| Universally Implantable Regenerative Human Tissue |
| 2 Disclaimer These slides and the accompanying oral presentation contain forward-looking statements. All statements, other than statements of historical fact, included in these slides and the accompanying oral presentation are forward-looking statements reflecting management’s current beliefs and expectations. In some cases, you can identify forward-looking statements by terminology such as “will,” “anticipate,” “expect,” “believe,” “intend” and “should” or the negative of these terms or other comparable terminology. Forward-looking statements in these slides and the accompanying oral presentation include, but are not limited to, statements about our plans and ability to commercialize our bioengineered acellular tissue engineered vessels (“ATEV s”) in the United States under the brand name Symvess in vascular trauma repair; the anticipated commercialization of our ATEVs and our ability to manufacture ATEVs and other product candidates in sufficient quantities to satisfy our clinical trial and commercial needs; our plans and ability to execute product development, process development and preclinical development efforts successfully and on our anticipated timelines; our plans, anticipated timelines and ability to obtain marketing approval from the U.S. Food and Drug Administration (“FDA”) and other regulatory authorities, including the European Medicines Agency and Israel, for our ATEVs in other indications and other product candidates; our ability to design, initiate and successfully complete clinical trials and other studies for our product candidates and our plans and expectations regarding our ongoing or planned preclinical and clinical trials; the outcome of our ongoing discussions with the FDA concerning the design of our clinical trials; our anticipated growth rate and market opportunities; the potential liquidity and trading of our securities; our ability to raise additional capital in the future; our ability to use our proprietary scientific technology platform to build a pipeline of additional product candidates; the anticipated characteristics and performance of our ATEVs; the expected size of the target populations and addressable markets for our product candidates; the anticipated benefits of our ATEVs relative to existing alternatives; our assessment of the competitive landscape; the degree of market acceptance of ATEVs and the availability of third-party coverage and reimbursement; the implementation of our business model and strategic plans for our business; our expectations regarding our strategic partnership with Fresenius Medical Care Holdings, Inc. to sell, market and distribute our 6 millimeter ATEV for certain specified indications and in specified markets; the performance of other third parties on which we rely, including our third-party manufacturers, our licensors, our suppliers and the organizations conducting our clinical trials; our ability to obtain and maintain intellectual property protection for our product candidates as well as our ability to operate our business without infringing, misappropriating or otherwise violating the intellectual property rights of others; our ability to maintain the confidentiality of our trade secrets, particularly with respect to our manufacturing process; our compliance with applicable laws and regulatory requirements, including FDA regulations, healthcare laws and regulations, and anti-corruption laws; our ability to attract, retain and motivate qualified personnel and to manage our growth effectively; our future financial performance and capital requirements; our ability to implement and maintain effective internal controls; and the impact of the overall global economy and increasing interest rates and inflation on our business. These statements relate to future events or to our future financial performance and involve known and unknown risks, uncertainties and other factors that may cause our actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by these forward-looking statements. The potential risks and uncertainties that could cause actual results to differ from the results predicted include, among others, those risks and uncertainties included under the captions “Risk Factors” and “Management’s Discussion and Analysis of Financial Condition and Results of Operations” in our Form 10-K for the year ended December 31, 2024, our quarterly report on Form 10-Q for the quarter ended September 30, 2025, each filed by Humacyte with the Securities and Exchange Commission, and in future filings made with the Securities and Exchange Commission from time to time. Any forward-looking statements contained herein are based on assumptions that we believe to be reasonable as of the date hereof. Except as required by law, we assume no obligation to update these forward-looking statements, even if new information becomes available in the future. This presentation shall not constitute an offer to sell or the solicitation of an offer to buy, nor shall there be any sale of our securities, in any state or other jurisdiction in which such offer, solicitation or sale would be unlawful prior to the reregistration or qualification under the securities laws of any such state or other jurisdiction. |
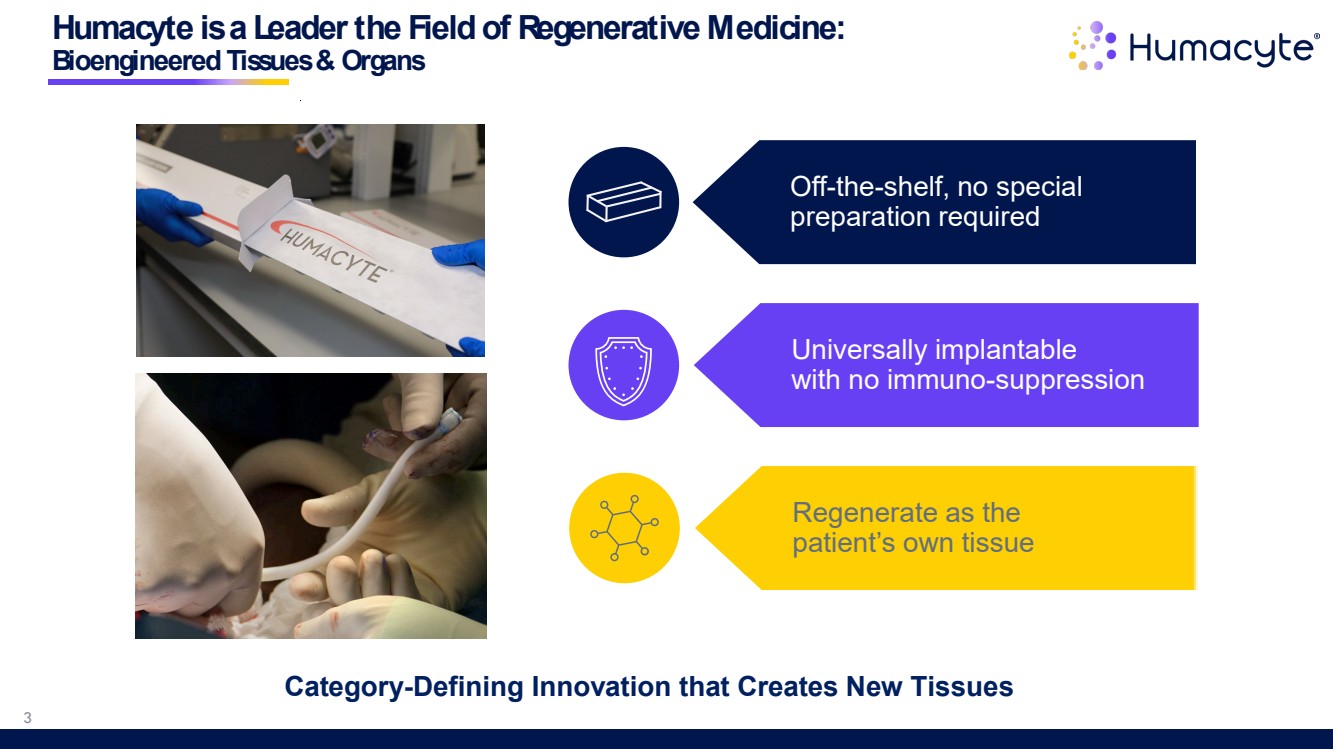
| Humacyte is a Leader the Field of Regenerative Medicine: Bioengineered Tissues & Organs Off-the-shelf, no special preparation required Universally implantable with no immuno-suppression Regenerate as the patient’s own tissue Category-Defining Innovation that Creates New Tissues 3 |
| Universally Implantable Regenerative Human Tissue U.S. FDA Approved FDA approved Symvess® (ATEV) BLA in December 2024 for treatment of extremity vascular trauma; U.S. market launch commenced 2025 First-in-Class Technology and Manufacturing Platform Large addressable markets trauma, dialysis, peripheral artery disease, diabetes, coronary bypass Commercial-Scale Manufacturing Commercial-scale manufacturing in place with annual capacity of up to 40,000 ATEVs in existing facility Validated through Multiple Partnerships 4 |
| Humacyte Leadership & Board Leadership Team Board of Directors Kathleen Sebelius Chair of the Board John P. Bamforth, PhD Emery N. Brown, MD, PhD Michael T. Constantino Brady W. Dougan Charles Bruce Green, MD Keith Anthony Jones, M.D., Laura E. Niklason, MD, PhD Todd M. Pope Diane Seimetz, PhD Max Wallace, JD Susan Windham-Bannister, PhD Laura E. Niklason, MD, PhD Founder, President, Chief Executive Officer Dale Sander Chief Financial Officer, Chief Corporate Development Officer Shamik Parikh, MD Chief Medical Officer Sabrina Osborne Chief People Officer Heather Connelly Chief Quality Officer Prior Experience 5 Rick McElheny SVP, Business Development Lisa Molyneux EVP, Enterprise Planning & Analysis |
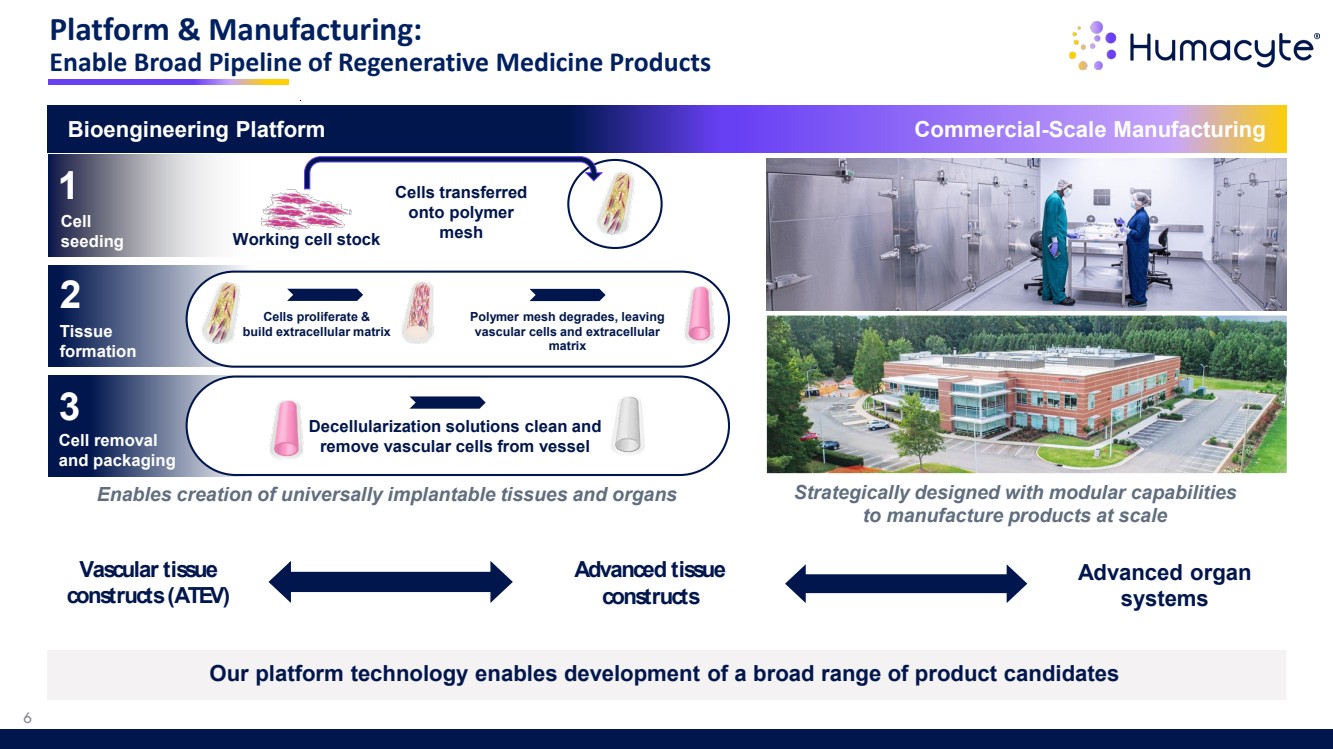
| Platform & Manufacturing: Enable Broad Pipeline of Regenerative Medicine Products Vascular tissue constructs (ATEV) Advanced tissue constructs Advanced organ systems Bioengineering Platform Cell seeding Tissue formation Cell removal and packaging 1 2 3 Working cell stock Cells transferred onto polymer mesh Cells proliferate & build extracellular matrix Polymer mesh degrades, leaving vascular cells and extracellular matrix Decellularization solutions clean and remove vascular cells from vessel Commercial-Scale Manufacturing Strategically designed with modular capabilities to manufacture products at scale Enables creation of universally implantable tissues and organs Our platform technology enables development of a broad range of product candidates 6 |
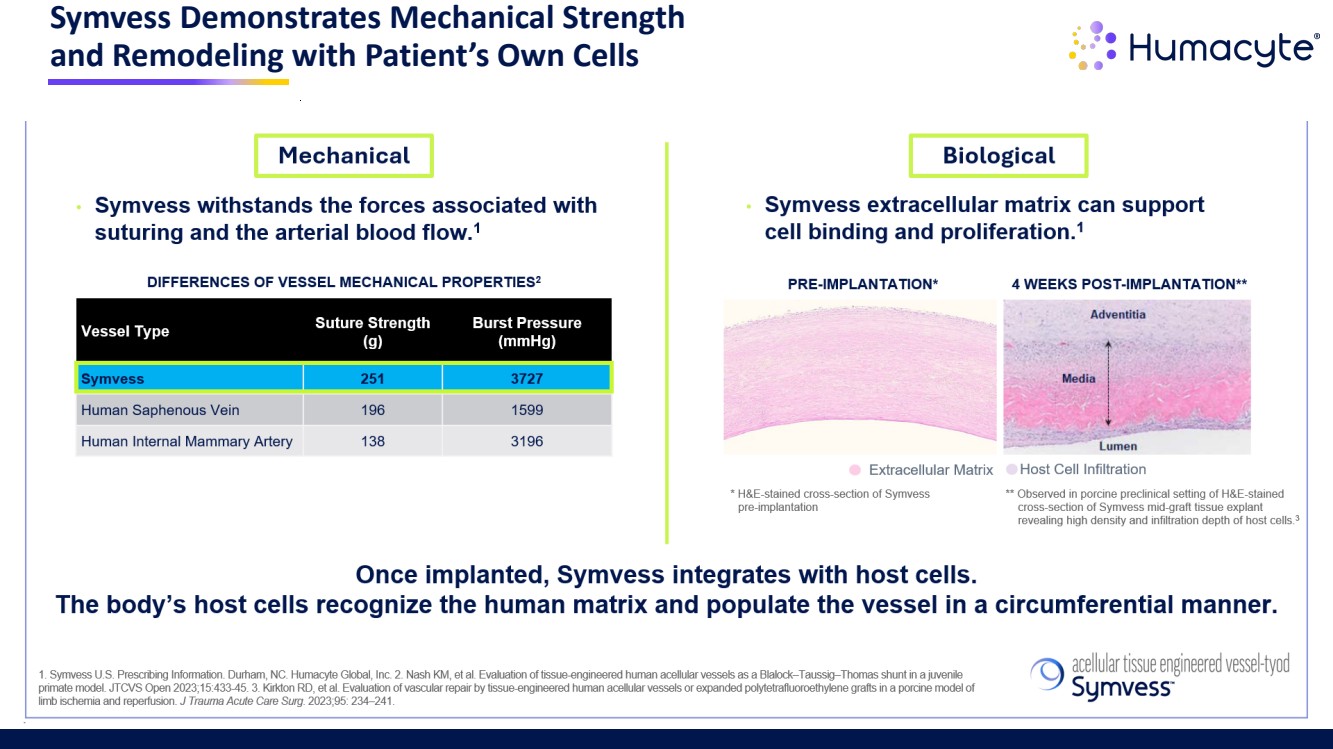
| Symvess Demonstrates Mechanical Strength and Remodeling with Patient’s Own Cells 7 |
| Pipeline with Multiple Potential Commercial Launches Preclinical Phase 1/2 Phase 3 Approved Vascular Tissue Constructs Trauma Dialysis (AV Access) PAD CABG Pediatric Heart Disease Complex Tissue Constructs Urinary Conduit Tracheal Replacement Esophageal Replacement Complex Organ Systems BioVascular Pancreas (T1D) Lung Approved by FDA V007 Phase 3 Trial Met Co-Primary Endpoints Three Phase 2 Studies Completed 8 V012 Phase 3 Trial in Women – Interim Results 2Q26 Dialysis (AV Access) ATEV CTEV |
| 9 Vascular Trauma FDA Approved in Extremity Vascular Trauma Symvess acellular tissue engineered vessel-tyod |
| Symvess is FDA Approved in Extremity Vascular Trauma Repopulates with the patient’s cells1-2,3 Low susceptibility to infection4 No immune response observed1-3,5 Off-the-shelf, ready to use1,3 Low amputation results1 INDICATION SYMVESS is an acellular tissue engineered vessel indicated for use in adults as a vascular conduit for extremity arterial injury when urgent revascularization is needed to avoid imminent limb loss, and autologous vein graft is not feasible. PLEASE SEE ACCOMPANYING FULL PRESCRIBING INFORMATION AT SYMVESS.COM, INCLUDING BOXED WARNING. REFERENCES: 1. Symvess U.S. Prescribing Information. Durham, NC. Humacyte Global, Inc. 2. Kirkton RD, et al. Bioengineered human acellular vessels recellularize and evolve into living blood vessels after human implantation. Sci Transl Med. 2019;11(485):eaau6934. 3. Dahl S, et al. Readily available tissue-engineered vascular grafts. Sci Transl Med. 2011 Feb 2;3(68):68ra9. 4. Wang J, et al. Biological mechanisms of infection resistance in tissue engineered blood vessels compared to synthetic expanded polytetrafluoroethylene grafts. JVS Vasc Sci. 2023;4:100120. 5. Moore EE, et al. Bioengineered Human Arteries for the Repair of Vascular Injuries. JAMA Surg. 2024 Nov 20:e244893. 10 |
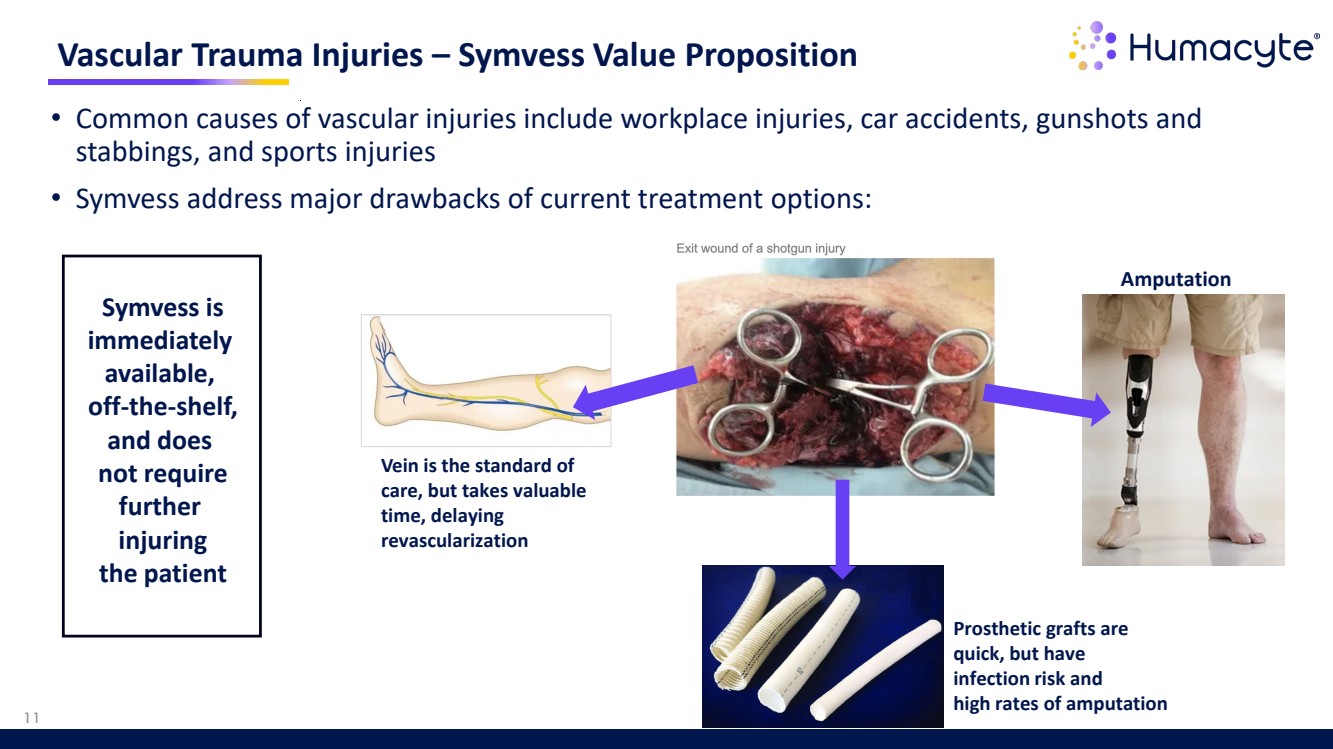
| • Common causes of vascular injuries include workplace injuries, car accidents, gunshots and stabbings, and sports injuries • Symvess address major drawbacks of current treatment options: Vascular Trauma Injuries – Symvess Value Proposition Vein is the standard of care, but takes valuable time, delaying revascularization Prosthetic grafts are quick, but have infection risk and high rates of amputation Amputation 11 Symvess is immediately available, off-the-shelf, and does not require further injuring the patient |
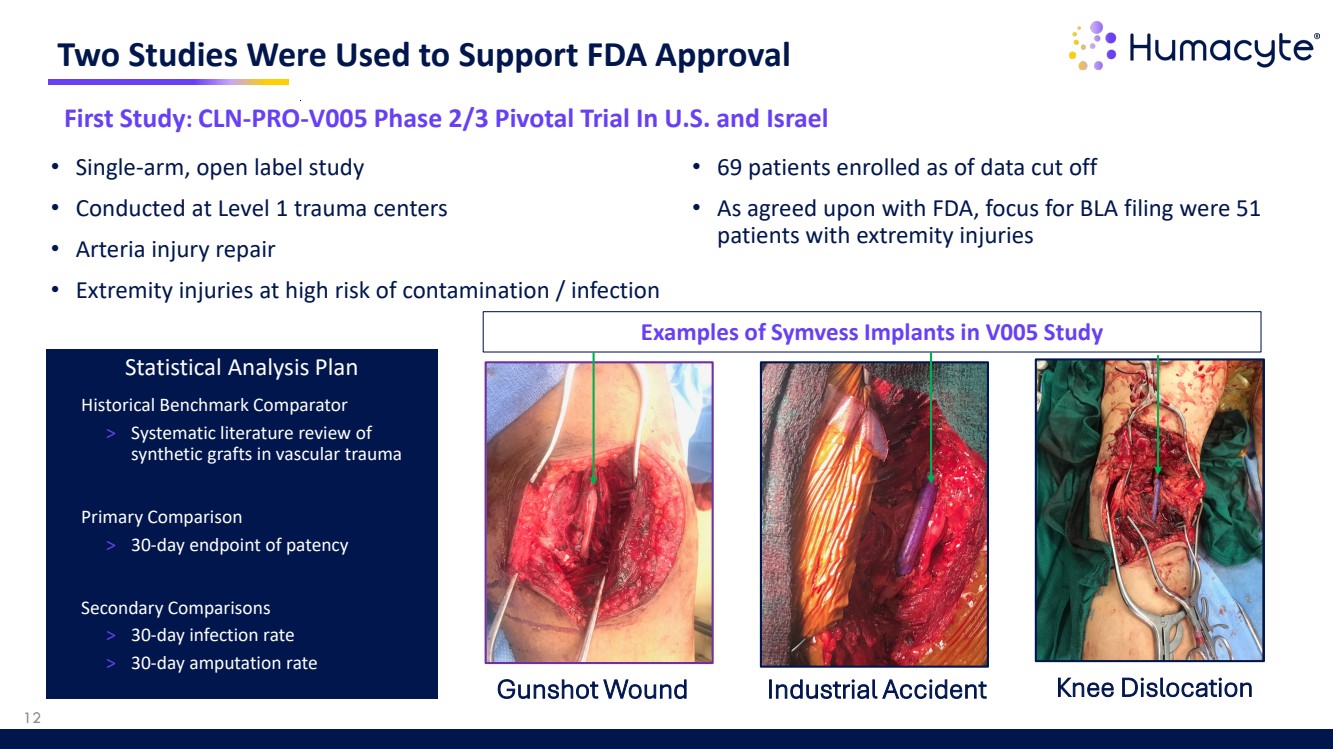
| Two Studies Were Used to Support FDA Approval Gunshot Wound Industrial Accident Knee Dislocation 12 First Study: CLN-PRO-V005 Phase 2/3 Pivotal Trial In U.S. and Israel • Single-arm, open label study • Conducted at Level 1 trauma centers • Arteria injury repair • Extremity injuries at high risk of contamination / infection Statistical Analysis Plan • Historical Benchmark Comparator > Systematic literature review of synthetic grafts in vascular trauma • Primary Comparison > 30-day endpoint of patency • Secondary Comparisons > 30-day infection rate > 30-day amputation rate • 69 patients enrolled as of data cut off • As agreed upon with FDA, focus for BLA filing were 51 patients with extremity injuries Examples of Symvess Implants in V005 Study |
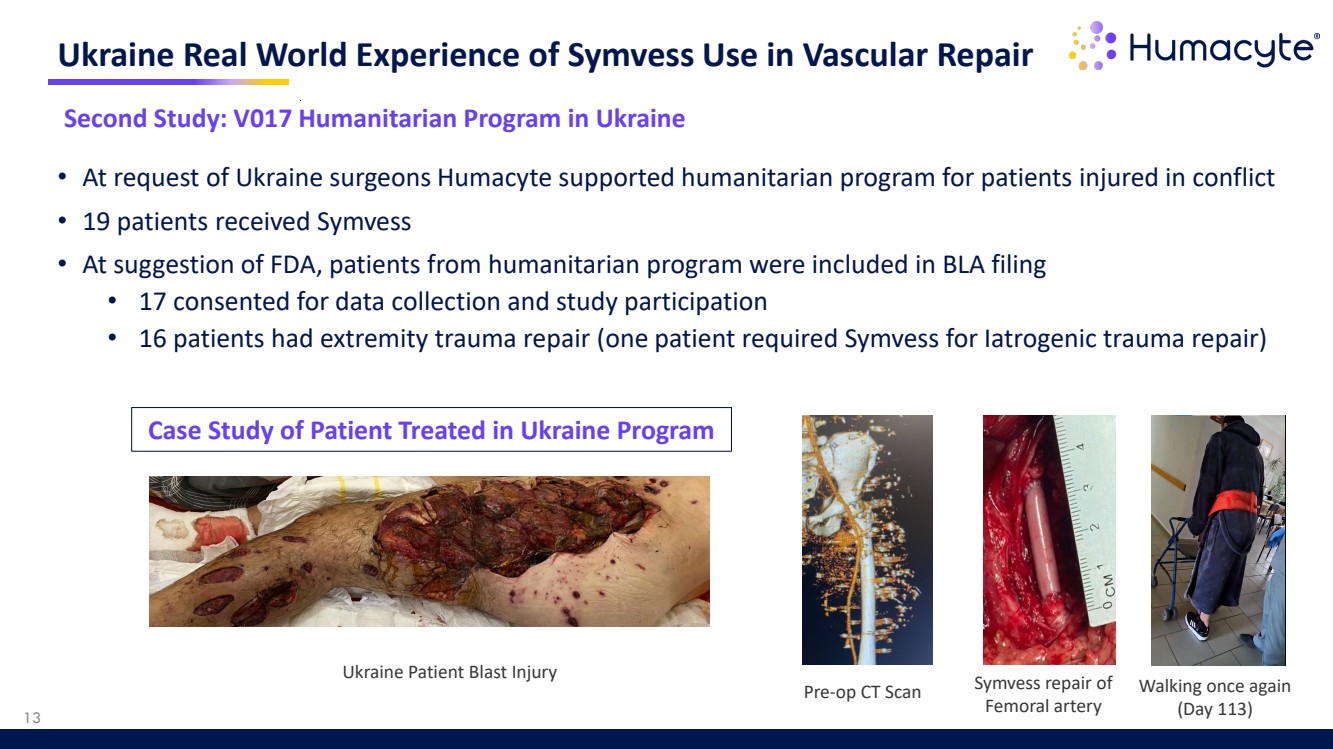
| • At request of Ukraine surgeons Humacyte supported humanitarian program for patients injured in conflict • 19 patients received Symvess • At suggestion of FDA, patients from humanitarian program were included in BLA filing • 17 consented for data collection and study participation • 16 patients had extremity trauma repair (one patient required Symvess for Iatrogenic trauma repair) Ukraine Real World Experience of Symvess Use in Vascular Repair Pre-op CT Scan Symvess repair of Femoral artery Ukraine Patient Blast Injury Walking once again (Day 113) 13 Second Study: V017 Humanitarian Program in Ukraine Case Study of Patient Treated in Ukraine Program |
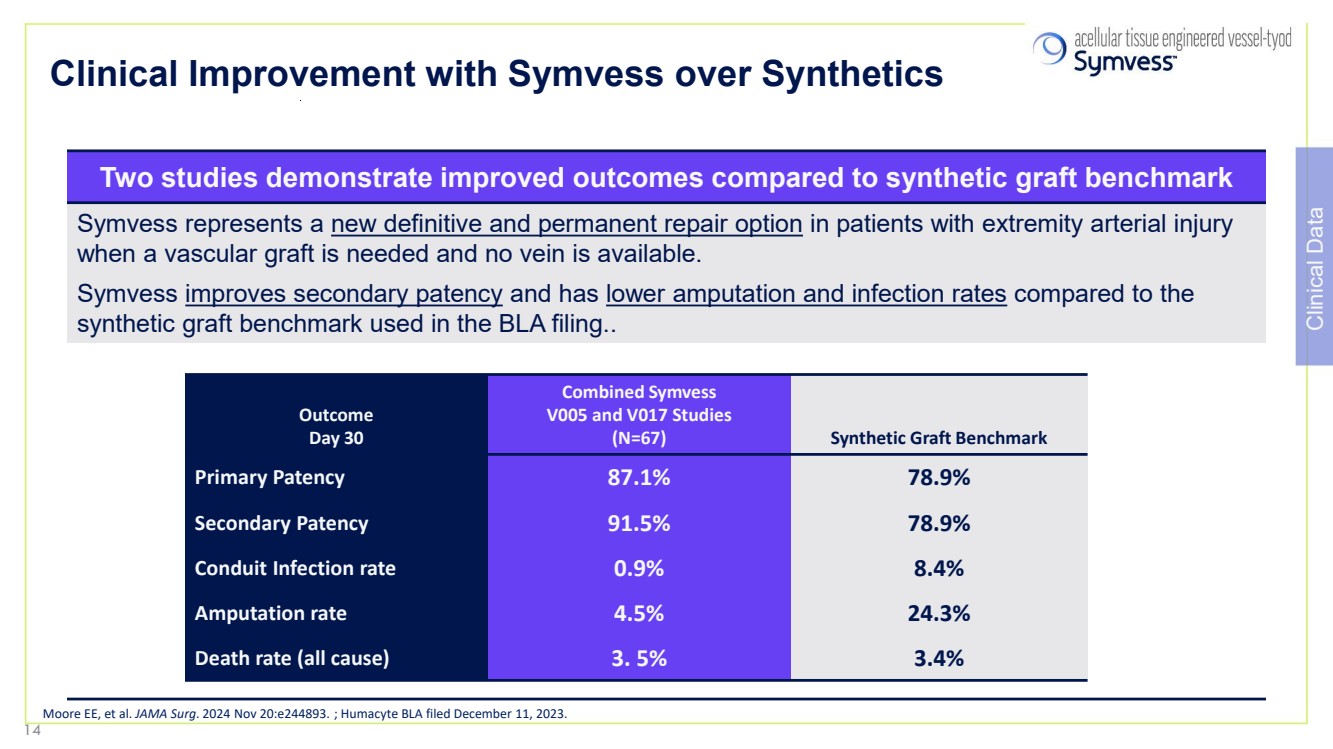
| Clinical Improvement with Symvess over Synthetics Two studies demonstrate improved outcomes compared to synthetic graft benchmark Symvess represents a new definitive and permanent repair option in patients with extremity arterial injury when a vascular graft is needed and no vein is available. Symvess improves secondary patency and has lower amputation and infection rates compared to the synthetic graft benchmark used in the BLA filing.. Outcome Day 30 Combined Symvess V005 and V017 Studies (N=67) Synthetic Graft Benchmark Primary Patency 87.1% 78.9% Secondary Patency 91.5% 78.9% Conduit Infection rate 0.9% 8.4% Amputation rate 4.5% 24.3% Death rate (all cause) 3. 5% 3.4% Moore EE, et al. JAMA Surg. 2024 Nov 20:e244893. ; Humacyte BLA filed December 11, 2023. Clinical Data 14 |
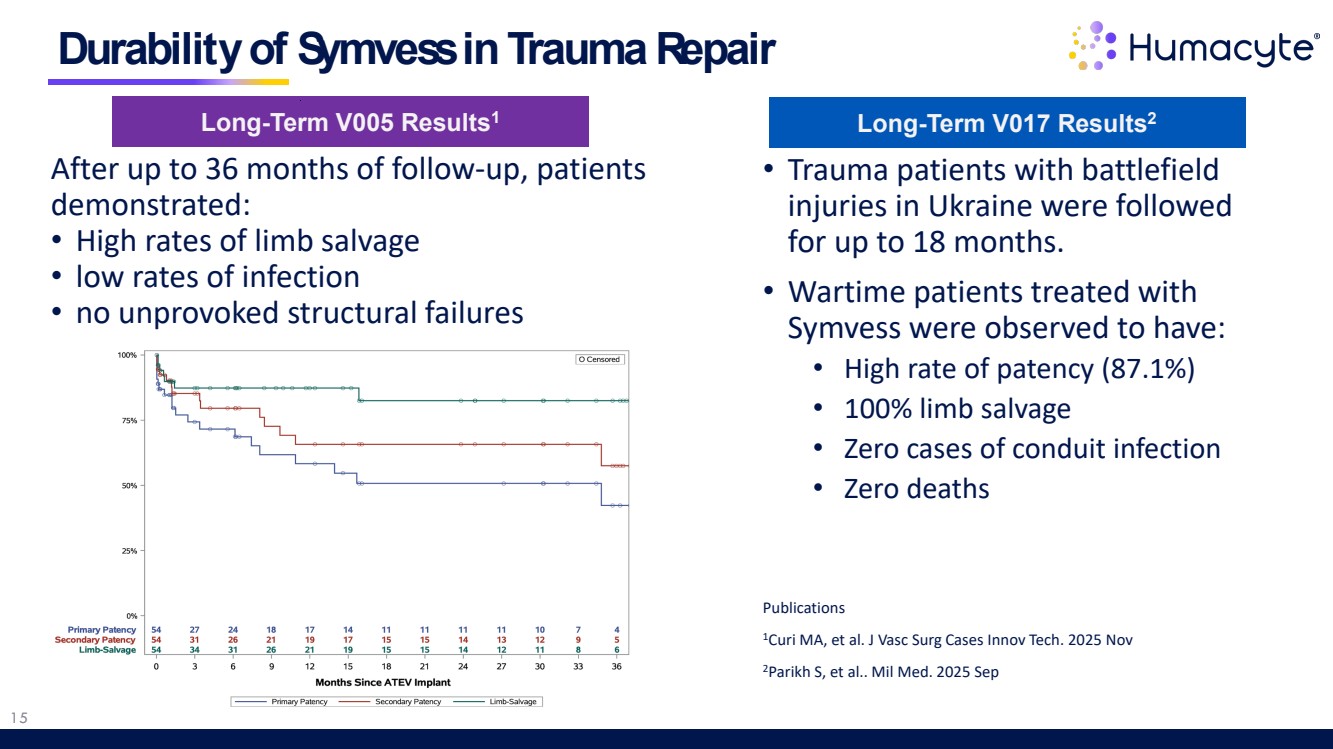
| 15 After up to 36 months of follow-up, patients demonstrated: • High rates of limb salvage • low rates of infection • no unprovoked structural failures Durability of Symvess in Trauma Repair Long-Term V005 Results1 Long-Term V017 Results2 • Trauma patients with battlefield injuries in Ukraine were followed for up to 18 months. • Wartime patients treated with Symvess were observed to have: • High rate of patency (87.1%) • 100% limb salvage • Zero cases of conduit infection • Zero deaths Publications 1Curi MA, et al. J Vasc Surg Cases Innov Tech. 2025 Nov 2Parikh S, et al.. Mil Med. 2025 Sep |
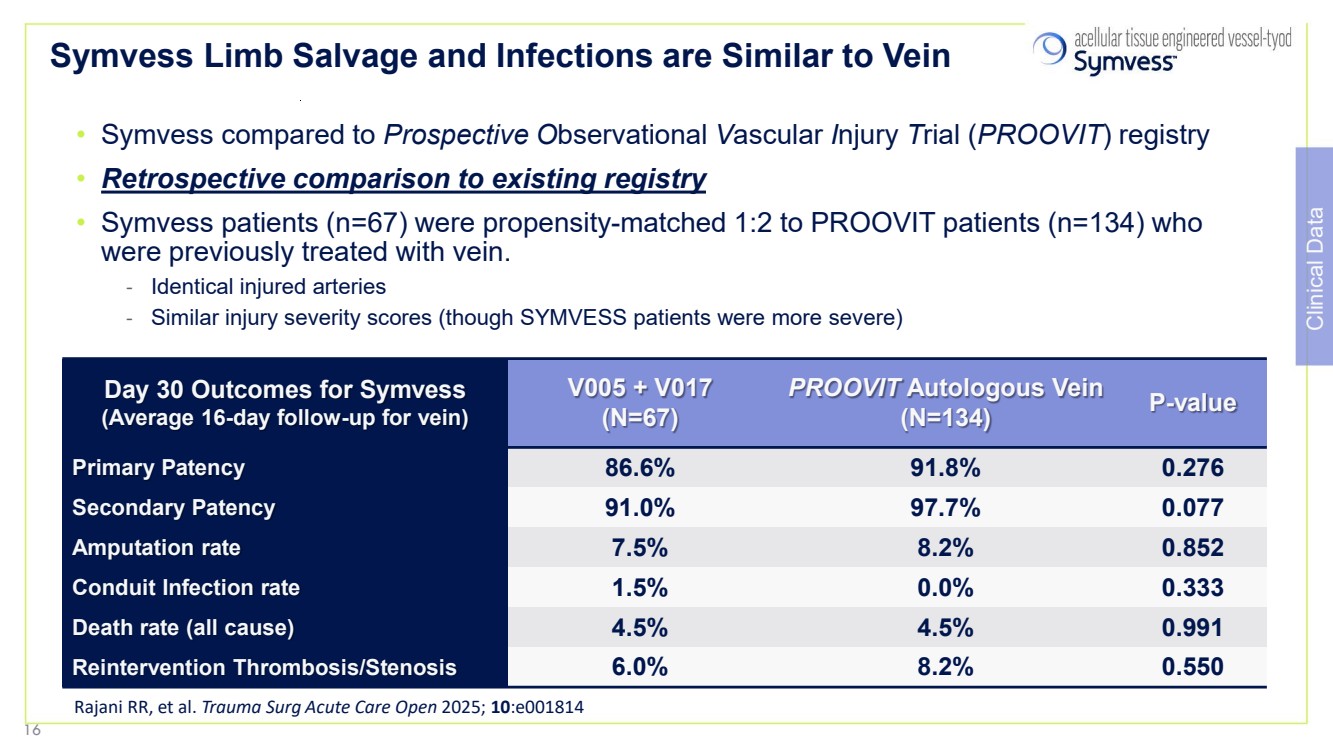
| Symvess Limb Salvage and Infections are Similar to Vein • Symvess compared to Prospective Observational Vascular Injury Trial (PROOVIT) registry • Retrospective comparison to existing registry • Symvess patients (n=67) were propensity-matched 1:2 to PROOVIT patients (n=134) who were previously treated with vein. - Identical injured arteries - Similar injury severity scores (though SYMVESS patients were more severe) Day 30 Outcomes for Symvess (Average 16-day follow-up for vein) V005 + V017 (N=67) PROOVIT Autologous Vein (N=134) P-value Primary Patency 86.6% 91.8% 0.276 Secondary Patency 91.0% 97.7% 0.077 Amputation rate 7.5% 8.2% 0.852 Conduit Infection rate 1.5% 0.0% 0.333 Death rate (all cause) 4.5% 4.5% 0.991 Reintervention Thrombosis/Stenosis 6.0% 8.2% 0.550 Rajani RR, et al. Trauma Surg Acute Care Open 2025; 10:e001814 Clinical Data 16 |
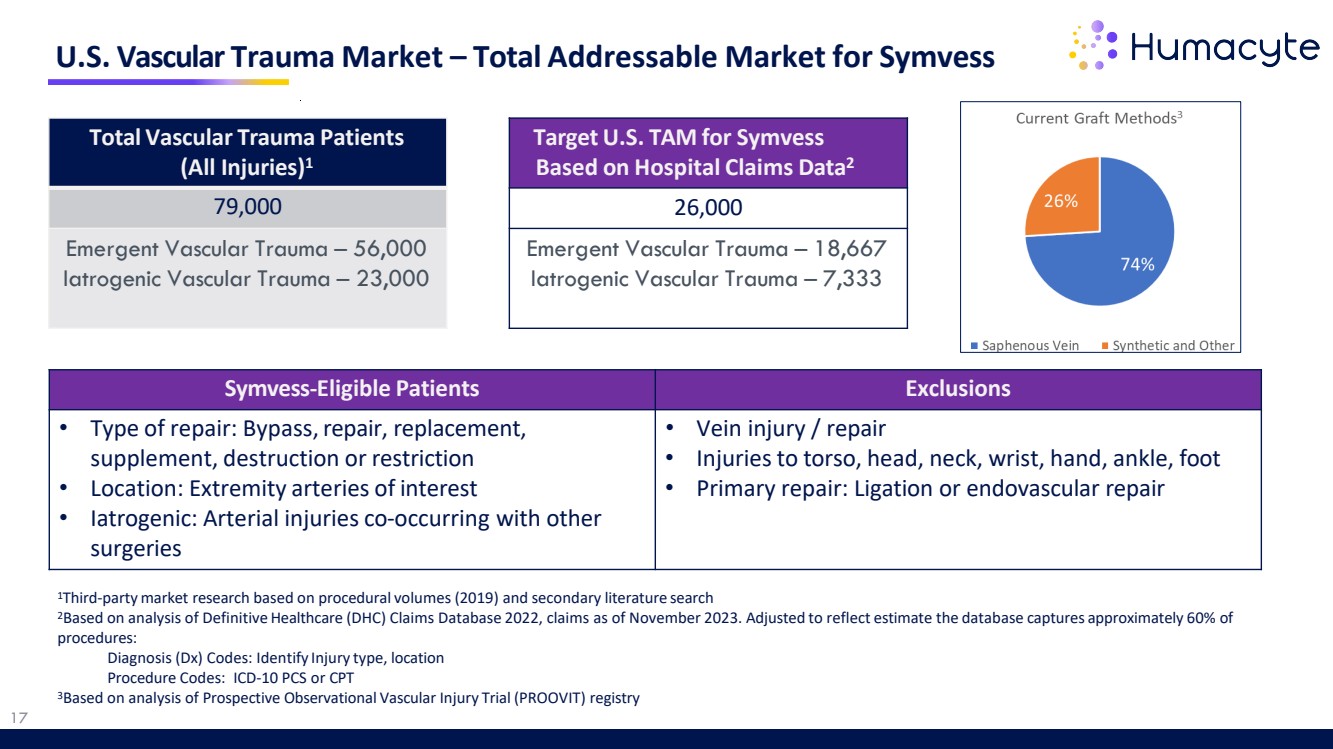
| U.S. Vascular Trauma Market – Total Addressable Market for Symvess Total Vascular Trauma Patients (All Injuries)1 79,000 Emergent Vascular Trauma – 56,000 Iatrogenic Vascular Trauma – 23,000 Target U.S. TAM for Symvess Based on Hospital Claims Data2 26,000 Emergent Vascular Trauma – 18,667 Iatrogenic Vascular Trauma – 7,333 1Third-partymarket research based on procedural volumes (2019) and secondary literature search 2Based on analysis of Definitive Healthcare (DHC) Claims Database 2022, claims as of November 2023. Adjusted to reflect estimate the database captures approximately 60% of procedures: Diagnosis (Dx) Codes: Identify Injury type, location Procedure Codes: ICD-10 PCS or CPT 3Based on analysis of Prospective Observational Vascular Injury Trial (PROOVIT) registry Symvess-Eligible Patients Exclusions • Type of repair: Bypass,repair, replacement, supplement, destruction or restriction • Location: Extremity arteries of interest • Iatrogenic: Arterial injuries co-occurring with other surgeries • Vein injury / repair • Injuries to torso, head, neck, wrist, hand, ankle, foot • Primary repair: Ligation or endovascular repair 17 |
| Drivers of U.S. Commercial Launch in Vascular Trauma 18 The Right Team Sales team of 12 executives who are experienced in vascular and/or trauma surgery and regenerative therapies Sales team is complemented by Medical Affairs, market access, and marketing teams Health Economics Budget Impact Model projects that the per-patient cost of treating patients with Symvess is estimated to be less than the cost of treating with synthetic grafts and other conduits Concentrated Market Approximately 200 Level 1 trauma centers in U.S. Approximately 3,000 vascular surgeons across civilian and military market opportunities Strong Clinical Results In the civilian and military clinical studies, Symvess was observed to have high rates of patency and low rates of amputation and infection Symvess acellulartissue engineered vessel-tyod |
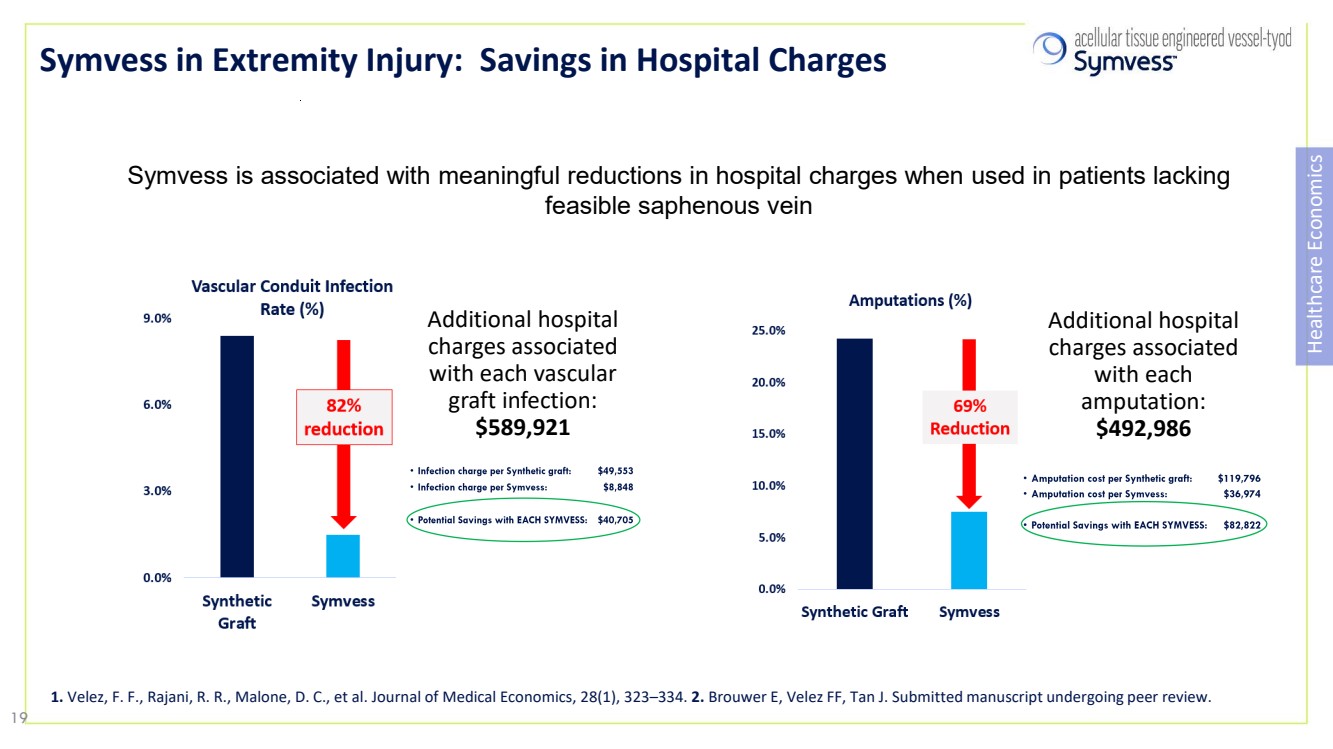
| Symvess in Extremity Injury: Savings in Hospital Charges Healthcare Economics Symvess is associated with meaningful reductions in hospital charges when used in patients lacking feasible saphenous vein Additional hospital charges associated with each vascular graft infection: $589,921 Additional hospital charges associated with each amputation: $492,986 1. Velez, F. F., Rajani, R. R., Malone, D. C., et al. Journal of Medical Economics, 28(1), 323–334. 2. Brouwer E, Velez FF, Tan J. Submitted manuscript undergoing peer review. 19 |
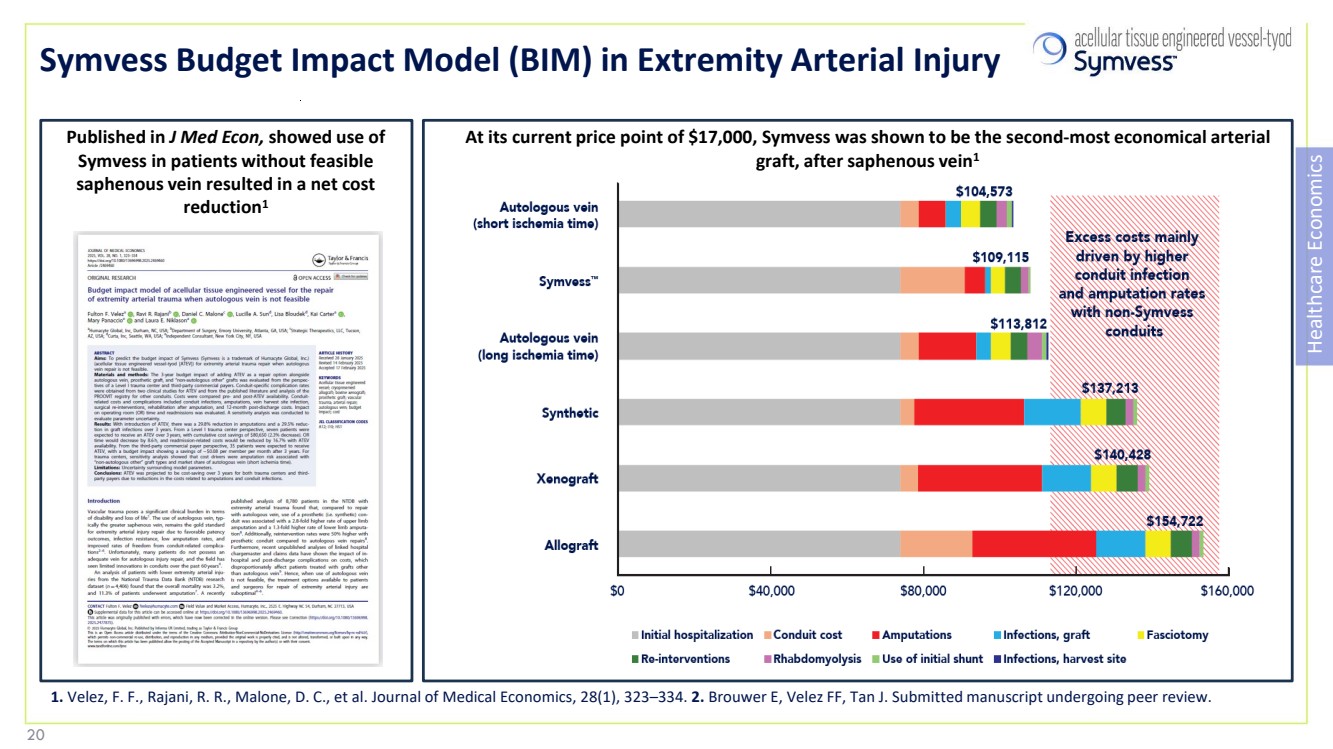
| Symvess Budget Impact Model (BIM) in Extremity Arterial Injury Healthcare Economics At its current price point of $17,000, Symvess was shown to be the second-most economical arterial graft, after saphenous vein1 Published in J Med Econ, showed use of Symvess in patients without feasible saphenous vein resulted in a net cost reduction1 1. Velez, F. F., Rajani, R. R., Malone, D. C., et al. Journal of Medical Economics, 28(1), 323–334. 2. Brouwer E, Velez FF, Tan J. Submitted manuscript undergoing peer review. 20 20 |
| Department of Defense Support DEPARTMENT OF DEFENSE SUPPORT Symvess (ATEV) for Vascular Trauma designated as a “Priority Product” by DoD • designation created by Public Law 115-92 to expedite the development and FDA review of DoD priority technologies V005 Phase 3 clinical trial was partially funded by the DoD Symvess successfully treated Ukrainian warfighters, resulting in 100% limb salvage FY 2026 DoD Appropriations Act includes funding for the evaluation and incorporation of biologic vascular repair technologies for warfighters The Department of Defense (DoD) invested in Symvess in recognition of its benefit in battlefield injuries for warfighters In civilian mass-casualty situations, having Symvess on the shelf can also help with response to terrorism/other threats, since surgeons can operate more quickly and treat more patients, not having to take time to harvest vein 21 |
| 22 AV Access for Dialysis |
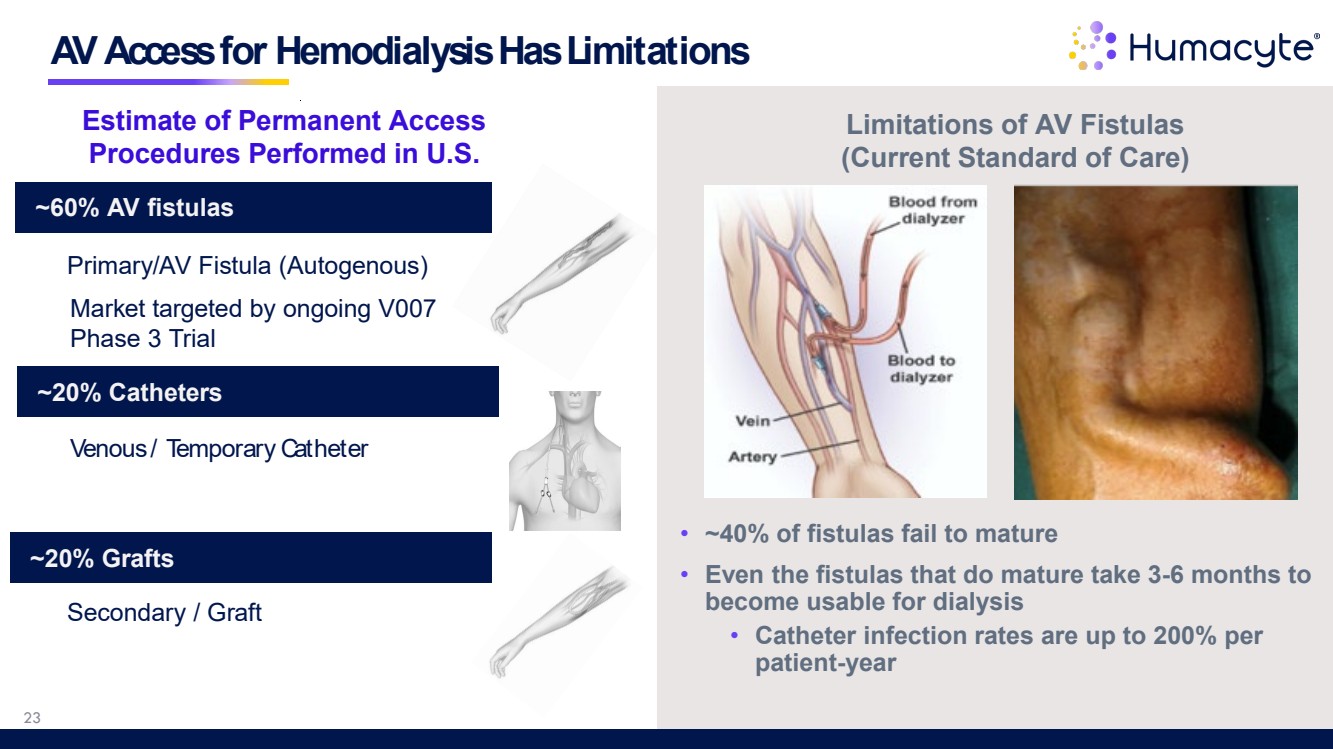
| AV Access for Hemodialysis Has Limitations Estimate of Permanent Access Procedures Performed in U.S. ~20% Grafts ~20% Catheters ~60% AV fistulas Venous / Temporary Catheter Primary/AV Fistula (Autogenous) Secondary / Graft Market targeted by ongoing V007 Phase 3 Trial • ~40% of fistulas fail to mature • Even the fistulas that do mature take 3-6 months to become usable for dialysis • Catheter infection rates are up to 200% per patient-year Limitations of AV Fistulas (Current Standard of Care) 23 |
| ATEV is Designed to Address Failures in AV Access • ATEV usable for dialysis after only four weeks • ATEV reduces catheter contact time, thereby reducing risk of catheter infection • >80% of ATEVs functional for dialysis at 6 months • ATEV infection rate is comparable to AVF • Opportunity to reduce cost of access failures and other complications: • Access failures and complications • Dialysis complications • Infections RMAT RMAT designation granted by FDA 24 ATEV provides potential for improved patient outcomes Strategic collaboration with FMC, the largest provider of renal care services |
| Partnered with Fresenius / Frenova Renal Research to identify the hemodialysis subpopulations with highest unmet needs Analysis of 178,575 adults with in-center hemodialysis established that: • Women are more likely to use AVG ± CVC for access within 90 days of initiation • Women have up to 90% increased risk of AVG ± CVC use, as compared to men • AVG ± CVC access has much higher complication rates: ~2X higher than AVF • Nearly $3 billion spent by Medicare in 2013 for on access complications/maintenance • Top quintile of dialysis patients cost between $91,841 to >$155,632 annually to maintain access • Women are more likely to fail AVF maturation: Cost >$30,000 in first year • Women are 20% more likely to fail AVF maturation • Women are 20% more likely to have multiple access failures in the first 6 months • Women are 24% more likely to have multiple hospitalizations for access complications • Some female sub-groups are at especially high risk • Example: Obese, diabetic women have excess costs of ~$27,000 to $91,000 during the first year Current AV Access in Women Work Poorly and is Expensive 25 |
| • More adverse events were reported in patients on the ATEV treatment arm than those on the AV fistula treatment arm: • More thromboses in the ATEV group, but virtually all were resolved • A number of serious events occurred more frequently in the AVF arm: • Two ruptures of AVF (a potentially fatal event), none for ATEV • Substantially more “steal” (ischemia of the hand), surgical revisions, and balloon-assisted maturation in the AVF group compared to the ATEV group V007 Top-Line Results – ATEV Met Co-Primary Endpoints 26 ATEV demonstrated superior function and patency at six and 12 months (co-primary endpoints) compared to autogenous fistula, the current standard of care for hemodialysis Co-Primary Endpoints ATEV (N=123) AVF (N=119) p-value Functional Patency at Month 6 81.3% 66.4% 0.0071 Secondary Patency at Month 12 68.3% 62.2% • Subjects with end-stage renal disease in need ATEV of dialysis • Enrollment completed April 2023, 242 total subjects Single-stage AV Fistula |
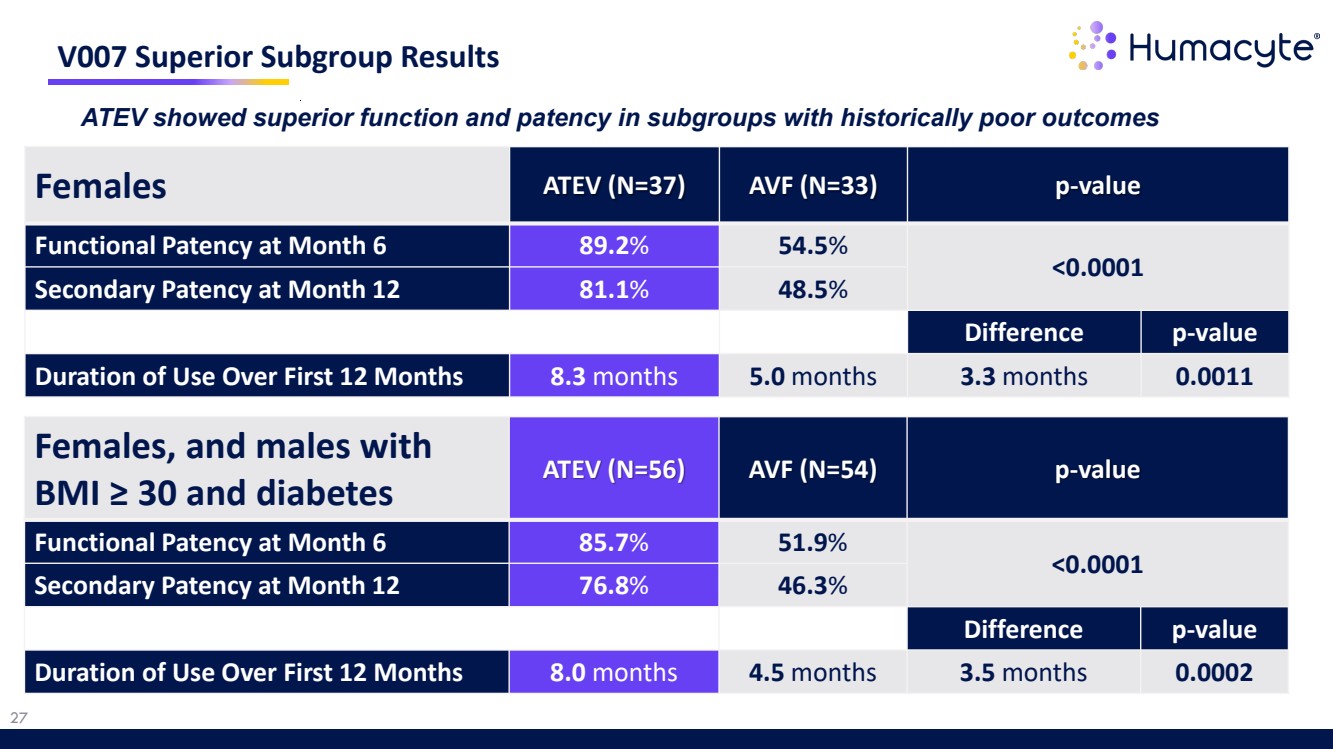
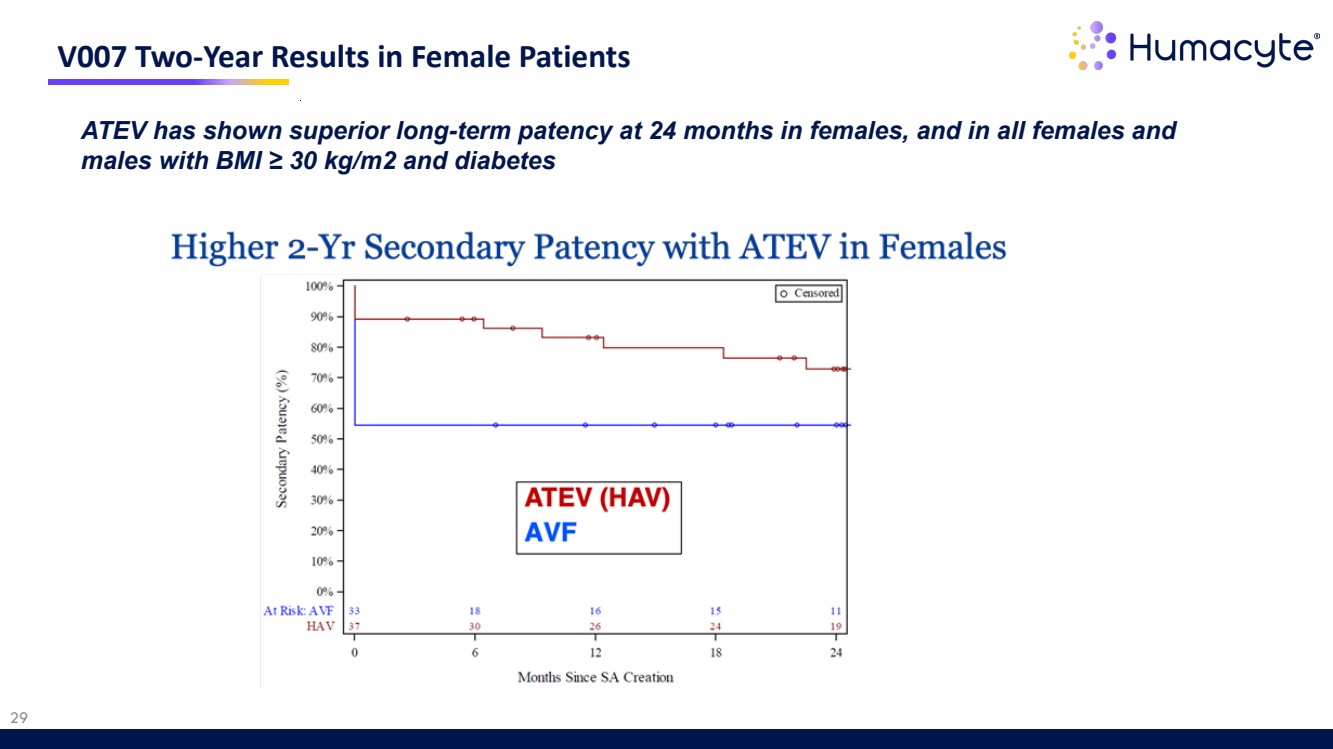
| 27 V007 Superior Subgroup Results Females ATEV (N=37) AVF (N=33) p-value Functional Patency at Month 6 89.2% 54.5% <0.0001 Secondary Patency at Month 12 81.1% 48.5% Difference p-value Duration of Use Over First 12 Months 8.3 months 5.0 months 3.3 months 0.0011 ATEV showed superior function and patency in subgroups with historically poor outcomes Females, and males with BMI ≥ 30 and diabetes ATEV (N=56) AVF (N=54) p-value Functional Patency at Month 6 85.7% 51.9% <0.0001 Secondary Patency at Month 12 76.8% 46.3% Difference p-value Duration of Use Over First 12 Months 8.0 months 4.5 months 3.5 months 0.0002 |
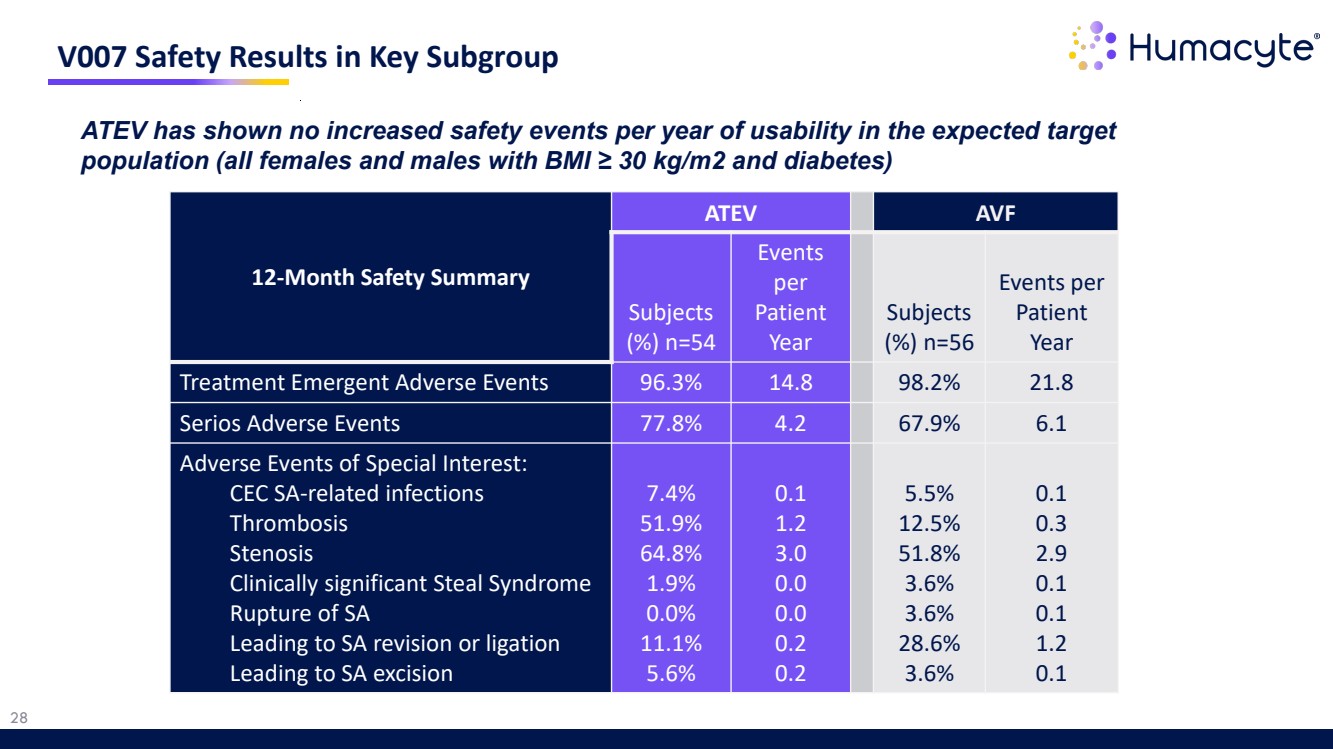
| 28 V007 Safety Results in Key Subgroup ATEV has shown no increased safety events per year of usability in the expected target population (all females and males with BMI ≥ 30 kg/m2 and diabetes) 12-Month Safety Summary ATEV AVF Subjects (%) n=54 Events per Patient Year Subjects (%) n=56 Events per Patient Year Treatment Emergent Adverse Events 96.3% 14.8 98.2% 21.8 Serios Adverse Events 77.8% 4.2 67.9% 6.1 Adverse Events of Special Interest: CEC SA-related infections Thrombosis Stenosis Clinically significant Steal Syndrome Rupture of SA Leading to SA revision or ligation Leading to SA excision 7.4% 51.9% 64.8% 1.9% 0.0% 11.1% 5.6% 0.1 1.2 3.0 0.0 0.0 0.2 0.2 5.5% 12.5% 51.8% 3.6% 3.6% 28.6% 3.6% 0.1 0.3 2.9 0.1 0.1 1.2 0.1 |
| 29 V007 Two-Year Results in Female Patients ATEV has shown superior long-term patency at 24 months in females, and in all females and males with BMI ≥ 30 kg/m2 and diabetes |
| • Enrollment: • Target 150 total subjects (interim analysis after 80 patients) • 1:1 Prospective randomization ATEV vs. Autogenous fistula V012 (HUMAXX) Trial in Women Dialysis Patients To Compare the Efficacy and Safety of the ATEV With AVF in Female Patients With End-Stage Renal Disease Requiring Hemodialysis (HUMAXX) Female patients currently receiving hemodialysis via catheter and who are candidates for creation of an AVF or implantation of an ATEV. Objectives: • Primary Efficacy: Total days free from in-dwelling catheter (“catheter-free days”) until 365 days, or until access abandonment, whichever occurs first. • Primary Safety: Number and severity of infections related to all accesses (including catheters) from access creation until 365 days. Trial comparing Humacyte’s (ATEV ) to AVF in women 30 Expect to file supplemental BLA based after interim analysis of V012 study results: Planned supplemental BLA filling in 2nd half of 2026 Target subgroups in which the ATEV showed the best results in the V007 study: All females, and potentially males with one or more risk factors |
| 31 Peripheral Arterial Disease (PAD) |
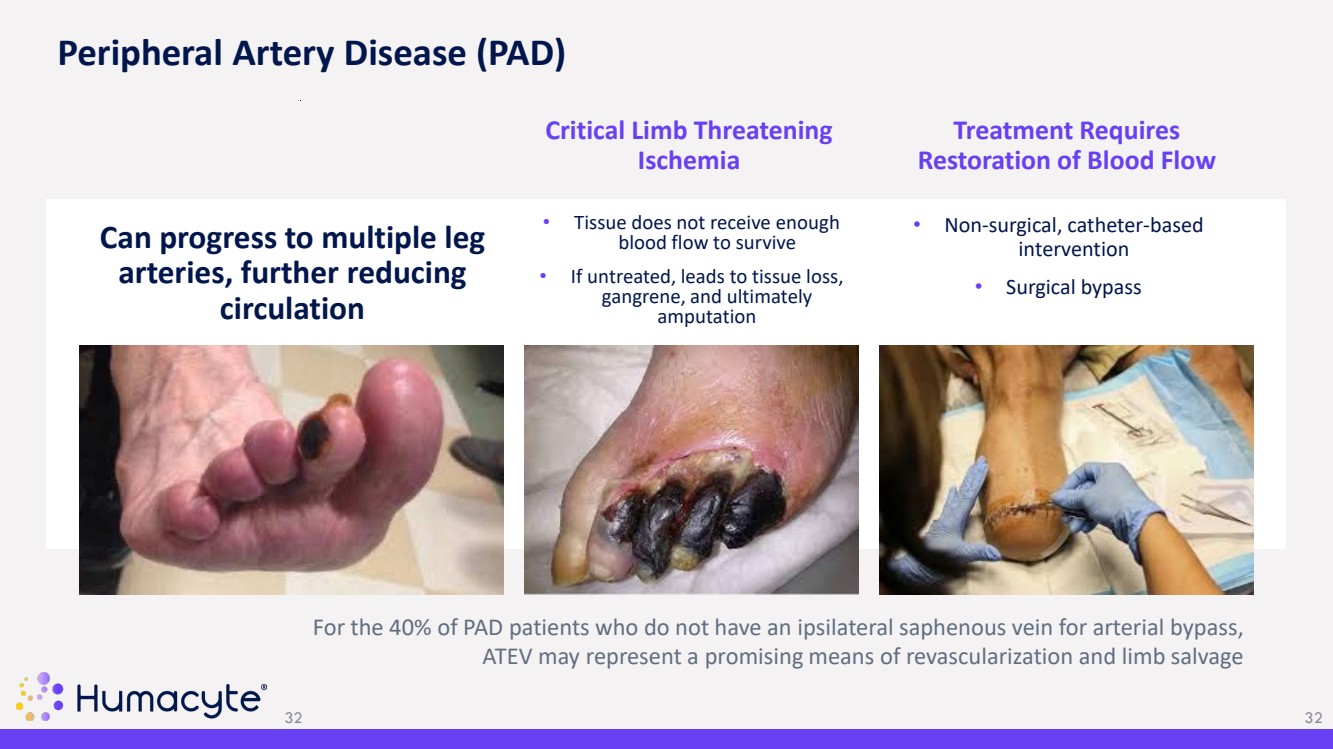
| • Tissue does not receive enough blood flow to survive • If untreated, leads to tissue loss, gangrene, and ultimately amputation Critical Limb Threatening Ischemia • Non-surgical, catheter-based intervention • Surgical bypass Treatment Requires Restoration of Blood Flow 32 Can progress to multiple leg arteries, further reducing circulation For the 40% of PAD patients who do not have an ipsilateral saphenous vein for arterial bypass, ATEV may represent a promising means of revascularization and limb salvage Peripheral Artery Disease (PAD) 32 |
| Current Clinical Experience with ATEV in Peripheral Arterial Disease 1Piotr Gutowski, et al, 6-Year Outcomes of a Phase 2 Study of Human-Tissue Engineered Blood Vessels for Peripheral Arterial Bypass, JVS: Vascular Science (2023) 2Lauria A, Kersey A, Propper B, et al. Annals of Vascular Surgery. 2022 Apr 6:S0890-5096(22)00180-7 • V002 – 20 patients (EU) • V004 – 15 patients (US) Phase 2 Trials Over 20 U.S. patients with critical limb ischemia treated under FDA Expanded Access program Investigator-sponsored IND • 29 patients with severe PAD at risk of limb loss • Patients did no have saphenous vein available EA Mayo IND • Six-year results from V002 published in Journal of Vascular Surgery – Vascular Science1 • Publication of First Eight Expanded Access Cases in Annals of Vascular Surgery2 • Outcomes published in Midwestern Vascular Surgical Society showing 86% limb salvage rate 33 |
| Expanded Access: Restoring Mobility with ATEV • The ATEV was used under compassionate use program in 70-year-old patient with severe vascular disease • No vein was available to perform a bypass, as the vein was previously harvested for a CABG • A right distal superficial femoral artery-to-peroneal artery bypass was performed using an ATEV • The patient’s postoperative course was unremarkable • At 1-year follow-up the angiography showed a patent ATEV without significant stenosis at the distal anastomosis • Four years after ATEV implantation, the patient continues to do well and is walking. This case was included in ATEV results in critical limb ischemia presented at VESS meeting in January 2022 Knee joint Staples at incision ATEV Angiogram at 1 year Bypass performed using the ATEV in patient with severe vascular disease 34 |
| 35 Pipeline: CTEV for Coronary Bypass Graft Surgery |
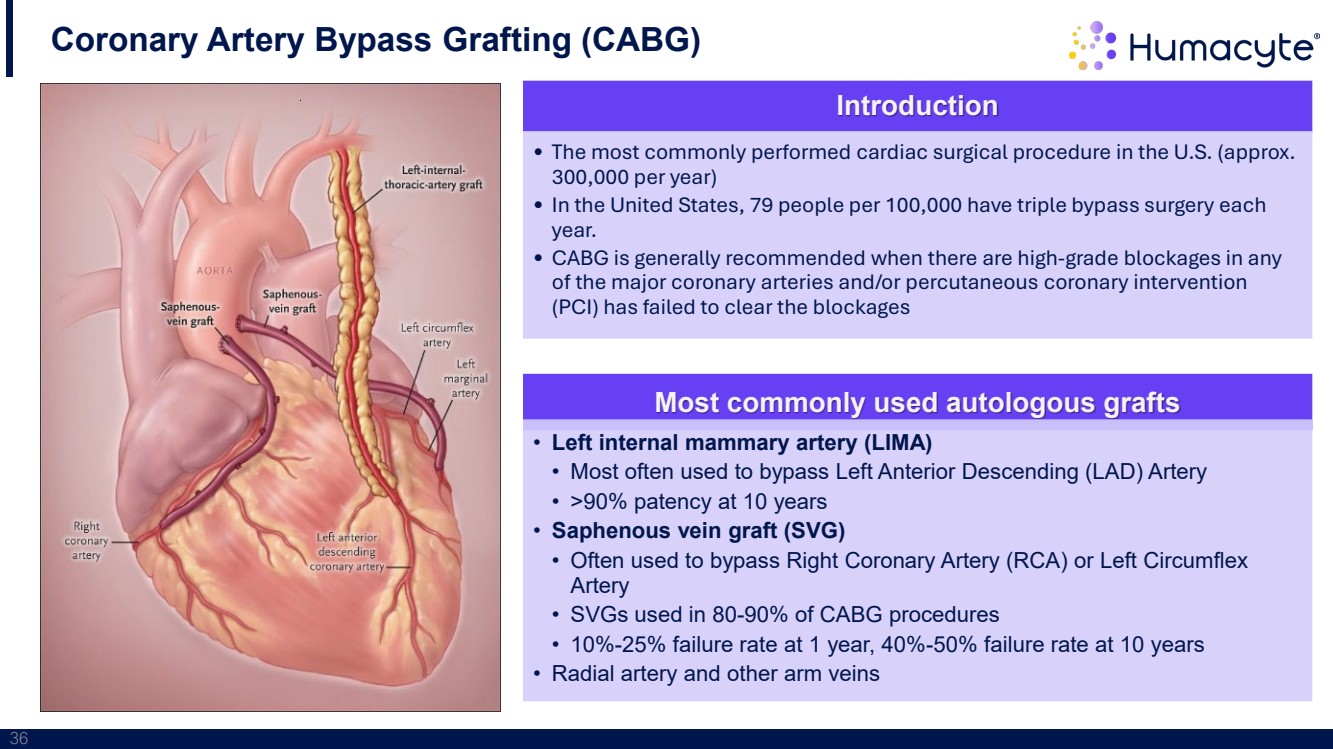
| 36 Coronary Artery Bypass Grafting (CABG) Introduction • The most commonly performed cardiac surgical procedure in the U.S. (approx. 300,000 per year) • In the United States, 79 people per 100,000 have triple bypass surgery each year. • CABG is generally recommended when there are high-grade blockages in any of the major coronary arteries and/or percutaneous coronary intervention (PCI) has failed to clear the blockages Most commonly used autologous grafts • Left internal mammary artery (LIMA) • Most often used to bypass Left Anterior Descending (LAD) Artery • >90% patency at 10 years • Saphenous vein graft (SVG) • Often used to bypass Right Coronary Artery (RCA) or Left Circumflex Artery • SVGs used in 80-90% of CABG procedures • 10%-25% failure rate at 1 year, 40%-50% failure rate at 10 years • Radial artery and other arm veins |
| Concerns with Saphenous Vein Grafts Vein Quality Issues • Varicosities (20-30% of patients) • Previous vein stripping or ablation • Small diameter (<3mm) • Sclerotic or diseased veins • Peripheral vascular disease effects Medical Co-morbidities • Bilateral leg amputations • Need to preserve vein for future peripheral bypass • Prior vein harvest for CABG or peripheral surgery • Obesity (difficult harvest) • Patients with diabetes (higher failure rates) Harvest-Related Morbidity • Wound complications (5-10%) • Leg edema and pain • Infection risk • Nerve injury (saphenous nerve) • Prolonged recovery time Long-Term Clinical Impact • Need for repeat revascularization • Recurrent angina (20-30% at 5 years) • Risk of graft atherosclerosis • Reduced event-free survival • Higher healthcare costs • Zenati MA et al. N Engl J Med. 2019;380:2069-77 • Hess CN et al. Circulation. 2014;130:815-27 • Lopes RD et al. JAMA. 2012;307:265-74 |
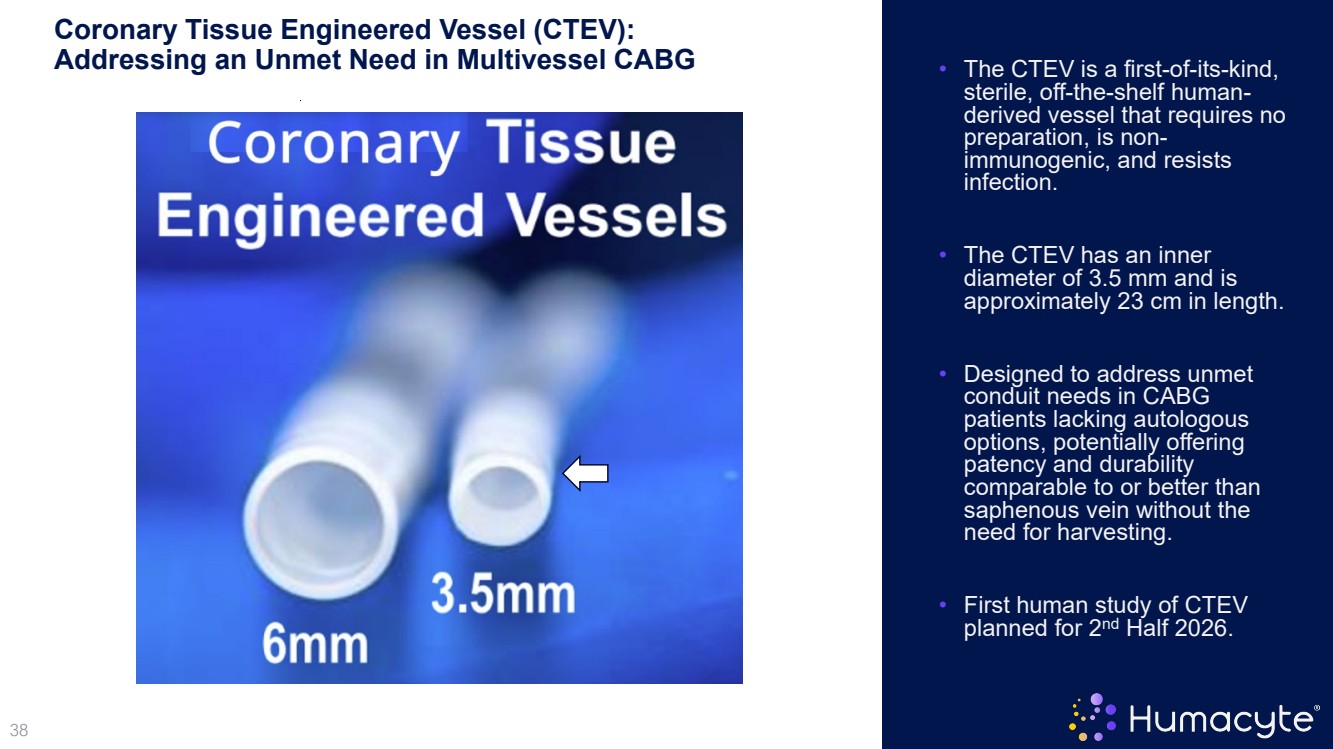
| 38 Coronary Tissue Engineered Vessel (CTEV): Addressing an Unmet Need in Multivessel CABG • The CTEV is a first-of-its-kind, sterile, off-the-shelf human- derived vessel that requires no preparation, is non- immunogenic, and resists infection. • The CTEV has an inner diameter of 3.5 mm and is approximately 23 cm in length. • Designed to address unmet conduit needs in CABG patients lacking autologous options, potentially offering patency and durability comparable to or better than saphenous vein without the need for harvesting. • First human study of CTEV planned for 2nd Half 2026. |
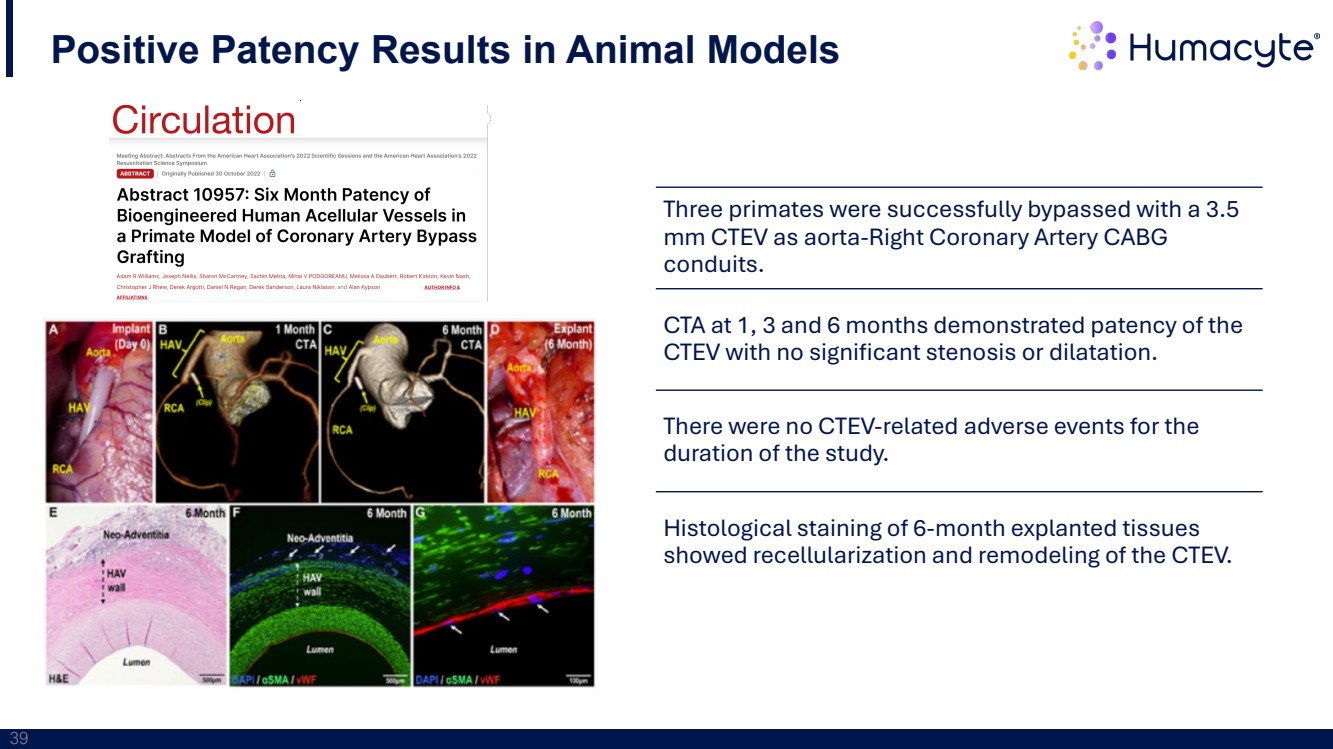
| 39 Three primates were successfully bypassed with a 3.5 mm CTEV as aorta-Right Coronary Artery CABG conduits. CTA at 1, 3 and 6 months demonstrated patency of the CTEV with no significant stenosis or dilatation. There were no CTEV-related adverse events for the duration of the study. Histological staining of 6-month explanted tissues showed recellularization and remodeling of the CTEV. Positive Patency Results in Animal Models |
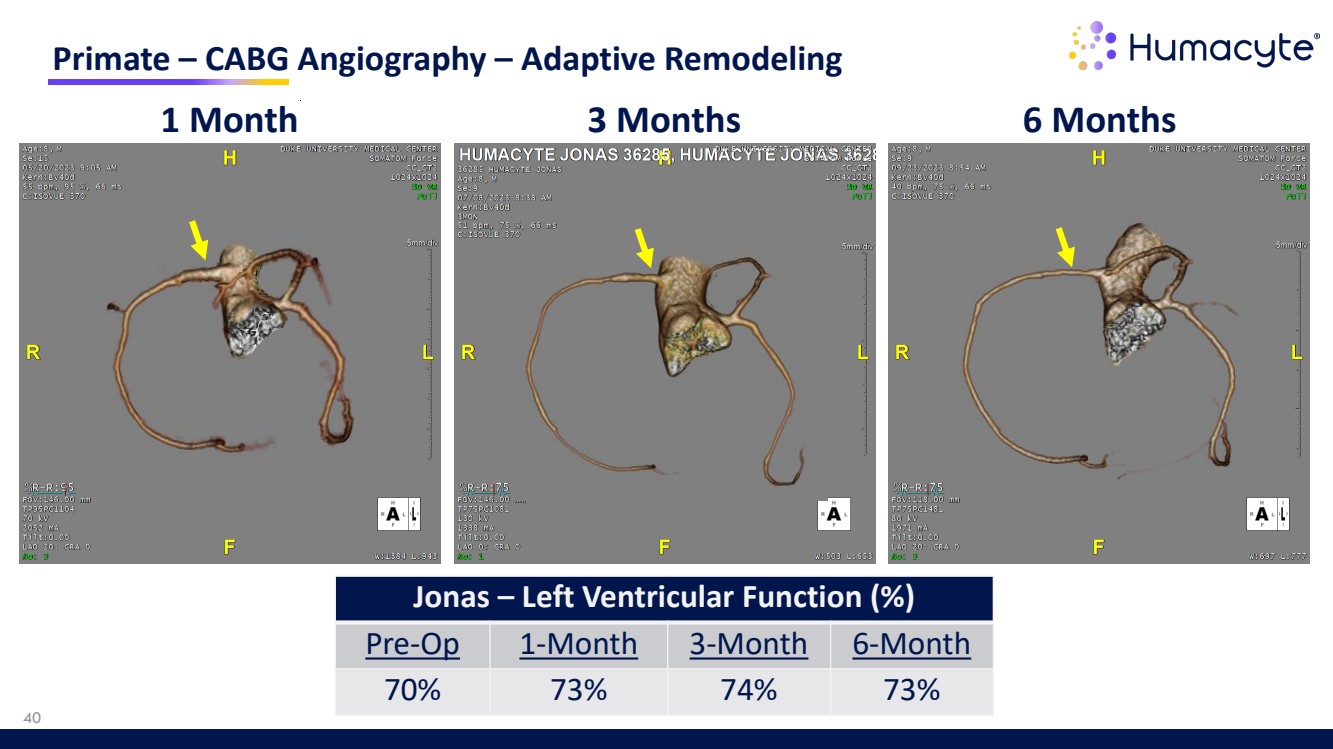
| Primate – CABG Angiography – Adaptive Remodeling 1 Month 3 Months 6 Months Jonas – Left Ventricular Function (%) Pre-Op 1-Month 3-Month 6-Month 70% 73% 74% 73% 40 |
| 41 Pipeline: BioVascular Pancreas |
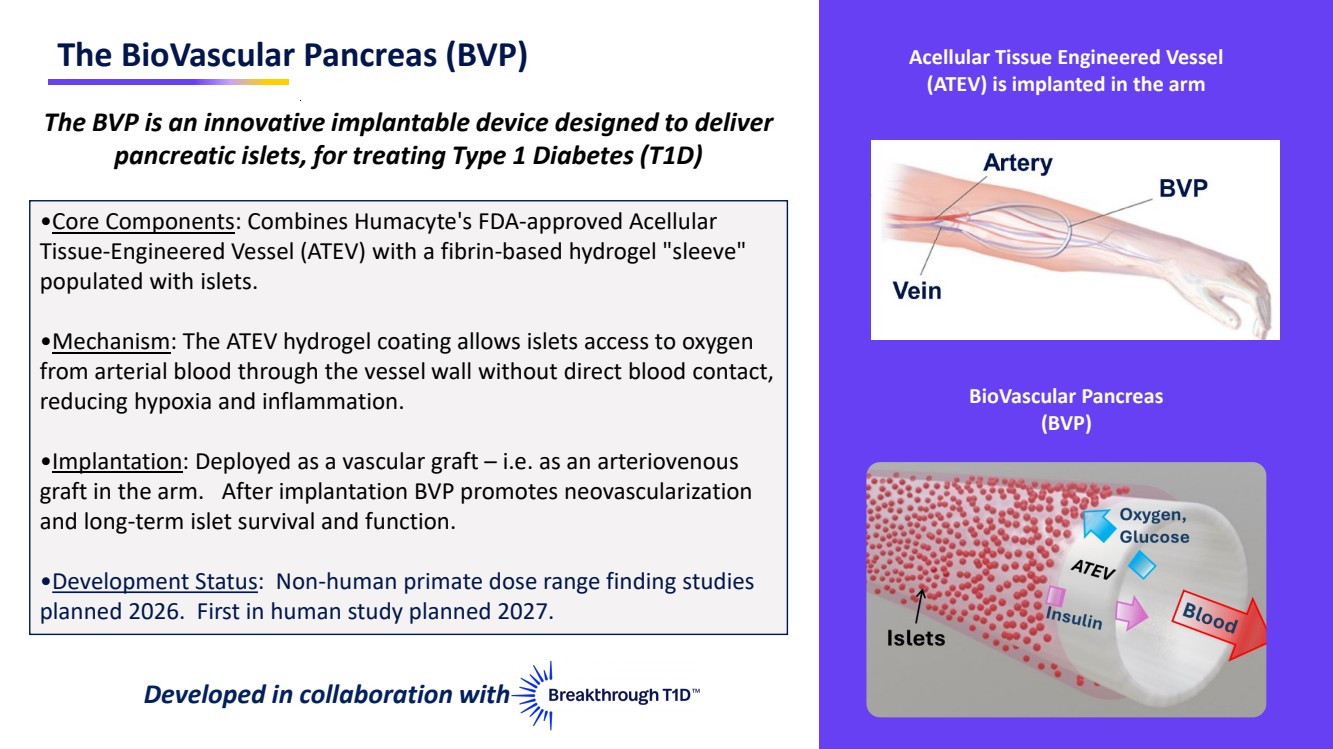
| The BioVascular Pancreas (BVP) Acellular Tissue Engineered Vessel (ATEV) is implanted in the arm BioVascular Pancreas (BVP) The BVP is an innovative implantable device designed to deliver pancreatic islets, for treating Type 1 Diabetes (T1D) •Core Components: Combines Humacyte's FDA-approved Acellular Tissue-Engineered Vessel (ATEV) with a fibrin-based hydrogel "sleeve" populated with islets. •Mechanism: The ATEV hydrogel coating allows islets access to oxygen from arterial blood through the vessel wall without direct blood contact, reducing hypoxia and inflammation. •Implantation: Deployed as a vascular graft – i.e. as an arteriovenous graft in the arm. After implantation BVP promotes neovascularization and long-term islet survival and function. •Development Status: Non-human primate dose range finding studies planned 2026. First in human study planned 2027. Developed in collaboration with |
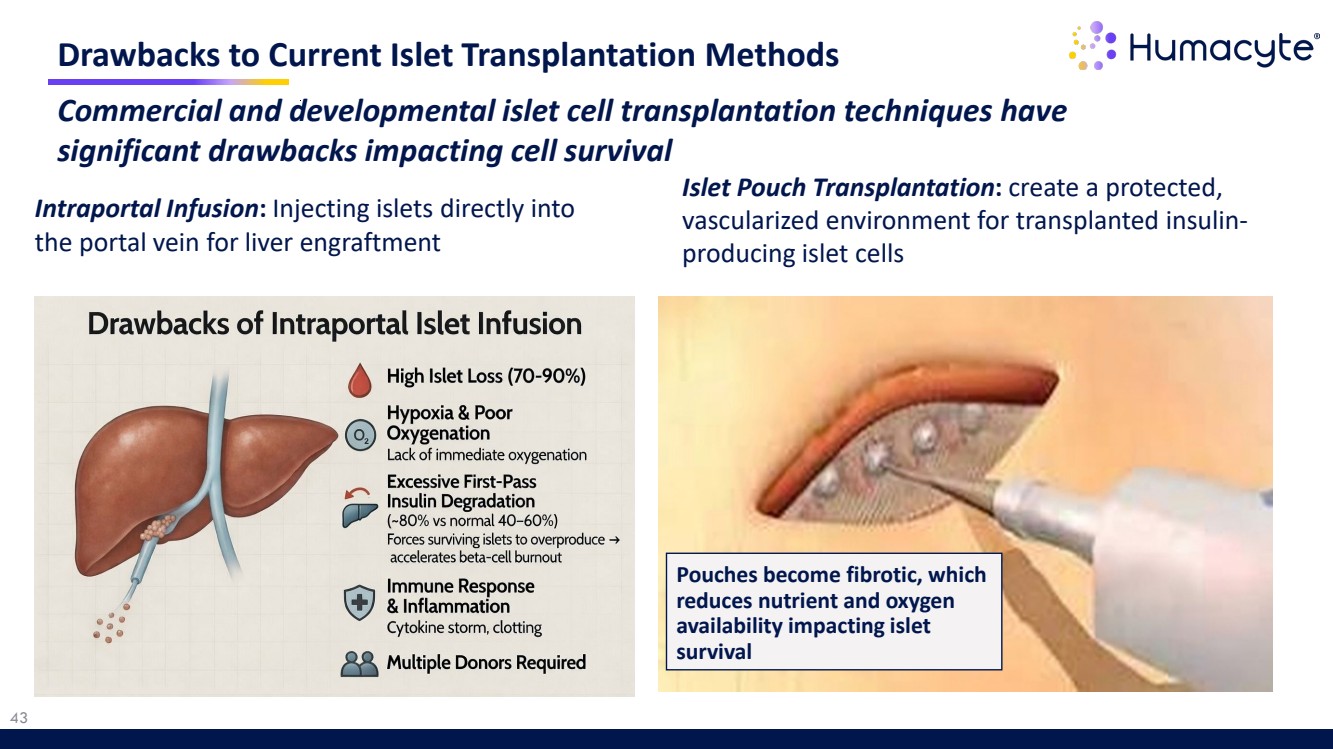
| 43 Drawbacks to Current Islet Transplantation Methods Intraportal Infusion: Injecting islets directly into the portal vein for liver engraftment ECTRA Commercial and developmental islet cell transplantation techniques have significant drawbacks impacting cell survival Islet Pouch Transplantation: create a protected, vascularized environment for transplanted insulin-producing islet cellsTRA Pouches become fibrotic, which reduces nutrient and oxygen availability impacting islet survival |
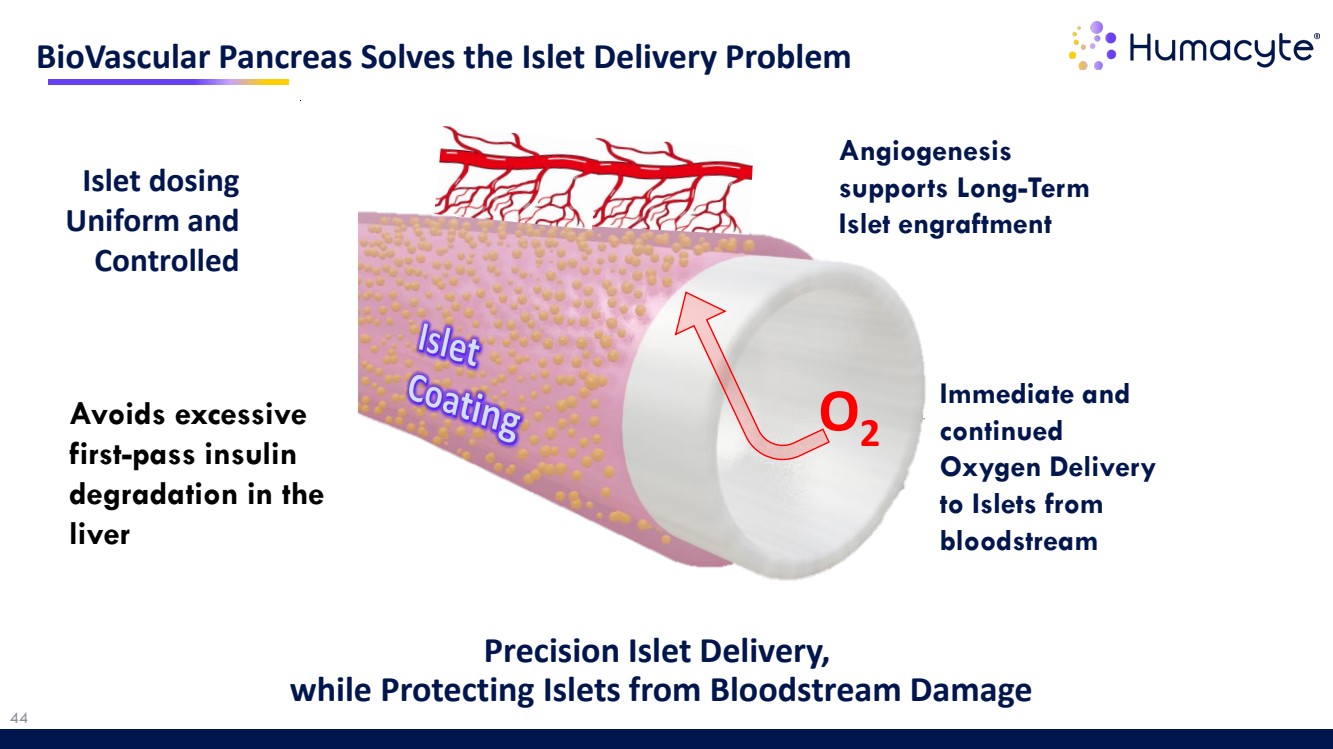
| 44 BioVascular Pancreas Solves the Islet Delivery Problem Precision Islet Delivery, while Protecting Islets from Bloodstream Damage Islet dosing Uniform and Controlled Immediate and continued Oxygen Delivery to Islets from bloodstream Angiogenesis supports Long-Term Islet engraftment O2 Avoids excessive first-pass insulin degradation in the liver |
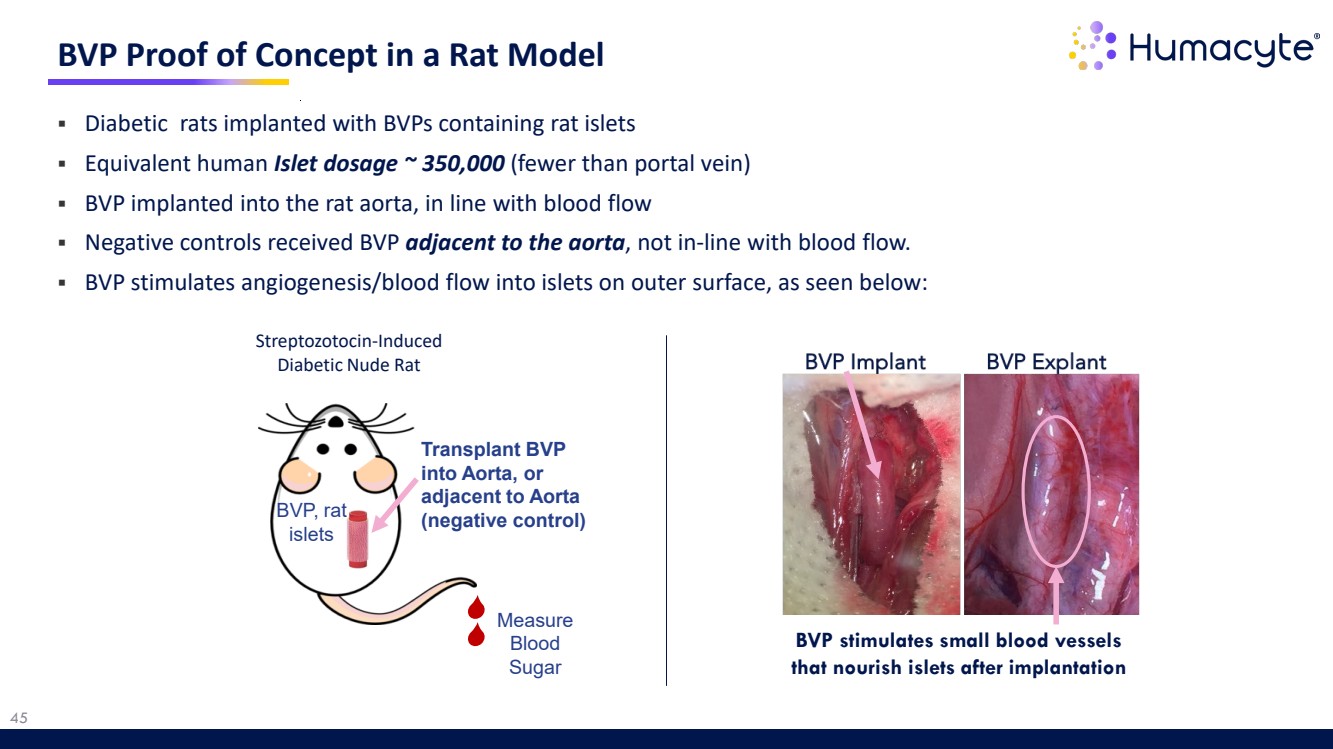
| 45 BVP Proof of Concept in a Rat Model BVP stimulates small blood vessels that nourish islets after implantation Measure Blood Sugar Streptozotocin-Induced Diabetic Nude Rat BVP, rat islets Transplant BVP into Aorta, or adjacent to Aorta (negative control) Diabetic rats implanted with BVPs containing rat islets Equivalent human Islet dosage ~ 350,000 (fewer than portal vein) BVP implanted into the rat aorta, in line with blood flow Negative controls received BVP adjacent to the aorta, not in-line with blood flow. BVP stimulates angiogenesis/blood flow into islets on outer surface, as seen below: |
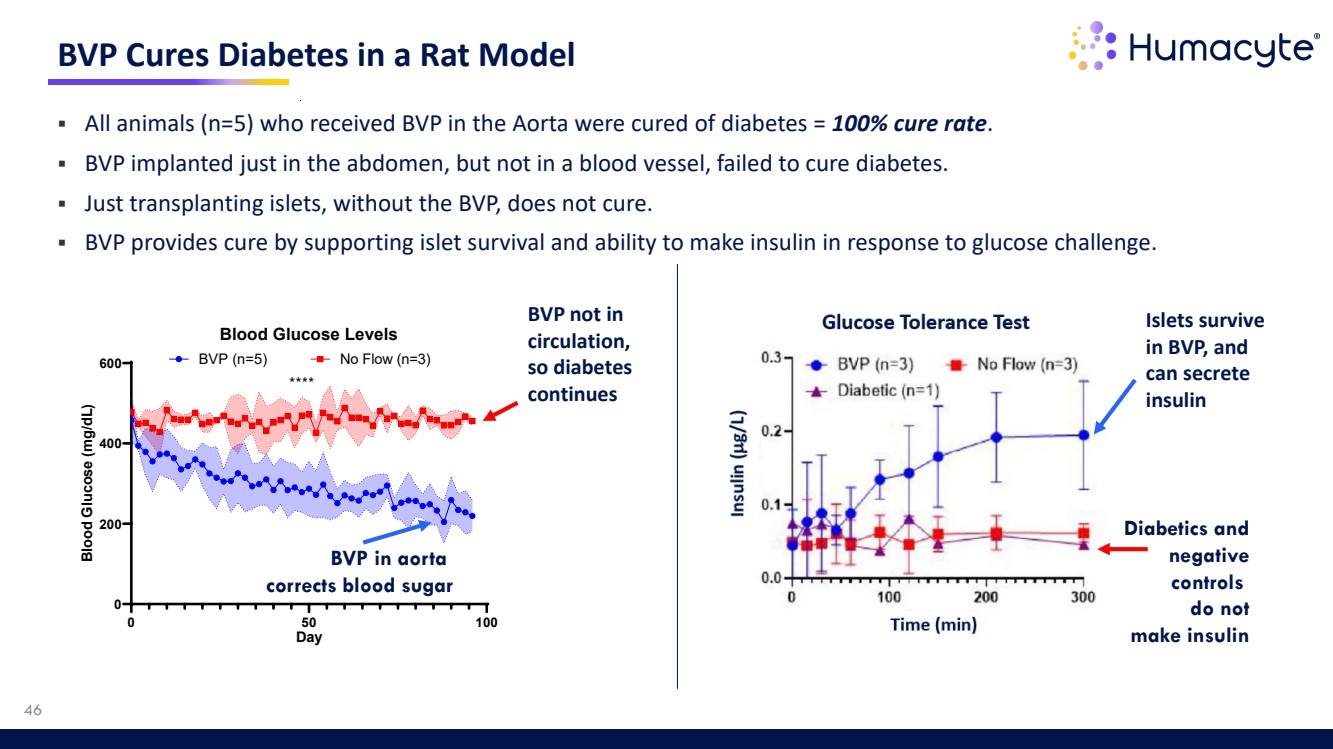
| 46 BVP Cures Diabetes in a Rat Model 0 50 100 0 200 400 600 Blood Glucose Levels Day Blood Glucose (mg/dL) BVP (n=5) No Flow (n=3) **** BVP not in circulation, so diabetes continues BVP in aorta corrects blood sugar All animals (n=5) who received BVP in the Aorta were cured of diabetes = 100% cure rate. BVP implanted just in the abdomen, but not in a blood vessel, failed to cure diabetes. Just transplanting islets, without the BVP, does not cure. BVP provides cure by supporting islet survival and ability to make insulin in response to glucose challenge. Diabetics and negative controls do not make insulin Islets survive in BVP, and can secrete insulin |
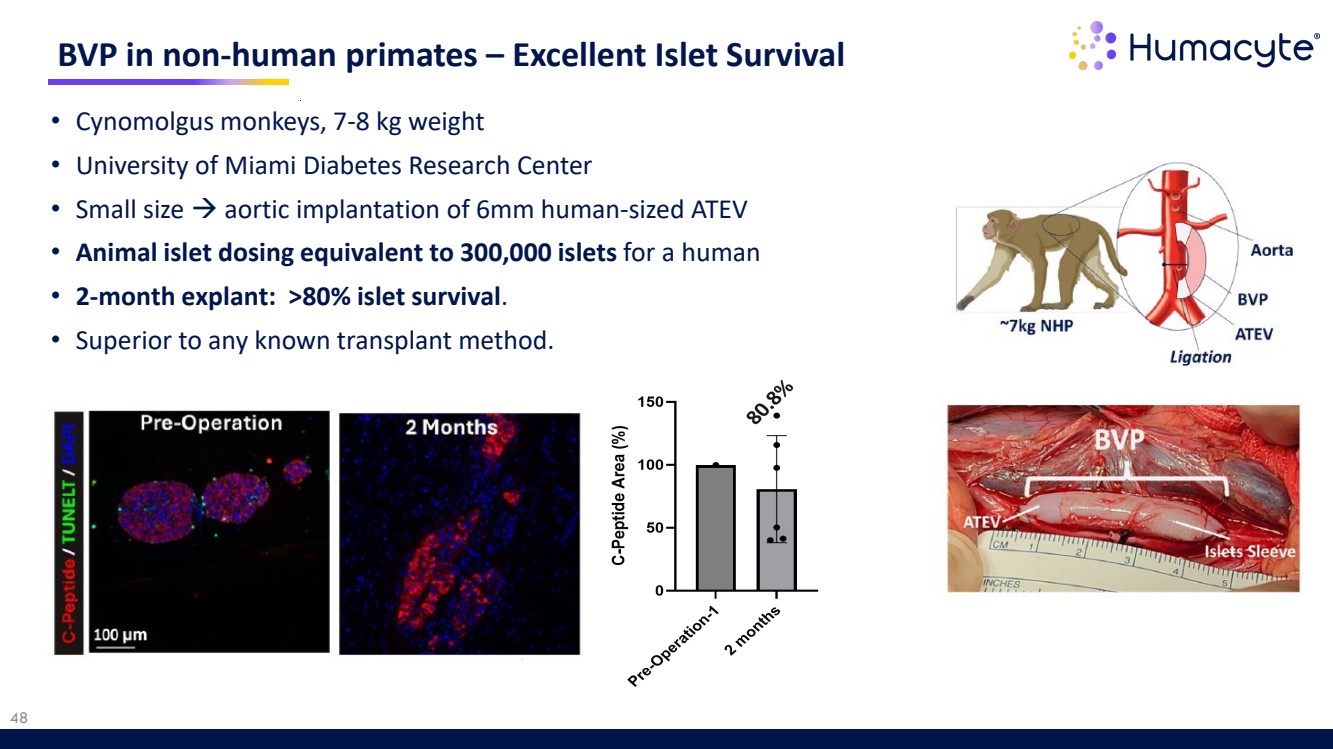
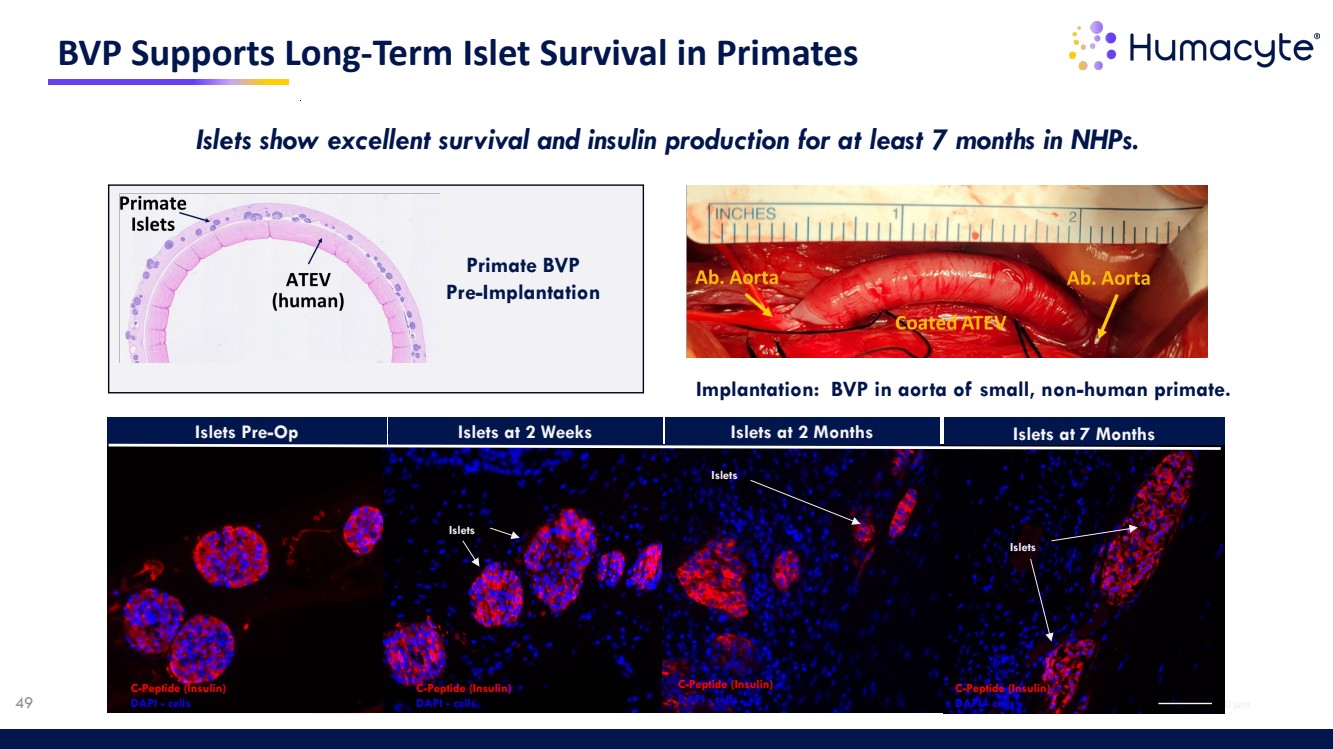
| 47 BVP NHP Preclinical Data • BVP Non-Human Primate (NHP) model: human 6mm ATEV combined with primate islets • Subjects: Cynomolgus macaques weighing 7-8 kg with streptozotocin-induced T1D • Treatment with allogeneic Biovascular Pancreas (BVP) delivering ~3840 islet equivalents/KG supported islet survival for up to 7 months post-implantation • Histology showed healthy and C-peptide-positive islets with few apoptotic cells • Greater than 80% islet retention observed at 2 months. • Significant neovascularization near islets by 7 months • Functional Outcomes Correlated with detectable plasma C-peptide levels starting at 4 months with reduced needs for exogenous insulin Greater than 80% |
| 48 • Cynomolgus monkeys, 7-8 kg weight • University of Miami Diabetes Research Center • Small size aortic implantation of 6mm human-sized ATEV • Animal islet dosing equivalent to 300,000 islets for a human • 2-month explant: >80% islet survival. • Superior to any known transplant method. BVP in non-human primates – Excellent Islet Survival |
| Islets 100 µm 100 µm 100 µm 100 µm BVP Supports Long-Term Islet Survival in Primates Islets Islets Pre-Op Islets at 2 Weeks Islets at 2 Months Islets Islets at 7 Months Islets C-Peptide (Insulin) DAPI - cells C-Peptide (Insulin) DAPI - cells C-Peptide (Insulin) DAPI - cells C-Peptide (Insulin) 49 DAPI - cells Implantation: BVP in aorta of small, non-human primate. I Primate BVP Pre-Implantation Islets show excellent survival and insulin production for at least 7 months in NHPs. |
| 50 Milestones |
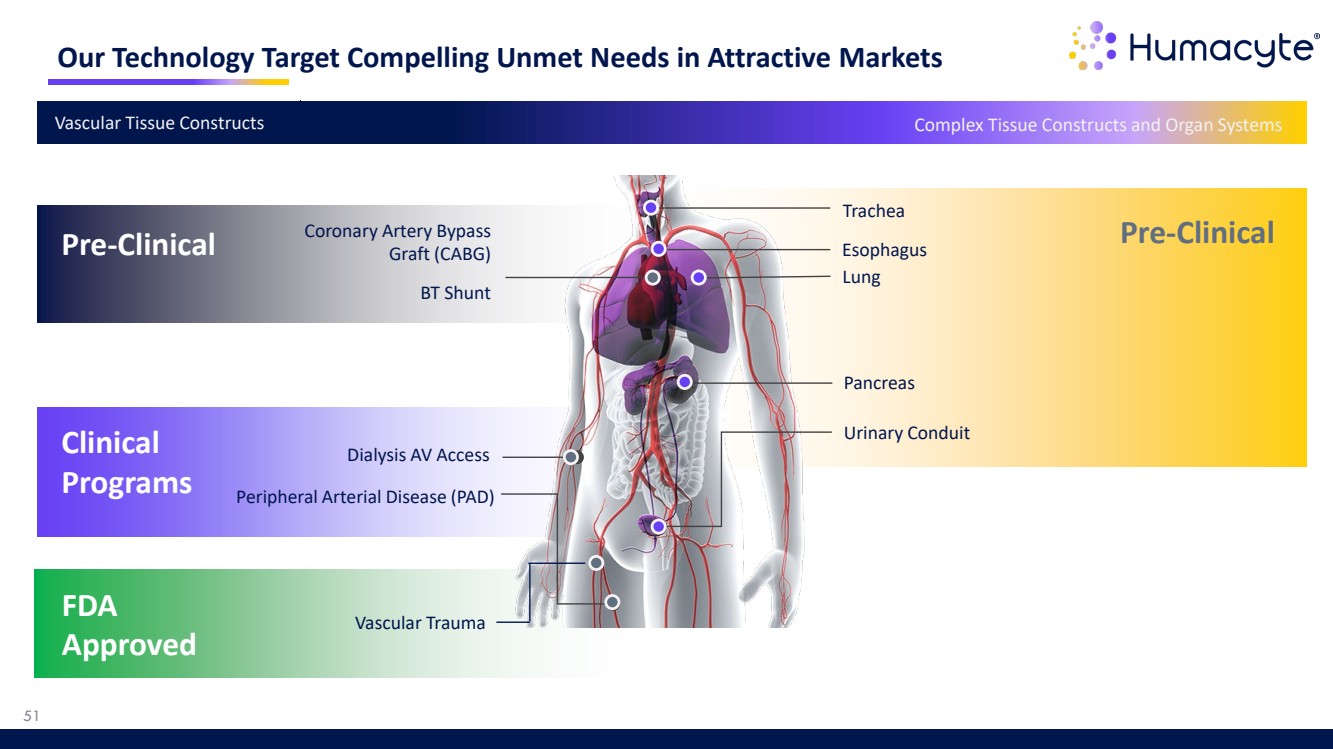
| Our Technology Target Compelling Unmet Needs in Attractive Markets Pre-Clinical Trachea Pancreas Esophagus Urinary Conduit Lung Peripheral Arterial Disease (PAD) Dialysis AV Access Coronary Artery Bypass Graft (CABG) BT Shunt Clinical Programs Pre-Clinical Vascular Tissue Constructs Complex Tissue Constructs and Organ Systems 51 FDA Approved Vascular Trauma |
| Commercial Manufacturing Scale – LUNA200 System Commercial 83,000 sq ft Bioprocessing Facility • Currently operating 8 LUNA200 systems • Annual capacity expected to exceed 40,000 ATEVs • Functionally closed system with state-of-the-art process automation Bioreactor bag Each bioreactor bag contains a single polymer mesh scaffold, seeded with banked human cells 10 bioreactor bags per growth drawer; tubing connects to shared nutritive media Each LUNA200 can produce 200 ATEVs per batch (or ~1,000 ATEVs annually) Growth drawer LUNA200 System 52 |
| Anticipated 2026 Milestones Vascular Trauma (Symvess): • U.S. commercial launch growth • Expansion into international markets Dialysis (ATEV): • Publication of V007 Phase 3 results • Interim results from V012 Phase 3 trial in female patients • Supplemental BLA filing with FDA All milestone dates are only management estimates Vascular Trauma - Symvess: • U.S. commercial launch • Long-term results showing durability of Symvess V007 dialysis positive Phase 3 ATEV two-year results Cardiac Bypass Graft Surgery (CABG) CTEV preclinical results from large-animal studies Preclinical BVP results showing survival and function of islets in large animals Completed in 2025 Planned for 2026 CABG (CTEV): • Commencement of first-in-human study • First patient results BioVascular Pancreas (BVP) for type-1 diabetes: • Preparation for first human study Publications & Presentations (Multiple other clinical and preclinical publications and presentations expected for 2026) 53 |
| The Promise of Regenerative Medicine Broad platform of universally implantable off-the-shelf bioengineered human tissues and organs Bioengineering Platform Platform targets extensive markets across multiple indications Extensive Markets Existing facilities expected to support anticipated commercial launch with room for modular expansion Commercial Scale Manufacturing 54 |
| Universally Implantable Regenerative Human Tissue Thank You |