

Building a Differentiated ADC Company Nasdaq: PYXS March 2026 .2

Forward Looking Statement This presentation contains forward-looking statements for the purposes of the safe harbor provisions under The Private Securities Litigation Reform Act of 1995 and other federal securities laws. All statements other than statements of historical facts contained in this presentation, including without limitation statements regarding the Company’s plans to develop, manufacture and commercialize its product candidate, including micvotabart pelidotin (‘MICVO’); preliminary data, timing and progress of the Company’s ongoing clinical trials; the expected results of the Company’s clinical trials; the ability of preliminary, initial and topline clinical data to de-risk MICVO and be confirmed with clinical trial progression, including the safety, tolerability, and potential efficacy of MICVO; the potential differentiation, advantage or effectiveness of MICVO compared to other approved products or products in development; the dosage and treatment potential of MICVO; the size and future of the market; the plans and objectives of management, and the future results of operations and financial position of the Company, are forward-looking statements. These statements are neither promises nor guarantees, but are statements that involve known and unknown risks, uncertainties and other important factors that are in some cases beyond the Company’s control that may cause actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward-looking statements, including, but not limited to, the following: the risks inherent in drug research and development, the Company’s projected cash runway and potential needs for additional funding; the lengthy, expensive, and uncertain process of clinical drug development, including potential delays in or failure to obtain regulatory approvals; the Company’s reliance on third parties and collaborators to conduct clinical trials, manufacture their product candidate, and develop and commercialize their product candidate; and the Company’s ability to compete successfully against other drug candidate. Accordingly, investors should not rely upon forward-looking statements as predictions of future events. Except as required by applicable law, the Company undertakes no obligation to update publicly or revise any forward-looking statements contained herein, whether as a result of any new information, future events, changed circumstances or otherwise. Additionally, investors should read risk factors in the section titled “Risk Factors” set forth in Part II, Item 1A. of the Company’s Annual Report on Form 10-K filed on March 23, 2026, and our other filings, each of which is on file with the Securities and Exchange Commission.
Multiple Clinical Data Catalysts Expected in 2026 Positioned to be a Differentiated ADC Company First-in-Concept Extracellular ADC Technology Clinical focus on significant unmet need in R/M HNSCC Validated monotherapy & combination efficacy signal in R/M HNSCC

MICVO is a First-in-Concept ADC
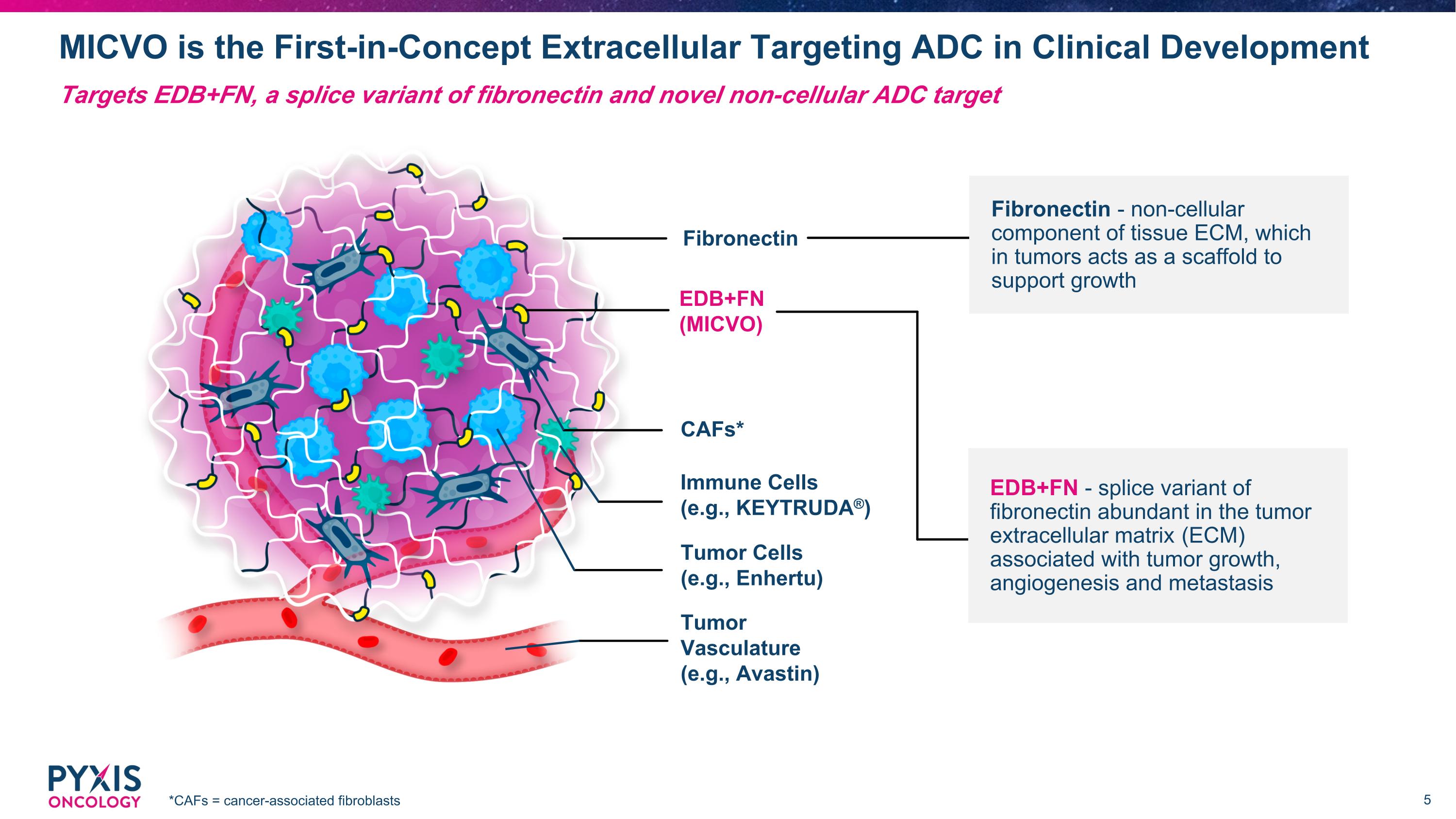
MICVO is the First-in-Concept Extracellular Targeting ADC in Clinical Development *CAFs = cancer-associated fibroblasts Targets EDB+FN, a splice variant of fibronectin and novel non-cellular ADC target Tumor Vasculature (e.g., Avastin) Fibronectin CAFs* EDB+FN (MICVO) Immune Cells (e.g., KEYTRUDA®) Tumor Cells (e.g., Enhertu) EDB+FN - splice variant of fibronectin abundant in the tumor extracellular matrix (ECM) associated with tumor growth, angiogenesis and metastasis Fibronectin - non-cellular component of tissue ECM, which in tumors acts as a scaffold to support growth
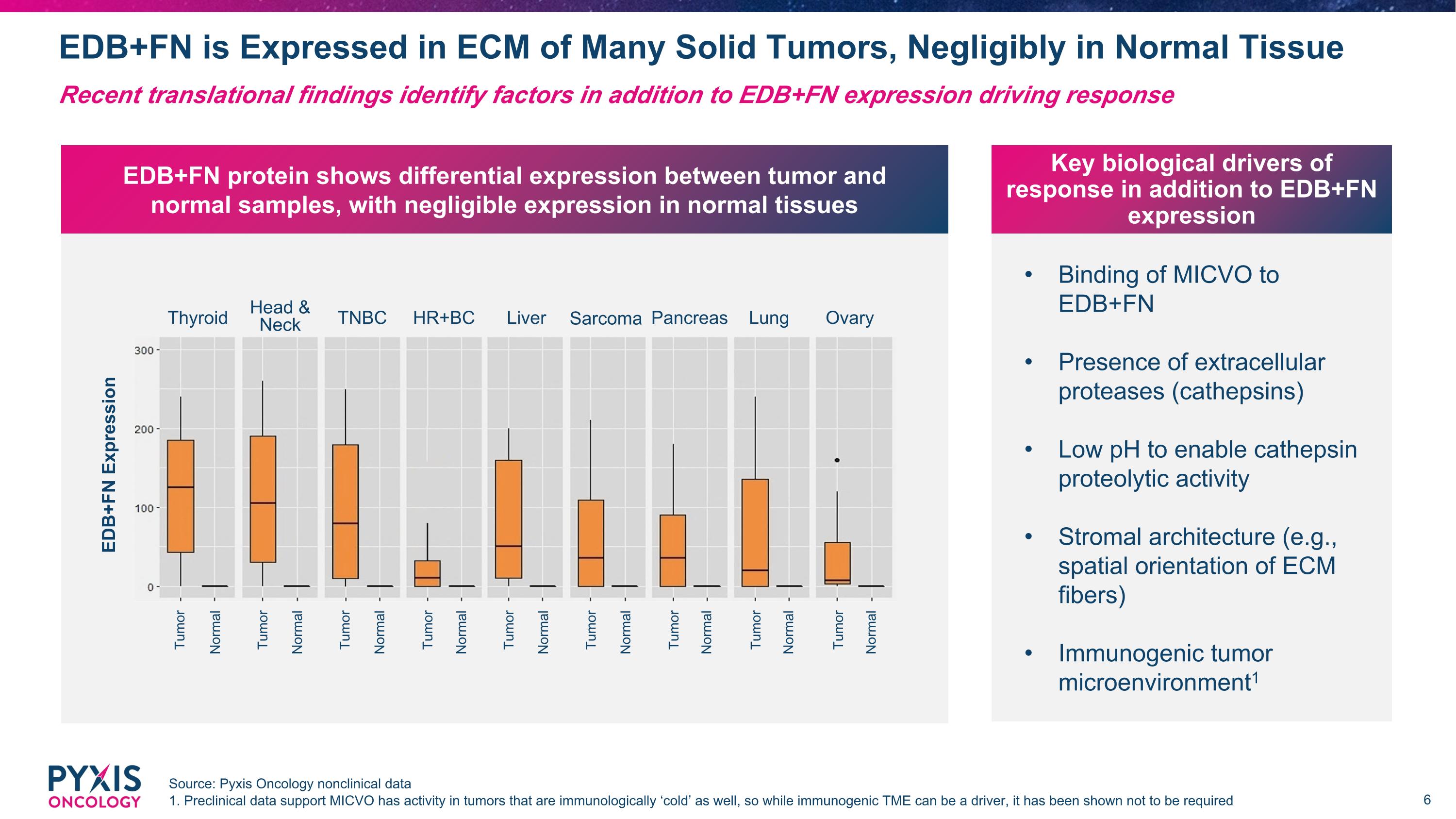
Binding of MICVO to EDB+FN Presence of extracellular proteases (cathepsins) Low pH to enable cathepsin proteolytic activity Stromal architecture (e.g., spatial orientation of ECM fibers) Immunogenic tumor microenvironment1 Key biological drivers of response in addition to EDB+FN expression EDB+FN is Expressed in ECM of Many Solid Tumors, Negligibly in Normal Tissue Source: Pyxis Oncology nonclinical data 1. Preclinical data support MICVO has activity in tumors that are immunologically ‘cold’ as well, so while immunogenic TME can be a driver, it has been shown not to be required Recent translational findings identify factors in addition to EDB+FN expression driving response EDB+FN protein shows differential expression between tumor and normal samples, with negligible expression in normal tissues EDB+FN Expression Head & Neck Thyroid TNBC Liver Sarcoma Lung Ovary Pancreas HR+BC Tumor Normal Tumor Normal Tumor Normal Tumor Normal Tumor Normal Tumor Normal Tumor Normal Tumor Normal Tumor Normal
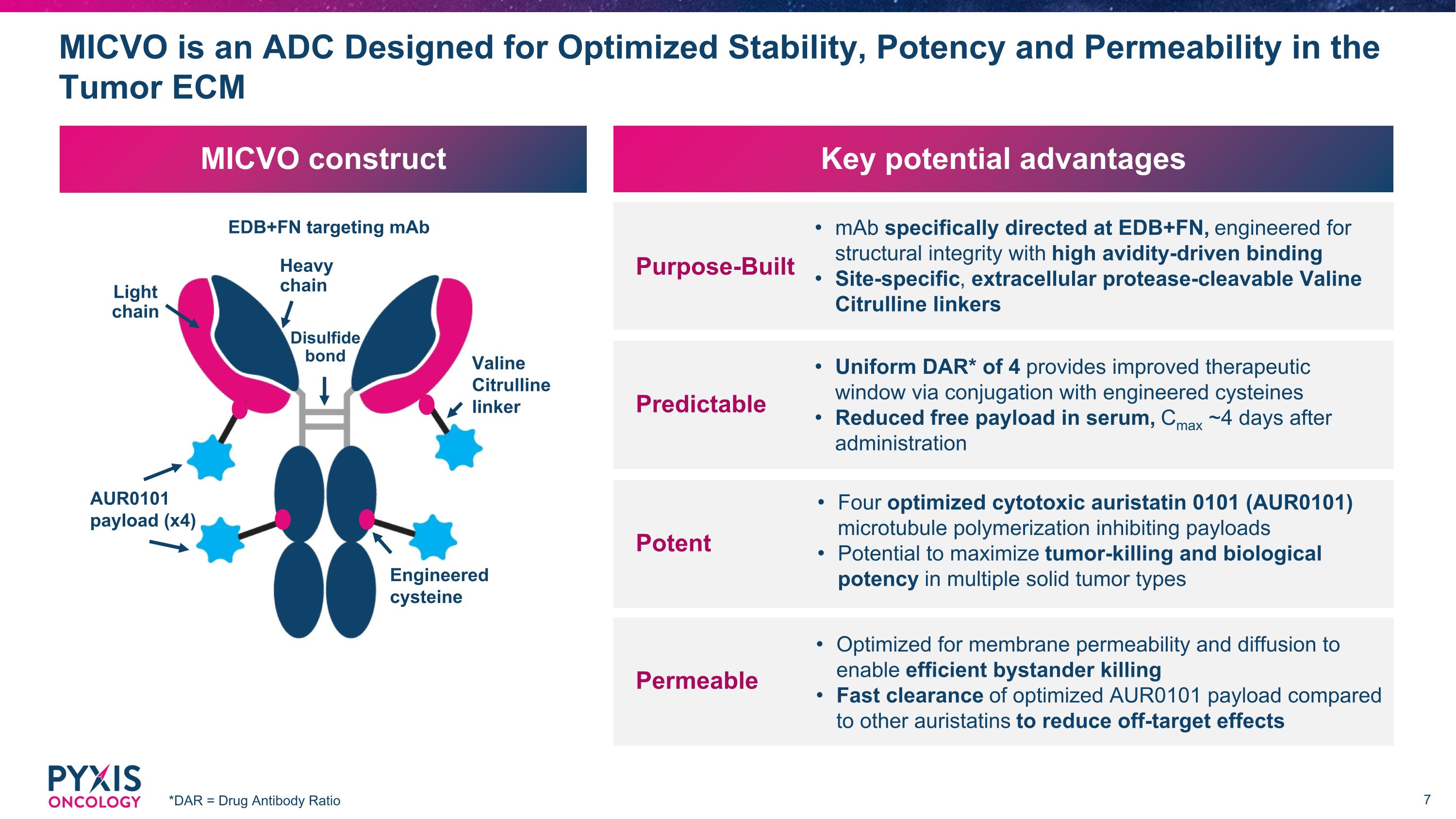
MICVO construct MICVO is an ADC Designed for Optimized Stability, Potency and Permeability in the Tumor ECM *DAR = Drug Antibody Ratio EDB+FN targeting mAb AUR0101 payload (x4) Valine Citrulline linker Light chain Heavy chain Engineered cysteine Disulfide bond Key potential advantages mAb specifically directed at EDB+FN, engineered for structural integrity with high avidity-driven binding Site-specific, extracellular protease-cleavable Valine Citrulline linkers Purpose-Built Uniform DAR* of 4 provides improved therapeutic window via conjugation with engineered cysteines Reduced free payload in serum, Cmax ~4 days after administration Predictable Four optimized cytotoxic auristatin 0101 (AUR0101) microtubule polymerization inhibiting payloads Potential to maximize tumor-killing and biological potency in multiple solid tumor types Potent Optimized for membrane permeability and diffusion to enable efficient bystander killing Fast clearance of optimized AUR0101 payload compared to other auristatins to reduce off-target effects Permeable
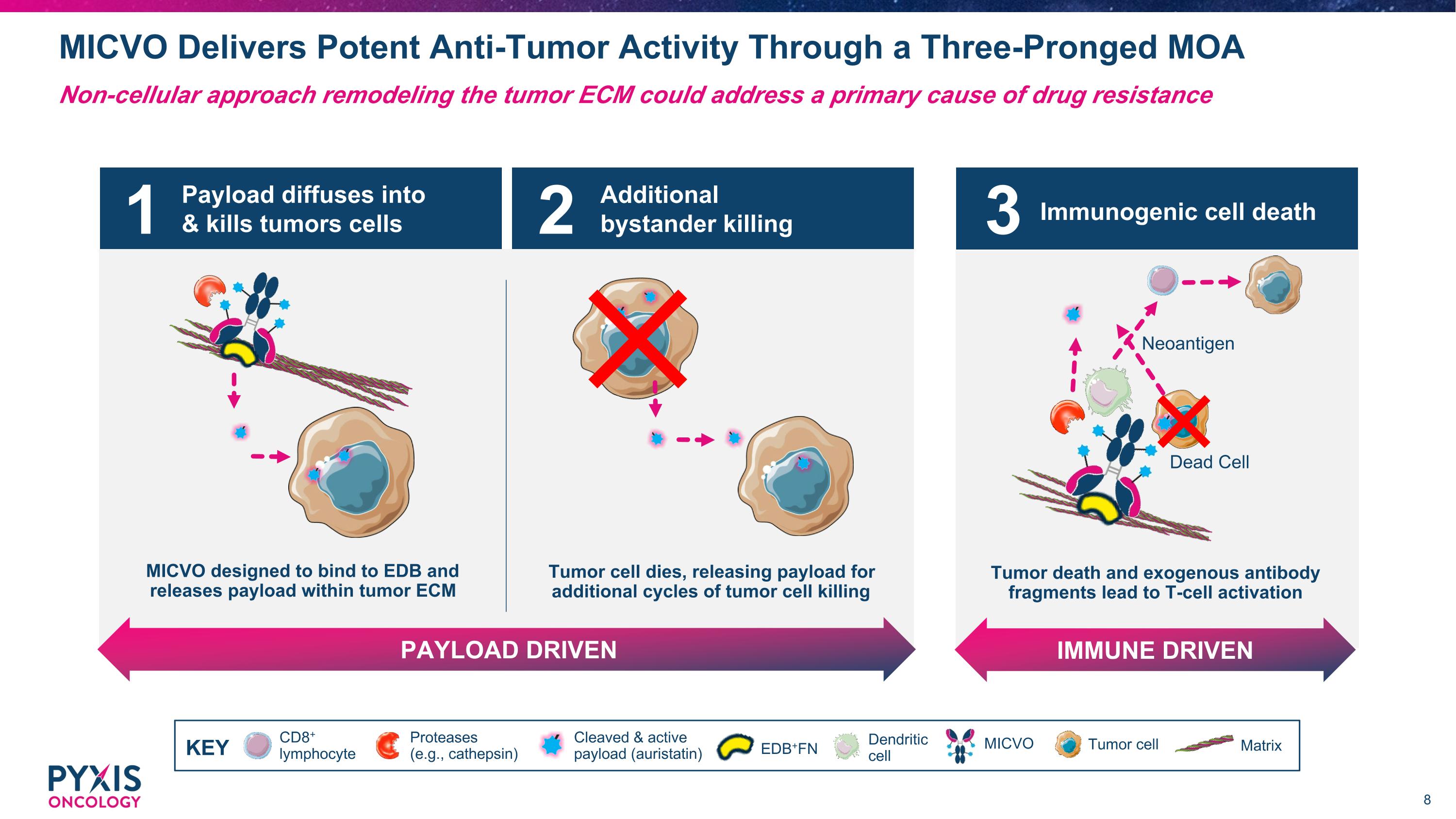
Payload diffuses into & kills tumors cells MICVO designed to bind to EDB and releases payload within tumor ECM Additional bystander killing PAYLOAD DRIVEN Tumor cell dies, releasing payload for additional cycles of tumor cell killing 1 2 MICVO Delivers Potent Anti-Tumor Activity Through a Three-Pronged MOA Non-cellular approach remodeling the tumor ECM could address a primary cause of drug resistance CD8+ lymphocyte Dendritic cell MICVO Cleaved & active payload (auristatin) Tumor cell Matrix Proteases (e.g., cathepsin) KEY EDB+FN Immunogenic cell death IMMUNE DRIVEN Tumor death and exogenous antibody fragments lead to T-cell activation Dead Cell Neoantigen 3
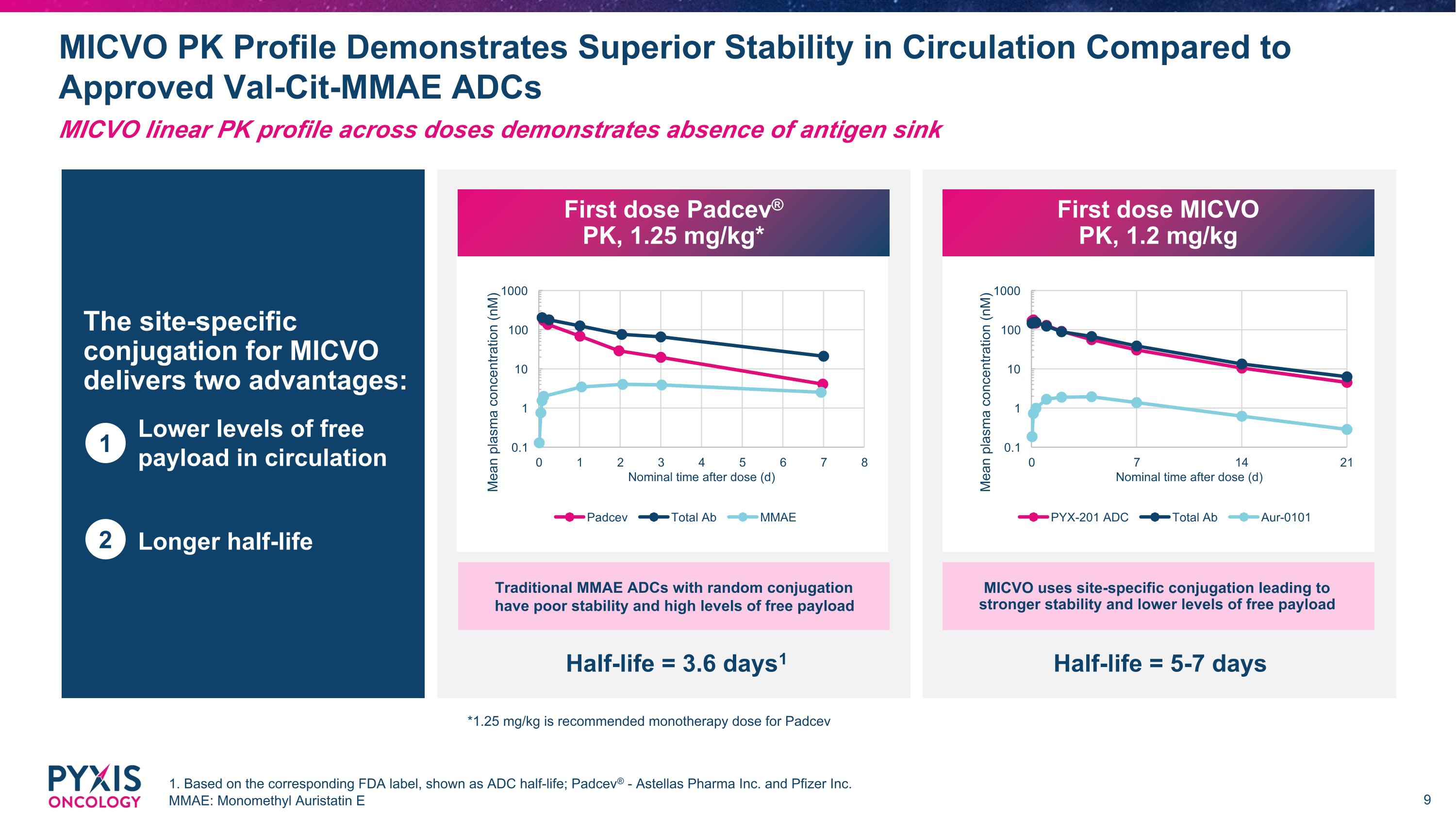
MICVO PK Profile Demonstrates Superior Stability in Circulation Compared to Approved Val-Cit-MMAE ADCs 1. Based on the corresponding FDA label, shown as ADC half-life; Padcev® - Astellas Pharma Inc. and Pfizer Inc. MMAE: Monomethyl Auristatin E MICVO linear PK profile across doses demonstrates absence of antigen sink The site-specific conjugation for MICVO delivers two advantages: Lower levels of free payload in circulation Longer half-life 1 2 Half-life = 3.6 days1 Half-life = 5-7 days MICVO *1.25 mg/kg is recommended monotherapy dose for Padcev First dose Padcev® PK, 1.25 mg/kg* First dose MICVO PK, 1.2 mg/kg Traditional MMAE ADCs with random conjugation have poor stability and high levels of free payload MICVO uses site-specific conjugation leading to stronger stability and lower levels of free payload

Initial MICVO Clinical Data Informs Path Forward
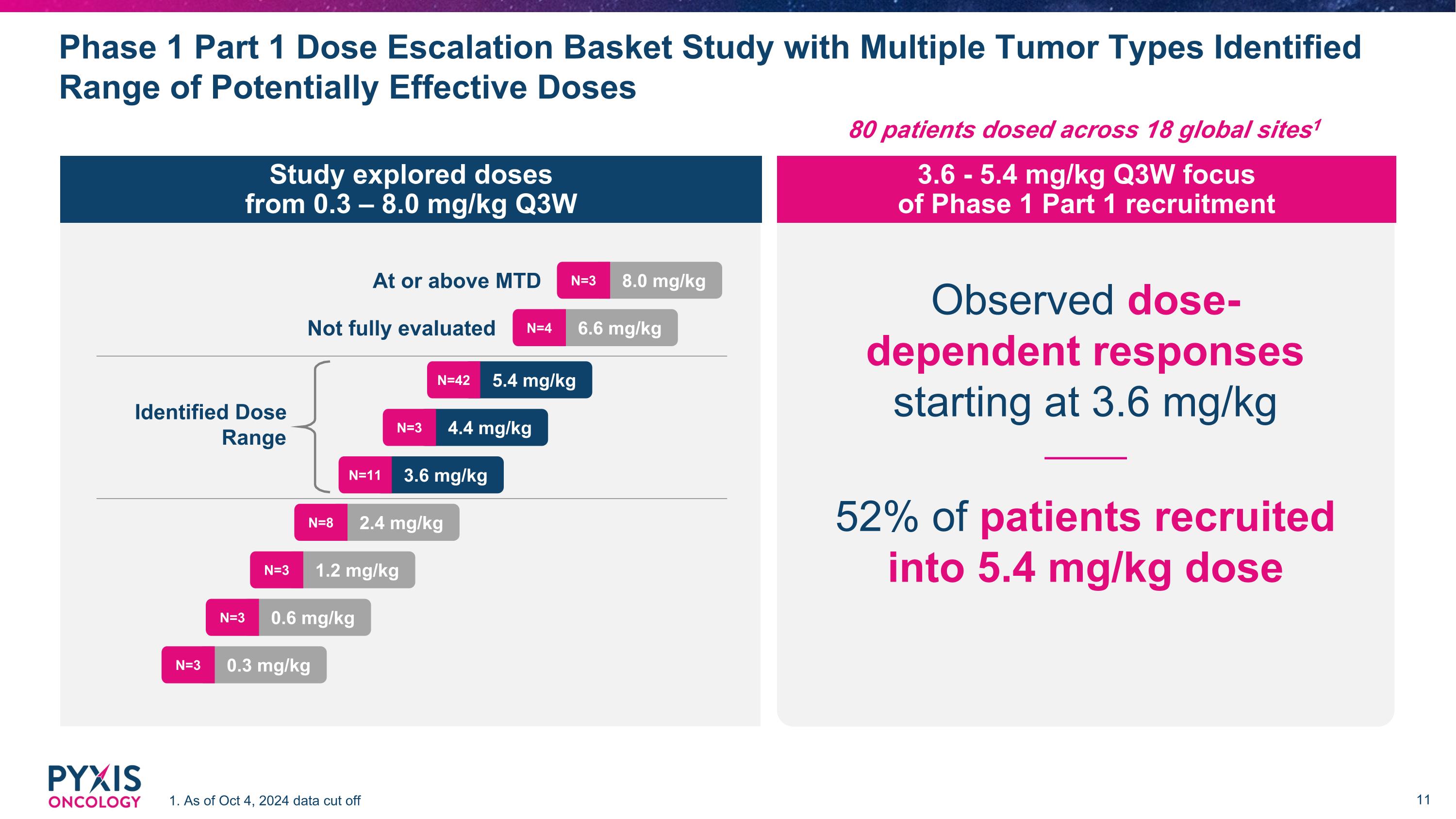
Phase 1 Part 1 Dose Escalation Basket Study with Multiple Tumor Types Identified Range of Potentially Effective Doses 1. As of Oct 4, 2024 data cut off Observed dose- dependent responses starting at 3.6 mg/kg 52% of patients recruited into 5.4 mg/kg dose 8.0 mg/kg N=3 Identified Dose Range Not fully evaluated At or above MTD 6.6 mg/kg N=4 5.4 mg/kg N=42 4.4 mg/kg N=3 3.6 mg/kg N=11 2.4 mg/kg N=8 1.2 mg/kg N=3 0.6 mg/kg N=3 0.3 mg/kg N=3 80 patients dosed across 18 global sites1 Study explored doses from 0.3 – 8.0 mg/kg Q3W 3.6 - 5.4 mg/kg Q3W focus of Phase 1 Part 1 recruitment
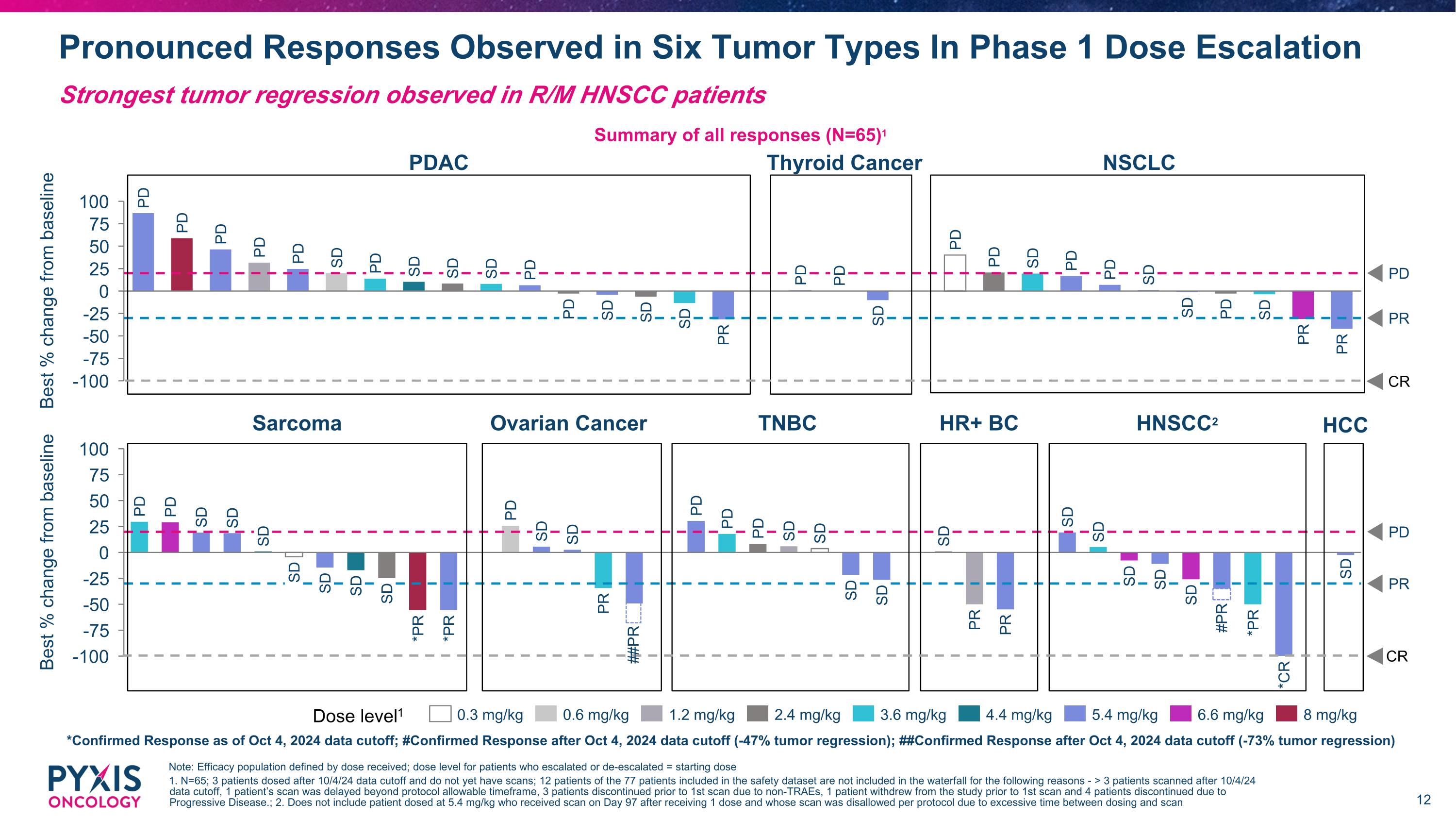
Pronounced Responses Observed in Six Tumor Types In Phase 1 Dose Escalation Note: Efficacy population defined by dose received; dose level for patients who escalated or de-escalated = starting dose 1. N=65; 3 patients dosed after 10/4/24 data cutoff and do not yet have scans; 12 patients of the 77 patients included in the safety dataset are not included in the waterfall for the following reasons - > 3 patients scanned after 10/4/24 data cutoff, 1 patient’s scan was delayed beyond protocol allowable timeframe, 3 patients discontinued prior to 1st scan due to non-TRAEs, 1 patient withdrew from the study prior to 1st scan and 4 patients discontinued due to Progressive Disease.; 2. Does not include patient dosed at 5.4 mg/kg who received scan on Day 97 after receiving 1 dose and whose scan was disallowed per protocol due to excessive time between dosing and scan Strongest tumor regression observed in R/M HNSCC patients Summary of all responses (N=65)1 Best % change from baseline PD PR PD PD PD PD PD SD PD SD SD SD PD PD SD SD SD PR PD PD SD PD PD SD PD PD SD SD PD SD PR PR Dose level1 0.3 mg/kg 0.6 mg/kg 1.2 mg/kg 2.4 mg/kg 3.6 mg/kg 4.4 mg/kg 5.4 mg/kg 6.6 mg/kg 8 mg/kg PDAC Thyroid Cancer NSCLC CR *Confirmed Response as of Oct 4, 2024 data cutoff; #Confirmed Response after Oct 4, 2024 data cutoff (-47% tumor regression); ##Confirmed Response after Oct 4, 2024 data cutoff (-73% tumor regression) PR Best % change from baseline PD PD SD SD SD SD SD SD SD *PR *PR PD SD SD PR PD PD PD SD SD PD SD SD PR PR SD SD SD SD SD *PR *CR SD SD Sarcoma Ovarian Cancer TNBC HR+ BC HNSCC2 ##PR #PR CR HCC
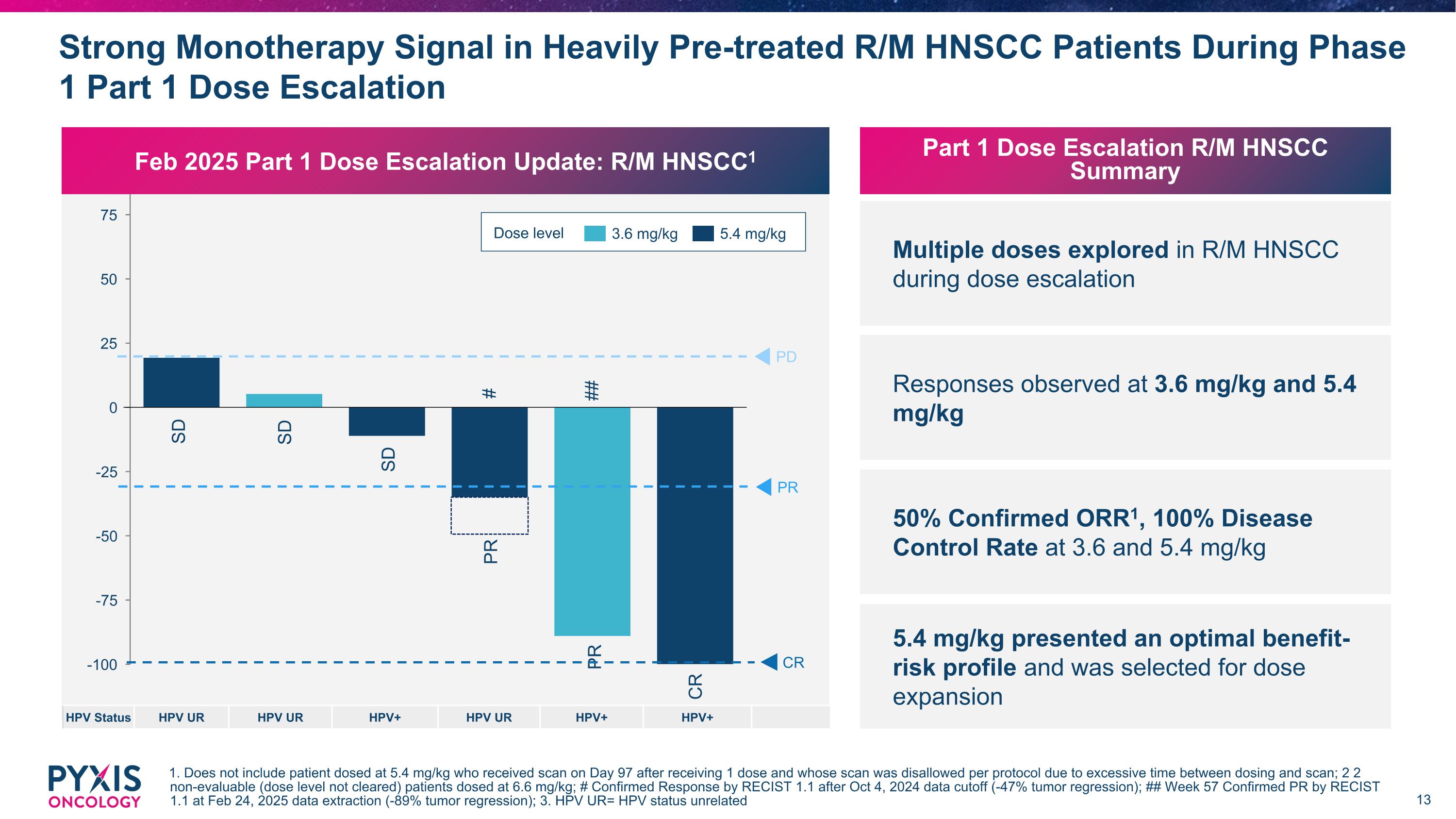
Strong Monotherapy Signal in Heavily Pre-treated R/M HNSCC Patients During Phase 1 Part 1 Dose Escalation 1. Does not include patient dosed at 5.4 mg/kg who received scan on Day 97 after receiving 1 dose and whose scan was disallowed per protocol due to excessive time between dosing and scan; 2 2 non-evaluable (dose level not cleared) patients dosed at 6.6 mg/kg; # Confirmed Response by RECIST 1.1 after Oct 4, 2024 data cutoff (-47% tumor regression); ## Week 57 Confirmed PR by RECIST 1.1 at Feb 24, 2025 data extraction (-89% tumor regression); 3. HPV UR= HPV status unrelated Dose level 3.6 mg/kg 5.4 mg/kg PD SD SD PR CR PR CR SD PR # ## HPV Status HPV UR HPV UR HPV+ HPV UR HPV+ HPV+ Feb 2025 Part 1 Dose Escalation Update: R/M HNSCC1 Part 1 Dose Escalation R/M HNSCC Summary Multiple doses explored in R/M HNSCC during dose escalation Responses observed at 3.6 mg/kg and 5.4 mg/kg 50% Confirmed ORR1, 100% Disease Control Rate at 3.6 and 5.4 mg/kg 5.4 mg/kg presented an optimal benefit-risk profile and was selected for dose expansion

Unmet Need in R/M HNSCC
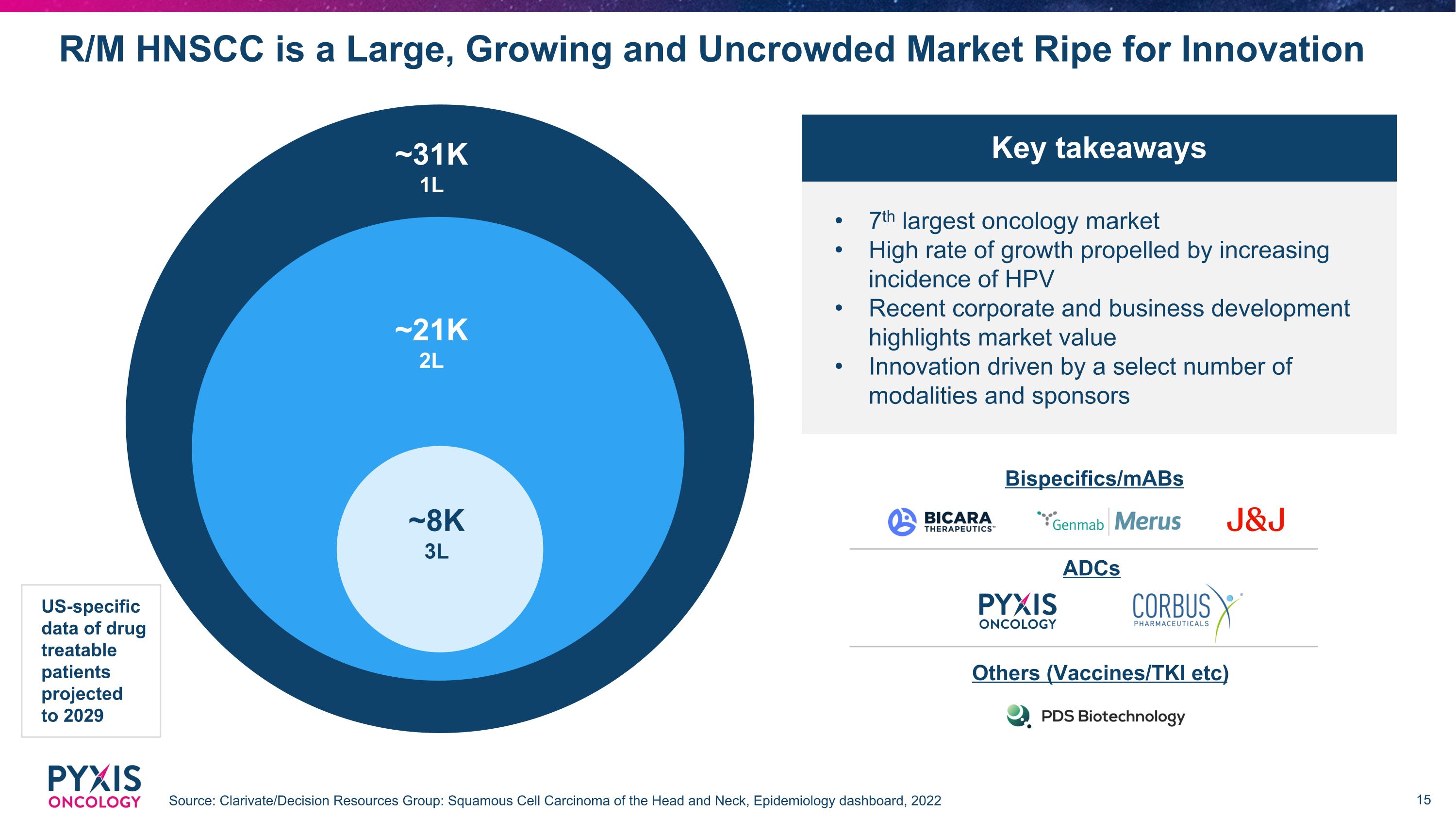
R/M HNSCC is a Large, Growing and Uncrowded Market Ripe for Innovation Source: Clarivate/Decision Resources Group: Squamous Cell Carcinoma of the Head and Neck, Epidemiology dashboard, 2022 ~31K 1L ~21K 2L ~8K 3L US-specific data of drug treatable patients projected to 2029 7th largest oncology market High rate of growth propelled by increasing incidence of HPV Recent corporate and business development highlights market value Innovation driven by a select number of modalities and sponsors Bispecifics/mABs ADCs Others (Vaccines/TKI etc) Key takeaways

Current US R/M HNSCC Standard of Care Leaves Patients with Few Options and Poor Efficacy CPI: Checkpoint Inhibitor; Cetux: Cetuximab CPI 3.0K 1L CPS<1 CPS≥1 CPI +/- Chemo ORR: 36% Chemo ORR: 20% 2L Cetux (EGFRi) and/or Chemo ORR: 11% CPI ORR: 13% 3L Cetux ORR<10% No established SOC
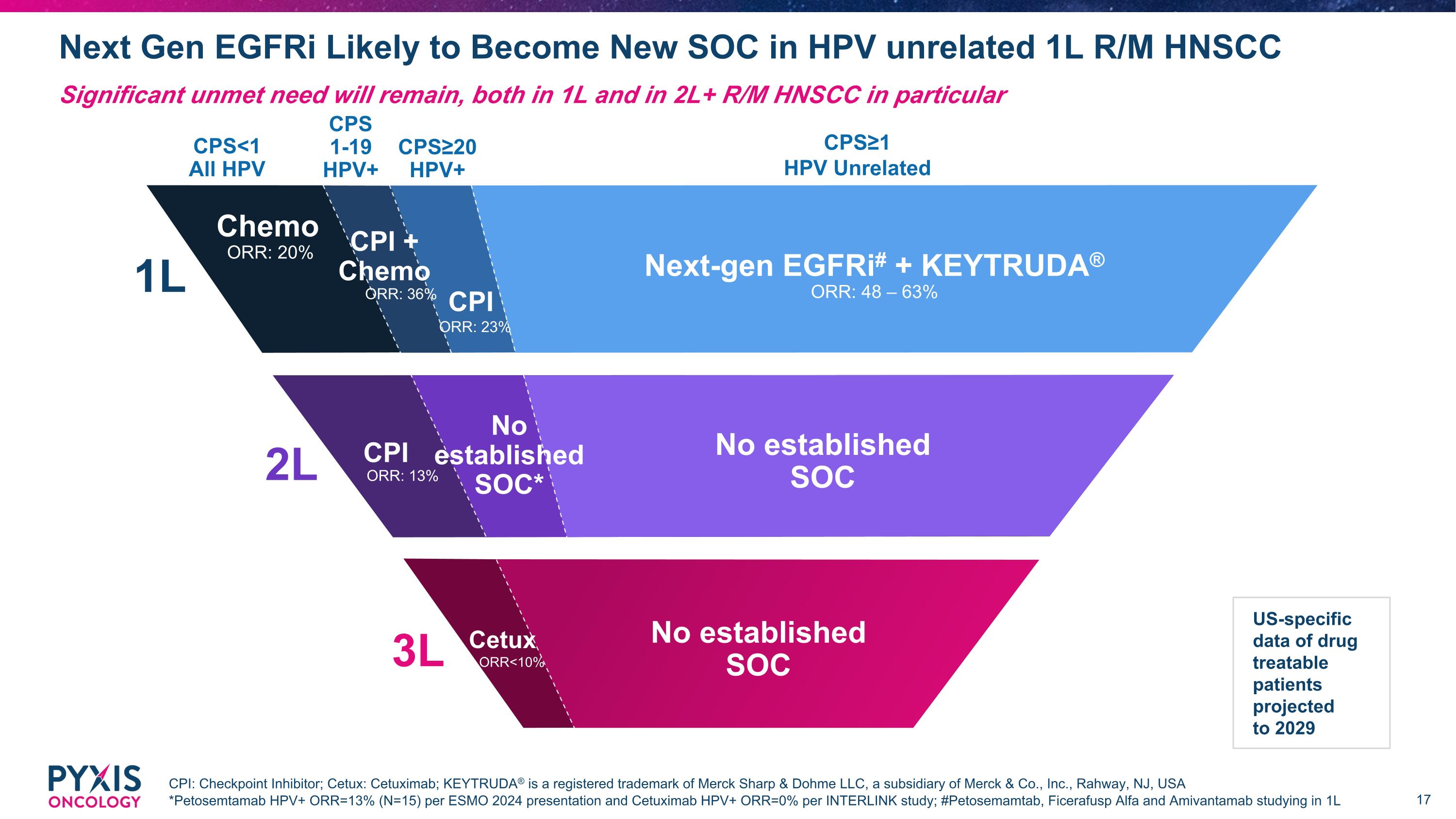
Next Gen EGFRi Likely to Become New SOC in HPV unrelated 1L R/M HNSCC CPI: Checkpoint Inhibitor; Cetux: Cetuximab; KEYTRUDA® is a registered trademark of Merck Sharp & Dohme LLC, a subsidiary of Merck & Co., Inc., Rahway, NJ, USA *Petosemtamab HPV+ ORR=13% (N=15) per ESMO 2024 presentation and Cetuximab HPV+ ORR=0% per INTERLINK study; #Petosemamtab, Ficerafusp Alfa and Amivantamab studying in 1L Significant unmet need will remain, both in 1L and in 2L+ R/M HNSCC in particular CPI 3.0K 1L CPS<1 All HPV CPS≥20 HPV+ CPS 1-19 HPV+ CPS≥1 HPV Unrelated Next-gen EGFRi# + KEYTRUDA® ORR: 48 – 63% Chemo ORR: 20% CPI ORR: 23% CPI + Chemo ORR: 36% 2L No established SOC* No established SOC CPI ORR: 13% 3L No established SOC Cetux ORR<10% US-specific data of drug treatable patients projected to 2029
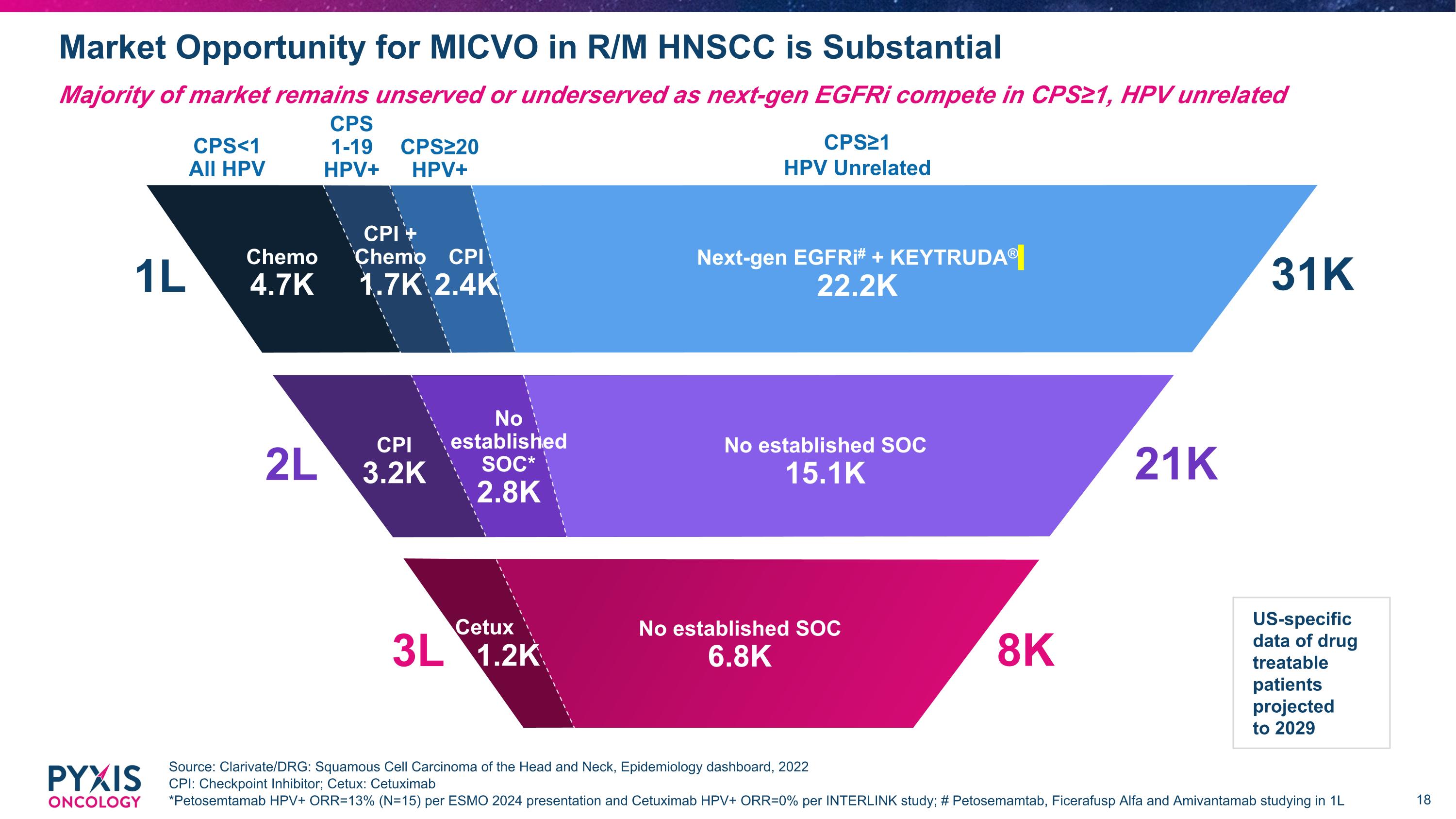
Market Opportunity for MICVO in R/M HNSCC is Substantial Source: Clarivate/DRG: Squamous Cell Carcinoma of the Head and Neck, Epidemiology dashboard, 2022 CPI: Checkpoint Inhibitor; Cetux: Cetuximab *Petosemtamab HPV+ ORR=13% (N=15) per ESMO 2024 presentation and Cetuximab HPV+ ORR=0% per INTERLINK study; # Petosemamtab, Ficerafusp Alfa and Amivantamab studying in 1L Majority of market remains unserved or underserved as next-gen EGFRi compete in CPS≥1, HPV unrelated US-specific data of drug treatable patients projected to 2029 CPI 3.0K 1L CPS<1 All HPV CPS≥20 HPV+ CPS 1-19 HPV+ CPS≥1 HPV Unrelated Next-gen EGFRi# + KEYTRUDA® 22.2K Chemo 4.7K CPI 2.4K CPI + Chemo 1.7K 31K 2L No established SOC* 2.8K No established SOC 15.1K 21K CPI 3.2K 8K 3L Cetux 1.2K No established SOC 6.8K
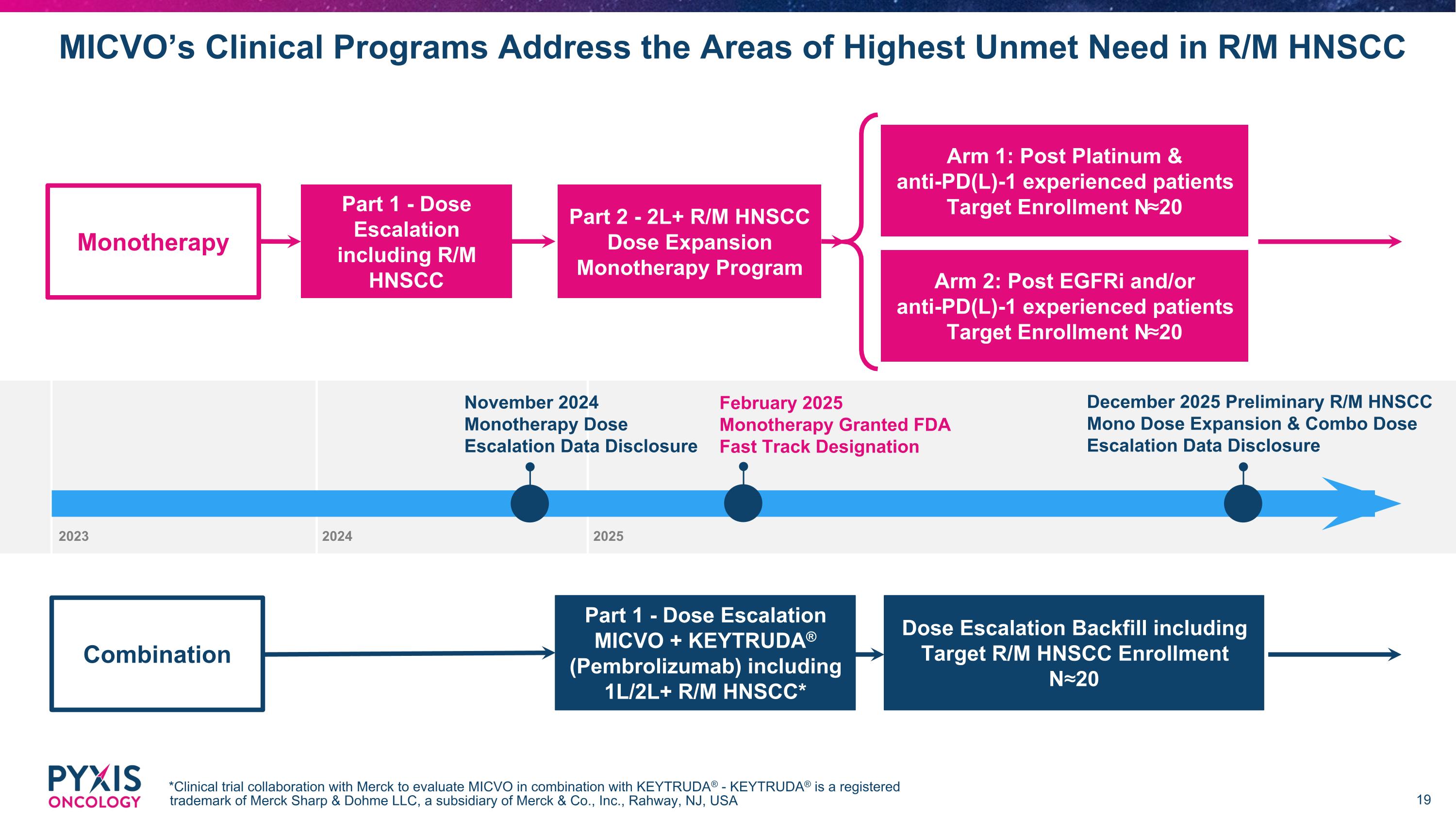
November 2024 Monotherapy Dose Escalation Data Disclosure December 2025 Preliminary R/M HNSCC Mono Dose Expansion & Combo Dose Escalation Data Disclosure MICVO’s Clinical Programs Address the Areas of Highest Unmet Need in R/M HNSCC *Clinical trial collaboration with Merck to evaluate MICVO in combination with KEYTRUDA® - KEYTRUDA® is a registered trademark of Merck Sharp & Dohme LLC, a subsidiary of Merck & Co., Inc., Rahway, NJ, USA Arm 1: Post Platinum & anti-PD(L)-1 experienced patients Target Enrollment N≈20 Part 2 - 2L+ R/M HNSCC Dose Expansion Monotherapy Program Part 1 - Dose Escalation MICVO + KEYTRUDA® (Pembrolizumab) including 1L/2L+ R/M HNSCC* Part 1 - Dose Escalation including R/M HNSCC Dose Escalation Backfill including Target R/M HNSCC Enrollment N≈20 Monotherapy Combination Arm 2: Post EGFRi and/or anti-PD(L)-1 experienced patients Target Enrollment N≈20 2023 2024 2025 February 2025 Monotherapy Granted FDA Fast Track Designation

MICVO Monotherapy in 2L+ R/M HNSCC December 2025 Preliminary Data

MICVO Preliminary Data Revealed Unsurpassed Efficacy in 2L+ R/M HNSCC 1. Efficacy evaluable (N=13) does not include N=1 dose escalation patient dosed at 5.4 mg/kg who received scan on Day 97 after receiving 1 dose and whose scan was disallowed per protocol due to excessive time between dosing and scan and N=4 patients in dose expansion who have not yet received 1st scan and are ongoing Phase 1: 46% Confirmed ORR and 92% Disease Control Rate (N=13, Efficacy Evaluable1) Correlation identified between high body weight patients and AEs has been linked to increased drug exposure Dosing interventions including dose capping and adjusted ideal body weight (AIBW) dosing are being employed to optimize MICVO benefit/risk profile moving forward Data as of Nov 3, 2025
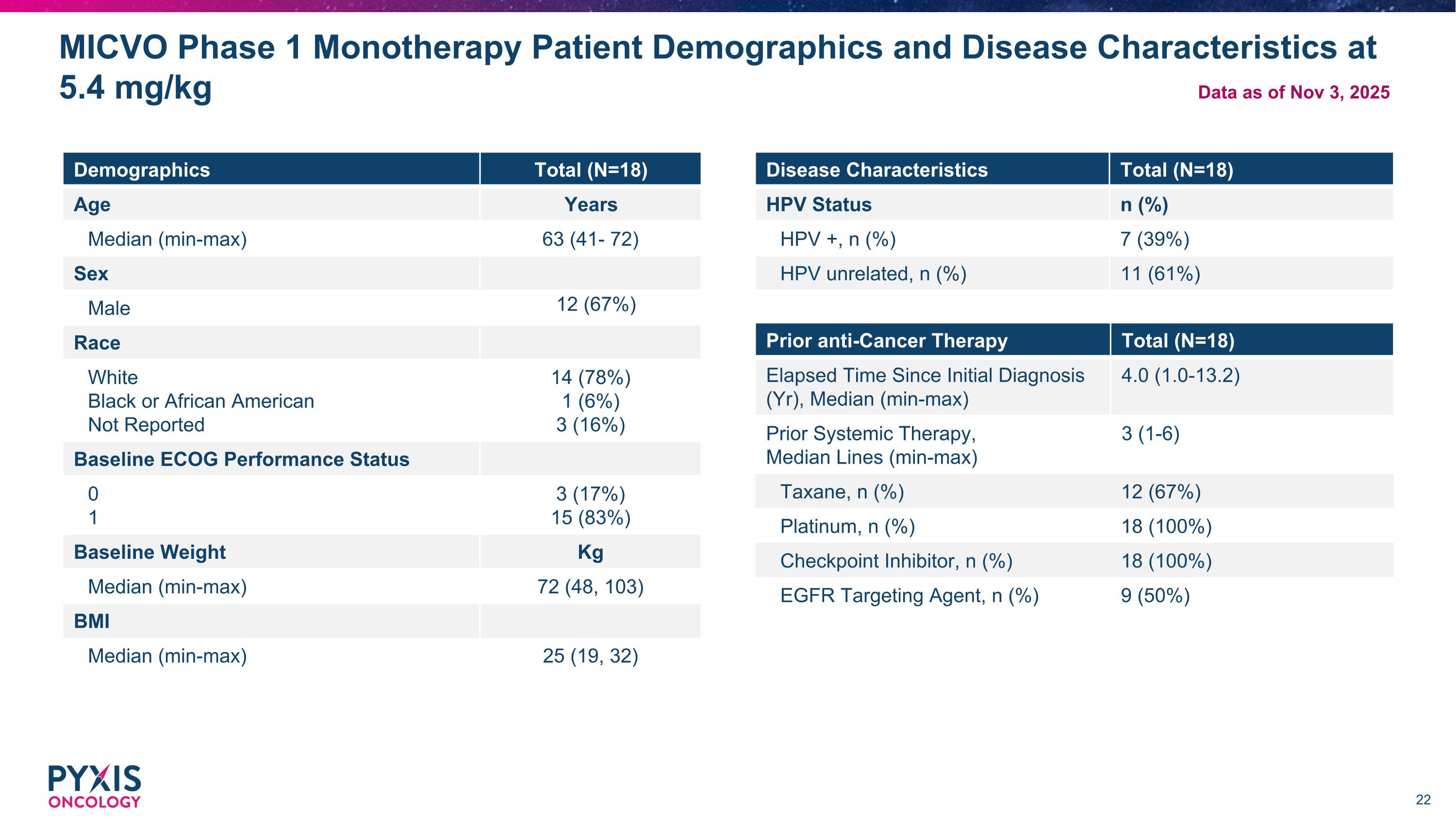
MICVO Phase 1 Monotherapy Patient Demographics and Disease Characteristics at 5.4 mg/kg Demographics Total (N=18) Age Years Median (min-max) 63 (41- 72) Sex Male 12 (67%) Race White Black or African American Not Reported 14 (78%) 1 (6%) 3 (16%) Baseline ECOG Performance Status 0 1 3 (17%) 15 (83%) Baseline Weight Kg Median (min-max) 72 (48, 103) BMI Median (min-max) 25 (19, 32) Prior anti-Cancer Therapy Total (N=18) Elapsed Time Since Initial Diagnosis (Yr), Median (min-max) 4.0 (1.0-13.2) Prior Systemic Therapy, Median Lines (min-max) 3 (1-6) Taxane, n (%) 12 (67%) Platinum, n (%) 18 (100%) Checkpoint Inhibitor, n (%) 18 (100%) EGFR Targeting Agent, n (%) 9 (50%) Disease Characteristics Total (N=18) HPV Status n (%) HPV +, n (%) 7 (39%) HPV unrelated, n (%) 11 (61%) Data as of Nov 3, 2025
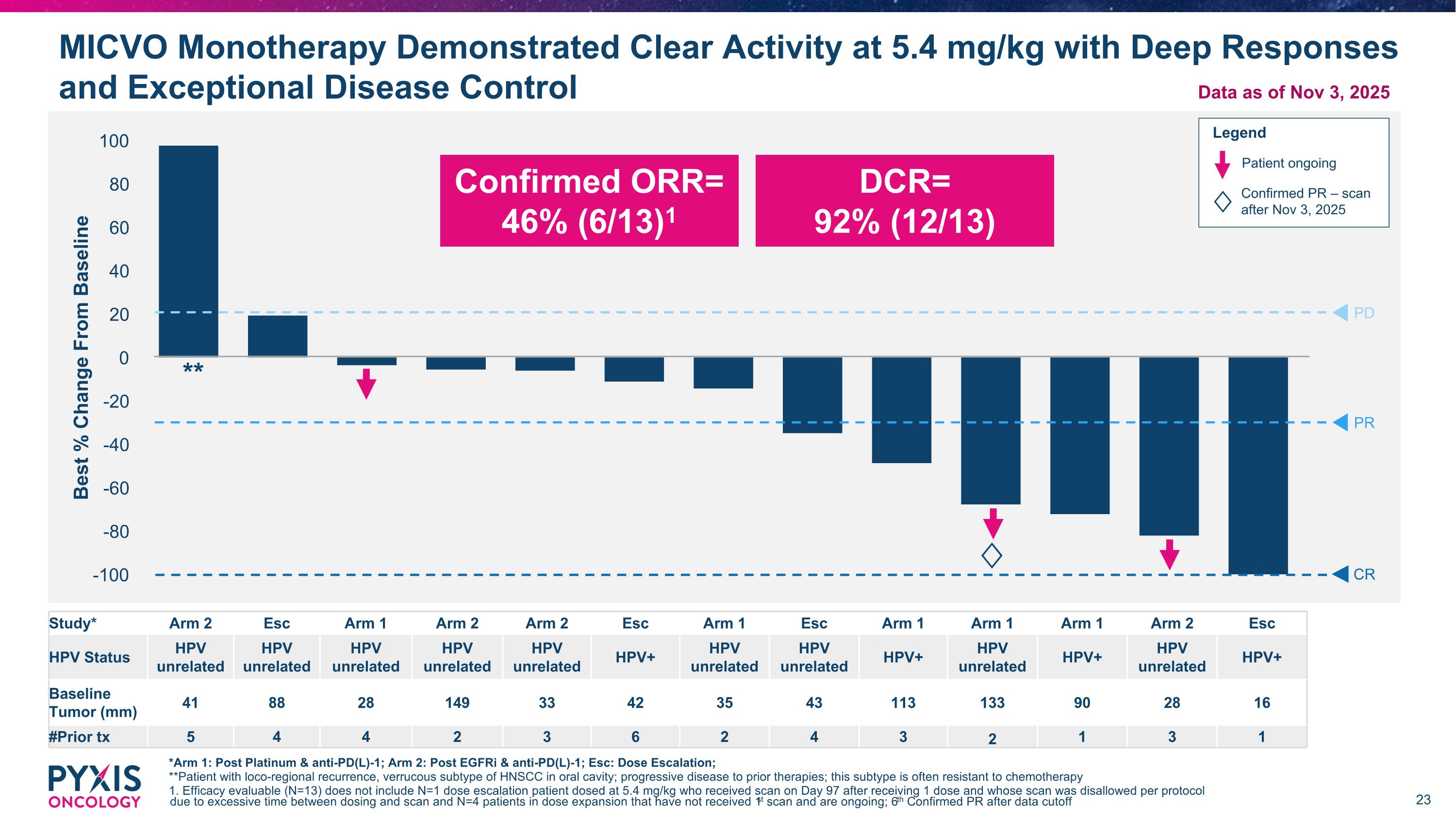
MICVO Monotherapy Demonstrated Clear Activity at 5.4 mg/kg with Deep Responses and Exceptional Disease Control *Arm 1: Post Platinum & anti-PD(L)-1; Arm 2: Post EGFRi & anti-PD(L)-1; Esc: Dose Escalation; **Patient with loco-regional recurrence, verrucous subtype of HNSCC in oral cavity; progressive disease to prior therapies; this subtype is often resistant to chemotherapy 1. Efficacy evaluable (N=13) does not include N=1 dose escalation patient dosed at 5.4 mg/kg who received scan on Day 97 after receiving 1 dose and whose scan was disallowed per protocol due to excessive time between dosing and scan and N=4 patients in dose expansion that have not received 1st scan and are ongoing; 6th Confirmed PR after data cutoff PD PR CR ♢ Study* Arm 2 Esc Arm 1 Arm 2 Arm 2 Esc Arm 1 Esc Arm 1 Arm 1 Arm 1 Arm 2 Esc HPV Status HPV unrelated HPV unrelated HPV unrelated HPV unrelated HPV unrelated HPV+ HPV unrelated HPV unrelated HPV+ HPV unrelated HPV+ HPV unrelated HPV+ Baseline Tumor (mm) 41 88 28 149 33 42 35 43 113 133 90 28 16 #Prior tx 5 4 4 2 3 6 2 4 3 2 1 3 1 Legend Patient ongoing Confirmed ORR= 46% (6/13)1 DCR= 92% (12/13) Confirmed PR – scan after Nov 3, 2025 ♢ ** Data as of Nov 3, 2025
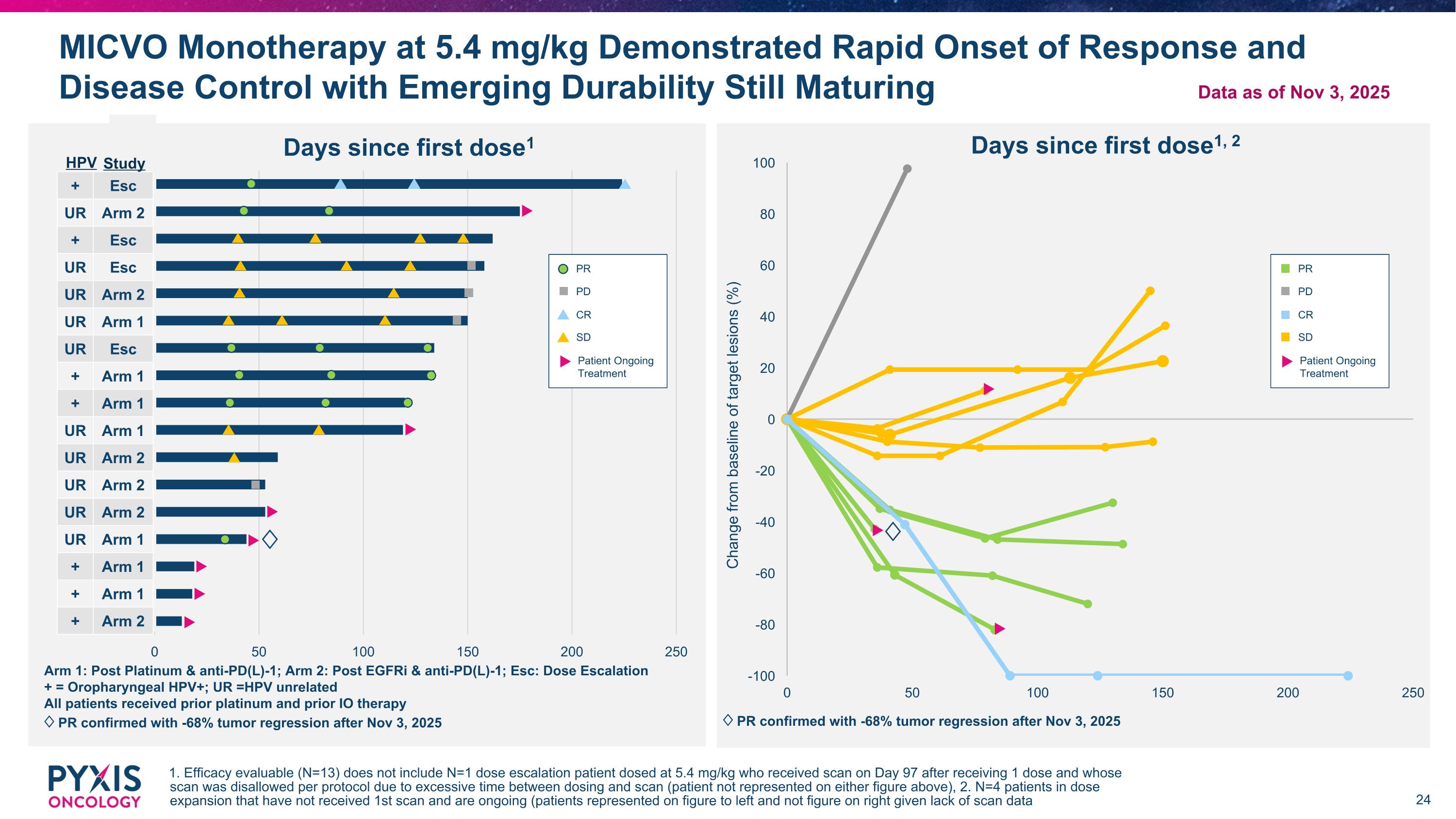
MICVO Monotherapy at 5.4 mg/kg Demonstrated Rapid Onset of Response and Disease Control with Emerging Durability Still Maturing 1. Efficacy evaluable (N=13) does not include N=1 dose escalation patient dosed at 5.4 mg/kg who received scan on Day 97 after receiving 1 dose and whose scan was disallowed per protocol due to excessive time between dosing and scan (patient not represented on either figure above), 2. N=4 patients in dose expansion that have not received 1st scan and are ongoing (patients represented on figure to left and not figure on right given lack of scan data Arm 1: Post Platinum & anti-PD(L)-1; Arm 2: Post EGFRi & anti-PD(L)-1; Esc: Dose Escalation + = Oropharyngeal HPV+; UR =HPV unrelated All patients received prior platinum and prior IO therapy ♢ PR confirmed with -68% tumor regression after Nov 3, 2025 HPV + Esc UR Arm 2 + Esc UR Esc UR Arm 2 UR Arm 1 UR Esc + Arm 1 + Arm 1 UR Arm 1 UR Arm 2 UR Arm 2 UR Arm 2 UR Arm 1 + Arm 1 + Arm 1 + Arm 2 PR PD CR SD Patient Ongoing Treatment Study ♢ ♢ ♢ PR confirmed with -68% tumor regression after Nov 3, 2025 Data as of Nov 3, 2025 PR PD CR SD Patient Ongoing Treatment
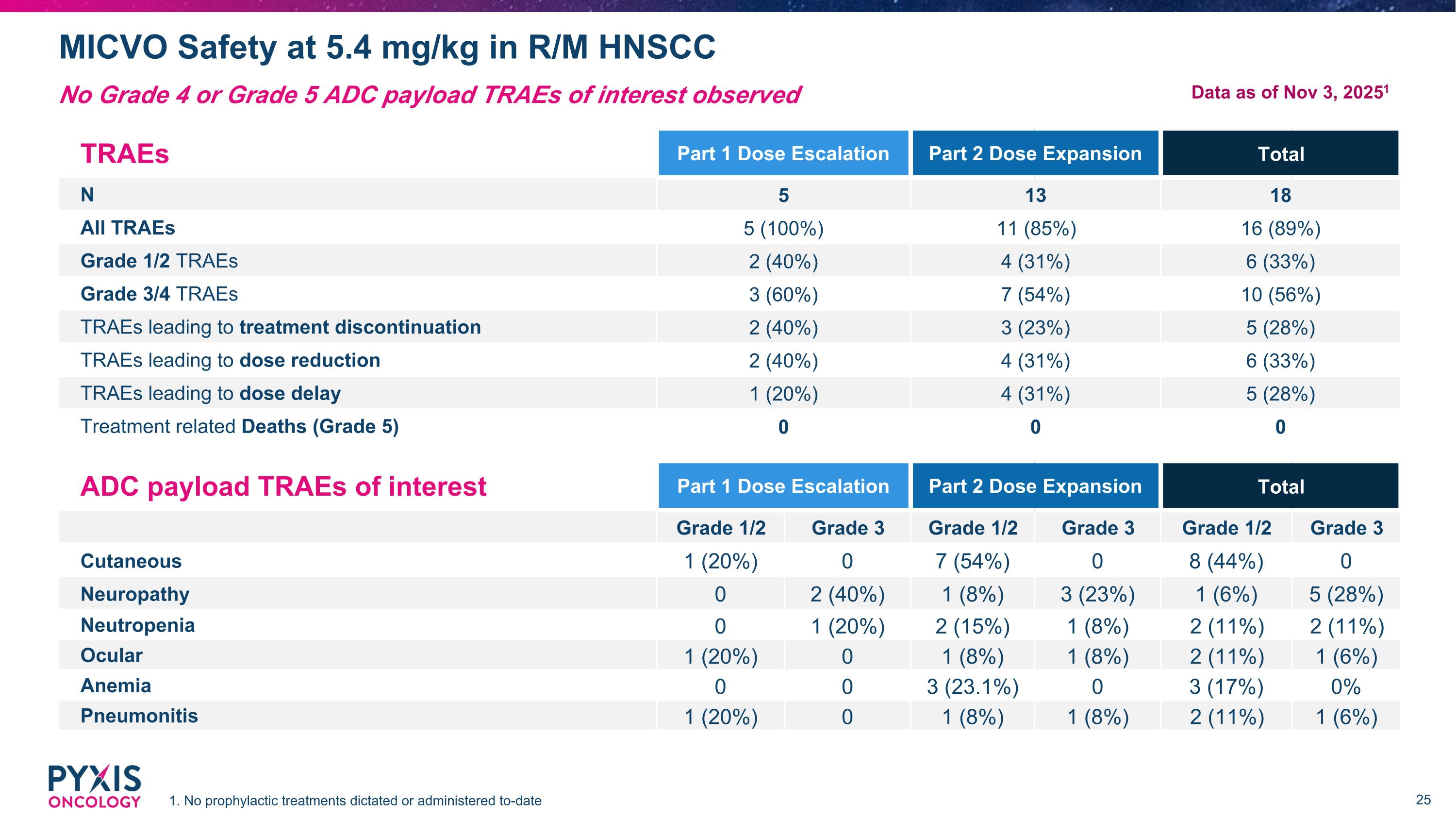
MICVO Safety at 5.4 mg/kg in R/M HNSCC 1. No prophylactic treatments dictated or administered to-date No Grade 4 or Grade 5 ADC payload TRAEs of interest observed TRAEs Part 1 Dose Escalation Part 2 Dose Expansion Total N 5 13 18 All TRAEs 5 (100%) 11 (85%) 16 (89%) Grade 1/2 TRAEs 2 (40%) 4 (31%) 6 (33%) Grade 3/4 TRAEs 3 (60%) 7 (54%) 10 (56%) TRAEs leading to treatment discontinuation 2 (40%) 3 (23%) 5 (28%) TRAEs leading to dose reduction 2 (40%) 4 (31%) 6 (33%) TRAEs leading to dose delay 1 (20%) 4 (31%) 5 (28%) Treatment related Deaths (Grade 5) 0 0 0 ADC payload TRAEs of interest Part 1 Dose Escalation Part 2 Dose Expansion Total Grade 1/2 Grade 3 Grade 1/2 Grade 3 Grade 1/2 Grade 3 Cutaneous 1 (20%) 0 7 (54%) 0 8 (44%) 0 Neuropathy 0 2 (40%) 1 (8%) 3 (23%) 1 (6%) 5 (28%) Neutropenia 0 1 (20%) 2 (15%) 1 (8%) 2 (11%) 2 (11%) Ocular 1 (20%) 0 1 (8%) 1 (8%) 2 (11%) 1 (6%) Anemia 0 0 3 (23.1%) 0 3 (17%) 0% Pneumonitis 1 (20%) 0 1 (8%) 1 (8%) 2 (11%) 1 (6%) Data as of Nov 3, 20251

MICVO Modified Weight-Based Dosing
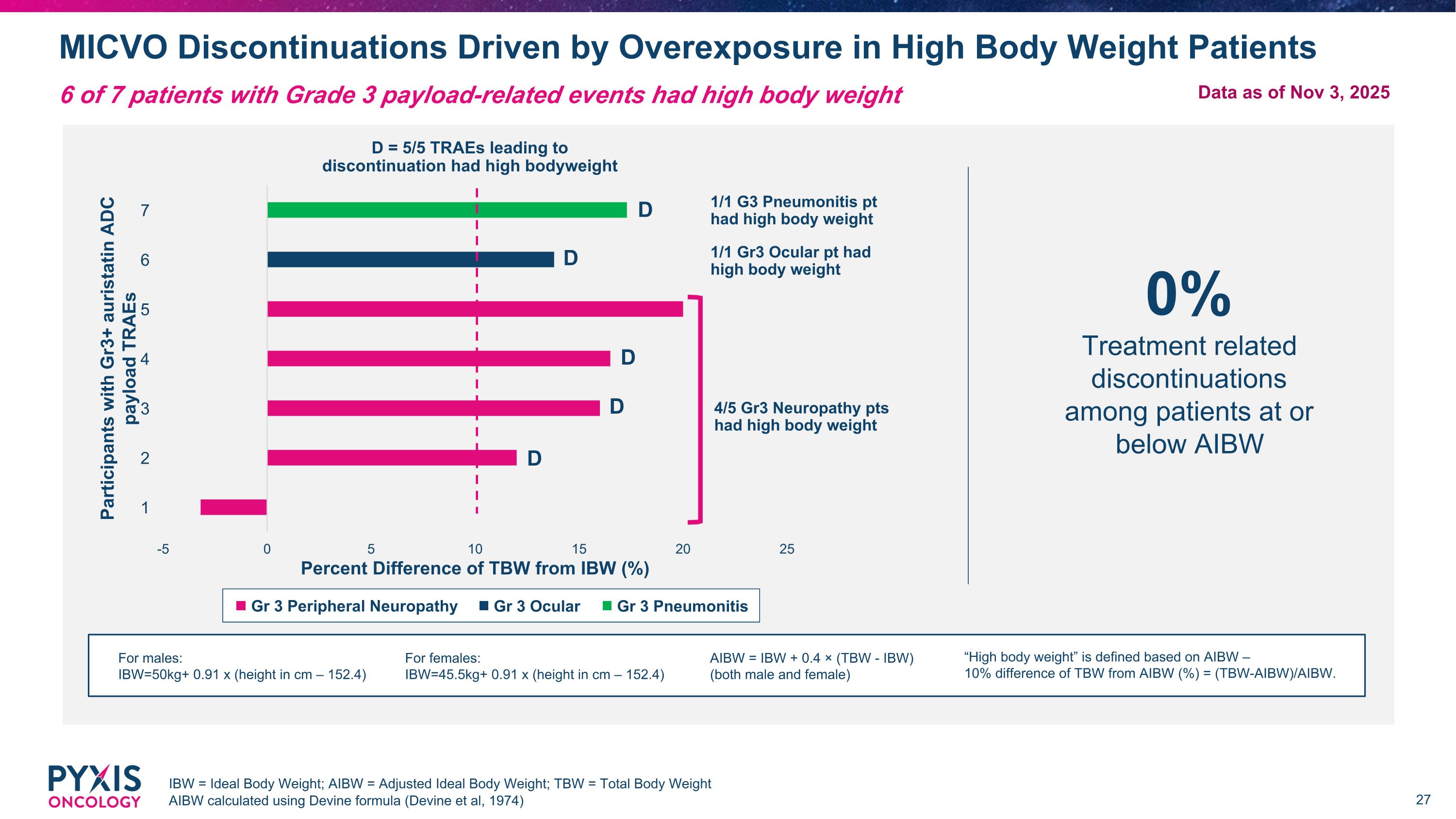
MICVO Discontinuations Driven by Overexposure in High Body Weight Patients IBW = Ideal Body Weight; AIBW = Adjusted Ideal Body Weight; TBW = Total Body Weight AIBW calculated using Devine formula (Devine et al, 1974) 6 of 7 patients with Grade 3 payload-related events had high body weight 0% Treatment related discontinuations among patients at or below AIBW Gr 3 Ocular Gr 3 Pneumonitis Gr 3 Peripheral Neuropathy 1/1 G3 Pneumonitis pt had high body weight 1/1 Gr3 Ocular pt had high body weight 4/5 Gr3 Neuropathy pts had high body weight D D D D D D = 5/5 TRAEs leading to discontinuation had high bodyweight For males: IBW=50kg+ 0.91 x (height in cm – 152.4) For females: IBW=45.5kg+ 0.91 x (height in cm – 152.4) AIBW = IBW + 0.4 × (TBW - IBW) (both male and female) “High body weight” is defined based on AIBW – 10% difference of TBW from AIBW (%) = (TBW-AIBW)/AIBW. Data as of Nov 3, 2025
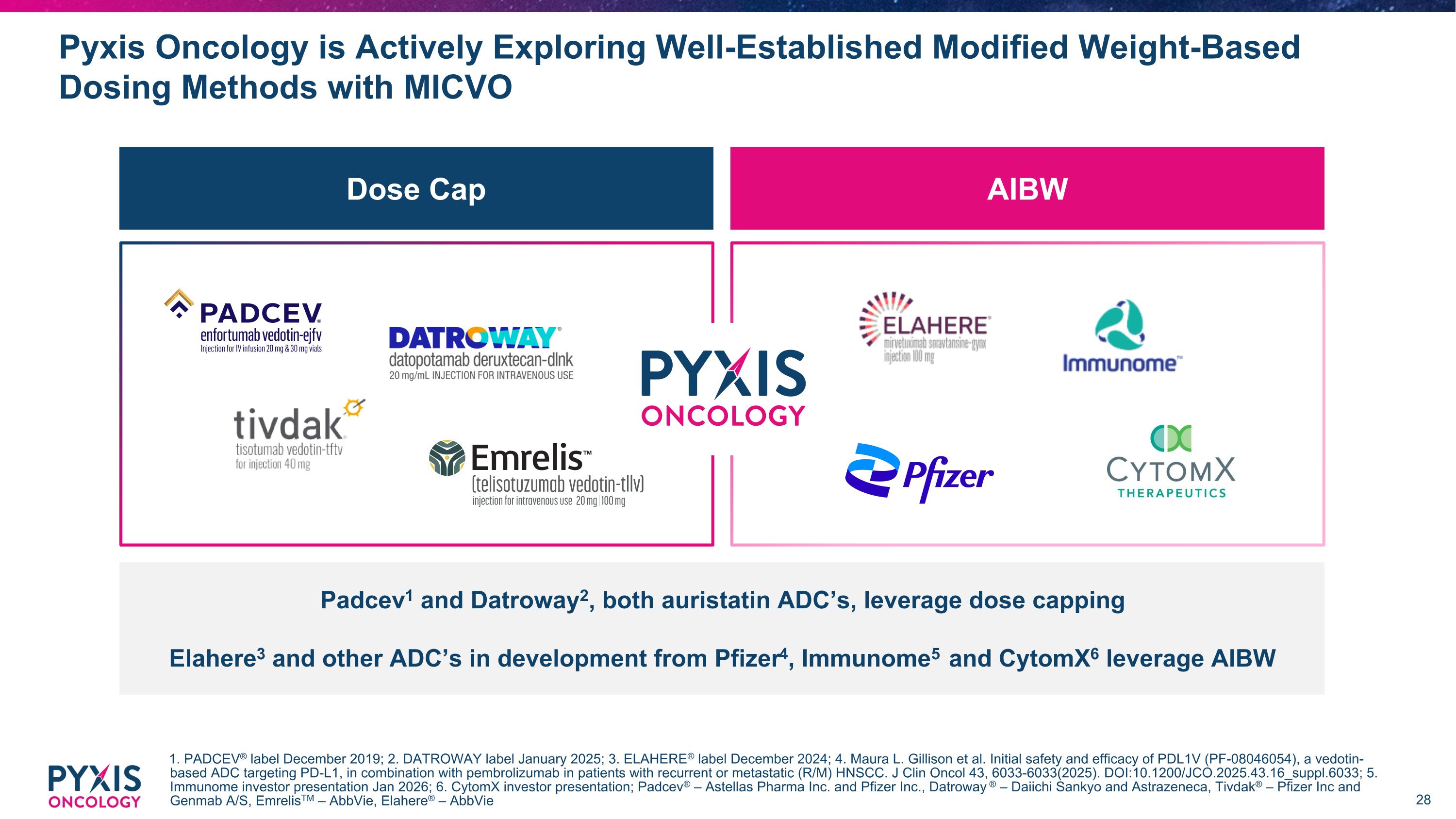
AIBW Dose Cap Pyxis Oncology is Actively Exploring Well-Established Modified Weight-Based Dosing Methods with MICVO 1. PADCEV® label December 2019; 2. DATROWAY label January 2025; 3. ELAHERE® label December 2024; 4. Maura L. Gillison et al. Initial safety and efficacy of PDL1V (PF-08046054), a vedotin-based ADC targeting PD-L1, in combination with pembrolizumab in patients with recurrent or metastatic (R/M) HNSCC. J Clin Oncol 43, 6033-6033(2025). DOI:10.1200/JCO.2025.43.16_suppl.6033; 5. Immunome investor presentation Jan 2026; 6. CytomX investor presentation; Padcev® – Astellas Pharma Inc. and Pfizer Inc., Datroway ® – Daiichi Sankyo and Astrazeneca, Tivdak® – Pfizer Inc and Genmab A/S, EmrelisTM – AbbVie, Elahere® – AbbVie Padcev1 and Datroway2, both auristatin ADC’s, leverage dose capping Elahere3 and other ADC’s in development from Pfizer4, Immunome5 and CytomX6 leverage AIBW
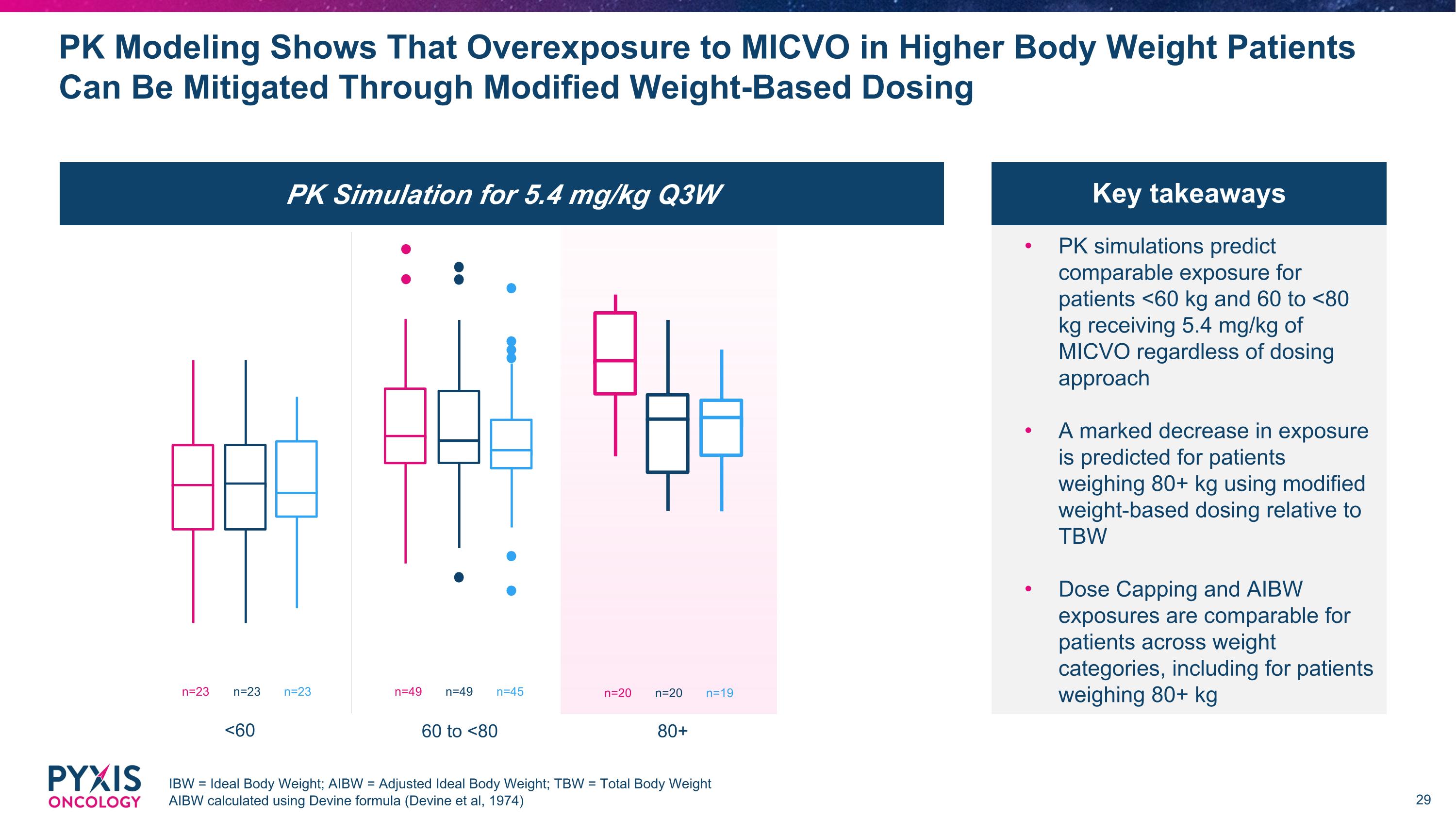
PK Simulation for 5.4 mg/kg Q3W PK Modeling Shows That Overexposure to MICVO in Higher Body Weight Patients Can Be Mitigated Through Modified Weight-Based Dosing IBW = Ideal Body Weight; AIBW = Adjusted Ideal Body Weight; TBW = Total Body Weight AIBW calculated using Devine formula (Devine et al, 1974) PK simulations predict comparable exposure for patients <60 kg and 60 to <80 kg receiving 5.4 mg/kg of MICVO regardless of dosing approach A marked decrease in exposure is predicted for patients weighing 80+ kg using modified weight-based dosing relative to TBW Dose Capping and AIBW exposures are comparable for patients across weight categories, including for patients weighing 80+ kg Key takeaways <60 n=23 n=23 n=23 60 to <80 n=49 n=49 n=45 80+ n=20 n=20 n=19
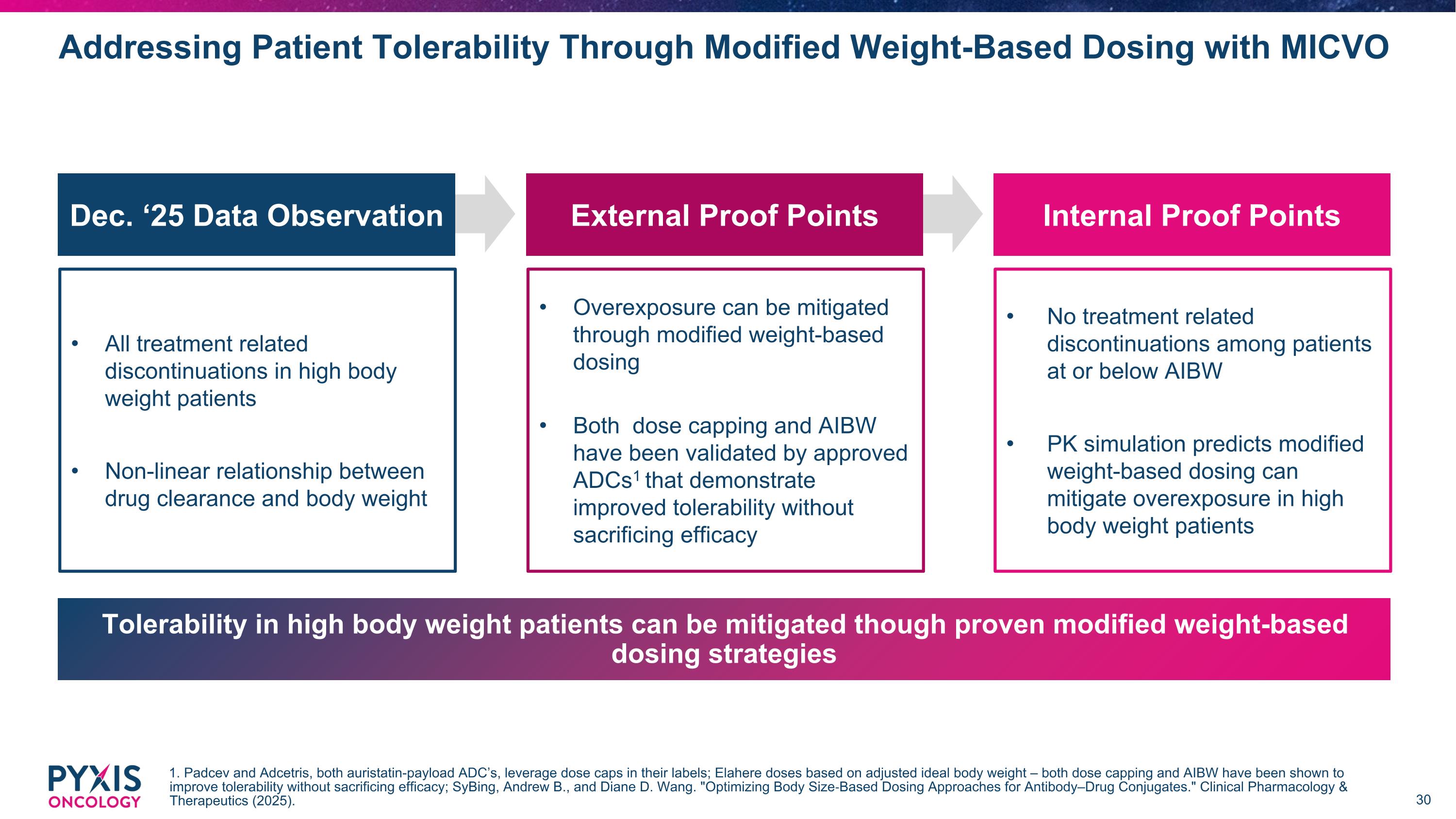
Overexposure can be mitigated through modified weight-based dosing Both dose capping and AIBW have been validated by approved ADCs1 that demonstrate improved tolerability without sacrificing efficacy Internal Proof Points No treatment related discontinuations among patients at or below AIBW PK simulation predicts modified weight-based dosing can mitigate overexposure in high body weight patients Addressing Patient Tolerability Through Modified Weight-Based Dosing with MICVO 1. Padcev and Adcetris, both auristatin-payload ADC’s, leverage dose caps in their labels; Elahere doses based on adjusted ideal body weight – both dose capping and AIBW have been shown to improve tolerability without sacrificing efficacy; SyBing, Andrew B., and Diane D. Wang. "Optimizing Body Size‐Based Dosing Approaches for Antibody–Drug Conjugates." Clinical Pharmacology & Therapeutics (2025). All treatment related discontinuations in high body weight patients Non-linear relationship between drug clearance and body weight External Proof Points Dec. ‘25 Data Observation Tolerability in high body weight patients can be mitigated though proven modified weight-based dosing strategies
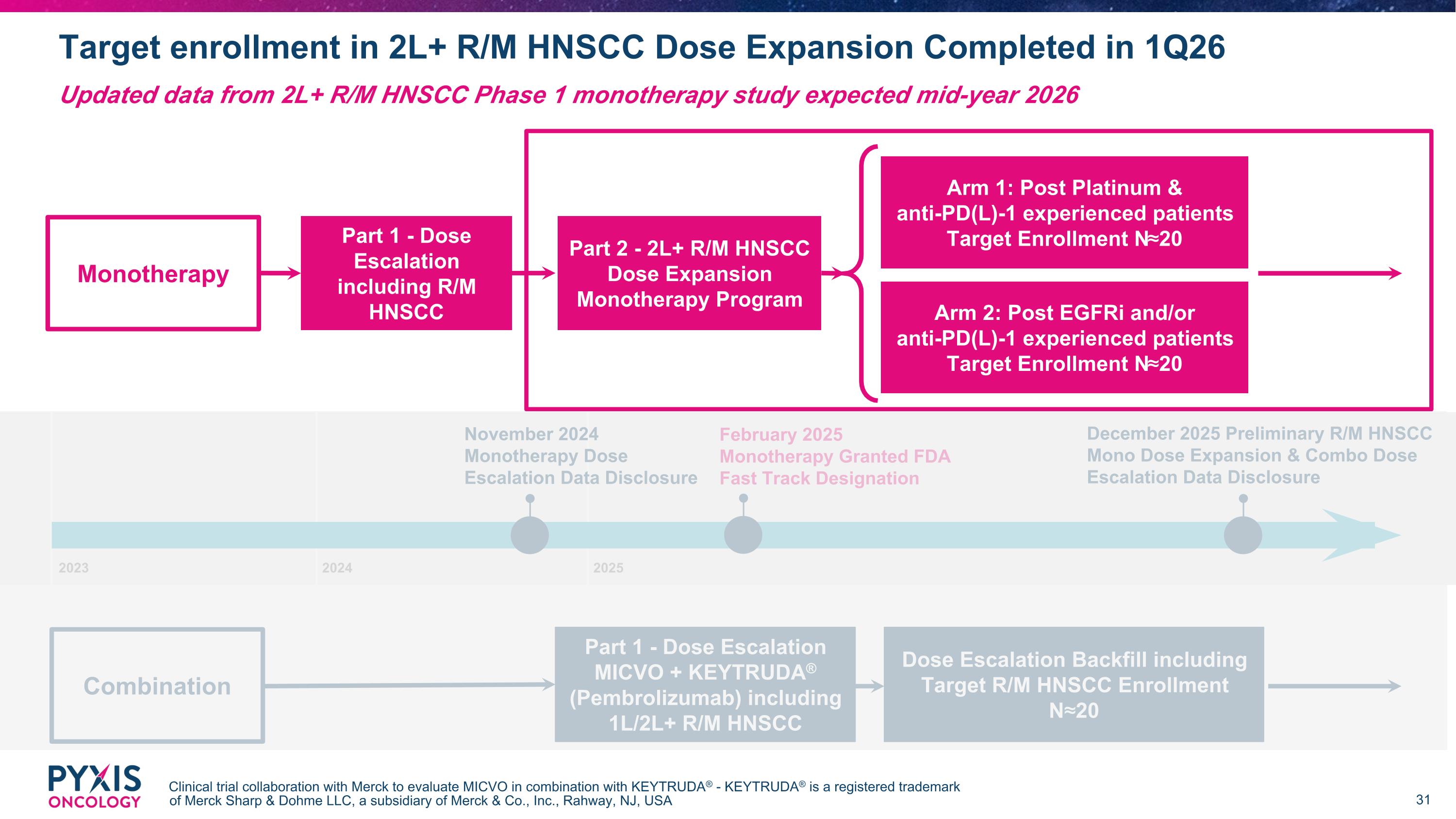
November 2024 Monotherapy Dose Escalation Data Disclosure December 2025 Preliminary R/M HNSCC Mono Dose Expansion & Combo Dose Escalation Data Disclosure Target enrollment in 2L+ R/M HNSCC Dose Expansion Completed in 1Q26 Clinical trial collaboration with Merck to evaluate MICVO in combination with KEYTRUDA® - KEYTRUDA® is a registered trademark of Merck Sharp & Dohme LLC, a subsidiary of Merck & Co., Inc., Rahway, NJ, USA Updated data from 2L+ R/M HNSCC Phase 1 monotherapy study expected mid-year 2026 Arm 1: Post Platinum & anti-PD(L)-1 experienced patients Target Enrollment N≈20 Part 2 - 2L+ R/M HNSCC Dose Expansion Monotherapy Program Part 1 - Dose Escalation MICVO + KEYTRUDA® (Pembrolizumab) including 1L/2L+ R/M HNSCC Part 1 - Dose Escalation including R/M HNSCC Dose Escalation Backfill including Target R/M HNSCC Enrollment N≈20 Monotherapy Combination Arm 2: Post EGFRi and/or anti-PD(L)-1 experienced patients Target Enrollment N≈20 2023 2024 2025 February 2025 Monotherapy Granted FDA Fast Track Designation

MICVO + Keytruda® in R/M HNSCC December 2025 Preliminary Data

MICVO + KEYTRUDA® Combination Summary in R/M HNSCC KEYTRUDA® is a registered trademark of Merck Sharp & Dohme LLC, a subsidiary of Merck & Co., Inc., Rahway, NJ, USA 71% Confirmed ORR, 100% DCR (n=7, 3.6 mg/kg & 4.4 mg/kg) Initial data support lack of overlapping toxicities observed between MICVO + KEYTRUDA® Significant potential in 1L+ in underserved patient populations Anticipated enrollment of HPV unrelated patients provides potential to build on promising HPV+ efficacy signal Future MICVO combinations may provide a further differentiated benefit/risk profile Data as of Nov 3, 2025
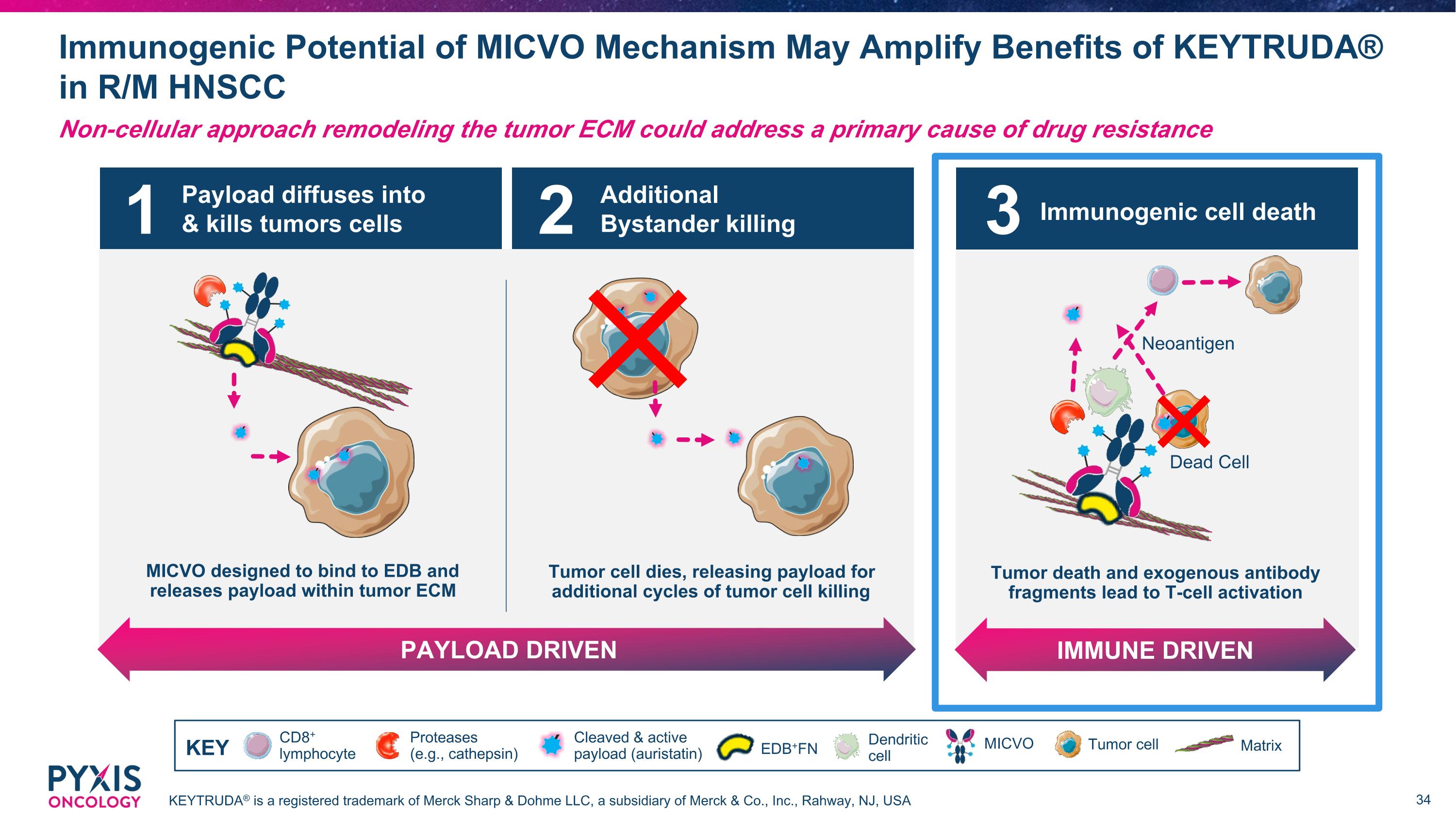
Payload diffuses into & kills tumors cells MICVO designed to bind to EDB and releases payload within tumor ECM Additional Bystander killing PAYLOAD DRIVEN Tumor cell dies, releasing payload for additional cycles of tumor cell killing 1 2 Immunogenic Potential of MICVO Mechanism May Amplify Benefits of KEYTRUDA® in R/M HNSCC KEYTRUDA® is a registered trademark of Merck Sharp & Dohme LLC, a subsidiary of Merck & Co., Inc., Rahway, NJ, USA Non-cellular approach remodeling the tumor ECM could address a primary cause of drug resistance CD8+ lymphocyte Dendritic cell MICVO Cleaved & active payload (auristatin) Tumor cell Matrix Proteases (e.g., cathepsin) KEY EDB+FN Immunogenic cell death IMMUNE DRIVEN Tumor death and exogenous antibody fragments lead to T-cell activation Dead Cell Neoantigen 3
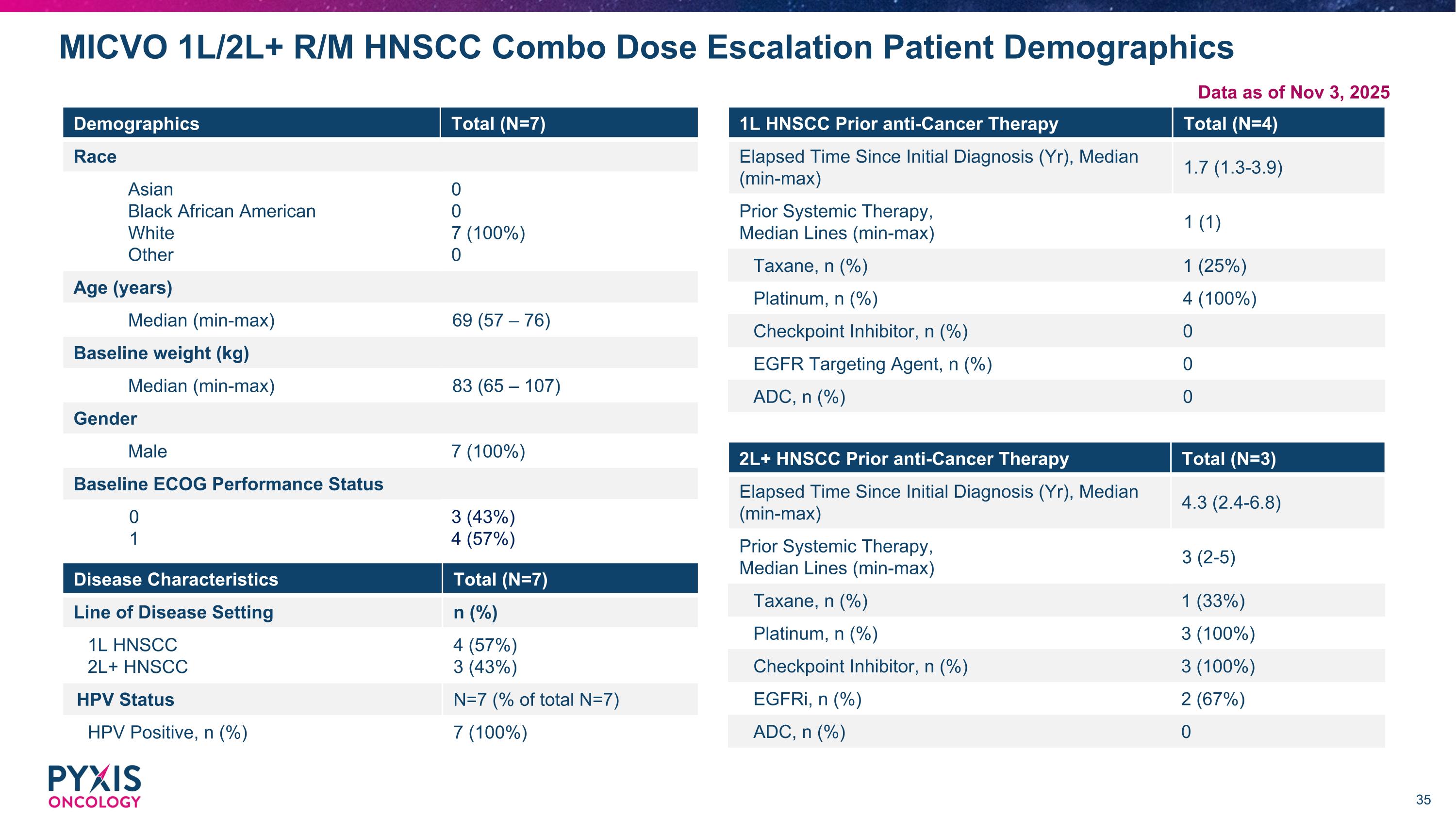
Demographics Total (N=7) Race Asian Black African American White Other 0 0 7 (100%) 0 Age (years) Median (min-max) 69 (57 – 76) Baseline weight (kg) Median (min-max) 83 (65 – 107) Gender Male 7 (100%) Baseline ECOG Performance Status 0 1 3 (43%) 4 (57%) MICVO 1L/2L+ R/M HNSCC Combo Dose Escalation Patient Demographics Disease Characteristics Total (N=7) Line of Disease Setting n (%) 1L HNSCC 2L+ HNSCC 4 (57%) 3 (43%) HPV Status N=7 (% of total N=7) HPV Positive, n (%) 7 (100%) 2L+ HNSCC Prior anti-Cancer Therapy Total (N=3) Elapsed Time Since Initial Diagnosis (Yr), Median (min-max) 4.3 (2.4-6.8) Prior Systemic Therapy, Median Lines (min-max) 3 (2-5) Taxane, n (%) 1 (33%) Platinum, n (%) 3 (100%) Checkpoint Inhibitor, n (%) 3 (100%) EGFRi, n (%) 2 (67%) ADC, n (%) 0 1L HNSCC Prior anti-Cancer Therapy Total (N=4) Elapsed Time Since Initial Diagnosis (Yr), Median (min-max) 1.7 (1.3-3.9) Prior Systemic Therapy, Median Lines (min-max) 1 (1) Taxane, n (%) 1 (25%) Platinum, n (%) 4 (100%) Checkpoint Inhibitor, n (%) 0 EGFR Targeting Agent, n (%) 0 ADC, n (%) 0 Data as of Nov 3, 2025
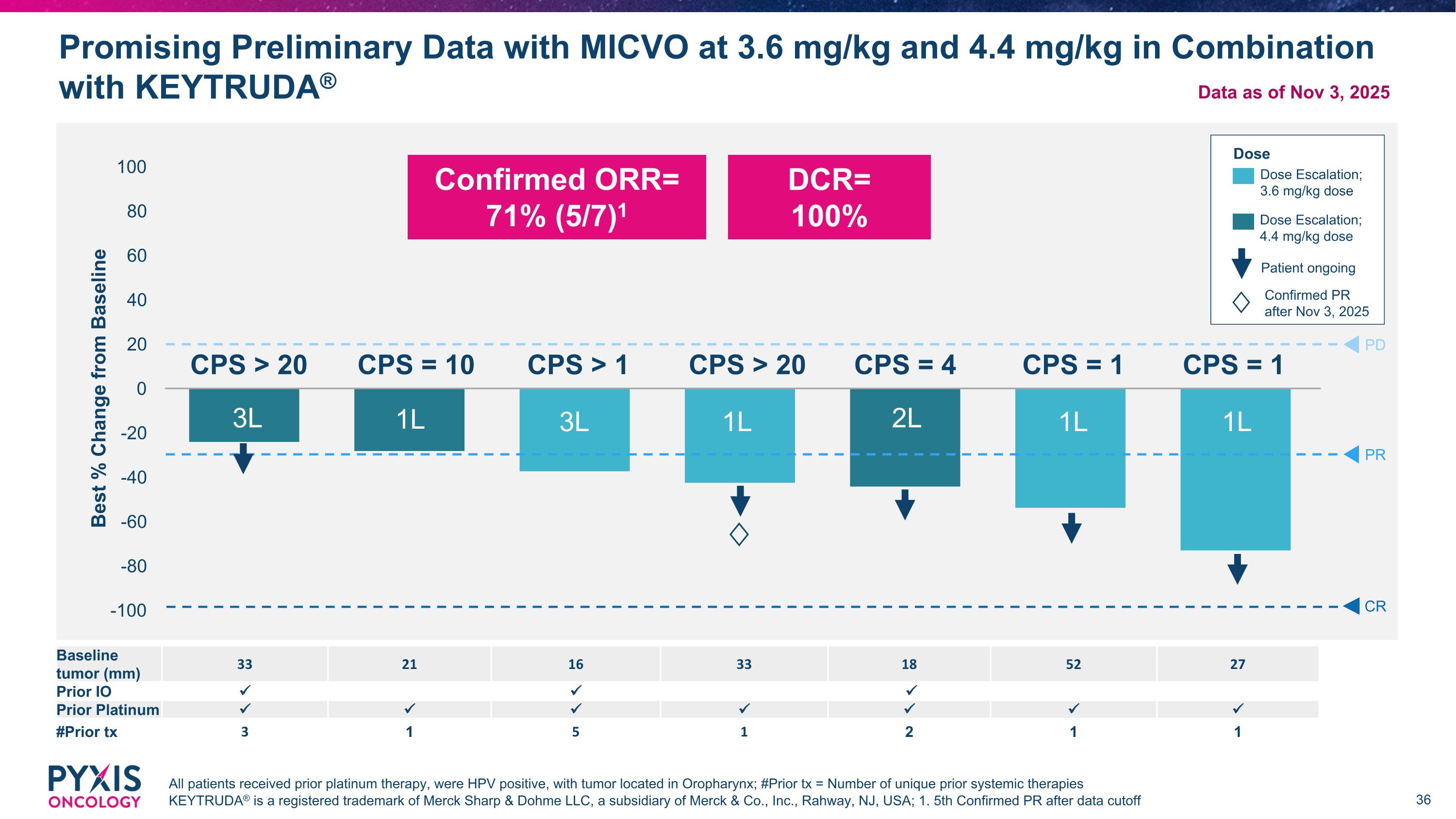
Promising Preliminary Data with MICVO at 3.6 mg/kg and 4.4 mg/kg in Combination with KEYTRUDA® All patients received prior platinum therapy, were HPV positive, with tumor located in Oropharynx; #Prior tx = Number of unique prior systemic therapies KEYTRUDA® is a registered trademark of Merck Sharp & Dohme LLC, a subsidiary of Merck & Co., Inc., Rahway, NJ, USA; 1. 5th Confirmed PR after data cutoff PD PR CR CPS = 1 CPS = 1 CPS = 10 3L 1L 3L 1L 2L 1L 1L Baseline tumor (mm) 33 21 16 33 18 52 27 Prior IO Prior Platinum #Prior tx 3 1 5 1 2 1 1 Dose Dose Escalation; 3.6 mg/kg dose Dose Escalation; 4.4 mg/kg dose DCR= 100% CPS > 20 CPS > 1 CPS > 20 CPS = 4 Confirmed ORR= 71% (5/7)1 Patient ongoing Confirmed PR after Nov 3, 2025 ♢ ♢ Data as of Nov 3, 2025
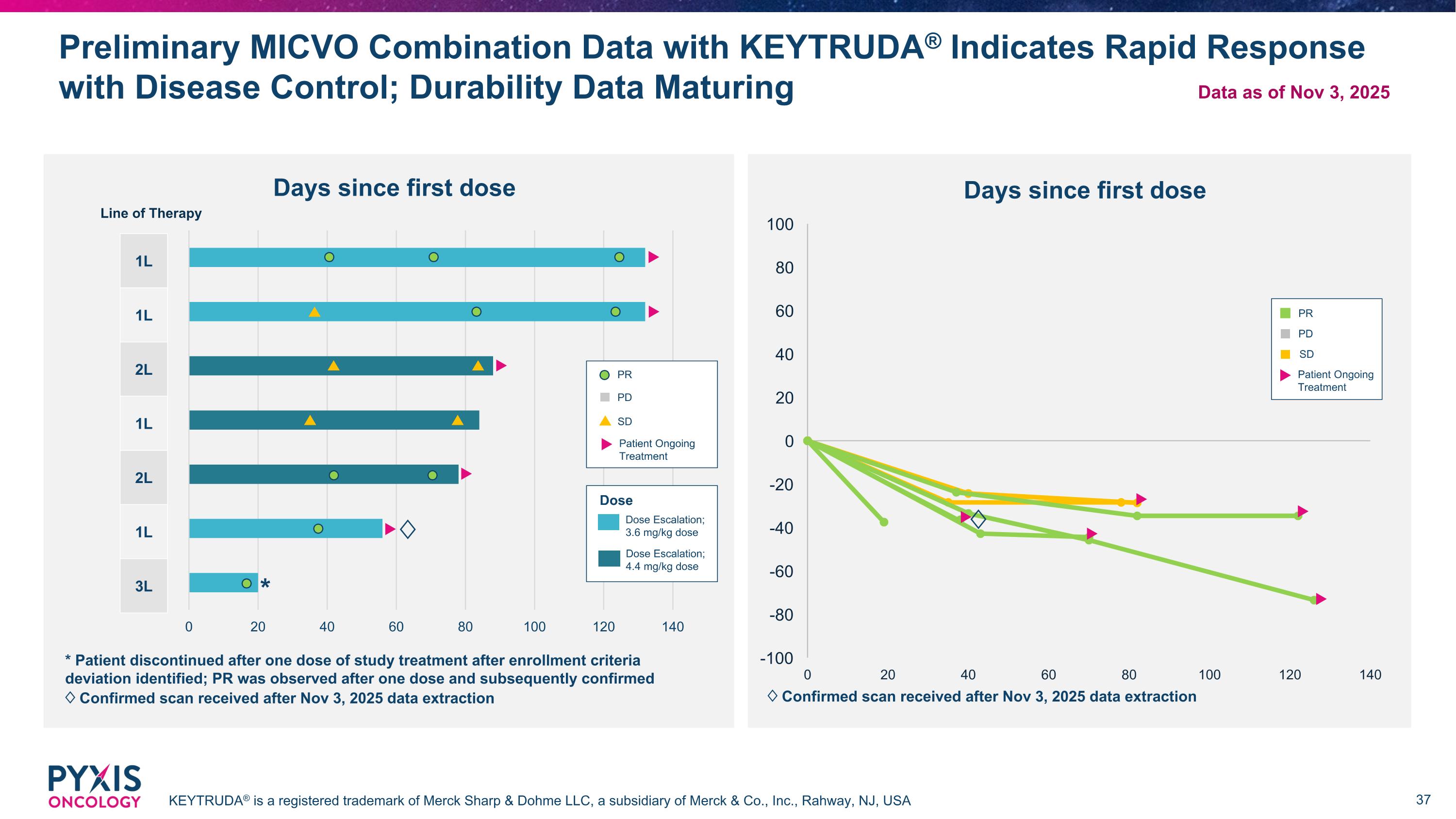
Preliminary MICVO Combination Data with KEYTRUDA® Indicates Rapid Response with Disease Control; Durability Data Maturing KEYTRUDA® is a registered trademark of Merck Sharp & Dohme LLC, a subsidiary of Merck & Co., Inc., Rahway, NJ, USA * Patient discontinued after one dose of study treatment after enrollment criteria deviation identified; PR was observed after one dose and subsequently confirmed ♢ Confirmed scan received after Nov 3, 2025 data extraction Dose Dose Escalation; 3.6 mg/kg dose Dose Escalation; 4.4 mg/kg dose 1L 1L 2L 1L 2L 1L 3L PR PD SD Patient Ongoing Treatment Line of Therapy PR SD PD Patient Ongoing Treatment * ♢ ♢ ♢ Confirmed scan received after Nov 3, 2025 data extraction Data as of Nov 3, 2025 Days since first dose
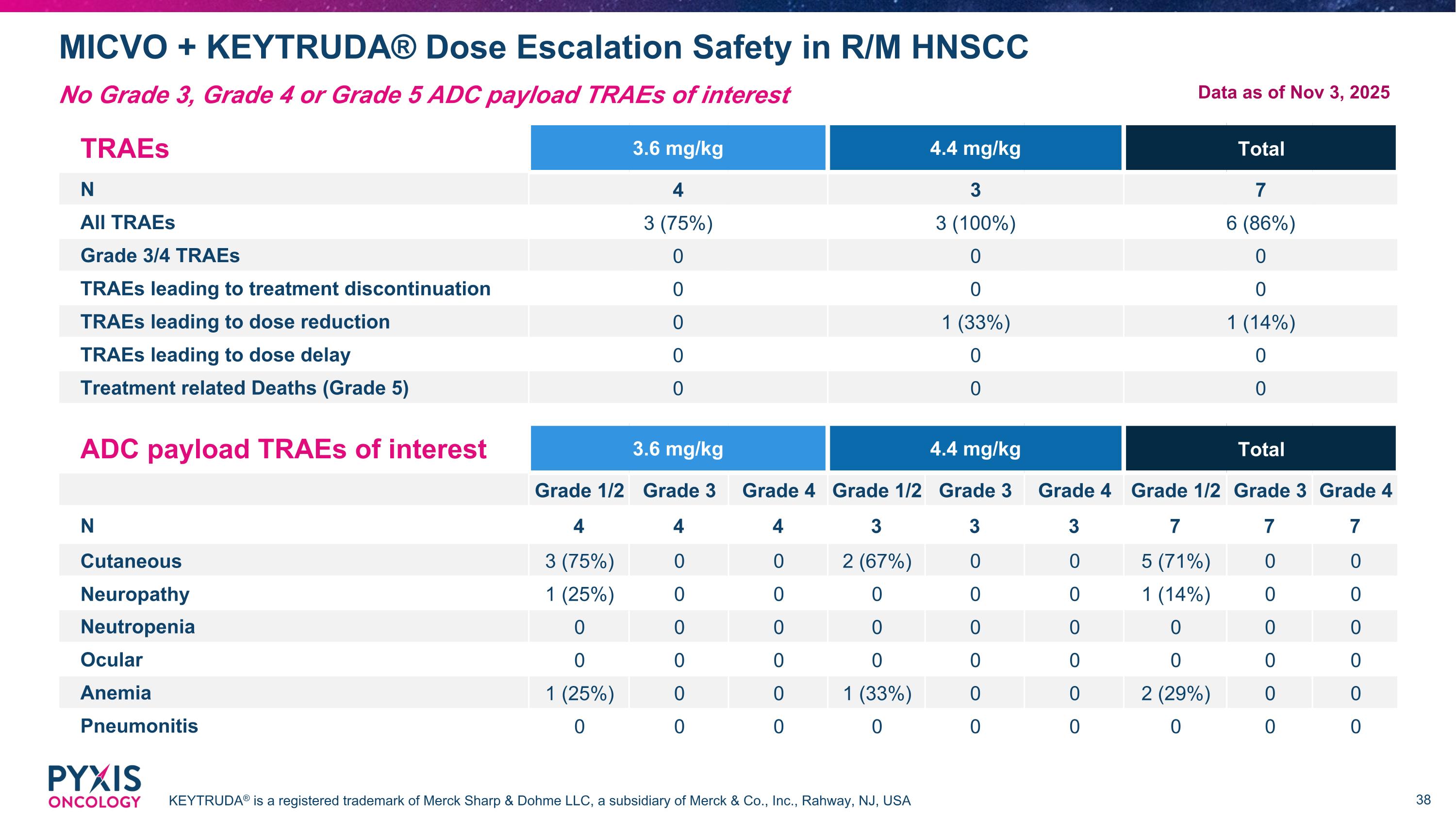
MICVO + KEYTRUDA® Dose Escalation Safety in R/M HNSCC KEYTRUDA® is a registered trademark of Merck Sharp & Dohme LLC, a subsidiary of Merck & Co., Inc., Rahway, NJ, USA No Grade 3, Grade 4 or Grade 5 ADC payload TRAEs of interest TRAEs 3.6 mg/kg 4.4 mg/kg Total N 4 3 7 All TRAEs 3 (75%) 3 (100%) 6 (86%) Grade 3/4 TRAEs 0 0 0 TRAEs leading to treatment discontinuation 0 0 0 TRAEs leading to dose reduction 0 1 (33%) 1 (14%) TRAEs leading to dose delay 0 0 0 Treatment related Deaths (Grade 5) 0 0 0 ADC payload TRAEs of interest 3.6 mg/kg 4.4 mg/kg Total Grade 1/2 Grade 3 Grade 4 Grade 1/2 Grade 3 Grade 4 Grade 1/2 Grade 3 Grade 4 N 4 4 4 3 3 3 7 7 7 Cutaneous 3 (75%) 0 0 2 (67%) 0 0 5 (71%) 0 0 Neuropathy 1 (25%) 0 0 0 0 0 1 (14%) 0 0 Neutropenia 0 0 0 0 0 0 0 0 0 Ocular 0 0 0 0 0 0 0 0 0 Anemia 1 (25%) 0 0 1 (33%) 0 0 2 (29%) 0 0 Pneumonitis 0 0 0 0 0 0 0 0 0 Data as of Nov 3, 2025

Multiple MICVO Clinical Data Milestones Expected in 2026 Updated Data from 2L+ R/M HNSCC Phase 1 Monotherapy Study 2H 2026 Updated Data from 1L/2L+ R/M HNSCC Phase 1/2 Dose Escalation Combination Study Mid 2026

Appendix
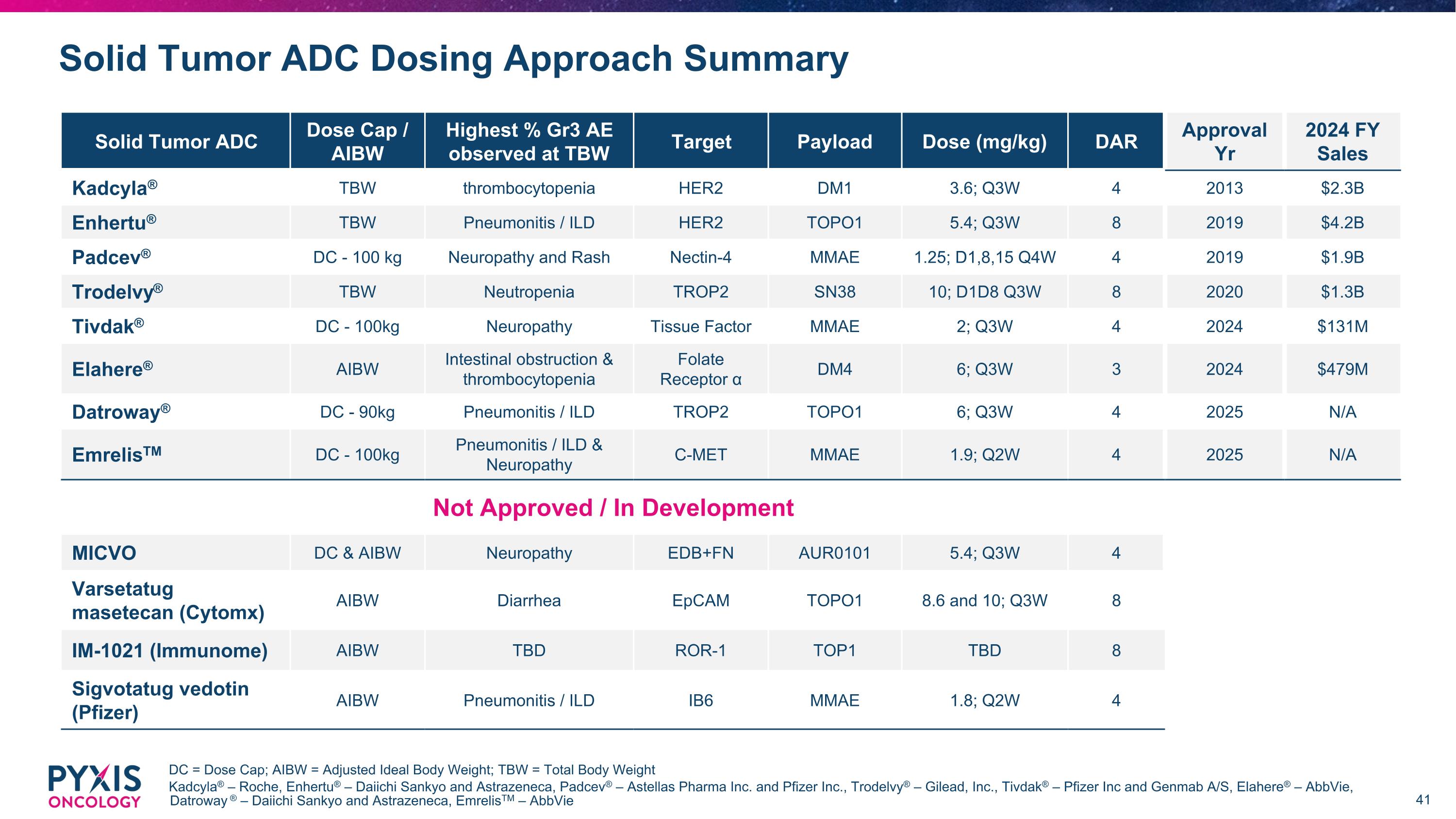
Solid Tumor ADC Dosing Approach Summary DC = Dose Cap; AIBW = Adjusted Ideal Body Weight; TBW = Total Body Weight Kadcyla® – Roche, Enhertu® – Daiichi Sankyo and Astrazeneca, Padcev® – Astellas Pharma Inc. and Pfizer Inc., Trodelvy® – Gilead, Inc., Tivdak® – Pfizer Inc and Genmab A/S, Elahere® – AbbVie, Datroway ® – Daiichi Sankyo and Astrazeneca, EmrelisTM – AbbVie Solid Tumor ADC Dose Cap / AIBW Highest % Gr3 AE observed at TBW Target Payload Dose (mg/kg) DAR Approval Yr 2024 FY Sales Kadcyla® TBW thrombocytopenia HER2 DM1 3.6; Q3W 4 2013 $2.3B Enhertu® TBW Pneumonitis / ILD HER2 TOPO1 5.4; Q3W 8 2019 $4.2B Padcev® DC - 100 kg Neuropathy and Rash Nectin-4 MMAE 1.25; D1,8,15 Q4W 4 2019 $1.9B Trodelvy® TBW Neutropenia TROP2 SN38 10; D1D8 Q3W 8 2020 $1.3B Tivdak® DC - 100kg Neuropathy Tissue Factor MMAE 2; Q3W 4 2024 $131M Elahere® AIBW Intestinal obstruction & thrombocytopenia Folate Receptor α DM4 6; Q3W 3 2024 $479M Datroway® DC - 90kg Pneumonitis / ILD TROP2 TOPO1 6; Q3W 4 2025 N/A EmrelisTM DC - 100kg Pneumonitis / ILD & Neuropathy C-MET MMAE 1.9; Q2W 4 2025 N/A Not Approved / In Development MICVO DC & AIBW Neuropathy EDB+FN AUR0101 5.4; Q3W 4 Varsetatug masetecan (Cytomx) AIBW Diarrhea EpCAM TOPO1 8.6 and 10; Q3W 8 IM-1021 (Immunome) AIBW TBD ROR-1 TOP1 TBD 8 Sigvotatug vedotin (Pfizer) AIBW Pneumonitis / ILD IB6 MMAE 1.8; Q2W 4

Recent Translational Posters Build on Previous Publications to Further Support Three-Pronged MOA of MICVO [1] Hooper A et al., Mol Cancer Ther (2022) 21(9):1462-1472 [2] Shen C & Iovino M et al., AACR-NCI-EORTC (2025) [3] Graziani EI et al., Mol Cancer Ther (2020) 19: 2068-78 [4] Iovino M et al., AACR-NCI-EORTC (2025) [5] Lewandowski S et al, AACR (2025). [6] Maderna A et al., J Med Chem (2014) 57: 10527-10543 [7] Trickett J et al, AACR-NCI-EORTC (2025) [8] Lewandowski S et al, AACR-NCI-EORTC (2025) [9] Rodriguez A et al, AACR-NCI-EORTC (2025) MICVO acts as a driver for the cancer-immunity cycle, inducing immunogenic cell death, activating immune cells and allowing tumor infiltration of T cells [4] Preclinical data support complementary potential with immune checkpoint inhibitors Mouse analog of MICVO showed immune response in tumors that had been refractory to anti-PD1 [9] Synergistic antitumor activity when combined with anti-PD1 [9] 1 2 3 Payload diffuses into & kills tumors cells Additional Bystander killing Immunogenic cell death Highly specific and avidity driven binding to EDB+FN [1, 2] Lack of drug sink and no off-target binding support minimal off target effects Strong binding strength to EDB+FN fibrils predicts the prolonged drug retention in TME for heightened clinical efficacy Extracellular payload release mediated by tumor-specific cathepsins [2] Improved membrane permeability for cancer cell diffusion and efficient bystander killing [2,3,4,5] Optimized payload potency by rational structure-based drug design (SBDD) to increase tumor cell killing [6] pH-dependency favors linker cleavage in acidic TME to minimize off-target toxicity [2] Observed changes to tumor stromal architecture indicate potential for extracellular mechanism to lead to unique TME remodeling and improve tumor response [7,8] Payload Driven Immune Driven
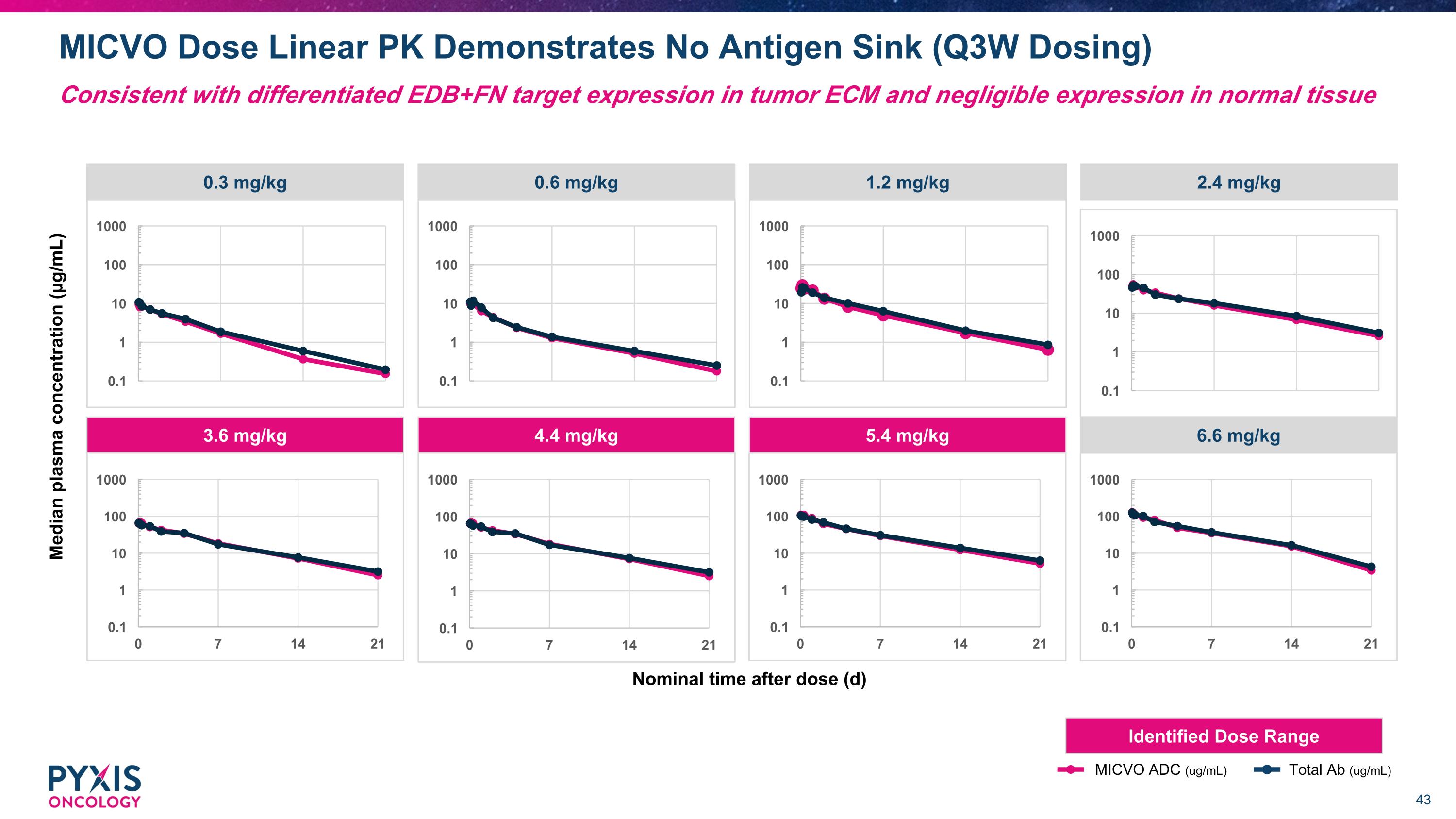
Nominal time after dose (d) Median plasma concentration (µg/mL) 0.3 mg/kg 0.6 mg/kg 1.2 mg/kg 2.4 mg/kg 3.6 mg/kg 4.4 mg/kg 5.4 mg/kg 6.6 mg/kg MICVO ADC (ug/mL) Total Ab (ug/mL) Identified Dose Range MICVO Dose Linear PK Demonstrates No Antigen Sink (Q3W Dosing) Consistent with differentiated EDB+FN target expression in tumor ECM and negligible expression in normal tissue