

MARCH 2026 We have an audacious mission to develop novel therapies for diseases with high unmet needs, with a focus on serious and fatal endocrine conditions and neurodegenerative diseases. Maggie, a mom and advocate living with post-bariatric hypoglycemia (PBH). Pancreatic islet cells

DISCLAIMER Statements contained in this presentation regarding matters that are not historical facts are “forward-looking statements” within the meaning of the Private Securities Litigation Reform Act of 1995, as amended. Because such statements are subject to risks and uncertainties, actual results may differ materially from those expressed or implied by such forward-looking statements. Such statements include, but are not limited to, the Company’s plans to explore the use of avexitide as a treatment for post-bariatric hypoglycemia (PBH) and congenital hyperinsulinism, AMX0035 for Wolfram syndrome, AMX0114 for ALS, and AMX0318 for PBH and other rare diseases; statements regarding the timing of clinical trials for PBH, Wolfram syndrome and/or ALS; expectation for regulatory action; and expectations regarding our longer-term strategy and expected cash runway. Any forward-looking statements in this presentation are based on management’s current expectations of future events and are subject to a number of risks and uncertainties that could cause actual results to differ materially and adversely from those set forth in or implied by such forward- looking statements. Risks that contribute to the uncertain nature of the forward-looking statements include: the success, cost, and timing of Amylyx’s program development activities, including ongoing and planned clinical trials, Amylyx’s ability to execute on its development and regulatory strategy, regulatory developments, Amylyx’s cash runway and ability to fund operations, as well as the risks and uncertainties set forth in Amylyx’s United States Securities and Exchange Commission (SEC) filings, including Amylyx’s Annual Report on Form 10-K for the year ended December 31, 2025, and subsequent filings with the SEC. All forward-looking statements contained in this presentation speak only as of the date on which they were made. Amylyx undertakes no obligation to update such statements to reflect events that occur or circumstances that exist after the date on which they were made. 2

A Growing Pipeline of Therapies to Serve Communities with High Unmet Needs Led by an experienced team with a proven track record of commercialization in rare diseases IND-ENABLING PRECLINICAL PHASE 1 PHASE 2 PHASE 3 COMMERCIAL STUDIES • Phase 3 LUCIDITY trial underway. Enrollment completed. Topline Post-Bariatric AVEXITIDE data expected in Q3 2026, and if approved, commercial launch Hypoglycemia LUCIDITY PHASE 3 CLINICAL TRIAL anticipated in 2027 GLP-1 receptor (PBH) antagonist• FDA-agreed upon primary outcome of reduction in the composite of Level 2 and Level 3 hypoglycemic events through Week 16 FDA Breakthrough Congenital Therapy and Orphan • Engaging physician and community experts around next steps Hyperinsulinism Drug Designations for clinical development (HI) • Pending alignment with the FDA, plan to initiate a focused, pivotal Phase 3 trial of AMX0035 in Wolfram syndrome AMX0035 Wolfram • Positive Phase 2 HELIOS data showed improvement or stabilization HELIOS PHASE 2 CLINICAL TRIAL Sodium phenylbutyrate and Syndrome across all disease measures at Week 24 (N=11) and sustained taurursodiol (also known as improvement or stabilization at Week 48 (N=10) ursodoxicoltaurine) • Early data from Cohort 1 (n=12) demonstrated AMX0114 was generally well-tolerated, with no treatment-related SAEs in Phase 1 AMX0114 Amyotrophic LUMINA trial ASO targeting calpain-2, a Lateral Sclerosis LUMINA PHASE 1 CLINICAL TRIAL protein involved in axonal • Completed enrollment in Cohort 2 (n=12) in March 2026 (ALS) degeneration • Granted FDA Fast Track designation • As part of research collaboration with Gubra, AMX0318 demonstrated a robust chemical stability profile, strong in vitro potency, evidence of AMX0318 PBH and Other in vivo efficacy and tolerability, high solubility, and a favorable Long-acting GLP-1 Rare Diseases pharmacokinetic profile consistent with a long-acting peptide receptor antagonist • IND-enabling studies underway, with an IND filing targeted for 2027 ASO=antisense oligonucleotide; FDA=U.S. Food and Drug Administration; GLP-1=glucagon-like peptide-1; IND=investigational new drug; SAE=serious adverse events. 3

AVEXITIDE Investigational, First-in-Class GLP-1 Receptor Antagonist with FDA Breakthrough Therapy Designation Pancreatic islet cells
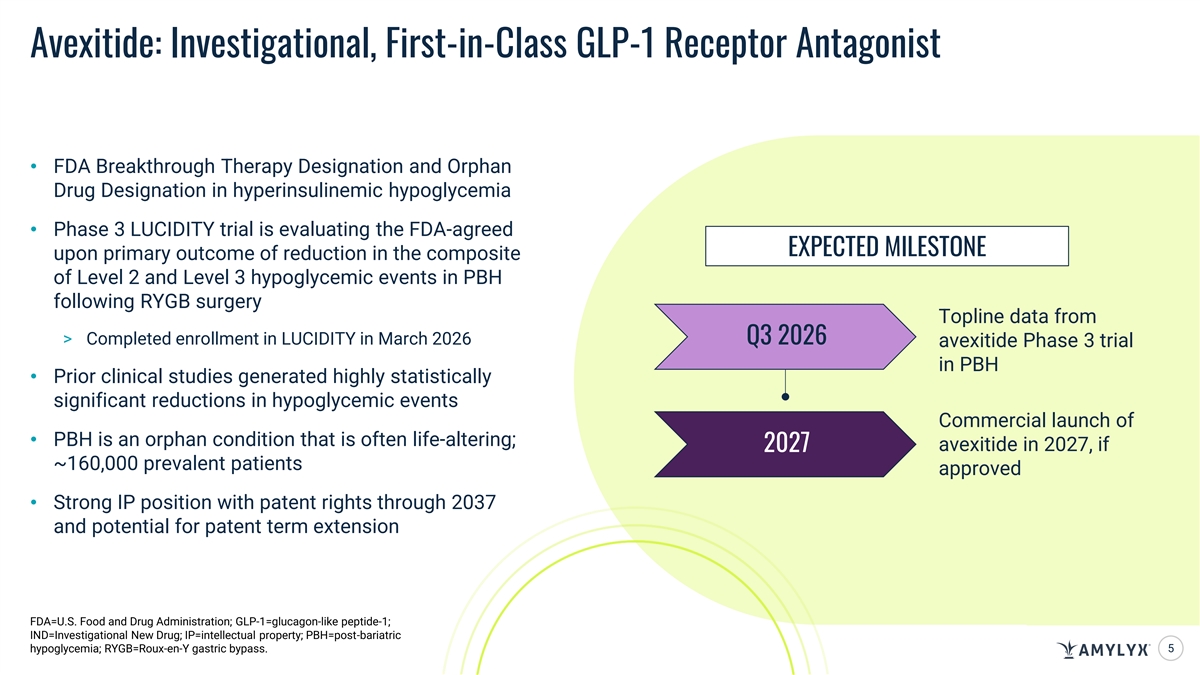
Avexitide: Investigational, First-in-Class GLP-1 Receptor Antagonist • FDA Breakthrough Therapy Designation and Orphan Drug Designation in hyperinsulinemic hypoglycemia • Phase 3 LUCIDITY trial is evaluating the FDA-agreed EXPECTED MILESTONE upon primary outcome of reduction in the composite of Level 2 and Level 3 hypoglycemic events in PBH following RYGB surgery Topline data from > Completed enrollment in LUCIDITY in March 2026 Q3 2026 avexitide Phase 3 trial in PBH • Prior clinical studies generated highly statistically significant reductions in hypoglycemic events Commercial launch of • PBH is an orphan condition that is often life-altering; avexitide in 2027, if 2027 ~160,000 prevalent patients approved • Strong IP position with patent rights through 2037 and potential for patent term extension FDA=U.S. Food and Drug Administration; GLP-1=glucagon-like peptide-1; IND=Investigational New Drug; IP=intellectual property; PBH=post-bariatric hypoglycemia; RYGB=Roux-en-Y gastric bypass. 5
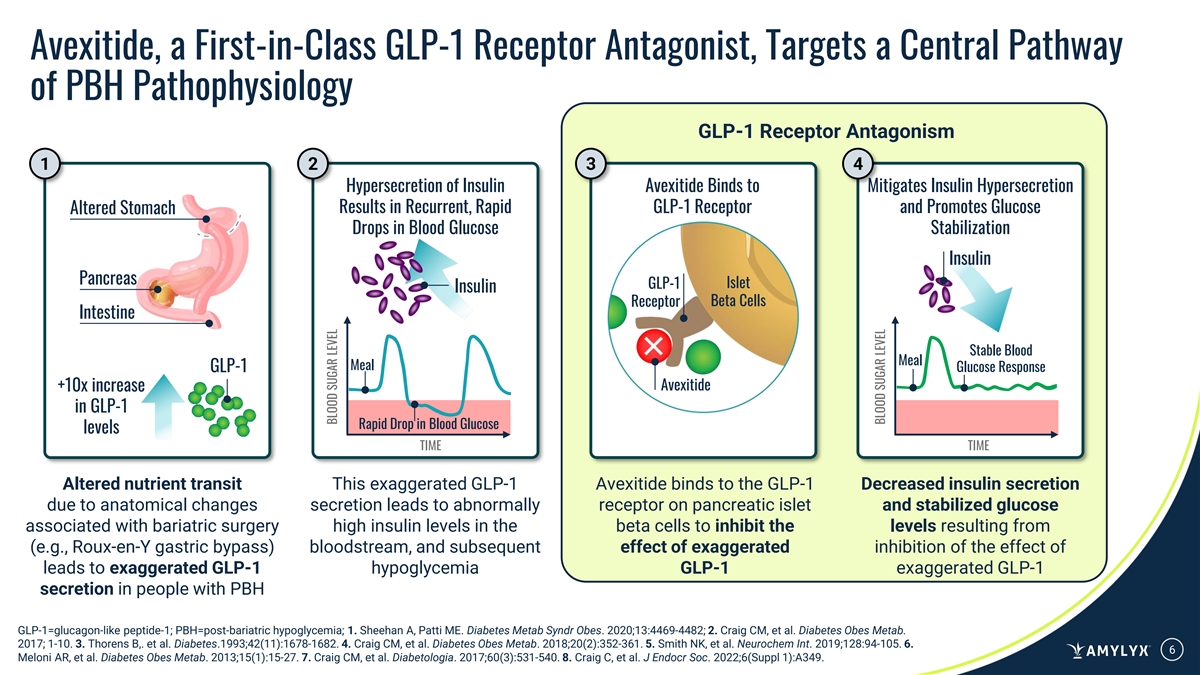
Avexitide, a First-in-Class GLP-1 Receptor Antagonist, Targets a Central Pathway of PBH Pathophysiology GLP-1 Receptor Antagonism 1 2 3 4 Hypersecretion of Insulin Avexitide Binds to Mitigates Insulin Hypersecretion Results in Recurrent, Rapid GLP-1 Receptor and Promotes Glucose Altered Stomach Drops in Blood Glucose Stabilization Insulin Pancreas GLP-1 Islet Insulin Beta Cells Receptor Intestine Stable Blood Meal Meal GLP-1 Glucose Response +10x increase Avexitide in GLP-1 Rapid Drop in Blood Glucose levels TIME TIME Altered nutrient transit This exaggerated GLP-1 Avexitide binds to the GLP-1 Decreased insulin secretion due to anatomical changes secretion leads to abnormally receptor on pancreatic islet and stabilized glucose associated with bariatric surgery high insulin levels in the beta cells to inhibit the levels resulting from (e.g., Roux-en-Y gastric bypass) bloodstream, and subsequent effect of exaggerated inhibition of the effect of leads to exaggerated GLP-1 hypoglycemia GLP-1 exaggerated GLP-1 secretion in people with PBH GLP-1=glucagon-like peptide-1; PBH=post-bariatric hypoglycemia; 1. Sheehan A, Patti ME. Diabetes Metab Syndr Obes. 2020;13:4469-4482; 2. Craig CM, et al. Diabetes Obes Metab. 2017; 1-10. 3. Thorens B,. et al. Diabetes.1993;42(11):1678-1682. 4. Craig CM, et al. Diabetes Obes Metab. 2018;20(2):352-361. 5. Smith NK, et al. Neurochem Int. 2019;128:94-105. 6. 6 Meloni AR, et al. Diabetes Obes Metab. 2013;15(1):15-27. 7. Craig CM, et al. Diabetologia. 2017;60(3):531-540. 8. Craig C, et al. J Endocr Soc. 2022;6(Suppl 1):A349. BLOOD SUGAR LEVEL BLOOD SUGAR LEVEL
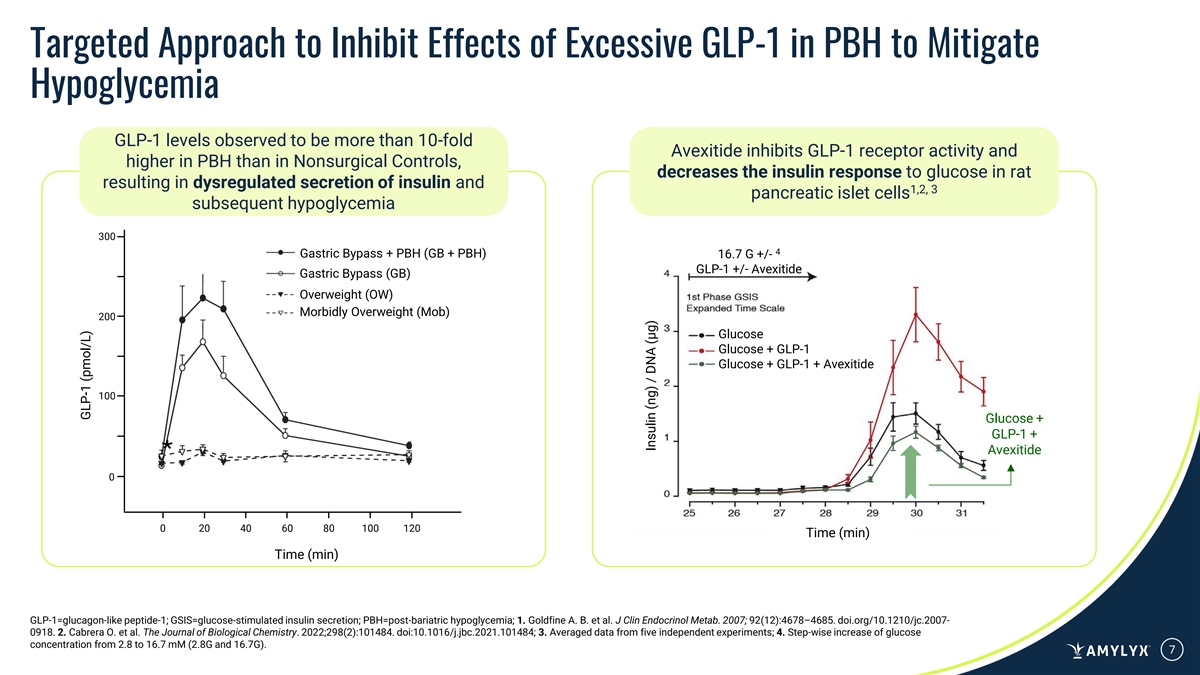
Targeted Approach to Inhibit Effects of Excessive GLP-1 in PBH to Mitigate Hypoglycemia GLP-1 levels observed to be more than 10-fold Avexitide inhibits GLP-1 receptor activity and higher in PBH than in Nonsurgical Controls, decreases the insulin response to glucose in rat resulting in dysregulated secretion of insulin and 1,2, 3 pancreatic islet cells subsequent hypoglycemia 300 4 Gastric Bypass + PBH (GB + PBH) 16.7 G +/- GLP-1 +/- Avexitide Gastric Bypass (GB) Overweight (OW) Morbidly Overweight (Mob) 200 Glucose Glucose + GLP-1 Glucose + GLP-1 + Avexitide 100 Glucose + GLP-1 + Avexitide 0 0 20 40 60 80 100 120 Time (min) Time (min) GLP-1=glucagon-like peptide-1; GSIS=glucose-stimulated insulin secretion; PBH=post-bariatric hypoglycemia; 1. Goldfine A. B. et al. J Clin Endocrinol Metab. 2007; 92(12):4678–4685. doi.org/10.1210/jc.2007- 0918. 2. Cabrera O. et al. The Journal of Biological Chemistry. 2022;298(2):101484. doi:10.1016/j.jbc.2021.101484; 3. Averaged data from five independent experiments; 4. Step-wise increase of glucose concentration from 2.8 to 16.7 mM (2.8G and 16.7G). 7 GLP-1 (pmol/L) Insulin (ng) / DNA (µg)
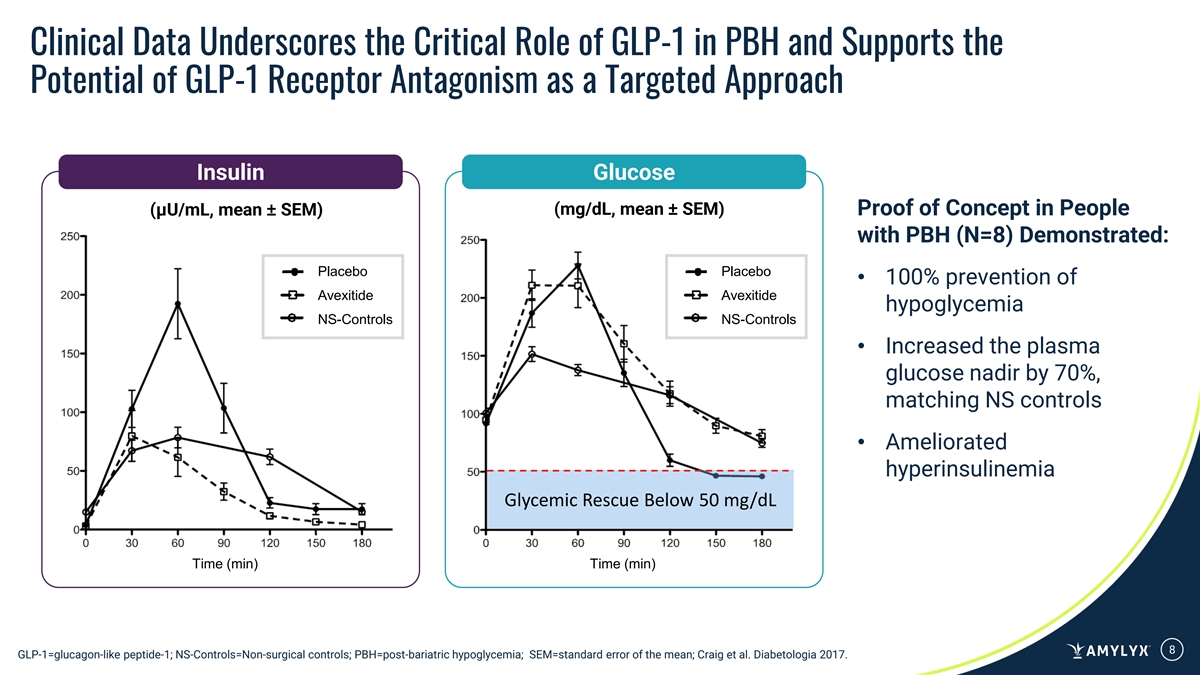
Clinical Data Underscores the Critical Role of GLP-1 in PBH and Supports the Potential of GLP-1 Receptor Antagonism as a Targeted Approach Insulin Glucose (mg/dL, mean ± SEM) Proof of Concept in People (μU/mL, mean ± SEM) with PBH (N=8) Demonstrated: Placebo Placebo • 100% prevention of Avexitide Avexitide hypoglycemia NS-Controls NS-Controls • Increased the plasma glucose nadir by 70%, matching NS controls • Ameliorated hyperinsulinemia Glycemic Rescue Below 50 mg/dL Time (min) Time (min) 8 GLP-1=glucagon-like peptide-1; NS-Controls=Non-surgical controls; PBH=post-bariatric hypoglycemia; SEM=standard error of the mean; Craig et al. Diabetologia 2017.
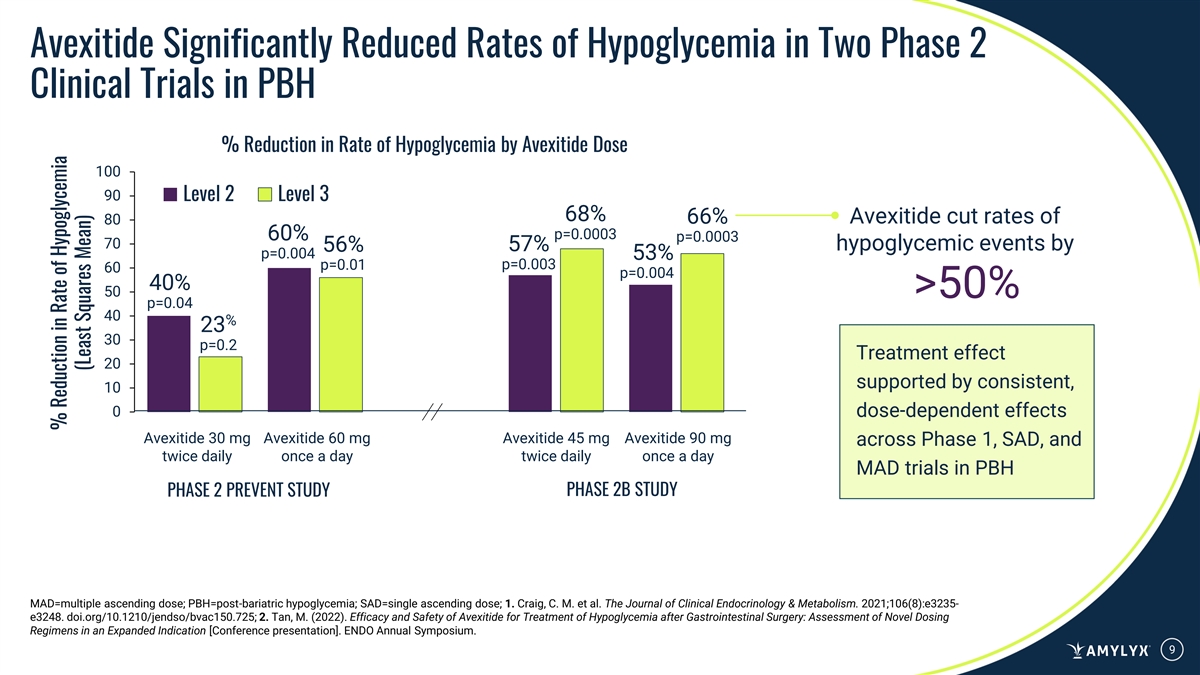
Avexitide Significantly Reduced Rates of Hypoglycemia in Two Phase 2 Clinical Trials in PBH % Reduction in Rate of Hypoglycemia by Avexitide Dose 100 90 Level 2 Level 3 68% 66% Avexitide cut rates of 80 p=0.0003 60% p=0.0003 70 57% hypoglycemic events by 56% p=0.004 53% p=0.01 p=0.003 60 p=0.004 40% 50 >50% p=0.04 40 % 23 30 p=0.2 Treatment effect 20 supported by consistent, 10 0 dose-dependent effects Avexitide 30 mg Avexitide 60 mg Avexitide 45 mg Avexitide 90 mg across Phase 1, SAD, and twice daily once a day twice daily once a day MAD trials in PBH PHASE 2B STUDY PHASE 2 PREVENT STUDY MAD=multiple ascending dose; PBH=post-bariatric hypoglycemia; SAD=single ascending dose; 1. Craig, C. M. et al. The Journal of Clinical Endocrinology & Metabolism. 2021;106(8):e3235- e3248. doi.org/10.1210/jendso/bvac150.725; 2. Tan, M. (2022). Efficacy and Safety of Avexitide for Treatment of Hypoglycemia after Gastrointestinal Surgery: Assessment of Novel Dosing Regimens in an Expanded Indication [Conference presentation]. ENDO Annual Symposium. 9 % Reduction in Rate of Hypoglycemia (Least Squares Mean)
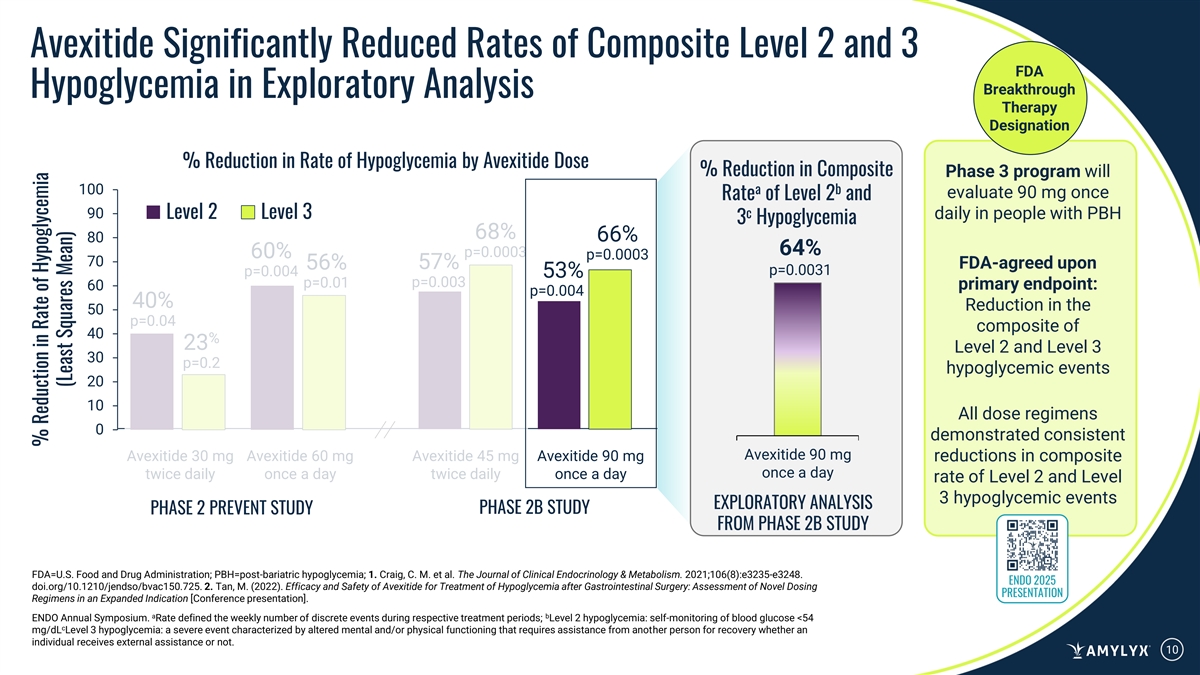
Avexitide Significantly Reduced Rates of Composite Level 2 and 3 FDA Breakthrough Hypoglycemia in Exploratory Analysis Therapy Designation % Reduction in Rate of Hypoglycemia by Avexitide Dose % Reduction in Composite Phase 3 program will 100 a b evaluate 90 mg once Rate of Level 2 and 90 Level 2 Level 3 c daily in people with PBH 3 Hypoglycemia 68% 66% 80 64% p=0.0003 60% p=0.0003 70 57% FDA-agreed upon 56% p=0.0031 p=0.004 53% p=0.01 p=0.003 60 primary endpoint: p=0.004 40% Reduction in the 50 p=0.04 composite of 40 % 23 Level 2 and Level 3 30 p=0.2 hypoglycemic events 20 10 All dose regimens 0 demonstrated consistent Avexitide 90 mg Avexitide 30 mg Avexitide 60 mg Avexitide 45 mg Avexitide 90 mg reductions in composite once a day twice daily once a day twice daily once a day rate of Level 2 and Level 3 hypoglycemic events EXPLORATORY ANALYSIS PHASE 2B STUDY PHASE 2 PREVENT STUDY FROM PHASE 2B STUDY FDA=U.S. Food and Drug Administration; PBH=post-bariatric hypoglycemia; 1. Craig, C. M. et al. The Journal of Clinical Endocrinology & Metabolism. 2021;106(8):e3235-e3248. ENDO 2025 doi.org/10.1210/jendso/bvac150.725. 2. Tan, M. (2022). Efficacy and Safety of Avexitide for Treatment of Hypoglycemia after Gastrointestinal Surgery: Assessment of Novel Dosing PRESENTATION Regimens in an Expanded Indication [Conference presentation]. a b ENDO Annual Symposium. Rate defined the weekly number of discrete events during respective treatment periods; Level 2 hypoglycemia: self-monitoring of blood glucose <54 c mg/dL Level 3 hypoglycemia: a severe event characterized by altered mental and/or physical functioning that requires assistance from another person for recovery whether an individual receives external assistance or not. 10 % Reduction in Rate of Hypoglycemia (Least Squares Mean)
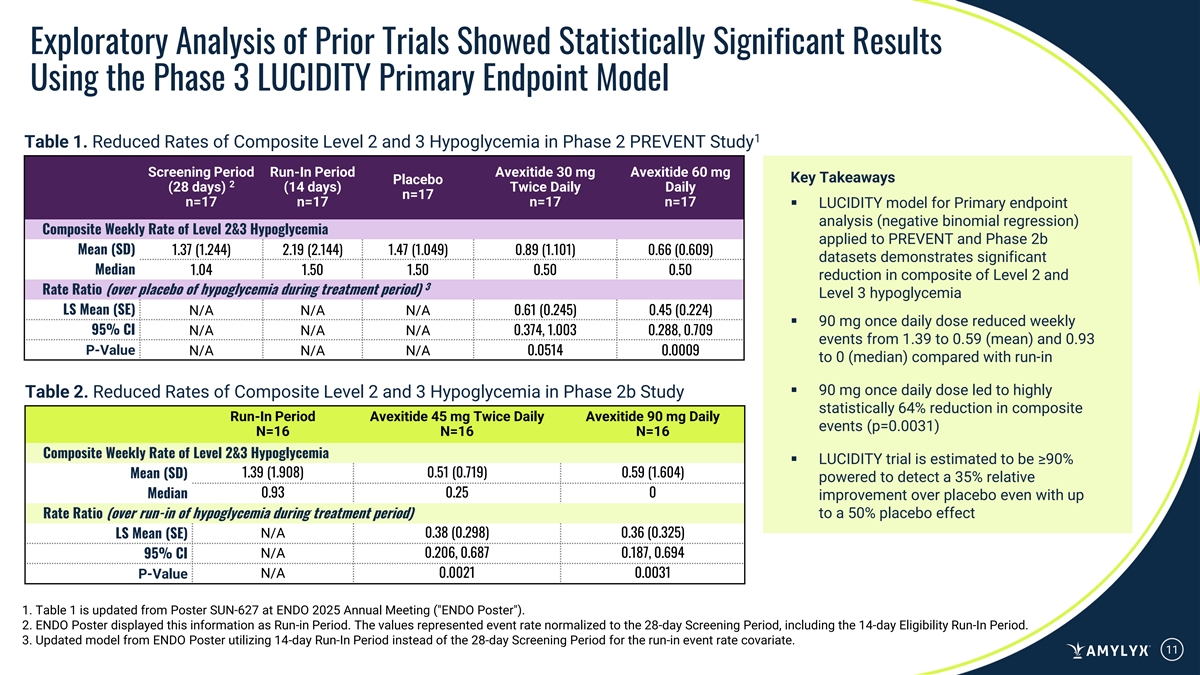
Exploratory Analysis of Prior Trials Showed Statistically Significant Results Using the Phase 3 LUCIDITY Primary Endpoint Model 1 Table 1. Reduced Rates of Composite Level 2 and 3 Hypoglycemia in Phase 2 PREVENT Study Screening Period Run-In Period Avexitide 30 mg Avexitide 60 mg Key Takeaways Placebo 2 (28 days) (14 days) Twice Daily Daily n=17 n=17 n=17 n=17 n=17 ▪ LUCIDITY model for Primary endpoint analysis (negative binomial regression) Composite Weekly Rate of Level 2&3 Hypoglycemia applied to PREVENT and Phase 2b Mean (SD) 1.37 (1.244) 2.19 (2.144) 1.47 (1.049) 0.89 (1.101) 0.66 (0.609) datasets demonstrates significant Median 1.04 1.50 1.50 0.50 0.50 reduction in composite of Level 2 and 3 Rate Ratio (over placebo of hypoglycemia during treatment period) Level 3 hypoglycemia LS Mean (SE) N/A N/A N/A 0.61 (0.245) 0.45 (0.224) ▪ 90 mg once daily dose reduced weekly 95% CI N/A N/A N/A 0.374, 1.003 0.288, 0.709 events from 1.39 to 0.59 (mean) and 0.93 P-Value N/A N/A N/A 0.0514 0.0009 to 0 (median) compared with run-in ▪ 90 mg once daily dose led to highly Table 2. Reduced Rates of Composite Level 2 and 3 Hypoglycemia in Phase 2b Study statistically 64% reduction in composite Run-In Period Avexitide 45 mg Twice Daily Avexitide 90 mg Daily events (p=0.0031) N=16 N=16 N=16 Composite Weekly Rate of Level 2&3 Hypoglycemia ▪ LUCIDITY trial is estimated to be ≥90% 1.39 (1.908) 0.51 (0.719) 0.59 (1.604) Mean (SD) powered to detect a 35% relative 0.93 0.25 0 Median improvement over placebo even with up Rate Ratio (over run-in of hypoglycemia during treatment period) to a 50% placebo effect N/A 0.38 (0.298) 0.36 (0.325) LS Mean (SE) N/A 0.206, 0.687 0.187, 0.694 95% CI N/A 0.0021 0.0031 P-Value 1. Table 1 is updated from Poster SUN-627 at ENDO 2025 Annual Meeting ( ENDO Poster ). 2. ENDO Poster displayed this information as Run-in Period. The values represented event rate normalized to the 28-day Screening Period, including the 14-day Eligibility Run-In Period. 3. Updated model from ENDO Poster utilizing 14-day Run-In Period instead of the 28-day Screening Period for the run-in event rate covariate. 11

CONSISTENT RESULTS IN PBH CLINICAL TRIALS Avexitide Reproducibly Improved Insulin and Glucose Responses During Standardized Meal Tests in People with PBH 1 2 3 4 Phase 1 SAD MAD Phase 2 PREVENT Avexitide Avexitide Avexitide Avexitide Avexitide IV infusion SC injection 30 mg BID 30 mg BID 60 mg QD SC injection SC injection SC injection (n=8) (N=8) 5 5 (n=5) (N=17) (N=17) Improvement vs. Placebo Postprandial Increase Increase Increase Increase Increase Glucose Nadir (p<0.001) (p<0.001) (p<0.05) (p=0.001) (p=0.0002) BID=twice daily; IV=intravenous; MAD=multiple ascending dose; PBH=post-bariatric hypoglycemia; QD=once daily; SAD=single ascending dose; SC=subcutaneous; 1. Craig, C. M. et al. Diabetologia. 2017;60(3):531-540. doi:10.1007/s00125-016-4179-x; 2. Craig, C. M. et al. Diabetes, Obesity & Metabolism. 2018;20:352–361. doi.org/10.1111/dom.13078; 3. Tan, M. et al. Diabetes, Obesity & Metabolism. 2020;22(8):1406-1416. doi:10.1111/dom.14048; 4. Craig, C. M. et al. The Journal of Clinical Endocrinology & Metabolism. 2021;106(8):e3235-e3248. doi:10.1210/clinem/dgab103; 5.18 participants were randomized and completed the trial with 17 included in the efficacy analysis due to a major protocol deviation (glycemic rescue 12 was not administered as indicated per protocol during the Period 1 placebo MMTT).

Avexitide was Generally Well-Tolerated with a Favorable Safety Profile Across Both Phase 2 Trials 1 2 Phase 2 PREVENT Study Phase 2b Study AEs generally mild to moderate and AEs generally mild to moderate and transient transient No clinically meaningful increases were observed in No treatment-related serious AEs • 1 serious adverse event (presyncope fasting or peak postprandial during avexitide 60 mg once daily) No serious AEs plasma glucose levels occurred; reported as unrelated to study (i.e., no hyperglycemia drug and self-limited observed) Most common AEs were diarrhea, Most common AEs were injection site* headache, bloating, and injection* site bruising, headache, and nausea reaction/bruising No participant discontinuations No participant discontinuations *Injection site reactions generally mild and transient with no grade 3 events or resulting discontinuations AE=adverse event; 1. Craig, C. M. et al. The Journal of Clinical Endocrinology & Metabolism. 2021;106(8):e3235-e3248. doi.org/10.1210/jendso/bvac150.725. 2. Tan, M. (2022). Efficacy and Safety of Avexitide for Treatment of Hypoglycemia after Gastrointestinal Surgery: Assessment of Novel Dosing Regimens in an Expanded Indication [Conference presentation]. ENDO Annual Symposium. 13
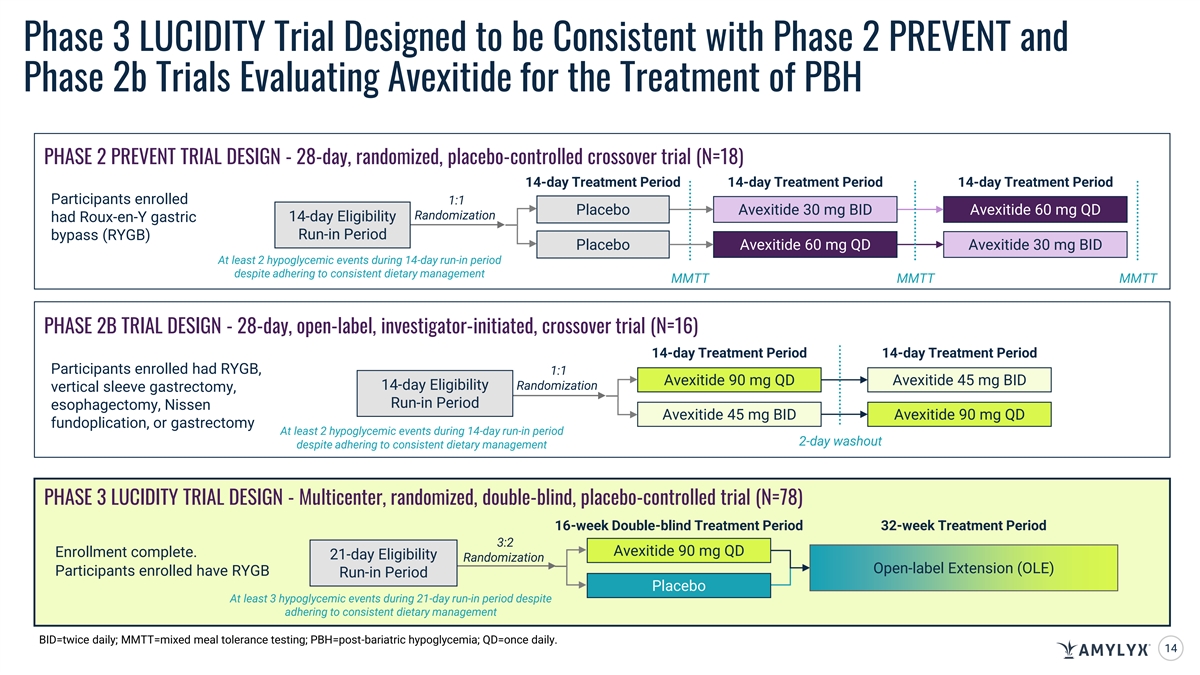
Phase 3 LUCIDITY Trial Designed to be Consistent with Phase 2 PREVENT and Phase 2b Trials Evaluating Avexitide for the Treatment of PBH PHASE 2 PREVENT TRIAL DESIGN - 28-day, randomized, placebo-controlled crossover trial (N=18) 14-day Treatment Period 14-day Treatment Period 14-day Treatment Period Participants enrolled 1:1 Placebo Avexitide 30 mg BID Avexitide 60 mg QD Randomization 14-day Eligibility had Roux-en-Y gastric bypass (RYGB) Run-in Period Placebo Avexitide 60 mg QD Avexitide 30 mg BID At least 2 hypoglycemic events during 14-day run-in period despite adhering to consistent dietary management MMTT MMTT MMTT PHASE 2B TRIAL DESIGN - 28-day, open-label, investigator-initiated, crossover trial (N=16) 14-day Treatment Period 14-day Treatment Period Participants enrolled had RYGB, 1:1 Avexitide 90 mg QD Avexitide 45 mg BID 14-day Eligibility Randomization vertical sleeve gastrectomy, Run-in Period esophagectomy, Nissen Avexitide 45 mg BID Avexitide 90 mg QD fundoplication, or gastrectomy At least 2 hypoglycemic events during 14-day run-in period 2-day washout despite adhering to consistent dietary management PHASE 3 LUCIDITY TRIAL DESIGN - Multicenter, randomized, double-blind, placebo-controlled trial (N=78) 16-week Double-blind Treatment Period 32-week Treatment Period 3:2 Avexitide 90 mg QD Enrollment complete. 21-day Eligibility Randomization Open-label Extension (OLE) Participants enrolled have RYGB Run-in Period Placebo At least 3 hypoglycemic events during 21-day run-in period despite adhering to consistent dietary management BID=twice daily; MMTT=mixed meal tolerance testing; PBH=post-bariatric hypoglycemia; QD=once daily. 14
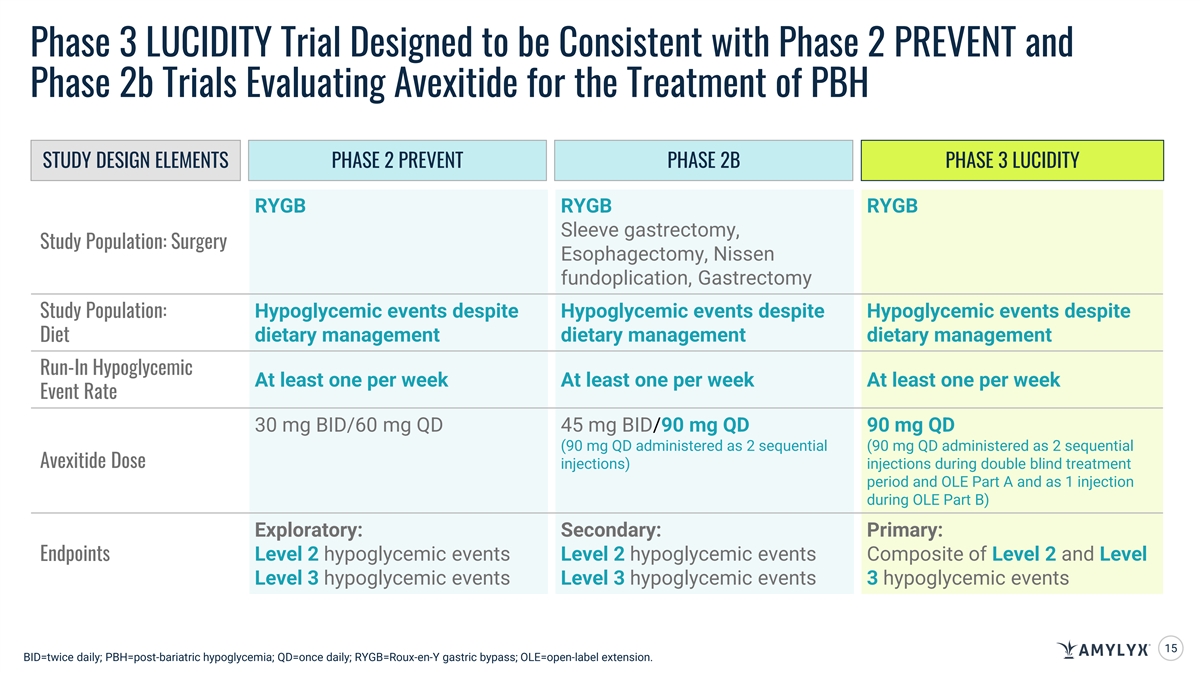
Phase 3 LUCIDITY Trial Designed to be Consistent with Phase 2 PREVENT and Phase 2b Trials Evaluating Avexitide for the Treatment of PBH STUDY DESIGN ELEMENTS PHASE 2 PREVENT PHASE 2B PHASE 3 LUCIDITY RYGB RYGB RYGB Sleeve gastrectomy, Study Population: Surgery Esophagectomy, Nissen fundoplication, Gastrectomy Hypoglycemic events despite Hypoglycemic events despite Hypoglycemic events despite Study Population: dietary management dietary management dietary management Diet Run-In Hypoglycemic At least one per week At least one per week At least one per week Event Rate 30 mg BID/60 mg QD 45 mg BID/90 mg QD 90 mg QD (90 mg QD administered as 2 sequential (90 mg QD administered as 2 sequential Avexitide Dose injections) injections during double blind treatment period and OLE Part A and as 1 injection during OLE Part B) Exploratory: Secondary: Primary: Endpoints Level 2 hypoglycemic events Level 2 hypoglycemic events Composite of Level 2 and Level Level 3 hypoglycemic events Level 3 hypoglycemic events 3 hypoglycemic events 15 BID=twice daily; PBH=post-bariatric hypoglycemia; QD=once daily; RYGB=Roux-en-Y gastric bypass; OLE=open-label extension.
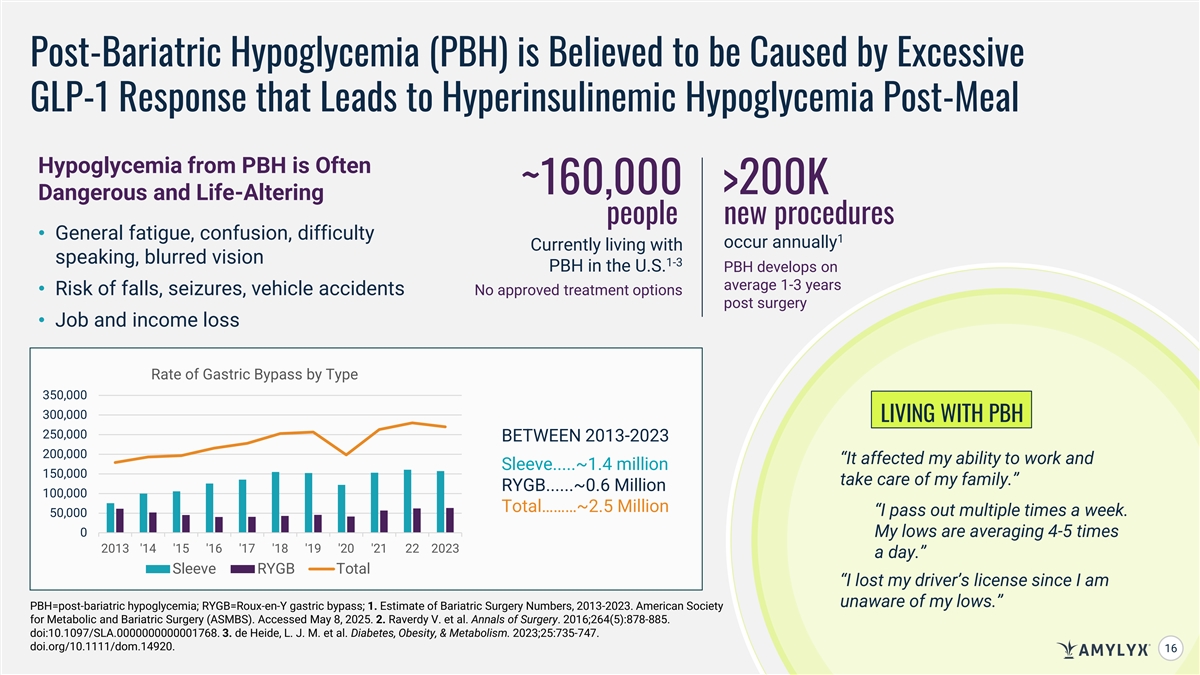
Post-Bariatric Hypoglycemia (PBH) is Believed to be Caused by Excessive GLP-1 Response that Leads to Hyperinsulinemic Hypoglycemia Post-Meal Hypoglycemia from PBH is Often ~160,000 >200K Dangerous and Life-Altering people new procedures • General fatigue, confusion, difficulty 1 occur annually Currently living with speaking, blurred vision 1-3 PBH in the U.S. PBH develops on average 1-3 years • Risk of falls, seizures, vehicle accidents No approved treatment options post surgery • Job and income loss Rate of Gastric Bypass by Type 350,000 300,000 LIVING WITH PBH 250,000 BETWEEN 2013-2023 200,000 “It affected my ability to work and Sleeve.....~1.4 million 150,000 take care of my family.” RYGB......~0.6 Million 100,000 Total………~2.5 Million “I pass out multiple times a week. 50,000 0 My lows are averaging 4-5 times 2013 '14 '15 '16 '17 '18 '19 '20 '21 22 2023 a day.” Sleeve RYGB Total “I lost my driver’s license since I am unaware of my lows.” PBH=post-bariatric hypoglycemia; RYGB=Roux-en-Y gastric bypass; 1. Estimate of Bariatric Surgery Numbers, 2013-2023. American Society for Metabolic and Bariatric Surgery (ASMBS). Accessed May 8, 2025. 2. Raverdy V. et al. Annals of Surgery. 2016;264(5):878-885. doi:10.1097/SLA.0000000000001768. 3. de Heide, L. J. M. et al. Diabetes, Obesity, & Metabolism. 2023;25:735-747. doi.org/10.1111/dom.14920. 16

Source Finding 2.5M bariatric surgeries of which 2M were either sleeve gastrectomy ~160K People Live With PBH in U.S. 1a ASMBS Registry of Bariatric Surgery (SG) or Roux-en-Y gastric bypass (RYGB) 56.1% prevalence of hypoglycemia in studies specifically examining 2 8-Study Meta-Analysis (N=280) RYGB and 54.3% in those examining SG 3 Prospective, Longitudinal Cohort (N=1,448) 43.2% prevalence of post-RYGB hypoglycemia symptoms ~2 Million from 2013-2023 Total U.S. Bariatric Surgeries 32.8% of participants had at least one OGTT (oral glucose tolerance 4 Prospective 1-Year Study (N=186) test)-related hypoglycemic event after laparoscopic sleeve gastrectomy 32.6% showed post-challenge hypoglycemia after RYGB; 5 Prospective 2-Year Study (N=281) ~20- 22.6% after SG Prevalence of Hypoglycemia ~400K – 1M 50% 6 Retrospective Survey (N=341) 29% with new-onset hypoglycemia symptoms post-RYGB or SG Following Bariatric Surgery Of 107 individuals with PBH treated with acarbose, 37% had 7 persistent/unacceptable frequency of hypoglycemic events Retrospective Study (N=120) b [Note: Equates to ~8-16% of total bariatric surgery population ] ~160K PBH Prevalence ~8% 13.1% met criteria for PBH 5 years post-op and 5% 8 Retrospective Study (N=1,206) c of those with PBH had severe symptoms 9 Prospective, Longitudinal Cohort (N=177) 7.9% met criteria for PBH 5-years post-RYGB Symptoms of PBH requiring hospitalization or ER visit occurred in 2.6- 3 Prospective, Longitudinal Cohort (N=1,448) 3.6% of people who underwent RYGB after 5 years Additional Corroborating Data 1 2 Incidence Prevalence 12d Preliminary data from Data from a Recent Academic Review : RYGB SG RYGB SG Total multiple industry- Any PBH leading medical claims • >3 hypoglycemia symptoms providers align with • Neuroglycopenia 27.3% 10.3% 275,717 111,370 387,087 current literature • Need for assistance or • Glucose ≤54 mg/Dl understanding of ~160K 10 prevalent population Medically-Important PBH 11.8% 4.4% 118,843 48,026 166,869 Visit to inpatient or outpatient facility a b c The ASMBS total bariatric procedure numbers are based on the best estimation from available data (BOLD,ACS/MBSAQIP, National Inpatient Sample Data and outpatient estimations) Assumes 20-40% post-surgical hypoglycemic symptom prevalence; Severe symptoms defined as glucose <40 mg/dL or d emergency room/hospital visit; Analysis incorporated historical census data dating back to 1993, life expectancy estimates, and disease-state modeling to inform projections 17 1.C Eonf stim id ae te ntial of B. aF ror i iatric nte Su rnal rgeryp N urp um ose bers s,only 2013 . -2023. American Society for Metabolic and Bariatric Surgery (ASMBS). Accessed May 8, 2025. 2. Lupoli R, et al. Nutr Metab Cardiovasc Dis. 2022;32(1):32-39. 3. Fischer LE, et al. Surg Obes Relat Dis.2021;17(10):1787-1798. 4. Belilgoli A, et al. Obes Surg. 2017;27:3179-3186 797-802.5. Brix JM, et al. Obes Facts. 2019;12:397-406. 6. Lee CJ, et al. Surg Obes and Relat Dis. 2018;14(6):797-802. 7. de Heide LJM, et al. Diabetes Obes Metab.2023;25:735-747.8. Lee CJ, et al. Obesity.2016.24(6):1342-1348. 9. Raverdy V, et al. Annals Surg. 2016.264(5):878-885.10. Data on file. 11. Hazelhurst J, et al. Endocr Connect. 2024;13(5):e230285. 12. McLaughlin, T et al. (2025, July 12-15). Prevalence of Post-bariatric Hypoglycemia in the United States, [Poster presentation]. ENDO 2025.
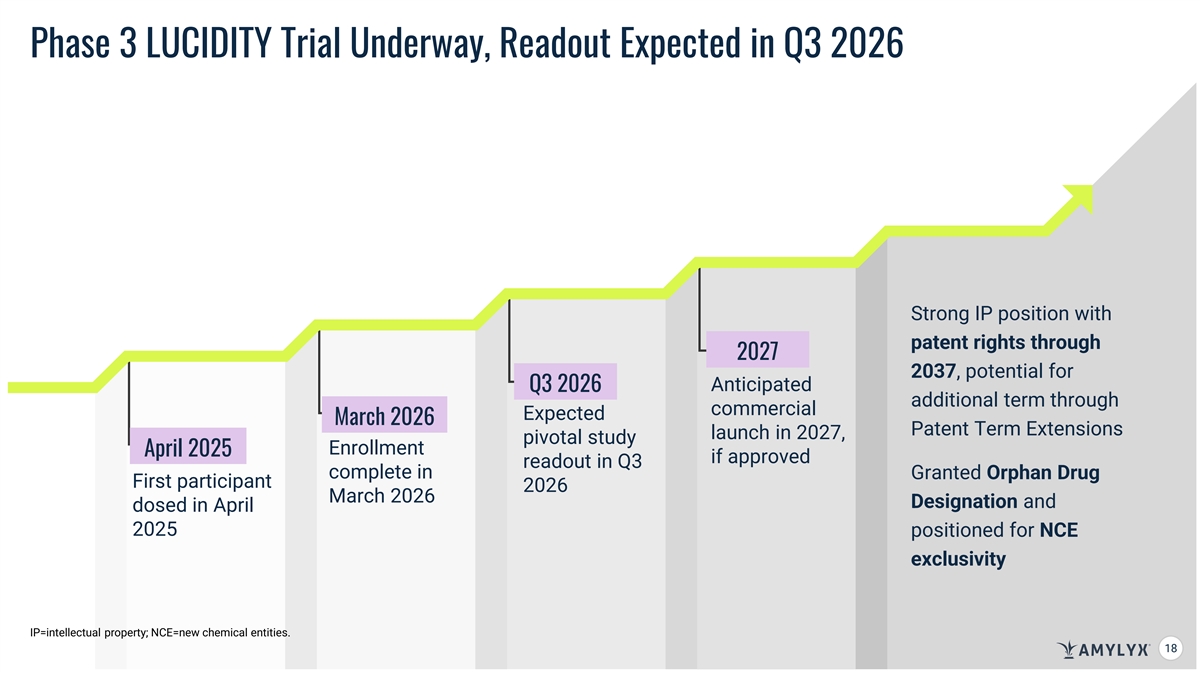
Phase 3 LUCIDITY Trial Underway, Readout Expected in Q3 2026 Strong IP position with patent rights through 2027 2037, potential for Anticipated Q3 2026 additional term through commercial Expected March 2026 Patent Term Extensions launch in 2027, pivotal study Enrollment April 2025 if approved readout in Q3 complete in Granted Orphan Drug First participant 2026 March 2026 Designation and dosed in April 2025 positioned for NCE exclusivity IP=intellectual property; NCE=new chemical entities. 18 18

AMX0035 Fixed-Dose Combination of Sodium Phenylbutyrate and Taurursodiol Designed to Slow or Mitigate Neurodegeneration Raquel, a college student and martial artist living with Wolfram syndrome. Retinal ganglion cells
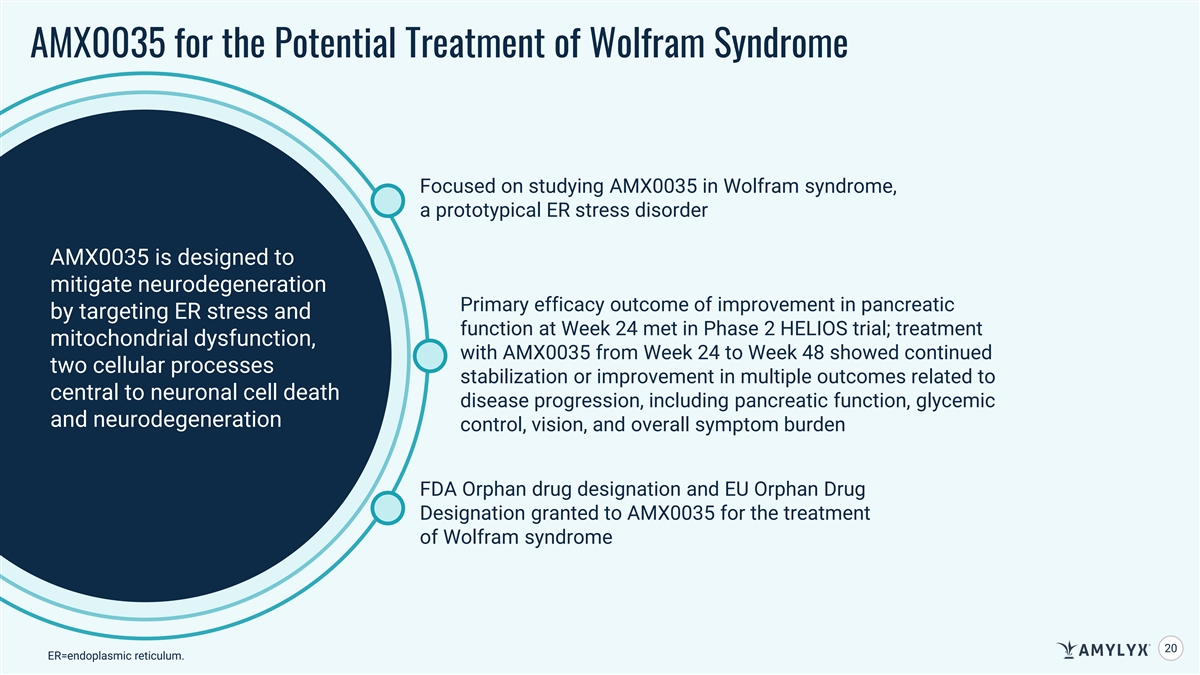
AMX0035 for the Potential Treatment of Wolfram Syndrome Focused on studying AMX0035 in Wolfram syndrome, a prototypical ER stress disorder AMX0035 is designed to mitigate neurodegeneration Primary efficacy outcome of improvement in pancreatic by targeting ER stress and function at Week 24 met in Phase 2 HELIOS trial; treatment mitochondrial dysfunction, with AMX0035 from Week 24 to Week 48 showed continued two cellular processes stabilization or improvement in multiple outcomes related to central to neuronal cell death disease progression, including pancreatic function, glycemic and neurodegeneration control, vision, and overall symptom burden FDA Orphan drug designation and EU Orphan Drug Designation granted to AMX0035 for the treatment of Wolfram syndrome 20 ER=endoplasmic reticulum.
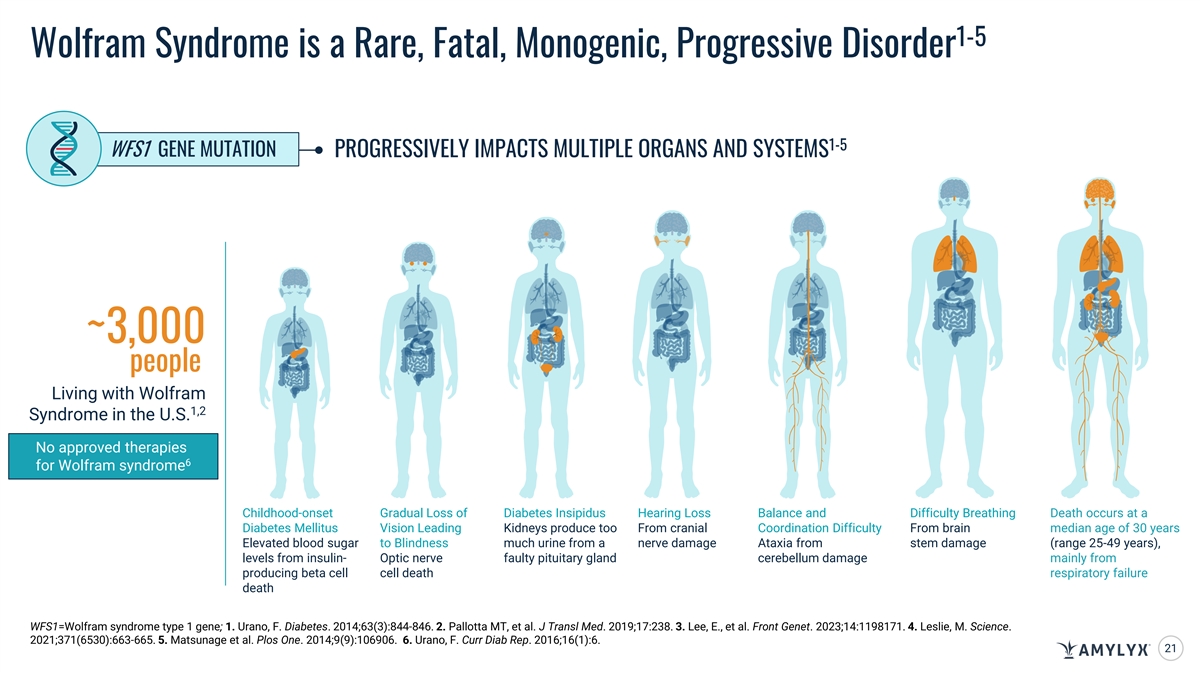
1-5 Wolfram Syndrome is a Rare, Fatal, Monogenic, Progressive Disorder 1-5 WFS1 GENE MUTATION PROGRESSIVELY IMPACTS MULTIPLE ORGANS AND SYSTEMS ~3,000 people Living with Wolfram 1,2 Syndrome in the U.S. No approved therapies 6 for Wolfram syndrome Childhood-onset Gradual Loss of Diabetes Insipidus Hearing Loss Balance and Difficulty Breathing Death occurs at a Diabetes Mellitus Vision Leading Kidneys produce too From cranial Coordination Difficulty From brain median age of 30 years Elevated blood sugar to Blindness much urine from a nerve damage Ataxia from stem damage (range 25-49 years), levels from insulin- Optic nerve faulty pituitary gland cerebellum damage mainly from producing beta cell cell death respiratory failure death WFS1=Wolfram syndrome type 1 gene; 1. Urano, F. Diabetes. 2014;63(3):844-846. 2. Pallotta MT, et al. J Transl Med. 2019;17:238. 3. Lee, E., et al. Front Genet. 2023;14:1198171. 4. Leslie, M. Science. 2021;371(6530):663-665. 5. Matsunage et al. Plos One. 2014;9(9):106906. 6. Urano, F. Curr Diab Rep. 2016;16(1):6. 21
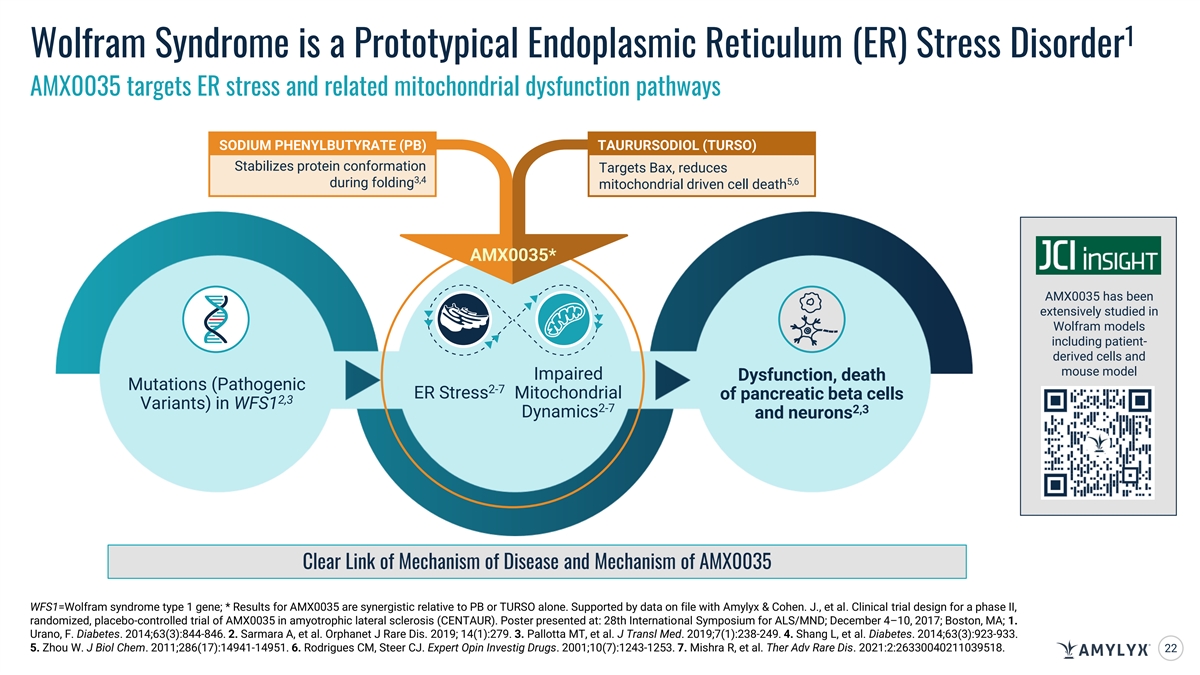
1 Wolfram Syndrome is a Prototypical Endoplasmic Reticulum (ER) Stress Disorder AMX0035 targets ER stress and related mitochondrial dysfunction pathways SODIUM PHENYLBUTYRATE (PB) TAURURSODIOL (TURSO) Stabilizes protein conformation Targets Bax, reduces 3,4 5,6 during folding mitochondrial driven cell death AMX0035* AMX0035 has been extensively studied in Wolfram models including patient- derived cells and mouse model Impaired Dysfunction, death Mutations (Pathogenic 2-7 ER Stress Mitochondrial of pancreatic beta cells 2,3 Variants) in WFS1 2-7 2,3 Dynamics and neurons Clear Link of Mechanism of Disease and Mechanism of AMX0035 WFS1=Wolfram syndrome type 1 gene; * Results for AMX0035 are synergistic relative to PB or TURSO alone. Supported by data on file with Amylyx & Cohen. J., et al. Clinical trial design for a phase II, randomized, placebo-controlled trial of AMX0035 in amyotrophic lateral sclerosis (CENTAUR). Poster presented at: 28th International Symposium for ALS/MND; December 4–10, 2017; Boston, MA; 1. Urano, F. Diabetes. 2014;63(3):844-846. 2. Sarmara A, et al. Orphanet J Rare Dis. 2019; 14(1):279. 3. Pallotta MT, et al. J Transl Med. 2019;7(1):238-249. 4. Shang L, et al. Diabetes. 2014;63(3):923-933. 5. Zhou W. J Biol Chem. 2011;286(17):14941-14951. 6. Rodrigues CM, Steer CJ. Expert Opin Investig Drugs. 2001;10(7):1243-1253. 7. Mishra R, et al. Ther Adv Rare Dis. 2021:2:26330040211039518. 22
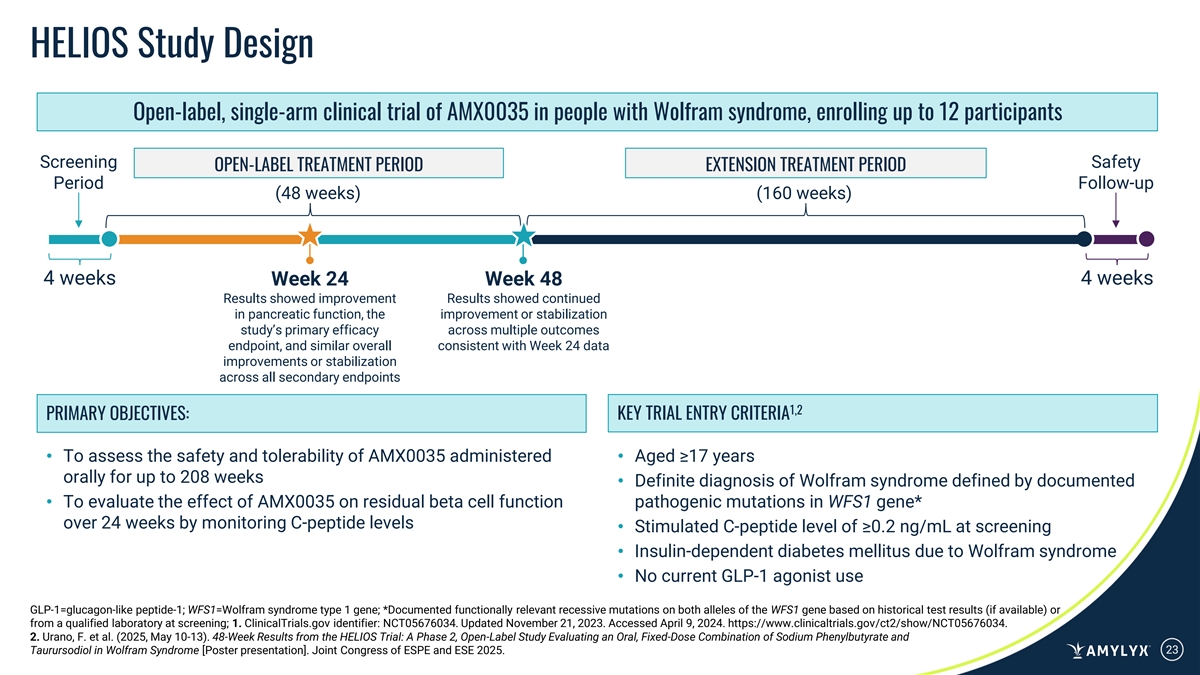
HELIOS Study Design Open-label, single-arm clinical trial of AMX0035 in people with Wolfram syndrome, enrolling up to 12 participants Screening Safety OPEN-LABEL TREATMENT PERIOD EXTENSION TREATMENT PERIOD Period Follow-up (48 weeks) (160 weeks) 4 weeks 4 weeks Week 24 Week 48 Results showed improvement Results showed continued in pancreatic function, the improvement or stabilization study’s primary efficacy across multiple outcomes endpoint, and similar overall consistent with Week 24 data improvements or stabilization across all secondary endpoints 1,2 PRIMARY OBJECTIVES: KEY TRIAL ENTRY CRITERIA • To assess the safety and tolerability of AMX0035 administered • Aged ≥17 years orally for up to 208 weeks • Definite diagnosis of Wolfram syndrome defined by documented • To evaluate the effect of AMX0035 on residual beta cell function pathogenic mutations in WFS1 gene* over 24 weeks by monitoring C-peptide levels • Stimulated C-peptide level of ≥0.2 ng/mL at screening • Insulin-dependent diabetes mellitus due to Wolfram syndrome • No current GLP-1 agonist use GLP-1=glucagon-like peptide-1; WFS1=Wolfram syndrome type 1 gene; *Documented functionally relevant recessive mutations on both alleles of the WFS1 gene based on historical test results (if available) or from a qualified laboratory at screening; 1. ClinicalTrials.gov identifier: NCT05676034. Updated November 21, 2023. Accessed April 9, 2024. https://www.clinicaltrials.gov/ct2/show/NCT05676034. 2. Urano, F. et al. (2025, May 10-13). 48-Week Results from the HELIOS Trial: A Phase 2, Open-Label Study Evaluating an Oral, Fixed-Dose Combination of Sodium Phenylbutyrate and Taurursodiol in Wolfram Syndrome [Poster presentation]. Joint Congress of ESPE and ESE 2025. 23
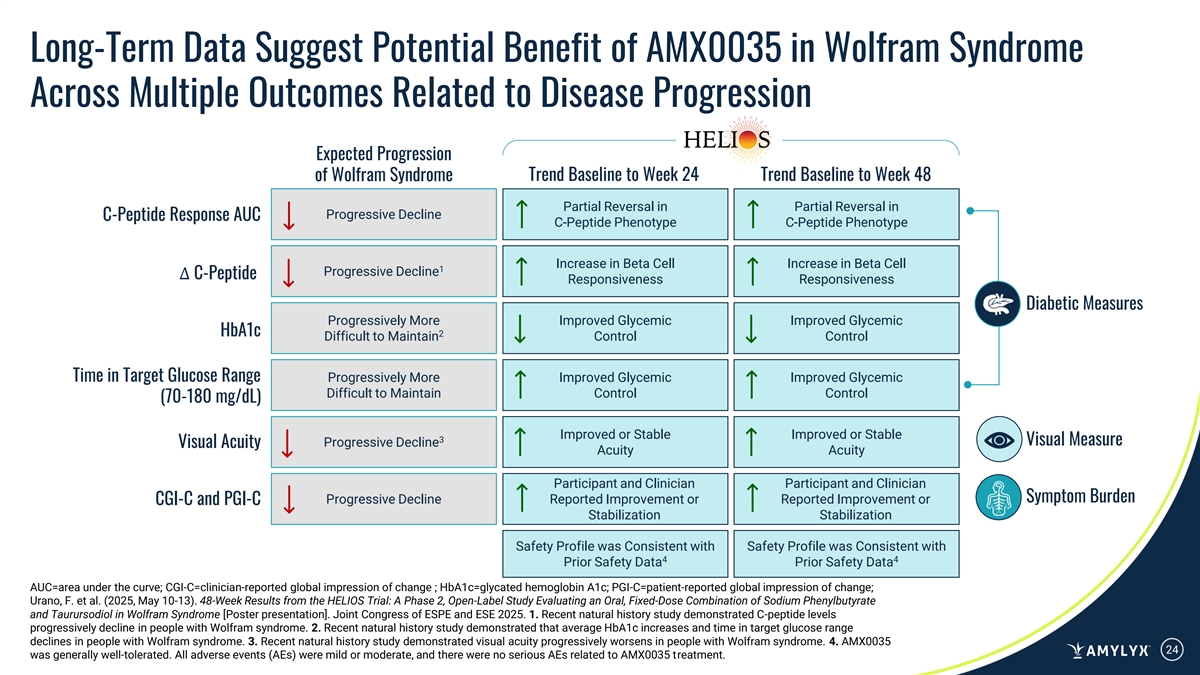
Long-Term Data Suggest Potential Benefit of AMX0035 in Wolfram Syndrome Across Multiple Outcomes Related to Disease Progression Expected Progression of Wolfram Syndrome Trend Baseline to Week 24 Trend Baseline to Week 48 Partial Reversal in Partial Reversal in Progressive Decline C-Peptide Response AUC C-Peptide Phenotype C-Peptide Phenotype Increase in Beta Cell Increase in Beta Cell 1 Progressive Decline ∆ C-Peptide Responsiveness Responsiveness Diabetic Measures Progressively More Improved Glycemic Improved Glycemic HbA1c 2 Difficult to Maintain Control Control Time in Target Glucose Range Progressively More Improved Glycemic Improved Glycemic Difficult to Maintain Control Control (70-180 mg/dL) Improved or Stable Improved or Stable 3 Visual Measure Progressive Decline Visual Acuity Acuity Acuity Participant and Clinician Participant and Clinician Symptom Burden Progressive Decline Reported Improvement or Reported Improvement or CGI-C and PGI-C Stabilization Stabilization Safety Profile was Consistent with Safety Profile was Consistent with 4 4 Prior Safety Data Prior Safety Data AUC=area under the curve; CGI-C=clinician-reported global impression of change ; HbA1c=glycated hemoglobin A1c; PGI-C=patient-reported global impression of change; Urano, F. et al. (2025, May 10-13). 48-Week Results from the HELIOS Trial: A Phase 2, Open-Label Study Evaluating an Oral, Fixed-Dose Combination of Sodium Phenylbutyrate and Taurursodiol in Wolfram Syndrome [Poster presentation]. Joint Congress of ESPE and ESE 2025. 1. Recent natural history study demonstrated C-peptide levels progressively decline in people with Wolfram syndrome. 2. Recent natural history study demonstrated that average HbA1c increases and time in target glucose range declines in people with Wolfram syndrome. 3. Recent natural history study demonstrated visual acuity progressively worsens in people with Wolfram syndrome. 4. AMX0035 24 was generally well-tolerated. All adverse events (AEs) were mild or moderate, and there were no serious AEs related to AMX0035 treatment.
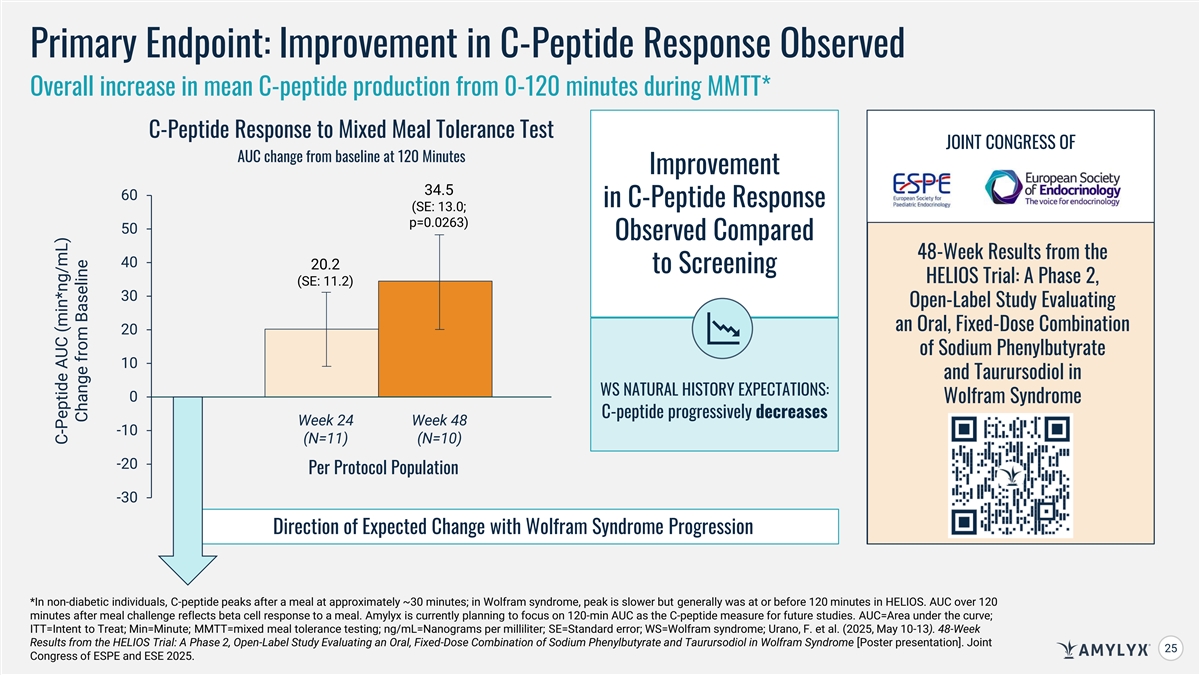
Primary Endpoint: Improvement in C-Peptide Response Observed Overall increase in mean C-peptide production from 0-120 minutes during MMTT* C-Peptide Response to Mixed Meal Tolerance Test JOINT CONGRESS OF AUC change from baseline at 120 Minutes Improvement 34.5 & 60 in C-Peptide Response (SE: 13.0; p=0.0263) 50 Observed Compared 48-Week Results from the 40 20.2 to Screening HELIOS Trial: A Phase 2, (SE: 11.2) 30 Open-Label Study Evaluating an Oral, Fixed-Dose Combination 20 of Sodium Phenylbutyrate 10 and Taurursodiol in WS NATURAL HISTORY EXPECTATIONS: 0 Wolfram Syndrome C-peptide progressively decreases Week 24 Week 48 -10 (N=11) (N=10) -20 Per Protocol Population -30 Direction of Expected Change with Wolfram Syndrome Progression *In non-diabetic individuals, C-peptide peaks after a meal at approximately ~30 minutes; in Wolfram syndrome, peak is slower but generally was at or before 120 minutes in HELIOS. AUC over 120 minutes after meal challenge reflects beta cell response to a meal. Amylyx is currently planning to focus on 120-min AUC as the C-peptide measure for future studies. AUC=Area under the curve; ITT=Intent to Treat; Min=Minute; MMTT=mixed meal tolerance testing; ng/mL=Nanograms per milliliter; SE=Standard error; WS=Wolfram syndrome; Urano, F. et al. (2025, May 10-13). 48-Week Results from the HELIOS Trial: A Phase 2, Open-Label Study Evaluating an Oral, Fixed-Dose Combination of Sodium Phenylbutyrate and Taurursodiol in Wolfram Syndrome [Poster presentation]. Joint 25 25 Congress of ESPE and ESE 2025. C-Peptide AUC (min*ng/mL) Change from Baseline
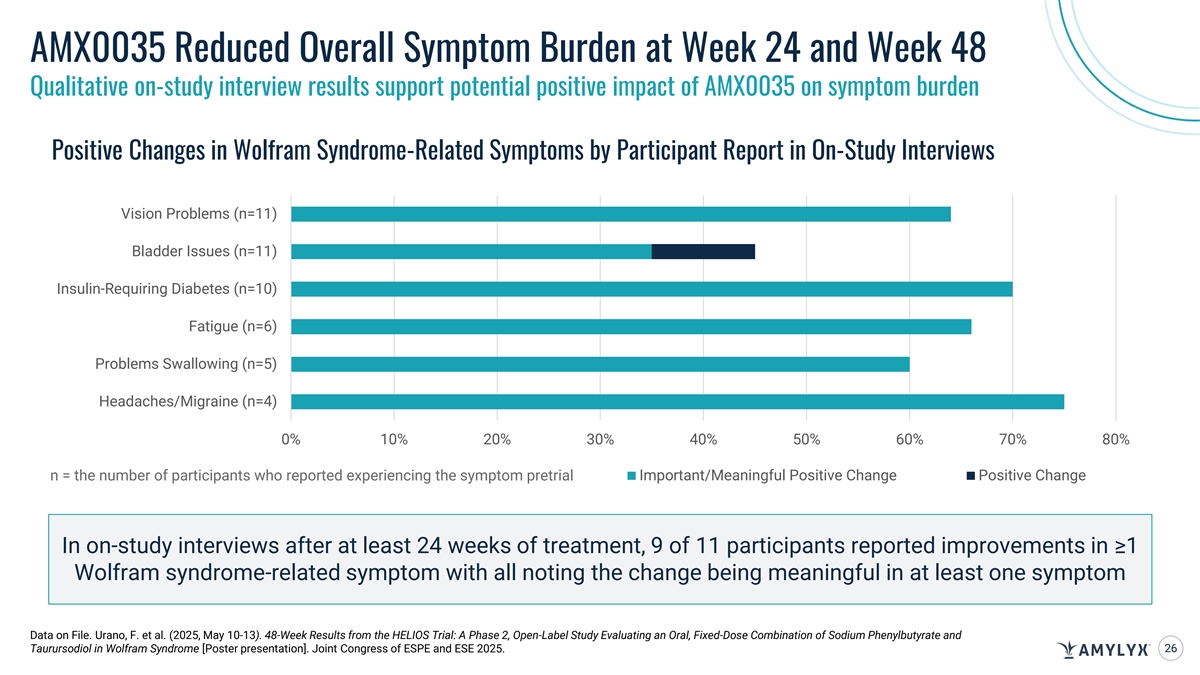
AMX0035 Reduced Overall Symptom Burden at Week 24 and Week 48 Qualitative on-study interview results support potential positive impact of AMX0035 on symptom burden Positive Changes in Wolfram Syndrome-Related Symptoms by Participant Report in On-Study Interviews Vision Problems (n=11) Bladder Issues (n=11) Insulin-Requiring Diabetes (n=10) Fatigue (n=6) Problems Swallowing (n=5) Headaches/Migraine (n=4) 0% 10% 20% 30% 40% 50% 60% 70% 80% Important/Meaningful Positive Change Positive Change n = the number of participants who reported experiencing the symptom pretrial In on-study interviews after at least 24 weeks of treatment, 9 of 11 participants reported improvements in ≥1 Wolfram syndrome-related symptom with all noting the change being meaningful in at least one symptom Data on File. Urano, F. et al. (2025, May 10-13). 48-Week Results from the HELIOS Trial: A Phase 2, Open-Label Study Evaluating an Oral, Fixed-Dose Combination of Sodium Phenylbutyrate and Taurursodiol in Wolfram Syndrome [Poster presentation]. Joint Congress of ESPE and ESE 2025. 26

AMX0035 Safety and Tolerability in HELIOS Summary of Treatment Emergent Adverse Events (TEAEs) AMX0035 • AMX0035 was generally well tolerated (N=12)* n (%) > Diarrhea was the most common TEAE (58.3%); all cases were of mild severity Participants with ≥1 TEAE 11 (91.7) > All TEAEs were graded mild or moderate TEAE related to study drug** 10 (83.3) • No new safety signals were identified • Nearly all participants reported ≥1 TEAE during Serious adverse events 0 (0) the trial Drug interrupted owing to TEAE 3 (25.0) > Most did not lead to modification or interruption of AMX0035 dosing and none Dose reduced owing to TEAE 3 (25.0) led to drug discontinuation Drug discontinued owing to TEAE 0 (0) *All available safety data as of January 10, 2025 included **Includes those with TEAEs considered at least possibly related to treatment Urano, F. et al. (2025, May 10-13). 48-Week Results from the HELIOS Trial: A Phase 2, Open-Label Study Evaluating an Oral, Fixed-Dose Combination of Sodium Phenylbutyrate and Taurursodiol in Wolfram Syndrome [Poster presentation]. Joint Congress of ESPE and ESE 2025. 27

AMX0114 Potent Antisense Oligonucleotide (ASO) Targeting Calpain-2 In memory of Mick, a husband and father, who was a gifted tattoo artist and musician. Nerve cells in the brain
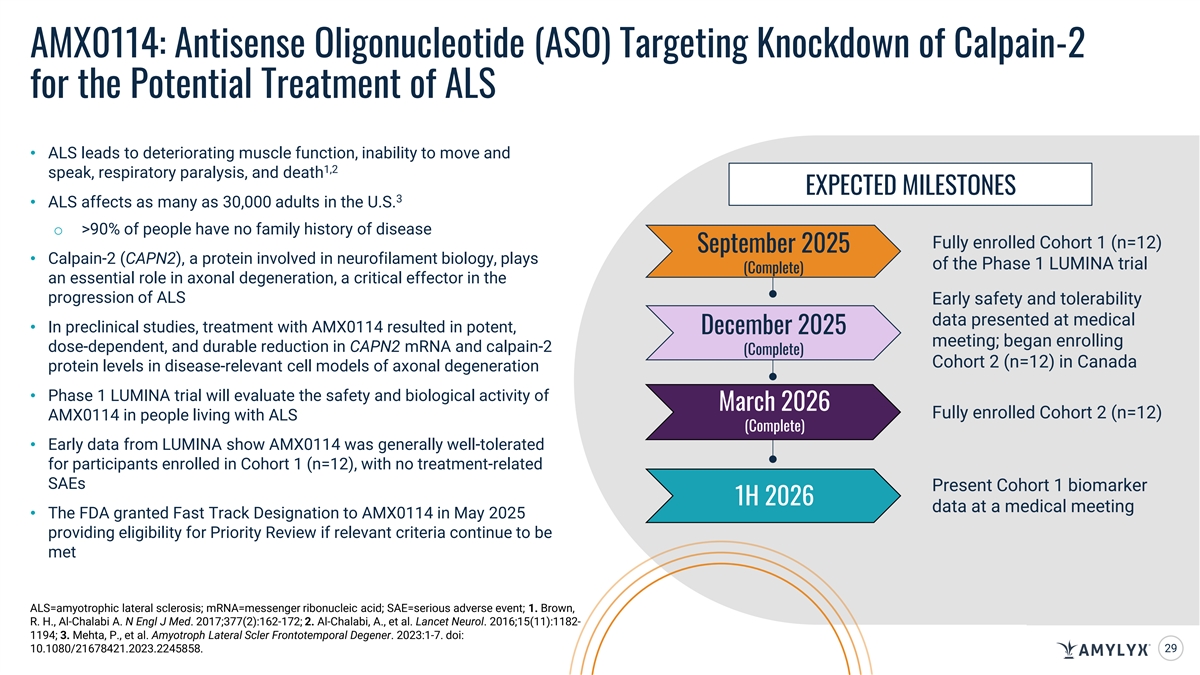
AMX0114: Antisense Oligonucleotide (ASO) Targeting Knockdown of Calpain-2 for the Potential Treatment of ALS • ALS leads to deteriorating muscle function, inability to move and 1,2 speak, respiratory paralysis, and death EXPECTED MILESTONES 3 • ALS affects as many as 30,000 adults in the U.S. o >90% of people have no family history of disease Fully enrolled Cohort 1 (n=12) September 2025 • Calpain-2 (CAPN2), a protein involved in neurofilament biology, plays of the Phase 1 LUMINA trial (Complete) an essential role in axonal degeneration, a critical effector in the progression of ALS Early safety and tolerability data presented at medical • In preclinical studies, treatment with AMX0114 resulted in potent, December 2025 meeting; began enrolling dose-dependent, and durable reduction in CAPN2 mRNA and calpain-2 (Complete) Cohort 2 (n=12) in Canada protein levels in disease-relevant cell models of axonal degeneration • Phase 1 LUMINA trial will evaluate the safety and biological activity of March 2026 Fully enrolled Cohort 2 (n=12) AMX0114 in people living with ALS (Complete) • Early data from LUMINA show AMX0114 was generally well-tolerated for participants enrolled in Cohort 1 (n=12), with no treatment-related SAEs Present Cohort 1 biomarker 1H 2026 data at a medical meeting • The FDA granted Fast Track Designation to AMX0114 in May 2025 providing eligibility for Priority Review if relevant criteria continue to be met ALS=amyotrophic lateral sclerosis; mRNA=messenger ribonucleic acid; SAE=serious adverse event; 1. Brown, R. H., Al-Chalabi A. N Engl J Med. 2017;377(2):162-172; 2. Al-Chalabi, A., et al. Lancet Neurol. 2016;15(11):1182- 1194; 3. Mehta, P., et al. Amyotroph Lateral Scler Frontotemporal Degener. 2023:1-7. doi: 10.1080/21678421.2023.2245858. 29
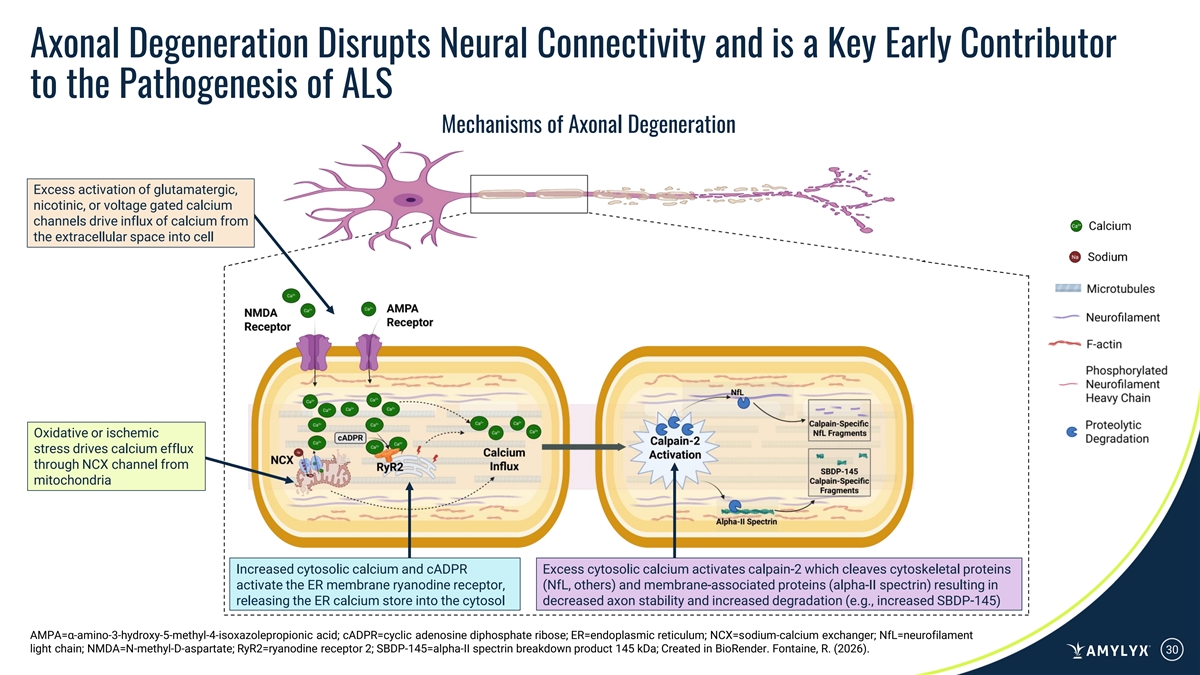
Axonal Degeneration Disrupts Neural Connectivity and is a Key Early Contributor to the Pathogenesis of ALS Mechanisms of Axonal Degeneration Excess activation of glutamatergic, nicotinic, or voltage gated calcium channels drive influx of calcium from the extracellular space into cell Oxidative or ischemic stress drives calcium efflux through NCX channel from mitochondria Increased cytosolic calcium and cADPR Excess cytosolic calcium activates calpain-2 which cleaves cytoskeletal proteins activate the ER membrane ryanodine receptor, (NfL, others) and membrane-associated proteins (alpha-II spectrin) resulting in releasing the ER calcium store into the cytosol decreased axon stability and increased degradation (e.g., increased SBDP-145) AMPA=α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid; cADPR=cyclic adenosine diphosphate ribose; ER=endoplasmic reticulum; NCX=sodium-calcium exchanger; NfL=neurofilament light chain; NMDA=N-methyl-D-aspartate; RyR2=ryanodine receptor 2; SBDP-145=alpha-II spectrin breakdown product 145 kDa; Created in BioRender. Fontaine, R. (2026). 30
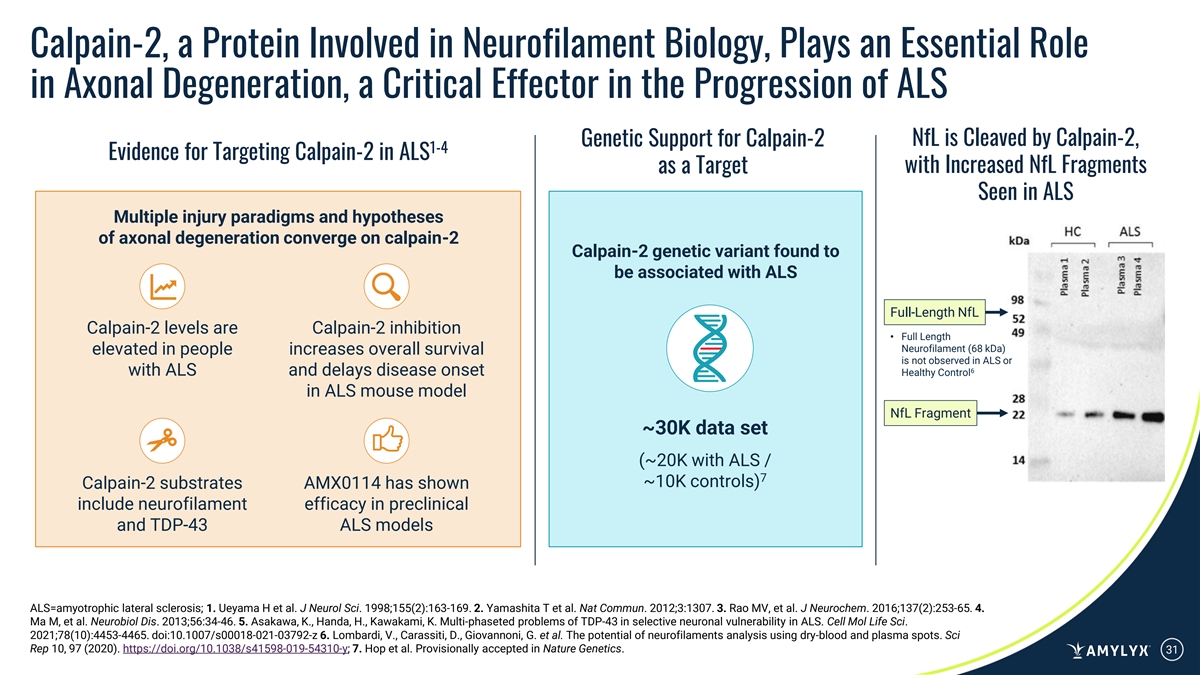
Calpain-2, a Protein Involved in Neurofilament Biology, Plays an Essential Role in Axonal Degeneration, a Critical Effector in the Progression of ALS Genetic Support for Calpain-2 NfL is Cleaved by Calpain-2, 1-4 Evidence for Targeting Calpain-2 in ALS as a Target with Increased NfL Fragments Seen in ALS Multiple injury paradigms and hypotheses of axonal degeneration converge on calpain-2 Calpain-2 genetic variant found to be associated with ALS Full-Length NfL Calpain-2 levels are Calpain-2 inhibition • Full Length Neurofilament (68 kDa) elevated in people increases overall survival is not observed in ALS or 6 with ALS and delays disease onset Healthy Control in ALS mouse model NfL Fragment ~30K data set (~20K with ALS / 7 ~10K controls) Calpain-2 substrates AMX0114 has shown include neurofilament efficacy in preclinical and TDP-43 ALS models ALS=amyotrophic lateral sclerosis; 1. Ueyama H et al. J Neurol Sci. 1998;155(2):163-169. 2. Yamashita T et al. Nat Commun. 2012;3:1307. 3. Rao MV, et al. J Neurochem. 2016;137(2):253-65. 4. Ma M, et al. Neurobiol Dis. 2013;56:34-46. 5. Asakawa, K., Handa, H., Kawakami, K. Multi-phaseted problems of TDP-43 in selective neuronal vulnerability in ALS. Cell Mol Life Sci. 2021;78(10):4453-4465. doi:10.1007/s00018-021-03792-z 6. Lombardi, V., Carassiti, D., Giovannoni, G. et al. The potential of neurofilaments analysis using dry-blood and plasma spots. Sci Rep 10, 97 (2020). https://doi.org/10.1038/s41598-019-54310-y; 7. Hop et al. Provisionally accepted in Nature Genetics. 31
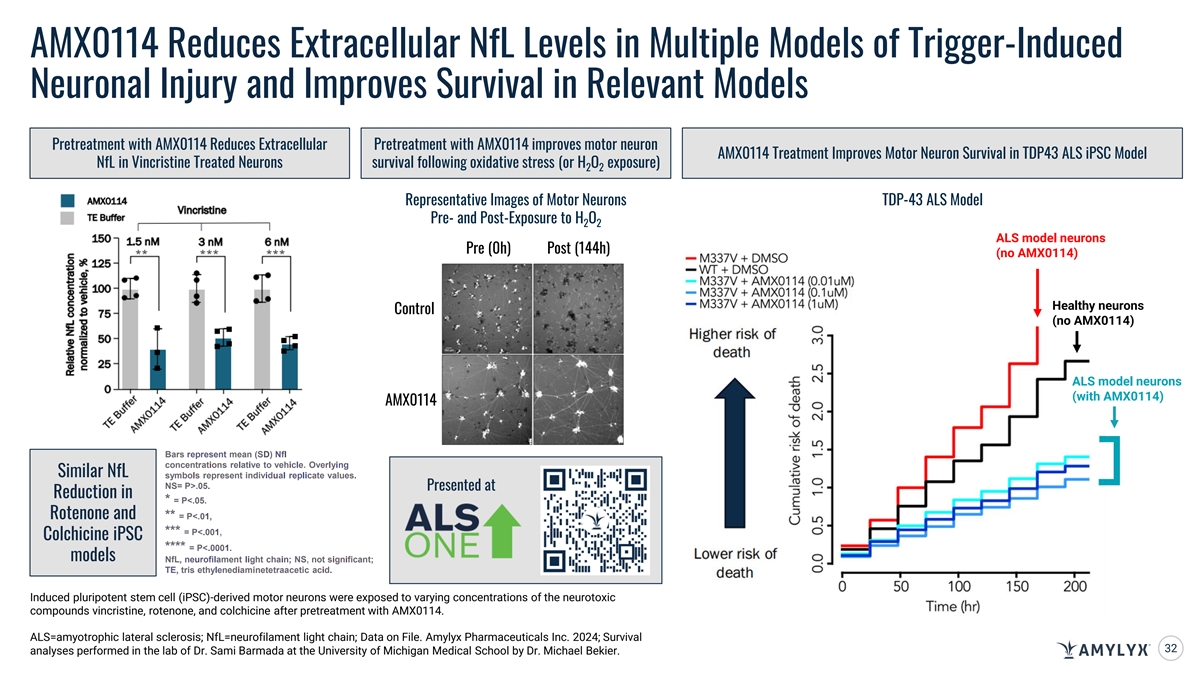
AMX0114 Reduces Extracellular NfL Levels in Multiple Models of Trigger-Induced Neuronal Injury and Improves Survival in Relevant Models Pretreatment with AMX0114 Reduces Extracellular Pretreatment with AMX0114 improves motor neuron AMX0114 Treatment Improves Motor Neuron Survival in TDP43 ALS iPSC Model NfL in Vincristine Treated Neurons survival following oxidative stress (or H O exposure) 2 2 Representative Images of Motor Neurons TDP-43 ALS Model Pre- and Post-Exposure to H O 2 2 ALS model neurons Pre (0h) Post (144h) (no AMX0114) Healthy neurons Control (no AMX0114) ALS model neurons (with AMX0114) AMX0114 Bars represent mean (SD) Nfl concentrations relative to vehicle. Overlying Similar NfL symbols represent individuaI repIicate values. NS= P>.05. Presented at Reduction in * = P<.05. Rotenone and ** = P<.01, *** = P<.001, Colchicine iPSC **** = P<.0001. models NfL, neurofilament light chain; NS, not significant; TE, tris ethylenediaminetetraacetic acid. Induced pluripotent stem cell (iPSC)-derived motor neurons were exposed to varying concentrations of the neurotoxic compounds vincristine, rotenone, and colchicine after pretreatment with AMX0114. ALS=amyotrophic lateral sclerosis; NfL=neurofilament light chain; Data on File. Amylyx Pharmaceuticals Inc. 2024; Survival 32 analyses performed in the lab of Dr. Sami Barmada at the University of Michigan Medical School by Dr. Michael Bekier.
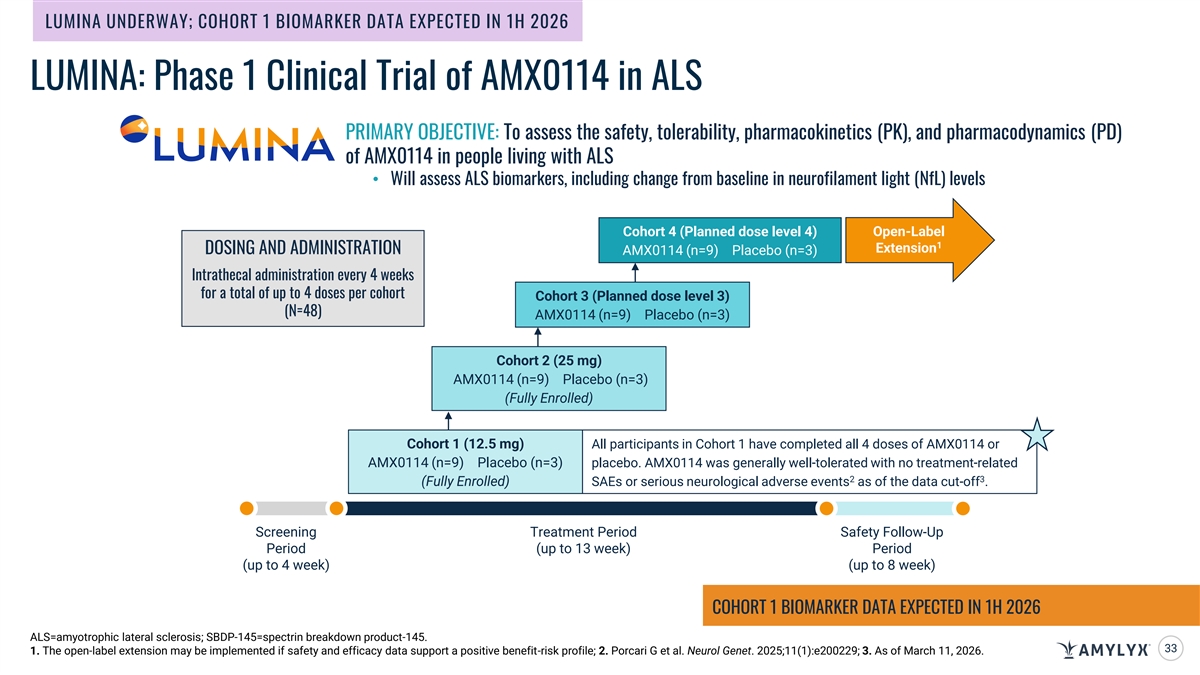
LUMINA UNDERWAY; COHORT 1 BIOMARKER DATA EXPECTED IN 1H 2026 LUMINA: Phase 1 Clinical Trial of AMX0114 in ALS PRIMARY OBJECTIVE: To assess the safety, tolerability, pharmacokinetics (PK), and pharmacodynamics (PD) of AMX0114 in people living with ALS • Will assess ALS biomarkers, including change from baseline in neurofilament light (NfL) levels Cohort 4 (Planned dose level 4) Open-Label 1 Extension DOSING AND ADMINISTRATION AMX0114 (n=9) Placebo (n=3) Intrathecal administration every 4 weeks for a total of up to 4 doses per cohort Cohort 3 (Planned dose level 3) (N=48) AMX0114 (n=9) Placebo (n=3) Cohort 2 (25 mg) AMX0114 (n=9) Placebo (n=3) (Fully Enrolled) Cohort 1 (12.5 mg) All participants in Cohort 1 have completed all 4 doses of AMX0114 or AMX0114 (n=9) Placebo (n=3) placebo. AMX0114 was generally well-tolerated with no treatment-related 2 3 (Fully Enrolled) SAEs or serious neurological adverse events as of the data cut-off . Screening Treatment Period Safety Follow-Up Period (up to 13 week) Period (up to 4 week) (up to 8 week) COHORT 1 BIOMARKER DATA EXPECTED IN 1H 2026 ALS=amyotrophic lateral sclerosis; SBDP-145=spectrin breakdown product-145. 33 1. The open-label extension may be implemented if safety and efficacy data support a positive benefit-risk profile; 2. Porcari G et al. Neurol Genet. 2025;11(1):e200229; 3. As of March 11, 2026.

AMX0318 Novel, Long-Acting GLP-1 Receptor Antagonist
AMX0318: A Novel, Long-Acting Glucagon-Like Peptide-1 (GLP-1) Receptor Antagonist • Entered into a collaboration with Gubra A/S in December 2024 to develop a long-acting GLP-1 receptor antagonist and in January 2026, AMX0318 was nominated as a EXPECTED MILESTONES development candidate • AMX0318 completed an extensive preclinical evaluation in collaboration with Gubra A/S Initiated IND-enabling 2026 • In preclinical studies, AMX0318 met key preclinical criteria studies (Complete) for a long-acting GLP-1 receptor antagonist, including: > Robust chemical stability profile > High solubility Submission of IND, pending successful completion of 2027 > Strong in vitro potency IND-enabling studies > Evidence of in vivo activity and tolerability > Favorable pharmacokinetic profile consistent with a long-acting peptide IND=investigational new drug. 35

Key Corporate Highlights

Advancing Novel Therapies for Diseases with High Unmet Needs Expected Upcoming Milestones Focus on diseases with well-defined mechanistic Phase 3 LUCIDITY topline data Q3 2026 Avexitide PBH Clinical anticipated in Q3 2026 rationale, clear clinical outcomes and biomarkers, and rigorous preclinical data Commercial launch, if approved, 2027 Avexitide PBH Regulatory anticipated in 2027 First-in-class GLP-1 receptor antagonist with FDA Breakthrough Therapy and Orphan Drug AVEXITIDE designations Phase 1 LUMINA trial Cohort 1 (n=12) biomarker data to be presented at a 1H 2026 AMX0114 ALS Clinical Oral, fixed-dose combination of two medical meeting expected in 1H 2026 small molecules, sodium AMX0035 phenylbutyrate and taurursodiol PBH & Rare IND-enabling studies underway with an Antisense oligonucleotide designed to 2027 AMX0318 Preclinical target calpain-2, a protein involved in IND filing targeted for 2027 Diseases AMX0114 axonal degeneration & neurofilament biology with FDA Fast Track Designation Novel, long-acting GLP-1 AMX0318 receptor antagonist ALS=amyotrophic lateral sclerosis; GLP-1=glucagon-like peptide-1; 37 PBH=post-bariatric hypoglycemia.

Avexitide, AMX0035, and AMX0114 are Protected by Robust Global IP Portfolio AVEXITIDE AMX0035 AMX0114 granted patents and over granted patents and over Pending composition of matter 40 pending applications 50 pending applications patent provides potential patent >80 >200 worldwide* worldwide term through 2043 if granted • Granted US patent rights through • Granted US patent rights • Positioned for NCE 2037** through 2040 exclusivity • Positioned for NCE exclusivity• Granted Orphan Drug Designation for the treatment • Granted Orphan Drug of Wolfram syndrome Designation for the treatment of hyperinsulinemic hypoglycemia * Includes in-licensed patents ** Additional patent term potentially available through patent term extension NCE=new chemical entities. 38

As of December 31, 2025 CASH RUNWAY EXPECTED INTO 2028 • $317M in cash, cash equivalents, and short-term investments THROUGH THE POTENTIAL APPROVAL • ~110M shares outstanding AND LAUNCH OF AVEXITIDE IN 2027 39

Team Leadership Team Tenured in Rare Disease with Commercial and Clinical Development Capabilities Joshua Cohen, BSE Justin Klee, ScB Jim Frates Camille L. Bedrosian, MD Co-CEO and Director Co-CEO and Director Chief Financial Officer Chief Medical Officer Co-Founded Amylyx, Co-CEO since Co-Founded Amylyx, Co-CEO since 22-year CFO at Alkermes; grew to Nearly 30 years of experience within the 2013, led preclinical, clinical and 2013, led preclinical, clinical and >$1B in annual revenue and biotech industry; Former CMO at commercial development of RELYVRIO commercial development of RELYVRIO >2,000 employees worldwide Ultragenyx, Alexion, and ARIAD as well as IPO and ~$1B in financing as well as IPO and ~$1B in financing Dan Monahan Tom Holmes Gina M. Mazzariello Linda Arsenault Chief Commercial Officer Chief Technical Operations Officer Chief Legal Officer and General Counsel Chief Human Resources Officer 20+ years of commercial leadership More than 25 years of biotech 20+ years of corporate and 25+ years of global HR experience at experience; Former commercial lead for experience; Former Head of Global commercial legal experience within multibillion-dollar life sciences and technology multiple industry-leading medicines at External Manufacturing at Biogen the healthcare industry, including at companies, including at Sumitomo Pharma Otsuka, Novartis, and Sanofi Boehringer Ingelheim America Holdings (SMPA) 40

Ushering in a New Era for Treating Diseases with High Unmet Needs