
Earnings 1Q26 MAY 6, 2026

This presentation of Recursion Pharmaceuticals, Inc. (“Recursion,” “we,” “us,” or “our”) and any accompanying discussion contain statements that are not historical facts may be considered forward-looking statements under federal securities laws and may be identified by words such as “anticipates,” “believes,” “estimates,” “expects,” “intends,” “plans,” “potential,” “predicts,” “projects,” “seeks,” “should,” “will,” or words of similar meaning and include, but are not limited to, statements regarding the impact of the acceptance of the fifth milestone by Sanofi on future developments and potential treatments; the impact of FAP trial on the Recursion OS and other clinical and preclinical programs; financial position, cash runway, and ability to reduce our cash expense; our ability to use AI to translate complex science into medicines faster and better; Recursion’s OS industrializing first- and best-in-class drug discovery; our ability to industrialize clinical development and the effect of doing so on clinical trial outcomes; the occurrence or realization of potential milestones and their potential timing or amounts; current and future preclinical and clinical studies, including timelines for enrollment in studies, data readouts, progression toward IND- enabling and other potential studies, and engagement with the FDA; advancements of and other decisions regarding our pipeline, partnerships, and data strategies; the potential size of the market opportunity for our drug candidates; outcomes and benefits from licenses, partnerships and collaborations, including option exercises by partners; the initiation, timing, progress, results, and cost of our research and development programs; advancements of our Recursion OS; and many others. Other important factors and information are contained in Recursion’s most recent Annual Report on Form 10-K, and the Company’s other filings with the U.S. Securities and Exchange Commission (the “SEC”), which can be accessed at https://ir.recursion.com, or www.sec.gov. All forward-looking statements are qualified by these cautionary statements and apply only as of the date they are made. Recursion does not undertake any obligation to update any forward- looking statement, whether as a result of new information, future events or otherwise. Certain information contained in this presentation relates to or is based on studies, publications, surveys and other data obtained from third-party sources and the company’s own internal estimates and research. While the company believes these third-party sources to be reliable as of the date of this presentation, it has not independently verified, and makes no representation as to the adequacy, fairness, accuracy or completeness of, any information obtained from third- party sources. In addition, all of the market data included in this presentation involves a number of assumptions and limitations, and there can be no guarantee as to the accuracy or reliability of such assumptions. Finally, while the company believes its own internal research is reliable, such research has not been verified by any independent source. Information contained in, or that can be accessed through our website is not a part of and is not incorporated into this presentation. Cross-trial or cross-candidate information about other investigational assets and drug candidates are not based on head-to-head studies and are presented for informational purposes; data presented are based on publicly available information for other clinical trials and other drug candidates. ® 2026 Recursion Pharmaceuticals, Inc. Recursion and its related logo are registered trademarks of Recursion Pharmaceuticals, Inc. All other trademarks are property of their respective owners. Important Information 2
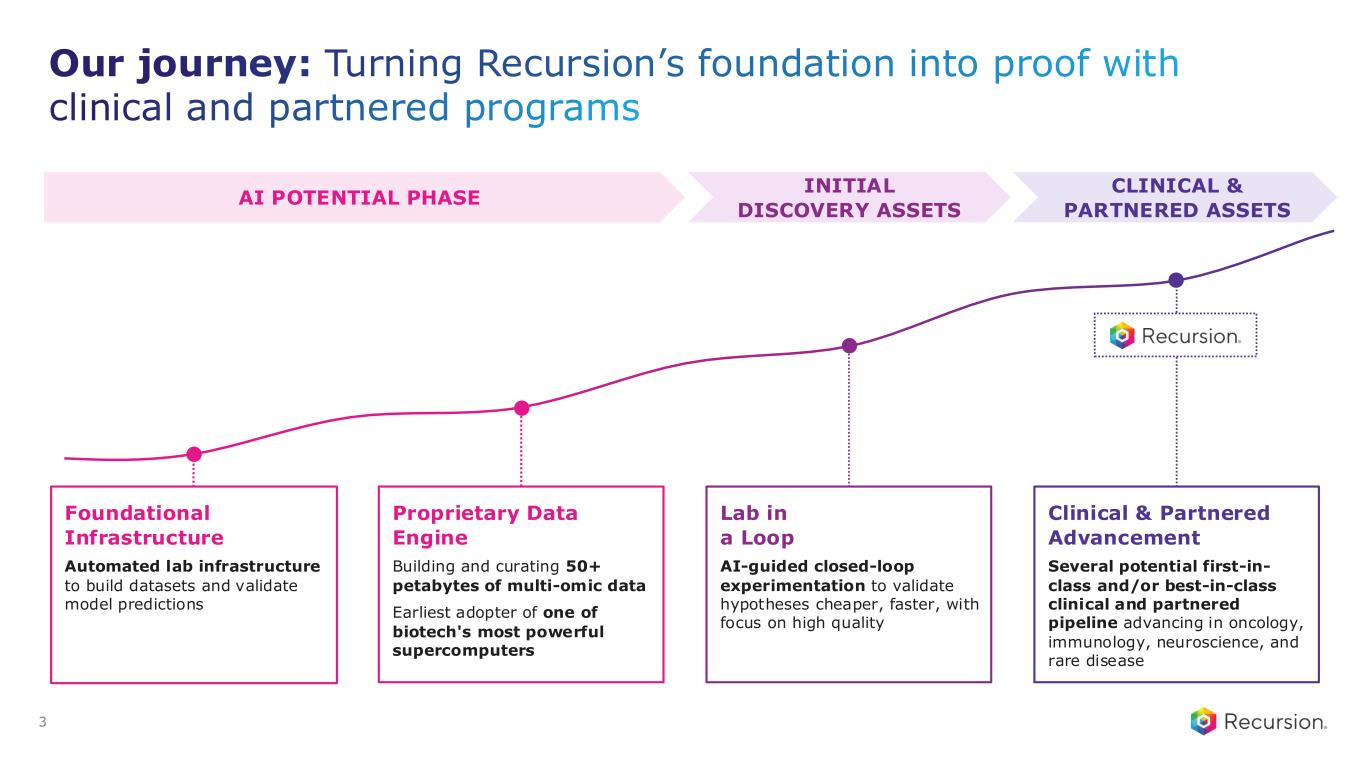
Our journey: Turning Recursion’s foundation into proof with clinical and partnered programs 3 AI POTENTIAL PHASE CLINICAL & PARTNERED ASSETS Foundational Infrastructure Automated lab infrastructure to build datasets and validate model predictions Proprietary Data Engine Building and curating 50+ petabytes of multi-omic data Earliest adopter of one of biotech's most powerful supercomputers Lab in a Loop AI-guided closed-loop experimentation to validate hypotheses cheaper, faster, with focus on high quality Clinical & Partnered Advancement Several potential first-in- class and/or best-in-class clinical and partnered pipeline advancing in oncology, immunology, neuroscience, and rare disease INITIAL DISCOVERY ASSETS
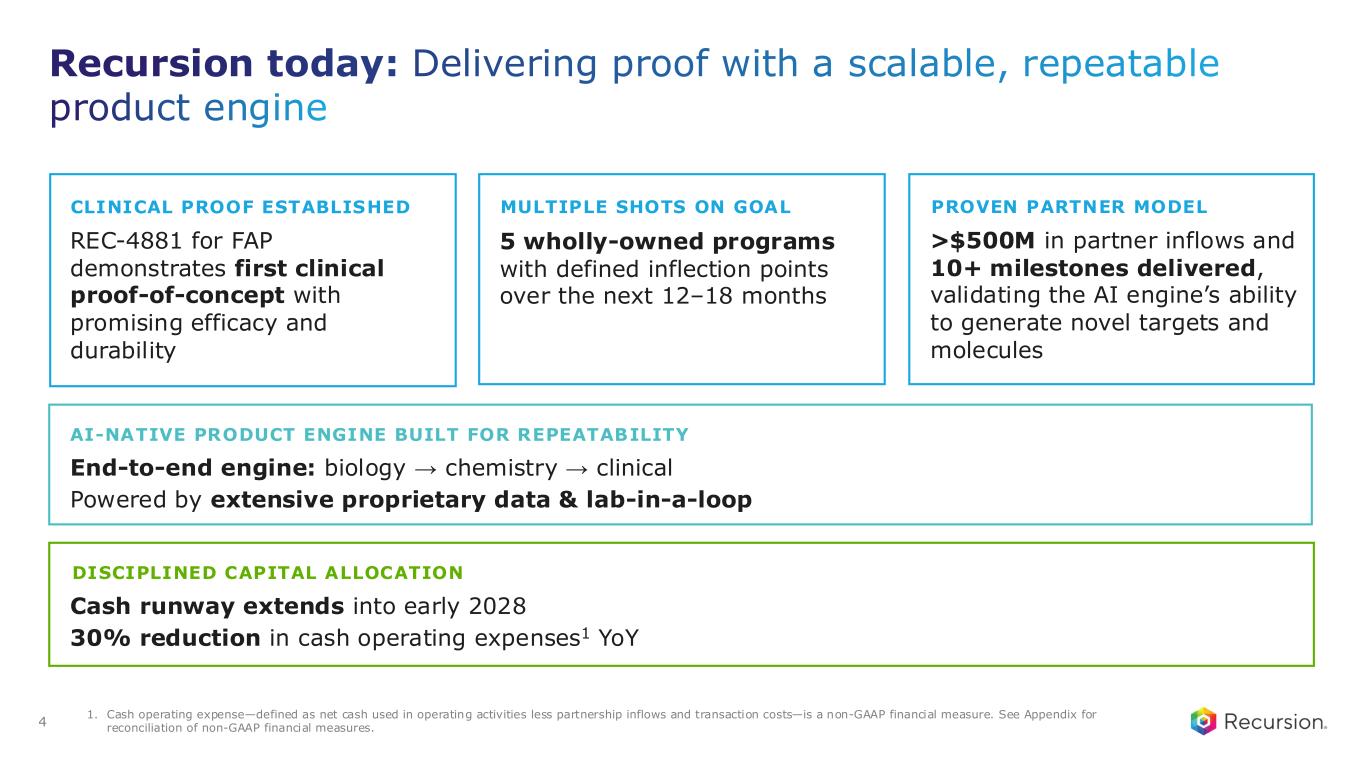
Recursion today: Delivering proof with a scalable, repeatable product engine 4 CLINICAL PROOF ESTABLISHED REC-4881 for FAP demonstrates first clinical proof-of-concept with promising efficacy and durability MULTIPLE SHOTS ON GOAL 5 wholly-owned programs with defined inflection points over the next 12–18 months PROVEN PARTNER MODEL >$500M in partner inflows and 10+ milestones delivered, validating the AI engine’s ability to generate novel targets and molecules AI-NATIVE PRODUCT ENGINE BUILT FOR REPEATABILITY End-to-end engine: biology → chemistry → clinical Powered by extensive proprietary data & lab-in-a-loop DISCIPLINED CAPITAL ALLOCATION Cash runway extends into early 2028 30% reduction in cash operating expenses1 YoY 1. Cash operating expense—defined as net cash used in operating activities less partnership inflows and transaction costs—is a non-GAAP financial measure. See Appendix for reconciliation of non-GAAP financial measures.
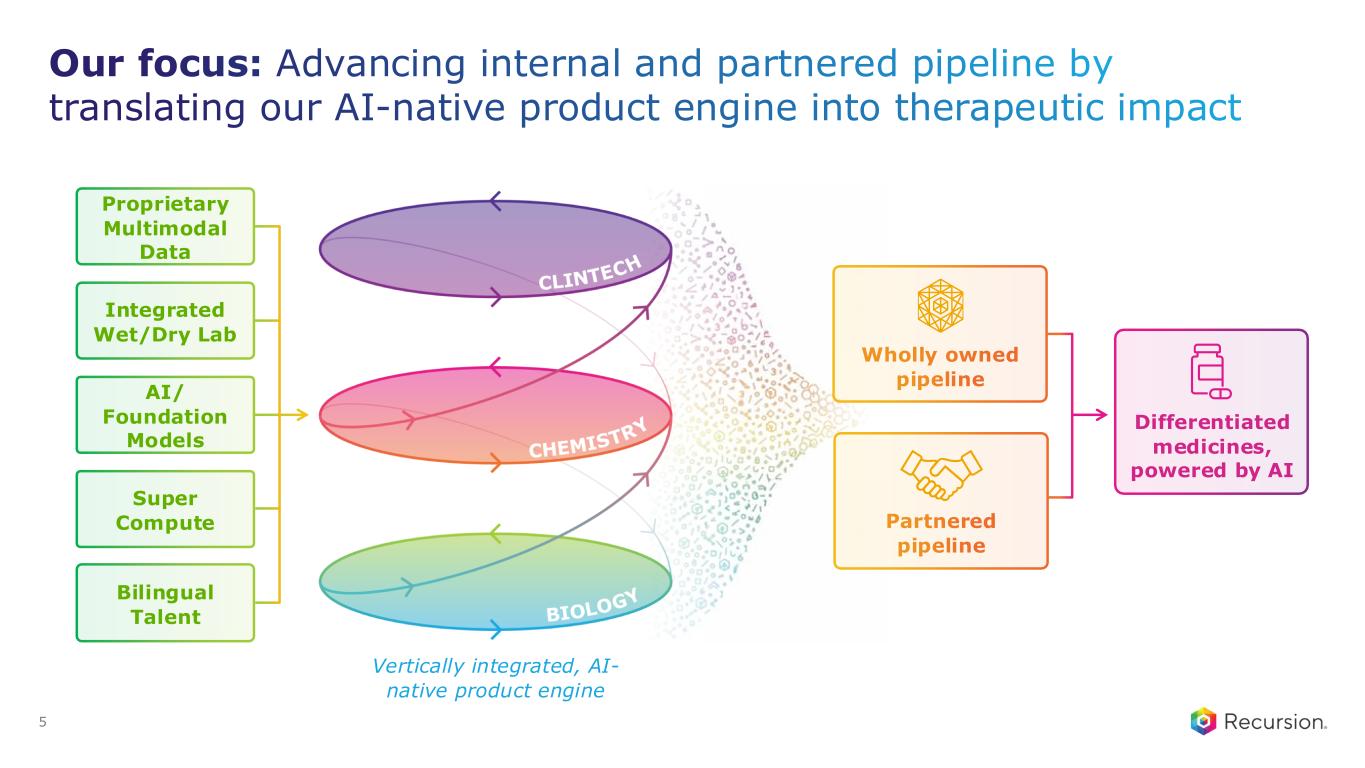
Our focus: Advancing internal and partnered pipeline by translating our AI-native product engine into therapeutic impact 5 Integrated Wet/Dry Lab AI/ Foundation Models Super Compute Bilingual Talent Proprietary Multimodal Data Wholly owned pipeline Partnered pipeline Vertically integrated, AI- native product engine Differentiated medicines, powered by AI
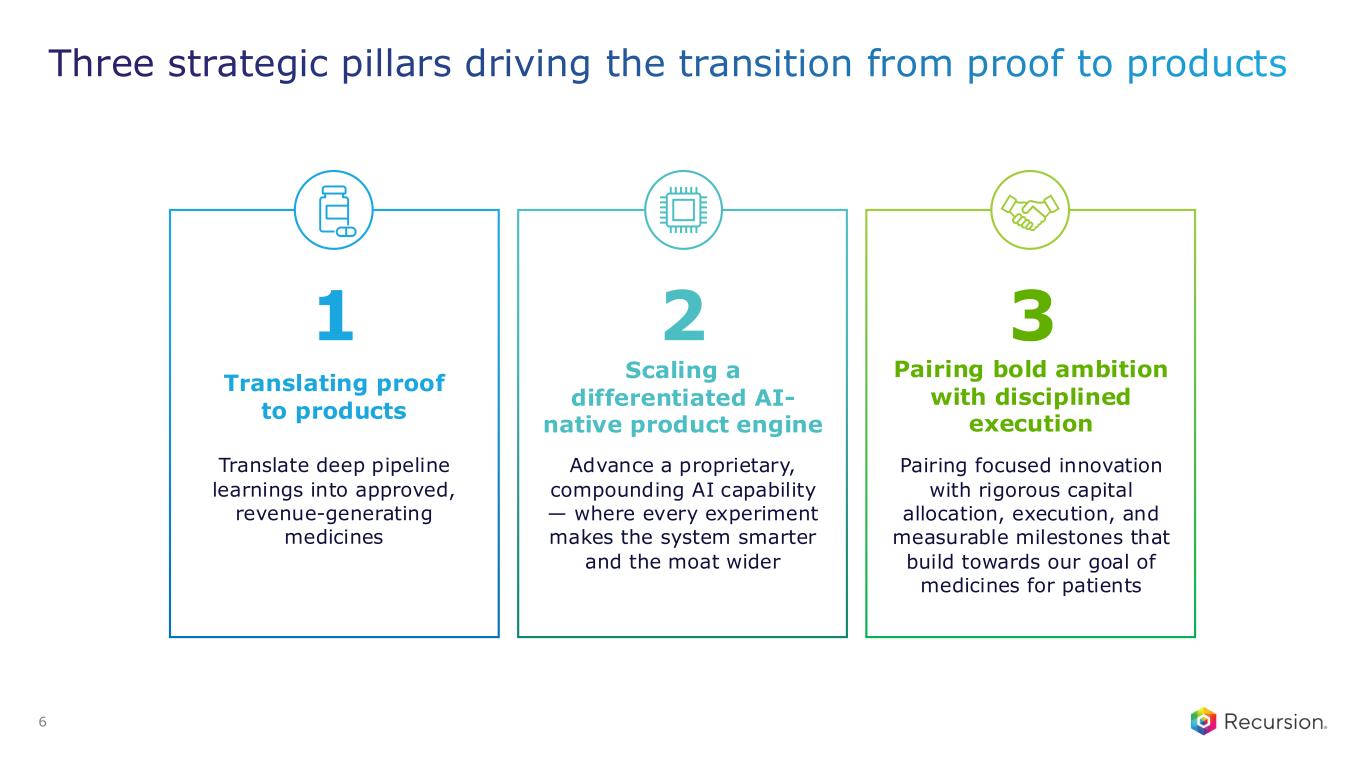
6 Translating proof to products Scaling a differentiated AI- native product engine Pairing bold ambition with disciplined execution Translate deep pipeline learnings into approved, revenue-generating medicines Advance a proprietary, compounding AI capability — where every experiment makes the system smarter and the moat wider Pairing focused innovation with rigorous capital allocation, execution, and measurable milestones that build towards our goal of medicines for patients 1 2 3 Three strategic pillars driving the transition from proof to products
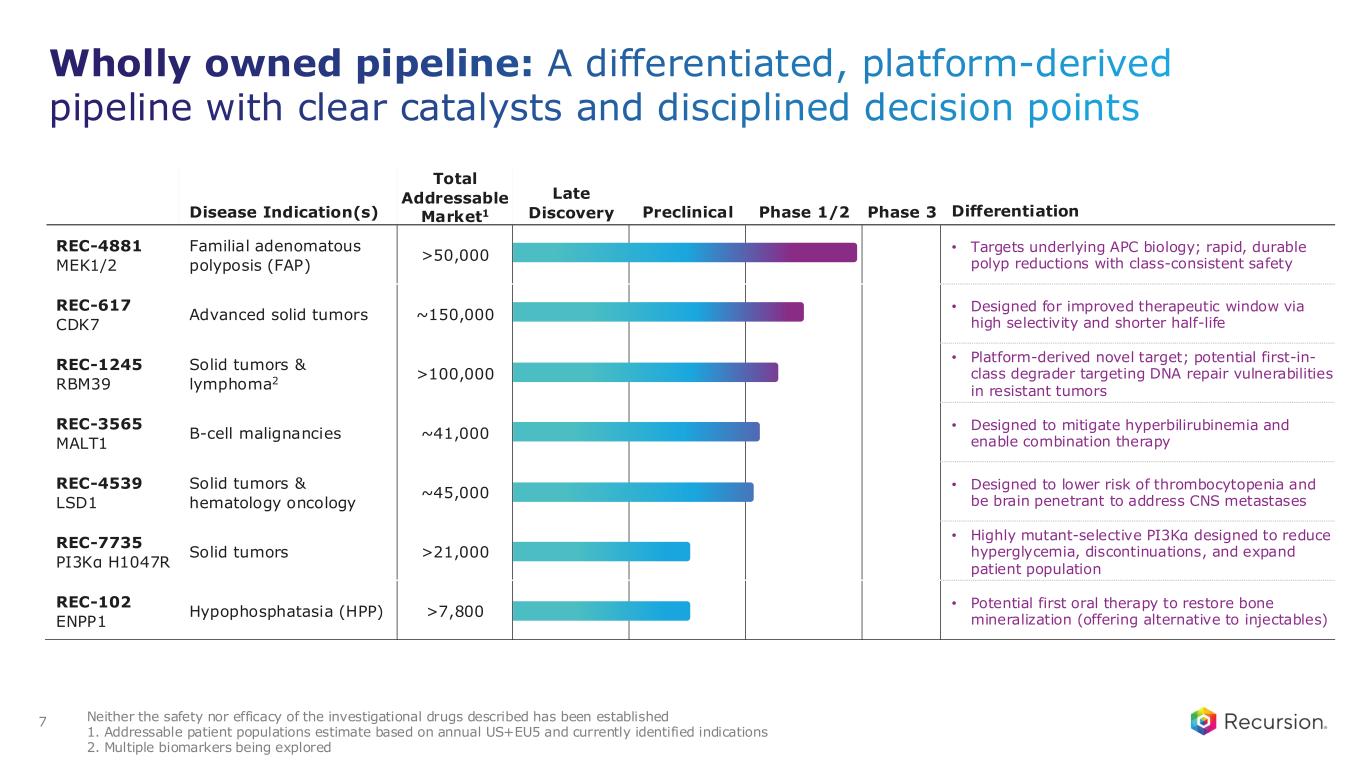
Wholly owned pipeline: A differentiated, platform-derived pipeline with clear catalysts and disciplined decision points 7 Disease Indication(s) Total Addressable Market1 Late Discovery Preclinical Phase 1/2 Phase 3 Differentiation REC-4881 MEK1/2 Familial adenomatous polyposis (FAP) >50,000 • Targets underlying APC biology; rapid, durable polyp reductions with class-consistent safety REC-617 CDK7 Advanced solid tumors ~150,000 • Designed for improved therapeutic window via high selectivity and shorter half-life REC-1245 RBM39 Solid tumors & lymphoma2 >100,000 • Platform-derived novel target; potential first-in- class degrader targeting DNA repair vulnerabilities in resistant tumors REC-3565 MALT1 B-cell malignancies ~41,000 • Designed to mitigate hyperbilirubinemia and enable combination therapy REC-4539 LSD1 Solid tumors & hematology oncology ~45,000 • Designed to lower risk of thrombocytopenia and be brain penetrant to address CNS metastases REC-7735 PI3Kα H1047R Solid tumors >21,000 • Highly mutant-selective PI3Kα designed to reduce hyperglycemia, discontinuations, and expand patient population REC-102 ENPP1 Hypophosphatasia (HPP) >7,800 • Potential first oral therapy to restore bone mineralization (offering alternative to injectables) Neither the safety nor efficacy of the investigational drugs described has been established 1. Addressable patient populations estimate based on annual US+EU5 and currently identified indications 2. Multiple biomarkers being explored
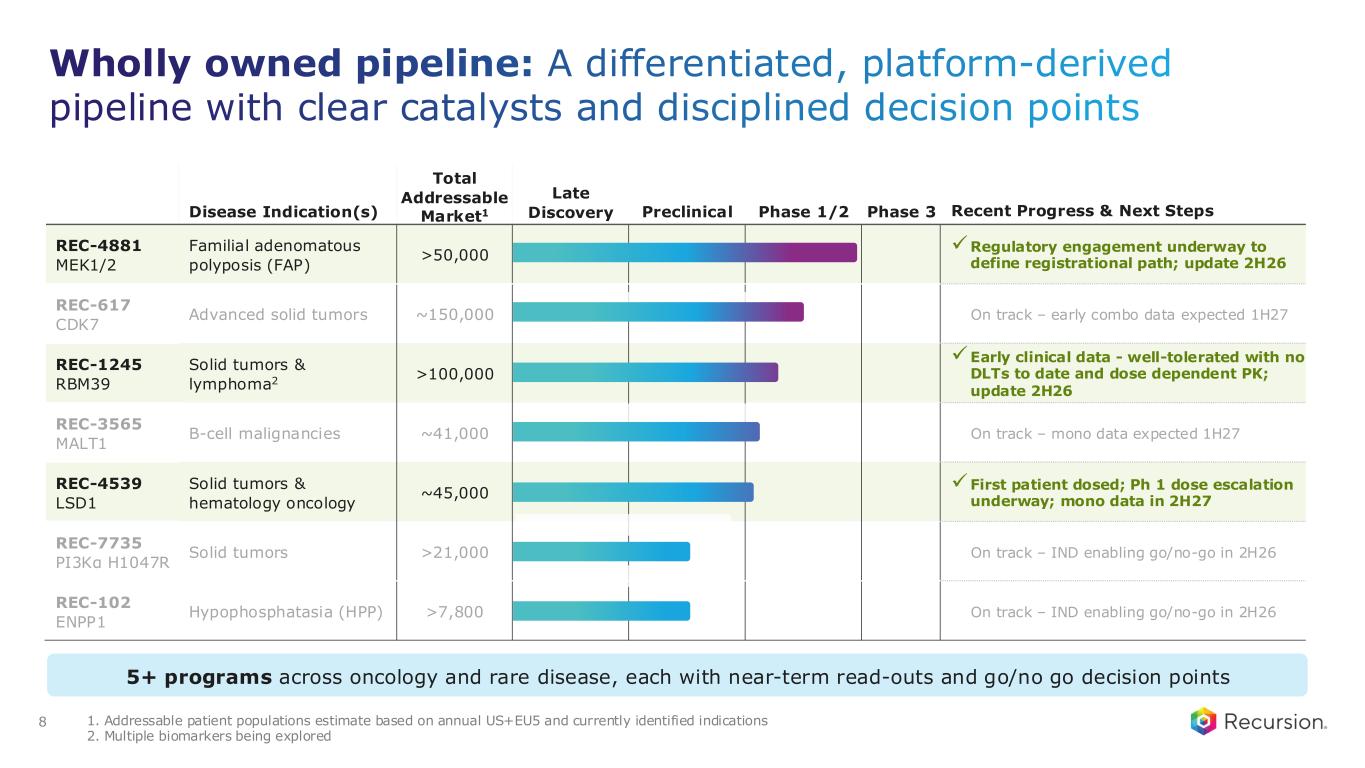
Wholly owned pipeline: A differentiated, platform-derived pipeline with clear catalysts and disciplined decision points 8 Disease Indication(s) Total Addressable Market1 Late Discovery Preclinical Phase 1/2 Phase 3 Recent Progress & Next Steps REC-4881 MEK1/2 Familial adenomatous polyposis (FAP) >50,000 ✓Regulatory engagement underway to define registrational path; update 2H26 REC-617 CDK7 Advanced solid tumors ~150,000 On track – early combo data expected 1H27 REC-1245 RBM39 Solid tumors & lymphoma2 >100,000 ✓Early clinical data - well-tolerated with no DLTs to date and dose dependent PK; update 2H26 REC-3565 MALT1 B-cell malignancies ~41,000 On track – mono data expected 1H27 REC-4539 LSD1 Solid tumors & hematology oncology ~45,000 ✓First patient dosed; Ph 1 dose escalation underway; mono data in 2H27 REC-7735 PI3Kα H1047R Solid tumors >21,000 On track – IND enabling go/no-go in 2H26 REC-102 ENPP1 Hypophosphatasia (HPP) >7,800 On track – IND enabling go/no-go in 2H26 5+ programs across oncology and rare disease, each with near-term read-outs and go/no go decision points 1. Addressable patient populations estimate based on annual US+EU5 and currently identified indications 2. Multiple biomarkers being explored
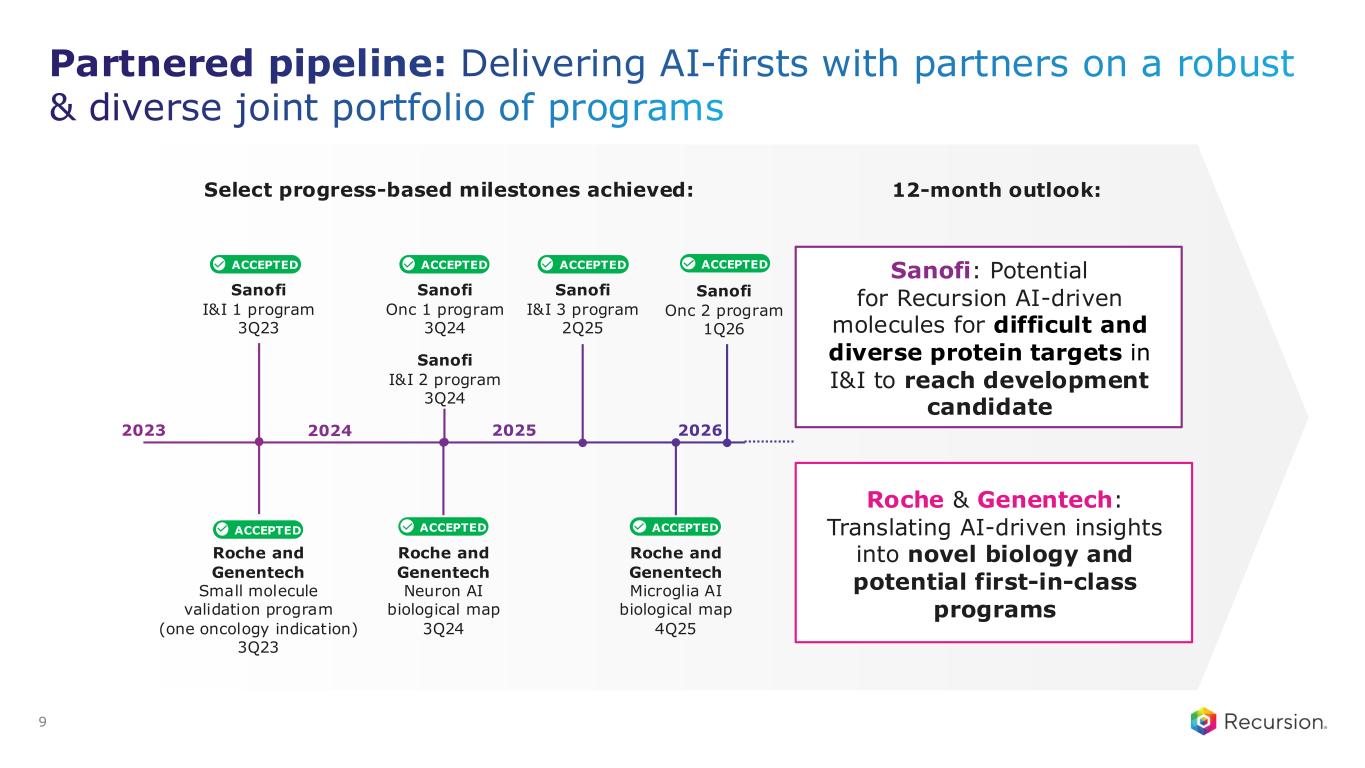
Partnered pipeline: Delivering AI-firsts with partners on a robust & diverse joint portfolio of programs 9 Sanofi: Potential for Recursion AI-driven molecules for difficult and diverse protein targets in I&I to reach development candidate Roche & Genentech: Translating AI-driven insights into novel biology and potential first-in-class programs Select progress-based milestones achieved: 2023 Sanofi I&I 1 program 3Q23 Roche and Genentech Neuron AI biological map 3Q24 Sanofi I&I 3 program 2Q25 Roche and Genentech Microglia AI biological map 4Q25 Sanofi Onc 2 program 1Q26 Sanofi I&I 2 program 3Q24 Sanofi Onc 1 program 3Q24 202620252024 ACCEPTED ACCEPTED ACCEPTED ACCEPTED ACCEPTEDACCEPTEDACCEPTED Roche and Genentech Small molecule validation program (one oncology indication) 3Q23 12-month outlook:

Vicki Goodman, M.D. appointed as Recursion CMO 10 • Seasoned Physician Executive: More than two decades of experience in oncology drug development and medical leadership, across Merck, Bristol Myers Squibb, GlaxoSmithKline, Exelixis, and Mural Oncology • Strategic Clinical Leadership: Former CMO and EVP; oversaw early- to late-stage clinical development, regulatory affairs, and biometrics across multi-asset pipelines • Proven Track Record: Development of KEYTRUDA® (pembrolizumab) (Merck), OPDIVO® (nivolumab) and YERVOY® (ipilimumab) (BMS), and guided dabrafenib (GSK) from early clinical expansion through regulatory approval • Regulatory Insight: Strategic background as a former Medical Officer at the U.S. Food and Drug Administration (FDA) • Clinical Foundation: M.D. from Albert Einstein College of Medicine; clinical training in internal medicine and hematology/oncology at the University of Michigan “I look forward to working with Najat, the leadership team, and the broader organization to advance the pipeline, support smart and disciplined development decisions, and help bring impactful new therapies to patients.”

REC-1245 Translating proof to products
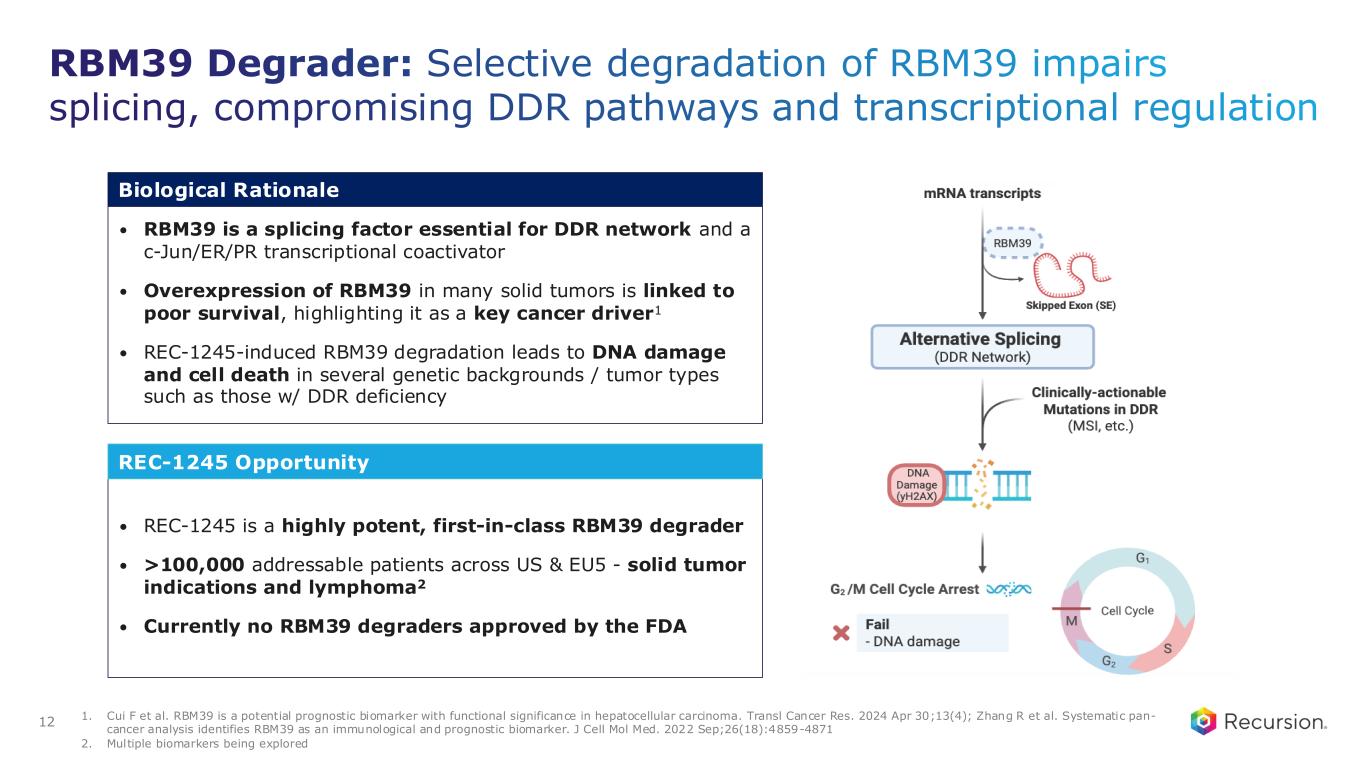
RBM39 Degrader: Selective degradation of RBM39 impairs splicing, compromising DDR pathways and transcriptional regulation • RBM39 is a splicing factor essential for DDR network and a c-Jun/ER/PR transcriptional coactivator • Overexpression of RBM39 in many solid tumors is linked to poor survival, highlighting it as a key cancer driver1 • REC-1245-induced RBM39 degradation leads to DNA damage and cell death in several genetic backgrounds / tumor types such as those w/ DDR deficiency Biological Rationale • REC-1245 is a highly potent, first-in-class RBM39 degrader • >100,000 addressable patients across US & EU5 - solid tumor indications and lymphoma2 • Currently no RBM39 degraders approved by the FDA REC-1245 Opportunity 12 1. Cui F et al. RBM39 is a potential prognostic biomarker with functional significance in hepatocellular carcinoma. Transl Cancer Res. 2024 Apr 30;13(4); Zhang R et al. Systematic pan- cancer analysis identifies RBM39 as an immunological and prognostic biomarker. J Cell Mol Med. 2022 Sep;26(18):4859-4871 2. Multiple biomarkers being explored
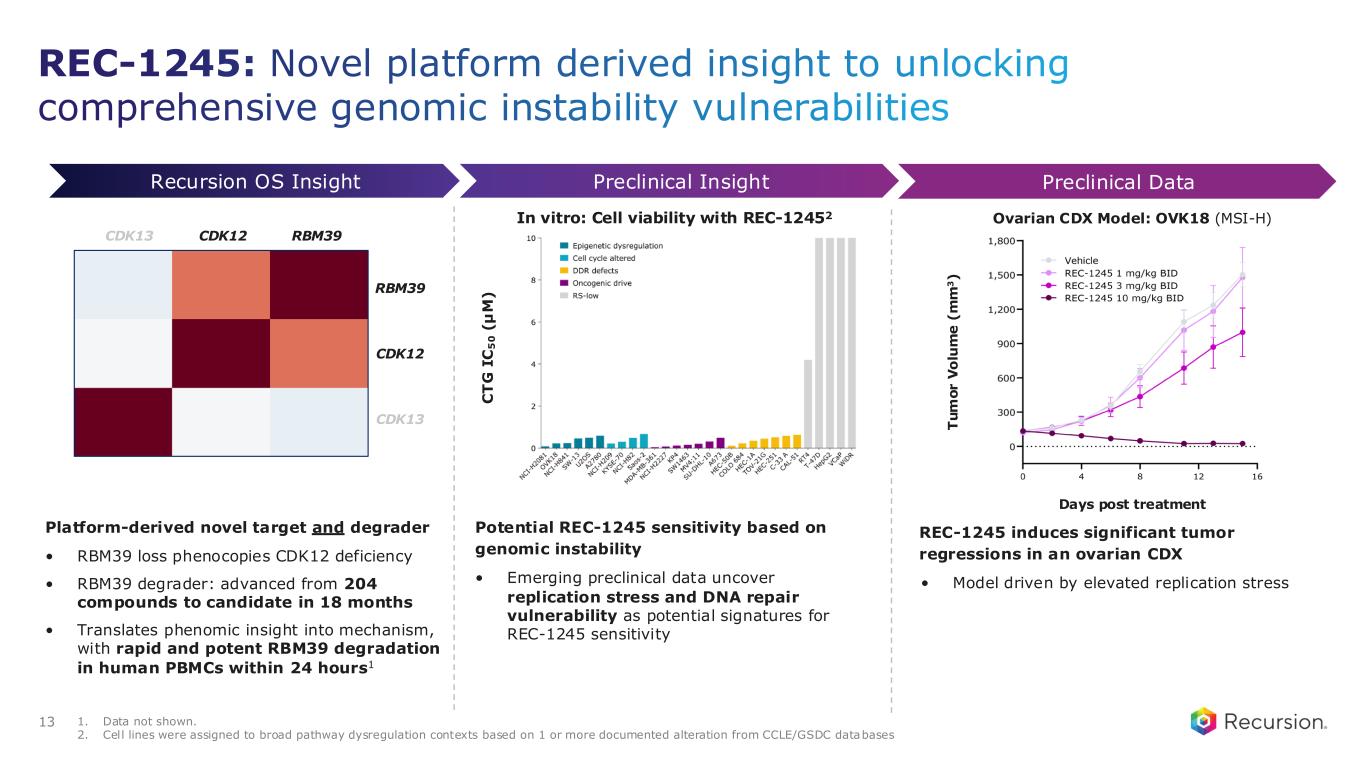
REC-1245: Novel platform derived insight to unlocking comprehensive genomic instability vulnerabilities 13 1. Data not shown. 2. Cell lines were assigned to broad pathway dysregulation contexts based on 1 or more documented alteration from CCLE/GSDC databases REC-1245 induces significant tumor regressions in an ovarian CDX • Model driven by elevated replication stress Preclinical InsightRecursion OS Insight Preclinical Data Days post treatment T u m o r V o lu m e ( m m 3 ) Ovarian CDX Model: OVK18 (MSI-H) Platform-derived novel target and degrader • RBM39 loss phenocopies CDK12 deficiency • RBM39 degrader: advanced from 204 compounds to candidate in 18 months • Translates phenomic insight into mechanism, with rapid and potent RBM39 degradation in human PBMCs within 24 hours1 CDK12 RBM39CDK13 RBM39 CDK12 CDK13 In vitro: Cell viability with REC-12452 C T G I C 5 0 ( µ M ) Potential REC-1245 sensitivity based on genomic instability • Emerging preclinical data uncover replication stress and DNA repair vulnerability as potential signatures for REC-1245 sensitivity
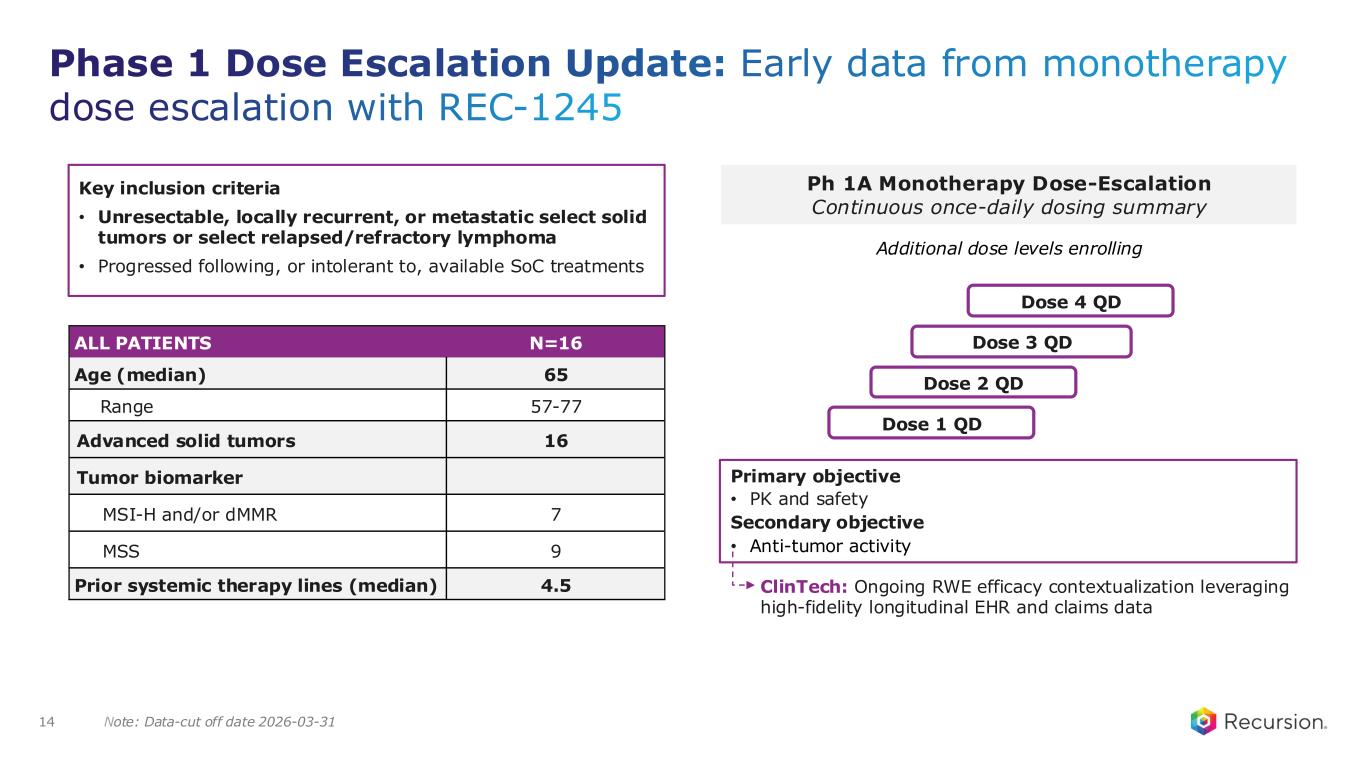
Note: Data-cut off date 2026-03-31 Phase 1 Dose Escalation Update: Early data from monotherapy dose escalation with REC-1245 14 Ph 1A Monotherapy Dose-Escalation Continuous once-daily dosing summary Dose 1 QD Dose 2 QD Dose 3 QD Dose 4 QD Additional dose levels enrolling Primary objective • PK and safety Secondary objective • Anti-tumor activity ALL PATIENTS N=16 Age (median) 65 Range 57-77 Advanced solid tumors 16 Tumor biomarker MSI-H and/or dMMR 7 MSS 9 Prior systemic therapy lines (median) 4.5 Key inclusion criteria • Unresectable, locally recurrent, or metastatic select solid tumors or select relapsed/refractory lymphoma • Progressed following, or intolerant to, available SoC treatments ClinTech: Ongoing RWE efficacy contextualization leveraging high-fidelity longitudinal EHR and claims data
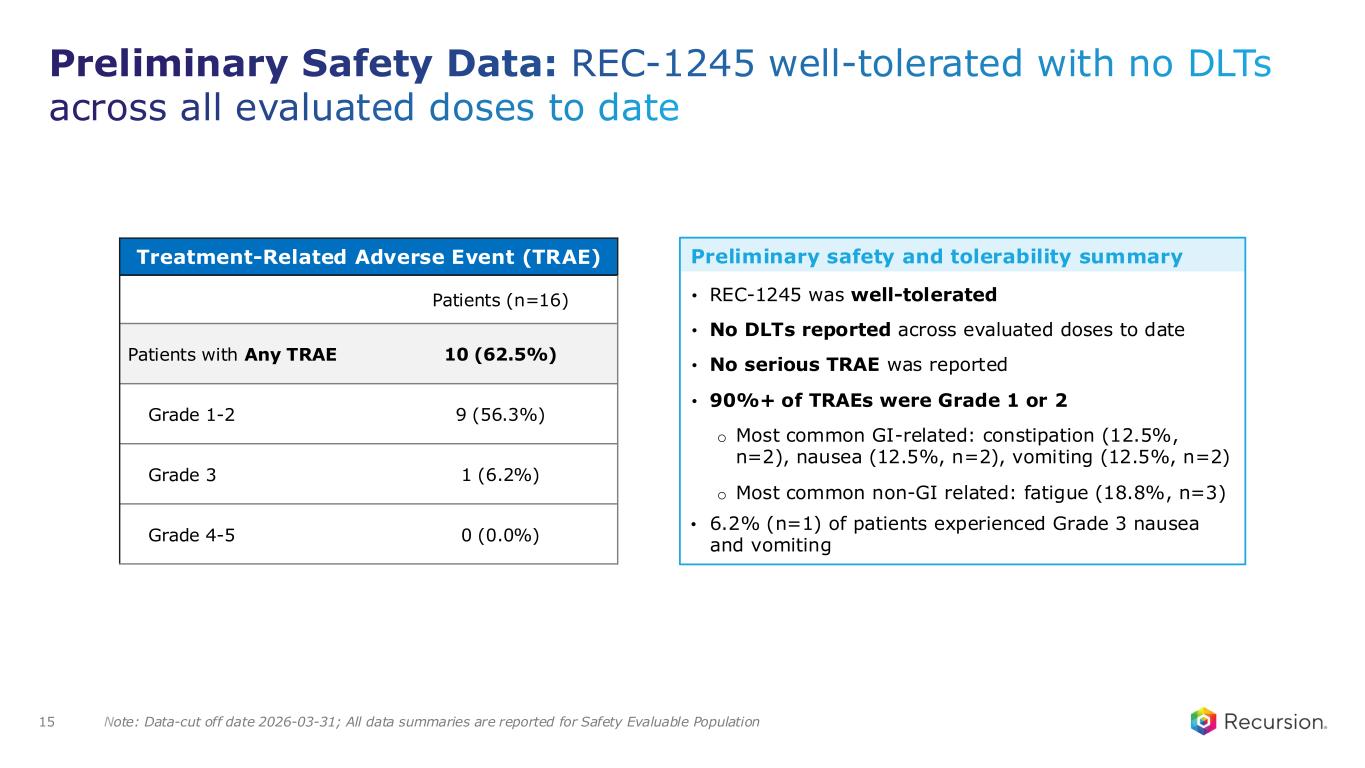
15 Note: Data-cut off date 2026-03-31; All data summaries are reported for Safety Evaluable Population Preliminary Safety Data: REC-1245 well-tolerated with no DLTs across all evaluated doses to date Treatment-Related Adverse Event (TRAE) Patients (n=16) Patients with Any TRAE 10 (62.5%) Grade 1-2 9 (56.3%) Grade 3 1 (6.2%) Grade 4-5 0 (0.0%) • REC-1245 was well-tolerated • No DLTs reported across evaluated doses to date • No serious TRAE was reported • 90%+ of TRAEs were Grade 1 or 2 o Most common GI-related: constipation (12.5%, n=2), nausea (12.5%, n=2), vomiting (12.5%, n=2) o Most common non-GI related: fatigue (18.8%, n=3) • 6.2% (n=1) of patients experienced Grade 3 nausea and vomiting Preliminary safety and tolerability summary

Preliminary PK/PD summary • Predictable, dose-dependent exposure across evaluated patients to date • PK is supportive of QD dosing and exposures continue to increase with dose • Expect to achieve exposures consistent with tumor regression in mice within the next two dose levels • Pharmacodynamic assessments demonstrate target engagement 16 Early data suggests REC-1245 has predictable, dose dependent exposure Note: Data-cut off date 2026-03-31; N=16
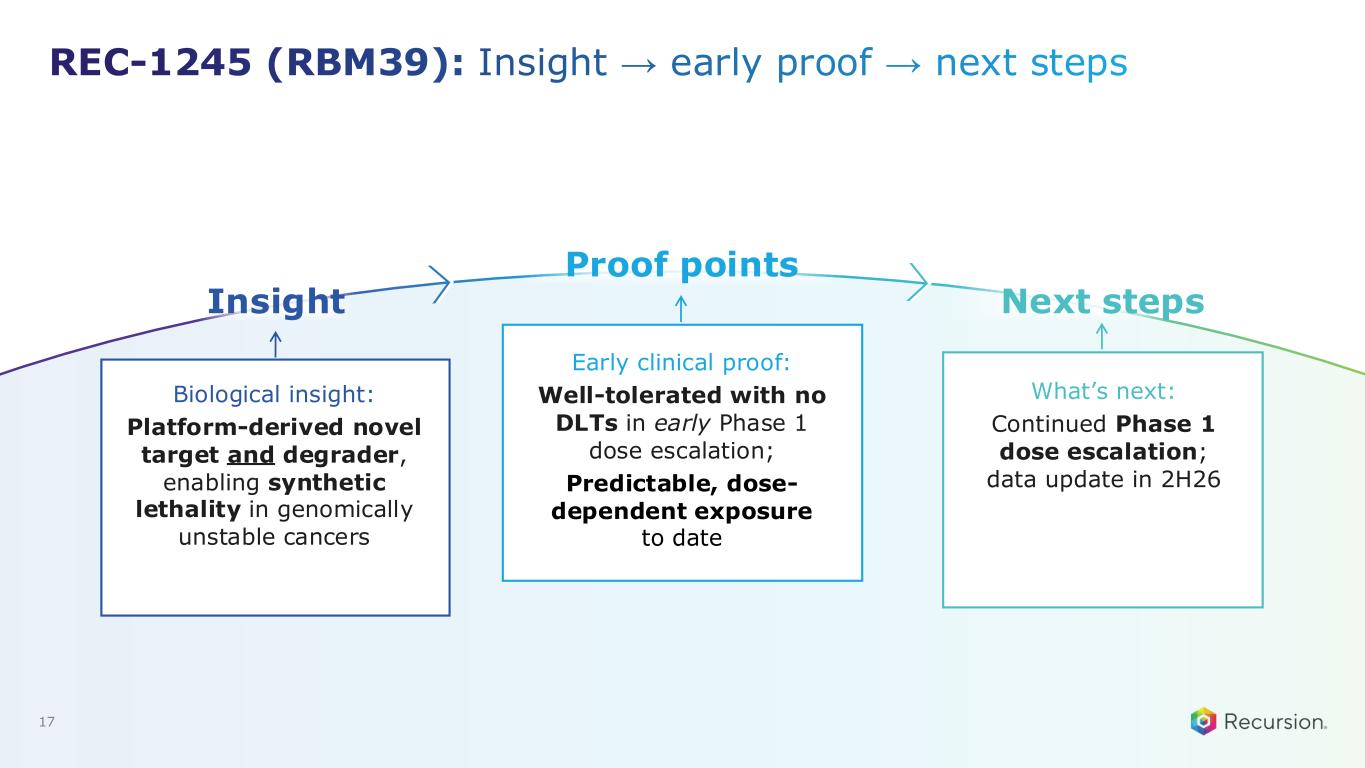
REC-1245 (RBM39): Insight → early proof → next steps Biological insight: Platform-derived novel target and degrader, enabling synthetic lethality in genomically unstable cancers Early clinical proof: Well-tolerated with no DLTs in early Phase 1 dose escalation; Predictable, dose- dependent exposure to date What’s next: Continued Phase 1 dose escalation; data update in 2H26 17 Insight Proof points Next steps

REC-4539 Translating proof to products
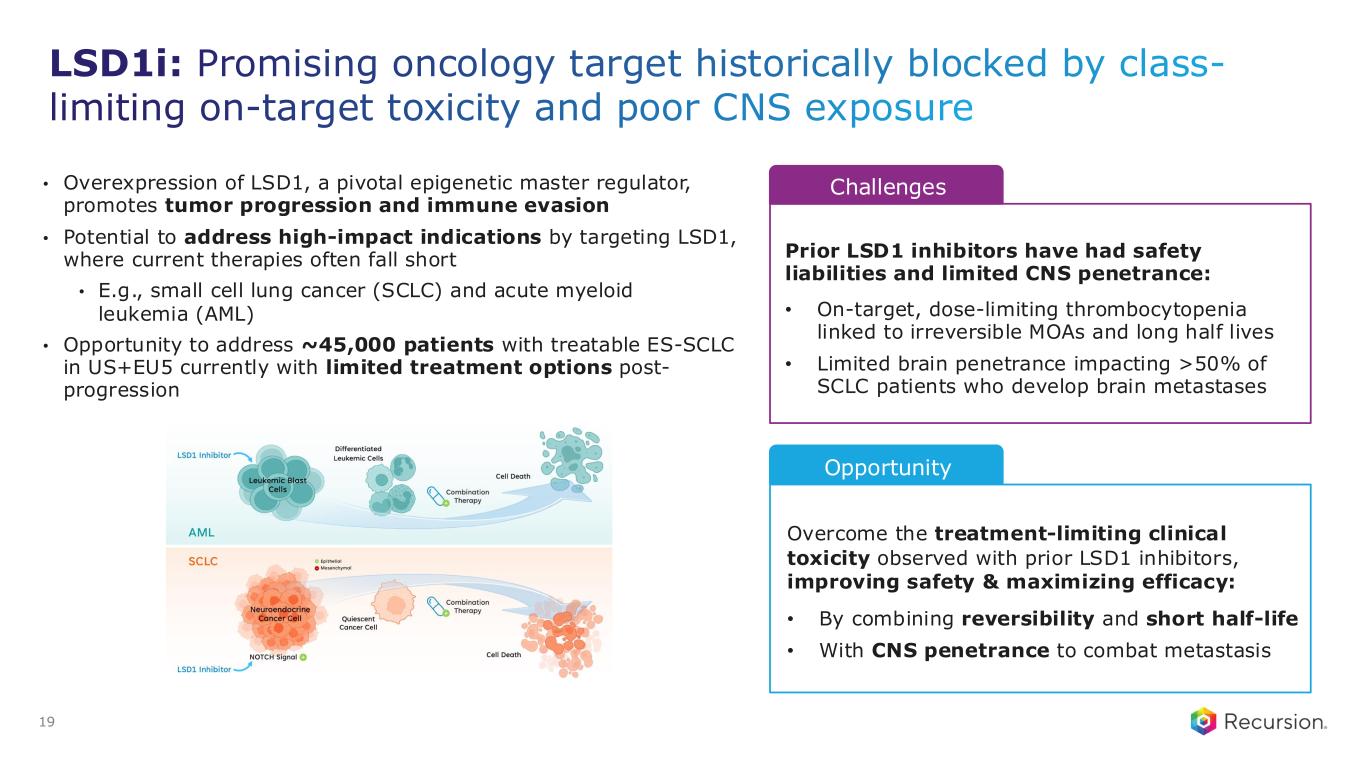
LSD1i: Promising oncology target historically blocked by class- limiting on-target toxicity and poor CNS exposure • Overexpression of LSD1, a pivotal epigenetic master regulator, promotes tumor progression and immune evasion • Potential to address high-impact indications by targeting LSD1, where current therapies often fall short • E.g., small cell lung cancer (SCLC) and acute myeloid leukemia (AML) • Opportunity to address ~45,000 patients with treatable ES-SCLC in US+EU5 currently with limited treatment options post- progression LS1 Opportunity Overcome the treatment-limiting clinical toxicity observed with prior LSD1 inhibitors, improving safety & maximizing efficacy: • By combining reversibility and short half-life • With CNS penetrance to combat metastasis 19 Challenges Prior LSD1 inhibitors have had safety liabilities and limited CNS penetrance: • On-target, dose-limiting thrombocytopenia linked to irreversible MOAs and long half lives • Limited brain penetrance impacting >50% of SCLC patients who develop brain metastases
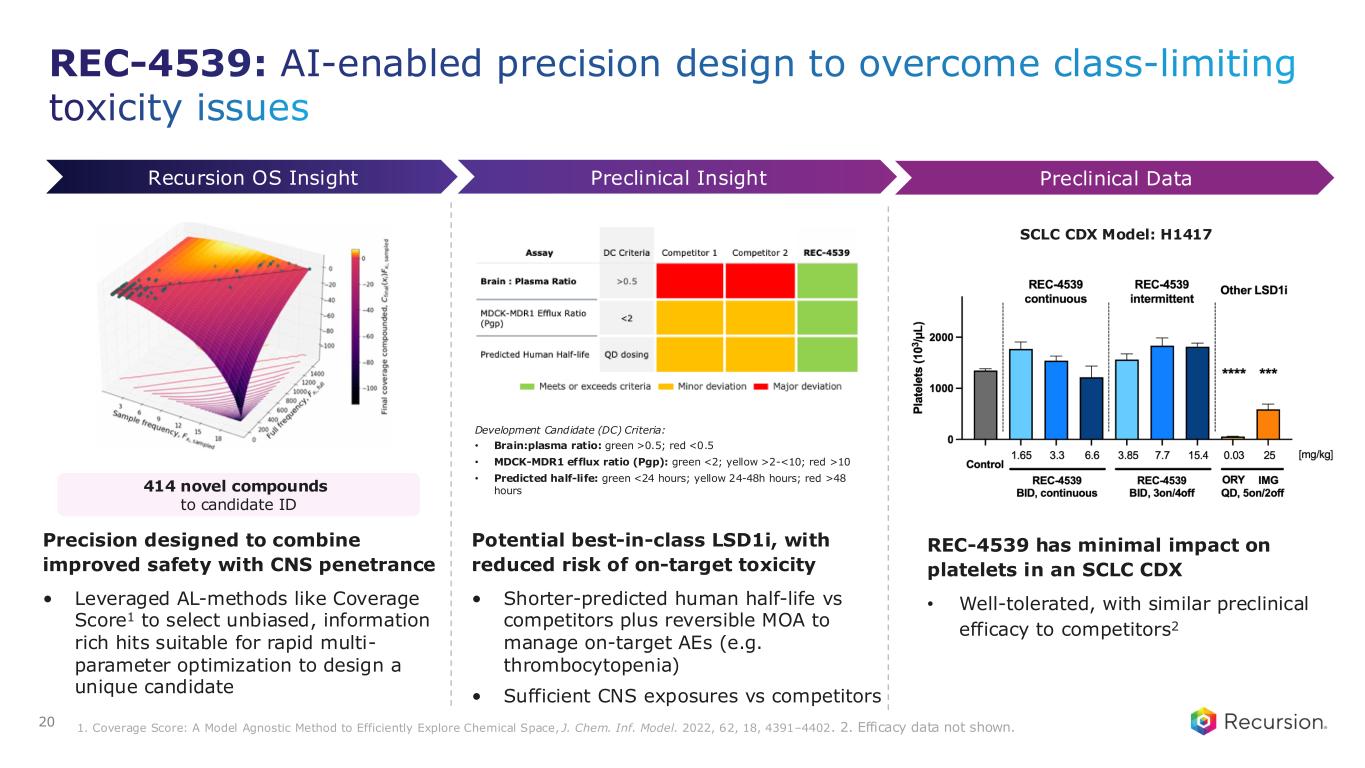
REC-4539: AI-enabled precision design to overcome class-limiting toxicity issues 20 REC-4539 has minimal impact on platelets in an SCLC CDX • Well-tolerated, with similar preclinical efficacy to competitors2 Preclinical InsightRecursion OS Insight Preclinical Data Precision designed to combine improved safety with CNS penetrance • Leveraged AL-methods like Coverage Score1 to select unbiased, information rich hits suitable for rapid multi- parameter optimization to design a unique candidate Potential best-in-class LSD1i, with reduced risk of on-target toxicity • Shorter-predicted human half-life vs competitors plus reversible MOA to manage on-target AEs (e.g. thrombocytopenia) • Sufficient CNS exposures vs competitors 414 novel compounds to candidate ID Development Candidate (DC) Criteria: • Brain:plasma ratio: green >0.5; red <0.5 • MDCK-MDR1 efflux ratio (Pgp): green <2; yellow >2-<10; red >10 • Predicted half-life: green <24 hours; yellow 24-48h hours; red >48 hours 1. Coverage Score: A Model Agnostic Method to Efficiently Explore Chemical Space, J. Chem. Inf. Model. 2022, 62, 18, 4391–4402. 2. Efficacy data not shown. SCLC CDX Model: H1417
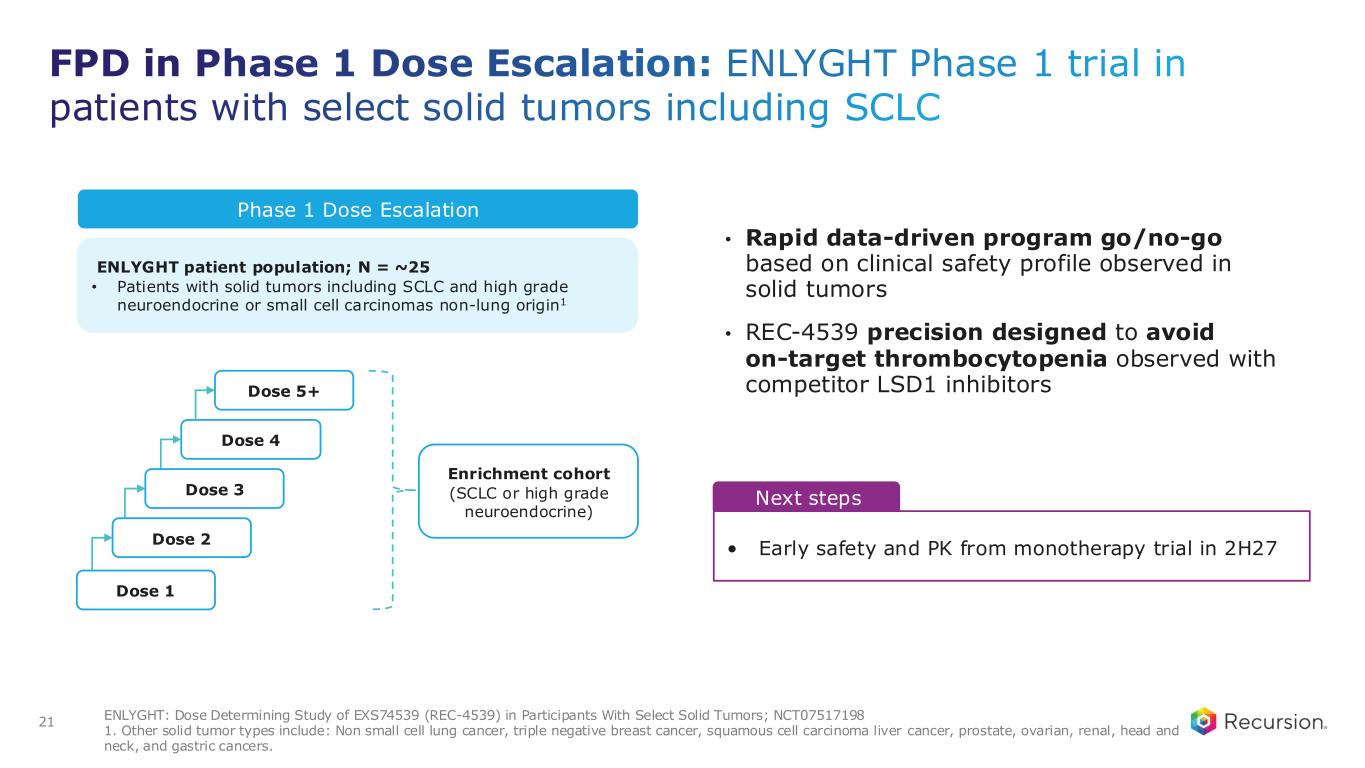
FPD in Phase 1 Dose Escalation: ENLYGHT Phase 1 trial in patients with select solid tumors including SCLC • Rapid data-driven program go/no-go based on clinical safety profile observed in solid tumors • REC-4539 precision designed to avoid on-target thrombocytopenia observed with competitor LSD1 inhibitors 21 ENLYGHT: Dose Determining Study of EXS74539 (REC-4539) in Participants With Select Solid Tumors; NCT07517198 1. Other solid tumor types include: Non small cell lung cancer, triple negative breast cancer, squamous cell carcinoma liver cancer, prostate, ovarian, renal, head and neck, and gastric cancers. Phase 1 Dose Escalation ENLYGHT patient population; N = ~25 • Patients with solid tumors including SCLC and high grade neuroendocrine or small cell carcinomas non-lung origin1 Dose 1 Dose 2 Dose 3 Dose 4 Dose 5+ Enrichment cohort (SCLC or high grade neuroendocrine) Next steps • Early safety and PK from monotherapy trial in 2H27
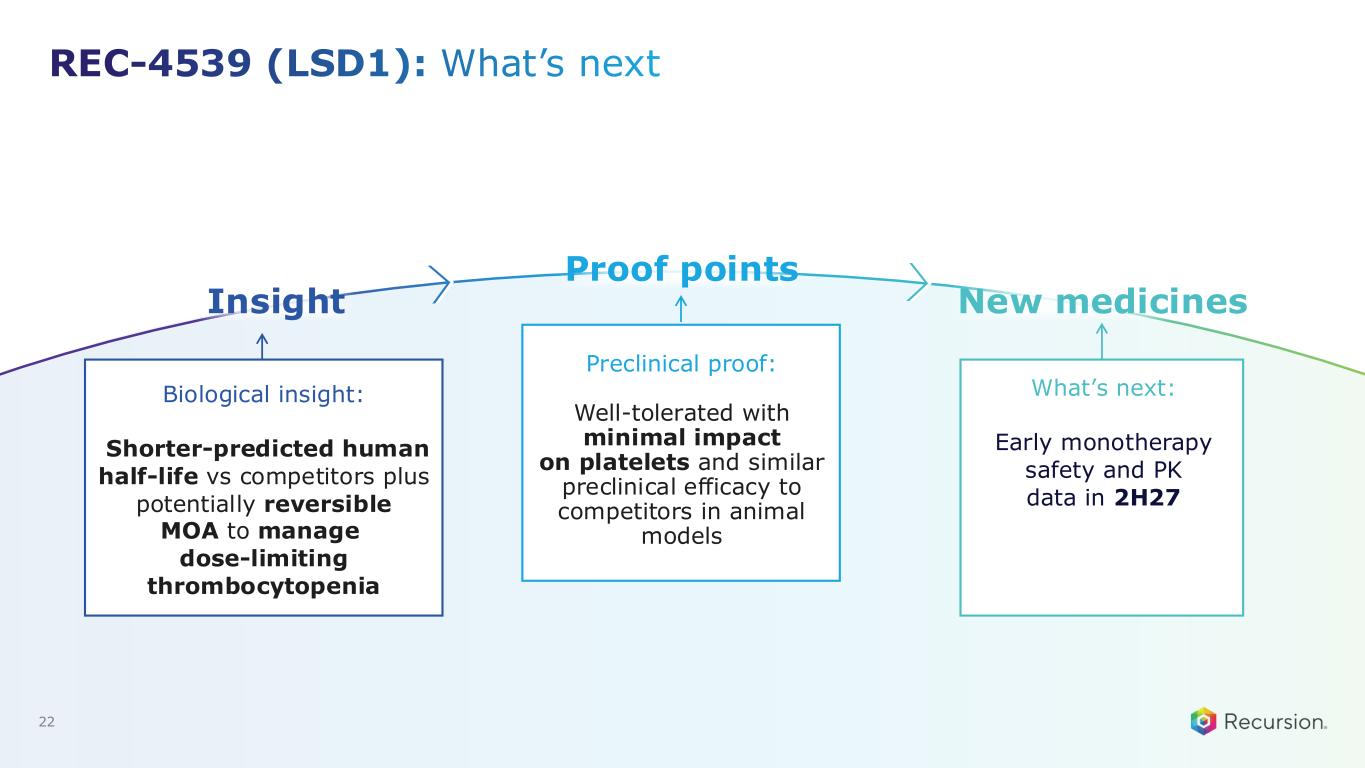
REC-4539 (LSD1): What’s next Biological insight: Shorter-predicted human half-life vs competitors plus potentially reversible MOA to manage dose-limiting thrombocytopenia Preclinical proof: Well-tolerated with minimal impact on platelets and similar preclinical efficacy to competitors in animal models What’s next: Early monotherapy safety and PK data in 2H27 22 Insight Proof points New medicines

Recursion OS Platform Scaling a differentiated AI-native product engine
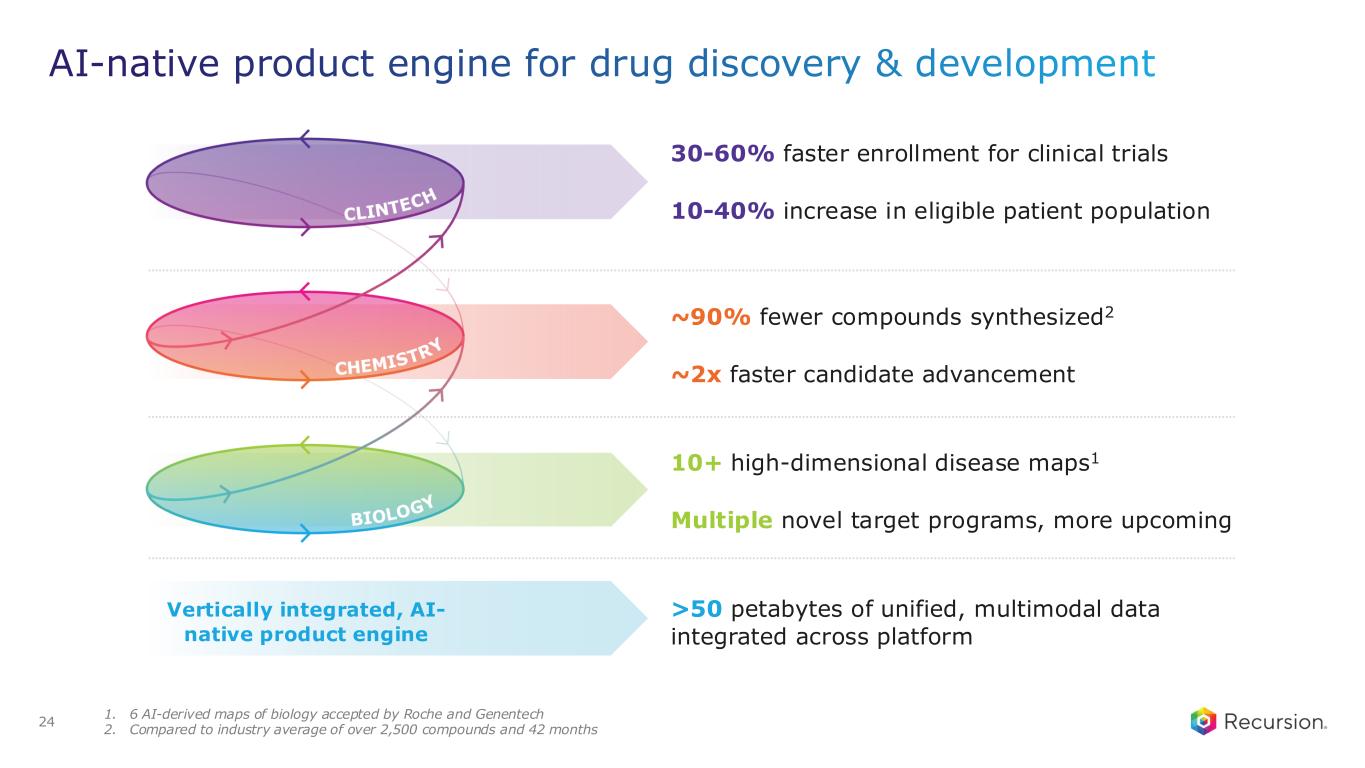
30-60% faster enrollment for clinical trials 10-40% increase in eligible patient population ~90% fewer compounds synthesized2 ~2x faster candidate advancement AI-native product engine for drug discovery & development 24 1. 6 AI-derived maps of biology accepted by Roche and Genentech 2. Compared to industry average of over 2,500 compounds and 42 months 10+ high-dimensional disease maps1 Multiple novel target programs, more upcoming Vertically integrated, AI- native product engine >50 petabytes of unified, multimodal data integrated across platform
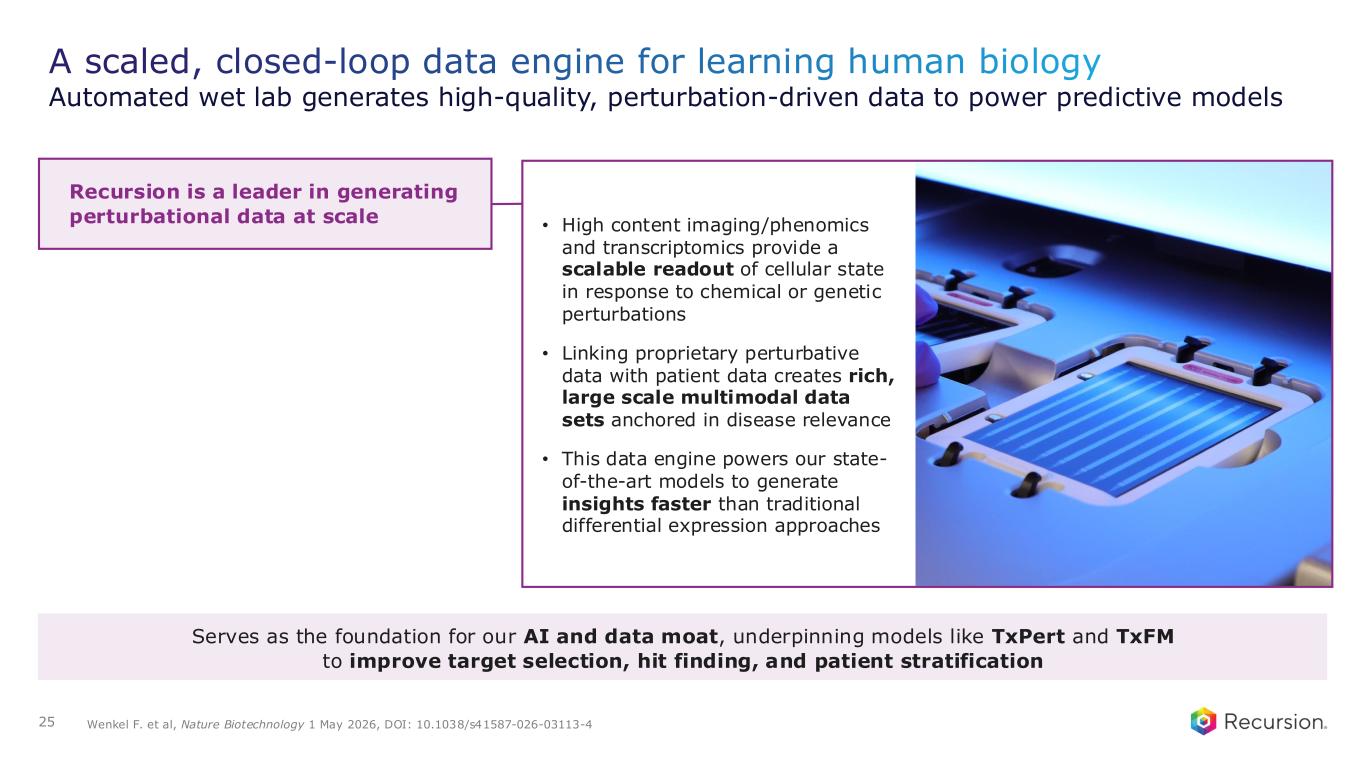
A scaled, closed-loop data engine for learning human biology Automated wet lab generates high-quality, perturbation-driven data to power predictive models 25 Wenkel F. et al, Nature Biotechnology 1 May 2026, DOI: 10.1038/s41587-026-03113-4 Recursion is a leader in generating perturbational data at scale • High content imaging/phenomics and transcriptomics provide a scalable readout of cellular state in response to chemical or genetic perturbations • Linking proprietary perturbative data with patient data creates rich, large scale multimodal data sets anchored in disease relevance • This data engine powers our state- of-the-art models to generate insights faster than traditional differential expression approaches Serves as the foundation for our AI and data moat, underpinning models like TxPert and TxFM to improve target selection, hit finding, and patient stratification

TxPert: Predicting biology before we run the experiment New model predicts transcriptomic readouts for unseen perturbations 26 Wenkel F. et al, Nature Biotechnology 1 May 2026, DOI: 10.1038/s41587-026-03113-4 TxPert predicts cellular response to unseen perturbations Learns complex biology, moving beyond simple pattern recognition • Grounded in biological knowledge graphs, including proprietary perturbation maps Generalizes beyond training data • SOTA published model in predicting unseen single perturbations; also on combinatorial effects and unseen cell contexts Approaching experimental accuracy • Predicted experimental results across 3 of 4 standard benchmark cell lines Published in Nature Biotechnology Application: Models like TxPert unlock perturbation space too vast to test experimentally—predicting and prioritizing the right experiments before running them to improve speed, cost, and probability of success
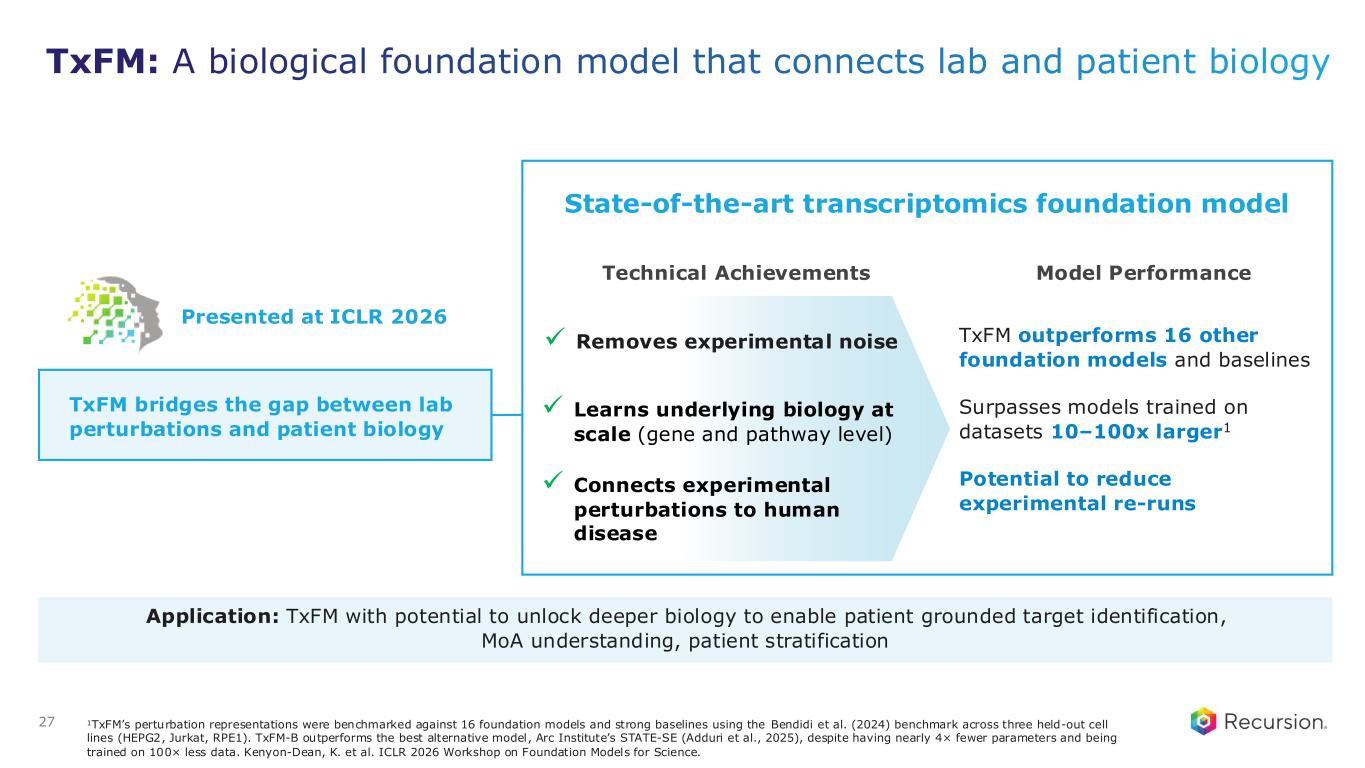
27 State-of-the-art transcriptomics foundation model Technical Achievements Model Performance TxFM outperforms 16 other foundation models and baselines Surpasses models trained on datasets 10–100x larger1 Potential to reduce experimental re-runs ✓ Removes experimental noise ✓ Learns underlying biology at scale (gene and pathway level) ✓ Connects experimental perturbations to human disease TxFM bridges the gap between lab perturbations and patient biology 1TxFM’s perturbation representations were benchmarked against 16 foundation models and strong baselines using the Bendidi et al. (2024) benchmark across three held-out cell lines (HEPG2, Jurkat, RPE1). TxFM-B outperforms the best alternative model, Arc Institute’s STATE-SE (Adduri et al., 2025), despite having nearly 4× fewer parameters and being trained on 100× less data. Kenyon-Dean, K. et al. ICLR 2026 Workshop on Foundation Models for Science. Application: TxFM with potential to unlock deeper biology to enable patient grounded target identification, MoA understanding, patient stratification TxFM: A biological foundation model that connects lab and patient biology Presented at ICLR 2026

Financials Pairing bold ambition with disciplined execution

Cash runway to deliver on upcoming milestones 29 1. Cash, cash equivalents and restricted cash 2. Cash operating expense—defined as net cash used in operating activities less partnership inflows and transaction costs—is a non-GAAP financial measure. See Appendix for reconciliation of non-GAAP financial measures. 1Q26 expense and cash1 update • ~30% reduction in YoY cash opex2 to $85m in 1Q26 • $665 million in cash1 as of March 31, 2026 1Q26 partnership highlights • 5th Sanofi milestone: $4M for a lead series in a potential first-in-class oncology program Expected cash runway into early 2028, without additional financing Reiterating 2026 cash opex guidance of <$390m

Looking ahead

Expected upcoming milestones 2026 and 2027 pipeline and partnership catalysts 31 1. Data-driven decision for potential Phase 1 initiation Translate proof to products Partner & Product catalysts – 2026 & 2027 Potential for AI-driven molecules to reach development candidate and late-stage discovery milestones Translating AI-driven insights from biology maps into potential first-in-class programs REC-4539 (LSD1i) Mono - Early safety and PK 2H 2026 1H 2026 2H 2027 1H 2027 REC-4881 (MEK1/2i) Additional clinical data REC-617 (CDK7i) Combo - early safety and PK REC-3565 (MALT1i) Mono - early safety and PK REC-4881 (MEK1/2i) Update on FDA engagement 2026 2027 Go/no-go decision1 REC-7735 (PI3K⍺ H1047Ri) REC-102 (ENPP1i) REC-1245 (RBM39 degrader) Additional Ph 1 dose escalation data REC-4881 (MEK1/2i) Initial engagement with FDA REC-1245 (RBM39 degrader) Mono - early safety and PK Scaling a differentiated AI-native product engine

32 THANK YOU


Non-GAAP Financial Measures To supplement our financial statements prepared in accordance with U.S. GAAP, we monitor and consider operating cash burn, which is a non-GAAP financial measure. We define operating cash expense as the net cash used in operating activities, excluding non-ordinary course transaction costs and partnership cash inflows. This non-GAAP financial measure is not based on any standardized methodology prescribed by U.S. GAAP and is not necessarily comparable to similarly- titled measures presented by other companies. We believe operating cash expense to be a liquidity measure that provides useful information to management and investors about the amount of cash consumed by the operations of the business. A limitation of using this non-U.S. GAAP measure is that operating cash expense does not represent the total change in cash and cash equivalents for the period because it excludes cash provided by or used for other investing and financing activities. We account for this limitation by providing information about our capital expenditures and other investing and financing activities in the statements of cash flows in our financial statements. Additionally, we reconciled operating cash burn below to net cash used in operating activities, the most directly comparable U.S. GAAP financial measure. In addition, it is important to note that other companies, including companies in our industry, may not use operating cash expense, may calculate operating cash expense in a different manner than we do or may use other financial measures to evaluate their performance, all of which could reduce the usefulness of operating cash expense as a comparative measure. Because of these limitations, operating cash burn should not be considered in isolation from, or as a substitute for, financial information prepared in accordance with U.S. GAAP. Appendix 34