

Disclaimer PDF print – this document is only a “printed version” and is not the original annual financial reporting including the audited financial statements pursuant to Article 361 of Book 2 of the Dutch Civil Code. These original annual financial reporting included in the audited financial statements and the auditor’s report thereto, are included in the single report package which can be found at https://www.argenx.com/investors/financial-reports

2025 Annual Report including the Annual Financial Statements for the year ended December 31, 2025 This Annual Report is filed with the Dutch Authority for the Financial Markets (Stichting Autoriteit Financiële Markten, AFM). The following main items included in our annual report on Form 20-F for the year ended December 31, 2025 (2025 20-F) filed with the United States Securities and Exchange Commission (SEC) on or about the date of this Annual Report have not been included in this Annual Report: • Form 20-F cover page; • Item 7 – Major Shareholders and Related Party Transactions; • Item 10E – Taxation; • Item 16E – Purchases of Equity Securities by the Issuer and Affiliated Purchasers; • Item 16G – Corporate Governance; • Report of Independent Registered Public Accounting Firm in respect of Internal Control over Financial Reporting for the SEC filing; • Report of Independent Registered Public Accounting Firm in respect of the PCAOB audits of the 2025 financial statements for the SEC filing; • Exhibits; and • Signatures. The following main sections of our Annual Report have not been included in our 2025 20-F: • Shareholder Letter; • Outlook 2026; • Statement of the Board of Directors; • Risk Appetite and Control; • Share Classes and Principal Shareholders; • Non-Financial Information (including Sustainability Statement); • The Company Financial Statements under Section Financial Statements (prepared pursuant to Dutch law); • Independent auditor’s report - Report on the audit of the financial statements 2025 included in the Annual Report with respect to the AFM Filing; • Limited Assurance Report of the Independent Auditor on the Sustainability Statement; and • Glossary. References to the content of this Annual Report Financial Statements The Financial Statements of argenx SE, which are subject to adoption by the Company’s shareholders at the 2026 General Meeting, are included in Section 6 this Annual Report. Board Report The board Report, within the meaning of Article 2:391 of the Dutch Civil Code, consists of the following sections of this Annual Report: To our Shareholders, Section 1 through Section 5 (excluding Section 3.3), and Section 7 through Section 8. The Report of the Non-Executive Directors is included in Section 3.3. argenx Annual Report 2025 2

Sustainability Statement The Sustainability Statement is included in Section 7.1 of this Annual Report. The Sustainability Statement includes a contents table in Section 7.1.1, which expressly incorporates by reference the relevant sections of this Annual Report. Certain defined terms Unless otherwise indicated, “argenx,” “argenx SE,” “the Company,” “our company,” “we,” “us”, “our” our “Group” refer to argenx SE and its consolidated subsidiaries. argenx SE is a European public company (Societas Europaea) incorporated under the laws of the Netherlands with its statutory seat in Amsterdam, the Netherlands. It is publicly listed in Belgium and the United States of America (the U.S.). The applicable regulations with respect to public information and protection of investors, as well as the commitments we make to securities and market authorities, are described in this Annual Report. We own various trademark registrations and applications, and unregistered trademarks, including but not limited to VYVGART®, VYVGART HYTRULO™, VYVDURA®, ARGENX™, ABDEG™, NHANCE™, SIMPLE ANTIBODY™, ARGENXMEDHUB™, MG UNITED™, SHINING THROUGH CIDP™, VYVIDLY™ and our corporate logo. Trade names, trademarks and service marks of other companies appearing in this Annual Report are the property of their respective holders. Solely for convenience, the trademarks and trade names in this Annual Report may be referred to without the ® and ™ symbols, but such references should not be construed as any indicator that their respective owners will not assert, to the fullest extent under applicable law, their rights thereto. We do not intend to use or display other companies’ trademarks and trade names to imply a relationship with, or endorsement or sponsorship, any other companies. VYVGART® (efgartigimod alfa-fcab) (VYVGART) has been approved in the U.S., Japan, the European Union (the EU) and several other countries such as the United Kingdom (UK), Switzerland, Israel, mainland China (Mainland China), Canada, South Korea and United Arab Emirates for the intravenous treatment of generalized myasthenia gravis (gMG). VYVGART is approved in Japan for the treatment of immune thrombocytopenia (ITP). VYVGART subcutaneous (SC) (efgartigimod alfa + hyaluronidase qvfc) (VYVGART SC) has been approved in the U.S. and China as VYVGART HYTRULO™ (VYVGART HYTRULO) and in several other countries, in Japan as VYVDURA® (VYVDURA) and in the EU and the UK as VYVGART for the treatment of gMG. VYVGART SC has also been approved in the U.S., China, Japan and the EU for the treatment of chronic inflammatory demyelinating polyneuropathy (CIDP). Lastly, VYVGART HYTRULO has also been approved as a prefilled syringe (PFS) in the U.S. for self-injection by adult patients with gMG and CIDP. For more information on the approval and commercialization status in several jurisdictions, please refer to Section 1.3.1. Unless otherwise specified, references in this Annual Report to VYVGART should be read as references to VYVGART and/or VYVGART SC, including VYVGART HYTRULO in relation to the U.S. and China, VYVGART in relation to the EU and the UK and VYVDURA in relation to Japan, depending on the context. Basis of preparation of our audited consolidated financial statements Our consolidated financial statements are prepared in accordance with the IFRS® Accounting Standards (IFRS) as issued by the International Accounting Standards Board (IASB) as adopted by the EU (EU-IFRS) and in accordance with the legal requirements of Part 9 of Book 2 of the Dutch Civil Code. argenx Annual Report 2025 3

Our consolidated financial statements are presented in this Annual Report in U.S. dollars. All references in this Annual Report to “$,” “US$,” “U.S.$,” “U.S. dollars,” “dollars” and “USD” mean U.S. dollars and all references to “€,” “EUR,” and “euros” mean euros, unless otherwise noted. Throughout this Annual Report, references to ADSs mean American depositary shares (ADSs) or ordinary shares represented by ADSs, as the case may be. Forward-looking Statements This Annual Report contains certain forward-looking statements. A forward-looking statement is any statement that does not relate to historical facts or events or to facts or events as of the date of this Annual Report or that are derived from our management’s beliefs and assumptions based on information currently available to our management. Forward-looking statements are generally identified by the use of forward- looking words, such as “aim”, “anticipate”, “aspire”, “believe”, “can”, “continue”, “could”, “estimate”, “expect”, “entail”, “forecast”, “future”, “goals”, “hope”, “intend”, “is designed to”, “likely”, “may”, “might”, “objective”, “plan”, “potential”, “pursue”, “project”, “predict”, “seek”, “should”, “strategy”, “target”, “will” or other or comparable variations or the negative of such terms, or by discussion of strategy, plans, objectives, goals, future events or intentions, although not all forward-looking statements contain these identifying words. These statements relate to our future results of operations and financial positions, prospects, developments, growth, business strategies, plans and our objectives for future operations, results of clinical trials and regulatory approvals, and are based on analyses or forecasts of future developments and estimates of amounts not yet determinable. These forward-looking statements represent the view of management only as of the date of this Annual Report, and we expressly disclaim any obligation or undertaking to update, review or revise forward-looking statements (whether as a result of new information, future developments or otherwise), except as may be otherwise required by applicable law. The forward-looking statements in this Annual Report involve known and unknown risks, future events, assumptions, uncertainties and other factors that could cause our actual future results of operations and financial positions, prospects, developments, growth, business strategies, plans and our objectives for future operations, results of clinical trials and regulatory approvals to differ materially from those forecasted or suggested herein. Forward-looking statements include, but are not limited to, statements about: • the initiation, timing, progress, development and results of clinical trials of our product candidates, including new indications, alternative dosing regimens, treatment modalities, and methods of administration, including statements regarding when results or interim analysis of the clinical trials will be available or made public; • the expansion of our business, including the further development of our sales and marketing abilities and our IIP, and the value of our pipeline; • the potential attributes, benefits, and side effects of our products and product candidates, including new indications, alternative dosing regimens and treatment modalities, and their competitive position with respect to other alternative treatments; • our ability to advance product candidates into, and successfully complete, clinical trials; • our estimates of the number of patients who suffer from the diseases we are targeting and the number of patients that will enroll in our clinical trials; • the demand and commercialization of our products and product candidates, including new indications, alternative dosing regimens, treatment modalities, and methods of administration, if approved; • the anticipated timing or likelihood of market or regulatory decisions relating to or of our products, including new indications, alternative dosing regimens, treatment modalities, and methods of administration; • the anticipated pricing and reimbursement of our products and product candidates, if approved; • our plans to have various programs to help patients afford our products, including patient assistance and co-pay coupon programs for eligible patients; • our ability to establish sales, marketing and distribution capabilities for any of our products and product candidates that achieve regulatory approval; argenx Annual Report 2025 4

• our regulatory strategy and our ability to establish and maintain manufacturing arrangements for our products and product candidates; • the scope and duration of protection, including any exclusivity period, we are able to establish and maintain for intellectual property rights covering our products and product candidates, platform and technology, including our intention to seek patent term extensions where available; • our estimates regarding expenses, future revenues, cash flow, capital requirements and our needs for additional financing; • our expectation that we will benefit from the Belgian innovation income deduction; • our financial performance, including potential volatility in the price of our ordinary shares and ADSs; • the competition we face in our drug discovery, development, and commercialization efforts; • the rate and degree of market acceptance of our products and product candidates, if approved; • the potential benefits of our current collaborations, including the possibility to access partner technology platforms or capabilities; • our plans and ability to enter into or maintain current collaborations for additional programs or product candidates; • our plans and ability to enter into or maintain current new distribution partnerships; • our long-term growth strategy to develop and market additional products and product candidates, including efgartigimod for new indications, empasiprubart and adimanebart; • the impact of government laws and regulations on our business; • our expectations with respect to the timing and amount of any dividends (if any); • our plans regarding our supply chain, including our reliance on third parties, including contract manufacturing organizations (CMOs); and • our business strategies, including Vision 2030, plans, projects, goals and targets and the timing, outcomes and benefits thereof. These include changes in general economic and business conditions. You should refer to Section 2 ”Risk Factors” of this Annual Report for a discussion of important factors that may cause our actual results to differ materially from those expressed or implied by our forward-looking statements. As a result of these factors, we cannot assure you that the forward-looking statements in this Annual Report will prove to be accurate. Furthermore, if our forward-looking statements prove to be inaccurate, the inaccuracy may be material. In light of the significant uncertainties in these forward-looking statements, you should not regard these statements as a representation or warranty by us or any other person that we will achieve our objectives and plans in any specified time frame or at all. We undertake no obligation to publicly update any forward-looking statements, whether as a result of new information, future events or otherwise, except as required by law. You should read this Annual Report and the documents that we reference in this Annual Report and have filed as exhibits to the Annual Report completely and with the understanding that our actual future results may be materially different from what we expect. We qualify all of our forward-looking statements by these cautionary statements. Information regarding market and industry statistics contained in this Annual Report is included based on information available to us that we believe is accurate. Forecasts and other forward-looking information obtained from this available information is subject to the same qualifications and the additional uncertainties accompanying any estimates of future market size, revenue and market acceptance of products and services. In addition, statements that include “we believe” and similar statements reflect our beliefs and opinions on the relevant subject. These statements are based upon information available to us as of the date of this Annual Report, and while we believe such information forms a reasonable basis for such statements, such information may be limited or incomplete, and our statements should not be read to indicate that we have conducted an exhaustive inquiry into, or review of, all potentially available relevant information. These statements are inherently uncertain and you are cautioned not to unduly rely upon these statements. argenx Annual Report 2025 5

Table of Contents To our Shareholders Shareholder Letter 10 2025 In Brief 11 2026 Outlook 17 1 Presentation of the Group 1.1 Company Profile 19 1.2 Strategy and Objectives 23 1.3 Our Products and Products Candidates 25 1.4 Collaborations and licenses 34 1.5 Manufacturing and Supply 36 1.6 Intellectual Property 37 1.7 Regulation 40 1.8 Documents on display 60 2 Risk Factors 2.1 Summary Risk Factors 62 2.2 Risk Factors Related to Commercialization of argenx’s Products and Product Candidates, Including for New Indications 64 2.3 Risk Factors Related to the Development and Clinical Testing of argenx’s Products and Product Candidates 71 2.4 Risk Factors Related to argenx’s Dependence on Third Parties 74 2.5 Risk Factors Related to Other Government Regulations 77 2.6 Risk Factors Related to argenx’s Financial Position 82 2.7 Risk Factors Related to argenx’s Business and Industry 83 2.8 Risk Factors Related to argenx’s Intellectual Property 86 2.9 Risk Factors Related to argenx’s Organization and Operations 90 2.10 Risk Factors Related to the ADSs 93 2.11 Risk Factors Related to being a Foreign Private Issuer or a Dutch Company 95 argenx Annual Report 2025 6

3 Corporate Governance 3.1 Dutch Corporate Governance Code 99 3.2 Management Structure 100 3.3 Report of the Non-Executive Directors 118 3.4 Remuneration Report and Compensation Statement 121 3.5 Corporate Governance – Nasdaq Listing Rules 158 3.6 Share Ownership 158 3.7 Insider Trading 158 3.8 Cybersecurity 159 3.9 Risk Appetite & Control 160 4 General Description of the Company and its Share Capital 4.1 Legal Information on the Company 166 4.2 Share Capital 166 4.3 Share Classes and Principal Shareholders 170 4.4 Limitations on the right to hold securities 172 4.5 General Meeting, Voting Rights and Admission 173 4.6 Anti-Takeover Provisions 175 4.7 Change of Control 175 4.8 Exchange Controls 175 4.9 Amendments of Articles of Association 175 4.10 Transparency Directive 175 4.11 Dutch Financial Reporting Supervision Act 176 4.12 Dividends and Other Distributions 176 4.13 Right to a surplus in the event of a liquidation 177 4.14 Material Modifications to the Rights of Security Holders and Use of Proceeds 177 4.15 Enforcement of civil liabilities 178 4.16 Controls and Procedures 179 4.17 Financial Calendar 2026 180 argenx Annual Report 2025 7

5 Operating and Financial Review and Prospects 5.1 Overview 182 5.2 Basis of presentation 183 5.3 Critical Accounting Judgements and Major Sources of Estimation Uncertainty 183 5.4 Results of Operation 184 5.5 Liquidity and Capital Resources 186 5.6 Research and development, patents and licenses 188 5.7 Trend information 189 5.8 Off-Balance Sheet Arrangements 189 5.9 Contractual Obligations 189 5.10 Information Regarding the Independent Auditor 189 5.11 Material Contracts and Related Party Transactions 190 5.12 Employees 192 5.13 Insurance 193 5.14 Legal and Arbitration Proceedings 193 5.15 Taxation 193 6 Financial Statements 6.1 Consolidated Financial Statements 212 6.2 Notes to the Consolidated Financial Statements 219 6.3 Company Financial Statements of argenx SE for the Year ended December 31, 2025 255 6.4 Other information 261 7 Non-Financial Information 7.1 Sustainability Statement 274 7.2 Environment 287 7.3 Social 301 7.4 Governance 316 7.5 Appendix 323 8 Glossary 8.1 Management Confirmations 328 8.2 Definitions 329 argenx Annual Report 2025 8

To our Shareholders Shareholder Letter 10 2025 In Brief 11 2026 Outlook 17 argenx Annual Report 2025 9

Shareholder Letter Dear Shareholder, 2025 was a remarkable year of execution, grounded in our urgent commitment to deliver meaningful innovation for patients. We delivered across our strategic priorities and continued to make meaningful progress toward our Vision 2030 goal of reaching 50,000 patients – strengthening the core of our business while advancing the science that will define our future. We treated 19,000 patients globally with VYVGART, and we successfully launched the VYVGART HYTRULO pre-filled syringe in gMG and CIDP, continuing to drive new patient and prescriber demand while underscoring the trust we believe physicians place in our medicine. At the same time, we built the foundation for our next wave of growth by advancing a robust, diversified pipeline of precision medicines. We enter 2026 with 10 ongoing registrational clinical trials, each in indications of high unmet need, and brought four new molecules into the pipeline in 2025, three of which are already entering patient clinical trials. All of these programs are rooted in our Immunology Innovation Program (IIP), representing more than a decade of disciplined discovery and entrepreneurial investment. Our progress would not be possible without the dedication of argonauts and our partners across our global organization. This year, our teams worked with focus and urgency - advancing clinical trials, submitting data packages for regulatory approval, scaling commercial operations, preparing for new launches, and building infrastructure to support our long-term sustainable growth. Looking forward, we have clear strategic priorities to guide our growth strategy into 2026 and beyond. First, we aim to expand the global impact of VYVGART by driving broader adoption and unlocking new opportunities, including expansions in both AChR-antibody seronegative gMG (Seronegative gMG) and ocular MG (Ocular MG) patients, following positive data from both Phase 3 clinical trials. We are also advancing into rheumatology, with pivotal data expected in autoimmune inflammatory myopathies (AIM or Myositis) and Sjögren’s disease (SjD). Second, we plan to shape the long-term future of neonatal Fc receptor (FcRn) medicines through combination strategies, the development of next-generation FcRn molecules such as ARGX-213 and ARGX-124, and preparations for an autoinjector launch in 2027. Beyond FcRn, we plan to deliver the next wave of immunology innovation, progressing towards the first pivotal readout of empasiprubart in multifocal motor neuropathy (MMN) and bringing three new molecules into Phase 1. Finally, as we announced in early 2026, Tim Van Hauwermeiren shared his decision to transition out of his role as CEO and into a new role as Non-Executive Chairperson of the Board, subject to shareholder approval at the annual general meeting on May 6, 2026. Karen Massey, who has served as argenx’ Chief Operating Officer since 2023, will be our next Chief Executive Officer. Over the past three years, Karen has delivered exceptional results - accelerating the VYVGART launch, building a scalable commercial engine, and architecting Vision 2030 to connect today’s business to long-term growth. Her steady leadership and dedication to how we work make her the right person to guide argenx through our next chapter. To our shareholders: thank you for your continued partnership and confidence in our long-term vision. To our employees: thank you for your dedication and your deep commitment to making a meaningful difference for patients. We are entering a defining period for our company – and we have the talent, pipeline, and purpose needed to continue building argenx for the long term. Thank you for your continued support. Sincerely, Tim Van Hauwermeiren & Peter Verhaeghe argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Shareholder Letter 10 Peter Verhaeghe Tim Van Hauwermeiren

2025 In Brief 2025 was a year of strong execution as we advanced our long-term commitment to patients under Vision 2030: aiming to treat 50,000 patients globally, secure 10 labeled indications across all approved medicines, and advance five pipeline candidates into Phase 3 development by 2030. We made significant progress executing against this goal throughout the year, by expanding our global reach with VYVGART in two blockbuster indications, advancing 10 ongoing registrational clinical trials, and completed our goal to bring forward four Phase 1 molecules by the end of the decade. Throughout 2025, VYVGART continued to deliver meaningful impact for patients globally, reaching more than 19,000 patients across three indications (gMG, CIDP, and ITP) and three product presentations. We successfully launched our PFS, which expanded access to new patient segments and enabled more convenient treatment options. In gMG, we strengthened our position as the #1 prescribed and fastest-growing biologic, supported by increasingly earlier use in the treatment paradigm and continued expansion into broader patient populations. Positive topline data from the Seronegative gMG clinical trial strengthened our ambition to be the treatment of choice and to pursue the broadest MG label to date. In CIDP, real-world outcomes continued to validate the ADHERE results, with physicians reporting sustained functional improvement and patients experiencing greater independence and quality of life. This strong commercial execution resulted in a milestone for the Company, with VYVGART surpassing $1 billion in product net sales in a single quarter for the first time in the third quarter of 2025. Across the pipeline, we made meaningful progress on a broad set of programs. We advanced efgartigimod through additional Phase 3 clinical trials that are expected to read out in 2026: Myositis and ITP, each supported by compelling biology and robust clinical or proof-of-concept data. Together, we believe these programs strengthen efgartigimod’s position as a foundational FcRn-based therapy with potential across multiple high-need autoimmune diseases. Our second asset, empasiprubart, advanced notably with three Phase 3 clinical trials underway in MMN and CIDP. As a first-in-class antibody targeting complement C2, we further expanded our understanding of C2 biology and its potential to set a new bar for treatment outcomes in immune-mediated neuromuscular diseases. Momentum also continued with adimanebart (ARGX-119), which entered Phase 3 development in CMS and progressed in a proof-of-concept clinical trial in spinal muscular atrophy (SMA). Across our early-stage portfolio, we advanced key next-generation programs, which include ARGX-213 and ARGX-124. We have a highly productive IIP, our engine for sourcing novel biology and accelerating differentiated medicines, with over 25 active programs. Together, these achievements and the progress across our pipeline position us well for the year ahead as we prepare for multiple registrational readouts and continued expansion of our global patient impact. argenx Annual Report 2025 2025 In Brief 11 Operational Highlights

Corporate Achievements Karen Massey Tim Van Hauwermeiren Subject to shareholder approval, Karen Massey, current COO, will transition to CEO and Executive Director and Tim Van Hauwermeiren, current CEO, will transition to Non-Executive Chairperson of the Board of Directors. Sandrine Piret-Gérard In 2025, Sandrine Piret-Gérard was appointed Chief Commercialization Officer. Sandrine joins from Gilead, where she lead the U.S. commercial organization. Anthony Rosenberg Anthony Rosenberg, who has served as a non- executive director since April 2017, was reappointed as a non-executive director and vice-chairperson of the Board of Directors for a term of 2 years. 1,863 Expansion to 1,863 full-time employees (as of December 31, 2025) to support further growth of our business, including fully staffed commercial teams in the U.S., Europe, Japan and Canada.Employees argenx Annual Report 2025 Corporate Achievements 12
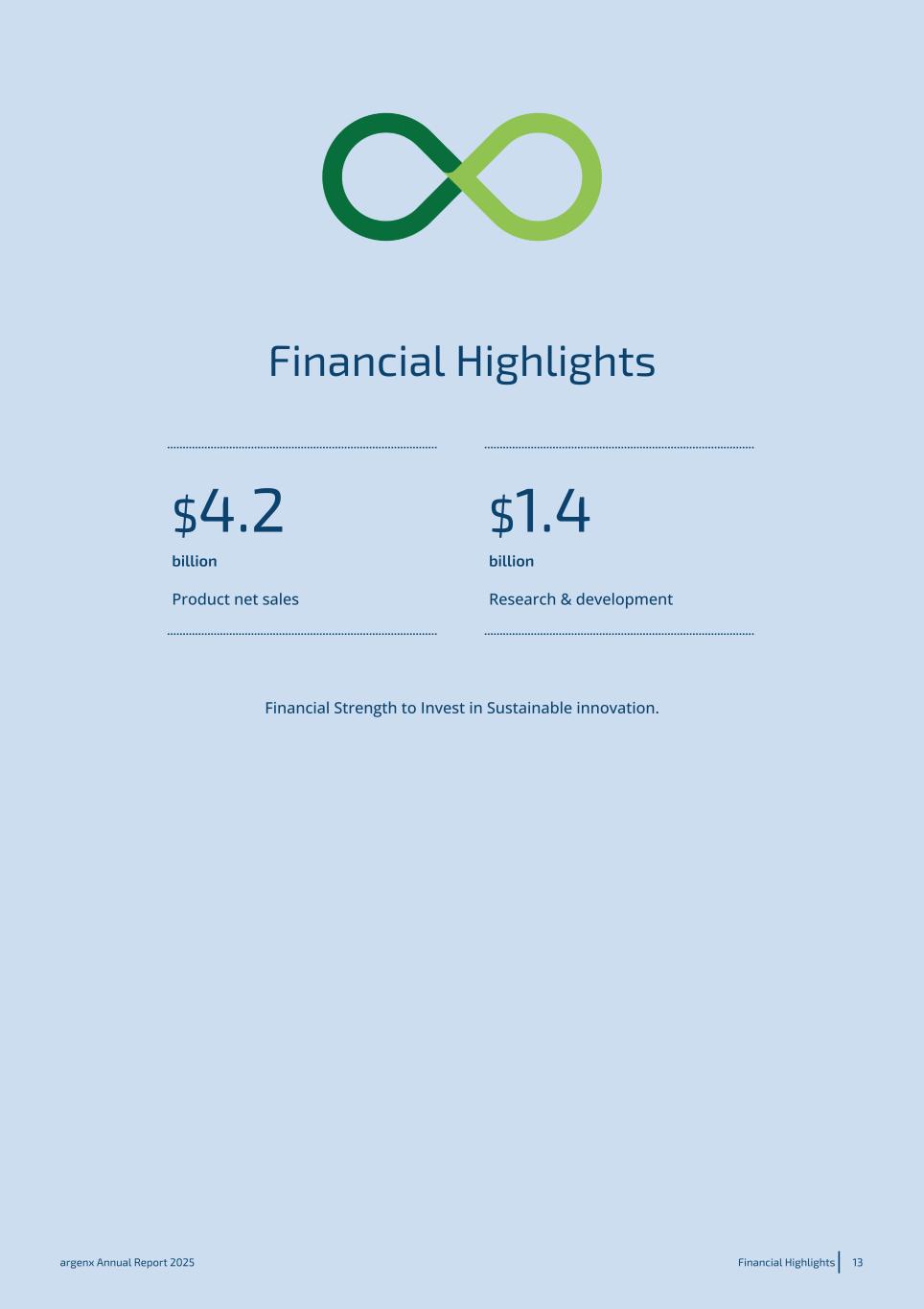
Financial Highlights $4.2 $1.4 billion billion Product net sales Research & development Financial Strength to Invest in Sustainable innovation. argenx Annual Report 2025 Financial Highlights 13

argenx Annual Report 2025 This is the story of Lucille 14 Despite living with CIDP (chronic inflammatory demyelinating polyneuropathy) for about a decade, Lucille’s can-do attitude and deep determination have helped her continue to live life on her own terms. She continues to stay busy, active, and positive about what her life can be, even with CIDP. How Lucille’s Determination Helps Her Travel the World, Even With CIDP

A long path to diagnosis At first, Lucille’s doctor thought her numbness and balance issues were being caused by her diabetes. As they continued to get worse, however, they both knew something else was going on. “The doctor would hold on to me and tell me to close my eyes,” she said, “and I just couldn’t stay standing. I’d fall down if he wasn’t holding me up.” Her doctor wasn’t familiar with CIDP, and he couldn’t figure out what was wrong. So for 5 years, Lucille’s symptoms continued to worsen. Eventually, he referred her to a colleague who happened to be familiar with CIDP and was able to help Lucille finally get the right diagnosis. “I wish I would have gotten a diagnosis sooner,” she said. Seeing more than one doctor is sometimes necessary to get a CIDP diagnosis. Not all doctors are familiar enough with CIDP to suggest the needed testing. Diagnosis received, challenge accepted. Being a very determined person, Lucille made the conscious decision to face her diagnosis head-on. Instead of focusing on the condition and how it could limit her, she focused on the many things she wanted to do and how she could limit the impact of CIDP on her lifestyle. She committed to a schedule of regular, ongoing treatments that have enabled her to continue doing the things that give her joy. “CIDP’s not going to get me down, until it actually takes me down,” she said. Lucille loves to travel. She has a very adventurous spirit, and her travels are not for the faint of heart. Not only has she made it to the top of Machu Picchu since being diagnosed, but she’s also visited Morocco, climbed the Great Pyramid in Giza, Egypt, and flew in a hot air balloon in Turkey, among many other exotic places. Unlike many people’s vacations, when Lucille travels, there’s no sitting around and taking it easy. That’s not to say CIDP hasn’t affected her abilities. These days, whenever she walks for a distance, she uses walking sticks for balance and support. She also has trouble walking down stairs, due to unsteadiness in her legs. These CIDP symptoms may have slowed her down, but they haven’t defeated her. Lucille has been able to manage her symptoms and keep moving with the help of caregivers, which include her children and her siblings. While traveling with her son in England, the two walked over 49 miles, despite Lucille falling on day 2. “My son helps me a lot. He knows that I can get tired. He knows that if I'm walking down stairs, to be in front of me so that I don't fall down the stairs.” Lucille recognizes the importance of a good support system. In addition to her son and siblings, with whom she’s very close, she also has a group of longtime friends and a loving support dog she can depend on for help and comfort. argenx Annual Report 2025 This is the story of Lucille 15

Because she’s so focused on doing what she loves, though, she often forgets about CIDP and the fact that her legs are weaker than they used to be. “I need to remind myself that, even though I’m not letting CIDP stop me, I still need to be careful and take my walking sticks or a cane with me,” she said. When hiking, “I may be the last person in the group to make it to the destination,” she said, “but I’m never far behind.” Hiking down a mountain in Peru was a difficult and scary adventure, but she set her sights on the goal and forged ahead. “I wasn’t going to give in to fear.” When CIDP symptoms start to get her down, Lucille’s confidence helps her get through. “To live with CIDP, I have to push through each day—bad days as well as good ones.” Lucille sums up her experience by saying, “As much as I can, I try to keep doing the things I love to do, even though I have CIDP.” She knows that’s not always possible, but that doesn’t stop her from living life on her terms. She already has several more adventures planned, including a canoe trip down the Amazon River. argenx Annual Report 2025 This is the story of Lucille 16

2026 Outlook 2026 marks a defining year on the path to Vision 2030 with three strategic priorities: • Impact more patients globally with VYVGART, driving broader adoption across current patient populations and unlocking new opportunities with potential label expansions • Shape the long-term future of FcRn medicines, advancing future FcRn molecules, innovative delivery modalities and combination approaches designed to transform patient outcomes • Deliver next wave of immunology innovation, accelerating empasiprubart and diversified pipeline of first-in-class molecules to drive sustainable value creation 12-18 Month Catalyst Outlook Phase 3 Data Readouts EFGARTIGIMOD EMPASIPRUBART Myositis 3Q 2026 MMN 4Q 2026 ITP 4Q 2026 CIDP 2H 2027 SjD 2H 2027 Other 2026 and 2027 Milestones: • Seronegative gMG FDA decision on approval expected on May 10, 2026 • Registrational clinical trial in Graves’ disease expected to initiate in 2026 • adimanebart CMS registrational clinical trial on track to start in third quarter of 2026 • ARGX-213 expected to enter patient clinical trials in 2026 • ARGX-124 expected to complete Phase 1 by end of 2026 • ARGX-121 Phase 2 in IgA nephropathy expected to start in 2026 • Three new molecules expected to enter Phase 1 in 2026: ARGX-118, ARGX-125, TSP-101 argenx Annual Report 2025 2026 Outlook 17

Presentation of the Group 1.1 Company Profile 19 1.2 Strategy and Objectives 23 1.3 Our Products and Product Candidates 25 1.4 Collaborations and licenses 34 1.5 Manufacturing and Supply 36 1.6 Intellectual Property 37 1.7 Regulation 40 1.8 Documents on display 60 argenx Annual Report 2025 18 1

1 Presentation of the Group 1.1 Company Profile 1.1.1 General At argenx, we are defining the future of immunology with an entrepreneurial mindset, bringing together agility, focus, and a collaboration in all that we do. We are a global, commercial-stage, entrepreneurial science company. Our co-creative discovery engine, the Immunology Innovation Program, is powered by partnerships between leading disease biologists and our antibody engineers, enabling us to advance with urgency a robust pipeline of differentiated therapies for severe diseases. We are committed to accelerating innovation, breaking down barriers to access, and building lasting trust through transparency and evidence. Our dynamic pipeline and evidence-driven approach position argenx to deliver sustainable growth and long-term value for patients, healthcare providers, and investors alike. We developed and commercialized the first approved FcRn blocker and we are evaluating efgartigimod in multiple serious autoimmune diseases. We are also advancing our second and third assets, empasiprubart, a complement 2 (C2) inhibitor and adimanebart, a muscle-specific kinase (MuSK) agonist, both of which are now in Phase 3 clinical trials. Our legal and commercial name is argenx SE. We were incorporated under the laws of the Netherlands on April 25, 2008, as a private company with limited liability (besloten vennootschap met beperkte aansprakelijkheid). From incorporation until August 28, 2009, our research and development activities were initially performed in the Netherlands, then Belgium, by argenx N.V. and its legal predecessors. Since August 28, 2009, all our research and development activities have been performed by our wholly-owned subsidiary, argenx BV, under a license provided by argenx N.V. Throughout this time, argenx BV assigned all resulting intellectual property to argenx N.V. On May 28, 2014, we converted to a Dutch public company with limited liability (naamloze vennootschap). On April 26, 2017, we converted to a Dutch European public company with limited liability (Societas Europaea or SE). On May 5, 2017, we transferred the legal ownership of all intellectual property rights of argenx SE to argenx BV, effective retroactively as of January 1, 2017. As a result, since January 1, 2017, (i) argenx BV holds all legal and economic ownership of our intellectual property rights, and (ii) the research and development agreement between argenx SE and argenx BV has been terminated. Our official seat is in Amsterdam, the Netherlands, and our registered office is at Laarderhoogtweg 25, 1101 EB Amsterdam, the Netherlands. We are registered with the trade register of the Dutch Chamber of Commerce under number 24435214. Our European legal entity identifier number (LEI) is 7245009C5FZE6G9ODQ71. Our telephone number is +31 (0) 10 70 38 441. Our website address is www.argenx.com. This website is not incorporated by reference in this Annual Report. The SEC maintains an Internet site that contains reports, proxy and information statements, and other information regarding issuers that file electronically with the SEC at www.sec.gov. The registered agent for service of process in the U.S. is CT Corporation System, with an address at 111 8th Avenue, New York, NY 10011. Our ordinary shares are listed on the regulated market of Euronext Brussels in Belgium under ISIN NL0010832176 under the symbol “ARGX” since 2014 and ADSs, each representing one ordinary share in argenx (or a right to receive such share), are listed on the Nasdaq Global Select Market (Nasdaq) under the symbol “ARGX” since 2017. argenx SE is the parent entity of the Group and the sole shareholder of: • argenx B.V., a private company with limited liability (besloten vennootschap/société à responsabilité limitée) incorporated under the laws of Belgium, having its registered seat in Zwijnaarde, Belgium and its address at Industriepark-Zwijnaarde 7, 9052 Zwijnaarde, Belgium. argenx B.V. is the sole shareholder of: ◦ argenx Australia Pty. Ltd.,incorporated under the laws of Australia, having its registered office and address at Level 14, 2 Riverside Quay, Melbourne VIC 3006, Australia; argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Company Profile 19

◦ argenx Austria Services GmbH, incorporated under the laws of Austria, having its registered office and address at Graben 19, 4th & 5th floor Vienna A-1010 Austria; ◦ argenx Benelux B.V. (prior to October 31, 2022 known as argenx IIP BV), incorporated under the laws of Belgium, having its registered seat in Zwijnaarde, Belgium and its address at Industriepark- Zwijnaarde 7, 9052 Zwijnaarde, Belgium; ◦ argenx Biotechnology (Shanghai) Co., Ltd., incorporated under the laws of China, having its registered office and address at Room 301-3, No. 481-479 Ping Xing Guan Road, Jingan District, Shanghai, China; ◦ argenx Brasil Produtos Farmacêuticos Ltda, incorporated under the laws of Brazil, having its registered office in Sao Paulo, Brazil and its address at Estrade da Lagoinha, 489 – Bloco 4, Bairro Lagoa CEP 06730-000, City of Vargem Grande Paulista, Sao Paulo, Brazil; ◦ argenx Canada Inc., incorporated under the laws of Ontario, having its registered office in Ontario, Canada and its address at 9131 Keele Street Suite A4, Vaughan, Ontario, Canada, L4K 0G7; ◦ argenx France SAS, incorporated under the laws of France, having its registered office in Issy-les- Moulineaux, France and its address at 24 rue Gouverneur Général Félix Éboué, 92130 Issy-les- Moulineaux, France; ◦ argenx Germany GmbH, incorporated under the laws of Germany, having its registered office in Munich, Germany and its address at Konrad-Zuse-Platz 8, 81829 Munich, Germany; ◦ argenx Italy S.r.l., incorporated under the laws of Italy, having its registered office in Milan, Italy and its address at Largo Francesco Richini 6 CAP, 20122 Milan, Italy; ◦ argenx Japan KK., incorporated under the laws of Japan, having its registered office in Tokyo, Japan and its address at HULIC JP Akasaka Building 2-5-8, Akasaka, Minato-ku, Tokyo, 107-0052, Japan; ◦ argenx Netherlands Services B.V., incorporated under the laws of the Netherlands having its registered office at Laarderhoogtweg 25, 1101 EB Amsterdam, the Netherlands; ◦ argenx Spain S.L., incorporated under the laws of Spain, having its registered office in Madrid, Spain and its address at Paseo dela Castellana 200, Planta 8a, Oficina 819, 28046 Madrid, Spain, with the branch office: argenx Spain S.L. - Sucursal em Portugal, organized under the laws of Portugal, having its registered office and address at Palácio Sottomayor, Rua Sousa Martins, nº1, 1º esquerdo 1050 217, Lisboa, Portugal; ◦ argenx Switzerland, S.A., incorporated under the laws of Switzerland, having its registered office in Geneva, Switzerland and its address at Rue du Pré-de-la-Bichette 1, 1202 Geneva, Switzerland; ◦ argenx UK Ltd., incorporated under the laws of the UK, having its registered office in Gerrards Cross, UK and its address at Spaces Gerrards Cross Chalfont Park, Building 1 Gerrards Cross, SL9 0BG, UK; ◦ argenx US, Inc., incorporated under the laws of the state of Delaware, U.S., having its registered office in Wilmington, Delaware and its address at 33 Arch Street, Boston, Massachusetts 02110and; ◦ Broteio Pharma B.V., incorporated under the laws of the Netherlands, having its registered office at Laarderhoogtweg 25, 1101 EB Amsterdam, the Netherlands. The following chart provides an overview of the Group as of the date of this Annual Report. Percentages refer to both the share of capital and voting rights. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Company Profile 20
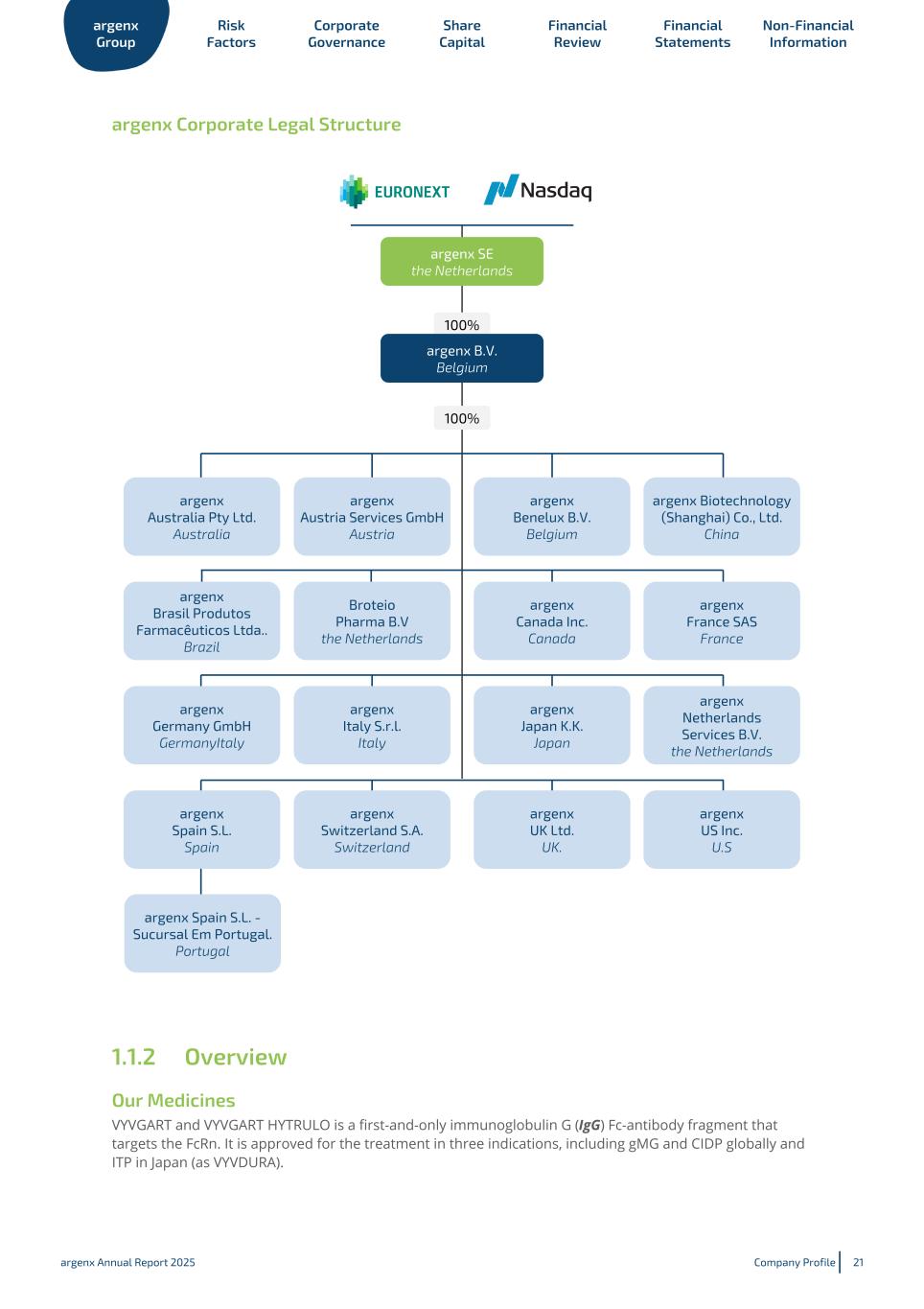
argenx Corporate Legal Structure 1.1.2 Overview Our Medicines VYVGART and VYVGART HYTRULO is a first-and-only immunoglobulin G (IgG) Fc-antibody fragment that targets the FcRn. It is approved for the treatment in three indications, including gMG and CIDP globally and ITP in Japan (as VYVDURA). argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Company Profile 21 argenx SE the Netherlands 100% argenx B.V. Belgium argenx Australia Pty Ltd. Australia argenx Austria Services GmbH Austria argenx Benelux B.V. Belgium argenx Biotechnology (Shanghai) Co., Ltd. China 100% argenx Germany GmbH GermanyItaly argenx Brasil Produtos Farmacêuticos Ltda.. Brazil Broteio Pharma B.V the Netherlands argenx Canada Inc. Canada argenx France SAS France argenx Netherlands Services B.V. the Netherlands argenx Spain S.L. Spain argenx Japan K.K. Japan argenx US Inc. U.S argenx Switzerland S.A. Switzerland argenx UK Ltd. UK. argenx Italy S.r.l. Italy argenx Spain S.L. - Sucursal Em Portugal. Portugal

Our Pipeline • efgartigimod is an IgG1 antibody Fc fragment that has been engineered for increased affinity to FcRn compared to endogenous IgG. Efgartigimod selectively reduces IgG by blocking FcRn-mediated IgG recycling without impacting antibody production or affecting other parts of the immune system. It is approved in three indications, including gMG, CIDP and ITP, and is being evaluated in more than ten additional serious autoimmune indications. • empasiprubart (C2 inhibitor) is a novel complement inhibitor targeting C2, blocking the function of both the classical and lectin pathways while leaving the alternative pathway intact. We believe empasiprubart has the potential to be a pipeline-in-a-product candidate and is being evaluated in two indications currently in Phase 3 clinical trials. • adimanebart (MusK agonist): adimanebart is an agonist SIMPLE ANTIBODY™ to the MuSK receptor with potential in multiple neuromuscular indications. It is currently in clinical trials for CMS (Phase 3 clinical trial) and SMA (Phase 2). • Earlier Stage Programs: ◦ Two future FcRn molecules are progressing: ARGX-213, an FcRn-targeted antibody engineered for half-life extension and sustained IgG reduction, and ARGX-124, a first-in-class FcRn pipeline candidate. ◦ ARGX-109 (targeting IL-6) and ARGX-121 (a first-in-class molecule targeting immunoglobulin A (IgA)) are also progressing. ◦ Entered into a research collaboration with Tensegrity Pharma, including an option for future acquisition, to advance Tensegrity’s lead program TSP-101 in autoimmune disease and other indications. ◦ Three new molecules expected to enter Phase 1 clinical trials in 2026, including ARGX-118, a first-in- class molecule targeting Galectin-10, ARGX-125, a first-in-class bispecific antibody, and TSP-101, targeting Fn14. • In addition to our wholly-owned pipeline, we have candidates that emerged from our IIP that we out- licensed to a partner for further development and for which we have milestone, royalty or profit-share agreements. These candidates include, amongst others: cusatuzumab (anti-CD70 antibody – OncoVerity), ARGX-112 (LP-0145 – anti-IL-22R antibody – LEO Pharma), ARGX-114 (AGMB-101 – agonistic anti-MET antibody – Agomab) and ARGX-115 (ABBV-151 – anti-GARP antibody – AbbVie). Immunology Innovation Program (IIP) Our IIP is the engine behind our robust and expansive pipeline. By fostering deep, ongoing collaboration between leading academic researchers and our in-house antibody engineers, we aim to translate breakthrough science into first-in-class therapies across multiple indications. This co-creation model has enabled every candidate in our wholly owned and partnered pipelines to emerge from IIP collaborations, underscoring our ability to identify and advance novel targets with speed and precision. Our approach is designed for scale and sustainability: we run parallel development programs, optimize trial design for efficiency, and maintain a relentless focus on unmet patient needs. This strategy has delivered measurable results – accelerating our path to profitability, driving strong commercial growth, and positioning argenx as a leader in immunology innovation. By integrating the aspirations of patients and the insights of healthcare professionals into every stage of discovery and development, we are not only building a differentiated pipeline but also setting new standards for impact and value creation in the sector. We bring to the collaboration our unique suite of antibody discovery and antibody engineering technologies and experience in clinical development to complement our partners’ expertise in disease and target biology. Our suite of technologies includes amongst others our SIMPLE ANTIBODY™ platform technology and NHANCE™, ABDEG™, POTELLIGENT®, and DHS mutations that focus on engineering the Fc region of antibodies in order to augment their intrinsic therapeutic properties. Our Suite of Technologies • SIMPLE ANTIBODY™ platform technology: Our proprietary SIMPLE ANTIBODY™ platform technology, based on the powerful llama immune system, allows us to exploit novel and complex disease biology targets. The platform sources antibody variable regions (V-regions) from the immune system of outbred llamas, each of which has a different genetic background. The llama produces highly diverse panels of argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Company Profile 22

antibodies with a high human homology, or similarity, in their V-regions when immunized with targets of human disease. Our SIMPLE ANTIBODY™ platform technology allows us to access and explore a broad target universe while potentially minimizing the long timelines associated with generating antibody candidates using traditional methods. • NHANCE™, ABDEG™, POTELLIGENT®, and DHS mutations focus on engineering the Fc region of antibodies in order to augment their intrinsic therapeutic properties. In addition, we obtained a non- exclusive research license and option from Chugai Pharmaceutical Co., Ltd. for the SMART-Ig® (“Recycling Antibody” and part of “Sweeping Antibody”) and ACT-Ig® (Antibody half-life extending) technologies. These technologies are designed to enable us to expand the therapeutic index of our product candidates, which is the ratio between toxic and therapeutic dose, by potentially modifying their half-life, tissue penetration, rate of disease target clearance and potency. 1.2 Strategy and Objectives 1.2.1 Company’s Strategies Our objective is to transform the lives of at least 50,000 patients and their communities before 2030 by providing them with life-changing medicines built on scientific breakthroughs in immunology. To reach this, we aim to deliver on a set of different goals: • Transform the lives of 50,000 patients, by redefining treatment expectations in MG & CIDP and delivering at least eight additional labeled indications and a second self-administered FcRn medicine by 2030. • Be the precision complement inhibitor that sets a new SOC and improves patient experience and outcomes across indications, with 3+ labeled indications and five total indications in development by 2030. • Be the leader in neuromuscular junction (NMJ) therapeutics, redefining patient expectations and reigniting hope in one labeled indication and at least four total indications by 2030. • Expand our pipeline of transformational innovation to enable consistent cadence of value creation for patients; >5 new molecules in late stage by 2030. • Scale in The argenx Way to remain a unique, independent company. • Solidify our place in the biotech ecosystem as the benchmark for entrepreneurial science delivering value for patients. 1.2.2 Competitive position We participate in a highly innovative industry characterized by a rapidly growing understanding of disease biology, quickly changing technologies, strong intellectual property barriers to entry, and a multitude of companies involved in the creation, development and commercialization of novel therapeutics. Many of these companies are highly sophisticated and often strategically collaborate with each other. Competition in the autoimmune field is intense and involves multiple monoclonal antibodies (mAbs), other biologics and small molecules either already marketed or in development by many different companies, including large pharmaceutical companies. We compete with a wide range of biopharmaceutical companies that are developing products for the treatment of gMG, CIDP, ITP and other autoimmune diseases, including products that are in the same class as VYVGART, as well as products that are similar to some of our product candidates. We are aware of several FcRn inhibitors that are in clinical development or marketed. In addition, we may face future competition from biosimilar versions of approved biologics in the autoimmune and immunology fields. The regulatory frameworks in the United States, Europe and other key markets could evolve in ways that may facilitate the entry of biosimilars once reference products lose market exclusivity. While FcRn inhibitors are a relatively new therapeutic class, the broader biologics market argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Company Profile 23

has seen an increase in biosimilar development and commercial activity, supported by maturing regulatory pathways, expanding manufacturing capabilities and ongoing payer and health-system initiatives aimed at reducing the cost of care. Competitive product launches may erode future sales of our products, including our existing products and those currently under development, or result in unanticipated product obsolescence. Such launches continue to occur, and potentially competitive products are in various stages of development. We could also face competition for use of limited international infusion sites, particularly in new markets as competitors launch new products. We cannot predict with accuracy the timing or impact of the introduction of competitive products that treat diseases and conditions like those treated by our products or product candidates. In addition, our competitors compete with us to recruit and retain qualified scientific and management personnel, establish clinical trial sites and patient registration for clinical trials, as well as in acquiring technologies complementary to, or necessary for, the development of our products. Please see the risk factor titled ”We face significant competition for our drug discovery and development efforts”. We compete in this dynamic landscape by aiming to advancing differentiated, first-in-class and best-in-class therapies grounded in novel biology, enabled by our deep scientific expertise and our technology platforms. Our strategy is to innovate ahead of the field—identifying new pathways, engineering unique mechanisms of action, and continuously expanding our understanding of Fc-mediated biology and related immunology. We also actively protect and strengthen our intellectual property through a comprehensive global portfolio of patents, trade secrets and proprietary know-how covering our product candidates, platforms and manufacturing processes. This commitment to scientific innovation and robust IP protection is central to sustaining our competitive position and delivering long-term value. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Strategy and Objectives 24
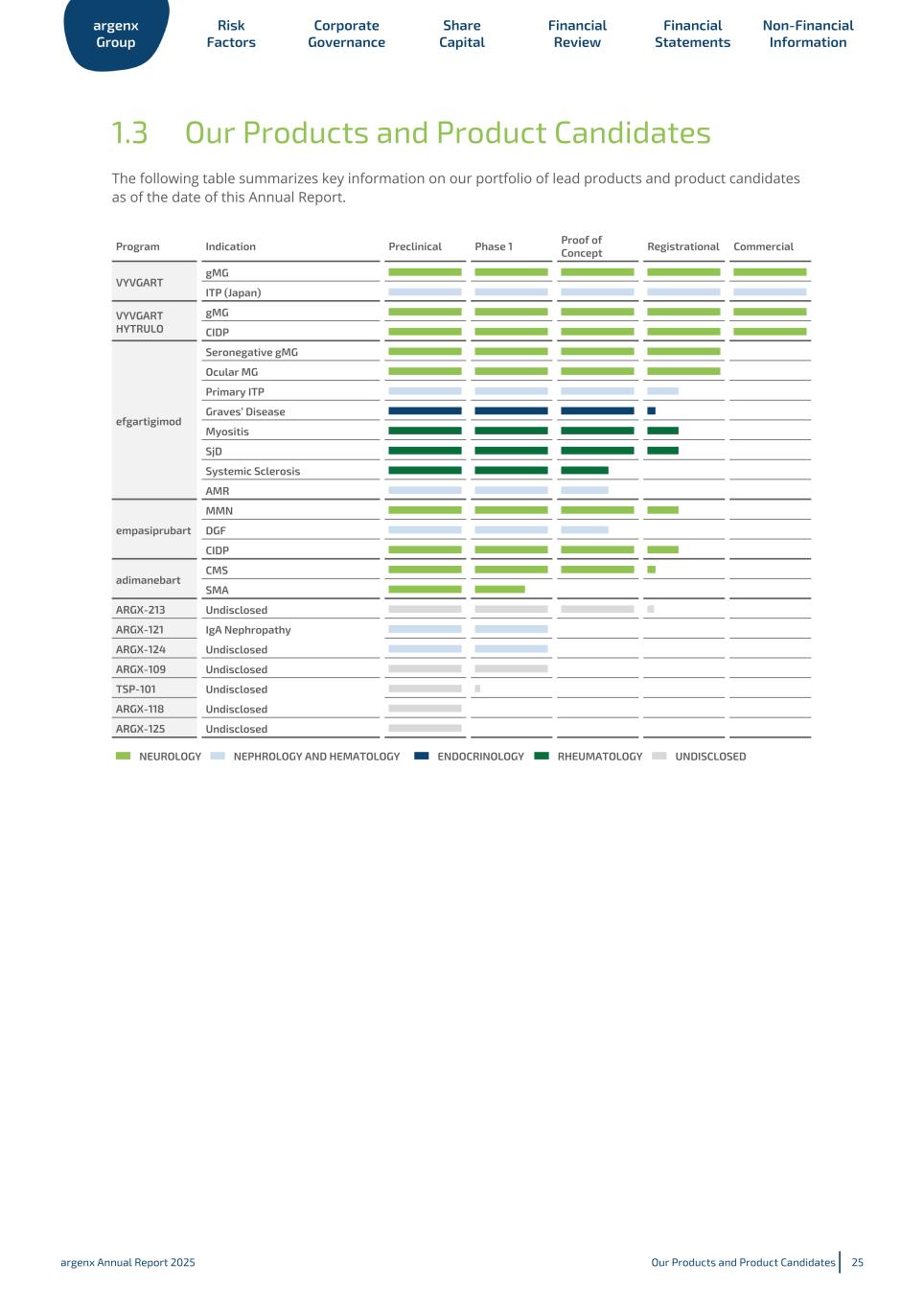
1.3 Our Products and Product Candidates The following table summarizes key information on our portfolio of lead products and product candidates as of the date of this Annual Report. Program Indication Preclinical Phase 1 Proof of Concept Registrational Commercial VYVGART gMG ITP (Japan) VYVGART HYTRULO gMG CIDP efgartigimod Seronegative gMG Ocular MG Primary ITP Graves’ Disease Myositis SjD Systemic Sclerosis AMR empasiprubart MMN DGF CIDP adimanebart CMS SMA ARGX-213 Undisclosed ARGX-121 IgA Nephropathy ARGX-124 Undisclosed ARGX-109 Undisclosed TSP-101 Undisclosed ARGX-118 Undisclosed ARGX-125 Undisclosed NEUROLOGY NEPHROLOGY AND HEMATOLOGY ENDOCRINOLOGY RHEUMATOLOGY UNDISCLOSED argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Our Products and Product Candidates 25
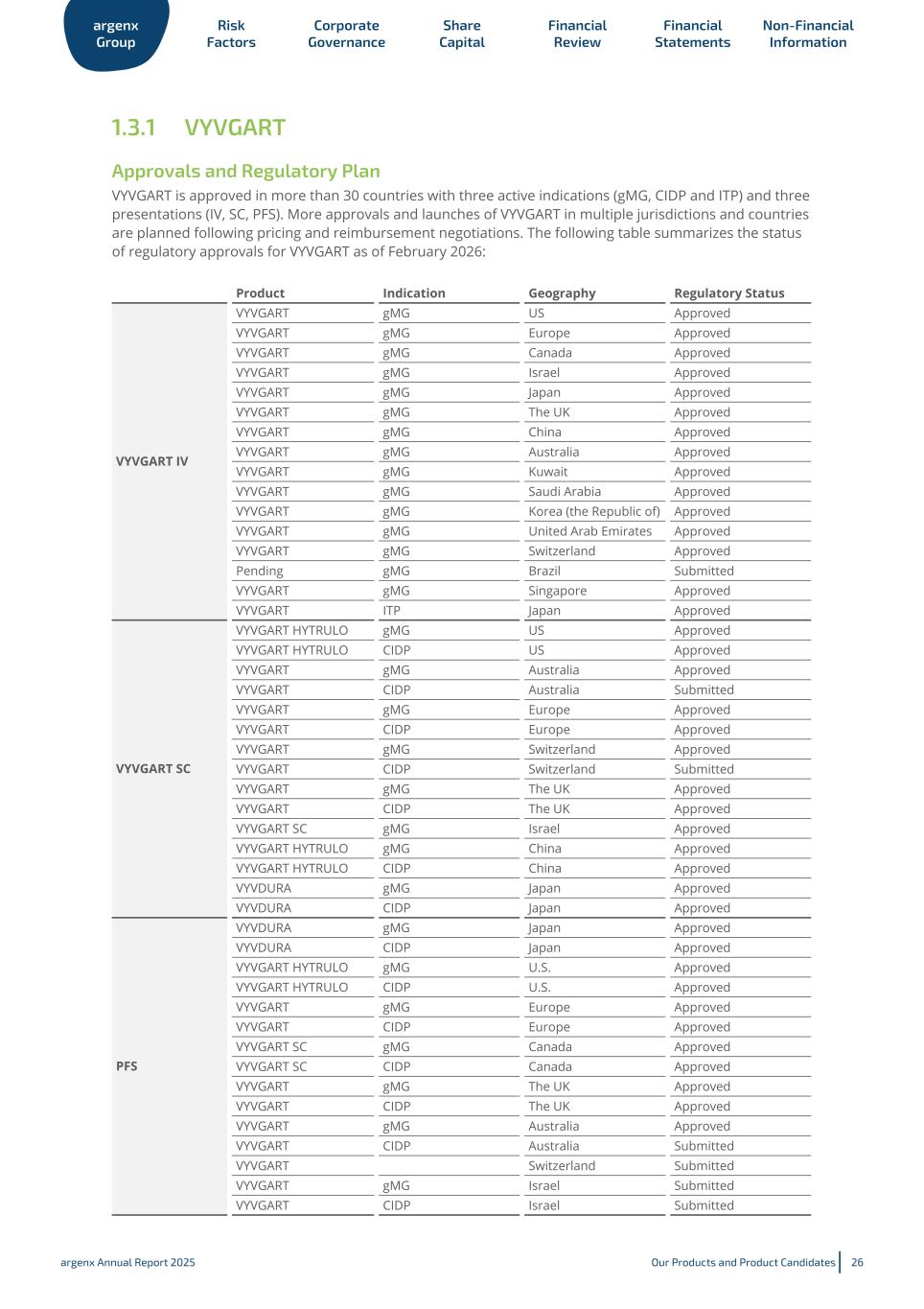
1.3.1 VYVGART Approvals and Regulatory Plan VYVGART is approved in more than 30 countries with three active indications (gMG, CIDP and ITP) and three presentations (IV, SC, PFS). More approvals and launches of VYVGART in multiple jurisdictions and countries are planned following pricing and reimbursement negotiations. The following table summarizes the status of regulatory approvals for VYVGART as of February 2026: Product Indication Geography Regulatory Status VYVGART IV VYVGART gMG US Approved VYVGART gMG Europe Approved VYVGART gMG Canada Approved VYVGART gMG Israel Approved VYVGART gMG Japan Approved VYVGART gMG The UK Approved VYVGART gMG China Approved VYVGART gMG Australia Approved VYVGART gMG Kuwait Approved VYVGART gMG Saudi Arabia Approved VYVGART gMG Korea (the Republic of) Approved VYVGART gMG United Arab Emirates Approved VYVGART gMG Switzerland Approved Pending gMG Brazil Submitted VYVGART gMG Singapore Approved VYVGART ITP Japan Approved VYVGART SC VYVGART HYTRULO gMG US Approved VYVGART HYTRULO CIDP US Approved VYVGART gMG Australia Approved VYVGART CIDP Australia Submitted VYVGART gMG Europe Approved VYVGART CIDP Europe Approved VYVGART gMG Switzerland Approved VYVGART CIDP Switzerland Submitted VYVGART gMG The UK Approved VYVGART CIDP The UK Approved VYVGART SC gMG Israel Approved VYVGART HYTRULO gMG China Approved VYVGART HYTRULO CIDP China Approved VYVDURA gMG Japan Approved VYVDURA CIDP Japan Approved PFS VYVDURA gMG Japan Approved VYVDURA CIDP Japan Approved VYVGART HYTRULO gMG U.S. Approved VYVGART HYTRULO CIDP U.S. Approved VYVGART gMG Europe Approved VYVGART CIDP Europe Approved VYVGART SC gMG Canada Approved VYVGART SC CIDP Canada Approved VYVGART gMG The UK Approved VYVGART CIDP The UK Approved VYVGART gMG Australia Approved VYVGART CIDP Australia Submitted VYVGART Switzerland Submitted VYVGART gMG Israel Submitted VYVGART CIDP Israel Submitted argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Our Products and Product Candidates 26

Commercialization We have established our own sales force in the U.S., Japan, Europe and Canada for VYVGART for the treatment of gMG and CIDP (where approved). We plan to expand our own sales and marketing capabilities and promote our products and product candidates in other regions if we decide there is a business case to do so after regulatory approval has been obtained. Development and commercialization may also be done through collaborations with third parties. In January 2021, we entered into an exclusive out-license agreement with Zai Lab (Zai Lab Agreement), a commercial- stage biopharmaceutical company, for the development and commercialization of efgartigimod in Greater China, (which includes Mainland China, Hong Kong, Taiwan and Macau, Greater China). Zai Lab announced approval of VYVGART in Mainland China in June 2023 for the treatment of adult gMG patients and in 2024 Zai Lab also announced the approval of VYVGART SC for gMG and CIDP. Under the Zai Lab Agreement, we received and continue to be eligible for certain sales-based milestone payments and royalties based on annual product net sales of efgartigimod in Greater China. We intend to continue expanding into new markets and will evaluate the most appropriate commercialization approach for each territory, whether through our own commercial organization or through additional distribution partnerships. In the U.S., argenx advertises certain products via digital and traditional media channels, including the internet and television. For a discussion of total revenues by geographic market, please see “Note 16 Segment Reporting” in our consolidated financial statements which are included in our Annual Report for the period ended December 31, 2025. Pre-Approval Access Program We are committed to improving the lives of people suffering from rare diseases. We are driven to discover new treatment approaches fueled by the resilience of patients to urgently deliver them. We aim to do this in partnership; we listen to patients, supporters and advocacy communities, and we hear their stories. Their insights guide us as we develop our investigational therapies and motivate us to advance the understanding of rare diseases. We have a Pre-Approval Access program (PAA) for patients with gMG which opened on February 21, 2021 for patients who are unable to participate in an ongoing clinical trial. In 2024, we approved access to this PAA for over 403 gMG patients in 14 countries. The PAA program remains open in countries where VYVGART is not yet launched or reimbursed. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Our Products and Product Candidates 27

1.3.2 efgartigimod (ARGX-113) Development Mechanism of Action As shown in Figure 1, efgartigimod is a human IgG1 Fc fragment equipped with our ABDEG™ mutations that is designed to target the FcRn and reduce IgG. FcRn is foundational to the immune system and functions to recycle IgG, extending its serum half-life over other IgGs that are not recycled by FcRn. IgGs that bind to FcRn are rescued from lysosomal degradation. By binding to FcRn, efgartigimod can reduce IgG recycling and increase IgG degradation. Figure 1: efgartigimod’s mechanism of action blocks the recycling of IgG antibodies and removes them from circulation. FcRn, neonatal Fc receptor; Ig, immunoglobulin; LDL, low-density lipoprotein. 1) Ulrichts P, et al. J Clin Invest. 2018;128:4372–86 6) Roopenian DC, Akilesh S. Nat Rev Immunol. 2007;7:715–25. 7) Ward ES, Ober RJ. Trends Pharmacol Sci. 2018;39:892–904. Formulations Overview We are developing two formulations of efgartigimod to address the needs of patients, physicians, and payers across indications and geographies, including efgartigimod IV (VYVGART) and efgartigimod SC (VYVGART SC). argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Our Products and Product Candidates 28
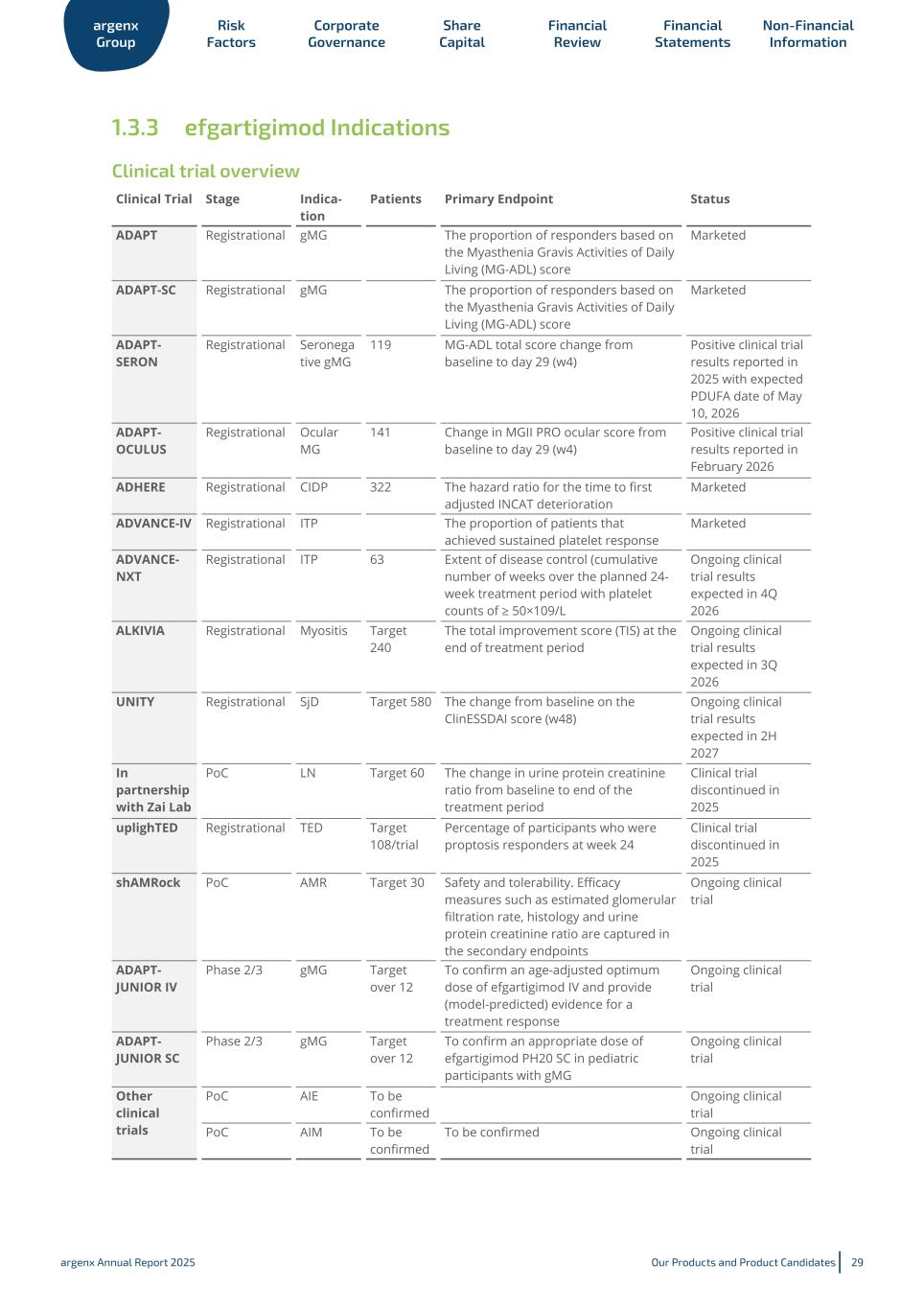
1.3.3 efgartigimod Indications Clinical trial overview ADAPT Registrational gMG The proportion of responders based on the Myasthenia Gravis Activities of Daily Living (MG-ADL) score Marketed ADAPT-SC Registrational gMG The proportion of responders based on the Myasthenia Gravis Activities of Daily Living (MG-ADL) score Marketed ADAPT- SERON Registrational Seronega tive gMG 119 MG-ADL total score change from baseline to day 29 (w4) Positive clinical trial results reported in 2025 with expected PDUFA date of May 10, 2026 ADAPT- OCULUS Registrational Ocular MG 141 Change in MGII PRO ocular score from baseline to day 29 (w4) Positive clinical trial results reported in February 2026 ADHERE Registrational CIDP 322 The hazard ratio for the time to first adjusted INCAT deterioration Marketed ADVANCE-IV Registrational ITP The proportion of patients that achieved sustained platelet response Marketed ADVANCE- NXT Registrational ITP 63 Extent of disease control (cumulative number of weeks over the planned 24- week treatment period with platelet counts of ≥ 50×109/L Ongoing clinical trial results expected in 4Q 2026 ALKIVIA Registrational Myositis Target 240 The total improvement score (TIS) at the end of treatment period Ongoing clinical trial results expected in 3Q 2026 UNITY Registrational SjD Target 580 The change from baseline on the ClinESSDAI score (w48) Ongoing clinical trial results expected in 2H 2027 In partnership with Zai Lab PoC LN Target 60 The change in urine protein creatinine ratio from baseline to end of the treatment period Clinical trial discontinued in 2025 uplighTED Registrational TED Target 108/trial Percentage of participants who were proptosis responders at week 24 Clinical trial discontinued in 2025 shAMRock PoC AMR Target 30 Safety and tolerability. Efficacy measures such as estimated glomerular filtration rate, histology and urine protein creatinine ratio are captured in the secondary endpoints Ongoing clinical trial ADAPT- JUNIOR IV Phase 2/3 gMG Target over 12 To confirm an age-adjusted optimum dose of efgartigimod IV and provide (model-predicted) evidence for a treatment response Ongoing clinical trial ADAPT- JUNIOR SC Phase 2/3 gMG Target over 12 To confirm an appropriate dose of efgartigimod PH20 SC in pediatric participants with gMG Ongoing clinical trial Other clinical trials PoC AIE To be confirmed Ongoing clinical trial PoC AIM To be confirmed To be confirmed Ongoing clinical trial Clinical Trial Stage Indica tion Patients Primary Endpoint Status argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Our Products and Product Candidates 29

gMG Overview gMG is a rare, chronic autoimmune disease in which pathogenic IgG autoantibodies disrupt neuromuscular signaling, leading to fluctuating and sometimes life-threatening muscle weakness. Autoantibodies block or remove acetylcholine receptors and activate complement, damaging the neuromuscular junction. MG often begins with ocular symptoms such as ptosis and diplopia, and approximately 85% of patients progress to generalized MG (gMG), which can impair bulbar, limb, and respiratory function. Respiratory crises occur in 15–20% of patients. MG prevalence in the U.S. is estimated at approximately 20 per 100,000, and roughly 85% of gMG patients have detectable AChR antibodies. efgartigimod has demonstrated consistent and robust clinical benefit across MG populations. Pivotal ADAPT data formed the basis for global approvals of VYVGART IV, and positive ADAPT-SC results supported approval of the subcutaneous formulation. In 2025, we reported positive topline results from ADAPT-SERON, our Phase 3 clinical trial in anti-AChR antibody-negative gMG, demonstrating a clear treatment effect and reinforcing the broad applicability of FcRn across MG subtypes. These data support our supplemental regulatory submission to expand VYVGART into the seronegative population, which was accpeted for priority review with an expected PDUFA target action date of May 10, 2026. We also reported positive data from our ADAPT OCULUS clinical trial, which met its primary endpoint, showing that patients living with oMG and treated with VYVGART demonstrated statistically significant improvement from baseline in Myasthenia Impairment Index (MGII) Patient Reported Outcome (PRO) ocular scores at Week 4 compared to placebo. The results support our supplemental regulatory submissions to expand VYVGART into the ocular MG population. We also have ongoing clinical trials in pediatric gMG patients (ADAPT- JUNIOR) with efgartigimod IV and efgartigimod SC. CIDP Overview CIDP is a chronic autoimmune disorder of peripheral nerves and nerve roots caused by an autoimmune- mediated destruction of the myelin sheath, or myelin producing cells, insulating the axon of the nerves and enabling speed of signal transduction. The cause of CIDP is unknown, but abnormalities in both cellular and humoral immunity have been shown. CIDP is a chronic and progressive disease: onset and progression occur over at least eight weeks in contrast with the more acute Guillain-Barré-syndrome. Demyelination and axonal damage in CIDP lead to loss of sensory and/or motor neuron function, which can lead to weakness, sensory loss, imbalance and/or pain. The U.S. prevalence is estimated at approximately 42,000 patients, of whom roughly 24,000 receive treatment. Most patients rely on IVIg as first-line therapy, while glucocorticoids, plasma exchange, and other immunosuppressants are used less frequently given safety, tolerability, and access limitations. In July 2023, the pivotal ADHERE clinical trial demonstrated that VYVGART SC significantly reduced the risk of relapse compared to placebo and provided evidence that pathogenic IgG autoantibodies play an important role in CIDP biology. Sixty-seven percent of patients entering the open-label Stage A improved clinically, and efgartigimod SC was well tolerated with a safety profile consistent with prior clinical trials. Nearly all eligible patients (99%) continued into the ADHERE-Plus OLE. Based on these data, VYVGART SC received regulatory approvals in the U.S. in June 2024, in China in November 2024, and in Japan in December 2024, with regulatory review ongoing in additional jurisdictions, including the EU. Primary ITP Overview Primary ITP is an acquired autoimmune bleeding disorder, characterized by a low platelet count (<100×109/L) in the absence of other causes associated with thrombocytopenia. In most patients, IgG autoantibodies directed against platelet receptors can be detected. They accelerate platelet clearance and destruction, inhibit platelet production, and impair platelet function, resulting in increased risk of bleeding and impaired quality of life. Primary ITP is differentiated from secondary ITP, which is associated with other illnesses, such as infections or autoimmune diseases, or which occurs after transfusion or taking other drugs, such as cancer drugs. Platelet deficiency, or thrombocytopenia, can cause bleeding in tissues, bruising and slow blood clotting after injury. Patients may suffer from depression and fatigue as well as side effects of existing therapies, impairing their quality of life. Current therapeutic approaches include non- argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Our Products and Product Candidates 30

specific immunosuppression (e.g., steroids and rituximab), inhibition of platelet clearance (e.g., splenectomy, IVIg, anti-D globulin, and spleen tyrosine kinase inhibitor fostamatinib13) or stimulation of platelet production (e.g., thrombopoietin receptor agonist TPO-RA). Splenectomy remains the only treatment that provides sustained remission off therapy for one year or longer for a high proportion of patients. ITP affects approximately 72,000 patients in the U.S. In 2022, the Phase 3 ADVANCE (IV) clinical trial met its primary endpoint, demonstrating that a higher proportion of chronic ITP patients receiving efgartigimod achieved a sustained platelet count response compared to placebo. These results supported approval of efgartigimod for ITP in Japan. In 2023, the accompanying subcutaneous clinical trial, ADVANCE-SC, did not meet its primary endpoint. To fulfill the requirement for two well-controlled trials needed for global registration, argenx is now conducting ADVANCE-NEXT, a Phase 3, randomized, double-blinded, placebo-controlled trial evaluating efgartigimod IV in adults with primary ITP. ADVANCE-NEXT remains ongoing, with topline Phase 3 results expected in the fourth quarter of 2026. AIM Overview AIM are a rare and heterogeneous group of autoimmune diseases that can affect muscle alone or multiple organ systems, including the skin, joints, lungs, gastrointestinal tract, and heart. These conditions are severe, disabling, and materially impact quality of life. Advances in understanding disease biology and the discovery of characteristic autoantibodies have led to clearer differentiation of AIM into clinically meaningful subtypes, including immune-mediated necrotizing myopathy (IMNM), antisynthetase syndrome (ASyS), and dermatomyositis (DM). Each subtype presents with distinct autoantibody profiles and manifestations, though proximal muscle weakness remains a defining feature across AIM. Today, there are no FDA-approved therapies for IMNM or ASyS, and treatment is largely dependent on steroids or broad immunosuppressants; IVIg was approved for DM in 2021. argenx is advancing the registrational ALKIVIA clinical trial of efgartigimod SC for the treatment of AIM. ALKIVIA is a seamless Phase 2/3 clinical trial enrolling approximately 240 patients across IMNM, ASyS, and DM, with Total Improvement Score (TIS) as the primary endpoint and a broad set of functional and quality-of-life secondary measures. In November 2024, following achievement of statistical significance on the primary endpoint in the Phase 2 portion and consistent improvement across all six core components of the TIS, argenx announced a ‘GO’ decision to proceed with the Phase 3 portion in all three AIM subtypes. Safety and tolerability were consistent with the known profile of efgartigimod. ALKIVIA remains ongoing, with topline Phase 3 results expected in the third quarter of 2026. SjD Overview SjD is a chronic, progressive autoimmune disease, characterized by lymphocytic infiltration and progressive destruction of exocrine glands. B-cells play a pivotal role in the development of the disease and this results amongst others in production of IgG autoantibodies, especially those which target SSA/Ro, SSB/La ribonuclear complexes. In addition to symptoms of dry eyes, dry mouth, chronic pain and fatigue, a substantial subset of patients suffer from extraglandular systemic disease. There are no FDA-approved treatments currently registered for the treatment of SjD. argenx is advancing the registrational UNITY clinical trial of efgartigimod SC for the treatment of SjD. UNITY is a Phase 3, randomized, placebo-controlled, double-blind clinical trial assessing the safety and efficacy of efgartigimod SC in 480 patients with at least moderate systemic disease (ClinESSDAI ≥6) who are on stable background therapy and positive for anti-SSA/Ro. After the 48-week treatment period, eligible participants may roll over into an OLE. The primary endpoint is change from baseline in clinESSDAI, with key secondary endpoints focused on patient-reported outcomes, ESSDAI, and STAR. UNITY remains ongoing, with topline Phase 3 results expected in the second half of 2027. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Our Products and Product Candidates 31

1.3.4 empasiprubart (ARGX-117) Development Mechanism of Action empasiprubart is a differentiated therapeutic mAb targeting C2 equipped with our proprietary NHANCE™ mutations. By addressing a novel target at the intersection of the complement and lectin pathways of the complement cascade, we believe empasiprubart represents a broad pipeline opportunity across several severe autoimmune indications. Activation of the classical and lectin pathway of complement may contribute to tissue damage and organ dysfunction in a number of autoimmune inflammatory diseases and ischemia-reperfusion conditions. Targeting C2 also leaves the alternative pathway of the complement system intact, which is an important component of the innate defense system. empasiprubart exhibits both pH- and calcium dependent binding. These unique characteristics enable empasiprubart to capture free C2 in circulation and release it in the endosome to be sorted for degradation in the lysosome. empasiprubart is equipped with NHANCE™ mutations increasing its affinity for FcRn and allowing it to recycle back into circulation to capture more C2. In addition to an IV formulation, we have exclusive access to Halozyme’s ENHANZE® SC drug delivery technology for the C2 target. Figure 2: empasiprubart mechanism of action. C2, complement component 2; FcRn, neonatal Fc receptor; IgG, immunoglobulin G. 1) Van de Walle I, et al. J Allergy Clin Immunol. 2021;147:1420–9. 2) Vaccaro C, et al. Proc Natl Acad Sci. 2006;103:18709–14. 3) Brinkhaus M, et al. Nat Commun. 2022;13:6073. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Our Products and Product Candidates 32

empasiprubart Indications MMN Overview MMN is a debilitating neuromuscular autoimmune disorder that is characterized by slowly progressive muscle weakness due to motor neuron degeneration. It mainly affects hands and forearms, mainly in males, and the median age of diagnosis is around 40 years. Diagnosis takes about a year and a half and is often misdiagnosed as ALS. There are estimated to be around 12,000 patients across key markets. Specific pathophysiologic characteristics of MMN include the presence of IgM autoantibodies against the ganglioside GM1 and conduction block, i.e., impaired propagation of action potentials along the axon. GM1 is widely expressed in the nervous system by neurons, particularly around the nodes of Ranvier, and Schwann cells. IVIg is the only approved treatment for MMN and needs to be dosed frequently to address the disease’s progressive nature. Phase 2 POC ARDA Clinical Trial The Phase 2 POC ARDA clinical trial was a randomized, double-blinded, placebo-controlled multicenter clinical trial evaluating the safety and tolerability, efficacy, PK, PD, and immunogenicity of two dose regimens of empasiprubart in adults with MMN. Safety and tolerability were the primary endpoint and additional endpoints included time to IVIg retreatment, biomarker analyses of C2 levels, and changes in key functional scores (modified medical research council-10 sum score, grip strength, MMN-RODS) as well as several patient-reported quality-of-life measures (fatigue severity score (FSS), chronic acquired polyneuropathy patient-reported index (CAP-PRI), and patient global impression change scale). In 2024, argenx announced positive data from the first cohort (n=16), which were confirmed with the second cohort (n=16) in July 2024, establishing POC in MMN, with empasiprubart demonstrating a 91% reduction in the need for IVIg rescue compared to placebo [HR (95% CI)=0.09 (0.02; 0.44)] in cohort 1 and an 84% reduction in IVIg rescue compared to placebo [HR (95% CI)=0.16 (0.02; 1.54)] in cohort 2. Based on these results argenx initiated the EMPASSION Phase 3 clinical trial evaluating empasiprubart in MMN head to head with IVIg at the end of 2024. Phase 3 EMPASSION Clinical Trial Design A Phase 3, randomized, double-blinded, double-dummy clinical trial evaluating the efficacy and safety of empasiprubart versus intravenous immunoglobulin in adults with multifocal motor neuropathy. The clinical trial comprises a screening period of up to 15 weeks, including a minimum of 2 IVIg cycles; a 24-week (6-month), randomized, double-blinded, double-dummy treatment period (part A) evaluating the efficacy and safety of empasiprubart vs IVIg continuation; a 24-month OLE period (part B); and a 15-month safety follow-up period starting after the last dose of IMP. The primary objective is to demonstrate the efficacy of empasiprubart compared to IVIg in improving functional ability. This will be measured by change from baseline in the 25-item MMN-RODS centile score at week 24. Additional key secondary endpoints include changes in measurements on key functional scores (modified medical research council -14 sum score, grip strength) as well as patient-reported quality of life outcome measures (polyneuropathy patient- reported index, and values of the patient global impression change scale and evaluation of manual dexterity using 9HPT. DGF Delayed graft function (DGF), defined as the need for dialysis in the first week after kidney transplant, affects up to 40% of deceased-donor recipients and is associated with poorer long-term outcomes. Decision for Phase 2 VARVARA clinical trial is now expected mid-year 2026 to complete 52-week efficacy analysis argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Our Products and Product Candidates 33

CIDP Overview Please refer to Section “1.3.3 efgartigimod Indications” (CIDP) for more information on CIDP. Phase 3 EMVIGORATE and EMNERGIZE Clinical Trials argenx is advancing two Phase 3 clinical trials of empasiprubart in CIDP: EMVIGORATE and EMNERGIZE. EMVIGORATE is a head-to-head clinical trial comparing empasiprubart to IVIg in adults with CIDP. EMNERGIZE is a randomized, placebo-controlled clinical trial evaluating the efficacy and safety of empasiprubart. Topline results from both EMVIGORATE and EMNERGIZE are expected in the second half of 2027. 1.3.5 adimanebart (ARGX-119) Development adimanebart is a humanized agonist monoclonal antibody that specifically targets and activates MuSK to promote maturation and stabilization of the NMJ, with planned development across severe neuromuscular diseases including CMS, ALS, and SMA. It is the first highly specific agonist mAb targeting human MuSK and was developed using the SIMPLE ANTIBODY™ platform in collaboration with leading experts, with preclinical proof-of-concept demonstrated in a DOK7-CMS model. A Phase 3 clinical trial in CMS is expected to initiate in the third quarter of 2026, following positive results from the Phase 1b clinical trial. A proof-of-concept clinical trial is also ongoing in SMA. 1.4 Collaborations and licenses At argenx, our approach to collaboration and licensing is rooted in the conviction that progress accelerates when boundaries are challenged and expertise is shared. We follow a disciplined strategy to maximize the value of our pipeline. We retain full development and commercialization rights for programs where we believe our platform and capabilities can deliver the greatest impact, ensuring we capture the full value of our innovation. At the same time, we actively seek out partnerships with organizations that share our drive to redefine what’s possible, leveraging complementary strengths to unlock new opportunities for patients. Our licensing strategy is dynamic and pragmatic: we license out select intellectual property to expand the reach of our science, while we also in-license or acquire technologies and assets that can amplify our pipeline or accelerate development. We have partnered, and plan to continue to partner, to develop products and product candidates that we believe have promising utility in disease areas or have patient populations that may benefit from resources of other biopharmaceutical companies. We believe every agreement is shaped by a clear-eyed focus on execution, mutual benefit, and the potential to create lasting change. We aim to be disciplined in our diligence and financial commitments, but not at the expense of agility or ambition. By building alliances that transcend traditional hierarchies and by staying relentlessly focused on unmet needs, we are not just advancing our own portfolio, we are helping to reshape the landscape of immunology for the long term. We also have several license agreements in place, under which we license patents, patent applications and other intellectual property to third parties. We have also entered into several license agreements under which we license patents, patent applications and other intellectual property from third parties. License agreements can relate to research and development and/or commercialization of the relevant product candidates (and technologies) or products. The licensed intellectual property covers some of our product candidates and some of the antibody engineering technologies that we use. Some of these licenses impose various diligence and financial payment obligations on us. We expect to continue to enter into these types of license agreements in the future. We have entered into multiple collaboration agreements with pharmaceutical partners and license agreements, some of which are described below. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Our Products and Product Candidates 34

1.4.1 Our Strategic Partnership with Zai Lab for efgartigimod Pursuant to the Zai Lab Agreement, Zai Lab obtained the exclusive right to develop and commercialize efgartigimod in Greater China. Zai Lab will also contribute patients to our global Phase 3 clinical trials of efgartigimod. Our Zai Lab strategic collaboration allows us to accelerate development of efgartigimod into new autoimmune indications with Zai Lab taking operational leadership of selected Phase 2 POC Clinical trials. We are eligible to receive a one-time sales based milestone and tiered royalties based on annual net sales of efgartigimod in Greater China thereafter. 1.4.2 Our Exclusive License with Halozyme for ENHANZE® In February 2019, we entered into an in-license agreement with Halozyme for the use of certain patents, materials and know-how owned by Halozyme and relating to its ENHANZE®, for application in the field of prevention and treatment of human diseases (the ENHANZE® License Agreement). Pursuant to the ENHANZE® License Agreement, we were granted exclusive rights to apply ENHANZE® to biologic products against pre-specified targets, in order to research, develop and commercialize SC formulations of our therapeutic antibody-based product candidates. Our first therapeutic target for which we received an exclusive license from Halozyme was FcRn, which allows us to apply ENHANZE® to efgartigimod and any other product candidates selective and specific for FcRn. Moreover, the breadth of our exclusive license to FcRn precludes either Halozyme itself or any of its current or future partners from utilizing ENHANZE® in the context of an FcRn-targeted product. Our second therapeutic target for which we received an exclusive license from Halozyme was human C2 associated with the product candidate empasiprubart, which is being developed to treat severe autoimmune diseases. Pursuant to the ENHANZE® License Agreement, we also have the right to nominate future targets for an exclusive ENHANZE® license if the target in question has not already been licensed by Halozyme or is not already being pursued by Halozyme. We have expanded our collaboration with Halozyme for ENHANZE® drug delivery technology to additional targets for a total of six, including FcRn and C2. We may terminate the ENHANZE® License Agreement at any time, either in its entirety or on a target-by- target basis, by sending Halozyme prior written notice. Absent early termination, the ENHANZE® License Agreement will automatically expire upon the expiry of our royalty payment obligations under the agreement. In the event the ENHANZE® License Agreement is terminated for any reason, the license granted to us would terminate but Halozyme would grant our sublicensees a direct license following such termination. In the event the ENHANZE® License Agreement is terminated other than for our breach, we would retain the right to sell licensed products then on hand for a certain period of time post-termination. 1.4.3 Our Exclusive License with the University of Texas for NHANCE™ and ABDEG™ In February 2012, we entered into an exclusive in-license with the Board of Regents of the University of Texas System (UT BoR) for the use of certain patent rights relating to the NHANCE™ platform for any use worldwide (the UT Agreement). The UT Agreement was amended on December 23, 2014 to also include certain additional patent rights relating to the ABDEG™ platform. Upon commercialization of any of our products that use the in-licensed patent rights, we will be obligated to pay UT BoR a percentage of net sales as a royalty until the expiration of any patents covering the product. This royalty varies with net sales volume and is subject to an adjustment for royalties we receive from a sublicensee of our rights under the UT Agreement, but in any event does not exceed 1%. In addition, we must make annual license maintenance payments to UT BoR until termination of the UT Agreement and we have assumed certain development and commercial milestone payment and reimbursement obligations. We also have diligence requirements with respect to development and commercialization of products which use the in-licensed patent rights. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Collaborations and licenses 35

Pursuant to the UT Agreement, we may grant sublicenses to third parties. If we receive any non-royalty income in connection with such sublicenses, we must pay UT BoR a percentage of such income varying from low-middle single digits to middle-upper single digits depending on the nature of the sublicense. Such fees are waived if a sublicensee agrees to pay the milestone payments as set forth in the UT Agreement. We may unilaterally terminate the UT Agreement for convenience upon prior written notice. Absent early termination, the UT Agreement will automatically expire upon the expiration of all issued patents and filed patent applications within the patent rights covered by the UT Agreement. Our royalty payment obligations expire, on a product-by-product and country-by-country basis, at such time as there are no valid claims covering such product. 1.4.4 OncoVerity for cusatuzumab In 2022, we, the University of Colorado Anschutz Medical Campus and the University of Colorado Health (UCHealth) created an asset-centric spin-off, OncoVerity, Inc (OncoVerity), focused on optimizing and advancing the development of cusatuzumab, a novel anti-CD70 antibody, in acute myeloid leukemia (AML). OncoVerity is an entity of co-creation, combining the extensive translational biology insights from Dr. Clayton Smith, M.D. from the University of Colorado with our experience on the CD70/CD27 pathway. In 2023, we granted an exclusive license for cusatuzumab to OncoVerity and provided, together with a joint venture of University of Colorado Health and University License Equity Holdings, Inc. on the University of Colorado Anschutz Medical Campus, and funding for ongoing clinical development of cusatuzumab. In 2024 and 2025, we participated in a further funding round to support the continued, ongoing, clinical development of cusatuzumab by OncoVerity. 1.4.5 Our Strategic Partnership with AbbVie for ARGX-115 (ABBV-151) In 2016, we entered into a collaboration agreement with AbbVie for ARGX-115 (ABBV-151), targeting GARP in oncology (the AbbVie Collaboration Agreement). After completing IND-enabling work, AbbVie exercised its option and assumed full responsibility for global development and commercialization. We are eligible for up to $625 million in potential development, regulatory and commercial milestones, plus tiered royalties from the mid-single digits to lower teens. We also retain co-promotion rights in the EEA and Switzerland. The agreement continues on a product-by-product basis until AbbVie’s payment obligations expire, and AbbVie may terminate the AbbVie Collaboration Agreement with prior notice. 1.5 Manufacturing and Supply At argenx, our manufacturing and supply strategy is built for scale, resilience, and speed, reflecting our commitment to move science forward and deliver for patients worldwide. We utilize third-party contract manufacturers who act in accordance with the FDA’s current good manufacturing practices (cGMPs) for the manufacture of drug substances and drug products. We partner with a global network of contract manufacturers who share our standards for quality and innovation, with the goal that every step, from cell bank development to large-scale drug substance production, meets the highest industry benchmarks. Our global supply chain and distribution strategy is to serve patients in region for that region. We work with Lonza teams based in Slough, UK, Portsmouth, U.S., Singapore and Visp, Switzerland for activities relating to the development of cell banks, development of our manufacturing processes and the manufacturing of drug substance, thereby using validated and scalable systems broadly accepted in our industry. In 2022, we started our collaboration with FUJIFILM Diosynth Biotechnologies Denmark ApS (Fujifilm) based in Hillerød, Denmark, for activities relating to the large-scale manufacturing of efgartigimod drug substance. In 2025, we expanded our partnership with Fujifilm to include new manufacturing site in North Carolina, U.S., strengthening our global supply chain and supporting anticipated growth in efgartigimod and pipeline assets. We use additional contract manufacturers to fill, label, package, store and distribute (investigational) drug products. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Collaborations and licenses 36

1.6 Intellectual Property 1.6.1 Introduction We strive to protect and maintain exclusivity for the proprietary technologies that we believe are important to our patients, business, and shareholders. We continue to pursue and maintain patent protection intended to cover core platform technologies incorporated into, or used to produce, our product candidates and commercial products. We will seek protection for our patient innovations in key global jurisdictions. We continue to focus our exclusivity strategies on all aspects of our assets, including our compositions of matter, methods of use for our approved products, and other inventions that are important to our business (e.g., the patient innovations described in our product labels/product inserts and our core manufacturing technologies). Our intellectual property portfolio continues to grow and keep pace with the innovations arising from our discovery, development, and commercial efforts. We expect the total volume of patent positions under our management to increase each year as our pipeline evolves. We currently oversee more than 500 pending applications and granted patents. More importantly, as we continue to innovate for patients, we will work to protect our patient innovations with new intellectual property filings to enable future reinvestment for patients. In addition to patent protection, we rely on trademarks and trade secrets to protect aspects of our business that are not amenable to, or that we do not consider appropriate for, patent protection, including certain aspects of our llama immunization and antibody affinity maturation approaches. Our commercial success depends in part upon our ability to obtain and maintain exclusivity, including regulatory exclusivities, patent, trade secret, and other proprietary protection for commercially important technologies, inventions and know-how related to our business. We will defend and enforce our intellectual property rights, particularly our patent rights, and preserve the confidentiality of our trade secrets while operating without infringing valid and enforceable intellectual property rights of others. Specifically, we are materially dependent on elements of our regulatory, patent and other proprietary protection, including certain of those related to our core platform technologies, described in Section 1.6.2 “Platform Technologies” below and our product candidates, as described in Section 1.6.3 “Our Internal Programs” below and Section 1.6.4 “Our Partnered Programs” below. The patent positions for biotechnology companies like us are generally uncertain and can involve complex legal, scientific, and factual issues. In addition, the coverage recited in the claims in a patent application can be significantly reduced before a patent is issued, and claim scope can be reinterpreted and even challenged after issuance. As a result, we cannot guarantee that any of our platform technologies and product candidates, or products will be protectable or remain protected by valid and enforceable patents. We cannot accurately predict whether pending patent applications will issue as patents in any particular jurisdiction or whether the claims of any issued patents will provide sufficient protection from infringing competitors. Any patents we hold may be challenged, circumvented, limited or invalidated by third parties. The term of individual patents depends on the patent laws in the countries in which they are obtained. In most countries, the patent term is 20 years from the earliest date of filing a non-provisional patent application. In the U.S., the term of a patent covering an FDA-approved drug may be eligible for a limited patent term extension under the Drug Price Competition and Patent Term Restoration Act of 1984 (Hatch-Waxman Act) as compensation for the loss of patent term during the FDA regulatory review process as described in Section 1.7.1 “Licensure and Regulation of Biologics in the U.S” below. Similar provisions are available in the EU and in other jurisdictions to extend the term of a patent that covers an approved drug and/or its use. It is possible that issued U.S. patents covering each of our products/product candidates may be entitled to patent term extensions. If our product candidates receive FDA approval, we intend to apply for patent term extensions, if available, to extend the term of patents that cover the approved product candidates and/or their uses. We also intend to seek patent term extensions in any jurisdictions where available. There is no guarantee that the applicable authorities, including the FDA, will agree with our assessment of whether such extensions should be granted, and if granted, the length of such extensions. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information

1.6.2 Platform Technologies With regard to our platform technologies, we own or control intellectual property rights directed to our SIMPLE ANTIBODY™ discovery platform, the ABDEG™ and NHANCE™ technologies. With regard to our SIMPLE ANTIBODY™ discovery platform, we have a broad patent portfolio providing exclusivity on the SIMPLE ANTIBODY™ platform. We expect to enjoy exclusivity under this patent portfolio until between 2029 and 2033. With regard to the ABDEG™ platform, we co-own the technology with the University of Texas Southwestern Medical Center and enjoy certain exclusive license rights. We have a broad patent portfolio covering the composition of matter and uses of certain FcRn antagonists to achieve certain biological effects. A composition of matter patent expires in 2036 in the U.S., whereas in many other countries the base expiry date is 2034. With regard to the NHANCE™ platform, we exclusively licensed two U.S. patents from the University of Texas Southwestern Medical Center with composition of matter claims directed to an IgG molecule comprising a variant human Fc domain, and method of use claims directed to a method of blocking FcRn function in a subject by providing to the subject such an IgG molecule. The U.S. patents are expected to expire between 2027 to 2028. The patent family also includes a granted European patent. 1.6.3 Our Internal Programs efgartigimod efgartigimod incorporates the ABDEG™ platform technology, for which we co-own the technology with the University of Texas Southwestern Medical Center and enjoy certain exclusive license rights. We have a broad patent portfolio with multiple patent families covering the composition of matter and uses of certain FcRn antagonists to achieve disease-modifying effects. A composition of matter and other relevant patents arising from the same patent family in the U.S. expire in 2036 and 2037 in Europe whereas in many other countries the base expiry date is 2034. We anticipate several more patient innovations to evolve during development and commercialization for which we will seek additional patent protection with later expiration dates. Our ARGX-109 Product Candidate With regard to our wholly-owned ARGX-109 product candidate, we have one patent family with composition of matter claims directed to ARGX-109. The patent family has a base expiry date in 2033. We anticipate several more patient innovations to evolve during development for which we will seek additional patent protection. Furthermore, ARGX-109 incorporates or employs the SIMPLE ANTIBODY™ platform technology and the NHANCE™ platform technology. empasiprubart Product Candidate With regard to the empasiprubart product candidate, we own or have rights to multiple patent families (with several granted patents and pending patent applications in multiple jurisdictions in North America, South America, the EU and Asia, directed to composition of matter claims and method of treatment claims. The patent families have base expiry dates in 2034, 2039 and 2040. We anticipate several more patient innovations to evolve during development for which we will seek additional patent protection. empasiprubart product candidate incorporates or employs the NHANCE™ platform technology. adimanebart Product Candidate With regard to the adimanebart product candidate, we in-licensed patent families from/with New York University Langone Health, a U.S. medical center based in New York, and additional patent families from/ with the Leiden University Medical Centre, with a U.S. granted patent and several pending applications in multiple jurisdictions . We anticipate several more patient innovations to evolve during development for which we will seek additional patent protection. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Intellectual Property 38

Our ARGX-118 Product Candidate With regard to the ARGX-118 product candidate, we co-own a patent portfolio with VIB, an inflammation research center in Ghent, Brussels, and Ghent University, with one U.S. granted patent and pending patent applications in multiple jurisdictions in North America, South America, the EU and Asia. The patent family has a base expiry date in 2039. 1.6.4 Our Partnered Programs Our cusatuzumab (ARGX-110) Product Candidate With regard to the cusatuzumab product candidate, we have a broad patent portfolio that include claims to the composition of matter, uses of the molecule, and other important inventions. The issued U.S. patents expire in 2032 and 2033, without taking a potential patent term extension into account. cusatuzumab incorporates or employs the SIMPLE ANTIBODY™ and POTELLIGENT® platform technologies. Our ARGX-115 (ABBV-151) Product Candidate With regard to the ARGX-115 (ABBV-151) product candidate that we co-own with, and exclusively license from, the Ludwig Institute for Cancer Research and UCL, we have a patent portfolio that includes a U.S. patent with a base expiry date in 2034, without taking a potential patent term extension into account. There is a second family with meaningful patent coverage to the composition of matter and epitope claims that are expected to expire in 2036 and 2038. Furthermore, ARGX-115 (ABBV-151) incorporates or employs the SIMPLE ANTIBODY™ platform technology. Our ARGX-112 (LP-0145) Product Candidate With regard to the ARGX-112 (LP-0145) product candidate, we have one patent family with composition of matter claims directed to an antibody that binds human IL-22R. The patent family has a base expiry date in 2037. Furthermore, ARGX-112 (LP-0145) incorporates the SIMPLE ANTIBODY™ platform technology. 1.6.5 Trade Secret Protection In addition to patent protection, we rely on trade secret protection to ensure exclusivity for our proprietary information that is not amenable to, or that we do not consider appropriate for, patent protection, including, for example, certain aspects of our llama immunization and antibody affinity maturation approaches. However, trade secrets can be difficult to protect. Although we take steps to protect our proprietary information, including restricting access to our premises and our confidential information, as well as entering into agreements with our employees, consultants, advisors and potential collaborators, third parties may independently develop the same or similar proprietary information or may otherwise gain access to our proprietary information. As a result, we may be unable to meaningfully protect our trade secrets and proprietary information. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Intellectual Property 39

1.7 Regulation Government authorities in the U.S., at the federal, state and local level, and in the EU and its Member States and other countries and jurisdictions, extensively regulate, among other things, the research, development, testing, manufacture, quality control, approval, packaging, storage, recordkeeping, labeling, advertising, promotion, distribution, marketing, post-approval monitoring and reporting, and import and export of pharmaceutical products, including biological products. In addition, many countries and jurisdictions regulate the pricing of pharmaceutical products. The processes for obtaining marketing approvals in the U.S. and in other countries and jurisdictions, along with subsequent compliance with applicable statutes and regulations and other regulatory authorities, require the expenditure of substantial personnel and financial resources, and breach of which can result in enforcement activity under civil, administrative and/ or criminal law. 1.7.1 Licensure and Regulation of Biologics in the U.S. In the U.S., biological products used for the prevention, treatment, or cure of a disease or condition in a human being are subject to regulation under the U.S. Federal Food, Drug, and Cosmetic Act (FDCA) and its implementing regulations. Biologics are approved for marketing under provisions of the Public Health Service Act (PHSA) via biologics license applications (BLAs). An applicant seeking approval to market and distribute a new biologic in the U.S. generally must satisfactorily complete each of the following steps: • preclinical laboratory tests, animal studies and formulation studies all performed in accordance with applicable requirements, including the GLPs; • submission to the FDA of an IND application for human clinical testing, which contains results of the preclinical tests, together with manufacturing information and analytical data and must become effective before human clinical trials may begin; • approval by an institutional review board (IRB) representing each clinical site before each clinical trial may be initiated; • performance of adequate and well-controlled human clinical trials to establish the safety, potency and purity of the product candidate for each proposed indication, in accordance with good clinical practices (GCPs); • preparation and submission to the FDA of a BLA for a biological product requesting marketing for one or more proposed indications, including submission of detailed information on the manufacture and composition of the product in clinical development and proposed labeling; • one or more FDA inspections of the manufacturing facility or facilities, including those of third parties, at which the product, or components thereof, are produced to assess compliance with cGMP requirements and to assure that the facilities, methods and controls are adequate to preserve the product’s identity, potency, quality and purity; • FDA inspections of the clinical trial sites and/or sponsor to assure compliance with GCPs, and the integrity of clinical data in support of the BLA; • payment of user fees and securing FDA approval of the BLA and licensure of the new biological product; and • compliance with any post-approval requirements, including the potential requirement to implement a risk evaluation and mitigation strategy (REMS) and any post-approval studies required by the FDA. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Regulation 40

Human Clinical Trials in Support of a BLA Clinical trials typically are conducted in three sequential phases, but the phases may overlap or be combined. Additional clinical trials may be required after approval. • Phase 1 clinical trials are initially conducted in a limited population to test the product candidate for safety, including adverse effects, dose tolerance, absorption, metabolism, distribution, excretion and PD in healthy humans or, in patients. • Phase 2 clinical trials are generally conducted in a limited patient population to identify possible adverse effects and safety risks, evaluate the efficacy of the product candidate for specific targeted indications and determine dose tolerance and optimal dosage. Multiple Phase 2 clinical trials may be conducted by the sponsor to obtain information prior to beginning larger Phase 3 clinical trials. • Phase 3 clinical trials are undertaken within an expanded patient population to gather additional information about safety and effectiveness necessary to evaluate the overall benefit-risk relationship of the drug and to provide an adequate basis for physician labeling. A sponsor who wishes to conduct a clinical trial outside the U.S. may, but is not required to, obtain FDA clearance to conduct the clinical trial under an effective IND. If a foreign clinical trial is not conducted under an IND, the sponsor may submit data from the clinical trial to the FDA in support of the BLA so long as the clinical trial is well-designed and well-conducted in accordance with GCPs, including review and approval by an independent ethics committee, and the FDA is able to validate the clinical trial data through an onsite inspection, if necessary. In some cases, the FDA may approve a BLA for a product candidate but require the sponsor, or the sponsor may otherwise choose, to conduct additional clinical trials to further assess, amongst other things, the product candidate’s safety and effectiveness after approval. Such post-approval clinical trials are typically referred to as Phase 4 clinical trials. Failure to exhibit due diligence with regard to conducting required Phase 4 clinical trials could result in FDA enforcement, including withdrawal of approval for products. Review and Approval of a BLA The results of product candidate development, preclinical testing and clinical trials, including negative or ambiguous results as well as positive findings, are submitted to the FDA as part of a BLA requesting a license to market the product. The BLA also must contain extensive manufacturing information and detailed information on the composition of the product and proposed labeling as well as payment of a user fee, unless exempt. The FDA has 60 days after submission of the application to conduct an initial review to determine whether the BLA is sufficient to file based on the agency’s threshold determination that it is sufficiently complete to permit substantive review. If the FDA determines the BLA is not sufficiently complete, it will refuse to file the BLA. Once the submission has been filed, the FDA begins an in-depth review of the application. Under the goals agreed to by the FDA under the PDUFA, the FDA has 10 months from the filing date in which to complete its initial review of a standard application and respond to the applicant, and six months from the filing date for an application granted priority review. The FDA does not always meet its PDUFA goal dates and they may be extended in certain circumstances. After the FDA’s evaluation of the application and accompanying information, including the results of any necessary inspections, the FDA will issue an approval letter, or a complete response letter. An approval letter authorizes commercial marketing of the product with specific prescribing information for specific indications. Under the PHSA, the FDA may approve a BLA if it determines that the product is safe, pure and potent and the facility where the product will be manufactured meets standards designed to ensure that it continues to be safe, pure and potent. If the application is not approved, the FDA will issue a complete response letter, which will identify the deficiencies in the application. Sponsors that receive a complete response letter may resubmit to the FDA information addressing the issues identified by the FDA, withdraw the application, or request a hearing. Even if a BLA is resubmitted with data and information addressing the deficiencies, the FDA may decide that the BLA does not satisfy the criteria for approval. The FDA may also refer the application to an advisory committee, consisting of independent experts, for review, evaluation and recommendation as to whether the application should be approved, particularly when applications present difficult or novel questions of safety or efficacy. The FDA is not bound by the argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Regulation 41

recommendations of an advisory committee, but it considers such recommendations carefully when making decisions. If the FDA approves a new product, it may require testing and surveillance programs to monitor the product after commercialization, or impose other conditions, including distribution restrictions or other risk management mechanisms, including REMS, to help ensure that the benefits of the product outweigh the potential risks. REMS can include medication guides, communication plans for healthcare professionals, and/or elements to assure safe use. This can include, but are not limited to, special training or certification for prescribing or dispensing, dispensing only under certain circumstances, special monitoring, and the use of patent registries. The FDA may prevent or limit further marketing of a product based on the results of post-market studies or surveillance programs. After approval, many types of changes to the approved product, such as adding new indications, certain manufacturing changes, and additional labeling claims, are subject to further testing requirements and FDA review and approval. Expedited Development and Review Programs The FDA is authorized to designate products meeting certain criteria for expedited development and review programs. Even if a product qualifies for one or more of these programs, the FDA may later decide that the product no longer meets the conditions for qualification, or the time period for FDA review or approval may not be shortened. The FDA may designate a product for fast track review if it is intended, whether alone or in combination with one or more other products, for the treatment of a serious or life-threatening disease or condition, and demonstrates the potential to address unmet medical needs for such a disease or condition. For fast track products, sponsors may have more frequent interactions with the FDA and the FDA may initiate review of sections of a fast track product’s application before the application is complete (rolling review). The sponsor must also provide, and the FDA must approve, a schedule for the submission of the remaining information and the sponsor must pay applicable user fees. However, the FDA’s PDUFA clock for a rolling review application does not begin until the last section of the application is submitted. A product may be designated as a breakthrough therapy if it is intended, either alone or in combination with one or more other products, to treat a serious or life-threatening disease or condition and preliminary clinical evidence indicates that the product may demonstrate substantial improvement over existing therapies on one or more clinically significant endpoints, such as substantial treatment effects observed early in clinical development. The FDA may take certain actions with respect to breakthrough therapies, including holding meetings with the sponsor throughout the development process; providing timely advice to the product sponsor regarding development and approval; involving more senior staff in the review process; assigning a cross-disciplinary project lead for the review team; and taking other steps to design the clinical trials in an efficient manner. Breakthrough therapy designation also comes with all of the benefits of fast-track designation. The FDA may designate a product for priority review if it is a product that treats a serious condition and, if approved, would provide a significant improvement in safety or effectiveness. The FDA determines, on a case-by-case basis, whether the proposed product represents a significant improvement when compared with other available therapies. Significant improvement may be illustrated by evidence of increased effectiveness in the treatment of a condition, elimination or substantial reduction of a treatment-limiting product reaction, documented enhancement of patient compliance that may lead to improvement in serious outcomes, or evidence of safety and effectiveness in a new subpopulation. A priority designation is intended to direct overall attention and resources to the evaluation of such applications, and to shorten the FDA’s goal for taking action on a marketing application from 10 months to six months after accepting the application for filing. The FDA may grant accelerated approval to a product for a serious or life-threatening condition that provides meaningful therapeutic advantage to patients over existing treatments based upon a determination that the product has an effect on a surrogate endpoint that is reasonably likely to predict clinical benefit or on a clinical endpoint that can be measured earlier than an effect on irreversible morbidity or mortality (IMM) and that is reasonably likely to predict an effect on IMM or other clinical benefit (intermediate clinical endpoint), taking into account the severity, rarity, or prevalence of the argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Regulation 42

condition and the availability or lack of alternative treatments. Products granted accelerated approval must meet the same statutory standards for safety and effectiveness as those granted traditional approval. For the purposes of accelerated approval, a surrogate endpoint is a marker, such as a laboratory measurement, radio-graphic image, physical sign, or other measure that is thought to predict clinical benefit but is not itself a measure of clinical benefit. Surrogate endpoints can often be measured more easily or more rapidly than clinical endpoints. An intermediate clinical endpoint is a measurement of a therapeutic effect that is considered reasonably likely to predict the clinical benefit of a product, such as an effect on IMM. The accelerated approval pathway is most often used in settings in which the course of a disease is long and an extended period of time is required to measure the intended clinical benefit of a product, even if the effect on the surrogate or intermediate clinical endpoint occurs rapidly. Thus, accelerated approval has been used extensively in the development and approval of products for treatment of a variety of cancers in which the goal of therapy is generally to improve survival or decrease morbidity and the duration of the typical disease course requires lengthy and sometimes large clinical trials to demonstrate a clinical or survival benefit. The accelerated approval pathway is usually contingent on a sponsor’s agreement to conduct, in a diligent manner, a post-approval confirmatory clinical trial or studies to verify and describe the product’s clinical benefit. These confirmatory clinical trials must be completed with due diligence, and the FDA may require that the confirmatory clinical trial be designed, initiated, and/or fully enrolled prior to, or within a certain period following, approval. The FDA must also specify the conditions of any required post-approval clinical trial. Sponsors are required to submit progress reports for required post-approval studies, and the failure to conduct with due diligence a required post-approval clinical trial, including a failure to meet any required conditions specified by the FDA, or to submit timely reports, are prohibited acts under the FDCA. Failure to conduct required post-approval studies, or confirm a clinical benefit during post-marketing studies, would allow the FDA to withdraw the product from the market on an expedited basis. Unless otherwise informed by the FDA, all promotional materials for product candidates approved under accelerated approval are subject to prior review by the agency. Orphan Drug Designation and Exclusivity Orphan drug designation in the U.S. is designed to encourage sponsors to develop products intended for rare diseases or conditions. In the U.S., a rare disease or condition is statutorily defined as a condition that affects fewer than 200,000 individuals in the U.S. or that affects more than 200,000 individuals in the U.S. and for which there is no reasonable expectation that the cost of developing and making available the product for the disease or condition will be recovered from sales of the product in the U.S. An application for designation as an orphan product can be made any time prior to the filing of an application for approval to market the product. If the FDA grants orphan drug designation, the generic identity of the product and its potential orphan use are disclosed publicly by the FDA. Orphan drug designation qualifies a company for tax credits. Orphan drug designation does not convey any advantage in or shorten the duration of the regulatory review and approval process. If a product that has orphan drug designation subsequently receives the first FDA approval for the disease or condition for which it has such designation, the product is entitled to orphan drug exclusivity, which means that the FDA may not approve any other application to market the same drug for the same indication for seven years from the date of such approval, except in limited circumstances, such as a showing of clinical superiority to the product with orphan exclusivity by means of greater effectiveness, greater safety or providing a major contribution to patient care, or if the holder of the orphan exclusivity is unable to supply the market. Competitors, however, may receive approval of either a different product for the same indication or the same product for a different indication, which could be used off-label in the orphan indication. Orphan drug exclusivity also could block the approval of one of our products for seven years if a competitor obtains approval before we do for the same product, as defined by the FDA, for the same indication we are seeking approval, or if our product is determined to be contained within the scope of the approval of the competitor’s product for the same indication or disease. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Regulation 43

Post-Approval Regulation If regulatory approval for marketing of a product or new indication for an existing product is obtained, the sponsor will be required to comply with all post-approval regulatory requirements, including those that the FDA has imposed as part of the approval process. The sponsor will be required to report certain adverse reactions and production problems to the FDA, provide updated safety and efficacy information and comply with requirements concerning advertising and promotional labeling. Manufacturers and other parties involved in the drug supply chain for prescription drug and biological products must also comply with product tracking and tracing requirements and must notify the FDA of counterfeit, diverted, stolen and intentionally adulterated products or products that are otherwise unfit for distribution in the U.S. Manufacturers and certain of their subcontractors are required to register their establishments with the FDA and certain state agencies, and are subject to periodic unannounced inspections by the FDA and certain state agencies for compliance with ongoing regulatory requirements, including cGMPs. Accordingly, the sponsor and its third-party manufacturers must continue to expend time, money and effort in the areas of production and quality control to maintain compliance with cGMPs and other regulatory requirements. A biological product may also be subject to official lot release, meaning that the manufacturer is required to perform certain tests on each lot of the product before it is released for distribution. If the product is subject to official lot release, the manufacturer must submit samples of each lot, together with a release protocol showing a summary of the history of manufacture of the lot and the results of all of the manufacturer’s tests performed on the lot, to the FDA. The FDA may in addition perform certain confirmatory tests on lots of some products before releasing the lots for distribution. Finally, the FDA will conduct laboratory research related to the safety, purity, potency and effectiveness of biological products. Any distribution of biological products and samples must comply with the U.S. Prescription Drug Marketing Act and the PHSA. Once approval of a BLA is granted, the FDA may revoke or suspend the approval if compliance with regulatory requirements and standards is not maintained or if problems occur after the product reaches the market. Later discovery of previously unknown problems with a product, including adverse events of unanticipated severity or frequency, or with manufacturing processes, or failure to comply with regulatory requirements, may result in revisions to the approved labeling to add new safety information; imposition of post-market studies or clinical trials to assess new safety risks; or imposition of distribution or other restrictions under a REMS program. FDA also has authority to require post-market studies, in certain circumstances, on reduced effectiveness of a product and may require labeling changes related to new reduced effectiveness information. Other potential consequences for a failure to maintain regulatory compliance include, among other things: • restrictions on the marketing or manufacturing of the product, complete withdrawal of the product from the market, or product recalls; • fines, untitled letters, or warning letters; • refusal of the FDA to approve pending applications or supplements to approved applications, or suspension or revocation of product license approvals; • product seizure or detention, or refusal to permit the import or export of products; or • injunctions or the imposition of civil or criminal penalties. Pediatric Studies and Exclusivity Under the Pediatric Research Equity Act of 2003, as amended (PREA), certain BLAs or supplements thereto must contain data that are adequate to assess the safety and effectiveness of the product for the claimed indications in all relevant pediatric sub-populations, and to support dosing and administration for each pediatric subpopulation for which the product is safe and effective. Sponsors must also submit an initial Pediatric Study Plan (PSP), within 60 days of an end-of-Phase 2 meeting or, if there is no such meeting, as early as practicable before the initiation of the Phase 3 or Phase 2/3 clinical trial. The initial PSPs must contain an outline of the proposed pediatric clinical trial or studies the applicant plans to conduct, including clinical trial objectives and design, any deferral or waiver requests and other information required by regulation. The applicant and the FDA must agree upon a final plan. The FDA or the applicant may request an amendment to the plan at any time. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Regulation 44

The FDA may, on its own initiative or at the request of the applicant, grant deferrals for submission of some or all pediatric data until after approval of the product for use in adults, or full or partial waivers from the pediatric data requirements. Unless otherwise required by regulation, PREA does not apply to a biologic for an indication for which orphan designation has been granted, except that PREA will apply to an original BLA for a new active ingredient that is orphan-designated if the biologic is a molecularly targeted cancer product intended for the treatment of an adult cancer and is directed at a molecular target that FDA determines to be substantially relevant to the growth or progression of a pediatric cancer. Pediatric exclusivity is another type of non-patent regulatory exclusivity in the U.S. and, if granted for a biologic, provides for the attachment of an additional six months of protection to the term of any existing regulatory exclusivity (i.e., reference product exclusivity and orphan drug exclusivity) that has at least 9 months left to expiration. This six-month exclusivity may be granted if a BLA sponsor submits reports of pediatric studies that fairly respond to a written request from the FDA for such studies, were conducted in accordance with commonly accepted scientific principles and protocols, and have been reported in accordance with filing requirements. Biosimilars and Exclusivity The Biologics Price Competition and Innovation Act (BPCIA) established a regulatory scheme authorizing the FDA to approve biosimilars and interchangeable biosimilars. Under the BPCIA, an applicant may submit an application for licensure of a biologic product that is “biosimilar to” or “interchangeable with” a previously approved biological product or “reference product.” For the FDA to approve a biosimilar product, it must find that the proposed biosimilar is highly similar to the reference product notwithstanding minor differences in clinically inactive components and that there are no clinically meaningful differences between the product and the reference product in terms of safety, purity, or potency. For the FDA to approve a biosimilar product as interchangeable with a reference product, the agency must find that the biosimilar product is biosimilar to the reference product and that it can be expected to produce the same clinical results as the reference product in any given patient, and (for products administered multiple times) that the biologic and the reference biologic may be alternated or switched after one has been previously administered without increasing safety risks or risks of diminished efficacy relative to exclusive use of the reference biologic without such alternation or switch. Under the BPCIA, an application for a biosimilar product may not be submitted to the FDA until four years following the date of approval of the reference product. The FDA may not approve a biosimilar product until 12 years from the date on which the reference product was approved. Even if a product is considered to be a reference product eligible for exclusivity, another company could market a competing version of that product if the FDA approves a full BLA for such product containing the sponsor’s own preclinical data and data from adequate and well-controlled clinical trials to demonstrate the safety, purity and potency of their product. We note that patent positions may be available to preclude the introduction into commerce of such competing product independent of any FDA exclusivities. The BPCIA also created certain exclusivity periods for biosimilars approved as interchangeable products. Products deemed interchangeable by the FDA may be substituted by pharmacies as dictated by individual state law. U.S. Patent Term Restoration Depending upon the timing, duration, and specifics of FDA review and approval of our product candidates, some of our U.S. patents may be eligible for limited patent term extension under the Hatch-Waxman Act that permits restoration of the patent term of up to five years as compensation for patent term lost during the FDA regulatory review process. Patent-term restoration, however, cannot extend the remaining term of a patent beyond a total of 14 years from the product’s approval date, and only those claims covering such approved product, a method for using it or a method for manufacturing it may be extended. The patent- term restoration period is generally one-half the time between the effective date of an IND and the submission date of a BLA plus the time between the submission date of a BLA and the approval of that application, except that the review period is reduced by any time during which the applicant failed to exercise due diligence. Only one patent applicable to an approved biologic is eligible for the extension and the application for the extension must be submitted within 60 days of approval from FDA and prior to the expiration of the patent. The U.S. Patent and Trademark Office (USPTO), in consultation with the FDA, reviews and approves the application for any patent term extension or restoration. In the future, we may apply for restoration of patent term for our currently owned or licensed patents to add patent life beyond argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Regulation 45

the current expiration date, depending on the expected length of the clinical trials and other factors involved in the filing of the relevant BLA. 1.7.2 Regulation and Procedures Governing Approval of Medicinal Products in the European Union Similar to the U.S., the EU comprehensively regulates, among other things, the development, manufacturing, placing on the market, advertising, distribution, import and export of medicinal products. Particularly, the placing on the market of a medicinal product for human use in the EU requires a marketing authorization (MA). Main provisions governing medicinal products in the EU are Directive 2001/83/EC and Regulation (EC) No 726/2004 (each as amended). Regulation (EC) No 141/2000 and Regulation (EC) No. 847/2000 (each as amended) are also of particular relevance for orphan medicinal products. While directives need to be transposed into national law by member states of the EU (EU Member States) before they are applicable, regulations directly apply in the EU Member States once these have been enacted. The process governing approval of MA applications (MAA) for the placing on the market of medicinal products in the EU generally follows the same lines as in the U.S. It entails satisfactory completion of pharmaceutical development, pre-clinical trials and adequate and well-controlled clinical trials to establish the safety and efficacy of the medicinal product for each proposed indication. The EU also requires an application for authorization of clinical trials to relevant competent authorities and the submission of an MAA to the European Medicines Agency (EMA) or to competent authorities in EU member states and granting of such MA by the European Commission or relevant national authorities before the medicinal product can be marketed and sold in the EU or the relevant EU Member States. The below mentioned principles and rules generally apply within the EEA, i.e., the EU including Iceland, Liechtenstein and Norway. Clinical Trial Approval Both non-clinical and clinical data are generally required to support an MAA for a medicinal product in the EU. Non-clinical investigations are performed to demonstrate the health or environmental safety of new biological substances. Non-clinical (pharmaco-toxicological) investigations must generally be conducted in compliance with the principles of good laboratory practice (GLP) as set forth in EU Directive 2004/10/EC (as amended). Clinical trials are comprehensively regulated in the EU under the Clinical Trials Regulation (EU) No 536/2014 (CTR), which entered into application on January 31, 2022, and gradually replaced the Clinical Trials Directive 2001/20/EC (CTD). As before, many of the CTR’s legal obligations are on the so-called sponsor, which is defined as the individual, company, institution, or organization that takes responsibility for the initiation, for the management and for setting up the financing of a clinical trial. The sponsor must obtain an authorization from the competent authority in the EU Member State(s) in which the clinical trial will be conducted as well as an approval from the competent national ethics committee in accordance with relevant national legislation in each of the relevant member states, before the commencement of such clinical trial. The CTR also imposes requirements, among others, regarding the conduct of a clinical trial (which must be conducted in accordance with the protocol and good clinical practice to generate acceptable data for MA submission), safety reporting of adverse events and reactions, changes to clinical trials, protection and informed consent of clinical trial subjects. Clinical trials conducted outside the EEA must follow the principles set forth in EU legislation if their results are to be submitted in an MAA in the EU. Orphan Designation and Exclusivity Regulations (EC) No. 141/2000 and No. 847/2000 (each as amended) provide that a product can be designated as an orphan medicinal product by the European Commission if its sponsor can establish: (i) that the product is intended for the diagnosis, prevention or treatment of a life-threatening or chronically debilitating condition, (ii) either (a) the prevalence of the condition is not more than five in ten thousand persons in the EU when the application is made, or (b) without incentives it is unlikely that the marketing of the product in the EU would generate sufficient return to justify the necessary investment in its argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Regulation 46

development and (iii) there exists no satisfactory method of diagnosis, prevention, or treatment of the condition in question that has been authorized in the EU or, if such method exists, the product has to be of a significant benefit compared to products available for the condition. An orphan designation provides a number of benefits, including fee reductions and, regulatory assistance. If an MA is granted for an orphan medicinal product, this generally results in a ten-year period of market exclusivity for the approved orphan indication. It is, however, not possible to combine non-orphan and orphan indications within the same MA. Thus, for non-orphan indications treated with the same active pharmaceutical ingredient, a separate MA has to be sought. Alternatively, the orphan designation may be waived to allow for the addition of non-orphan indications to an existing MA. As a result, the approved medicinal product would no longer profit from the orphan designation’s benefits. During an orphan medicinal product’s market exclusivity period, neither the EMA, the European Commission nor the EU Member States can accept an application or grant an MA for a “similar medicinal product.” A “similar medicinal product”, i.e., a medicinal product containing a similar active substance or substances as contained in an authorized orphan medicinal product, and which is intended for the same therapeutic indication. The market exclusivity period for the authorized therapeutic indication may, however, be reduced to six years if, at the end of the fifth year, it is established that the product no longer meets the criteria for orphan designation. For orphan medicinal products intended for pediatric use, the market exclusivity period may be prolonged by additional two years if they are authorized with a pediatric indication based on the results from studies conducted under an EMA-approved pediatric investigation plan or if they are authorized without a pediatric indication but the results of the studies conducted under the EMA-approved pediatric investigation plan are reflected in the summary of product characteristic and, if appropriate, in the package leaflet. Market exclusivity may also be revoked in very select cases, such as if (i) it is established that a similar medicinal product is safer, more effective or otherwise clinically superior; (ii) the MA holder (MAH) for the authorized orphan medicinal product consents to the second orphan application; or (iii) the MAH for the authorized orphan medicinal product cannot supply sufficient quantities. Orphan designation must be requested before submitting an MAA and is reconfirmed during the MAA process. Orphan designation does not convey any advantage in, or shorten the duration of, the regulatory review and MA approval process. Marketing Authorization To obtain an MA for a medicinal product under the EU regulatory framework, an applicant must submit an MAA, either to the EMA using the centralized procedure or to competent authorities in the EU Member States using the other procedures (decentralized procedure, national procedure, or mutual recognition procedure). An MA may be granted only to an applicant established in the EU. Regulation (EC) No. 1901/2006 provides that prior to obtaining an MA in the EU, an applicant must demonstrate compliance with all measures included in an EMA-approved pediatric investigation plan, covering all subsets of the pediatric population, unless the EMA has granted a product-specific waiver, class waiver, or a deferral for one or more of the measures included in the pediatric investigation plan. The centralized procedure provides for the grant of a single MA by the European Commission that is valid for all EEA Member States. Pursuant to Regulation (EC) No. 726/2004 (as amended), the centralized procedure is compulsory for specific products, including for medicines produced by certain biotechnological processes, products designated as orphan medicinal products, advanced therapy medicinal products (gene therapy, somatic cell therapy or tissue engineered products) and products with a new active substance indicated for the treatment of certain diseases, including products for the treatment of cancer and auto-immune diseases and other immune dysfunctions and neurodegenerative disorders. The centralized procedure is optional for certain other medicinal products. Under the centralized procedure, the EMA’s Committee for Medicinal Products for Human Use (CHMP) is responsible for conducting the assessment of a product to define its risk/benefit profile. The CHMP recommendation is then sent to the European Commission, which adopts a decision binding in all EEA Member States. Under the centralized procedure, the maximum timeframe for the evaluation of an MAA is 210 days, excluding clock stops when additional information or written or oral explanation is to be provided by the applicant in response to questions asked by the CHMP, which can considerably extend the 210 days. Accelerated evaluation (150 days excluding clock stops) may be granted by the CHMP in exceptional cases, when a medicinal product is of major interest from the point of view of public health and, in particular, from the viewpoint of therapeutic innovation. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Regulation 47

MAs have an initial validity for five years, in principle, and they may be renewed after five years on the basis of a reevaluation of the risk benefit balance by the EMA, or by the competent authority of the EU Member State. Once renewed, the MA is valid for an unlimited period, unless the European Commission or the competent authority decides, on justified grounds relating to pharmacovigilance, to proceed with one additional five-year renewal period. Any MA that is not followed by the placement of the medicinal product on the EU market or on the market of the authorizing EU Member State(s) within three years after authorization, or if the drug is removed from the market for three consecutive years, ceases to be valid. European Data and Market Exclusivity In the EU, innovative medicinal products, approved on the basis of a complete independent data package, qualify for eight years of data exclusivity upon MA and an additional two years of market exclusivity (for the more comprehensive protections applying to orphan medicinal products, please refer to Section 1.7.2 “Orphan Designation and Exclusivity” above). The data exclusivity, if granted, prevents generic or biosimilar applicants from referencing the innovator’s preclinical and clinical trial data contained in the dossier of the reference product when applying for a generic or biosimilar MA in the EU, for a period of eight years from the date on which the reference product was first authorized in the EU. During the additional two-year period of market exclusivity, a generic or biosimilar MAA can be submitted, and the innovator’s data may be referenced, but no generic or biosimilar product can be marketed in the EU until the expiration of the market exclusivity period. The overall ten-year period will be extended to a maximum of 11 years if, during the first eight years of those 10 years, the MAH obtains an MA for one or more new therapeutic indications which, during the scientific evaluation prior to their authorization, are determined to bring a significant clinical benefit in comparison with currently approved therapies. There is no guarantee that a product will be considered by the EMA to be an innovative medicinal product, and products may not qualify for data exclusivity. Even if a product is considered to be an innovative medicinal product so that the innovator gains the prescribed period of data exclusivity, another company nevertheless could also market another version of the product if such company obtained an MA based on an MAA with a complete independent data package of pharmaceutical tests, preclinical tests and clinical trials. Regulatory Requirements after Marketing Authorization Following MA approval, the MAH is required to comply with a range of requirements applicable to the manufacturing, marketing, promotion and sale of the medicinal product. These include compliance with the EU’s stringent pharmacovigilance or safety reporting rules under Directive 2001/83/EC and Regulation (EU) 726/2004 (each as amended) and the associated guideline on good pharmacovigilance practices (as amended), pursuant to which post-authorization studies and additional monitoring obligations can be imposed. In addition, the manufacturing of authorized medicinal products, for which a separate manufacturer’s license is mandatory, must also be conducted in strict compliance with the principles of good manufacturing practice (GMP) set forth in Commission Directive 2017/1572 GMP and comparable requirements of other regulatory bodies in the EU, which mandate the methods, facilities and controls used in manufacturing, processing and packing of products to assure their safety and identity. Further, the wholesale distribution of authorized medicinal products requires a separate distribution license and must be conducted in strict compliance with good distribution practice standards. Finally, the marketing and promotion of authorized medicinal products is strictly regulated under Directive 2001/83/EC, (as amended) and as transposed into national laws. Potential consequences for a failure to maintain regulatory compliance mainly depend on the relevant regulations in the EU Member States, but are, for example, in Germany, similar to those in the U.S. Please refer to Section 1.7.1. “Post-Approval Regulations” above. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Regulation 48

Agreement on new EU Pharmaceutical Legislation On December 11, 2025, the EU legislative bodies agreed to overhaul, modernize, and streamline the existing general pharmaceutical legislation, including e.g., Directive 2001/83/EC, as well as Regulations (EC) No. 726/2004, No. 141/2000, or No. 1901/2006 (EU Pharmaceutical Legislation). This agreement is still subject to formal approval by the European Parliament and the Council of the EU, before being formally adopted. It is expected that the EU Pharmaceutical Legislation will become applicable in 2028. Although the final text has not yet been published, agreed key elements appear to include, among others, certain changes to the baseline marketing exclusivity periods, the streamlining of regulatory procedures as well as a broadening of the so-called “Bolar exemption”, which allows developers to undertake testing and to prepare for regulatory submissions before patent expiry. 1.7.3 Regulation and Procedures Governing Approval of Medicinal Products in Japan In order to market any medical products in Japan, a company must comply with numerous and varying regulatory requirements regarding quality, safety and efficacy in the context, among other things, of clinical trials, marketing approval, commercial sales and distribution of products. A person who manufactures or markets medical products in Japan is subject to the supervision of the Ministry of Health, Labour and Welfare (MHLW), primarily under the Act on Securing Quality, Efficacy and Safety of Pharmaceuticals and Medical Devices (Pharmaceutical and Medical Devices Act). This entails the satisfactory completion of pharmaceutical development, preclinical studies and adequate and well-controlled clinical trials to establish the safety and efficacy of the medical product for each proposed indication. It also requires the filing of a notification of clinical trials with the Pharmaceuticals and Medical Devices Agency (Japan) (PMDA) and the obtaining of marketing approval from the relevant authorities before the product can be marketed and sold in the Japanese market. Business License Under the Pharmaceutical and Medical Devices Act, a company or individual must obtain a Marketing Authorization Holder (MAH) license from the MHLW to engage in the marketing or provision of medical products. This requirement applies to medical products that are either manufactured by the company itself outsourced to a third party for manufacturing or imported. To manufacture medical products for the Japanese market, a company must obtain a manufacturing license from the MHLW for each production facility. This license is separate from the marketing authorization and is required for both domestic and foreign manufacturing sites. Marketing Approval Under the Pharmaceutical and Medical Devices Act, it is generally required to obtain marketing approval from the MHLW for the marketing of each medical product. An application for marketing approval must be made through the PMDA, which implements a marketing approval review. Clinical Trial Under the Pharmaceutical and Medical Devices Act, it is required to file notification of clinical trials with the PMDA. The data of clinical trials and other pertinent data, which must be attached to an application for marketing approval, must be obtained in compliance with the standards established by the MHLW, such as GLPs and GCPs stipulated by the ministerial ordinances of the MHLW. Regulatory Requirements after Marketing Approval A MAH that has obtained marketing approval for a new pharmaceutical is subject to re-examination by the PMDA for a specified period after receiving marketing approval. Such re-examination period for VYVGART is stated to be 10 years after the marketing approval in January 2022. The purpose of this re-examination process is to ensure the safety and efficacy of a newly approved pharmaceutical by imposing on the MAH the obligation to gather clinical data for a certain period after the marketing approval was granted to enable the PMDA to re-examine the product. Results of use and other pertinent data must be attached to argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Regulation 49

an application for a re-examination. An MAH that has obtained a marketing approval is also required to investigate, among other things, the results of use and to periodically report to the PMDA pursuant to the Pharmaceutical and Medical Devices Act. Price Regulation Japan's public medical insurance systems cover virtually the entire Japanese population. The public medical insurance system, however, does not cover any medical product which is not listed on the National Health Insurance (NHI) price list published by the Minister of the MHLW. Accordingly, an MAH of medical products must first have a new medical product listed on the NHI price list to obtain coverage under the public medical insurance system. VYVGART was listed on the NHI price list in April 2022 and the price was adjusted in February 2024. VYVDURA was listed in April 2024. The NHI price of a medical product is determined either by price comparison of comparable medical products with necessary adjustments for innovation, usefulness or size of the market; or, in the absence of comparable medical products, by the cost calculation method, determined after considering of the opinion of the manufacturer. Prices on the NHI price list are subject to revision, generally once every year, based on the actual prices at which the medical products are purchased by medical institutions. 1.7.4 Coverage, Pricing and Reimbursement Significant uncertainty exists as to the coverage and reimbursement status of any product candidates for which we may obtain regulatory approval. Even if our product candidates are approved for marketing, sales of such product candidates will depend, in part, on the extent to which third-party payors, including government health programs in the U.S. (such as Medicare and Medicaid), commercial health insurers, and managed care organizations, provide coverage and establish adequate reimbursement levels for such product candidates. Moreover, increasing efforts by governmental and third-party payors in the EU, the U.S. and other markets to cap or reduce healthcare costs may cause such organizations to limit both coverage and the level of reimbursement for newly approved products and, as a result, they may not cover or provide adequate payment for our product candidates. We expect to experience pricing pressures in connection with the sale of any of our product candidates due to the trend toward managed healthcare, the increasing influence of health maintenance organizations and additional legislative changes. The downward pressure on healthcare costs in general, particularly prescription drugs and surgical procedures and other treatments, has become very intense. As a result, increasingly high barriers are being erected to the entry of new products. In the U.S. and markets in other countries, patients generally rely on third-party payors to reimburse all or part of the costs associated with their treatment. Adequate coverage and reimbursement from governmental healthcare programs, such as Medicare and Medicaid, and commercial payors is critical to new product acceptance. Patients are unlikely to use any product candidates we may develop unless coverage is provided and reimbursement is adequate to cover a significant portion of the cost of such product candidates. Factors payors consider in determining reimbursement are based on whether the product is (i) a covered benefit under its health plan; (ii) safe, effective and medically necessary; (iii) appropriate for the specific patient; (iv) cost-effective; and (v) neither experimental nor investigational. The Medicare and Medicaid programs increasingly are used as models for how private payors and other governmental payors develop their coverage and reimbursement policies for drugs and biologics. Some third-party payors may require pre-approval of coverage for new or innovative devices or drug therapies before they will reimburse healthcare providers who use such therapies. It is difficult to predict at this time what third-party payors will decide with respect to the coverage and reimbursement for our product candidates. No uniform policy for coverage and reimbursement for drug products exists among third-party payors in the U.S. Therefore, coverage and reimbursement for drug products can differ significantly from payor to payor including formulary tier placement and utilization management requirements (if any). As a result, the coverage determination process is often a time-consuming and costly process that will require us to provide scientific and clinical support for the use of our products to each payor separately, with no assurance that coverage and adequate reimbursement will be applied consistently or obtained in the first argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Regulation 50

instance. The position of a product on a formulary generally determines the co-payment that a patient will need to make to obtain the product and can strongly influence the adoption of a product by patients and physicians. Third-party payors may limit coverage to specific products on a formulary, which might not include all of the approved products for a particular indication. Additionally, a payor’s decision to provide coverage for a product does not imply that an adequate reimbursement rate will be approved or that cost- sharing will be acceptable for patients. Coverage policies and third-party reimbursement rates may change at any time. Even if favorable coverage and reimbursement status is attained for one or more of our products for which we or our collaborators receive marketing approval, less favorable coverage policies and reimbursement rates may be implemented in the future. Third-party payors are increasingly challenging the price and examining the medical necessity and cost- effectiveness of medical products and services and imposing controls to manage costs, especially drugs when an equivalent generic drug or a less expensive therapy is available. It is possible that a third-party payor may consider our product candidate and other therapies (in some cases even off-label treatments) as substitutable and only offer to reimburse patients for the less expensive product. Even if we show improved efficacy or improved convenience of administration with our product candidate, pricing of existing drugs may limit the amount we will be able to charge for our product candidate. These payors may deny or revoke the reimbursement status of a given drug product or establish prices for new or existing marketed products at levels that are too low to enable us to realize an appropriate return on our investment in product development. If reimbursement is not available or is available only at limited levels, we may not be able to successfully commercialize our product candidates and may not be able to obtain a satisfactory financial return on products that we may develop. In Mainland China, VYVGART IV has been included in the National Reimbursement Drug List (NRDL) for the treatment of adults with gMG who are AChR-AB+ after going through price negotiations with the National Healthcare Security Administration (NHSA) since January 2024, which means that the price of this drug can be (partly) reimbursed by the social security program of Mainland China for the treatment of this indication in accordance with relevant rules within certain period. According to the current regulations of Mainland China, if we want our products in addition to VYVGART IV to be included in the NRDL or want VYVGART to be included in the NRDL for the treatment of other indications, we will need to go through price negotiations with the NHSA, for which purpose we will likely need to significantly reduce their prices. Although the inclusion of our products in the NRDL may increase the demand for the relevant products, our potential revenue from the sales of these products may still decrease as a result of lower prices. Furthermore, rules and regulations regarding reimbursement change frequently, in some cases at short notice, and we believe that changes in these rules and regulations are likely. Outside the U.S., we will face challenges in ensuring obtaining adequate coverage and payment for any product candidates we may develop. Pricing of prescription pharmaceuticals is subject to governmental control in many countries. In order to secure coverage and reimbursement for any product that might be approved for sale, we have needed and may need to conduct expensive pharmacoeconomic studies in order to demonstrate the medical necessity and cost-effectiveness of the product, and the cost of these studies would be in addition to the costs required to obtain FDA or other comparable marketing approvals. Conducting such studies could be expensive, involve additional risk and result in delays in our commercialization efforts. Even after pharmacogenomic studies are conducted, product candidates may not be considered medically necessary or cost-effective. A decision by a third-party payor not to cover any product candidates we may develop could reduce physician utilization of such product candidates once approved and have a material adverse effect on our sales, results of operations and financial condition. Third-party reimbursement and coverage may not be adequate to enable us to maintain price levels sufficient to realize an appropriate return on our investment in product development. The insurance coverage and reimbursement status of newly approved products for orphan diseases is particularly uncertain, and failure to obtain or maintain adequate coverage and reimbursement for any such product candidates could limit our ability to generate revenue. As noted above, in the U.S., we plan to have various programs to help patients afford our products, including patient assistance programs and co-pay coupon programs for eligible patients. More specifically, patients can enroll into MY VYVGART PATH™, a patient support program that provides personalized support from a nurse case manager and committed support team. In addition to providing support on questions on the treatment and on navigating the insurance process, the program provides a VYVGART Co-pay Program to eligible patients, aids in referring patients to charitable foundations that may be able to help with out-of- pocket costs and informs patients of financial assistance programs that may be available. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Regulation 51

The containment of healthcare costs also has become a priority of U.S. federal, state and international governments and the prices of pharmaceuticals have been a focus in this effort. Governments have shown significant interest in implementing cost-containment programs, including price controls, restrictions on reimbursement and requirements for substitution of generic products. Net prices for drugs may be reduced by mandatory discounts or rebates required by government healthcare programs or private payors and by any future relaxation of laws that presently restrict imports of drugs from countries where they may be sold at lower prices than in the U.S. Increasingly, third-party payors are requiring that drug companies provide them with predetermined discounts from list prices and are challenging the prices charged for medical products. We cannot be sure that reimbursement will be available for any future product candidate that we commercialize and, if reimbursement is available, the level of reimbursement. In addition, many pharmaceutical manufacturers must calculate and report certain price reporting metrics to the government, such as average sales price and best price. Penalties may apply in some cases when such metrics are not submitted accurately and timely. Adoption of price controls and cost-containment measures, and adoption of more restrictive policies in jurisdictions with existing controls and measures, could further limit our potential revenue from the sale of any products for which we may obtain approval. The delivery of healthcare in the EU, including the establishment and operation of health services and the pricing and reimbursement of medicinal products, is almost exclusively governed by national laws, rather than EU legislation. National governments and health service providers have different priorities and approaches to the delivery of healthcare and the pricing and reimbursement of products in that context. Therefore, in the EU, pricing and reimbursement schemes vary widely from EU Member State to another. Some EU Member States provide that products may be marketed only after a reimbursement price has been agreed. Some EU Member States may require the completion of additional studies that compare the cost-effectiveness of a particular product candidate to currently available therapies (so called health technology assessments) in order to obtain reimbursement or pricing approval. EU Member States may approve a specific price for a product or may instead adopt a system of direct or indirect controls on the profitability of the company placing the medicinal product on the market. Other EU Member States allow companies to fix their own prices for products but monitor and control prescription volumes and issue guidance to physicians to limit prescriptions. Recently, many EU Member States have increased the amount of discounts required on medicinal products and these efforts could continue as Member States attempt to further manage healthcare expenditures. For example, Germany introduced a specific discount on certain combination products with new active ingredients. The downward pressure on healthcare costs in general, particularly medicinal prescription products, has become intense. As a result, increasingly high barriers are being erected to the entry of new products. Political, economic and regulatory developments may further complicate pricing negotiations, and pricing negotiations may continue after reimbursement has been obtained. Reference pricing used by various EU Member States and parallel trade (arbitrage between low-priced and high-priced Member States) can further reduce prices. Special pricing and reimbursement rules may apply to orphan medicinal products. Inclusion of orphan drugs in reimbursement systems tend to focus on the medical usefulness, need, quality and economic benefits to patients and the healthcare system as for any drug. Acceptance of any medicinal product for reimbursement may come with cost, use and often volume restrictions, which again can vary by country. In addition, results-based rules of reimbursement may apply. There can be no assurance that any EU Member State that has price controls or reimbursement limitations for pharmaceutical products will allow favorable reimbursement and pricing arrangements for any of our products, if approved in those countries. Historically, products launched in the EU do not follow price structures of the U.S. and generally prices tend to be significantly lower. The above underlines that, outside the U.S., international operations are generally subject to extensive governmental price controls and other market regulations, and we believe the increasing emphasis on cost- containment initiatives in Europe, Canada and other countries has and will continue to put pressure on the pricing and usage of our product candidates. In many countries, the prices of medical products are subject to varying price control mechanisms as part of national health systems. Other countries allow companies to fix their own prices for medical products but monitor and control company profits. Additional foreign price controls or other changes in pricing regulation could restrict the amount that we are able to charge for our product candidates. Accordingly, in markets outside the U.S., the reimbursement for our products may be argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Regulation 52

reduced compared with the U.S. and may be insufficient to generate commercially reasonable revenue and profits. 1.7.5 Government Pricing and Reimbursement Programs for Marketed Drugs in the U.S. Medicaid, the 340B Drug Pricing Program, and Medicare Federal law requires that a pharmaceutical manufacturer, as a condition of having its drug and biological products receive federal reimbursement under Medicaid and Medicare Part B, must pay rebates to state Medicaid programs for all units of its covered outpatient drugs dispensed to Medicaid beneficiaries and paid for by a state Medicaid program under either a fee-for-service arrangement or through a managed care organization. This federal requirement is effectuated through a Medicaid drug rebate agreement between the manufacturer and the Secretary of U.S. Department of Health and Human Services (HHS). The Centers for Medicare & Medicaid Services (CMS) administers the Medicaid drug rebate agreements, which provide, among other things, that the drug manufacturer will pay rebates to each state Medicaid agency on a quarterly basis and report certain price information on a monthly and quarterly basis. The rebates are based on prices reported to CMS by manufacturers for their covered outpatient drugs, including average manufacturer price (AMP) and best price. Effective January 1, 2024, the Medicaid total rebate amount is no longer capped at 100% of a covered outpatient drug’s AMP, which means that a manufacturer could pay a total rebate amount on a unit of the drug that is greater than the average price the manufacturer receives for the drug. The terms of participation in the Medicaid drug rebate program impose an obligation to correct the prices reported in previous quarters, as may be necessary. Any such corrections could result in additional or lesser rebate liability, depending on the direction of the correction. In addition to retroactive rebates, if a manufacturer were found to have knowingly submitted false information to the government, federal law provides for civil monetary penalties for failing to provide required information, late submission of required information, and false information. A manufacturer must also participate in a federal program known as the 340B drug pricing program in order for federal funds to be available to pay for the manufacturer’s drug and biological products under Medicaid and Medicare Part B. Under this program, the participating manufacturer agrees to charge certain safety net healthcare providers no more than an established discounted price for its covered outpatient drugs. The formula for determining the discounted price is defined by statute and is based on the AMP and the unit rebate amount as calculated under the Medicaid drug rebate program, discussed above. Manufacturers are required to report pricing information to the Health Resources and Services Administration on a quarterly basis. The Health Resources and Services Administration has also issued regulations relating to the calculation of the ceiling price as well as imposition of civil monetary penalties for each instance of knowingly and intentionally overcharging a 340B covered entity. Federal law also requires that manufacturers report data on a quarterly basis to CMS regarding the pricing of drugs that are separately reimbursable under Medicare Part B. These are generally drugs and biologics, such as injectable products, that are administered incident to a physician service and are not generally self- administered. The pricing information submitted by manufacturers is the basis for reimbursement to physicians and suppliers for drugs covered under Medicare Part B. Under the Inflation Reduction Act (IRA), manufacturers are also required to provide quarterly rebates for certain single-source drugs and biologics (including biosimilars) covered under Medicare Part B with prices that increase faster than the rate of inflation. This requirement started on January 1, 2023 for drugs approved on or before December 1, 2020 and begins six quarters after a drug is first marketed for all other drugs. As with the Medicaid drug rebate program, federal law provides for civil monetary penalties for failing to provide required information, late submission of required information, and false information. Additionally, the Infrastructure Investment and Jobs Act added a requirement, effective January 1, 2023, for manufacturers of certain single-source drugs (including biologics and biosimilars) separately paid for under Medicare Part B for at least 18 months and marketed in single-dose containers or packages (known as refundable single-dose containers or single-use package drugs) to provide annual refunds for any portions of the dispensed drug that are unused and discarded if those unused or discarded portions exceed an argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Regulation 53

applicable percentage defined by statute or regulation. Manufacturers will be subject to periodic audits and those that fail to pay refunds for their refundable single-dose containers or single-use package drugs shall be subject to civil monetary penalties. Medicare Part D provides prescription drug benefits for seniors and people with disabilities. Beginning in 2025, the IRA eliminates the coverage gap phase and associated manufacturer discounts under Medicare Part D, significantly lowers the enrollee maximum out-of-pocket cost and establishes a new manufacturer discount program, which requires 10% discounts in the initial phase, and 20% discounts in the catastrophic phase. Although these discounts represent a lower percentage of enrollees’ costs than coverage gap discounts, the new manufacturer contribution during the catastrophic phase could be considerable for certain high-cost drugs and the total contributions by manufacturers to a Part D enrollee’s drug expenses may exceed those currently provided. The IRA also requires manufacturers to provide annual Medicare Part D rebates for single-source drugs and biological products with prices that increase faster than the rate of inflation. The IRA also allows HHS to directly negotiate the selling price of a statutorily specified number of drugs and biologics each year that CMS reimburses under Medicare Part B and Part D. Only high-expenditure single- source biologics that have been approved for at least 11 years (7 years for single-source drugs) can qualify for negotiation, with the negotiated price taking effect two years after the selection year. In July 2025, Congress expanded the IRA’s orphan drug exclusion to protect from selection drugs that are indicated only for orphan indications, as well as to extend the time before an orphan drug may be selected if it is later approved for a non-orphan indication. Negotiations for Medicare Part D products began in 2023 with the negotiated price taking effect in 2026, and negotiations for Medicare Part B products will begin in 2026 with the negotiated price taking effect in 2028. U.S. Federal Contracting and Pricing Requirements Manufacturers are also required to make their covered drugs, which are generally drugs approved under NDAs or BLAs, available to authorized users of the Federal Supply Schedule (FSS) of the General Services Administration. The law also requires manufacturers to offer deeply discounted FSS contract pricing for purchases of their covered drugs by the Department of Veterans Affairs, the Department of Defense, the Coast Guard, and the Public Health Service (including the Indian Health Service) in order for federal funding to be available for reimbursement or purchase of the manufacturer’s drugs under certain federal programs. FSS pricing to those four federal agencies for covered drugs must be no more than the Federal Ceiling Price (FCP), which is at least 24% below the Non-Federal Average Manufacturer Price (Non-FAMP) for the prior year. The Non-FAMP is the average price for covered drugs sold to wholesalers or other middlemen, net of any price reductions. The accuracy of a manufacturer’s reported Non-FAMPs, FCPs, or FSS contract prices may be audited by the government. Among the remedies available to the government for inaccuracies is recoupment of any overcharges to the four specified federal agencies based on those inaccuracies. If a manufacturer were found to have knowingly reported false prices, in addition to other penalties available to the government, the law provides for significant civil monetary penalties per incorrect item. Finally, manufacturers are required to disclose in FSS contract proposals all commercial pricing that is equal to or less than the proposed FSS pricing, and subsequent to award of an FSS contract, manufacturers are required to monitor certain commercial price reductions and extend commensurate price reductions to the government, under the terms of the FSS contract Price Reductions Clause. Among the remedies available to the government for any failure to properly disclose commercial pricing and/or to extend FSS contract price reductions is recoupment of any FSS overcharges that may result from such omissions. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Regulation 54

1.7.6 Healthcare Law and Regulation Healthcare providers and third-party payors play a primary role in the recommendation and prescription of pharmaceutical products that are granted marketing approval. Our current and future arrangements with providers, researchers, consultants, third-party payors and customers are subject to broadly applicable federal and state fraud and abuse, anti-kickback, false claims, transparency and patient privacy laws and regulations and other healthcare laws and regulations that may constrain our business and/or financial arrangements. Restrictions under applicable federal and state healthcare laws and regulations include, without limitation, the following: • the U.S. federal Anti-Kickback Statute (AKS) prohibits, among other things, persons and entities from knowingly and willfully soliciting, receiving, offering, or paying remuneration, directly or indirectly, in cash or in kind, to induce or reward either the referral of an individual for, or the purchase, lease, order or recommendation of, any good facility, item, or service, for which payment may be made, in whole or in part, under a federal healthcare program such as Medicare and Medicaid. This statute has been interpreted to apply to arrangements between pharmaceutical manufacturers on the one hand and prescribers, purchasers formulary managers and other persons and entities on the other. Although there are a number of statutory exceptions and regulatory safe harbors protecting certain activities from prosecution, the exceptions and safe harbors are drawn narrowly, and arrangements may be subject to scrutiny or penalty if they do not fully satisfy all elements of an available exception or safe harbor. A person or entity can be found guilty of violating the AKS without actual knowledge of the statute or specific intent to violate it. In addition, the government may assert that a claim including items or services resulting from a violation of the AKS constitutes a false or fraudulent claim for purposes of the federal False Claims Act or federal civil money penalties statute. Violations of the AKS carry potentially significant civil and criminal penalties, including imprisonment, fines, administrative civil monetary penalties, and exclusion from participation in federal healthcare programs. • the U.S. federal false claims and civil monetary penalties laws, including the civil False Claims Act and federal civil monetary penalty laws, which, among other things, impose criminal and civil penalties, including through civil whistleblower or qui tam actions, against individuals or entities for knowingly presenting, or causing to be presented, to the U.S. federal government, claims for payment or approval that are false or fraudulent, knowingly making, using or causing to be made or used, a false record or statement material to a false or fraudulent claim or obligation to pay or transmit money to the federal government, or from knowingly making a false statement to avoid, decrease or conceal an obligation to pay money to the U.S. federal government. In addition, the government may assert that a claim including items and services resulting from a violation of the AKS constitutes a false or fraudulent claim for purposes of the False Claims Act. Manufacturers can be held liable under the False Claims Act even when they do not submit claims directly to government payors if they are deemed to “cause” the submission of false or fraudulent claims. The False Claims Act also permits a private individual acting as a “whistleblower” to bring qui tam actions on behalf of the federal government alleging violations of the False Claims Act and to share in any monetary recovery. When an entity is determined to have violated the federal civil False Claims Act, the government may impose civil fines and penalties for each false claim, plus treble damages, and exclude the entity from participation in Medicare, Medicaid and other federal healthcare programs; • the U.S. federal Health Insurance Portability and Accountability Act of 1996 (HIPAA) which imposes criminal and civil liability for, among other things, knowingly and willfully executing, or attempting to execute, a scheme to defraud any healthcare benefit program, or obtaining by means of false or fraudulent pretenses, representations, or promises, any of the money or property owned by, or under the custody or control of, any healthcare benefit program, regardless of the pay (e.g., public or private) or knowingly and willfully falsifying, concealing or covering up a material fact or making any materially false statement, in connection with the delivery of, or payment for, healthcare benefits, items or services relating to healthcare matters; similar to the AKS, a person or entity does not need to have actual knowledge of the statute or specific intent to violate it in order to have committed a violation; • HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act of 2009 (HITECH) and its implementing regulations, and as amended again by the Omnibus Rule in 2013, which imposes certain obligations, including mandatory contractual terms, with respect to safeguarding the privacy, security and transmission of individually identifiable health information without appropriate authorization by covered entities subject to the Final HIPAA Omnibus Rule, i.e., certain covered health plans, healthcare clearinghouses and healthcare providers, as well as their business associates, those argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Regulation 55

independent contractors or agents of covered entities that perform certain services for or on their behalf involving the use or disclosure of individually identifiable health information. HITECH also created new tiers of civil monetary penalties, amended HIPAA to make civil and criminal penalties directly applicable to business associates and possibly other persons, and gave state attorneys general new authority to file civil actions for damages or injunctions in federal courts to enforce the federal HIPAA laws and seek attorneys’ fees and costs associated with pursuing federal civil actions; • the federal transparency requirements known as the federal Physician Payments Sunshine Act, under the Patient Protection and Affordable Care Act, as amended by the Health Care and Education Reconciliation Act of 2010 (collectively, the ACA), which requires certain manufacturers of drugs, devices, biologics and medical supplies to report annually to CMS information related to payments and other transfers of value made by that entity to physicians (currently defined to include doctors, dentists, optometrists, podiatrists and chiropractors), certain non-physician providers such as physician assistants and nurse practitioners and teaching hospitals, as well as ownership and investment interests held by physicians and their immediate family members. Failure to submit required information may result in civil monetary penalties for all payments, transfers of value or ownership or investment interests that are not timely, accurately, and completely reported in an annual submission; • federal government price reporting laws, which require us to calculate and report complex pricing metrics in an accurate and timely manner to government programs; • federal consumer protection and unfair competition laws, which broadly regulate marketplace activities and activities that potentially harm consumers; • analogous state and local laws and regulations, including: state anti-kickback and false claims laws; state laws that require pharmaceutical companies to comply with the pharmaceutical industry’s voluntary compliance guidelines and the relevant compliance guidance promulgated by the U.S. federal government, or otherwise restrict payments that may be made to healthcare providers and other potential referral sources; state and local laws that require the licensure of sales representatives; state laws that require drug manufacturers to report information related to payments and other transfers of value to physicians and other healthcare providers or marketing expenditures and pricing information; state laws governing the privacy and security of health information in certain circumstances, many of which differ from each other in significant ways and may not have the same effect; and state laws related to insurance fraud in the case of claims involving private insurers; and • EU, UK and other foreign law equivalents, including reporting requirements detailing interactions with and payments to healthcare providers and data privacy and security laws and regulations that may be more stringent than those in the U.S. State and foreign laws, including for example the EU General Data Protection Regulation (GDPR), also govern the privacy and security of health information in some circumstances, many of which differ from each other in significant ways and often are not preempted by HIPAA, thus complicating compliance efforts. There are ambiguities as to what is required to comply with these state requirements and if we fail to comply with an applicable state law requirement, we could be subject to penalties. We have and will continue to spend substantial time and money to ensure that our business arrangements with third parties comply with applicable healthcare laws and regulations. Recent healthcare reform legislation has strengthened these federal and state healthcare laws. Because of the breadth of these laws and the narrowness of the statutory exceptions and safe harbors available, it is possible that some of our business activities could be subject to challenge under one or more of such laws. Other laws that may affect our ability to operate include: • the anti-inducement law prohibits, among other things, the offering or giving of remuneration, which includes, without limitation, any transfer of items or services for free or for less than fair market value (with limited exceptions), to a Medicare or Medicaid beneficiary that the person know or should know is likely to influence the beneficiary’s selection of a particular supplier of items or services reimbursable by a federal or state governmental program; and • European and other foreign law equivalents of each of the laws, including reporting requirements detailing interactions with and payments to healthcare providers. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Regulation 56

In the U.S., to help patients afford our approved product, we may utilize programs to assist them, including patient assistance programs and co-pay coupon programs for eligible patients. Government enforcement agencies have shown increased interest in pharmaceutical companies’ product and patient assistance programs, including reimbursement support services, and a number of investigations into these programs have resulted in significant civil and criminal settlements. In addition, at least one insurer has directed its network pharmacies to no longer accept co-pay coupons for certain specialty drugs the insurer identified. Our co-pay coupon programs could become the target of similar insurer actions. In addition, in November 2013, the CMS issued guidance to the issuers of qualified health plans sold through the ACA’s marketplaces encouraging such plans to reject patient cost-sharing support from third parties and indicating that the CMS intends to monitor the provision of such support and may take regulatory action to limit it in the future. The CMS subsequently issued a rule requiring individual market qualified health plans to accept third-party premium and cost-sharing payments from certain government-related entities. In September 2014, the Office of the Inspector General of the HHS issued a Special Advisory Bulletin warning manufacturers that they may be subject to sanctions under the AKS and/or civil monetary penalty laws if they do not take appropriate steps to exclude Part D beneficiaries from using co-pay coupons. Accordingly, companies exclude these Part D beneficiaries from using co-pay coupons. Additionally, certain third-party payors are modifying benefit designs based on the availability of manufacturer cost-sharing assistance (e.g., copay accumulator or maximizer programs). Following a federal district court decision vacating the provisions of the 2021 Notice of Benefit and Payment Parameter final rule that provided health plans with discretion whether to include manufacturer assistance toward the cost-sharing limit, CMS stated its intent to address this issue in future rulemaking. It is possible that changes in insurer policies regarding co-pay coupons and/or the introduction and enactment of new legislation or regulatory action could restrict or otherwise negatively affect these patient support programs, which could result in fewer patients using affected products, and therefore could have a material adverse effect on our sales, business, and financial condition. Third-party patient assistance programs that receive financial support from companies have become the subject of enhanced government and regulatory scrutiny. The Office of the Inspector General of the HHS has established guidelines that suggest that it is lawful for pharmaceutical manufacturers to make donations to charitable organizations who provide co-pay assistance to Medicare patients, provided that such organizations, among other things, are bona fide charities, are entirely independent of and not controlled by the manufacturer, provide aid to applicants on a first-come basis according to consistent financial criteria and do not link aid to use of a donor’s product. However, donations to patient assistance programs have received some negative publicity and have been the subject of multiple government enforcement actions, related to allegations regarding their use to promote branded pharmaceutical products over other less costly alternatives. Specifically, in recent years, there have been multiple settlements resulting out of government claims challenging the legality of their patient assistance programs under a variety of federal and state laws. It is possible that we may make grants to independent charitable foundations that help financially needy patients with their premium, co-pay, and co-insurance obligations. If we choose to do so, and if we or our vendors or donation recipients are deemed to fail to comply with relevant laws, regulations or evolving government guidance in the operation of these programs, we could be subject to damages, fines, penalties, or other criminal, civil, or administrative sanctions or enforcement actions. We cannot ensure that our compliance controls, policies, and procedures will be sufficient to protect against acts of our employees, business partners, or vendors that may violate the laws or regulations of the jurisdictions in which we operate. Regardless of whether we have complied with the law, a government investigation could impact our business practices, harm our reputation, divert the attention of management, increase our expenses, and reduce the availability of foundation support for our patients who need assistance. Violations of these laws or any future enacted laws can subject us to criminal, civil and administrative sanctions including monetary penalties, damages, fines, disgorgement, individual imprisonment and exclusion from participation in government funded healthcare programs, such as Medicare and Medicaid, additional reporting requirements and oversight if we become subject to a corporate integrity agreement or similar agreement to resolve allegations of non-compliance with these laws, reputational harm, and we may be required to curtail or restructure our operations. Moreover, we expect that there will continue to be federal and state laws and regulations, proposed and implemented, that could impact our future operations and business. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Regulation 57

Because of the breadth of these laws and the narrowness of the statutory exceptions and safe harbors available, it is possible that some of our business activities could be subject to challenge under one or more of such laws. Ensuring that our internal operations and future business arrangements with third parties comply with applicable healthcare laws and regulations will involve substantial costs. It is possible that governmental authorities will conclude that our business practices do not comply with current or future statutes, regulations, agency guidance or case law involving applicable fraud and abuse or other healthcare laws and regulations. If our operations are found to be in violation of any of the laws described above or any other governmental laws and regulations that may apply to us, we may be subject to significant penalties, including administrative, civil and criminal penalties, damages, fines, disgorgement, the exclusion from participation in federal and state healthcare programs, individual imprisonment, reputational harm, and the curtailment or restructuring of our operations, as well as additional reporting obligations and oversight if we become subject to a corporate integrity agreement or other agreement to resolve allegations of non-compliance with these laws. Further, defending against any such actions can be costly and time-consuming, and may require significant financial and personnel resources. Therefore, even if we are successful in defending against any such actions that may be brought against us, our business may be impaired. If any of the physicians or other providers or entities with whom we expect to do business are found to not be in compliance with applicable laws, they may be subject to criminal, civil or administrative sanctions, including exclusions from government funded healthcare programs and imprisonment. If any of the above occur, our ability to operate our business and our results of operations could be adversely affected. 1.7.7 Healthcare Reform In the U.S., the EU and other foreign jurisdictions, there have been a number of legislative and regulatory changes to the healthcare systems that could affect our future results of operations. In particular, there have been and continue to be a number of initiatives at the U.S. federal and state levels that seek to reduce healthcare costs and improve the quality of healthcare. For example, the ACA, effective since March 2010, is a sweeping law intended to broaden access to health insurance, reduce or constrain the growth of healthcare spending, enhance remedies against fraud and abuse, add new transparency requirements for the healthcare and health insurance industries, impose new taxes and fees on the health industry and impose additional health policy reforms. Healthcare reforms that have been adopted, and that may be adopted in the future, could result in further reductions in coverage and levels of reimbursement for pharmaceutical products, increases in rebates payable under U.S. government rebate programs and additional downward pressure on pharmaceutical product prices. As discussed above, in August 2022, the IRA was enacted codifying, among other things: a Medicare drug price negotiation program, under which HHS directly negotiates the selling price of statutorily specified number of Part B and Part D drugs and biologics each year; inflation rebates which penalizes drug manufacturers that increase prices of Medicare Part B and Part D drugs at a rate greater than the rate of inflation; and a redesign of the Part D benefit. The IRA permits the Secretary of HHS to implement many of these provisions through guidance, as opposed to regulation, for the initial years. Manufacturers that fail to comply with the IRA may be subject to various penalties, including civil monetary penalties. The IRA also extends enhanced subsidies for individuals purchasing health insurance coverage in ACA marketplaces through plan year 2025. To date, none of the legislative attempts to extend the subsidies has been enacted. These IRA provisions began taking effect progressively starting in 2023, although certain policies have been subject to legal challenges. For example, the provisions related to the negotiation of selling prices of high-expenditure single-source drugs and biologics have been challenged in multiple lawsuits. Additionally, we cannot predict whether the U.S. Congress will further amend the IRA or if the government will adopt new or different interpretations of the law in future guidance or rulemaking. However, at this time, the Trump administration is continuing to implement the IRA and to defend the law in litigation. While it is unclear how the IRA will be implemented in the future and the outcome of the litigation, it will likely have a significant impact on the pharmaceutical industry. In addition, the Trump administration has taken several steps to try to align U.S. drug prices with drug prices in other countries through an approach known as most favored nation (MFN) pricing. For example, on May 12, 2025, the current Presidential administration published an executive order which, among other actions, instructed HHS to communicate MFN price targets. The executive order also directed certain steps if “significant progress towards [MFN] pricing . . . is not delivered.” On July 31, 2025, the U.S. President issued letters to 17 pharmaceutical companies (not including argenx), calling on those manufacturers and argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Regulation 58

“every manufacturer” to take the following steps within 60 days: extend MFN pricing to Medicaid for all of their existing drugs; guarantee Medicare, Medicaid, and commercial payors receive MFN pricing for newly- launched drugs; return increased revenues abroad to American patients and taxpayers; and participate in direct-to-consumer or direct-to-business distribution models to provide “high-volume, high rebate” drugs at MFN pricing. Certain manufacturers have entered into direct agreements with the government. On November 6, 2025, CMS announced the GENErating cost Reductions fOr U.S. Medicaid (GENEROUS) Model under its Center for Medicare and Medicaid Innovation authority (CMMI). The GENEROUS Model is a voluntary model that tests the impact of CMS-facilitated supplemental rebate agreements that align the Medicaid net price with a defined MFN price. In December 2025, CMS issued the Global Benchmark for Efficient Drug Pricing (GLOBE) Model and Guarding U.S. Medicare Against Rising Drug Costs (GUARD) Model proposed rules under its Center for Medicare and Medicaid Innovation authority. The GLOBE and GUARD models would require manufacturers to pay additional rebates for certain drugs based on the difference between the Medicare price and the price in market basket countries. CMS proposes that the agency would apply the new rebate requirement to utilization by approximately 25% of Medicare Part B fee-for-service enrollees (under GLOBE) and 25% of Medicare Part D enrollees (under GUARD). It is uncertain if these proposed rules will be finalized and if they are, how they will impact our business. Additionally, in Congress, there are pending legislative proposals that, if enacted, would require MFN pricing in certain healthcare programs. We cannot predict if any of these legislative proposals will be enacted, how they would be implemented, and how they could impact our business. We expect that additional U.S. federal healthcare reform measures will be adopted in the future, any of which could limit the amounts that the U.S. federal government will pay for healthcare products and services, which could result in reduced demand for our product candidates or additional pricing pressures. Further, legislative and regulatory proposals have been made to expand post-approval requirements and restrict sales and promotional activities for pharmaceutical products. We cannot be sure whether additional legislative changes will be enacted, or whether FDA regulations, guidance or interpretations will be changed, or what the impact of such changes on the marketing approvals, if any, of our product candidates, may be. In addition, increased scrutiny by the U.S. Congress of the FDA’s approval process may significantly delay or prevent marketing approval, as well as subject us to more stringent product labeling and post- marketing conditions and other requirements. Individual states in the U.S. have also become increasingly aggressive in passing legislation and implementing regulations designed to control pharmaceutical and biological product pricing, including price or patient reimbursement constraints, affordability review boards, discounts, restrictions on certain product access and marketing cost disclosure and transparency measures, and, in some cases, designed to encourage importation from other countries and bulk purchasing. Legally mandated price controls on payment amounts by third-party payors or other restrictions could harm our business, results of operations, financial condition and prospects. In addition, regional healthcare authorities and individual hospitals are increasingly using bidding procedures to determine what pharmaceutical products and which suppliers will be included in their prescription drug and other healthcare programs. This could reduce the ultimate demand for our products or put pressure on our product pricing, which could negatively affect our business, results of operations, financial condition and prospects. In international markets, reimbursement and healthcare payment systems vary significantly by country (including across the EU’s individual member states), and many countries have instituted price ceilings on specific products and therapies. Future political, economic, and regulatory developments may further affect the ability of pharmaceutical companies to profitably commercialize current and future products. 1.7.8 Environmental Aspects which may Influence the Use of our Material Fixed Assets Our primary research and development activities take place in our facilities in Zwijnaarde, Belgium. For these activities we require, and have obtained, the necessary environmental and biohazard permits from the responsible governments, required by us for the manner in which we use said facilities. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Regulation 59

1.8 Documents on display We are subject to the information reporting requirements of the U.S. Securities Exchange Act of 1934, as amended (Exchange Act) applicable to foreign private issuers. Accordingly, we are required to file reports and other information with the SEC. Those reports may be inspected without charge at the locations described below. As a foreign private issuer, we are exempt from the rules under the Exchange Act related to the furnishing and content of proxy statements, and our officers, directors and principal shareholders are exempt from the reporting and short-swing profit recovery provisions contained in Section 16 of the Exchange Act. In addition, we are not required under the Exchange Act to file periodic reports and financial statements with the SEC as frequently or as promptly as U.S. companies whose securities are registered under the Exchange Act. Nevertheless, we will file with the SEC an Annual Report containing financial statements that have been examined and reported on, with an opinion expressed by an independent registered public accounting firm. We maintain a corporate website at www.argenx.com. We make available on our website, free of charge, our Annual Report and the text of our reports on Form 6-K, including any amendments to these reports, as well as certain other SEC filings, as soon as reasonably practicable after they are electronically filed with or furnished to the SEC. Information contained on, or that can be accessed through, our website does not constitute a part of this Annual Report. We have included our website address in this Annual Report solely as an inactive textual reference. The SEC maintains a website (www.sec.gov) that contains reports and other information regarding registrants, such as argenx SE, that file electronically with the SEC. With respect to references made in this Annual Report to any contract or other document of argenx SE, such references are not necessarily complete and you should refer to the exhibits attached or included elsewhere to this Annual Report for copies of the actual contract or document. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Documents on display 60

Risk Factors 2.1 Summary Risk Factors 62 2.2 Risk Factors Related to Commercialization of argenx’s Products and Product Candidates, Including for New Indications 64 2.3 Risk Factors Related to the Development and Clinical Testing of argenx’s Products and Product Candidates 71 2.4 Risk Factors Related to argenx’s Dependence on Third Parties 74 2.5 Risk Factors Related to Other Government Regulations 77 2.6 Risk Factors Related to argenx’s Financial Position 82 2.7 Risk Factors Related to argenx’s Business and Industry 83 2.8 Risk Factors Related to argenx’s Intellectual Property 86 2.9 Risk Factors Related to argenx’s Organization and Operations 90 2.10 Risk Factors Related to the ADSs 93 2.11 Risk Factors Related to being a Foreign Private Issuer or a Dutch Company 95 argenx Annual Report 2025 61 2

2 Risk Factors Our business faces significant risks, including those described below. You should carefully consider all of the information set forth in this Annual Report and in our other filings with the SEC, including the following risk factors. Our business, financial condition or results of operations could be materially and adversely affected if any of these risks occur. These are not the only risks argenx faces. Additional risks and uncertainties not presently known to argenx or that it currently considers immaterial or not specific may also impair its business, results of operation and financial condition. This report also contains forward- looking statements that involve risks and uncertainties. Our actual results could differ materially and adversely from those anticipated in these forward-looking statements as a result of certain factors including the risks described below and elsewhere in this Annual Report and as may be described in our subsequent SEC filings. See “Forward-Looking Statements”. 2.1 Summary Risk Factors Our ability to implement our business strategy is subject to numerous risks that you should be aware of before making an investment decision. These risks are described more fully below. These risks include, among others: • The commercial success of our products and product candidates, including in new indications or methods of administration, will depend on the degree of market acceptance. • We face significant competition for our drug discovery and development efforts. • We will face significant challenges in successfully commercializing our products and additional product candidates after they are launched. • Our products and product candidates for which we have obtained or intend to seek approval as biological products, including for new indications, may face biosimilar competition. • Enacted and future legislation and regulations could impact demand for our products which could impact our business and future results of operations. • We are subject to government pricing laws, regulation and enforcement, which affect the prices we may charge the government for our products and the reimbursement our customers may obtain from the government. Changes in such laws, regulation, and enforcement may place downward pressure on the prices we can charge in the marketplace, and our failure to comply with these laws could harm our results, operations and/or financial conditions. • We may not obtain or maintain adequate pricing and coverage or reimbursement status for our products and product candidates. • If we fail to obtain orphan drug designation or we do not have valid and enforceable patents covering our products and their uses and product candidates and fail to obtain and/or maintain orphan drug exclusivity for our products or product candidates, our competitors may be able to sell products to treat the same conditions and our revenue may be reduced. • Failure to successfully identify, select and develop our products in other indications, or additional products or product candidates could impair our ability to grow. • Failure to successfully develop or obtain marketing approval for our products and product candidates could negatively impact our business. • Certain of our clinical trials have not succeeded, and may in the future also not succeed, and even if they succeed, we may not obtain regulatory approval for our products or product candidates or regulatory approval may be delayed. • If we decide to pursue accelerated approval for any of our product candidates, it may not lead to faster development or regulatory review or approval and we may still need to conduct additional clinical trials, which could increase the expense of obtaining, if at all, necessary marketing approvals. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Summary Risk Factors 62

• Our products and product candidates may have serious adverse, undesirable or unacceptable side effects, and we or others may identify undesirable or unacceptable side effects caused by any of our products or product candidates before and after they have received marketing approval. • If our target patient population is smaller than expected, we are unable to successfully enroll and retain patients in our clinical trials, or experience significant delays in doing so, we may not realize the full commercial potential of any products or product candidates. • We rely, and expect to continue to rely, on third parties to conduct some of our research activities, manufacturing and clinical trials and for parts of the development and commercialization of our existing and future research programs, products and product candidates. If our relationships with such third parties are not successful, our business may be adversely affected. • Disruptions caused by our reliance on third parties for our raw materials and manufacturing process may delay or disrupt our business, product development and commercialization efforts. • Accuracy and timing of our financial reporting is partially dependent on information received from third- party partners, which we do not control. • We and our third-party manufacturers and suppliers may become exposed to liability, fines, penalties or other sanctions and substantial expenses in connection with environmental compliance or remediation activities. • We are subject to healthcare laws, regulation and potential enforcement. The failure to comply with these laws could harm our results, operations and/or financial condition. • Our performance tracked by our Environmental, Social and Governance metrics is subject to risks and the outcomes may not achieve the anticipated benefits or align with new regulations and stakeholders’ expectations. • We may not be able to be profitable or sustain net profitability in the future and may require additional financing to fund our operations. • We may become exposed to costly and damaging liability claims. • We may engage in strategic transactions, including acquisitions, collaborations, licenses or investments in other companies or technologies, and we may not realize the benefits of such transactions. • Our business and operations could suffer in the event of system failures or unauthorized or inappropriate use of or access to our systems. • We may be unable to adequately maintain, enforce or protect our intellectual property rights in products, product candidates and platform technologies which could adversely affect our ability to maximize the value for patients in our marketed products and product candidates. • Intellectual property litigation could lead to substantial resource diversion or issued patents could be found invalid, not infringed, or unenforceable if challenged in the applicable patent office or court. • Our future growth and ability to compete depends on maintaining our culture, retaining our key personnel and recruiting additional qualified personnel. • Global geo- and socio-political threats and macro-economic uncertainty and other unforeseen political crises could materially and adversely affect our business and financial performance. • Holders of our ADSs have fewer rights than our ordinary shareholders. • The price of our ADSs may be volatile and may fluctuate due to factors beyond our control. An active public trading market may not be sustained. • Claims of U.S. civil liabilities may not be enforceable against us or the members of our Senior Management Team and our Board of Directors. • As a foreign private issuer, we are exempt from various rules and regulations that a U.S. domestic public company would be required to follow, including those requirements under U.S. securities laws and Nasdaq listing standards. • We may lose our foreign private issuer status which would then require us to comply with the Exchange Act’s domestic reporting regime and cause us to incur significant legal, accounting and other expenses. • If we were to be classified as a passive foreign investment company for U.S. federal income tax purposes, this could result in adverse U.S. tax consequences to certain U.S. holders. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Summary Risk Factors 63

2.2 Risk Factors Related to Commercialization of argenx’s Products and Product Candidates, Including for New Indications The commercial success of our products and product candidates, including in new indications or methods of administration, will depend on the degree of market acceptance. Our products and product candidates, including for any new indications or methods of administration, if and when approved and available on the market, may never achieve an adequate level of acceptance by physicians, patients, the medical community, or healthcare payors for us to be profitable or sustain net profitability in the future. This will depend on a number of factors, many of which are beyond our control, including, but not limited to: • consumer perceptions or publicity regarding our business or the efficacy, safety and quality of the products and product candidates in our profile, our clinical trials for new indications, or any similar products distributed by other companies, and the prevalence and severity of any adverse effects discovered before or after marketing approval has been received; • approval may be for indications, dosage and methods of administration or patient populations that are not as broad as intended or desired; • changes in the standard of care for the targeted indications for any product and product candidate; • relative availability, cost, and convenience of alternative approved therapies; • labeling may require significant use or distribution restrictions or safety warnings; • acceptance by physicians, public health bodies, patients and healthcare payors of each product as safe, effective and cost-effective; and • patients continued commitment required to receive periodic in-center infusions. In addition, because we are developing our products and product candidates for the treatment of different indications, negative results in a clinical trial evaluating the efficacy and safety of a product or product candidate for one indication, including by one of our competitors, could negatively impact the perception of the efficacy and safety of such product or product candidate in a different indication, which could have an adverse effect on our reputation, commercialization efforts and financial condition. Moreover, efforts to educate the medical community and third-party payors on the benefits of our products and product candidates may require significant resources and may never be successful. If our product candidates or methods of use of existing products or new indications fail to gain market acceptance, it will have a material adverse impact on our ability to generate revenues. Even if some products achieve market acceptance, they may not be able to retain market acceptance and/or the market may prove not to be large enough to allow us to generate significant revenues. We face significant competition for our drug discovery and development efforts. The market for pharmaceutical products is highly competitive and characterized by rapidly growing understanding of disease biology, quickly changing technologies, strong intellectual property barriers to entry, and a multitude of companies involved in the creation, development, and commercialization of novel therapeutics. Many of these companies are highly sophisticated and often strategically collaborate with each other. Competition in the autoimmune field is intense and involves multiple mAbs, other biologics and small molecules either already marketed or in development by many different companies including, but not limited to, large pharmaceutical companies such as AstraZeneca plc, AbbVie, Inc., Amgen, Inc., Biogen Inc., GlaxoSmithKline plc, F. Hoffman-La Roche AG, Johnson & Johnson Innovation, Inc. and Novartis AG. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Risk Factors Related to Commercialization of argenx’s Products and Product Candidates, Including for New Indications 64

In addition, these and other pharmaceutical companies have mAbs or other biologics in clinical development for the treatment of autoimmune diseases. Currently, our commercial revenue is generated by VYVGART and VYVGART SC in gMG, CIDP and ITP (Japan only). We face and expect to continue to face intense competition from other biopharmaceutical companies, who have launched or are developing products for the treatment of gMG and/or CIDP and other autoimmune diseases, including products that are in the same class as VYVGART, as well as products that are similar to some of our product candidates. Competition for other potential future indications is also fierce, with significant development by competitors in almost all of the indications we are currently developing or planning to develop for our product or product candidates. For example, we are aware of certain biopharmaceutical companies selling products for the treatment of adult patients with gMG, and several biopharmaceutical companies are developing drugs that may have utility for the treatment of gMG and/or CIDP. Competitive product launches may erode future sales of our products, including our existing products and those currently under development, or result in unanticipated product obsolescence. Such launches continue to occur, and potentially competitive products are in various stages of development. We could also face competition for use of limited international infusion sites, particularly in new markets as competitors launch new products. We cannot predict with accuracy the timing or impact of the introduction of competitive products that treat diseases and conditions like those treated by our products or product candidates. In addition, our competitors and potential competitors compete with us in recruiting and retaining qualified personnel in all areas of our business, establishing clinical trial sites, registering patients for clinical trials, as well as in acquiring technologies complementary to, or necessary for, the development of our products. Competition is also increasing from companies that are utilizing artificial intelligence and other computational approaches for the development of products. These competitors may incorporate artificial intelligence into their businesses more quickly or more successfully than us, which could impair our ability to compete effectively and adversely affect our results of operations. We anticipate that we will continue to face increasing competition in the future as new companies enter our market and scientific developments surrounding the pharmaceutical market continue to accelerate. We cannot predict the extent to which these developments will impact potential future sales of our products or our product candidates. There can be no assurance that our competitors are not currently developing, or will not in the future develop, technologies and products that are equally or more effective, are more economically attractive, and can be administered more easily than any of our current or future technologies or products. Such competing products or technology platforms may gain faster or greater market acceptance than our products or technology platforms. If we, our products and product candidates or our technology platforms do not compete effectively, it is likely to have a material adverse effect on our business, financial condition and results of operation. We will face significant challenges in successfully commercializing our products and additional product candidates after they are launched. The commercialization of VYVGART in new indications or other product candidates if and when approved, or entrance of any of our products or product candidates into new markets will require us to further expand our sales and marketing organization, enter into collaboration arrangements with third parties, outsource certain functions to third parties, or use some combination of each. We have built, and continue to expand, our sales force in certain of the countries where VYVGART is approved and plan to further develop our sales and marketing capabilities to promote our products, and product candidates, including new indications, if and when marketing approval has been obtained in other relevant jurisdictions. Even if we successfully expand our sales and marketing capabilities, either on our own or in collaboration with third parties, we may fail to launch or market our products effectively. Recruiting and training a specialized sales force is expensive and the costs of expanding an independent sales, marketing and/or promotion organization could be greater than we anticipate. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Risk Factors Related to Commercialization of argenx’s Products and Product Candidates, Including for New Indications 65

We could further encounter difficulties in our sales or marketing, due to regulatory actions, shut-downs, work stoppages or strikes, approval delays, withdrawals, recalls, penalties, supply disruptions, shortages or stock-outs at our facilities or third-party facilities that we rely on, reputational harm, the impact to our facilities due to natural or man-made disasters, product liability, and/or unanticipated costs. In addition, recruiting and training a sales force is time-consuming and could delay any product launch. In the event that any such launch is delayed or does not occur for any reason, we would have prematurely or unnecessarily incurred these commercialization expenses, and our investment would be lost if we cannot retain or reposition our sales and marketing personnel. Our products and product candidates for which we have obtained or intend to seek approval as biological products, including for new indications, may face biosimilar competition. In the U.S., the Biologics Price Competition and Innovation Act (BPCIA) created an abbreviated approval pathway for biological products that are demonstrated to be “biosimilar” to or interchangeable with a U.S. FDA-licensed reference biological product. However, during the 12-year regulatory exclusivity period applicable to reference biological products, another company may still market a competing version of the reference product if the FDA approves a full BLA for the competing product containing the sponsor’s own preclinical data and data from adequate and well-controlled clinical trials of their product. We believe that any of our product candidates approved as a biological product under a BLA in the U.S. should qualify for the Biologics Price Competition and Innovation Act 12-year period of exclusivity, as is the case with VYVGART. The base regulatory exclusivity period for VYVGART is expected to extend until December 2033 in the U.S. whereas regulatory protection in the EU is expected to expire in August 2032. However, in the U.S., there is a risk that this exclusivity could be shortened due to congressional action or otherwise, or that the FDA will not consider our product candidates to be reference products for competing products, potentially creating the opportunity for competition by biosimilar products sooner than anticipated. The same applies to the EU, as there is also a risk that this exclusivity could be shortened due to legislative actions. We are aware that some of our competitors may be actively developing competing or biosimilar products for VYVGART. It is possible our competitors will be successful in developing biosimilar or interchangeable products for our products and product candidates, and the approval of such competing products may lead to substantial competition in the market, a decrease in sales, or force us to make VYVGART available at lower prices due to competitive pressures. Moreover, an interchangeable biosimilar product, once approved, may be substituted under existing state laws for any one of our reference products. In addition, the Further Consolidated Appropriations Act, 2020, which incorporated the framework from the Creating and Restoring Equal Access To Equivalent Samples (CREATES) legislation, allows biosimilar developers to obtain access to reference biological products, which may facilitate the development of biosimilars to our products. The FDA has also recently issued guidance eliminating the need for data from comparative clinical efficiency studies to demonstrate biosimilarity in many circumstances, which may accelerate biosimilar market entry. If competing or biosimilar products are approved, the market position of our products for existing and recently approved indications may be adversely affected. In the EU, biosimilars are evaluated for marketing authorization pursuant to a set of general and product class-specific guidelines. Moreover, the EU’s legislative bodies are currently working toward finalizing a reform of the EU Pharmaceutical Legislation, likely shortening the baseline of market exclusivity periods for medicinal products. In addition, some EU Member States have adopted, or are considering the adoption of, biosimilar uptake measures or may impose automatic price reductions upon market entry of one or more biosimilar competitors. While the degree of competitive effects of biosimilar competition among EU Member States may vary, continuation of policies promoting biosimilar products in the EU and in EU Member States could erode market share or introduce competitive pricing pressures for our products and product candidates. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Risk Factors Related to Commercialization of argenx’s Products and Product Candidates, Including for New Indications 66

Enacted and future legislation and regulations could impact demand for our products which could impact our business and future results of operations. In the U.S. and the EU and other jurisdictions, there have been a number of legislative and regulatory changes to healthcare systems that could affect our future results of operations. Governmental regulations that mandate price controls or limitations on patient access to our products or establish prices paid by government entities or programs for our products could impact our business, and our future results of operations could be adversely affected by changes in such regulations or policies. In particular, there have been and continue to be a number of initiatives at the U.S. federal and state levels that seek to reduce healthcare costs in general and the cost of pharmaceuticals in particular, including pharmaceutical pricing reforms under the IRA. The IRA authorizes Medicare drug price negotiation, imposes inflation-based rebate obligations and significantly redesigns the Medicare Part D benefit, including establishing manufacturer discount requirements and capping beneficiary out-of-pocket costs. Although the program remains subject to legal challenges, the IRA is being implemented and may materially reduce the prices we are able to charge for our products, increase our rebate and discount obligations, and affect coverage, formulary placement and demand for our products and product candidates. See Section 1.7.4 “Regulatory Framework – Coverage, Pricing, and Reimbursement” for additional details. The HHS has and will continue to issue and update guidance and rulemaking as these IRA programs are implemented. We cannot predict how the HHS will interpret the IRA in the future, or whether the U.S. Congress will enact legislation that further amends the law. However, at this time, the Trump administration is continuing to implement the IRA. Manufacturers that fail to comply with the IRA may be subject to significant penalties, including civil monetary penalties and excise taxes. The IRA also extends enhanced subsidies for individuals purchasing health insurance coverage in ACA (as defined below) marketplaces through plan year 2025. To date, none of the legislative attempts to extend the subsidies has been enacted. While the full economic impact of IRA is unknown at this time, the law’s passage is likely to affect the pricing of our products and product candidates. The adoption of restrictive price controls in new jurisdictions, more restrictive controls in existing jurisdictions, the adoption of these lower prices by commercial payors, or the failure to obtain or maintain timely or adequate pricing could also adversely impact revenue. We expect pricing pressures will continue globally. Further, at the U.S. state level, legislatures are increasingly enacting laws and implementing regulations designed to control pharmaceutical and biological product pricing, including price or reimbursement constraints, discount requirements, price transparency reporting, and programs designed to encourage importation from other countries and bulk purchasing. States are also enacting laws modeled on federal policies, such as the IRA and the 340B drug discount program. We expect that additional state and federal healthcare reform measures will be adopted in the future, any of which could limit the amounts that federal and state governments will pay for healthcare products and services, including pharmaceuticals, which could result in reduced demand for our products and product candidates or additional pricing pressures. The EU, on the other hand, is in the final stages of adopting new EU Pharmaceutical Legislation in 2026, reforming its legislative framework for medicinal products (see “Agreement on new EU Pharmaceutical Legislation”). Depending on the final text, which has yet to be published and is still subject to formal approval, the impact could be positive with respect to certain regulatory processes. Other aspects may, however, have a negative impact on innovative pharma and biotech companies such as argenx. In particular, this may be the case for the envisaged shorter baseline market exclusivity periods. In any case, these legislative changes will only enter into effect after a (not yet specified) transitional period, which will most likely conclude in 2028 the earliest. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Risk Factors Related to Commercialization of argenx’s Products and Product Candidates, Including for New Indications 67

We are subject to government pricing laws, regulation and enforcement, which affect the prices we may charge the government for our products and the reimbursement our customers may obtain from the government. Changes in such laws, regulation, and enforcement may place downward pressure on the prices we can charge in the marketplace, and our failure to comply with these laws could harm our results, operations and/or financial conditions. In the U.S., we are required to participate in various government programs for our products to be reimbursed or purchased by the federal government. We participate in programs such as the Medicaid Drug Rebate Program, the 340B drug discount program, Medicare Part B, Medicare Part D and the U.S. Department of Veterans Affairs Federal Supply Schedule pricing program. The requirements vary by program, but we are, among other things, required to enter into agreements with and calculate and report prices and other information to certain government agencies, charge no more than statutorily mandated ceiling prices and calculate and pay rebates and refunds for certain products. The calculations are complex and are often subject to interpretation by us, governmental agencies and the courts. If we determine that the prices we reported were in error, we may be required to restate those prices and pay additional rebates or refunds to the extent we understated the rebate or overcharged the government due to the error. Additionally, there are penalties associated with submission of incorrect pricing or other data by the specified deadline, as well as potential allegations under the False Claims Act and other laws and regulations. Statutory or regulatory changes, including changes in CMS guidance, could affect the average sales price calculations and the resulting Medicare payment rate for VYVGART and our potential future products. Any such measures may increase our financial obligations to government payors, reduce net realized prices, and adversely affect the demand, coverage and overall sales of our products. In addition, maintaining compliance with these government price reporting and discounting obligations is time-consuming and costly, and a failure to comply can result in substantial fines, penalties, all of which could adversely impact our financial results. See Section 1.7.4 “Coverage, Pricing and Reimbursement” for additional details on the regulatory framework regarding the coverage, pricing and reimbursement of our products and product candidates. In addition, the current U.S. Presidential administration has taken several steps to try to align U.S. drug prices with drug prices in other countries through an approach known as MFN pricing, On November 6, 2025, CMS announced the GENEROUS Model under its CMMI authority. The GENEROUS Model is a voluntary model that tests the impact of CMS-facilitated supplemental rebate agreements that align the Medicaid net price with a defined MFN price. In December 2025, CMS issued proposed rules for the GLOBE Model and the GUARD Model under its CMMI authority. The GLOBE and GUARD Models are mandatory models that, if finalized, would require manufacturers of certain drugs to pay additional rebates based on the difference between the Medicare price and the price in market basket countries. CMS proposes that the new rebate would apply to utilization by approximately 25% of Medicare Part B fee-for-service beneficiaries and 25% of Medicare Part D enrollees. In Congress, there also are pending legislative proposals that, if enacted, would require MFN pricing in certain healthcare programs. It is also currently uncertain how these U.S. policy efforts to align US pharmaceutical pricing more closely to international benchmarks from countries with competing healthcare cost containment measures will affect our business. Any expansion, finalization or implementation of these or similar MFN-based pricing initiatives could subject our products to additional rebate obligations, negatively impact our pricing strategies, product demand, or competitive positioning across global markets, and may result in reduced revenue in critical markets. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Risk Factors Related to Commercialization of argenx’s Products and Product Candidates, Including for New Indications 68

We may not obtain or maintain adequate pricing and coverage or reimbursement status for our products and product candidates. Sales of VYVGART and our product candidates, if approved, will depend, in part, on the extent to which third-party payors, including government health programs in the U.S. (such as Medicare Parts B and D and Medicaid) and other countries, commercial health insurers, and managed care organizations, provide coverage and establish adequate reimbursement levels for such products and product candidates. Patients generally rely on third-party payors to reimburse all or part of the associated healthcare costs, and are unlikely to use our products unless coverage is provided and reimbursement is adequate to cover a significant portion of the cost of our products. In the U.S., no uniform policy of coverage and reimbursement for products exists among commercial third- party payors. Commercial third-party payors decide which products they will pay for and establish reimbursement levels, often relying upon Medicare coverage policy and payment limitations. However, decisions regarding the extent of coverage, formulary tier placement, utilization management requirements (including step therapy), and the amount of reimbursement to be provided for any product candidate that we develop through approval will be made on a plan-by-plan basis. Even under U.S. government healthcare programs such as Medicare and Medicaid, coverage and reimbursement policies can vary significantly. Medicare Part D is administered by commercial insurance companies under contract with the CMS, and their coverage and reimbursement policies may vary, subject to certain statutory and regulatory requirements. Additionally, Medicaid programs vary from state to state in their coverage policies and reimbursement rates, subject to certain federal requirements. Further, from time to time, typically on an annual basis, payment rates are updated and revised by third-party payors. Such updates could impact the demand for our products, to the extent that patients who are prescribed our products, if approved, are not separately reimbursed for the cost of the product. The process for determining whether a third-party payor will provide coverage for a product may be separate from the process for setting the price of a product or for establishing the reimbursement rate that such a payor will pay for the product. Even if we do obtain adequate levels of reimbursement, third-party payors, such as government or private healthcare insurers, carefully review and increasingly question the coverage of, and challenge the prices charged for, products. Increasingly, third-party payors are requiring that biopharmaceutical companies provide them with predetermined discounts from list prices and are challenging the prices charged for products. We may also be required to conduct expensive pharmacoeconomic studies to justify the coverage and the amount of reimbursement for particular medications. We cannot be sure that coverage and reimbursement will be available for any product that we commercialize and, if reimbursement is available, what the level of reimbursement will be. Moreover, coverage policies and third-party payor reimbursement rates may change at any time. Therefore, even if favorable coverage and reimbursement status is attained for one or more products for which we receive marketing approval in one or more indications, less favorable coverage policies and reimbursement rates may be implemented in the future. For instance, even though favorable coverage and reimbursement status has been attained for VYVGART for the treatment of gMG and CIDP in the U.S., access to VYVGART for any other indication may be reduced or restricted by limited payor coverage due to treatment criteria, which may prevent us from realizing its full commercial potential. In addition, the coverage and reimbursement levels for our products for the treatment in one indication may have an adverse impact on the coverage and reimbursement levels of such products or product candidates in other indications for which marketing approval has previously been or may subsequently be obtained. Inadequate coverage or reimbursement may diminish or prevent altogether any significant demand for our products and/or may prevent us entirely from entering certain markets or indications, which would prevent us from generating significant revenues or sustaining net profitability in the future, which would adversely affect our business, financials and results of operations. In many foreign countries, pricing, coverage, and level of reimbursement of prescription drugs are subject to governmental and/or third-party payor control, and we and our collaborators may be unable to obtain coverage, pricing, and/or reimbursement on terms that are favorable to us or necessary for us or our collaborators to successfully commercialize our marketed products in those countries. In some foreign countries, the proposed pricing for a drug must be approved before it may be lawfully marketed. The requirements governing drug pricing and reimbursement vary widely from country to country, and may take into account the clinical effectiveness, cost, and service impact of existing, new, and emerging drugs argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Risk Factors Related to Commercialization of argenx’s Products and Product Candidates, Including for New Indications 69

and treatments. For example, in the EU, pricing and reimbursement of medicinal products, is almost exclusively a matter for national, rather than EU, provisions and regulations. EU Member States may restrict the range of medicinal products for which their national health insurance systems provide reimbursement and may control the prices of medicinal products for human use. An EU Member State may approve a specific price for the medicinal product or it may instead adopt a system of direct or indirect controls on the profitability of the company placing the medicinal product on the market. Our results of operations may suffer if we or our collaborators are unable to market our products in foreign countries or if coverage and reimbursement for our marketed products in foreign countries is limited or delayed. Downward price pressures in the EU and other countries may, in turn, contribute to downward price pressure in the U.S. because of MFN initiatives. If we fail to obtain orphan drug designation or we do not have valid and enforceable patents covering our products and their uses and product candidates and fail to obtain and/or maintain orphan drug exclusivity for our products or product candidates, our competitors may be able to sell products to treat the same conditions and our revenue may be reduced. We have and may from time to time seek orphan drug designation in the U.S., Japan and, the EU for certain indications addressed by our products and product candidates. With regard to these designations or future designations we may obtain, we may not be the first to obtain marketing approval of these drugs for such indication due to the uncertainties associated with developing therapeutic products, and we may not obtain orphan exclusivity upon approval. In the U.S., orphan drug exclusivity applies only to the specific uses or indications for which a drug is approved within the designated rare disease or condition, and therefore may be narrower than the scope of the original orphan designation. In addition, exclusive marketing rights in the U.S. may be limited if we seek approval for an indication broader than the orphan-designated indication, or may be lost if the FDA later determines that the request for designation was materially defective or if we are unable to assure sufficient quantities of the product to meet the needs of patients with the rare disease or condition. Further, even if we obtain orphan drug exclusivity for a product, that exclusivity may not effectively protect the product from competition because different drugs with different active moieties or different principal molecular structural features can be approved for the same condition. Even after an orphan drug is approved, the MHRA, the EMA, respectively the European Commission, the FDA, the MHLW (collectively, the Relevant Regulatory Authorities) or other comparable regulatory authorities can subsequently approve the same drug with the same principal molecular structural features for the same condition if the regulator concludes that the later drug is safer, more effective, or makes a major contribution to patient care. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Risk Factors Related to Commercialization of argenx’s Products and Product Candidates, Including for New Indications 70

2.3 Risk Factors Related to the Development and Clinical Testing of argenx’s Products and Product Candidates Failure to successfully identify, select and develop our products in other indications, or additional products or product candidates could impair our ability to grow. Our long-term growth strategy entails developing and marketing additional products and product candidates, including efgartigimod for new indications, empasiprubart and adimanebart. This requires substantial resources, whether or not any product candidates or new indications are ultimately identified. The success of this strategy depends partly upon our ability to identify, select, develop, and ultimately, commercialize promising product candidates. We are heavily dependent on precise, accurate and reliable scientific data to identify, select and develop promising product candidates and products. Our business decisions may therefore be adversely influenced by inaccurate, improper or fraudulent scientific data, including data sourced from third parties. Even with accurate scientific data, our technology platforms may fail to discover and to generate additional products and products candidates, that are suitable for further development. Even if we identify additional product candidates, they may not be suitable for clinical development as a result of harmful side effects, limited efficacy or other characteristics that indicate that it is unlikely to be a product that will receive approval by the Relevant Regulatory Authorities, and other comparable regulatory authorities or achieve market acceptance. If we do not successfully identify, develop and commercialize product candidates and VYVGART in new indications based upon our technological approach, we may not be able to obtain product or collaboration revenues in future periods. Obtaining regulatory approval for our products and product candidates is inherently uncertain. To obtain the requisite regulatory approvals to market and sell any of our products and product candidates, we or our collaborators for such candidates must successfully demonstrate that our products are safe and effective in humans. Clinical trials are expensive and can take many years to complete, and their outcome is inherently uncertain. Further, success in early clinical trials or in one indication does not guarantee success in later clinical trials or in other indications. Failure to successfully develop or obtain marketing approval for our products and product candidates could negatively impact our business. The time required to obtain approval by the Relevant Regulatory Authorities and other comparable regulatory authorities is unpredictable but typically takes many years, if obtained at all, following the commencement of clinical trials and depends upon numerous factors, including the substantial discretion or interpretation of the regulatory authorities. This lengthy approval process as well as the unpredictability of future clinical trial results may result in our failing to obtain regulatory approval to market any of our product candidates, including for new indications. We have experienced delays in our prior clinical trials, and we may experience delays in our ongoing or planned clinical trials, for a large variety of reasons outside our control in complying with regulatory approvals which can adversely affect the timing of clinical trials, including as described in Section 2.5 “Risk Factors Related to Other Government Regulations — All aspects of our business ranging from preclinical, clinical trials, marketing and commercialization are highly regulated and any delay by relevant regulatory authorities could jeopardize our development and approval process or result in other suspensions, refusals or withdrawal of approvals.” Over the last several years, the U.S. government has shut down several times and average review times at the FDA have fluctuated in recent years as a result. If a prolonged government shutdown occurs it could significantly impact the ability of the FDA to timely review and process our regulatory submissions. Inadequate funding for the FDA and other government agencies, or other disruptions to these agencies’ operations, could also prevent new products and services from being developed or commercialized in a timely manner or otherwise prevent those agencies from performing normal functions on which the operation of our business may rely, which could negatively impact our business. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Risk Factors Related to the Development and Clinical Testing of argenx’s Products and Product Candidates 71

In addition, ongoing efforts by the current U.S. Presidential administration to limit the size of the FDA and other agencies of HHS, including through reductions in staff, may further increase the unpredictability in approval timelines for our products and product candidates. In 2025, HHS announced and implemented a restructuring that included significant reductions in the FDA’s workforce pursuant to an executive order directing federal workforce optimization. These staffing reductions together with resignations, resulting in leadership turnover and loss of institutional knowledge, may disrupt agency operations and adversely affect the timing, consistency or outcome of regulatory review. Ongoing initiatives by the current U.S. Presidential administration to deregulate and to review the scientific basis for, and otherwise attempt to influence, FDA decisions and policies create regulatory uncertainty for pharmaceutical companies. Additionally, uncertainty remains as to how the FDA’s agency-wide implementation of ELSA, a generative artificial intelligence tool, including for review of drug product applications, as well as its deployment of agentic artificial intelligence capabilities, will impact the outcomes and timeliness of FDA reviews and other activities. In addition, the future of the currently applicable Prescription Drug User Fee Act construct to ensure timely FDA review of applications may be impacted due to expressed concerns about the effect on industry-FDA relations, and staffing shortages. If we are unable to obtain regulatory approval of our products and product candidates on a timely basis or at all, our business, financial operations and/or financial condition may be impacted. Certain of our clinical trials have not succeeded, and may in the future also not succeed, and even if they succeed, we may not obtain regulatory approval for our products or product candidates or regulatory approval may be delayed. Certain of our clinical trials have not succeeded, and may in the future also not succeed. We could experience operational challenges as we undertake an increasing number of clinical trials, including those conducted in countries outside the EU, UK and the U.S. that may subject us to further delays and expenses as a result of increased shipment costs, additional regulatory requirements and the engagement of non-EU, non-UK and non-U.S. contract research organizations (CROs). Such trials may also expose us to risks associated with clinical investigators and institutions who apply different standards of diagnosis, screening and medical care or are otherwise unfamiliar with standards and requirements imposed by the Relevant Regulatory Authorities. If we experience delays in the completion of, or termination of, any clinical trial of our products or product candidates, may increase our costs, slow down our product candidate development and approval process and jeopardize our ability to commence product sales and generate revenues. Many of the factors that cause, or lead to, a delay in the commencement or completion of clinical trials may also ultimately lead to the denial of regulatory approval of our product candidates or result in the development of our product candidates being stopped early. Significant clinical trial delays could also allow our competitors to bring products to market before we do or shorten any periods during which we have the exclusive right to commercialize our products and product candidates. Even if clinical trials are initiated, our development efforts may not be successful. Even if we obtain positive results from preclinical trials or initial clinical trials, we may not achieve the same success in future clinical trials, which may negatively impact the price of our ordinary shares or ADSs. Regulatory approval of our products or product candidates may be delayed or refused for many reasons, including for reasons outside our control. Some of the reasons for regulatory delay or refusal include: • the Relevant Regulatory Authorities or other comparable regulatory authorities may disagree with the design or implementation of our clinical trials; • we may be unable to demonstrate, to the satisfaction of the Relevant Regulatory Authorities or other comparable regulatory authorities, that our product candidates are safe, pure, potent and effective for any of their proposed indications; • the results of clinical trials may not meet the level of statistical significance required by the Relevant Regulatory Authorities or other comparable regulatory authorities for approval; • the chemistry, manufacturing and controls information submitted in an application is insufficient; and argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Risk Factors Related to the Development and Clinical Testing of argenx’s Products and Product Candidates 72

• the facilities of third-party manufacturers with which we contract for the manufacture of our product candidates are not adequate to support approval of our product candidates. Any of these occurrences may harm our business, results of operations and financial condition significantly. If we decide to pursue accelerated approval for any of our product candidates, it may not lead to faster development or regulatory review or approval and we may still need to conduct additional clinical trials, which could increase the expense of obtaining, if at all, necessary marketing approvals. The accelerated approval pathway has come under scrutiny by various stakeholders, and the Food and Drug Omnibus Reform Act of 2022 (FDORA) revised the requirements for this pathway. Although this legislation did not change the standard for accelerated approval, it authorized the FDA to require a post- approval clinical trial to be underway prior to approval or within a specified time period following approval, and must specify conditions of any required post-approval clinical trial. FDORA also requires sponsors to submit progress reports for required post-approval studies. Failure to conduct due diligence for required post-approval studies is deemed a prohibited act under the FDCA. FDORA also details procedures the FDA must follow to withdraw an accelerated approval on an expedited basis, including where the required post- approval studies are not conducted with due diligence or fail to verify clinical benefit, other evidence demonstrates that the product is not shown to be safe or effective under the conditions of use, or the sponsor disseminates false or misleading promotional materials with respect to the product. FDA has been exercising these authorities and has issued guidance documents regarding the accelerated approval pathway. If we decide to pursue accelerated approval for any of our product candidates, the failure to obtain accelerated approval (or the withdrawal of any accelerated approval) could result in a longer time period to commercialization of such product candidate, if any, and could increase the cost of development of such product candidate and harm our competitive position in the marketplace. For example, if standard of care were to evolve or if any of our competitors were to receive approval for a drug or biological product for a disease or condition for which we are seeking accelerated approval before we receive accelerated approval, we may not be able to demonstrate that our product candidate provides a meaningful advantage over other available therapies and accelerated approval may not occur. Our products and product candidates may have adverse, undesirable or unacceptable side effects, and we or others may identify undesirable or unacceptable side effects caused by any of our products or product candidates before and after they have received marketing approval. Undesirable side effects that may be caused by our product candidates, or by the combination of our product candidates with other medical products, could cause us or regulatory authorities to interrupt, delay or halt clinical trials and could result in more restrictive labeling or the delay or denial of regulatory approval by the Relevant Regulatory Authorities or other comparable regulatory authorities. We have observed adverse events and treatment emergent adverse events in our clinical trials, and we may see additional adverse events and treatment emergent adverse events in our ongoing and future clinical trials. Such side effects may be more serious than those observed to date, and as a result, our ongoing and future clinical trials may be negatively impacted. Moreover, as we seek to develop product candidates, including products in new indications, patients may experience new or more serious effects. Drug-related side effects caused by any of our products or product candidates that we or others identify could, among other things, affect patient recruitment, the ability of enrolled patients to complete the clinical trial, result in potential product liability claims, damage sales of our existing products, result in significant reputational damage for us and our product development, and other issues including the delay of other programs. Any undesirable drug-related side effects can also cause the Relevant Regulatory Authorities or other comparable regulatory authorities to withdraw approvals or revoke licenses of such products and require us to take such products off the market, require the addition of labeling statements, specific warnings, or a contraindication or other modification of the product labeling, request the issuance of safety alerts, require a REMS to ensure that the benefits of the product outweigh its risks, and/or require us to change the way the product is administered, conduct additional clinical trials or change the labeling of the product. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Risk Factors Related to the Development and Clinical Testing of argenx’s Products and Product Candidates 73

If our target patient population is smaller than expected, we are unable to successfully enroll and retain patients in our clinical trials, or experience significant delays in doing so, we may not realize the full commercial potential of any products or product candidates. Currently, we mainly develop products or product candidates for the treatment of rare diseases for which the target patient population can be small. If the actual number of patients with these disorders is smaller than we expected, we may encounter difficulties in enrolling sufficient patients in our clinical trials, thereby delaying or preventing development and approval of our products or product candidates. Physicians, who are an important source of patients for clinical trials, may also be less familiar with these rare diseases and may therefore fail to identify these conditions in their patients and therefore may not refer them to our clinical trials. Patient enrollment, a significant factor in the timing of clinical trials, depends on many factors, including the size and nature of the patient population, eligibility criteria for the clinical trial, the proximity of patients to clinical sites, competition for patient recruitment from competing clinical trials, the design of the clinical trial, the availability of alternate approved therapies for the indication the clinical trial is investigating, and clinicians’ and patients’ perceptions as to the potential advantages of the drug being studied in relation to other available therapies. We compete with other companies to enroll target patient populations, as set forth in Section 2.1 “Risk Factors Related to Commercialization of argenx’s Products and Product Candidates, Including for New Indications—We face significant competition for our drug discovery and development efforts.” Even if product candidates obtain significant market share for their approved indications, because certain potential target populations are small, we may never recoup our investment in such product candidate without obtaining regulatory approval for additional indications for such product candidates. Even once enrolled, we may be unable to retain a sufficient number of patients to complete any of our clinical trials. In addition, any negative results we may report in clinical trials of our drug candidates may make it difficult or impossible to recruit and retain patients in other clinical trials of that same drug candidate. Delays in the completion of any clinical trial of our product candidates will increase our costs, slow down our product candidate development and approval process and delay or potentially jeopardize our ability to commence product sales and generate revenue. In addition, some of the factors that cause, or lead to, a delay in the commencement or completion of clinical trials may also ultimately lead to the denial of regulatory approval of our product candidates. 2.4 Risk Factors Related to argenx’s Dependence on Third Parties We rely, and expect to continue to rely, on third parties to conduct some of our research activities, manufacturing and clinical trials and for parts of the development and commercialization of our existing and future research programs, products and product candidates. If our relationships with such third parties are not successful, our business may be adversely affected. We have relied upon and plan to continue to rely upon third parties, including independent clinical investigators, CROs, CMOs and other third-party service providers with the applicable protocol, legal and regulatory requirements and scientific standards, and our reliance on these third parties does not relieve us of our regulatory responsibilities. To the extent our collaborators or the CROs or investigators fail to enroll participants for our clinical trials, fail to conduct the clinical trial to GCP standards or in full compliance with legal and regulatory requirements or are delayed for a significant time in the execution of clinical trials, including achieving full enrollment, we may be affected by increased costs, program delays or both, which may harm our business. In addition, we are, and expect to continue to be, dependent on partnerships with partners and licensees relating to the development and commercialization of our existing and future research programs, products and product candidates. We currently have collaborative research relationships with various argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Risk Factors Related to the Development and Clinical Testing of argenx’s Products and Product Candidates 74

pharmaceutical companies such as AbbVie, Zai Lab and with various academic and research institutions worldwide for the development of product candidates resulting from such collaborations. We also have distribution agreements in place with several distribution partners for VYVGART. We have and will continue to have discussions on potential partnering opportunities with various pharmaceutical companies. If we fail to enter into or maintain collaborations on reasonable terms or at all, our ability to develop our existing or future research programs and product candidates and to commercialize our existing or future products could be delayed, the commercial potential of our products could change and our costs of development and commercialization could increase. While we have agreements governing our relationships with these third parties, we have limited influence over their actual performance and control only certain aspects of their activities. If independent investigators, third-party service providers or CROs fail to devote sufficient resources to the development of our product candidates, or if their performance is substandard, it may delay or compromise the prospects for approval and commercialization of any product candidates that we develop. In addition, our collaborators, CROs, CMOs, distributors, and other third-party service providers are subject to extensive healthcare, regulatory, data privacy, manufacturing, anti-corruption and other laws and regulations. Although we seek to structure our relationships in compliance with applicable laws and regulations and monitor the activities of these third parties, we do not control their day-to-day operations and cannot guarantee that they will comply with all applicable legal and regulatory requirements. Any failure by our third-party partners to comply with such requirements could result in regulatory enforcement actions, monetary penalties, exclusion from government healthcare programs, reputational harm, delays in development or commercialization, or other adverse consequences. In particular, regulatory authorities enforce GCP requirements through periodic inspections of clinical trial sponsors, principal investigators and clinical trial sites. If we, our investigators or any of our CROs fail to comply with applicable GCPs, the clinical data generated in our clinical trials may be deemed unreliable and the Relevant Regulatory Authorities or comparable regulatory authorities may require us to perform additional clinical trials before approving our marketing applications. In addition, our collaborative partners may not adhere to, or may terminate collaboration agreements with, all associated consequences or disagree on the interpretation of contractual terms. We may not be able to control our collaborative partners’ compliance with all applicable requirements for the commercialization of our products, which could adversely affect such commercialization and the profitability of such products. Failures by our collaborative partners to meet their contractual, regulatory, or other obligations to us, or any disruption in the relationships between us and our collaborative partners, could have a material adverse effect on our product pipeline and business. We face significant competition in establishing successful relationships with third-party service providers and appropriate collaborative partners. These third-party service providers may have contractual relationships with other entities, some of which may be our competitors, which may draw their time and resources away from our programs. In addition, some of our third-party service providers or CROs have the ability to terminate their respective agreements with us, and if such agreements terminate, we may not be able to enter into arrangements with alternative CROs or investigators or to do so on commercially reasonable terms. In addition, we may not be able to find appropriate collaboration partners. Disruptions caused by our reliance on third parties for our raw materials and manufacturing process may delay or disrupt our business, product development and commercialization efforts. We do not have the ability to internally source the raw materials necessary to produce our products or product candidates, and do not currently have, nor do we plan to acquire, the infrastructure or capability internally to manufacture our products or product candidates and depend on a worldwide supply chain and third parties for both. Disruptions caused by our reliance on such third-party suppliers, service providers and manufacturers may delay or disrupt our business, product development and commercialization efforts. Reliance on Third-Party Suppliers and Service Providers For some of our raw materials, we rely on a single source of supply and there are limited supplies of the raw materials. If prices increased, or we were to experience an unexpected loss of supply of or if any supplier was unable to meet our demand for any of our products and product candidates, including increased demand if VYVGART is approved for additional indications, we could experience delays in our research or planned clinical trials or risk shortages in commercial supply which could materially impact our argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Risk Factors Related to argenx’s Dependence on Third Parties 75

revenue potential. These issues could be exacerbated by pressure on the supply chain, for example due to power shortages, natural disasters, extreme weather conditions, public health crises, changed laws or regulations, military conflicts, executive orders, or geopolitical events, including trade disputes, embargoes or economic or financial sanctions enacted as a result of international conflict. The cost of our raw materials may also increase based on actual or threatened trade restrictions or increased tariffs on foreign exports. As we continue to grow our business we may need to establish additional sources of supply for our products. The lead time needed to establish a relationship with a new supplier can be lengthy and require us to devote substantial time and resources, which could result in additional costs, or delays and adversely affect our business. Additionally, certain of the raw materials required in the manufacture and the formulation of our products and product candidates may be derived from biological sources, including mammalian tissues, bovine serum and human serum albumin. There are certain European regulatory restrictions on using these biological source materials including rigorous testing requirements, which could limit or delay production. Regulatory authorities may require additional studies if we adopt a new supplier. If there are changes in the regulation requirements that our suppliers are unable to meet, our clinical development or commercial activities may be delayed or interrupted. We may not be able to engage a back-up or alternative supplier or service provider in a timely manner or at all if any of these third parties were to cease or interrupt production or otherwise fail to supply these materials, products, or services to us for any reasons, including due to regulatory requirements or actions (including recalls), adverse financial developments at or affecting the supplier, failure by the supplier to comply with cGMPs, contamination, business interruptions, or labor shortages or disputes. Interruptions in the supply of these materials, products or services may also result from international conflict, trade disputes, embargoes or economic or financial sanctions imposed, administered or enforced by the UN Security Council, the U.S., the UK, the EU, or the respective governmental institutions of any of the foregoing including, without limitation, the Office of Foreign Assets Control of the US Department of the Treasury, the US Department of Commerce, the US Department of State and any other agency of the US government. Reliance on Third-Party Manufacturing We rely on and expect to continue to rely on CMOs. We also rely on certain third parties to perform filling, finishing, distribution, laboratory testing and other services related to the manufacture and supply of our products and product candidates. We do not control the manufacturing process at our CMOs and are completely dependent on them for the production of our products and product candidates in accordance with relevant regulations (such as cGMPs), although we are responsible for ensuring that our products comply with regulatory requirements. If our CMOs cannot successfully manufacture material that conforms to our specifications and the strict regulatory requirements of the Relevant Regulatory Authorities or other comparable regulatory authorities, our business could be adversely affected, including an inability to initiate or continue clinical trials of product candidates under development, delay in submitting regulatory applications, or receiving regulatory approvals for product candidates, including new indications, subjecting third-party manufacturing facilities to additional inspections by regulatory authorities, requirements to cease distribution or to recall batches of our products or product candidates and an inability to meet commercial demands for our marketed products. Most notably, we contract with Lonza for their manufacturing sites in Slough, UK, Portsmouth, U.S., Singapore and Visp, Switzerland as well as with Fujifilm, based in Denmark, for activities relating to the development of cell banks, development of our manufacturing processes and the manufacturing of drug substance. We use additional contract manufacturers to fill, test, label, package, store and distribute our (investigational) drug products. Our products and product candidates are biologics and require multiple processing steps that are more difficult than those required for most small molecule chemical pharmaceuticals. While we work with our CMOs and partners on optimization, strengthening and upscaling our manufacturing, problems with these manufacturing processes, such as capacity issues, or even minor deviations from the normal process or from the materials used in the manufacturing process, which may not be detectable by us in a timely manner, could lead to manufacturing failures or product defects, resulting in lot failures, product recalls, product liability claims and insufficient inventory. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Risk Factors Related to argenx’s Dependence on Third Parties 76

We face risks inherent in relying on limited CMOs, as any failure in their ability to successfully manufacture our products or product candidates as described above or any disruption, such as supply shortages or disruptions of raw materials, fires, pandemics, natural hazards or acts of vandalism at the CMO could significantly interrupt our manufacturing capability. Alternative production plans in place or disaster- recovery facilities available to us may not be sufficient. In case of a disruption, we may have to establish additional alternative manufacturing sources. This would require substantial investment on our part, which we may not be able to obtain on commercially acceptable terms or at all. Additionally, we may experience significant manufacturing delays as we build or locate replacement facilities and seek and obtain necessary regulatory approvals. If this occurs, we will be unable to satisfy manufacturing needs on a timely basis, if at all. Also, operating any new facilities may be more expensive than operating at our current facilities. Further, business interruption insurance may not adequately compensate us for any losses that may occur, and we would have to bear the additional cost of any disruption. For these reasons, a significant disruptive event of the manufacturing facility could have drastic consequences, including placing our financial stability at risk. Accuracy and timing of our financial reporting is partially dependent on information received from third-party partners, which we do not control. We have collaborated, and plan to continue to collaborate, with third parties, including distributor and licensing partners, on certain product candidates. As part of some of these collaborations, our collaboration partners are responsible for providing us with financial information regarding specific projects, including funds spent, liabilities incurred and expected future costs, on which we rely for our own financial reporting. If our collaboration partners fail to provide us with the necessary financial information within the agreed upon timeframes, or if such financial information proves inaccurate, it would adversely impact the timing and accuracy of our own financial reporting. Any inaccuracy in our financial reporting could cause investors to lose confidence in our financial reporting, reputational damage or affect our ability to obtain, and the terms of, any future financing, which may harm our business. 2.5 Risk Factors Related to Other Government Regulations We are subject to healthcare laws, regulation and enforcement. The failure to comply with these laws could harm our results, operations and/or financial condition. Our current and future operations are and may become directly, or indirectly through our customers and third-party payors, subject to various U.S. federal and state, EU and EU Member State, Japanese, Chinese, UK, Canadian and Israeli healthcare laws, and healthcare laws of other jurisdictions in which we conduct our business. This includes, but is not limited to, the U.S. FDCA, the U.S. False Claims Act and EU Directive 2001/83/EC. In particular, our sales, marketing and business arrangements are subject to healthcare fraud and abuse, anti-kickback and similar laws designed to prevent fraud, misconduct, improper inducements and other abusive practices. For example, EU Directive 2001/83/EC restricts the provision of gifts, pecuniary advantages or other benefits to persons qualified to prescribe or supply medicinal products, subject to limited exceptions. Compliance with these complex and evolving requirements requires significant resources and may constrain our commercial activities. Any action against us for violation of these laws, even if we successfully defend against it, could cause us to incur significant legal expenses and divert our management’s attention from the operation of our business. The shifting compliance environment and the need to maintain robust and expandable systems to comply with multiple jurisdictions with different compliance or reporting requirements increases the possibility that we or our collaborative partners may run afoul of one or more of these requirements. We continue to expand, enhance and refine our internal ethics and compliance function and program to ensure compliance with the different healthcare laws and regulations. As we continue to grow our headcount to argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Risk Factors Related to argenx’s Dependence on Third Parties 77

support our business, we face increased compliance risk as we need to train and supervise additional personnel to comply with relevant healthcare laws and regulations. This involves substantial costs and, notwithstanding our investment, there can be no assurance that our policies and procedures will be followed at all times or will effectively detect and/or prevent all compliance violations by our employees, consultants, subcontractors, agents and partners. It is possible that governmental authorities will conclude that our business practices do not comply with current or future statutes, regulations or case law involving applicable fraud and abuse or other healthcare laws and regulations. If our operations are found to be in violation of any of these laws or any other governmental regulations applicable to us, we may be subject to significant civil, criminal and administrative investigations, penalties, damages, fines, disgorgement, imprisonment, exclusion of drugs from government funded healthcare programs, such as Medicare and Medicaid in the U.S., additional reporting requirements and oversight, reputational harm and the curtailment or restructuring of our operations. Managing such investigations and defending against or appealing any such actions or penalties can be costly and time-consuming and may require significant financial and personnel resources. Therefore, even if we are successful in managing any such governmental investigations and/or defending against or appealing any such actions or penalties that may be brought against or imposed upon us, our business may be impaired. Efforts to ensure that our business arrangements with third parties comply with applicable healthcare laws and regulations also involve substantial costs, and because we do not fully control the operations or compliance practices of these third parties, we cannot assure you that they will comply with all applicable healthcare laws and regulatory requirements. The scope, interpretation and enforcement of healthcare laws remain uncertain and subject to change, particularly in the current environment of healthcare reform. Federal and state authorities in the U.S. have increased scrutiny of interactions between healthcare companies and healthcare providers, as well as promotional practices, including direct-to-consumer prescription drug advertising and manufacturer- sponsored platforms that facilitate patient access to products. The FDA has also heightened its review of the data supporting advertising and promotional claims. Compliance with these evolving requirements, and responding to investigations or enforcement actions, may require significant time and resources and could divert management attention, result in penalties or otherwise adversely affect our business. All aspects of our business, including preclinical research, clinical trials, marketing and commercialization, are highly regulated, and any delay by relevant regulatory authorities could jeopardize our development and approval process and/or result in suspensions of marketing authorizations, refusals to approve our products, or withdrawal of existing approvals. Before we can commence clinical trials for a product candidate, we must complete extensive preclinical testing to support our IND or planned IND applications in the U.S. or Japan, or our clinical trial applications (CTAs) in the EU, or comparable applications in other jurisdictions. We cannot be sure that we will be able to submit INDs or CTAs or comparable applications for our development programs on the timelines we expect, if at all. We also cannot guarantee that submission of INDs or CTAs or comparable applications will result in the Relevant Regulatory Authorities or other regulatory authorities allowing clinical trials to begin. Clinical trials must be conducted in accordance with applicable laws and regulations, Relevant Regulatory Authorities' and other comparable regulatory authorities’ legal requirements and regulations and are subject to oversight by Relevant Regulatory Authorities and other comparable regulatory authorities as well as IRBs and ethics committees. In particular, clinical trials must be conducted in compliance with GCPs and clinical supplies of our products and product candidates must be produced under cGMPs and other regulations. We could encounter delays if a clinical trial is suspended or terminated, by us, by the IRB or ethics committee, by the data review committee or data safety monitoring board for such clinical trial, or by the Relevant Regulatory Authorities or other comparable regulatory authorities. Such authorities may impose a suspension or termination due to a number of factors, including failure to conduct the clinical trial in accordance with regulatory requirements or our clinical protocols, inspection of the clinical trial operations or clinical trial site by the Relevant Regulatory Authorities or other comparable regulatory authorities resulting in the imposition of a clinical hold, unforeseen safety issues or adverse side effects, including those relating to the class to which our products and product candidates belong, failure to demonstrate a benefit from using the product or product candidate, changes in governmental regulations or administrative actions, or lack of adequate funding to continue the clinical trial. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Risk Factors Related to Other Government Regulations 78

If we experience delays in the completion of, or termination of, any clinical trial of our products or product candidates, the costs of our clinical programs may increase, the commercial prospects of our products and product candidates may be harmed, and our ability to generate product revenues from any of these products and product candidates may be delayed. Significant clinical trial delays could also allow our competitors to bring products to market before we do, or shorten any periods during which we have the exclusive right to commercialize our products and product candidates. Moreover, we must obtain separate regulatory approvals in each jurisdiction where we want to market, and approval by one regulatory authority does not ensure approval by any other regulatory authority. As approval procedures can vary among countries and may change over time, this can require additional clinical testing, and the time required to obtain approval may differ. We can provide no assurances that such approval will be obtained on the timeline that we expect or at all. In addition, we anticipate submitting applications for approval of VYVGART in new indications, but can provide no assurances that such applications will be accepted or that we will receive approval on our anticipated timeline, or at all. If VYVGART or any new formulations of VYVGART are not approved in one or more jurisdictions including beyond the countries where VYVGART is approved, or if such approvals are significantly delayed, it could have a material adverse effect on our business. It is possible that none of our other existing product candidates or any product candidates we may seek to develop in the future will ever obtain regulatory approval in any other jurisdiction for any indication. Even if approval is obtained, the Relevant Regulatory Authorities or other comparable regulatory authorities may approve the product for fewer or more limited indications or patient sub-segments than requested and/or with a label that does not include the labeling claims necessary or desirable for the successful commercialization of that product. Further, the Relevant Regulatory Authorities or other comparable regulatory authorities may impose extensive and ongoing unique regulatory requirements, such as granting approval contingent on the performance of costly post-marketing clinical trials, including Phase 4 clinical trials, and surveillance to monitor the safety and efficacy of the product. The costs of compliance with all Relevant Regulatory Authorities' and other applicable authorities' regulations, requirements or guidelines could be substantial, and failure to comply could result in sanctions, including fines, injunctions, civil penalties, denial of applications for marketing authorization of our products, delays, suspension or withdrawal of approvals, license revocation, seizures or recalls of products, operating restrictions and criminal prosecutions, any of which could significantly increase our and/or our collaborative partners’ costs or delay or prevent the development and commercialization of our product candidates. At this time, we cannot guarantee or know the exact nature, precise timing and detailed costs of the efforts that will be necessary to complete the remainder of the development of our research programs and product candidates. We are subject to privacy, cybersecurity and AI laws, regulation and potential enforcement. The failure to comply with these laws could harm our results, operations and/or financial conditions. Privacy laws, regulations and related enforcement are particularly relevant to our business as we collect, store and process personal data and in particular data of vulnerable groups such as patients, and sensitive information including health data as well as human biological samples. We also collaborate with third parties where we may seek to use data collected by third parties on our or their behalf, or we may seek to share data collected by us with such third parties to further our research or commercial initiatives. The GDPR imposes strict requirements, including access controls, impact assessment and safeguards on cross-border transfers of personal data and imposes substantial penalties in the event of non-compliance. We face uncertainty as to the exact interpretation of the requirements under the GDPR, and we may be unsuccessful in implementing all measures required by data protection authorities or courts. Any investigation by a data protection authority could result in fines and other penalties. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Risk Factors Related to Other Government Regulations 79

The data privacy landscape is complex and fragmented. In the EU, national laws may impose stricter obligations than the GDPR, particularly for sensitive genetic, ethnic origin or race data, while regulations like the Directive 2002/58/EC of the European Parliament and of the Council of July 12, 2002 (as amended, the e-Privacy Directive) add further compliance risks. This trend expands globally, with evolving laws in countries such as the U.S., Japan, Canada and China. For example, China has passed a number of laws concerning data protection and the collection, use and transfer of personal data (including data considered to be relevant as Chinese human genetic resources), and restricting the transfer of this data outside of China. These evolving laws and regulations impose increasing restrictions on the processing of personal data, which may require us to modify our data collection or processing practices and to incur significant expenses associated with our compliance efforts. Moreover, in the current digital and regulatory landscape, privacy obligations are increasingly interconnected with broader cybersecurity laws and requirements, reflecting the close link between the protection of personal data and the security of information systems. Failure to comply with applicable cybersecurity frameworks and directives, including Directive (EU) 2022/2555 on Network and Information Security (NIS2), could result in significant legal, regulatory, and operational risks. In addition, inadequate cybersecurity and non-compliance with data protection laws and regulations increases the risk of personal data breaches, potentially resulting in regulatory sanctions, civil claims, reputational damage and loss of trust. Furthermore, our integration of AI solutions into certain of aspects our business, introduce a new layer of regulatory risk. Most significantly, Regulation (EU) 2024/1689 (AI Act) establishes the world’s first comprehensive legal framework for AI, impacting how we develop and deploy AI systems in the future. In particular, high-risk AI systems must comply with strict regulatory requirements to ensure safety, transparency, and fundamental rights protection. Non-compliance with the AI Act may result in significant penalties. As most provisions will take effect from August 2, 2026, we must continuously assess and adapt our compliance strategy to mitigate risks from this evolving legal landscape. If we violate existing laws and regulations or fail to comply with changing laws and regulations, we might be subject to fines, penalties and other adverse consequences, which could have a material adverse effect on our reputation, business, results of operations, cash flows or financial condition. Failure to comply with anti-corruption laws and regulations, anti-money laundering laws and regulations, economic or financial sanctions, trade embargoes and/or export control regulations and other laws governing our operations could have an adverse impact on our business, financial conditions and operations. We are or may become subject to various laws and regulations regarding anti-corruption, anti-money laundering, economic or financial sanctions, trade embargoes, investment restrictions, anti-fraud, other comprehensive prohibitions against transaction activity pursuant to anti-terrorism laws or export control laws and regulations issued by multiple jurisdictions. These include the UK Bribery Act 2010 and the U.S. Foreign Corrupt Practices Act of 1977, in each case, as amended, as well as comparable laws and regulations in other countries in which we do business, including in the European Union and China, which prohibit, among other things, payments, offers, or promises made for the purpose of improperly influencing any act or decision of a foreign government official. In the UK, since September 1, 2025, it is an offense under the Economic Crime and Corporate Transparency Act 2023 for a large organization to fail to prevent certain fraudulent activities by an associated person (such as an employee, agent, or subsidiary), unless it can demonstrate that it had reasonable prevention procedures in place to prevent the fraudulent activity. The nature of our business means that we engage in significant interactions with foreign officials. Compliance with these laws and regulations in the U.S. and in foreign jurisdictions is complex, and may increase our cost of doing business internationally. We are also subject to economic and financial sanctions, trade embargoes, other comprehensive prohibitions against transaction activity pursuant to anti-terrorism laws and export control rules and regulations, including those imposed, administered or enforced by the UN Security Council, the, U.S., the UK, and the EU. Any change in export or import regulations, economic or financial sanctions regulations, trade embargo regulations or related legislation, shift in the enforcement or scope of existing regulations, argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Risk Factors Related to Other Government Regulations 80

or change in the countries, governments, persons or technologies targeted by such regulations, could decrease our ability to conduct our planned research and development activities and to manufacture, import, export or sell our products internationally, which could require us to expend additional resources to achieve our goals and adversely affect our business, financial conditions and operations. We have mechanisms in place to promote compliance with such rules and regulations. However, there can be no assurance that our policies and procedures will be followed at all times or will effectively detect and/ or prevent violations of applicable compliance regimes by our employees, consultants, sub-contractors, agents and partners. In the event of non-compliance, we could be subject to substantial civil or criminal penalties, including sanctions against us, incarceration for responsible employees and managers, the possible loss of export or import privileges, debarment from participation in government contracting, reputational harm, and resulting loss of revenue and profits, which could have a material adverse impact on our business, financial conditions and operations. Our performance tracked by our Environmental, Social and Governance metrics is subject to risks and the outcomes may not achieve the anticipated benefits or align with new regulations and stakeholders’ expectations. There has been an increasing focus from stakeholders and regulators relating to environmental, social and governance (ESG) matters across all industries in recent years. The standards and stakeholder expectations continue to evolve, sometimes with contradictory expectations, and criteria to evaluate ESG practices may change rapidly. We are subject to evolving rules, including the European Union’s Corporate Sustainability Reporting Directive (CSRD) and ancillary European Union legislation. The SEC adopted rules in 2024 requiring enhanced climate-related disclosures, but after legal challenges, subsequently announced that it would end its defense of such climate disclosure rule. As a result, various U.S. states have enacted or proposed climate- and sustainability-related disclosure laws that may apply to companies doing business in those jurisdictions, such as California’s climate and carbon market disclosure laws, which would require in- scope companies to report on greenhouse gas emissions, climate-related financial risks, and the use of carbon offsets and emissions reduction claims relating to their operations or products. The future of the California climate disclosure laws is uncertain, as these laws are subject to ongoing litigation. In response to new ESG initiatives and regulations we may voluntarily elect, or be required, to adopt strategies, policies, or procedures related to ESG matters. Such efforts could divert management’s attention from central operational matters and cause us to expend significant capital and human resources. Moreover, increasingly, different stakeholder groups and regulators have divergent views, particularly in the U.S., on ESG matters, which increases the risk that any action or lack thereof with respect to sustainability or ESG matters will be perceived negatively by at least some stakeholders and regulators and adversely impact our business and reputation. The current sociopolitical landscape has led to rapid and unpredictable shifts in public sentiment, which has resulted in dynamics that increase the risk of reputational damage, boycotts and shifts in consumer behavior that could adversely affect our business and reputation. Reports could also lead to the disclosure of information that may have a negative impact on our operations and reputation which may lead to additional exposure. In addition, any required disclosures and measurements of ESG metrics are highly dependent on third-parties, such as our suppliers and CROs, that we do not control. Failure to accurately comply with any sustainability reporting obligations may result in enforcement actions, sanctions, fines and penalties, reputational harm or private litigation. We may become exposed to liability and substantial expenses in connection with environmental compliance or remediation activities. Our operations, including our research, development and testing, and our third-party manufacturers’ and suppliers’ operations, are subject to numerous environmental, health and safety laws and regulations and for which we may become liable. These laws and regulations govern, among other things, the controlled use, handling, release and disposal of and the maintenance of a registry for, hazardous materials and biological materials, laboratory procedures and exposure to pathogens. We do not have control over our manufacturers’ or suppliers’ compliance with environmental, health and safety laws and regulations. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Risk Factors Related to Other Government Regulations 81

If we or one of our CMOs or third-party distributors, manufacturers, suppliers, licensees or co-marketers fail to comply with such laws and regulations, such failure could result in substantial liability, fines, penalties or other sanctions and incur substantial expenses, and could also face significant reputational loss. We face a risk of environmental liability inherent in our current and historical activities, including liability relating to releases of our exposure to hazardous or biological materials. Furthermore, environmental, health and safety laws and regulations are becoming more stringent. Both us and our third-party manufacturers and suppliers may be required to incur substantial expenses in connection with future environmental compliance or remediation activities, in which case, our production and development efforts may be interrupted or delayed, and our financial condition and results of operations may be materially adversely affected. 2.6 Risk Factors Related to argenx’s Financial Position We may not be able to be profitable or sustain net profitability in the future and may require additional financing to fund our operations. To be profitable or sustain net profitability in the future, we must continue to succeed in commercializing products that generate significant product net sales. Our future results of operations and profitability may fluctuate from period to period, and we will need to generate significant revenues to be profitable or sustain net profitability in the future. We may not be able to generate these revenues, and we may never achieve profitability on a sustained basis in the future. If we do not succeed in sustaining profitability or in funding our operations, we would not be able to use deferred tax assets against taxable profits which would result in a de-recognition of our deferred tax asset balance. In addition, we intend to continue to conduct research and development, preclinical testing, clinical trials and regulatory compliance activities as well as the continued commercialization of VYVGART and other products candidates, for current and future indications, and we intend to continue our efforts to expand our sales, marketing and distribution infrastructure. As a result, we anticipate that our operating expenses will increase as we execute on our strategic objectives and could increase more significantly if we experience delays or encounter issues relating thereto, including failed clinical trials, ambiguous clinical trial results, safety issues or other regulatory challenges. To finance our operations, particularly if we are unable to generate sufficient product net sales or otherwise control expenses, we may need to raise additional capital through a combination of public or private equity or debt financings or other sources, which may include collaborations with third parties. Our ability to raise additional funds on acceptable terms or at all will depend on financial, economic and market conditions and other factors, over which we may have no or limited control. If we cannot raise additional capital when needed on acceptable terms, we may be required to delay, reduce or terminate research and development programs; defer or forego commercialization of our products and product candidates (including for new indications); and limit expansion or otherwise fail to capitalize on business opportunities, any of which could have a material adverse effect on our business, financial condition and results of operations. Our assets, earnings and cash flows and the investment of our cash and cash equivalents may be subject to risks which may cause losses and affect the liquidity of these investments. We invest our cash in accordance with an established internal investment policy. Currently, substantially all of our available cash and cash equivalents and current financial assets are invested in either current accounts, savings accounts, term accounts or highly liquid money market funds. Any future investments may include term deposits, corporate bonds, commercial paper, certificates of deposit, government securities and money market funds in accordance with our cash investment policy. These investments may be subject to general credit, liquidity, market, inflation, foreign currency and interest rate risks and we may realize losses in the fair value of these investments or a complete loss of these investments. The argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Risk Factors Related to Other Government Regulations 82

aforementioned risks associated with our cash flows and investment portfolio may adversely affect our results of operations, liquidity and financial condition. Due to the international scope of our operations, our assets, earnings and cash flows are influenced by movements in exchange rates of several currencies, particularly the euro and Japanese Yen, versus the U.S. dollar. Our revenue from outside of the U.S. will increase as our products, whether commercialized by us or our business partners or our collaborators gain marketing approval in such jurisdictions. If the U.S. dollar weakens against a specific foreign currency, our revenues will increase, having a positive impact on net income, but our overall expenses will increase, having a negative impact on expenses. Conversely, if the U.S. dollar strengthens against a specific foreign currency, our revenues will decrease, having a negative impact on net income, but our overall expenses will decrease, having a positive impact on expenses. Continued volatility in foreign exchange rates is likely to impact our operating results and financial condition. 2.7 Risk Factors Related to argenx’s Business and Industry We may become exposed to costly and damaging liability claims. We are exposed to potential product liability and professional indemnity risks that are inherent in the research, development, manufacturing, marketing and use of pharmaceutical products and marketing of human therapeutic products. The current and future use of products and product candidates by us and our collaborators in clinical trials and the sale of any approved products may further expose us to liability claims. If any of our products or product candidates were to cause adverse side effects during clinical trials or after approval of the product candidate, we may be exposed to substantial liabilities. These claims might be made by patients who use the product, healthcare providers, pharmaceutical companies, physicians, payors, caregivers, investors, employees, government agencies, or our collaborators or others selling such products. Physicians and patients may not comply with any warnings that identify known potential adverse effects and patients who should not use our product candidates. Any claims against us, regardless of their merit, could be difficult and costly to defend and could materially adversely affect the market for our products and product candidates or any prospects for commercialization of our products and product candidates. Any such claims, regardless of their merit, could also adversely affect our reputation and the trust that physician and patients place in our products. Product liability risk in the EU will increase in the future once plaintiff-friendly reforms, such as Directive (EU) 2024/2853 (the new Product Liability Directive), take effect. The new Product Liability Directive introduces claimant-friendly changes. This includes, for instance, the expansion of the definition of “damage” (e.g. by including medically recognized psychological harm), creating rebuttable presumptions as to defect and causation to help claimants prove their case (e.g. if the claimant faces excessive difficulties to prove this due to scientific complexity) and abolishing minimum or maximum financial thresholds for claims. The new Product Liability Directive, like its predecessor, provides that claims shall expire if the injured person does not initiate proceedings within ten years after the defective product was placed on the market. However, it extends this long stop period (a statute of limitations) to 25 years if this is due to the latency of the underlying personal injury. Member States must transpose the Product Liability Directive into national law by December 2026. Regardless of the merits or eventual outcome, litigation or liability claims may result in: • decreased demand for our products due to negative public perception; • damage to our reputation; • withdrawal of clinical trial participants or difficulties in recruiting new clinical trial participants; • initiation of investigations by regulators; • costs to defend or settle the related litigation; • a diversion of management’s time and our resources; argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Risk Factors Related to argenx’s Financial Position 83

• substantial monetary awards to clinical trial participants or patients; • product recalls, withdrawals or labeling, marketing or promotional restrictions; • loss of revenues from product sales; and • the inability to successfully commercialize our product candidates, if approved. Although we maintain product liability insurance, we may not be able to maintain insurance coverage at a reasonable cost or to obtain adequate insurance coverage to satisfy any liability that may arise. Product liability claims could delay or prevent completion of our clinical development programs. In addition, claims made by patients, healthcare professionals or others might not be fully covered by product liability insurance and could result in investigations of the safety of our products or product candidates or may result in recalls. If a successful product liability claim or series of claims is brought against us for uninsured liabilities or in excess of insured liabilities, our assets may not be sufficient to cover such claims and our business, financial condition and results of operations would be adversely affected. In the ordinary course of business we may also face substantial, complex or extended litigation that could cause us to incur significant costs and distract our management. This is especially relevant for biopharmaceutical companies. Such litigation or proceedings could substantially increase our operating expenses and could adversely affect our business. We may engage in strategic transactions, including acquisitions, collaborations, licenses or investments in other companies or technologies, and we may not realize the benefits of such transactions. We may enter into strategic transactions, including acquisitions, collaborations, licenses or investments for or in other companies or technologies that complement or augment our existing business and facilitate our access to new products, research projects or geographical areas. However, we may not be able to identify appropriate targets or enter into such transactions under satisfactory conditions. We may be unable to complete a proposed transaction if we or our shareholders are unable to obtain required regulatory approvals in the various jurisdictions in which we or a potential acquisition target or acquirer operate. In addition, we may need additional funding to finance these transactions including through issuances of public or private equity or convertible debt securities, which could be dilutive to our shareholders and ADS holders. Integrating any newly acquired companies, business, technologies or products could be expensive, time- consuming, and may never be successful. Integration efforts often take a significant amount of time, place a significant strain on managerial, operational and financial resources, result in loss of key personnel and could prove to be more difficult or expensive than we predict. The diversion of our management’s attention and any delay or difficulties encountered in connection with any future transactions we may consummate could result in the disruption of our ongoing business or inconsistencies in standards and controls that could negatively affect our ability to maintain third-party relationships. We cannot assure that we will achieve the expected synergies to justify any such transaction, which could have a material adverse effect on our business, financial condition, results of operations and future growth prospects and our investors’ ability to realize on their investment. Our business and operations could suffer in the event of system failures or unauthorized or inappropriate use of or access to our systems. We are increasingly dependent on our and our third-party partners’ information technology systems and infrastructure for our business. We collect, store and transmit sensitive information including intellectual property, proprietary business information, including highly sensitive clinical trial data, and personal data in connection with business operations. The secure maintenance of this information is critical to our operations and business strategy. Some of this information could be an attractive target of criminal attack or unauthorized access and use by third parties with a wide range of motives and expertise, including organized criminal groups, “hacktivists”, patient groups, disgruntled current or former employees and others. Cyber-attacks are of ever-increasing levels of sophistication, and despite our security measures, our information technology and infrastructure may be vulnerable to such attacks or may be breached, including due to employee error or malfeasance. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Risk Factors Related to argenx’s Business and Industry 84

In addition to these threats, we are subject to stringent cybersecurity laws, most notably NIS2 with respect to our activities in the European Union. As a company involved in the development and manufacturing of pharmaceutical and medicinal products, our operations could be brought within the scope of NIS2. This would require us to implement robust measures to secure our network and information systems. We are also subject to cybersecurity laws in other international jurisdictions in which we conduct our operations, such as the Cybersecurity Review Measures in China. Failure to comply with these cybersecurity standards could result in significant penalties. See Section 2.5 “Risk Factors Related to Other Government Regulations—We are subject to privacy, cybersecurity and AI laws, regulation and potential enforcement. The failure to comply with these laws could harm our results, operations and/or financial conditions.” Although we are making significant efforts to maintain the security and integrity of our information systems and the information systems of our third-party partners and are exploring various measures to manage the risk of a security breach or disruption, there can be no assurance that our security efforts and measures will be effective or that attempted security breaches or disruptions would not be successful or damaging. Despite the implementation of security measures, our internal computer systems and those of our third- party partners, contractors and consultants are vulnerable to damage or interruption from computer viruses, unauthorized or inappropriate access or use, natural disasters, pandemics, terrorism, war (including the ongoing conflict in Ukraine and conflict in the Middle East), and telecommunication and electrical failures. For example, the loss of pre-clinical trial data or data from completed or ongoing clinical trials for our product candidates could result in delays in our regulatory filings and development efforts, as well as delays in the commercialization of our products, and significantly increase our costs. To the extent that any disruption, security breach or unauthorized or inappropriate use or access to our systems were to result in a loss of or damage to our data, or inappropriate disclosure of confidential, personal or proprietary information, we could incur notification obligations to affected individuals and government agencies, liability, including potential lawsuits from patients, collaborators, employees, stockholders or other third parties and liability under foreign, federal and state laws that protect the privacy and security of personal data, and the development and potential commercialization of our product candidates could be delayed. Disruptions in the our and our third-party partners’ information technology systems could adversely affect our business operations and harm our competitive position. Not all of our contracts contain limitations of liability, and even where they do, there can be no assurance that limitations of liability in our contracts are sufficient to protect us from liabilities, damages or claims related to our data privacy and security obligations. We cannot be sure that our insurance coverage will be adequate or sufficient to protect us from or to mitigate liabilities arising out of our privacy and security practices. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Risk Factors Related to argenx’s Business and Industry 85

2.8 Risk Factors Related to argenx’s Intellectual Property We may be unable to adequately maintain, enforce or protect our intellectual property rights in products, product candidates and platform technologies which could adversely affect our ability to maximize the value for patients in our marketed products and product candidates. Our commercial success depends in part on obtaining and maintaining patents and other forms of intellectual property rights for our products, product candidates and platform technologies. Failure to obtain, maintain, enforce, protect, or extend adequate patent and other intellectual property rights, which can be challenging and costly, could adversely affect our ability to develop and market our products and product candidates and reduce any competitive advantage we may have. We cannot be certain that patents will be issued or granted with respect to applications that are currently pending and we may not be the first to file patent applications related to our product candidates and products. The scope of patent protection that the European Patent Office and the USPTO will grant with respect to products in our product pipeline is uncertain and may vary. It is possible that the European Patent Office and USPTO will not allow broad claims that cover molecules closely related to our products and product candidates as well as the specific molecule, and competitors may be free to market substantially similar molecules if granted approval, thereby reducing our market potential. We and our current or future licensors, licensees or collaboration partners may not be able to prepare, file, prosecute and maintain all necessary or desirable patent applications at a reasonable cost or in a timely manner. Our current and future licensors’, licensees’ or collaboration partners’ ability to ensure the issuance, scope, validity, enforceability and commercial value of technology licenses is uncertain and we may need to rely on them to obtain costly additional IP licenses. Additionally, such parties may not fully comply with applicable patent rules or laws, which could result in loss of patent rights, or such parties may disagree with us as to the strategy for prosecution, maintenance or enforcement of any such patent rights. Filing, prosecuting, and defending patents on product candidates in all jurisdictions throughout the world would be prohibitively expensive and the laws of certain jurisdictions may not protect our rights to the same extent as the laws of the U.S., UK or EU. We may face difficulties in enforcing patent rights in the future, including in certain jurisdictions where we have not yet filed patent applications. Competitors may use our and our licensors’ or collaboration partners’ technologies in jurisdictions where we have not obtained patent protection, or where broad research exemptions are available, to develop their own products. Third parties may export otherwise infringing products to territories where we, our licensors or collaboration partners have patent protection, but where enforcement is not as strong as that in the U.S., UK and the EU. In such cases, we would have little effective recourse to prevent such products from competing with ours. In addition, some countries have compulsory licensing laws under which a patent owner may be compelled to grant licenses to third parties, and other countries limit the enforceability of patents against government agencies or government contractors. In these countries, the patent owner may have limited remedies, which could materially diminish the value of such patent. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Risk Factors Related to argenx’s Intellectual Property 86

Intellectual property litigation could lead to substantial resource diversion or issued patents could be found invalid, not infringed, or unenforceable if challenged in the applicable patent office or court. Our patents may remain open to invalidity challenges after allowance or grant, whereby third parties can challenge the scope or validity of such granted patent. In the course of such proceedings, we may be compelled to limit the scope of patent claims thus challenged or may lose the claims altogether. We may elect to initiate adversarial proceedings in order to enforce or defend any intellectual property rights owned by or licensed to us, or to determine or challenge the scope or validity of intellectual property rights of third parties to protect our competitive position. We may need to divert substantial time and resources to the enforcement and protection of our or our collaboration partners’ intellectual property rights. In addition, the outcomes of any proceedings could be uncertain and any remedies or damages awarded may not be meaningful. An adverse ruling of non-infringement, limiting claim scope, or invalidating one or more of our issued patents could allow third parties to commercialize our products after the expiration of our market exclusivity or use our platform technologies to compete directly with us, without payment to us. In many jurisdictions (including the Unified Patent Court), an action to enforce a patent may not be brought until immediately before, or even after, the infringing product of a competitor has been launched in the market. In these jurisdictions, infringement rulings and injunctions (even in preliminary injunction proceedings) are often issued only after the competing product has already been launched and potentially impaired the competitive advantage on the market. We may be subject to claims challenging the inventorship or ownership of our intellectual property or be required to make additional payments to secure intellectual property from collaborators. Many of our consultants and employees, including in the senior management team (consisting of our CEO and senior personnel reporting directly to the CEO) (Senior Management Team), were previously employed at other competing or potentially competing biotechnology or pharmaceutical companies and some have executed proprietary rights, non-disclosure and non-competition agreements in connection with such previous employment. Although we take measures to ensure third parties, consultants and employees do not use such proprietary information in their work for us, we may be subject to claims that we or these consultants and employees have improperly used or disclosed confidential information or intellectual property of their former employer. Additionally, many of our collaborators do not commit to assigning all intellectual property arising out of our collaborations to us and, instead, grant us options to acquire intellectual property or commit to making such intellectual property available to us at a fair price. As such, we may be required to make additional payments to secure valuable intellectual property rights under our existing collaborations or become subject to inventorship disputes. In addition, although we take steps to ensure that our collaborators do not use our intellectual property rights other than for the purposes of our collaboration, there may be instances where former or current collaborators or other third parties nevertheless apply for or obtain patent protection for inventions to which we believe we have rights, in whole or in part. In such cases, we may elect to assert our ownership of such intellectual property. There is no guarantee that we will be successful in asserting such claims. In addition, while it is our policy to require our employees and contractors who may be involved in the development of intellectual property to execute agreements assigning such intellectual property to us, we may be unsuccessful in ensuring effective assignment of intellectual property under such agreements. Our assignment agreements may not be self-executing, or may be breached, and we may be forced to bring or defend against claims to assert ownership of such intellectual property. There is no guarantee we will be successful in pursuing such claims, which could result in us paying monetary damages or losing valuable personnel or intellectual property rights. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Risk Factors Related to argenx’s Intellectual Property 87

Third-party intellectual property rights could adversely affect our ability to commercialize our products and product candidates. Our revenue generated may suffer if valid and enforceable third-party intellectual property rights cover our products, product candidates, manufacturing processes, or those of our partners. In such cases, our freedom to develop or commercialize products or product candidates may require obtaining a license, designing around third party intellectual property rights with significant time and materials costs, or invalidating the third party rights. If our products are found to infringe a valid and enforceable patent claim, we and our partners could be prevented from continuing to develop or commercialize the affected product without an appropriate license, which may be costly or unavailable on commercially reasonable terms, if at all. Similarly, other companies may have filed patent applications or have patents directed toward molecules and/or uses of those molecules that modulate similar targets modulated by our products and we may not be aware of unpublished pending patent applications or patent applications that are amended to cover our products or platform technologies. Even if we or our partners can obtain the appropriate license, it may be non-exclusive, thereby providing third parties with the opportunity to access the same licensed technology. If the breadth and scope of protection provided by our or our partners’ patents, licensed patents, or patent applications is threatened or limited, it could dissuade companies from future collaborations with us to license, develop, or commercialize products and product candidates which would have an adverse effect on our business position. Furthermore, because of the substantial amount of discovery required in connection with intellectual property litigation, some of our confidential information could be disclosed in any such proceedings. We may not be successful in obtaining or maintaining necessary rights to our products and product candidates through acquisitions and in-licenses. We may be unable to acquire or in-license third-party intellectual property rights necessary or useful for development or commercialization of our product, product candidates or technology. We sometimes collaborate with U.S. and non-U.S. academic institutions and typically receive an option to negotiate a license to the institution’s proprietary interest in any collaboration technology. However, we may be unable to successfully negotiate such license and the institution may offer such intellectual property rights to third parties thereby devaluing our exclusive rights to pursue the applicable program. In addition, our competitor companies may be unwilling to license desirable or necessary intellectual property rights to us, or we may be otherwise unable to license or acquire other third party intellectual property rights on commercially reasonable terms which could negatively impact current development or hinder our ability to pursue development of new programs. Under our existing licenses, failure to comply with our obligations thereunder could result in termination of such licenses, thereby limiting our ability to develop and commercialize products covered by such licensed technology. Moreover, despite our efforts to comply with our contractual obligations, our licensors could conclude we have materially breached any such agreement and we could incur significant costs and disruption to our business defending against any breach alleged by the licensor. Moreover, several of our existing license agreements are sub-licenses from third parties. We have little control if our licensors fail to comply with their obligations under their upstream license agreements, whereby the original third-party licensor may have the right to terminate the original license and possibly our sub-license. In such cases, we may not be able to procure a direct license covering such intellectual property possibly materially affecting our ability to develop and commercialize certain products and product candidates. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Risk Factors Related to argenx’s Intellectual Property 88

If our brand protection strategies, including the filing, prosecution and enforcement of trademarks and trade names, are not adequately executed, we may not be able to build name recognition for approved products in our markets of interest in line with our strategic priorities. Third parties may seek to oppose, attempt to cancel our trademark applications, or challenge, infringe or circumvent our registered and unregistered trademarks, including through counterfeiting of our products. In the event that our trademarks are successfully challenged, we may not be able to use these trademarks to continue to effectively market our branded products and could be forced to rebrand them, which could result in loss of their brand recognition or require us to devote resources to develop new brand profiles. Such efforts could also hinder our efforts to commit to and deliver on strategic internal initiatives. If we attempt to enforce our trademarks or assert trademark infringement claims, a court may determine that our trademarks are invalid or unenforceable or that the party against whom we have asserted trademark infringement has superior rights with respect to such marks. If we are unable to establish name recognition and adequately protect and enforce our trademark portfolio, we may not be able to compete effectively in the market or build brand recognition for new products globally. We may not be able to obtain protection under the U.S. Drug Price Competition and Patent Term Restoration Act of 1984 (Hatch-Waxman Act) and similar non- U.S. legislation for extending the term of patents covering each of our products and product candidates. Depending upon the timing, duration and conditions of FDA marketing approval of our product candidates, one or more of our U.S. patents may be eligible for limited patent term extension under the Hatch-Waxman Act and similar legislation in the EU and the Asia Pacific region. However, the patent term extension under the Hatch Waxman Act cannot extend the remaining term of a patent beyond a total of 14 years from the date of product approval, and only one patent applicable to an approved drug may be extended. If we are unable to obtain patent term extension, the term of any such extension is less than we request, or the statutes governing patent term extension are amended to reduce the term of such extensions, our patent exclusivity for that product will be shortened and our competitors may obtain approval to market competing products sooner than we expect. Changes in patent laws or patent jurisprudence could diminish the value of patents in general thereby impairing our ability to protect our products. Changes in patent law across jurisdictions, or changes in any relevant government’s enforcement procedure may weaken our ability to obtain new patents or to enforce rights in our owned and licensed patents. For example, the U.S. Supreme Court has ruled on several patent cases in recent years, either narrowing the scope of patent protection available in certain circumstances or weakening the rights of patent owners in certain situations. Relatedly, the U.S. Congress is considering multiple draft bills that, if passed, may have a significant impact on U.S. patent laws. Any such changes by the U.S. Congress or U.S. courts and the relevant law-making bodies in other countries may materially affect our patents or patent applications and we cannot predict the effects of future changes in patent law. We may be unable to protect the confidentiality of our trade secrets and their disclosure to competitors, harming our market position. In addition to patent protection, we rely on trade secret protection to preserve the value of our proprietary information supporting our business model. We take balanced measures to preserve the commercial value of our trade secrets and prevent their misappropriation. However,despite these measures, which include segregation of key files, requiring our licensors, collaborators, suppliers, consultants and advisors to execute confidentiality agreements, we cannot fully protect against willful or inadvertent unauthorized disclosure of our trade secrets. Trade secret enforcement proceedings can be unpredictable and vary across geographies and we may not be able to secure adequate legal or equitable remedies to prevent lost value in our misappropriated trade secrets. Any misappropriation and use, whether willful or inadvertent, could result in lost value to the company and enable our competitors to duplicate or build upon our technology. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Risk Factors Related to argenx’s Intellectual Property 89

2.9 Risk Factors Related to argenx’s Organization and Operations Our future growth and ability to compete depends on maintaining our culture, retaining our key personnel and recruiting additional qualified personnel. We believe that our corporate culture has been, and will continue to be a key contributor to our success. However, as we implement more complex organizational structures, and increase our headcount to support the growth in our business, our ability to foster our key values - innovation, co-creation, empowerment, excellence and humility that we believe are important to support our growth - may be impacted. We may find it increasingly difficult to maintain the beneficial aspects of our corporate culture, which could similarly negatively impact our ability to attract, retain and motivate qualified employees and our future success. As a global organization in a highly competitive and specialized industry, our success also depends upon the continued contributions of our key management, scientific, medical and technical personnel, many of whom have been instrumental for us and have substantial experience with our product and related technologies. These key management individuals include the members of the Board of Directors and Senior Management Team. Difficulties in recruiting or the loss of key managers, scientific, medical or technical personnel could delay our research and development activities. In addition, it may be difficult to attract and retain highly qualified management, scientific and medical personnel, particularly if we expand into fields that will require additional skills. In addition, future leadership transitions and management changes may cause uncertainty in, or a disruption to, our business, and may increase the likelihood of senior management or other employee turnover. We are currently undergoing a leadership transition, as in early January 2026, we announced that Karen Massey, our current COO, will transition to the role of CEO and Executive Director and Tim Van Hauwermeiren, our current CEO, will transition to the role of Non-Executive Director and Chairperson of the Board of Directors. Both transitions are subject to shareholder approval at the 2026 General Meeting. Any failure to ensure an effective transition or any future management changes could adversely affect our business, and we cannot predict the likelihood, timing or effect of future transitions among our executive leadership. As a Dutch company listed on Euronext Brussels and Nasdaq, our remuneration practices and policies may be limited by local governance rules or shareholder guidance for EU companies. Such limitations may make it more difficult to successfully compete for key talent in a number of markets with differing remuneration practices and policies compared to our competitors. For example, the Dutch Corporate Governance Code 2025 (DCGC) places certain limitations on the ability to grant equity incentives to Non-Executive Directors, while Belgian law requires Non-Executive Directors to receive part of their remuneration in the forms of shares, but not stock options. The DCGC also places limitations on amount of severance payment permitted in the event of dismissal. Many other biotechnology and pharmaceutical companies and academic institutions that we compete against for qualified personnel have greater financial and other resources, different risk profiles and a longer history in the industry than we do. Additionally, inflationary pressures and a tight labor market for skilled workers may increase compensation expectations, requiring us to raise operating costs in order to attract and retain employees. Therefore, we might not be able to attract or retain these key persons or other employees on conditions that are economically acceptable. Global geo- and socio-political threats and macro-economic uncertainty and other unforeseen political crises could materially and adversely affect our business and financial performance. Many geo- and socio-political threats and macro-economic uncertainties are outside of our control and could adversely affect consumer confidence and disposable income levels, increase difficulty in forecasting our financial results and have other impacts on our business and financial performance. Such geo- and socio-political threats could also result in volatility in stock markets in general, causing our stock to have extreme price and volume fluctuations unrelated to our business and financial performance. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Risk Factors Related to argenx’s Organization and Operations 90

Such geo- and socio-political threats and uncertainties include: • general economic and market conditions, including government shutdowns, including the U.S. government shutdown, instability resulting from inflationary pressures, and increasing interest rates; • geopolitical events, including natural disasters, public health emergencies, acts of war, and terrorism; • economic and financial sanctions, trade embargoes, import and export regulations, tariffs, customs, and inbound and outbound investment restrictions; • restrictions, changes in trade agreements, trade barriers or other restrictions on foreign trade, and changes in trade regulations and restrictions, including between the U.S. and other countries; • global or regional economic conditions that impact companies and customers with which we do business; • political or social unrest, economic instability, repression, or human rights issues; • disruptions in supply chains; • risks related to other government regulation or required compliance with local laws; • consumer and commercial credit availability, unemployment, and consumer debt levels; and • local licensing and reporting obligations. Due to our international operations and the fact that we run clinical trials across multiple jurisdictions, geopolitical conflicts and related sanctions may, in certain circumstances, impact our ability to conduct or complete clinical trials in the affected regions. For example, General License 6D issued by the U.S. Department of the Treasury’s Office of Foreign Assets Control currently authorizes certain clinical trial and medical research activities that would otherwise be restricted by U.S. sanctions targeting Russia, the scope, availability, or duration of such authorizations may change. We also perform development activities in a number of countries exposed to geopolitical risk and if conflicts in those countries were to escalate further and impact neighboring countries, it could impact our development activities in those countries. Changes in U.S.-Mainland China relations, including tariffs, export controls, economic and financial sanctions, and other regulations may adversely impact our collaboration with Zai Lab in Mainland China, Hong Kong, Taiwan and Macau (together, Greater China). The U.S. government has taken steps and continues to take steps with regard to U.S.-Mainland China relations that will impact companies with connections to the U.S. or Mainland China, including by imposing tariffs affecting certain products manufactured in Mainland China, imposing economic and financial sanctions on certain individuals and entities in the Mainland China, and issuing statements indicating enhanced review of companies with significant Mainland China-based operations. The U.S. government has also passed laws, including the BIOSECURE Act, which was signed into law on December 18, 2025, that could limit or restrict our ability to purchase products or services from, or otherwise collaborate with, certain Chinese biotechnology companies without losing our ability to contract with or receive funding from the U.S. government. Such restrictions could have an adverse impact on our operations. Several countries are considering or have implemented tariffs, trade barriers or restrictions, as well as other measures impacting cross-border commerce, which could negatively affect our business, financial conditions and results of operations, including by disrupting our research and development activities, affecting our suppliers and negatively impacting our supply chain, impacting our ability to sell our products outside of the United States and negatively impacting our revenues from product sales or our cost of goods sold. The U.S. federal government has implemented tariffs on certain foreign goods and may implement additional or revised tariffs in the future. Such actions could give rise to retaliatory tariffs imposed by foreign governments and an escalation of trade measures by the U.S. and impacted countries. Developments with regard to the timing and manner in which tariffs will be implemented; the amount, scope, and nature of tariffs; the countries subject to new or additional tariffs imposed by the U.S.; tariffs imposed by other countries on goods imported from the U.S.; and other wide-ranging retaliatory measures are rapidly evolving and may change unexpectedly at any time. For example, the U.S. President has repeatedly announced plans to impose 100% tariffs on imported branded or patented pharmaceuticals, unless the importing company is building U.S. manufacturing capacity. It is not yet clear whether these tariffs would apply to the importation of active pharmaceutical ingredients and possibly bulk drug products argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Risk Factors Related to argenx’s Organization and Operations 91

that are intended for use in clinical trials and not for commercial sale, which could increase the costs of materials for our clinical trials. Any direct tariffs, if imposed on pharmaceutical products, may result in increased costs for raw materials and contract manufacturing services, reduced ability to source critical CMOs, and a delay in our development timelines. Although on February 20, 2026, the U.S. Supreme Court struck down certain tariffs imposed by the Trump Administration pursuant to the International Emergency Economic Powers Act, the Trump Administration responded by announcing new tariffs pursuant to another statute. Other countries may, in turn, retaliate with new tariffs against US exports. These events increase the uncertainty and the risks we face resulting from tariffs described above. Any new legislation, executive orders, tariffs, export controls, economic and financial sanctions, trade embargoes and/or other regulations that may be implemented, any unfavorable government policies on international trade, including tariffs and export controls, the renegotiation of existing trade agreements, any increased scrutiny on companies with significant Mainland China-based operations, and any retaliatory actions taken by the U.S., EU, Chinese or other governments due to trade tensions could have an adverse effect on our business, including the development and commercialization of products containing argenx- licensed material. Further, general political uncertainty may have an adverse impact on our business, financial condition and results of operations. We face risks related to natural disasters and public health issues, that could negatively affect our business and financial condition. Our business could be adversely impacted by the effects of catastrophic global events including natural disasters such as earthquakes, fires, hurricanes, tornados, floods or significant power outages and public health crises. For example, the manufacturing of all of our products and product candidates requires using cells which are stored in a cell bank. We have one master cell bank for each product manufactured in accordance with cGMPs. However, it is possible that we could lose multiple cell banks and have our manufacturing significantly impacted by the need to replace these cell banks, which could materially adversely affect our business, prospects, financial condition and results of operations. Public health issues could also negatively affect our business and financial condition. We operate and conduct our clinical trials globally. We cannot presently predict the scope and severity of any potential future business shutdowns or disruptions as a result of public health issues. If we or any of the third parties with whom we engage, including the suppliers, contract manufacturers, clinical trial sites, regulators and other third parties, were to experience shutdowns, quarantines, or other business disruptions due to natural disasters or global public health issues, it may impair our or our third-party partners’ ability to initiate clinical trials and recruit and retain patients, particularly if quarantine or travel restrictions impede healthcare provider or patient movement, impact the usability of the data due to treatment interruptions and require protocol amendments. In addition, regulatory authorities may restrict their operations or be delayed in their operations during a pandemic, the outbreak of new variants or other public health issues, including further to travel restrictions which could adversely affect our ability to obtain regulatory approval for and to commercialize our products and product candidates and have a material adverse effect on our business and financial results. We are exposed globally to unanticipated changes in tax laws and regulations, adjustments to our tax provisions, exposure to additional tax liabilities, or adjustments of our tax assets. As a company active in research and development, we have benefited from certain research and development tax incentives including tax credits and a payroll withholding tax exemption. We also expect to benefit from the Belgian innovation income deduction. The determination of our provision for income taxes and other tax liabilities requires judgment, including the adoption of certain accounting policies and our determination of whether our deferred tax assets are, and will remain, fully available in future periods. We cannot guarantee that our interpretation of applicable tax laws (including with respect to our eligibility for, or our calculation of, tax incentives such as the Belgian R&D tax credit, the Belgian payroll withholding tax exemption for R&D personnel, the Belgian innovation income deduction and similar tax incentives in other jurisdictions in which we have material operations or sales), our transfer pricing policies or our organizational and operational structure will not be questioned by the relevant tax authorities, or that the relevant tax laws and regulations, or the interpretation thereof, argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Risk Factors Related to argenx’s Organization and Operations 92

including through tax rulings, will not be subject to change. Our effective tax rates could be adversely affected, now or in the future, by changes in tax laws, treaties and regulations or the interpretation thereof by the relevant tax authorities in countries where we have material operations. A successful challenge to tax positions in Belgium or other country where we have material operations may lead to adjustments in the amounts recorded in our financial statements and could have a significant impact on our effective tax rate and on our deferred tax assets. An increase of the effective tax rates could have an adverse effect on our business, financial position, results of operations and cash flows. In case of a change of control, we could be exposed to the risk of losing any unused tax credit and innovation income deduction. Furthermore, if any legislator decides to eliminate, or change the conditions for claiming such tax incentives, or reduce the scope or the rate of such incentives, any of which it could decide to do at any time, our results of operations could be adversely affected including through the de- recognition of deferred tax assets. We may encounter difficulties efficiently managing our growth and our increasing development, regulatory, and sales and marketing capabilities, which could disrupt our operations. We have grown, and expect to continue to grow globally, significantly in the number of employees and scope of operations over recent years, particularly in the areas of drug research, drug development, regulatory affairs, and sales and marketing. To manage our anticipated future growth and support our expanding global footprint, we must continue to enhance our managerial, operational and financial systems, expand our facilities and recruit and train additional qualified personnel and build the internal infrastructure necessary. Any inability to manage growth effectively could delay the execution of our strategic objectives or disrupt our operations, which in turn could materially harm our business and prospects. In particular, our planned international expansion subjects us to a number of risks, including risks following complexities in monitoring and coordinating research and development, marketing, supply chain and other operations in a large number of jurisdictions, risks related to laws, regulations and policies, including those implemented following changes in political leadership and trade, risks related to varying standards and practices in the legal, regulatory and business cultures in which we operate, risks related to capital and exchange controls, cross-border taxes and tariffs, cross-border data transfer restrictions, and complex sanctions regimes in various countries such as the U.S., the EU and other jurisdictions, violations of which could lead to fines or other penalties, risks related to geopolitical and local political instability and uncertain business environments and risks related to complexities associated with managing local personnel and preventing misconduct by local third-party partners. The application of laws and regulations impacting global business operations is often unclear and may at times conflict. Compliance with these laws and regulations may involve significant costs or require changes in our business practices that result in reduced revenue and profitability. Non-compliance could also result in fines, damages, criminal sanctions, prohibited business conduct, and damage to our reputation. 2.10 Risk Factors Related to the ADSs Holders of our ADSs have fewer rights than our ordinary shareholders. Except as described in this Annual Report or any deposit agreements, holders of ADSs are not treated as our shareholders unless they withdraw the ordinary shares underlying their ADSs in accordance with the deposit agreement and applicable laws and regulations. The depositary, or its nominee, is the holder of the ordinary shares underlying the ADSs. ADSs are transferable on the books of the depositary. The depositary may refuse to deliver, transfer or register transfers of ADSs. Temporary delays in the cancellation of ADSs and withdrawal of the underlying ordinary shares may arise. In addition, ADS holders may not be able to cancel their ADSs and withdraw the underlying ordinary shares when they owe money for fees, taxes and similar charges and when it is necessary to prohibit withdrawals in order to comply with any laws or governmental regulations that apply to ADSs or to the withdrawal of ordinary shares or other deposited securities. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Risk Factors Related to argenx’s Organization and Operations 93

Holders of ADSs may vote them in person or by proxy in accordance with applicable laws and regulations and our articles of association (Articles of Association). We cannot guarantee that holders of ADSs will receive the voting materials in time to ensure that they can instruct the depositary to vote the ordinary shares underlying their ADSs. Holders of our ADSs may not be able to exercise their right to give voting instructions or to vote in person or by proxy and they may not have any recourse against the depositary or us if their ordinary shares are not voted as they have requested or if their shares cannot be voted. The price of our ADSs may be volatile and may fluctuate due to factors beyond our control. An active public trading market may not be sustained. The stock markets in general, and biopharmaceutical companies in particular, have experienced extreme price and volume fluctuations that have often been unrelated or disproportionate to the operating performance of these companies. The trading price of our ADSs depends on a number of factors, including those described elsewhere in this “Risk Factors” section, many of which are beyond our control and may not be related to our operating performance, which may limit or prevent investors from readily selling their ADSs or ordinary shares and may otherwise negatively affect the liquidity of our ADSs and ordinary shares. We provide guidance regarding our cash and expenses, which are inherently uncertain. Any guidance that we provide may not always be accurate or may vary. If we fail to meet our guidance, or if we have to revise such guidance, the price of our ADSs or ordinary shares could decline. Sales of a substantial number of ADSs or our ordinary shares in the public market, or the perception that these sales might occur, could depress the market price of ADSs or our ordinary shares and could impair the market price of our securities or our ability to raise capital through the sale of additional equity securities. In addition, an active public trading market for our ADSs may not be sustained. Further, fluctuations in exchange rates may also impact the price of our ADSs and ordinary shares which may result in heavy trading by investors seeking to exploit such differences, or impact the proceeds holders receive. If securities or industry analysts cease coverage of us, or publish inaccurate or unfavorable research about our business, the price of our ADSs or ordinary shares and our trading volume could decline. The trading market for the ADSs and ordinary shares depends in part on the research and reports that securities or industry analysts publish about us or our business. We do not have control over these analysts. If no or too few securities or industry analysts cover us, the trading price of our ADSs and ordinary shares would likely be negatively affected. If one or more of the analysts who cover us downgrade our ADSs or ordinary shares or publish inaccurate or unfavorable research about our business, the price of our ADSs or ordinary shares would likely decline. If we fail to maintain an effective system of disclosure controls and internal control over financial reporting, our ability to comply with applicable regulations could be impaired, and the trading price of our ADSs may be negatively impacted. We are required to comply with various corporate governance and financial requirements under the Sarbanes-Oxley Act of 2002, the Dodd-Frank Wall Street Reform and Consumer Protection Act, the listing rules of the Nasdaq Global Market (the Nasdaq Listing Rules) and requirements, and other applicable securities rules and regulations. We are required to furnish a report by management on, among other things, the effectiveness of our internal control over financial reporting and an attestation report on internal control over financial reporting issued by our independent registered public accounting firm. Undetected material weaknesses in our internal controls could lead to financial statement restatements and require us to incur the expense of remediation. Moreover, any failure to maintain internal control over financial reporting or any material weaknesses or significant deficiency thereover, could result in a loss of investors’ in the accuracy, completeness and reliability of our financial statements, subject us to sanctions or investigations, or negatively impact the trading price of our ADSs or ordinary shares. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Risk Factors Related to the ADSs 94

2.11 Risk Factors Related to being a Foreign Private Issuer or a Dutch Company The risks in this subsection that relate to our status as a foreign private issuer will change if we lose our status as a foreign private issuer under U.S. law. We are a Dutch European public company with limited liability. The rights of our shareholders may be different from the rights of shareholders in companies governed by the laws of U.S. jurisdictions. We are a Dutch European public company with limited liability (Societas Europeae). The rights of shareholders and the responsibilities of members of our Board of Directors may be different from the rights and obligations of shareholders in companies governed by the laws of U.S. jurisdictions. As a result of these differences between Dutch corporate law and our Articles of Association, on the one hand, and the U.S. federal and state laws, on the other hand, in certain instances, our shareholders and holders of our ADSs could receive less protection than they would as shareholders or ADS holders of a listed U.S. company. For example, provisions of our Articles of Association may make it more difficult for a third party to acquire control of us or effect a change in our Board of Directors. We have adopted several provisions that may have the effect of making a takeover of our Company more difficult or less attractive. These provisions could discourage potential takeover attempts that other shareholders may consider to be in their best interest and could adversely affect the market price of our securities. Holders of our ordinary shares outside the Netherlands, and holders of ADSs may not be able to exercise pre-emptive rights or preferential subscription rights, respectively. In the event of an increase in our share capital, holders of our ordinary shares are generally entitled under Dutch law to full pre-emptive rights, unless these rights are excluded either by a resolution of the shareholders at a General Meeting, or by a resolution of the Board of Directors (if the Board of Directors has been designated by the shareholders at a General Meeting for this purpose). However, making pre-emptive rights available to holders of ordinary shares or ADSs representing ordinary shares also requires compliance with applicable securities laws in the jurisdictions where holders of those securities are located, which we may be unable or unwilling to do. In particular, holders of ordinary shares or ADSs located in the U.S. would not be able to participate in a pre-emptive rights offering unless we registered the securities to which the rights relate under the U.S. Securities Act of 1933, as amended (Securities Act) or an exemption from the registration requirements. In addition, ADS holders would not be able to participate in a pre-emptive rights offering unless we made arrangements with the depositary to extend that offering to holders of ADSs, which we are not required to do. Claims of U.S. civil liabilities may not be enforceable against us or the members of our Senior Management Team and our Board of Directors. A significant amount of our assets are located outside the U.S. The majority of the members of our Senior Management Team and our directors are not U.S. residents and we do not have significant assets in the U.S. As a result, it may not be possible, or more difficult, for investors to enforce against them or us in U.S. courts, including judgments predicated upon the civil liability provisions of the U.S. federal securities laws. There are no treaties between the U.S. with either the Netherlands or Belgium providing for the reciprocal recognition and enforcement of judgments, other than arbitration awards, in civil and commercial matters. Consequently, a final judgment for payment given by a court in the U.S. based on civil liability, whether or not predicated solely upon U.S. securities laws, would not automatically be recognized or enforceable in the Netherlands or in Belgium unless the underlying claim was re-litigated before a Dutch or Belgian court of competent jurisdiction. This will depend on the applicable Dutch or Belgian national rules. In addition, there is doubt as to whether a Dutch or Belgian court would impose civil liability on us or the members of our argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Risk Factors Related to being a Foreign Private Issuer or a Dutch Company 95

management or of our Board of Directors in an original action predicated solely upon the U.S. federal securities laws brought in a court of competent jurisdiction against us, our management or directors. As a foreign private issuer, we are exempt from various rules and regulations that a U.S. domestic public company would be required to follow, including those requirements under U.S. securities laws and Nasdaq listing standards. As a “foreign private issuer” defined in the SEC’s rules and regulations, we are not subject to all of the disclosure and corporate governance requirements applicable to companies organized within the United States. For example, we are exempt from certain provisions of the U.S. Securities Exchange Act of 1934, as amended (Exchange Act), that are applicable to U.S. domestic public companies. In addition, our executive officers, directors and principal shareholders are exempt from the short-swing profit and recovery provisions contained in Section 16(b) of the Exchange Act. We are subject to Dutch laws and regulations with regard to such matters. While we furnish quarterly unaudited financial information to the SEC on Form 6-K, the information we furnish to the SEC is less extensive and less timely compared to that required to be filed with the SEC by U.S. domestic issuers. As a foreign private issuer listed on Nasdaq, we are subject to corporate governance listing standards. However, we are permitted to rely on home country governance requirements and certain exemptions thereunder. Certain of our corporate governance practices may differ significantly from the Nasdaq corporate governance listing standards. These practices may afford less protection to shareholders than they would enjoy if we complied fully with corporate governance listing standards. As a Dutch public company with limited liability, we are not obligated to, and do not comply with, all the best practice provisions of the DCGC, which may affect shareholders’ rights. We are required to disclose in our annual report, filed in the Netherlands, whether we comply with the provisions of the DCGC. If we do not comply with the provisions of the DCGC, we must list the reasons for any deviation from the DCGC in our annual report filed in the Netherlands. We may lose our foreign private issuer status which would then require us to comply with the Exchange Act’s domestic reporting regime and cause us to incur significant legal, accounting and other expenses. In order to maintain our current status as a foreign private issuer, either (a) a majority of our ordinary shares must be either directly or indirectly owned of record by non-residents of the U.S. or (b) none of the following conditions may be true: (i) a majority of our executive officers or directors are U.S. citizens or residents, (ii) more than 50% of our assets are located in the U.S. and (iii) our business is administered principally in the U.S. As of February 19, 2025, we believe at least 50% of our outstanding ordinary shares were held by U.S. residents (assuming that all our ordinary shares represented by ADSs were held by residents of the U.S.). In June 2025, the SEC issued a concept release soliciting public comment on potential changes to the definition of a foreign private issuer. This release is the first review of the foreign private issuer framework since 2008, and the SEC is considering revisions that could significantly impact which foreign companies qualify for the more accommodative U.S. reporting, corporate governance requirements afforded to foreign private issuers, as well as the lack of Section 16(b) liability for certain FPI officers, directors and shareholders. The SEC may revise the requirements to qualify as a foreign private issuer in a way that changes our status as a foreign private issuer and results in us qualifying as a U.S. domestic issuer. This could cause us to have to comply with regulations applicable to U.S. domestic issuers in a very compressed timeframe. The regulatory and compliance costs to us as a U.S. domestic issuer may be significantly higher than those we incur as a foreign private issuer. We also expect that if we were required to comply with the rules and regulations applicable to U.S. domestic issuers, it would make it more difficult and expensive for us to obtain director and officer liability insurance, and we may be required to accept reduced coverage or incur substantially higher costs to obtain coverage. These rules and regulations could also make it more difficult for us to attract and retain qualified members of our Board of Directors. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Risk Factors Related to being a Foreign Private Issuer or a Dutch Company 96

If we were to be classified as a passive foreign investment company for U.S. federal income tax purposes, this could result in adverse U.S. tax consequences to certain U.S. holders. If the Company is classified as a passive foreign investment company (PFIC) for any taxable year, U.S. investors may be subject to adverse U.S. federal income tax consequences described below under Section 5.15.1 “Taxation – U.S. Federal Income Tax Considerations – Passive Foreign Investment Company Considerations” The Company will be classified as a PFIC for U.S. federal income tax purposes for any taxable year in which, taking into account a pro rata portion of the income and assets of 25% or more owned subsidiaries, either (i) at least 75% of its gross income consists of “passive income” or (ii) at least 50% of the average quarterly value of its assets is attributable to assets that produce, or are held for the production of, passive income. Based on our historic and anticipated operations, the composition of our income and the projected composition and estimated fair market values of our assets, we do not believe that we were a PFIC for our most recent taxable year and do not expect to be classified as a PFIC for the current taxable year or for the foreseeable future. However, our status as a PFIC is a factual determination made on an annual basis, and we cannot provide any assurances regarding our PFIC status for the current or future taxable years. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Risk Factors Related to being a Foreign Private Issuer or a Dutch Company 97

Corporate Governance 3.1 Dutch Corporate Governance Code 99 3.2 Management Structure 100 3.3 Report of the Non-Executive Directors 118 3.4 Remuneration Report and Compensation Statement 121 3.5 Corporate Governance – Nasdaq Listing Rules 158 3.6 Share Ownership 158 3.7 Insider Trading 158 3.8 Cybersecurity 159 3.9 Risk Appetite & Control 160 argenx Annual Report 2025 98 3

3 Corporate Governance 3.1 Dutch Corporate Governance Code As a European public company (Societas Europaea) incorporated under the laws of the Netherlands, we are subject to the DCGC. A copy of the DCGC can be found at www.mccg.nl. The DCGC is based on the notion that a company is a long-term alliance between the various stakeholders of the company. Stakeholders are groups and individuals who, directly or indirectly, influence – or are influenced by – the attainment of our objectives: employees, shareholders and other capital providers, suppliers, patients, healthcare community, academic partners and other stakeholders. The DCGC is based on a “comply or explain” principle. Accordingly, companies are required to state the extent to which they comply with the principles and best practice provisions of the DCGC in their annual report and, where they do not comply with them, why and to what extent they deviate from them. We acknowledge the importance of good corporate governance and we fully endorse the underlying principles of the DCGC, which is reflected in a policy that complies with the best practice provisions as stated in the DCGC (the Board By-Laws). However, we deviate from the best practice provisions in the areas set out below, for the reasons explained in this section. • Pursuant to best practice provision 2.3.2 of the DCGC, if the Board of Directors comprises more than 4 Non-Executive Directors, it should appoint a nomination committee, an audit committee and a remuneration committee. However, the Board of Directors has combined the tasks and duties of the nomination committee and the remuneration committee into one committee, being the Remuneration and Nomination Committee for efficiency purposes. • Pursuant to best practice provisions 3.1.2 under vi of the DCGC, shares should be held by directors for at least five years after they are awarded. Whereas we do have minimum holding requirements requiring our directors and executive management to hold minimum levels of ownership in the company during their time in function, we do not have a generic restriction on selling shares within five years after they are granted. We regularly benchmark our equity incentive practices, and in 2025 even more closely so in connection with our efforts to update our remuneration policy, and note that an all-out selling restriction of five years post grant is significantly more restrictive than restrictions applied by a large majority of our peer group. We believe we have several measures in place to effectively ensure long-term alignment of interests and we do not expect to implement a general five-year holding requirement for all equity in the foreseeable future. With the approval of our remuneration policy during the extraordinary general meeting on November 18, 2025, (the 2025 Extraordinary General Meeting), we have introduced cliff vesting periods of three years for stock options and performance share units (PSUs) for statutory executive directors (Executive Directors) and the requirement that restricted shares granted to Non-Executive Directors must be held for four years (except to the extent necessary to cover immediate tax obligations resulting from the immediate vesting). In addition, we also increased the minimum holding requirements to 6x base salary for Executive Directors and 5x annual board membership retainer fees for Non-Executive Directors. • Pursuant to best practice provision 3.2.3 of the DCGC, the severance payment in the event of dismissal should not exceed one year’s base compensation. The management agreement with our current chief executive officer and sole Executive Director (CEO) contains a legacy provision, setting the contractual notice period for termination and severance arrangements at 18 months, which we will continue to respect during the duration of his management agreement, which will be dissolved as of the date of the 2026 General Meeting. For any other Executive Director(s) (including Karen Massey who will be nominated for appointment as Executive Director at our upcoming 2026 General Meeting), severance arrangements will not exceed 12 months unless required by local laws. • Pursuant to best practice provision 3.3.2. of the DCGC, Non-Executive Directors should not be granted any shares or rights to shares as remuneration. When attracting qualified Non-Executive Directors, we directly compete with other companies who like us, are listed on a major U.S. stock exchange and face the corresponding stringent regulatory and legal environments. In order to be competitive, it is essential to be able to attract Non-Executive Directors who can navigate these complex requirements along with argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Dutch Corporate Governance Code 99

the accompanying responsibility and liability risks. We annually benchmark and review Non-Executive Director total remuneration against our peer group, which is selected based on objective criteria that we disclose annually in the remuneration report. We realize that granting equity to Non-Executive Directors is viewed differently in the Dutch context and is a deviation from this best practice provision. However, considering our peer group, the international context in which we operate and compete with for talent, and the fact that the corporate governance code principles in our country of primary listing (Belgium) actually require paying part of the non-executive fees in the form of equity, granting equity in the form of restricted shares is a well-considered deviation from the DCGC. The immediate vest at grant of the restricted shares, combined with a holding period of four years (except to the extent necessary to cover immediate tax obligations resulting from the immediate vest) ensures alignment of interest between our Non-Executive Directors and our shareholders. We do not expect to change this practice in the foreseeable future. 3.2 Management Structure 3.2.1 General As at December 31, 2025, we have a one-tier board structure consisting of 1 Executive Director and 8 Non-Executive Directors, and a Senior Management Team responsible for the day-to-day operations. Set out below is a summary of certain provisions of Dutch corporate law as of the date of this Annual Report, as well as a summary of relevant information concerning our Board of Directors and certain provisions of our Articles of Association and the Board By-Laws. This summary does not purport to give a complete overview and should be read in conjunction with and is qualified in its entirety by reference to the relevant provisions of Dutch law as in force on the date of this Annual Report, the Articles of Association and Board By-Laws. The Articles of Association are available in the governing Dutch language and an unofficial English translation thereof, and the Board By-Laws are available in English, on our website. 3.2.2 Statement of the Board of Directors Responsibilities for the Financial Statements and Board Report In accordance with Article 5:25c(2)(c) of the Dutch Financial Supervision Act (Wet op het financieel toezicht) (DFSA), the Board of Directors hereby certifies that, to the best of our knowledge, our consolidated financial statements as of December 31, 2025, prepared in accordance with IFRS as adopted by the EU, and with the legal requirements applicable in the Netherlands, give a true and fair view of the assets, liabilities, financial position and profit or loss of the company and the undertakings included in the consolidation taken as a whole, and that the board report includes a fair review of the development and performance of the business and the position of argenx and the undertakings included in the consolidation taken as a whole, together with a description of the principal risks and uncertainties that they face. Responsibility for this Annual Report The Board of Directors declares that the information contained in this Annual Report, including our consolidated financial statements as of December 31, 2025 and the board report, is, to the best of its knowledge, in accordance with the facts and contains no omission likely to affect its import. The Board of Directors is responsible for the information given in this Annual Report. In Control Statement Our Board of Directors is responsible for the oversight of our risk management activities and has specifically designated the audit and compliance committee (the Audit and Compliance Committee) to assist our Board of Directors in this task and prepare recommendations in this respect to the Board of Directors. While our Board of Directors oversees our risk management, our Senior Management Team is argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Dutch Corporate Governance Code 100

responsible for day-to-day risk management processes. Our Board of Directors expects our Senior Management Team to consider risk and risk management in each business decision, to proactively develop and monitor risk management strategies and processes for day-to-day activities and to effectively implement risk management strategies adopted by the Board of Directors. We believe this division of responsibilities is the most effective approach for addressing the risks we face. 3.2.3 Board of Directors Responsibilities Pursuant to the Dutch Civil Code (DCC), our Board of Directors is collectively responsible for our general affairs. Our Board of Directors, our Executive Director as well as our Non-Executive Directors, define our strategy (as further set out in Section 1.2 “Strategy and objectives”). Our strategy is regularly discussed and monitored at our Board of Directors meetings. Please refer to Section 7.4 “Governance” for more details regarding our governance and oversight with regard to sustainability matters. Pursuant to our Articles of Association, our Board of Directors will divide its duties among its members, with our day-to-day management entrusted to the Executive Director(s). The Non-Executive Directors are tasked with supervising our management and advising the Executive Director(s). In addition, both the Executive Director(s) and the Non-Executive Directors must perform the duties assigned to them pursuant to the Articles of Association. The division of tasks within our Board of Directors is determined (and amended, if necessary) by our Board of Directors. Each director has a duty to properly perform the duties assigned to him or her and to act in our corporate interest. Under Dutch law, the corporate interest extends to the interests of all corporate stakeholders, such as shareholders, creditors, employees and other stakeholders. Composition, Appointment and Dismissal The Articles of Association provide that our Board of Directors will consist of our Executive Director(s) and Non-Executive Directors. The number of Executive Directors must at all times be less than the number of Non-Executive Directors. The number of directors, as well as the number of Executive Directors and Non- Executive Directors, is determined by our Board of Directors, provided that the Board of Directors must consist of at least three members. Our directors are appointed by the General Meeting for a period of four years as either Executive Directors or as Non-Executive Directors. This four-year term aligns with best practice 2.2.1 of the DCGC, which stipulates that executive and Non-Executive Directors may be appointed for a maximum period of four years. We believe that appointing directors for a four-year term, rather than for example annual (re-)appointments, promotes stability and continuity within the Board of Directors. It also allows deserving candidates to be appointed for more than one year, enhancing our position in recruitment processes, as longer appointment periods are generally more attractive to candidates. Additionally, it contributes to the Board of Directors' and, by extension, the Company's ability to focus on long-term goals, in line with the DCGC's principle that a company's strategy should aim for sustainable long-term value creation. In accordance with best practice provision 2.2.1 of the DCGC, Executive Directors may be reappointed for periods not more than four years at a time. In accordance with best practice provision 2.2.2 of the DCGC, Non-Executive Directors may be reappointed once for a period of four years, after which the Non-Executive Director may be reappointed again for a period of two years, which reappointment may be extended by at most two years. In the event of a reappointment after an eight-year period, reasons will be given in the report of the Board of Directors. The Board of Directors is required to make one or more proposals for each seat on our Board of Directors to be filled. A resolution to nominate a director by our Board of Directors (with support from the remuneration and nomination committee (the Remuneration and Nomination Committee) may be adopted by a simple majority of the votes cast. Our Board of Directors conducts evaluations of all its directors and director candidates to create a well- rounded board, designed to promote long-term shareholder value creation through strong leadership and oversight. The Board of Directors recognizes that directors who serve on the board for longer terms can be valuable sources of continuity, understanding of the business and historical insight. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Management Structure 101

Our Board of Directors designates one Executive Director as CEO and may grant other titles to Executive Directors (if appointed). Our Board of Directors also designates a Non-Executive Director as chairperson of the Board of Directors and a Non-Executive Director as vice chairperson of the Board of Directors. The legal relationship between an executive member of the Board of Directors and argenx SE will not be considered as an employment agreement. Employment agreements between an Executive Director and a Group company (other than argenx SE) are permitted. In the absence of an employment agreement, members of a board of directors generally do not enjoy the same protection as employees under Dutch labor law. For a discussion of date of expiration of the current term of office and the period during which the person has served in that office, see Section 3.2.4 “Non-Executive Directors” and Section 3.2.5 “Senior Management Team”. Except for the arrangements described in Section 5.11.2 “Related Party Transactions”, subsection “Agreements with Our Senior Management Team”, there are no arrangements or understanding between us and any of the Executive Directors providing for benefits upon termination of their employment, other than as required by applicable law. In addition, the contracts between us and our Non-Executive Directors do not provide for any benefits upon termination. In addition, the Company is not party to any agreement with a director or employee providing compensation if his or her employment is terminated because of a public takeover offer in respect of the Company. As a foreign private issuer, under the Nasdaq Listing Rules, we are not required to have a majority independent directors on our Board of Directors, except that Audit and Compliance Committee is required to consist fully of independent directors. However, our Board of Directors has determined that, taking into account any applicable committee independence standards, at the date of this Annual Report, all of our Non-Executive Directors, including the members of Audit and Compliance Committee, are “independent directors” under Rule 10A-3 of the Exchange Act and the applicable rules of Nasdaq and of the DCGC. In making such determination, our Board of Directors considered the relationships that each Non-Executive Director has with us and all other facts and circumstances our Board of Directors deemed relevant in determining the director’s independence, including the number of ordinary shares beneficially owned by the director and his or her affiliated entities (if any). The DCGC requires that the composition of Non-Executive Directors is such that the members are able to operate independently and critically vis-à-vis one another, the Executive Directors, and any particular interests involved. As of the date of this Annual Report, all Non-Executive Directors meet the independence criteria contained in the DCGC. Therefore, in the opinion of the Non-Executive Directors, the composition of our Non-Executive Directors complies with the independence requirements of best practice provisions 2.1.7 to 2.1.9 of the DCGC. Our Board of Directors has consequently also determined that all members of our committees are independent under the applicable rules of the DCGC. As of the date of this Annual Report (or in any period before), none of the members of our Board of Directors and Senior Management Team has or has had a family relationship with any other member of our Board of Directors or Senior Management Team. Directors may be suspended or removed by the General Meeting at any time, with or without cause, by means of a resolution passed by a simple majority of the votes cast. Pursuant to the DCC, Executive Directors may also be suspended by the Board of Directors. The suspension of an Executive Director by the Board of Directors may be discontinued by the General Meeting. Diversity In accordance with applicable Dutch legislation, we are required to report annually to the Social Economic Council (Sociaal-Economische Raad) on (i) the gender ratio, i.e., the male and female Executive Directors and Non-Executive Directors, as well as employees in managerial positions at the end of the financial year, (ii) the Company’s self-imposed appropriate and ambitious targets in the form of a target figure to make the ratio between the number of male and female Executive Directors and Non-Executive Directors, as well as in categories of employees in managerial positions to be determined by the Company, more balanced, and (iii) the plan of action to achieve these targets or quotas. If we have not complied with one or more of the foregoing, we are required to report on the reasons for this non-compliance. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Management Structure 102

In accordance with our Diversity, Equity and Inclusion Policy, we aim to foster an inclusive work environment in support of our strategic plan and priorities. We continue to raise the bar in this regard, and to commit to measures and goals designed to support our maturing company culture. We aim to have an equal gender balance in our Board of Directors and in our Company leadership (including functional leaders and project leaders). As of December 31, 2025, our Board of Directors consisted of 9 directors, including 1 Executive Director and 8 Non-Executive Directors. Of the directors who chose to disclose their gender, the Board of Directors contained 5 male directors and 3 female directors (Non-Executive Directors), translating into a 55.56% male/33.33% female balance for our full Board of Directors (compared to 6 males and 3 females (Non- Executive Directors) (60.00%/30.00%) as of December 31, 2024) and a 62.50% male / 37.50% female balance for our Non-Executive Directors (compared to 66.67% male/33.33% female as of December 31, 2024). 90% of our directors in the Board of Directors are independent. In line with our reporting to the Social Economic Council, our annual objective is to attain an equal gender balance (50% male and 50% female) by 2050 in our Board of Directors and in our Company leadership (including functional and project leaders). In 2025, one Non-Executive Director (Anthony Rosenberg) was re-appointed. Although the gender balance was not equal, our Board of Directors nominated Anthony Rosenberg for appointment at the 2025 General Meeting. Our Board of Directors highly valued the skills, knowledge and expertise built up during his career and his contribution and performance as Non-Executive Director. The Board of Directors will take the targets into account for future nomination appointments. As of December 31, 2025, our Company leadership team consisted of 78 persons, comprised of a mix of 39 males and 39 females, (50% / 50% respectively) while 0 positions remained vacant. Compared to 57 persons as of December 31, 2024, comprised of a mix of 24 males and 28 females, (42% / 49% respectively) while 5 positions remained vacant. Our leadership consists of all full time employees reporting directly to our CEO, as well as all (other) leaders of our largest functions and projects. Each of these positions is characterized by a high impact across the organization, leading a global and cross functional team and having a global reach. As of December 31, 2025, 61% of our workforce were female and 39% were male (compared to 58% female and 42% male as of December 31, 2024). Committees In accordance with the DCGC, our Non-Executive Directors can set up specialized committees to analyze specific issues and advise the Non-Executive Directors on those issues and prepare resolutions with respect thereto. The committees are advisory bodies only, and the decision-making remains within the collegial responsibility of the Board of Directors. The Non-Executive Directors determine the terms of reference of each committee with respect to the organization, procedures, policies and activities of the committee. Our Board of Directors has established and appointed (i) an Audit and Compliance Committee; and (ii) the Remuneration and Nomination Committee. The composition and function of these committees complies with all applicable requirements of Euronext Brussels, the DCGC, the Exchange Act, the exchange on which the ordinary shares and the ADSs are listed and U.S. SEC rules and regulations. Only Non-Executive Directors qualify for membership of these committees. The Audit and Compliance Committee and the Remuneration and Nomination Committee may not be chaired by the chairperson of the Board of Directors or by a former Executive Director of the Company. In addition to the aforementioned legally required subcommittees, our Board of Directors may also opt to incorporate informal committees consisting of Non-Executive Directors and other internal and external persons in argenx, in order to facilitate discussions and act as a sounding board on specific projects, as well as on a more permanent basis. Our Board of Directors has incorporated a research and development committee and a commercialization committee. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Management Structure 103

Meetings and decision-making Our Board By-Laws describe, inter alia, the procedure for holding meetings of the Board of Directors, for the decision-making by the Board of Directors and the Board of Directors’ operating procedures. In accordance with our Articles of Association, our Board of Directors meets at least once every three months to discuss the state of affairs within the Company and the expected developments. Under our Board By-Laws, the members of our Board of Directors must endeavor, insofar as is possible, to ensure that resolutions are adopted unanimously. Where unanimity cannot be achieved and Dutch law, the Articles of Association or the Board By-Laws do not prescribe a larger majority, all resolutions of our Board of Directors must be adopted by a simple majority of the votes cast in a meeting at which at least a majority of the members of our Board of Directors then in office are present or represented. The Articles of Association provide that in case of a tie of votes, the chairperson does not have a casting vote and as such the proposal will be rejected in case of a tie. Under the Board By-Laws, some specific matters require approval of the majority of the Non-Executive Directors. These matters are set out in Schedule 1 of our Board By-Laws. Our Board By-Laws are available on our website. The Non-Executive Directors may also determine that certain other matters shall require approval of a certain majority of the Non-Executive Directors. Such matters shall be clearly specified and notified to the Executive Director(s) in writing. Resolutions of the Board of Directors may also be adopted outside of a meeting in writing, provided that all directors in office (in respect of whom no conflict of interest exists as referred to in the Articles of Association) have consented in writing to this manner of decision-making. A director may issue a proxy for a specific Board of Directors meeting to another director in writing. A director having a direct or indirect personal interest that conflicts with the interest of the Company and its affiliated enterprise has a conflict of interest. Each director shall inform all other directors of a conflict of interest without delay. A director shall not participate in the deliberations and decision-making process in relation to an item if he has a conflict of interest with respect thereto. In such case, the other directors shall resolve the item. In case because of this no resolution can be adopted by the Executive Director(s), the Non- Executive Directors will resolve on the matter. In case because of this no resolution can be adopted by the Non-Executive Directors, the Board of Directors will resolve on the matter as if there were no conflict of interest. The Executive Director(s) are required to be asked their vision on their own remuneration in accordance with best practice provision 3.2.2 of the DCGC but may not participate in the adoption of resolutions (including any deliberations in respect of such resolutions) relating to their remuneration. Audit and Compliance Committee Our Audit and Compliance Committee consists of four members: Steve Krognes (chairperson), Peter Verhaeghe, Anthony Rosenberg and James Daly. Our Board of Directors previously established that Peter Verhaeghe, Anthony Rosenberg, James Daly and Steve Krognes satisfy the independence requirements set forth in Rule 10A-3 of the Exchange Act and that Steve Krognes qualifies as “audit committee financial experts” as defined by SEC rules and Article 39 paragraph 1 of Directive 2014/56/EU of the European Parliament and of the Council of April 16, 2014 amending Directive 2006/43/EC on statutory audits of annual accounts and consolidated accounts (which has been laid down in Dutch law by the Decree establishment audit committee (Besluit instelling auditcommissie) and has the requisite financial sophistication under the applicable Nasdaq rules and regulations. Further, our Board of Directors established that the composition of the Audit and Compliance Committee meets the requirements under the Dutch Decree on Establishing Audit Committees. Our Audit and Compliance Committee assists our Board of Directors in overseeing the accuracy and integrity of our accounting, financial and non-financial (including sustainability) reporting processes and audits and reviews of our (consolidated) financial statements as well as non-financial information, the implementation and effectiveness of an internal control system and our compliance with legal and regulatory requirements, the independent auditors’ qualifications and independence and the performance argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Management Structure 104

of the independent auditors. Our Audit and Compliance Committee is also responsible for monitoring the status of, and compliance with, our global ethics and compliance program and meets with the head of our ethics and compliance function at least quarterly to discuss the status and overall effectiveness of the program as well as any issues or incidents that occurred and remedial actions needed (if applicable). The Committee furthermore oversees climate-related risks and supervises the status of the Company’s cybersecurity program and regularly (at least quarterly) discusses the status thereof with management. Our Audit and Compliance Committee is governed by a charter that complies with the Nasdaq Listing Rules and the DCGC and is publicly available on our website. It is responsible for, among other things, establishing methods and procedures for supervising, and where necessary requiring improvements of, our financial reporting, risk management, ethics and compliance and organization for the purpose of making appropriate recommendations to our Board of Directors in that regard. Our Audit and Compliance Committee meets as often as is required for its proper functioning, but at least four times a year and at least once a year meets separately with our independent auditor. Our Audit and Compliance Committee reports regularly to our Board of Directors on the exercise of its functions. It informs our Board of Directors about all areas in which action or improvement is necessary in its opinion and produces recommendations concerning the necessary steps or resolutions that need to be taken. The audit review and the reporting on that review cover us and our subsidiaries as a whole. The members of the Audit and Compliance Committee are entitled to receive all information which they need for the performance of their function, from our Board of Directors and employees. Every member of the Audit and Compliance Committee shall exercise this right in consultation with the chairperson of the Audit and Compliance Committee. Please refer to Section 3.3.5 “Report Audit and Compliance Committee” for an overview of the number of meetings and attendance rates. Remuneration and Nomination Committee We have established a Remuneration and Nomination Committee, which serves as both the remuneration committee and selection and appointment committee as prescribed by the DCGC. Our Remuneration and Nomination Committee currently consists of three members: Ana Céspedes (chairperson), Peter Verhaeghe and Steve Krognes. Donald deBethizy retired from the Board of Directors and the Remuneration and Nomination Committee on May 27, 2025. Consequently, Ana Céspedes succeeded Donald deBethizy as the chairperson of the Remuneration and Nomination Committee and Steve Krognes became a member of the Remuneration and Nomination Committee. Our Remuneration and Nomination Committee is responsible for, among other things: • regularly reviewing the remuneration policy and practices in light of all relevant circumstances and benchmarks, and recommending to the Non-Executive Directors the remuneration of the individual Executive Directors; • advising our Board of Directors in respect of the remuneration for the Non-Executive Directors; • preparing the remuneration report to be included in our annual report; and • drawing up selection criteria and appointment procedures for directors and making proposals for appointment and re-appointment of the directors. The Remuneration and Nomination Committee consists of at least three members. The Remuneration and Nomination Committee meets as often as is required for its proper functioning, but at least once per year to evaluate its functioning. Please refer to Section 3.3.6 “Report Remuneration and Nomination Committee” for an overview of the number of meetings and attendance rates. Informal subcommittees Research and Development Committee The research and development committee consists of members of our Board of Directors and other persons, which composition may vary from time to time. Currently, the research and development committee consists of three members who are also members of our Board of Directors: Brian Kotzin (chairperson), Pamela Klein and Tim Van Hauwermeiren. Non-board member advisors of the research and argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Management Structure 105

development committee include David Lacey, Prof. Hans de Haard, Wim Parys and Mary Lynne Hedley. Ad- hoc participants to the committee meetings include a variety of employees and/or external advisors, depending on the needs of the committee and the topics under discussion. Donald deBethizy retired from the Board of Directors and the Research and Development Committee on May 27, 2025. The research and development committee is responsible for, among other things: • monitoring and overseeing our research and development goals, strategies and measures; • serving as a sounding board to our research and development management, general management and Board of Directors; and • performing strategic reviews of our key research and development programs. The research and development committee also promotes transparency in R&D practices, ensuring that findings, both positive and negative, are reported accurately and openly, and reviews, comments on and makes recommendations in respect of our non-financial reporting on R&D related topics to the Audit and Compliance Committee and/or the Board of Directors. All members of the research and development committee shall have adequate industrial, academic and/or practical experience with the research and development of biopharmaceuticals. Our research and development committee meets as often as is required for its proper functioning, but typically meets at least once prior to each meeting of our Board of Directors and reports regularly to our Board of Directors on the outcome of its deliberations, including any recommendations to the Board of Directors or the Senior Management Team. The chairperson of our research and development committee reports to our Board of Directors on the research and development committee’s discussions and strategic advice after each meeting on all matters within its duties and responsibilities. Please refer to Section 3.3.7 “Report Research and Development Committee” for an overview of the number of meetings and attendance rates. Commercialization Committee Our commercialization committee consists of members of our Board of Directors and other persons, which composition may vary from time to time. As of the date of this Annual Report, the commercialization committee consists of three members: James Daly (chairperson), Anthony Rosenberg and Camilla Sylvest. The commercialization committee is responsible for, among other things: • reviewing and guiding the commercialization strategies and to promote and support innovation within commercialization efforts; • providing guidance on the global product launch strategies, global manufacturing, packaging, labeling and distribution strategies, patient support programs and end-of-life product management; • sales and marketing activities, including engagement of downstreams payors and stakeholders. Our commercialization committee meets as often as is required for its proper functioning and in practice meets at least once per quarter. The commercialization committee reports regularly to our Board of Directors on the outcome of its strategic reviews and any recommendations to the Board of Directors or Senior Management Team. Please refer to Section 3.3.8 “Report Commercialization Committee” for an overview of the number of meetings and attendance rates. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Management Structure 106

3.2.4 Non-Executive Directors Our Board of Directors as at December 31, 2025 comprised the following 8 Non-Executive Directors. As announced on January 5, 2026, there will be a leadership transition in May 2026. Peter Verhaeghe will step down as Non-Executive Director and chairperson of the Board of Directors at the 2026 General Meeting. Tim Van Hauwermeiren will transition from his current CEO role to the position of Non-Executive chairperson of the Board of Directors after receiving approval from the shareholders during the 2026 General Meeting. Peter Verhaeghe Peter Verhaeghe has served as a member and chairperson of the board of arGEN-X B.V. since October 2008 and as Non- Executive Director on our Board of Directors since July 2014. Peter Verhaeghe is the managing partner of VVGB Advocaten- Avocats, a corporate finance law and tax law firm, a position he has held since July 1999. He is currently lead counsel to a number of Belgian, Dutch, French, U.S. and Swiss life sciences companies. Peter Verhaeghe has served on the boards of directors of Participatiemaatschappij Vlaanderen NV since May 2018 and miDiagnostics NV since April 2020. He has also served as chairman of the board of Haretis SA (Luxembourg) since March 2011 and as chairman of the LP & advisory committee of Bioqube Factory Fund I NV since September 2020. Peter Verhaeghe previously served as a member of the board of directors of CzechPak Manufacturing s.r.o., Fujirebio Europe N.V., Tibotec-Virco NV, and Biocartis SA. He was also the president of the board of directors of Merisant France SAS, a member of the management board of Merisant Company 2 S.à. rl., and chairman of the board of directors of PharmaNeuroBoost NV. He holds a degree in law from the University of Leuven and an LL.M. degree from Harvard Law School. Ana Céspedes Ana Céspedes has served as a member of our Board of Directors since December 2022 and as the chairperson of the Remuneration and Nomination Committee since May 2025. Ana Céspedes is the chief executive officer and president of Vitamin Angels, a global health organization dedicated to addressing malnutrition among women and children worldwide. Prior to joining Vitamin Angels, Ana Céspedes served as chief operating officer of the International AIDS Vaccine Initiative, where she oversaw global operations across the United States, Europe, Africa and India. Her responsibilities included strategy and business development, access and commercialization, government affairs and external relations, communications, legal, human resources, finance, administration, business technology, and regional operations. Prior to that, Ana Céspedes held multiple senior leadership roles at Merck KGaA, most recently as Global Head of Strategy and External Engagement, Government and Public Affairs. She also worked as a senior consultant at Arthur Andersen. She currently serves as President of the Scientific Committee of the ProPatiens Institute and has led multiple international initiatives aimed at strengthening collaboration between the pharmaceutical industry and healthcare systems and advancing professional capabilities in market access and corporate affairs. She holds a Bachelor’s degree in Pharmacy and a Ph.D. from the Complutense University of Madrid, a Master in General Management (PDG) from IESE Business School, and an Executive Certificate in Strategy and Innovation from the Massachusetts Institute of Technology. She obtained the NACD.DC™ (National Association of Corporate Directors Directorship Certification®) in July 2024. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Management Structure 107

James Daly James Daly has served as a member of our Board of Directors since May 2018. James Daly currently also serves as a director of Acadia Pharmaceuticals, Inc., Madrigal Pharmaceuticals, Inc. and Cytokinetics, Inc. He was formerly a member of the board of Halozyme Therapeutics, Inc., Bellicum Pharmaceuticals, Inc. and Chimerix, Inc. In 1985, he joined GlaxoSmithKline where he held various positions, including senior vice president of the respiratory division with full responsibility for sales, marketing and medical affairs. James Daly moved to Amgen Inc. in 2001 where he was senior vice president for the North America commercial operations until 2011. In 2012, he joined Incyte Corp, a publicly-traded company focused on oncology and inflammation, where he was chief commercial officer until June 2015. Mr. Daly holds a Bachelor’s of Science and an MBA from the University at Buffalo, State University of New York. Pamela Klein Pamela Klein has served as a member of our Board of Directors since April 2016. Since 2008, Pamela Klein has been a principal and founder of PMK BioResearch, a company offering strategic consulting in oncology drug development to corporate boards, management teams and the investment community. She has also been a venture partner in Ysios Capital Partners, SGIEC, S.A.U. since 2023. She currently serves as a member of the board of directors of several companies including Shasqi, Frontier Medicines Corp and Ona Therapeutics. Pamela KleinPreviously, Pamela Klein served on the board of directors of FStar, Sardona Therapeutics, Patrys Therapeutics, Inc, Jiya Acquisition Corp, and Spring Bank Pharmaceuticals, Inc. until its merger with F-Star Therapeutics in July 2020. Pamela Klein previously spent seven years at the National Cancer Institute as research director of the NCI-Navy Breast Center, after which she joined Genentech as vice president of development until 2001. She also served as chief medical officer for Intellikine, Inc., which was acquired by Takeda American Holdings. She holds a Bachelor’s degree in biology from California State University and an M.D. from Stritch School of Medicine, Loyola University Chicago and is trained in internal medicine and medical oncology.She continues to serve as an advisor for both private and public biotech companies. Brian Kotzin Brian Kotzin has served as a member of our Board of Directors and as a chairperson of our research and development committee since May 7, 2024. He is a former member of the board of directors at Vera Therapeutics, Inc., Rigel Pharmaceuticals, Inc. and Kyverna Therapeutics, Inc. He served as Senior Vice President for Nektar Therapeutics, Inc. from April 2017 to June 2023, and has held various leadership positions at Nektar Therapeutics, Inc., including serving as Chief Medical Officer and Head of Clinical Development from January 2021 to September 2021, again from May 2022 to June 2023, and again from February 2025 to November. He currently is the interim chief medical officer at Nektar Therapeutics, Inc. From 2004 to 2015, Brian Kotzin was argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Management Structure 108

Vice President, Global and Clinical Development and Head, Inflammation Therapeutic Area at Amgen Inc., directing the global development efforts for product candidates in the inflammation area. During his employment at Amgen Inc, he also served as Vice President of Translational Sciences and Head of Medical Sciences from 2006 to 2011. Prior to entering the life sciences industry, Brian Kotzin held several positions as a professor at the University of Colorado Health Sciences Center, where his research focused on immunopathogenesis of inflammatory diseases. He has also held leadership roles at several national organizations, including as a member of the American College of Rheumatology (ACR) Board of Directors, Member and Chairperson of the NIH Immunological Sciences Study Section, Chairperson of the NIH Autoimmunity Centers of Excellence, and Member of the Board of Directors for the Federation of Clinical Immunology Societies. He is currently an elected Master of the ACR. He received an M.D. from Stanford University and a Bachelor’s degree in mathematics from the University of Southern California. Steve Krognes Steve Krognes has served as a member of our Board of Directors and as a chairperson of our Audit and Compliance Committee since February 2023. Steve Krognes also serves on the boards of directors of Guardant Health, Inc., Denali Therapeutics, Inc., and Pliant Therapeutics, Inc. In September 2023, he also was appointed to the board of directors of ClayvstBio and in March 2026 he was appointed to the Board of Alveus Therapeutics Inc. He previously served on the boards of directors of RLS Global AB and Corvus Pharmaceuticals, Inc. and Gritstone Bio, Inc. Steve Krognes was the chief financial officer of Denali Therapeutics, Inc. from 2015 until retiring from that position in April 2022. Steve Krognes led successful financings for Denali Therapeutics, Inc., including its initial public offering in 2017, and contributed significantly to the company’s strategy, growth and strong financial position. His extensive leadership experience in the biotech and pharmaceutical industries includes 12 years in total at Roche and Genentech, Inc., during which Steve Krognes served as chief financial officer of Genentech, Inc. for six years and global head of Roche’s mergers & acquisition team for six years. He also chaired the Genentech Access to Care Foundation and represented Genentech on the board and executive committee of the California Life Science Association. Before that, Steve Krognes worked as an investment banker at Goldman Sachs, as a management consultant at McKinsey & Company, and as a venture capitalist in Scandinavia. Mr. Krognes holds a Master’s of Business Administration (MBA) from Harvard Business School and a Bachelor’s of Science in economics from the Wharton School of the University of Pennsylvania. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Management Structure 109

Anthony Rosenberg Anthony Rosenberg has served as a member of our Board of Directors since April 2017 and as vice-chairperson of our Board of Directors since May 2025. He currently serves as chief executive officer of TR Advisory Services GmbH, his own consultancy firm advising on business development, licensing, and mergers and acquisitions. Anthony Rosenberg also currently serves as chairman of the boards of directors of NUCLIDIUM AG, Oculis SA and Cullinan Therapeutics Inc. Previously Anthony Rosenberg held the positions of Managing Director at MPM Capital, a venture capital firm (2015 until 2020); head of M&A and Licensing of Novartis International (2013 to 2015); and head of business development and licensing at Novartis Pharma (2005 to 2012). Anthony Rosenberg also previously served on the boards of directors of SiO2 Material Science (until March 2023), Radius Health Inc., TriNetX, Inc., iOmx Therapeutics AG, and Clinical Ink, Inc. Mr. Rosenberg has a Bachelor of Science with honors from the University of Leicester and a Master’s of Science in physiology from the University of London. Camilla Sylvest Camilla Sylvest previously served as the executive vice president of commercial strategy and corporate affairs of Novo Nordisk A/S until April 2025. Camilla Sylvest has more than 29 years of working experience at Novo Nordisk A/S during which time she was based in Switzerland, Denmark, Germany, Malaysia, and Mainland China. Over the years, Camilla Sylvest has headed up Novo Nordisk A/S affiliates of growing size and complexity in Europe. She was also corporate vice president of the business area Oceania and Southeast Asia and senior vice president and general manager of the Novo Nordisk A/S region of Mainland China. Camilla Sylvest also served as a member of the board of Danish Crown A/S until September 2025. She holds a Master’s of Science in economics from the University of Southern Denmark and an executive MBA from the Scandinavian Management Institute. Camilla Sylvest has served as a member of our Board of Directors since September 2022. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Management Structure 110
The following table sets forth certain information with respect to the current Non-Executive Directors, including their ages, as at December 31, 2025: Name Age Gender Position Nationality Date of Initial Appointment Date of last (re-) Appointment Term expiration Peter Verhaeghe 67 M Non-Executive Director (chairperson) Belgium October 15, 2008 May 7, 2024 AGM 20261) Ana Céspedes 52 F Non-Executive director Spain December 12, 2022 December 12, 2022 AGM 2026 James Daly 64 M Non-Executive Director U.S. May 8, 2018 May 10, 2022 AGM 2026 Donald deBethizy2) 75 M Non-Executive Director U.S. May 13, 2015 May 2, 2023 AGM 2025 Pamela Klein 64 F Non-Executive Director U.S. April 28, 2016 May 7, 2024 AGM 2026 Brian Kotzin 77 M Non-Executive Director U.S. May 7, 2024 May 7, 2024 AGM 2028 Steve Krognes 57 M Non-Executive Director U.S. and Norway February 27, 2023 February 27, 2023 AGM 2027 Anthony Rosenberg2) 72 M Non-Executive Director (vice- chairperson) UK April 26, 2017 May 27, 2025 AGM 2027 Camilla Sylvest 53 F Non-Executive director Denmark September 8, 2022 September 8, 2022 AGM 2026 1) Peter Verhaeghe will step down as Non-Executive Director and chairperson of the Board of Directors at the 2026 General Meeting. 2) Donald deBethizy retired from the Board of Directors on May 27, 2025. Anthony Rosenberg succeeded him as the vice-chairperson of the Board of Directors. The address for our Non-Executive Directors is our registered office, Laarderhoogtweg 25, 1101 EB Amsterdam, the Netherlands. The following table sets forth the companies and partnerships of which the current Non-Executive Directors have been a member of the administrative, management or supervisory bodies or partner at any time in the previous five years, indicating whether or not the individual is still a member of the administrative, management or supervisory bodies or partner, as of the date of this Annual Report, other than argenx or our subsidiaries: Peter Verhaeghe • VVGB Advocaten – Avocats • Participatiemaatschappij Vlaanderen NV • miDiagnostics NV • Bioqube Factory Fund I • Haretis SA Ana Céspedes • Vitamin Angels • ProPatiens Institute • Living Mindfulness S.L. • International AIDS Vaccine Initiative (IAVI) James Daly • Acadia Pharmaceuticals Inc. • Madrigal Pharmaceuticals, Inc. • Cytokinetics, Inc. • Halozyme Therapeutics, Inc. • Bellicum Pharmaceuticals, Inc. Donald deBethizy • White City Consulting ApS • Cereno Scientific AB • Protteris, Inc. • Lophora ApS • Rigotec GmbH • TME Pharma NV and AG • Saniona AB • Albumedix Ltd. • Asceneuron SA • Albumin Holdings ApS • Innovent LLC Name Current Past argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Management Structure 111
Pamela Klein • Ysios Capital Partners, SGIEC, S.A.U. • Shasqi Inc. • Frontier Medicines Corp. • Ona Therapeutics SL • F-Star Therapeutics, Inc. • Jiya Acquisition Corp. • Sardona Therapeutics, Inc. • I-MAB • Patrys Therapeutics, Inc. Brian Kotzin • Biora Therapeutics, Inc. • Genascence Corporation • Nektar Therapeutics, Inc. • Kyverna Therapeutics, Inc. • Rigel Pharmaceuticals, Inc. Steve Krognes • Denali Therapeutics Inc. • Guardant Health, Inc. • Pliant Therapeutics, Inc. • ClavystBio Pte. Ltd. • Alveus Therapeutics Inc • RLS Global AB • Corvus Pharmaceuticals Inc. • Gritstone Bio, Inc. Anthony Rosenberg • TR Advisory Services GmbH • Cullinan Therapeutics Inc • Oculis SA • NUCLIDIUM AG • SiO2 Material Science Camilla Sylvest • World Diabetes Foundation • World Diabetes Foundation • Novo Nordisk A/S • Danish Crown A/S Name Current Past argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Management Structure 112

3.2.5 Senior Management Team Our Senior Management Team acts as our executive management. Our Senior Management Team comprised of the following persons in 2025 and on the date of this Annual Report (appointment and retirement dates noted as applicable). Of our Senior Management Team, only our CEO, Tim Van Hauwermeiren, is currently part of our Board of Directors as Executive Director. Tim Van Hauwermeiren will transition from his current CEO role to the position of Non-Executive chairperson of the Board of Directors after receiving approval from the shareholders during the 2026 General Meeting. It is also envisaged that Karen Massey, our current COO, will be appointed as an Executive Director at the 2026 General Meeting and subsequently elected as CEO by the Board of Directors. Tim Van Hauwermeiren Tim Van Hauwermeiren co-founded our Company in 2008 and has served as our CEO since July 2008. He has served as a member of our Board of Directors since July 2014. Tim Van Hauwermeiren has almost 31 years of general management and business development experience across the life sciences and consumer goods sectors. He also serves on the board of directors of Lexeo Therapeutics, Inc. and Denali Therapeutics, Inc. Karen Massey Karen Massey has served as our COO since March 2023. Ms. Massey has over 25 years of experience in the pharmaceutical and biotechnology industry, including in commercial, product development, corporate strategy, and innovation roles. Prior to joining argenx, Ms. Massey was with Genentech (Roche Group) for over nine years, where she most recently served as senior vice president of product development and global clinical operations and previously held various commercial leadership roles across marketing and business operations, including as the vice president of the multiple sclerosis and neuromyelitis optica business. Ms. Massey started her biopharmaceutical career in marketing at Pfizer Inc., and returned there, after two years as a management consultant at Bain & Company, to take on leadership positions in corporate strategy and sales and as a commercial lead in Latin America. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Management Structure 113

Karl Gubitz Karl Gubitz has served as our CFO since June 2021. Mr. Gubitz previously worked at Pfizer Inc. for nearly 20 years, most recently as vice president of finance within the global oncology business. Within Pfizer Inc., Mr. Gubitz held country, regional, and global positions, and consistently delivered top-line growth. He managed teams of over 250 colleagues in financial leadership roles within the global internal medicine and global innovative products businesses. Prior to joining Pfizer Inc. in 2003, Mr. Gubitz held various management roles at PricewaterhouseCoopers LLP. Peter Ulrichts Peter Ulrichts has served as our chief scientific officer since January 2023. In this role, he oversees the development of all clinical and pre-clinical compounds within our pipeline. Dr. Ulrichts previously served in various roles at the Company since he joined us in 2010, including, most recently, as our head of clinical science. As a research scientist, Dr. Ulrichts was involved in the development of various therapeutic antibodies for the treatment of cancer and autoimmune diseases. In 2013, he headed the development of our FcRn antagonist efgartigimod until the first-in-human clinical trial. He subsequently transitioned to become the lead scientist of our efgartigimod program. Malini Moorthy Malini Moorthy has served as our general counsel and corporate secretary since February 2022. She has over 26 years of extensive global legal and compliance experience in the biopharmaceutical and medical device industries. She was most recently senior vice president and chief deputy general counsel of legal, compliance, and government affairs at Medtronic plc, where she played a pivotal role in shaping and driving enterprise and functional strategies. Before joining Medtronic plc, Ms. Moorthy spent four years at Bayer Corporation as the head of global litigation and investigations and 10 years at Pfizer Inc., where she progressed to lead civil litigation globally. Ms. Moorthy began her career as a law firm associate, first with McCarthy Tétrault LLP and Genest Murray Desbrisay Lamek LLP in Toronto, Canada and then Salans LLP (now Dentons US LLP) in New York City. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Management Structure 114

Luc Truyen Luc Truyen has served as our chief medical officer since April 2022 and previously served as our head of research and development operations management from September 2021 to April 2022. Prior to this, Dr. Truyen was with Johnson & Johnson (and its subsidiary companies) for over 21 years holding various leadership positions, primarily within neuroscience. In his most recent position prior to joining argenx, Dr. Truyen was global head of development and external affairs for neuroscience, managing the strategy and delivery of the early and late portfolio of assets for mood disorders, schizophrenia, and neurodegenerative and neuroinflammatory disorders. Besides Dr. Truyen’s strong track record in clinical development resulting in several globally innovative drug approvals, his broad-based experience also includes leading global clinical development operations for the whole Johnson & Johnson pharmaceutical group as well as serving as the head of research and development and chief medical officer of Janssen Alzheimer Immunotherapy Research & Development LLC, an internal spin- out from Johnson & Johnson. Arjen Lemmen Arjen Lemmen joined argenx in 2016 and has served as our vice president of corporate development & strategy since 2019. He has successfully executed several transactions including a number of programs within the IIP. Prior to joining the Company, Mr. Lemmen served as a corporate finance specialist at Kempen & Co NV focusing on mergers and acquisitions, equity capital markets and strategic advisory transactions in the European life sciences industry. Andria Wilk Andria Wilk joined argenx as global head of quality in January 2020. Ms. Wilk has more than 26 years of experience in quality assurance within the pharmaceutical industry. Most recently, Ms. Wilk served as senior director, head of medical, regulatory & clinical quality assurance at H Lundbeck A/S, where she managed the global medical, regulatory & clinical quality assurance group based in the EU, U.S., and Asia. In this role, she was responsible for the global audit programs and quality assurance support for all clinical trial and post-marketing activities and related computerized systems. Prior to H Lundbeck A/S, she held various quality assurance positions of increasing responsibility within AstraZeneca plc, Takeda Global Research, Development Centre Europe, and Astellas Pharma Inc. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Management Structure 115
The following table sets forth certain information with respect to the members of our Senior Management Team, including their ages, as of December 31, 2025: Name Age Position Nationality Date of Initial Appointment Tim Van Hauwermeiren1) 53 CEO and Executive Director Belgium July 15, 2008 Karen Massey2) 47 COO Australia March 13, 2023 Karl Gubitz 56 CFO South Africa and U.S. June 1, 2021 Peter Ulrichts 46 Chief Scientific Officer Belgium January 1, 2023 Malini Moorthy 56 General Counsel and Corporate Secretary Canada and U.S. February 14, 2022 Luc Truyen 61 Chief Medical Officer Belgium and U.S. April 1, 2022 Arjen Lemmen 41 Vice-President Corporate Development & Strategy The Netherlands May 1, 2016 Andria Wilk 53 Global Head of Quality UK January 13, 2020 1) Tim Van Hauwermeiren will transition from his current CEO role to the position of Non-Executive chairperson of the Board of Directors after receiving approval from the shareholders during the 2026 General Meeting. 2) It is envisaged that Karen Massey, our current COO, will be appointed as an Executive Director at the 2026 General Meeting and subsequently elected as CEO by the Board of Directors. The address for our Senior Management Team is Industriepark-Zwijnaarde 7, 9052 Zwijnaarde (Ghent), Belgium. The following table sets forth the companies and partnerships of which the members of our Senior Management Team (or persons who have been members of our Senior Management Team in 2025) have been a member of the administrative, management or supervisory bodies or partner at any time in the previous five years, indicating whether or not the individual is still a member of the administrative, management or supervisory bodies or partner, as of the date of this Annual Report, other than argenx or our subsidiaries: Tim Van Hauwermeiren Lexeo Therapeutics, Inc. Aelin Therapeutics NV Denali Therapeutics Inc. RayzeBio, Inc. – Iteos Therapeutics, Inc. Karen Massey – Genentech, Inc. Karl Gubitz – Pfizer Inc. Peter Ulrichts – – Malini Moorthy – Medtronic plc Luc Truyen – Johnson & Johnson Arjen Lemmen OncoVerity Inc. – Andria Wilk European Forum for Good Clinical Practice (EFGCP) – Name Current Past 3.2.6 Conflict-of-Interest and Related Party Transactions Directors must immediately report any (potential) direct or indirect personal interest in a matter that conflicts with the interests of the Company and the business connected with it to the chairperson of our Board of Directors and to the other directors. The Non-Executive Directors will decide, without the director concerned being present, whether there is a conflict of interest. A director will not participate in any discussions and decision making if he or she has a conflict of interest in the matter being discussed. In case because of this no resolution can be adopted by the Executive Directors, the Non-Executive Directors will resolve on the matter. Decisions to enter into transactions in which there are conflicts of interest with directors that are of material significance to us or to the relevant director require the approval of the Non-Executive Directors. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Management Structure 116

Dutch law provides that transactions with related parties are material if (i) information on the transaction qualifies as inside information under the MAR and (ii) such transaction is entered into with one or more holders of shares in the Company representing at least 10% if the issued share capital, or a member of our Board of Directors. Material related party transactions that are not concluded in the ordinary course of business or on normal market terms must be made public by the Company at the time the transaction is entered into, subject to certain exceptions. The Board of Directors has established an internal procedure to periodically assess whether transactions are concluded in the ordinary course of business and on normal market terms. Transactions that are individually non-material, but which are entered into with the same related party during the same fiscal year, must be evaluated in the aggregate to determine if they are material. There are no arrangements or understandings in place with major shareholders, customers, suppliers or others pursuant to which any member of our Board of Directors or Senior Management Team has been appointed. There are no conflicts of interests between the Company and any administrative, management and supervisory bodies and Senior Management Team, nor are there any potential conflicts of interests of the members of our Board of Directors and Senior Management Team between any duties to the Company and their private interests and or other duties. The Board of Directors has established a related party transaction policy in accordance with applicable law. 3.2.7 Code of Business Conduct and Ethics We adopted a Code of Business Conduct and Ethics (Code of Conduct), that is applicable to all of our employees and directors. Our Code of Conduct translates the core values into a set of clear standards to help guide our conduct as we navigate the complexities of the highly regulated and competitive global marketplace in which we operate as we work to become an independent, fully integrated, and global immunology company. Its purpose is not to exhaustively list all the behaviors we as a company expect from each other. Rather, the Code of Conduct contains key principles for us to live by as individuals, to ensure that we maximally contribute to argenx’s collective success. [As part of their onboarding, all employees receive a training on the Code of Conduct and the Directors receive a copy of the Code of Conduct and need to comply with its terms.] The Code of Conduct is available on our website at www.argenx.com/ investors/governance/rules-codes-compliance. The Audit and Compliance Committee of our Board of Directors (i) is responsible for overseeing and evaluating the Code of Conduct and (ii) is required to approve any waivers of the Code of Conduct for employees and directors. The Audit and Compliance Committee has not received requests for material waivers of the Code of Conduct in the financial year 2025. We expect that any amendments to the Code of Conduct, and any waivers of its requirements, will be disclosed on our website. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Management Structure 117
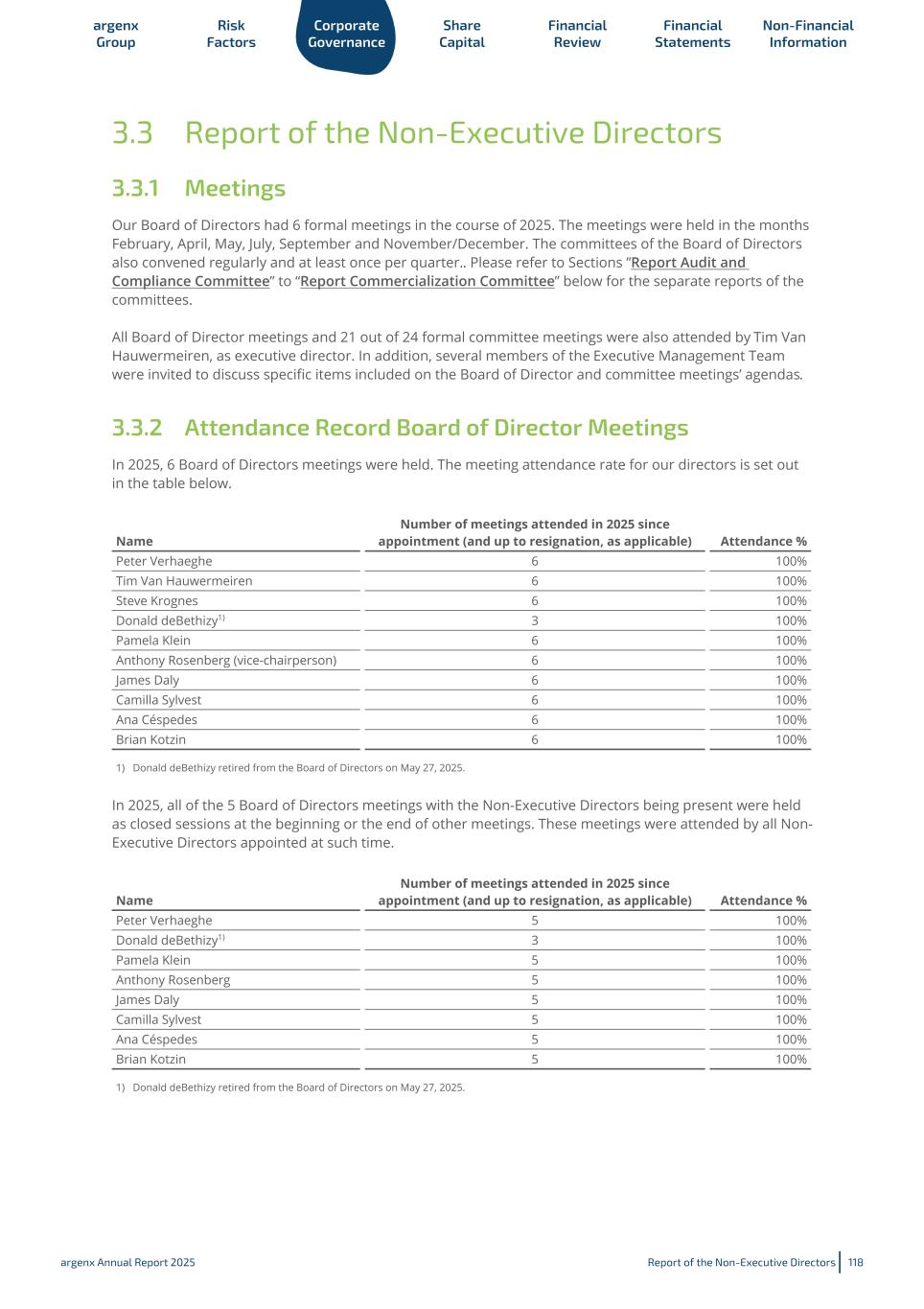
3.3 Report of the Non-Executive Directors 3.3.1 Meetings Our Board of Directors had 6 formal meetings in the course of 2025. The meetings were held in the months February, April, May, July, September and November/December. The committees of the Board of Directors also convened regularly and at least once per quarter.. Please refer to Sections ”Report Audit and Compliance Committee” to “Report Commercialization Committee” below for the separate reports of the committees. All Board of Director meetings and 21 out of 24 formal committee meetings were also attended by Tim Van Hauwermeiren, as executive director. In addition, several members of the Executive Management Team were invited to discuss specific items included on the Board of Director and committee meetings’ agendas. 3.3.2 Attendance Record Board of Director Meetings In 2025, 6 Board of Directors meetings were held. The meeting attendance rate for our directors is set out in the table below. Name Number of meetings attended in 2025 since appointment (and up to resignation, as applicable) Attendance % Peter Verhaeghe 6 100% Tim Van Hauwermeiren 6 100% Steve Krognes 6 100% Donald deBethizy1) 3 100% Pamela Klein 6 100% Anthony Rosenberg (vice-chairperson) 6 100% James Daly 6 100% Camilla Sylvest 6 100% Ana Céspedes 6 100% Brian Kotzin 6 100% 1) Donald deBethizy retired from the Board of Directors on May 27, 2025. In 2025, all of the 5 Board of Directors meetings with the Non-Executive Directors being present were held as closed sessions at the beginning or the end of other meetings. These meetings were attended by all Non- Executive Directors appointed at such time. Name Number of meetings attended in 2025 since appointment (and up to resignation, as applicable) Attendance % Peter Verhaeghe 5 100% Donald deBethizy1) 3 100% Pamela Klein 5 100% Anthony Rosenberg 5 100% James Daly 5 100% Camilla Sylvest 5 100% Ana Céspedes 5 100% Brian Kotzin 5 100% 1) Donald deBethizy retired from the Board of Directors on May 27, 2025. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Report of the Non-Executive Directors 118

3.3.3 Activities The agenda for the Board of Directors centers around the key business objectives for long-term value creation and the key risks involved, as well as the manner in which the Executive Management Team implements our strategy including our research and development pipeline and the commercialization of our products, our culture to ensure proper monitoring by the Non-Executive Directors, our financial position as well as the results of our subsidiaries, significant investment proposals, yearly budgets, the internal risk management and control system, talent development, succession planning and remuneration and appointment matters. In 2025, the Board of Directors discussed a multitude of different themes. The Board of Directors primarily supervised and discussed the Company's innovation mission and objectives towards our long-term strategic Vision 2030, reviewed the scientific pipeline and regulatory developments for all product candidates in several geographies, ensuring the required progression thereof. The Board of Directors furthermore reviewed and discussed our strategy for manufacturing processes, our supply chain and identified potential corresponding risks following the geopolitical environment. The Board also focused on the commercialization strategies and opportunities, contributing to our successful product launches, including the launch of the PFS in 2025, supporting innovation within commercialization efforts. The Board spent a significant amount of time on themes around cybersecurity and AI, including on NIS2 Directive requirements. In line with previous years, the Board of Directors also discussed talent development and succession planning, both for the senior leaders within the Company (within and beyond the Executive Management Team) and the Board of Directors. This led to Brian Kotzinthe renewal of the appointment of Anthony Rosenberg as Non-Executive Director and the attraction of several new members in the Company’s broader leadership team. Time was also spent discussing and evaluating the Company's strategy surrounding the continued rapid growth and the measures undertaken to preserve our unique company culture. Finally, the Board of Directors spent a lot time discussing shareholder feedback on our journey on getting the remuneration policy approved during the 2025 Extraordinary General Meeting. 3.3.4 Board Evaluation The Board of Directors evaluates its functioning and the functioning of its committees and of each individual director annually. The evaluation process is performed with the help of an external professional board evaluation consultant. In 2025, the evaluation was performed by Nasdaq Center for Board Excellence. The evaluation includes preparing specific questionnaires focusing on the skills and competences most relevant to us, and the most material board topics and challenges we face. The written questionnaire is then followed up by one-to-one interviews with the representative of Nasdaq Center for Board Excellence with each member of the Board of Directors, followed by a debrief and discussion held with the external evaluator and the entire Board of Directors both in writing (in form of a report) and in the form of a live discussion of the evaluation report aimed at distilling specific learnings and conclusions. Based on the self-evaluation performed, the Non-Executive Directors concluded that the Board of Directors and its committees had properly discharged their responsibilities during 2025. The Board of Directors identified certain strengths and weaknesses and adopted a plan for further board development and succession in 2026. All directors consider the Board of Directors, in line with previous years to have high integrity with a continued commitment to high quality governance and a shared desire to continuously improve the Board of Directors. The importance to preserve this was highlighted by Nasdaq Center for Board Excellence. All Non-Executive Directors consider fostering further development and education of great importance, which can be developed in 2026 through advisory board sessions, deep-dives and other external educational courses. Lastly, all Non-Executive Directors will continue discussions on Board of Directors and the Executive Management team succession and focus on further developing profiles for future Board of Director candidates. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Report of the Non-Executive Directors 119

3.3.5 Report Audit and Compliance Committee The Audit and Compliance Committee reports regularly to our Board of Directors on the exercise of its functions. It informs our Board of Directors about all areas in which action or improvement is necessary in its opinion and produces recommendations concerning the necessary steps that need to be taken. The audit review and the reporting on that review cover the Company and its subsidiaries as a whole and the external auditor is also present during these meetings. In 2025, the main topics of discussion at the meetings were the 2024 consolidated financial statements and press release as well as interim consolidated financial statements and press releases, internal audit and external auditors’ reports, the review of quarterly forecasts and financial plan, tax updates, cash management, compliance with CSRD and updating the double materiality assessment), the Company’s ethics and compliance program, the Company’s cyber security program and risks related to AI, the Company’s privacy program and the certification of the recently included VOR (Verklaring Omtrent Risicobeheersing/statement on risk management) statement in the DCGC. In 2025, 8 formal Audit and Compliance Committee meetings were held as well as multiple informal meetings. The formal meeting attendance rate for our Non-Executive Directors is set out in the table below. Name Number of meetings attended in 2025 since appointment Attendance % Steve Krognes (chairperson) 8 100% Peter Verhaeghe 7 87.5% Anthony Rosenberg 8 100% James Daly 7 87.5% 3.3.6 Report Remuneration and Nomination Committee The Remuneration and Nomination Committee assists the Board of Directors by, amongst other matters, regularly reviewing our remuneration policy, preparing remuneration proposals and periodically assessing the size and composition of the Board of Directors and the Executive Management Team and development of talent throughout the Company. During their deliberations in 2025, the main topics of discussion at the meetings were long-term succession and development planning for key Company leadership and proposing appropriate remuneration policies during the annual general meeting (AGM) held on May 27, 2025 (the 2025 General Meeting) and the EGM, taking into account stakeholder feedback following extensive engagement efforts and benchmarking all remuneration against our peer group. In 2025, 6 formal Remuneration and Nomination Committee meetings were held as well as multiple informal meetings. The formal meeting attendance rate for our Directors is set out in the table below. Name Number of meetings attended in 2025 since appointment (and up to resignation or since joining, as applicable) Attendance % Ana Céspedes1) 6 100% Donald deBethizy2) (chairperson until May 27, 2025) 3 100% Peter Verhaeghe 6 100% Steve Krognes3) 3 100% 1) Ana Céspedes succeeded Donald deBethizy as chairperson of the Remuneration and Nomination Committee effective May 27, 2025. 2) Donald deBethizy retired from the Board of Directors and the Remuneration and Nomination Committee on May 27, 2025. 3) Steve Krognes joined the Remuneration and Nomination Committee effective May 27, 2025. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Report of the Non-Executive Directors 120

3.3.7 Report Research and Development Committee The research and development committee functions as a sounding board to our research and development management, the Executive Management Team and the Board of Directors, and monitors our research and development goals, strategies and measures. In 2025, the committee held 5 formal meetings, in which it focused mainly on the vision and strategy on science, the Company’s research and development pipeline including its preclinical and clinical stage product-candidates, potential future indications for its commercial stage products and developments in relation to our IIP. The meeting attendance rate for our directors is set out in the table below. Name Number of meetings attended in 2025 since appointment (and up to resignation or since joining, as applicable) Attendance % Brian Kotzin (chairperson) 5 100% Donald deBethizy1) 2 100% Pamela Klein 5 100% Tim Van Hauwermeiren2) 5 100% 1) Donald deBethizy retired from the Board of Directors and the Research and Development Committee on May 27, 2025. 2) Tim Van Hauwermeiren joined the Research and Development Committee as a member as on May 27, 2025. 3.3.8 Report Commercialization Committee The commercialization committee functions as a sounding board on branded and unbranded strategic marketing plans for the Board of Directors. In 2025, the committee held 5 formal meetings, in which it focused mainly on the continued commercialization efforts of VYVGART in gMG and CIDP, the execution of our launch of the PFS in the U.S. as well as the preparation for potential future launches, subject to obtaining further approvals. The meeting attendance rate for our directors is set out in the table below. Name Number of meetings attended in 2025 since appointment Attendance % James Daly (chairperson) 5 100% Anthony Rosenberg 5 100% Camilla Sylvest 5 100% 3.4 Remuneration Report and Compensation Statement 3.4.1 Letter from the Chairperson of the Remuneration and Nomination Committee Dear Stakeholders, Introduction On behalf of the Remuneration and Nomination Committee, I am pleased to present the 2025 remuneration report (the 2025 Remuneration Report), which outlines the Committee’s activities over the past year and provides insight into how the Company’s achievements and continued progress in 2025 have shaped the remuneration of our CEO and Board of Directors. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Report of the Non-Executive Directors 121

In compliance with article 2:135b of the Dutch Civil Code, the European Shareholder Rights Directive and the DCGC, this 2025 Remuneration Report discloses how we implemented our 2021 Remuneration Policy for the Board of Directors and CEO in 2025 and explains how we will move forward with the 2025 Remuneration Policy, which became effective upon receiving shareholder approval at the 2025 Extraordinary General Meeting, held on November 18, 2025. In so doing so we hope that we can demonstrate the CEO is not only rewarded for immediate achievements, but also for sustained progress in our business strategies, individual objectives, and key strategic non-financial metrics that we believe underpin our long-term mission and strategy. argenx performance in 2025 2025 was a year of strong execution and strategic delivery across argenx’ core priorities. We made substantial progress toward our Vision 2030 ambition of reaching 50,000 patients globally, treating approximately 19,000 patients across three indications (gMG, CIDP and ITP) and three product presentations. We successfully launched the VYVGART Hytrulo pre-filled syringe in MG and CIDP, which expanded access to new patient segments and enabled more convenient treatment options. At the same time, the Company continued to invest in long-term value creation by advancing a robust and diversified pipeline, entering 2026 with 10 ongoing registrational clinical trials and adding four new molecules to the pipeline. Our Senior Management Team, including the CEO, CFO and COO (Named Executive Officers or NEOs), guided the organization through a year of operational intensity, progressing late-stage clinical programs, supporting regulatory submissions, scaling commercial operations and building the infrastructure required for sustainable growth, while positioning argenx for its next phase of leadership and innovation. In gMG, we strengthened our position as the #1 prescribed and fastest-growing biologic, supported by increasingly earlier use in the treatment paradigm and continued expansion into broader patient populations. Positive topline data from the Seronegative gMG clinical trial strengthened our ambition to be the treatment of choice and to pursue the broadest MG label to date. In CIDP, real-world outcomes continued to validate the ADHERE results, with physicians reporting sustained functional improvement and patients experiencing greater independence and quality of life. This strong commercial execution resulted in a historic milestone for the company, with VYVGART surpassing $1 billion in product net sales in a single quarter for the first time in the third quarter. Fixed base pay After our annual comprehensive base pay review, in 2025 the CEO, CFO and COO’s base pay increased from EUR 700,000 to EUR 732,000 ($827,160), $553,000 to $578,000 and CHF 594,000 to CHF 615,000 ($741,641), which is an increase of 4.6%, 4.5% and 3.5% respectively. These increases are determined in accordance with the Company’s global base pay increase principles and guidelines and are consistent with the methodology used to determine base pay increases for employees across the organization. In addition, they reinforce our commitment to a balanced, performance-driven remuneration structure that supports sustainable long-term value creation, while maintaining fairness and transparency and taking into account annually performed scenario analyses, including benchmarking exercises in setting total remuneration levels. Even with the above-referenced increase, the CEO’s base pay, at his own request, remained below the 25th percentile of the 2024 peer group. Short Term Performance Pay The CEO, CFO and COO delivered strong performance outcomes in 2025, resulting in STI payouts of 150% of target for the CEO, 150% for the CFO and 175% for the COO. These outcomes reflect the successful launch of the pre-filled syringe on April 10, 2025, associated revenue growth and continued delivery against key strategic priorities. The Remuneration and Nomination Committee considers that these results accurately reflect performance against the targets set at the start of the year and demonstrate clear alignment between pay and performance. Further details on individual achievements are provided in the main body of the report. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Remuneration Report and Compensation Statement 122

Long Term Performance Pay Over 2025, argenx’s share price rose by 20% from €600.00 to €716.80. In fact, over a three-year period, from December 31, 2022 to December 31, 2025, the share price has risen by approximately 106%, from €348.30 to €716.80 per share, further underscoring the NEOs successful long-term performance. Under the 2021 Remuneration Policy, as part of the long-term incentive pay (LTIP), the NEOs received RSUs and stock option grants. For disclosure on these awards vesting in 2025, please refer to ‘‘Section 3.4.3 LTIP’’ of this 2025 Remuneration Report. Under the 2025 Remuneration Policy approved in November 2025, RSUs have been replaced by PSUs. As a result, the LTIP is now fully ‘at-risk’ as it is entirely performance-based and a mixture of stock options (no more than 50% of the annual grant) and PSUs (at least 50% of the annual grant). For the PSU portion of the 2025 grant, they will be assessed on the following performance metrics and targets: • Maximize the VYVGART opportunity consisting of the following targets: 2027 annual revenue (50%) and gMG Label Expansion (15%); • Build a portfolio of breakthrough antibody-based products consisting of the following target: FDA submissions (15%); • Ensure long-term sustainability as an independent company consisting of the following target: Pipeline progression (10%); and • Scaling the argenx way consisting of the following target: talent retention (10%). For further information on the 2026 PSU awards, please refer to ‘‘Section 3.4.3 LTIP - PSUs’’ of this 2025 Remuneration Report. Stakeholder Engagement Shareholders play a crucial role in our success by providing invaluable support and fostering strong partnerships that are essential to our growth. We deeply appreciate their continued commitment and strive to keep them well-informed, ensuring a lasting and productive relationship. During 2025, we directly engaged with our top 60 shareholders and other shareholders who previously engaged with us (holding a total of approximately 70% of our shares) as well as with proxy advisors, resulting in strong shareholder endorsement, with approximately 96% of votes cast in favor of the 2025 Remuneration Policy during the 2025 Extraordinary General Meeting held on November 18, 2025, reflecting broad support for the Company’s approach to executive remuneration. Looking Forward As announced on January 5, 2026, our current CEO, Tim Van Hauwermeiren, will transition from his CEO role to the position of Non-Executive chairperson of the Board of Directors, subject to receiving approval from the shareholders during the 2026 General Meeting. I would like to express the Committee’s immense appreciation for his great leadership, contributions and accomplishments delivered since the Company’s founding in 2008. The Board of Directors will appoint our current COO, Karen Massey, as our new CEO, following the approval of her nomination to Executive Director by the shareholders during the 2026 General Meeting. We are delighted to receive overwhelmingly positive feedback from shareholders since the announcement of the leadership transition and strong support for both Tim Van Hauwermeiren and Karen Massey’s new roles. Tim Van Hauwermeiren’s anticipated CEO remuneration for 2026 will be in accordance with the provisions of the 2025 Remuneration Policy and is summarized below: • Treatment of base pay: The annual base pay for 2026 will be paid, pro-rated, up to and including the date of his resignation as CEO at the 2026 General Meeting. • Treatment of 2026 STI: The 2026 STI will be paid on a pro-rated basis up to and including the date of his resignation as CEO at the 2026 General Meeting, in accordance with the Founder CEO legacy agreement. Full details will be disclosed in the 2026 Remuneration Report. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Remuneration Report and Compensation Statement 123

• Treatment of LTI: All unvested equity other than PSUs will immediately and fully vest at the time of the 2026 General Meeting. The PSUs granted in 2025 will remain subject to their normal three-year performance period and will vest in 2027, based on performance and pro-rated for time served as CEO in calendar years 2025 and 2026 (up to and including the date of his resignation as CEO at the 2026 General Meeting). Tim Van Hauwermeiren will not receive a new pro-rated 2026 LTIP grant, including both stock options and PSUs. Subject to approval by the shareholders at the 2026 General Meeting, Tim Van Hauwermeiren’s remuneration arrangements as Non-Executive chairperson of the Board of Directors will be in accordance with the 2025 Remuneration Policy. For more information regarding Tim Van Hauwermeiren’s 2026 compensation as CEO and as Non-Executive chairperson of the Board of Directors, including when details on achievement and pay-out will be available, please refer to section ‘‘Section 3.4.3 LTIP - Looking Forward’’ below. More information on Karen Massey’s remuneration will be disclosed in the convocation notice for the 2026 General Meeting, which will become available on or around the date of this 2025 Remuneration Report. I share the enthusiasm and excitement expressed by our shareholders about the upcoming leadership transition, and I am confident that with Tim Van Hauwermeiren as the Non-Executive chairperson of the Board of Directors and Karen Massey as CEO, argenx is extremely well-positioned for the long term. We will continue to engage actively on this leadership transition and other relevant topics with our key stakeholders and proxy advisors throughout 2026 and beyond. We remain available to address any questions regarding corporate governance and executive compensation. On behalf of the Remuneration and Nomination Committee I would like to thank all our investors, employees and stakeholders for their continued support and I ask that shareholders vote to approve this report at the 2026 General Meeting. Ana Céspedes Chairperson, Remuneration and Nomination Committee 3.4.2 Remuneration Policy Summaries for each approved policy, which guided our backward-looking and forward-looking pay decisions during the year-under-review are below. For more details on these policies, please refer to the below links provided • The 2021 Remuneration Policy: https://argenx.com/content/dam/argenx-corp/media-documents/ argenx_remuneration_policy_final_approved_11_May_2021.pdf. • The 2025 Remuneration Policy: https://argenx.com/content/dam/argenx-corp/media-documents/ EGM_November_2025-Remuneration_Policy.pdf. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Remuneration Report and Compensation Statement 124
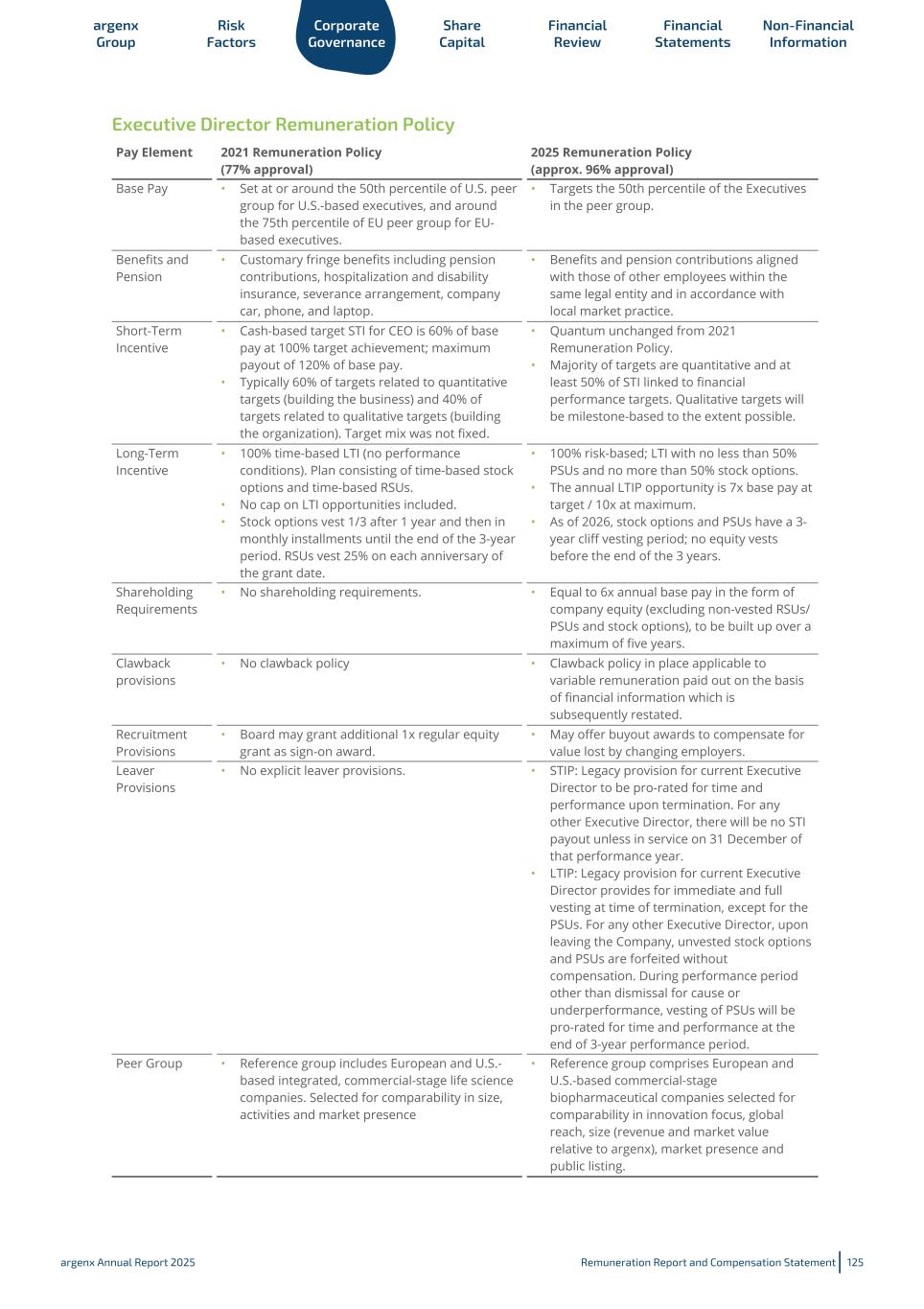
Executive Director Remuneration Policy Pay Element 2021 Remuneration Policy (77% approval) 2025 Remuneration Policy (approx. 96% approval) Base Pay • Set at or around the 50th percentile of U.S. peer group for U.S.-based executives, and around the 75th percentile of EU peer group for EU- based executives. • Targets the 50th percentile of the Executives in the peer group. Benefits and Pension • Customary fringe benefits including pension contributions, hospitalization and disability insurance, severance arrangement, company car, phone, and laptop. • Benefits and pension contributions aligned with those of other employees within the same legal entity and in accordance with local market practice. Short-Term Incentive • Cash-based target STI for CEO is 60% of base pay at 100% target achievement; maximum payout of 120% of base pay. • Typically 60% of targets related to quantitative targets (building the business) and 40% of targets related to qualitative targets (building the organization). Target mix was not fixed. • Quantum unchanged from 2021 Remuneration Policy. • Majority of targets are quantitative and at least 50% of STI linked to financial performance targets. Qualitative targets will be milestone-based to the extent possible. Long-Term Incentive • 100% time-based LTI (no performance conditions). Plan consisting of time-based stock options and time-based RSUs. • No cap on LTI opportunities included. • Stock options vest 1/3 after 1 year and then in monthly installments until the end of the 3-year period. RSUs vest 25% on each anniversary of the grant date. • 100% risk-based; LTI with no less than 50% PSUs and no more than 50% stock options. • The annual LTIP opportunity is 7x base pay at target / 10x at maximum. • As of 2026, stock options and PSUs have a 3- year cliff vesting period; no equity vests before the end of the 3 years. Shareholding Requirements • No shareholding requirements. • Equal to 6x annual base pay in the form of company equity (excluding non-vested RSUs/ PSUs and stock options), to be built up over a maximum of five years. Clawback provisions • No clawback policy • Clawback policy in place applicable to variable remuneration paid out on the basis of financial information which is subsequently restated. Recruitment Provisions • Board may grant additional 1x regular equity grant as sign-on award. • May offer buyout awards to compensate for value lost by changing employers. Leaver Provisions • No explicit leaver provisions. • STIP: Legacy provision for current Executive Director to be pro-rated for time and performance upon termination. For any other Executive Director, there will be no STI payout unless in service on 31 December of that performance year. • LTIP: Legacy provision for current Executive Director provides for immediate and full vesting at time of termination, except for the PSUs. For any other Executive Director, upon leaving the Company, unvested stock options and PSUs are forfeited without compensation. During performance period other than dismissal for cause or underperformance, vesting of PSUs will be pro-rated for time and performance at the end of 3-year performance period. Peer Group • Reference group includes European and U.S.- based integrated, commercial-stage life science companies. Selected for comparability in size, activities and market presence • Reference group comprises European and U.S.-based commercial-stage biopharmaceutical companies selected for comparability in innovation focus, global reach, size (revenue and market value relative to argenx), market presence and public listing. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Remuneration Report and Compensation Statement 125

Non-Executive Director Remuneration Policy Pay Element 2021 Policy (77% approval) 2025 Policy (96% approval) Cash • Benchmarked regularly fees could be adjusted as necessary based on regular benchmarking exercises to ensure continued fair and competitive remuneration. • Annual cash retainer fee targets the 50th percentile of the peer group and benchmarked annually. 2025 Board of Directors membership fee: $60k per year, with additional $59.5k for the chairperson. Committee members receive (depending on the committee) $10,000-12,500 for membership or $20,000-25,000 for chairmanship. Equity • Share options and/or restricted share units in an amount that is at or around the 50th percentile of the U.S. companies in our reference group. Per 2021, we granted 2,700 stock options and 600 RSUs to each Non- Executive Director. Equity granted was adjusted on the basis of benchmark outcomes and in consideration of developments in the composition of equity incentives offered by argenx to key persons outside the Board of Directors, including company employees. • RSUs vest over 4 years and stock options vesting over 3 years. • Since 2024, stock options no longer granted to Non-Executive Directors to avoid any perceived effect on independence; switched to granting RSUs. • Annual equity grant of $400,000 in the form of restricted shares with no vesting conditions, targeting the 50th percentile of the peer group and subject to annual review. No shares may be sold until after the 4th anniversary of the grant date, except to the extent necessary to cover immediate tax obligations resulting from the vest. • Restricted shares are not subject to vesting conditions. 4-year holding requirement for equity grants (except sales to cover immediate tax obligations). Shareholding Requirements • No shareholding requirements. • Holding of 5x annual Board of Director membership retainer fees (as at the date of the 2025 Remuneration Policy, the annual retainer fee was $60,000), to be built over a period of 5 years. Special travel allowance • No special travel allowance. • Special travel allowance of $5,000 for in- person attendance at each board meeting held outside of a Non-Executive Director’s official continent of residence. Reference Peer Group Selection Criteria The argenx peer group is based on the following selection criteria: • Sector (Biopharmaceutical companies, excluding diagnostics and animal health companies); • Innovation focus (at least 25% of revenue is spent on R&D); • Global reach (generates product revenues both within and outside the US); • Revenue (1/4x – 4x of our annual revenue); • Market capitalization (1/4x – 4x of our 30-day average market cap on the last business day prior to the date of the Company-wide grant in June); and • Listing location (listed on a major US Stock Exchange). If there are fewer than 15 companies meeting all six criteria, the peer list will be supplemented with companies that meet all but one criterion with the least relevant criterion dropped first, in the order as displayed above (from most to least relevant). For the 2025 Peer Group that was selected in Q3 2025, we made adjustments to our benchmarking methodology. In response to shareholder feedback, we shifted the US listing criterion from the second argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Remuneration Report and Compensation Statement 126

most important criterion to the criterion with the least significance. This resulted in an increase from 26.67% European peers in the 2025 Peer Group to 40% European peers in the 2025 Peer Group. The 2025 Peer Group was composed of the following global reference companies • US Companies: Alnylam Pharmaceuticals, Biogen, BioMarin Pharmaceutical, Incyte, Insmed, Moderna, Regeneron Pharmaceuticals, Sarepta Therapeutics, Vertex Pharmaceuticals. • European Companies: Ascendis Pharma, BeOne Medicines, BioNTech, Genmab, Jazz Pharmaceuticals, UCB. Each year, the Remuneration and Nomination Committee will validate the peer group to ensure its relevance and the Remuneration and Nomination Committee may recommend adjustments to the Board of Directors, if deemed necessary. 3.4.3 NEO Remuneration in FY25 Base pay After our annual comprehensive base pay review, the CEO, CFO and COO’s base pay increased from EUR 700,000 to EUR 732,000 ($827,160), $553,000 to $578,000 and CHF 594,000 to CHF 615,000 ($741,641), which is an increase of 4.6%, 4.5% and 3.5% respectively. These increases are determined in accordance with the Company’s global base pay increase principles and guidelines and are consistent with the methodology and base pay increases for employees across the organization. In addition, they reinforce our commitment to a balanced, performance-driven remuneration structure that supports sustainable long- term value creation, while maintaining fairness and transparency and taking into account annually performed scenario analyses including benchmarking exercises in setting total remuneration levels. Pension and fringe benefits The benefits paid to the NEOs are jurisdiction dependent. For the CEO, these included benefits customary in the Belgian market, and which are standard components of Belgian-based employee packages: pension contributions, a hospitalization insurance, a representation allowance and a company car. The Company pension contribution percentage of base pay for the CEO is equal to the Company pension contribution percentage for all employees in Belgium. For the COO, these included benefits customary in the Swiss market, and which are standard components of Switzerland-based employee packages: car allowance, lunch allowance, health insurance allowance, representation allowance and pension contributions. For the CFO, these included benefits customary in the U.S. market, and which are standard components of our U.S.- based employee packages: a company-administered health benefit plan and 401k plan. Short Term Incentive Pay The CEO, CFO and COO participated in the 2025 STIP with target opportunities of 60%, 40% and 50% of base pay, and maximum opportunities of 120%, 80% and 100% of base pay, respectively. In assessing performance against the Company’s 2025 business plan, the Board determined that the NEOs delivered a strong year of execution, resulting in STIP outcomes of 150% of target for the CEO and CFO and 175% for the COO. Specifically for the CEO, he delivered strong performance against the measures. He exceeded the annual operating budget revenue targets, successfully executing the PFS launch in the U.S., resulting in achieving the maximum opportunity for the revenue goal. Target achievement was reached for the pipeline goal as the MG combination clinical trial was launched and 3 new candidates were nominated and 3 new molecules graduated. The Innovation goal was also met at target through championing key innovation projects and embedding innovation goals into the performance targets of employees. Lastly, succession plans for key senior leaders as well as the CEO succession were accomplished at target for scaling the argenx way goal to secure long-term leadership strength & organizational capabilities. The overall strong performance resulted in a total weighted achievement of 150% of target opportunity. Specifically for the CFO, he delivered a strong performance against the measures. He also exceeded the annual operating budget revenue targets, successfully executing the PFS launch in the US, resulting in achieving the maximum opportunity for the revenue goal. Maximum opportunity was also achieved for argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Remuneration Report and Compensation Statement 127

making substantial progress on the digital finance transformation goal, including reductions in financial closing timelines, automation of financial processes and transforming the planning cycle. Target achievement was reached for the P&L goal by effectively managing the Company’s tax commitments and managing headcount growth within the approved budget parameters, supporting disciplined scaling for scaling the argenx way goal. The overall strong performance resulted in a total weighted achievement of 150% of target opportunity. Specifically for the COO, she delivered a very strong performance against the measures. Just like the CEO and CFO, she exceeded the annual operating budget revenue targets, successfully executing the PFS launch in the US, resulting in achieving the maximum opportunity for both the revenue and pipeline acceleration goals. Maximum opportunity was also achieved for operational and digital transformation priorities, including the successful onboarding of the BIS leaders and execution of key transformation objectives. Lastly, critical leadership hires were successfully onboarded and operational excellence initiatives were embedded to accelerate novel therapies to patients, resulting in target achievement for scaling the argenx way goal. The overall very strong performance resulted in a total weighted achievement of 175% of target opportunity. The Board considers the selected performance metrics to remain the most appropriate measures of NEO performance, as they reflect the Company’s key strategic, operational and financial priorities. The tables below outline the targets set and the corresponding achievements for each NEO. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Remuneration Report and Compensation Statement 128
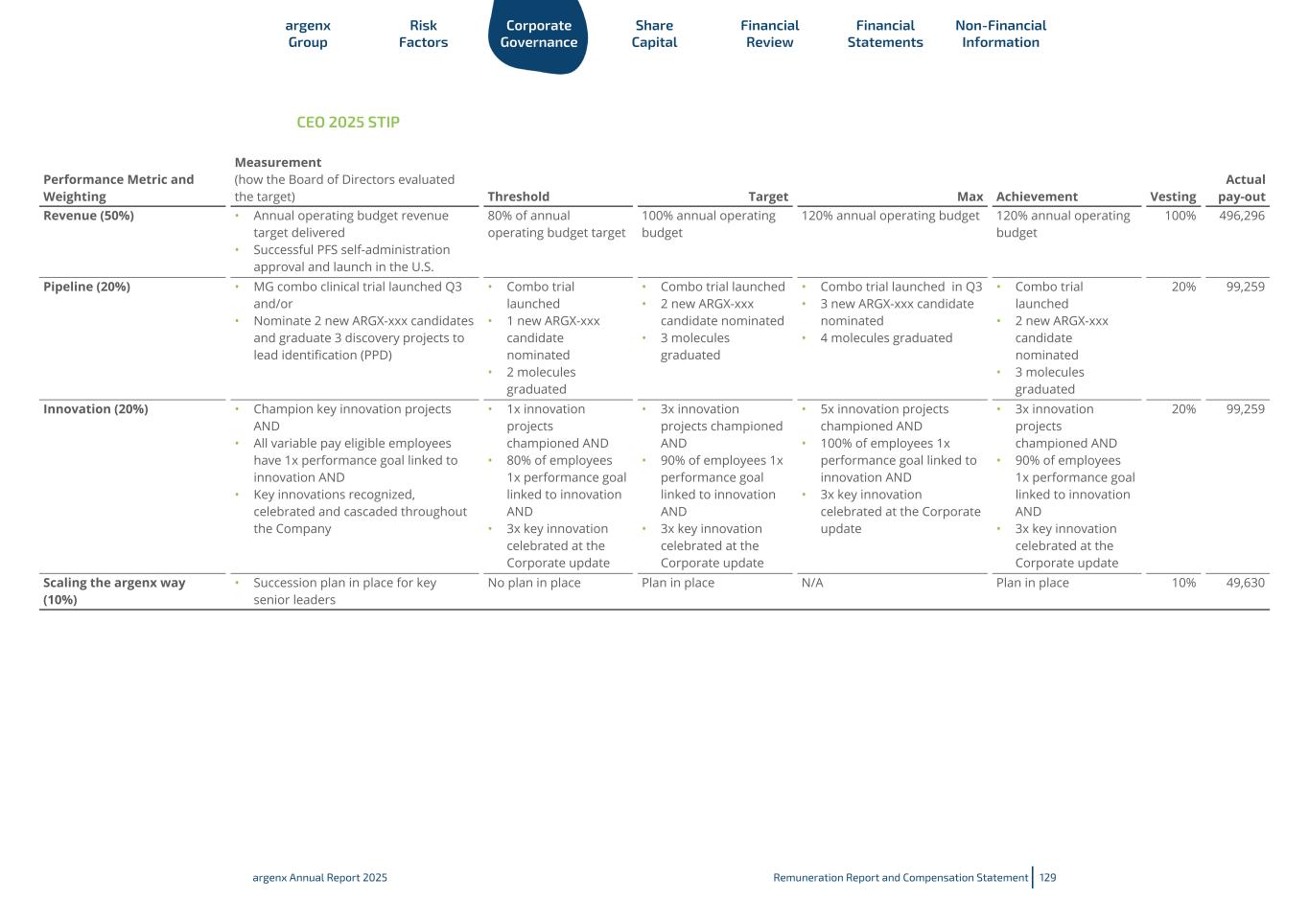
CEO 2025 STIP Revenue (50%) • Annual operating budget revenue target delivered • Successful PFS self-administration approval and launch in the U.S. 80% of annual operating budget target 100% annual operating budget 120% annual operating budget 120% annual operating budget 100% 496,296 Pipeline (20%) • MG combo clinical trial launched Q3 and/or • Nominate 2 new ARGX-xxx candidates and graduate 3 discovery projects to lead identification (PPD) • Combo trial launched • 1 new ARGX-xxx candidate nominated • 2 molecules graduated • Combo trial launched • 2 new ARGX-xxx candidate nominated • 3 molecules graduated • Combo trial launched in Q3 • 3 new ARGX-xxx candidate nominated • 4 molecules graduated • Combo trial launched • 2 new ARGX-xxx candidate nominated • 3 molecules graduated 20% 99,259 Innovation (20%) • Champion key innovation projects AND • All variable pay eligible employees have 1x performance goal linked to innovation AND • Key innovations recognized, celebrated and cascaded throughout the Company • 1x innovation projects championed AND • 80% of employees 1x performance goal linked to innovation AND • 3x key innovation celebrated at the Corporate update • 3x innovation projects championed AND • 90% of employees 1x performance goal linked to innovation AND • 3x key innovation celebrated at the Corporate update • 5x innovation projects championed AND • 100% of employees 1x performance goal linked to innovation AND • 3x key innovation celebrated at the Corporate update • 3x innovation projects championed AND • 90% of employees 1x performance goal linked to innovation AND • 3x key innovation celebrated at the Corporate update 20% 99,259 Scaling the argenx way (10%) • Succession plan in place for key senior leaders No plan in place Plan in place N/A Plan in place 10% 49,630 Performance Metric and Weighting Measurement (how the Board of Directors evaluated the target) Threshold Target Max Achievement Vesting Actual pay-out argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Remuneration Report and Compensation Statement 129
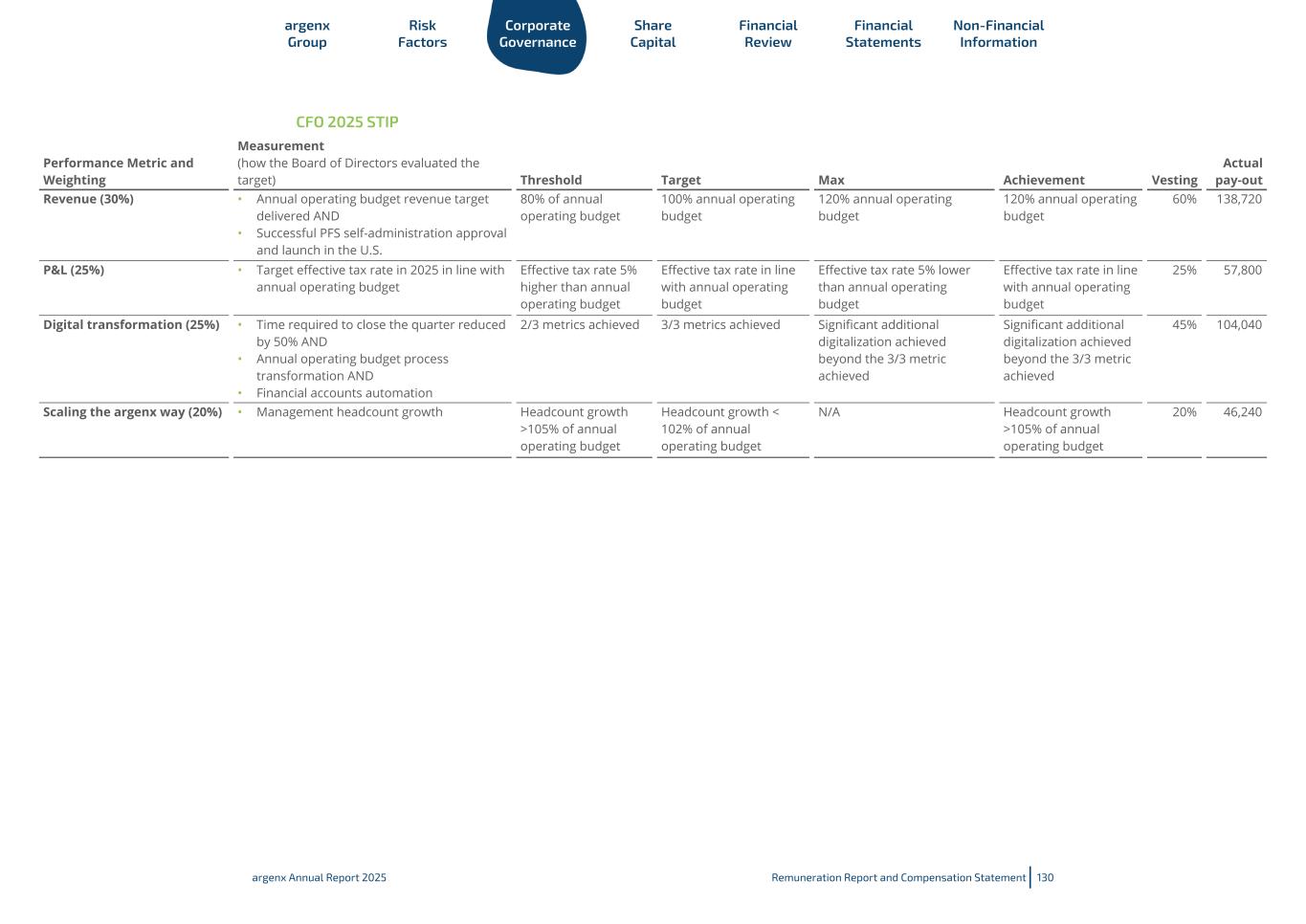
CFO 2025 STIP Revenue (30%) • Annual operating budget revenue target delivered AND • Successful PFS self-administration approval and launch in the U.S. 80% of annual operating budget 100% annual operating budget 120% annual operating budget 120% annual operating budget 60% 138,720 P&L (25%) • Target effective tax rate in 2025 in line with annual operating budget Effective tax rate 5% higher than annual operating budget Effective tax rate in line with annual operating budget Effective tax rate 5% lower than annual operating budget Effective tax rate in line with annual operating budget 25% 57,800 Digital transformation (25%) • Time required to close the quarter reduced by 50% AND • Annual operating budget process transformation AND • Financial accounts automation 2/3 metrics achieved 3/3 metrics achieved Significant additional digitalization achieved beyond the 3/3 metric achieved Significant additional digitalization achieved beyond the 3/3 metric achieved 45% 104,040 Scaling the argenx way (20%) • Management headcount growth Headcount growth >105% of annual operating budget Headcount growth < 102% of annual operating budget N/A Headcount growth >105% of annual operating budget 20% 46,240 Performance Metric and Weighting Measurement (how the Board of Directors evaluated the target) Threshold Target Max Achievement Vesting Actual pay-out argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Remuneration Report and Compensation Statement 130
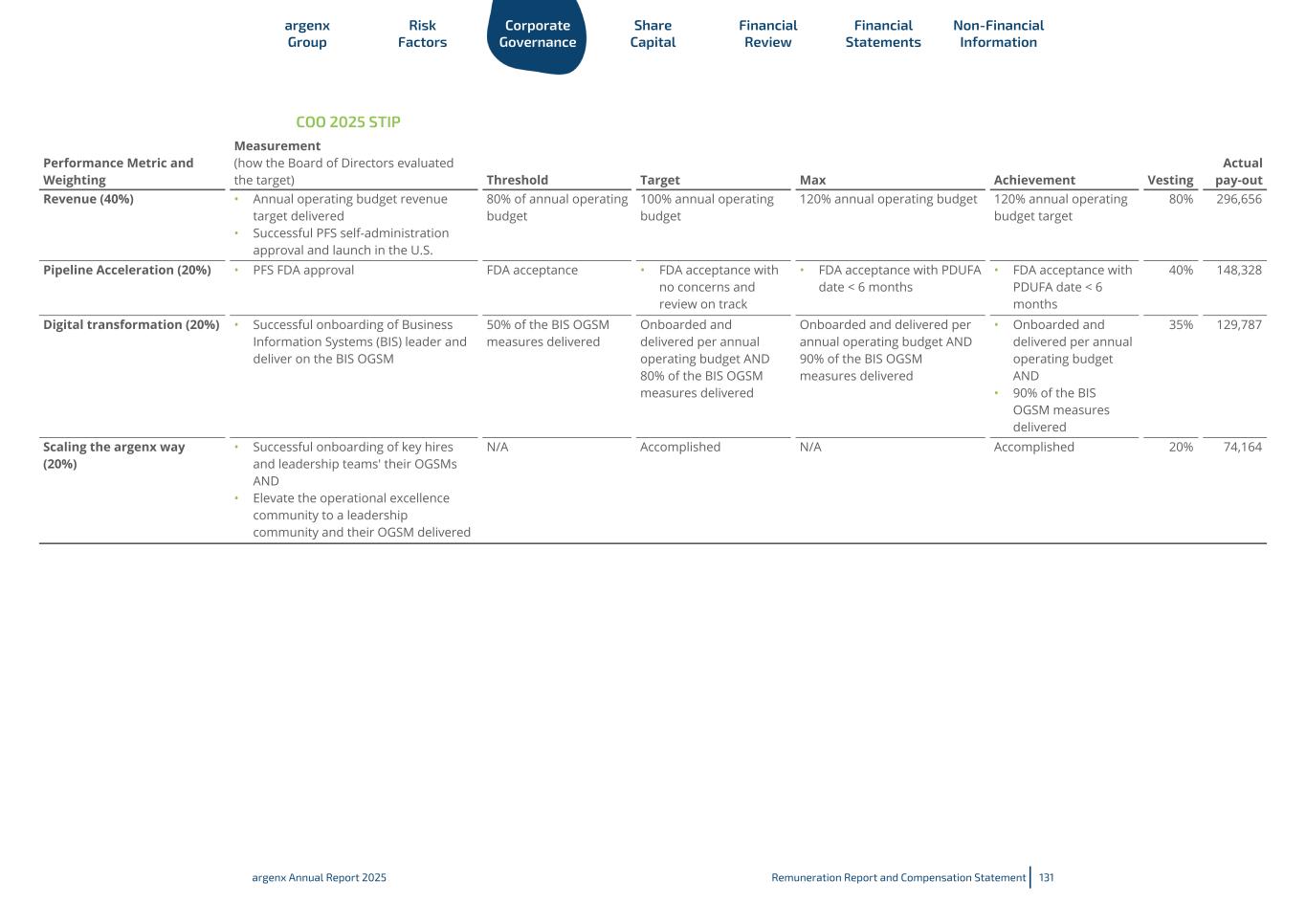
COO 2025 STIP Performance Metric and Weighting Measurement (how the Board of Directors evaluated the target) Threshold Target Max Achievement Vesting Actual pay-out Revenue (40%) • Annual operating budget revenue target delivered • Successful PFS self-administration approval and launch in the U.S. 80% of annual operating budget 100% annual operating budget 120% annual operating budget 120% annual operating budget target 80% 296,656 Pipeline Acceleration (20%) • PFS FDA approval FDA acceptance • FDA acceptance with no concerns and review on track • FDA acceptance with PDUFA date < 6 months • FDA acceptance with PDUFA date < 6 months 40% 148,328 Digital transformation (20%) • Successful onboarding of Business Information Systems (BIS) leader and deliver on the BIS OGSM 50% of the BIS OGSM measures delivered Onboarded and delivered per annual operating budget AND 80% of the BIS OGSM measures delivered Onboarded and delivered per annual operating budget AND 90% of the BIS OGSM measures delivered • Onboarded and delivered per annual operating budget AND • 90% of the BIS OGSM measures delivered 35% 129,787 Scaling the argenx way (20%) • Successful onboarding of key hires and leadership teams' their OGSMs AND • Elevate the operational excellence community to a leadership community and their OGSM delivered N/A Accomplished N/A Accomplished 20% 74,164 argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Remuneration Report and Compensation Statement 131
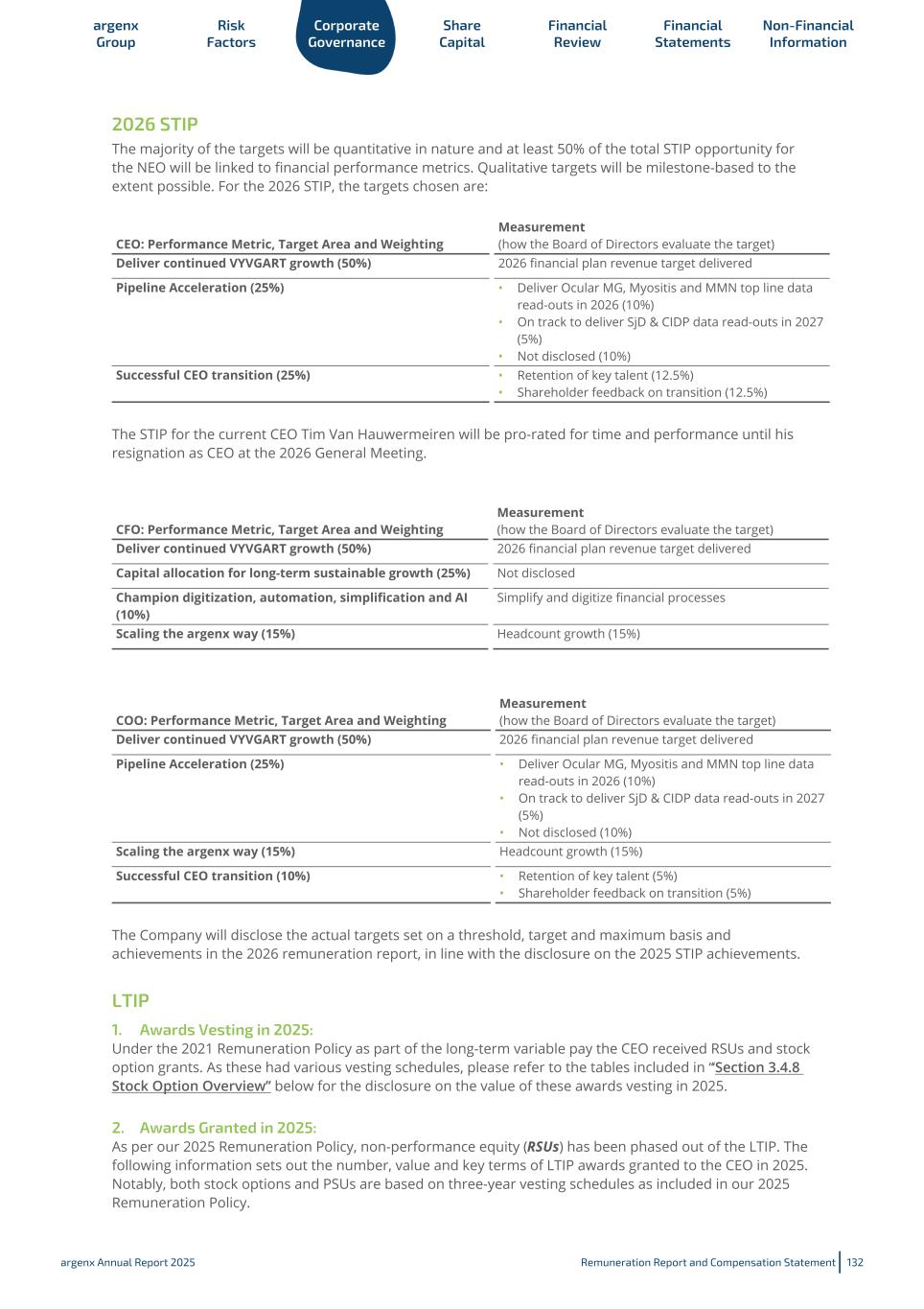
2026 STIP The majority of the targets will be quantitative in nature and at least 50% of the total STIP opportunity for the NEO will be linked to financial performance metrics. Qualitative targets will be milestone-based to the extent possible. For the 2026 STIP, the targets chosen are: CEO: Performance Metric, Target Area and Weighting Measurement (how the Board of Directors evaluate the target) Deliver continued VYVGART growth (50%) 2026 financial plan revenue target delivered Pipeline Acceleration (25%) • Deliver Ocular MG, Myositis and MMN top line data read-outs in 2026 (10%) • On track to deliver SjD & CIDP data read-outs in 2027 (5%) • Not disclosed (10%) Successful CEO transition (25%) • Retention of key talent (12.5%) • Shareholder feedback on transition (12.5%) The STIP for the current CEO Tim Van Hauwermeiren will be pro-rated for time and performance until his resignation as CEO at the 2026 General Meeting. CFO: Performance Metric, Target Area and Weighting Measurement (how the Board of Directors evaluate the target) Deliver continued VYVGART growth (50%) 2026 financial plan revenue target delivered Capital allocation for long-term sustainable growth (25%) Not disclosed Champion digitization, automation, simplification and AI (10%) Simplify and digitize financial processes Scaling the argenx way (15%) Headcount growth (15%) COO: Performance Metric, Target Area and Weighting Measurement (how the Board of Directors evaluate the target) Deliver continued VYVGART growth (50%) 2026 financial plan revenue target delivered Pipeline Acceleration (25%) • Deliver Ocular MG, Myositis and MMN top line data read-outs in 2026 (10%) • On track to deliver SjD & CIDP data read-outs in 2027 (5%) • Not disclosed (10%) Scaling the argenx way (15%) Headcount growth (15%) Successful CEO transition (10%) • Retention of key talent (5%) • Shareholder feedback on transition (5%) The Company will disclose the actual targets set on a threshold, target and maximum basis and achievements in the 2026 remuneration report, in line with the disclosure on the 2025 STIP achievements. LTIP 1. Awards Vesting in 2025: Under the 2021 Remuneration Policy as part of the long-term variable pay the CEO received RSUs and stock option grants. As these had various vesting schedules, please refer to the tables included in ‘‘‘Section 3.4.8 Stock Option Overview’’ below for the disclosure on the value of these awards vesting in 2025. 2. Awards Granted in 2025: As per our 2025 Remuneration Policy, non-performance equity (RSUs) has been phased out of the LTIP. The following information sets out the number, value and key terms of LTIP awards granted to the CEO in 2025. Notably, both stock options and PSUs are based on three-year vesting schedules as included in our 2025 Remuneration Policy. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Remuneration Report and Compensation Statement 132

• Stock Options: The number of stock options was calculated by dividing the target value through the then applicable Black-Scholes value based on 30 calendar days preceding the 15th day of the month in which the grant occurs (the Reference Date), rounded up to the nearest whole number. The stock options granted on June 30, 2025 to the CEO have an exercise price of €479.30 / $561.74. • PSUs: The numbers of PSUs was calculated by dividing the target value through the average closing price 30 calendar days preceding the Reference Date, rounded up to the nearest whole number. • Target Value: Using the above methodology, the total LTIP target grant was valued at $5,790,000, $3,395,000, and $3,895,000 which is 7.0, 5.9 and 5.3 times the CEO’s, CFO’s and COO’s base salaries, respectively. The CEO and CFO received their respective equity grants converted into a number of stock options and PSUs on the Reference Date of the 30-days average share price of 510.88 EUR/$569.17 per share preceding the Reference Date and the Black-Scholes model fair market value of 172.92 EUR/ $192.65 per stock option. Consequently, 15,027, 8,812, 10,110 stock options (50% of the LTIP grant) and 5,085, 2,983, 3,423 PSUs (50% of the LTIP grant) were granted to the CEO, CFO and COO, respectively. • NB: Relevant for CEO only as he is a Belgian beneficiary: these amounts do not reflect the actual economic value realized by the beneficiary. Amounts included represent the expenses with respect to the assumptions used in the Black-Scholes model which differ between Belgian beneficiaries versus non- Belgian beneficiaries, resulting in the CEO’s stock based compensation expenses being higher than other beneficiaries. For a description of the assumptions used, refer to “Note 13 Share-Based Payment in Section 6 ”Consolidated Financial Statements” which are included to our Annual Report for the period ended December 31, 2025. PSUs Together with the rest of the Senior Management Team, the CFO and the COO Karen Massey, who is envisaged to be elected as an Executive Director during the 2026 General Meeting and subsequently transition into the role of CEO, are eligible to receive a PSU grant in 2026. The current CEO, Tim van Hauwermeiren, will not receive a pro-rated PSU grant for 2026 in connection with his transition into the role of Non-Executive Director and Chairperson of the Board of Directors at the 2026 General Meeting. The four measures of the 2026–2028 PSUs are based on the following principles: • at least 50% of the pay opportunity will be linked to financial performance metrics such as revenue growth; • at least 40% of the pay opportunity will be linked to innovation and pipeline progression metrics, such as delivering clinical and regulatory milestones; and • up to 10% of the pay opportunity will be linked to people and culture metrics essential for sustainable, long-term value creation. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Remuneration Report and Compensation Statement 133

Performance Metric Target Measurement (how the Board of Directors will evaluate the metric and why it has been chosen) Threshold Target Max Maximize the VYVGART opportunity (50%) 2028 annual revenue Minimum product net sales of undisclosed amount Targets and Executive Director achievement will be disclosed retroactively in the 2028 remuneration report, published in 2029 Pipeline progression (40%) (s)BLA Approvals (in addition to potential Seronegative gMG and Ocular MG) (20%) Undisclosed number of new approvals Phase (ii) Progression and/or IND / CTA Assets Submissions (20%) Undisclosed number of new pipeline assets into phase 2 and/or undisclosed number of new additional pipeline assets IND / clinical trial application submitted Scaling the argenx way (10%) Talent retention Three-year average voluntary employee turnover equal to or below 6.5% (target) or equal to or below 8.5% (floor) Shareholding Requirements The remuneration policy requires an Executive Director to build up a shareholding requirement 6x their base pay. On December 31, 2025, the CEO is in compliance with this requirement. Clawback policy In the year ended December 31, 2025, no variable remuneration was clawed back and no variable remuneration was adjusted (retroactively). Looking Forward – Mr Van Hauwermeiren 2026 CEO position As Tim Van Hauwermeiren will be treated as a good leaver, his anticipated CEO remuneration for 2026 will be delivered in accordance with the provisions of the 2025 Remuneration Policy and is outlined below: • Treatment of base pay: The annual base pay for 2026 will be paid, pro-rated, up to and including the date of his resignation as CEO at the 2026 General Meeting. This is expected to amount to EUR 261,665, being a 126 day pro-rated pay out of the full 2026 base pay of EUR 758,000. • Treatment of 2026 STI: The 2026 STI will be paid on a pro-rated basis up to and including the date of his resignation as CEO at the 2026 General Meeting. Achievement and pay-out will be determined on the date of resignation at the 2026 General Meeting and will occur shortly thereafter to facilitate his full transition into a Non-Executive Director and chairperson of the Board of Directors. Since at the date of this 2025 Remuneration Report the performance period is still running until May 6, 2026, disclosure will be included in the 2026 remuneration report, available in the 2027 annual report. • Treatment of 2025 LTI: All unvested equity other than PSUs will immediately and fully vest at the time of the 2026 General Meeting. The PSUs granted in 2025 will remain subject to their normal three-year performance period and will vest in 2027, based on performance and pro-rated for time served as CEO in the calendar years 2025 and 2026 (up to and including the date of his resignation as CEO at the 2026 General Meeting). The achievement and pay-out will be determined based on the information available to us on May 6, 2026 and the actual vesting and pay-out will occur at the end of the three-year performance period in 2027. Disclosure will be included in the 2027 remuneration report, available in the 2028 annual report. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Remuneration Report and Compensation Statement 134
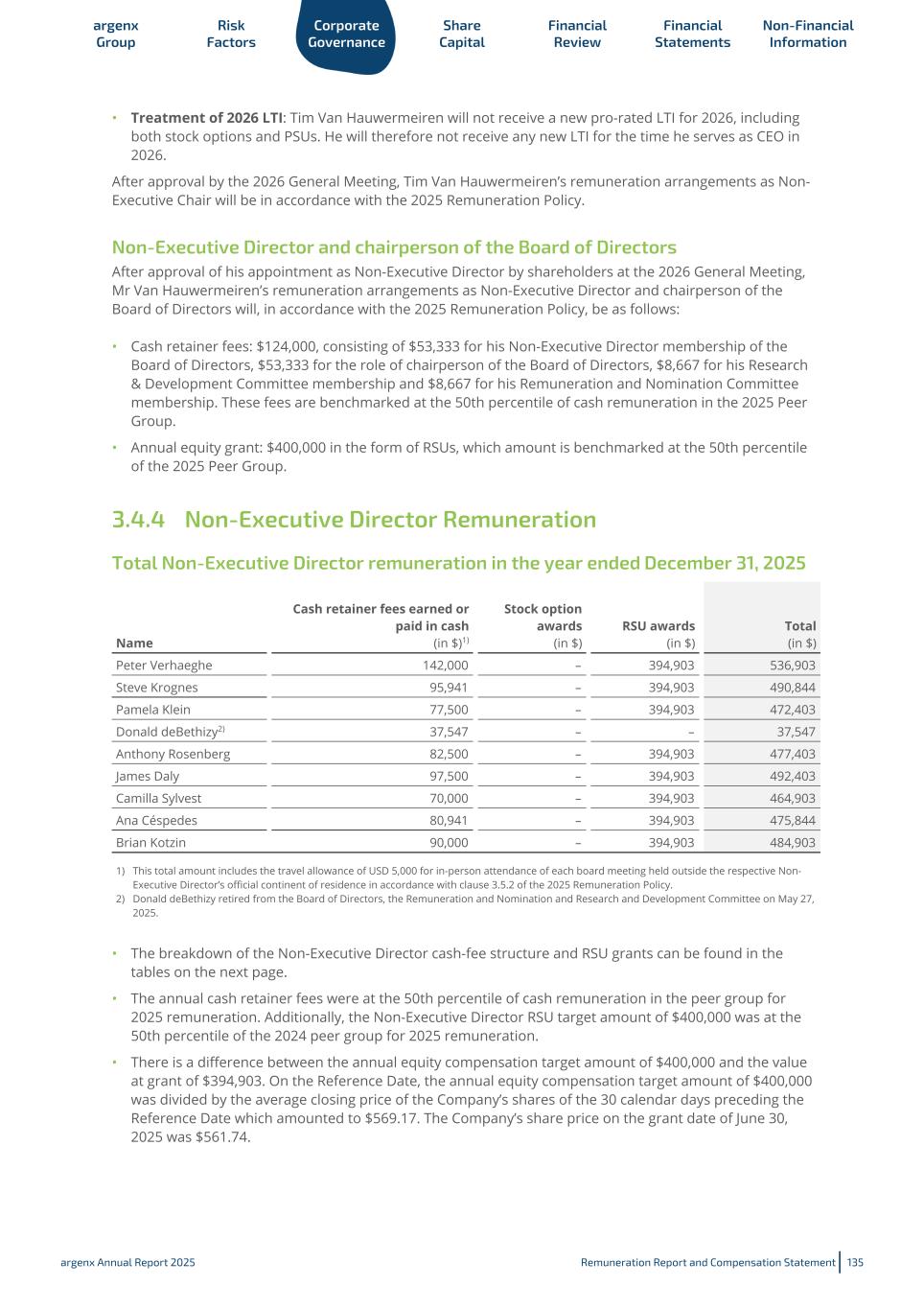
• Treatment of 2026 LTI: Tim Van Hauwermeiren will not receive a new pro-rated LTI for 2026, including both stock options and PSUs. He will therefore not receive any new LTI for the time he serves as CEO in 2026. After approval by the 2026 General Meeting, Tim Van Hauwermeiren’s remuneration arrangements as Non- Executive Chair will be in accordance with the 2025 Remuneration Policy. Non-Executive Director and chairperson of the Board of Directors After approval of his appointment as Non-Executive Director by shareholders at the 2026 General Meeting, Mr Van Hauwermeiren’s remuneration arrangements as Non-Executive Director and chairperson of the Board of Directors will, in accordance with the 2025 Remuneration Policy, be as follows: • Cash retainer fees: $124,000, consisting of $53,333 for his Non-Executive Director membership of the Board of Directors, $53,333 for the role of chairperson of the Board of Directors, $8,667 for his Research & Development Committee membership and $8,667 for his Remuneration and Nomination Committee membership. These fees are benchmarked at the 50th percentile of cash remuneration in the 2025 Peer Group. • Annual equity grant: $400,000 in the form of RSUs, which amount is benchmarked at the 50th percentile of the 2025 Peer Group. 3.4.4 Non-Executive Director Remuneration Total Non-Executive Director remuneration in the year ended December 31, 2025 Name Cash retainer fees earned or paid in cash (in $)1) Stock option awards (in $) RSU awards (in $) Total (in $) Peter Verhaeghe 142,000 – 394,903 536,903 Steve Krognes 95,941 – 394,903 490,844 Pamela Klein 77,500 – 394,903 472,403 Donald deBethizy2) 37,547 – – 37,547 Anthony Rosenberg 82,500 – 394,903 477,403 James Daly 97,500 – 394,903 492,403 Camilla Sylvest 70,000 – 394,903 464,903 Ana Céspedes 80,941 – 394,903 475,844 Brian Kotzin 90,000 – 394,903 484,903 1) This total amount includes the travel allowance of USD 5,000 for in-person attendance of each board meeting held outside the respective Non- Executive Director’s official continent of residence in accordance with clause 3.5.2 of the 2025 Remuneration Policy. 2) Donald deBethizy retired from the Board of Directors, the Remuneration and Nomination and Research and Development Committee on May 27, 2025. • The breakdown of the Non-Executive Director cash-fee structure and RSU grants can be found in the tables on the next page. • The annual cash retainer fees were at the 50th percentile of cash remuneration in the peer group for 2025 remuneration. Additionally, the Non-Executive Director RSU target amount of $400,000 was at the 50th percentile of the 2024 peer group for 2025 remuneration. • There is a difference between the annual equity compensation target amount of $400,000 and the value at grant of $394,903. On the Reference Date, the annual equity compensation target amount of $400,000 was divided by the average closing price of the Company’s shares of the 30 calendar days preceding the Reference Date which amounted to $569.17. The Company’s share price on the grant date of June 30, 2025 was $561.74. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Remuneration Report and Compensation Statement 135
Non-Executive Director Shareholding Non-Executive Directors are required to hold at least 5x annual Board of Director membership retainer fees ($60,000 in 2025) worth of Company stock for the duration of their role. As of December 31, 2025, all Non- Executive Directors comply with this requirement. Deviations In the year ended December 31, 2025, the Company did not deviate from the decision-making process for Executive and Non-Executive Director pay and no deviations took place from the 2021 Remuneration Policy or the 2025 Remuneration Policy. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Remuneration Report and Compensation Statement 136
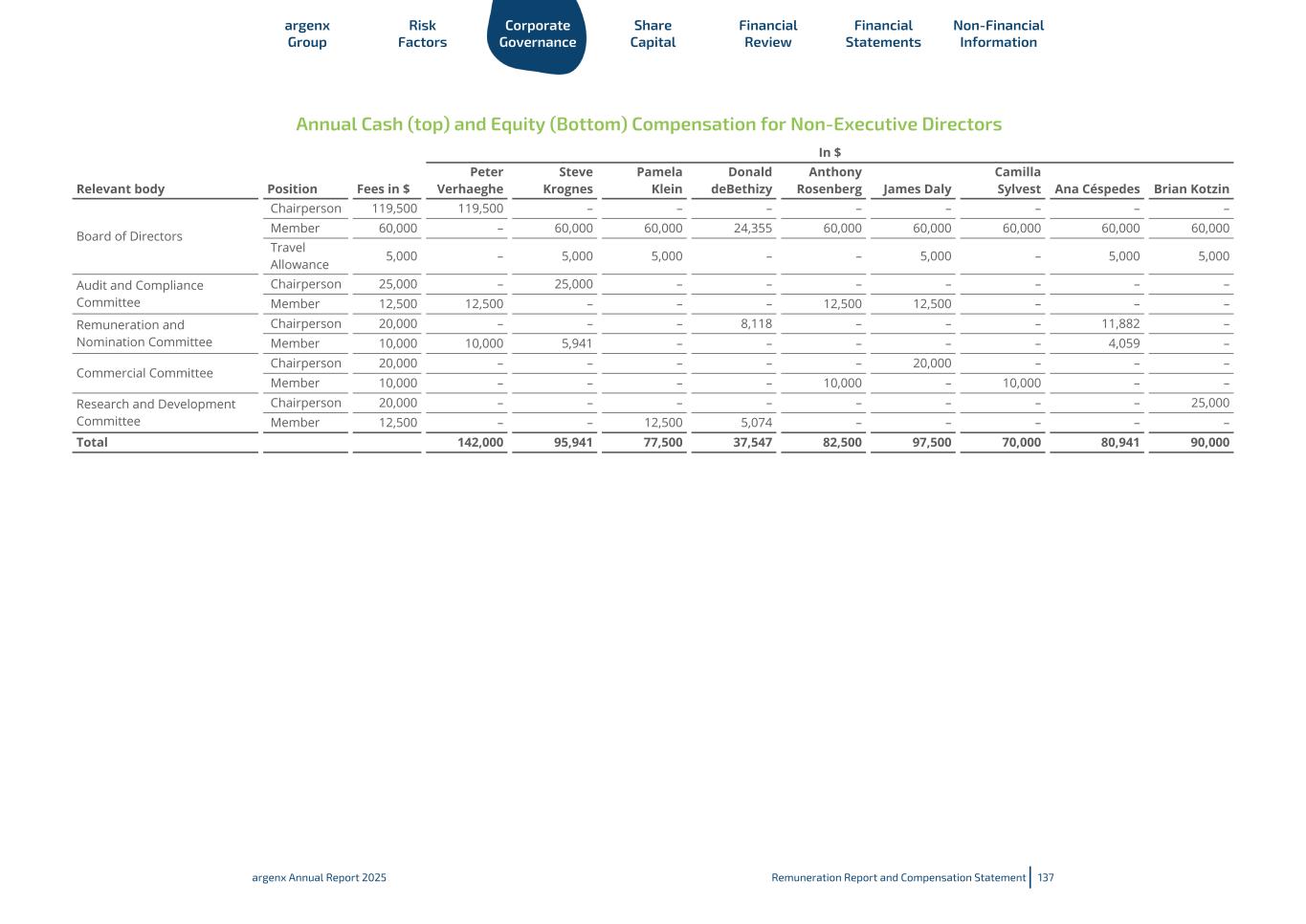
Annual Cash (top) and Equity (Bottom) Compensation for Non-Executive Directors In $ Relevant body Position Fees in $ Peter Verhaeghe Steve Krognes Pamela Klein Donald deBethizy Anthony Rosenberg James Daly Camilla Sylvest Ana Céspedes Brian Kotzin Board of Directors Chairperson 119,500 119,500 – – – – – – – – Member 60,000 – 60,000 60,000 24,355 60,000 60,000 60,000 60,000 60,000 Travel Allowance 5,000 – 5,000 5,000 – – 5,000 – 5,000 5,000 Audit and Compliance Committee Chairperson 25,000 – 25,000 – – – – – – – Member 12,500 12,500 – – – 12,500 12,500 – – – Remuneration and Nomination Committee Chairperson 20,000 – – – 8,118 – – – 11,882 – Member 10,000 10,000 5,941 – – – – – 4,059 – Commercial Committee Chairperson 20,000 – – – – – 20,000 – – – Member 10,000 – – – – 10,000 – 10,000 – – Research and Development Committee Chairperson 20,000 – – – – – – – – 25,000 Member 12,500 – – 12,500 5,074 – – – – – Total 142,000 95,941 77,500 37,547 82,500 97,500 70,000 80,941 90,000 argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Remuneration Report and Compensation Statement 137
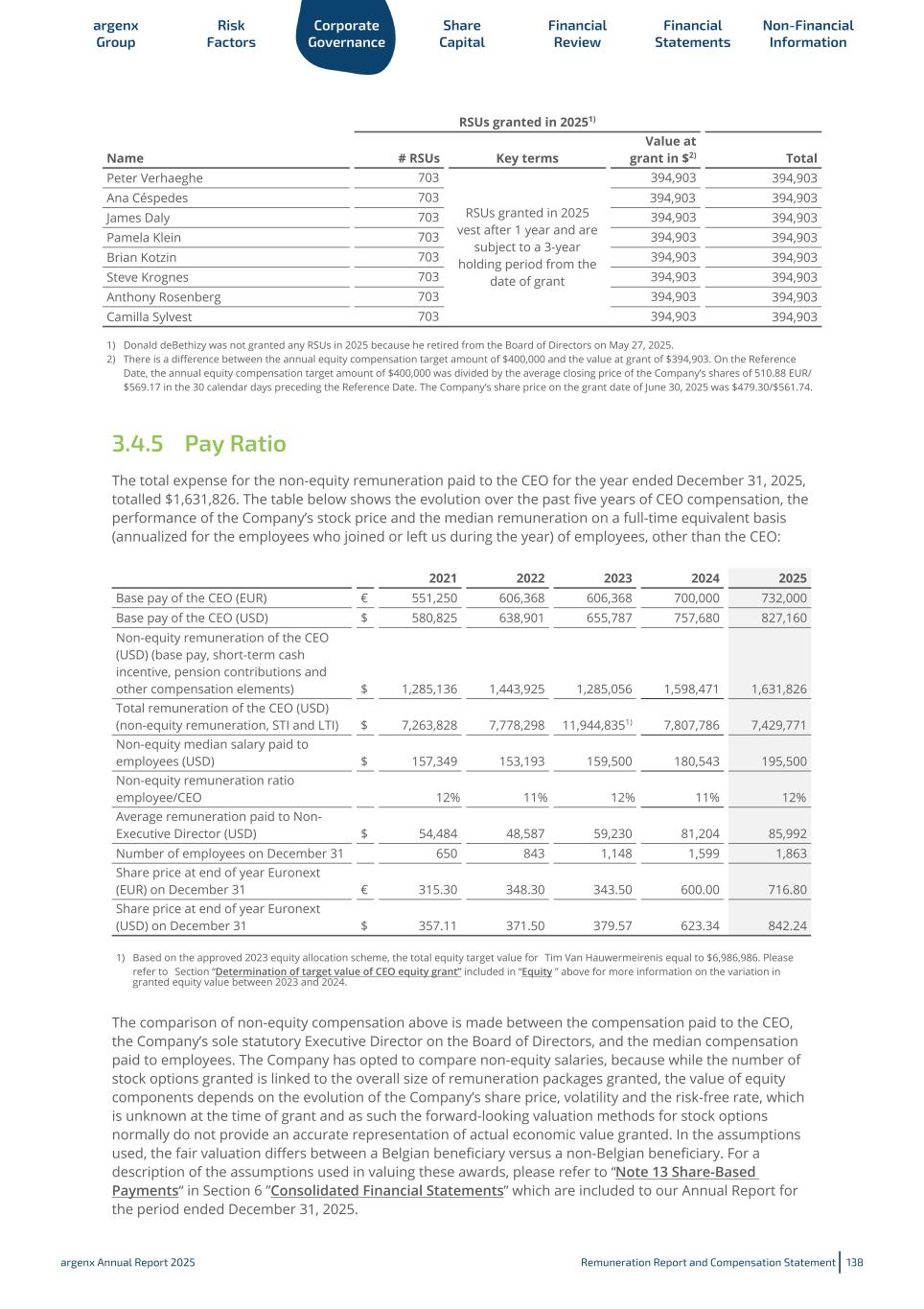
RSUs granted in 20251) Name # RSUs Key terms Value at grant in $2) Total Peter Verhaeghe 703 RSUs granted in 2025 vest after 1 year and are subject to a 3-year holding period from the date of grant 394,903 394,903 Ana Céspedes 703 394,903 394,903 James Daly 703 394,903 394,903 Pamela Klein 703 394,903 394,903 Brian Kotzin 703 394,903 394,903 Steve Krognes 703 394,903 394,903 Anthony Rosenberg 703 394,903 394,903 Camilla Sylvest 703 394,903 394,903 1) Donald deBethizy was not granted any RSUs in 2025 because he retired from the Board of Directors on May 27, 2025. 2) There is a difference between the annual equity compensation target amount of $400,000 and the value at grant of $394,903. On the Reference Date, the annual equity compensation target amount of $400,000 was divided by the average closing price of the Company’s shares of 510.88 EUR/ $569.17 in the 30 calendar days preceding the Reference Date. The Company’s share price on the grant date of June 30, 2025 was $479.30/$561.74. 3.4.5 Pay Ratio The total expense for the non-equity remuneration paid to the CEO for the year ended December 31, 2025, totalled $1,631,826. The table below shows the evolution over the past five years of CEO compensation, the performance of the Company’s stock price and the median remuneration on a full-time equivalent basis (annualized for the employees who joined or left us during the year) of employees, other than the CEO: 2021 2022 2023 2024 2025 Base pay of the CEO (EUR) € 551,250 606,368 606,368 700,000 732,000 Base pay of the CEO (USD) $ 580,825 638,901 655,787 757,680 827,160 Non-equity remuneration of the CEO (USD) (base pay, short-term cash incentive, pension contributions and other compensation elements) $ 1,285,136 1,443,925 1,285,056 1,598,471 1,631,826 Total remuneration of the CEO (USD) (non-equity remuneration, STI and LTI) $ 7,263,828 7,778,298 11,944,8351) 7,807,786 7,429,771 Non-equity median salary paid to employees (USD) $ 157,349 153,193 159,500 180,543 195,500 Non-equity remuneration ratio employee/CEO 12% 11% 12% 11% 12% Average remuneration paid to Non- Executive Director (USD) $ 54,484 48,587 59,230 81,204 85,992 Number of employees on December 31 650 843 1,148 1,599 1,863 Share price at end of year Euronext (EUR) on December 31 € 315.30 348.30 343.50 600.00 716.80 Share price at end of year Euronext (USD) on December 31 $ 357.11 371.50 379.57 623.34 842.24 1) Based on the approved 2023 equity allocation scheme, the total equity target value for Tim Van Hauwermeirenis equal to $6,986,986. Please refer to Section “Determination of target value of CEO equity grant” included in “Equity ” above for more information on the variation in granted equity value between 2023 and 2024. The comparison of non-equity compensation above is made between the compensation paid to the CEO, the Company’s sole statutory Executive Director on the Board of Directors, and the median compensation paid to employees. The Company has opted to compare non-equity salaries, because while the number of stock options granted is linked to the overall size of remuneration packages granted, the value of equity components depends on the evolution of the Company’s share price, volatility and the risk-free rate, which is unknown at the time of grant and as such the forward-looking valuation methods for stock options normally do not provide an accurate representation of actual economic value granted. In the assumptions used, the fair valuation differs between a Belgian beneficiary versus a non-Belgian beneficiary. For a description of the assumptions used in valuing these awards, please refer to “Note 13 Share-Based Payments“ in Section 6 ”Consolidated Financial Statements” which are included to our Annual Report for the period ended December 31, 2025. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Remuneration Report and Compensation Statement 138

Regional pay ratios Due to the global spread of employees over multiple continents, we have also included the above comparison to a regional basis for U.S. employees, EU employees and Japanese employees. Due to the overall higher compensation level in the business sector in the U.S. compared to the EU and Japan, there is a significant difference in the pay ratio when the CEO’s compensation is compared to the median compensation of all employees, compared to employees in the U.S. The following information is provided for reference purposes: Ratio of non-equity compensation of the median employee compared to the CEO for the year ended December 31, 2025 All employees 12% North American employees 16% European employees 9% Japanese employees 5% Rest of the World employees 13% Total employment costs (excluding any costs related stock options and RSUs) paid in the year ended December 31, 2025 was split between regions as follows: Total employment costs in the year ended December 31, 2025 (in millions of $) North-America 272 Europe 247 Japan 16 Rest of the World 3 Share-based payment ratios 2021 2022 2023 2024 2025 Stock options granted to the CEO 25,000 25,000 30,000 18,279 15,027 RSUs granted to the CEO 5,700 5,700 6,700 6,762 – PSUs granted to the CEO N/A N/A N/A N/A 5,085 Median stock options granted to employees 981 900 600 306 221 Median RSUs granted to employees 200 200 94 148 75 Ratio employee/CEO for stock options 4% 4% 2% 2% 1% Ratio employee/CEO for RSUs1) 4% 4% 1% 2% N/A Ratio employee RSUs/CEO for PSUs1) N/A N/A N/A N/A 1% Median number of stock options granted to Non-Executive Directors2) 2,700 2,700 1,600 N/A N/A Median number of RSUs granted to Non- Executive Directors 600 600 350 1,124 703 Median stock options granted to employees 981 900 600 306 120 Ratio Non-Executive Directors/employee stock options2) 36% 33% 38% N/A N/A Ratio Non-Executive Directors/employee RSUs 33% 33% 27% 13% 11% 1) In 2025, PSUs were only granted to members of the Executive Management Team and therefore, the median employee has not received PSUs. 2) In 2024 and 2025, the Non-Executive Directors only received RSUs and no longer were granted stock options. 3.4.6 Other Disclosures Remuneration by subsidiaries In the year ended December 31, 2025, no remuneration was granted and allocated by subsidiaries or other companies whose financials are consolidated, other than the regular remuneration payments made by the entities with whom members of Senior Management Team have their employment contracts. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Remuneration Report and Compensation Statement 139

Severance arrangements In the year ended ended December 31, 2025, no severance payments were granted to the Non-Executive Directors. No loans or guarantees In the year ended December 31, 2025, no loans or guarantees or the like were provided to the NEOs or the Non-Executive Directors. 3.4.7 Key terms of equity plan applicable to grants in 2025 Stock options granted pursuant to the Equity Incentive Plan shall vest over a 36-month period, with 12/36ths of the total grant vesting on the first anniversary of the grant date and the remaining 24/36ths vesting in equal monthly installments of 1/36th each month thereafter. The number of Stock Options that vest on each vesting date is rounded to the nearest whole number. Fractions below 0.5000 are rounded down, while fractions of 0.5000 or above are rounded up. If rounding down, the difference is added to the next vesting date; if rounding up, the difference is deducted from the next vesting date. Any remaining unvested equity is fully vested on the final day of the applicable vesting period—36 months for Stock Options, subject, in each case, to the plan participant’s continued employment or mandate. Stock options are exercisable when vested, and in any case not after the stock option expiration date included in each individual stock option grant, which is 10 years, or in the case of Belgian tax resident employees, at their election either 5 years or 10 years from the date of grant. Each stock option shall be granted with an exercise price equal to the fair market value upon the date of grant and shall have a term equal to five or 10 years from the date of grant. Plan participants may prefer to elect the five-year period as this may limit their personal tax obligations in respect of the stock option in respect to the jurisdiction where stock options are taxed at grant, compared to a ten-year stock option. Stock options granted to Belgian tax resident beneficiaries (including the CEO) are not exercisable prior to the fourth year following the year of the grant. More specifically, stock options granted to an Executive Director cliff vests on the third anniversary of the grant date. Non-Executive Directors are not eligible to receive any stock option grants. RSUs granted under the Equity Incentive Plan shall vest over a period of four years with respect to one fourth of the shares upon each anniversary of the date of grant. At the time of vesting, the holder of such RSUs receives shares in the share capital of the Company for free equal to the number of RSUs vested minus a certain number of shares required to cover employee taxes payable by us on behalf of the holder of RSUs, if applicable. Since 2025, RSUs are no longer granted to the NEOs. Any RSUs granted to Non-Executive Directors in 2025 vested after one year instead of four years and are subject to a three-year holding period. In accordance with our 2025 Remuneration Policy, any RSUs granted in 2026 and beyond to Non-Executive Directors are not subject to any vesting conditions and the shares must be held until the fourth anniversary of the grant date, except to the extent necessary to cover immediate tax obligations resulting from the immediate vest. Since 2025, PSUs are granted to NEOs under the Equity Incentive Plan. PSUs cliff vest at the end of their three-year performance period. Pay-out levels depend upon the achievement of the Executive Director’s measures relative to the threshold, target and maximum levels that were determined by the Board. Unvested equity incentives shall vest in the event of a (i) sale, merger, consolidation, tender offer or similar acquisition of shares or other transaction or series of related transactions as a result of which a change in control occurs, (ii) sale or other disposition of all or substantially all of the Company’s assets or (iii) the Company’s dissolution and/or liquidation. The Board of Directors, upon approval of a majority of the Non-Executive Directors, may amend or terminate the Equity Incentive Plan or may amend the terms of the Equity Incentive Plan, or any outstanding stock options or RSUs, provided that the Company will compensate any affected individual for any direct negative impact of such amendment. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Remuneration Report and Compensation Statement 140
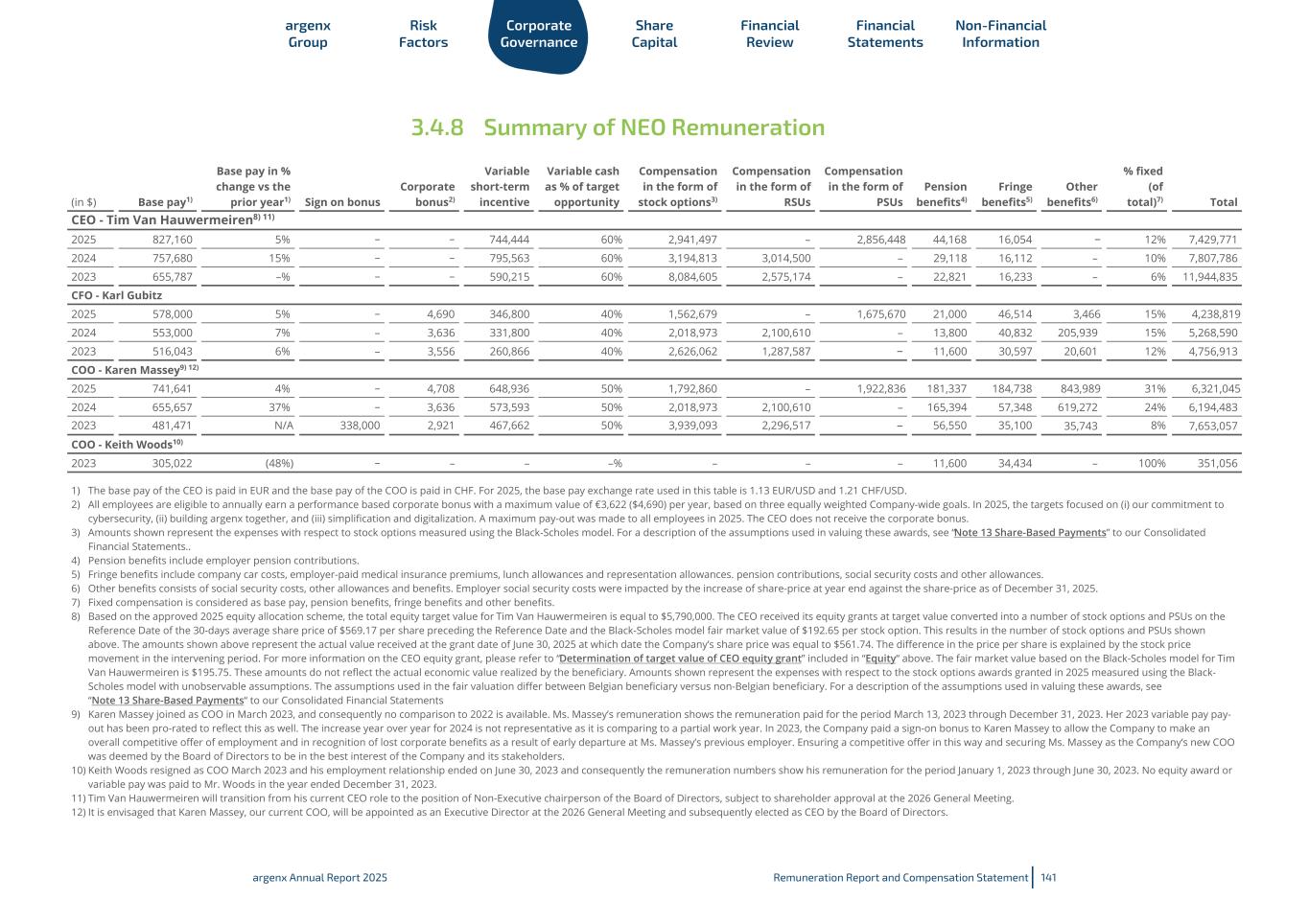
3.4.8 Summary of NEO Remuneration (in $) Base pay1) Base pay in % change vs the prior year1) Sign on bonus Corporate bonus2) Variable short-term incentive Variable cash as % of target opportunity Compensation in the form of stock options3) Compensation in the form of RSUs Compensation in the form of PSUs Pension benefits4) Fringe benefits5) Other benefits6) % fixed (of total)7) Total CEO - Tim Van Hauwermeiren8) 11) 2025 827,160 5% – – 744,444 60% 2,941,497 – 2,856,448 44,168 16,054 – 12% 7,429,771 2024 757,680 15% – – 795,563 60% 3,194,813 3,014,500 – 29,118 16,112 – 10% 7,807,786 2023 655,787 –% – – 590,215 60% 8,084,605 2,575,174 – 22,821 16,233 – 6% 11,944,835 CFO - Karl Gubitz 2025 578,000 5% – 4,690 346,800 40% 1,562,679 – 1,675,670 21,000 46,514 3,466 15% 4,238,819 2024 553,000 7% – 3,636 331,800 40% 2,018,973 2,100,610 – 13,800 40,832 205,939 15% 5,268,590 2023 516,043 6% – 3,556 260,866 40% 2,626,062 1,287,587 – 11,600 30,597 20,601 12% 4,756,913 COO - Karen Massey9) 12) 2025 741,641 4% – 4,708 648,936 50% 1,792,860 – 1,922,836 181,337 184,738 843,989 31% 6,321,045 2024 655,657 37% – 3,636 573,593 50% 2,018,973 2,100,610 – 165,394 57,348 619,272 24% 6,194,483 2023 481,471 N/A 338,000 2,921 467,662 50% 3,939,093 2,296,517 – 56,550 35,100 35,743 8% 7,653,057 COO - Keith Woods10) 2023 305,022 (48%) – – – –% – – – 11,600 34,434 – 100% 351,056 1) The base pay of the CEO is paid in EUR and the base pay of the COO is paid in CHF. For 2025, the base pay exchange rate used in this table is 1.13 EUR/USD and 1.21 CHF/USD. 2) All employees are eligible to annually earn a performance based corporate bonus with a maximum value of €3,622 ($4,690) per year, based on three equally weighted Company-wide goals. In 2025, the targets focused on (i) our commitment to cybersecurity, (ii) building argenx together, and (iii) simplification and digitalization. A maximum pay-out was made to all employees in 2025. The CEO does not receive the corporate bonus. 3) Amounts shown represent the expenses with respect to stock options measured using the Black-Scholes model. For a description of the assumptions used in valuing these awards, see “Note 13 Share-Based Payments” to our Consolidated Financial Statements.. 4) Pension benefits include employer pension contributions. 5) Fringe benefits include company car costs, employer-paid medical insurance premiums, lunch allowances and representation allowances. pension contributions, social security costs and other allowances. 6) Other benefits consists of social security costs, other allowances and benefits. Employer social security costs were impacted by the increase of share-price at year end against the share-price as of December 31, 2025. 7) Fixed compensation is considered as base pay, pension benefits, fringe benefits and other benefits. 8) Based on the approved 2025 equity allocation scheme, the total equity target value for Tim Van Hauwermeiren is equal to $5,790,000. The CEO received its equity grants at target value converted into a number of stock options and PSUs on the Reference Date of the 30-days average share price of $569.17 per share preceding the Reference Date and the Black-Scholes model fair market value of $192.65 per stock option. This results in the number of stock options and PSUs shown above. The amounts shown above represent the actual value received at the grant date of June 30, 2025 at which date the Company’s share price was equal to $561.74. The difference in the price per share is explained by the stock price movement in the intervening period. For more information on the CEO equity grant, please refer to “Determination of target value of CEO equity grant” included in “Equity“ above. The fair market value based on the Black-Scholes model for Tim Van Hauwermeiren is $195.75. These amounts do not reflect the actual economic value realized by the beneficiary. Amounts shown represent the expenses with respect to the stock options awards granted in 2025 measured using the Black- Scholes model with unobservable assumptions. The assumptions used in the fair valuation differ between Belgian beneficiary versus non-Belgian beneficiary. For a description of the assumptions used in valuing these awards, see “Note 13 Share-Based Payments“ to our Consolidated Financial Statements 9) Karen Massey joined as COO in March 2023, and consequently no comparison to 2022 is available. Ms. Massey’s remuneration shows the remuneration paid for the period March 13, 2023 through December 31, 2023. Her 2023 variable pay pay- out has been pro-rated to reflect this as well. The increase year over year for 2024 is not representative as it is comparing to a partial work year. In 2023, the Company paid a sign-on bonus to Karen Massey to allow the Company to make an overall competitive offer of employment and in recognition of lost corporate benefits as a result of early departure at Ms. Massey’s previous employer. Ensuring a competitive offer in this way and securing Ms. Massey as the Company’s new COO was deemed by the Board of Directors to be in the best interest of the Company and its stakeholders. 10) Keith Woods resigned as COO March 2023 and his employment relationship ended on June 30, 2023 and consequently the remuneration numbers show his remuneration for the period January 1, 2023 through June 30, 2023. No equity award or variable pay was paid to Mr. Woods in the year ended December 31, 2023. 11) Tim Van Hauwermeiren will transition from his current CEO role to the position of Non-Executive chairperson of the Board of Directors, subject to shareholder approval at the 2026 General Meeting. 12) It is envisaged that Karen Massey, our current COO, will be appointed as an Executive Director at the 2026 General Meeting and subsequently elected as CEO by the Board of Directors. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Remuneration Report and Compensation Statement 141
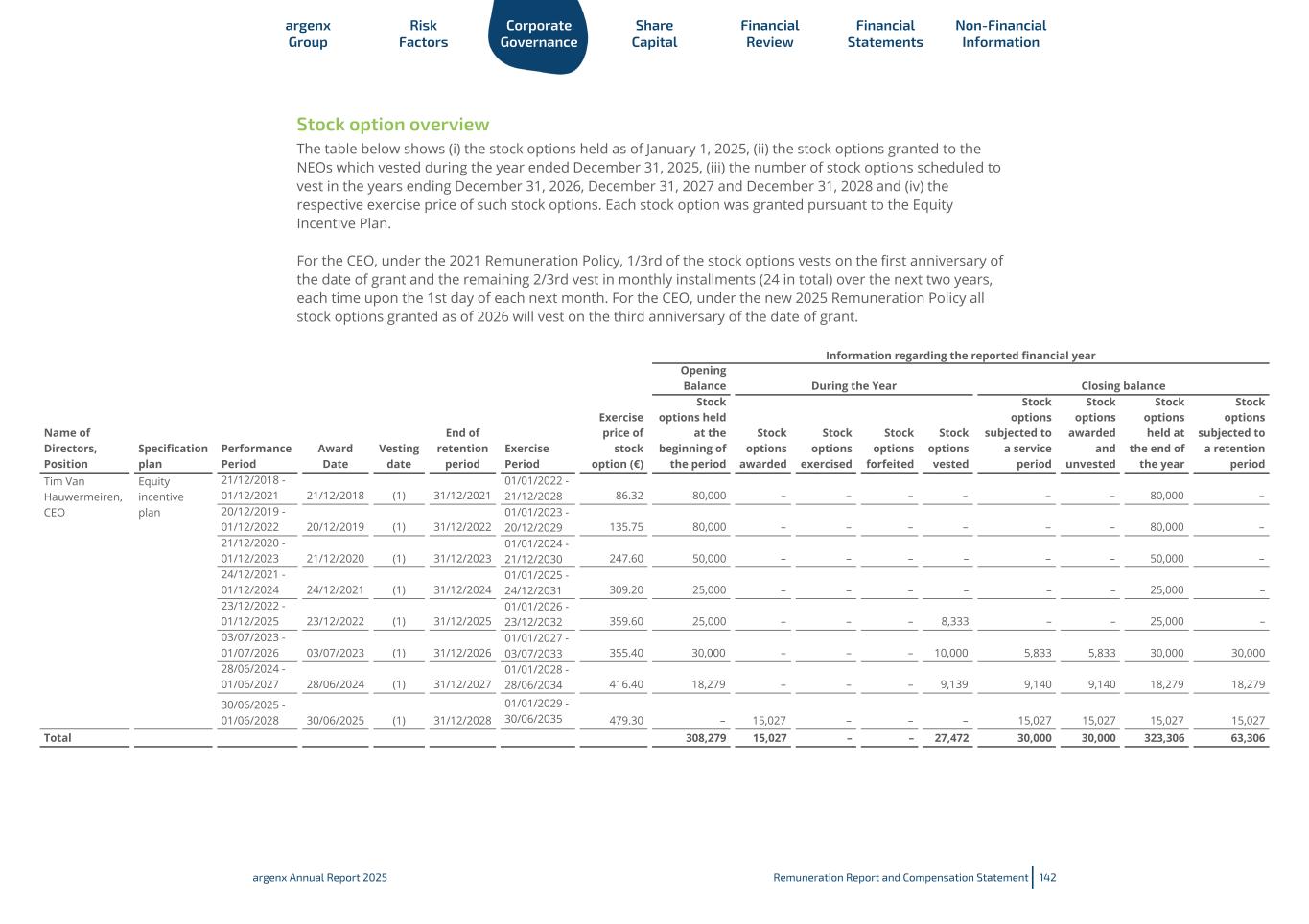
Stock option overview The table below shows (i) the stock options held as of January 1, 2025, (ii) the stock options granted to the NEOs which vested during the year ended December 31, 2025, (iii) the number of stock options scheduled to vest in the years ending December 31, 2026, December 31, 2027 and December 31, 2028 and (iv) the respective exercise price of such stock options. Each stock option was granted pursuant to the Equity Incentive Plan. For the CEO, under the 2021 Remuneration Policy, 1/3rd of the stock options vests on the first anniversary of the date of grant and the remaining 2/3rd vest in monthly installments (24 in total) over the next two years, each time upon the 1st day of each next month. For the CEO, under the new 2025 Remuneration Policy all stock options granted as of 2026 will vest on the third anniversary of the date of grant. Tim Van Hauwermeiren, CEO Equity incentive plan 21/12/2018 - 01/12/2021 21/12/2018 (1) 31/12/2021 01/01/2022 - 21/12/2028 86.32 80,000 – – – – – – 80,000 – 20/12/2019 - 01/12/2022 20/12/2019 (1) 31/12/2022 01/01/2023 - 20/12/2029 135.75 80,000 – – – – – – 80,000 – 21/12/2020 - 01/12/2023 21/12/2020 (1) 31/12/2023 01/01/2024 - 21/12/2030 247.60 50,000 – – – – – – 50,000 – 24/12/2021 - 01/12/2024 24/12/2021 (1) 31/12/2024 01/01/2025 - 24/12/2031 309.20 25,000 – – – – – – 25,000 – 23/12/2022 - 01/12/2025 23/12/2022 (1) 31/12/2025 01/01/2026 - 23/12/2032 359.60 25,000 – – – 8,333 – – 25,000 – 03/07/2023 - 01/07/2026 03/07/2023 (1) 31/12/2026 01/01/2027 - 03/07/2033 355.40 30,000 – – – 10,000 5,833 5,833 30,000 30,000 28/06/2024 - 01/06/2027 28/06/2024 (1) 31/12/2027 01/01/2028 - 28/06/2034 416.40 18,279 – – – 9,139 9,140 9,140 18,279 18,279 30/06/2025 - 01/06/2028 30/06/2025 (1) 31/12/2028 01/01/2029 - 30/06/2035 479.30 – 15,027 – – – 15,027 15,027 15,027 15,027 Total 308,279 15,027 – – 27,472 30,000 30,000 323,306 63,306 Information regarding the reported financial year Opening Balance During the Year Closing balance Name of Directors, Position Specification plan Performance Period Award Date Vesting date End of retention period Exercise Period Exercise price of stock option (€) Stock options held at the beginning of the period Stock options awarded Stock options exercised Stock options forfeited Stock options vested Stock options subjected to a service period Stock options awarded and unvested Stock options held at the end of the year Stock options subjected to a retention period argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Remuneration Report and Compensation Statement 142
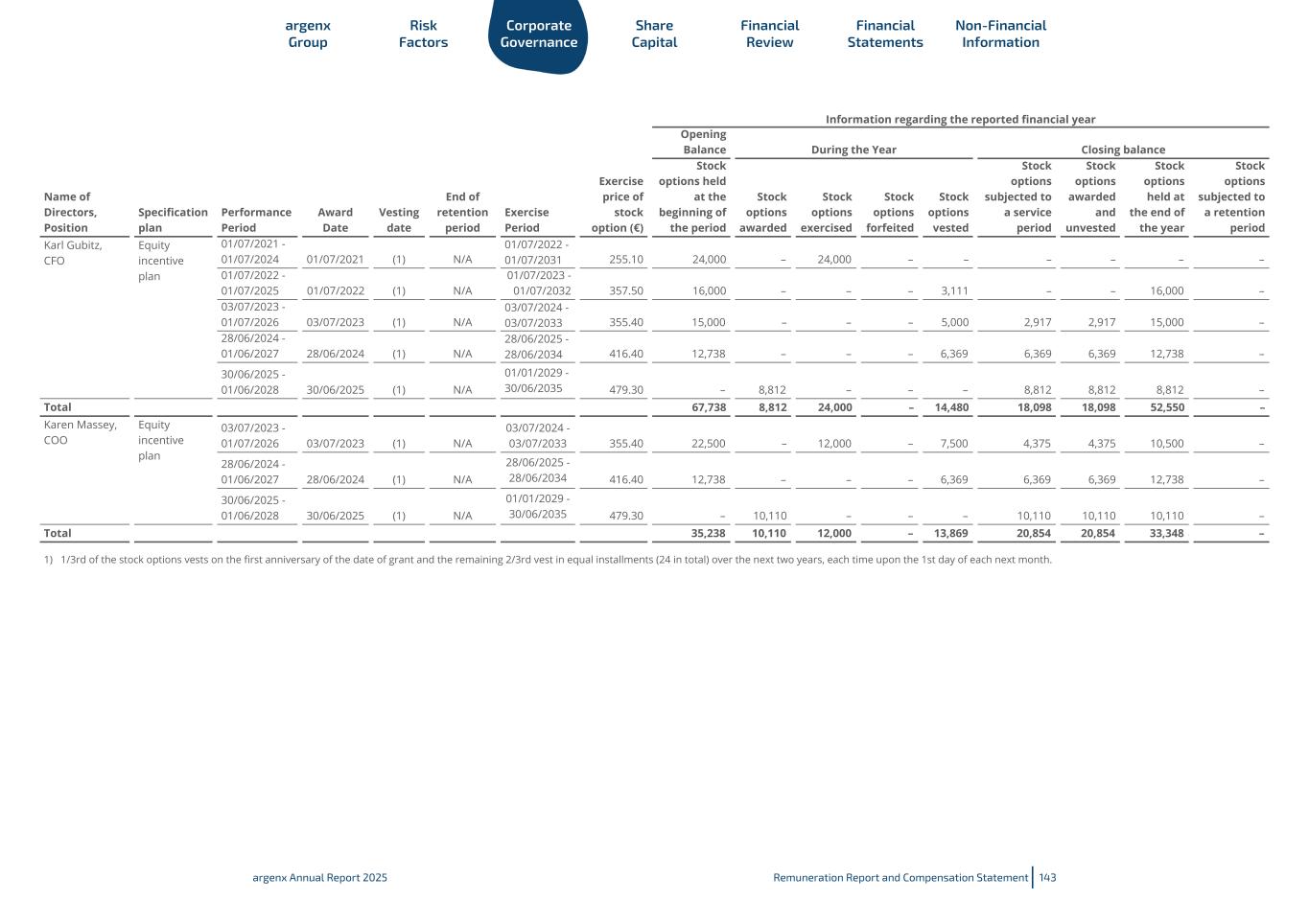
Karl Gubitz, CFO Equity incentive plan 01/07/2021 - 01/07/2024 01/07/2021 (1) N/A 01/07/2022 - 01/07/2031 255.10 24,000 – 24,000 – – – – – – 01/07/2022 - 01/07/2025 01/07/2022 (1) N/A 01/07/2023 - 01/07/2032 357.50 16,000 – – – 3,111 – – 16,000 – 03/07/2023 - 01/07/2026 03/07/2023 (1) N/A 03/07/2024 - 03/07/2033 355.40 15,000 – – – 5,000 2,917 2,917 15,000 – 28/06/2024 - 01/06/2027 28/06/2024 (1) N/A 28/06/2025 - 28/06/2034 416.40 12,738 – – – 6,369 6,369 6,369 12,738 – 30/06/2025 - 01/06/2028 30/06/2025 (1) N/A 01/01/2029 - 30/06/2035 479.30 – 8,812 – – – 8,812 8,812 8,812 – Total 67,738 8,812 24,000 – 14,480 18,098 18,098 52,550 – Karen Massey, COO Equity incentive plan 03/07/2023 - 01/07/2026 03/07/2023 (1) N/A 03/07/2024 - 03/07/2033 355.40 22,500 – 12,000 – 7,500 4,375 4,375 10,500 – 28/06/2024 - 01/06/2027 28/06/2024 (1) N/A 28/06/2025 - 28/06/2034 416.40 12,738 – – – 6,369 6,369 6,369 12,738 – 30/06/2025 - 01/06/2028 30/06/2025 (1) N/A 01/01/2029 - 30/06/2035 479.30 – 10,110 – – – 10,110 10,110 10,110 – Total 35,238 10,110 12,000 – 13,869 20,854 20,854 33,348 8 – 1) 1/3rd of the stock options vests on the first anniversary of the date of grant and the remaining 2/3rd vest in equal installments (24 in total) over the next two years, each time upon the 1st day of each next month. Information regarding the reported financial year Opening Balance During the Year Closing balance Name of Directors, Position Specification plan Performance Period Award Date Vesting date End of retention period Exercise Period Exercise price of stock option (€) Stock options held at the beginning of the period Stock options awarded Stock options exercised Stock options forfeited Stock options vested Stock options subjected to a service period Stock options awarded and unvested Stock options held at the end of the year Stock options subjected to a retention period argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Remuneration Report and Compensation Statement 143
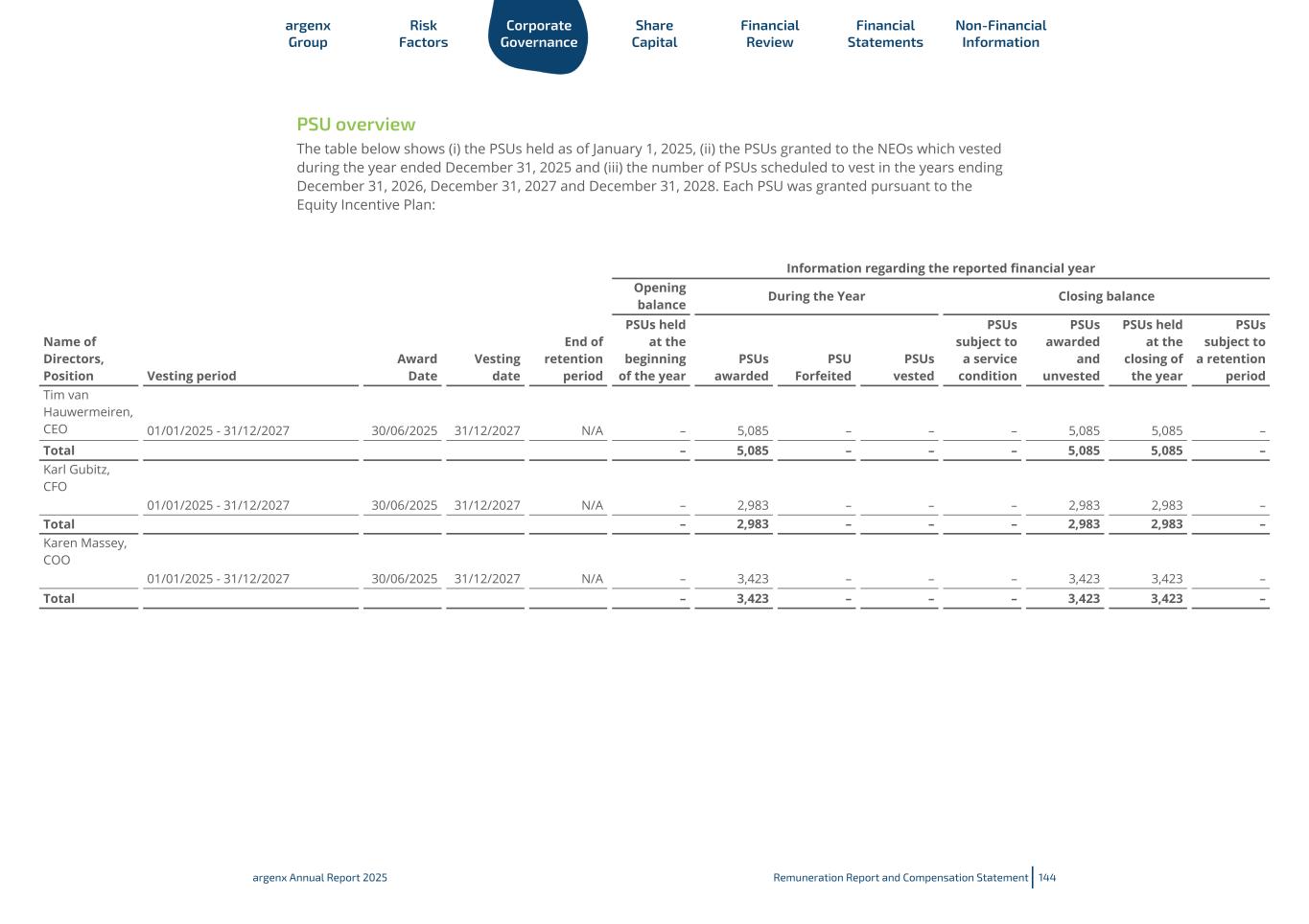
PSU overview The table below shows (i) the PSUs held as of January 1, 2025, (ii) the PSUs granted to the NEOs which vested during the year ended December 31, 2025 and (iii) the number of PSUs scheduled to vest in the years ending December 31, 2026, December 31, 2027 and December 31, 2028. Each PSU was granted pursuant to the Equity Incentive Plan: Information regarding the reported financial year The main conditions of PSU plan Opening balance During the Year Closing balance Name of Directors, Position Vesting period Award Date Vesting date End of retention period PSUs held at the beginning of the year PSUs awarded PSU Forfeited PSUs vested PSUs subject to a service condition PSUs awarded and unvested PSUs held at the closing of the year PSUs subject to a retention period Tim van Hauwermeiren, CEO 01/01/2025 - 31/12/2027 30/06/2025 31/12/2027 N/A – 5,085 – – – 5,085 5,085 – Total – 5,085 – – – 5,085 5,085 – Karl Gubitz, CFO 01/01/2025 - 31/12/2027 30/06/2025 31/12/2027 N/A – 2,983 – – – 2,983 2,983 – Total – 2,983 – – – 2,983 2,983 – Karen Massey, COO 01/01/2025 - 31/12/2027 30/06/2025 31/12/2027 N/A – 3,423 – – – 3,423 3,423 – Total – 3,423 – – – 3,423 3,423 – argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Remuneration Report and Compensation Statement 144
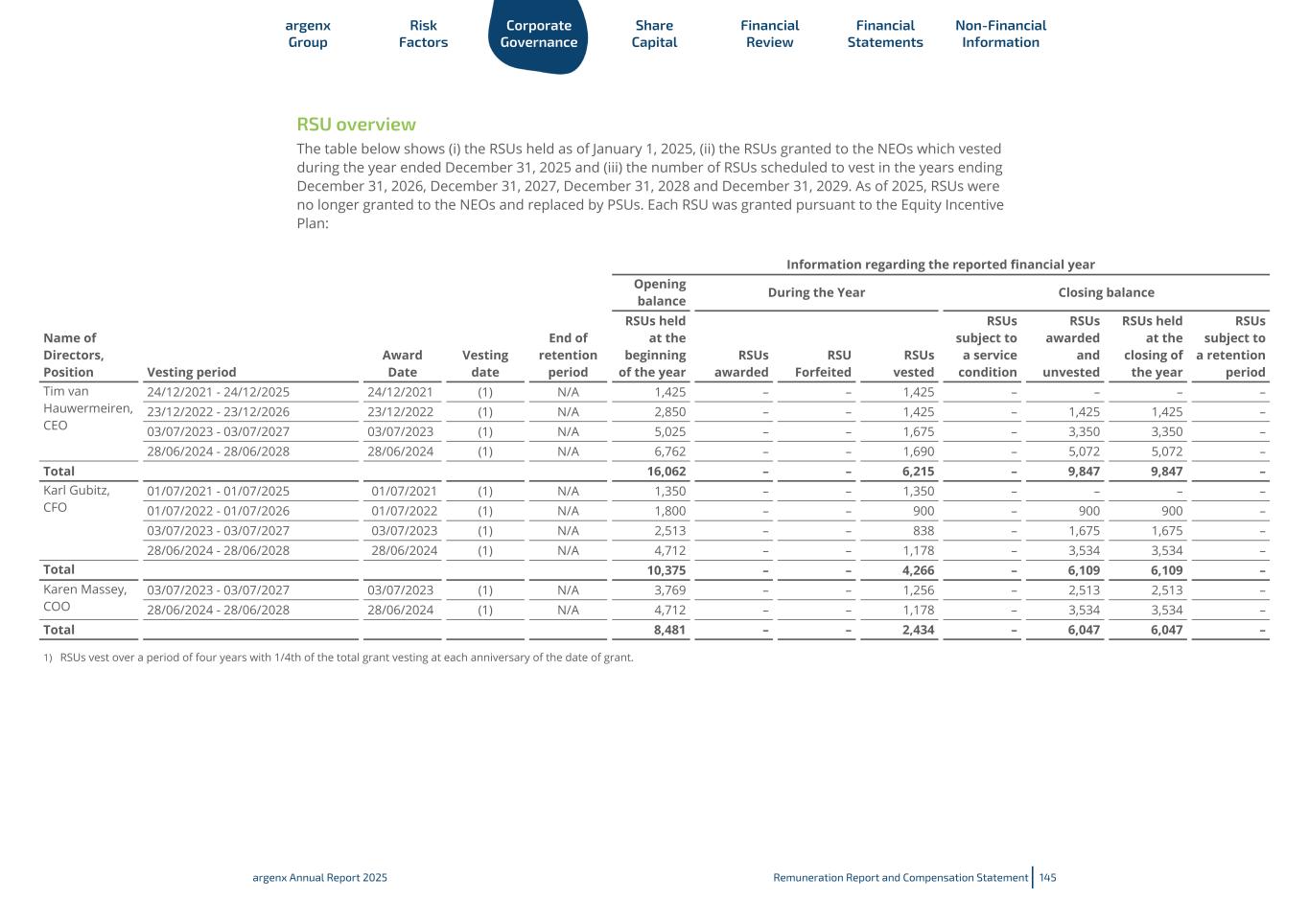
RSU overview The table below shows (i) the RSUs held as of January 1, 2025, (ii) the RSUs granted to the NEOs which vested during the year ended December 31, 2025 and (iii) the number of RSUs scheduled to vest in the years ending December 31, 2026, December 31, 2027, December 31, 2028 and December 31, 2029. As of 2025, RSUs were no longer granted to the NEOs and replaced by PSUs. Each RSU was granted pursuant to the Equity Incentive Plan: Information regarding the reported financial year The main conditions of RSU plan Opening balance During the Year Closing balance Name of Directors, Position Vesting period Award Date Vesting date End of retention period RSUs held at the beginning of the year RSUs awarded RSU Forfeited RSUs vested RSUs subject to a service condition RSUs awarded and unvested RSUs held at the closing of the year RSUs subject to a retention period Tim van Hauwermeiren, CEO 24/12/2021 - 24/12/2025 24/12/2021 (1) N/A 1,425 – – 1,425 – – – – 23/12/2022 - 23/12/2026 23/12/2022 (1) N/A 2,850 – – 1,425 – 1,425 1,425 – 03/07/2023 - 03/07/2027 03/07/2023 (1) N/A 5,025 – – 1,675 – 3,350 3,350 – 28/06/2024 - 28/06/2028 28/06/2024 (1) N/A 6,762 – – 1,690 – 5,072 5,072 – Total 16,062 – – 6,215 – 9,847 9,847 – Karl Gubitz, CFO 01/07/2021 - 01/07/2025 01/07/2021 (1) N/A 1,350 – – 1,350 – – – – 01/07/2022 - 01/07/2026 01/07/2022 (1) N/A 1,800 – – 900 – 900 900 – 03/07/2023 - 03/07/2027 03/07/2023 (1) N/A 2,513 – – 838 – 1,675 1,675 – 28/06/2024 - 28/06/2028 28/06/2024 (1) N/A 4,712 – – 1,178 – 3,534 3,534 – Total 10,375 – – 4,266 – 6,109 6,109 – Karen Massey, COO 03/07/2023 - 03/07/2027 03/07/2023 (1) N/A 3,769 – – 1,256 – 2,513 2,513 – 28/06/2024 - 28/06/2028 28/06/2024 (1) N/A 4,712 – – 1,178 – 3,534 3,534 – Total 8,481 – – 2,434 – 6,047 6,047 – 1) RSUs vest over a period of four years with 1/4th of the total grant vesting at each anniversary of the date of grant. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Remuneration Report and Compensation Statement 145

3.4.9 Remuneration of Other Members of the Senior Management Team For the purposes of U.S. governance reporting requirements, all senior level employees reporting directly to the CEO qualify as the Company’s ‘executives’. The remuneration disclosures in relation to this more extensive group of senior personnel (excluding the NEOs) in this 2025 Remuneration Report is presented on an aggregated basis, with the exception of equity remuneration, which is presented on an individual basis. Aggregate compensation for other members of the Senior Management Team The following table sets forth information regarding aggregate compensation paid to members of the Senior Management Team (other than the NEOs) during the year ended December 31, 2025. (in $) Compensation Base pay 3,138,512 Variable STI1) 1,526,247 Compensation in the form of stock options 10,521,797 Compensation in the form of PSUs 7,443,617 Pension benefits2) 80,045 Fringe benefits3) 107,679 Other benefits4) 3,187,985 Total 26,005,882 1) Variable STI includes a performance based Company wide corporate bonus of €3,622 ($4,690) per member of the Senior Management Team. 2) Pension benefits include employer pension contributions. 3) Fringe benefits include company car costs, employer-paid medical insurance premiums, lunch allowances and representation allowances. pension contributions, social security costs and other allowances. 4) Other benefits consists of social security costs, other allowances and benefits. Employer social security costs were impacted by the increase of share-price at year end against the share-price as of December 31, 2025. For more information on equity granted to members of the Senior Management Team (other than the NEOs), during 2025, please refer to Section 3.4.12 “Summary of Other members of the Senior Management Team” below. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Remuneration Report and Compensation Statement 146
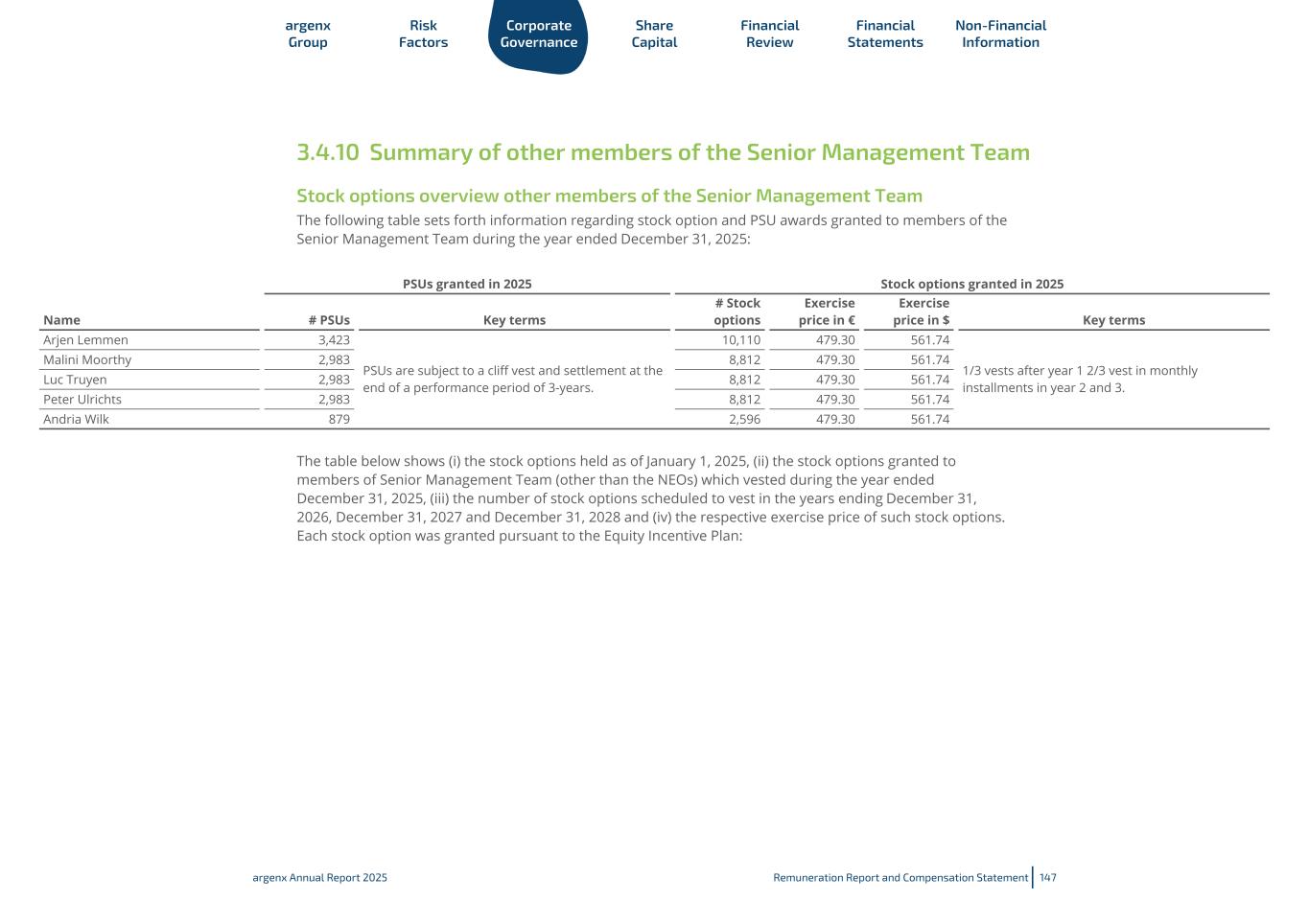
3.4.10 Summary of other members of the Senior Management Team Stock options overview other members of the Senior Management Team The following table sets forth information regarding stock option and PSU awards granted to members of the Senior Management Team during the year ended December 31, 2025: PSUs granted in 2025 Stock options granted in 2025 Name # PSUs Key terms # Stock options Exercise price in € Exercise price in $ Key terms Arjen Lemmen 3,423 PSUs are subject to a cliff vest and settlement at the end of a performance period of 3-years. 10,110 479.30 561.74 1/3 vests after year 1 2/3 vest in monthly installments in year 2 and 3. Malini Moorthy 2,983 8,812 479.30 561.74 Luc Truyen 2,983 8,812 479.30 561.74 Peter Ulrichts 2,983 8,812 479.30 561.74 Andria Wilk 879 2,596 479.30 561.74 The table below shows (i) the stock options held as of January 1, 2025, (ii) the stock options granted to members of Senior Management Team (other than the NEOs) which vested during the year ended December 31, 2025, (iii) the number of stock options scheduled to vest in the years ending December 31, 2026, December 31, 2027 and December 31, 2028 and (iv) the respective exercise price of such stock options. Each stock option was granted pursuant to the Equity Incentive Plan: argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Remuneration Report and Compensation Statement 147
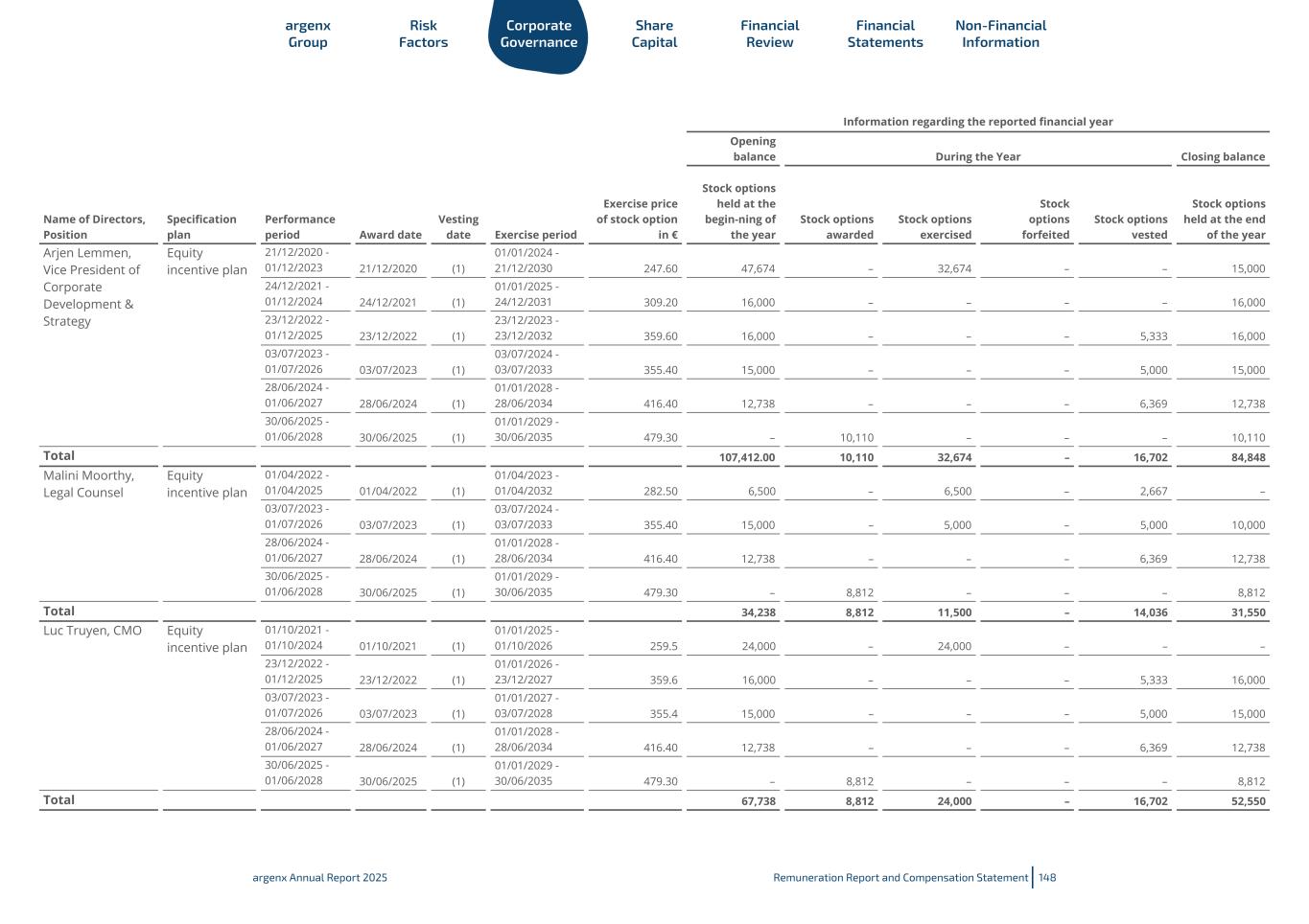
Arjen Lemmen, Vice President of Corporate Development & Strategy Equity incentive plan 21/12/2020 - 01/12/2023 21/12/2020 (1) 01/01/2024 - 21/12/2030 247.60 47,674 – 32,674 – – 15,000 24/12/2021 - 01/12/2024 24/12/2021 (1) 01/01/2025 - 24/12/2031 309.20 16,000 – – – – 16,000 23/12/2022 - 01/12/2025 23/12/2022 (1) 23/12/2023 - 23/12/2032 359.60 16,000 – – – 5,333 16,000 03/07/2023 - 01/07/2026 03/07/2023 (1) 03/07/2024 - 03/07/2033 355.40 15,000 – – – 5,000 15,000 28/06/2024 - 01/06/2027 28/06/2024 (1) 01/01/2028 - 28/06/2034 416.40 12,738 – – – 6,369 12,738 30/06/2025 - 01/06/2028 30/06/2025 (1) 01/01/2029 - 30/06/2035 479.30 – 10,110 – – – 10,110 Total 107,412.00 10,110 32,674 – 16,702 84,848 Malini Moorthy, Legal Counsel Equity incentive plan 01/04/2022 - 01/04/2025 01/04/2022 (1) 01/04/2023 - 01/04/2032 282.50 6,500 – 6,500 – 2,667 – 03/07/2023 - 01/07/2026 03/07/2023 (1) 03/07/2024 - 03/07/2033 355.40 15,000 – 5,000 – 5,000 10,000 28/06/2024 - 01/06/2027 28/06/2024 (1) 01/01/2028 - 28/06/2034 416.40 12,738 – – – 6,369 12,738 30/06/2025 - 01/06/2028 30/06/2025 (1) 01/01/2029 - 30/06/2035 479.30 – 8,812 – – – 8,812 Total 34,238 8,812 11,500 – 14,036 31,550 Luc Truyen, CMO Equity incentive plan 01/10/2021 - 01/10/2024 01/10/2021 (1) 01/01/2025 - 01/10/2026 259.5 24,000 – 24,000 – – – 23/12/2022 - 01/12/2025 23/12/2022 (1) 01/01/2026 - 23/12/2027 359.6 16,000 – – – 5,333 16,000 03/07/2023 - 01/07/2026 03/07/2023 (1) 01/01/2027 - 03/07/2028 355.4 15,000 – – – 5,000 15,000 28/06/2024 - 01/06/2027 28/06/2024 (1) 01/01/2028 - 28/06/2034 416.40 12,738 – – – 6,369 12,738 30/06/2025 - 01/06/2028 30/06/2025 (1) 01/01/2029 - 30/06/2035 479.30 – 8,812 – – – 8,812 Total 67,738 8,812 24,000 – 16,702 52,550 Information regarding the reported financial year Opening balance During the Year Closing balance Name of Directors, Position Specification plan Performance period Award date Vesting date Exercise period Exercise price of stock option in € Stock options held at the beginning of the year Stock options awarded Stock options exercised Stock options forfeited Stock options vested Stock options held at the end of the year argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Remuneration Report and Compensation Statement 148
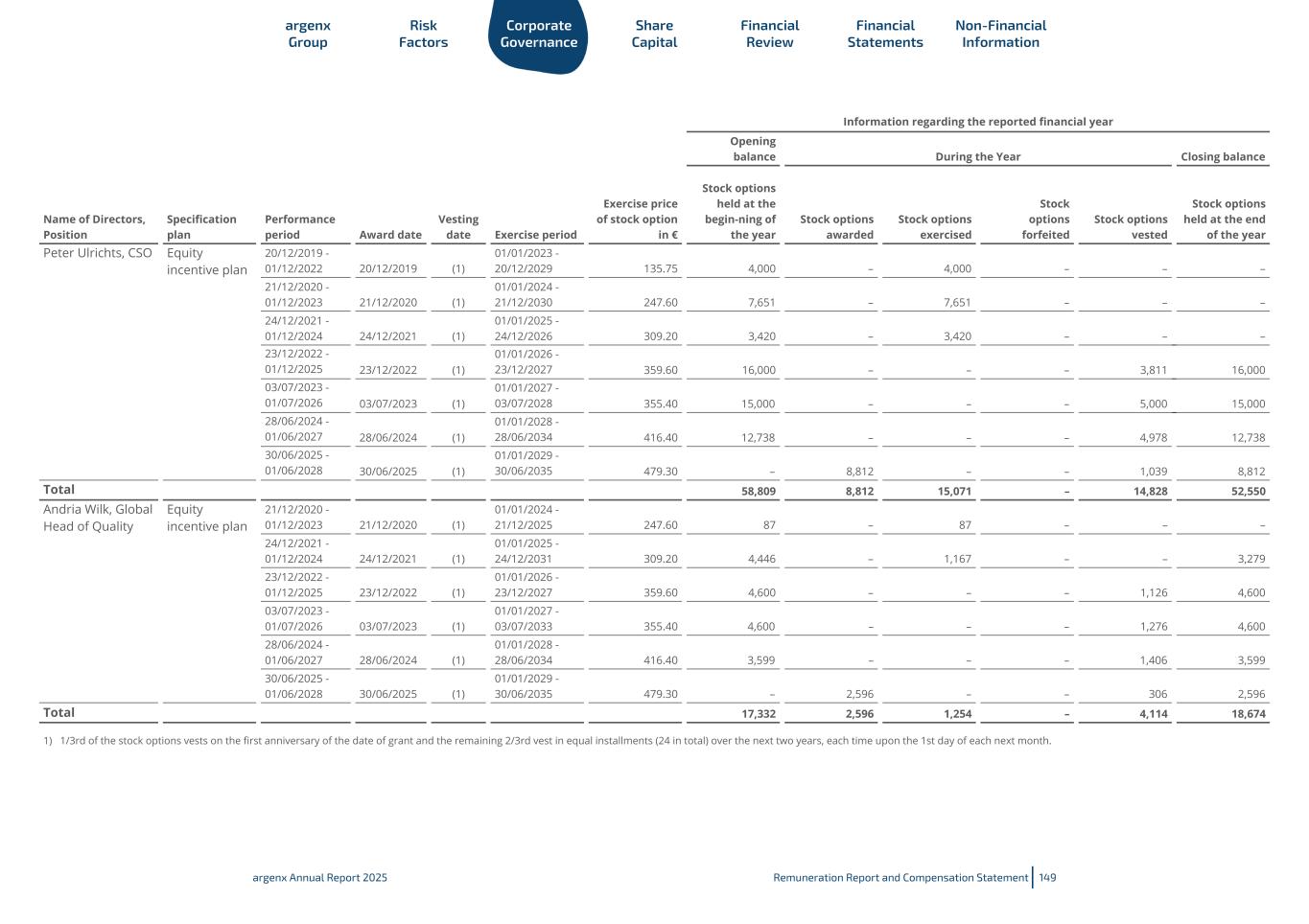
Peter Ulrichts, CSO Equity incentive plan 20/12/2019 - 01/12/2022 20/12/2019 (1) 01/01/2023 - 20/12/2029 135.75 4,000 – 4,000 – – – 21/12/2020 - 01/12/2023 21/12/2020 (1) 01/01/2024 - 21/12/2030 247.60 7,651 – 7,651 – – – 24/12/2021 - 01/12/2024 24/12/2021 (1) 01/01/2025 - 24/12/2026 309.20 3,420 – 3,420 – – – 23/12/2022 - 01/12/2025 23/12/2022 (1) 01/01/2026 - 23/12/2027 359.60 16,000 – – – 3,811 16,000 03/07/2023 - 01/07/2026 03/07/2023 (1) 01/01/2027 - 03/07/2028 355.40 15,000 – – – 5,000 15,000 28/06/2024 - 01/06/2027 28/06/2024 (1) 01/01/2028 - 28/06/2034 416.40 12,738 – – – 4,978 12,738 30/06/2025 - 01/06/2028 30/06/2025 (1) 01/01/2029 - 30/06/2035 479.30 – 8,812 – – 1,039 8,812 Total 58,809 8,812 15,071 – 14,828 52,550 Andria Wilk, Global Head of Quality Equity incentive plan 21/12/2020 - 01/12/2023 21/12/2020 (1) 01/01/2024 - 21/12/2025 247.60 87 – 87 – – – 24/12/2021 - 01/12/2024 24/12/2021 (1) 01/01/2025 - 24/12/2031 309.20 4,446 – 1,167 – – 3,279 23/12/2022 - 01/12/2025 23/12/2022 (1) 01/01/2026 - 23/12/2027 359.60 4,600 – – – 1,126 4,600 03/07/2023 - 01/07/2026 03/07/2023 (1) 01/01/2027 - 03/07/2033 355.40 4,600 – – – 1,276 4,600 28/06/2024 - 01/06/2027 28/06/2024 (1) 01/01/2028 - 28/06/2034 416.40 3,599 – – – 1,406 3,599 30/06/2025 - 01/06/2028 30/06/2025 (1) 01/01/2029 - 30/06/2035 479.30 – 2,596 – – 306 2,596 Total 17,332 2,596 1,254 – 4,114 18,674 1) 1/3rd of the stock options vests on the first anniversary of the date of grant and the remaining 2/3rd vest in equal installments (24 in total) over the next two years, each time upon the 1st day of each next month. Information regarding the reported financial year Opening balance During the Year Closing balance Name of Directors, Position Specification plan Performance period Award date Vesting date Exercise period Exercise price of stock option in € Stock options held at the beginning of the year Stock options awarded Stock options exercised Stock options forfeited Stock options vested Stock options held at the end of the year argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Remuneration Report and Compensation Statement 149
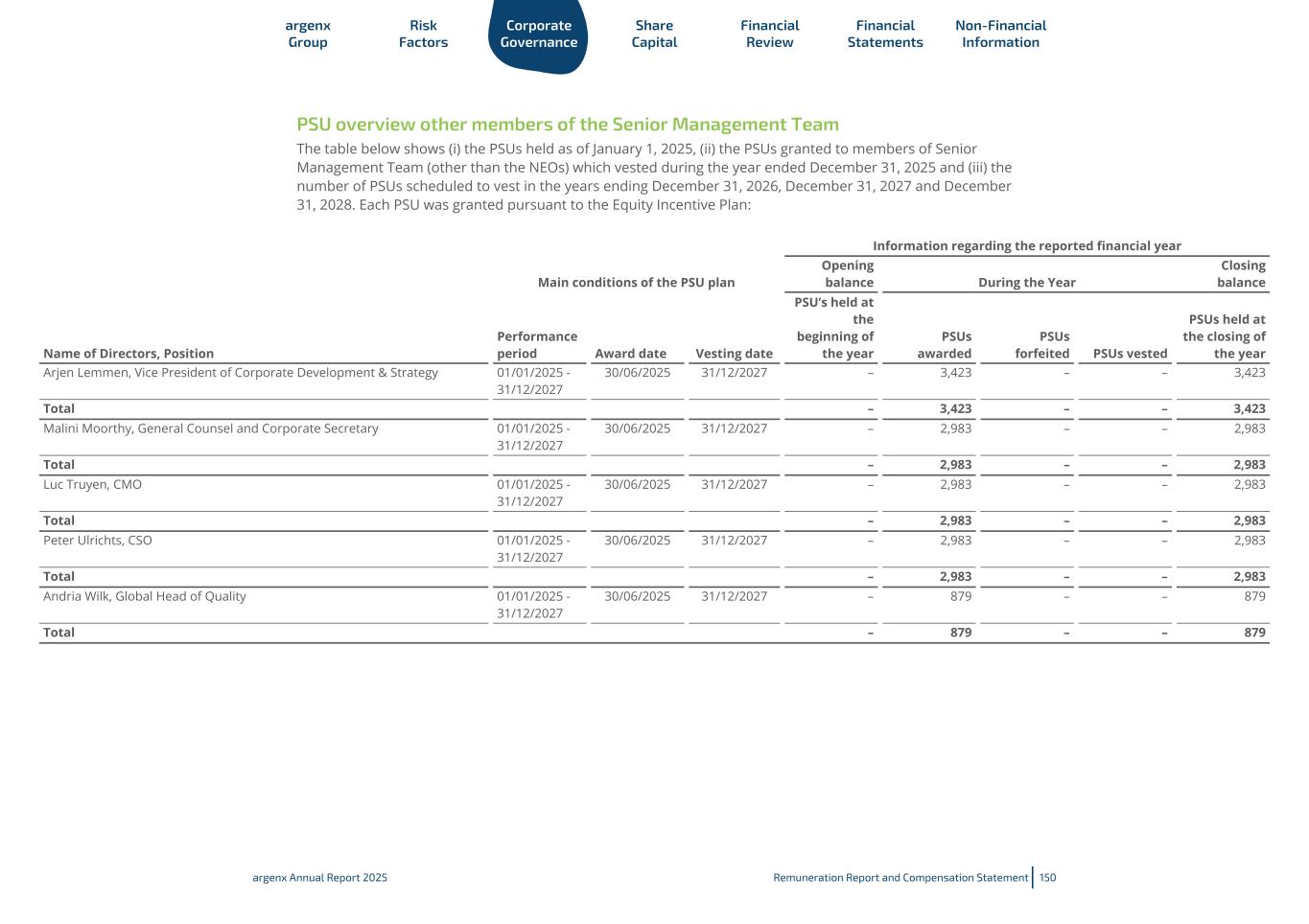
PSU overview other members of the Senior Management Team The table below shows (i) the PSUs held as of January 1, 2025, (ii) the PSUs granted to members of Senior Management Team (other than the NEOs) which vested during the year ended December 31, 2025 and (iii) the number of PSUs scheduled to vest in the years ending December 31, 2026, December 31, 2027 and December 31, 2028. Each PSU was granted pursuant to the Equity Incentive Plan: Arjen Lemmen, Vice President of Corporate Development & Strategy 01/01/2025 - 31/12/2027 30/06/2025 31/12/2027 – 3,423 – – 3,423 Total – 3,423 – – 3,423 Malini Moorthy, General Counsel and Corporate Secretary 01/01/2025 - 31/12/2027 30/06/2025 31/12/2027 – 2,983 – – 2,983 Total – 2,983 – – 2,983 Luc Truyen, CMO 01/01/2025 - 31/12/2027 30/06/2025 31/12/2027 – 2,983 – – 2,983 Total – 2,983 – – 2,983 Peter Ulrichts, CSO 01/01/2025 - 31/12/2027 30/06/2025 31/12/2027 – 2,983 – – 2,983 Total – 2,983 – – 2,983 Andria Wilk, Global Head of Quality 01/01/2025 - 31/12/2027 30/06/2025 31/12/2027 – 879 – – 879 Total – 879 – – 879 Information regarding the reported financial year Main conditions of the PSU plan Opening balance During the Year Closing balance Name of Directors, Position Performance period Award date Vesting date PSU’s held at the beginning of the year PSUs awarded PSUs forfeited PSUs vested PSUs held at the closing of the year argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Remuneration Report and Compensation Statement 150
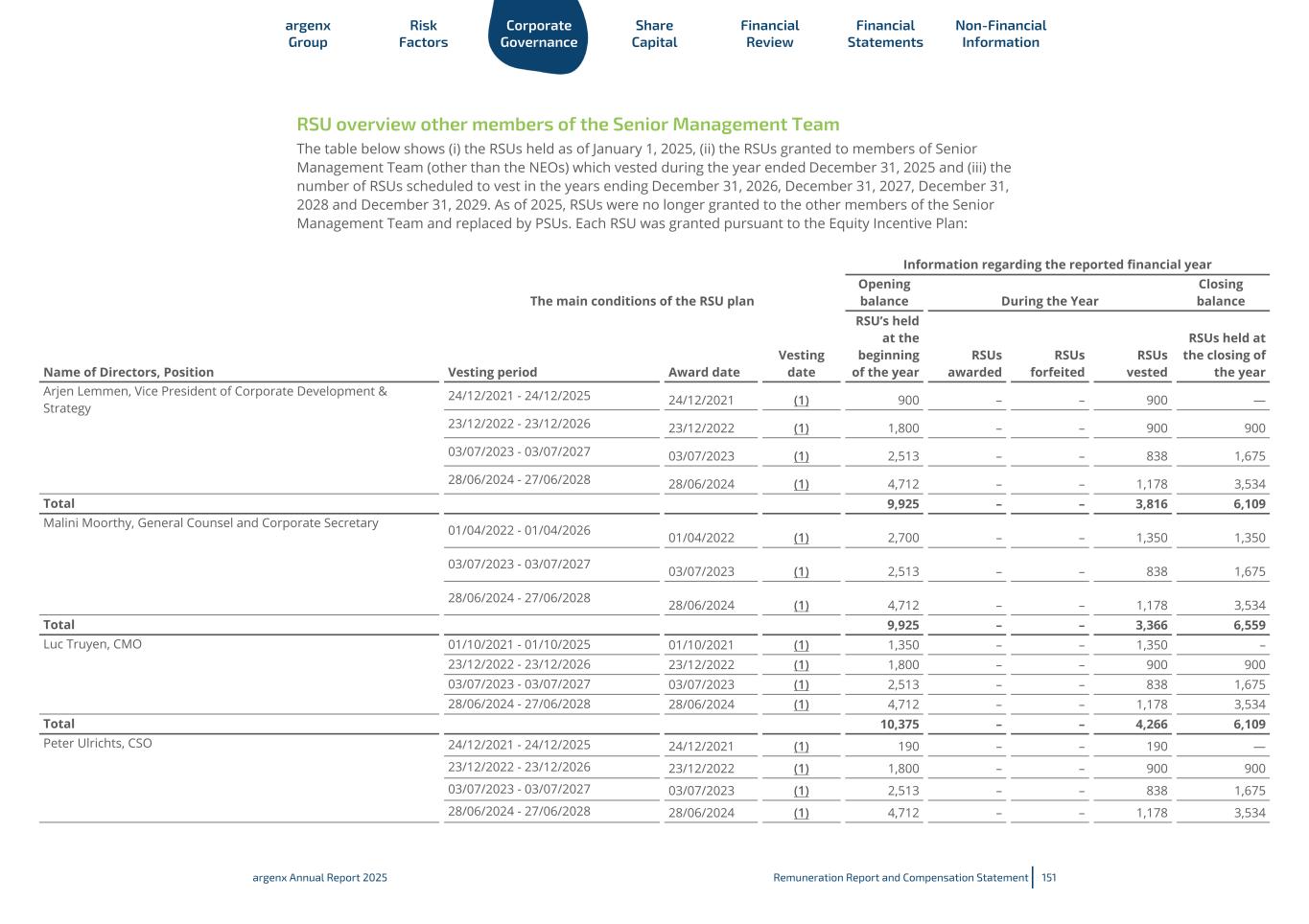
RSU overview other members of the Senior Management Team The table below shows (i) the RSUs held as of January 1, 2025, (ii) the RSUs granted to members of Senior Management Team (other than the NEOs) which vested during the year ended December 31, 2025 and (iii) the number of RSUs scheduled to vest in the years ending December 31, 2026, December 31, 2027, December 31, 2028 and December 31, 2029. As of 2025, RSUs were no longer granted to the other members of the Senior Management Team and replaced by PSUs. Each RSU was granted pursuant to the Equity Incentive Plan: Arjen Lemmen, Vice President of Corporate Development & Strategy 24/12/2021 - 24/12/2025 24/12/2021 (1) 900 – – 900 — 23/12/2022 - 23/12/2026 23/12/2022 (1) 1,800 – – 900 900 03/07/2023 - 03/07/2027 03/07/2023 (1) 2,513 – – 838 1,675 28/06/2024 - 27/06/2028 28/06/2024 (1) 4,712 – – 1,178 3,534 Total 9,925 – – 3,816 6,109 Malini Moorthy, General Counsel and Corporate Secretary 01/04/2022 - 01/04/2026 01/04/2022 (1) 2,700 – – 1,350 1,350 03/07/2023 - 03/07/2027 03/07/2023 (1) 2,513 – – 838 1,675 28/06/2024 - 27/06/2028 28/06/2024 (1) 4,712 – – 1,178 3,534 Total 9,925 – – 3,366 6,559 Luc Truyen, CMO 01/10/2021 - 01/10/2025 01/10/2021 (1) 1,350 – – 1,350 – 23/12/2022 - 23/12/2026 23/12/2022 (1) 1,800 – – 900 900 03/07/2023 - 03/07/2027 03/07/2023 (1) 2,513 – – 838 1,675 28/06/2024 - 27/06/2028 28/06/2024 (1) 4,712 – – 1,178 3,534 Total 10,375 – – 4,266 6,109 Peter Ulrichts, CSO 24/12/2021 - 24/12/2025 24/12/2021 (1) 190 – – 190 — 23/12/2022 - 23/12/2026 23/12/2022 (1) 1,800 – – 900 900 03/07/2023 - 03/07/2027 03/07/2023 (1) 2,513 – – 838 1,675 28/06/2024 - 27/06/2028 28/06/2024 (1) 4,712 – – 1,178 3,534 Information regarding the reported financial year The main conditions of the RSU plan Opening balance During the Year Closing balance Name of Directors, Position Vesting period Award date Vesting date RSU’s held at the beginning of the year RSUs awarded RSUs forfeited RSUs vested RSUs held at the closing of the year argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Remuneration Report and Compensation Statement 151
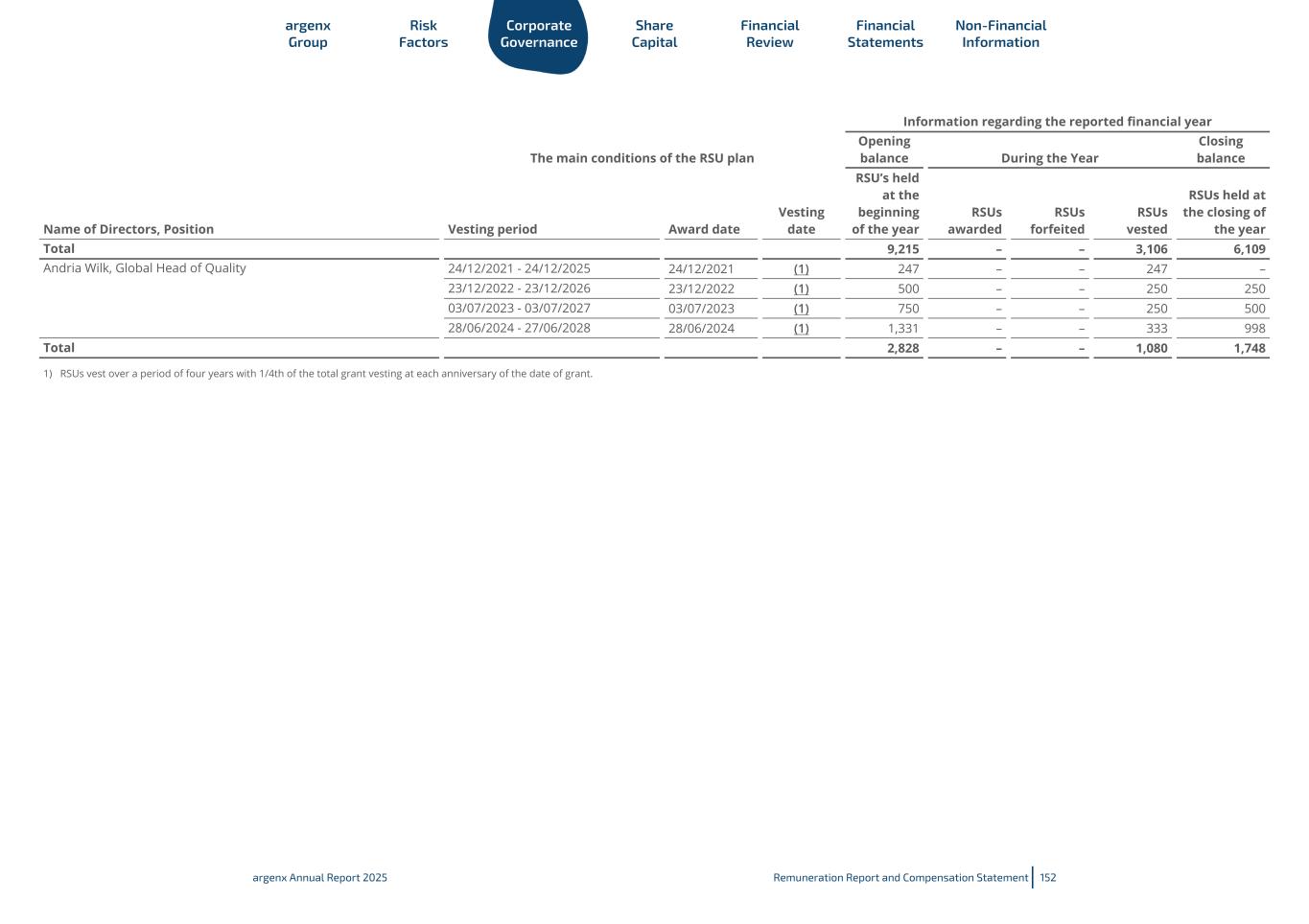
Total 9,215 – – 3,106 6,109 Andria Wilk, Global Head of Quality 24/12/2021 - 24/12/2025 24/12/2021 (1) 247 – – 247 – 23/12/2022 - 23/12/2026 23/12/2022 (1) 500 – – 250 250 03/07/2023 - 03/07/2027 03/07/2023 (1) 750 – – 250 500 28/06/2024 - 27/06/2028 28/06/2024 (1) 1,331 – – 333 998 Total 2,828 – – 1,080 1,748 1) RSUs vest over a period of four years with 1/4th of the total grant vesting at each anniversary of the date of grant. Information regarding the reported financial year The main conditions of the RSU plan Opening balance During the Year Closing balance Name of Directors, Position Vesting period Award date Vesting date RSU’s held at the beginning of the year RSUs awarded RSUs forfeited RSUs vested RSUs held at the closing of the year argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Remuneration Report and Compensation Statement 152
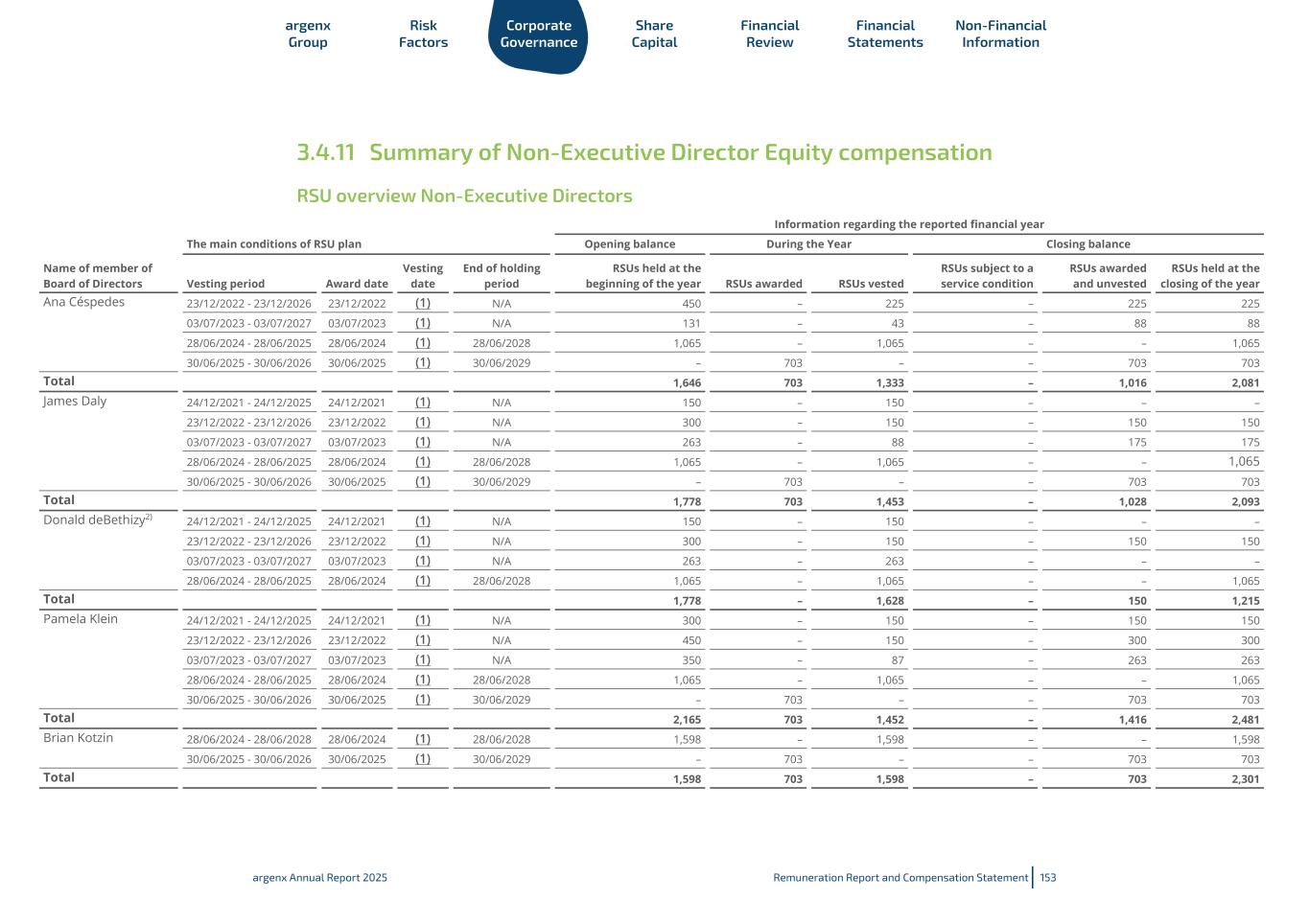
3.4.11 Summary of Non-Executive Director Equity compensation RSU overview Non-Executive Directors Ana Céspedes 23/12/2022 - 23/12/2026 23/12/2022 (1) N/A 450 – 225 – 225 225 03/07/2023 - 03/07/2027 03/07/2023 (1) N/A 131 – 43 – 88 88 28/06/2024 - 28/06/2025 28/06/2024 (1) 28/06/2028 1,065 – 1,065 – – 1,065 30/06/2025 - 30/06/2026 30/06/2025 (1) 30/06/2029 – 703 – – 703 703 Total 1,646 703 1,333 – 1,016 2,081 James Daly 24/12/2021 - 24/12/2025 24/12/2021 (1) N/A 150 – 150 – – – 23/12/2022 - 23/12/2026 23/12/2022 (1) N/A 300 – 150 – 150 150 03/07/2023 - 03/07/2027 03/07/2023 (1) N/A 263 – 88 – 175 175 28/06/2024 - 28/06/2025 28/06/2024 (1) 28/06/2028 1,065 – 1,065 – – 1,065 30/06/2025 - 30/06/2026 30/06/2025 (1) 30/06/2029 – 703 – – 703 703 Total 1,778 703 1,453 – 1,028 2,093 Donald deBethizy2) 24/12/2021 - 24/12/2025 24/12/2021 (1) N/A 150 – 150 – – – 23/12/2022 - 23/12/2026 23/12/2022 (1) N/A 300 – 150 – 150 150 03/07/2023 - 03/07/2027 03/07/2023 (1) N/A 263 – 263 – – – 28/06/2024 - 28/06/2025 28/06/2024 (1) 28/06/2028 1,065 – 1,065 – – 1,065 Total 1,778 – 1,628 – 150 1,215 Pamela Klein 24/12/2021 - 24/12/2025 24/12/2021 (1) N/A 300 – 150 – 150 150 23/12/2022 - 23/12/2026 23/12/2022 (1) N/A 450 – 150 – 300 300 03/07/2023 - 03/07/2027 03/07/2023 (1) N/A 350 – 87 – 263 263 28/06/2024 - 28/06/2025 28/06/2024 (1) 28/06/2028 1,065 – 1,065 – – 1,065 30/06/2025 - 30/06/2026 30/06/2025 (1) 30/06/2029 – 703 – – 703 703 Total 2,165 703 1,452 – 1,416 2,481 Brian Kotzin 28/06/2024 - 28/06/2028 28/06/2024 (1) 28/06/2028 1,598 – 1,598 – – 1,598 30/06/2025 - 30/06/2026 30/06/2025 (1) 30/06/2029 – 703 – – 703 703 Total 1,598 703 1,598 – 703 2,301 Information regarding the reported financial year The main conditions of RSU plan Opening balance During the Year Closing balance Name of member of Board of Directors Vesting period Award date Vesting date End of holding period RSUs held at the beginning of the year RSUs awarded RSUs vested RSUs subject to a service condition RSUs awarded and unvested RSUs held at the closing of the year argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Remuneration Report and Compensation Statement 153
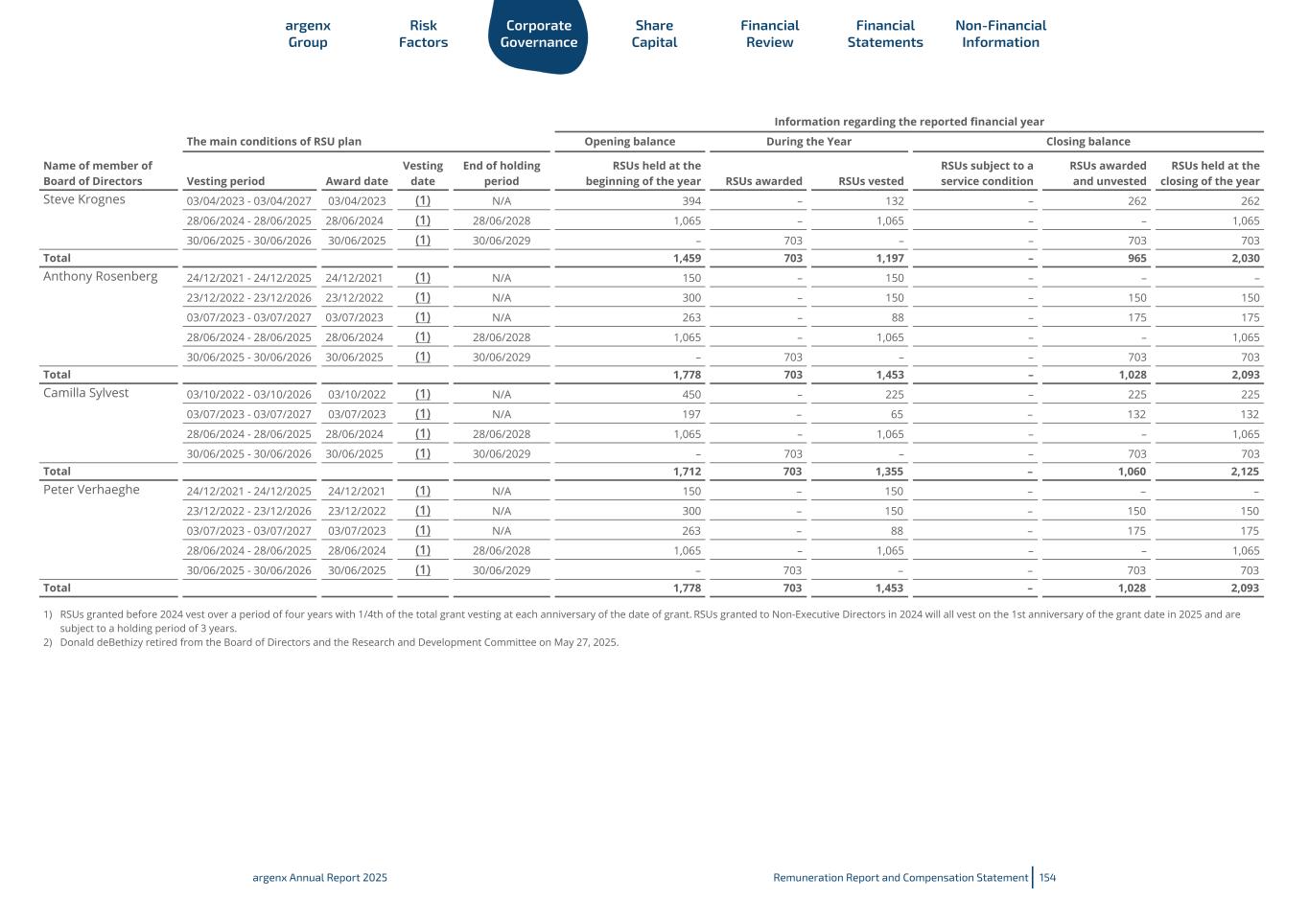
Steve Krognes 03/04/2023 - 03/04/2027 03/04/2023 (1) N/A 394 – 132 – 262 262 28/06/2024 - 28/06/2025 28/06/2024 (1) 28/06/2028 1,065 – 1,065 – – 1,065 30/06/2025 - 30/06/2026 30/06/2025 (1) 30/06/2029 – 703 – – 703 703 Total 1,459 703 1,197 – 965 2,030 Anthony Rosenberg 24/12/2021 - 24/12/2025 24/12/2021 (1) N/A 150 – 150 – – – 23/12/2022 - 23/12/2026 23/12/2022 (1) N/A 300 – 150 – 150 150 03/07/2023 - 03/07/2027 03/07/2023 (1) N/A 263 – 88 – 175 175 28/06/2024 - 28/06/2025 28/06/2024 (1) 28/06/2028 1,065 – 1,065 – – 1,065 30/06/2025 - 30/06/2026 30/06/2025 (1) 30/06/2029 – 703 – – 703 703 Total 1,778 703 1,453 – 1,028 2,093 Camilla Sylvest 03/10/2022 - 03/10/2026 03/10/2022 (1) N/A 450 – 225 – 225 225 03/07/2023 - 03/07/2027 03/07/2023 (1) N/A 197 – 65 – 132 132 28/06/2024 - 28/06/2025 28/06/2024 (1) 28/06/2028 1,065 – 1,065 – – 1,065 30/06/2025 - 30/06/2026 30/06/2025 (1) 30/06/2029 – 703 – – 703 703 Total 1,712 703 1,355 – 1,060 2,125 Peter Verhaeghe 24/12/2021 - 24/12/2025 24/12/2021 (1) N/A 150 – 150 – – – 23/12/2022 - 23/12/2026 23/12/2022 (1) N/A 300 – 150 – 150 150 03/07/2023 - 03/07/2027 03/07/2023 (1) N/A 263 – 88 – 175 175 28/06/2024 - 28/06/2025 28/06/2024 (1) 28/06/2028 1,065 – 1,065 – – 1,065 30/06/2025 - 30/06/2026 30/06/2025 (1) 30/06/2029 – 703 – – 703 703 Total 1,778 703 1,453 – 1,028 2,093 1) RSUs granted before 2024 vest over a period of four years with 1/4th of the total grant vesting at each anniversary of the date of grant. RSUs granted to Non-Executive Directors in 2024 will all vest on the 1st anniversary of the grant date in 2025 and are subject to a holding period of 3 years. 2) Donald deBethizy retired from the Board of Directors and the Research and Development Committee on May 27, 2025. Information regarding the reported financial year The main conditions of RSU plan Opening balance During the Year Closing balance Name of member of Board of Directors Vesting period Award date Vesting date End of holding period RSUs held at the beginning of the year RSUs awarded RSUs vested RSUs subject to a service condition RSUs awarded and unvested RSUs held at the closing of the year argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Remuneration Report and Compensation Statement 154
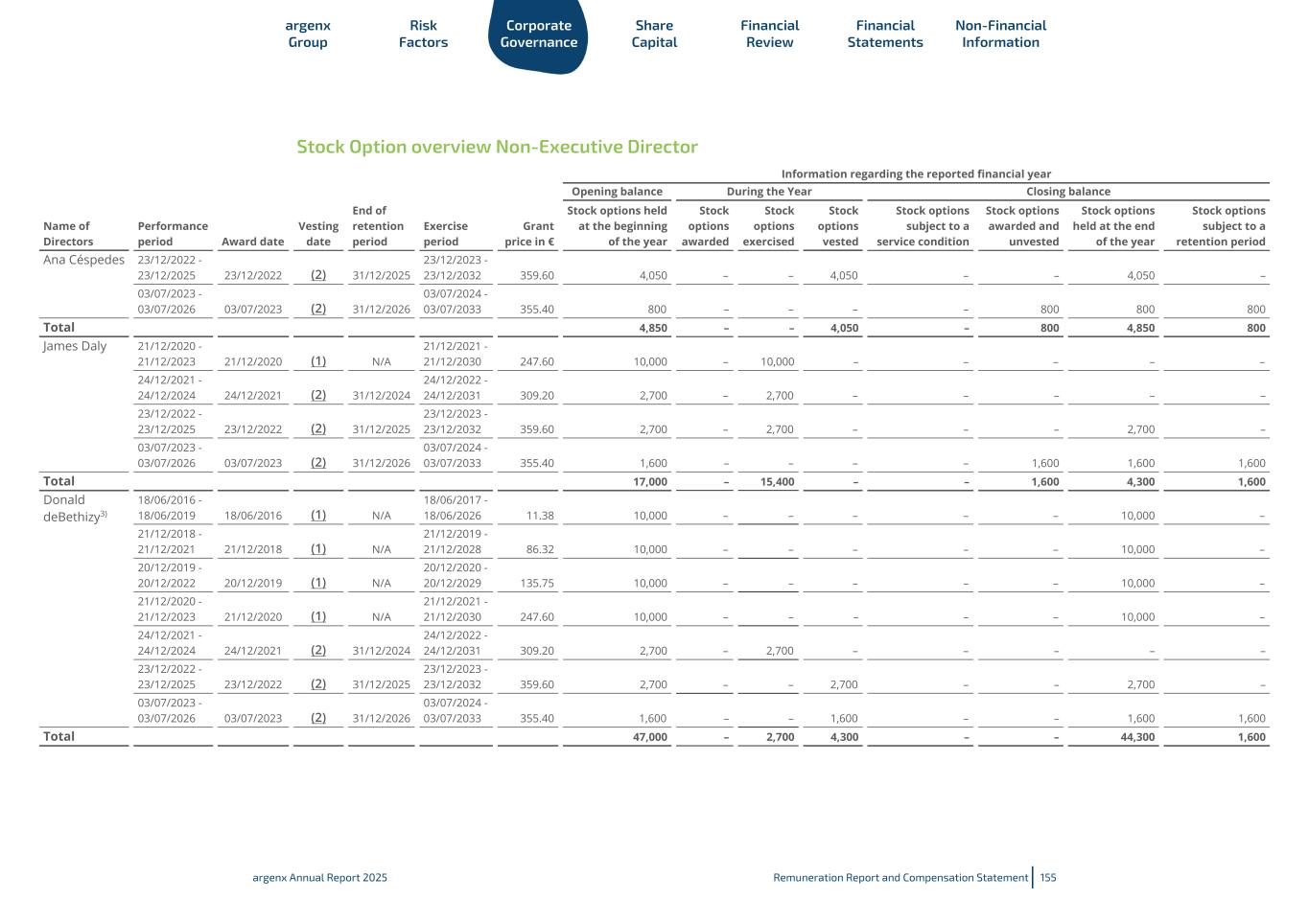
Stock Option overview Non-Executive Director Ana Céspedes 23/12/2022 - 23/12/2025 23/12/2022 (2) 31/12/2025 23/12/2023 - 23/12/2032 359.60 4,050 – – 4,050 – – 4,050 – 03/07/2023 - 03/07/2026 03/07/2023 (2) 31/12/2026 03/07/2024 - 03/07/2033 355.40 800 – – – – 800 800 800 Total 4,850 – – 4,050 – 800 4,850 800 James Daly 21/12/2020 - 21/12/2023 21/12/2020 (1) N/A 21/12/2021 - 21/12/2030 247.60 10,000 – 10,000 – – – – – 24/12/2021 - 24/12/2024 24/12/2021 (2) 31/12/2024 24/12/2022 - 24/12/2031 309.20 2,700 – 2,700 – – – – – 23/12/2022 - 23/12/2025 23/12/2022 (2) 31/12/2025 23/12/2023 - 23/12/2032 359.60 2,700 – 2,700 – – – 2,700 – 03/07/2023 - 03/07/2026 03/07/2023 (2) 31/12/2026 03/07/2024 - 03/07/2033 355.40 1,600 – – – – 1,600 1,600 1,600 Total 17,000 – 15,400 – – 1,600 4,300 1,600 Donald deBethizy3) 18/06/2016 - 18/06/2019 18/06/2016 (1) N/A 18/06/2017 - 18/06/2026 11.38 10,000 – – – – – 10,000 – 21/12/2018 - 21/12/2021 21/12/2018 (1) N/A 21/12/2019 - 21/12/2028 86.32 10,000 – – – – – 10,000 – 20/12/2019 - 20/12/2022 20/12/2019 (1) N/A 20/12/2020 - 20/12/2029 135.75 10,000 – – – – – 10,000 – 21/12/2020 - 21/12/2023 21/12/2020 (1) N/A 21/12/2021 - 21/12/2030 247.60 10,000 – – – – – 10,000 – 24/12/2021 - 24/12/2024 24/12/2021 (2) 31/12/2024 24/12/2022 - 24/12/2031 309.20 2,700 – 2,700 – – – – – 23/12/2022 - 23/12/2025 23/12/2022 (2) 31/12/2025 23/12/2023 - 23/12/2032 359.60 2,700 – – 2,700 – – 2,700 – 03/07/2023 - 03/07/2026 03/07/2023 (2) 31/12/2026 03/07/2024 - 03/07/2033 355.40 1,600 – – 1,600 – – 1,600 1,600 Total 47,000 – 2,700 4,300 – – 44,300 1,600 Information regarding the reported financial year Opening balance During the Year Closing balance Name of Directors Performance period Award date Vesting date End of retention period Exercise period Grant price in € Stock options held at the beginning of the year Stock options awarded Stock options exercised Stock options vested Stock options subject to a service condition Stock options awarded and unvested Stock options held at the end of the year Stock options subject to a retention period argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Remuneration Report and Compensation Statement 155
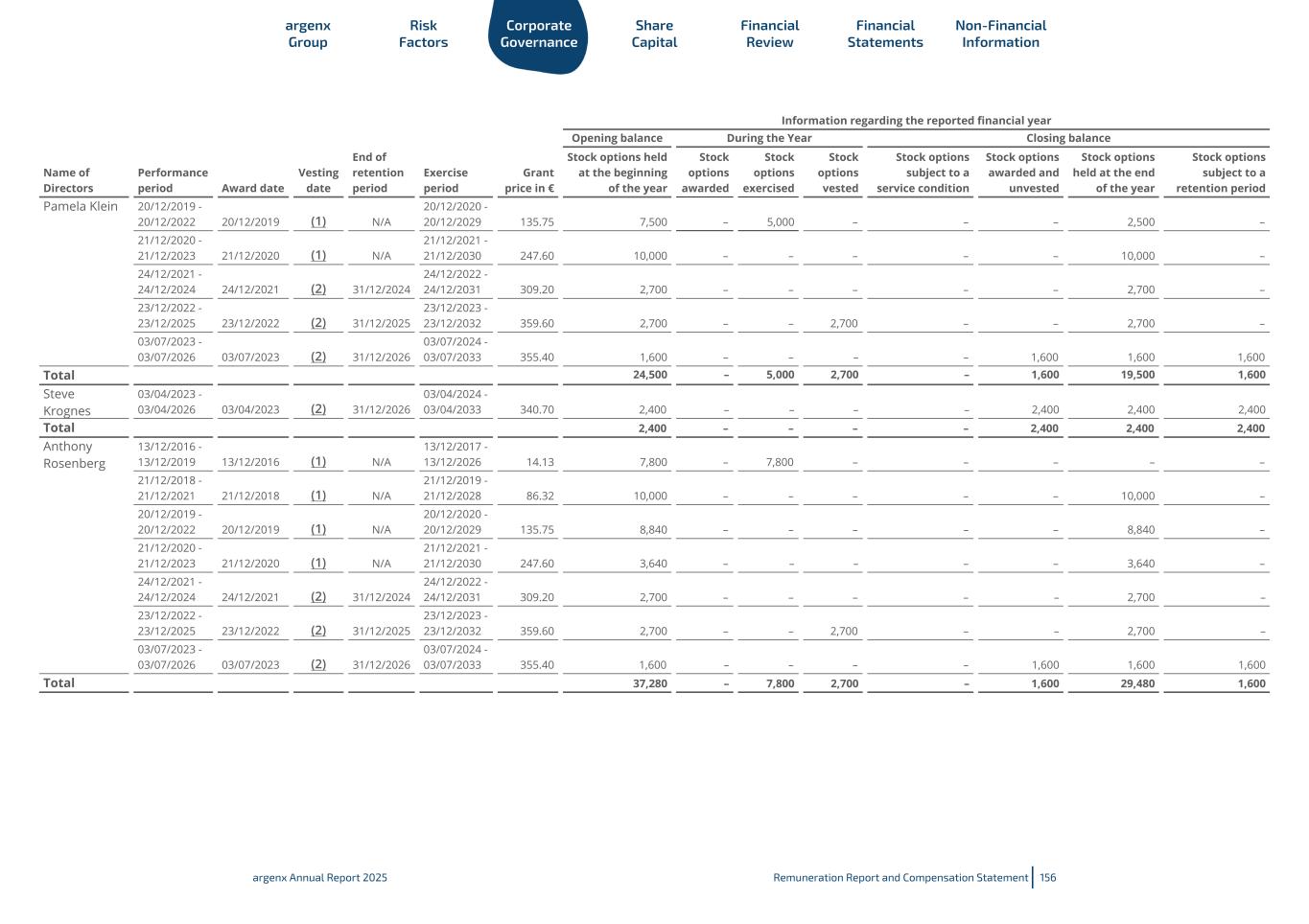
Pamela Klein 20/12/2019 - 20/12/2022 20/12/2019 (1) N/A 20/12/2020 - 20/12/2029 135.75 7,500 – 5,000 – – – 2,500 – 21/12/2020 - 21/12/2023 21/12/2020 (1) N/A 21/12/2021 - 21/12/2030 247.60 10,000 – – – – – 10,000 – 24/12/2021 - 24/12/2024 24/12/2021 (2) 31/12/2024 24/12/2022 - 24/12/2031 309.20 2,700 – – – – – 2,700 – 23/12/2022 - 23/12/2025 23/12/2022 (2) 31/12/2025 23/12/2023 - 23/12/2032 359.60 2,700 – – 2,700 – – 2,700 – 03/07/2023 - 03/07/2026 03/07/2023 (2) 31/12/2026 03/07/2024 - 03/07/2033 355.40 1,600 – – – – 1,600 1,600 1,600 Total 24,500 – 5,000 2,700 – 1,600 19,500 1,600 Steve Krognes 03/04/2023 - 03/04/2026 03/04/2023 (2) 31/12/2026 03/04/2024 - 03/04/2033 340.70 2,400 – – – – 2,400 2,400 2,400 Total 2,400 – – – – 2,400 2,400 2,400 Anthony Rosenberg 13/12/2016 - 13/12/2019 13/12/2016 (1) N/A 13/12/2017 - 13/12/2026 14.13 7,800 – 7,800 – – – – – 21/12/2018 - 21/12/2021 21/12/2018 (1) N/A 21/12/2019 - 21/12/2028 86.32 10,000 – – – – – 10,000 – 20/12/2019 - 20/12/2022 20/12/2019 (1) N/A 20/12/2020 - 20/12/2029 135.75 8,840 – – – – – 8,840 – 21/12/2020 - 21/12/2023 21/12/2020 (1) N/A 21/12/2021 - 21/12/2030 247.60 3,640 – – – – – 3,640 – 24/12/2021 - 24/12/2024 24/12/2021 (2) 31/12/2024 24/12/2022 - 24/12/2031 309.20 2,700 – – – – – 2,700 – 23/12/2022 - 23/12/2025 23/12/2022 (2) 31/12/2025 23/12/2023 - 23/12/2032 359.60 2,700 – – 2,700 – – 2,700 – 03/07/2023 - 03/07/2026 03/07/2023 (2) 31/12/2026 03/07/2024 - 03/07/2033 355.40 1,600 – – – – 1,600 1,600 1,600 Total 37,280 – 7,800 2,700 – 1,600 29,480 1,600 Information regarding the reported financial year Opening balance During the Year Closing balance Name of Directors Performance period Award date Vesting date End of retention period Exercise period Grant price in € Stock options held at the beginning of the year Stock options awarded Stock options exercised Stock options vested Stock options subject to a service condition Stock options awarded and unvested Stock options held at the end of the year Stock options subject to a retention period argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Remuneration Report and Compensation Statement 156

Camilla Sylvest 03/10/2022 - 03/10/2025 03/10/2022 (2) 31/12/2025 03/10/2023 - 03/10/2032 368.50 4,050 – – 4,050 – – 4,050 – 03/07/2023 - 03/07/2026 03/07/2023 (2) 31/12/2026 03/07/2024 - 03/07/2033 355.40 1,200 – – – – 1,200 1,200 1,200 Total 5,250 – – 4,050 – 1,200 5,250 1,200 Peter Verhaeghe 18/06/2016 - 18/06/2019 18/06/2016 (1) 31/12/2019 01/01/2020 - 18/06/2026 11.38 4,000 – 4,000 – – – – – 21/12/2018 - 21/12/2021 21/12/2018 (1) 31/12/2021 01/01/2022 - 21/12/2028 86.32 10,000 – – – – – 10,000 – 20/12/2019 - 20/12/2022 20/12/2019 (1) 31/12/2022 01/01/2023 - 20/12/2029 135.75 10,000 – – – – – 10,000 – 21/12/2020 - 21/12/2023 21/12/2020 (1) 31/12/2023 01/01/2024 - 21/12/2030 247.60 10,000 – – – – – 10,000 – 24/12/2021 - 24/12/2024 24/12/2021 (2) 31/12/2024 01/01/2025 - 24/12/2031 309.20 2,700 – – – – – 2,700 – 23/12/2022 - 23/12/2025 23/12/2022 (2) 31/12/2025 01/01/2026 - 23/12/2032 359.60 2,700 – – 2,700 – – 2,700 – 03/07/2023 - 03/07/2026 03/07/2023 (2) 31/12/2026 01/01/2027 - 03/07/2033 355.40 1,600 – – – – 1,600 1,600 1,600 Total 41,000 – 4,000 2,700 – 1,600 37,000 1,600 1) 1/3 of stock options vests on the first anniversary of the grant and the remaining 2/3rd vest in equal monthly installments (24 in total) over the next two years, each time upon the 1st day of the month. 2) Stock options vest upon third anniversary of the grant. 3) Donald deBethizy retired from the Board of Directors, the Remuneration and Nomination and Research and Development Committee on May 27, 2025. Information regarding the reported financial year Opening balance During the Year Closing balance Name of Directors Performance period Award date Vesting date End of retention period Exercise period Grant price in € Stock options held at the beginning of the year Stock options awarded Stock options exercised Stock options vested Stock options subject to a service condition Stock options awarded and unvested Stock options held at the end of the year Stock options subject to a retention period argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Remuneration Report and Compensation Statement 157

3.5 Corporate Governance – Nasdaq Listing Rules As a foreign private issuer, the Nasdaq Listing Rules include certain accommodations in the corporate governance requirements that allow foreign private issuers to follow “home country” corporate governance practices in lieu of the otherwise applicable Nasdaq corporate governance standards. We intend to rely on certain exemptions for foreign private issuers and to follow Dutch corporate governance practices in lieu of the Nasdaq corporate governance rules. The following is a summary of the significant ways in which our corporate governance practices differ from those required by the Nasdaq Listing Rules with which we are not required to comply: • Quorum at Shareholder Meetings. In accordance with Dutch law and generally accepted business practices in the Netherlands, our Articles of Association do not provide quorum requirements generally applicable to general meetings of shareholders. To that extent, our practice varies from the requirement of Nasdaq Listing Rule 5620(c), which requires an issuer to provide in its bylaws for a generally applicable quorum, and that such quorum may not be less than one-third of the outstanding voting stock. • Solicitation of Proxies. Although we must provide shareholders with an agenda and other relevant documents ahead of any General Meeting, Dutch law does not have a regulatory regime for the solicitation of proxies, and the solicitation of proxies is not a generally accepted business practice in the Netherlands. Thus, our practice varies from the requirement of Nasdaq Listing Rule 5620(b). • Shareholder Approval. We follow certain Dutch shareholder approval requirements for the issuance of securities in connection with certain events such as the acquisition of stock or assets of another company, the establishment of or amendments to equity-based compensation plans for employees, a change of control of us and certain private placements. To this extent, our practice varies from the requirements of Nasdaq Rule 5635, which generally requires an issuer to obtain shareholder approval for the issuance of securities in connection with such events. • Distribution of Annual Reports. We do not follow Nasdaq Listing Rule 5250(d), which requires companies to make available copies of their annual reports containing audited financial statements to their shareholders. The distribution of our annual reports to shareholders is not required under Dutch corporate law or Dutch securities laws. Furthermore, it is generally accepted business practice for Dutch companies not to distribute annual reports. In part, this is because the Dutch system of bearer shares has made it impractical to keep a current list of holders of the bearer shares in order to distribute the annual reports. Instead, we make our Annual Report available at our corporate head office in the Netherlands (and at the offices of our Dutch listing agent as stated in the convening notice for the meeting) no later than 42 days prior to convocation of any annual General Meeting. In addition, we post a copy of our annual reports on our website prior to our annual General Meeting. 3.6 Share Ownership For information regarding the share ownership of our directors and members of our executive committee, please refer to Section “Remuneration Report and Compensation Statement” and Section “Share Classes and Principal Shareholders”, Subsection “Major Shareholders”. 3.7 Insider Trading We have an insider trading policy in place that complies with MAR. The insider trading policy is intended to maintain confidentiality of inside information (as defined under MAR), refrain from market manipulation and comply with the obligations of argenx under MAR, the Exchange Act and other applicable securities laws. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Corporate Governance – Nasdaq Listing Rules 158

3.8 Cybersecurity 3.8.1 Information Security Risk Management and Strategy Our approach to risk management is designed to identify, assess, prioritize and manage major risk exposures that could affect our ability to execute our corporate strategy and fulfill our business objectives. As part of our information security and privacy program, the Information Security and Management System (the ISMS), we perform risk assessments in which we map and prioritize information security risks identified through the processes described below, including risks associated with our use of third-party service providers. These assessments inform our ISMS strategies and oversight processes and are included with other enterprise risks as part of our broader enterprise risk management. We view information security risks as one of the key risks categories we face. IT system vendors are subject to security review and audits. For more information regarding the cybersecurity-related risks we face, please refer to Section 2.7 “Our business and operations could suffer in the event of system failures or unauthorized or inappropriate use of or access to our systems”. Our processes for assessing, identifying and managing information security risks and vulnerabilities are embedded across our business as part of our ISMS. Among other things, we conduct audits and tests of our information systems (including review and assessment by independent third-party advisors, who assess and report on the maturity of our security measures and help identify areas for continued focus and improvement) and review information security threat information published by government entities and other organizations in which we participate. We conduct training on data security matters for our employees to be aware and vigilant against potential data security risks and data privacy is incorporated into our overall compliance training, such as through privacy-specific training for employees and contractors. Phishing training is also implemented regularly, which includes mock phishing emails to test employee vigilance. In addition, employees are required to read and acknowledge information security policies that are relevant to their specific role. We also have implemented and maintain information security incident response plans, which include processes to triage, assess severity for, escalate, contain, investigate and remediate information security incidents, as well as to comply with potentially applicable legal obligations and mitigate brand and reputational damage. 3.8.2 Information Security Governance and Oversight Our ISMS enables our Board of Directors to establish a mutual understanding with our Senior Management Team of the effectiveness of our information security risk management practices and capabilities, including the division of responsibilities for reviewing our information security risk exposure and risk tolerance, tracking emerging information risks and ensuring proper escalation of certain key risks for periodic review by the Board of Directors and its committees. As part of its broader risk oversight activities, the Board of Directors oversees risks from information security threats, both directly and through the Audit and Compliance Committee. The Audit and Compliance Committee also oversees our internal control over financial reporting. As an element of its cybersecurity oversight activities, the Audit and Compliance Committee regularly reviews the results of our enterprise risk assessments, including information security risk assessments, as well as management's strategies to detect, monitor and manage such risks and related risk assessment and risk management policies. The Digital Technology lead for Security and Compliance provides regular updates on cyber risks, cyber security matters and progress on the cybersecurity programme to the Global Risk Management Committee. Additionally, the data protection officer provides regular updates to the Senior Management Team, and the Audit and Compliance Committee as a component of the Audit and Compliance Committee’s compliance updates. The data protection officer also regularly reports to the Global Corporate Compliance Committee, the Global Risk Management Committee and the General Counsel and Corporate Secretary on matters such as the status of the organizational privacy plan, data breaches and routine programs. In addition to these regularly scheduled updates from the data protection officer, the Global Head of Digital Technologies reports to the Audit and Compliance Committee or the full Board of Directors, as appropriate, on how certain information security risks are being managed and progress towards agreed mitigation goals, as well as any potential material risks from cybersecurity threats that have been detected by the information security team. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Cybersecurity 159

Our information security team is responsible for day-to-day identification, assessment and management of the information security risks we face. Our Global Head of Digital Technologies has almost 25 years of experience in information management systems in life sciences and the managers reporting to the Global Head of Digital Technologies have over 25 cumulative years of experience in information security. Our incident response and data breach procedures seek to promote the timely detection, reporting, and investigation of all security incidents, as well as the timely notification of any reportable breaches (including any material cybersecurity incidents and personal data breaches) to the competent authorities and the timely communication to the affected individuals, where relevant. We maintain records of breaches on our quarterly corporate risk dashboard and our personal data breach register, and we monitor and regularly report our data breach metrics to the Senior Management Team, including the Audit and Compliance Committee, the global corporate compliance committee, and the global risk management committee. In addition to the ordinary-course Board of Directors and Audit and Compliance Committee reporting and oversight described above, we also maintain disclosure controls and procedures designed for prompt reporting to the Board of Directors and timely public disclosure, as appropriate, of material events, including information security risks. 3.9 Risk Appetite & Control Before reading this section, please carefully review the following cautionary statement: In this section we will make the required disclosures regarding our risk appetite and mitigating actions. We fully take the risk mitigation actions and risk management described in this section into account while preparing the description of the main risks and uncertainties we face, as set out in Section “Risk Factors”. Any mitigating language used in this section does not have any impact on the risks and uncertainties we face or their potential adverse effects as they are described in Section “Risk Factors”. Section “Risk Factors” describes the main risks and uncertainties we face already fully having taken into account our risk management and the risk mitigating actions described herein. 3.9.1 Introduction This section Section 3provides a general description of our willingness to mitigate the risks and uncertainties we face (also called our ‘risk appetite’), and to give a description of the mitigating actions we have taken with regard to our most relevant risks. 3.9.2 Controlling Actions We Take with Regard to Our Most Relevant Risks and Uncertainties The following is a description of the main risks and uncertainties we face (being the first risk of each category of risk factors set out in Section “Risk Factors”) and a description of the measures we took to control them. A description of the expected impact upon materialization of these risks is included for each risk in Section “Risk Factors”. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Cybersecurity 160

The commercial success of our products and product candidates, including in new indications or methods of administration, will depend on the degree of market acceptance. We plan to focus on the successful commercialization of the products and product candidates after they are launched. We aim to further expand and develop our sales and marketing organization, including by attracting and retaining top talent for key positions such as the recent hire for the position of the chief commercial officer, enter into and/or amend where necessary collaboration arrangements with third parties, outsource certain functions to third parties, or use some combination of each to promote market acceptance of our products and product candidates. We seek to execute on our business model and long term version by advancing our core focus on innovation through identifying and selecting new product candidates in our IIP program, expanding our intellectual property portfolio, further expanding our labels and developing next-generation products such as the PFS. Failure to successfully identify, select and develop our products in other indications, or additional products or product candidates could impair our ability to grow. We will continue to focus on the IIP program, which is part of our method of identifying, selecting and developing product candidates and product based on scientific data and rationale. In addition, we remain committed to data driven decision making by driving innovation through cultivation of strategic opportunities across clinical stage development pathways. As part of this strategy, we aim to create optionality within our product portfolio, which is why we are advancing and prioritizing the continued development of adimanebart and empasiprubart. We rely, and expect to continue to rely, on third parties to conduct some of our research activities, manufacturing and clinical trials and for parts of the development and commercialization of our existing and future research programs, products and product candidates. If our relationships with such third parties are not successful, our business may be adversely affected. Project management is crucial to our success, and thus we seek to maintain a rich pipeline of possible collaboration partners, as well foster good relationships with existing and potential future partners to spread operational risks. As part of our contracting strategy, we seek to align incentives with our partners by structuring agreements that emphasize payment for outcomes rather than activities. Both during selection of our partners as well during the duration of our contracts with them, we apply continued and integrated quality management focused oversight to support performance and mitigate any potential risks. RISK FACTOR MEASURES TAKEN TO CONTROL THESE RISKS argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Risk Appetite & Control 161

We may be unable to adequately maintain, enforce or protect our intellectual property rights in products, product candidates and platform technologies which could adversely affect our ability to maximize the value for patients in our marketed products and product candidates. We strive to protect the proprietary technologies that we believe are important to our business. We aim to achieve this by filing patent positions covering key patient innovations. Those key patient innovations are often embodied in the product labels describing how our products should be deployed to treat disease including the structure of our molecules in our products, and the formulation, dosing, administration, and other key innovations establishing the utility of our products for patients. In addition, we invest deeply in our manufacturing technologies and strive to protect new innovations with the potential to bring options and value to the company. These patent positions may prove useful in preventing an infringing third party from introducing a product into commerce during the term of our granted patent positions. In addition to patent protection, we also rely on trademarks to protect the goodwill and brand recognition the company brings to health care providers and patients. In particular, we aim to protect the hard- earned goodwill we earned with patient groups, our IIP partners, and other key partners that support the co- creation of our business. We also rely on trade secrets to protect valuable aspects of our business that are not amenable to, or that we do not consider appropriate for, patent protection, including certain aspects of our antibody engineering technologies, assay development, product development, biological insights, and multiple aspects of our business operations that have made a positive patient and commercial impact. Our future growth and ability to compete depends on maintaining our culture, retaining our key personnel and recruiting additional qualified personnel. We prioritize building and maintaining a strong, values-driven culture, which is reflected in the way we hire, reward, and promote talent. Cultural fit, integrity, and alignment with our five cultural pillars are key criteria in our talent processes, and we continue to invest in programs that strengthen our culture and support the development of our employees. As part of this broader talent framework, we seek to offer competitive remuneration packages and share-based incentives in the form of the Equity Incentive Plan. We perform periodic benchmark analyses with an external independent service provider to promote the competitiveness of the compensation offered to our key personnel in comparison to other (reference group) companies. RISK FACTOR MEASURES TAKEN TO CONTROL THESE RISKS 3.9.3 General Description of Our Risk Appetite Our risk appetite is defined by risk category and serves as a framework for assessing and managing risks in line with our strategy and priorities. While certain risks and uncertainties are inherent to our business and beyond our control, others may be influenced or mitigated. The Global Risk Management Committee regularly assesses whether risk exposures given the risk appetite remain within the levels approved by the Global Risk Management Committee. Key risks and related mitigating measures are monitored through the Enterprise Risk Management framework and reported to the Audit and Compliance Committee. In accordance with guideline 400.1054 of the Dutch Counsel for Annual Reporting (Raad voor de Jaarverslaggeving), this risk management section provides an overview of the risk mitigating actions taken or planned to be taken by us. The mentioning of these mitigating actions may not in any way be viewed as an implied or express guarantee that such mitigation will in practice be effective in limiting the risk exposure and/or the potential damage to us from any such risk materializing. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Risk Appetite & Control 162

3.9.4 Material Impact of Risk Materialization in 2025 During the period between January 1, 2025 and December 31, 2025, we did not identify any material impact as a result of materialization of previously identified risks and uncertainties. 3.9.5 Financial Risks and Controls In running our business, we seek to implement a sustainable policy regarding internal control and risk management. Our Board of Directors has delegated an active role to our Audit and Compliance Committee in the design, implementation and monitoring of an internal risk management and control system to manage the significant risks to which we are exposed. Our financial reporting is structured within a tight framework of budgeting, reporting and forecasting. A distinction is made between reports for internal and external use. External reporting at group level consists of an annual report (in the form of this Annual Report), including financial statements audited by the independent auditor, as well semi-annual reporting and quarterly updates, containing summarized financial information. The external reports are based on the internal financial reporting. Internal financial reporting consists of extensive consolidated monthly reports in which current developments are compared to the monthly (cumulative) budgets and previous forecasts. In addition, each quarter we reiterate or update our forecast for the annual results, including the cash flow position at the end of the year. The quarterly budgets are part of the annual group budget, which is prepared every year by our Senior Management Team and approved by our Board of Directors. Our specialized finance and administration department are primarily responsible for evaluating the draft internal and external reporting, before these are finally approved by our Board of Directors. Our Board of Directors discusses the financial results of the group at all formal board meetings, which meetings are minuted. Our internal controls over financial reporting are a subset of internal controls and include policies and procedures that: • pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of our assets; • provide reasonable assurance that transactions are recorded as necessary to permit preparation of our financial statements in accordance with IFRS as issued by the International Accounting Standards Board and as adopted by the EU, and that receipts and expenditures are being made only by authorized persons; and • provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use or disposition of our assets that could have a material effect on the financial statements. Since we have securities registered with the SEC and are a large accelerated filer within the meaning of Rule 12b-2 of the Exchange Act, we need to assess the effectiveness of our internal controls over financial reporting and provide a report on the results of our assessment. Our Board of Directors reviewed its internal controls over financial reporting based on criteria established in the Internal Control – Integrated Framework (2013) issued by the Committee of Sponsoring Organizations of the Treadway Commission and engaged an external advisor to help assess the effectiveness of its controls. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Risk Appetite & Control 163

3.9.6 Recent or Current Developments in our System of Risk Management We pay attention to proactive risk management by continuing to have the evaluation of our core risks and uncertainties as a standing discussion topic for our Board of Directors. In addition, since 20242025, we have added quarterly updates for specific risks to our Board of Directors agendas, including cyber security, privacy and healthcare compliance risks. 3.9.7 Framework and Effectiveness of Internal Risk Management and Control Systems According to best practice provision 1.4.2(ii) of the DCGC, the Board of Directors must report in the board report on the design and operating effectiveness of the internal risk management and control systems during the past financial year. The revised DCGC of March 2025 added that the frameworks that have been used for this purpose (such as the COSO framework for internal control) must be reported on. It is expected that the management board indicates, in the reporting on the design and operating effectiveness of the internal risk management and control systems, which reference or standards framework (e.g. the COSO framework for internal control) has been used. It is also expected that the Board of Directors clearly explains how it has assessed the operating effectiveness of the internal risk management and control systems. Additionally, the revised DCGC of March 2025 added that, pursuant to best practice provision 1.4.2(iii), the Board of Directors must report in their board report on its assessment of the effectiveness of the internal risk management and control systems in relation to operational, compliance and reporting risks for the past financial year. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Risk Appetite & Control 164

General Description of the Company and its Share Capital 4.1 Legal Information on the Company 166 4.2 Share Capital 166 4.3 Share Classes and Principal Shareholders 170 4.4 Limitations on the right to hold securities 172 4.5 General Meeting, Voting Rights and Admission 173 4.6 Anti-Takeover Provisions 175 4.7 Change of Control 175 4.8 Exchange Controls 175 4.9 Amendments of Articles of Association 175 4.10 Transparency Directive 175 4.11 Dutch Financial Reporting Supervision Act 176 4.12 Dividends and Other Distributions 176 4.13 Right to a surplus in the event of a liquidation 177 4.14 Material Modifications to the Rights of Security Holders and Use of Proceeds 177 4.15 Enforcement of civil liabilities 178 4.16 Controls and Procedures 179 4.17 Financial Calendar 2026 180 argenx Annual Report 2025 165 4

4 General Description of the Company and its Share Capital 4.1 Legal Information on the Company 4.1.1 General We were incorporated on April 25, 2008 in the Netherlands and under Dutch law. Our commercial name is ‘argenx’ and since April 26, 2017, our corporate name is ‘argenx SE’. We are a European public company (Societas Europaea or SE), with our corporate seat in Amsterdam, the Netherlands, and are registered with the trade register of the Dutch Chamber of Commerce under number 24435214. Our registered office is at Laarderhoogtweg 25, 1101 EB Amsterdam, the Netherlands and our telephone number is +31 (0) 10 70 38 441. Our website address is http://www.argenx.com. Our LEI is 7245009C5FZE6G9ODQ71. Our ordinary shares are listed on Euronext Brussels under ISIN NL0010832176 under the symbol “ARGX” since July 10, 2014. The ADSs are listed on Nasdaq, under the symbol “ARGX” since May 18, 2017. 4.1.2 Statutory/Corporate Objectives Pursuant to Article 3 of our Articles of Association, our corporate objectives are, amongst others: (a) to exploit, including all activities relating to research, development, production, marketing and commercial exploitation; biological, chemical or other products, processes and technologies in the life sciences sector in general, and more specifically in the diagnostic, pharmaceutical, medical, cosmetic, chemical and agricultural sector; (b) to design and develop instruments which may be used in medical diagnosis and affiliated areas; (c) the worldwide distribution of, sale of and rendering services relating to our products and subsidiaries directly to customers as well as through third parties; and (d) to act as the holding company of the Group. 4.2 Share Capital 4.2.1 Authorized and Issued Share Capital Under Dutch Law, a company’s authorized share capital sets out the maximum amount and number of shares that it may issue without amending its articles of association. Our Articles of Association provide for an authorized share capital in the amount of €9.0 million divided into 90 million shares, each with a nominal value of €0.10. All issued and outstanding shares have been fully paid up and the shares are held in dematerialized form. As of December 31, 2025 our issued and paid up share capital amounted to €6,188,331 ($7,353,827), represented by 61,883,306 ordinary shares with a nominal value of €0.10, each representing an identical fraction of our share capital. As of December 31, 2025, neither we nor any of our subsidiaries held any of our own shares. During the year ended December 31, 2025 and as of the date of this Annual Report, we did not purchase any shares in the Company. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Legal Information on the Company 166

4.2.2 Stock Options, Restricted Stock Units and Performance Stock Units In addition to the shares already outstanding, we have granted stock options which upon exercise will lead to an increase in the number of our outstanding shares. 21,469 stock options were granted on March 31, 2025, 593,475 on June 30, 2025, 30,762on September 30, 2025 and17,595 on December 31, 2025. A total of 3,883,114 stock options (where each stock option entitles the holder to subscribe for one ordinary share) were outstanding and granted as of December 31, 2025. Upon exercise of these 3,883,114 stock options, we will receive a total amount of €1.2 billion ($1.2 billion) in stock option exercise price, thereby increasing our share capital and share premium by the same amount. Further, we have granted RSUs which upon vesting will lead to an increase in the number of our outstanding shares. RSUs were granted on March 31, 2025, on June 30, 2025, on September 30, 2025 and on December 31, 2025. A total of 584,653 RSUs (where the holder receives an equal number of ordinary shares, minus a certain number of shares required to cover certain costs, if applicable) were outstanding and granted as of December 31, 2025. Lastly, we have granted PSUs, which at grant are a conditional right to receive ordinary shares upon vest, based on achievement of performance measures. Vested PSUs will lead to an increase in the number of our outstanding shares. PSUs were granted on 30 June 2025. A total of 30,360 PSUs (where the holder receives, an equal number ordinary share depending on the achievement of performance measures, minus a certain number of shares required to cover certain costs, if applicable) were outstanding and granted as of December 31, 2025. Apart from the stock options, PSUs and RSUs granted under our Equity Incentive Plan, we do not currently have other stock options, RSUs, PSUs options to purchase securities, convertible securities or other rights to subscribe for or purchase securities outstanding. For stock option information through December 31, 2025, see ”Note 13 Share-Based Payments” in our consolidated financial statements which are included to our Annual Report for the year ended December 31, 2025. 4.2.3 American Depositary Shares In connection with our initial public offering on Nasdaq, the Bank of New York Mellon, as depositary, registered and delivered ADSs. Each ADS represents one share (or a right to receive one share) deposited with ING Bank N.V., as custodian for the depositary in the Netherlands. Each ADS also represents any other securities, cash or other property which may be held by the depositary. The deposited shares together with our other securities, cash and other property held by the depositary, are referred to as the deposited securities. The depositary’s office at which the ADSs are administered is located at 101 Barclay Street, New York, New York 10286. The Bank of New York Mellon’s principal executive office is located at 225 Liberty Street, New York, New York 10286. A deposit agreement among us, the depositary, ADS holders and all other persons indirectly or beneficially holding ADSs sets out ADS holder rights as well as the rights and obligations of the depositary. New York law governs the deposit agreement and the ADSs. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Share Capital 167

4.2.4 Fees and Charges Persons depositing or withdrawing shares or ADS holders must pay: For: $5.00 (or less) per 100 ADSs (or portion of 100 ADSs) Issuance of ADSs, including issuances resulting from a distribution of shares or rights or other property Cancellation of ADSs for the purpose of withdrawal, including if the deposit agreement terminates $.05 (or less) per ADS Any cash distribution to ADS holders A fee equivalent to the fee that would be payable if securities distributed to you had been shares and the shares had been deposited for issuance of ADSs Distribution of securities distributed to holders of deposited securities (including rights) that are distributed by the depositary to ADS holders $.05 (or less) per ADS per calendar year Depositary services Registration or transfer fees Transfer and registration of shares on our share register to or from the name of the depositary or its agent when you deposit or withdraw shares Expenses of the depositary Cable, telex and facsimile transmissions (when expressly provided in the deposit agreement) Converting foreign currency to USDs Taxes and other governmental charges the depositary or the custodian has to pay on any ADSs or shares underlying ADSs, such as stock transfer taxes, stamp duty or withholding taxes As necessary Any charges incurred by the depositary or its agents for servicing the deposited securities As necessary The depositary collects its fees for delivery and surrender of ADSs directly from investors depositing shares or surrendering ADSs for the purpose of withdrawal or from intermediaries acting for them. The depositary collects fees for making distributions to investors by deducting those fees from the amounts distributed or by selling a portion of distributable property to pay the fees. The depositary may collect its annual fee for depositary services by deduction from cash distributions or by directly billing investors or by charging the book-entry system accounts of participants acting for them. The depositary may collect any of its fees by deduction from any cash distribution payable (or by selling a portion of securities or other property distributable) to ADS holders that are obligated to pay those fees. The depositary may generally refuse to provide fee-attracting services until its fees for those services are paid. From time to time, the depositary may make payments to us to reimburse us for costs and expenses generally arising out of establishment and maintenance of the ADS program, waive fees and expenses for services provided to us by the depositary or share revenue from the fees collected from ADS holders. In performing its duties under the deposit agreement, the depositary may use brokers, dealers, foreign currency dealers or other service providers that are owned by or affiliated with the depositary and that may earn or share fees, spreads or commissions. The depositary may convert currency itself or through any of its affiliates and, in those cases, acts as principal for its own account and not as agent, advisor, broker or fiduciary on behalf of any other person and earns revenue, including, without limitation, transaction spreads, that it will retain for its own account. The revenue is based on, among other things, the difference between the exchange rate assigned to the currency conversion made under the deposit agreement and the rate that the depositary or its affiliate receives when buying or selling foreign currency for its own account. The depositary makes no representation that the exchange rate used or obtained in any currency conversion under the deposit agreement will be the most favorable rate that could be obtained at the time or that the method by which that rate will be determined will be the most favorable to ADS holders, subject to the depositary’s obligations under the deposit agreement. The methodology used to determine exchange rates used in currency conversions is available upon request. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Share Capital 168
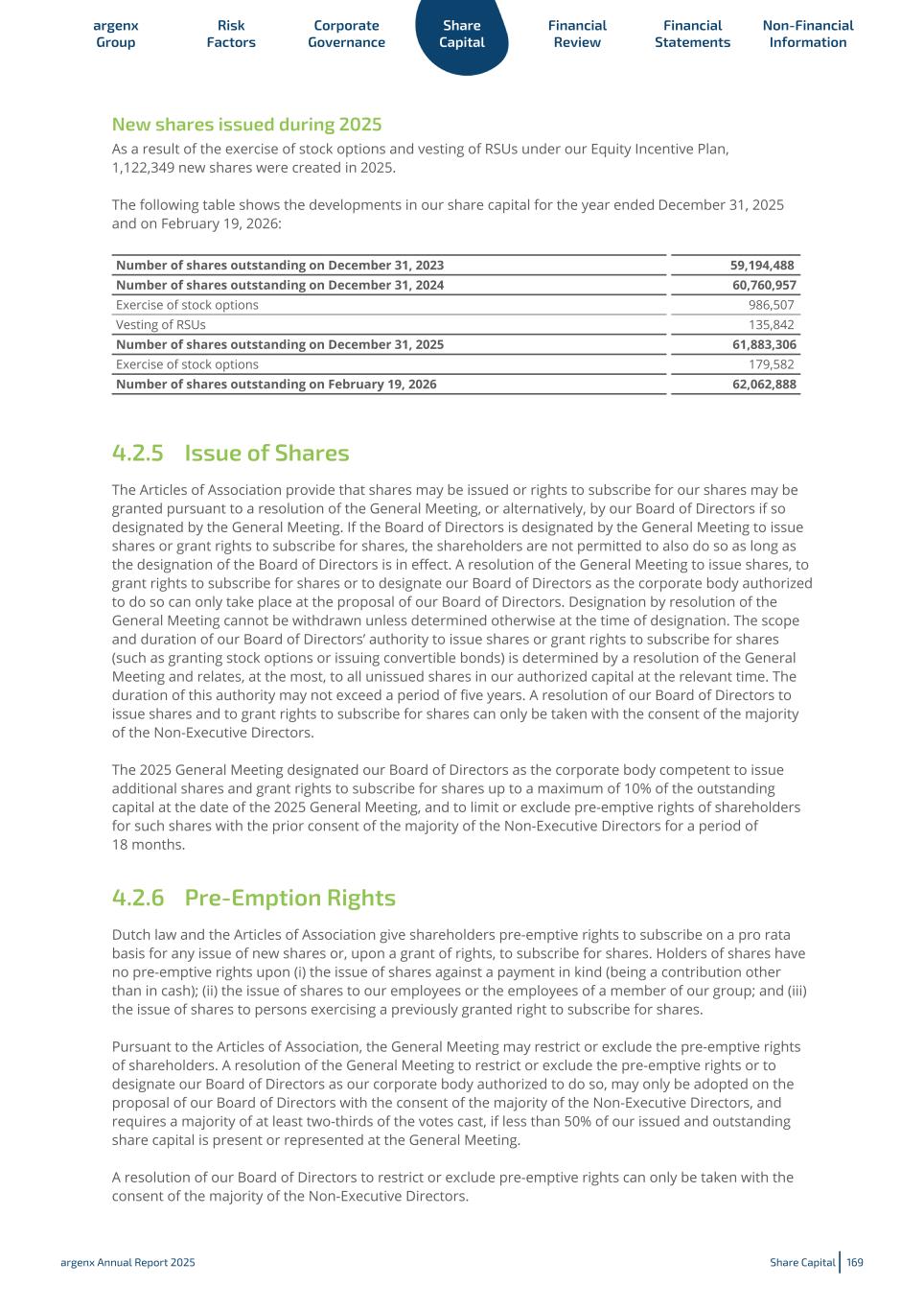
New shares issued during 2025 As a result of the exercise of stock options and vesting of RSUs under our Equity Incentive Plan, 1,122,349 new shares were created in 2025. The following table shows the developments in our share capital for the year ended December 31, 2025 and on February 19, 2026: Number of shares outstanding on December 31, 2023 59,194,488 Number of shares outstanding on December 31, 2024 60,760,957 Exercise of stock options 986,507 Vesting of RSUs 135,842 Number of shares outstanding on December 31, 2025 61,883,306 Exercise of stock options 179,582 Number of shares outstanding on February 19, 2026 62,062,888 4.2.5 Issue of Shares The Articles of Association provide that shares may be issued or rights to subscribe for our shares may be granted pursuant to a resolution of the General Meeting, or alternatively, by our Board of Directors if so designated by the General Meeting. If the Board of Directors is designated by the General Meeting to issue shares or grant rights to subscribe for shares, the shareholders are not permitted to also do so as long as the designation of the Board of Directors is in effect. A resolution of the General Meeting to issue shares, to grant rights to subscribe for shares or to designate our Board of Directors as the corporate body authorized to do so can only take place at the proposal of our Board of Directors. Designation by resolution of the General Meeting cannot be withdrawn unless determined otherwise at the time of designation. The scope and duration of our Board of Directors’ authority to issue shares or grant rights to subscribe for shares (such as granting stock options or issuing convertible bonds) is determined by a resolution of the General Meeting and relates, at the most, to all unissued shares in our authorized capital at the relevant time. The duration of this authority may not exceed a period of five years. A resolution of our Board of Directors to issue shares and to grant rights to subscribe for shares can only be taken with the consent of the majority of the Non-Executive Directors. The 2025 General Meeting designated our Board of Directors as the corporate body competent to issue additional shares and grant rights to subscribe for shares up to a maximum of 10% of the outstanding capital at the date of the 2025 General Meeting, and to limit or exclude pre-emptive rights of shareholders for such shares with the prior consent of the majority of the Non-Executive Directors for a period of 18 months. 4.2.6 Pre-Emption Rights Dutch law and the Articles of Association give shareholders pre-emptive rights to subscribe on a pro rata basis for any issue of new shares or, upon a grant of rights, to subscribe for shares. Holders of shares have no pre-emptive rights upon (i) the issue of shares against a payment in kind (being a contribution other than in cash); (ii) the issue of shares to our employees or the employees of a member of our group; and (iii) the issue of shares to persons exercising a previously granted right to subscribe for shares. Pursuant to the Articles of Association, the General Meeting may restrict or exclude the pre-emptive rights of shareholders. A resolution of the General Meeting to restrict or exclude the pre-emptive rights or to designate our Board of Directors as our corporate body authorized to do so, may only be adopted on the proposal of our Board of Directors with the consent of the majority of the Non-Executive Directors, and requires a majority of at least two-thirds of the votes cast, if less than 50% of our issued and outstanding share capital is present or represented at the General Meeting. A resolution of our Board of Directors to restrict or exclude pre-emptive rights can only be taken with the consent of the majority of the Non-Executive Directors. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Share Capital 169

The designation of our Board of Directors as the body competent to restrict or exclude the pre-emptive rights may not exceed a period of five years. Designation by resolution of the shareholders at a General Meeting cannot be withdrawn unless determined otherwise at the time of designation. Please refer to section “Issue of Shares” with respect to the current right of the Board of Directors to limit or exclude pre-emptive rights. 4.2.7 Acquisition of Shares in our Capital We may not subscribe for our own shares on issue. We may acquire fully paid-up shares at any time for no consideration or, if: • our shareholders’ equity less the payment required to make the acquisition, does not fall below the sum of called-up and paid-in share capital and any statutory reserves; • we and our subsidiaries would thereafter not hold shares or hold a pledge over shares with an aggregate nominal value exceeding 50% of our issued share capital; and • our Board of Directors has been authorized thereto by the General Meeting. As part of the authorization, the General Meeting must specify the number of shares that may be repurchased, the manner in which the shares may be acquired and the price range within which the shares may be acquired. An authorization by the General Meeting to our Board of Directors for the repurchase of shares can be granted for a maximum period of 18 months. No authorization of the General Meeting is required if ordinary shares are acquired by us with the intention of transferring such ordinary shares to our employees under the Equity Incentive Plan. A resolution of our Board of Directors to repurchase shares can only be taken with the consent of the majority of the Non-Executive Directors. Shares held by us in our own share capital do not carry a right to any distribution. Please refer to Section 4.5 “General Meeting, Voting Rights and Admission" with respect to the exercising voting rights for the shares held by us. 4.2.8 Reduction of Share Capital The shareholders at a General Meeting may, upon a proposal by our Board of Directors, resolve to reduce the issued share capital by cancelling shares or by amending the Articles of Association to reduce the nominal value of the shares. 4.3 Share Classes and Principal Shareholders As at February 19, 2026 our issued share capital amounted to €6,206,288.80 and was represented by 62,062,888 ordinary shares. There is only one class of shares (ordinary shares, including ordinary shares represented by ADSs), and there are no special rights attached to any of the ordinary shares, nor special shareholder rights, including voting rights, for any of our shareholders. Each shareholder has one vote. 4.3.1 Disclosure of holdings Pursuant to the DFSA, any person who, directly or indirectly, acquires or disposes of an (actual or deemed) interest in the capital, voting rights or gross short position of the Company must immediately give written notice to the AFM by means of a standard form, if, as a result of such acquisition or disposal, the percentage of capital interest or voting rights held by such person meets, exceeds or falls below the following thresholds: 3%, 5%, 10%, 15%, 20%, 25%, 30%, 40%, 50%, 60%, 75% and 95%. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Share Capital 170

Any person whose interest in the capital, voting rights or gross short position in the Company meets, exceeds or falls below one or several of the above-mentioned thresholds due to a change in the Company’s outstanding capital, or in voting rights attached to the shares as notified to the AFM by the Company, should notify the AFM no later than the fourth trading day after the AFM has published the notification by the Company. Furthermore, each director must notify the AFM of each change in the number of shares he or she holds and of each change in the number of votes he or she is entitled to cast in respect of our issued and outstanding share capital, immediately after the relevant change. The AFM does not issue separate public announcements of the notifications. It does, however, keep a public register of and publishes all notifications made pursuant to the DFSA at its website (www.afm.nl). Third parties can request to be notified automatically by email of changes to the public register in relation to a particular company’s shares or a particular notifying party. Non-compliance with these notification obligations is an economic offense and may lead to criminal prosecution. The AFM may impose administrative penalties for non-compliance, and the publication thereof. In addition, a civil court can impose measures against any person who fails to notify or incorrectly notifies the AFM of matters required to be notified. A claim requiring that such measures be imposed may be instituted by us, or by one or more of our shareholders who alone or together with others represent at least 3% of our issued and outstanding share capital of or voting rights. Shareholders are advised to consult with their own legal advisors to determine whether the notification obligations apply to them. 4.3.2 Short positions Pursuant to EU Regulation No. 236/2012, each person (legal entities as well as natural persons) holding a net short position attaining 0.1% of our issued share capital must report it to the AFM. Each subsequent increase of this position by 0.1% above 0.1% will also have to be reported. Each net short position equal to 0.5% of our issued share capital and any subsequent increase of that position by 0.1% will be made public via the AFM short selling register. To calculate whether a natural person or legal person has a net short position, their short positions and long positions must be set off. A short transaction in a share can only be contracted if a reasonable case can be made that the shares sold can actually be delivered, which requires confirmation of a third party that the shares have been located. The notification shall be made no later than 15:30 central European time on the following trading day. Furthermore, each person holding a gross short position in relation to our issued share capital that reaches, exceeds or falls below one of the following thresholds: 3%, 5%, 10%, 15%, 20%, 25%, 30%, 40%, 50%, 60%, 75% and 95%, must immediately give written notice to the AFM. If a person’s gross short position reaches, exceeds or falls below one of the above mentioned thresholds as a result of a change in our issued share capital, such person is required to make a notification not later than on the fourth trading day after the AFM has published our notification in the public register of the AFM. The AFM keeps a public register of the short selling notifications. Shareholders are advised to consult with their own legal advisors to determine whether any of the above short selling notification obligations apply to them. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Share Classes and Principal Shareholders 171
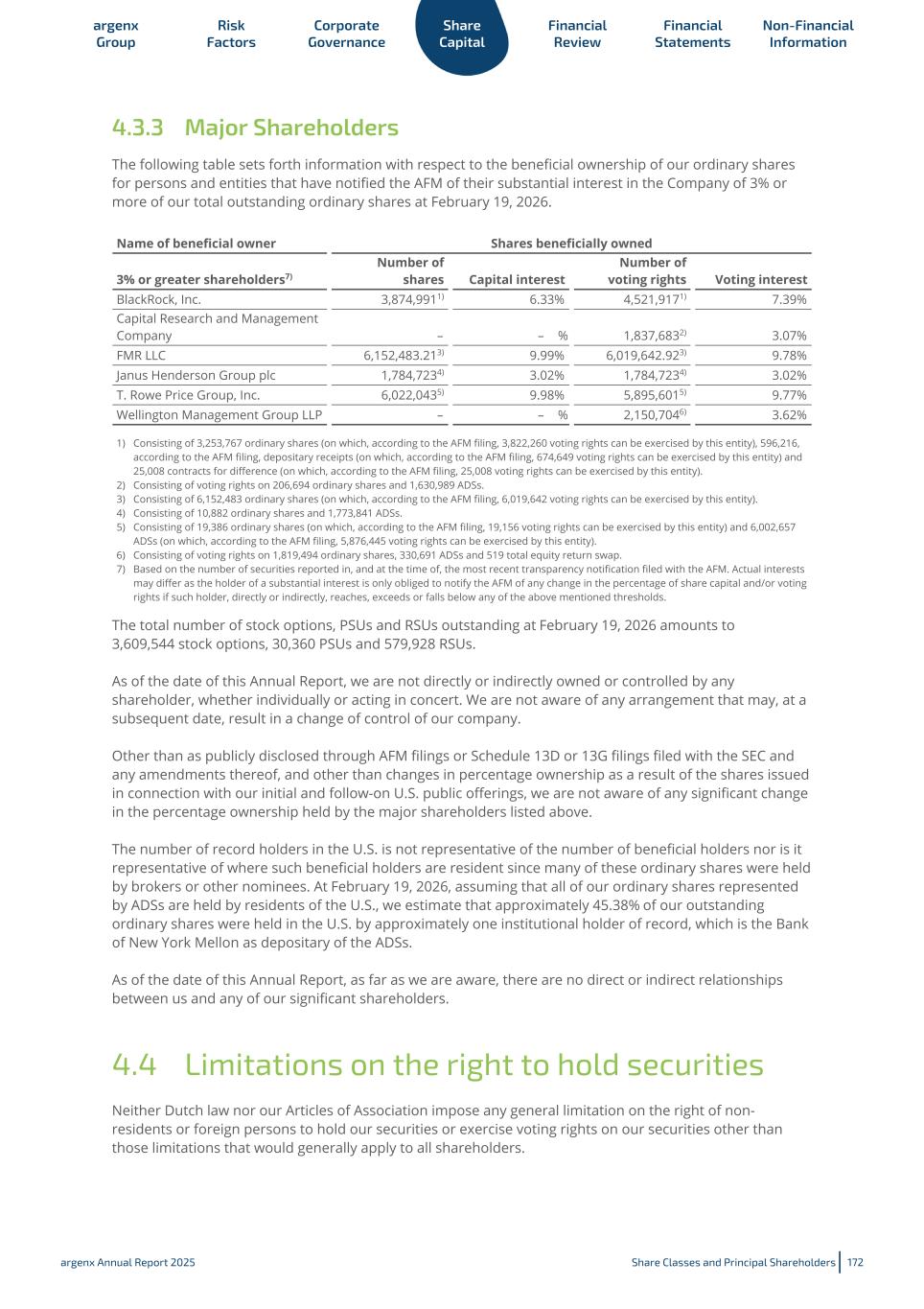
4.3.3 Major Shareholders The following table sets forth information with respect to the beneficial ownership of our ordinary shares for persons and entities that have notified the AFM of their substantial interest in the Company of 3% or more of our total outstanding ordinary shares at February 19, 2026. Name of beneficial owner Shares beneficially owned 3% or greater shareholders7) Number of shares Capital interest Number of voting rights Voting interest BlackRock, Inc. 3,874,9911) 6.33% 4,521,9171) 7.39% Capital Research and Management Company – – % 1,837,6832) 3.07% FMR LLC 6,152,483.213) 9.99% 6,019,642.923) 9.78% Janus Henderson Group plc 1,784,7234) 3.02% 1,784,7234) 3.02% T. Rowe Price Group, Inc. 6,022,0435) 9.98% 5,895,6015) 9.77% Wellington Management Group LLP – – % 2,150,7046) 3.62% 1) Consisting of 3,253,767 ordinary shares (on which, according to the AFM filing, 3,822,260 voting rights can be exercised by this entity), 596,216, according to the AFM filing, depositary receipts (on which, according to the AFM filing, 674,649 voting rights can be exercised by this entity) and 25,008 contracts for difference (on which, according to the AFM filing, 25,008 voting rights can be exercised by this entity). 2) Consisting of voting rights on 206,694 ordinary shares and 1,630,989 ADSs. 3) Consisting of 6,152,483 ordinary shares (on which, according to the AFM filing, 6,019,642 voting rights can be exercised by this entity). 4) Consisting of 10,882 ordinary shares and 1,773,841 ADSs. 5) Consisting of 19,386 ordinary shares (on which, according to the AFM filing, 19,156 voting rights can be exercised by this entity) and 6,002,657 ADSs (on which, according to the AFM filing, 5,876,445 voting rights can be exercised by this entity). 6) Consisting of voting rights on 1,819,494 ordinary shares, 330,691 ADSs and 519 total equity return swap. 7) Based on the number of securities reported in, and at the time of, the most recent transparency notification filed with the AFM. Actual interests may differ as the holder of a substantial interest is only obliged to notify the AFM of any change in the percentage of share capital and/or voting rights if such holder, directly or indirectly, reaches, exceeds or falls below any of the above mentioned thresholds. The total number of stock options, PSUs and RSUs outstanding at February 19, 2026 amounts to 3,609,544 stock options, 30,360 PSUs and 579,928 RSUs. As of the date of this Annual Report, we are not directly or indirectly owned or controlled by any shareholder, whether individually or acting in concert. We are not aware of any arrangement that may, at a subsequent date, result in a change of control of our company. Other than as publicly disclosed through AFM filings or Schedule 13D or 13G filings filed with the SEC and any amendments thereof, and other than changes in percentage ownership as a result of the shares issued in connection with our initial and follow-on U.S. public offerings, we are not aware of any significant change in the percentage ownership held by the major shareholders listed above. The number of record holders in the U.S. is not representative of the number of beneficial holders nor is it representative of where such beneficial holders are resident since many of these ordinary shares were held by brokers or other nominees. At February 19, 2026, assuming that all of our ordinary shares represented by ADSs are held by residents of the U.S., we estimate that approximately 45.38% of our outstanding ordinary shares were held in the U.S. by approximately one institutional holder of record, which is the Bank of New York Mellon as depositary of the ADSs. As of the date of this Annual Report, as far as we are aware, there are no direct or indirect relationships between us and any of our significant shareholders. 4.4 Limitations on the right to hold securities Neither Dutch law nor our Articles of Association impose any general limitation on the right of non- residents or foreign persons to hold our securities or exercise voting rights on our securities other than those limitations that would generally apply to all shareholders. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Share Classes and Principal Shareholders 172

4.5 General Meeting, Voting Rights and Admission General Meetings are held at the place where the Company has its official seat (being Amsterdam) or at Schiphol Airport (municipality of Haarlemmermeer), the Netherlands. The Articles of Association provide that at least one annual General Meeting shall be held within six months after the close of each fiscal year. Additional extraordinary General Meetings may be held whenever our Board of Directors deems such to be necessary. Shareholders representing alone or in aggregate at least one-tenth of our issued and outstanding share capital may, pursuant to the DCC, request that a General Meeting be convened. If our Board of Directors has not taken the steps necessary to ensure that a General Meeting will be held within the relevant statutory period after the request, the requesting persons may, at his/her/their request, be authorized by a court in preliminary relief proceedings to convene a General Meeting. We will give notice of any General Meeting by publication on our website and furthermore, to the extent required, in another manner in accordance with the applicable stock exchange regulations. The notice convening any General Meeting must include, among other items, an agenda indicating the place and date of the meeting, the items for discussion and voting, the proceedings for registration including the registration date, as well as any proposals for the agenda made by the Board of Directors or shareholders holding at least 3% of the issued share capital. For an annual General Meeting, the agenda shall include, among other things, the adoption of the annual accounts, appropriation of our profits and proposals relating to the composition of our Board of Directors. Pursuant to Dutch law, shareholders holding at least 3% of our issued and outstanding share capital have a right to request our Board of Directors to include items on the agenda of any General Meeting. Our Board of Directors must agree to these requests, provided that (i) the request was made in writing and motivated, and (ii) the request was received by the chairperson of our Board of Directors at least 60 days prior to the date of a General Meeting. No resolutions shall be adopted on items other than those which have been included in the agenda. In accordance with the DCGC, a shareholder may include an item on the agenda only after consulting our Board of Directors in that respect. If one or more shareholders intends to request that an item be put on the agenda that may result in a change in the Company’s strategy, our Board of Directors may invoke a response time of a maximum of 180 days until the day of a General Meeting. In addition, pursuant to the DCC, our Board of Directors may invoke a statutory cooling-off period up to a maximum of 250 days (wettelijke bedenktijd). For the Company, this will apply in case: • shareholders request our Board of Directors to have a General Meeting consider a proposal for the appointment, suspension or dismissal of one or more directors, or a proposal for the amendment of one or more provisions in the Articles of Association relating thereto; or • a public offering of shares in the capital of the Company is announced or made without the bidder and the Company having been reached agreement about the offering; and • only if our Board of Directors also considers the relevant situation to be substantially contrary to the interests of the Company and its affiliated enterprises. If our Board of Directors invokes such a cooling-off period, this causes the powers of the General Meeting to appoint, suspend or dismiss directors (and to amend the Articles of Association in this respect) to be suspended. General Meetings are presided over by the chairperson of the Board of Directors or, if he/she is absent, by the vice chairperson of the Board of Directors. If both the chairperson and the vice chairperson are absent, the Non-Executive Directors present at the General Meeting shall appoint one of them to be chairperson. In General Meetings, members of the Board of Directors have an advisory vote. The chairperson of the General Meeting may decide at his/her discretion to admit other persons to the General Meeting. The external auditor of the Company shall attend a General Meeting in which the annual accounts are discussed. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 General Meeting, Voting Rights and Admission 173

Our Board of Directors must give notice of a General Meeting, by at least such number of days prior to the day of the meeting as required by Dutch law, which is currently 42 days. Shareholders (as well as other persons with voting rights or meeting rights) may attend a General Meeting, to address the General Meeting and, in so far as they have such right, to exercise voting rights pro rata to its shareholding, either in person or by proxy. Shareholders may exercise these rights, if they are the holders of shares on the registration date which is currently the 28th day before the day of a General Meeting, and they or their proxy have notified our Board of Directors of their intention to attend a General Meeting in writing at the address and by the date specified in the notice of said meeting. All shareholders, and each usufructuary and pledgee to whom the right to vote on our shares accrues, are entitled, in person or represented by a proxy authorized in writing, to attend and address a General Meeting and exercise voting rights pro rata to their shareholding. Shareholders may exercise their rights if they are the holders of our shares on the record date as required by Dutch law, which is currently the 28th day before the day of a General Meeting, and they or their proxy have notified us of their intention to attend such General Meeting in writing or by any other electronic means that can be reproduced on paper ultimately at a date set for that purpose by our Board of Directors which date may not be earlier than the seventh day prior to such General Meeting, specifying such person’s name and the number of shares for which such person may exercise the voting rights and/or meeting rights at such General Meeting. The convocation notice shall state the record date and the manner in which the persons entitled to attend a General Meeting may register and exercise their rights. Each ordinary share confers the right on the holder to cast one vote at the General Meeting. Shareholders may vote by proxy. The voting rights attached to any shares held by us are suspended as long as they are held in treasury. Nonetheless, the holders of a right of usufruct (vruchtgebruik) in shares belonging to another and the holders of a right of pledge in respect of ordinary shares held by us are not excluded from any right they may have to vote on such ordinary shares, if the right of usufruct (vruchtgebruik) or the right of pledge was granted prior to the time such ordinary share was acquired by us. We may not cast votes in respect of a share in respect of which there is a right of usufruct (vruchtgebruik) or a right of pledge. Shares which are not entitled to voting rights pursuant to the preceding sentences will not be taken into account for the purpose of determining the number of shareholders that vote and that are present or represented, or the amount of the share capital that is provided or that is represented at a General Meeting. Decisions of the General Meeting are taken by an absolute majority of votes cast, except where Dutch law or the Articles of Association provide for a qualified majority or unanimity. In accordance with Dutch law and generally accepted business practices, our Articles of Association do not provide quorum requirements generally applicable to a General Meeting. To this extent, our practice varies from the requirement of Nasdaq Listing Rule 5620(c), which requires an issuer to provide in its bylaws for a generally applicable quorum, and that such quorum may not be less than one-third of the outstanding voting stock. Two General Meetings were held in 2025. At the 2025 General Meeting, our annual report and annual accounts for the year ended December 31, 2024 were approved, the allocation of profits of the year ended December 31, 2024 to the retained earnings of the Company was approved. Anthony Rosenberg was reappointed as a Non-Executive Director to the Board of Directors for a term of two years, and the Board of Directors was authorized to issue shares and grant rights to subscribe for shares in our share capital for up to 10% of the outstanding share capital at the date of the meeting and for a period of 18 months from the meeting and to limit or exclude statutory pre-emptive rights with regard to such (rights to subscribe for) shares. At the 2025 Extraordinary General Meeting, the 2025 Remuneration Policy was approved. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 General Meeting, Voting Rights and Admission 174

4.6 Anti-Takeover Provisions Various protective measures are possible and permissible within the boundaries set by Dutch law and Dutch case law. We have not implemented specific measures with the aim of deterring takeover attempts. However, we have adopted several provisions that may have the effect of making a takeover of argenx more difficult or less attractive, including requirements that certain matters, including an amendment of our Articles of Association, may only be brought to our shareholders for a vote upon a proposal by our Board of Directors. No takeover bid has been instigated by third parties in respect of our equity during the current or previous fiscal years. 4.7 Change of Control The Company is not a party to any significant agreements which will take effect, be altered or terminated upon a change of control of the Company as a result of a public offer. 4.8 Exchange Controls Under Dutch law, subject to the 1977 Sanction Act (Sanctiewet 1977) and other international economic or financial sanctions, there are no exchange control restrictions on investments in, or payments on, shares (except as to cash amounts). There are no special restrictions in our Articles of Association or Dutch law that limit the right of shareholders who are not citizens or residents of the Netherlands to hold or vote shares. 4.9 Amendments of Articles of Association The shareholders at a General Meeting may amend the Articles of Association, at the proposal of our Board of Directors, with the consent of the majority of the Non-Executive Directors. A resolution by the shareholders at a General Meeting to amend the Articles of Association requires a simple majority of the votes cast in a meeting in which at least half of our issued and outstanding capital is present or represented, or at least two-thirds of the votes cast, if less than half of our issued and outstanding capital is present or represented at that meeting. The Articles of Association were last amended in 2024 pursuant to the notarial deed of partial amendment of the Articles of Association, executed on May 7, 2024. The full text of the Articles of Association and an unofficial English translation thereof are available on our website (www.argenx.com/investors). 4.10 Transparency Directive We are a European public company with limited liability (Societas Europaea or SE) incorporated and existing under the laws of the Netherlands. The Netherlands is our EU home member state (lidstaat van herkomst) for the purposes of Directive 2004/109/EC (as amended by Directive 2013/50/EU), or the Transparency Directive, as a consequence of which we are subject to the DFSA in respect to certain ongoing transparency and disclosure obligations. In addition, as long as our shares are listed on Euronext Brussels and the ADSs on Nasdaq, we are required to disclose any regulated information which has been disclosed pursuant to the DFSA as well as in accordance with the Belgian Law of May 2, 2007, the Belgian Royal Decree of November 14, 2007 as well as Nasdaq Listing Rules. We must publish our annual accounts within four months after the end of each financial year and our half-yearly figures within three months after the end of the first six months of each financial year. Within five calendar days after adoption of our annual accounts, we must file our adopted annual accounts with the AFM. Pursuant to the DFSA, we will be required, among other things, to make public without delay any change in the rights attaching to our shares or any rights to subscribe our shares. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Anti-Takeover Provisions 175

4.11 Dutch Financial Reporting Supervision Act Pursuant to the Dutch Financial Reporting Supervision Act (Wet toezicht financiële verslaggeving), the AFM supervises the application of financial reporting standards and has an independent right to (i) request an explanation from the Company regarding its application of the applicable financial reporting standards if, based on publicly known facts or circumstances, it has reason to doubt that the issuer’s financial reporting meets such standards and (ii) make a notification to the Company that its financial reports do not meet the applicable financial reporting standards, which notification may be accompanied by a recommendation to the Company to issue a press release on the subject matter. If the Company does not comply with such a request or recommendation, the AFM may request the Enterprise Chamber of the Court of Appeal in Amsterdam (Ondernemingskamer van het Gerechtshof te Amsterdam) (the Enterprise Chamber) to order the Company to (a) provide an explanation regarding its application of the applicable financial reporting standards to its financial reports or (b) prepare its financial reports in accordance with the Enterprise Chamber’s instructions. This Annual Report also concerns the annual financial reporting within the meaning of 5:25c(2) DFSA. 4.12 Dividends and Other Distributions Pursuant to Dutch law and the Articles of Association, the distribution of profits will take place following the adoption of our annual accounts, from which we will determine whether such distribution is permitted. We may only make distributions to the shareholders, whether from profits or from its freely distributable reserves, only insofar as its shareholders’ equity exceeds the sum of the paid-up and called-up share capital plus the reserves required to be maintained by Dutch law. The General Meeting may determine which part of our profits will be added to the reserves in consideration of our reserves and dividends policy. The remaining part of the profits after the addition to the reserves will be at the disposal of the General Meeting. Distributions of dividends will be made pro rata to the nominal value of each share. Subject to Dutch law and the Articles of Association, our Board of Directors, with the consent of the majority of the Non-Executive Directors, may resolve to distribute an interim dividend if it determines such interim dividend to be justified by our profits. For this purpose, our Board of Directors must prepare an interim statement of assets and liabilities. Such interim statement shall show our financial position not earlier than on the first day of the third month before the month in which the resolution to make the interim distribution is announced. An interim dividend can only be paid if (a) an interim statement of assets and liabilities is drawn up showing that the funds available for distribution are sufficient, and (b) our shareholders’ equity exceeds the sum of the paid-up and called-up share capital plus the reserves required to be maintained by Dutch law. Our Board of Directors, with the consent of the majority of the Non-Executive Directors, may resolve that we make distributions to shareholders from one or more of our freely distributable reserves, other than by way of profit distribution, subject to the due observance of our policy on reserves and dividends. Any such distributions will be made pro rata to the nominal value of each share. Dividends and other distributions shall be made payable not later than the date determined by our Board of Directors. Claims to dividends and other distributions not made within five years from the date that such dividends or distributions became payable, will lapse and any such amounts will be considered to have been forfeited to us (verjaring). argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Dutch Financial Reporting Supervision Act 176

Our Board of Directors has declared a series of interim distributions on account of the Company’s freely distributable reserves for such amounts as was required to pay up the aggregate nominal value of all such shares that were issued to holders of vested RSUs, all in accordance with our Equity Incentive Plan. In accordance with Dutch law, our Board of Directors prepared and filed an interim simplified balance sheet demonstrating that there were sufficient freely distributable reserves for such interim distributions. Such interim simplified balance sheet was filed with the Dutch trade register. The aggregate amount of these interim distributions amounted to approximately €13,584 ($15,961) in 2025. Other than these interim distributions, we have not paid or declared any cash dividends on our ordinary shares, and we do not anticipate paying any cash dividends in the foreseeable future. All of our outstanding shares have the same dividend rights. We intend to retain all available funds and any future earnings to fund the development and expansion of our business. Even if future operations lead to significant levels of distributable profits, we currently intend that any earnings will be reinvested in our business and that cash dividends will not be paid until we have an established revenue stream to support continuing cash dividends. In addition, payment of any future dividends to shareholders would be subject to shareholder approval at a General Meeting, upon proposal of our Board of Directors, which proposal would be subject to the approval of the majority of the Non- Executive Directors after taking into account various factors including our business prospects, cash requirements, financial performance and new product development. Our Articles of Association, as available on our website, contain the provision on the distribution of profits in article 20 (profits, distributions and losses). 4.13 Right to a surplus in the event of a liquidation Any surplus remaining after settlement of all debts and liquidation costs will be distributed to the shareholders in proportion to the nominal value of their shareholdings. 4.14 Material Modifications to the Rights of Security Holders and Use of Proceeds On July 18, 2023, we entered into an Underwriting Agreement with J.P. Morgan Securities LLC, Morgan Stanley & Co. LLC, Goldman Sachs & Co. LLC, BofA Securities, Inc., Cowen and Company, LLC, as representatives of the several underwriters named therein, relating to a global offering of an aggregate of 2,244,899 ordinary shares of the Company, nominal value €0.10 per share, including ordinary shares represented by ADSs, comprised of (i) 1,580,981 ADSs at a public offering price of $490.00 per ADS in the U.S. and countries outside the EEA and (ii) 663,918 ordinary shares at an offering price of €436.37 per ordinary share in a concurrent private placement in the EEA to certain legal entities all of which are qualified investors within the meaning of Regulation 2017/1129 of the European Parliament and of the Council of June 14, 2017, as amended. The offering was made pursuant to our effective shelf registration statement on Form F-3ASR (File No. 333-258251) filed on July 29, 2021, as supplemented by a preliminary prospectus supplement dated July 17, 2023, filed with the SEC on July 17, 2023, and a final prospectus supplement dated July 18, 2023, filed with the SEC on July 20, 2023. In connection with this offering, we granted the underwriters a 30-day option to purchase up to 336,734 additional ordinary shares (which may be represented by ADSs), which was exercised in full. The net proceeds to us from the sale of the ADSs and ordinary shares in this offering, after deducting the underwriting discounts and commissions and estimated offering expenses payable by the Company, was $1.2 billion (€1.1 billion). The offering closed on July 24, 2023. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Dividends and Other Distributions 177

We have not used any of the net proceeds from the offering to make payments, directly or indirectly, to any director, officer or general partner of ours or to their associates, persons owning 10% or more of any class of our equity securities, or to any of our affiliates. We have invested the net proceeds from the offering in cash and cash equivalents and current financial assets. There has been no material change in our planned use of the net proceeds from the offering as described in our final prospectus supplement filed pursuant to Rule 424(b)(5) under the Securities Act with the SEC on July 20, 2023 (File No.333-258251). The registration statement was effective on July 29, 2021. 4.15 Enforcement of civil liabilities We are a European public company with limited liability (Societas Europaea or SE) incorporated under the laws of the Netherlands. Substantially all of our assets are located outside the U.S. The majority of our directors reside outside the U.S. As a result, it may not be possible for investors to effect service of process within the U.S. upon such persons or to enforce against them or us in U.S. courts, including judgments predicated upon the civil liability provisions of the federal securities laws of the U.S. The U.S. and the Netherlands currently do not have a treaty providing for the reciprocal recognition and enforcement of judgments, other than arbitration awards, in civil and commercial matters. Consequently, a final judgment for payment given by a court in the U.S., whether or not predicated solely upon U.S. securities laws, would not automatically be recognized or enforceable in the Netherlands. In order to obtain a judgment which is enforceable in the Netherlands, the party in whose favor a final and conclusive judgment of the U.S. court has been rendered will be required to file its claim with a court of competent jurisdiction in the Netherlands. Such party may submit to the Dutch court the final judgment rendered by the U.S. court. This court will have a level of discretion in its assessment of the judgment rendered by the relevant U.S. court. On the basis of case law by the Dutch Supreme Court, Dutch courts will in principle have to give conclusive effect to a final and enforceable judgment of such court in respect of the contractual obligations thereunder without re-examination or re-litigation of the substantive matters adjudicated upon, provided that: (i) the U.S. court involved accepted jurisdiction on the basis of internationally recognized grounds to accept jurisdiction, (ii) the proceedings before such court being in compliance with principles of proper procedure (behoorlijke rechtspleging), (iii) such judgment not being contrary to the public policy of the Netherlands and (iv) such judgment not being incompatible with a judgment given between the same parties by a Netherlands court or with a prior judgment given between the same parties by a foreign court in a dispute concerning the same subject matter and based on the same cause of action, provided such prior judgment fulfills the conditions necessary for it to be given binding effect in the Netherlands. Dutch courts may deny the recognition and enforcement of punitive damages or other awards that do not fit to the Dutch legal order. Moreover, a Dutch court may reduce the amount of damages granted by a U.S. court and recognize damages only to the extent that they are necessary to compensate actual losses or damages. Enforcement and recognition of judgments of U.S. courts in the Netherlands are solely governed by the provisions of the Dutch Civil Procedure Code. Original actions or actions for the enforcement of judgments of U.S. courts relating to the civil liability provisions of the federal or state securities laws of the U.S. are not directly enforceable in Belgium. The U.S. and Belgium currently do not have a treaty providing for reciprocal recognition and enforcement of judgments, other than arbitral awards, in civil and commercial matters. Consequently, a final judgment for payment given by a court in the U.S., whether or not predicated solely upon U.S. securities laws, would not automatically be recognized or enforceable in Belgium. In order for a final judgment for the payment of money rendered by U.S. courts based on civil liability to produce any effect on Belgian soil, it is accordingly required that this judgment be recognized and be declared enforceable by a Belgian court pursuant to the relevant provisions of the PIL Code. Recognition or enforcement does not imply a review of the merits of the case and is irrespective of any reciprocity requirement. A U.S. judgment will, however, not be recognized or declared enforceable in Belgium if it infringes upon one or more of the grounds for refusal which are exhaustively listed in article 25 of the PIL Code. In addition to recognition or enforcement, a judgment by a federal or state court in the U.S. against us may also serve as evidence in a similar action in a Belgian court if it meets the conditions required for the authenticity of judgments according to the law of the state where it was rendered. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Material Modifications to the Rights of Security Holders and Use of Proceeds 178

In addition, with regard to enforcements by legal proceedings in Belgium (including the recognition of foreign court decisions in Belgium), a registration tax at the rate of 3% of the amount of the judgment is payable by the debtor, if the sum of money which the debtor is ordered to pay by a Belgian court, or by a foreign court judgment that is either (i) automatically enforceable and registered in Belgium, or (ii) rendered enforceable by a Belgian court, exceeds €12,500. The registration tax is payable by the debtor. The debtor is liable for the payment of the registration tax, in the proportion determined by the decision ordering payment or liquidation or determining priority for creditors made or established against it. The debtor(s) are jointly and severally liable in the event that they are ordered to pay jointly and severally. A stamp duty is payable as of the second certified copy of an enforcement judgment rendered by a Belgian court, with a maximum of €1,450. Dutch and Belgian civil procedure differ substantially from U.S. civil procedure in a number of respects. Insofar as the production of evidence is concerned, U.S. law and the laws of several other jurisdictions based on common law provide for pre-trial discovery, a process by which parties to the proceedings may prior to trial compel the production of documents by adverse or third parties and the deposition of witnesses. Evidence obtained in this manner may be decisive in the outcome of any proceeding. No such pre-trial discovery process exists under Dutch or Belgian law. Subject to the foregoing and service of process in accordance with applicable treaties, investors may be able to enforce in the Netherlands or Belgium judgments in civil and commercial matters obtained from U.S. federal or state courts. However, no assurance can be given that those judgments will be enforceable. In addition, it is doubtful whether a Dutch or Belgian court would accept jurisdiction and impose civil liability in an original action commenced in the Netherlands or Belgium and predicated solely upon U.S. federal securities laws. 4.16 Controls and Procedures 4.16.1 Disclosure Controls and Procedures Our management, with the participation of our CEO and CFO, has evaluated the effectiveness of the design and operation of our disclosure controls and procedures pursuant to Exchange Act Rule 13a-15(b) as of December 31, 2025. While there are inherent limitations to the effectiveness of any system of disclosure controls and procedures, including the possibility of human error and the circumvention or overriding of the controls and procedures, our disclosure controls and procedures are designed to provide reasonable assurance of achieving their objectives. Based upon our evaluation, as of December 31, 2025, our CEO and CFO have concluded that the disclosure controls and procedures, in accordance with Exchange Act Rule 13a-15(e), are (i) effective at the level of reasonable assurance in ensuring that information required to be disclosed in the reports that are filed or submitted under the Exchange Act, is recorded, processed, summarized and reported within the time periods specified in the Commission’s rules and forms, and (ii) are effective at the level of reasonable assurance in ensuring that information to be disclosed in the reports that are filed or submitted under the Exchange Act is accumulated and communicated to the management of our company, including our CEO and CFO, to allow timely decisions regarding required disclosure. 4.16.2 Management’s Annual Report on Internal Control Over Financial Reporting Our management is responsible for establishing and maintaining adequate internal control over financial reporting (as such term is defined in Rule 13a-15(f) and Rule 15d-15(f) under the Exchange Act). Our internal control over financial reporting is a process designed, under the supervision of our CEO and CFO, to provide reasonable assurance regarding the reliability of financial reporting and preparation of financial statements for external reporting purposes in accordance with IFRS, as issued by the IASB. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Enforcement of civil liabilities 179

Our internal control over financial reporting includes policies and procedures that pertain to the maintenance of records that, in reasonable detail, accurately and fairly, reflect transactions and dispositions of assets, provide reasonable assurance that transactions are recorded in the manner necessary to permit the preparation of financial statements in accordance with IFRS, as issued by the IASB, and that receipts and expenditures are only carried out in accordance with the authorization of our management and directors, and provide reasonable assurance regarding the prevention or timely detection of any unauthorized acquisition, use or disposition of our assets that could have a material effect on our consolidated financial statements. Because of its inherent limitations, internal control over financial reporting may not prevent or detect all misstatements. Moreover, projections of any evaluation of the effectiveness of internal control to future periods are subject to a risk that controls may become inadequate because of changes in conditions and that the degree of compliance with the policies or procedures may deteriorate. Our management has assessed the effectiveness of internal control over financial reporting based on the Internal Control-Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (COSO) in 2013. Based on this assessment, our management has concluded that our internal control over financial reporting as of December 31, 2025 was effective. 4.16.3 Changes in Internal Control Over Financial Reporting During the period covered by this Annual Report, we have not made any change to our internal control over financial reporting that has materially affected, or is reasonably likely to materially affect, our internal control over financial reporting. 4.16.4 Internal Control Over Non-Financial Information The Company has implemented internal controls over non-financial information, namely Sustainability Reporting based on the Internal Control-Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (COSO) in 2013. The Company continues to implement improvements to its internal control over non-financial reporting. 4.17 Financial Calendar 2026 May 6, 2026 Annual General Meeting of Shareholders in Amsterdam, the Netherlands May 7, 2026 First Quarter 2026 Financial Results and Business Update July 23, 2026 Half Year and Second Quarter 2026 Financial Results and Business Update October 22, 2026 Third Quarter 2026 Financial Results and Business Update argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Controls and Procedures 180

Operating and Financial Review and Prospects "Operating and Financial Review and Prospects” should be read together with the information in our financial statements and related notes included elsewhere in this Annual Report. The following discussion is based on our financial information prepared in accordance with the International Financial Reporting Standards as issued by the IASB’s and adopted by the European Union (EU-IFRS) and in accordance with the legal requirements of Part 9 of Book 2 of the Dutch Civil Code. The following discussion includes forward-looking statements that involve risks, uncertainties and assumptions. Our actual results may differ materially from those anticipated in these forward-looking statements as a result of many factors, including but not limited to those described in section “Risk Factors” and elsewhere in this Annual Report. See “Forward-Looking Statements” in this Annual Report. 5.1 Overview 182 5.2 Basis of presentation 183 5.3 Critical Accounting Judgments and Major Sources of Estimation Uncertainty 183 5.4 Results of Operations 184 5.5 Liquidity and Capital Resources 186 5.6 Research and development, patents and licenses 188 5.7 Trend information 189 5.8 Off-Balance Sheet Arrangements 189 5.9 Contractual Obligations 189 5.10 Information Regarding the Independent Auditor 189 5.11 Material Contracts and Related Party Transactions 190 5.12 Employees 192 5.13 Insurance 193 5.14 Legal and Arbitration Proceedings 193 5.15 Taxation 193 argenx Annual Report 2025 181 5

5 Operating and Financial Review and Prospects 5.1 Overview 2025 was a transformative year for argenx as we advanced our mission to deliver innovation to patients. Our commercial execution reached new heights with the successful expansion of VYVGART, the first-in-class FcRn blocker, which now offers three administration options, including self-injection with the PFS. The PFS launch began in the U.S. in April of this year, followed by the EU and Japan. This evolution reflects our commitment to innovating the patient experience in our two blockbuster indications and pursuing the broadest label for our medicines. The commercialization of the VYVGART franchise generated global product net sales of $4.2 billion in 2025 as compared to $2.2 billion in 2024. Beyond commercial achievements, we continued to execute on a pipeline with breadth and depth, reinforcing our leadership in immunology. We announced positive topline results from the ADAPT SERON clinical trial of VYVGART in Seronegative gMG, further expanding the potential reach of VYVGART. In December of this year, we filed the Seronegative gMG supplemental BLA in pursuit of the broadest MG label of any biologic. In January 2026, the FDA has accepted for priority review a supplemental BLA for VYVGART for the treatment of adults with Seronegative gMG. The application has been granted an expected PDUFA date of May 10, 2026. Additionally, we showcased innovation through our R&D webinar highlighting adimanebart (ARGX-119), a first-in-class agonistic antibody targeting and MuSK to promote maturation and stabilization of the neuromuscular junction, with advancement inCMS. These programs underscore our strategy of entrepreneurial clinical development and commitment to addressing unmet needs across a spectrum of autoimmune and neuromuscular diseases. Looking ahead, in 2026 our teams will strive to continue delivering VYVGART to as many patients as possible. We also expect results to be delivered on four registrational readouts in 2026 and two more in 2027: • Topline results expected in the first quarter of 2026 for Ocular MG (ADAPT OCULUS) with efgartigimod • Topline results expected in the third quarter of 2026 from ALKIVIA clinical trial evaluating three myositis subsetsIMNM,ASyS and DM with efgartigimod • Topline results expected in the fourth quarter of 2026 for EMPASSION clinical trial (MMN) with empasiprubart • Topline results expected in the fourth quarter of 2026 for primary ITP (ADVANCE-NEXT) with efgartigimod • Topline results from UNITY clinical trial (SjD) expected in second half of 2027 with efgartigimod • Topline results from EMVIGORATE clinical trial for empasiprubart (CIDP) expected in second half of 2027 Our Vision 2030 sets the goal to have five new molecules in Phase 3, ten labeled indications, and 50,000 patients on treatment by 2030. It provides a clear roadmap for scaling impact on patients, physicians, and the innovation ecosystem we’re shaping. Achieving this vision will be driven by our core competencies: building winning molecules, entrepreneurial clinical development, and delivering a differentiated patient experience. We plan to continue to prioritize innovation, expand global access, and leverage partnerships to accelerate growth. On our research and development, we continue towards advancing a deep pipeline of both clinical and preclinical-stage product candidates for the treatment of severe autoimmune diseases. Leveraging our technology suite, our ecosystem of partnerships and clinical expertise, we have advanced several argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Overview 182

candidates into late-stage clinical development, and we currently have multiple programs in the discovery stage. argenx continues to target one Investigational New Drug (IND) application per year in generating a world-class pipeline. We enter the next phase of our ambitious innovation agenda with a solid balance sheet, providing financial strength and flexibility to invest confidently in our pipeline and global expansion. As of December 31, 2025 and December 31, 2024, we had cash and cash equivalents amounting to $3.5 billion and $1.5 billion, respectively; in addition to current financial assets of $0.9 billionand $1.9 billion, respectively. As outlined in ‘‘Section 6.1.1 Consolidated Statements of Financial Position’’, the Company held total assets of $8.7 billion as of the year ended December 31, 2025, compared to $6.2 billion as of the year ended December 31, 2024. The main reason for the material change in balance sheet total is the operational growth of the Company in the period. For the year ended December 31, 2025 the Company recorded a second year of profitability with $1 billion compared to our first annual profit for the year ended December 31, 2024 in the amount of $1 billion. This was the Company’s first year of operational profitability. As of December 31, 2025, we had accumulated losses of $0.3 billion. We expect our expenses to continue to increase as we continue to execute registrational and proof-of- concept studies across efgartigimod, empasiprubart and adimanebart, as well as the continued investment in our IIP. We anticipate that our expenses will increase if and as we execute on our research and development activities, pre-commercial and commercial activities and various other activities. We are actively engaged in the maintenance, expansion and protection of our intellectual property portfolio, including litigation costs associated with defending against alleged patent infringement claims or enforcing our IP rights against third parties. We expect that the costs of development and commercialization might also increase due to current and future collaborations with research and development partners as well as commercial partners. 5.2 Basis of presentation ‘‘Section 5 Operating and Financial Review and Prospects’’ should be read in parallel to our consolidated financial statements and ‘‘Section 2 Risk Factors’’ which are included to our Annual Report for the period ended December 31, 2025. We specifically, but not exhaustively, indicate the following references to the notes of the financial statements ‘‘Note 2 Material Accounting Policy Information’’: • the basis of presentation, • the material accounting policies, and • the description of the composition of material accounts, which is further detailed in the notes to the consolidated financial statements. 5.3 Critical Accounting Judgments and Major Sources of Estimation Uncertainty In the application of the Company’s accounting policies, which are described above, the Company is required to make judgments, estimates and assumptions about the carrying amounts of assets and liabilities that are not readily apparent from other sources. The estimates and associated assumptions are based on historical experience and other factors that are considered to be relevant. Actual results may differ from these estimates. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Overview 183
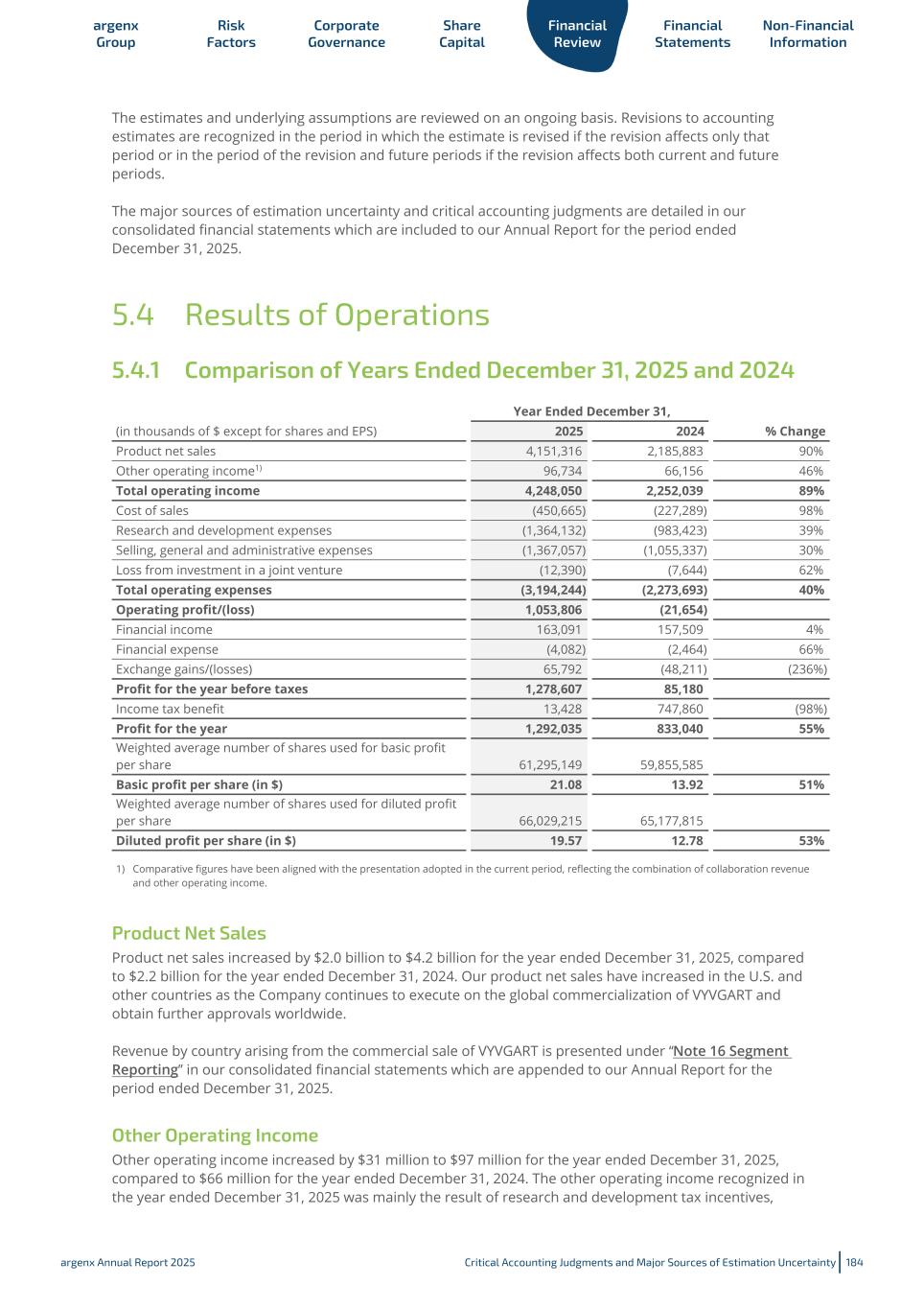
The estimates and underlying assumptions are reviewed on an ongoing basis. Revisions to accounting estimates are recognized in the period in which the estimate is revised if the revision affects only that period or in the period of the revision and future periods if the revision affects both current and future periods. The major sources of estimation uncertainty and critical accounting judgments are detailed in our consolidated financial statements which are included to our Annual Report for the period ended December 31, 2025. 5.4 Results of Operations 5.4.1 Comparison of Years Ended December 31, 2025 and 2024 Year Ended December 31, (in thousands of $ except for shares and EPS) 2025 2024 % Change Product net sales 4,151,316 2,185,883 90% Other operating income1) 96,734 66,156 46% Total operating income 4,248,050 2,252,039 89% Cost of sales (450,665) (227,289) 98% Research and development expenses (1,364,132) (983,423) 39% Selling, general and administrative expenses (1,367,057) (1,055,337) 30% Loss from investment in a joint venture (12,390) (7,644) 62% Total operating expenses (3,194,244) (2,273,693) 40% Operating profit/(loss) 1,053,806 (21,654) Financial income 163,091 157,509 4% Financial expense (4,082) (2,464) 66% Exchange gains/(losses) 65,792 (48,211) (236%) Profit for the year before taxes 1,278,607 85,180 Income tax benefit 13,428 747,860 (98%) Profit for the year 1,292,035 833,040 55% Weighted average number of shares used for basic profit per share 61,295,149 59,855,585 Basic profit per share (in $) 21.08 13.92 51% Weighted average number of shares used for diluted profit per share 66,029,215 65,177,815 Diluted profit per share (in $) 19.57 12.78 53% 1) Comparative figures have been aligned with the presentation adopted in the current period, reflecting the combination of collaboration revenue and other operating income. Product Net Sales Product net sales increased by $2.0 billion to $4.2 billion for the year ended December 31, 2025, compared to $2.2 billion for the year ended December 31, 2024. Our product net sales have increased in the U.S. and other countries as the Company continues to execute on the global commercialization of VYVGART and obtain further approvals worldwide. Revenue by country arising from the commercial sale of VYVGART is presented under ‘‘Note 16 Segment Reporting’’ in our consolidated financial statements which are appended to our Annual Report for the period ended December 31, 2025. Other Operating Income Other operating income increased by $31 million to $97 million for the year ended December 31, 2025, compared to $66 million for the year ended December 31, 2024. The other operating income recognized in the year ended December 31, 2025 was mainly the result of research and development tax incentives, argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Critical Accounting Judgments and Major Sources of Estimation Uncertainty 184

payroll tax rebates and the clinical supply of product on product net sales of VYVGART in Greater China through Zai Lab. Other operating income is presented under ‘‘Note 15 Other Operating Income’’ in our consolidated financial statements which are appended to our Annual Report for the period ended December 31, 2025. Research and Development Expenses Research and development expenses consist principally of: • external research and development expenses related to (i) chemistry, manufacturing and control costs for our product candidates, both for preclinical and clinical testing, all of which is conducted by specialized contract manufacturers, (ii) fees and other costs paid to CROs in connection with preclinical testing and the performance of clinical trials for our product candidates, (iii) costs associated with regulatory submissions and approvals, QA and pharmacovigilance and (iv) costs associated with post- approval clinical trials; • personnel expenses related to compensation of research and development staff and related expenses, including salaries, benefits and share-based payment expenses; and • other expenses. Our research and development expenses totaled $1.4 billion and $1.0 billion for the years ended December 31, 2025 and 2024, respectively. The increase of $0.4 billion in 2025 as compared to 2024 is primarily driven by an increase in personnel expenses and external research and development expenses. Our external research and development expenses for the year ended December 31, 2025 totaled to $0.4 billion, compared to $0.3 billion for the year ended December 31, 2024. The expenses reflect clinical trial costs and manufacturing expenses related to the development of our product candidate portfolio. Personnel expenses relate to internal and external R&D personnel. The expenses also include share-based compensation expenses related to our research and development employees. Our research and development expenses may vary substantially from period to period based on the timing of our research and development activities, including the timing of the initiation of clinical trials, material used in R&D phase and enrollment of patients in clinical trials. Research and development expenses are expected to increase as we advance the clinical development of efgartigimod, empasiprubart, adimanebart and further advance the research and development of our other early-stage pipeline candidates. The successful development of our product candidates is highly uncertain. At this time, we cannot reasonably estimate the nature, timing and estimated costs of the efforts that will be necessary to complete the development of, or the period, if any, in which material net cash inflows may commence from any of our product candidates. This is due to numerous risks and uncertainties associated with developing drugs, as further described in “ Section 2 Risk Factors”. Research and development expenses are presented under ‘‘Note 17 Research and Development Expenses’’ in our consolidated financial statements which are included to our Annual Report for the period ended December 31, 2025. Selling, General and Administrative Expenses Selling, general and administrative expenses consist primarily of: • personnel expenses related to commercial and enabling functions, as well as their related expenses, including salaries, benefits and share-based payment expenses; • marketing and promotional activities related to the global commercialization of VYVGART; • professional fees related to commercial and enabling functions; • Board of Directors expenses consisting of directors’ fees, travel expenses and share-based compensation for non-executive directors; and • other expenses. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Results of Operations 185

Our selling, general and administrative expenses totaled $1.4 billion and $1.1 billion for the years ended December 31, 2025 and 2024, respectively. The increase of $0.3 billion for the year ended December 31, 2025 principally resulted from: • increased professional and marketing fees, including promotional and marketing costs primarily due to the scaling of our commercial operations relating to VYVGART; • increased costs of personnel expenses, related to planned increase in the headcount of our Selling, general and administrative employees recruited to strengthen our enabling functions and the scaling of our commercial operations relating to VYVGART; and • continued investment in our Digital Technology infrastructure. Selling, general and administrative expenses are presented under ‘Note 18 Selling, General and Administrative Expenses’’ in our consolidated financial statements which are included to our Annual Report for the period ended December 31, 2025. Financial Income and (Expense) For the year ended December 31, 2025, financial income amounted to $163 million compared to $158 million for the year ended December 31, 2024. The increase of $6 million in 2025 related primarily to the capital increase of our cash, cash equivalents, and current financial assets. 5.5 Liquidity and Capital Resources 5.5.1 Sources of Funds Our capitalization is detailed in the “Consolidated Statements of Financial Position” which are included to our Annual Report for the period ended December 31, 2025. As of December 31, 2025 on an actual basis, the Company had a total equity amount of $7.3 billion. Since our inception in 2008, we have invested most of our resources in developing our product candidates, building our intellectual property portfolio, developing our supply chain, conducting business planning, raising capital and providing general and administrative support for these operations. To date, we have funded our operations through (i) public and private placements of equity securities, (ii) upfront, milestone and expense reimbursement payments received from our collaborators, (iii) funding from governmental bodies, (iv) proceeds from exercise of employee stock options and (v) interest income from the investment of our cash and cash equivalents, in addition to current financial assets. Through December 31, 2025, we have raised gross proceeds of $5.9 billion from private and public offerings of equity securities. Our commercial operations have also started to contribute to the funding of our operations based on positive cash flow from operating activities as of the year ended December 31, 2025. As we continue to invest in innovation, our cash flows may fluctuate, are difficult to forecast and will depend on many factors. We have no ongoing material financing commitments, such as lines of credit or guarantees, that are expected to affect our liquidity over the next five years, other than leases and commitments as part of our operations, which are detailed in “Note 27 Commitments” and “Note 24 Financial Risk Management” in our consolidated financial statements which are included to our Annual Report for the period ended December 31, 2025. For more information as to the risks associated with our future funding needs, see “Risk Factors — Risk Factors Related to argenx’s Financial Position”. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Results of Operations 186

For more information as to our financial instruments, please see “Note 24 Financial Risk Management” in our consolidated financial statements which are included to our Annual Report for the period ended December 31, 2025. 5.5.2 Cash Flows Comparison for the Years Ended December 31, 2025 and 2024 As of December 31, 2025, the Company had $3.5 billion of cash and cash equivalents compared to $1.5 billion as of December 31, 2024. The Company’s cash and cash equivalents increased by $2.0 billion year-over-year mainly resulting from positive cash flow from operating activities and a higher amount of capital held in cash and cash equivalents as opposed to current financial assets. The Company’s net cash flow from operating activities increased by $0.8 billion for the year ended December 31, 2025 compared to the year ended December 31, 2024 mainly due to increased product net sales of VYVGART partially offset by buildup of working capital. Net cash flow used in investing activities increased by $1.7 billion for the year ended December 31, 2025 compared to the year ended December 31, 2024 mainly due to the nature of financial instruments held as of the reporting date classified as cash and cash equivalents coming from capital held in the year as current financial assets. This is partially offset by payments related to regulatory and sales based milestones to Halozyme. Net cash flow from financing activities decreased by $47 million for the year ended December 31, 2025 compared to the year ended December 31, 2024 mainly due to proceeds from the exercise of stock options. For more information, please see “Consolidated Statements of Cash Flows” and “Note 11 Cash and Cash Equivalents” in our consolidated financial statements which are included to our Annual Report for the period ended December 31, 2025. 5.5.3 Operating and Capital Expenditure Requirements We recorded a profit of $1.3 billion for the year ended December 31, 2025. Our operating expenditures are detailed above in our research and development expenses along with our Selling, general and administrative expenses. We anticipate that our operating expenses will increase as we intend to continue conducting research and development, as well as continuing our efforts to expand our sales & marketing and establish our distribution infrastructure. Although we have generated product net sales of $4.2 billion from global product net sales of VYVGART for the year ended December 31, 2025, which supports our current profitability, we cannot provide assurances that we will be profitable or able to sustain net profitability in the future based on these indications alone. Furthermore, we cannot provide any assurances that we will receive the regulatory approvals to commercialize VYVGART in other indications or in other countries. On the basis of current assumptions, we expect that our existing cash and cash equivalents and current financial assets will enable us to fund our operating expenses and capital expenditure requirements through at least the next twelve months. The adequacy of our available funds to meet our future operating expenses and capital expenditures will depend on numerous risks and uncertainties associated with the development and commercialization of efgartigimod and our other product candidates and discovery stage programs and because the extent to which we may enter into collaborations with third parties for the development of these product candidates is unknown. We are unable to estimate the amounts of increased capital outlays and operating expenses associated with completing the research and development of our product candidates. Our future capital requirements for efgartigimod, empasiprubart, adimanebart, our other product candidates and discovery stage programs will depend on many factors, including: argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Liquidity and Capital Resources 187

• the progress, timing and completion of preclinical testing and clinical trials for our current or any future product candidates; • the number of potential new product candidates we identify and decide to develop; • the time and costs involved in obtaining regulatory approvals for our product candidates and any delays we may encounter as a result of evolving regulatory requirements or adverse results with respect to any of our product candidates; • selling and marketing activities undertaken in connection with the commercialization of VYVGART or potential commercialization of any of our current or any future product candidates, if approved, and costs involved in the creation of an effective sales and marketing organization; • manufacturing activities undertaken for VYVGART and potential commercialization of any of our current or any future product candidates, if approved, and costs involved in the creation of an effective supply chain; • the costs involved in growing our organization to the size needed to allow for the research, development and potential commercialization of our current or any future product candidates; • the costs involved in filing patent applications and maintaining and enforcing patents or defending against claims or infringements raised by third parties; • the maintenance of our existing collaboration agreements, the entry into new collaboration agreements and the pursuit of other strategic business development opportunities; and • developments related to global economic uncertainties and political instability. For more information as to the risks associated with our future funding needs, see “Risk Factors — Risk Factors Related to argenx’s Financial Position". 5.5.4 Working capital statement In our opinion, the working capital of the Company is sufficient for the Company’s present requirements, at least for a period of 12 months from the date of this Annual Report. 5.5.5 Cash Investment Policy The Company has adopted a policy whereby cash and cash equivalents and current financial assets are invested with several highly reputable banks and financial institutions. The main purpose of the Cash Investment Policy is to preserve the available cash and to ensure sufficient short-term liquidity at all times. Therefore, the Company holds its cash, cash equivalents and current financial assets mainly with banks which are independently rated A- or higher. Amounts of cash held with banks rated lower than A- are limited to insignificant balances. The maximum amount and tenor of time deposits depends on the rating of the counterparty bank. The Company also holds cash equivalents in the form of money market funds with a low historical volatility. These money market funds are highly liquid investments and can be readily convertible into a known amount of cash. The Company has adopted a policy whereby money market funds must have a minimum rating of A of which 95% should have a AAA-rating. For more information as to our treasury policy and liquidity, please see “Note 24 Financial Risk Management” in our consolidated financial statements which are included to our Annual Report for the period ended December 31, 2025. 5.6 Research and development, patents and licenses For a discussion of our research and development policies, refer to the “Overview” and “Results of Operations” within Section 5. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Liquidity and Capital Resources 188

5.7 Trend information Other than as disclosed elsewhere in this Annual Report, we are not aware of any trends, uncertainties, demands, commitments or events for the current financial period that are reasonably likely to have a material effect on our net revenues, income, profitability, liquidity, capital resources or prospects, or that caused the disclosed financial information to be not necessarily indicative of future operating results or financial conditions. There has been no significant change in the financial performance or the financial position of the Group since the balance sheet date of December 31, 2025. For more information, please refer to “Overview”, “Operating Results”,“Liquidity and Capital Resources” within Section 5 and to ‘‘Note 27 Commitments’’ in our consolidated financial statements which are appended to our Annual Report for the period ended December 31, 2025. 5.8 Off-Balance Sheet Arrangements During the periods presented we did not have, and we do not currently have, any off-balance sheet arrangements, as defined in the applicable rules and regulations, such as relationships with unconsolidated entities or financial partnerships, which are often referred to as structured finance or special purpose entities, established for the purpose of facilitating financing transactions that are not required to be reflected on our balance sheets. Our scope of consolidated entities is disclosed in “Note 29 Overview of Consolidation Scope”. 5.9 Contractual Obligations For a discussion of contractual obligations, please see ‘‘Note 27 Commitments” in our consolidated financial statements which are appended to our Annual Report for the period ended December 31, 2025. 5.10 Information Regarding the Independent Auditor The audited financial statements as of and for the year ended December 31, 2025 have been audited by our independent auditor, EY Accountants B.V. (EY). The partner of EY who signed the auditor’s reports is a member of the Netherlands Institute of Chartered Accountants (Koninklijke Nederlandse Beroepsorganisatie van Accountants). The office of EY is located at Prof. Dr. Dorgelolaan 12, 5613 AM, Eindhoven, the Netherlands. The audited consolidated financial statements as of and for the year ended December 31, 2024 and 2023 have been audited by our (previously appointed) independent auditor, Deloitte Accountants B.V. (Deloitte), who rendered unqualified audit reports on these financial statements. The partner of Deloitte who signed the auditors’ reports is a member of the Netherlands Institute of Chartered Accountants (Koninklijke Nederlandse Beroepsorganisatie van Accountants). The office of Deloitte is located at Wilhelminakade 1, 3072 AP Rotterdam, the Netherlands. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Trend information 189

5.11 Material Contracts and Related Party Transactions 5.11.1 Material Contracts Our material contracts are described in sections “Collaborations and Licenses”. 5.11.2 Related Party Transactions Since January 1, 2025, we have not entered into any transactions with any related parties which are, as a single transaction or in their entirety, material to us other than that which is described in ‘‘Note 25 Related Party Transactions“ in our consolidated financial statements which are appended to our Annual Report for the period ended December 31, 2025. In addition, since January 1, 2025, there has not been, nor is there currently proposed, any material transaction or series of similar material transactions to which we were or are a party in which any of the members of our Board of Directors or senior management, holders of more than 10% of any class of our voting securities, or any member of the immediate family of any of the foregoing persons, had or will have a direct or indirect material interest, other than the compensation and shareholding arrangements we describe in section “Share Classes and Principal Shareholders”, and the transactions we describe below. Transactions with Related Companies From time to time, in the ordinary course of our business, we may contract for services from companies in which certain of the members of our senior management or directors may serve as director or advisor. The costs of these services are negotiated on an arm’s length basis and none of these arrangements are material to us. Agreements with Our Senior Management Team Other than as set forth in this Annual Report, there are no arrangements or understandings in place with major shareholders, customers, suppliers or others pursuant to which any member of our Board of Directors or Senior Management Team has been appointed. We have entered into a management agreement with Tim Van Hauwermeiren as our CEO, our sole Executive Director. The key terms of his agreement are as follows: Tim Van Hauwermeiren Fixed-base compensation $827,160 Short-term variable compensation A target of 60% of the fixed-base compensation based on previously determined bonus targets established by the non-executive directors Pension contributions1) $44,168 Duration Indefinite 1) Amounts shown represent pension contributions paid during the year ended December 31, 2025. As announced on January 5, 2026, there will be a leadership transition in May 2026. Peter Verhaeghe will step down as Non-Executive Director and chairperson of the Board of Directors at the 2026 General Meeting. Tim Van Hauwermeiren will step down from his current CEO role and it is envisaged that he will be appointed as a Non-Executive Director at the 2026 General Meeting and subsequently elected as by the Board of Directors as the chairperson of the Board of Directors. Tim van Hauwermeiren’s management agreement will therefore be terminated. It is also envisaged that Karen Massey, our current COO, will be appointed as an Executive Director at the 2026 General Meeting and subsequently elected as CEO by the Board of Directors. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Material Contracts and Related Party Transactions 190

Karen Massey currently holds the role of COO and has an employment contract with our subsidiary, argenx Switzerland SA, for an indefinite term. The 2026 annual report will reflect the main terms of her agreements with the Company and/or its subsidiaries. Karl Gubitz, our chief financial officer, has an employment contract with our subsidiary, argenx US Inc., for an indefinite term. Peter Ulrichts, our chief scientific officer has an employment contract with our subsidiary, argenx B.V., for an indefinite term. Arjen Lemmen, our vice president corporate development and strategy, has an employment contract with our subsidiary, argenx B.V., for an indefinite term. Andria Wilk, our global head of quality, has an employment contract with our subsidiary, argenx B.V., for an indefinite term. Malini Moorthy, our general counsel and corporate secretary has an employment contract with our subsidiary, argenx US, for an indefinite term. Ms. Moorthy has also entered into a secondment agreement with argenx US, under which Ms. Moorthy was seconded from argenx US to argenx B.V. and was based in Belgium for the period of April 1, 2023 through December 31, 2024. This secondment was extended through December 31, 2026. Luc Truyen, our head of research and development management operations and our chief medical officer, has an employment contract with our subsidiary, argenx US Inc. for an indefinite term. Mr. Truyen entered into a secondment agreement with argenx US Inc., under which Mr. Truyen has been seconded from argenx US Inc. to argenx B.V. and is based in Belgium for the period of April 1, 2022 through November 30, 2026 (unless otherwise extended by the parties). Indemnification Agreements In connection with our initial U.S. public offering, we entered into indemnification agreements with each of our Non-Executive Directors and each member of our Senior Management Team. We have entered into such agreements with each new non-executive director or member of our senior management when they have joined us since our initial U.S. public offering. Insofar as indemnification for liabilities arising under the Securities Act may be permitted to non-executive directors, officers or persons controlling us pursuant to the foregoing provisions, we have been informed that, in the opinion of the SEC such indemnification is against public policy as expressed in the Securities Act and is therefore unenforceable. Related Party Transactions Policy In connection with our initial U.S. public offering, we entered into a related party transaction policy. Our Code of Conduct and our Board Rules also include specific rules of transactions with related parties. Property, plants and equipment Our principal executive, operational offices and laboratory space are located in Zwijnaarde, Belgium. In 2024, we added new office space in Zwijnaarde. The total future cash flows related to these leases are represented below in Note 20 Leases in our consolidated financial statements which are included to our Annual Report for the period ended December 31, 2025. We also lease office space in Amsterdam (the Netherlands), Boston (U.S.), Tokyo (Japan), Geneva (Switzerland), Munich (Germany), Issy-Les-Moulineaux (France), Vaughan, Ontario (Canada), Gerrards Cross (UK), Milan (Italy), Madrid (Spain) and Sydney (Australia). In addition, our lease liabilities include a lease plan for company cars with maturity dates up to four years. For a discussion of contractual obligations, please see Note 27 Commitments in our consolidated financial statements which are included to our Annual Report for the period ended December 31, 2025. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Material Contracts and Related Party Transactions 191
Below are the key facilities worldwide leased as of December 31, 2025: Facility location Use Approx. size (m2) Lease expiry Zwijnaarde, Belgium (leased) Operations and Laboratory Space 5,339 September 30, 2031 Zwijnaarde, Belgium (leased) Office Space 3,765 September 30, 2036 Boston, Massachusetts (leased) Office Space 2,379 August 31, 2030 Tokyo, Japan (leased) Office Space 546 January 17, 2027 Environment, Health and Safety Our primary research and development activities take place in our facilities in Zwijnaarde, Belgium. For these activities we require, and have obtained, the necessary environmental and biohazard permits from the responsible governments, required by us for the manner in which we use said facilities. See section “Risk Factors”. 5.12 Employees As of December 31, 2025, we had 1,863 employees and 1659 consultants, which we refer to as “contingent workers”. At each date shown below, we had the following number of employees, broken out by department and geography. As of December 31, 2025 2024 2023 Function: Research and development 773 644 653 Selling, general and administrative 1,090 955 495 Total 1,863 1,599 1,148 Geography: U.S. 789 694 454 Belgium 565 466 355 Japan 146 139 116 Switzerland 71 49 28 Germany 55 41 25 UK 48 44 37 Italy 39 33 27 France 38 38 40 The Netherlands 38 34 22 Spain 34 32 20 Canada 24 19 16 Rest of the World/Remote 16 10 8 Total 1,863 1,599 1,148 Collective bargaining agreements (CBAs) can be entered into in Belgium and other jurisdictions at the national, industry, or company levels. These CBAs are binding on both employers and employees. We have no trade union representation or CBAs at the company level, but we are subject to the national and chemical industry CBAs. The CBAs currently applicable to us relate to employment conditions such as wages, working time, job security, innovation and supplementary pensions. We have not had, and do not anticipate having, disputes on any of these subjects. CBAs may, however, change the employment conditions of our employees in the future and hence adversely affect our employment relationships. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Material Contracts and Related Party Transactions 192

5.13 Insurance We maintain an insurance portfolio that is common and appropriate for our business. Our main insurances are commercial general liability insurances, including products liability insurance, director and officer liability insurance and our maritime insurance covering the risk of loss of product during transit and storage. 5.14 Legal and Arbitration Proceedings From time to time we may become involved in legal, governmental or arbitration proceedings or be subject to claims arising in the ordinary course of our business. Regardless of the outcome, litigation can have an adverse impact on us because of defense and settlement costs, diversion of management resources and other factors. During the previous 12 months, there have not been any legal, governmental or arbitration proceedings (including any such proceedings which are pending or threatened of which we are aware) which may have, or have had in the recent past, significant effects on argenx and/or the Group’s financial position or profitability. 5.15 Taxation This summary does not consider your particular circumstances. We urge you to consult your own independent tax advisors about the income, capital gains and/or transfer tax consequences to you in light of your particular circumstances of purchasing, holding and disposing of ordinary shares or ADSs. 5.15.1 U.S. Federal Income Tax Considerations The following discussion is a summary under present law of certain material U.S. federal income tax considerations relating to the ownership and disposition of ADSs by a U.S. holder (as defined below). This summary addresses only the U.S. federal income tax considerations for U.S. holders that hold ADSs as capital assets (generally, property held for investment) and use the U.S. dollar as their functional currency. This summary does not address all U.S. federal income tax matters that may be relevant to a particular U.S. holder and is not a substitute for tax advice. This summary does not address tax considerations applicable to a holder of ADSs that may be subject to special tax rules including, without limitation, banks, financial institutions or insurance companies, brokers, dealers or traders in securities, currencies, commodities, or notional principal contracts, traders in securities that elect to mark-to-market, tax-exempt entities or organizations, including “individual retirement accounts” or “Roth IRAs”, real estate investment trusts, regulated investment companies, persons that hold the ADSs as part of a “hedging,” “integrated” or “conversion” transaction or as a position in a “straddle”, partnerships (including entities or arrangements classified as partnerships for U.S. federal income tax purposes) or other pass-through entities (including S-corporations), or persons that will hold the ADSs through such an entity, certain former citizens or long-term residents of the United States, persons that received the ADSs as compensation for the performance of services, persons subject to special tax accounting rules as a result of any item of gross income with respect to the shares being taken into account in an applicable financial statement, and holders that own directly, indirectly, or through attribution 10% or more of the voting power or value of our ordinary shares and ADSs. This summary does not address U.S. federal taxes other than the income tax (such as the Medicare surtax on net investment income, the estate, gift, or alternative minimum tax), any election to apply Section 1400Z-2 of the U.S. Internal Revenue Code of 1986, as amended (the Code) to gains recognized with respect to ADSs, or any U.S. state, local, or non-U.S. tax considerations of the ownership and disposition of ADSs. For the purposes of this summary, a “U.S. holder” is a beneficial owner of ADSs that is (or is treated as), for U.S. federal income tax purposes, (i) an individual who is a citizen or resident of the United States, (ii) a corporation, or any other entity treated as a corporation for U.S. federal income tax purposes, created or argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Insurance 193

organized in or under the laws of the United States, any state thereof, or the District of Columbia, (iii) an estate, the income of which is subject to U.S. federal income taxation regardless of its source, or a trust, if a court within the United States is able to exercise primary supervision over its administration and one or more U.S. persons have the authority to control all of the substantial decisions of such trust. If a partnership (or other entity or arrangement treated as a partnership for U.S. federal income tax purposes) holds ADSs, the U.S. federal income tax consequences relating to an investment in those ADSs will depend in part upon the status of the partner and the activities of the partnership. A partnership that holds ADSs should consult its tax advisor regarding the U.S. federal income tax considerations for it and for its partners of owning and disposing of ADSs in its and their particular circumstances. In general, a U.S. holder that owns ADSs will be treated as the beneficial owner of the underlying shares represented by those ADSs for U.S. federal income tax purposes. Accordingly, no gain or loss will generally be recognized if a U.S. holder exchanges ADSs for the underlying shares represented by those ADSs. Persons considering an investment in the ADSs should consult their own tax advisors as to the particular tax consequences applicable to them relating to the ownership and disposition of ADSs, including the applicability of U.S. federal, state and local tax laws and non-U.S. tax laws. Distributions Although we do not currently plan to pay dividends, and subject to the discussion under “Passive Foreign Investment Company Considerations” below, the gross amount of distributions paid with respect to our ordinary shares including Dutch or Belgian tax withheld therefrom, if any (other than pro rata distribution), generally will be included in a U.S. holder’s gross income as foreign source ordinary dividend income when actually or constructively received to the extent such distribution is paid out of our current and accumulated earnings and profits as determined under U.S. federal income tax principles. Distributions in excess of our current and accumulated earnings and profits will be treated as a non-taxable return of capital and will be applied against and reduce, the U.S. holder’s adjusted tax basis in ADSs (but not below zero) and distributions in excess of earnings and profits and a U.S. holder’s adjusted tax basis will generally be taxable to the U.S. holder as either long-term or short-term capital gain depending upon whether the U.S. holder has held the ADSs for more than one year as of the time such distribution is received. However, since we do not calculate our earnings and profits under U.S. federal income tax principles, it is expected that any distribution will be reported as a dividend, even if that distribution would otherwise be treated as a non-taxable return of capital or as capital gain. Our dividends will not be eligible for the dividends-received deduction generally allowed to U.S. corporations. Dividends paid to non-corporate U.S. holders that satisfy a minimum holding period (during which they are not protected from the risk of loss) and certain other requirements may qualify for the preferential favorable tax rates applicable to qualified dividend income, provided that we are a “qualified foreign corporation” and we are not a PFIC as to the non-corporate U.S. holder in the taxable year of the dividend or the preceding taxable year. A qualified foreign corporation includes a non-U.S. corporation that is eligible for the benefits of a comprehensive income tax treaties with the United States. A non-U.S. corporation also will be considered to be a qualified foreign corporation with respect to any dividend it pays on shares which are readily tradable on an established securities market in the United States. Our ADSs are listed on Nasdaq, which is an established securities market in the United States, and we expect our ADSs to be readily tradable on Nasdaq. However, there can be no assurance that the ADSs will be considered readily tradable on an established securities market in the United States in any taxable year. U.S. holders should consult their own tax advisors regarding the application of these rules given their particular circumstances. Subject to generally applicable limitations, a U.S. holder may be entitled to claim a U.S. foreign tax credit for Dutch withholding tax imposed at the appropriate rate. U.S. holders who do not elect to claim a credit for any foreign income taxes paid or accrued during the taxable year may instead claim a deduction of such taxes. The rules relating to the foreign tax credit are complex. Each U.S. holder should consult its own tax advisors regarding the foreign tax credit rules. In general, the amount of a distribution paid to a U.S. holder in a foreign currency will be the dollar value of the foreign currency calculated by reference to the applicable exchange rate on the day the U.S. holder receives the distribution, regardless of whether the foreign currency is converted into U.S. dollars at that time. Any foreign currency gain or loss a U.S. holder realizes on a subsequent conversion of foreign argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Taxation 194

currency into U.S. dollars will be U.S. source ordinary income or loss. If dividends received in a foreign currency are converted into U.S. dollars on the day they are received, a U.S. holder should not be required to recognize foreign currency gain or loss in respect of the dividend. Sale, Exchange or Other Taxable Disposition of ADSs Subject to the discussion under “Passive Foreign Investment Company Considerations” below, a U.S. holder will generally recognize capital gain or loss on the sale, exchange or other taxable disposition of ADSs in an amount equal to the difference between the amount realized from such sale or exchange and the U.S. holder’s adjusted basis in the ADSs, each amount determined in USD. The adjusted tax basis in ADSs generally will be equal to the U.S. dollar cost of such ADSs. Any such capital gain or loss generally will be long-term capital gain or loss if the U.S. holder’s holding period for such ADSs exceeds one year as of the date of sale or other disposition. Long-term capital realized by a non-corporate U.S. holder is generally eligible for a preferential reduced rates. The deductibility of capital losses for U.S. federal income tax purposes is subject to certain limitations. Any such gain or loss that a U.S. holder recognizes generally will be treated as U.S. source income or loss for foreign tax credit limitation purposes. Passive Foreign Investment Company Considerations In general, a non-U.S. corporation will be classified as a passive foreign investment company, or PFIC, for any taxable year in which, after applying certain look-through rules with respect to certain dividends, rents, interest or royalties received from its affiliates and taking into account its proportionate share of the income and assets of its 25% or more owned subsidiaries, either: (i) at least 75% of its gross income is “passive income” or (ii) at least 50% of the average quarterly value of its total gross assets is attributable to cash in excess of working capital requirements or assets that produce “passive income” or are held for the production of “passive income”. Passive income for this purpose generally includes dividends, interest, royalties, rents, gains from commodities and securities transactions, the excess of gains over losses from the disposition of assets which produce passive income. While we are treated as a publicly traded company for these purposes, the value of our assets, including goodwill and other intangibles, will be based on their fair market value, which will depend on the market value of our ordinary shares and ADSs, which are subject to change. Based on our historic and anticipated operations, the composition of our income and the projected composition and estimated fair market values of our assets, we do not believe that we were a PFIC for our most recent taxable year and do not expect to be classified as a PFIC for the current taxable year or for the foreseeable future. However, our possible status as a PFIC is a factual determination made annually after the close of each taxable year and, therefore, may be subject to change. Accordingly, there can be no assurance that we will not be a PFIC for any year in which a U.S. holder holds ADSs. The Company does not intend to provide any annual assessments of its PFIC status. If we were to be classified as a PFIC for any taxable year during which a U.S. holder owns ADSs, gain recognized on a sale or other disposition (including certain pledges) of such U.S. holder’s ADSs would be allocated ratably over such U.S. holder’s holding period. Amounts allocated to the taxable year of the sale or disposition and to any year before we became a PFIC would be taxed as ordinary income and the amount allocated to each other taxable year would be subject to tax at the highest rate in effect for individuals or corporations, as appropriate, for that taxable year, and an interest charge will be imposed on the resulting tax liability for each such year. In addition, to the extent that distributions received by a U.S. holder on its ADSs in any taxable year exceed 125% of the average of the annual distributions on such holder’s ADSs received during the preceding three taxable years (or, if shorter, the U.S. holder’s holding period), such excess distributions will be subject to taxation in the same manner. Furthermore, dividends that are not excess distributions would not be eligible for the preferential tax rate applicable to qualified dividend income received by individuals and certain other non-corporate persons. If the Company is a PFIC for any taxable year during which you own ADSs, the Company will generally continue to be treated as a PFIC with respect to you for all succeeding years during which you own the ADSs, even if the Company ceases to meet the threshold requirements for PFIC status. Certain elections may be available that will result in alternative treatments (such as mark-to-market treatment) of the Shares. U.S. holders should consult their own tax advisors concerning the Company’s possible PFIC status and the consequences to them if the Company were a PFIC for any taxable year, including whether any of these argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Taxation 195

elections will be available, and, if so, what the consequences of the alternative treatments will be in your particular circumstances. Backup Withholding and Information Reporting U.S. holders generally will be subject to information reporting requirements with respect to dividends on ADSs and on the proceeds from the sale, exchange or disposition of the ADSs that are paid within the United States or through U.S.- related financial intermediaries, unless the U.S. holder is a corporation or other “exempt recipient.” In addition, U.S. holders may be subject to backup withholding on such payments, unless the U.S. holder provides a correct taxpayer identification number and a duly executed IRS Form W-9 or otherwise establishes an exemption. Backup withholding is not an additional tax, and the amount of any backup withholding will be allowed as a credit against a U.S. holder’s U.S. federal income tax liability and may entitle such holder to a refund, provided that the required information is timely furnished to the IRS. Foreign Asset Reporting Certain U.S. holders who are individuals and certain entities controlled by individuals may be required to report information relating to an interest in ADSs, subject to certain exceptions (including an exception for shares held in accounts maintained by U.S. financial institutions) by filing IRS Form 8938 (Statement of Specified Foreign Financial Assets) with their federal income tax return. Investors who fail to report required information could become subject to substantial penalties. U.S. holders are urged to consult their tax advisors regarding their information reporting obligations, if any, with respect to their ownership and disposition of the ADSs. 5.15.2 Material Dutch Tax Consequences The following summary outlines certain material Dutch tax consequences in connection with the acquisition, ownership and disposal of the ADSs. All references in this summary to the Netherlands and Dutch law are to the European part of the Kingdom of the Netherlands and its law, respectively, only. The summary does not purport to present any comprehensive or complete picture of all Dutch tax aspects that could be of relevance to the acquisition, ownership and disposal of the ADSs by a (prospective) holder of the ADSs. Depending on the particular situation of a holder of ADSs, this summary may not describe all potentially relevant Dutch tax consequences in light of such a holder of ADSs’ (specific) circumstances. The summary is based on the tax laws and practice of the Netherlands as in effect on the date of this Annual Report, which are subject to changes that could prospectively or retrospectively affect the Dutch tax consequences. This summary does not address the Dutch tax consequences for a holder of ADSs that is considered to be affiliated (gelieerd) to the Company within the meaning of the Dutch Withholding Tax Act 2021 (Wet bronbelasting 2021). Generally, a holder of ADSs is considered to be affiliated to the Company for these purposes if (i) it has a qualifying interest in the Company, (ii) the Company has a qualifying interest in such party, or (iii) a third party has a qualifying interest in both the Company and such party. A party is equated with any qualifying unity (kwalificerende eenheid) of parties of which it forms part. A qualifying unity is defined as entities that have been established and/or are acting jointly with the primary purpose, or one of the primary purposes, to avoid the imposition of tax on one or more of such entities, for example where the controlling interest (to be) held is divided into various non-controlling interests with the primary purpose, or one of the primary purposes, to avoid the aforementioned tax. A qualifying interest is an interest that allows the holder to have a decisive influence over the other party’s decisions, in such a way that it is able to determine the activities of the other party. A party is in any case considered to have a qualifying interest in another party if it (directly or indirectly) owns more than 50 per cent. of the voting rights in such other party. For purposes of Dutch income and corporate income tax, shares, or certain other assets, which may include depositary receipts in respect of shares, legally owned by a third party such as a trustee, foundation or similar entity or arrangement, a “Third Party”, may under certain circumstances have to be allocated to the (deemed) settlor, grantor or similar originator, the “Settlor”, or, upon the death of the Settlor, such Settlor’s beneficiaries, the “Beneficiaries”, in proportion to their entitlement to the estate of the Settlor of such trust or similar arrangement, the “Separated Private Assets”. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Taxation 196

The summary does not address the Dutch tax consequences of a holder of the ADSs who is an individual and who has a substantial interest (aanmerkelijk belang) in the Company. Generally, a holder of the ADSs will have a substantial interest in the Company if such holder of the ADSs, whether alone or together with such holder’s spouse or partner and/or certain other close relatives, holds directly or indirectly, or as Settlor or Beneficiary of Separated Private Assets (i) (x) the ownership of, (y) certain other rights, such as usufruct, over, or (z) rights to acquire (whether or not already issued), shares (including the ADSs) representing 5% or more of the total issued and outstanding capital (or the issued and outstanding capital of any class of shares) of the Company or (ii) (x) the ownership of, or (y) certain other rights, such as usufruct over, profit participating certificates (winstbewijzen) that relate to 5% or more of the annual profit of the Company or to 5% or more of the liquidation proceeds of the Company. In addition, a holder of the ADSs has a substantial interest in the Company if such holder, whether alone or together with such holder’s spouse or partner and/or certain other close relatives, has the ownership of, or other rights over, shares, or depositary receipts in respect of shares, in, or profit certificates issued by, the Company that represent less than 5% of the relevant aggregate that either (a) qualified as part of a substantial interest as set forth above and where shares, or depositary receipts in respect of shares, profit certificates and/or rights there over have been, or are deemed to have been, partially disposed of, or (b) have been acquired as part of a transaction that qualified for non-recognition of gain treatment. Furthermore, this summary does not address the Dutch tax consequences of a holder of the ADSs: • who is an individual and receives income or realizes capital gains in respect of the ADSs in connection with such holder’s employment activities or in such holder’s capacity as (former) board member or (former) supervisory board member; • who is a resident of any non-European part of the Kingdom of the Netherlands; or • in respect of the Dutch Minimum Taxation Act 2024 (Wet minimumbelasting 2024), (which is the Dutch implementation of Council Directive (EU) 2022/2523 of 14 December 2022 on ensuring a global minimum level of taxation for multinational enterprise groups and large-scale domestic groups in the EU). Dividend Withholding Tax General The Company is generally required to withhold dividend withholding tax imposed by the Netherlands at a rate of 15% on dividends distributed by the Company in respect of our ordinary shares underlying the ADSs. The expression “dividends distributed by the Company” as used herein includes, but is not limited to: (a) distributions in cash or in kind, deemed and constructive distributions and repayments of paid-in capital (gestort kapitaal) not recognized for Dutch dividend withholding tax purposes; (b) liquidation proceeds, proceeds of redemption of our ordinary shares or, as a rule, consideration for the repurchase of our ordinary shares by the Company in excess of the average paid-in capital recognized for Dutch dividend withholding tax purposes; (c) the par value of our ordinary shares issued to a holder of our ordinary shares or an increase of the par value of our ordinary shares, to the extent that it does not appear that a contribution, recognized for Dutch dividend withholding tax purposes, has been made or will be made; and (d) partial repayment of paid-in capital, recognized for Dutch dividend withholding tax purposes, if and to the extent that there are net profits (zuivere winst), unless (i) the shareholders at a General Meeting have resolved in advance to make such repayment and (ii) the par value of our ordinary shares concerned has been reduced by an equal amount by way of an amendment of our articles of association. Holders of the ADSs Resident in the Netherlands A holder of the ADSs that is an individual that is resident or deemed to be resident in the Netherlands for Dutch tax purposes is generally entitled, subject to the anti-dividend stripping rules described below, to a full credit against its income tax liability, or a full refund, of the Dutch dividend withholding tax. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Taxation 197

A holder of the ADSs that is a legal entity that is resident or deemed to be resident in the Netherlands for Dutch tax purposes is generally entitled, subject to the anti-dividend stripping rules described below, to a full credit against its corporate income tax liability of the Dutch dividend withholding tax. If and to the extent such legal entity cannot credit the full amount of Dutch dividend withholding tax in a given year, the Dutch dividend withholding tax may be carried forward and credited against its corporate income tax liability in subsequent years (without time limitation). The two previous paragraphs generally apply to holders of the ADSs that are neither resident nor deemed to be resident in the Netherlands for Dutch tax purposes if the ADSs are attributable to a permanent establishment (vaste inrichting) or a permanent representative (vaste vertegenwoordiger) in the Netherlands of such non-resident holder of ADSs. A holder of the ADSs that is a legal entity that is resident or deemed to be resident in the Netherlands for Dutch tax purposes that is exempt from Dutch corporate income tax but that is not qualifying exempt investment institution (vrijgestelde beleggingsinstelling), is generally entitled, subject to the anti-dividend stripping rules described below, to an exemption at source (subject to the completion of necessary procedural formalities) or a full refund of Dutch dividend withholding tax on dividends received. Holders of the ADSs Resident Outside the Netherlands A holder of the ADSs that is resident in a country for tax purposes with which the Netherlands has a tax treaty in effect, may, depending on the terms of such tax treaty and subject to the anti-dividend stripping rules described below, be eligible for a full or partial exemption from, or full or partial refund of, Dutch dividend withholding tax on dividends received. A holder of the ADSs, that is a legal entity (a) tax resident in (i) an EU Member State, (ii) Iceland, Norway or Liechtenstein, or (iii) a country with which the Netherlands has concluded a tax treaty that includes an article on dividends and (b) that is in its state of residence under the terms of a tax treaty concluded with a third state, not considered to be resident for tax purposes in a country with which the Netherlands has not concluded a tax treaty that includes an article on dividends (i.e., not an EU Member State, Iceland, Norway or Liechtenstein), is generally entitled, subject to the anti-abuse rules and the anti-dividend stripping rules described below, to a full exemption from Dutch dividend withholding tax on dividends received if it holds an interest of, generally, at least 5% of the nominal share capital of the Company in respect of which, had such holder been a tax resident in the Netherlands, the participation exemption (deelnemingsvrijstelling) would have applied. The full exemption from Dutch dividend withholding tax on dividends received by a holder of the ADSs is not granted if (x) the interest held by such holder (i) is held for the avoidance of Dutch dividend withholding tax of another person as (one of) the main purpose(s) and (ii) forms part of an artificial structure or series of structures (such as structures which are not put into place for valid business reasons reflecting economic reality), or (y) the holder of ADSs has a similar function to a qualifying investment institution (fiscale beleggingsinstelling) or a qualifying exempt investment institution (vrijgestelde beleggingsinstelling). A holder of the ADSs, that is an entity tax resident in (i) an EU Member State or (ii) Iceland, Norway or Liechtenstein, or (iii) in a jurisdiction which has an arrangement for the exchange of tax information with the Netherlands (and such holder as described under (iii) holds the ADSs as a portfolio investment (i.e., such holding is not acquired with a view to the establishment or maintenance of lasting and direct economic links between the holder of the ADSs and the Company and does not allow the holder of the ADSs to participate effectively in the management or control of the Company)), which is exempt from tax in its country of residence and does not have a similar function to a qualifying investment institution (fiscale beleggingsinstelling) or a qualifying exempt investment institution (vrijgestelde beleggingsinstelling), and that would have been exempt from Dutch corporate income tax if it had been a resident of the Netherlands, is generally entitled, subject to the anti-dividend stripping rules described below, to an exemption or a full refund of Dutch dividend withholding tax on dividends received. This exemption or full refund will in general benefit certain foreign pension funds, government agencies and certain government controlled commercial entities. No exemption, reduction, credit or refund of Dutch dividend withholding tax will be granted if the recipient of the dividend paid by the Company is not considered the beneficial owner (uiteindelijk gerechtigde) of the dividend. A recipient of a dividend is in any case not considered the beneficial owner of the dividend argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Taxation 198

pursuant to the anti-dividend stripping rules if, as a consequence of a combination of transactions and tested at group level, (i) a person (other than the holder of the dividend coupon), directly or indirectly, partly or wholly benefits from the dividend, (ii) such person directly or indirectly retains or acquires a comparable interest in the ADSs, and (iii) such person is entitled to a less favorable exemption, refund or credit of dividend withholding tax than the recipient of the dividend distribution. The term “combination of transactions” includes transactions that have been entered into by parties related to the recipient of the dividend, that have been entered into in the anonymity of a regulated stock market, the sole acquisition of one or more dividend coupons and the establishment of short-term rights or enjoyment on the ADSs (e.g., usufruct). Holders of the ADSs Resident in the U.S. Dividends distributed by the Company to U.S. resident holders of the ADSs that are eligible for benefits under the Convention between the Netherlands and the U.S. for the avoidance of Double Taxation and the Prevention of Fiscal Evasion with respect to Taxes and Income, dated December 18, 1992 as amended by the protocol of March 8, 2004 (U.S. Tax Treaty), generally will be entitled to a reduced dividend withholding tax rate of 5% in case of certain U.S. corporate shareholders owning at least 10% of the Company’s total voting power. Certain U.S. pension funds and tax-exempt organizations may qualify for a complete exemption from Dutch dividend withholding tax. Under the U.S. Tax Treaty such benefits are generally available to U.S. residents if such resident is the beneficial owner of the dividends, provided that such shareholder does not have an enterprise or an interest in an enterprise that is, in whole or in part, carried on through a permanent establishment or permanent representative in the Netherlands and to which enterprise or part of an enterprise the ADSs are attributable. A person may, however, not claim the benefits of the U.S. Tax Treaty if such person’s entitlement to such benefits is limited by the provisions of Article 26 (the limitation on benefits provision) of the U.S. Tax Treaty. The reduced dividend withholding tax rate can generally be applied at source upon the distribution of the dividends, provided that the proper forms have been filed in advance of the distribution. In the case of certain tax-exempt organizations, as a general rule, the so-called refund method applies; only when certain administrative conditions have been fulfilled may such tax-exempt organization use the exemption method. Irrespective of meeting the conditions of the relevant provisions of the U.S. Tax Treaty, dividends distributed by the Company to a U.S. resident holder meeting the criteria set out in the second and third paragraphs under “Holders of the ADSs Resident Outside the Netherlands” above are generally, subject to the anti-dividend stripping rules described above, fully exempt from Dutch dividend withholding tax. Taxes on Income and Capital Gains Holders of the ADSs Resident in the Netherlands: Individuals A holder of the ADSs, who is an individual resident or deemed to be resident in the Netherlands for Dutch tax purposes will be subject to regular Dutch income tax on the income derived from the ADSs and the gains realized upon the redemption and/or disposal of the ADSs by the holder thereof, if: (a) such holder of the ADSs has an enterprise or an interest in an enterprise, to which enterprise the ADSs are attributable; and/or (b) such income or capital gain forms “a benefit from miscellaneous activities” (resultaat uit overige werkzaamheden) which, for instance, would be the case if the activities with respect to the ADSs exceed “normal active asset management” (normaal, actief vermogensbeheer) or if income and gains are derived from the holding, whether directly or indirectly, of (a combination of) shares, debt claims or other rights (together, a “lucrative interest” (lucratief belang)) that the holder thereof has acquired under such circumstances that such income and gains are intended to be remuneration for work or services performed by such holder (or a related person), whether within or outside an employment relation, where such lucrative interest provides the holder thereof, economically speaking, with certain benefits that have a relation to the relevant work or services. If either of the above mentioned conditions (a) or (b) applies, income derived from the ADSs and the gains realized upon the redemption and/or disposal of the ADSs will in general be subject to Dutch income tax at the progressive rates up to 49.5%. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Taxation 199

If the above mentioned conditions (a) and (b) do not apply, a holder of the ADSs who is an individual, resident or deemed to be resident in the Netherlands for Dutch tax purposes will not be subject to taxes on income and capital gains in the Netherlands. Instead, such individual is generally taxed at a flat rate of 36% on deemed income from “savings and investments” (sparen en beleggen), which deemed income is determined on the basis of the amount included in the individual’s “yield basis” (rendementsgrondslag) at the beginning of the calendar year (minus a tax-free threshold; the yield basis minus such threshold being the tax basis). For 2026, the deemed income derived from savings and investments will be a percentage of the tax basis up to % that is determined based on the actual allocation of (i) savings, (ii) other investments, and (iii) debts/liabilities within the individual’s yield basis. The tax-free threshold for 2026 is €59,357. The percentages to determine the deemed income will be reassessed every year (with possible retroactive effect to 1 January). A holder of the ADSs that is able to demonstrate that its tax liability is determined on the basis of the deemed income derived from savings and investments that exceeds the “actual returns” (werkelijk rendement) of such individual may under certain circumstances elect to be taxed on the basis of such “actual returns” (werkelijk rendement) instead. These rules are subject to ongoing litigation and may therefore change. A holder of ADSs may need to file (protective) appeals to any assessments based on these rules to benefit from any beneficial case law. Holders of the ADSs Resident in the Netherlands: Corporate Entities The income derived from the ADSs and the gains realized upon the redemption and/or disposal of the ADSs by any holder of the ADSs that is an entity subject to corporate income tax in the Netherlands is generally subject to Dutch corporate income tax levied at a rate of 25.8% (19% over profits up to and including €200,000), unless, and to the extent that, the participation exemption (deelnemingsvrijstelling) applies. Holders of the ADSs Resident Outside the Netherlands: Individuals A holder of the ADSs who is an individual, not resident or deemed to be resident in the Netherlands will not be subject to any Dutch taxes on income derived from the ADSs and the gains realized upon the redemption and/or disposal of the ADSs, unless: (a) such holder has an enterprise or an interest in an enterprise that is, in whole or in part, carried on through a permanent establishment (vaste inrichting) or a permanent representative (vaste vertegenwoordiger) in the Netherlands and to which enterprise or part of an enterprise, as the case may be, the ADSs are attributable; or (b) such income or capital gain forms a “benefit from miscellaneous activities in the Netherlands” (resultaat uit overige werkzaamheden in Nederland) which would for instance be the case if the activities in the Netherlands with respect to the ADSs exceed “normal active asset management” (normaal, actief vermogensbeheer) or if such income and gains are derived from the holding, whether directly or indirectly, of (a combination of) shares, debt claims or other rights (together, a “lucrative interest” (lucratief belang)) that the holder thereof has acquired under such circumstances that such income and gains are intended to be remuneration for work or services performed by such holder (or a related person), in whole or in part, in the Netherlands, whether within or outside an employment relation, where such lucrative interest provides the holder thereof, economically speaking, with certain benefits that have a relation to the relevant work or services. If either of the above mentioned conditions (a) or (b) applies, income derived from the ADSs and the gains realized upon the redemption and/or disposal of the ADSs will in general be subject to Dutch income tax at the progressive rates up to 49.5%. Holders of the ADSs Resident Outside the Netherlands: Legal and Other Entities A holder of the ADSs, that is not an individual and that is not resident or deemed to be resident in the Netherlands for corporate income tax purposes, will not be subject to any Dutch taxes on income derived from the ADSs and the gains realized upon the redemption and/or disposal of the ADSs, unless: • such holder has an enterprise or an interest in an enterprise that is, in whole or in part, carried on through a permanent establishment (vaste inrichting) or a permanent representative (vaste vertegenwoordiger) in the Netherlands and to which enterprise or part of an enterprise, as the case may be, the ADSs are attributable; or argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Taxation 200

• such holder has a substantial interest (aanmerkelijk belang) in the Company, that (i) is held for the avoidance of Dutch income tax of another person as (one of) the main purpose(s) and (ii) forms part of an artificial structure or series of structures (such as structures which are not put into place for valid business reasons reflecting economic reality). If either of the above mentioned conditions applies, income derived from the ADSs and the gains realized upon the acquisition, redemption and/or disposal of the ADSs will, in general, be subject to Dutch regular corporate income tax, levied at a rate of 25.8% (19% over profits up to and including €200,000), unless, and to the extent that, with respect to a holder as described under (a), the participation exemption (deelnemingsvrijstelling) applies. Gift, Estate and Inheritance Taxes Holders of the ADSs Resident in the Netherlands Gift tax may be due in the Netherlands with respect to an acquisition of the ADSs by way of a gift by a holder of the ADSs who is resident or deemed to be resident of the Netherlands at the time of the gift. Inheritance tax may be due in the Netherlands with respect to an acquisition or deemed acquisition of the ADSs by way of an inheritance or bequest on the death of a holder of the ADSs who is resident or deemed to be resident of the Netherlands, or in case of a gift by an individual who at the date of the gift was neither resident nor deemed to be resident in the Netherlands, such individual dies within 180 days after the date of the gift, while that individual, at the time of the individual’s death, is resident or deemed to be resident in the Netherlands. For purposes of Dutch gift and inheritance tax, an individual with the Dutch nationality will be deemed to be resident in the Netherlands if such individual has been resident in the Netherlands at any time during the 10 years preceding the date of the gift or such individual’s death. For purposes of Dutch gift tax, an individual not holding the Dutch nationality will be deemed to be resident of the Netherlands if such individual has been resident in the Netherlands at any time during the 12 months preceding the date of the gift. Holders of the ADSs Resident Outside the Netherlands No gift, estate or inheritance taxes will arise in the Netherlands with respect to an acquisition of the ADSs by way of a gift by, or on the death of, a holder of the ADSs who is neither resident nor deemed to be resident of the Netherlands, unless, in the case of a gift of the ADSs by an individual who at the date of the gift was neither resident nor deemed to be resident in the Netherlands, such individual dies within 180 days after the date of the gift, while being resident or deemed to be resident in the Netherlands. Certain Special Situations For purposes of Dutch gift, estate and inheritance tax, (i) a gift by a third party will be construed as a gift by the settlor, and (ii) upon the death of the settlor, as a rule such settlor’s beneficiaries will be deemed to have inherited directly from the settlor. Subsequently, such beneficiaries will be deemed the settlor, grantor or similar originator of the separated private assets for purposes of the Dutch gift, estate and inheritance tax in case of subsequent gifts or inheritances. For the purposes of the Dutch gift and inheritance tax, a gift that is made under a condition precedent is deemed to have been made at the moment such condition precedent is satisfied. If the condition precedent is fulfilled after the death of the donor, the gift is deemed to be made upon the death of the donor. Value Added Tax No Dutch value added tax will arise in respect of or in connection with the subscription, issue, placement, allotment or delivery of the ADSs. Other Taxes and Duties No Dutch registration tax, capital tax, customs duty, transfer tax, stamp duty or any other similar documentary tax or duty, other than court fees, will be payable in the Netherlands in respect of or in connection with the subscription, issue, placement, allotment or delivery of the ADSs. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Taxation 201

Residency A holder of the ADSs will not be treated as a resident, or a deemed resident, of the Netherlands for tax purposes by reason only of the acquisition, or the holding, of the ADSs or the performance by the Company under the ADSs. 5.15.3 Material Belgian Tax Consequences The paragraphs below present a summary of certain Belgian federal income tax consequences of the ownership and disposal of ADSs by an investor. This summary does not describe the tax treatment of investors that are subject to special rules, such as banks, insurance companies, collective investment undertakings, dealers in securities or currencies, persons that hold, or will hold, ADSs as a position in a straddle, share-repurchase transaction, conversion transactions, synthetic security or other integrated financial transactions. The summary is based on laws, treaties and regulatory interpretations in effect in Belgium on the date of this Annual Report, all of which are subject to change, including changes that could have retroactive effect. Investors should appreciate that, as a result of evolutions in law or practice, the eventual tax consequences may be different from what is stated below. For the purposes of this summary, a resident investor is: • an individual subject to Belgian personal income tax (personenbelasting/impôt des personnes physiques), i.e., (i) an individual having its domicile in Belgium, (ii) when not having its domicile in Belgium, an individual having its seat of wealth in Belgium, or (iii) an individual assimilated to a resident for purposes of Belgian tax law; • a company subject to Belgian corporate income tax (vennootschapsbelasting/impôt des sociétés), i.e., a corporate entity having its principal establishment, administrative seat or effective place of management in Belgium (and that is not excluded from the scope of the Belgian corporate income tax); or • a legal entity subject to the Belgian tax on legal entities (rechtspersonenbelasting/impôt des personnes morales), i.e., a legal entity other than a company subject to Belgian corporate income tax having its principal establishment, administrative seat or effective place of management in Belgium. A non-resident investor is any person that is not a Belgian resident investor. Investors should consult their own advisors regarding the tax consequences of an investment in the ADSs in light of their particular situation, including the effect of any state, local or other national laws, treaties and regulatory interpretations thereof. Dividends For Belgian income tax purposes, the gross amount of all benefits paid on or attributed to the ADSs is generally treated as a dividend distribution. By way of exception, the repayment of capital carried out in accordance with applicable Dutch company law provisions is not treated as a dividend distribution to the extent that such repayment is imputed on fiscal capital. This fiscal capital includes, in principle, the actual paid-up statutory share capital and, subject to certain conditions, the paid-up share premiums and the cash amounts subscribed to at the time of the issue of profit-sharing certificates. However, a repayment of capital is not fully imputed on fiscal capital if the company also has certain reserves. Indeed, in such case, a reimbursement of capital is proratedly imputed on, on the one hand, fiscal capital and, on the other hand, taxed reserves (whether or not incorporated in capital) and tax-exempt reserves incorporated in capital (according to a specific priority rule). The part imputed on the reserves is treated as a dividend distribution subject to applicable tax rules. In general, a Belgian withholding tax of (currently) 30% is normally levied on dividends by any intermediary established in Belgium that is in any way involved in the processing of the payment of non-Belgian sourced dividends (e.g., a Belgian financial institution). For this purpose, “dividends” also include the price paid in case of a redemption of ADSs (after deduction of the part of the fiscal capital represented by the redeemed ADSs) and, in the event of our liquidation, any amounts distributed in excess of the fiscal capital. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Taxation 202

However, no withholding tax will be triggered in case of a redemption which is carried out on a stock exchange and meets certain conditions. Further, the withholding tax rate is subject to such relief as may be available under applicable domestic or tax treaty provisions. Under Belgian law, non-Belgian dividend withholding tax is not creditable against Belgian income tax and is not reimbursable to the extent that it exceeds Belgian income tax. Please refer to Section 5.15 “Taxation— Material Dutch Tax Consequences—Dividend Withholding Tax” for a description of withholding tax that may be imposed on dividends by the Netherlands. Belgian Resident Individuals For Belgian resident individuals who acquire and hold ADSs as a private investment, the Belgian dividend withholding tax fully discharges their personal income tax liability. If (and only if) the dividend income would be declared in the personal income tax return, it will be taxed at the lower of the generally applicable 30% Belgian withholding tax rate on dividends or, in case globalization is more advantageous, at the progressive personal income tax rates applicable to the taxpayer’s overall declared income. The first €833 (for income year 2026) (amount applicable per year and per taxpayer) of the reported ordinary dividend income will be exempt from tax, subject to certain conditions. For the avoidance of doubt, all reported dividends (not only dividends distributed on our ADSs) are taken into account to assess whether the said maximum amount is reached. If the dividends are reported, the Belgian dividend withholding tax levied at source may be credited against the personal income tax due and is reimbursable to the extent that it exceeds the personal income tax due, provided that the dividend distribution does not result in a reduction in value of or a capital loss on our ADSs. The latter condition is not applicable if the individual can demonstrate that it has held ADSs in full legal ownership for an uninterrupted period of 12 months prior to the payment or attribution of the dividends. Belgian resident individual investors who acquire and hold the ADSs for professional purposes must always declare the dividend income in their personal income tax return and will be taxable at the investor’s personal income tax rate increased with local surcharges. Belgian withholding tax levied may be credited against the personal income tax due and is reimbursable to the extent that it exceeds the income tax due, subject to two conditions: (i) the taxpayer must own the ADSs in full legal ownership on the dividend record date and (ii) the dividend distribution may not result in a reduction in value of or a capital loss on the ADSs. The latter condition is not applicable if the investor can demonstrate that it has held the full legal ownership of the ADSs for an uninterrupted period of 12 months prior to the payment or attribution of the dividends. Belgian Resident Companies Dividends received by Belgian resident companies are exempt from Belgian withholding tax provided that the investor satisfies the identification requirements in Article 117, §11 of the Royal Decree implementing the Belgian Income Tax Code (the BITC). For Belgian resident companies, the gross dividend income (after deduction of any non-Belgian withholding tax but including any Belgian withholding tax) must be declared in the corporate income tax return and will be subject to a corporate income tax rate of 25%. Subject to certain conditions, a reduced corporate income tax rate of 20% applies on the first €100,000 of taxable profits if the shareholder qualifies as a small company (kleine vennootschap), being a company that, on the balance sheet date of the last completed financial year, has not exceeded more than one of the following thresholds on a consolidated basis for two consecutive financial years: (i) an average annual number of 50 employees, (ii) an annual turnover excluding VAT of EUR 11,250,000 and (iii) a balance sheet total of €6,000,000 (cf. Article 2, §1,5°, c)bis of the BITC in conjunction with Article 1:24, §1 to §6 of the Belgian Code on Companies and Associations (BCCA) (a Small Company)). As a general rule, Belgian resident companies may generally (although subject to certain limitations) deduct 100% of the gross dividend received from their taxable income (Dividend Received Deduction) provided that at the time of a dividend payment or attribution: argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Taxation 203

i. the Belgian resident company holds (A) ADSs representing at least 10% of our share capital or (B) a participation with an acquisition value of at least €2,500,000 which (unless the shareholder is a Small Company) qualifies as “fixed financial asset” (financiële vaste activa). The condition relating to the qualification as “fixed financial asset” applies as of assessment year 2026; ii. the ADSs representing our share capital have been or will be held in full ownership for an uninterrupted period of at least one year; and iii. the conditions described in Article 203 of the BITC (relating to the taxation of the underlying distributed income and the absence of abuse), are met, (together, the Conditions for the Application of the Dividend Received Deduction Regime). Conditions (i) and (ii) above are, in principle, not applicable for dividends received by an investment company in the meaning of Article 2, §1, 5°, f) BITC. The Conditions for the Application of the Dividend Received Deduction Regime depend on a factual analysis and for this reason the availability of this regime should be verified upon each dividend distribution. Any Belgian dividend withholding tax levied at source can be credited against the ordinary Belgian corporate income tax and is reimbursable to the extent it exceeds such corporate income tax, subject to two conditions: (i) the taxpayer must own the ADSs in full legal ownership on the dividend record date and (ii) the dividend distribution does not result in a reduction in value of or a capital loss on the ADSs. The latter condition is not applicable: (i) if the taxpayer demonstrates that it has held the ADSs in full legal ownership for an uninterrupted period of 12 months immediately prior to the payment or attribution of the dividends or (ii) if, during that period, the ADSs never belonged to a taxpayer other than a Belgian resident company or a non-resident company that has, in an uninterrupted manner, invested the ADSs in a PE in Belgium. Belgian resident Organizations for Financing Pensions For organizations for financing pensions (OFPs) i.e., Belgian pension funds incorporated under the form of an OFP (organisme voor de financiering van pensioenen/organisme de financement de pensions) within the meaning of Article 8 of the Belgian Law of October 27, 2006, dividend income is generally tax exempt. Subject to certain limitations, any Belgian dividend withholding tax levied at source may be credited against the corporate income tax due and is reimbursable to the extent that it exceeds the corporate income tax due. Belgian (or foreign) OFPs not holding the ADSs for an uninterrupted period of 60 days in full ownership results in a rebuttable presumption that the arrangement (or a series of arrangements) is not genuine (kunstmatig/pas authentique) and has been put in place for the main purpose or one of the main purposes of obtaining this withholding tax credit. The withholding tax exemption will in such case not apply and/or any Belgian dividend withholding tax levied at source on the dividends will in such case not be credited against the corporate income tax, unless counterproof is provided that the arrangement or series of arrangements are genuine. Other Belgian resident Taxable Legal Entities For taxpayers subject to the Belgian income tax on legal entities, the Belgian dividend withholding tax in principle fully discharges their income tax liability. If the dividend is paid outside Belgium without the intervention of a Belgian paying agent and without the deduction of Belgian withholding tax, the legal entity is in principle required to declare and pay the 30% withholding tax to the Belgian tax authorities. Belgian Non-Resident Individuals and Companies Dividend payments on the ADSs through a professional intermediary in Belgium will, in principle, be subject to the 30% withholding tax, unless the shareholder is resident in a country with which Belgium has concluded a double taxation agreement and delivers the requested affidavit. Non-resident investors can also obtain an exemption of Belgian dividend withholding tax if they are the owners or usufructors of the ADSs and they deliver an affidavit confirming that they have not allocated the ADSs to business activities in Belgium and that they are non-residents, provided that the dividend is paid through a Belgian credit institution, stock market company or recognized clearing or settlement institution. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Taxation 204

If the ADSs are acquired by a non-resident investor in connection with a business in Belgium, the investor must report any dividends received, which are taxable at the applicable non-resident individual or corporate income tax rate, as appropriate. Any Belgian withholding tax levied at source may be credited against the non-resident individual or corporate income tax and is reimbursable to the extent it exceeds the income tax due, subject to two conditions: (i) the taxpayer must own the ADSs in full legal ownership on the dividend record date and (ii) the dividend distribution does not result in a reduction in value of or a capital loss on the ADSs. The latter condition is not applicable if (i) the non-resident individual or the non-resident company demonstrates that the ADSs were held in full legal ownership for an uninterrupted period of 12 months immediately prior to the payment or attribution of the dividends or (ii) with regard to non-resident companies only, if, during the said period, the ADSs have not belonged to a taxpayer other than a resident company or a non-resident company which has, in an uninterrupted manner, invested the ADSs in a Belgian PE. Non-resident companies that have invested the ADSs in a Belgian establishment may deduct up to 100% of the gross dividends included in their taxable profits if, at the date dividends are paid or attributed, the Conditions for Application of the Dividend Received Deduction Regime are satisfied. Application of the Dividend Received Deduction depends, however, on a factual analysis to be made upon each distribution and its availability should be verified upon each distribution. Capital Gains and Losses on ADSs Belgian Resident Individuals Under the current legislation, Belgian resident individuals acquiring the ADSs as a private investment should not be subject to Belgian capital gains tax on the disposal of the ADSs; capital losses are not tax deductible. However, the Belgian federal government has agreed to introduce a capital gains tax on financial assets (such as the ADSs) for capital gains realized as from January 1, 2026 (the New Capital Gains Tax). This New Capital Gains Tax would only apply to capital gains accrued as from January 1, 2026 (i.e. historical capital gains accrued until December 31, 2025 would not be subject to the tax). The New Capital Gains Tax must be adopted by the Belgian parliament prior to coming into effect. It is expected that the New Capital Gains Tax will be adopted in the coming months and, once adopted, it will apply to capital gains realized as from January 1, 2026. Based on the draft texts currently available, the New Capital Gains Tax would comprise the following three categories of capital gains: • “Internal” capital gains: capital gains on ADSs sold to a transferee over which the transferor, either individually or together with his spouse or descendants, ascendants, collateral relatives up to and including the second degree and those of his spouse, exercises direct or indirect control as defined in article 1:14 of the BCCA. Such “internal” capital gains would be subject to a 33% tax rate; • “Significant Stake” capital gains: capital gains on ADSs realized within the ‘normal management of private estate’ if the transferor, at the moment of the transfer, holds a participation of at least 20% in the company whose shares are being transferred (a Significant Stake). The first €1,000,000 of capital gain on a Significant Stake would be exempt (such exempt tranche being available per period of five years). The capital gain exceeding €1,000,000 would be subject to progressive rates: 1.25% on capital gains between €1,000,000 and €2,500,000; 2.5% on capital gains between €2,500,000 and EUR 5,000,000; 5% on capital gains between €5,000,000 and €10,000,000 and 10% on capital gains exceeding €10,000,000; • “Other” capital gains: capital gains on financial assets (such as the ADSs) realized within the ‘normal management of private estate’, other than “internal” capital gains or capital gains on a “Significant Stake” (as described above). The applicable rate would be 10%. An annual exemption will be provided for up to €10,000, which may be increased by up to €1,000 for each year in which the exemption is not (fully) used, up to a maximum of €15,000 after five years (amounts to be indexed). Based on the draft texts currently available, it is intended that the tax on ‘other’ capital gains would generally be levied via withholding tax (subject to certain exceptions). argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Taxation 205

Capital losses on the disposal of the ADSs would be deductible from capital gains realized in the same taxable year, by the same taxpayer and within the same “category” of taxable capital gains on financial assets (as described above) However, capital gains which are deemed to be realized outside the scope of the ‘normal management of the individual’s private estate’ are taxable at 33% (plus local surcharges). Under the current legislation, Belgian resident individuals who hold the ADSs for professional purposes are taxable at the ordinary progressive personal income tax rates (plus local surcharges) on any capital gains realized upon the disposal of the ADSs, except for shares held for more than five years, which are taxable at a separate rate of 16.5% or (if the capital gain is realized in the framework of the cessation of activities and under certain circumstances) 10% (in each case plus local surcharges). Capital losses on the ADSs incurred by Belgian resident individuals who hold the ADSs for professional purposes are in principle tax deductible. Based on the draft texts currently available, the application of the New Capital Gains Tax could be triggered if Belgian resident individuals transfer their place of residence or seat of wealth outside of Belgium, subject to conditions and exemptions. Belgian Resident Companies Belgian resident companies are normally not subject to Belgian capital gains taxation on gains realized upon the disposal of our ADSs provided that the Conditions for the Application of the Dividend Received Deduction Regime (see above under “Dividends—Belgian Resident Companies”) are met. If one of the Conditions for Dividend Received Deduction is not met, the capital gains realized upon the disposal of our ADSs by a Belgian resident company are taxable at the ordinary corporate income tax rate of, currently, 25%, unless the reduced corporate income tax rate of 20% on the first €100,000 of taxable profits applies (see above). Capital losses on our ADSs incurred by resident companies are as a general rule not tax deductible. Our ADSs held in the trading portfolios (handelsportefeuille/portefeuille commercial) of qualifying credit institutions, investment enterprises and management companies of collective investment undertakings which are subject to the Royal Decree of 23 September 1992 on the annual accounts of credit institutions, investment firms and management companies of collective investment undertakings (Koninklijk besluit van 23 september 1992 op de jaarrekening van de kredietinstellingen, de beleggingsondernemingen en de beheervennootschappen van instellingen voor collectieve belegging/ arrêté royal du 23 septembre 1992 relatif aux comptes annuels des établissements de crédit, des entreprises d’investissement et des sociétés de gestion d’organismes de placement collectif) are subject to a different regime. The capital gains on such shares are taxable at the ordinary corporate income tax rate of 25%. Capital losses on such shares are tax deductible. Internal transfers to and from the trading portfolio are assimilated to a realization. Capital gains realized by Belgian resident companies (both ordinary Belgian resident companies and qualifying credit institutions, investment enterprises and management companies of collective investment undertakings) upon the redemption of our ADSs or upon our liquidation are, in principle, subject to the same taxation regime as dividends. Refer to Section 5.15 “Taxation — Dividends”. Belgian resident OFPs OFPs are, in principle, not subject to Belgian capital gains taxation realized upon the disposal of the ADSs, and capital losses are not tax deductible. Capital gains realized by Belgian OFPs upon the redemption of ADSs or upon our liquidation will in principle be taxed as dividends. Other Belgian Taxable Legal Entities Under the current legislation, Belgian resident legal entities subject to the legal entities income tax are, in principle, not subject to Belgian capital gains taxation on the disposal of ADSs. Capital losses on ADSs incurred by Belgian resident legal entities are not tax deductible. However, the Belgian government has agreed to introduce a New Capital Gains Tax on financial assets (such as the ADSs) from January 1, 2026 onwards (see “Belgian Resident Individuals” above). Based on the argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Taxation 206

draft texts currently available, this capital gains tax will also be due by legal entities subject to Belgian legal entities tax, except entities that are entitled to receive tax-deductible gifts. Capital gains realized by Belgian resident legal entities upon the redemption of ADSs or upon our liquidation will in principle be taxed as dividends. Belgian Non-Resident Individuals and Companies Non-resident individuals or companies are, in principle, not subject to Belgian income tax on capital gains realized upon disposal of the ADSs, unless such ADSs are held as part of a business conducted in Belgium through a Belgian establishment. In such a case, the same principles apply as described with regard to Belgian individuals (holding the shares for professional purposes) or Belgian companies. Non-resident individuals who do not use the shares for professional purposes and who have their fiscal residence in a country with which Belgium has not concluded a tax treaty or with which Belgium has concluded a tax treaty that confers the authority to tax capital gains on the ADSs to Belgium, might be subject to tax in Belgium if the capital gains are obtained or received in Belgium and arise from transactions which are to be considered speculative or beyond the normal management of one’s private estate. Refer to Section “Taxation— Capital Gains and Losses on ADSs - Belgian Resident Individuals". Such non-resident individuals might therefore be obliged to file a tax return and should consult their own tax advisor. However, Belgium has concluded tax treaties with more than 95 countries which generally provide for a full exemption from Belgian capital gains taxation on such gains realized by residents of those countries. However, the draft texts relating to the New Capital Gains Tax provide for the repeal of the provisions of the BITC that allow capital gains realized by Belgian non-residents who do not hold the ADSs for professional purposes to be subject to tax in Belgium. As a result, capital gains realized by Belgian non-residents outside the scope of a professional activity could no longer be taxable in Belgium. Capital gains realized by non-resident individuals or non-resident companies upon the redemption of ADSs or upon our liquidation will, in principle, be subject to the same taxation regime as dividends. Tax on Stock Exchange Transactions Upon the issue of the ADSs (primary market), no Tax on Stock Exchange Transactions (taks op beursverrichtingen/taxe sur opérations de bourse) is due. The purchase and the sale and any other acquisition or transfer for consideration of ADSs (secondary market transactions) is subject to the Tax on Stock Exchange Transactions if (i) it is executed in Belgium through a professional intermediary, or (ii) deemed to be executed in Belgium, which is the case if the order is directly or indirectly made to a professional intermediary established outside of Belgium, either by private individuals with habitual residence in Belgium, or legal entities for the account of their seat or establishment in Belgium (both, a Belgian Investor). The Tax on Stock Exchange Transactions is levied at a rate of 0.35% of the purchase price, capped at €1,600 per transaction and per party. A separate tax is due by each party to the transaction, and both taxes are collected by the professional intermediary. However, if the intermediary is established outside of Belgium, the tax will in principle be due by the Belgian Investor, unless that Belgian Investor can demonstrate that the tax has already been paid. Professional intermediaries established outside of Belgium can, subject to certain conditions and formalities, appoint a Belgian Stock Exchange Tax Representative, which will be liable for the Tax on Stock Exchange Transactions in respect of the transactions executed through the professional intermediary. If the Stock Exchange Tax Representative would have paid the Tax on Stock Exchange Transactions due, the Belgian Investor will, as per the above, no longer be the debtor of the Tax on Stock Exchange Transactions. No Tax on Stock Exchange Transactions is due on transactions entered into by the following parties, provided they are acting for their own account: (i) professional intermediaries described in Article 2, 9° and 10° of the Belgian Law of August 2, 2002; (ii) insurance companies described in Article 2, §1 of the Belgian Law of July 9, 1975; (iii) professional retirement institutions referred to in Article 2, 1° of the Belgian Law of October 27, 2006 concerning the supervision on institutions for occupational pension; (iv) collective investment institutions; (v) regulated real estate companies; and (vi) Belgian non-residents provided they deliver a certificate to their financial intermediary in Belgium confirming their non-resident status. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Taxation 207

Annual Tax on Securities Accounts The Belgian Annual Tax on Securities Accounts is a subscription tax, levied on securities accounts and not on the holders thereof. A securities account is defined as an account on which financial instruments can be credited and debited. The tax applies to securities accounts held both in Belgium and abroad when the account holder is a Belgian resident or when the account forms part of the assets of a Belgian establishment of a non-Belgian resident. The tax applies to natural persons residing in Belgium, as well as to companies and legal entities (subject to the tax for legal entities) that are established in Belgium. The tax is also applicable to securities accounts held by non-Belgian residents (both natural persons and legal persons) if the securities account is held in Belgium. If the applicable double tax treaty however allocates the right to tax capital to the jurisdiction of residence, Belgium would be prevented from applying the Annual Tax on Securities Accounts to the Belgian securities accounts held by non-Belgian residents. As described above, the tax applies whether or not the account is held in Belgium if the account forms part of the assets of a Belgian establishment of a non-Belgian resident. The Annual Tax on Securities Accounts is applicable to securities accounts of which the average value of the assets amounts to more than €1,000,000 during the reference period. In principle, this reference period starts on 1 October and ends on 30 September of the following year. The aforementioned threshold is assessed on the average value of the assets in the securities account at reference points within the reference period (in principle December 31st, March 31st, June 30th and September 30th). The threshold is assessed per securities account and not per account holder. The applicable tax rate is 0.15%, which is levied on the average value of the assets held in the securities account that exceeds the €1,000,000 threshold. It is however limited to 10% of the difference between the average value and the threshold of €1,000,000, in order to avoid that the Annual Tax on Securities Accounts would result in reducing the value of the securities account below the €1,000,000 threshold. However, as part of the federal budget agreement, the Belgian government announced that the applicable tax rate would be increased from 0.15% to 0.30%. This change should nevertheless first be adopted by the Belgian parliament prior to coming into effect. The Annual Tax is in principle withheld, reported and paid by the Belgian intermediary. If the intermediary is established outside of Belgium, the tax must in principle be reported and paid by the account holder, unless the account holder can demonstrate that the tax has already been reported and paid by an intermediary. Intermediaries established outside of Belgium can, subject to certain conditions and formalities, appoint a Belgian Annual Tax on Securities Accounts Representative, which will be liable for reporting and paying the Annual Tax on Securities Accounts in respect of securities accounts in scope of the Annual Tax that are held through such intermediaries. If the Annual Tax on Securities Accounts Representative would have paid the Annual Tax on Securities Accounts due, the account holder will, as per the above, no longer be the debtor of the Annual Tax on Securities Accounts. The Annual Tax on Securities Accounts is however not applicable to securities accounts held by certain categories of account holders active in the financial or fund sector, as listed in the relevant legislation (e.g. credit institutions, insurance companies, investment companies, and certain collective investment undertakings). These exemptions do however not apply if a non-qualifying third party has a direct or indirect claim on the value of the securities account. Applicable as of July 29, 2025, a new specific anti-abuse rule (SAAR) in relation to the annual tax on securities accounts was introduced. The SAAR introduces a presumption of abuse in case of (i) a conversion of financial instruments registered in a securities account into similar instruments that are not registered in such an account (e.g. dematerialized securities into registered securities), if before the conversion the total value of the taxable financial instruments in the account exceeded €1,000,000, and (ii) a transfer of securities from one securities account to one or more other securities account(s), if before such transfer the total value of taxable instruments in the account exceeded €1,000,000 and provided that (a) the securities account holders of the accounts involved are the same, or (b) the transferring account holder is a joint holder of the receiving account. The application of the SAAR may be refuted if it is demonstrated that the transaction is mainly justified by a motive other than the avoidance of the annual tax on securities accounts. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Taxation 208

The financial intermediary established or located in Belgium or the Belgian annual tax on securities accounts representative must notify the transactions mentioned under (i) and (ii) above to the tax authorities ultimately by the last day of the month following the end of the relevant reference period. If it concerns a foreign securities account for which no Belgian annual tax on securities accounts representative is indicated, the notification should occur by the holder of the securities accounts itself. Prospective investors are strongly advised to seek their own professional advice in relation to the possible impact of the Annual Tax on Securities Accounts on their own personal tax position. 5.15.4 Enforcement of civil liabilities We are a European public company with limited liability (Societas Europaea or SE) incorporated under the laws of the Netherlands. A majority of our assets are located outside the U.S. As a result, it may not be possible or it may be difficult for investors to effect service of process within the U.S. upon such persons or the Group, or to enforce against them or us in U.S. courts, including judgments predicated upon the civil liability provisions of the federal securities laws of the U.S. The U.S. and the Netherlands currently do not have a treaty providing for the reciprocal recognition and enforcement of judgments, other than arbitration awards, in civil and commercial matters. Consequently, a final judgment (for payment) given by a court in the U.S., whether or not predicated solely upon U.S. securities laws, would not automatically be recognized or enforceable in the Netherlands. In order to obtain a judgment which is enforceable in the Netherlands, the party in whose favor a final and conclusive judgment of the U.S. court has been rendered will be required to file its claim with a court of competent jurisdiction in the Netherlands. Such party may submit to the Dutch court the final judgment rendered by the U.S. court. This court will have a level of discretion in its assessment of the judgment rendered by the relevant U.S. court. On the basis of case law by the Dutch Supreme Court, Dutch courts will in principle have to give conclusive effect to a final and enforceable judgment of such court in respect of the contractual obligations thereunder without re-examination or re-litigation of the substantive matters adjudicated upon, provided that: (i) the jurisdiction of the U.S. court has been based on a ground of jurisdiction that is generally acceptable according to international standards, (ii) the judgment by the U.S. court was rendered in legal proceedings that comply with the Dutch standards of the proper administration of justice that includes sufficient safeguards (behoorlijke rechtspleging), (iii) the judgment by the U.S. court does not contravene Dutch public policy (openbare orde), and (iv) the judgment by the U.S. court is not irreconcilable with a judgment of a Dutch court or an earlier judgment of a foreign court rendered between the same parties that is capable of being recognized in the Netherlands. Even if such foreign judgments is given binding effect, a claim based thereon may, however, still be rejected if the foreign judgment is not or no longer formally enforceable in the country of origin. In addition, there can be no assurance that civil liabilities predicated upon the federal or state securities laws of the United States will be enforceable in the Netherlands or any other jurisdiction. Enforcement and recognition of judgments of U.S. courts in the Netherlands are solely governed by the provisions of the Dutch Civil Procedure Code (Wetboek van Burgerlijke Rechtsvordering). Judgments may be rendered in a foreign currency, but enforcement is executed in euro at the applicable rate of exchange. Under certain circumstances, a Dutch court has the power to stay proceedings (aanhouden) or to declare that it has no jurisdiction if concurrent proceedings are being brought elsewhere. Moreover, a Dutch court may reduce the amount of damages granted by a U.S. court and recognize damages only to the extent that they are necessary to compensate actual losses or damages. Original actions or actions for the enforcement of judgments of U.S. courts relating to the civil liability provisions of the federal or state securities laws of the U.S. are not directly enforceable in Belgium. The U.S. and Belgium currently do not have a treaty providing for reciprocal recognition and enforcement of judgments, other than arbitral awards, in civil and commercial matters. Consequently, a final judgment for payment given by a court in the U.S., whether or not predicated solely upon U.S. securities laws, would not automatically be recognized or enforceable in Belgium. In order for a final judgment for the payment of money rendered by U.S. courts based on civil liability to produce any effect on Belgian soil, it is accordingly required that this judgment be recognized and be declared enforceable by a Belgian court pursuant to the relevant provisions of the PIL Code. Recognition or enforcement does not imply a review of the merits of the case and is irrespective of any reciprocity requirement. A U.S. judgment will, however, not be recognized or declared enforceable in Belgium if it infringes upon one or more of the grounds for refusal which are exhaustively listed in article 25 of the PIL Code. In addition to recognition or enforcement, a judgment by a federal or state court in the U.S. against us may also serve as evidence in a similar action in a argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Taxation 209

Belgian court if it meets the conditions required for the authenticity of judgments according to the law of the state where it was rendered. In addition, with regard to enforcements by legal proceedings in Belgium (including the recognition of foreign court decisions in Belgium), a registration tax at the rate of 3% of the amount of the judgment is payable by the debtor, if the sum of money which the debtor is ordered to pay by a Belgian court, or by a foreign court judgment that is either (i) automatically enforceable and registered in Belgium, or (ii) rendered enforceable by a Belgian court, exceeds €12,500. The registration tax is payable by the debtor. The debtor is liable for the payment of the registration tax, in the proportion determined by the decision ordering payment or liquidation or determining priority for creditors made or established against it. The debtor(s) are jointly and severally liable in the event that they are ordered to pay jointly and severally. A stamp duty is payable as of the second certified copy of an enforcement judgment rendered by a Belgian court, with a maximum of €1,450. Dutch and Belgian civil procedure differ substantially from U.S. civil procedure in a number of respects. Insofar as the production of evidence is concerned, U.S. law and the laws of several other jurisdictions based on common law provide for pre-trial discovery, a process by which parties to the proceedings may prior to trial compel the production of documents by adverse or third parties and the deposition of witnesses. Evidence obtained in this manner may be decisive in the outcome of any proceeding. No such pre-trial discovery process exists under Dutch or Belgian law. Subject to the foregoing and service of process in accordance with applicable treaties, investors may be able to enforce in the Netherlands or Belgium judgments in civil and commercial matters obtained from U.S. federal or state courts. However, no assurance can be given that those judgments will be enforceable. In addition, it is doubtful whether a Dutch or Belgian court would accept jurisdiction and impose civil liability in an original action commenced in the Netherlands or Belgium and predicated solely upon U.S. federal securities laws. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Taxation 210

Financial Statements 6.1 Consolidated Financial Statements 212 6.2 Notes to the Consolidated Financial Statements 219 6.3 Company Financial Statements of argenx SE for the Year ended December 31, 2025 255 6.4 Other information 261 argenx Annual Report 2025 211 6
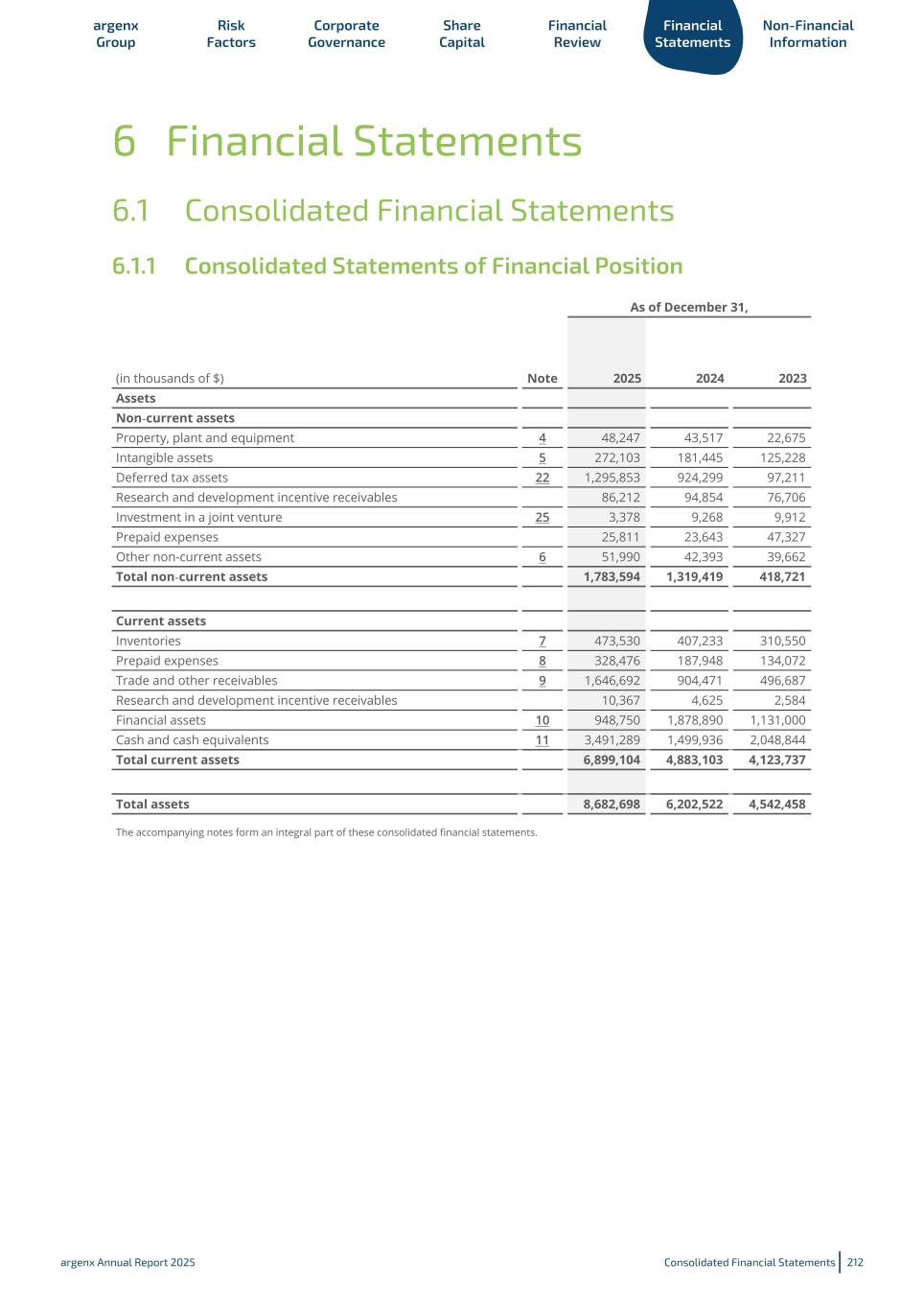
6 Financial Statements 6.1 Consolidated Financial Statements 6.1.1 Consolidated Statements of Financial Position As of December 31, (in thousands of $) Note 2025 2024 2023 Assets Non-current assets Property, plant and equipment 4 48,247 $ 43,517 $ 22,675 Intangible assets 5 272,103 181,445 125,228 Deferred tax assets 22 1,295,853 924,299 97,211 Research and development incentive receivables 86,212 94,854 76,706 Investment in a joint venture 25 3,378 9,268 9,912 Prepaid expenses 25,811 23,643 47,327 Other non-current assets 6 51,990 42,393 39,662 Total non-current assets 1,783,594 1,319,419 418,721 Current assets Inventories 7 473,530 $ 407,233 $ 310,550 Prepaid expenses 8 328,476 187,948 134,072 Trade and other receivables 9 1,646,692 904,471 496,687 Research and development incentive receivables 10,367 4,625 2,584 Financial assets 10 948,750 1,878,890 1,131,000 Cash and cash equivalents 11 3,491,289 1,499,936 2,048,844 Total current assets 6,899,104 4,883,103 4,123,737 Total assets 8,682,698 $ 6,202,522 $ 4,542,458 The accompanying notes form an integral part of these consolidated financial statements. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Consolidated Financial Statements 212
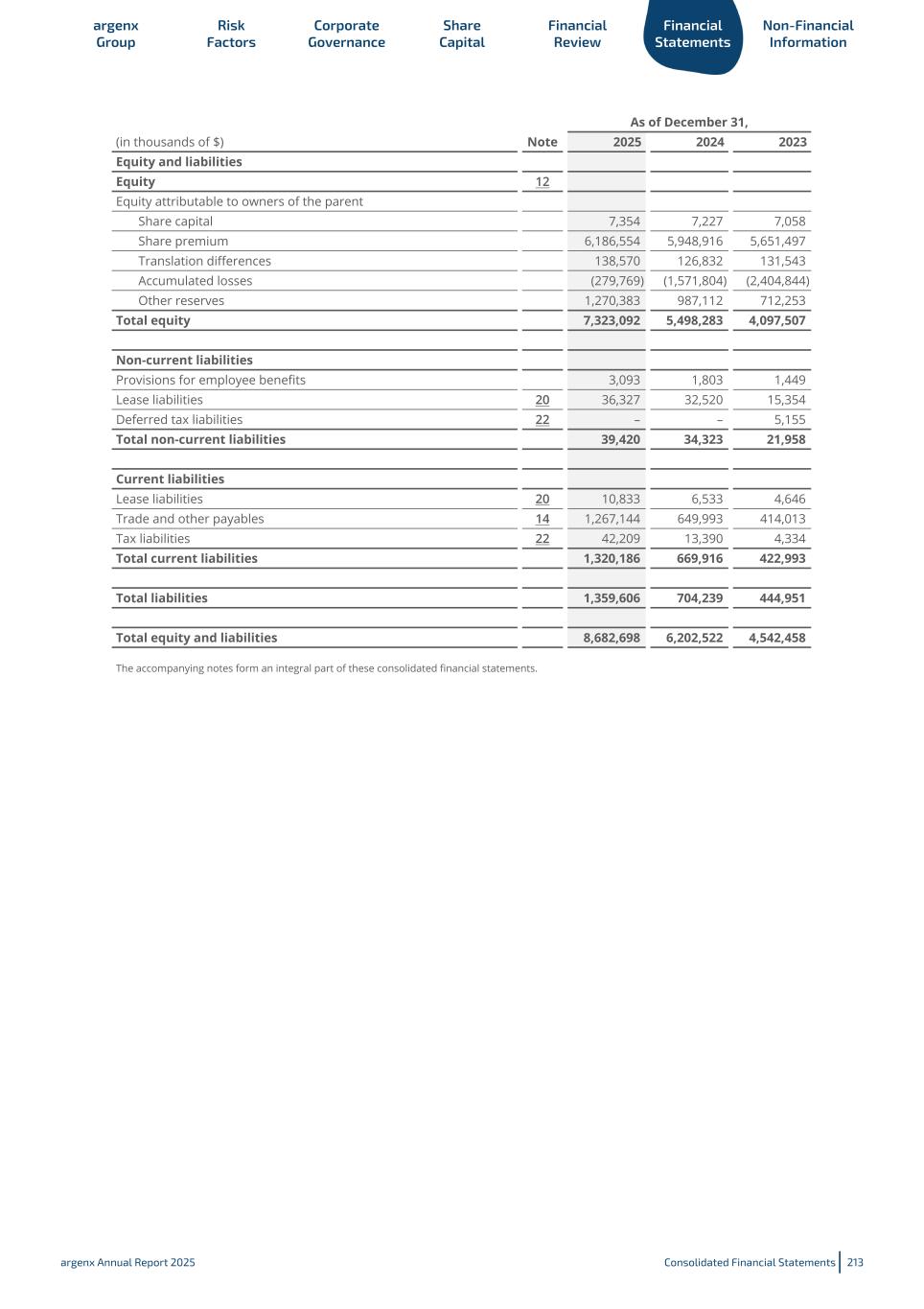
As of December 31, (in thousands of $) Note 2025 2024 2023 Equity and liabilities Equity 12 Equity attributable to owners of the parent Share capital 7,354 7,227 7,058 Share premium 6,186,554 5,948,916 5,651,497 Translation differences 138,570 126,832 131,543 Accumulated losses (279,769) (1,571,804) (2,404,844) Other reserves 1,270,383 987,112 712,253 Total equity 7,323,092 5,498,283 4,097,507 Non-current liabilities Provisions for employee benefits 3,093 1,803 1,449 Lease liabilities 20 36,327 32,520 15,354 Deferred tax liabilities 22 – – 5,155 Total non-current liabilities 39,420 34,323 21,958 Current liabilities Lease liabilities 20 10,833 6,533 4,646 Trade and other payables 14 1,267,144 649,993 414,013 Tax liabilities 22 42,209 13,390 4,334 Total current liabilities 1,320,186 669,916 422,993 Total liabilities 1,359,606 704,239 444,951 Total equity and liabilities 8,682,698 6,202,522 4,542,458 The accompanying notes form an integral part of these consolidated financial statements. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Consolidated Financial Statements 213
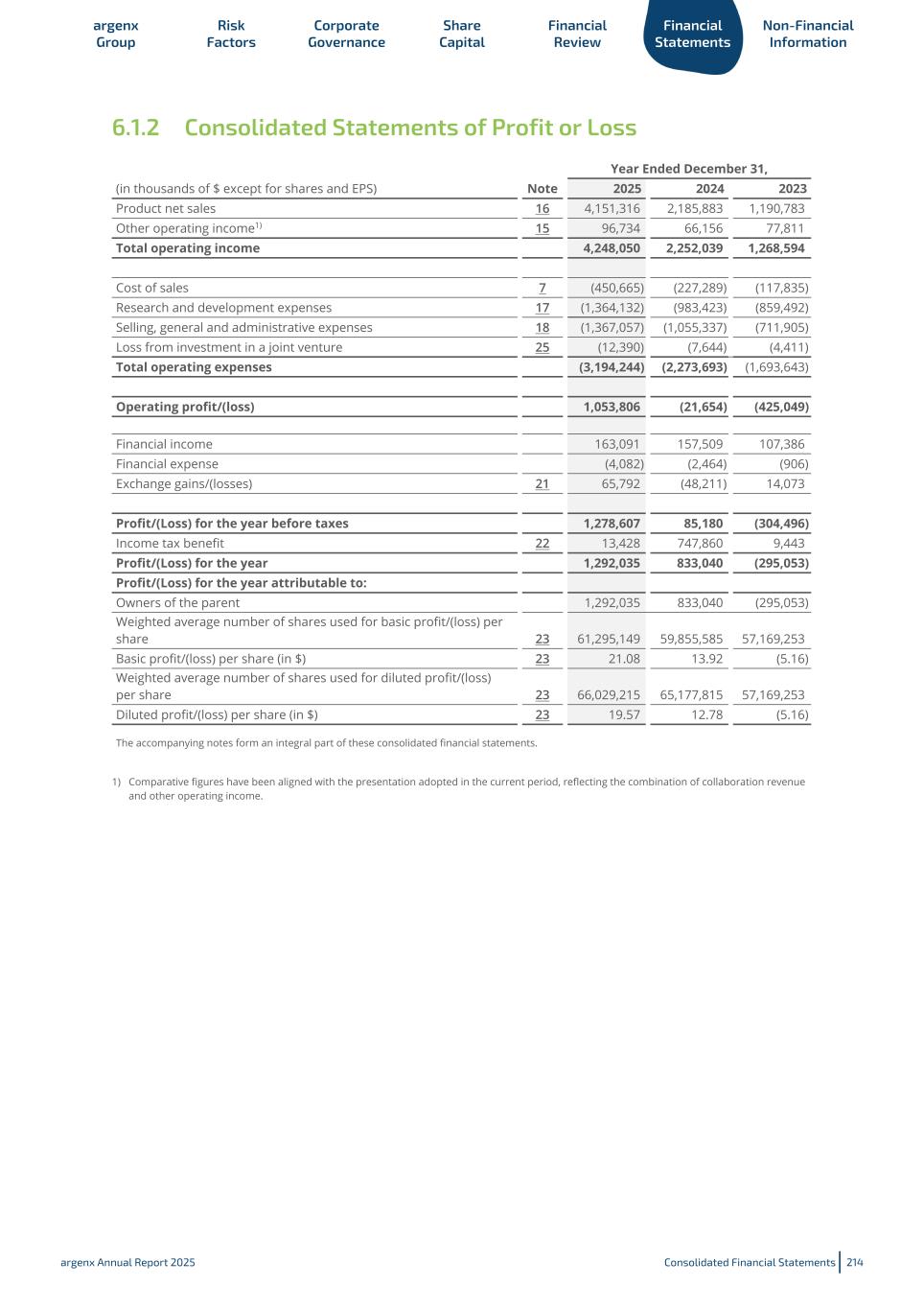
6.1.2 Consolidated Statements of Profit or Loss Year Ended December 31, (in thousands of $ except for shares and EPS) Note 2025 2024 2023 Product net sales 16 4,151,316 2,185,883 1,190,783 Other operating income1) 15 96,734 66,156 77,811 Total operating income 4,248,050 2,252,039 1,268,594 Cost of sales 7 (450,665) (227,289) (117,835) Research and development expenses 17 (1,364,132) (983,423) (859,492) Selling, general and administrative expenses 18 (1,367,057) (1,055,337) (711,905) Loss from investment in a joint venture 25 (12,390) (7,644) (4,411) Total operating expenses (3,194,244) (2,273,693) (1,693,643) Operating profit/(loss) 1,053,806 (21,654) (425,049) Financial income 163,091 157,509 107,386 Financial expense (4,082) (2,464) (906) Exchange gains/(losses) 21 65,792 (48,211) 14,073 Profit/(Loss) for the year before taxes 1,278,607 85,180 (304,496) Income tax benefit 22 13,428 747,860 9,443 Profit/(Loss) for the year 1,292,035 833,040 (295,053) Profit/(Loss) for the year attributable to: Owners of the parent 1,292,035 833,040 (295,053) Weighted average number of shares used for basic profit/(loss) per share 23 61,295,149 59,855,585 57,169,253 Basic profit/(loss) per share (in $) 23 21.08 13.92 (5.16) Weighted average number of shares used for diluted profit/(loss) per share 23 66,029,215 65,177,815 57,169,253 Diluted profit/(loss) per share (in $) 23 19.57 12.78 (5.16) The accompanying notes form an integral part of these consolidated financial statements. 1) Comparative figures have been aligned with the presentation adopted in the current period, reflecting the combination of collaboration revenue and other operating income. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Consolidated Financial Statements 214
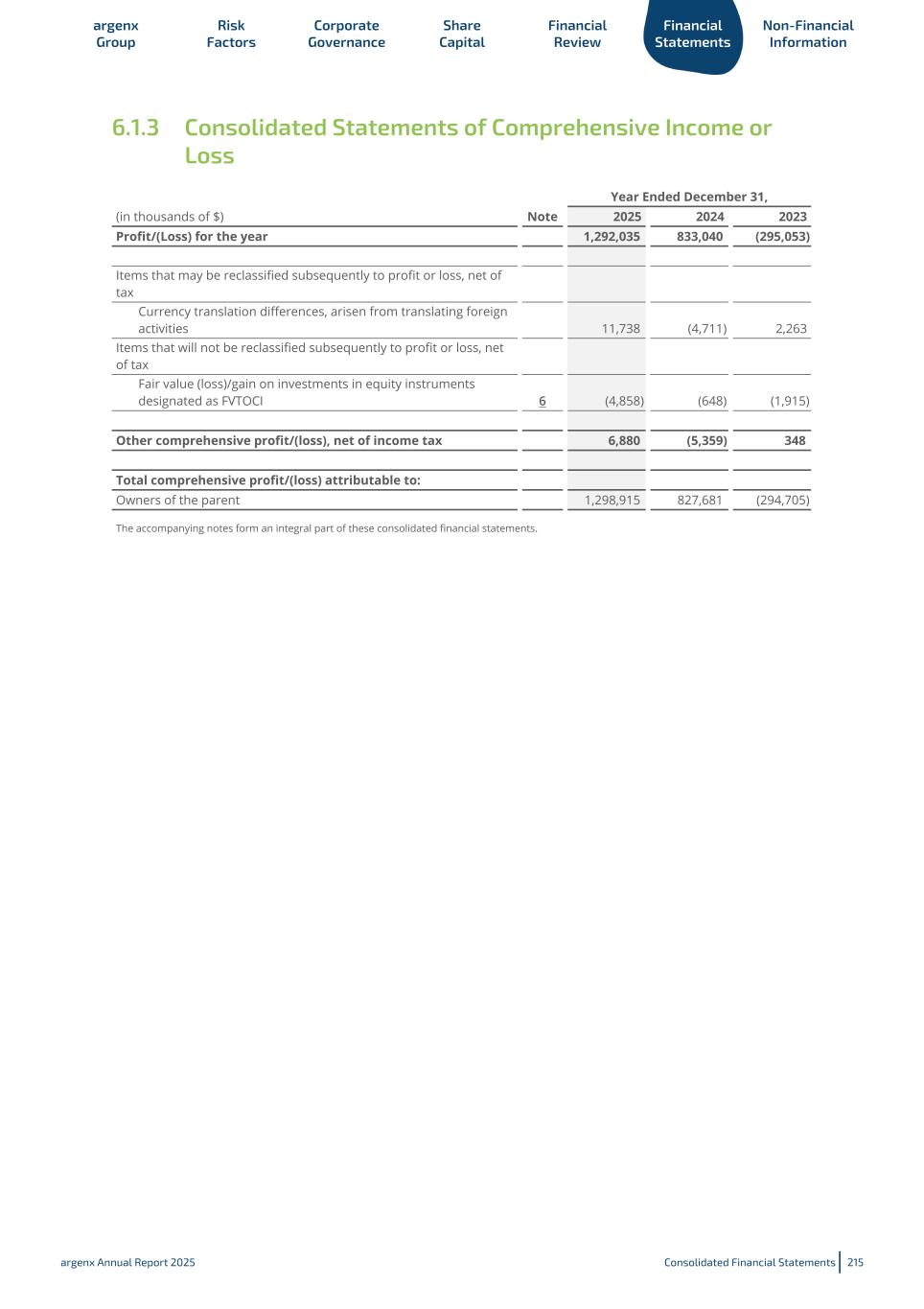
6.1.3 Consolidated Statements of Comprehensive Income or Loss Year Ended December 31, (in thousands of $) Note 2025 2024 2023 Profit/(Loss) for the year 1,292,035 833,040 (295,053) Items that may be reclassified subsequently to profit or loss, net of tax Currency translation differences, arisen from translating foreign activities 11,738 (4,711) 2,263 Items that will not be reclassified subsequently to profit or loss, net of tax Fair value (loss)/gain on investments in equity instruments designated as FVTOCI 6 (4,858) (648) (1,915) Other comprehensive profit/(loss), net of income tax 6,880 (5,359) 348 Total comprehensive profit/(loss) attributable to: Owners of the parent 1,298,915 827,681 (294,705) The accompanying notes form an integral part of these consolidated financial statements. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Consolidated Financial Statements 215
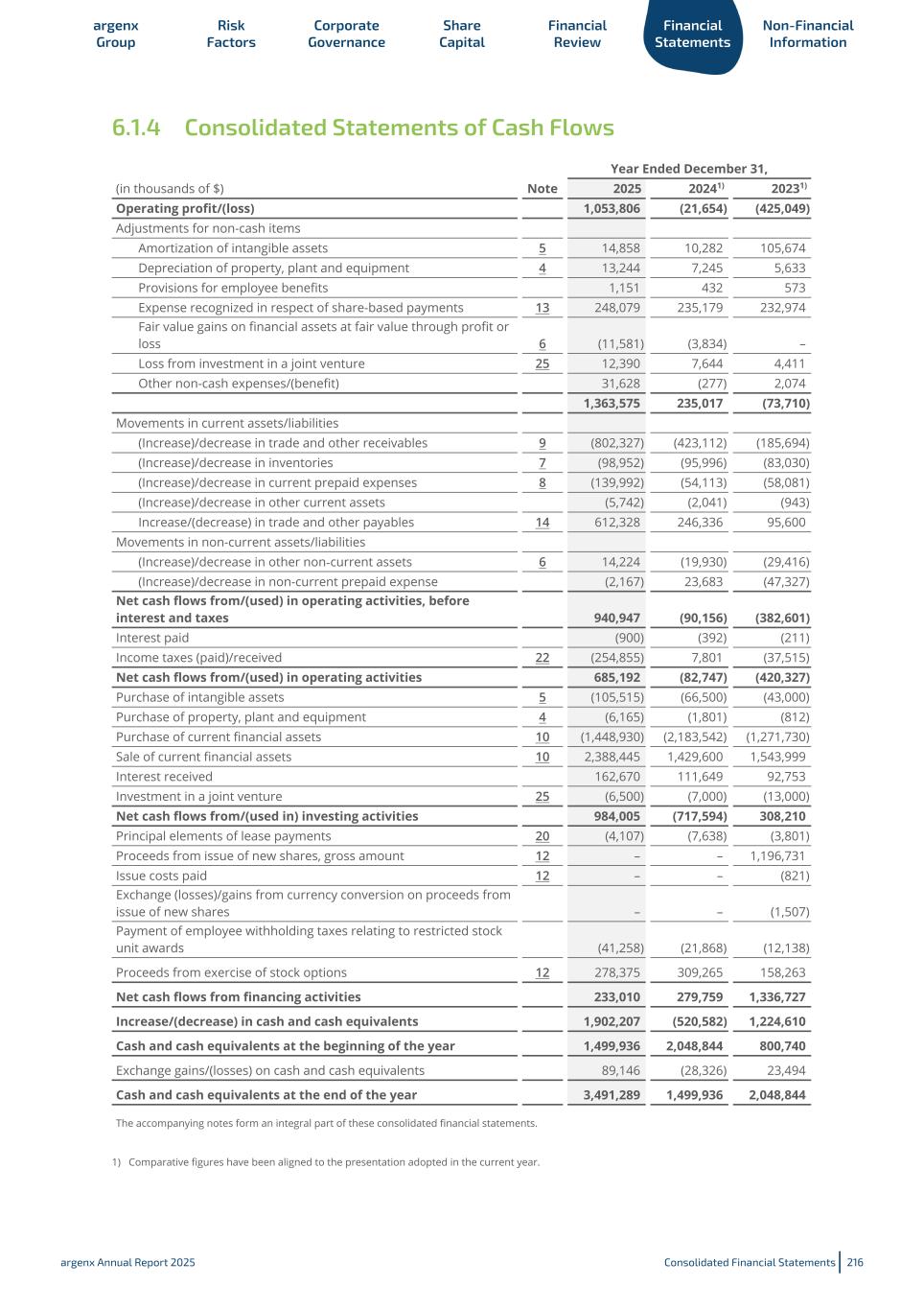
6.1.4 Consolidated Statements of Cash Flows Operating profit/(loss) 1,053,806 (21,654) (425,049) Adjustments for non-cash items Amortization of intangible assets 5 14,858 10,282 105,674 Depreciation of property, plant and equipment 4 13,244 7,245 5,633 Provisions for employee benefits 1,151 432 573 Expense recognized in respect of share-based payments 13 248,079 235,179 232,974 Fair value gains on financial assets at fair value through profit or loss 6 (11,581) (3,834) – Loss from investment in a joint venture 25 12,390 7,644 4,411 Other non-cash expenses/(benefit) 31,628 (277) 2,074 1,363,575 235,017 (73,710) Movements in current assets/liabilities (Increase)/decrease in trade and other receivables 9 (802,327) (423,112) (185,694) (Increase)/decrease in inventories 7 (98,952) (95,996) (83,030) (Increase)/decrease in current prepaid expenses 8 (139,992) (54,113) (58,081) (Increase)/decrease in other current assets (5,742) (2,041) (943) Increase/(decrease) in trade and other payables 14 612,328 246,336 95,600 Movements in non-current assets/liabilities (Increase)/decrease in other non-current assets 6 14,224 (19,930) (29,416) (Increase)/decrease in non-current prepaid expense (2,167) 23,683 (47,327) Net cash flows from/(used) in operating activities, before interest and taxes 940,947 (90,156) (382,601) Interest paid (900) (392) (211) Income taxes (paid)/received 22 (254,855) 7,801 (37,515) Net cash flows from/(used) in operating activities 685,192 (82,747) (420,327) Purchase of intangible assets 5 (105,515) (66,500) (43,000) Purchase of property, plant and equipment 4 (6,165) (1,801) (812) Purchase of current financial assets 10 (1,448,930) (2,183,542) (1,271,730) Sale of current financial assets 10 2,388,445 1,429,600 1,543,999 Interest received 162,670 111,649 92,753 Investment in a joint venture 25 (6,500) (7,000) (13,000) Net cash flows from/(used in) investing activities 984,005 (717,594) 308,210 Principal elements of lease payments 20 (4,107) (7,638) (3,801) Proceeds from issue of new shares, gross amount 12 – – 1,196,731 Issue costs paid 12 – – (821) Exchange (losses)/gains from currency conversion on proceeds from issue of new shares – – (1,507) Payment of employee withholding taxes relating to restricted stock unit awards (41,258) (21,868) (12,138) Proceeds from exercise of stock options 12 278,375 309,265 158,263 Net cash flows from financing activities 233,010 279,759 1,336,727 Increase/(decrease) in cash and cash equivalents 1,902,207 (520,582) 1,224,610 Cash and cash equivalents at the beginning of the year 1,499,936 2,048,844 800,740 Exchange gains/(losses) on cash and cash equivalents 89,146 (28,326) 23,494 Cash and cash equivalents at the end of the year 3,491,289 1,499,936 2,048,844 The accompanying notes form an integral part of these consolidated financial statements. Year Ended December 31, (in thousands of $) Note 2025 20241) 20231) 1) Comparative figures have been aligned to the presentation adopted in the current year. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Consolidated Financial Statements 216
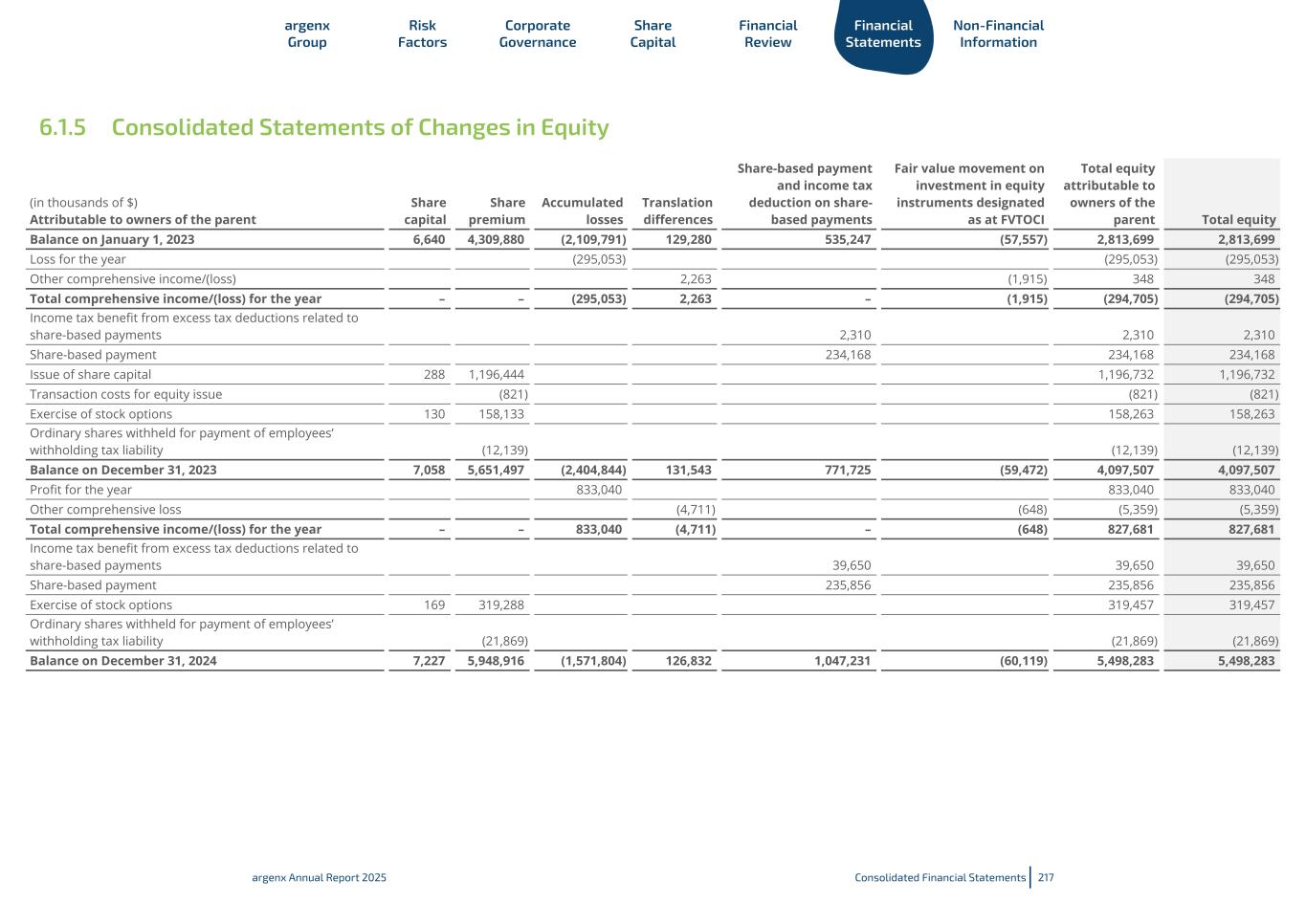
6.1.5 Consolidated Statements of Changes in Equity Balance on January 1, 2023 6,640 4,309,880 (2,109,791) 129,280 535,247 (57,557) 2,813,699 2,813,699 Loss for the year (295,053) (295,053) (295,053) Other comprehensive income/(loss) 2,263 (1,915) 348 348 Total comprehensive income/(loss) for the year – – (295,053) 2,263 – (1,915) (294,705) (294,705) Income tax benefit from excess tax deductions related to share-based payments 2,310 2,310 2,310 Share-based payment 234,168 234,168 234,168 Issue of share capital 288 1,196,444 1,196,732 1,196,732 Transaction costs for equity issue (821) (821) (821) Exercise of stock options 130 158,133 158,263 158,263 Ordinary shares withheld for payment of employees’ withholding tax liability (12,139) (12,139) (12,139) Balance on December 31, 2023 7,058 5,651,497 (2,404,844) 131,543 771,725 (59,472) 4,097,507 4,097,507 Profit for the year 833,040 833,040 833,040 Other comprehensive loss (4,711) (648) (5,359) (5,359) Total comprehensive income/(loss) for the year – – 833,040 (4,711) – (648) 827,681 827,681 Income tax benefit from excess tax deductions related to share-based payments 39,650 39,650 39,650 Share-based payment 235,856 235,856 235,856 Exercise of stock options 169 319,288 319,457 319,457 Ordinary shares withheld for payment of employees’ withholding tax liability (21,869) (21,869) (21,869) Balance on December 31, 2024 7,227 5,948,916 (1,571,804) 126,832 1,047,231 (60,119) 5,498,283 5,498,283 (in thousands of $) Attributable to owners of the parent Share capital Share premium Accumulated losses Translation differences Share-based payment and income tax deduction on share- based payments Fair value movement on investment in equity instruments designated as at FVTOCI Total equity attributable to owners of the parent Total equity argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Consolidated Financial Statements 217
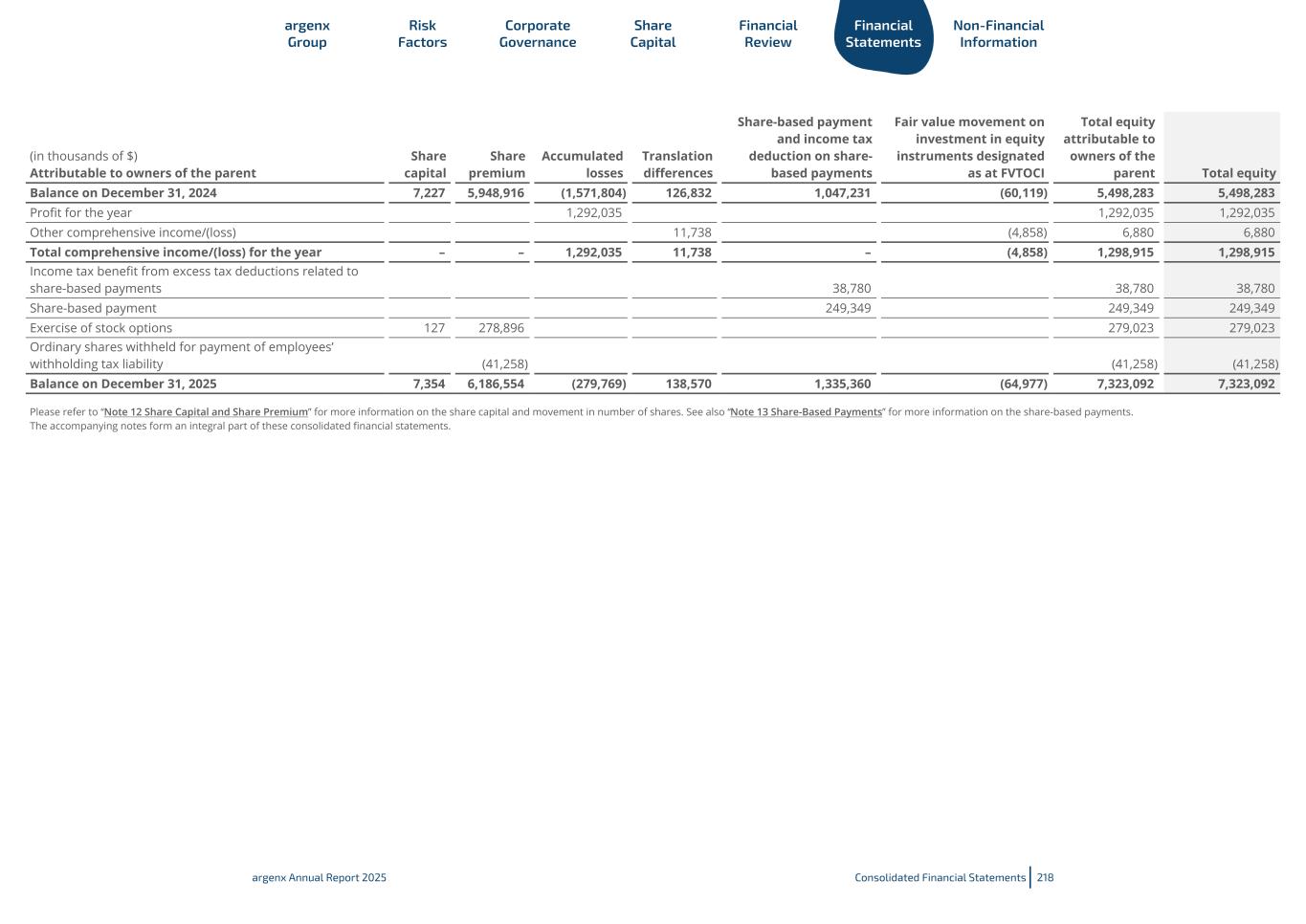
Balance on December 31, 2024 0 7,227 5,948,916 (1,571,804) 126,832 – 1,047,231 – (60,119) 5,498,283 – 5,498,283 Profit for the year 1,292,035 1,292,035 1,292,035 Other comprehensive income/(loss) 11,738 (4,858) 6,880 6,880 Total comprehensive income/(loss) for the year – – 1,292,035 11,738 – (4,858) 1,298,915 1,298,915 Income tax benefit from excess tax deductions related to share-based payments 38,780 38,780 38,780 Share-based payment 249,349 249,349 249,349 Exercise of stock options 127 278,896 279,023 279,023 Ordinary shares withheld for payment of employees’ withholding tax liability (41,258) (41,258) (41,258) Balance on December 31, 2025 7,354 6,186,554 (279,769) 138,570 1,335,360 (64,977) 7,323,092 7,323,092 Please refer to ‘‘Note 12 Share Capital and Share Premium’’ for more information on the share capital and movement in number of shares. See also ‘‘Note 13 Share-Based Payments’’ for more information on the share-based payments. The accompanying notes form an integral part of these consolidated financial statements. (in thousands of $) Attributable to owners of the parent Share capital Share premium Accumulated losses Translation differences Share-based payment and income tax deduction on share- based payments Fair value movement on investment in equity instruments designated as at FVTOCI Total equity attributable to owners of the parent Total equity argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Consolidated Financial Statements 218

6.2 Notes to the Consolidated Financial Statements 1. General Information about the Company argenx SE is a Dutch European public company with limited liability incorporated under the laws of the Netherlands. The company (COC 24435214) has its official seat in Amsterdam, The Netherlands, and its registered office is at Laarderhoogtweg 25, 1101 EB Amsterdam, the Netherlands. An overview of the company and its subsidiaries (the Company) are described in ‘‘Note 29 Overview of Consolidation Scope’’. argenx SE is a publicly traded company with ordinary shares listed on Euronext Brussels under the symbol “ARGX” since July 2014 and with American Depositary Shares listed on Nasdaq under the symbol “ARGX” since May 2017. 2. Material Accounting Policy Information The Company’s material accounting policies are summarized below. 2.1 Statement of compliance and basis of preparation The consolidated financial statements are prepared in accordance with the IFRS® Accounting Standards (IFRS) as adopted by the European Union (EU-IFRS) and in accordance with the legal requirements of Part 9 of Book 2 of the Dutch Civil Code. The consolidated financial statements provide a general overview of the Company’s activities and the results achieved. They present fairly the entity’s financial position, its financial performance and cash flows, on a going concern basis. The material accounting policy information applied in the preparation of the above consolidated financial statements are set out below. All amounts are presented in thousands of US dollar, unless otherwise indicated, rounded to the nearest $ ‘000. The consolidated financial statements have been approved for issue by the Company’s Board of Directors (the “Board”) on March 18, 2026. 2.2 Adoption of new and revised standards New standards and interpretations applicable for the annual period beginning on January 1, 2025 In the current year, the Group has assessed and adopted amendments to IFRS that are mandatorily effective for accounting periods that begin on or after January 1, 2025. Their adoption has not had any material impact on the disclosures or on the amounts reported in these consolidated financial statements. New standards and interpretations issued, but not yet applicable for the annual period beginning on January 1, 2025 • IFRS 18 Presentation and Disclosures in Financial Statements IFRS 18 replaces IAS 1, carrying forward many of the requirements in IAS 1 unchanged and complementing them with new requirements. In addition, some IAS 1 paragraphs have been moved to IAS 8 Accounting Policies, Changes in Accounting Estimates and Errors and IFRS 7 Financial Instruments: Disclosures. Furthermore, the IASB has made minor amendments to IAS 7 Statement of Cash Flows and IAS 33 Earnings per Share. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Notes to the Consolidated Financial Statements 219

IFRS 18 introduces new requirements to: • present specified categories and defined subtotals in the statement of profit or loss • provide disclosures on management-defined performance measures (MPMs) in the notes to the financial statements • improve aggregation and disaggregation. An entity is required to apply IFRS 18 for annual reporting periods beginning on or after January 1, 2027, with earlier application permitted. The amendments to IAS 7 and IAS 33, as well as the revised IAS 8 and IFRS 7, become effective when an entity applies IFRS 18. IFRS 18 requires retrospective application with specific transition provisions. The Company is currently working to identify all impacts the new standard and amendments will have on the primary financial statements and notes to the financial statements. The initial expected material impact on the Company’s financial statements is, as follows: • Foreign exchange differences will be classified in the same category as the income and expenses from the items giving rising to the foreign exchange differences. • Amendments to the Classification and Measurement of Financial Instruments (Amendments to IFRS 9 and IFRS 7) The amendments address matters identified during the post-implementation review of the classification and measurement requirements of IFRS 9 Financial Instruments. The amendments are effective for annual periods starting on or after 1 January 2026. The Company does not anticipate that the amendments will have a material effect on the Company’s financial statements in future periods. We have not early adopted any standard, interpretation, or amendment that has been issued but is not yet effective. 2.3 Basis of consolidation The consolidated financial statements include the financial statements of the Company and entities controlled by the Company (its subsidiaries). Control is achieved when the Company: • has power over the investee; • is exposed, or has rights, to variable returns from its involvement with the investee; and • has the ability to use its power to affect its returns. The Company reassesses whether or not it controls an investee if facts and circumstances indicate that there are changes to one or more of the three elements of control listed above. The results of the subsidiaries are included in the consolidated statements of profit or loss and consolidated statements of other comprehensive income or loss from the effective date of acquisition up to the date when control ceases to exist. When necessary, adjustments are made to the financial statements of subsidiaries to bring their accounting policies into line with those used by other members of the Group. All intercompany transactions and unrealized gains on transactions between group companies are eliminated. Unrealized losses are also eliminated unless the transaction provides evidence of an impairment of the transferred asset. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Notes to the Consolidated Financial Statements 220

2.4 Foreign currency transactions 2.4.1 Functional and presentation currency Items included in the consolidated financial statements of each of the entities are valued using the currency of their economic environment in which the entity operates. The consolidated financial statements are presented in USD ($), which is the Company’s functional and presentation currency. 2.4.2 Transactions and balances Transactions in foreign currencies are translated at the exchange rate ruling at the date of the transaction. Monetary assets and liabilities denominated in foreign currencies are translated at the exchange rate ruling at the reporting date. Foreign exchange differences arising on translation are recognized in the consolidated statements of profit or loss and the consolidated statements of other comprehensive income or loss as “Exchange (losses)/gains”. Non-monetary assets and liabilities denominated in foreign currencies are translated at the foreign exchange rate applicable at the date of the transaction. 2.4.3 Financial statements of foreign entities For foreign entities using a different functional currency than USD: • assets and liabilities for each balance sheet presented are translated at the closing rate at the date of the balance sheet; • income and expenses for each statement presenting profit or loss and statements of other comprehensive income or loss are translated at average exchange rates. 2.5 Intangible assets 2.5.1 Internally generated intangible assets Expenditure on research activities is recognized as an expense in the period in which it is incurred. 2.5.2 Acquired In-Process R&D and Acquired R&D available for use Upfront payments and development milestone payments for “Acquired In-Process R&D” obtained through in-licensing arrangements are capitalized as intangible assets under “Acquired In-Process R&D” upon meeting the IAS 38 capitalization criteria. These intangibles are considered as intangible assets with definite useful lives and are carried at cost less amortization and accumulated impairment losses. The Company has not started to amortize “Acquired In-Process R&D” as they are not available for use until regulatory approval has been obtained or the asset is ready for its intended use, but they are evaluated for potential impairment on an annual basis or when facts and circumstances may indicate a risk of impairment. Any impairment charge is recorded in the consolidated statements of profit or loss and the consolidated statements of other comprehensive income or loss under “Research and development expense”. Once an asset included in “Acquired In-Process R&D” has received marketing approval from a regulatory authority or is ready for its intended use, it is recorded under “Acquired R&D available for use” category. Regulatory milestone payments and sales-based milestone payments for R&D obtained through in-licensing arrangements acquired are capitalized intangible assets under “Acquired R&D available for use” upon meeting the IAS 38 capitalization criteria. All intangibles classified under “Acquired R&D available for use” are considered as intangible assets with finite useful lives and are carried at cost less accumulated amortization and accumulated impairment losses. “Acquired R&D available for use” is evaluated for potential impairment when the Company identifies indications based on facts and circumstances of the asset. Any impairment charge is recorded in the consolidated statements of profit or loss and the consolidated statements of other comprehensive income or loss under “Cost of sales” for assets which are currently generating product net sales. “Acquired R&D available for use” is amortized under “Cost of sales” for assets which are currently generating product net sales on a straight-line basis over the estimated useful life, being the longer of the current patent protection life of the acquired R&D and patent protection life of the combined product. Impairment and amortization relating to assets available for use but not generating sales are recorded under “Research & Development Expenses”. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Notes to the Consolidated Financial Statements 221

2.6 Research and development incentives receivables The current and non-current research and development incentive receivables relate to refunds resulting from research and development incentives on Research and development expenses in Belgium and are credited to the consolidated statements of profit or loss and the consolidated statements of other comprehensive income or loss under the line “Other operating income” when the relevant expenditure has been incurred and there is a reasonable assurance that the research and development incentives will be received. 2.7 Inventories Inventories are valued at the lower of cost and net realizable value. Costs incurred in bringing each product to its present location and condition are accounted for, and include purchase cost, cost of direct materials and labor, and a proportion of manufacturing overheads based on the normal operating capacity. Net realizable value is the estimated selling price in the ordinary course of business, less estimated costs of completion and the estimated costs necessary to make the sale. When the net realizable value falls below the carrying amount, or when inventories become obsolete or excessive, a write-down is recognized. Inventories include products which could be used, besides in commercial activities, in preclinical and clinical programs, in free-of-charge, compassionate use or pre-approval access program. These products, when they are used, are expensed either through “Research & development expenses” or “Selling, general and administrative expenses”. The Company capitalizes inventory costs associated with products prior to the regulatory approval of these products, or for inventory produced in production facilities not yet approved, when it is highly probable that the pre-approval inventories will be sellable. The determination to capitalize is based on the particular facts and circumstances relating to the expected regulatory approval of the product or production facility being considered. The assessment of whether or not the product is considered highly probable to be sellable is made and includes, but is not limited to, how far a particular product or facility has progressed along the approval process, any known safety or efficacy concern and other impediments. Capitalized costs related to pre-launch inventories could be written down upon a change in facts and circumstances. The write-down would be recorded under “Research and development expenses” in the consolidated statements of profit or loss and the consolidated statements of other comprehensive income or loss. 2.8 Trade and other receivables Trade and other receivables are designated as financial assets measured at amortized cost. They are initially measured either at their invoiced amounts or at transaction price, in the absence of a significant financing component less adjustments for estimated revenue deductions such as rebates, chargebacks and returns. All receivables are subsequently measured at amortized cost, which generally corresponds to nominal value less expected credit loss provision. Loss allowance for expected credit losses are established using a simplified approach of forward-looking expected credit loss model (ECL), which includes possible default events on the trade receivables over the entire holding period of the trade receivable. These provisions represent the difference between the trade receivable’s carrying amount in the consolidated statements of financial position and the estimated collectible amount. Charges for loss allowance for expected credit losses are recorded under “Selling, general and administrative expenses” in the consolidated statements of profit or loss and consolidated statements of other comprehensive income or loss. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Notes to the Consolidated Financial Statements 222

2.9 Current financial assets Current financial assets measured at amortized costs comprise of term accounts that have an initial maturity equal or less than twelve months, but exceeding three months. Current financial assets measured at fair value through profit or loss comprise of money market funds. Interests on Current financial assets are reported under Cash Flow from investment activities under “Interest received” and under “Financial income’’ in the Consolidated Statement of Profit or Loss. 2.10 Cash and cash equivalents Cash are financial assets measured at amortized cost and comprise of cash at bank. Cash equivalents measured at amortized cost comprise of term accounts that have an initial maturity of less than three months that are subject to an insignificant risk of changes in values. Cash equivalents are determined at inception and measured at fair value through profit or loss comprise of money market funds that are readily convertible to cash. They are subject to insignificant risk of changes in value and which are used by the Company in the management of its short-term commitments. Cash and cash equivalents exclude restricted cash, which is presented in the consolidated statements of financial position under the line “Other non-current assets”. Interests on Cash and cash equivalents is reported under Cash Flow from investment activities under “Interest received” and under “Financial income’’ in the Consolidated Statement of Profit or Loss. 2.11 Trade and other payables Trade and other payables are comprised of liabilities that are due less than one year from the balance sheet date and are in general not interest bearing and settled on an ongoing basis during the financial year. They also include accrued expenses related to the Company’s operating activities. Trade and other payables are initially measured at their transaction price, which are subsequent to initial recognition measured at amortized cost. Short-term employee benefits include payables and accruals for salaries and bonuses to be paid to the employees of the Company. They are recognized as expenses for the period in which employees perform the corresponding services. 2.12 Leases The Company assesses at contract inception whether a contract is, or contains, a lease. That is, if the contract conveys the right to control the use of an identified asset for a period of time in exchange for consideration. The Company applies a single recognition and measurement approach for all leases, except for short-term leases and leases of low-value assets. The Company recognizes lease liabilities to make lease payments and right-of-use assets representing the right to use the underlying assets. The Company recognizes right-of-use assets at the commencement date of the lease (i.e., the date the underlying asset is available for use). Right-of-use assets are measured at cost, less any accumulated depreciation and impairment losses, and adjusted for any remeasurement of lease liabilities. The cost of right-of-use assets includes the amount of lease liabilities recognized, initial direct costs incurred, and lease payments made at or before the commencement date less any lease incentives received. Right-of-use assets are depreciated on a straight-line basis over the shorter of the lease term and the estimated useful lives of the assets, as follows: • Buildings 2 to 15 years • Vehicles 2 to 4 years • Equipment 2 to 5 years argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Notes to the Consolidated Financial Statements 223

If ownership of the leased asset transfers to the Company at the end of the lease term or the cost reflects the exercise of a purchase option, depreciation is calculated using the estimated useful life of the asset. The Company’s right-of-use assets are included in “Property, Plant and Equipment”. At the commencement date of the lease, the Company recognizes lease liabilities measured at the present value of lease payments to be made over the lease term. The lease payments include fixed payments, and payments of penalties for terminating the lease, if the lease term reflects the Company exercising the option to terminate. In calculating the present value of of lease payments, the Company uses its incremental borrowing rate at the lease commencement date if the interest rate implicit in the lease is not readily determinable. After the commencement date, the amount of lease liabilities is reduced for the lease payments made. In addition, the carrying amount of lease liabilities is remeasured if there is a modification, a change in the lease term, a change in the lease payments, or a change in the assessment of an option to purchase the underlying asset. The Company’s lease liabilities are included in “Lease Liabilities Non-Current” and “Lease Liabilities Current”. The Company applies the short-term lease recognition exemption (i.e., those leases that have a lease term of 12 months or less from the commencement date and do not contain a purchase option) and the lease of low-value assets recognition exemption to leases that are considered to be of low value. Lease payments on short-term leases and leases of low-value assets are recognized as expense within the operating category in the statement of profit or loss, on a straight-line basis over the lease term. 2.13 Financial instruments Financial instruments are initially recognized either at fair value or at transaction price and subsequently measured at either amortized cost or fair value under IFRS 9 on the basis of both the Company’s model for managing the financial assets and the contractual cash flow characteristics of the financial asset. A financial asset is classified as current when the cash flows expected to flow from the instrument mature within one year. Profit share in AgomAb Therapeutics NV: The Company holds investments in non-current financial assets, which based on IFRS 9, are designated as financial assets at fair value through profit or loss. The fair value of listed investments is based upon the closing price of such securities at each reporting date. As there is no active market for this equity instrument, the Company establishes the fair value by using valuation techniques. The changes to the fair valuation is recorded under “Other operating income” in the consolidated statements of profit or loss. Shares of Zai Lab Ltd: Based on IFRS 9, the Company irrevocably elected to designate this specific investment as a financial asset at fair value through OCI as the participation is not held for trading purposes nor contingent consideration recognized by an acquirer in a business combination. The investment is recorded under “Other non-current assets” in consolidated statements of financial position and changes to the fair valuation is recorded under “Fair value gain/(loss) on investments in equity instruments designated as at FVTOCI” in the consolidated statements of other comprehensive income or loss. 2.14 Shareholder’s equity An equity instrument is any contract that evidences a residual interest in the assets of an entity after deducting all of its liabilities. Equity instruments issued by the Company are recognized at the proceeds received, net of direct issue costs. The Company has never distributed any dividends to its shareholders. As of December 31, 2025, no profits were available for distribution. 2.15 Share-based payments Equity-settled share-based payments to employees and others providing similar services are measured at the fair value of the equity instruments at the acceptance date. Equity settled share-based payments argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Notes to the Consolidated Financial Statements 224

includes expenses related to stock options, restricted stock units and performance stock units granted by the Company. The fair value determined at the acceptance date of the equity-settled share-based payments is expensed over the vesting period, based on the Company’s estimate of equity instruments that will eventually vest, with a corresponding increase in equity. At the end of each reporting period, the Company revises its estimate of the number of equity instruments expected to vest. The impact of the revision of the original estimates, if any, is recognized in the consolidated statements of profit or loss and the consolidated statements of other comprehensive income or loss such that the cumulative expense reflects the revised estimate, with a corresponding adjustment to the equity-settled share-based payment reserve. The share-based payment expense is recorded in the “Consolidated Statements of Profit or Loss” depending on the nature of the services provided by each beneficiary. 2.16 Income taxes Income tax in the consolidated statements of profit or loss and the consolidated statements of other comprehensive income or loss represents the total of the current tax and deferred tax. The current tax is based on taxable profit for the year. Taxable profit differs from profit as reported in the consolidated statements of profit or loss and consolidated statements of other comprehensive income or loss as it excludes items of income or expense that are taxable or deductible in other years and items that are never taxable or deductible. The Company’s liability for current tax is calculated using tax rates that have been enacted or substantively enacted by the end of the reporting period. Deferred tax is recognized on temporary differences between the carrying amounts of assets and liabilities in the consolidated financial statements and the corresponding tax basis used in the computation of taxable profit. The Company recognizes deferred tax assets, including the tax base of tax loss carryforwards, if management assesses that these tax assets can be offset against positive taxable profits in the future. This judgment is made on an ongoing basis, considering actual results, budgets, and business plans for the coming years. The realization of deferred tax assets depends on all available factors as of reporting date. The carrying amount of deferred tax assets is reviewed at the end of each reporting period and reduced to the extent that it is not probable that sufficient taxable profits will be available to allow all or part of the asset to be recovered. Deferred tax assets and liabilities are offset if there is a legally enforceable right and intention to offset the income taxes and relates to same tax jurisdiction. Deferred tax assets and liabilities are measured at the tax rates that are expected to apply in the period in which the liability is settled or the asset realized, based on tax rates (and tax laws) that have been enacted or substantially enacted by the end of the reporting period. The Company records uncertain tax positions in accordance with IAS 12 Income Taxes using the two-step test whereby (1) the Company determines whether it is probable that the tax positions will be accepted by relevant taxing authorities, and (2) for those tax positions that are not probable that a tax authority will accept in full the position, the Company recognizes uncertain tax positions using either the most likely amount or the expected value, depending on specific facts and circumstances. 2.17 Product net sales Revenue from the sale of products is recognized at an amount that reflects the consideration that the Company expects to be entitled to receive in exchange for transferring goods to a customer, at the time when the customer obtains control of the goods rendered. The consideration that is committed in a contract with a customer can include fixed amounts, variable amounts, or both. The amount of the consideration may vary due to discounts, rebates, returns, chargebacks or other similar items. Contingent consideration is included in the transaction price when it is highly probable that the amount of revenue recognized is not subject to significant future reversals. Product net sales are recognized once we satisfy the performance obligation at a point in time under the revenue recognition criteria in accordance with IFRS 15 Revenue from contracts with customers. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Notes to the Consolidated Financial Statements 225

Revenue arising from the sale of commercial product is presented in the consolidated statements of profit or loss and the consolidated statements of other comprehensive income or loss under “Product net sales”. In accordance with IFRS 15, such revenue is recognized when the product is transferred, in accordance with the delivery and acceptance terms agreed with the customer. Payment of the transaction price is payable as from the point the customer obtains the legal title to the goods. A deferred revenue is recognized if the Company receives consideration, or has an unconditional right to receive revenue, prior to the completion of all performance obligation. The amount of revenue recognized reflects the various types of price reductions or rights of return offered by the Company to its customers. Such price reductions and rights of return qualify as variable consideration under IFRS 15. Products sold are covered by various Government and State programs for which specific discounts are applied. Rebates are granted to healthcare authorities, and under contractual arrangements with certain customers. Some wholesalers are entitled to chargeback incentives based on the selling price to the end customer, under specific contractual arrangements. Rebates, chargebacks and other incentives are recognized in the period in which the underlying sales are recognized as a reduction of product sales. The significant components of variable consideration are as follows: Rebates & Discounts: We are subject to government mandated rebates & discounts in multiple jurisdictions globally including in the U.S. for Medicaid Drug Rebate Program, Medicare Part D Manufacturer Discount Program, and other government health care programs. Rebate amounts are based upon contractual agreements and/or legal requirements with public sector benefit providers. We use the expected-value method for estimating these rebates. The structure of the Medicare Part D Manufacturer Discount Program was updated to reflect provisions of the Inflation Reduction Act of 2022 (IRA) that became effective January 1, 2025. The Part D redesign sunset the Coverage Gap Discount Program (CGDP) and established the Manufacturer Discount Program. The Medicare Part D Manufacturer Discount Program is a federal program to subsidize the costs of prescription drugs for Medicare beneficiaries in the U.S., which mandates manufacturers to fund a portion of the Medicare Part D coverage for prescription drugs sold to eligible patients. The expected utilization is estimated based on available third-party data and/or historical data. Estimates for these rebates and discounts are adjusted quarterly to reflect the most recent information. We record an accrued liability and reduction of product sales for unpaid rebates related to products for which control has been transferred to customers. Commercial Rebates: Commercial rebates are arrangements with third party payors where the Company will pay the third-party payors rebates and other fees on eligible purchases of the Company’s product. In consideration for the rebates and fees paid, the third-party payors will cover its’ patient purchases made of the Company’s products. The rebates and fees paid will be treated as variable consideration and a reduction to the transaction price. We use the expected-value method for estimating the ultimate rebate and fee paid, which are based on the volume of product sold. We apply the applicable rebate rate against a payor mix factor for the relevant patient populations and to the vials sold in the effective plan year of the rebate to derive a liability recorded. Estimates for these agreements are adjusted quarterly to reflect the most recent information. We record an accrued liability for unpaid commercial rebates. Chargebacks: Chargebacks are discounts that occur in multiple jurisdictions globally, whereby contracted parties purchase directly from a specialty distributor. Contracted parties primarily consist of public health service institutions and government entities. In the U.S., 340B and Veterans Affairs (VA) chargebacks are initiated through arrangements between manufacturers and the government, for which the product is purchased at an agreed discounted price. The reserves for chargeback are based on known sales to contracted parties. We establish the reserves for chargebacks in the same period that the related revenue is recognized, resulting in an accrued liability and reduction of product gross sales. Distributor fees: Specialty distributors provide distribution services to the Company for a fee, based on a contractually determined fixed percentage of sales. As the services being provided by the specialty distributor are not distinct, the recurring service fees paid to specialty distributors are treated as variable argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Notes to the Consolidated Financial Statements 226

consideration and a reduction to the transaction price. We estimate these distributor fees and record such estimates in the same period the related revenue is recognized, resulting in a reduction of product gross sales. We record an accrued liability for unpaid distributor fees. Other components of the variable considerations include co-pay assistance program, Medicare Part B discarded drug refund program in the U.S., and product returns. The estimated amounts described above are recognized in the consolidated statements of profit or loss and the consolidated statements of other comprehensive income or loss within “Product net sales” as a reduction of gross sales, and generally within “Trade and other payables” in the consolidated statements of financial position. They are subject to regular review and adjustment as appropriate based on the most recent data available to management. The Company has determined that sales rebates and reserves constitute a major source of estimation uncertainty as disclosed in ‘‘Note 3 Critical accounting judgments and major sources of estimation uncertainty’’. The major source of estimation uncertainty has been isolated to rebates and chargebacks as disclosed in ‘‘Note 14 Trade and Other Payables’’. If management’s estimates differ from actual results, we will record adjustments that would affect product sales in the period of adjustment. 2.18 Other operating income - Collaboration and license agreements Amongst the Company’s collaboration and license agreements in scope of IFRS 15, the following two have generated revenue in the reporting periods: Zai Lab Under the collaboration agreement, the Company provides clinical and commercial supply to Zai Lab. The Company concludes to recognize such sales as revenue given that the Company acts as principal in the transaction as the risk related to inventory is borne by the Company until the inventory is transferred to Zai Lab. The revenue related to clinical supply is recorded under line item “Other operating income”. The revenue related to commercial supply is recorded under line item “Product net sales” in the Consolidated Statements of Profit or Loss. The income related to royalties or sales-based milestones on sales made in China is recorded under line item “Other operating income”. AbbVie For the AbbVie Collaboration Agreement the Company has determined that the transfer of license combined with the performance of research and development activities represent one single performance obligation. The Company concluded that the license is not distinct in the context of the contract. The transaction price is composed of a fixed part, being an upfront license fee, and a variable part, being milestone payments and cost reimbursements for research and development activities delivered. Milestone payments are only included in the transaction price to the extent it is highly probable that a significant reversal in the amount of cumulative revenue recognition will not occur when the uncertainty associated with the variable consideration is subsequently resolved. Management estimates the amount to be included in the transaction price upon achievement of the milestone event. Sales-based milestones and sales-based royalties are a part of the Company’s arrangements but are not yet included in its revenues. The transaction price has been allocated to the single performance obligation and revenues have been recognized over the estimated service period based on an input model, being the percentage of completion method. The upfront license fee has been fully recognized since 2021 as the performance obligation has been fulfilled at that time. Milestone payments that become highly probable after the performance obligation has been fulfilled are therefore recognized at that point in time. 2.19 Cost of Sales Cost of sales are recognized when the associated revenue from product net sales is recognized. Cost of sales include material, manufacturing costs and other costs attributable to production, including shipping costs relevant amortizations, as well as royalties payable on sold products. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Notes to the Consolidated Financial Statements 227

3. Critical accounting judgments and major sources of estimation uncertainty In the application of the Company’s accounting policies, which are described above, the Company is required to make judgments, estimates and assumptions about the carrying amounts of assets and liabilities that are not readily apparent from other sources. The estimates and associated assumptions are based on historical experience and other factors that are considered to be relevant. Actual results may differ from these estimates. The estimates and underlying assumptions are reviewed on an ongoing basis. Revisions to accounting estimates are recognized in the period in which the estimate is revised if the revision affects only that period or in the period of the revision and future periods if the revision affects both current and future periods. Major sources of estimation uncertainty Sales rebates and reserves Product Sales are recognized when the Company has transferred control of the goods to the customer. Product Sales are subject to various deductions, which are primarily composed of rebates to government agencies, distributors, health insurance companies and managed healthcare organizations to arrive to ‘‘Product net sales’’. Certain deductions from Product Sales are subject to payment based on claims after the initial recognition of the sale due to the time lag between the point of sale and receipt of a claim. Upon initial recognition of the Product Sales, the Company recognizes a liability for the variable consideration based on the Company’s best estimate of expected claims. This estimate is a source of complexity and uncertainty as the Company specifically estimates the payor mix. Additionally, the transaction price is based upon contracts with customers, healthcare providers, payors and government agencies, regulated discounts applicable to government-funded programs, historical experience of claims received and other relevant factors. These open claims are recorded as liabilities under “Sales rebates and reserves’’ in the ‘‘Consolidated Statements of Financial Position’’. The Company reviews these liabilities at each reporting period to take into account potential changes in the programs, the volume of claims and/or the most probable final outcome associated to each sale. In line with IFRS 15, the Company applies constraint in recognition of variable compensation on Product Net Sales. Due to the nature of these liabilities it is not practicable to give meaningful sensitivity estimates due to the large volume of variables that contribute to Medicare Part D, commercial rebates and chargebacks as outlined in “Note 2.17 Product net sales”. Future events could cause the assumptions within our valuation models to change and materially affect the future results of the Company. Please refer to “Note 14 Trade and Other Payables” for the movement over the period and the ending balance of the sales rebates and reserves. Critical accounting judgment The Company has not exercised a critical accounting judgment in the current year ended December 31, 2025. As of the year ended December 31, 2024, the Company had applied judgment related to the number of years of forecasted future taxable profits to be considered as reliable as positive evidence towards its estimate on recognition of deferred tax assets. The Company considers that as of December 31, 2025, the recognition of its deferred tax assets do not constitute a critical accounting judgment based on the facts and circumstances; specifically, significant growth in the U.S. based Product Net Sales, current and future taxable profit levels and the current external competitive landscape. No other Critical accounting judgments and major sources of estimation uncertainty have been made in the current period by the Company. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Notes to the Consolidated Financial Statements 228
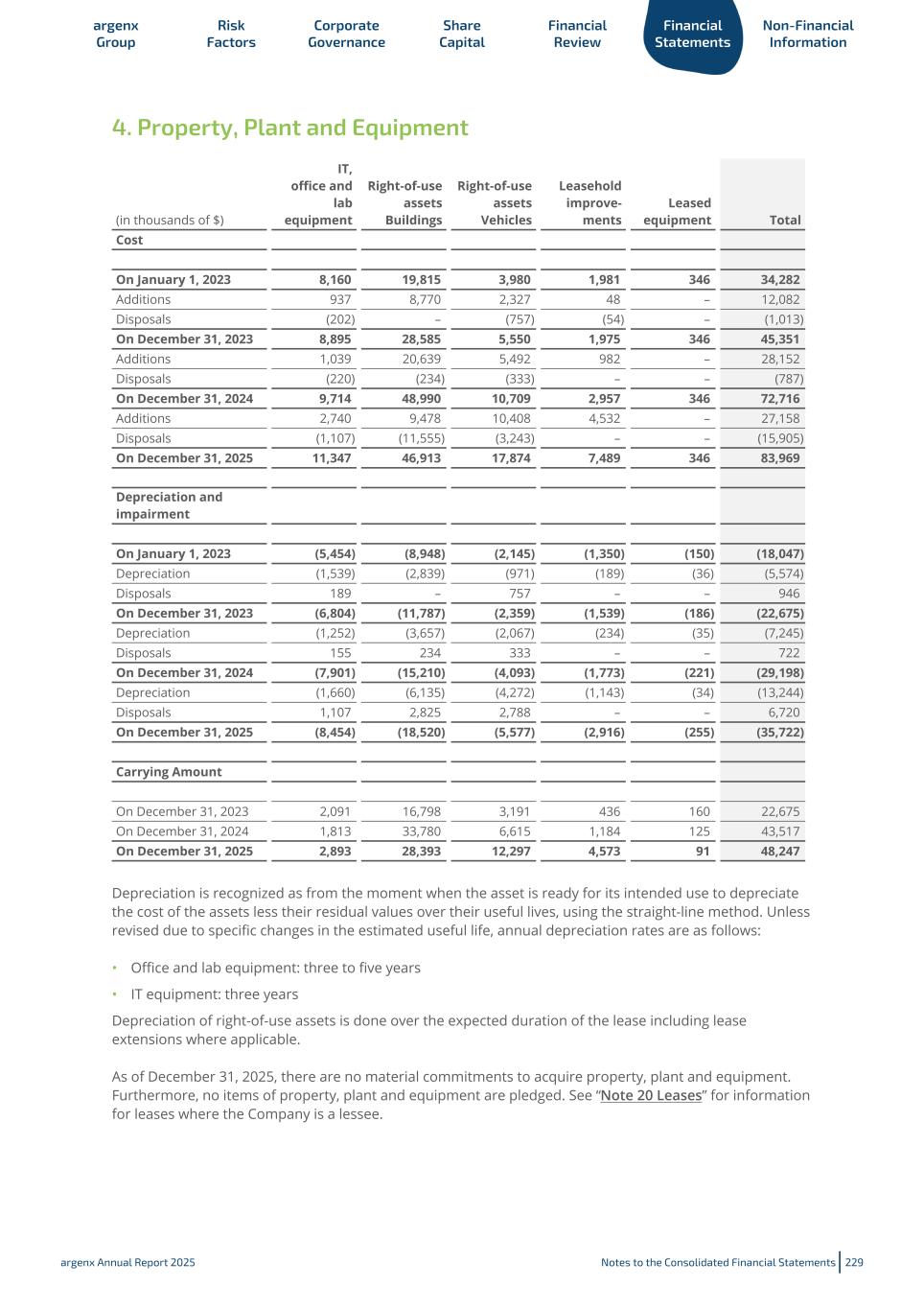
4. Property, Plant and Equipment (in thousands of $) IT, office and lab equipment Right-of-use assets Buildings Right-of-use assets Vehicles Leasehold improve ments Leased equipment Total Cost On January 1, 2023 8,160 19,815 3,980 1,981 346 34,282 Additions 937 8,770 2,327 48 – 12,082 Disposals (202) – (757) (54) – (1,013) On December 31, 2023 8,895 28,585 5,550 1,975 346 45,351 Additions 1,039 20,639 5,492 982 – 28,152 Disposals (220) (234) (333) – – (787) On December 31, 2024 9,714 48,990 10,709 2,957 346 72,716 Additions 2,740 9,478 10,408 4,532 – 27,158 Disposals (1,107) (11,555) (3,243) – – (15,905) On December 31, 2025 11,347 46,913 17,874 7,489 346 83,969 Depreciation and impairment On January 1, 2023 (5,454) (8,948) (2,145) (1,350) (150) (18,047) Depreciation (1,539) (2,839) (971) (189) (36) (5,574) Disposals 189 – 757 – – 946 On December 31, 2023 (6,804) (11,787) (2,359) (1,539) (186) (22,675) Depreciation (1,252) (3,657) (2,067) (234) (35) (7,245) Disposals 155 234 333 – – 722 On December 31, 2024 (7,901) (15,210) (4,093) (1,773) (221) (29,198) Depreciation (1,660) (6,135) (4,272) (1,143) (34) (13,244) Disposals 1,107 2,825 2,788 – – 6,720 On December 31, 2025 (8,454) (18,520) (5,577) (2,916) (255) (35,722) Carrying Amount On December 31, 2023 2,091 16,798 3,191 436 160 22,675 On December 31, 2024 1,813 33,780 6,615 1,184 125 43,517 On December 31, 2025 2,893 28,393 12,297 4,573 91 48,247 Depreciation is recognized as from the moment when the asset is ready for its intended use to depreciate the cost of the assets less their residual values over their useful lives, using the straight-line method. Unless revised due to specific changes in the estimated useful life, annual depreciation rates are as follows: • Office and lab equipment: three to five years • IT equipment: three years Depreciation of right-of-use assets is done over the expected duration of the lease including lease extensions where applicable. As of December 31, 2025, there are no material commitments to acquire property, plant and equipment. Furthermore, no items of property, plant and equipment are pledged. See “Note 20 Leases” for information for leases where the Company is a lessee. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Notes to the Consolidated Financial Statements 229
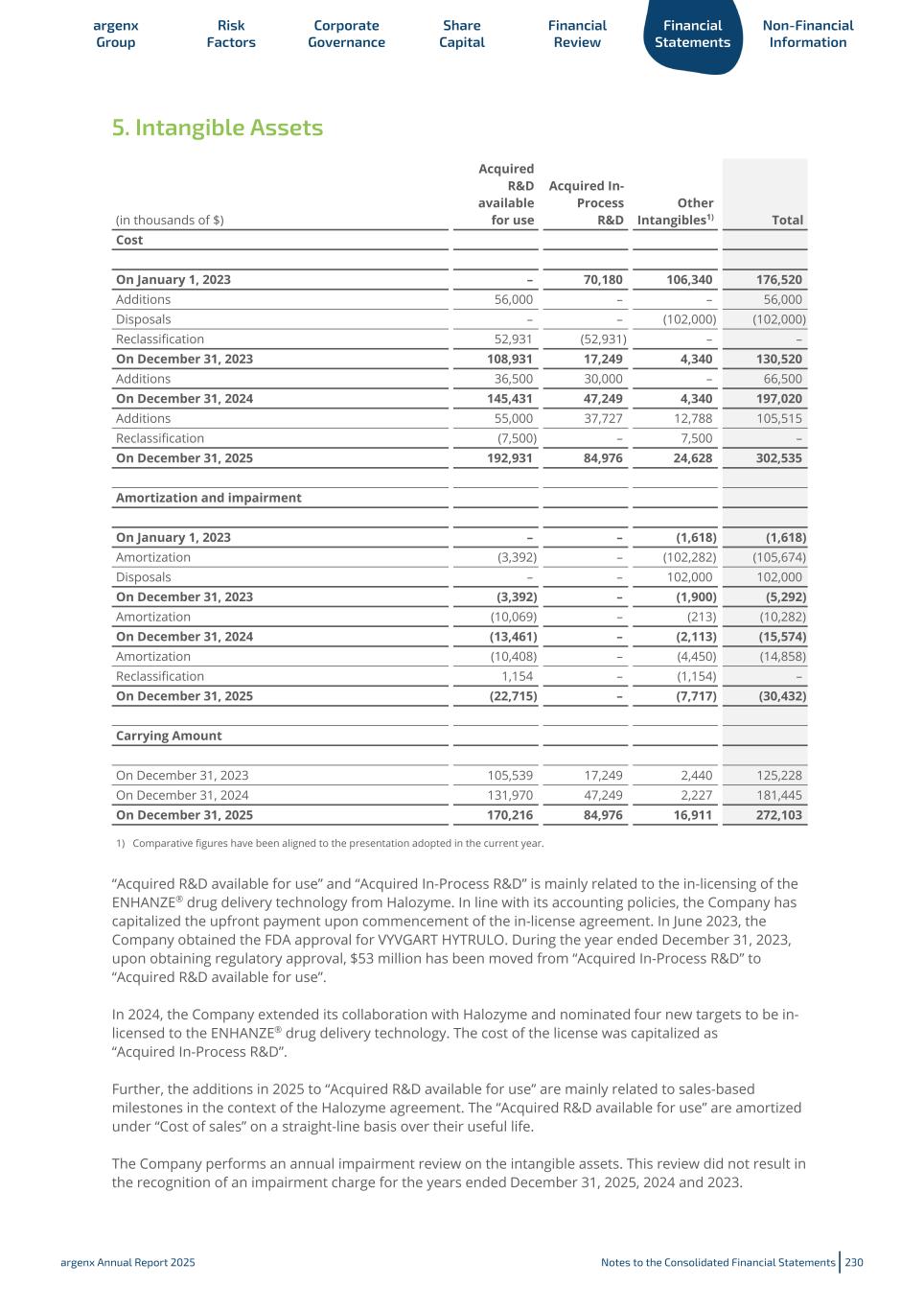
5. Intangible Assets (in thousands of $) Acquired R&D available for use Acquired In- Process R&D Other Intangibles1) Total Cost On January 1, 2023 – 70,180 106,340 176,520 Additions 56,000 – – 56,000 Disposals – – (102,000) (102,000) Reclassification 52,931 (52,931) – – On December 31, 2023 108,931 17,249 4,340 130,520 Additions 36,500 30,000 – 66,500 On December 31, 2024 145,431 47,249 4,340 197,020 Additions 55,000 37,727 12,788 105,515 Reclassification (7,500) – 7,500 – On December 31, 2025 192,931 84,976 24,628 302,535 Amortization and impairment On January 1, 2023 – – (1,618) (1,618) Amortization (3,392) – (102,282) (105,674) Disposals – – 102,000 102,000 On December 31, 2023 (3,392) – (1,900) (5,292) Amortization (10,069) – (213) (10,282) On December 31, 2024 (13,461) – (2,113) (15,574) Amortization (10,408) – (4,450) (14,858) Reclassification 1,154 – (1,154) – On December 31, 2025 (22,715) – (7,717) (30,432) Carrying Amount On December 31, 2023 105,539 17,249 2,440 125,228 On December 31, 2024 131,970 47,249 2,227 181,445 On December 31, 2025 170,216 84,976 16,911 272,103 1) Comparative figures have been aligned to the presentation adopted in the current year. “Acquired R&D available for use” and “Acquired In-Process R&D” is mainly related to the in-licensing of the ENHANZE® drug delivery technology from Halozyme. In line with its accounting policies, the Company has capitalized the upfront payment upon commencement of the in-license agreement. In June 2023, the Company obtained the FDA approval for VYVGART HYTRULO. During the year ended December 31, 2023, upon obtaining regulatory approval, $53 million has been moved from “Acquired In-Process R&D” to “Acquired R&D available for use”. In 2024, the Company extended its collaboration with Halozyme and nominated four new targets to be in- licensed to the ENHANZE® drug delivery technology. The cost of the license was capitalized as “Acquired In-Process R&D”. Further, the additions in 2025 to “Acquired R&D available for use” are mainly related to sales-based milestones in the context of the Halozyme agreement. The “Acquired R&D available for use” are amortized under “Cost of sales” on a straight-line basis over their useful life. The Company performs an annual impairment review on the intangible assets. This review did not result in the recognition of an impairment charge for the years ended December 31, 2025, 2024 and 2023. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Notes to the Consolidated Financial Statements 230

In the fourth quarter of 20232024, the Company utilized the PRV submitted with the sBLA filing for VYVGART HYTRULO for the treatment of CIDP, which resulted in the amortization of $102 million of intangible assets which is recognized under “Research and development expenses” within the consolidated statements of profit or loss and the consolidated statements of other comprehensive income or loss and subsequent derecognition of $102 million of intangibles included under “Other intangibles” on the consolidated statements of financial position. The Company continually assesses the useful life of intangible assets based on the shorter of its expected useful life and legal life. Currently, intangible assets are amortized over a range of three to more than fifteen years. As of December 31, 2025, there are no material commitments to acquire intangible assets, except as set forth in “Note 27 Commitments”. No intangible assets are pledged as security for liabilities nor are there any intangible assets whose title is restricted. 6. Other Non-Current Assets Other non-current assets consisted of non-current restricted cash and financial assets held at fair value through profit or loss or through OCI. As of December 31, (in thousands of $) 2025 2024 2023 Non-current restricted assets 4,838 1,964 2,419 Non-current financial assets held at fair value through profit or loss 37,130 25,549 21,715 Non-current financial assets held at fair value through OCI 10,022 14,880 15,528 Total other non-current assets 51,990 42,393 39,662 Non-current restricted assets on December 31, 2025 was mainly composed of deposit guarantees paid under the lease agreements for the laboratory and offices of the Company. Non-current financial assets held at fair value through profit or loss is comprised of the profit share in AgomAb Therapeutics NV. In March 2019, the Company entered into a license agreement with AgomAb Therapeutics NV for the use of HGF-mimetic SIMPLE Antibodies™, developed under the Company’s Immunology Innovative Program. In exchange for granting this license, the Company received a profit share in AgomAb Therapeutics NV. Since AgomAb Therapeutics NV is a private company, the valuation of the profit share is based on the post-money valuation coming from its most recent financing round. In October 2023, AgomAb Therapeutics NV secured €100 million as a result of a Series C financing round. The Company’s profit share was diluted, but resulting in no change of the fair value. In October 2024, AgomAb Therapeutics NV secured $89 million as a result of a Series D financing round. The Company reassessed the fair value of the asset as of December 31, 2025 based on publicly available clinical announcements as of the reporting date. Fair value changes on non-current financial assets with fair value through profit or loss are recognized in the consolidated statements of profit or loss and the consolidated statements of other comprehensive income or loss under “Other operating income”. As part of the license agreement for the development and commercialization for efgartigimod in Greater China, in 2021 the Company obtained, amongst others, 568,182 newly issued Zai Lab shares calculated at a price of €132 per share. The fair value of the equity instrument at reporting date is determined by reference to the closing price of such securities at each reporting date (classified as level 1 in the fair value hierarchy). The Company made the irrevocable election to recognize subsequent changes in fair value through OCI under “Fair value gain/(loss) on investments in equity instruments designated as at FVTOCI”. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Notes to the Consolidated Financial Statements 231
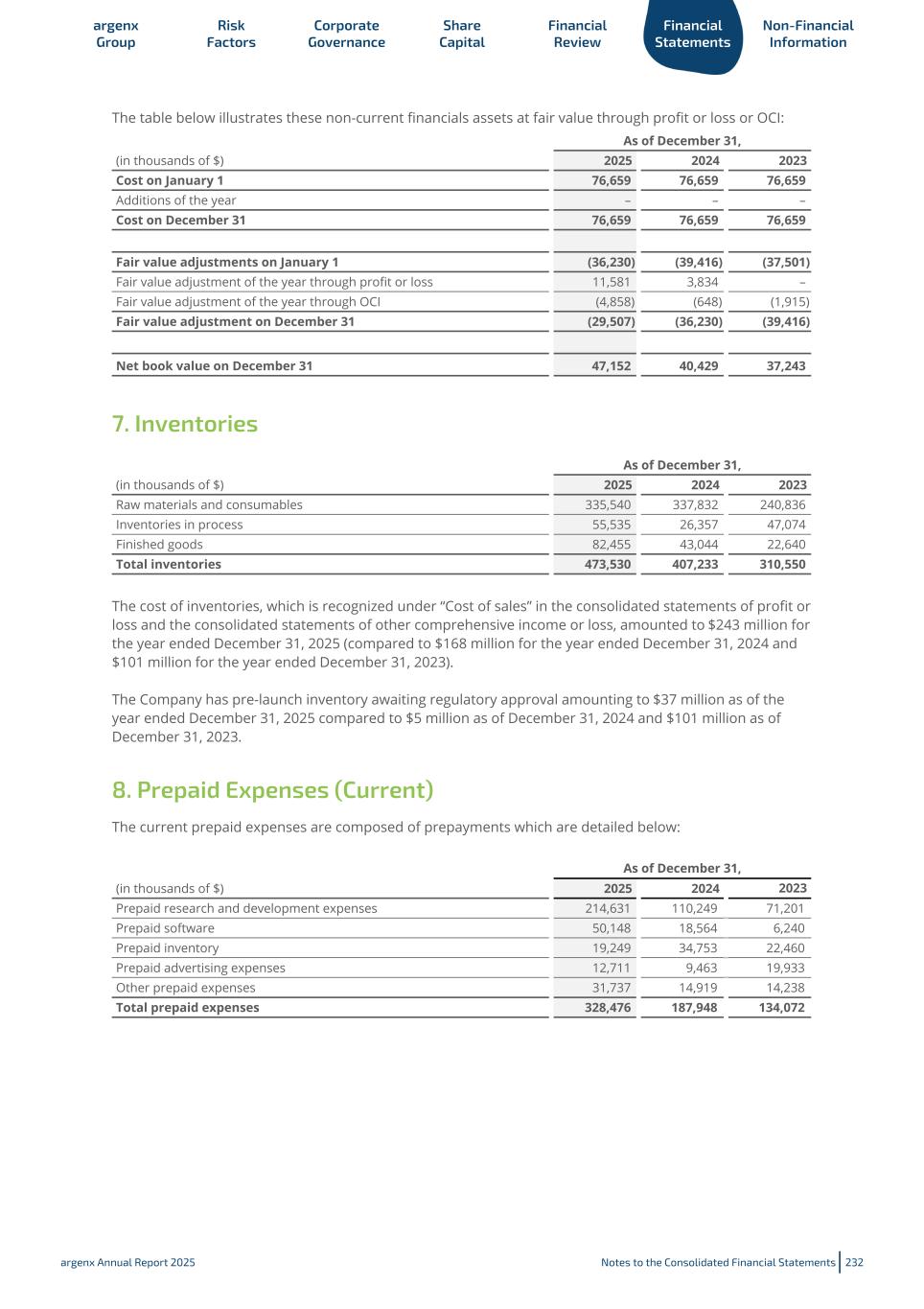
The table below illustrates these non-current financials assets at fair value through profit or loss or OCI: As of December 31, (in thousands of $) 2025 2024 2023 Cost on January 1 76,659 76,659 76,659 Additions of the year – – – Cost on December 31 76,659 76,659 76,659 Fair value adjustments on January 1 (36,230) (39,416) (37,501) Fair value adjustment of the year through profit or loss 11,581 3,834 – Fair value adjustment of the year through OCI (4,858) (648) (1,915) Fair value adjustment on December 31 (29,507) (36,230) (39,416) Net book value on December 31 47,152 40,429 37,243 7. Inventories As of December 31, (in thousands of $) 2025 2024 2023 Raw materials and consumables 335,540 337,832 240,836 Inventories in process 55,535 26,357 47,074 Finished goods 82,455 43,044 22,640 Total inventories 473,530 407,233 310,550 The cost of inventories, which is recognized under “Cost of sales” in the consolidated statements of profit or loss and the consolidated statements of other comprehensive income or loss, amounted to $243 million for the year ended December 31, 2025 (compared to $168 million for the year ended December 31, 2024 and $101 million for the year ended December 31, 2023). The Company has pre-launch inventory awaiting regulatory approval amounting to $37 million as of the year ended December 31, 2025 compared to $5 million as of December 31, 2024 and $101 million as of December 31, 2023. 8. Prepaid Expenses (Current) The current prepaid expenses are composed of prepayments which are detailed below: As of December 31, (in thousands of $) 2025 2024 2023 Prepaid research and development expenses 214,631 110,249 71,201 Prepaid software 50,148 18,564 6,240 Prepaid inventory 19,249 34,753 22,460 Prepaid advertising expenses 12,711 9,463 19,933 Other prepaid expenses 31,737 14,919 14,238 Total prepaid expenses 328,476 187,948 134,072 argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Notes to the Consolidated Financial Statements 232

9. Trade and Other Receivables The trade and other receivables are composed of receivables which are detailed below: As of December 31, (in thousands of $) 2025 2024 2023 Trade receivables 1,489,572 817,707 417,994 Tax receivables 123,123 40,886 63,605 Interest receivables 33,533 40,214 13,126 Other receivables 464 5,664 1,962 Total trade and other receivables 1,646,692 904,471 496,687 The carrying amounts of trade and other receivables approximate their respective fair values. On December 31, 2025, 2024 and 2023, we did not have a material provision for expected credit losses. Please also refer to “Note 24 Financial Risk Management” for more information on the financial risk management. 10. Financial Assets — Current These current financial assets relate to term accounts with an initial maturity longer than three months and less than 12 months and money market funds that do not qualify as cash equivalents as they are not expected to be used to meet short-term commitments. As of December 31, (in thousands of $) 2025 2024 2023 Term accounts 948,750 1,878,890 1,131,000 Total current financial assets 948,750 1,878,890 1,131,000 On December 31, 2025, the current financial assets included $59 million (€50 million) held in EUR (compared to $104 million (€100 million) for the year ended December 31, 2024 and $221 million (€200 million) for the year ended December 31, 2023) which could generate a foreign currency exchange gain or loss in the financial results in accordance with the fluctuations of the USD/EUR exchange rate as the Company’s functional currency is USD. Please also refer to “Note 24 Financial Risk Management” for more information on the financial risk management. 11. Cash and Cash Equivalents As of December 31, (in thousands of $) 2025 2024 2023 Money market funds 2,541,112 1,394,409 1,678,100 Term accounts 945,001 100,000 350,000 Cash and bank balances 5,176 5,527 20,744 Total cash and cash equivalents 3,491,289 1,499,936 2,048,844 Cash and cash equivalents comprise of cash and bank balances, term accounts with an original maturity not exceeding three months and money market funds that are readily convertible to cash and are subject to an insignificant risk of changes in value. Cash positions are invested with preferred financial partners, which are considered to be high quality financial institutions with sound credit ratings to reduce credit risk. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Notes to the Consolidated Financial Statements 233

On December 31, 2025, the cash and cash equivalents included $97 million (€83 million) held in EUR (compared to $653 million (€628 million) for the year ended December 31, 2024 and $703 million (€636 million) for the year ended December 31, 2023) which could generate a foreign currency exchange gain or loss in the financial results in accordance with the fluctuation of the USD/EUR exchange rate as the Company’s functional currency is USD. Please also refer to “Note 24 Financial Risk Management” for more information on the financial risk management. 12. Share Capital and Share Premium As of December 31, 2025, the Company’s share capital was represented by 61,883,306 shares. All shares were issued, fully paid up and of the same class. The table below summarizes the share issuances as a result of offerings, exercise of stock options and the vesting of restricted stock units under the Company’s Employee Stock Option Plan. Roll forward of number of shares outstanding: Number of shares outstanding on January 1, 2023 55,395,856 Exercise of stock options 1,137,439 Vesting of RSUs 79,560 Global public offering on Nasdaq on July 18, 2023 2,244,899 Over-allotment option exercised by underwriters on July 19, 2023 336,734 Number of shares outstanding on December 31, 2023 59,194,488 Exercise of stock options 1,478,225 Vesting of RSUs 88,244 Number of shares outstanding on December 31, 2024 60,760,957 Exercise of stock options 986,507 Vesting of RSUs 135,842 Number of shares outstanding on December 31, 2025 61,883,306 On July 18, 2023, argenx SE offered 2,244,899 of its ordinary shares through a global offering which consisted of 1,580,981 ADSs in the U.S. at a price of $490.00 per ADS, before underwriting discounts and commissions and offering expenses; and 663,918 ordinary shares in the European Economic Area at a price of €436.37 per share, before underwriting discounts and commissions and offering expenses. On July 19, 2023, the underwriters of the offering exercised their overallotment option to purchase 336,734 additional ADSs in full. As a result, argenx SE received $1.3 billion in gross proceeds from this offering, decreased by $66 million of underwriter discounts and commissions, and offering expenses, of which $1 million has been deducted from equity. The total net cash proceeds from the offering amounted to $1.2 billion. On May 27, 2025, at the annual general meeting, the shareholders of the Company approved the authorization to the Board to issue up to a maximum of 10% of the then-outstanding share capital, for a period of 18 months. On December 31, 2025, an amount of €532,863, represented by 5,328,634 shares, still remained available under the authorization to issue shares as granted to the Board by the shareholders of the Company. 13. Share-Based Payments The Company has an equity incentive plan for the employees, key consultants, board members, senior management and key outside advisors (‘’key persons”) of the Company and its subsidiaries. In accordance with the terms of the plan, as approved by shareholders, employees may be granted stock options and/or restricted stock units and/or performance stock units. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Notes to the Consolidated Financial Statements 234

13.1. Stock Options The stock options are granted to key persons of the Company and its subsidiaries. The stock options may be granted to purchase ordinary shares at an exercise price. The stock options have been granted free of charge. Each employee’s stock option converts into one ordinary share of the Company upon exercise. The stock options carry neither rights to dividends nor voting rights. Stock options may be exercised at any time from the date of vesting to the date of their expiry. The stock options granted vest, in principle, as follows: • 1/3rd of the total stock options granted will vest on the first anniversary of the granting of the stock options, and • 1/36th of the total stock options granted will vest on the first day of each month following the first anniversary of the granting of the stock options. Stock options granted to non-executive directors vest on the third anniversary of the date of grant. Upon leave of the key persons stock options must be exercised before the later of (i) 90 days after the last working day at argenx, or (ii) March 31 of the fourth year following the date of grant of those stock options, and in any case no later than the expiration date of the option. In order to pre-finance the taxes that are paid upon the grant of stock options, Belgian employees have the ability, in exchange for the taxes due upon the grant of the stock options, to transfer the economic benefits related to part of those stock options to a third party. In the year ending December 31, 2025, the economic benefits of 12,951 stock options, for which accelerated vesting applies, were transferred to a third party. No other conditions are attached to stock options. The following stock option arrangements were in existence during the current and prior years and which are exercisable at the end of each period presented: 2024 2.87 – – 3,308 2024 4.64 – – 532 2024 8.43 – – 81,500 2025 13.44 – 400 1,600 2025 11.13 – 78,690 99,326 2026 13.48 75,643 93,378 97,972 2026 13.37 10,000 14,000 24,400 2026 16.61 95,749 103,859 111,811 2027 21.63 31,646 35,046 38,434 2027 24.87 136,705 152,085 225,852 2028 94.96 6,670 7,370 13,890 2028 101.43 187,281 190,011 225,457 2024 133.35 – – 26,171 2029 133.35 33,513 44,158 71,573 2024 159.51 – – 104,176 2029 159.51 251,019 275,154 370,566 2025 140.45 – 3,758 16,712 2030 140.45 18,291 30,675 50,801 2025 230.48 – 7,926 126,331 2030 230.48 69,359 79,691 160,677 2025 235.26 – 5,629 31,424 2030 235.26 31,388 47,908 78,534 2025 290.93 – 90,425 202,205 2030 290.93 246,192 351,911 559,173 Outstanding stock options on December 31, Expiry date Exercise price per stock options (in $)¹⁾ 2025 2024 2023 argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Notes to the Consolidated Financial Statements 235
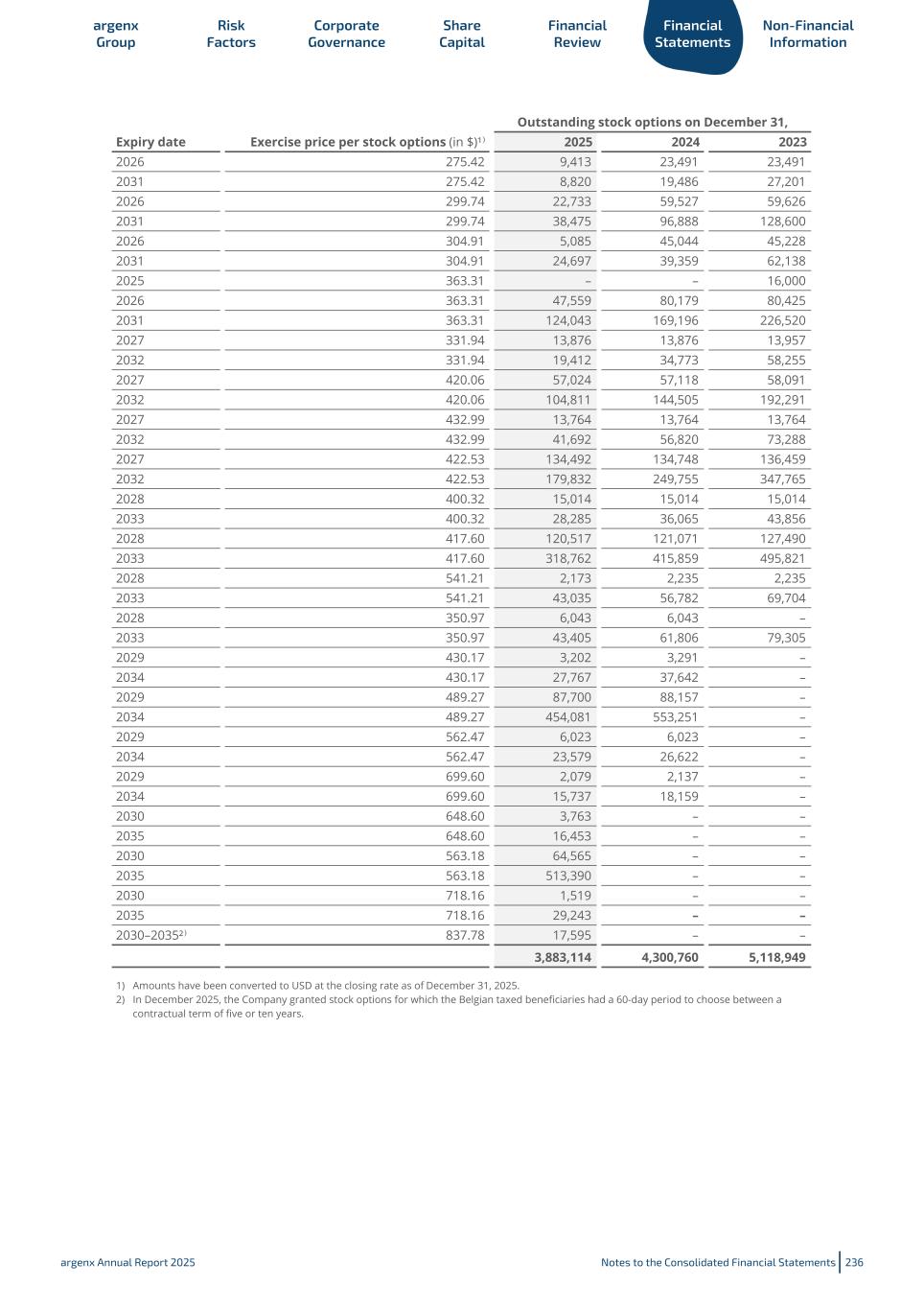
2026 275.42 9,413 23,491 23,491 2031 275.42 8,820 19,486 27,201 2026 299.74 22,733 59,527 59,626 2031 299.74 38,475 96,888 128,600 2026 304.91 5,085 45,044 45,228 2031 304.91 24,697 39,359 62,138 2025 363.31 – – 16,000 2026 363.31 47,559 80,179 80,425 2031 363.31 124,043 169,196 226,520 2027 331.94 13,876 13,876 13,957 2032 331.94 19,412 34,773 58,255 2027 420.06 57,024 57,118 58,091 2032 420.06 104,811 144,505 192,291 2027 432.99 13,764 13,764 13,764 2032 432.99 41,692 56,820 73,288 2027 422.53 134,492 134,748 136,459 2032 422.53 179,832 249,755 347,765 2028 400.32 15,014 15,014 15,014 2033 400.32 28,285 36,065 43,856 2028 417.60 120,517 121,071 127,490 2033 417.60 318,762 415,859 495,821 2028 541.21 2,173 2,235 2,235 2033 541.21 43,035 56,782 69,704 2028 350.97 6,043 6,043 – 2033 350.97 43,405 61,806 79,305 2029 430.17 3,202 3,291 – 2034 430.17 27,767 37,642 – 2029 489.27 87,700 88,157 – 2034 489.27 454,081 553,251 – 2029 562.47 6,023 6,023 – 2034 562.47 23,579 26,622 – 2029 699.60 2,079 2,137 – 2034 699.60 15,737 18,159 – 2030 648.60 3,763 – – 2035 648.60 16,453 – – 2030 563.18 64,565 – – 2035 563.18 513,390 – – 2030 718.16 1,519 – – 2035 718.16 29,243 – – 2030–2035²⁾ 837.78 17,595 – – 3,883,114 4,300,760 5,118,949 1) Amounts have been converted to USD at the closing rate as of December 31, 2025. 2) In December 2025, the Company granted stock options for which the Belgian taxed beneficiaries had a 60-day period to choose between a contractual term of five or ten years. Outstanding stock options on December 31, Expiry date Exercise price per stock options (in $)¹⁾ 2025 2024 2023 argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Notes to the Consolidated Financial Statements 236
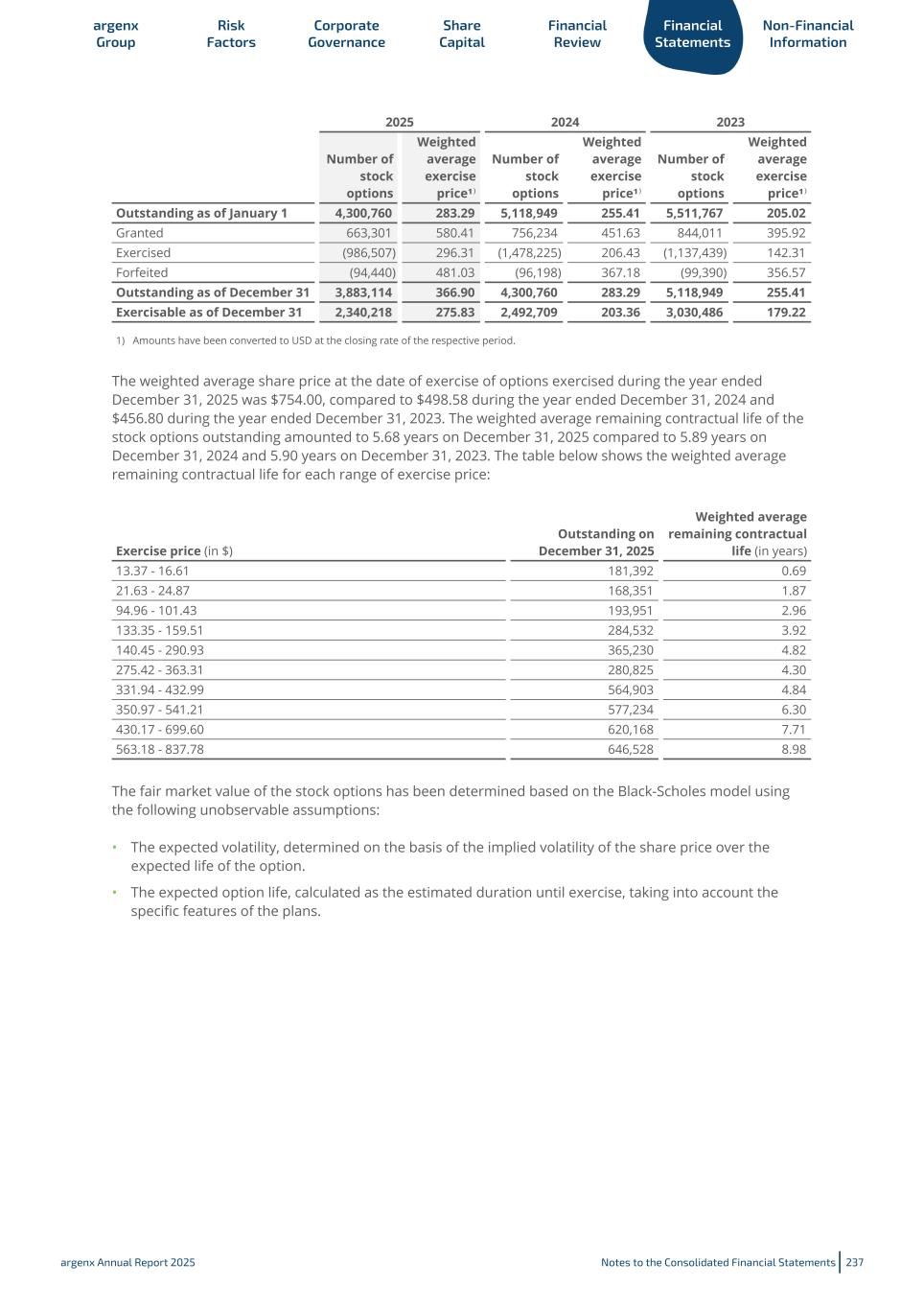
2025 2024 2023 Number of stock options Weighted average exercise price¹⁾ Number of stock options Weighted average exercise price¹⁾ Number of stock options Weighted average exercise price¹⁾ Outstanding as of January 1 4,300,760 283.29 5,118,949 255.41 5,511,767 205.02 Granted 663,301 580.41 756,234 451.63 844,011 395.92 Exercised (986,507) 296.31 (1,478,225) 206.43 (1,137,439) 142.31 Forfeited (94,440) 481.03 (96,198) 367.18 (99,390) 356.57 Outstanding as of December 31 3,883,114 366.90 4,300,760 283.29 5,118,949 255.41 Exercisable as of December 31 2,340,218 275.83 2,492,709 203.36 3,030,486 179.22 1) Amounts have been converted to USD at the closing rate of the respective period. The weighted average share price at the date of exercise of options exercised during the year ended December 31, 2025 was $754.00, compared to $498.58 during the year ended December 31, 2024 and $456.80 during the year ended December 31, 2023. The weighted average remaining contractual life of the stock options outstanding amounted to 5.68 years on December 31, 2025 compared to 5.89 years on December 31, 2024 and 5.90 years on December 31, 2023. The table below shows the weighted average remaining contractual life for each range of exercise price: Exercise price (in $) Outstanding on December 31, 2025 Weighted average remaining contractual life (in years) 13.37 - 16.61 181,392 0.69 21.63 - 24.87 168,351 1.87 94.96 - 101.43 193,951 2.96 133.35 - 159.51 284,532 3.92 140.45 - 290.93 365,230 4.82 275.42 - 363.31 280,825 4.30 331.94 - 432.99 564,903 4.84 350.97 - 541.21 577,234 6.30 430.17 - 699.60 620,168 7.71 563.18 - 837.78 646,528 8.98 The fair market value of the stock options has been determined based on the Black-Scholes model using the following unobservable assumptions: • The expected volatility, determined on the basis of the implied volatility of the share price over the expected life of the option. • The expected option life, calculated as the estimated duration until exercise, taking into account the specific features of the plans. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Notes to the Consolidated Financial Statements 237
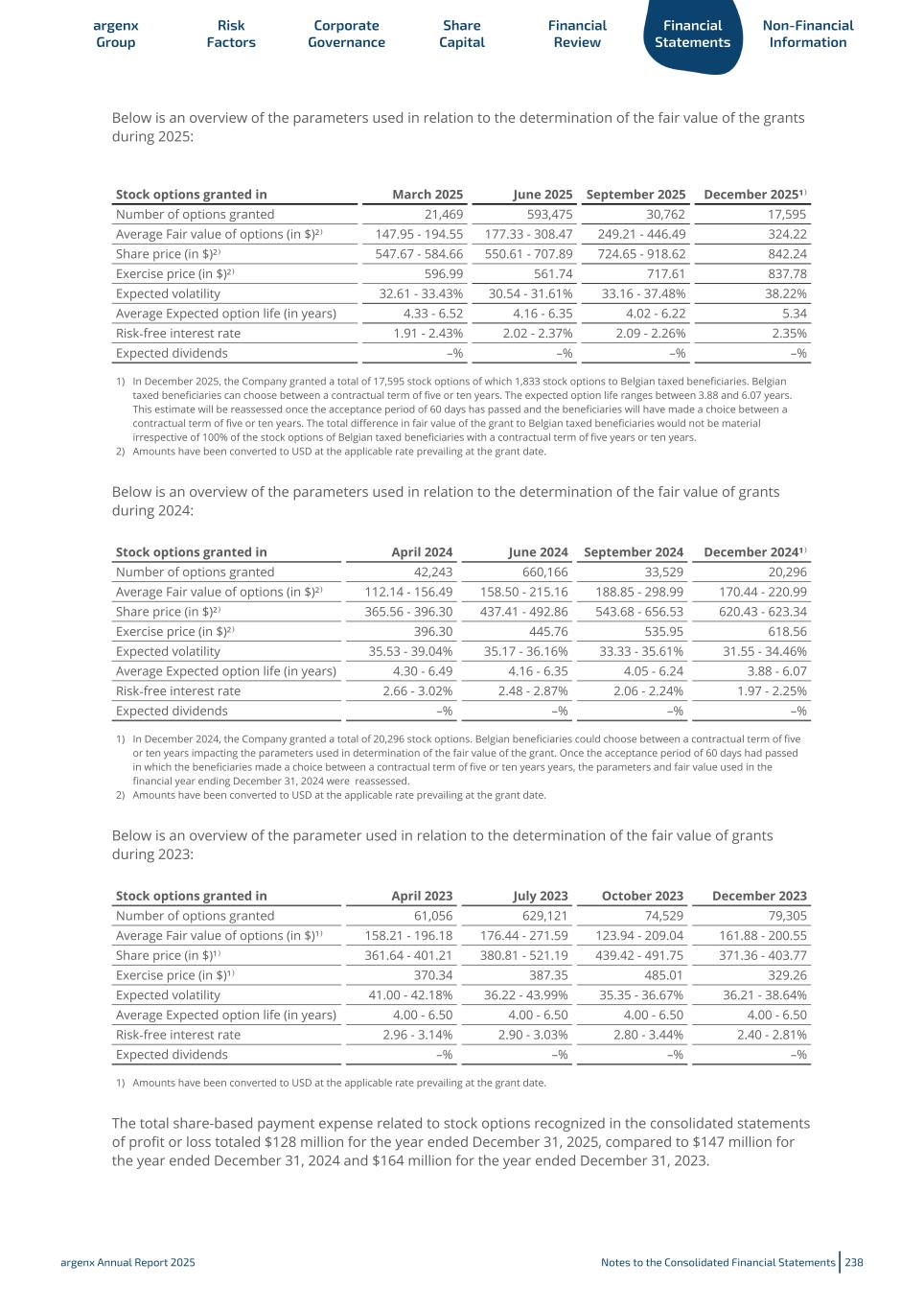
Below is an overview of the parameters used in relation to the determination of the fair value of the grants during 2025: Stock options granted in March 2025 June 2025 September 2025 December 2025¹⁾ Number of options granted 21,469 593,475 30,762 17,595 Average Fair value of options (in $)²⁾ 147.95 - 194.55 177.33 - 308.47 249.21 - 446.49 324.22 Share price (in $)²⁾ 547.67 - 584.66 550.61 - 707.89 724.65 - 918.62 842.24 Exercise price (in $)²⁾ 596.99 561.74 717.61 837.78 Expected volatility 32.61 - 33.43% 30.54 - 31.61% 33.16 - 37.48% 38.22% Average Expected option life (in years) 4.33 - 6.52 4.16 - 6.35 4.02 - 6.22 5.34 Risk-free interest rate 1.91 - 2.43% 2.02 - 2.37% 2.09 - 2.26% 2.35% Expected dividends –% –% –% –% 1) In December 2025, the Company granted a total of 17,595 stock options of which 1,833 stock options to Belgian taxed beneficiaries. Belgian taxed beneficiaries can choose between a contractual term of five or ten years. The expected option life ranges between 3.88 and 6.07 years. This estimate will be reassessed once the acceptance period of 60 days has passed and the beneficiaries will have made a choice between a contractual term of five or ten years. The total difference in fair value of the grant to Belgian taxed beneficiaries would not be material irrespective of 100% of the stock options of Belgian taxed beneficiaries with a contractual term of five years or ten years. 2) Amounts have been converted to USD at the applicable rate prevailing at the grant date. Below is an overview of the parameters used in relation to the determination of the fair value of grants during 2024: Stock options granted in April 2024 June 2024 September 2024 December 2024¹⁾ Number of options granted 42,243 660,166 33,529 20,296 Average Fair value of options (in $)²⁾ 112.14 - 156.49 158.50 - 215.16 188.85 - 298.99 170.44 - 220.99 Share price (in $)²⁾ 365.56 - 396.30 437.41 - 492.86 543.68 - 656.53 620.43 - 623.34 Exercise price (in $)²⁾ 396.30 445.76 535.95 618.56 Expected volatility 35.53 - 39.04% 35.17 - 36.16% 33.33 - 35.61% 31.55 - 34.46% Average Expected option life (in years) 4.30 - 6.49 4.16 - 6.35 4.05 - 6.24 3.88 - 6.07 Risk-free interest rate 2.66 - 3.02% 2.48 - 2.87% 2.06 - 2.24% 1.97 - 2.25% Expected dividends –% –% –% –% 1) In December 2024, the Company granted a total of 20,296 stock options. Belgian beneficiaries could choose between a contractual term of five or ten years impacting the parameters used in determination of the fair value of the grant. Once the acceptance period of 60 days had passed in which the beneficiaries made a choice between a contractual term of five or ten years years, the parameters and fair value used in the financial year ending December 31, 2024 were reassessed. 2) Amounts have been converted to USD at the applicable rate prevailing at the grant date. Below is an overview of the parameter used in relation to the determination of the fair value of grants during 2023: Stock options granted in April 2023 July 2023 October 2023 December 2023 Number of options granted 61,056 629,121 74,529 79,305 Average Fair value of options (in $)¹⁾ 158.21 - 196.18 176.44 - 271.59 123.94 - 209.04 161.88 - 200.55 Share price (in $)¹⁾ 361.64 - 401.21 380.81 - 521.19 439.42 - 491.75 371.36 - 403.77 Exercise price (in $)¹⁾ 370.34 387.35 485.01 329.26 Expected volatility 41.00 - 42.18% 36.22 - 43.99% 35.35 - 36.67% 36.21 - 38.64% Average Expected option life (in years) 4.00 - 6.50 4.00 - 6.50 4.00 - 6.50 4.00 - 6.50 Risk-free interest rate 2.96 - 3.14% 2.90 - 3.03% 2.80 - 3.44% 2.40 - 2.81% Expected dividends –% –% –% –% 1) Amounts have been converted to USD at the applicable rate prevailing at the grant date. The total share-based payment expense related to stock options recognized in the consolidated statements of profit or loss totaled $128 million for the year ended December 31, 2025, compared to $147 million for the year ended December 31, 2024 and $164 million for the year ended December 31, 2023. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Notes to the Consolidated Financial Statements 238
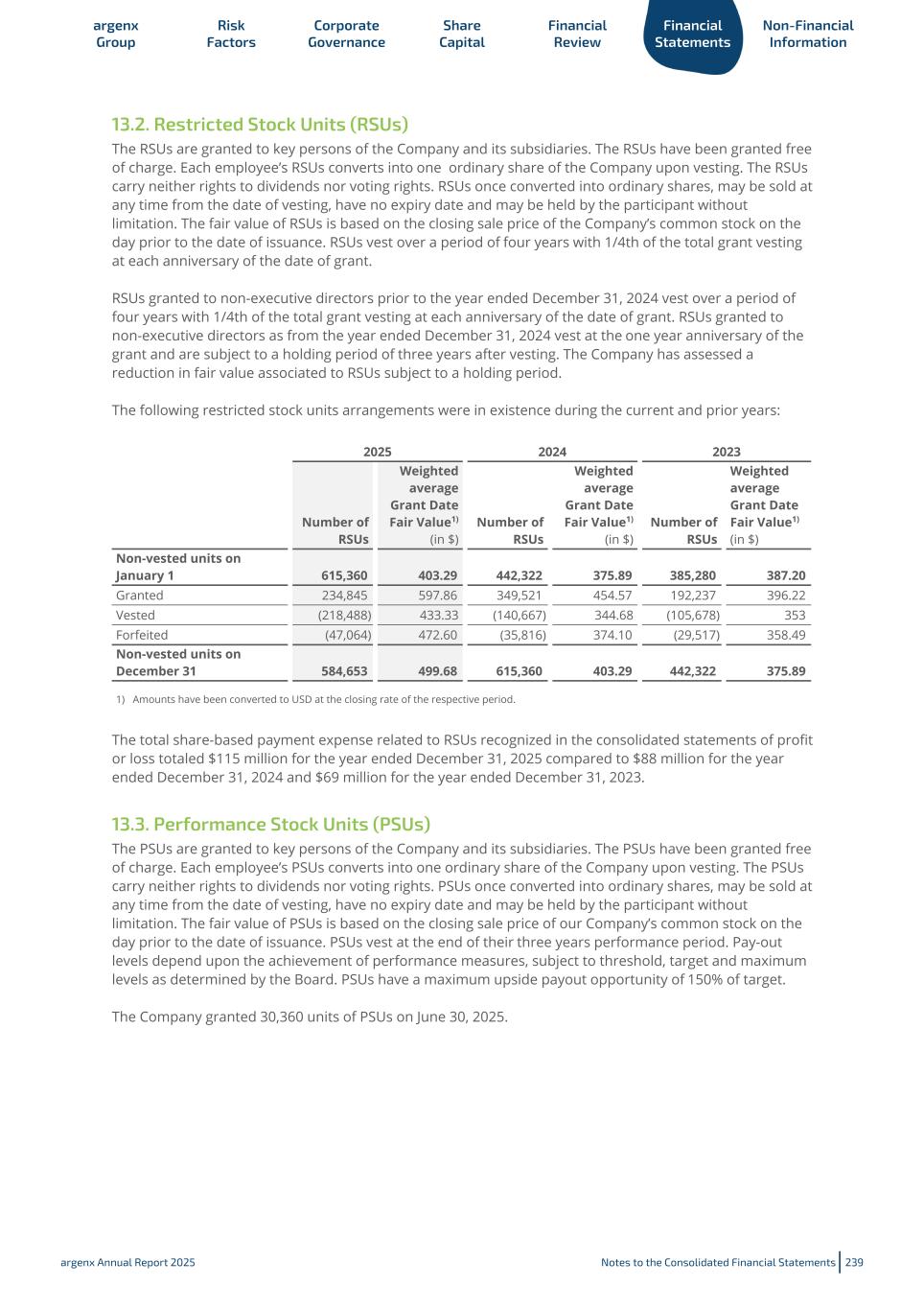
13.2. Restricted Stock Units (RSUs) The RSUs are granted to key persons of the Company and its subsidiaries. The RSUs have been granted free of charge. Each employee’s RSUs converts into one ordinary share of the Company upon vesting. The RSUs carry neither rights to dividends nor voting rights. RSUs once converted into ordinary shares, may be sold at any time from the date of vesting, have no expiry date and may be held by the participant without limitation. The fair value of RSUs is based on the closing sale price of the Company’s common stock on the day prior to the date of issuance. RSUs vest over a period of four years with 1/4th of the total grant vesting at each anniversary of the date of grant. RSUs granted to non-executive directors prior to the year ended December 31, 2024 vest over a period of four years with 1/4th of the total grant vesting at each anniversary of the date of grant. RSUs granted to non-executive directors as from the year ended December 31, 2024 vest at the one year anniversary of the grant and are subject to a holding period of three years after vesting. The Company has assessed a reduction in fair value associated to RSUs subject to a holding period. The following restricted stock units arrangements were in existence during the current and prior years: 2025 2024 2023 Number of RSUs Weighted average Grant Date Fair Value1) (in $) Number of RSUs Weighted average Grant Date Fair Value1) (in $) Number of RSUs Weighted average Grant Date Fair Value1) (in $) Non-vested units on January 1 615,360 403.29 442,322 375.89 385,280 387.20 Granted 234,845 597.86 349,521 454.57 192,237 396.22 Vested (218,488) 433.33 (140,667) 344.68 (105,678) 353 Forfeited (47,064) 472.60 (35,816) 374.10 (29,517) 358.49 Non-vested units on December 31 584,653 499.68 615,360 403.29 442,322 375.89 1) Amounts have been converted to USD at the closing rate of the respective period. The total share-based payment expense related to RSUs recognized in the consolidated statements of profit or loss totaled $115 million for the year ended December 31, 2025 compared to $88 million for the year ended December 31, 2024 and $69 million for the year ended December 31, 2023. 13.3. Performance Stock Units (PSUs) The PSUs are granted to key persons of the Company and its subsidiaries. The PSUs have been granted free of charge. Each employee’s PSUs converts into one ordinary share of the Company upon vesting. The PSUs carry neither rights to dividends nor voting rights. PSUs once converted into ordinary shares, may be sold at any time from the date of vesting, have no expiry date and may be held by the participant without limitation. The fair value of PSUs is based on the closing sale price of our Company’s common stock on the day prior to the date of issuance. PSUs vest at the end of their three years performance period. Pay-out levels depend upon the achievement of performance measures, subject to threshold, target and maximum levels as determined by the Board. PSUs have a maximum upside payout opportunity of 150% of target. The Company granted 30,360 units of PSUs on June 30, 2025. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Notes to the Consolidated Financial Statements 239

2025 Number of PSUs Weighted average Grant Date Fair Value1) (in $) Non-vested units on January 1 – – Granted 30,360 563.18 Vested – – Forfeited – – Non-vested units on December 31 30,360 563.18 1) Amounts have been converted to USD at the closing rate of the respective period. This was the first grant of PSUs by the Company, there are therefore no comparable periods. The total share-based payment expense related to PSUs recognized in the consolidated statements of profit or loss totaled $6 million for the year ended December 31, 2025. 14. Trade and Other Payables As of December 31, (in thousands of $) 2025 2024 2023 Trade payables 554,268 342,228 245,557 Sales rebates and reserves 402,032 140,474 55,788 Short-term employee benefits 212,344 150,818 95,104 Other 98,500 16,473 17,564 Total trade and other payables 1,267,144 649,993 414,013 The carrying amounts of trade and other payables approximate their respective fair values. Trade payables correspond primarily to R&D, commercial and manufacturing activities and include accrued expenses related to these activities. Short-term employee benefits include payables and accruals for salaries and bonuses to be paid to the employees of the Company. The following table summarizes the movement in the sales rebates and reserves for the year ended December 31, 2025, 2024 and 2023: argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Notes to the Consolidated Financial Statements 240

(in thousands of $) Rebates and chargebacks Distribution fees, product returns Total sales rebates and reserves Balance on January 1, 2023 15,398 4,079 19,478 Current estimate related to the sales made in the current year 123,542 26,427 149,969 Adjustment for prior periods (4,041) (883) (4,924) Credits or payments1) (85,237) (23,497) (108,734) Balance on December 31, 2023 49,662 6,126 55,788 Current estimate related to the sales made in the current year 285,863 50,239 336,102 Adjustment for prior periods (10,912) (162) (11,074) Credits or payments1) (197,202) (43,140) (240,342) Balance on December 31, 2024 127,411 13,063 140,474 Current estimate related to the sales made in the current period 824,251 132,171 956,422 Adjustment for prior periods (6,507) 2,038 (4,469) Credits or payments (583,779) (112,685) (696,464) Foreign currency translation differences 6,584 (515) 6,069 Balance on December 31, 2025 367,960 34,072 402,032 1) Comparative figures have been aligned to the presentation adopted in the current year. 15. Other Operating Income Year Ended December 31, (in thousands of $) 2025 20241) 20231) Research and development incentives 62,503 46,106 27,815 Payroll tax rebates 19,061 11,855 11,925 Collaboration revenue 2,166 4,348 35,533 Change in fair value on non-current financial assets 11,581 3,834 – Other operating income 1,423 13 2,538 Total other operating income 96,734 66,156 77,811 1) Comparative figures have been aligned with the presentation adopted in the current year. For the year ended December 31, 2025, the collaboration revenue was generated under the agreement with Zai Lab. This note should be read alongside ‘‘Note 2.18 Other operating income - Collaboration and license agreements’’. 15.1. Research and development incentives The Company has accounted for tax incentives following a research and development tax incentive scheme in Belgium according to which the incentive will be refunded after a five years period, if not offset against the current tax payable over the period. 15.2. Payroll tax rebates The Company accounted for payroll tax rebates as a reduction in withholding income taxes for its highly qualified personnel employed in its research and development department. 15.3. Collaboration revenue - AbbVie In April 2016, the Company entered into the AbbVie Collaboration Agreement to develop and commercialize ARGX-115 (ABBV-151). In October 2023, the Company achieved the second development milestone upon initiation of a non-pivotal clinical trial, triggering a $30 million payment. Subject to the continuing progress of ARGX-115 (ABBV-151) by AbbVie, the Company is eligible to receive future development, regulatory and commercial milestone payments in aggregate amounts of up to $50 million, $190 million and $325 million, respectively, as well as tiered royalties on sales at percentages ranging from the mid-single digits to the lower teens, subject to customary reductions. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Notes to the Consolidated Financial Statements 241
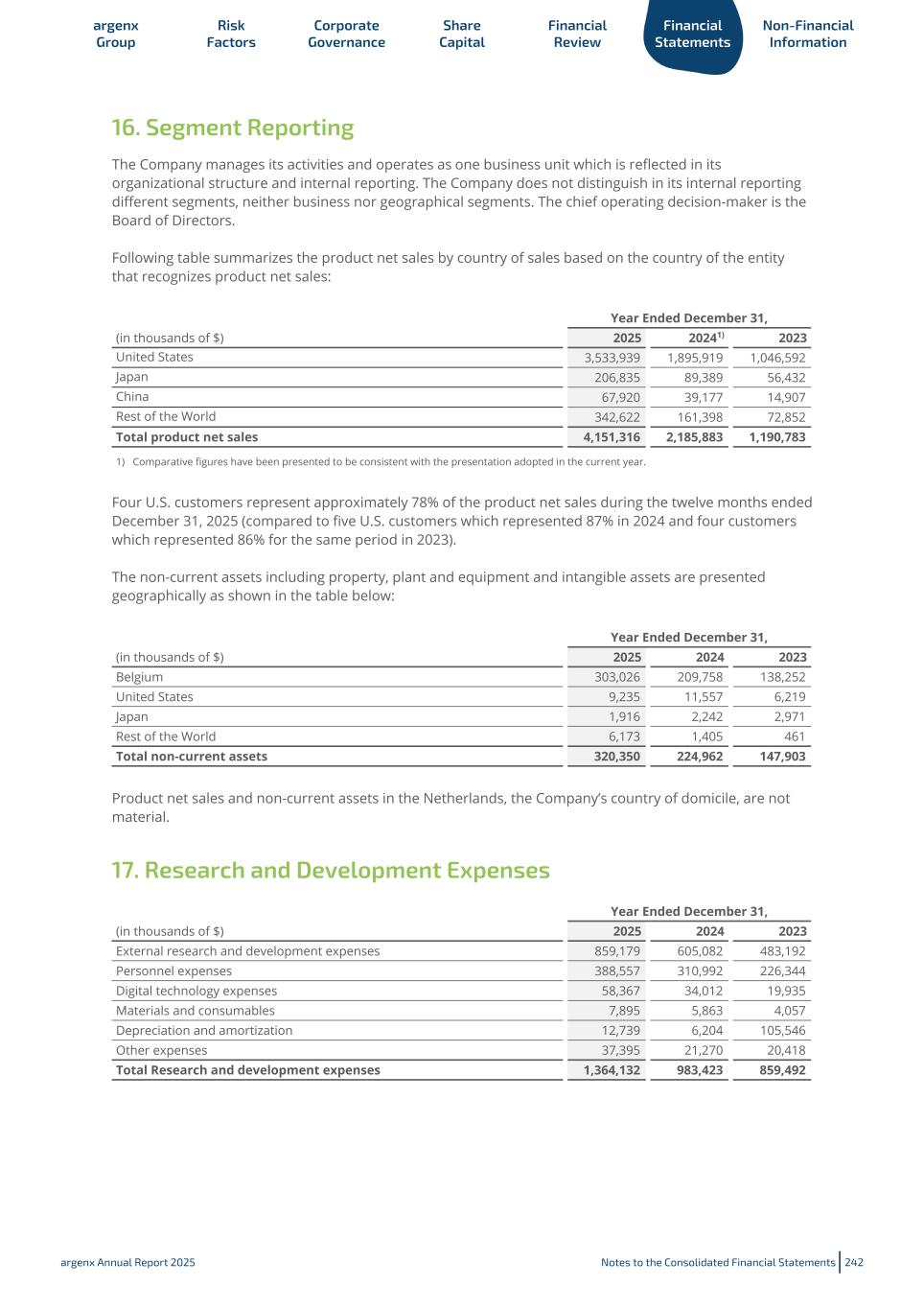
16. Segment Reporting The Company manages its activities and operates as one business unit which is reflected in its organizational structure and internal reporting. The Company does not distinguish in its internal reporting different segments, neither business nor geographical segments. The chief operating decision-maker is the Board of Directors. Following table summarizes the product net sales by country of sales based on the country of the entity that recognizes product net sales: Year Ended December 31, (in thousands of $) 2025 20241) 2023 United States 3,533,939 1,895,919 1,046,592 Japan 206,835 89,389 56,432 China 67,920 39,177 14,907 Rest of the World 342,622 161,398 72,852 Total product net sales 4,151,316 2,185,883 1,190,783 1) Comparative figures have been presented to be consistent with the presentation adopted in the current year. Four U.S. customers represent approximately 78% of the product net sales during the twelve months ended December 31, 2025 (compared to five U.S. customers which represented 87% in 2024 and four customers which represented 86% for the same period in 2023). The non-current assets including property, plant and equipment and intangible assets are presented geographically as shown in the table below: Year Ended December 31, (in thousands of $) 2025 2024 2023 Belgium 303,026 209,758 138,252 United States 9,235 11,557 6,219 Japan 1,916 2,242 2,971 Rest of the World 6,173 1,405 461 Total non-current assets 320,350 224,962 147,903 Product net sales and non-current assets in the Netherlands, the Company’s country of domicile, are not material. 17. Research and Development Expenses Year Ended December 31, (in thousands of $) 2025 2024 2023 External research and development expenses 859,179 605,082 483,192 Personnel expenses 388,557 310,992 226,344 Digital technology expenses 58,367 34,012 19,935 Materials and consumables 7,895 5,863 4,057 Depreciation and amortization 12,739 6,204 105,546 Other expenses 37,395 21,270 20,418 Total Research and development expenses 1,364,132 983,423 859,492 argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Notes to the Consolidated Financial Statements 242
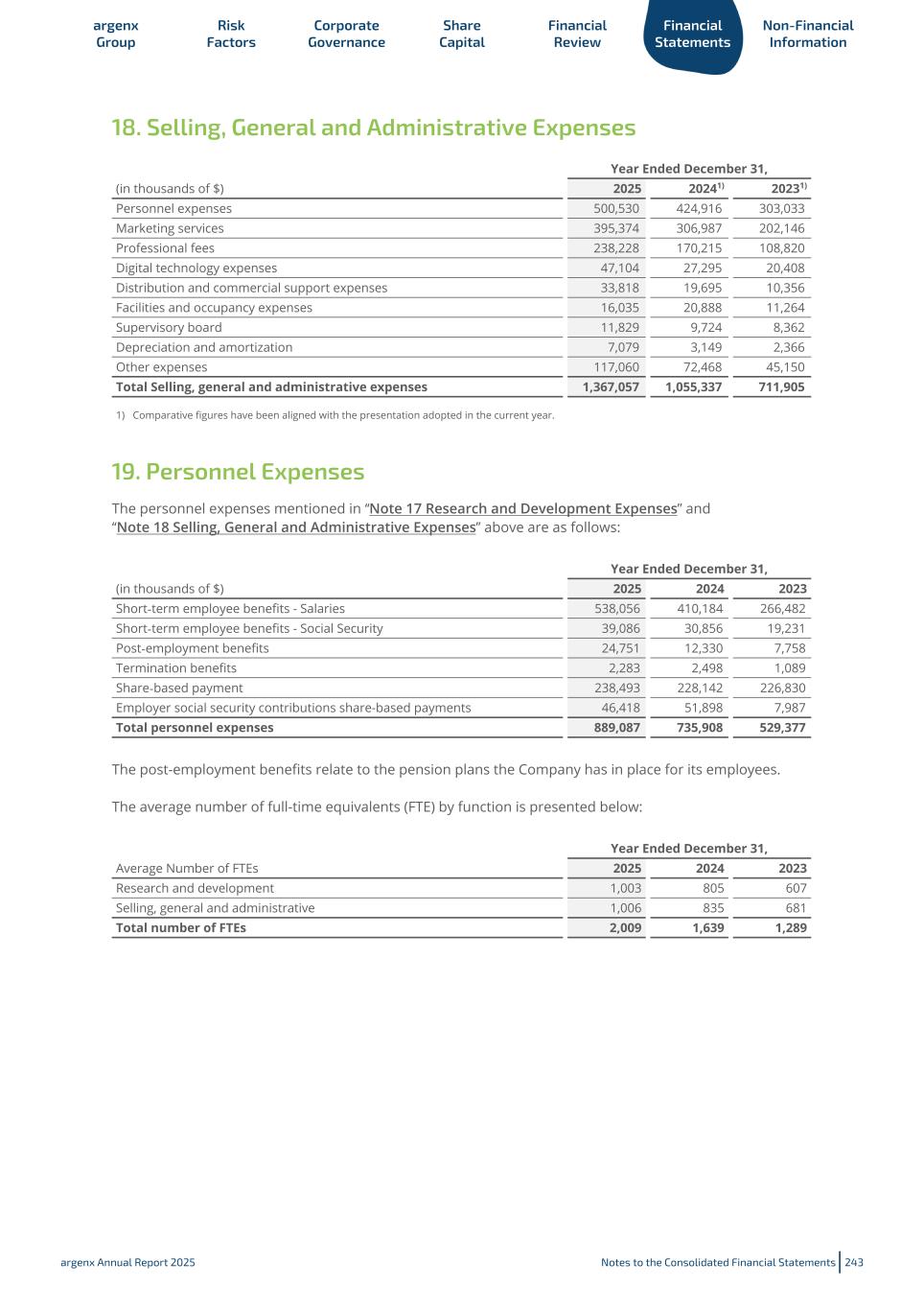
18. Selling, General and Administrative Expenses Year Ended December 31, (in thousands of $) 2025 20241) 20231) Personnel expenses 500,530 424,916 303,033 Marketing services 395,374 306,987 202,146 Professional fees 238,228 170,215 108,820 Digital technology expenses 47,104 27,295 20,408 Distribution and commercial support expenses 33,818 19,695 10,356 Facilities and occupancy expenses 16,035 20,888 11,264 Supervisory board 11,829 9,724 8,362 Depreciation and amortization 7,079 3,149 2,366 Other expenses 117,060 72,468 45,150 Total Selling, general and administrative expenses 1,367,057 1,055,337 711,905 1) Comparative figures have been aligned with the presentation adopted in the current year. 19. Personnel Expenses The personnel expenses mentioned in ‘‘Note 17 Research and Development Expenses” and ‘‘Note 18 Selling, General and Administrative Expenses’’ above are as follows: Year Ended December 31, (in thousands of $) 2025 2024 2023 Short-term employee benefits - Salaries 538,056 410,184 266,482 Short-term employee benefits - Social Security 39,086 30,856 19,231 Post-employment benefits 24,751 12,330 7,758 Termination benefits 2,283 2,498 1,089 Share-based payment 238,493 228,142 226,830 Employer social security contributions share-based payments 46,418 51,898 7,987 Total personnel expenses 889,087 735,908 529,377 The post-employment benefits relate to the pension plans the Company has in place for its employees. The average number of full-time equivalents (FTE) by function is presented below: Year Ended December 31, Average Number of FTEs 2025 2024 2023 Research and development 1,003 805 607 Selling, general and administrative 1,006 835 681 Total number of FTEs 2,009 1,639 1,289 argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Notes to the Consolidated Financial Statements 243
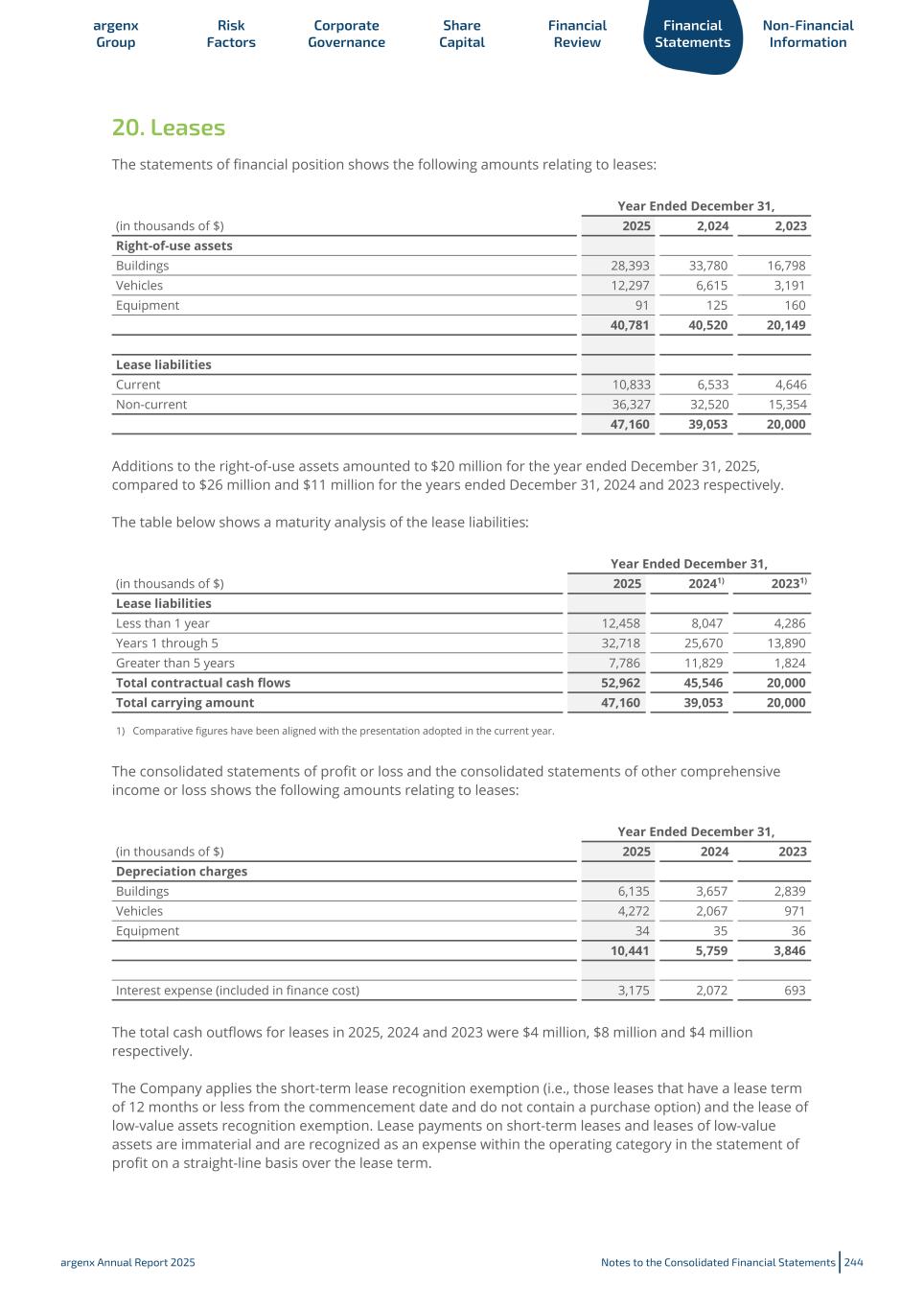
20. Leases The statements of financial position shows the following amounts relating to leases: Year Ended December 31, (in thousands of $) 2025 2,024 2,023 Right-of-use assets Buildings 28,393 33,780 16,798 Vehicles 12,297 6,615 3,191 Equipment 91 125 160 40,781 40,520 20,149 Lease liabilities Current 10,833 6,533 4,646 Non-current 36,327 32,520 15,354 47,160 39,053 20,000 Additions to the right-of-use assets amounted to $20 million for the year ended December 31, 2025, compared to $26 million and $11 million for the years ended December 31, 2024 and 2023 respectively. The table below shows a maturity analysis of the lease liabilities: Year Ended December 31, (in thousands of $) 2025 20241) 20231) Lease liabilities Less than 1 year 12,458 8,047 4,286 Years 1 through 5 32,718 25,670 13,890 Greater than 5 years 7,786 11,829 1,824 Total contractual cash flows 52,962 45,546 20,000 Total carrying amount 47,160 39,053 20,000 1) Comparative figures have been aligned with the presentation adopted in the current year. The consolidated statements of profit or loss and the consolidated statements of other comprehensive income or loss shows the following amounts relating to leases: Year Ended December 31, (in thousands of $) 2025 2024 2023 Depreciation charges Buildings 6,135 3,657 2,839 Vehicles 4,272 2,067 971 Equipment 34 35 36 10,441 5,759 3,846 Interest expense (included in finance cost) 3,175 2,072 693 The total cash outflows for leases in 2025, 2024 and 2023 were $4 million, $8 million and $4 million respectively. The Company applies the short-term lease recognition exemption (i.e., those leases that have a lease term of 12 months or less from the commencement date and do not contain a purchase option) and the lease of low-value assets recognition exemption. Lease payments on short-term leases and leases of low-value assets are immaterial and are recognized as an expense within the operating category in the statement of profit on a straight-line basis over the lease term. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Notes to the Consolidated Financial Statements 244
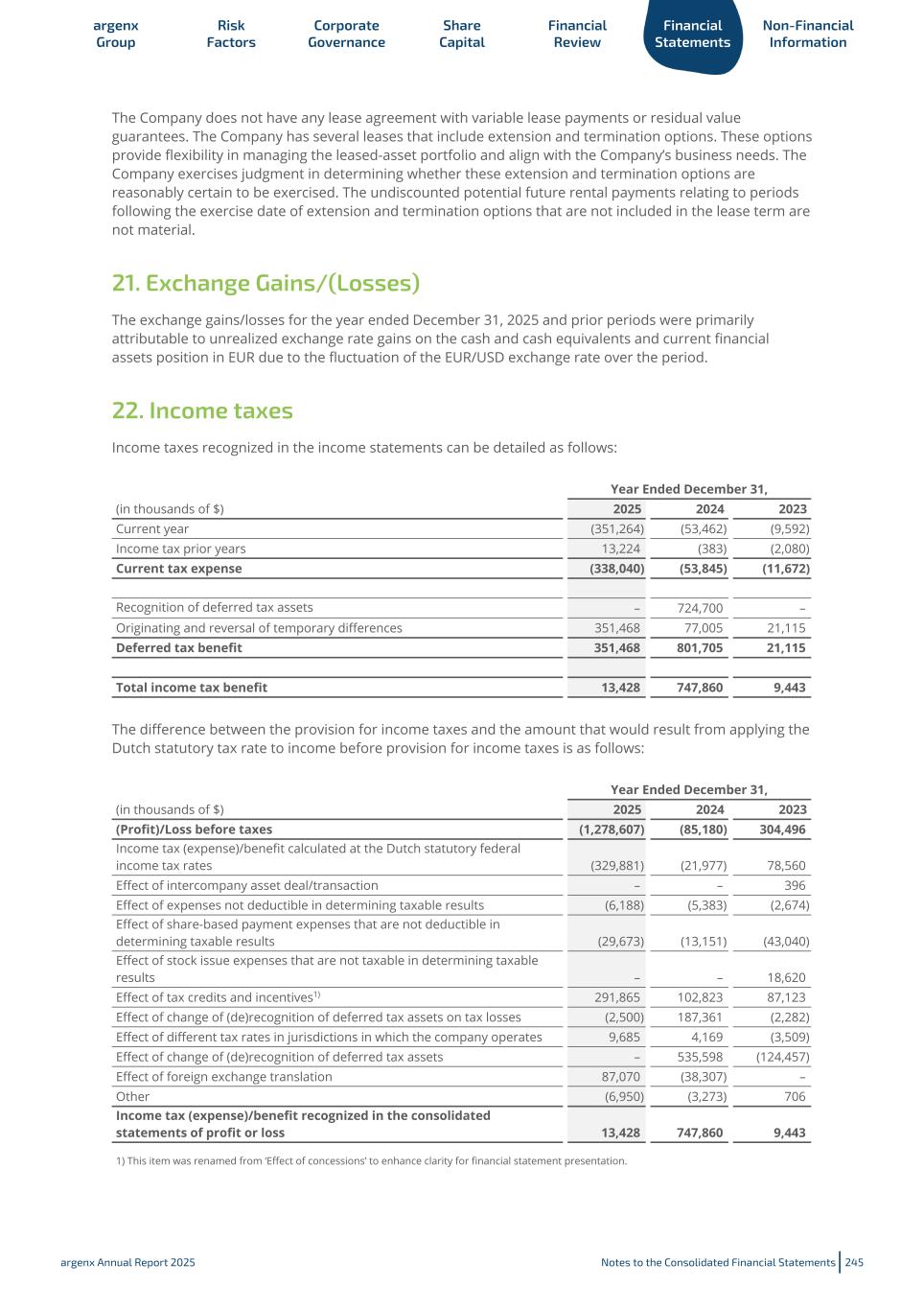
The Company does not have any lease agreement with variable lease payments or residual value guarantees. The Company has several leases that include extension and termination options. These options provide flexibility in managing the leased-asset portfolio and align with the Company’s business needs. The Company exercises judgment in determining whether these extension and termination options are reasonably certain to be exercised. The undiscounted potential future rental payments relating to periods following the exercise date of extension and termination options that are not included in the lease term are not material. 21. Exchange Gains/(Losses) The exchange gains/losses for the year ended December 31, 2025 and prior periods were primarily attributable to unrealized exchange rate gains on the cash and cash equivalents and current financial assets position in EUR due to the fluctuation of the EUR/USD exchange rate over the period. 22. Income taxes Income taxes recognized in the income statements can be detailed as follows: Year Ended December 31, (in thousands of $) 2025 2024 2023 Current year (351,264) (53,462) (9,592) Income tax prior years 13,224 (383) (2,080) Current tax expense (338,040) (53,845) (11,672) Recognition of deferred tax assets – 724,700 – Originating and reversal of temporary differences 351,468 77,005 21,115 Deferred tax benefit 351,468 801,705 21,115 Total income tax benefit 13,428 747,860 9,443 The difference between the provision for income taxes and the amount that would result from applying the Dutch statutory tax rate to income before provision for income taxes is as follows: Year Ended December 31, (in thousands of $) 2025 2024 2023 (Profit)/Loss before taxes (1,278,607) (85,180) 304,496 Income tax (expense)/benefit calculated at the Dutch statutory federal income tax rates (329,881) (21,977) 78,560 Effect of intercompany asset deal/transaction – – 396 Effect of expenses not deductible in determining taxable results (6,188) (5,383) (2,674) Effect of share-based payment expenses that are not deductible in determining taxable results (29,673) (13,151) (43,040) Effect of stock issue expenses that are not taxable in determining taxable results – – 18,620 Effect of tax credits and incentives1) 291,865 102,823 87,123 Effect of change of (de)recognition of deferred tax assets on tax losses (2,500) 187,361 (2,282) Effect of different tax rates in jurisdictions in which the company operates 9,685 4,169 (3,509) Effect of change of (de)recognition of deferred tax assets – 535,598 (124,457) Effect of foreign exchange translation 87,070 (38,307) – Other (6,950) (3,273) 706 Income tax (expense)/benefit recognized in the consolidated statements of profit or loss 13,428 747,860 9,443 1) This item was renamed from ‘Effect of concessions’ to enhance clarity for financial statement presentation. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Notes to the Consolidated Financial Statements 245
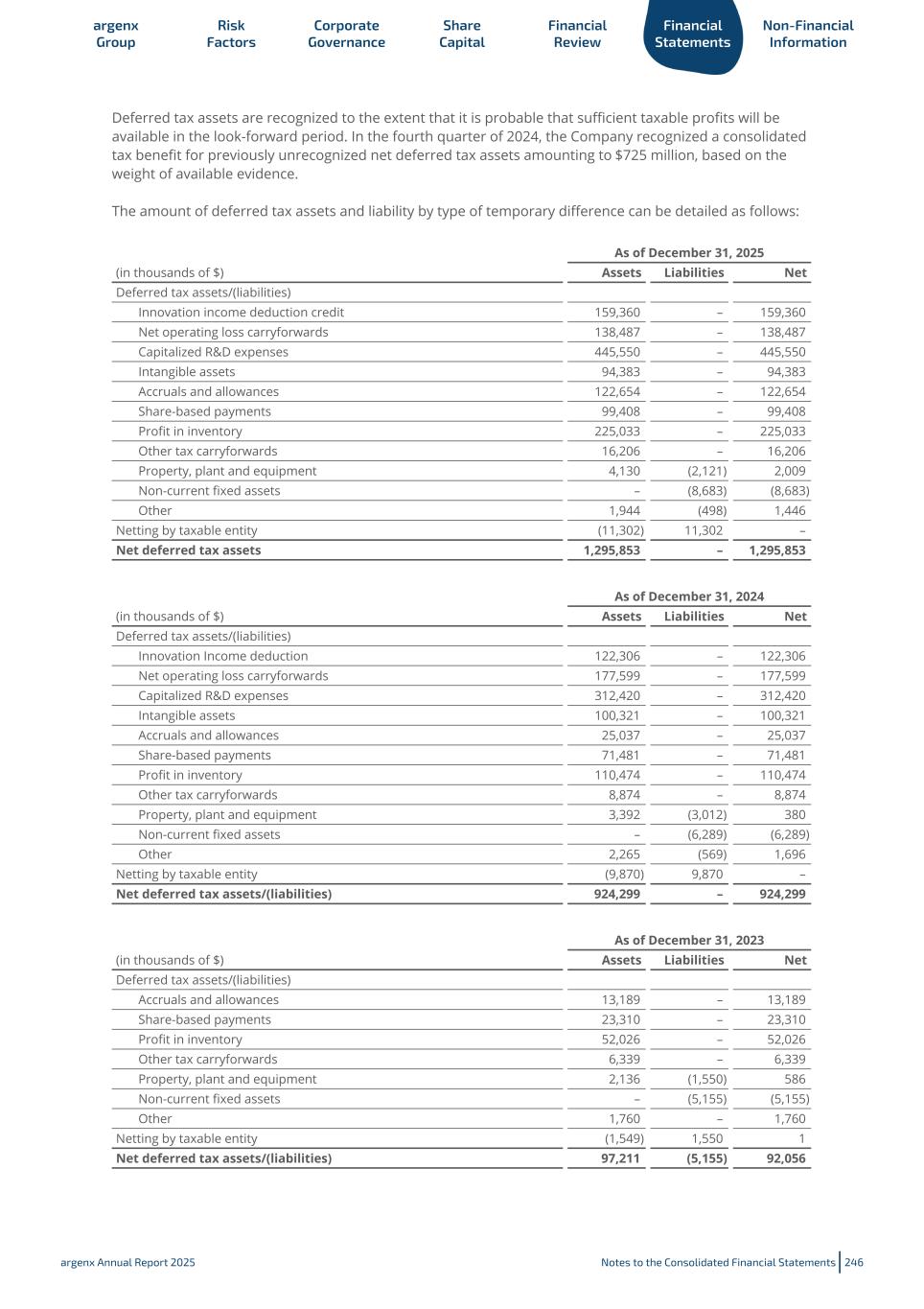
Deferred tax assets are recognized to the extent that it is probable that sufficient taxable profits will be available in the look-forward period. In the fourth quarter of 2024, the Company recognized a consolidated tax benefit for previously unrecognized net deferred tax assets amounting to $725 million, based on the weight of available evidence. The amount of deferred tax assets and liability by type of temporary difference can be detailed as follows: As of December 31, 2025 (in thousands of $) Assets Liabilities Net Deferred tax assets/(liabilities) Innovation income deduction credit 159,360 – 159,360 Net operating loss carryforwards 138,487 – 138,487 Capitalized R&D expenses 445,550 – 445,550 Intangible assets 94,383 – 94,383 Accruals and allowances 122,654 – 122,654 Share-based payments 99,408 – 99,408 Profit in inventory 225,033 – 225,033 Other tax carryforwards 16,206 – 16,206 Property, plant and equipment 4,130 (2,121) 2,009 Non-current fixed assets – (8,683) (8,683) Other 1,944 (498) 1,446 Netting by taxable entity (11,302) 11,302 – Net deferred tax assets 1,295,853 – 1,295,853 As of December 31, 2024 (in thousands of $) Assets Liabilities Net Deferred tax assets/(liabilities) Innovation Income deduction 122,306 – 122,306 Net operating loss carryforwards 177,599 – 177,599 Capitalized R&D expenses 312,420 – 312,420 Intangible assets 100,321 – 100,321 Accruals and allowances 25,037 – 25,037 Share-based payments 71,481 – 71,481 Profit in inventory 110,474 – 110,474 Other tax carryforwards 8,874 – 8,874 Property, plant and equipment 3,392 (3,012) 380 Non-current fixed assets – (6,289) (6,289) Other 2,265 (569) 1,696 Netting by taxable entity (9,870) 9,870 – Net deferred tax assets/(liabilities) 924,299 – 924,299 As of December 31, 2023 (in thousands of $) Assets Liabilities Net Deferred tax assets/(liabilities) Accruals and allowances 13,189 – 13,189 Share-based payments 23,310 – 23,310 Profit in inventory 52,026 – 52,026 Other tax carryforwards 6,339 – 6,339 Property, plant and equipment 2,136 (1,550) 586 Non-current fixed assets – (5,155) (5,155) Other 1,760 – 1,760 Netting by taxable entity (1,549) 1,550 1 Net deferred tax assets/(liabilities) 97,211 (5,155) 92,056 argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Notes to the Consolidated Financial Statements 246
The change in net deferred taxes recorded in the consolidated statements of financial position can be detailed as follows: (in thousands of $) Deferred tax assets Deferred tax liabilities Balance on January 1, 2025 924,299 – Recognized in profit or loss 264,021 – Recognized in equity 20,780 – Effects of change in foreign exchange rate 86,753 – Balance on December 31, 2025 1,295,853 – (in thousands of $) Deferred tax assets Deferred tax liabilities Balance on January 1, 2024 97,211 (5,155) Recognized in profit or loss 758,264 5,155 Recognized in equity 30,846 – Effects of change in foreign exchange rate 37,978 – Balance on December 31, 2024 924,299 – (in thousands of $) Deferred tax assets Deferred tax liabilities Balance on January 1, 2023 79,222 (8,406) Recognized in profit or loss 17,685 3,430 Recognized in equity 381 – Effects of change in foreign exchange rate (77) (179) Balance on December 31, 2023 97,211 (5,155) The Company also has unrecognized tax losses carried forward in the Netherlands in the amount of $56 million as of December 31, 2025, compared to $46 million on December 31, 2024 and $33 million on December 31, 2023. These losses carried forward do not have an expiration date based upon the applicable enacted tax legislation in the Netherlands. As of December 31, 2025, the Company has $209 million of undistributed earnings attributable to foreign subsidiaries (compared to $125 million on December 31, 2024 and $128 million on December 31, 2023) for which no provision for deferred tax liabilities have been recognized because the Company has control over the timing of the reversal of the temporary differences and there are no plans of distributions in the foreseeable future. The Company is subject to the OECD Pillar Two Directive and implementing domestic laws in 2025. The Pillar Two Rules does not have a material impact on our effective tax rate or the recognition of our deferred tax assets. The Company continues to apply the exception relating to recognizing and disclosing information about deferred tax assets and liabilities related to legislation that is enacted to implement the OECD Pillar Two model rules. 23. Earnings per Share Year Ended December 31 (in thousands of $ except for shares and EPS) 2025 2024 2023 Profit/(Loss) for the period 1,292,035 833,040 (295,053) Weighted average number of shares used for basic profit/(loss) per share 61,295,149 59,855,585 57,169,253 Basic profit/(loss) per share (in $) 21.08 13.92 (5.16) Weighted average number of shares used for diluted profit/(loss) per share 66,029,215 65,177,815 57,169,253 Diluted profit/(loss) per share (in $) 19.57 12.78 (5.16) argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Notes to the Consolidated Financial Statements 247
Profit/(loss) per ordinary share is calculated by dividing the profit/(loss) for the period by the weighted average number of ordinary shares during the year. Diluted profit/(loss) per share is calculated by adjusting the weighted average number of shares by in the money outstanding dilutive stock options, RSUs and PSUs. As the Company reported a net loss in 2023, stock options and RSUs had an anti-dilutive effect rather than a dilutive effect. As such, there is no difference between basic and diluted loss per ordinary share for this period. 24. Financial Risk Management The financial risks are managed centrally. The Company coordinates the access to national and international financial markets and considers and manages continuously the financial risks concerning the Company’s activities. These relate to credit risk, liquidity risk, interest rate risk and currency risk. The Company does not buy or trade financial instruments for speculative purposes. Categories of financial assets and liabilities: Measurement category Carrying amount on December 31 (in thousands of $) 2025 2024 2023 Financial assets - non-current FVTPL 37,130 25,549 21,715 Financial assets - non-current FVTOCI 10,022 14,880 15,528 Research and development incentive receivables - non- current Amortized cost 86,212 94,854 76,706 Restricted assets - non-current Amortized cost 4,838 1,964 2,419 Trade and other receivables Amortized cost 1,646,692 904,471 496,687 Financial assets - current Amortized cost 948,750 1,878,890 1,131,000 Research and development incentive receivables - current Amortized cost 10,367 4,625 2,584 Cash and bank balances Amortized cost 5,176 5,527 20,744 Cash equivalents FVTPL 2,541,112 1,394,409 1,678,100 Cash equivalents Amortized cost 945,001 100,000 350,000 Trade and other payables Amortized cost 1,267,144 649,993 414,013 The carrying amounts of research and development incentive receivables, financial assets, trade and other receivables, and trade and other payables are considered to be the same as their fair values, due to their short-term nature. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Notes to the Consolidated Financial Statements 248
Financial assets held at fair value through profit or loss or OCI Financial assets held at fair value through profit or loss or OCI consisted of equity instruments of listed and non-listed companies and money market funds. The Company has no restrictions on the sale of these equity instruments and the assets are not pledged under any of its liabilities. These instruments are classified as financial assets held at fair value through profit or loss or OCI which qualify for: • Level 1 fair value measurement with respect to current financial assets and cash equivalents based upon the closing price (net asset value) of such securities at each reporting date. • Level 3 fair value measurement with respect to non-current financial assets. The market price of these financial instruments might face fluctuations and might be affected by a variety of factors, such as the global economic situation. Current financial assets and cash equivalents include collective investment funds denominated in € and $ of which the underlying investments include bonds and other international debt securities. Based on the weighted average maturity of the underlying instruments, amongst others, these investments are either classified as current financial assets or cash equivalents. The maximum exposure to credit risk is the carrying amount at reporting date. The Company carried the following assets at fair value on December 31, 2025, 2024 and 2023 respectively: As of December 31, 2025 (in thousands of $) Level 1 Level 2 Level 3 Non-current financial assets 10,022 – 37,130 Cash and cash equivalents 2,541,112 – – Assets carried at fair value 2,551,134 – 37,130 As of December 31, 2024 (in thousands of $) Level 1 Level 2 Level 3 Non-current financial assets 14,880 – 25,549 Cash and cash equivalents 1,394,409 – – Assets carried at fair value 1,409,289 – 25,549 As of December 31, 2023 (in thousands of $) Level 1 Level 2 Level 3 Non-current financial assets 15,528 – 21,715 Cash and cash equivalents 1,678,100 – – Assets carried at fair value 1,693,628 – 21,715 During the disclosed calendar year, no transfers occurred between the applicable categories. Non-current financial assets – Level 1 The Company owns shares of Zai Lab due to its license and collaboration agreement. The fair value shares of the equity instrument at period-end is determined by reference to the closing price of such securities at each reporting date (classified as level 1 in the fair value hierarchy), resulting in a change in fair value. The Company made the irrevocable election to recognize subsequent changes in fair value through OCI. Non-current financial assets – Level 3 The Company has a profit share in AgomAb Therapeutics NV which is a non-publicly listed company valued using certain unobservable inputs and assumptions. The changes in the value of these investments are detailed in ‘‘Note 6 Other Non-Current Assets”. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Notes to the Consolidated Financial Statements 249

Capital risk The Company manages its capital to ensure that it will be able to continue as a going concern. The capital structure of the Company consists of equity attributed to the holders of equity instruments of the Company, such as capital, reserves and accumulated losses as mentioned in the consolidated statements of changes in equity. The Company makes the necessary adjustments in light of changes in the economic circumstances, risks associated to the different assets and the projected cash needs of the current and projected research activities. On December 31, 2025, cash and cash equivalents amounted to $3.5 billion, current financial assets amounted to $0.9 billion and total capital amounted to $7.3 billion. The current cash situation and the anticipated cash generation and usage are the most important parameters in assessing the capital structure. The Company’s objective is to maintain the capital structure at a level to be able to finance its activities for at least twelve months. Cash income from operations is taken into account and, if needed and possible, the Company can enter into financing agreements or issue new shares. Credit risk Credit risk refers to the risk that a counterparty will default on its contractual obligations resulting in financial loss to the Company. The Company has adopted a policy of only dealing with creditworthy counterparties and obtaining sufficient collateral, where appropriate, as a means of mitigating the risk of financial loss from defaults. Concentrations in credit risk are determined based on an analysis of counterparties and their importance on the overall outstanding contractual obligations at year-end. The Company's commercial revenue are concentrated as discussed in “Note 16 Segment Reporting”, on a limited number of U.S. customers with high quality creditworthiness. The Company sets customer specific credit limits in order to reduce credit risk from commercial payors. The Company applied the IFRS 9 simplified approach to measuring expected credit losses which uses a lifetime expected loss allowance for trade receivables. To measure the expected credit losses, receivables have been grouped based on credit risk characteristics and the days past due. The provision for expected credit losses was not significant given that there have been no significant credit losses over the last three years and the high quality nature of the Company’s customers. Cash and cash equivalents and current financial assets are invested with several highly reputable banks and financial institutions. The main purpose of the Cash Investment Policy is to preserve the available cash and to ensure sufficient short-term liquidity at all times. Therefore, the Company holds its cash and cash equivalents, in addition to current financial assets mainly with banks which are independently rated A- or higher. Amounts of cash held with banks rated lower than A- are limited to insignificant balances. The maximum amount and tenor of term accounts depends on the rating of the counterparty bank. The Company also holds cash equivalents in the form of money market funds with a low historical volatility. These money market funds are highly liquid investments and can be readily convertible into a known amount of cash. The company has adopted a policy whereby money market funds must have a minimum rating of A, and whereby 95% of its money market funds should have a AAA-rating. Liquidity risk The Company manages liquidity risk by maintaining adequate reserves, by continuously monitoring forecast and actual cash flows, and by matching the maturity profile of financial assets and liabilities. The Company’s main sources of cash are the sale of commercial product and exercise of stock options. This cash is invested in savings accounts, term accounts and money market funds. These money market funds represent the majority of the Company’s available sources of liquidity. Since all of these are immediately tradable and convertible in cash they have an important mitigating effect on any short-term liquidity risk. As of December 31, 2025, the Company had lines of credit totaling $29 million with financial institutions mainly relating to leasing guarantees. Interest rate risk The only variable interest-bearing financial instruments are cash and cash equivalents and current financial assets. Changes in interest rates may cause variations in interest income resulting from short-term interest- bearing assets. Lower short-term interests may have a negative impact on the interest income of the Company. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Notes to the Consolidated Financial Statements 250

For the year ended December 31, 2025, if applicable interest rates would increase/decrease by 50 basis points, this would have a positive/negative impact of $22 million (compared to $8 million for the year ended December 31, 2024 and $8 million for the year ended December 31, 2023 if applicable interest rates would increase/decrease by 25 basis points). Foreign exchange risk The Company undertakes transactions denominated in foreign currencies, causing exposures to exchange rate fluctuations. The Company is mainly exposed to the Euro, Japanese yen, British pound and Swiss franc. To limit this risk, the Company attempts to align incoming and outgoing cash flows in currencies other than USD. The Company further limits its non-USD liquidity holdings when possible. The net exposure to exchange differences of the monetary assets (being from cash and cash equivalents, in addition to current financial assets) of the Company at the end of the reporting period are as follows: As of December 31, (in thousands of $) 2025 2024 2023 EUR 155,757 756,676 923,773 Other currencies 1,258 1,679 8,708 On December 31, 2025, if the EUR would have strengthened/weakened versus the USD by 10%, this would have had a negative/positive impact of $16 million, compared to $76 million and $92 million on December 31, 2024 and December 31, 2023, respectively.If other currencies would have strengthen or weakened against the USD by 10%, this would have had no significant impact in all reported periods. 25. Related Party Transactions 25.1. Relationship and transactions with joint venture entity In 2022, the University of Colorado Anschutz Medical Campus and the University of Colorado Health (UCHealth) created an asset-centric spin-off, OncoVerity, Inc (OncoVerity), focused on optimizing and advancing the development of cusatuzumab, a novel anti-CD70 antibody, in AML. OncoVerity is an entity of co-creation, combining the extensive translational biology insights from Dr. Clayton Smith, M.D. from the University of Colorado with our experience on the CD70/CD27 pathway. argenx contributed $7 million in 2025 ($7 million and $13 million in 2024 and 2023 respectively). The investment has been accounted under IAS 28 Investment in associates and Joint Ventures using the equity method of accounting and has been designated as an “Investment in a joint venture” in the consolidated statements of financial position. The share of net loss resulting from investment in joint ventures is presented in consolidated statements of profit or loss as “Loss from investment in a joint venture”. The cash contributions made by the Company to the Joint Venture is reported under Cash flow from investing activities under “Investment in a joint venture”. 25.2. Relationship and transactions with subsidiaries See ‘‘Note 29 Overview of Consolidation Scope’’ for an overview of the consolidated companies of the group, which are all wholly-owned subsidiaries of argenx SE. Balances and transactions between the Company and its subsidiaries, which are related parties of the Company, have been eliminated on consolidation and are not disclosed in this note. 25.3. Relationship and transactions with key personnel The Company’s key management personnel consists of the members of the management team and the members of the board of directors. Remuneration of key management personnel On December 31, 2025, the Senior Management Team consisted of eight members: Chief Executive Officer, Chief Operating Officer, Chief Financial Officer, Chief Scientific Officer, General Counsel, Chief Medical argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Notes to the Consolidated Financial Statements 251
Officer, Vice President Corporate Development and Strategy and Global Head of Quality Assurance. They provide their services on a full-time basis. On December 31, 2025, the board of directors consisted of eight Non-Executive Directors: Peter Verhaeghe, Pamela Klein, Anthony Rosenberg, James Daly, Camilla Sylvest, Ana Céspedes, Steve Krognes, Brian Kotzin, and one executive director, Tim Van Hauwermeiren. Only the CEO is a member of both the Senior Management Team and the Board of Directors. The CEO does not receive any remuneration for his membership of the Board of Directors, as this is part of his total remuneration package in his capacity as member of the Senior Management Team. The remuneration package of the members of key management personnel comprises: Year Ended December 31, (in thousands of $, except for the number of stock options & RSUs) 2025 2024 2023 Remuneration of key management personnel Short-term benefits for the Senior Management Team Gross salary 5,285 4,529 4,161 Variable pay 3,276 3,084 2,816 Employer social security 2,497 1,473 807 Other short term benefits 497 672 545 Post-employment benefits for the Senior Management Team 327 274 167 Cost of stock options granted in the year for the Senior Management Team 16,819 17,758 27,983 Cost of restricted stock units granted in the year for the Senior Management Team – 16,211 11,694 Cost of performance stock units granted in the year for the Senior Management Team 13,899 – – Employer social security cost related to stock options 1,432 2,825 (494) Total benefits for key management personnel 44,032 46,826 47,679 Numbers of stock options granted in the year Senior Management Team 73,091 98,306 132,100 Numbers of restricted stock units granted in the year Senior Management Team – 36,365 30,425 Numbers of performance stock units granted in the year Senior Management Team 24,742 – – Remuneration of Non-Executive Directors Board fees and other short-term benefits for Non-Executive Directors 774 731 533 Cost of stock options granted in the year for Non-Executive Directors – – 2,280 Cost of restricted stock units granted in the year for Non-Executive Directors 3,159 4,511 1,034 Total benefits for Non-Executive Directors 3,933 5,242 3,846 Numbers of stock options granted in the year Non-Executive Directors – – 12,400 Numbers of restricted stock units granted in the year Non-Executive Directors 5,624 10,118 2,713 Other No loans, quasi-loans or other guarantees were given by the Company or any of its subsidiaries to members of the Senior Management Team or the Board of Directors. We have not entered into transactions with the Company’s key management personnel, other than as described above with respect to remuneration arrangements relating to the exercise of their mandates as members of the Senior Management Team and the Board of Directors. 26. Contingencies The Company is currently not facing any outstanding claims or litigation that may have a significant adverse impact on the Company’s consolidated financial position. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Notes to the Consolidated Financial Statements 252

27. Commitments In February 2019, the Company entered into a global collaboration and license agreement with Halozyme Therapeutics, which was later amended in September 2020 and again in September 2024. Under the terms of the agreement, the Company will pay up to $40 million to achievement of specific regulatory and sales-based milestones related specifically to its FcRn target. This amount represents the maximum amount that would be paid if all milestones would be achieved but excludes variable royalty payments based on unit sales. Further, the Company will pay up to $78 million per other non-FcRn target subject to achievement of specified development, regulatory and sales-based milestones. This amount represents the maximum amount that would be paid per target if all milestones would be achieved but excludes variable royalty payments based on unit sales. The Company has a total of six nominated targets under this agreement including its FcRn target. The Company’s commercial supply is manufactured in collaboration with Lonza and Fujifilm. In the aggregate, the Company has outstanding commitments for efgartigimod under these commercial supply agreements amounting to approximately $1.3 billion. These agreements provide commercial supply of efgartigimod to the Company’s global commercial operations through facilities in the U.S., Europe and Asia. 28. Audit Fees The following auditors’ fees were expensed in the consolidated statements of profit or loss and the consolidated statements of other comprehensive income or loss: Year Ended December 31, (in thousands of $) 20251) 20242) 20232) Audit fees 2,633 2,657 1,979 Audit-related fees 705 597 330 Total 3,338 3,254 2,309 1) Audit fees reported are audit services performed by EY Accountants B.V. as the external auditor in 2025 referred to in Section 1 of the Dutch Accounting Firms Oversight Act (Wta) as well as by the EY network. In 2025, audit and audit related fees of EY Accountants B.V. (excluding its member firms and/or affiliates) amounted to $973 and $677, respectively. 2) Audit fees reported are audit services performed by Deloitte Accountants B.V. as the external auditor in 2024 and 2023 referred to in Section 1 of the Dutch Accounting Firms Oversight Act (Wta) as well as by the Deloitte network. At the meeting held on May 7, 2024, the Company’s general assembly of shareholders appointed EY Accountants B.V. as external auditor for the financial year ending December 31, 2025. Deloitte Accountants B.V. completed its mandate as external auditor as of the financial year ending December 31, 2024. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Notes to the Consolidated Financial Statements 253
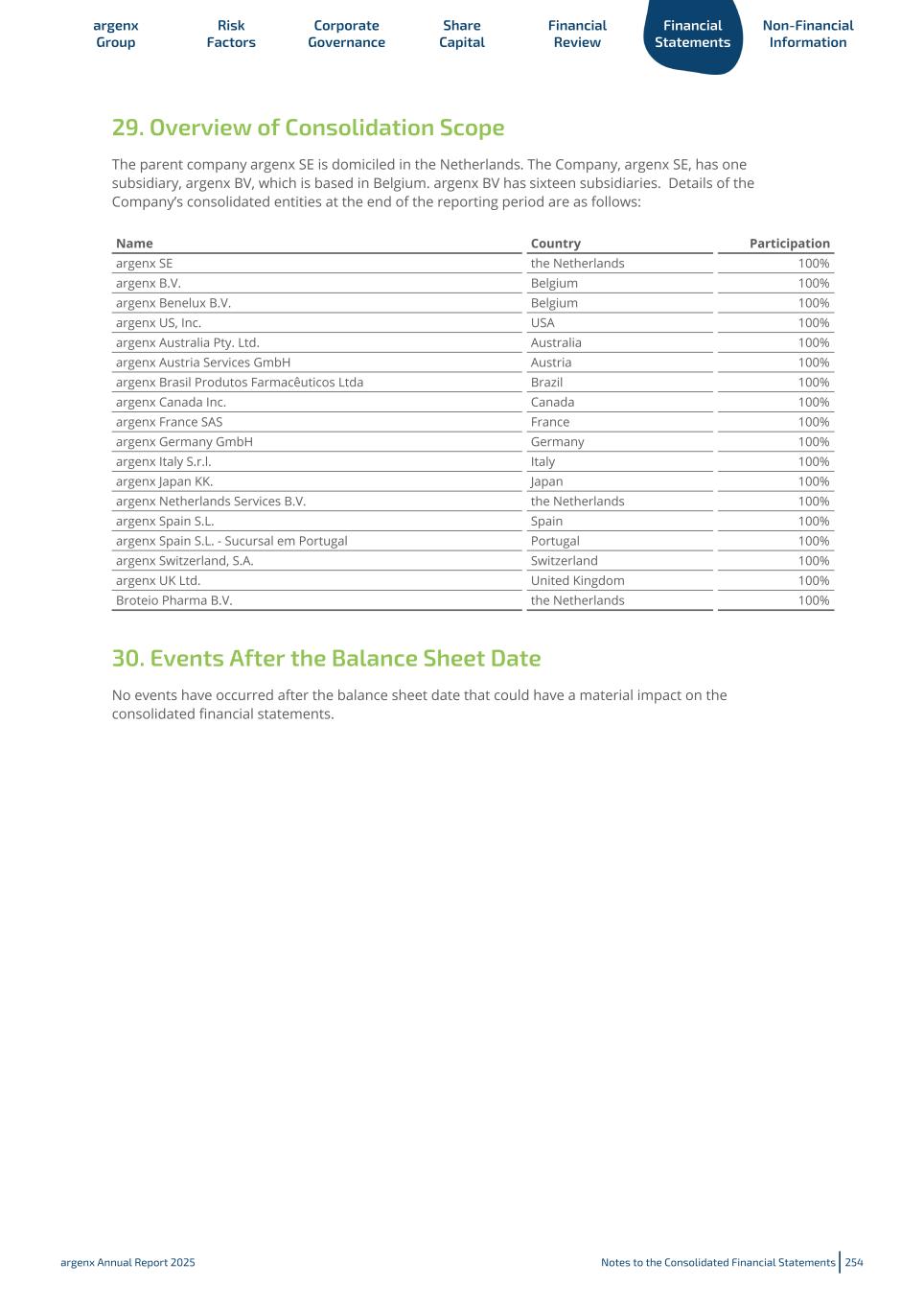
29. Overview of Consolidation Scope The parent company argenx SE is domiciled in the Netherlands. The Company, argenx SE, has one subsidiary, argenx BV, which is based in Belgium. argenx BV has sixteen subsidiaries. Details of the Company’s consolidated entities at the end of the reporting period are as follows: Name Country Participation argenx SE the Netherlands 100% argenx B.V. Belgium 100% argenx Benelux B.V. Belgium 100% argenx US, Inc. USA 100% argenx Australia Pty. Ltd. Australia 100% argenx Austria Services GmbH Austria 100% argenx Brasil Produtos Farmacêuticos Ltda Brazil 100% argenx Canada Inc. Canada 100% argenx France SAS France 100% argenx Germany GmbH Germany 100% argenx Italy S.r.l. Italy 100% argenx Japan KK. Japan 100% argenx Netherlands Services B.V. the Netherlands 100% argenx Spain S.L. Spain 100% argenx Spain S.L. - Sucursal em Portugal Portugal 100% argenx Switzerland, S.A. Switzerland 100% argenx UK Ltd. United Kingdom 100% Broteio Pharma B.V. the Netherlands 100% 30. Events After the Balance Sheet Date No events have occurred after the balance sheet date that could have a material impact on the consolidated financial statements. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Notes to the Consolidated Financial Statements 254
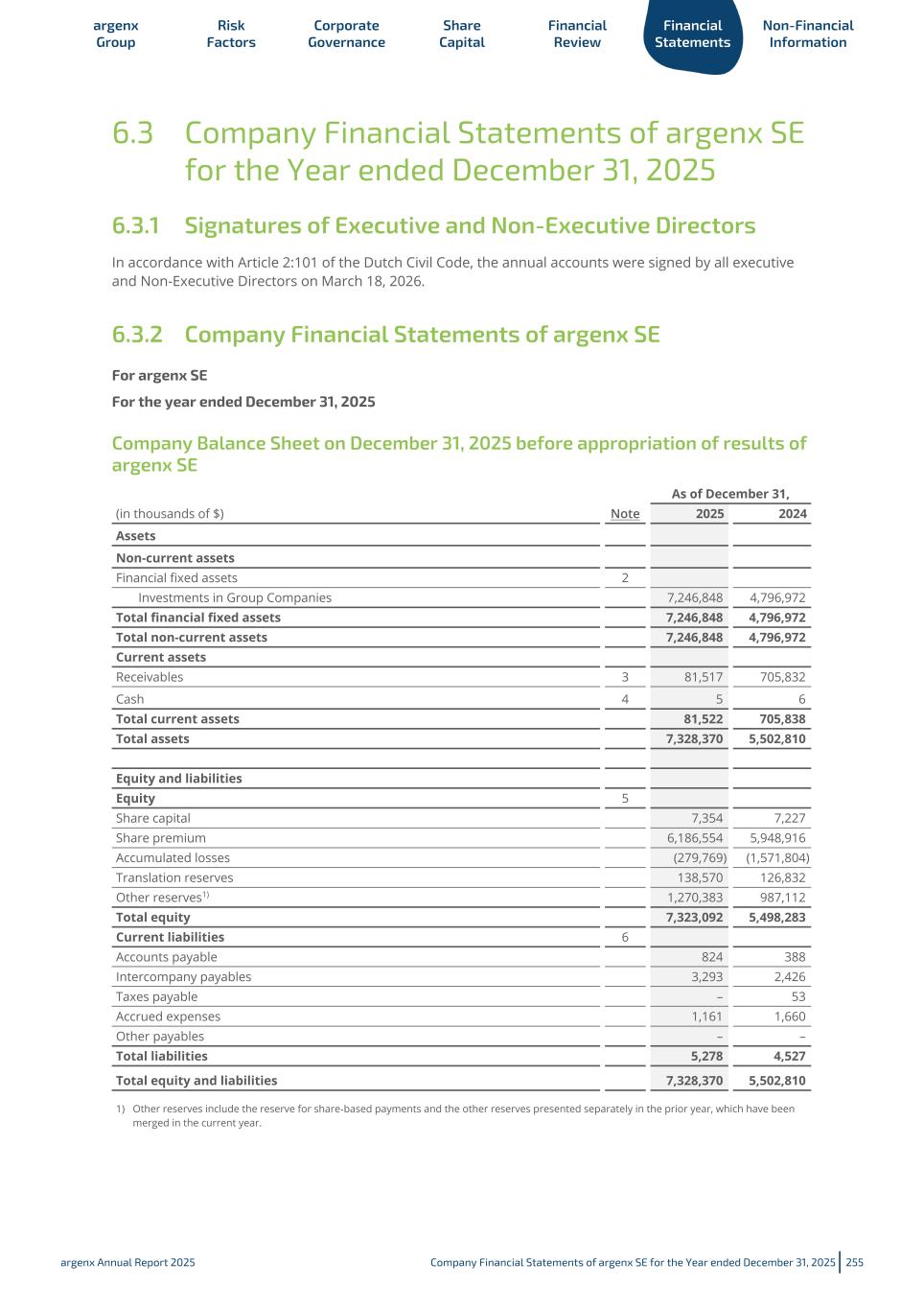
6.3 Company Financial Statements of argenx SE for the Year ended December 31, 2025 6.3.1 Signatures of Executive and Non-Executive Directors In accordance with Article 2:101 of the Dutch Civil Code, the annual accounts were signed by all executive and Non-Executive Directors on March 18, 2026. 6.3.2 Company Financial Statements of argenx SE For argenx SE For the year ended December 31, 2025 Company Balance Sheet on December 31, 2025 before appropriation of results of argenx SE As of December 31, (in thousands of $) Note 2025 2024 Assets Non-current assets Financial fixed assets 2 Investments in Group Companies 7,246,848 4,796,972 Total financial fixed assets 7,246,848 4,796,972 Total non-current assets 7,246,848 4,796,972 Current assets Receivables 3 81,517 705,832 Cash 4 5 6 Total current assets 81,522 705,838 Total assets 7,328,370 5,502,810 Equity and liabilities Equity 5 Share capital 7,354 7,227 Share premium 6,186,554 5,948,916 Accumulated losses (279,769) (1,571,804) Translation reserves 138,570 126,832 Other reserves1) 1,270,383 987,112 Total equity 7,323,092 5,498,283 Current liabilities 6 Accounts payable 824 388 Intercompany payables 3,293 2,426 Taxes payable – 53 Accrued expenses 1,161 1,660 Other payables – – Total liabilities 5,278 4,527 Total equity and liabilities 7,328,370 5,502,810 1) Other reserves include the reserve for share-based payments and the other reserves presented separately in the prior year, which have been merged in the current year. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Company Financial Statements of argenx SE for the Year ended December 31, 2025 255

Company Profit and Loss Account for the Year Ended December 31, 2025 of argenx SE Year Ended December 31, (in thousands of $) Note 2025 20241) Intercompany recharges 98 – Total operating income 98 – General & administrative expenses (24,167) (21,594) Total operating expenses (24,167) (21,594) Operating result (24,069) (21,594) Financial income and expense 7 4,744 2,161 Result before taxation (19,325) (19,433) Taxation on result of ordinary activities 7 23 Result after taxation (19,318) (19,410) Share in result of subsidiaries 8 1,311,353 852,450 Net Result 1,292,035 833,040 1) Comparative figures have been aligned with the presentation adopted in the current year. 6.3.3 Notes to the Company Financial Statements of argenx SE 1. Acounting information and Policies 1.1 Basis of Preparation The company financial statements of argenx SE (hereafter: ‘‘the Company’’) have been prepared in accordance with Part 9, Book 2 of the Dutch Civil Code. In accordance with article 362 sub8, Book 2 of the Dutch Civil Code, the company’s financial statements are prepared based on the accounting principles of recognition, measurement and determination of profit, as applied in the Consolidated IFRS financial statements. 1.2 Summary of Significant Accounting Policies In case no other policies are mentioned, refer to the accounting policies as described in the summary of significant accounting policies in the Consolidated IFRS financial statements. For an appropriate interpretation, the company’s financial statements argenx SEshould be read in conjunction with the Consolidated IFRS financial statements. Participating Interests in Group Companies Participating interests in group companies are valued using the equity method, applying the IFRS accounting policies endorsed by the European Union. Following the adoption of IFRS 9 by the group, and our interpretation of the Dutch Accounting Standard 100.108, the company shall, upon identification of a credit loss on an intercompany loan and/or receivable, eliminate the carrying amount of the intercompany loan and/or receivable for the value of the identified credit loss. Result of Participating Interests The share in the result of participating interests consists of the share of the Company in the result of these participating interests. In so far as gains or losses on transactions involving the transfer of assets and liabilities between the Company and its participating interests or between participating interests themselves can be considered unrealized, they have not been recognized. All amounts are presented in thousands of USD, unless stated otherwise. The balance sheet and profit and loss statement references have been included. These refer to the notes herein. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Company Financial Statements of argenx SE for the Year ended December 31, 2025 256
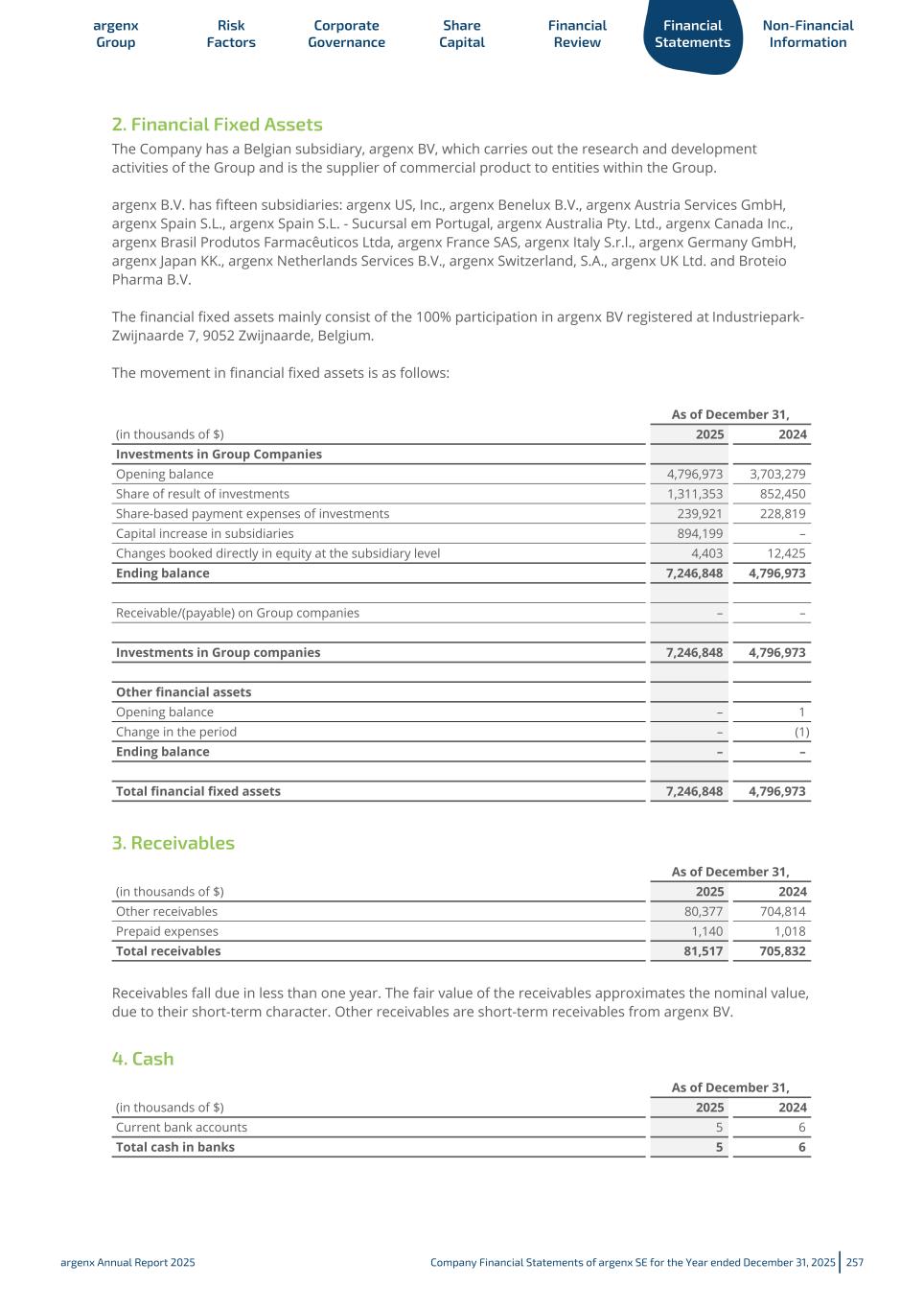
2. Financial Fixed Assets The Company has a Belgian subsidiary, argenx BV, which carries out the research and development activities of the Group and is the supplier of commercial product to entities within the Group. argenx B.V. has fifteen subsidiaries: argenx US, Inc., argenx Benelux B.V., argenx Austria Services GmbH, argenx Spain S.L., argenx Spain S.L. - Sucursal em Portugal, argenx Australia Pty. Ltd., argenx Canada Inc., argenx Brasil Produtos Farmacêuticos Ltda, argenx France SAS, argenx Italy S.r.l., argenx Germany GmbH, argenx Japan KK., argenx Netherlands Services B.V., argenx Switzerland, S.A., argenx UK Ltd. and Broteio Pharma B.V. The financial fixed assets mainly consist of the 100% participation in argenx BV registered at Industriepark- Zwijnaarde 7, 9052 Zwijnaarde, Belgium. The movement in financial fixed assets is as follows: As of December 31, (in thousands of $) 2025 2024 Investments in Group Companies Opening balance 4,796,973 3,703,279 Share of result of investments 1,311,353 852,450 Share-based payment expenses of investments 239,921 228,819 Capital increase in subsidiaries 894,199 – Changes booked directly in equity at the subsidiary level 4,403 12,425 Ending balance 7,246,848 4,796,973 Receivable/(payable) on Group companies – – Investments in Group companies 7,246,848 4,796,973 Other financial assets Opening balance – 1 Change in the period – (1) Ending balance – – Total financial fixed assets 7,246,848 4,796,973 3. Receivables As of December 31, (in thousands of $) 2025 2024 Other receivables 80,377 704,814 Prepaid expenses 1,140 1,018 Total receivables 81,517 705,832 Receivables fall due in less than one year. The fair value of the receivables approximates the nominal value, due to their short-term character. Other receivables are short-term receivables from argenx BV. 4. Cash As of December 31, (in thousands of $) 2025 2024 Current bank accounts 5 6 Total cash in banks 5 6 argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Company Financial Statements of argenx SE for the Year ended December 31, 2025 257
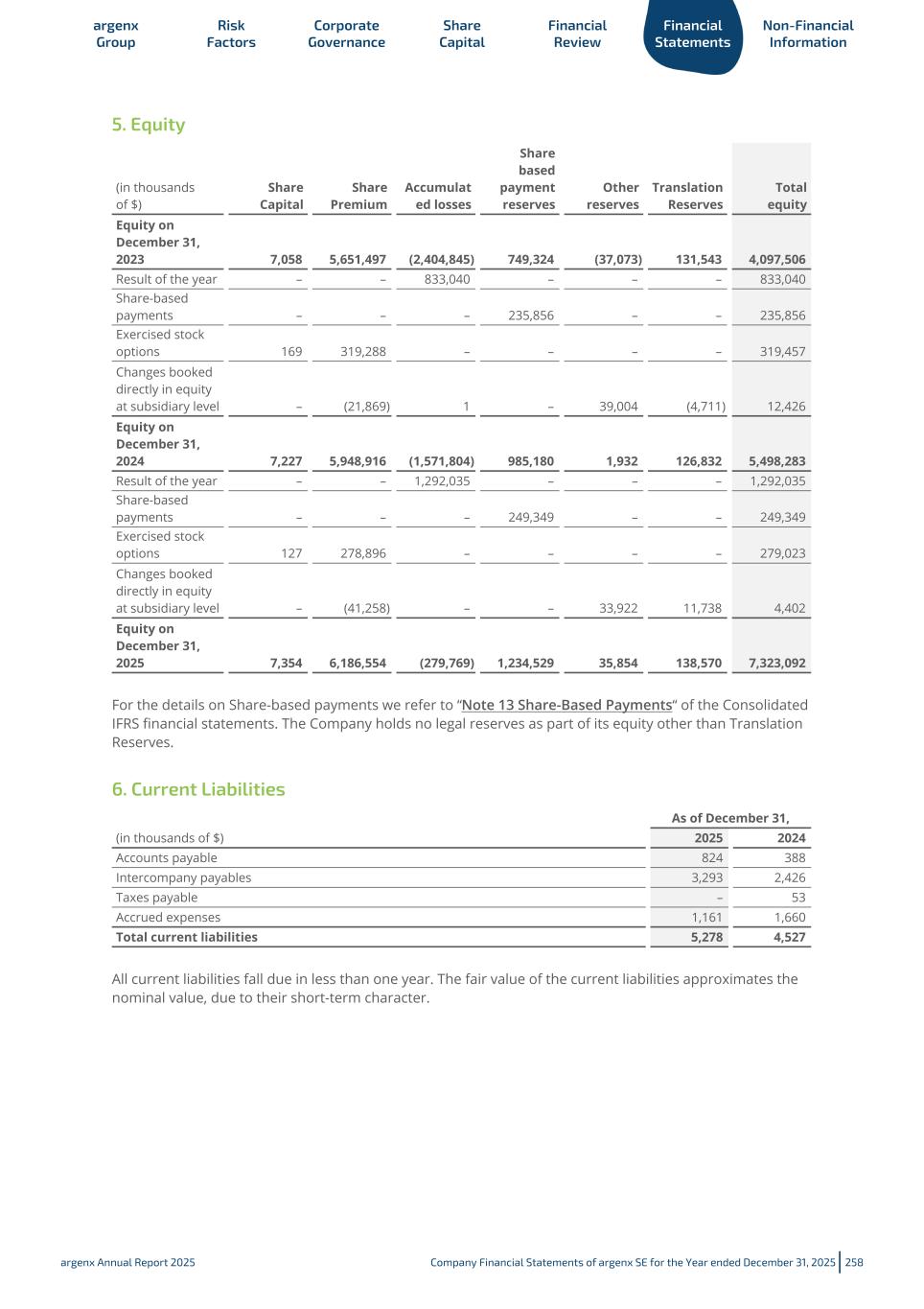
5. Equity (in thousands of $) Share Capital Share Premium Accumulat ed losses Share based payment reserves Other reserves Translation Reserves Total equity Equity on December 31, 2023 7,058 5,651,497 (2,404,845) 749,324 (37,073) 131,543 4,097,506 Result of the year – – 833,040 – – – 833,040 Share-based payments – – – 235,856 – – 235,856 Exercised stock options 169 319,288 – – – – 319,457 Changes booked directly in equity at subsidiary level – (21,869) 1 – 39,004 (4,711) 12,426 Equity on December 31, 2024 7,227 5,948,916 (1,571,804) 985,180 1,932 126,832 5,498,283 Result of the year – – 1,292,035 – – – 1,292,035 Share-based payments – – – 249,349 – – 249,349 Exercised stock options 127 278,896 – – – – 279,023 Changes booked directly in equity at subsidiary level – (41,258) – – 33,922 11,738 4,402 Equity on December 31, 2025 7,354 6,186,554 (279,769) 1,234,529 35,854 138,570 7,323,092 For the details on Share-based payments we refer to “Note 13 Share-Based Payments“ of the Consolidated IFRS financial statements. The Company holds no legal reserves as part of its equity other than Translation Reserves. 6. Current Liabilities As of December 31, (in thousands of $) 2025 2024 Accounts payable 824 388 Intercompany payables 3,293 2,426 Taxes payable – 53 Accrued expenses 1,161 1,660 Total current liabilities 5,278 4,527 All current liabilities fall due in less than one year. The fair value of the current liabilities approximates the nominal value, due to their short-term character. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Company Financial Statements of argenx SE for the Year ended December 31, 2025 258
7. Financial Result and exchange Gains/(Losses) As of December 31, (in thousands of $) 2025 2024 Net gains on investments held at FVTPL 2 88 Fees collected from ADS holders 307 509 Interest on intercompany current account 2,600 6,236 Financial income 2,909 6,833 Other financial expenses (5) (8) Financial expenses (5) (8) Exchange gains/(losses) 1,840 (4,664) Financial income and expense 4,744 2,161 8. Share in Result of Subsidiaries The Company has one Belgian subsidiary, argenx BV, which carries out the research and development activities of the Group and its commercial supply. Year ended December 31, (in thousands of $) 2025 2024 argenx BV 1,311,353 852,450 Total share in result of subsidiaries 1,311,353 852,450 9. Other Disclosures Contingent Liabilities The contingent liabilities of the Company consist of a rental agreement for office space in Amsterdam for an immaterial amount. Related-Party Transactions All legal entities that can be controlled, jointly controlled or significantly influenced are considered as a related party. Also, entities which can control the company are considered a related party. In addition, directors, other key management of argenx SE and close relatives are regarded as related parties. Other than the intercompany cross-charges, there were no related party transactions. Remuneration Remuneration of the Executive Director for 2025 and 2024 is as follows: (in $) 2025 2024 Base pay 827,160 757,679 Variable short-term incentive 744,444 795,563 Stock options granted 2,941,497 3,194,813 Restricted stock units (RSUs) granted – 3,014,500 Performance stock units (PSU) granted 2,856,448 – Pension contributions 44,168 29,118 Fringe benefits 16,054 16,112 Total remuneration of the executive director 7,429,773 7,807,785 Part of the remuneration of the Executive Director is being paid by subsidiaries of argenx SE. See “Note 25 Related Party Transactions“ of the notes to the Consolidated IFRS financial statements for the remuneration of non-executive Board of directors. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Company Financial Statements of argenx SE for the Year ended December 31, 2025 259

Information Relating to Employees During the year 2025, the Company had an average of 0.25 FTE (2024: 0.25 FTE). The employee is based in Belgium. Auditor’s Fees See “Note 28 Audit Fees“ of the notes to the Consolidated IFRS financial statements. Proposal for Appropriation of the Result The Company reported a net result for the year of $1.3 billion for the year ended on December 31, 2025. The Board of Directors proposes to carry forward the net profit of the year 2025 to the accumulated losses. Anticipating the approval of the financial statements by the shareholders at the annual general meeting of shareholders, this proposal has already been reflected in the 2025 financial statements. Events after the balance sheet date For the events after balance sheet date, we refer to “Note 30 Events After the Balance Sheet Date“ of the Consolidated IFRS financial statements. Amsterdam, March 19, 2026 The Director Tim Van Hauwermeiren, CEO argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Company Financial Statements of argenx SE for the Year ended December 31, 2025 260

6.4 Other information 6.4.1 Provision in the articles of association governing the appropriation of results 1. The company shall have a policy on reserves and dividends which shall be determined and may be amended by the board of directors. The adoption and thereafter each material change of the policy on reserves and dividends shall be discussed at the general meeting under a separate agenda item. 2. From the profits, shown in the annual accounts, as adopted, the board of directors shall determine which part shall be reserved. Any profits remaining thereafter shall be at the disposal of the general meeting. The board of directors shall make a proposal for that purpose. A proposal to pay a dividend shall be dealt with as a separate agenda item at the general meeting. 3. Distribution of dividends on the shares shall be made in proportion to the nominal value of each share. 4. Distributions may be made only insofar as the Company’s equity exceeds the amount of the paid in and called up part of the issued capital, increased by the reserves which must be kept by virtue of the law. 5. If a loss was suffered during any one year, the board of directors may resolve to offset such loss by writing it off against a reserve which the company is not required to keep by virtue of the law. 6. The distribution of profits shall be made after the adoption of the annual accounts, from which it appears that the same is permitted. 7. The board of directors may, subject to due observance of the policy of the Company on reserves and dividends, resolve to make an interim distribution, provided the requirement of paragraph 4 of this article has been complied with, as shown by interim accounts. Such interim accounts shall show the financial position of the Company not earlier than on the first day of the third month before the month in which the resolution to make the interim distribution is announced. Such interim accounts shall be signed by all members of the board of directors. If the signature of one or more of them is missing, this shall be stated and reasons for this omission shall be given. The interim accounts shall be deposited in the offices of the trade register within eight days after the day on which the resolution to make the interim distribution has been announced. 8. At the proposal of the board of directors, the general meeting may resolve to make a distribution on shares wholly or partly not in cash but in shares. 9. The board of directors may, subject to due observance of the policy of the Company on reserves and dividends, resolve that distributions to holders of shares shall be made out of one or more reserves. 10. A claim of a shareholder for payment of a distribution shall be barred after five years have elapsed. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Other information 261

6.4.2 Independent Auditor's report To: the Shareholders and the Board of Directors of argenx SE Report on the audit of the financial statements 2025 included in the Annual Report Our opinion We have audited the accompanying financial statements 2025 of argenx SE based in Amsterdam, the Netherlands. The financial statements comprise the consolidated financial statements and the company financial statements. In our opinion: • The consolidated financial statements give a true and fair view of the financial position of argenx SE as at December 31, 2025 and of its result and its cash flows for 2025 in accordance with IFRS Accounting Standards as adopted in the European Union (IFRS Accounting Standards) and with Part 9 of Book 2 of the Dutch Civil Code • The company financial statements give a true and fair view of the financial position of argenx SE as at December 31, 2025 and of its result for 2025 in accordance with Part 9 of Book 2 of the Dutch Civil Code The consolidated financial statements comprise: • The consolidated statement of financial position as at December 31, 2025 • The following statements for the year ended December 31, 2025: the consolidated statements of profit or loss, comprehensive income or loss, changes in equity and cash flows • The notes comprising material accounting policy information and other explanatory information The company financial statements comprise: • The company balance sheet on December 31, 2025 • The company profit and loss account for the year ended December 31, 2025 • The notes comprising a summary of the accounting policies and other explanatory information Basis for our opinion We conducted our audit in accordance with Dutch law, including the Dutch Standards on Auditing. Our responsibilities under those standards are further described in the Our responsibilities for the audit of the financial statements section of our report. We are independent of argenx SE in accordance with the EU Regulation on specific requirements regarding statutory audit of public-interest entities, the Wet toezicht accountantsorganisaties (Wta, Audit firms supervision act), the Verordening inzake de onafhankelijkheid van accountants bij assurance-opdrachten (ViO, Code of Ethics for Professional Accountants, a regulation with respect to independence) and other relevant independence regulations in the Netherlands. Furthermore, we have complied with the Verordening gedrags- en beroepsregels accountants (VGBA, Dutch Code of Ethics for Professional Accountants). We believe the audit evidence we have obtained is sufficient and appropriate to provide a basis for our opinion. Information in support of our opinion We designed our audit procedures in the context of our audit of the financial statements as a whole and in forming our opinion thereon. The following information in support of our opinion and any findings were addressed in this context, and we do not provide a separate opinion or conclusion on these matters. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Other information 262

Our understanding of the business argenx SE (“the company”, and, together with its consolidated subsidiaries, “the group”) is a commercial- stage biopharma company developing and marketing therapies for the treatment of severe autoimmune diseases. We paid specific attention in our audit to a number of areas driven by the operations of the group and our risk assessment. We determined materiality and identified and assessed the risks of material misstatement of the financial statements, whether due to fraud or error in order to design audit procedures responsive to those risks and to obtain audit evidence that is sufficient and appropriate to provide a basis for our opinion. Materiality Materiality $73 million Benchmark applied 7% of operating profit for the year ended December 31, 2025 Explanation When determining the appropriate measurement basis, we considered which key performance indicators are the focus of the users of the financial statements. As a significant amount of the profit/(loss) for the year before taxes is composed of financial income, financial expense and exchange gains/(losses), which we determined does not appropriately reflect the operating performance of the company, we concluded that the most appropriate materiality base is operating profit. We have also taken into account misstatements and/or possible misstatements that in our opinion are material for the users of the financial statements for qualitative reasons. We agreed with the Board of Directors that misstatements in excess of $3.65 million, which are identified during the audit, would be reported to them, as well as smaller misstatements that in our view must be reported on qualitative grounds. Scope of the group audit argenx SE is at the head of a group of entities. The company has its official seat in the Netherlands. The group’s headquarters and the primary research and development activities are in Belgium. The majority of the group’s product net sales is recognized from the United States. The group relies on contract manufacturing organizations (CMOs) with manufacturing sites in the UK, U.S., Singapore, Switzerland and Denmark. The financial information of this group is included in the financial statements. We are responsible for planning and performing the group audit to obtain sufficient appropriate audit evidence regarding the financial information of the entities or business units within the group as a basis for forming an opinion on the financial statements. We are also responsible for the direction, supervision, review and evaluation of the audit work performed for purposes of the group audit. We bear the full responsibility for the auditor’s report. Based on our understanding of the group and its environment, the applicable financial framework and the group’s system of internal control, we identified and assessed risks of material misstatement of the financial statements and the significant accounts and disclosures. Based on this risk assessment, we determined the nature, timing and extent of audit work performed, including the entities or business units within the group (components) at which to perform audit work. For this determination we considered the nature of the relevant events and conditions underlying the identified risks of material misstatements for the financial statements, the association of these risks to components and the materiality or financial size of the components relative to the group. We performed the (centralized) audit work ourselves at the group’s headquarters in Belgium, including (centralized) audit work for selected components and financial statement account balances such as product net sales and deferred tax assets. For the financial information of argenx B.V., comprising the research and development activities in Belgium, we made use of an EY Firm (component auditor). We communicated the audit work to be performed and identified risks through instructions for the component auditor as well as requesting the component auditor to communicate matters related to the financial information of the component that is relevant to identifying and assessing risks. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Other information 263

This resulted in a coverage of 100% of operating profit/(loss), 88% of total operating income and 94% of total assets. For other components, we performed specified audit procedures and analytical procedures to corroborate that our risk assessment and scoping remained appropriate throughout the audit. We performed site visits to meet with local management of argenx US, Inc. (USA) and with local management and the component auditor of argenx B.V. (Belgium). These site visits encompassed some, or all, of the following activities: observing the component operations, discussing the group risk assessment and the risks of material misstatements. We frequently communicated with the component team in Belgium, reviewed and evaluated the adequacy of the deliverables and reviewed working papers to address the risks of material misstatement. We held planning meetings, key meetings required based on circumstances and we attended closing meetings with local management and the component team. By performing the audit work mentioned above at the entities or business units within the group, together with additional work at group level, we have been able to obtain sufficient and appropriate audit evidence about the group’s financial information to provide an opinion on the financial statements. Teaming, use of specialists and internal audit We ensured that the audit teams both at group and at component levels included the appropriate skills and competences which are needed for the audit of a listed client in the life sciences industry. We included specialists in the areas of IT audit, forensics, and income tax. We performed our audit in cooperation with Internal Audit of argenx SE, leveraging their in-depth knowledge of the group and work performed. We agreed on the joint coordination of the audit planning, the nature and scope of the work to be performed, reporting and documentation. We evaluated and tested the relevant work performed by Internal Audit to satisfy ourselves that the work was adequate for our purposes and established what work had to be performed by our own professionals. Our focus on climate-related risks and the energy transition Climate change and the energy transition are high on the public agenda. Issues such as CO2 reduction impact financial reporting, as these issues entail risks for the business operation, the valuation of assets and provisions or the sustainability of the business model and access to financial markets of companies with a larger CO2 footprint. The Board of Directors reported in the section 7.2 of the Non-financial information how the company is addressing climate-related and environmental risks. As part of our audit of the financial statements, we evaluated the extent to which climate-related risks and the effects of the energy transition are taken into account in estimates and significant assumptions as well as in the design of relevant internal control measures. Furthermore, we read the non-financial information in the annual report and considered whether there is any material inconsistency between the non-financial information and the financial statements. Based on the audit procedures performed, we do not deem climate-related risks to have a material impact on the financial reporting judgements, estimates or significant assumptions as at December 31, 2025. Our focus on fraud and non-compliance with laws and regulations Our responsibility Although we are not responsible for preventing fraud or non-compliance and we cannot be expected to detect non-compliance with all laws and regulations, it is our responsibility to obtain reasonable assurance that the financial statements, taken as a whole, are free from material misstatement, whether caused by fraud or error. The risk of not detecting a material misstatement resulting from fraud is higher than for one resulting from error, as fraud may involve collusion, forgery, intentional omissions, misrepresentations, or the override of internal control. Our audit response related to fraud risks We identified and assessed the risks of material misstatements of the financial statements due to fraud. During our audit we obtained an understanding of the company and its environment and the components argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Other information 264

of the system of internal control, including the risk assessment process and the board of director’s process for responding to the risks of fraud and monitoring the system of internal control, as well as the outcomes. We refer to Section 2 ‘‘Risk Factors’’ of the Annual Report for the Board of Directors’ (fraud) risk assessment. We evaluated the design and relevant aspects of the system of internal control and in particular the fraud risk assessment, as well as the argenx Code of Business Conduct and Ethics, Global Speak Up and Anti- Retaliation Policy and incident registration. We evaluated the design and the implementation and, where considered appropriate, tested the operating effectiveness, of internal controls designed to mitigate fraud risks. As part of our process of identifying fraud risks, we evaluated fraud risk factors with respect to financial reporting fraud, misappropriation of assets and bribery and corruption in close co-operation with our forensic specialists. We evaluated whether these factors indicate that a risk of material misstatement due to fraud is present. We incorporated elements of unpredictability in our audit. We also considered the outcome of our other audit procedures and evaluated whether any findings were indicative of fraud or non-compliance. We addressed the risks related to management override of controls, as this risk is present in all organizations. For these risks we have, among other things, performed procedures to evaluate whether the selection and application of accounting policies by the company, particularly those relating to subjective measurements and complex transactions, as disclosed in “Note 3 - Critical accounting judgments and major sources of estimation uncertainty” to the consolidated financial statements, may be indicative to fraudulent financial reporting. We have also used data analysis to identify and address high-risk journal entries and other adjustments made in the financial reporting process. We evaluated the business rationale (or the lack thereof) of significant extraordinary transactions, including those with related parties. When identifying and assessing fraud risks we presumed that there are risks of fraud in revenue recognition. We evaluated the risk of management manipulating the payor mix assumption for the US Sales rebates and reserves related to Medicare Part D, in particular give rise to such risks. We describe the audit procedures responsive to the risk of fraud in revenue recognition in the description of our audit approach for the key audit matter U.S. Sales Rebates and Reserves –Medicare Part D Claims. We considered available information and made enquiries of relevant individuals including the Board of Directors (including the Chair of Audit and Compliance Committee), the Chief Executive Officer, Chief Operating Officer, the Chief Financial Officer, Global Group Controller, Head of Internal Control, Head of Global Ethics and Compliance, General Counsel, and Internal Audit. The fraud risks we identified, enquiries and other available information did not lead to specific indications for fraud or suspected fraud potentially materially impacting the view of the financial statements. Our audit response related to risks of non-compliance with laws and regulations We performed appropriate audit procedures regarding compliance with the provisions of those laws and regulations that have a direct effect on the determination of material amounts and disclosures in the financial statements. Furthermore, we assessed factors related to the risks of non-compliance with laws and regulations that could reasonably be expected to have a material effect on the financial statements from our general industry experience, through discussions with the Board of Directors, reading minutes, inspection of internal audit and compliance reports, and performing substantive tests of details of classes of transactions, account balances or disclosures. We also inspected lawyers’ letters and correspondence with regulatory authorities. We remained alert to any indication of (suspected) non-compliance throughout the audit. Finally, we obtained written representations that all known instances of non-compliance with laws and regulations have been disclosed to us. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Other information 265

Our audit response related to going concern As disclosed in “Note 2.1 – Statement of compliance and basis of preparation” to the consolidated financial statements, the financial statements have been prepared on a going concern basis. When preparing the financial statements, the Board of Directors made a specific assessment of the company’s ability to continue as a going concern and to continue its operations for the foreseeable future. We discussed and evaluated the specific assessment with the Board of Directors exercising professional judgment and maintaining professional skepticism. We considered whether the Board of Directors’ going concern assessment, based on our knowledge and understanding obtained through our audit of the financial statements or otherwise, contains all relevant events or conditions that may cast significant doubt on the company’s ability to continue as a going concern. If we conclude that a material uncertainty exists, we are required to draw attention in our auditor’s report to the related disclosures in the financial statements or, if such disclosures are inadequate, to modify our opinion. Based on our procedures performed, we did not identify material uncertainties about going concern or the Board of Directors’ use of the going concern basis of accounting. Our conclusions are based on the audit evidence obtained up to the date of our auditor’s report. However, future events or conditions may cause a company to cease to continue as a going concern. Our key audit matters Key audit matters are those matters that, in our professional judgment, were of most significance in our audit of the financial statements. We have communicated the key audit matter to the Board of Directors. The key audit matter is not a comprehensive reflection of all matters discussed. U.S. Sales Rebates and Reserves – Medicare Part D Claims Risk As described in “Note 2.17 – Product Net Sales’’, “Note 3 – Critical Accounting Judgments”, and “Note 14 – Trade and Other Payables” to the consolidated financial statements, the Company recognizes revenue net of price reductions (product net sales), including, among others, estimates of Medicare Part D Manufacturer Discount Program claims and also recognizes an accrued liability, in sales rebates and reserves, for the estimated claims amount. These claim estimates are based on the expected value method, taking into account the payor mix. At December 31, 2025, the Company had a total of $402 million in liabilities related to sales rebates and reserves, which includes Medicare Part D Claims. We consider the U.S. Sales Rebates and Reserves - Medicare Part D Claims a key audit matter as the related rebates and reserves are significant to the financial statements due to the subjectivity related to the payor mix assumption used in determining the rebates and reserves as well as a result of the presumed risk of fraud in revenue recognition. Our audit approach We evaluated the appropriateness of the group’s revenue recognition accounting policies, in particular relating to the U.S. sales rebates and reserves, in accordance with IFRS 15 ‘Revenue from Contracts with Customers’ and tested the Company’s internal controls over the Medicare Part D sales rebates and reserves process. This included testing controls over the data used to determine the payor mix, management's review of the expected-value method model and the payor mix assumption, and the comparison of actual claim payments to the estimated reserves. Our audit procedures to test Medicare Part D sales rebates and reserves included, among others, independently developing an estimate of the claims amount and testing the mathematical accuracy of the model used by management. We assessed the reasonableness of the Company's Medicare Part D claims by comparing previous estimates to actual claims and agreeing a sample of those claims to source documents. We evaluated the adequacy of Company’s disclosures related to sales rebates and reserves. Key observations Based on our procedures performed, we have not identified any material misstatements relating to the U.S. Sales Rebates and Reserves – Medicare Part D Claims. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Other information 266

Report on other information included in the annual report The annual report contains other information in addition to the financial statements and our auditor’s report thereon. Based on the following procedures performed, we conclude that the other information: • Is consistent with the financial statements and does not contain material misstatements • Contains the information as required by Part 9 of Book 2 of the Dutch Civil Code for the management report (excluding the sustainability statement) and the other information as required by Part 9 of Book 2 of the Dutch Civil Code and as required by Sections 2:135b and 2:145 sub-section 2 of the Dutch Civil Code for the remuneration report. We have read the other information. Based on our knowledge and understanding obtained through our audit of the financial statements or otherwise, we have considered whether the other information contains material misstatements. By performing these procedures, we comply with the requirements of Part 9 of Book 2 and Section 2:135b sub-Section 7 of the Dutch Civil Code and the Dutch Standard 720. The scope of the procedures performed is substantially less than the scope of those performed in our audit of the financial statements. The Board of Directors is responsible for the preparation of the other information, including the management report in accordance with Part 9 of Book 2 of the Dutch Civil Code and other information required by Part 9 of Book 2 of the Dutch Civil Code. The Board of Directors is responsible for ensuring that the remuneration report is drawn up and published in accordance with Sections 2:135b and 2:145 sub-section 2 of the Dutch Civil Code. Description of responsibilities regarding the financial statements Responsibilities of the Board of Directors for the financial statements The Board of Directors is responsible for the preparation and fair presentation of the financial statements in accordance with IFRS Accounting Standards and Part 9 of Book 2 of the Dutch Civil Code. Furthermore, the Board of Directors is responsible for such internal control as the Board of Directors determines is necessary to enable the preparation of the financial statements that are free from material misstatement, whether due to fraud or error. As part of the preparation of the financial statements, the Board of Directors is responsible for assessing the company’s ability to continue as a going concern. Based on the financial reporting framework mentioned, the Board of Directors should prepare the financial statements using the going concern basis of accounting unless the Board of Directors either intends to liquidate the company or to cease operations, or has no realistic alternative but to do so. The Board of Directors should disclose events and circumstances that may cast significant doubt on the company’s ability to continue as a going concern in the financial statements. The Audit and Compliance Committee advises the Board of Directors on matters relating to the oversight of the quality, integrity, functioning and effectiveness of the company’s financial reporting, internal risk management and control systems over financial and non-financial matters. Our responsibilities for the audit of the financial statements Our objective is to plan and perform the audit engagement in a manner that allows us to obtain sufficient and appropriate audit evidence for our opinion. Our audit has been performed with a high, but not absolute, level of assurance, which means we may not detect all material misstatements, whether due to fraud or error during our audit. Misstatements can arise from fraud or error and are considered material if, individually or in the aggregate, they could reasonably be expected to influence the economic decisions of users taken on the basis of these financial statements. The materiality affects the nature, timing and extent of our audit procedures and the evaluation of the effect of identified misstatements on our opinion. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Other information 267

We have exercised professional judgment and have maintained professional skepticism throughout the audit, in accordance with Dutch Standards on Auditing, ethical requirements and independence requirements. The Information in support of our opinion section above includes an informative summary of our responsibilities and the work performed as the basis for our opinion. Our audit further included among others: • Performing audit procedures responsive to the risks identified, and obtaining audit evidence that is sufficient and appropriate to provide a basis for our opinion • Obtaining an understanding of internal control relevant to the audit in order to design audit procedures that are appropriate in the circumstances, but not for the purpose of expressing an opinion on the effectiveness of the company’s internal control • Evaluating the appropriateness of accounting policies used and the reasonableness of accounting estimates and related disclosures made by management • Evaluating the overall presentation, structure and content of the financial statements, including the disclosures • Evaluating whether the financial statements represent the underlying transactions and events in a manner that achieves fair presentation Communication We communicate with the Board of Directors regarding, among other matters, the planned scope and timing of the audit and significant audit findings, including any significant findings in internal control that we identify during our audit. In this respect we also submit an additional report to the Audit and Compliance Committee of the Board of Directors in accordance with Article 11 of the EU Regulation on specific requirements regarding statutory audit of public-interest entities. The information included in this additional report is consistent with our audit opinion in this auditor’s report. We provide the Audit and Compliance Committee of the Board of Directors with a statement that we have complied with relevant ethical requirements regarding independence, and to communicate with them all relationships and other matters that may reasonably be thought to bear on our independence, and where applicable, related safeguards. From the matters communicated with the Board of Directors, we determine the key audit matters: those matters that were of most significance in the audit of the financial statements. We describe these matters in our auditor’s report unless law or regulation precludes public disclosure about the matter or when, in extremely rare circumstances, not communicating the matter is in the public interest. Report on other legal and regulatory requirements and ESEF Engagement We were appointed by the general meeting as auditor of argenx SE on May 7, 2024, as of the audit for the year 2025. No prohibited non-audit services We have not provided prohibited non-audit services as referred to in Article 5(1) of the EU Regulation on specific requirements regarding statutory audit of public-interest entities. European Single Electronic Reporting Format (ESEF) argenx SE has prepared the annual report in ESEF. The requirements for this are set out in the Delegated Regulation (EU) 2019/815 with regard to regulatory technical standards on the specification of a single electronic reporting format (hereinafter: the RTS on ESEF). In our opinion the annual report prepared in the XHTML format, including the (partially) marked-up consolidated financial statements as included in the reporting package by argenx SE, complies in all material respects with the RTS on ESEF. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Other information 268

Management is responsible for preparing the annual report, including the financial statements, in accordance with the RTS on ESEF, whereby management combines the various components into a single reporting package. Our responsibility is to obtain reasonable assurance for our opinion whether the annual report in this reporting package complies with the RTS on ESEF. We performed our examination in accordance with Dutch law, including Dutch Standard 3950N, ”Assurance-opdrachten inzake het voldoen aan de criteria voor het opstellen van een digitaal verantwoordingsdocument” (assurance engagements relating to compliance with criteria for digital reporting). Our examination included amongst others: • Obtaining an understanding of the company’s financial reporting process, including the preparation of the reporting package • Identifying and assessing the risks that the annual report does not comply in all material respects with the RTS on ESEF and designing and performing further assurance procedures responsive to those risks to provide a basis for our opinion, including: ◦ Obtaining the reporting package and performing validations to determine whether the reporting package containing the Inline XBRL instance document and the XBRL extension taxonomy files, has been prepared in accordance with the technical specifications as included in the RTS on ESEF ◦ Examining the information related to the consolidated financial statements in the reporting package to determine whether all required mark-ups have been applied and whether these are in accordance with the RTS on ESEF. Eindhoven, March 19, 2026 EY Accountants B.V. signed by J. C. F. Lemmens argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Other information 269

6.4.3 Limited Assurance Report of the Independent Auditor on the Sustainability Statement To: the Shareholders and the Board of Directors of argenx SE Our conclusion We have performed a limited assurance engagement on the consolidated sustainability statement for the year ended December 31, 2025 of argenx SE based in Amsterdam (hereinafter: the company) in section 7 ‘Non-Financial information’ of the accompanying management report including the information incorporated in the sustainability statement by reference (hereinafter: the sustainability statement). Based on our procedures performed and the evidence obtained, nothing has come to our attention that causes us to believe that the sustainability statement is not, in all material respects: • prepared in accordance with the European Sustainability Reporting Standards (ESRS) as adopted by the European Commission and compliant with the double materiality assessment process carried out by the company to identify the information reported pursuant to the ESRS; and • compliant with the reporting requirements provided for in Article 8 of Regulation (EU) 2020/852 (Taxonomy Regulation). Our conclusion has been formed on the basis of the matters outlined in this limited assurance report. Basis for our conclusion We have performed our limited assurance engagement on the sustainability statement in accordance with Dutch law, including Dutch Standard 3810N, “Assurance-opdrachten inzake duurzaamheidsverslaggeving” (Assurance engagements relating to sustainability reporting), which is a specified Dutch standard that is based on the International Standard on Assurance Engagements (ISAE) 3000 (Revised), “Assurance engagements other than audits or reviews of historical financial information”. Our assurance engagement was aimed to obtain a limited level of assurance that the sustainability statement is free from material misstatements. The procedures vary in nature and timing from, and are less in extent, than for a reasonable assurance engagement. Consequently, the level of assurance obtained in a limited assurance engagement is substantially lower than the assurance that would have been obtained had a reasonable assurance engagement been performed. Our responsibilities in this regard are further described in the section ‘Our responsibilities for the limited assurance engagement on the sustainability statement’ of our report. We are independent of argenx SE in accordance with the Verordening inzake de onafhankelijkheid van accountants bij assurance-opdrachten (ViO, Code of Ethics for Professional Accountants, a regulation with respect to independence) and other relevant independence regulations in the Netherlands. This includes that we do not perform any activities that could result in a conflict of interest with our independent assurance engagement and we are not involved in the preparation of the sustainability statement, as doing so may compromise our independence. Furthermore, we have complied with the Verordening gedrags- en beroepsregels accountants (VGBA, Dutch Code of Ethics for Professional Accountants). The ViO and VGBA are at least as demanding as the International code of ethics for professional accountants (including International independence standards) of the International Ethics Standards Board for Accountants (the IESBA Code) as relevant to limited assurance engagements on sustainability statements of public interest entities in the European Union. We believe that the assurance evidence we have obtained is sufficient and appropriate to provide a basis for our conclusion. Inherent limitations associated with measurement or evaluation of sustainability information Significant uncertainties affecting the quantitative metrics and monetary amounts Section 7.1.2 .2 “Disclosures in relation to specific circumstances” in the sustainability statement identifies the quantitative metrics and monetary amounts that are subject to a high level of measurement argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Other information 270

uncertainty and discloses information about the sources of measurement uncertainty and the assumptions, approximations and judgments the company has made in measuring these in compliance with the ESRS. Comparability may be limited for entity-specific sustainability information The company provides additional entity-specific sustainability information in Section 7.3.2 “S4 Patients”. The comparability of entity-specific sustainability information between entities and over time may be affected by the absence of a uniform practice or availability of external information sources to measure or evaluate this information that can support comparability. This allows for the application of different, but acceptable, measurement techniques. Inherent limitations of a double materiality assessment process The sustainability statement may not include every impact, risk and opportunity or additional entity-specific disclosure that each individual stakeholder (group) may consider important in its own particular assessment. Inherent limitations of forward-looking information In reporting forward-looking information in accordance with the ESRS, management describes the underlying assumptions and methods of producing the information, as well as other factors that provide evidence that it reflects the actual plans or decisions made by the company (actions). Forward-looking information relates to events and actions that have not yet occurred and may never occur. The actual outcome is likely to be different since anticipated events frequently do not occur as expected. Responsibilities of the Board of Directors for the sustainability statement The executive directors of the Board of Directors are responsible for the preparation of the sustainability statement in accordance with the ESRS, including the double materiality assessment process carried out by the company as the basis for the sustainability statement and disclosure of material impacts, risks and opportunities in accordance with the ESRS. As part of the preparation of the sustainability statement, the executive directors are responsible for compliance with the reporting requirements provided for in Article 8 of Regulation (EU) 2020/852 (Taxonomy Regulation). The executive directors are also responsible for selecting and applying additional entity-specific disclosures to enable users to understand the company’s sustainability-related impacts, risks or opportunities and for determining that these additional entity-specific disclosures are suitable in the circumstances and in accordance with the ESRS. Furthermore, the executive directors are responsible for such internal control as they determine is necessary to enable the preparation of the sustainability statement that is free from material misstatement, whether due to fraud or error. The non-executive directors of the Board of Directors are responsible for overseeing the sustainability reporting process including the double materiality assessment process carried out by the company. Our responsibilities for the limited assurance engagement on the Sustainability Statement Our responsibility is to plan and perform the limited assurance engagement in a manner that allows us to obtain sufficient and appropriate assurance evidence for our conclusion. We apply the applicable quality management requirements pursuant to the Nadere voorschriften kwaliteitsmanagement (NVKM, regulations for quality management) and the International Standard on Quality Management (ISQM) 1, and accordingly maintain a comprehensive system of quality management including documented policies and procedures regarding compliance with ethical requirements, professional standards and other relevant legal and regulatory requirements. Our limited assurance engagement included amongst others: • Performing inquiries and an analysis of the external environment and obtaining an understanding of relevant sustainability themes and issues, the characteristics of the company, its activities and the value chain and its key intangible resources in order to assess the double materiality assessment process argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Other information 271

carried out by the company as the basis for the sustainability statement and disclosure of all material sustainability-related impacts, risks and opportunities in accordance with the ESRS. • Obtaining through inquiries a general understanding of the internal control environment, the company’s processes for gathering and reporting entity-related and value chain information, the information systems and the company’s risk assessment process relevant to the preparation of the sustainability statement and for identifying the company’s activities, determining eligible and aligned economic activities and prepare the disclosures provided for in Article 8 of Regulation (EU) 2020/852 (Taxonomy Regulation), without obtaining assurance information about the implementation or testing the operating effectiveness of controls. • Assessing the double materiality assessment process carried out by the company and identifying and assessing areas of the sustainability statement, including the disclosures provided for in Article 8 of Regulation (EU) 2020/852 (Taxonomy Regulation), where misleading or unbalanced information or material misstatements, whether due to fraud or error, are likely to arise (‘selected disclosures’). Designing and performing further assurance procedures aimed at assessing that the sustainability statement is free from material misstatements responsive to this risk analysis. • Considering whether the description of the double materiality assessment process in the sustainability statement made by the Board of Directors appears consistent with the process carried out by the company. • Performing analytical review procedures on quantitative information in the sustainability statement, including consideration of data and trends. • Assessing whether the company’s methods for developing estimates are appropriate and have been consistently applied for selected disclosures. We considered data and trends, however our procedures did not include testing the data on which the estimates are based or separately developing our own estimates against which to evaluate the Board of Director’s estimates. • Analyzing, on a limited sample basis, relevant internal and external documentation available to the company (including publicly available information or information from actors throughout its value chain) for selected disclosures. • Reading the other information in the annual report to identify material inconsistencies, if any, with the sustainability statement. • Considering whether the disclosures provided to address the reporting requirements provided for in Article 8 of Regulation (EU) 2020/852 (Taxonomy Regulation) for each of the environmental objectives, reconcile with the underlying records of the company and are consistent or coherent with the sustainability statement, appear reasonable, in particular whether the eligible economic activities meet the cumulative conditions to qualify as aligned and whether the technical screening criteria are met, and whether the key performance indicators disclosures have been defined and calculated in accordance with the Taxonomy delegated acts, and comply with the reporting requirements provided for in Article 8 of Regulation (EU) 2020/852 (Taxonomy Regulation), including the format in which the activities are presented. • Considering the overall presentation, structure and fundamental qualitative characteristics of information (relevance and faithful representation: complete, neutral and accurate) reported in the sustainability statement, including the reporting requirements provided for in Article 8 of Regulation (EU) 2020/852 (Taxonomy Regulation). • Considering, based on our limited assurance procedures and evaluation of the evidence obtained, whether the sustainability statement as a whole, is free from material misstatements and prepared in accordance with the ESRS. Communication We communicate with the Board of Directors regarding, among other matters, the planned scope and timing of the assurance engagement and significant findings that we identify during our assurance engagement. Eindhoven, March 19, 2026 EY Accountants B.V. signed by J.C.F. Lemmens argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Other information 272

Non-Financial Information 7.1 Sustainability Statement 274 7.2 Environment 287 7.3 Social 301 7.4 Governance 316 7.5 Appendix 323 argenx Annual Report 2025 273 7

7 Non-Financial Information 7.1 Sustainability Statement 7.1.1 General Information At argenx, we are on a mission to transform the lives of patients by translating immunology breakthroughs into novel antibody-based medicines, guided by a responsible approach to bringing medicines to patients. In 2025, we continued to uphold our commitment to transparency and accountability in line with the European Union’s Non-Financial Reporting Directive (NFRD), as implemented into Dutch legislation. We have integrated our Sustainability Statement into this Annual Report in alignment with the general principles of the CSRD. The Dutch government has stated that companies that voluntarily applied the CSRD in 2024 and 2025— including by (i) reporting in accordance with the European Sustainability Reporting Standards (ESRS), (ii) publishing a sustainability report, and (iii) engaging an external auditor to provide assurance on that report —will, in principle, be considered to have complied with their statutory reporting obligations for those years. This reflects the expectation that Dutch legislation will include retrospective application of the CSRD for financial years beginning after January 1, 2024 and January 1, 2025 for entities within scope. This Sustainability Statement is prepared in accordance with the ESRS and is compliant with the reporting requirements provided for in Article 8 of Regulation (EU) 2020/852. The statement applies the European Commission’s “quick fix” amendment (approved November 2025), which provides transitional relief for companies preparing their second CSRD-aligned disclosures. The EU Taxonomy disclosures are presented within the Environment section of this report and have been prepared in accordance with the reporting rules as applicable until December 31, 2025. Our Sustainability Statement addresses the interests of key stakeholders, including patients, healthcare communities, employees, investors, and business partners, and is guided by the material topics identified through our double materiality assessment. We prioritize ESG topics that most effectively support our mission and deliver value to stakeholders. Our ESG reporting approach is designed to be transparent, purposeful, and aligned with regulatory requirements, while maintaining our focus on patient outcomes, employee engagement, and the interests of our broader stakeholder community. This chapter outlines our ESG principles and the methodologies used to measure progress, reflecting our ongoing efforts to generate positive outcomes for patients, employees, and communities. Content BP-1 General basis for preparation of sustainability statement General Basis of Preparation of the Sustainability Statement (BP-1) BP-2 Disclosures in relation to specific circumstances Disclosures in Relation to Specific Circumstances (BP-2) GOV-1 The role of administrative, management and supervisory bodies Management of Material Risks, Impacts and Opportunities by Administrative, Management and Supervisory Bodies (GOV-1, GOV-2) GOV-2 Information provided to and sustainability matters addressed by the undertaking’s administrative, management and supervisory bodies Management of Material Risks, Impacts and Opportunities by Administrative, Management and Supervisory Bodies (GOV-1, GOV-2) Disclosure Requirements Section ESRS 2 argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Sustainability Statement 274
GOV-3 Integration of sustainability-related performance in incentive schemes Integration of Sustainability-Related Performance in Incentive Schemes (GOV-3) GOV-4 Statement on due diligence Due Diligence (GOV-4) GOV-5 Risk management and internal controls over sustainability reporting Risk Management and Internal Controls Over Sustainability Reporting (GOV-5) SBM-1 Strategy, business model and value chain Sustainability Strategy and Our Business Model (SBM-1) SBM-2 Interests and views of stakeholders Stakeholder Engagement (SBM-2) SBM-3 Material impacts, risks and opportunities and their interaction with strategy and business model Double Materiality Assessment Results (SBM-3) IRO-1 Description of process to identify and assess material impacts, risks, and opportunities Double Materiality Assessment (IRO-1, IRO-2) IRO-2 Disclosure requirements in ESRS covered by the undertaking’s sustainability statement Double Materiality Assessment (IRO-1, IRO-2) Incorporation by reference ESRS 2 BP-1 General basis for preparation of sustainability statement 1.1.1 General, Note 29 Overview of Consolidation Scope SBM-1 Sustainability Strategy and Our Business Model 1.1.2 Our Medicines, 1.3 Our Products and Product Candidates, 5.12 Employees, 6.1 Consolidated Statements of Financial Position GOV-1-2 Board of director’s skills and expertise 3.2 Management Structure, 3.2.4 Non-Executive Directors, 3.3 Report of the Non-Executive Directors, Note 25.3 Relationships and transactions with key personnel GOV 3 Integration of sustainability-related performance in incentive schemes 3.4.2 Remuneration Policy IRO-1, IRO-2 Double Materiality Assessment Note 29 Overview of Consolidation Scope E1-6 Gross Scopes 1, 2, 3 and Total GHG emissions 6.1 Consolidated Statements of Profit or Loss EU Taxonomy Denominator for calculation of turnover Note 16 Segment Reporting and Note 15 Other Operating Income EU Taxonomy Denominator for CapEx calculation Note 4 Property, Plant and Equipment and Note 5 Intangible Assets EU Taxonomy Denominator for OpEx calculation Note 17 Research and Development Expenses and Note 18 Selling, General and Administrative Expenses S1-16 Remuneration metrics (pay gap and total remuneration) 3.4 Remuneration Report and Compensation Statement S1-6 Characteristics of the undertaking’s employees Note 19 Personnel Expenses S4 Patients 1.7.4 Coverage, Pricing and Reimbursement, 1.7.5 Government Pricing and Reimbursement Programs for Marketed Drugs in the U.S., and 1.2.1 Company’s Strategies G1 Business Conduct 3.2.4 Non-Executive Directors ESRS E1 - Climate Change ESRS 2, GOV-3 Integration of sustainability-related performance in incentive schemes Integration of Sustainability-Related Performance Incentive Schemes (GOV-3) ESRS 2 SBM-3 Material Impacts, Risks and Opportunities and Their Interaction with Strategy and Business Model Material Impacts, Risks and Opportunities (SBM-3), Climate Change Mitigation (IRO-1, SBM-3) ESRS 2, IRO-1 Description of the processes to identify and assess material impacts, risks, and opportunities Climate Change Mitigation (IRO-1, SBM-3) E1-1 Transition Plan for Climate Change Mitigation Transition Plan for Climate Change Mitigation (E1-1) E1-2 Policies related to climate change mitigation and adaptation Emissions (E1-2) E1-3 Actions and resources in relation to climate change policies Emissions (E1-3) Disclosure Requirements Section ESRS 2 argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Sustainability Statement 275
E1-4 Targets related to climate change mitigation and adaptation Emissions (E1-4) E1-5 Energy consumption and mix Energy Consumption and Mix (E1-5) E1-6 Gross Scopes 1, 2, 3 and Total GHG emissions Gross Scopes 1, 2, 3 and Total GHG Emissions (E1-6) S1 - Own Workforce ESRS 2 SBM-2 Interests and views of stakeholders Stakeholder Engagement (SBM-2) ESRS 2 SBM-3 Material impacts, risk and opportunities and their interaction with strategy and business models Material Impacts, Risks and Opportunities (SBM-3) S1-1 Policies relating to own workforce Equal Treatment and Opportunities for All (S1-1, MDR-P), Own Workforce Health and Safety (S1-1, MDR-P) S1-2 Processes for engaging with workers about impacts Processes for Engaging With Own Workforce and Workers’ Representatives About Impacts (S1-2) S1-3 Processes to remediate impacts & channels to raise concerns Processes to Remediate Negative Impacts and Channels for Own Workforce to Raise Concerns (S1-3) S1-4 Taking action on material negative impacts, advancing positive impacts, and approaches to mitigating material risks and pursuing material opportunities relating to own workforce Equal Treatment and Opportunities for All (S1-4, MDR-A), Own Workforce Health and Safety (S1-4, MDR-A) S1-5 Targets Equal Treatment and Opportunities for All (S1-5, MDR-T), Own Workforce Health and Safety (S1-5, MDR-T) S1-6 Characteristics of employees Characteristics of Own Employees (S1-6) S1-7 Characteristics of non-employees in the workforce Characteristics of Non-Employees in the Workforce (S1-7) S1-9 Diversity metrics Employee Age Distribution (S1-9), Gender Distribution of Top Management (S1-9) S1-13 Training and skills development metrics Training and Skills Development Metrics (S1-13) S1-14 Health and safety metrics Work-Related Fatalities and Injuries (S1-14) S1-16 Remuneration Metrics Remuneration (Pay Gap and Total Remuneration) (S1-16) S1-17 Incidents, complaints and severe human rights impacts Discrimination Incidents Reported and Complaints Filed (S1-17) S4 - Patients ESRS 2 SBM-3 Materials, impacts, risks and opportunities and their interaction with strategy and business model Material Impacts, Risks and Opportunities (SBM-3) ESRS 2 BP 2-17 Disclosures in relation to specific circumstances Patients (Phase in-relief applied) G1 - Business Conduct ESRS 2 GOV 1 The role of administrative, management and supervisory bodies Sustainability Governance and Oversight (GOV-1, GOV-2) SBM-3 Materials, impacts, risks and opportunities and their interaction with strategy and business model Material Impacts, Risks and Opportunities (SBM-3) G1-1 Business conduct policies and corporate culture Corporate Culture (G1-1, MDR-P), Protection of Whistleblowers (G1-1, MDR-P), Measures to Protect Whistleblowers (G1-1), Animal Welfare (G1-1, MDR-P) G1-2 Management of relationships with suppliers Supply Chain Management (G1-2, MDR-P, G1-2, MDR-A, MDR-T) G1-3 Prevention and detection of corruption and bribery Anti-Corruption and Anti-Bribery (G1-3, MDR-P, G1-3, G1-3, MDR-A) Disclosure Requirements Section ESRS 2 argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Sustainability Statement 276

G1-4 Incidents of corruption of bribery Anti-Corruption and Anti-Bribery (G1- 4, MDR-M) G1-6 Payment practices Payment Practices (G1-6, MDR-T) Disclosure Requirements Section ESRS 2 7.1.2 Basis for Preparation 7.1.2.1. General Basis of Preparation of the Sustainability Statement The scope of consolidation for our 2025 Sustainability Statement aligns with our financial statements, and the reporting period covers January 1 to December 31, 2025. The statement also includes comparative data for the fiscal year ending December 31, 2024 to illustrate year-over-year performance. Select information is incorporated by reference from the annual report and falls within the scope of the limited assurance engagement. For more information on the consolidated accounting group, see Section 1.1.1 “General”, and “Note 29 Overview of Consolidation Scope”. Methodology We reference industry-specific frameworks where applicable, including the Sustainability Accounting Standards Board (SASB) Biotechnology & Pharmaceuticals Standard, to provide stakeholders with comparable and meaningful insights into our sustainability performance. Future iterations of this statement may incorporate methodological refinements as the regulatory environment and related guidance continue to evolve. In defining the scope and boundaries of this Sustainability Statement, impacts, risks, and opportunities were identified across all three areas in line with ESRS 1 Section 5.1. Accordingly, the information presented throughout this statement reflects the full value chain where material impacts, risks, and opportunities were identified. Further information on the extent to which value chain data are required and reported is provided within the relevant topical ESRS sections. We have not omitted any disclosures because of ongoing negotiations, nor have we omitted any information due to reasons of intellectual property. Several assumptions and estimation techniques were used where precise information was unavailable, including cases where third-party data had not yet been provided. Additional details on assumptions and estimation techniques are provided in the relevant accounting policies sections. For example, Scope 3 greenhouse gas (GHG) emission calculations incorporate supplier-reported data, industry averages, and extrapolated estimates. Methodologies for calculating sustainability data, including carbon accounting, continue to evolve. Future reports may reflect refinements to these methodologies, and any significant methodological updates will be disclosed along with their impact on reported figures. 7.1.2.2. Disclosures in Relation to Specific Circumstances Time Horizons The time horizons applied are consistent with those defined by the ESRS for reporting purposes: • Short-term: The period covered by the undertaking's financial statements, typically one year. • Medium-term: From the end of the short-term period up to five years. • Long-term: More than five years. Sources of Estimation and Outcome Uncertainty We have identified areas involving estimation uncertainty, primarily related to environmental and climate data, due to limitations in data availability, evolving methodologies, and reliance on secondary sources. Where significant measurement uncertainty exists, we apply reasonable estimations and disclose them alongside the relevant metric. Additional details on assumptions and estimation techniques are provided in the relevant accounting policies sections. For example, data limitations affected the completeness of information available, increasing uncertainty in our GHG calculations. We have implemented systems to argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Sustainability Statement 277 BP-1 BP-2

estimate and monitor value chain data, covering key metrics such as energy use and GHG emissions. These estimates, which are discussed in more detail in the emissions accounting policies, draw on a combination of supplier data, industry averages, and reasonable estimation methods to approximate upstream and downstream impacts. Actions are underway to improve data availability in future reporting periods. Unless otherwise stated, metrics disclosed have not been validated by an external body. Forward-looking information, including targets and projections, is inherently subject to uncertainty due to potential changes in market, regulatory, technological, and environmental conditions. Despite these limitations, we continue to enhance data quality, transparency, and alignment with recognized sustainability standards. Changes in Preparation or Presentation and Errors in Prior Periods Where restatements are deemed material, they will be disclosed within the accounting policies section of the relevant topical standards, together with an explanation of the underlying reasons. The Company identified a discrepancy in the Scope 3 GHG emissions reported in 2024 due to an inconsistency in information directly provided by a supplier in the prior year. This increased our emissions for our Scope 3 Purchased Goods and Services from 183,781 tCO2e to 236, 582 tCO2e and our Category 4 Upstream Transportation and Distribution emissions from 24,556 to 24,587 tCO2e. The restated emissions can be found in Table E1-6. This affected the percentage of emissions covered by supplier-specific data in 2024, which has been updated and is reflected in the accounting policies. The Company has outlined improvements in its internal control over Sustainability Reporting as disclosed in Section 7.1.4.4. “Risk Management and Internal Controls Over Sustainability Reporting (GOV-5)”. In addition, in 2025, we restated our 2024 energy consumption (E1–5) metrics, due to a methodological change. Application of Transitional Relief In accordance with the European Commission’s “quick-fix” amendment, we have applied the phase-in relief for ESRS S4 Patients. Given the central importance of patients to our business model, we will continue to disclose the information of importance to our patients and users in the S4 section, ensuring that reporting remains focused on the most relevant impacts and stakeholder information needs. 7.1.3 Sustainability Strategy and Our Business Model 7.1.3.1. Business Model, Value Chain and Products Our business model centers on scientific innovation and co-creation, bringing together research, technology, and collaboration to engineer life-changing immunology solutions for patients. We bring antibody-engineering expertise to pioneering researchers to help advance immunology breakthroughs into differentiated medicines. We also collaborate with healthcare providers, regulators, and patient advocacy groups to align our research and commercialization activities with patient needs across regions. Our value chain is structured around two core components: 1. Research and Development (R&D): Includes early-stage research, biotechnology sourcing, pre-clinical studies, clinical trials, and collaboration with CROs and CMOs. These activities culminate in regulatory submissions that enable product commercialization. 2. Commercial Operations: Includes manufacturing, packaging, labeling, global distribution, patient support programs, and end-of-life product management. Sales and marketing activities engage downstream payers and stakeholders. Further information on our products and product candidates can be found in Section 1.1.2 ““Our Medicines”, and Section 1.3 “Our Products and Product Candidates”. Details on headcount and financial performance are available in Section 5.12 “Employees” and Section 6.1 “Consolidated Statements of Financial Position” respectively. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Sustainability Statement 278 SBM-1

7.1.3.2. Sustainability Strategy Our sustainability strategy is grounded in transformational innovation, patient-focused impact, and operational integrity. Our approach is guided by regulatory compliance and supported by internal processes and controls that promote transparency and accountability. Collaboration and thoughtful design are central to our approach. We work across internal functions and with external partners to enhance efficiency and scale impact. We foster a culture that values people across our organization and wider ecosystem, recognizing that continued progress depends on collective effort. Training and development promote continuous improvement and enhance innovation capabilities across teams. By staying aligned with industry developments and an evolving regulatory landscape, we maintain our license to operate and support responsible value creation. Our sustainability strategy provides a foundation for continued innovation and patient-centered progress, supporting our long-term vision and commitment to global health improvement. 7.1.4 Sustainability Governance and Oversight 7.1.4.1. Management of Material Risks, Impacts and Opportunities by Administrative, Management and Supervisory Bodies The Board of Directors, the Company’s highest governance body, operates as a one-tier board under Dutch law. The Board of Directors is collectively responsible for overseeing our general affairs, including governance and oversight of sustainability matters. For information on the composition of the Board of Directors, see Section 3.2 “Management Structure”, Section 3.3 “Report of the Non-Executive Directors” and Section 6.2 “Note 25.3 Relationship and transactions with key personnel”. The Audit and Compliance Committee holds ultimate responsibility for the integrity and design of our sustainability reporting, while specific ESG topics are managed by designated committees, as illustrated in the chart below. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Sustainability Statement 279 GOV-1 GOV-2
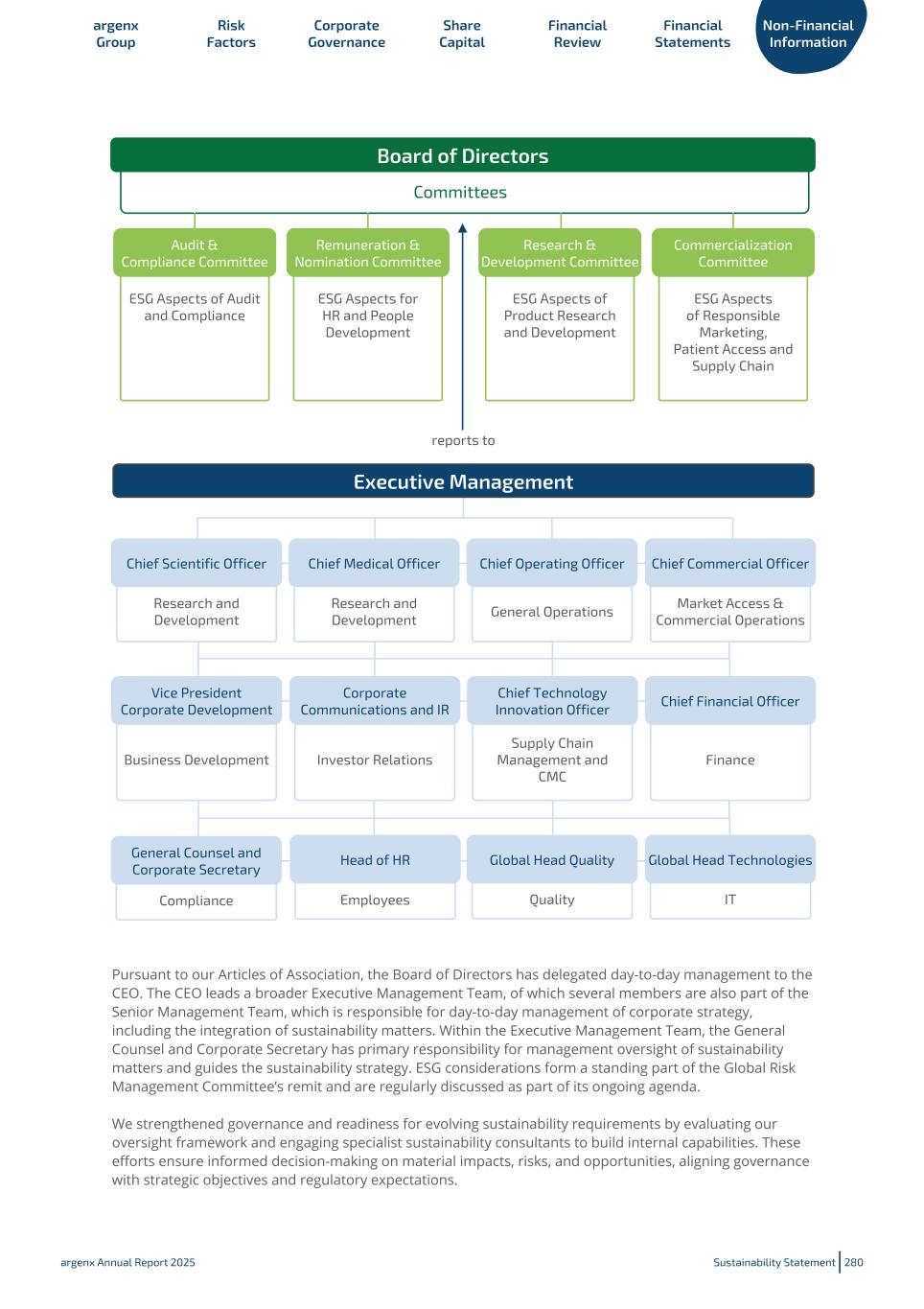
Pursuant to our Articles of Association, the Board of Directors has delegated day-to-day management to the CEO. The CEO leads a broader Executive Management Team, of which several members are also part of the Senior Management Team, which is responsible for day-to-day management of corporate strategy, including the integration of sustainability matters. Within the Executive Management Team, the General Counsel and Corporate Secretary has primary responsibility for management oversight of sustainability matters and guides the sustainability strategy. ESG considerations form a standing part of the Global Risk Management Committee’s remit and are regularly discussed as part of its ongoing agenda. We strengthened governance and readiness for evolving sustainability requirements by evaluating our oversight framework and engaging specialist sustainability consultants to build internal capabilities. These efforts ensure informed decision-making on material impacts, risks, and opportunities, aligning governance with strategic objectives and regulatory expectations. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Sustainability Statement 280 Employees Quality Research and Development Research and Development General Operations Investor Relations Supply Chain Management and CMC Finance ESG Aspects for HR and People Development ESG Aspects of Product Research and Development ESG Aspects of Responsible Marketing, Patient Access and Supply Chain Executive Management ESG Aspects of Audit and Compliance Audit & Compliance Committee Remuneration & Nomination Committee Research & Development Committee Commercialization Committee reports to Chief Scientific Officer Chief Medical Officer Chief Operating Officer Compliance General Counsel and Corporate Secretary Head of HR Global Head Quality Business Development Vice President Corporate Development Corporate Communications and IR Chief Technology Innovation Officer Chief Financial Officer Board of Directors Committees Market Access & Commercial Operations Chief Commercial Officer IT Global Head Technologies

While not formally represented within the administrative, management, or supervisory bodies, employees’ perspectives are incorporated into decision-making through various channels, including regular meetings, feedback sessions, and committee participation. In 2025, the Executive Management Team provided periodic updates to the Audit and Compliance Committee and the Board of Directors, with sustainability topics regularly included on meeting agendas. Key topics discussed in 2025 included regulatory compliance (including the Quick Fix amendment, and more generally the EU Omnibus Simplification Package), corporate culture, scientific innovation, and product affordability and pricing. Discussions addressed both direct and indirect matters related to material impacts, risks, and opportunities. The Board of Directors and Audit and Compliance Committee bring significant industry and compliance expertise that supports oversight of ethical business practices. The Audit and Compliance Committee works closely with the Ethics and Compliance function, receiving quarterly updates on anti-bribery, anti- corruption, and related business-conduct topics to ensure emerging risks are effectively addressed. For more information on our Board of Directors, see Section 3.2.4 “Non-Executive Directors”. 7.1.4.2. Integration of Sustainability-Related Performance Incentive Schemes In 2025, the short-term and long-term incentive compensation for the Board of Directors and Executive Management Team included performance metrics specifically tied to talent retention. For more information on incentive schemes related to talent management, see Section 3.4.3 “NEO Remuneration in FY25”. 7.1.4.3. Due Diligence In 2025, we refined our sustainability due diligence processes to improve the identification, assessment, and management of ESG-related impacts, risks, and opportunities, in alignment with ESRS requirements. We refreshed our double materiality assessment (DMA) to ensure key topics were comprehensively covered and appropriately prioritized across our value chain. The process was documented, reviewed by the Global Risk Management Committee and validated by the Audit and Compliance Committee, ensuring governance oversight of both the process and its outcomes. To improve the quality and consistency of data used in our assessments, we worked closely with internal data owners and functional leads across departments, while engaging with key suppliers and partners to strengthen visibility into upstream and downstream impacts. As part of our ongoing due diligence efforts, we continue to refine our internal control systems for sustainability reporting, integrating them more closely with our broader risk management and governance processes to ensure accuracy, accountability, and continuous improvement. Core Elements of Due Diligence Related Paragraphs Embedding due diligence in governance, strategy and business model GOV-1, GOV-2, GOV-5 Engaging with affected stakeholders in all key steps of the due diligence process ESRS 2 SBM-2, S1-2 Identifying and assessing negative impacts on people and the environment IRO-1, S1-3, G1-2 Taking action to address negative impacts S1-4, S4-4, G1-1, G1-3 Tracking the effectiveness of actions S1-9, S1-14, S1-17, S4 (MDR-M), G1-3, G1-4, G1-6 7.1.4.4. Risk Management and Internal Controls Over Sustainability Reporting Our 2025 Sustainability Statement data collection process was designed to ensure compliance with ESRS requirements, and we established a governance and control framework to support the accuracy, consistency, and transparency of our reporting. We engaged our statutory auditor, EY Accountants B.V., to provide limited assurance as outlined in the independent assurance report. The CFO oversees sustainability reporting and auditing as part of our integrated reporting process. Day-to- day responsibility lies with the Finance Team, which coordinates data collection and validation across the argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Sustainability Statement 281 GOV-4 GOV-3 GOV-5

business, supported by external advisors. Internal controls are embedded in key reporting activities, including data collection, consolidation, review, and approval. We also engaged external advisors to identify, evaluate, and prioritize risks that could affect the quality and completeness of our reporting. This included a combination of top-down and bottom-up reviews, stakeholder interviews, and documentation analysis. The following core risks were identified and are being mitigated as follows: Risk Mitigation Regulatory and legal risks: Non-compliance with evolving CSRD and ESRS requirements. Continuous monitoring of regulatory changes, staff training, and engagement of external advisors. Data accuracy and integrity: Errors from manual inputs or inconsistent data sources. Implementation of validation checks, defined data ownership, and system-based controls. IT and systems risks: Failures or inefficiencies in reporting tools. Use of a secure, centralized reporting platform and regular system testing. Operational and organizational risks: Lack of clear responsibilities or resources. Defined roles, escalation procedures, and management oversight. Findings from risk assessments and internal reviews are integrated into broader risk management and reporting processes. External advisors work closely with Finance, Legal, IT, and Operations to ensure corrective actions are implemented. Controls are refined annually based on audit results, feedback, and lessons learned. 7.1.5 Stakeholder Engagement We engage with a broad range of stakeholders, including patients, healthcare providers, employees, suppliers, and (potential) investors, to understand and incorporate their perspectives into our strategy and business model. Stakeholder engagement is managed through business units and cross-functional teams focused on alliances, partnerships, healthcare professionals, patients, and other stakeholder groups, and is guided by our policy on bilateral contacts and dialogue with shareholders and stakeholders (the Bilateral Shareholders and Stakeholders Contacts Policy). The Bilateral Shareholders and Stakeholders Contacts Policy sets out the principles and processes for engaging with (potential) shareholders and (potential) stakeholders, under which the Board of Directors may confirm the appropriate means for engagement with such (potential) shareholders and (potential) stakeholders. In addition, our Interactions with the Healthcare Community Global Policy outlines the principles and processes for interacting with members of the healthcare community, while our Interactions with the Patient Community Global Policy outlines guidelines for interacting with patient advocacy organizations and the patient community. While external stakeholders were not directly consulted for the double materiality assessment, their perspectives were represented through business units that maintain ongoing dialogue with them. We have outlined below a non-exhaustive list of key elements of our engagement with stakeholders: argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Sustainability Statement 282 SBM-2
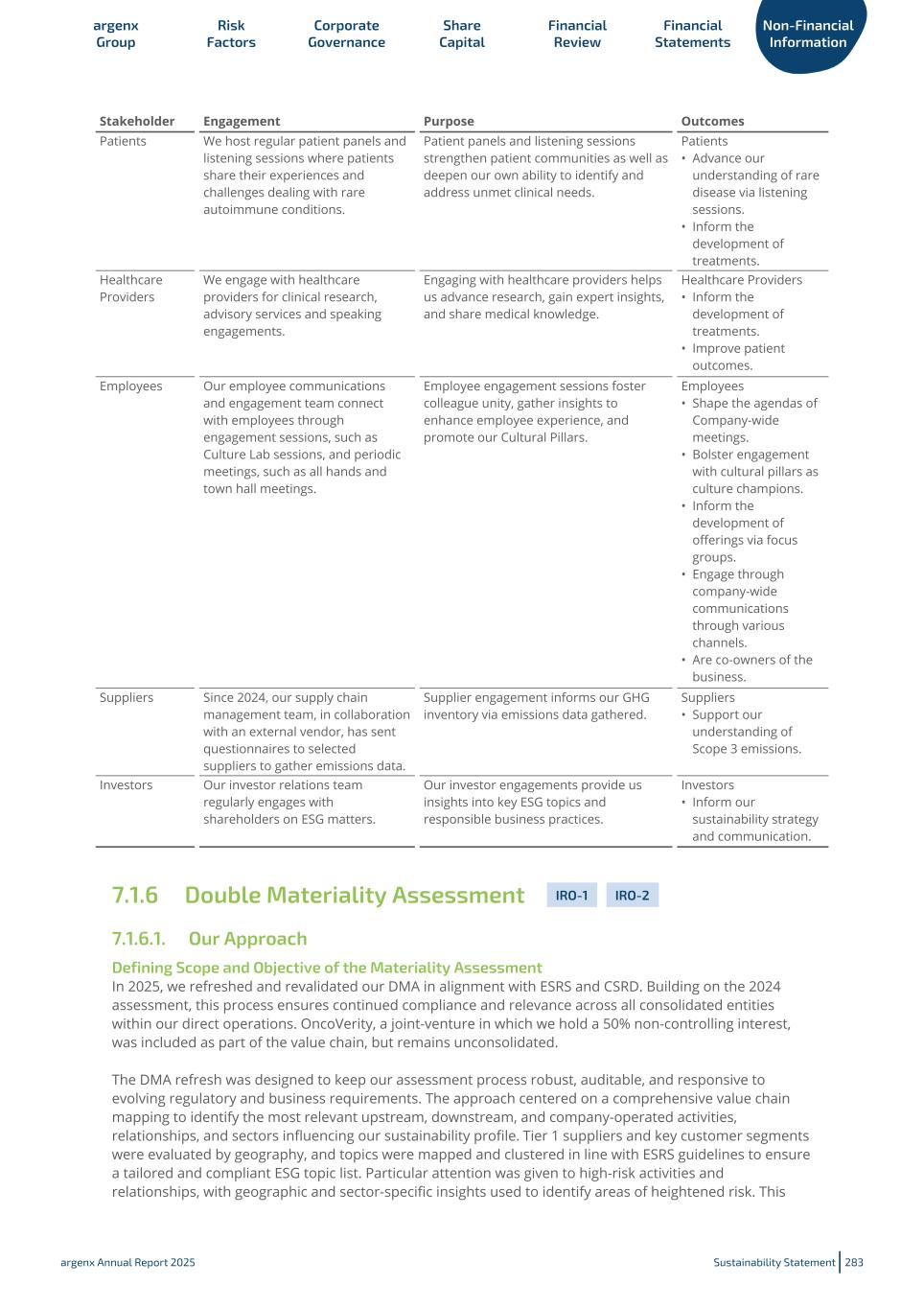
Stakeholder Engagement Purpose Outcomes Patients We host regular patient panels and listening sessions where patients share their experiences and challenges dealing with rare autoimmune conditions. Patient panels and listening sessions strengthen patient communities as well as deepen our own ability to identify and address unmet clinical needs. Patients • Advance our understanding of rare disease via listening sessions. • Inform the development of treatments. Healthcare Providers We engage with healthcare providers for clinical research, advisory services and speaking engagements. Engaging with healthcare providers helps us advance research, gain expert insights, and share medical knowledge. Healthcare Providers • Inform the development of treatments. • Improve patient outcomes. Employees Our employee communications and engagement team connect with employees through engagement sessions, such as Culture Lab sessions, and periodic meetings, such as all hands and town hall meetings. Employee engagement sessions foster colleague unity, gather insights to enhance employee experience, and promote our Cultural Pillars. Employees • Shape the agendas of Company-wide meetings. • Bolster engagement with cultural pillars as culture champions. • Inform the development of offerings via focus groups. • Engage through company-wide communications through various channels. • Are co-owners of the business. Suppliers Since 2024, our supply chain management team, in collaboration with an external vendor, has sent questionnaires to selected suppliers to gather emissions data. Supplier engagement informs our GHG inventory via emissions data gathered. Suppliers • Support our understanding of Scope 3 emissions. Investors Our investor relations team regularly engages with shareholders on ESG matters. Our investor engagements provide us insights into key ESG topics and responsible business practices. Investors • Inform our sustainability strategy and communication. 7.1.6 Double Materiality Assessment 7.1.6.1. Our Approach Defining Scope and Objective of the Materiality Assessment In 2025, we refreshed and revalidated our DMA in alignment with ESRS and CSRD. Building on the 2024 assessment, this process ensures continued compliance and relevance across all consolidated entities within our direct operations. OncoVerity, a joint-venture in which we hold a 50% non-controlling interest, was included as part of the value chain, but remains unconsolidated. The DMA refresh was designed to keep our assessment process robust, auditable, and responsive to evolving regulatory and business requirements. The approach centered on a comprehensive value chain mapping to identify the most relevant upstream, downstream, and company-operated activities, relationships, and sectors influencing our sustainability profile. Tier 1 suppliers and key customer segments were evaluated by geography, and topics were mapped and clustered in line with ESRS guidelines to ensure a tailored and compliant ESG topic list. Particular attention was given to high-risk activities and relationships, with geographic and sector-specific insights used to identify areas of heightened risk. This argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Sustainability Statement 283 IRO-1 IRO-2

targeted focus strengthens the assessment of operations and partnerships and ensures continued alignment with CSRD requirements. To learn more about our consolidation scope, see “Note 29 Overview of Consolidation Scope”. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Sustainability Statement 284 Own Operations argenx is a global immunology company advancing antibody-based therapies for autoimmune diseases through integrated R&D, sourcing innovation, outsourced manufacturing, and expanding commercial operations Upstream argenx’s upstream operations center on early-stage research, working with academic partners, sourcing of biotechnology and raw materials, and strategic partnerships to support antibody discovery and development Downstream argenx’s downstream operations focus on global distribution, patient support, and lifecycle management of its antibody therapies, ensuring broad access and sustained impact Stakeholders Category Activities R&D Commercialization Enabling functions Business Development R&D and Commercialization Employees Investors Regulators Contract Research Organizations Contract Manufacturing Organizations Distributors Patients Healthcare Providers Distribution and Marketing Patient Use Direct suppliers Early-Stage Research and Development Pre-Clinical studies Clinical Trials IP Licensing Direct suppliers Manufacturing Processes (Bulk production, Packaging, Labeling) Direct suppliers Marketing Licensing Product IP Customers Global Distribution Indirect suppliers Sourcing Raw Materials Healthcare Participants Patient Support Programs Patient Product Usage Product End-of-Life Management Drug Discovery and Development Process Commercial Operations

7.1.6.2. Identifying Topics and Impacts, Risks, and Opportunities The identification of impacts, risks, and opportunities (IROs) was guided by an updated assessment of the sustainability challenges and opportunities most relevant to our operations and business activities. Existing sustainability matters were reviewed and validated for completeness and accuracy, with updates made to IRO descriptions, time horizons and value chain attributions where necessary. Stakeholders were consulted to validate the long list of IROs, ensuring that all relevant impacts, risks, and opportunities were captured and appropriately prioritized. IROs were grouped by topic, type, time horizon, geographical scope, and primary impact area, considering both our own operations and value chain activities. Dependencies on natural, human, and social/relationship capital were identified and validated. Potential impacts, risks and opportunities were systematically mapped to the dependencies from which they arise, ensuring each was directly linked to a defined impact. This process reinforces the connection between sustainability dependencies and potential business effects. Stakeholder engagement included direct consultation with internal experts and indirect consultation with credible proxies. Internal stakeholders represented a broad range of functions, while external perspectives were incorporated through desktop research and use of credible proxies. 7.1.6.3. Scoring and Thresholds The 2024 scoring methodology was retained with minor refinements for consistency. Impact materiality was assessed by assigning scores to each IRO on a 1 to 5 scale across scale, scope, and irremediable character which were combined into a severity score. For negative impacts, this severity score was then multiplied by the likelihood of the impact occurring. Financial materiality of risks and opportunities was evaluated through considering likelihood and potential financial impact, aligned with our Enterprise Risk Management framework. Each identified IRO was assessed across three time horizons; short term (0-2 years), medium term (3-5 years), or long term (more than 5 years). Stakeholders were re-engaged during the scoring phase to challenge and validate the scoring of IROs, ensuring that prioritization reflected both internal expertise and external perspectives. Materiality thresholds for both sustainability impacts and financial effects were established using a matrix-based approach grounded in the quantitative scoring results. These thresholds were reviewed, and stakeholder input was used to validate the inclusion of borderline topics. The process and scoring criteria were reviewed and approved by the Senior Management Team and the Board of Directors. Validation workshops provided qualitative feedback and ensured alignment between stakeholder perspectives and scoring outcomes. 7.1.6.4. Strategic Integration of Material IROs Findings were validated by the Senior Management Team and the Board of Directors to ensure alignment with strategic objectives. Material IROs are integrated into our risk management and strategic planning processes, informing the Sustainability Statement and ongoing risk profile. Internal controls, validation workshops, and governance oversight ensure the integrity of the process. Methodologies, input parameters, and assumptions are regularly reviewed and updated to reflect evolving business, regulatory, and sustainability contexts. 7.1.6.5. Process Evolution Compared with the prior year, the 2025 refresh included enhanced documentation, refined value chain mapping, adjustments to IRO time horizons, and strengthened stakeholder engagement. The scoring methodology and thresholds were reviewed and updated to reflect evolving business, regulatory, and sustainability contexts. These refinements improve audit readiness, reporting quality, and alignment with stakeholder priorities. 7.1.6.6. Double Materiality Assessment Results Through this double materiality re-assessment, which addressed both impact and financial materiality in accordance with the ESRS, we identified our material sustainability topics across ESRS categories for disclosure. New IROs were identified in 2025, and the IRO language was refined to improve relevance to our business model and operating environment. Waste-related IROs were removed from scope and deemed argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Sustainability Statement 285 SBM-3

not to be material due to the low volume of waste generated in our business model. This conclusion was informed by waste audits completed as part of the 2024 reporting process. Equal treatment-related IROs (Training and Skills Development and Diversity), which were previously identified as positive impacts in 2024, have been reframed as potential negative impacts in 2025. Animal welfare was added as a new material risk under G1 for 2025. The materiality results are summarized in the following matrix, prepared in line with ESRS 2 SBM-3. For detailed descriptions of our material impacts, risks, and opportunities, see the relevant topical sections. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Sustainability Statement 286 Financial Materiality Double Materiality Impact Materiality No topics Patients* Business Conduct Climate Change Own Workforce E1 S1 S4 G1 *For reporting purposes, we are disclosing information related to the Entity Specific Topic of Innovation under the S4 Patients section.
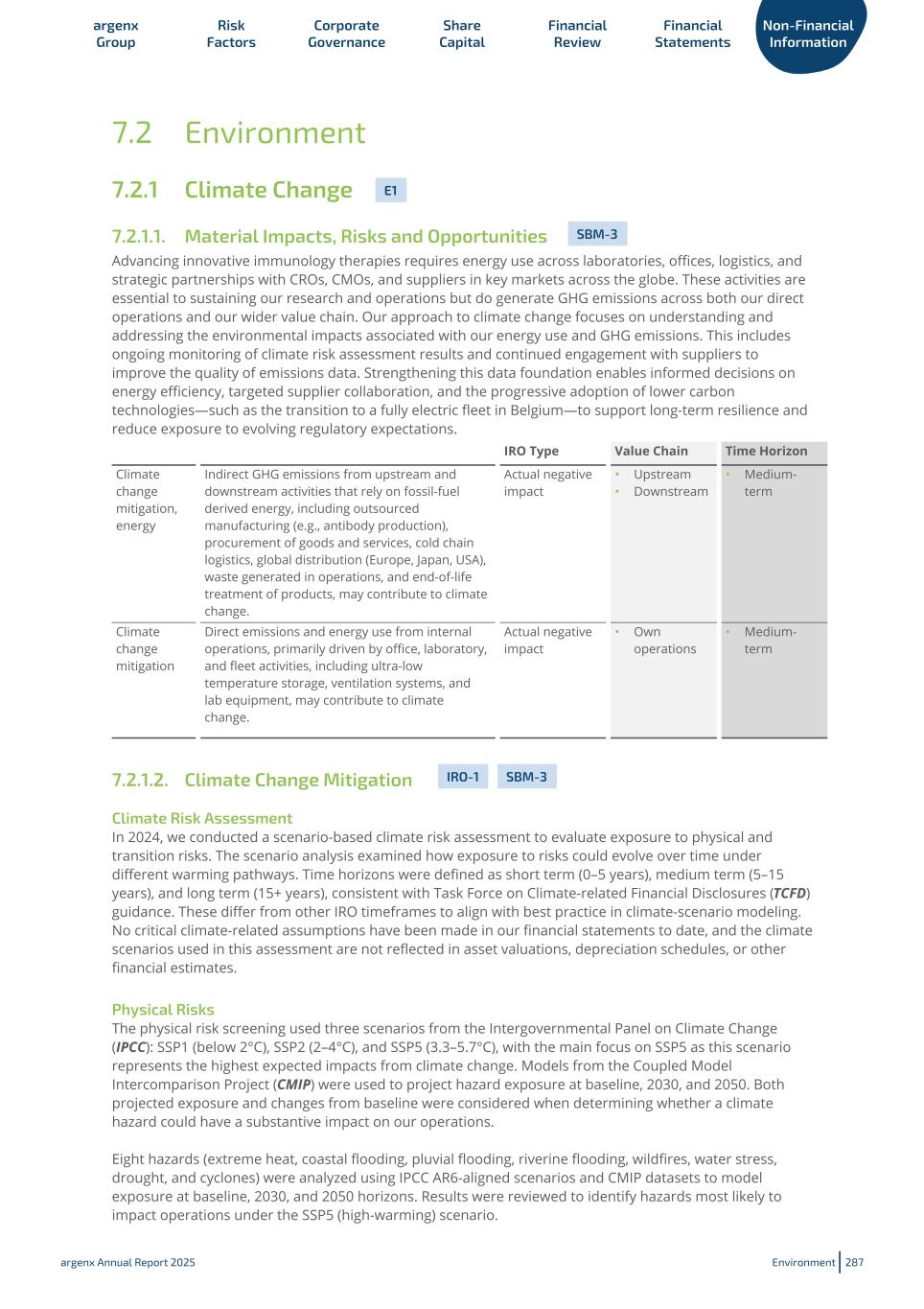
7.2 Environment 7.2.1 Climate Change 7.2.1.1. Material Impacts, Risks and Opportunities Advancing innovative immunology therapies requires energy use across laboratories, offices, logistics, and strategic partnerships with CROs, CMOs, and suppliers in key markets across the globe. These activities are essential to sustaining our research and operations but do generate GHG emissions across both our direct operations and our wider value chain. Our approach to climate change focuses on understanding and addressing the environmental impacts associated with our energy use and GHG emissions. This includes ongoing monitoring of climate risk assessment results and continued engagement with suppliers to improve the quality of emissions data. Strengthening this data foundation enables informed decisions on energy efficiency, targeted supplier collaboration, and the progressive adoption of lower carbon technologies—such as the transition to a fully electric fleet in Belgium—to support long-term resilience and reduce exposure to evolving regulatory expectations. IRO Type Value Chain Time Horizon Climate change mitigation, energy Indirect GHG emissions from upstream and downstream activities that rely on fossil-fuel derived energy, including outsourced manufacturing (e.g., antibody production), procurement of goods and services, cold chain logistics, global distribution (Europe, Japan, USA), waste generated in operations, and end-of-life treatment of products, may contribute to climate change. Actual negative impact • Upstream • Downstream • Medium- term Climate change mitigation Direct emissions and energy use from internal operations, primarily driven by office, laboratory, and fleet activities, including ultra-low temperature storage, ventilation systems, and lab equipment, may contribute to climate change. Actual negative impact • Own operations • Medium- term 7.2.1.2. Climate Change Mitigation Climate Risk Assessment In 2024, we conducted a scenario-based climate risk assessment to evaluate exposure to physical and transition risks. The scenario analysis examined how exposure to risks could evolve over time under different warming pathways. Time horizons were defined as short term (0–5 years), medium term (5–15 years), and long term (15+ years), consistent with Task Force on Climate-related Financial Disclosures (TCFD) guidance. These differ from other IRO timeframes to align with best practice in climate-scenario modeling. No critical climate-related assumptions have been made in our financial statements to date, and the climate scenarios used in this assessment are not reflected in asset valuations, depreciation schedules, or other financial estimates. Physical Risks The physical risk screening used three scenarios from the Intergovernmental Panel on Climate Change (IPCC): SSP1 (below 2°C), SSP2 (2–4°C), and SSP5 (3.3–5.7°C), with the main focus on SSP5 as this scenario represents the highest expected impacts from climate change. Models from the Coupled Model Intercomparison Project (CMIP) were used to project hazard exposure at baseline, 2030, and 2050. Both projected exposure and changes from baseline were considered when determining whether a climate hazard could have a substantive impact on our operations. Eight hazards (extreme heat, coastal flooding, pluvial flooding, riverine flooding, wildfires, water stress, drought, and cyclones) were analyzed using IPCC AR6-aligned scenarios and CMIP datasets to model exposure at baseline, 2030, and 2050 horizons. Results were reviewed to identify hazards most likely to impact operations under the SSP5 (high-warming) scenario. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Environment 287 E1 SBM-3 IRO-1 SBM-3

The assessment covered 43 key locations within our value chain, including own operations (12 office sites), suppliers (28 sites), and customers (3 sites) across North America, Europe, and Asia Pacific. The assessment was conducted at the inherent level, without considering existing adaptation or mitigation measures. Sensitivity to hazards was evaluated based on historical exposure and potential operational disruption. Results may inform strategic and risk management decisions, including contingency planning, supply source diversification, and location selection. For example, office sites were found to have low sensitivity, whereas some contract manufacturers could face greater disruption from extreme weather events. Future assessments may incorporate adaptive measures to further refine understanding of climate resilience. Transition Risks Transition risks were assessed qualitatively across four categories (policy and legal, market, technology, and reputation) based on the TCFD framework. Thirteen sub-categories including carbon pricing, emissions reporting obligations, product regulation, litigation, changing customer behavior, increased cost of raw materials, technology substitution, and reputation factors, were evaluated to understand potential exposure. We analyzed our emissions, revenue, market presence, and stakeholder priority data against the International Energy Agency (IEA) Stated Policies , Announced Pledges, and net zero emissions by 2050 scenarios for 2030, 2040, and 2050. The transition risk screening used all three IEA scenarios, with the main focus on the net zero scenario, as this is where the highest transition risks are expected. These scenarios provide medium- to long-term energy trend projections, allowing us to explore the potential implications of various policy choices, investment trends, and technology dynamics. The assessment assigned a baseline score for each risk reflecting current exposure, and projected future exposure using proxy indicators from the IEA for 2030, 2040, and 2050. The net zero emissions scenario, which aligns with global decarbonization objectives, was prioritized to inform our strategic and regulatory planning. The analysis established baseline exposure levels for integration into our broader risk-management processes and future scenario updates. At this stage, we have not conducted a specific assessment to identify assets or business activities that may be incompatible with a transition to a climate-neutral economy. Results Based on the physical and transition risk assessments, the following climate-related risks were identified as relevant but not material under our double materiality assessment. • Climate-related regulations: Evolving disclosure and reporting requirements (e.g., CSRD in Europe, SB 219 in California, and Australia’s Climate-related Reporting Bill) may increase compliance costs and resource needs. Non-compliance poses potential risks, including litigation and reputational damage. • Raw materials costs: Rising costs and reduced availability of fossil-fuel-derived inputs, driven by carbon pricing or supply disruptions, could affect procurement in the short to medium term. We mitigate these risks by maintaining multiple qualified suppliers across different regions to avoid single-source dependencies and reduce simultaneous exposure to climate events. The following risks were identified as potentially relevant at an inherent level, where exposure to these risks was found to be higher. • Physical (acute): Some sites are expected to experience greater extreme heat under high-warming scenarios by 2030-2050. However, due to existing HVAC systems, heat protocols, and backup generators at R&D sites, the overall operational impact is expected to remain limited. • Physical risk (chronic): Water stress could affect certain sites in high-warming scenarios, although impacts are expected to remain low given current adaptation measures and low water dependence in office operations. Contract manufacturers are expected to bear most potential cost increases. While we have not conducted a standalone resilience analysis, the climate risk assessment considered the effect of existing adaptation and mitigation measures (such as HVAC systems, heat protocols, backup generators, and diversified supply chains) when evaluating the likely operational impact of physical risks. These measures are expected to limit the potential disruption from acute and chronic climate hazards, and their effectiveness was qualitatively assessed following the scenario analysis. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Environment 288

We have applied the phase-in relief under ESRS 1 Appendix C for our second year of Sustainability Statement preparation, allowing the omission of anticipated financial effects. We may continue refining our analysis to enhance understanding of these potential risks and prepare for future disclosure requirements. 7.2.1.3. Transition Plan for Climate Change Mitigation In accordance with paragraph 17 of ESRS E1-1, we have not yet developed a climate transition plan for climate change mitigation. We are monitoring regulatory developments and may adapt our approach once clearer guidance is available. 7.2.2 Emissions 7.2.2.1. Policies Policy Company Car Policy (EMEA) Purpose Defines the principles, rules, and expectations for company cars across EMEA, with regional nuances. Employees must acknowledge reviewing the policy prior to ordering a vehicle. Scope Applies to all employees in EMEA who are eligible for a company car. Most senior level accountable Compensation & Benefits is responsible for policy ownership. Daily fleet operations are managed by Finance Operations in line with the policy. Availability Internally available via the Company intranet and the Fleetpack tool. Process for monitoring An external fleet management provider operates within the parameters of argenx’s car policy and supports compliance through embedded system controls and approval processes. Applicability across sustainability statement Section 7.2.2 “Emissions (E1)” We currently do not have policies linked to managing upstream or downstream emissions. 7.2.2.2. Actions EV Program We are taking steps to reduce the environmental impacts associated with our employee vehicle fleet. In Belgium, charging wall boxes are provided to employees with a company car, supporting the adoption of lower-emission vehicles. Fixed car lists have been implemented for all eligible employees, removing internal combustion engine vehicles and significantly reducing plug-in hybrid electric vehicles . This approach prioritizes electric vehicles and reflects our longer-term plan to transition toward a fully electric fleet in Belgium, contributing to reduced fleet emissions. Charging infrastructure constraints in other countries of operation currently limit the applicability of similar measures outside Belgium. As infrastructure becomes more accessible, we may assess opportunities to expand these actions when feasible. Emission-reduction impacts associated with fleet changes have not yet been quantified. For additional information on how employee travel and commuting contribute to our overall GHG emissions, see Section 7.2.2.4 “Gross Scopes 1, 2, 3 and Total GHG emissions (E1-6). Alinso Building Retrofit We have undertaken a retrofitting program at the PolyTower offices in Ghent to improve energy efficiency and reduce operational emissions. This initiative supports our broader efforts to mitigate climate change and enhance resource efficiency across our facilities. The retrofitting program includes the following measures: 1. HVAC System: Fossil-free system based on reversible heat pumps, managed through a building management system that adjusts ventilation rates based on CO₂ levels and occupancy. 2. Lighting: 100% LED lighting with timer or sensor-based controls. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Environment 289 E1-1 E1-2 E1-3
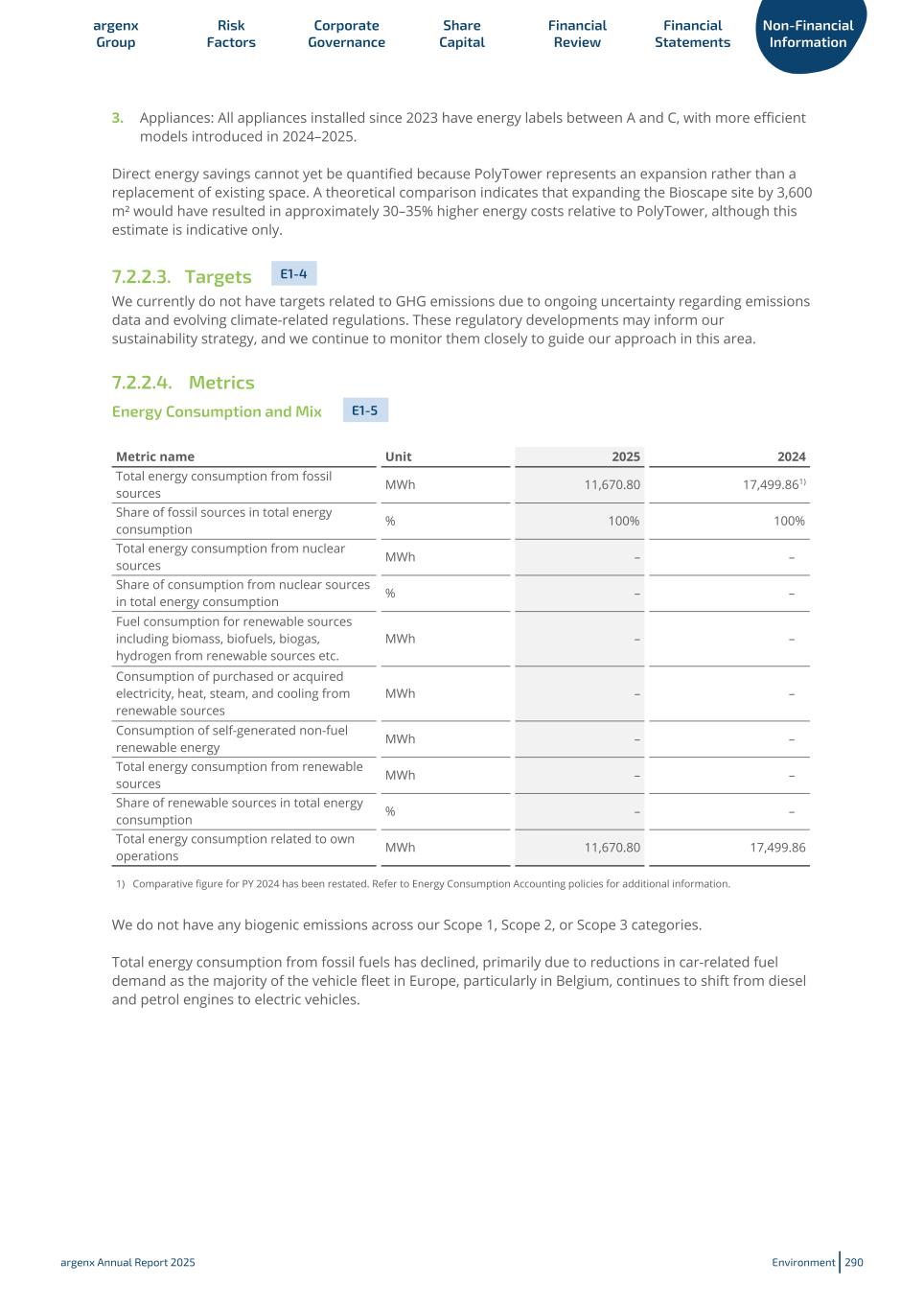
3. Appliances: All appliances installed since 2023 have energy labels between A and C, with more efficient models introduced in 2024–2025. Direct energy savings cannot yet be quantified because PolyTower represents an expansion rather than a replacement of existing space. A theoretical comparison indicates that expanding the Bioscape site by 3,600 m² would have resulted in approximately 30–35% higher energy costs relative to PolyTower, although this estimate is indicative only. 7.2.2.3. Targets We currently do not have targets related to GHG emissions due to ongoing uncertainty regarding emissions data and evolving climate-related regulations. These regulatory developments may inform our sustainability strategy, and we continue to monitor them closely to guide our approach in this area. 7.2.2.4. Metrics Energy Consumption and Mix Metric name Unit 2025 2024 Total energy consumption from fossil sources MWh 11,670.80 17,499.861) Share of fossil sources in total energy consumption % 100% 100% Total energy consumption from nuclear sources MWh – – Share of consumption from nuclear sources in total energy consumption % – – Fuel consumption for renewable sources including biomass, biofuels, biogas, hydrogen from renewable sources etc. MWh – – Consumption of purchased or acquired electricity, heat, steam, and cooling from renewable sources MWh – – Consumption of self-generated non-fuel renewable energy MWh – – Total energy consumption from renewable sources MWh – – Share of renewable sources in total energy consumption % – – Total energy consumption related to own operations MWh 11,670.80 17,499.86 1) Comparative figure for PY 2024 has been restated. Refer to Energy Consumption Accounting policies for additional information. We do not have any biogenic emissions across our Scope 1, Scope 2, or Scope 3 categories. Total energy consumption from fossil fuels has declined, primarily due to reductions in car-related fuel demand as the majority of the vehicle fleet in Europe, particularly in Belgium, continues to shift from diesel and petrol engines to electric vehicles. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Environment 290 E1-4 E1-5
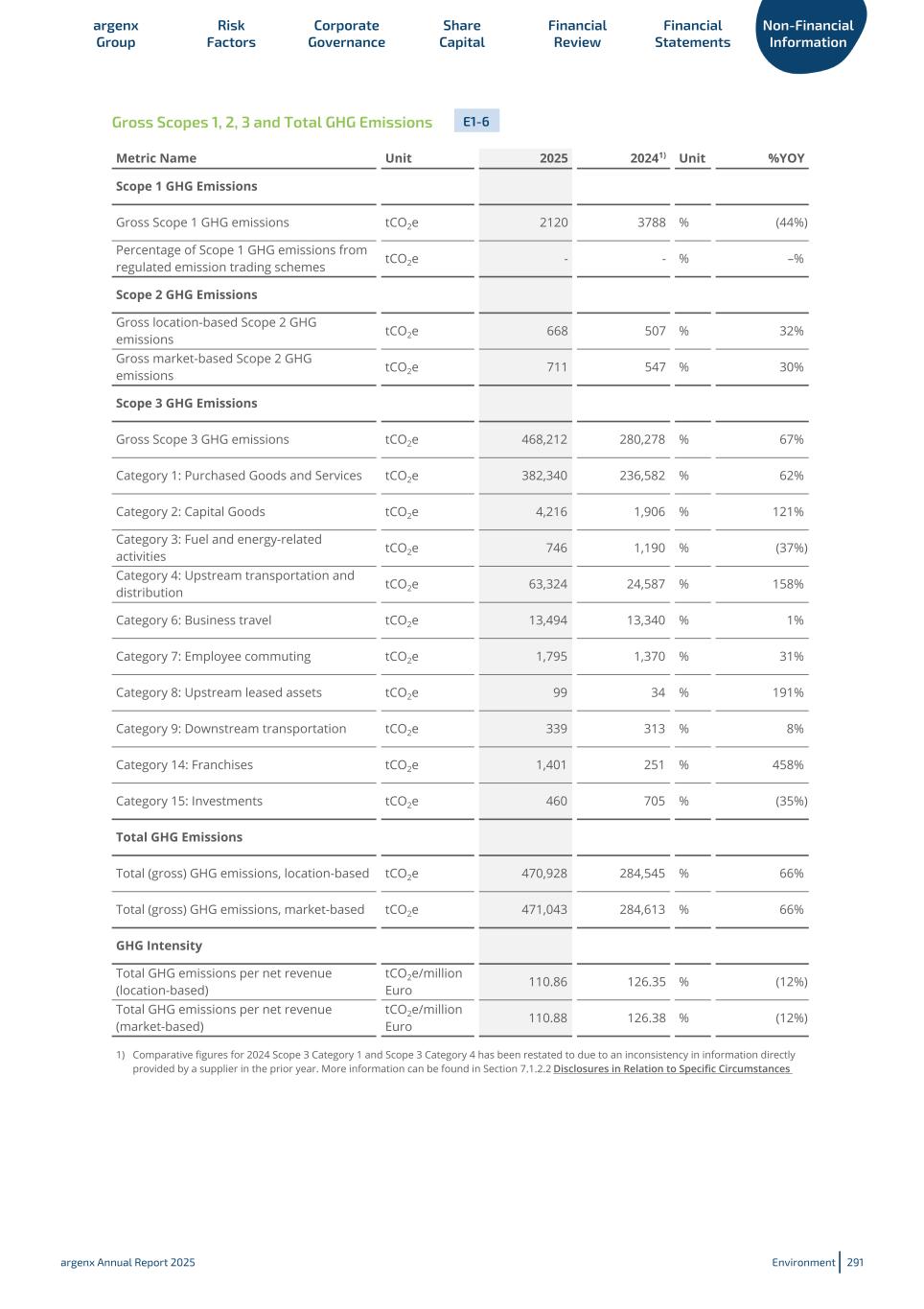
Gross Scopes 1, 2, 3 and Total GHG Emissions Metric Name Unit 2025 20241) Unit %YOY Scope 1 GHG Emissions Gross Scope 1 GHG emissions tCO2e 2120 3788 % (44%) Percentage of Scope 1 GHG emissions from regulated emission trading schemes tCO2e - - % –% Scope 2 GHG Emissions Gross location-based Scope 2 GHG emissions tCO2e 668 507 % 32% Gross market-based Scope 2 GHG emissions tCO2e 711 547 % 30% Scope 3 GHG Emissions Gross Scope 3 GHG emissions tCO2e 468,212 280,278 % 67% Category 1: Purchased Goods and Services tCO2e 382,340 236,582 % 62% Category 2: Capital Goods tCO2e 4,216 1,906 % 121% Category 3: Fuel and energy-related activities tCO2e 746 1,190 % (37%) Category 4: Upstream transportation and distribution tCO2e 63,324 24,587 % 158% Category 6: Business travel tCO2e 13,494 13,340 % 1% Category 7: Employee commuting tCO2e 1,795 1,370 % 31% Category 8: Upstream leased assets tCO2e 99 34 % 191% Category 9: Downstream transportation tCO2e 339 313 % 8% Category 14: Franchises tCO2e 1,401 251 % 458% Category 15: Investments tCO2e 460 705 % (35%) Total GHG Emissions Total (gross) GHG emissions, location-based tCO2e 470,928 284,545 % 66% Total (gross) GHG emissions, market-based tCO2e 471,043 284,613 % 66% GHG Intensity Total GHG emissions per net revenue (location-based) tCO2e/million Euro 110.86 126.35 % (12%) Total GHG emissions per net revenue (market-based) tCO2e/million Euro 110.88 126.38 % (12%) 1) Comparative figures for 2024 Scope 3 Category 1 and Scope 3 Category 4 has been restated to due to an inconsistency in information directly provided by a supplier in the prior year. More information can be found in Section 7.1.2.2 Disclosures in Relation to Specific Circumstances argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Environment 291 E1-6

Accounting Policies The GHG intensity is calculated as the total GHG emissions divided by total operating income. The reported figure for total operating income can be found in Section 6.1 “Consolidated Statements of Profit or Loss”. We define our organizational boundaries using the Operational Control approach, as outlined in the GHG Protocol developed by the World Resources Institute and World Business Council for Sustainable Development (WBCSD). Under this approach, we account for 100% of GHG emissions from operations under our control. Emissions from our joint venture, OncoVerity, are included in Scope 3, Category 15 – Investments. Energy Consumption As seen in the E1-5 metrics table, 100% of the purchased electricity, heat, steam and cooling comes from fossil sources. In 2025, there was no energy consumption purchased from renewable sources or on-site self-generated. We have revised the energy consumption calculations to only capture facilities under scope 1 and Scope 2 and subsequently restated our 2024 figures to align with this approach. Scope 1 Emissions Scope 1 emissions include all direct GHG emissions associated with sources owned or controlled by the Company. Our Scope 1 emissions are primarily associated with leased employee vehicles. As all sites are leased, emissions from purchased heating and cooling are classified under Scope 2, consistent with the GHG Protocol Scope 2 Guidance ("Identifying Scope 2 Emissions and Setting the Scope 2 Boundary"). In 2025, vehicle fuel consumption was estimated using average daily fuel consumption from 2024. Leased vehicle data, including contract dates and fuel type, was used to calculate the number of days each vehicle was in use during 2025. This was multiplied by the corresponding 2024 average daily fuel consumption for each fuel type to estimate total fuel consumption for the year. Scope 2 Emissions Scope 2 emissions include indirect GHG emissions from purchased or acquired energy such as electricity, heating, and cooling, and electricity used to charge leased electric vehicles. These emissions occur at the point of generation rather than within our operations. Although we do not own or control these sources, they result from our energy consumption. We collect invoiced utility data for properties under operational control. Where data is unavailable, consumption is estimated using floor area and energy intensity benchmarks from the Better Buildings Partnership and the World Bank. For facilities with reported data, information is obtained from landlords, utility bills, or supplier invoices. In addition, because the data collection and calculations were completed in Q4 2025, some facilities’ reported energy consumption data for Q4 was missing. The missing values were estimated using average consumption from comparable months with available data. Electricity-related emissions are calculated by multiplying total site energy consumption by the relevant regional or country-specific emission factors. Location-based factors are sourced from the IEA, CO₂emissiefactoren, and the U.S. Environmental Protection Agency (EPA) eGRID. As we do not currently procure renewable energy, market-based emissions reflect the residual mix where available and default to location-based factors where residuals are not available, following the GHG Protocol market-based emission factor hierarchy. We also track the mileage for our leased electric vehicles which are charged using grid electricity. Data collection and emissions calculations for these vehicles follow a same approach to that used for vehicle fuel consumption reported under Scope 1. Emissions from the electricity used to charge leased electric vehicles are reported under Scope 2, in line with the GHG Protocol Scope 2 Guidance. Emissions from heating and cooling systems (e.g., HVAC or boilers) are calculated using annual facility consumption data and emission factors from the UK Department for Energy Security and Net Zero and global warming potentials from the IPCC AR6. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Environment 292

Location-based emissions represent grid-average factors for the regions where we operate, while market- based emissions reflect our purchasing choices. When direct data are unavailable, estimates are based on IEA and Department for Environment, Food and Rural Affairs (DEFRA) factors, with residual factors applied where available. Scope 3 Emissions Scope 3 emissions account for all other indirect emissions across our value chain. In line with WBCSD and GHG Protocol best practices, we engage external partners to support the development of our Scope 3 emissions inventory by collecting supplier-reported emissions data. This resulted in 48% supplier-specific data coverage, remaining consistent with 2024. Our Scope 3 emissions include the following categories and methodologies: • Category 1 – Purchased Goods and Services (PG&S): Includes emissions from the production of goods and services procured by argenx. A hybrid approach combines supplier-specific data which account for 57% of PG&S emissions data (compared to 55% in 2024). For the remaining spend, emissions were estimated using Environmentally Extended Input-Output (EEIO) emission factors from the EPA Supply Chain Emission Factors, applied to cash-based, cost-incurred financial data. Due to limited granularity in spend categories, a supplier-based approach was applied, with spend matched to appropriate EEIO factors using inflation-adjusted values. In line with the principle of materiality, the inventory prioritized suppliers representing the top 90% of total spend. To allocate emissions between Category 1 (Purchased Goods and Services) and Category 4 (Upstream Transportation and Distribution), historical proxy percentages from the FY24 spend report were used. • Category 2 – Capital Goods: Includes upstream (cradle-to-gate) emissions from the production of capital goods purchased or acquired during the reporting year. Emissions are calculated using a spend-based EEIO analysis, applying EPA Supply Chain Emission Factors and financial data. • Category 3 – Fuel- and Energy-Related Activities Not Included in Scope 1 or 2: Represents upstream emissions from fuels and energy used in Scopes 1 and 2, calculated using an average-data approach. Calculations are based on Scope 1 and 2 consumption, with fuel factors from DEFRA and electricity factors sourced from the IEA and country-specific sources where available. • Category 4 – Upstream Transportation and Distribution: Calculated on a well-to-wheel (WTW) basis in alignment with the Science Based Targets initiative (SBTi), covering the extraction, refinement, distribution, and combustion of fuels. Supplier-specific emissions data were applied where available, representing approximately 6% of total Category 4 emissions (down from 14% in 2024). Where supplier data were unavailable, emissions were estimated using distance-based calculations with DEFRA emission factors, and spend-based estimates using EPA Supply Chain Emission Factors where distance data could not be obtained. Spend-based calculations were derived from cost-incurred financial data. Consistent with the FY24 GHG inventory, assumptions were made that pallet weights and Biocair transportation lane distances remained unchanged. • Category 6 – Business Travel: Includes air, rail, car, and other business travel (e.g., ride-share, rental, taxi), calculated on a WTW basis using distance- or spend-based data in alignment with the SBTi. Emissions from hotel stays, while optional under the GHG Protocol, have been included using a spend- based approach which relies upon our cash-out financial data as the underlying source for these calculations. Air-travel data are exported from Business Travel Insights, categorizing flights by haul length (short, medium, long) and cabin class; distances are multiplied by DEFRA well-to-tank (WTT) and tank-to-wheel (TTW) emission factors. • Category 7 – Employee Commute and Work from Home (WFH): Employee commute emissions are calculated using a distance-based methodology based on employee location and regional transport patterns, on a WTW basis in alignment with the SBTi and applying DEFRA emission factors. Emissions from teleworking (WFH), while optional under the GHG Protocol, have been included by estimating the incremental increase in household energy use and remote-work frequency. Emissions from leased commuting vehicles are excluded to avoid double counting with Scope 1. • Category 8 – Upstream Leased Assets: Includes shared workspaces; emissions are calculated consistent with Scopes 1 and 2 methodologies, using actual activity data where available and estimates otherwise. Emission factors are primarily sourced from the IEA and DEFRA, with residual factors used for market- based calculations where available. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Environment 293

• Category 9 – Downstream Transportation and Distribution: Represents outbound transportation not paid for by us, calculated on a distance-based WTW basis in alignment with the SBTi and using DEFRA emission factors. • Category 14 – Franchises: Reflects the license granted to Zai Lab to sell and distribute VYVGART in China in return for sales-based royalties and a one-time milestone payment. Emissions are calculated using the franchise-specific method, based on Zai Lab’s Scope 1 and 2 data allocated to us. • Category 15 – Investments: Includes emissions from the OncoVerity joint venture (calculated using the average-data method and EPA Supply Chain Emission Factors) and Zai Lab (calculated using the investment-specific method). The following categories have been excluded: • Category 5 - Waste: Not relevant; our emissions related to waste is minimal. • Category 10 – Processing of Sold Products: Not applicable; we do not sell intermediate products. • Category 11 – Use of Sold Products: Not applicable; our products do not consume energy. • Category 12 – End of Life Treatment of Sold Products: Not relevant; our emissions related to the disposal of sold products is minimal. • Category 13 – Downstream Leased Assets: Not applicable; we do not lease assets to other entities. 7.2.3 EU Taxonomy 7.2.3.1. Introduction to the EU Taxonomy Regulation The EU Taxonomy is a classification system for environmentally sustainable economic activities. It provides a common framework for determining when an activity contributes substantially to one or more environmental objectives, does no significant harm to others, and complies with minimum social safeguards. By defining these technical screening criteria, the Taxonomy aims to direct investments into sustainable activities, increase transparency and improve comparability. The EU Taxonomy Regulation identifies six environmental objectives: 1. Climate change mitigation 2. Climate change adaptation 3. Sustainable use and protection of water and marine resources 4. Transition to a circular economy 5. Pollution prevention and control 6. Protection and restoration of biodiversity and ecosystems As a non-financial undertaking, argenx is required to disclose the proportion of its turnover, capital expenditure (CapEx) and operational expenditure (OpEx) associated with Taxonomy-eligible or Taxonomy- aligned economic activities listed under these six objectives. The methodology applied has remained consistent with the previous reporting period, ensuring comparability and continuity across reporting periods. In 2025, argenx closely monitored the development of the Omnibus Delegated Act, which simplifies the EU Taxonomy Regulation and entered into force on 28 January 2026. Although the Delegated Act applies retrospectively from 1 January 2026, Article 4 provides a transitional option allowing reporting undertakings to continue applying the earlier reporting rules for the 2025 financial year. For the purposes of this report, argenx has exercised this transitional option and will adopt the amended rules in the next reporting period. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Environment 294

7.2.3.2. Eligibility and Alignment Eligibility In 2025, we reviewed our activities against the economic activities listed under the six environmental objectives covered by the Climate, Environmental and Complementary Climate Delegated Acts. Potential eligible activities were identified through an initial screening process of all activities and finalized based on the activity descriptions in the Delegated Acts. There were no notable changes in our assessment from the previous financial year. Two eligible activities were identified as relevant: • 1.2. Manufacture of medicinal products (Pollution prevention and control) • 6.5. Transport by motorbikes, passenger cars and light commercial vehicles (Climate Change Mitigation). These correspond to our turnover derived from sales of medicinal products and R&D activities (associated with activity 1.2), and to vehicle leases (associated with activity 6.5). argenx Eligible Activities Economic Activity Environmental Objective Description of argenx’s Economic Activities KPI 1.2. Manufacture of medicinal products Pollution prevention and control Contract manufacturing of medicinal products Research and development activities related to medicinal products Turnover, OpEx 6.5. Transport by motorbikes, passenger cars and light commercial vehicles Climate change mitigation Vehicles leases CapEx Alignment The Taxonomy assessment was conducted in collaboration with legal, financial, and internal ESG experts, with additional support from external specialists. The Minimum Safeguards establish criteria to ensure entities carrying out environmentally sustainable activities labeled as Taxonomy-aligned meet certain social and governance standards. These criteria are centered around four key themes: human rights, corruption, taxation and fair competition. We conducted a thorough assessment of whether it meets the Minimum Safeguards criteria as laid out in the Final Report on Minimum Safeguards published by the EU platform on Sustainable Finance in October 2022. We are considered compliant with criteria related to corruption, taxation, and fair competition through our Global Tax Policy and Code of Business Conduct and Ethics, which covers human rights, anti-corruption, and bribery as well as fair competition. No breaches of the Minimum Safeguards were identified. We remain committed to respecting human rights and working with partners who share this commitment. However, we have not yet implemented a formal Human Rights Due Diligence process fully aligned with the six-step approach outlined in the UN Guiding Principles on Business and Human Rights (UNGPs) and the OECD Guidelines, as required by the Minimum Safeguards criteria. Based on this outcome, full alignment with Taxonomy requirements for turnover and OpEx associated with activity 1.2 (Manufacture of medicinal products), and CapEx associated with activity 6.5 (Transport by motorbikes, passenger cars and light commercial vehicles) could not be demonstrated at this time. Accordingly, we have reported zero percent alignment for turnover and OpEx KPIs. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Environment 295

KPI Eligible (USD million) Aligned (USD million) Non-eligible (USD million) 2025 2024 2025 2024 2025 2024 Turnover 4,151.3 (99.9%) 2,185.9 (99.8%) 0 (0%) 0 (0%) 2.2 (0.1%) 4.3 (0.2%) CapEx 10.4 (7.8%) 5.5 (5.8%) 0 (0%) 0 (0%) 122.3 (92.2%) 89.2 (94.2%) OpEx 859.2 (99.7%) 605.1 (99.9%) 0 (0%) 0 (0%) 2.3 (0.3%) 0.7 (0.1%) Accounting Policies Turnover Turnover consists of net turnover derived from products or services. In line with our revised approach to assessing taxonomy eligibility for turnover during the previous financial year, contract manufacturing is included in the KPI calculation. Revenue from sales of the manufactured products (arising from the economic activity 1.2.) is considered eligible. We fully recognizes the revenue under the principles set out in IFRS 15, and therefore, all product net sales are considered eligible under activity 1.2. Manufacture of medicinal products. Numerator: Consists of the external product net sales (associated with activity 1.2. Manufacture of medicinal products) and totals $4.2 billion. Denominator: Consists of product net sales and collaboration revenue (as listed in Annex I, point 1.1.1 of Disclosures Delegated Act), and totals $4.2 billion. Refer to ‘‘Note 16 Segment Reporting’’ and “Note 15 Other Operating Income’’ in the consolidated financial statements. CapEx CapEx covers additions to tangible and intangible assets including right-of-use assets during the fiscal year considered before depreciation, amortization, and any re-measurements. We have considered leased vehicles that result in the recognition of a right-of-use of asset and are recognized under IFRS 16 Leases as eligible CapEx per the definition in Taxonomy Disclosures Delegated Act. All leased vehicles are considered eligible under 6.5. Transport by motorbikes, passenger cars and light commercial vehicles. Numerator: Additions to leased vehicles (associated with activity 6.5. Transport by motorbikes, passenger cars and light commercial vehicles), totaling $10.4 million. Denominator: Additions to tangible and intangible assets during the fiscal year (as listed in Annex I, point 1.1.2.1 of Disclosures Delegated Act), totaling $132.7 million. Refer to “Note 4 Property, Plant and Equipment” and “Note 5 Intangible Assets” in the consolidated financial statements. OpEx OpEx covers direct non-capitalized costs related to research and development, building renovation measures, short-term lease, maintenance and repair, and any other direct expenditures relating to the day- to-day servicing of assets of property, plant, and equipment. We considered direct costs related to research and development associated with activity 1.2. Manufacture of medicinal products as eligible OpEx. Research and development are a key activity in our strategic business model and value chain. It consists of multi-phase clinical trials, regulatory approval processes, research of pre-clinical stage product candidates, and discovery stage programs, all with the eventual goal to manufacture medicinal products and treat patients globally. Please refer to “Note 17 Research and Development Expenses” for eligible R&D expenses. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Environment 296

For 2025, specifically, R&D related to evaluating the use of efgartigimod in 15 severe autoimmune diseases (including MG, CIDP, and ITP), empasiprubart is currently being evaluated in four diseases, proof-of-concept clinical trials in adimanebart, and other pre-clinical research, was considered eligible OpEx. Numerator: Direct R&D expenses related to efgartigimod and other pre-clinical candidates (associated with activity 1.2. Manufacture of medicinal products) totaling $859.2 million. Denominator: R&D, maintenance, and repair (as listed in Annex I, point 1.1.3.1 of Disclosures Delegated Act), totaling $861.4 million. For the denominator, refer to “Note 17 Research and Development Expenses" and” “Note 18 Selling, General and Administrative Expenses’’ in the consolidated financial statements. Double counting is avoided as none of the eligible activities contribute to multiple environmental objectives and each KPI includes only one eligible activity. 7.2.3.3. Changes From the Previous Reporting Period We reviewed the taxonomy-eligibility and alignment of our economic activities under all six environmental objectives. No notable changes were identified compared to the previous fiscal year. Row Nuclear energy related activities 1 The undertaking carries out, funds, or has exposures to research, development, demonstration and deployment of innovative electricity generation facilities that produce energy from nuclear processes with minimal waste from the fuel cycle. NO 2 The undertaking carries out, funds, or has exposures to construction and safe operation of new nuclear installations to produce electricity or process heat, including for the purposes of district heating or industrial processes such as hydrogen production, as well as their safety upgrades, using best available technologies. NO 3 The undertaking carries out, funds, or has exposures to safe operation of existing nuclear installations that produce electricity or process heat, including for the purposes of district heating or industrial processes such as hydrogen production from nuclear energy, as well as their safety upgrades. NO Fossil gas related activities 4 The undertaking carries out, funds, or has exposures to construction or operation of electricity generation facilities that produce electricity using fossil gaseous fuels. NO 5 The undertaking carries out, funds, or has exposures to construction, refurbishment, and operation of combined heat/cool and power generation facilities using fossil gaseous fuels. NO 6 The undertaking carries out, funds, or has exposures to construction, refurbishment and operation of heat generation facilities that produce heat/cool using fossil gaseous fuels. NO argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Environment 297
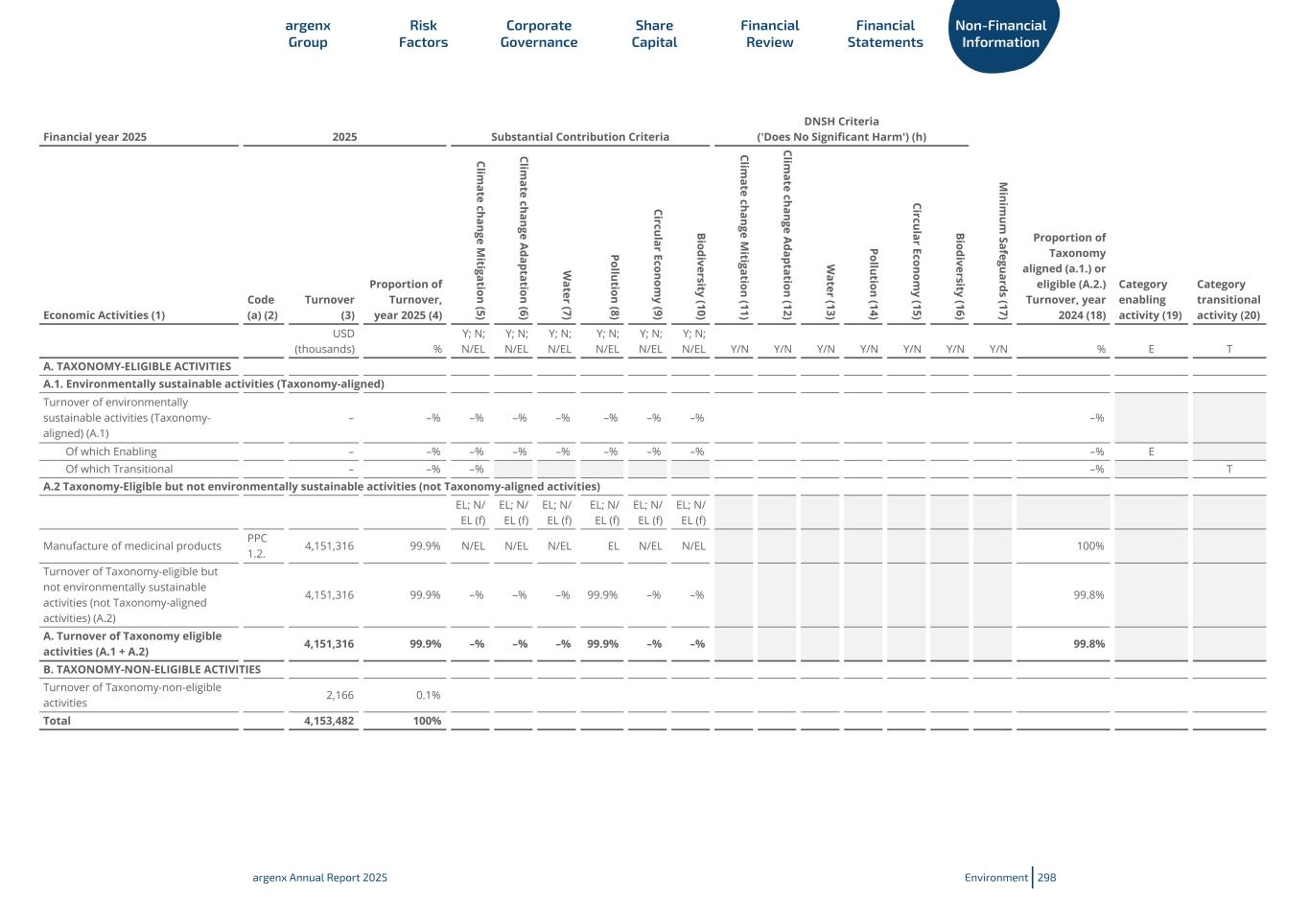
Financial year 2025 2025 Substantial Contribution Criteria DNSH Criteria ('Does No Significant Harm') (h) Economic Activities (1) Code (a) (2) Turnover (3) Proportion of Turnover, year 2025 (4) Clim ate change M itigation (5) Clim ate change A daptation (6) W ater (7) Pollution (8) Circular Econom y (9) Biodiversity (10) Clim ate change M itigation (11) Clim ate change A daptation (12) W ater (13) Pollution (14) Circular Econom y (15) Biodiversity (16) M inim um Safeguards (17) Proportion of Taxonomy aligned (a.1.) or eligible (A.2.) Turnover, year 2024 (18) Category enabling activity (19) Category transitional activity (20) USD (thousands) % Y; N; N/EL Y; N; N/EL Y; N; N/EL Y; N; N/EL Y; N; N/EL Y; N; N/EL Y/N Y/N Y/N Y/N Y/N Y/N Y/N % E T A. TAXONOMY-ELIGIBLE ACTIVITIES A.1. Environmentally sustainable activities (Taxonomy-aligned) Turnover of environmentally sustainable activities (Taxonomy- aligned) (A.1) – –% –% –% –% –% –% –% –% Of which Enabling – –% –% –% –% –% –% –% –% E Of which Transitional – –% –% –% T A.2 Taxonomy-Eligible but not environmentally sustainable activities (not Taxonomy-aligned activities) EL; N/ EL (f) EL; N/ EL (f) EL; N/ EL (f) EL; N/ EL (f) EL; N/ EL (f) EL; N/ EL (f) Manufacture of medicinal products PPC 1.2. 4,151,316 99.9% N/EL N/EL N/EL EL N/EL N/EL 100% Turnover of Taxonomy-eligible but not environmentally sustainable activities (not Taxonomy-aligned activities) (A.2) 4,151,316 99.9% –% –% –% 99.9% –% –% 99.8% A. Turnover of Taxonomy eligible activities (A.1 + A.2) 4,151,316 99.9% –% –% –% 99.9% –% –% 99.8% B. TAXONOMY-NON-ELIGIBLE ACTIVITIES Turnover of Taxonomy-non-eligible activities 2,166 0.1% Total 4,153,482 100% argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Environment 298
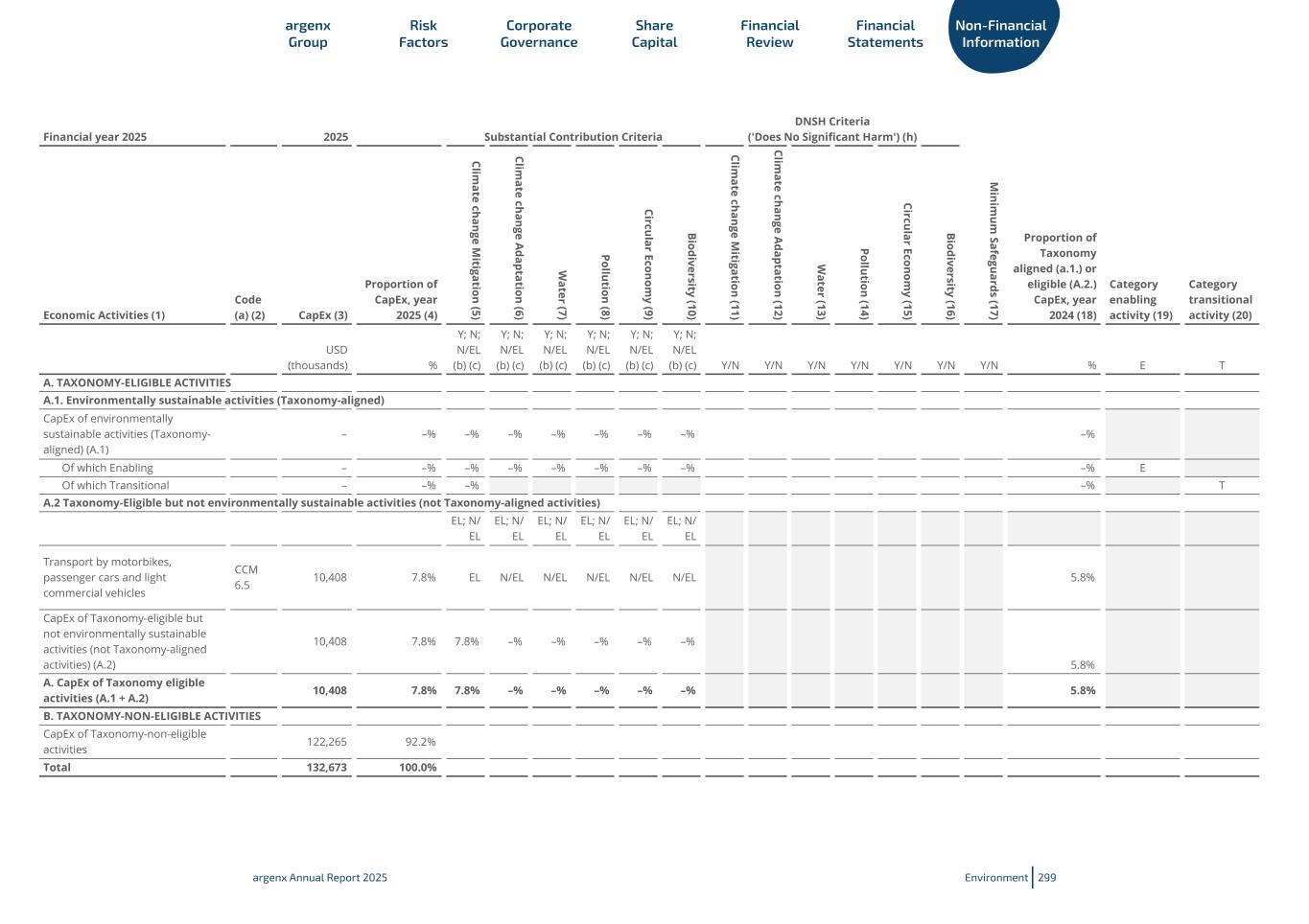
Financial year 2025 2025 Substantial Contribution Criteria DNSH Criteria ('Does No Significant Harm') (h) Economic Activities (1) Code (a) (2) CapEx (3) Proportion of CapEx, year 2025 (4) Clim ate change M itigation (5) Clim ate change A daptation (6) W ater (7) Pollution (8) Circular Econom y (9) Biodiversity (10) Clim ate change M itigation (11) Clim ate change A daptation (12) W ater (13) Pollution (14) Circular Econom y (15) Biodiversity (16) M inim um Safeguards (17) Proportion of Taxonomy aligned (a.1.) or eligible (A.2.) CapEx, year 2024 (18) Category enabling activity (19) Category transitional activity (20) USD (thousands) % Y; N; N/EL (b) (c) Y; N; N/EL (b) (c) Y; N; N/EL (b) (c) Y; N; N/EL (b) (c) Y; N; N/EL (b) (c) Y; N; N/EL (b) (c) Y/N Y/N Y/N Y/N Y/N Y/N Y/N % E T A. TAXONOMY-ELIGIBLE ACTIVITIES A.1. Environmentally sustainable activities (Taxonomy-aligned) CapEx of environmentally sustainable activities (Taxonomy- aligned) (A.1) – –% –% –% –% –% –% –% –% Of which Enabling – –% –% –% –% –% –% –% –% E Of which Transitional – –% –% –% T A.2 Taxonomy-Eligible but not environmentally sustainable activities (not Taxonomy-aligned activities) EL; N/ EL EL; N/ EL EL; N/ EL EL; N/ EL EL; N/ EL EL; N/ EL Transport by motorbikes, passenger cars and light commercial vehicles CCM 6.5 10,408 7.8% EL N/EL N/EL N/EL N/EL N/EL 5.8% CapEx of Taxonomy-eligible but not environmentally sustainable activities (not Taxonomy-aligned activities) (A.2) 10,408 7.8% 7.8% –% –% –% –% –% 5.8% A. CapEx of Taxonomy eligible activities (A.1 + A.2) 10,408 7.8% 7.8% –% –% –% –% –% 5.8% B. TAXONOMY-NON-ELIGIBLE ACTIVITIES CapEx of Taxonomy-non-eligible activities 122,265 92.2% Total 132,673 100.0% argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Environment 299
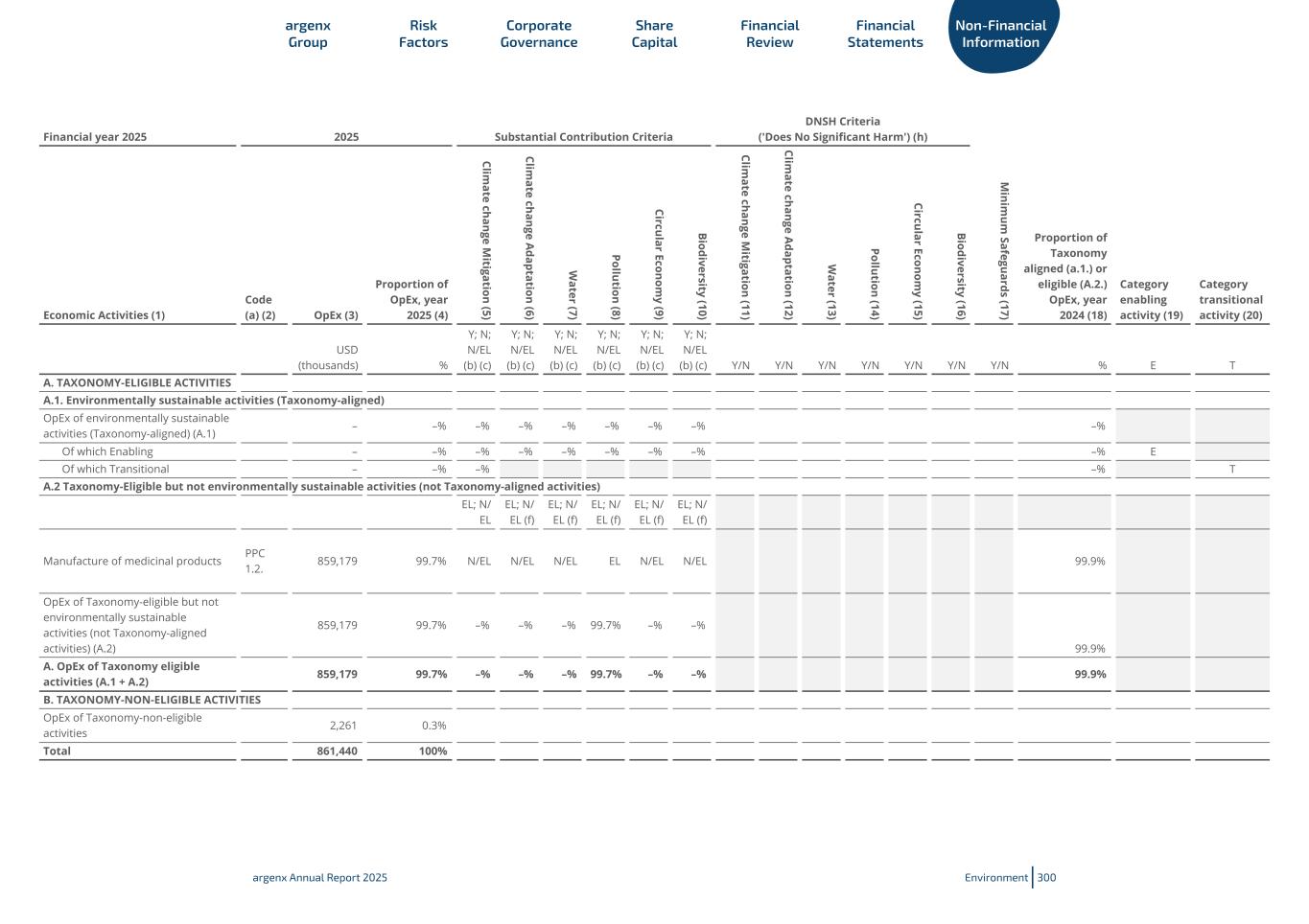
Financial year 2025 2025 Substantial Contribution Criteria DNSH Criteria ('Does No Significant Harm') (h) Economic Activities (1) Code (a) (2) OpEx (3) Proportion of OpEx, year 2025 (4) Clim ate change M itigation (5) Clim ate change A daptation (6) W ater (7) Pollution (8) Circular Econom y (9) Biodiversity (10) Clim ate change M itigation (11) Clim ate change A daptation (12) W ater (13) Pollution (14) Circular Econom y (15) Biodiversity (16) M inim um Safeguards (17) Proportion of Taxonomy aligned (a.1.) or eligible (A.2.) OpEx, year 2024 (18) Category enabling activity (19) Category transitional activity (20) USD (thousands) % Y; N; N/EL (b) (c) Y; N; N/EL (b) (c) Y; N; N/EL (b) (c) Y; N; N/EL (b) (c) Y; N; N/EL (b) (c) Y; N; N/EL (b) (c) Y/N Y/N Y/N Y/N Y/N Y/N Y/N % E T A. TAXONOMY-ELIGIBLE ACTIVITIES A.1. Environmentally sustainable activities (Taxonomy-aligned) OpEx of environmentally sustainable activities (Taxonomy-aligned) (A.1) – –% –% –% –% –% –% –% –% Of which Enabling – –% –% –% –% –% –% –% –% E Of which Transitional – –% –% –% T A.2 Taxonomy-Eligible but not environmentally sustainable activities (not Taxonomy-aligned activities) EL; N/ EL EL; N/ EL (f) EL; N/ EL (f) EL; N/ EL (f) EL; N/ EL (f) EL; N/ EL (f) Manufacture of medicinal products PPC 1.2. 859,179 99.7% N/EL N/EL N/EL EL N/EL N/EL 99.9% OpEx of Taxonomy-eligible but not environmentally sustainable activities (not Taxonomy-aligned activities) (A.2) 859,179 99.7% –% –% –% 99.7% –% –% 99.9% A. OpEx of Taxonomy eligible activities (A.1 + A.2) 859,179 99.7% –% –% –% 99.7% –% –% 99.9% B. TAXONOMY-NON-ELIGIBLE ACTIVITIES OpEx of Taxonomy-non-eligible activities 2,261 0.3% Total 861,440 100% argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Environment 300
7.3 Social 7.3.1 Own Workforce 7.3.1.1. Material Impacts, Risks and Opportunities Innovation is at the core of our business, and investing in our people is essential to sustaining it. Our business model depends on cutting-edge research and the continuous development of scientific and technical expertise to advance innovation in immunology. Our success relies on a healthy and highly skilled workforce including employees, contingent workers, independent consultants, and partners who collectively drive our progress. We strive to maintain an inclusive, feedback-driven culture and maintain clear expectations for safety, conduct, and well-being to strengthen our ability to navigate workforce-related risks that could affect continuity, compliance, or innovation capacity. This focus on people helps us to remain equipped with the talent, expertise, and culture necessary to advance immunology innovation and deliver on our long-term strategic objectives. IRO Type Value Chain Time Horizon Equal treatment and opportunities for all Training and skills development Unequal opportunities for training, skill development, and internal advancement across regions or roles, particularly in fast-paced or resource-constrained teams, may limit employee growth and satisfaction. Potential negative impact • Own operations • Medium- term Diversity A lack of diversity, inclusion, and equity across the global workforce, especially within global and specialized Research and Development teams, may undermine employee well-being. Potential negative impact • Own operations • Short-term Working conditions Health and safety Exposure to hazardous substances and biological agents in research and operational roles within the Company's own operations may compromise the physical and psychological well-being of employees. Potential negative impact • Own operations • Short-term 7.3.1.2. Processes for Engaging With Own Workforce and Workers’ Representatives About Impacts All employees are co-owners who contribute to our broader purpose and success. We engage directly with employees and encourage open dialogue through regular forums such as quarterly Company-wide meetings, focus groups, and new-hire check-ins. • Employees can contribute to the agenda of quarterly Company-wide meetings by submitting questions in advance and can also raise additional questions live during the meeting. • We have established a network of ‘Culture Champions’—nominated employees—who host ongoing cultural dialogue sessions. Insights from these sessions inform employee programs and help guide leadership in advancing employee engagement opportunities. • Office and campus site teams host quarterly focus groups on topics including well-being, development, health and safety, and other employee initiatives. • New-hire check-ins are hosted by members of the HR team after the first three months. • CEO welcome sessions are held to introduce new hires to argenx. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Social 301 S1 SBM-3 S1-2

We also engage employees through various ongoing communication channels. All employees are invited to share ideas and questions through ‘Engage’, our internal social networking platform. The Employee Communications team provides weekly Company-wide updates and a monthly ‘In Case You Missed It’ recap. Employees also receive an internal notification whenever a company press release is issued. We assess the effectiveness of employee engagement, with oversight from the Global Head of Human Resources, by gathering feedback through focus group discussions, employee questions and comments submitted before the quarterly Company-wide meetings, and analysis of engagement with internal communication channels. To strengthen communication and collaboration, employees are trained on the situation-behavior-impact feedback model, which promotes clear, actionable, and constructive feedback. We do not currently have a separate process to gain insights into the perspectives of particularly vulnerable employee groups. 7.3.1.3. Processes to Remediate Negative Impacts and Channels for Own Workforce to Raise Concerns We provide multiple channels for employees to raise questions, concerns, or file reports. For health and safety related matters, employees can contact their respective HR Business Partner or contact the Global Facilities and Employee Health and Safety Lead directly if involved in an incident. For other matters, communication channels include the argenx Helpline, HR and Legal teams. These channels are introduced to all employees during onboarding. Our Anti-Retaliation Policy, detailed in Section 7.4.1.3. “Protection of Whistleblowers”, strictly prohibits retaliation against anyone who raises a concern in good faith. For more information on our anti-retaliation procedures and how we enable employees to speak up, see the Governance section of this Sustainability Statement. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Social 302 S1-3
7.3.1.4. Equal Treatment and Opportunities for All Policies We uphold the right to freedom of association and maintain a zero-tolerance stance on workplace discrimination. Our Code of Conduct and Business Ethics, as detailed in Section 7.4.1.2. “Corporate Culture”, outlines our policies for prohibiting and preventing discrimination based on categories, including but not limited to race, religion, color, political convictions, gender, sex, pregnancy, ethnicity or national origin, civil state, social status, sexual orientation, disability (or handicap), or age. Our Anti-Retaliation Policy prohibits discrimination against employees who raise complaints. Our approach to equal treatment and opportunity is further supported by the following policies: Policy Managing Training standard operating procedure Purpose Establish a uniform approach to training, including how training is managed and assigned. Promote equal treatment and professional conduct through uniform training. Scope All employees, consultants, interns, and contractors Most senior level accountable Head of Quality Availability Internal document-sharing platform Process for monitoring Quarterly Quality Management Review Applicability across sustainability statement Section 7.3.1 “Own Workforce (S1)” Policy Diversity, Equity and Inclusion Policy Purpose Support a workforce that is composed of members that can provide broad and complementary perspectives of the various business goals and strategic objectives of our company. Comply with the requirement we are subject to pursuant to the Dutch Civil Code and the Dutch Decree on the Content of the Management Report to disclose our diversity policy with respect to our directors and employees in managerial positions to the Dutch Social and Economic Council (Sociaal Economische Raad). Scope All global operations Most senior level accountable Head of HR and General Counsel and Corporate Secretary Availability Externally available: Diversity Equity and Inclusion Policy Process for monitoring No formal monitoring Applicability across sustainability statement Section 7.3.1 “Own Workforce (S1)” We do not have a separate policy or program that manages impacts related to human rights. We engage with third parties who align with our values and endeavor to not conduct business with any individual or organization that participates in activities we prohibit. We comply with international labor standards and applicable employment laws in our regions of operation. This includes, but is not limited to, the prohibition of child exploitation and child labor, forced, bonded, or indentured labor and involuntary prison labor, harsh or inhumane treatment, the threat thereof, or any form of modern slavery or human trafficking. We review our initiatives to facilitate compliance with local laws, and individual employment decisions are based on merit, consistent with our philosophy and applicable laws. We have not conducted a direct assessment of whether any of our operations are at heightened risk of forced, compulsory, or child labor. However, our owned operations are located in geographies and involve activities that are not typically associated with elevated risks of these issues. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Social 303 S1-1 MDR-P
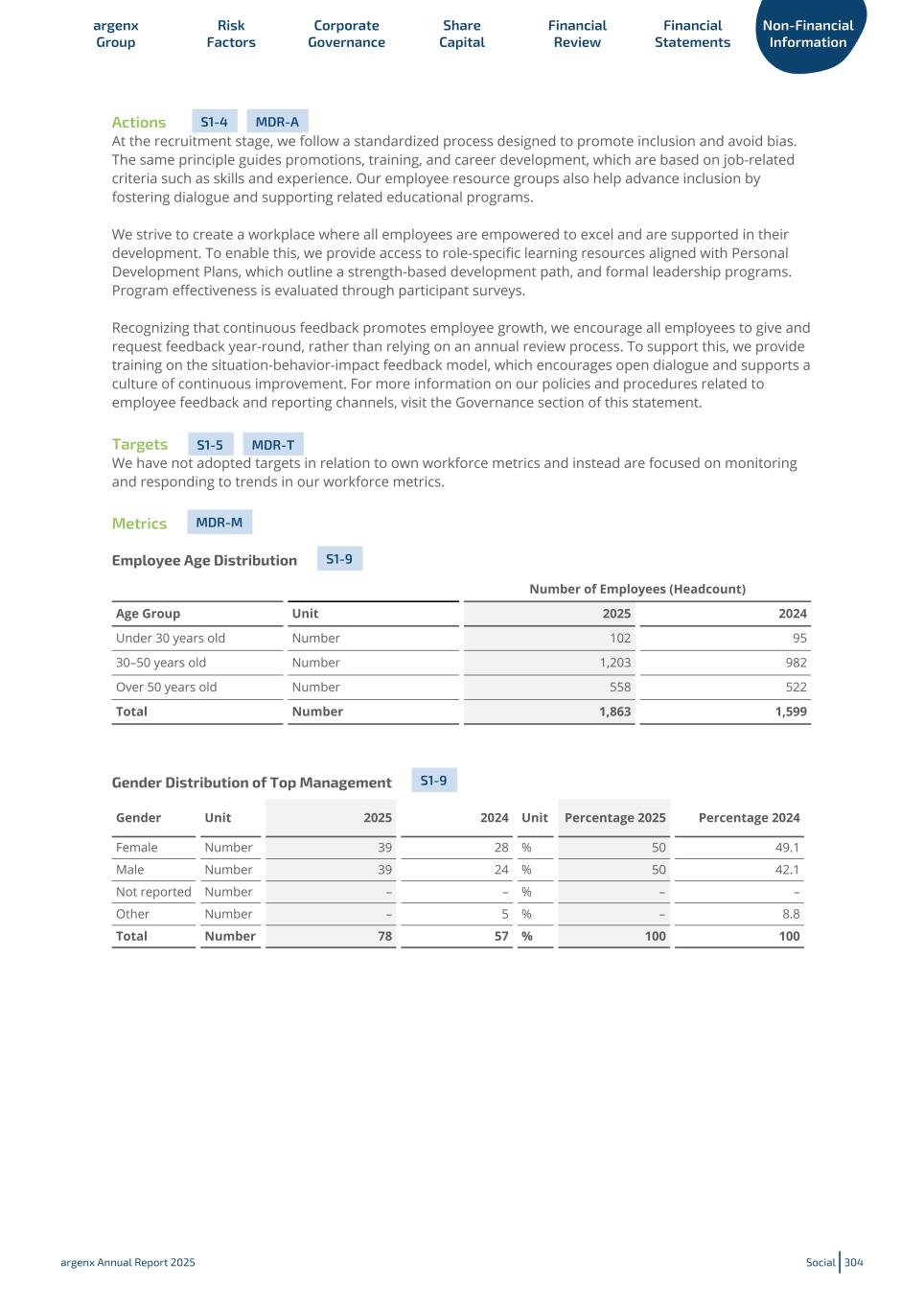
Actions At the recruitment stage, we follow a standardized process designed to promote inclusion and avoid bias. The same principle guides promotions, training, and career development, which are based on job-related criteria such as skills and experience. Our employee resource groups also help advance inclusion by fostering dialogue and supporting related educational programs. We strive to create a workplace where all employees are empowered to excel and are supported in their development. To enable this, we provide access to role-specific learning resources aligned with Personal Development Plans, which outline a strength-based development path, and formal leadership programs. Program effectiveness is evaluated through participant surveys. Recognizing that continuous feedback promotes employee growth, we encourage all employees to give and request feedback year-round, rather than relying on an annual review process. To support this, we provide training on the situation-behavior-impact feedback model, which encourages open dialogue and supports a culture of continuous improvement. For more information on our policies and procedures related to employee feedback and reporting channels, visit the Governance section of this statement. Targets We have not adopted targets in relation to own workforce metrics and instead are focused on monitoring and responding to trends in our workforce metrics. Metrics Employee Age Distribution Number of Employees (Headcount) Age Group Unit 2025 2024 Under 30 years old Number 102 95 30–50 years old Number 1,203 982 Over 50 years old Number 558 522 Total Number 1,863 1,599 Gender Distribution of Top Management Gender Unit 2025 2024 Unit Percentage 2025 Percentage 2024 Female Number 39 28 % 50 49.1 Male Number 39 24 % 50 42.1 Not reported Number – – % – – Other Number – 5 % – 8.8 Total Number 78 57 % 100 100 argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Social 304 MDR-M S1-9 S1-9 S1-4 MDR-A S1-5 MDR-T

Training and Skills Development Metrics We have applied transitional relief under ESRS S1-13 for the second year Sustainability Statement preparation Remuneration (Pay Gap and Total Remuneration) The remuneration ratio below is defined under ESRS and is presented differently under the Remuneration and Compensation Statement in Section 3.4 of this Annual Report. The figure for 2025 is 25. In 2024, this figure was 23. Our commitment to pay equity is deeply rooted in our core values and cultural foundation. We ensure that our remuneration practices are fair, reflecting team and individual impact. They are also based on skills and market competitiveness relevant to the responsibilities held. The gender pay gap as presented in the below table reflects the adjusted gender pay gap for comparable positions, responsibilities, skill sets and experiences following the ESRS methodology but clustered in the following five categories: • Individual contributor; • Managers; • Directors; • Vice-Presidents; • Executives (excluding the CEO). The metric is calculated as the average male gross hourly pay level less the average female gross hourly pay level expressed as a percentage of the average male gross hourly pay level (Average gross hourly pay for male employees - Average gross hourly pay for female employees) / (Average gross hourly pay for male employees) * 100. Using this methodology the gender pay gaps range from -7% to +2.5% with a weighted average of 4.6% in favor of women, reflecting our commitment and continuous monitoring of the core principles as laid out above. Level Gender Pay Gap - 2025 Gender Pay Gap - 2024 Individual Contributors (3.9%) (5.1%) Managers (7.1%) (6.0%) Directors (3.8%) (5.6%) Vice-Presidents 2.5% 1.0% Executives excluding CEO (5.4%) (2.7%) Weighted average gender pay gap (4.6%) (5.4%) argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Social 305 S1-16 S1-13

We believe that examining gender pay gaps on a purely total population basis without adequate detail and precision, as required by ESRS, does not offer a meaningful metric or insight into the fairness of our employee compensation. It disregards experience, seniority, and cost-of-living differences by country. We believe that the unadjusted gender pay gap ratio provides an inaccurate and overly simplistic representation of a complex measure. If all relevant factors are disregarded, the value for 2025 would be 18%, as calculated under ESRS. This figure for 2024 was 18%. Accounting Policies Top management refers to the leadership team, including the Executive Management Team, major global and commercial leaders, major development project leaders, and key R&D leaders. “Other” within gender distribution of top management refers to vacant top-management positions. 7.3.1.5. Own Workforce Health and Safety Policies While we do not maintain a formal, standalone workplace accident prevention policy, our Code of Business Conduct and Ethics includes a commitment to maintaining a safe and healthy workplace. We comply with applicable health and safety regulations and have implemented processes to prevent work-related incidents. Actions We provide laboratory employees with training on safe chemical handling, waste management, and biosafety practices. As a preventative measure, we also conduct lab audits to raise awareness, verify that policies are understood and applied in daily work, and identify opportunities for improvement. Targets We have not adopted targets in relation to workforce health and safety and instead maintain an objective to minimize incidents across our operations. Metrics Work-Related Fatalities and Injuries Metric Description Unit 2025 2024 Number of fatalities in own workforce as result of work-related injuries and work-related ill health Number – – Number of recordable work-related accidents for own workforce Number – 1 Percentage of own workforce who are covered by health and safety management system based on recognized standards or guidelines and which have been internally audited and/or audited or certified by an external party % – – Rate of recordable work-related accidents for own workforce Number – 0.000018 Accounting Policies Work-related fatalities and injuries reported in headcount reflect employees and do not include contractors or others from third-party companies. Percentage of own workforce who are covered by health and safety management system based on recognized standards or guidelines and which have been internally audited and/or audited or certified by an external party only reflects coverage by recognized standards and guidelines, this metric does not relate to legal requirements. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Social 306 S1-1 MDR-P S1-4 MDR-A MDR-M S1-14 S1-5 MDR-T
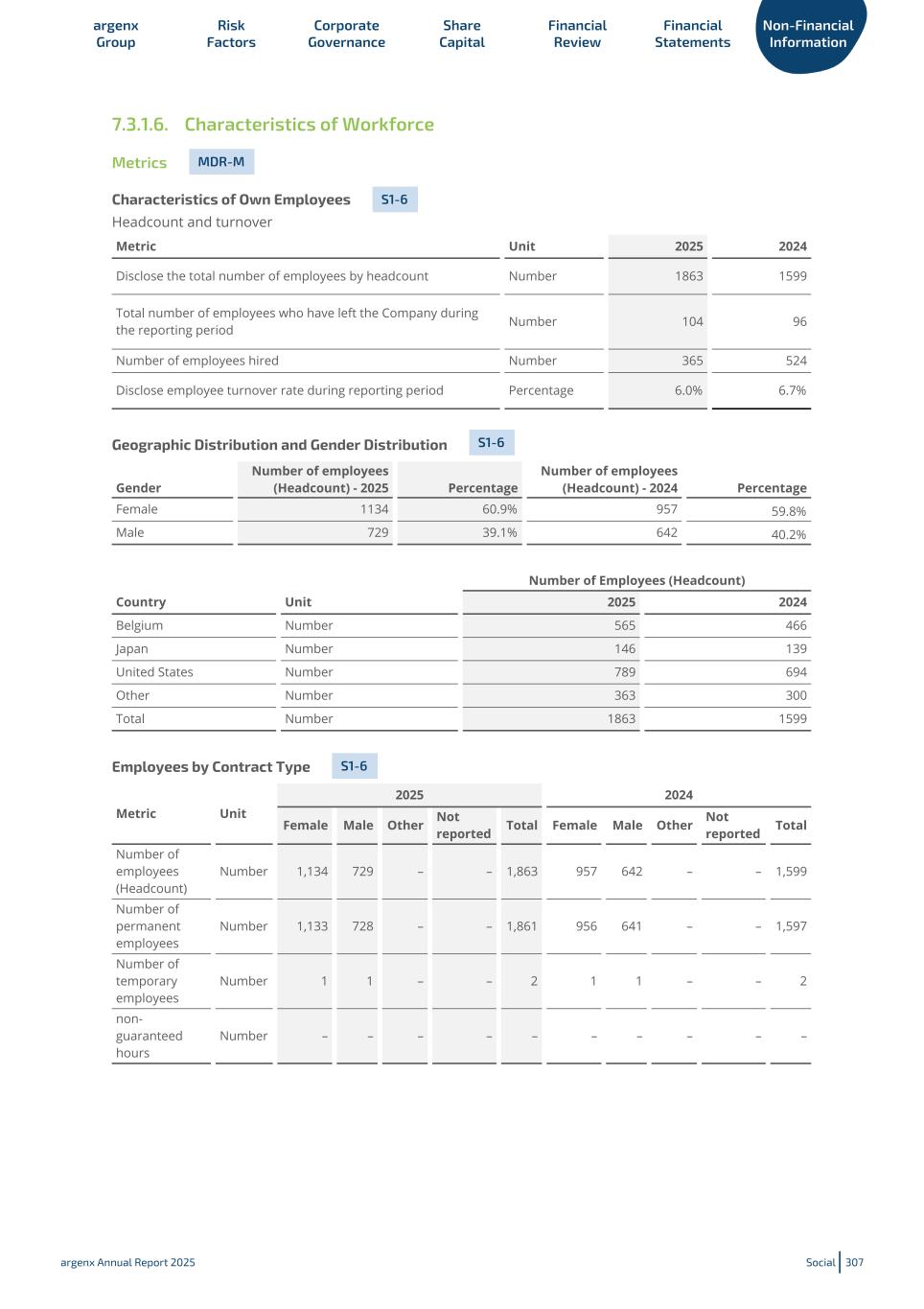
7.3.1.6. Characteristics of Workforce Metrics Characteristics of Own Employees Headcount and turnover Metric Unit 2025 2024 Disclose the total number of employees by headcount Number 1863 1599 Total number of employees who have left the Company during the reporting period Number 104 96 Number of employees hired Number 365 524 Disclose employee turnover rate during reporting period Percentage 6.0% 6.7% Geographic Distribution and Gender Distribution Gender Number of employees (Headcount) - 2025 Percentage Number of employees (Headcount) - 2024 Percentage Female 1134 60.9% 957 59.8% Male 729 39.1% 642 40.2% Number of Employees (Headcount) Country Unit 2025 2024 Belgium Number 565 466 Japan Number 146 139 United States Number 789 694 Other Number 363 300 Total Number 1863 1599 Employees by Contract Type Metric Unit 2025 2024 Female Male Other Not reported Total Female Male Other Not reported Total Number of employees (Headcount) Number 1,134 729 – – 1,863 957 642 – – 1,599 Number of permanent employees Number 1,133 728 – – 1,861 956 641 – – 1,597 Number of temporary employees Number 1 1 – – 2 1 1 – – 2 Number of non- guaranteed hours Number – – – – – – – – – – argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Social 307 MDR-M S1-6 S1-6 S1-6

Accounting Policies Headcount represents the total number of employees as of December 31st, 2025. Turnover is calculated using average headcount at year-end as the denominator. Data is compiled from our internal HR system (Workday). For additional employee data, see “Note 19 Personnel Expenses”. The geographic breakdown includes countries with more than 50 employees, that represent at least 10% of our total workforce. Entities with fewer than 50 employees are consolidated and reported under "Other.” For 2025, “Other” includes Australia, Austria, Brazil, Canada, France, Germany, Greece, Ireland, Italy, Netherlands, Poland, Portugal, Spain, Sweden, Switzerland, United Kingdom. Within contract type by gender, “Not reported” indicates employees who chose not to disclose this information. Characteristics of Non-Employees in the Workforce We have applied transitional relief under ESRS S1-7 for the second year Sustainability Statement preparation. Discrimination Incidents Reported and Complaints Filed Metric 2025 2024 Total number of incidents of discrimination including harassment reported in the reporting period 131) 11 Number of complaints filed through channels for workforce 61) – Total amount of fines, penalties, and compensation for damages due to incidents of discrimination, including harassment and complaints filed – – Total amount of fines, penalties and compensation for damages due to cases of severe human rights incidents – – Number of severe human rights incidents including an indication of how many are cases of non-respect of the UN Guiding Principles on Business and Human Rights, ILO Declaration on Fundamental Principles and Rights at Work, or OECD Guidelines for Multinational Enterprises - 2025 – – 1) None of the matters were substantiated as discrimination, harassment, a severe human rights incident, or any other instance of unlawful conduct or activity. The Company may become aware of workforce concerns from time to time, both internally and externally. We take these matters seriously. In line with our commitment to maintaining a safe workplace, respecting human rights, and providing equal treatment and equal employment opportunities,we take prompt and appropriate action when such concerns are raised. Accounting Policies We track workforce concerns, including equal employment opportunity matters and complaints or incidents related to discrimination and harassment. In 2025, 19 such matters were reported, including 6 complaints and 13 incidents of discrimination and/or harassment. None of the 19 matters were substantiated as discrimination, harassment, a severe human rights incident, or any other instance of unlawful conduct or activity. In 2024, 11 matters were reported, all relating to discrimination or harassment, and none were substantiated. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Social 308 S1-7 S1-17
7.3.2 Patients In accordance with the European Commission’s “Quick-Fix” amendment, we have applied the phase-in relief for ESRS S4 Patients. Given the central importance of patients to our business model, we have included the most salient and decision-useful information in this S4 section to ensure that reporting remains focused on the most relevant impacts and stakeholder information needs. This section provides a summary of the material impacts, risks, and opportunities identified, together with an overview of related policies, actions, targets, and entity-specific metrics, in line with the disclosure requirements set out in ESRS 2 BP-2 (17). 7.3.2.1. Material Impacts, Risks and Opportunities IRO Type Value Chain Time Horizon Information- related impacts for patients Privacy Inadvertent exposure of sensitive patient information may increase the risk of fines and penalties, lawsuits, remediation costs and reputational damage to the Company, and may undermine patient trust in healthcare innovation. Risk • Upstream • Own operations • Downstream • Short-term Access to quality information Failure to ensure transparent, accurate, and compliant product information (including labelling, usage instructions, and risk disclosures) may compromise patient safety, damage trust with healthcare professionals and regulators, and lead to financial and regulatory penalties. Risk • Own operations • Downstream • Short-term Access to quality information Misleading or incomplete product information (e.g., improper dosing or contraindication risks) can lead to patient harm, particularly in complex autoimmune treatments. Potential negative impact • Downstream • Medium- term Personal safety of patients Health and safety Failing to ensure the safety of clinical trial participants, whether due to investigational product issues or inadequate oversight of contract research partners, can lead to direct harm to patient health and well-being. Potential negative impact • Downstream • Short-term Health and safety Variability in product quality across a globally distributed manufacturing network could compromise treatment efficacy and patient safety. Potential negative impact • Upstream • Own operations • Downstream • Medium- term Health and safety Insufficient product traceability and transparency may enable counterfeit or compromised drugs to enter the supply chain, posing serious risks to patient safety. Potential negative impact • Upstream • Downstream • Medium- term Health and safety Risk of clinical trial suspension, reputational damage and future regulatory hurdles as a result of adverse side-effects observed during clinical trials, including unforeseen reactions, if not appropriately addressed. Risk • Own operations • Downstream • Medium- term argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Social 309 S4 SBM-3
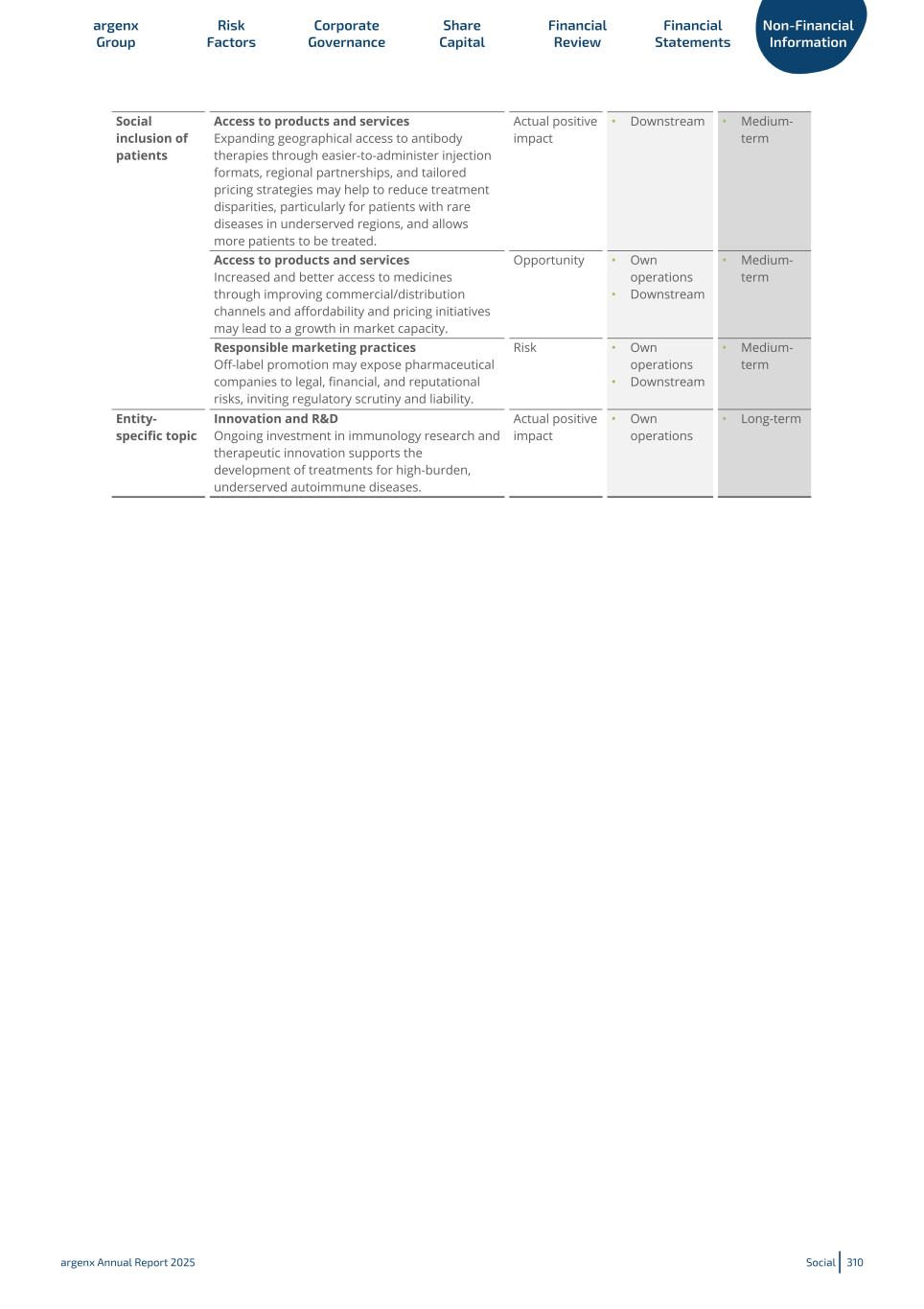
Social inclusion of patients Access to products and services Expanding geographical access to antibody therapies through easier-to-administer injection formats, regional partnerships, and tailored pricing strategies may help to reduce treatment disparities, particularly for patients with rare diseases in underserved regions, and allows more patients to be treated. Actual positive impact • Downstream • Medium- term Access to products and services Increased and better access to medicines through improving commercial/distribution channels and affordability and pricing initiatives may lead to a growth in market capacity. Opportunity • Own operations • Downstream • Medium- term Responsible marketing practices Off-label promotion may expose pharmaceutical companies to legal, financial, and reputational risks, inviting regulatory scrutiny and liability. Risk • Own operations • Downstream • Medium- term Entity- specific topic Innovation and R&D Ongoing investment in immunology research and therapeutic innovation supports the development of treatments for high-burden, underserved autoimmune diseases. Actual positive impact • Own operations • Long-term argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Social 310
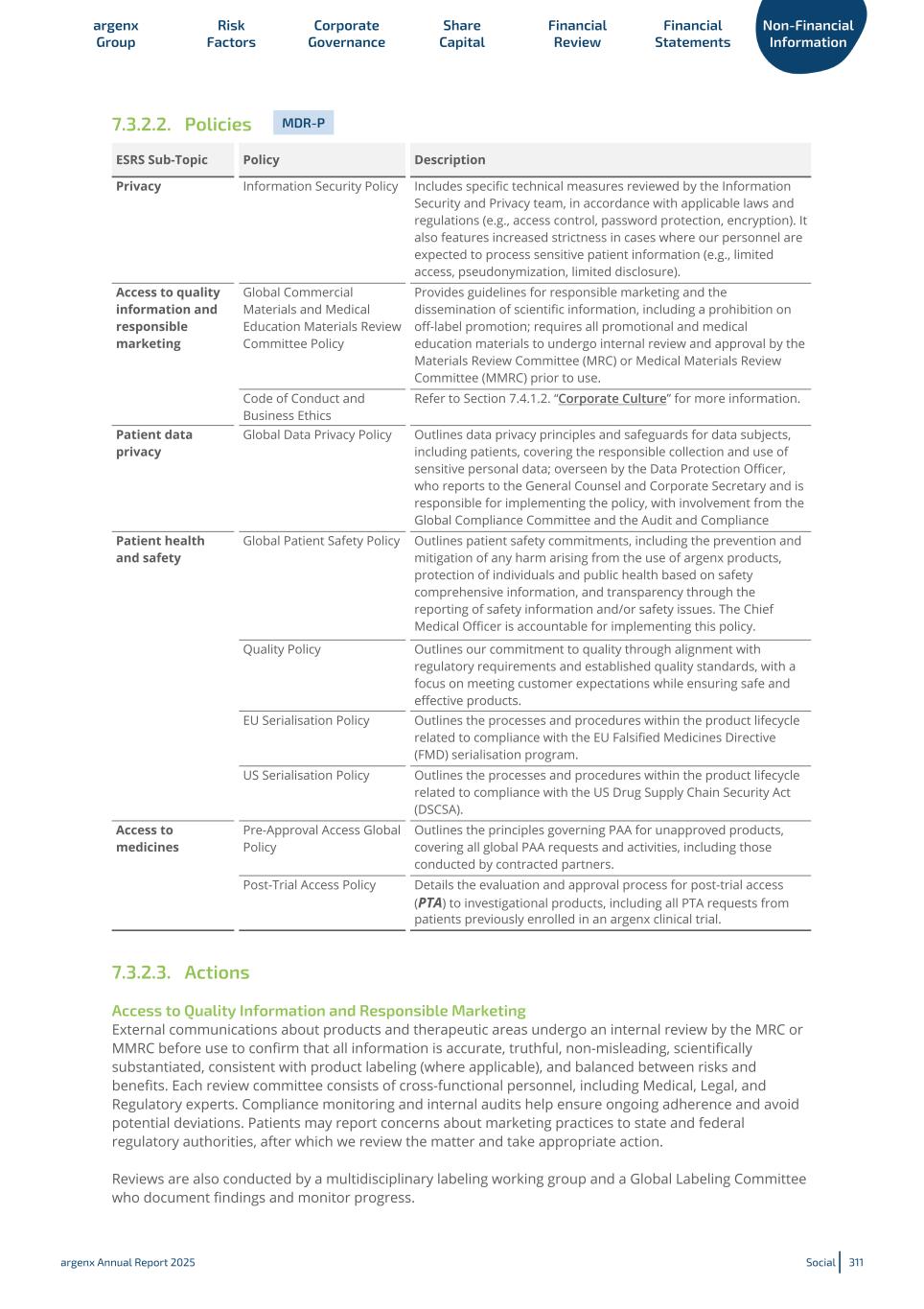
7.3.2.2. Policies ESRS Sub-Topic Policy Description Privacy Information Security Policy Includes specific technical measures reviewed by the Information Security and Privacy team, in accordance with applicable laws and regulations (e.g., access control, password protection, encryption). It also features increased strictness in cases where our personnel are expected to process sensitive patient information (e.g., limited access, pseudonymization, limited disclosure). Access to quality information and responsible marketing Global Commercial Materials and Medical Education Materials Review Committee Policy Provides guidelines for responsible marketing and the dissemination of scientific information, including a prohibition on off-label promotion; requires all promotional and medical education materials to undergo internal review and approval by the Materials Review Committee (MRC) or Medical Materials Review Committee (MMRC) prior to use. Code of Conduct and Business Ethics Refer to Section 7.4.1.2. “Corporate Culture” for more information. Patient data privacy Global Data Privacy Policy Outlines data privacy principles and safeguards for data subjects, including patients, covering the responsible collection and use of sensitive personal data; overseen by the Data Protection Officer, who reports to the General Counsel and Corporate Secretary and is responsible for implementing the policy, with involvement from the Global Compliance Committee and the Audit and Compliance Patient health and safety Global Patient Safety Policy Outlines patient safety commitments, including the prevention and mitigation of any harm arising from the use of argenx products, protection of individuals and public health based on safety comprehensive information, and transparency through the reporting of safety information and/or safety issues. The Chief Medical Officer is accountable for implementing this policy. Quality Policy Outlines our commitment to quality through alignment with regulatory requirements and established quality standards, with a focus on meeting customer expectations while ensuring safe and effective products. EU Serialisation Policy Outlines the processes and procedures within the product lifecycle related to compliance with the EU Falsified Medicines Directive (FMD) serialisation program. US Serialisation Policy Outlines the processes and procedures within the product lifecycle related to compliance with the US Drug Supply Chain Security Act (DSCSA). Access to medicines Pre-Approval Access Global Policy Outlines the principles governing PAA for unapproved products, covering all global PAA requests and activities, including those conducted by contracted partners. Post-Trial Access Policy Details the evaluation and approval process for post-trial access (PTA) to investigational products, including all PTA requests from patients previously enrolled in an argenx clinical trial. 7.3.2.3. Actions Access to Quality Information and Responsible Marketing External communications about products and therapeutic areas undergo an internal review by the MRC or MMRC before use to confirm that all information is accurate, truthful, non-misleading, scientifically substantiated, consistent with product labeling (where applicable), and balanced between risks and benefits. Each review committee consists of cross-functional personnel, including Medical, Legal, and Regulatory experts. Compliance monitoring and internal audits help ensure ongoing adherence and avoid potential deviations. Patients may report concerns about marketing practices to state and federal regulatory authorities, after which we review the matter and take appropriate action. Reviews are also conducted by a multidisciplinary labeling working group and a Global Labeling Committee who document findings and monitor progress. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Social 311 MDR-P
We adhere to the European Federation of Pharmaceutical Industries and Associations Code of Conduct, which defines ethical interactions with healthcare professionals, healthcare organizations, and patient organizations, supporting responsible promotion of medicinal products. Patient Health and Safety We have procedures in place to safeguard patient health and to align with global and local pharmacovigilance regulations and standards. This includes signal detection—identifying, evaluating, and acting on potential associations between medicinal products and adverse events, and determining recommendations. To mitigate the risk of falsified or counterfeit medicines, we follow a standardized reporting process. In suspected cases, affected batches are isolated, quarantined, and investigated. If counterfeit products are confirmed, relevant supply-chain stakeholders and authorities are notified, and market actions are taken in consultation with authorities. In line with the Global Patient Safety Policy, we communicate significant safety findings that affect the benefit-risk balance of our products to all applicable parties, including health authorities, patients, and healthcare providers. To protect clinical trial participants, we train clinical investigators and site staff on study protocols and trial requirements and regularly monitor trial sites for compliance. We monitor pharmacovigilance compliance through defined metrics and implement corrective and preventive actions when thresholds are not met. Each instance is recorded in our internal system, and a corrective and preventative action is created which covers root-cause analysis, immediate corrective measures, preventive actions, and defined timelines. Patients can report adverse events or other concerns through multiple channels, including their physician or nurse; MyVyvgartPath Nurse Case Managers (for enrolled, post-prescription U.S. patients); argenx patient-advocacy representatives; or via our website, email, phone, or social media. Adverse events or quality issues at any stage of the product lifecycle—including clinical trials, pre-approval access, and post-marketing—are reported through various channels to Global Patient Safety and Global Quality. These reports are managed in accordance with pharmacovigilance regulations, guidelines, and internal quality procedures. We assess our pharmacovigilance system through compliance monitoring and quality-management audits, evaluating product safety profiles and benefit-risk balance using all available data. The internal Benefit-Risk Committee reviews findings quarterly or as needed for each approved and investigational product. Access to Medicines MyVyvgartPath provides patients with education on insurance coverage, financial-assistance programs, commercial co-pay support, and expected out-of-pocket costs to help them navigate the complexities of the healthcare system in the United States. We collaborate with distribution partners, healthcare systems, and payers to optimize supply chains and reduce access barriers, including formulary restrictions, unfavorable medical policies, new-to-market blocks, prior-authorization requirements, and step edits. Market-specific affordability programs help us reach more patients and advance rare-disease care. In the United States, we participate in Medicare and Medicaid programs, which cap patient and manufacturer out- of-pocket contributions, and in the 340B program, which provides upfront product discounts to covered providers serving low-income communities. Licensed physicians may request access to investigational medicines under development at argenx for patients with serious or immediately life-threatening conditions who are unable to participate in clinical trials or who have exhausted available treatment options. Unsolicited requests are reviewed in accordance with the Pre-Approval Access Policy, which governs all global requests. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Social 312

Additionally, patients enrolled in argenx clinical trials may qualify for Post-Trial Access during temporary treatment gaps before medicines are approved or reimbursed in their country. The Post-Trial Access Program is managed by Medical Affairs and Evidence Generation under the oversight of the Chief Medical Officer. Please refer to Section 1.7.4 "Coverage, Pricing and Reimbursement” and Section 1.7.5 “Government Pricing and Reimbursement Programs for Marketed Drugs in the U.S.” for additional information. Innovation and R&D Our Vision 2030 outlines our long-term ambition to transform the treatment for patients living with autoimmune diseases. Please refer to Section 1.2.1 “Company’s Strategies” for additional information. 7.3.2.4. Targets Our Vision 2030 aims to treat 50,000 patients with our medicines, achieve ten labeled indications across our approved medicines, and advance five pipeline candidates into Phase 3 development by 2030. Please refer to Section 1.2.1 “Company’s Strategies” for additional information. There are currently no targets in relation to access to quality information and responsible marketing, patient data privacy, patient health and safety, and access to medicines. 7.3.2.5. Metrics MDR-M Legal Proceedings Relating to Ethical Marketing Metric 2025 2024 Total amount of monetary losses as a result of legal proceedings associated with false marketing claims – – Data Privacy Metric 2025 2024 Number of data breaches that were detected within argenx that involved the exposure of sensitive patient information 2 – Number of data breaches that were reportable to authorities or data subjects under applicable law – – argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Social 313
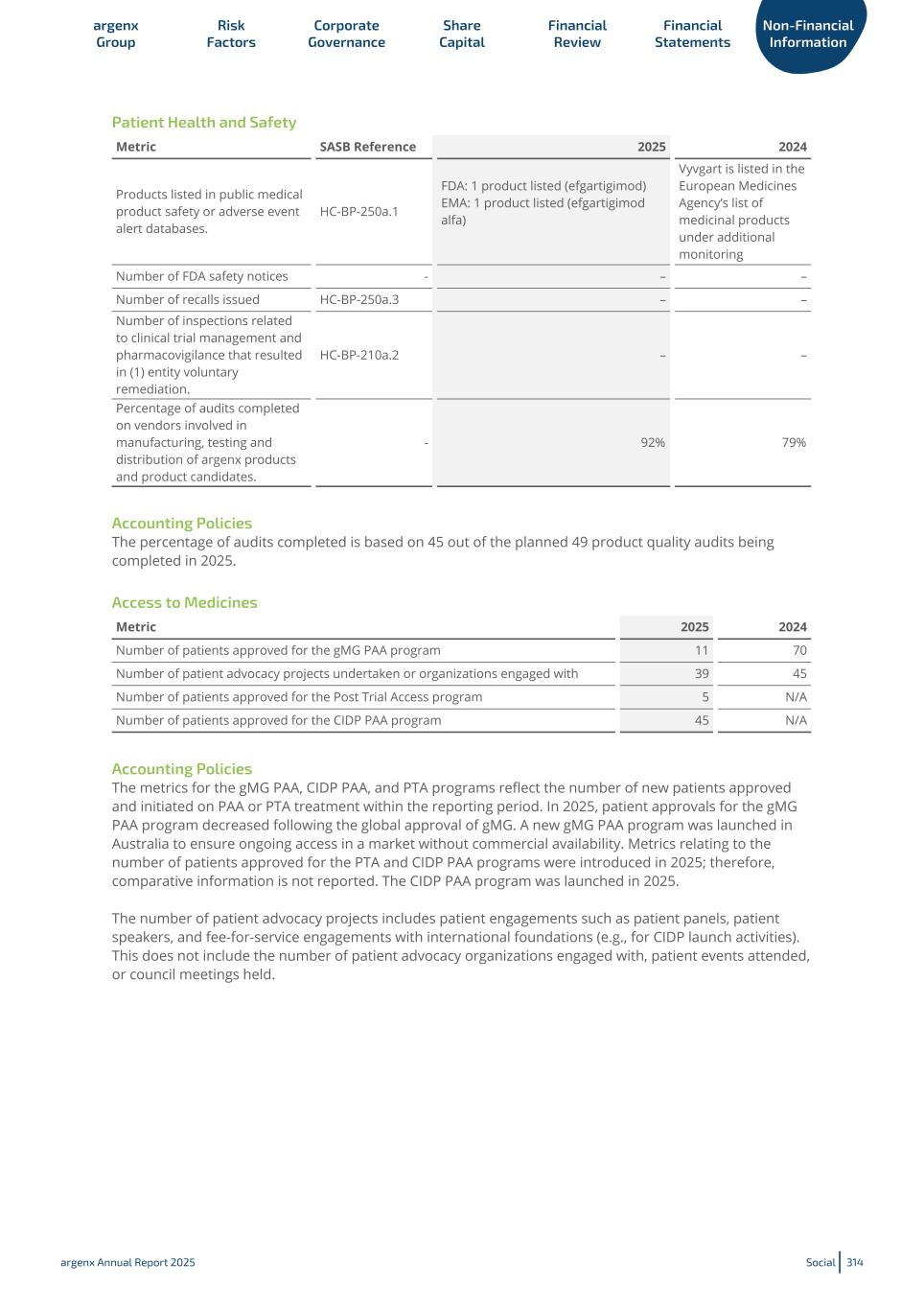
Patient Health and Safety Metric SASB Reference 2025 2024 Products listed in public medical product safety or adverse event alert databases. HC-BP-250a.1 FDA: 1 product listed (efgartigimod) EMA: 1 product listed (efgartigimod alfa) Vyvgart is listed in the European Medicines Agency’s list of medicinal products under additional monitoring Number of FDA safety notices - – – Number of recalls issued HC-BP-250a.3 – – Number of inspections related to clinical trial management and pharmacovigilance that resulted in (1) entity voluntary remediation. HC-BP-210a.2 – – Percentage of audits completed on vendors involved in manufacturing, testing and distribution of argenx products and product candidates. - 92% 79% Accounting Policies The percentage of audits completed is based on 45 out of the planned 49 product quality audits being completed in 2025. Access to Medicines Metric 2025 2024 Number of patients approved for the gMG PAA program 11 70 Number of patient advocacy projects undertaken or organizations engaged with 39 45 Number of patients approved for the Post Trial Access program 5 N/A Number of patients approved for the CIDP PAA program 45 N/A Accounting Policies The metrics for the gMG PAA, CIDP PAA, and PTA programs reflect the number of new patients approved and initiated on PAA or PTA treatment within the reporting period. In 2025, patient approvals for the gMG PAA program decreased following the global approval of gMG. A new gMG PAA program was launched in Australia to ensure ongoing access in a market without commercial availability. Metrics relating to the number of patients approved for the PTA and CIDP PAA programs were introduced in 2025; therefore, comparative information is not reported. The CIDP PAA program was launched in 2025. The number of patient advocacy projects includes patient engagements such as patient panels, patient speakers, and fee-for-service engagements with international foundations (e.g., for CIDP launch activities). This does not include the number of patient advocacy organizations engaged with, patient events attended, or council meetings held. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Social 314
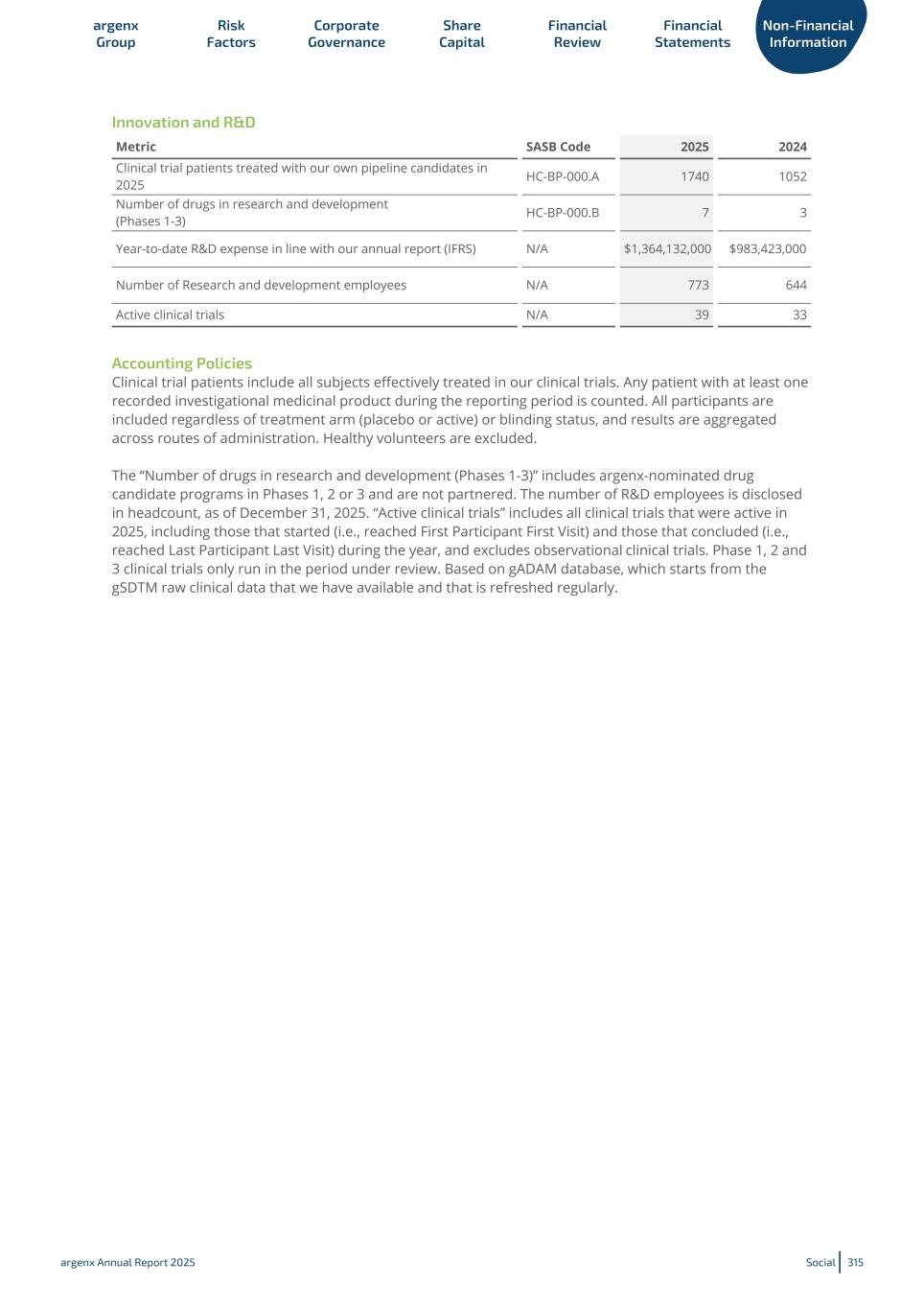
Innovation and R&D Metric SASB Code 2025 2024 Clinical trial patients treated with our own pipeline candidates in 2025 HC-BP-000.A 1740 1052 Number of drugs in research and development (Phases 1-3) HC-BP-000.B 7 3 Year-to-date R&D expense in line with our annual report (IFRS) N/A $ 1,364,132,000 $983,423,000 Number of Research and development employees N/A 773 644 Active clinical trials N/A 39 33 Accounting Policies Clinical trial patients include all subjects effectively treated in our clinical trials. Any patient with at least one recorded investigational medicinal product during the reporting period is counted. All participants are included regardless of treatment arm (placebo or active) or blinding status, and results are aggregated across routes of administration. Healthy volunteers are excluded. The “Number of drugs in research and development (Phases 1-3)” includes argenx-nominated drug candidate programs in Phases 1, 2 or 3 and are not partnered. The number of R&D employees is disclosed in headcount, as of December 31, 2025. “Active clinical trials” includes all clinical trials that were active in 2025, including those that started (i.e., reached First Participant First Visit) and those that concluded (i.e., reached Last Participant Last Visit) during the year, and excludes observational clinical trials. Phase 1, 2 and 3 clinical trials only run in the period under review. Based on gADAM database, which starts from the gSDTM raw clinical data that we have available and that is refreshed regularly. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Social 315
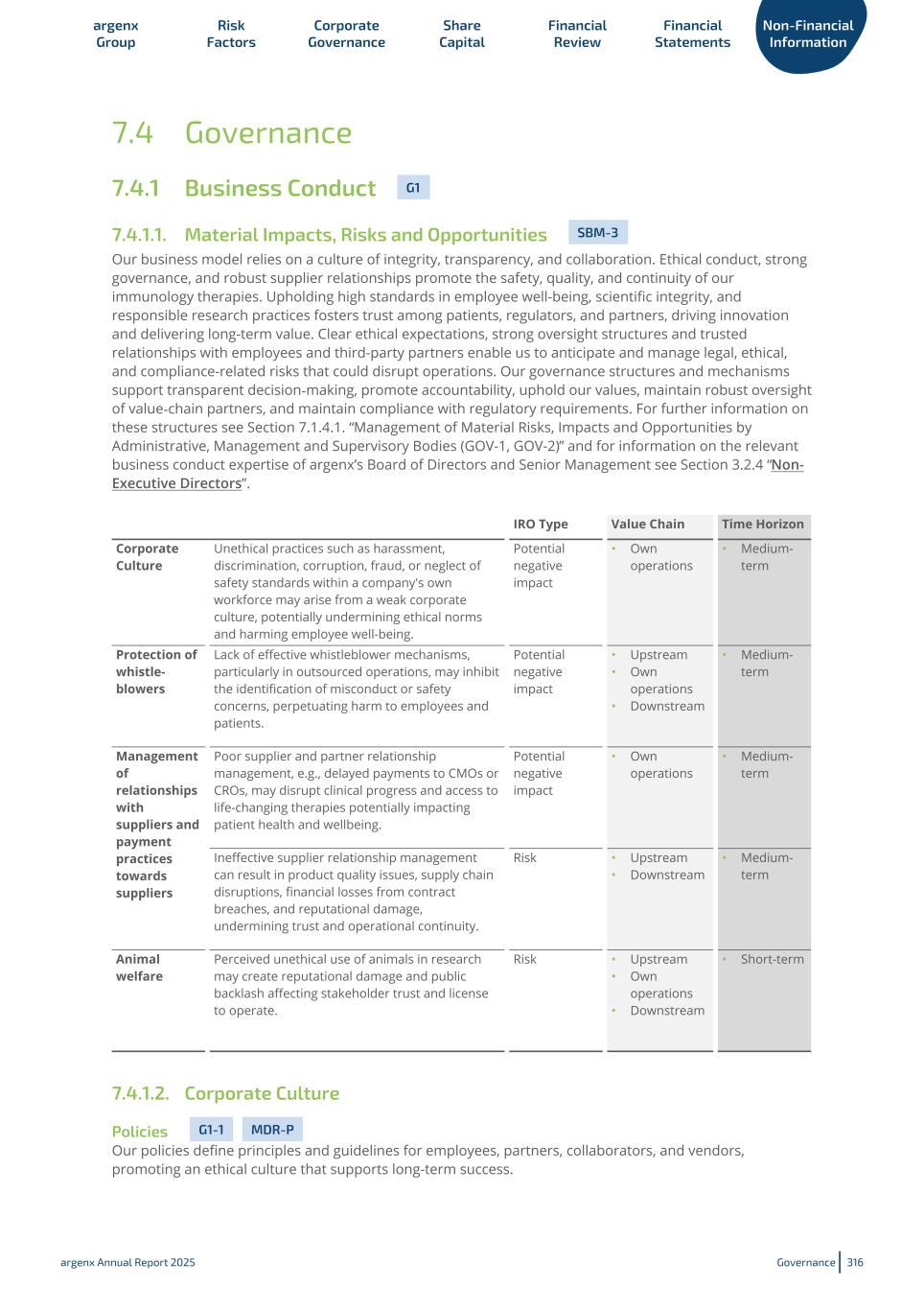
7.4 Governance 7.4.1 Business Conduct 7.4.1.1. Material Impacts, Risks and Opportunities Our business model relies on a culture of integrity, transparency, and collaboration. Ethical conduct, strong governance, and robust supplier relationships promote the safety, quality, and continuity of our immunology therapies. Upholding high standards in employee well-being, scientific integrity, and responsible research practices fosters trust among patients, regulators, and partners, driving innovation and delivering long-term value. Clear ethical expectations, strong oversight structures and trusted relationships with employees and third-party partners enable us to anticipate and manage legal, ethical, and compliance-related risks that could disrupt operations. Our governance structures and mechanisms support transparent decision-making, promote accountability, uphold our values, maintain robust oversight of value-chain partners, and maintain compliance with regulatory requirements. For further information on these structures see Section 7.1.4.1. “Management of Material Risks, Impacts and Opportunities by Administrative, Management and Supervisory Bodies (GOV-1, GOV-2)” and for information on the relevant business conduct expertise of argenx’s Board of Directors and Senior Management see Section 3.2.4 “Non- Executive Directors”. IRO Type Value Chain Time Horizon Corporate Culture Unethical practices such as harassment, discrimination, corruption, fraud, or neglect of safety standards within a company's own workforce may arise from a weak corporate culture, potentially undermining ethical norms and harming employee well-being. Potential negative impact • Own operations • Medium- term Protection of whistle- blowers Lack of effective whistleblower mechanisms, particularly in outsourced operations, may inhibit the identification of misconduct or safety concerns, perpetuating harm to employees and patients. Potential negative impact • Upstream • Own operations • Downstream • Medium- term Management of relationships with suppliers and payment practices towards suppliers Poor supplier and partner relationship management, e.g., delayed payments to CMOs or CROs, may disrupt clinical progress and access to life-changing therapies potentially impacting patient health and wellbeing. Potential negative impact • Own operations • Medium- term Ineffective supplier relationship management can result in product quality issues, supply chain disruptions, financial losses from contract breaches, and reputational damage, undermining trust and operational continuity. Risk • Upstream • Downstream • Medium- term Animal welfare Perceived unethical use of animals in research may create reputational damage and public backlash affecting stakeholder trust and license to operate. Risk • Upstream • Own operations • Downstream • Short-term 7.4.1.2. Corporate Culture Policies Our policies define principles and guidelines for employees, partners, collaborators, and vendors, promoting an ethical culture that supports long-term success. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Governance 316 SBM-3 G1 G1-1 MDR-P
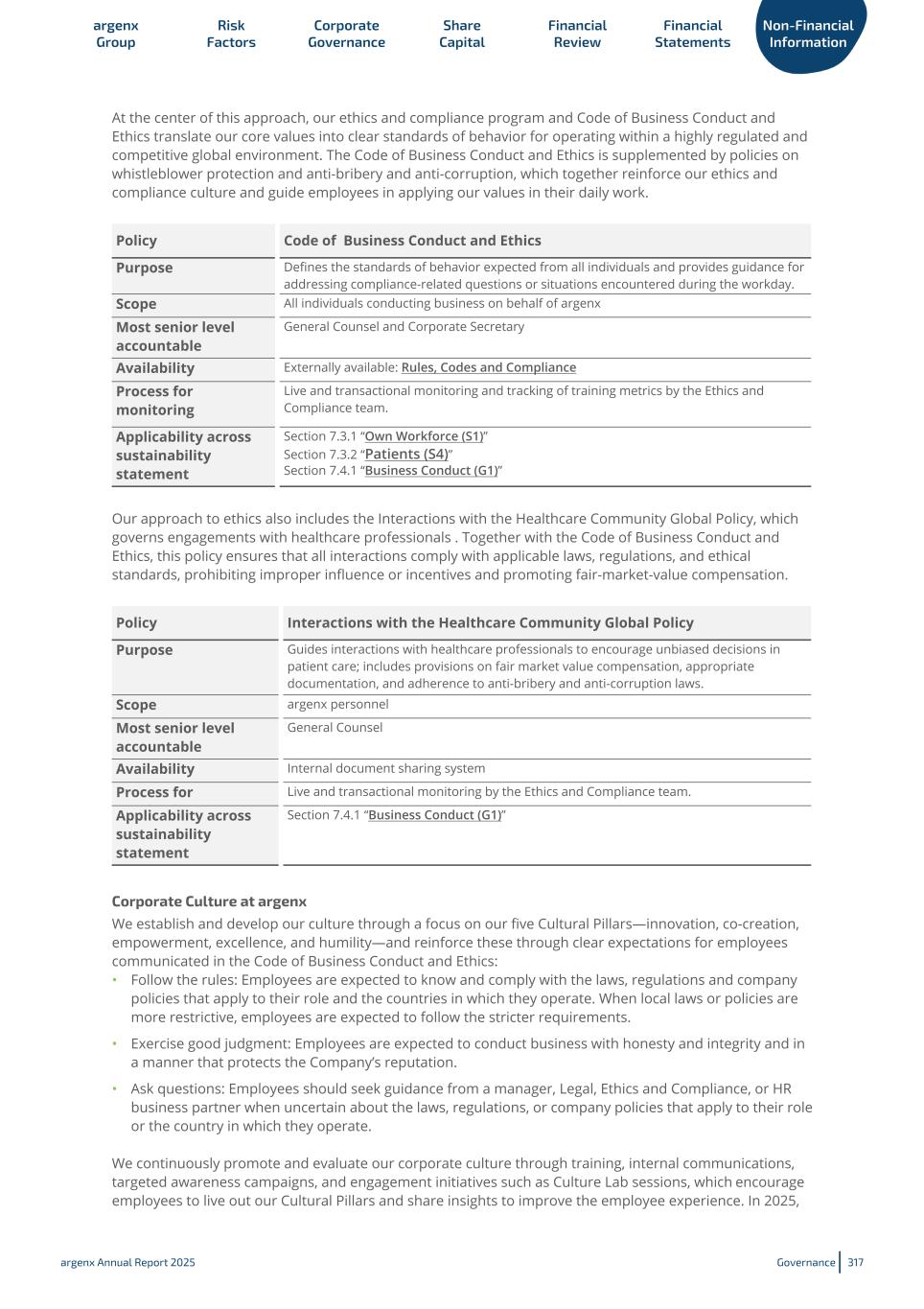
At the center of this approach, our ethics and compliance program and Code of Business Conduct and Ethics translate our core values into clear standards of behavior for operating within a highly regulated and competitive global environment. The Code of Business Conduct and Ethics is supplemented by policies on whistleblower protection and anti-bribery and anti-corruption, which together reinforce our ethics and compliance culture and guide employees in applying our values in their daily work. Policy Code of Business Conduct and Ethics Purpose Defines the standards of behavior expected from all individuals and provides guidance for addressing compliance-related questions or situations encountered during the workday. Scope All individuals conducting business on behalf of argenx Most senior level accountable General Counsel and Corporate Secretary Availability Externally available: Rules, Codes and Compliance Process for monitoring Live and transactional monitoring and tracking of training metrics by the Ethics and Compliance team. Applicability across sustainability statement Section 7.3.1 “Own Workforce (S1)” Section 7.3.2 “Patients (S4)” Section 7.4.1 “Business Conduct (G1)” Our approach to ethics also includes the Interactions with the Healthcare Community Global Policy, which governs engagements with healthcare professionals . Together with the Code of Business Conduct and Ethics, this policy ensures that all interactions comply with applicable laws, regulations, and ethical standards, prohibiting improper influence or incentives and promoting fair-market-value compensation. Policy Interactions with the Healthcare Community Global Policy Purpose Guides interactions with healthcare professionals to encourage unbiased decisions in patient care; includes provisions on fair market value compensation, appropriate documentation, and adherence to anti-bribery and anti-corruption laws. Scope argenx personnel Most senior level accountable General Counsel Availability Internal document sharing system Process for monitoring Live and transactional monitoring by the Ethics and Compliance team. Applicability across sustainability statement Section 7.4.1 “Business Conduct (G1)” Corporate Culture at argenx We establish and develop our culture through a focus on our five Cultural Pillars—innovation, co-creation, empowerment, excellence, and humility—and reinforce these through clear expectations for employees communicated in the Code of Business Conduct and Ethics: • Follow the rules: Employees are expected to know and comply with the laws, regulations and company policies that apply to their role and the countries in which they operate. When local laws or policies are more restrictive, employees are expected to follow the stricter requirements. • Exercise good judgment: Employees are expected to conduct business with honesty and integrity and in a manner that protects the Company’s reputation. • Ask questions: Employees should seek guidance from a manager, Legal, Ethics and Compliance, or HR business partner when uncertain about the laws, regulations, or company policies that apply to their role or the country in which they operate. We continuously promote and evaluate our corporate culture through training, internal communications, targeted awareness campaigns, and engagement initiatives such as Culture Lab sessions, which encourage employees to live out our Cultural Pillars and share insights to improve the employee experience. In 2025, argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Governance 317
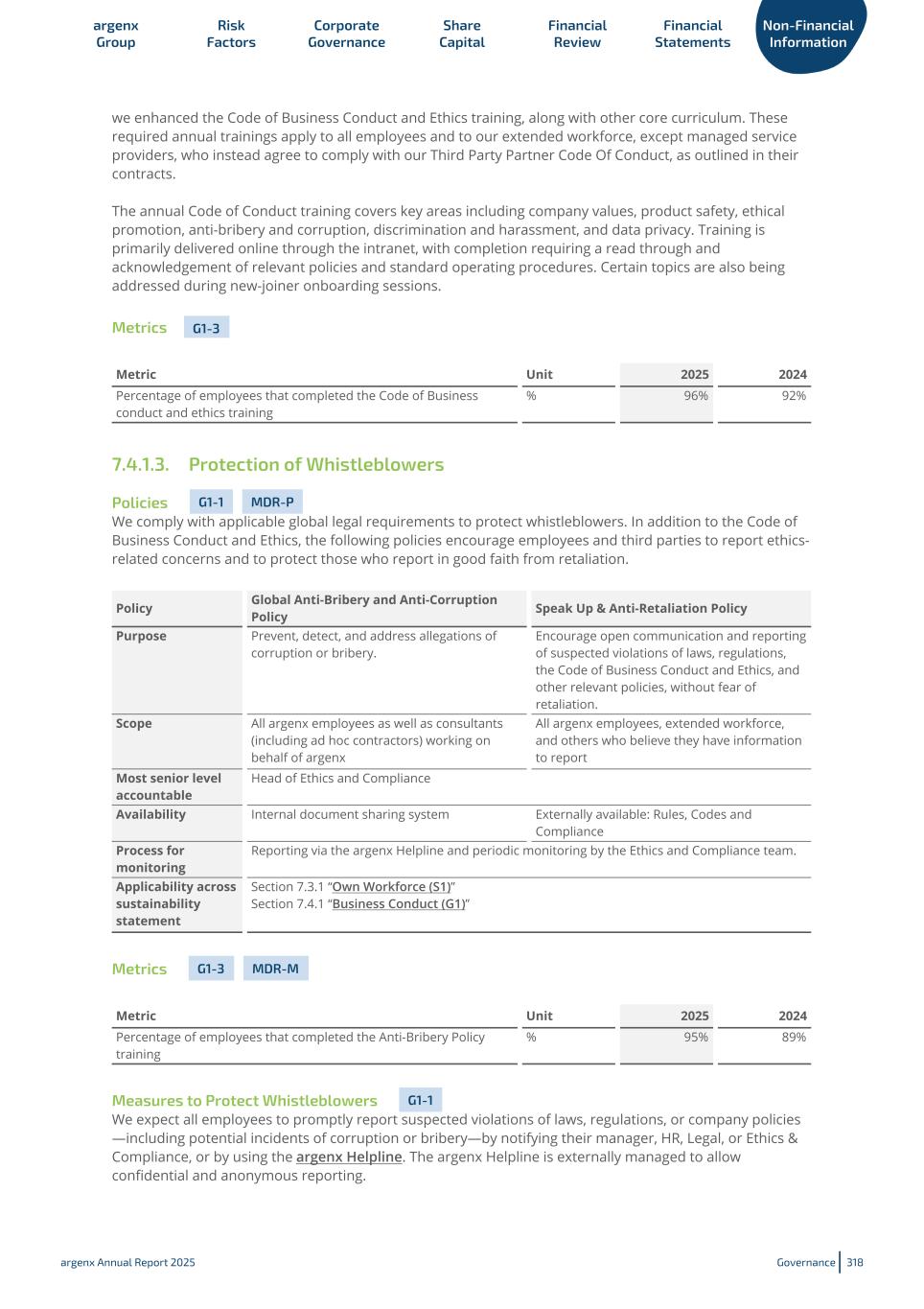
we enhanced the Code of Business Conduct and Ethics training, along with other core curriculum. These required annual trainings apply to all employees and to our extended workforce, except managed service providers, who instead agree to comply with our Third Party Partner Code Of Conduct, as outlined in their contracts. The annual Code of Conduct training covers key areas including company values, product safety, ethical promotion, anti-bribery and corruption, discrimination and harassment, and data privacy. Training is primarily delivered online through the intranet, with completion requiring a read through and acknowledgement of relevant policies and standard operating procedures. Certain topics are also being addressed during new-joiner onboarding sessions. Metrics Metric Unit 2025 2024 Percentage of employees that completed the Code of Business conduct and ethics training % 96% 92% 7.4.1.3. Protection of Whistleblowers Policies We comply with applicable global legal requirements to protect whistleblowers. In addition to the Code of Business Conduct and Ethics, the following policies encourage employees and third parties to report ethics- related concerns and to protect those who report in good faith from retaliation. Policy Global Anti-Bribery and Anti-Corruption Policy Speak Up & Anti-Retaliation Policy Purpose Prevent, detect, and address allegations of corruption or bribery. Encourage open communication and reporting of suspected violations of laws, regulations, the Code of Business Conduct and Ethics, and other relevant policies, without fear of retaliation. Scope All argenx employees as well as consultants (including ad hoc contractors) working on behalf of argenx All argenx employees, extended workforce, and others who believe they have information to report Most senior level accountable Head of Ethics and Compliance Availability Internal document sharing system Externally available: Rules, Codes and Compliance Process for monitoring Reporting via the argenx Helpline and periodic monitoring by the Ethics and Compliance team. Applicability across sustainability statement Section 7.3.1 “Own Workforce (S1)” Section 7.4.1 “Business Conduct (G1)” Metrics Metric Unit 2025 2024 Percentage of employees that completed the Anti-Bribery Policy training % 95% 89% Measures to Protect Whistleblowers We expect all employees to promptly report suspected violations of laws, regulations, or company policies —including potential incidents of corruption or bribery—by notifying their manager, HR, Legal, or Ethics & Compliance, or by using the argenx Helpline. The argenx Helpline is externally managed to allow confidential and anonymous reporting. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Governance 318 G1-3 G1-1 MDR-P G1-3 MDR-M G1-1

Our policies, together with our Ethics and Compliance Investigations Global Procedure, prohibit retaliation against any individual who reports a concern or any person assists a reporter, cooperates with an investigation, responds to a request from regulators or government authorities, or exercises a legally protected right to report evidence of violations. Any form of retaliation will result in disciplinary action, up to and including termination of employment, revocation of site access, or discontinuance of services. All employees, consultants, and ad-hoc contractors are assigned an e-learning module as part of the annual Code of Conduct training, which includes whistleblowing procedures for raising concerns. Employees receiving whistleblowing reports are trained on a dedicated investigations standard operating procedure. 7.4.1.4. Anti-Corruption and Anti-Bribery Policies Anti-corruption and anti-bribery are addressed and communicated through our Code of Business Conduct and Ethics, (see Section 7.4.1.2. “Corporate Culture”), our Global Anti-Bribery and Anti-Corruption Policy and Speak Up & Anti-Retaliation Policy (see Section 7.4.1.3. “Protection of Whistleblowers”), our Third Party Partner Code of Conduct (see Section 7.4.1.5. “Supply Chain Management”), and additionally communicated via the intranet and onboarding sessions for new joiners. These policies aim to prevent, detect, and address potential allegations of corruption or bribery. We have reviewed these policies against peer practices to ensure consistency with industry standards. Further analysis is ongoing to assess alignment with the UN Convention against Corruption and Bribery. Actions All employees and our extended workforce, excluding managed service providers, participate in annual training related to anti-corruption and anti-bribery through an e-learning module, which includes information on the process for reporting concerns. New members of the Board of Directors are walked through the Global Anti-Bribery and Anti-Corruption Policy and the Code of Business Conduct and Ethics as part of their onboarding. They are informed of any key policy updates and receive regular updates on ethics and compliance matters. The Ethics and Compliance (E&C) function oversees the argenx Helpline, which serves as the central reporting channel for managing and triaging cases. The argenx Helpline was transitioned to a new system and enhanced in 2025 through integration with other argenx systems to improve reporting efficiency. All reporters are assured of confidentiality and non-retaliation. The E&C function is responsible for managing compliance investigations and consults with additional stakeholders, including Legal and HR, where necessary. If an allegation or concern is cross-functional and involves issues relevant to more than one function (e.g., includes HR-related issues or potential law violations), E&C will confer with HR and/or Legal to determine which business function will lead the investigation. We investigate all allegations and incidents of corruption and bribery with thoroughness, fairness, transparency and confidentiality. Investigations follow standardized procedures guided by the Ethics & Compliance Global Investigations Procedure, Speak Up & Anti-Retaliation Policy, and Code of Business Conduct and Ethics. All reports are reviewed and managed in accordance with applicable policies and legal requirements. All investigations are conducted by qualified personnel, and the specific individuals involved varies on a case by case basis. Investigations are tracked from intake to remediation and documented in a Compliance Investigation Report. Allegations or concerns received through the various reporting channels are promptly communicated to E&C for triage. Upon intake, E&C confirms receipt to reporters and issues an acknowledgment that reports of misconduct are taken seriously. If, based on the initial evaluation, it is determined that no further investigation is required, E&C will document the matter as closed. If the reporter wishes to remain anonymous, their anonymity will be protected to the fullest extent possible, unless disclosure is required by law or necessary to conduct the investigation and any related proceedings. Reports are shared regularly with the GCC and the Audit and Compliance Committee. E&C also conducts periodic monitoring which includes live monitoring of speaker events and advisory boards to ensure compliance with promotional, branding, and regulatory standards. Transaction argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Governance 319 G1-3 MDR-P G1-3 MDR-A

monitoring includes of sponsorships, donations, grants, donations, speaker programs, fee-for-service engagements, research grants and meals. Metrics Finance, HR, Procurement, Sales and Marketing and Supply Chain Management are considered to be functions at-risk due to the nature of roles involving more frequent interaction with high risk parties or patients. 100% of these functions-at-risk are covered by the anti-bribery and corruption training programs. The training program involves an e-learning that provides an overview of the Global Anti-Bribery and Anti- Corruption Policy and asks employees to confirm they have read the policy. Please refer to the G1-3 metric table which shows completion rate of the anti-bribery and corruption training by all argenx employees. Accounting Policies Training program figures include employees, contractors and extended workforce, excluding managed service providers. Metrics Incidents of corruption and bribery Metric Unit 2025 2024 Number of convictions for violation of anti-corruption and anti- bribery laws Number – – Amount of fines for violation of anti-corruption and anti-bribery laws € – – Accounting Policies Number of convictions is tracked through the argenx Helpline. Fines for violations are tracked through the argenx Helpline system, Legal team and outside counsel. Investigations are tracked in accordance with our Speak Up & Anti-Retaliation Policy and value chain violations are tracked through the argenx Helpline. 7.4.1.5. Supply Chain Management Policies Our Code of Business Conduct and Ethics, detailed in Section 7.4.1.2. “Corporate Culture”, outlines ethical standards for engaging with all third parties. It establishes that third parties are selected based on clear and objective criteria such as quality, capability, reputation, past performance, and price. This is applied in conjunction with our Third Party Partner Code of Conduct. Policy Third Party Partner Code of Conduct Purpose Outlines standards expected of third-party partners, including guidance around anti- bribery and anti-corruption. Scope All global third-party partners and those engaged by third parties on behalf of argenx Most senior level accountable General Counsel and Corporate Secretary Availability Externally available: Rules, Codes and Compliance Process for monitoring Reporting via the argenx Helpline Applicability across sustainability statement Section 7.4.1 “Business Conduct (G1)” The standard payment terms, which apply to all supplier categories, are 30-days in instances where other terms have not been contractually agreed upon. While we view payment practices as a fundamental component of vendor management, we do not currently maintain a formal payment practices policy or a separate policy for preventing late payments to small or medium-sized enterprises , instead our standard payment terms and contractual agreements are applied to all suppliers, including small or medium-sized enterprises. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Governance 320 G1-4 MDR-M G1-2 MDR-P G1-3 MDR-M

Actions Outsourcing and co-creation are key elements of our business strategy, enabling us to leverage external expertise and resources. Recognizing the important role of suppliers in this model, we have developed a supply chain management approach focused on defining expectations, qualifying suppliers, and monitoring performance. Our vendor qualification and due diligence process assesses new suppliers for compliance with regulatory and company standards, including reference checks, screening, and where applicable review of the third- party’s own Code of Conduct. Social due diligence includes verification of adherence to mandatory regulatory requirements such as worker rights. Environmental criteria are not currently used in supplier selection. The Global Sourcing and Vendor Alliance Management team oversees relationships with Development suppliers, along with functional business owners who manage collaboration with their respective third parties. We engage suppliers directly through regular meetings with business owners who oversee the operational aspects of service performance and delivery. Partnership governance structures are established with key suppliers to define collaboration from strategic through operational levels, including objectives, meeting cadence, and accountability. This governance structure is continuously evaluated. We have established a working group focused on strengthening third-party risk-management, including early risk detection, compliance control, and process simplification. Risk management is also integrated into the supply chain management approach through the supply chain maps process, which visualizes the VYVGART supply chain, logistics and quality-assurance requirements. Suppliers across our supply chain, from development vendors, manufacturing, and distribution to our commercial and technology vendors, undergo a qualification process, periodic audits, and performance reviews to maintain quality standards and manage supplier performance. Quality or compliance issues are escalated through governance channels. If low-quality performance, supply chain disruptions, or non-compliance with supplier agreements are identified (e.g.,through audits), corrective action plans are implemented and monitored. Persistent under-performance without resolution may result in termination of a supplier relationship. Payment Practices We follow standardized procure-to-pay processes to prevent late payments. Payment runs are conducted according to a standard payment cycle determined by country of jurisdiction. Weekly payment runs were conducted for all European and US entities, while in Japan, all duly approved invoices were paid once a month. Targets We currently apply a qualitative approach to management of suppliers and have not yet established formal quantitative targets. Metrics Payment practices Metric Unit 2025 2024 Standard payment terms in number of days by main category of suppliers Days 30 30 Average time to pay an invoice from the invoice date Days 35 30 Invoices paid within standard payment terms % 76 76 Number of legal proceedings currently outstanding for late payments Number – – Accounting Policies Our payment practices, including average payment time compared to standard payment terms, were calculated using data extracted from our payment system software. The analysis covered all invoices paid to registered vendors and excluded payments to employees and intercompany payments. Standard argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Governance 321 G1-2 MDR-A G1-6 G1-6 MDR-T G1-1 MDR-M

payment terms are 30-days, unless alternative terms are contractually agreed. The average payment time was determined by dividing the total number of calendar days between invoice date and payment date by the total number of paid invoices, calculated for each business unit, region, and the entire group. 7.4.1.6. Animal Welfare Policies The Animal Welfare Policy outlines our approach to safeguarding the welfare of animals used in research. The policy is assigned as a read-and-acknowledge requirement for all employees. Policy Animal Welfare Policy Purpose Provide guidance and define key principles of replacement, reduction and refinement (the “3Rs”) aimed at promoting and safeguarding the welfare of animals used in research by or on behalf of argenx. Scope All individuals conducting business on behalf of argenx Most senior level accountable Head of Pharmtox Availability Externally available: Rules, Codes and Compliance Process for monitoring Accreditation requirements for CROs, collaborators and vendors and a due diligence process to monitor the implementation of the policy. Applicability across sustainability statement Section 7.4.1 “Business Conduct (G1)” argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Governance 322 G1-1 MDR-P
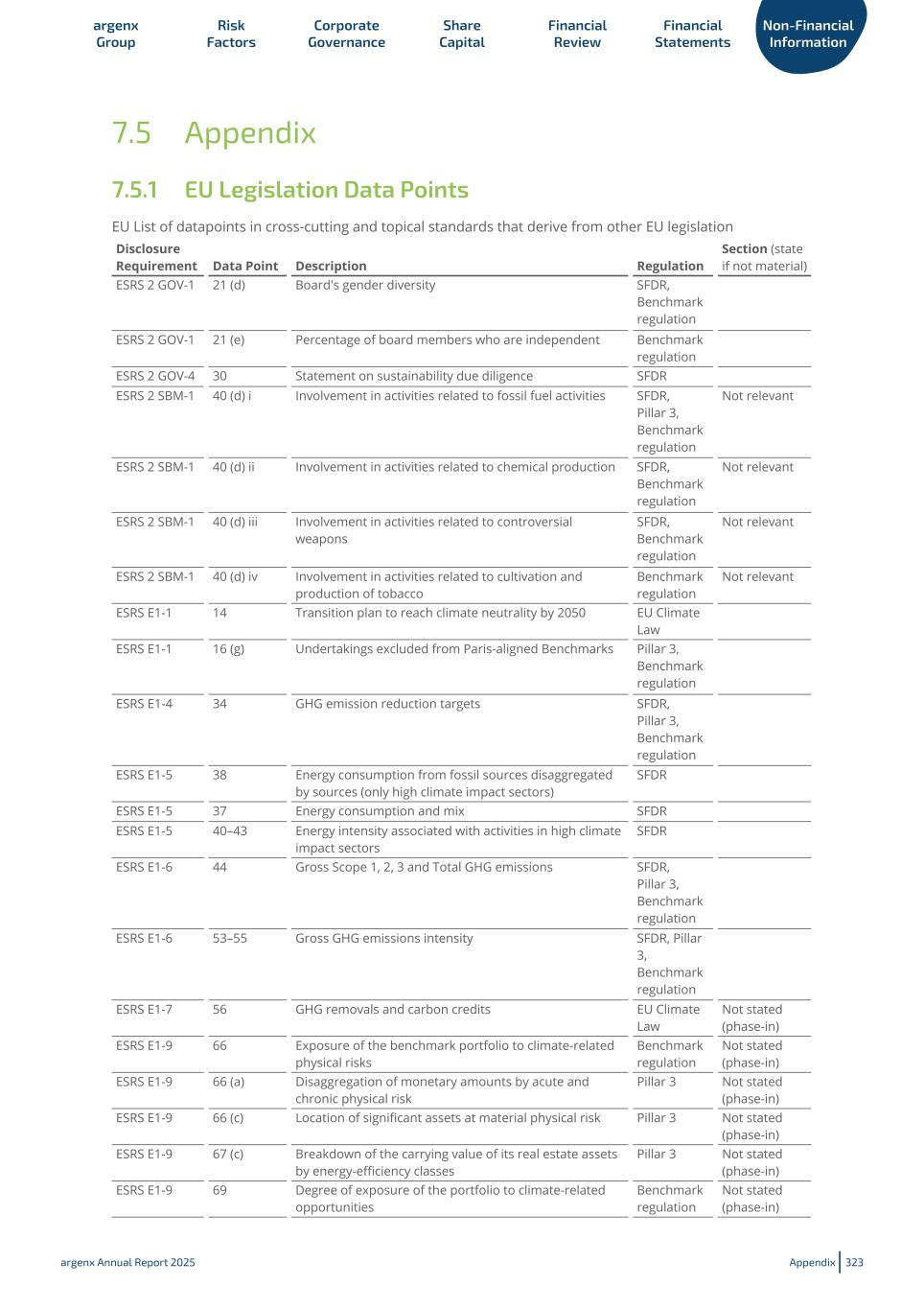
7.5 Appendix 7.5.1 EU Legislation Data Points EU List of datapoints in cross-cutting and topical standards that derive from other EU legislation ESRS 2 GOV-1 21 (d) Board's gender diversity SFDR, Benchmark regulation ESRS 2 GOV-1 21 (e) Percentage of board members who are independent Benchmark regulation ESRS 2 GOV-4 30 Statement on sustainability due diligence SFDR ESRS 2 SBM-1 40 (d) i Involvement in activities related to fossil fuel activities SFDR, Pillar 3, Benchmark regulation Not relevant ESRS 2 SBM-1 40 (d) ii Involvement in activities related to chemical production SFDR, Benchmark regulation Not relevant ESRS 2 SBM-1 40 (d) iii Involvement in activities related to controversial weapons SFDR, Benchmark regulation Not relevant ESRS 2 SBM-1 40 (d) iv Involvement in activities related to cultivation and production of tobacco Benchmark regulation Not relevant ESRS E1-1 14 Transition plan to reach climate neutrality by 2050 EU Climate Law ESRS E1-1 16 (g) Undertakings excluded from Paris-aligned Benchmarks Pillar 3, Benchmark regulation ESRS E1-4 34 GHG emission reduction targets SFDR, Pillar 3, Benchmark regulation ESRS E1-5 38 Energy consumption from fossil sources disaggregated by sources (only high climate impact sectors) SFDR ESRS E1-5 37 Energy consumption and mix SFDR ESRS E1-5 40–43 Energy intensity associated with activities in high climate impact sectors SFDR ESRS E1-6 44 Gross Scope 1, 2, 3 and Total GHG emissions SFDR, Pillar 3, Benchmark regulation ESRS E1-6 53–55 Gross GHG emissions intensity SFDR, Pillar 3, Benchmark regulation ESRS E1-7 56 GHG removals and carbon credits EU Climate Law Not stated (phase-in) ESRS E1-9 66 Exposure of the benchmark portfolio to climate-related physical risks Benchmark regulation Not stated (phase-in) ESRS E1-9 66 (a) Disaggregation of monetary amounts by acute and chronic physical risk Pillar 3 Not stated (phase-in) ESRS E1-9 66 (c) Location of significant assets at material physical risk Pillar 3 Not stated (phase-in) ESRS E1-9 67 (c) Breakdown of the carrying value of its real estate assets by energy-efficiency classes Pillar 3 Not stated (phase-in) ESRS E1-9 69 Degree of exposure of the portfolio to climate-related opportunities Benchmark regulation Not stated (phase-in) Disclosure Requirement Data Point Description Regulation Section (state if not material) argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Appendix 323
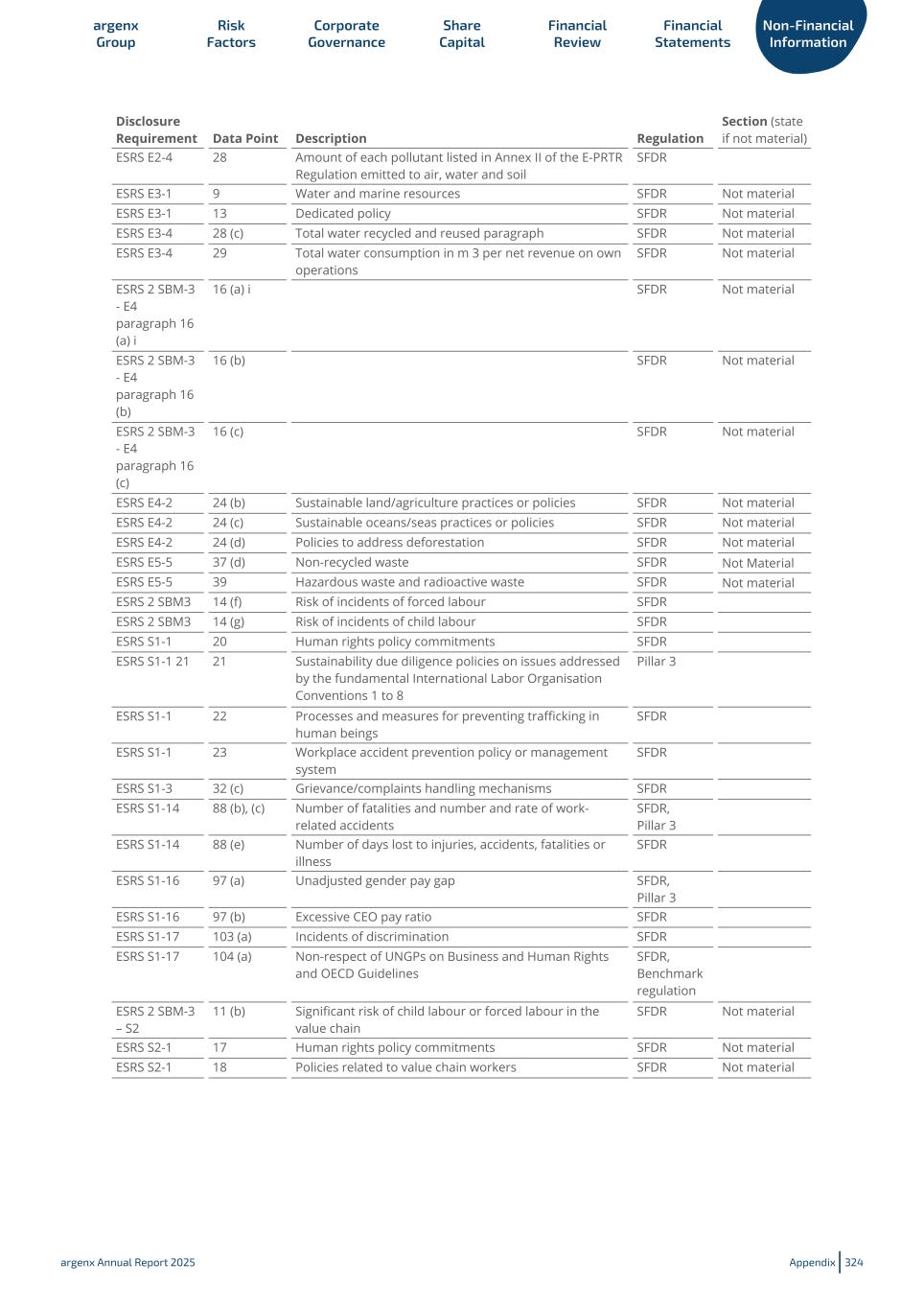
ESRS E2-4 28 Amount of each pollutant listed in Annex II of the E-PRTR Regulation emitted to air, water and soil SFDR ESRS E3-1 9 Water and marine resources SFDR Not material ESRS E3-1 13 Dedicated policy SFDR Not material ESRS E3-4 28 (c) Total water recycled and reused paragraph SFDR Not material ESRS E3-4 29 Total water consumption in m 3 per net revenue on own operations SFDR Not material ESRS 2 SBM-3 - E4 paragraph 16 (a) i 16 (a) i SFDR Not material ESRS 2 SBM-3 - E4 paragraph 16 (b) 16 (b) SFDR Not material ESRS 2 SBM-3 - E4 paragraph 16 (c) 16 (c) SFDR Not material ESRS E4-2 24 (b) Sustainable land/agriculture practices or policies SFDR Not material ESRS E4-2 24 (c) Sustainable oceans/seas practices or policies SFDR Not material ESRS E4-2 24 (d) Policies to address deforestation SFDR Not material ESRS E5-5 37 (d) Non-recycled waste SFDR Not Material ESRS E5-5 39 Hazardous waste and radioactive waste SFDR Not material ESRS 2 SBM3 14 (f) Risk of incidents of forced labour SFDR ESRS 2 SBM3 14 (g) Risk of incidents of child labour SFDR ESRS S1-1 20 Human rights policy commitments SFDR ESRS S1-1 21 21 Sustainability due diligence policies on issues addressed by the fundamental International Labor Organisation Conventions 1 to 8 Pillar 3 ESRS S1-1 22 Processes and measures for preventing trafficking in human beings SFDR ESRS S1-1 23 Workplace accident prevention policy or management system SFDR ESRS S1-3 32 (c) Grievance/complaints handling mechanisms SFDR ESRS S1-14 88 (b), (c) Number of fatalities and number and rate of work- related accidents SFDR, Pillar 3 ESRS S1-14 88 (e) Number of days lost to injuries, accidents, fatalities or illness SFDR ESRS S1-16 97 (a) Unadjusted gender pay gap SFDR, Pillar 3 ESRS S1-16 97 (b) Excessive CEO pay ratio SFDR ESRS S1-17 103 (a) Incidents of discrimination SFDR ESRS S1-17 104 (a) Non-respect of UNGPs on Business and Human Rights and OECD Guidelines SFDR, Benchmark regulation ESRS 2 SBM-3 – S2 11 (b) Significant risk of child labour or forced labour in the value chain SFDR Not material ESRS S2-1 17 Human rights policy commitments SFDR Not material ESRS S2-1 18 Policies related to value chain workers SFDR Not material Disclosure Requirement Data Point Description Regulation Section (state if not material) argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Appendix 324
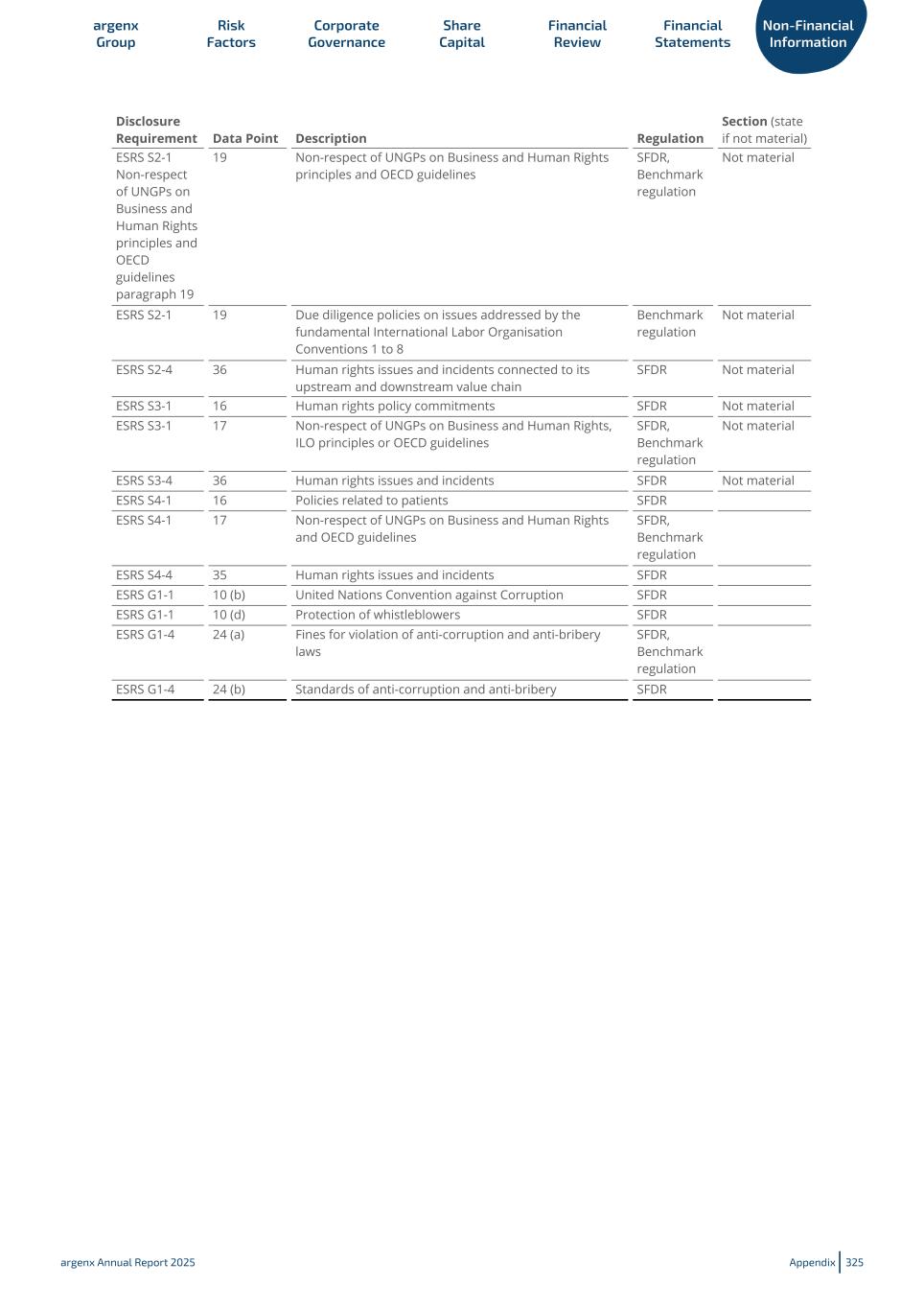
ESRS S2-1 Non-respect of UNGPs on Business and Human Rights principles and OECD guidelines paragraph 19 19 Non-respect of UNGPs on Business and Human Rights principles and OECD guidelines SFDR, Benchmark regulation Not material ESRS S2-1 19 Due diligence policies on issues addressed by the fundamental International Labor Organisation Conventions 1 to 8 Benchmark regulation Not material ESRS S2-4 36 Human rights issues and incidents connected to its upstream and downstream value chain SFDR Not material ESRS S3-1 16 Human rights policy commitments SFDR Not material ESRS S3-1 17 Non-respect of UNGPs on Business and Human Rights, ILO principles or OECD guidelines SFDR, Benchmark regulation Not material ESRS S3-4 36 Human rights issues and incidents SFDR Not material ESRS S4-1 16 Policies related to patients SFDR ESRS S4-1 17 Non-respect of UNGPs on Business and Human Rights and OECD guidelines SFDR, Benchmark regulation ESRS S4-4 35 Human rights issues and incidents SFDR ESRS G1-1 10 (b) United Nations Convention against Corruption SFDR ESRS G1-1 10 (d) Protection of whistleblowers SFDR ESRS G1-4 24 (a) Fines for violation of anti-corruption and anti-bribery laws SFDR, Benchmark regulation ESRS G1-4 24 (b) Standards of anti-corruption and anti-bribery SFDR Disclosure Requirement Data Point Description Regulation Section (state if not material) argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Appendix 325
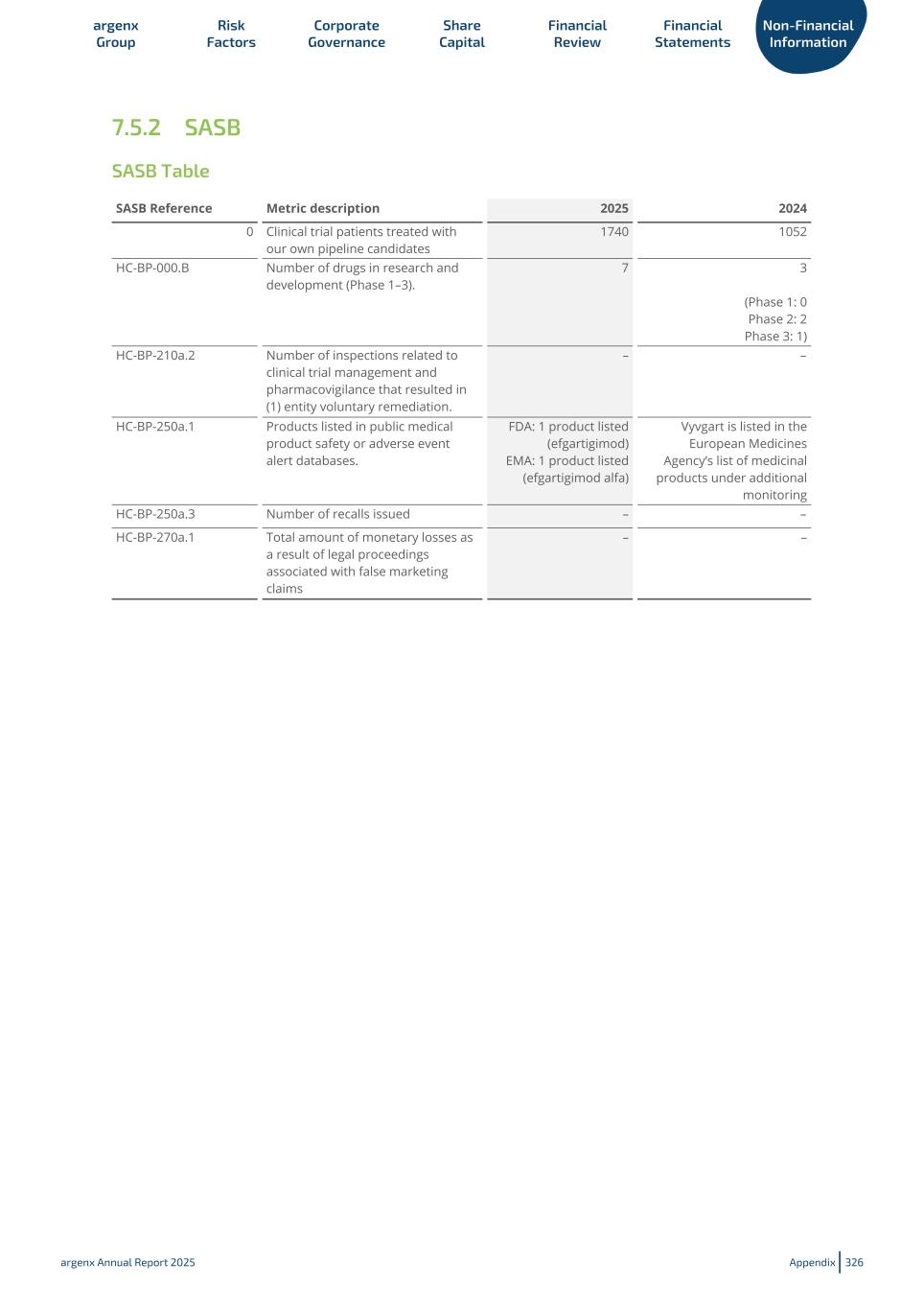
7.5.2 SASB SASB Table 0 Clinical trial patients treated with our own pipeline candidates 1740 1052 HC-BP-000.B Number of drugs in research and development (Phase 1–3). 7 3 (Phase 1: 0 Phase 2: 2 Phase 3: 1) HC-BP-210a.2 Number of inspections related to clinical trial management and pharmacovigilance that resulted in (1) entity voluntary remediation. – – HC-BP-250a.1 Products listed in public medical product safety or adverse event alert databases. FDA: 1 product listed (efgartigimod) EMA: 1 product listed (efgartigimod alfa) Vyvgart is listed in the European Medicines Agency’s list of medicinal products under additional monitoring HC-BP-250a.3 Number of recalls issued – – HC-BP-270a.1 Total amount of monetary losses as a result of legal proceedings associated with false marketing claims – – SASB Reference Metric description 2025 2024 argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Appendix 326

Glossary 8.1 Management Confirmations 328 8.2 Definitions 329 argenx Annual Report 2025 327 8

8 Glossary 8.1 Management Confirmations With due regard to best practice provision 1.4.3 of the DCGC, we confirm that: i. the report of the board of directors provides sufficient insights into any deficiency in the effectiveness of the internal risk management and control systems, as is further substantiated in Section 2 “Risk Factors” and Section 3 “Corporate Governance”. The Audit and Compliance Committee executes the oversight with respect to management’s implementation and substantiation of DCGC. ii. the internal risk and control systems described herein, particularly in paragraph 3.9.5 “Financial Risks and Controls” provide reasonable assurance that the financial reporting does not contain any material inaccuracies; iii. the internal risk and control systems provide limited assurance that sustainability reporting is free from material misstatements; iv. in light of and as set out in this report, while the company seeks to control operational and compliance risks in the manner and to the extent as described in this report, the Board is not aware that our internal risk management and control systems, would not provide sufficient comfort1) that the material operational and compliance risks faced by the company are effectively controlled in line with the risk appetite as of December 31, 2025; v. we confirm that we expect that our existing cash and cash equivalents and current financial assets will enable us to fund our operating expenses and capital expenditure requirements through at least the next 12 months. On the basis of the current state of affairs, it is justified that the financial reporting is prepared on a going concern basis; and vi. the report of the board of directors, particularly Section 2 “Risk Factors” includes the material risks, as referred to in best practice provision 1.2.1 of the DCGC as well as uncertainties, to the extent that they are relevant for our continuity for a period of 12 months after the preparation of this Annual Report. The aforementioned statement does not in any way limit the relevance or applicability of the Risk Factors set out in this Annual Report to the aforementioned period of 12 months. 1) Sufficient comfort is to be read as: comfort considering our risk appetite, the complexity of our enterprise, inherent limitations to these systems and other disclosures on these systems in our board report. argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Cross Reference table for annual reporting requirements 328
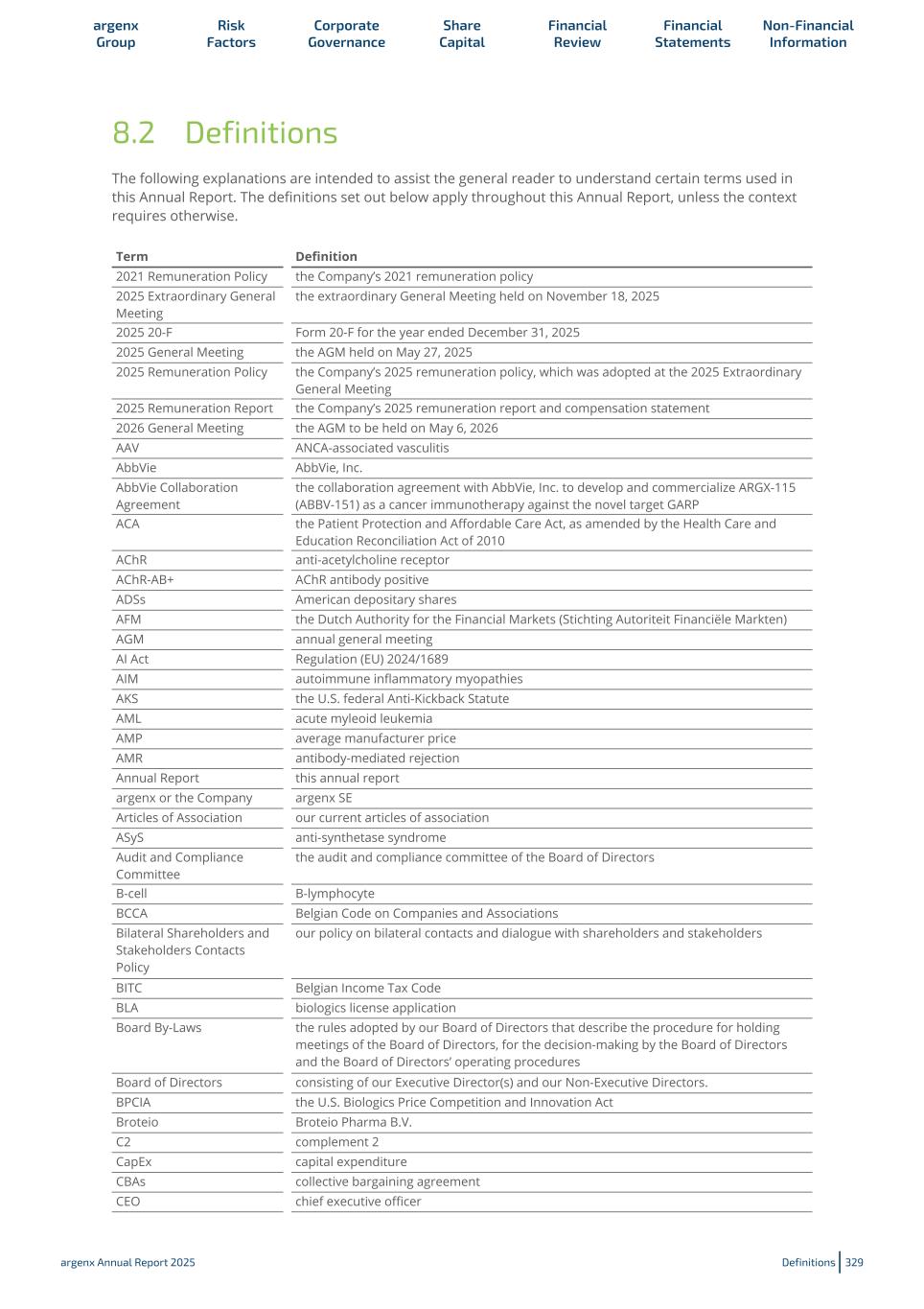
8.2 Definitions The following explanations are intended to assist the general reader to understand certain terms used in this Annual Report. The definitions set out below apply throughout this Annual Report, unless the context requires otherwise. 2021 Remuneration Policy the Company’s 2021 remuneration policy 2025 Extraordinary General Meeting the extraordinary General Meeting held on November 18, 2025 2025 20-F Form 20-F for the year ended December 31, 2025 2025 General Meeting the AGM held on May 27, 2025 2025 Remuneration Policy the Company’s 2025 remuneration policy, which was adopted at the 2025 Extraordinary General Meeting 2025 Remuneration Report the Company’s 2025 remuneration report and compensation statement 2026 General Meeting the AGM to be held on May 6, 2026 AAV ANCA-associated vasculitis AbbVie AbbVie, Inc. AbbVie Collaboration Agreement the collaboration agreement with AbbVie, Inc. to develop and commercialize ARGX-115 (ABBV-151) as a cancer immunotherapy against the novel target GARP ACA the Patient Protection and Affordable Care Act, as amended by the Health Care and Education Reconciliation Act of 2010 AChR anti-acetylcholine receptor AChR-AB+ AChR antibody positive ADSs American depositary shares AFM the Dutch Authority for the Financial Markets (Stichting Autoriteit Financiële Markten) AGM annual general meeting AI Act Regulation (EU) 2024/1689 AIM autoimmune inflammatory myopathies AKS the U.S. federal Anti-Kickback Statute AML acute myleoid leukemia AMP average manufacturer price AMR antibody-mediated rejection Annual Report this annual report argenx or the Company argenx SE Articles of Association our current articles of association ASyS anti-synthetase syndrome Audit and Compliance Committee the audit and compliance committee of the Board of Directors B-cell B-lymphocyte BCCA Belgian Code on Companies and Associations Bilateral Shareholders and Stakeholders Contacts Policy our policy on bilateral contacts and dialogue with shareholders and stakeholders BITC Belgian Income Tax Code BLA biologics license application Board By-Laws the rules adopted by our Board of Directors that describe the procedure for holding meetings of the Board of Directors, for the decision-making by the Board of Directors and the Board of Directors’ operating procedures Board of Directors consisting of our Executive Director(s) and our Non-Executive Directors. BPCIA the U.S. Biologics Price Competition and Innovation Act Broteio Broteio Pharma B.V. C2 complement 2 CapEx capital expenditure CBAs collective bargaining agreement CEO chief executive officer Term Definition argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Definitions 329
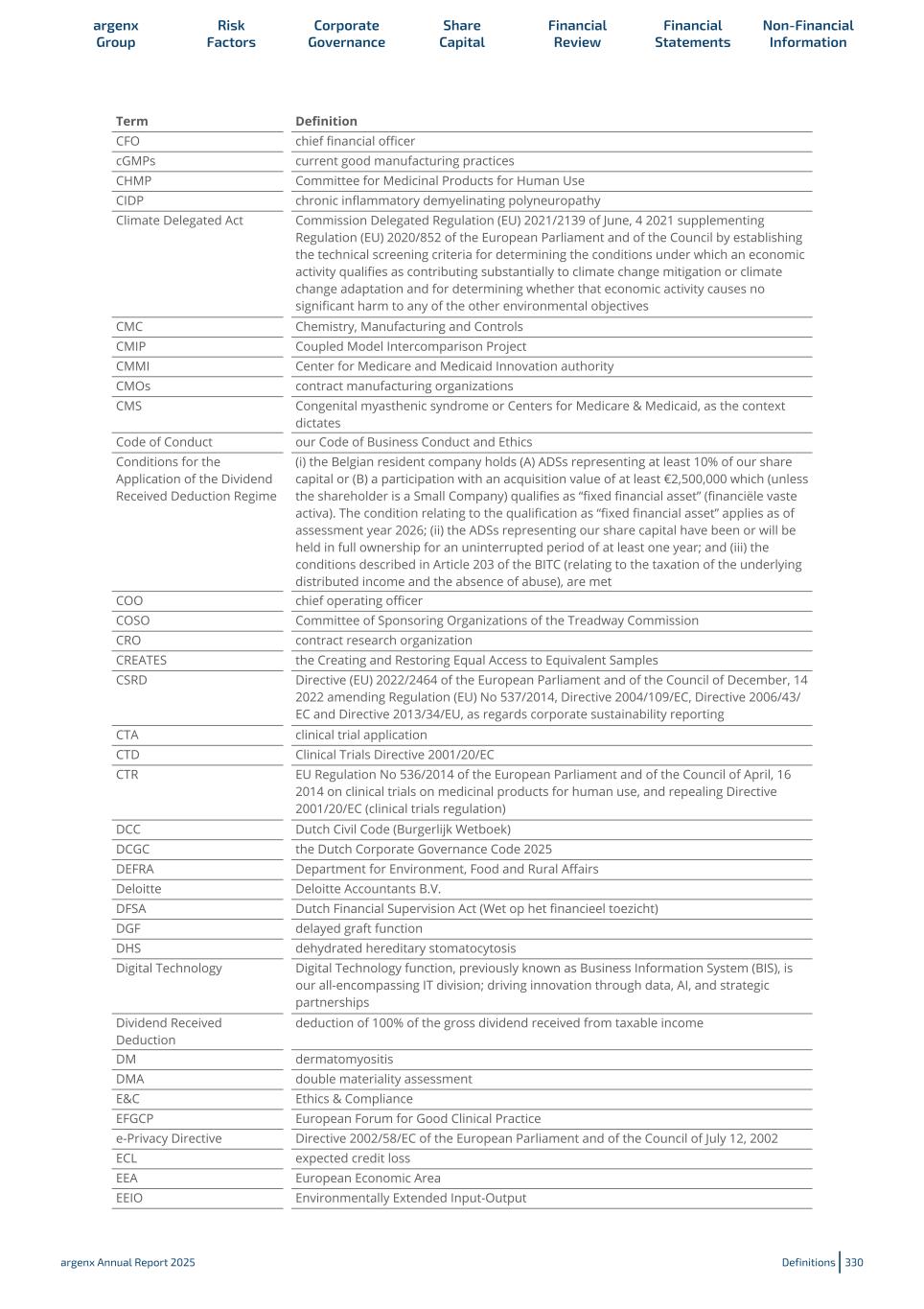
CFO chief financial officer cGMPs current good manufacturing practices CHMP Committee for Medicinal Products for Human Use CIDP chronic inflammatory demyelinating polyneuropathy Climate Delegated Act Commission Delegated Regulation (EU) 2021/2139 of June, 4 2021 supplementing Regulation (EU) 2020/852 of the European Parliament and of the Council by establishing the technical screening criteria for determining the conditions under which an economic activity qualifies as contributing substantially to climate change mitigation or climate change adaptation and for determining whether that economic activity causes no significant harm to any of the other environmental objectives CMC Chemistry, Manufacturing and Controls CMIP Coupled Model Intercomparison Project CMMI Center for Medicare and Medicaid Innovation authority CMOs contract manufacturing organizations CMS Congenital myasthenic syndrome or Centers for Medicare & Medicaid, as the context dictates Code of Conduct our Code of Business Conduct and Ethics Conditions for the Application of the Dividend Received Deduction Regime (i) the Belgian resident company holds (A) ADSs representing at least 10% of our share capital or (B) a participation with an acquisition value of at least €2,500,000 which (unless the shareholder is a Small Company) qualifies as “fixed financial asset” (financiële vaste activa). The condition relating to the qualification as “fixed financial asset” applies as of assessment year 2026; (ii) the ADSs representing our share capital have been or will be held in full ownership for an uninterrupted period of at least one year; and (iii) the conditions described in Article 203 of the BITC (relating to the taxation of the underlying distributed income and the absence of abuse), are met COO chief operating officer COSO Committee of Sponsoring Organizations of the Treadway Commission CRO contract research organization CREATES the Creating and Restoring Equal Access to Equivalent Samples CSRD Directive (EU) 2022/2464 of the European Parliament and of the Council of December, 14 2022 amending Regulation (EU) No 537/2014, Directive 2004/109/EC, Directive 2006/43/ EC and Directive 2013/34/EU, as regards corporate sustainability reporting CTA clinical trial application CTD Clinical Trials Directive 2001/20/EC CTR EU Regulation No 536/2014 of the European Parliament and of the Council of April, 16 2014 on clinical trials on medicinal products for human use, and repealing Directive 2001/20/EC (clinical trials regulation) DCC Dutch Civil Code (Burgerlijk Wetboek) DCGC the Dutch Corporate Governance Code 2025 DEFRA Department for Environment, Food and Rural Affairs Deloitte Deloitte Accountants B.V. DFSA Dutch Financial Supervision Act (Wet op het financieel toezicht) DGF delayed graft function DHS dehydrated hereditary stomatocytosis Digital Technology Digital Technology function, previously known as Business Information System (BIS), is our all-encompassing IT division; driving innovation through data, AI, and strategic partnerships Dividend Received Deduction deduction of 100% of the gross dividend received from taxable income DM dermatomyositis DMA double materiality assessment E&C Ethics & Compliance EFGCP European Forum for Good Clinical Practice e-Privacy Directive Directive 2002/58/EC of the European Parliament and of the Council of July 12, 2002 ECL expected credit loss EEA European Economic Area EEIO Environmentally Extended Input-Output Term Definition argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Definitions 330
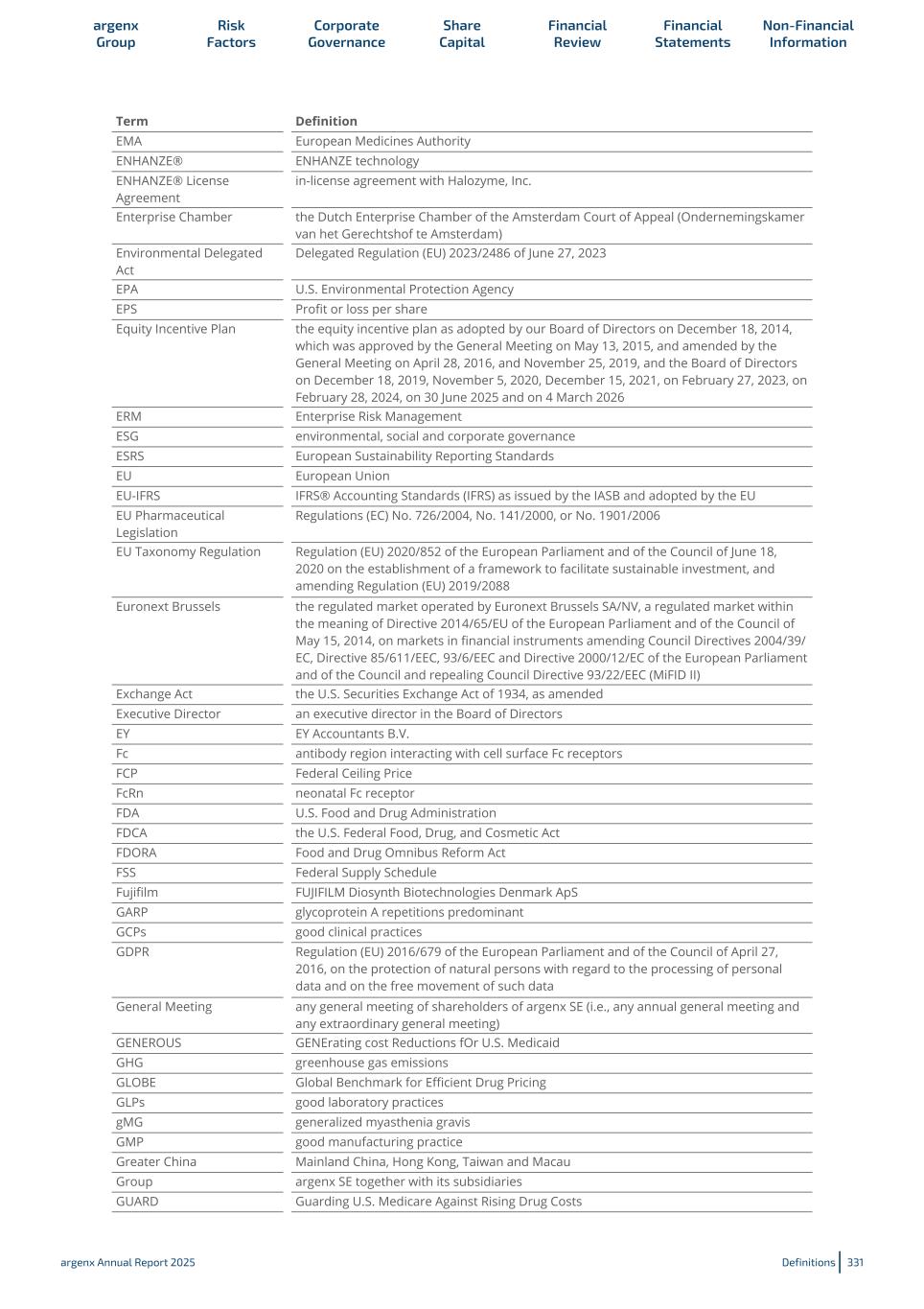
EMA European Medicines Authority ENHANZE® ENHANZE technology ENHANZE® License Agreement in-license agreement with Halozyme, Inc. Enterprise Chamber the Dutch Enterprise Chamber of the Amsterdam Court of Appeal (Ondernemingskamer van het Gerechtshof te Amsterdam) Environmental Delegated Act Delegated Regulation (EU) 2023/2486 of June 27, 2023 EPA U.S. Environmental Protection Agency EPS Profit or loss per share Equity Incentive Plan the equity incentive plan as adopted by our Board of Directors on December 18, 2014, which was approved by the General Meeting on May 13, 2015, and amended by the General Meeting on April 28, 2016, and November 25, 2019, and the Board of Directors on December 18, 2019, November 5, 2020, December 15, 2021, on February 27, 2023, on February 28, 2024, on 30 June 2025 and on 4 March 2026 ERM Enterprise Risk Management ESG environmental, social and corporate governance ESRS European Sustainability Reporting Standards EU European Union EU-IFRS IFRS® Accounting Standards (IFRS) as issued by the IASB and adopted by the EU EU Pharmaceutical Legislation Regulations (EC) No. 726/2004, No. 141/2000, or No. 1901/2006 EU Taxonomy Regulation Regulation (EU) 2020/852 of the European Parliament and of the Council of June 18, 2020 on the establishment of a framework to facilitate sustainable investment, and amending Regulation (EU) 2019/2088 Euronext Brussels the regulated market operated by Euronext Brussels SA/NV, a regulated market within the meaning of Directive 2014/65/EU of the European Parliament and of the Council of May 15, 2014, on markets in financial instruments amending Council Directives 2004/39/ EC, Directive 85/611/EEC, 93/6/EEC and Directive 2000/12/EC of the European Parliament and of the Council and repealing Council Directive 93/22/EEC (MiFID II) Exchange Act the U.S. Securities Exchange Act of 1934, as amended Executive Director an executive director in the Board of Directors EY EY Accountants B.V. Fc antibody region interacting with cell surface Fc receptors FCP Federal Ceiling Price FcRn neonatal Fc receptor FDA U.S. Food and Drug Administration FDCA the U.S. Federal Food, Drug, and Cosmetic Act FDORA Food and Drug Omnibus Reform Act FSS Federal Supply Schedule Fujifilm FUJIFILM Diosynth Biotechnologies Denmark ApS GARP glycoprotein A repetitions predominant GCPs good clinical practices GDPR Regulation (EU) 2016/679 of the European Parliament and of the Council of April 27, 2016, on the protection of natural persons with regard to the processing of personal data and on the free movement of such data General Meeting any general meeting of shareholders of argenx SE (i.e., any annual general meeting and any extraordinary general meeting) GENEROUS GENErating cost Reductions fOr U.S. Medicaid GHG greenhouse gas emissions GLOBE Global Benchmark for Efficient Drug Pricing GLPs good laboratory practices gMG generalized myasthenia gravis GMP good manufacturing practice Greater China Mainland China, Hong Kong, Taiwan and Macau Group argenx SE together with its subsidiaries GUARD Guarding U.S. Medicare Against Rising Drug Costs Term Definition argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Definitions 331

Halozyme Halozyme Therapeutics, Inc. Hatch-Waxman Act the U.S. Drug Price Competition and Patent Term Restoration Act of 1984 HHS U.S. Department of Health and Human Services HIPAA the U.S. federal Health Insurance Portability and Accountability Act of 1996 HITECH Health Information Technology for Economic and Clinical Health Act of 2009 IASB International Accounting Standards Board IEA International Energy Agency IFRS IFRS® Accounting Standards IgA Immunoglobulin A IgG Immunoglobulin G IgM Immunoglobulin M IIP immunology innovation program IMM irreversible morbidity or mortality IMNM immune-mediated necrotizing myopathy IND investigational new drug IPCC Intergovernmental Panel on Climate Change IRA Inflation Reduction Act IRB institutional review board IROs impacts, risks, and opportunities ISAE International Standard on Assurance Engagements ISMS Information Security and Management System ITP immune thrombocytopenia IV intravenous IVIg intravenous IgG KPI key performance indicator LEI European legal entity identifier number Lonza Lonza Sales AG LTIP Long term incentive plan MA marketing authorization MAA marketing authorization application mAb monoclonal antibody MAH marketing authorization holder Mainland China mainland China MAR Regulation (EU) No 596/2014 of the European Parliament and of the Council of April 2014 on market abuse (market abuse regulation) and repealing Directive 2003/6/EC European Parliament and of the Council and Commission Directives 2003/124/EC, 2003/ EC and 2004/72/EC, and the rules and regulations promulgated pursuant thereto MFN most favored nation MG myasthenia gravis MG-ADL Myasthenia Gravis Activities of Daily Living MHLW Ministry of Health, Labour and Welfare MMN multifocal motor neuropathy MN membranous nephropathy MSE minimal symptom expression MuSK muscle-specific kinase Myositis idiopathic inflammatory myopathies Nasdaq the Nasdaq Global Select Market Nasdaq Listing Rules the listing rules of the Nasdaq Global Market NDA new drug application NEO named executive officer New Capital Gains Tax the capital gains tax on financial assets (such as the ADSs) for capital gains realized as from January 1, 2026 as agreed to be introduced by the Belgian federal government new Product Liability Directive Directive (EU) 2024/2853 Term Definition argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Definitions 332
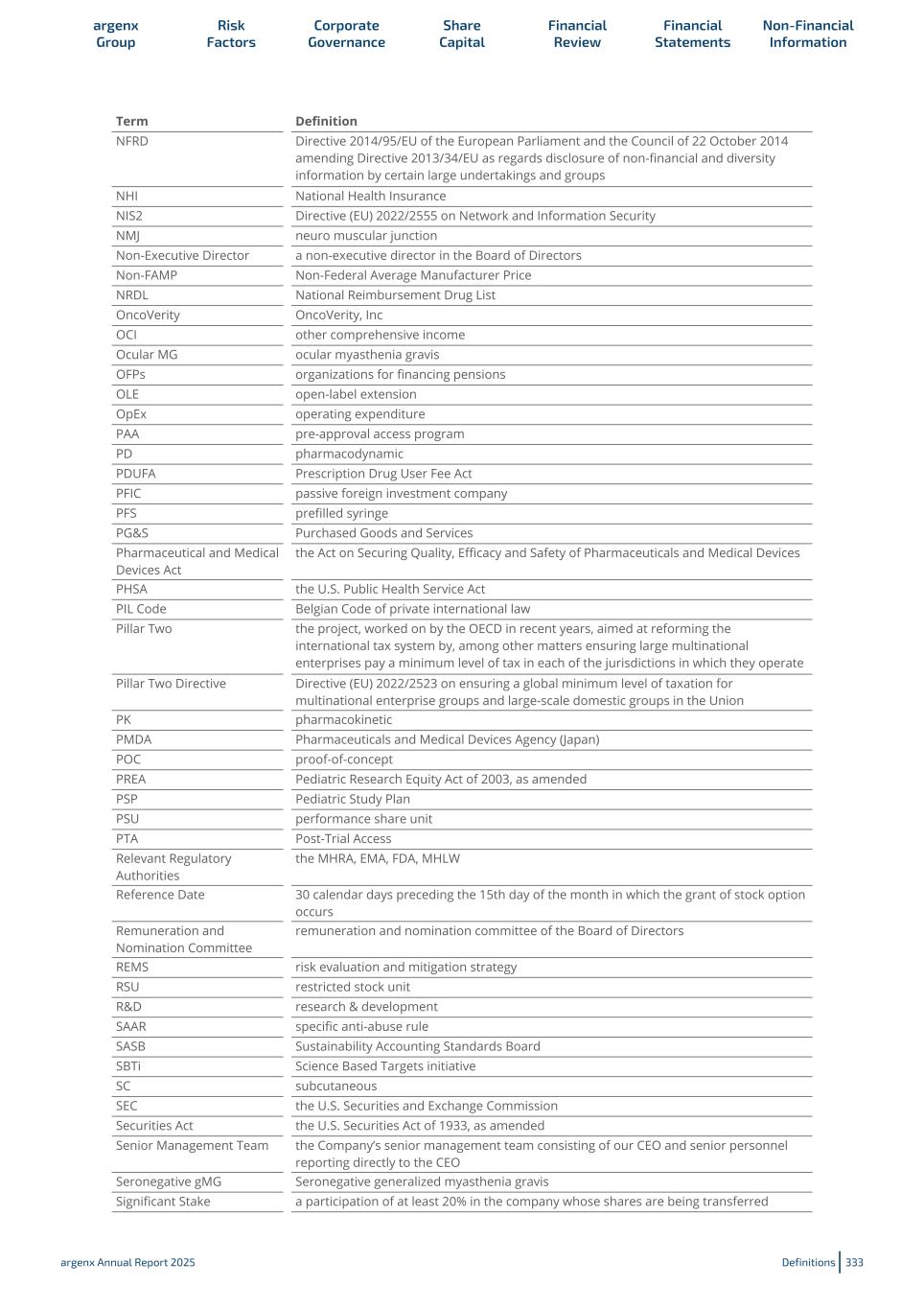
NFRD Directive 2014/95/EU of the European Parliament and the Council of 22 October 2014 amending Directive 2013/34/EU as regards disclosure of non-financial and diversity information by certain large undertakings and groups NHI National Health Insurance NIS2 Directive (EU) 2022/2555 on Network and Information Security NMJ neuro muscular junction Non-Executive Director a non-executive director in the Board of Directors Non-FAMP Non-Federal Average Manufacturer Price NRDL National Reimbursement Drug List OncoVerity OncoVerity, Inc OCI other comprehensive income Ocular MG ocular myasthenia gravis OFPs organizations for financing pensions OLE open-label extension OpEx operating expenditure PAA pre-approval access program PD pharmacodynamic PDUFA Prescription Drug User Fee Act PFIC passive foreign investment company PFS prefilled syringe PG&S Purchased Goods and Services Pharmaceutical and Medical Devices Act the Act on Securing Quality, Efficacy and Safety of Pharmaceuticals and Medical Devices PHSA the U.S. Public Health Service Act PIL Code Belgian Code of private international law Pillar Two the project, worked on by the OECD in recent years, aimed at reforming the international tax system by, among other matters ensuring large multinational enterprises pay a minimum level of tax in each of the jurisdictions in which they operate Pillar Two Directive Directive (EU) 2022/2523 on ensuring a global minimum level of taxation for multinational enterprise groups and large-scale domestic groups in the Union PK pharmacokinetic PMDA Pharmaceuticals and Medical Devices Agency (Japan) POC proof-of-concept PREA Pediatric Research Equity Act of 2003, as amended PSP Pediatric Study Plan PSU performance share unit PTA Post-Trial Access Relevant Regulatory Authorities the MHRA, EMA, FDA, MHLW Reference Date 30 calendar days preceding the 15th day of the month in which the grant of stock option occurs Remuneration and Nomination Committee remuneration and nomination committee of the Board of Directors REMS risk evaluation and mitigation strategy RSU restricted stock unit R&D research & development SAAR specific anti-abuse rule SASB Sustainability Accounting Standards Board SBTi Science Based Targets initiative SC subcutaneous SEC the U.S. Securities and Exchange Commission Securities Act the U.S. Securities Act of 1933, as amended Senior Management Team the Company’s senior management team consisting of our CEO and senior personnel reporting directly to the CEO Seronegative gMG Seronegative generalized myasthenia gravis Significant Stake a participation of at least 20% in the company whose shares are being transferred Term Definition argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Definitions 333
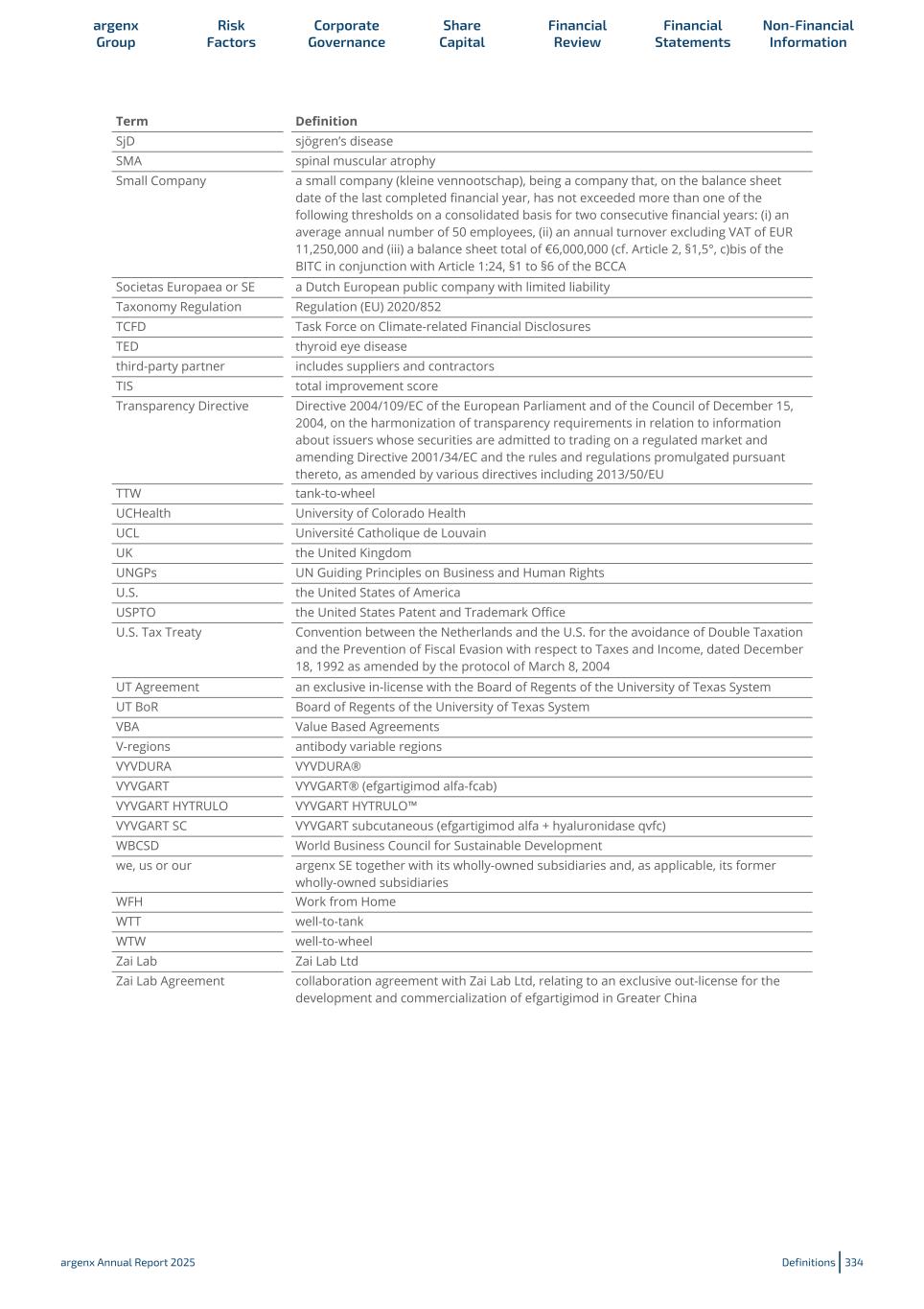
SjD sjögren’s disease SMA spinal muscular atrophy Small Company a small company (kleine vennootschap), being a company that, on the balance sheet date of the last completed financial year, has not exceeded more than one of the following thresholds on a consolidated basis for two consecutive financial years: (i) an average annual number of 50 employees, (ii) an annual turnover excluding VAT of EUR 11,250,000 and (iii) a balance sheet total of €6,000,000 (cf. Article 2, §1,5°, c)bis of the BITC in conjunction with Article 1:24, §1 to §6 of the BCCA Societas Europaea or SE a Dutch European public company with limited liability Taxonomy Regulation Regulation (EU) 2020/852 TCFD Task Force on Climate-related Financial Disclosures TED thyroid eye disease third-party partner includes suppliers and contractors TIS total improvement score Transparency Directive Directive 2004/109/EC of the European Parliament and of the Council of December 15, 2004, on the harmonization of transparency requirements in relation to information about issuers whose securities are admitted to trading on a regulated market and amending Directive 2001/34/EC and the rules and regulations promulgated pursuant thereto, as amended by various directives including 2013/50/EU TTW tank-to-wheel UCHealth University of Colorado Health UCL Université Catholique de Louvain UK the United Kingdom UNGPs UN Guiding Principles on Business and Human Rights U.S. the United States of America USPTO the United States Patent and Trademark Office U.S. Tax Treaty Convention between the Netherlands and the U.S. for the avoidance of Double Taxation and the Prevention of Fiscal Evasion with respect to Taxes and Income, dated December 18, 1992 as amended by the protocol of March 8, 2004 UT Agreement an exclusive in-license with the Board of Regents of the University of Texas System UT BoR Board of Regents of the University of Texas System VBA Value Based Agreements V-regions antibody variable regions VYVDURA VYVDURA® VYVGART VYVGART® (efgartigimod alfa-fcab) VYVGART HYTRULO VYVGART HYTRULO™ VYVGART SC VYVGART subcutaneous (efgartigimod alfa + hyaluronidase qvfc) WBCSD World Business Council for Sustainable Development we, us or our argenx SE together with its wholly-owned subsidiaries and, as applicable, its former wholly-owned subsidiaries WFH Work from Home WTT well-to-tank WTW well-to-wheel Zai Lab Zai Lab Ltd Zai Lab Agreement collaboration agreement with Zai Lab Ltd, relating to an exclusive out-license for the development and commercialization of efgartigimod in Greater China Term Definition argenx Group Risk Factors Corporate Governance Share Capital Financial Review Financial Statements Non-Financial Information argenx Annual Report 2025 Definitions 334
Contact us via argenx.com/contact-us You can find the annual report 2025 online at reports.argenx.com/2025