.3

Corporate Responsibility & P|ESG Report 2025 www.ascendispharma.com
.3

Corporate Responsibility & P|ESG Report 2025 www.ascendispharma.com

Patients| Environmental, Social and Governance Report (P|ESG) Introduction Introduction 03 A message from our Executive Management Team 04 Who We Are 05 Mission & Vision 06 Innovation 07 A Brief History 08 Global Presence 09 Products, Programs & Pipeline P|ESG Approach P|ESG Approach 20 2025 Corporate Responsibility & P|ESG Highlights 21 Our Corporate Responsibility & P|ESG Approach 23 Key Topics 24 P|ESG Risks Impact Impact 25 Patients 32 Environmental 36 Social 42 Governance Ambitions Ambitions 50 2026 Corporate Responsibility & P|ESG Ambitions This report, included in the Ascendis Pharma A/S 2025 Annual Report, covers the Corporate Responsibility & P|ESG activities across all Ascendis Pharma Group entities for the year 2025. Through this report, we fulfill our compliance with Section 99a (CSR), Section 99b (Gender Diversity at Group Level), and Section 99d (Data Ethics) of the Danish Financial Statements Act. While some topics discussed in this report are significant, they should not be interpreted as material under U.S. federal securities laws and regulations. The concept of materiality used in this report reflects the interests of various stakeholders and other relevant definitions, which may differ from those applied under U.S. securities law. In addition, certain P|ESG information is based on methodologies and data that continue to evolve and are therefore subject to uncertainty and change over time. 2

A message from our Executive Management Team At Ascendis Pharma, we remain dedicated to improving the lives of patients by applying our innovative TransCon® technology platform to Introduction develop new therapies demonstrating best-in-class potential to address unmet medical needs. Corporate responsibility is embedded in how we operate and in our ability to provide safe, With our innovative technology effective treatments for the people who depend “ and passion for listening to on us. As our organization expands, we recognize that our influence – on both local and global patients and following the communities and on the environment – also science to address major P|ESG Approach increases. unmet medical needs, Ascendis Pharma is uniquely positioned to This continued evolution drives us to regularly review and strengthen our ESG reporting design and advance only those practices to ensure that our P|ESG framework therapies demonstrating develops in step with our growth and the best-in-class potential to benefit expectations of our stakeholders. Our intention health and well-being. is to prioritize the areas where we can create the Jan Mikkelsen Impact greatest positive impact. Founder, President & CEO Ascendis Pharma This report highlights our 2025 performance and reaffirms our dedication to making ongoing At Ascendis Pharma, we’re just getting started. progress in our P|ESG journey in alignment with current and emerging requirements. “ We are grateful to everyone who has contributed Ambitions to this work and look forward to building on these foundations in the years ahead. 3

Who We Are Ascendis Pharma was founded in 2007. We are a global biopharmaceutical company focused on applying our innovative TransCon® technology platform to make a meaningful difference for patients and their families. Guided by our core values of Patients, Science and Passion, and following our algorithm for product innovation, we develop TransCon® based therapies that demonstrate best-in-class potential to address major unmet medical needs. Our portfolio of Endocrinology Rare Disease approved products and product candidates addresses hypoparathyroidism and growth disorders. To create additional value, we have established partnerships to develop and bring to market TransCon® based products in large therapeutic areas, including Metabolic and Cardiovascular diseases and Ophthalmology. Our business model is built on fast, successful drug development and commercial therapeutic synergies. With an expanding global presence, we are positioned to reach patients worldwide. In the U.S., we have established a multifaceted organization to support ongoing commercialization efforts and serve as a foundation for future endocrinology rare disease product launches. In Europe, we are establishing our presence by building integrated organizations to commercialize approved Endocrinology Rare Disease products in select countries, including DACH (Germany, Austria & Switzerland), France & BeNeLux (Belgium, the Netherlands and Luxembourg), Iberia (Portugal & Spain), Italy, Nordics (Denmark, Norway, Sweden, Iceland & Finland), and the United Kingdom & Ireland. Beyond the U.S. and Europe “Direct Markets”, we are expanding global reach for our Endocrinology Rare Disease products through exclusive sales and distribution agreements with geographic market leaders, which we call “International Markets”. We currently have agreements covering over 75 countries. Finally, we are making our Endocrinology Rare Disease products commercially available in select markets under exclusive license agreements with partners with local development and commercialization expertise and infrastructure, which we call “Strategic Collaborations”. In Japan, Teijin, and in Greater China, VISEN, holds exclusive license rights to develop and commercialize TransCon® hGH, TransCon® PTH and TransCon® CNP. Ascendis Pharma in 2025 Introduction 18 Ascendis Pharma Direct Market Presence P|ESG €720M Approach Total Revenue 1,180+ Impact Employees 12 Clinical Development Ambitions Programs 4

Our Mission We are applying our innovative TransCon® technology platform to develop novel therapies demonstrating best-in-class potential to address unmet medical needs. Our Vision Introduction Our vision is to create new therapies focused on making a meaningful difference in patients’ lives. Vision 2030 P|ESG Achieve blockbuster status for multiple products and expand our engine for future innovation. Approach Be the Leading Endocrinology Rare Disease Company • Achieve >€5B for TransCon® PTH, TransCon® hGH and TransCon® CNP through worldwide commercialization. • Be the leader in Growth Disorders and Hypoparathyroidism, pursuing clinical conditions, innovative LCM and complementary patient offerings. • Expand pipeline with Endocrinology Rare Disease blockbuster product opportunities. Impact Create Value in Additional Therapeutic Areas through Innovative Business Models • Obtain accelerated approval in oncology with registrational trials ongoing. • Pursue TransCon® product opportunities in >€5B indications. • Maximize value creation of these product opportunities through collaboration with therapeutic area market leaders. Differentiate with Ascendis Fundamentals Ambitions • Outperform industry drug development benchmarks with Ascendis’ product innovation algorithm. • Remain independent as a profitable biopharma through lean and flexible ways of working. • Let our values Patients, Science, Passion drive our decisions to success. 5
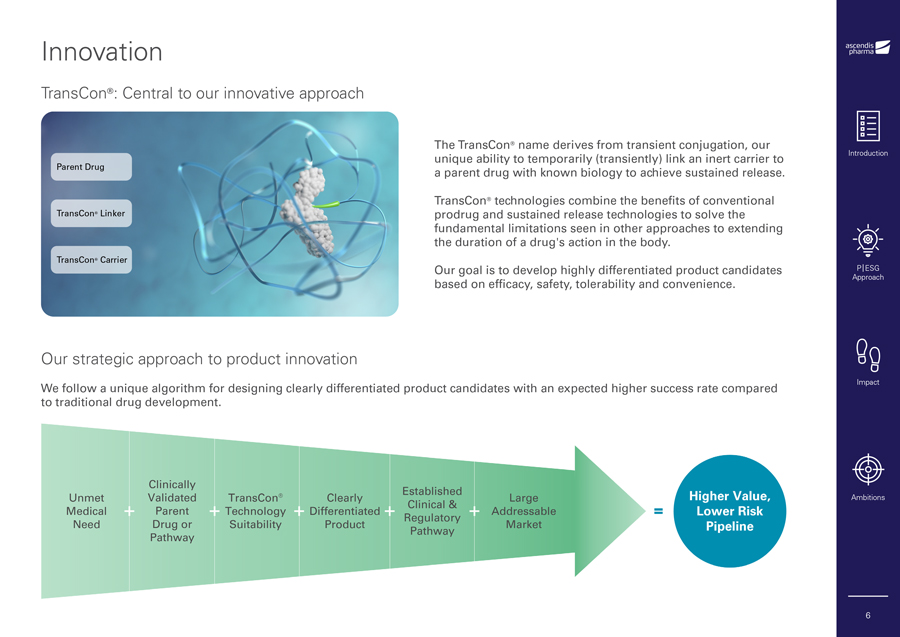
Innovation TransCon®: Central to our innovative approach The TransCon® name derives from transient conjugation, our unique ability to temporarily (transiently) link an inert carrier to Introduction Parent Drug a parent drug with known biology to achieve sustained release. TransCon® technologies combine the benefits of conventional TransCon® Linker prodrug and sustained release technologies to solve the fundamental limitations seen in other approaches to extending the duration of a drug’s action in the body. TransCon® Carrier Our goal is to develop highly differentiated product candidates P|ESG based on efficacy, safety, tolerability and convenience. Approach Our strategic approach to product innovation We follow a unique algorithm for designing clearly differentiated product candidates with an expected higher success rate compared Impact to traditional drug development. Ambitions 6
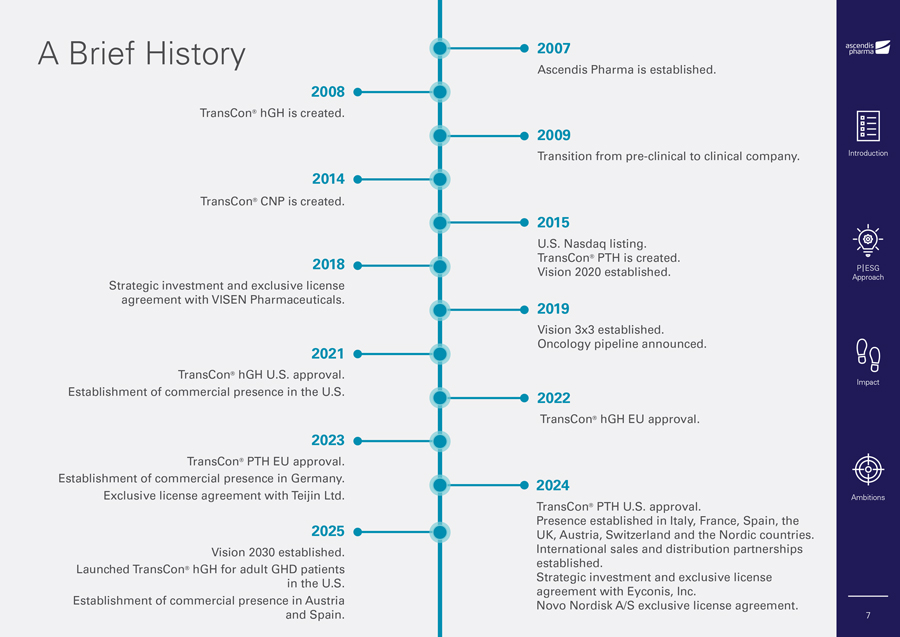
A Brief History 2007 Ascendis Pharma is established. 2008 TransCon® hGH is created. 2009 Transition from pre-clinical to clinical company. Introduction 2014 TransCon® CNP is created. 2015 U.S. Nasdaq listing. 2018 TransCon® PTH is created. Vision 2020 established. P|ESG Approach Strategic investment and exclusive license agreement with VISEN Pharmaceuticals. 2019 Vision 3x3 established. Oncology pipeline announced. 2021 TransCon® hGH U.S. approval. Impact Establishment of commercial presence in the U.S. 2022 TransCon® hGH EU approval. 2023 TransCon® PTH EU approval. Establishment of commercial presence in Germany. 2024 Exclusive license agreement with Teijin Ltd. Ambitions TransCon® PTH U.S. approval. Presence established in Italy, France, Spain, the 2025 UK, Austria, Switzerland and the Nordic countries. Vision 2030 established. International sales and distribution partnerships established. Launched TransCon® hGH for adult GHD patients Strategic investment and exclusive license in the U.S. agreement with Eyconis, Inc. Establishment of commercial presence in Austria Novo Nordisk A/S exclusive license agreement. and Spain. 7
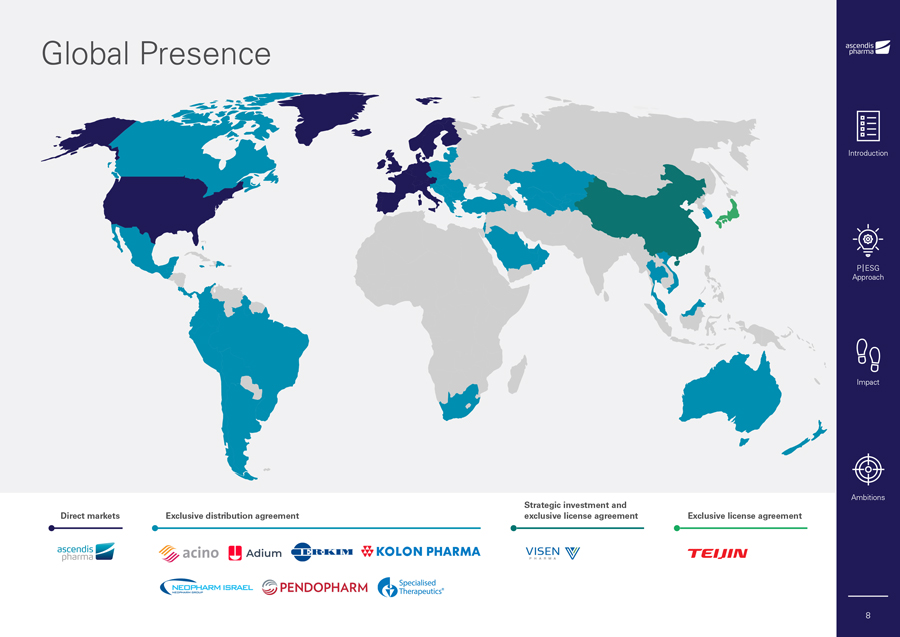
Global Presence Introduction P|ESG Approach Impact Ambitions Strategic investment and Direct markets Exclusive distribution agreement exclusive license agreement Exclusive license agreement 8

Introduction P|ESG Approach Products, Programs Impact & Pipeline Ambitions We currently have two marketed products and a diversified portfolio of product candidates in clinical development. 9

Endocrinology Rare Disease We built our first pipeline in Endocrinology Rare Diseases because it is an area where we saw the potential of TransCon® technology to address unmet medical needs. Each of our endocrinology product candidates has best-in-class potential, and each leverages established biology to help make a difference for patients. Introduction Growth Hormone Deficiency (GHD) Compound Trade Name Authorized in Available in Skytrofa® United States United States lonapegsomatropin* Developed asTransCon® hGH European Union & EEA** Austria & Germany P|ESG Approach *lonapegsomatropin-tcgd in the U.S. **EEA = European Economic Area Hypoparathyroidism Compound Trade Name Authorized in Available in Impact Yorvipath® United States United States palopegteriparatide Developed asTransCon® PTH European Union, EEA & the UK** Austria, Germany & Spain **EEA = European Economic Area. UK = United Kingdom Achondroplasia Ambitions Compound Trade Name Investigational Available in N/A Submitted NDA to FDA navepegritide N/A Developed asTransCon® CNP Submitted MMA to EMA Note: This overview is for Ascendis Pharma Direct Markets only. 10
Growth Hormone Deficiency What is Growth Hormone Deficiency (GHD)? Growth hormone is essential in overall endocrine health, promoting growth and maintaining normal body composition, cardiovascular function, and metabolism. For children with GHD, their bodies do not make or release enough growth hormone to keep up with normal growth, strength or body fat distribution. Children with GHD are characterized by short stature, metabolic and cardiovascular abnormalities, cognitive deficiencies, and poor quality of life. Children with GHD may experience: • Short stature • Delayed puberty • Bone development challenges • Decreased muscle mass and weakness • Emotional challenges • Low blood sugar • Reduced strength and endurance GHD is the most common pituitary hormone deficiency in children, with a prevalence of approximately 1 in 4,000 to 1 in 10,000.1,2 Nearly all forms of congenital GHD are believed to affect males and females equally, yet most societies express greater concern for short stature in males. As a result, the diagnosis of GHD favors males over females. In the National Cooperative Growth Study, 73% of individuals diagnosed with idiopathic GHD were male.3 1. Brinkman JE, et al. Physiology, Growth Hormone. In: StatPearls. Treasure Island (FL): StatPearls Publishing; May 1, 2023. 2. Brod M, et al. Qual Life Res. 2017;26(7):1673-86. 3. National Organization for Rare Disorders (NORD). Rare Disease Database: Growth Hormone Deficiency. Introduction P|ESG Approach Impact Ambitions 11

Growth Hormone Deficiency 2025 Highlights • Since its launch in 2021, Skytrofa® has been prescribed to more than 11,000 patients. • Received FDA approval of Skytrofa® for adult growth hormone deficiency, which represents the first of multiple planned label expansions. • Phase 3 trials with TransCon® CNP in achondroplasia and hypochondroplasia. • Initiated Phase 3 basket trial in four additional indications: idiopathic short stature (ISS), SHOX deficiency, Turner syndrome, and small for gestational age (SGA). About Skytrofa® TransCon® hGH is a prodrug composed of somatropin that is transiently bound to a carrier by a proprietary linker. TransCon® hGH is administered once weekly and is designed to maintain the same mode of action as daily therapies by providing sustained release of active, unmodified somatropin, the same recombinant growth hormone molecule used in the daily hGH therapies that have historically been the standard of care. Our first marketed product, Skytrofa® (lonapegsomatropin; developed as TransCon® hGH) is approved by the FDA for the treatment of pediatric patients one year and older who weigh at least 11.5 kg and have growth failure due to inadequate secretion of endogenous growth hormone, also known as growth hormone deficiency or GHD. Skytrofa® is also approved by the FDA for replacement of endogenous growth hormone in adults with GHD. The European Commission (EC) authorized Skytrofa® in the EU for the treatment of children and adolescents 3-18 years of age with growth failure due to GHD. Skytrofa® has also been authorized by other regulatory authorities globally. Skytrofa® is commercially available for prescription in Ascendis Pharma Direct Markets, the U.S., and Germany. Introduction P|ESG Approach Impact Ambitions 12

Hypoparathyroidism What is Hypoparathyroidism? Hypoparathyroidism is a rare endocrine disease, caused by insufficient levels of parathyroid hormone (PTH) in the body.1 The parathyroid glands lie behind the thyroid gland in the neck. They produce PTH, which is the primary regulator of calcium and phosphate in the body by acting directly on bones and kidneys and indirectly on the intestine.2,3 If the parathyroid glands are removed, destroyed or defective, this may lead to insufficient levels of PTH.2,4 Hypoparathyroidism can arise from genetic causes, autoimmune causes and other causes. Most commonly, hypoparathyroidism results following neck surgery constituting approximately 75% of all cases.2,5 The number of individuals living withhypoparathyroidism is estimated to be ~0.8M—1.1M across Ascendis Pharma’s geographic regions*. Hypoparathyroidism affectsnumerous systems in the body and is associated with a range of short-term symptoms and long-term complications.2,4,6 Brain • Symptoms of anxiety and depression • Fatigue • Cognitive impairment, ‘brain fog’ Central nervous system • Seizures • Calcifications • Parkinsonism or dystonia Lungs • Laryngospasm Kidneys** • Nephrocalcinosis • Kidney stones • Chronic kidney disease Peripheralnervous system • Paresthesia • Muscle cramps • Tetany Eyes • Cataracts • Papilledema Teeth • Altered tooth morphology Heart • Cardiac arrhythmias • Hypocalcaemia associated dilated cardiomyopathy Skin • Dry skin •Pustular psoriasis Brittle nails and prone to onycholysis • Coarse, thin hair Musculoskeletal • Myopathy • Spondyloarthropathy * Germany, Austria, France, Spain, Portugal, Italy, United Kingdom, Switzerland, Belgium, Netherlands, Luxembourg, Denmark, Finland, Iceland, Norway, Sweden, U.S., LATAM/Canada, CEE/Eurasia, MENA, ANZ, Israel, South Korea, Japan, China. ** These manifestations are mostly the result of treatment with calcium and active vitamin D rather than of the disease itself. 1. Bollerslev et al. European Society of Endocrinology Clinical Guideline: Treatment of chronic hypoparathyroidism in adults. Eur J Endocrinol. 2015 Aug;173(2):G1-20. 2. Brandi ML et al. Summary Statement and Guidelines. The Journal of Clinical Endocrinology & Metabolism. 2016 Jun 1;101(6):2273–83. 3. Chen K et al. Clinical burden and healthcare resource utilization among patients with chronic hypoparathyroidism, overall and by adequately vs not adequately controlled disease: a multi-country chart review. Journal of Medical Economics. 2019 Jun 17;22(11):1141–52. 4. Mannstadt M et al. Hypoparathyroidism. Nat Rev Dis Primers. 2017 Aug 31;3:17055. 5. Clarke BL, et al. Epidemiology and Diagnosis of Hypoparathyroidism, The Journal of Clinical Endocrinology & Metabolism, Volume 101, Issue 6, 1 June 2026, Pages 2284-2299. 6. Shoback DM et al. Presentation of Hypoparathyroidism: Etiologies and Clinical Features. J Clin Endocrinol Metab. 2016;101(6):2300-12. Introduction P|ESG Approach Impact Ambitions 13

Hypoparathyroidism 2025 Highlights • 2025 was the first full year of the U.S. launch, with more than 5,300 patients being prescribed Yorvipath® by more than 2,400 unique prescribers. • Outside the U.S. and select EU countries, we have expanded our international sales footprint with distribution agreements in place covering more than 75 countries and a commercial launch in Japan through our partner Teijin. • The PaTHway60 Trial is an ongoing safety and efficacy trial to enable dose titration up to 60 mcg/day in adults with hypoparathyroidism, intended to support U.S. label expansion. • The PaTHway Adolescent Trial is an ongoing trial intended to support U.S. label expansion to include the treatment of adolescents with hypoparathyroidism 12 to less than 18 years of age. About Yorvipath® TransCon® PTH (palopegteriparatide) is a prodrug of PTH (1-34), administered once daily and designed to provide sustained release of active PTH to maintain continuous exposure within the physiological range for 24 hours/day in individuals with hypoparathyroidism, thereby more fully addressing aspects of the disease. Yorvipath® (palopegteriparatide; developed as TransCon® PTH) is approved by the U.S. Food & Drug Administration (FDA) and authorized by the European Commission (EC) and other regulatory agencies for the treatment of adults with hypoparathyroidism. Yorvipath® is commercially available in the U.S. and Japan, and in the European Union (EU) in Germany, Austria and Spain. It is also available in more than 30 countries through Named Patient Programs. Introduction P|ESG Approach Impact Ambitions 14

Living with Hypoparathyroidism – Sabine’s Story Sabine has been living with hypoparathyroidism for 17 years. She is 66 years old. She describes her journey as a long and challenging path back to a self-determined life. Her story began in January 2008, when she underwent thyroid surgery because of multiple nodules. Shortly afterward, it was discovered that her parathyroid glands no longer worked. Overnight, her life changed. “I experienced constant fatigue and lethargy. What affected me most was the change in my personality. I barely recognized myself.” Sabine recalls. Tasks that once brought her joy, such as exercising or spending time with her family, suddenly became exhausting. Everyday activities, even walking short distances, left her breathless, with her heart racing and her energy depleted. She remembers feeling anxious in situations that had never worried her before, for example driving alone or going into the city. Her condition affected her family as well. Her husband had to take on additional responsibilities and provide continuous emotional support. At times he felt he no longer recognized the life they once shared, because the illness changed their routines and Sabine’s overall well-being. Conventional therapy with oral calcium and active vitamin D initially provided only short-term relief. Over time, Sabine became frustrated and saddened as she watched her quality of life decline. “I noticed every day what hypoparathyroidism was doing to me and to my body,” she explains. She struggled with muscle cramps, tingling and painful spasms in her hands. Fatigue and emotional strain added to the burden. The symptoms were invisible to others. Communicating the severity of her condition was often difficult. Sabine My motto is: I only have this “one life and even with a chronic condition, I want to make the very best of it. “ Introduction P|ESG Approach Impact Ambitions 15

Living with Hypoparathyroidism – Sabine’s Story Sabine remained determined to regain control. She actively sought help from specialists, wrote letters and advocated for the care she needed. “I reached a point where I was desperate. I wrote to my endocrinologist: I have palpitations. I feel depressed. I used to be agile and joyful. Now I am sad and unmotivated. Please help me.” Her persistence became the key to finding the right support and guidance. Today, Sabine looks back with resilience and hope. She emphasizes the importance of understanding and compassionate healthcare. “Hypoparathyroidism is not just about lab values. It affects every part of your life, physically, mentally and emotionally. It is crucial to find doctors who truly listen.” Sabine’s story shows how a chronic and invisible condition can change a person’s daily life, personality, and independence. Through determination, self-advocacy and appropriate care, she learned to navigate the challenges of hypoparathyroidism while protecting her spirit and sense of identity. Introduction P|ESG Approach Impact Ambitions 16

Achondroplasia What is Achondroplasia? Achondroplasia is a rare genetic condition arising from a systemic fibroblast growth factor receptor 3 (FGFR3) variant that leads to an imbalance in the effects of the FGFR3 and C-type natriuretic peptide signaling pathways, estimated to affect more than 250,000 people worldwide.1-11 While historically considered a skeletal condition, the FGFR3 variant seen in achondroplasia is expressed in tissues throughout the body, causing serious muscular, neurological, and cardiorespiratory complications in addition to skeletal dysplasia.1-8 Medical complications of achondroplasia vary across different stages of life.4,19 Throughout infancy and childhood, observed complications include spinal abnormalities, enlarged brain ventricles, impaired muscle strength and stamina, hearing deficits and chronic ear infections, upper airway obstructions, sleep-disordered breathing, hip problems, leg bowing, and chronic pain; many of these persist or worsen in adulthood.4,12,10 These medical complications can have detrimental effects on quality of life, physical functioning, and psychosocial function.1,2,4,18,19 Individuals with achondroplasia often require multiple surgeries and procedures to alleviate the condition’s many complications.6 Condition overview and medical complications A rare genetic condition that leads to well-characterized skeletal dysplasia and serious muscular, neurological, and cardiorespiratory complications. Achondroplasia arises from a systemic variant in the FGFR3 gene4,8 leading to the constitutive activation of FGFR3.9-11 FGFR3 is expressed in multiple tissues throughout the body.1-4,8 Achondroplasia is the most common form of short- limbed disproportionate short stature.4 Other clinical manifestations include:4,12,10 • Shortening of proximal long bones • Spinal abnormalities • Tibial bowing Achondroplasia affects more than 250,000 people worldwide2,5, with an estimated pediatric (<18 years) prevalence of: US – 3.5 in 100,000.a,14,15 Europe – 3.3 in 100,000.16,17 Clinical manifestations of achondroplasia are associated with a wide range of significant, potentially life-threatening medical complications that negatively impact health-related quality of life.1,2,4,18,19 CNP, c-type natriuretic peptide; FGFR3, fibroblast growth factor 3. a. Estimated prevalence rate is based on the United States Census Bureau; Komodo claims data (range from January 2016 to March 2025) in addition to Stevenson et al. 201210 and Waller et al. 2008.11 1. Ireland PJ, et al, Appl Clin Genet. 2014;7:117-25; 2. Horton WA, et al, Lancet. 2007; 2007;370(9582):162-72; 3. Sims D T, et al, J Appl Physiol. 2018; 124(3): 696-703; 4. Pauli RM, Orphanet J Rare Dis 2019; 5. Baujat G, et al, Best Pract Res Clin Rheumatol 2008; 22(1): 3-18. 6. Savarirayan R, et al, Nat Rev Endocrinol. 2022;18(3):173-89. 7. Cormier-Daire V, et al, OrphanetJ Rare Dis. 2022;17(1):293. 8. Cormier-Daire V, et al, Orphanet J Rare Dis. 2021;16(1):333. 9. Wrobel W, et al. Int J Mol Sci 2021; 22(11). 10. Rintz E, et al. Int J Mol Sci 2022; 23(11). 11. Krejci P, et al. PLoS One 2008; 3(12): e3961. 12. Hoover-Fong J, et al, Bone 2021; 146: 115872. 13. Shelmerdine SC, et al, Am J Med Genet A 2016; 170(8): 2039-43. 14. Stevenson DA, et al. Am J Med Genet A 2012; 158A(5): 1046-54. 15. Waller DK, et al. Am J Med Genet A 2008; 146A(18): 2385-9. 16. Coi A, et al. Am J Med Genet A. 2019; 179(9):1791–8. 17. Stoll C, et al. Eur J Med Genet. 2022; 65(11):104612. 18. Murton MC, et al. Adv Ther 2023; 40(9): 3639-80. 19. McGraw SA, et al, Adv Ther 2022; 39(7): 3378-91. Introduction P|ESG Approach Impact Ambitions 17

Achondroplasia 2025 Highlights TransCon® CNP • Submitted new drug application (NDA) to the U.S. Food and Drug Administration (FDA). • Submitted marketing authorization application (MAA) to the European Medicines Agency (EMA). • Pivotal ApproaCH Trial demonstrated significant improvements in linear growth and body proportionality, as well as benefits beyond linear growth compared to placebo. • Analyses from our pivotal ApproaCH Trial showed that children treated with TransCon® CNP had improvements in the Physical Functioning domain of the Achondroplasia Child Experience Measure (ACEM-PF), with greatest benefits in younger children who had the most pronounced leg bowing angulation (≥5 degrees) at baseline, supporting benefits beyond linear growth. • Long-term extension data suggests durable effect of TransCon® CNP monotherapy up to 3 years of treatment. TransCon® CNP & TransCon® hGH Dual Therapy • Week 26 data from our Phase 2 COACH trial studying TransCon® CNP in combination with TransCon® hGH demonstrated that TransCon® hGH boosted treatment benefits of TransCon® CNP, resulting in significant growth and proportionality improvements in children with achondroplasia, with a safety and tolerability profile consistent with those observed for TransCon® hGH and TransCon® CNP monotherapies. • Submitted a protocol and held an end-of-Phase 2 meeting with the FDA regarding a Phase 3 trial of TransCon® CNP and TransCon® hGH in pediatric achondroplasia. • Submitted an application to investigate TransCon® CNP alone and in combination with TransCon® hGH for the treatment of hypochondroplasia. About TransCon® CNP TransCon® CNP (navepegritide) is an investigational prodrug of C-type natriuretic peptide (CNP) administered once weekly, designed for continuous inhibition of the overactive FGFR3 pathway in achondroplasia by providing continuous exposure of active CNP to receptors on tissues throughout the body. TransCon® CNP is currently under review in the U.S. and the EU for the treatment of children with achondroplasia. About TransCon® CNP and TransCon® hGH Dual Therapy TransCon® CNP provides holistic treatment of achondroplasia and enables complementary effect of TransCon® hGH. In achondroplasia, overactive FGFR3 signaling acts as a brake, inhibiting bone growth1,2. TransCon® CNP releases the brake enabling GH to accelerate outcomes. 1. Rintz E, et al. Int J Mol Sci. 2022; 23(11); 2. Krejci P, et al. PLoS One. 2008; 3(12): E3961. Introduction P|ESG Approach Impact Ambitions 18
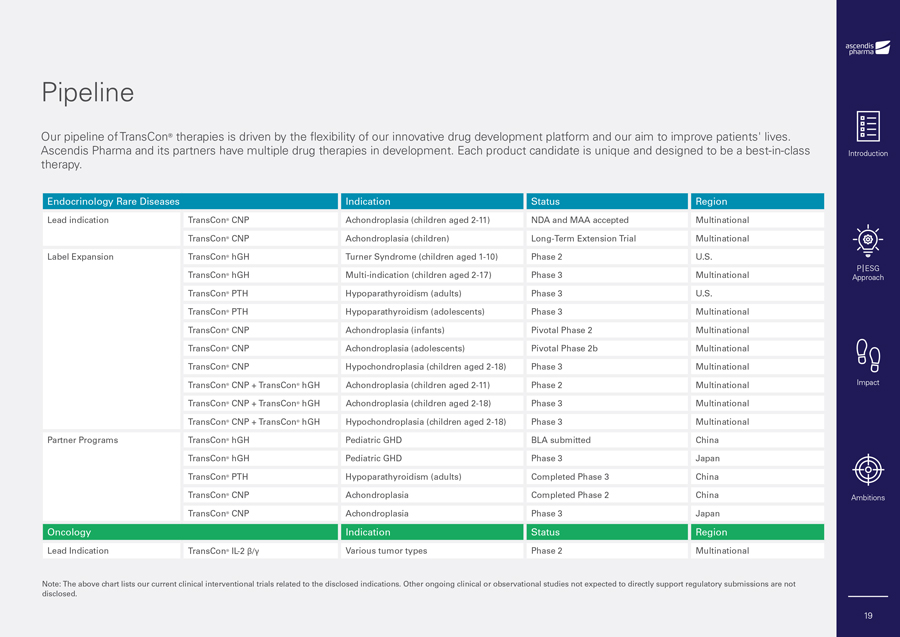
Pipeline Our pipeline of TransCon® therapies is driven by the flexibility of our innovative drug development platform and our aim to improve patients’ lives. Ascendis Pharma and its partners have multiple drug therapies in development. Each product candidate is unique and designed to be a best-in-class therapy. Endocrinology Rare Diseases Indication Status Region Lead indication TransCon® CNP Achondroplasia (children aged 2-11) NDA and MAA accepted Multinational TransCon® CNP Achondroplasia (children) Long-Term Extension Trial Multinational Label Expansion TransCon® hGH Turner Syndrome (children aged 1-10) Phase 2 U.S. TransCon® hGH Multi-indication (children aged 2-17) Phase 3 Multinational TransCon® PTH Hypoparathyroidism (adults) Phase 3 U.S. TransCon® PTH Hypoparathyroidism (adolescents) Phase 3 Multinational TransCon® CNP Achondroplasia (infants) Pivotal Phase 2 Multinational TransCon® CNP Achondroplasia (adolescents) Pivotal Phase 2b Multinational TransCon® CNP Hypochondroplasia (children aged 2-18) Phase 3 Multinational TransCon® CNP + TransCon® hGH Achondroplasia (children aged 2-11) Phase 2 Multinational TransCon® CNP + TransCon® hGH Achondroplasia (children aged 2-18) Phase 3 Multinational TransCon® CNP + TransCon® hGH Hypochondroplasia (children aged 2-18) Phase 3 Multinational Partner Programs TransCon® hGH Pediatric GHD BLA submitted China TransCon® hGH Pediatric GHD Phase 3 Japan TransCon® PTH Hypoparathyroidism (adults) Completed Phase 3 China TransCon® CNP Achondroplasia Completed Phase 2 China TransCon® CNP Achondroplasia Phase 3 Japan Oncology Indication Status Region Lead Indication TransCon® IL-2 â/ã Various tumor types Phase 2 Multinational Note: The above chart lists our current clinical interventional trials related to the disclosed indications. Other ongoing clinical or observational studies not expected to directly support regulatory submissions are not disclosed. Introduction P|ESG Approach Impact Ambitions 19

2025 Corporate Responsibility & P|ESG Highlights In 2025, we continued to advance our Corporate Responsibility and P|ESG efforts, focusing on strengthening existing processes and maintaining alignment with evolving regulatory and stakeholder expectations. Patients • Launched Yorvipath® in the U.S., Austria and Spain. • Launched TransCon® hGH for adult GHD patients in the U.S. • Submitted TransCon® CNP NDA to FDA. • Submitted TransCon® CNP MMA to EMA. • Launched second generation auto-injector for Skytrofa®. • Continued support through Named Patient Programs across Europe and International Markets for TransCon® PTH and TransCon® hGH. • Yorvipath® patient support launched in the U.S. • Continued collaborative relationships with Patient Advocacy groups. • Continued activities for long-term Skytrofa® registries in the U.S. and support to German registry INSIGHTS-GHT. • Launched the Patient Support Program in Spain for patients under Yorvipath® treatment. Environmental • Successfully reported on our Scope 1 and 2 emissions data, representing climate impacts related to on-site energy consumption and purchased electricity, heat and cooling. Social • Successfully hired and onboarded 319 employees. • Continued focus on the attraction and retention of talent. • Focus on capability building in the organization – delivered training programs on Leadership Development, Employeeship, Project Management and the Let’s Talk concept. Governance • Further strengthened compliance implementation to support decision-makers in doing the right thing for the patients. • Established Compliance Committees in European Direct Markets. • Finalized the first version of an approved list of vendors of animal matrices and implemented this list at internal laboratories. Introduction P|ESG Approach Impact Ambitions 20

Our Corporate Responsibility & P|ESG Approach Our 2025 Corporate Responsibility & P|ESG Report is structured around our four key focus areas: Patients, Environmental, Social and Governance. Through these pillars, we aim to uphold our environmental and social license to operate while supporting long-term value creation for both our operations and the patients we serve. About this Report This report reflects our continued commitment to transparency and accountability as we work toward alignment with evolving sustainability regulations, including the European Union’s Corporate Sustainability Reporting Directive (CSRD). We recognize the increasing expectations of the users of this report and our affected stakeholders and remain committed to meeting these responsibly. In 2024, we completed a pilot Double Materiality Assessment (DMA) to begin identifying and evaluating our potentially material impacts, risks and opportunities. While this provided valuable early insights, 2025 brought significant regulatory developments, particularly the ESRS revisions introduced through the Omnibus Directive, which resulted in substantial changes to reporting expectations and methodologies. Given this shifting landscape, we chose not to conduct a full DMA in 2025. Instead, we deliberately paused to allow the regulatory framework to stabilize and to ensure that our future assessment is performed using the most up-to-date requirements and best practices, while continuing to seek a meaningful balance between Ascendis Pharma’s strategic priorities and focus areas and the evolving reporting requirements. In the meantime, we remain committed to refining our methodology and improving the accuracy and relevance of our current and future disclosures. As part of our continued due diligence efforts, this report presents our key P|ESG topics, our primary P|ESG risks and an overview of the policies, processes and performance that underpin our work. It also outlines forward-looking ambitions across each pillar of our framework. Each section includes updates on the commitments we made last year, and the progress made toward achieving them. While not all ambitions were fully met due to a combination of internal priorities and external developments, we remain committed to continuous improvement and transparency as we navigate an evolving regulatory environment and a dynamic business landscape. We appreciate your understanding as we continue to mature our approach to corporate responsibility. Vision & Values Patients Patient & Caregiver Clinical Trials Product Safety Access & Affordability Engagement Environmental Social Governance • Climate Impact • Employee Engagement & Development • Business Conduct & Ethics • Suppliers & Business Partners • Inclusive Culture • Anti-Corruption & -Bribery • Health & Safety • Responsible Marketing • People in our Value Chain • Data Governance • Human Rights • Cybersecurity • Animal Welfare Due Diligence, Double Materiality Reporting & Communication Introduction P|ESG Approach Impact Ambitions 21
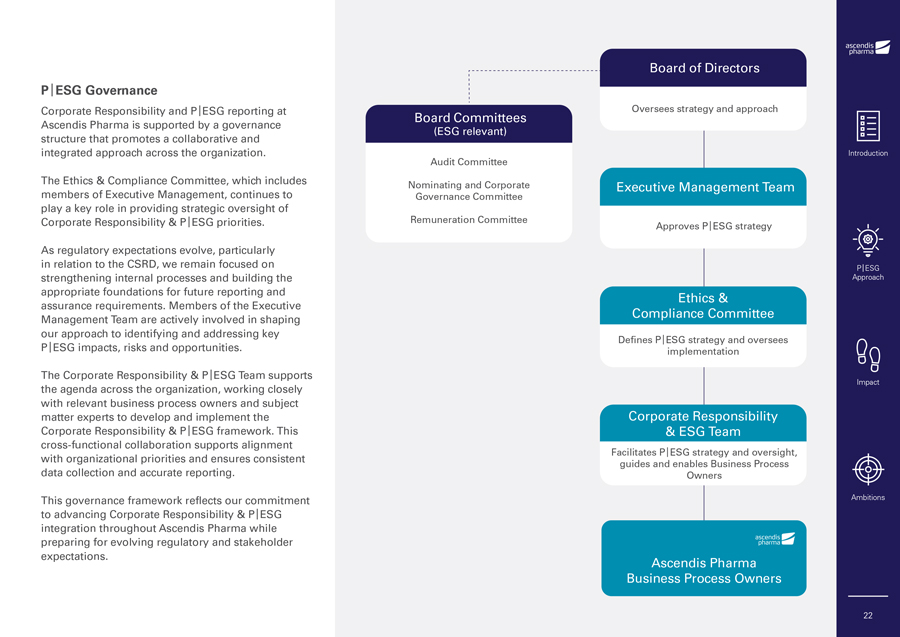
P|ESG Governance Corporate Responsibility and P|ESG reporting at Ascendis Pharma is supported by a governance structure that promotes a collaborative and integrated approach across the organization. The Ethics & Compliance Committee, which includes members of Executive Management, continues to play a key role in providing strategic oversight of Corporate Responsibility & P|ESG priorities. As regulatory expectations evolve, particularly in relation to the CSRD, we remain focused on strengthening internal processes and building the appropriate foundations for future reporting and assurance requirements. Members of the Executive Management Team are actively involved in shaping our approach to identifying and addressing key P|ESG impacts, risks and opportunities. The Corporate Responsibility & P|ESG Team supports the agenda across the organization, working closely with relevant business process owners and subject matter experts to develop and implement the Corporate Responsibility & P|ESG framework. This cross-functional collaboration supports alignment with organizational priorities and ensures consistent data collection and accurate reporting. This governance framework reflects our commitment to advancing Corporate Responsibility & P|ESG integration throughout Ascendis Pharma while preparing for evolving regulatory and stakeholder expectations. Board Committees (ESG relevant) Audit Committee Nominating and Corporate Governance Committee Remuneration Committee Board of Directors Oversees strategy and approach Executive Management Team Approves P|ESG strategy Ethics & Compliance Committee Defines P|ESG strategy and oversees implementation Corporate Responsibility & ESG Team Facilitates P|ESG strategy and oversight, guides and enables Business Process Owners Ascendis Pharma Business Process Owners Introduction P|ESG Approach Impact Ambitions 22
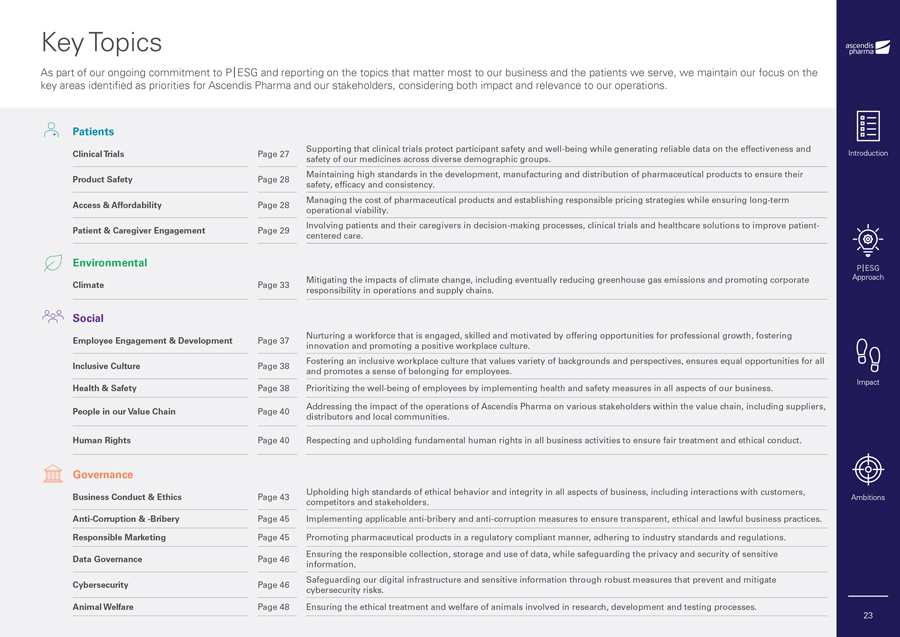
Key Topics As part of our ongoing commitment to P|ESG and reporting on the topics that matter most to our business and the patients we serve, we maintain our focus on the key areas identified as priorities for Ascendis Pharma and our stakeholders, considering both impact and relevance to our operations. Patients Supporting that clinical trials protect participant safety and well-being while generating reliable data on the effectiveness and Clinical Trials Page 27 safety of our medicines across diverse demographic groups. Maintaining high standards in the development, manufacturing and distribution of pharmaceutical products to ensure their Product Safety Page 28 safety, efficacy and consistency. Managing the cost of pharmaceutical products and establishing responsible pricing strategies while ensuring long-term Access & Affordability Page 28 operational viability. Involving patients and their caregivers in decision-making processes, clinical trials and healthcare solutions to improve patient- Patient & Caregiver Engagement Page 29 centered care. Environmental Mitigating the impacts of climate change, including eventually reducing greenhouse gas emissions and promoting corporate Climate Page 33 responsibility in operations and supply chains. Social Nurturing a workforce that is engaged, skilled and motivated by offering opportunities for professional growth, fostering Employee Engagement & Development Page 37 innovation and promoting a positive workplace culture. Fostering an inclusive workplace culture that values variety of backgrounds and perspectives, ensures equal opportunities for all Inclusive Culture Page 38 and promotes a sense of belonging for employees. Health & Safety Page 38 Prioritizing the well-being of employees by implementing health and safety measures in all aspects of our business. Addressing the impact of the operations of Ascendis Pharma on various stakeholders within the value chain, including suppliers, People in our Value Chain Page 40 distributors and local communities. Human Rights Page 40 Respecting and upholding fundamental human rights in all business activities to ensure fair treatment and ethical conduct. Governance Upholding high standards of ethical behavior and integrity in all aspects of business, including interactions with customers, Business Conduct & Ethics Page 43 competitors and stakeholders. Anti-Corruption & -Bribery Page 45 Implementing applicable anti-bribery and anti-corruption measures to ensure transparent, ethical and lawful business practices. Responsible Marketing Page 45 Promoting pharmaceutical products in a regulatory compliant manner, adhering to industry standards and regulations. Ensuring the responsible collection, storage and use of data, while safeguarding the privacy and security of sensitive Data Governance Page 46 information. Safeguarding our digital infrastructure and sensitive information through robust measures that prevent and mitigate Cybersecurity Page 46 cybersecurity risks. Animal Welfare Page 48 Ensuring the ethical treatment and welfare of animals involved in research, development and testing processes. Introduction P|ESG Approach Impact Ambitions 23
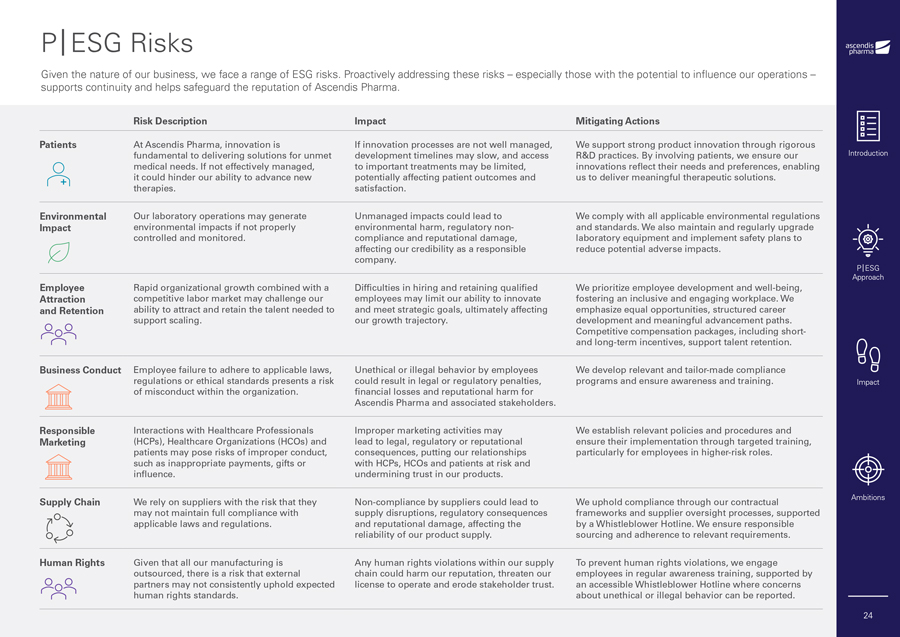
P|ESG Risks Given the nature of our business, we face a range of ESG risks. Proactively addressing these risks – especially those with the potential to influence our operations – supports continuity and helps safeguard the reputation of Ascendis Pharma. Risk Description Impact Mitigating Actions Patients At Ascendis Pharma, innovation is If innovation processes are not well managed, We support strong product innovation through rigorous fundamental to delivering solutions for unmet development timelines may slow, and access R&D practices. By involving patients, we ensure our medical needs. If not effectively managed, to important treatments may be limited, innovations reflect their needs and preferences, enabling it could hinder our ability to advance new potentially affecting patient outcomes and us to deliver meaningful therapeutic solutions. therapies. satisfaction. Environmental Our laboratory operations may generate Unmanaged impacts could lead to We comply with all applicable environmental regulations Impact environmental impacts if not properly environmental harm, regulatory non- and standards. We also maintain and regularly upgrade controlled and monitored. compliance and reputational damage, laboratory equipment and implement safety plans to affecting our credibility as a responsible reduce potential adverse impacts. company. Employee Rapid organizational growth combined with a Difficulties in hiring and retaining qualified We prioritize employee development and well-being, Attraction competitive labor market may challenge our employees may limit our ability to innovate fostering an inclusive and engaging workplace. We and Retention ability to attract and retain the talent needed to and meet strategic goals, ultimately affecting emphasize equal opportunities, structured career support scaling. our growth trajectory. development and meaningful advancement paths. Competitive compensation packages, including short- and long-term incentives, support talent retention. Business Conduct Employee failure to adhere to applicable laws, Unethical or illegal behavior by employees We develop relevant and tailor-made compliance regulations or ethical standards presents a risk could result in legal or regulatory penalties, programs and ensure awareness and training. of misconduct within the organization. financial losses and reputational harm for Ascendis Pharma and associated stakeholders. Responsible Interactions with Healthcare Professionals Improper marketing activities may We establish relevant policies and procedures and Marketing (HCPs), Healthcare Organizations (HCOs) and lead to legal, regulatory or reputational ensure their implementation through targeted training, patients may pose risks of improper conduct, consequences, putting our relationships particularly for employees in higher-risk roles. such as inappropriate payments, gifts or with HCPs, HCOs and patients at risk and influence. undermining trust in our products. Supply Chain We rely on suppliers with the risk that they Non-compliance by suppliers could lead to We uphold compliance through our contractual may not maintain full compliance with supply disruptions, regulatory consequences frameworks and supplier oversight processes, supported applicable laws and regulations. and reputational damage, affecting the by a Whistleblower Hotline. We ensure responsible reliability of our product supply. sourcing and adherence to relevant requirements. Human Rights Given that all our manufacturing is Any human rights violations within our supply To prevent human rights violations, we engage outsourced, there is a risk that external chain could harm our reputation, threaten our employees in regular awareness training, supported by partners may not consistently uphold expected license to operate and erode stakeholder trust. an accessible Whistleblower Hotline where concerns human rights standards. about unethical or illegal behavior can be reported. Introduction P|ESG Approach Impact Ambitions 24

Patients Patient Focus Clinical Trials • Clinical Trial Safety, Data Integrity and Transparency • Clinical Trial Representation Product Safety Access & Affordability • Named Patient Programs • Access Support in the U.S. Patient & Caregiver Engagement • Patient Support Programs – EU Direct Markets • Patient Registry Introduction P|ESG Approach Impact Ambitions 25

At Ascendis Pharma, patients are at the heart of all that we do. From the development of safe and effective medicines to their responsible commercialization, we strive to understand, support and advocate for the communities we serve. Listen No one knows the disease journey better than patients, families and caregivers. Understanding their needs and experiences provides critical insights that shape our work. Learn As a patient- and science-driven company, we actively seek to expand our knowledge through advisory boards, market research and community engagement. Advocate We collaborate to drive awareness and advance shared goals. By working together, we aspire to create lasting positive impact for those who rely on our therapies. Introduction P|ESG Approach Impact Ambitions 26

At the heart of our decision-making is a steadfast Ascendis Pharma aims to follow the principles We believe in clinical trial transparency as a commitment to addressing unmet medical needs of the Declaration of Helsinki and all relevant scientific, ethical and legal commitment and are and improving the lives of patients. We work ethical standards, laws and regulations when fully dedicated to disclosing clinical trial designs, to unlock the full potential of our products and conducting clinical trials with patients or results and outcomes through relevant channels, product opportunities, ensuring they deliver volunteers. This entails always obtaining approval including scientific journals, conferences and meaningful benefits. This patient-centric focus is for the trial from regulatory authorities and ethics public databases. not only integral to our mission and values, it is a committees. Our priority is to safeguard the guiding principle that informs every aspect of our rights, safety and well-being of all participants, Clinical Trial Representation work. It drives us to develop safe and effective including ensuring fully informed consent is We recognize the important role of patient medicines and to commercialize them responsibly collected from participants prior to trial start. To representation in clinical trials, including in so we can make a positive and lasting impact on the extent possible we are fully committed to the rare disease area, which in itself presents patients’ lives. providing our trial participants post-trial access to unique challenges and opportunities. Recruiting the medication until marketed either in long-term a meaningfully representative patient population The Patients chapter of this report reflects this open-labels cohorts or through local early access can be difficult due to the small numbers affected commitment and highlights our efforts across the programs. by rare diseases. However, our work in this following key areas: area has led us to expand recruitment efforts, Fully dedicated to allowing us to reach patients from a variety of • Clinical Trials disclosing clinical trial social backgrounds, ethnicities and nationalities • Product Safety designs to further strengthen the outcomes of our clinical • Access & Affordability trials. • Patient & Caregiver Engagement To protect the integrity of clinical data and Because rare disease patients are frequently We also provide insight into our broader business outcomes, we have established robust and seen at centralized treatment centers, we provide governance and policies. For further information, risk-based processes for collecting, processing, necessary and relevant support. we encourage readers to consult our Code of monitoring and analyzing information. In many Business Conduct & Ethics, which includes a instances, we collaborate with third-party We are also committed to exploring decentralized range of publicly available policies. vendors who act on our behalf. We hold them clinical trials to further enhance inclusivity. to the same high standards as we do ourselves. Decentralization strategies, like direct-to-patient Clinical Trials Before partnering with them, we carefully assess shipping of study medications and virtual their commitment to quality and compliance. site visits, allow patients to participate from a Clinical Trial Safety, Data Integrity and Afterward, we monitor that these standards are distance, reducing barriers tied to travel and Transparency met through established oversight and audit proximity to trial sites. However, these models As a science-based company, we are dedicated processes. This monitoring involves various introduce risks and compliance considerations to conducting trials with high quality and respect levels of the organization, including senior that we carefully assess to uphold patient safety for the participants and the scientific hypothesis management, through vendor governance and data integrity. being evaluated. models. Introduction P|ESG Approach Impact Ambitions 27

Lastly, we recognize that the broader challenge corrective and preventive actions to maintain Named Patient Programs of finding trial sites that serve a representative high levels of product safety. To reinforce our For some countries there is a significant gap range of populations is a global issue that commitment to safety, we provide regular between regulatory approval and product calls for collaborative action between industry pharmacovigilance training to employees and availability. In 2025, Ascendis Pharma continued and national regulatory agencies to improve relevant external partners. to support hypoparathyroid patients with inclusivity in clinical research. immediate treatment needs through Named Through these measures, we strive to safeguard Patient Programs, setting up logistical frameworks Product Safety patient well-being and ensure that healthcare to respond to physician requests wherever professionals have accurate and timely feasible. In these programs, it is the physicians At Ascendis Pharma, patient safety remains information regarding the safety profile of our who take the lead, coordinating with healthcare at the core of everything we do. We operate products. systems to ensure access. in compliance with all relevant health and safety regulations to ensure that our products We operate in alignment with the following Access support in the U.S. – Ascendis consistently uphold high standards of quality, quality and safety frameworks: Signature Access Program® (ASAP) safety and efficacy. In 2025, we advanced our commitment to • Good Clinical Practice (GCP) We maintain robust systems for monitoring patient access by fully executing the Yorvipath® • Good Laboratory Practice (GLP) and managing any adverse events connected patient support launch in the U.S. Building on • Good Manufacturing Practice (GMP) to our products and services. Our Global Safety the foundation laid in late 2024, we delivered • Good Distribution Practice (GDP) Database serves as a central platform for a comprehensive, high-touch support model • Good Pharmacovigilance Practice (GVP) collecting and processing adverse event data. that has already begun to make a meaningful • Requirements for the development of By analyzing aggregated reports and engaging difference for patients and healthcare providers combination products in signal detection activities, we proactively navigating this new therapy. identify emerging safety trends or potential risks. Additional details and related policies can be When a safety signal is observed, it is carefully As we look ahead, we remain focused on found in our Code of Business Conduct & Ethics. assessed, and appropriate measures are taken to further refining our offerings to meet the address them. These may include notifications evolving needs of patients prescribed Ascendis Access & Affordability to healthcare professionals, communication Pharma medications. This includes continued with regulatory authorities, or broader public At Ascendis Pharma, we are committed to enhancements to our services, technology and notifications as needed. advancing timely access to our innovative support model to ensure every patient receives medicines for patients. Where possible, we work timely, reliable and compassionate assistance Our safety measures include ongoing monitoring to help address access-related barriers that may throughout their treatment journey. of reported adverse events, supported by regular limit availability for patients and healthcare updates and safety guidance when required. We providers. conduct thorough investigations and implement Introduction P|ESG Approach Impact Ambitions 28

Patient & Caregiver Engagement reflects our belief that meaningful progress • Patient empowerment with knowledge and happens only when we work in true partnership. resources Over the past year, we strengthened our • Enhanced patient satisfaction commitment to improving the lives of individuals Our 2026 ambitions build on the foundation • Reduced healthcare expenses living with rare diseases by continuing to engage of championing ethical, evidence-driven • Enhanced health outcomes with the patient and caregiver communities scientific exchange, strengthening collaboration we serve. Their lived experiences remain with healthcare professionals and advancing Depending on local implementation, Ascendis central to how we design, deliver and refine insights that uplift rare disease communities. Pharma PSPs may include elements such as: innovative healthcare solutions. Through ongoing By remaining aligned with our core values collaboration with advocacy groups, advisory of trust, integrity and collaboration, we will • Education and training on device handling panels and community partners, we ensure that continue evolving our approach to make a and injection technique patient perspectives meaningfully inform our lasting, meaningful difference in the lives of the • Emotional and practical support services from scientific and strategic priorities. individuals and families we serve. trained personnel • Educational materials In 2025, we further advanced our efforts to build Patient Support Programs—EU Direct • Regular monitoring to improve therapeutic trusted, long-term relationships with advocacy Markets adherence and patient understanding organizations. By listening closely to their evolving needs and supporting initiatives that Driven by our commitment to ensuring patient Currently, Ascendis Pharma has established PSPs elevate community voices, we worked to amplify safety, Ascendis Pharma may offer Patient in Germany and Austria, supporting patients their impact across the healthcare ecosystem. Support Programs (PSP) aimed at improving across both hypoparathyroidism and growth These partnerships remain essential in addressing treatment adherence, enhancing clinical hormone deficiency. Across these markets, shared challenges and shaping patient-centered outcomes and empowering patients to effectively the programs support nearly 400 patients and initiatives that create real-world value. manage their disease. Studies consistently focus on education and guidance related to safe indicate that such programs have positive effects product use. Continue evolving our on patients’ adherence to medications, increasing satisfaction and improving their health-related In 2025, a PSP was also launched in approach to make a lasting, quality of life. Additionally, these programs have meaningful difference demonstrated improvements in clinical outcomes Spain to support patients with chronic and have reduced resource utilization leading to hypoparathyroidism, supporting safe and correct As we continue expanding our commercial cost savings. use of the pre-filled Yorvipath® pen by patients in the initial phase after treatment start and when presence in select global markets, we remain insecurities or problems arise. In other words, such programs may provide the focused on ensuring equitable access for following benefits: underserved communities. Our work is guided Through these patient support initiatives, by a commitment to ethical collaboration, Ascendis Pharma seeks to contribute to patient • Education and training transparent communication and unwavering safety, while respecting local regulations and • Improved treatment adherence respect for regulatory and industry standards. ensuring that all activities are implemented in a • Emotional, lifestyle and psychological support Every interaction with patient organizations compliant and responsible manner. Introduction P|ESG Approach Impact Ambitions 29
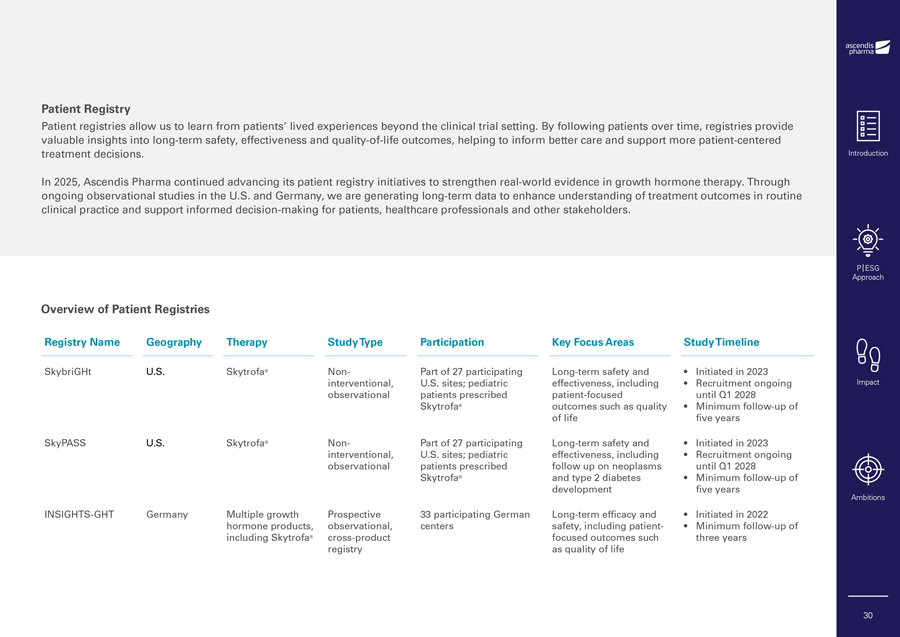
Patient Registry Patient registries allow us to learn from patients’ lived experiences beyond the clinical trial setting. By following patients over time, registries provide valuable insights into long-term safety, effectiveness and quality-of-life outcomes, helping to inform better care and support more patient-centered treatment decisions. In 2025, Ascendis Pharma continued advancing its patient registry initiatives to strengthen real-world evidence in growth hormone therapy. Through ongoing observational studies in the U.S. and Germany, we are generating long-term data to enhance understanding of treatment outcomes in routine clinical practice and support informed decision-making for patients, healthcare professionals and other stakeholders. Overview of Patient Registries Registry Name Geography Therapy Study Type Participation Key Focus Areas Study Timeline SkybriGHt U.S. Skytrofa® Non- Part of 27 participating Long-term safety and • Initiated in 2023 interventional, U.S. sites; pediatric effectiveness, including • Recruitment ongoing observational patients prescribed patient-focused until Q1 2028 Skytrofa® outcomes such as quality • Minimum follow-up of of life five years SkyPASS U.S. Skytrofa® Non- Part of 27 participating Long-term safety and • Initiated in 2023 interventional, U.S. sites; pediatric effectiveness, including • Recruitment ongoing observational patients prescribed follow up on neoplasms until Q1 2028 Skytrofa® and type 2 diabetes • Minimum follow-up of development five years INSIGHTS-GHT Germany Multiple growth Prospective 33 participating German Long-term efficacy and • Initiated in 2022 hormone products, observational, centers safety, including patient- • Minimum follow-up of including Skytrofa® cross-product focused outcomes such three years registry as quality of life Introduction P|ESG Approach Impact Ambitions 30

2025 and 2026 Corporate Responsibility and P|ESG Ambitions 2025 Ambitions Status Description 2026 Ambitions Significant regulatory developments, including Review the material impacts, risks and the evolving CSRD and ESRS requirements, led In opportunities related to our patients as us to pause our DMA work to ensure alignment progress part of our 2025 DMA. with updated regulatory expectations and the Ascendis Pharma strategy. Continue to refine our approach Monitor the regulatory landscape Due to shifting regulatory expectations around to identifying and prioritizing and seek input from industry peers population diversity in clinical trials, industry patient-related impacts, risks and patient advocacy groups on the In activity in this area has slowed. We are and opportunities that support developing field of population diversity progress currently maintaining a monitoring position, our patient-centric strategy. in clinical trials with specific focus on tracking evolving regulatory frameworks and rare diseases, including oncology. gathering informal input from peers. Continued enhancements to our services, technology and We have been able to create pathways to Establish access pathways to our entire support model to ensure every In access in over 75 countries across international endocrinology portfolio across the world patient receives timely, reliable progress markets through strategic partnerships. To date to all patients in need. and compassionate assistance we are treating patients in 25 countries. throughout their treatment journey. Ascendis Pharma is treating patients globally Add innovative technology based on with TransCon® based products, reflecting TransCon® to the HCPs’ armamentarium, In Amplify awareness of unmet continued progress toward establishing establishing it as the gold standard of progress medical needs in growth these therapies as a standard of care for rare care for treating rare endocrine diseases. disorders through publication endocrine diseases. of scientific data, educational events to reinforce scientific Hosted “Innovation through Science” event credibility and sustained for endocrinologists and other specialists, engagement with patients and Champion ethical, meaningful and fostering dialogue around clinical evidence HCPs. evidence-driven scientific exchange with generated by our data-driven approach, long-HCPs, anchored in our deep knowledge Achieved term patient benefit with focus on clearly of medical science, to improve outcomes defined unmet medical needs, and responsible in individuals with rare diseases. innovation through the application of our TransCon® technology to design product candidates around real patient priorities. Introduction P|ESG Approach Impact Ambitions 31

Environmental Climate Impact Suppliers & Business Partners Introduction P|ESG Approach Impact Ambitions 32

At Ascendis Pharma, we understand the importance of managing our environmental impact across the full value chain. Although we do not own manufacturing sites, we work closely with Contract Development and Manufacturing Organizations (CDMOs), logistics providers, research partners and other third-party collaborators to uphold environmental principles and comply with applicable laws, industry standards and internal guidelines. As we continue to assess our environmental footprint, we are focusing on strengthening our understanding of our impacts so we can identify opportunities for future improvement. As our business grows, we remain committed to exploring opportunities to deepen environmental responsibility across our operations and value chain while maintaining compliance with regulatory expectations. This year’s Environmental chapter provides insights into: • Climate Impact • Suppliers & Business Partners Further details can be found in our Environmental Policy in our Code of Business Conduct & Ethics. Climate Impact Our attention to climate impact spans our offices, laboratories and the broader network of partners that support our work. All our facilities operate in leased spaces, and we continue to integrate sustainability considerations into their management. Over the past year, we collected Scope 1 and 2 emissions data, allowing us to better assess our direct and indirect climate impact. In 2025, total energy consumption increased from 4,880 MWh to 6,397 MWh, alongside an increase in total Scope 1 and 2 emissions from 925 tCO e to 1,124 tCO e. This 2 2 year-on-year increase primarily reflects business growth during the reporting period, including the addition of new office locations and an expanded operational footprint. The resulting increase in absolute emissions is therefore driven by growth in activities rather than changes in operational efficiency. Continue to prioritize improved data robustness Although we do not own or operate manufacturing sites, we remain diligent in understanding the environmental footprint associated with the production and commercialization of our products. This year, we continued collecting greenhouse gas emissions data from our largest CDMOs and logistics providers to strengthen our understanding of the upstream and downstream value chain of our commercially approved products. This ongoing Introduction P|ESG Approach Impact Ambitions 33
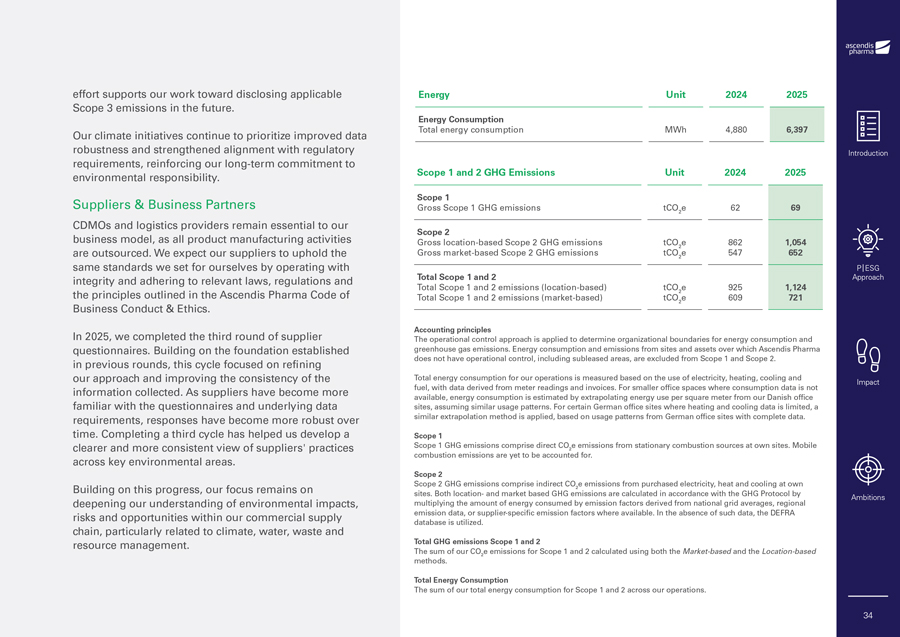
effort supports our work toward disclosing applicable Scope 3 emissions in the future. Our climate initiatives continue to prioritize improved data robustness and strengthened alignment with regulatory requirements, reinforcing our long-term commitment to environmental responsibility. Suppliers & Business Partners CDMOs and logistics providers remain essential to our business model, as all product manufacturing activities are outsourced. We expect our suppliers to uphold the same standards we set for ourselves by operating with integrity and adhering to relevant laws, regulations and the principles outlined in the Ascendis Pharma Code of Business Conduct & Ethics. In 2025, we completed the third round of supplier questionnaires. Building on the foundation established in previous rounds, this cycle focused on refining our approach and improving the consistency of the information collected. As suppliers have become more familiar with the questionnaires and underlying data requirements, responses have become more robust over time. Completing a third cycle has helped us develop a clearer and more consistent view of suppliers’ practices across key environmental areas. Building on this progress, our focus remains on deepening our understanding of environmental impacts, risks and opportunities within our commercial supply chain, particularly related to climate, water, waste and resource management. Energy Unit 2024 2025 Energy Consumption Total energy consumption MWh 4,880 6,397 Scope 1 and 2 GHG Emissions Unit 2024 2025 Scope 1 Gross Scope 1 GHG emissions tCO e 62 69 2 Scope 2 Gross location-based Scope 2 GHG emissions tCO e 862 1,054 2 Gross market-based Scope 2 GHG emissions tCO e 547 652 2 Total Scope 1 and 2 Total Scope 1 and 2 emissions (location-based) tCO e 925 1,124 2 Total Scope 1 and 2 emissions (market-based) tCO e 609 721 2 Accounting principles The operational control approach is applied to determine organizational boundaries for energy consumption and greenhouse gas emissions. Energy consumption and emissions from sites and assets over which Ascendis Pharma does not have operational control, including subleased areas, are excluded from Scope 1 and Scope 2. Total energy consumption for our operations is measured based on the use of electricity, heating, cooling and fuel, with data derived from meter readings and invoices. For smaller office spaces where consumption data is not available, energy consumption is estimated by extrapolating energy use per square meter from our Danish office sites, assuming similar usage patterns. For certain German office sites where heating and cooling data is limited, a similar extrapolation method is applied, based on usage patterns from German office sites with complete data. Scope 1 Scope 1 GHG emissions comprise direct CO e emissions from stationary combustion sources at own sites. Mobile 2 combustion emissions are yet to be accounted for. Scope 2 Scope 2 GHG emissions comprise indirect CO e emissions from purchased electricity, heat and cooling at own 2 sites. Both location- and market based GHG emissions are calculated in accordance with the GHG Protocol by multiplying the amount of energy consumed by emission factors derived from national grid averages, regional emission data, or supplier-specific emission factors where available. In the absence of such data, the DEFRA database is utilized. Total GHG emissions Scope 1 and 2 The sum of our CO e emissions for Scope 1 and 2 calculated using both the Market-based and the Location-based 2 methods. Total Energy Consumption The sum of our total energy consumption for Scope 1 and 2 across our operations. Introduction P|ESG Approach Impact Ambitions 34

2025 and 2026 Corporate Responsibility and P|ESG Ambitions 2025 Ambitions Status Description 2026 Ambitions Continue and further standardize Scope 1 and Scope Significant regulatory developments, 2 greenhouse gas emissions Review the material environmental including the evolving CSRD and ESRS reporting. impacts, risks and opportunities within our In requirements, led us to pause our DMA operations and commercial supply chains progress work to ensure alignment with updated Further refine and improve as part of our 2025 DMA. regulatory expectations and the Ascendis Scope 3 data collection through Pharma strategy. engagement with key suppliers and partners. Introduction P|ESG Approach Impact Ambitions 35

Social Employee Engagement & Development Inclusive Culture Health & Safety People in our Value Chain Human Rights Introduction P|ESG Approach Impact Ambitions 36
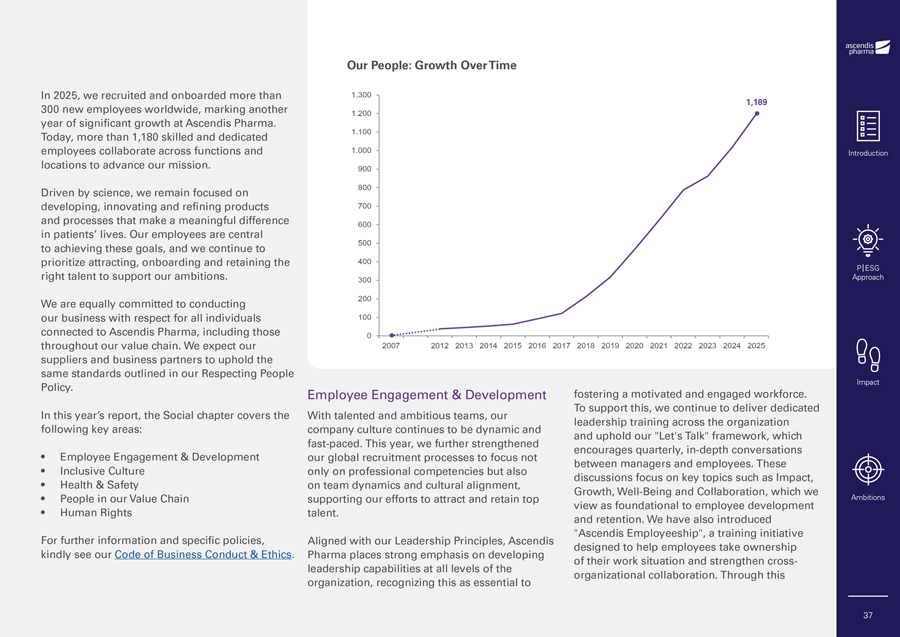
In 2025, we recruited and onboarded more than 300 new employees worldwide, marking another year of significant growth at Ascendis Pharma. Today, more than 1,180 skilled and dedicated employees collaborate across functions and locations to advance our mission. Driven by science, we remain focused on developing, innovating and refining products and processes that make a meaningful difference in patients’ lives. Our employees are central to achieving these goals, and we continue to prioritize attracting, onboarding and retaining the right talent to support our ambitions. We are equally committed to conducting our business with respect for all individuals connected to Ascendis Pharma, including those throughout our value chain. We expect our suppliers and business partners to uphold the same standards outlined in our Respecting People Policy. In this year’s report, the Social chapter covers the following key areas: • Employee Engagement & Development • Inclusive Culture • Health & Safety • People in our Value Chain • Human Rights For further information and specific policies, kindly see our Code of Business Conduct & Ethics. Our People: Growth Over Time 1.300 1,189 1.200 1.100 1.000 900 800 700 600 500 400 300 200 100 0 2007 2012 2013 2014 2015 2016 2017 2018 2019 2020 2021 2022 2023 2024 2025 Employee Engagement & Development With talented and ambitious teams, our company culture continues to be dynamic and fast-paced. This year, we further strengthened our global recruitment processes to focus not only on professional competencies but also on team dynamics and cultural alignment, supporting our efforts to attract and retain top talent. Aligned with our Leadership Principles, Ascendis Pharma places strong emphasis on developing leadership capabilities at all levels of the organization, recognizing this as essential to fostering a motivated and engaged workforce. To support this, we continue to deliver dedicated leadership training across the organization and uphold our “Let’s Talk” framework, which encourages quarterly, in-depth conversations between managers and employees. These discussions focus on key topics such as Impact, Growth, Well-Being and Collaboration, which we view as foundational to employee development and retention. We have also introduced “Ascendis Employeeship”, a training initiative designed to help employees take ownership of their work situation and strengthen cross-organizational collaboration. Through this Introduction P|ESG Approach Impact Ambitions 37

initiative, we aim to create opportunities for personal development that ultimately contribute to the collective success of Ascendis Pharma. Employee feedback remains an important part of how we evolve our culture and practices, and we invite employees to share their perspectives through various surveys, including onboarding and offboarding surveys and interviews. Employee turnover is monitored as a key indicator of workforce engagement and organizational health. Insights from onboarding and offboarding processes, together with employee feedback, help inform our efforts to strengthen retention and continuously improve the employee experience. In 2025, employee turnover decreased to 11.3% from 14.0% in 2024, reflecting improved retention and organizational stability. We believe that attracting and maintaining an engaged workforce depends on being an equitable and fair employer. As part of this commitment, we continue to emphasize pay equity across the organization by assessing and addressing any potential pay disparities. Our initial analysis has been encouraging, showing little to no systemic variation between female and male employees. This work is ongoing, and we will continue our efforts in the coming year to strengthen objective and gender-neutral pay practices, transparency, and internal monitoring in line with evolving regulatory requirements. Finally, at the discretion of our Board of Directors and upon management’s recommendation, employees are eligible to participate in our short-term and long-term incentive programs, which support performance, engagement and motivation across Ascendis Pharma. Inclusive Culture We are committed to fostering an inclusive workplace where every individual can thrive and contribute fully. This ambition is reflected in our culture, policies and everyday practices. Ascendis Pharma is dedicated to ensuring equal opportunities and fair treatment for all individuals based solely on merit, free from any discrimination based on race, color, religion, national origin, gender identity or expression, including pregnancy, sexual orientation, age, disability, veteran status or any other characteristic protected by law. Dedicated to ensuring equal opportunities Our focus on inclusion is embedded throughout our people practices, including recruiting, people development, leadership development and succession planning in compliance with applicable guidelines. In line with this commitment, we maintain a policy for our Board of Directors that emphasizes selecting the most qualified candidates to support our business. Gender diversity across leadership positions at Ascendis Pharma meets Danish gender diversity requirements. Consistent with guidelines from the Danish Business Authority, we define equal representation as an acceptable gender distribution within a 40/60 range. We monitor this distribution continuously and conduct a formal biannual evaluation to identify and implement initiatives as needed. As a result, we currently achieve equal gender representation not only on our Board of Directors but also across all management levels within the organization. Aligned with our ambition to remain an attractive and fair employer, our reward philosophy is built on offering competitive and equitable compensation based on gender-neutral principles. Compensation decisions are informed by role evaluations, individual qualifications, experience and performance, ensuring fair and consistent recognition of our employees. Health & Safety At Ascendis Pharma, we are committed to conducting our business in a way that safeguards the health, safety and well-being of our employees, in full compliance with applicable health and safety laws and regulations. Health and safety considerations are integrated into our daily operations, and we regularly incorporate feedback from employees and external Introduction P|ESG Approach Impact Ambitions 38
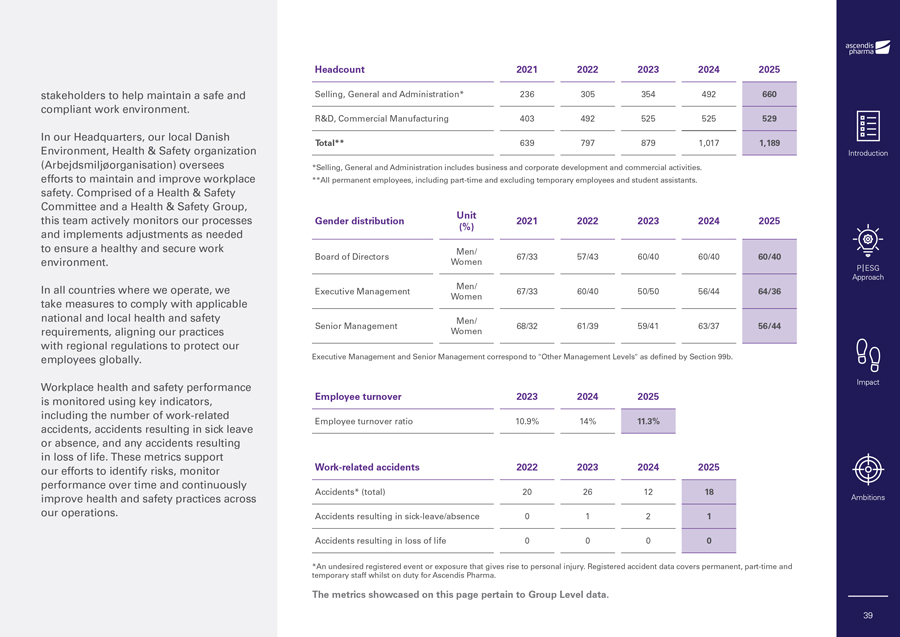
stakeholders to help maintain a safe and compliant work environment. In our Headquarters, our local Danish Environment, Health & Safety organization (Arbejdsmiljøorganisation) oversees efforts to maintain and improve workplace safety. Comprised of a Health & Safety Committee and a Health & Safety Group, this team actively monitors our processes and implements adjustments as needed to ensure a healthy and secure work environment. In all countries where we operate, we take measures to comply with applicable national and local health and safety requirements, aligning our practices with regional regulations to protect our employees globally. Workplace health and safety performance is monitored using key indicators, including the number of work-related accidents, accidents resulting in sick leave or absence, and any accidents resulting in loss of life. These metrics support our efforts to identify risks, monitor performance over time and continuously improve health and safety practices across our operations. Headcount 2021 2022 2023 2024 2025 Selling, General and Administration* 236 305 354 492 660 R&D, Commercial Manufacturing 403 492 525 525 529 Total** 639 797 879 1,017 1,189 *Selling, General and Administration includes business and corporate development and commercial activities. **All permanent employees, including part-time and excluding temporary employees and student assistants. Unit Gender distribution 2021 2022 2023 2024 2025 (%) Men/ Board of Directors 67/33 57/43 60/40 60/40 60/40 Women Men/ Executive Management 67/33 60/40 50/50 56/44 64/36 Women Men/ Senior Management 68/32 61/39 59/41 63/37 56/44 Women Executive Management and Senior Management correspond to “Other Management Levels” as defined by Section 99b. Employee turnover 2023 2024 2025 Employee turnover ratio 10.9% 14% 11.3% Work-related accidents 2022 2023 2024 2025 Accidents* (total) 20 26 12 18 Accidents resulting in sick-leave/absence 0 1 2 1 Accidents resulting in loss of life 0 0 0 0 *An undesired registered event or exposure that gives rise to personal injury. Registered accident data covers permanent, part-time and temporary staff whilst on duty for Ascendis Pharma. The metrics showcased on this page pertain to Group Level data. Introduction P|ESG Approach Impact Ambitions 39

People in our Value Chain As outlined in the Environmental section, our business relies on a network of partners and suppliers who are responsible for manufacturing our products. We expect these partners and suppliers to uphold strong practices and standards of integrity, including compliance with applicable laws, regulations and social responsibility principles. We remain committed to strengthening our understanding of the social impacts, risks and opportunities within our commercial supply chain. This approach ensures that our commitment to ethical and socially responsible practices extends across our broader value chain. Our efforts focus on identifying areas where we can make a meaningful difference, including promoting fair labor practices, upholding human rights and ensuring workplace safety across our supply chain. By fostering strong and transparent relationships with our partners and suppliers, we strive to uphold our values and contribute to a more responsible and sustainable value chain. Human Rights Our commitment to human rights is grounded in internationally recognized standards, including the Universal Declaration of Human Rights (UNDHR), the International Covenant on Civil and Political Rights (ICCPR) and its Second Optional Protocol, and the International Covenant on Economic, Social and Cultural Rights (ICESCR). We also align with the fundamental conventions of the International Labor Organization (ILO), which play a central role in safeguarding workers’ rights and form an integral part of our human rights approach. Guided by the UN Guiding Principles, we incorporate human rights considerations into our third-party compliance approach. Our goal is to ensure that our partners and suppliers uphold relevant human rights standards and principles throughout their operations. Our Respecting People Policy, including our comprehensive Human Rights Policy, is embedded in our Code of Business Conduct & Ethics and provides employees and business partners with clear expectations regarding human rights and ethical conduct. The Code outlines mechanisms for raising concerns, including access to a Whistleblower Hotline, which enables confidential reporting of potential unethical or illegal behavior. The availability of this mechanism has established a formal channel for identifying and addressing concerns, and reports received are reviewed and handled in accordance with established internal procedures. Through these measures, we aim to prevent human rights violations, strengthen transparency, and protect our reputation, license to operate, and stakeholder trust. Introduction P|ESG Approach Impact Ambitions 40

2025 and 2026 Corporate Responsibility and P|ESG Ambitions 2025 Ambitions Status Description 2026 Ambitions Ascendis Pharma continues to grow with a Attraction, onboarding and Attraction, onboarding and retention of talent continued focus to attract and retain the right retention of talent to ensure we to ensure we have the right people to deliver on Ongoing talent to deliver on the Ascendis Pharma have the right people to deliver on our ambitions. ambitions. our ambitions. Continuous focus on leadership and employeeship development to enable the development, Continuous focus on leadership and performance and well-being of our employeeship development to enable the Throughout the year, training in all areas has people. Ongoing development, performance and well-being of our been carried out across locations. people. Maintain a strong focus on advancing gender pay initiatives and meeting evolving gender pay reporting requirements. We have further developed our gender neutral Investigation of our pay policies, emphasizing pay structures and carried out ongoing Continue to develop our human pay equity across the organization and taking Ongoing monitoring of gender pay differentials. We are rights approach by strengthening actionable steps towards ensuring a clear, furthermore preparing for upcoming gender pay third-party compliance practices objective and transparent pay approach. reporting requirements under EU legislation. and maintaining accessible mechanisms for raising concerns. Introduction P|ESG Approach Impact Ambitions 41

Governance Business Conduct & Ethics • The Ascendis Pharma Business Ethics & Compliance Program • Effective and Tailored Compliance Training Anti-Corruption & -Bribery Responsible Marketing • Transparent Interactions • Promotional Review & Compliance Data Governance • Data Ethics • Data Privacy Cybersecurity Third-Party Compliance Speak Up Policy & Whistleblower Hotline Animal Welfare Introduction P|ESG Approach Impact Ambitions 42

At Ascendis Pharma, we remain dedicated to conducting our business responsibly, upholding high ethical standards and complying with applicable laws and regulations. Compliant behavior is fundamental to our Corporate Responsibility & P|ESG Reporting framework and central to how we operate as a biopharmaceutical company. Acting with integrity is essential to maintaining our license to operate and is a key driver of our continued success. This year’s Governance chapter reaffirms our commitment to responsible business practices and covers the following areas: • Business Conduct & Ethics • Anti-Bribery & -Corruption • Responsible Marketing, including transparent transactions • Data Governance, including data ethics and data privacy • Cybersecurity • Animal welfare Business Conduct & Ethics The Ascendis Pharma Code of Business Conduct & Ethics provides the foundation for our actions and establishes a set of global policies within many areas across our business. It translates our company values into practical guidance and establishes clear expectations for compliant behavior across our value chain. All employees are required to follow the Code of Business Conduct & Ethics and its supporting Content Overview of the Ascendis Pharma Code of Business Conduct & Ethics policies and procedures at all times. Any compliance may lead to contract termination or identified or potential breaches are carefully legal action, where applicable. assessed and investigated, with corrective actions taken when necessary, and may include As part of our life-cycle management program, additional training, formal warnings or, in certain the Code of Business Conduct & Ethics will be circumstances, termination of employment. reviewed, updated as necessary, and approved by the Board of Directors in 2026. We also expect our business partners to act in accordance with the principles outlined in the Further details on our governance structure, Code of Business Conduct & Ethics. Any identified policies and procedures can be found in our or potential violations are investigated, and non- Code of Business Conduct & Ethics which is available on our website. Introduction P|ESG Approach Impact Ambitions 43
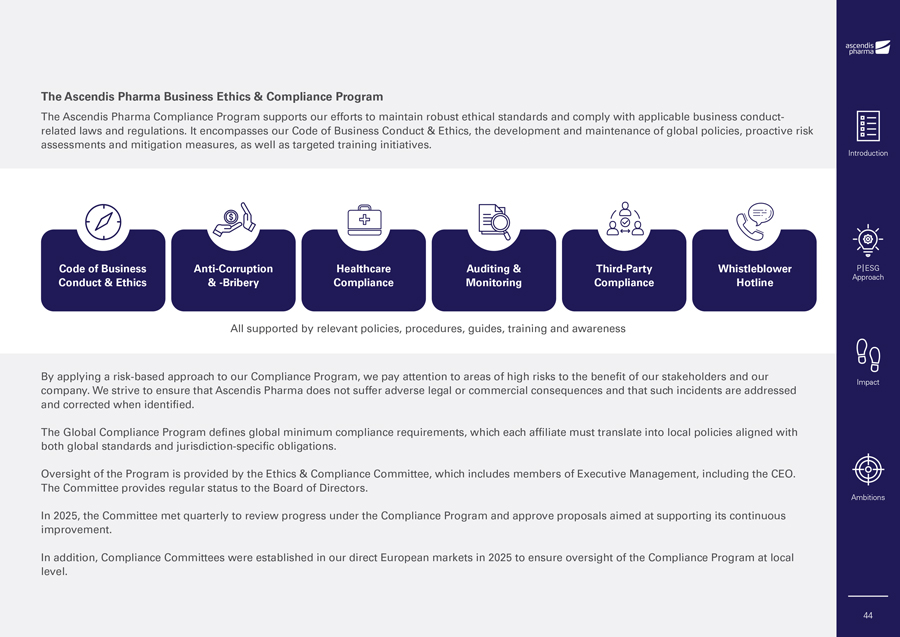
The Ascendis Pharma Business Ethics & Compliance Program The Ascendis Pharma Compliance Program supports our efforts to maintain robust ethical standards and comply with applicable business conduct-related laws and regulations. It encompasses our Code of Business Conduct & Ethics, the development and maintenance of global policies, proactive risk assessments and mitigation measures, as well as targeted training initiatives. Introduction Code of Business Anti-Corruption Healthcare Auditing & Third-Party Whistleblower P|ESG Conduct & Ethics & -Bribery Compliance Monitoring Compliance Hotline Approach All supported by relevant policies, procedures, guides, training and awareness By applying a risk-based approach to our Compliance Program, we pay attention to areas of high risks to the benefit of our stakeholders and our Impact company. We strive to ensure that Ascendis Pharma does not suffer adverse legal or commercial consequences and that such incidents are addressed and corrected when identified. The Global Compliance Program defines global minimum compliance requirements, which each affiliate must translate into local policies aligned with both global standards and jurisdiction-specific obligations. Oversight of the Program is provided by the Ethics & Compliance Committee, which includes members of Executive Management, including the CEO. The Committee provides regular status to the Board of Directors. Ambitions In 2025, the Committee met quarterly to review progress under the Compliance Program and approve proposals aimed at supporting its continuous improvement. In addition, Compliance Committees were established in our direct European markets in 2025 to ensure oversight of the Compliance Program at local level. 44

Effective and Tailored Compliance Training Anti-Corruption & -Bribery Policy All interactions with these stakeholders must have a legitimate purpose and fully comply with Training plays a critical role in maintaining a The Anti-Corruption & -Bribery Policy strictly the applicable regulations governing the Ascendis strong compliance culture. Employees undergo a prohibits bribery, corruption and kickbacks in any form, whether involving public officials or Pharma entity, the participants involved and the range of mandatory training programs tailored to private-sector individuals, and whether carried location of the interaction. Introduction their roles and associated risk areas. out directly or indirectly through a third party. We value transparency in these interactions In 2025, mandatory training in the Code of Under this policy, employees are not allowed to offer, promise or authorize anything of value and disclose transfers of value to healthcare Business Conduct & Ethics was provided to new to influence the actions of government officials professionals, healthcare organizations, and employees as part of the onboarding process or healthcare professionals, seek preferential patient organizations in accordance with legal and through an e-learning module followed by an treatment or express gratitude for favorable regulatory requirements. assessment requiring a 100% passing score. actions. Similarly, employees cannot solicit The overall completion rate reached 99%, with or accept any form of payment or valuable To ensure compliance with applicable regulations the remaining 1% accounted for by employees item intended to sway their responsibilities P|ESG we have built a compliance framework to support Approach on leave or other extenuating circumstances. or express gratitude for acting in a way that such interactions. Existing employees complete this training on a improperly benefited that person. biennial basis, with the next scheduled refresher The policy also prohibits the use of third parties Promotional Review & Compliance planned for 2026. to facilitate payments or actions that would Responsible marketing is essential to how we In addition, face-to-face training workshops were otherwise be impermissible if performed operate. Information about products must meet directly by Ascendis Pharma. legal and regulatory requirements and maintain conducted in selected Direct Markets providing a forum to discuss practical compliance dilemmas compliant and ethical practices. Impact and everyday challenges. Building on this experience, in-person training on high-risk areas Our policy underscores our commitment Therefore, we are committed to ensuring that will remain a focus area in 2026. to transparency, integrity and adherence to our external communication and messaging ethical standards and legal requirements in all are consistent in our business operations, and Anti-Corruption & -Bribery interactions. For more details, please refer to the that our educational efforts benefit patients Anti-Corruption & -Bribery Policy in our Code of and caregivers, and that we do not have, or As part of the Ascendis Pharma Compliance Business Conduct & Ethics. appear to have, an undue and illegal influence Program, all employees undergo biennial training on medical judgment, prescribing or product in the Anti-Corruption & Bribery Policy, which Responsible Marketing Ambitions recommendations. outlines the expected behavior as representatives of the company. However, employees with Transparent Interactions To that end, we are guided by our values, our potential exposure, such as field-based staff, Collaborating with healthcare professionals, high ethical standards and our commitment to receive additional role-specific training to ensure healthcare organizations and patients is comply with applicable legal, regulatory and they understand and can navigate relevant risks. fundamental to our mission to develop innovative professional requirements in the countries technologies and treatments. in which we operate. Our relationship with 45

others who administer, prescribe, purchase or Our focus continues to be on strengthening data risk management program designed to recommend regulated healthcare products, must ethics so that employees and partners clearly safeguard the confidentiality and integrity of the meet similar high standards of integrity and must understand our standards when collecting, information we collect and process, maintain comply with applicable laws, regulations and processing and managing data with an emphasis resilient Information Technology (IT) operations industry codes. on quality, integrity, transparency and security in and respond effectively to potential threats. every aspect of data handling. Introduction We therefore maintain formal review processes Our program is guided by internationally for medical and promotional materials before use. Data Privacy recognized frameworks. On a strategic and In 2024, we updated these processes to reflect tactical level, we have designed our security As data privacy laws and regulations continue to our evolving global footprint and the growing program around the International Organization evolve globally, our commitment to safeguarding reach of our products. In 2025, we implemented for Standardization/International Electrotechnical personal data remains robust as we keep on applicable processes in relevant Direct Markets to Commission (ISO/IEC) 27001 standard, while our strengthening our privacy practices. ensure compliance with local requirements. operational program is maintained in accordance with the Center for Internet Security (CIS) Critical Our policies and procedures set clear P|ESG Data Governance expectations for both employees and business Security Controls framework. Approach Data Ethics partners to uphold our high standards when Our Information Security Policy for Global IT handling personal data. By having a clear As a forward-thinking biopharma company outlines the organizational responsibilities for framework for collection, storage, use, transfer guided by our core principles of patients, maintaining a strong security posture for our IT and disposal of personal data, we continue to science and passion, data lies at the heart of our systems and sets forth the IT security measures promote security, accuracy and transparency mission to deliver meaningful improvements and controls that are required to be in place. throughout the personal data lifecycle. in patients’ lives. We are therefore dedicated to This policy covers all IT systems that process managing data responsibly while maintaining full Impact Ascendis Pharma information. Our Security Our commitment also extends to third-party compliance with applicable laws, regulations and Management team is responsible for ensuring data processing where we continue to conduct guidelines. internal security compliance and for managing IT thorough evaluations to confirm our partners’ vendors. Continue to embed commitment to protecting personal data. To enforce these standards, we enter into data responsible AI principles Our cybersecurity risk management program processing and, where applicable, data transfer includes the following core elements: agreements, complemented by ongoing With the rapid progress of artificial intelligence, monitoring to ensure compliance. • Risk assessments designed to identify Ambitions we continue to embed responsible AI principles into our data governance approach. Our work Cybersecurity cybersecurity risks across our critical includes building an AI framework designed systems, data, and broader IT environment. Safeguarding the confidentiality, integrity and • A dedicated Security Management to ensure transparency, ethical practices and availability of our information and systems is team responsible for managing our risk safe deployment of AI systems, reinforcing our essential to our ability to operate responsibly. We assessment processes, security controls, and commitment to data ethics. have established a comprehensive cybersecurity incident response activities. 46

Whistleblower Hotline Policy • An external and internal Security Operations Speak Up Policy & Whistleblower Hotline Our Whistleblower Hotline provides a secure Center (SOC) performing advanced and trusted channel for raising concerns. We encourage and expect employees to raise Employees as well as external parties, including threat detection and response across our concerns about unethical behavior or potential or former or prospective employees, members of environment. actual breaches of the Code of Business Conduct the Board of Directors, consultants, and other Introduction • A defined process for registration, & Ethics or related policies. third parties, can submit reports safely and, classification and escalation of any incidents, if they choose, anonymously. The platform managed by a designated IT Incident Manager While direct managers are generally the first point also enables direct communication with the and cross-functional incident response team. of contact, we recognize that reporting through investigation team throughout the process. The • Employee awareness initiatives, deployed a line manager may not always be comfortable. hotline is available at all times, 24/7. Concerns across various internal channels to reinforce Employees can therefore also contact HR or can be submitted either through a secure online strong security behaviors. Compliance confidentially. Additionally, the form or by phone via the protected hotline Whistleblower Hotline provides an anonymous service. • Secure access control measures applied to critical IT systems, devices, and equipment to channel for employees and external parties to P|ESG Whistleblower Hotline Reports Approach prevent unauthorized access. report concerns. To promote accountability and transparency, 2025 2 Oversight of our program begins at the Board of Directors, with more detailed supervision we disclose key insights from the Speak Up 2024 0 delegated to the Executive Board. The Executive Policy and Whistleblower Hotline over the past 2023 0 Board receives regular updates from our five years, emphasizing our commitment to Security Management team and is briefed on maintaining an environment where concerns can 2022 1 any significant cybersecurity incidents; it in turn be raised without fear of negative consequences. 2021 2 Impact reports key developments to the full Board. This In 2025, two reports were received through governance structure ensures alignment between the Whistleblower Hotline. Both reports were our cybersecurity program, our operational investigated and concluded in accordance with our procedures, and appropriate actions were Protection of good faith reporters priorities, and our long-term business strategy. taken where relevant. Ascendis Pharma strictly prohibits any form Third-Party Compliance of retaliation against individuals who, in good Our Whistleblower Hotline can be accessed faith, seek guidance or report concerns or Our commitment to ethical conduct extends to 24/7 through our website and can be used by potential violations. Any retaliatory behavior will Ambitions our external partners. We expect third parties employees and external parties to report potential lead to disciplinary action, which may include acting on our behalf to adhere to the same violations of our Code of Business Conduct & termination of employment. We are committed standards that we apply internally. To this end, Ethics, applicable laws, or internal policies. to maintaining confidentiality in accordance with we ensure that relevant contracts with partners applicable laws and safeguarding the privacy of include clauses that stipulate this requirement those who report concerns. However, knowingly and reserves the right for us to audit sales and submitting false reports is considered a serious distribution partners to ensure alignment with our misconduct and may result in appropriate ethical and legal expectations. disciplinary measures. 47

Animal Welfare our high standards for the housing and care of experimental animals. To address animal welfare Animal studies are required by regulatory at suppliers of animal-derived biological matrices authorities and play an important role in the for Ascendis Pharma studies, an audit program development of new pharmaceuticals. The has been initiated to evaluate suppliers by means Introduction Ascendis Pharma Internal Committee for Animal of questionnaires, on site or virtual audits. Those Welfare reinforces our continued commitment vendors that comply with our high standards of to apply the best possible welfare for animals animal welfare have been added to an approved used in the development of safe and effective vendor list and are utilized for the purchase of human treatments. Our principles adhere to animal derived material internally. Efforts are the 3R framework: Replacement, Reduction and underway in establishing a similar approach for Refinement, and we emphasize the conduct of external bioanalytical laboratories contracted by only necessary and scientifically sound animal Ascendis Pharma. studies, minimizing the use of animals in our P|ESG Approach research whenever possible. The Committee is further dedicated to animal welfare on a Emphasize the conduct global scale by the signing of the “Marseille of only necessary and Declaration on the worldwide implementation scientifically sound animal of high standards for animals housed and used studies internally and externally by the industry for scientific purposes” in 2024 and the continuous collaboration with the other signatories in With the aim of increasing company-wide Impact matters concerning animal welfare. awareness of the work of the Committee and the 3R initiatives implemented in 2025, the Internal The Internal Committee for Animal Welfare Committee for Animal Welfare undertook the commits to the 3R principles when providing in- following activities: house guidance on all matters related to animal studies. All animal study protocols are reviewed • Shared information on animal welfare by the Committee to ensure that only necessary initiatives via internal communication and scientifically sound animal studies are platforms. Ambitions conducted and that the studies comply with a high animal welfare standard. All animal studies • Launched animal welfare training material as conducted by Ascendis Pharma are performed part of general employee onboarding. at external Contract Research Organizations • Hosted an internal knowledge-sharing session (CROs). These CROs are audited regularly by focused on animal welfare initiatives within members of the Committee for compliance with the company. 48

2025 and 2026 Corporate Responsibility and P|ESG Ambitions 2025 Ambitions Status Description 2026 Ambitions Continuous work with the establishment as well Further strengthen our Global Compliance Continue building and Ongoing as updating of global compliance framework Framework. strengthening our Compliance concurrently with our business development. Framework in accordance with Further strengthen our Compliance Frameworks Identified all relevant compliance elements, compliance areas concurrently in our EU Direct Markets to prepare for future Ongoing in the process of implementation, established with Ascendis Pharma growth. launches. Compliance Committees in all Direct Markets. Continue raising internal awareness of animal welfare and animal welfare initiatives. Generate an audit plan of vendors for biological Audits have been performed on site, virtually, matrices and initiate the conduct of on-site audits Achieved Complete the implementation and by questionnaire-based assessments. of these vendors. of the approved vendor list at external laboratories and conduct Implement use of approved vendor list for An approved vendor list has been implemented additional audits to expand list of biological matrices across organization and at Achieved across the organization, and implementation approved vendors of biological external vendors. efforts are ongoing for external laboratories. matrices. Introduction P|ESG Approach Impact Ambitions 49

2026 Corporate Responsibility & P|ESG Ambitions In 2026, we will continue strengthening our Corporate Responsibility and P|ESG framework by ensuring strong alignment with the Ascendis Pharma strategy and evolving regulatory requirements, and by advancing the processes that support consistent, accurate and transparent reporting across the organization. Patients Environmental • Continue to refine our • Continue and further standardize Scope 1 and Scope 2 greenhouse gas emissions reporting. approach to identifying • Further refine and improve Scope 3 data collection through engagement with key suppliers and partners. and prioritizing patient-related impacts, risks and opportunities that support our Social patient-centric strategy. • Attraction, onboarding, and retention of talent to ensure we have the right people to deliver on our • Continued enhancements to ambitions. our services, technology and • Continuous focus on leadership and employeeship development to enable the development, performance support model to ensure every and well-being of our people. patient receives timely, reliable and compassionate assistance • Maintain a strong focus on advancing gender pay initiatives and meeting evolving gender pay reporting throughout their treatment requirements. journey. • Amplify awareness of unmet • Continue to develop our human rights approach by strengthening third-party compliance practices and medical needs in growth maintaining accessible mechanisms for raising concerns. disorders through publication Governance of scientific data, educational events to reinforce scientific credibility and sustained • Continue building and strengthening our Compliance Framework in accordance with compliance areas engagement with patients and concurrently with Ascendis Pharma growth. HCPs. • Continue raising internal awareness of animal welfare and animal welfare initiatives. • Complete the implementation of the approved vendor list at external laboratories and conduct additional audits to expand list of approved vendors of biological matrices. Introduction P|ESG Approach Impact Ambitions 50

This report may contain forward-looking statements concerning our business, operations and financial performance and condition, as well as our plans, objectives and expectations for our business operations and financial performance and condition, including with relation to our corporate responsibility efforts. Any statements contained herein that are not statements of historical facts may be deemed to be forward-looking statements. In some cases, you can identify forward-looking statements by terminology such as “aim”, “anticipate”, “assume”, “believe”, “commit”, “contemplate”, “continue”, “could”, “due”, “estimate”, “expect”, “goal”, “intend”, “may”, “objective”, “plan”, “predict”, “potential”, “positioned”, “seek”, “should”, “target”, “will”, “would”, and other similar expressions that are predictions or indicate future events and future trends, or the negative of these terms or other comparable terminology. These forward-looking statements include our plans for 2026 and onwards with respect to our Corporate Responsibility and P|ESG strategy and ambitions. These forward-looking statements are based on senior management’s current expectations, estimates, forecasts and projections about our business and the industry in which we operate and involve known and unknown risks, uncertainties and other factors that are in some cases beyond our control. As a result, any or all of our forward-looking statements in this report may turn out to be inaccurate, perhaps materially so. The forward-looking statements speak only as of the date of this report. Except as required by law, we assume no obligation to update or revise these forward-looking statements for any reason, even if new information becomes available in the future. Given these risks and uncertainties, you are cautioned not to rely on such forward-looking statements as predictions of future events. Additionally, while various matters discussed herein may be significant to various stakeholders, such significance should not be understood as necessarily rising to the level of materiality under U.S. federal securities laws or other regimes. Materiality, particularly in the ESG context, is subject to various definitions, several of which are broader than the definition for SEC reporting purposes. We also make certain disclosures based on policies and practices that we believe provide reasonable support for certain matters; however, we cannot guarantee (nor should any language of “ensure” or similar be taken to mean) that such efforts will be successful in all circumstances. Moreover, certain of this information is subject to assumptions, estimates, or third-party information that we have not independently verified, as well as standards that are still evolving and may be subject to change. Moreover, while we aim to align certain of our disclosures and initiatives with the recommendations and expectations of various third-party frameworks, we cannot guarantee strict adherence to these frameworks’ recommendations. Our disclosures, as well as relevant internal controls, based on any standards may change due to revisions in framework requirements, availability or quality of information, changes in our business or applicable government policies, or other factors, some of which may be beyond our control. Finally, website and document references in this report are provided for convenience only; absent express language to the contrary, such materials are not incorporated to this report by reference. Introduction P|ESG Approach Impact Ambitions 51

Ascendis® Ascendis Pharma A/S (Company Reg. No. 29918791) Tuborg Boulevard 12 2900 Hellerup Denmark Tel: +45 70 22 22 44 Ascendis, Ascendis Pharma, Ascendis Pharma logo, the company logo, Skytrofa, Yorvipath, Ascendis Signature Access Program and TransCon are trademarks owned by the Ascendis Pharma Group. ©Ascendis Pharma A/S. February 2026. Version 1.0