
4th Quarter & Full Year 2025 Financial Results & Corporate Update March 10th, 2026 .2

This presentation contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995, as amended, including, but not limited to, statements concerning: expected changes to BioNTech’s leadership and the transition of responsibilities at the Management Board; preliminary discussions between BioNTech and the co-founders regarding the potential contribution of certain BioNTech assets to an independent company; BioNTech’s expected revenues and net profit/(loss) related to sales of BioNTech’s COVID-19 vaccine, referred to as COMIRNATY where approved for use under full or conditional marketing authorization, in territories controlled by BioNTech’s collaboration partners, particularly for those figures that are derived from preliminary estimates provided by BioNTech’s partners; the rate and degree of market acceptance of BioNTech’s COVID-19 vaccine and, if approved, BioNTech’s investigational medicines; expectations regarding anticipated changes in COVID-19 vaccine demand; the initiation, timing, progress, results, and cost of BioNTech’s research and development programs, including BioNTech’s current and future preclinical studies and clinical trials, including statements regarding the expected timing of initiation, enrollment, and completion of studies or trials and related preparatory work and the availability of results, and the timing and outcome of applications for regulatory approvals and marketing authorizations; BioNTech’s expectations regarding potential future commercialization in oncology, including goals regarding timing and indications; the targeted timing and number of additional potentially registrational trials, and the registrational potential of any trial BioNTech may initiate; discussions with regulatory agencies; BioNTech’s expectations with respect to intellectual property; the impact of BioNTech’s collaboration and licensing agreements, including BioNTech’s partnership with BMS; BioNTech's expectations with respect to tariff policy; BioNTech’s estimates of revenues, research and development expenses, selling, general and administrative expenses, and capital expenditures for operating activities; BioNTech's expectations for upcoming scientific and investor presentations; and BioNTech’s expectations of net profit / (loss). In some cases, forward-looking statements can be identified by terminology such as “will,” “may,” “should,” “expects,” “intends,” “plans,” “aims,” “anticipates,” “believes,” “estimates,” “predicts,” “potential,” “continue,” or the negative of these terms or other comparable terminology, although not all forward-looking statements contain these words. The forward-looking statements in this presentation are based on BioNTech’s current expectations and beliefs of future events and are neither promises nor guarantees. You should not place undue reliance on these forward-looking statements because they involve known and unknown risks, uncertainties, and other factors, many of which are beyond BioNTech’s control, and which could cause actual results to differ materially and adversely from those expressed or implied by these forward- looking statements. These risks and uncertainties include, but are not limited to: BioNTech’s ability to successfully identify and recruit successors for the CEO and CMO positions; the uncertainties inherent in research and development, including the ability to meet anticipated clinical endpoints, commencement and/or completion dates for clinical trials, projected data release timelines, regulatory submission dates, regulatory approval dates and/or launch dates, as well as risks associated with preclinical and clinical data, including the data discussed in this release, and including the possibility of unfavorable new preclinical, clinical or safety data and further analyses of existing preclinical, clinical or safety data; the nature of the clinical data, which is subject to ongoing peer review, regulatory review and market interpretation; BioNTech’s pricing and coverage negotiations with governmental authorities, private health insurers and other third-party payors; the future commercial demand and medical need for initial or booster doses of a COVID-19 vaccine; the impact of tariffs and escalations in trade policy; competition from other COVID-19 vaccines or related to BioNTech’s other product candidates; the timing of and BioNTech’s ability to obtain and maintain regulatory approval for its product candidates; the ability of BioNTech’s COVID-19 vaccines to prevent COVID-19 caused by emerging virus variants; BioNTech’s ability to identify research opportunities and discover and develop investigational medicines; the ability and willingness of BioNTech’s third-party collaborators to continue research and development activities relating to BioNTech's development candidates and investigational medicines; unforeseen safety issues and potential claims that are alleged to arise from the use of products and product candidates developed or manufactured by BioNTech; BioNTech’s and its collaborators’ ability to commercialize and its product candidates, if approved; BioNTech’s ability to manage its development and related expenses; regulatory and political developments in the United States and other countries; BioNTech’s ability to effectively scale its production capabilities and manufacture its products and product candidates; risks relating to the global financial system and markets; and other factors not known to BioNTech at this time. You should review the risks and uncertainties described under the heading “Risk Factors” in BioNTech’s Annual Report on Form 20-F for the period ended December 31, 2025, and in subsequent filings made by BioNTech with the SEC, which are available on the SEC’s website at www.sec.gov. These forward-looking statements speak only as of the date hereof. Except as required by law, BioNTech disclaims any intention or responsibility for updating or revising any forward-looking statements contained in this presentation in the event of new information, future developments or otherwise. Furthermore, certain statements contained in this presentation relate to or are based on studies, publications, surveys and other data obtained from third-party sources and BioNTech’s own internal estimates and research. While BioNTech believes these third-party sources to be reliable as of the date of this presentation, it has not independently verified, and makes no representation as to the adequacy, fairness, accuracy or completeness of, any information obtained from third-party sources. In addition, any market data included in this presentation involves assumptions and limitations, and there can be no guarantee as to the accuracy or reliability of such assumptions. While BioNTech believes its own internal research is reliable, such research has not been verified by any independent source. In addition, BioNTech is the owner of various trademarks, trade names and service marks that may appear in this presentation. Certain other trademarks, trade names and service marks appearing in this presentation are the property of third parties. Solely for convenience, the trademarks and trade names in this presentation may be referred to without the ® and TM symbols, but such references should not be construed as any indicator that their respective owners will not assert, to the fullest extent under applicable law, their rights thereto. 2 An abbreviation directory of defined terms can be found at the end of the presentation. To be updated by Legal This Slide Presentation Includes Forward-Looking Statements

1 Progress Highlights Prof. Ugur Sahin, Co-founder & Chief Executive Officer 2 Oncology Execution Prof. Özlem Türeci, Co-founder & Chief Medical Officer 3 Financial Performance Ramón Zapata, Chief Financial Officer 3

Progress Highlights Ugur Sahin, Co-founder & Chief Executive Officer 1 4

Translating Science into Survival Building a Global Immunotherapy Powerhouse 5

2025 and Recent Achievements: Strong Performance and Pipeline Momentum 6 COVID-19 Market Leadership Advanced Key Oncology Programs Executed Key Strategic Deals Strengthened Financial Position Launched variant-adapted COVID-19 vaccine Leading COVID-19 vaccine market share1 Over 25 phase 2 & 3 oncology trials ongoing2 10 novel-combination trials ongoing with pumitamig3 Strategic BMS partnership Acquired Biotheus Acquired CureVac Beat increased 2025 revenue guidance €17.2 billion in cash, cash equivalents and securities4 1. Over 50%, including Italy, Spain, France, Germany, USA, Japan, Australia; 2. Includes Phase 2 or 3 trials for BNT111, BNT113, autogene cevumeran (partnered with Genentech, a member of the Roche Group), gotistobart (partnered with OncoC4), trastuzumab pamirtecan (partnered with DualityBio) and pumitamig (partnered with Bristol Myers Squibb) 3. Partnered with Bristol Myers Squibb (BMS); 4. Cash and cash equivalents plus security investments as of December 31, 2025, reached €17,235.6 million, comprising €7,675.4 million in cash and cash equivalents, €7,158.5 million in security investments disclosed as current financial assets and €2,401.7 million in security investments disclosed as non-current financial assets.
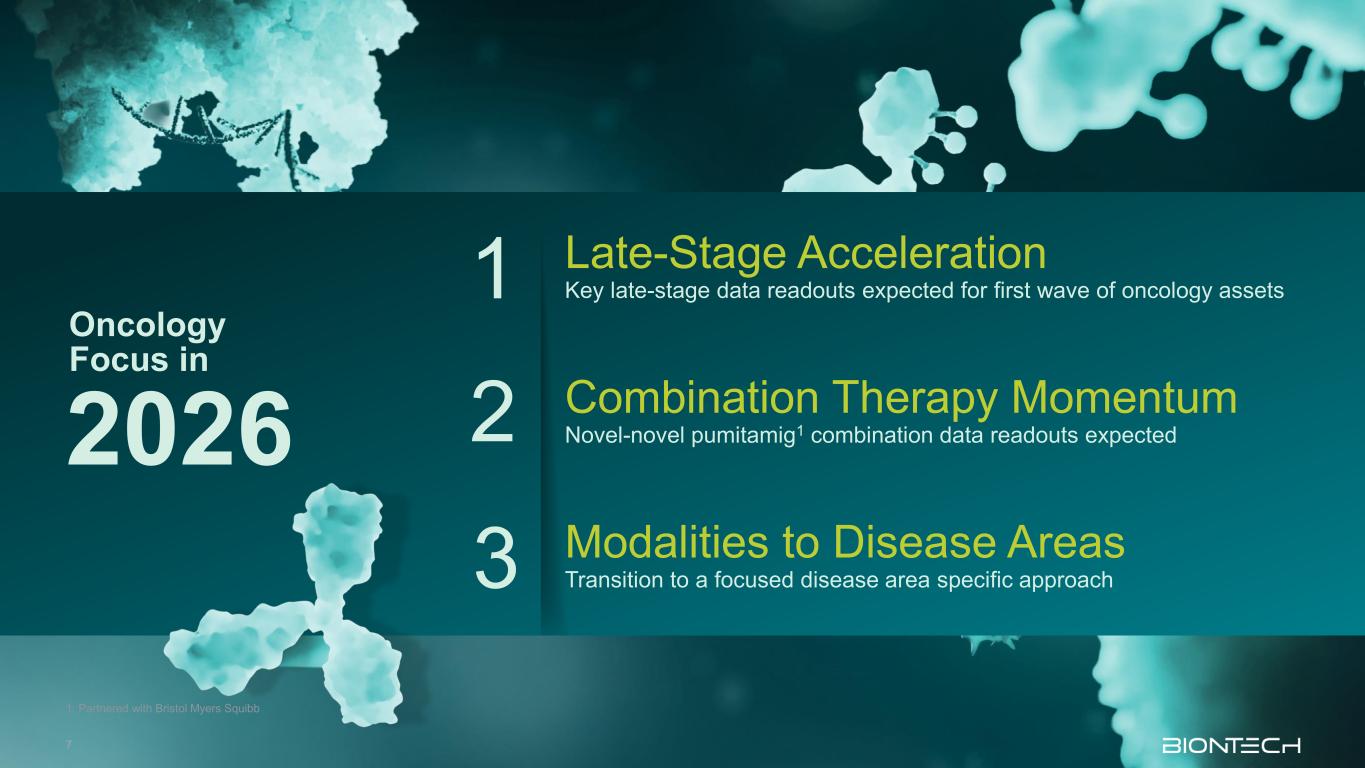
Modalities to Disease Areas Transition to a focused disease area specific approach Late-Stage Acceleration Key late-stage data readouts expected for first wave of oncology assets Oncology Focus in 2026 Combination Therapy Momentum Novel-novel pumitamig1 combination data readouts expected 1 2 3 1. Partnered with Bristol Myers Squibb 7
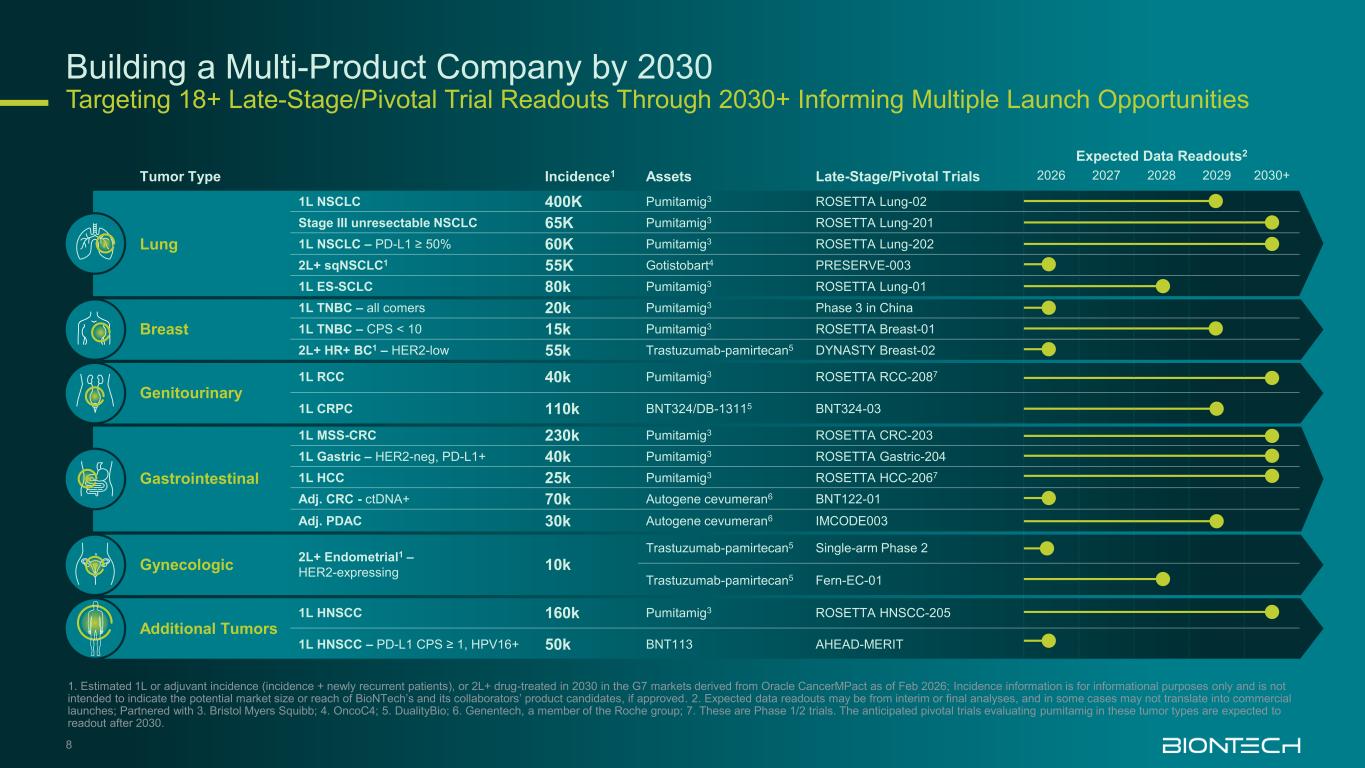
Building a Multi-Product Company by 2030 Targeting 18+ Late-Stage/Pivotal Trial Readouts Through 2030+ Informing Multiple Launch Opportunities 8 1. Estimated 1L or adjuvant incidence (incidence + newly recurrent patients), or 2L+ drug-treated in 2030 in the G7 markets derived from Oracle CancerMPact as of Feb 2026; Incidence information is for informational purposes only and is not intended to indicate the potential market size or reach of BioNTech’s and its collaborators’ product candidates, if approved. 2. Expected data readouts may be from interim or final analyses, and in some cases may not translate into commercial launches; Partnered with 3. Bristol Myers Squibb; 4. OncoC4; 5. DualityBio; 6. Genentech, a member of the Roche group; 7. These are Phase 1/2 trials. The anticipated pivotal trials evaluating pumitamig in these tumor types are expected to readout after 2030. Tumor Type Incidence1 Assets Late-Stage/Pivotal Trials Expected Data Readouts2 2026 2027 2028 2029 2030+ Lung 1L NSCLC 400K Pumitamig3 ROSETTA Lung-02 Stage III unresectable NSCLC 65K Pumitamig3 ROSETTA Lung-201 1L NSCLC – PD-L1 ≥ 50% 60K Pumitamig3 ROSETTA Lung-202 2L+ sqNSCLC1 55K Gotistobart4 PRESERVE-003 1L ES-SCLC 80k Pumitamig3 ROSETTA Lung-01 Breast 1L TNBC – all comers 20k Pumitamig3 Phase 3 in China 1L TNBC – CPS < 10 15k Pumitamig3 ROSETTA Breast-01 2L+ HR+ BC1 – HER2-low 55k Trastuzumab-pamirtecan5 DYNASTY Breast-02 Genitourinary 1L RCC 40k Pumitamig3 ROSETTA RCC-2087 1L CRPC 110k BNT324/DB-13115 BNT324-03 Gastrointestinal 1L MSS-CRC 230k Pumitamig3 ROSETTA CRC-203 1L Gastric – HER2-neg, PD-L1+ 40k Pumitamig3 ROSETTA Gastric-204 1L HCC 25k Pumitamig3 ROSETTA HCC-2067 Adj. CRC - ctDNA+ 70k Autogene cevumeran6 BNT122-01 Adj. PDAC 30k Autogene cevumeran6 IMCODE003 Gynecologic 2L+ Endometrial1 – HER2-expressing 10k Trastuzumab-pamirtecan5 Single-arm Phase 2 Trastuzumab-pamirtecan5 Fern-EC-01 Additional Tumors 1L HNSCC 160k Pumitamig3 ROSETTA HNSCC-205 1L HNSCC – PD-L1 CPS ≥ 1, HPV16+ 50k BNT113 AHEAD-MERIT

BioNTech New Company Focus Becoming a multi-product company by 2030 Sharpening focus on growing late-stage clinical pipeline spanning immunomodulator, ADC and mRNA candidates Pioneering next-generation mRNA innovations with disruptive potential Next Steps CEO and CMO transition by end of 2026 Executive search underway BioNTech planning to contribute related rights and mRNA technologies in exchange for a minority stake Signing of binding agreements expected by end of H1 2026 Strategic Focus to Maximize Value for Patients and Shareholders 9 The information above is based on a non-binding letter of intent and is subject to the relevant parties entering into a final, definitive agreement.

Oncology Execution Özlem Türeci, Co-founder & Chief Medical Officer 2 10
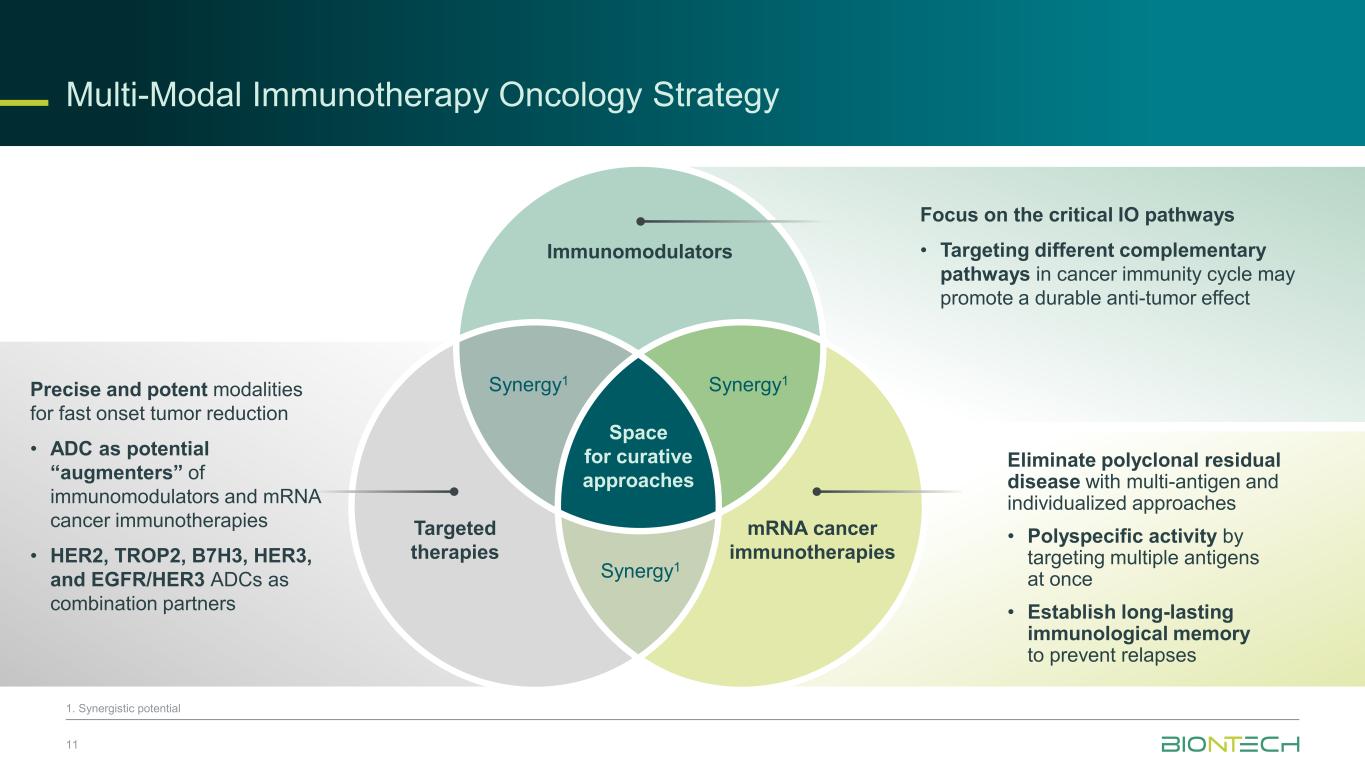
11 1. Synergistic potential Space for curative approaches Immunomodulators Targeted therapies Synergy1Synergy1 Synergy1 mRNA cancer immunotherapies Focus on the critical IO pathways • Targeting different complementary pathways in cancer immunity cycle may promote a durable anti-tumor effect Eliminate polyclonal residual disease with multi-antigen and individualized approaches • Polyspecific activity by targeting multiple antigens at once • Establish long-lasting immunological memory to prevent relapses Precise and potent modalities for fast onset tumor reduction • ADC as potential “augmenters” of immunomodulators and mRNA cancer immunotherapies • HER2, TROP2, B7H3, HER3, and EGFR/HER3 ADCs as combination partners Multi-Modal Immunotherapy Oncology Strategy

Establish ElevateExpand SCLC 1L Ph3 (Global) 2L Ph3 (China) NSCLC 1L Ph2/3 (Global) 1L NSCLC Stage III unres. Ph3 (Global) 1L NSCLC PD-L1 ≥ 50% Ph3 (Global) TNBC 1L Ph3 trial (Global) 1L Ph3 (China) Pumitamig Strategy to Build a Proprietary IO Franchise 12 1. Partnered with Bristol Myers Squibb. Registrational-Intent 1L Gastric Ph2/3 (Global) 1L CRC Ph2/3 (Global) 1L HNSCC Ph2/3 (Global) Signal-Seeking 1L PDAC Ph2 (China) 1L GBM Ph2 (China) 1L RCC Ph1/2 (Global) 1L HCC Ph1/2 (Global) And others Combining with our ADCs targeting • HER2 • TROP2 • B7H3 Exploring potential synergies with our IO agents • EpCam x 4-1BB • TIGIT x PVRIG • mRNA cancer immunotherapy Broad Pan-Tumor Applicability With Standard-of-Care Chemotherapy 12+ trials exploring pumitamig1 in 10+ new indications Potential New Standards of Care 10+ novel-novel combinations • HER3 • EGFR x HER3 • Novel targets Foundational Registrations Registrational trials with pumitamig1 ongoing in 3 high-impact tumors

Establish ElevateExpand SCLC 1L Ph3 (Global) 2L Ph3 (China) NSCLC 1L Ph2/3 (Global) 1L NSCLC Stage III unres. Ph3 (Global) 1L NSCLC PD-L1 ≥ 50% Ph3 (Global) TNBC 1L Ph3 trial (Global) 1L Ph3 (China) Pumitamig Strategy to Build a Proprietary IO Franchise 13 Registrational-Intent 1L Gastric Ph2/3 (Global) 1L CRC Ph2/3 (Global) 1L HNSCC Ph2/3 (Global) Signal-Seeking 1L PDAC Ph2 (China) 1L GBM Ph2 (China) 1L RCC Ph1/2 (Global) 1L HCC Ph1/2 (Global) And others Combining with our ADCs targeting • HER2 • TROP2 • B7H3 Exploring potential synergies with our IO agents • EpCam x 4-1BB • TIGIT x PVRIG • mRNA cancer immunotherapy Potential New Standards of Care 10+ novel-novel combinations • HER3 • EGFR x HER3 • Novel targets Foundational Registrations Registrational trials with pumitamig1 ongoing in 3 high-impact tumors Broad Pan-Tumor Applicability With Standard-of-Care Chemotherapy 12+ trials exploring pumitamig1 in 10+ new indications 1. Partnered with Bristol Myers Squibb.
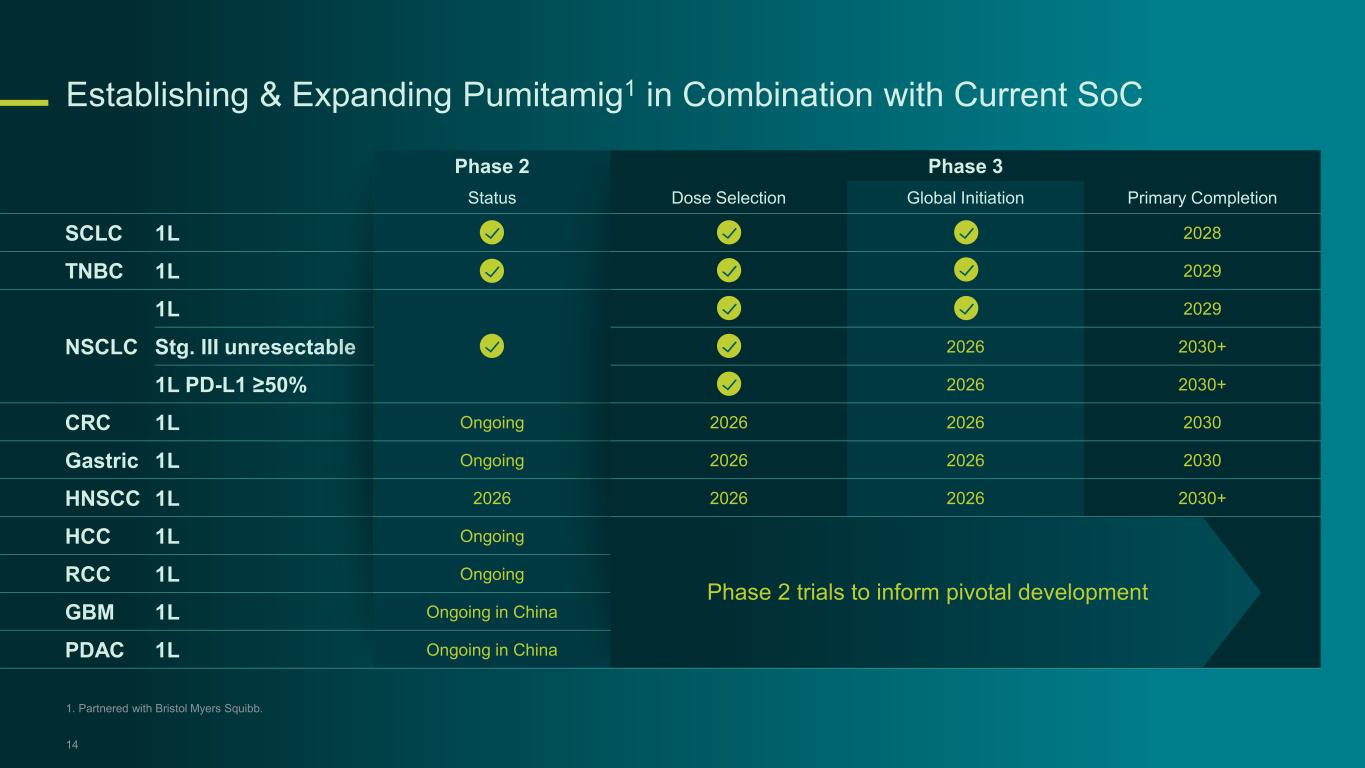
Establishing & Expanding Pumitamig1 in Combination with Current SoC 14 1. Partnered with Bristol Myers Squibb. Phase 2 Phase 3 Status Dose Selection Global Initiation Primary Completion SCLC 1L 2028 TNBC 1L 2029 NSCLC 1L 2029 Stg. III unresectable 2026 2030+ 1L PD-L1 ≥50% 2026 2030+ CRC 1L Ongoing 2026 2026 2030 Gastric 1L Ongoing 2026 2026 2030 HNSCC 1L 2026 2026 2026 2030+ HCC 1L Ongoing RCC 1L Ongoing GBM 1L Ongoing in China PDAC 1L Ongoing in China Phase 2 trials to inform pivotal development
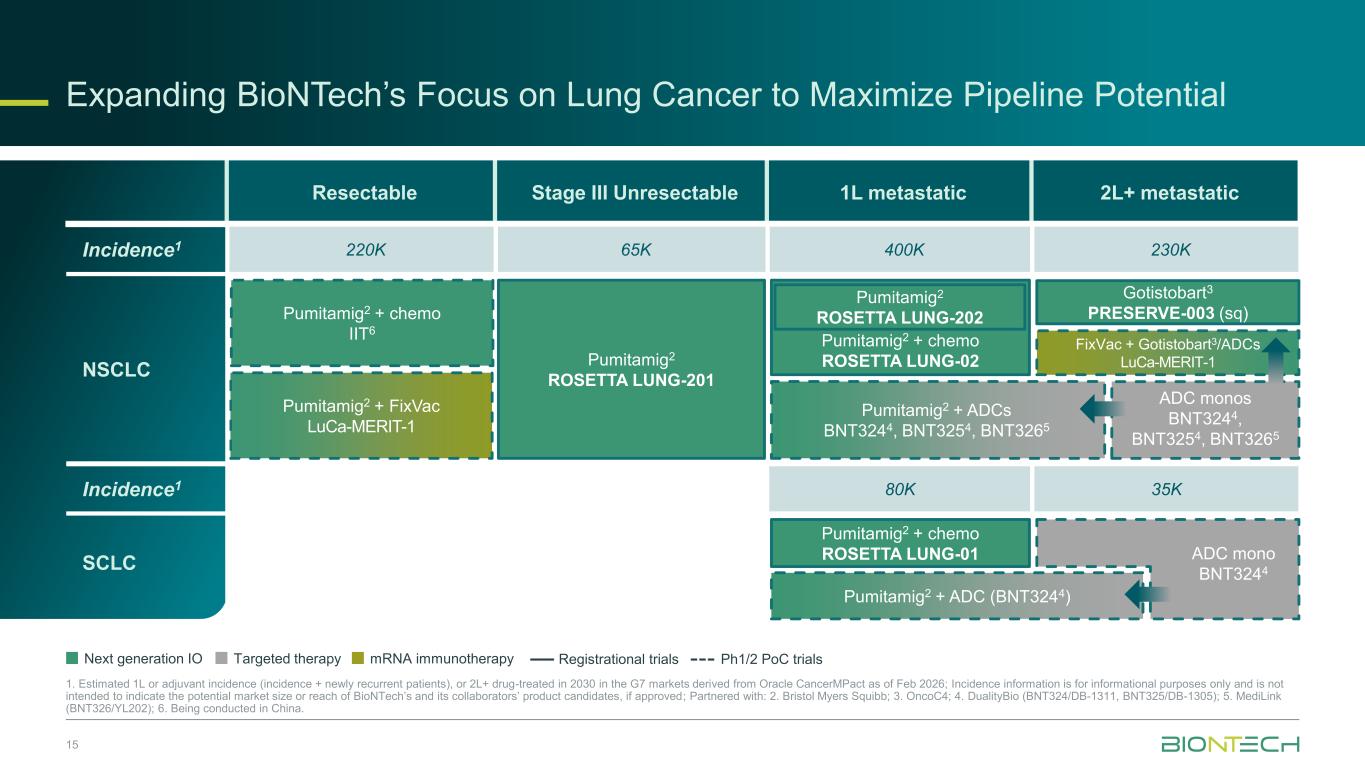
Expanding BioNTech’s Focus on Lung Cancer to Maximize Pipeline Potential 15 1. Estimated 1L or adjuvant incidence (incidence + newly recurrent patients), or 2L+ drug-treated in 2030 in the G7 markets derived from Oracle CancerMPact as of Feb 2026; Incidence information is for informational purposes only and is not intended to indicate the potential market size or reach of BioNTech’s and its collaborators’ product candidates, if approved; Partnered with: 2. Bristol Myers Squibb; 3. OncoC4; 4. DualityBio (BNT324/DB-1311, BNT325/DB-1305); 5. MediLink (BNT326/YL202); 6. Being conducted in China. Resectable Stage III Unresectable 1L metastatic 2L+ metastatic Next generation IO Targeted therapy mRNA immunotherapy Registrational trials Ph1/2 PoC trials Gotistobart3 PRESERVE-003 (sq) FixVac + Gotistobart3/ADCs LuCa-MERIT-1 Pumitamig2 + chemo ROSETTA LUNG-01 Pumitamig2 + chemo IIT6 Pumitamig2 + chemo ROSETTA LUNG-02 ADC monos BNT3244, BNT3254, BNT3265 ADC mono BNT3244 Pumitamig2 + ADC (BNT3244) Pumitamig2 + ADCs BNT3244, BNT3254, BNT3265 Pumitamig2 ROSETTA LUNG-201 Pumitamig2 ROSETTA LUNG-202 Pumitamig2 + FixVac LuCa-MERIT-1 Incidence1 80K 35K SCLC Incidence1 220K 65K 400K 230K NSCLC

Establish ElevateExpand SCLC 1L Ph3 (Global) 2L Ph3 (China) NSCLC 1L Ph2/3 (Global) 1L NSCLC St III unres. Ph3 (Global) 1L NSCLC PD-L1 ≥ 50% Ph3 (Global) TNBC 1L Ph3 trial (Global) 1L Ph3 (China) Executing a Parallel Three-Wave Strategy to Build a Proprietary IO Franchise 16 Registrational-Intent 1L Gastric Ph2/3 (Global) 1L CRC Ph2/3 (Global) 1L HNSCC Ph2/3 (Global) Signal-Seeking 1L PDAC Ph2 (China) 1L GBM Ph2 (China) 1L RCC Ph1/2 (Global) 1L HCC Ph1/2 (Global) And others Combining with our ADCs targeting • HER2 • TROP2 • B7H3 Exploring potential synergies with our IO agents • EpCam x 4-1BB • TIGIT x PVRIG • mRNA cancer immunotherapy Potential New Standards of Care 10+ novel-novel combinations • HER3 • EGFR x HER3 • Novel targets 1. Partnered with Bristol Myers Squibb. Foundational Registrations Registrational trials with pumitamig1 ongoing in 3 high-impact tumors Broad Pan-Tumor Applicability With Standard-of-Care Chemotherapy 12+ trials exploring pumitamig1 in 10+ new indications
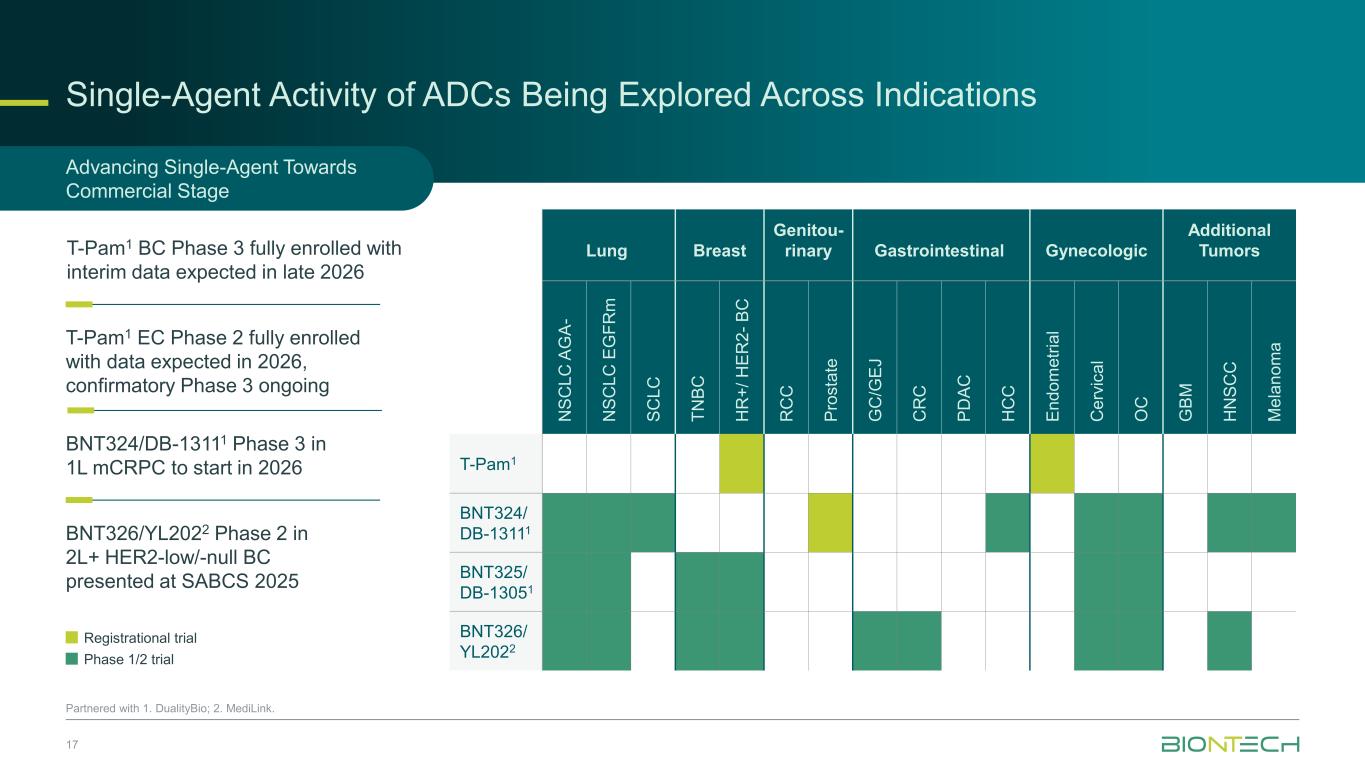
Single-Agent Activity of ADCs Being Explored Across Indications 17 Partnered with 1. DualityBio; 2. MediLink. Registrational trial Phase 1/2 trial Advancing Single-Agent Towards Commercial Stage T-Pam1 EC Phase 2 fully enrolled with data expected in 2026, confirmatory Phase 3 ongoing T-Pam1 BC Phase 3 fully enrolled with interim data expected in late 2026 BNT324/DB-13111 Phase 3 in 1L mCRPC to start in 2026 BNT326/YL2022 Phase 2 in 2L+ HER2-low/-null BC presented at SABCS 2025 Lung Breast Genitou- rinary Gastrointestinal Gynecologic Additional Tumors N S C L C A G A - N S C L C E G F R m S C L C T N B C H R + / H E R 2 - B C R C C P ro s ta te G C /G E J C R C P D A C H C C E n d o m e tr ia l C e rv ic a l O C G B M H N S C C M e la n o m a T-Pam1 BNT324/ DB-13111 BNT325/ DB-13051 BNT326/ YL2022
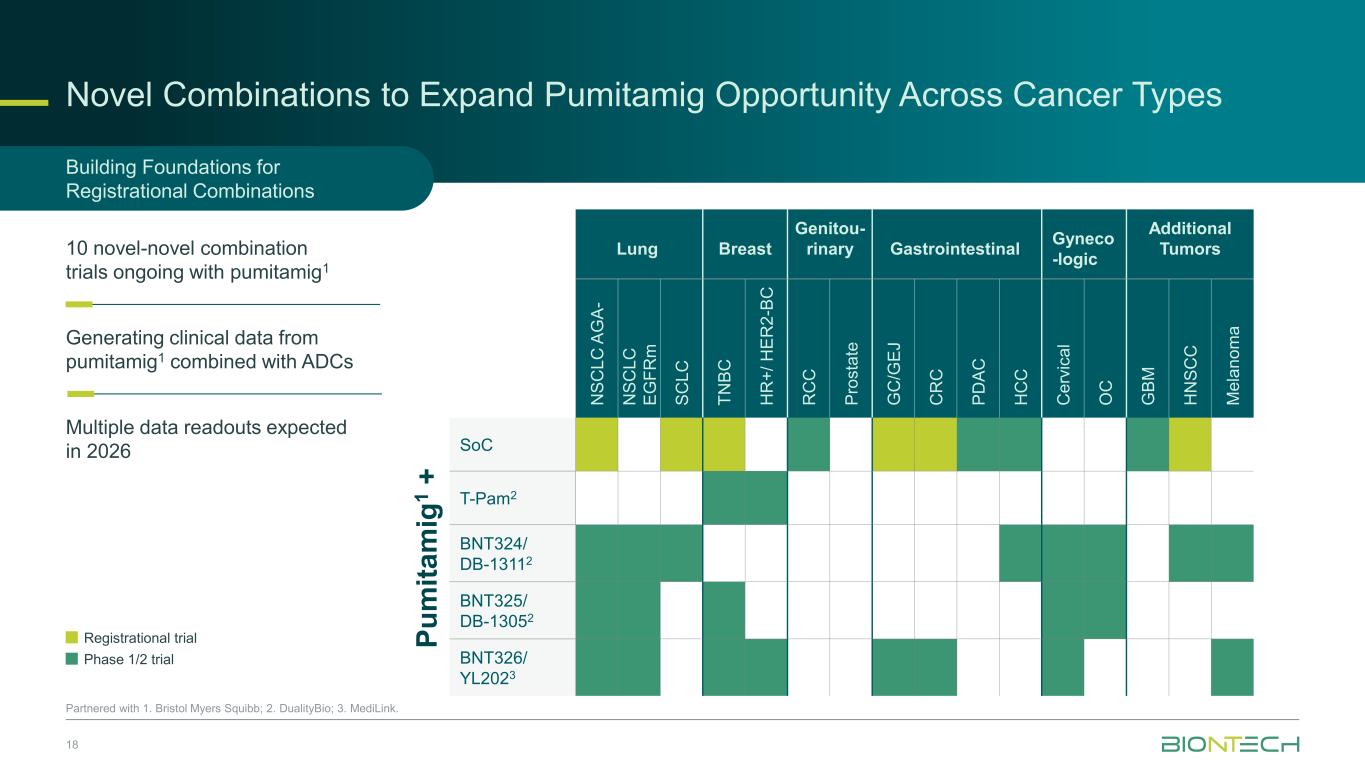
Novel Combinations to Expand Pumitamig Opportunity Across Cancer Types 18 Partnered with 1. Bristol Myers Squibb; 2. DualityBio; 3. MediLink. Registrational trial Phase 1/2 trial Building Foundations for Registrational Combinations Generating clinical data from pumitamig1 combined with ADCs 10 novel-novel combination trials ongoing with pumitamig1 Multiple data readouts expected in 2026 P u m it a m ig 1 + Lung Breast Genitou- rinary Gastrointestinal Gyneco -logic Additional Tumors N S C L C A G A - N S C L C E G F R m S C L C T N B C H R + / H E R 2 -B C R C C P ro s ta te G C /G E J C R C P D A C H C C C e rv ic a l O C G B M H N S C C M e la n o m a SoC T-Pam2 BNT324/ DB-13112 BNT325/ DB-13052 BNT326/ YL2023
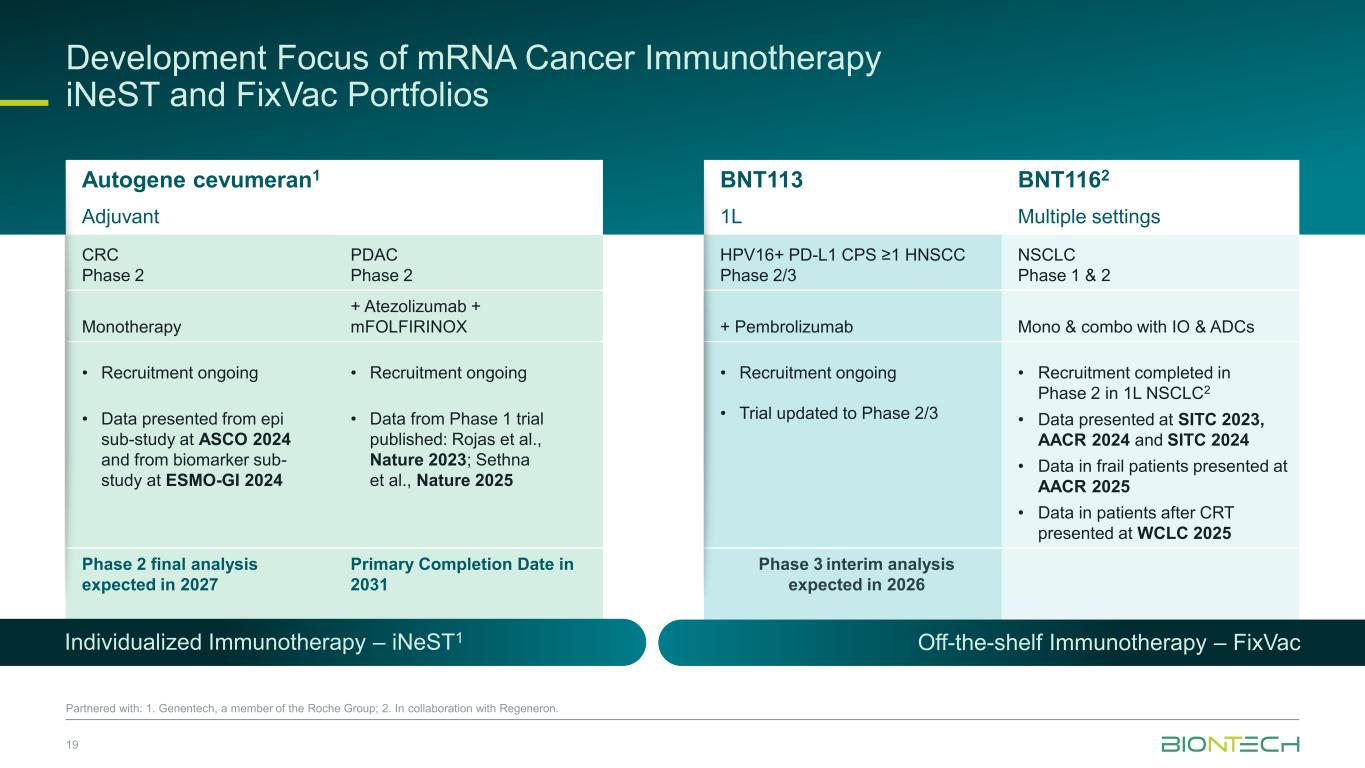
Development Focus of mRNA Cancer Immunotherapy iNeST and FixVac Portfolios 19 Partnered with: 1. Genentech, a member of the Roche Group; 2. In collaboration with Regeneron. Autogene cevumeran1 BNT113 BNT1162 Adjuvant 1L Multiple settings CRC Phase 2 PDAC Phase 2 HPV16+ PD-L1 CPS ≥1 HNSCC Phase 2/3 NSCLC Phase 1 & 2 Monotherapy + Atezolizumab + mFOLFIRINOX + Pembrolizumab Mono & combo with IO & ADCs • Recruitment ongoing • Data presented from epi sub-study at ASCO 2024 and from biomarker sub- study at ESMO-GI 2024 • Recruitment ongoing • Data from Phase 1 trial published: Rojas et al., Nature 2023; Sethna et al., Nature 2025 • Recruitment ongoing • Trial updated to Phase 2/3 • Recruitment completed in Phase 2 in 1L NSCLC2 • Data presented at SITC 2023, AACR 2024 and SITC 2024 • Data in frail patients presented at AACR 2025 • Data in patients after CRT presented at WCLC 2025 Phase 2 final analysis expected in 2027 Primary Completion Date in 2031 Phase 3 interim analysis expected in 2026 Individualized Immunotherapy – iNeST1 Off-the-shelf Immunotherapy – FixVac
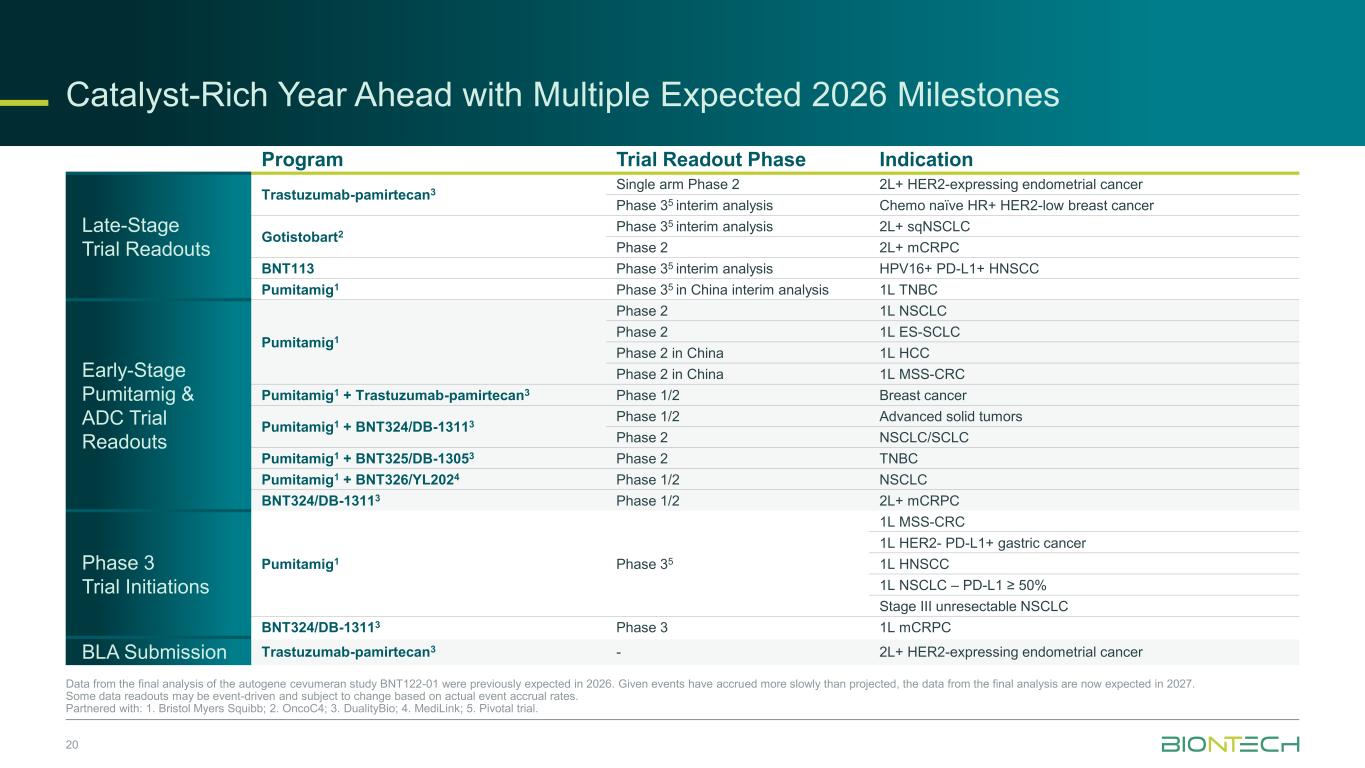
20 Data from the final analysis of the autogene cevumeran study BNT122-01 were previously expected in 2026. Given events have accrued more slowly than projected, the data from the final analysis are now expected in 2027. Some data readouts may be event-driven and subject to change based on actual event accrual rates. Partnered with: 1. Bristol Myers Squibb; 2. OncoC4; 3. DualityBio; 4. MediLink; 5. Pivotal trial. Program Trial Readout Phase Indication Late-Stage Trial Readouts Trastuzumab-pamirtecan3 Single arm Phase 2 2L+ HER2-expressing endometrial cancer Phase 35 interim analysis Chemo naïve HR+ HER2-low breast cancer Gotistobart2 Phase 35 interim analysis 2L+ sqNSCLC Phase 2 2L+ mCRPC BNT113 Phase 35 interim analysis HPV16+ PD-L1+ HNSCC Pumitamig1 Phase 35 in China interim analysis 1L TNBC Early-Stage Pumitamig & ADC Trial Readouts Pumitamig1 Phase 2 1L NSCLC Phase 2 1L ES-SCLC Phase 2 in China 1L HCC Phase 2 in China 1L MSS-CRC Pumitamig1 + Trastuzumab-pamirtecan3 Phase 1/2 Breast cancer Pumitamig1 + BNT324/DB-13113 Phase 1/2 Advanced solid tumors Phase 2 NSCLC/SCLC Pumitamig1 + BNT325/DB-13053 Phase 2 TNBC Pumitamig1 + BNT326/YL2024 Phase 1/2 NSCLC BNT324/DB-13113 Phase 1/2 2L+ mCRPC Phase 3 Trial Initiations Pumitamig1 Phase 35 1L MSS-CRC 1L HER2- PD-L1+ gastric cancer 1L HNSCC 1L NSCLC – PD-L1 ≥ 50% Stage III unresectable NSCLC BNT324/DB-13113 Phase 3 1L mCRPC BLA Submission Trastuzumab-pamirtecan3 - 2L+ HER2-expressing endometrial cancer Catalyst-Rich Year Ahead with Multiple Expected 2026 Milestones

Financial Performance Ramón Zapata, Chief Financial Officer 3 21

Full Year 2025 Financial Results Compared to Guidance 22 1. Numbers have been rounded. More information can be found in BioNTech’s Report on Form 20-F for the year ended December 31, 2025, filed on March 10, 2026, which is available at www.sec.gov. In € millions FY 2025 IFRS Results1 FY 2025 IFRS Guidance Total Revenues 2,870 2,600 – 2,800 R&D Expenses 2,105 2,000 – 2,200 SG&A Expenses 624 550 – 650 Capital Expenditures for Operating Activities 198 200 – 250
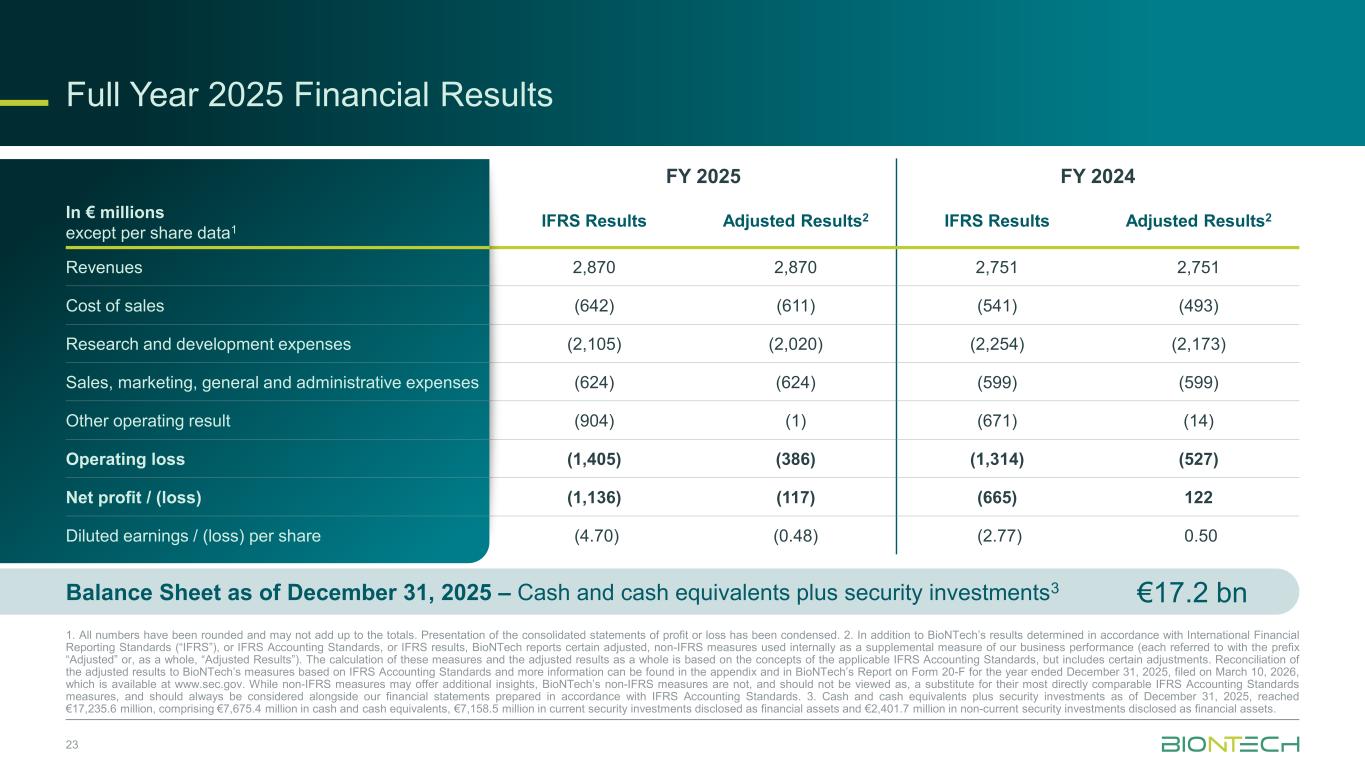
Full Year 2025 Financial Results 23 1. All numbers have been rounded and may not add up to the totals. Presentation of the consolidated statements of profit or loss has been condensed. 2. In addition to BioNTech’s results determined in accordance with International Financial Reporting Standards (“IFRS”), or IFRS Accounting Standards, or IFRS results, BioNTech reports certain adjusted, non-IFRS measures used internally as a supplemental measure of our business performance (each referred to with the prefix “Adjusted” or, as a whole, “Adjusted Results”). The calculation of these measures and the adjusted results as a whole is based on the concepts of the applicable IFRS Accounting Standards, but includes certain adjustments. Reconciliation of the adjusted results to BioNTech’s measures based on IFRS Accounting Standards and more information can be found in the appendix and in BioNTech’s Report on Form 20-F for the year ended December 31, 2025, filed on March 10, 2026, which is available at www.sec.gov. While non-IFRS measures may offer additional insights, BioNTech’s non-IFRS measures are not, and should not be viewed as, a substitute for their most directly comparable IFRS Accounting Standards measures, and should always be considered alongside our financial statements prepared in accordance with IFRS Accounting Standards. 3. Cash and cash equivalents plus security investments as of December 31, 2025, reached €17,235.6 million, comprising €7,675.4 million in cash and cash equivalents, €7,158.5 million in current security investments disclosed as financial assets and €2,401.7 million in non-current security investments disclosed as financial assets. In € millions except per share data1 FY 2025 FY 2024 IFRS Results Adjusted Results2 IFRS Results Adjusted Results2 Revenues 2,870 2,870 2,751 2,751 Cost of sales (642) (611) (541) (493) Research and development expenses (2,105) (2,020) (2,254) (2,173) Sales, marketing, general and administrative expenses (624) (624) (599) (599) Other operating result (904) (1) (671) (14) Operating loss (1,405) (386) (1,314) (527) Net profit / (loss) (1,136) (117) (665) 122 Diluted earnings / (loss) per share (4.70) (0.48) (2.77) 0.50 €17.2 bnBalance Sheet as of December 31, 2025 – Cash and cash equivalents plus security investments3
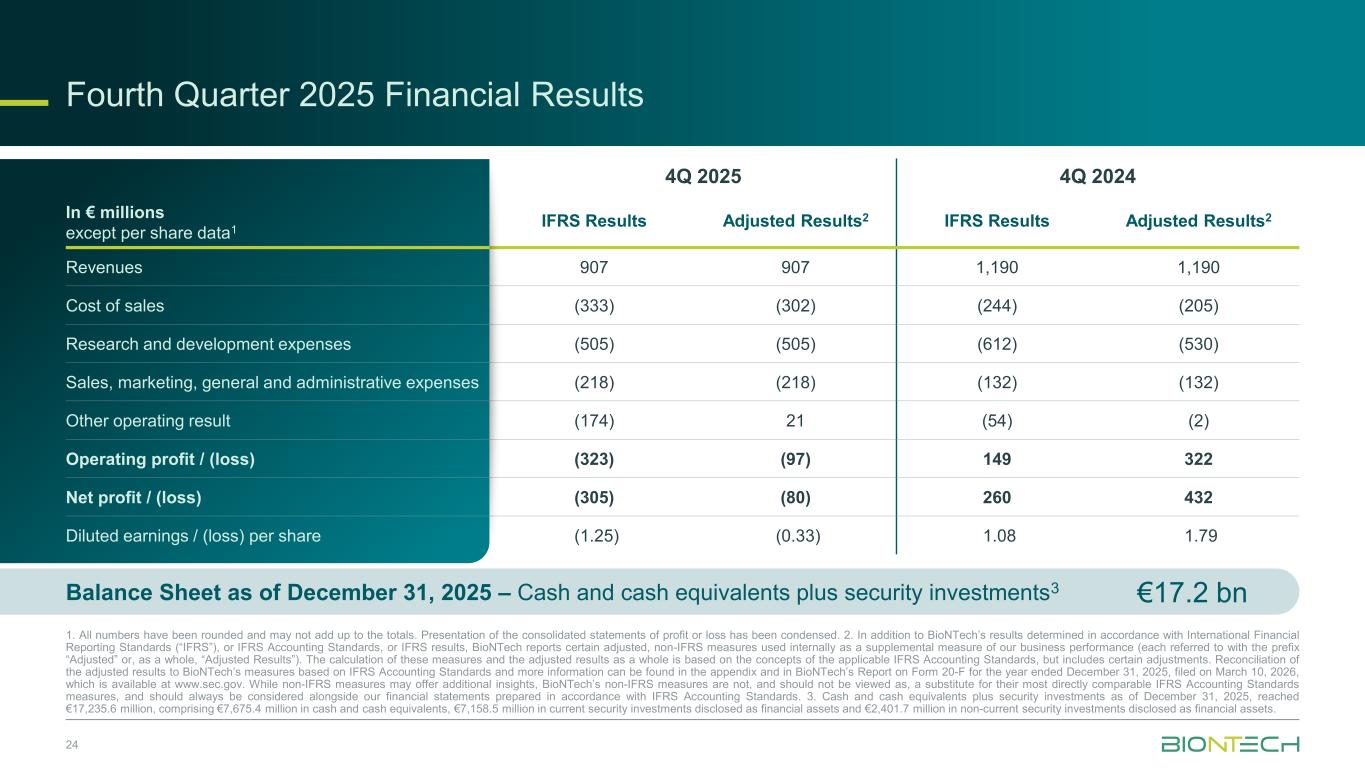
Fourth Quarter 2025 Financial Results 24 1. All numbers have been rounded and may not add up to the totals. Presentation of the consolidated statements of profit or loss has been condensed. 2. In addition to BioNTech’s results determined in accordance with International Financial Reporting Standards (“IFRS”), or IFRS Accounting Standards, or IFRS results, BioNTech reports certain adjusted, non-IFRS measures used internally as a supplemental measure of our business performance (each referred to with the prefix “Adjusted” or, as a whole, “Adjusted Results”). The calculation of these measures and the adjusted results as a whole is based on the concepts of the applicable IFRS Accounting Standards, but includes certain adjustments. Reconciliation of the adjusted results to BioNTech’s measures based on IFRS Accounting Standards and more information can be found in the appendix and in BioNTech’s Report on Form 20-F for the year ended December 31, 2025, filed on March 10, 2026, which is available at www.sec.gov. While non-IFRS measures may offer additional insights, BioNTech’s non-IFRS measures are not, and should not be viewed as, a substitute for their most directly comparable IFRS Accounting Standards measures, and should always be considered alongside our financial statements prepared in accordance with IFRS Accounting Standards. 3. Cash and cash equivalents plus security investments as of December 31, 2025, reached €17,235.6 million, comprising €7,675.4 million in cash and cash equivalents, €7,158.5 million in current security investments disclosed as financial assets and €2,401.7 million in non-current security investments disclosed as financial assets. In € millions except per share data1 4Q 2025 4Q 2024 IFRS Results Adjusted Results2 IFRS Results Adjusted Results2 Revenues 907 907 1,190 1,190 Cost of sales (333) (302) (244) (205) Research and development expenses (505) (505) (612) (530) Sales, marketing, general and administrative expenses (218) (218) (132) (132) Other operating result (174) 21 (54) (2) Operating profit / (loss) (323) (97) 149 322 Net profit / (loss) (305) (80) 260 432 Diluted earnings / (loss) per share (1.25) (0.33) 1.08 1.79 €17.2 bnBalance Sheet as of December 31, 2025 – Cash and cash equivalents plus security investments3

Full Year 2026 Financial Guidance1 25 1. Excludes risks that are not yet known and/or quantifiable and related activities. It includes effects identified from licensing arrangements, collaborations and Merger & Acquisitions (“M&A”) transactions to the extent disclosed. The guidance is based on non-IFRS measures and excludes certain effects compared to measures based on IFRS Accounting Standards. More information can be found in BioNTech’s Report on Form 20-F for the year ended December 31, 2025 filed on March 10, 2026, which is available at www.sec.gov. In € millions FY 2026 non-IFRS Guidance Total Revenues 2,000 – 2,300 Adjusted R&D Expenses 2,200 – 2,500 Adjusted SG&A Expenses 700 – 800 Revenue Guidance Considerations • Competitive market dynamics in the United States • Begin managing transition of multi-year contracts in Europe, and specifically in Germany where BioNTech recognizes direct sales • Stable revenues from the collaboration with BMS, from a pandemic preparedness contract with the German government, and from the BioNTech Group service businesses • No one-time revenue from Pfizer opt-out from further development of shingles program
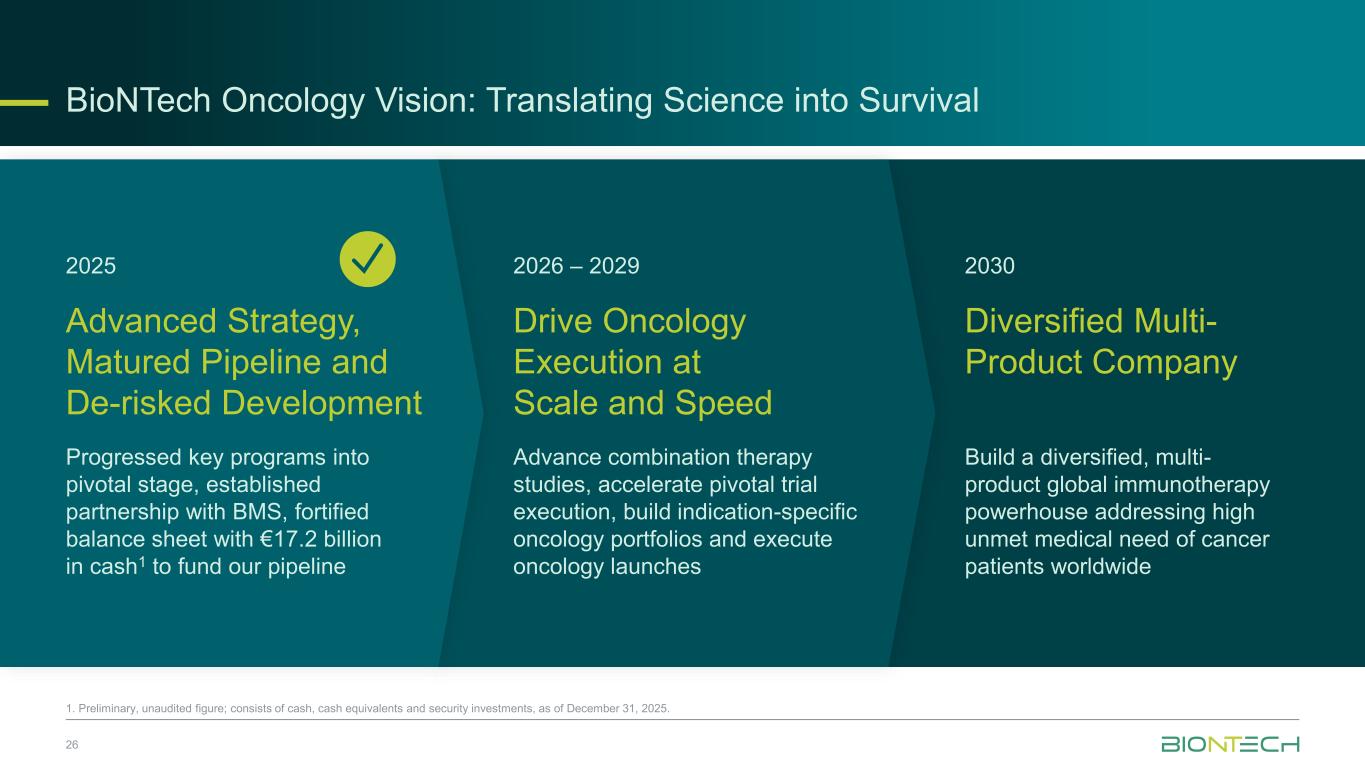
2025 2026 – 2029 2030 Advanced Strategy, Matured Pipeline and De-risked Development Drive Oncology Execution at Scale and Speed Diversified Multi- Product Company Progressed key programs into pivotal stage, established partnership with BMS, fortified balance sheet with €17.2 billion in cash1 to fund our pipeline Advance combination therapy studies, accelerate pivotal trial execution, build indication-specific oncology portfolios and execute oncology launches Build a diversified, multi- product global immunotherapy powerhouse addressing high unmet medical need of cancer patients worldwide BioNTech Oncology Vision: Translating Science into Survival 26 1. Preliminary, unaudited figure; consists of cash, cash equivalents and security investments, as of December 31, 2025. i l i i : r l ti i i t r i l

Thank you

Appendix
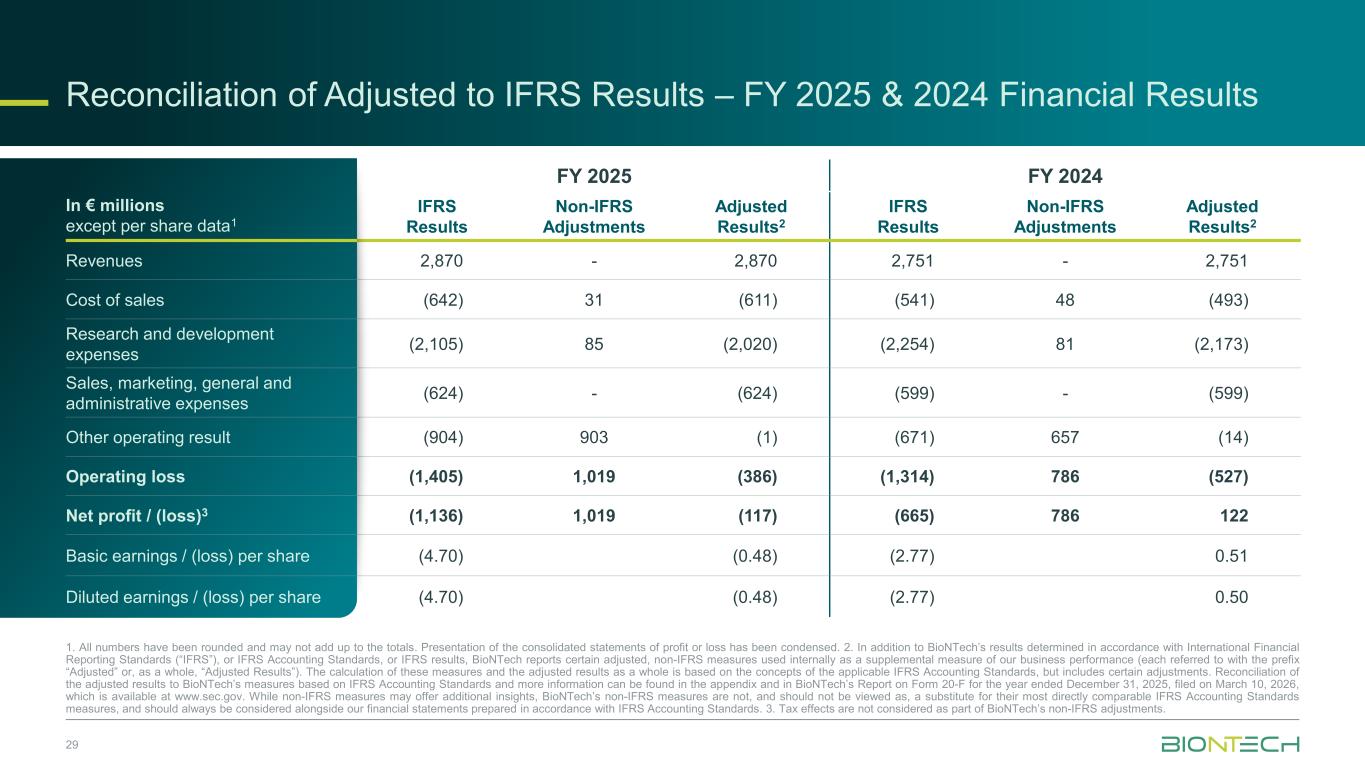
Reconciliation of Adjusted to IFRS Results – FY 2025 & 2024 Financial Results 29 In € millions except per share data1 FY 2025 FY 2024 IFRS Results Non-IFRS Adjustments Adjusted Results2 IFRS Results Non-IFRS Adjustments Adjusted Results2 Revenues 2,870 - 2,870 2,751 - 2,751 Cost of sales (642) 31 (611) (541) 48 (493) Research and development expenses (2,105) 85 (2,020) (2,254) 81 (2,173) Sales, marketing, general and administrative expenses (624) - (624) (599) - (599) Other operating result (904) 903 (1) (671) 657 (14) Operating loss (1,405) 1,019 (386) (1,314) 786 (527) Net profit / (loss)3 (1,136) 1,019 (117) (665) 786 122 Basic earnings / (loss) per share (4.70) (0.48) (2.77) 0.51 Diluted earnings / (loss) per share (4.70) (0.48) (2.77) 0.50 1. All numbers have been rounded and may not add up to the totals. Presentation of the consolidated statements of profit or loss has been condensed. 2. In addition to BioNTech’s results determined in accordance with International Financial Reporting Standards (“IFRS”), or IFRS Accounting Standards, or IFRS results, BioNTech reports certain adjusted, non-IFRS measures used internally as a supplemental measure of our business performance (each referred to with the prefix “Adjusted” or, as a whole, “Adjusted Results”). The calculation of these measures and the adjusted results as a whole is based on the concepts of the applicable IFRS Accounting Standards, but includes certain adjustments. Reconciliation of the adjusted results to BioNTech’s measures based on IFRS Accounting Standards and more information can be found in the appendix and in BioNTech’s Report on Form 20-F for the year ended December 31, 2025, filed on March 10, 2026, which is available at www.sec.gov. While non-IFRS measures may offer additional insights, BioNTech’s non-IFRS measures are not, and should not be viewed as, a substitute for their most directly comparable IFRS Accounting Standards measures, and should always be considered alongside our financial statements prepared in accordance with IFRS Accounting Standards. 3. Tax effects are not considered as part of BioNTech’s non-IFRS adjustments.
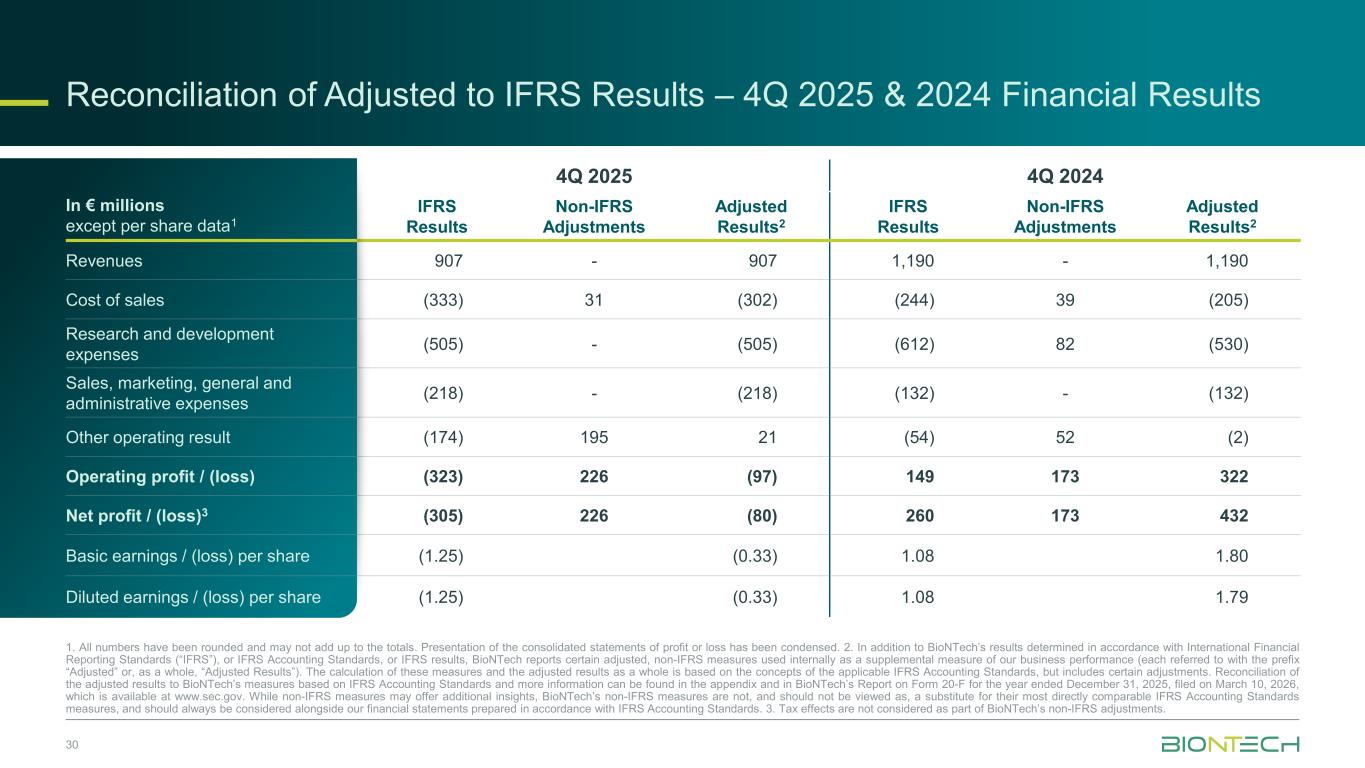
Reconciliation of Adjusted to IFRS Results – 4Q 2025 & 2024 Financial Results 30 In € millions except per share data1 4Q 2025 4Q 2024 IFRS Results Non-IFRS Adjustments Adjusted Results2 IFRS Results Non-IFRS Adjustments Adjusted Results2 Revenues 907 - 907 1,190 - 1,190 Cost of sales (333) 31 (302) (244) 39 (205) Research and development expenses (505) - (505) (612) 82 (530) Sales, marketing, general and administrative expenses (218) - (218) (132) - (132) Other operating result (174) 195 21 (54) 52 (2) Operating profit / (loss) (323) 226 (97) 149 173 322 Net profit / (loss)3 (305) 226 (80) 260 173 432 Basic earnings / (loss) per share (1.25) (0.33) 1.08 1.80 Diluted earnings / (loss) per share (1.25) (0.33) 1.08 1.79 1. All numbers have been rounded and may not add up to the totals. Presentation of the consolidated statements of profit or loss has been condensed. 2. In addition to BioNTech’s results determined in accordance with International Financial Reporting Standards (“IFRS”), or IFRS Accounting Standards, or IFRS results, BioNTech reports certain adjusted, non-IFRS measures used internally as a supplemental measure of our business performance (each referred to with the prefix “Adjusted” or, as a whole, “Adjusted Results”). The calculation of these measures and the adjusted results as a whole is based on the concepts of the applicable IFRS Accounting Standards, but includes certain adjustments. Reconciliation of the adjusted results to BioNTech’s measures based on IFRS Accounting Standards and more information can be found in the appendix and in BioNTech’s Report on Form 20-F for the year ended December 31, 2025, filed on March 10, 2026, which is available at www.sec.gov. While non-IFRS measures may offer additional insights, BioNTech’s non-IFRS measures are not, and should not be viewed as, a substitute for their most directly comparable IFRS Accounting Standards measures, and should always be considered alongside our financial statements prepared in accordance with IFRS Accounting Standards. 3. Tax effects are not considered as part of BioNTech’s non-IFRS adjustments.
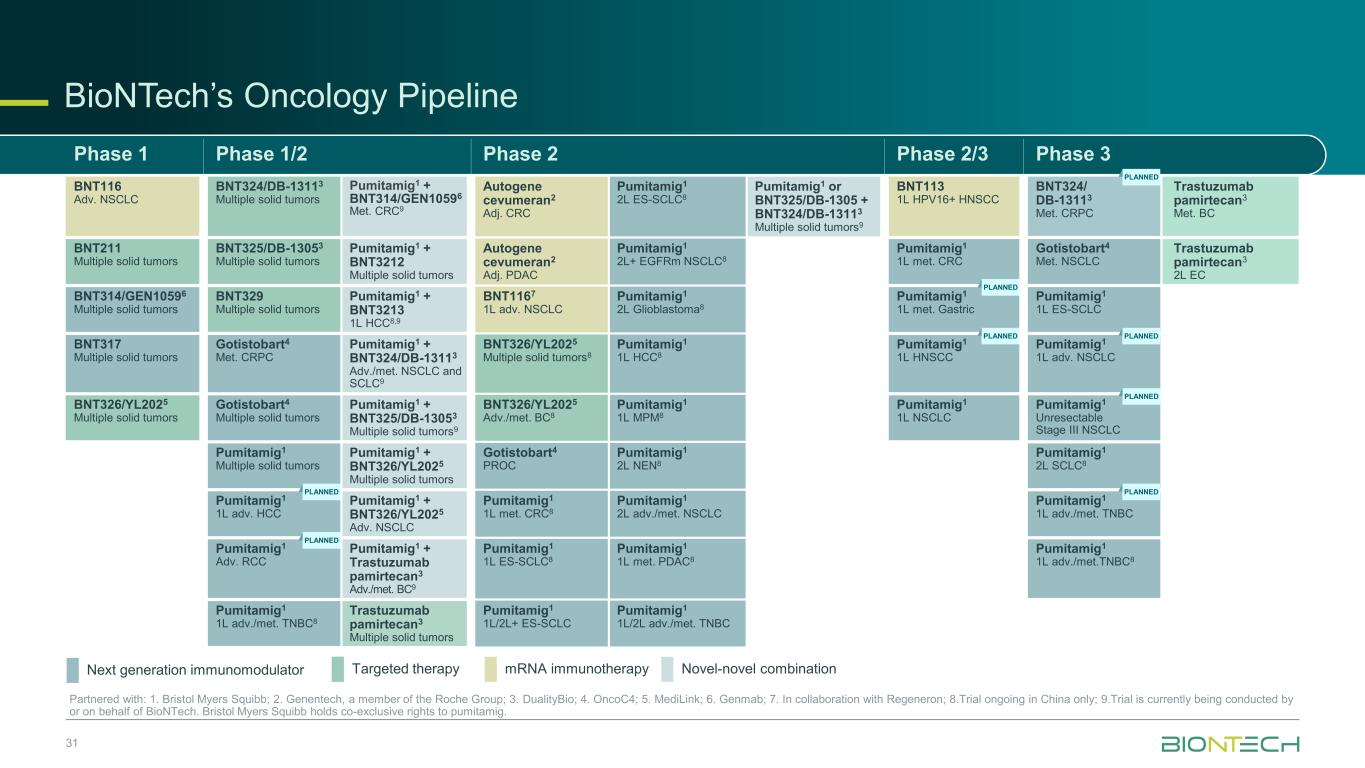
BioNTech’s Oncology Pipeline 31 Partnered with: 1. Bristol Myers Squibb; 2. Genentech, a member of the Roche Group; 3. DualityBio; 4. OncoC4; 5. MediLink; 6. Genmab; 7. In collaboration with Regeneron; 8.Trial ongoing in China only; 9.Trial is currently being conducted by or on behalf of BioNTech. Bristol Myers Squibb holds co-exclusive rights to pumitamig. Phase 1 Phase 1/2 Phase 2 Phase 2/3 Phase 3 BNT116 Adv. NSCLC BNT324/DB-13113 Multiple solid tumors Pumitamig1 + BNT314/GEN10596 Met. CRC9 Autogene cevumeran2 Adj. CRC Pumitamig1 2L ES-SCLC8 Pumitamig1 or BNT325/DB-1305 + BNT324/DB-13113 Multiple solid tumors9 BNT113 1L HPV16+ HNSCC BNT324/ DB-13113 Met. CRPC Trastuzumab pamirtecan3 Met. BC BNT211 Multiple solid tumors BNT325/DB-13053 Multiple solid tumors Pumitamig1 + BNT3212 Multiple solid tumors Autogene cevumeran2 Adj. PDAC Pumitamig1 2L+ EGFRm NSCLC8 Pumitamig1 1L met. CRC Gotistobart4 Met. NSCLC Trastuzumab pamirtecan3 2L EC BNT314/GEN10596 Multiple solid tumors BNT329 Multiple solid tumors Pumitamig1 + BNT3213 1L HCC8,9 BNT1167 1L adv. NSCLC Pumitamig1 2L Glioblastoma8 Pumitamig1 1L met. Gastric Pumitamig1 1L ES-SCLC BNT317 Multiple solid tumors Gotistobart4 Met. CRPC Pumitamig1 + BNT324/DB-13113 Adv./met. NSCLC and SCLC9 BNT326/YL2025 Multiple solid tumors8 Pumitamig1 1L HCC8 Pumitamig1 1L HNSCC Pumitamig1 1L adv. NSCLC BNT326/YL2025 Multiple solid tumors Gotistobart4 Multiple solid tumors Pumitamig1 + BNT325/DB-13053 Multiple solid tumors9 BNT326/YL2025 Adv./met. BC8 Pumitamig1 1L MPM8 Pumitamig1 1L NSCLC Pumitamig1 Unresectable Stage III NSCLC Pumitamig1 Multiple solid tumors Pumitamig1 + BNT326/YL2025 Multiple solid tumors Gotistobart4 PROC Pumitamig1 2L NEN8 Pumitamig1 2L SCLC8 Pumitamig1 1L adv. HCC Pumitamig1 + BNT326/YL2025 Adv. NSCLC Pumitamig1 1L met. CRC8 Pumitamig1 2L adv./met. NSCLC Pumitamig1 1L adv./met. TNBC Pumitamig1 Adv. RCC Pumitamig1 + Trastuzumab pamirtecan3 Adv./met. BC9 Pumitamig1 1L ES-SCLC8 Pumitamig1 1L met. PDAC8 Pumitamig1 1L adv./met.TNBC8 Pumitamig1 1L adv./met. TNBC8 Trastuzumab pamirtecan3 Multiple solid tumors Pumitamig1 1L/2L+ ES-SCLC Pumitamig1 1L/2L adv./met. TNBC mRNA immunotherapyNext generation immunomodulator Targeted therapy Novel-novel combination PLANNED PLANNED PLANNED PLANNED PLANNED PLANNED PLANNED PLANNED
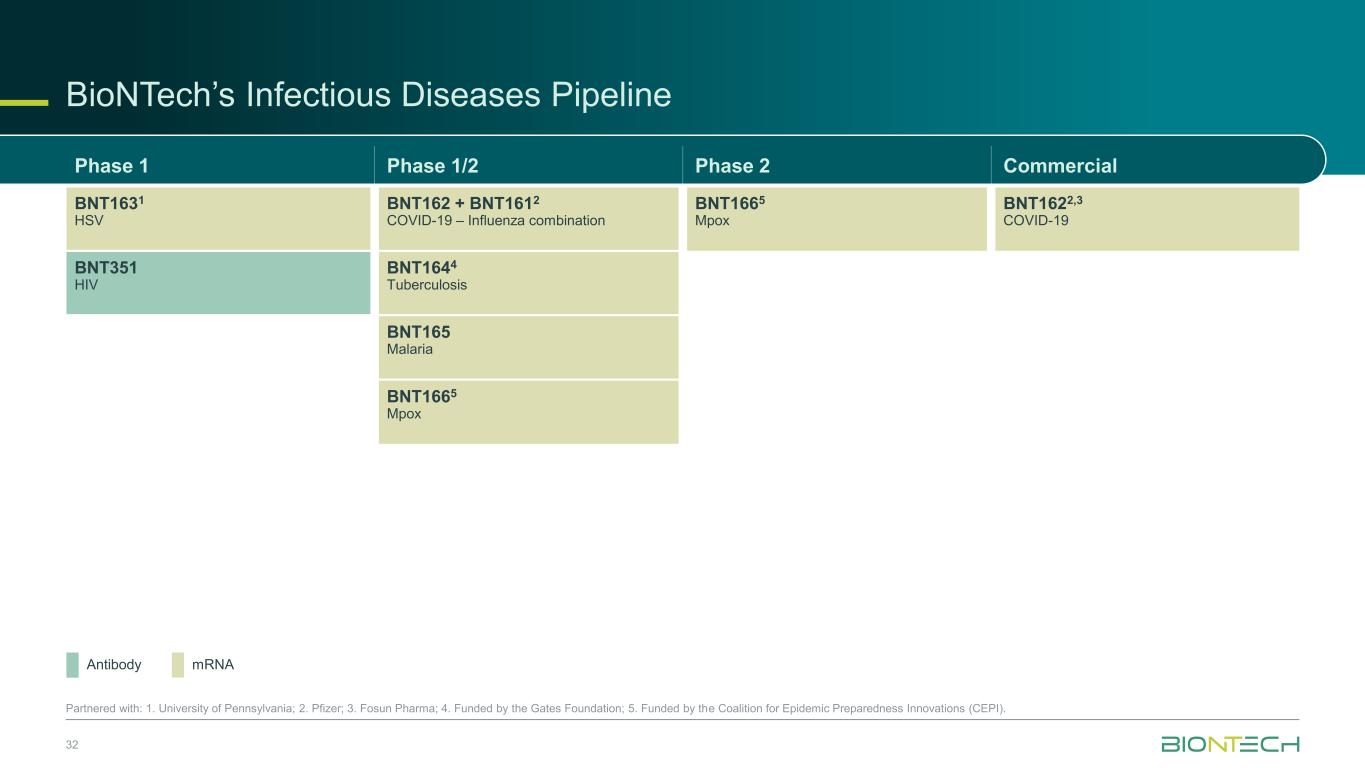
32 Partnered with: 1. University of Pennsylvania; 2. Pfizer; 3. Fosun Pharma; 4. Funded by the Gates Foundation; 5. Funded by the Coalition for Epidemic Preparedness Innovations (CEPI). mRNAAntibody BioNTech’s Infectious Diseases Pipeline Phase 1 Phase 1/2 Phase 2 Commercial BNT1631 HSV BNT162 + BNT1612 COVID-19 – Influenza combination BNT1665 Mpox BNT1622,3 COVID-19 BNT351 HIV BNT1644 Tuberculosis BNT165 Malaria BNT1665 Mpox
33 4-1BB CD137 G7 markets Canada, France, Germany, Italy, Japan, GB, USA (sq) NSCLC (squamous) Non-small cell lung cancer n L nth line GB Great Britain OC Ovarian cancer AACR American Association for Cancer Research GBM Glioblastoma PCD Projected Commercialization Date ADC Antibody-drug conjugate GC/GEJ Gastric/Gastro-esophageal junction cancer PD-(L)1 Programmed cell death protein (ligand) 1 adj. Adjuvant HCC Hepatocellular carcinoma PDAC Pancreatic ductal adenocarcinoma adv. Advanced HER2 (or 3) Human epidermal growth factor receptor 2 (or 3) PoC Proof of concept AGA Actionable oncogenic alteration HIV Human immunodeficiency virus PROC Platinum-resistant ovarian cancer ASCO American Society of Clinical Oncology HNC Head and neck cancer PVRIG Poliovirus receptor-related immunoglobulin B7-H3 B7 Homolog 3 HNSCC Head and neck squamous cell carcinoma R&D Research and development BC Breast cancer HPV Human papilloma virus (ncc/cc)RCC ((non-)clear cell) Renal cell carcinoma BLA Biologics License Applications HR Hormone receptor SABCS San Antonio Breast Cancer Symposium BMS Bristol Myers Squibb HSV Herpes simplex virus (ES)SCLC (Extensive stage) small cell lung cancer CPS Combined positive score IFRS International financial reporting standards SEC U.S. Securities and Exchange Commission CRC Colorectal cancer IIT Investigator initiated trial SG&A Selling, general and administrative expenses (m)CRPC (met.) Castration resistant prostate cancer iNeST Individualized NeoAntigen-Specific Therapy SITC Society of Immunotherapy of Cancer CRT Chemoradiation therapy IO Immuno-oncology SoC Standard of care ctDNA Circulating tumor DNA M&A Merger and acquisitions TIGIT T cell immunoreceptor with Ig and ITIM domains EC Endometrial cancer met. Metastatic TM Trademark EGFR(m) (mutated) Epidermal growth factor receptor MIUC Muscle-invasive urothelial carcinoma TNBC Triple-negative breast cancer EpCAM Epithelial cell adhesion molecule MPM Malignant pleural mesothelioma T-Pam Trastuzumab pamirtecan ESMO European Society for Medical Oncology Mpox Monkeypox TROP2 Trophoblast cell-surface antigen 2 EU4(5) Germany, France, Italy, Spain, (UK) mRNA Messenger ribonucleic acid U.S. United States FixVac Fixed Antigen Vaccine MSS Microsatellite stability UK United Kingdom FY Fiscal year NEN Neuroendocrine neoplasm WCLC World Conference of Lung Cancer Abbreviation Directory