

Corporate Presentation January 2026 Modifying Disease Delaying Progression Transforming Treatment Company Logo © 2026 SAB BIOTHERAPEUTICS, INC.

The material in this presentation has been prepared by SAB Biotherapeutics, Inc., doing business as SAB BIO (“SAB”) and is general background information about SAB’s activities current as of the date of this presentation. This information is given in summary form and is not intended to be complete. Information in this presentation, including financial forecasts, should not be considered advice or a recommendation to investors or potential investors in relation to holding, purchasing, or selling securities or other financial products or instruments and does not take into account any particular investment objectives, financial situation or needs. This presentation is for informational purposes only and does not constitute an offer to sell or a solicitation of an offer to buy any securities. This presentation may contain forward-looking statements including statements regarding our intent, belief, or current expectations with respect to SAB’s businesses and operations, market conditions, the exercise of outstanding warrants for cash, results of operations and financial condition, capital adequacy, specific provisions, and risk management practices. Readers are cautioned not to place undue reliance on these forward-looking statements. SAB does not undertake any obligation to update any information herein for any reason or to publicly release the result of any revisions to these forward-looking statements to reflect events or circumstances after the date hereof to reflect the occurrence of unanticipated events unless required by law. While due care has been used in the preparation of forecast information, actual results may vary in a materially positive or negative manner and the presentation may contain errors or omissions. Forecasts and hypothetical examples are subject to uncertainty and contingencies outside SAB’s control. Past performance is not a reliable indication of future performance. The forward-looking statements contained or implied in this presentation are subject to other risks and uncertainties, including those discussed under the heading "Risk Factors" in SAB’s most recent Annual Report on Form 10-K with the Securities and Exchange Commission (the “SEC”) and in other filings and reports that SAB makes with the SEC. Unless otherwise specified, information is current at the date hereof. The SAB logo and other trademarks of SAB appearing in this presentation are the property of SAB. All other trademarks, services marks, and trade names in this presentation are the property of their respective owners. Forward-Looking Statements © 2026 SAB BIOTHERAPEUTICS, INC. 2

Transforming Treatment for People Living with Autoimmune Diseases through a Unique Disease-modifying Therapy At SAB BIO, our mission is to dramatically redefine what it means to be diagnosed with Type 1 Diabetes by developing a medicine to change the course of disease, not just treat symptoms Company Logo 3
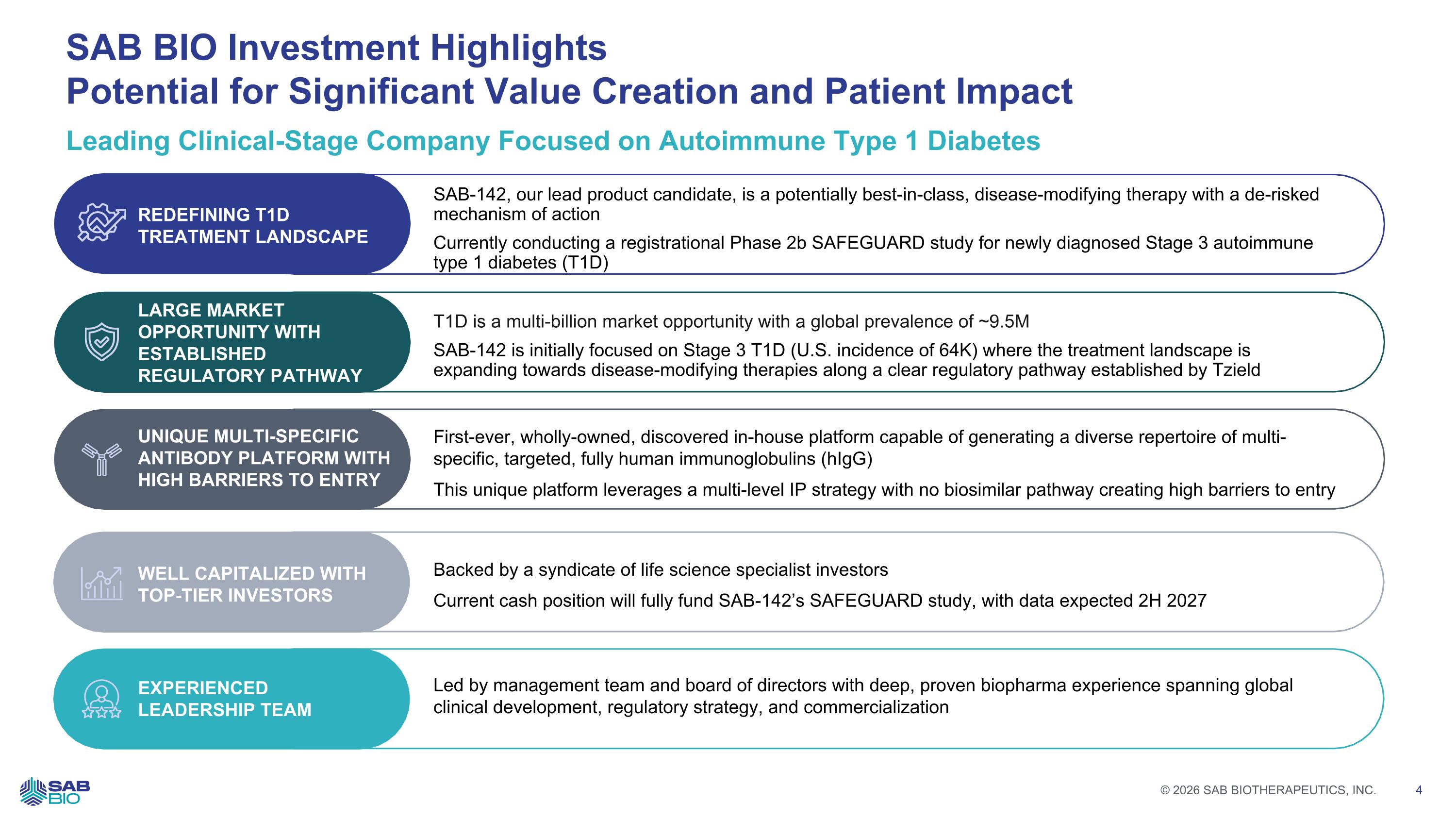
Leading Clinical-Stage Company Focused on Autoimmune Type 1 Diabetes SAB BIO Investment Highlights Potential for Significant Value Creation and Patient Impact REDEFINING T1D TREATMENT LANDSCAPE SAB-142, our lead product candidate, is a potentially best-in-class, disease-modifying therapy with a de-risked mechanism of action Currently conducting a registrational Phase 2b SAFEGUARD study for newly diagnosed Stage 3 autoimmune type 1 diabetes (T1D) LARGE MARKET OPPORTUNITY WITH ESTABLISHED REGULATORY PATHWAY T1D is a multi-billion market opportunity with a global prevalence of ~9.5M SAB-142 is initially focused on Stage 3 T1D (U.S. incidence of 64K) where the treatment landscape is expanding towards disease-modifying therapies along a clear regulatory pathway established by Tzield UNIQUE MULTI-SPECIFIC ANTIBODY PLATFORM WITH HIGH BARRIERS TO ENTRY First-ever, wholly-owned, discovered in-house platform capable of generating a diverse repertoire of multi-specific, targeted, fully human immunoglobulins (hIgG) This unique platform leverages a multi-level IP strategy with no biosimilar pathway creating high barriers to entry WELL CAPITALIZED WITH TOP-TIER INVESTORS Backed by a syndicate of life science specialist investors Current cash position will fully fund SAB-142’s SAFEGUARD study, with data expected 2H 2027 EXPERIENCED LEADERSHIP TEAM Led by management team and board of directors with deep, proven biopharma experience spanning global clinical development, regulatory strategy, and commercialization Company Logo © 2026 SAB BIOTHERAPEUTICS, INC. 4
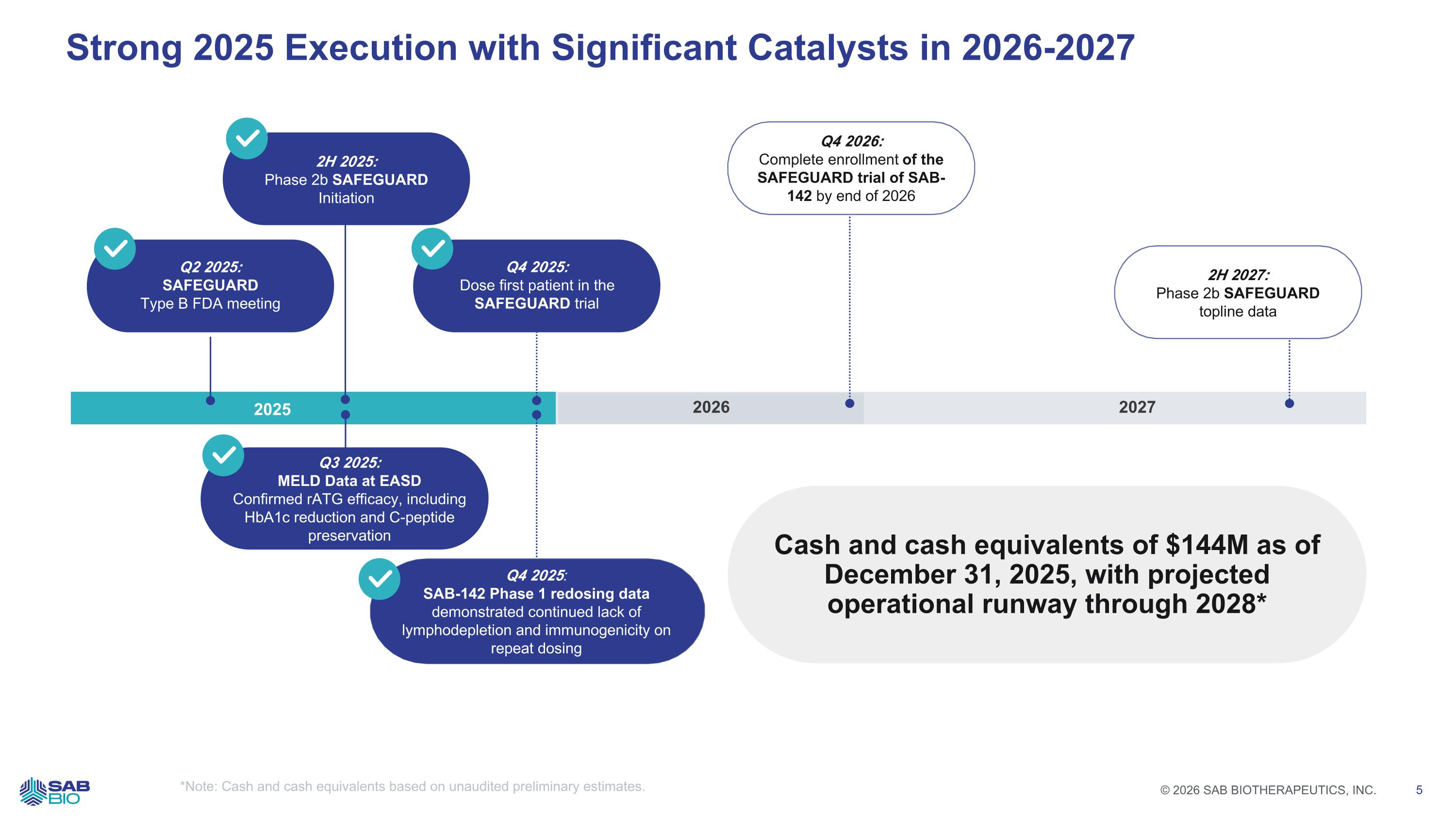
Strong 2025 Execution with Significant Catalysts in 2026-2027 2025 2026 2027 2H 2025: Phase 2b SAFEGUARD Initiation Q2 2025: SAFEGUARD Type B FDA meeting Q3 2025: MELD Data at EASD Confirmed rATG efficacy, including HbA1c reduction and C-peptide preservation Q4 2025: SAB-142 Phase 1 redosing data demonstrated continued lack of lymphodepletion and immunogenicity on repeat dosing 2H 2027: Phase 2b SAFEGUARD topline data Cash and cash equivalents of $144M as of December 31, 2025, with projected operational runway through 2028* Q4 2025: Dose first patient in the SAFEGUARD trial Q4 2026: Complete enrollment of the SAFEGUARD trial of SAB-142 by end of 2026 *Note: Cash and cash equivalents based on unaudited preliminary estimates. Company Logo © 2026 SAB BIOTHERAPEUTICS, INC. 5
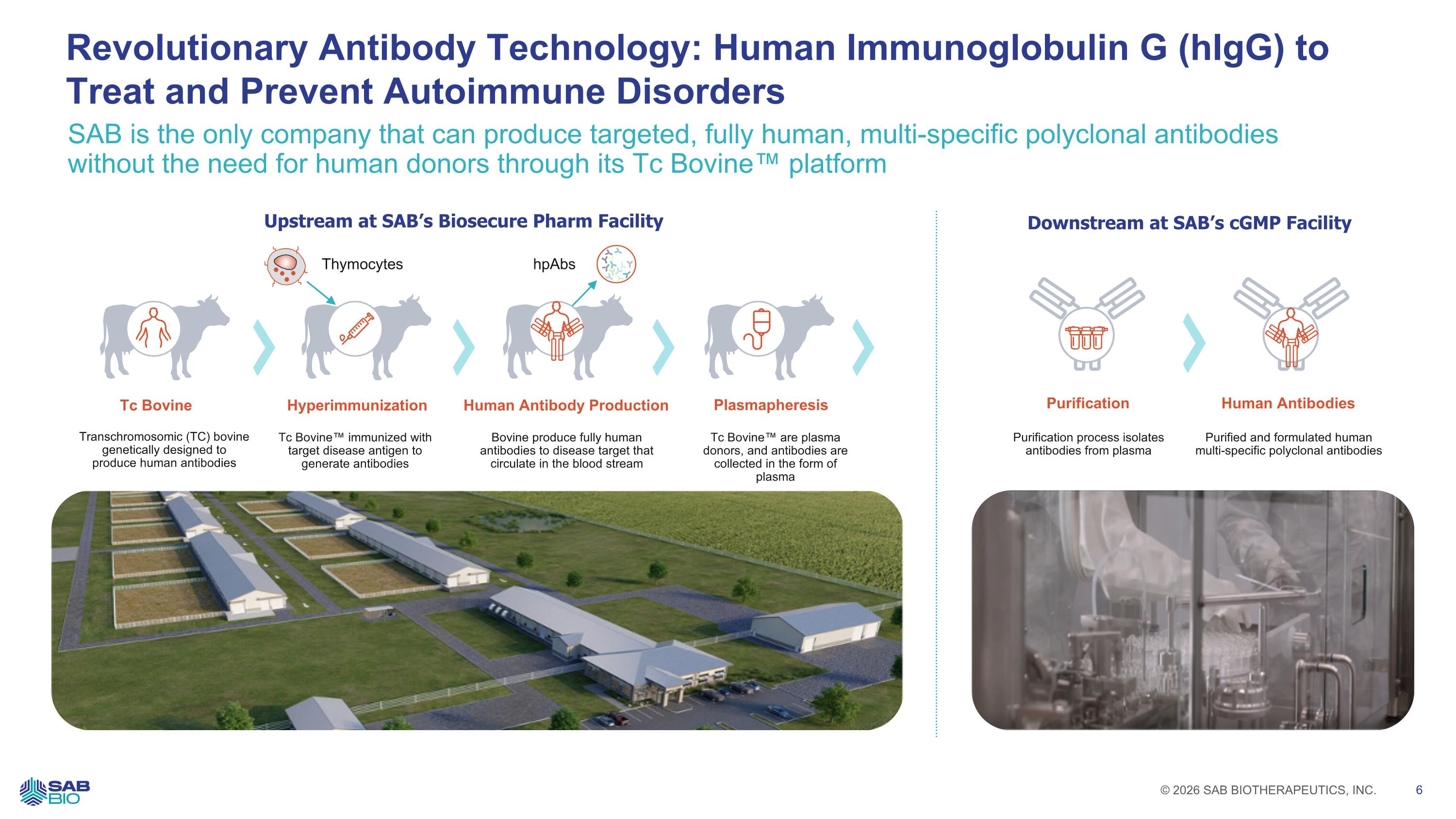
SAB is the only company that can produce targeted, fully human, multi-specific polyclonal antibodies without the need for human donors through its Tc Bovine™ platform Revolutionary Antibody Technology: Human Immunoglobulin G (hIgG) to Treat and Prevent Autoimmune Disorders Upstream at SAB’s Biosecure Pharm Facility Downstream at SAB’s cGMP Facility Thymocytes Tc Bovine Transchromosomic (TC) bovine genetically designed to produce human antibodies Hyperimmunization Tc Bovine™ immunized with target disease antigen to generate antibodies Human Antibody Production Bovine produce fully human antibodies to disease target that circulate in the blood stream Plasmapheresis Tc Bovine™ are plasma donors, and antibodies are collected in the form of plasma hpAbs Purification Purification process isolates antibodies from plasma Human Antibodies Purified and formulated human multi-specific polyclonal antibodies Company Logo © 2026 SAB BIOTHERAPEUTICS, INC. 6
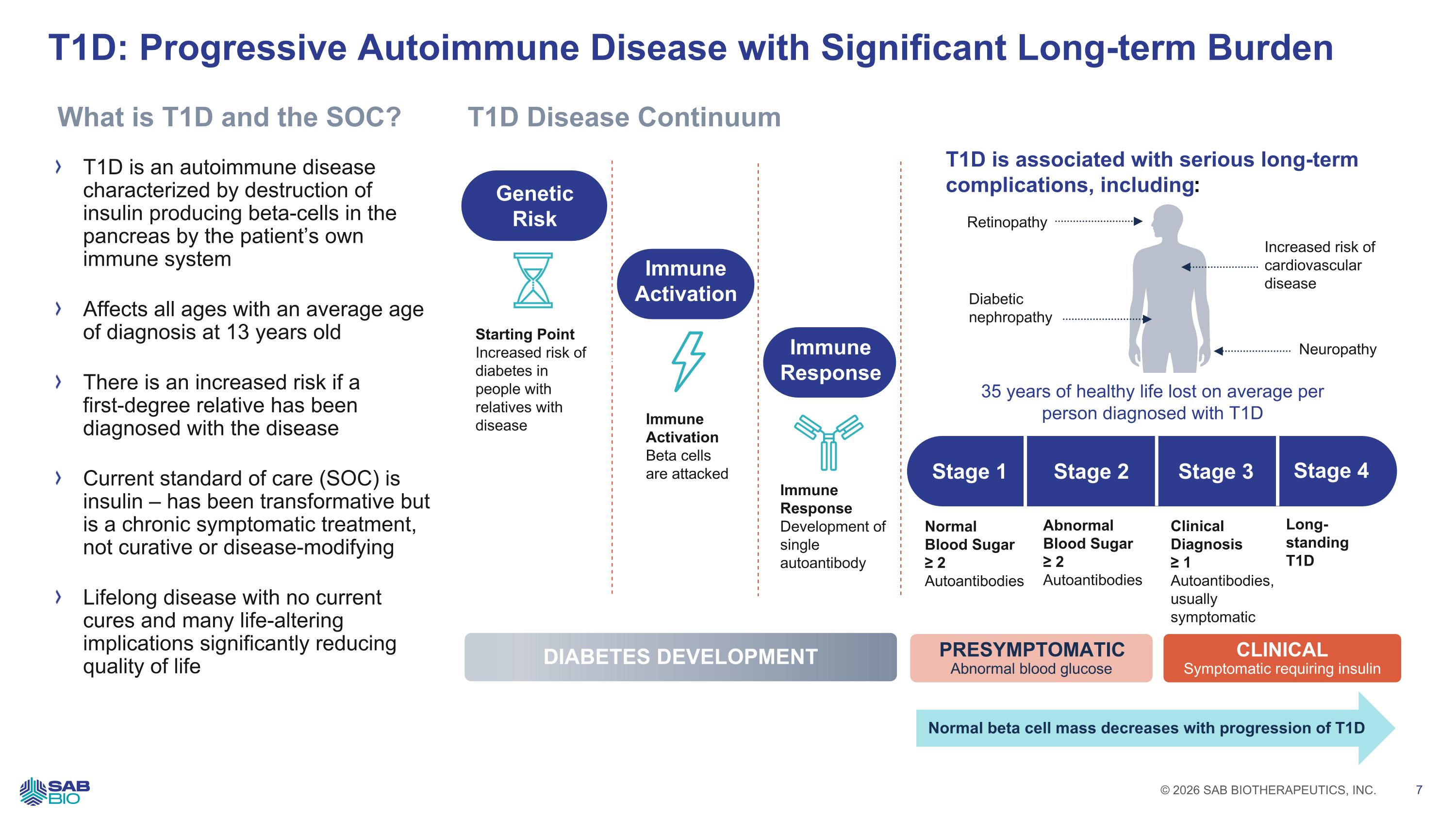
T1D is associated with serious long-term complications, including: T1D: Progressive Autoimmune Disease with Significant Long-term Burden T1D is an autoimmune disease characterized by destruction of insulin producing beta-cells in the pancreas by the patient’s own immune system Affects all ages with an average age of diagnosis at 13 years old There is an increased risk if a first-degree relative has been diagnosed with the disease Current standard of care (SOC) is insulin – has been transformative but is a chronic symptomatic treatment, not curative or disease-modifying Lifelong disease with no current cures and many life-altering implications significantly reducing quality of life 64,000 US Patients Diagnosed Annually2 Genetic Risk Immune Activation Immune Response Starting Point Increased risk of diabetes in people with relatives with disease Immune Activation Beta cells are attacked Immune Response Development of single autoantibody Stage 1 Stage 2 Stage 3 Stage 4 Normal Blood Sugar ≥ 2 Autoantibodies Abnormal Blood Sugar ≥ 2 Autoantibodies Clinical Diagnosis ≥ 1 Autoantibodies, usually symptomatic Long-standing T1D PRESYMPTOMATIC Abnormal blood glucose CLINICAL Symptomatic requiring insulin DIABETES DEVELOPMENT Normal beta cell mass decreases with progression of T1D T1D Disease Continuum What is T1D and the SOC? Retinopathy Diabetic nephropathy 35 years of healthy life lost on average per person diagnosed with T1D Increased risk of cardiovascular disease Neuropathy Company Logo © 2026 SAB BIOTHERAPEUTICS, INC. 7
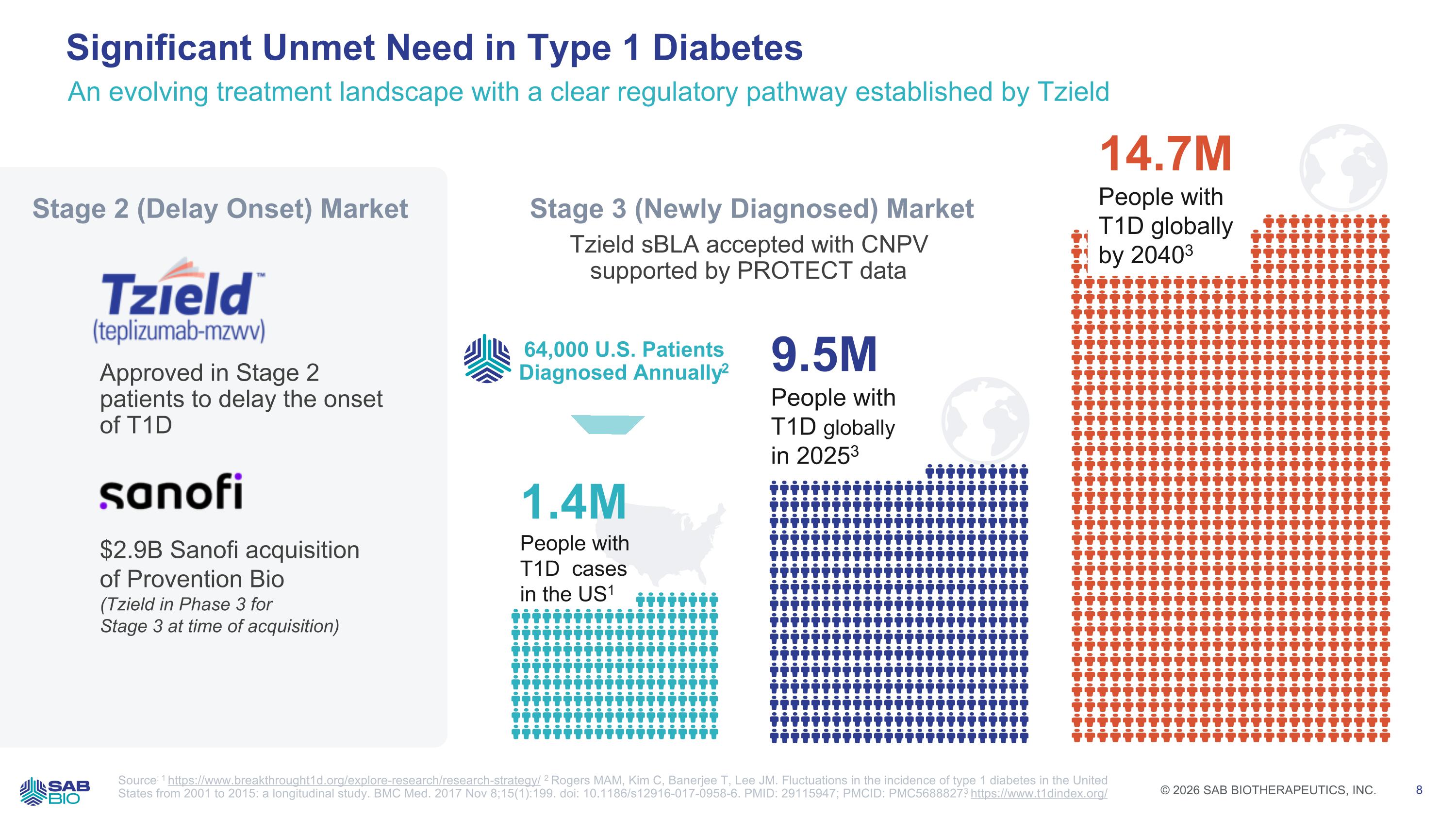
Significant Unmet Need in Type 1 Diabetes Source: 1 https://www.breakthrought1d.org/explore-research/research-strategy/ 2 Rogers MAM, Kim C, Banerjee T, Lee JM. Fluctuations in the incidence of type 1 diabetes in the United States from 2001 to 2015: a longitudinal study. BMC Med. 2017 Nov 8;15(1):199. doi: 10.1186/s12916-017-0958-6. PMID: 29115947; PMCID: PMC5688827. 3 https://www.t1dindex.org/ 9.5M People with T1D globally in 20253 1.4M People with T1D cases in the US1 14.7M People with T1D globally by 20403 64,000 U.S. Patients Diagnosed Annually2 $2.9B Sanofi acquisition of Provention Bio (Tzield in Phase 3 for Stage 3 at time of acquisition) Stage 2 (Delay Onset) Market Approved in Stage 2 patients to delay the onset of T1D Stage 3 (Newly Diagnosed) Market An evolving treatment landscape with a clear regulatory pathway established by Tzield Tzield sBLA accepted with CNPV supported by PROTECT data Company Logo © 2026 SAB BIOTHERAPEUTICS, INC. 8
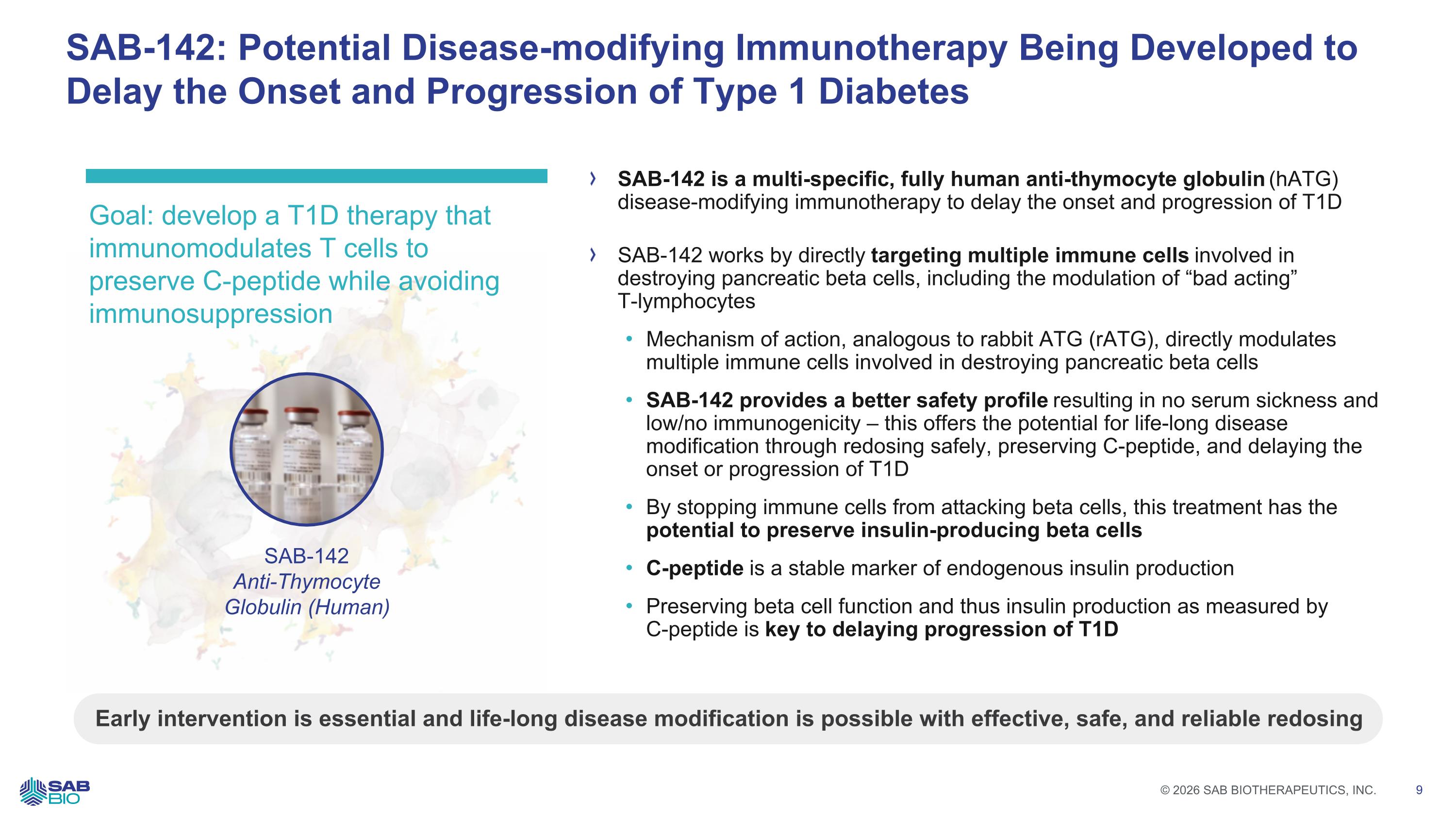
SAB-142: Potential Disease-modifying Immunotherapy Being Developed to Delay the Onset and Progression of Type 1 Diabetes Early intervention is essential and life-long disease modification is possible with effective, safe, and reliable redosing Goal: develop a T1D therapy that immunomodulates T cells to preserve C-peptide while avoiding immunosuppression SAB-142 is a multi-specific, fully human anti-thymocyte globulin (hATG) disease-modifying immunotherapy to delay the onset and progression of T1D SAB-142 works by directly targeting multiple immune cells involved in destroying pancreatic beta cells, including the modulation of “bad acting” T-lymphocytes Mechanism of action, analogous to rabbit ATG (rATG), directly modulates multiple immune cells involved in destroying pancreatic beta cells SAB-142 provides a better safety profile resulting in no serum sickness and low/no immunogenicity – this offers the potential for life-long disease modification through redosing safely, preserving C-peptide, and delaying the onset or progression of T1D By stopping immune cells from attacking beta cells, this treatment has the potential to preserve insulin-producing beta cells C-peptide is a stable marker of endogenous insulin production Preserving beta cell function and thus insulin production as measured by C-peptide is key to delaying progression of T1D SAB-142 Anti-Thymocyte Globulin (Human) Company Logo © 2026 SAB BIOTHERAPEUTICS, INC. 9
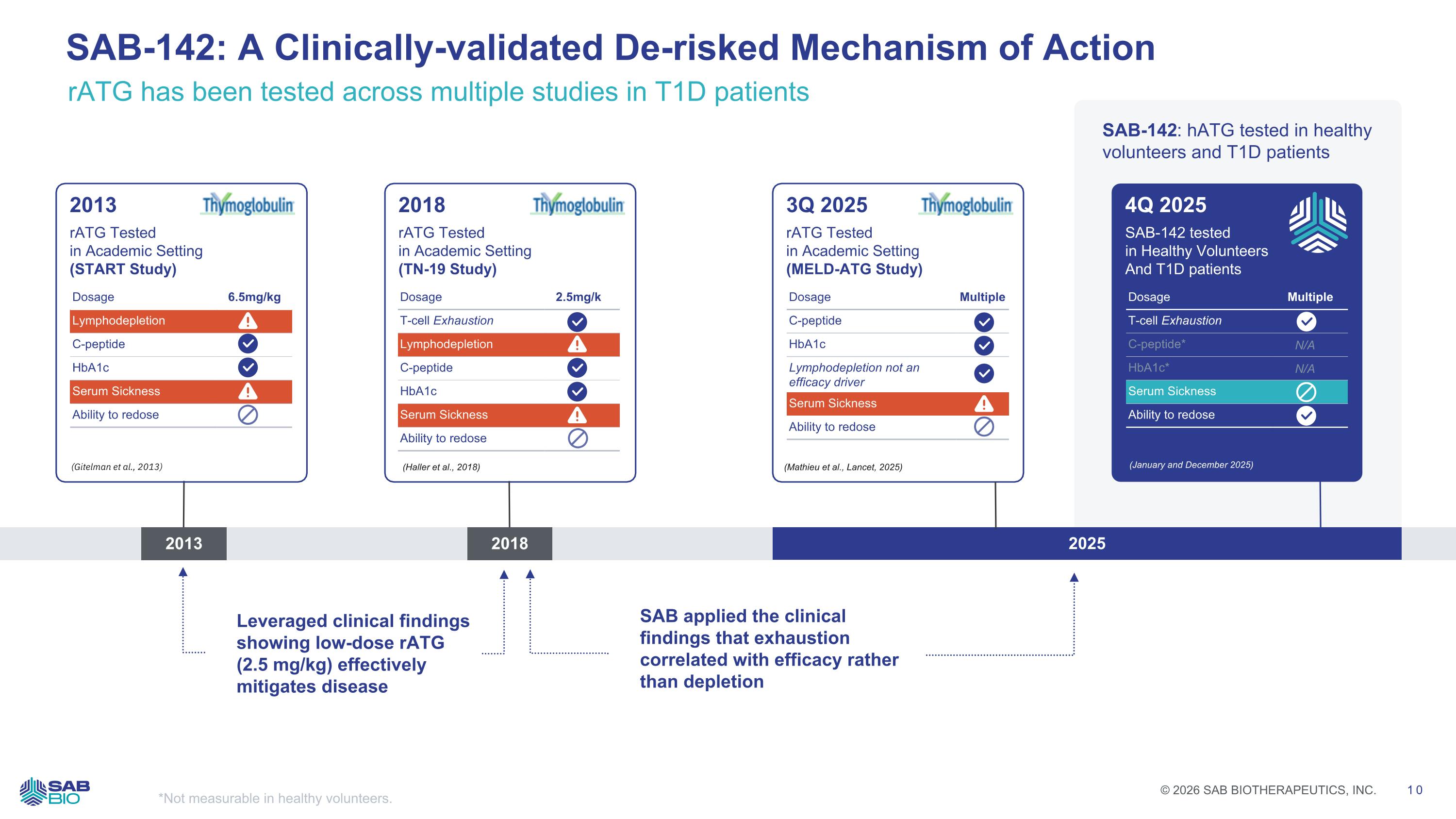
*Not measurable in healthy volunteers. 2013 2018 2025 2013 rATG Tested in Academic Setting (START Study) (Gitelman et al., 2013) Dosage 6.5mg/kg Lymphodepletion C-peptide HbA1c Serum Sickness Ability to redose 2018 rATG Tested in Academic Setting (TN-19 Study) (Haller et al., 2018) Dosage 2.5mg/kg T-cell Exhaustion Lymphodepletion C-peptide HbA1c Serum Sickness Ability to redose 3Q 2025 rATG Tested in Academic Setting (MELD-ATG Study) (Mathieu et al., Lancet, 2025) Dosage Multiple C-peptide HbA1c Lymphodepletion not an efficacy driver Serum Sickness Ability to redose 4Q 2025 SAB-142 tested in Healthy Volunteers And T1D patients (January and December 2025) Dosage Multiple T-cell Exhaustion C-peptide* N/A HbA1c* N/A Serum Sickness Ability to redose Leveraged clinical findings showing low-dose rATG (2.5 mg/kg) effectively mitigates disease SAB applied the clinical findings that exhaustion correlated with efficacy rather than depletion SAB-142: hATG tested in healthy volunteers and T1D patients rATG has been tested across multiple studies in T1D patients SAB-142: A Clinically-validated De-risked Mechanism of Action Company Logo © 2026 SAB BIOTHERAPEUTICS, INC. 10
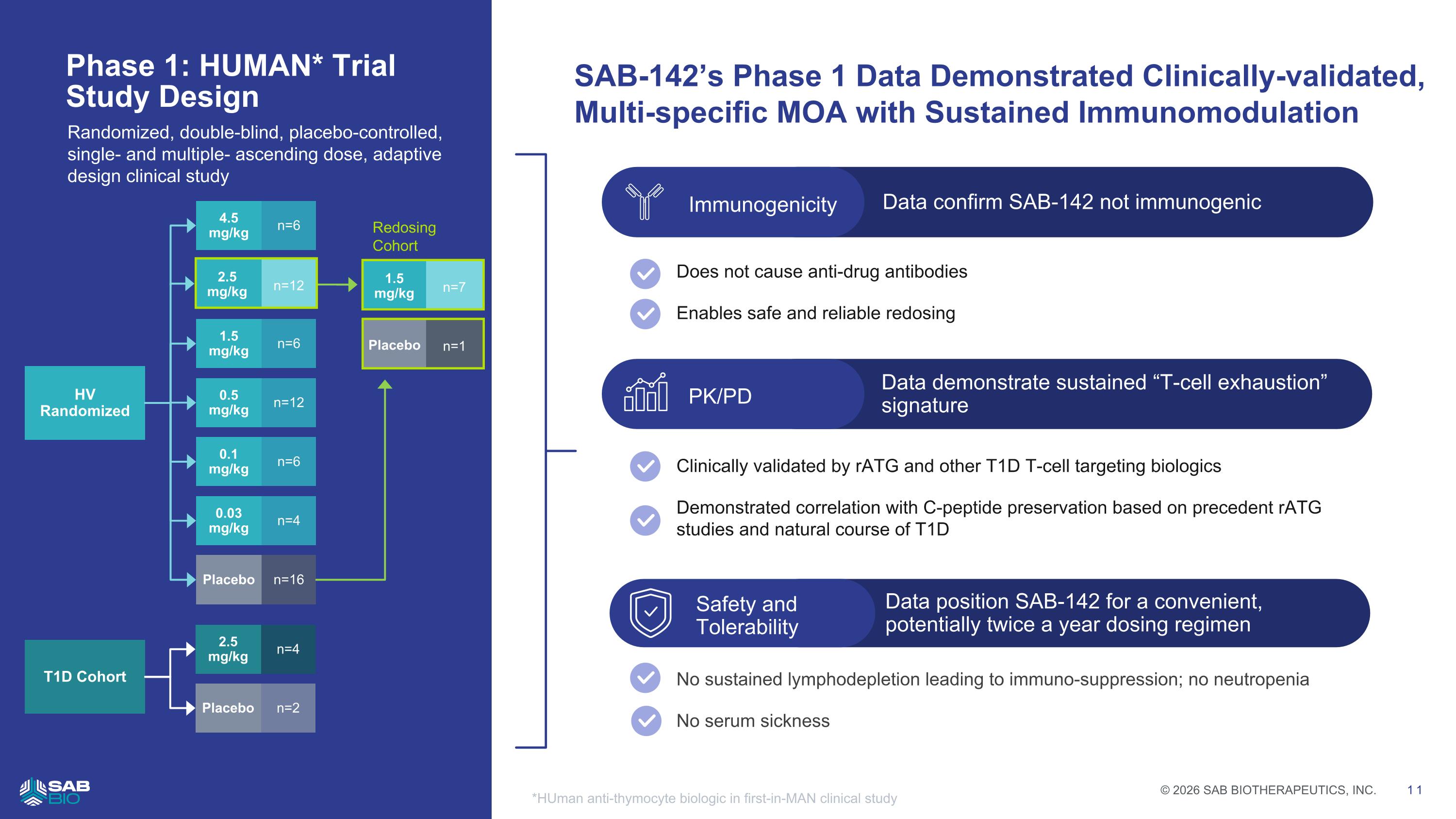
Data position SAB-142 for a convenient, potentially twice a year dosing regimen Safety and Tolerability Data demonstrate sustained “T-cell exhaustion” signature Data confirm SAB-142 not immunogenic Immunogenicity Does not cause anti-drug antibodies Enables safe and reliable redosing Clinically validated by rATG and other T1D T-cell targeting biologics Demonstrated correlation with C-peptide preservation based on precedent rATG studies and natural course of T1D No sustained lymphodepletion leading to immuno-suppression; no neutropenia No serum sickness PK/PD SAB-142’s Phase 1 Data Demonstrated Clinically-validated, Multi-specific MOA with Sustained Immunomodulation Phase 1: HUMAN* Trial Study Design *HUman anti-thymocyte biologic in first-in-MAN clinical study Randomized, double-blind, placebo-controlled, single- and multiple- ascending dose, adaptive design clinical study HV Randomized 1.5 mg/kg n=6 0.5 mg/kg n=12 0.1 mg/kg n=6 0.03 mg/kg n=4 Placebo n=16 T1D Cohort 4.5 mg/kg n=6 2.5 mg/kg n=4 Placebo n=2 Redosing Cohort 1.5 mg/kg n=7 Placebo n=1 2.5 mg/kg n=12 Company Logo © 2026 SAB BIOTHERAPEUTICS, INC. 11
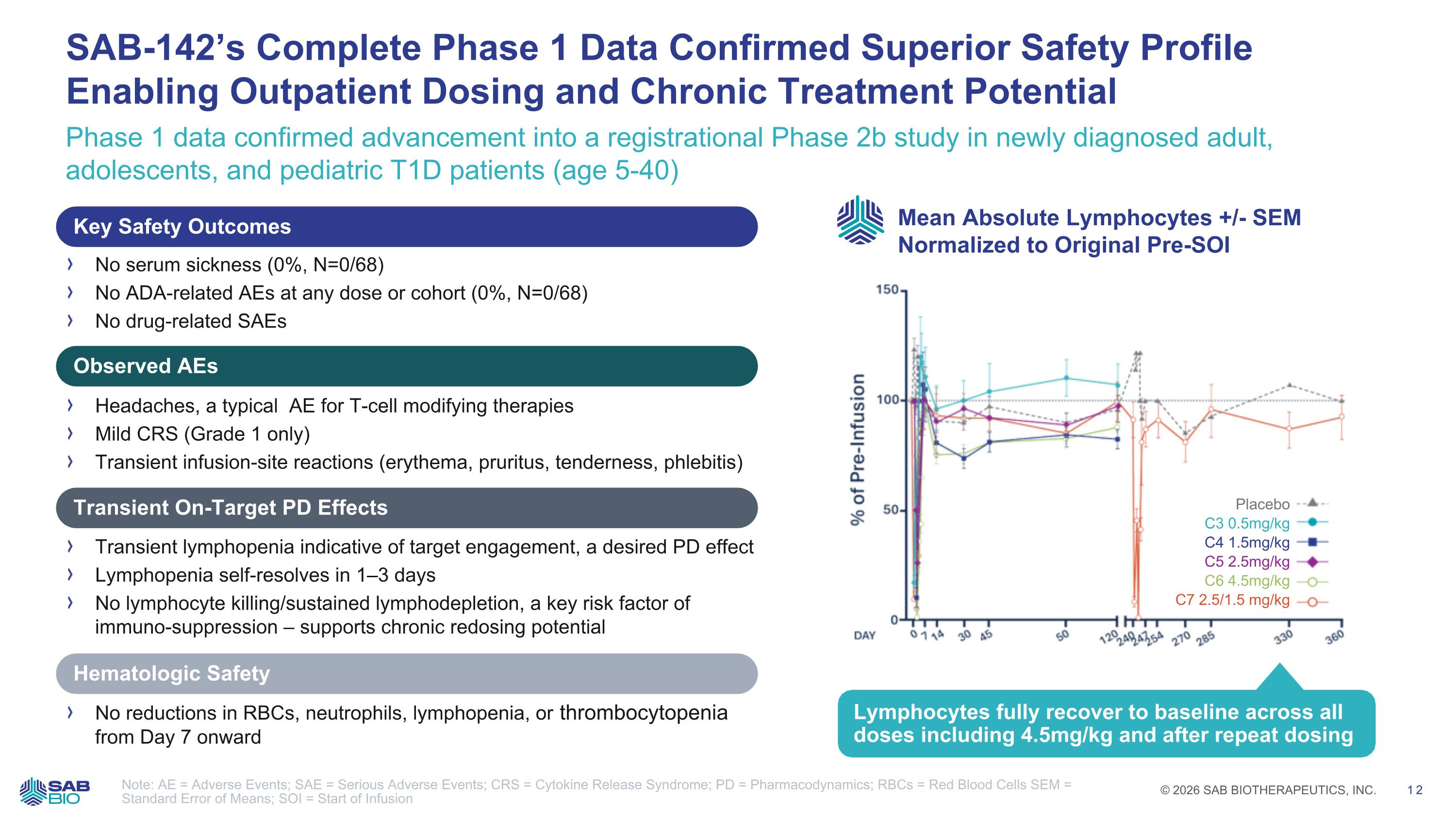
Phase 1 data confirmed advancement into a registrational Phase 2b study in newly diagnosed adult, adolescents, and pediatric T1D patients (age 5-40) SAB-142’s Complete Phase 1 Data Confirmed Superior Safety Profile Enabling Outpatient Dosing and Chronic Treatment Potential Note: AE = Adverse Events; SAE = Serious Adverse Events; CRS = Cytokine Release Syndrome; PD = Pharmacodynamics; RBCs = Red Blood Cells SEM = Standard Error of Means; SOI = Start of Infusion Safety profile enables outpatient dosing and redosing No serum sickness (0%, N=0/68) No ADA-related AEs at any dose or cohort (0%, N=0/68) No drug-related SAEs Headaches, a typical AE for T-cell modifying therapies Mild CRS (Grade 1 only) Transient infusion-site reactions (erythema, pruritus, tenderness, phlebitis) Transient lymphopenia indicative of target engagement, a desired PD effect Lymphopenia self-resolves in 1–3 days No lymphocyte killing/sustained lymphodepletion, a key risk factor of immuno-suppression – supports chronic redosing potential No reductions in RBCs, neutrophils, lymphopenia, or thrombocytopenia from Day 7 onward Key Safety Outcomes Observed AEs Transient On-Target PD Effects Hematologic Safety Mean Absolute Lymphocytes +/- SEM Normalized to Original Pre-SOI Lymphocytes fully recover to baseline across all doses including 4.5mg/kg and after repeat dosing Placebo C3 0.5mg/kg C4 1.5mg/kg C5 2.5mg/kg C6 4.5mg/kg C7 2.5/1.5 mg/kg Company Logo © 2026 SAB BIOTHERAPEUTICS, INC. 12
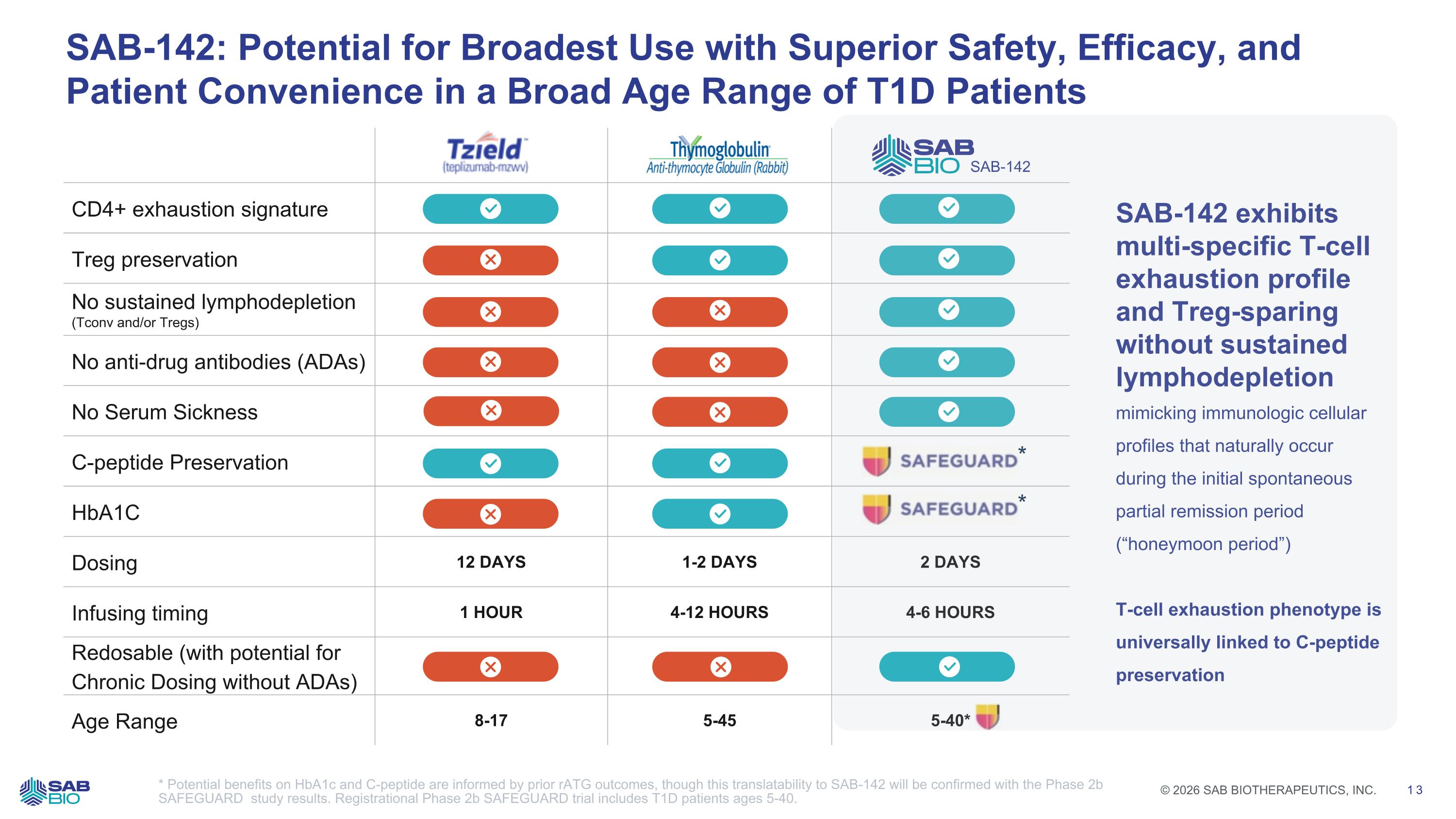
CD4+ exhaustion signature Treg preservation No sustained lymphodepletion (Tconv and/or Tregs) No anti-drug antibodies (ADAs) No Serum Sickness C-peptide Preservation HbA1C Dosing 12 DAYS 1-2 DAYS 2 DAYS Infusing timing 1 HOUR 4-12 HOURS 4-6 HOURS Redosable (with potential for Chronic Dosing without ADAs) Age Range 8-17 5-45 5-40* SAB-142: Potential for Broadest Use with Superior Safety, Efficacy, and Patient Convenience in a Broad Age Range of T1D Patients * Potential benefits on HbA1c and C-peptide are informed by prior rATG outcomes, though this translatability to SAB-142 will be confirmed with the Phase 2b SAFEGUARD study results. Registrational Phase 2b SAFEGUARD trial includes T1D patients ages 5-40. SAB-142 * * SAB-142 exhibits multi-specific T-cell exhaustion profile and Treg-sparing without sustained lymphodepletion mimicking immunologic cellular profiles that naturally occur during the initial spontaneous partial remission period (“honeymoon period”) T-cell exhaustion phenotype is universally linked to C-peptide preservation Company Logo © 2026 SAB BIOTHERAPEUTICS, INC. 13
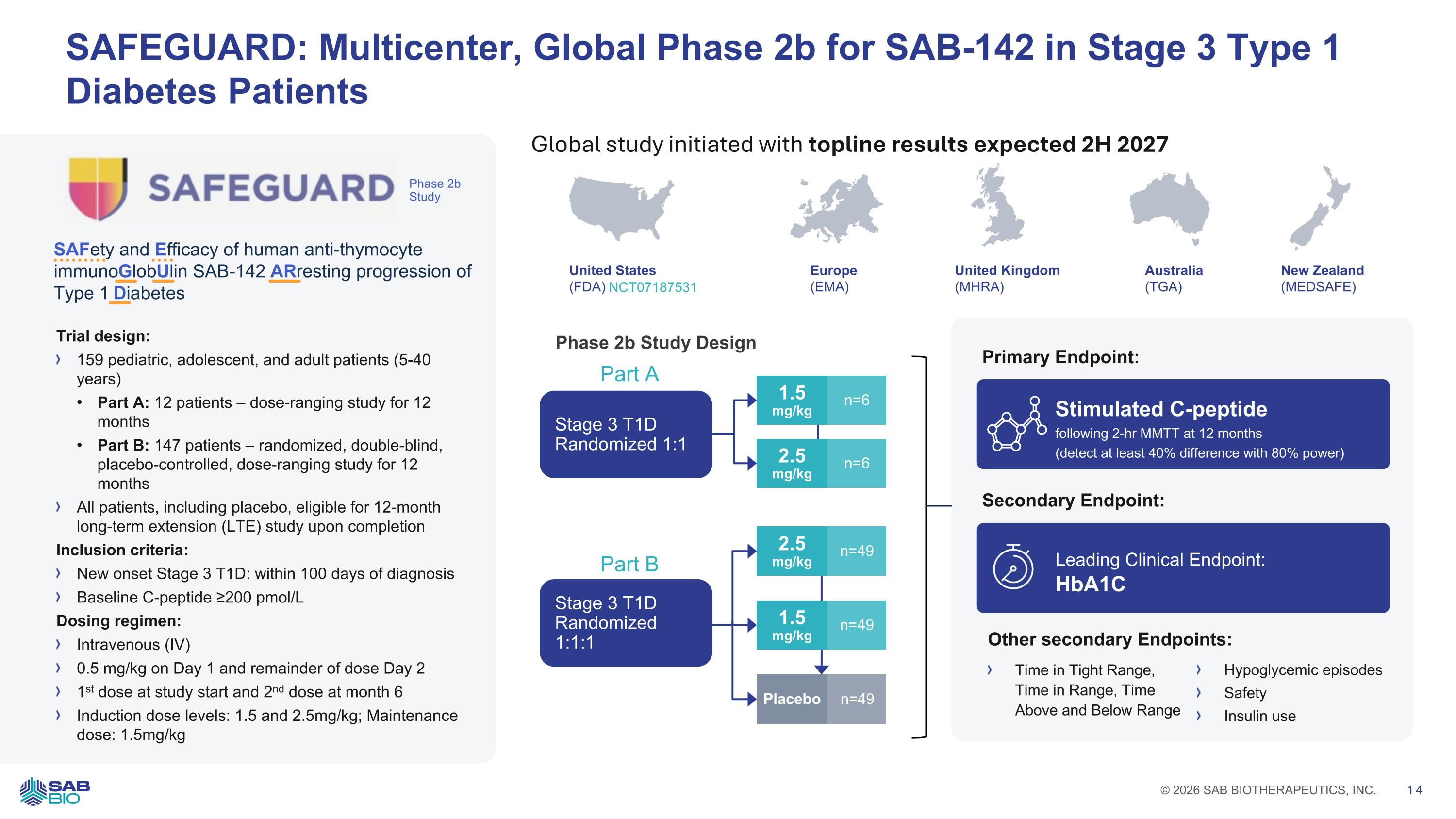
SAFEGUARD: Multicenter, Global Phase 2b for SAB-142 in Stage 3 Type 1 Diabetes Patients Trial design: 159 pediatric, adolescent, and adult patients (5-40 years) Part A: 12 patients – dose-ranging study for 12 months Part B: 147 patients – randomized, double-blind, placebo-controlled, dose-ranging study for 12 months All patients, including placebo, eligible for 12-month long-term extension (LTE) study upon completion Inclusion criteria: New onset Stage 3 T1D: within 100 days of diagnosis Baseline C-peptide ≥200 pmol/L Dosing regimen: Intravenous (IV) 0.5 mg/kg on Day 1 and remainder of dose Day 2 1st dose at study start and 2nd dose at month 6 Induction dose levels: 1.5 and 2.5mg/kg; Maintenance dose: 1.5mg/kg Phase 2b Study SAFety and Efficacy of human anti-thymocyte immunoGlobUlin SAB-142 ARresting progression of Type 1 Diabetes Phase 2b Study Design 1.5 mg/kg n=49 Stimulated C-peptide following 2-hr MMTT at 12 months (detect at least 40% difference with 80% power) Leading Clinical Endpoint: HbA1C Primary Endpoint: Secondary Endpoint: Time in Tight Range, Time in Range, Time Above and Below Range Hypoglycemic episodes Safety Insulin use Other secondary Endpoints: Part A Part B 2.5 mg/kg n=6 1.5 mg/kg n=6 2.5 mg/kg n=49 Placebo n=49 United States (FDA) United Kingdom (MHRA) Europe (EMA) Australia (TGA) New Zealand (MEDSAFE) NCT07187531 Global study initiated with topline results expected 2H 2027 Stage 3 T1D Randomized 1:1 Stage 3 T1D Randomized 1:1:1 Company Logo © 2026 SAB BIOTHERAPEUTICS, INC. 14
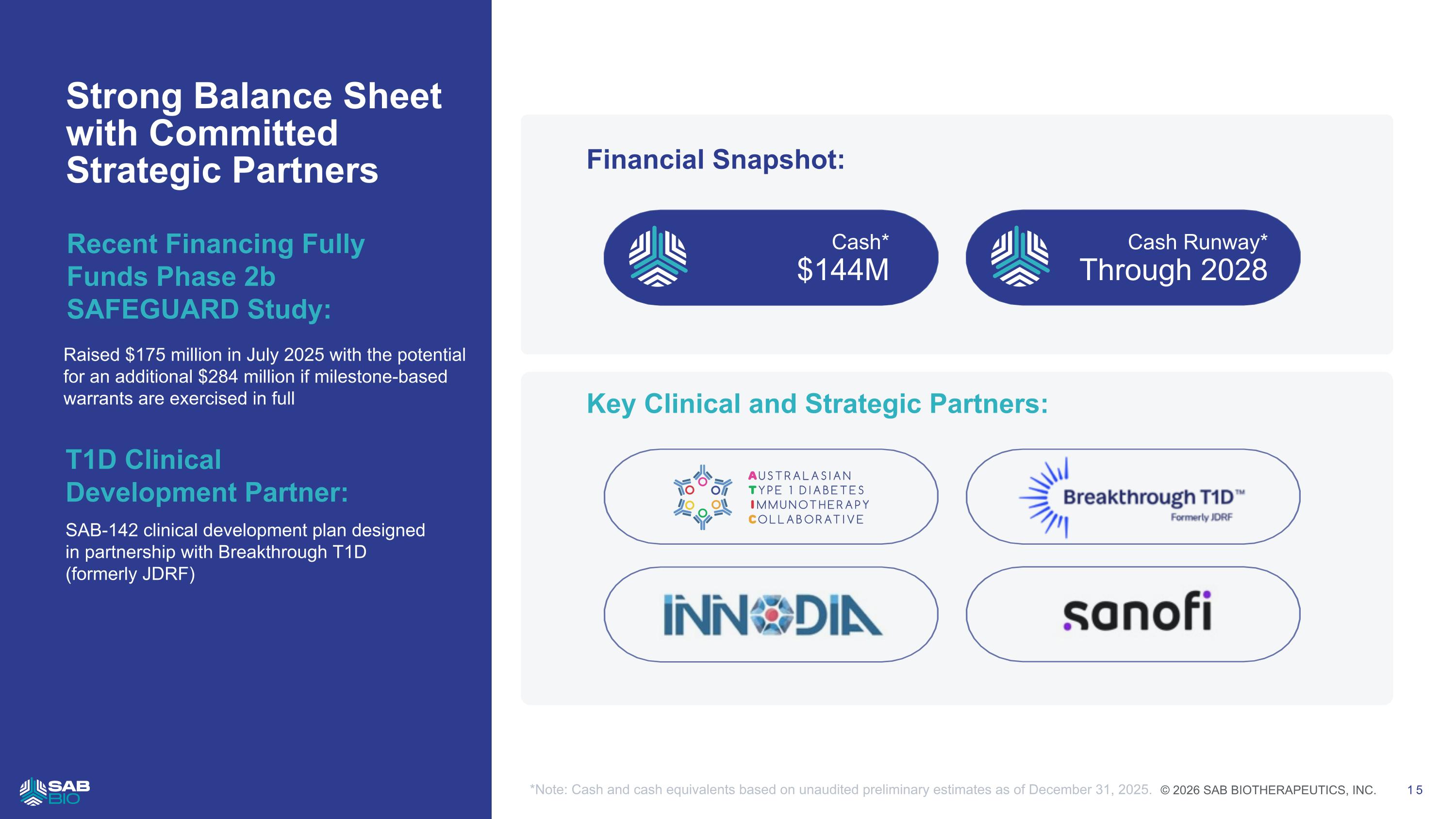
Strong Balance Sheet with Committed Strategic Partners Raised $175 million in July 2025 with the potential for an additional $284 million if milestone-based warrants are exercised in full Financial Snapshot: Cash* $144M Cash Runway* Through 2028 T1D Clinical Development Partner: SAB-142 clinical development plan designed in partnership with Breakthrough T1D (formerly JDRF) Recent Financing Fully Funds Phase 2b SAFEGUARD Study: Key Clinical and Strategic Partners: *Note: Cash and cash equivalents based on unaudited preliminary estimates as of December 31, 2025. Company Logo © 2026 SAB BIOTHERAPEUTICS, INC. 15
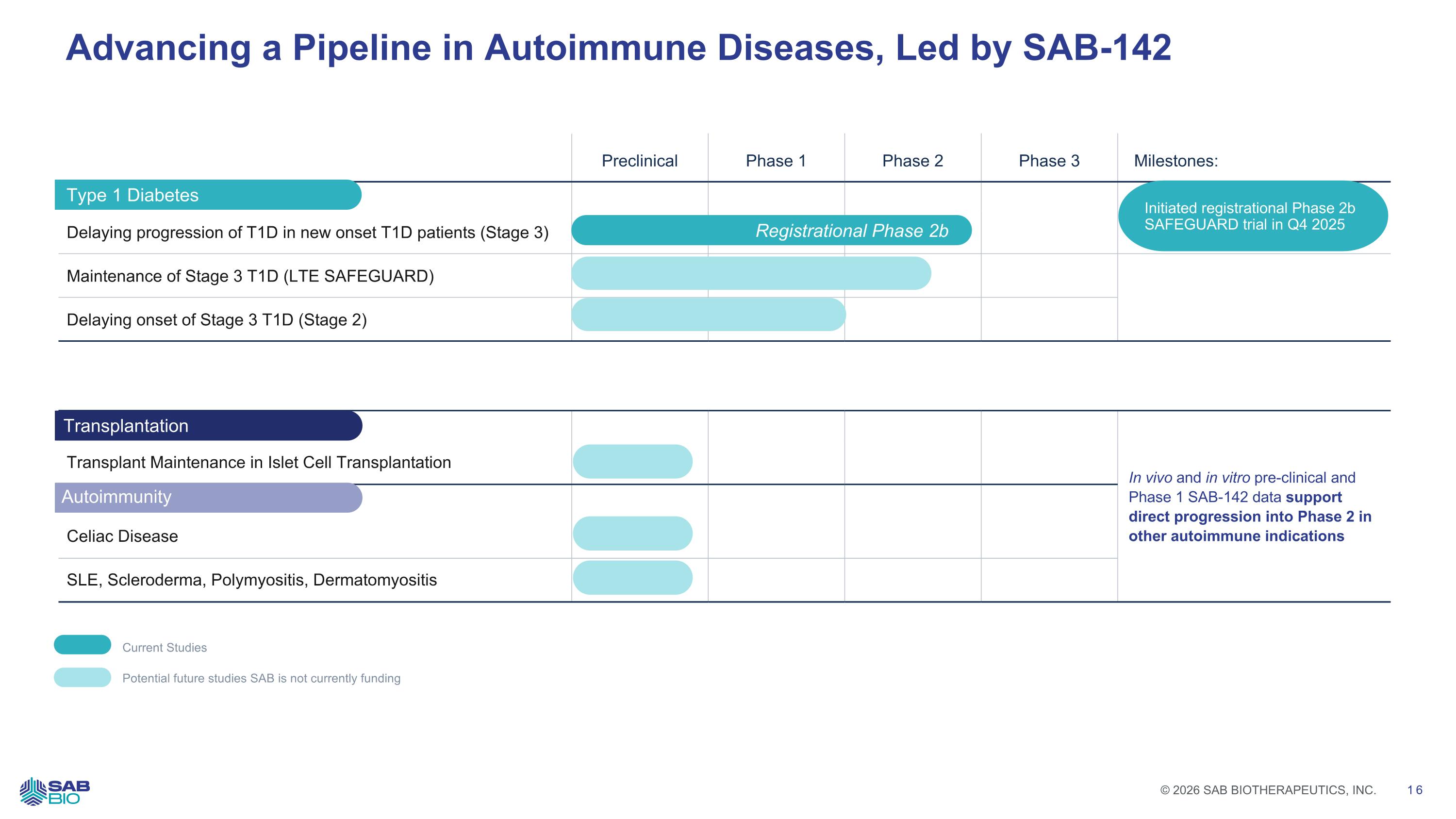
Preclinical Phase 1 Phase 2 Phase 3 Milestones: Delaying progression of T1D in new onset T1D patients (Stage 3) Maintenance of Stage 3 T1D (LTE SAFEGUARD) Delaying onset of Stage 3 T1D (Stage 2) Advancing a Pipeline in Autoimmune Diseases, Led by SAB-142 In vivo and in vitro pre-clinical and Phase 1 SAB-142 data support direct progression into Phase 2 in other autoimmune indications Transplant Maintenance in Islet Cell Transplantation Celiac Disease SLE, Scleroderma, Polymyositis, Dermatomyositis Current Studies Potential future studies SAB is not currently funding Type 1 Diabetes Transplantation Autoimmunity Registrational Phase 2b Initiated registrational Phase 2b SAFEGUARD trial in Q4 2025 Company Logo © 2026 SAB BIOTHERAPEUTICS, INC. 16
Leading Clinical-Stage Company Focused on Autoimmune Type 1 Diabetes SAB BIO Investment Highlights Potential for Significant Value Creation and Patient Impact REDEFINING T1D TREATMENT LANDSCAPE SAB-142, our lead product candidate, is a potentially best-in-class, disease-modifying therapy with a de-risked mechanism of action Currently conducting a registrational Phase 2b SAFEGUARD study for newly diagnosed Stage 3 autoimmune type 1 diabetes (T1D) LARGE MARKET OPPORTUNITY WITH ESTABLISHED REGULATORY PATHWAY T1D is a multi-billion market opportunity with a global prevalence of ~9.5M SAB-142 is initially focused on Stage 3 T1D (U.S. incidence of 64K) where the treatment landscape is expanding towards disease-modifying therapies along a clear regulatory pathway established by Tzield UNIQUE MULTI-SPECIFIC ANTIBODY PLATFORM WITH HIGH BARRIERS TO ENTRY First-ever, wholly-owned, discovered in-house platform capable of generating a diverse repertoire of multi-specific, targeted, fully human immunoglobulins (hIgG) This unique platform leverages a multi-level IP strategy with no biosimilar pathway creating high barriers to entry WELL CAPITALIZED WITH TOP-TIER INVESTORS Backed by a syndicate of life science specialist investors Current cash position will fully fund SAB-142’s SAFEGUARD study, with data expected 2H 2027 EXPERIENCED LEADERSHIP TEAM Led by management team and board of directors with deep, proven biopharma experience spanning global clinical development, regulatory strategy, and commercialization Company Logo © 2026 SAB BIOTHERAPEUTICS, INC. 17

Modifying Disease Delaying Progression Transforming Treatment THANK YOU Company Logo © 2026 SAB BIOTHERAPEUTICS, INC.

Appendix
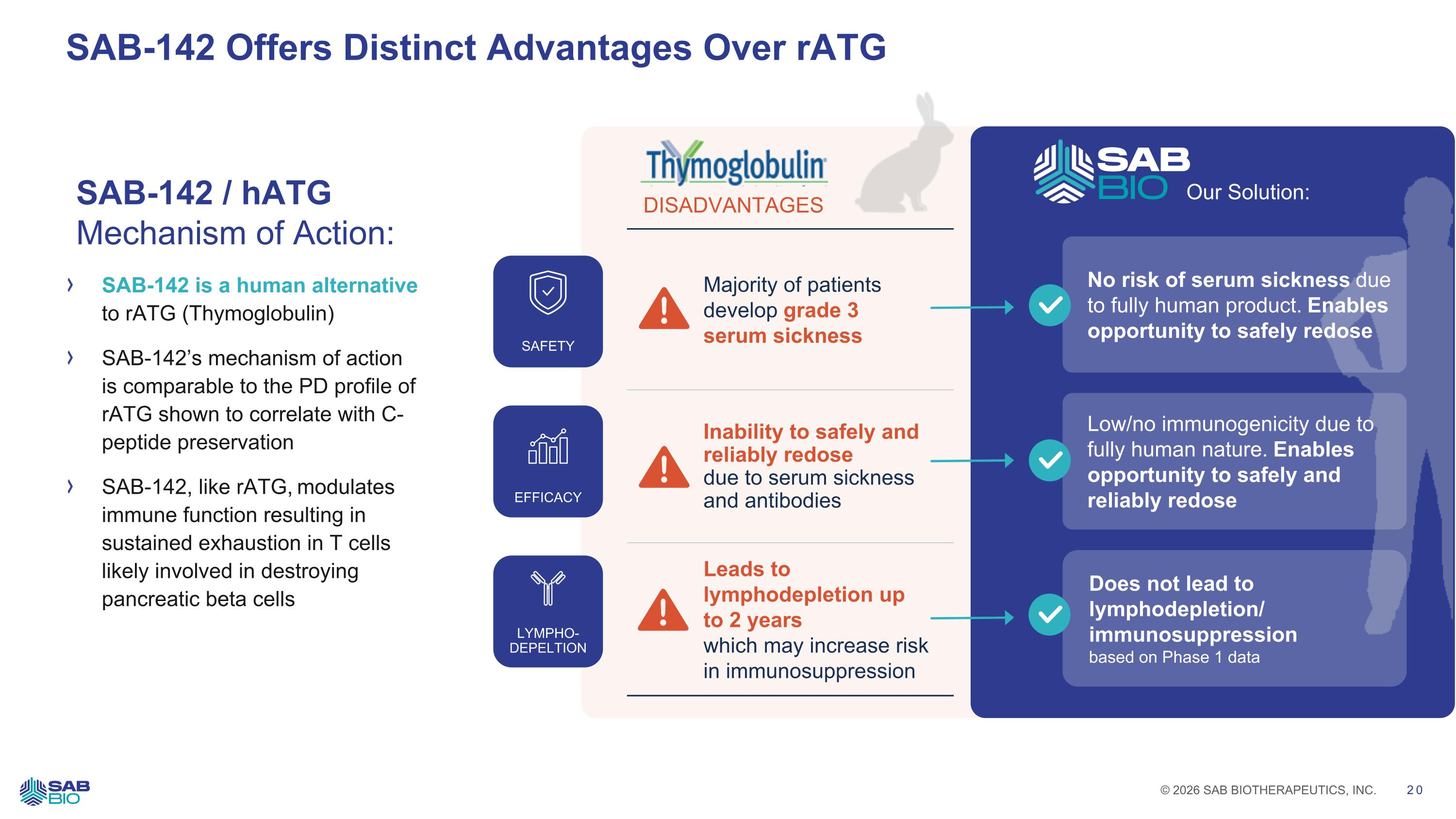
SAB-142 Offers Distinct Advantages Over rATG DISADVANTAGES Majority of patients develop grade 3 serum sickness Inability to safely and reliably redose due to serum sickness and antibodies Leads to lymphodepletion up to 2 years which may increase risk in immunosuppression Low/no immunogenicity due to fully human nature. Enables opportunity to safely and reliably redose Does not lead to lymphodepletion/ immunosuppression based on Phase 1 data Our Solution: SAB-142 is a human alternative to rATG (Thymoglobulin) SAB-142’s mechanism of action is comparable to the PD profile of rATG shown to correlate with C-peptide preservation SAB-142, like rATG, modulates immune function resulting in sustained exhaustion in T cells likely involved in destroying pancreatic beta cells SAB-142 / hATG Mechanism of Action: No risk of serum sickness due to fully human product. Enables opportunity to safely redose SAFETY EFFICACY LYMPHO- DEPELTION Company Logo © 2026 SAB BIOTHERAPEUTICS, INC. 20
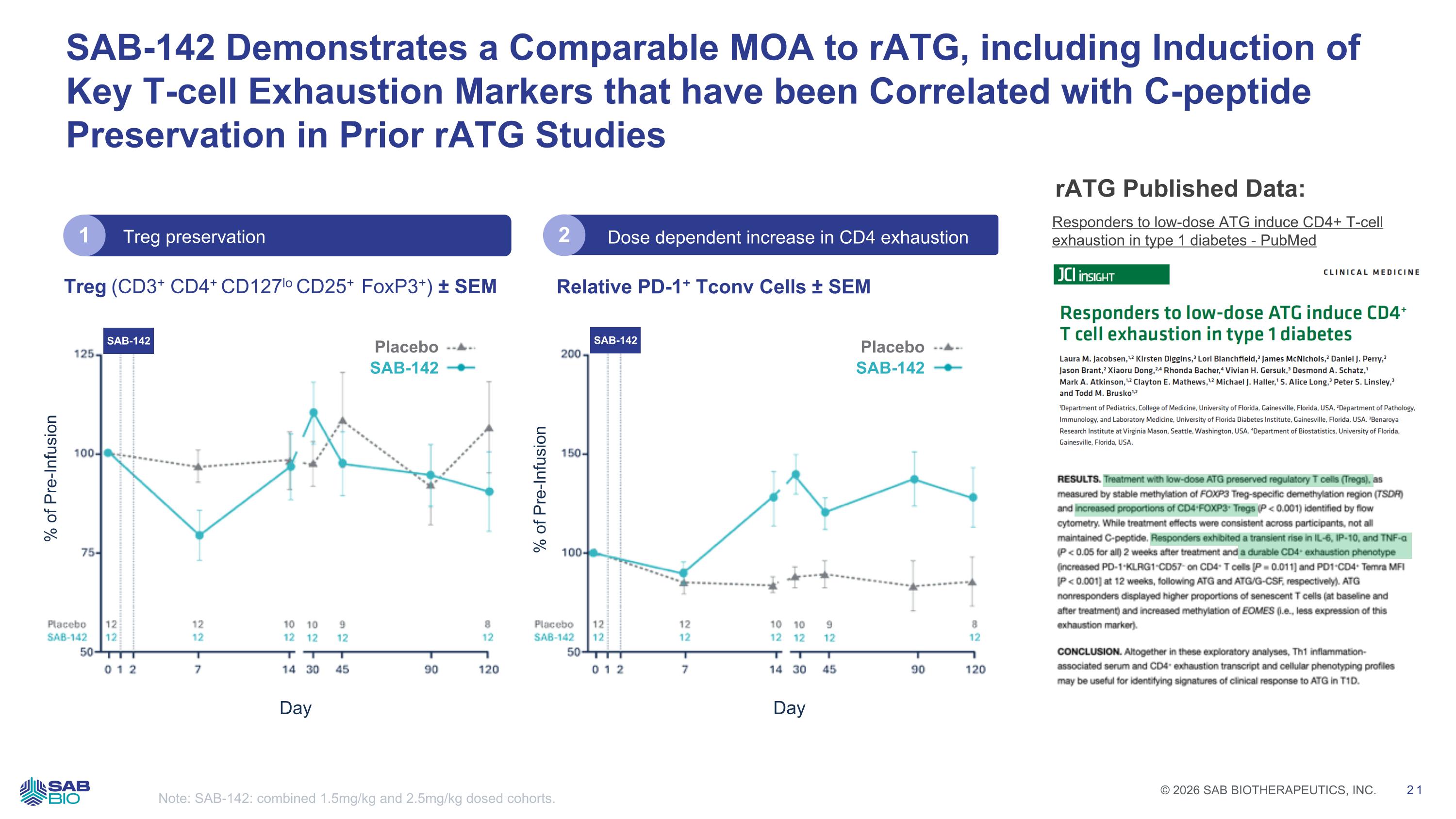
SAB-142 Demonstrates a Comparable MOA to rATG, including Induction of Key T-cell Exhaustion Markers that have been Correlated with C-peptide Preservation in Prior rATG Studies Note: SAB-142: combined 1.5mg/kg and 2.5mg/kg dosed cohorts. Treg (CD3+ CD4+ CD127lo CD25+ FoxP3+) ± SEM Relative PD-1+ Tconv Cells ± SEM Dose dependent increase in CD4 exhaustion Responders to low-dose ATG induce CD4+ T-cell exhaustion in type 1 diabetes - PubMed rATG Published Data: Placebo SAB-142 % of Pre-Infusion Day SAB-142 % of Pre-Infusion Placebo SAB-142 Day SAB-142 Treg preservation 1 2 JCI INSIGHT CLINICAL MEDICINE Responders to low-dose ATG induce CD4+ T cell exhaustion in type 1 diabetes Laura M. Jacobsen, Kirsten Diggins, Lori Blanchfield, James McNichols, Daniel J. Perry, Jason Brant, Xiaoru Dong, Rhonda Bacher, Vivian H. Gersuk, Desmond A. Schatz, Mark A. Atkinson, Clayton E. Mathews, Michael J. Haller, S. Alice Long, Peter S. Linsley, and Todd M. Brusko 'Department of Pediatrics, College of Medicine, University of Florida, Gainesville, Florida, USA. Department of Pathology, Immunology, and Laboratory Medicine, University of Florida Diabetes Institute, Gainesville, Florida, USA. 'Benaroya Research Institute at Virginia Mason, Seattle, Washington, USA. 'Department of Biostatistics, University of Florida, Gainesville, Florida, USA. RESULTS. Treatment with low-dose ATG preserved regulatory T cells (Tregs), as measured by stable methylation of FOXP3 Treg-specific demethylation region (TSDR) and increased proportions of CD4+FOXP3+ Tregs (P < 0.001) identified by flow cytometry. While treatment effects were consistent across participants, not all maintained C-peptide. Responders exhibited a transient rise in IL-6, IP-10, and TNF-a (P < 0.05 for all) 2 weeks after treatment and a durable CD4+ exhaustion phenotype (increased PD-1+KLRG1-CD57- on CD4+ T cells [P = 0.011] and PD1+CD4- Temra MFI [P < 0.001] at 12 weeks, following ATG and ATG/G-CSF, respectively). ATG nonresponders displayed higher proportions of senescent T cells (at baseline and after treatment) and increased methylation of EOMES (i.e., less expression of this exhaustion marker). CONCLUSION. Altogether in these exploratory analyses, Th1 inflammation-associated serum and CD4+ exhaustion transcript and cellular phenotyping profiles may be useful for identifying signatures of clinical response to ATG in T1D. Company Logo © 2026 SAB BIOTHERAPEUTICS, INC. 21
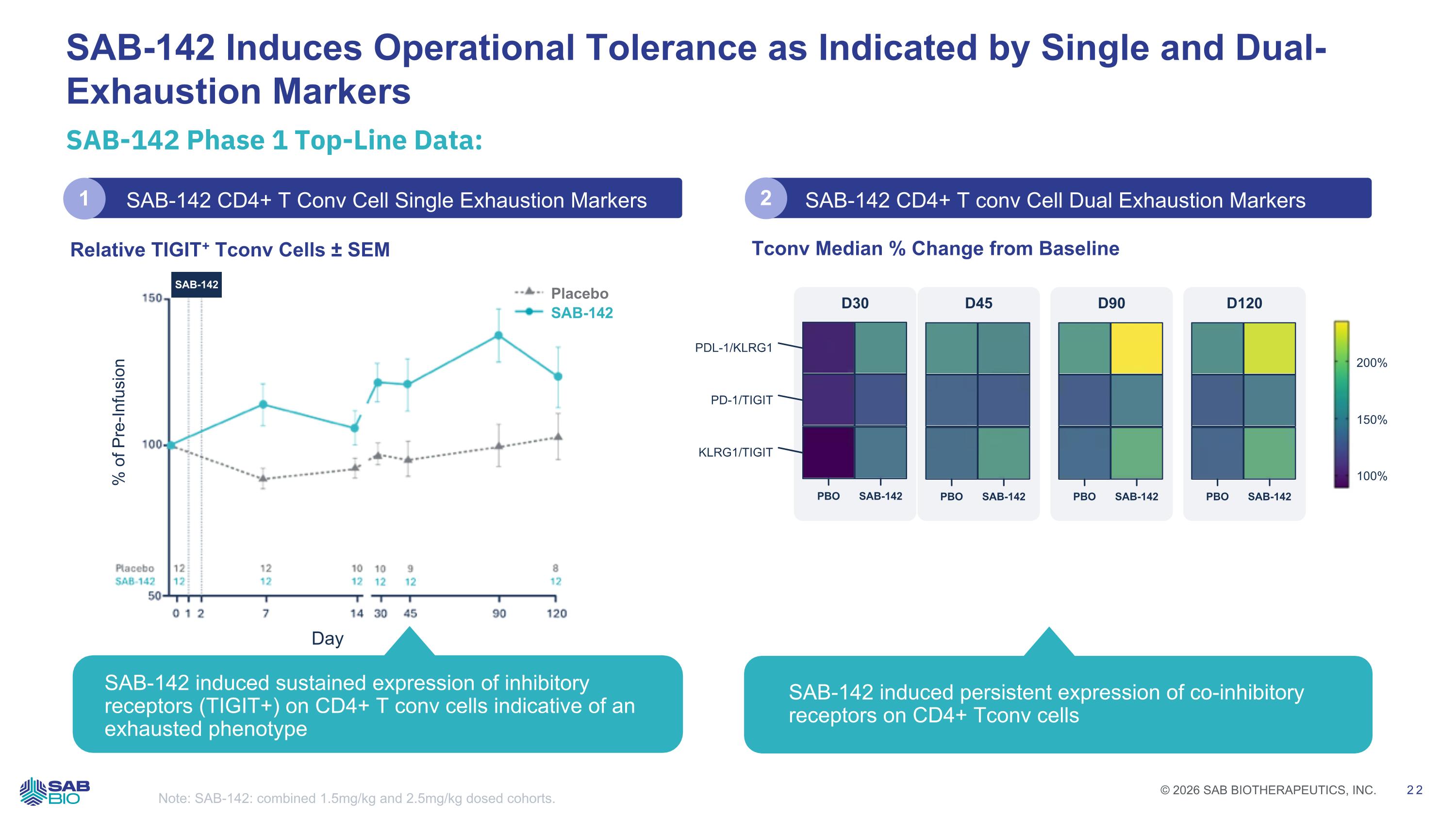
SAB-142 Phase 1 Top-Line Data: SAB-142 Induces Operational Tolerance as Indicated by Single and Dual-Exhaustion Markers Note: SAB-142: combined 1.5mg/kg and 2.5mg/kg dosed cohorts. Placebo SAB-142 % of Pre-Infusion Day SAB-142 Tconv Median % Change from Baseline PDL-1/KLRG1 PD-1/TIGIT KLRG1/TIGIT D30 PBO SAB-142 D45 PBO SAB-142 D90 PBO SAB-142 D120 PBO SAB-142 200% 150% 100% SAB-142 CD4+ T Conv Cell Single Exhaustion Markers SAB-142 CD4+ T conv Cell Dual Exhaustion Markers Relative TIGIT+ Tconv Cells ± SEM SAB-142 induced sustained expression of inhibitory receptors (TIGIT+) on CD4+ T conv cells indicative of an exhausted phenotype SAB-142 induced persistent expression of co-inhibitory receptors on CD4+ Tconv cells 1 2 Company Logo © 2026 SAB BIOTHERAPEUTICS, INC. 22
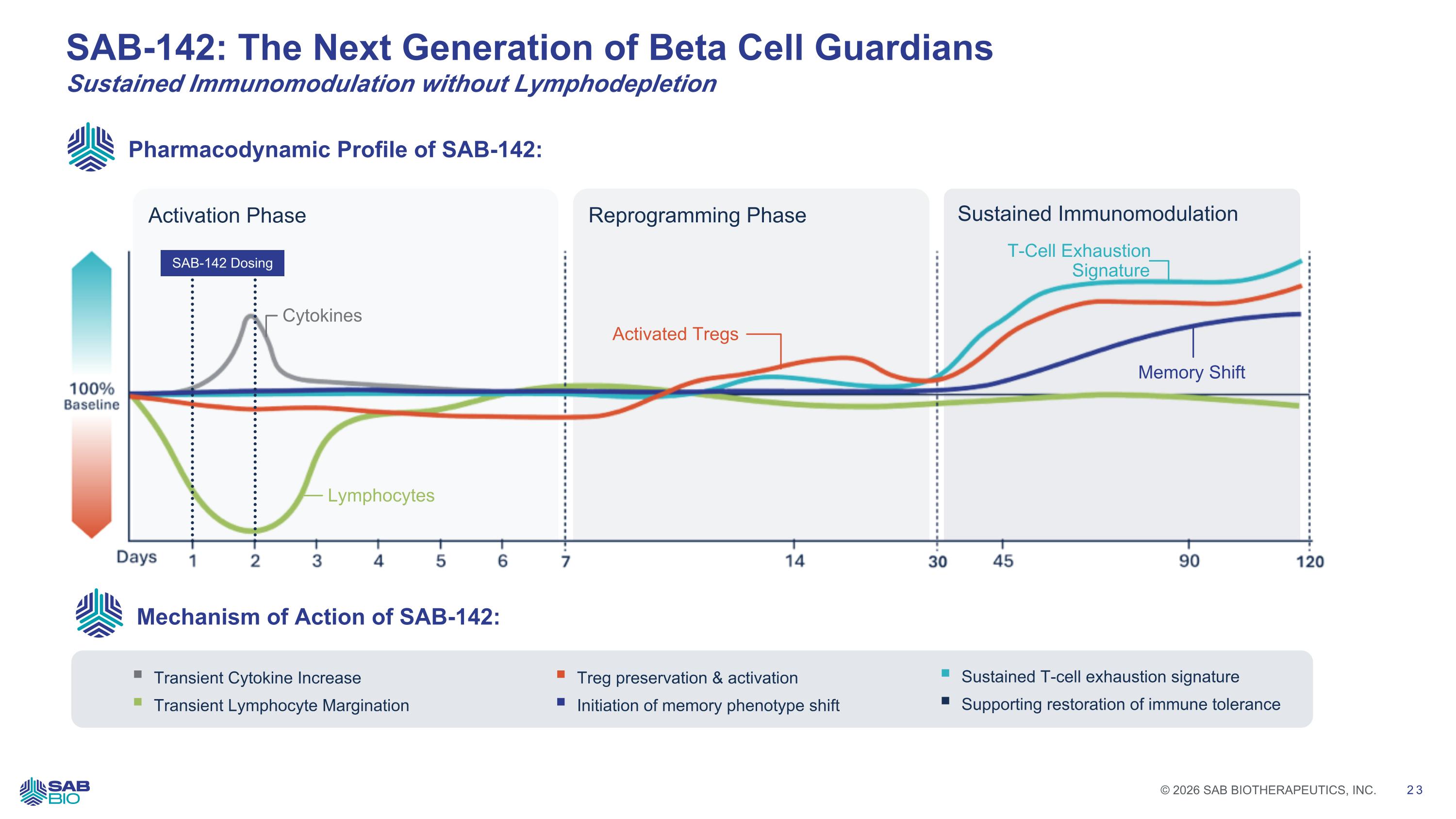
Transient Cytokine Increase Transient Lymphocyte Margination SAB-142: The Next Generation of Beta Cell Guardians Sustained Immunomodulation without Lymphodepletion Treg preservation & activation Initiation of memory phenotype shift Reprogramming Phase Sustained Immunomodulation Activation Phase Lymphocytes Activated Tregs T-Cell Exhaustion Signature Memory Shift Pharmacodynamic Profile of SAB-142: Mechanism of Action of SAB-142: Sustained T-cell exhaustion signature Supporting restoration of immune tolerance Cytokines SAB-142 Dosing Company Logo © 2026 SAB BIOTHERAPEUTICS, INC. 23
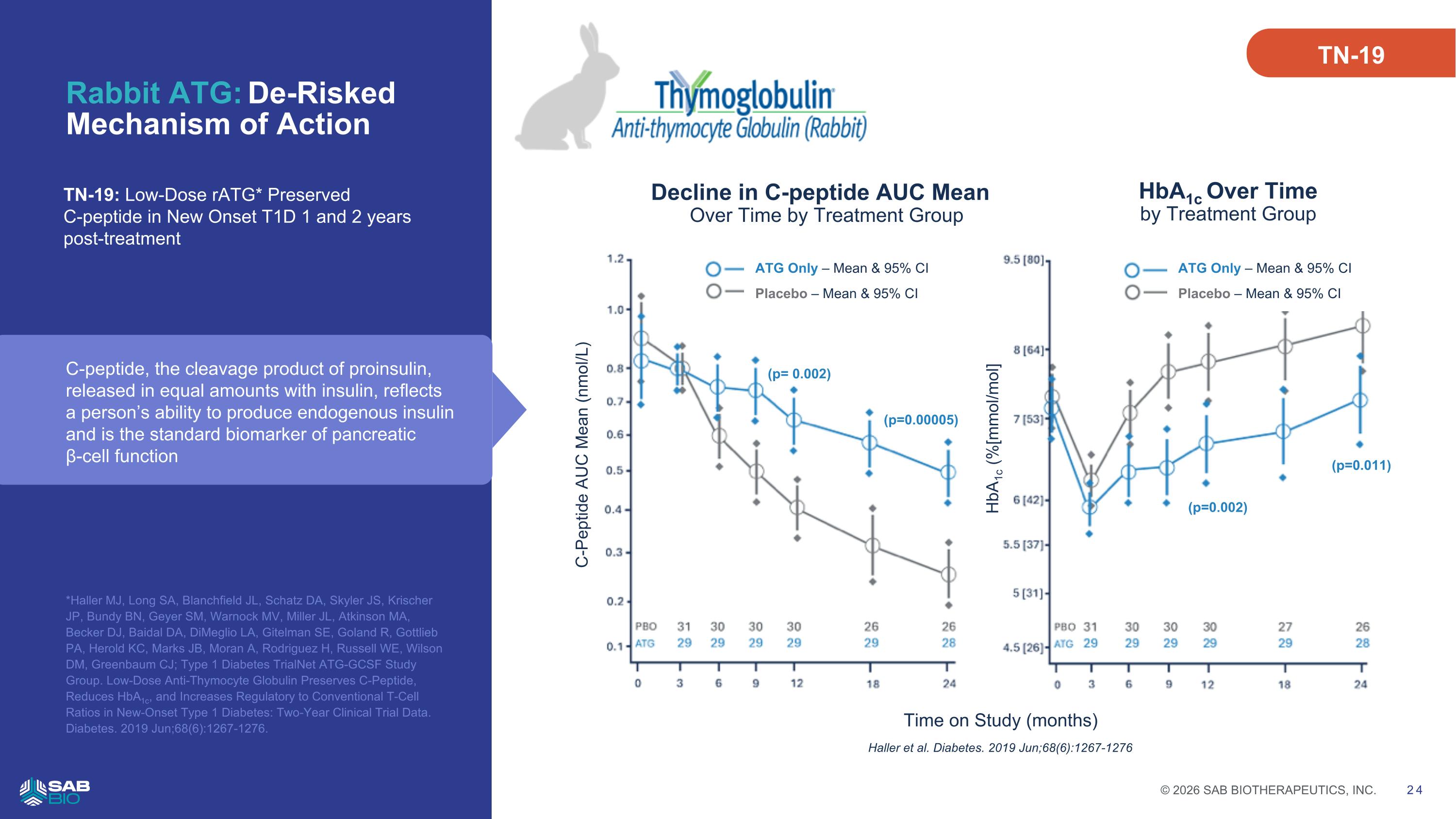
Rabbit ATG: De-Risked Mechanism of Action *Haller MJ, Long SA, Blanchfield JL, Schatz DA, Skyler JS, Krischer JP, Bundy BN, Geyer SM, Warnock MV, Miller JL, Atkinson MA, Becker DJ, Baidal DA, DiMeglio LA, Gitelman SE, Goland R, Gottlieb PA, Herold KC, Marks JB, Moran A, Rodriguez H, Russell WE, Wilson DM, Greenbaum CJ; Type 1 Diabetes TrialNet ATG-GCSF Study Group. Low-Dose Anti-Thymocyte Globulin Preserves C-Peptide, Reduces HbA1c, and Increases Regulatory to Conventional T-Cell Ratios in New-Onset Type 1 Diabetes: Two-Year Clinical Trial Data. Diabetes. 2019 Jun;68(6):1267-1276. TN-19: Low-Dose rATG* Preserved C-peptide in New Onset T1D 1 and 2 years post-treatment ATG Only – Mean & 95% CI Placebo – Mean & 95% CI 24 HbA1c Over Time by Treatment Group HbA1c (%[mmol/mol] Time on Study (months) ATG Only – Mean & 95% CI Placebo – Mean & 95% CI C-Peptide AUC Mean (nmol/L) (p=0.00005) Haller et al. Diabetes. 2019 Jun;68(6):1267-1276 Decline in C-peptide AUC Mean Over Time by Treatment Group (p=0.002) (p= 0.002) (p=0.011) TN-19 C-peptide, the cleavage product of proinsulin, released in equal amounts with insulin, reflects a person’s ability to produce endogenous insulin and is the standard biomarker of pancreatic β-cell function Company Logo © 2026 SAB BIOTHERAPEUTICS, INC. 24
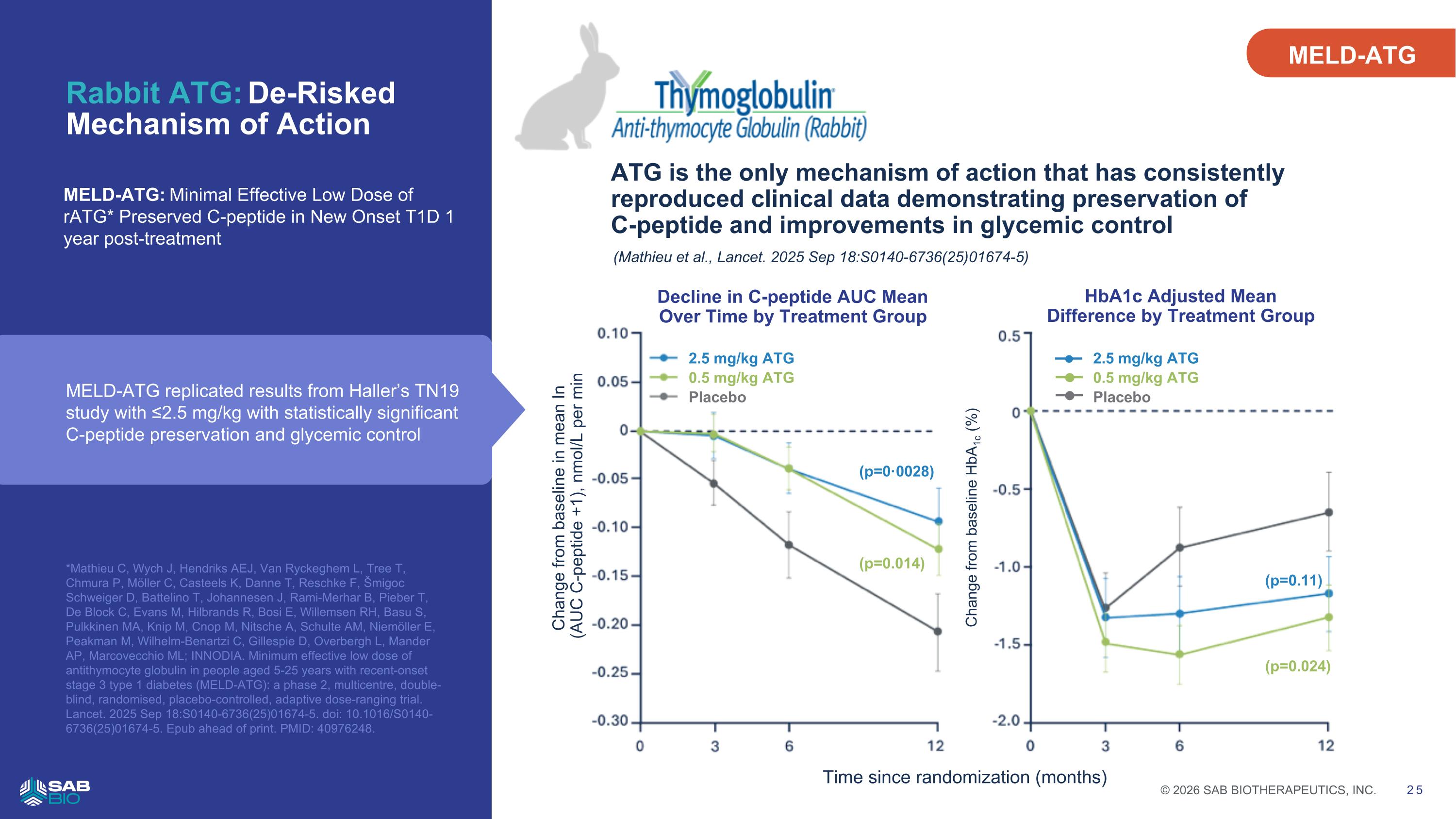
Rabbit ATG: De-Risked Mechanism of Action MELD-ATG: Minimal Effective Low Dose of rATG* Preserved C-peptide in New Onset T1D 1 year post-treatment 25 ATG is the only mechanism of action that has consistently reproduced clinical data demonstrating preservation of C-peptide and improvements in glycemic control (Mathieu et al., Lancet. 2025 Sep 18:S0140-6736(25)01674-5) 2.5 mg/kg ATG 0.5 mg/kg ATG Placebo Change from baseline in mean In (AUC C-peptide +1), nmol/L per min Time since randomization (months) Change from baseline HbA1c (%) Decline in C-peptide AUC Mean Over Time by Treatment Group HbA1c Adjusted Mean Difference by Treatment Group (p=0·0028) (p=0.014) (p=0.11) (p=0.024) 2.5 mg/kg ATG 0.5 mg/kg ATG Placebo MELD-ATG replicated results from Haller’s TN19 study with ≤2.5 mg/kg with statistically significant C-peptide preservation and glycemic control MELD-ATG *Mathieu C, Wych J, Hendriks AEJ, Van Ryckeghem L, Tree T, Chmura P, Möller C, Casteels K, Danne T, Reschke F, Šmigoc Schweiger D, Battelino T, Johannesen J, Rami-Merhar B, Pieber T, De Block C, Evans M, Hilbrands R, Bosi E, Willemsen RH, Basu S, Pulkkinen MA, Knip M, Cnop M, Nitsche A, Schulte AM, Niemöller E, Peakman M, Wilhelm-Benartzi C, Gillespie D, Overbergh L, Mander AP, Marcovecchio ML; INNODIA. Minimum effective low dose of antithymocyte globulin in people aged 5-25 years with recent-onset stage 3 type 1 diabetes (MELD-ATG): a phase 2, multicentre, double-blind, randomised, placebo-controlled, adaptive dose-ranging trial. Lancet. 2025 Sep 18:S0140-6736(25)01674-5. doi: 10.1016/S0140-6736(25)01674-5. Epub ahead of print. PMID: 40976248. Company Logo © 2026 SAB BIOTHERAPEUTICS, INC. 25
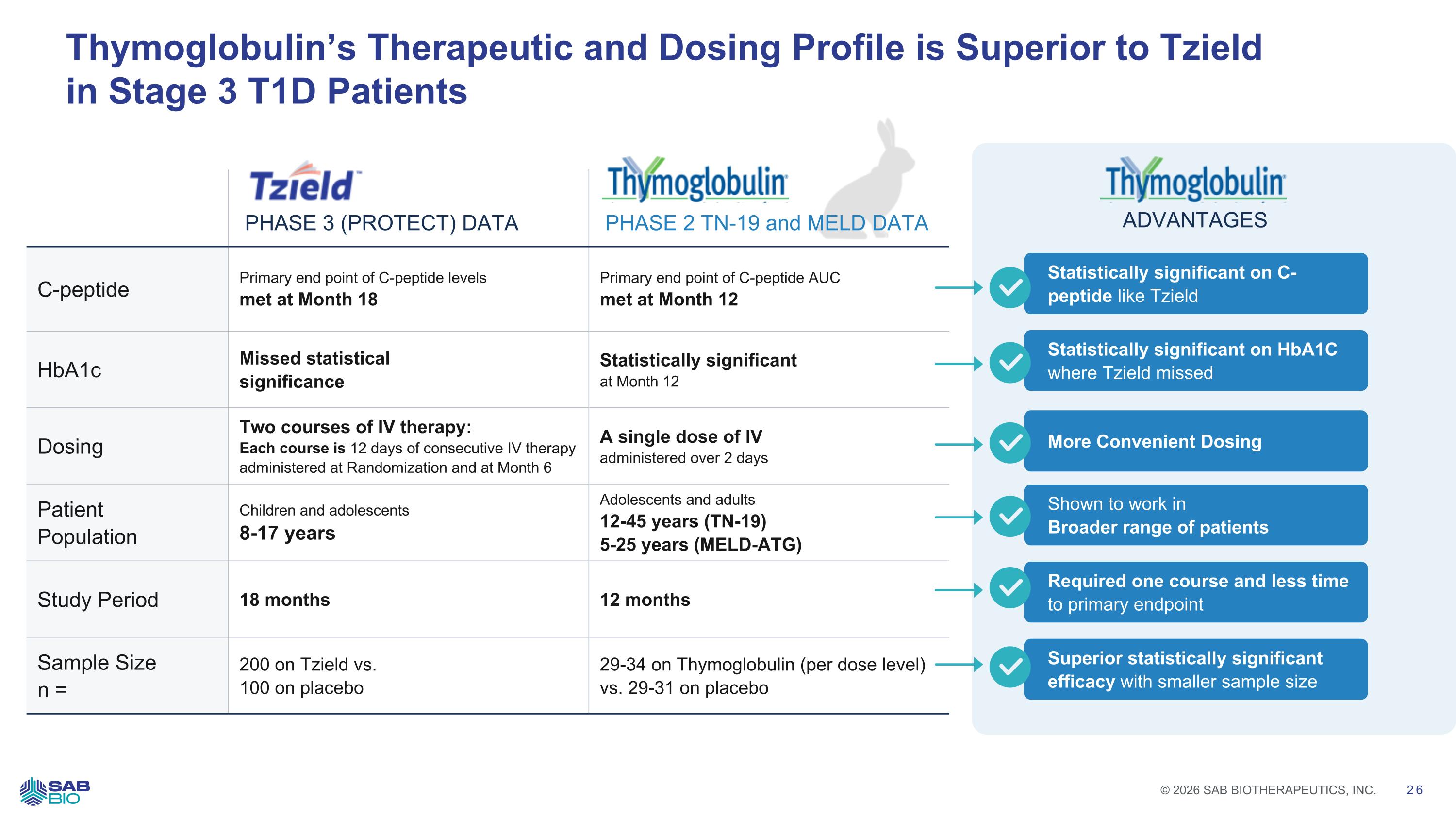
Thymoglobulin’s Therapeutic and Dosing Profile is Superior to Tzield in Stage 3 T1D Patients PHASE 3 (PROTECT) DATA PHASE 2 TN-19 and MELD DATA C-peptide Primary end point of C-peptide levels met at Month 18 Primary end point of C-peptide AUC met at Month 12 HbA1c Missed statistical significance Statistically significant at Month 12 Dosing Two courses of IV therapy: Each course is 12 days of consecutive IV therapy administered at Randomization and at Month 6 A single dose of IV administered over 2 days Patient Population Children and adolescents 8-17 years Adolescents and adults 12-45 years (TN-19) 5-25 years (MELD-ATG) Study Period 18 months 12 months Sample Size n = 200 on Tzield vs. 100 on placebo 29-34 on Thymoglobulin (per dose level) vs. 29-31 on placebo Statistically significant on C-peptide like Tzield Statistically significant on HbA1C where Tzield missed More Convenient Dosing Shown to work in Broader range of patients Required one course and less time to primary endpoint Superior statistically significant efficacy with smaller sample size ADVANTAGES Company Logo © 2026 SAB BIOTHERAPEUTICS, INC. 26