

Transaction and Company Overview March 2, 2026 .2

Disclaimer This presentation and the accompanying slides and oral commentary (this “Presentation”), which have been prepared by Candid Therapeutics, Inc. (the “Company”), are for informational purposes only, and shall not form the basis for or be relied on in connection with any investment decision with respect to the Company, Rallybio Corporation (“Rallybio”) or the combined company. This Presentation has been prepared by the Company based on information and data which the Company considers reliable, but no reliance shall be placed on, and no representation or warranty, express or implied, whatsoever is or will be given by the Company or any of its affiliates, directors, officers, employees or advisers or any other person as to the truth, accuracy, completeness, fairness and reasonableness of the contents of this Presentation. This Presentation may not be all inclusive and does not purport to contain all of the information that may be required to evaluate a possible investment decision with respect to the Company. The recipient agrees and acknowledges that (i) this Presentation is not intended to form the basis of any investment decision by the recipient and does not constitute investment, tax or legal advice, and (ii) the information contained in this Presentation is subject to change, and any such changes may be material. Certain matters discussed in this Presentation may contain forward-looking statements that are, by their nature, subject to significant risks and uncertainties. Forward-looking statements can be identified by words such as “may,” “will,” “should,” “would,” “could,” “believe,” “expect,” “anticipate,” “intend,” “plan,” “continue,” “seek,” “estimate,” “potential” or the negative of these terms or other similar terms. Forward-looking statements in this Presentation include, but are not limited to, statements about: expectations with respect to the proposed merger with Rallybio (the “Merger”) and the proposed concurrent financing, the structure and timing thereof, the use of proceeds therefrom, the ability of the parties to consummate the transactions and the expected post-closing ownership of the combined company; the combined company's listing in Nasdaq after the closing of the proposed Merger; the expected management team of the combined company; the combined company's expected cash runway; the potential of Rallybio stockholders to receive consideration pursuant to the Contingent Value Rights ("CVRs"); the Company’s product candidates and the potential benefits thereof and potential new indications; the Company’s expectations with regard to the design and results of its research and development programs, preclinical studies, and clinical trials, including the timing and availability of data from such studies and trials; the potential for the Company’s portfolio to deliver clinical milestones across multiple programs with first or best in class potential; the potential market size and size of the potential patient populations for the Company’s product candidates and any future product candidates; and the Company’s business strategy. Such forward-looking statements reflect the current views of the Company’s management regarding future events; they are not guarantees of future performance. These forward-looking statements are subject to a number of risks and uncertainties, many of which involve factors or circumstances that are beyond Rallybio’s and the Company’s control. The Company’s and the combined company’s actual results could differ materially from those stated or implied in forward-looking statements due to a number of factors, including but not limited to (i) the risk that the conditions to closing of the proposed Merger are not satisfied, including the failure to timely obtain stockholder approval for the merger agreement and the transactions contemplated thereby, if at all; (ii) uncertainties as to the timing of the consummation of the proposed Merger and the ability of each of Rallybio and the Company to consummate the proposed Merger; (iii) risks related to Rallybio’s ability to manage its operating expenses and its expenses associated with the proposed Merger pending closing; (iv) risks related to the failure or delay in obtaining required approvals from any governmental or regulatory entity necessary to consummate the proposed Merger; (v) the risk that as a result of adjustments to the exchange ratio, Rallybio’s stockholders and the Company’s stockholders could own more or less of the combined company than is currently anticipated; (vi) risks related to the market price of Rallybio’s common stock relative to the value suggested by the exchange ratio; (vii) unexpected costs, charges or expenses resulting from the proposed transactions; (viii) potential adverse reactions or changes to business relationships resulting from the announcement or completion of the proposed Merger; (ix) the uncertainties associated with the Company’s product candidates and platform technologies, as well as risks associated with the clinical development and regulatory approval of product candidates, including potential delays in the commencement, enrollment and completion of clinical trials; (x) risks related to the inability of the combined company to obtain sufficient additional capital to continue to advance these product candidates and its preclinical programs; (xi) uncertainties in obtaining successful clinical results for product candidates and unexpected costs that may result therefrom; (xii) risks related to the failure to realize any value from product candidates and preclinical programs being developed and anticipated to be developed in light of inherent risks and difficulties involved in successfully bringing product candidates to market; (xiii) risks associated with the possible failure to realize certain anticipated benefits of the proposed Merger, including with respect to future financial and operating results; (xiv) the risk that the concurrent financing is not consummated; (xv) the potential for the occurrence of any event, change or other circumstance or condition that could give rise to the termination of the merger agreement and any agreements entered into in connection therewith; and (xvi) the possibility that holders of CVRs may never receive any proceeds therefrom. Actual results and the timing of events could differ materially from those anticipated in such forward-looking statements as a result of these risks and uncertainties. These and other risks and uncertainties are more fully described in periodic filings with the SEC, including the factors described in the section titled “Risk Factors” in Rallybio’s Annual Report on Form 10-K for the year ended December 31, 2024 and Quarterly Report on Form 10-Q for the quarter ended September 30, 2025, each filed with the Securities and Exchange Commission (the “SEC”), and in other filings that Rallybio makes and will make with the SEC in connection with the proposed transactions, including the Proxy Statement described below under “Additional Information and Where to Find It.” You should not place undue reliance on these forward-looking statements, which are made only as of the date hereof or as of the dates indicated in the forward-looking statements. Each of the Company and Rallybio expressly disclaim any obligation or undertaking to release publicly any updates or revisions to any forward-looking statements contained herein to reflect any change in their expectations with regard thereto or any change in events, conditions or circumstances on which any such statements are based. This Presentation does not purport to summarize all of the conditions, risks and other attributes of an investment in Rallybio or the Company.

Disclaimer (cont.) This Presentation may contain trademarks, service marks, trade names and copyrights of other companies, which are the property of their respective owners. Solely for convenience, some of the trademarks, service marks, trade names and copyrights referred to in this Presentation may be listed without the TM, SM or © or ® symbols, but the Company will assert, to the fullest extent under applicable law, the rights of the owners to these trademarks, service marks, trade names and copyrights. Participants in the Solicitation This Presentation relates to the proposed transactions involving Rallybio and the Company and may be deemed to be solicitation material in respect of the proposed transactions. In connection with the proposed transactions, Rallybio will file relevant materials with the SEC, including a registration statement on Form S-4 (the “Form S-4”) that will contain a proxy statement (the “Proxy Statement”) and prospectus. This Presentation is not a substitute for the Form S-4, the Proxy Statement or for any other document that Rallybio may file with the SEC and/or send to Rallybio’s stockholders in connection with the proposed transactions. Rallybio, the Company, and their respective directors and certain of their executive officers may be considered participants in the solicitation of proxies from Rallybio’s stockholders with respect to the proposed transactions under the rules of the SEC. Information about the directors and executive officers of Rallybio is set forth in its proxy statement, which was filed with the SEC on April 7, 2025, and in subsequent documents filed with the SEC. Additional information regarding the persons who may be deemed participants in the proxy solicitations and a description of their direct and indirect interests, by security holdings or otherwise, will also be included in the Form S-4, the Proxy Statement and other relevant materials to be filed with the SEC when they become available. You may obtain free copies of this document as described below. BEFORE MAKING ANY VOTING DECISION, INVESTORS AND SECURITY HOLDERS OF RALLYBIO ARE URGED TO READ THE FORM S-4, THE PROXY STATEMENT AND OTHER DOCUMENTS FILED WITH THE SEC CAREFULLY AND IN THEIR ENTIRETY WHEN THEY BECOME AVAILABLE BECAUSE THEY WILL CONTAIN IMPORTANT INFORMATION ABOUT RALLYBIO, THE PROPOSED TRANSACTIONS AND RELATED MATTERS. No Offer or Solicitation This Presentation does not constitute an offer to sell or the solicitation of an offer to buy any securities nor a solicitation of any vote or approval with respect to the proposed transactions herein or otherwise. No offering of securities shall be made except by means of a prospectus meeting the requirements of Section 10 of the U. S. Securities Act of 1933, as amended, and otherwise in accordance with applicable law. Additional Information and Where to Find It Investors and security holders will be able to obtain free copies of the Form S-4, the Proxy Statement and other documents filed by Rallybio with the SEC through the website maintained by the SEC at http://www.sec.gov. Copies of the documents filed by Rallybio with the SEC will also be available free of charge on Rallybio’s website at investors.rallybio.com, or by contacting Rallybio’s Investor Relations at investors@rallybio.com.

The proceeds from the private financing are expected to be primarily used to advance Candid’s pipeline programs through various milestones Transaction Highlights Rallybio to acquire 100% of Candid equity interests in reverse-triangular merger with Candid surviving the merger as a wholly owned subsidiary of Rallybio Post-closing, Rallybio will be renamed Candid Therapeutics, Inc. and trade on Nasdaq under the ticker symbol “CDRX” In connection with the Merger, Candid entered into subscription agreements for a concurrent oversubscribed and upsized private financing of over $505 million in gross proceeds Syndicate includes leading healthcare institutional investors and mutual funds, including Venrock Healthcare Capital Partners, RA Capital Management, Janus Henderson Investors, accounts advised by T. Rowe Price Associates, Inc., venBio Partners, Viking Global Investors, Cormorant Asset Management, Foresite Capital, Soleus Capital, TCGX, Vivo Capital, a life sciences focused institutional investor, several additional mutual funds and other institutional investors Structure Financing Primary Use of Proceeds Candid management team, led by Dr. Ken Song, will continue to lead the combined company post closing of transaction Management Team
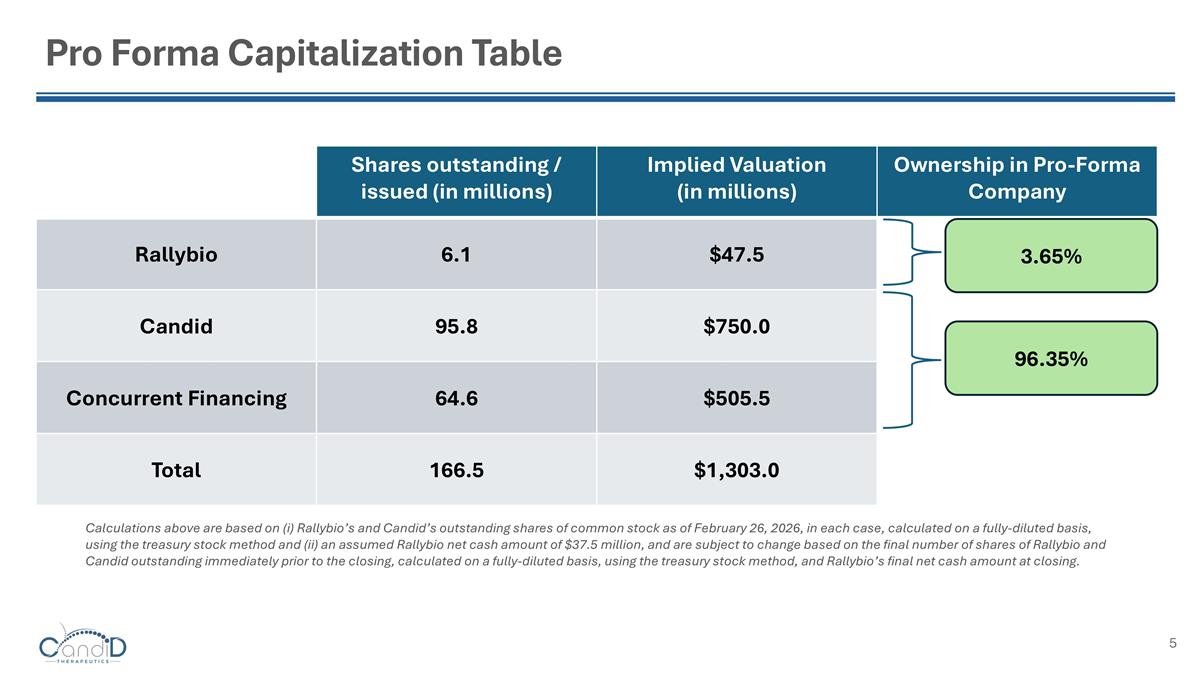
Pro Forma Capitalization Table Calculations above are based on (i) Rallybio’s and Candid’s outstanding shares of common stock as of February 26, 2026, in each case, calculated on a fully-diluted basis, using the treasury stock method and (ii) an assumed Rallybio net cash amount of $37.5 million, and are subject to change based on the final number of shares of Rallybio and Candid outstanding immediately prior to the closing, calculated on a fully-diluted basis, using the treasury stock method, and Rallybio’s final net cash amount at closing. Shares outstanding / issued (in millions) Implied Valuation (in millions) Ownership in Pro-Forma Company Rallybio 6.1 $47.5 Candid 95.8 $750.0 Concurrent Financing 64.6 $505.5 Total 166.5 $1,303.0 3.65% 96.35%

Entrepreneurial team with proven development track record and recent exits Wholly-owned China entity for early clinical development Candid Therapeutics – TCEs for Autoimmune Diseases (AIDs) Broad pipeline with deep clinical experience in TCEs B-cell and plasma cell depletion has demonstrated substantial clinical efficacy in multiple AIDs, opening up a large market opportunity that can potentially parallel success of anti-tumor necrosis alpha therapies T-cell engager (TCE) antibodies are well positioned to be a primary modality for B-cell depletion BCMA TCE (cizutamig) – Potential first and best-in-class for AID entering global Phase 2 studies 87 patients dosed including 47 patients with AID* with low rates of cytokine release syndrome (CRS) Initial deeper therapeutic activity with less dosing frequency than anti-FcRn drug class CD19 and CD20 TCEs (CND261, CND319) – Potential for best-in-class profile for AID CND261, CD20 TCE, with 100+ patients dosed in oncology and AID* with low rates of CRS CND319, CD19/CD20 TCE, with encouraging non-human primate data. First-in-human studies planned for mid ’26. BCMAxCD19 TCE (CND460) – IND enabling studies initiated with first-in-human studies 1H ‘27 Ongoing clinical studies in over 10 indications with global Phase 2 studies launching in mid ‘26 for myasthenia gravis and interstitial lung disease Transformative Opportunity Differentiated Portfolio Approach (5 TCE programs) Strong Foundation *As of February 2, 2026
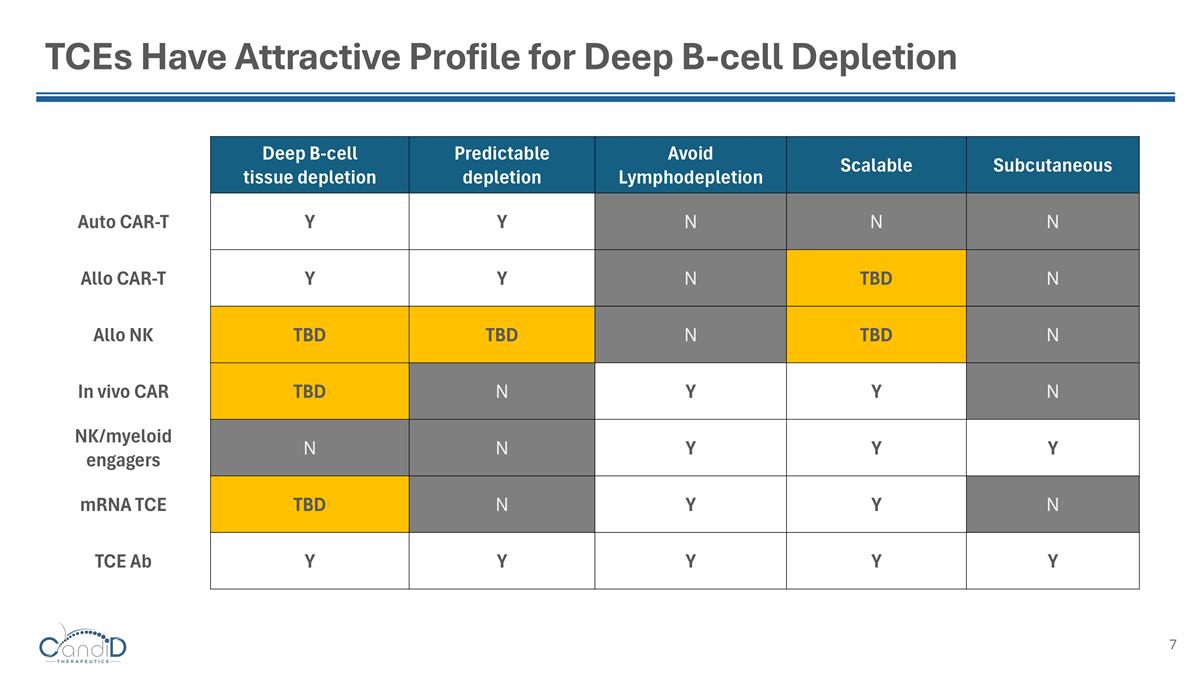
TCEs Have Attractive Profile for Deep B-cell Depletion Deep B-cell tissue depletion Predictable depletion Avoid Lymphodepletion Scalable Subcutaneous Auto CAR-T Y Y N N N Allo CAR-T Y Y N TBD N Allo NK TBD TBD N TBD N In vivo CAR TBD N Y Y N NK/myeloid engagers N N Y Y Y mRNA TCE TBD N Y Y N TCE Ab Y Y Y Y Y
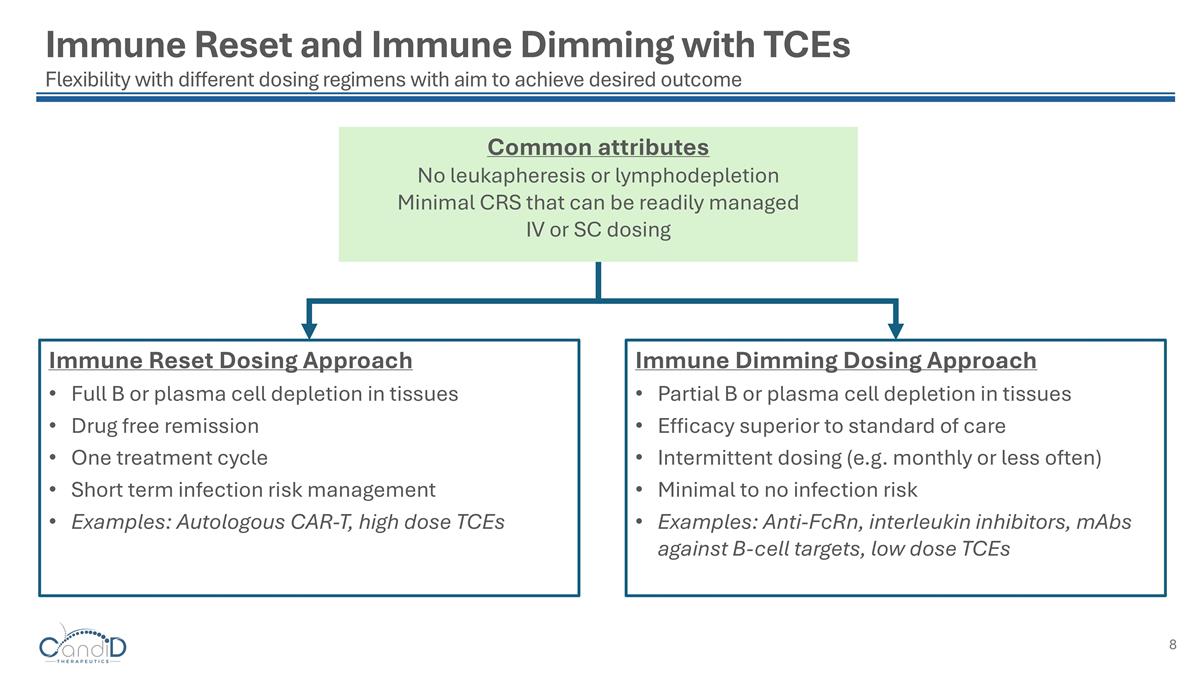
Immune Reset and Immune Dimming with TCEs Flexibility with different dosing regimens with aim to achieve desired outcome Immune Reset Dosing Approach Full B or plasma cell depletion in tissues Drug free remission One treatment cycle Short term infection risk management Examples: Autologous CAR-T, high dose TCEs Immune Dimming Dosing Approach Partial B or plasma cell depletion in tissues Efficacy superior to standard of care Intermittent dosing (e.g. monthly or less often) Minimal to no infection risk Examples: Anti-FcRn, interleukin inhibitors, mAbs against B-cell targets, low dose TCEs Common attributes No leukapheresis or lymphodepletion Minimal CRS that can be readily managed IV or SC dosing
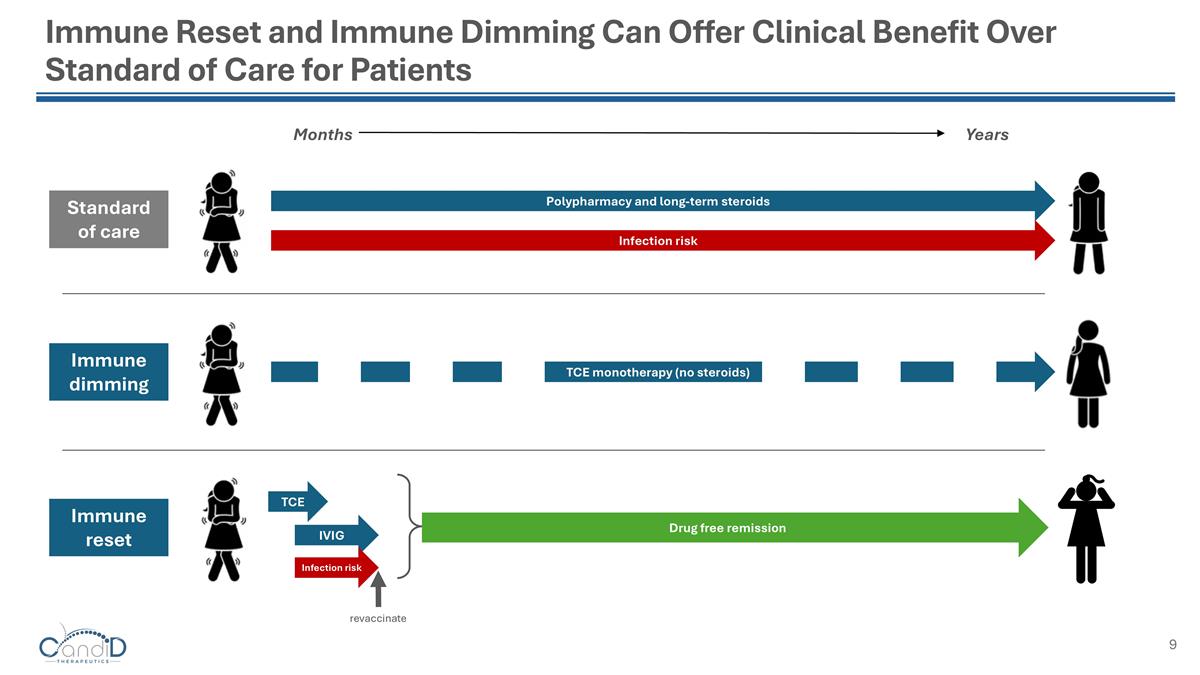
Immune Reset and Immune Dimming Can Offer Clinical Benefit Over Standard of Care for Patients Immune dimming TCE monotherapy (no steroids) Months Years Polypharmacy and long-term steroids Infection risk Standard of care Immune reset Drug free remission revaccinate TCE IVIG Infection risk
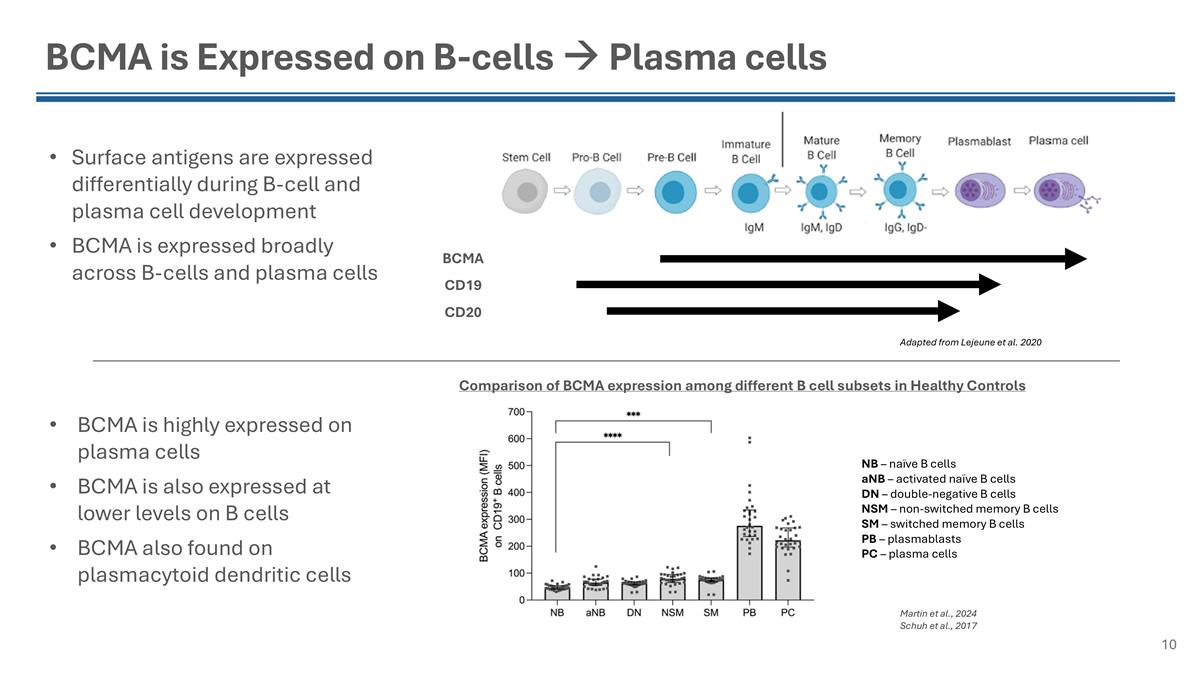
BCMA is Expressed on B-cells à Plasma cells Surface antigens are expressed differentially during B-cell and plasma cell development BCMA is expressed broadly across B-cells and plasma cells BCMA is highly expressed on plasma cells BCMA is also expressed at lower levels on B cells BCMA also found on plasmacytoid dendritic cells Comparison of BCMA expression among different B cell subsets in Healthy Controls NB – naïve B cells aNB – activated naïve B cells DN – double-negative B cells NSM – non-switched memory B cells SM – switched memory B cells PB – plasmablasts PC – plasma cells Martin et al., 2024 Schuh et al., 2017 Adapted from Lejeune et al. 2020 BCMA CD19 CD20
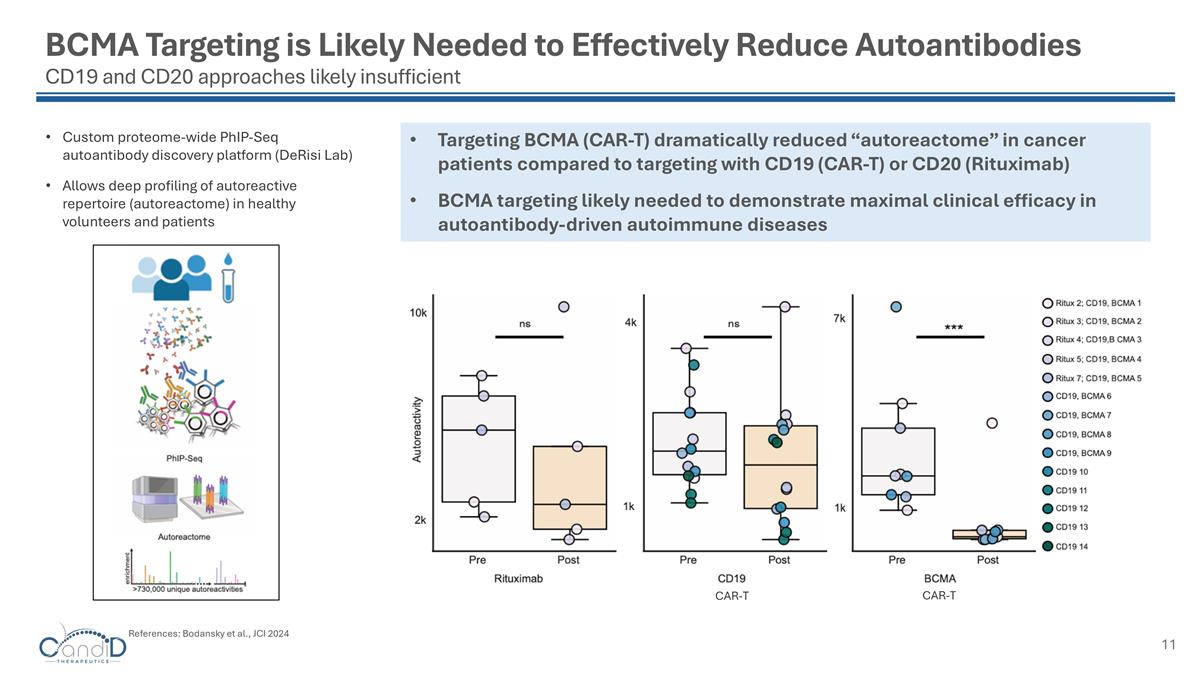
BCMA Targeting is Likely Needed to Effectively Reduce Autoantibodies CD19 and CD20 approaches likely insufficient Targeting BCMA (CAR-T) dramatically reduced “autoreactome” in cancer patients compared to targeting with CD19 (CAR-T) or CD20 (Rituximab) BCMA targeting likely needed to demonstrate maximal clinical efficacy in autoantibody-driven autoimmune diseases Custom proteome-wide PhIP-Seq autoantibody discovery platform (DeRisi Lab) Allows deep profiling of autoreactive repertoire (autoreactome) in healthy volunteers and patients References: Bodansky et al., JCI 2024 CAR-T CAR-T
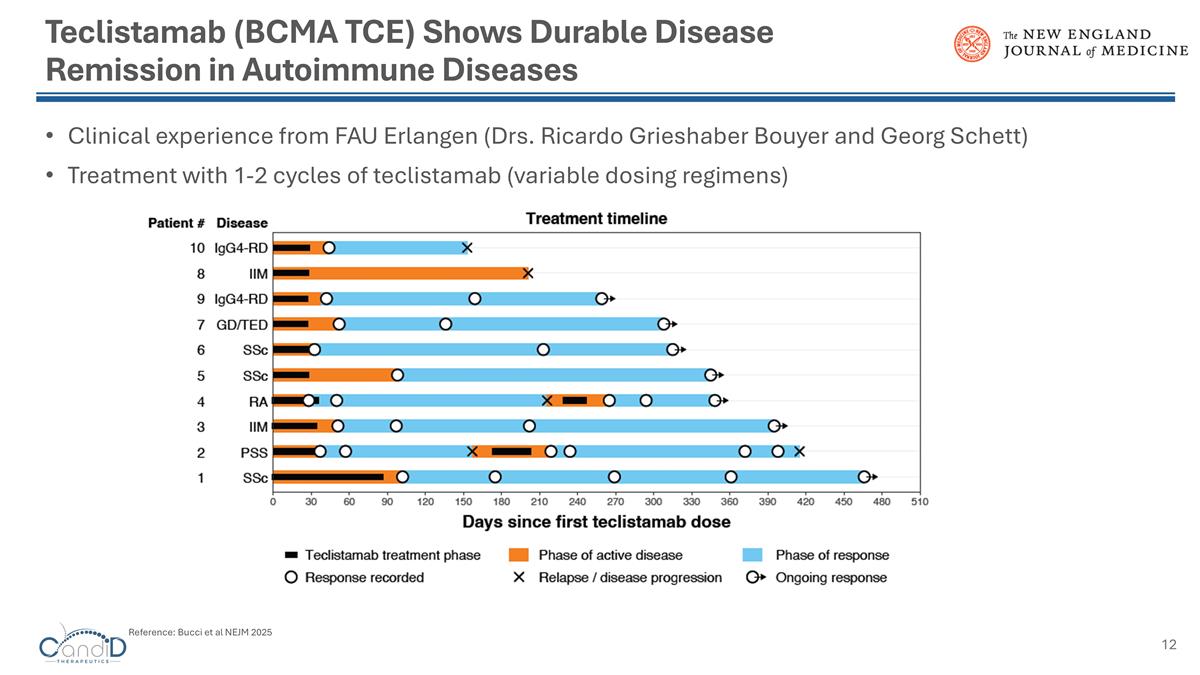
Teclistamab (BCMA TCE) Shows Durable Disease Remission in Autoimmune Diseases Clinical experience from FAU Erlangen (Drs. Ricardo Grieshaber Bouyer and Georg Schett) Treatment with 1-2 cycles of teclistamab (variable dosing regimens) Reference: Bucci et al NEJM 2025
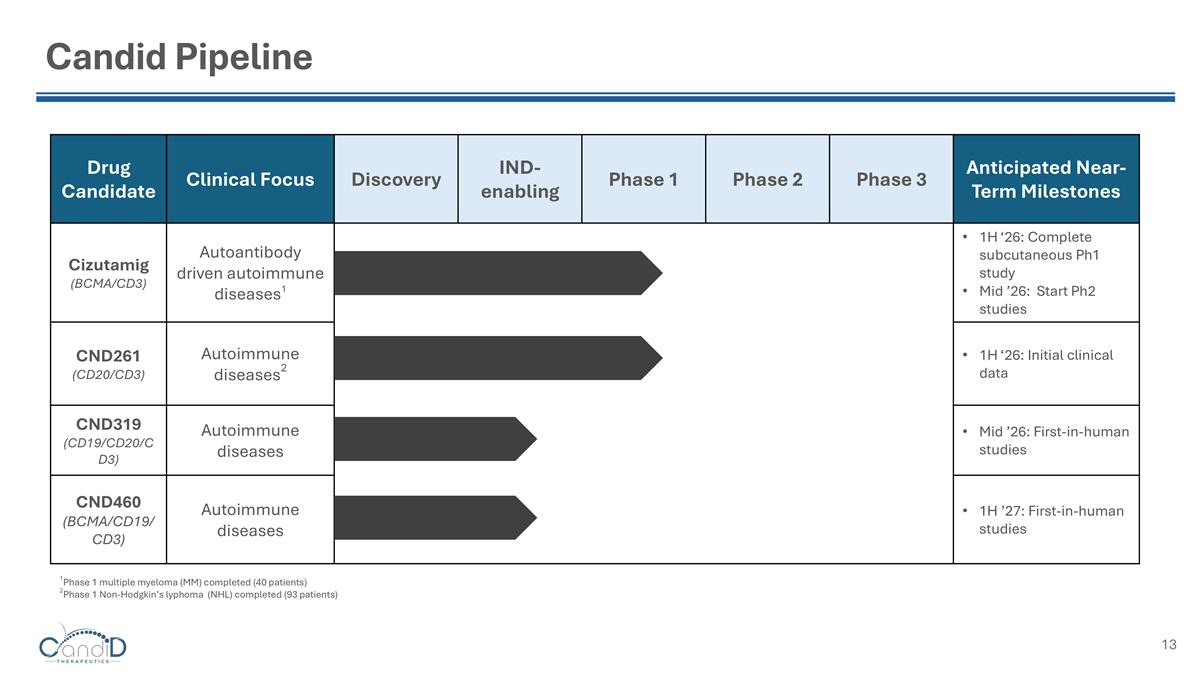
Candid Pipeline Drug Candidate Clinical Focus Discovery IND-enabling Phase 1 Phase 2 Phase 3 Anticipated Near-Term Milestones Cizutamig (BCMA/CD3) Autoantibody driven autoimmune diseases1 1H ‘26: Complete subcutaneous Ph1 study Mid ’26: Start Ph2 studies CND261 (CD20/CD3) Autoimmune diseases2 1H ‘26: Initial clinical data CND319 (CD19/CD20/CD3) Autoimmune diseases Mid ’26: First-in-human studies CND460 (BCMA/CD19/CD3) Autoimmune diseases 1H ’27: First-in-human studies 1Phase 1 multiple myeloma (MM) completed (40 patients) 2Phase 1 Non-Hodgkin’s lyphoma (NHL) completed (93 patients)
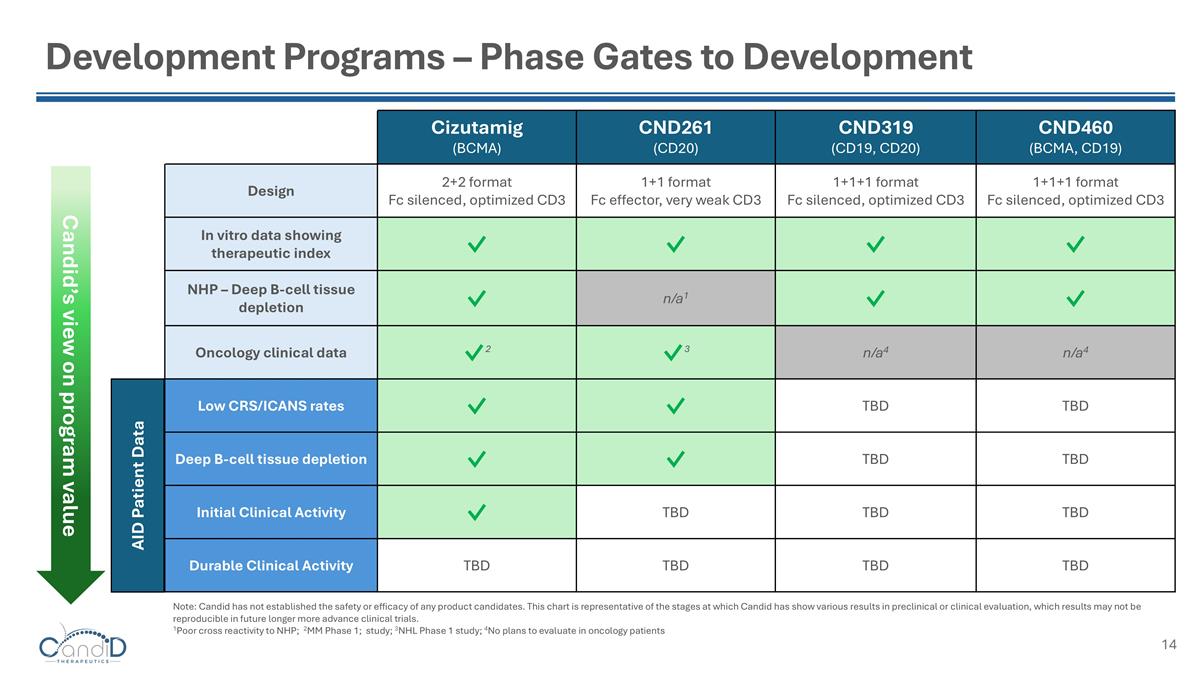
Development Programs – Phase Gates to Development Cizutamig (BCMA) CND261 (CD20) CND319 (CD19, CD20) CND460 (BCMA, CD19) Design 2+2 format Fc silenced, optimized CD3 1+1 format Fc effector, very weak CD3 1+1+1 format Fc silenced, optimized CD3 1+1+1 format Fc silenced, optimized CD3 In vitro data showing therapeutic index ✓ ✓ ✓ ✓ NHP – Deep B-cell tissue depletion ✓ n/a1 ✓ ✓ Oncology clinical data ✓2 ✓3 n/a4 n/a4 Low CRS/ICANS rates ✓ ✓ TBD TBD Deep B-cell tissue depletion ✓ ✓ TBD TBD Initial Clinical Activity ✓ TBD TBD TBD Durable Clinical Activity TBD TBD TBD TBD AID Patient Data Candid’s view on program value Note: Candid has not established the safety or efficacy of any product candidates. This chart is representative of the stages at which Candid has show various results in preclinical or clinical evaluation, which results may not be reproducible in future longer more advance clinical trials. 1Poor cross reactivity to NHP; 2MM Phase 1; study; 3NHL Phase 1 study; 4No plans to evaluate in oncology patients
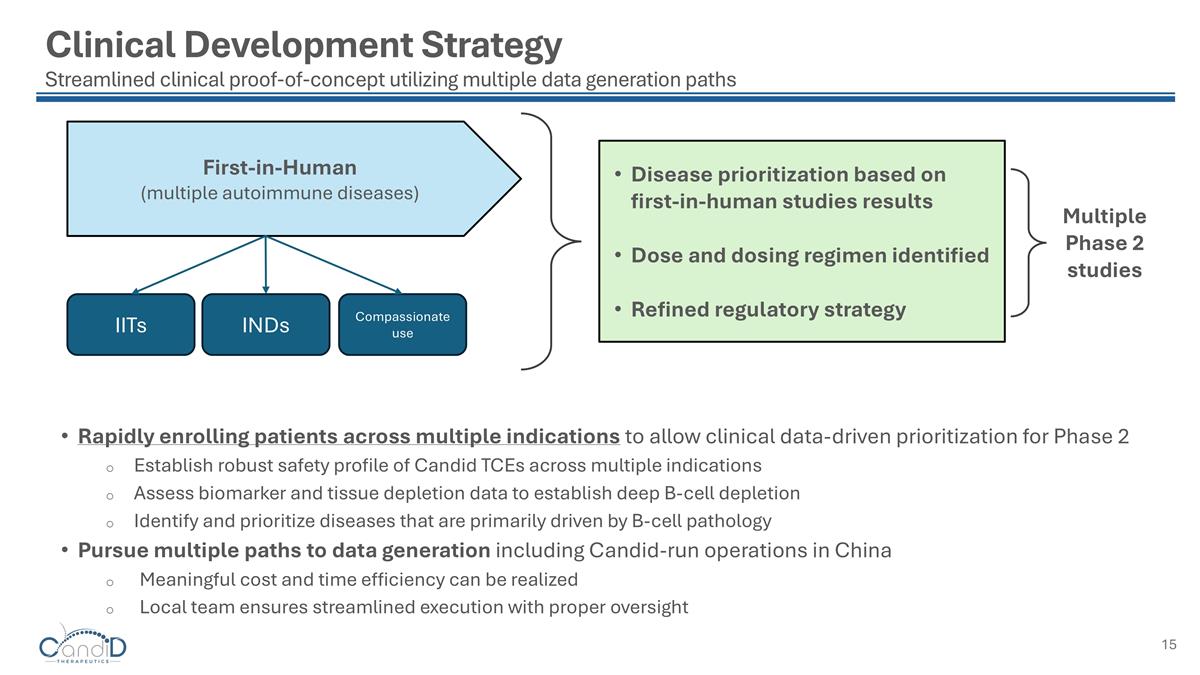
First-in-Human (multiple autoimmune diseases) Rapidly enrolling patients across multiple indications to allow clinical data-driven prioritization for Phase 2 Establish robust safety profile of Candid TCEs across multiple indications Assess biomarker and tissue depletion data to establish deep B-cell depletion Identify and prioritize diseases that are primarily driven by B-cell pathology Pursue multiple paths to data generation including Candid-run operations in China Meaningful cost and time efficiency can be realized Local team ensures streamlined execution with proper oversight Clinical Development Strategy Streamlined clinical proof-of-concept utilizing multiple data generation paths Multiple Phase 2 studies Disease prioritization based on first-in-human studies results Dose and dosing regimen identified Refined regulatory strategy IITs INDs Compassionate use
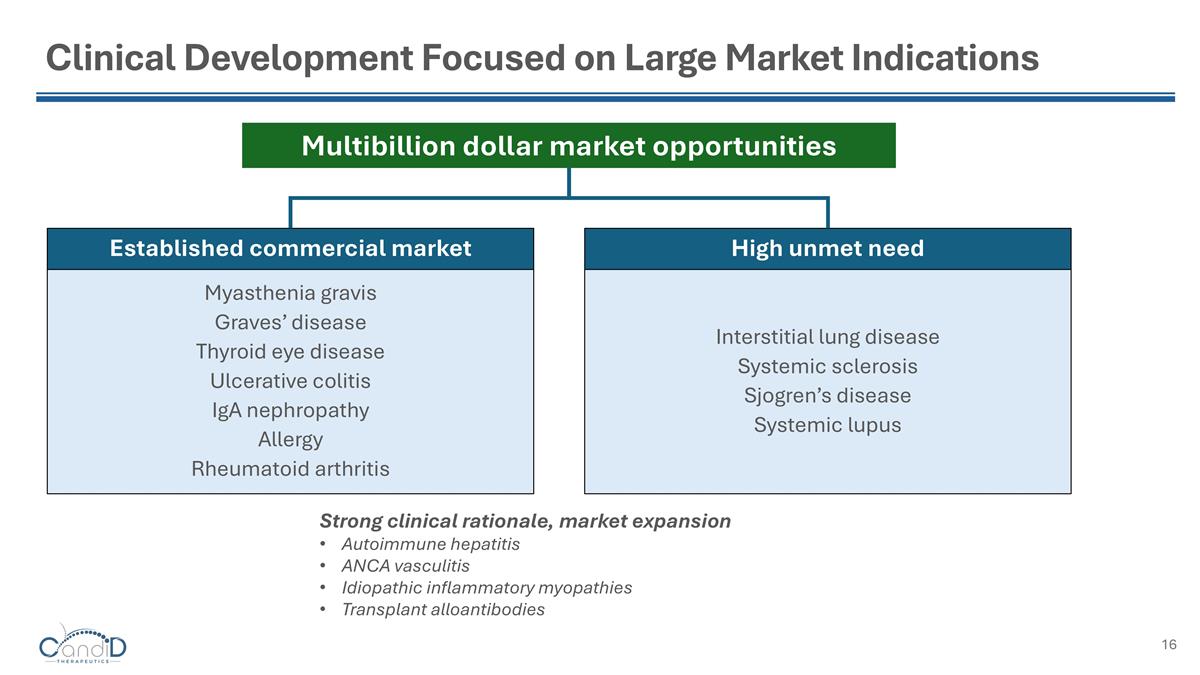
Clinical Development Focused on Large Market Indications Myasthenia gravis Graves’ disease Thyroid eye disease Ulcerative colitis IgA nephropathy Allergy Rheumatoid arthritis Interstitial lung disease Systemic sclerosis Sjogren’s disease Systemic lupus Strong clinical rationale, market expansion Autoimmune hepatitis ANCA vasculitis Idiopathic inflammatory myopathies Transplant alloantibodies Established commercial market High unmet need Multibillion dollar market opportunities

Cizutamig – BCMA/CD3 Clinical Stage Antibody Design elements High affinity binding to BCMA which is expressed on B and plasma cells Optimized CD3 binding to maximize direct cell killing and minimize release of cytokines which can cause side effects In vitro data à reduced cytokine release while maintaining B-cell cytotoxicity as compared to other BCMA TCEs Clinical data in oncology and autoimmune Ph 1 dose escalation (0.2mg to 300mg, 10 cohorts, n=40) completed in patients with multiple myeloma in Australia and China 47 patients dosed across multiple autoimmune diseases* Manufacturing – Subcutaneous formulation developed (2+2 binding) Purposefully designed to mitigate cytokine release while maintaining cytotoxicity Fc silenced CD3 optimized *As of February 2, 2026
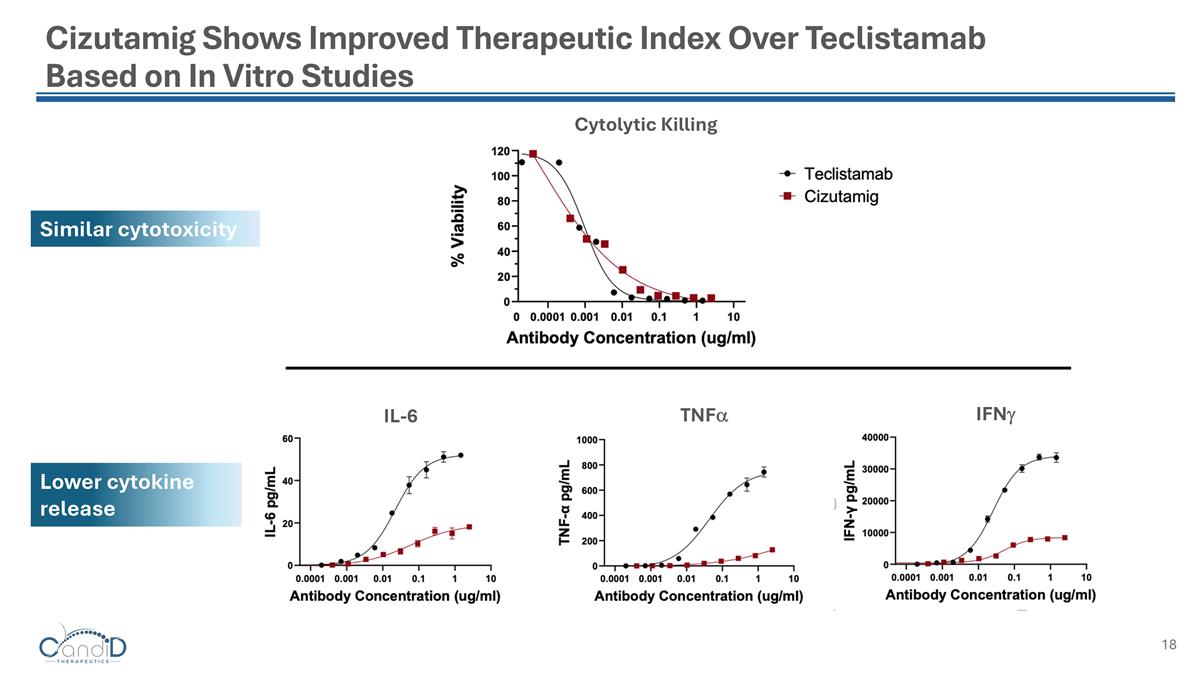
Cizutamig Shows Improved Therapeutic Index Over Teclistamab Based on In Vitro Studies IL-6 TNFa IFNg Cytolytic Killing Similar cytotoxicity Lower cytokine release
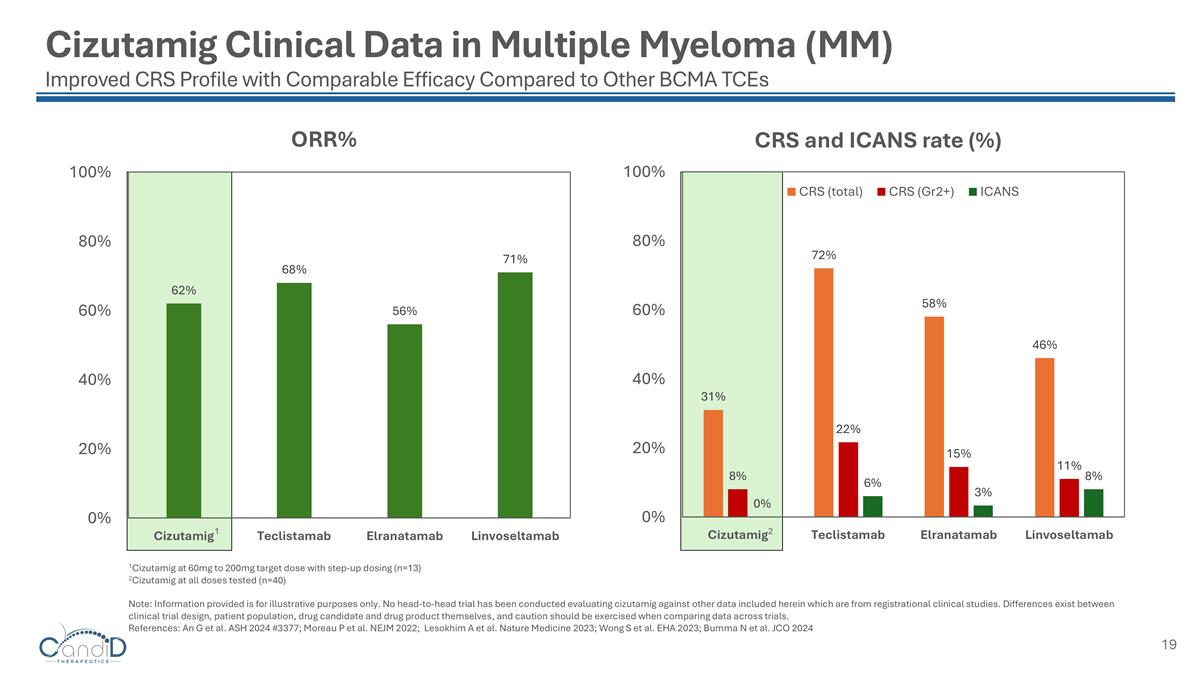
Cizutamig Clinical Data in Multiple Myeloma (MM) Improved CRS Profile with Comparable Efficacy Compared to Other BCMA TCEs 1Cizutamig at 60mg to 200mg target dose with step-up dosing (n=13) 2Cizutamig at all doses tested (n=40) Note: Information provided is for illustrative purposes only. No head-to-head trial has been conducted evaluating cizutamig against other data included herein which are from registrational clinical studies. Differences exist between clinical trial design, patient population, drug candidate and drug product themselves, and caution should be exercised when comparing data across trials. References: An G et al. ASH 2024 #3377; Moreau P et al. NEJM 2022; Lesokhim A et al. Nature Medicine 2023; Wong S et al. EHA 2023; Bumma N et al. JCO 2024 1 2
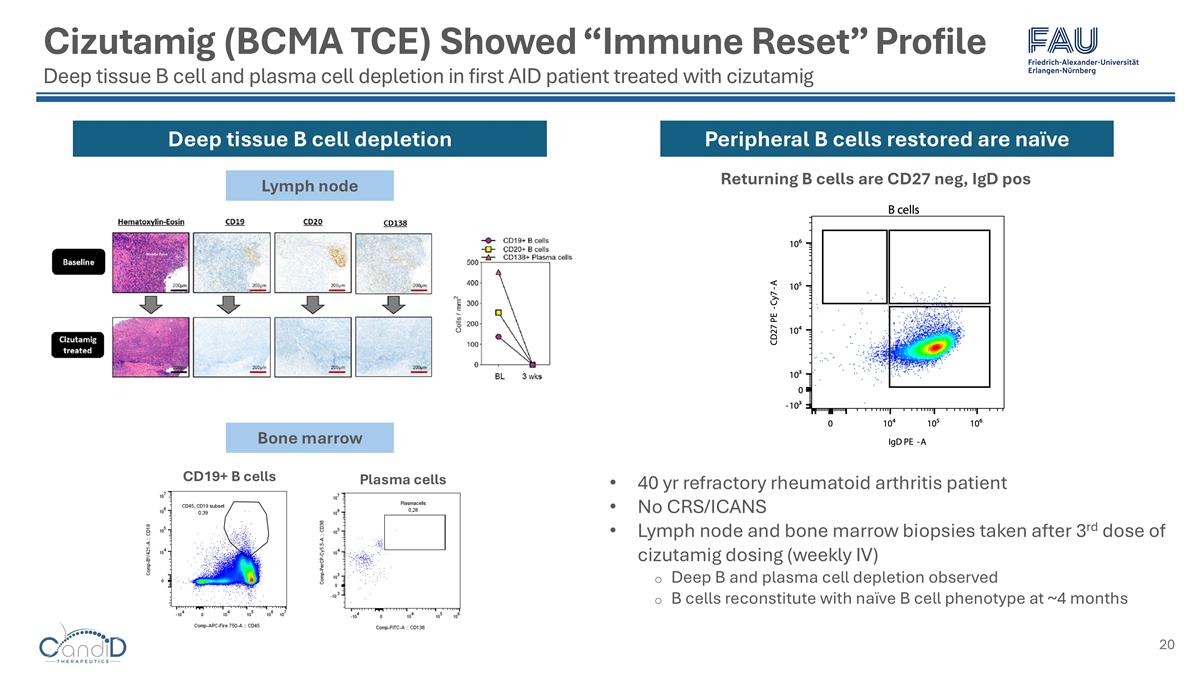
Cizutamig (BCMA TCE) Showed “Immune Reset” Profile Deep tissue B cell and plasma cell depletion in first AID patient treated with cizutamig CD19+ B cells Plasma cells Lymph node Bone marrow Deep tissue B cell depletion 40 yr refractory rheumatoid arthritis patient No CRS/ICANS Lymph node and bone marrow biopsies taken after 3rd dose of cizutamig dosing (weekly IV) Deep B and plasma cell depletion observed B cells reconstitute with naïve B cell phenotype at ~4 months Returning B cells are CD27 neg, IgD pos Peripheral B cells restored are naïve
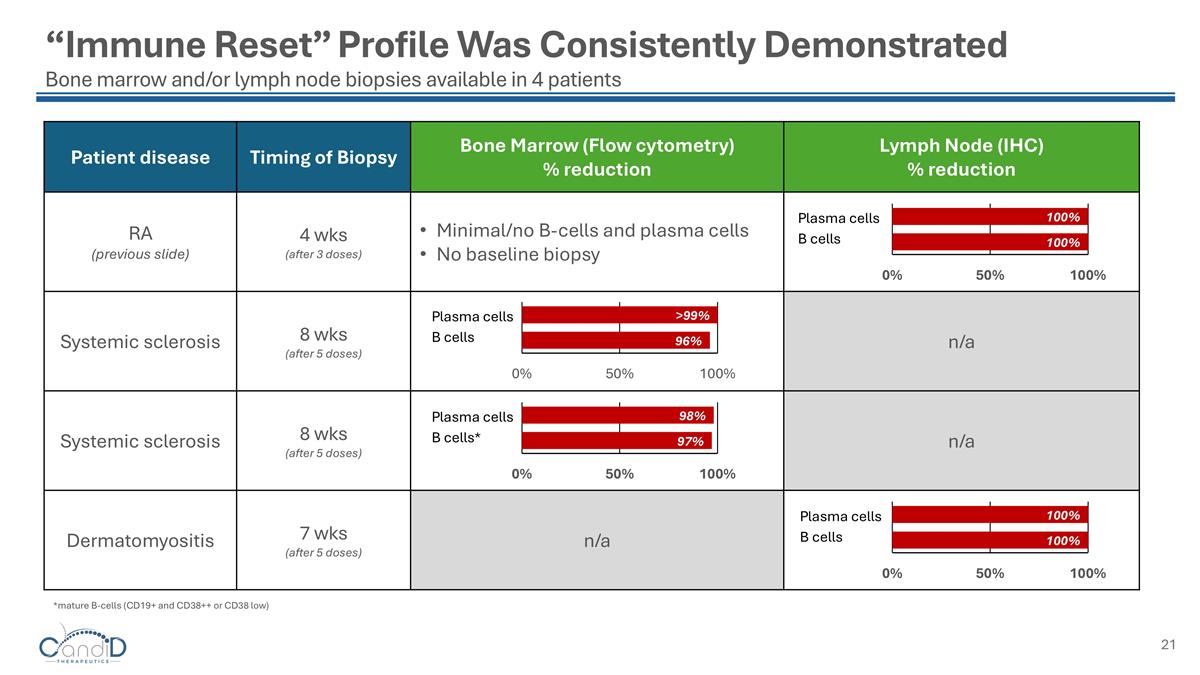
“Immune Reset” Profile Was Consistently Demonstrated Bone marrow and/or lymph node biopsies available in 4 patients Patient disease Timing of Biopsy Bone Marrow (Flow cytometry) % reduction Lymph Node (IHC) % reduction RA (previous slide) 4 wks (after 3 doses) Minimal/no B-cells and plasma cells No baseline biopsy Systemic sclerosis 8 wks (after 5 doses) n/a Systemic sclerosis 8 wks (after 5 doses) n/a Dermatomyositis 7 wks (after 5 doses) n/a Plasma cells B cells Plasma cells B cells Plasma cells B cells* Plasma cells B cells *mature B-cells (CD19+ and CD38++ or CD38 low)
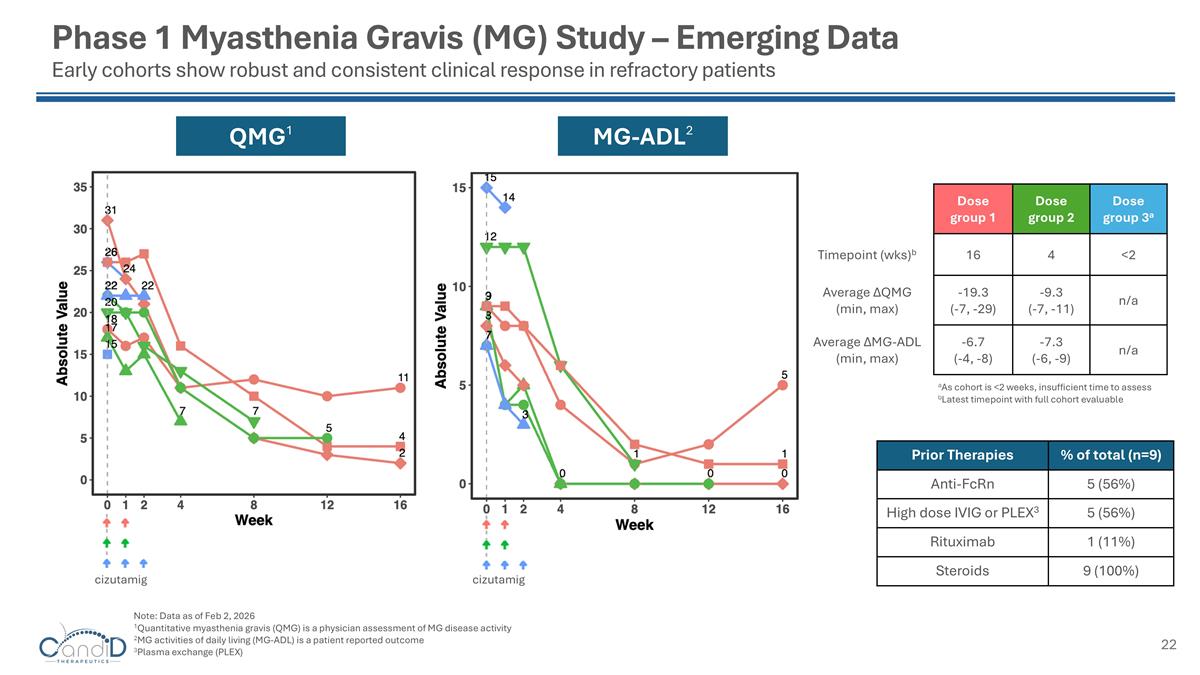
Dose group 1 Dose group 2 Dose group 3a Timepoint (wks)b 16 4 <2 Average ∆QMG (min, max) -19.3 (-7, -29) -9.3 (-7, -11) n/a Average ∆MG-ADL (min, max) -6.7 (-4, -8) -7.3 (-6, -9) n/a MG-ADL2 QMG1 cizutamig Phase 1 Myasthenia Gravis (MG) Study – Emerging Data Early cohorts show robust and consistent clinical response in refractory patients aAs cohort is <2 weeks, insufficient time to assess bLatest timepoint with full cohort evaluable Prior Therapies % of total (n=9) Anti-FcRn 5 (56%) High dose IVIG or PLEX3 5 (56%) Rituximab 1 (11%) Steroids 9 (100%) cizutamig Note: Data as of Feb 2, 2026 1Quantitative myasthenia gravis (QMG) is a physician assessment of MG disease activity 2MG activities of daily living (MG-ADL) is a patient reported outcome 3Plasma exchange (PLEX)
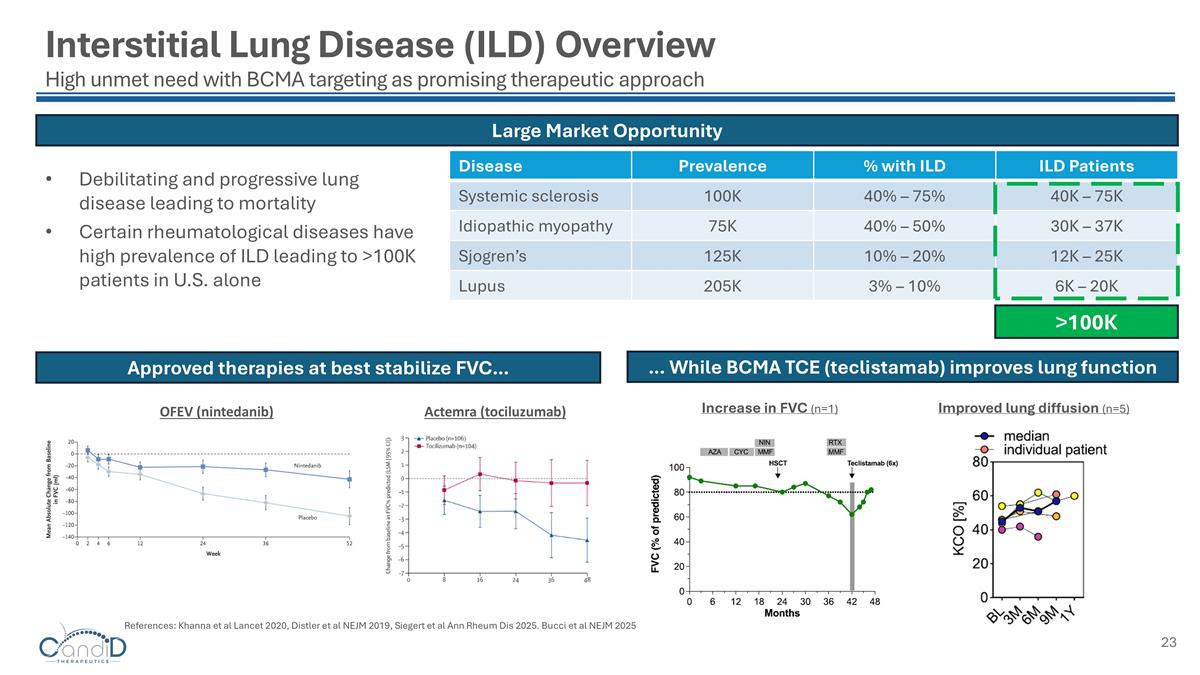
Debilitating and progressive lung disease leading to mortality Certain rheumatological diseases have high prevalence of ILD leading to >100K patients in U.S. alone Approved therapies at best stabilize FVC… Disease Prevalence % with ILD ILD Patients Systemic sclerosis 100K 40% – 75% 40K – 75K Idiopathic myopathy 75K 40% – 50% 30K – 37K Sjogren’s 125K 10% – 20% 12K – 25K Lupus 205K 3% – 10% 6K – 20K … While BCMA TCE (teclistamab) improves lung function Increase in FVC (n=1) Large Market Opportunity >100K Interstitial Lung Disease (ILD) Overview High unmet need with BCMA targeting as promising therapeutic approach References: Khanna et al Lancet 2020, Distler et al NEJM 2019, Siegert et al Ann Rheum Dis 2025. Bucci et al NEJM 2025 Improved lung diffusion (n=5) OFEV (nintedanib) Actemra (tociluzumab)
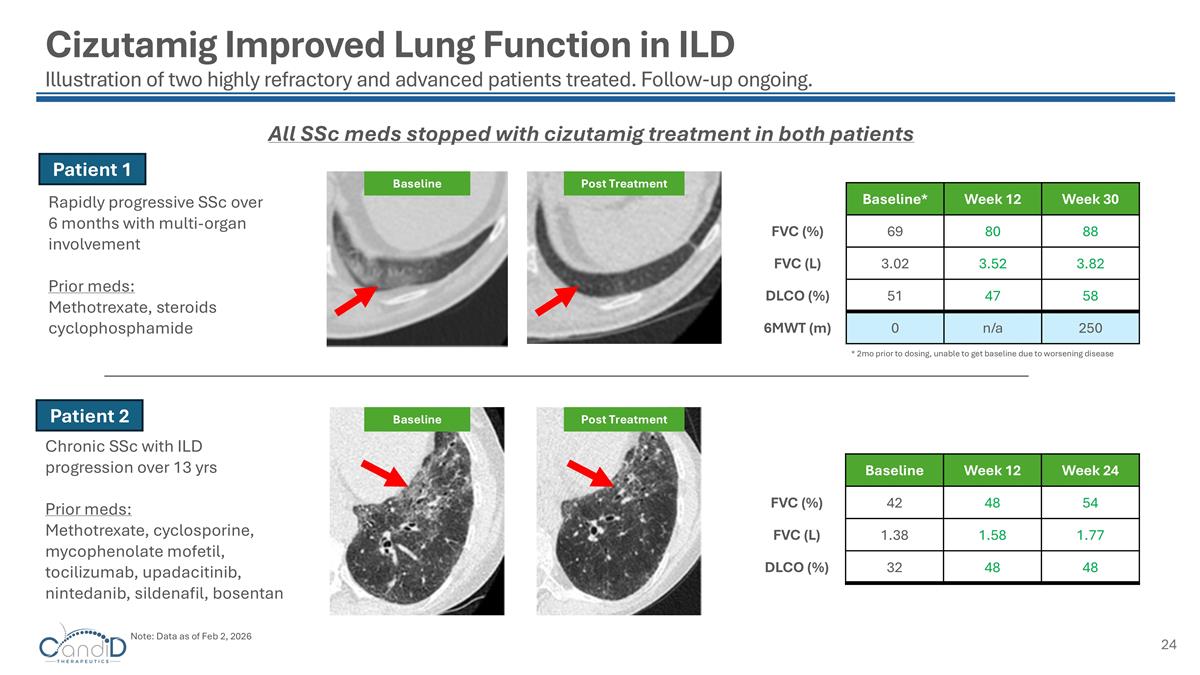
Cizutamig Improved Lung Function in ILD Illustration of two highly refractory and advanced patients treated. Follow-up ongoing. Patient 1 Patient 2 Rapidly progressive SSc over 6 months with multi-organ involvement Prior meds: Methotrexate, steroids cyclophosphamide Chronic SSc with ILD progression over 13 yrs Prior meds: Methotrexate, cyclosporine, mycophenolate mofetil, tocilizumab, upadacitinib, nintedanib, sildenafil, bosentan Baseline Post Treatment Baseline Week 12 Week 24 FVC (%) 42 48 54 FVC (L) 1.38 1.58 1.77 DLCO (%) 32 48 48 Baseline* Week 12 Week 30 FVC (%) 69 80 88 FVC (L) 3.02 3.52 3.82 DLCO (%) 51 47 58 6MWT (m) 0 n/a 250 * 2mo prior to dosing, unable to get baseline due to worsening disease Baseline Post Treatment All SSc meds stopped with cizutamig treatment in both patients Note: Data as of Feb 2, 2026
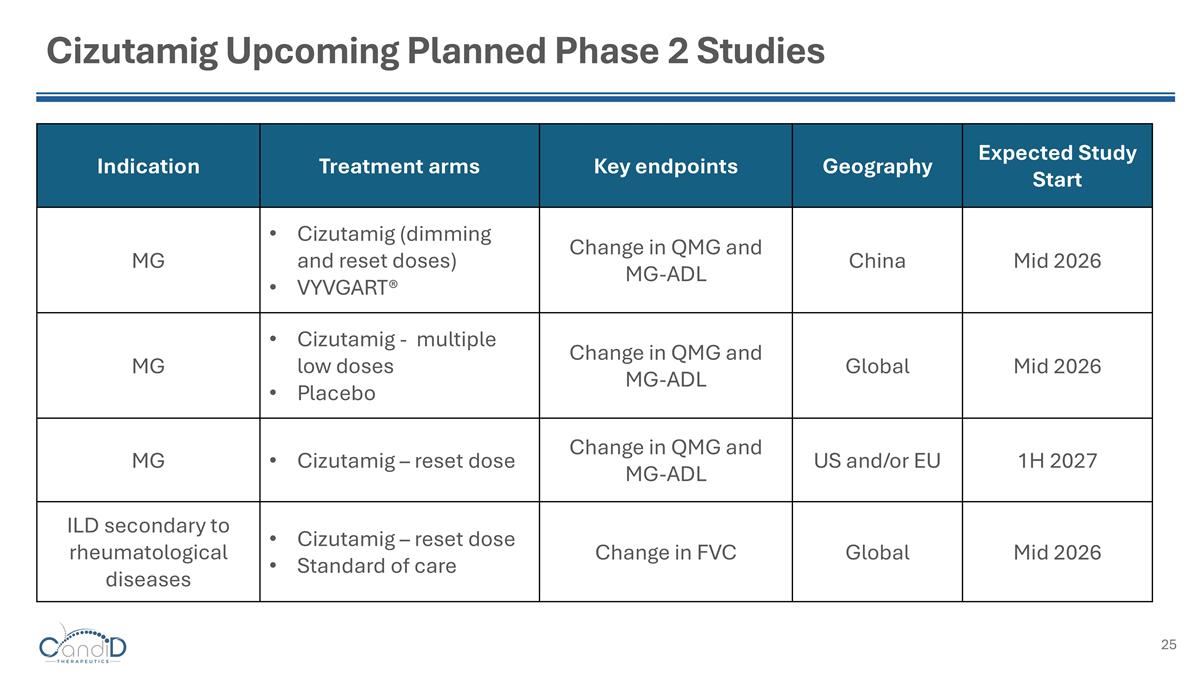
Cizutamig Upcoming Planned Phase 2 Studies Indication Treatment arms Key endpoints Geography Expected Study Start MG Cizutamig (dimming and reset doses) VYVGART® Change in QMG and MG-ADL China Mid 2026 MG Cizutamig - multiple low doses Placebo Change in QMG and MG-ADL Global Mid 2026 MG Cizutamig – reset dose Change in QMG and MG-ADL US and/or EU 1H 2027 ILD secondary to rheumatological diseases Cizutamig – reset dose Standard of care Change in FVC Global Mid 2026
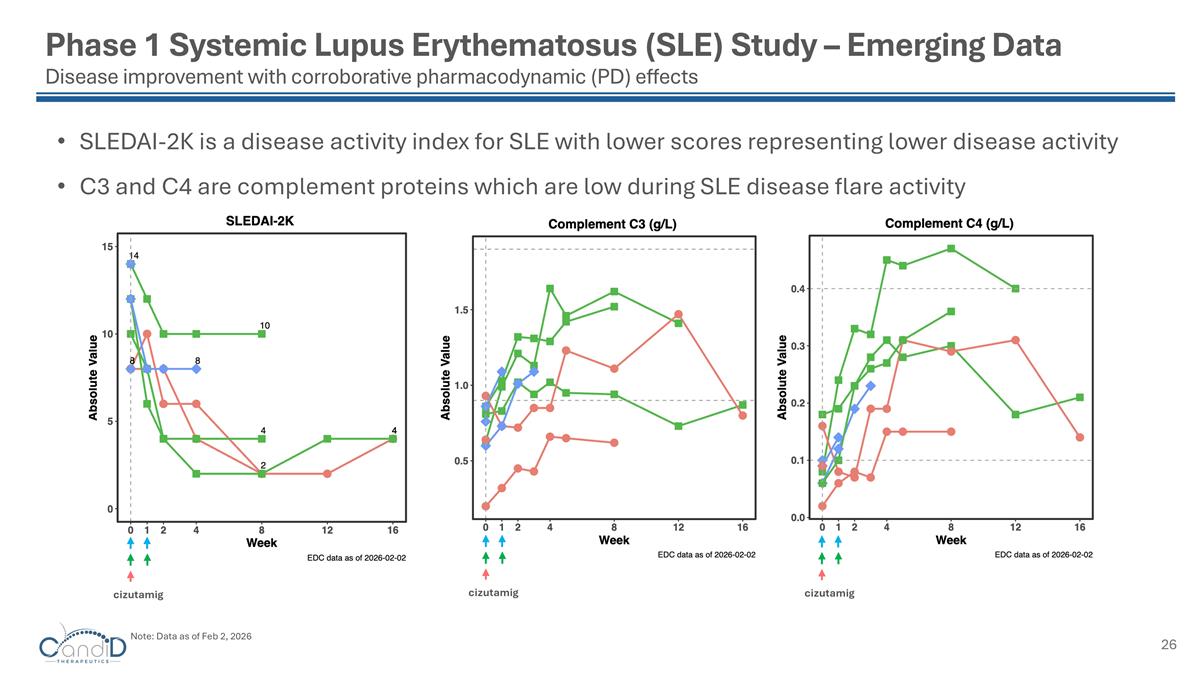
Phase 1 Systemic Lupus Erythematosus (SLE) Study – Emerging Data Disease improvement with corroborative pharmacodynamic (PD) effects SLEDAI-2K is a disease activity index for SLE with lower scores representing lower disease activity C3 and C4 are complement proteins which are low during SLE disease flare activity cizutamig cizutamig cizutamig Note: Data as of Feb 2, 2026
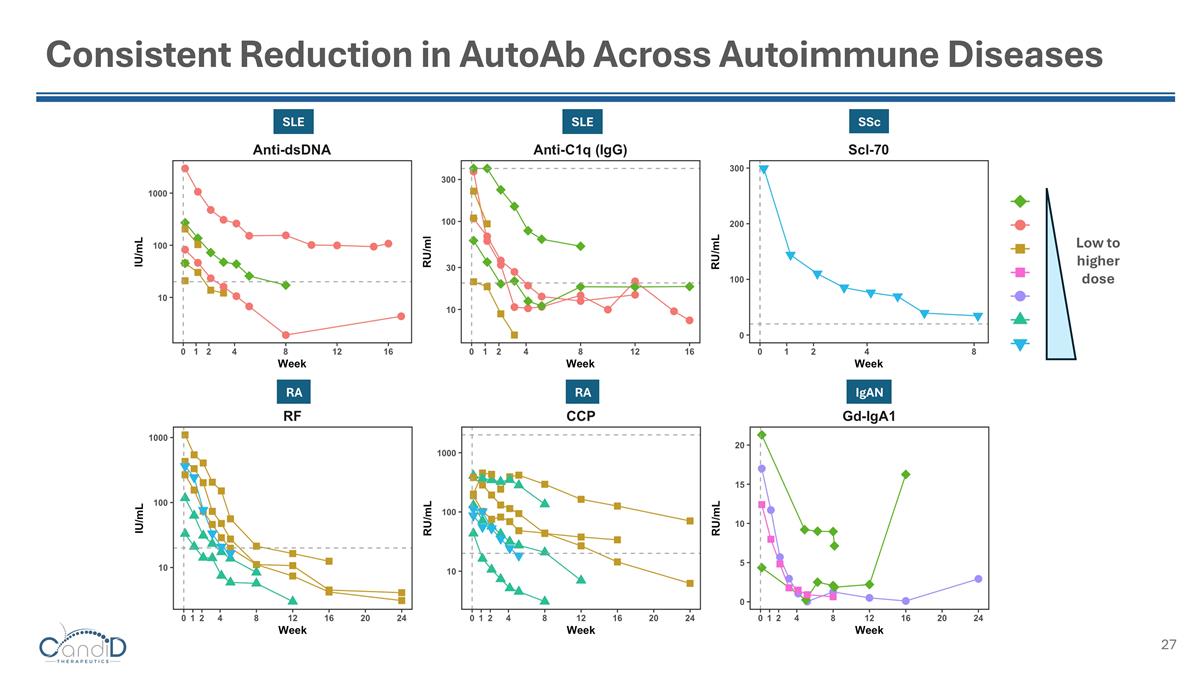
Consistent Reduction in AutoAb Across Autoimmune Diseases SLE SLE SSc RA RA IgAN Low to higher dose
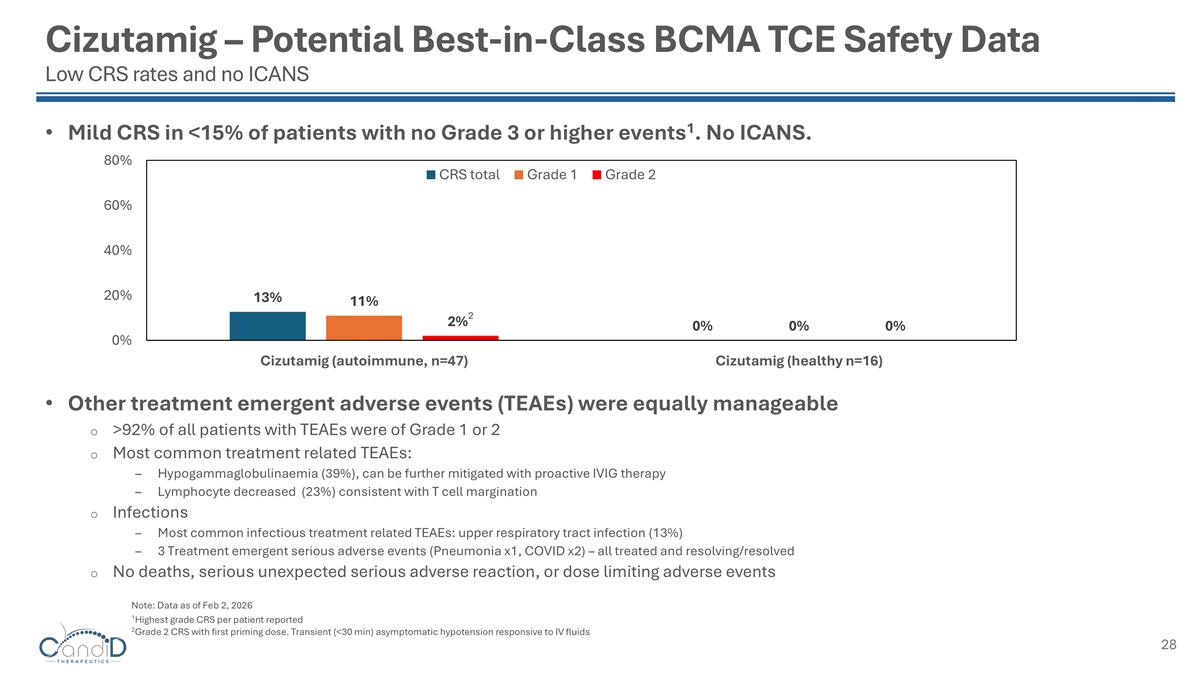
Cizutamig – Potential Best-in-Class BCMA TCE Safety Data Low CRS rates and no ICANS Mild CRS in <15% of patients with no Grade 3 or higher events1. No ICANS. Other treatment emergent adverse events (TEAEs) were equally manageable >92% of all patients with TEAEs were of Grade 1 or 2 Most common treatment related TEAEs: Hypogammaglobulinaemia (39%), can be further mitigated with proactive IVIG therapy Lymphocyte decreased (23%) consistent with T cell margination Infections Most common infectious treatment related TEAEs: upper respiratory tract infection (13%) 3 Treatment emergent serious adverse events (Pneumonia x1, COVID x2) – all treated and resolving/resolved No deaths, serious unexpected serious adverse reaction, or dose limiting adverse events 1Highest grade CRS per patient reported 2Grade 2 CRS with first priming dose. Transient (<30 min) asymptomatic hypotension responsive to IV fluids Note: Data as of Feb 2, 2026
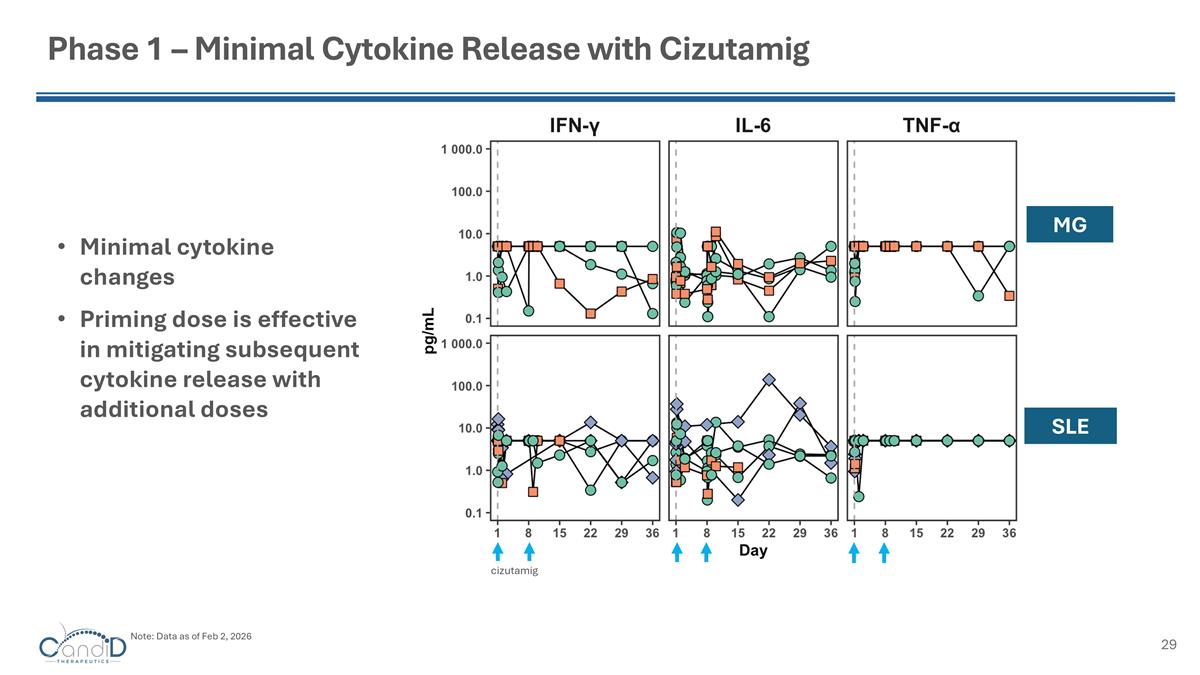
Phase 1 – Minimal Cytokine Release with Cizutamig Minimal cytokine changes Priming dose is effective in mitigating subsequent cytokine release with additional doses MG SLE cizutamig Note: Data as of Feb 2, 2026

CND261– CD20/CD3 Clinical Stage Antibody Design elements High affinity binding to CD20 which is expressed broadly on B-cells Retained Fc effector function with very weak CD3 affinity In vitro data à reduced cytokine release while maintaining B-cell cytotoxicity as compared to other CD20 TCEs Clinical data in oncology and autoimmune Ph 1 dose escalation (1mg to 300mg, 9 cohorts, n=94) completed in patients with lymphoma in Australia and China 26 patients dosed in rheumatoid arthritis, systemic lupus, and vasculitis Manufacturing – GMP manufacturing currently at 500L scale with ability to further scale-up as needed (1+1 binding) Purposefully designed to mitigate cytokine release while maintaining cytotoxicity Fc effector intact CD3 very weak
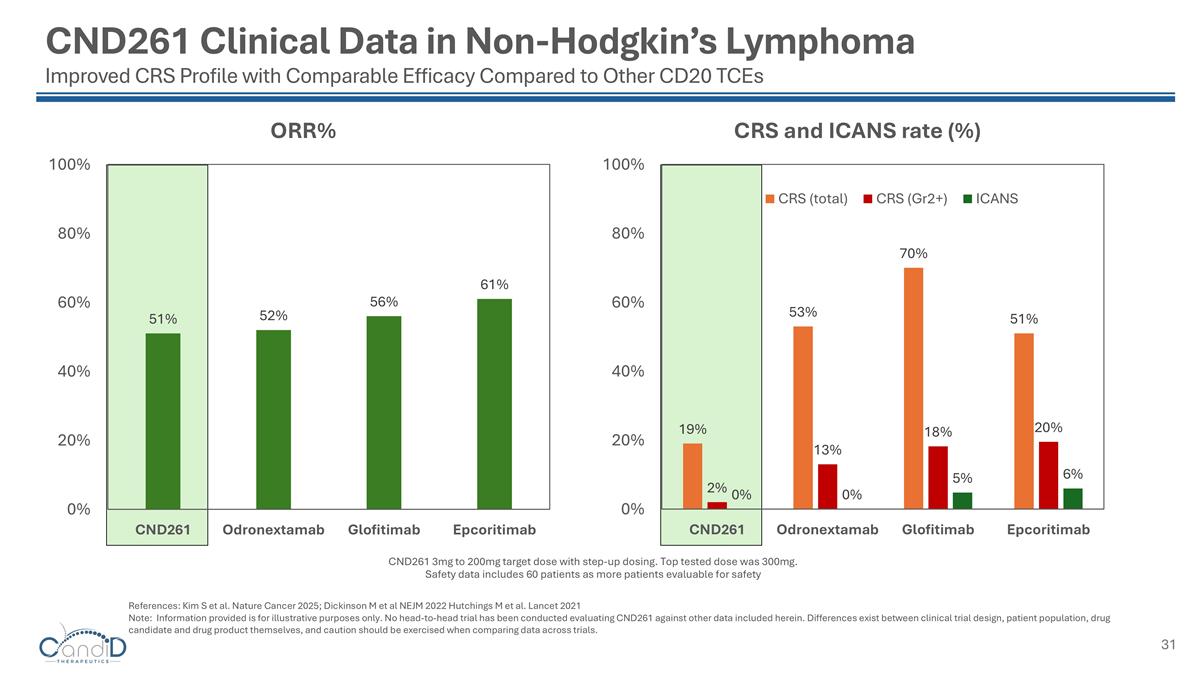
CND261 Clinical Data in Non-Hodgkin’s Lymphoma Improved CRS Profile with Comparable Efficacy Compared to Other CD20 TCEs References: Kim S et al. Nature Cancer 2025; Dickinson M et al NEJM 2022 Hutchings M et al. Lancet 2021 Note: Information provided is for illustrative purposes only. No head-to-head trial has been conducted evaluating CND261 against other data included herein. Differences exist between clinical trial design, patient population, drug candidate and drug product themselves, and caution should be exercised when comparing data across trials. CND261 3mg to 200mg target dose with step-up dosing. Top tested dose was 300mg. Safety data includes 60 patients as more patients evaluable for safety

CND261 – Tissue B-cell Depletion Observed and Well Tolerated Biopsy data available in four patients – deep CD20+ B-cell depletion observed CRS limited to Grade 1 only (19%) with No ICANS CND261 (autoimmune, n=26) CRS (total) 5 (19%) Grade 1 5 (19%) Grade 2 0 (0%) Grade 3+ 0 (0%) ICANS 0 (0%) Patient disease Timing of Biopsy Lymph Node Findings RA 8 wks Post Rx biopsy shows no B-cell reduction. No baseline biopsy. +ADA with impact on drug PK confirmed. RA 6 wks Post Rx biopsy shows full depletion of CD19 and CD20 B-cells. No baseline biopsy. SLE 4 wks 100% depletion of CD20+ B-cells, some CD19+ B-cells remain (preliminary) SLE 4 wks 100% depletion of CD20+ B-cells, some CD19+ B-cells remain (preliminary) Note: Data as of Feb 2, 2026
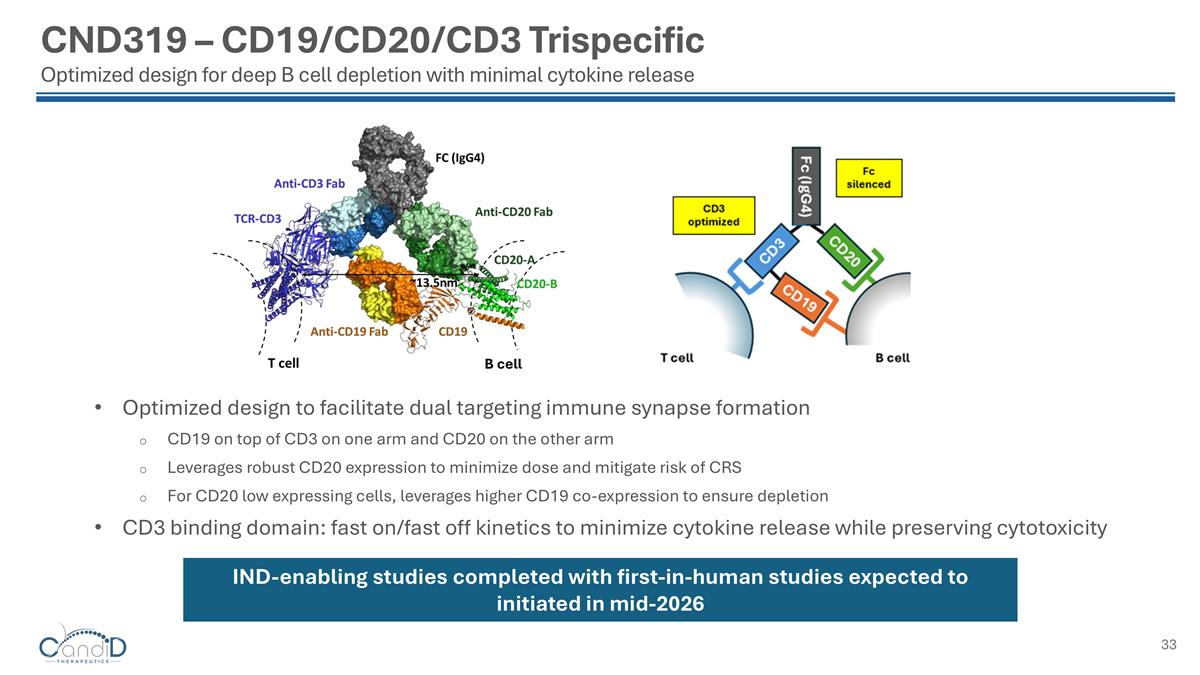
CND319 – CD19/CD20/CD3 Trispecific Optimized design for deep B cell depletion with minimal cytokine release Optimized design to facilitate dual targeting immune synapse formation CD19 on top of CD3 on one arm and CD20 on the other arm Leverages robust CD20 expression to minimize dose and mitigate risk of CRS For CD20 low expressing cells, leverages higher CD19 co-expression to ensure depletion CD3 binding domain: fast on/fast off kinetics to minimize cytokine release while preserving cytotoxicity IND-enabling studies completed with first-in-human studies expected to initiated in mid-2026 B cell
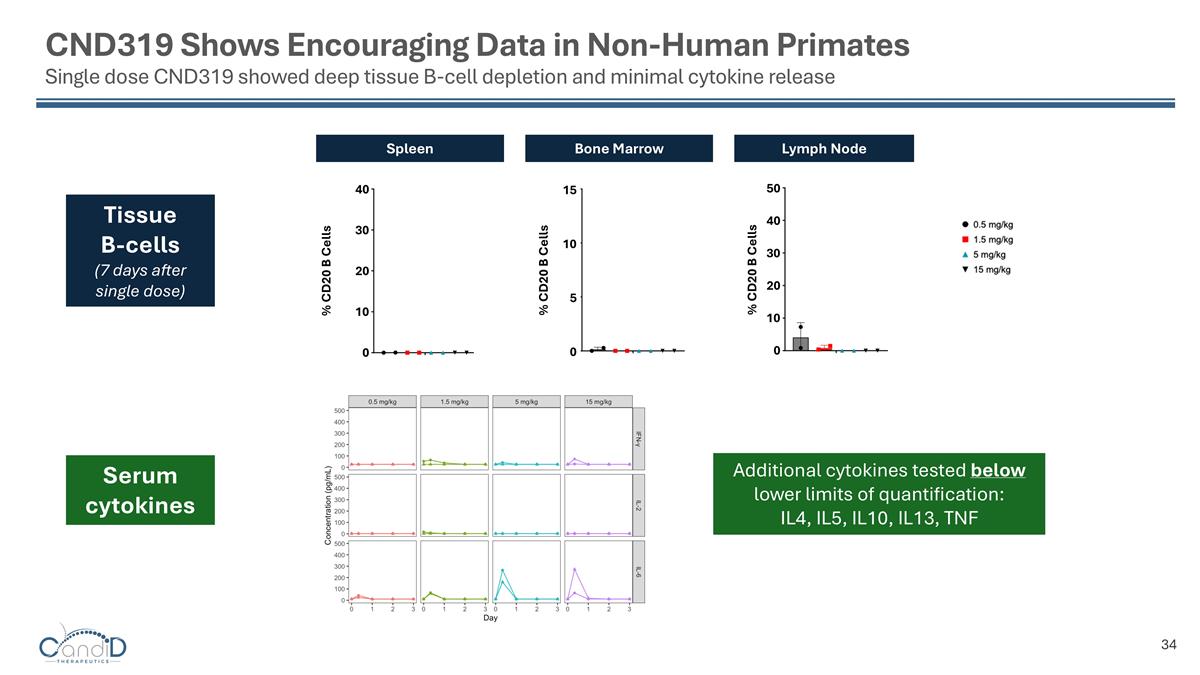
CND319 Shows Encouraging Data in Non-Human Primates Single dose CND319 showed deep tissue B-cell depletion and minimal cytokine release Spleen Bone Marrow Lymph Node Tissue B-cells (7 days after single dose) 40 30 20 10 0 15 10 5 0 40 30 20 10 0 50 % CD20 B Cells % CD20 B Cells % CD20 B Cells Additional cytokines tested below lower limits of quantification: IL4, IL5, IL10, IL13, TNF Serum cytokines
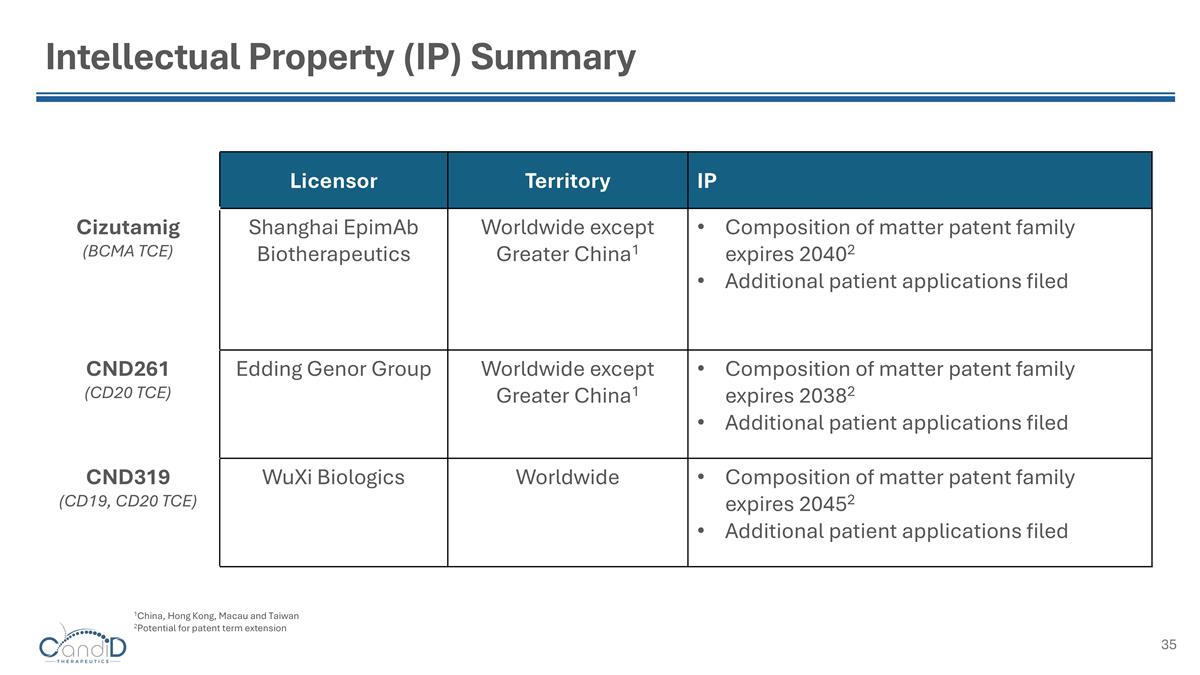
Intellectual Property (IP) Summary Licensor Territory IP Cizutamig (BCMA TCE) Shanghai EpimAb Biotherapeutics Worldwide except Greater China1 Composition of matter patent family expires 20402 Additional patient applications filed CND261 (CD20 TCE) Edding Genor Group Worldwide except Greater China1 Composition of matter patent family expires 20382 Additional patient applications filed CND319 (CD19, CD20 TCE) WuXi Biologics Worldwide Composition of matter patent family expires 20452 Additional patient applications filed 1China, Hong Kong, Macau and Taiwan 2Potential for patent term extension
Candid’s Strategic Playbook to Win Playbook Elements T-cell engager (TCE) antibodies Emerging clinical data show TCEs can match auto CAR-T on deep B cell depletion for immune reset We believe TCEs have an attractive profile for broad outpatient adoption – scalable and subcutaneous Portfolio approach Multiple drugs targeting different B cell antigens allows one to maximize potential value Not all diseases driven primarily by B cell pathology so obtaining clinical data across diseases is key Clinical data to drive decisions Clinical data is paramount to guide dose optimization and indication selection Many companies have been slow to recruit autoimmune patients for deep B-cell depletion Solely focused on TCEs for autoimmune diseases 70+ patients dosed since starting clinical evaluation Q2 ‘25 Fully operational team in China is capitalizing on the efficiencies to rapidly generate clinical data Potential first and best-in-class BCMA TCE in AID Potential best-in-class CD19 and/or CD20 TCE Double digit clinical studies ongoing