| 1 | © 2026 Aprea Therapeutics, Inc. All Rights Reserved A clinical-stage precision medicine oncology company focused on the discovery and development of targeted therapies for patients with biomarker-defined cancers March 2026 |
| 2 | © 2026 Aprea Therapeutics, Inc. All Rights Reserved Forward-Looking Statements Certain information contained in this presentation includes “forward-looking statements”, within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended related to our study analyses, clinical trials, regulatory submissions, and projected cash position. We may, in some cases use terms such as “future,” “predicts,” “believes,” “potential,” “continue,” “anticipates,” “estimates,” “expects,” “plans,” “intends,” “targeting,” “confidence,” “may,” “could,” “might,” “likely,” “will,” “should” or other words that convey uncertainty of the future events or outcomes to identify these forward-looking statements. Our forward-looking statements are based on current beliefs and expectations of our management team and on information currently available to management that involve risks, potential changes in circumstances, assumptions, and uncertainties. All statements contained in this presentation other than statements of historical fact are forward-looking statements, including statements regarding our ability to develop, commercialize, and achieve market acceptance of our current and planned products and services, our research and development efforts, including timing considerations and other matters regarding our business strategies, use of capital, results of operations and financial position, and plans and objectives for future operations. Any or all of the forward-looking statements may turn out to be wrong or be affected by inaccurate assumptions we might make or by known or unknown risks and uncertainties. These forward-looking statements are subject to risks and uncertainties including, without limitation, risks related to the success, timing, and cost of our ongoing clinical trials and anticipated clinical trials for our current product candidates, including statements regarding the timing of initiation, pace of enrollment and completion of the trials (including our ability to fully fund our disclosed clinical trials, which assumes no material changes to our currently projected expenses), futility analyses, presentations at conferences and data reported in an abstract, and receipt of interim or preliminary results (including, without limitation, any preclinical results or data), which are not necessarily indicative of the final results of our ongoing clinical trials, our understanding of product candidates mechanisms of action and interpretation of preclinical and early clinical results from its clinical development programs and our ability to predict clinical outcomes based on such preclinical and early clinical result, and our ability to continue as a going concern, and the other risks, uncertainties, and other factors described under “Risk Factors,” “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and elsewhere in the documents we file with the U.S. Securities and Exchange Commission. For all these reasons, actual results and developments could be materially different from those expressed in or implied by our forward-looking statements. You are cautioned not to place undue reliance on these forward-looking statements, which are made only as of the date of this presentation. We undertake no obligation to update such forward-looking statements for any reason, except as required by law. This presentation shall not constitute an offer to sell or the solicitation of an offer to buy any securities, nor shall there be any sale of any securities in any state or jurisdiction in which such offer, solicitation or sale would be unlawful prior to registration or qualification under the securities laws of any such state or jurisdiction. This presentation may not be reproduced, forwarded to any person or published, in whole or in part. |
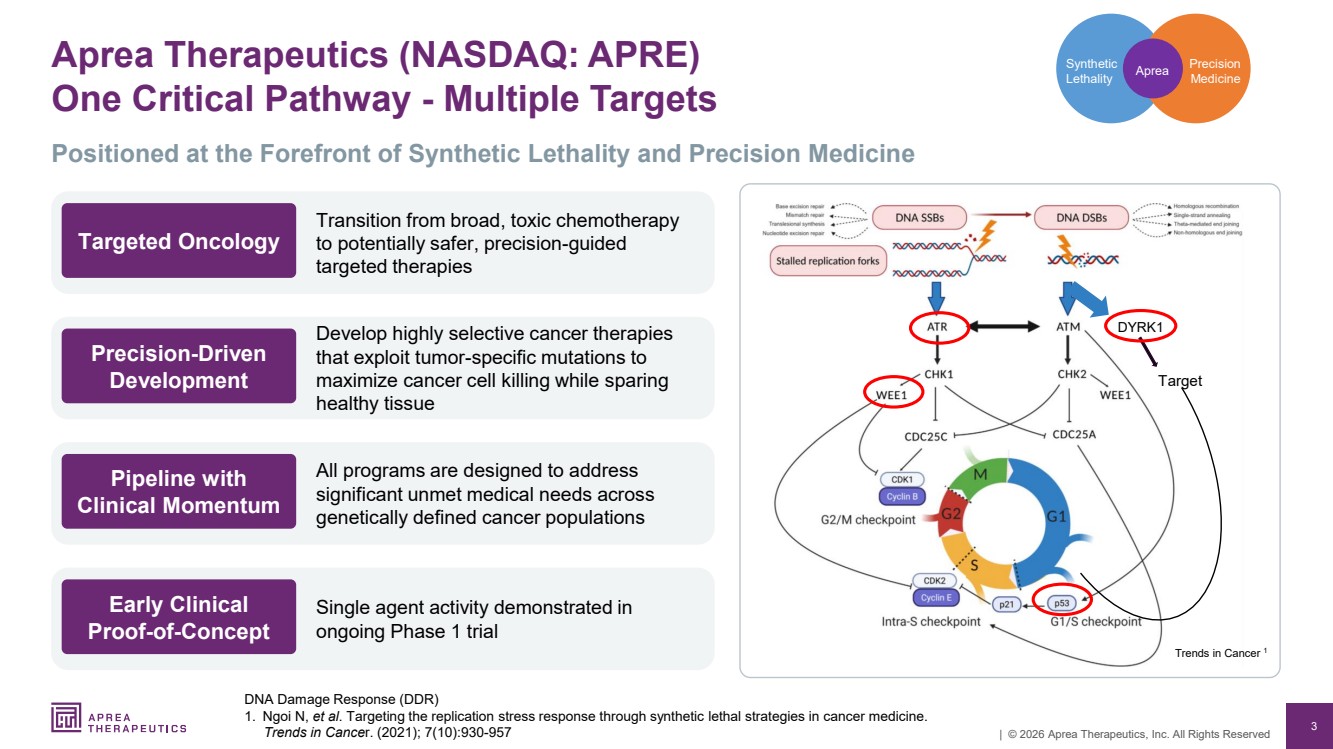
| 3 | © 2026 Aprea Therapeutics, Inc. All Rights Reserved Aprea Therapeutics (NASDAQ: APRE) One Critical Pathway - Multiple Targets DNA Damage Response (DDR) 1. Ngoi N, et al. Targeting the replication stress response through synthetic lethal strategies in cancer medicine. Trends in Cancer. (2021); 7(10):930-957 Precision Medicine Synthetic Lethality Aprea DYRK1 Target Trends in Cancer 1 Positioned at the Forefront of Synthetic Lethality and Precision Medicine Transition from broad, toxic chemotherapy to potentially safer, precision-guided targeted therapies Targeted Oncology Develop highly selective cancer therapies that exploit tumor-specific mutations to maximize cancer cell killing while sparing healthy tissue Precision-Driven Development All programs are designed to address significant unmet medical needs across genetically defined cancer populations Pipeline with Clinical Momentum Single agent activity demonstrated in ongoing Phase 1 trial Early Clinical Proof-of-Concept |
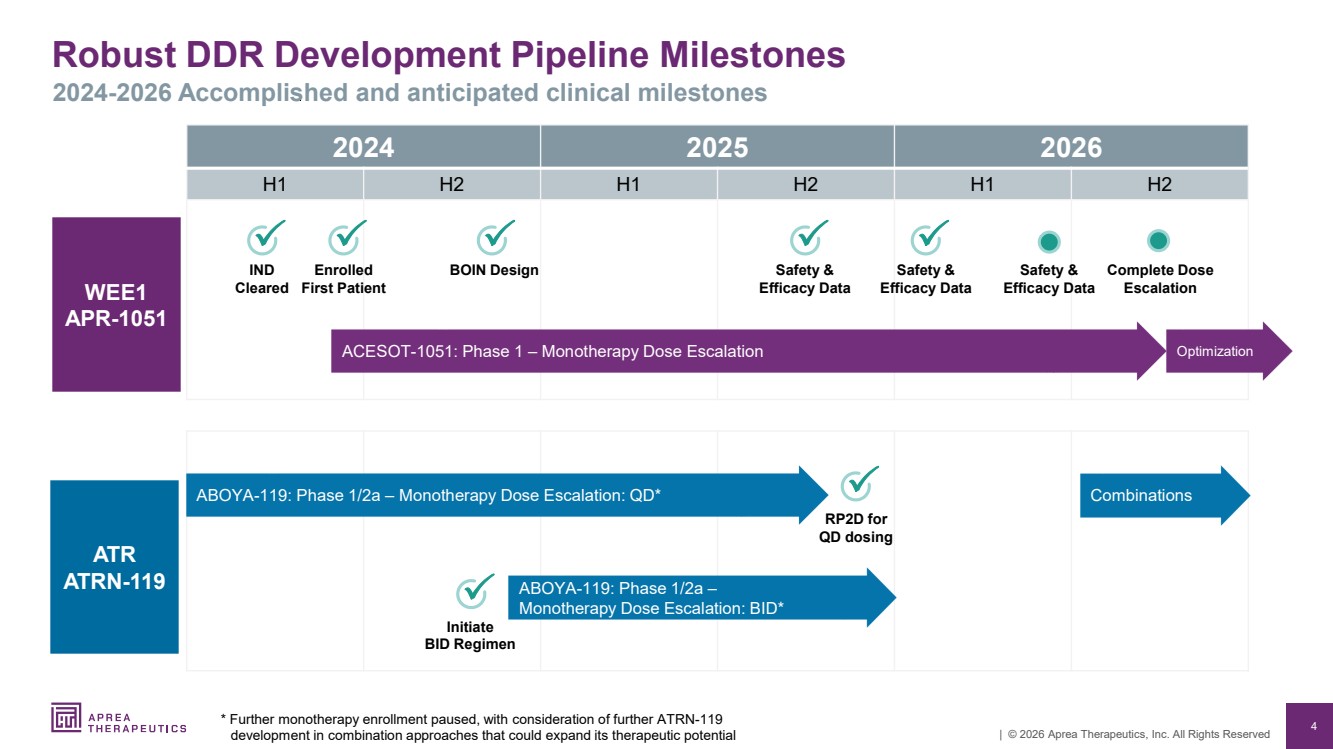
| 4 | © 2026 Aprea Therapeutics, Inc. All Rights Reserved Robust DDR Development Pipeline Milestones 2024-2026 Accomplished and anticipated clinical milestones 2024 2025 2026 H1 H2 H1 H2 H1 H2 ATR ATRN-119 WEE1 APR-1051 RP2D for QD dosing ACESOT-1051: Phase 1 – Monotherapy Dose Escalation Enrolled First Patient Complete Dose Escalation IND Cleared ABOYA-119: Phase 1/2a – Monotherapy Dose Escalation: QD* BOIN Design Initiate BID Regimen Optimization ABOYA-119: Phase 1/2a – Monotherapy Dose Escalation: BID* * Further monotherapy enrollment paused, with consideration of further ATRN-119 development in combination approaches that could expand its therapeutic potential Safety & Efficacy Data Safety & Efficacy Data Combinations Safety & Efficacy Data |
| 5 | © 2026 Aprea Therapeutics, Inc. All Rights Reserved Strong Drug Development and Commercial Expertise Experienced team in synthetic lethality and targeted therapy Management Board of Directors Richard Peters, M.D., Ph.D. Chairman of the Board Oren Gilad, Ph.D. President and CEO Jean-Pierre Bizzari, M.D. Director Marc Duey Director Michael Grissinger Director Gabriela Gruia, M.D. Director John Henneman Director Rifat Pamukcu, M.D. Director Bernd R. Seizinger, M.D., Ph.D. Director Oren Gilad, Ph.D. President and CEO John P. Hamill Sr. Vice President and CFO Eugene Kennedy, MD Chief Medical Advisor Ze’ev Weiss, CPA, B.Sc. Chief Business Advisor Mike Carleton, Ph.D. Translational Medicine Advisor Brian Wiley SVP, Corporate Strategy |
| 6 | © 2026 Aprea Therapeutics, Inc. All Rights Reserved 6 WEE1 Inhibitor: APR-1051 ACESOT-1051: Clinical Proof-Of-Concept |
| 7 | © 2026 Aprea Therapeutics, Inc. All Rights Reserved APR-1051: Next–Generation WEE1 Inhibitor Expanding the Therapeutic Index 1. Zentalis Corporate Presentation, January 2026 2. Debio 0123-101, A Phase 1 Trial of Debio 0123 In Combination With Carboplatin In Advanced Solid Tumors: Safety, Pharmacokinetic, And Preliminary Antitumor Activity Data, Poster ASCO 2023 3. Results of A Phase 1, Dose-Finding Study of Debio 0123 As Monotherapy In Adult Patients With Advanced Solid Tumors —Safety, Pharmacokinetic, And Preliminary Antitumor Activity Data, Poster ASCO 2024 Targeting the clinically validated WEE1 pathway with an engineered profile aimed at widening the therapeutic window and improving tolerability versus earlier inhibitors Program Clinical Limitation Strategic Outcome What It Signals Adavosertib (AstraZeneca) Hematologic & GI toxicity limited dose intensity Terminated further clinical development Returned by AstraZeneca to Merck & Co. Biology works, narrow therapeutic window Azenosertib (Zentalis) Continuous dosing not tolerated1 Ongoing dosing and schedule optimization Biology works, therapeutic window still being defined Debio 0123 (Debiopharm) QT prolongation liability at high doses2 Limited single-agent activity – no clinical responses at doses up to and including at MTD3 Cardiac safety restricts exposure APR-1051 (Aprea Therapeutics) Early signals of tumor reduction without class-limiting tox to date Dose escalation progressing Potentially widened therapeutic window No head-to-head studies have been conducted. Trial information is based on publicly available data and should be interpreted cautiously. |
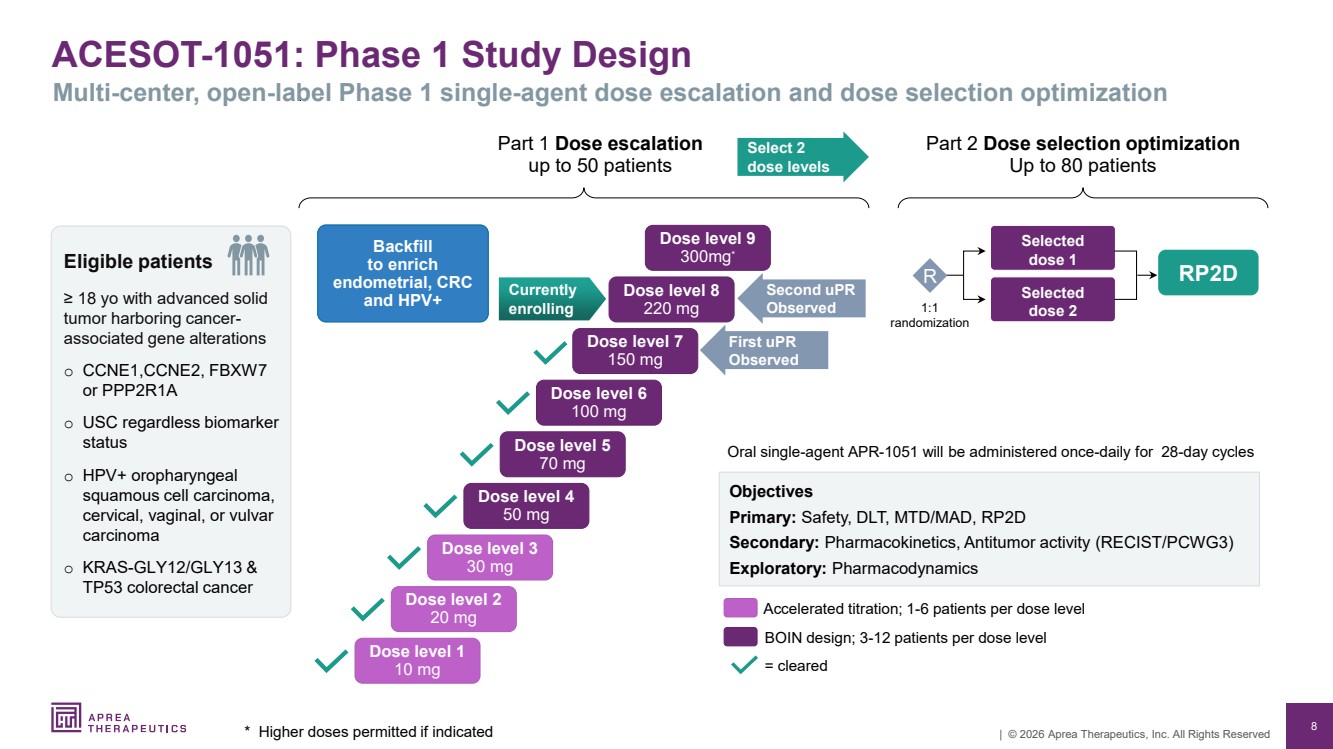
| 8 | © 2026 Aprea Therapeutics, Inc. All Rights Reserved ACESOT-1051: Phase 1 Study Design * Higher doses permitted if indicated Multi-center, open-label Phase 1 single-agent dose escalation and dose selection optimization Part 1 Dose escalation up to 50 patients RP2D Oral single-agent APR-1051 will be administered once-daily for 28-day cycles Objectives Primary: Safety, DLT, MTD/MAD, RP2D Secondary: Pharmacokinetics, Antitumor activity (RECIST/PCWG3) Exploratory: Pharmacodynamics = cleared Accelerated titration; 1-6 patients per dose level BOIN design; 3-12 patients per dose level Part 2 Dose selection optimization Up to 80 patients Selected dose 2 Selected dose 1 1:1 randomization R Currently enrolling Dose level 1 10 mg Dose level 2 20 mg Dose level 3 30 mg Dose level 4 50 mg Dose level 5 70 mg Dose level 6 100 mg Dose level 7 150 mg Dose level 8 220 mg Dose level 9 300mg* Backfill to enrich endometrial, CRC and HPV+ Second uPR Observed First uPR Observed Select 2 dose levels Eligible patients ≥ 18 yo with advanced solid tumor harboring cancer-associated gene alterations o CCNE1,CCNE2, FBXW7 or PPP2R1A o USC regardless biomarker status o HPV+ oropharyngeal squamous cell carcinoma, cervical, vaginal, or vulvar carcinoma o KRAS-GLY12/GLY13 & TP53 colorectal cancer |
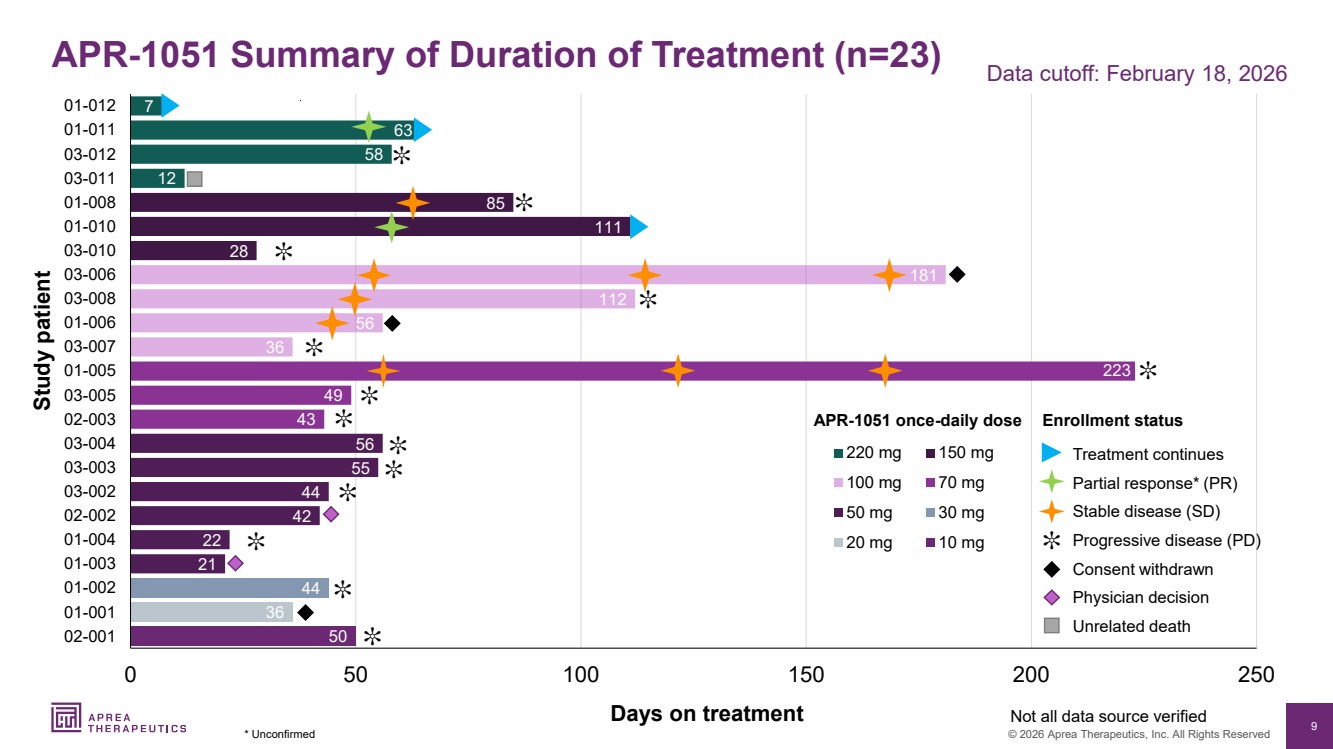
| 9 © 2026 Aprea Therapeutics, Inc. All Rights Reserved 50 36 44 21 22 42 44 55 56 43 49 223 36 56 112 181 28 111 85 12 58 63 7 0 50 100 150 200 250 02-001 01-001 01-002 01-003 01-004 02-002 03-002 03-003 03-004 02-003 03-005 01-005 03-007 01-006 03-008 03-006 03-010 01-010 01-008 03-011 03-012 01-011 01-012 220 mg 150 mg 100 mg 70 mg 50 mg 30 mg 20 mg 10 mg Days on treatment Study patient APR-1051 once-daily dose Not all data source verified ✼ Data cutoff: February 18, 2026 ✼ Progressive disease (PD) Stable disease (SD) Consent withdrawn Treatment continues Physician decision Unrelated death Partial response* (PR) APR-1051 Summary of Duration of Treatment (n=23) * Unconfirmed ✼ ✼ ✼ ✼ ✼ ✼ ✼ ✼ ✼ ✼ ✼ ✼ ✼ Enrollment status |
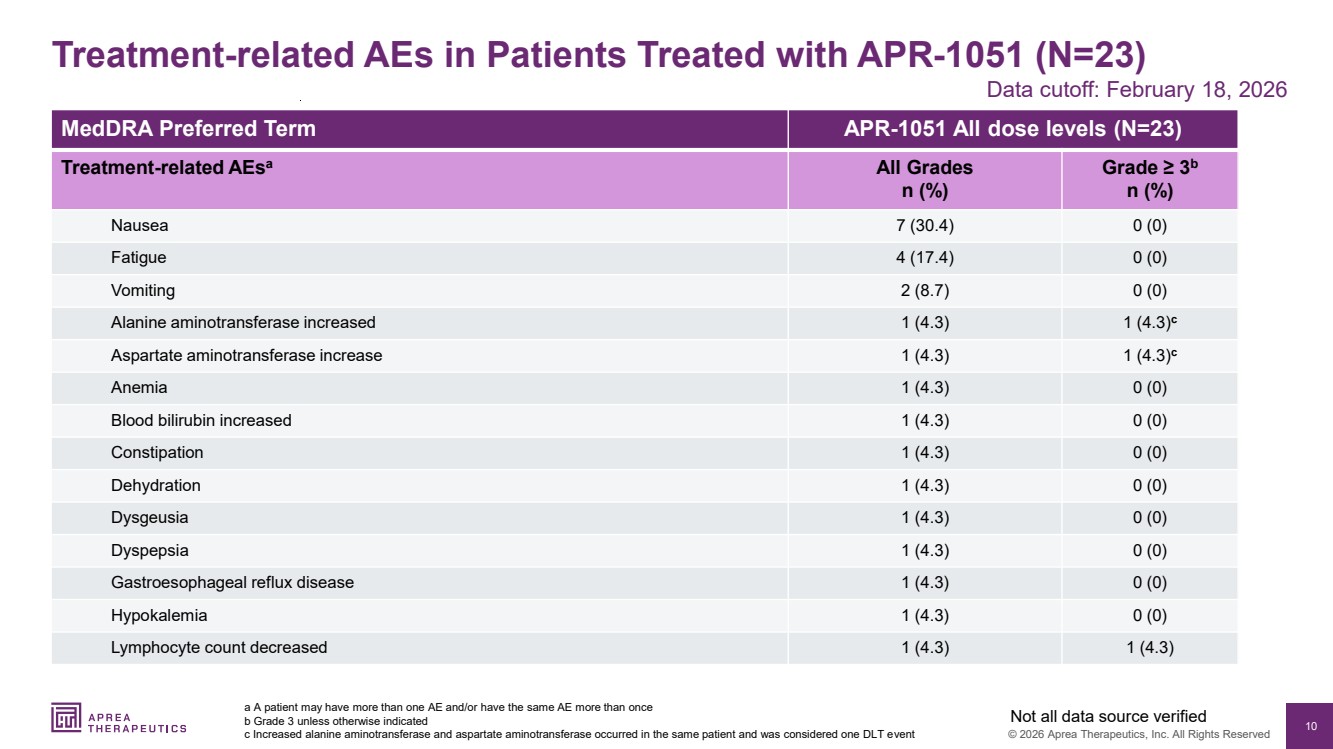
| 10 © 2026 Aprea Therapeutics, Inc. All Rights Reserved MedDRA Preferred Term APR-1051 All dose levels (N=23) Treatment-related AEsa All Grades n (%) Grade ≥ 3b n (%) Nausea 7 (30.4) 0 (0) Fatigue 4 (17.4) 0 (0) Vomiting 2 (8.7) 0 (0) Alanine aminotransferase increased 1 (4.3) 1 (4.3)c Aspartate aminotransferase increase 1 (4.3) 1 (4.3)c Anemia 1 (4.3) 0 (0) Blood bilirubin increased 1 (4.3) 0 (0) Constipation 1 (4.3) 0 (0) Dehydration 1 (4.3) 0 (0) Dysgeusia 1 (4.3) 0 (0) Dyspepsia 1 (4.3) 0 (0) Gastroesophageal reflux disease 1 (4.3) 0 (0) Hypokalemia 1 (4.3) 0 (0) Lymphocyte count decreased 1 (4.3) 1 (4.3) Treatment-related AEs in Patients Treated with APR-1051 (N=23) a A patient may have more than one AE and/or have the same AE more than once b Grade 3 unless otherwise indicated c Increased alanine aminotransferase and aspartate aminotransferase occurred in the same patient and was considered one DLT event Not all data source verified Data cutoff: February 18, 2026 |
| 11 | © 2026 Aprea Therapeutics, Inc. All Rights Reserved Patient Example #1 (On Treatment): Partial Response Observed in Target Indication and Alteration APR-1051 shows single agent activity in cancer cells with mutated PPP2R1A Clinical Data 220 mg Cohort Patient: 63-year-old Female Diagnosis: Uterine Serous Carcinoma (form of endometrial cancer (EC)) Key Mutations: PPP2R1A Treatment History: 4 prior lines - heavily pretreated • Line 1: Carboplatin + Paclitaxel → 126 days, PD • Line 2: Pembrolizumab + Lenvatinib → ~17 months, PD • Line 3: Doxorubicin → 56 days, PD • Line 4: Topotecan → 70 days, PD APR-1051 Activity: • Current Status: On treatment • Best Response: uPR at first scan (-50% tumor response) Notes: uPR achieved with 50% tumor shrinkage and >85% reduction in CA-125 tumor marker in heavily pretreated 63-year-old patient. Well tolerated with minimal toxicity. PPP2R1A may be relevant to response. |
| 12 | © 2026 Aprea Therapeutics, Inc. All Rights Reserved Patient Example #2 (On Treatment): Partial Response Observed in Target Indication and Alteration Clinical Data 150 mg Cohort Patient: 68-year-old Female Diagnosis: Uterine Serous Carcinoma (form of EC) Key Mutations: PPP2R1A Treatment History: 4 prior lines - heavily pretreated • Line 1: Paclitaxel/carboplatin → 105 days, CR (Completed Regimen) • Line 2: Docetaxel/carboplatin → 134 days, PR (Completed Regimen) • Line 3: Letrozole → 79 days, PD • Line 4: Investigational agent (ATK-160) → 55 days, PD APR-1051 Activity: • Current Status: On treatment • Best Response: uPR at first scan (-50% tumor response) Notes: uPR achieved with 50% tumor shrinkage and >90% reduction in CA-125 tumor marker in heavily pretreated 68-year-old patient. Well tolerated with minimal toxicity. PPP2R1A may be relevant to response. APR-1051 shows single agent activity in cancer cells with mutated PPP2R1A |
| 13 | © 2026 Aprea Therapeutics, Inc. All Rights Reserved Patient Example #3: Disease Control Observed in Early Patient Outcomes APR-1051 shows single agent activity in cancer cells with mutated FBXW7 Clinical Data 100 mg Cohort Patient: 86-year-old Female Diagnosis: Rectal Cancer Key Mutations: FBXW7 (Drives Cyclin E accumulation and overexpression) Treatment History: 5 prior lines - heavily pretreated • Line 1: Capecitabine/XRT → 191 days, PD • Line 2: Capecitabine/oxaliplatin/bevacizumab → 45 days, PD • Line 3: FOLFIRI + bevacizumab → 43 days, PD • Line 4: Local XRT (lung mets) → 12 days, not evaluable • Line 5: Tretinoin/bevacizumab/Tecentriq (ATRT trial) → 50 days, PD APR-1051 Activity: • Current Status: Consent withdrawn after 181 days • Best Response: SD at third scan (-15% tumor response) Notes: Durable SD maintained 181 days in a heavily pretreated 86-year-old patient; well tolerated with minimal toxicity. FBXW7 mutation may be relevant to response |
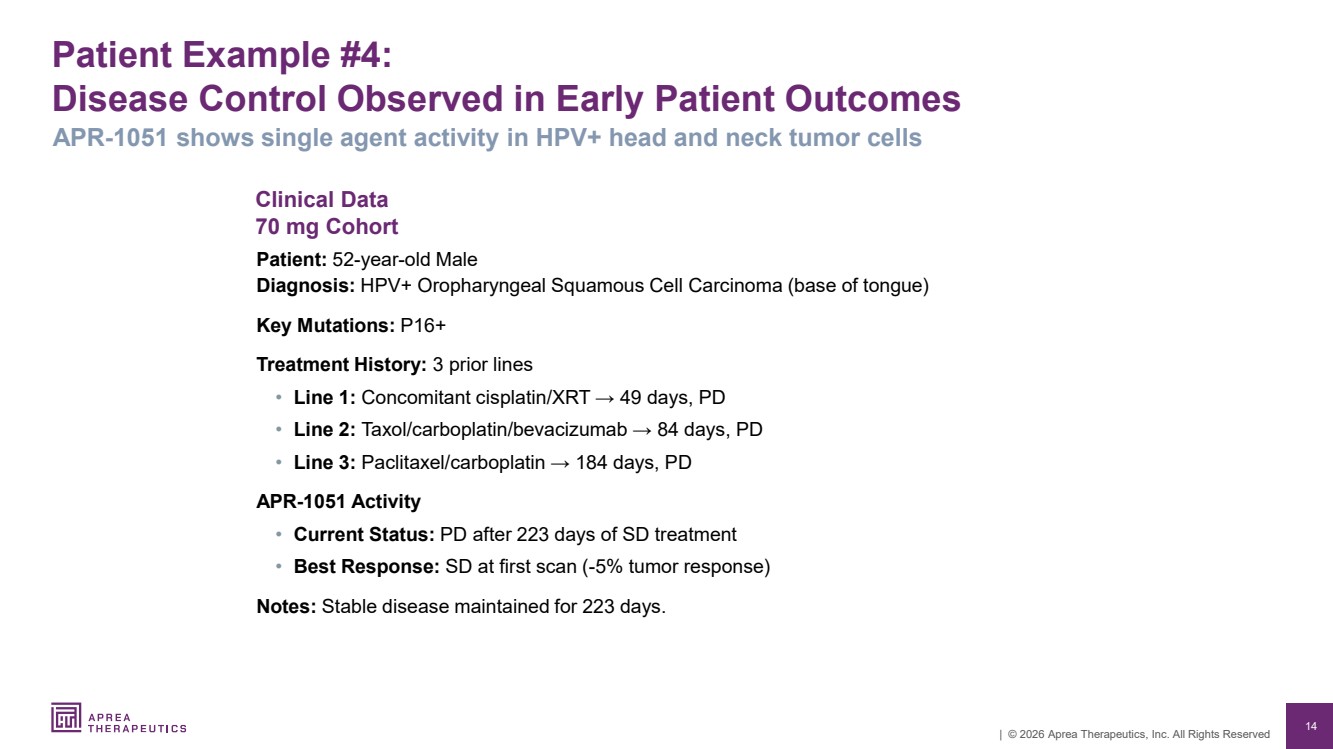
| 14 | © 2026 Aprea Therapeutics, Inc. All Rights Reserved Clinical Data 70 mg Cohort Patient Example #4: Disease Control Observed in Early Patient Outcomes Patient: 52-year-old Male Diagnosis: HPV+ Oropharyngeal Squamous Cell Carcinoma (base of tongue) Key Mutations: P16+ Treatment History: 3 prior lines • Line 1: Concomitant cisplatin/XRT → 49 days, PD • Line 2: Taxol/carboplatin/bevacizumab → 84 days, PD • Line 3: Paclitaxel/carboplatin → 184 days, PD APR-1051 Activity • Current Status: PD after 223 days of SD treatment • Best Response: SD at first scan (-5% tumor response) Notes: Stable disease maintained for 223 days. APR-1051 shows single agent activity in HPV+ head and neck tumor cells |
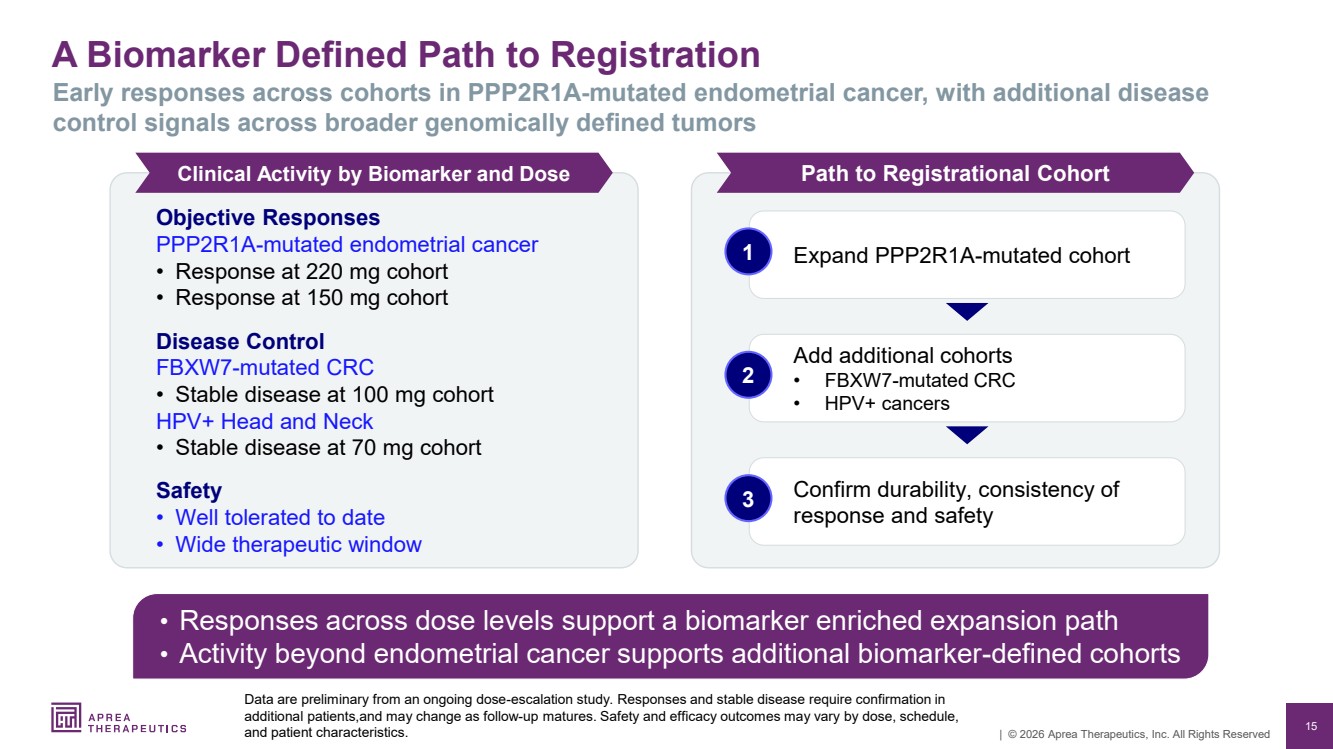
| 15 | © 2026 Aprea Therapeutics, Inc. All Rights Reserved A Biomarker Defined Path to Registration Data are preliminary from an ongoing dose-escalation study. Responses and stable disease require confirmation in additional patients,and may change as follow-up matures. Safety and efficacy outcomes may vary by dose, schedule, and patient characteristics. Early responses across cohorts in PPP2R1A-mutated endometrial cancer, with additional disease control signals across broader genomically defined tumors Objective Responses PPP2R1A-mutated endometrial cancer • Response at 220 mg cohort • Response at 150 mg cohort Disease Control FBXW7-mutated CRC • Stable disease at 100 mg cohort HPV+ Head and Neck • Stable disease at 70 mg cohort Safety • Well tolerated to date • Wide therapeutic window • Responses across dose levels support a biomarker enriched expansion path • Activity beyond endometrial cancer supports additional biomarker-defined cohorts Clinical Activity by Biomarker and Dose Path to Registrational Cohort 1 Expand PPP2R1A-mutated cohort Add additional cohorts • FBXW7-mutated CRC • HPV+ cancers 2 Confirm durability, consistency of response and safety 3 |
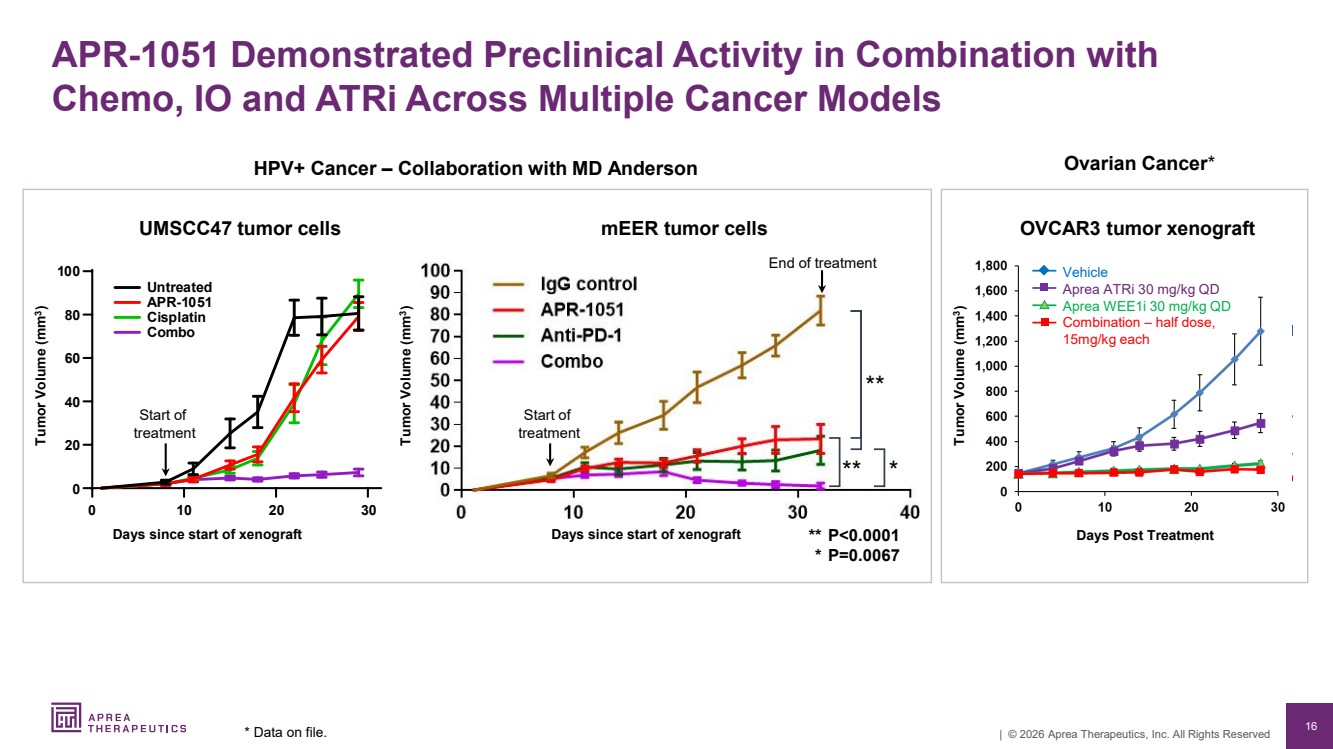
| 16 | © 2026 Aprea Therapeutics, Inc. All Rights Reserved HPV+ Cancer – Collaboration with MD Anderson Ovarian Cancer* UMSCC47 tumor cells OVCAR3 tumor xenograft Tumor Volume (mm3 ) Vehicle Aprea ATRi 30 mg/kg QD Aprea WEE1i 30 mg/kg QD Combination – half dose, 15mg/kg each Days Post Treatment Tumor Volume (mm3 ) Days since start of xenograft mEER tumor cells Tumor Volume (mm3 ) End of treatment Days since start of xenograft APR-1051 Demonstrated Preclinical Activity in Combination with Chemo, IO and ATRi Across Multiple Cancer Models * Data on file. Start of treatment 3 rea ed AP Cis a i C Start of treatment ** P<0.0001 * P=0.0067 ** ** * |
| 17 | © 2026 Aprea Therapeutics, Inc. All Rights Reserved Summary APR-1051 aims to be the first WEE1 inhibitor to translate validated biology into a scalable commercial asset 1 Clinically validated target • WEE1 inhibition has shown promising activity in genomically defined tumors APR-1051 opportunity • Early clinical proof-of-concept at 150 mg and 220 mg dose levels • Potentially favorable safety profile at active dose levels • Enrollment continues, additional clinical data expected over the next 3-6 months • Clinical team strengthened to drive next phase of development • Capital in place to achieve meaningful inflection points • Valuation lags fundamentals, creating an asymmetric opportunity Competitor programs constrained by low therapeutic index • Dose intensity and duration limited by hematologic, GI or cardiac toxicity 2 3 |
| 18 | © 2026 Aprea Therapeutics, Inc. All Rights Reserved 18 APR-1051: Potentially Differentiated WEE1 Inhibitor Pre-Clinical |
| 19 | © 2026 Aprea Therapeutics, Inc. All Rights Reserved APR-1051: Potentially Best-in-Class WEE1 Inhibitor Potent and structurally differentiated, with high selectivity to limit off-target toxicity AstraZeneca Adavosertib (AZD-1775) Zentalis Azenosertib (ZN-c3) Aprea APR-1051 Undisclosed |
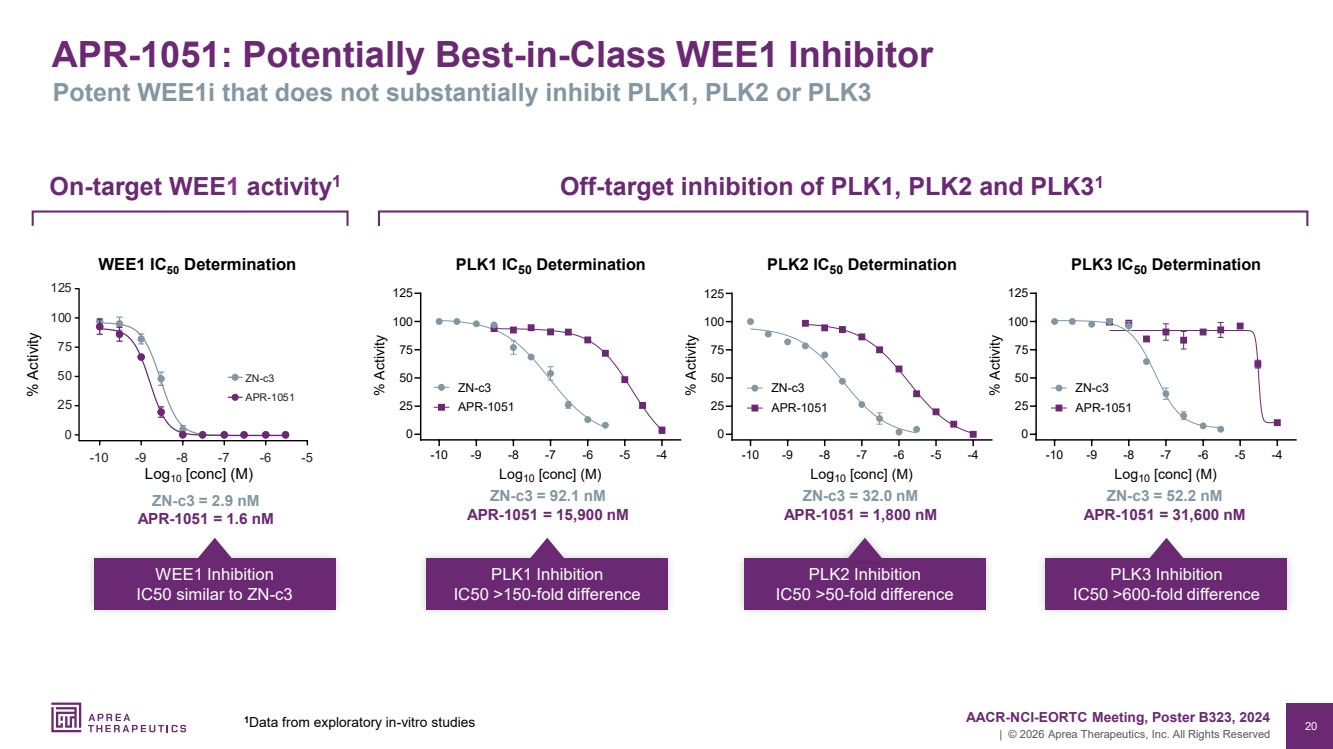
| 20 | © 2026 Aprea Therapeutics, Inc. All Rights Reserved APR-1051: Potentially Best-in-Class WEE1 Inhibitor Potent WEE1i that does not substantially inhibit PLK1, PLK2 or PLK3 -10 -9 -8 -7 -6 -5 -4 0 25 50 75 100 125 PLK1 IC50 Determination Log10 [conc] (M) % Activity ZN-c3 APR-1051 -10 -9 -8 -7 -6 -5 -4 0 25 50 75 100 125 PLK2 IC50 Determination Log10 [conc] (M) % Activity ZN-c3 APR-1051 -10 -9 -8 -7 -6 -5 -4 0 25 50 75 100 125 PLK3 IC50 Determination Log10 [conc] (M) % Activity ZN-c3 APR-1051 ZN-c3 = 92.1 nM APR-1051 = 15,900 nM PLK1 Inhibition IC50 >150-fold difference ZN-c3 = 32.0 nM APR-1051 = 1,800 nM PLK2 Inhibition IC50 >50-fold difference ZN-c3 = 52.2 nM APR-1051 = 31,600 nM PLK3 Inhibition IC50 >600-fold difference Off-target inhibition of PLK1, PLK2 and PLK3 On 1 -target WEE1 activity1 ZN-c3 = 2.9 nM APR-1051 = 1.6 nM WEE1 Inhibition IC50 similar to ZN-c3 125 100 75 50 25 0 10 9 8 7 6 5 APR 1051 c3 WEE1 IC50 Determination % Activity Log10 [conc] (M) 1 AACR-NCI-EORTC Meeting, Poster B323, 2024 Data from exploratory in-vitro studies |
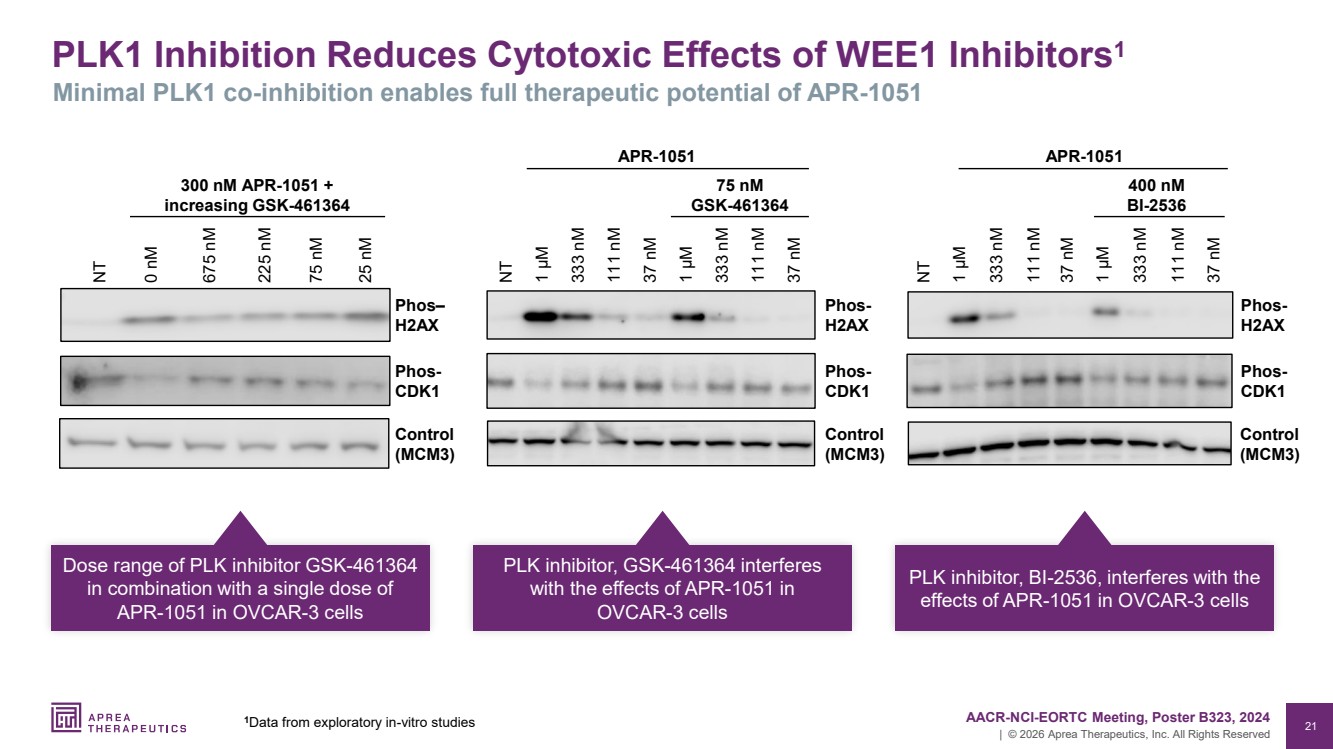
| 21 | © 2026 Aprea Therapeutics, Inc. All Rights Reserved PLK1 Inhibition Reduces Cytotoxic Effects of WEE1 Inhibitors1 Minimal PLK1 co-inhibition enables full therapeutic potential of APR-1051 37 nM 111 nM 333 nM 1 µM 37 nM 111 nM 333 nM 1 µM Phos-H2AX Control (MCM3) NT Phos-CDK1 APR-1051 75 nM GSK-461364 0 nM NT 675 nM 25 nM 225 nM 75 nM Phos– H2AX Control (MCM3) Phos-CDK1 300 nM APR-1051 + increasing GSK-461364 Control (MCM3) 37 nM 111 nM 333 nM 1 µM 37 nM 111 nM 333 nM NT 1 µM Phos-H2AX 400 nM BI-2536 APR-1051 Phos-CDK1 Dose range of PLK inhibitor GSK-461364 in combination with a single dose of APR-1051 in OVCAR-3 cells PLK inhibitor, GSK-461364 interferes with the effects of APR-1051 in OVCAR-3 cells PLK inhibitor, BI-2536, interferes with the effects of APR-1051 in OVCAR-3 cells 1 AACR-NCI-EORTC Meeting, Poster B323, 2024 Data from exploratory in-vitro studies |
| 22 | © 2026 Aprea Therapeutics, Inc. All Rights Reserved Studies Show PLK1 Suppression is Associated with Sepsis-Induced Loss of Intestinal Barrier Function 1 PLK1 protects against sepsis-induced intestinal barrier dysfunction, Cao et al, Scientific Reports (2018). 2 PLK1 protects intestinal barrier function in sepsis: A translational research, Cao et al, Cytokine (2023). 3 PLK1 protects intestinal barrier function in sepsis: A translational research, Cao et al, Molecular Medicine (2022). 4 LncRNA DANCR improves the dysfunction of the intestinal barrier and alleviates epithelial injury by targeting the miR‐1306‐5p/PLK1 axis in sepsis, Wang et al., Cell Biology International (2021). |
| 23 | © 2026 Aprea Therapeutics, Inc. All Rights Reserved 23 Intellectual Property Portfolio Financial Summary & Capitalization Investment Highlights |
| 24 | © 2026 Aprea Therapeutics, Inc. All Rights Reserved Robust Global Intellectual Property Protection Family 1: Ataxia Telangiectasia and Rad3-Related (ATR) Protein Kinase Inhibitors • Macrocyclic inhibitors of ATR & methods of using them to treat various cancers, filed on Oct. 13th, 2015 • Patents granted in AU, BR, CA, CN, EP, IL, IN, JP, KR, MX, HK. • 1.1: Issued on May 30, 2017 as U.S. Patent 9,663,535 • 1.2: Issued on May 29, 2018 as U.S. Patent 9,981,989 • 1.3: Issued on Feb. 5, 2019 as U.S. Patent 10,196,405 Family 2: ATR Inhibitors and Methods of Use • Carboxylic acid-containing macrocyclic ATR inhibitors, and prodrugs; methods of using these inhibitors to treat various cancers; filed on Apr. 12th, 2017 • Issued on May 28th , 2019 as U.S. Patent 10,301,324 Family 3: ATR Inhibitor Pharmaceutical Composition and Methods • International application filed on Apr. 14th, 2023 • Pharmaceutical formulation and composition of our lead ATR inhibitor in the clinic • Patent granted in JP; Applications pending US, AU, BR, CA, CN, EA, EP, HK, IL, IN, KR, MX, NZ, PH, SG, ZA Family 4: WEE1 Inhibitor Pharmaceutical Compositions and Methods • International Application filed on Jun. 3rd, 2022 • Composition of our lead WEE1 inhibitor compounds • Patent granted in AU; Applications pending in US, AU, BR, CA, CN, EP, HK, IL, IN, JP, KR, MX, ZA Family 5: Methods of Treating Cancer • International application filed on Sept. 19, 2026 • Clinical methods of treating advanced solid cancer tumors using lead ATR inhibitor Family 6: Macrocyclic Undisclosed DDR target Inhibitors and Methods of their Preparation and Use • International application filed on Jan. 22, 2026 • U.S. Provisional Applications filed on Jun. 6, 2025, and Sep. 19, 2025 |
| 25 | © 2026 Aprea Therapeutics, Inc. All Rights Reserved Securities Common Equivalents as of March 9, 2026 Preferred Stock (as converted) 15,596 Common Stock (1) 11,452,452 Warrants (2) 16,212,687 Options 840,121 Restricted Stock Units 25,176 Fully Diluted Equivalents 28,546,032 Aprea Therapeutics (NASDAQ: APRE) Financial Summary and Capitalization 1. 400,000,000 common shares authorized 2. Total warrants include pre-funded, Tranche A, Tranche B and Purchase Cash and Equivalents of ~$14.6M as of December 31, 2025 $5.6M in gross proceeds raised in private placement Jan 30, 2026 |
| 26 | © 2026 Aprea Therapeutics, Inc. All Rights Reserved Expected cash runway into Q1 2027 • Achieve near term inflection points and catalysts • Evaluate optimal strategic partnerships Near term catalysts • APR-1051: Q1 2026 Safety/efficacy data ✓ Q3 2026 Complete dose escalation • ATRN-119: October 2025 RP2D ✓ H2 2026 Potential collaborations on combinations Diversified portfolio with best in class, de-risked clinical and preclinical programs • Highly potent and selective WEE1 (APR-1051) and ATR (ATRN-119) inhibitors • Early evidence of clinical activity including two uPRs with APR-1051 • Single agent and combination potential therapies Technology developed by pioneers in synthetic lethality • Management with strong drug development and commercial expertise Investment Highlights |
| 27 | © 2026 Aprea Therapeutics, Inc. All Rights Reserved Aprea Therapeutics (NASDAQ: APRE) |