
Dianthus Therapeutics Early GO Decision in CAPTIVATE CIDP Trial March 9, 2026 .3

Forward-looking statements Certain statements in this presentation, other than purely historical information, may constitute “forward-looking statements” within the meaning of the federal securities laws, including for purposes of the safe harbor provisions under the United States Private Securities Litigation Reform Act of 1995, express or implied statements regarding future plans and prospects, including statements regarding the expectations or plans for discovery, preclinical studies, clinical trials and research and development programs, in particular with respect to claseprubart and DNTH212, and any developments or results in connection therewith, including the target product profile and administration of claseprubart and DNTH212; the anticipated timing of the initiation and results from those studies and trials; expectations regarding the clinical trial designs or indications; expectations regarding the time period over which the Company’s capital resources are expected to be sufficient to fund its anticipated operations; and expectations regarding market size, patient population size, and potential opportunities for complement therapies, in particular with respect to claseprubart and DNTH212. Claseprubart and DNTH212 are investigational agents that are not approved as therapies in any indication in any jurisdiction worldwide. The words “opportunity,” “potential,” “milestones,” “runway,” “will,” “anticipate,” “achieve,” “near-term,” “catalysts,” “pursue,” “pipeline,” “believe,” “continue,” “could,” “estimate,” “expect,” “intend,” “may,” “might,” “plan,” “possible,” “predict,” “project,” “should,” “strive,” “would,” “aim,” “target,” “commit,” and similar expressions (including the negatives of these terms or variations of them) generally identify forward-looking statements, but the absence of these words does not mean that statement is not forward looking. Actual results could differ materially from those included in the forward-looking statements due to various factors, risks and uncertainties, including, but not limited to, that preclinical testing of claseprubart and DNTH212 and data from clinical trials may not be predictive of the results or success of ongoing or later clinical trials, that the preliminary interim analysis based on a limited number of patients from the Part A open label portion of the claseprubart CAPTIVATE study in patients with chronic inflammatory demyelinating polyneuropathy may not be predictive of the results or success of the remaining patients treated in Part A or patients treated in Part B of the CAPTIVATE study, that the development of claseprubart or DNTH212 may take longer and/or cost more than planned, that the Company or its partner may be unable to successfully complete the clinical development of the Company’s compounds, that the Company or its partner may be delayed in initiating, enrolling or completing its planned clinical trials, and that the Company's compounds may not receive regulatory approval or become commercially successful products. These and other risks and uncertainties are identified under the heading "Risk Factors" included in the Company’s Annual Report on Form 10-K for the period ended December 31, 2025, and other filings that the Company has made and may make with the SEC in the future. Nothing in this presentation should be regarded as a representation by any person that the forward-looking statements set forth herein will be achieved or that any of the contemplated results of such forward-looking statements will be achieved. Nothing in this Presentation should be regarded as a representation by any person that the forward-looking statements set forth herein will be achieved or that any of the contemplated results of such forward-looking statements will be achieved. Dianthus undertakes no obligation to publicly update or revise any forward-looking statement, whether as a result of new information, future events or otherwise, except as required by law. This Presentation contains limited disclosure with respect to an interim responder analysis in the open-label Part A of the registrational claseprubart CAPTIVATE study. The level of detail in this Presentation is limited to preserve the integrity of the trial as a two-part registrational trial.

Agenda Introduction Marino Garcia, Chief Executive Officer Early GO Decision in CAPTIVATE Simrat Randhawa, MD, Head of Research & Development Closing Remarks Marino Garcia, Chief Executive Officer Analyst Q&A Marino Garcia, Chief Executive Officer Simrat Randhawa, MD, Head of Research & Development John King, Chief Commercial Officer Ryan Savitz, Chief Financial Officer & Chief Business Officer

Introduction Marino Garcia, CEO
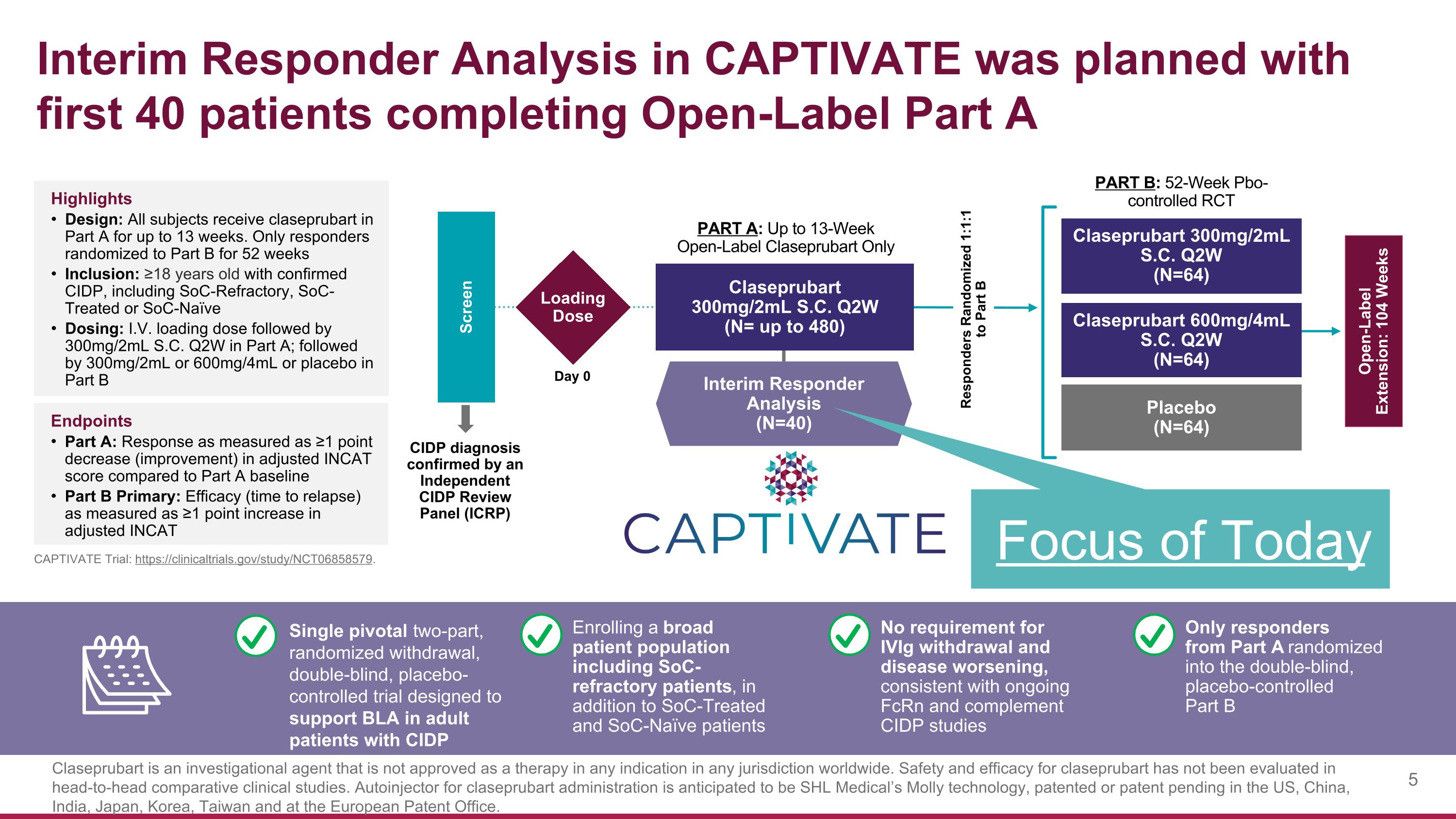
Interim Responder Analysis in CAPTIVATE was planned with first 40 patients completing Open-Label Part A Claseprubart 300mg/2mL S.C. Q2W (N=64) Placebo (N=64) Claseprubart 600mg/4mL S.C. Q2W (N=64) Screen PART A: Up to 13-Week Open-Label Claseprubart Only PART B: 52-Week Pbo- controlled RCT CIDP diagnosis confirmed by an Independent CIDP Review Panel (ICRP) Open-Label Extension: 104 Weeks Loading Dose Day 0 Interim Responder Analysis (N=40) Claseprubart 300mg/2mL S.C. Q2W (N= up to 480) Highlights Design: All subjects receive claseprubart in Part A for up to 13 weeks. Only responders randomized to Part B for 52 weeks Inclusion: ≥18 years old with confirmed CIDP, including SoC-Refractory, SoC-Treated or SoC-Naïve Dosing: I.V. loading dose followed by 300mg/2mL S.C. Q2W in Part A; followed by 300mg/2mL or 600mg/4mL or placebo in Part B Endpoints Part A: Response as measured as ≥1 point decrease (improvement) in adjusted INCAT score compared to Part A baseline Part B Primary: Efficacy (time to relapse) as measured as ≥1 point increase in adjusted INCAT Responders Randomized 1:1:1 to Part B Enrolling a broad patient population including SoC-refractory patients, in addition to SoC-Treated and SoC-Naïve patients No requirement for IVIg withdrawal and disease worsening, consistent with ongoing FcRn and complement CIDP studies Only responders from Part A randomized into the double-blind, placebo-controlled Part B Single pivotal two-part, randomized withdrawal, double-blind, placebo-controlled trial designed to support BLA in adult patients with CIDP Focus of Today CAPTIVATE Trial: https://clinicaltrials.gov/study/NCT06858579. Claseprubart is an investigational agent that is not approved as a therapy in any indication in any jurisdiction worldwide. Safety and efficacy for claseprubart has not been evaluated in head-to-head comparative clinical studies. Autoinjector for claseprubart administration is anticipated to be SHL Medical’s Molly technology, patented or patent pending in the US, China, India, Japan, Korea, Taiwan and at the European Patent Office.

Significant differences between CAPTIVATE and ADHERE Source: Company filings, presentations and clinicaltrials.gov. Ig refers to IVIg and SCIg Data are derived from different clinical trials conducted at different times, with differences in trial design and patient populations. As a result, cross-trial comparisons cannot be made, and no head-to-head clinical trials have been conducted. ADHERE required discontinuation of IVIg or SCIg and evidence of clinically meaningful deterioration before dosing in Part A Defined as a clinical improvement on the parameters that the participant worsened in during run-in (≥4-point increase in I-RODS and/or ≥8-kPa increase in mean grip strength) or clinical improvement (≥1-point decrease) in INCAT Empasiprubart and riliprubart studies Considerations Efgartigimod (FcRn) S.C. QW Claseprubart (aC1s) 300mg/2mL S.C. Q2W Key Differentiators of CAPTIVATE Ph. 3 Study Populations Evaluating claseprubart in SoC-Refractory CIDP patients, in addition to a broader CIDP patient population including SoC-Treated and SoC-Naïve Require IVIg or SCIg Withdrawal and Relapse Prior to Enrolling in Part A of Study1 YES NO Immediate switch 7 days from last Ig dose to claseprubart; consistent with other ongoing complement CIDP studies3 Study Endpoints / Results Confirmed ECI2 Ph. 3 Stage A results: 66.5% ECI (wk 12) Switching Ig patients to claseprubart 7 days after last dose Aiming for ≥1-point adj. INCAT improvement OVER SoC/Ig in ≥50% of patients in Part A Part A designed to evaluate clinically meaningful improvement over Ig after immediate switch 7 days after last dose SoC-Treated Off Treatment SoC-Treated SoC-Naïve SoC-Refractory ~1/3 of pts did not return to pre-Ig washout baseline
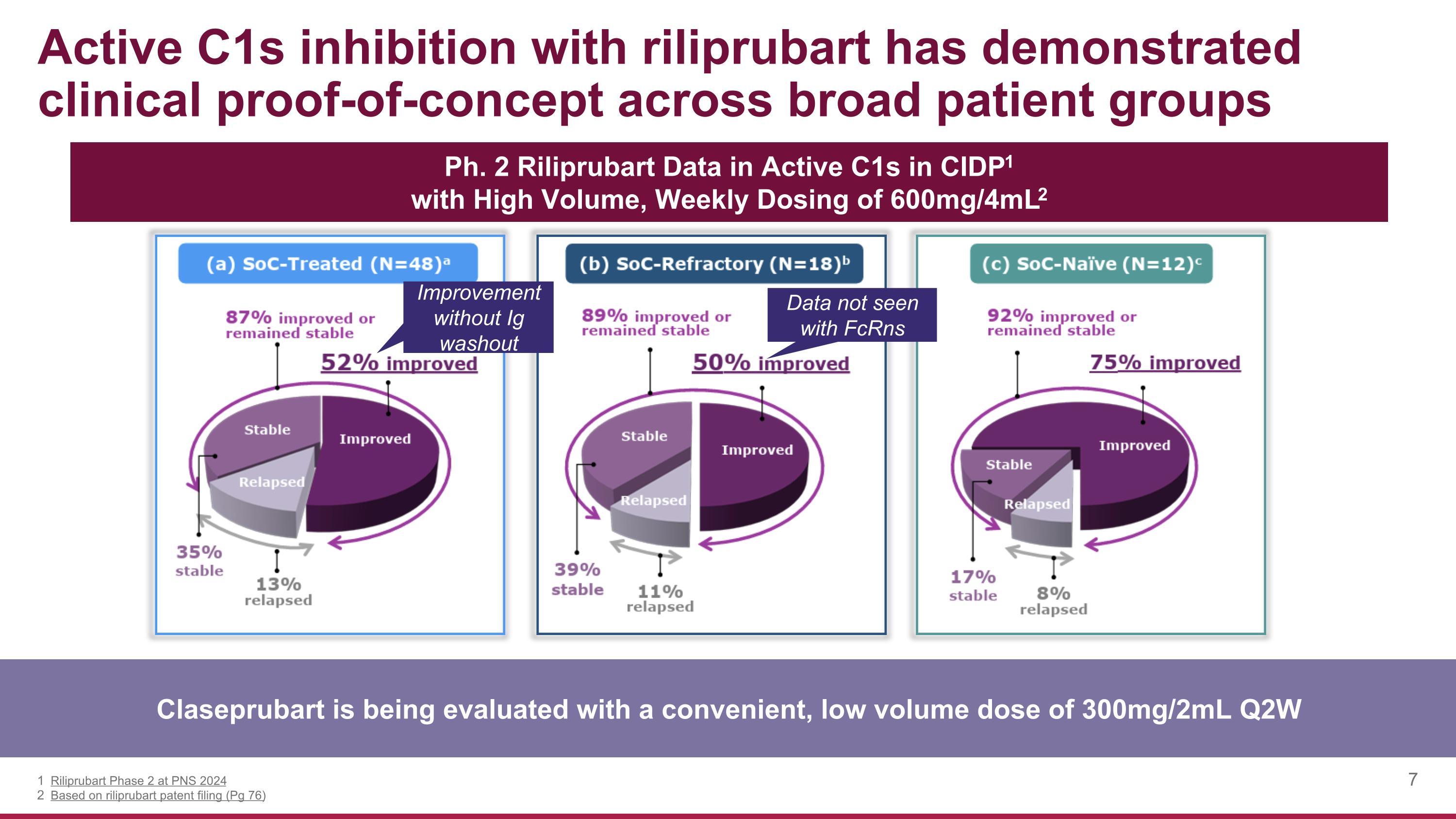
Active C1s inhibition with riliprubart has demonstrated clinical proof-of-concept across broad patient groups Claseprubart is being evaluated with a convenient, low volume dose of 300mg/2mL Q2W Riliprubart Phase 2 at PNS 2024 Based on riliprubart patent filing (Pg 76) Ph. 2 Riliprubart Data in Active C1s in CIDP1 with High Volume, Weekly Dosing of 600mg/4mL2 Data not seen with FcRns Improvement without Ig washout

Targeting response rate of >50% (>20 responders) out of first 40 participants to complete Part A GO: However, increase dose as responder rate below precedent Less than 20 Responders 25% 30% 45% 55% 60% 65% 70% 75% 20 or More Responders Data from CAPTIVATE and riliprubart Ph. 2 are derived from different clinical trials conducted at different times, with differences in trial design and patient populations. As a result, cross-trial comparisons cannot be made, and no head-to-head clinical trials have been conducted. Based on SoC-Refractory and SoC-Treated response rates from riliprubart Phase 2 posters; Riliprubart Phase 2 at AANEM 2023 and Riliprubart Phase 2 at PNS 2024 35% 40% 80% 85% 90% 100% Riliprubart Ph. 2 benchmark response rates: ~50%1 50% Interim Responder Rate in CAPTIVATE Part A GO: Keep dose 300mg/2mL Q2W as target responder rate met or exceeded 95%

Early GO Decision in CAPTIVATE Simrat Randhawa, MD, Head of Research & Development

Early GO decision reached with less than 40 planned participants completing Part A GO decision reached early after 20 confirmed responders were achieved with less than 40 planned participants completing Part A Independent DSMB reviewed the data to date and confirmed GO decision; no related serious infections, no clinical symptoms of autoimmune activation, no related SAEs or discontinuations GO decision supports continued development of claseprubart at 300mg/2mL Q2W S.C. in CIDP targeting a potentially best-in-disease biologic profile Safety / Tolerability Update CAPTIVATE Interim Analysis Objective Targeting response rate of 50% or greater (≥20 patients out of first 40 participants in Part A) based on precedent set with aC1s inhibition GO Decision CAPTIVATE Interim Analysis data cut off as of March 4, 2026
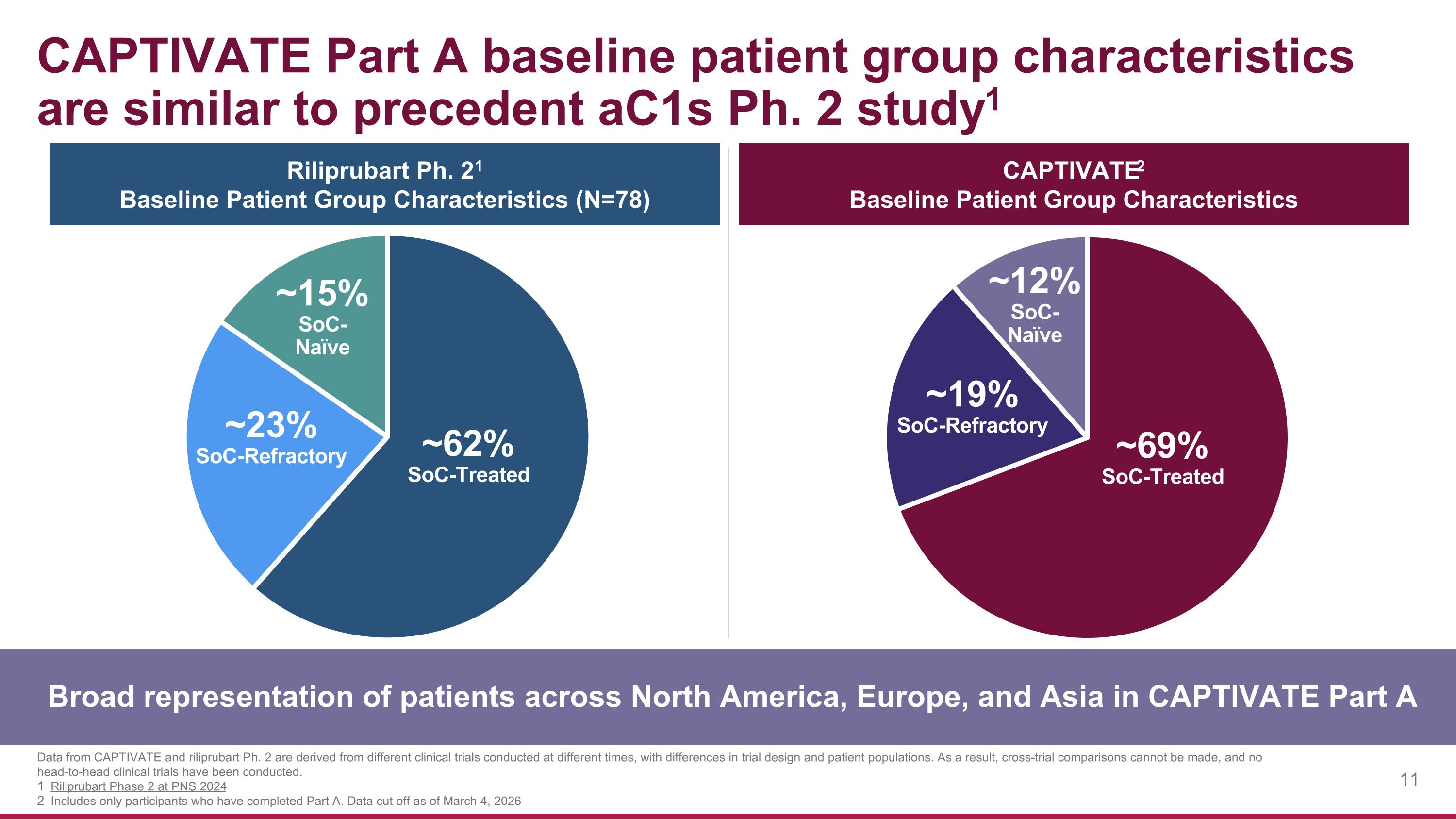
CAPTIVATE Part A baseline patient group characteristics are similar to precedent aC1s Ph. 2 study1 Broad representation of patients across North America, Europe, and Asia in CAPTIVATE Part A Data from CAPTIVATE and riliprubart Ph. 2 are derived from different clinical trials conducted at different times, with differences in trial design and patient populations. As a result, cross-trial comparisons cannot be made, and no head-to-head clinical trials have been conducted. Riliprubart Phase 2 at PNS 2024 Includes only participants who have completed Part A. Data cut off as of March 4, 2026 CAPTIVATE2 Baseline Patient Group Characteristics ~69% SoC-Treated ~19% SoC-Refractory ~12% SoC- Naïve ~62% SoC-Treated ~23% SoC-Refractory ~15% SoC- Naïve Riliprubart Ph. 21 Baseline Patient Group Characteristics (N=78)
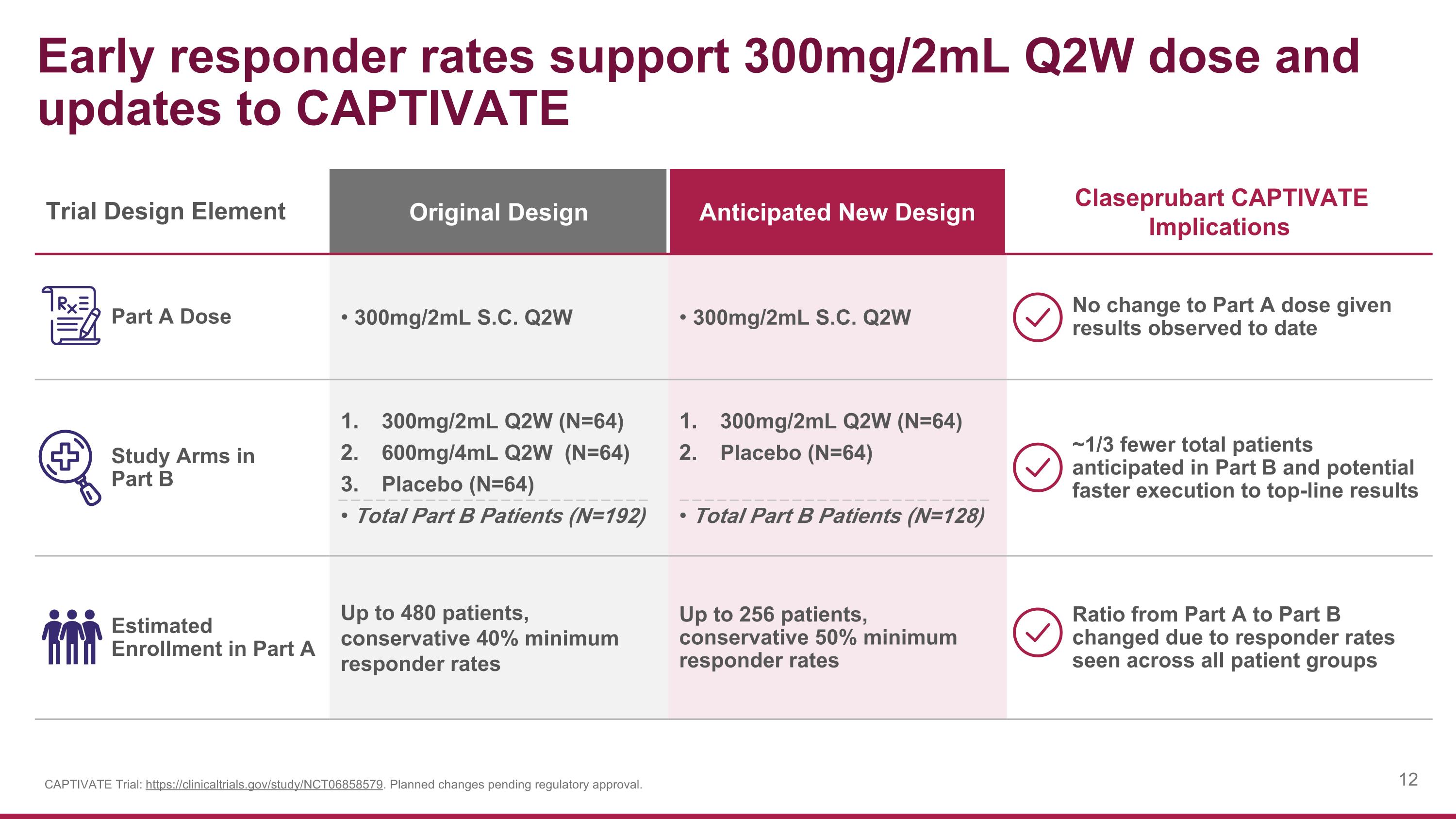
Early responder rates support 300mg/2mL Q2W dose and updates to CAPTIVATE Trial Design Element Original Design Anticipated New Design Claseprubart CAPTIVATE Implications Part A Dose 300mg/2mL S.C. Q2W 300mg/2mL S.C. Q2W No change to Part A dose given results observed to date Study Arms in Part B 300mg/2mL Q2W (N=64) 600mg/4mL Q2W (N=64) Placebo (N=64) Total Part B Patients (N=192) 300mg/2mL Q2W (N=64) Placebo (N=64) Total Part B Patients (N=128) ~1/3 fewer total patients anticipated in Part B and potential faster execution to top-line results Estimated Enrollment in Part A Up to 480 patients, conservative 40% minimum responder rates Up to 256 patients, conservative 50% minimum responder rates Ratio from Part A to Part B changed due to responder rates seen across all patient groups CAPTIVATE Trial: https://clinicaltrials.gov/study/NCT06858579. Planned changes pending regulatory approval.
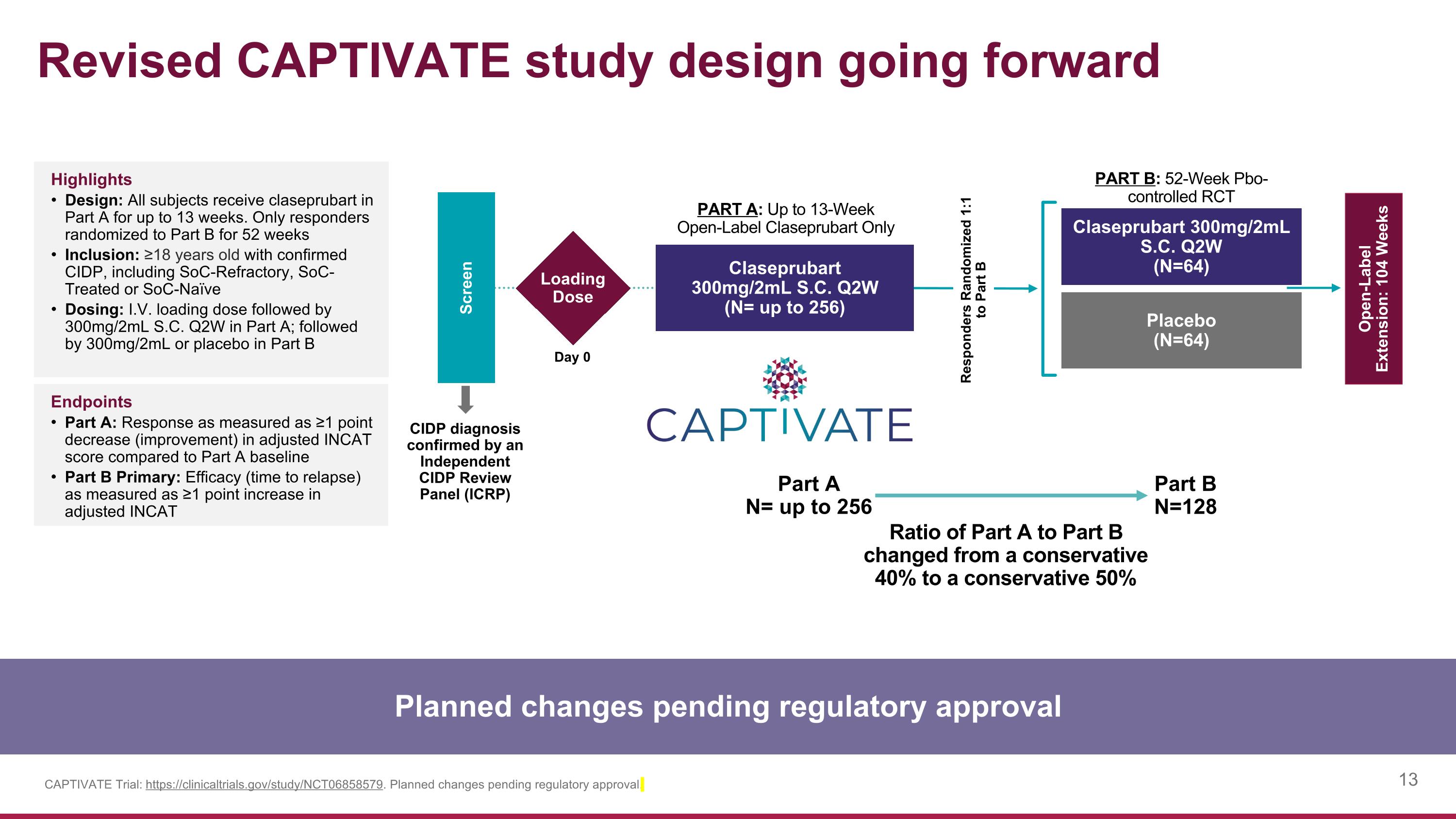
PART B: 52-Week Pbo- controlled RCT Revised CAPTIVATE study design going forward Claseprubart 300mg/2mL S.C. Q2W (N=64) Placebo (N=64) Screen PART A: Up to 13-Week Open-Label Claseprubart Only CIDP diagnosis confirmed by an Independent CIDP Review Panel (ICRP) Open-Label Extension: 104 Weeks Loading Dose Day 0 Claseprubart 300mg/2mL S.C. Q2W (N= up to 256) CAPTIVATE Trial: https://clinicaltrials.gov/study/NCT06858579. Planned changes pending regulatory approval. Highlights Design: All subjects receive claseprubart in Part A for up to 13 weeks. Only responders randomized to Part B for 52 weeks Inclusion: ≥18 years old with confirmed CIDP, including SoC-Refractory, SoC-Treated or SoC-Naïve Dosing: I.V. loading dose followed by 300mg/2mL S.C. Q2W in Part A; followed by 300mg/2mL or placebo in Part B Endpoints Part A: Response as measured as ≥1 point decrease (improvement) in adjusted INCAT score compared to Part A baseline Part B Primary: Efficacy (time to relapse) as measured as ≥1 point increase in adjusted INCAT Responders Randomized 1:1 to Part B Planned changes pending regulatory approval Part A N= up to 256 Part B N=128 Ratio of Part A to Part B changed from a conservative 40% to a conservative 50%

Closing Remarks Marino Garcia, CEO
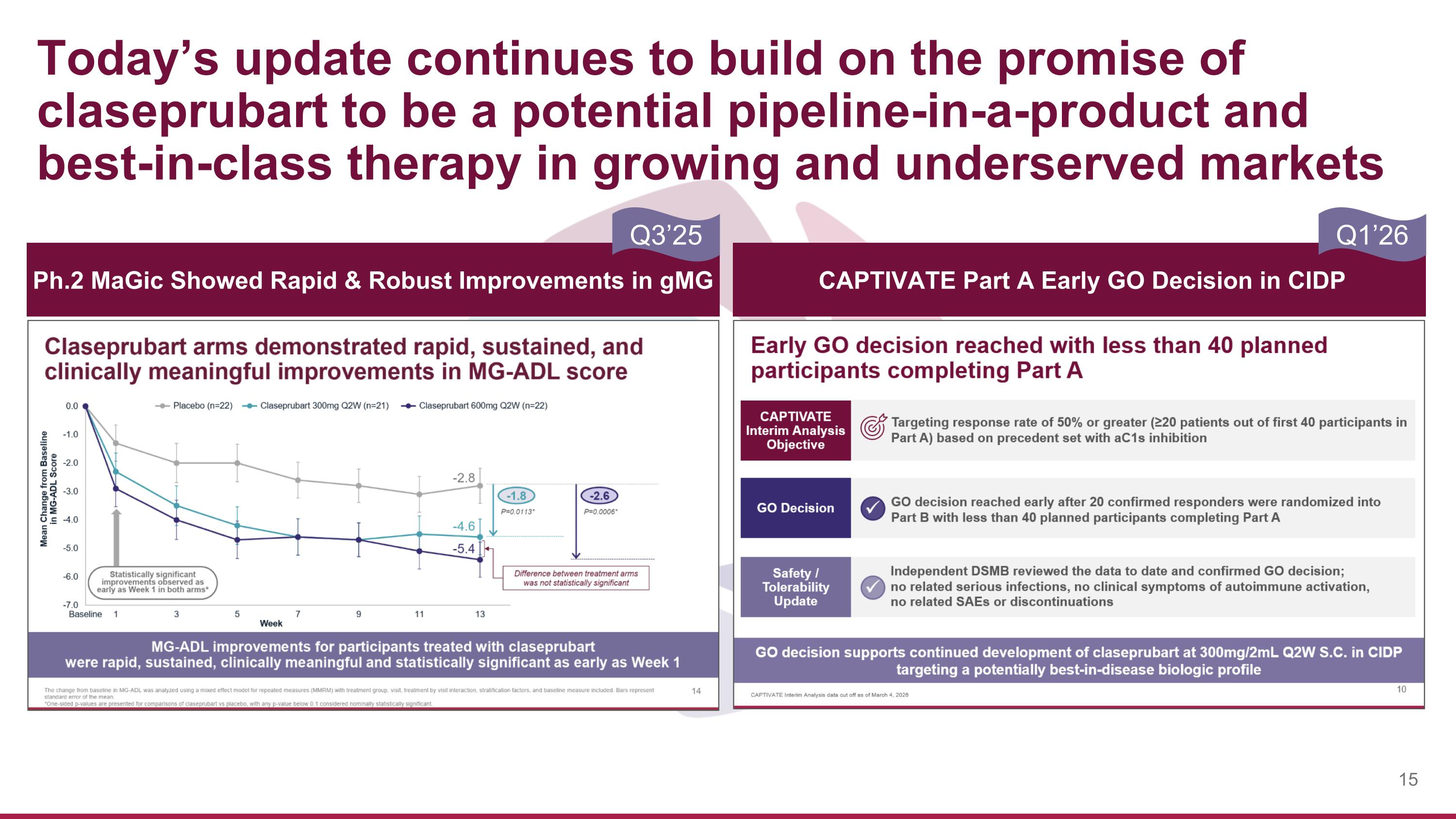
Today’s update continues to build on the promise of claseprubart to be a potential pipeline-in-a-product and best-in-class therapy in growing and underserved markets CAPTIVATE Part A Early GO Decision in CIDP Ph.2 MaGic Showed Rapid & Robust Improvements in gMG Q3’25 Q1’26

US CIDP market offers substantial growth potential given high unmet need and limitations of current standard of care Opportunity for an active C1s inhibitor with the target profile of claseprubart to replace the standard of care Current Ig and biologics account for >$3.5B5,6 Despite SoC, many (30-50%) patients are refractory, face risk of relapse, and confront adverse effects of long-term treatment7-9 FcRn is considered more of an alternative than improvement over IVIg10 Active C1s inhibition has demonstrated ~50% improvement in both SoC-Treated and SoC-Refractory patients in clinical trials11 Opportunity for S.C. self-administer autoinjector US CIDP Market Opportunity 1. Komodo claims data 2013-2025, adjusted to account for 70% capture of real-world patient counts for biologic treated patients, adjusted to account for 27% misdiagnosed; 2. Other Tx: PLEX/Splenectomy/Thymectomy, Rituximab, Biologic; 3. Argenx Vyvgart Hytrulo HCP website; 4. Fierce Pharma, CIDP Pricing; 5. Argenx 4Q 2025 Financial Results, Feb 26, 2026; 6. Immunoglobulin – Global Market Analysis, Fortune Business Insights; 7. Mair D, et al. Novel therapies in CIDP. Journal of Neurology, Neurosurgery & Psychiatry 2025;96:38-46.; 8. Gogia B, et al. Chronic Inflammatory Demyelinating Polyradiculoneuropathy, StatPearls Publishing.; 9. Bus, S.R.M., et al. Clinical outcome of CIDP one year after start of treatment. J Neurol.; 10. Levine T, et al. Early deterioration of CIDP following transition from IVIg to FcRn inhibitor, Journal of the Neurological Sciences; 11. Novel therapies in CIDP, Journal of Neurology, Neurosurgery, and Psychiatry (2024). Claseprubart target profile Est. >$1B4,5 FcRn >2,000 pts3 I.V., S.C., Oral QW S.C. PFS Rapid [<10s] Q2W S.C. Autoinjector CIDP Patients1 Opportunity to Reach More Patients Ig ~19,000 pts Steroids, ISTs, other2 ~10,000 pts Standard of care1 ~40,000 (~5% CAGR) + Steroids (~6,000 pts)

Survey of US Neurologists supports potential transformative opportunity in CIDP of Neurologists strongly prefer treatment options without a boxed warning or REMS program ~54% Total Neurologists 80 Neuromuscular Specialist 81% Generalist 19% Academic 58% Community based 42% Sample Demographics ~13 years in active clinical practice (post-residency), on average ~90% of professional time spent providing direct patient care, on average ~60 CIDP patients seen in the past 12 months, on average of Neurologists strongly believe patients prefer treatments with more consistent and sustained symptom control ~79% of Neurologists strongly believe there is high unmet need for therapies with greater efficacy ~66% Dianthus 2026 Neurologist CIDP and MMN quantitative survey (n=80, N = 65 Neuromuscular specialists, N = 15 General Neurologists), fielded Q1 Data represents % of Neurologists selecting 7-9 on a 9-pt. agreement scale of Neurologists strongly believe patients prefer therapies that are more convenient and easier to administer ~75% Claseprubart aims to differentiate and effectively address the significant unmet needs in the CIDP market

% of Surveyed Neurologists Who Strongly Believe… Surveyed Neurologists want safer, more effective and convenient treatment options than IVIg for CIDP patients Significant unmet need for effective therapies for patients who don’t respond to IVIg 75% IVIg imposes a high treatment burden due to frequent and time-consuming infusions 59% IVIg boxed warning is concerning due to potential risk of kidney failure or thrombosis 55% Overall, surveyed Neurologists believe ~50% patients on IVIg have partial or no response to treatment Dianthus 2026 Neurologist CIDP and MMN quantitative survey (n=80, N = 65 Neuromuscular specialists, N = 15 General Neurologists), fielded Q1 Data represents % of Neurologists selecting 7-9 on a 9-pt. agreement scale Patients on IVIg commonly report waning efficacy prior to their next dose 56%
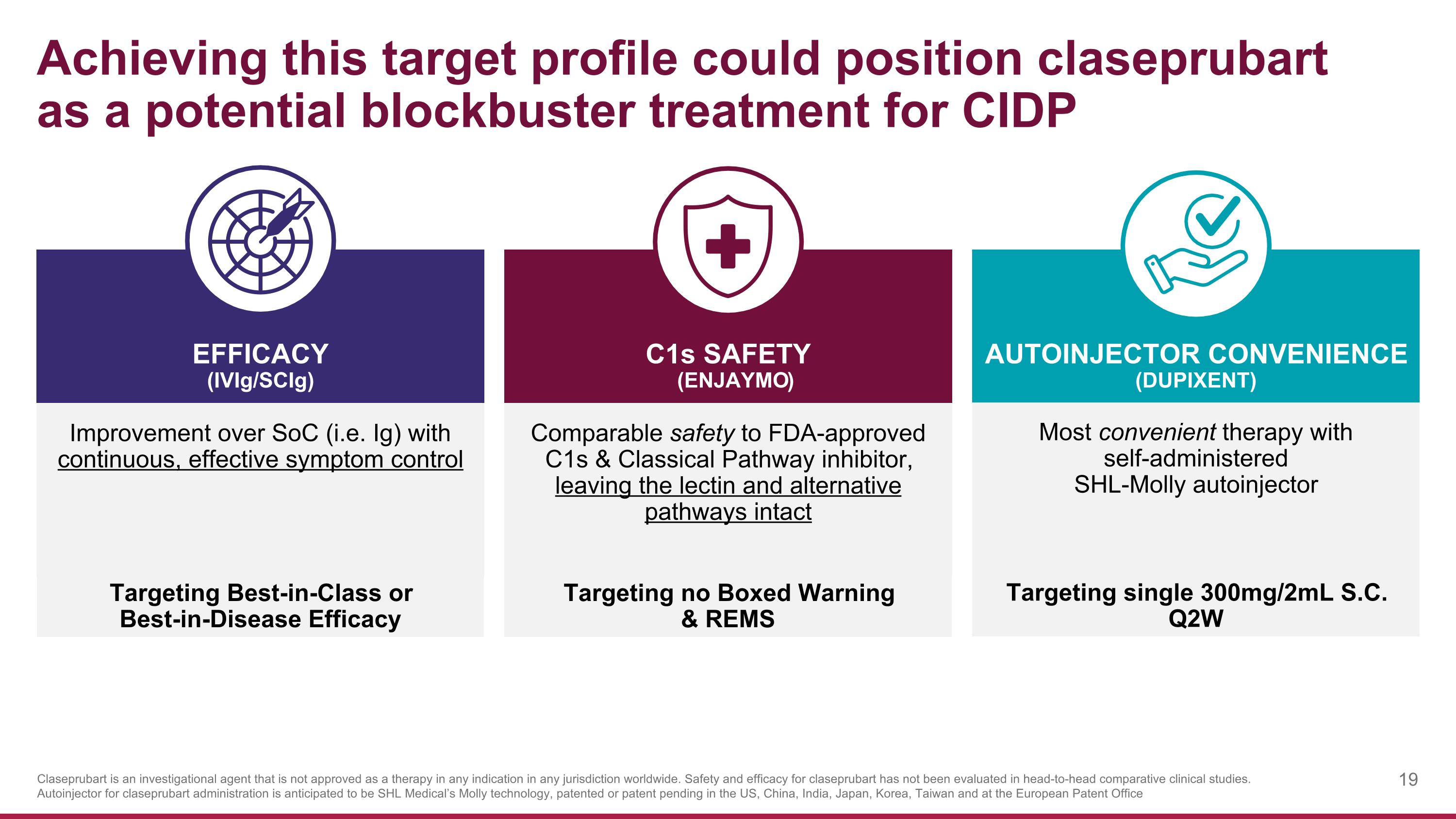
Achieving this target profile could position claseprubart as a potential blockbuster treatment for CIDP Improvement over SoC (i.e. Ig) with continuous, effective symptom control C1s SAFETY (Enjaymo) EFFICACY (IVIg/SCIg) AUTOINJECTOR CONVENIENCE (DUPIXENT) Comparable safety to FDA-approved C1s & Classical Pathway inhibitor, leaving the lectin and alternative pathways intact Most convenient therapy with self-administered SHL-Molly autoinjector Claseprubart is an investigational agent that is not approved as a therapy in any indication in any jurisdiction worldwide. Safety and efficacy for claseprubart has not been evaluated in head-to-head comparative clinical studies. Autoinjector for claseprubart administration is anticipated to be SHL Medical’s Molly technology, patented or patent pending in the US, China, India, Japan, Korea, Taiwan and at the European Patent Office Targeting Best-in-Class or Best-in-Disease Efficacy Targeting no Boxed Warning & REMS Targeting single 300mg/2mL S.C. Q2W
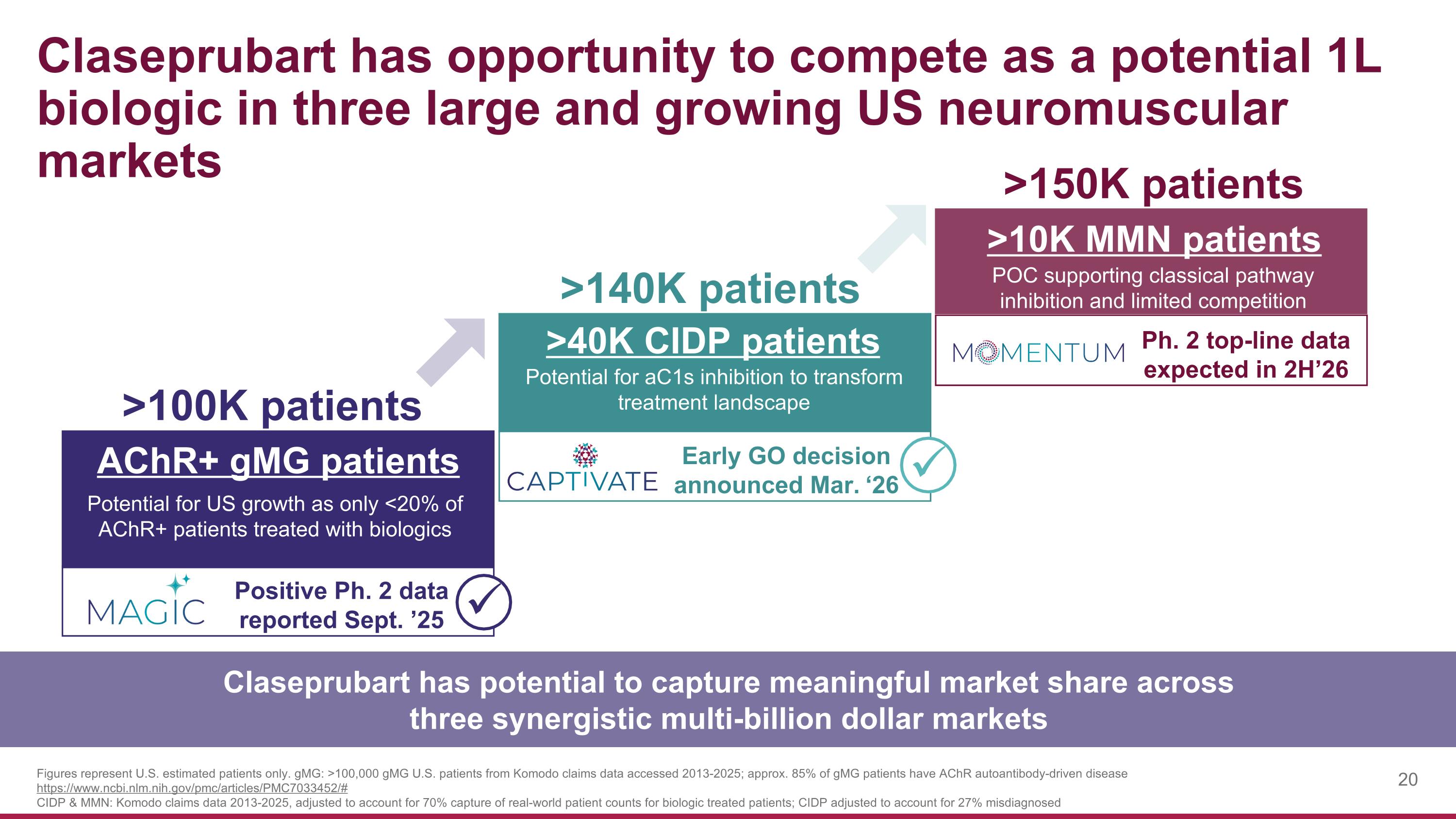
Claseprubart has opportunity to compete as a potential 1L biologic in three large and growing US neuromuscular markets >100K patients >140K patients >150K patients AChR+ gMG patients Positive Ph. 2 data reported Sept. ’25 >40K CIDP patients Early GO decision announced Mar. ‘26 Ph. 2 top-line data expected in 2H’26 Potential for US growth as only <20% of AChR+ patients treated with biologics >10K MMN patients Potential for aC1s inhibition to transform treatment landscape POC supporting classical pathway inhibition and limited competition Figures represent U.S. estimated patients only. gMG: >100,000 gMG U.S. patients from Komodo claims data accessed 2013-2025; approx. 85% of gMG patients have AChR autoantibody-driven disease https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7033452/# CIDP & MMN: Komodo claims data 2013-2025, adjusted to account for 70% capture of real-world patient counts for biologic treated patients; CIDP adjusted to account for 27% misdiagnosed Claseprubart has potential to capture meaningful market share across three synergistic multi-billion dollar markets
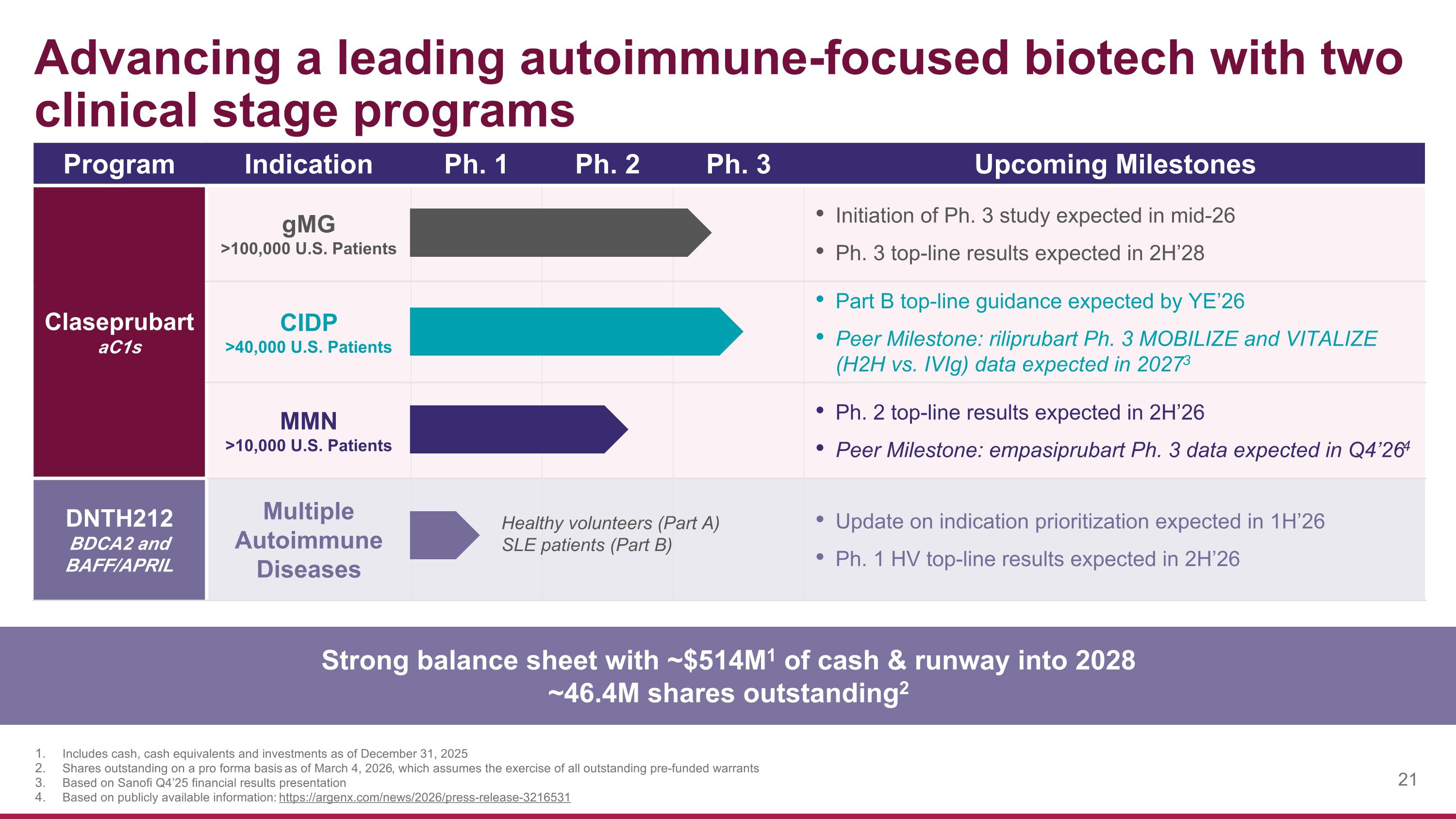
Advancing a leading autoimmune-focused biotech with two clinical stage programs Program Indication Ph. 1 Ph. 2 Ph. 3 Upcoming Milestones Claseprubart aC1s gMG >100,000 U.S. Patients Initiation of Ph. 3 study expected in mid-26 Ph. 3 top-line results expected in 2H’28 CIDP >40,000 U.S. Patients Part B top-line guidance expected by YE’26 Peer Milestone: riliprubart Ph. 3 MOBILIZE and VITALIZE (H2H vs. IVIg) data expected in 20273 MMN >10,000 U.S. Patients Ph. 2 top-line results expected in 2H’26 Peer Milestone: empasiprubart Ph. 3 data expected in Q4’264 DNTH212 BDCA2 and BAFF/APRIL Multiple Autoimmune Diseases Update on indication prioritization expected in 1H’26 Ph. 1 HV top-line results expected in 2H’26 Healthy volunteers (Part A) SLE patients (Part B) Strong balance sheet with ~$514M1 of cash & runway into 2028 ~46.4M shares outstanding2 Includes cash, cash equivalents and investments as of December 31, 2025 Shares outstanding on a pro forma basis as of March 4, 2026, which assumes the exercise of all outstanding pre-funded warrants Based on Sanofi Q4’25 financial results presentation Based on publicly available information: https://argenx.com/news/2026/press-release-3216531
Q&A Early GO Decision in CAPTIVATE CIDP Trial March 9, 2026