

Investor Presentation February 26, 2026 Indivior, Powering Recovery, Renewing Hope. .3

IMPORTANT CAUTIONARY STATEMENT REGARDING FORWARD-LOOKING STATEMENTS INVESTOR PRESENTATION | February 2026 This presentation contains certain statements that are forward-looking. Forward-looking statements include, among other things, express and implied statements regarding: the Company’s financial guidance for 2026, including total net revenue, SUBLOCADE® net revenue, non-GAAP gross margin, non-GAAP operating expenses, adjusted EBITDA, and cash flow from operations; expected acceleration of SUBLOCADE U.S. dispense unit and net revenue growth in 2026; expected future acceleration in the growth of adjusted EBITDA and cash flow; planned initiatives to accelerate SUBLOCADE growth; our expectation that we can grow and accelerate SUBLOCADE net revenue, generate immediate accretion from profitability and cash flow growth exceeding revenue growth, and leverage strengthened financial profile to acquire next growth drivers; expectations of increased LAI usage; our intention to invest in SUBLOCADE at sustained levels; expected future operating expense savings; potential future patents that might be awarded; our expectation that our financial profile with strengthen and that this will enable us to acquire our next growth drivers; potential share repurchases; potential deployment of capital to create long-term value for shareholders; our product development pipeline and potential future products, the timing of clinical trials, expectations regarding regulatory approval of such product candidates, the timing of such approvals, and the timing of commercial launch of such products or product candidates, and eventual annual revenues of such future products; and other statements containing the words "believe," "anticipate," "plan," "expect," "intend," "estimate," "forecast," “strategy,” “target,” “guidance,” “outlook,” “potential,” "project," "priority," "may," "will," "should," "would," "could," "can," the negatives thereof, and variations thereon and similar expressions. By their nature, forward-looking statements involve risks and uncertainties as they relate to events or circumstances that may or may not occur in the future. Actual results may differ materially from those expressed or implied in these forward-looking statements due to a number of factors, including: lower than expected future sales of our products; greater than expected impacts from competition; unanticipated costs including the effects of potential tariffs and potential retaliatory tariffs; whether we are able to identify efficiencies and fund additional investments that we expect to generate increased revenue, and the timing of such actions; market acceptance of long-acting injectables; cash available for share repurchases in the future, and the market price of our common stock in the future; our ability to identify accretive investment opportunities, to negotiate with third parties to acquire such assets, and our ability to efficiently manage such assets and execute upon opportunities; and the results of pending and future clinical trials, and the decisions of relevant regulators. For additional information about some of the risks and important factors that could affect our future results and financial condition, see "Risk Factors" in our Annual Report on Form 10-K filed March 3, 2025, in our Quarterly Reports on Forms 10-Q filed May 1, 2025, July 31, 2025, and October 30, 2025, our other filings with the U.S. Securities and Exchange Commission. Forward-looking statements speak only as of the date that they are made and should be regarded solely as our current plans, estimates and beliefs. Except as required by law, we do not undertake and specifically decline any obligation to update, republish or revise forward-looking statements to reflect future events or circumstances or to reflect the occurrences of unanticipated events.
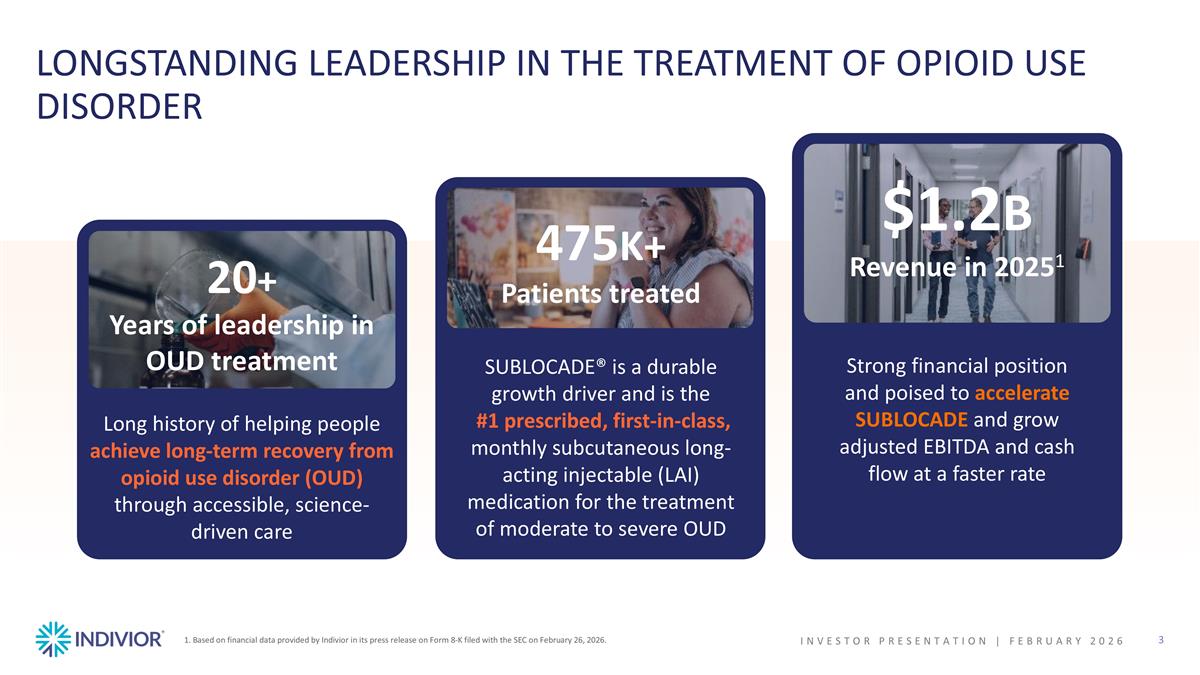
LONGSTANDING LEADERSHIP IN THE TREATMENT OF OPIOID USE DISORDER 20+ Years of leadership in OUD treatment Long history of helping people achieve long-term recovery from opioid use disorder (OUD) through accessible, science-driven care 475K+ Patients treated SUBLOCADE® is a durable growth driver and is the #1 prescribed, first-in-class, monthly subcutaneous long-acting injectable (LAI) medication for the treatment of moderate to severe OUD $1.2B Revenue in 20251 Strong financial position and poised to accelerate SUBLOCADE and grow adjusted EBITDA and cash flow at a faster rate INVESTOR PRESENTATION | February 2026 1. Based on financial data provided by Indivior in its press release on Form 8-K filed with the SEC on February 26, 2026.

EXECUTING THE INDIVIOR ACTION AGENDA AND ENTERING 2026 AS A FOCUSED, SIMPLIFIED AND STRONGER INDIVIOR New operating model in place to drive significant bottom-line growth and cash flow generation Improved financial profile and strength enables capital allocation optionality INVESTOR PRESENTATION | February 2026 Sharpened focus on highest growth opportunity – U.S. SUBLOCADE

THE INDIVIOR ACTION AGENDA I II III Grow U.S. SUBLOCADE net revenue Simplify the organization and establish “go-forward” operating model Determine actions and investments necessary to expand LAI penetration in U.S. BMAT category to accelerate U.S. SUBLOCADE net revenue Accelerate U.S. SUBLOCADE dispense unit and net revenue throughout 2026 Immediately accelerate adjusted EBITDA and cash flow at a faster rate Phase II – Accelerate (Began Jan. 2026) Leverage strengthened financial profile to acquire next growth drivers Phase III – Breakout (H2’26 – Beyond) Phase I – Generate Momentum (Completed) LAI: long-acting injectable. BMAT: buprenorphine medication assisted treatment. INVESTOR PRESENTATION | February 2026
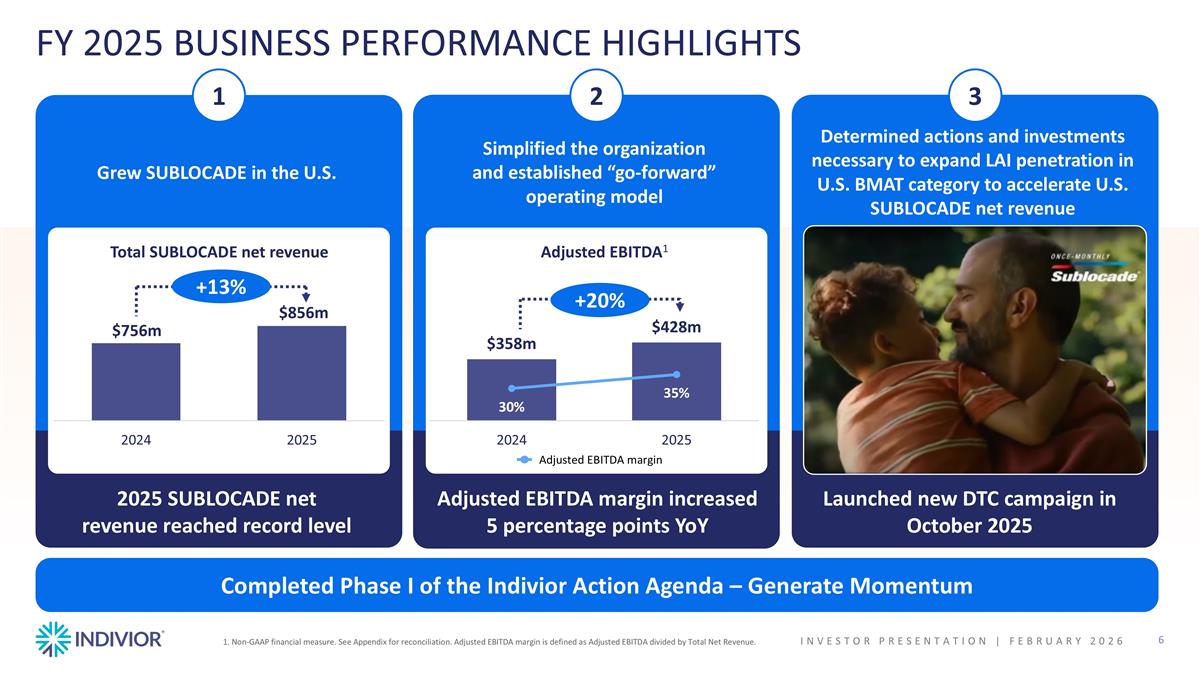
FY 2025 BUSINESS PERFORMANCE HIGHLIGHTS Grew SUBLOCADE in the U.S. Simplified the organization and established “go-forward” operating model Determined actions and investments necessary to expand LAI penetration in U.S. BMAT category to accelerate U.S. SUBLOCADE net revenue 2025 SUBLOCADE net revenue reached record level Adjusted EBITDA margin increased 5 percentage points YoY Launched new DTC campaign in October 2025 1 2 3 +13% Adjusted EBITDA1 1. Non-GAAP financial measure. See Appendix for reconciliation. Adjusted EBITDA margin is defined as Adjusted EBITDA divided by Total Net Revenue. Total SUBLOCADE net revenue +20% Adjusted EBITDA margin Completed Phase I of the Indivior Action Agenda – Generate Momentum INVESTOR PRESENTATION | February 2026

ENTERED PHASE II – ACCELERATE – ON JANUARY 1, 2026 Accelerate U.S. SUBLOCADE +8% +30% Non-GAAP operating expenses will not exceed $450m; ~$300m in cash flow from operations expected in 20262 Total SUBLOCADE Net Revenue Adjusted EBITDA3 1. Based on financial guidance ranges provided by Indivior in its press release on Form 8-K filed with the SEC on February 26, 2026. 2. Excludes cash flows from investing and financing activities. 3. Adjusted EBITDA is a non-GAAP financial measure. See non-GAAP Financial Measures in the Appendix for reconciliation to the most comparable GAAP measures. For non-GAAP guidance items, the Company has relied upon the exception in item 10(e)(1)(i)(B) of Regulation S-K to exclude such reconciliations, as the reconciliations of these non-GAAP guidance metrics to their corresponding GAAP equivalents are not available without unreasonable effort; See Appendix for details. Expect to accelerate SUBLOCADE dispense unit growth from 7% in 2025 to the mid-teens in 2026 Immediately Accelerate Adjusted EBITDA and Cash Generation at a Faster Rate than Net Revenue INVESTOR PRESENTATION | February 2026

2026 FINANCIAL GUIDANCE REFLECTS SIGNIFICANT MARGIN EXPANSION AS PART OF PHASE II – ACCELERATE Guidance Range1 YoY Change2 Total Net Revenue $1,125m - $1,195m -6% SUBLOCADE Net Revenue $905m - $945m +8% Non-GAAP Operating Expenses3 $430m - $450m -29% Adjusted EBITDA3 $535m - $575m +30% 1. As of February 26, 2026, before exceptional items and assuming no material change in key FX rates vs. FY 2025 average rates. Financial data provided by Indivior in its press release on Form 8-K filed with the SEC on February 26, 2026. 2. Represents the midpoint of 2026 guidance ranges compared to 2025 actuals. 3. For non-GAAP guidance items, the Company has relied upon the exception in item 10(e)(1)(i)(B) of Regulation S-K to exclude such reconciliations, as the reconciliations of these non-GAAP guidance metrics to their corresponding GAAP equivalents are not available without unreasonable effort; See slides 20 to 27 for details. INVESTOR PRESENTATION | February 2026

SUBLOCADE®
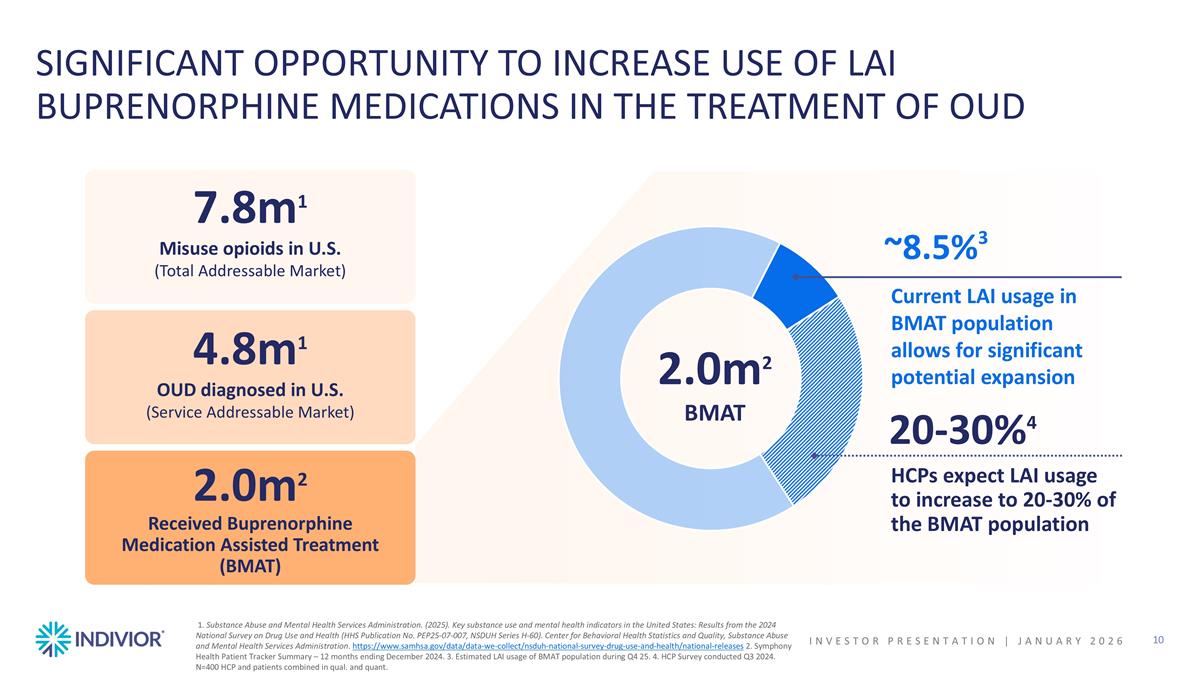
1. Substance Abuse and Mental Health Services Administration. (2025). Key substance use and mental health indicators in the United States: Results from the 2024 National Survey on Drug Use and Health (HHS Publication No. PEP25-07-007, NSDUH Series H-60). Center for Behavioral Health Statistics and Quality, Substance Abuse and Mental Health Services Administration. https://www.samhsa.gov/data/data-we-collect/nsduh-national-survey-drug-use-and-health/national-releases 2. Symphony Health Patient Tracker Summary – 12 months ending December 2024. 3. Estimated LAI usage of BMAT population during Q4 25. 4. HCP Survey conducted Q3 2024. N=400 HCP and patients combined in qual. and quant. SIGNIFICANT OPPORTUNITY TO INCREASE USE OF LAI BUPRENORPHINE MEDICATIONS IN THE TREATMENT OF OUD INVESTOR PRESENTATION | JANUARY 2026 7.8m1 Misuse opioids in U.S. (Total Addressable Market) 4.8m1 OUD diagnosed in U.S. (Service Addressable Market) 2.0m2 Received Buprenorphine Medication Assisted Treatment (BMAT) ~8.5%3 Current LAI usage in BMAT population allows for significant potential expansion HCPs expect LAI usage to increase to 20-30% of the BMAT population 20-30%4 2.0m2 BMAT
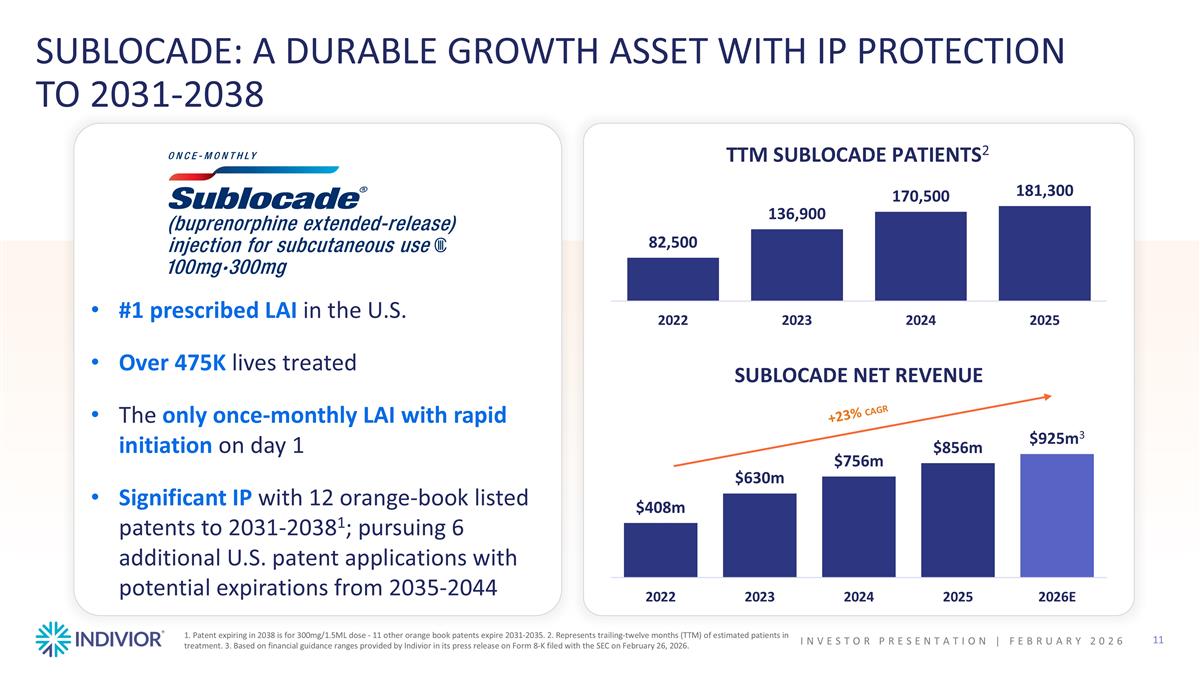
#1 prescribed LAI in the U.S. Over 475K lives treated The only once-monthly LAI with rapid initiation on day 1 Significant IP with 12 orange-book listed patents to 2031-20381; pursuing 6 additional U.S. patent applications with potential expirations from 2035-2044 SUBLOCADE: A DURABLE GROWTH ASSET WITH IP PROTECTION TO 2031-2038 SUBLOCADE NET REVENUE +23% CAGR 1. Patent expiring in 2038 is for 300mg/1.5ML dose - 11 other orange book patents expire 2031-2035. 2. Represents trailing-twelve months (TTM) of estimated patients in treatment. 3. Based on financial guidance ranges provided by Indivior in its press release on Form 8-K filed with the SEC on February 26, 2026. TTM SUBLOCADE PATIENTS2 INVESTOR PRESENTATION | February 2026
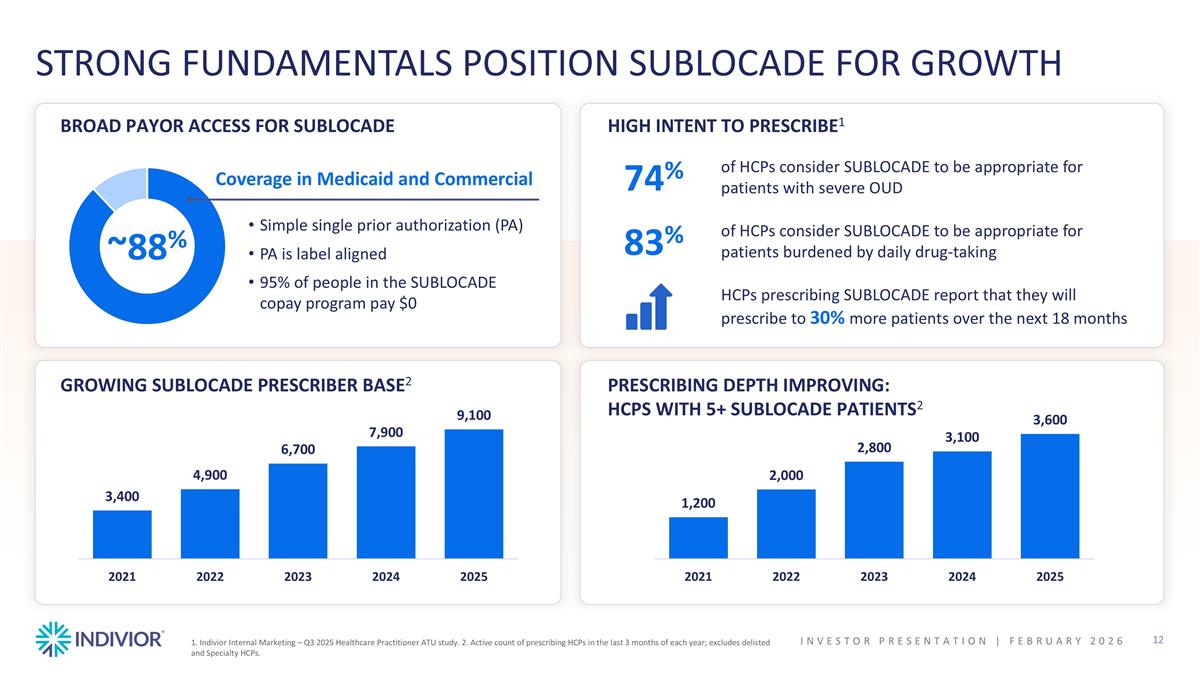
1. Indivior Internal Marketing – Q3 2025 Healthcare Practitioner ATU study. 2. Active count of prescribing HCPs in the last 3 months of each year; excludes delisted and Specialty HCPs. STRONG FUNDAMENTALS POSITION SUBLOCADE FOR GROWTH GROWING SUBLOCADE PRESCRIBER BASE2 PRESCRIBING DEPTH IMPROVING: HCPS WITH 5+ SUBLOCADE PATIENTS2 BROAD PAYOR ACCESS FOR SUBLOCADE HIGH INTENT TO PRESCRIBE1 ~88% Coverage in Medicaid and Commercial Simple single prior authorization (PA) PA is label aligned 95% of people in the SUBLOCADE copay program pay $0 74% 83% of HCPs consider SUBLOCADE to be appropriate for patients with severe OUD of HCPs consider SUBLOCADE to be appropriate for patients burdened by daily drug-taking HCPs prescribing SUBLOCADE report that they will prescribe to 30% more patients over the next 18 months INVESTOR PRESENTATION | February 2026
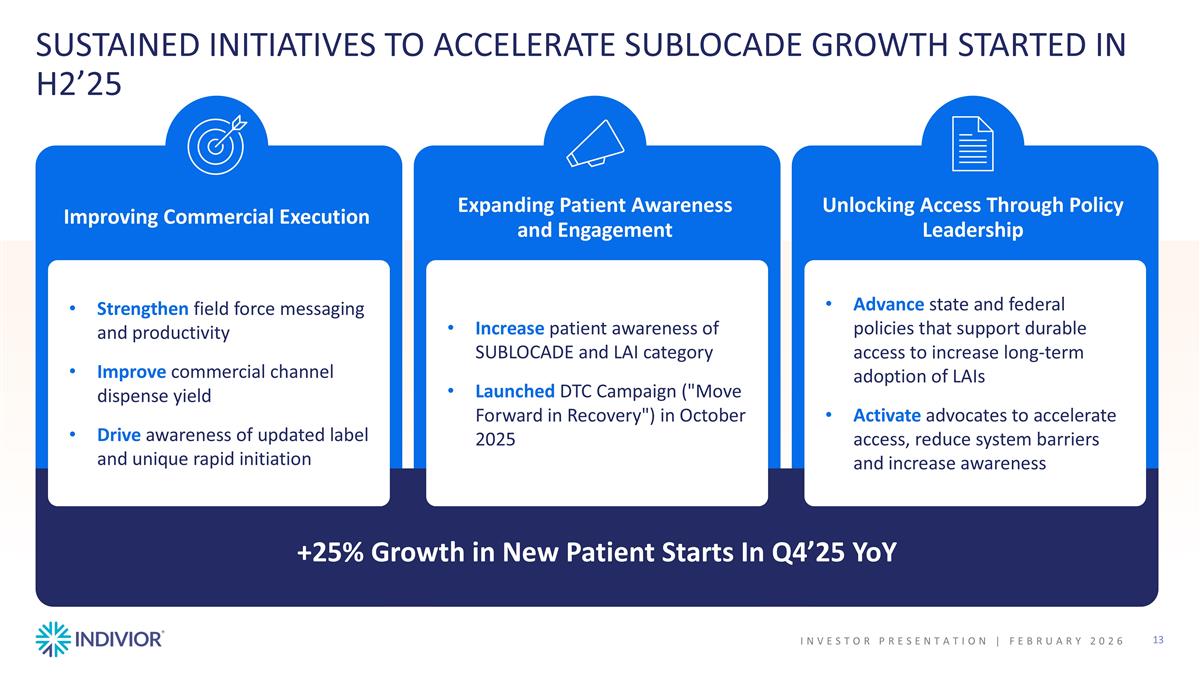
SUSTAINED INITIATIVES TO ACCELERATE SUBLOCADE GROWTH STARTED IN H2’25 Unlocking Access Through Policy Leadership Advance state and federal policies that support durable access to increase long-term adoption of LAIs Activate advocates to accelerate access, reduce system barriers and increase awareness +25% Growth in New Patient Starts In Q4’25 YoY Expanding Patient Awareness and Engagement Increase patient awareness of SUBLOCADE and LAI category Launched DTC Campaign ("Move Forward in Recovery") in October 2025 Improving Commercial Execution Strengthen field force messaging and productivity Improve commercial channel dispense yield Drive awareness of updated label and unique rapid initiation INVESTOR PRESENTATION | February 2026

Source: Indivior internal analytics and patient ATU research (Q4’25). FASTP: Find a SUBLOCADE Treatment Provider. CRM: Customer Relationship Management. SUBLOCADE ON TRACK TO ACCELERATE IN 2026 DRIVEN BY PATIENT EDUCATION & ACTIVATION EFFORTS INVESTOR PRESENTATION | February 2026 Launched new DTC campaign, Move Forward in Recovery, on October 1, 2025 Positive early indicators of success: Patient prompted awareness increased to 44% in Q4’25 vs. 15% in Q1’25 ~60% increase in branded SUBLOCADE online search volume in Q4’25 vs. Q3’25 ~70% growth in FASTP physician locator usage in Q4’25 vs. Q3’25 CRM enrollments surged to ~1,400 people/month in Q4’25 from ~60 people/month pre-campaign

Pipeline
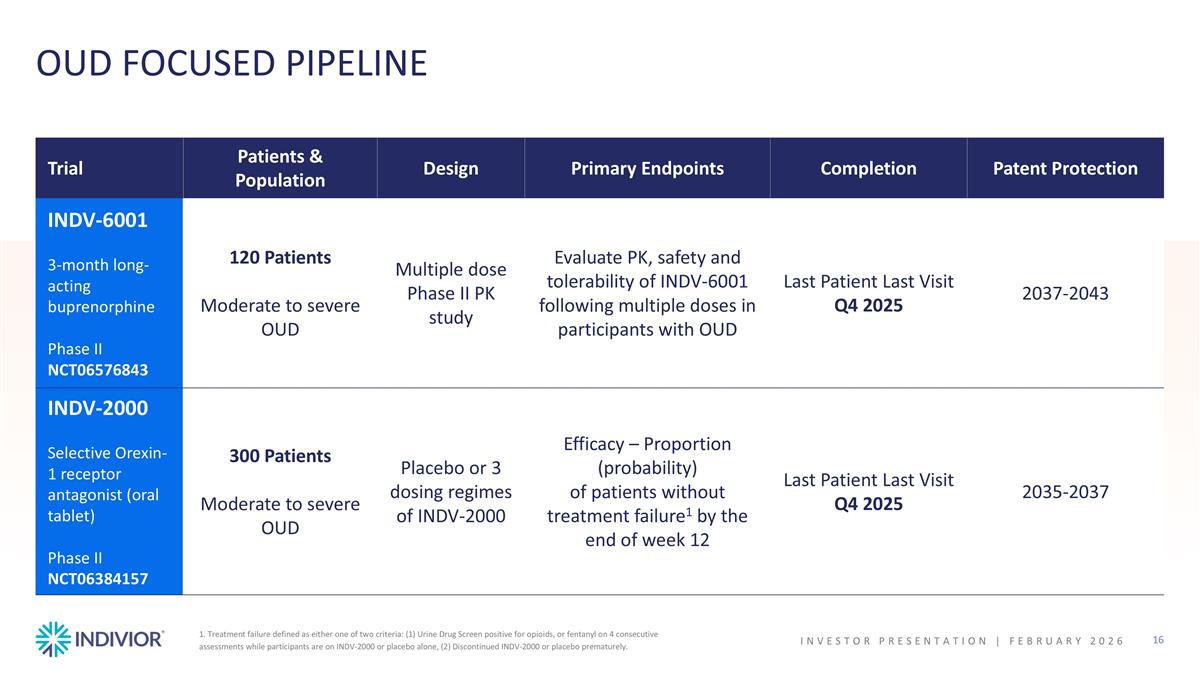
1. Treatment failure defined as either one of two criteria: (1) Urine Drug Screen positive for opioids, or fentanyl on 4 consecutive assessments while participants are on INDV-2000 or placebo alone, (2) Discontinued INDV-2000 or placebo prematurely. Trial Patients & Population Design Primary Endpoints Completion Patent Protection INDV-6001 3-month long-acting buprenorphine Phase II NCT06576843 120 Patients Moderate to severe OUD Multiple dose Phase II PK study Evaluate PK, safety and tolerability of INDV-6001 following multiple doses in participants with OUD Last Patient Last Visit Q4 2025 2037-2043 INDV-2000 Selective Orexin-1 receptor antagonist (oral tablet) Phase II NCT06384157 300 Patients Moderate to severe OUD Placebo or 3 dosing regimes of INDV-2000 Efficacy – Proportion (probability) of patients without treatment failure1 by the end of week 12 Last Patient Last Visit Q4 2025 2035-2037 OUD FOCUSED PIPELINE INVESTOR PRESENTATION | February 2026

Financials
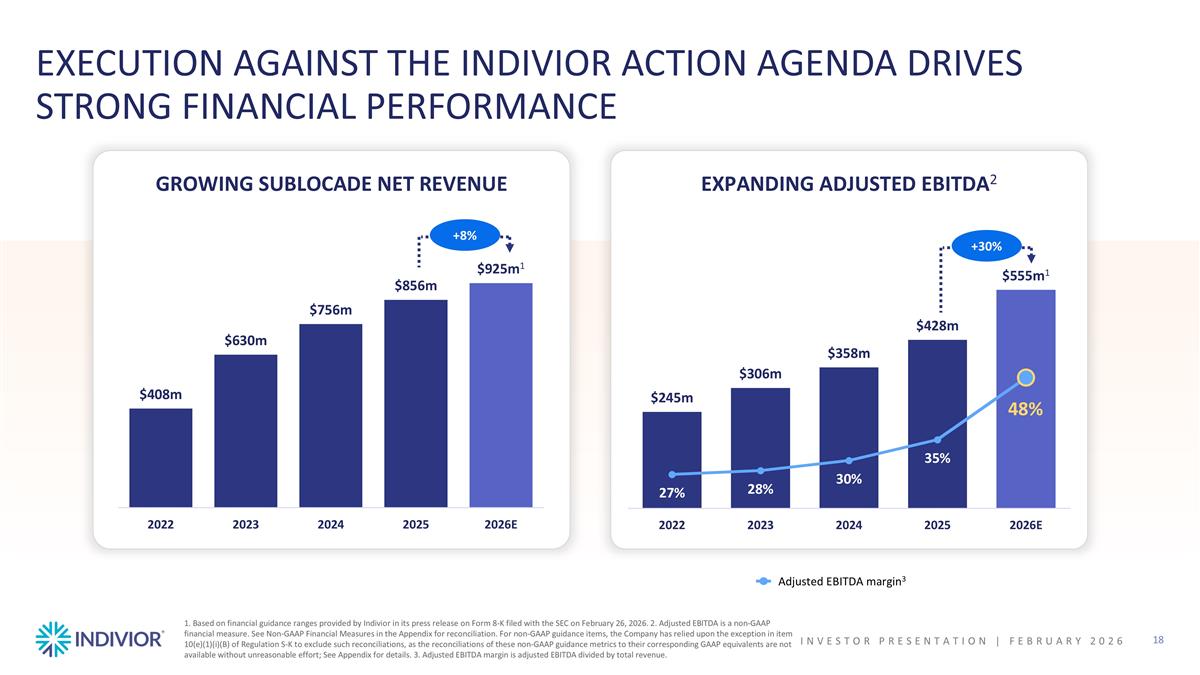
Adjusted EBITDA margin3 EXECUTION AGAINST THE INDIVIOR ACTION AGENDA DRIVES STRONG FINANCIAL PERFORMANCE GROWING SUBLOCADE NET REVENUE EXPANDING ADJUSTED EBITDA2 +8% +30% 1. Based on financial guidance ranges provided by Indivior in its press release on Form 8-K filed with the SEC on February 26, 2026. 2. Adjusted EBITDA is a non-GAAP financial measure. See Non-GAAP Financial Measures in the Appendix for reconciliation. For non-GAAP guidance items, the Company has relied upon the exception in item 10(e)(1)(i)(B) of Regulation S-K to exclude such reconciliations, as the reconciliations of these non-GAAP guidance metrics to their corresponding GAAP equivalents are not available without unreasonable effort; See Appendix for details. 3. Adjusted EBITDA margin is adjusted EBITDA divided by total revenue. INVESTOR PRESENTATION | February 2026
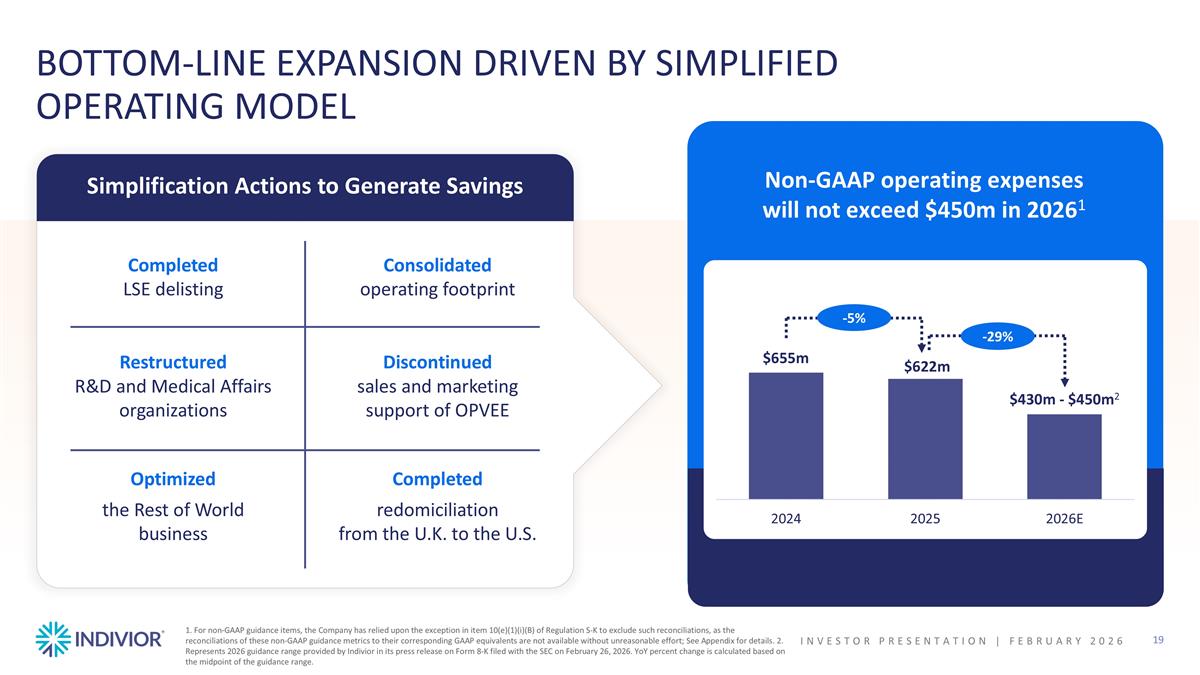
1. For non-GAAP guidance items, the Company has relied upon the exception in item 10(e)(1)(i)(B) of Regulation S-K to exclude such reconciliations, as the reconciliations of these non-GAAP guidance metrics to their corresponding GAAP equivalents are not available without unreasonable effort; See Appendix for details. 2. Represents 2026 guidance range provided by Indivior in its press release on Form 8-K filed with the SEC on February 26, 2026. YoY percent change is calculated based on the midpoint of the guidance range. Completed LSE delisting Restructured R&D and Medical Affairs organizations Optimized the Rest of World business BOTTOM-LINE EXPANSION DRIVEN BY SIMPLIFIED OPERATING MODEL Simplification Actions to Generate Savings Consolidated operating footprint Discontinued sales and marketing support of OPVEE Completed redomiciliation from the U.K. to the U.S. Non-GAAP operating expenses will not exceed $450m in 20261 -5% -29% INVESTOR PRESENTATION | February 2026

2026 CAPITAL DEPLOYMENT STRATEGY $222m in cash and investments as of 12/31/25 ~$300m in cash flow from operations expected in 20261 $295m Payment to DOJ on 11/20/25 eliminated legacy matter 0.7x leverage ratio2 DEBT MANAGEMENT $350m term loan maturing in 2030 with $50m revolving credit facility SHARE REPURCHASES Authorized ~$400m share repurchase program with a term of up to 18 months in February 2026 BUSINESS DEVELOPMENT Earning our way to Phase III of Indivior Action Agenda – Breakout – to acquire next commercial stage growth drivers 1. Excludes cash flows from investing and financing activities. 2. Defined as Net Debt as of December 31, 2025, of $283m, divided by 2025 Adjusted EBITDA of $428m; See Non-GAAP Financial Measures in the Appendix for reconciliation on slide 34. INVESTOR PRESENTATION | February 2026

Summary

DELIVERING ON STRATEGIC PRIORITIES TO ACCELERATE IN 2026 Maximize the potential of the business Create long-term value for shareholders Make a positive difference in the lives of people living with OUD INVESTOR PRESENTATION | February 2026

Appendix
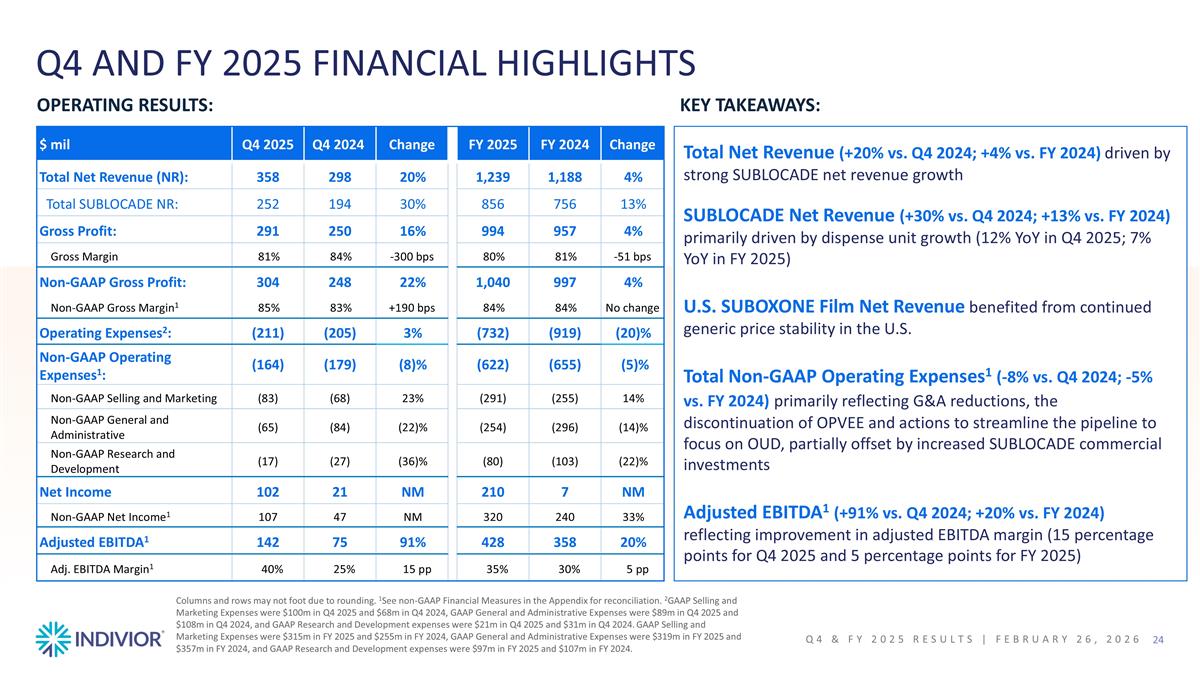
Q4 AND FY 2025 FINANCIAL HIGHLIGHTS OPERATING RESULTS: KEY TAKEAWAYS: Total Net Revenue (+20% vs. Q4 2024; +4% vs. FY 2024) driven by strong SUBLOCADE net revenue growth SUBLOCADE Net Revenue (+30% vs. Q4 2024; +13% vs. FY 2024) primarily driven by dispense unit growth (12% YoY in Q4 2025; 7% YoY in FY 2025) U.S. SUBOXONE Film Net Revenue benefited from continued generic price stability in the U.S. Total Non-GAAP Operating Expenses1 (-8% vs. Q4 2024; -5% vs. FY 2024) primarily reflecting G&A reductions, the discontinuation of OPVEE and actions to streamline the pipeline to focus on OUD, partially offset by increased SUBLOCADE commercial investments Adjusted EBITDA1 (+91% vs. Q4 2024; +20% vs. FY 2024) reflecting improvement in adjusted EBITDA margin (15 percentage points for Q4 2025 and 5 percentage points for FY 2025) Columns and rows may not foot due to rounding. 1See non-GAAP Financial Measures in the Appendix for reconciliation. 2GAAP Selling and Marketing Expenses were $100m in Q4 2025 and $68m in Q4 2024, GAAP General and Administrative Expenses were $89m in Q4 2025 and $108m in Q4 2024, and GAAP Research and Development expenses were $21m in Q4 2025 and $31m in Q4 2024. GAAP Selling and Marketing Expenses were $315m in FY 2025 and $255m in FY 2024, GAAP General and Administrative Expenses were $319m in FY 2025 and $357m in FY 2024, and GAAP Research and Development expenses were $97m in FY 2025 and $107m in FY 2024. Q4 & FY 2025 Results | February 26, 2026 $ mil Q4 2025 Q4 2024 Change FY 2025 FY 2024 Change Total Net Revenue (NR): 358 298 20% 1,239 1,188 4% Total SUBLOCADE NR: 252 194 30% 856 756 13% Gross Profit: 291 250 16% 994 957 4% Gross Margin 81% 84% -300 bps 80% 81% -51 bps Non-GAAP Gross Profit: 304 248 22% 1,040 997 4% Non-GAAP Gross Margin1 85% 83% +190 bps 84% 84% No change Operating Expenses2: (211) (205) 3% (732) (919) (20)% Non-GAAP Operating Expenses1: (164) (179) (8)% (622) (655) (5)% Non-GAAP Selling and Marketing (83) (68) 23% (291) (255) 14% Non-GAAP General and Administrative (65) (84) (22)% (254) (296) (14)% Non-GAAP Research and Development (17) (27) (36)% (80) (103) (22)% Net Income 102 21 NM 210 7 NM Non-GAAP Net Income1 107 47 NM 320 240 33% Adjusted EBITDA1 142 75 91% 428 358 20% Adj. EBITDA Margin1 40% 25% 15 pp 35% 30% 5 pp
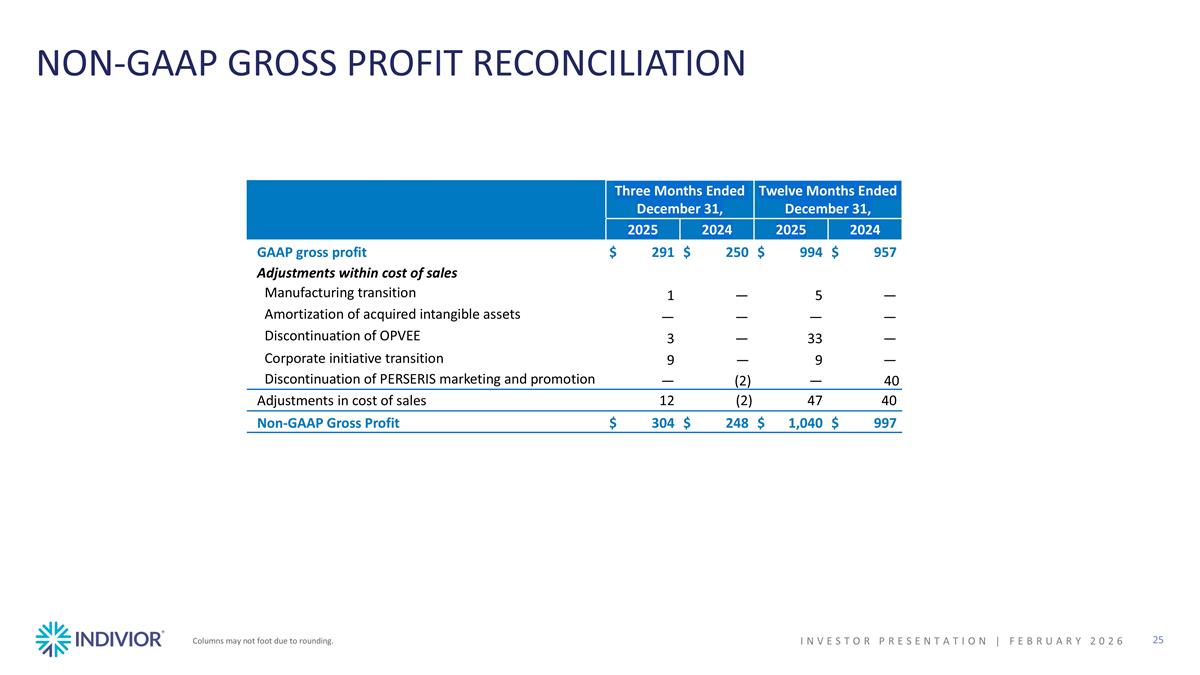
NON-GAAP GROSS PROFIT RECONCILIATION Columns may not foot due to rounding. INVESTOR PRESENTATION | February 2026 Three Months Ended December 31, Twelve Months Ended December 31, 2025 2024 2025 2024 GAAP gross profit $291 $250 $994 $957 Adjustments within cost of sales Manufacturing transition 1 — 5 — Amortization of acquired intangible assets — — — — Discontinuation of OPVEE 3 — 33 — Corporate initiative transition 9 — 9 — Discontinuation of PERSERIS marketing and promotion — (2) — 40 Adjustments in cost of sales 12 (2) 47 40 Non-GAAP Gross Profit $304 $248 $1,040 $997
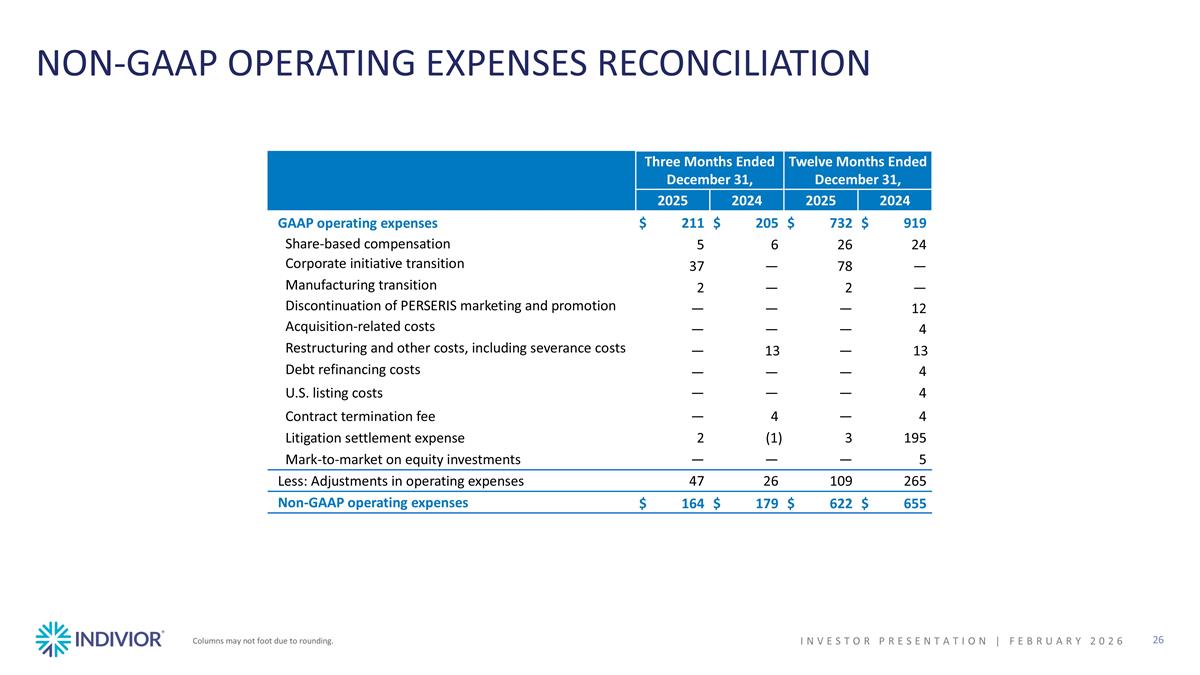
NON-GAAP OPERATING EXPENSES RECONCILIATION Columns may not foot due to rounding. INVESTOR PRESENTATION | February 2026 Three Months Ended December 31, Twelve Months Ended December 31, 2025 2024 2025 2024 GAAP operating expenses $211 $205 $732 $919 Share-based compensation 5 6 26 24 Corporate initiative transition 37 — 78 — Manufacturing transition 2 — 2 — Discontinuation of PERSERIS marketing and promotion — — — 12 Acquisition-related costs — — — 4 Restructuring and other costs, including severance costs — 13 — 13 Debt refinancing costs — — — 4 U.S. listing costs — — — 4 Contract termination fee — 4 — 4 Litigation settlement expense 2 (1) 3 195 Mark-to-market on equity investments — — — 5 Less: Adjustments in operating expenses 47 26 109 265 Non-GAAP operating expenses $164 $179 $622 $655
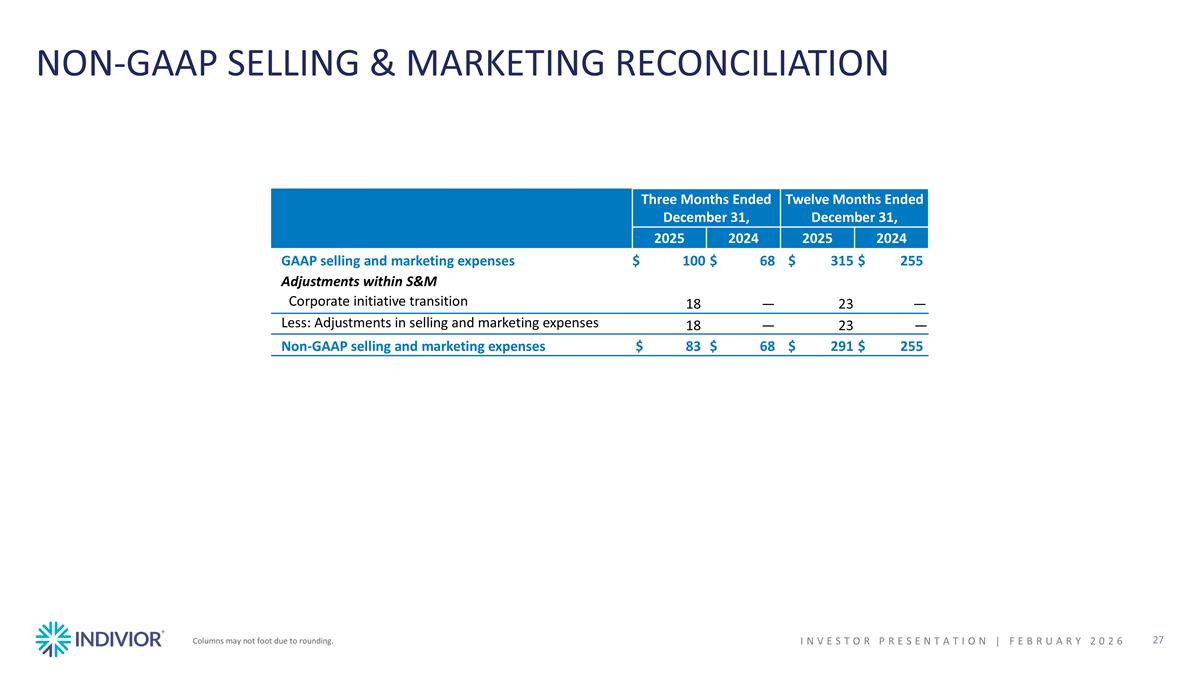
NON-GAAP SELLING & MARKETING RECONCILIATION Columns may not foot due to rounding. Three Months Ended December 31, Twelve Months Ended December 31, 2025 2024 2025 2024 GAAP selling and marketing expenses $100 $68 $315 $255 Adjustments within S&M Corporate initiative transition 18 — 23 — Less: Adjustments in selling and marketing expenses 18 — 23 — Non-GAAP selling and marketing expenses $83 $68 $291 $255 INVESTOR PRESENTATION | February 2026
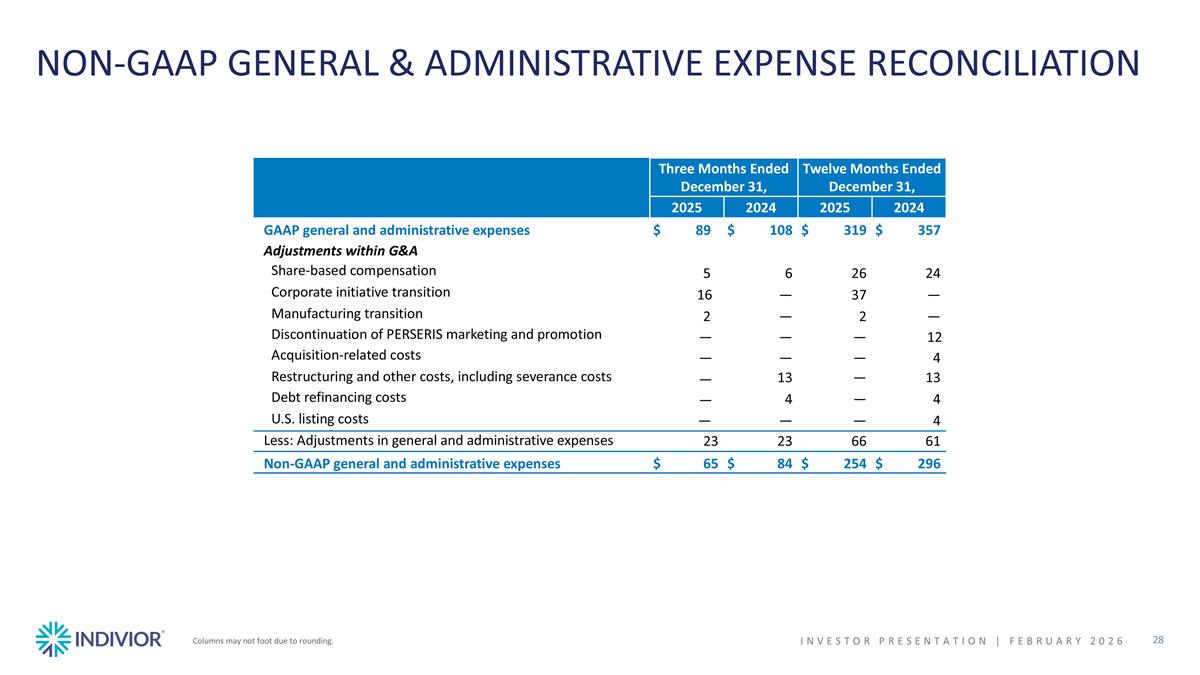
NON-GAAP GENERAL & ADMINISTRATIVE EXPENSE RECONCILIATION Columns may not foot due to rounding. INVESTOR PRESENTATION | February 2026 Three Months Ended December 31, Twelve Months Ended December 31, 2025 2024 2025 2024 GAAP general and administrative expenses $89 $108 $319 $357 Adjustments within G&A Share-based compensation 5 6 26 24 Corporate initiative transition 16 — 37 — Manufacturing transition 2 — 2 — Discontinuation of PERSERIS marketing and promotion — — — 12 Acquisition-related costs — — — 4 Restructuring and other costs, including severance costs — 13 — 13 Debt refinancing costs — 4 — 4 U.S. listing costs — — — 4 Less: Adjustments in general and administrative expenses 23 23 66 61 Non-GAAP general and administrative expenses $65 $84 $254 $296

NON-GAAP RESEARCH & DEVELOPMENT RECONCILIATION Columns may not foot due to rounding. INVESTOR PRESENTATION | February 2026 Three Months Ended December 31, Twelve Months Ended December 31, 2025 2024 2025 2024 GAAP research and development expenses $21 $31 $97 $107 Adjustments within R&D Impairment of products in development and related fees — 4 — 4 Corporate initiative transition 4 — 17 — Less: Adjustments in research and development expenses 4 4 17 4 Non-GAAP research and development expenses $17 $27 $80 $103
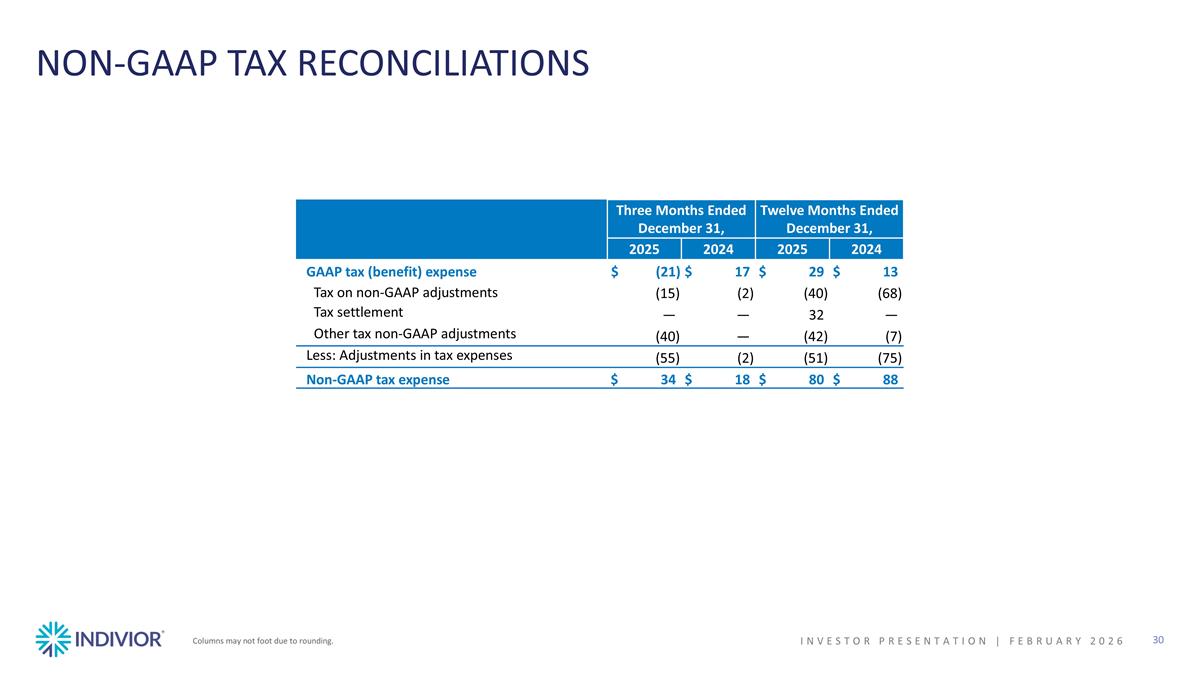
NON-GAAP TAX RECONCILIATIONS Columns may not foot due to rounding. INVESTOR PRESENTATION | February 2026 Three Months Ended December 31, Twelve Months Ended December 31, 2025 2024 2025 2024 GAAP tax (benefit) expense $(21) $17 $29 $13 Tax on non-GAAP adjustments (15) (2) (40) (68) Tax settlement — — 32 — Other tax non-GAAP adjustments (40) — (42) (7) Less: Adjustments in tax expenses (55) (2) (51) (75) Non-GAAP tax expense $34 $18 $80 $88
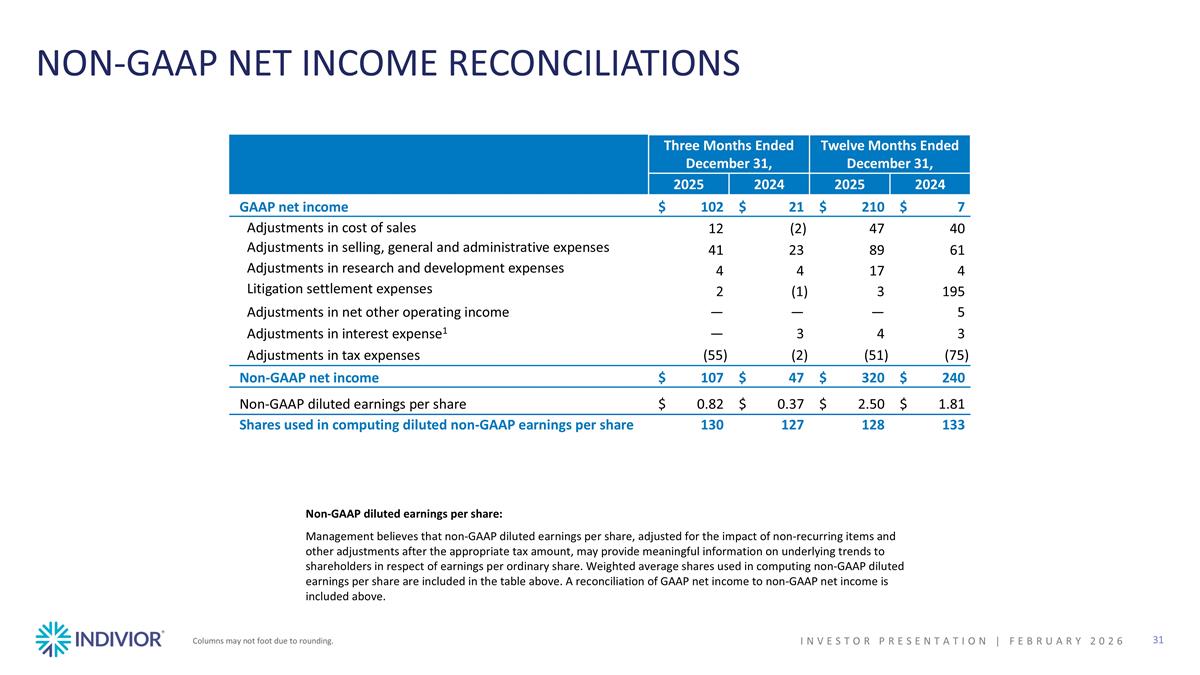
NON-GAAP NET INCOME RECONCILIATIONS Non-GAAP diluted earnings per share: Management believes that non-GAAP diluted earnings per share, adjusted for the impact of non-recurring items and other adjustments after the appropriate tax amount, may provide meaningful information on underlying trends to shareholders in respect of earnings per ordinary share. Weighted average shares used in computing non-GAAP diluted earnings per share are included in the table above. A reconciliation of GAAP net income to non-GAAP net income is included above. Columns may not foot due to rounding. INVESTOR PRESENTATION | February 2026 Three Months Ended December 31, Twelve Months Ended December 31, 2025 2024 2025 2024 GAAP net income $102 $21 $210 $7 Adjustments in cost of sales 12 (2) 47 40 Adjustments in selling, general and administrative expenses 41 23 89 61 Adjustments in research and development expenses 4 4 17 4 Litigation settlement expenses 2 (1) 3 195 Adjustments in net other operating income — — — 5 Adjustments in interest expense1 — 3 4 3 Adjustments in tax expenses (55) (2) (51) (75) Non-GAAP net income $107 $47 $320 $240 Non-GAAP diluted earnings per share $0.82 $0.37 $2.50 $1.81 Shares used in computing diluted non-GAAP earnings per share 130 127 128 133
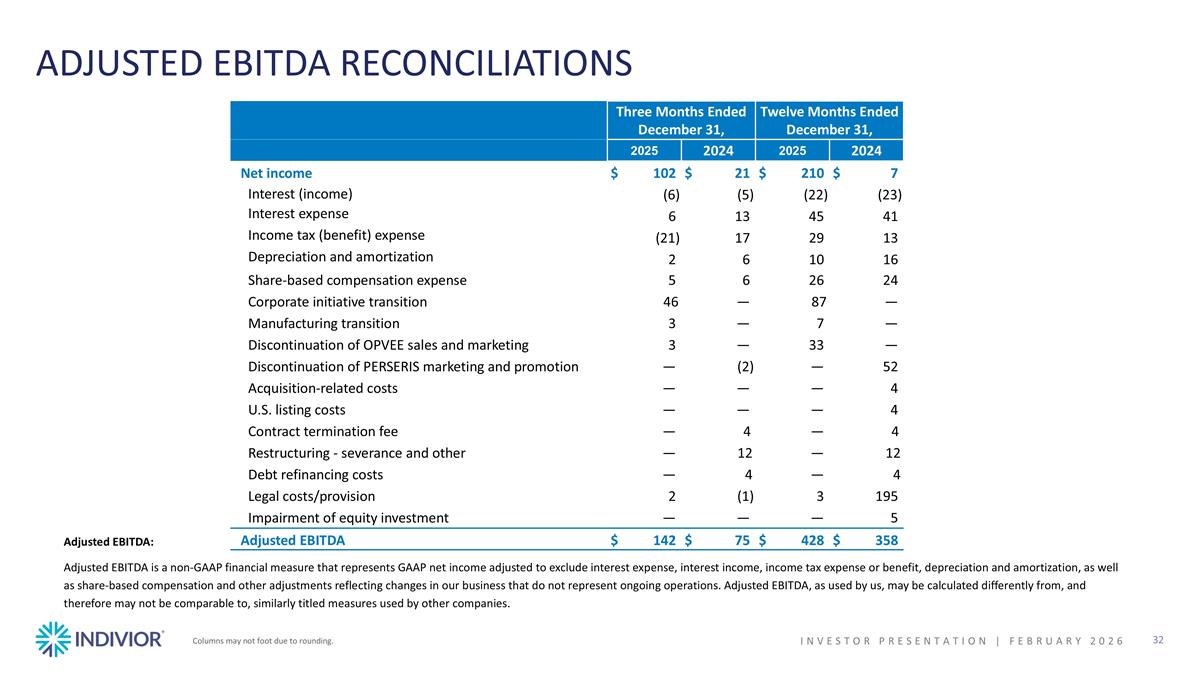
ADJUSTED EBITDA RECONCILIATIONS Adjusted EBITDA: Adjusted EBITDA is a non-GAAP financial measure that represents GAAP net income adjusted to exclude interest expense, interest income, income tax expense or benefit, depreciation and amortization, as well as share-based compensation and other adjustments reflecting changes in our business that do not represent ongoing operations. Adjusted EBITDA, as used by us, may be calculated differently from, and therefore may not be comparable to, similarly titled measures used by other companies. Columns may not foot due to rounding. INVESTOR PRESENTATION | February 2026 Three Months Ended December 31, Twelve Months Ended December 31, 2025 2024 2025 2024 Net income $102 $21 $210 $7 Interest (income) (6) (5) (22) (23) Interest expense 6 13 45 41 Income tax (benefit) expense (21) 17 29 13 Depreciation and amortization 2 6 10 16 Share-based compensation expense 5 6 26 24 Corporate initiative transition 46 — 87 — Manufacturing transition 3 — 7 — Discontinuation of OPVEE sales and marketing 3 — 33 — Discontinuation of PERSERIS marketing and promotion — (2) — 52 Acquisition-related costs — — — 4 U.S. listing costs — — — 4 Contract termination fee — 4 — 4 Restructuring - severance and other — 12 — 12 Debt refinancing costs — 4 — 4 Legal costs/provision 2 (1) 3 195 Impairment of equity investment — — — 5 Adjusted EBITDA $142 $75 $428 $358
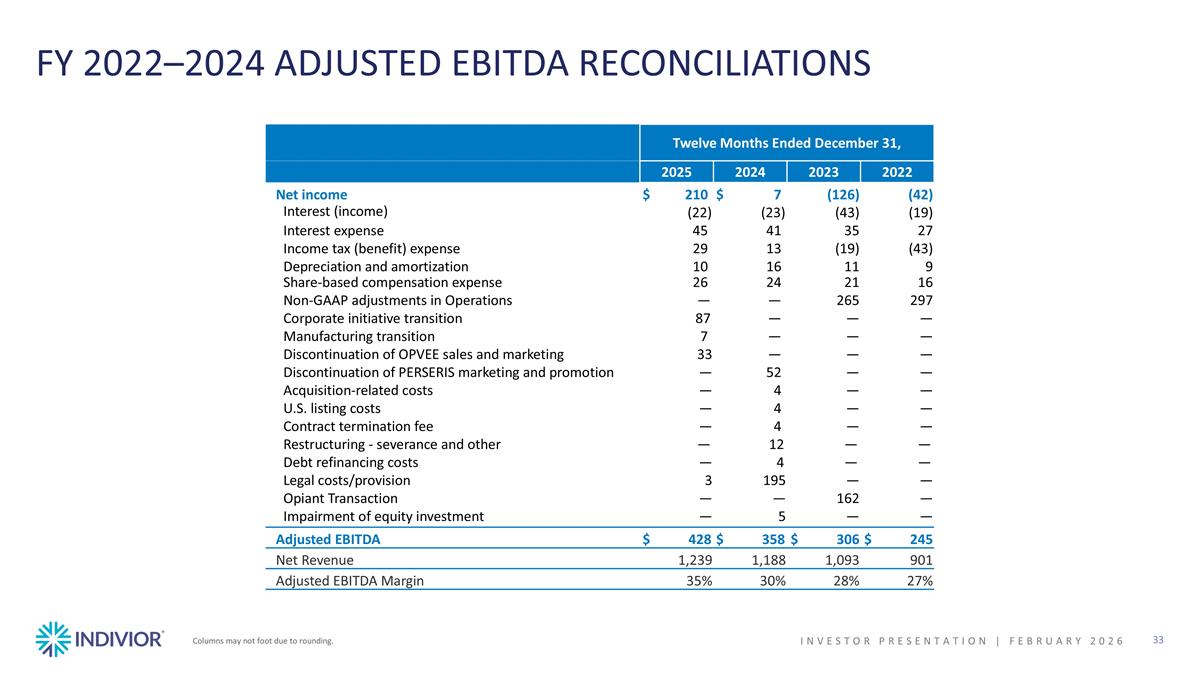
FY 2022–2024 ADJUSTED EBITDA RECONCILIATIONS Columns may not foot due to rounding. INVESTOR PRESENTATION | February 2026 Twelve Months Ended December 31, 2025 2024 2023 2022 Net income $210 $7 (126) (42) Interest (income) (22) (23) (43) (19) Interest expense 45 41 35 27 Income tax (benefit) expense 29 13 (19) (43) Depreciation and amortization 10 16 11 9 Share-based compensation expense 26 24 21 16 Non-GAAP adjustments in Operations — — 265 297 Corporate initiative transition 87 — — — Manufacturing transition 7 — — — Discontinuation of OPVEE sales and marketing 33 — — — Discontinuation of PERSERIS marketing and promotion — 52 — — Acquisition-related costs — 4 — — U.S. listing costs — 4 — — Contract termination fee — 4 — — Restructuring - severance and other — 12 — — Debt refinancing costs — 4 — — Legal costs/provision 3 195 — — Opiant Transaction — — 162 — Impairment of equity investment — 5 — — Adjusted EBITDA $ 428 $ 358 $ 306 $ 245 Net Revenue 1,239 1,188 1,093 901 Adjusted EBITDA Margin 35% 30% 28% 27%

TRAILING TWELVE MONTHS LEVERAGE RECONCILIATION 1. Net Debt represents $333m of the outstanding balance of the note purchase agreement less $50m of cash. Columns may not foot due to rounding. INVESTOR PRESENTATION | February 2026 ($ in mil.) 2025 Net Debt1 $283 Net income 210 Adjustments: Interest income (22) Interest expense 45 Income tax expense 29 Depreciation and amortization 10 Non-GAAP adjustments in operating income 127 Share-based compensation expense 26 Legal costs/provision 3 Total Adjustments 218 Adjusted EBITDA $428 Adjusted Leverage 0.7

SUBLOCADE® (buprenorphine extended-release) injection, for subcutaneous use (CIII) INDICATION SUBLOCADE is indicated for the treatment of moderate to severe opioid use disorder in patients who have initiated treatment with a single dose of a transmucosal buprenorphine product or who are already being treated with buprenorphine. SUBLOCADE should be used as part of a complete treatment plan that includes counseling and psychosocial support. HIGHLIGHTED SAFETY INFORMATION WARNING: RISK OF SERIOUS HARM OR DEATH WITH INTRAVENOUS ADMINISTRATION; SUBLOCADE RISK EVALUATION AND MITIGATION STRATEGY See full prescribing information for complete boxed warning. Serious harm or death could result if administered intravenously. SUBLOCADE is only available through a restricted program called the SUBLOCADE REMS Program. Healthcare settings and pharmacies that order and dispense SUBLOCADE must be certified in this program and comply with the REMS requirements. CONTRAINDICATIONS Hypersensitivity to buprenorphine or any other ingredients in SUBLOCADE. WARNINGS AND PRECAUTIONS Addiction, Abuse, and Misuse: SUBLOCADE contains buprenorphine, a Schedule III controlled substance that can be abused in a manner similar to other opioids. Monitor patients for conditions indicative of diversion or progression of opioid dependence and addictive behaviors. Respiratory Depression: Life threatening respiratory depression and death have occurred in association with buprenorphine. Warn patients of the potential danger of self-administration of benzodiazepines or other CNS depressants while under treatment with SUBLOCADE. Risk of Serious Injection Site Reactions: Likelihood of may increase with inadvertent intramuscular or intradermal administration. Evaluate and treat as appropriate. The most common injection site reactions are pain, erythema and pruritus with some involving abscess, ulceration and necrosis. Neonatal Opioid Withdrawal Syndrome: Neonatal opioid withdrawal syndrome (NOWS) is an expected and treatable outcome of prolonged use of opioids during pregnancy. Adrenal Insufficiency: If diagnosed, treat with physiologic replacement of corticosteroids, and wean patient off the opioid. Risk of Opioid Withdrawal With Abrupt Discontinuation: If treatment with SUBLOCADE is discontinued, monitor patients for several months for withdrawal and treat appropriately. Risk of Hepatitis, Hepatic Events: Monitor liver function tests prior to and during treatment. Risk of Withdrawal in Patients Dependent on Full Agonist Opioids: Verify that patients have tolerated transmucosal buprenorphine before injecting SUBLOCADE. Treatment of Emergent Acute Pain: Treat pain with a non-opioid analgesic whenever possible. If opioid therapy is required, monitor patients closely because higher doses may be required for analgesic effect. ADVERSE REACTIONS Adverse reactions commonly associated with SUBLOCADE (in ≥5% of subjects) were constipation, headache, nausea, injection site pruritus, vomiting, increased hepatic enzymes, fatigue, and injection site pain. For more information about SUBLOCADE, the full Prescribing Information including BOXED WARNING, and Medication Guide, visit www.sublocade.com. INVESTOR PRESENTATION | February 2026