LICENSE AGREEMENT
This LICENSE AGREEMENT (the “Agreement”) is executed as of December 18, 2025 (the “Effective Date”) by and between Ligand Pharmaceuticals Incorporated, a corporation organized under the laws of Delaware and having a place of business at 555 Heritage Dr., Suite 200, Jupiter, FL 33458 (“Ligand”) and Athira Pharma, Inc., a corporation organized under the laws of Delaware and having a place of business at 18706 North Creek Parkway, Suite 104, Bothell, WA, 98011(“Licensee”). Ligand and Licensee are each referred to herein by name or, individually, as a “Party” or, collectively, as “Parties.”
BACKGROUND
WHEREAS, Ligand Controls (as defined below) certain patent rights and know-how, which relate to Lasofoxifene (as defined below), including those rights it obtained pursuant to that certain Patent License Agreement by and between Ligand and Pfizer, Inc. (“Pfizer”) dated July 15, 2011 (the “Pfizer License”);
WHEREAS, Ligand and Sermonix Pharmaceuticals, Inc. (“Sermonix”) entered into a License Agreement, dated February 1, 2015 (as amended) (such date the “Original Effective Date” and such agreement, the “Original Agreement”) pursuant to which Ligand granted to Sermonix a worldwide exclusive license, under such patent rights and know-how, to develop and commercialize Lasofoxifene;
WHEREAS, Licensee and Sermonix are entering into a transaction pursuant to which Licensee is obtaining the exclusive right to develop and commercialize Lasofoxifene in the Territory (as defined below) (the “Sermonix-Athira License”) and, instead of receiving a sublicense from Sermonix under the Original Agreement, Licensee desires to obtain directly from Ligand an exclusive license, under Ligand’s patent rights and know-how, to develop and commercialize Lasofoxifene in the Territory and Sermonix desires to amend and restate the Original License Agreement to exclude the Territory; and
WHEREAS, Ligand desires to grant such license to Licensee, all in accordance with the terms and conditions herein, and to separately enter into such amended and restated agreement (the “A&R Agreement”) with Sermonix as of the Effective Date.
NOW, THEREFORE, in consideration of the mutual covenants and agreements provided herein below and other consideration, the receipt and sufficiency of which is hereby acknowledged, Ligand and Licensee hereby agree as follows:
As used in this Agreement, capitalized terms shall have the meanings indicated in this Article 1 or as specified elsewhere in this Agreement:
1.1“Additional Third Party License” has the meaning set forth in Section 3.4(g).
1.2“Affiliate” means, with respect to a Person, any Person that is controlled by, controls, or is under common control with such first Person, as the case may be. For purposes of this Section1.2, the term “control” means (a) direct or indirect ownership of fifty percent (50%) or more of the voting interest in the entity in question, or fifty percent (50%) or more interest in the income of the entity in question; provided, however, that if local Law requires a minimum percentage of local ownership of greater than fifty percent (50%), control will be established by direct or indirect beneficial ownership of one hundred percent (100%) of the maximum ownership percentage that may, under such local Law, be owned by foreign interests, or (b) possession, directly or indirectly, of the power to direct or cause the direction of management or policies of the entity in question (whether through ownership of securities or other ownership interests, by contract or otherwise).
1.4“Challenge” means if Licensee, its Affiliates or any Sublicensee: (a) institutes, or causes its counsel to institute, any interference, opposition, re-examination, review or similar proceeding with respect to any Licensed Patent with the U.S. Patent and Trademark Office or any foreign patent office; (b) makes any filing or institutes any legal proceeding, or causes its counsel to make any filing or institute any legal proceeding, with a court or other governmental body (including, the U.S. Patent and Trademark Office or any foreign patent office) in which one or more claims or allegations challenges the validity or enforceability of any Licensed Patent; or (c) makes any filing with or certification to, or causes its representative to make any filing with or certification to, the FDA pursuant to 21 U.S.C. § 355(b)(2)(A)(iv) or 21 U.S.C. § 355(j)(2)(A)(vii)(IV) with respect to any Licensed Patent.
1.5“Change of Control” means, with respect to a Party, (a) a merger or consolidation of such Party with a Third Party which results in the stockholders or equity holders of such Party not owning at least fifty percent (50%) of the combined voting power of the surviving entity immediately after such merger or consolidation, or (b) except in the case of a bona fide equity or debt financings, whether private or public, in which a Party issues new shares of its capital stock or securities convertible into shares of such Party, a transaction or series of related transactions in which a Third Party, together with its Affiliates, becomes the beneficial owner of fifty percent (50%) or more of the combined voting power of the outstanding securities of such Party, or (c) the sale or other transfer to a Third Party of all or substantially all of such Party’s business to which the subject matter of this Agreement relates.
1.6“Clinical Trial” means an investigation in human subjects and/or patients intended to discover or verify the clinical, pharmacological and/or other pharmacodynamic effects of
Product, and/or to identify any adverse reactions to Product, and/or to study absorption, distribution, metabolism, and/or excretion of Product with the objective of ascertaining its safety, activity and/or efficacy.
1.7“Commercially Reasonable Efforts” means [***].
1.8“Confidential Information” means any information of a confidential and proprietary nature, including know-how, information, invention disclosures, patent applications, proprietary materials and/or technologies, economic information, business or research strategies, trade secrets, and material embodiments thereof, disclosed by a Party to the other Party and characterized to the receiving Party as confidential. For clarity, [***].
1.9“Control” or “Controlled” means, with respect to any information, material or intellectual property right, that a Party owns or has a license to such information, material or intellectual property right, as applicable, and has the ability to grant to the other Party access to, or a license or sublicense under, such information, material or intellectual property right as provided under the terms of this Agreement. Notwithstanding the foregoing if use of any information, material or intellectual property right by Licensee requires that Ligand pay royalties, milestone payments or other consideration to a Third Party, then such information, material or intellectual property right shall only be deemed to be Controlled by Ligand if Licensee has agreed to pay its pro rata share of the financial consideration due to such Third Party.
1.10“Develop” or “Development” means pre-clinical and clinical research and development activities, including toxicology and other pre-clinical development efforts, stability testing, process development, formulation development, delivery system development, quality assurance and quality control development, statistical analysis, clinical pharmacology, clinical studies (including Clinical Trials), regulatory affairs, and Regulatory Approval and clinical study regulatory activities.
1.11 “Executive” shall mean for Ligand, [***] (or such individual’s designee), and, for Licensee, [***] (or such individual’s designee). [***].
1.12“FCPA” has the meaning set forth in Section 7.2(b).
1.13“FD&C Act” means the U.S. Federal Food, Drug, and Cosmetic Act (21 U.S.C. §301, et seq.), including any amendments or supplements thereto.
1.14“FDA” means the U.S. Food and Drug Administration, or any successor agency thereto.
1.15“Field” means the treatment or prevention of human diseases.
1.16“First Commercial Sale” means the first sale of Product by Licensee or its Affiliates or Sublicensees to a Third Party for which payment (cash or non-cash) has been received in any country in the Territory.
1.17“Generic Product” means any pharmaceutical product that (a) is sold by [***], (b) contains [***], (c) has [***] and (d) [***] or, [***].
1.18“Geographical Asia” means the countries and territories set forth on Exhibit A.
1.19“Governmental Entity” means any regional, central, federal, state, provincial or local court, commission or governmental, regulatory or administrative body, board, bureau, agency, instrumentality, authority or tribunal or any subdivision thereof.
1.20“Government Official” has the meaning set forth in Section 7.2(a).
1.21“Improvement” means any discovery, invention, contribution, method, finding, or improvement, whether or not patentable, and all intellectual property therein, that is conceived, reduced to practice, or otherwise developed by or on behalf of a Party, during the Term, that is a modification, improvement or enhancement to the Licensed Patents and is dominated by the claims of one or more of the Patent rights described in Section 1.30.
1.22“IND” means an Investigational New Drug application, Clinical Study Application, Clinical Trial Exemption, or similar application or submission for approval to conduct Clinical Trials filed with or submitted to a Regulatory Authority in the applicable jurisdiction in conformance with the requirements of such Regulatory Authority.
1.23 “Indication” means a medically recognized disease or disorder which in all cases requires a Pivotal Clinical Study for Regulatory Approval for the relevant Product label. Notwithstanding the foregoing, [***].
1.24“Infringement Claim” has the meaning set forth in Section 8.1(b).
1.25“Intellectual Property Rights” means Patents, copyrights, database rights, Know-How and similar rights of any type (excluding trademarks) under the laws of any Governmental Entity, including all applications, registrations, extensions and renewals relating to any of the foregoing.
1.26“Know-How” means all technical information and other technical subject matter, proprietary methods, ideas, concepts, formulations, discoveries, inventions, devices, technology, trade secrets, compositions, designs, formulae, know-how, show-how, specifications, drawings, techniques, results, data, processes, methods, procedures, designs and regulatory correspondence and information (including pharmacological, toxicological, pre-clinical, clinical and manufacturing test data, manufacturing protocols, analytical methods and data, quality control data and process validation) whether or not patentable.
1.27“Lasofoxifene” means that compound known as lasofoxifene tartrate as identified in Exhibit B.
1.28“Law” means, individually and collectively, any and all laws, ordinances, orders, rules, rulings, directives and regulations of any kind whatsoever of any Governmental Entity or Regulatory Authority within the applicable jurisdiction.
1.29 “Licensed Know-How” means all Know-How Controlled by Ligand or any of its Affiliates as of the Original Effective Date that is (a) necessary or useful in connection with developing, making, using, selling, offering to sell, exporting and importing Product in the Territory and (b) not included in the Licensed Patents.
1.30“Licensed Patents” means those Patents Controlled by Ligand or any of its Affiliates listed in Schedule 1.30 attached hereto.
1.31“Licensed Technology” means the Licensed Know-How and the Licensed Patents.
1.32“Licensee Improvements” means any and all Improvements to the Licensed Technology and any and all Licensee Patents that claim any composition or method of making or method of using Lasofoxifene or otherwise necessary in connection with the Development, manufacture, use or commercialization of Lasofoxifene and/or Product, which are Controlled or owned by Licensee or any of its Affiliates during the Term (other than Ligand Patent rights pursuant to the licenses granted hereunder).
1.33“Licensee Indemnitees” has the meaning set forth in Section 8.2.
1.34“Ligand Indemnitees” has the meaning set forth in Section 8.1(a).
1.35“Litigation Costs” has the meaning set forth in Section 8.1(a).
1.36“Losses” has the meaning set forth in Section 8.1(a).
1.37“NDA” means a “New Drug Application,” as defined in the FD&C Act and applicable regulations promulgated thereunder by the FDA and all amendments and supplements thereto filed with the FDA, or the equivalent application filed with any Regulatory Authority, including all documents, data, and other information concerning Product, which are necessary for gaining Regulatory Approval to market and sell Product in the relevant jurisdiction.
1.38“Net Sales” means [***], less the following deductions, [***].
In the event a Product is sold in a package or formulated in combination with one or more other active ingredients that are not Products (as used in this definition of Net Sales, a “Combination Product”), then [***].
1.41“Other Covered Party” has the meaning set forth in Section 7.2(a).
1.42“Patents” means all: (a) United States and foreign patents, re-examinations, reissues, renewals, extensions and term restorations, inventors’ certificates and counterparts thereof; and (b) pending applications for United States and foreign patents, including, provisional applications, continuations, continued prosecution, divisional and substitute applications, and counterparts thereof.
1.43“Phase 1a Trial” means a Clinical Trial that would satisfy the requirements of 21 C.F.R. Part 312.21(a) (as amended from time to time) or other comparable regulation imposed by an applicable Regulatory Authority in any country other than the United States, the principal purposes of which are to generate sufficient data on safety in humans to commence a Phase 2 Trial. For purposes of this Agreement, ‘initiation’ of a Phase 1a Trial means the first [***] a human subject in a Phase 1a Trial.
1.44“Phase 2 Trial” means a Clinical Trial that would satisfy the requirements of 21 C.F.R. Part 312.21(b) (as amended from time to time) or other comparable regulation imposed by an applicable Regulatory Authority in any country other than the United States, the principal purposes of which are to make a preliminary determination that a Product is safe for its intended use and to obtain sufficient information about such Product’s efficacy to permit the design of further clinical trials. For purposes of this Agreement, ‘initiation’ of a Phase 2 Trial means the first [***] a human subject in a Phase 2 Trial.
1.45“Phase 3 Trial” means a Clinical Trial on sufficient numbers of patients that is designed to establish that a Product is safe and efficacious for its intended use, and to define warnings, precautions and adverse reactions that are associated with such Product in the dosage range to be prescribed, and to support Regulatory Approval of such Product or label expansion of such Product. For purposes of this Agreement, ‘initiation’ of a Phase 3 Trial means the first [***] a human subject in a Phase 3 Trial.
1.46“Person” means any individual, corporation, partnership, association, joint-stock company, trust, unincorporated organization or government or political subdivision thereof.
1.47“Pivotal Clinical Study” means a Phase 3 Trial that [***].
1.48“Product” means, subject to Section 2.1(b), all preparations, compositions and formulations intended for oral use in the Field that contain Lasofoxifene, whether alone or in combination with other active pharmaceutical ingredients. [***].
1.49“Prosecute” or “Prosecution” means, with respect to Patents, the filing for, prosecuting, responding to oppositions, nullity actions, re-examinations, revocation actions and similar proceedings (including conducting or participating in interference and oppositions) filed by Third Parties against, and maintaining, Patents.
1.50“Regulatory Approval” means, with respect to a country or jurisdiction within the Territory, any approvals, licenses, registrations or authorizations necessary for the manufacture, marketing and sale of a Product in such country or jurisdiction.
1.51“Regulatory Authority” means any national (e.g., the FDA), supranational (e.g., the European Medicines Agency), regional, state or local regulatory agency, department bureau, commission, council or other Governmental Entity in any jurisdiction of the world involved in the granting of Regulatory Approval for pharmaceutical products.
1.52“Regulatory Documentation” means all submissions to Regulatory Authorities and other Governmental Entities, including for Clinical Trials, preclinical trials, tests, and biostudies, relating to a Product, including all INDs, NDAs and Regulatory Approvals, as well as all correspondence with Governmental Entities (registration and licenses, pricing and reimbursement correspondence, regulatory drug lists, advertising and promotion documents), adverse event files, complaint files, manufacturing records and inspection reports.
1.53 “Research Plan” has the meaning set forth in Section 4.2(a).
1.54“Retained Field” means all preparations, compositions and formulations of product containing Lasofoxifene other than for oral use in the Field in the Territory.
1.55“Royalty Term” has the meaning set forth in Section 3.4(d).
1.56“Sublicense Agreement” has the meaning set forth in Section 2.4(a).
1.57“Sublicense Revenues” means amounts (including, any licensing fees, or license maintenance fees, or milestone payments) paid to the Licensee by any Sublicensee under a Sublicense Agreement, provided that Sublicense Revenues will not include [***].
1.58“Sublicensee” means a Third Party to whom Licensee grants a sublicense hereunder to sell, offer for sale or import a Product, but excluding wholesalers and other physical distributors. For the avoidance of doubt, [***].
1.59“Term” has the meaning set forth in Section 10.1.
1.60“Territory” means worldwide other than Geographical Asia.
1.61“Third Party” means any Person other than Ligand, Licensee or any Affiliate of either Ligand or Licensee.
1.63 “Valid Claim” means (a) any claim of [***] patent within the Licensed Patents that has not been [***], and which patent has not been [***], or (b) [***].
1.64Unless the context of this Agreement otherwise requires: (a) words of any gender include each other gender; (b) words using the singular or plural number also include the plural or
singular number, respectively; (c) the terms “hereof,” “herein,” “hereby” and derivative or similar words refer to this entire Agreement; (d) the terms “Article,” “Section” or “Exhibit” refer to the specified Article, Section or Exhibit of this Agreement; and (e) the term “including” means “including without limitation.” Whenever this Agreement refers to a number of days, such number shall refer to calendar days.
ARTICLE 2
LICENSES AND TECHNOLOGY TRANSFER
2.1Exclusive License for the Products.
(a)During the Term, subject to the terms and conditions of this Agreement, Ligand hereby grants to Licensee an exclusive, royalty-bearing right and license under the Licensed Technology to develop, make, have made, use, sell, have sold, import and export Products in the Field in the Territory. For clarity, in the foregoing sentence, “exclusive” means that Ligand shall not for its own account, and shall not grant to any Third Party the right and license under the Licensed Technology to, develop, make, have made, use, sell, have sold, import and export Products in the Field in the Territory.
2.2Rights to Improvements. Licensee and its permitted Sublicensees shall have the right to make Improvements to the Licensed Technology, and to utilize such Improvements to develop, make, have made, use, sell, have sold, import and export Products in the Field in the Territory.
2.3License Back to Ligand. Subject to the terms and conditions of this Agreement, Licensee hereby grants Ligand a non-exclusive, perpetual, irrevocable, royalty-free, fully paid-up worldwide license, with the right to sublicense through multiple tiers, under the Licensee Improvements to develop, seek regulatory approval for, make, have made, import, export, use, offer for sale and sell products comprising Lasofoxifene other than Products.
(a)The rights and licenses granted pursuant to Section 2.1 include the right to grant sublicenses pursuant to a written sublicense agreement (each a “Sublicense Agreement”); provided, however, that (i) any such Sublicense Agreement shall be consistent with and subject to the terms and conditions of this Agreement; (ii) Licensee shall remain fully responsible to Ligand for the performance of its Sublicensee(s) with respect to Licensee’s obligations under the terms of this Agreement; (iii) [***].
(b)Should this Agreement terminate for any reason, at the request of Licensee or any of its Sublicensees, Ligand shall grant such Sublicensee a direct license to the Licensed Technology in the Territory and the Field on the same terms applicable to Licensee under this Agreement and limited to the scope of rights granted by Licensee to a Sublicensee. In no event shall Ligand have greater obligations under any such written agreement than Ligand has under this Agreement.
2.5Technology Transfer. Ligand acknowledges and agrees that Sermonix has or will disclose and provide to Licensee all tangible embodiments of data and information concerning the Licensed Technology and Regulatory Documentation (including without limitation all safety data for the Product) that is in Sermonix’s possession as of the Effective Date and was disclosed and provided to Sermonix by Ligand pursuant to the Original Agreement. Licensee is permitted to maintain a global safety database for the Product throughout the Term and to receive safety data for the Product from Sermonix for inclusion in the global safety database and to disclose safety data from the global safety database to Sermonix for its use in Geographical Asia.
2.7Manufacturing. Licensee shall be solely responsible for manufacturing and supplying Lasofoxifene and finished dosage form of Product for commercialization in the Territory. In this role, Licensee shall identify and manage Third Party contract manufacturers, as well as lead all supply chain management and quality control activities.
2.8No Other Rights. Ligand and Licensee each acknowledges and agrees that, except as expressly granted under this Agreement, no right, title, or interest of any nature whatsoever is granted whether by implication, estoppel, reliance, or otherwise, by either Party to the other Party. All rights with respect to technology, Patents or other Intellectual Property Rights that are not specifically granted herein are reserved.
2.9Bankruptcy. All rights and licenses granted under or pursuant to this Agreement, including amendments hereto, are, for all purposes of 11 U.S.C. § 365(n), licenses of rights to intellectual property as defined in the United States Bankruptcy Code, and any comparable Law of a relevant jurisdiction. Each Party may elect to retain and may fully exercise all of its rights and elections under 11 U.S.C. § 365(n). The Parties further agree that, in the event of the commencement a bankruptcy proceeding by or against Ligand under the United States Bankruptcy Code, Licensee shall be entitled to a complete duplicate of (or complete access to, as appropriate) any intellectual property licensed to Licensee and all embodiments of such intellectual property, which, if not already in Licensee’s possession, shall be promptly delivered to it (a) upon any such commencement of a bankruptcy proceeding upon Licensee’s written request therefor, unless Ligand elects to continue to perform all of its obligations under this Agreement or (b) if not delivered under clause (a), following the rejection of this Agreement by Ligand upon written request therefor by Licensee.
3.2Regulatory Milestone Payments. In further consideration of the rights and licenses granted by Ligand hereunder, on a Product-by-Product basis, Licensee shall pay to Ligand the non-refundable and non-creditable milestone payments set forth below for the achievement by Licensee or its Affiliates or Sublicensees of each of the corresponding events, said milestone payments to be made upon no later [***] after [***] provided such milestone payments shall be accelerated and due and payable in their entirety within [***] after either (i) [***] or (ii) [***]:
|
|
Milestone |
Payment |
[***] |
[***] |
[***] |
[***] |
3.3Commercial Milestone Payments. In further consideration of the rights and licenses granted by Ligand hereunder, on a Product-by-Product basis, Licensee shall pay to Ligand the non-refundable and non-creditable milestone payments set forth below within [***] of the achievement by Licensee or its Affiliates or Sublicensees of each of the corresponding events:
|
|
Milestone |
Payment |
[***] |
[***] |
[***] |
[***] |
[***] |
[***] |
(a)Royalty Rates. In further consideration of the rights and licenses granted by Ligand hereunder, Licensee shall pay to Ligand a tiered royalty on aggregate Net Sales of Product sold by Licensee, its Affiliates and Sublicensees during the Royalty Term as follows:
|
|
Net Sales in a Single Calendar Year |
Royalty rate |
[***] |
6% |
[***] |
[***] |
[***] |
10% |
(b)Sublicensing. In the event Licensee grants a sublicense under Section 2.4 to a Sublicensee to develop, make, use, sell, offer to sell, import or export a Product, the applicable Sublicense Agreement shall require the Sublicensee to [***].
(c)Payment of Royalties. Licensee shall pay on a [***] basis all royalties due and payable on Net Sales of Product in each [***] pursuant to this Section 3.4 within [***] after [***]; provided, that [***].
(d)Royalty Term. Notwithstanding anything to the contrary, the royalties under this Section 3.4 shall be payable in respect of each Product until the end of the Term; provided, however, that the royalty rate for Licensee shall be reduced by [***] on a country-by-country, Product-by-Product basis upon the later of (i) the expiry of the last Valid Claim in the last Licensed Patent in such country including any extensions thereto, (ii) the expiration of any market exclusivity or data exclusivity granted by the applicable Regulatory Authority for such Product in the Territory, and (iii) fifteen (15) years after the First Commercial Sale of such Product in such country (the “Royalty Term”). Licensee acknowledges that such reduced royalty is fair and reasonable in order to compensate Ligand for Licensee's continuing license of the Ligand Know-How.
(e)Third Party Payments on Products. Licensee shall be responsible for [***] in connection with [***].
(f)Generic Entry. Notwithstanding the foregoing, (i) for Net Sales based on sales of a Product in a country in the Territory, any payments owed with respect to such Product pursuant to this Section 3.4 will be reduced by [***] for [***], if at any time a Generic Product [***].
(g)Third Party Patents. If the Intellectual Property Rights from any Third Party cover the composition of matter of Lasofoxifene in a country in the Territory, then Licensee may negotiate and obtain a license under, or otherwise pay amounts with respect to any litigation regarding, such Third Party’s Intellectual Property Right(s) (each such Third Party license or payment referred to herein as an “Additional Third Party License”). Any royalty otherwise payable to Ligand under this Agreement with respect to Net Sales of any Product by Licensee, its Affiliates or Sublicensees in such country will be reduced by [***] of [***] to Third Parties pursuant to any Additional Third Party Licenses, such reduction to continue (and be carried forward for use) until all such amounts have been expended, provided that, in no event will [***] be less than [***].
3.5Sublicense Revenues. In addition to any payments due under Sections 3.2, 3.3 or 3.4 as a result of a Sublicensee’s activities, the Licensee shall pay to Ligand [***] of all Sublicense Revenues within [***].
3.6Payment Method. All payments made by Licensee under this Agreement shall be made in U.S. Dollars, and such payments shall be made by check or wire transfer to one or more bank accounts to be designated in writing by Ligand.
3.7Currency Conversion. In the event that Products are sold in currencies other than U.S. Dollars, Net Sales shall be calculated by Licensee in accordance with [***]. Net Sales in currencies other than U.S. Dollars shall be converted into U.S. Dollars using [***].
3.8Late Payment Interest. Any payment due and payable to Ligand under the terms and conditions of this Agreement, including any royalty payment, made by Licensee after the date such payment is due and payable shall bear interest as of the day after the date such payment was due and payable and shall continue to accrue such interest until such payment is made at a rate equal to [***].
3.9Records and Reports. All payments made to Ligand hereunder shall be accompanied by a written statement setting forth in reasonable detail the calculation thereof, including, [***]. Licensee shall maintain complete and accurate records sufficient to enable accurate calculation of royalties and other payments due Ligand hereunder. Such records and books of account shall be preserved by Licensee for a period of [***], which obligation shall survive termination of this Agreement. Licensee must ensure that its Sublicensees provide reports and keep records in a manner consistent with this Section 3.9. Licensee shall provide reports received from Sublicensees to Ligand with the applicable payment.
3.10Taxes. Licensee may withhold from payment made to Ligand under this Agreement any income tax required to be withheld by Licensee under the laws of the country or jurisdiction where Licensee has commercially sold Products. If any tax is withheld by Licensee, Licensee shall provide Ligand receipts or other evidence of such withholding and payment to the appropriate tax authorities on a timely basis following that tax payment. Each Party agrees to cooperate with the other Party in claiming refunds or exemptions from such deductions or withholdings under any relevant agreement or treaty which is in effect. The Parties shall discuss applicable mechanisms for minimizing such taxes to the extent possible in compliance with applicable Law. In addition, the Parties shall cooperate in accordance with applicable Law to minimize indirect taxes (such as value added tax, sales tax, consumption tax and other similar taxes) in connection with this Agreement.
3.11Audit Rights. Licensee shall permit an independent public accountant designated by Ligand and reasonably acceptable to Licensee, to have access, no more than [***], to Licensee’s records and books to the extent necessary to determine the accuracy of Net Sales reported, and payments made, by Licensee to Ligand within [***]. The independent public accountant shall disclose to Ligand only (a) the accuracy of Net Sales reported and the basis for royalty and other payments made to Ligand under this Agreement and (b) the difference, if any, such reported and paid amounts vary from amounts determined as a result of the audit. If such examination results in a determination that Net Sales or payments have been misstated, over or under paid amounts due shall be paid promptly to the appropriate Party. If [***], the fees and expenses of such accountant shall be paid by Licensee; otherwise the fees and expenses of such accountant shall be paid by Ligand. All matters reviewed by such independent public accountant shall be deemed Confidential Information of Licensee subject to Article 6.
ARTICLE 4
PRODUCT ACTIVITIES
(a)Licensee shall diligently Develop, manufacture and sell Products, and shall use Commercially Reasonable Efforts to develop markets for Products, in both cases either directly or through a Sublicensee. Licensee, either directly or through a Sublicensee, shall obtain all necessary Regulatory Approvals in each country where Products are made, used, sold, imported, or offered for sale.
(b)In addition, without limiting Licensee’s obligations under Section 4.1(a), Licensee will use its Commercially Reasonably Efforts to develop the Product for the treatment of metastatic breast cancer.
(d)Licensee shall use Commercially Reasonable Efforts to obtain Regulatory Approval, including [***], for the Product in Europe.
4.2Research Plan; Progress Reports.
(a)Licensee shall develop a research plan detailing the work it will perform and associated timelines to Develop Products and to obtain Regulatory Approval and sell Products throughout the Territory (the “Research Plan”). Licensee will provide a copy of the current Research Plan to Ligand within [***] and any updates on [***] basis.
(b)By [***], Licensee shall submit a written report to Ligand covering [***]. Each report will describe: [***]. Licensee shall also provide to Ligand copies of any similar progress reports received from its Sublicensees, within [***]. In addition, upon the reasonable request of Ligand but no more frequently than [***], Licensee and Ligand shall meet in-person at a mutually agreeable location to discuss [***].
4.3Development. Licensee shall be solely responsible for the Development of all Products in the Field in the Territory. Licensee shall bear one hundred percent (100%) of all costs and expenses associated with the Development of Products.
4.4Regulatory Responsibilities.
(a)Licensee shall bear one hundred percent (100%) of all costs and expenses associated with regulatory activities related to the Products in the Field in the Territory.
(b)Ligand has the right to request copies of [***] and significant correspondence to and from all Regulatory Authorities related to [***]; provided, [***]. Each Party shall provide the other Party with reasonable cooperation and assistance in connection with regulatory activities for Lasofoxifene, including [***].
(c)Licensee shall be responsible for ensuring, at its sole expense, that the Development and commercialization of all Products in the applicable jurisdiction within the Territory are in compliance with applicable Laws in all material respects, including all rules and regulations promulgated by applicable Regulatory Authorities. Specifically and without limiting the foregoing, Licensee shall file all compliance filings, certificates and safety reporting for the Products at its sole expense in the Territory.
(d)Licensee shall be responsible for taking all actions related to adverse event reporting and other regulatory obligations that are legally required of the holder of a Regulatory Approval application, license, registration or authorization in the Territory.
4.5Commercialization Plan. Commencing [***], Licensee shall commence preparing a Commercialization Plan (“Commercialization Plan”) for such Product. No later than [***], Licensee shall submit such Commercialization Plan to Ligand for review. After the launch of such Product, on [***] basis, Licensee shall [***].
4.6Commercialization. Licensee shall be solely responsible for the commercialization of all Products in the Field in the Territory. Licensee shall bear one hundred percent (100%) of all costs and expenses associated with commercialization of all Products in the Field in the Territory.
4.7Transfer; Rights of Reference. The Parties hereby acknowledge and agree that (a) pursuant to the Original Agreement, Ligand transferred, on behalf of itself and its Affiliates, to Sermonix the INDs and NDAs for the Product set forth on Exhibit C and (b) pursuant to the Sermonix-Athira License, Sermonix has or will transfer to Licensee such INDs and NDAs. Ligand, its Affiliates and its sublicensees shall have a right of reference to the Regulatory Documentation for Lasofoxifene controlled by Licensee, to rely upon, and otherwise use, an investigation for the purpose of obtaining approval of an application for a product in the Retained Field, including the ability to make available the underlying raw data in its possession in documented or electronic form from the investigation for FDA audit, if necessary. Licensee, its Affiliates and its sublicensees shall have a right of reference to the Regulatory Documentation for Lasofoxifene controlled by Ligand, its Affiliates and/or its sublicensees, to rely upon, and otherwise use, an investigation for the purpose of obtaining approval of an application for a Product in the Field in the Territory, including the ability to make available the underlying raw data in its possession in documented or electronic form from the investigation for FDA audit, if necessary.
ARTICLE 5
INTELLECTUAL PROPERTY
5.1Patent Maintenance and Prosecution.
(a)Ligand shall, [***] Prosecute the Licensed Patents; provided, however, (1) subject to clause 3 below, in the event that [***] in any country [***], then Licensee and its Sublicensees shall have the right, [***], to Prosecute the Licensed Patents in such country; (2) subject to clause 3 below, Licensee shall have the right to Prosecute the Licensed Patents in the event [***]; and (3) [***]. The Prosecuting Party shall make available to the Non-Prosecuting
Party copies of material correspondence with any patent office regarding the Licensed Patents. The Party that does not possess the right to Prosecute the Licensed Patents as provided above (the “Non-Prosecuting Party”) [***], and the Party that does possess the right to Prosecute the Licensed Patents [***]. Solely for the period of time [***], for as long as Ligand is the Prosecuting Party, Licensee shall [***]. In the event that the Prosecuting Party (which for purposes of this Section 5.1(a) shall include all applicable licensors) decides to cease activities relating to Prosecuting any Licensed Patents, the Prosecuting Party shall provide written notice thereof to the Non-Prosecuting Party and, prior to taking action that would result in the abandonment of any such Licensed Patent, the Prosecuting Party shall [***] with the Non-Prosecuting Party, [***] at least [***]. Thereafter, the Non-Prosecuting Party may, but is not required to, undertake, [***], the Prosecution of such Licensed Patent, and the Prosecuting Party shall cooperate. In addition, if Licensee no longer desires a license to one or more Licensed Patents, Licensee shall provide written notice thereof to Licensee and effective [***], such Patent(s) shall no longer be deemed Licensed Patent(s).
(b)Licensee shall, [***], Prosecute any Patents covering Improvements.
5.2Licensed Patents and Licensed Know-How Enforcement and Defense.
(a)Notification. Each Party shall notify the other Party of any infringement of any of the Licensed Patents or misappropriation of any of the Licensed Know-How by a Third Party in the Field that becomes known to such Party, and of any claim by a Third Party that the activities of a Party relating to a Product infringe Patent rights or misappropriate other Intellectual Property Rights of such Third Party.
(b)Licensed Patents. [***], as between the Parties, Licensee (which for purposes of this Section 5.2(b) shall include all Sublicensees) shall have the first right, but not an obligation, to initiate, maintain and control, [***], legal action against any infringement of the Licensed Patents by a Third Party Product in the Field in the Territory. In the event that Licensee initiates legal action against infringement of the Licensed Patents by a Third Party, Licensee shall notify Ligand in writing. In order to establish standing, Ligand, upon request of Licensee, agrees to [***] commence or to join in any such litigation, [***], and in any event to cooperate with Licensee in such litigation [***]. If Licensee does not take steps to defend or enforce the Licensed Patents, Ligand shall have the right, but not an obligation, to initiate, maintain and control, [***], legal action against any infringement of the Licensed Patents by a Third Party Product in the Field in the Territory. Any recovery received by a Party from legal action initiated pursuant to this Section 5.2, whether by judgment, award, decree or settlement, shall be [***]. For the avoidance of doubt, as between the Parties, Ligand shall retain the sole right, but not an obligation, to initiate, maintain and control legal action against any infringement of the Licensed Patents by a Third Party in the Retained Field in the Territory or in any field outside the Territory.
5.3Cooperation. In any suit, proceeding or dispute involving the infringement of any of the Licensed Patents in the Field or misappropriation of any of the Licensed Know-How in the Field, the Parties shall provide each other with [***] cooperation, and, upon the request [***] of the Party bringing suit, the other Party shall make available to the Party bringing suit, [***].
6.1Confidentiality Obligations. Each Party agrees that, during the Term and for [***] thereafter, all Confidential Information of the other Party shall be maintained in strict confidence, and shall not be used for any purpose other than the purposes expressly permitted by this Agreement, and, subject to Section 6.2, shall not be disclosed to any Third Party. The foregoing obligations will not apply to any portion of Confidential Information to the extent that it can be established by competent proof that such portion:
(a)was already known to the recipient as evidenced by its written records, other than under an obligation of confidentiality, at the time of disclosure;
(b)was generally available to the public or was otherwise part of the public domain at the time of its disclosure to the recipient;
(c)became generally available to the public or otherwise becomes part of the public domain after its disclosure and other than through any act or omission of the recipient in breach of this Agreement; or
(d)was subsequently lawfully disclosed to the recipient by a Third Party other than in contravention of a confidentiality obligation of such Third Party to the disclosing party.
6.2Permitted Usage. Each Party may use and disclose Confidential Information of the other Party, in accordance with this Agreement, as follows: (a) under appropriate confidentiality provisions no less restrictive than those in this Agreement, in connection with the performance of its obligations or exercise of rights granted to or retained by such Party; (b) in connection with the Prosecution or enforcement of Licensed Patents or Improvements, in accordance with this Agreement; or (c) in connection with prosecuting or defending litigation, complying with applicable governmental regulations, filing for, obtaining and maintaining Regulatory Approvals, or as otherwise required by Law, but provided that if a Party is required by Law to make any disclosure of the other Party’s Confidential Information, it will give [***] notice to the other Party of such disclosure requirement, it will disclose only [***], and it will [***] to secure confidential treatment of such Confidential Information required to be disclosed.
6.3Terms of Agreement. The terms of this Agreement shall be Confidential Information of both Parties, and subject to the terms of this Article 6. Notwithstanding the foregoing, [***]. Except as otherwise permitted for disclosures pursuant to this Article 6, the disclosing Party shall [***] to preserve the confidentiality of this Agreement and the terms thereof notwithstanding any required disclosure. A Party will give the other Party written notice of any required disclosure under (ii) above, which notice shall [***]. In the event either Party is required to file this Agreement with the U.S. Securities and Exchange Commission or any comparable non-U.S. Governmental Entity, such Party shall apply for confidential treatment of this Agreement to the fullest extent permitted by applicable Law, shall provide the other Party a copy of the confidential treatment request [***], and [***].
6.4Public Announcements. Upon [***], Ligand shall be permitted to issue a press release which press release may contain, [***]. Neither Party shall make any subsequent public announcement concerning this Agreement or the terms hereof not previously made public without [***]. Notwithstanding the foregoing, any [***] shall not require the prior approval of Licensee.
ARTICLE 7
Representations, Warranties and Covenants
7.1General. Each Party represents and warrants to the other that:
(a)it is duly organized and validly existing under the Law of the jurisdiction of its incorporation, and has full corporate power and authority to enter into this Agreement and to carry out the provisions hereof;
(b)it is qualified to do business and is in good standing in each jurisdiction in which it conducts business;
(c)it is duly authorized to execute and deliver this Agreement and to perform its obligations hereunder, and the individual executing this Agreement on its behalf has been duly authorized to do so by all requisite corporate action;
(d)this Agreement is legally binding upon it and enforceable in accordance with its terms and the execution, delivery and performance of this Agreement by it does not conflict with any agreement, instrument or understanding, oral or written, to which it is a party or by which it may be bound, nor violate any material Law; and
(e)it is not aware of any action, suit or inquiry or investigation instituted by any Person which questions or threatens the validity of this Agreement.
7.2Representations and Covenants of Licensee.
(a)Licensee has not, directly or indirectly, offered, promised, paid, authorized or given, and will not in the future, offer, promise, pay, authorize or give, money or anything of value, directly or indirectly, to any Government Official (as defined below) or Other Covered Party (as defined below) for the purpose of: (i) influencing any act or decision of the Government Official or Other Covered Party; (ii) inducing the Government Official or Other Covered Party to do or omit to do an act in violation of a lawful duty; (iii) securing any improper advantage; or (iv) inducing the Government Official or Other Covered Party to influence the act or decision of a government or government instrumentality, in order to obtain or retain business, or direct business to, any person or entity, in any way related to this Agreement.
For purposes of this Agreement: (i) “Government Official” means any official, officer, employee or representative of: (A) any federal, state, provincial, county or municipal government or any department or agency thereof; (B) any public international organization or any department or agency thereof; or (C) any company or other entity owned or controlled by any government; and
(ii) “Other Covered Party” means any political party or party official, or any candidate for political office.
(b)Anti-Corruption Compliance.
(1)In performing under this Agreement, Licensee and its Affiliates agree to comply with all applicable anti-corruption laws, including the Foreign Corrupt Practices Act of 1977, as amended (“FCPA”) and all laws enacted to implement the OECD Convention on Combating Bribery of Foreign Officials in International Business Transactions.
(2)Licensee is not aware of any Government Official or Other Covered Party having any financial interest in the subject matter of this Agreement or in any way personally benefiting, directly or indirectly, from this Agreement.
(3)No political contributions or charitable donations shall be given, offered, promised or paid at the request of any Government Official or Other Covered Party that is in any way related to this Agreement or any related activity, without Ligand's prior written approval.
(4)In the event that Licensee violates the FCPA or any applicable anti-corruption law or breaches any provision in this Section, Ligand shall have the right to unilaterally terminate this Agreement. In addition, Licensee shall defend, indemnify and hold harmless Ligand from and against any and all costs, damages, losses, liabilities, expenses, judgments, fines, settlements and any other amounts of any nature, including reasonable attorneys' fees arising from any improper payment made in violation of the FCPA, any applicable anti-corruption laws or this Section, directly or indirectly, by, on behalf of or with the knowledge of the Licensee, in relation to this Agreement.
7.3Representations of Ligand. Ligand hereby represents and warrants to Licensee that:
(a)except for the Licensed Technology [***] expressly disclosed in Schedule 7.3(a), [***] Licensed Technology [***];
(b)it has and will have the full right, power and authority to grant all of the right, title and interest in the licenses and other rights granted or to be granted to Licensee, Licensee’s Affiliates or Licensee’s Sublicensees under this Agreement (provided [***]);
(c)there are no adverse actions, suits, or claims pending or to [***], threatened against Ligand in any court or by or before any Governmental Entity with respect to Lasofoxifene or the Licensed Technology and, to [***], there are no Third Party Patents that would reasonably be expected to give rise to such actions, suits or claims. No Third Party has challenged the ownership, scope, duration, validity enforceability, priority or right to use the Licensed Technology;
(d)Ligand has not initiated or been involved in any proceedings or claims in which it alleges that any Third Party is or was infringing or misappropriating the Licensed Technology, nor have any proceedings been threatened by Ligand, nor to [***] is there any valid basis for any such proceeding;
(e)to [***] and notwithstanding 35 USC §271(e)(1) or any comparable Laws, the commercialization of Lasofoxifene does not infringe or misappropriate any Patent rights, know-how or other Intellectual Property Rights of any Third Party;
(f)the execution, delivery and performance by Ligand of this Agreement and its compliance with the terms and provisions hereof does not and will not violate or result, in any material respect, in a breach of or default under any binding obligation or agreement of Ligand existing as of the Effective Date;
(a)Ligand covenants that it will not, during the Term, undertake any obligation, or grant any right, license, interest or lien, that conflicts with its obligations, or the rights and licenses granted to Licensee, under the terms of this Agreement, or impairs the rights granted by Ligand to Licensee under the terms of this Agreement.
(b)Ligand has not, directly or indirectly, offered, promised, paid, authorized or given, and will not in the future, offer, promise, pay, authorize or give, money or anything of value, directly or indirectly, to any Government Official or Other Covered Party for the purpose of: (i) influencing any act or decision of the Government Official or Other Covered Party; (ii) inducing the Government Official or Other Covered Party to do or omit to do an act in violation of a lawful duty; (iii) securing any improper advantage; or (iv) inducing the Government Official or Other Covered Party to influence the act or decision of a government or government instrumentality, in order to obtain or retain business, or direct business to, any person or entity, in any way related to this Agreement.
(c)Anti-Corruption Compliance.
(1)In performing under this Agreement, Ligand and its Affiliates agree to comply with all applicable anti-corruption laws, including the FCPA and all laws enacted to implement the OECD Convention on Combating Bribery of Foreign Officials in International Business Transactions.
(2)Ligand is not aware of any Government Official or Other Covered Party having any financial interest in the subject matter of this Agreement or in any way personally benefiting, directly or indirectly, from this Agreement.
(3)No political contributions or charitable donations shall be given, offered, promised or paid at the request of any Government Official or Other Covered Party that is in any way related to this Agreement or any related activity, without Licensee's prior written approval.
(4)In the event that Ligand violates the FCPA or any applicable anti-corruption law or breaches any provision in this Section, Licensee shall have the right to unilaterally terminate this Agreement. In addition, Ligand shall defend, indemnify and hold harmless Licensee from and against any and all costs, damages, losses, liabilities, expenses, judgments, fines, settlements and any other amounts of any nature, including reasonable attorneys' fees arising from any improper payment made in violation of the FCPA, any applicable anti-corruption laws or this Section, directly or indirectly, by, on behalf of or with the knowledge of the Ligand, in relation to this Agreement.
7.5Disclaimer. EXCEPT AS PROVIDED IN THIS ARTICLE 7, NEITHER PARTY MAKES ANY REPRESENTATION OR WARRANTY (EXPRESS, IMPLIED, STATUTORY OR OTHERWISE) WITH RESPECT TO THE SUBJECT MATTER OF THIS AGREEMENT, AND EACH PARTY SPECIFICALLY DISCLAIMS ANY AND ALL IMPLIED WARRANTIES OR CONDITIONS OF MERCHANTABILITY AND FITNESS FOR A PARTICULAR PURPOSE, AND ALL WARRANTIES AND CONDITIONS OF THE VALIDITY OF THE LICENSED PATENTS OR NONINFRINGEMENT OF THIRD PARTY INTELLECTUAL PROPERTY RIGHTS. This SECTION 7.5 shall not be construed to limit either Party’s obligations under ARTICLE 8.
ARTICLE 8
INDEMNIFICATION; INSURANCE
(a)Indemnification by Licensee. Licensee shall indemnify, hold harmless, and defend Ligand, its Affiliates, and their respective equity holders, partners (general and/or limited), managers, directors, officers, employees and agents (“Ligand Indemnitees”) from and against any and all Third Party claims, suits, losses, liabilities, damages, costs, fees and expenses (including reasonable attorneys’ fees) (collectively, “Losses”) finally awarded to a Third Party by a court of competent jurisdiction or agreed to in a settlement approved by Licensee that result from any claim made or brought against a Ligand Indemnitee by or on behalf of such Third Party, and subject to Section 8.3, any direct out-of-pocket costs and expenses (including reasonable attorneys’ fees) (“Litigation Costs”) incurred by a Ligand Indemnitee while investigating or conducting the defense of such Third Party claim, in any such case, solely to the extent such claim is directly based on or directly arises out of (a) any material breach of, or inaccuracy in, any representation or warranty made by Licensee in this Agreement, or any breach or violation of any covenant or agreement of Licensee or any of its Affiliates or Sublicensees in or pursuant to this Agreement, (b) the negligence or willful misconduct by or of Licensee or its Affiliates, and their respective directors, officers, employees and agents, (c) the Development, manufacturing and/or
commercialization of a Product by Licensee or its Affiliates or Sublicensees (including product liability), and/or (d) Licensee’s contractual agreements with Third Parties during the Term. Furthermore, Licensee shall have no obligation to indemnify the Ligand Indemnitees to the extent that the Losses arise out of or result from, directly or indirectly, any material breach of, or inaccuracy in, any representation or warranty made by Ligand in this Agreement, or any breach or violation of any covenant or agreement of Ligand in or pursuant to this Agreement, or the negligence or willful misconduct by or of any of the Ligand Indemnitees, or the Development, manufacturing and/or commercialization of Lasofoxifene by Ligand at any time.
(b)Claimed Infringement of Third Party Rights. In the event that a Third Party at any time provides written notice of a claim to, or brings an action, suit or proceeding against, any Party, or any of such Party’s respective Affiliates or sublicensees, claiming infringement of its Patent rights or unauthorized use or misappropriation of its Know-How, based upon an assertion or claim arising out of the Development, manufacture or commercialization of a Product in the Territory (“Infringement Claim”), such Party shall promptly notify the other Party of the Infringement Claim or the commencement of such action, suit or proceeding, enclosing a copy of the Infringement Claim and all papers served. Each Party agrees to make available to the other Party its advice and counsel regarding the technical merits of any such claim [***] and to offer reasonable assistance to the other Party [***].
(c)Obligation to Defend. Licensee shall have the obligation to defend all Infringement Claims brought against either Party or any of its Affiliates or sublicensees arising out of the Development, manufacture or commercialization of a Product in the Field in the Territory.
(d)Procedure. Licensee shall have the sole and exclusive right to select counsel to defend any Infringement Claim brought via litigation or other formal dispute resolution procedure; provided [***]. Licensee shall keep Ligand informed, and shall from time to time consult with Ligand regarding the status of any such claims and shall provide Ligand with copies of all material documents filed in, and all material written communications relating to, any suit brought in connection with such claims. Ligand shall also have the right to participate and be represented in any such claim or related suit, at its own expense. Licensee shall not settle any Infringement Claims that would adversely impact any of the Licensed Patents (such as invalidation of or narrowing the scope of any claim of any of the Licensed Patents) or purport to impose any obligations on Ligand, without obtaining the prior written consent of Ligand or its Affiliate, as applicable, [***]. All litigation costs and expenses incurred by Licensee in connection with such Infringement Claim, and all damages payable by Licensee to the Third Party in respect of such Infringement Claims shall be borne by Licensee.
8.2Indemnification by Ligand. Ligand shall indemnify, hold harmless, and defend Licensee, its Affiliates and their respective equity holders, partners (general and/or limited), directors, managers, officers, employees and agents (“Licensee Indemnitees”) from and against any and all Losses finally awarded to a Third Party by a court of competent jurisdiction or agreed to in a settlement approved by Ligand that result from any claim made or bought against a Licensee Indemnitee by or on behalf of such Third Party, and subject to Section 8.3, any Litigation Costs incurred by a Licensee Indemnitee while investigating or conducting the defense of such Third Party claim, in any such case, solely to the extent such claim is directly based on or directly arises out of (a) the material breach by Ligand of any representation, warranty or covenant contained in this Agreement; (b) the commercialization of Lasofoxifene by Ligand, its Affiliates or its sublicensees outside the Field, and/or (c) Ligand’s contractual agreements with Third Parties; provided, however, that such indemnification right shall not apply to any Losses or litigation costs (i) to the extent directly attributable to the negligence, reckless misconduct, or intentional misconduct of a Licensee Indemnitee, (ii) for which Licensee is obligated to indemnify Ligand under Section 8.1(a), or (iii) resulting from any Third Party claim arising out of such Third Party’s contractual agreement(s) with a Licensee Indemnitee. Furthermore, Ligand shall have no obligation to indemnify the Licensee Indemnitees to the extent that the Losses arise out of or result from, directly or indirectly, any material breach of, or inaccuracy in, any representation or warranty made by Licensee in this Agreement, or any breach or violation of any covenant or agreement of Licensee in or pursuant to this Agreement, or the negligence or willful misconduct by or of any of the Licensee Indemnitees.
8.3Procedure. In the event of any such claim against any Licensee Indemnitee or Ligand Indemnitee (individually, an “Indemnitee”), the indemnified Party shall promptly notify the other Party in writing of the claim and the indemnifying Party shall manage and control, at its sole expense, the defense of the claim and its settlement; provided that the failure to so notify promptly shall not relieve the indemnifying Party of its obligations under this Article 8 except to the extent of the actual prejudice suffered by such Party as a result of such failure; and further provided that the indemnifying Party shall not have the right to assume the defense of such claim if such claim relates to an Infringement Claim. The Indemnitee shall cooperate with the indemnifying Party and may, at its option and expense, be represented in and participate in any such action or proceeding. The indemnifying Party shall not be liable for any settlements, litigation costs or expenses incurred by any Indemnitee without the indemnifying Party’s written authorization. [***].
(a)Licensee shall [***] to name and cause Ligand and the Ligand Indemnitees to be named as “additional insureds” on any commercial general liability and product liability insurance policies maintained by Licensee, its Affiliates and Sublicensees applicable to the Products.
(b)Licensee (or an Affiliate or Sublicensee, as applicable) shall, at its sole expense, procure and maintain commercial general liability insurance with reputable insurers in usual and customary amounts based on the stage of development for the Products. If Licensee elects to self-insure all or part of the limits described above, such self-insurance program must be acceptable to Ligand [***]. The maintenance of such insurance policies shall not in any way limit Licensee’s liability with respect to indemnification under this Agreement.
(c)Licensee (or an Affiliate or Sublicensee, as applicable) shall maintain such commercial general liability insurance beyond the termination of this Agreement during (a) the period that any Product is being commercially distributed or sold by or on behalf of Licensee, its Affiliates or a Sublicensee; and (b) a reasonable period after the period referred to in sub-clause (a) above, which in no event shall be [***].
ARTICLE 9
LIMITATION OF LIABILITY
9.1EXCEPT FOR ANY LIABILITY THAT IS THE CONSEQUENCE OF WILLFUL MISCONDUCT OF A PARTY, IN NO EVENT SHALL EITHER PARTY BE LIABLE TO THE OTHER PARTY FOR ANY SPECIAL, CONSEQUENTIAL, EXEMPLARY OR INCIDENTAL DAMAGES (INCLUDING LOST OR ANTICIPATED REVENUES OR PROFITS RELATING TO THE SAME), HOWEVER CAUSED AND ON ANY THEORY OF LIABILITY ARISING OUT OF THIS AGREEMENT, WHETHER SUCH CLAIM IS BASED ON CONTRACT, TORT (INCLUDING NEGLIGENCE) OR OTHERWISE, AND WHETHER OR NOT SUCH PARTY HAS BEEN ADVISED OF THE POSSIBILITY OF SUCH DAMAGE. THESE LIMITATIONS SHALL APPLY NOTWITHSTANDING ANY FAILURE OF ESSENTIAL PURPOSE OF ANY LIMITED REMEDY PROVIDED HEREIN. THIS ARTICLE 9 SHALL NOT BE CONSTRUED TO LIMIT EITHER PARTY’S OBLIGATIONS UNDER ARTICLE 8.
TERM AND TERMINATION
10.1Term. This Agreement shall commence on the Effective Date and on a country-by-country basis, shall be in full force and effect until terminated by either Party pursuant to this Article 10, for so long as Licensee (or an Affiliate or Sublicensee) is developing, making, using or selling a Product in the Territory (the “Term”).
(a)Termination by Licensee for Clinical Failure. Licensee may terminate this Agreement in its entirety or on a Product-by- Product or country-by-country basis by providing written notice to Ligand if, prior to Regulatory Approval, Licensee determines [***] that there are material concerns regarding (i) the safety of any Product or (ii) the failure to substantially achieve any of the primary efficacy endpoints of any clinical trial involving any Product, provided that [***].
(b)For Material Breach. If either Party shall at any time breach any material term, condition or agreement herein, and shall fail to have cured any such default or breach within [***] (the “Cure Period”), or if the default or breach is not capable of being cured during the Cure Period, failed to have initiated and actively pursued remedy of such default or breach during the Cure Period, in each case after receipt of written notice thereof by the other Party, then the other Party may, at its option, terminate this Agreement and revoke any rights and licenses herein; provided, [***]. Any termination of this Agreement under this Section 10.2 shall not, however, prejudice the right of the Party who terminates this Agreement to recover any payment due at the time of such cancellation. [***].
(c)For Bankruptcy. Ligand may terminate this Agreement upon the occurrence of one or more of the following: (i) immediately upon written notice to Licensee in the event Licensee is insolvent or [***]; or (ii) [***].
(d)For Challenge. If Licensee, its Affiliates or a Sublicensee institutes a Challenge, Ligand may, at its option, terminate this Agreement and revoke any rights and licenses herein with [***] prior written notice.
10.3Effect of Termination.
(a)Rights and Obligations Upon Termination. As of the effective date of a termination of this Agreement for any reason: (i) this Agreement and all rights and licenses granted to Licensee under Section 2.1 shall terminate and all rights in the Licensed Technology shall revert to Ligand; (ii) Licensee shall return to Ligand the Licensed Know-How; (iii) to transfer to Ligand copies of all Development data generated by or on behalf of Licensee in connection with a Product (including reports of clinical studies and all other documentation containing or embodying any preclinical, clinical and manufacturing data); (iv) transfer ownership to Ligand of all Regulatory Approval applications and Regulatory Approvals, including any IND and/or NDA, for a Product in the Territory, not then owned by, and otherwise transferred to, Ligand; (v) transfer (1) ownership to Ligand of any (a) [***] and (b) trademarks owned by Licensee and/or its Affiliates, but in each case (a)-(b) solely if utilized exclusively in connection with a Product and not in connection with Licensee’s and/or its Affiliates’ other products or services, (vi) [***] notify all applicable Regulatory Authorities of such transfer; (vii) [***] execute and deliver any and all documents necessary to effectuate such transfer and otherwise reasonably assist Ligand in effectuating such transfer; and (viii) each Party shall return to the other Party and cease using all Confidential Information of the other; provided, however, each Party may retain one (1) copy of such Confidential Information for archival purposes.
(b)Accrued Rights. Termination of this Agreement for any reason will be without prejudice to any rights that will have accrued to the benefit of a Party prior to such termination. Such termination will not relieve a Party from accrued payment obligations or from obligations which are expressly indicated to survive termination of this Agreement.
(c)Survival. All rights and obligations of the Parties which by intent or meaning have validity beyond or by their nature apply or are to be performed or exercised after the termination of this Agreement shall survive the termination of this Agreement for the period so specified, if any, or for perpetuity.
ARTICLE 11
GENERAL PROVISIONS
11.1Entire Agreement. The Parties acknowledge that this Agreement, together with the exhibits and schedules attached hereto, sets forth the entire agreement and understanding of the Parties as to the subject matter hereof, and supersedes all prior and contemporaneous discussions, agreements and writings in respect hereto.
11.2Modification; Waiver. No waiver, modification, amendment or alteration of any provision of this Agreement will be valid or effective unless made in writing and signed by each of the Parties. The failure of a Party to enforce any rights or provisions of the Agreement shall not be construed to be a waiver of such rights or provisions, or a waiver by such Party to thereafter enforce such rights or provisions or any other rights or provisions hereunder.
11.3Further Assurances. Each Party agrees to execute, acknowledge, and deliver such further instruments and to do all such other acts as may be necessary or appropriate in order to carry out the express provisions of this Agreement.
11.4Force Majeure. Neither Party shall be held responsible for any delay or failure in performance hereunder caused by strikes, embargoes, unexpected government requirements, civil or military authorities, acts of God, earthquake, or by the public enemy or other causes reasonably beyond such Party’s control and without such Party’s fault or negligence; provided that the affected Party notifies the unaffected Party [***], and resumes performance hereunder [***] following cessation of such force majeure event.
11.5Assignments. Neither this Agreement nor any interest hereunder may be assigned, nor any other obligation delegated, by a Party without the prior written consent of the other Party. Notwithstanding the foregoing, (a) [***]; (b) either Party shall have the right to assign this Agreement without consent of the other Party to an Affiliate of the assigning Party or to any successor in interest to the assigning Party by operation of law, merger, consolidation, or other business reorganization or the sale of all or substantially all of its assets relating to the subject matter of this Agreement in a manner such that the assigning Party will remain liable and responsible for the performance and observance of all of its duties and obligations hereunder; and (c) and Licensee shall have the right to assign this Agreement to Sermonix without the consent of Ligand in the event that the Sermonix-Athira Agreement is terminated for any reason, provided that [***]. This Agreement shall be binding upon successors and permitted assigns of the Parties. Any assignment not in accordance with this Section 11.5 will be null and void.
11.6Performance by Affiliates. The Parties recognize that each may perform some or all of its obligations under this Agreement through its Affiliates or may exercise some or all of its rights under this Agreement through its Affiliates; provided, however, that each Party shall remain
responsible and be the guarantor of the performance by its Affiliates and shall cause its Affiliates to comply with the provisions of this Agreement in connection with such performance. In particular and without limitation, all Affiliates of a Party that receive Confidential Information of the other Party pursuant to this Agreement shall be governed and bound by all obligations set forth in Article 6. Each Party will prohibit all of its Affiliates from taking any action that such Party is prohibited from taking under this Agreement as if such Affiliates were parties to this Agreement.
11.7Relationship of the Parties. The Parties shall perform their obligations under this Agreement as independent contractors and nothing in this Agreement is intended or will be deemed to constitute a partnership, agency or employer-employee relationship between the Parties. Neither Party will have any right, power or authority to assume, create, or incur any expense, liability, or obligation, express or implied, on behalf of the other.
11.8No Use of Names. Except as otherwise required under applicable Law, or as otherwise permitted under Section 6.4, neither Party will use the name of the other Party in its advertising, press releases or promotional materials without the prior written consent of such other Party.
11.9Notices. Any notice, request, delivery, approval or consent required or permitted to be given under this Agreement will be in writing and will be deemed to have been sufficiently given if delivered in person (in which case, it will be effective [***]), transmitted by facsimile, if facsimile number is provided below (receipt verified; in which case, it will be effective [***]) or by express courier service (signature required; in which case, it will be effective [***]), to the Party to which it is directed at its address or facsimile number shown below or such other address or facsimile number as such Party will have last given by notice to the other Party.
If to Ligand: [***]
[***]
[***]
[***]
Attention: [***]
Email: [***]
With a copy to (which shall not constitute notice hereunder):
[***]
[***]
[***]
Attn: [***]
Email: [***]
If to Licensee: [***]
[***]
[***]
[***]
Attention: [***]
Email: [***]
With a copy to (which shall not constitute notice hereunder):
[***]
[***]
[***]
Attention: [***]
Email: [***]
If to Sermonix (pursuant
to Section 10.2(b)): [***]
[***]
[***]
[***]
Attention: [***]
Email: [***]
With a copy to (which shall not constitute notice hereunder):
[***]
[***]
[***]
Attn: [***]
Email: [***]
11.10Governing Law. The rights and obligations of the Parties under this Agreement shall be governed, and shall be interpreted, construed, and enforced, in all respects by the Law of the State of California, without giving effect to any conflict of Law rule that would result in the application of the Law of any jurisdiction other than the internal Law of the State of California to the rights and duties of the Parties.
11.11Dispute Resolution. With respect to any disputes between the Parties concerning this Agreement, the dispute shall be submitted to escalating levels of Licensee and Ligand senior management for review. If the dispute cannot be resolved despite such escalation, then the matter may be referred by either Party to the Executive Officers to be resolved by negotiation [***]. Such resolution, if any, by the Executive Officers shall be final and binding on the Parties. If the Executive Officers are unable to resolve such dispute within such [***] period, each Party will be free to pursue all rights available to it under law or equity, provided that it has complied with this Section 11.11. Notwithstanding the foregoing, either Party may seek emergency or temporary injunctive relief in any court of competent jurisdiction.
11.12Headings. The article, section and subsection headings contained herein are for the purposes of convenience only and are not intended to define or limit the contents of the articles, sections or subsections to which such headings apply.
11.13Severability. When possible, each provision of this Agreement will be interpreted in such manner as to be effective and valid under Law, but, if any provision of this Agreement is held to be prohibited by or invalid under Law, such provision will be ineffective but only to the extent of such prohibition or invalidity, without invalidating the remainder of such provision or of this Agreement. The Parties will make a good faith effort to replace the invalid or unenforceable provision with a valid one which in its economic effect is most consistent with the invalid or unenforceable provision.
11.14Counterparts. This Agreement may be executed in counterparts (including by facsimile or electronic signature), each of which shall be deemed an original and all of which together shall constitute one instrument.
[Signature Page Follows]
IN WITNESS WHEREOF, the Parties have executed this Agreement in duplicate originals by their duly authorized representatives as of the Effective Date.
LIGAND PHARMACEUTICALS ATHIRA PHARMA, INC.
INCORPORATED
(“Ligand”) (“Licensee”)
By: By:
Name: Name:
Title: Title:
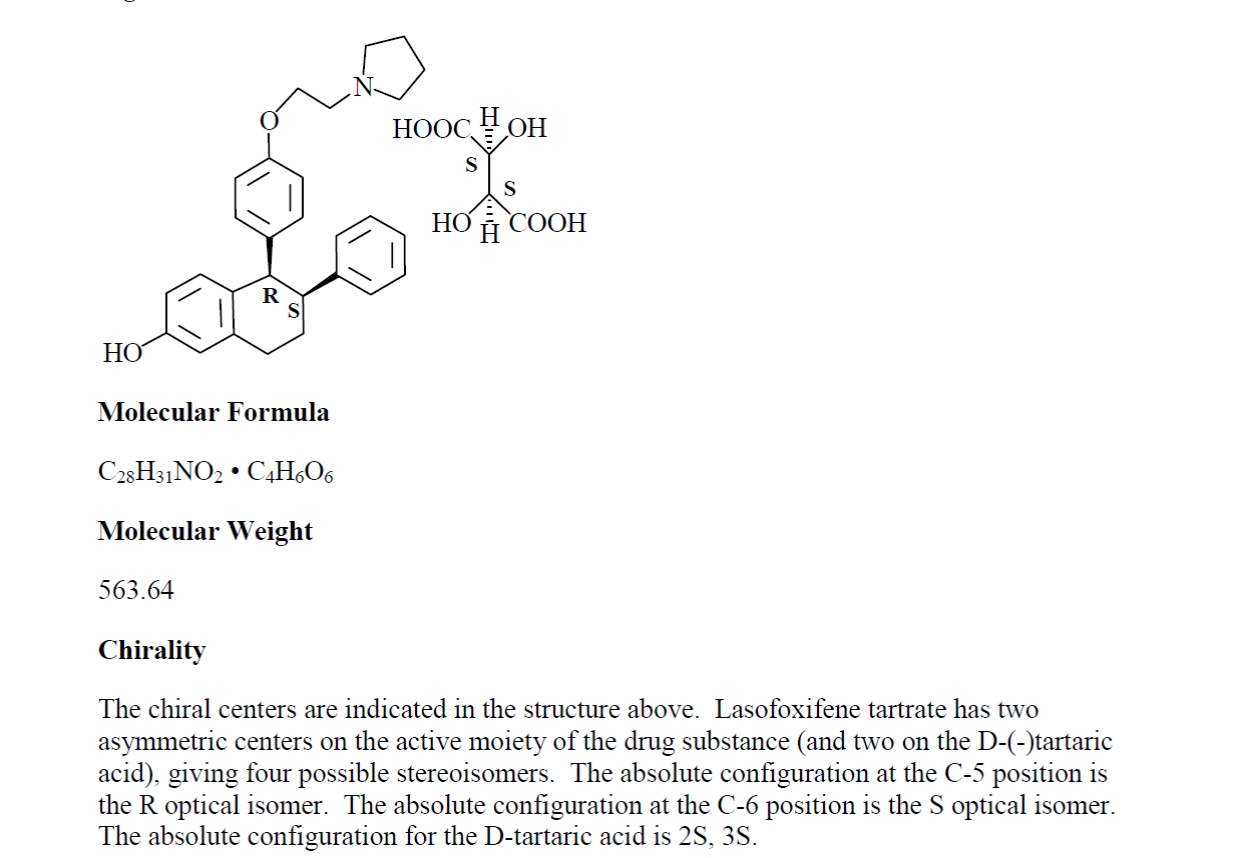
EXHIBIT B
Lasofoxifene
Chemical Structure of Lasofoxifene Tartrate