
Leading a new era of innovation in immunology 1 4 Q 2 0 2 5 F I N A N C I A L R E S U L T S C A L L F E B R U A R Y 2 6 , 2 0 2 6

Forward Looking Statements This presentation has been prepared by argenx se (“argenx” or the “company”) for informational purposes only and not for any other purpose. Nothing contained in this presentation is, or should be construed as, a recommendation, promise or representation by the presenter or the company or any director, employee, agent, or adviser of the company. This presentation does not purport to be all-inclusive or to contain all of the information you may desire. Certain information contained in this presentation relates to or is based on studies, publications, surveys and other data obtained from third-party sources and the company’s own internal estimates and research. While argenx believes these third-party studies, publications, surveys and other data to be reliable as of the date of this presentation, it has not independently verified, and makes no representation as to the adequacy, fairness, accuracy or completeness of, any information obtained from third-party sources. In addition, no independent source has evaluated the reasonableness or accuracy of argenx’s internal estimates or research, and no reliance should be made on any information or statements made in this presentation relating to or based on such internal estimates and research. Certain statements contained in this presentation, other than present and historical facts and conditions independently verif iable at the date hereof, may constitute forward-looking statements. These forward-looking statements can be identified by the use of forward-looking terminology, including the terms “advance,” “broaden,” “build,” “deliver,” and “expand,” and include statements argenx makes regarding its new era of innovation in immunology; its advancement of its Vision 2030, including (1) 5 new molecules in Phase 3; (2) 10 labeled indications; and (3) 50,000 patients on treatment, supported by its 2025 execution; its 2026 strategic priorities, including its goals to: (1) impact more patients with VYVGART through delivery of broadest MG label, AIM and ITP Phase 3 readouts, and expansion into rheumatology; (2) shape long-term future of FcRn, including advancement of combination approaches and 3 clinical-stage FcRn molecules; and (3) deliver the next wave of innovation, including first empa Phase 3 readout (MMN), 4 Phase 3 molecules, and 10 clinical molecules; its commitment to key expansion opportunities in autoimmune myositis and multifocal motor neuropathy, including redefining biology, treatment, and patient outcomes; its investment in sustainable innovation; its expected timing of its sBLA submission by Q3 2026 based on its positive data from the ADAPT-OCULUS Phase 3 Clinical Trial in ocular MG; its belief that its prefilled syringe launch is increasing patient impact and broadening prescriber base; its commitment to building durable leadership with the broadest label in MG, with an expected total addressable market of 60,000 U.S. MG patients in 2030, as well as its redefinition of patient outcomes through its (1) expected launch in seronegative MG; (2) Ocular MG Phase 3 readout; (3) empowerment of HCP treatment choice; and (4) combination of empasiprubart and efgartigimod; and its clear path to CIDP market expansion by redefining: (1) treatment through evidence generation and HCP prescriber growth and (2) patient outcomes through its biomarker exploration and progression of multiple mechanisms of action. By their nature, forward-looking statements involve risks and uncertainties and readers are cautioned that any such forward-looking statements are not guarantees of future performance. argenx’s actual results may differ materially from those predicted by the forward-looking statements as a result of various important factors, including the results of argenx's clinical trials; expectations regarding the inherent uncertainties associated with the development of novel drug therapies; preclinical and clinical trial and product development activities and regulatory approval requirements in products and product candidates; the acceptance of argenx's products and product candidates by patients as safe, effective and cost-effective; the impact of governmental laws and regulations on our business, including tariffs, export controls, sanctions and other regulations on its business; disruptions caused on our reliance of third parties suppliers, service provides and manufacturing; inflation and deflation and the corresponding fluctuations in interest rates; and regional instability and conflicts. A further list and description of these risks, uncertainties and other risks can be found in argenx’s U.S. Securities and Exchange Commission (the “SEC”) filings and reports, including in argenx’s most recent annual report on Form 20-F filed with the SEC as well as subsequent filings and reports filed by argenx with the SEC. Given these uncertainties, the reader is advised not to place any undue reliance on such forward-looking statements. These forward-looking statements speak only as of the date of publication of this document. argenx undertakes no obligation to publicly update or revise the information in this presentation, including any forward-looking statements, except as may be required by law. This presentation contains trademarks, trade names and service marks of other companies, which are the property of their respective owners. 2

Advancing Vision 2030 10 New Molecules Entered Pipeline 4 Ongoing Registrational Studies ~19K Patients Treated Globally* 5 10 50 New Molecules in Phase 3 Patients on TreatmentK Labeled Indications VISION 2030 Michele, CIDP Patient Execution in 2025

2026 Strategic Priorities Impact More Patients with VYVGART Deliver Next Wave of Innovation Deliver broadest MG label AIM and ITP Phase 3 readouts Expand into rheumatology Shape Long-Term Future of FcRn 3 Clinical-stage FcRn molecules Advance combination approaches First empa Phase 3 readout (MMN) 4 Phase 3 molecules 10 Clinical-stage molecules 4
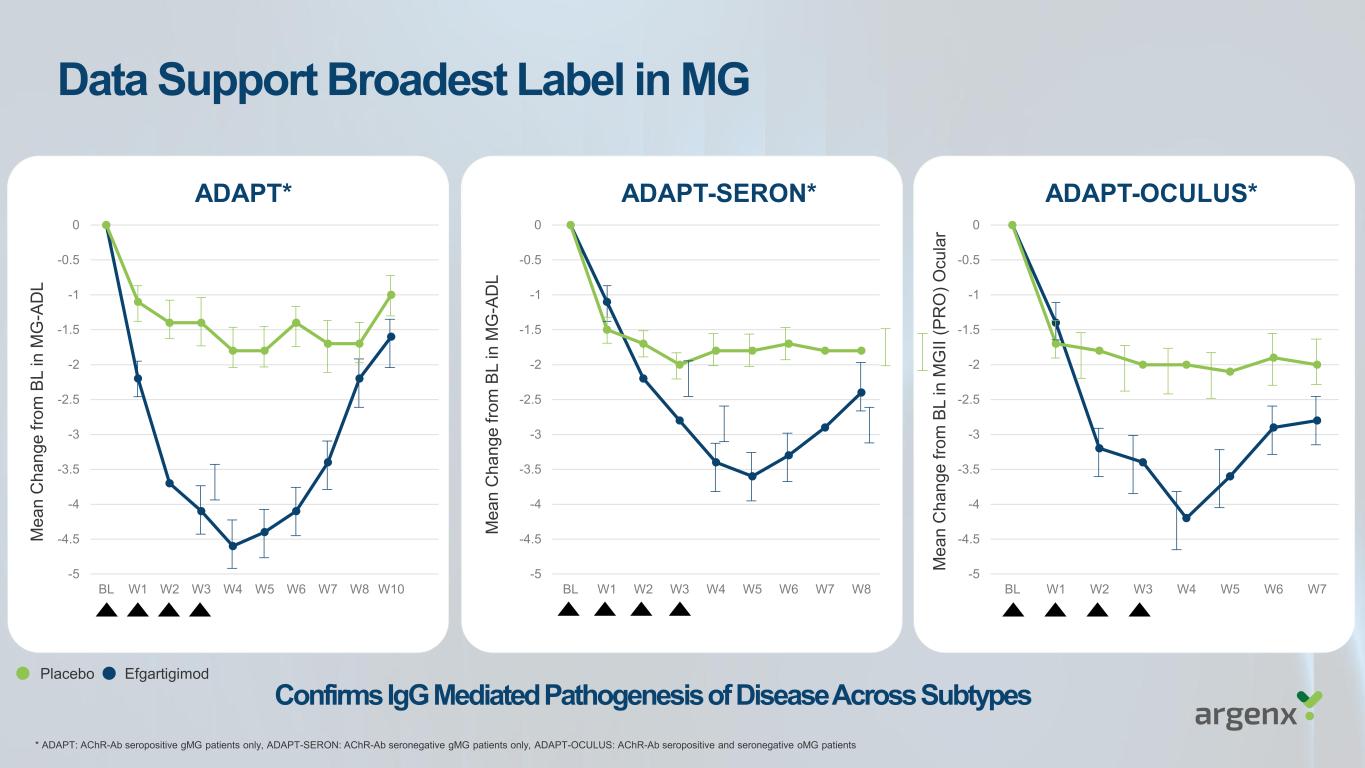
-5 -4.5 -4 -3.5 -3 -2.5 -2 -1.5 -1 -0.5 0 BL W1 W2 W3 W4 W5 W6 W7 W8 ADAPT-SERON* -5 -4.5 -4 -3.5 -3 -2.5 -2 -1.5 -1 -0.5 0 BL W1 W2 W3 W4 W5 W6 W7 W8 W10 ADAPT* -5 -4.5 -4 -3.5 -3 -2.5 -2 -1.5 -1 -0.5 0 BL W1 W2 W3 W4 W5 W6 W7 ADAPT-OCULUS* M e a n C h a n g e f ro m B L i n M G -A D L M e a n C h a n g e f ro m B L i n M G -A D L M e a n C h a n g e f ro m B L i n M G II ( P R O ) O c u la r Confirms IgG Mediated Pathogenesis of Disease Across Subtypes Data Support Broadest Label in MG Placebo Efgartigimod * ADAPT: AChR-Ab seropositive gMG patients only, ADAPT-SERON: AChR-Ab seronegative gMG patients only, ADAPT-OCULUS: AChR-Ab seropositive and seronegative oMG patients

6 Registrational Readouts Over Next 24 Months Phase 3 Data Readouts Sjogren’s Disease Myositis ITP CIDP MMN EFGARTIGIMOD EMPASIPRUBART AChR- gMG (MuSK+, LRP4+, triple seronegative) PDUFA date of May 10, 2026 Decision on Approval 2H 2027 3Q 2026 4Q 2026 2H 2027 4Q 2026 6
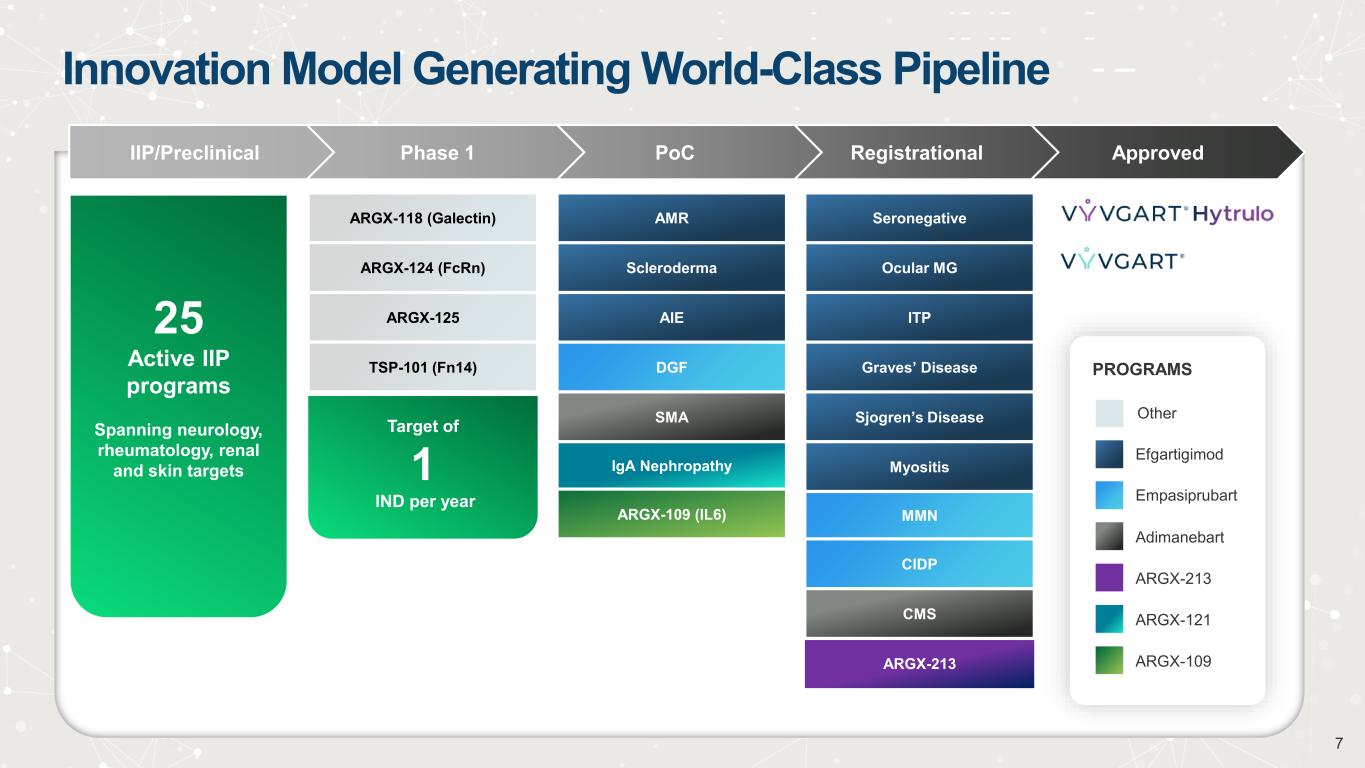
Innovation Model Generating World-Class Pipeline ApprovedRegistrationalPoCPhase 1IIP/Preclinical Seronegative Ocular MG ITP Graves’ Disease Sjogren’s Disease Myositis MMN CIDP CMS ARGX-213 25 Active IIP programs Spanning neurology, rheumatology, renal and skin targets ARGX-118 (Galectin) ARGX-124 (FcRn) ARGX-125 TSP-101 (Fn14) AMR Scleroderma AIE DGF SMA IgA Nephropathy ARGX-109 (IL6) Efgartigimod Empasiprubart Adimanebart Other PROGRAMS ARGX-213 ARGX-121 ARGX-109 Target of 1 IND per year 7
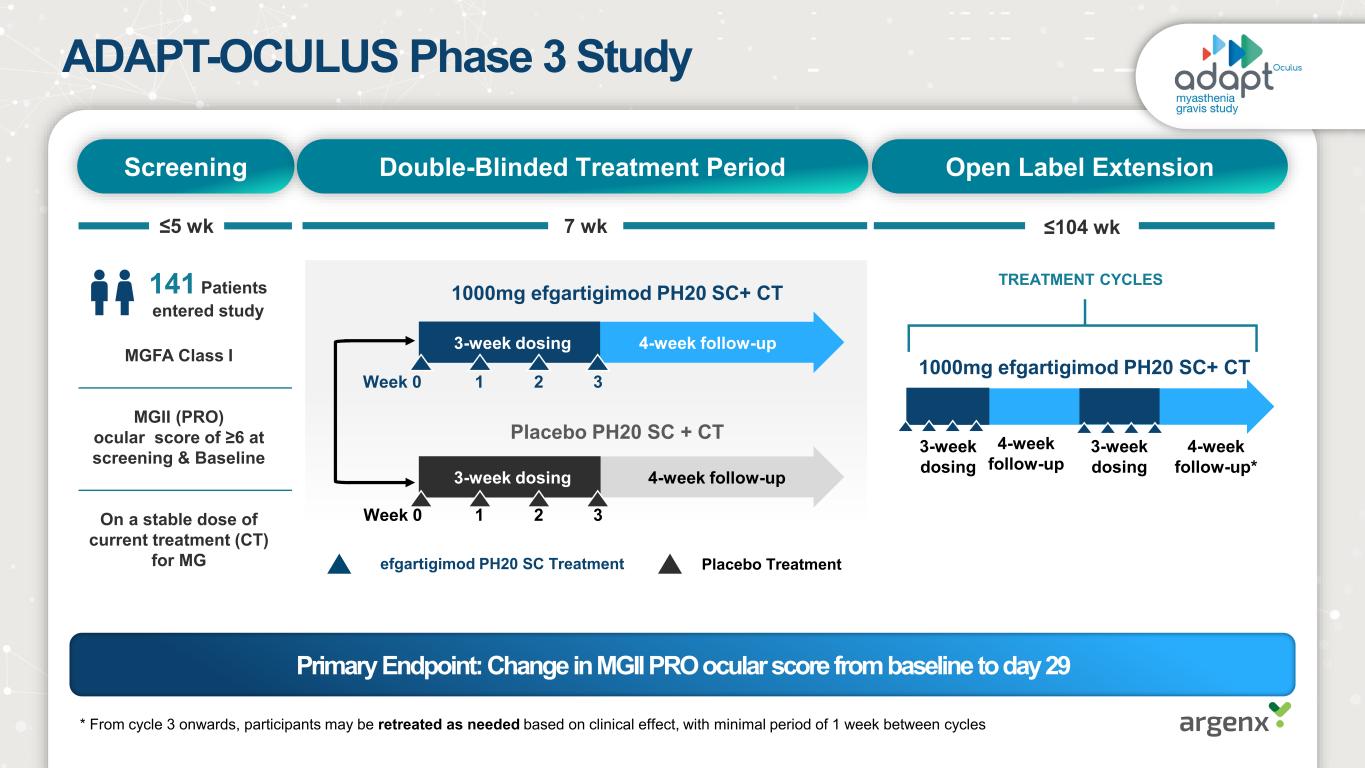
ADAPT-OCULUS Phase 3 Study MGFA Class I MGII (PRO) ocular score of ≥6 at screening & Baseline On a stable dose of current treatment (CT) for MG Screening Open Label ExtensionDouble-Blinded Treatment Period 7 wk≤5 wk ≤104 wk 1000mg efgartigimod PH20 SC+ CT 4-week follow-up 4-week follow-up* Week 0 1 2 3 Week 0 1 2 3 Placebo PH20 SC + CT 1000mg efgartigimod PH20 SC+ CT 4-week follow-up 4-week follow-up efgartigimod PH20 SC Treatment Placebo Treatment 3-week dosing 3-week dosing 3-week dosing 3-week dosing TREATMENT CYCLES141 Patients entered study * From cycle 3 onwards, participants may be retreated as needed based on clinical effect, with minimal period of 1 week between cycles Primary Endpoint: Change in MGII PRO ocular score from baseline to day 29
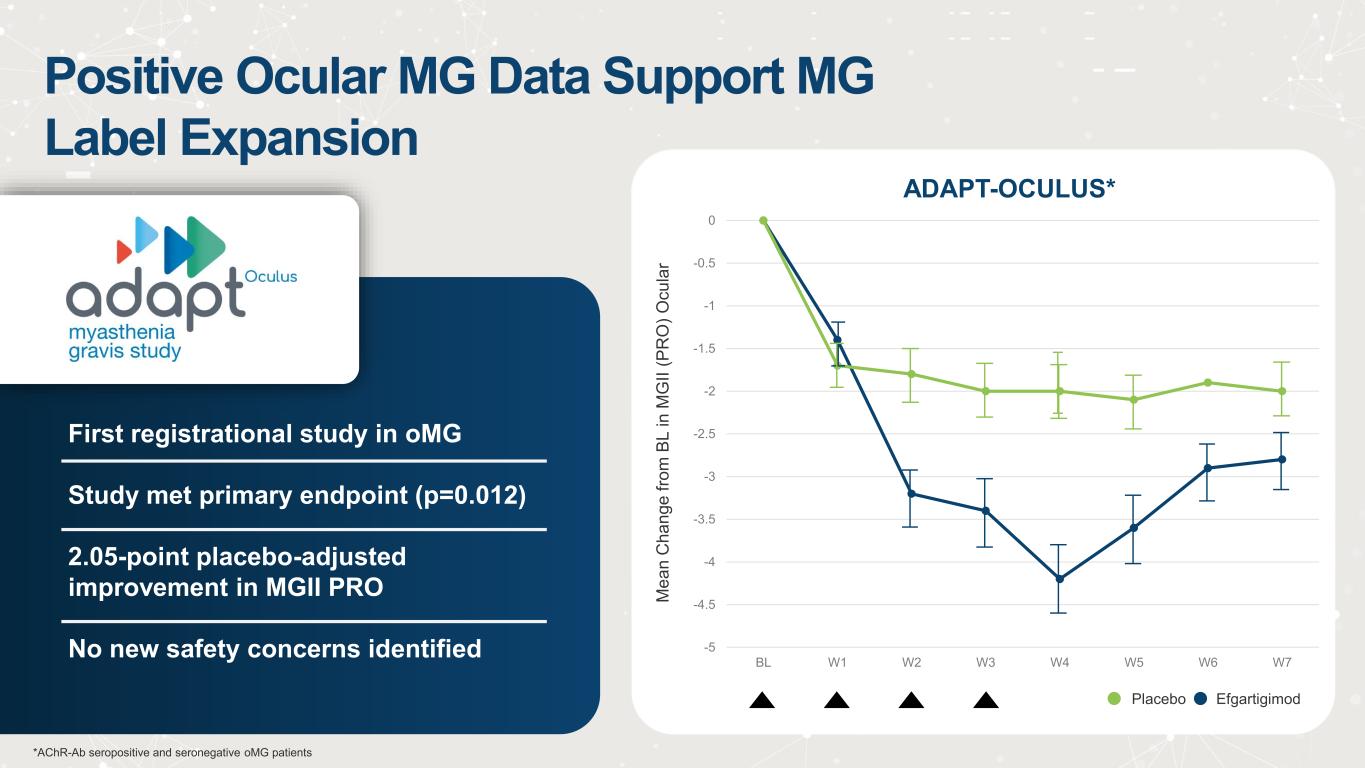
Positive Ocular MG Data Support MG Label Expansion First registrational study in oMG Study met primary endpoint (p=0.012) 2.05-point placebo-adjusted improvement in MGII PRO No new safety concerns identified -5 -4.5 -4 -3.5 -3 -2.5 -2 -1.5 -1 -0.5 0 BL W1 W2 W3 W4 W5 W6 W7 ADAPT-OCULUS* M e a n C h a n g e f ro m B L i n M G II ( P R O ) O c u la r Placebo Efgartigimod *AChR-Ab seropositive and seronegative oMG patients
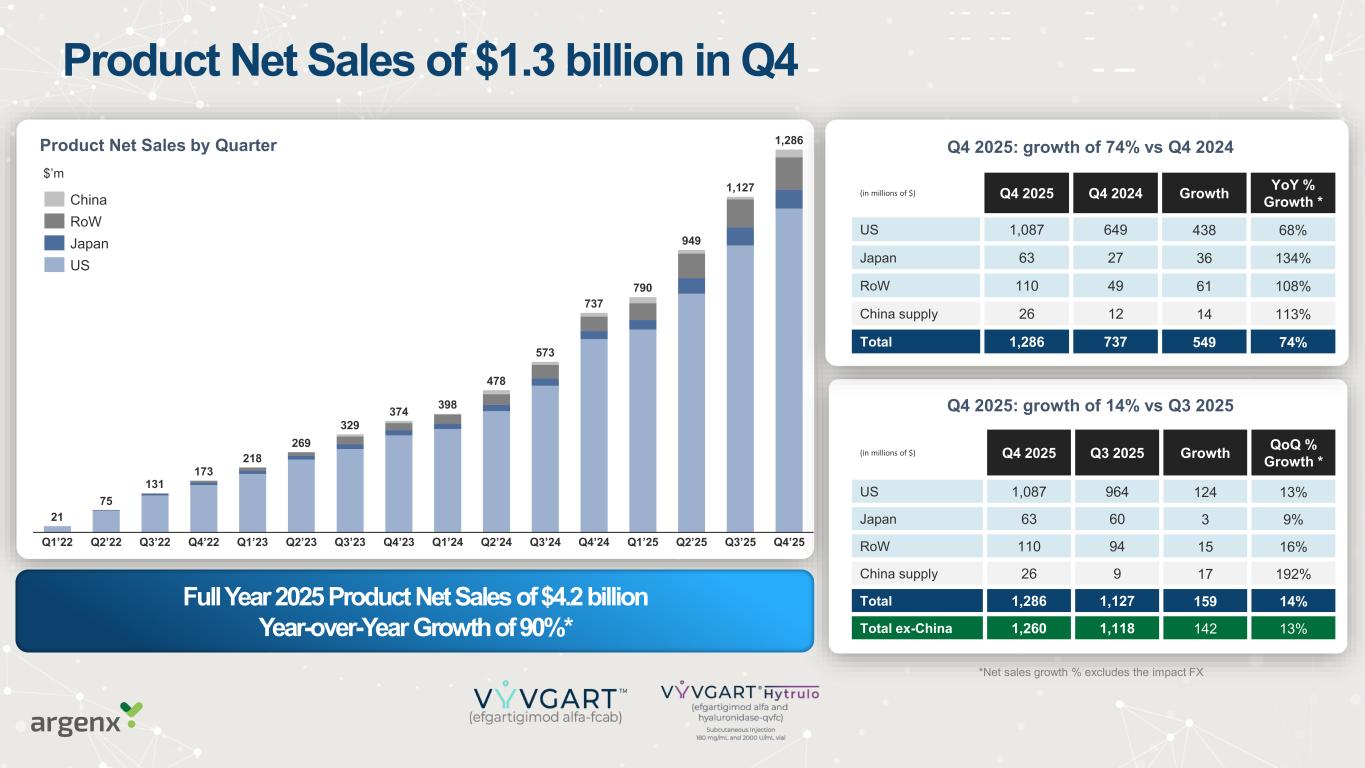
Product Net Sales of $1.3 billion in Q4 Q4 2025: growth of 14% vs Q3 2025 *All growth is operational and excludes the impact of FX (in millions of $) Q4 2025 Q3 2025 Growth QoQ % Growth * US 1,087 964 124 13% Japan 63 60 3 9% RoW 110 94 15 16% China supply 26 9 17 192% Total 1,286 1,127 159 14% Total ex-China 1,260 1,118 142 13% (in millions of $) Q4 2025 Q4 2024 Growth YoY % Growth * US 1,087 649 438 68% Japan 63 27 36 134% RoW 110 49 61 108% China supply 26 12 14 113% Total 1,286 737 549 74% Q4 2025: growth of 74% vs Q4 2024Product Net Sales by Quarter *Net sales growth % excludes the impact FX Full Year 2025 Product Net Sales of $4.2 billion Year-over-Year Growth of 90%* Q2’22 Q3’22 Q4’22 Q1’23 Q2’23 Q3’23 Q4’23 Q1’24 Q2’24 Q3’24 Q4’24 Q1’25 Q2’25 Q3’25 Q4’25Q1’22 75 131 173 218 269 329 374 398 478 573 737 790 949 1,127 1,286 21 China RoW Japan US $’m

Q4 2025 Financial Summary Financial Strength to Invest in Sustainable Innovation (in million of $) 2025 2024 2025 2024 Product net sales 1,286 737 4,151 2,186 Other operating income 36 24 97 66 Total operating income 1,322 761 4,248 2,252 Cost of sales (150) (73) (451) (227) Research and development expenses (372) (297) (1,364) (983) Selling, general and administrative expenses (430) (286) (1,367) (1,055) Loss from investment in a joint venture (4) (2) (12) (8) Total operating expenses (955) (658) (3,194) (2,274) Operating profit/(loss) 368 103 1,054 (22) Financial income 45 39 163 158 Financial expense (1) (1) (4) (2) Exchange (losses)/gains (8) (55) 66 (48) Profit for the period before taxes 403 87 1,279 85 Income tax benefit 130 688 13 748 Profit for the period 533 774 1,292 833 Comparative figures have been presented to be consistent with the one adopted in the current period with respect to the combination of co llaboration revenue and other operating income. Three months ended Twelve months ended December 31 December 31 $1.1 billion in operating income in 2025, marking first year of operating profitability (in million of $) 2025 2024 2025 2024 Product net sales 1,286 737 4,151 2,186 Other operating income* 36 24 97 66 Total operating income 1,322 761 4,248 2,252 Cost of sales (150) (73) (451) (227) Research and development expenses (372) (297) (1,364) (983) Selling, general and administrative expenses (430) (286) (1,367) (1,055) Loss from investment in a joint venture (4) (2) (12) (8) Total operating expenses (955) (658) (3,194) (2,274) Operating profit/(loss) 368 103 1,054 (22) Financial income 45 39 163 158 Financial expense (1) (1) (4) (2) Exchange (losses)/gains (8) (55) 66 (48) Profit for the period before taxes 403 87 1,279 85 Income tax benefit 130 688 13 748 Profit for the period 533 774 1,292 833 Three months ended Twelve months ended December 31 December 31

Innovation has no value unless it provides meaningful benefit to patients

20% YoY increase in new prescribers >4,700 Prescribers in the US NEW PRESCRIBER AND PATIENT GROWTH Prefilled Syringe Launch is Increasing Patient Impact and Broadening Prescriber Base 70% Source: argenx market research Achieved Blockbuster Status as of 3Q 2025 MG EXPANSION Earlier Line Patients #1 PRESCRIBED BIOLOGIC VYVGART drove 60% Growth in overall MG biologics share CIDP EXPANSION Patients directly from orals As of 4Q 2025 Financial Results Mary Beth, MG Patient 13
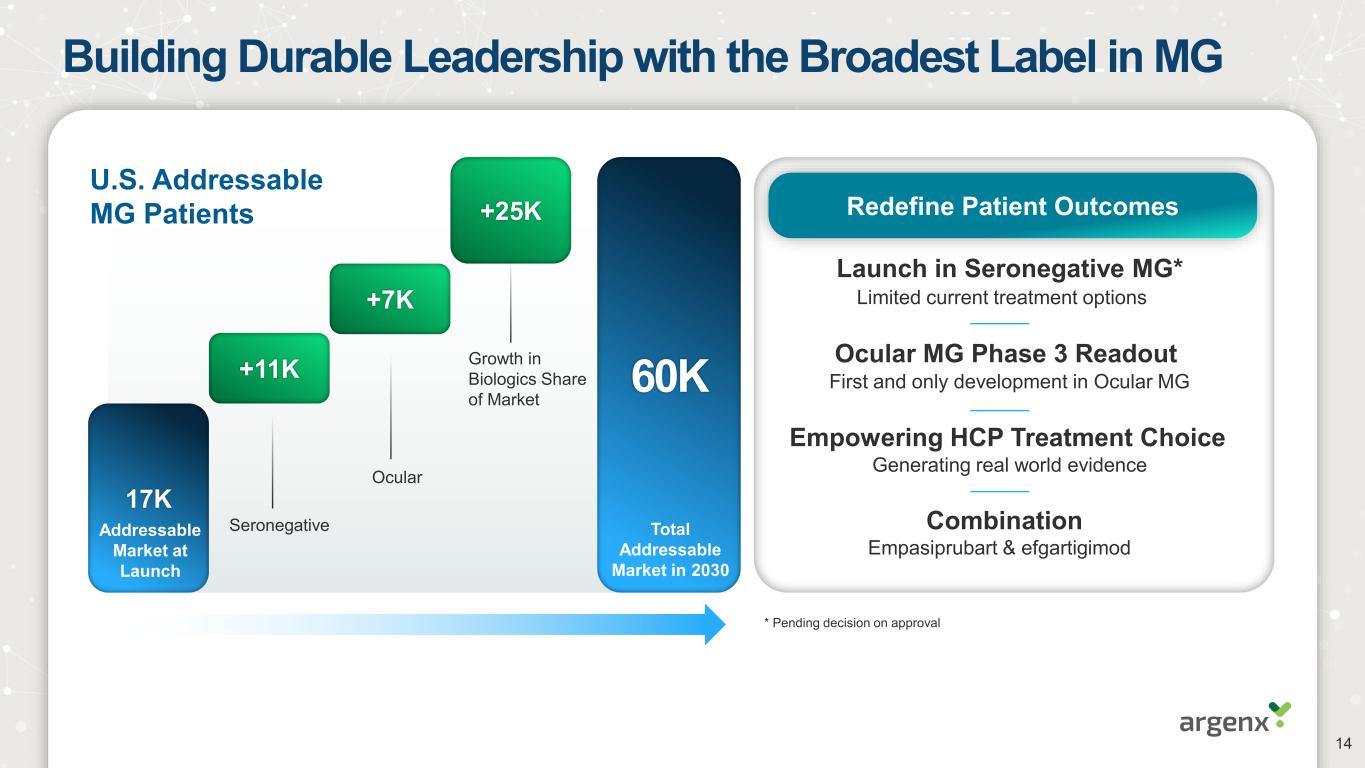
Building Durable Leadership with the Broadest Label in MG Launch in Seronegative MG* Ocular MG Phase 3 Readout Limited current treatment options First and only development in Ocular MG * Pending decision on approval Redefine Patient Outcomes U.S. Addressable MG Patients 17K +11K +7K +25K 60K Seronegative Ocular Growth in Biologics Share of Market Combination Empasiprubart & efgartigimod Generating real world evidence Empowering HCP Treatment Choice Addressable Market at Launch Total Addressable Market in 2030 14
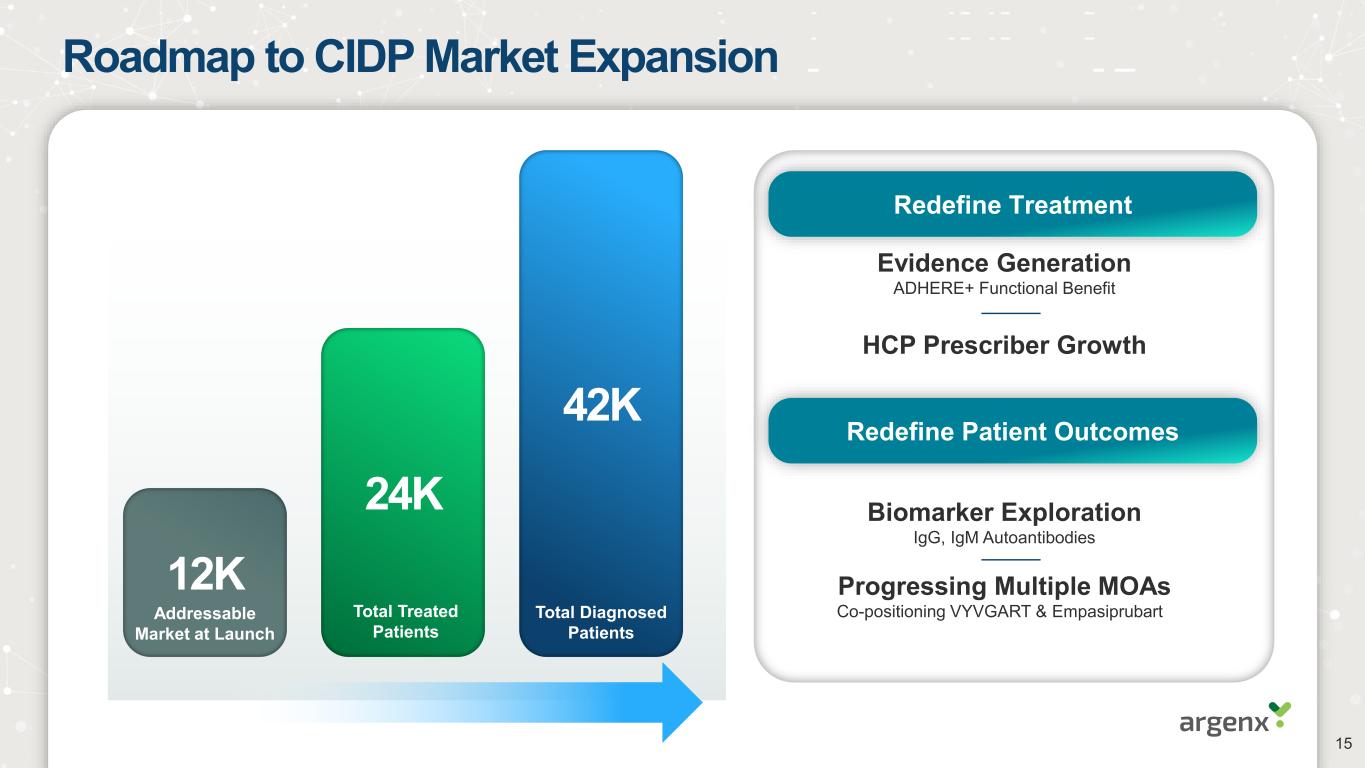
Roadmap to CIDP Market Expansion Evidence Generation ADHERE+ Functional Benefit 42K 24K 12K Total Treated Patients Addressable Market at Launch Total Diagnosed Patients HCP Prescriber Growth Biomarker Exploration IgG, IgM Autoantibodies Progressing Multiple MOAs Co-positioning VYVGART & Empasiprubart Redefine Treatment Redefine Patient Outcomes 15

HumilityInnovation ExcellenceCo-Creation Empowerment We are on a bold mission