

.2 –– Transaction & Company Overview CONFIDENTIAL 1

–– Disclaimers The information in this Presentation has been prepared by Sensei Biotherapeutics, Inc. ( Sensei ) and Faeth Holdings Therapeutics, Inc. ( Faeth and, together with Sensei and each of Sensei's subsidiaries, the combined company ) and contains information pertaining to the business and operations of the combined company. The information contained in this Presentation: (a) is provided as at the date hereof, is subject to change without notice, and is based on publicly available data, internally developed data as well as third party information from other sources; (b) does not purpose to contain all the information that may be necessary or desirable to fully and accurately evaluate an investment in the combined company; (c) is not to be considered a recommendation by the combined company that any person make an investment in the combined company; (d) is for information purposes only and shall not constitute an offer to buy, sell, issue or subscribe for, or the solicitation of an offer to buy, sell or issue, or subscribe for any securities of the combined company in any jurisdiction in which such offer solicitation or sale would be unlawful. Where any opinion or belief is expressed in this Presentation, it is based on certain assumptions and limitations and is an expression of present opinion or belief only. This Presentation should not be construed as legal, financial or tax advice to any individual, as each individual's circumstances are different. This Presentation is for informational purposes only and should not be considered a solicitation or recommendation to purchase, sell or hold a security. Certain matters discussed in this Presentation may contain forward-looking statements that are, by their nature, subject to significant risks and uncertainties. Forward-looking statements can be identified by words such as “may,” “will,” “should,” “would,” “could,” “believe,” “expect,” “anticipate,” “intend,” “plan,” “continue,” “seek,” “estimate,” “potential” or the negative of these terms or other similar terms. Forward-looking statements in this Presentation include, but are not limited to, statements about: Sensei, Faeth, the concurrent financing and the acquisition of Faeth by Sensei (the Transactions ), including the closing of the concurrent financing, if any, and the expected effects, perceived benefits or opportunities and related timing with respect thereto; expectations regarding the use of proceeds from the concurrent financing and cash runway expectations therefrom, including such proceeds funding the combined company through key clinical milestones; the combined company’s product candidates and the potential benefits thereof; planned and ongoing pre-clinical and clinical studies, including the timing for data readouts and future development milestones; the potential peak sales for the combined company’s product candidates; the combined company’s CMC infrastructure plans and positioning for growth, including expectations regarding DS/DP supply for ongoing and proposed clinical trials; and expectations regarding patent protection for the combined company’s product candidates, including plans to file patent applications and the timing thereof. Such forward-looking statements reflect the current views of the combined company’s management regarding future events; they are not guarantees of future performance. These forward-looking statements involve significant risks and uncertainties that could cause actual results to differ materially and adversely from results expressed or implied by this Presentation, including, amongst others: the inability to complete the concurrent financing; costs related to the proposed transaction; the ability of the combined company to obtain sufficient additional capital to further advance its clinical programs; the ability of the combined company’s clinical trials to demonstrate acceptable safety and efficacy of its product candidates and other positive results; the progress of the combined company’s preclinical studies and clinical trials; risks related to clinical development and regulatory approval of the Company's product candidates, including potential delays in the commencement, enrollment and completion of clinical trials; the size of the market opportunities for the combined company’s product candidates; competition in the combined company’s industry; changes in applicable laws or regulations and other risks and uncertainties from time to time described in Sensei's Quarterly Report on Form 10-Q for the quarter ended September 30, 2025, including those under the Risk Factors section therein, and in Sensei's other filings with the U.S. Securities and Exchange Commission. The combined company assumes no obligation to update any forward-looking information contained in this Presentation. Certain information contained in this Presentation related to or are based on studies, publications and other data obtained from third party sources as well as our own internal estimates and research. While the combined company believes that such third-party sources are reliable, there can be no assurance as to the accuracy or completeness of the indicated information. The combined company has not independently verified the information provided by such third-party sources. This Presentation may contain trademarks, service marks, trade names and copyrights of other companies, which are the property of their respective owners. Solely for convenience, some of the trademarks, service marks, trade names and copyrights referred to in this Presentation may be listed without the TM, SM or © or ® symbols, but the combined company will assert, to the fullest extent under applicable law, the rights of the owners to these trademarks, service marks, trade names and copyrights. CONFIDENTIAL 2

–– Transaction Highlights The acquisition of Faeth was structured as a stock-for-stock transaction whereby all of Faeth’s outstanding equity interests were exchanged for a combination of shares of Sensei common stock Structure and a newly created, Series B non-voting convertible preferred stock. Concurrent with the acquisition of Faeth, Sensei executed a definitive agreement for a $200 Financing million private placement with a group of institutional accredited investors. Continuing leadership includes Chris Gerry, President, Josiah Craver, SVP, Finance and Anand Management Parikh, Chief Operating Officer. Anand Parikh will also be joining the Sensei board. and BOD The proceeds from the private placement are expected to be primarily used to advance PIKTOR Primary Use of and deliver the following anticipated milestones: Phase 2 topline data in endometrial cancer and the initiation of a Phase 1b trial in HR+/HER2- advanced breast cancer, both expected by YE Proceeds 2026. CONFIDENTIAL 3
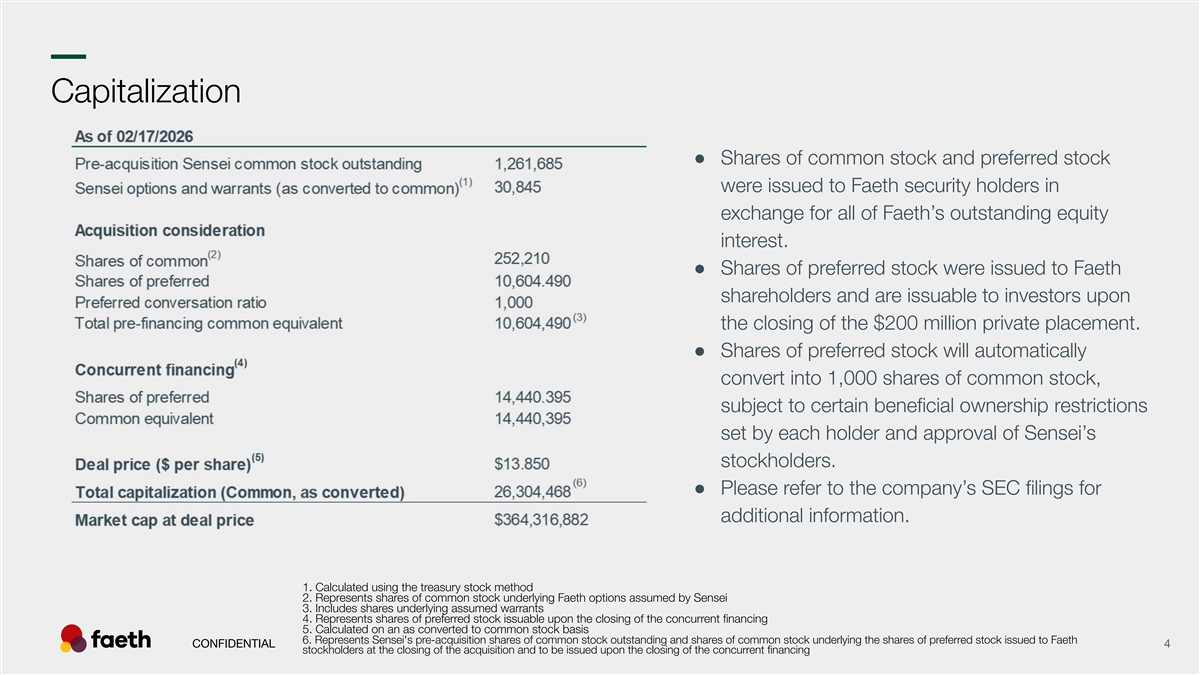
–– Capitalization ● Shares of common stock and preferred stock were issued to Faeth security holders in exchange for all of Faeth’s outstanding equity interest. ● Shares of preferred stock were issued to Faeth shareholders and are issuable to investors upon the closing of the $200 million private placement. ● Shares of preferred stock will automatically convert into 1,000 shares of common stock, subject to certain beneficial ownership restrictions set by each holder and approval of Sensei’s stockholders. ● Please refer to the company’s SEC filings for additional information. 1. Calculated using the treasury stock method 2. Represents shares of common stock underlying Faeth options assumed by Sensei 3. Includes shares underlying assumed warrants 4. Represents shares of preferred stock issuable upon the closing of the concurrent financing 5. Calculated on an as converted to common stock basis 6. Represents Sensei's pre-acquisition shares of common stock outstanding and shares of common stock underlying the shares of preferred stock issued to Faeth CONFIDENTIAL 4 stockholders at the closing of the acquisition and to be issued upon the closing of the concurrent financing

–– Faeth Overview ● Oral multi-node inhibition of PI3K/AKT/mTOR pathway; designed for more complete inhibition, less toxicity ● PIKTOR—investigational combination therapy consisting of two oral small molecules—hitting PI3K-alpha, mTORC1, mTORC2 ● Recent and anticipated readouts: ○ Phase 2 platinum resistant ovarian cancer (ESMO Oct 2025) - met primary endpoint ○ Phase 2 in endometrial cancer reads out EOY 2026 ● Raise is expected to fund the combined company’s product candidates through key clinical readouts, including a Phase 2 trial in second-line advanced endometrial cancer through topline data and initiation of a Phase 1b trial in first- and second-line HR+/HER2- advanced breast cancer, with remaining proceeds for general corporate purposes and completion of Sensei's ongoing Phase 1/2 trial of solnerstotug. CONFIDENTIAL 5

–– Focused pipeline of potentially best-in-class product candidates, leveraging Faeth’s unique metabolic insights IND Discovery Phase I Phase II Phase III Target Population enabling Oncology: Endometrial, Breast, PIKTOR: Serabelisib + Sapanisertib Ovarian, Lung NEAAR: Formulated amino acids Oncology: Rectal (+ Chemo / Radiation) Pediatric Rare Novel small molecule targeting Disease / Neuro amino acid catabolism CONFIDENTIAL 6

–– Faeth scientific founders: world leaders in cancer biology Lew Cantley, PhD Sid Mukherjee, MD PhD Oliver Maddocks, PhD Karen Vousden, PhD Scott Lowe, PhD Greg Hannon, PhD Dana Farber Cancer Institute, Asst. Prof., Columbia, Pulitzer Prize CSO & Co-Founder Group Leader, Crick Institute, Chair of Cancer Biology & Genetics at Director of CRUK Cambridge Harvard. Founder: Agios Winner, Time 100 Most Influential Honorary Professor of Cancer Biology & Former Chief Scientist of CRUK, Memorial Sloan Kettering, Institute (NASDAQ), People, Founder: Vor (NASDAQ) Metabolism, University of Glasgow Director, Bristol Myers Squibb Founder: ORIC (NASDAQ) Petra, Volastra CONFIDENTIAL 7
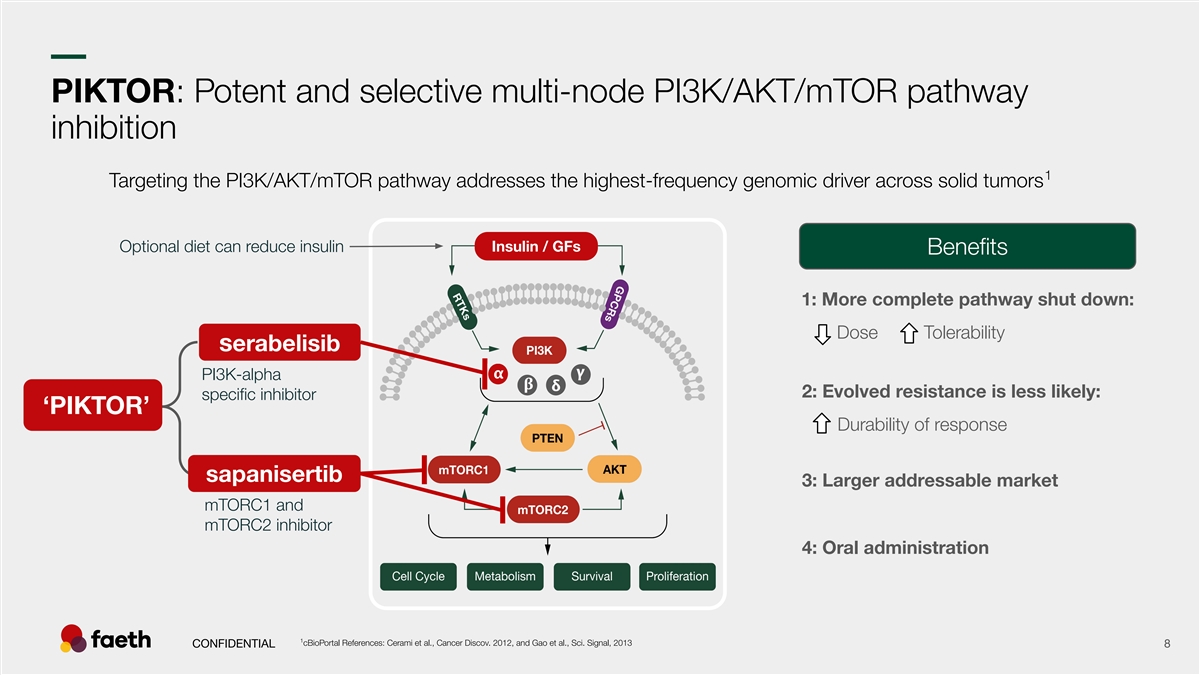
–– PIKTOR: Potent and selective multi-node PI3K/AKT/mTOR pathway inhibition 1 Targeting the PI3K/AKT/mTOR pathway addresses the highest-frequency genomic driver across solid tumors Optional diet can reduce insulin Insulin / GFs Benefits 1: More complete pathway shut down: Dose Tolerability serabelisib PI3K-alpha 2: Evolved resistance is less likely: specific inhibitor ‘PIKTOR’ Durability of response sapanisertib 3: Larger addressable market mTORC1 and mTORC2 inhibitor 4: Oral administration 1 cBioPortal References: Cerami et al., Cancer Discov. 2012, and Gao et al., Sci. Signal, 2013 CONFIDENTIAL 8
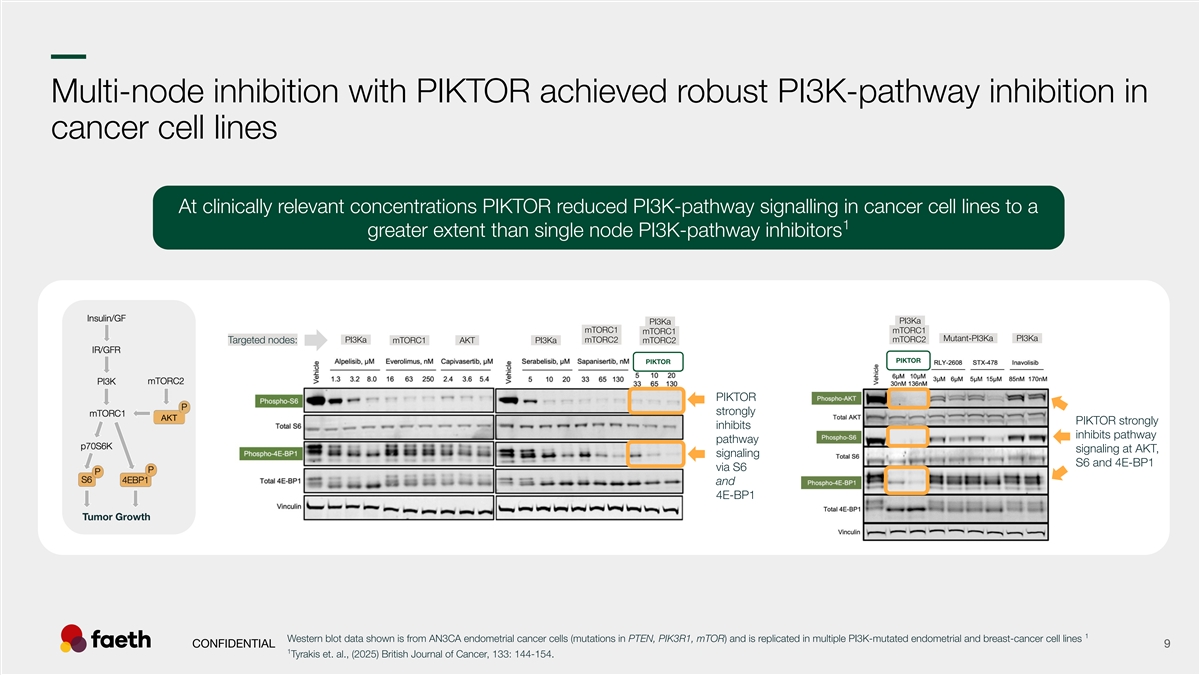
–– umor GrTowth Multi-node inhibition with PIKTOR achieved robust PI3K-pathway inhibition in cancer cell lines At clinically relevant concentrations PIKTOR reduced PI3K-pathway signalling in cancer cell lines to a 1 greater extent than single node PI3K-pathway inhibitors –PI3Ka– –PI3Ka– -mTORC1- -mTORC1- -mTORC1- –Mutant-PI3Ka– –PI3Ka– –PI3Ka– -mTORC2- -mTORC2- Targeted nodes: -mTORC1- –AKT– –PI3Ka– -mTORC2- PIKTOR PIKTOR PIKTOR strongly AKT PIKTOR strongly inhibits inhibits pathway pathway signaling at AKT, signaling S6 and 4E-BP1 via S6 and 4E-BP1 1 Western blot data shown is from AN3CA endometrial cancer cells (mutations in PTEN, PIK3R1, mTOR) and is replicated in multiple PI3K-mutated endometrial and breast-cancer cell lines CONFIDENTIAL 9 1 Tyrakis et. al., (2025) British Journal of Cancer, 133: 144-154.
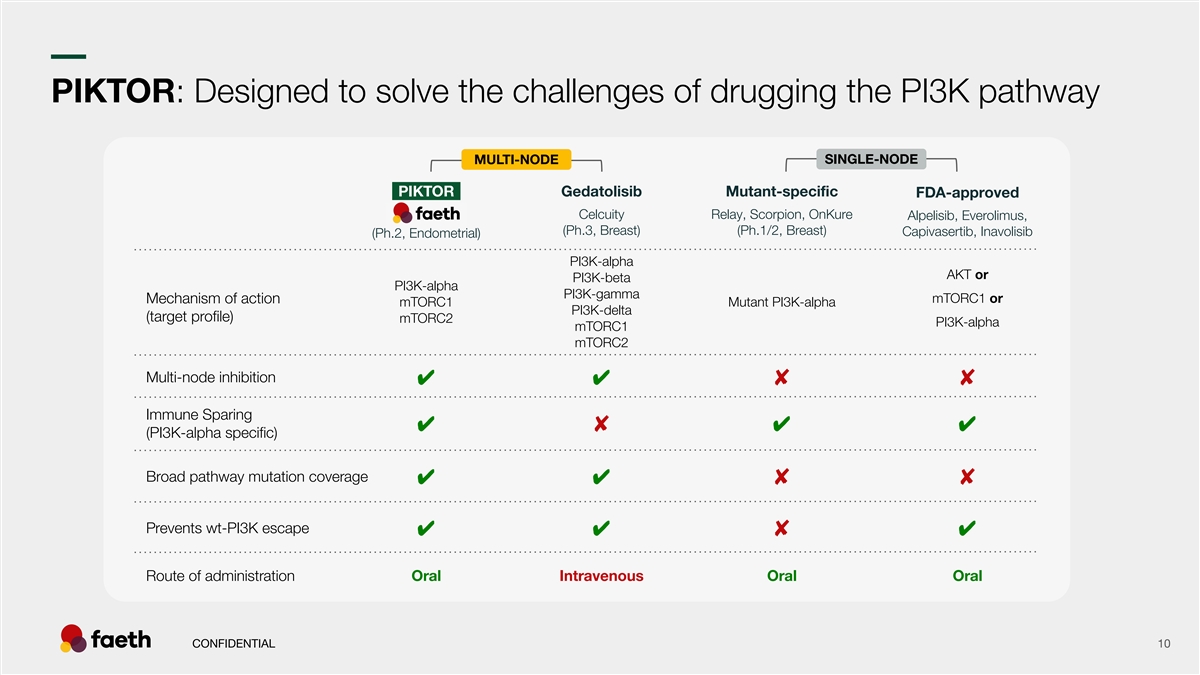
–– PIKTOR: Designed to solve the challenges of drugging the PI3K pathway SINGLE-NODE MULTI-NODE -PIKTOR- Gedatolisib Mutant-specific FDA-approved Celcuity Relay, Scorpion, OnKure Alpelisib, Everolimus, Faeth (Ph.3, Breast) (Ph.1/2, Breast) Capivasertib, Inavolisib (Ph.2, Endometrial) PI3K-alpha AKT or PI3K-beta PI3K-alpha PI3K-gamma mTORC1 or Mechanism of action mTORC1 Mutant PI3K-alpha PI3K-delta (target profile) mTORC2 PI3K-alpha mTORC1 mTORC2 Multi-node inhibition ✔✔ ✘✘ Immune Sparing ✔✔✔ ✘ (PI3K-alpha specific) Broad pathway mutation coverage ✔✔ ✘✘ Prevents wt-PI3K escape ✔✔✔ ✘ Route of administration Oral Intravenous Oral Oral CONFIDENTIAL 10
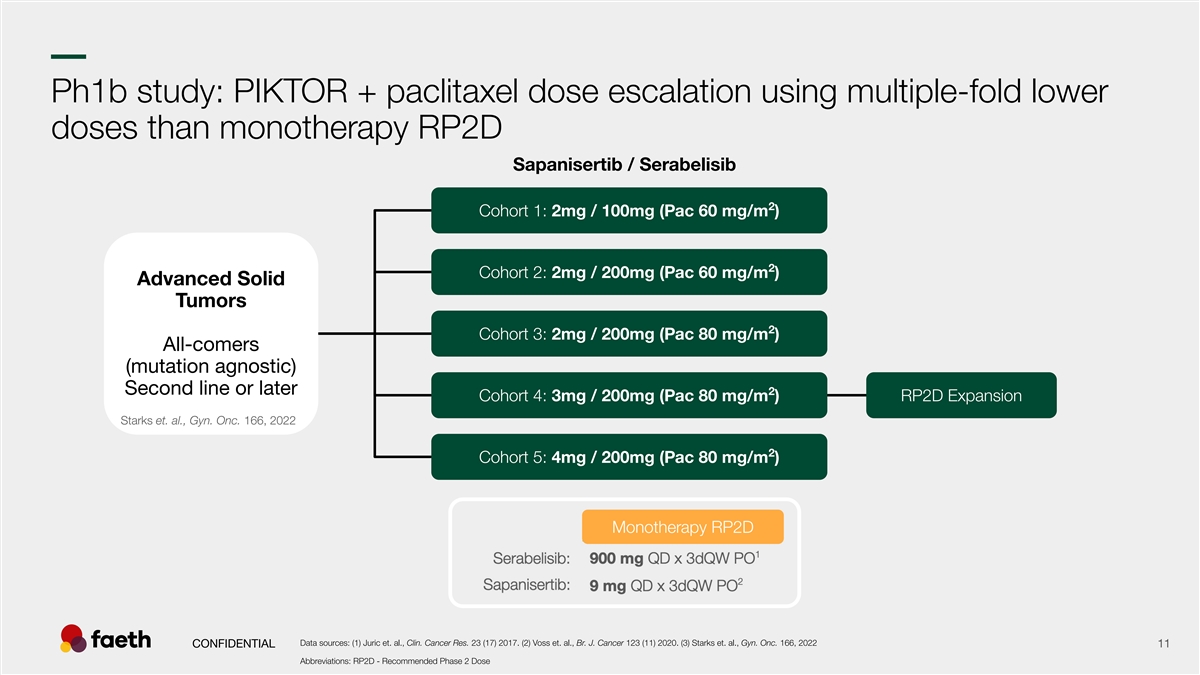
–– Ph1b study: PIKTOR + paclitaxel dose escalation using multiple-fold lower doses than monotherapy RP2D Sapanisertib / Serabelisib 2 Cohort 1: 2mg / 100mg (Pac 60 mg/m ) 2 Cohort 2: 2mg / 200mg (Pac 60 mg/m ) Advanced Solid Tumors 2 Cohort 3: 2mg / 200mg (Pac 80 mg/m ) All-comers (mutation agnostic) Second line or later 2 Cohort 4: 3mg / 200mg (Pac 80 mg/m ) RP2D Expansion Starks et. al., Gyn. Onc. 166, 2022 2 Cohort 5: 4mg / 200mg (Pac 80 mg/m ) Monotherapy RP2D Data sources: (1) Juric et. al., Clin. Cancer Res. 23 (17) 2017. (2) Voss et. al., Br. J. Cancer 123 (11) 2020. (3) Starks et. al., Gyn. Onc. 166, 2022 CONFIDENTIAL 11 Abbreviations: RP2D - Recommended Phase 2 Dose
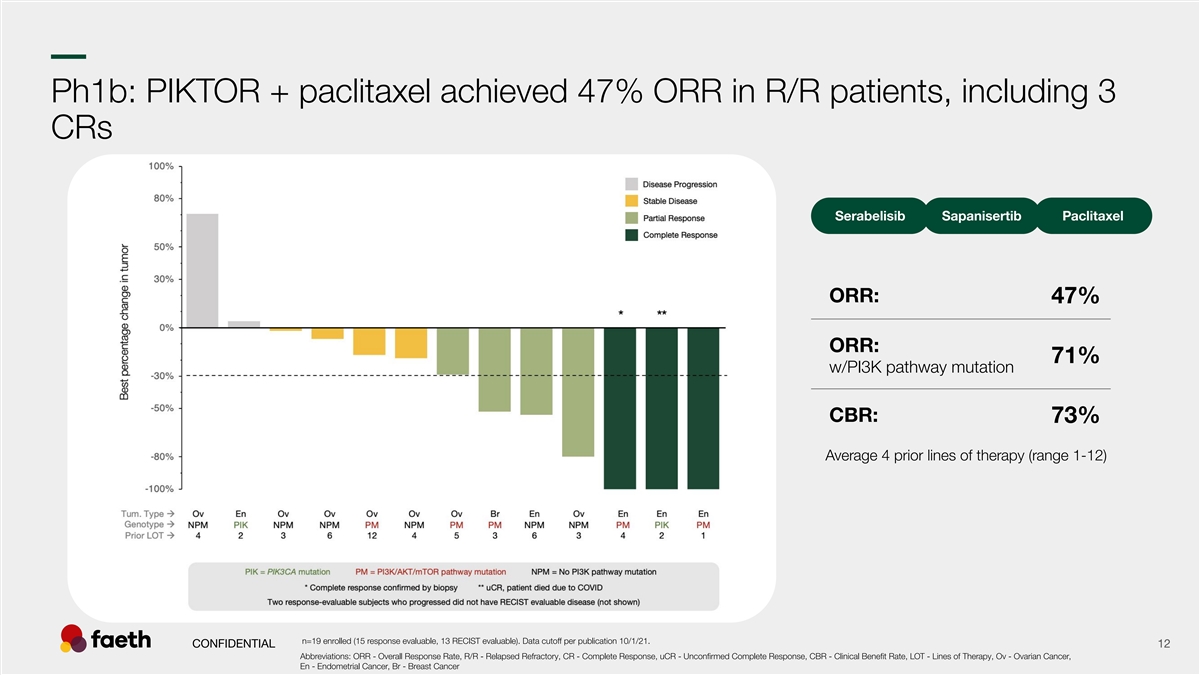
–– Ph1b: PIKTOR + paclitaxel achieved 47% ORR in R/R patients, including 3 CRs Serabelisib Sapanisertib Paclitaxel ORR: 47% 12 ORR: 71% w/PI3K pathway mutation CBR: 73% Average 4 prior lines of therapy (range 1-12) n=19 enrolled (15 response evaluable, 13 RECIST evaluable). Data cutoff per publication 10/1/21. CONFIDENTIAL 12 Abbreviations: ORR - Overall Response Rate, R/R - Relapsed Refractory, CR - Complete Response, uCR - Unconfirmed Complete Response, CBR - Clinical Benefit Rate, LOT - Lines of Therapy, Ov - Ovarian Cancer, En - Endometrial Cancer, Br - Breast Cancer
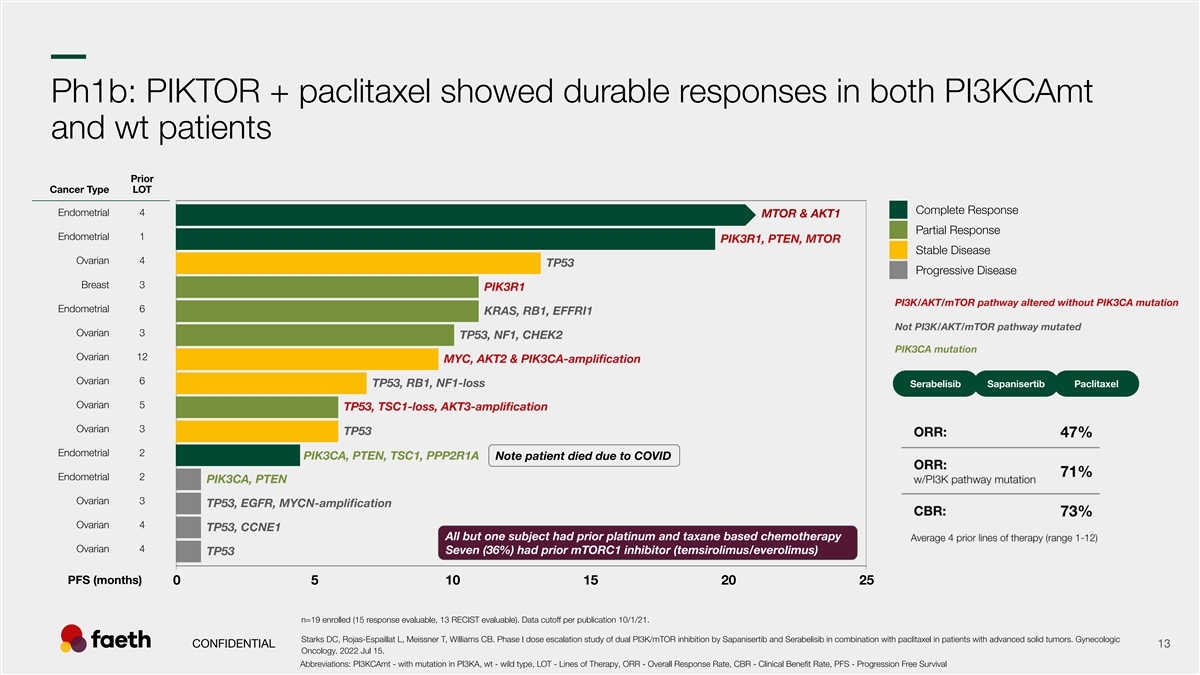
–– Ph1b: PIKTOR + paclitaxel showed durable responses in both PI3KCAmt and wt patients Prior Cancer Type LOT Complete Response Endometrial 4 MTOR & AKT1 Partial Response Endometrial 1 PIK3R1, PTEN, MTOR Stable Disease Ovarian 4 TP53 Progressive Disease Breast 3 PIK3R1 PI3K/AKT/mTOR pathway altered without PIK3CA mutation Endometrial 6 KRAS, RB1, EFFRI1 Not PI3K/AKT/mTOR pathway mutated Ovarian 3 TP53, NF1, CHEK2 PIK3CA mutation Ovarian 12 MYC, AKT2 & PIK3CA-amplification Ovarian 6 TP53, RB1, NF1-loss Serabelisib Sapanisertib Paclitaxel Ovarian 5 TP53, TSC1-loss, AKT3-amplification Ovarian 3 TP53 Endometrial 2 PIK3CA, PTEN, TSC1, PPP2R1A Note patient died due to COVID Endometrial 2 PIK3CA, PTEN Ovarian 3 TP53, EGFR, MYCN-amplification Ovarian 4 TP53, CCNE1 All but one subject had prior platinum and taxane based chemotherapy Ovarian 4 Seven (36%) had prior mTORC1 inhibitor (temsirolimus/everolimus) TP53 PFS (months) 0 5 10 15 20 25 n=19 enrolled (15 response evaluable, 13 RECIST evaluable). Data cutoff per publication 10/1/21. Starks DC, Rojas-Espaillat L, Meissner T, Williams CB. Phase I dose escalation study of dual PI3K/mTOR inhibition by Sapanisertib and Serabelisib in combination with paclitaxel in patients with advanced solid tumors. Gynecologic CONFIDENTIAL 13 Oncology. 2022 Jul 15. Abbreviations: PI3KCAmt - with mutation in PI3KA, wt - wild type, LOT - Lines of Therapy, ORR - Overall Response Rate, CBR - Clinical Benefit Rate, PFS - Progression Free Survival

–– Ph 1b Safety Highlights: PIKTOR was generally well tolerated ● Favorable toxicity profile compared to approved agents Single agent Lenvatinib and PIKTOR and chemo in 2L 2 1 Pembrolizumab Paclitaxel 3 endometrial Grade 3 AEs 58% 89% 73% Discontinuation ~5% 33% 8% ● Primarily low grade AEs - Common AEs included Gr 1, 2 events for nausea, decreased appetite, diarrhea, fatigue, neutropenia and anemia (potentially due to 1 paclitaxel) The results are presented from different clinical trials at different points in time with differences in trial design. No head-to-head trials have been conducted among the results shown and cross-trial comparisons must be interpreted with caution. As a result, conclusive cross-trial comparisons cannot be made. Data sources: (1) Starks et al. Phase I dose escalation study of dual PI3K/mTOR inhibition by Sapanisertib and Serabelisib in combination with paclitaxel in patients with advanced solid tumors. Gynecologic Oncology. 2022 Jul 15. (2) Makker et al., NEJM 2022 (KEYNOTE-775) (3) Chemotherapy: Control arm from KEYNOTE trial; Treating investigator’s choice (doxorubicin 60 mg/m2 IV weekly or paclitaxel 80 mg/m2 IV weekly with 3 weeks on and 1 week off. Data cutoff CONFIDENTIAL 14 per publication 10/1/21. Abbreviations: AE - Adverse Events
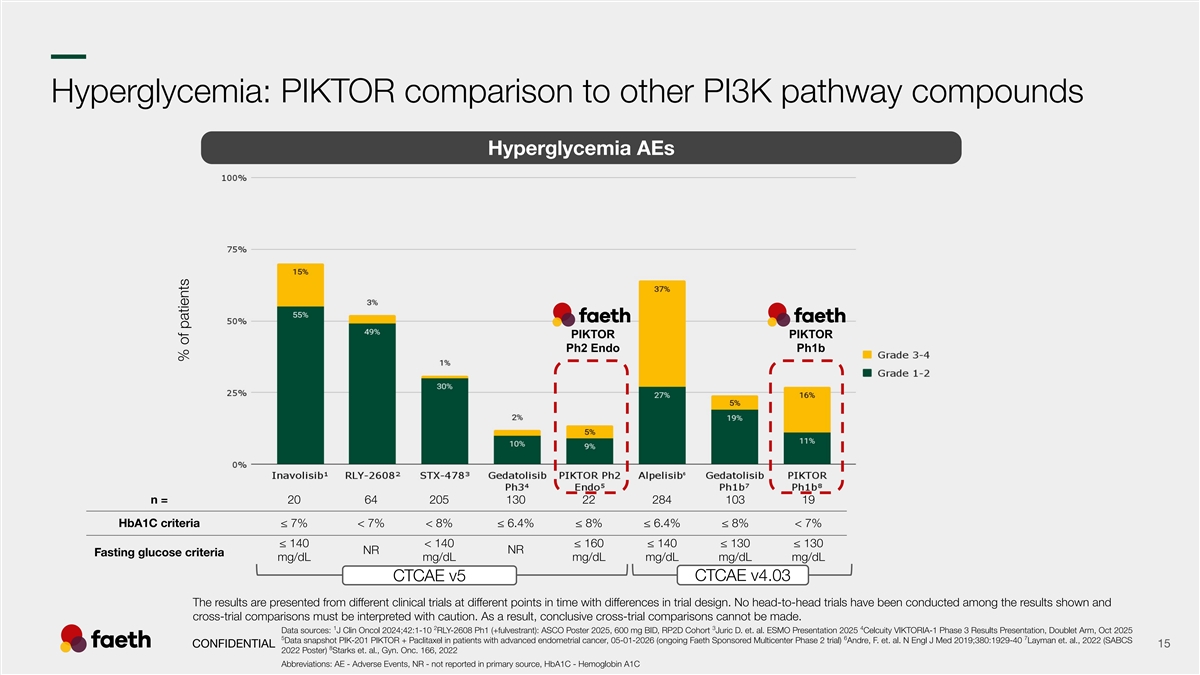
–– Hyperglycemia: PIKTOR comparison to other PI3K pathway compounds Hyperglycemia AEs PIKTOR PIKTOR Ph2 Endo Ph1b n = 20 64 205 130 22 284 103 19 HbA1C criteria ≤ 7% < 7% < 8% ≤ 6.4% ≤ 8% ≤ 6.4% ≤ 8% < 7% ≤ 140 < 140 ≤ 160 ≤ 140 ≤ 130 ≤ 130 NR NR Fasting glucose criteria mg/dL mg/dL mg/dL mg/dL mg/dL mg/dL CTCAE v4.03 CTCAE v5 The results are presented from different clinical trials at different points in time with differences in trial design. No head-to-head trials have been conducted among the results shown and cross-trial comparisons must be interpreted with caution. As a result, conclusive cross-trial comparisons cannot be made. 1 2 3 4 Data sources: J Clin Oncol 2024;42:1-10 RLY-2608 Ph1 (+fulvestrant): ASCO Poster 2025, 600 mg BID, RP2D Cohort Juric D. et. al. ESMO Presentation 2025 Celcuity VIKTORIA-1 Phase 3 Results Presentation, Doublet Arm, Oct 2025 5 6 7 Data snapshot PIK-201 PIKTOR + Paclitaxel in patients with advanced endometrial cancer, 05-01-2026 (ongoing Faeth Sponsored Multicenter Phase 2 trial) Andre, F. et. al. N Engl J Med 2019;380:1929-40 Layman et. al., 2022 (SABCS CONFIDENTIAL 15 8 2022 Poster) Starks et. al., Gyn. Onc. 166, 2022 Abbreviations: AE - Adverse Events, NR - not reported in primary source, HbA1C - Hemoglobin A1C % of patients
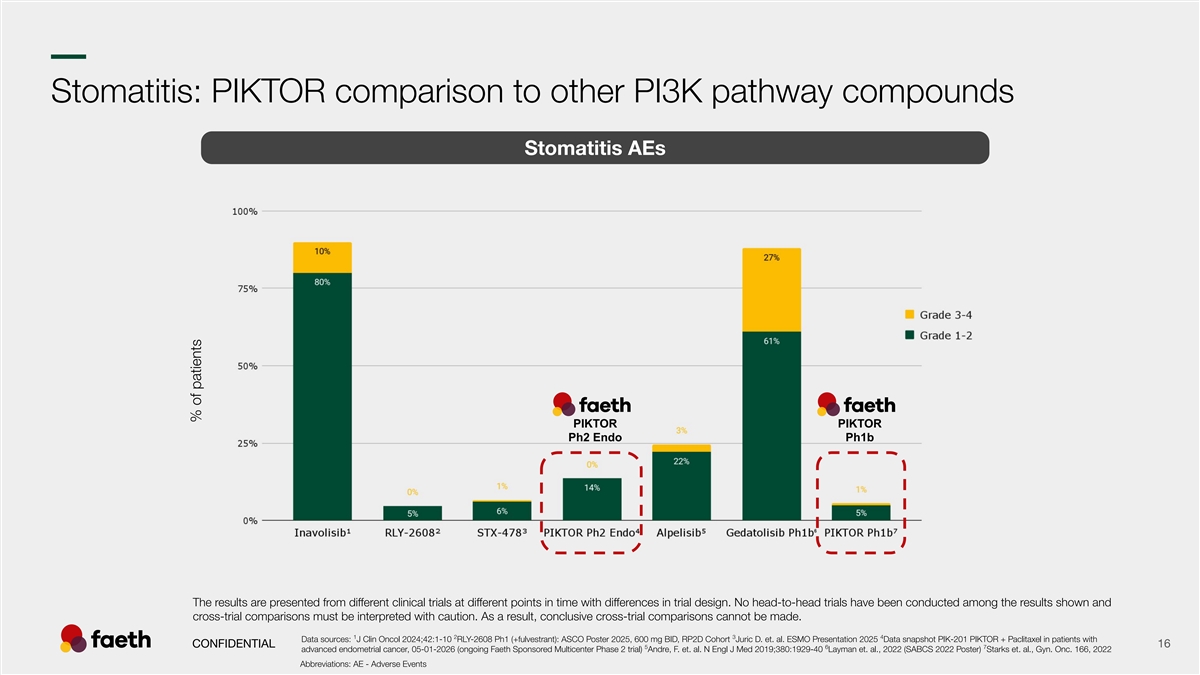
–– Stomatitis: PIKTOR comparison to other PI3K pathway compounds Stomatitis AEs PIKTOR PIKTOR Ph2 Endo Ph1b The results are presented from different clinical trials at different points in time with differences in trial design. No head-to-head trials have been conducted among the results shown and cross-trial comparisons must be interpreted with caution. As a result, conclusive cross-trial comparisons cannot be made. 1 2 3 4 Data sources: J Clin Oncol 2024;42:1-10 RLY-2608 Ph1 (+fulvestrant): ASCO Poster 2025, 600 mg BID, RP2D Cohort Juric D. et. al. ESMO Presentation 2025 Data snapshot PIK-201 PIKTOR + Paclitaxel in patients with CONFIDENTIAL 16 5 6 7 advanced endometrial cancer, 05-01-2026 (ongoing Faeth Sponsored Multicenter Phase 2 trial) Andre, F. et. al. N Engl J Med 2019;380:1929-40 Layman et. al., 2022 (SABCS 2022 Poster) Starks et. al., Gyn. Onc. 166, 2022 Abbreviations: AE - Adverse Events % of patients % of patients
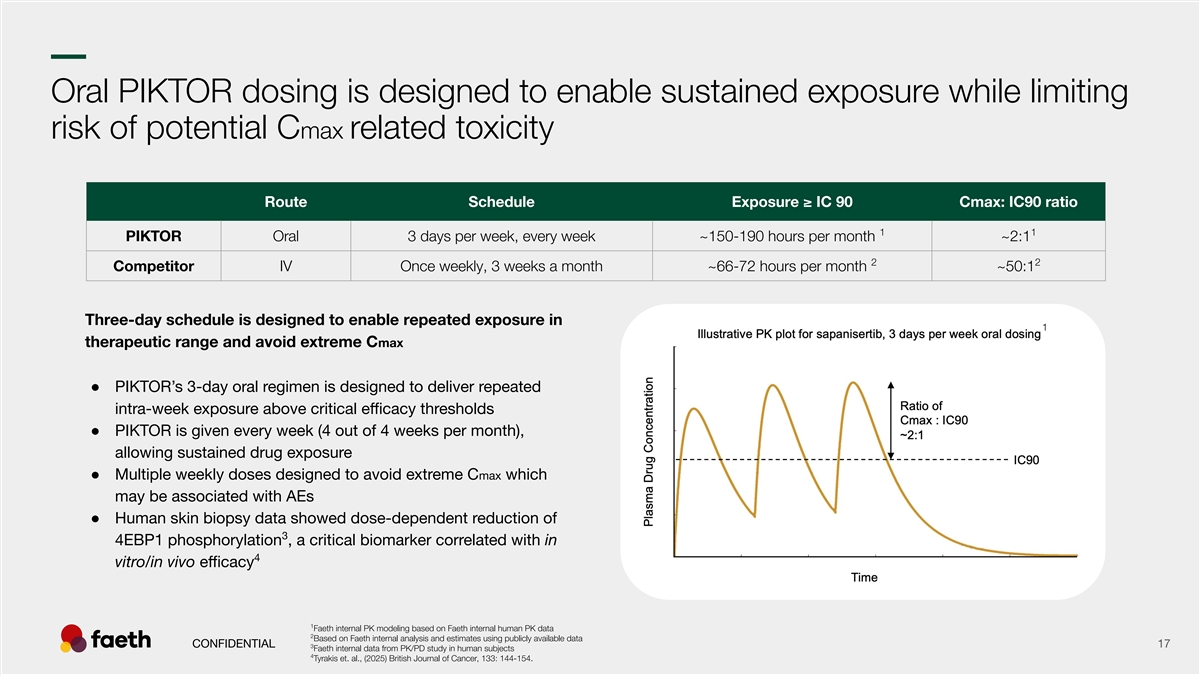
–– Oral PIKTOR dosing is designed to enable sustained exposure while limiting risk of potential Cmax related toxicity Route Schedule Exposure ≥ IC 90 Cmax: IC90 ratio 1 1 PIKTOR Oral 3 days per week, every week ~150-190 hours per month ~2:1 2 2 Competitor IV Once weekly, 3 weeks a month ~66-72 hours per month ~50:1 Three-day schedule is designed to enable repeated exposure in 1 therapeutic range and avoid extreme Cmax ● PIKTOR’s 3-day oral regimen is designed to deliver repeated intra-week exposure above critical efficacy thresholds ● PIKTOR is given every week (4 out of 4 weeks per month), allowing sustained drug exposure ● Multiple weekly doses designed to avoid extreme Cmax which may be associated with AEs ● Human skin biopsy data showed dose-dependent reduction of 3 4EBP1 phosphorylation , a critical biomarker correlated with in 4 vitro/in vivo efficacy 1 Faeth internal PK modeling based on Faeth internal human PK data 2 Based on Faeth internal analysis and estimates using publicly available data CONFIDENTIAL 17 3 Faeth internal data from PK/PD study in human subjects 4 Tyrakis et. al., (2025) British Journal of Cancer, 133: 144-154.

–– PIKTOR clinical development ● We believe this indication has the greatest First Indication: 2L advanced EC potential for first approval Phase 2 enrolling Endometrial ● Large unmet clinical need in 2L 1 Estimated market size of ~$1-1.5B ● >80% PI3K/AKT/mTOR pathway mutated First Indication:1L/ 2L HR+/HER2- ● Well understood mechanism with potential for Phase 1b advanced BC broad label protocol drafted Breast ● 60% PI3K/AKT/mTOR pathway mutated 1 Estimated market size of ~$8-9B ● Oral dosage form advantage vs. gedatolisib Phase 2 complete ● Demonstrated activity in an all-comers First Indication: Advanced (Sapa + Pac); population platinum-resistant OC Ovarian FDA interaction ● 60% PI3K/AKT/mTOR pathway mutated 1 planned in 2026 ● Successful Phase 2 complete Estimated market size of ~$1.5-2B Phase 2 protocol First Indication: NFE2L2/KEAP1 advanced drafted - ● Promising single agent activity of sapanisertib NSCLC Lung LungMAP in NFE2L2/KEAP1 mutated NSCLC consortium IIT 1 Estimated market size of ~$1-1.5B 1 Based on estimated sales data, assuming FDA approval, contained in Deallus’ February 12, 2024 report to the Company CONFIDENTIAL 18
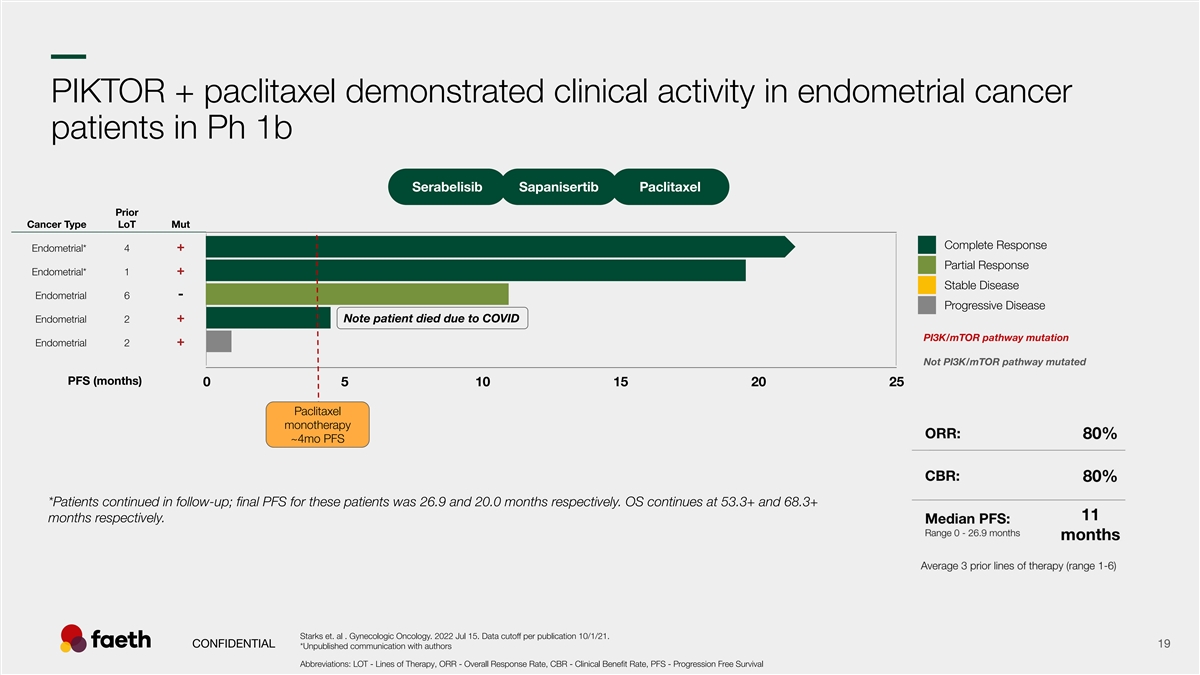
–– PIKTOR + paclitaxel demonstrated clinical activity in endometrial cancer patients in Ph 1b Serabelisib Sapanisertib Paclitaxel Prior Cancer Type LoT Mut Complete Response + Endometrial* 4 Partial Response + Endometrial* 1 Stable Disease - Endometrial 6 Progressive Disease + Endometrial 2 Note patient died due to COVID PI3K/mTOR pathway mutation + Endometrial 2 Not PI3K/mTOR pathway mutated PFS (months) 0 5 10 15 20 25 Paclitaxel monotherapy ~4mo PFS *Patients continued in follow-up; final PFS for these patients was 26.9 and 20.0 months respectively. OS continues at 53.3+ and 68.3+ months respectively. Starks et. al . Gynecologic Oncology. 2022 Jul 15. Data cutoff per publication 10/1/21. CONFIDENTIAL 19 *Unpublished communication with authors Abbreviations: LOT - Lines of Therapy, ORR - Overall Response Rate, CBR - Clinical Benefit Rate, PFS - Progression Free Survival
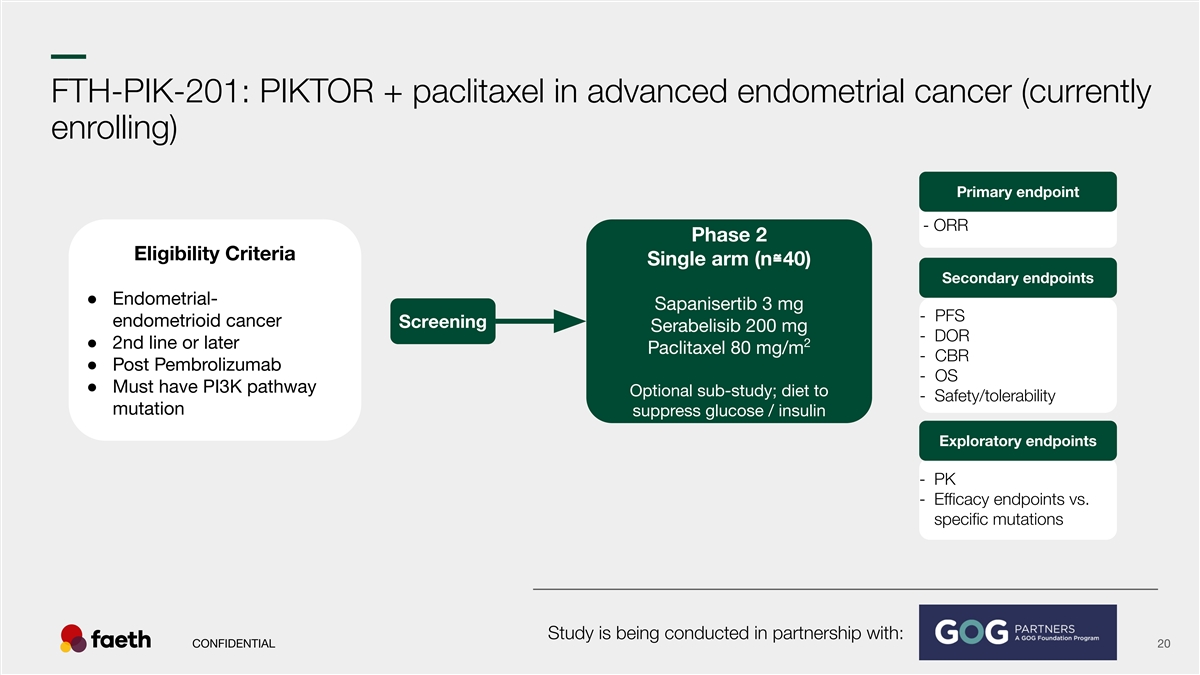
–– FTH-PIK-201: PIKTOR + paclitaxel in advanced endometrial cancer (currently enrolling) Primary endpoint - ORR Phase 2 Eligibility Criteria Single arm (n≅40) Secondary endpoints ● Endometrial- Sapanisertib 3 mg - PFS endometrioid cancer Screening Serabelisib 200 mg - DOR 2 ● 2nd line or later Paclitaxel 80 mg/m - CBR ● Post Pembrolizumab - OS ● Must have PI3K pathway Optional sub-study; diet to - Safety/tolerability mutation suppress glucose / insulin Exploratory endpoints - PK - Efficacy endpoints vs. specific mutations Study is being conducted in partnership with: CONFIDENTIAL 20

–– PIKTOR clinical development ● We believe this indication has the greatest First Indication: 2L advanced EC potential for first approval Phase 2 enrolling Endometrial ● Large unmet clinical need in 2L 1 Estimated market size of ~$1-1.5B ● >80% PI3K/AKT/mTOR pathway mutated First Indication:1L/ 2L HR+/HER2- ● Well understood mechanism with potential for Phase 1b advanced BC broad label protocol drafted Breast ● 60% PI3K/AKT/mTOR pathway mutated 1 Estimated market size of ~$8-9B ● Oral dosage form advantage vs. gedatolisib ● Demonstrated activity in an all-comers Phase 2 complete First Indication: Advanced population platinum-resistant OC (Sapa + Pac); Ovarian ● 60% PI3K/AKT/mTOR pathway mutated FDA interaction 1 ● Successful Phase 2 complete Estimated market size of ~$1.5-2B planned in 2026 Phase 2 protocol First Indication: NFE2L2/KEAP1 advanced drafted - ● Promising single agent activity of sapanisertib NSCLC Lung LungMAP in NFE2L2/KEAP1 mutated NSCLC consortium IIT 1 Estimated market size of ~$1-1.5B 1 Based on estimated sales data, assuming FDA approval, contained in Deallus’ February 12, 2024 report to the Company CONFIDENTIAL 21
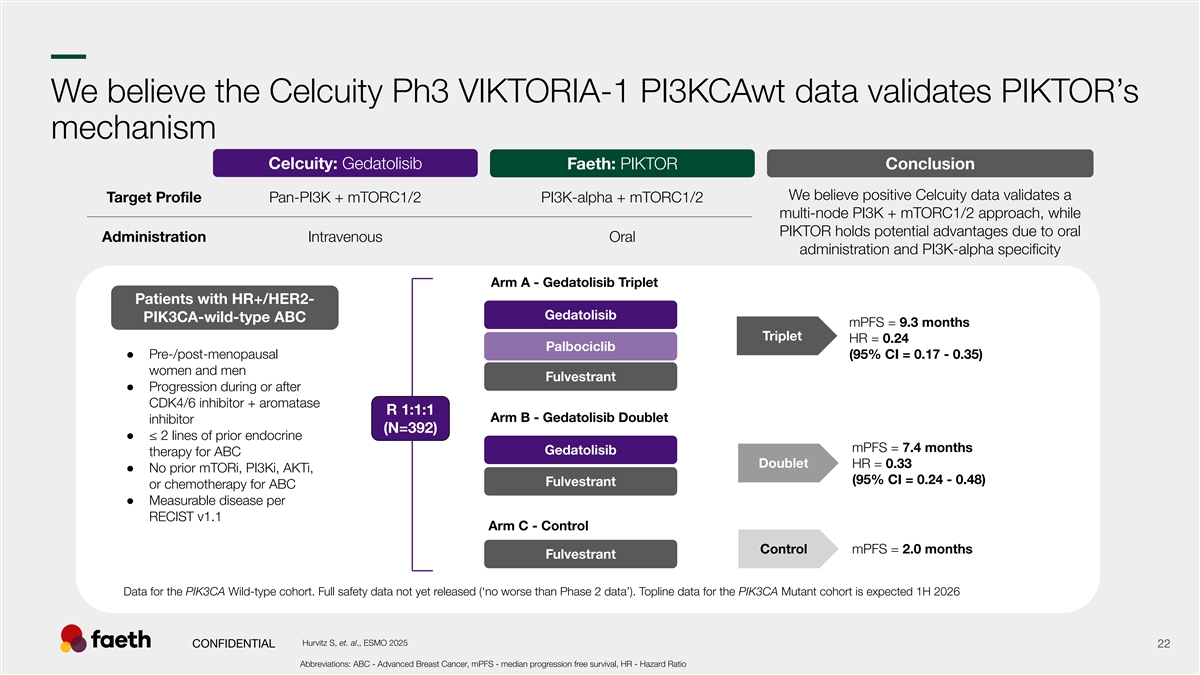
–– We believe the Celcuity Ph3 VIKTORIA-1 PI3KCAwt data validates PIKTOR’s mechanism Celcuity: Gedatolisib Faeth: PIKTOR Conclusion We believe positive Celcuity data validates a Target Profile Pan-PI3K + mTORC1/2 PI3K-alpha + mTORC1/2 multi-node PI3K + mTORC1/2 approach, while PIKTOR holds potential advantages due to oral Administration Intravenous Oral administration and PI3K-alpha specificity Arm A - Gedatolisib Triplet Patients with HR+/HER2- Gedatolisib PIK3CA-wild-type ABC mPFS = 9.3 months Triplet HR = 0.24 Palbociclib ● Pre-/post-menopausal (95% CI = 0.17 - 0.35) women and men Fulvestrant ● Progression during or after CDK4/6 inhibitor + aromatase R 1:1:1 Arm B - Gedatolisib Doublet inhibitor (N=392) ● ≤ 2 lines of prior endocrine mPFS = 7.4 months Gedatolisib therapy for ABC Doublet HR = 0.33 ● No prior mTORi, PI3Ki, AKTi, (95% CI = 0.24 - 0.48) Fulvestrant or chemotherapy for ABC ● Measurable disease per RECIST v1.1 Arm C - Control Control mPFS = 2.0 months Fulvestrant Data for the PIK3CA Wild-type cohort. Full safety data not yet released (‘no worse than Phase 2 data’). Topline data for the PIK3CA Mutant cohort is expected 1H 2026 Hurvitz S, et. al., ESMO 2025 CONFIDENTIAL 22 Abbreviations: ABC - Advanced Breast Cancer, mPFS - median progression free survival, HR - Hazard Ratio
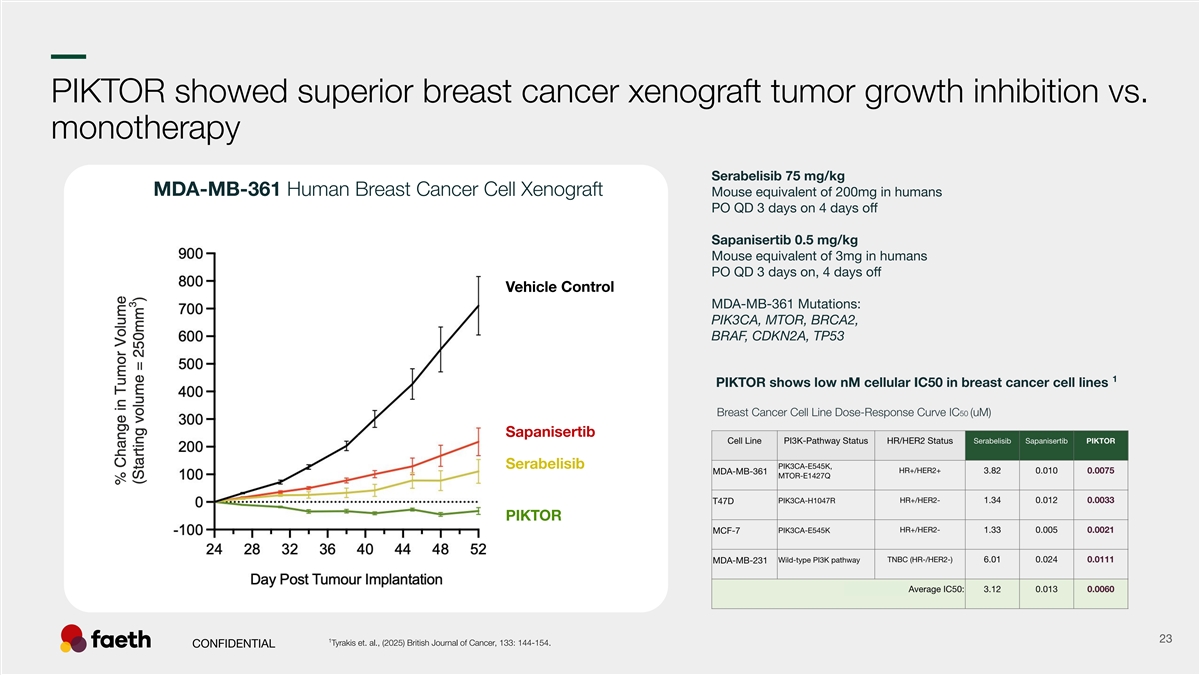
–– PIKTOR showed superior breast cancer xenograft tumor growth inhibition vs. monotherapy Serabelisib 75 mg/kg MDA-MB-361 Human Breast Cancer Cell Xenograft Mouse equivalent of 200mg in humans PO QD 3 days on 4 days off Sapanisertib 0.5 mg/kg Mouse equivalent of 3mg in humans PO QD 3 days on, 4 days off MDA-MB-361 Mutations: PIK3CA, MTOR, BRCA2, BRAF, CDKN2A, TP53 1 PIKTOR shows low nM cellular IC50 in breast cancer cell lines 1 23 Tyrakis et. al., (2025) British Journal of Cancer, 133: 144-154. CONFIDENTIAL Vehicle Control Sapanisertib Serabelisib PIKTOR
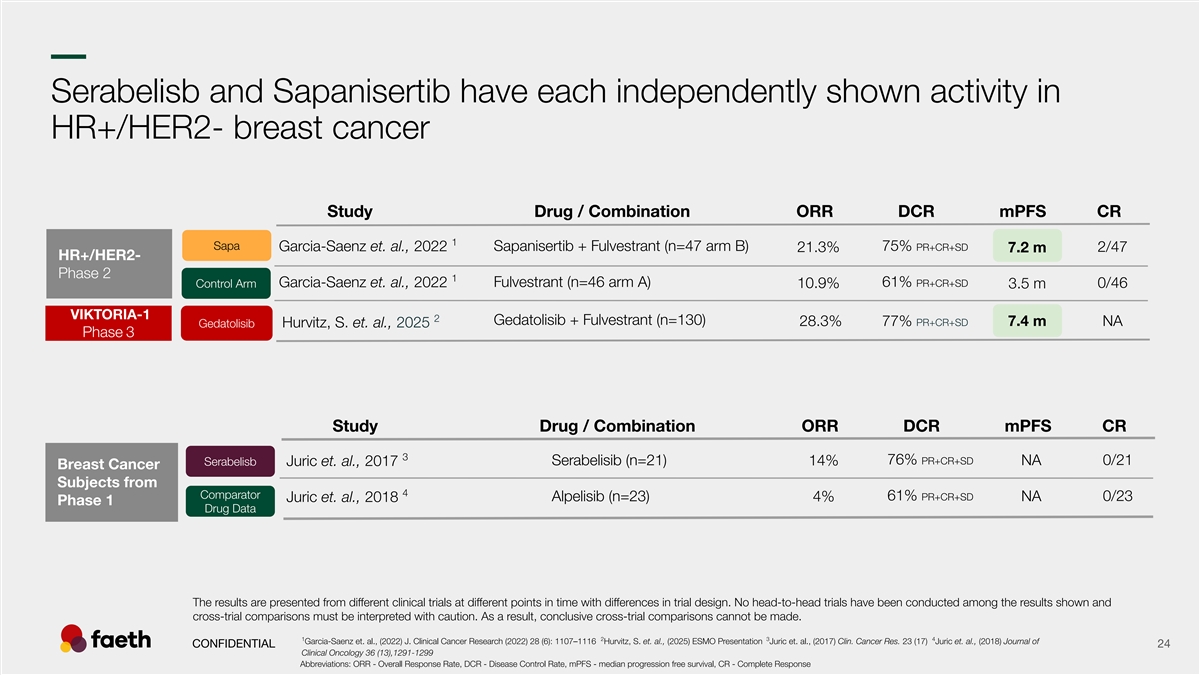
–– Serabelisb and Sapanisertib have each independently shown activity in HR+/HER2- breast cancer Study Drug / Combination ORR DCR mPFS CR 1 Sapa Garcia-Saenz et. al., 2022 Sapanisertib + Fulvestrant (n=47 arm B) 75% PR+CR+SD 21.3% 2/47 7.2 m HR+/HER2- Phase 2 1 Fulvestrant (n=46 arm A) 61% PR+CR+SD Control Arm Garcia-Saenz et. al., 2022 0/46 10.9% 3.5 m VIKTORIA-1 2 Gedatolisib + Fulvestrant (n=130) 28.3% 77% PR+CR+SD 7.4 m NA Gedatolisib Hurvitz, S. et. al., 2025 Phase 3 Study Drug / Combination ORR DCR mPFS CR 3 76% PR+CR+SD Serabelisib (n=21) 14% NA 0/21 Serabelisb Juric et. al., 2017 Breast Cancer Subjects from 4 Comparator 61% PR+CR+SD Alpelisib (n=23) NA 0/23 Juric et. al., 2018 4% Phase 1 Drug Data The results are presented from different clinical trials at different points in time with differences in trial design. No head-to-head trials have been conducted among the results shown and cross-trial comparisons must be interpreted with caution. As a result, conclusive cross-trial comparisons cannot be made. 1 2 3 4 Garcia-Saenz et. al., (2022) J. Clinical Cancer Research (2022) 28 (6): 1107–1116 Hurvitz, S. et. al., (2025) ESMO Presentation Juric et. al., (2017) Clin. Cancer Res. 23 (17) Juric et. al., (2018) Journal of CONFIDENTIAL 24 Clinical Oncology 36 (13),1291-1299 Abbreviations: ORR - Overall Response Rate, DCR - Disease Control Rate, mPFS - median progression free survival, CR - Complete Response
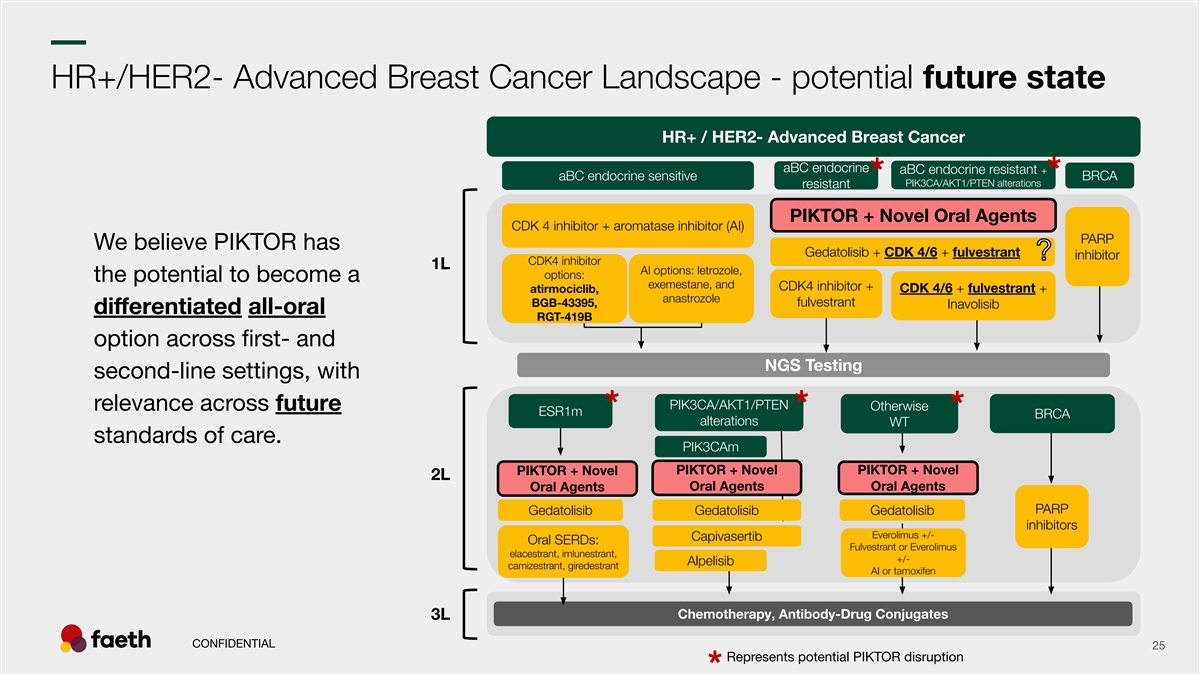
–– HR+/HER2- Advanced Breast Cancer Landscape - potential future state HR+ / HER2- Advanced Breast Cancer aBC endocrine aBC endocrine resistant + aBC endocrine sensitive BRCA * * PIK3CA/AKT1/PTEN alterations resistant PIKTOR + Novel Oral Agents CDK 4 inhibitor + aromatase inhibitor (AI) PARP We believe PIKTOR has Gedatolisib + CDK 4/6 + fulvestrant inhibitor CDK4 inhibitor 1L AI options: letrozole, options: the potential to become a exemestane, and CDK4 inhibitor + atirmociclib, CDK 4/6 + fulvestrant + anastrozole BGB-43395, fulvestrant Inavolisib differentiated all-oral RGT-419B option across first- and NGS Testing second-line settings, with PIK3CA/AKT1/PTEN relevance across future Otherwise ESR1m * * * BRCA alterations WT standards of care. PIK3CAm PIKTOR + Novel PIKTOR + Novel PIKTOR + Novel 2L Oral Agents Oral Agents Oral Agents PARP Gedatolisib Gedatolisib Gedatolisib inhibitors Everolimus +/- Capivasertib Oral SERDs: Fulvestrant or Everolimus elacestrant, imlunestrant, +/- Alpelisib camizestrant, giredestrant AI or tamoxifen Chemotherapy, Antibody-Drug Conjugates 3L CONFIDENTIAL 25 Represents potential PIKTOR disruption *

–– PIKTOR clinical development ● We believe this indication has the greatest First Indication: 2L advanced EC potential for first approval Phase 2 enrolling Endometrial ● Large unmet clinical need in 2L 1 Estimated market size of ~$1-1.5B ● >80% PI3K/AKT/mTOR pathway mutated First Indication:1L/ 2L HR+/HER2- ● Well understood mechanism with potential for Phase 1b advanced BC broad label protocol drafted Breast ● 60% PI3K/AKT/mTOR pathway mutated 1 Estimated market size of ~$8-9B ● Oral dosage form advantage vs. gedatolisib ● Demonstrated activity in an all-comers Phase 2 complete First Indication: Advanced population (Sapa + Pac); platinum-resistant OC Ovarian ● 60% PI3K/AKT/mTOR pathway mutated FDA interaction 1 ● Successful Phase 2 complete Estimated market size of ~$1.5-2B planned in 2026 Phase 2 protocol First Indication: NFE2L2/KEAP1 advanced drafted - ● Promising single agent activity of sapanisertib NSCLC Lung LungMAP in NFE2L2/KEAP1 mutated NSCLC consortium IIT 1 Estimated market size of ~$1-1.5B 1 Based on estimated sales data, assuming FDA approval, contained in Deallus’ February 12, 2024 report to the Company CONFIDENTIAL 26
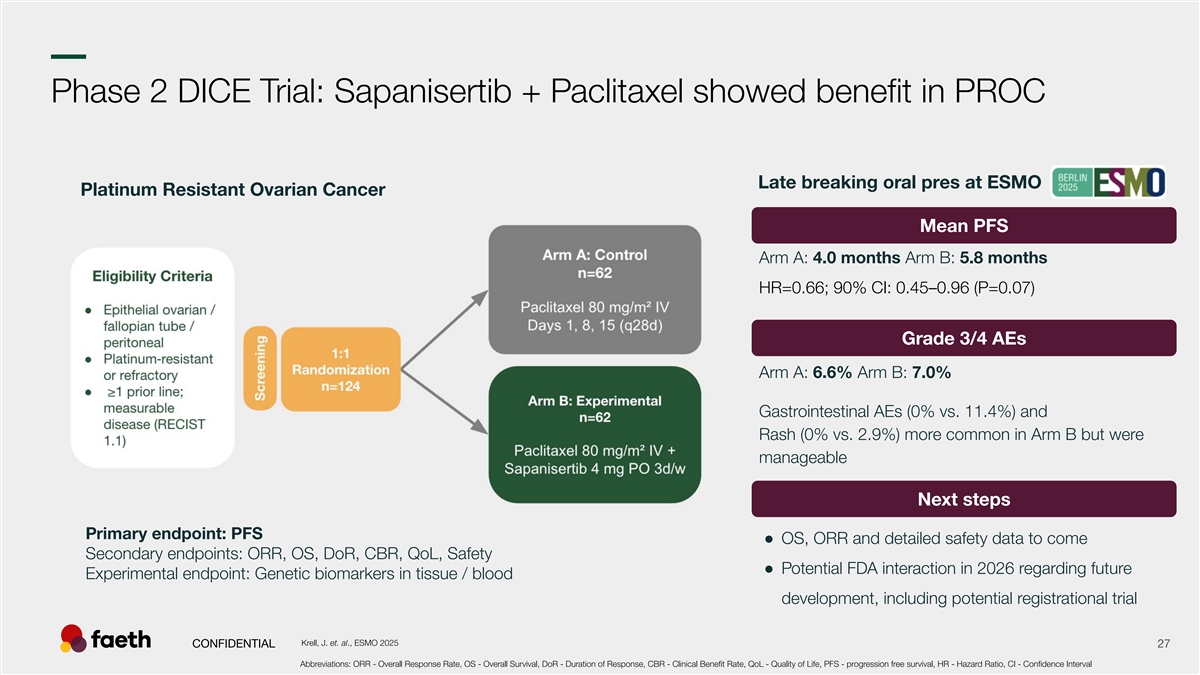
–– Phase 2 DICE Trial: Sapanisertib + Paclitaxel showed benefit in PROC Late breaking oral pres at ESMO Platinum Resistant Ovarian Cancer Mean PFS Arm A: 4.0 months Arm B: 5.8 months HR=0.66; 90% CI: 0.45–0.96 (P=0.07) Grade 3/4 AEs Arm A: 6.6% Arm B: 7.0% Gastrointestinal AEs (0% vs. 11.4%) and Rash (0% vs. 2.9%) more common in Arm B but were manageable Next steps Primary endpoint: PFS ● OS, ORR and detailed safety data to come Secondary endpoints: ORR, OS, DoR, CBR, QoL, Safety ● Potential FDA interaction in 2026 regarding future Experimental endpoint: Genetic biomarkers in tissue / blood development, including potential registrational trial Krell, J. et. al., ESMO 2025 CONFIDENTIAL 27 Abbreviations: ORR - Overall Response Rate, OS - Overall Survival, DoR - Duration of Response, CBR - Clinical Benefit Rate, QoL - Quality of Life, PFS - progression free survival, HR - Hazard Ratio, CI - Confidence Interval
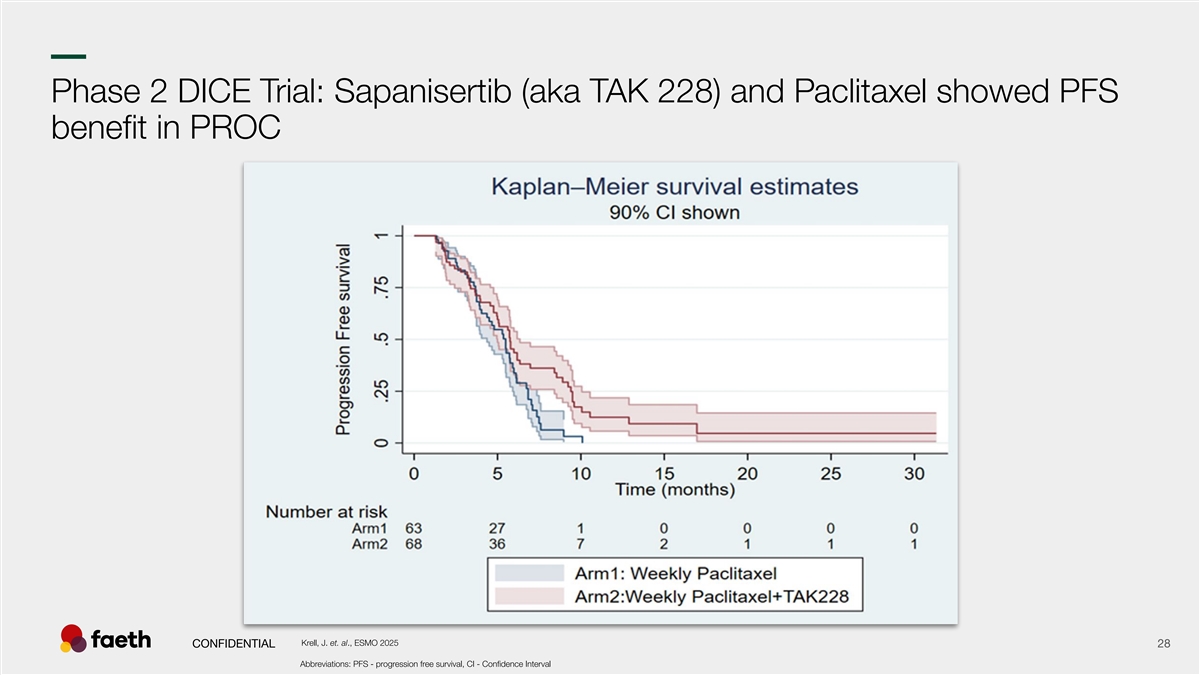
–– Phase 2 DICE Trial: Sapanisertib (aka TAK 228) and Paclitaxel showed PFS benefit in PROC Krell, J. et. al., ESMO 2025 CONFIDENTIAL 28 Abbreviations: PFS - progression free survival, CI - Confidence Interval 28

–– We believe regulatory positioning supports accelerated path to registrational trial Precedent supports a PI3K/mTOR multi-node strategy Combination Dosing 1✔ ● Recent Gedatolisib Ph3 success reinforces the class and mechanism Precedent Combination acceptable for HV studies Clin Pharm Requirements ✔ Defined ● FDA has previously allowed dosing of serabelisib in healthy volunteers providing robust PK/BA data alongside extensive PK/BA data available Safety Database Supports 2 from historical sapanisertib clinical studies ✔ Advancement Clin Pharm gaps are well-defined and addressable ✔ CMC Program on Track Strong safety foundation ● More than 240 subjects exposed; no Hy’s law cases, minimal bilirubin Path to Registrational Trial ✔ elevations, predictable toxicity on intermittent dosing to date Additional contribution of components work may be needed Contribution of Components but could be accelerated with capital 1 Hurvitz, S. et. al., (2025) ESMO Presentation 2 CONFIDENTIAL 29 Patel et. al., 2019, Clin Pharmacol Drug Dev Jul;8(5):637-646., Voss et al. 2020 British Journal of Cancer Nov;123(11):1590-1598 Abbreviations: PK/BA - Pharmacokinetics / Bioavailability
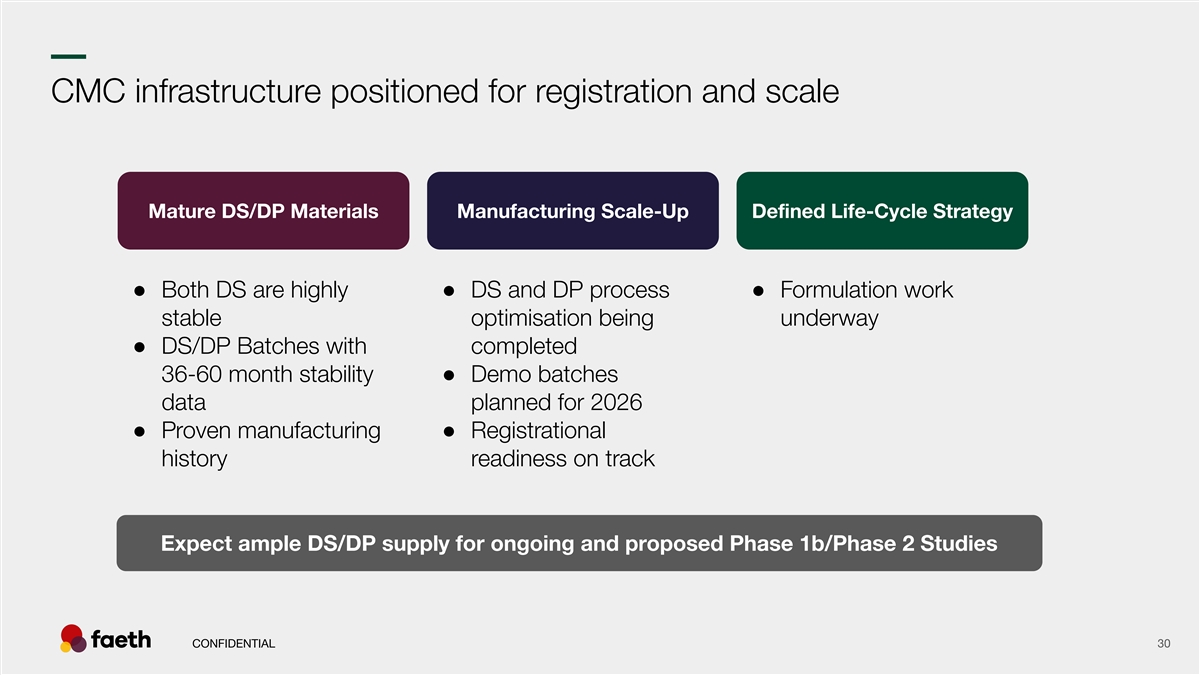
–– CMC infrastructure positioned for registration and scale Mature DS/DP Materials Manufacturing Scale-Up Defined Life-Cycle Strategy ● Both DS are highly ● DS and DP process ● Formulation work stable optimisation being underway ● DS/DP Batches with completed 36-60 month stability ● Demo batches data planned for 2026 ● Proven manufacturing ● Registrational history readiness on track Expect ample DS/DP supply for ongoing and proposed Phase 1b/Phase 2 Studies CONFIDENTIAL 30
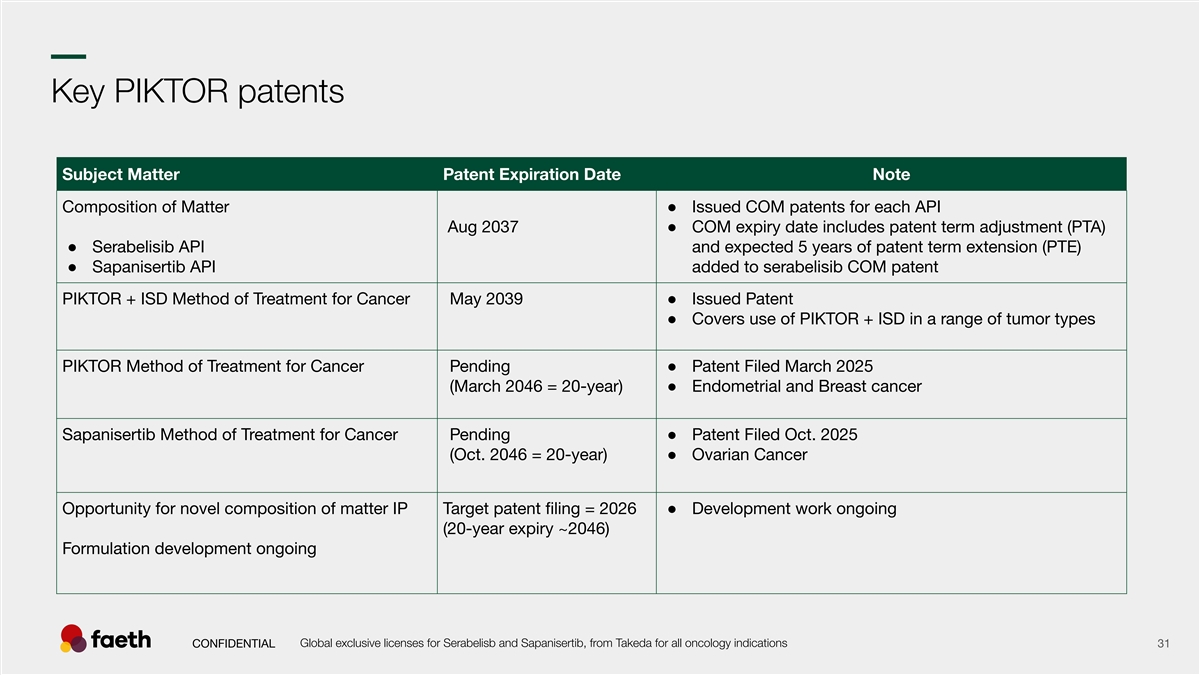
–– Key PIKTOR patents Subject Matter Patent Expiration Date Note Composition of Matter ● Issued COM patents for each API Aug 2037● COM expiry date includes patent term adjustment (PTA) ● Serabelisib API and expected 5 years of patent term extension (PTE) ● Sapanisertib API added to serabelisib COM patent PIKTOR + ISD Method of Treatment for Cancer May 2039● Issued Patent ● Covers use of PIKTOR + ISD in a range of tumor types PIKTOR Method of Treatment for Cancer Pending ● Patent Filed March 2025 (March 2046 = 20-year)● Endometrial and Breast cancer Sapanisertib Method of Treatment for Cancer Pending ● Patent Filed Oct. 2025 (Oct. 2046 = 20-year)● Ovarian Cancer Opportunity for novel composition of matter IP Target patent filing = 2026 ● Development work ongoing (20-year expiry ~2046) Formulation development ongoing Global exclusive licenses for Serabelisb and Sapanisertib, from Takeda for all oncology indications CONFIDENTIAL 31
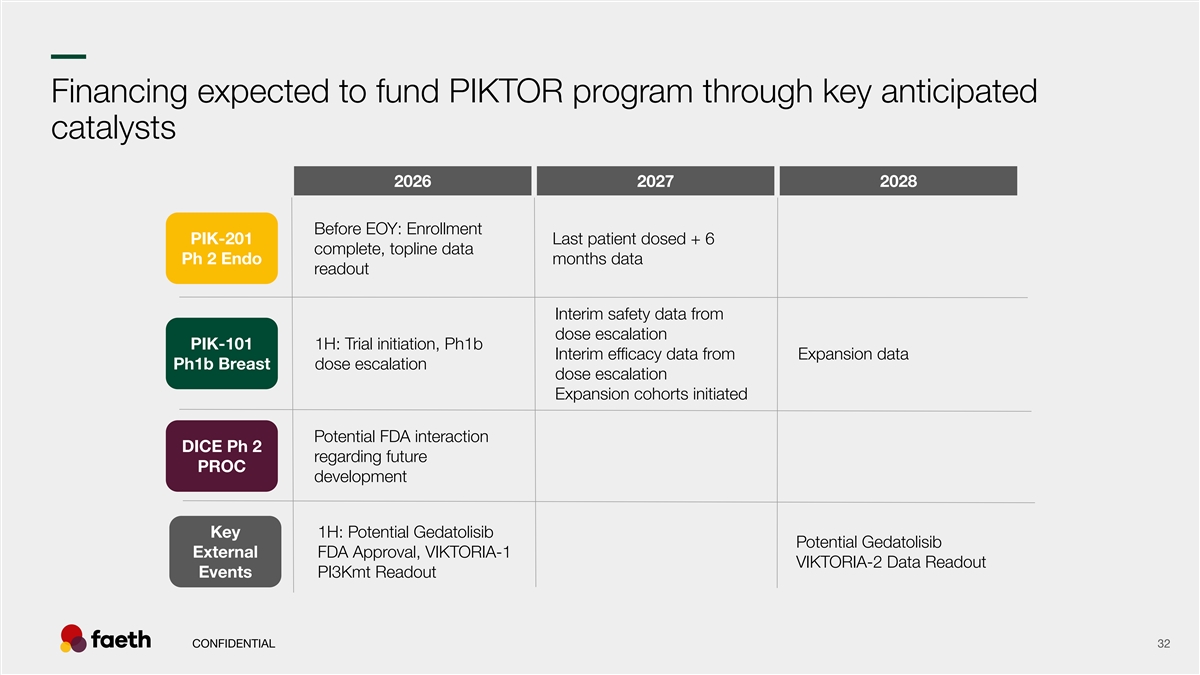
–– Financing expected to fund PIKTOR program through key anticipated catalysts 2026 2027 2028 Before EOY: Enrollment PIK-201 Last patient dosed + 6 complete, topline data Ph 2 Endo months data readout Interim safety data from dose escalation PIK-101 1H: Trial initiation, Ph1b Interim efficacy data from Expansion data Ph1b Breast dose escalation dose escalation Expansion cohorts initiated Potential FDA interaction DICE Ph 2 regarding future PROC development Key 1H: Potential Gedatolisib Potential Gedatolisib External FDA Approval, VIKTORIA-1 VIKTORIA-2 Data Readout Events PI3Kmt Readout CONFIDENTIAL 32

–– Faeth team is well rounded with deep experience in drug development Management Team Key Faeth Employees Board Bob Holmen, JD Christopher Gerry, JD Tom Ricks, MBA Board Chair President & General Counsel Oliver Maddocks, MPharm PhD Chief Scientific Officer Anand Parikh, JD Kristian Humer, MBA Anand Parikh, JD Chief Operating Officer Debbie Chirnomas, MD MPH Chief Medical Officer Phil Donenberg Christopher Gerry, JD Josiah Craver, CPA SVP, Finance CONFIDENTIAL 33

–– Thank you 34 CONFIDENTIAL