetukalner; MPC: median percent change. All doses administered once daily with food and no titration. Post-hoc week 8 values for X-TOLE2 AZK 25 mg: Focal Seizure Frequency During the first 8 weeks in the Modified MARCH 2026 20 Intent-to-Treat Population. Phase 2b X-TOLE Source: French et al., JAMA Neurol. 2023;80(11):1145-1154; Phase 2b X-TOLE results: -34.6% placebo-adjusted MPC over 8 weeks in the 25 mg group (-52.8% from baseline in 25 mg group and -18.2% from baseline in placebo group) Please try to avoid covering the logo Placebo-Adjusted MPC (%)

Summary of X-TOLE2 Topline Results Study met primary endpoint in both dose groups, including -53.2% MPC from baseline in monthly FOS frequency with 25 mg dose vs. 10.4% for placebo (p=0.000000000006) X-TOLE2 outperformed X-TOLE study with a placebo-adjusted MPC of -42.7% in the 25 mg group in X-TOLE2 compared to -34.6% in X-TOLE Azetukalner was generally well-tolerated with a safety profile consistent with X-TOLE X-TOLE results: -34.6% placebo-adjusted MPC at Week 8 in the 25 mg group (-52.8% from baseline in 25 mg group and -18.2% from baseline in placebo group) MARCH 2026 21

Moving Toward Commercialization Darren Cline Chief Commercial Officer
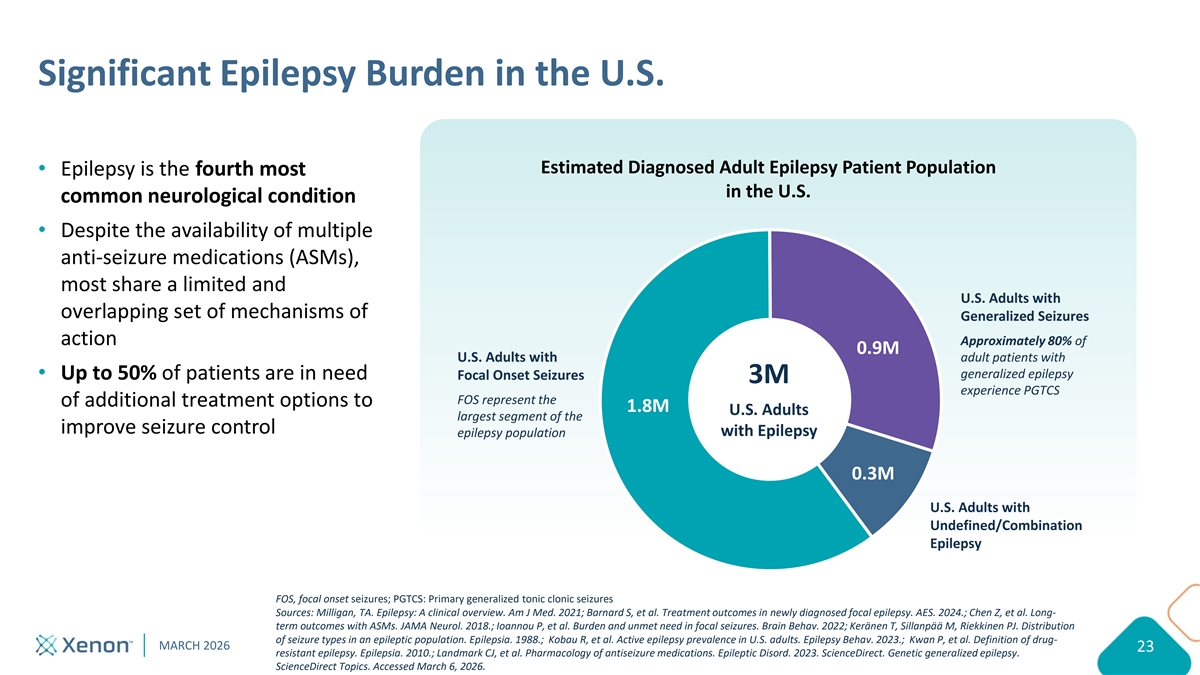
Significant Epilepsy Burden in the U.S. Estimated Diagnosed Adult Epilepsy Patient Population Epilepsy is the fourth most in the U.S. common neurological condition Despite the availability of multiple anti-seizure medications (ASMs), most share a limited and U.S. Adults with overlapping set of mechanisms of Generalized Seizures action Approximately 80% of 0.9M adult patients with U.S. Adults with generalized epilepsy Up to 50% of patients are in need Focal Onset Seizures 3M experience PGTCS FOS represent the of additional treatment options to 1.8M U.S. Adults largest segment of the improve seizure control with Epilepsy epilepsy population 0.3M U.S. Adults with Undefined/Combination Epilepsy FOS, focal onset seizures; PGTCS: Primary generalized tonic clonic seizures Sources: Milligan, TA. Epilepsy: A clinical overview. Am J Med. 2021; Barnard S, et al. Treatment outcomes in newly diagnosed focal epilepsy. AES. 2024.; Chen Z, et al. Long- term outcomes with ASMs. JAMA Neurol. 2018.; Ioannou P, et al. Burden and unmet need in focal seizures. Brain Behav. 2022; Keränen T, Sillanpää M, Riekkinen PJ. Distribution of seizure types in an epileptic population. Epilepsia. 1988.; Kobau R, et al. Active epilepsy prevalence in U.S. adults. Epilepsy Behav. 2023.; Kwan P, et al. Definition of drug- MARCH 2026 23 resistant epilepsy. Epilepsia. 2010.; Landmark CJ, et al. Pharmacology of antiseizure medications. Epileptic Disord. 2023. ScienceDirect. Genetic generalized epilepsy. ScienceDirect Topics. Accessed March 6, 2026.

Market Research Suggests Both Epilepsy Specialists and General Neurologists May Find Value in Potential Azetukalner Attributes 1 1 Epilepsy Specialists General Neurologists Perceptions of Unfavorable Favorable Unfavorable Favorable “Product X” 2 Novel Mechanism of Action Ease of Use “Product X” Dosing Flexibility to Balance Efficacy and Safety Manageable Safety Profile Value Drivers Branded ASM-like Efficacy Fast Onset of Action The MoA is interesting to me. I The side effect profile of Product X Product X is distinguished from Product X has strong efficacy, great onset and [manageable] side like to choose something that is is tolerable. I also don’t have to other brands from a safety effects, it would be a 2/3L agent. different than what my patients wait to get my patients to an perspective, along with the lack – General Neurologist have received. This product gives effective dose, but have some of titration and rapid onset. me more flexibility. flexibility in the dose I choose. – General Neurologist –Epilepsy Specialist –Epilepsy Specialist 1. Research sample includes 14 general neurologists and 4 epileptologists. 2. Lack of titration schedule and limited DDIs drove physician perceptions of Product X as an “easy to MARCH 2026 24 use” treatment option with simple onboarding. Source: Physician Interviews; ClearView Analysis. Market research referenced herein is preliminary and qualitative in nature.
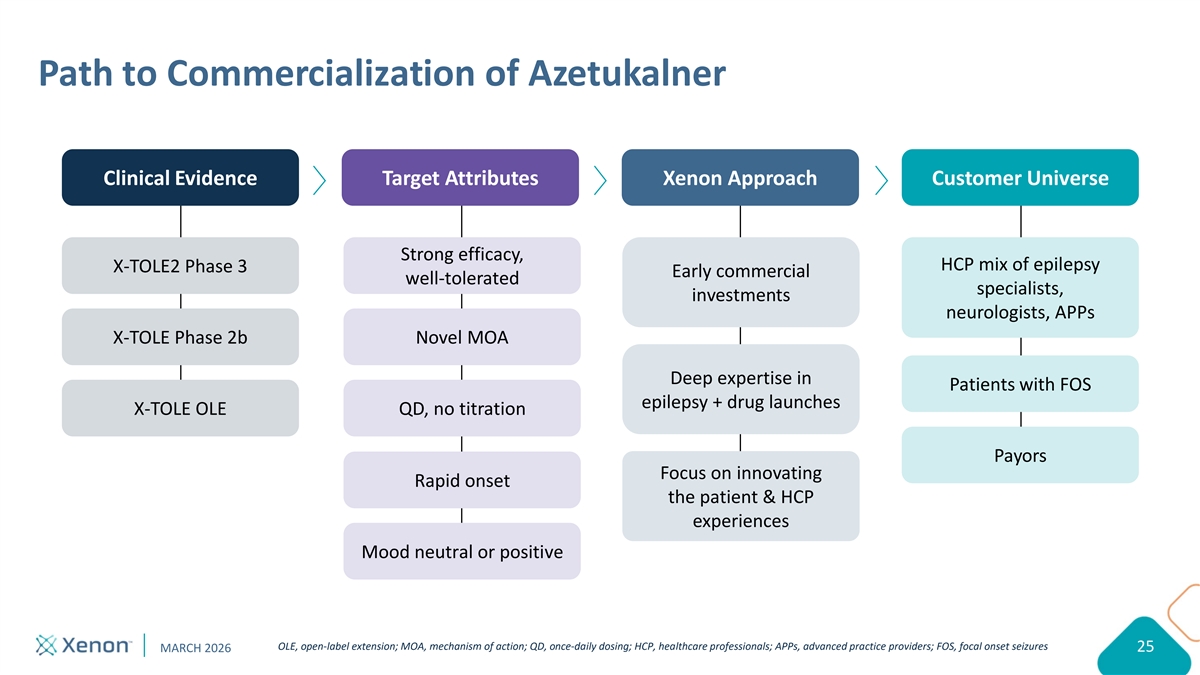
Path to Commercialization of Azetukalner Clinical Evidence Target Attributes Xenon Approach Customer Universe Strong efficacy, HCP mix of epilepsy X-TOLE2 Phase 3 Early commercial well-tolerated specialists, investments neurologists, APPs X-TOLE Phase 2b Novel MOA Deep expertise in Patients with FOS epilepsy + drug launches X-TOLE OLE QD, no titration Payors Focus on innovating Rapid onset the patient & HCP experiences Mood neutral or positive OLE, open-label extension; MOA, mechanism of action; QD, once-daily dosing; HCP, healthcare professionals; APPs, advanced practice providers; FOS, focal onset seizures MARCH 2026 25

Looking Toward the Future Ian Mortimer President & Chief Executive Officer

X-TOLE2 Results Summary Positive Phase 3 study results support an anticipated NDA submission in Q3 2026 PRIMARY ENDPOINT WAS MET: Highly statistically significant, dose-dependent reduction from baseline in median monthly FOS frequency (MPC) over the 12-week treatment period vs. placebo Responder rate 50: Statistically significant, dose-dependent (15 and 25 mg) increase in number of responders with >50% reduction in monthly FOS frequency Onset of efficacy: Rapid onset of response as assessed by statistically significant MPC achieved within one week for AZK 25 mg vs. placebo Overall health status: Significant improvements in PGI-C/CGI-C for both 15 and 25 mg treatment groups vs. placebo Safety and tolerability: AZK profile consistent with Phase 2b X-TOLE study AZK, azetukalner; PGI-C, Patient Global Impression of Change; CGI-C, Clinical Global Impression of Change; MPC, median percent change; FOS, focal onset seizure MARCH 2026 27

Thank you to our partners in the epilepsy community, including patient advocates, clinicians and study participants. Hadley, living with epilepsy

POSITIVE TOPLINE DATA Azetukalner in Focal Onset Seizures

For more information INVESTORS@XENON-PHARMA.COM