

This presentation of Recursion Pharmaceuticals, Inc. (“Recursion,” “we,” “us,” or “our”) and any accompanying discussion contain statements that are not historical facts may be considered forward-looking statements under federal securities laws and may be identified by words such as “anticipates,” “believes,” “estimates,” “expects,” “intends,” “plans,” “potential,” “predicts,” “projects,” “seeks,” “should,” “will,” or words of similar meaning and include, but are not limited to, statements regarding the impact of the acceptance of the fifth milestone by Sanofi on future developments and potential treatments; the impact of FAP trial on the Recursion OS and other clinical and preclinical programs; financial position, cash runway, and ability to reduce our cash expense; our ability to use AI to translate complex science into medicines faster and better; Recursion’s OS industrializing first- and best-in-class drug discovery; our ability to industrialize clinical development and the effect of doing so on clinical trial outcomes; the occurrence or realization of potential milestones and their potential timing or amounts; current and future preclinical and clinical studies, including timelines for enrollment in studies, data readouts, progression toward IND- enabling and other potential studies, and engagement with the FDA; advancements of and other decisions regarding our pipeline, partnerships, and data strategies; the potential size of the market opportunity for our drug candidates; outcomes and benefits from licenses, partnerships and collaborations, including option exercises by partners; the initiation, timing, progress, results, and cost of our research and development programs; advancements of our Recursion OS; and many others. Other important factors and information are contained in Recursion’s most recent Annual Report on Form 10-K, and the Company’s other filings with the U.S. Securities and Exchange Commission (the “SEC”), which can be accessed at https://ir.recursion.com, or www.sec.gov. All forward-looking statements are qualified by these cautionary statements and apply only as of the date they are made. Recursion does not undertake any obligation to update any forward- looking statement, whether as a result of new information, future events or otherwise. Certain information contained in this presentation relates to or is based on studies, publications, surveys and other data obtained from third-party sources and the company’s own internal estimates and research. While the company believes these third-party sources to be reliable as of the date of this presentation, it has not independently verified, and makes no representation as to the adequacy, fairness, accuracy or completeness of, any information obtained from third- party sources. In addition, all of the market data included in this presentation involves a number of assumptions and limitations, and there can be no guarantee as to the accuracy or reliability of such assumptions. Finally, while the company believes its own internal research is reliable, such research has not been verified by any independent source. Information contained in, or that can be accessed through our website is not a part of and is not incorporated into this presentation. Cross-trial or cross-candidate comparisons against other clinical trials and other drug candidates are not based on head-to-head studies and are presented for informational purposes; comparisons are based on publicly available information for other clinical trials and other drug candidates. Any non-Recursion logos or trademarks included herein are the property of the owners thereof and are used for reference purposes only. Important Information 2
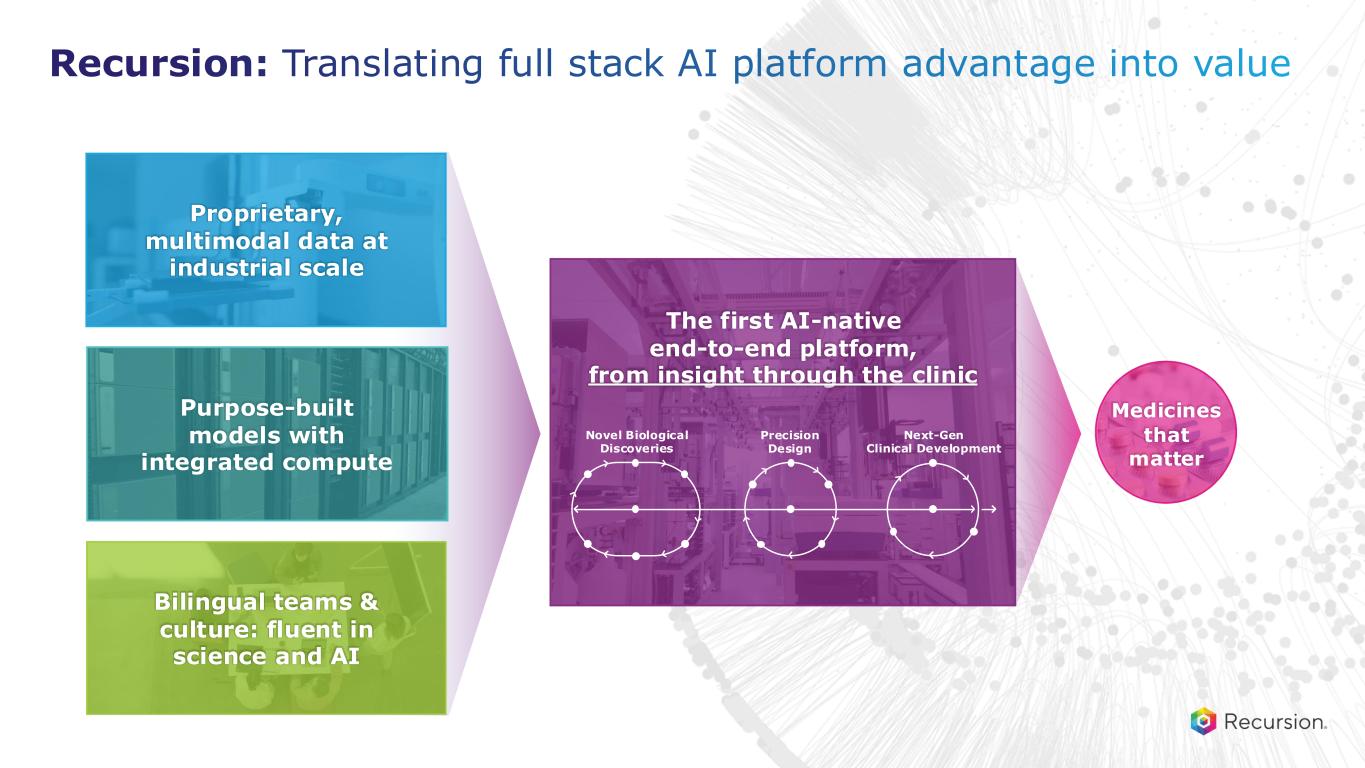
The first AI-native end-to-end platform, from insight through the clinic Precision Design Novel Biological Discoveries Next-Gen Clinical Development Bilingual teams & culture: fluent in science and AI Purpose-built models with integrated compute Proprietary, multimodal data at industrial scale Recursion: Translating full stack AI platform advantage into value Medicines that matter

1. Includes preclinical programs that are expected to enter the clinic within the next 18 months 2. Milestones: Potential Roche and Genentech and Sanofi milestones per small molecule program. Royalties: Recursion is eligible for tiered royalties up to high single digits (Roche and Genentech) and up to double digits (Sanofi) 3. Cash, cash equivalents and restricted cash as of December 31, 2025 4. Runway guidance includes risk-adjusted cash inflows from partnerships Recursion: Progress, by the numbers Wholly owned1~5 CLINICAL DEVELOPMENT ✓REC-4881: FIRST AI-enabled clinical proof-of- concept with potential for first-in-class for FAP ✓>$500M in upfront & milestones achieved • >$300M in potential milestones + tiered royalties per program2 • Several high value opportunities Partnered Wholly owned ~15 DISCOVERY The first AI-native end-to-end platform, from idea through the clinic $754 million in YE 2025 cash3, providing expected runway into early 20284 4
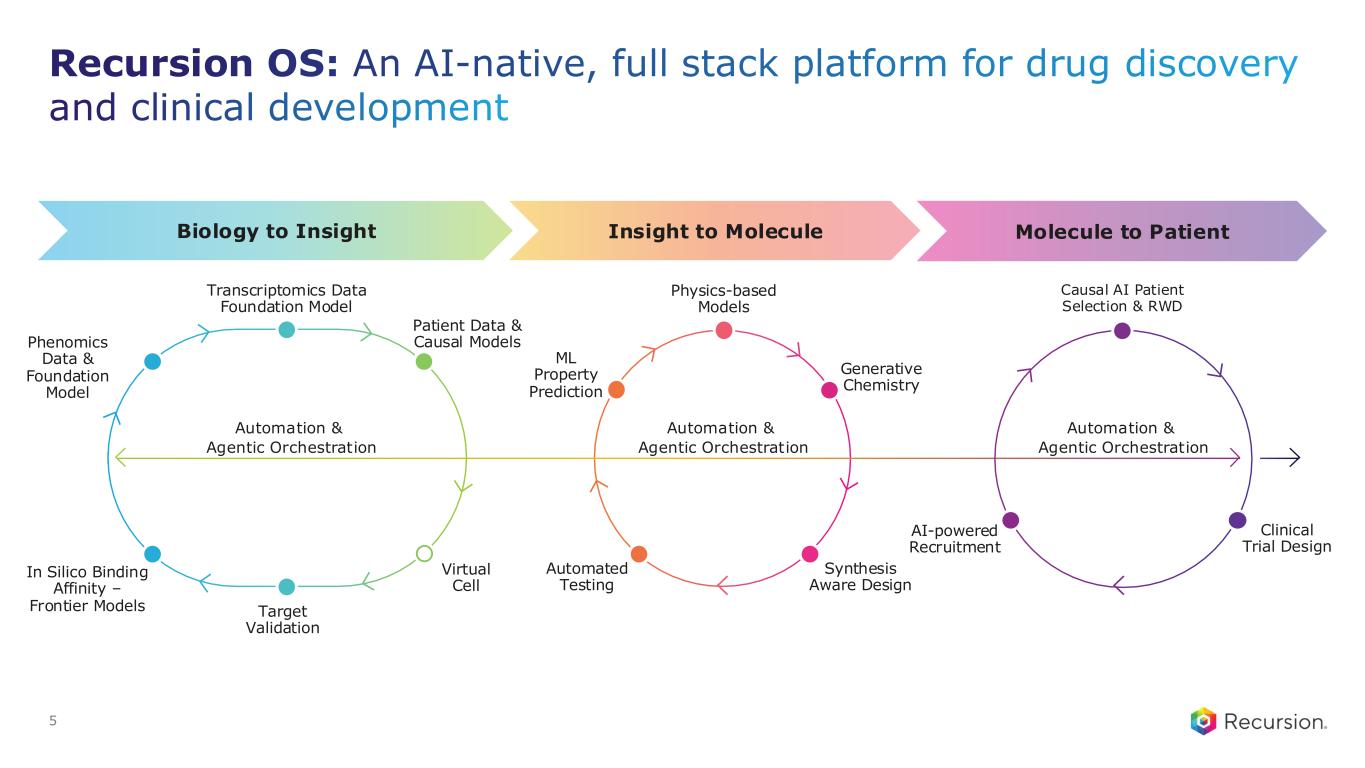
Recursion OS: An AI-native, full stack platform for drug discovery and clinical development 5 In Silico Binding Affinity – Frontier Models Automated Testing Synthesis Aware Design Physics-based Models ML Property Prediction Transcriptomics Data Foundation Model Virtual Cell Target Validation Phenomics Data & Foundation Model Causal AI Patient Selection & RWD Insight to MoleculeBiology to Insight Molecule to Patient Generative Chemistry Automation & Agentic Orchestration Automation & Agentic Orchestration Automation & Agentic Orchestration AI-powered Recruitment Clinical Trial Design Patient Data & Causal Models
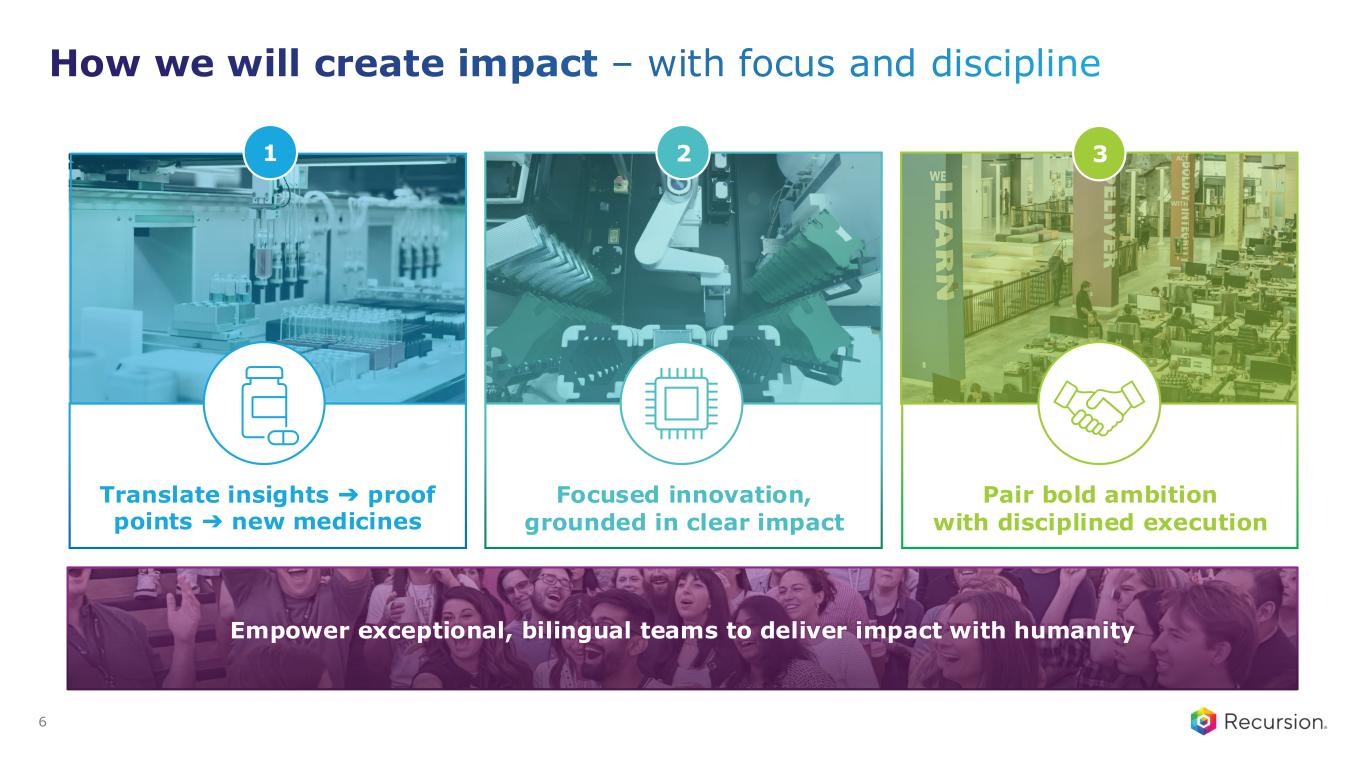
How we will create impact – with focus and discipline 6 Translate insights ➔ proof points ➔ new medicines Focused innovation, grounded in clear impact Empower exceptional, bilingual teams to deliver impact with humanity Pair bold ambition with disciplined execution 1 2 3
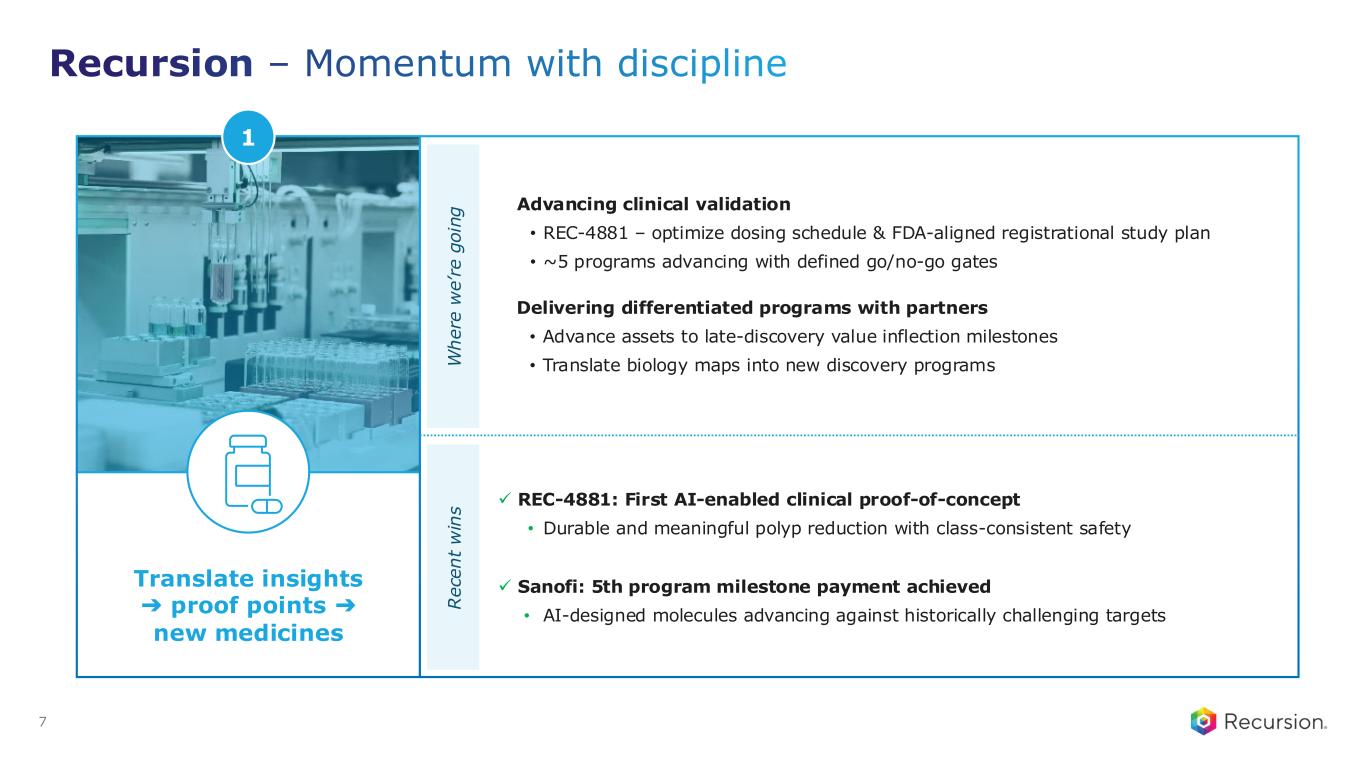
Recursion – Momentum with discipline 7 Advancing clinical validation • REC-4881 – optimize dosing schedule & FDA-aligned registrational study plan • ~5 programs advancing with defined go/no-go gates Delivering differentiated programs with partners • Advance assets to late-discovery value inflection milestones • Translate biology maps into new discovery programsW h e re w e ’r e g o in g ✓ REC-4881: First AI-enabled clinical proof-of-concept • Durable and meaningful polyp reduction with class-consistent safety ✓ Sanofi: 5th program milestone payment achieved • AI-designed molecules advancing against historically challenging targets R e ce n t w in s Translate insights ➔ proof points ➔ new medicines 1
Recursion – Momentum with discipline 8 W h e re w e ’r e g o in g R e ce n t w in s ✓ Precision AI-molecule design delivering differentiated assets • ~90% fewer compounds synthesized and 2x faster candidate advancement vs industry ✓ Roche and Genentech: First-in-class CRISPR phenomap • Whole genome human neuronal and microglial biology maps ✓ ClinTech: Contextualized FAP efficacy signal • High quality registry + AI-enabled RWE strengthened interpretation of single-arm data Mature, actionable portfolio of high-quality targets • Integrate omics and patient data with purpose-built models Generative drug design at scale • Next-gen models and agentic systems for design AI-powered Clinical Development at scale • Increased automation and in silico trial design and execution Focused innovation, grounded in clear impact 2

Recursion – Momentum with discipline 9 ✓ ~35% reduction (~$200M) in pro forma operating expenses YoY • Driven by sharper portfolio focus, G&A optimization, improved platform efficiency ✓ Runway extended to early 2028 W h e re w e ’r e g o in g R e ce n t w in s Pair bold ambition with disciplined execution Relentless capital discipline • Portfolio decisions anchored in automated Target Product Profiles • Objective, data-driven go/no-go gates • Platform investment tied directly to measurable impact Operational leverage at scale • Deploy AI agents to compress timelines and reduce cost • Embed automation across workflows 3

Wholly Owned Clinical Pipeline Translate insights ➔ proof points ➔ new medicines
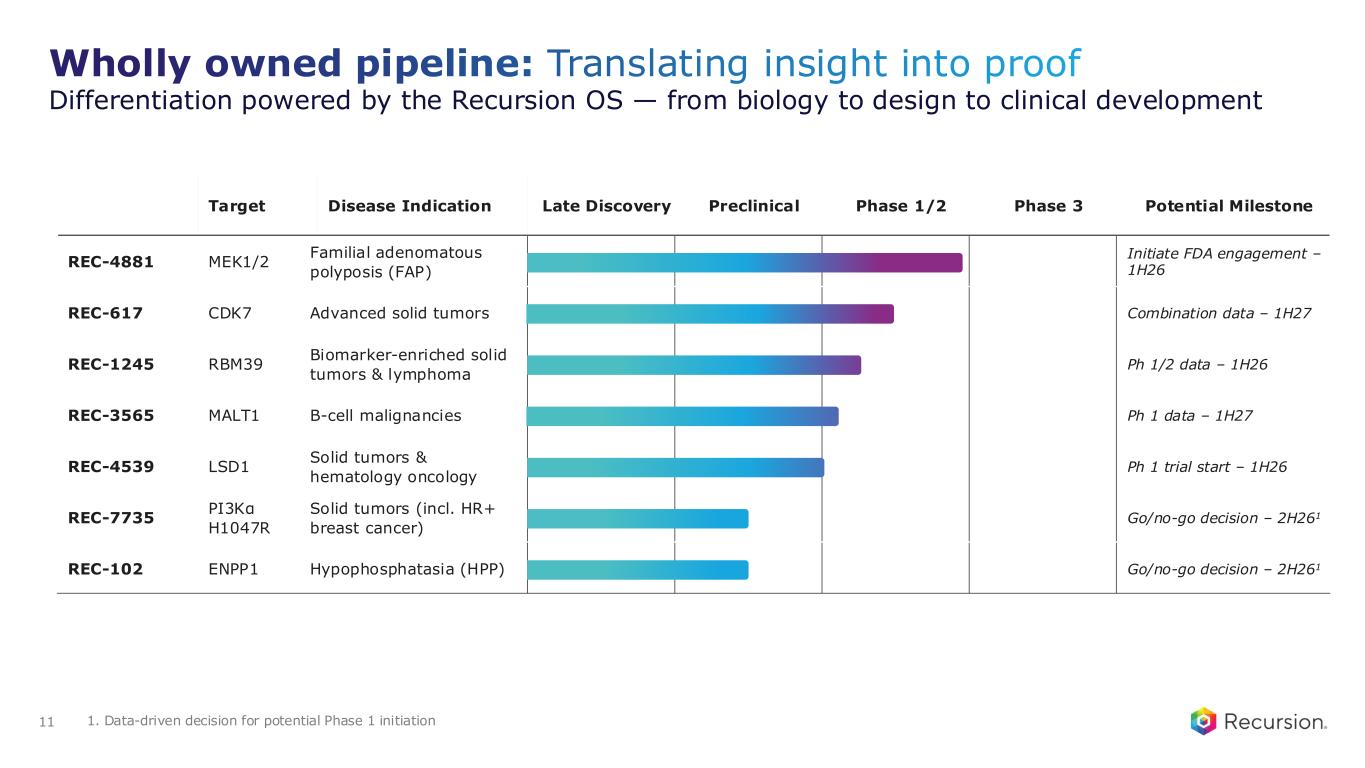
Wholly owned pipeline: Translating insight into proof Differentiation powered by the Recursion OS — from biology to design to clinical development 11 Target Disease Indication Late Discovery Preclinical Phase 1/2 Phase 3 Potential Milestone REC-4881 MEK1/2 Familial adenomatous polyposis (FAP) Initiate FDA engagement – 1H26 REC-617 CDK7 Advanced solid tumors Combination data – 1H27 REC-1245 RBM39 Biomarker-enriched solid tumors & lymphoma Ph 1/2 data – 1H26 REC-3565 MALT1 B-cell malignancies Ph 1 data – 1H27 REC-4539 LSD1 Solid tumors & hematology oncology Ph 1 trial start – 1H26 REC-7735 PI3Kα H1047R Solid tumors (incl. HR+ breast cancer) Go/no-go decision – 2H261 REC-102 ENPP1 Hypophosphatasia (HPP) Go/no-go decision – 2H261 1. Data-driven decision for potential Phase 1 initiation
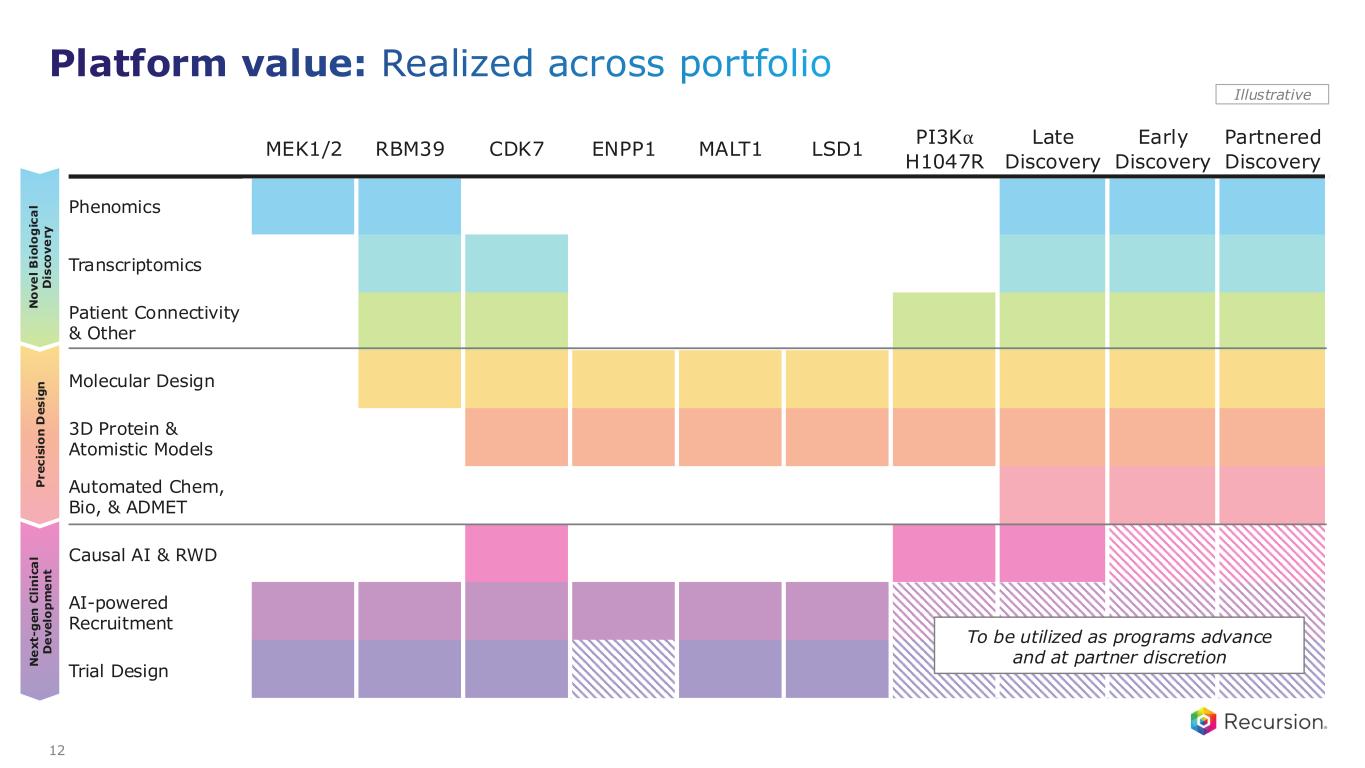
MEK1/2 RBM39 CDK7 ENPP1 MALT1 LSD1 PI3K⍺ H1047R Late Discovery Early Discovery Partnered Discovery Phenomics Transcriptomics Patient Connectivity & Other Molecular Design 3D Protein & Atomistic Models Automated Chem, Bio, & ADMET Causal AI & RWD AI-powered Recruitment Trial Design P r e c is io n D e s ig n N o v e l B io lo g ic a l D is c o v e ry N e x t- g e n C li n ic a l D e v e lo p m e n t Illustrative To be utilized as programs advance and at partner discretion 12 Platform value: Realized across portfolio
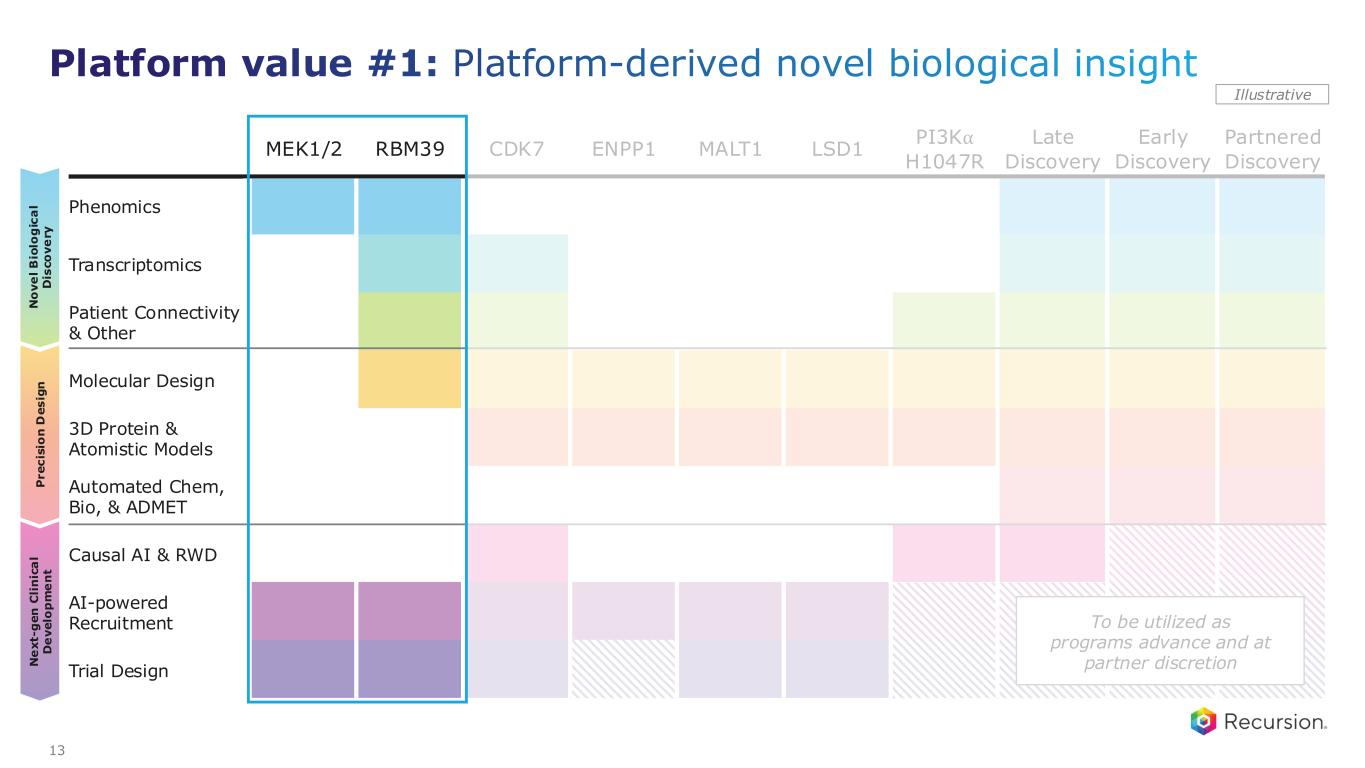
MEK1/2 RBM39 CDK7 ENPP1 MALT1 LSD1 PI3K⍺ H1047R Late Discovery Early Discovery Partnered Discovery Phenomics Transcriptomics Patient Connectivity & Other Molecular Design 3D Protein & Atomistic Models Automated Chem, Bio, & ADMET Causal AI & RWD AI-powered Recruitment Trial Design P r e c is io n D e s ig n N o v e l B io lo g ic a l D is c o v e ry N e x t- g e n C li n ic a l D e v e lo p m e n t Illustrative 13 Platform value #1: Platform-derived novel biological insight To be utilized as programs advance and at partner discretion

Platform value #1: Platform-derived novel biological insight 14 Addressable patient populations estimate based on annual US+EU5 and currently identified indications Preclinical Clinical REC-1245 | RBM39, Solid tumors & lymphoma1 Why it matters: • Synthetic-lethal vulnerability in genomically unstable cancers • ~100,000+ addressable patients RXRX Differentiation: Phenomics discovered novel MOA in “undruggable” space (CDK12) Progress: Ph 1 monotherapy dose escalation ongoing Next steps: Early Ph 1 update on safety and PK monotherapy expected 1H26 REC-4881 | MEK1/2 Familial adenomatous polyposis (FAP) • Significant unmet need, no approved pharmacotherapies • >50,000 addressable patients • Phenomics uncovered MEK1/2 inhibition as a novel MOA that rescues APC-deficient cells • Clinical POC with durable polyp burden reduction and safety profile in line with class • Initiate FDA engagement on registrational path in 1H26 • Expanding population to 18+ and optimizing dose REC-1245 | RBM39 Solid tumors & lymphoma • Synthetic-lethal vulnerability in genomically unstable cancers • >100,000 addressable patients • Phenomics discovered novel MOA in “undruggable” space (CDK12) • Ph 1 monotherapy dose escalation ongoing • Early Ph 1 update on safety and PK monotherapy expected 1H26 Why it matters RXRX differentiation Progress Next steps
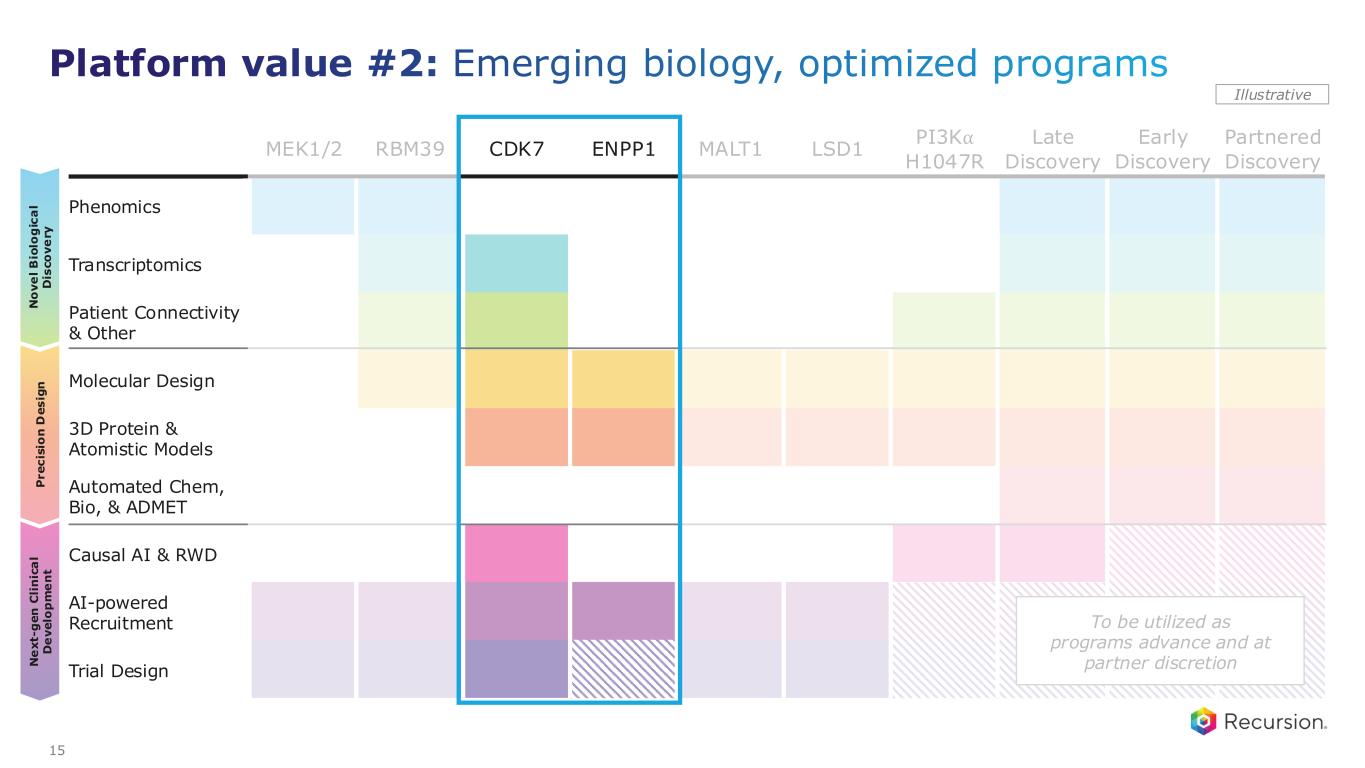
MEK1/2 RBM39 CDK7 ENPP1 MALT1 LSD1 PI3K⍺ H1047R Late Discovery Early Discovery Partnered Discovery Phenomics Transcriptomics Patient Connectivity & Other Molecular Design 3D Protein & Atomistic Models Automated Chem, Bio, & ADMET Causal AI & RWD AI-powered Recruitment Trial Design P r e c is io n D e s ig n N o v e l B io lo g ic a l D is c o v e ry N e x t- g e n C li n ic a l D e v e lo p m e n t Illustrative 15 Platform value #2: Emerging biology, optimized programs To be utilized as programs advance and at partner discretion
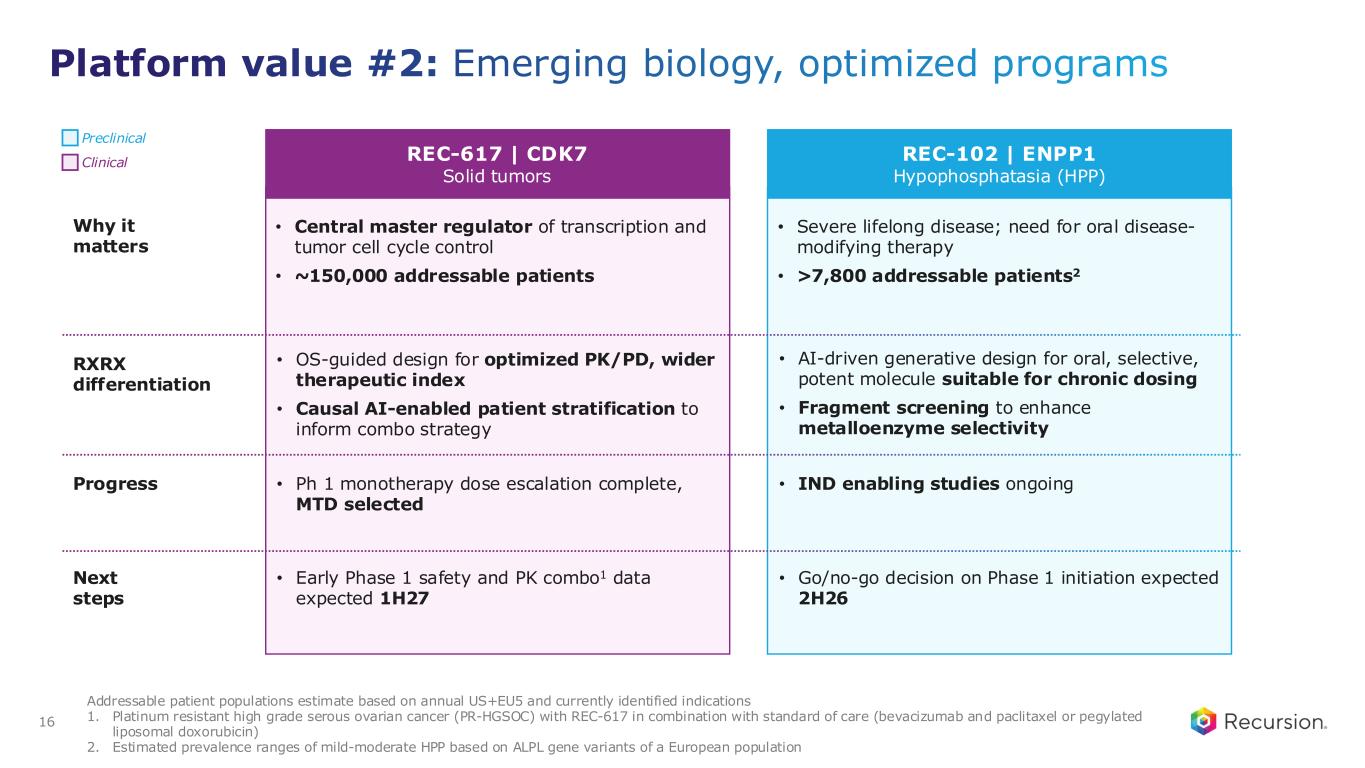
Platform value #2: Emerging biology, optimized programs 16 REC-617 | CDK7 Solid tumors • Central master regulator of transcription and tumor cell cycle control • ~150,000 addressable patients • OS-guided design for optimized PK/PD, wider therapeutic index • Causal AI-enabled patient stratification to inform combo strategy • Ph 1 monotherapy dose escalation complete, MTD selected • Early Phase 1 safety and PK combo1 data expected 1H27 REC-102 | ENPP1 Hypophosphatasia (HPP) • Severe lifelong disease; need for oral disease- modifying therapy • >7,800 addressable patients2 • AI-driven generative design for oral, selective, potent molecule suitable for chronic dosing • Fragment screening to enhance metalloenzyme selectivity • IND enabling studies ongoing • Go/no-go decision on Phase 1 initiation expected 2H26 Addressable patient populations estimate based on annual US+EU5 and currently identified indications 1. Platinum resistant high grade serous ovarian cancer (PR-HGSOC) with REC-617 in combination with standard of care (bevacizumab and paclitaxel or pegylated liposomal doxorubicin) 2. Estimated prevalence ranges of mild-moderate HPP based on ALPL gene variants of a European population Preclinical Clinical Why it matters RXRX differentiation Progress Next steps
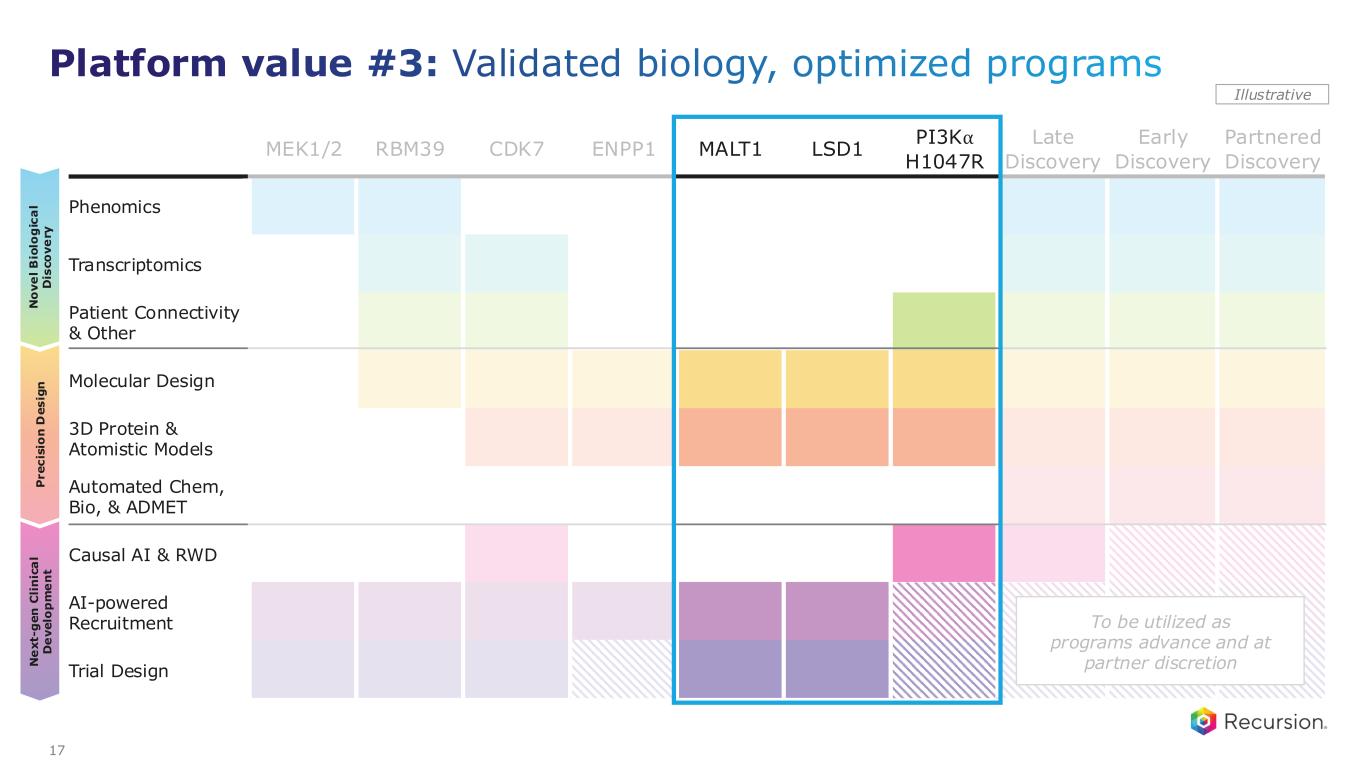
MEK1/2 RBM39 CDK7 ENPP1 MALT1 LSD1 PI3K⍺ H1047R Late Discovery Early Discovery Partnered Discovery Phenomics Transcriptomics Patient Connectivity & Other Molecular Design 3D Protein & Atomistic Models Automated Chem, Bio, & ADMET Causal AI & RWD AI-powered Recruitment Trial Design P r e c is io n D e s ig n N o v e l B io lo g ic a l D is c o v e ry N e x t- g e n C li n ic a l D e v e lo p m e n t Illustrative To be utilized as programs advance and at partner discretion 17 Platform value #3: Validated biology, optimized programs
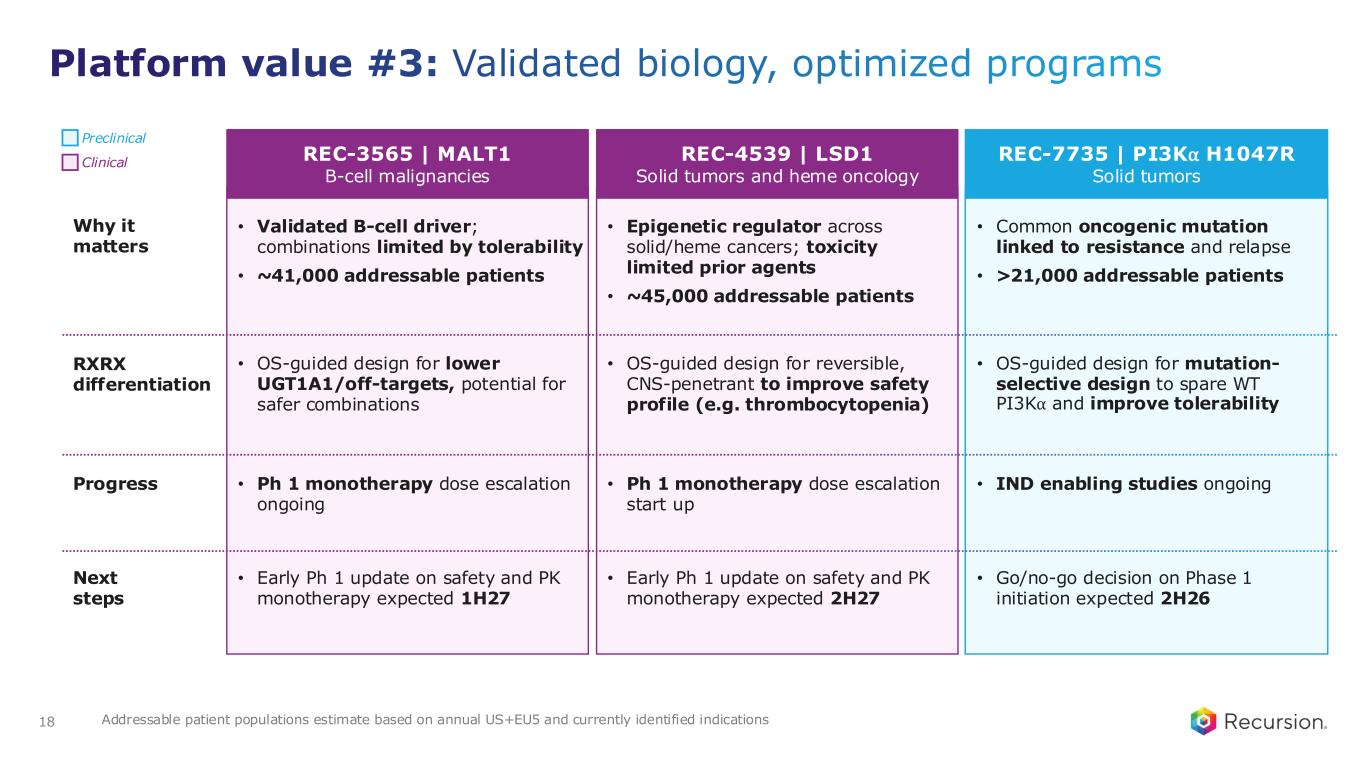
Platform value #3: Validated biology, optimized programs 18 Preclinical Clinical REC-1245 | RBM39, Solid tumors & lymphoma1 Why it matters: • Synthetic-lethal vulnerability in genomically unstable cancers • ~100,000+ addressable patients RXRX Differentiation: Phenomics discovered novel MOA in “undruggable” space (CDK12) Progress: Ph 1 monotherapy dose escalation ongoing Next steps: Early Ph 1 update on safety and PK monotherapy expected 1H26 REC-3565 | MALT1 B-cell malignancies • Validated B-cell driver; combinations limited by tolerability • ~41,000 addressable patients • OS-guided design for lower UGT1A1/off-targets, potential for safer combinations • Ph 1 monotherapy dose escalation ongoing • Early Ph 1 update on safety and PK monotherapy expected 1H27 REC-7735 | PI3K⍺ H1047R Solid tumors • Common oncogenic mutation linked to resistance and relapse • >21,000 addressable patients • OS-guided design for mutation- selective design to spare WT PI3K⍺ and improve tolerability • IND enabling studies ongoing • Go/no-go decision on Phase 1 initiation expected 2H26 Addressable patient populations estimate based on annual US+EU5 and currently identified indications REC-4539 | LSD1 Solid tumors and heme oncology • Epigenetic regulator across solid/heme cancers; toxicity limited prior agents • ~45,000 addressable patients • OS-guided design for reversible, CNS-penetrant to improve safety profile (e.g. thrombocytopenia) • Ph 1 monotherapy dose escalation start up • Early Ph 1 update on safety and PK monotherapy expected 2H27 Preclinical Clinical Why it matters RXRX differentiation Progress Next steps

REC-4881 Translate insights ➔ proof points ➔ new medicines
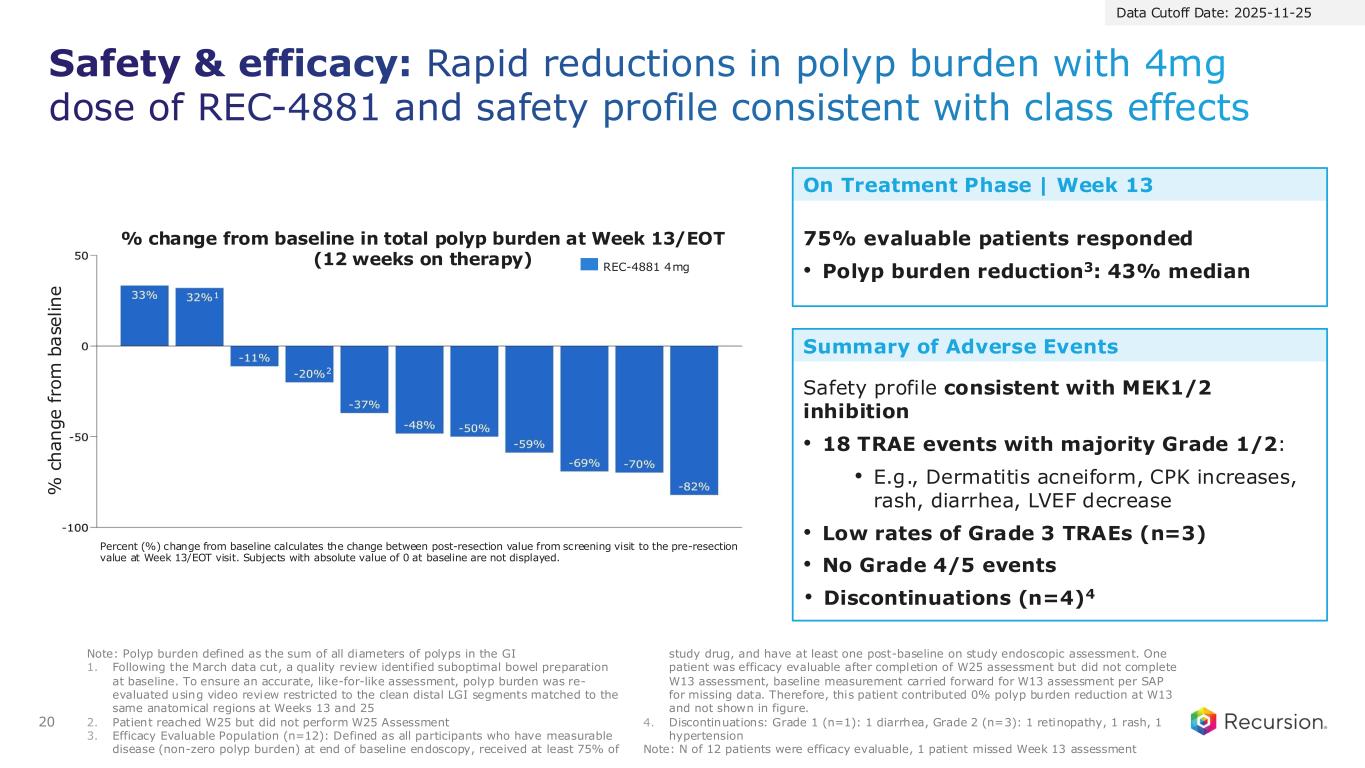
20 Safety & efficacy: Rapid reductions in polyp burden with 4mg dose of REC-4881 and safety profile consistent with class effects Note: Polyp burden defined as the sum of all diameters of polyps in the GI 1. Following the March data cut, a quality review identified suboptimal bowel preparation at baseline. To ensure an accurate, like-for-like assessment, polyp burden was re- evaluated using video review restricted to the clean distal LGI segments matched to the same anatomical regions at Weeks 13 and 25 2. Patient reached W25 but did not perform W25 Assessment 3. Efficacy Evaluable Population (n=12): Defined as all participants who have measurable disease (non-zero polyp burden) at end of baseline endoscopy, received at least 75% of study drug, and have at least one post-baseline on study endoscopic assessment. One patient was efficacy evaluable after completion of W25 assessment but did not complete W13 assessment, baseline measurement carried forward for W13 assessment per SAP for missing data. Therefore, this patient contributed 0% polyp burden reduction at W13 and not shown in figure. 4. Discontinuations: Grade 1 (n=1): 1 diarrhea, Grade 2 (n=3): 1 retinopathy, 1 rash, 1 hypertension Note: N of 12 patients were efficacy evaluable, 1 patient missed Week 13 assessment Data Cutoff Date: 2025-11-25 Percent (%) change from baseline calculates the change between post-resection value from screening visit to the pre-resection value at Week 13/EOT visit. Subjects with absolute value of 0 at baseline are not displayed. REC-4881 4mg % change from baseline in total polyp burden at Week 13/EOT (12 weeks on therapy) % c h a n g e f ro m b a se li n e 1 2 Safety profile consistent with MEK1/2 inhibition • 18 TRAE events with majority Grade 1/2: • E.g., Dermatitis acneiform, CPK increases, rash, diarrhea, LVEF decrease • Low rates of Grade 3 TRAEs (n=3) • No Grade 4/5 events • Discontinuations (n=4)4 75% evaluable patients responded • Polyp burden reduction3: 43% median Summary of Adverse Events On Treatment Phase | Week 13
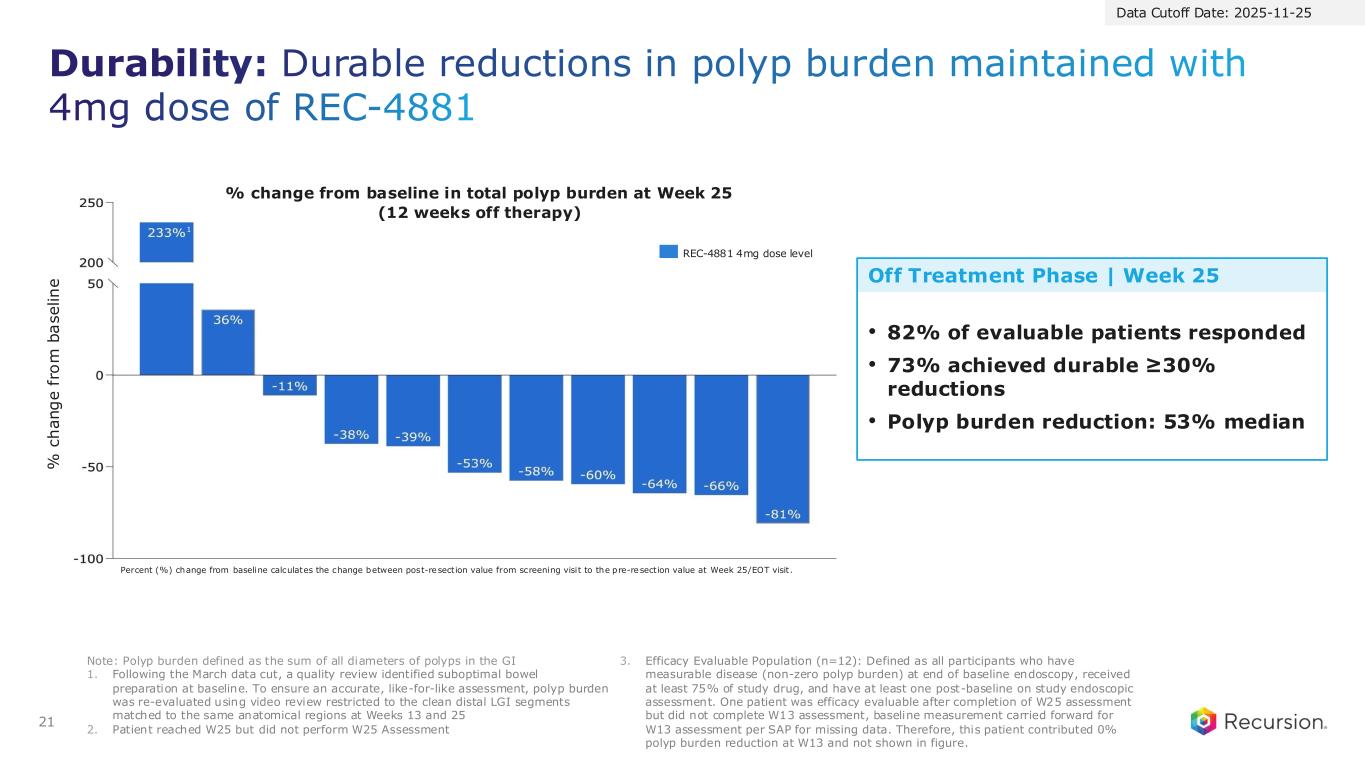
21 Durability: Durable reductions in polyp burden maintained with 4mg dose of REC-4881 Note: Polyp burden defined as the sum of all diameters of polyps in the GI 1. Following the March data cut, a quality review identified suboptimal bowel preparation at baseline. To ensure an accurate, like-for-like assessment, polyp burden was re-evaluated using video review restricted to the clean distal LGI segments matched to the same anatomical regions at Weeks 13 and 25 2. Patient reached W25 but did not perform W25 Assessment 3. Efficacy Evaluable Population (n=12): Defined as all participants who have measurable disease (non-zero polyp burden) at end of baseline endoscopy, received at least 75% of study drug, and have at least one post-baseline on study endoscopic assessment. One patient was efficacy evaluable after completion of W25 assessment but did not complete W13 assessment, baseline measurement carried forward for W13 assessment per SAP for missing data. Therefore, this patient contributed 0% polyp burden reduction at W13 and not shown in figure. Data Cutoff Date: 2025-11-25 • 82% of evaluable patients responded • 73% achieved durable ≥30% reductions • Polyp burden reduction: 53% median REC-4881 4mg dose level % change from baseline in total polyp burden at Week 25 (12 weeks off therapy) % c h a n g e f ro m b a s e li n e Percent (%) change from baseline calculates the change between post-resection value from screening visit to the pre-resection value at Week 25/EOT visit. 1 Off Treatment Phase | Week 25
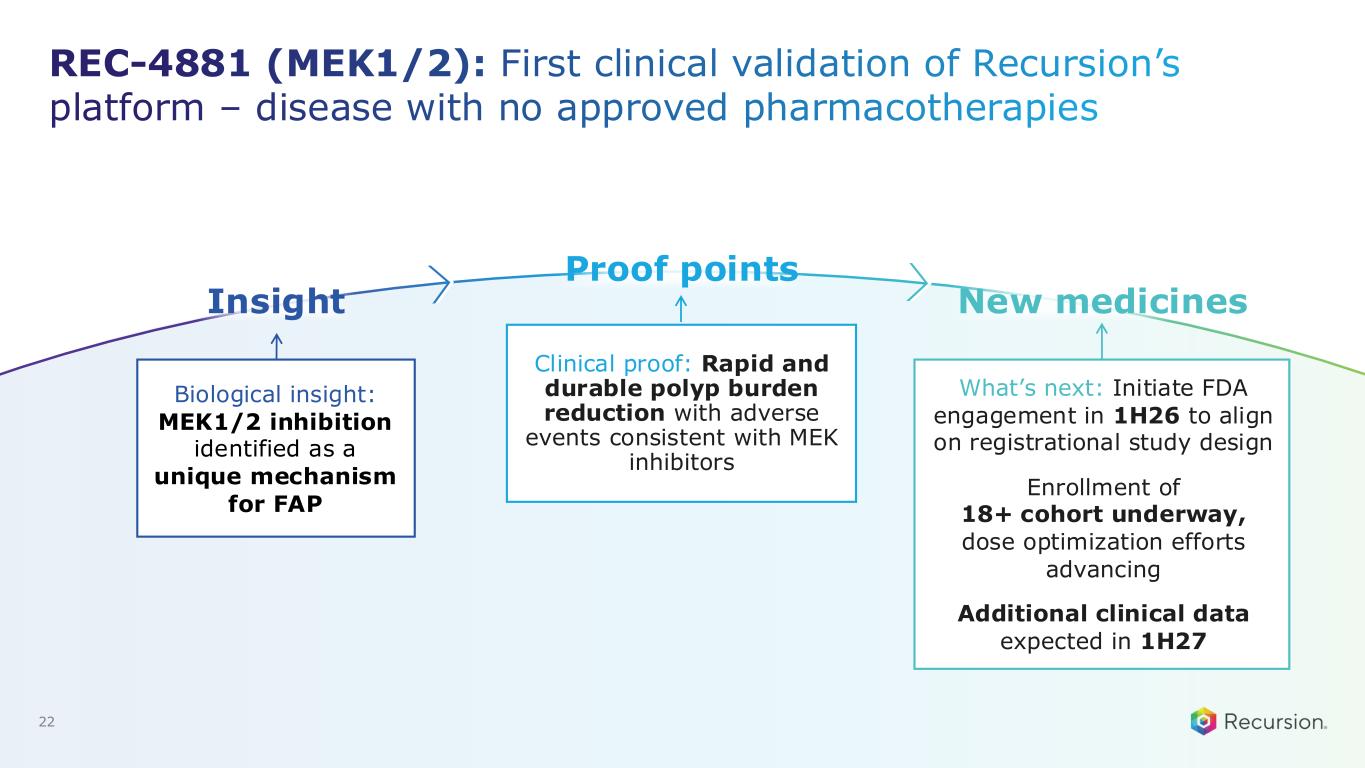
REC-4881 (MEK1/2): First clinical validation of Recursion’s platform – disease with no approved pharmacotherapies Biological insight: MEK1/2 inhibition identified as a unique mechanism for FAP Clinical proof: Rapid and durable polyp burden reduction with adverse events consistent with MEK inhibitors What’s next: Initiate FDA engagement in 1H26 to align on registrational study design Enrollment of 18+ cohort underway, dose optimization efforts advancing Additional clinical data expected in 1H27 22 Insight Proof points New medicines

REC-7735 Translate insights ➔ proof points ➔ new medicines

REC-7735: PI3K⍺ H1047R mutant selective inhibitor designed to improve therapeutic index Unmet need • >21,0001 patients with H1047R-mutant solid tumors • Current PI3K⍺ inhibitors are constrained by: o Hyperglycemia and metabolic toxicity o Dose interruptions and reductions o Limited treatment duration Thesis & differentiation based on preclinical data • >100x mutant selectivity vs WT PI3K⍺, minimizing risk for AEs • Potential for superior efficacy and synergistic effect in combination with SOC • Limited to no impact on hyperglycemia markers 1. Addressable patient populations estimate based on annual US+EU5 and currently identified indications24 Recursion approach • AI-powered precision design to optimize selectivity and limit metabolic liabilities such as hyperglycemia • 242 novel compounds synthesized from first novel hit to REC-7735 Program Status • IND enabling studies ongoing • Go/no-go decision 2H26
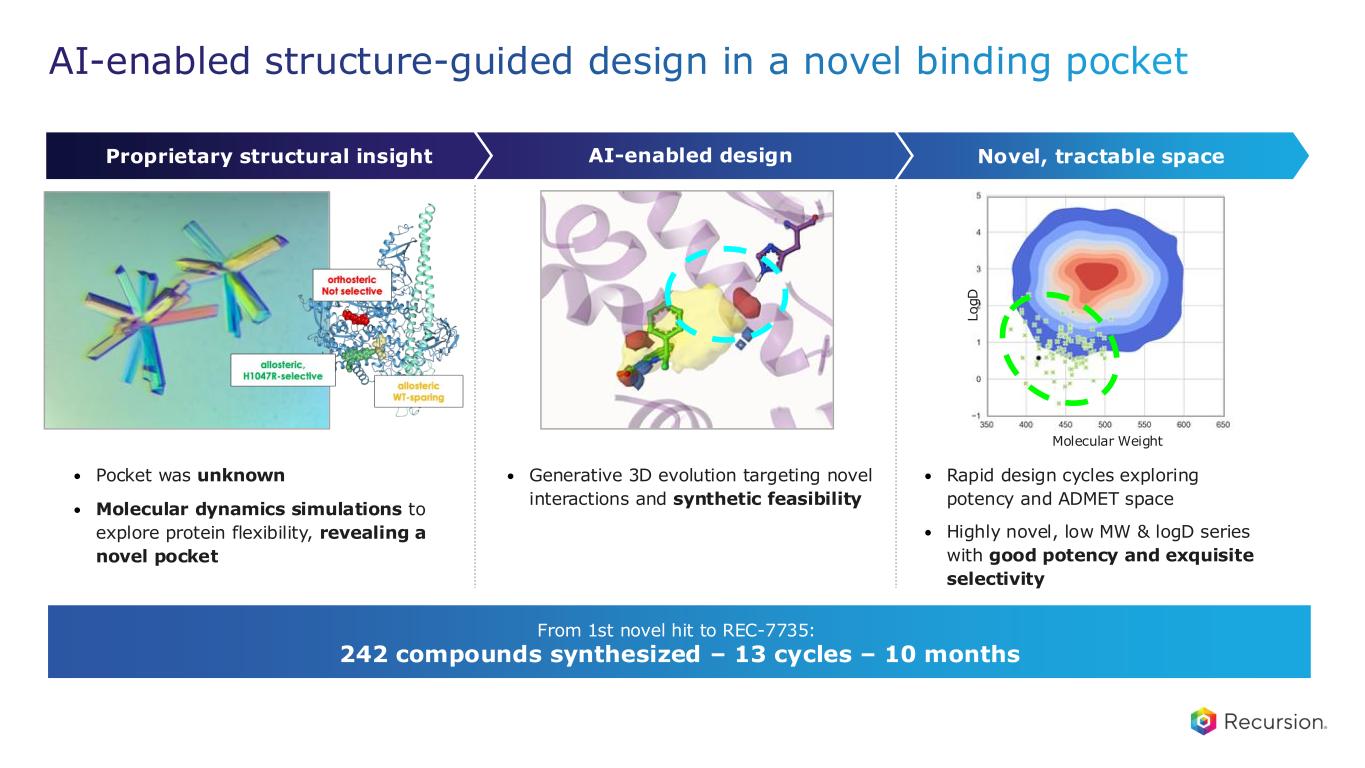
• Generative 3D evolution targeting novel interactions and synthetic feasibility • Pocket was unknown • Molecular dynamics simulations to explore protein flexibility, revealing a novel pocket • Rapid design cycles exploring potency and ADMET space • Highly novel, low MW & logD series with good potency and exquisite selectivity L o g D Molecular Weight AI-enabled structure-guided design in a novel binding pocket Novel, tractable spaceAI-enabled designProprietary structural insight From 1st novel hit to REC-7735: 242 compounds synthesized – 13 cycles – 10 months
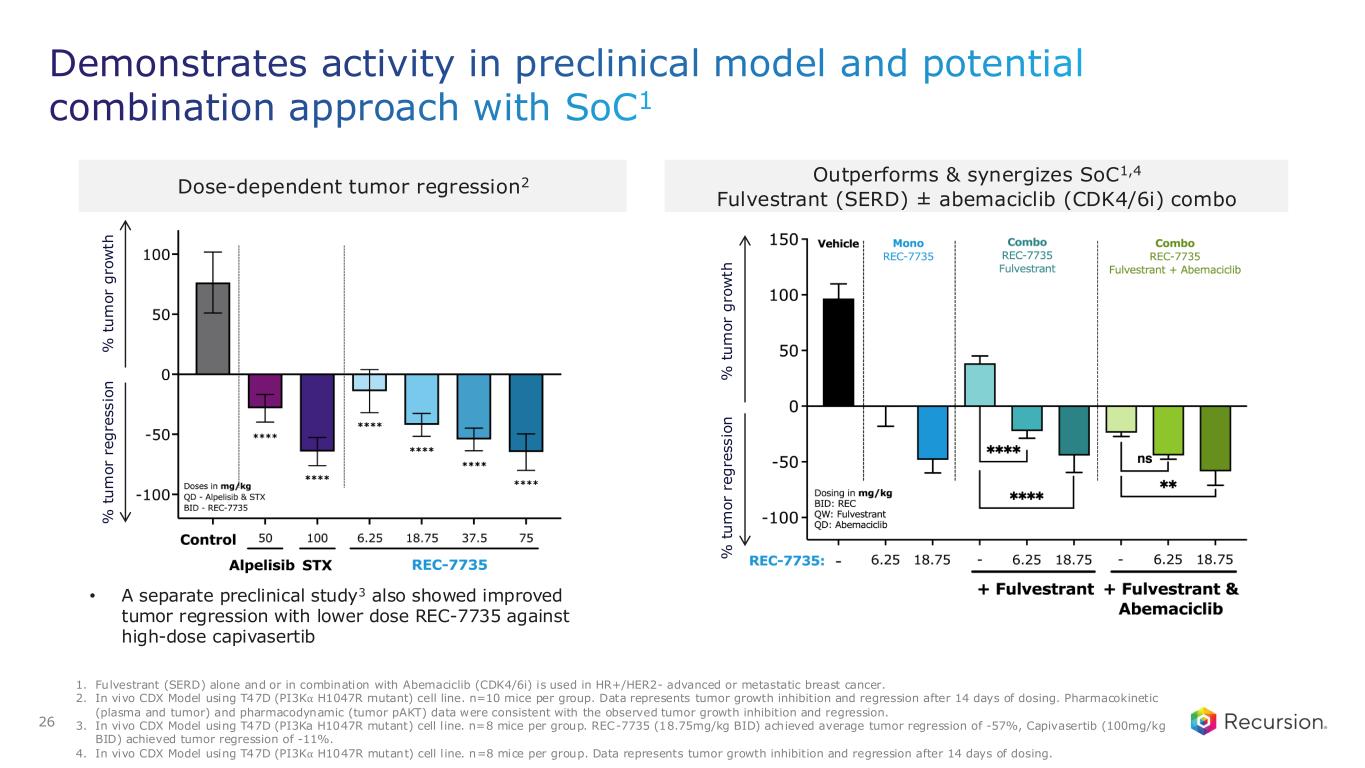
Demonstrates activity in preclinical model and potential combination approach with SoC1 26 1. Fulvestrant (SERD) alone and or in combination with Abemaciclib (CDK4/6i) is used in HR+/HER2- advanced or metastatic breast cancer. 2. In vivo CDX Model using T47D (PI3K⍺ H1047R mutant) cell line. n=10 mice per group. Data represents tumor growth inhibition and regression after 14 days of dosing. Pharmacokinetic (plasma and tumor) and pharmacodynamic (tumor pAKT) data were consistent with the observed tumor growth inhibition and regression. 3. In vivo CDX Model using T47D (PI3Ka H1047R mutant) cell l ine. n=8 mice per group. REC-7735 (18.75mg/kg BID) achieved average tumor regression of -57%, Capivasertib (100mg/kg BID) achieved tumor regression of -11%. 4. In vivo CDX Model using T47D (PI3K⍺ H1047R mutant) cell l ine. n=8 mice per group. Data represents tumor growth inhibition and regression after 14 days of dosing. Dose-dependent tumor regression2 Outperforms & synergizes SoC1,4 Fulvestrant (SERD) ± abemaciclib (CDK4/6i) combo % t u m o r g ro w th % t u m o r re g re s s io n • A separate preclinical study3 also showed improved tumor regression with lower dose REC-7735 against high-dose capivasertib % t u m o r g ro w th % t u m o r re g re s s io n
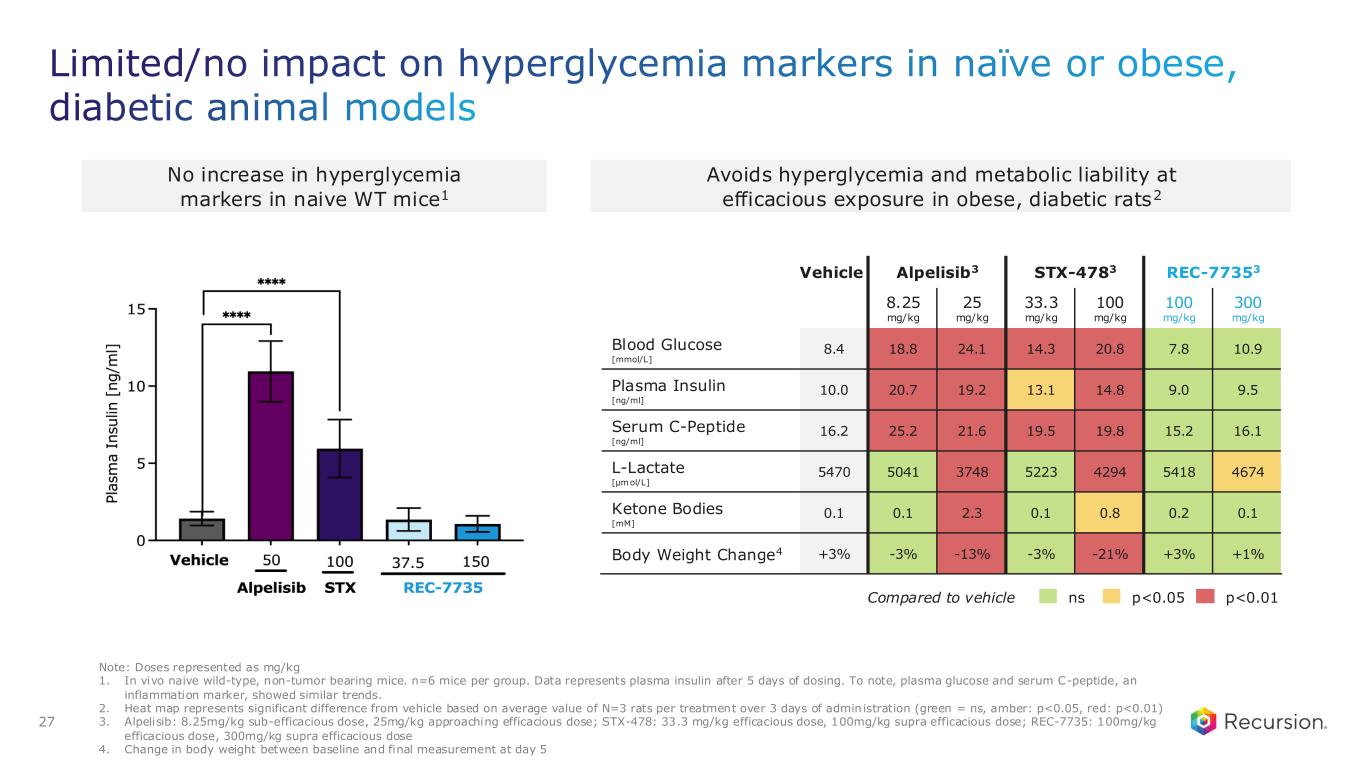
Limited/no impact on hyperglycemia markers in naïve or obese, diabetic animal models 27 Note: Doses represented as mg/kg 1. In vivo naive wild-type, non-tumor bearing mice. n=6 mice per group. Data represents plasma insulin after 5 days of dosing. To note, plasma glucose and serum C-peptide, an inflammation marker, showed similar trends. 2. Heat map represents significant difference from vehicle based on average value of N=3 rats per treatment over 3 days of admin istration (green = ns, amber: p<0.05, red: p<0.01) 3. Alpelisib: 8.25mg/kg sub-efficacious dose, 25mg/kg approaching efficacious dose; STX-478: 33.3 mg/kg efficacious dose, 100mg/kg supra efficacious dose; REC-7735: 100mg/kg efficacious dose, 300mg/kg supra efficacious dose 4. Change in body weight between baseline and final measurement at day 5 Vehicle Alpelisib3 STX-4783 REC-77353 8.25 mg/kg 25 mg/kg 33.3 mg/kg 100 mg/kg 100 mg/kg 300 mg/kg Blood Glucose [mmol/L] 8.4 18.8 24.1 14.3 20.8 7.8 10.9 Plasma Insulin [ng/ml] 10.0 20.7 19.2 13.1 14.8 9.0 9.5 Serum C-Peptide [ng/ml] 16.2 25.2 21.6 19.5 19.8 15.2 16.1 L-Lactate [μmol/L] 5470 5041 3748 5223 4294 5418 4674 Ketone Bodies [mM] 0.1 0.1 2.3 0.1 0.8 0.2 0.1 Body Weight Change4 +3% -3% -13% -3% -21% +3% +1% No increase in hyperglycemia markers in naive WT mice1 Avoids hyperglycemia and metabolic liability at efficacious exposure in obese, diabetic rats2 nsCompared to vehicle p<0.05 p<0.01

Potential to expand the treatable population through improved therapeutic index 28 1. ITOVEBI FDA Label 2. PIQRAY FDA Label; Narayan P et al. Clin Cancer Res (2021) 27 (7): 1842–1849 3. Cross-sectional, Kaplan-Meier analysis of a large administrative health insurance database, calculating time from first exposure to discontinuation for currently approved PI3K inhibitors; median time to discontinuation suggests 50% continue with treatment in this timeframe ~11,000 of HR+, HER2- BC population 14-55% Dose reduction1,2 65-85% Experienced hyperglycemia1,2 66-69% Dose interruption1,2 Current PI3K⍺ inhibitors (SoC) REC-7735 expansion opportunity through improved therapeutic index 3-6 mos Tolerability-driven limitations: >21,000 Across HER2+ BC, TNBC, colorectal, endometrial & uterine, and ovarian If tolerability improves: • Broader patient eligibility • Longer treatment duration • Expanded combination potential Clinical validation of improved tolerability required to confirm expansion thesisReal-world median time to discontinuation3
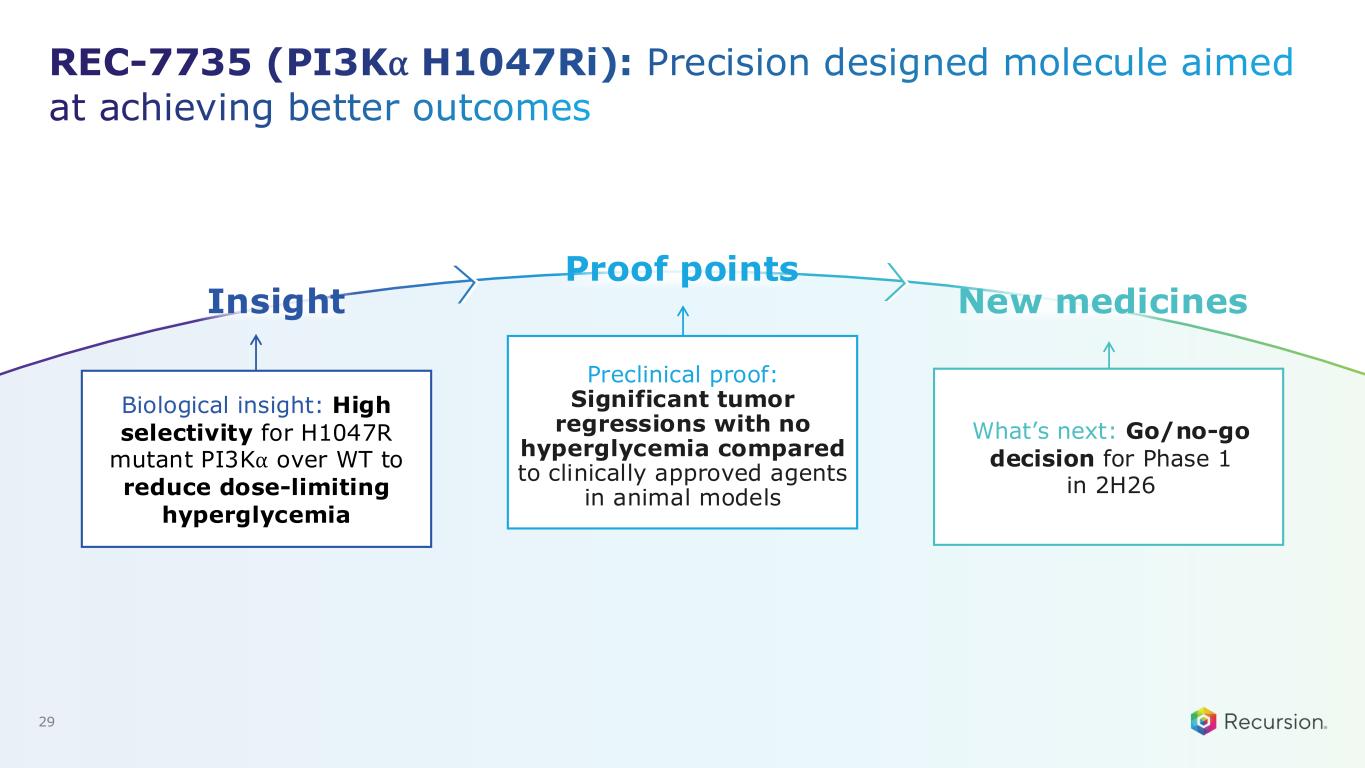
REC-7735 (PI3K⍺ H1047Ri): Precision designed molecule aimed at achieving better outcomes 29 Biological insight: High selectivity for H1047R mutant PI3K⍺ over WT to reduce dose-limiting hyperglycemia What’s next: Go/no-go decision for Phase 1 in 2H26 Preclinical proof: Significant tumor regressions with no hyperglycemia compared to clinically approved agents in animal models Insight Proof points New medicines

Translate insights ➔ proof points ➔ new medicines Partnered Discovery
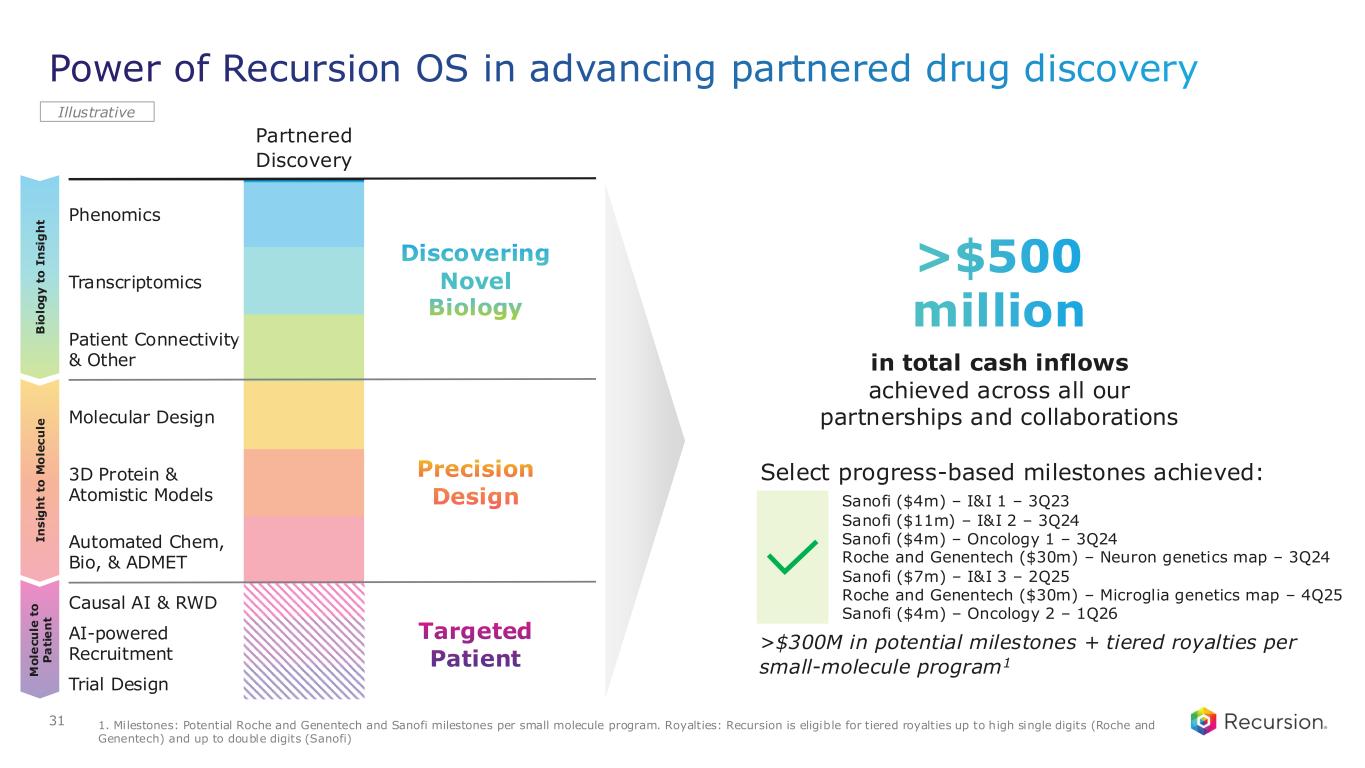
Power of Recursion OS in advancing partnered drug discovery Partnered Discovery Phenomics Transcriptomics Patient Connectivity & Other Molecular Design 3D Protein & Atomistic Models Automated Chem, Bio, & ADMET Causal AI & RWD AI-powered Recruitment Trial Design I n s ig h t to M o le c u le B io lo g y t o I n s ig h t M o le c u le t o P a ti e n t Illustrative Discovering Novel Biology Precision Design Targeted Patient in total cash inflows achieved across all our partnerships and collaborations >$500 million 31 Select progress-based milestones achieved: Roche and Genentech ($30m) – Neuron genetics map – 3Q24 Sanofi ($7m) – I&I 3 – 2Q25 Roche and Genentech ($30m) – Microglia genetics map – 4Q25 Sanofi ($4m) – Oncology 2 – 1Q26 Sanofi ($4m) – I&I 1 – 3Q23 Sanofi ($11m) – I&I 2 – 3Q24 Sanofi ($4m) – Oncology 1 – 3Q24 >$300M in potential milestones + tiered royalties per small-molecule program1 1. Milestones: Potential Roche and Genentech and Sanofi milestones per small molecule program. Royalties: Recursion is eligible for tiered royalties up to high single digits (Roche and Genentech) and up to double digits (Sanofi)

Challenging targets Differentiated, potentially best-in- class and first-in-class molecules Programs aim to address specific unmet clinical needs not served by current SoC 5 advanced lead packages delivered by Recursion accepted by Sanofi to date $34M in milestones to date, in addition to $100M upfront Potential for later stage discovery milestones over next 18 months Fifth milestone with Sanofi: Advancing a joint portfolio of differentiated molecules for challenging targets in I&I and oncology 32 Indication Most Recent Milestone Achieved I&I 1 $4M – 3Q 23 I&I 2 $11M – 3Q 24 I&I 3 $7M – 2Q 25 Oncology 1 $4M – 3Q 24 Oncology 2 $4M – 1Q 26 Multiple early discovery projects Advanced Lead Development Candidate Partnership Highlights
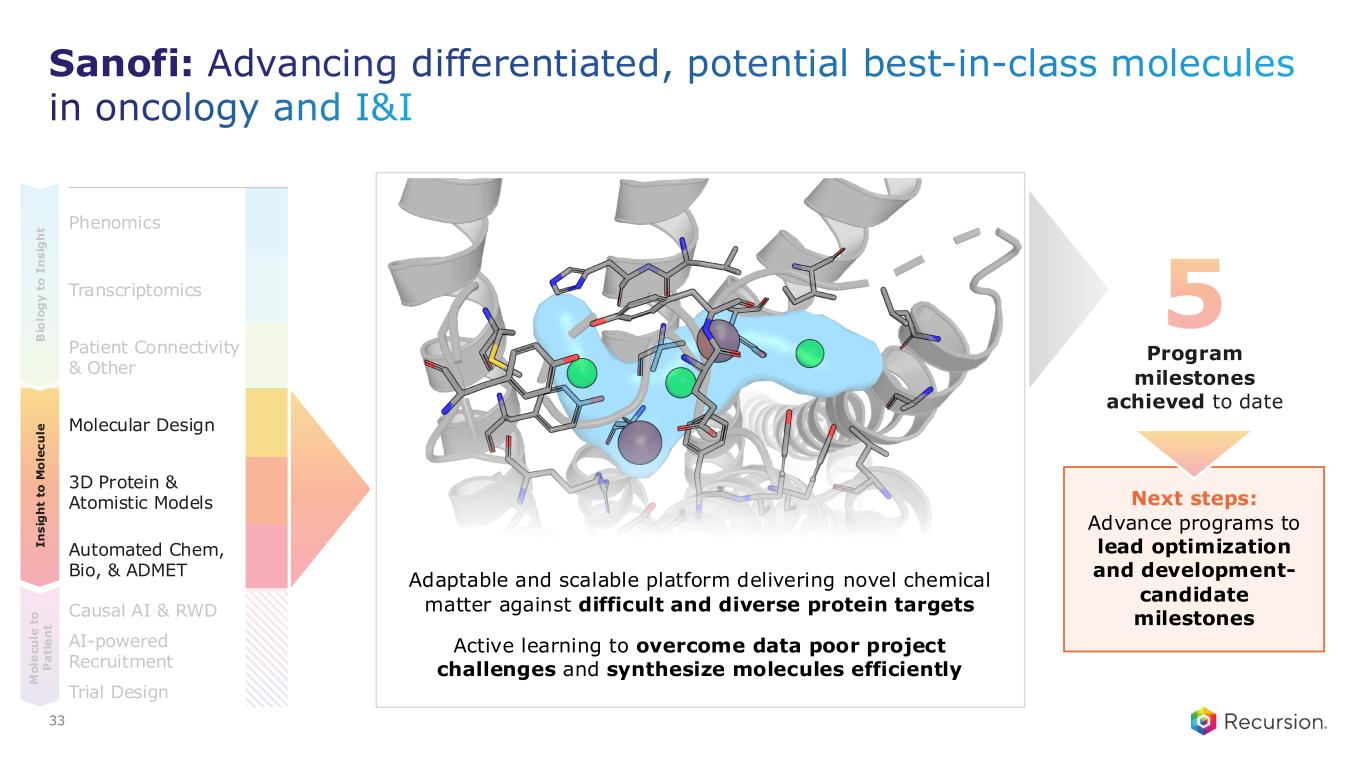
Sanofi: Advancing differentiated, potential best-in-class molecules in oncology and I&I Phenomics Transcriptomics Patient Connectivity & Other Molecular Design 3D Protein & Atomistic Models Automated Chem, Bio, & ADMET Causal AI & RWD AI-powered Recruitment Trial Design I n s ig h t to M o le c u le B io lo g y t o I n s ig h t M o le c u le t o P a ti e n t 5 Program milestones achieved to date Next steps: Advance programs to lead optimization and development- candidate milestones Adaptable and scalable platform delivering novel chemical matter against difficult and diverse protein targets Active learning to overcome data poor project challenges and synthesize molecules efficiently 33

Recursion OS Platform Focused innovation, grounded in clear impact
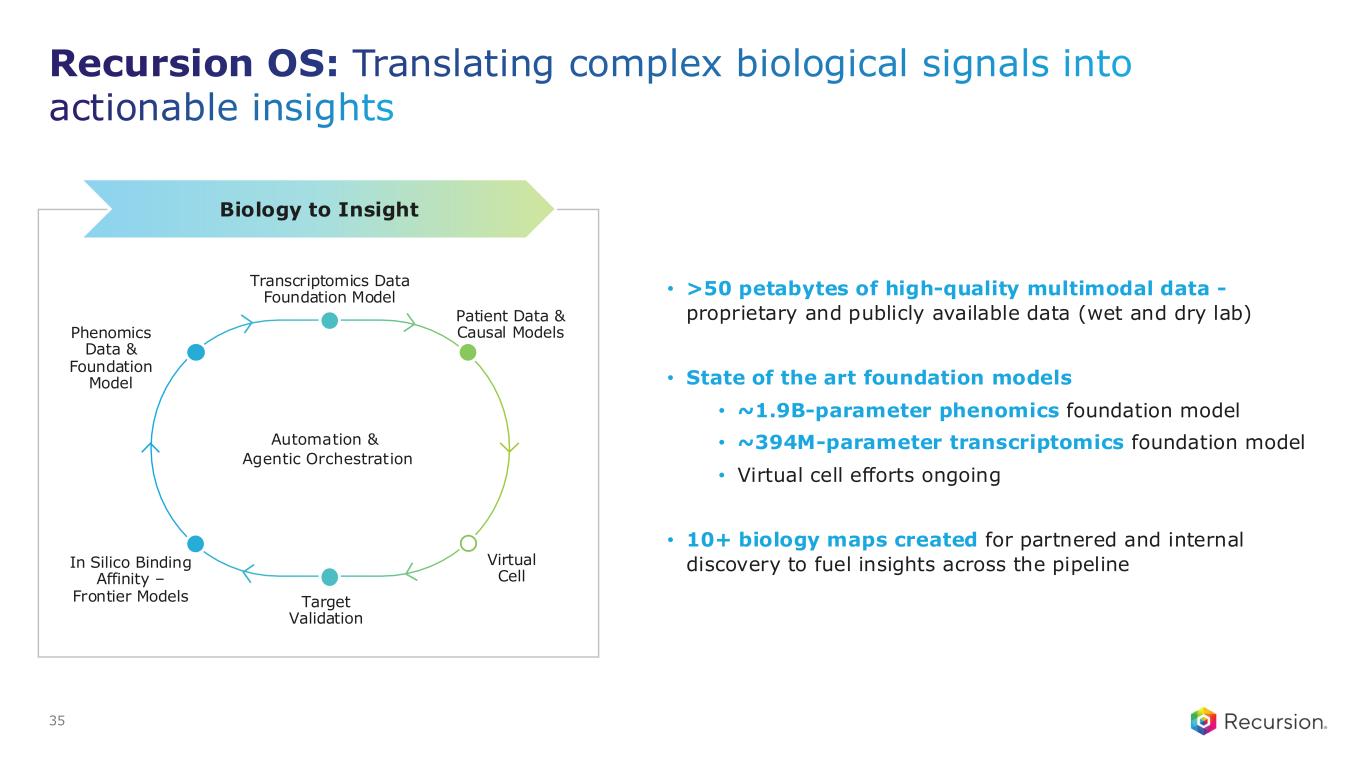
Recursion OS: Translating complex biological signals into actionable insights 35 Biology to Insight • >50 petabytes of high-quality multimodal data - proprietary and publicly available data (wet and dry lab) • State of the art foundation models • ~1.9B-parameter phenomics foundation model • ~394M-parameter transcriptomics foundation model • Virtual cell efforts ongoing • 10+ biology maps created for partnered and internal discovery to fuel insights across the pipelineIn Silico Binding Affinity – Frontier Models Transcriptomics Data Foundation Model Virtual Cell Target Validation Phenomics Data & Foundation Model Automation & Agentic Orchestration Patient Data & Causal Models
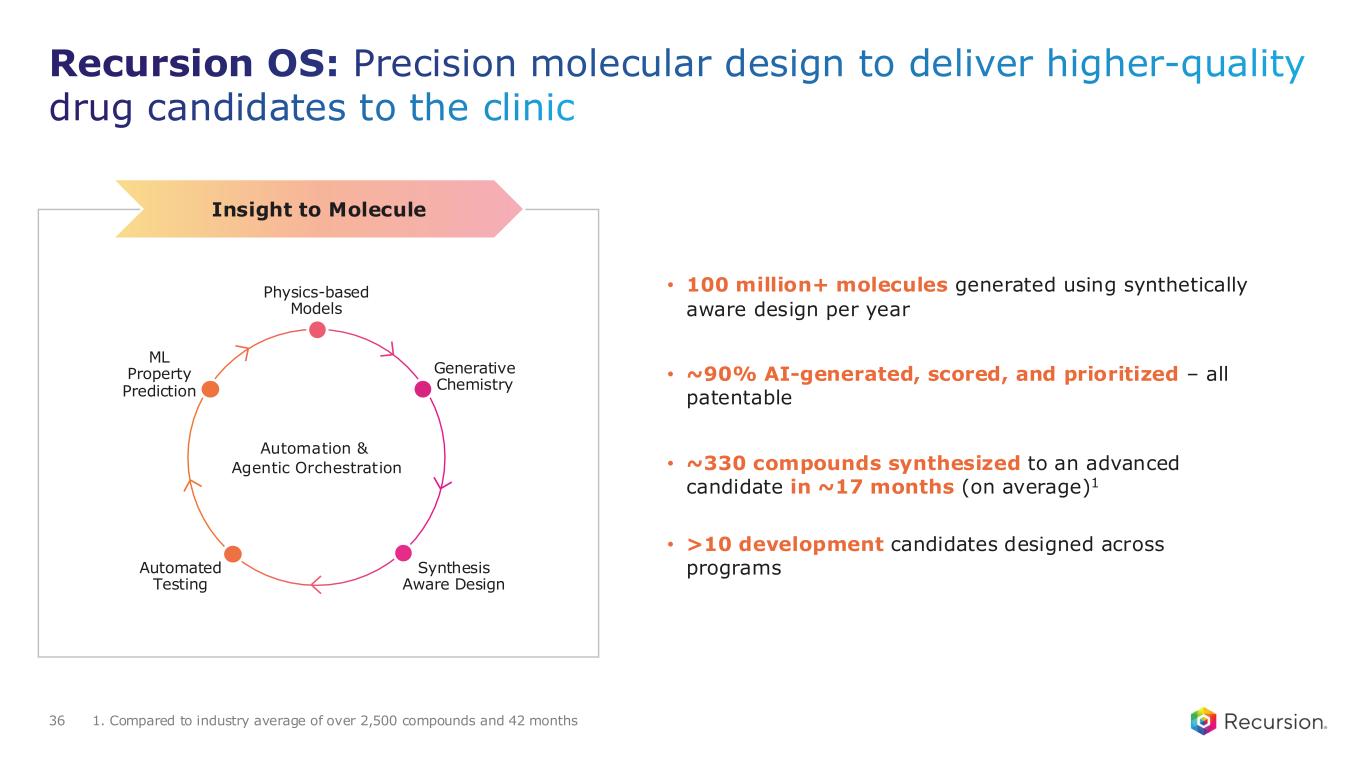
Recursion OS: Precision molecular design to deliver higher-quality drug candidates to the clinic 36 • 100 million+ molecules generated using synthetically aware design per year • ~90% AI-generated, scored, and prioritized – all patentable • ~330 compounds synthesized to an advanced candidate in ~17 months (on average)1 • >10 development candidates designed across programs 1. Compared to industry average of over 2,500 compounds and 42 months Insight to Molecule Automated Testing Synthesis Aware Design Physics-based Models ML Property Prediction Generative Chemistry Automation & Agentic Orchestration

Recursion OS: Designed to improve trial efficiency, patient access, and probability of clinical success 37 Causal AI Patient Selection & RWD Automation & Agentic Orchestration AI-powered Recruitment Clinical Trial Design Molecule to Patient Real-World Scale: • ~300M real-world lives accessible through integrated data partnerships Clinical Execution Improvements (Early Results): • 10–40% increase in eligible patient population • ~1.3–1.6× improvement in enrollment rates • Site & country selection reduced from months to hours • Study startup reductions of up to 3 months Precision & Evidence Integration: • Causal AI-driven patient selection deployed across programs • Natural history and EHR analyses contextualizing trial results with RWD
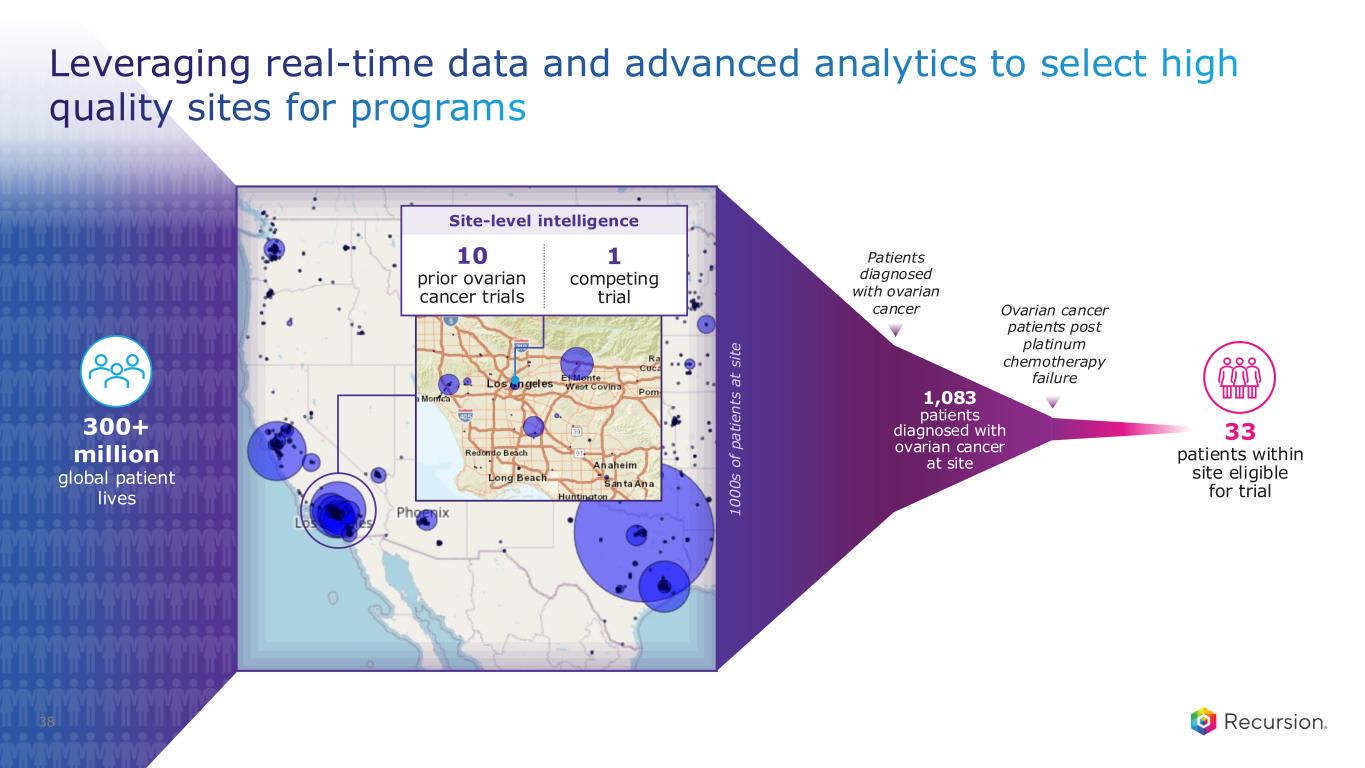
38 Leveraging real-time data and advanced analytics to select high quality sites for programs Ovarian cancer patients post platinum chemotherapy failure 1,083 patients diagnosed with ovarian cancer at site 300+ million global patient lives Patients diagnosed with ovarian cancer 33 patients within site eligible for trial 1 0 0 0 s o f p a ti e n ts a t s it e 10 prior ovarian cancer trials 1 competing trial Site-level intelligence

Financials

Extended cash runway to deliver on upcoming milestones 40 1. Cash, cash equivalents and restricted cash 2. Cash operating expense—defined as net cash used in operating activities less partnership inflows and transaction costs—is a non-GAAP financial measure. See Appendix for reconciliation of non-GAAP financial measures. • Expected 2026 cash operating expense2 of <$390 million 2025 partnership inflows: highlights 2025 cash1 and expense update Reiterating cash operating expense guidance • $754 million in cash1 as of December 31, 2025 • 2025 cash operating expense2 of $399 million: 10% below guidance and a ~35% reduction year-over-year • Total partnership inflows >$500 million • $30 million milestone from Roche for microglia map • $7 million milestone payment from Sanofi for I&I program Expected cash runway into early 2028, without additional financing

Looking ahead

Expected upcoming milestones 2026 and 2027 pipeline and partnership catalysts 42 1. Data-driven decision for potential Phase 1 initiation 2. Cash, cash equivalents and restricted cash as of December 31, 2025 3. Runway guidance includes risk-adjusted cash inflows from partnerships 4. Cash operating expense—defined as net cash used in operating activities less partnership inflows and transaction costs—is a non-GAAP financial measure. See Appendix for reconciliation of non-GAAP financial measures. Translate insights ➔ proof points ➔ new medicines Pair bold ambition with disciplined execution Focused innovation, grounded in clear impact 1H 2026 REC-4881 (MEK1/2i) Initial engagement with FDA REC-1245 (RBM39 degrader) Mono - early safety and PK 2H 2027 REC-4539 (LSD1i) Mono - Early safety and PK 1H 2027 REC-4881 (MEK1/2i) Additional clinical data REC-617 (CDK7i) Combo - early safety and PK REC-3565 (MALT1i) Mono - early safety and PK • $754M in YE 2025 cash2 with expected runway into early 20283 • Expected 2026 cash operating expense4 of <$390 million • Biology foundation models and patient data enabling scalable, high-quality target discovery • Generative AI design at scale (next-gen models and agentic systems) • Clinical Development AI at scale Partner catalysts – 2026 & 2027 Later-stage discovery milestones Advancing maps to early-stage programs Anticipated multiple new project initiations 2H 2026 Go/no-go decision1 REC-7735 (PI3K⍺ H1047Ri) REC-102 (ENPP1i)

43 THANK YOU

Non-GAAP Financial Measures To supplement our financial statements prepared in accordance with U.S. GAAP, we monitor and consider operating cash burn, which is a non-GAAP financial measure. We define operating cash expense as the net cash used in operating activities, excluding non-ordinary course transaction costs and partnership cash inflows. This non-GAAP financial measure is not based on any standardized methodology prescribed by U.S. GAAP and is not necessarily comparable to similarly- titled measures presented by other companies. We believe operating cash expense to be a liquidity measure that provides useful information to management and investors about the amount of cash consumed by the operations of the business. A limitation of using this non-U.S. GAAP measure is that operating cash expense does not represent the total change in cash and cash equivalents for the period because it excludes cash provided by or used for other investing and financing activities. We account for this limitation by providing information about our capital expenditures and other investing and financing activities in the statements of cash flows in our financial statements. Additionally, we reconciled operating cash burn below to net cash used in operating activities, the most directly comparable U.S. GAAP financial measure. In addition, it is important to note that other companies, including companies in our industry, may not use operating cash expense, may calculate operating cash expense in a different manner than we do or may use other financial measures to evaluate their performance, all of which could reduce the usefulness of operating cash expense as a comparative measure. Because of these limitations, operating cash burn should not be considered in isolation from, or as a substitute for, financial information prepared in accordance with U.S. GAAP. Appendix 44