

FEBRUARY 2026 Fourth Quarter & Full-Year 2025 Financial Results & Corporate Progress

Forward Looking Statements This presentation and the accompanying oral commentary contain forward-looking statements that are based on our management’s beliefs and assumptions and on information currently available to our management. Forward-looking statements are inherently subject to risks and uncertainties, some of which cannot be predicted or quantified. In some cases, you can identify forward-looking statements by terminology such as “may,” “will,” “should,” “could,” “expect,” “plan,” anticipate,” “believe,” “estimate,” “predict,” “intend,” “potential,” “would,” “continue,” “ongoing” or the negative of these terms or other comparable terminology. Forward-looking statements include all statements other than statements of historical fact contained in this presentation, including information concerning our future financial performance, including the sufficiency of our cash, cash equivalents and short-term investments to fund our operations, business plans and objectives, timing and success of our commercialization and marketing efforts, timing and success of our planned nonclinical and clinical development activities, the potential benefits and therapeutic scope of our acquisitions and collaborations, including our acquisition of Mersana Therapeutics, Inc. and its lead product candidate, emiltatug ledadotin (XMT-1660), or Emi-Le, the results of any of our strategic collaborations, including the potential achievement of milestones and provision of royalty payments thereunder, efficacy and safety profiles of our products and product candidates, the ability of OJEMDA™ (tovorafenib) to treat pediatric low-grade glioma (pLGG) or related indications, the potential therapeutic benefits and economic value of our products and product candidates, potential growth opportunities, competitive position, industry environment and potential market opportunities, our ability to protect intellectual property and the impact of global business or macroeconomic conditions, including as a result of inflation, fluctuating interest rates, supply chain disruptions, labor shortages or displacement, high unemployment rates, government shutdowns, significant political, trade or regulatory developments, including tariffs, shifting priorities within the U.S. Food and Drug Administration and reduced funding of federal healthcare programs, and global regional conflicts, on our business and operations. Forward-looking statements are subject to known and unknown risks, uncertainties, assumptions and other factors. It is not possible for our management to predict all risks, nor can we assess the impact of all factors on our business or the extent to which any factor, or combination of factors, may cause actual results to differ materially from those contained in any forward-looking statements we may make. These factors, together with those that are described under the heading “Risk Factors” contained in our most recent Annual Report on Form 10-K filed with the Securities and Exchange Commission (SEC) and other documents we file from time to time with the SEC, may cause our actual results, performance or achievements to differ materially and adversely from those anticipated or implied by our forward-looking statements. In addition, statements that “we believe” and similar statements reflect our beliefs and opinions on the relevant subject. These statements are based upon information available to us as of the date of this presentation, and although we believe such information forms a reasonable basis for such statements, such information may be limited or incomplete, and our statements should not be read to indicate that we have conducted a thorough inquiry into, or review of, all potentially available relevant information. These statements are inherently uncertain and investors are cautioned not to unduly rely upon these statements. Furthermore, if our forward-looking statements prove to be inaccurate, the inaccuracy may be material. In light of the significant uncertainties in these forward-looking statements, you should not regard these statements as a representation or warranty by us or any other person that we will achieve our objectives and plans in any specified time frame, or at all. We undertake no obligation to publicly update any forward-looking statements, whether as a result of new information, future events or otherwise, except as required by law. This presentation also contains estimates and other statistical data made by independent parties and by us relating to market size and growth and other data about our industry. This data involves a number of assumptions and limitations, and you are cautioned not to give undue weight to such estimates. In addition, projections, assumptions and estimates of our future performance and the future performance of the markets in which we operate are necessarily subject to a high degree of uncertainty and risk.
Agenda & Day One Participants Opening Remarks Jeremy Bender (Chief Executive Officer) OJEMDATM 3-Year Data Summary Michael Vasconcelles (Head of R&D) R&D Overview Michael Vasconcelles (Head of R&D) Financial Performance Charles York (Chief Operating & Chief Financial Officer) Q&A Session All Participants OJEMDA Performance Lauren Merendino (Chief Commercial Officer)

Opening Remarks Jeremy Bender Chief Executive Officer

Q4 & FY 2025 – A Transformational Year For Day One EMA: European Medicines Agency 1 Represents the comparison of full year 2024 (April 2024 approval) to full year 2025. 2 Regulatory submission process executed by Ipsen. 3 Represents cash, cash equivalents and short-term investments as of December 31, 2025. r/r, relapsed or refractory pLGG. Financial Position Pipeline Progress $155.4M (+172%1) net product revenue in 2025 4,635 (+181%1) prescriptions in 2025 2026 guidance of $225M - $250M net product revenue >$400M product opportunity in r/r pLGG FIREFLY-2 trial enrollment completion expected 1H 2026 Tovorafenib EMA regulatory decision expected in 20262 Emi-Le, B7-H4-targeted ADC, Phase 1 data expected mid-2026 DAY301, PTK7-targeted ADC, initial Phase 1a data update expected 2H 2026 Strong balance sheet with $441M in cash3 Disciplined investment and execution across commercial, clinical and corporate development

OJEMDA 3-Year Data Summary Michael Vasconcelles Head of R&D
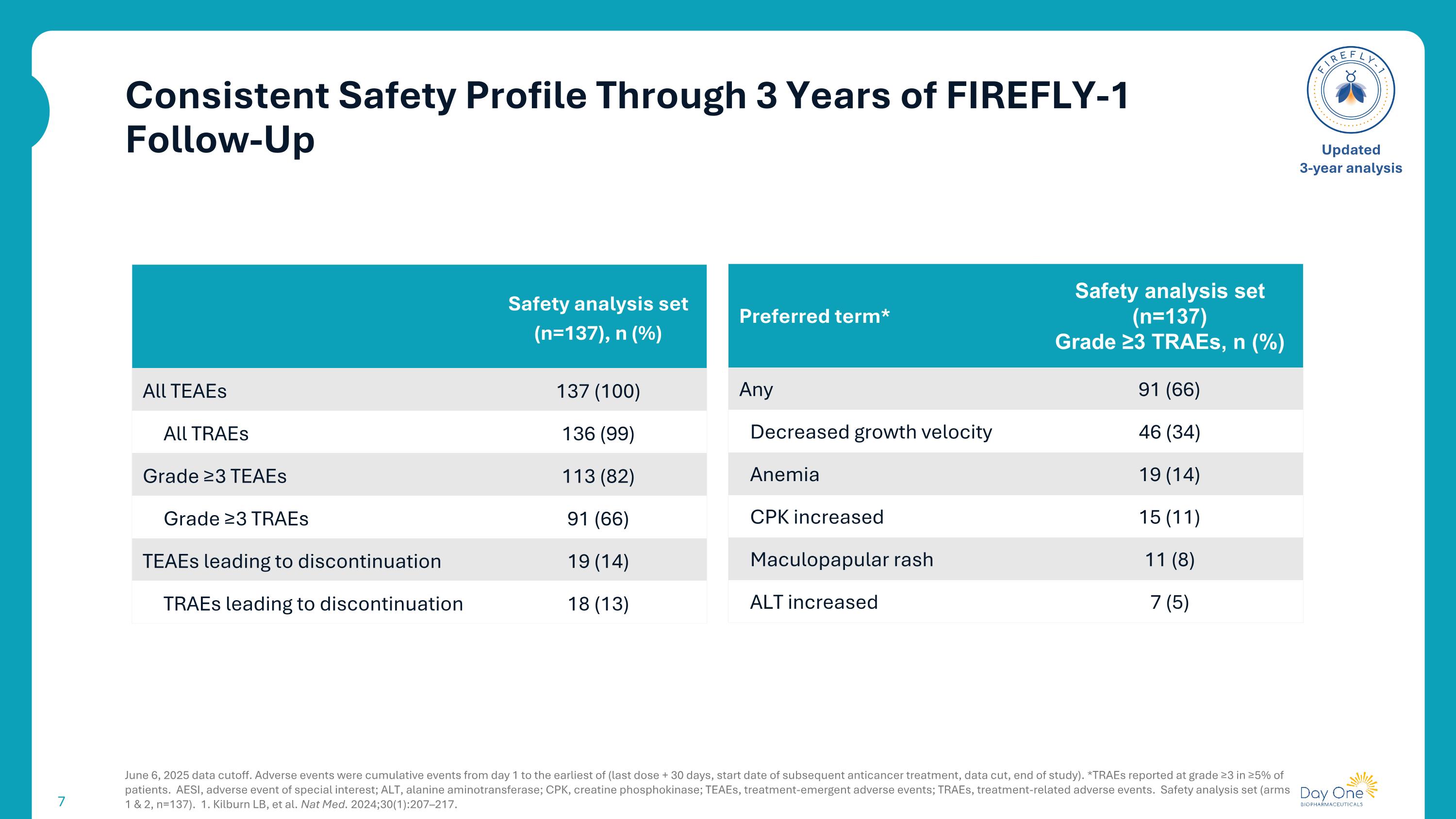
Consistent Safety Profile Through 3 Years of FIREFLY-1 Follow-Up June 6, 2025 data cutoff. Adverse events were cumulative events from day 1 to the earliest of (last dose + 30 days, start date of subsequent anticancer treatment, data cut, end of study). *TRAEs reported at grade ≥3 in ≥5% of patients. AESI, adverse event of special interest; ALT, alanine aminotransferase; CPK, creatine phosphokinase; TEAEs, treatment-emergent adverse events; TRAEs, treatment-related adverse events. Safety analysis set (arms 1 & 2, n=137). 1. Kilburn LB, et al. Nat Med. 2024;30(1):207–217. Updated 3-year analysis Safety analysis set (n=137), n (%) All TEAEs 137 (100) All TRAEs 136 (99) Grade ≥3 TEAEs 113 (82) Grade ≥3 TRAEs 91 (66) TEAEs leading to discontinuation 19 (14) TRAEs leading to discontinuation 18 (13) Preferred term* Safety analysis set (n=137) Grade ≥3 TRAEs, n (%) Any 91 (66) Decreased growth velocity 46 (34) Anemia 19 (14) CPK increased 15 (11) Maculopapular rash 11 (8) ALT increased 7 (5)
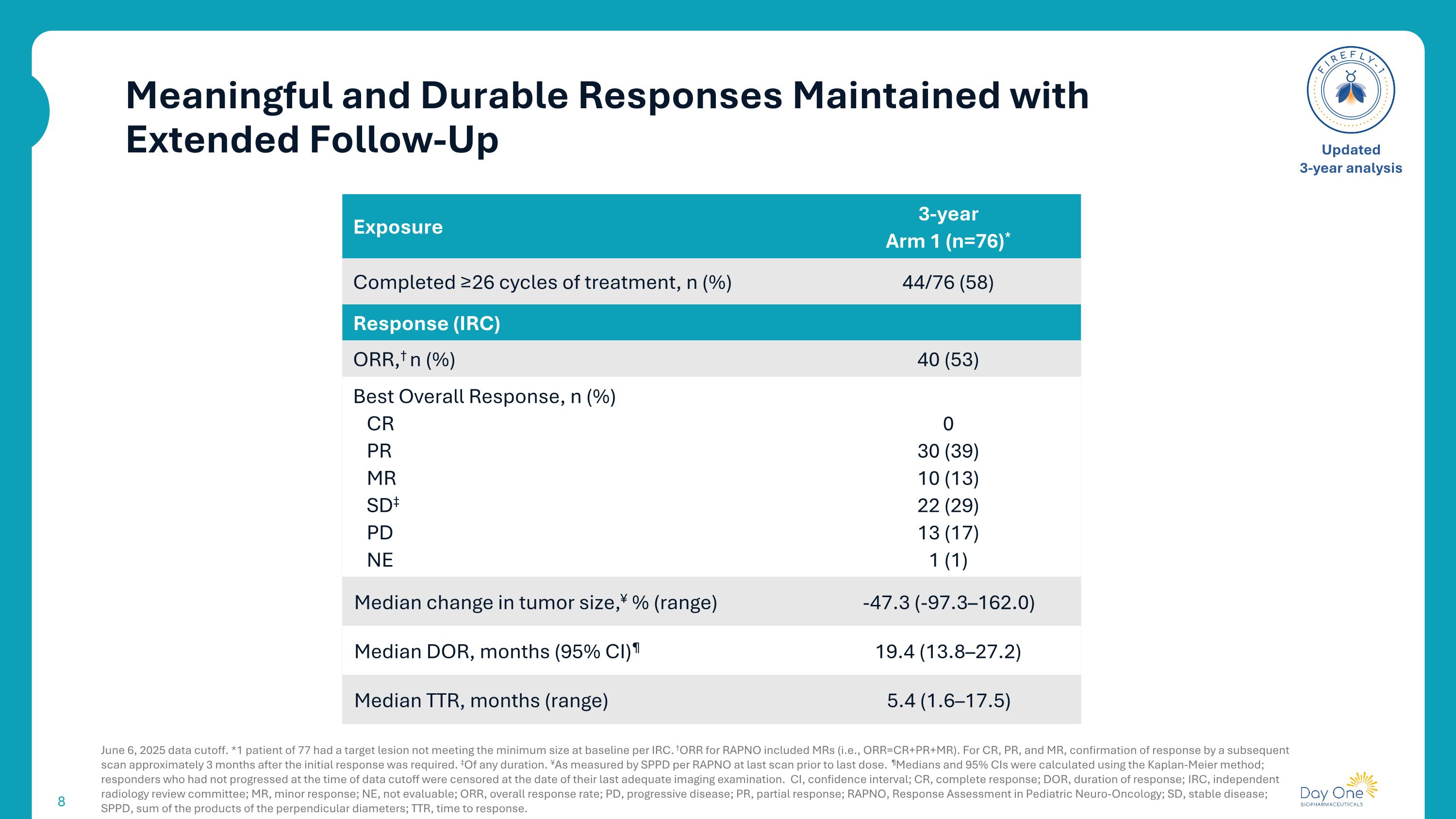
Meaningful and Durable Responses Maintained with Extended Follow-Up June 6, 2025 data cutoff. *1 patient of 77 had a target lesion not meeting the minimum size at baseline per IRC. †ORR for RAPNO included MRs (i.e., ORR=CR+PR+MR). For CR, PR, and MR, confirmation of response by a subsequent scan approximately 3 months after the initial response was required. ‡Of any duration. ¥As measured by SPPD per RAPNO at last scan prior to last dose. ¶Medians and 95% CIs were calculated using the Kaplan-Meier method; responders who had not progressed at the time of data cutoff were censored at the date of their last adequate imaging examination. CI, confidence interval; CR, complete response; DOR, duration of response; IRC, independent radiology review committee; MR, minor response; NE, not evaluable; ORR, overall response rate; PD, progressive disease; PR, partial response; RAPNO, Response Assessment in Pediatric Neuro-Oncology; SD, stable disease; SPPD, sum of the products of the perpendicular diameters; TTR, time to response. Updated 3-year analysis Exposure 3-year Arm 1 (n=76)* Completed ≥26 cycles of treatment, n (%) 44/76 (58) Response (IRC) ORR,† n (%) 40 (53) Best Overall Response, n (%) CR PR MR SD‡ PD NE 0 30 (39) 10 (13) 22 (29) 13 (17) 1 (1) Median change in tumor size,¥ % (range) -47.3 (-97.3–162.0) Median DOR, months (95% CI)¶ 19.4 (13.8–27.2) Median TTR, months (range) 5.4 (1.6–17.5)
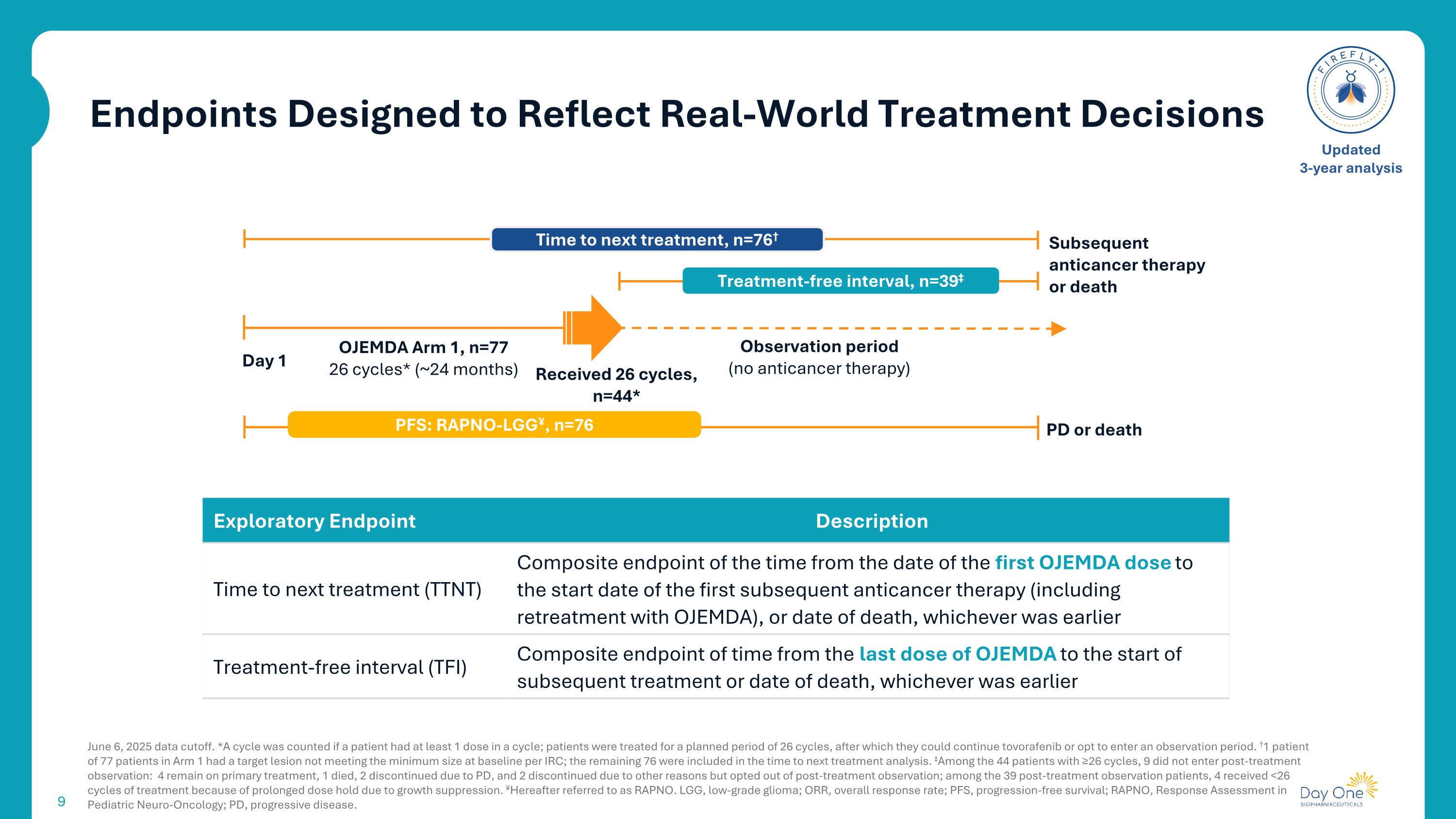
Endpoints Designed to Reflect Real-World Treatment Decisions June 6, 2025 data cutoff. *A cycle was counted if a patient had at least 1 dose in a cycle; patients were treated for a planned period of 26 cycles, after which they could continue tovorafenib or opt to enter an observation period. †1 patient of 77 patients in Arm 1 had a target lesion not meeting the minimum size at baseline per IRC; the remaining 76 were included in the time to next treatment analysis. ‡Among the 44 patients with ≥26 cycles, 9 did not enter post-treatment observation: 4 remain on primary treatment, 1 died, 2 discontinued due to PD, and 2 discontinued due to other reasons but opted out of post-treatment observation; among the 39 post-treatment observation patients, 4 received <26 cycles of treatment because of prolonged dose hold due to growth suppression. ¥Hereafter referred to as RAPNO. LGG, low-grade glioma; ORR, overall response rate; PFS, progression-free survival; RAPNO, Response Assessment in Pediatric Neuro-Oncology; PD, progressive disease. Updated 3-year analysis Subsequent anticancer therapy or death OJEMDA Arm 1, n=77 26 cycles* (~24 months) Received 26 cycles, n=44* Observation period (no anticancer therapy) Time to next treatment, n=76† Treatment-free interval, n=39‡ Day 1 PD or death PFS: RAPNO-LGG¥, n=76 Exploratory Endpoint Description Time to next treatment (TTNT) Composite endpoint of the time from the date of the first OJEMDA dose to the start date of the first subsequent anticancer therapy (including retreatment with OJEMDA), or date of death, whichever was earlier Treatment-free interval (TFI) Composite endpoint of time from the last dose of OJEMDA to the start of subsequent treatment or date of death, whichever was earlier
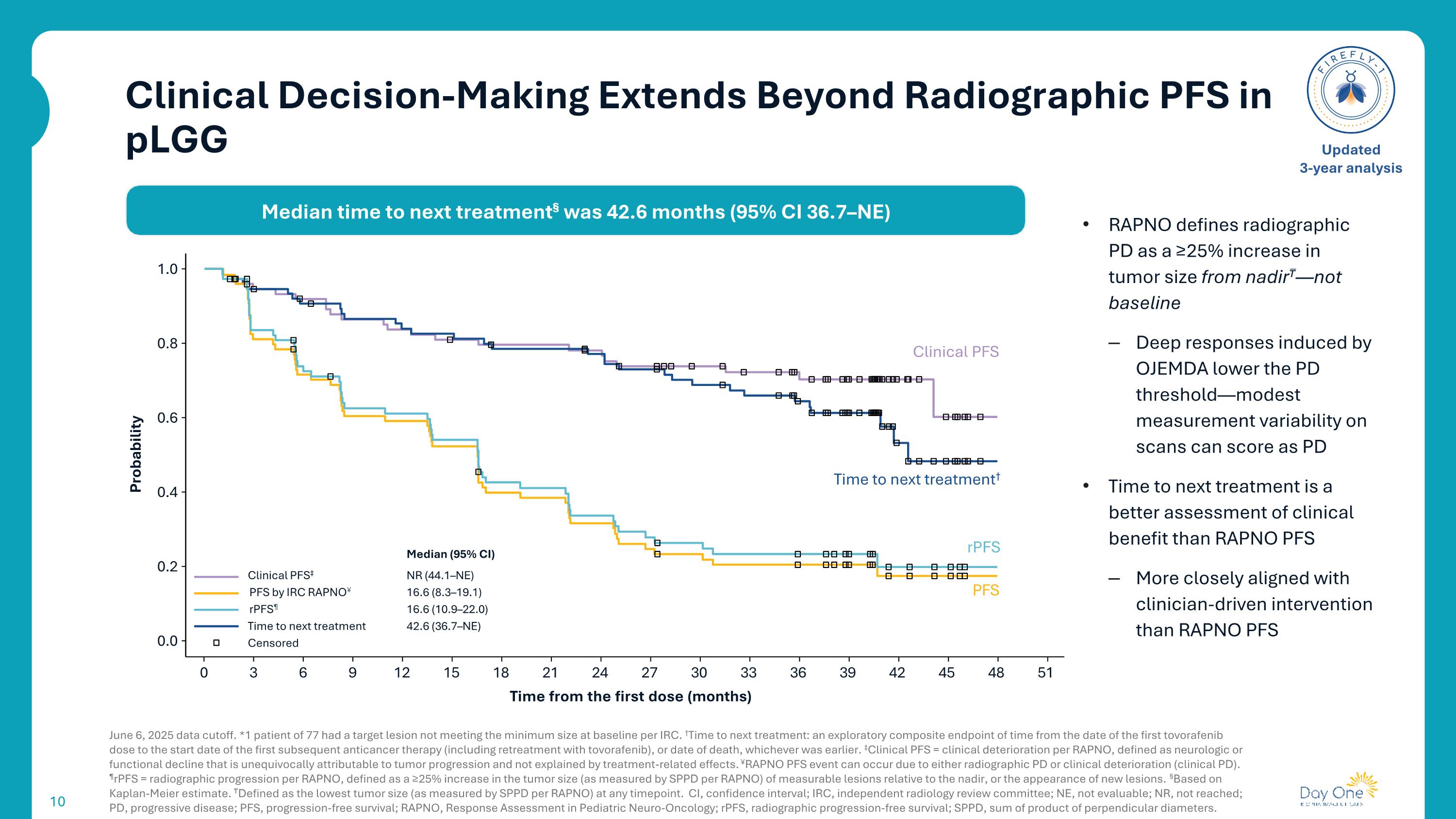
Clinical Decision-Making Extends Beyond Radiographic PFS in pLGG June 6, 2025 data cutoff. *1 patient of 77 had a target lesion not meeting the minimum size at baseline per IRC. †Time to next treatment: an exploratory composite endpoint of time from the date of the first tovorafenib dose to the start date of the first subsequent anticancer therapy (including retreatment with tovorafenib), or date of death, whichever was earlier. ‡Clinical PFS = clinical deterioration per RAPNO, defined as neurologic or functional decline that is unequivocally attributable to tumor progression and not explained by treatment-related effects. ¥RAPNO PFS event can occur due to either radiographic PD or clinical deterioration (clinical PD). ¶rPFS = radiographic progression per RAPNO, defined as a ≥25% increase in the tumor size (as measured by SPPD per RAPNO) of measurable lesions relative to the nadir, or the appearance of new lesions. §Based on Kaplan-Meier estimate. ₸Defined as the lowest tumor size (as measured by SPPD per RAPNO) at any timepoint. CI, confidence interval; IRC, independent radiology review committee; NE, not evaluable; NR, not reached; PD, progressive disease; PFS, progression-free survival; RAPNO, Response Assessment in Pediatric Neuro-Oncology; rPFS, radiographic progression-free survival; SPPD, sum of product of perpendicular diameters. Updated 3-year analysis 1.0 0.0 0 3 6 9 12 15 18 21 24 27 30 33 36 39 42 45 48 51 Time from the first dose (months) 0.8 0.6 0.4 0.2 Probability Clinical PFS rPFS PFS Time to next treatment† Median (95% CI) Clinical PFS‡ NR (44.1–NE) PFS by IRC RAPNO¥ 16.6 (8.3–19.1) rPFS¶ 16.6 (10.9–22.0) Time to next treatment 42.6 (36.7–NE) Censored Median time to next treatment§ was 42.6 months (95% CI 36.7–NE) RAPNO defines radiographic PD as a ≥25% increase in tumor size from nadir₸—not baseline Deep responses induced by OJEMDA lower the PD threshold—modest measurement variability on scans can score as PD Time to next treatment is a better assessment of clinical benefit than RAPNO PFS More closely aligned with clinician-driven intervention than RAPNO PFS

OJEMDA Performance Lauren Merendino Chief Commercial Officer
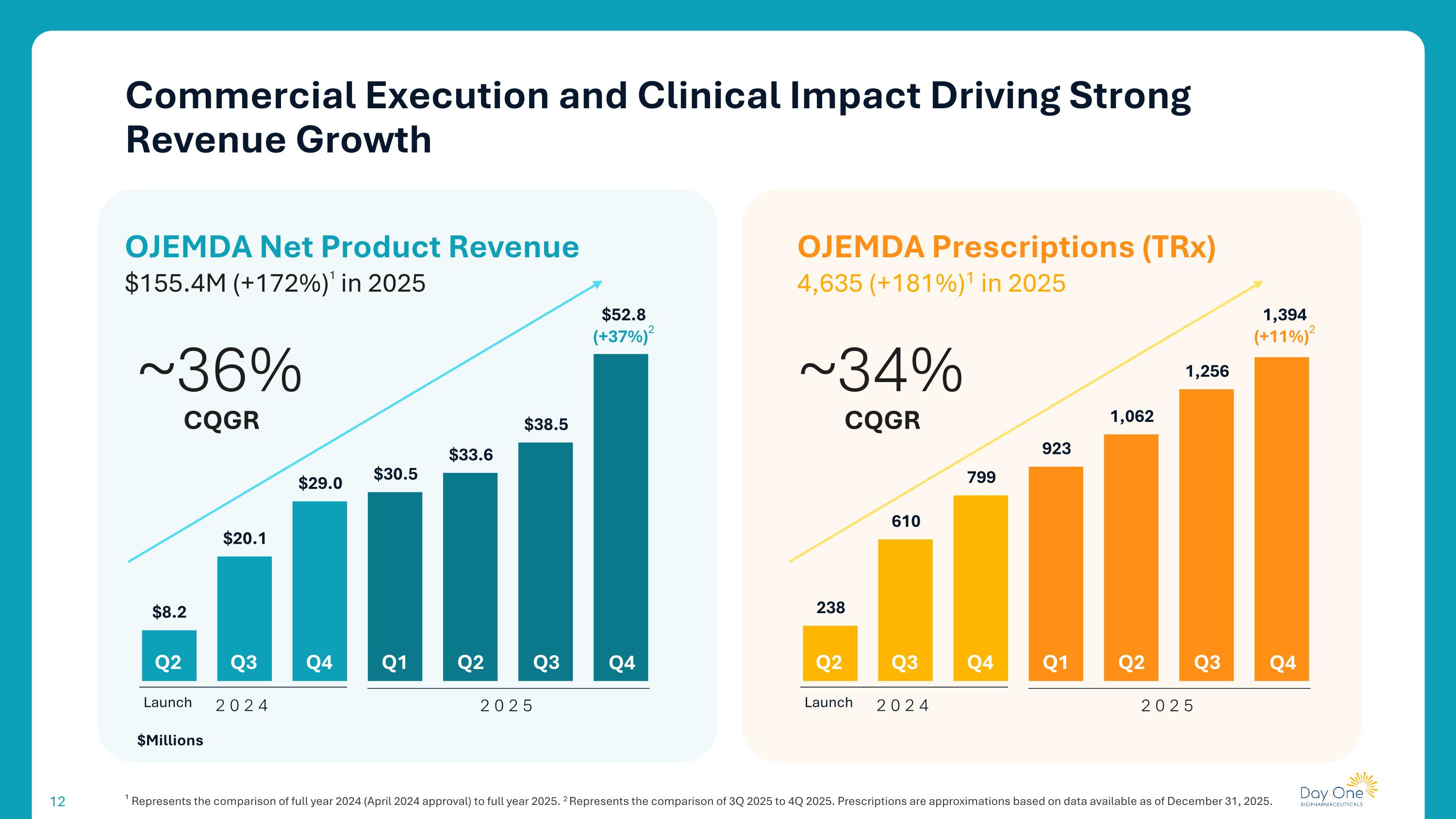
Commercial Execution and Clinical Impact Driving Strong Revenue Growth ~36% Q2 Q3 Q4 Q1 Q2 Q3 Q4 2024 2025 OJEMDA Net Product Revenue $155.4M (+172%)1 in 2025 CQGR Launch $52.8 (+37%)2 $Millions OJEMDA Prescriptions (TRx) 4,635 (+181%)1 in 2025 ~34% Q2 Q3 Q4 Q1 Q2 Q3 Q4 2024 2025 CQGR Launch 1,394 (+11%)2 ¹ Represents the comparison of full year 2024 (April 2024 approval) to full year 2025. 2 Represents the comparison of 3Q 2025 to 4Q 2025. Prescriptions are approximations based on data available as of December 31, 2025.
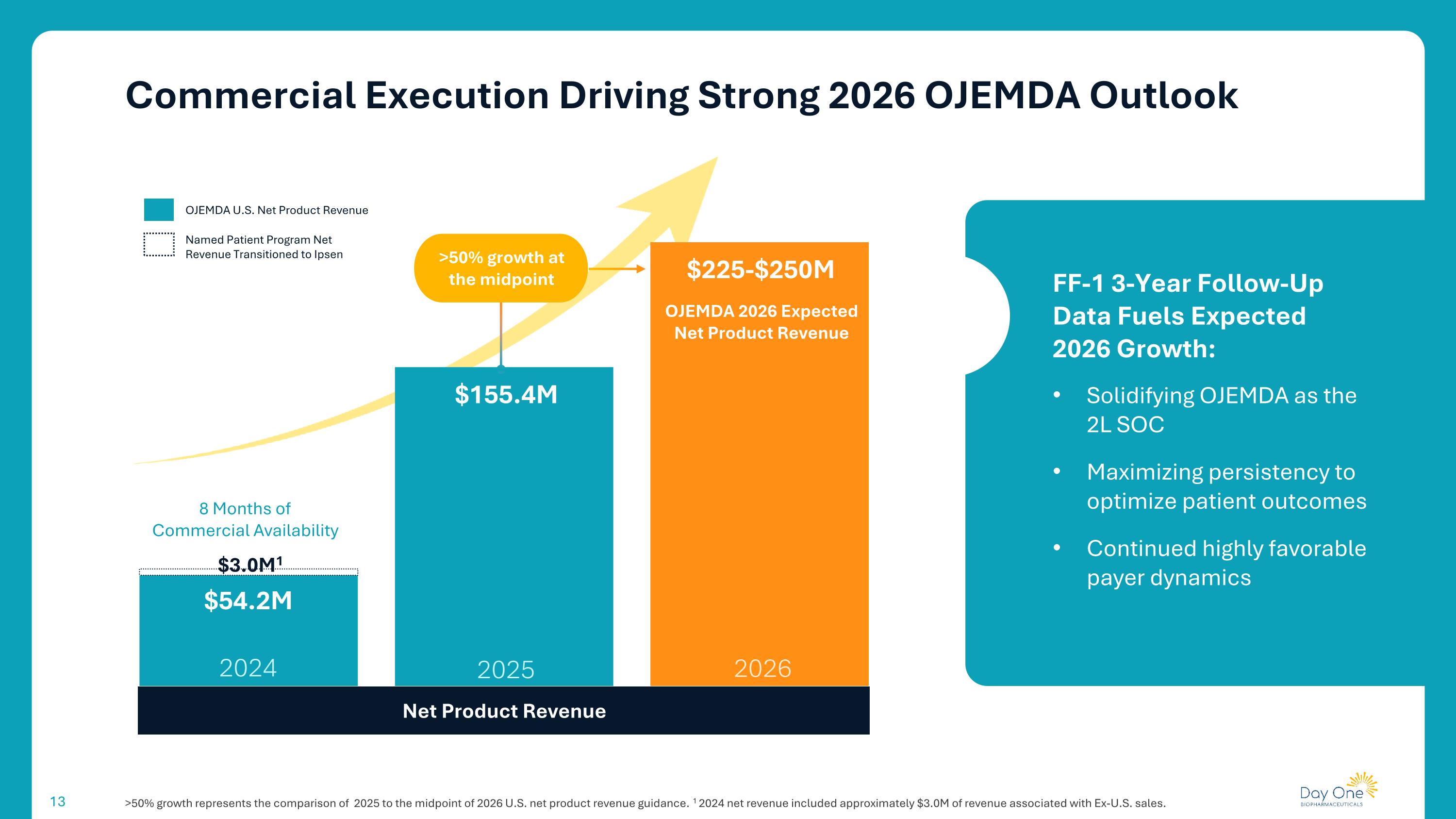
Commercial Execution Driving Strong 2026 OJEMDA Outlook >50% growth represents the comparison of 2025 to the midpoint of 2026 U.S. net product revenue guidance. 1 2024 net revenue included approximately $3.0M of revenue associated with Ex-U.S. sales. FF-1 3-Year Follow-Up Data Fuels Expected 2026 Growth: Solidifying OJEMDA as the 2L SOC Maximizing persistency to optimize patient outcomes Continued highly favorable payer dynamics Net Product Revenue 2026 2024 2025 8 Months of Commercial Availability OJEMDA 2026 Expected Net Product Revenue $155.4M $54.2M $225-$250M >50% growth at the midpoint $3.0M1 OJEMDA U.S. Net Product Revenue Named Patient Program Net Revenue Transitioned to Ipsen
Our Strategy: Focused Execution to Solidify OJEMDA as 2L Standard of Care * On label patients only. Drive New Patient Starts Optimize Persistence Increase depth of prescribing through continued evidence generation that reinforces how OJEMDA’s clinical profile and benefits align with attributes physicians prioritize when treating pLGG patients* Support physicians and patients to optimize their experience on OJEMDA including effective AE management and appropriate dose-adjustments

R&D Overview Michael Vasconcelles Head of R&D
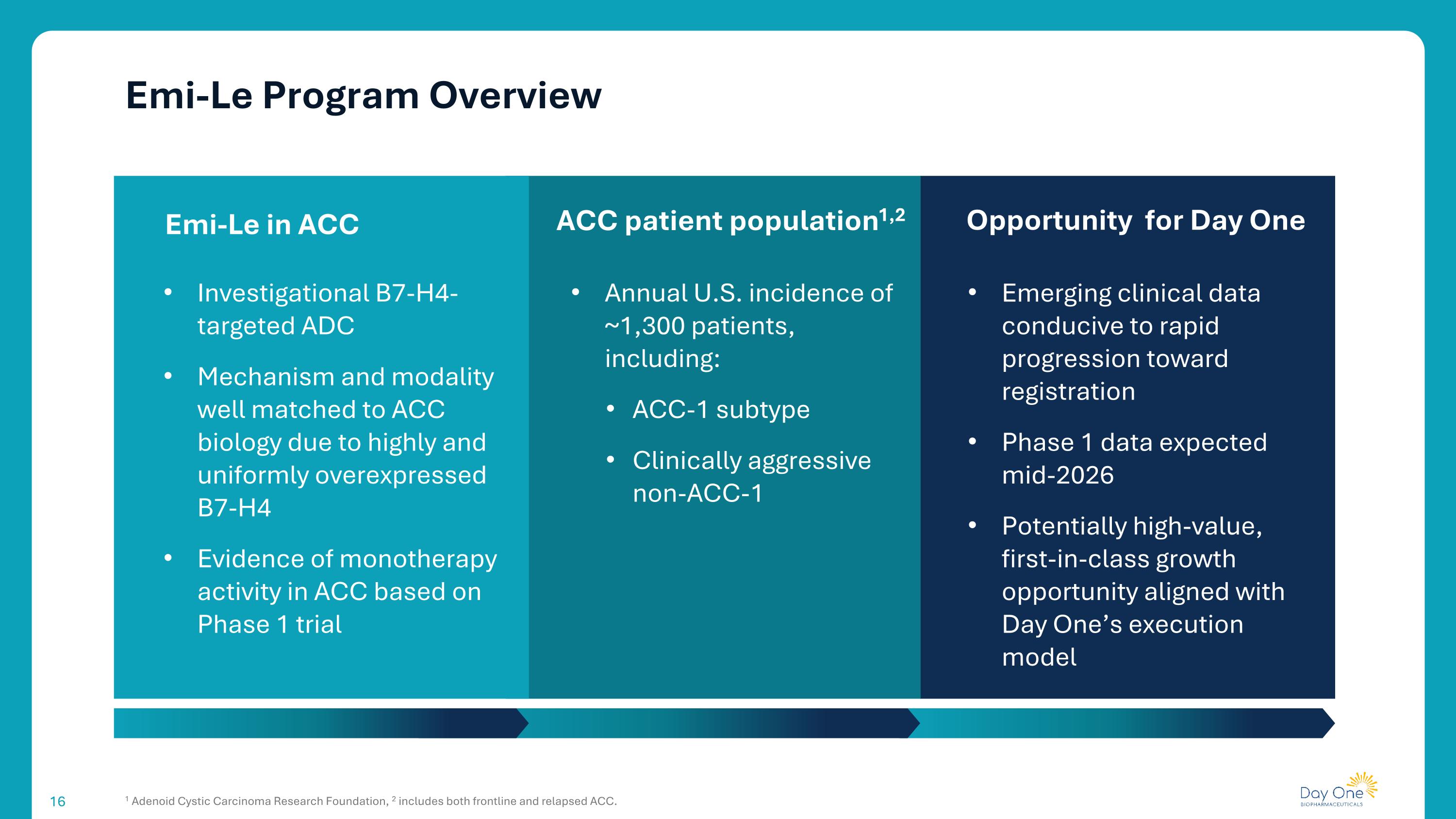
Emi-Le Program Overview 1 Adenoid Cystic Carcinoma Research Foundation, 2 includes both frontline and relapsed ACC. ACC patient population1,2 Investigational B7-H4-targeted ADC Mechanism and modality well matched to ACC biology due to highly and uniformly overexpressed B7-H4 Evidence of monotherapy activity in ACC based on Phase 1 trial Emi-Le in ACC Opportunity for Day One Annual U.S. incidence of ~1,300 patients, including: ACC-1 subtype Clinically aggressive non-ACC-1 Emerging clinical data conducive to rapid progression toward registration Phase 1 data expected mid-2026 Potentially high-value, first-in-class growth opportunity aligned with Day One’s execution model
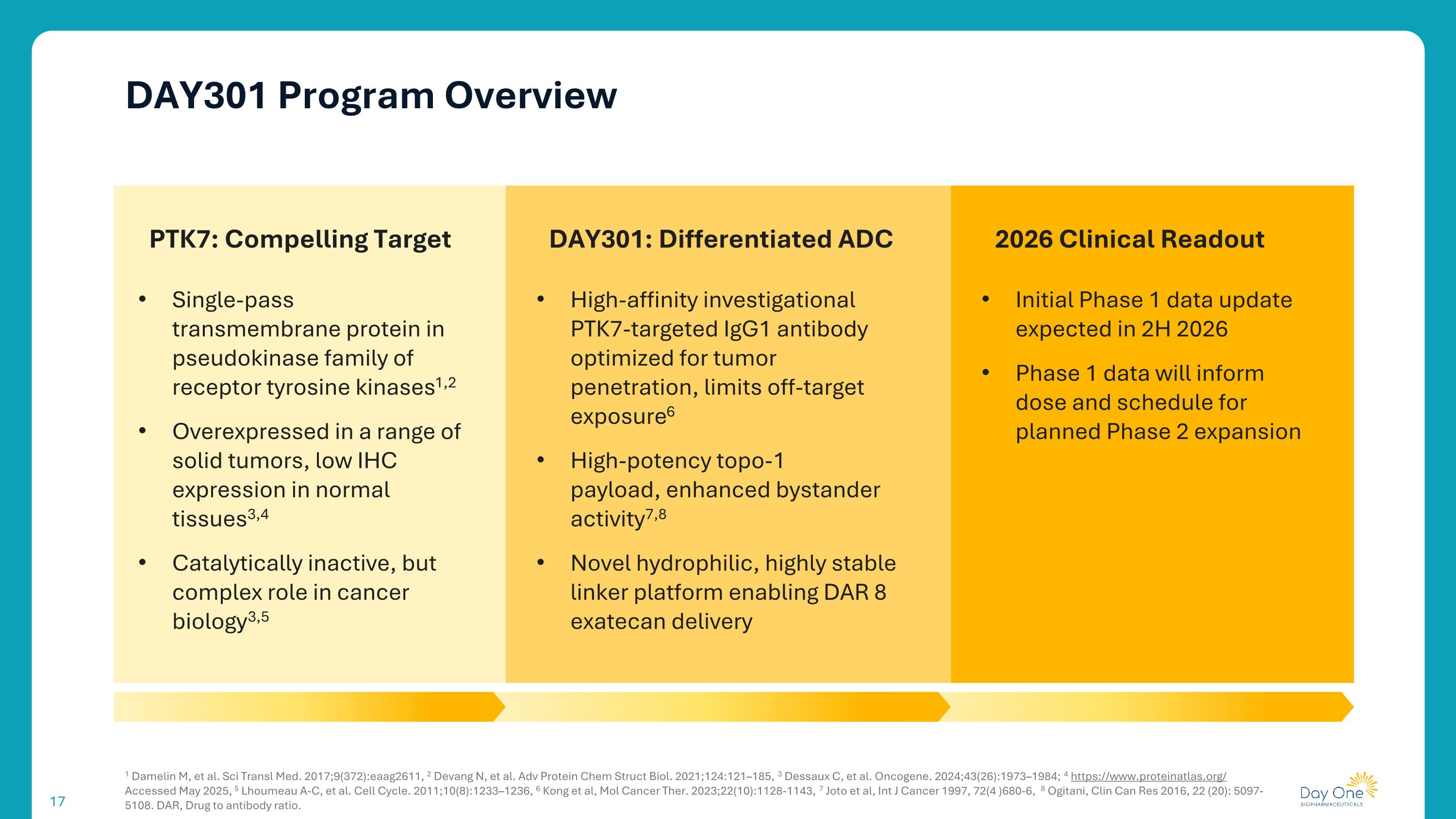
DAY301 Program Overview 1 Damelin M, et al. Sci Transl Med. 2017;9(372):eaag2611, 2 Devang N, et al. Adv Protein Chem Struct Biol. 2021;124:121–185, 3 Dessaux C, et al. Oncogene. 2024;43(26):1973–1984; 4 https://www.proteinatlas.org/ Accessed May 2025, 5 Lhoumeau A-C, et al. Cell Cycle. 2011;10(8):1233–1236, 6 Kong et al, Mol Cancer Ther. 2023;22(10):1128-1143, 7 Joto et al, Int J Cancer 1997, 72(4 )680-6, 8 Ogitani, Clin Can Res 2016, 22 (20): 5097-5108. DAR, Drug to antibody ratio. PTK7: Compelling Target Single-pass transmembrane protein in pseudokinase family of receptor tyrosine kinases1,2 Overexpressed in a range of solid tumors, low IHC expression in normal tissues3,4 Catalytically inactive, but complex role in cancer biology3,5 DAY301: Differentiated ADC 2026 Clinical Readout High-affinity investigational PTK7-targeted IgG1 antibody optimized for tumor penetration, limits off-target exposure6 High-potency topo-1 payload, enhanced bystander activity7,8 Novel hydrophilic, highly stable linker platform enabling DAR 8 exatecan delivery Initial Phase 1 data update expected in 2H 2026 Phase 1 data will inform dose and schedule for planned Phase 2 expansion

Financial Performance Charles York Chief Operating Officer & Chief Financial Officer
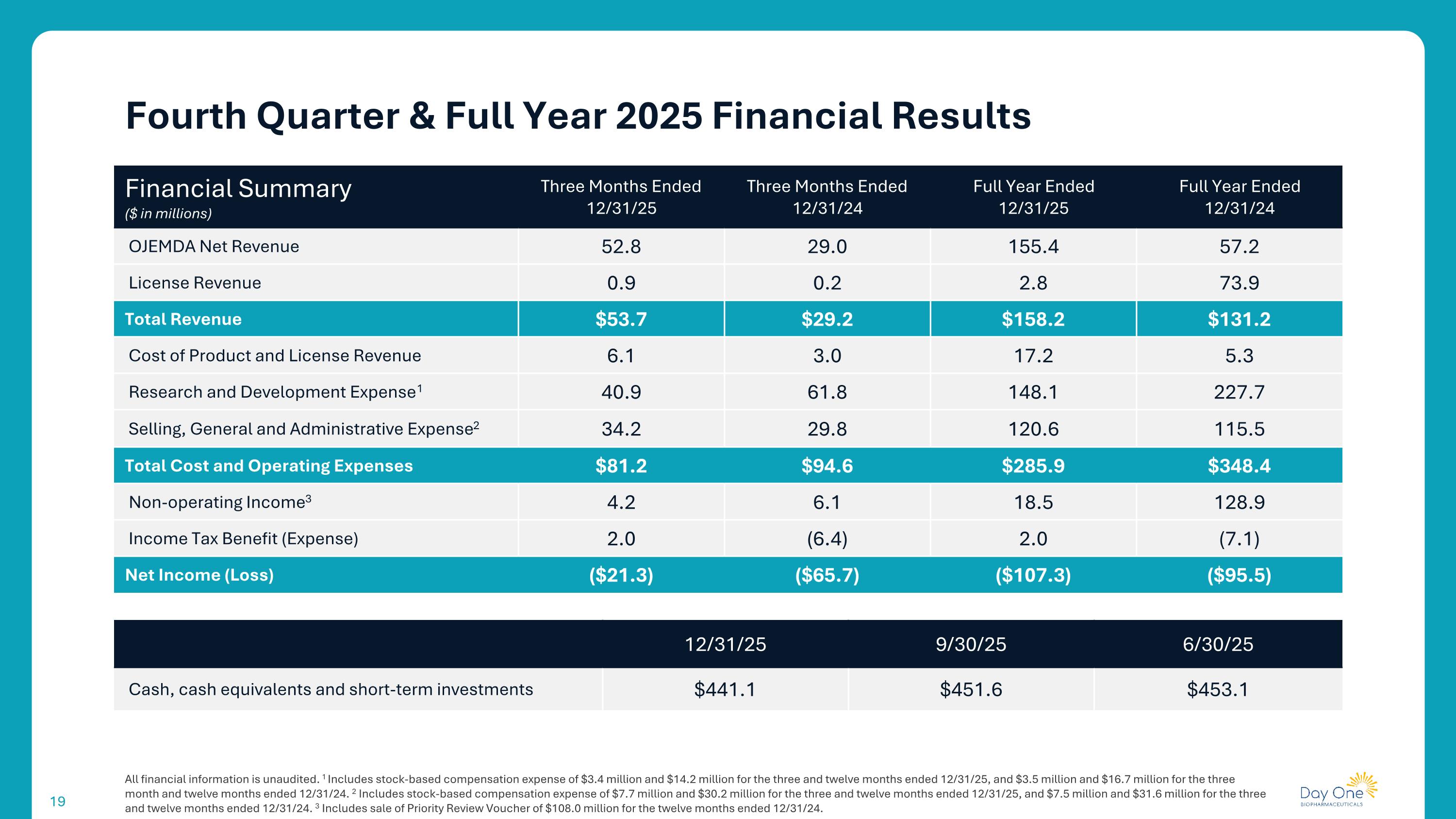
All financial information is unaudited. 1 Includes stock-based compensation expense of $3.4 million and $14.2 million for the three and twelve months ended 12/31/25, and $3.5 million and $16.7 million for the three month and twelve months ended 12/31/24. 2 Includes stock-based compensation expense of $7.7 million and $30.2 million for the three and twelve months ended 12/31/25, and $7.5 million and $31.6 million for the three and twelve months ended 12/31/24. 3 Includes sale of Priority Review Voucher of $108.0 million for the twelve months ended 12/31/24. Fourth Quarter & Full Year 2025 Financial Results Financial Summary ($ in millions) Three Months Ended 12/31/25 Three Months Ended 12/31/24 Full Year Ended 12/31/25 Full Year Ended 12/31/24 OJEMDA Net Revenue 52.8 29.0 155.4 57.2 License Revenue 0.9 0.2 2.8 73.9 Total Revenue $53.7 $29.2 $158.2 $131.2 Cost of Product and License Revenue 6.1 3.0 17.2 5.3 Research and Development Expense1 40.9 61.8 148.1 227.7 Selling, General and Administrative Expense2 34.2 29.8 120.6 115.5 Total Cost and Operating Expenses $81.2 $94.6 $285.9 $348.4 Non-operating Income3 4.2 6.1 18.5 128.9 Income Tax Benefit (Expense) 2.0 (6.4) 2.0 (7.1) Net Income (Loss) ($21.3) ($65.7) ($107.3) ($95.5) 12/31/25 9/30/25 6/30/25 Cash, cash equivalents and short-term investments $441.1 $451.6 $453.1

Thank You